EIF5A2 acts as a potential marker for prognosis and immunity in human cancers
Abstract
EIF5A2, a key member of the EIF family, has not been extensively studied regarding its role and mechanism in pan-cancer. TCGA and GTEx analyses were performed to investigate the differential expression of EIF5A2 in tumors and normal tissues. cBioportal was used to investigate the gene alterations of EIF5A2 in tumors. We used Cox regression and Kaplan–Meier analyses to discuss the impact of EIF5A2 expression on prognosis. Quantitative real-time PCR (qRT-PCR) was used to detect EIF5A2 mRNA levels in 12 cases of fresh liver hepatocellular carcinoma (LIHC) and corresponding adjacent non-tumor tissues. Immunohistochemistry (IHC) was employed to evaluate the expression levels of EIF5A2 in 284 cases of LIHC as well as in adjacent non-tumorous tissues. Furthermore, the study explored the association between EIF5A2 expression and various clinicopathological parameters, along with its prognostic implications. Spearman correlation analysis was used to estimate the relationship between EIF5A2 expression and tumor mutation burden (TMB), microsatellite instability (MSI), and immunologic features. “pRRophetic”R package was utilized to obtain the sensitivity of common drugs. The Gene Set Enrichment Analysis (GSEA) was used to study the functional enrichment analysis of EIF5A2-related genes. EIF5A2 was overexpressed in most tumors using TCGA and GTEx databases. Cox regression analysis demonstrated that high EIF5A2 expression was associated with unfavorable overall survival (OS) and disease-specific survival (DSS) in head and neck squamous cell carcinoma (HNSC), brain lower grade glioma (LGG), and LIHC. The frequency of EIF5A2 gene alterations was the highest in lung squamous cell carcinoma (LUSC). EIF5A2 expression was associated with TMB, MSI, and immune cell infiltration in some tumors. We performed IHC and qRT-PCR to evaluate EIF5A2 expression in HCC and normal tissues, and found upregulation of EIF5A2 expression at the mRNA and protein levels in LIHC. There was a correlation between EIF5A2 expression and tumor size, tumor grade, and TNM stage in LIHC. Kaplan–Meier survival suggested that the overexpression EIF5A2 group had unfavorable outcomes in LIHC. EIF5A2 expression was correlated with immune cell infiltration in LIHC. The high EIF5A2 expression group was more sensitive to cisplatin, crizontinib, gemcitabine, and nilotinib in LIHC. High EIF5A2 expression was associated with several pathways, including cell cycle, proteasome, DNA replication, primary immunodeficiency and oocyte meiosis. EIF5A2 may serve as a potential prognostic marker and a latent focus for cancer immunological treatment.
Article type: Research Article
Keywords: EIF5A2, Pan-cancer, Prognosis, Immunity
Affiliations: https://ror.org/026e9yy16grid.412521.10000 0004 1769 1119Department of Pathology, The Affiliated Hospital of Qingdao University, 16 Jiangsu Road, Qingdao, 266000 People’s Republic of China; https://ror.org/021cj6z65grid.410645.20000 0001 0455 0905Department of Pathology, School of Basic Medicine, Qingdao University, 308 Ningxia Road, Qingdao, 266071 People’s Republic of China; https://ror.org/013xs5b60grid.24696.3f0000 0004 0369 153XDepartment of Pathology, Beijing Ditan Hospital, Capital Medical University, No. 8 Jing Shun East Street, Chaoyang District, Beijing, 100015 China
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1007/s13205-025-04509-w | PubMed: 40964657 | PMC: PMC12436671
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.8 MB)
Introduction
Liver cancer is one of the five deadliest cancers globally and its incidence is increasing every year. Despite extensive research for decades, cancer continues to be a major global health problem (Prajapati et al. ref. 2025). This is particularly prevalent in developing countries (Siegel et al. ref. 2019; Starley et al. ref. 2010a; Wu et al. ref. 2019). Liver hepatocellular carcinoma (LIHC) is the most common histological subtype of liver cancer (Starley et al. 2010b). Most patients are identified with the disease in its later stages. Significant progress has been made in diagnosing diseases early through standard interventions, such as radiation, surgery, personalized strategies, and chemotherapy (Mishra et al ref. 2023a). However, postoperative recurrence and chemotherapy resistance lead to a poor overall prognosis. Therefore, identification of therapeutic targets is crucial for molecular-targeted therapy for cancer.
Eukaryotic translation initiation factor 5A2 (EIF5A2) was first sequenced and isolated from human chromosome 3q26 in 2000 (Llovet et al. ref. 2016; Guan et al. ref. 2001). The EIF5A family consists of two members (EIF5A1 and EIF5A2), both of which contain a special amino acid, 8-hydroxy-2, 7, 10-triamino-capric acid (hypusine), which is vital for post-translational modification and activation of EIF5A2 (Jenkins et al. ref. 2001). Unlike EIF5A1, which is typically expressed in normal humans, EIF5A2 has tissue-specific and cell-specific characteristics, and its mRNA is expressed only in specific tissues (such as testes, brains, and some malignant tumors) and their cells (Clement et al. ref. 2003). Research has shown that EIF5A2 is elevated in multiple cancers, including cervical, ovarian, colorectal, gastric, liver, melanoma, lung, nasopharyngeal, gallbladder, and esophageal squamous cell carcinoma (Wang et al. ref. 2013; Liu et al. ref. 2017; Yang et al. ref. 2009, ref. 2016a, ref. 2016b; Quanico et al. ref. 2017; Zhu et al. 2012; Meng et al. ref. 2015; Tang et al. ref. 2010; Cao et al. ref. 2017; Khosravi et al. ref. 2014, ref. 2016; Chen et al. ref. 2018; Huang et al. ref. 2016; Zheng et al. ref. 2020; Li et al. ref. 2014). In bladder cancer cells, EIF5A2 promotes DOX resistance, EIF5A2 causes DOX resistance through activation of the TGF-β pathway (Yang et al. ref. 2023). EIF5A2 upregulates metastasis-associated protein 1 (MTAl) through C-MYC, thereby inducing EMT and consequently enhancing the invasion and metastasis ability of colorectal cancer cells (Zhu et al. ref. 2011). EIF5A2 mRNA expression is significantly increased in LIHC (Yang et al. ref. 2016b), and EIF5A2 overexpression can lead to shorter survival times (Tang et al. ref. 2010). High expression of EIF5A2 enhances the migration and invasion ability of LIHC cells by inducing epithelial–mesenchymal transition (EMT) (Yang et al. ref. 2016b). In this study, EIF5A2 expression, prognosis, mutation, and its relationship with tumor mutation burden (TMB), microsatellite instability (MSI), immunity, and drug sensitivity were studied from a pan-cancer perspective.
Materials and methods
Data preparation
We obtained the expression data of 33 types of tumors from the TCGA (version: GDC2.0) and GTEx (version 8) databases in TPM format and Batch effects were addressed via ComBat. Then the differential expression of EIF5A2 in different cancers and normal tissues was discussed using the Wilcoxon test. Meanwhile, EIF5A2 expression in human cancers and matched normal tissue from TCGA database was studied by paired t test.
Correlative analysis of EIF5A2 expression with pathological stage in pan-cancer
To study whether EIF5A2 expression was linked with pathological stage in tumors, we acquired clinical features of 33 tumors from TCGA database. The Wilcoxon rank-sum test was used to investigate the correlation between EIF5A2 expression and clinicopathological characteristics.
Prognostic value of EIF5A2 in pan-cancer
Each tumor was classified into low- and high-expression groups. This study selected overall survival (OS) and disease-specific survival (DSS) as survival indicators through univariate Cox regression, and the results were presented in the form of a forest plot. Cox regression analysis was conducted using the survival package in the R language (version 4.2.1), and a forest map and Kaplan–Meier analysis were used for visualization.
Genes alterations, TMB, MSI analysis in pan-cancer
We used the cBioportal database (version 4.0) to analyze EIF5A2 gene alterations in pan-cancer. We used the R package maftools (version 2.8.05) to obtain tumor TMB and MSI from the literature and utilized Spearman’s correlation analysis to study the correlation between EIF5A2 expression and TMB and MSI in pan-cancer.
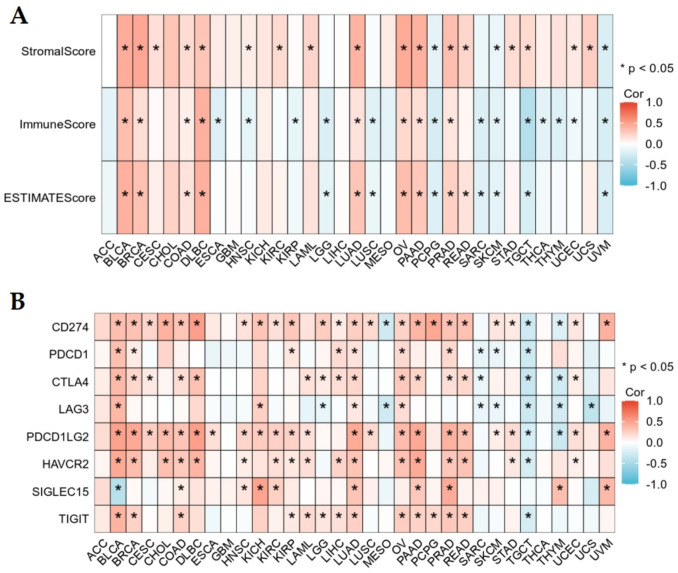
Correlative analysis of EIF5A2 with immunological features in pan-cancer
The relationship between EIF5A2 expression and the immune score, stromal score, and estimate score was studied using the ESTIMATE algorithm in cancers. Moreover, the correlation between EIF5A2 and the 24 types of immune cell infiltration was assessed using ssGSEA. Immunotherapy using immune checkpoint inhibitors (ICIs) has opened a new era in the treatment of malignant tumors. Therefore, it is necessary to investigate the link between EIF5A2 and immune checkpoints in tumors.
Immunohistochemical staining
We collected LIHC specimens and corresponding adjacent normal liver tissues from 284 patients surgically resected at the Affiliated Hospital of Qingdao University from February 2014 to October 2016. None of the patients had received radiotherapy or chemotherapy before surgery. Detailed patient demographics, inclusion/exclusion criteria, and clinicopathological data are summarized in Table 1. Patients were regularly followed up until April 29, 2020. There were 83 patients had complete follow-up data 5 years after surgery, and 31 of the patients died. The study was authorized by local institutional review boards (batch number: NO.ZYFYWZLL29132), and written informed consent was obtained from all patients before surgery. The tissues were collected, embedded, sliced at 4 μm, dewaxed with xylene, hydrated with gradient alcohol, soaked in 3%H2O2 for 20 min, and subjected to microwave repair (1 mmol/L EDTA, pH8.0). Rabbit monoclonal antibody EIF5A2 (EPR7412-50 cat.no.ab150439; 1:50, Abcam, UK) was added and incubated overnight at 4 °C. The secondary antibody (PV6000, Zhongshan Jinqiao Company) was followed by incubation for 30 min, color development by DAB, and tablet sealing. The slices were observed under a microscope. EIF5A2 expression was positive when a brownish-yellow or brown color appeared in the cytoplasm or nucleus. The evaluation criteria are described in the literature (He et al. ref. 2011), and the scoring criteria for positive cell staining intensity were 0 (negative), 1 (weak staining), 2 (medium intensity staining), and 3 (strong staining). The scoring criteria for the percentage of positive cells were as follows: 1 point (< 25%), 2 points (25–50%), 3 points (≥ 50–75%), and 4 points (≥ 75%). EIF5A2 immunohistochemical staining was based on the percentage of the staining intensity fraction multiplied by the positive cell fraction. A score ≤ 4 was considered as low expression, and > 4 was considered as high expression.
Table 1: Association between EIF5A2 and clinicolpathological features in LIHC using IHC
| Variables | n | Low expression | High expression | χ2 | P |
|---|---|---|---|---|---|
| Type | |||||
| Liver cancer | 284 | 107(37.68) | 177(62.32) | 20.48 | < 0.001 |
| Paracarcinoma | 284 | 273(96.13) | 11(3.87) | ||
| Gender | 1.914 | 0.167 | |||
| Male | 244 | 88 (36.07) | 156 (63.93) | ||
| Female | 40 | 19 (47.50) | 21 (52.50) | ||
| Age (years) | 3.102 | 0.078 | |||
| < 57 | 127 | 55 (43.31) | 72 (56.69) | ||
| ≥ 57 | 157 | 52 (33.12) | 105 (66.88) | ||
| AFP (ng/ml) | 0.017 | 0.896 | |||
| ≤ 20 | 126 | 48 (38.10) | 78 (61.90) | ||
| > 20 | 158 | 59 (37.34) | 99 (62.66) | ||
| Tumor size (cm) | 16.280 | < 0.001 | |||
| < 5 | 178 | 83 (46.07) | 95 (53.93) | ||
| ≥ 5 | 106 | 24 (24.53) | 82 (75.47) | ||
| Vascular invasion | 1.580 | 0.209 | |||
| No | 159 | 65 (40.88) | 94 (59.12) | ||
| Yes | 125 | 42 (33.60) | 83 (66.40) | ||
| Tumor differentiation | 5.523 | 0.019 | |||
| Well—moderate | 166 | 72 (43.37) | 94 (56.63) | ||
| Poor | 118 | 35 (29.66) | 83 (70.34) | ||
| TNM stage | 4.663 | 0.031 | |||
| I–II | 257 | 102 (29.96) | 155 (70.04) | ||
| III–IV | 27 | 5 (7.41) | 22 (92.59) |
Quantitative real-time PCR (qRT-PCR)
Twelve pairs of fresh LIHC and non-tumorous tissues were randomly collected from patients during surgical resection and stored at − 80 °C for qRT-PCR analysis. Total RNA was extracted using TRIzol, and its purity and concentration were measured using a UV spectrophotometer. Real-time fluorescent PCR was performed after reverse transcription to obtain cDNA. Three duplicate wells were set for each sample, and the reaction conditions were as follows: pre-denaturation at 95 °C for 30 s; denaturation at 95 °C for 5 s, annealing at 60 °C for 30 s, and extension at 72 °C for 30 s; a total of 40 cycles. Wilcoxon rank sum test was applied to study the differential expression between LIHC and normal tissues.
The following primers were used in this study:
EIF5A2: 5’-TATGCAGTGCTCGGCCTTG-3’.
Reverse: 5’-TTGGAACATCCATGTTGTGAGTAGA-3’.
GAPDH: 5’-GCACCGTCAAGGCTGAGAAC-3’.
Reverse: 5’-TGGTGAAGACGCCAGTGGA-3’.
Relationship between EIF5A2 and drug sensitivity in LIHC
To explore the application of EIF5A2 in clinical therapy, we used “pRRophetic” R package to obtain the sensitivity of common drugs. The half-inhibitory concentration (IC50) is an important indicator of the efficacy of drug therapy.
Functional enrichment analysis of EIF5A2-related genes in LIHC
The GSEA was used to study the functional enrichment analysis of EIF5A2-related genes. Significance was defined with thresholds of P < 0.05 and FDR < 0.25.
Statistical analysis
SPSS23.0 software was utilized to study the experimental data, and the difference in EIF5A2 expression between the two groups was explored by paired t test and Wilcoxon rank sum test. The relationship between EIF5A2 and clinicopathological parameters was examined using χ2 tests. Survival was assessed using the Kaplan–Meier survival curve and log-rank tests. Spearman correlative analysis was applied to discuss the relationship of EIF5A2 with immunity. Statistical significance was set at P < 0.05.
Results
Expression of EIF5A2 in 33 types of tumors
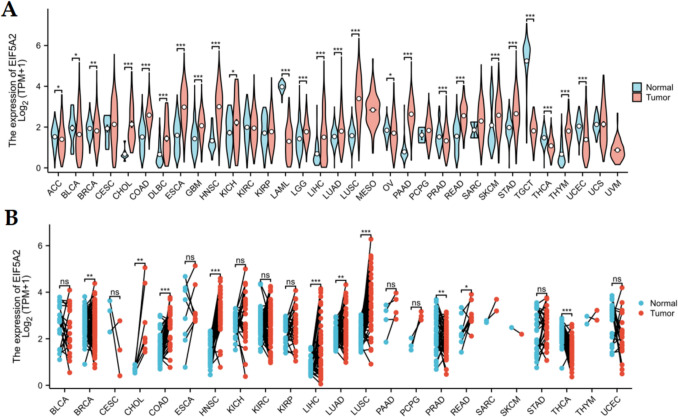
Using TCGA and GTEx databases, we found that EIF5A2 expression was elevated in 16 tumors relative to normal tissues, including CHOL, COAD, DLBC, ESCA, GBM, HNSC, KICH, LGG, LIHC, LUAD, LUSC, PAAD, READ, SKCM, STAD, and THYM, but decreased in 9 tumors, including ACC, BLCA, BRCA, LAML, OV, PRAD, TGCT, THCA, and UCEC (Fig. 1A). In addition, match paired analysis showed that EIF5A2 was overexpressed in CHOL, COAD, HNSC, LIHC, LUAD, LUSC, and READ and low expression in BRCA, PRAD, and THCA (Fig. 1B).
EIF5A2 expression was related to clinicopathologic stage in some tumors
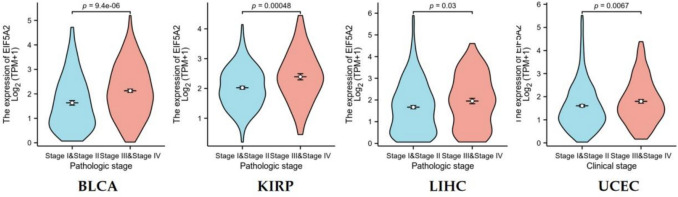
We selected pathological stage as a clinical index to explore its correlation with EFI5A2 expression. The results suggested that EIF5A2 expression increased in the advanced stages of BLCA, KIRP, LIHC, and UCEC (Fig. 2).
EIF5A2 overexpression suggests an unfavorable prognosis in several tumors
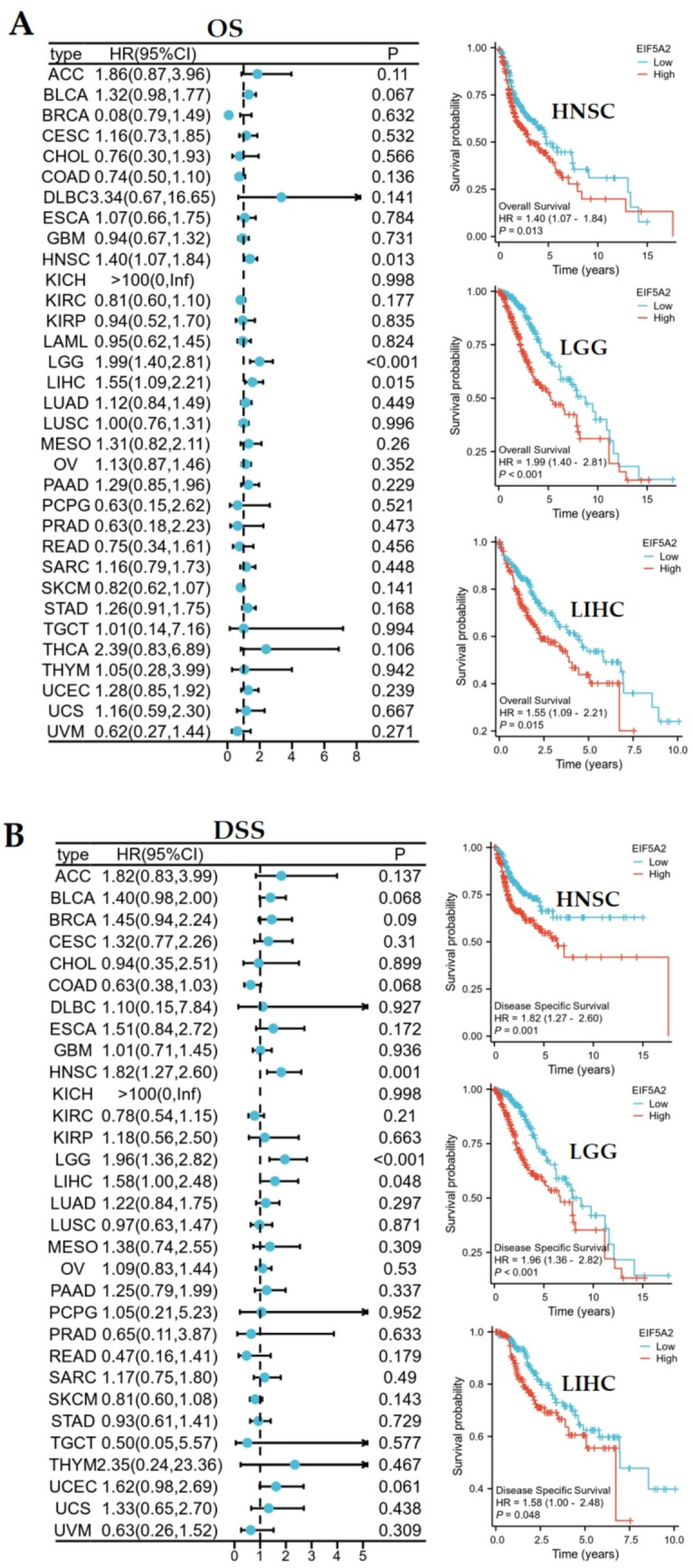
Overexpression of EIF5A2 is a risk factor for OS in HNSC (HR = 1.4, 95%CI 1.07–1.84, P = 0.013), LGG (HR = 1.99, 95%CI 1.40–2.81, P < 0.01), and LIHC (HR = 1.55, 95%CI 1.09–2.21, P = 0.015). Kaplan–Meier survival curves showed that patients with high EIF5A2 expression had shorter OSin HNSC, LGG, and LIHC (Fig. 3A). DSS analysis demonstrated that high EIF5A2 expression was a hazard index for HNSC (HR = 1.82, 95%CI 1.27–2.60, P = 0.001), LGG (HR = 1.96, 95%CI 1.36–2.82, P < 0.001), and LIHC (HR = 1.58, 95%CI 1.00–2.48, P = 0.048), and Kaplan–Meier analysis also revealed that overexpression of EIF5A2 had poor prognosis for these 3 tumors (Fig. 3B).
EIF5A2 genetic alteration, TMB, and MSI analysis in various tumors
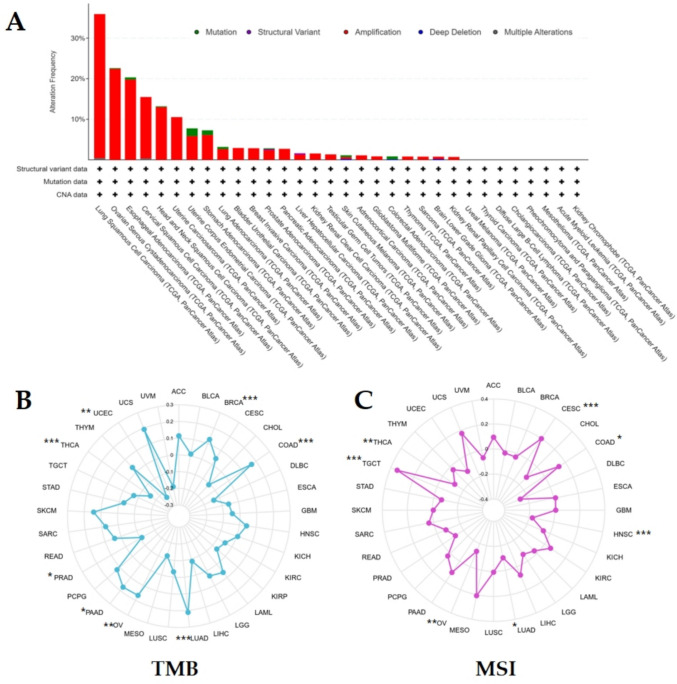
The cBioportal database showed that EIF5A2 was mainly present in various tumors in the form of amplification. LUSC showed the highest frequency of genetic changes, followed by OV and ESCA. No genetic alterations in EIF5A2 were observed in UVM, THCA, DLBC, CHOL, PCPG, MESO, LAML, and KICH (Fig. 4A). In addition, EIF5A2 expression was positively correlated with TMB in BRCA, COAD, LUAD, OV, and PAAD and negatively correlated with TMB in PRAD, THCA, and UCEC (Fig. 4B). MSI analysis demonstrated that EIF5A2 was positively correlated with MSI in CESC, COAD, and TGCT and negatively correlated with MSI in HNSC, LUAD, OV, and THCA (Fig. 4C).
The relationship of EIF5A2 expression with immunological features in pan-cancer
ESTIMATE analysis demonstrated a positive association between EIF5A2 expression and stromal score, immune score, and estimate score in BLCA, BRCA, COAD, DLBC, LUAD, OV, PAAD, and PRAD, and a negative association in SKCM and UVM. EIF5A2 expression was not correlated with three scores in ACC, CHOL, GBM, KICH, LIHC, and MESO (Fig. 5A). We examined the relationship between EIF5A2 and immune checkpoints using Spearman analysis and found that EIF5A2 was correlated with CD274, PDCD1LG2, and HAVCR2 in the majority of cancers and PDCD1, CTLA4, LAG3, SIGLEC15, and TIGIT in some tumors. EIF5A2 was positively linked to CD274, PDCD1, CTLA4, HAVCR2, and TIGIT in LIHC (Fig. 5B).
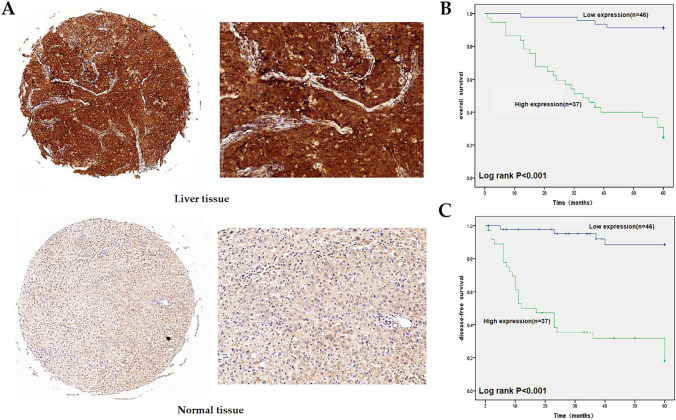
Expression of EIF5A2, prognosis and relationship with clinicopathology in LIHC by immunohistochemistry (IHC)
The IHC results indicated that EIF5A2 was expressed in both the cytoplasm and nucleus, and its expression was higher than that in normal liver tissue (Fig. 6A). In addition, EIF5A2 expression was associated with tumor size, tumor grade, and TNM stage in LIHC (P < 0.05), but not with sex, age, AFP level, and vascular invasion (Table 1). The median OS and DFS for the high-expression group were 34.7 months and 20.3 months, respectively, whereas the medians for the low-expression group were 61.7 months and 60.2 months. The group with high EIF5A2 expression had poor OS according to Kaplan–Meier survival analysis (Fig. 6B). The group with low EIF5A2 expression had better DFS than the group with high EIF5A2 expression (Fig. 6C).
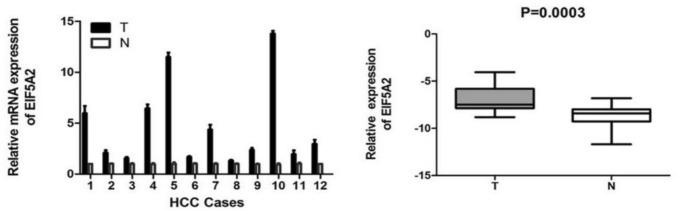
EIF5A2 was up-regulated at level of mRNA in LIHC
EIF5A2 mRNA expression in 12 LIHC and normal tissues was observed by qRT-PCR. The findings suggested that EIF5A2 mRNA expression was significantly higher in LIHC tissues than in normal tissues (t = 5.305, P = 0.0003) (Fig. 7).
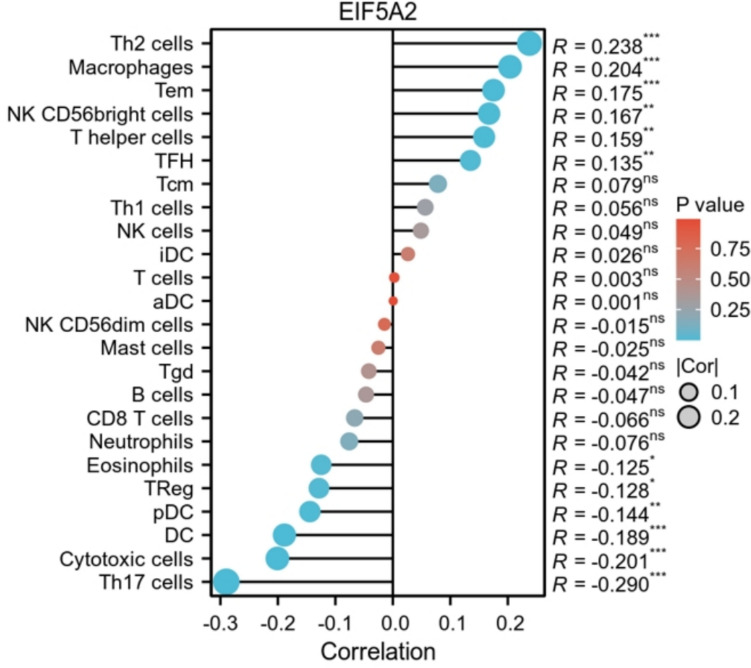
The relationship of EIF5A2 expression with immunity in LIHC
ssGSEA analysis demonstrated that EIF5A2 expression was positively linked with Th2 cells, macrophages, Tem, NK CD56 bright cells, T helper cells, and TFH, and negatively associated with Th17 cells, cytotoxic cells, DC, pDC, Tregs, and eosinophils (Fig. 8).
Relationship between EIF5A2 and drug sensitivity in LIHC
We used IC50 to assess chemotherapeutic drugs for LIHC. The lower the IC50 value of a drug, the more sensitive the patient is to the drug. The results showed that EIF5A2 expression had a negative correlation with the IC50 values of Cisplatin, Crizontinib, Gemcitabine, Nilotinib , suggesting that patients with high EIF5A2 expression were more susceptible to these drugs (Fig. 9).
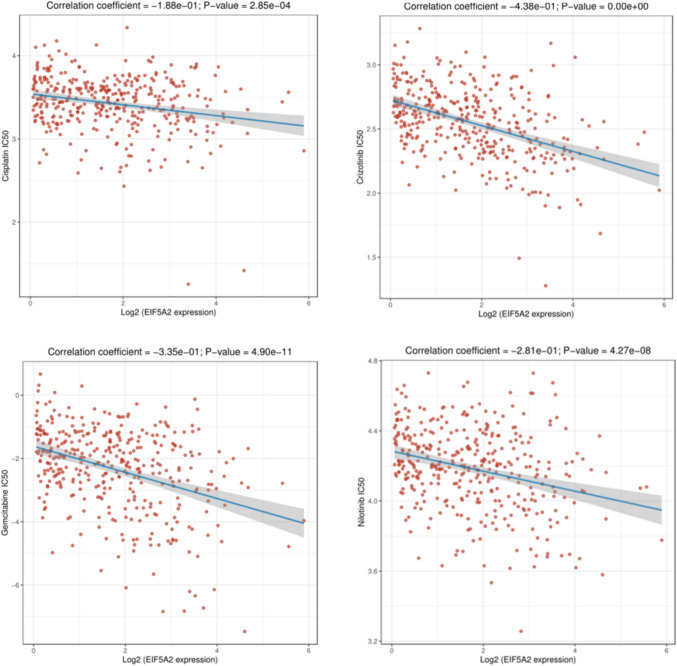
Function enrichment analysis of EIF5A2-related genes in LIHC and other tumors
To elucidate the bioprocesses involved in both groups, we utilized the KEGG set of GSEA for enrichment analysis. In LIHC, GSEA suggested that EIF5A2-related genes were involved in cell cycle, proteasome, DNA replication, primary immunodeficiency and oocyte meiosis. In most tumors, EIF5A2 was mostly enriched in ECM receptor interaction (Fig. 10).
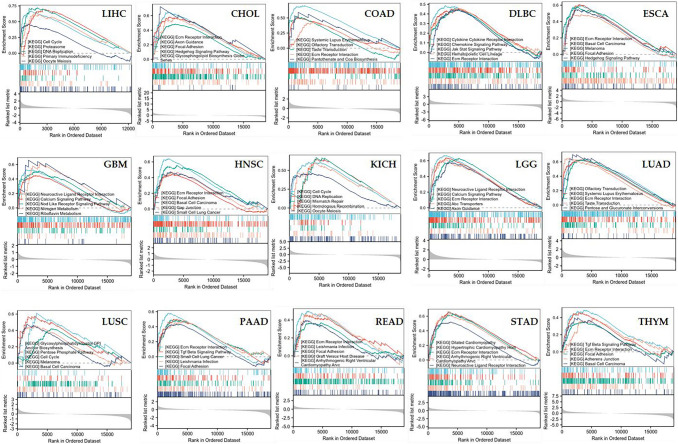
Discussion
Cancer is a severe disease that results in the loss of millions of lives each year (Mishra and et al ref. 2023b; Divya Mishra et al. ref. 2023), and according to the Global Cancer Observatory (GLOBOCAN) of the World Health Organization, a total of 18.08 million new cancer cases have been diagnosed worldwide in 2018, with liver cancer ranking fourth among men (600,000 cases) (Mattiuzzi and Lippi ref. 2019). In China, the incidence rate of liver cancer ranks second among men and fifth among women (Feng et al. ref. 2019). Since the early symptoms of LIHC patients are not typical, about half of the patients are in the middle and late periods when they visit a doctor, the recurrence and metastasis rates are high after surgery, and the prognosis is poor (Zeng et al. ref. 2018). Less than 15% of patients with early liver cancer have the opportunity to undergo liver resection, while advanced patients have to choose transcatheter arterial chemoembolization (TACE) and the oral chemotherapy drug sorafenib. However, it has limited curative effects and obvious drug resistance (Anwanwan et al. ref. 2020; El-Serag et al. ref. 2008). Molecular targeted therapy uses drugs to prevent cancerous cell growth, and the emergence of this therapy is a milestone (El-Serag et al. ref. 2008). At present, there is a pressing need to elucidate the pathogenic mechanisms of LIHC and identify the available targeting markers.
The EIF5A family consists of two members: EIF5A1 and EIF5A2. Recent evidence suggests that EIF5A1 is a shuttle protein present in the nucleus, cytoplasm, and mitochondria (Jao and Yu Chen ref. 2002; Wu et al. ref. 2020). Like the EIF5A1 protein, EIF5A2 protein is also a shuttle protein, and knockdown of expotin-4 led to nuclear accumulation of EIF5A2 protein, indicating that expotin-4 mediated EIF5A2 protein export from the nucleus (Zender et al. ref. 2008). As an oncogene, EIF5A2 can promote the growth, invasion, and metastasis of tumor cells (Tang et al. ref. 2010). Studies have demonstrated that EIF5A2 expression is elevated in ovarian cancer, cervical cancer, gastric cancer, lung cancer, liver cancer, and other cancers (Quanico et al. ref. 2017; Yang et al. ref. 2009, ref. 2016b, ref. 2023; Zhu et al. ref. 2011, 2012; Meng et al. ref. 2015; Tang et al. ref. 2010; Cao et al. ref. 2017; Khosravi et al. ref. 2014, ref. 2016; Chen et al. ref. 2018; Huang et al. ref. 2016; Zheng et al. ref. 2020; Li et al. ref. 2014), and plays a pivotal role in many biological processes, such as tumor formation, growth, and metastasis. Overexpression of EIF5A2 can induce EMT to promote liver cancer cell metastasis, and silencing of EIF5A2 can enhance the sensitivity to 5-fluorouracil (5-FU) in liver cancer cells by blocking the p38 MAPK and JNK/c-Jun/MMP-2 signaling pathways (Wang et al. ref. 2014). Knocking down EIF5A2 can reverse EMT by inhibiting ROS pathway, thereby inhibiting liver cancer cells’ invasion and metastasis (Yang et al. ref. 2016b; Liu et al. ref. 2016). Compared to normal gallbladder tissues, EIF5A2 expression is elevated in gallbladder cancer (Huang et al. ref. 2016).
We analyzed the expression, prognosis, and mutation of EIF5A2 and its relationship with TMB and MSI in pan-cancers. EIF5A2 expression was upregulated in 16 tumors and downregulated in 9 tumors by TCGA and GTEx. In addition, we detected EIF5A2 mRNA expression levels in 12 pairs of fresh HCC and corresponding non-tumor tissues by qRT-PCR and found that EIF5A2 mRNA levels were upregulated in LIHC, indicating that EIF5A2 participates in LIHC progression. Cox regression analysis showed that highly expressed EIF5A2 was associated with poor OS and DSS in HNSC, LGG, and LIHC. We applied Kaplan–Meier survival curve analysis and found that patients with high EIF5A2 expression had poorer OS and DFS. We also conducted a correlation analysis between EIF5A2 and clinicopathological characteristics and showed that EIF5A2 expression was correlated with tumor size, differentiation degree, and TNM stage, but not with gender, age, AFP level, and vascular invasion. These findings demonstrate that EIF5A2 is involved in LIHC progression and may be a latent prognostic biomarker for LIHC.
Tumorigenesis is a complicated process that involves genetic changes. Gene alterations included amplification, mutations, deep deletions, structural variants, and multiple alterations. The cBioportal database has shown that EIF5A2 exists primarily in amplified forms in different tumors. LUSC showed the highest frequency of gene changes, followed by OV and ESCA. TMB is an index used to evaluate the frequency of gene mutations, and has become a latent marker for predicting the efficacy of cancer immunotherapy in clinical practice. Previous studies have confirmed that a high tumor mutation load is positively correlated with immune treatment effects (Jiao et al. ref. 2019; Hellmann et al. ref. 2018). This study elucidated that EIF5A2 expression was positively correlated with TMB in 5 tumors, and negatively correlated with TMB in 3 tumors. This indicates that the high expression of EIF5A2 in BRCA, COAD, LUAD, OV, and PAAD has shown good efficacy in immunotherapy. MSI is defined as a type of high microsatellite mutation rate caused by MMR defects in phenotypes (Baretti and Le ref. 2018). Tumor cells are more prone to gene mutations due to MMR defects; therefore, MSI is one of the main factors in the development of tumor cells and is also a biological marker for predicting the effectiveness of immunotherapy (Lower et al. ref. 2018). In CESC, COAD, and TGCT, EIF5A2 expression was positively correlated with MSI and negatively correlated with HNSC, LUAD, OV, and THCA, indicating that high EIF5A2 expression in CESC, COAD and TGCT could benefit from immunotherapy. Tumors exhibiting high TMB and high MSI show enhanced responsiveness to immunotherapy (Filipovic et al. ref. 2020; Reck et al. ref. 2019). High EIF5A2 expression may indirectly increase TMB by regulating DNA damage repair-related genes, thereby generating more new antigens. However, it may also activate immune checkpoint molecules (such as PD-L1) to evade immune surveillance.
The tumor microenvironment (TME) is defined as the internal and external environment of tumor cells during tumor occurrence and metastasis. Immunocytes play an important role in TME. The results demonstrated that EIF5A2 expression was positively associated with stromal score, immune core, and estimate score in some tumors by ESTIMATE analysis. We used ssGSEA to discuss the relationship between EIF5A2 expression and immune cells in LIHC and revealed that EIF5A2 expression was positively correlated with Th2 cells, macrophages, Tem, NK CD56 bright cells, T helper cells, and TFH, and negatively correlated with Th17 cells, cytotoxic cells, DC, pDC, Tregs, and eosinophils. This suggests that immune cell interactions with tumor cells can lead to tumor development. Along with the basis and continuous breakthrough of clinical research, immunotherapies such as immune checkpoint inhibitors (ICI) based on T cells have shown good efficacy in cancers. In this study, EIF5A2 expression was positively associated with common immune checkpoints in some tumors. In addition, we conducted a correlation analysis of the drug sensitivity of EIF5A2 in LIHC. In LIHC, Cisplatin, Crizontinib, Gemcitabine and Nilotinib may have a good therapeutic effect in patients with high EIF5A2 expression. To clarify the biological process of EIF5A2 in LIHC, we conducted GSEA functional enrichment analysis and found that the group with high EIF5A2 expression participated in the following pathways: cell cycle, proteasome, DNA replication, primary immunodeficiency and oocyte meiosis.
In this study, we examined EIF5A2 expression, prognosis, clinicopathological characteristics, and immunity from a pan-cancer perspective. However, some limitations of this study should be considered. First, we only examined the overexpression of EIF5A2 at the mRNA and protein levels in LIHC. The biological mechanism of EIF5A2 involvement in LIHC has not yet been elucidated and requires further study. Second, confounding factors such as tumor heterogeneity or stromal contamination in bulk RNA-seq were not considered. Further investigation is warranted to elucidate the pathological significance of EIF5A2 across a diverse range of tumors.
Conclusion
Thus, EIF5A2 is a potential prognostic biomarker for cancer. Moreover, EIF5A2 expression was elevated at the mRNA and protein levels in LIHC tissues compared to that in normal tissues. Overexpression of EIF5A2 leads to unfavorable prognosis in some tumors. EIF5A2 expression correlates with immune cell infiltration and immune checkpoints in several cancers. This study sheds light on EIF5A2’s role in various tumors and serves as a latent biomarker for prognosis and immunotherapy.
References
- D Anwanwan, SK Singh, S Singh. Challenges in liver cancer and possible treatment approaches. Biochimica Et Biophysica Acta (BBA), 2020
- M Baretti, DT Le. DNA mismatch repair in cancer. Pharmacol Ther, 2018. [DOI | PubMed]
- TT Cao, SH Lin, L Fu. Eukaryotic translation initiation factor 5A2 promotes metabolic reprogramming in hepatocellular carcinoma cells. Carcinogenesis, 2017. [DOI | PubMed]
- C Chen, B Zhang, S Wu. Knockdown of EIF5A2 inhibits the malignant potential of non-small cell lung cancer cells. Oncol Lett, 2018. [PubMed]
- PM Clement, CA Henderson, ZA Jenkins. Identification and characterization of eukaryotic initiation factor 5a–2. Eur J Biochem, 2003. [DOI | PubMed]
- HB El-Serag, JA Marrero, L Rudolph. Diagnosis and treatment of hepatocellular carcinoma. Gastroenterology, 2008. [DOI | PubMed]
- RM Feng, YN Zong, SM Cao. Current cancer situation in China: good or bad news from the 2018 global cancer statistics?. Cancer Commun (Lond), 2019. [PubMed]
- A Filipovic, G Miller, J Bolen. Progress toward identifying exact proxies for predicting response to immunotherapies. Front Cell Dev Biol, 2020. [DOI | PubMed]
- XY Guan, JS Sham, TC Tang. Isolation of a novel candidate oncogene within a frequently amplified region at 3q26 in ovarian cancer. Can Res, 2001
- LR He, HY Zhao, BK Li. Overexpression of eIF5A-2 is an adverse prognostic marker of survival in stage I non-small cell lung cancer patients. Int J Cancer, 2011. [DOI | PubMed]
- MD Hellmann, T Ciuleanu, A Pluzanski. Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N Engl J Med, 2018. [DOI | PubMed]
- PY Huang, TT Zeng, X Ban. Expression of EIF5A2 associates with poor survival of nasopharyngeal carcinoma patients treated with induction chemotherapy. BMC Cancer, 2016. [DOI | PubMed]
- DL Jao, K Yu Chen. Subcellular localization of the hypusine-containing eukaryotic initiation factor 5A by immunofluorescent staining and green fluorescent protein tagging. J Cell Biochem, 2002. [DOI | PubMed]
- ZA Jenkins, PG HååG, HE Johansson. Human eIF5A2 on chromosome 3q25-q27 is a phylogenetically conserved vertebrate variant of eukaryotic translation initiation factor 5A with tissue-specific expression. Genomics, 2001. [DOI | PubMed]
- X-D Jiao, Xi He, B-D Qin. The prognostic value of tumor mutation burden in EGFR-mutant advanced lung adenocarcinoma,an analysis based on cBioPortal data base. J Thorac Dis, 2019. [DOI | PubMed]
- S Khosravi, RP Wong, GS Ardekani. Role of EIF5A2, a downstream target of Akt, in promoting melanoma cell invasion. Br J Cancer, 2014. [DOI | PubMed]
- S Khosravi, M Martinka, Y Zhou. Prognostic significance of the expression of nuclear eukaryotic translation initiation factor 5A2 in human melanoma. Oncol Lett, 2016. [DOI | PubMed]
- Y Li, L Fu, JB Li. Increased expression of EIF5A2, via hypoxia or gene amplification, contributes to metastasis and angiogenesis of esophageal squamous cell carcinoma. Gastroenterology, 2014. [DOI | PubMed]
- RR Liu, YS Lv, YX Tang. Eukaryotic translation initiation factor 5A2 regulates the migration and invasion of hepatocellular carcinoma cells via pathways involving reactive oxygen species. Oncotarget, 2016. [DOI | PubMed]
- X Liu, D Chen, J Liu. Blocking modification of eukaryotic Initiation 5A2 antagonizes cervical carcinoma via inhibition of RhoA/ROCK signal transduction pathway. Technol Cancer Res Treat, 2017. [DOI | PubMed]
- JM Llovet, J Zucman-Rossi, E Pikarsky. Hepatocellular carcinoma. Nat Rev Dis Primers, 2016. [DOI | PubMed]
- SS Lower, MP Mcgurk, AG Clark. Satellite DNA evolution: old ideas, new approaches. Curr Opin Genet Dev, 2018. [DOI | PubMed]
- C Mattiuzzi, G Lippi. Current cancer epidemiology. J Epidemiol Glob Health, 2019. [DOI | PubMed]
- QB Meng, WM Kang, JC Yu. Overexpression of eukaryotic translation initiation factor 5A2 (EIF5A2) correlates with cell aggressiveness and poor survival in gastric cancer. PLoS ONE, 2015. [DOI | PubMed]
- D Mishra, A Mishra, SN Rai, SK Singh. In silico insight to identify potential inhibitors of BUB1B from mushroom bioactive compounds to prevent breast cancer metastasis. Front Biosci (Landmark Ed), 2023. [DOI | PubMed]
- D Mishra, A Mishra, SN Rai, E Vamanu. Identification of prognostic biomarkers for suppressing tumorigenesis and metastasis of hepatocellular carcinoma through transcriptome analysis. Diagnostics, 2023. [DOI | PubMed]
- D Mishra, A Mishra, S Nand Rai, E Vamanu. Demystifying the role of prognostic biomarkers in breast cancer through integrated transcriptome and pathway enrichment analyses. Diagnostics, 2023. [DOI | PubMed]
- C Prajapati, SN Rai, AK Singh, BA Chopade, Y Singh. An update of fungal endophyte diversity and strategies for augmenting therapeutic potential of their potent metabolites: recent advancement. Appl Biochem Biotechnol, 2025. [DOI | PubMed]
- J Quanico, J Franck, T Cardon. NanoLC-MS coupling of liquid microjunction microextraction for on-tissue proteomic analysis. Biochim Biophys Acta Proteins Proteomics, 2017. [DOI | PubMed]
- M Reck, M Schenker, KH Lee, M Provencio, M Nishio, K Lesniewski-Kmak, R Sangha, S Ahmed, J Raimbourg, K Feeney. Nivolumab plus ipilimumab versus chemotherapy as first-line treatment in advanced non–small-cell lung cancer with high tumour mutational burden: patient-reported outcomes results from the randomised, open-label, phase III CheckMate 227 trial. Eur J Cancer, 2019. [DOI | PubMed]
- RL Siegel, KD Miller, A Jemal. Cancer statistics, 2019. CA Cancer J Clin, 2019. [PubMed]
- BQ Starley, CJ Calcagno, SA Harrison. Nonalcoholic fatty liver disease and hepatocellular carcinoma: a weighty connection. Hepatology, 2010. [DOI | PubMed]
- DJ Tang, SS Dong, NF Ma. Overexpression of eukaryotic initiation factor 5A2 enhances cell motility and promotes tumor metastasis in hepatocellular carcinoma. Hepatology, 2010. [DOI | PubMed]
- FW Wang, XY Guan, D Xie. Roles of eukaryotic initiation factor 5A2 in human cancer. Int J Biol Sci, 2013. [DOI | PubMed]
- FW Wang, MY Cai, SJ Mai. Ablation of EIF5A2 induces tumor vasculature remodeling and improves tumor response to chemotherapy via regulation of matrix metalloproteinase 2 expression. Oncotarget, 2014. [DOI | PubMed]
- C Wu, M Li, H Meng. Analysis of status and countermeasures of cancer incidence and mortality in China. Sci China Life Sci, 2019. [DOI | PubMed]
- GQ Wu, YM Xu, ATY Lau. Recent insights into eukaryotic translation initiation factors 5A1 and 5A2 and their roles in human health and disease. Cancer Cell Int, 2020. [DOI | PubMed]
- GF Yang, D Xie, JH Liu. Expression and amplification of eIF-5A2 in human epithelial ovarian tumors and overexpression of EIF-5A2 is a new independent predictor of outcome in patients with ovarian carcinoma. Gynecol Oncol, 2009. [DOI | PubMed]
- SS Yang, Y Gao, DY Wang. Overexpression of eukaryotic initiation factor 5A2 (EIF5A2) is associated with cancer progression and poor prognosis in patients with early-stage cervical cancer. Histopathology, 2016. [DOI | PubMed]
- Q Yang, Z Ye, Q Zhang. Expression of eukaryotic translation initiation factor 5A–2 (eIF5A-2) associated with poor survival in gastric cancer. Tumor Biol, 2016. [DOI]
- J Yang, X Jiang, Y Chen, L Teng. EIF5A2 promotes doxorubicin resistance in bladder cancer cells through the TGF-β signaling pathway. Discov Med, 2023. [DOI | PubMed]
- L Zender, W Xue, J Zuber. An oncogenomics-based in vivo RNAi screen identifies. tumor suppressors in liver cancer. Cell, 2008. [DOI | PubMed]
- H Zeng, W Chen, R Zheng. Changing cancer survival in China during 2003–15: a pooled analysis of 17 population-based cancer registries. Lancet Glob Health, 2018. [DOI]
- X Zheng, L Gao, BT Wang. Overexpression of EIF5A2 is associated with poor survival and aggressive tumor biology in gallbladder cancer. Histol Histopathol, 2020. [PubMed]
- W Zhu, MY Cai, ZT Tong. Overexpression of EIF5A2 promotes colorectal careinoma cell aggressiveness by upregnlating MTAl through C—MYC to induce epithelial-mesenchymahransition. Gut, 2011. [DOI | PubMed]
