Substance use and use disorders among Veterans on long-term opioid therapy
Abstract
Background:
Few studies have reported on the prevalence and health risks associated with substance use and substance use disorder (SU/SUD) in Veterans who use long-term opioid therapy (LTOT). We leveraged health record data to estimate SU/SUD prevalence and its association with mortality among Veterans on LTOT.
Methods:
We conducted a secondary analysis of cohort data for Veterans on LTOT within Veterans Health Administration outpatient settings (2014–2019). SU/SUD was defined as a positive screen for risky alcohol use; a positive urine drug screen for cannabis, benzodiazepines, or stimulants; or a documented SUD diagnosis. We fit Cox models for all-cause mortality, fatal overdose, and traumatic deaths, comparing Veterans on LTOT with SU/SUD vs. LTOT-only.
Results:
One in four (25.0 %) Veterans on LTOT have risky alcohol use, tested positive for other substances, or had a diagnosed SUD. Alcohol was the most common SU/SUD (9.8 %), followed by sedative (8.1 %), cannabis (6.6 %), and stimulant (0.6 %). Relative to Veterans on LTOT only, mortality rates were higher for Veterans on LTOT with cannabis (HR=1.16, 95 % CI=1.03, 1.30), sedative (HR=1.29, 95 % CI=1.10, 1.52), or stimulant SU/SUD (HR=1.54, 95 % CI=1.17, 2.02). Fatal overdose rates were higher for LTOT with alcohol (HR=1.43, 95 % CI=1.10, 1.86), sedatives (HR=1.40, 95 % CI=1.04, 1.91), or stimulant SU/SUD (HR=3.29, 95 % CI=1.60, 6.77). LTOT with sedative SU/SUD was associated with traumatic death rates (HR=1.30, 95 % CI=1.05, 1.61).
Conclusion:
Substance co-use is common among Veterans on LTOT and is associated with elevated mortality and overdose risks. Comprehensive screening and targeted interventions may be needed.
Article type: Research Article
Keywords: Veterans, Long-term opioid therapy, Substance co-use, Mortality, Fatal overdose, Traumatic deaths
Affiliations: Center for Data to Discovery and Delivery Innovation (3DI), San Francisco VA Health Care System, CA, USA; National Clinician Scholars Program (NCSP) at UCSF, San Francisco, CA, USA; School of Medicine, University of California, San Francisco, CA, USA
License: CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.dadr.2025.100347 | PubMed: 40520458 | PMC: PMC12166433
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.1 MB)
Introduction
Substance use disorder (SUD) is pervasive in the U.S., affecting over 48 million Americans aged 12 and above in 2022 (ref. Substance Abuse and Mental Health Services Administration, 2023). The impact of SUD on healthcare systems and society is immense, with opioids and stimulants accounting for 81.8 % and 57.1 % of drug overdose deaths, respectively in 2022 (ref. Centers for Disease Control and Prevention, 2023). SUD-related healthcare costs are estimated at $35 billion, including $10.2 billion for alcohol-related disorders and $7.3 billion for opioid-related disorders (ref. Li et al., 2023). SUD also contributes to increased hospitalizations and emergency department visits (ref. Lewer et al., 2020), incurring significant costs for healthcare system.
Veterans represent a particularly vulnerable subpopulation for opioid misuse. Veterans have high rates of chronic pain, often due to service-related injuries and trauma, and many rely on opioid therapy for pain management. Despite reductions in opioid prescriptions over the past decade, more than 50 % of Veterans prescribed opioids in 2023 were on long-term opioid therapy (LTOT) (ref. U.S. Department of Veterans Affairs, 2023), which is using opioids for pain for more than three months (ref. Karmali et al., 2020). Many Veterans initiated on LTOT maintain persistently high opioid use for over two years (ref. Hayes et al., 2021), increasing their risk for opioid use disorder (OUD). In addition to chronic pain, many Veterans experience high rates of PTSD and depression (ref. Athey and Overholser, 2018, ref. Lan et al., 2016), as well as socioeconomic vulnerabilities, such as housing instability and financial insecurity (ref. Austin et al., 2021), which also increase the risk for opioid and other SUD. Overall,18 % of Veterans diagnosed with SUD in the past year in 2022, with one study revealed 58.8 % of Veterans diagnosed with OUD are also diagnosed with at least one additional SUD (ref. Lin, Bohnert, et al., 2021).
The health risks associated with LTOT may be greater for the many patients who use other substances (ref. Dobscha et al., 2013, ref. Vowles et al., 2022). Furthermore, co-use of other substances, such as sedatives, in addition to LTOT, have been shown to increase risk adverse health outcomes, including falls and emergency department visits (ref. Yarborough et al., 2019). Current opioid prescribing guidelines primarily address risks associated with opioid co-use with alcohol and sedatives (e.g., benzodiazepines) and pay less attention to co-use with other substances, such as cannabis and stimulants (ref. Dowell et al., 2022, ref. VA/DoD Clinical Practice Guideline, T. L. G., 2022). This oversight is concerning, as stimulants and other substances were detected in 63 % of opioid-related overdose deaths across 25 U.S. states (ref. Gladden et al., 2019). Additionally, overdose mortality involving cocaine and psychostimulants (e.g., methamphetamine) alone among Veterans increased by over 200 % and 600 %, respectively, from 2010 to 2019 (ref. Begley et al., 2022).
To date, comprehensive data on the prevalence and mortality risks of SU/SUD among Veterans on LTOT remains limited. While prior research has identified risk factors for multiple SU/SUD in Veterans (ref. Bhalla et al., 2017, ref. Cypel et al., 2023), explored overdose mortality trends (ref. Begley et al., 2022), and examined the association between SUDs and suicide mortality (ref. Bohnert et al., 2017), these studies do not focus on Veterans receiving LTOT with other forms of substance use (i.e., multiple SU/SUDs) using biomarker data. In addition, existing studies examining substance co-use with LTOT have largely relied on self-reported data or medical records without biomarker confirmation (e.g., urine drug screening), which may underestimate the true prevalence of substance use.
This study addressed these gaps by leveraging longitudinal data from Veterans Health Administration, incorporating urine drug screens to provide biomarker confirmation of substance use. This study specifically aimed to examine Veterans on LTOT to estimate the prevalence of co-occurring SU/SUD and to assess whether SU/SUD was associated with increased rates of all-cause or cause-specific mortality (fatal overdose and traumatic deaths).
Methods
Sample
This is a secondary analysis of national VA and Medicare datasets from a retrospective cohort study on the associations of cannabis use with mortality and hospital visits among Veterans on LTOT (ref. Keyhani et al., 2022a). The cohort included all Veterans who completed their first urine drug screening (UDS) in a VA outpatient setting between January 1, 2014, and December 31, 2019 and who were prescribed LTOT (defined as those receiving opioid therapy for at least 84 days within a 90-day period, aligning with typical VA prescribing practices of 30-day dispensing intervals and accounting for refill delays or prescription gaps (ref. Karmali et al., 2020)). The date of the UDS was the “index date” to anchor the assessment of other variables and indicate the start of follow-up for mortality outcomes. Patients were excluded if they were potentially at the end of life (e.g., those residing in a VA community living center, receiving palliative care, or undergoing inpatient cancer chemotherapy) or had a Care Assessment Needs (CAN) score of 99, indicating a very high likelihood of death within 90 days (ref. Wang et al., 2013). We also excluded patients prescribed THC-containing medications (e.g., dronabinol) to avoid false-positive urine drug screens for THC and individuals treated in opiate treatment programs (e.g., methadone clinics) as their primary reason for receiving opioids was opioid use disorder, not pain management. We further excluded opioids formulations inconsistent with chronic pain treatment, including single-agent and combination products primarily used for palliative care or opioid use disorder (e.g., buprenorphine) (Supplemental). However, we did not exclude individuals prescribed stimulants or benzodiazepines, as the study aimed to examine their potential associations with the outcomes. Data were obtained from the VA Corporate Data Warehouse, which contains comprehensive patient demographic information, health-related records, and diagnostic codes, with additional data on Medicare utilized by Veterans in the cohort. The study was approved by institutional review board at the University of California, San Francisco.
Exposure
The primary exposures were for co-use of alcohol, cannabis, stimulants (cocaine and amphetamine), and sedatives (primarily benzodiazepine) along with LTOT. At the VA, urine drug screening (UDS) has been implemented as a risk mitigation strategy for patients prescribed opioids for pain. Engagement with this screening has been monitored since 2013 and became mandatory in 2014. Currently, over 90 % of patients receiving long-term opioids for chronic pain undergo UDS (ref. Sandbrink et al., 2020). Risky alcohol use (consumption above recommended limits) was measured using the 3-item Alcohol Use Disorder Identification Test (AUDIT-C), with a cut-off score of 4 for men and 3 for women (ref. Bradley et al., 1998, ref. Bush et al., 1998). Opioids, cannabis, stimulant, and benzodiazepine use was detected by at least one positive UDS. Substance-specific use disorders were measured based on the International Classification of Diseases (ICD) 9 and 10 codes for alcohol, cannabis, stimulant, and sedative use disorders within the prior two years before the index date (Supplemental).
We categorized Veterans on LTOT into five groups, focusing specifically on those with LTOT and one additional SU/SUD. These groups were: (1) LTOT-only as the reference group (without positive UDS for other substance or a ICD-recorded SUD diagnosis) (2) alcohol group (risky alcohol use or ICD-recorded alcohol use disorder), (3) cannabis group (positive cannabis use on UDS or ICD-recorded cannabis use disorder), (4) sedative group (positive benzodiazepine on UDS or ICD-recorded sedative use disorder), and (5) stimulant group (positive stimulant use on UDS or ICD-recorded stimulant use disorders). The sedative group captures primarily benzodiazepines and may include barbiturates.
Outcomes
The outcomes were all-cause mortality, fatal overdose (either intentional, unintentional, or provisional drug-related deaths), and traumatic deaths (accidents or unintentional injuries, suicide, and homicide). These outcomes were extracted from the VA Mortality Data repository, and ICD-9 and 10 codes were used to categorize data related to causes of death (Supplemental).
Covariates
Demographic variables included age, sex, race, ethnicity, and marital status (Table 1). Health-related factors comprised mental health conditions (psychosis, depression, bipolar disorder, posttraumatic stress disorder, anxiety, and self-harm) and pain-related diagnoses (back and spine disorders, neck and spine disorders, osteoarthritis, neuropathy, headache, and traumatic brain injury). These conditions were identified using the ICD-9 and 10 codes. Social factors were housing insecurity and lack of social support, which both were also confirmed with ICD codes. The ICD-coded health and social factors were assessed for the two years prior to the index UDS. The Care Assessment Need (CAN) score integrates patient demographics, comorbidities, healthcare utilization, and socioeconomic factors to predict hospitalization and estimate 90-day mortality risk (ref. Fihn and Box, 2013, ref. Fihn and Box, 2016). The score ranges from 0 (low) to 99 (high mortality risk), updated weekly, and patients with a score of 99 were excluded.
Table 1: Baseline characteristics of Veterans on long term opioid therapy (LTOT, ≥ 84 Days) in past 90 days (N = 167,547).
| Characteristics | Total Sample (N = 167,547)N (%) or Mean (SD, range) | With Any Other SU/SUD (N = 128,185)N (%) or Mean (SD) | Without Other SU/SUD (N = 39,362)N (%) or Mean (SD) | P-value | ||
|---|---|---|---|---|---|---|
| Demographics | ||||||
| Age | 62.7 (11.4, 21–117) | 60.5 (11.7) | 60.5 (10.2) | < 0.001 | ||
| 18–34 | 3307 (2.0 %) | 978 (2.5 %) | 2329 (1.8 %) | < 0.001 | ||
| 35–49 | 14,951 (8.9 %) | 3670 (9.3 %) | 11,281 (8.8 %) | |||
| 50–64 | 60,290 (36.0 %) | 17,492 (44.4 %) | 42,798 (33.4 %) | |||
| 65–74 | 67,375 (40.2 %) | 15,109 (38.4 %) | 52,266 (40.8 %) | |||
| 75 or older | 21,624 (12.9 %) | 2113 (5.4 %) | 19,511 (15.2 %) | |||
| Sex | ||||||
| Male | 157,026 (93.7 %) | 37,438 (95.1 %) | 119,588 (93.3 %) | < 0.001 | ||
| Female | 10,521 (6.3 %) | 1924 (4.9 %) | 8597 (6.7 %) | |||
| Race | ||||||
| American Indian or Alaska Native | 1729 (1.0 %) | 512 (1.3 %) | 1217 (1.0 %) | < 0.001 | ||
| Asian | 396 (0.2 %) | 87 (0.2 %) | 309 (0.2 %) | |||
| Black or African American | 19,854 (11.9 %) | 5231 (13.3 %) | 14,623 (11.4 %) | |||
| Native Hawaiian or Other Pacific Islander | 1168 (0.7 %) | 281 (0.7 %) | 887 (0.7 %) | |||
| White | 134,282 (80.2 %) | 30,776 (78.2 %) | 103,506 (80.8 %) | |||
| More than one race/missing | 10,118 (6.0 %) | 2475 (6.3 %) | 7643 (6.0 %) | |||
| Hispanic/Latinx | 5628 (3.4 %) | 1539 (3.9 %) | 4089 (3.2 %) | < 0.001 | ||
| Married | 88,931 (53.1 %) | 16,795 (42.7 %) | 72,136 (56.3 %) | < 0.001 | ||
| Health-Related Factors | ||||||
| Mental health diagnosesa (prior 2 years) | ||||||
| Psychosis | 4209 (2.5 %) | 1457 (3.7 %) | 2752 (2.2 %) | < 0.001 | ||
| Depression | 60,435 (36.1 %) | 17,221 (43.8 %) | 43,214 (33.7 %) | < 0.001 | ||
| Bipolar | 6391 (3.8 %) | 2418 (6.1 %) | 3973 (3.1 %) | < 0.001 | ||
| PTSDb | 17,481 (10.4 %) | 5469 (13.9 %) | 12,012 (9.4 %) | < 0.001 | ||
| Anxiety | 34,010 (20.3 %) | 9930 (25.2 %) | 24,080 (18.8 %) | < 0.001 | ||
| Self-harm | 572 (0.3 %) | 301 (0.8 %) | 271 (0.2 %) | < 0.001 | ||
| Pain-related diagnosesa (prior 2 years) | ||||||
| Back and spine disorders | 117,679 (70.2 %) | 27,638 (70.2 %) | 90,041 (70.2 %) | 0.915 | ||
| Neck and spine disorders | 39,476 (23.6 %) | 9770 (24.8 %) | 29,706 (21.2 %) | < 0.001 | ||
| Osteoarthritis | 57,273 (34.2 %) | 12,654 (32.2 %) | 44,619 (34.8 %) | < 0.001 | ||
| Neuropathy | 33,909 (20.2 %) | 6792 (17.3 %) | 27,117 (21.2 %) | < 0.001 | ||
| Headache | 16,903 (10.1 %) | 3936 (10.0 %) | 12,967 (10.1 %) | 0.503 | ||
| Traumatic brain injury | 225 (0.1 %) | 71 (0.2 %) | 154 (0.1 %) | 0.004 | ||
| Care Assessment Needs (CAN) score | 51.4 (27.4, 0–99) | 60.5 (10.2) | 63.4 (11.7) | < 0.001 | ||
| Hospitalization in the past year | 28,969 (17.3 %) | 7824 (19.9 %) | 21,145 (16.5 %) | < 0.001 | ||
| Social-Related Factors | ||||||
| Homelessness and marginally houseda (prior 2 years) | 7803 (4.7 %) | 3374 (4.7 %) | 4429 (3.5 %) | < 0.001 | ||
| Lack of social supporta (prior 2 years) | 1575 (0.9 %) | 556 (1.4 %) | 1019 (0.8 %) | < 0.001 | ||
| Substance-Related Conditions | ||||||
| Substance use disordersa (prior 2 years) | ||||||
| Alcohol use disorder | 18,601 (11.1 %) | 18,601 (47.3 %) | 0 (0.0 %) | < 0.001 | ||
| Cannabis use disorder | 7163 (4.3 %) | 7163 (18.2 %) | 0 (0.0 %) | < 0.001 | ||
| Cocaine use disorder | 2314 (1.4 %) | 1655 (4.2 %) | 659 (0.5 %) | < 0.001 | ||
| Opioid use disorder | 12,373 (7.4 %) | 4363 (11.1 %) | 8010 (6.3 %) | < 0.001 | ||
| Sedative use disorder | 1212 (0.7 %) | 843 (2.1 %) | 369 (0.3 %) | < 0.001 | ||
| Stimulant use disorder | 917 (0.5 %) | 917 (2.3 %) | 0 (0.0 %) | < 0.001 | ||
| Other drug use disorder | 6095 (3.6 %) | 3292 (8.4 %) | 2803 (2.2 %) | < 0.001 | ||
| Substance use, self-reported (past year) | ||||||
| Elevated AUDIT-C score(≥ 3 for women and ≥ 4 for men) | 6557 (3.9 %) | 6.557 (16.7 %) | 0 (0.0 %) | < 0.001 | ||
| Substance use, UDS-screened (past year) | ||||||
| Positive amphetamine | 1305 (0.8 %) | 1305 (3.3 %) | 0 (0.0 %) | < 0.001 | ||
| Positive benzodiazepine | 17,112 (10.2 %) | 6947 (17.7 %) | 10,165 (7.93 %) | < 0.001 | ||
| Positive cannabis | 13,870 (8.3 %) | 13,870 (35.2 %) | 0 (0.0 %) | < 0.001 | ||
| Positive cocaine | 787 (0.5 %) | 787 (2.0 %) | 0 (0.0 %) | < 0.001 | ||
| Psychoactive drugs (past 90 days) | ||||||
| Morphine milligram-equivalents | 224.5 (410.3, 0–20,400) | 230.9 (2.2) | 222.5 (1.1) | < 0.001 | ||
| Long-acting opioid | 49,718 (29.7 %) | 11,148 (28.3 %) | 38,570 (30.1 %) | < 0.001 | ||
| Benzodiazepine | 30,920 (18.5 %) | 7054 (17.9 %) | 23,866 (18.6 %) | 0.002 | ||
| GABA drugc | 50,347 (30.1 %) | 11,268 (28.6 %) | 39,079 (30.5 %) | < 0.001 | ||
| Muscle relaxant | 35,859 (21.4 %) | 8397 (21.3 %) | 27,462 (21.4 %) | 0.700 | ||
| Antidepressant | 57,312 (34.2 %) | 14,236 (36.2 %) | 43,076 (33.6 %) | < 0.001 | ||
| Antipsychotic | 12,269 (7.3 %) | 3971 (10.1 %) | 8298 (6.5 %) | < 0.001 | ||
| Sedatives | 13,145 (7.6 %) | 2987 (7.6 %) | 10,158 (7.9 %) | 0.030 | ||
| Smoking status | ||||||
| Current tobacco use | 75,350 (45.0 %) | 23,004 (58.4 %) | 52,346 (40.8 %) | < 0.001 | ||
| History of tobacco use | 36,735 (21.9 %) | 7046 (17.9 %) | 29,689 (23.2 %) | |||
| No history of tobacco use | 35,589 (21.2 %) | 5597 (14.2 %) | 29,992 (23.4 %) | |||
| Unknown status | 19,873 (11.9 %) | 1272 (3.2 %) | 5329 (4.2 %) | |||
a Diagnosed based on ICD-9 and ICD-10 codes
Statistical analysis
We assessed differences in the distribution of all-cause mortality and other causes of death across the different SU/SUD groups using chi-square tests for categorical variables and t-tests or Wilcoxon rank-sum tests for continuous variables, as appropriate. We estimated the association between SU/SUD and all-cause mortality using the Cox proportional hazards model. Since the proportional hazards assumption was violated, time-varying covariates were included in the model. For fatal overdose and traumatic deaths, we used the ref. Fine and Gray’s (1999) sub-distribution hazards model to account for competing risks (i.e., all-cause mortality not related to the primary event). All models included demographic covariates, smoking status, psychoactive drugs, CAN score, and hospitalizations within one year, in addition to variables for the different exposure groups. Hazard ratios (HR) and 95 % confidence intervals (CIs) were reported for each outcome. All analyses were conducted using STATA, v18.
Results
Baseline characteristics
A total of 167,547 Veterans were included in the study. The average age of Veterans on LTOT was 62.7 years (ranged from 21 to 117), predominantly male (93.7 %) and White (80.2 %) (Table 1). Over half were married (53.1 %). Common mental health diagnoses were depression (36.1 %), anxiety (20.3 %), and PTSD (10.4 %). More than two-thirds were diagnosed with back and spine disorders (70.2 %), followed by osteoarthritis (34.2 %) and neck and spine disorders (23.6 %).
Alcohol use disorder was the most prevalent substance-specific disorder in Veterans on LTOT (11.1 %), followed by opioid use disorder (7.4 %), cannabis use disorder (4.3 %), cocaine use disorder (1.4 %), sedative use disorder (0.7 %), and stimulant use disorder (0.5 %). About 3.9 % of Veterans on LTOT had risky alcohol use (based on AUDIT-C scores), 10.2 % of them had UDS-positive benzodiazepine use, and 8.3 % had UDS-positive cannabis use.
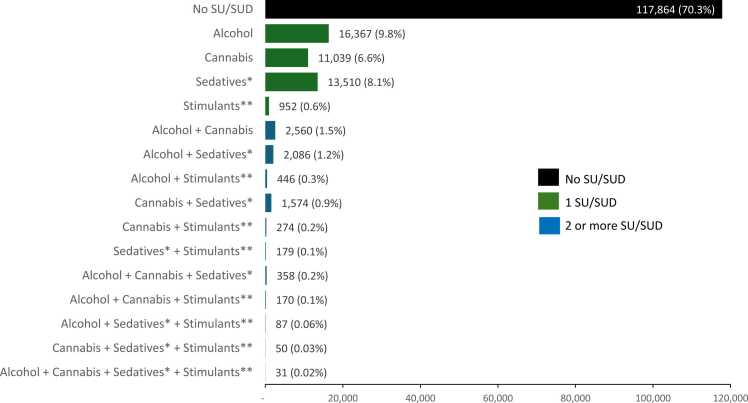
Combination of multiple substance use and disorders
The majority of Veterans (79.5 %) were on LTOT and did not have additional SU/SUD (LTOT-only group) (Fig. 1). Among Veterans with one additional SU/SUD, the most common substance group was alcohol (9.8 %), followed by sedative (8.1 %), cannabis (6.6 %), and stimulant (0.6 %). More complex combinations involving two or more substances or diagnoses were less common, with the most common combssinations being alcohol and cannabis (1.5 %), alcohol and sedatives (1.2 %), and cannabis and sedatives (0.9 %) (Fig. 2).
Prevalence of all-cause mortality and primary causes of deaths
Approximately, 1 out of 7 Veterans (14.2 %, n = 23,813 all-cause mortality) died within the follow-up period (Table 2). Mortality was higher among Veterans with alcohol (16.4 %) and sedative SU/SUD (15.0 %) compared to LTOT-only (14.2 %). The prevalence of fatal overdose was significantly higher among those co-using substances compared to the LTOT-only group (χ²=93.76, p < 0.001). Veterans with stimulant SU/SUD had the highest fatal overdose (0.74 %), followed by those with sedative (0.58 %), alcohol (0.48 %), and cannabis SU/SUD (0.24 %). For traumatic deaths, Veterans with stimulant SU/SUD had a higher prevalence of accidents or unintentional injuries (0.53 %) and homicide (0.11 %), while those with sedative SU/SUD had the highest prevalence of suicide (0.47 %) compared to Veterans not using any additional substances. Table 3
Table 2: Prevalence of all-cause mortality, fatal overdose, and traumatic deaths among veterans on long-term opioid therapy and multiple substance use and disorders between January 1, 2014, to December 31, 2019 (N = 167,547).
| Category (N, %) | Overall N (%) | No SU/SUD(n = 117,864) | Alcohola(n = 16,367) | Cannabisa(n = 11,039) | Sedativea(n = 13,510) | Stimulanta(n = 952) | Chi-square (X2), p values |
|---|---|---|---|---|---|---|---|
| All-Cause Mortality | 23,813 (14.20 %) | 17,868 (15.16 %) | 2685 (16.40 %) | 1129 (10.23 %) | 2020 (14.95 %) | 111 (11.66 %) | X2= 219.46,p < 0.001 |
| Fatal Overdose | 463 (0.28 %) | 272 (0.23 %) | 79 (0.48 %) | 27 (0.24 %) | 78 (0.58 %) | 7 (0.74 %) | X2= 93.76,p < 0.001 |
| Traumatic Deathsb | 1103 (0.66 %) | 763 (0.65 %) | 138 (0.84 %) | 63 (0.57 %) | 131 (0.97 %) | 8 (0.84 %) | X2= 29.95,p < 0.001 |
| Accidents/Unintentional Injuries | 691 (0.41 %) | 493 (0.42 %) | 90 (0.55 %) | 40 (0.36 %) | 63 (0.47 %) | 5 (0.53 %) | |
| Suicide | 376 (0.22 %) | 253 (0.21 %) | 38 (0.23 %) | 19 (0.17 %) | 64 (0.47 %) | 2 (0.21 %) | |
| Homicide | 36 (0.02 %) | 17 (0.01 %) | 10 (0.06 %) | 4 (0.04 %) | 4 (0.03 %) | 1 (0.11 %) |
SU/SUD = Substance use or substance use disorder
Table 3: Survival analysis for all-cause mortality (23,813), fatal overdose (n = 463), and traumatic deaths (n = 1103) among Veterans on long-term opioid therapy (LTOT) adjusted for demographics, smoking status, psychoactive drugs, care assessment needs score, and hospitalization within one year between January 1, 2014, to December 31, 2019.
| LTOT Categories | All-cause mortality(HR [95 % CI], p-value)a | Fatal overdose(HR [95 % CI], p-value)b | Traumatic deaths(HR [95 % CI], p-value)c |
|---|---|---|---|
| No SU/SUD | (reference) | (reference) | (reference) |
| Alcohold | 1.06 (0.99, 1.14), p = 0.073 | 1.43 (1.10, 1.86), p = 0.008 | 1.15 (0.96, 1.39), p = 0.140 |
| Cannabisd | 1.16 (1.03, 1.30), p = 0.018 | 1.09 (0.71, 1.65), p = 0.700 | 1.10 (0.85, 1.42), p = 0.484 |
| Sedatived | 1.29 (1.10, 1.52), p = 0.002 | 1.40 (1.04 1.91), p = 0.029 | 1.30 (1.05, 1.61), p = 0.017 |
| Stimulantd | 1.54 (1.17, 2.02), p = 0.002 | 3.29 (1.60, 6.77), p = 0.003 | 1.55 (0.76, 3.17), p = 0.229 |
HR = Hazard ratio; CI = Confidence intervals; SU/SUD = Substance use or substance use disorder
Notes: Results from competing risks model where non-overdose and non-traumatic deaths were treated as competing risks.
Survival analysis of all-cause mortality, fatal overdose, and traumatic deaths
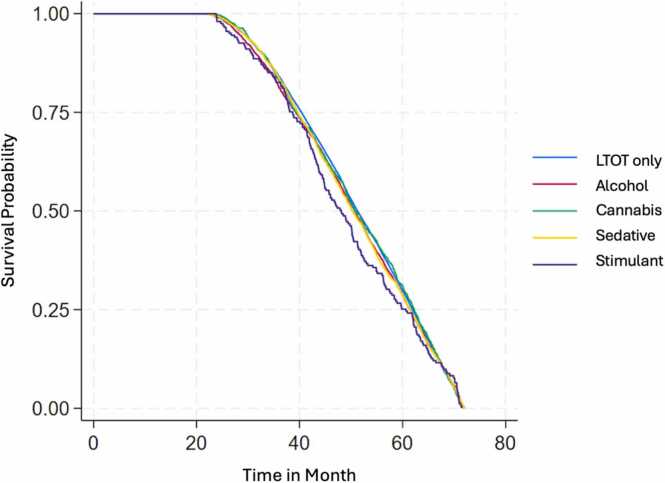
In the survival analysis, Veterans with cannabis (hazard ratio [HR]=1.16, 95 % CI=1.03, 1.30, p = 0.018), sedative (HR=1.29, 95 % CI=1.10, 1.52, p = 0.002), and stimulant SU/SUD (HR=1.54, 95 % CI=1.17, 2.02, p = 0.002) showed higher hazard rates of all-cause mortality compared to the LTOT-only group. For fatal overdose, Veterans with alcohol (HR=1.43, 95 % CI=1.10, 1.86, p = 0.008) and sedative SU/SUD (HR=1.40, 95 % CI=1.04, 1.91, p = 0.029) had a nearly 40 % higher hazard rate, while those with stimulant SU/SUD had more than triple the hazard rate (HR=3.29, 95 % CI=1.60, 6.77, p = 0.003) relative to the LTOT-only group. Additionally, Veterans with sedative SU/SUD showed a higher hazard rate of traumatic death compared to LTOT-only (HR=1.30, 95 % CI=1.05, 1.61, p = 0.017).
Discussion
This study addresses important gaps in the literature regarding substance co-use with opioids among Veterans on LTOT. Despite VA guidelines discouraging opioid use in patients with substance use disorders (SUDs) (ref. VA/VA/DoD Clinical Practice Guideline, T. L. G., 2022), we found one in four Veterans on LTOT either co-used one additional substance detectable by urine drug screen (UDS), reported risky alcohol consumption, or had a diagnosis of a SUD. Among these other substances, alcohol and sedative SU/SUD was the most common, followed by cannabis. Consistent with prior studies (ref. Hawkins et al., 2019, ref. National Academies of Sciences, Engineering, and Medicine, 2025, ref. Park et al., 2015), sedative SU/SUD was associated with higher mortality rates among Veterans on LTOT for all three outcomes, while Veterans with alcohol SU/SUD exhibited an increased mortality rate for fatal overdose relative to Veterans on LTOT who did not use other substances. Moreover, we found that cannabis and stimulant SU/SUD were associated with high rates of all-cause mortality, which to our knowledge is a novel finding specific to Veterans on LTOT. Cannabis use was associated with a 16 % increase in all-cause mortality rate, while stimulant use was associated with a 54 % increase in all-cause mortality rate and a threefold higher rate for fatal overdose.
While previous studies suggest that clinicians are more likely to discontinue opioids among Veterans with SUD (ref. Lovejoy et al., 2017, ref. Wyse et al., 2018), the high prevalence of substance co-use among Veterans on LTOT highlights the need for enhanced screening and harm reduction interventions to address co-occurring substance use patterns. This statistic is particularly concerning because Veterans on LTOT with a diagnosed SUD are more likely to follow riskier opioid trajectories (ref. Axson et al., 2024), increasing their risk for developing opioid use disorder (OUD). We also found that 7.4 % of Veterans on LTOT had an OUD diagnosis, which is lower than prior estimates suggesting that 41.3 % of Veterans on LTOT met criteria for lifetime OUD (ref. Boscarino et al., 2015). This difference is likely due to our study’s focus on OUD diagnoses within the past two years rather than a lifetime prevalence, as well as the exclusion of Veterans receiving treatment for OUD. While LTOT prescribing has declined, over 50 % of Veterans prescribed opioids remain on LTOT (ref. U.S. Department of Veterans Affairs, 2023). Given the risks of tapering LTOT, including overdose and mental health crises (ref. Agnoli et al., 2021) and the VA guidelines of addressing OUD before tapering, pain management should emphasize a patient-centered approach and shared decision-making to balance between risks and benefits of continued LTOT or tapering to discontinuation (ref. Himstreet et al., 2023).
The high prevalence of cannabis SU/SUD among Veterans on LTOT in our study is likely influenced by the increasing accessibility of cannabis due to the legalization of medical and recreational use in many states (ref. Schaeffer, 2024). Many patients now use cannabis for pain management (ref. Campbell et al., 2018), and some Veterans express interest in, or are actively using, cannabis to replace opioids (ref. Nevedal et al., 2023). However, both the VA and Centers for Disease Control and Prevention (CDC) offer limited guidance on prescribing cannabis for pain management and the approach providers should take to co-use of cannabis and prescription opioids. Moreover, there is a lack of evidence that cannabis is effective on managing pain, either as an adjunct or replacement to LTOT. For example, one study found that cannabis use was not associated with improved pain interference in Veterans on LTOT (ref. Becker et al., 2022). Furthermore, combining cannabis with opioids may increase short-term mortality risk, especially among older Veterans (ref. Keyhani et al., 2022b).
Stimulant SU/SUD among Veterans on LTOT is associated with significantly higher fatal overdose rates, which were three times higher than among Veterans with LTOT-only in our study. This finding aligns with national trends, which show a dramatic rise in opioid-involved stimulant overdoses–quadrupled between 2012 and 2018–and often involving heroin as well as synthetic and natural opioids (ref. Coughlin et al., 2022). Similarly, another study conducted in the general population reported a twofold increase in the hazard of fatal overdose for individuals combining stimulants with opioids compared to opioids alone (ref. Palis et al., 2022). While current VA guidelines recommend avoiding LTOT in patients with known SUD and advise against concurrent alcohol or benzodiazepine use (ref. VA/VA/DoD Clinical Practice Guideline, T. L. G., 2022), they do not provide specific recommendations for regular screening of substances like cannabis and stimulants. Expanding these guidelines to include routine screening for a broader range of substances is particularly urgent given the growing prevalence of cannabis use among Veterans on LTOT and the significant association between stimulant co-use and overdose.
In addition to cannabis and stimulants, our study confirmed the well-documented associations of alcohol and sedative SU/SUD in combination with opioids. Alcohol SU/SUD increased the rate of fatal overdose by 35 %, while sedative SU/SUD doubled the fatal overdose rate and was associated with a 30 % higher rate of traumatic deaths. These findings are consistent with prior research showing that co-prescription of benzodiazepines is associated with a higher mortality rates than opioids alone (ref. Gaither et al., 2016b, ref. Gaither et al., 2016a). Furthermore, hazardous alcohol consumption among LTOT patients is often under-addressed, as these patients are less likely to receive alcohol-related interventions compared to those not on opioids (ref. Joudrey et al., 2024). Veterans who co-use alcohol and opioids experience disproportionately high fatal overdose rates, with a particularly high prevalence among Black and Hispanic Veterans (ref. Lin, Bonar, et al., 2021). One study conducted in the general population found that individuals who co-use alcohol and opioids had a fatal opioid overdose rate of approximately 15 %, while those who co-use benzodiazepines with opioids had a rate exceeding 20 % in 2017 (ref. Tori et al., 2020). Although not specific to overdose-related deaths, another study found that individuals who co-use sedatives and opioids had a 20 % higher likelihood of experiencing an opioid overdose than those using opioids alone (ref. Cho et al., 2020).
Given the high prevalence of SUSUD among Veterans and the association between additional substance use and overdose, our findings suggest an urgent need to expand guidelines to include routine screening for a broader range of substances and substance use disorders. While risk mitigation strategies like urine drug screening (UDS) are widely utilized (ref. Sandbrink et al., 2020), there is variability in their implementation across VA facilities. This variation is influenced by differences in provider practices and patient preferences and lack of conclusive evidence supporting uniform protocols (ref. VA/VA/DoD Clinical Practice Guideline, T. L. G., 2022). These inconsistencies may hinder clinicians’ ability to standardize monitoring and adequately address the full spectrum of overdose risks. Clearer and more actionable guidance is needed, with specific recommendations on screening frequency, methods (biomarkers, self-report, or both), and follow-up interventions. Expanding and standardizing these screening protocols would enable earlier identification of high-risk behaviors, empowering clinicians to implement harm reduction measures tailored to the individual. Such measures could include tapering LTOT, increasing patient monitoring, and providing tailored substance use treatment. Future research should prioritize evaluating the effectiveness of these expanded protocols in reducing overdose risks, as well as assessing their feasibility and acceptability in clinical practice.
Since overdose risk is also shaped by physiological, psychological, and socio-structural factors, expanding harm reduction efforts should include services such as naloxone distribution, medications for opioid use disorder (MOUD), and holistic and complementary approaches (ref. Bennett et al., 2022). Additionally, peer-delivered interventions led by Veterans with lived experience of substance use may mitigate barriers to overdose prevention (ref. Ranney et al., 2024), particularly for socially isolated Veterans or those reluctant to engage with traditional healthcare services. Community-based care models are also important for Veterans who are not connected to VA, as nearly one-fifth of VA mental health patients receive community-based services that address major social determinants of health and co-occurring substance use disorders (ref. Bhalla et al., 2020).
Limitations
Our findings are subject to certain caveats. Although urine drug screens (UDS) provide objective substance use data, they may underestimate SU/SUD prevalence due intermittent use and co-use that go undetected, and 10 % of Veterans on LTOT may not have provided a UDS (ref. Sandbrink et al., 2020). Additionally, the lack of UDS frequency data per person limits assessment of screening consistency. We did not categorize opioid UDS results as consistent or aberrant relative to prescriptions or stratify by opioid dose (e.g., low-dose oxycodone), and thus our findings do not speak to issues of prescription adherence and potential diversion. We excluded Morphine Equivalent Daily Dose (MEDD) as an exposure due to extreme skewness from large methadone prescriptions, limiting dose-related risk assessment. There is likely under-assessment of SU/SUD. We also excluded patients prescribed buprenorphine due to its primary use for opioid use disorder, limiting generalizability to those treated with buprenorphine for chronic pain. We used a retrospective design, excluded patients in treatment for opioid use, and did not have measures for community substance use treatment outside the VA system, all of which affect documentation of SU/SUD in VA medical records. Our use of ICD-coded SUD diagnoses from the prior two years does not distinguish between active and past conditions, potentially misclassifying SUD status and underestimating active cases.
For alcohol use, VA clinician-administered screenings may underreport hazardous alcohol use compared to self-reports (ref. Bradley et al., 2011), as ICD-coded AUD diagnoses likely incorporate multiple assessments beyond AUDIT-C, making direct comparisons between screening-based and diagnostic measures challenging. Our classification of sedative SU/SUD relied on UDS-detected benzodiazepines and ICD-coded sedative use disorders, potentially underestimating use. The discrepancy between UDS-positive benzodiazepine use (10.2 %) and prescriptions (18.5 %) suggests variability in adherence, detection sensitivity, or UDS accuracy. Additionally, we could not distinguish prescribed from non-prescribed benzodiazepines. However, given the high prevalence of opioid and benzodiazepine co-prescribing (ref. Sun et al., 2017) and the ongoing misuse of these prescribed medications (ref. Taha et al., 2024), including prescribed benzodiazepines would allow for a more comprehensive assessment of their association with overdose. Our mutually exclusive SU/SUD categorization do not account for the co-occurrent of multiple types of SU/SUD in addition to LTOT, limiting the ability to isolate the effects of individual substances. Finally, our findings are specific to VA-enrolled Veterans, who have distinct demographic and health profiles, and may not generalize to non-Veteran populations.
Conclusion
Our study highlights the high prevalence of SU/SUD among patients on LTOT and the increased risk of early mortality for patients with co-occurring LTOT and SU/SUD. These findings support the need for comprehensive and systematic screening within VA to address co-occurring SU/SUD in this population. Healthcare providers and policymakers should consider incorporating routine screening for a broader range of substances and adopting evidence-based approaches to intervention and harm reduction. Future research should focus on evaluating standardized screening protocols and identifying tailored strategies to reduce overdose risk for patients on LTOT with SU/SUD. These efforts are crucial to improving outcomes for Veterans on LTOT and ensuring equitable, patient-centered care.
CRediT authorship contribution statement
Salomeh Keyhani: Writing – review & editing, Validation, Supervision, Funding acquisition. Thye Peng Ngo: Writing – review & editing, Writing – original draft, Methodology, Formal analysis, Conceptualization. Hoggatt Katherine J: Writing – review & editing, Validation, Supervision. Samuel Leonard: Writing – review & editing, Validation, Data curation.
Author disclosures
This work was supported by VA Health Services Research and Development IIR grant 18–231–2 from the US Department of Veterans Affairs Health Services. Support for US Department of Veterans Affairs (VA) and Centers for Medicare & Medicaid Services data was provided by the VA, VA Health Services Research and Development Service, and VA Information Resource Center (project numbers SDR 02–237 and 98–004)
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- A. Agnoli, G. Xing, D.J. Tancredi, E. Magnan, A. Jerant, J.J. Fenton. Association of dose tapering with overdose or mental health crisis among patients prescribed long-term opioids. JAMA, 2021. [DOI | PubMed]
- A. Athey, J. Overholser. A systematic review of suicide risk in veterans: depression is a more powerful predictor than comorbid psychiatric disorders. Mil. Behav. Health, 2018. [DOI]
- A.E. Austin, K.Y. Shiue, R.B. Naumann, M.C. Figgatt, C. Gest, M.E. Shanahan. Associations of housing stress with later substance use outcomes: a systematic review. Addict. Behav., 2021. [DOI]
- S.A. Axson, W.C. Becker, J.S. Merlin, K.A. Lorenz, A.M. Midboe, A. C. Black. Long-term opioid therapy trajectories in Veteran patients with and without substance use disorder. Addict. Behav., 2024. [DOI]
- W.C. Becker, Y. Li, E.C. Caniglia, R. Vickers-Smith, T. Feinberg, B.D.L. Marshall, E.J. Edelman. Cannabis use, pain interference, and prescription opioid receipt among persons with HIV: a target trial emulation study. AIDS Care, 2022. [DOI | PubMed]
- M.R. Begley, C. Ravindran, T. Peltzman, S.W. Morley, B.M. Stephens, L. Ashrafioun, J.F. McCarthy. Veteran drug overdose mortality, 2010–2019. Drug Alcohol Depend., 2022. [DOI]
- A.S. Bennett, H. Guarino, P.C. Britton, D. O’Brien-Mazza, S.H. Cook, F. Taveras, J. Cortez, L. Elliott. U.S. Military veterans and the opioid overdose crisis: a review of risk factors and prevention efforts. Ann. Med., 2022. [DOI | PubMed]
- I.P. Bhalla, E.A. Stefanovics, R.A. Rosenheck. Clinical epidemiology of single versus multiple substance use disorders: polysubstance use disorder. Med. Care, 2017. [PubMed]
- I.P. Bhalla, E.A. Stefanovics, R.A. Rosenheck. Social determinants of mental health care systems: intensive community based Care in the Veterans Health Administration. BMC Public Health, 2020. [DOI | PubMed]
- K.M. Bohnert, M.A. Ilgen, S. Louzon, J.F. McCarthy, I.R. Katz. Substance use disorders and the risk of suicide mortality among men and women in the US Veterans Health Administration. Addiction, 2017. [DOI | PubMed]
- J.A. Boscarino, S.N. Hoffman, J.J. Han. Opioid-use disorder among patients on long-term opioid therapy: impact of final DSM-5 diagnostic criteria on prevalence and correlates. Subst. Abus. Rehabil., 2015. [DOI]
- K.A. Bradley, K.R. Bush, M.B. McDonell, T. Malone, S.D. Fihn, the Ambulatory Care Quality Improvement Project (ACQUIP). Screening for problem drinking. J. Gen. Intern. Med., 1998. [DOI]
- K.A. Bradley, G.T. Lapham, E.J. Hawkins, C.E. Achtmeyer, E.C. Williams, R.M. Thomas, D.R. Kivlahan. Quality concerns with routine alcohol screening in VA clinical settings. J. Gen. Intern. Med., 2011. [DOI | PubMed]
- K. Bush, D.R. Kivlahan, M.B. McDonell, S.D. Fihn, K.A. Bradley, for the Ambulatory Care Quality Improvement Project (ACQUIP). The AUDIT alcohol consumption questions (AUDIT-C): an effective brief screening test for problem drinking. Arch. Intern. Med., 1998. [DOI | PubMed]
- G. Campbell, W.D. Hall, A. Peacock, N. Lintzeris, R. Bruno, B. Larance, S. Nielsen, M. Cohen, G. Chan, R.P. Mattick, F. Blyth, M. Shanahan, T. Dobbins, M. Farrell, L. Degenhardt. Effect of cannabis use in people with chronic non-cancer pain prescribed opioids: findings from a 4-year prospective cohort study. Lancet Public Health, 2018. [DOI | PubMed]
- Centers for Disease Control and Prevention. 2023. State Unintentional Drug Overdose Reporting System (SUDORS). https://www.cdc.gov/drugoverdose/fatal/dashboard/index.html
- J. Cho, M.M. Spence, F. Niu, R.L. Hui, P. Gray, S. Steinberg. Risk of overdose with exposure to prescription opioids, benzodiazepines, and non-benzodiazepine sedative-hypnotics in adults: a retrospective cohort study. J. Gen. Intern. Med., 2020. [DOI | PubMed]
- L.N. Coughlin, L. Zhang, A.S.B. Bohnert, D.T. Maust, J. Goldstick, L. ( A. Lin. Patient characteristics and treatment utilization in fatal stimulant-involved overdoses in the United States Veterans Health Administration. Addiction, 2022. [DOI | PubMed]
- Y.S. Cypel, D. DePhilippis, V.J. Davey. Substance use in U.S. Vietnam War era veterans and nonveterans: results from the Vietnam era health retrospective observational study. Subst. Use Misuse, 2023. [DOI | PubMed]
- S.K. Dobscha, B.J. Morasco, J.P. Duckart, T. Macey, R.A. Deyo. Correlates of prescription opioid initiation and long-term opioid use in Veterans with persistent pain. Clin. J. Pain., 2013. [DOI | PubMed]
- D. Dowell, K.R. Ragan, C.M. Jones, G.T. Baldwin, R. Chou. CDC clinical practice guideline for prescribing opioids for pain—United States, 2022. Mmwr. Recomm. Rep., 2022. [DOI]
- Fihn, S.D., & Box, T. 2013. Care Assessment Need (CAN) score and the Patient Care Assessment System (PCAS): Tools for Care Management. Office of Informatics and Analytics, Veterans Health Administration. https://www.hsrd.research.va.gov/for_researchers/cyber_seminars/archives/713-notes.pdf
- Fihn, S.D., & Box, T. 2016. Update on the Care Assessment Need Score–CAN 2.0 and the Patient Care Assessment System (PCAS). Office of Analytics and Business Intelligence, Veterans Health Administration. https://www.hsrd.research.va.gov/for_researchers/cyber_seminars/archives/1088-notes.pdf
- J.P. Fine, R.J. Gray. A proportional hazards model for the subdistribution of a competing risk. J. Am. Stat. Assoc., 1999. [DOI]
- J.R. Gaither, J.L. Goulet, W.C. Becker, S. Crystal, E.J. Edelman, K. Gordon, R.D. Kerns, D. Rimland, M. Skanderson, A.C. Justice, D.A. Fiellin. The effect of substance use disorders on the association between guideline-concordant long-term opioid therapy and all-cause mortality. J. Addict. Med., 2016. [DOI | PubMed]
- J.R. Gaither, J.L. Goulet, W.C. Becker, S. Crystal, E.J. Edelman, K. Gordon, R.D. Kerns, D. Rimland, M. Skanderson, A.C. Justice, D.A. Fiellin. The association between receipt of guideline-concordant long-term opioid therapy and all-cause mortality. J. Gen. Intern. Med., 2016. [DOI | PubMed]
- R.M. Gladden, J. O’Donnell, C.L. Mattson, P. Seth. Changes in opioid-involved overdose deaths by opioid type and presence of benzodiazepines, cocaine, and methamphetamine—25 states, July–December 2017 to January–June 2018. Mmwr. Morb. Mortal. Wkly. Rep., 2019. [DOI]
- E.J. Hawkins, S.B. Goldberg, C.A. Malte, A.J. Saxon. New coprescription of opioids and benzodiazepines and mortality among Veterans Affairs patients with posttraumatic stress disorder. J. Clin. Psychiatry, 2019. [DOI]
- C.J. Hayes, L.E. Gressler, B. Hu, B.L. Jones, J.S. Williams, B.C. Martin. Trajectories of opioid coverage after long-term opioid therapy initiation among a national cohort of US veterans. J. Pain. Res., 2021. [DOI | PubMed]
- Himstreet, J.E., Wells, D.L., & Popish, S.J. 2023. Opioid deprescribing discussion tool: A VA clinician guide (pp. 1–25). U.S. Department of Veterans Affairs,. https://www.pbm.va.gov/PBM/AcademicDetailingService/Documents/508/10-1548_PAIN_OpioidDeprescribingDiscussionTool_P97068.pdf
- P.J. Joudrey, M. Wang, E. DeRycke, E.C. Williams, E.J. Edelman. Alcohol-related care among veterans with unhealthy alcohol use: the role of long-term opioid therapy receipt. J. Addict. Med., 2024. [DOI | PubMed]
- R.N. Karmali, C. Bush, S.R. Raman, C.I. Campbell, A.C. Skinner, A.W. Roberts. Long-term opioid therapy definitions and predictors: a systematic review. Pharmacoepidemiol. Drug Saf., 2020. [DOI | PubMed]
- S. Keyhani, S. Leonard, A.L. Byers, T. Zaman, E. Krebs, P.C. Austin, T. Moss-Vazquez, C. Austin, F. Sandbrink, D.M. Bravata. Association of a positive drug screening for cannabis with mortality and hospital visits among Veterans Affairs enrollees prescribed opioids. JAMA Netw. Open, 2022. [DOI]
- S. Keyhani, S. Leonard, A.L. Byers, T. Zaman, E. Krebs, P.C. Austin, T. Moss-Vazquez, C. Austin, F. Sandbrink, D.M. Bravata. Association of a positive drug screening for cannabis with mortality and hospital visits among veterans affairs enrollees prescribed opioids. JAMA Netw. Open, 2022. [DOI]
- C.-W. Lan, D.A. Fiellin, D.T. Barry, K.J. Bryant, A.J. Gordon, E.J. Edelman, J.R. Gaither, S.A. Maisto, B.D.L. Marshall. The epidemiology of substance use disorders in US Veterans: a systematic review and analysis of assessment methods. Am. J. Addict., 2016. [DOI | PubMed]
- D. Lewer, J. Freer, E. King, S. Larney, L. Degenhardt, E.J. Tweed, V.D. Hope, M. Harris, T. Millar, A. Hayward, D. Ciccarone, K.I. Morley. Frequency of health-care utilization by adults who use illicit drugs: a systematic review and meta-analysis. Addiction, 2020. [DOI | PubMed]
- M. Li, C. Peterson, L. Xu, C.A. Mikosz, F. Luo. Medical costs of substance use disorders in the US employer-sponsored insurance population. JAMA Netw. Open, 2023. [DOI]
- L.A. Lin, A.S.B. Bohnert, F.C. Blow, A.J. Gordon, R.V. Ignacio, H.M. Kim, M.A. Ilgen. Polysubstance use and association with opioid use disorder treatment in the US Veterans Health Administration. Addiction, 2021. [DOI | PubMed]
- L.A. Lin, E.E. Bonar, L. Zhang, R. Girard, L. Coughlin. Alcohol-involved overdose deaths in US Veterans. Drug Alcohol Depend., 2021. [DOI]
- T.I. Lovejoy, B.J. Morasco, M.I. Demidenko, T.H.A. Meath, J.W. Frank, S.K. Dobscha. Reasons for discontinuation of long-term opioid therapy in patients with and without substance use disorders. PAIN, 2017. [DOI | PubMed]
- National Academies of Sciences, Engineering, and Medicine. 2025. [DOI]
- A.L. Nevedal, C. Timko, M.C. Lor, K.J. Hoggatt. Patient and provider perspectives on benefits and harms of continuing, tapering, and discontinuing long-term opioid therapy. J. Gen. Intern. Med., 2023. [DOI | PubMed]
- H. Palis, C. Xavier, S. Dobrer, R. Desai, K. Sedgemore, M. Scow, K. Lock, W. Gan, A. Slaunwhite. Concurrent use of opioids and stimulants and risk of fatal overdose: a cohort study. BMC Public Health, 2022. [DOI | PubMed]
- T.W. Park, R. Saitz, D. Ganoczy, M.A. Ilgen, A.S.B. Bohnert. Benzodiazepine prescribing patterns and deaths from drug overdose among US veterans receiving opioid analgesics: case-cohort study. BMJ, 2015. [DOI]
- Ranney, R.M., Goldberg, R., Maguen, S., & Blonigen, D. 2024. Peer specialist-led interventions in primary care at the Veterans Health Administration: An integrative review. Psychological Services, No Pagination Specified-No Pagination Specified. https://doi.org/10.1037/ser0000858
- F. Sandbrink, E.M. Oliva, T.L. McMullen, A.R. Aylor, M.A. Harvey, M.L. Christopher, F. Cunningham, T. Minegishi, T. Emmendorfer, J.M. Perry. Opioid prescribing and opioid risk mitigation strategies in the Veterans Health Administration. J. Gen. Intern. Med., 2020. [DOI | PubMed]
- Schaeffer, K. 2024. 9 facts about Americans and marijuana. Pew Research Center. https://www.pewresearch.org/short-reads/2024/04/10/facts-about-marijuana/
- Substance Abuse and Mental Health Services Administration. 2023. Key substance use and mental health indicators in the United States: Results from the 2022 National Survey on Drug Use and Health. https://www.samhsa.gov/data/release/2022-national-survey-drug-use-and-health-nsduh-releases
- E.C. Sun, A. Dixit, K. Humphreys, B.D. Darnall, L.C. Baker, S. Mackey. Association between concurrent use of prescription opioids and benzodiazepines and overdose: retrospective analysis. BMJ, 2017. [DOI]
- S.A. Taha, J.R. Westra, D.H. Tacker, M.A. Raji, Y.-F. Kuo. Trends in co-prescribed opioids and benzodiazepines, non-prescribed opioids and benzodiazepines, and schedule-I drugs in the United States, 2013 to 2019. Prev. Med. Rep., 2024. [DOI]
- M.E. Tori, M.R. Larochelle, T.S. Naimi. Alcohol or benzodiazepine co-involvement with opioid overdose deaths in the United States, 1999-2017. JAMA Netw. Open, 2020. [DOI]
- U.S. Department of Veterans Affairs. 2023. VA reduces number of Veterans prescribed opioids by 67% since 2012 [VA News]. https://news.va.gov/press-room/va-reduces-opioids-by-67-since-2012/
- VA/DoD Clinical Practice Guideline, T. L. G 2022. VA/DOD Clinical Practice Guideline for the use of opioids in the management of chronic pain. U.S. Government Printing Office.
- K.E. Vowles, Z.S. Schmidt, C.G. Ford. Opioid and alcohol misuse in Veterans with chronic pain: a risk screening study. J. Pain., 2022. [DOI | PubMed]
- L. Wang, B. Porter, C. Maynard, G. Evans, C. Bryson, H. Sun, I. Gupta, E. Lowy, M. McDonell, K. Frisbee, C. Nielson, F. Kirkland, S.D. Fihn. Predicting risk of hospitalization or death among patients receiving primary care in the Veterans Health Administration. Med. Care, 2013. [PubMed]
- J.J. Wyse, B.J. Morasco, S.K. Dobscha, M.I. Demidenko, T.H.A. Meath, T.I. Lovejoy. Provider reasons for discontinuing long-term opioid therapy following aberrant urine drug tests differ based on the type of substance identified. J. Opioid Manag., 2018. [DOI | PubMed]
- B.J.H. Yarborough, S.P. Stumbo, A. Stoneburner, N. Smith, S.K. Dobscha, R.A. Deyo, B.J. Morasco. Correlates of benzodiazepine use and adverse outcomes among patients with chronic pain prescribed long-term opioid therapy. Pain. Med., 2019. [DOI | PubMed]
