Plant-parasitic nematodes on hemp in the Pacific Northwest of the United States
Abstract
Background:
Plant-parasitic nematodes are one of the most important biotic factors that impact crop production globally. Since hemp cultivation in the U.S. was banned from 1970 to 2018, little information is available about current plant-parasitic nematode pressure on U.S. hemp production. The production of hemp has gained interest in Washington and Oregon, states where several genera of plant-parasitic nematodes have been associated with various crops. This report is the first to define plant-parasitic nematodes associated with hemp in these states in the Pacific Northwest.
Methods:
Soil and root samples from hemp fields were collected in early autumn in 2021, 2022, and 2023. The occurrence, population density, and identity of plant-parasitic nematodes in these samples were determined using morphological and molecular identification methods. A Bayesian analysis of available sequence data was used to analyze phylogenetic relationships of nematode species found in hemp fields. Additionally, the host status of hemp ‘Alpha Explorer’ to three plant-parasitic nematodes, Meloidogyne chitwoodi, M. hapla, and Pratylenchus neglectus, was tested under greenhouse conditions. The occurrence of plant-parasitic nematodes and reproduction factor (final population density/initial population density) values of the three nematode species were analyzed with non-parametric methods.
Results:
Pratylenchus spp. were the most frequent plant-parasitic nematodes recovered from soil samples, being present in ~ 63% of samples (n = 107). Only two endoparasitic nematodes, Meloidogyne spp. and Pratylenchus spp., were found in root samples, with Pratylenchus spp. as the most frequent (20 out of 24 fields). A large diversity of Pratylenchus spp. was detected in hemp root samples. Hemp ‘Alpha Explorer’ was a poor host for P. neglectus, resulting in low reproduction values (< 1). Additionally, results of our study indicated that hemp is not a host for M. chitwoodi.
Conclusions:
Pratylenchus spp. were the most frequent plant-parasitic nematodes found in hemp fields in Oregon and Washington. This study reports for the first time five Pratylenchus species (Pratylenchus crenatus, P. fallax, P. hexincisus, P. neglectus, and P. scribneri) associated with hemp in Oregon and Washington; P. penetrans was also found in the region on hemp. The host status results indicate that hemp can be considered a non-host for M. chitwoodi and a poor host for M. hapla and P. neglectus.
Supplementary Information:
The online version contains supplementary material available at 10.1186/s42238-025-00301-y.
Article type: Research Article
Keywords: Hemp, Identification, Plant-parasitic nematodes
Affiliations: https://ror.org/00ysfqy60grid.4391.f0000 0001 2112 1969Department of Botany and Plant Pathology, Oregon State University, 2701 SW Campus Way, Corvallis, OR 97331 USA; https://ror.org/00qv2zm13grid.508980.cHorticultural Crops Disease and Pest Management Unit, USDA-ARS, 3420 Orchard Ave, Corvallis, OR 97330 USA; https://ror.org/00qv2zm13grid.508980.cForage Seed and Cereal Research Unit, USDA-ARS, 3450 SW Campus Way, Corvallis, OR 97331 USA
License: © This is a U.S. Government work and not under copyright protection in the US; foreign copyright protection may apply 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1186/s42238-025-00301-y | PubMed: 40671069 | PMC: PMC12265197
Relevance: Core topic: mentioned in title and keywords
Full text: PDF (1.5 MB)
Introduction
Hemp (Cannabis sativa), an herbaceous annual crop, is one of two crops from the Cannabaceae family cultivated in the Pacific Northwest (PNW; Oregon and Washington) of the United States (Punja ref. 2021). Hemp cultivation in the United States has been historically for fiber production. However, the 1937 Marihuana Tax Act classified all hemp crops as marijuana. Therefore, subject to the new drug enforcement laws with the Controlled Substance Act of 1970, hemp cultivation was banned, regardless of end use (Cranshaw et al. ref. 2019). It was not until the passage of the 2014 Farm Bill (U.S. H.R. 2642 – Agricultural Act of 2014 113th Congress [2013–2014]) that hemp was included in pilot research programs to reincorporate this crop into agriculture systems of the U.S. The 2018 Farm Bill removed hemp and hemp seeds from the Drug Enforcement Administration’s schedule of controlled substances, defining hemp as a hemp plant or any part of such plant with a maximum concentration of delta-9 tetrahydrocannabinol (THC) of 0.3% in terms of dry weight (Congress ref. 2018).
After the 2014 and 2018 Farm Bills, the production of hemp in the U.S. tripled from 10,406 ha in 2017 to 31,637 ha in 2018. In 2019, the hemp acreage reached a maximum peak of 81,393 ha (Vote, Hemp ref. 2021). After this production peak, hemp acreage decreased to a total of 11,458 ha in 2022. Hemp acreage in Oregon and Washington in 2022 was 850 ha and 152 ha, respectively (USDA-NASS ref. 2023). Although hemp is grown for different purposes in the PNW, the industry is dominated by outdoor floral hemp production for essential oil extraction valued at ~ $93 million (USDA-NASS ref. 2023). Because hemp has only been widely produced recently in the U.S., there is little information about the biotic challenges hemp growers face, including plant-parasitic nematodes (Bernard et al. ref. 2022). These organisms are estimated to cause more than $170 billion in economic losses to overall crop production globally per year (Kantor et al. ref. 2022).
A study conducted in Florida, a state with a subtropical climate, reported seven different genera of plant-parasitic nematodes, Nanidorus, Rotylenchulus, Helicotylenchus, ring nematode (Criconemella or Mesocriconema), Belonolaimus, Tylenchorhynchus, and Meloidogyne, associated with hemp (Desaeger et al. ref. 2023). Pratylenchus spp. have also been reported in hemp (Bernard et al. ref. 2022; Núnez-Rodríguez et al. ref. 2023), as well as Meloidogyne spp. (Nunez-Rodriguez et al. ref. 2024). Most of these nematodes are present in the PNW of the U.S. with Pratylenchus and Meloidogyne being as the most common genera (Zasada et al. ref. 2019). Despite the recent interest of hemp as a new crop in the region, no studies have been conducted to identify plant-parasitic nematodes in this crop or to evaluate the host status of hemp for plant-parasitic nematodes of concern. This study aimed to provide information on the presence of plant-parasitic nematodes in hemp fields in Oregon and Washington and determine the host status of hemp to three plant-parasitic nematodes commonly found in the PNW, Meloidogyne chitwoodi, M. hapla, and Pratylenchus neglectus.
Materials and methods
Sample collection
Composite soil samples from hemp fields were collected towards the end of the growing season (September) in 2021, 2022, and 2023, just before commercial harvest of the plants occurred in late September to early October. One or two 100-plant transects (~ 70 cm between plants) per field were selected for sampling based on field size, and 10 soil cores were collected per transect. In fields with one transect, one composite soil sample was collected, whereas two composite soil samples were collected in fields with two sampling transects. Soil sampling was performed using a 2.5-cm-diameter soil probe with a probe length of 30 cm (12” L soil sampler; JMC, Newton, IA). Cores were collected from every 10th plant in each transect and as close to the base of the plant crowns as possible. The 10 cores collected per transect were mixed in a bucket and then placed into a labeled plastic bag. In 2022 and 2023, root sampling was allowed by hemp growers. Roots were collected using a shovel. When possible, hemp roots were collected from the same plants near where soil samples were collected. When growers were concerned about potential damage caused by root sampling, only five plants out of the 10 were sampled. Samples were stored in a cooler during transport and then stored in at 4° C until processing.
Nematode extraction and enumeration
Soil samples (n = 45) collected in 2021 were extracted using a semi-automatic elutriator followed by sugar centrifugation with a 1 M sugar solution to recover vermiform nematodes from 250 g of wet soil (East et al. ref. 2019). At the time of processing, fine roots (n = 42, no roots were obtained from three fields) recovered on the upper sieve when elutriating were collected to extract nematodes using the Baermann funnel method (Ingham ref. 1994), with the main goal of generating preliminary information on the occurrence of plant-parasitic nematodes in hemp roots and to justify future requests to growers for hemp root collections. Soil samples collected in 2022 (n = 44) and 2023 (n = 18) were decant-sieved followed by sugar centrifugation with a 1 M sugar solution (Jenkins ref. 1964). Root samples from Washington (n = 7 collected in 2022) and Oregon (n = 10 and n = 7 collected in 2022 and 2023, respectively) were washed, cut into 1–2 cm long pieces, mixed, and placed under intermittent mist for nematode extraction for seven days (Zasada et al. ref. 2015). Plant-parasitic nematodes were enumerated and identified to the genus level based on morphological features using an inverted microscope. Frequency of occurrence of plant-parasitic nematodes in soil was analyzed for effects using χ2 analysis in RStudio v4.3.1 (R Core Team ref. 2023).
Molecular identification of plant-parasitic nematodes
Three Pratylenchus populations recovered from fine roots collected in 2021 were selected for molecular identification using species-specific primers from the β-1,4-endoglucanase gene region. Eight nematodes from each population were hand-picked and cut prior to DNA extraction (Peetz and Zasada ref. 2016); each nematode represented a soil sample. Fifteen Pratylenchus populations (eight and seven populations from Oregon and Washington, respectively) obtained from the 2022 hemp roots were identified via molecular techniques. The partial gene cytochrome oxidase I (COX1) was used for identification, which was amplified using the primers JB3 (5’- TTTTTTGGGCATCCTGAGGTTTAT − 3’)/JB4.5 (5’- TAAAGAAAGAACATAATGAAA ATG − 3’) (Bowles et al. ref. 1992), F7bp (GGDTGRACWTTHTAYCCNCC − 3’)/B4.5 (Bowles et al. ref. 1992; Ozbayrak et al. ref. 2019), or COIFGED (5’- CCTTTGGGCATCCNGARGTNTAT − 3’)/B5GED (5’- ACCTAAACTTARWACRTARTGAAAATG − 3’) (Bowles et al. ref. 1992; Ren et al. ref. 2024). Only one set of primers was used per population. Samples were sent for bidirectional sequencing to the Oregon State University Center for Quantitative Life Science (Corvallis, OR). Sequence identification was determined using the NCBI Basic Local Alignment Search Tool (BLAST).
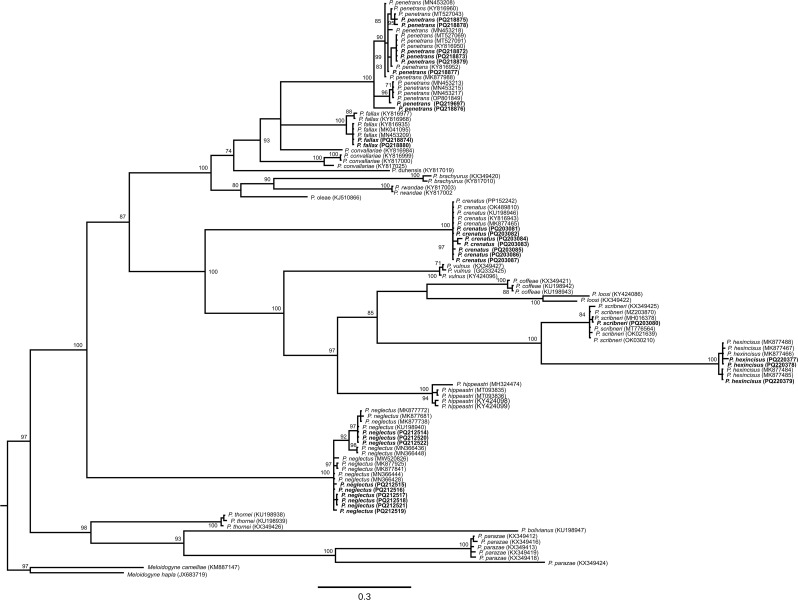
Reference sequences of Pratylenchus spp. were retrieved from GenBank to estimate phylogenetic relationships. The algorithm Clustal W was used for sequence alignment (Thompson et al. ref. 1994) and trimmed using BioEdit v.7.0.5.3 (Hall ref. 1999). jModelTest 2.1.10 v20160303 was used to select the best substitution model (Darriba et al. ref. 2012) to perform the phylogenetic analyses using a Bayesian analysis method (Larget and Simon ref. 1999). Finally, the phylogenetic tree was visualized using FigTree v1.4.3 (Rambaut ref. 2016). Sequences obtained in this study were deposited to GenBank (accession numbers PQ203080- PQ203087, PQ212514-, PQ212522, PQ218872- PQ218880, PQ219697, and PQ220377- PQ220379).
Host status of hemp for plant-parasitic nematodes
The pathogenicity of Meloidogyne hapla (reported in Núñez-Rodríguez et al. ref. 2024), M. chitwoodi and Pratylenchus neglectus were tested under greenhouse conditions in 2023. Briefly, eggs of Meloidogyne spp. were extracted from pure greenhouse cultures using the NaOCl method (Hussey and Baker ref. 1973), whereas P. neglectus was obtained from pure carrot disc cultures (Coyne et al. ref. 2014). The M. hapla population was originally collected from a vineyard in Alderdale, WA. The M. chitwoodi population was originally collected from a potato field in Prosser, WA. The P. neglectus population was originally collected from a wheat field in Pendelton, OR. All nematodes were molecularly identified to the species level by the North Carolina Department of Agriculture and Consumer Services based upon sequencing of the internal transcribed spacer (ITS) region (Raleigh, NC). Twenty 3-L plastic pots were filled with 2.1 kg of steam-pasteurized 1:1 sand/loam soil mixture. One five-week-old hemp ‘Alpha Explorer’ seedling was transplanted into each pot. Five pots per nematode species were inoculated with 4,000 eggs (M. chitwoodi and M. hapla) or 2,000 nematodes (mix of juveniles and adults of P. neglectus), and the five remaining pots were left non-inoculated. Three positive controls for each nematode species, tomato ‘Rutgers’ for Meloidogyne spp. and wheat ‘Yuma’ for P. neglectus, were planted in 0.6-L plastic pots filled with 0.4 kg of the soil mixture. Positive control tomato plants were inoculated with 1,100 Meloidogyne eggs per nematode species and positive control wheat plants were inoculated with 500 P. neglectus. The experimental design in this experiment was a completely randomized design. The average greenhouse temperature after inoculation was 20.8 ± 3.2°C. Plants were harvested 60 days after inoculation. Meloidogyne spp. were extracted from roots using the NaOCl method (Hussey and Baker ref. 1973), while P. neglectus was extracted from roots and soil using the intermittent mist and Baermann funnel methods, respectively, as described above. The reproduction factor (RF = final population density/initial population density) was calculated to determine the host status of hemp to these nematodes. The experiment was conducted twice.
The RF of each trial was analyzed separately since RF values of plant-parasitic nematodes on hemp did not meet the assumptions of normality (Shapiro test) and homoscedasticity (Levene test). Data was analyzed using the non-parametric Kruskal-Wallis and Mann-Whitney tests in RStudio v4.3.1 (R Core Team ref. 2023).
Results
Occurrence of plant-parasitic nematodes in hemp fields
A total of 107 soil samples (77 from Oregon and 30 from Washington) were processed, representing 66 hemp fields (50 from Oregon and 16 from Washington), from which seven different plant-parasitic nematodes were identified. The endoparasitic nematode Pratylenchus spp. were the most frequently detected nematodes, present in ~ 63% of the samples, followed by the ectoparasitic nematodes, Paratylenchus and Tylenchorhynchus (Table 1; P < 0.05). Four other plant-parasitic nematodes were detected at < 5% occurrence, including Criconematidae, Helicotylenchus, Meloidogyne, and Xiphinema. There were no statistical differences observed when the frequency of occurrence of plant-parasitic nematodes was analyzed by state or combined over both states (Supplementary Table 1; P > 0.05).
Table 1: Frequency of occurrence (FO%), maximum population density (Max.), and mean population density (nematodes/250 g of soil) of plant-parasitic nematodes when present in soil samples from hemp fields in the Pacific Northwest (Oregon and Washington) collected in September of 2021 and 2023
| Plant-parasitic nematodes | FO%* | Max. | Mean |
|---|---|---|---|
| Criconematidae | 3.7 a | 27 | 9 |
| Helicotylenchus | 3.7 a | 20 | 10 |
| Meloidogyne | 3.7 a | 26 | 20 |
| Paratylenchus | 22.4 b | 12,325 | 988 |
| Pratylenchus | 62.6 c | 228 | 338 |
| Tylenchorhynchus | 16.8 b | 320 | 37 |
| Xiphinema | 3.7 a | 27 | 15 |
*Frequency of occurrence data were analyzed for effects using χ2 analysis. Values followed by the same letter in the same column are not significantly different from each other (P < 0.05)
Pratylenchus spp. were the only endoparasitic nematodes obtained from the fine root samples collected in 2021, present in 30 out of the 42 samples (results not shown). In 2022, root samples were collected from 17 fields; 10 fields in Oregon and 7 fields in Washington. In Washington, Pratylenchus spp. were the only endoparasitic nematodes found in hemp root samples detected in all fields with population densities ranging from 1,195 to 13,362 nematodes in 100 g of wet root (Table 2). Two genera of endoparasitic nematodes were found in root samples collected in 2022 in Oregon, Pratylenchus spp. and Meloidogyne spp. Pratylenchus spp. were found in all 10 fields and three of these fields also contained Meloidogyne spp. The population densities of Pratylenchus and Meloidogyne ranged from 43 to 6,461 nematodes and 6 to 21,336 s-stage juveniles (J2) in 100 g of wet root, respectively (Table 2). Nematodes obtained in 2022 were selected for species identification using molecular methods.
Table 2: Population density of endoparasitic nematodes in roots expressed in 100 g of wet root obtained from hemp fields (n = 24)
| Field | Year | State | Pratylenchus spp. | Meloidogyne spp. |
|---|---|---|---|---|
| WA1 | 2022 | Washington | 1,195 | 0 |
| WA2 | 2022 | Washington | 5,071 | 0 |
| WA3 | 2022 | Washington | 2,085 | 0 |
| WA4 | 2022 | Washington | 13,362 | 0 |
| WA5 | 2022 | Washington | 12,886 | 0 |
| WA6 | 2022 | Washington | 5,040 | 0 |
| WA8 | 2022 | Washington | 5,446 | 0 |
| OR2 | 2022 | Oregon | 385 | 0 |
| OR3 | 2022 | Oregon | 532 | 24 |
| OR5 | 2022 | Oregon | 3,871 | 0 |
| OR6 | 2022 | Oregon | 43 | 0 |
| OR7 | 2022 | Oregon | 116 | 0 |
| OR9 | 2022 | Oregon | 128 | 0 |
| OR10 | 2022 | Oregon | 6,461 | 21,336 |
| OR11 | 2022 | Oregon | 3,813 | 0 |
| OR12 | 2022 | Oregon | 43 | 0 |
| OR18 | 2022 | Oregon | 841 | 6 |
| OR6 | 2023 | Oregon | 2,737 | 0 |
| OR10 | 2023 | Oregon | 2,107 | 296 |
| OR12 | 2023 | Oregon | 0 | 0 |
| OR15 | 2023 | Oregon | 0 | 0 |
| OR16 | 2023 | Oregon | 0 | 0 |
| OR18 | 2023 | Oregon | 498 | 0 |
| OR19 | 2023 | Oregon | 0 | 0 |
In 2023, Pratylenchus spp. and Meloidogyne spp. were the only endoparasitic nematodes found in the root samples collected in Oregon (Table 2). Pratylenchus spp. were detected in 3 of 7 fields with a range of population densities from 498 to 2,737 nematodes in 100 g of wet root. Meloidogyne spp. were detected in 1 of 6 fields with a population density of 296 J2 in 100 g of wet root.
Molecular identification of plant-parasitic nematodes
In 2021, three Pratylenchus spp. populations collected from Oregon were selected for molecular identification. These populations were identified as Pratylenchus penetrans (results not shown). In 2022, 15 populations of Pratylenchus spp. were molecularly identified, which included a P. penetrans population reported in Núñez-Rodríguez et al. (ref. 2023). In general, BLAST results ranged from 98.7 to 100% identity (Table 3). In Washington, four Pratylenchus spp. were identified: P. hexincisus (one sample out of seven), P. neglectus (five samples), P. penetrans (three samples), and P. scribneri (one sample). There were two samples with mixed Pratylenchus spp. One sample had three different Pratylenchus species (P. hexincisus, P. neglectus, and P. scribneri), whereas, the other sample contained two species (P. neglectus and P. penetrans). In Oregon, three Pratylenchus spp. were identified, P. crenatus (four out of eight samples), P. fallax (two samples), and P. penetrans (two samples) in root samples collected in 2022. No mixed Pratylenchus spp. populations were found in Oregon. Three samples had Meloidogyne spp. and one of these populations was identified as Meloidogyne hapla by Núñez-Rodríguez et al. (ref. 2024).
Table 3: Sequenced Pratylenchus spp. Populations recovered from hemp root samples collected in the Pacific Northwest (Oregon and Washington) including the top match percent identity based on the NCBI basic local alignment search tool
| PopulationCode | Pratylenchusspecies | Accession number | Match inGenBank | % Identity |
|---|---|---|---|---|
| WA1 | P. hexincisus | PQ220377 | MK877488 | 98.7 |
| WA1 | P. hexincisus | PQ220378 | MK877488 | 100 |
| WA1 | P. hexincisus | PQ220379 | MK877485 | 100 |
| WA1 | P. neglectus | PQ212514 | MK877772 | 99.8 |
| WA1 | P. scribneri | PQ203080 | MZ203870 | 99.8 |
| WA2 | P. neglectus | PQ212515 | MN366428 | 99.8 |
| WA2 | P. neglectus | PQ212516 | MN366428 | 100 |
| WA2 | P. neglectus | PQ212517 | MN366428 | 99.8 |
| WA3 | P. penetrans | PQ218872 | MN453208 | 99.3 |
| WA3 | P. neglectus | PQ212518 | MK877798 | 99.8 |
| WA3 | P. neglectus | PQ212519 | MN366444 | 99.8 |
| WA4 | P. neglectus | PQ212520 | MK877772 | 99.8 |
| WA4 | P. neglectus | PQ212521 | MN366428 | 99.8 |
| WA5 | P. penetrans | PQ218873 | MN453208 | 99.3 |
| WA5 | P. penetrans | PQ219697 | MN453217 | 99.8 |
| WA6 | P. neglectus | PQ212520 | MK877772 | 99.8 |
| WA8* | P. penetrans | OP801849 | MN453217 | 100 |
| OR5 | P. fallax | PQ218874 | MN453209 | 100 |
| OR6 | P. crenatus | PQ203081 | PP152242 | 99.8 |
| OR7 | P. crenatus | PQ203082 | PP152242 | 99.8 |
| OR9 | P. penetrans | PQ218875 | MN453208 | 99.5 |
| OR9 | P. penetrans | PQ218876 | KY816942 | 100 |
| OR9 | P. penetrans | PQ218877 | MN453208 | 99.8 |
| OR9 | P. penetrans | PQ218878 | MT527066 | 100 |
| OR10 | P. crenatus | PQ203083 | MK877465 | 99.5 |
| OR10 | P. crenatus | PQ203084 | PP152242 | 99.5 |
| OR10 | P. crenatus | PQ203085 | PP152242 | 99.8 |
| OR10 | P. crenatus | PQ203086 | PP152242 | 99.8 |
| OR11 | P. penetrans | PQ218879 | MK877988 | 99.1 |
| OR12 | P. crenatus | PQ203087 | MK877465 | 99.7 |
| OR18 | P. fallax | PQ218880 | MN453209 | 100 |
*Núnez-Rodríguez et al. (ref. 2023)
Pratylenchus penetrans was found relatively frequently in samples collected from both states. However, except for P. neglectus, the other Pratylenchus spp. were not recovered from both states (Table 3). The phylogenetic analysis placed the P. fallax and P. scribneri populations in clades with P. fallax and P. scribneri sequences with posterior probability (PP) values of 100 and 84, respectively. All P. fallax and P. penetrans populations identified in this study were placed within the Penetrans group (PP = 100; Fig. 1). Sequences of P. penetrans were placed in a clade with other P. penetrans sequences (PP = 100), with P. penetrans haplotypes forming subclades within this clade (Fig. 1). Populations of P. crenatus, P. hexincisus, and P. neglectus were also placed in clades with sequences of their respective species (PP = 100).
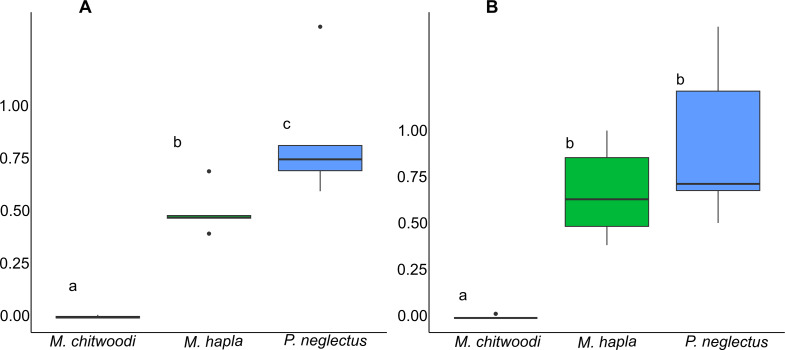
Host status of hemp for plant-parasitic nematodes.
Positive controls confirmed the viability of the nematode inoculum for both experiments. In these positive controls, the RF values (expressed as mean ± standard deviation) for M. hapla were 247.7 ± 17.7 and 169.4 ± 9.5 in the first and second trials, respectively. Meloidogyne chitwoodi had RF values of 118.9 ± 7.2 and 58.6 ± 10.7, whereas P. neglectus had RF values of 7.0 ± 3.5 and 8.4 ± 3.0 in the first and second trials, respectively. In both trials, M. chitwoodi had the lowest RF value (0.001 ± 0.006 and 0.004 ± 0.01 in first and second trials, respectively; P < 0.05). In the first trial, RF values for the three plant-parasitic nematodes were significantly different from each other (P = 0.002). However, in the second trial, RF values for M. hapla and P. neglectus were not statistically different (P > 0.05; Fig. 2). In both experiments, M. hapla and P. neglectus had RF values below 1 but not smaller than 0.5 (Fig. 2).
Discussion
In 2014, hemp cultivation was allowed in the U.S. after being banned since 1970 (Johnson ref. 2019). Therefore, there is limited information about factors that impact hemp production, including plant-parasitic nematodes. In the PNW, this is the first survey of plant-parasitic nematodes associated with hemp. Determining the occurrence and distribution of plant-parasitic nematodes is a crucial step towards determining the potential importance of these soilborne pathogens for crop production and future research efforts to manage nematodes (Zasada et al. ref. 2010). All seven genera of plant-parasitic nematodes identified in soil samples in this study have been reported in the PNW of the U.S. as affecting different crops (Zasada et al. ref. 2019). Pratylenchus was the most frequent genus recovered from both soil and hemp roots. This nematode is known to have a wide host range (> 2,000 plant species), including non-agricultural and economically important agricultural crops (Agrios ref. 2005; Núñez-Rodríguez et al. ref. 2022). This result is similar to what has been observed in the region across commodities, with Pratylenchus spp. being found in ~ 70% of samples considered in the PNW (Zasada et al. ref. 2019).
Although Meloidogyne spp. are considered the most important plant-parasitic nematodes worldwide (Jones et al. ref. 2013), Meloidogyne was not commonly recovered in this survey. This result is opposite to what has been reported by Desaeger et al. (ref. 2024) in Florida, where Meloidogyne spp. were the most frequently encountered plant-parasitic nematodes. This finding highlights the importance of region-specific sampling to assess the presence of plant-parasitic nematodes where little or no information is available.
Three samples from Oregon had both Meloidogyne and Pratylenchus, but one sample contained both nematodes with densities in the thousands: Meloidogyne sp. at 21,335 J2 and Pratylenchus sp. at 6,461 per 100 g of wet root (OR10 during 2022 in Table 2). The Meloidogyne population was identified as M. hapla (Núñez-Rodríguez et al. ref. 2024). Paratylenchus spp. and Tylenchorhynchus spp. are two ectoparasitic nematodes that are commonly found in the PNW (Zasada et al. ref. 2019). Both of these plant-parasitic nematodes were also commonly found in our study. There was a sample with an average of 12,325 nematodes per 250 g of soil (~ 149 Paratylechus spp. per g of soil). On pea, Upadhaya et al. (ref. 2019) demonstrated that an initial population of 4.5 P. nanus/g of soil caused a reduction of plant height, dry shoot weight, and seed weight. The population density we observed on hemp was 10 times higher than the initial population reported by Upadhaya et al. (ref. 2019).
The low occurrence of Criconematidae, Helicotylenchus spp., and Xiphinema spp. is in agreement with what has been reported in the PNW (Zasada et al. ref. 2019). However, the low occurrence of these plant-parasitic nematodes does not mean that they do not pose a risk for hemp production. For example, Xiphinema spp. may be of concern for hemp growers due to the ability of this group of nematodes to transmit viruses that have been reported in hemp, such as Arabis mosaic virus (Harrison et al. ref. 1974; McPartland ref. 2000).
The partial COX1 gene allowed for the identification of all 15 Pratylenchus spp. populations selected for this purpose, including the population found in OR10, which was identified as P. penetrans (Núñez-Rodríguez et al. ref. 2024). The phylogenetic tree obtained in this study had a similar topology to the one reported in Janssen et al. (ref. 2017), which demonstrated the importance for this region to separate between closely related species such as P. penetrans and P. fallax.
The most frequent Pratylenchus spp. identified from hemp roots was P. neglectus, which was found in 10 samples (one sample from Oregon and nine from Washington), and P. penetrans in eight samples (five and three samples from Oregon and Washington, respectively). These species are two of the most import Pratylenchus species worldwide (Jones et al. ref. 2013), including in the PNW region, where they and P. crenatus (another species identified in this study) can commonly be found associated with various crops (Zasada et al. ref. 2019). Pratylenchus fallax, P. hexincisus, and P. scribneri were also identified in this study. This is the first time P. fallax, P. scribneri, P. crenatus, and P. neglectus have been reported on hemp in the PNW (Zasada et al. ref. 2018). To our knowledge, this is the first time that hemp is reported to be a host for these nematodes. This is also the first report of P. hexincisus in the PNW or on hemp (Zasada et al. ref. 2018).
The pathogenicity of P. neglectus, M. hapla (Núñez-Rodríguez et al. ref. 2024)d chitwoodi (Zasada et al. ref. 2019), the latter being a nematode of concern for potato growers in the PNW, was tested under greenhouse conditions. Hemp was a non-host for M. chitwoodi with RF values close to 0, whereas hemp was a poor host for M. hapla and P. neglectus. The poor host status of hemp for M. hapla was also observed by Coburn et al. (ref. 2024). However, reproduction of M. hapla has been reported to vary between hemp cultivars, from moderately susceptible to highly resistant (de Meijer ref. 1993). In the case of M. chitwoodi, although not with the same hemp cultivar, our results agree with Kock et al. (1994), who reported hemp as non-host for this nematode.
This is the first research effort defining the occurrence, identity, and interaction of plant-parasitic nematodes on hemp in Oregon and Washington. Our study determined that Pratylenchus spp. were the most frequent plant-parasitic nematodes found across all hemp fields sampled. Additionally, five Pratylenchus species were found in hemp fields in the region. Based upon the host status of only one hemp cultivar, hemp seems to be a promising crop to consider for crop rotation systems in fields where M. chitwoodi is of concern, since it appears to be a non-host. Further studies should be conducted in microplot or field environments to evaluate the potential role of hemp to reduce population densities of Meloidogyne spp. and Pratylenchus spp. in the PNW.
Supplementary Materials
References
- Agrios. Plant pathology, 2005
- EC Bernard, AG Chaffin, KD Gwinn. Review of nematode interactions with hemp (Cannabis sativa). J Nematol, 2022. [DOI | PubMed]
- J Bowles, D Blair, DP McManus. Genetic variants within the genus Echinococcus identified by mitochondrial DNA sequencing. Mol Biochem Parasitol, 1992. [DOI | PubMed]
- Coburn J, Desaeger J. Host status and susceptibility of Cannabis sativa cultivars to root-knot nematodes. J Nematol. 2024;56. 10.2478/jofnem-2024-0003. e2024-1.
- Congress US. S.2667-Hemp Farming Act of. 2018. S.2667–115th Congress (2017–2018). https://www.congress.gov/bill/115th-congress/house-bill/1
- Coyne DL, Adewuyi O, Mbiru E. Protocol for in vitro culturing of lesion nematodes: Radopholus similis and Pratylenchus spp. on carrot discs. International Institute of Tropical Agriculture, Ibadan, Nigeria. 2014. https://cgspace.cgiar.org/server/api/core/bitstreams/ba6eaad6-6677-4313-9839-7914b5568b64/content
- W Cranshaw, M Schreiner, K Britt, KP Kuhar, J McPartland, J Grant. Developing insect pest management systems for hemp in the united states: A work in progress. J Integr Pest Manag, 2019. [DOI]
- D Darriba, GL Taboada, R Doallo, D Posada. jModelTest 2: more models, new heuristics and parallel computing. Nat Meth, 2012. [DOI]
- de Meijer. Evaluation and verification of resistance to Meloidogyne hapla Chitwood in a Cannabis germplasm collection. Euphytica, 1993. [DOI]
- J Desaeger, J Coburn, J Freeman, Z Brym. Plant-parasitic nematodes associated with Cannabis sativa in Florida. J Nematol, 2023. [DOI | PubMed]
- KE East, IA Zasada, RP Schreiner, MM Moyer. Developmental dynamics of Meloidogyne hapla in Washington wine grapes. Plant Dis, 2019. [DOI | PubMed]
- Hall. BioEdit: A user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucleic Acids Symp, 1999
- B Harrison, W Robertson, C Taylor. Specificity of retention and transmission of viruses by nematodes. J Nematol, 1974. [PubMed]
- R Hussey, KR Barker. A comparison of methods of collecting inocula of Meloidogyne spp. Including a new technique. Plant Dis Rptr, 1973
- RE Ingham. Nematodes. Methods of soil analysis: part 2, Microbiological and biochemical properties, 1994
- T Janssen, G Karssen, V Orlando, S Subbotin, W Bert. Molecular characterization and species delimiting of plant-parasitic nematodes of the genus Pratylenchus from the penetrans group (Nematoda: Pratylenchidae). Mol Phylogenet Evol, 2017. [DOI | PubMed]
- WR Jenkins. A rapid centrifugal-flotation technique for separating nematodes from soil. Plant Dis Rptr, 1964
- R Johnson. Defining hemp: a fact sheet, 2019
- J Jones, A Haegeman, E Danchin, H Gaur, J Helder, M Jones. Review: top 10 plant-parasitic nematodes in molecular plant pathology. Mol Plant Pathol, 2013. [DOI | PubMed]
- M Kantor, Z Handoo, C Kantor, L Carta. Top ten most important U.S.-regulated and emerging plant-parasitic nematodes. Horticulturae, 2022. [DOI]
- B Larget, D Simon. Markov chain Monte Carlo algorithms for the bayesian analysis of phylogenetic trees. Mol Biol Evol, 1999. [DOI]
- JM McPartland, RC Clarke, DP Watson. Hemp diseases and pests: management and biological control, 2000
- LA Núñez-Rodríguez, DA Humphreys-Pereira, AM Rodríguez-Ruiz, L Flores-Chaves. Plant-parasitic nematodes associated with weeds in potato (Solanum tuberosum L.) fields from the Northern area of cartago, Costa Rica. Nematropica, 2022
- LA Núñez-Rodríguez, HM Rivedal, A Peetz, C Ocamb, I Zasada. First report of the root-lesion nematode, Pratylenchus penetrans, parasitizing hemp (Cannabis sativa) in the united States. Plant Health Prog, 2023. [DOI]
- L Núñez-Rodríguez, H Rivedal, A Peetz, CM Ocamb, I Zasada. First report of Meloidogyne hapla on hemp (Cannabis sativa) in Oregon. J Nematol, 2024. [DOI | PubMed]
- M Ozbayrak, TC Todd, T Harris, RS Higgins, KS Powers, PG Mullin. A COI DNA barcoding survey of Pratylenchus species in the great plains region of North America. J Nematol, 2019. [DOI]
- AB Peetz, IA Zasada. Species-specific diagnostics using a β-1,4-endoglucanase gene for Pratylenchus spp. Occurring Pac Northwest North Am Nematology, 2016. [DOI]
- Press USDA-NASS. Release. 2023;https://www.nass.usda.gov/Statistics_by_State/Idaho/Publications/Crops_Press_Releases/2023/HEMP.pdf
- Punja ZK. Emerging disease of Cannibis sativus and sustainable management. Pest Manag Sci. 2021;77:3857–3870. 10.1002/ps.6307.
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. 2023; https://www.R-project.org/
- Rambaut A. FigTree v1.4.3: Tree Figure Drawing Tool. 2016; http://tree.bio.ed.ac.uk/software/figtree/
- Y Ren, DL Porazinska, Q Ma, S Liu, H Li, X Qing. A single degenerated primer significantly improves COX1 barcoding performance in soil nematode community profiling. Soil Ecol Lett, 2024. [DOI]
- Thompson JD, Higgins DG, Gibson TJ. CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position speci ic gap penalties and weight matrix choice. Nuc Acids Res. 1994;22:4673–4680. 10.1093/nar/22.22.4673.
- A Upadhaya, G Yan, J Pasche. Reproduction ability and growth effect of pin nematode, Paratylenchus nanus, with selected field pea cultivars. Plant Dis, 2019. [DOI | PubMed]
- Vote, Hemp. U.S. hemp crop report. 2021;https://www.votehemp.com/u-s-hemp-crop-report/
- IA Zasada, JN Pinkerton, TA Forge. Plant-parasitic nematodes associated with highbush blueberries (Vaccinium corymbosum) in the Pacific Northwest of North America. Int J Fruit Sci, 2010. [DOI]
- IA Zasada, JE Weiland, Z Han, TW Walters, P Moore. Impact of Pratylenchus penetrans on establishment of red raspberry. Plant Dis, 2015. [DOI | PubMed]
- IA Zasada, LM Dandurand, C Gleason, CH Hagerty, RE Ingham. Plant parasitic nematodes of the Pacific northwest: idaho, Oregon and Washington. Plant-parasitic nematodes in sustainable agriculture of North america, volume 2: Western USDA and Mexico, 2018
- IA Zasada, M Kitner, C Wram, N Wade, RE Ingham, S Hafez. Trends in occurrence, distribution, and population densities of plant-parasitic nematodes in the Pacific Northwest of the united States from 2012 to 2016. Plant Health Prog, 2019. [DOI]
