Safety of Drugs Used in Difficult-to-Treat Epileptic Syndromes: A Disproportionality Analysis Using the Eudravigilance Database
Abstract
Background/Objectives: Difficult-to-treat epileptic syndromes include conditions typically emerging in the first years of life and are characterized by a high rate of drug refractoriness. This study aimed to better define the safety profile of drugs used as adjunctive therapies for seizures associated with these syndromes using real-world pharmacovigilance data. Methods: We retrospectively analyzed the publicly available data regarding Individual Case Safety Reports (ICSRs), presenting stiripentol, cannabidiol, or fenfluramine as suspected drugs, reported on the Eudravigilance database until the third quarter of 2024. Data were evaluated with descriptive analyses and then with disproportionality measures, including the reporting odds ratio. Results: A total of 5986 ICSRs met the inclusion criteria (71.6% from cannabidiol, 14.5% fenfluramine, and 13.9% stiripentol). Significantly higher probabilities of reporting Cardiac disorders, Vascular disorders, and Respiratory, thoracic, and mediastinal disorders were observed with fenfluramine. Cannabidiol was associated with Product issues, whereas stiripentol was associated with injury, poisoning, procedural complications, Metabolism and nutrition disorders, and Blood and lymphatic system disorders. Conclusions: Our analysis did not highlight new and unexpected serious safety signals but confirmed the need to strictly monitor patients for the risk of adverse events. However, further prospective studies are required to better clarify the safety profile of these drugs in order to optimize their use.
Article type: Research Article
Keywords: epileptic syndromes, stiripentol, cannabidiol, fenfluramine, adverse events, eudravigilance, disproportionality analysis
Affiliations: Department of Public Health, University of Naples Federico II, 80131 Naples, Italy; ariannascala7@gmail.com (A.S.); teresa.trunfio@gmail.com (T.A.T.); giovanni.improta@unina.it (G.I.); mariodamiano.toro@unina.it (M.D.T.); Department of Medicine and Surgery, University of Enna “Kore”, 94100 Enna, Italy; chiara.pennisi@unikore.it (C.P.); giorgia.f1d@gmail.com (G.F.); adriana.graziano@unikore.it (A.C.E.G.); marilena.briglia@unikore.it (M.B.); fabio.allia@unikore.it (F.A.); giovanni.giurdanella@unikore.it (G.G.); roberta.malaguarnera@unikore.it (R.M.); rosalia.battaglia@unikore.it (R.B.); giovanniluca.romano@unikore.it (G.L.R.); Department of Biomedical and Biotechnological Sciences, School of Medicine, University of Catania, 95123 Catania, Italy; vmicale@unict.it (V.M.); serena.dm.92@gmail.com (S.D.M.); prof.filippodrago@gmail.com (F.D.); Radiology Unit, Humanitas Istituto Clinico Catanese, 95045 Catania, Italy; ceciliagozzo91@gmail.com; Division of Hematology, A.O.U. Policlinico “G.Rodolico—S. Marco”, 95125 Catania, Italy; fannypalumbo@gmail.com; Hematology and Bone Marrow Transplantation Unit, Hospital of Bolzano (SABES-Azienda Sanitaria dell’Alto Adige), Teaching Hospital of Paracelsus Medical University, 39100 Bolzano, Italy; gerovetro@gmail.com; Department of Special Surgery Ophthalmology Department, University of Jordan, Amman 11942, Jordan; Clinical Trial Unit, University Hospital of Catania, 95125 Catania, Italy
License: © 2025 by the authors. CC BY 4.0 Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.3390/ph18121895 | PubMed: 41471384 | PMC: PMC12736221
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (479 KB)
1. Introduction
Epileptic encephalopathies are severe forms of epilepsy which typically occur early in infancy and result in reduced cognitive function [ref. 1,ref. 2,ref. 3,ref. 4]. These disorders can be part of epileptic syndromes and can be characterized by generalized or recurrent focal seizures and being severe and usually refractory to standard antiepileptic drugs (AEDs), including benzodiazepines, phenobarbital, sodium valproate, lamotrigine, and topiramate.
Dravet syndrome (DS) and Lennox–Gastaut syndrome (LGS) are two rare but very severe epileptic syndromes [ref. 5]. DS, also known as severe myoclonic epilepsy of infancy (SMEI), is a genetic condition caused frequently by the loss-of-function of SCN1A variants, which typically develops in the first year of a life with frequent, prolonged seizures commonly triggered by hyperthermia [ref. 6,ref. 7,ref. 8]. LGS is a severe form of epilepsy, characterized by multiple types of seizures (tonic axial, atonic, absence, myoclonic, and generalized tonic–clonic seizures) and intellectual disability [ref. 9]. It can be associated with encephalitis, meningitis, tuberous sclerosis, brain malformations, and brain injury. Even in this case, treatment with AEDs is often ineffective.
Patients with DS and LGS are usually in polypharmacy, treated with a median of three AEDs [ref. 10,ref. 11,ref. 12,ref. 13,ref. 14,ref. 15].
The use of these drugs should be optimized to reduce the burden of seizures, as well as to minimize adverse events (AEs).
Valproate (VPA) is recognized as the first-line medication [ref. 11,ref. 16]; for subsequent lines, various AEDs have been approved as adjunctive therapies.
Among the pharmacological agents approved in the European Union as adjunctive therapy for the management of pediatric epilepsy-related seizures, in particular for DS, we can find stiripentol, cannabidiol, and fenfluramine.
In 2007, stiripentol (Diacomit®) received approval in Europe as an orphan drug for managing bilateral tonic–clonic seizures (BTCS) in children with DS [ref. 17]. It is intended for use as an add-on therapy alongside clobazam (CLB) and VPA, in cases where these drugs are not sufficient to control seizures [ref. 18,ref. 19]. Several mechanisms of action have been proposed for stiripentol, including enhanced GABAergic neurotransmission, inhibition of lactate dehydrogenase, reduction in calcium-mediated toxicity in hippocampal neurons with NMDA receptors, and the inhibition of calcium channels [ref. 20]. Several studies have highlighted that stiripentol is a generally well-tolerated and effective medication for reducing the frequency and duration of epileptic seizures [ref. 21,ref. 22], with certain AEs occurring more frequently, such as weight loss, decreased appetite, and drowsiness [ref. 23,ref. 24].
Cannabidiol, along with Δ9-tetrahydrocannabinol (THC), is one of the bioactive compounds found in the Cannabis plant [ref. 25]. Unlike THC, cannabidiol is not a psychoactive substance and exhibits anti-inflammatory, neuroprotective, and antioxidant properties [ref. 26]. Although the exact anticonvulsant mechanism of action has not yet been fully elucidated, cannabidiol has proven effective in suppressing seizures in animal models of epilepsy [ref. 26]. It has been proposed that cannabidiol may modulate neuronal excitability by interacting with various targets involved in the functional regulation of neuronal excitability, such as the transient receptor potential vanilloid 1 (TRPV1), the equilibrative nucleoside transporter 1 (ENT1), the orphan G protein-coupled receptor 55 (GPR55) [ref. 27], the voltage-gated sodium channels [ref. 28], the dopamine D2/D3 receptors [ref. 29,ref. 30], and the cannabinoid CB1 receptors [ref. 31,ref. 32,ref. 33]. Cannabidiol has been approved in the European Union under the name Epidyolex® (GW Pharma [International] B.V.), in combination with CLB, for the treatment of seizures associated with LGS and DS in patients aged two years and older [ref. 34]. Additionally, in Europe, cannabidiol is authorized as an adjunct therapy for seizures associated with the tuberous sclerosis complex (TSC) in patients over the age of two [ref. 35]. The primary AEs associated with cannabidiol use include drowsiness, diarrhea, vomiting, and pyrexia [ref. 36,ref. 37,ref. 38,ref. 39].
Fenfluramine (Fintepla®) is an anticonvulsant drug with a dual mechanism of action, functioning both as an agonist of the serotonergic system and a positive allosteric modulator of sigma-1 receptors (σ1-Rs) [ref. 40]. It has been suggested that its interaction with serotonin (5-HT) receptors enhances inhibitory transmission mediated by γ-aminobutyric acid (GABA), while the activation of σ1-Rs is predominantly associated with a reduction in excitatory glutamatergic signaling [ref. 40]. Previously, the drug was used at high doses as an appetite suppressant in adults with obesity, but its marketing was discontinued due to an increased risk of serious cardiovascular events, valvular heart disease (VHD), and pulmonary arterial hypertension (PAH). More recently, it has been approved in Europe at significantly lower doses for the treatment of seizures associated with DS and LGS, as an adjunctive therapy to other anticonvulsants in patients older than 2 years [ref. 41]. Fenfluramine exhibits a favorable tolerability profile, with the most commonly reported AEs across multiple studies being reduced appetite, fatigue, and somnolence [ref. 40,ref. 42,ref. 43].
Although the tolerability of these drugs has been assessed in several clinical studies, post-marketing surveillance is essential to ensure the safe use of these medications. Indeed, post-marketing studies on pharmacovigilance databases potentially enable the identification of rare or severe AEs, which may not have emerged during the drug development phase due to time constraints or limited study populations [ref. 44].
The aim of this study was to analyze data from the Eudravigilance database to comprehensively assess the safety profile of these drugs.
2. Results
Our analysis showed a high prevalence of ICSRs from cannabidiol, followed by fenfluramine and stiripentol. Significantly higher probabilities of reporting Cardiac disorders, Vascular disorders, and Respiratory, thoracic, and mediastinal disorders were observed with fenfluramine. Cannabidiol was associated with Product issues, whereas stiripentol was associated with Injury, poisoning, procedural complications, Metabolism and nutrition disorders, and Blood and lymphatic system disorders.
Overall, 5896 ICSRs related to stiripentol (n = 822; 13.9%), cannabidiol (n = 4222; 71.6%), and fenfluramine (n = 852; 14.5%) were retrieved from the Eudravigilance database for the reference period (Figure 1). The number of cases reported increased year by year, with the highest number of reports observed in 2021 for cannabidiol (n = 1065; 25.2%), and in 2024 for fenfluramine (n = 213; 25%) and stiripentol (n = 183; 22.3%).
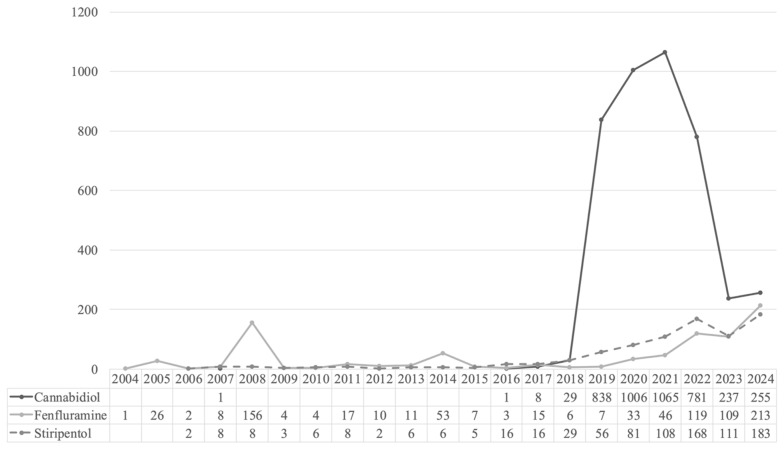
Table 1 summarizes the main characteristics of the ICSRs for each drug.
Table 1: Characteristics of Individual Case Safety Reports (ICSRs).
| Stiripentol | Cannabidiol | Fenfluramine | |
|---|---|---|---|
| Age group | 822 (100) | 4222 (100) | 852 (100) |
| Not Specified | 226 (27.49) | 581 (13.76) | 216 (25.35) |
| 0–1 Month | 0 (0) | 2 (0.05) | 2 (0.23) |
| 2 Months–2 Years | 94 (11.44) | 157 (3.72) | 28 (3.29) |
| 3–11 Years | 262 (31.87) | 991 (23.47) | 130 (15.26) |
| 12–17 Years | 113 (13.75) | 661 (15.66) | 92 (10.8) |
| 18–64 Years | 126 (15.33) | 1732 (41.02) | 356 (41.78) |
| 65–85 Years | 1 (0.12) | 93 (2.2) | 27 (3.17) |
| More than 85 Years | 0 (0) | 5 (0.12) | 1 (0.12) |
| Sex | 822 (100) | 4222 (100) | 852 (100) |
| Female | 383 (46.59) | 1948 (46.14) | 506 (59.39) |
| Male | 423 (51.46) | 2149 (50.9) | 243 (28.52) |
| Not Specified | 16 (1.95) | 125 (2.96) | 103 (12.09) |
| Reporter group | 822 (100) | 4222 (100) | |
| Healthcare Professional | 444 (54.01) | 3974 (94.13) | 692 (81.22) |
| Non-Healthcare Professional | 378 (45.99) | 248 (5.87) | 121 (14.2) |
| Not Specified | 0 (0) | 0 (0) | 39 (4.58) |
| EEA/Non-EEA | 822 (100) | 4222 (100) | 852 (100) |
| European Economic Area | 229 (27.86) | 865 (20.49) | 340 (39.91) |
| Non-European Economic Area | 593 (72.14) | 3357 (79.51) | 512 (60.09) |
| Country | |||
| France | 66 (8.03) | 416 (9.85) | 94 (11.03) |
| Germany | 57 (6.93) | 116 (2.75) | 132 (15.49) |
| UK | 16 (1.95) | 101 (2.39) | 0 |
| Italy | 28 (3.41) | 64 (1.52) | 24 (2.82) |
| Spain | 30 (3.65) | 50 (1.18) | 17 (2) |
| Netherlands | 0 | 32 (0.76) | 0 |
| Austria | 0 | 15 (0.36) | 0 |
| Others | 32 (3.89) | 71 (1.68) | 61 (7.16) |
| Seriousness | 822 (100) | 4222 (100) | 852 (100) |
| Non-Serious | 93 (11.3) | 603 (14.3) | 137 (16.1) |
| Serious | 728 (88.6) | 3619 (85.7) | 713 (83.7) |
| Not Specified | 1 (0.1) | 0 (0) | 2 (0.2) |
| Outcome | 2220 (100) | 8823 (100) | 2081 (100) |
| Fatal | 42 (1.89) | 503 (5.7) | 67 (3.22) |
| Not Recovered/Not Resolved | 138 (6.22) | 1169 (13.25) | 278 (13.36) |
| Not Specified | 0 (0) | 0 (0) | 37 (1.78) |
| Recovered/Resolved | 300 (13.51) | 1423 (16.13) | 320 (15.38) |
| Recovered/Resolved with Sequelae | 6 (0.27) | 16 (0.18) | 19 (0.91) |
| Recovering/Resolving | 134 (6.04) | 455 (5.16) | 93 (4.47) |
| Unknown | 1600 (72.07) | 5257 (59.58) | 1267 (60.88) |
The incidence of events in males accounted for a larger proportion than females, except for fenfluramine (stiripentol n = 423; 51.46%; cannabidiol n = 2149; 50.9%; fenfluramine n = 243; 28.52%; p < 0.001). Adult patients (≥18 years old) were more represented than the pediatric population (<18 years old), except for stiripentol.
In terms of seriousness, more than 80% of ICSRs indicated at least one ADR classifiable as serious, although less frequently in the fenfluramine group (stiripentol n = 728, 88.6%; cannabidiol n = 3619, 85.7%; fenfluramine n = 713, 83.7%; p = 0.015). The outcome recovered/resolved was reported for 2043 cases, more frequently for cannabidiol (n = 1423, 16.1%; p = 0.009), whereas a fatal event was reported for 612 cases, more frequently for cannabidiol (n = 503, 5.7%; p = 0.001). However, outcome data were available in less than 50% of ICSRs (stiripentol n = 620, 27.9%; cannabidiol n = 3566, 40.4%; fenfluramine n = 814, 39.1%).
More than 80% of ADRs were reported by healthcare professionals, apart from stiripentol (stiripentol n = 444, 54.01%; cannabidiol n = 3974, 94.13%; fenfluramine n = 692, 81.22%). Unfortunately, further information about the sender is not available. ICSRs are reported according to the sender, identified only as a healthcare or non-healthcare professional.
The largest number of AEs related to stiripentol and cannabidiol came from France (8.03% and 9.85, respectively), whereas in Germany, ICSRs associated with fenfluramine were reported more frequently (15.49%).
“Nervous system disorders” was the most represented reaction group (stiripentol n = 563; 26.6%; cannabidiol n = 2292; 27.1%; fenfluramine n = 368; 19.1%; Table 2).
Table 2: Individual Case Safety Reports (ICSRs) stratified by system organ class (SOC). The number of reports with at least one adverse drug reaction (ADR) related to the SOCs are reported. The sum of the events by SOC (%) is higher than the total number of reports, since a single ICSR could include ADRs related to more than one SOC.
| SOC | Stiripentol (%) | Cannabidiol (%) | Fenfluramine (%) |
|---|---|---|---|
| Blood and lymphatic system disorders | 26 (1.2) | 47 (0.6) | 15 (0.8) |
| Cardiac disorders | 2 (0.1) | 85 (1) | 269 (14) |
| Congenital, familial, and genetic disorders | 1 (0.0) | 13 (0.2) | 8 (0.4) |
| Ear and labyrinth disorders | 1 (0.0) | 24 (0.3) | 5 (0.3) |
| Endocrine disorders | 3 (0.1) | 12 (0.1) | 4 (0.2) |
| Eye disorders | 5 (0.2) | 59 (0.7) | 11 (0.6) |
| Gastrointestinal disorders | 103 (4.9) | 550 (6.5) | 76 (4) |
| General disorders and administration site conditions | 313 (14.8) | 1200 (14.2) | 234 (12.2) |
| Hepatobiliary disorders | 15 (0.7) | 61 (0.7) | 15 (0.8) |
| Immune system disorders | 5 (0.2) | 50 (0.6) | 6 (0.3) |
| Infections and infestations | 117 (5.5) | 551 (6.5) | 83 (4.3) |
| Injury, poisoning, and procedural complications | 435 (20.6) | 1232 (14.5) | 140 (7.3) |
| Investigations | 105 (5) | 428 (5.1) | 112 (5.8) |
| Metabolism and nutritional disorders | 127 (6) | 207 (2.4) | 97 (5) |
| Musculoskeletal and connective tissue disorders | 27 (1.3) | 80 (0.9) | 20 (1) |
| Neoplasms: benign, malignant, and unspecified | 1 (0.0) | 26 (0.3) | 7 (0.4) |
| Nervous system disorders | 563 (26.6) | 2292 (27.1) | 368 (19.1) |
| Pregnancy, puerperium, and perinatal conditions | 1 (0.0) | 10 (0.1) | 3 (0.2) |
| Product issues | 6 (0.3) | 203 (2.4) | 8 (0.4) |
| Psychiatric disorders | 117 (5.5) | 566 (6.7) | 109 (5.7) |
| Renal and urinary disorders | 16 (0.8) | 68 (0.8) | 19 (1) |
| Reproductive system and breast disorders | 2 (0.1) | 16 (0.2) | 7 (0.4) |
| Respiratory, thoracic, and mediastinal disorders | 26 (1.2) | 249 (2.9) | 181 (9.4) |
| Skin and subcutaneous tissue disorders | 32 (1.5) | 151 (1.8) | 22 (1.1) |
| Social circumstances | 3 (0.1) | 49 (0.6) | 5 (0.3) |
| Surgical and medical procedures | 60 (2.8) | 173 (2) | 36 (1.9) |
| Vascular disorders | 4 (0.2) | 70 (0.8) | 62 (3.2) |
The most frequent suspected reactions related to this SOC were ‘seizure’ (stiripentol n = 423; cannabidiol n = 1609; fenfluramine n = 151) and ‘somnolence’ (stiripentol n = 84; cannabidiol n = 207; fenfluramine n = 32) (Table 3).
Table 3: Reported suspected reactions included in the group “Nervous system disorders”. Reactions with less than 10 cases have been excluded.
| Suspected Reaction | Stiripentol | Cannabidiol | Fenfluramine |
|---|---|---|---|
| Amnesia | / | 11 | / |
| Ataxia | 11 | 10 | / |
| Atonic seizures | / | 19 | / |
| Balance disorder | 14 | 33 | / |
| Cognitive disorder | / | 15 | / |
| Coma | / | 15 | / |
| Depressed level of consciousness | / | 14 | / |
| Disturbance in attention | / | 15 | / |
| Dizziness | / | 42 | 22 |
| Drooling | / | 11 | / |
| Dysarthria | / | 12 | / |
| Dyskinesia | / | 11 | / |
| Epilepsy | 17 | 61 | 11 |
| Generalized tonic–clonic seizure | 14 | 79 | 15 |
| Headache | / | 38 | 14 |
| Hypersomnia | / | 25 | / |
| Hypotonia | 14 | 10 | / |
| Lethargy | 16 | 60 | / |
| Loss of consciousness | / | 17 | / |
| Memory impairment | / | 14 | / |
| Myoclonic epilepsy | / | 12 | / |
| Partial seizures | / | 12 | / |
| Petit mal epilepsy | / | 24 | / |
| Psychomotor hyperactivity | / | 11 | / |
| Sedation | / | 47 | / |
| Seizure | 423 | 1609 | 151 |
| Seizure cluster | / | 47 | / |
| Somnolence | 84 | 207 | 32 |
| Speech disorder | / | 10 | / |
| Status epilepticus | 10 | 60 | 23 |
| Syncope | / | / | 11 |
| Tonic convulsion | / | 14 | / |
| Tremor | 10 | 26 | / |
Significantly disproportionately, reporting for fenfluramine-related reports compared to the other drugs was observed for “Cardiac disorders” (ROR = 26.29; 95% CI 25.422–27.187), “Vascular disorders” (ROR = 5.271; 4.959–5.602), and “Respiratory, thoracic, and mediastinal disorders” (ROR = 4.678; 4.579–4.779) (Table 4; Tables S1–S4). The most frequently suspected reactions related to these SOCs were pulmonary hypertension (‘pulmonary arterial hypertension’, PAH, n = 27, and ‘pulmonary hypertension’ n = 62) and valvular heart disease (VHD), including ‘aortic valve incompetence’ (n = 79), ‘cardiac valve disease’ (n = 51), ‘mitral valve incompetence’ (n = 86) ‘mitral valve stenosis’ (n = 11), and ‘tricuspid valve incompetence’ (n = 44; Table 5). Moreover, significantly higher probabilities of reporting were observed for “Product issues” with cannabidiol (ROR = 5.989; 5.148–6.968), and “Injury, poisoning, and procedural complications” (ROR = 3.033; 2.998–3.068), “Metabolism and nutrition disorders” (ROR = 2.867; 22.796–2.94), and “Blood and lymphatic system disorders” (ROR = 2.64; 2.366–2.947) with stiripentol (Table 6 and Table 7).
Table 4: Signal strength at the system organ class (SOC) level. ROR = reporting odds ratio.
| SOC | ROR | ||
|---|---|---|---|
| Cannabidiol vs. Others | Fenfluramine vs. Others | Stiripentol vs. Others | |
| Blood and lymphatic system disorders | 0.448 [0.409; 0.491] | 1.22 [1.04; 1.432] | 2.64 [2.366; 2.947] |
| Cardiac disorders | 0.106 [0.103; 0.11] | 26.29 [25.422; 27.187] | 0.033 [0.012; 0.087] |
| Congenital, familial, and genetic disorders | 0.571 [0.395; 0.827] | 3.406 [2.311; 5.018] | 0.293 [0.037; 2.291] |
| Ear and labyrinth disorders | 1.589 [1.055; 2.395] | 1.185 [0.738; 1.902] | 0.212 [0.028; 1.614] |
| Endocrine disorders | 0.679 [0.435; 1.059] | 1.581 [0.848; 2.95] | 1.158 [0.532; 2.523] |
| Eye disorders | 1.469 [1.255; 1.719] | 1.018 [0.824; 1.258] | 0.437 [0.287; 0.668] |
| Gastrointestinal disorders | 1.251 [1.231; 1.272] | 0.659 [0.638; 0.68] | 1.018 [0.992; 1.044] |
| General disorders and administration site conditions | 0.818 [0.812; 0.824] | 0.884 [0.872; 0.896] | 1.561 [1.542; 1.58] |
| Hepatobiliary disorders | 0.803 [0.728; 0.887] | 1.171 [0.999; 1.374] | 1.222 [1.042; 1.433] |
| Immune system disorders | 1.812 [1.455; 2.256] | 0.643 [0.447; 0.927] | 0.548 [0.357; 0.843] |
| Infections and infestations | 1.106 [1.089; 1.123] | 0.707 [0.686; 0.728] | 1.162 [1.136; 1.189] |
| Injury, poisoning, and procedural complications | 0.788 [0.782; 0.793] | 0.398 [0.391; 0.406] | 3.033 [2.998; 3.068] |
| Investigations | 0.757 [0.746; 0.769] | 1.281 [1.25; 1.312] | 1.23 [1.199; 1.261] |
| Metabolism and nutritional disorders | 0.334 [0.327; 0.34] | 1.812 [1.76; 1.865] | 2.867 [2.796; 2.94] |
| Musculoskeletal and connective tissue disorders | 0.669 [0.625; 0.716] | 1.109 [0.985; 1.249] | 1.689 [1.536; 1.858] |
| Neoplasms benign, malignant, and unspecified (incl cysts and polyps) | 1.29 [0.935; 1.781] | 1.539 [1.079; 2.196] | 0.186 [0.025; 1.406] |
| Nervous system disorders | 0.948 [0.942; 0.954] | 0.583 [0.577; 0.589] | 1.973 [1.948; 1.998] |
| Pregnancy, puerperium, and perinatal conditions | 0.991 [0.498; 1.972] | 1.617 [0.702; 3.723] | 0.474 [0.057; 3.925] |
| Product issues | 5.989 [5.148; 6.968] | 0.219 [0.17; 0.284] | 0.169 [0.121; 0.238] |
| Psychiatric disorders | 0.992 [0.978; 1.006] | 0.937 [0.915; 0.959] | 1.082 [1.057; 1.107] |
| Renal and urinary disorders | 0.767 [0.703; 0.836] | 1.347 [1.184; 1.533] | 1.138 [0.982; 1.319] |
| Reproductive system and breast disorders | 0.704 [0.5; 0.991] | 2.313 [1.564; 3.422] | 0.536 [0.184; 1.558] |
| Respiratory, thoracic, and mediastinal disorders | 0.444 [0.436; 0.453] | 4.678 [4.579; 4.779] | 0.353 [0.325; 0.383] |
| Skin and subcutaneous tissue disorders | 1.113 [1.057; 1.171] | 0.704 [0.635; 0.78] | 1.148 [1.064; 1.237] |
| Social circumstances | 2.445 [1.836; 3.257] | 0.567 [0.368; 0.873] | 0.341 [0.17; 0.681] |
| Surgical and medical procedures | 0.702 [0.679; 0.726] | 0.911 [0.853; 0.973] | 1.833 [1.752; 1.917] |
| Vascular disorders | 0.411 [0.387; 0.436] | 5.271 [4.959; 5.602] | 0.183 [0.11; 0.304] |
Table 5: Suspected reactions reported for fenfluramine included in the groups “Cardiac disorders”, “Vascular disorders”, and “Respiratory, thoracic, and mediastinal disorders”. Reactions with less than 10 cases have been excluded.
| Cardiac Disorders | N Overall | N from 2021 |
|---|---|---|
| Aortic valve incompetence | 79 | 2 |
| Cardiac failure | 11 | / |
| Cardiac failure congestive | 15 | / |
| Cardiac valve disease | 51 | 2 |
| Cardiovascular disorder | 60 | / |
| Left atrial dilatation | 10 | / |
| Left ventricular hypertrophy | 13 | / |
| Mitral valve incompetence | 86 | 5 |
| Mitral valve stenosis | 11 | / |
| Palpitations | 16 | 1 |
| Right ventricular failure | 11 | / |
| Tachycardia | 19 | 4 |
| Tricuspid valve incompetence | 44 | 4 |
| Respiratory, thoracic, and mediastinal disorders | / | |
| Cough | 13 | 7 |
| Dyspnoea | 63 | / |
| Pulmonary arterial hypertension | 27 | / |
| Pulmonary hypertension | 62 | 3 |
| Vascular disorders | / | |
| Hypertension | 19 | 3 |
Table 6: Suspected reactions reported for cannabidiol included in the group ‘Product issues”. Reactions with less than 10 cases have been excluded.
| Product Issues | N |
|---|---|
| Liquid product physical issue | 14 |
| Product container issue | 10 |
| Product distribution issue | 29 |
| Product quality issue | 12 |
| Product supply issue | 96 |
Table 7: Suspected reactions reported for stiripentol included in the groups ‘Injury, poisoning, and procedural complications”, “Metabolism and nutrition disorders”, and “Blood and lymphatic system disorders”. Reactions with less than 10 cases have been excluded.
| Blood and Lymphatic System Disorders | N |
|---|---|
| Neutropenia | 13 |
| Thrombocytopenia | 11 |
| Injury, poisoning, and procedural complications | |
| Fall | 31 |
| Inappropriate schedule of product administration | 13 |
| Off label use | 20 |
| Product dose omission issue | 99 |
| Product preparation issue | 104 |
| Product use in unapproved indication | 233 |
| Product use issue | 19 |
| Wrong technique in product usage process | 19 |
| Metabolism and nutritional disorders | |
| Decreased appetite | 92 |
| Dehydration | 18 |
| Hyperammonaemia | 12 |
3. Discussion
A lot of AEDs can be used to control seizures in epileptic syndromes in the first or subsequent lines (such as clobazam or valproate). We focused our analysis on drugs with similar clinical applications in the context of pediatric epilepsy-related seizures. Stiripentol, cannabidiol, and fenfluramine are AEDs with different mechanisms of action that have been approved for specific difficult-to-treat epileptic syndromes. We found differences in terms of incidence according to age and gender, but we cannot draw final conclusions due to the lack of detailed information on the use of these drugs. In particular, adults were more represented than pediatric patients for fenfluramine and cannabidiol. These drugs are approved for epileptic syndromes, but their use is not limited to children and even adults or the elderly can also be treated according to the summary of the products’ characteristics. Indeed, these syndromes generally develop in the first years of life and can persist into adulthood, though seizure types and frequency may change [ref. 45,ref. 46].
The safety profile of these drugs is quite similar, including the occurrence of some common AEs such as weight loss, decreased appetite, and drowsiness/somnolence. Nevertheless, clinical trials and post-marketing surveillance allowed us to recognize specific ADRs, such as neutropenia, thrombocytopenia, nausea, and vomiting for stiripentol, anemia, diarrhea, and infections for cannabidiol, and gastrointestinal and cardiovascular disorders for fenfluramine.
Many of the above ADRs are often due to the other anticonvulsants used in combination (e.g., clobazam and valproate) and may resolve with dose reduction.
Our analysis showed increased reporting frequencies of “Nervous system disorders” for the three drugs. The most frequent events were ‘seizure’ and similar reactions (‘atonic seizure’, ‘epilepsy’, ‘generalized tonic–clonic seizure’, ‘myoclonic epilepsy’, ‘partial seizure’, ‘petit mal epilepsy’, ‘seizure cluster’, ‘status epilepticus’, and ‘tonic convulsion’). Most of these ADRs can be derived from insufficient therapeutic control of pre-existing diseases rather than from exposure to the drug. However, seizure aggravation (or the development of new types of seizures) can occur, in theory, with all AEDs [ref. 47]. The mechanisms of this paradoxical effect are mostly unknown and may be related to specific pharmacodynamic mechanisms, e.g., enhanced GABA transmission or the blockade of voltage-gated sodium channels [ref. 48].
The SmPC of fenfluramine describes a possible clinically relevant increase in seizure frequency, which may occur during treatment and may require dose adjustment or discontinuation, as with other AEDs [ref. 41]. Indeed, seizure was among the most frequent AEs leading to study withdrawal in clinical trials and status epilepticus was observed in three patients (3%) treated with the highest dose of fenfluramine [ref. 49].
Even for cannabidiol, a clinically relevant increase in seizure frequency may occur during treatment, but the frequency of status epilepticus in the phase 3 clinical trials was similar to that of the placebo [ref. 35].
To the best of our knowledge, stiripentol seems to not be associated with paradoxical seizures [ref. 17], and the reaction ‘seizure’ is not reported in its SmPC. Therefore, our findings should be further explored to figure out the link between the events that are more frequently reported on the Eudravigilance database and treatment with stiripentol.
As expected, ‘somnolence’ was the second most reported neurological event, being already recognized as a ‘very common’ ADR in the three SmPCs.
According to the disproportionality analysis, the most significant signals at the SOC level were “Cardiac disorders”, “Vascular disorders”, and “Respiratory, thoracic, and mediastinal disorders” for fenfluramine compared to the other drugs.
These results are in accordance with the known safety profile of fenfluramine used as an anorectic agent. The drug was approved for adults to reduce body weight, but it was withdrawn in 1997 based on the increased risk of VHD and PAH, even if most of the cardiopulmonary disorders improved following drug discontinuation [ref. 50,ref. 51]. In these patients, fenfluramine was prescribed at higher dosages (>40–60 mg daily or 0.5–2.1 mg/kg/day) than those approved for epileptic syndromes, ranging from 0.2 mg/kg/day–0.7 mg/kg/day, with a maximal recommended daily dose of 17–26 mg [ref. 48,ref. 52,ref. 53,ref. 54,ref. 55,ref. 56,ref. 57,ref. 58,ref. 59,ref. 60,ref. 61,ref. 62]. No cases of VHD or PAH were observed during the studies for epileptic syndromes. These results have been confirmed in long-term studies in patients treated for 3 years. These data support the cardiovascular safety of fenfluramine at lower dosages used for seizures compared to the higher dosages used for obesity [ref. 49,ref. 63,ref. 64]. However, post-marketing data showed that VHD and PAH can also occur with low doses of fenfluramine and cardiac monitoring using echocardiography is recommended prior to starting treatment to exclude any pre-existing diseases: every 6 months for the first 2 years and annually thereafter [ref. 41]. In the case of pathological abnormalities on the echocardiogram, it is recommended to evaluate the benefit of continuing treatment compared to the risk of ADRs.
To confirm that these ADRs were related to the use of fenfluramine for epilepsy, we selected the ICSRs from the year of approval for epileptic syndromes (2020), and we found a considerable reduction in the number of events (Table 5). However, this does not exclude the risk of VHD and PAH and patients should continue to be monitored.
The pathophysiology of these side effects is still under debate but may be linked to the interaction with serotonin receptors and the consequent growth of pulmonary smooth muscle cells [ref. 65,ref. 66]. Moreover, 5HT2B receptors can be specifically involved with the hyperplasia responsible for VHD [ref. 67].
Currently, these events are listed in the SmPC of fenfluramine with a frequency ‘not known’ [ref. 41], and further investigations are thus needed to define the burden of this association.
Significantly higher probabilities of reporting “Product issues” were observed with cannabidiol, in particular ‘product supply issue’ and ‘product distribution issue’. These events were observed only with cannabidiol and were reported for more than 85% of countries from the Non-European Economic Area (Table S5). As far as we know, these issues have not been raised previously, and we can only speculate that they may be related to the nature of the product as a cannabis-derived drug.
Finally, we found a significant association for stiripentol with the onset of “Blood and lymphatic system disorders”, “Injury, poisoning, and procedural complications”, and “Metabolism and nutrition disorders”.
Neutropenia is a common AE associated with the administration of stiripentol, and blood counts should be assessed prior to starting treatment, and then monitored every 6 months [ref. 17]. Instead, thrombocytopenia has been recognized as a rare event.
As regards to “Metabolism and nutrition disorders”, it is well known that decreased appetite represents an expected event in patients treated with stiripentol, but also with fenfluramine and cannabidiol, as reported in their SmPCs [ref. 17,ref. 35,ref. 41]. This represents a critical point, especially for the pediatric population, which may experience growth retardation given the frequency of gastrointestinal ADRs (nausea and vomiting). Therefore, the growth rate of children under these treatments should be carefully monitored.
Finally, ‘product use in unapproved indication’ (e.g., LGS, frontal lobe epilepsy, epileptic encephalopathy, partial seizures, febrile convulsion, and idiopathic generalized epilepsy) was the most reported ADR in the group “Injury, poisoning, and procedural complications”, supported by data emerging regarding off-label use in other forms of epilepsy. Indeed, stiripentol demonstrated to be effective in different seizure types for pediatric patients with drug-resistant epilepsy, apart from DS [ref. 18,ref. 68,ref. 69,ref. 70,ref. 71,ref. 72,ref. 73,ref. 74,ref. 75].
Although our findings provide a comprehensive perspective in the evaluation of ADRs related to the drugs under evaluation, the results of the present study should be observed in the light of some limitations, including the lack of detailed information about the treatment, the number of patients effectively treated in the reference period, the specific patients’ characteristics, the presence of multiple suspected drugs in ICSRs, the risk of under-reporting compared to the global clinical population, and the difficulty in identifying confounders. Indeed, at this access level, detailed information about drug use is not available. In particular, treatment duration was available only for a small percentage of ICSRs (8–13%). Therefore, it is difficult to conduct further analyses or construct hypotheses. Moreover, even when this information is available, is not possible to figure out if the treatment began in pediatric age, because we only have the treatment duration and the onset age of ADRs., e.g., for an ICSR, we know that the patient experienced the ADR at the age 18–64 years and that cannabidiol was used for 500 days, but we do not know the exact age and when cannabidiol was started.
4. Materials and Methods
4.1. Data Source and Study Design
This pharmacovigilance study was conducted by analyzing AE reports from the Eudravigilance database, from December 2001 until the third quarter of 2024. Data on Individual Case Safety Reports (ICSRs) presenting the suspected drugs of stiripentol, cannabidiol, or fenfluramine were retrieved using the publicly available Eudravigilance access platform (www.adrreports.eu). The data access level used for our analysis was the one indicated as “Stakeholder Group II: Healthcare professionals, patients and the general public” in the Eudravigilance access policy.
Suspected adverse drug reactions (ADRs) were grouped according to the Medical Dictionary for Regulatory Activities (MedDRA®) [ref. 75,ref. 76,ref. 77,ref. 78,ref. 79,ref. 80] and defined as serious if they were fatal, life-threatening, required hospitalization or prolonged existing hospitalization, resulted in persistent or significant disability, represented a congenital anomaly/birth defect, or other medically important condition [ref. 81,ref. 82].
For the outcomes, ADRs were classified using the standardized terminology: ‘recovered/resolved’, ‘recovering/resolving’, ‘recovered/resolved with sequelae’, ‘not recovered/not resolved’, ‘fatal’, and ‘unknown’, on the basis of what was reported in the ICSR.
The ADR expectedness was verified by consulting the Summary of Product Characteristics (SmPCs) available in the European Medicines Agency (EMA) database [ref. 17,ref. 35,ref. 41].
4.2. Data Analysis
Starting with an Excel spreadsheet (Microsoft Office, Excel, Microsoft Corporation, Redmond, Washington) where all variables of interest were collected, the analysis was performed by developing a special script using the open-source software RStudio, version 2025.05.0-496 [ref. 83]. Descriptive statistics were used to summarize data, reporting frequencies and percentages for categorical data and median values for continuous data.
In particular, descriptive statistics were implemented to represent how the three samples were distributed according to the different variables (such as demographic, reporting, or outcome) collected. In order to assess the differences between the active ingredients, the proportion test, mathematically equivalent to the chi-square test, was conducted [ref. 84]. The associations between the drugs and AEs were determined by the reporting odds ratio (ROR), the proportional reporting ratio (PRR), the Bayesian confidence propagation neural network (BCPNN), and the multi-item gamma Poisson shrinker (MGPS) algorithms, which were based on the disproportionality analysis (Table 8).
Table 8: Two-by-two contingency table for signal detection.
| SOC of Interest | Other SOCs | Total | |
|---|---|---|---|
| Active ingredient 1 | a | b | a + b |
| Active ingredient 2 + 3 | c | d | c + d |
| Total | a + c | b + d | N |
Various tables were obtained by isolating one active ingredient from the others and for the different system organ classes (SOCs). On these contingency tables, the formulae for calculating the signal detection methods were implemented using references from the literature [ref. 85]. For each, 95% confidence intervals were given, and the thresholds reported in Table 9 were applied to them.
Table 9: Thresholds of signal detection methods. BCPNN = Bayesian confidence propagation neural network; MGPS = multi-item gamma Poisson shrinker; PRR = proportional reporting ratio; ROR = reporting odds ratio.
| Signal Detection Methods | Thresholds |
|---|---|
| ROR | Lower limit of 95% CI of ROR > 1 and a ≥ 3/5 |
| PRR | Lower limit of 95% CI of PRR > 1 and a ≥ 3/5 |
| BCPNN | Lower limit of 95% CI of IC > 0 |
| MGPS | 5th percentile of EBGM (EB05) ≥ 1.8/2 and EBGM ≥ 2.5 |
5. Conclusions
To the best of our knowledge, this is one of the first pharmacovigilance studies to compare the safety and tolerability profiles of AEDs used in difficult-to-treat epileptic syndromes.
Our analysis did not reveal new and unexpected serious safety signals but confirmed the need to strictly monitor patients to optimize treatment and minimize AE onset.
References
- T.T. Sands, J.N. Gelinas. Epilepsy and Encephalopathy. Pediatr. Neurol., 2024. [DOI | PubMed]
- R.S. Fisher, W. van Emde Boas, W. Blume, C. Elger, P. Genton, P. Lee, J. Engel. Epileptic seizures and epilepsy: Definitions proposed by the International League Against Epilepsy (ILAE) and the International Bureau for Epilepsy (IBE). Epilepsia, 2005. [DOI | PubMed]
- A.T. Berg, J.T. Langfitt, F.M. Testa, S.R. Levy, F. DiMario, M. Westerveld, J. Kulas. Global cognitive function in children with epilepsy: A community-based study. Epilepsia, 2008. [DOI | PubMed]
- C. Reilly, P. Atkinson, K.B. Das, R.F. Chin, S.E. Aylett, V. Burch, C. Gillberg, R.C. Scott, B.G. Neville. Neurobehavioral comorbidities in children with active epilepsy: A population-based study. Pediatrics, 2014. [DOI | PubMed]
- S.M. Zuberi, E. Wirrell, E. Yozawitz, J.M. Wilmshurst, N. Specchio, K. Riney, R. Pressler, S. Auvin, P. Samia, E. Hirsch. ILAE classification and definition of epilepsy syndromes with onset in neonates and infants: Position statement by the ILAE Task Force on Nosology and Definitions. Epilepsia, 2022. [DOI | PubMed]
- R. Nabbout, N. Chemaly, M. Chipaux, G. Barcia, C. Bouis, C. Dubouch, D. Leunen, I. Jambaqué, O. Dulac, G. Dellatolas. Encephalopathy in children with Dravet syndrome is not a pure consequence of epilepsy. Orphanet J. Rare Dis., 2013. [DOI | PubMed]
- A.C. Bender, R.P. Morse, R.C. Scott, G.L. Holmes, P.P. Lenck-Santini. SCN1A mutations in Dravet syndrome: Impact of interneuron dysfunction on neural networks and cognitive outcome. Epilepsy Behav., 2012. [DOI | PubMed]
- C. Dravet. The core Dravet syndrome phenotype. Epilepsia, 2011. [DOI]
- J.A. Nelson, K.G. Knupp. Lennox-Gastaut Syndrome: Current Treatments, Novel Therapeutics, and Future Directions. Neurotherapeutics, 2023. [DOI | PubMed]
- S. Lattanzi, E. Trinka, E. Russo, C. Del Giovane, S. Matricardi, S. Meletti, P. Striano, P.T. Damavandi, M. Silvestrini, F. Brigo. Pharmacotherapy for Dravet Syndrome: A Systematic Review and Network Meta-Analysis of Randomized Controlled Trials. Drugs, 2023. [DOI | PubMed]
- E.C. Wirrell, V. Hood, K.G. Knupp, M.A. Meskis, R. Nabbout, I.E. Scheffer, J. Wilmshurst, J. Sullivan. International consensus on diagnosis and management of Dravet syndrome. Epilepsia, 2022. [DOI | PubMed]
- A. Fattorusso, S. Matricardi, E. Mencaroni, G.B. Dell’Isola, G. Di Cara, P. Striano, A. Verrotti. The Pharmacoresistant Epilepsy: An Overview on Existent and New Emerging Therapies. Front. Neurol., 2021. [DOI | PubMed]
- A. Verrotti, P. Striano, G. Iapadre, L. Zagaroli, P. Bonanni, G. Coppola, M. Elia, O. Mecarelli, E. Franzoni, P. Liso. The pharmacological management of Lennox-Gastaut syndrome and critical literature review. Seizure, 2018. [DOI | PubMed]
- S. Schubert-Bast, M. Wolff, A. Wiemer-Kruel, S. von Spiczak, R. Trollmann, P.S. Reif, C. Pritchard, T. Polster, B.A. Neubauer, T. Mayer. Seizure management and prescription patterns of anticonvulsants in Dravet syndrome: A multicenter cohort study from Germany and review of literature. Epilepsy Behav., 2019. [DOI | PubMed]
- A. Arzimanoglou, J. French, W.T. Blume, J.H. Cross, J.P. Ernst, M. Feucht, P. Genton, R. Guerrini, G. Kluger, J.M. Pellock. Lennox-Gastaut syndrome: A consensus approach on diagnosis, assessment, management, and trial methodology. Lancet Neurol., 2009. [DOI | PubMed]
- J.H. Cross, S. Auvin, M. Falip, P. Striano, A. Arzimanoglou. Expert Opinion on the Management of Lennox-Gastaut Syndrome: Treatment Algorithms and Practical Considerations. Front. Neurol., 2017. [DOI | PubMed]
- Diacomit: EPAR—Product Information. 2025
- S. Balestrini, V. Doccini, A. Boncristiano, M. Lenge, S. De Masi, R. Guerrini. Efficacy and Safety of Long-Term Treatment with Stiripentol in Children and Adults with Drug-Resistant Epilepsies: A Retrospective Cohort Study of 196 Patients. Drugs Real. World Outcomes, 2022. [DOI | PubMed]
- L. Moller, O.J. Simon, C. Junemann, M. Austermann-Menche, M.P. Bergmann, L. Habermehl, K. Menzler, L. Timmermann, A. Strzelczyk, S. Knake. Stiripentol for the treatment of refractory status epilepticus. Neurol. Res. Pract., 2024. [DOI | PubMed]
- A. Gil-Nagel, A. Aledo-Serrano, A. Beltran-Corbellini, L. Martinez-Vicente, A. Jimenez-Huete, R. Toledano-Delgado, I. Gacía-Morales, A. Valls-Carbó. Efficacy and tolerability of add-on stiripentol in real-world clinical practice: An observational study in Dravet syndrome and non-Dravet developmental and epileptic encephalopathies. Epilepsia Open, 2024. [DOI | PubMed]
- K.A. Myers, P. Lightfoot, S.G. Patil, J.H. Cross, I.E. Scheffer. Stiripentol efficacy and safety in Dravet syndrome: A 12-year observational study. Dev. Med. Child. Neurol., 2018. [DOI | PubMed]
- N. Specchio, S. Auvin, A. Strzelczyk, F. Brigo, V. Villanueva, E. Trinka. Efficacy and safety of stiripentol in the prevention and cessation of status epilepticus: A systematic review. Epilepsia Open, 2024. [DOI | PubMed]
- M. Yamada, K. Suzuki, D. Matsui, Y. Inoue, Y. Ohtsuka. Long-term safety and effectiveness of stiripentol in patients with Dravet syndrome: Interim report of a post-marketing surveillance study in Japan. Epilepsy Res., 2021. [PubMed]
- R. Guerrini, L. Chancharme, B. Serraz, C. Chiron. Additional Results from Two Randomized, Placebo-Controlled Trials of Stiripentol in Dravet Syndrome Highlight a Rapid Antiseizure Efficacy with Longer Seizure-Free Periods. Neurol. Ther., 2024. [DOI | PubMed]
- N. Stella. THC and CBD: Similarities and differences between siblings. Neuron, 2023. [DOI | PubMed]
- S. Liu, Z. He, J. Li. Long-term efficacy and adverse effects of cannabidiol in adjuvant treatment of drug-resistant epilepsy: A systematic review and meta-analysis. Ther. Adv. Neurol. Disord., 2023. [DOI | PubMed]
- A. Manduca, V. Buzzelli, A. Rava, A. Feo, E. Carbone, S. Schiavi, B. Peruzzi, V. D’Oria, M. Pezzullo, A. Pasquadibisceglie. Cannabidiol and positive effects on object recognition memory in an in vivo model of Fragile X Syndrome: Obligatory role of hippocampal GPR55 receptors. Pharmacol. Res., 2024. [DOI | PubMed]
- M.R. Ghovanloo, N.G. Shuart, J. Mezeyova, R.A. Dean, P.C. Ruben, S.J. Goodchild. Inhibitory effects of cannabidiol on voltage-dependent sodium currents. J. Biol. Chem., 2018. [DOI | PubMed]
- T. Stark, M. Di Bartolomeo, R. Di Marco, E. Drazanova, C.B.M. Platania, F.A. Iannotti, J. Ruda-Kucerova, C. D’Addario, L. Kratka, V. Pekarik. Altered dopamine D3 receptor gene expression in MAM model of schizophrenia is reversed by peripubertal cannabidiol treatment. Biochem. Pharmacol., 2020. [DOI | PubMed]
- M. Di Bartolomeo, T. Stark, O.M. Maurel, F.A. Iannotti, M. Kuchar, J. Ruda-Kucerova, F. Piscitelli, S. Laudani, V. Pekarik, S. Salomone. Crosstalk between the transcriptional regulation of dopamine D2 and cannabinoid CB1 receptors in schizophrenia: Analyses in patients and in perinatal Delta9-tetrahydrocannabinol-exposed rats. Pharmacol. Res., 2021. [PubMed]
- T. Stark, J. Ruda-Kucerova, F.A. Iannotti, C. D’Addario, R. Di Marco, V. Pekarik, E. Drazanova, F. Piscitelli, M. Bari, Z. Babinska. Peripubertal cannabidiol treatment rescues behavioral and neurochemical abnormalities in the MAM model of schizophrenia. Neuropharmacology, 2019. [DOI | PubMed]
- T. Stark, S. Di Martino, F. Drago, C.T. Wotjak, V. Micale. Phytocannabinoids and schizophrenia: Focus on adolescence as a critical window of enhanced vulnerability and opportunity for treatment. Pharmacol. Res., 2021. [DOI | PubMed]
- V. Micale, M. Di Bartolomeo, S. Di Martino, T. Stark, B. Dell’Osso, F. Drago, C. D’Addario. Are the epigenetic changes predictive of therapeutic efficacy for psychiatric disorders? A translational approach towards novel drug targets. Pharmacol. Ther., 2023. [DOI | PubMed]
- P.N. Patsalos, J.P. Szaflarski, B. Gidal, K. VanLandingham, D. Critchley, G. Morrison. Clinical implications of trials investigating drug-drug interactions between cannabidiol and enzyme inducers or inhibitors or common antiseizure drugs. Epilepsia, 2020. [DOI | PubMed]
- Epidyolex: EPAR—Product Information. 2025
- O. Devinsky, A.D. Patel, J.H. Cross, V. Villanueva, E.C. Wirrell, M. Privitera, S.M. Greenwood, C. Roberts, D. Checketts, K.E. VanLandingham. Effect of Cannabidiol on Drop Seizures in the Lennox-Gastaut Syndrome. N. Engl. J. Med., 2018. [DOI | PubMed]
- O. Devinsky, A.D. Patel, E.A. Thiele, M.H. Wong, R. Appleton, C.L. Harden, S. Greenwood, G. Morrison, K. Sommerville. Randomized, dose-ranging safety trial of cannabidiol in Dravet syndrome. Neurology, 2018. [DOI | PubMed]
- O. Devinsky, J.H. Cross, L. Laux, E. Marsh, I. Miller, R. Nabbout, I.E. Scheffer, E.A. Thiele, S. Wright. Trial of Cannabidiol for Drug-Resistant Seizures in the Dravet Syndrome. N. Engl. J. Med., 2017. [PubMed]
- L. Fiorillo, M. Cicciu, T.F. Tozum, M. Saccucci, C. Orlando, G.L. Romano, C. D’Amico, G. Cervino. Endosseous Dental Implant Materials and Clinical Outcomes of Different Alloys: A Systematic Review. Materials, 2022. [DOI | PubMed]
- J.E. Frampton. Fenfluramine: A Review in Dravet and Lennox-Gastaut Syndromes. Drugs, 2023. [PubMed]
- Fintepla: EPAR—Product Information. 2025
- K.G. Knupp, I.E. Scheffer, B. Ceulemans, J. Sullivan, K.C. Nickels, L. Lagae, R. Guerrini, S.M. Zuberi, R. Nabbout, K. Riney. Fenfluramine provides clinically meaningful reduction in frequency of drop seizures in patients with Lennox-Gastaut syndrome: Interim analysis of an open-label extension study. Epilepsia, 2023. [DOI | PubMed]
- R. Guerrini, N. Specchio, A. Aledo-Serrano, M. Pringsheim, F. Darra, T. Mayer, A. Gil-Nagel, T. Polster, S.M. Zuberi, A. Lothe. An examination of the efficacy and safety of fenfluramine in adults, children, and adolescents with Dravet syndrome in a real-world practice setting: A report from the Fenfluramine European Early Access Program. Epilepsia Open, 2022. [DOI | PubMed]
- M. Banovac, G. Candore, J. Slattery, F. Houyez, D. Haerry, G. Genov, P. Arlett. Patient Reporting in the EU: Analysis of EudraVigilance Data. Drug Saf., 2017. [DOI | PubMed]
- S. Balestrini, R. Guerrini, S.M. Sisodiya. Rare and Complex Epilepsies from Childhood to Adulthood: Requirements for Separate Management or Scope for a Lifespan Holistic Approach?. Curr. Neurol. Neurosci. Rep., 2021. [DOI | PubMed]
- H. Oguni, S. Ito, A. Nishikawa, Y. Otani, S. Nagata. Transition from pediatric to adult care in a Japanese cohort of childhood-onset epilepsy: Prevalence of epileptic syndromes and complexity in the transition. Seizure, 2021. [DOI | PubMed]
- M. Sazgar, B.F. Bourgeois. Aggravation of epilepsy by antiepileptic drugs. Pediatr. Neurol., 2005. [DOI | PubMed]
- J. Aicardi, H. Gastaut. Treatment of self-induced photosensitive epilepsy with fenfluramine. N. Engl. J. Med., 1985. [PubMed]
- K.G. Knupp, I.E. Scheffer, B. Ceulemans, J.E. Sullivan, K.C. Nickels, L. Lagae, R. Guerrini, S.M. Zuberi, R. Nabbout, K. Riney. Efficacy and Safety of Fenfluramine for the Treatment of Seizures Associated With Lennox-Gastaut Syndrome: A Randomized Clinical Trial. JAMA Neurol., 2022. [DOI | PubMed]
- D.D. Hensrud, H.M. Connolly, M. Grogan, F.A. Miller, K.R. Bailey, M.D. Jensen. Echocardiographic improvement over time after cessation of use of fenfluramine and phentermine. Mayo Clin. Proc., 1999. [DOI | PubMed]
- L. Abenhaim, Y. Moride, F. Brenot, S. Rich, J. Benichou, X. Kurz, T. Higenbottam, C. Oakley, E. Wouters, M. Aubier. Appetite-suppressant drugs and the risk of primary pulmonary hypertension. International Primary Pulmonary Hypertension Study Group. N. Engl. J. Med., 1996. [DOI | PubMed]
- C.F. Dahl, M.R. Allen, P.M. Urie, P.N. Hopkins. Valvular regurgitation and surgery associated with fenfluramine use: An analysis of 5743 individuals. BMC Med., 2008. [DOI | PubMed]
- P.N. Hopkins, G.I. Polukoff. Risk of valvular heart disease associated with use of fenfluramine. BMC Cardiovasc. Disord., 2003. [DOI | PubMed]
- H. Jick, C. Vasilakis, L.A. Weinrauch, C.R. Meier, S.S. Jick, L.E. Derby. A population-based study of appetite-suppressant drugs and the risk of cardiac-valve regurgitation. N. Engl. J. Med., 1998. [DOI | PubMed]
- S. Rich, L. Rubin, A.M. Walker, S. Schneeweiss, L. Abenhaim. Anorexigens and pulmonary hypertension in the United States: Results from the surveillance of North American pulmonary hypertension. Chest, 2000. [DOI | PubMed]
- A. Schoonjans, B.P. Paelinck, F. Marchau, B. Gunning, A. Gammaitoni, B.S. Galer, L. Lagae, B. Ceulemans. Low-dose fenfluramine significantly reduces seizure frequency in Dravet syndrome: A prospective study of a new cohort of patients. Eur. J. Neurol., 2017. [DOI | PubMed]
- Y. Xu, D. Chen, L. Liu. Optimal dose of fenfluramine in adjuvant treatment of drug-resistant epilepsy: Evidence from randomized controlled trials. Front. Neurol., 2024. [DOI | PubMed]
- A.S. Schoonjans, L. Lagae, B. Ceulemans. Low-dose fenfluramine in the treatment of neurologic disorders: Experience in Dravet syndrome. Ther. Adv. Neurol. Disord., 2015. [DOI | PubMed]
- B. Ceulemans, M. Boel, K. Leyssens, C. Van Rossem, P. Neels, P.G. Jorens, L. Lagae. Successful use of fenfluramine as an add-on treatment for Dravet syndrome. Epilepsia, 2012. [DOI | PubMed]
- Y.K. Loke, S. Derry, A. Pritchard-Copley. Appetite suppressants and valvular heart disease—A systematic review. BMC Clin. Pharmacol., 2002. [DOI | PubMed]
- P. Tabaee Damavandi, N. Fabin, R. Giossi, S. Matricardi, C. Del Giovane, P. Striano, S. Meletti, F. Brigo, E. Trinka, S. Lattanzi. Efficacy and Safety of Fenfluramine in Epilepsy: A Systematic Review and Meta-analysis. Neurol. Ther., 2023. [DOI | PubMed]
- I.K. Sharawat, P.K. Panda, A. Kasinathan, P. Panda, L. Dawman, K. Joshi. Efficacy and tolerability of fenfluramine in patients with Dravet syndrome: A systematic review and meta-analysis. Seizure, 2021. [DOI | PubMed]
- R. Nabbout, A. Mistry, S. Zuberi, N. Villeneuve, A. Gil-Nagel, R. Sanchez-Carpintero, U. Stephani, L. Laux, E. Wirrell, K. Knupp. Fenfluramine for Treatment-Resistant Seizures in Patients With Dravet Syndrome Receiving Stiripentol-Inclusive Regimens: A Randomized Clinical Trial. JAMA Neurol., 2020. [DOI | PubMed]
- A. Agarwal, G.M. Farfel, A.R. Gammaitoni, P.C. Wong, F.J. Pinto, B.S. Galer. Long-term cardiovascular safety of fenfluramine in patients with Dravet syndrome treated for up to 3 years: Findings from serial echocardiographic assessments. Eur. J. Paediatr. Neurol., 2022. [DOI | PubMed]
- M. McGee, N. Whitehead, J. Martin, N. Collins. Drug-associated pulmonary arterial hypertension. Clin. Toxicol., 2018. [DOI]
- R.B. Rothman, J.B. Redmon, S.K. Raatz, C.A. Kwong, J.E. Swanson, J.P. Bantle. Chronic treatment with phentermine combined with fenfluramine lowers plasma serotonin. Am. J. Cardiol., 2000. [DOI | PubMed]
- R.B. Rothman, M.H. Baumann, J.E. Savage, L. Rauser, A. McBride, S.J. Hufeisen, B.L. Roth. Evidence for possible involvement of 5-HT(2B) receptors in the cardiac valvulopathy associated with fenfluramine and other serotonergic medications. Circulation, 2000. [DOI | PubMed]
- L. Habermehl, P.M. Mross, K. Krause, I. Immisch, D. Chiru, F. Zahnert, I. Gorny, A. Strzelczyk, F. Rosenow, L. Möller. Stiripentol in the treatment of adults with focal epilepsy- a retrospective analysis. Seizure, 2021. [DOI | PubMed]
- A. Verrotti, G. Prezioso, S. Stagi, M.C. Paolino, P. Parisi. Pharmacological considerations in the use of stiripentol for the treatment of epilepsy. Expert. Opin. Drug Metab. Toxicol., 2016. [DOI | PubMed]
- M. Trivisano, N. Specchio, F. Vigevano. Extending the use of stiripentol to other epileptic syndromes: A case of PCDH19-related epilepsy. Eur. J. Paediatr. Neurol., 2015. [DOI | PubMed]
- A. Rosati, A. Boncristiano, V. Doccini, A. Pugi, T. Pisano, M. Lenge, S. De Masi, R. Guerrini. Long-term efficacy of add-on stiripentol treatment in children, adolescents, and young adults with refractory epilepsies: A single center prospective observational study. Epilepsia, 2019. [DOI | PubMed]
- V. Soto-Insuga, E. Gonzalez-Alguacil, M. Ballara-Petitbo, N. Lamagrande-Casanova, A. Duat-Rodriguez, C. Benitez-Provedo, E. Cardenal-Muñoz, J.J. García-Peñas. Efficacy of Stiripentol Beyond Dravet Syndrome: A Retrospective Medical Record Review of Patients with Drug-Resistant Epilepsies. Neurol. Ther., 2025. [DOI | PubMed]
- R. Amato, F. Lazzara, T.H. Chou, G.L. Romano, M. Cammalleri, M. Dal Monte, G. Casini, V. Porciatti. Diabetes Exacerbates the Intraocular Pressure-Independent Retinal Ganglion Cells Degeneration in the DBA/2J Model of Glaucoma. Investig. Ophthalmol. Vis. Sci., 2021. [DOI]
- L. Gozzo, G.L. Romano, F. Romano, S. Brancati, L. Longo, D.C. Vitale, F. Drago. Health Technology Assessment of Advanced Therapy Medicinal Products: Comparison Among 3 European Countries. Front. Pharmacol., 2021. [DOI | PubMed]
- L. Gozzo, L. Longo, D.C. Vitale, F. Drago. The Regulatory Challenges for Drug Repurposing During the Covid-19 Pandemic: The Italian Experience. Front. Pharmacol., 2020. [DOI | PubMed]
- S. Brancati, L. Gozzo, G.L. Romano, C. Vetro, I. Dulcamare, C. Maugeri, M. Parisi, L. Longo, D.C. Vitale, F. Di Raimondo. Venetoclax in Relapsed/Refractory Acute Myeloid Leukemia: Are Supporting Evidences Enough?. Cancers, 2021. [DOI | PubMed]
- E.G. Brown. Methods and pitfalls in searching drug safety databases utilising the Medical Dictionary for Regulatory Activities (MedDRA). Drug Saf., 2003. [DOI | PubMed]
- C. Vetro, A. Duminuco, L. Gozzo, C. Maugeri, M. Parisi, S. Brancati, L. Longo, D.C. Vitale, G.L. Romano, R. Ciuni. Pegylated Asparaginase-Induced Liver Injury: A Case-Based Review and Data From Pharmacovigilance. J. Clin. Pharmacol., 2022. [DOI | PubMed]
- L. Gozzo, C. Vetro, S. Brancati, L. Longo, D.C. Vitale, G.L. Romano, E. Mauro, P.F. Fiumara, C. Maugeri, M.S. Parisi. Off-Label Use of Venetoclax in Patients With Acute Myeloid Leukemia: Single Center Experience and Data From Pharmacovigilance Database. Front. Pharmacol., 2021. [DOI | PubMed]
- L. Gozzo, A. Nardo, S. Brancati, A. Judica, A. Duminuco, C. Maugeri, M. Parisi, L. Longo, D.C. Vitale, R. Ruscica. Severe Gastrointestinal Toxicity Following the Use of Gilteritinib: A Case Series and Analysis of Postmarketing Surveillance Data. Healthcare, 2023. [DOI | PubMed]
- MA Inclusion/Exclusion Criteria for the “Important Medical Events” List Criteria for MedDRA Terms on the IME List. 2025
- G. Minervini, M. Basili, R. Franco, P. Bollero, M. Mancini, L. Gozzo, G.L. Romano, M.M. Marrapodi, F. Gorassini, C. D’Amico. Periodontal Disease and Pregnancy: Correlation with Underweight Birth. Eur. J. Dent., 2023. [DOI | PubMed]
- RStudio: Integrated Development for R; RStudio, PBC: Boston, MA, USA, 2023
- J.A.L. Sauro, J.R. Lewis. Chapter 5-is there a statistical difference between designs. Quantifying the User Experience: Practical Statistics for User Research, 2016
- A. Bate, M. Lindquist, I.R. Edwards, S. Olsson, R. Orre, A. Lansner, R.M. De Freitas. A Bayesian neural network method for adverse drug reaction signal generation. Eur. J. Clin. Pharmacol., 1998. [DOI | PubMed]
