Immunomodulatory Properties of Human Breast Milk: MicroRNA Contents and Potential Epigenetic Effects
Abstract
Infants who are exclusively breastfed in the first six months of age receive adequate nutrients, achieving optimal immune protection and growth. In addition to the known nutritional components of human breast milk (HBM), i.e., water, carbohydrates, fats and proteins, it is also a rich source of microRNAs, which impact epigenetic mechanisms. This comprehensive work presents an up-to-date overview of the immunomodulatory constituents of HBM, highlighting its content of circulating microRNAs. The epigenetic effects of HBM are discussed, especially those regulated by miRNAs. HBM contains more than 1400 microRNAs. The majority of these microRNAs originate from the lactating gland and are based on the remodeling of cells in the gland during breastfeeding. These miRNAs can affect epigenetic patterns by several mechanisms, including DNA methylation, histone modifications and RNA regulation, which could ultimately result in alterations in gene expressions. Therefore, the unique microRNA profile of HBM, including exosomal microRNAs, is implicated in the regulation of the genes responsible for a variety of immunological and physiological functions, such as FTO, INS, IGF1, NRF2, GLUT1 and FOXP3 genes. Hence, studying the HBM miRNA composition is important for improving the nutritional approaches for pregnancy and infant’s early life and preventing diseases that could occur in the future. Interestingly, the composition of miRNAs in HBM is affected by multiple factors, including diet, environmental and genetic factors.
Article type: Review Article
Keywords: breastfeeding, lactation, epigenetics, miRNA, RNA regulation, DNA methylation, histone modification
Affiliations: Department of Medical Laboratory Sciences, Faculty of Applied Medical Sciences, The Hashemite University, P.O. Box 330127, Zarqa 13133, Jordan; nada.nawaf@ymail.com; Department of Immunology, School of Medical Sciences, Universiti Sains Malaysia, Kubang Kerian, Kota Bharu 16150, Malaysia; alhatamleh@student.usm.my; Department of Clinical Nutrition and Dietetics, Faculty of Applied Medical Sciences, The Hashemite University, P.O. Box 330127, Zarqa 13133, Jordan; aminolaimat@hu.edu.jo (A.N.O.); murad@hu.edu.jo (M.A.A.-H.); Cell Therapy Center (CTC), The University of Jordan, Amman 11942, Jordan; walhan.alshaer@ju.edu.jo; Department of Pathology, Microbiology and Forensic Medicine, School of Medicine, The University of Jordan, Amman 11942, Jordan; hananyalu97@gmail.com; Faculty of Medicine, The Hashemite University, P.O. Box 330127, Zarqa 13133, Jordan; khaledalbakri1999@gmail.com; Department of Pharmaceutical Sciences, Faculty of Pharmacy, The University of Jordan, Amman 11942, Jordan; alkhafajienas@gmail.com; Department of Biology and Biotechnology, Faculty of Sciences, The Hashemite University, P.O. Box 330127, Zarqa 13133, Jordan; salim_dr_1954@hotmail.com; Department of Biomedical Sciences, College of Health Sciences, QU Health, Qatar University, Doha 2713, Qatar; aabdallah@qu.edu.qa
License: © 2022 by the authors. CC BY 4.0 Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.3390/biomedicines10061219 | PubMed: 35740242 | PMC: PMC9219990
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (7.7 MB)
1. Introduction
Since ancient times, human breast milk (HBM) has been considered the best food for newborn nutrition. Breastfeeding is the process of feeding a young child (under the age of two years) directly from a woman’s breasts during lactation [ref. 1]. A report from the World Health Organization (WHO) indicates that feeding for the first six months from mothers’ own milk without any extra external supplements is the optimal nutrition for infants to get benefits for immunity and health outcomes [ref. 2]. It is recommended that children continue breastfeeding along with supplemental food until two years of age [ref. 1]. Several studies have linked the exclusive breastfeeding duration to protective effects against a wide range of diseases in newborns. These include cardiorespiratory disorders [ref. 3], malocclusions [ref. 4], pediatric sleep-disordered breathing [ref. 5], parent-reported behavioral difficulties [ref. 5], gains and losses in weight [ref. 6], intelligence and neurocognitive behavior changes [ref. 7], type 1 diabetes (T1D) [ref. 8] as well as infant mortality [ref. 9]. Some civilizations and religions (such as Arab and Islam) use the term “milk kinship” [ref. 10,ref. 11]. It was shown that individuals breastfeeding from the same woman might cause consanguinity even in cases in which they are not related by genetic background. The consequences of marriage between these individuals are the same as consanguineous marriage and put children born of such marriage at risk for certain disorders [ref. 10,ref. 12]. Moreover, breastfeeding has many benefits for the physical and emotional health of breastfeeding mothers [ref. 13], such as improving uterine involution, reducing bleeding, preventing anemia [ref. 14,ref. 15], reducing the risk of postpartum depression by regulating cortisol circadian rhythms [ref. 16,ref. 17], decreasing the risk of type 2 diabetes (T2D), cardiovascular diseases, and metabolic syndrome, and protecting against breast cancer (BC) [ref. 18].
Extensive studies have been performed to investigate the composition of HBM. The reviews of these studies concluded that it contains bioactive substances, critical macro and micronutrients, and immune-active factors required to ensure children’s optimal growth; they consider it critical for the early development of neonatal immunity [ref. 19,ref. 20,ref. 21]. More specifically, HBM is mainly composed of around 87–88% water and solid components such as macronutrients. These include nearly 7% (60–70 g/L) carbohydrates that supply energy for growth, body functions and activity, are necessary for providing the building blocks of essential body compounds and shape the metabolic activities of the human gut microbiota. The macronutrients also include 3.8% (35–40 g/L) fats for visual and brain development and 1% (8–10 g/L) proteins as crucial building blocks for growth and development [ref. 22,ref. 23,ref. 24,ref. 25].
Moreover, HBM contains essential vitamins for growth, including fat-soluble vitamins such as vitamins A, E and K, as well as water-soluble vitamins such as vitamins C, B2, B3 and B5 [ref. 26,ref. 27]. HBM is also rich in minerals such as sodium, potassium, chloride, calcium, iron, zinc, copper, magnesium and selenium [ref. 28]. These essential minerals are required for many vital mechanisms such as building and maintaining strong bones and teeth, red blood cell production, regulation of immune system function, promoting proper fluid balance, muscle contraction, nerve transmission and protection of cells from oxidative damage [ref. 29,ref. 30,ref. 31,ref. 32,ref. 33]. Studies have also detected some hormones in HBM, including leptin, adiponectin, gonadotropin-releasing hormone (GnRH), insulin, estrogen, androgens, gastrin, progesterone, resistin and ghrelin [ref. 34,ref. 35,ref. 36,ref. 37,ref. 38]. Although the roles of these hormones are not completely clear, it is suggested that they are involved in several developmental and growth processes, protecting infants from some diseases (such as obesity) and may forestall the infant’s maturation until they are ready for reproduction [ref. 34,ref. 35,ref. 36,ref. 37,ref. 38]. Furthermore, HBM contains a variety of compounds with immunological properties (e.g., antimicrobial and antioxidant compounds), viable leukocytes (i.e., macrophages, neutrophils and lymphocytes) and a vast range of soluble and cellular factors (e.g., cytokines, chemokines and nucleotides) to facilitate the development and maturation of immune system and immune functioning [ref. 39,ref. 40]. HBM contains oligosaccharides (bifidus factor) that are thought to stimulate the growth of more than 200 desirable bacterial strains, the most important being probiotic bacteria, including Bacteroides spp., which play a vital function in the early stages of newborns’ gut colonization [ref. 41]; Lactobacillus spp., which ferment lactic acid bacteria (metabolize lactose and other simple sugars); and Bifidobacterium spp., which promote gut barrier function and modulate the immune system response [ref. 42].
Unlike other body fluids, HBM is extremely rich in circulating RNA molecules [ref. 43]. In the recent two decades, studies have detected a large number of circulating microRNAs (miRNAs) in HBM [ref. 44,ref. 45,ref. 46,ref. 47]. Almost 1400 miRNAs have been identified in HBM, and some of them have verified functions in the health and disease of infants [ref. 48]. The miRNAs in HBM have emerged as potential immune-regulatory agents targeting immune cells and influencing the development of an infant’s immune system through immune modulation [ref. 49,ref. 50]. For example, high quantities of miRNAs have been detected in HBM that have potential regulatory effects on the differentiation, maturation, activation and suppression of B and T cells, as well as monocyte development [ref. 51,ref. 52].
Moreover, miRNAs play a vital role in modulating the expression of a wide range of genes by targeting DNA methyltransferases without permanent changes in the gene sequences [ref. 53,ref. 54]. This process is called epigenetic regulation, and thus, miRNAs are considered epigenetic modulators. Furthermore, miRNAs affect the protein expressions of the target mRNAs resulting in RNA modifications [ref. 55,ref. 56], and they may affect histone modifications [ref. 57]. Although the epigenetic modifications from HBM miRNA transmission on future generations are not yet fully understood, recent research supports the idea that factors (i.e., miRNAs) modifying the epigenetic mechanisms may be transmitted by HBM and that these epigenetic changes may be transferred transgenerationally [ref. 12]. Therefore, studies on the HBM content of miRNAs and their potential epigenetic modifications are of vital importance. This work presents a comprehensive critical review of the immunomodulatory properties of HBM in breastfed infants, with emphasis on the potential roles of milk-derived miRNAs. In this up-to-date review, the available literature regarding miRNAs in HBM are reviewed, and the miRNA–mediated epigenetic regulations are discussed in general and from immunological aspects. This work should be of particular interest to researchers who are investigating HBM miRNAs, as well as to the general public. It may help ensure the quality of future research on epigenetic programming through breastmilk miRNAs.
2. The Physiological Basis of Breastfeeding and Milk Composition
The human breast is composed of mammary tissue, areola, nipple, fat, connective tissues, nerves, lymphatic and blood vessels. The mammary tissue contains the alveoli, which are the milk storage and factory, and the ducts that transport the produced HBM outside the body. The contraction of myoepithelium, muscle cells surrounding the alveoli, compresses the collected milk located inside the alveoli to move through the nine ducts to pass across the nipple—which also has nerves and muscle fibers [ref. 58,ref. 59]. Furthermore, Montgomery’s glands are present in the circular areola surrounding the nipple [ref. 59]. These glands attract the baby to suck from the mother’s breast by secreting a special scent; furthermore, they produce an oily fluid to protect the nipple’s skin during the suckling process [ref. 58]. Figure 1 shows the anatomical features of the female breast and the process of lactation.
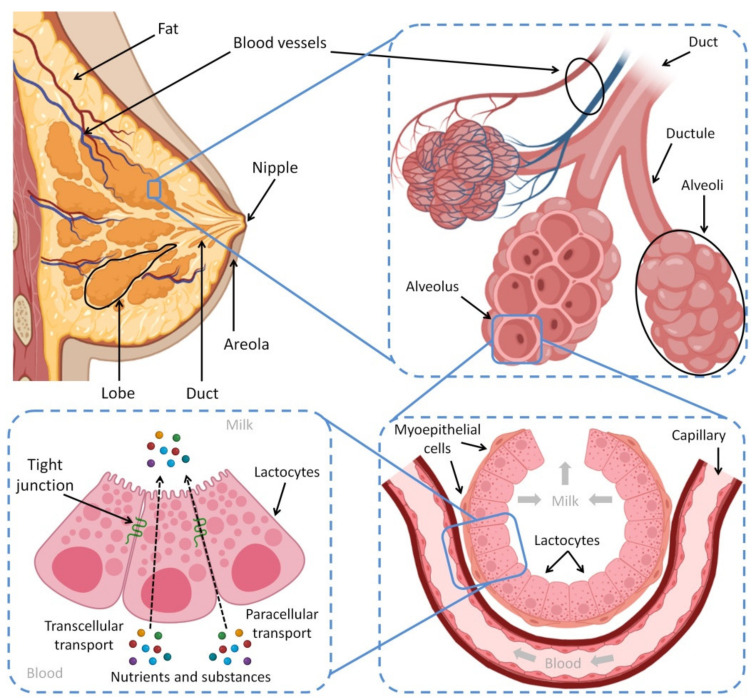
Oxytocin and prolactin are the most important hormones that have a direct impact on lactation, alongside other hormones like estrogen, which indirectly affect breastfeeding [ref. 63]. Suckling the nipple sends sensory impulses to the brain, which stimulates the pituitary gland to secrete oxytocin and prolactin from the posterior and anterior lobes, respectively [ref. 64]. The necessity of prolactin lies in the production of milk from the milk-producing cells present in the alveoli. During pregnancy, the blood’s prolactin levels continually increase, which ultimately initiate and control milk production in the mammary tissues [ref. 65]. Nevertheless, estrogen and progesterone prevent milk secretion by blocking the activity of prolactin. When their levels reduce after delivery, the secretion of milk begins [ref. 66]. Although prolactin is important for producing HBM, there is no longer a strong association between the amounts of milk and prolactin produced [ref. 67]. In fact, milk production will eventually stop once a mother stops breastfeeding; the rest of the produced milk will gradually dry up [ref. 68].
It is important to know that lactation at night is more helpful in preserving the supply of milk because prolactin production increases at night [ref. 69]. Oxytocin stimulates the contraction of myoepithelial cells around the alveoli. It pushes the existing milk inside the lumen of the alveoli to the mouth of the baby and facilitates feeding [ref. 70]. The reflex of oxytocin is conditioned to the feelings and sensations of the mother, such as seeing, smelling, and touching the baby. The oxytocin reflex could explain the importance of the baby being close to his or her mother and keeping skin-to-skin contact between them [ref. 71,ref. 72]. Moreover, lactation is influenced by the emotional changes of the mother; thus, an upset mood inhibits the oxytocin reflex, and otherwise, when the mother gets support and becomes more comfortable, the milk production and letdown will return [ref. 73].
3. HBM Composition
HBM is the standard for baby nutrition because it is filled with a variety of beneficial substances, including peptide growth factors, hormones [ref. 74], prebiotics and probiotics [ref. 75], enzymes, cytokines, chemokines, anti-inflammatory elements [ref. 76], antimicrobial proteins/peptides [ref. 77] and antioxidants [ref. 78]. HBM is a person-specific biofluid and includes various bioactive and nutritional components. Bioactive components are the ingredients that affect the body’s condition and function by influencing the biological elements and processes [ref. 79]. Different bioactive components are found in milk produced by different sources; some are synthesized by mammary epithelium, some are secreted by cells within the milk, and others come from maternal serum [ref. 19,ref. 80]. Additionally, milk fat globules secreted from mammary epithelium contain a variety of lipids and proteins [ref. 81]. Figure 2 summarizes the beneficial substances of HBM and the factors affecting its composition.
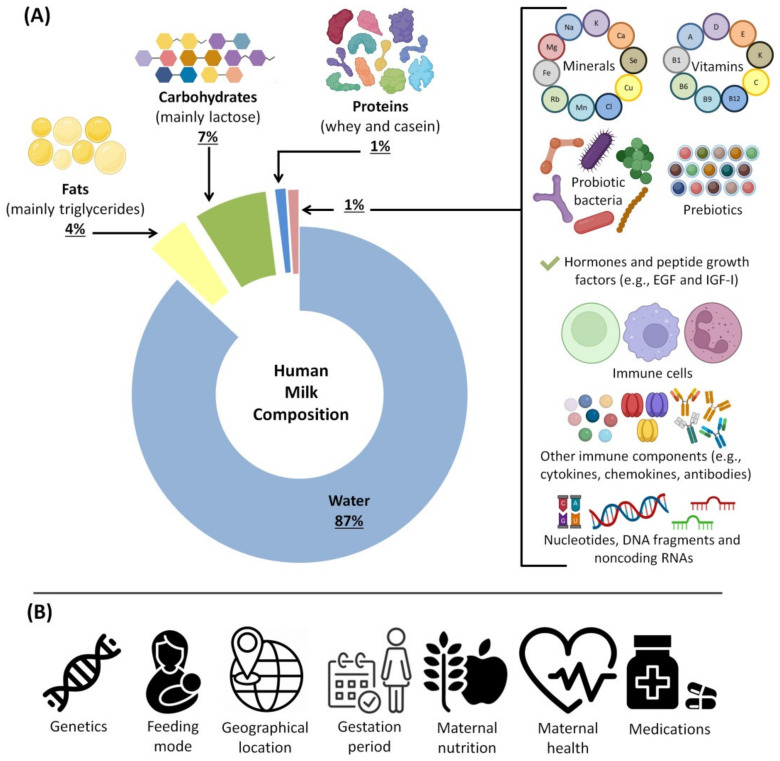
In HBM, fat provides approximately 50% of the total energy, while lactose provides 40% [ref. 83]. However, the composition of the milk is influenced by the length of gestation, lactation time, maternal diet, genotype, disease, age of the lactating baby and the onset of the menstrual cycle [ref. 87]. For instance, the fat content of preterm milk in early lactation is remarkably higher compared to that of term milk [ref. 88]. Furthermore, the average protein concentration gradually declines over the first six months and then stabilizes. Fat is associated with the mother’s diet and has a positive relationship with weight gain during pregnancy. Interestingly, the milk of the mother contains the required components for the development and growth of the infant, even if the mother’s diet is inadequate [ref. 83]. Moreover, the amount and nature of HBM components change over time to adapt to the varying needs of the infants’ development [ref. 83].
It is important to know the dynamic variability of HBM to reach the maximum benefits. For instance, it is thought that hindmilk enhances premature infants’ growth. It is also rich in lipids, providing the infant with the needs of energy and satiety. Therefore, mothers should completely empty one breast before feeding from the other [ref. 19]. Although the fat and protein contents vary regarding the conditions and time, the lactose concentration in mature milk is constant to preserve a stable osmotic pressure in the HBM. The absorption of calcium and other minerals is also mediated by lactose. Lactose attaches to many bioactive carbohydrates like oligosaccharides. Therefore, if there is not enough lactase in the small intestine, lactose malabsorption syndrome will develop. However, it was observed that exclusively breastfed infants rarely have lactase deficiency [ref. 83]. Furthermore, to enhance the health and growth of infants, milk components can be adjusted via dietary intake. For instance, maternal diet affects docosahexaenoic acid content. It is reported that 1 g of docosahexaenoic acid supplementation daily in maternal diet augments its level in HBM [ref. 89]. Furthermore, mother immunization significantly enhances the protective Igs levels in HBM and protects from influenza infection in both infant and mother [ref. 19].
4. Breastfeeding and Immunity
In 1891, researchers had shown that immunity was transferred from mother to newborn through breastfeeding [ref. 90], and in 1903, this was attributed to the presence of antibodies in mothers’ milk [ref. 91]. According to recent findings, HBM contains a rich supply of defensive elements that interact with one another, which serve as an innate immune response that protects against viruses [ref. 92,ref. 93]. Breastmilk’s innate immune system, in combination with the acquired immune system and intestinal flora, forms a strong component of the gut’s mucosal immunity, which protects breastfed newborns against microbial infections [ref. 92,ref. 94,ref. 95]. The incidence and mortality risks of pneumonia and diarrhea among completely breastfed infants are significantly lower than those who were fed with milk formula [ref. 96]. Further, breastfeeding has short- and long-term impacts such as optimal cognitive and behavioral development and protection against allergies and chronic diseases such as hypertension, obesity, diabetes, cardiovascular and autoimmune diseases [ref. 97]. Table 1 lists the most abundant immunomodulatory molecules in HBM and their main immunoregulatory functions.
Table 1: The main immunomodulators in HBM and their roles in improving the health and immune system.
| Component | Types | Major Immune-Related Functions | Reference |
|---|---|---|---|
| Fatty acids | – Monounsaturated (42%)- Medium-chain (42%)- Long-chain polyunsaturated (16%); linoleic acid (10%), arachidonic acid (1%), α-linolenic acid (>1%), eicosapentaenoic acid (0.1%), docosahexaenoic acid (0.4%), and others (3.7%) | – Maturation of immune system- Modulate the acquired immunological responses that affect the balance between Th1 and Th2 cells and Treg and Teff cells- Regulate the production of immunomodulatory cytokines (e.g., TGFs)- Enhance level of innate immune response (i.e., soluble CD14) and adaptative immune response (i.e., IgA)- Act as antiviral, antibacterial and antiprotozoal agents | [ref. 98,ref. 99,ref. 100,ref. 101] |
| Oligosaccharides | – Fucosylated (35% to 50%)- Sialylated (12–14%)- Nonfucosylated neutral (42–55%) | – Influence the expression of chemokines (e.g., CX3CL1, CCL5, CXCL2, CXCL3), cytokines (e.g., IL-4, IL-17C, IL-8, IL-1β, IL-10, IFN-γ), cellular receptors (IFNGR1), cell adhesion molecules (e.g., ICAM-1/2)- Reduce the infectivity of rotavirus, norovirus and influenza viruses- Exhibit antimicrobial and antibiofilm activities against S. agalactiae, E. coli, B. subtilis and S. aureus– Shape the gut microbiota in infants- Inhibit leukocyte adhesion to endothelial cells- Modulate TLR-4 signaling- Induce the production of cytokines (i.e., IL-4 and IFN-γ) required for expansion of Th1 and Th17 implicated in the pathogenesis of enterocolitis- Increase serum levels of IgG1 and IgG- Increase the expression of CD27 on splenic B-cells- Inhibit the adherence and binding of specific pathogens to the host cells in intestine | [ref. 19,ref. 102,ref. 103,ref. 104,ref. 105,ref. 106,ref. 107,ref. 108,ref. 109] |
| Hormones | Leptin, erythropoietin, adiponectin, ghrelin, IGFs, resistin and obestatin | – Erythropoietin prevents HIV transmission from mother to child- Adiponectin reduces inflammation, regulates infant metabolism (reducing later-life obesity) and inhibits production of TNF-α in intestinal epithelium and macrophages- Leptin regulates proinflammatory cytokines (e.g., TNF-α and IL-6) and Th1 responses; it also promotes proliferation and activation of monocytes and NK cells, neutrophils chemotaxis and T cell survival by modulating the expression of anti-apoptotic proteins (e.g., Bcl-xL)- IGFs play vital roles in the development and function of T cells- Resistin is involved in the anti-infection immune process by interacting with a variety of immune cells; can either directly or indirectly promote infiltration, adhesion and migration of monocytes, neutrophils and CD4+ T cells | [ref. 19,ref. 35,ref. 110,ref. 111,ref. 112,ref. 113] |
| Cells | Leukocytes (i.e., lymphocytes, neutrophils and macrophages), hematopoietic stem cells and hematopoietic progenitor cells | – Maternal leukocytes provide active immunity by fighting pathogens via phagocytosis and intracellular killing, produce microbicidal molecules, present antigens; also play vital role in shaping infant’s immune system, promoting development of immunocompetence and altering gut bacterial colonization | [ref. 19,ref. 114,ref. 115] |
| Proteins, glycoproteins and peptides | Cytokines, chemokines, soluble receptors, receptor agonists and antagonists, growth factors, immunoglobulin and others | – They enhance defense against pathogenic bacteria, viruses and yeasts and promote gut development and immune function. Cytokines and chemokines are the most redundant secreted proteins that provide active immunity to infants. For example, TGF-β prevents diseases induced by allergy and controls wound repair and inflammation; G-CSF plays a role in sepsis treatment and enhances cell prefoliation, crypt depth and villi; IL-6 (a key circulating pyrogen) activates CNS mechanisms in fever during infection and inflammation; IL-7 helps develop thymic; IL-8 protects from TNF-α-induced damage; IL-10 has anti-inflammatory activity; IFN-γ has pro-inflammatory activity as it inhibits the Th2/allergic response while increases the Th1/inflammation response- HBM contains glycoprotein cytokine receptors that modulate immune responses. For example, sTNFR1/2 and IL1Ra suppress pro-inflammatory TNF-α and IL-1 activity, respectively, decrease stimulation of IL-8 secretion from the intestinal epithelial of neonates and reduce necrotizing enterocolitis; TLR1-9 agonist and antagonist protect the infant from infections- Growth factors: TGF-β induces regulatory T cell production; epidermal GF inhibits apoptosis in intestinal cells; NGF enhances the outgrowth and survival of neurons; IGF alters intestinal atrophy, induces erythropoiesis; VEGF plays a role in angiogenesis regulation, decreasing the burden of premature retinopathy- Immunoglobulins: IgG, IgM and IgA account for 90% of HBM immunoglobulins and provide passive immunity to the newborn. The major function of IgA and IgG is neutralization of pathogens by binding to them and preventing them from binding to the epithelial cells in the gut mucosa. Moreover, by opsonizing the antigen for complement fixation and destruction, IgM suppresses microbial infections- Bile salt-dependent lipase blocks viral infection (such as HIV) by binding to the pathogen receptor DC-SIGN- Mucin1/4 protect infant from viral (e.g., rotavirus, norovirus and HIV) and bacterial (e.g., E. coli and S. enterica) infections- Cathelicidin-derived antimicrobial peptides produced by cells in breast milk protect infant from autoimmune diseases and have broad antimicrobial activities against Gram-positive and Gram-negative bacteria- α-lactalbumin is the major protein found in HBM that is converted in the stomach to HAMLET. In the presence of free oleic acid, HAMLET reduces the volume of >95% of skin papilloma- Soluble CD14 sensitizes the innate mucosal immune system to Gram-negative bacteria, such as E. coli, and mediates TLR4 binding to lipopolysaccharide of Gram-negative bacteria; inhibits TLR2 signaling and attenuates TLR4 signaling- HβD-2 is a peptide that inhibits TLR7 signaling and has antibacterial activities against Salmonella spp., E. coli and P. aeruginosa– Lactoferrin is a glycoprotein that has capacity against various fungi, viruses and bacteria; inhibitory effects reported against V. cholera and E. coli; responsible for sequestering iron needed by bacteria for growth and survival; influences TLR4 signaling- Lactadherin is a glycoprotein that protects neonates from rotavirus infection, mediates phagocytosis of apoptotic cells, blocks NF-κB and TLR4 signaling leading to a signaling cascade that reduces inflammation | [ref. 19,ref. 98,ref. 110,ref. 116,ref. 117,ref. 118,ref. 119,ref. 120,ref. 121,ref. 122,ref. 123] |
| Lysozymes | – Lysozymes hinder growth of many bacterial species by disrupting the proteoglycan layer of the cell wall- Lysozymes are characterized by a positive charge which can facilitate electrostatic interactions with the viral capsid blocking the viral fusion proteins (especially in HSV and HIV) | [ref. 124,ref. 125,ref. 126] | |
| Nucleotides | CMP, UMP, GMP, AMP | – Enhance immune responses and promote the development of a less pathogenic intestinal flora in infant | [ref. 127,ref. 128] |
| Nucleic acids | – DNA fragments- ncRNAs, including miRNA, siRNA, lncRNA, circRNA, piRNA, rRNA and tRNA | – miRNAs have direct impacts on immunological regulation, such as suppressing the production of essential transcription factors in immune cell polarization or altering the epigenetic state of immune cell lineages- Other ncRNAs are less studied than miRNA but have been found to be functionally involved in several regulatory mechanisms related to miRNA mechanisms and mRNA translation process | [ref. 129,ref. 130] |
Abbreviations: DC-SIGN, dendritic cell-specific intercellular adhesion molecule-3-grabbing non-integrin; HAMLET, human α-lactalbumin made lethal to tumor cells; IGF, insulin-like growth factor; TGF, transforming growth factor; TNF, tumor necrosis factor; Th; T helper cells; Teff, effector T cell; Treg, regulatory T cell; TLR, Toll-like receptor; HBM, human breast milk; HSV, herpes simplex virus; HIV, human immunodeficiency virus; HβD-2, human β-defensin 2; ncRNAs, non-coding RNAs; miRNA, microRNA; siRNA, small interfering RNA; lncRNA, long noncoding RNA; circRNA, circular RNA; piRNA, Piwi-interacting RNA; rRNA, ribosomal RNA; tRNA, transfer RNA; CMP, cytidine monophosphate; UMP, uridine monophosphate; GNP, guanosine monophosphate; AMP, adenosine monophosphate.
HBM contains two main groups of proteins: whey and casein. These two classes are present with ratios varying from 70/30 to 80/20 and 50/50 in early and late lactation, respectively [ref. 131,ref. 132]. Lactoferrin, one of the main proteins in the whey class, prevents the spread of bacterial infections among infants. However, there are other available proteins such as α-lactalbumin, cathelicidin-derived antimicrobial peptides and folate-binding protein in HBM [ref. 131]. α-lactalbumin is the principal protein found in HBM that is converted in the stomach to “human α-lactalbumin made lethal to tumor cells” (HAMLET). Cathelicidin-derived antimicrobial peptides are produced by HBM cells. They convey protection of the mother from the risk of BC, infection and allergy and protect infants from autoimmune diseases [ref. 19]. Furthermore, HBM can act as an inflammatory modulator by suppressing the interleukins participating in the regulation of proinflammatory mediators such as cytokine genes (e.g., IL-8 gene) [ref. 133].
HBM also contains several growth factors that widely impact nervous, vascular, endocrine and intestinal systems [ref. 19]. The epidermal growth factor (EGF) is located in the amniotic fluid and has higher levels in the colostrum and preterm milk compared to term milk [ref. 134,ref. 135]. The EGF stimulates intestinal cells to increase protein synthesis, water and glucose absorption, cell division and DNA synthesis [ref. 136]. Further, the EGF is essential for mucosal intestine healing [ref. 135]. In addition to IgA, which is the predominant antibody found in HBM [ref. 137], stem cell markers estrogen-related receptor beta (ESRRB), cytokeratin 5 (CK5) and myoepithelial marker CK14 are reported as components of HBM [ref. 138].
Cytokines are peptides that have many functions and work in both paracrine/autocrine ways [ref. 80]. Chemokines are a specific group of cytokines that stimulate the movement of other cells. HBM cytokines are classified into two wide classes: those that protect against pathogens or promote inflammation and those that decrease inflammation [ref. 139]. Transforming growth factor-beta (TGF-β) is one of the most common cytokines in HBM [ref. 140]. It is activated by the acidity of the stomach. Moreover, TGF-β aids in wound healing and allergic disease prevention [ref. 141].
HBM uniquely has a variety of structures and a high concentration of oligosaccharides as prebiotics (non-digestible food ingredients) that enhance the growth of probiotics (microorganisms that afford health benefits to the host when taken adequately) in the intestines [ref. 142]. It was reported that formula-fed infants have fewer oligosaccharides than breastfed infants [ref. 143]. Additionally, it is well known that breastfed newborns exhibit a high prevalence of the probiotic Lactobacillus species, especially Lactobacillus bifidus, which acidifies the gut and prevents enteric pathogens from infecting breastfed infants [ref. 144,ref. 145]. Furthermore, Bifidobacterium species are not dominant in the gut of formula-fed infants [ref. 146]. Giving probiotics with formula decreases the cases and severity of infant diarrhea [ref. 147]. However, most isolated probiotics are taken from fecal infant microbiota or foods [ref. 83]. The benefits of prebiotic and probiotic supplementation on infants were investigated by several studies [ref. 148,ref. 149]. Clostridium histolyticum was highly detected in the placebo (control) group compared to the probiotic administered group. Moreover, the prebiotic and probiotic groups were less vulnerable to infection with viral respiratory tract infections. Furthermore, the episodes induced by rhinovirus were significantly higher in the placebo group compared to the probiotics and prebiotics groups [ref. 150].
HBM oligosaccharides (HMOs), which mimic histo-blood group antigens (HBGAs) and behave as receptor decoys, interact with noroviruses. It was discovered that an HBM oligosaccharide (i.e., 2′-fucosyllactose (2′FL)) prevents the GI.1 and GII.17 noroviruses from attaching to HBGAs [ref. 151]. The results were supported by other studies; two HBM oligosaccharides, 2′FL and 3-fucosyllactose (3FL), have been found to prevent norovirus from binding to surrogate HBGA samples. X-ray crystallography revealed that 2′FL and 3FL bind to the same HBGA pockets on the norovirus capsid, as they structurally resemble HBGAs [ref. 152]. These findings show that 2′FL and 3FL may function as natural decoys in humans. Figure 3 illustrates the structural basis for norovirus inhibition by 2′FL and 3FL.
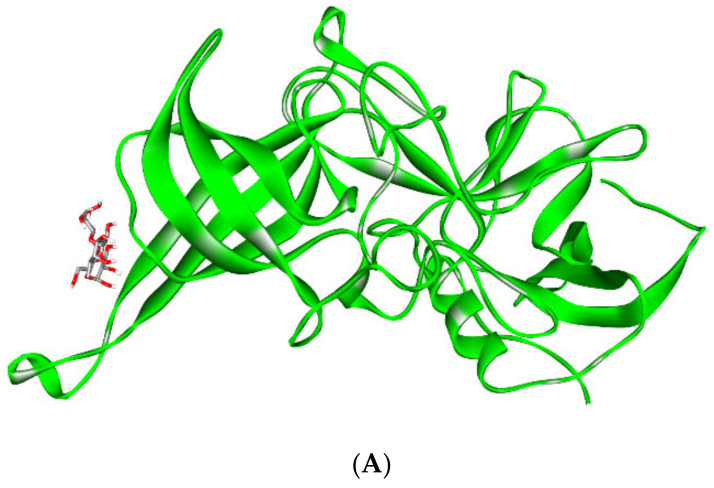
Moreover, catabolic pathways that help the growth of Roseburia and Eubacterium (gut flora linked to protection from immune and metabolic problems and from colorectal cancer) on distinct HBM oligosaccharides were detected [ref. 153]. During growth on selected HBM oligosaccharides and in co-cultures with Akkermansia muciniphila on mucin, the HBM oligosaccharides pathways were elevated along with additional glycan-utilization loci, suggesting an additional role in permitting cross-feeding and access to mucin O-glycans [ref. 153]. Furthermore, Bifidobacterium longum subsp. infantis also uses small-mass neutral HBM oligosaccharides, with several of them being fucosylated [ref. 154]. A time-dependent effect was discovered in a temporal glycan consumption profile. On the other hand, Bifidobacterium bifidum possesses a glycoside hydrolase family (i.e., lacto-N-biosidase) for degrading lacto-N-tetraose and liberating lacto-N-biose I [ref. 155]. Overall, this study shows possible symbiosis between humans and bifidobacterial species in the infant gut.
Reactive oxygen species (ROS) are highly oxidizing molecules involved in cellular signaling. Due to their oxidative impacts, high levels of ROS can cause damage to fundamental macromolecular components, including DNA, protein and lipids [ref. 156,ref. 157]. To override these negative effects, there is an established antioxidant system inside the body [ref. 158]. Many antioxidants were found in HBM, such as melatonin, glutathione S-transferase, glutathione peroxidase, catalase, glutathione reductase and superoxide dismutase [ref. 159]. They are classified into exogenous and endogenous and further grouped into enzymatic molecules, non-enzymatic molecules and hormones [ref. 158]. The antioxidant content is lower in mature milk compared to colostrum, and their activity declines over the breastfeeding period [ref. 160]. Regarding enzymatic molecules, catalase—which is composed of four protein subunits—participates in hydrogen peroxide detoxification and helps in ROS elimination [ref. 161,ref. 162]. Glutathione, one of the main non-enzymatic molecules, regenerates some antioxidants, including vitamin E and C, to their active forms [ref. 163]. Melatonin is an endocrine molecule produced by the pineal gland that has protective impacts against aging [ref. 164]. Accordingly, it is considered a promising molecule for protecting the nervous system in infants [ref. 158].
Overall, breastfeeding decreases the risk for different diseases. Table 2 compares breastfeeding and commercial infant formula feeding to health outcomes. The report was prepared by the Agency for Healthcare Research and Quality (AHRQ) of the US Department of Health Human Services [ref. 165,ref. 166].
Table 2: The odds ratio of different diseases among breastfed people compared with commercial infant formula-fed or referent group specified.
| Condition | Breastfeeding (Months) | Comments * | OR ** |
|---|---|---|---|
| Otitis media | Any | – | 0.77 |
| ≥3 | Exclusive BF | 0.50 | |
| Upper RTI | >6 | Exclusive BF | 0.30 |
| Lower RTI | ≥4 | Exclusive BF | 0.28 |
| Asthma | ≥3 | Atopic family history | 0.60 |
| No atopic family history | 0.74 | ||
| RSV bronchiolitis | >4 | – | 0.26 |
| NEC | NICU stay | Preterm infants with exclusive HBM | 0.23 |
| Atopic dermatitis | >3 | Exclusive BF negative family history | 0.84 |
| Exclusive BF positive family history | 0.58 | ||
| Gastroenteritis | Any | – | 0.36 |
| IBD | Any | – | 0.69 |
| Obesity | Any | – | 0.76 |
| Celiac disease | >2 | Gluten exposure when BF | 0.48 |
| T2D | >3 | Exclusive BF | 0.71 |
| Any | – | 0.61 | |
| ALL | >6 | – | 0.80 |
| – | 0.85 | ||
| SIDS | Any | – | 0.64 |
Abbreviations: ALL, acute lymphocytic leukemia; BF, breastfeeding; HBM, human breast milk; IBD, inflammatory bowel disease; RSV, respiratory syncytial virus; T2D, type 2 diabetes; RTI, respiratory tract infection; NEC, necrotizing enterocolitis; NICU, neonatal intensive care unit; SIDS sudden infants death syndrome. * Referent group is exclusive BF ≥ 6 months. ** OR, odds ratio: expressed as increased risk relative to commercial formula feeding.
Moreover, morbidity and mortality among breastfed newborns were shown to be several times lower compared to those non-breastfed [ref. 167]. This is viewed as a result of the existence of protective chemicals that play a critical role in safeguarding infants’ bodies against diseases, either directly by preventing pathogens from binding to their cellular receptors or indirectly by altering the gut flora [ref. 168,ref. 169]. Individual differences, mothers’ genotype, infant genotype, concentrations and digestion site are all considered when these bioactive substances act as immunomodulatory molecules [ref. 85,ref. 170].
5. Circulating miRNAs in HBM
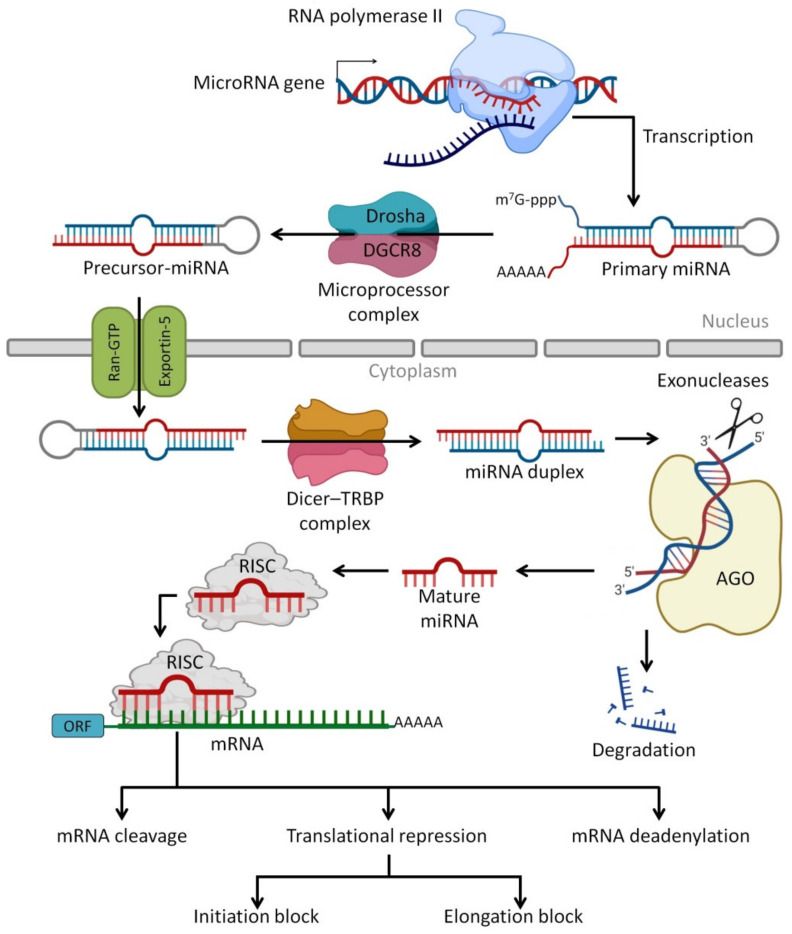
miRNAs are the most abundant class of very small regulatory non-coding RNA molecules that are composed of 20 to 24 nucleotides and are capable of controlling 40% to 60% of gene expression at the post-transcriptional level [ref. 171,ref. 172]. The miRNAs can be produced endogenously, delivered exogenously from neighbor cells as a cell–cell communication, delivered from foods such as plants and human HBM as cell-free miRNAs or via milk exosomes [ref. 173]. miRNAs regulate protein synthesis by base-pairing to target mRNAs [ref. 174] and lead to suppressed protein synthesis by different mechanisms, including translation repressing or targeted mRNA degradation [ref. 175]. Furthermore, there are some miRNAs found in pathways that improve and increase target mRNA expression [ref. 176]. In addition to miRNA functions in normal physiological processes such as regulation of gene expression [ref. 177], they are utilized as biomarkers for prognosis and diagnosis of cancer, gastrointestinal tract (GIT) disorders, autoimmunity and other diseases [ref. 178]. This is due to the presence of some specific tissue-derived miRNAs as extracellular circulating miRNA molecules that are found in body fluids such as plasma, saliva, urine and milk [ref. 179,ref. 180]. Figure 4 presents the processing pathways of miRNAs in the human body.
miRNAs have been found in higher concentrations in all milk fractions (i.e., cells, lipids and skim) than in other body fluids, including plasma [ref. 185]. Milk cells have the largest concentration and variety of miRNAs, while skim milk has the lowest [ref. 51,ref. 186]. About 1467 recognized miRNAs and 1996 novel miRNAs have been discovered in milk cells [ref. 185], while 429 mature miRNAs have been detected in skim milk [ref. 187]. In addition, 602 miRNAs were found in isolated exosomes in skim milk [ref. 188], and 308 miRNAs were found in milk lipids [ref. 189]. Although the substantial heterogeneity of miRNA profiles between different breastfeeding women has been documented [ref. 49], the causes for this variability have not been addressed to date, underscoring the importance of future research in this area. A maternal high-fat diet was shown to modulate miRNAs isolated from HBM fat globules, which can modify metabolic pathways in HBM-fed newborns [ref. 189].
The miRNA is an important substance in HBM because, firstly, the highest concentration of miRNAs is found in HBM (47,240 μg/L in HBM vs. 308 μg/L in plasma and 94 μg/L in urine) [ref. 190], which is attributed to the presence of stem cells in HBM [ref. 191,ref. 192], and the presence of HBM exosome-derived miRNAs [ref. 190]. Secondly, HBM miRNAs are very resistant to harsh conditions such as pasteurization and milk bank storage procedures [ref. 49,ref. 188], ultraviolet radiation [ref. 193], RNase digestion, high temperature, low pH and multiple freeze/thaw cycles [ref. 49,ref. 188]. Thirdly, high heterogeneity of miRNAs was detected in all fractions of milk [ref. 44,ref. 190]. Fourthly, the majority of miRNA originates from mammary epithelial cells, with only small fractions originating from maternal blood circulation [ref. 44]. That leads to supplying the HBM-fed infants with a wide spectrum of organ-derived miRNAs, such as pancreatic miR-216 and miR-217, hematopoietic-derived miR-142-5 and liver-derived miR-122 [ref. 49,ref. 188,ref. 194,ref. 195]. miRNAs in HBM are not limited to endogenous synthesis (either from lactating glands or maternal blood circulation), as food derived-miRNAs have also been detected [ref. 196]. This high concentration and heterogeneity of detected miRNAs in HBM exert regulatory functions by targeting a high spectrum of mRNA involved in adaptive and innate immune responses [ref. 49], regulating and inhibiting different types of cancers [ref. 197], regulating blood lipid profiles [ref. 196], regulating blood glucose levels [ref. 198] and protecting from cardiovascular disease and sickle cell disease [ref. 44].
Figure 5 depicts a scenario displaying the sources of exogenous miRNA for the infant (HBM and infant formulas) and their uptake in the infant’s GIT together with other macro/micronutrients (i.e., amino acids and fatty acids). It is worth noting that infant formulae have far lower levels of miRNAs than HBM, with possible changes in their biological activity that need to be investigated further [ref. 199,ref. 200,ref. 201,ref. 202,ref. 203,ref. 204].
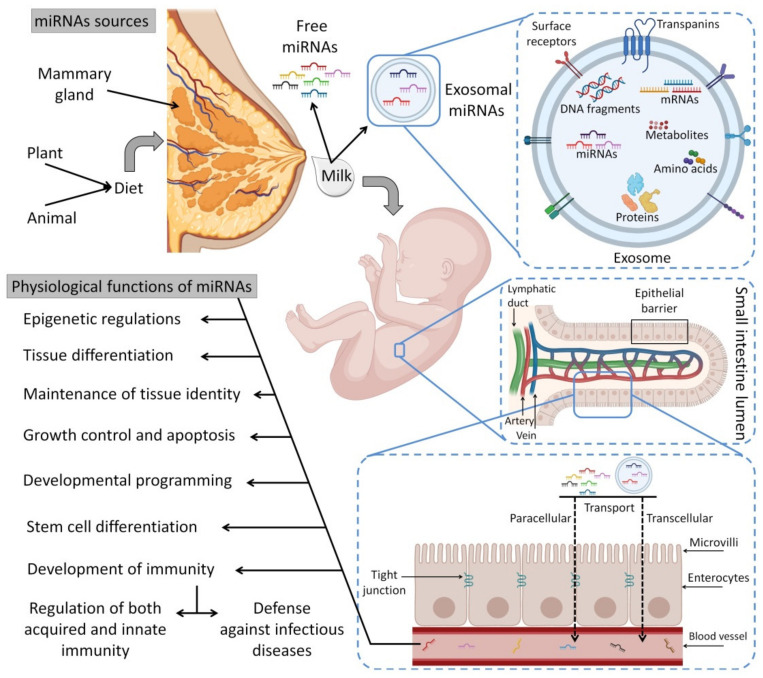
5.1. Exosomal miRNAs
Exosomes are small membrane vesicles (30 to 150 nm in diameter) produced from endosomes that are discharged into the extracellular environment by several different cells [ref. 208]. Exosomes that are found in different physiological fluids (i.e., milk, amniotic fluid, ascites fluid, blood, saliva and urine) have distinct subsets of miRNAs [ref. 190]. Exosomes and other extracellular vesicles attach to several categories of cells using endocytosis procedures to transport miRNAs [ref. 209,ref. 210,ref. 211]. Exosomal miRNAs can be protected from degradation in harsh environments [ref. 212]. Particularly, milk exosomes protect delivered miRNAs from RNAase digestion, varying pHs along the GIT and cycles of freezing/thawing in the case of frozen HBM [ref. 188,ref. 199,ref. 213,ref. 214]. Exosomes allow endocytosis to transport miRNAs from the GIT into the bloodstream and target tissues [ref. 215].
Additionally, other microvesicles, including apoptotic bodies (tiny vesicles resulting from the death of apoptotic cells), are also involved in miRNA protection [ref. 216]. Furthermore, HBM-derived miRNAs could be completely transported since they are protected within the cells and thus survive the offspring’s GI system and dwell in various organs [ref. 56,ref. 217]. A few theories have been suggested in relation to free miRNA in milk. RNase can be found in all fluids in the body [ref. 218] and degrades RNA molecules into tiny pieces, which indicates its importance in the maturation process of RNA [ref. 219]. RNAs, on the other hand, are known to be unstable in harsh environments [ref. 220,ref. 221]. However, HBM-derived miRNAs remain remarkably stable even when treated with RNase in vitro [ref. 222]. Given that milk miRNAs are surrounded by a lipid bi-layered membrane and are supplied with adherence molecules, it has been proposed that their packaging in “vehicle” structures, such as exosomes, somatic cells and other microvesicles, may be vital for their long-distance passage [ref. 49,ref. 188,ref. 223].
5.2. Sources of HBM miRNAs and the Effects of Different Conditions
The following factors have an impact on miRNAs in HBM from food sources: (1) foods manufacturing, which may include baking, frying, fermenting and a variety of other processing treatments that may compromise the integrity of small RNA structures; (2) unharmed passage via the GIT since the duodenum contains nuclease enzymes for DNA and RNA; (3) the absorption into the blood via different gastrointestinal barriers; (4) transferring into alveolar cells; and (5) milk secretion by alveolar cells [ref. 52,ref. 224,ref. 225,ref. 226]. These five factors contain a variety of chemical environments, some of which cause antagonistic effects against miRNAs. As a result, large miRNA levels should be available in the foods to establish a quantifiable titer in HBM [ref. 223]. However, since the neonates lack a well-developed gastrointestinal barrier, larger molecules (such as the mother’s antibodies) may be able to enter directly into the blood circulation. When miRNAs such as species-specific miRNAs are present in the milk, their entrance is enhanced into the bloodstream. Therefore, newborns could be able to pick up miRNAs more easily from food. However, transferring miRNAs from meals into HBM is only possible if they are present in large quantities [ref. 49,ref. 51,ref. 227].
Zhang et al. [ref. 228] identified plant food-derived miRNAs in human circulation and bodily fluids, which were tracked by several studies for further investigation to understand their role in mammalian gene regulation [ref. 223,ref. 225,ref. 227,ref. 229]. The inability of all plant-derived miRNAs to pass through the GIT into circulation is a key concern when contemplating miRNAs as biological modulators in humans [ref. 230], although it has been established that some plant food-derived miRNAs in HBM can target many human mRNAs. For example, miR-156a, miR-166a, miR-167a, miR-172a and miR-168a, target 271, 88, 15, 7 and 4 distinct human mRNAs, respectively [ref. 223]. The variety and concentration of human miRNAs may be influenced by factors such as the mothers’ age, body mass index (BMI), neonatal gender, the breastfeeding mother and infant health, term or preterm birth and lactation duration [ref. 45,ref. 231,ref. 232].
According to Carney’s study [ref. 46], significant correlations were found between gestational age and 21 of the 26 miRNAs altered in skim or lipid portions in samples of pre-mature infants’ maternal HBM. However, no miRNAs were associated with maternal ethnicity or race, twin pregnancy or maternal hypertension [ref. 46]. In either colostrum or mature milk, the intensity of miRNAs was not associated with maternal age at gestational or conception week. Moreover, the contents of miR-378 and miR-30b were higher in colostrum received by girls than in that received by boys. After correcting for maternal pre-pregnancy BMI, this pattern remained for miR-378 [ref. 45]. The levels of expression of let-7a, miR-30b and miR-378 were negatively associated with BMI of maternal pre-pregnancy and late pregnancy, but positively associated with maternal weight gain during pregnancy. Furthermore, the level of let-7a in mature milk at the late stage of pregnancy was adversely associated with maternal weight [ref. 45].
According to a recent study, there are 63 highly expressed miRNAs in HBM. Of them, 13 are colostrum-specific miRNAs, 13 are mature-specific miRNAs and the rest (37) are common miRNAs [ref. 233]. Table 3 lists these miRNAs and extensively discusses their physiological functions in normal and pathological conditions. In addition to the functions listed in Table 3, other studies have confirmed that miRNAs control the expression levels of target genes through synergism, especially knowing that several miRNAs can target 3’UTR of the same mRNA transcript [ref. 234,ref. 235,ref. 236].
Table 3: The abundantly expressed miRNAs in HBM and their physiological functions in normal and pathological conditions.
| miRNA [Sequence] | Function [Reference] |
|---|---|
| Colostrum-specific miRNAs | |
| hsa-let-7i-5p[UGAGGUAGUAGUUUGUGCUGUU] | Regulates cell morphology and migration through distinct signaling pathways in normal and pathogenic urethral fibroblasts [ref. 237]; protects against acute ischemic stroke [ref. 238]; controls the migration of head and neck cancer cells through downregulation of BMI1 protein [ref. 239]; inactivates localized scleroderma [ref. 240]; regulates MS pathogenesis by suppressing induction Treg by targeting IGF1R and TGFβR1 [ref. 241]; protects against pneumoconiosis caused by nanoparticles inhalation [ref. 242]; acts as an autophagy suppressor by targeting ATG10 and ATG16L1 in NPC and may represent a promising therapeutic target for NPC treatment [ref. 243]; targets HABP4 gene and functions as a tumor promoter in ccRCC, and thus offers a potential target for treatment [ref. 244]; inhibits granulosa-luteal cell proliferation and oestradiol biosynthesis by directly targeting IMP2 [ref. 245]; inhibits KGN proliferation and decreases estradiol production in an IMP2-dependent manner, providing insights into the pathogenesis of PCOS [ref. 246]; promotes differentiation of hESCs [ref. 247]; inhibits the metastasis of TNBC [ref. 248]. |
| hsa-miR-423-5p[AAAAGCUGGGUUGAGAGGGCAA] | Regulates ovarian response to ovulation [ref. 249]; targets ING-4 and upregulates signaling molecules such as p-AKT and p-ERK1/2, which support miR-423-5p functions as an oncogene in glioma and suggests targeting it as therapeutic potential for glioma [ref. 250]; targets PTTG1 and SYT1 mRNAs, thus induces cell apoptosis, inhibits cell proliferation and reduces growth hormone release and migration of GH3 cells [ref. 251]; regulates TGF-β signaling by targeting SMAD2, thus functions in the development of bicuspid aortic valve BAV disease and its complication, bicuspid aortopathy [ref. 252]; induces silencing of the nerve growth factor, which promotes retinal microvascular dysfunction, demonstrating the potential for miRNA-based therapy for treating diabetic retinopathy [ref. 253]; promotes BC invasion [ref. 254]. |
| hsa-miR-320b[UUCAAGUAAUUCAGGAUAGGU] | Negatively regulates normal human epidermal keratinocyte proliferation by targeting AKT3 to regulate the STAT3 and SAPK/JNK pathways, thus might participate in the pathogenesis of psoriasis, may act as a novel diagnostic marker or therapeutic target for this disease [ref. 255]; affects HCC radiosensitivity to ionizing radiation treatment through DNA damage repair signaling [ref. 256]; regulates osteoblast differentiation [ref. 257]; modulates cholesterol efflux and atherosclerosis [ref. 258]. |
| hsa-miR-26b-5p[UUCAAGUAAUUCAGGAUAGGU] | Controls the adipogenic differentiation of hADMSC [ref. 259]; acts as a tumor suppressor in PC [ref. 260]; affects cytokine secretion in RA [ref. 261]; modulates Th17 cell plasticity in RA [ref. 262]; inhibits proliferation, migration, invasion and apoptosis induction of osteosarcoma cells [ref. 263]. |
| hsa-miR-146a-5p[UGAGAACUGAAUUCCAUGGGUU] | Modulates androgen-independent PC cell apoptosis [ref. 264]; regulates KIR expression [ref. 265]; acts as a tumor suppressor in B-cell malignancies [ref. 266]; inhibits the metastasis of ccRCC [ref. 267]; inhibits NSCLC proliferation [ref. 268]; protects cardiomyocytes and myocardial tissues in polymicrobial sepsis [ref. 269]; associated with low-risk human PSCCs [ref. 270]; improves the decidual cytokine microenvironment [ref. 271]; acts as tumor suppressor in esophageal, prostatic, glioma and ovarian cancers [ref. 272,ref. 273,ref. 274,ref. 275]; suppresses osteoclastogenesis [ref. 276]. |
| hsa-let-7c-5p[UGAGGUAGUAGGUUGUAUGGUU] | Targets TGF-β signaling and contributes to the pathogenesis of renal fibrosis [ref. 277]; inhibits osteo/odontogenic differentiation of IGF-1-treated DPMSCs by targeting IGF-1 [ref. 278]; inhibits MAP4K4 expression; inhibits OSCC cell proliferation and migration [ref. 279]. |
| hsa-miR-200b-3p[UAAUACUGCCUGGUAAUGAUGA] | Inhibits epithelial-to-mesenchymal transition in TNBC [ref. 280]; inhibits human cytomegalovirus replication [ref. 281]; acts as a tumor suppressor in HCC [ref. 282]; regulates self-renewing divisions in PC cells by inducing less Notch signaling and promotes daughter cells to become asymmetric [ref. 283]; promotes endothelial cell apoptosis by targeting HDAC4 in atherosclerosis [ref. 284]; inhibits cell proliferation and Ca2+ influx in PASMCs [ref. 285]. |
| hsa-miR-151b[UCGAGGAGCUCACAGUCU] | Controls expression of GHR [ref. 189] and regulates proliferation and apoptosis of THCA cells through SNRPB axis [ref. 286]. |
| hsa-miR-24-3p[UGGCUCAGUUCAGCAGGAACAG] | Enhances NPC radiosensitivity by targeting both the 3’UTR and 5’UTR of Jab1/CSN5 [ref. 287]; enhances cell growth in HCC by targeting metallothionein 1M [ref. 288]; regulates lung adenocarcinoma progression through FGFR3 signaling [ref. 289]; regulates neuronal differentiation by regulating hippocalcin expression [ref. 290]; inhibits progression of pancreatic ductal adenocarcinoma through LAMB3 downregulation [ref. 291]; regulates proliferation, migration and invasion of cancer cells by directly targeting p130Cas [ref. 292]; suppresses proliferation and invasiveness of gastric mucosal lesions [ref. 293]. |
| hsa-miR-107[AGCAGCAUUGUACAGGGCUAUCA] | Regulates cellular migration by inducing CDK5 activity and the associated molecular pathways [ref. 294]; inhibits acute aortic dissection progression [ref. 295]; regulates chemo-drug sensitivity in BC cell by targeting TRIAP1 [ref. 296]; downregulates Cdc42 3’UTR and suppresses ESCC proliferation, migration and invasion [ref. 297]; modulates NeuroD1 and SOX6 genes affecting MSCs commitment toward insulin-producing cells [ref. 298]; modulates chondrocyte proliferation [ref. 299]; inhibits glioma cell migration and invasion [ref. 300]; regulates cisplatin chemosensitivity in NSLCC [ref. 301]; promotes tumor suppressor in GC [ref. 302]; inhibits endothelial progenitor cell differentiation [ref. 303]; contributes to post-stroke angiogenesis [ref. 304]; antagonizes profibrotic phenotypes of pericytes [ref. 305]. |
| hsa-miR-221-3p[AGCUACAUUGUCUGCUGGGUUUC] | Regulates apoptosis in ovarian granulosa cells [ref. 306]; regulates epithelial ovarian cancer progression [ref. 307]; affects proliferation and apoptosis of keratinocytes [ref. 308]; reduces airway eosinophilia and CXCL17 expression in asthma [ref. 309]; targets CACNA1C and KCNJ5 and alters cardiac ion channel expression [ref. 310]; downregulates EIF5A2 and inhibits cell proliferation in medulloblastoma [ref. 311], acts as a tumor suppressor and disease progression marker in prostate cancer [ref. 312]; down-modulates KIT receptor, which suggests a potential role in cancer therapy [ref. 313]; regulates CDKN1C/p57 and CDKN1B/p27 expression in HCC [ref. 314], suppresses HDAC6 providing a new target for the treatment of liver malignancies [ref. 315], targets KIT and ETV1 in gastrointestinal stromal tumors [ref. 316]. |
| hsa-miR-151a-5p[UCGAGGAGCUCACAGUCUAGU] | Regulates E-cadherin in NSCLC cells, which promotes partial EMT and thus acts as a therapeutic target [ref. 317]. |
| hsa-miR-378c[ACUGGACUUGGAGUCAGAAGAGUGG] | Suppresses stomach adenocarcinoma cell proliferation, migration, invasion and epithelial-mesenchymal transition [ref. 318]. |
| Mature milk-specific miRNAs | |
| hsa-miR-375[CCCCGCGACGAGCCCCUCGCACAAACCGGACCUGAGCGUUUUGUUCGUUCGGCUCGCGUGAGGC] | Induces generation of insulin-producing cells from human decidua basalis-derived stromal cells [ref. 319]; promotes pancreatic cell differentiation [ref. 320]; suppresses ESCC by direct targeting of SHOX2 [ref. 321], reduces viability of HCC under hypoxic conditions [ref. 322], suppresses bladder cancer via the Wnt/beta-catenin pathway [ref. 323]; enhances generation of insulin-producing cells from human MSCs [ref. 324]; promotes redifferentiation of adult human β cells [ref. 325]; enhances infant growth and development [ref. 189]; regulates expression of JAK2 [ref. 189]; activates p21; suppresses telomerase activity [ref. 326]. |
| hsa-miR-193b-3p[AACUGGCCCUCAAAGUCCCGCU] | Regulates matrix metalloproteinase in chondrocytes [ref. 327]; regulates chondrogenesis [ref. 328] and chondrocyte metabolism [ref. 329]; acts as tumor suppressor in ovarian carcinoma cells [ref. 330]; attenuates neuroinflammation in early brain injury after subarachnoid hemorrhage [ref. 331]. |
| hsa-miR-345-5p[GCUGACUCCUAGUCCAGGGCUC] | Acts as anti-osteogenic factor [ref. 332]. |
| hsa-miR-423-3p[AGCUCGGUCUGAGGCCCCUCAGU] | Activates oncogenic autophagy in GC [ref. 333] and enhances tumor growth in lung adenocarcinoma [ref. 334]. |
| hsa-miR-125a-5p[UCCCUGAGACCCUUUAACCUGUGA] | Decreases sensitivity of Treg cells toward IL-6-mediated conversion [ref. 332]; suppresses breast cancer by downregulating BAP1 [ref. 335,ref. 336]; suppresses bladder cancer by targeting FUT4 [ref. 97]; suppresses cervical carcinoma [ref. 337], HCC [ref. 338], GC [ref. 339] colon cancer [ref. 340], prostate carcinoma [ref. 341], bladder cancer [ref. 336] and CRC [ref. 342]; activates p53 and induces apoptosis in lung cancer cells [ref. 343,ref. 344]; contributes to hepatic stellate cell activation [ref. 345]; inhibits trophoblast cell migration and proliferation in preeclampsia [ref. 346]. |
| hsa-miR-148a-5p[AAAGUUCUGAGACACUCCGACU] | Regulates expression of SOCS-7 [ref. 189]; controls ATPase expression [ref. 189]; regulates triacylglycerol and long-chain acyl-CoA fatty acid synthesis [ref. 189]; regulates lactose synthesis [ref. 189]; promotes cartilage production [ref. 347]; relieves hepatic fibrosis [ref. 348]; regulates the stem cell-like side population distribution in ESCC [ref. 349]. |
| hsa-miR-29c-3p[UAGCACCAUUUGAAAUCGGUUA] | Regulates biological function of CRC [ref. 350]; suppresses gallbladder carcinoma [ref. 351], T-cell acute lymphoblastic leukemia [ref. 352], ovarian cancer [ref. 353] and melanoma [ref. 354]. |
| hsa-miR-27a-3p[UUCACAGUGGCUAAGUUCCGC] | Regulates expression of intercellular junctions at the brain endothelium and controls the endothelial barrier permeability [ref. 355]; suppresses osteoblastogenesis [ref. 356]; suppresses OSCCs [ref. 357] and HCC [ref. 358]; inhibits cell proliferation and inflammation of RA in synovial fibroblasts [ref. 356,ref. 359]; mediates of human adipogenesis [ref. 360]. |
| hsa-miR-365a-3p[UAAUGCCCCUAAAAAUCCUUAU] | Suppresses progression of PC [ref. 361] and GC [ref. 362]. |
| hsa-miR-365b-3p[UAAUGCCCCUAAAAAUCCUUAU] | Promotes HCC cell migration and invasion [ref. 363]. |
| hsa-miR-183-5p[UAUGGCACUGGUAGAAUUCACU] | Modulates cell adhesion [ref. 364]; regulates uterine receptivity and enhances embryo implantation [ref. 365]; promotes invasion of endometrial stromal cells [ref. 366]; regulates myogenic differentiation [ref. 367]. |
| hsa-miR-148b-3p[UCAGUGCAUCACAGAACUUUGU] | Stimulates osteogenesis [ref. 368] and suppresses glioma cells [ref. 369]. |
| hsa-miR-28-3p[CACUAGAUUGUGAGCUCCUGGA] | Inhibits diffuse large B-Cell lymphoma cell proliferation [ref. 370]. |
| Common miRNAs | |
| hsa-miR-141-3p[UAACACUGUCUGGUAAAGAUGG] | Suppresses ameloblastoma cell migration [ref. 371]; suppresses osteosarcoma cells [ref. 372], GC [ref. 373] and CRC [ref. 374]; promotes endothelial cell angiogenesis [ref. 375]; regulates myogenic differentiation in myoblasts [ref. 376]; regulates IL-13-induced airway mucus production [ref. 377]; regulates mesenchymal stem cell aging [ref. 378]. |
| hsa-miR-22-3p[AAGCUGCCAGUUGAAGAACUGU] | Suppresses endothelial progenitor cell proliferation and migration in venous thrombosis [ref. 379]; suppresses T-cells in ALL [ref. 380]; suppresses sepsis-induced acute kidney injury [ref. 381]. |
| hsa-miR-181a-5p[AACAUUCAACGCUGUCGGUGAGU] | Reduces oxidation resistance in osteoarthritis [ref. 382]; suppresses prostate cancer [ref. 383]; regulates several cancer genes [ref. 384]; regulates multiple malignant processes of breast cancer [ref. 385]; suppresses invasion and migration of HTR-8/SVneo in pre-eclampsia [ref. 386]. |
| hsa-miR-26a-5p[UUCAAGUAAUCCAGGAUAGGCU] | Regulates the glutamate transporter in multiple sclerosis [ref. 387]; regulates fatty acid and sterol metabolism in nonalcoholic fatty liver disease; regulates the expression of inducible nitric oxide synthase in human osteoarthritis chondrocytes [ref. 388]; suppresses breast cancer [ref. 389] and prostate cancer [ref. 390]. |
| hsa-miR-30a-5p[UGUAAACAUCCUCGACUGGAAG] | Suppresses CRC [ref. 391], lung squamous cell carcinoma [ref. 392], and renal cell carcinoma [ref. 393]. |
| hsa-let-7a-5p[UGAGGUAGUAGGUUGUAUAGUU] | Decreases cell proliferation and inhibits the expression of Bcl-2 in ovarian cancer cells [ref. 394]. |
| hsa-miR-148a-3p[UCAGUGCACUACAGAACUUUGU] | Suppresses GC [ref. 395]; promotes ADH4 expression [ref. 396]; regulates angiogenesis [ref. 397]. |
| hsa-miR-27b-3p[UUCACAGUGGCUAAGUUCUGC] | Suppresses glioma [ref. 398], lung cancer [ref. 399], CRC [ref. 400]; endothelial cell proliferation and migration in Kawasaki Disease [ref. 401]; suppresses Osteogenic differentiation of maxillary sinus membrane stem cells by targeting Sp7 [ref. 402]. |
| hsa-miR-146b-5p[UGAGAACUGAAUUCCAUAGGCUG] | Suppresses NSCLC [ref. 403], and glioma [ref. 404]; down-regulates BRCA1 expression in TNBC [ref. 405]; induces IL-6 [ref. 406]. |
| hsa-let-7f-5p[UGAGGUAGUAGAUUGUAUAGUU] | Promotes bone marrow MSCs survival in AD [ref. 407] and suppresses NSCLC [ref. 408]. |
| hsa-miR-21-5p[UAGCUUAUCAGACUGAUGUUGA] | Suppresses breast cancer cells [ref. 409]; induces angiogenesis [ref. 410]; regulates mesothelin expression [ref. 411]; promotes ThP-1 cell proliferation [ref. 412]; links EMT in keloid keratinocytes [ref. 413]. |
| hsa-miR-92a-3p[UAUUGCACUUGUCCCGGCCUGU] | Suppresses lymphoma [ref. 414]; promotes cell proliferation, invasion and metastasis, inhibiting cell apoptosis and serving as predictive biomarkers for tumor diagnosis or chemoresistance [ref. 415]; regulates angiogenesis in stromal cells [ref. 416]; relates to activated partial thromboplastin time, prothrombin activity and plasma lipocalin-2 level [ref. 417]; regulates cartilage-specific gene expression in chondrogenesis [ref. 418]; regulates aggrecanase-1 and 2 expressions in human articular chondrocytes [ref. 419]. |
| hsa-miR-16-5p[UAGCAGCACGUAAAUAUUGGCG] | Suppresses CRC [ref. 420], chordoma [ref. 421], neuroblastoma [ref. 422] and breast cancer [ref. 423]; involved in dilation of ischemic cardiomyopathy [ref. 424]; enhances radiosensitivity in prostate cancer [ref. 425]; prevents amyloid β-induce injury [ref. 426]; affects neurological function, autophagy and apoptosis of hippocampal neurons in AD [ref. 427]; controls development of osteoarthritis in chondrocytes [ref. 428]; suppresses myofibroblast activation in systemic sclerosis [ref. 429]; regulates the p53 signaling pathway in myoblast differentiation [ref. 430]; regulates postmenopausal osteoporosis [ref. 431]. |
| hsa-miR-101-3p[UACAGUACUGUGAUAACUGAA] | Suppresses HER2-positive BC [ref. 432], HCC [ref. 433,ref. 434], glioblastoma [ref. 435], endometrial carcinoma [ref. 436], NSCLC [ref. 437], renal cell carcinoma [ref. 438] and melanoma [ref. 439]; regulates cancer proliferation [ref. 440]; regulates mitochondrial metabolic function [ref. 440]; induces vascular endothelial cell dysfunction [ref. 441]; regulates osteogenesis [ref. 442]. |
| hsa-miR-30d-5p[UGUAAACAUCCCCGACUGGAAG] | Suppresses gallbladder carcinoma [ref. 443], rectal cancer [ref. 444], colon cancer [ref. 445], prostate cancer [ref. 446], ESCC [ref. 447], renal carcinoma [ref. 448], PC [ref. 449], HCC [ref. 450], THCA [ref. 451], LSCC [ref. 452] and NSCLC [ref. 453,ref. 454,ref. 455]. |
| hsa-miR-378a-3p[ACUGGACUUGGAGUCAGAAGGC] | Controls metabolism, muscle differentiation/regeneration and angiogenesis [ref. 456]; suppresses glioblastoma [ref. 457] and HCC [ref. 458]; protects against intestinal injury [ref. 459]; modulates keratinocytes cell cycle arrest in psoriasis keratinocytes [ref. 460]. |
| hsa-miR-191-5p[CAACGGAAUCCCAAAAGCAGCUG] | Inhibits replication of human immunodeficiency virus type 1 (HIV-1) [ref. 461]. |
| hsa-miR-10a-5p[UACCCUGUAGAUCCGAAUUUGUG] | Inhibits osteogenic differentiation [ref. 462]; inhibits keratinocyte proliferation in atopic dermatitis [ref. 463]; reduces IL-6-induced cartilage cell ferroptosis [ref. 464]; regulates BDNF expression in follicular fluid [ref. 465]; suppresses renal cell carcinoma [ref. 466]; mitigates Ca2+ entry in T cells through gut bacterial metabolite urolithin [ref. 467]; enhances viability and migration of human umbilical vein endothelial cells [ref. 468]. |
| hsa-let-7b-5p[UGAGGUAGUAGGUUGUGUGGUU] | Promotes protein processing in endoplasmic reticulum in acute pulmonary embolism [ref. 469]; promotes angiogenesis [ref. 470]; inhibits proliferation of leukemia [ref. 471]; inhibits proliferation of leukemia THP-1 Cells [ref. 471]. |
| hsa-miR-200a-3p[UAACACUGUCUGGUAACGAUGU] | Prevents MPP+-induced apoptotic cell death [ref. 472]. |
| hsa-miR-186-5p[CAAAGAAUUCUCCUUUUGGGCU] | Promotes apoptosis [ref. 473] and suppresses CRC [ref. 474]. |
| hsa-miR-320a[CUCCCCUCCGCCUUCUCUUCCCGGUUCUUCCCGGAGUCGGGAAAAGCUG] | Suppresses CRC [ref. 475,ref. 476], glioblastoma [ref. 477], GC [ref. 478], salivary adenoid cystic carcinoma [ref. 479] and CML [ref. 480]; targets genes in lithium response in bipolar disorder [ref. 481]; regulates fibrotic process in interstitial lung disease of systemic sclerosis [ref. 482]; regulates cell proliferation and apoptosis in multiple myeloma [ref. 483]; regulates erythroid differentiation [ref. 484]; controls glucagon expression [ref. 485]; regulates cell proliferation and apoptosis in multiple myeloma [ref. 483]; stimulates endometrial stromal cell migration during preimplantation embryo stage [ref. 486]; improves skeletal muscle mitochondrial metabolism [ref. 487]. |
| hsa-miR-181b-5p[AACAUUCAUUGCUGUCGGUGGGU] | Involved in Ang II-induced phenotypic transformation of smooth muscle cells in hypertension [ref. 488]; suppresses starvation-induced cardiomyocyte autophagy [ref. 488]; improves anti-tumor cytotoxic T cell response in B cells of CLL [ref. 489]; inhibits trophoblast cell migration and invasion in multiple abnormal trophoblast invasion [ref. 490]; modulates cell migratory proteins in endometrial stromal cells [ref. 491]; suppresses the progression of epilepsy [ref. 492]; suppresses gallbladder carcinoma [ref. 493]. |
| hsa-miR-30e-5p[UGUAAACAUCCUUGACUGGAAG] | Regulates angiogenesis, apoptosis, cell differentiation, oxidative stress and hypoxia [ref. 494]; regulates autophagy and apoptosis in contrast-induced acute kidney injury [ref. 495]; suppresses NSCLC [ref. 496] and squamous cell carcinoma of the head and neck [ref. 497]; enhances innate immune responses [ref. 498]; suppresses cancer cell adhesion, migration and invasion, and considered as a potential target for curbing metastatic spread in P53-deficient tumors [ref. 499]. |
| hsa-miR-103a-3p[AGCAGCAUUGUACAGGGCUAUGA] | Regulates BDNF expression in follicular fluid [ref. 465]; suppresses PC [ref. 500]; aggravates renal cell carcinoma [ref. 501]; regulates Wnt signaling pathway in colorectal carcinoma [ref. 502]. |
| hsa-miR-182-5p[UUUGGCAAUGGUAGAACUCACACU] | Mediates downregulation of BRCA1, impacting DNA repair and sensitivity to PARP inhibitors [ref. 503]; suppresses CRC [ref. 504], renal cell carcinoma [ref. 505], bladder cancer [ref. 506] and prostate cancer [ref. 507]; acts as glial cell line-derived neurotrophic factor GDNF mimics dopaminergic midbrain neurons [ref. 508]; associates with renal cancer cell mitotic arrest [ref. 509]. |
| hsa-miR-151a-3p[CUAGACUGAAGCUCCUUGAGG] | Enhances slug-dependent angiogenesis and regulates multiple functions in the lung, such as cell growth, motility, partial EMT and angiogenesis [ref. 510]. |
| hsa-miR-335-5p[UCAAGAGCAAUAACGAAAAAUGU] | Regulates bone homeostasis [ref. 511]; suppresses GC [ref. 512], uterine leiomyoma [ref. 513] and ESCC [ref. 514]; regulates cardiac mesoderm and progenitor cell differentiation [ref. 515]. |
| hsa-miR-25-3p[CAUUGCACUUGUCUCGGUCUGA] | Suppresses hepatocytes [ref. 516] and regulates osteoblast differentiation of human aortic valve interstitial cells [ref. 517]. |
| hsa-let-7g-5p[UGAGGUAGUAGUUUGUACAGUU] | Suppresses epithelial ovarian cancer [ref. 517] and glioblastoma [ref. 518]; and alleviates murine collagen-induced arthritis by inhibiting Th17 cell differentiation [ref. 519]. |
| hsa-miR-200c-3p[UAAUACUGCCGGGUAAUGAUGGA] | Suppresses prostate carcinoma [ref. 520], renal cell carcinoma [ref. 521] and epithelial ovarian cancer [ref. 522]; regulates integrin-mediated cell adhesion [ref. 523]; attenuates the tumor-infiltrating capacity of macrophages [ref. 524]. |
| hsa-miR-30c-5p[UGUAAACAUCCUACACUCUCAGC] | Suppresses GC [ref. 525] and prostate cancer [ref. 526]; protects cells from hypoxia-reoxygenation-induced apoptosis and induces cell proliferation and anti-apoptotic and proliferative effects [ref. 527]; reduces cellular migration and pro-angiogenic gene expression in extracellular vesicle EV-recipient cells [ref. 528]. |
| hsa-miR-429[UAAUACUGUCUGGUAAAACCGU] | Inhibits cell proliferation and Ca2+ influx by pulmonary artery smooth muscle cells [ref. 285]; regulates hypoxia [ref. 529]; suppresses breast cancer [ref. 530], osteosarcoma [ref. 531], THCA, soft tissue sarcoma [ref. 532], cervical cancer [ref. 533], GC [ref. 534], diffuse large B-cell lymphoma [ref. 535], esophageal carcinoma [ref. 536], HCC [ref. 537,ref. 538], glioblastoma [ref. 539], NPC [ref. 540], PC cancer [ref. 541], THCA [ref. 542], OSCC [ref. 543] and renal cell carcinoma [ref. 544]; inhibits bone metastasis in breast cancer [ref. 545]; regulates the transition between HIF1A and HIF3A expression in human endothelial cells [ref. 546]. |
| hsa-miR-99b-5p[CACCCGUAGAACCGACCUUGCG] | Suppresses primary myotubes [ref. 547], epidermal keratinocytes and cervical cancer cells [ref. 548]. |
| hsa-miR-29a-3p[UAGCACCAUCUGAAAUCGGUUA] | Modulates CYP2C19 in human liver cells [ref. 549]; suppresses cell proliferation [ref. 550]; activates hepatic stellate cells, which moderate their profibrogenic phenotype, supporting the use of miR-29a agonists for treating liver fibrosis [ref. 551]; regulates tumorigenicity and TME development [ref. 552]; involved in the progression of HCC, which elucidates its potential theragnostic implications [ref. 552]; suppresses GC [ref. 553] and renal cell carcinoma by regulating E2F1 expression by long non-coding RNA H19 [ref. 554]; induces TNFα in endothelial dysfunction [ref. 555]; mediates tumor immune infiltration in breast invasive carcinoma [ref. 556]; acts as a protective factor for fibrogenesis in gluteal muscle contracture [ref. 557]; regulates osteoblast differentiation and peri-implant osseointegration [ref. 558]; promotes intestinal epithelial apoptosis in ulcerative colitis [ref. 559]; regulates and restores endothelial function in normal people and cardiometabolic disorders, respectively [ref. 560]; regulates peripheral glucocorticoid receptor signaling [ref. 561]; suppresses CRC [ref. 562], gliomas [ref. 563], head and neck squamous cell carcinoma [ref. 564], prostate cancer [ref. 565], GC [ref. 553], PTC [ref. 566], lung cancer [ref. 567], cervical cancer [ref. 568] and endometrial cancer [ref. 569]; enhances the radiosensitivity of OSCC cells [ref. 570]; modulates ALDH5A1 and SLC22A7 in human liver cells [ref. 571]. |
| hsa-miR-30b-5p[UGUAAACAUCCUACACUCAGCU] | Suppresses HCC, which is sponged by long non-coding RNA HNF1A-AS1 oncogene [ref. 572]; inhibits GC cell migration [ref. 572]; regulates lipid metabolism [ref. 573]; involved in vascular smooth muscle cell differentiation [ref. 574]; involved in homocysteine-induced apoptosis in human coronary artery endothelial cells [ref. 575]; inhibits osteoblast differentiation [ref. 576]; inhibits proliferation and promotes apoptosis of medulloblastoma cells [ref. 577]; controls adverse effects of non-small cell lung cancer NSCLC radiotherapy [ref. 578]; mediates ferroptosis of trophoblasts, which is involved in the pathogenesis of preeclampsia [ref. 579]; suppresses expression of B-cell activating factor mRNA primary in Sjögren’s syndrome [ref. 580]. |
| hsa-miR-19b-3p[UGUGCAAAUCCAUGCAAAACUGA] | Suppresses cell mobility [ref. 581]; involved in proliferation, apoptosis and cycle of SH-SY5Y cells [ref. 582]; regulates neuropathic pain and neuroinflammation [ref. 583]; regulates cell cycle in CRC [ref. 584]; induces endothelial dysfunction and decreases lung injury, inflammation and permeability and improved hemodynamics [ref. 585]; regulates skeletal muscle anabolism [ref. 586]; regulates apoptosis in THCA [ref. 587]; interacts with environmental factors, such as maternal stress during pregnancy, neonatal jaundice and family psychiatric history, to impact risk of ASD [ref. 588]; stimulates cardiomyocyte apoptosis [ref. 589]. |
All sequences were retrieved from https://mirbase.org/ (accessed on 22 April 2022). The top 10 miRNAs in both colostrum and mature milk are highlighted in green. Abbreviations: AD, Alzheimer’s disease; ASD, autism spectrum disorder; ATG, autophagy related; BDNF, brain-derived neurotrophic factor; ESCC, esophageal squamous cell carcinoma; MSCs, mesenchymal stem cells; DPMSCs, dental pulp-derived MSCs; Treg, regulatory T cells; ccRCC, clear cell renal cell carcinoma; NSCLC, non-small cell lung cancer; IGF, insulin-like growth factor; FGFR3, fibroblast growth factor receptor 3; GC, gastric cancer; GHR, growth hormone receptor; SOCS-7, suppressor of cytokine signaling-7; EMT, epithelial-to-mesenchymal transition; HBP4, hyaluronan binding protein 4; HIF, hypoxia-inducible factor; JAK2, janus kinase 2; ALL, acute lymphoblastic leukemia; ADH4, alcohol dehydrogenases; BC, breast cancer; LSCC, lung squamous cell carcinoma; TGF, transforming growth factor; TNBC, triple negative BC; THCA, thyroid cancer; SNRPB, small nuclear ribonucleoprotein-associated protein B; PC, pancreatic cancer; PTC, papillary thyroid cancer; PASMCs, pulmonary artery smooth muscle cells; AML, acute myeloid leukemia; HCC, hepatocellular carcinoma; CRC, colorectal cancer; CML, chronic myeloid leukemia; HDAC4, histone deacetylase 4; KIR, killer immunoglobulin-like receptor; LAMB3, laminin subunit beta 3; MS, multiple sclerosis; MAP4K4, mitogen-activated protein kinase 4; NPC, nasopharyngeal carcinoma; OSCC, oral squamous cell carcinoma; PCOS, polycystic ovary syndrome; RA, rheumatoid arthritis.
5.3. Variability in miRNA Expressions in HBM
It has been reported that HBM miRNAs are differentially expressed during lactation stages, for example, the notable drop in the expression level of miR-181a and miR-155 after 6 months of lactation [ref. 44]. In a study involving 33 matched samples, the total concentration of miRNA in the fraction of colostrum whey was 87.78 ng/L, which was significantly higher than that in the fraction of mature milk whey (33.15 ng/L). miRNA-378 miRNA-30B and Let-7a were highly expressed in colostrum (4.64, 4.05 and 2.58, respectively) and mature milk (3.62, 4.92 and 2.39, respectively). However, the levels of miRNA-378 and let-7a significantly decreased with the lactation period, while levels of miRNA-30B in mature milk were higher than in colostrum [ref. 45].
The change of miRNA content in pre- and post-feeding is a consequence of the change of milk content (such as increase in the cells and fat content) during breastfeeding [ref. 590,ref. 591], where high content and composition of miRNAs are found in post-feeding [ref. 186]; which indicates that breastfeeding enhances the content of miRNAs in HBM. The milk cells and fat contain higher amounts of miRNAs. Those components are elevated in post-feeding due to cell turnover during breast sucking, migration of epithelial cells into milk channels and the process of milk synthesis [ref. 590,ref. 592]. Unlike miRNAs related to the cell content, elevated miRNAs related to milk fat are significantly correlated with milk volume intake by the infant [ref. 186].
Furthermore, in premature infant delivery, an exclusive profile of HBM miRNA with adaptive metabolic targets and functions for growth in premature infants was reported [ref. 46]. Several different physiological challenges may occur for premature infants compared to fully mature infants since they require different nutritional needs. There are several significant differences in the expression of 113 miRNAs in skim and lipid samples of mothers of preterm (pMBM) and term infants (tMBM) [ref. 46]. The regulation that occurs within the mammary epithelial cell nucleus may play a significant role in the differences in the miRNA composition of pMBM and tMBM. Furthermore, the environmental changes, including abrupt premature delivery, may partially alter the miRNA packaging and extrusion into MBM, which increases the differences in the composition [ref. 46]. For example, miRNAs are packaged in several ways, such as shedding, vesicles, RICS–complex protein binding and exosomal transfer [ref. 593]. Since each miRNA has a high affinity for specific packaging mechanisms [ref. 594], a difference in the carrier ratio may impact the specific secretion of miRNAs in pMBM but not in tMBM and thus increase the differences in macronutrient and micronutrient composition of both milks [ref. 88].
Premature delivery, on the other hand, could affect miRNA production within the cell nucleus. The change in maternal hormones could alter the transcription of miRNAs, given the hormonal changes that occur pre- and post-partum. Lactogenic hormones, for example, influence the expression of miRNA and its secretion in cultured mammary cells [ref. 165]. Mothers of preterm infants had reduced levels of prolactin [ref. 164], which may affect miRNA expression and release. Moreover, the exposure to estrogen hormone changes the content of miRNA in breast cells [ref. 595], while the exposure to progesterone changes the processing machinery concentrations of miRNA, which could result in a changed miRNA profile [ref. 596]. During pregnancy, the levels of estrogen and progesterone increase; however, lower circulating levels of both hormones in mothers of premature infants at delivery may permanently affect the miRNAs in pMBM and thus enhance the potential evolutionary advantages for the premature neonate, such as influencing glucose homeostasis, regulation of adipogenesis and B-cell proliferation [ref. 46].
A high-fat diet during pregnancy alters miRNA expression [ref. 597]. Target pathway analysis indicated that changes in miRNA expression due to changes in food consumption might affect the metabolic pathways of either mothers or newborns. High galactose and glucose diets had no significant effect on miRNA species in the milk of mothers [ref. 189]. However, the miR-27 and miR-67 expressions were significantly raised under a high-fat diet compared to a high carbohydrate diet [ref. 189], which indicated that HBM has a good epigenetic potential in breastfed infants. The changes in the type and quantity of miRNA expression in HBM are considered as a dynamic maternal regulation of infant gene expression based on environmental changes with significant maternal diet distresses. Lactation also enhances the adaptation of mothers and offspring to changes in food supply, which would potentially support an evolutionary advantage for the offspring [ref. 598]. Moreover, the impact of both maternal and post-natal diets on modification in the offspring’s hepatic epigenome in animal primate models has been reported [ref. 599,ref. 600]. It has been found that HBM composition differs with gestational period at delivery and through the first six months of infant age [ref. 601]. It is reasonable to suppose that the expression of miRNA would also change during the period of lactation until the weaning of the infant.
6. Immunoregulatory Roles of HBM-Derived miRNAs
In addition to the biological functions of miRNAs in cell differentiation, metabolism, proliferation, apoptosis, homeostasis and protection from some diseases [ref. 602], they are also considered immunoregulatory elements since they play a vital role in differentiation and regulation of the immunological functions in both adaptive and innate immunity [ref. 603]. More than 65% of HBM-derived miRNAs are related to immune function [ref. 188]. Most miRNAs in HBM are plentiful and known to have immunomodulatory functions (Table 3). These functions are summarized in Figure 6.
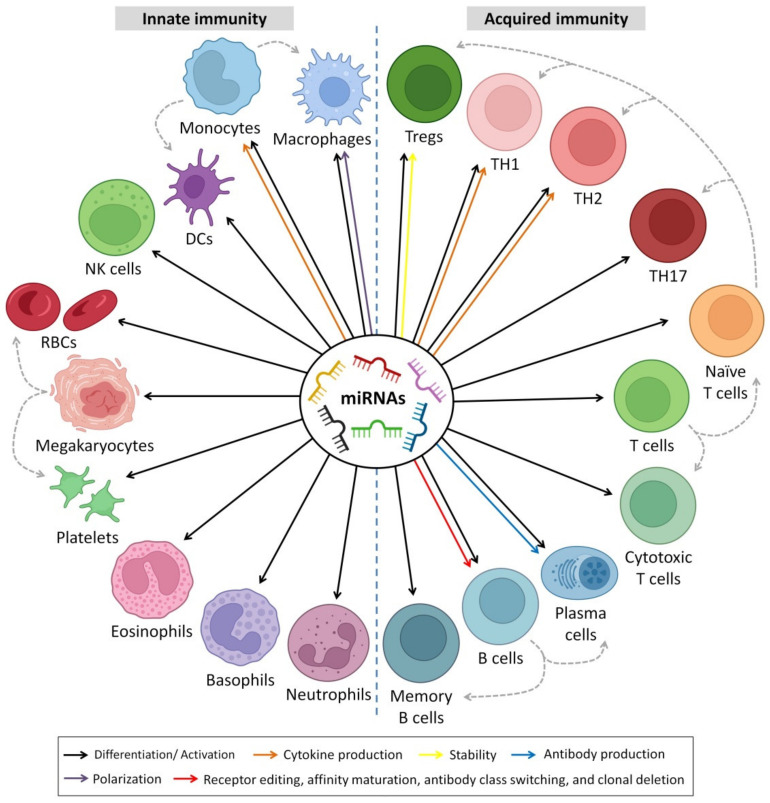
HBM-derived miRNAs play a substantial role in the early immune system maturation of infants. Several studies have examined the relationship between miRNAs and innate and adaptive immunological responses [ref. 603,ref. 616]. According to the Pathway Central database’ annotation (SABiosciences, Frederick, MD, USA), 4 of the top 10 most frequently expressed unique miRNAs (miR-148a-3p, miR-30b-5p, miR-182-5p and miR-200a-3p) are identified as immune-related pre-miRNAs [ref. 188]. They showed that milk miRNAs affect T and B cell development [ref. 617], neutrophil and monocyte proliferation [ref. 618], inflammatory mediators’ secretion [ref. 619] and macrophage differentiation [ref. 620]. In the mammalian immune system, control of miRNAs has emerged as a fundamental regulatory factor, with any dysregulation leading to immunological disorders and malignancies [ref. 3,ref. 10,ref. 13,ref. 621]. For example, the miRNA clusters miR-92 and miR-17 have been found in high concentrations in HBM, indicating their responsibility in the regulation of monocyte development as well as the maturation and differentiation of B and T cells [ref. 617,ref. 622]. Furthermore, miR-30b-5p increases the cellular invasion and immunosuppression [ref. 188,ref. 623], and miR-182-5p enhances T cell-mediated immune responses [ref. 624], whereas miR-200a-3p is associated with Hodgkin’s lymphoma [ref. 625].
MiR-223 is a hematopoietic-specific miRNA that interacts with lineage-specific transcription factors in regulatory signaling networks. In CD34+ human hematopoietic progenitors (HPCs) undergoing unilineage differentiation, miR-223 is increased more than 10-fold during granulopoiesis, 3-fold during monocytopoiesis and kept at low levels during erythropoiesis [ref. 626]. Perri et al. reported the presence of miR-223 (together with miR-181a) colostrum and HBM and suggested that they operate as selective targets on populations of T cells and granulocytes. As a result, these biomolecules may have an early impact on the immunological homeostasis of newborns. While there was variance in immune-related miRNAs in HBM across breastfeeding women, there was none in colostrum [ref. 627]. Furthermore, MiR-223 is thought to play a role in obstructive lung disease as altered expression levels have been observed in both asthma and chronic obstructive pulmonary disease (COPD) [ref. 628]. Moreover, miR-223 has been shown to be a potential diagnostic and prognostic marker for many cancers, and it has been reported to suppress osteosarcoma cell proliferation in vitro [ref. 629].
Furthermore, HBM contains significant quantities of miR-223, which is believed to trigger granulocyte proliferation [ref. 630]. B cell-related miRNAs, such as miR-155 and miR-181, are abundant in HBM [ref. 631,ref. 632], and they may trigger B cell differentiation. MiR 150, on the other hand, is known to behave as a suppressor of B cells [ref. 633,ref. 634], despite its lower concentration in HBM. Interestingly, Zhou et al. identified a large number of miRNAs in HBM exosomes [ref. 188]. Four miRNAs among the top abundant ten (i.e., miR-182-5p, miRNAs, miR-30b-5p, miR-148a-3p and miR-200a-3p) were linked with immunological processes [ref. 188]. MiR-30b-5p, in particular, induces immunosuppression and inhibits activation [ref. 623]. In contrast, miR-182-5p stimulates immune responses of T cells [ref. 624]. About 59 pre-miRNAs out of 87 (detected in HBM exosomes) showed immunological functions [ref. 188]. The miR-17-92 cluster, which was also extremely expressed in HBM exosomes, behaved as a developmental regulator of the immune system [ref. 635].
Several miRNA molecules involved in B-cell proliferation pathways, for example, the high expression of the miR-17-92 cluster, are associated with improving B-cell propagation and survival [ref. 635]. Furthermore, elevated CD19+ B cell expansion was noticed after ectopic high miR-181 levels [ref. 636]. In some cases, abnormal elevation of miRNA leads to B-cell tumorigenesis, such as Hodgkin’s lymphoma [ref. 637], Burkitt lymphoma [ref. 638] and lymphoblastic leukemia [ref. 639]. The regulatory mechanisms of miRNAs to B-cells are not limited to enhancing cell proliferation and survival; some miRNAs exert normal controlling functions when expressed at low levels. For example, miRNA-150 interferes with the nuclear transcription factor gene c-Myb, which involves B-cell differentiation [ref. 633,ref. 634]. The first study to understand the role of miRNA in B-lymphocyte differentiation stages was on the protein-coding gene argonaute RISC catalytic component 2 (AGO2), which leads to cells stuck at the pre-B-cell stage and failure of successful progression to mature B lymphocytes [ref. 622]. The AGO2 is important to the synthesis and functioning of miRNAs in hematopoietic stem cells AGO2 [ref. 622,ref. 640]. Moreover, the study showed that the Dicer gene deletion, an important gene for RNA interference molecules biogenesis, led to defects in B-lymphocyte differentiation, programmed cell apoptosis and antibody production [ref. 622].
Early T progenitor cells, with the deletion of the Dicer gene, led to a massive decrease in mature T cells without altering the expression patterns of CD8 and CD4 markers during T cell maturation [ref. 641]. Whereas deletion of the Dicer gene in the single-positive stage led to less reduction in T cell count in comparison with deletion at early stages [ref. 642]. miRNA-181 is involved in signal transduction during T cell differentiation and subsequently enhances positive and negative selection [ref. 643], sensitizing the T cell receptor to stimuli [ref. 643] and reaching hemostasis in cases of over-expression of T cells [ref. 635]. MiR-101 regulates the post-transcription of CD278; abnormal alteration of miRNA-101 leads to autoimmunity disease by the production of effector T cell (Teff) phenotype [ref. 644]. Targeting miRNA-155 to the protein-coding gene suppressor of cytokine signaling 1 (SOCS1) improves the response of regulatory T-lymphocytes to IL-2, which enhances cell survival [ref. 645].
In addition to the role of miRNAs in adaptive immune response, miRNAs are involved in several mechanisms in the innate immune response. miRNA-223 controls granulocytic differentiation and granulopoiesis [ref. 646]. Induced ablation of miRNA-223 leads to an elevated number of granulocyte progenitors and neutrophil hyperactivity, which leads to spontaneously developing inflammatory and exaggerated tissue destruction [ref. 630]. MiRNA-125 interferes with tumor necrosis factor-ɑ (TNF-ɑ) gene; therefore, a low expression level of miRNA-125 is required to establish a macrophage-mediated inflammatory response [ref. 647]. It has been reported that miR-146b-5p targets NF-κB signaling in innate immune responses [ref. 648]. The interplay of miRNA action mechanisms and their effect on downstream gene expression is not clear, especially those genes involved in innate immunity [ref. 649]. MiRNA-155, on the other hand, is found in significant abundance in HBM and has a regulatory role in cellular (B and T cells) and innate immune response. Moreover, some miRNAs may have roles in reshaping immune responses against microbial infections [ref. 650]. For example, it has been reported that miR-29a-3p can suppress the immune responses to intracellular pathogens by targeting IFN-γ [ref. 651].
Toll-like receptors (TLRs) are proteins that show a crucial function in the innate immune and digestive systems [ref. 652]. TLRs are a large collection of receptors that range from TLR1 to TLR13 [ref. 653,ref. 654]. HBM also inhibits the TLR signaling pathways of the intestinal epithelial cells, lowering the risk of enteric inflammation [ref. 102]. The presence of TLR regulatory components in HBM promotes the use of safe oral prophylactic and therapeutic treatments for inflammatory bowel disease and other gastrointestinal inflammatory disorders caused by aberrant TLR signaling. This was shown by inflammation suppression in rat gut models by using HBM [ref. 122]. It was found that miRNAs have a significant role in modulating TLRs; for example, the miR-146 (present in HBM) targets Traf6 and Irak1, components of the TLR signaling pathway activated by LPS, suggesting a negative feedback loop [ref. 655]. This field of research is still immature, and extensive investigations are needed to solve the mysteries behind the effects of breastfeeding, as these studies may be useful for manufacturing additives for formulas.
Furthermore, miRNA can influence the development or prevention of autoimmune disorders such as inflammatory bowel disease (IBD) and play a significant role in their development or prevention [ref. 49,ref. 656]. miRNAs could potentially be applied as biomarkers of milk to identify disorders in the immune system, including allergic illnesses [ref. 657,ref. 658]. Kosaka et al. [ref. 49] found high levels of miRNAs in HBM during the first 6 months of lactation with immune system functions; these miRNAs include miR-150, miR-181a, miR-155, miR-17 and miR-223. MiR-155 and miR-181, which are the most common in the control of B cell variation [ref. 607,ref. 635], were found in high concentrations in HBM [ref. 49,ref. 189], implying that they have a role in the immune system development of the infant.
7. Breastfeeding and Epigenetics
Epigenetics reflect all molecular mechanisms transforming the expression of genotype into phenotype [ref. 659]. This occurs by covalent modifications of DNA by methylation of cytosine [ref. 660], mainly at CpG dinucleotides [ref. 661]; adenine and guanine methylation [ref. 660]; or histone protein modification by deacetylation, methylation or phosphorylation [ref. 662], which regulates gene expression by chromatin remodeling [ref. 663]. The human epigenome functions as a connection between the inheritable genetic information of humans and its response to environmental factors. Furthermore, variations in human epigenome patterns have a key role in individual response and susceptibility to future toxicant exposure and consequent disease outcomes. Moreover, the epigenetic system consists of nuclear information, which is heritable during cell division and is responsible for controlling cell development, cellular responsiveness and tissue differentiation [ref. 664].
7.1. MiRNAs–Mediated Epigenetics and Immunity
Over the last few years, many researchers have reported interesting associations between a number of miRNAs with epigenetic alterations leading to the occurrence of various diseases such as cancers [ref. 665]. Furthermore, it has been shown that miRNAs are controlled by epigenetic mechanisms; they were also shown to have a reciprocal role in epigenetic regulations. Some of the miRNAs are called epigenetic-miRNAs (epi-miRNAs) as they can control and regulate the epigenetic enzymes and regulators [ref. 666,ref. 667]. On the other hand, epigenetic enzymes can control the expression level of tumor suppressor miRNAs, and vice versa, these enzymes can also be regulated by reverse responses of the targeted miRNA. Thus, since epi-miRNAs can affect gene expression, many studies showed that epi-miRNA could serve as a fascinating therapeutic tool for diseases initiated by epigenetic dysregulation, such as cancers [ref. 665,ref. 668].
It was shown that miRNAs could regulate and affect epigenetic DNA-methylation by targeting DNA methylation enzymes (DNA methyltransferases; DNMTs). DNMT-3A and B serve as targets for miRNA; miRNA-29 family members were the first discovered as epi-miRNAs due to their direct influences on DNMT-3A and B in lung cancer [ref. 666]. In this study, they found that the miRNA-29 family, with its subtypes 29a, 29b and 29c, has base pairing complementary to the 3′ end of UTRs in DNMT-3A and B. Thus, the expression of miR-29s led to decreased DNMT3A and B expression in lung cancer, supporting the vital role of miR-29s as epigenetic regulators. Furthermore, another study showed that the miRNA-290 family is mainly found in mammalian placenta, which directly targets and regulates the expression of post-transcriptional factor Rbl2 gene, which acts as a repressor for DNMT 3A and 3B leading to hypomethylation [ref. 669]. Thus, miRNA-290 indirectly acts as a regulator for DNA methylation via targeting transcriptional factors.
Moreover, Khraiwesh et al. proposed that the initiation of epigenetic silencing by DNA methylation is controlled by the ratio of the miRNA to its target mRNA [ref. 670]. In addition to the regulatory effects of miRNAs on DNA methylation, miRNAs can also mediate histone deacetylases (HDACs) modifications. For example, miRNA-1, miRNA-206 and miRNA-140 directly affect HDAC4 regulation. HDAC4 was shown to be a direct target of miR-1 and miR-140 in several research works [ref. 671,ref. 672]. HDAC4 contains two binding sites for miRNA-1 at its 3′UTR. Another study by Williams et al. [ref. 673] showed that amyotrophic lateral sclerosis (ALS) progression disease extensively depends on miRNA-206 level; miR-206 is shown to delay the progression of ALS by targeting HDAC4. The mechanisms of epigenetic regulation are shown in Figure 7.
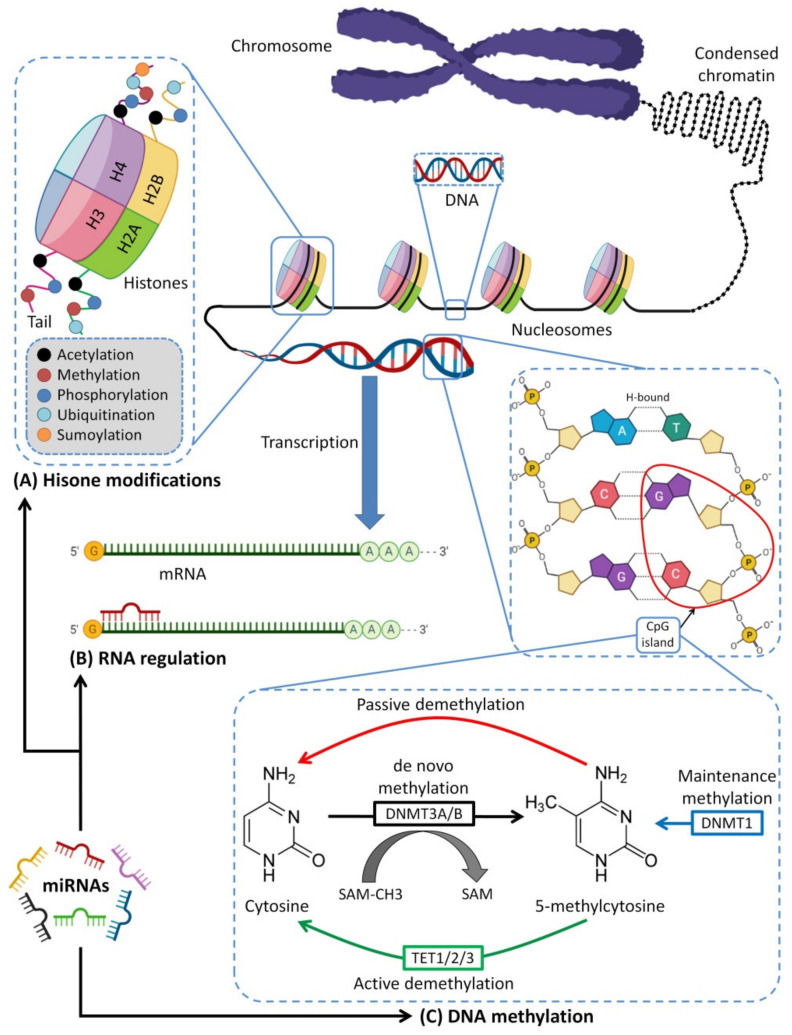
The initiation of distinct innate and adaptive immune responses can be affected and regulated by epigenetic mechanisms [ref. 682]. Epigenetic modifications can affect immune responses by improving the expression of immune-related genes and tumor-associated antigens, besides controlling the secretion of inflammatory mediators such as cytokines and chemokines that participate in the activation of the immune system [ref. 683]. Thus, the epigenetic system can produce more prominent immune responses and control the magnitude of inflammation and host immune responses. Moreover, epigenetics systems control other functions in immunity, such as producing monocyte-derived DCs, playing a role in the differentiation of “helper” CD4+ T cells and regulatory T cells, generating a memory T cell phenotype and also generating antibodies using plasma cells [ref. 682]. Epigenetic modifications, such as the regulation of cytokine production, DNA methylation/demethylation and numerous types of histone modifications in IFNG, IL-4 and IL-13 genes are involved in the differentiation of Th1 and Th2 cells from naive CD4+ T cells. The promoter of INFG is hypermethylated in naïve T cells and is only demethylated when the differentiation occurs in Th1 cells, while the promoter of IL4 is hypermethylated in naive T cells and Th1 cells, and only partial demethylation for Th2 cells [ref. 684,ref. 685].
The epigenetic mechanisms that are supposed to control the memory of the environmental impact may also promote the persistence of disease-associated phenotypes, even in the lack of the initial trigger [ref. 682]. There are also various examples of the link between epigenetic changes and autoimmune disorders. DNA hypomethylation of histone deacetylase (HDAC) 1 and 2 levels, hyperacetylation of histones 3 and 4 and decreased methylation rate of histone 3 at lysine 9 (H3K9) have been observed in synovial tissues of patients with rheumatoid arthritis (RA). Additionally, in patients with multiple sclerosis (MS), decreased DNA methylation in the central nervous system white-matter has been detected in comparison to healthy individuals [ref. 686]. Moreover, in systemic lupus erythematosus disorder, the prime targets of auto-antibodies are hypomethylated apoptotic DNA and modified histones [ref. 687]. Many studies have established a strong association between epigenetics alterations and allergic conditions such as asthma [ref. 688]. In paternal origin traits, asthma and epigenetic changes have been found to be passed more from an asthmatic mother than an asthmatic father [ref. 689], which can be related to the direct immune interactions between the fetus and its mother [ref. 690].
Indeed, various functions of immune cells such as hematopoietic lineage, antigen-receptor rearrangement, exclusion of allelic and immune response induction against pathogens are epigenetically controlled. Furthermore, understanding the epigenetic mechanisms involved in regulating the immune response could be very useful in the explanation of the immunological processes involved in the rejection and allograft tolerance and in developing novel therapeutic approaches [ref. 691].
7.2. Epigenetic Effects of HBM
In DNA methylation, a methyl group is added to the fifth position of the cytosine ring [ref. 10]. In many organisms, DNA methylation is essential for normal organism growth and cellular differentiation. The gene expression pattern in cells is permanently altered by DNA methylation. For example, cells programmed to be liver cells during embryonic development remain as liver cells throughout the organism’s life. DNA methylation is most commonly found in the context of the CpG dinucleotide. In mammals, over 75% of all CpGs are methylated. Over time, methylated C residues spontaneously deaminate to yield T residues. Therefore, CpG dinucleotides slowly mutate to TpG dinucleotides, as indicated by the human genome’s underrepresentation of CpG dinucleotides (only 21% of the expected frequency). Spontaneous deamination of unmethylated C residues, on the other hand, results in U residues, a mutation that the cell rapidly recognizes and corrects [ref. 692,ref. 693]. MiRNA-mediated post-transcriptional regulation and transcriptional control by epigenetic changes work together to regulate gene expression and sustain physiological functioning. If this circuit is interrupted, it can lead to a variety of diseases [ref. 10,ref. 173,ref. 190].
It was shown that breastfeeding affects DNA methylation of the human genome, especially genes that are involved in the immune response, particularly innate immunity attributed mainly to miRNA-148a-3p, miRNA-146b-5p and others [ref. 188]. A main function of 148a-3p is interfering with the function of DNA methyltransferase 3b (DNMT3b), which is important for de novo methylation during the embryonic stage of fetal development and for DNA methyltransferase 1 (DNMT1)-mediated methylation of the DNA after delivery [ref. 188,ref. 694]. It was found in mice where the knockout of DNMT3b promotes lymphomagenesis due to demethylation of the enhancer gene MENT (also known as Gm128) in normal thymocytes [ref. 695]. Changes in DNA methyltransferase (DNMT 1, DNMT 2 and DNMT 3) expression in the liver and skeletal muscle were shown to affect global DNA methylation in the offspring of pigs fed with a low-protein maternal diet [ref. 696,ref. 697,ref. 698,ref. 699]. These results may reveal the effect of the maternal diet on carbohydrate and fat metabolism. Figure 8 represents the main immunoregulatory functions of HBM-derived exosomal miRNA and their modulatory effects on DNTMs.
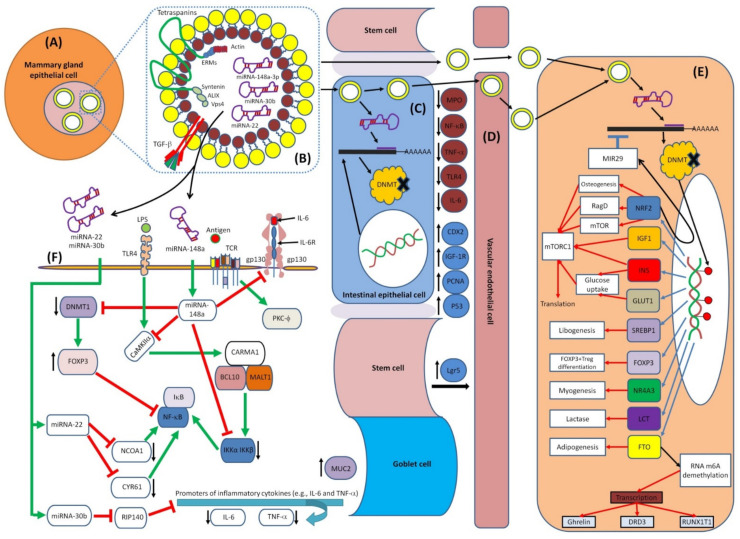
DNMT3b is required for genome-wide de novo methylation and the creation of DNA methylation patterns [ref. 719]. DNA methylation is coordinated with histone methylation. It may methylate nucleosomal DNA within the nucleosome core region preferentially, and it may act as a transcriptional co-repressor by interacting with CBX4. It appears to be involved in gene silencing and, in conjunction with DNMT1, to be involved in the stimulation of BAG1 gene expression via the recruitment of CTCFL/BORIS [ref. 720]. Figure 9 shows the main interactions of DNMT3b and DNMT1.
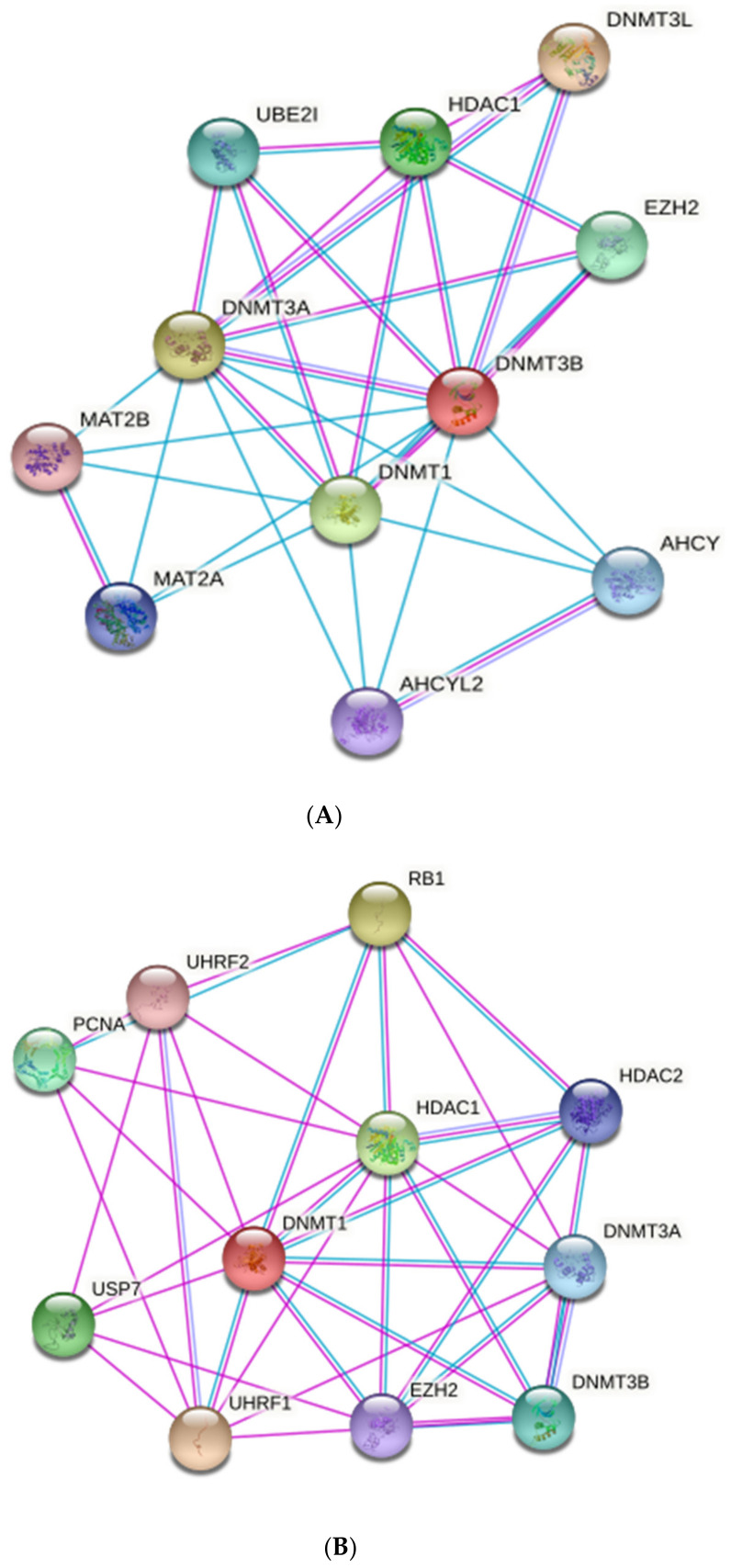
It is important to note that studies investigating the effects of breastfeeding are considerably diverse in terms of participant age, tissue investigated, DNA methylation targets and methodologies used for DNA methylation profiling, and breastfeeding categorization [ref. 727,ref. 728,ref. 729,ref. 730]. In a study where early exposures were investigated in relation to methylation of cancer-related genes in a case–control study of women with BC [ref. 728], it was found that premenopausal women who were never breastfed were nearly three times more likely to have promoter methylation in the p53 gene (an essential tumor suppressor) [ref. 728].
An epigenome-wide association study reported a link between breastfeeding duration and methylation levels at 4276 CpG sites, which are linked to 2635 genes [ref. 731]. These genes were primarily involved in the modulation of cell signaling systems, the formation of anatomical structures and cells and, most importantly, the development and function of the immune system and the CNS. These findings led to the conclusion that the oxytocin signaling pathway plays a unique role as a potential activator of coordinated epigenetic modifications in genes involved in CNS function in response to breastfeeding [ref. 731]. Breastfeeding appears to influence global methylation patterns, modulate epigenetic effects of some genetic variants and be negatively associated with promoter methylation of the leptin (LEP) (an anorexigenic hormone that regulates growth, hunger and insulin sensitivity), CDKN2A (implicated in tumor suppression) and Slc2a4 (which encodes an insulin-related glucose transporter) genes and positively associated with promoter methylation of Nyp gene (which produces an orexigenic neuropeptide) [ref. 732].
For the LEP gene, the methylation of its promoter was studied in toddlers in relation to breastfeeding duration [ref. 733]. Children who were breastfed for at least 1 to 3 months had lower LEP promoter methylation in white blood cells and higher serum levels of leptin than children who were never breastfed. Methylation of LEP was similarly reduced in children with higher birthweights [ref. 727]. Both total and exclusive breastfeeding duration were linked to DNA methylation at four LEP CpG sites at 10 years old but not at 18 years. Differential methylation region (DMR) analysis found five significant differentially methylated regions in the genome. The duration of breastfeeding was also linked to an early transitory overweight trajectory. LEP DNA methylation was also linked to this trajectory at one CpG position, and persistent obesity was linked to another; however, mediation analysis was not significant [ref. 733].
Furthermore, the impact of breastfeeding on newborn growth and methylation of obesity-related genes of buccal cells was investigated [ref. 734]. There was a significant difference in infant growth and buccal RXRA and LEP gene methylation after 12 months of breastfeeding. A positive association was identified between RXRA CpG2 methylation and breastfeeding duration. For CpG3 and CpG islands of the RXRA gene, methylation levels were significantly reduced for children breastfed for 4–6 months compared to non-breastfed ones (only CpG3), and those breastfed for 7–9, 10–12 or 1–3 months. However, children breastfed for 7–9 months (6.1%) had more LEP CpG3 methylation than those breastfed for 1–3 months (4.3%) and 10–12 months (4.6%). Furthermore, children who were breastfed for 10–12 months had much lower weight [ref. 734].
The relationship between the hypermethylation of sorting nexin 25 (SNX25) gene and allergic disease is exciting [ref. 735,ref. 736]. Increased promoter methylation causes a decrease in gene expression and, as a result, protein downregulation. TGF-β has been demonstrated to be downregulated by SNX25 [ref. 736,ref. 737]. TGF-β is a regulatory cytokine connected to immune regulation and inflammation that is detected in HBM. TGF-β has been implicated in the pathophysiology of allergic diseases [ref. 736,ref. 738,ref. 739].
Two differentially methylated positions (DMPs) in the genes SNX25 and LINC00840 were shown to be substantially linked with breastfeeding duration of more than 6 months, and the findings were reproduced for exclusive breastfeeding for more than 3 months. Furthermore, in 10-year-old children who were breastfed for more than three months, a substantial differentially methylated region (DMR) covering the FDFT1 gene was discovered [ref. 735]. During infancy, exclusive breastfeeding causes more significant DNA methylation changes than during later stages of child development. At the genome-wide analysis, 2 CpG sites in boys (NREP and IL16) and 13 CpG sites in girls (ATP6V0A1, DHX15/PPARGC1A, LINC00398/ALOX5AP, FAM238C, miR-21, SNAPC3, NATP/NAT2, CUX1, TRAPPC9, OSBPL1A, ZNF185, FAM84A, PDPK1) were found to mediate the association between longitudinal BMI and exclusive breastfeeding. Enrichment of CpG sites located within miRNAs and key pathways (AMPK signaling pathway, insulin signaling pathway and endocytosis) was detected. In a dose–response relationship with exclusive breastfeeding time, total DNA methylation variation corresponding to 3 to 5 months of exclusive breastfeeding was related to lower BMI growth in the first 6 years of life [ref. 740].
The number of weeks of breastfeeding in young women in the United Kingdom had minimal, non-statistically significant effects on methylation of the interleukin-4 receptor gene’s relevant CpG island, which was associated with asthma risk [ref. 730]. Various degrees of breastfeeding (none, less than 3 months, more than 3 months) were connected to different patterns of whole-genome methylation in a case–control study of asthma in 200 children from industrial vs. rural settings in the Czech Republic [ref. 729]. Another study looked at the age-related rise in global methylation in blood at birth, 7 and 17 years, in relation to a range of maternal, pregnancy and birth-related variables, such as whether the child had ever been breastfed. In this study, there was no significant association between breastfeeding and methylation differences [ref. 741].
Breastfeeding may potentially expose infants to epigenetic consequences from the mother’s environment or health habits. When compared to individuals who did not breastfeed, the methylation of DRD4 (a key dopamine receptor) in cheek cells was higher in eight-week-old children whose moms drank moderate amounts of alcohol during breastfeeding compared to those who did not drink [ref. 742]. Despite the fact that the brain is the most important tissue for studying dopamine-receptor methylation, sampling live infants is clearly invasive. Relapsed adult alcoholics exhibit equivalent alterations in the methylation of cheek-cell dopamine receptors because cheek cells are formed from the same primordial germ layer as the brain [ref. 743].
Separate linear regression models controlling for confounders were used to identify 87 differentially methylated CpGs in different breastfeeding and formula feeding children (exclusive breastfeeding (EBF): 27 CpGs, exclusive formula feeding (EFF): 48 CpGs and mixed: 12 CpGs) [ref. 744]. The EFF group had a significantly lower total of all methylation alterations from birth to the age of ten years old. As a result, the number of CpGs with a methylation reduction increased by 4.7% (13,683 CpGs). Future research is needed to lessen the negative health impacts of lower methylation associated with exclusive formula feeding and its negative potential for a child’s development [ref. 744]. Breastfeeding is linked to epigenetic changes in buccal cells in children. After controlling for child and maternal factors, four significant CpGs were related to breastfeeding in the subgroup of children less than ten years [ref. 745]. Methylation differences at these CpGs were smaller and non-significant in children beyond the age of ten years. Three of the previously published CpG sites were linked to breastfeeding in children under the age of ten years, indicating that these CpGs are linked to breastfeeding in buccal and blood cells [ref. 745].
Furthermore, researchers looked into the associations between breastfeeding length and DNA methylation at two sites in the promoter of the toll-like receptor-1 (TLR1) gene, as well as the link between TLR1 DNA methylation and illness risk [ref. 746]. Blood was drawn from 100 adults and divided into two groups based on the length of time they were breastfed (<6 months and ≥6 months), with 53 samples undergoing DNA extraction. This study found a significant association between longer breastfeeding length and decreased susceptibility to influenza and allergies, as well as a significant reduction in DNA methylation within the TLR1 gene promoter [ref. 746].
Researchers reported two differentially methylated sites to have directionally consistent associations with breastfeeding at the ages of 7 and 15–17 years, but not at birth [ref. 747]. Twelve differentially methylated regions were found in relation to breastfeeding, three of which showed signs of directional concordance with ages 7 and 15–17 years, but not at birth and age 7 years [ref. 747]. A study investigated whether DNA methylation, which is influenced by dietary intake, could play a role in the link between breastfeeding and child cognition [ref. 748]. The goal was to see if there was an association between global DNA methylation and cognition and behavior in 4-year-old children (73 children). No association was found between global DNA methylation and child behavior and cognition [ref. 748].
The impact of dietary intervention in the first year of life on global methylation levels in leukocyte samples from a group of infants born in southern Brazil between 2001 and 2002 was investigated [ref. 749]. Overall methylation assessments were performed on DNA samples from 237 4-year-old children using enzyme-linked immunosorbent assays. The intervention group’s mean methylation values were greater compared to the control group. As a result, these factors may play a role in the rise in global DNA methylation. The findings of this study support the idea that nutritional interventions should be made early in life [ref. 749]. In another study, Anderson et al. [ref. 750] found that maternal vitamin D supplementation affects the methylation of genes involved in developmental processes in mothers and breastfed babies. Loss of methylation was linked to genes involved in metabolic functions and signal transduction pathways. [ref. 750]. Table 4 summarizes studies that investigated the epigenetic effects of breastfeeding on different physiological and pathological functions.
Table 4: List of studies that investigated the epigenetic effects of breastfeeding on different physiological and pathological functions in infants and mothers.
| Disease/Condition | Type of Study | Target(s) | Study Criteria and Participants | Main Findings | Reference |
|---|---|---|---|---|---|
| Cancer | Case-control | The p53 gene (the guardian of the genome) | In archived tumor blocks from 803 cases, the promoter methylation of the p16 gene in connection to breastfeeding was examined | The p53 gene promoter was nearly three times more likely to be methylated in premenopausal women who had never been breastfed | [ref. 695] |
| Metabolism and growth | Cross-sectional | LEP gene | 120 Dutch children (50 girls) were included with average age of 1.4 years | Children who were breastfed for at least 1 to 3 months had lower LEP promoter methylation in white blood cells and higher serum levels of leptin than children who were never breastfed | [ref. 727] |
| Prospective observational cohort | RXRA and LEP genes | The effects of breastfeeding duration on infant growth and methylation in obesity-related genes of buccal cells (n = 101) were assessed | At 12 months, breastfeeding duration was associated with epigenetic changes in RXRA and LEP genes, as well as infant biometry and growth | [ref. 734] | |
| Cohort | LEP gene | 23 CpGs in the LEP gene in 297 samples of 10-year-old and 16 CpGs in 305 samples of 18-year-old were tested for association with breastfeeding duration | Breastfeeding length is associated with LEP methylation at 10 years of age and BMI trajectory; despite the small sample size, LEP DNA methylation is related to BMI trajectories throughout childhood | [ref. 733] | |
| Cohort | The protein coding genes FDFT1 and SNX25, as well as the ncRNA gene LINC00840 | A comprehensive Epigenome-Wide Association Study to identify associations between breastfeeding and DNA methylation patterns in childhood (at birth, 10, 18 and 26 years) was performed. Breastfeeding durations of >3 months and >6 months, as well as exclusive breastfeeding durations of >3 months, were used to categorize the feeding. | In 10-year-old children who were breastfed for more than three months, a substantial differentially methylated region covering the gene FDFT1 was discovered | [ref. 735] | |
| BMI and weight | Cohort | A total of 2 CpG sites in boys (NREP and IL16) and 13 CpG sites in girls (ATP6V0A1, DHX15/PPARGC1A, LINC00398/ALOX5AP, FAM238C, miR-21, SNAPC3, NATP/NAT2, CUX1, TRAPPC9, OSBPL1A, ZNF185, FAM84A, PDPK1) were investigated | The study comprised 15,454 pregnancies, with 15,589 known fetuses, 14,901 of whom were alive at one year. A total of 12,761 children were available for study after twins (n = 201) and children missing anthropometric measurements or age information were removed. The kids were tracked for more than two decades | CpG sites were shown to be enriched in miRNAs and critical pathways (AMPK signaling, insulin signaling and endocytosis). When compared to no breastfeeding, DNA methylation variation corresponding to 3 to 5 months of exclusive breastfeeding was linked to lower BMI growth in the first 6 years of life | [ref. 740] |
| Lung diseases and asthma | Cohort | This gene is linked to asthma risk | Blood samples were collected from 245 females at age 18 years randomly selected for methylation analysis from a birth cohort (n = 1456) | The number of weeks of breastfeeding had minor impacts on methylation of the interleukin-4 receptor gene’s relevant CpG island | [ref. 730] |
| Neurological disorder | Cohort | DRD4 gene | The data came from a large population-based triple B pregnancy cohort study (n = 844) that included thorough information on maternal alcohol intake throughout pregnancy and in the early postpartum period. The methylation of the DRD4 promoter DNA was investigated | The methylation of a DRD4 (a key dopamine receptor) in cheek cells was higher in eight-week-old children whose moms drank moderate amounts of alcohol during breastfeeding compared to those who did not drink | [ref. 742] |
| Cohort (bioinformatics study) | Many genes, particularly those involved in oxytocin signaling pathway | Investigating the association of breastfeeding and DNA methylation in the peripheral blood cells of 37 children aged 9 months to 4 years | In response to breastfeeding, the oxytocin signaling pathway serves a unique role as a possible activator of coordinated epigenetic alterations in genes essential to CNS function | [ref. 751] | |
| Immunity and allergy | Cross-sectional | TLR1 gene | In 57 adult adults, DNA methylation at two locations in the promoter of the TLR1 gene, as well as the relationship between DNA methylation of the TLR1 gene and illness susceptibility, were studied | The promoter of the TLR1 gene showed a considerable reduction in DNA methylation. There was no link discovered between DNA methylation and illness vulnerability | [ref. 746] |
Munch et al. [ref. 189] found that the genetic targets of 308 miRNAs in HBM had different functions, including gene expression and metabolic control, as well as immunological responses, implying that these miRNAs may be important for HBM-fed infants [ref. 189,ref. 752]. In addition to these roles, several miRNAs are thought to play a role in central nervous system (CNS) regulation. It has also been observed that HBM miR-118.2 targets Teneurin Transmembrane Protein 2 (TENM2), which is found in elevated concentrations in the CNS [ref. 753], implying a regulatory role in the infant’s brain nervous system connection development [ref. 754,ref. 755]. MiRNAs generated from milk can also be used to target adipogenesis in infants. Overexpression of miRNA-155 may target the adipogenic transcription factor CCAAT/enhancer-binding protein-β (C/EBPβ), which results in reducing the mass of brown adipose tissue [ref. 756]. Furthermore, milk-derived miRNA-29a is able to inhibit the INSIG-1 gene [ref. 189,ref. 190,ref. 757], which regulates adipogenesis processes [ref. 758]; this has been linked to body mass index (BMI) [ref. 759]. In addition, milk miRNAs also play a significant role in the control of short- and long-term appetite of breastfed infants via numerous appetite regulatory hormones of HBM, such as adiponectin, leptin, insulin, ghrelin and others [ref. 760].
HBM-derived miRNAs have been hypothesized to protect children from cancer from birth to adulthood [ref. 189]. MiR-21, for example, is found in both human and bovine milk [ref. 188] and has been shown to be overexpressed in human hepatocellular carcinoma (HCC). Any dysregulation of miR-21 can be linked to HCC increase by altering mTORC1 signaling, such as PTEN expression [ref. 761]. MiR-21 is a common miRNA found in bovine milk [ref. 757], as well as mature and colostrum HBM [ref. 190]. It is also plentiful in human plasma [ref. 762] and is assumed to have a role in postnatal growth promotion in newborns [ref. 705]. MiR-21 also has additional normal tissue activities, such as regulating adipogenic development in human adipose tissue mesenchymal stem cells (MSCs) [ref. 763]. Furthermore, HBM miRNAs may influence tumor suppressor genes directly [ref. 764], such as the let-7 family, which inhibits the growth of lung tumors by targeting the RAS oncogenes directly [ref. 765]. Further research into the natural roles of miRNA in the infant and the lactating breast is needed. Furthermore, based on the target predictions of novel-miR-118 in the nervous system, it seems that the presence of novel-miR-118.2 in HBM has neurocognitive advantages [ref. 189].
8. Conclusions
HBM contains several physiologically active components that aid in newborn growth, development and health. Most research studies have focused on milk exosomes and their miRNA content. The miRNAs in HBM emerged as potential immune-regulatory agents targeting the immune cells, influencing the development of an infant’s immune system through immune modulation. Moreover, miRNAs play crucial roles in modulating the expression of a wide range of genes by targeting DNA methyltransferases. Physiological miRNA transfer during breastfeeding delivers suitable signals for sufficient epigenetic programming of newborns.
References
- M.S. Kramer. “Breast is best”: The evidence. Early Hum. Dev., 2010. [DOI | PubMed]
- Infant and Young Child Nutrition: Global Strategy on Infant and Young Child Feeding
- I. Labayen, J.R. Ruiz, F.B. Ortega, H.M. Loit, J. Harro, I. Villa, T. Veidebaum, M. Sjostrom. Exclusive breastfeeding duration and cardiorespiratory fitness in children and adolescents. Am. J. Clin. Nutr., 2012. [DOI | PubMed]
- M. Boronat-Catalá, J.M. Montiel-Company, C. Bellot-Arcís, J.M. Almerich-Silla, M. Catalá-Pizarro. Association between duration of breastfeeding and malocclusions in primary and mixed dentition: A systematic review and meta-analysis. Sci. Rep., 2017. [DOI | PubMed]
- H.E. Montgomery-Downs, V.M. Crabtree, O. Sans Capdevila, D. Gozal. Infant-feeding methods and childhood sleep-disordered breathing. Pediatrics, 2007. [DOI | PubMed]
- H. Kalies, J. Heinrich, N. Borte, B. Schaaf, A. von Berg, R. von Kries, H.E. Wichmann, G. Bolte. The effect of breastfeeding on weight gain in infants: Results of a birth cohort study. Eur. J. Med. Res., 2005. [PubMed]
- M. Brown Belfort. The Science of Breastfeeding and Brain Development. Breastfeed. Med., 2017. [DOI | PubMed]
- N.A. Lund-Blix, S. Dydensborg Sander, K. Størdal, A.-M. Nybo Andersen, K.S. Rønningen, G. Joner, T. Skrivarhaug, P.R. Njølstad, S. Husby, L.C. Stene. Infant Feeding and Risk of Type 1 Diabetes in Two Large Scandinavian Birth Cohorts. Diabetes Care, 2017. [DOI | PubMed]
- G.A. Biks, Y. Berhane, A. Worku, Y.K. Gete. Exclusive breast feeding is the strongest predictor of infant survival in Northwest Ethiopia: A longitudinal study. J. Health Popul. Nutr., 2015. [DOI | PubMed]
- H. Ozkan, F. Tuzun, A. Kumral, N. Duman. Milk kinship hypothesis in light of epigenetic knowledge. Clin. Epigenetics, 2012. [DOI | PubMed]
- L. Moran, J. Gilad. From folklore to scientific evidence: Breast-feeding and wet-nursing in islam and the case of non-puerperal lactation. Int. J. Biomed. Sci. IJBS, 2007. [PubMed]
- H. Ozkan, F. Tuzun, S. Taheri, P. Korhan, P. Akokay, O. Yılmaz, N. Duman, Y. Özkul, E. Tufan, A. Kumral. Epigenetic Programming Through Breast Milk and Its Impact on Milk-Siblings Mating. Front. Genet., 2020. [DOI | PubMed]
- M.S. Kramer, R. Kakuma. Optimal duration of exclusive breastfeeding. Cochrane Database Syst. Rev., 2012. [DOI]
- G. Delli Carpini, S. Morini, M. Papiccio, M. Serri, V. Damiani, C. Grelloni, N. Clemente, A. Ciavattini. The association between childbirth, breastfeeding, and uterine fibroids: An observational study. Sci. Rep., 2019. [DOI | PubMed]
- P. Abedi, S. Jahanfar, F. Namvar, J. Lee. Breastfeeding or nipple stimulation for reducing postpartum haemorrhage in the third stage of labour. Cochrane Database Syst. Rev., 2016. [DOI]
- B. Figueiredo, C.C. Dias, S. Brandão, C. Canário, R. Nunes-Costa. Breastfeeding and postpartum depression: State of the art review. J. Pediatr., 2013. [DOI]
- S. Ahn, E.J. Corwin. The association between breastfeeding, the stress response, inflammation, and postpartum depression during the postpartum period: Prospective cohort study. Int. J. Nurs. Stud., 2015. [DOI | PubMed]
- E. Gouveri, N. Papanas, A.I. Hatzitolios, E. Maltezos. Breastfeeding and diabetes. Curr. Diabetes Rev., 2011. [DOI | PubMed]
- O. Ballard, A.L. Morrow. Human milk composition: Nutrients and bioactive factors. Pediatr. Clin. N. Am., 2013. [DOI]
- K.E. Lyons, C.A. Ryan, E.M. Dempsey, R.P. Ross, C. Stanton. Breast Milk, a Source of Beneficial Microbes and Associated Benefits for Infant Health. Nutrients, 2020. [DOI]
- J.D. Thai, K.E. Gregory. Bioactive Factors in Human Breast Milk Attenuate Intestinal Inflammation during Early Life. Nutrients, 2020. [DOI]
- S.Y. Kim, D.Y. Yi. Components of human breast milk: From macronutrient to microbiome and microRNA. Clin. Exp. Pediatrics, 2020. [DOI]
- R. Uauy, D.R. Hoffman, P. Peirano, D.G. Birch, E.E. Birch. Essential fatty acids in visual and brain development. Lipids, 2001. [DOI | PubMed]
- F. Haschke, N. Haiden, S.K. Thakkar. Nutritive and Bioactive Proteins in Breastmilk. Ann. Nutr. Metab., 2016. [DOI | PubMed]
- P.K. Berger, J.F. Plows, E.W. Demerath, D.A. Fields. Carbohydrate composition in breast milk and its effect on infant health. Curr. Opin. Clin. Nutr. Metab. Care, 2020. [DOI | PubMed]
- B.E. Young, J. Westcott, J. Kemp, L. Allen, D. Hampel, A.L. Garcés, L. Figueroa, S.S. Goudar, S.M. Dhaded, M. Somannavar. B-Vitamins and Choline in Human Milk Are Not Impacted by a Preconception Lipid-Based Nutrient Supplement, but Differ Among Three Low-to-Middle Income Settings-Findings From the Women First Trial. Front. Nutr., 2021. [DOI | PubMed]
- J.Y. de Vries, S. Pundir, E. McKenzie, J. Keijer, M. Kussmann. Maternal Circulating Vitamin Status and Colostrum Vitamin Composition in Healthy Lactating Women-A Systematic Approach. Nutrients, 2018. [DOI]
- C. Li, N.W. Solomons, M.E. Scott, K.G. Koski. Minerals and Trace Elements in Human Breast Milk Are Associated with Guatemalan Infant Anthropometric Outcomes within the First 6 Months. J. Nutr., 2016. [DOI | PubMed]
- N. Abbaspour, R. Hurrell, R. Kelishadi. Review on iron and its importance for human health. J. Res. Med. Sci., 2014. [PubMed]
- S. Khayat, H. Fanaei, A. Ghanbarzehi. Minerals in Pregnancy and Lactation: A Review Article. J. Clin. Diagn. Res., 2017. [DOI]
- M. Domellöf, B. Lönnerdal, K.G. Dewey, R.J. Cohen, O. Hernell. Iron, zinc, and copper concentrations in breast milk are independent of maternal mineral status. Am. J. Clin. Nutr., 2004. [DOI | PubMed]
- M.-E. Roumelioti, R.H. Glew, Z.J. Khitan, H. Rondon-Berrios, C.P. Argyropoulos, D. Malhotra, D.S. Raj, E.I. Agaba, M. Rohrscheib, G.H. Murata. Fluid balance concepts in medicine: Principles and practice. World J. Nephrol., 2018. [DOI | PubMed]
- A. Moretti. What is the role of magnesium for skeletal muscle cramps? A Cochrane Review summary with commentary. J. Musculoskelet. Neuronal Interact., 2021. [PubMed]
- A. Mazzocchi, M.L. Giannì, D. Morniroli, L. Leone, P. Roggero, C. Agostoni, V. De Cosmi, F. Mosca. Hormones in Breast Milk and Effect on Infants’ Growth: A Systematic Review. Nutrients, 2019. [DOI]
- F. Savino, S.A. Liguori, M.F. Fissore, R. Oggero. Breast milk hormones and their protective effect on obesity. Int. J. Pediatr. Endocrinol., 2009. [DOI | PubMed]
- M.A. Suwaydi, Z. Gridneva, S.L. Perrella, M.E. Wlodek, C.T. Lai, D.T. Geddes. Human Milk Metabolic Hormones: Analytical Methods and Current Understanding. Int. J. Mol. Sci., 2021. [DOI | PubMed]
- Y.O. Ilcol, Z.B. Hizli, T. Ozkan. Leptin concentration in breast milk and its relationship to duration of lactation and hormonal status. Int. Breastfeed. J., 2006. [DOI | PubMed]
- S. Pundir, Z. Gridneva, A. Pillai, E.B. Thorstensen, C.R. Wall, D.T. Geddes, D. Cameron-Smith. Human Milk Glucocorticoid Levels Are Associated with Infant Adiposity and Head Circumference Over the First Year of Life. Front. Nutr., 2020. [DOI | PubMed]
- P.A. Quitadamo, L. Comegna, P. Cristalli. Anti-Infective, Anti-Inflammatory, and Immunomodulatory Properties of Breast Milk Factors for the Protection of Infants in the Pandemic From COVID-19. Front. Public Health, 2020. [DOI | PubMed]
- C.J. Field. The immunological components of human milk and their effect on immune development in infants. J. Nutr., 2005. [DOI | PubMed]
- M. Toscano, R. De Grandi, E. Grossi, L. Drago. Role of the Human Breast Milk-Associated Microbiota on the Newborns’ Immune System: A Mini Review. Front. Microbiol., 2017. [DOI | PubMed]
- A. Soto, V. Martín, E. Jiménez, I. Mader, J.M. Rodríguez, L. Fernández. Lactobacilli and bifidobacteria in human breast milk: Influence of antibiotherapy and other host and clinical factors. J. Pediatric Gastroenterol. Nutr., 2014. [DOI | PubMed]
- M.R. Simpson, G. Brede, J. Johansen, R. Johnsen, O. Storrø, P. Sætrom, T. Øien. Human Breast Milk miRNA, Maternal Probiotic Supplementation and Atopic Dermatitis in Offspring. PLoS ONE, 2015. [DOI | PubMed]
- M. Alsaweed, C.T. Lai, P.E. Hartmann, D.T. Geddes, F. Kakulas. Human Milk Cells and Lipids Conserve Numerous Known and Novel miRNAs, Some of Which Are Differentially Expressed during Lactation. PLoS ONE, 2016. [DOI | PubMed]
- Y. Xi, X. Jiang, R. Li, M. Chen, W. Song, X. Li. The levels of human milk microRNAs and their association with maternal weight characteristics. Eur. J. Clin. Nutr., 2016. [DOI | PubMed]
- M.C. Carney, A. Tarasiuk, S.L. DiAngelo, P. Silveyra, A. Podany, L.L. Birch, I.M. Paul, S. Kelleher, S.D. Hicks. Metabolism-related microRNAs in maternal breast milk are influenced by premature delivery. Pediatric Res., 2017. [DOI | PubMed]
- Y. Liao, X. Du, J. Li, B. Lönnerdal. Human milk exosomes and their microRNAs survive digestion in vitro and are taken up by human intestinal cells. Mol. Nutr. Food Res., 2017. [DOI | PubMed]
- A. Benmoussa, P. Provost. Milk MicroRNAs in Health and Disease. Compr. Rev. Food Sci. Food Saf., 2019. [DOI | PubMed]
- N. Kosaka, H. Izumi, K. Sekine, T. Ochiya. microRNA as a new immune-regulatory agent in breast milk. Silence, 2010. [DOI | PubMed]
- S.D. Hicks, A. Confair, K. Warren, D. Chandran. Levels of Breast Milk MicroRNAs and Other Non-Coding RNAs Are Impacted by Milk Maturity and Maternal Diet. Front. Immunol., 2021. [DOI | PubMed]
- B.J. Stephen, N. Pareek, M. Saeed, M.A. Kausar, S. Rahman, M. Datta. Xeno-miRNA in Maternal-Infant Immune Crosstalk: An Aid to Disease Alleviation. Front. Immunol., 2020. [DOI | PubMed]
- E. Carrillo-Lozano, F. Sebastián-Valles, C. Knott-Torcal. Circulating microRNAs in Breast Milk and Their Potential Impact on the Infant. Nutrients, 2020. [DOI]
- E.K. Ng, W.P. Tsang, S.S. Ng, H.C. Jin, J. Yu, J.J. Li, C. Röcken, M.P. Ebert, T.T. Kwok, J.J. Sung. MicroRNA-143 targets DNA methyltransferases 3A in colorectal cancer. Br. J. Cancer, 2009. [DOI | PubMed]
- K. Huumonen, M. Korkalainen, M. Viluksela, T. Lahtinen, J. Naarala, J. Juutilainen. Role of microRNAs and DNA Methyltransferases in Transmitting Induced Genomic Instability between Cell Generations. Front. Public Health, 2014. [DOI | PubMed]
- L.-A. Macfarlane, P.R. Murphy. MicroRNA: Biogenesis, Function and Role in Cancer. Curr. Genom., 2010. [DOI]
- J. O’Brien, H. Hayder, Y. Zayed, C. Peng. Overview of MicroRNA Biogenesis, Mechanisms of Actions, and Circulation. Front. Endocrinol., 2018. [DOI]
- E. Navarro Quiroz, R. Navarro Quiroz, L. Pacheco Lugo, G. Aroca Martínez, L. Gómez Escorcia, H. Gonzalez Torres, A. Cadena Bonfanti, M.D.C. Marmolejo, E. Sanchez, J.L. Villarreal Camacho. Integrated analysis of microRNA regulation and its interaction with mechanisms of epigenetic regulation in the etiology of systemic lupus erythematosus. PLoS ONE, 2019. [DOI | PubMed]
- 58.Infant and Young Child Feeding: Model Chapter for Textbooks for Medical Students and Allied Health ProfessionalsWorld Health OrganizationGeneva, Switzerland2009
- G. Zucca-Matthes, C. Urban, A. Vallejo. Anatomy of the nipple and breast ducts. Gland Surg., 2016. [DOI | PubMed]
- K. Uvnas Moberg, A. Ekstrom-Bergstrom, S. Buckley, C. Massarotti, Z. Pajalic, K. Luegmair, A. Kotlowska, L. Lengler, I. Olza, S. Grylka-Baeschlin. Maternal plasma levels of oxytocin during breastfeeding-A systematic review. PLoS ONE, 2020. [DOI | PubMed]
- H. Macias, L. Hinck. Mammary gland development. Wiley Interdiscip. Rev. Dev. Biol., 2012. [DOI | PubMed]
- G. Colleluori, J. Perugini, G. Barbatelli, S. Cinti. Mammary gland adipocytes in lactation cycle, obesity and breast cancer. Rev. Endocr. Metab. Disord., 2021. [DOI | PubMed]
- R.A. Lawrence, R.M. Lawrence. Breastfeeding: A Guide for the Medical Profession, 2015
- J.L. Voogt. Control of hormone release during lactation. Clin. Obstet. Gynaecol., 1978. [DOI | PubMed]
- P.E. Hartmann, R.A. Owens, D.B. Cox, J.C. Kent. Breast Development and Control of Milk Synthesis. Food Nutr. Bull., 1996. [DOI]
- K.M. Ostrom. A review of the hormone prolactin during lactation. Prog. Food Nutr. Sci., 1990. [PubMed]
- M. Saleem, H. Martin, P. Coates. Prolactin Biology and Laboratory Measurement: An Update on Physiology and Current Analytical Issues. Clin. Biochem. Rev., 2018. [PubMed]
- J.C. Kent, D.K. Prime, C.P. Garbin. Principles for maintaining or increasing breast milk production. J. Obstet. Gynecol. Neonatal Nurs., 2012. [DOI]
- Y. Okatani, Y. Sagara. Role of melatonin in nocturnal prolactin secretion in women with normoprolactinemia and mild hyperprolactinemia. Am. J. Obstet. Gynecol., 1993. [DOI | PubMed]
- D.T. Ramsay, J.C. Kent, R.A. Owens, P.E. Hartmann. Ultrasound imaging of milk ejection in the breast of lactating women. Pediatrics, 2004. [DOI | PubMed]
- K.U. Moberg, D.K. Prime. Oxytocin effects in mothers and infants during breastfeeding. Infant, 2013
- E.N. Erickson, C.L. Emeis. Breastfeeding Outcomes After Oxytocin Use During Childbirth: An Integrative Review. J. Midwifery Women’s Health, 2017. [DOI | PubMed]
- A.M. Stuebe, K. Grewen, S. Meltzer-Brody. Association between maternal mood and oxytocin response to breastfeeding. J. Women’s Health, 2013. [DOI]
- S. Oguchi, K. Shinohara, Y. Yamashiro, W.A. Walker, I.R. Sanderson. Growth factors in breast milk and their effect on gastrointestinal development. Zhonghua Minguo Xiao Er Ke Yi Xue Hui Za Zhi, 1997. [PubMed]
- S. Moossavi, K. Miliku, S. Sepehri, E. Khafipour, M.B. Azad. The Prebiotic and Probiotic Properties of Human Milk: Implications for Infant Immune Development and Pediatric Asthma. Front. Pediatr., 2018. [DOI | PubMed]
- J. Burch, W. Karmaus, V. Gangur, N. Soto-Ramírez, M. Yousefi, L.M. Goetzl. Pre- and perinatal characteristics and breast milk immune markers. Pediatric Res., 2013. [DOI]
- S. Trend, T. Strunk, J. Hibbert, C.H. Kok, G. Zhang, D.A. Doherty, P. Richmond, D. Burgner, K. Simmer, D.J. Davidson. Antimicrobial protein and Peptide concentrations and activity in human breast milk consumed by preterm infants at risk of late-onset neonatal sepsis. PLoS ONE, 2015. [DOI | PubMed]
- A.M. Xavier, K. Rai, A.M. Hegde. Total antioxidant concentrations of breastmilk–an eye-opener to the negligent. J. Health Popul. Nutr., 2011. [DOI | PubMed]
- J. Schrezenmeir, H. Korhonen, C. Williams, H.S. Gill, N. Shah. Foreword. Br. J. Nutr., 2007. [DOI]
- R. Garofalo. Cytokines in human milk. J. Pediatr., 2010. [DOI | PubMed]
- M. Cavaletto, M.G. Giuffrida, A. Conti. The proteomic approach to analysis of human milk fat globule membrane. Clin. Chim. Acta, 2004. [DOI | PubMed]
- R. Jenness. The composition of human milk. Semin. Perinatol., 1979. [PubMed]
- C.R. Martin, P.R. Ling, G.L. Blackburn. Review of Infant Feeding: Key Features of Breast Milk and Infant Formula. Nutrients, 2016. [DOI]
- G. Vizzari, D. Morniroli, F. Ceroni, E. Verduci, A. Consales, L. Colombo, J. Cerasani, F. Mosca, M.L. Gianni. Human Milk, More Than Simple Nourishment. Children, 2021. [DOI | PubMed]
- Y. Golan, Y.G. Assaraf. Genetic and Physiological Factors Affecting Human Milk Production and Composition. Nutrients, 2020. [DOI | PubMed]
- K.E. Gregory, W.A. Walker. Immunologic Factors in Human Milk and Disease Prevention in the Preterm Infant. Curr. Pediatr. Rep., 2013. [DOI | PubMed]
- N.J. Andreas, B. Kampmann, K. Mehring Le-Doare. Human breast milk: A review on its composition and bioactivity. Early Hum. Dev., 2015. [DOI | PubMed]
- D.A. Gidrewicz, T.R. Fenton. A systematic review and meta-analysis of the nutrient content of preterm and term breast milk. BMC Pediatrics, 2014. [DOI | PubMed]
- M.C. Steinhoff, S.B. Omer. A review of fetal and infant protection associated with antenatal influenza immunization. Am. J. Obstet. Gynecol., 2012. [DOI | PubMed]
- S. Chheda, S.E. Keeney, A.S. Goldman. Immunology of Human Milk and Host Immunity. Fetal Neonatal Physiol., 2004. [DOI]
- L. M’Rabet, A.P. Vos, G.N. Boehm, J. Garssen. Breast-Feeding and Its Role in Early Development of the Immune System in Infants: Consequences for Health Later in Life. J. Nutr., 2008. [DOI | PubMed]
- B.M. Jakaitis, P.W. Denning. Human breast milk and the gastrointestinal innate immune system. Clin. Perinatol., 2014. [DOI | PubMed]
- B.M. Henrick, X.D. Yao, L. Nasser, A. Roozrogousheh, K.L. Rosenthal. Breastfeeding Behaviors and the Innate Immune System of Human Milk: Working Together to Protect Infants against Inflammation, HIV-1, and Other Infections. Front. Immunol., 2017. [DOI | PubMed]
- Y. Yao, X. Cai, Y. Ye, F. Wang, F. Chen, C. Zheng. The Role of Microbiota in Infant Health: From Early Life to Adulthood. Front. Immunol., 2021. [DOI | PubMed]
- L. Zhuang, H. Chen, S. Zhang, J. Zhuang, Q. Li, Z. Feng. Intestinal Microbiota in Early Life and Its Implications on Childhood Health. Genom. Proteom. Bioinform., 2019. [DOI]
- R.E. Black, L.H. Allen, Z.A. Bhutta, L.E. Caulfield, M. de Onis, M. Ezzati, C. Mathers, J. Rivera. Maternal and child undernutrition: Global and regional exposures and health consequences. Lancet, 2008. [DOI | PubMed]
- C.M. Dieterich, J.P. Felice, E. O’Sullivan, K.M. Rasmussen. Breastfeeding and health outcomes for the mother-infant dyad. Pediatr. Clin. N. Am., 2013. [DOI]
- D.S. Newburg. Innate immunity and human milk. J. Nutr., 2005. [DOI | PubMed]
- R. Bobinski, J. Bobinska. Fatty acids of human milk—A review. Int. J. Vitam. Nutr. Res., 2020. [DOI]
- J.V. Visentainer, O.O. Santos, L. Maldaner, C. Zappielo, V. Neia, L. Visentainer, L. Pelissari, J. Pizzo, A. Rydlewski, R. Silveira. Lipids and Fatty Acids in Human Milk: Benefits and Analysis. Biochemistry and Health Benefits of Fatty Acids, 2018
- F. Dilika, P.D. Bremner, J.J. Meyer. Antibacterial activity of linoleic and oleic acids isolated from Helichrysum pedunculatum: A plant used during circumcision rites. Fitoterapia, 2000. [DOI | PubMed]
- Y.Y. He, N.T. Lawlor, D.S. Newburg. Human Milk Components Modulate Toll-Like Receptor-Mediated Inflammation. Adv. Nutr. Int. Rev. J., 2016. [DOI]
- E. Kurakevich, T. Hennet, M. Hausmann, G. Rogler, L. Borsig. Milk oligosaccharide sialyl (α2,3) lactose activates intestinal CD11c+ cells through TLR4. Proc. Natl. Acad. Sci. USA, 2013. [DOI | PubMed]
- J. Plaza-Diaz, L. Fontana, A. Gil. Human Milk Oligosaccharides and Immune System Development. Nutrients, 2018. [DOI]
- D.R. Laucirica, V. Triantis, R. Schoemaker, M.K. Estes, S. Ramani. Milk Oligosaccharides Inhibit Human Rotavirus Infectivity in MA104 Cells. J. Nutr., 2017. [DOI | PubMed]
- L. Qian, Y. Zhou, Z. Teng, C.L. Du, C. Tian. Preparation and antibacterial activity of oligosaccharides derived from dandelion. Int. J. Biol. Macromol., 2014. [DOI | PubMed]
- J. Lis-Kuberka, M. Orczyk-Pawilowicz. Sialylated Oligosaccharides and Glycoconjugates of Human Milk. The Impact on Infant and Newborn Protection, Development and Well-Being. Nutrients, 2019. [DOI | PubMed]
- L. Bode, C. Kunz, M. Muhly-Reinholz, K. Mayer, W. Seeger, S. Rudloff. Inhibition of monocyte, lymphocyte, and neutrophil adhesion to endothelial cells by human milk oligosaccharides. Thromb. Haemost., 2004. [DOI | PubMed]
- T. Suligoj, L.K. Vigsnaes, P.V.D. Abbeele, A. Apostolou, K. Karalis, G.M. Savva, B. McConnell, N. Juge. Effects of Human Milk Oligosaccharides on the Adult Gut Microbiota and Barrier Function. Nutrients, 2020. [DOI | PubMed]
- D. Newburg, A. Walker. Protection of the neonate by the innate immune system of developing gut and of human milk. Pediatric Res., 2007. [DOI | PubMed]
- A. Mykoniatis, P.M. Anton, M. Wlk, C.C. Wang, L. Ungsunan, S. Blher, M. Venihaki, S. Simeonidis, J. Zacks, D. Zhao. Leptin mediates Clostridium difficile toxin A-induced enteritis in mice. Gastroenterology, 2003. [DOI | PubMed]
- A.T. Soliman, M. Yasin, A. Kassem. Leptin in pediatrics: A hormone from adipocyte that wheels several functions in children. Indian J. Endocrinol. Metab., 2012. [DOI | PubMed]
- T.J. Smith. Insulin-like growth factor-I regulation of immune function: A potential therapeutic target in autoimmune diseases?. Pharmacol. Rev., 2010. [DOI | PubMed]
- N.T. Cacho, R.M. Lawrence. Innate Immunity and Breast Milk. Front. Immunol., 2017. [DOI | PubMed]
- M. Witkowska-Zimny, E. Kaminska-El-Hassan. Cells of human breast milk. Cell. Mol. Biol. Lett., 2017. [DOI | PubMed]
- A.S. Goldman, C. Garza, B.L. Nichols, R.M. Goldblum. Immunologic factors in human milk during the first year of lactation. J. Pediatrics, 1982. [DOI]
- H.L. Yilmaz, E.S. Saygili-Yilmaz, R. Gunesacar. Interleukin-10 and-12 in human milk at 3 stages of lactation: A longitudinal study. Adv. Ther., 2007. [DOI | PubMed]
- M. Guo. Human Milk and Infant Formula. Functional Foods, 2009
- L.E. Carr, M.D. Virmani, F. Rosa, D. Munblit, K.S. Matazel, A.A. Elolimy, L. Yeruva. Role of Human Milk Bioactives on Infants’ Gut and Immune Health. Front. Immunol., 2021. [DOI | PubMed]
- E.M. Kosciuczuk, P. Lisowski, J. Jarczak, N. Strzalkowska, A. Jozwik, J. Horbanczuk, J. Krzyzewski, L. Zwierzchowski, E. Bagnicka. Cathelicidins: Family of antimicrobial peptides. A review. Mol. Biol. Rep., 2012. [DOI | PubMed]
- J.R. Zucali, H.E. Broxmeyer, D. Levy, C. Morse. Lactoferrin decreases monocyte-induced fibroblast production of myeloid colony-stimulating activity by suppressing monocyte release of interleukin-1. Blood, 1989. [DOI | PubMed]
- E.S. Buescher. Anti-inflammatory characteristics of human milk. Bioactive Components of Human Milk, 2001
- J. Brock. Lactoferrin in human milk: Its role in iron absorption and protection against enteric infection in the newborn infant. Arch. Dis. Child., 1980. [DOI | PubMed]
- B. Yang, J. Wang, B. Tang, Y. Liu, C. Guo, P. Yang, T. Yu, R. Li, J. Zhao, L. Zhang. Characterization of bioactive recombinant human lysozyme expressed in milk of cloned transgenic cattle. PLoS ONE, 2011. [DOI | PubMed]
- J. Malaczewska, E. Kaczorek-Lukowska, R. Wojcik, A.K. Siwicki. Antiviral effects of nisin, lysozyme, lactoferrin and their mixtures against bovine viral diarrhoea virus. BMC Vet. Res., 2019. [DOI | PubMed]
- S.A. Ragland, A.K. Criss. From bacterial killing to immune modulation: Recent insights into the functions of lysozyme. PLoS Pathog., 2017. [DOI | PubMed]
- L. Yang, Z. Guo, M. Yu, X. Cai, Y. Mao, F. Tian, W. Xu, G. Liu, X. Li, Y. Zhao. Profile of Nucleotides in Chinese Mature Breast Milk from Six Regions. Nutrients, 2022. [DOI | PubMed]
- R. Chaudhari, M. Fanion. Technical aspects of micronutrient addition to foods. Food Fortification and Supplementation, 2008
- L. Tingo, E. Ahlberg, L. Johansson, S.A. Pedersen, K. Chawla, P. Saetrom, E. Cione, M.R. Simpson. Non-Coding RNAs in Human Breast Milk: A Systematic Review. Front. Immunol., 2021. [DOI | PubMed]
- D.M. Haas, M. Daum, T. Skaar, S. Philips, D. Miracle, J.L. Renbarger. Human breast milk as a source of DNA for amplification. J. Clin. Pharmacol., 2011. [DOI | PubMed]
- M. Guo. Human Milk Biochemistry and Infant Formula Manufacturing Technology, 2014
- B. Lonnerdal. Nutritional and physiologic significance of human milk proteins. Am. J. Clin. Nutr., 2003. [DOI | PubMed]
- E. Verduci, G. Banderali, S. Barberi, G. Radaelli, A. Lops, F. Betti, E. Riva, M. Giovannini. Epigenetic effects of human breast milk. Nutrients, 2014. [DOI | PubMed]
- C.L. Wagner, S.N. Taylor, D. Johnson. Host factors in amniotic fluid and breast milk that contribute to gut maturation. Clin. Rev. Allergy Immunol., 2008. [DOI | PubMed]
- C.J. Chang, J.C. Chao. Effect of human milk and epidermal growth factor on growth of human intestinal Caco-2 cells. J. Pediatr. Gastroenterol. Nutr., 2002. [DOI | PubMed]
- B. Dvorak, C.C. Fituch, C.S. Williams, N.M. Hurst, R.J. Schanler. Increased epidermal growth factor levels in human milk of mothers with extremely premature infants. Pediatr. Res., 2003. [DOI | PubMed]
- D. Munblit, S. Sheth, P. Abrol, M. Treneva, D.G. Peroni, L.Y. Chow, A.L. Boner, A. Pampura, J.O. Warner, R.J. Boyle. Exposures influencing total IgA level in colostrum. J. Dev. Orig. Health Dis., 2016. [DOI | PubMed]
- A.J. Twigger, A.R. Hepworth, C.T. Lai, E. Chetwynd, A.M. Stuebe, P. Blancafort, P.E. Hartmann, D.T. Geddes, F. Kakulas. Gene expression in breastmilk cells is associated with maternal and infant characteristics. Sci. Rep., 2015. [DOI | PubMed]
- S. Agarwal, W. Karmaus, S. Davis, V. Gangur. Immune markers in breast milk and fetal and maternal body fluids: A systematic review of perinatal concentrations. J. Hum. Lact., 2011. [DOI | PubMed]
- I.A. Penttila. Milk-derived transforming growth factor-beta and the infant immune response. J. Pediatr., 2010. [DOI | PubMed]
- Y. Nakamura, M. Miyata, T. Ando, N. Shimokawa, Y. Ohnuma, R. Katoh, H. Ogawa, K. Okumura, A. Nakao. The latent form of transforming growth factor-beta administered orally is activated by gastric acid in mice. J. Nutr., 2009. [DOI | PubMed]
- S. Musilova, V. Rada, E. Vlkova, V. Bunesova. Beneficial effects of human milk oligosaccharides on gut microbiota. Benef. Microbes, 2014. [DOI | PubMed]
- E. Bezirtzoglou, A. Tsiotsias, G.W. Welling. Microbiota profile in feces of breast- and formula-fed newborns by using fluorescence in situ hybridization (FISH). Anaerobe, 2011. [DOI | PubMed]
- K. Lubiech, M. Twaruzek. Lactobacillus Bacteria in Breast Milk. Nutrients, 2020. [DOI]
- X. Zhang, S. Mushajiang, B. Luo, F. Tian, Y. Ni, W. Yan. The Composition and Concordance of Lactobacillus Populations of Infant Gut and the Corresponding Breast-Milk and Maternal Gut. Front. Microbiol., 2020. [DOI | PubMed]
- R. Oozeer, K. van Limpt, T. Ludwig, K. Ben Amor, R. Martin, R.D. Wind, G. Boehm, J. Knol. Intestinal microbiology in early life: Specific prebiotics can have similar functionalities as human-milk oligosaccharides. Am. J. Clin. Nutr., 2013. [DOI | PubMed]
- C. Chassard, T. de Wouters, C. Lacroix. Probiotics tailored to the infant: A window of opportunity. Curr. Opin. Biotechnol., 2014. [DOI | PubMed]
- X. Zou, J. Huang, Q. Jin, Z. Guo, Y. Liu, L. Cheong, X. Xu, X. Wang. Lipid composition analysis of milk fats from different mammalian species: Potential for use as human milk fat substitutes. J. Agric. Food Chem., 2013. [DOI | PubMed]
- R. Luoto, O. Ruuskanen, M. Waris, M. Kalliomaki, S. Salminen, E. Isolauri. Prebiotic and probiotic supplementation prevents rhinovirus infections in preterm infants: A randomized, placebo-controlled trial. J. Allergy Clin. Immunol., 2014. [DOI | PubMed]
- H. Ahanchian, S.A. Jafari. Probiotics and Prebiotics for Prevention of Viral Respiratory Tract Infections. Probiotics Prebiotics Synbiotics, 2016. [DOI]
- A. Koromyslova, S. Tripathi, V. Morozov, H. Schroten, G.S. Hansman. Human norovirus inhibition by a human milk oligosaccharide. Virology, 2017. [DOI | PubMed]
- S. Weichert, A. Koromyslova, B.K. Singh, S. Hansman, S. Jennewein, H. Schroten, G.S. Hansman. Structural Basis for Norovirus Inhibition by Human Milk Oligosaccharides. J. Virol., 2016. [DOI | PubMed]
- M.J. Pichler, C. Yamada, B. Shuoker, C. Alvarez-Silva, A. Gotoh, M.L. Leth, E. Schoof, T. Katoh, M. Sakanaka, T. Katayama. Butyrate producing colonic Clostridiales metabolise human milk oligosaccharides and cross feed on mucin via conserved pathways. Nat. Commun., 2020. [DOI | PubMed]
- D.A. Sela, D. Garrido, L. Lerno, S. Wu, K. Tan, H.J. Eom, A. Joachimiak, C.B. Lebrilla, D.A. Mills. Bifidobacterium longum subsp. infantis ATCC 15697 α-fucosidases are active on fucosylated human milk oligosaccharides. Appl. Environ. Microbiol., 2012. [DOI | PubMed]
- H. Sakurama, M. Kiyohara, J. Wada, Y. Honda, M. Yamaguchi, S. Fukiya, A. Yokota, H. Ashida, H. Kumagai, M. Kitaoka. Lacto-N-biosidase encoded by a novel gene of Bifidobacterium longum subspecies longum shows unique substrate specificity and requires a designated chaperone for its active expression. J. Biol. Chem., 2013. [DOI | PubMed]
- M.A.I. Al-Hatamleh, I. Ismail, O.M. Al-Shajrawi, T.M. Ariff. Effect of stress on alteration of haematological parameters: A preliminary study on preclinical medical students in Malaysia. J. Cell. Neurosci. Oxidative Stress, 2020. [DOI]
- M.A.I. Al-Hatamleh, M.M. Hatmal, K. Sattar, S. Ahmad, M.Z. Mustafa, M.D.C. Bittencourt, R. Mohamud. Antiviral and Immunomodulatory Effects of Phytochemicals from Honey against COVID-19: Potential Mechanisms of Action and Future Directions. Molecules, 2020. [DOI]
- A. Gila-Diaz, S.M. Arribas, A. Algara, M.A. Martin-Cabrejas, A.L. Lopez de Pablo, M. Saenz de Pipaon, D. Ramiro-Cortijo. A Review of Bioactive Factors in Human Breastmilk: A Focus on Prematurity. Nutrients, 2019. [DOI]
- P.C. Castillo-Castaneda, A. Garcia-Gonzalez, A.E. Bencomo-Alvarez, P. Barros-Nunez, R. Gaxiola-Robles, L.C. Mendez-Rodriguez, T. Zenteno-Savin. Micronutrient content and antioxidant enzyme activities in human breast milk. J. Trace Elem. Med. Biol., 2019. [DOI | PubMed]
- N.A. Ankrah, R. Appiah-Opong, C. Dzokoto. Human breastmilk storage and the glutathione content. J. Trop. Pediatr., 2000. [DOI | PubMed]
- M.M. Goyal, A. Basak. Human catalase: Looking for complete identity. Protein Cell, 2010. [DOI | PubMed]
- R. Brigelius-Flohe. Tissue-specific functions of individual glutathione peroxidases. Free Radic. Biol. Med., 1999. [DOI | PubMed]
- N. Ballatori, S.M. Krance, S. Notenboom, S. Shi, K. Tieu, C.L. Hammond. Glutathione dysregulation and the etiology and progression of human diseases. Biol. Chem., 2009. [DOI | PubMed]
- R.J. Reiter. Antioxidant actions of melatonin. Adv. Pharmacol., 1997. [DOI | PubMed]
- A.I. Eidelman, R.J. Schanler, M. Johnston, S. Landers, L. Noble, K. Szucs, L. Viehmann. Breastfeeding and the use of human milk. Pediatrics, 2012. [PubMed]
- S. Ip, M. Chung, G. Raman. Tufts-New England Medical Center Evidence-based Practice Center. Breastfeeding and maternal and infant health outcomes in developed countries. Evid. Rep. Technol. Assess., 2007
- C.G. Grulee, H.N. Sanford, P.H. Herron. Breast and artificial feeding: Influence on morbidity and mortality of twenty thousand infants. J. Am. Med. Assoc., 1934. [DOI]
- Z.Y. Kho, S.K. Lal. The Human Gut Microbiome-A Potential Controller of Wellness and Disease. Front. Microbiol., 2018. [DOI | PubMed]
- C.G. Turin, T.J. Ochoa. The Role of Maternal Breast Milk in Preventing Infantile Diarrhea in the Developing World. Curr. Trop. Med. Rep., 2014. [DOI | PubMed]
- T. Yahaya, U. Shemishere. Association between Bioactive Molecules in Breast Milk and Type 1 Diabetes Mellitus. Sultan Qaboos Univ. Med. J., 2020. [DOI | PubMed]
- S.-Y. Ying, D.C. Chang, S.-L. Lin. The microRNA (miRNA): Overview of the RNA genes that modulate gene function. Mol. Biotechnol., 2008. [DOI | PubMed]
- Z. Fang, R. Du, A. Edwards, E.K. Flemington, K. Zhang. The sequence structures of human microRNA molecules and their implications. PLoS ONE, 2013. [DOI | PubMed]
- C. Xiao, K. Rajewsky. MicroRNA control in the immune system: Basic principles. Cell, 2009. [DOI | PubMed]
- M.R. Fabian, N. Sonenberg, W. Filipowicz. Regulation of mRNA translation and stability by microRNAs. Annu. Rev. Biochem., 2010. [DOI | PubMed]
- W. Filipowicz, S.N. Bhattacharyya, N. Sonenberg. Mechanisms of post-transcriptional regulation by microRNAs: Are the answers in sight?. Nat. Rev. Genet., 2008. [DOI | PubMed]
- S. Vasudevan, Y. Tong, J.A. Steitz. Switching from repression to activation: MicroRNAs can up-regulate translation. Science, 2007. [DOI | PubMed]
- L. He, G.J. Hannon. MicroRNAs: Small RNAs with a big role in gene regulation. Nat. Rev. Genet., 2004. [DOI | PubMed]
- C.E. Condrat, D.C. Thompson, M.G. Barbu, O.L. Bugnar, A. Boboc, D. Cretoiu, N. Suciu, S.M. Cretoiu, S.C. Voinea. miRNAs as Biomarkers in Disease: Latest Findings Regarding Their Role in Diagnosis and Prognosis. Cells, 2020. [DOI]
- M. Lagos-Quintana, R. Rauhut, A. Yalcin, J. Meyer, W. Lendeckel, T. Tuschl. Identification of tissue-specific microRNAs from mouse. Curr. Biol., 2002. [DOI | PubMed]
- S. Kumar, P.H. Reddy. Are circulating microRNAs peripheral biomarkers for Alzheimer’s disease?. Biochim. Biophys. Acta, 2016. [DOI | PubMed]
- Y. Peng, C.M. Croce. The role of MicroRNAs in human cancer. Signal Transduct. Target. Ther., 2016. [DOI | PubMed]
- A. Valinezhad Orang, R. Safaralizadeh, M. Kazemzadeh-Bavili. Mechanisms of miRNA-Mediated Gene Regulation from Common Downregulation to mRNA-Specific Upregulation. Int. J. Genom., 2014. [DOI]
- M.W. Webster, Y.-H. Chen, J.A.W. Stowell, N. Alhusaini, T. Sweet, B.R. Graveley, J. Coller, L.A. Passmore. mRNA Deadenylation Is Coupled to Translation Rates by the Differential Activities of Ccr4-Not Nucleases. Mol. Cell, 2018. [DOI | PubMed]
- D. O’Carroll, A. Schaefer. General Principals of miRNA Biogenesis and Regulation in the Brain. Neuropsychopharmacology, 2013. [DOI | PubMed]
- M. Alsaweed, C.T. Lai, P.E. Hartmann, D.T. Geddes, F. Kakulas. Human milk miRNAs primarily originate from the mammary gland resulting in unique miRNA profiles of fractionated milk. Sci. Rep., 2016. [DOI | PubMed]
- M. Alsaweed, C.T. Lai, P.E. Hartmann, D.T. Geddes, F. Kakulas. Human milk cells contain numerous miRNAs that may change with milk removal and regulate multiple physiological processes. Int. J. Mol. Sci., 2016. [DOI]
- M. Alsaweed, A.R. Hepworth, C. Lefevre, P.E. Hartmann, D.T. Geddes, F. Hassiotou. Human Milk MicroRNA and Total RNA Differ Depending on Milk Fractionation. J. Cell. Biochem., 2015. [DOI | PubMed]
- Q. Zhou, M. Li, X. Wang, Q. Li, T. Wang, Q. Zhu, X. Zhou, X. Wang, X. Gao, X. Li. Immune-related microRNAs are abundant in breast milk exosomes. Int. J. Biol. Sci., 2012. [DOI | PubMed]
- E.M. Munch, R.A. Harris, M. Mohammad, A.L. Benham, S.M. Pejerrey, L. Showalter, M. Hu, C.D. Shope, P.D. Maningat, P.H. Gunaratne. Transcriptome profiling of microRNA by Next-Gen deep sequencing reveals known and novel miRNA species in the lipid fraction of human breast milk. PLoS ONE, 2013. [DOI | PubMed]
- J.A. Weber, D.H. Baxter, S. Zhang, D.Y. Huang, K.H. Huang, M.J. Lee, D.J. Galas, K. Wang. The microRNA spectrum in 12 body fluids. Clin. Chem., 2010. [DOI | PubMed]
- S. Mathivanan, H. Ji, R.J. Simpson. Exosomes: Extracellular organelles important in intercellular communication. J. Proteom., 2010. [DOI | PubMed]
- L.C. Cheng, M. Tavazoie, F. Doetsch. Stem cells: From epigenetics to microRNAs. Neuron, 2005. [DOI | PubMed]
- J. Pothof, N.S. Verkaik, W. Van Ijcken, E.A. Wiemer, V.T. Ta, G.T. van der Horst, N.G. Jaspers, D.C. van Gent, J.H. Hoeijmakers, S.P. Persengiev. MicroRNA-mediated gene silencing modulates the UV-induced DNA-damage response. EMBO J., 2009. [DOI | PubMed]
- Y. Gao, J. Schug, L.B. McKenna, J. Le Lay, K.H. Kaestner, L.E. Greenbaum. Tissue-specific regulation of mouse microRNA genes in endoderm-derived tissues. Nucleic Acids Res., 2011. [DOI | PubMed]
- E.S. Buescher, I. Malinowska. Soluble receptors and cytokine antagonists in human milk. Pediatr. Res., 1996. [DOI | PubMed]
- L. Zhang, D. Hou, X. Chen, D. Li, L. Zhu, Y. Zhang, J. Li, Z. Bian, X. Liang, X. Cai. Exogenous plant MIR168a specifically targets mammalian LDLRAP1: Evidence of cross-kingdom regulation by microRNA. Cell Res., 2012. [DOI | PubMed]
- G.A. Calin, C.M. Croce. MicroRNA signatures in human cancers. Nat. Rev. Cancer, 2006. [DOI | PubMed]
- T. Kinouchi, K. Koizumi, T. Kuwata, T. Yajima. Milk-borne insulin with trypsin inhibitor in milk induces pancreatic amylase development at the onset of weaning in rats. J. Pediatric Gastroenterol. Nutr., 2000. [DOI]
- M. Alsaweed, P.E. Hartmann, D.T. Geddes, F. Kakulas. MicroRNAs in Breastmilk and the Lactating Breast: Potential Immunoprotectors and Developmental Regulators for the Infant and the Mother. Int. J. Environ. Res. Public Health, 2015. [DOI | PubMed]
- S. Saurabh, A.S. Vidyarthi, D. Prasad. RNA interference: Concept to reality in crop improvement. Planta, 2014. [DOI | PubMed]
- B.P. Lewis, C.B. Burge, D.P. Bartel. Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell, 2005. [DOI | PubMed]
- R.C. Friedman, K.K. Farh, C.B. Burge, D.P. Bartel. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res., 2009. [DOI | PubMed]
- S. Griffiths-Jones, R.J. Grocock, S. van Dongen, A. Bateman, A.J. Enright. miRBase: MicroRNA sequences, targets and gene nomenclature. Nucleic Acids Res., 2006. [DOI | PubMed]
- I. Bentwich, A. Avniel, Y. Karov, R. Aharonov, S. Gilad, O. Barad, A. Barzilai, P. Einat, U. Einav, E. Meiri. Identification of hundreds of conserved and nonconserved human microRNAs. Nat. Genet., 2005. [DOI | PubMed]
- X. Jiang, L. You, Z. Zhang, X. Cui, H. Zhong, X. Sun, C. Ji, X. Chi. Biological Properties of Milk-Derived Extracellular Vesicles and Their Physiological Functions in Infant. Front. Cell Dev. Biol., 2021. [DOI | PubMed]
- J. Tome-Carneiro, N. Fernandez-Alonso, C. Tomas-Zapico, F. Visioli, E. Iglesias-Gutierrez, A. Davalos. Breast milk microRNAs harsh journey towards potential effects in infant development and maturation. Lipid encapsulation can help. Pharmacol. Res., 2018. [DOI | PubMed]
- C. Leroux, M.L. Chervet, J.B. German. Perspective: Milk microRNAs as Important Players in Infant Physiology and Development. Adv. Nutr., 2021. [DOI | PubMed]
- L.M. Doyle, M.Z. Wang. Overview of Extracellular Vesicles, Their Origin, Composition, Purpose, and Methods for Exosome Isolation and Analysis. Cells, 2019. [DOI]
- K. O’Brien, K. Breyne, S. Ughetto, L.C. Laurent, X.O. Breakefield. RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat. Rev. Mol. Cell Biol., 2020. [DOI | PubMed]
- S. Zhu, M.L. Si, H. Wu, Y.Y. Mo. MicroRNA-21 targets the tumor suppressor gene tropomyosin 1 (TPM1). J. Biol. Chem., 2007. [DOI | PubMed]
- S. Mishra, J.J. Deng, P.S. Gowda, M.K. Rao, C.L. Lin, C.L. Chen, T. Huang, L.Z. Sun. Androgen receptor and microRNA-21 axis downregulates transforming growth factor beta receptor II (TGFBR2) expression in prostate cancer. Oncogene, 2014. [DOI | PubMed]
- R. Bhome, F. Del Vecchio, G.H. Lee, M.D. Bullock, J.N. Primrose, A.E. Sayan, A.H. Mirnezami. Exosomal microRNAs (exomiRs): Small molecules with a big role in cancer. Cancer Lett., 2018. [DOI | PubMed]
- N. Kosaka, H. Iguchi, T. Ochiya. Circulating microRNA in body fluid: A new potential biomarker for cancer diagnosis and prognosis. Cancer Sci., 2010. [DOI | PubMed]
- L. Ji, X. Chen. Regulation of small RNA stability: Methylation and beyond. Cell Res., 2012. [DOI | PubMed]
- T. Wolf, S.R. Baier, J. Zempleni. The Intestinal Transport of Bovine Milk Exosomes Is Mediated by Endocytosis in Human Colon Carcinoma Caco-2 Cells and Rat Small Intestinal IEC-6 Cells. J. Nutr., 2015. [DOI | PubMed]
- M. Nazari-Jahantigh, V. Egea, A. Schober, C. Weber. MicroRNA-specific regulatory mechanisms in atherosclerosis. J. Mol. Cell. Cardiol., 2015. [DOI | PubMed]
- Y. Ito, K. Taniguchi, Y. Kuranaga, N. Eid, Y. Inomata, S.W. Lee, K. Uchiyama. Uptake of MicroRNAs from Exosome-Like Nanovesicles of Edible Plant Juice by Rat Enterocytes. Int. J. Mol. Sci., 2021. [DOI | PubMed]
- A. Blank, C. Dekker, G. Schieven, R. Sugiyama, M. Thelen. Human body fluid ribonucleases: Detection, interrelationships and significance. Nucleic Acids Symp. Ser., 1981. [PubMed]
- R.G. Matos, A. Barbas, C.M. Arraiano. RNase R mutants elucidate the catalysis of structured RNA: RNA-binding domains select the RNAs targeted for degradation. Biochem. J., 2009. [DOI | PubMed]
- H.S. Bernhardt, W.P. Tate. Primordial soup or vinaigrette: Did the RNA world evolve at acidic pH?. Biol. Direct, 2012. [DOI | PubMed]
- M. Levy, S.L. Miller. The stability of the RNA bases: Implications for the origin of life. Proc. Natl. Acad. Sci. USA, 1998. [DOI | PubMed]
- T. Chiba, A. Kooka, K. Kowatari, M. Yoshizawa, N. Chiba, A. Takaguri, Y. Fukushi, F. Hongo, H. Sato, S. Wada. Expression profiles of hsa-miR-148a-3p and hsa-miR-125b-5p in human breast milk and infant formulae. Int. Breastfeed. J., 2022. [DOI | PubMed]
- A. Lukasik, I. Brzozowska, U. Zielenkiewicz, P. Zielenkiewicz. Detection of Plant miRNAs Abundance in Human Breast Milk. Int. J. Mol. Sci., 2017. [DOI]
- F. Mar-Aguilar, A. Arreola-Triana, D. Mata-Cardona, V. Gonzalez-Villasana, C. Rodriguez-Padilla, D. Resendez-Perez. Evidence of transfer of miRNAs from the diet to the blood still inconclusive. PeerJ, 2020. [DOI | PubMed]
- Z. Li, R. Xu, N. Li. MicroRNAs from plants to animals, do they define a new messenger for communication?. Nutr. Metab., 2018. [DOI]
- H. Hayek, B. Kosmider, K. Bahmed. The role of miRNAs in alveolar epithelial cells in emphysema. Biomed. Pharmacol., 2021. [DOI]
- E. Diez-Sainz, S. Lorente-Cebrian, P. Aranaz, J.I. Riezu-Boj, J.A. Martinez, F.I. Milagro. Potential Mechanisms Linking Food-Derived MicroRNAs, Gut Microbiota and Intestinal Barrier Functions in the Context of Nutrition and Human Health. Front. Nutr., 2021. [DOI | PubMed]
- Q. Zhao, Y. Liu, N. Zhang, M. Hu, H. Zhang, T. Joshi, D. Xu. Evidence for plant-derived xenomiRs based on a large-scale analysis of public small RNA sequencing data from human samples. PLoS ONE, 2018. [DOI | PubMed]
- X. Chen, L. Liu, Q. Chu, S. Sun, Y. Wu, Z. Tong, W. Fang, M.P. Timko, L. Fan. Large-scale identification of extracellular plant miRNAs in mammals implicates their dietary intake. PLoS ONE, 2021. [DOI | PubMed]
- G.M. Sundaram. Dietary non-coding RNAs from plants: Fairy tale or treasure?. Noncoding RNA Res., 2019. [DOI | PubMed]
- C. Peila, S. Sottemano, F. Cesare Marincola, M. Stocchero, N.G. Pusceddu, A. Dessi, E. Baraldi, V. Fanos, E. Bertino. NMR Metabonomic Profile of Preterm Human Milk in the First Month of Lactation: From Extreme to Moderate Prematurity. Foods, 2022. [DOI | PubMed]
- R. Zamanillo, J. Sanchez, F. Serra, A. Palou. Breast Milk Supply of MicroRNA Associated with Leptin and Adiponectin Is Affected by Maternal Overweight/Obesity and Influences Infancy BMI. Nutrients, 2019. [DOI | PubMed]
- B. Yun, Y. Kim, D.J. Park, S. Oh. Comparative analysis of dietary exosome-derived microRNAs from human, bovine and caprine colostrum and mature milk. J. Anim. Sci. Technol., 2021. [DOI | PubMed]
- J. Xu, C.X. Li, Y.S. Li, J.Y. Lv, Y. Ma, T.T. Shao, L.D. Xu, Y.Y. Wang, L. Du, Y.P. Zhang. MiRNA-miRNA synergistic network: Construction via co-regulating functional modules and disease miRNA topological features. Nucleic Acids Res., 2011. [DOI | PubMed]
- X. Chen, W. Zhao, Y. Yuan, Y. Bai, Y. Sun, W. Zhu, Z. Du. MicroRNAs tend to synergistically control expression of genes encoding extensively-expressed proteins in humans. PeerJ, 2017. [DOI | PubMed]
- J. Zhang, V.V.H. Pham, L. Liu, T. Xu, B. Truong, J. Li, N. Rao, T.D. Le. Identifying miRNA synergism using multiple-intervention causal inference. BMC Bioinform., 2019. [DOI]
- K. Zhang, R. Yang, J. Chen, E. Qi, S. Zhou, Y. Wang, Q. Fu, R. Chen, X. Fang. Let-7i-5p Regulation of Cell Morphology and Migration Through Distinct Signaling Pathways in Normal and Pathogenic Urethral Fibroblasts. Front. Bioeng. Biotechnol., 2020. [DOI | PubMed]
- W. Xiang, C. Tian, J. Lin, X. Wu, G. Pang, L. Zhou, S. Pan, Z. Deng. Plasma let-7i and miR-15a expression are associated with the effect of recombinant tissue plasminogen activator treatment in acute ischemic stroke patients. Thromb. Res., 2017. [DOI | PubMed]
- L. Elkhadragy, M. Chen, K. Miller, M.H. Yang, W. Long. A regulatory BMI1/let-7i/ERK3 pathway controls the motility of head and neck cancer cells. Mol. Oncol., 2017. [DOI | PubMed]
- K. Wolska-Gawron, J. Bartosinska, M. Rusek, M. Kowal, D. Raczkiewicz, D. Krasowska. Circulating miRNA-181b-5p, miRNA-223-3p, miRNA-210-3p, let 7i-5p, miRNA-21-5p and miRNA-29a-3p in patients with localized scleroderma as potential biomarkers. Sci. Rep., 2020. [DOI | PubMed]
- K. Kimura, H. Hohjoh, M. Fukuoka, W. Sato, S. Oki, C. Tomi, H. Yamaguchi, T. Kondo, R. Takahashi, T. Yamamura. Circulating exosomes suppress the induction of regulatory T cells via let-7i in multiple sclerosis. Nat. Commun., 2018. [DOI | PubMed]
- L. Zhang, C. Hao, S. Yao, R. Tang, W. Guo, H. Cong, J. Li, L. Bao, D. Wang, Y. Li. Exosomal miRNA Profiling to Identify Nanoparticle Phagocytic Mechanisms. Small, 2018. [DOI | PubMed]
- B. You, P. Zhang, M. Gu, H. Yin, Y. Fan, H. Yao, S. Pan, H. Xie, T. Cheng, H. Liu. Let-7i-5p promotes a malignant phenotype in nasopharyngeal carcinoma via inhibiting tumor-suppressive autophagy. Cancer Lett., 2022. [DOI | PubMed]
- Y. Liu, X. Hu, L. Hu, C. Xu, X. Liang. Let-7i-5p enhances cell proliferation, migration and invasion of ccRCC by targeting HABP4. BMC Urol., 2021. [DOI | PubMed]
- X. Xu, H.R. Shen, M. Yu, M.R. Du, X.L. Li. MicroRNA let-7i inhibits granulosa-luteal cell proliferation and oestradiol biosynthesis by directly targeting IMP2. Reprod. Biomed. Online, 2022. [DOI | PubMed]
- X. Xu, H.-R. Shen, M. Yu, M.-R. Du, X.-L. Li. Let-7i Regulates KGN Proliferation and Estradiol Biosynthesis By Directly Targeting IMP2 in Polycystic Ovary Syndrome. Res. Sq., 2021. [DOI]
- M. Sahu, B. Mallick. Deciphering synergistic regulatory networks of microRNAs in hESCs and fibroblasts. Int. J. Biol. Macromol., 2018. [DOI | PubMed]
- S. Banerjee, S.R. Kalyani Yabalooru, D. Karunagaran. Identification of mRNA and non-coding RNA hubs using network analysis in organ tropism regulated triple negative breast cancer metastasis. Comput. Biol. Med., 2020. [DOI | PubMed]
- S. Xie, Q. Zhang, J. Zhao, J. Hao, J. Fu, Y. Li. MiR-423-5p may regulate ovarian response to ovulation induction via CSF1. Reprod. Biol. Endocrinol., 2020. [DOI | PubMed]
- S. Li, A. Zeng, Q. Hu, W. Yan, Y. Liu, Y. You. miR-423-5p contributes to a malignant phenotype and temozolomide chemoresistance in glioblastomas. Neuro Oncol., 2017. [DOI | PubMed]
- S. Zhao, J. Li, J. Feng, Z. Li, Q. Liu, P. Lv, F. Wang, H. Gao, Y. Zhang. Identification of Serum miRNA-423-5p Expression Signature in Somatotroph Adenomas. Int. J. Endocrinol., 2019. [DOI | PubMed]
- H. Zhang, D. Liu, S. Zhu, F. Wang, X. Sun, S. Yang, C. Wang. Plasma Exosomal Mir-423-5p Is Involved in the Occurrence and Development of Bicuspid Aortopathy via TGF-beta/SMAD2 Pathway. Front. Physiol., 2021. [DOI | PubMed]
- X. Wang, Y. Liu, Q. Yang, Y. Shu, C. Sun, L. Yin, J. Zou, P. Zhan, Y. Wang, M. Wu. Prostaglandin E2 Promotes Retinal Microvascular Endothelial cell-derived miR-423-5p-containing extracellular vesicles inducing Muller cell activation in diabetic retinopathy. Traffic, 2022. [DOI]
- T. Dai, X. Zhao, Y. Li, L. Yu, Y. Li, X. Zhou, Q. Gong. miR-423 Promotes Breast Cancer Invasion by Activating NF-kappaB Signaling. Onco Targets Ther., 2020. [DOI | PubMed]
- Y. Wang, X. Yu, L. Wang, W. Ma, Q. Sun. miR-320b Is Down-Regulated in Psoriasis and Modulates Keratinocyte Proliferation by Targeting AKT3. Inflammation, 2018. [DOI | PubMed]
- J. Wang, H. Zhao, J. Yu, X. Xu, H. Jing, N. Li, Y. Tang, S. Wang, Y. Li, J. Cai. MiR-320b/RAD21 axis affects hepatocellular carcinoma radiosensitivity to ionizing radiation treatment through DNA damage repair signaling. Cancer Sci., 2021. [DOI | PubMed]
- N. Laxman, H. Mallmin, O. Nilsson, A. Kindmark. miR-203 and miR-320 Regulate Bone Morphogenetic Protein-2-Induced Osteoblast Differentiation by Targeting Distal-Less Homeobox 5 (Dlx5). Genes, 2016. [DOI]
- X. Lu, B. Yang, H. Yang, L. Wang, H. Li, S. Chen, X. Lu, D. Gu. MicroRNA-320b Modulates Cholesterol Efflux and Atherosclerosis. J. Atheroscler. Thromb., 2022. [DOI | PubMed]
- Y. Luo, H. Ji, Y. Cao, X. Ding, M. Li, H. Song, S. Li, C. WaTableng, H. Wu, J. Meng. miR-26b-5p/TCF-4 Controls the Adipogenic Differentiation of Human Adipose-derived Mesenchymal Stem Cells. Cell Transplant., 2020. [DOI | PubMed]
- S. Jing, J. Tian, Y. Zhang, X. Chen, S. Zheng. Identification of a new pseudogenes/lncRNAs-hsa-miR-26b-5p-COL12A1 competing endogenous RNA network associated with prognosis of pancreatic cancer using bioinformatics analysis. Aging, 2020. [DOI | PubMed]
- P. Hu, Z.S. Dong, S. Zheng, X. Guan, L. Zhang, L. Li, Z. Liu. The effects of miR-26b-5p on fibroblast-like synovial cells in rheumatoid arthritis (RA-FLS) via targeting EZH2. Tissue Cell, 2021. [DOI | PubMed]
- M.F. Zhang, P. Yang, M.Y. Shen, X. Wang, N.X. Gao, X.P. Zhou, L.L. Zhou, Y. Lu. MicroRNA-26b-5p alleviates murine collagen-induced arthritis by modulating Th17 cell plasticity. Cell Immunol., 2021. [DOI | PubMed]
- J.Y. Du, L.F. Wang, Q. Wang, L.D. Yu. miR-26b inhibits proliferation, migration, invasion and apoptosis induction via the downregulation of 6-phosphofructo-2-kinase/fructose-2,6-bisphosphatase-3 driven glycolysis in osteosarcoma cells. Oncol. Rep., 2015. [DOI | PubMed]
- B. Xu, Y. Huang, X. Niu, T. Tao, L. Jiang, N. Tong, S. Chen, N. Liu, W. Zhu, M. Chen. Hsa-miR-146a-5p modulates androgen-independent prostate cancer cells apoptosis by targeting ROCK1. Prostate, 2015. [DOI | PubMed]
- S. Pesce, M. Squillario, M. Greppi, F. Loiacono, L. Moretta, A. Moretta, S. Sivori, P. Castagnola, A. Barla, S. Candiani. New miRNA Signature Heralds Human NK Cell Subsets at Different Maturation Steps: Involvement of miR-146a-5p in the Regulation of KIR Expression. Front. Immunol., 2018. [DOI | PubMed]
- J.R. Contreras, J.K. Palanichamy, T.M. Tran, T.R. Fernando, N.I. Rodriguez-Malave, N. Goswami, V.A. Arboleda, D. Casero, D.S. Rao. MicroRNA-146a modulates B-cell oncogenesis by regulating Egr1. Oncotarget, 2015. [DOI | PubMed]
- Z. Wotschofsky, L. Gummlich, J. Liep, C. Stephan, E. Kilic, K. Jung, J.N. Billaud, H.A. Meyer. Integrated microRNA and mRNA Signature Associated with the Transition from the Locally Confined to the Metastasized Clear Cell Renal Cell Carcinoma Exemplified by miR-146-5p. PLoS ONE, 2016. [DOI | PubMed]
- Y.L. Li, J. Wang, C.Y. Zhang, Y.Q. Shen, H.M. Wang, L. Ding, Y.C. Gu, J.T. Lou, X.T. Zhao, Z.L. Ma. MiR-146a-5p inhibits cell proliferation and cell cycle progression in NSCLC cell lines by targeting CCND1 and CCND2. Oncotarget, 2016. [DOI | PubMed]
- C. Liu, J. Xue, B. Xu, A. Zhang, L. Qin, J. Liu, Y. Yang. Exosomes Derived from miR-146a-5p-Enriched Mesenchymal Stem Cells Protect the Cardiomyocytes and Myocardial Tissues in the Polymicrobial Sepsis through Regulating MYBL1. Stem Cells Int., 2021. [DOI | PubMed]
- E. Peta, R. Cappellesso, G. Masi, A. Sinigaglia, M. Trevisan, A. Grassi, B. Di Camillo, E. Vassarotto, A. Fassina, G. Palu. Down-regulation of microRNA-146a is associated with high-risk human papillomavirus infection and epidermal growth factor receptor overexpression in penile squamous cell carcinoma. Hum. Pathol., 2017. [DOI | PubMed]
- H.X. Ye, L. Li, Y.J. Dong, P.H. Li, Q. Su, Y.H. Guo, Y.R. Lu, Y. Zhong, Y. Jia, J.Q. Cheng. miR-146a-5p improves the decidual cytokine microenvironment by regulating the toll-like receptor signaling pathway in unexplained spontaneous abortion. Int. Immunopharmacol., 2020. [DOI | PubMed]
- C. Wang, S. Guan, F. Liu, X. Chen, L. Han, D. Wang, E.U. Nesa, X. Wang, C. Bao, N. Wang. Prognostic and diagnostic potential of miR-146a in oesophageal squamous cell carcinoma. Br. J. Cancer, 2016. [DOI | PubMed]
- B. Xu, N. Wang, X. Wang, N. Tong, N. Shao, J. Tao, P. Li, X. Niu, N. Feng, L. Zhang. MiR-146a suppresses tumor growth and progression by targeting EGFR pathway and in a p-ERK-dependent manner in castration-resistant prostate cancer. Prostate, 2012. [DOI | PubMed]
- J. Mei, R. Bachoo, C.L. Zhang. MicroRNA-146a inhibits glioma development by targeting Notch1. Mol. Cell. Biol., 2011. [DOI | PubMed]
- Y. Cui, K. She, D. Tian, P. Zhang, X. Xin. miR-146a Inhibits Proliferation and Enhances Chemosensitivity in Epithelial Ovarian Cancer via Reduction of SOD2. Oncol. Res., 2016. [DOI | PubMed]
- Y. Araki, H. Aiba, T. Yoshida, N. Yamamoto, K. Hayashi, A. Takeuchi, S. Miwa, K. Igarashi, T.D. Nguyen, K.A. Ishii. Osteosarcoma-Derived Small Extracellular Vesicles Enhance Tumor Metastasis and Suppress Osteoclastogenesis by miR-146a-5p. Front. Oncol., 2021. [DOI | PubMed]
- Z. Wang, C. Zhou, Y. Sun, Y. Chen, D. Xue. Let-7c-5p Is Involved in Chronic Kidney Disease by Targeting TGF-beta Signaling. Biomed. Res. Int., 2020. [DOI | PubMed]
- G.X. Liu, S. Ma, Y. Li, Y. Yu, Y.X. Zhou, Y.D. Lu, L. Jin, Z.L. Wang, J.H. Yu. Hsa-let-7c controls the committed differentiation of IGF-1-treated mesenchymal stem cells derived from dental pulps by targeting IGF-1R via the MAPK pathways. Exp. Mol. Med., 2018. [DOI]
- Y. Wang, H. Ren, Q. Wang, S. Li, Y. Lu, W. Masu, J. Zhang, T. Wang. Let-7c-5p/IGF2BP1 axis in Oral Squamous Cell Carcinoma represses progressions in vitro. Trop. J. Pharm. Res., 2021. [DOI]
- L.V. Rhodes, E.C. Martin, H.C. Segar, D.F. Miller, A. Buechlein, D.B. Rusch, K.P. Nephew, M.E. Burow, B.M. Collins-Burow. Dual regulation by microRNA-200b-3p and microRNA-200b-5p in the inhibition of epithelial-to-mesenchymal transition in triple-negative breast cancer. Oncotarget, 2015. [DOI | PubMed]
- K.H. Lee, B.J. Lim, V.H. Ferreira, S.Y. Min, Y.M. Hong, J.H. Jo, S.H. Han. Expression of human miR-200b-3p and -200c-3p in cytomegalovirus-infected tissues. Biosci. Rep., 2018. [DOI | PubMed]
- A. Moh-Moh-Aung, M. Fujisawa, S. Ito, H. Katayama, T. Ohara, Y. Ota, T. Yoshimura, A. Matsukawa. Decreased miR-200b-3p in cancer cells leads to angiogenesis in HCC by enhancing endothelial ERG expression. Sci. Rep., 2020. [DOI | PubMed]
- C.C. Nwaeburu, A. Abukiwan, Z. Zhao, I. Herr. Quercetin-induced miR-200b-3p regulates the mode of self-renewing divisions in pancreatic cancer. Mol. Cancer, 2017. [DOI | PubMed]
- F. Zhang, N. Cheng, J. Du, H. Zhang, C. Zhang. MicroRNA-200b-3p promotes endothelial cell apoptosis by targeting HDAC4 in atherosclerosis. BMC Cardiovasc. Disord., 2021. [DOI | PubMed]
- C. Li, F. Qin, M. Xue, Y. Lei, F. Hu, H. Xu, G. Sun, T. Wang, M. Guo. miR-429 and miR-424-5p inhibit cell proliferation and Ca2+ influx by downregulating CaSR in pulmonary artery smooth muscle cells. Am. J. Physiol. Cell Physiol., 2019. [DOI | PubMed]
- Y. Deng, J. Wu, X. Li. lncRNA RUNDC3A-AS1 Regulates Proliferation and Apoptosis of Thyroid Cancer Cells via the miR-151b/SNRPB Axis. Int. J. Endocrinol., 2022. [DOI | PubMed]
- S. Wang, Y. Pan, R. Zhang, T. Xu, W. Wu, R. Zhang, C. Wang, H. Huang, C.A. Calin, H. Yang. Hsa-miR-24-3p increases nasopharyngeal carcinoma radiosensitivity by targeting both the 3’UTR and 5’UTR of Jab1/CSN5. Oncogene, 2016. [DOI | PubMed]
- X. Dong, W. Ding, J. Ye, D. Yan, F. Xue, L. Xu, J. Yin, W. Guo. MiR-24-3p enhances cell growth in hepatocellular carcinoma by targeting metallothionein 1M. Cell Biochem. Funct., 2016. [DOI | PubMed]
- P. Jing, N. Zhao, N. Xie, M. Ye, Y. Zhang, Z. Zhang, M. Li, X. Lai, J. Zhang, Z. Gu. miR-24-3p/FGFR3 Signaling as a Novel Axis Is Involved in Epithelial-Mesenchymal Transition and Regulates Lung Adenocarcinoma Progression. J. Immunol. Res., 2018. [DOI | PubMed]
- M.J. Kang, S.Y. Park, J.S. Han. MicroRNA-24-3p regulates neuronal differentiation by controlling hippocalcin expression. Cell Mol. Life Sci., 2019. [DOI | PubMed]
- W. Huang, J. Gu, T. Tao, J. Zhang, H. Wang, Y. Fan. MiR-24-3p Inhibits the Progression of Pancreatic Ductal Adenocarcinoma Through LAMB3 Downregulation. Front. Oncol., 2019. [DOI | PubMed]
- H. Kang, J.G. Rho, C. Kim, H. Tak, H. Lee, E. Ji, S. Ahn, A.R. Shin, H.I. Cho, Y.H. Huh. The miR-24-3p/p130Cas: A novel axis regulating the migration and invasion of cancer cells. Sci. Rep., 2017. [DOI | PubMed]
- Q. Li, N. Wang, H. Wei, C. Li, J. Wu, G. Yang. miR-24-3p Regulates Progression of Gastric Mucosal Lesions and Suppresses Proliferation and Invasiveness of N87 Via Peroxiredoxin 6. Dig. Dis. Sci., 2016. [DOI | PubMed]
- S. Moncini, A. Salvi, P. Zuccotti, G. Viero, A. Quattrone, S. Barlati, G. De Petro, M. Venturin, P. Riva. The role of miR-103 and miR-107 in regulation of CDK5R1 expression and in cellular migration. PLoS ONE, 2011. [DOI | PubMed]
- Z. Wang, X. Zhuang, B. Chen, D. Feng, G. Li, M. Wei. The Role of miR-107 as a Potential Biomarker and Cellular Factor for Acute Aortic Dissection. DNA Cell Biol., 2020. [DOI | PubMed]
- Y. Luo, T. Hua, X. You, J. Lou, X. Yang, N. Tang. Effects of MiR-107 on The Chemo-drug Sensitivity of Breast Cancer Cells. Open Med., 2019. [DOI]
- P. Sharma, N. Saini, R. Sharma. miR-107 functions as a tumor suppressor in human esophageal squamous cell carcinoma and targets Cdc42. Oncol. Rep., 2017. [DOI | PubMed]
- H. Rajabi, S. Aslani, R. Rahbarghazi. Level of miR-101a and miR-107 in Human Adipose Mesenchymal Stem Cells Committed to Insulin-producing Cells. Int. J. Mol. Cell. Med., 2021. [DOI | PubMed]
- F. Tian, J. Wang, Z. Zhang, J. Yang. miR-107 modulates chondrocyte proliferation, apoptosis, and extracellular matrix synthesis by targeting PTEN. Int. J. Clin. Exp. Pathol., 2019. [PubMed]
- L. Chen, X.R. Chen, R. Zhang, P. Li, Y. Liu, K. Yan, X.D. Jiang. MicroRNA-107 inhibits glioma cell migration and invasion by modulating Notch2 expression. J. Neurooncol., 2013. [DOI | PubMed]
- Z. Zhang, L. Zhang, Z.Y. Yin, X.L. Fan, B. Hu, L.Q. Wang, D. Zhang. miR-107 regulates cisplatin chemosensitivity of A549 non small cell lung cancer cell line by targeting cyclin dependent kinase 8. Int. J. Clin. Exp. Pathol., 2014. [PubMed]
- W. Ren, X. Zhang, W. Li, Q. Feng, H. Feng, Y. Tong, H. Rong, W. Wang, D. Zhang, Z. Zhang. Exosomal miRNA-107 induces myeloid-derived suppressor cell expansion in gastric cancer. Cancer Manag. Res., 2019. [DOI | PubMed]
- S. Meng, J. Cao, L. Wang, Q. Zhou, Y. Li, C. Shen, X. Zhang, C. Wang. MicroRNA 107 partly inhibits endothelial progenitor cells differentiation via HIF-1beta. PLoS ONE, 2012. [DOI | PubMed]
- Y. Li, L. Mao, Y. Gao, S. Baral, Y. Zhou, B. Hu. MicroRNA-107 contributes to post-stroke angiogenesis by targeting Dicer-1. Sci. Rep., 2015. [DOI | PubMed]
- Y.C. Wang, H. Xie, Y.C. Zhang, Q.H. Meng, M.M. Xiong, M.W. Jia, F. Peng, D.L. Tang. Exosomal miR-107 antagonizes profibrotic phenotypes of pericytes by targeting a pathway involving HIF-1alpha/Notch1/PDGFRbeta/YAP1/Twist1 axis in vitro. Am. J. Physiol. Heart Circ. Physiol., 2021. [DOI | PubMed]
- C. Wei, S. Xiang, Y. Yu, J. Song, M. Zheng, F. Lian. miR-221-3p regulates apoptosis of ovarian granulosa cells via targeting FOXO1 in older women with diminished ovarian reserve (DOR). Mol. Reprod. Dev., 2021. [DOI | PubMed]
- X. Li, M. Tang. Exosomes released from M2 macrophages transfer miR-221-3p contributed to EOC progression through targeting CDKN1B. Cancer Med., 2020. [DOI | PubMed]
- Y. Tu, W. Wu, Y. Guo, F. Lu, X. Li, D. Xu, D. Zou, Y. Tu, Y. Chai, L. He. Up-regulation of hsa-miR-221-3p induced by UVB affects proliferation and apoptosis of keratinocytes via Bcl-xL/Bax pathway. Photodermatol. Photoimmunol. Photomed., 2021. [DOI | PubMed]
- K. Zhang, Y. Liang, Y. Feng, W. Wu, H. Zhang, J. He, Q. Hu, J. Zhao, Y. Xu, Z. Liu. Decreased epithelial and sputum miR-221-3p associates with airway eosinophilic inflammation and CXCL17 expression in asthma. Am. J. Physiol. Lung Cell. Mol. Physiol., 2018. [DOI | PubMed]
- S. Binas, M. Knyrim, J. Hupfeld, U. Kloeckner, S. Rabe, S. Mildenberger, K. Quarch, N. Stratz, D. Misiak, M. Gekle. miR-221 and -222 target CACNA1C and KCNJ5 leading to altered cardiac ion channel expression and current density. Cell. Mol. Life Sci., 2020. [DOI | PubMed]
- Y. Yang, H. Cui, X. Wang. Downregulation of EIF5A2 by miR-221-3p inhibits cell proliferation, promotes cell cycle arrest and apoptosis in medulloblastoma cells. Biosci. Biotechnol. Biochem., 2019. [DOI | PubMed]
- Y. Goto, S. Kojima, R. Nishikawa, A. Kurozumi, M. Kato, H. Enokida, R. Matsushita, K. Yamazaki, Y. Ishida, M. Nakagawa. MicroRNA expression signature of castration-resistant prostate cancer: The microRNA-221/222 cluster functions as a tumour suppressor and disease progression marker. Br. J. Cancer, 2015. [DOI | PubMed]
- N. Felli, L. Fontana, E. Pelosi, R. Botta, D. Bonci, F. Facchiano, F. Liuzzi, V. Lulli, O. Morsilli, S. Santoro. MicroRNAs 221 and 222 inhibit normal erythropoiesis and erythroleukemic cell growth via kit receptor down-modulation. Proc. Natl. Acad. Sci. USA, 2005. [DOI | PubMed]
- F. Fornari, L. Gramantieri, M. Ferracin, A. Veronese, S. Sabbioni, G.A. Calin, G.L. Grazi, C. Giovannini, C.M. Croce, L. Bolondi. MiR-221 controls CDKN1C/p57 and CDKN1B/p27 expression in human hepatocellular carcinoma. Oncogene, 2008. [DOI | PubMed]
- H.J. Bae, K.H. Jung, J.W. Eun, Q. Shen, H.S. Kim, S.J. Park, W.C. Shin, H.D. Yang, W.S. Park, J.Y. Lee. MicroRNA-221 governs tumor suppressor HDAC6 to potentiate malignant progression of liver cancer. J. Hepatol., 2015. [DOI | PubMed]
- C.M. Gits, P.F. van Kuijk, M.B. Jonkers, A.W. Boersma, W.F. van Ijcken, A. Wozniak, R. Sciot, P. Rutkowski, P. Schoffski, T. Taguchi. MiR-17-92 and miR-221/222 cluster members target KIT and ETV1 in human gastrointestinal stromal tumours. Br. J. Cancer, 2013. [DOI | PubMed]
- I. Daugaard, K.J. Sanders, A. Idica, K. Vittayarukskul, M. Hamdorf, J.D. Krog, R. Chow, D. Jury, L.L. Hansen, H. Hager. miR-151a induces partial EMT by regulating E-cadherin in NSCLC cells. Oncogenesis, 2017. [DOI | PubMed]
- Y. Hu, M. Luo. NORAD-sponged miR-378c alleviates malignant behaviors of stomach adenocarcinoma via targeting NRP1. Cancer Cell Int., 2022. [DOI | PubMed]
- A. Shaer, N. Azarpira, A. Vahdati, M.H. Karimi, M. Shariati. miR-375 induces human decidua basalis-derived stromal cells to become insulin-producing cells. Cell. Mol. Biol. Lett., 2014. [DOI | PubMed]
- Z. Lu, J. Wang, X. Wang, Z. Li, D. Niu, M. Wang, J. Xiang, Y. Yue, Y. Xia, X. Li. miR-375 Promotes Pancreatic Differentiation In Vitro by Affecting Different Target Genes at Different Stages. Stem Cells Int., 2021. [DOI | PubMed]
- J. Yi, L. Jin, J. Chen, B. Feng, Z. He, L. Chen, H. Song. MiR-375 suppresses invasion and metastasis by direct targeting of SHOX2 in esophageal squamous cell carcinoma. Acta Biochim. Biophys. Sin., 2017. [DOI | PubMed]
- Y. Chang, W. Yan, X. He, L. Zhang, C. Li, H. Huang, G. Nace, D.A. Geller, J. Lin, A. Tsung. miR-375 inhibits autophagy and reduces viability of hepatocellular carcinoma cells under hypoxic conditions. Gastroenterology, 2012. [DOI | PubMed]
- Q. Li, T. Huyan, S. Cai, Q. Huang, M. Zhang, H. Peng, Y. Zhang, N. Liu, W. Zhang. The role of exosomal miR-375-3p: A potential suppressor in bladder cancer via the Wnt/beta-catenin pathway. FASEB J., 2020. [DOI | PubMed]
- A. Jafarian, M. Taghikani, S. Abroun, A. Allahverdi, M. Lamei, N. Lakpour, M. Soleimani. The Generation of Insulin Producing Cells from Human Mesenchymal Stem Cells by MiR-375 and Anti-MiR-9. PLoS ONE, 2015. [DOI | PubMed]
- G. Nathan, S. Kredo-Russo, T. Geiger, A. Lenz, H. Kaspi, E. Hornstein, S. Efrat. MiR-375 promotes redifferentiation of adult human beta cells expanded in vitro. PLoS ONE, 2015. [DOI | PubMed]
- H.M. Jung, B.L. Phillips, E.K. Chan. miR-375 activates p21 and suppresses telomerase activity by coordinately regulating HPV E6/E7, E6AP, CIP2A, and 14-3-3zeta. Mol. Cancer, 2014. [DOI | PubMed]
- Z.K. Chang, F.G. Meng, Z.Q. Zhang, G.P. Mao, Z.Y. Huang, W.M. Liao, A.S. He. MicroRNA-193b-3p regulates matrix metalloproteinase 19 expression in interleukin-1beta-induced human chondrocytes. J. Cell. Biochem., 2018. [DOI | PubMed]
- X. Chen, R. Zhang, Q. Zhang, Z. Xu, F. Xu, D. Li, Y. Li. Chondrocyte sheet in vivo cartilage regeneration technique using miR-193b-3p to target MMP16. Aging, 2019. [DOI | PubMed]
- F. Meng, Z. Li, Z. Zhang, Z. Yang, Y. Kang, X. Zhao, D. Long, S. Hu, M. Gu, S. He. MicroRNA-193b-3p regulates chondrogenesis and chondrocyte metabolism by targeting HDAC3. Theranostics, 2018. [DOI | PubMed]
- J. Zhang, J. Qin, Y. Su. miR-193b-3p possesses anti-tumor activity in ovarian carcinoma cells by targeting p21-activated kinase 3. Biomed. Pharmacol., 2017. [DOI | PubMed]
- N. Lai, D. Wu, T. Liang, P. Pan, G. Yuan, X. Li, H. Li, H. Shen, Z. Wang, G. Chen. Systemic exosomal miR-193b-3p delivery attenuates neuroinflammation in early brain injury after subarachnoid hemorrhage in mice. J. Neuroinflamm., 2020. [DOI | PubMed]
- Y. Wang, W. Ye, Y. Liu, B. Mei, X. Liu, Q. Huang. Osteoporosis genome-wide association study variant c.3781 C>A is regulated by a novel anti-osteogenic factor miR-345-5p. Hum. Mutat., 2020. [DOI | PubMed]
- P. Kong, X. Zhu, Q. Geng, L. Xia, X. Sun, Y. Chen, W. Li, Z. Zhou, Y. Zhan, D. Xu. The microRNA-423-3p-Bim Axis Promotes Cancer Progression and Activates Oncogenic Autophagy in Gastric Cancer. Mol. Ther., 2017. [DOI | PubMed]
- R. Aghdam, M. Habibi, G. Taheri. Using informative features in machine learning based method for COVID-19 drug repurposing. J. Cheminform., 2021. [DOI | PubMed]
- L. Yan, M.C. Yu, G.L. Gao, H.W. Liang, X.Y. Zhou, Z.T. Zhu, C.Y. Zhang, Y.B. Wang, X. Chen. MiR-125a-5p functions as a tumour suppressor in breast cancer by downregulating BAP1. J. Cell. Biochem., 2018. [DOI | PubMed]
- Y. Zhang, D. Zhang, J. Lv, S. Wang, Q. Zhang. MiR-125a-5p suppresses bladder cancer progression through targeting FUT4. Biomed. Pharmacol., 2018. [DOI]
- X. Qin, Y. Wan, S. Wang, M. Xue. MicroRNA-125a-5p modulates human cervical carcinoma proliferation and migration by targeting ABL2. Drug Des. Dev., 2016. [DOI]
- X. Xu, Y. Tao, Y. Niu, Z. Wang, C. Zhang, Y. Yu, L. Ma. miR-125a-5p inhibits tumorigenesis in hepatocellular carcinoma. Aging, 2019. [DOI | PubMed]
- Y. Cao, S. Tan, Y. Tu, G. Zhang, Y. Liu, D. Li, S. Xu, Z. Le, J. Xiong, W. Zou. MicroRNA-125a-5p inhibits invasion and metastasis of gastric cancer cells by targeting BRMS1 expression. Oncol. Lett., 2018. [DOI | PubMed]
- Z. Tong, N. Liu, L. Lin, X. Guo, D. Yang, Q. Zhang. miR-125a-5p inhibits cell proliferation and induces apoptosis in colon cancer via targeting BCL2, BCL2L12 and MCL1. Biomed. Pharmacol., 2015. [DOI]
- Y. Fu, F. Cao. MicroRNA-125a-5p regulates cancer cell proliferation and migration through NAIF1 in prostate carcinoma. Onco Targets Ther., 2015. [DOI | PubMed]
- X. Yang, J. Qiu, H. Kang, Y. Wang, J. Qian. miR-125a-5p suppresses colorectal cancer progression by targeting VEGFA. Cancer Manag. Res., 2018. [DOI | PubMed]
- L. Jiang, J. Chang, Q. Zhang, L. Sun, X. Qiu. MicroRNA hsa-miR-125a-3p activates p53 and induces apoptosis in lung cancer cells. Cancer Investig., 2013. [DOI | PubMed]
- L. Jiang, Q. Huang, J. Chang, E. Wang, X. Qiu. MicroRNA HSA-miR-125a-5p induces apoptosis by activating p53 in lung cancer cells. Exp. Lung Res., 2011. [DOI | PubMed]
- G. Li, J. Li, C. Li, H. Qi, P. Dong, J. Zheng, F. Yu. MicroRNA-125a-5p Contributes to Hepatic Stellate Cell Activation through Targeting FIH1. Cell. Physiol. Biochem., 2016. [DOI | PubMed]
- Z. Xueya, L. Yamei, C. Sha, C. Dan, S. Hong, Y. Xingyu, C. Weiwei. Exosomal encapsulation of miR-125a-5p inhibited trophoblast cell migration and proliferation by regulating the expression of VEGFA in preeclampsia. Biochem. Biophys. Res. Commun., 2020. [DOI | PubMed]
- L.A. Vonk, A.H. Kragten, W.J. Dhert, D.B. Saris, L.B. Creemers. Overexpression of hsa-miR-148a promotes cartilage production and inhibits cartilage degradation by osteoarthritic chondrocytes. Osteoarthr. Cartil., 2014. [DOI]
- J. Xuan, H. Xu, H. Li, D. Chen, Y. Qiu, X. Chen, M. Shao, X. Xia. Extracellular Vesicles from miR-148a-5p-Enriched Bone Marrow Mesenchymal Stem Cells Relieve Hepatic Fibrosis by Targeting Smad4. Mol. Biotechnol., 2022. [DOI | PubMed]
- Y. Tan, X. Lu, Z. Cheng, G. Pan, S. Liu, P. Apiziaji, H. Wang, J. Zhang, Y. Abulimiti. miR-148a Regulates the Stem Cell-Like Side Populations Distribution by Affecting the Expression of ACVR1 in Esophageal Squamous Cell Carcinoma. Onco Targets Ther., 2020. [DOI | PubMed]
- S. Zhang, J. Jin, X. Tian, L. Wu. hsa-miR-29c-3p regulates biological function of colorectal cancer by targeting SPARC. Oncotarget, 2017. [DOI | PubMed]
- K. Lu, F. Feng, Y. Yang, K. Liu, J. Duan, H. Liu, J. Yang, M. Wu, C. Liu, Y. Chang. High-throughput screening identified miR-7-2-3p and miR-29c-3p as metastasis suppressors in gallbladder carcinoma. J. Gastroenterol., 2020. [DOI | PubMed]
- M. Zhuang, Q. Chaolumen, L. Li, B. Chen, Q. Su, Y. Yang, X. Zhang. MiR-29b-3p cooperates with miR-29c-3p to affect the malignant biological behaviors in T-cell acute lymphoblastic leukemia via TFAP2C/GPX1 axis. Biochem. Biophys. Res. Commun., 2020. [DOI | PubMed]
- S. Feng, S. Luo, C. Ji, J. Shi. miR-29c-3p regulates proliferation and migration in ovarian cancer by targeting KIF4A. World J. Surg. Oncol., 2020. [DOI | PubMed]
- J. Liu, G. Tao, C. Zhong, X. Liu. Upregulation of miR-29c-3p Hinders Melanoma Progression by Inhibiting CDCA4 Expression. Biomed Res. Int., 2021. [DOI | PubMed]
- R. Harati, S. Hammad, A. Tlili, M. Mahfood, A. Mabondzo, R. Hamoudi. miR-27a-3p regulates expression of intercellular junctions at the brain endothelium and controls the endothelial barrier permeability. PLoS ONE, 2022. [DOI | PubMed]
- Y. Xu, D. Li, Z. Zhu, L. Li, Y. Jin, C. Ma, W. Zhang. miR27a3p negatively regulates osteogenic differentiation of MC3T3E1 preosteoblasts by targeting osterix. Mol. Med. Rep., 2020. [DOI | PubMed]
- G. Zeng, W. Xun, K. Wei, Y. Yang, H. Shen. MicroRNA-27a-3p regulates epithelial to mesenchymal transition via targeting YAP1 in oral squamous cell carcinoma cells. Oncol. Rep., 2016. [DOI | PubMed]
- J.M. Li, J. Zhou, Z. Xu, H.J. Huang, M.J. Chen, J.S. Ji. MicroRNA-27a-3p inhibits cell viability and migration through down-regulating DUSP16 in hepatocellular carcinoma. J. Cell. Biochem., 2018. [DOI | PubMed]
- L. Chen, Q. Lu, J. Chen, R. Feng, C. Yang. Upregulating miR-27a-3p inhibits cell proliferation and inflammation of rheumatoid arthritis synovial fibroblasts through targeting toll-like receptor 5. Exp. Med., 2021. [DOI]
- H. Wu, T. Pula, D. Tews, E.Z. Amri, K.M. Debatin, M. Wabitsch, P. Fischer-Posovszky, J. Roos. microRNA-27a-3p but Not -5p Is a Crucial Mediator of Human Adipogenesis. Cells, 2021. [DOI | PubMed]
- L. Yin, X. Xiao, C. Georgikou, Y. Yin, L. Liu, S. Karakhanova, Y. Luo, J. Gladkich, J. Fellenberg, C. Sticht. MicroRNA-365a-3p inhibits c-Rel-mediated NF-kappaB signaling and the progression of pancreatic cancer. Cancer Lett., 2019. [DOI | PubMed]
- R. Yang, G. Liu, L. Han, Y. Qiu, L. Wang, M. Wang. MiR-365a-3p-Mediated Regulation of HELLS/GLUT1 Axis Suppresses Aerobic Glycolysis and Gastric Cancer Growth. Front. Oncol., 2021. [DOI | PubMed]
- Q. Tian, H.F. Sun, W.J. Wang, Q. Li, J. Ding, W. Di. miRNA-365b promotes hepatocellular carcinoma cell migration and invasion by downregulating SGTB. Future Oncol., 2019. [DOI | PubMed]
- C. Oliveira-Rizzo, M.C. Ottati, R.S. Fort, S. Chavez, J.M. Trinidad, A. DiPaolo, B. Garat, J.R. Sotelo-Silveira, M.A. Duhagon. Hsa-miR-183-5p Modulates Cell Adhesion by Repression of ITGB1 Expression in Prostate Cancer. Non-Coding RNA, 2022. [DOI | PubMed]
- R. Akbar, K. Ullah, T.U. Rahman, Y. Cheng, H.Y. Pang, L.Y. Jin, Q.J. Wang, H.F. Huang, J.Z. Sheng. miR-183-5p regulates uterine receptivity and enhances embryo implantation. J. Mol. Endocrinol., 2020. [DOI | PubMed]
- J. Chen, L. Gu, J. Ni, P. Hu, K. Hu, Y.L. Shi. MiR-183 Regulates ITGB1P Expression and Promotes Invasion of Endometrial Stromal Cells. Biomed. Res. Int., 2015. [DOI | PubMed]
- M.T. Nguyen, K.H. Min, W. Lee. MiR-183-5p induced by saturated fatty acids regulates the myogenic differentiation by directly targeting FHL1 in C2C12 myoblasts. BMB Rep., 2020. [DOI | PubMed]
- S. Mollazadeh, B.S. Fazly Bazzaz, V. Neshati, A.A.F. de Vries, H. Naderi-Meshkin, M. Mojarad, M. Mirahmadi, Z. Neshati, M.A. Kerachian. Overexpression of MicroRNA-148b-3p stimulates osteogenesis of human bone marrow-derived mesenchymal stem cells: The role of MicroRNA-148b-3p in osteogenesis. BMC Med. Genet., 2019. [DOI | PubMed]
- G. Wang, Z. Li, N. Tian, L. Han, Y. Fu, Z. Guo, Y. Tian. miR-148b-3p inhibits malignant biological behaviors of human glioma cells induced by high HOTAIR expression. Oncol. Lett., 2016. [DOI | PubMed]
- S. Yan, Y. Chen, M. Yang, Q. Zhang, J. Ma, B. Liu, L. Yang, X. Li. hsa-MicroRNA-28-5p Inhibits Diffuse Large B-Cell Lymphoma Cell Proliferation by Downregulating 14-3-3zeta Expression. Evid. Based Complement. Altern. Med., 2022. [DOI | PubMed]
- G. Guan, X. Niu, X. Qiao, X. Wang, J. Liu, M. Zhong. Upregulation of Neural Cell Adhesion Molecule 1 (NCAM1) by hsa-miR-141-3p Suppresses Ameloblastoma Cell Migration. Med. Sci. Monit., 2020. [DOI | PubMed]
- N. Wang, P. Li, W. Liu, N. Wang, Z. Lu, J. Feng, X. Zeng, J. Yang, Y. Wang, W. Zhao. miR-141-3p suppresses proliferation and promotes apoptosis by targeting GLI2 in osteosarcoma cells. Oncol. Rep., 2018. [DOI | PubMed]
- M. Huang, L. Wu, Y. Qin, Z. Li, S. Luo, H. Qin, Y. Yang, J. Chen. Anti-proliferative role and prognostic implication of miR-141 in gastric cancer. Am. J. Transl. Res., 2016. [PubMed]
- Z. Liang, X. Li, S. Liu, C. Li, X. Wang, J. Xing. MiR-141-3p inhibits cell proliferation, migration and invasion by targeting TRAF5 in colorectal cancer. Biochem. Biophys. Res. Commun., 2019. [DOI | PubMed]
- S. Masoumi-Dehghi, S. Babashah, M. Sadeghizadeh. microRNA-141-3p-containing small extracellular vesicles derived from epithelial ovarian cancer cells promote endothelial cell angiogenesis through activating the JAK/STAT3 and NF-kappaB signaling pathways. J. Cell Commun. Signal., 2020. [DOI | PubMed]
- M.T. Nguyen, W. Lee. MiR-141-3p regulates myogenic differentiation in C2C12 myoblasts via CFL2-YAP-mediated mechanotransduction. BMB Rep., 2022. [DOI | PubMed]
- S. Siddiqui, K. Johansson, A. Joo, L.R. Bonser, K.D. Koh, O. Le Tonqueze, S. Bolourchi, R.A. Bautista, L. Zlock, T.L. Roth. Epithelial miR-141 regulates IL-13-induced airway mucus production. JCI Insight, 2021. [DOI]
- K.R. Yu, S. Lee, J.W. Jung, I.S. Hong, H.S. Kim, Y. Seo, T.H. Shin, K.S. Kang. MicroRNA-141-3p plays a role in human mesenchymal stem cell aging by directly targeting ZMPSTE24. J. Cell Sci., 2013. [DOI | PubMed]
- H. Liang, Y. Chen, H. Li, X. Yu, C. Xia, Z. Ming, C. Zhong. miR-22-3p Suppresses Endothelial Progenitor Cell Proliferation and Migration via Inhibiting Onecut 1 (OC1)/Vascular Endothelial Growth Factor A (VEGFA) Signaling Pathway and Its Clinical Significance in Venous Thrombosis. Med. Sci. Monit., 2020. [DOI | PubMed]
- V. Saccomani, A. Grassi, E. Piovan, D. Bongiovanni, L. Di Martino, S. Minuzzo, V. Tosello, P. Zanovello. miR-22-3p Negatively Affects Tumor Progression in T-Cell Acute Lymphoblastic Leukemia. Cells, 2020. [DOI]
- X. Wang, Y. Wang, M. Kong, J. Yang. MiR-22-3p suppresses sepsis-induced acute kidney injury by targeting PTEN. Biosci. Rep., 2020. [DOI | PubMed]
- J. Xue, Z. Min, Z. Xia, B. Cheng, B. Lan, F. Zhang, Y. Han, K. Wang, J. Sun. The hsa-miR-181a-5p reduces oxidation resistance by controlling SECISBP2 in osteoarthritis. BMC Musculoskelet. Disord., 2018. [DOI | PubMed]
- H. Shen, X.D. Weng, X.H. Liu, D. Yang, L. Wang, J. Guo, M. Wang, X. Wang, C.H. Diao. miR-181a-5p is downregulated and inhibits proliferation and the cell cycle in prostate cancer. Int. J. Clin. Exp. Pathol., 2018. [PubMed]
- S. He, S. Zeng, Z.W. Zhou, Z.X. He, S.F. Zhou. Hsa-microRNA-181a is a regulator of a number of cancer genes and a biomarker for endometrial carcinoma in patients: A bioinformatic and clinical study and the therapeutic implication. Drug Des. Dev., 2015. [DOI]
- K. Liu, F. Xie, A. Gao, R. Zhang, L. Zhang, Z. Xiao, Q. Hu, W. Huang, Q. Huang, B. Lin. SOX2 regulates multiple malignant processes of breast cancer development through the SOX2/miR-181a-5p, miR-30e-5p/TUSC3 axis. Mol. Cancer, 2017. [DOI | PubMed]
- L. Wu, W.Y. Song, Y. Xie, L.L. Hu, X.M. Hou, R. Wang, Y. Gao, J.N. Zhang, L. Zhang, W.W. Li. miR-181a-5p suppresses invasion and migration of HTR-8/SVneo cells by directly targeting IGF2BP2. Cell Death Dis., 2018. [DOI | PubMed]
- N. Potenza, N. Mosca, P. Mondola, S. Damiano, A. Russo, B. De Felice. Human miR-26a-5p regulates the glutamate transporter SLC1A1 (EAAT3) expression. Relevance in multiple sclerosis. Biochim. Biophys. Acta Mol. Basis Dis., 2018. [DOI | PubMed]
- Z. Rasheed, H.A. Al-Shobaili, N. Rasheed, A. Mahmood, M.I. Khan. MicroRNA-26a-5p regulates the expression of inducible nitric oxide synthase via activation of NF-kappaB pathway in human osteoarthritis chondrocytes. Arch. Biochem. Biophys., 2016. [DOI | PubMed]
- Z.M. Huang, H.F. Ge, C.C. Yang, Y. Cai, Z. Chen, W.Z. Tian, J.L. Tao. MicroRNA-26a-5p inhibits breast cancer cell growth by suppressing RNF6 expression. Kaohsiung J. Med. Sci., 2019. [DOI | PubMed]
- K. Erdmann, K. Kaulke, C. Rieger, K. Salomo, M.P. Wirth, S. Fuessel. MiR-26a and miR-138 block the G1/S transition by targeting the cell cycle regulating network in prostate cancer cells. J. Cancer Res. Clin. Oncol., 2016. [DOI | PubMed]
- W. Wei, Y. Yang, J. Cai, K. Cui, R.X. Li, H. Wang, X. Shang, D. Wei. MiR-30a-5p Suppresses Tumor Metastasis of Human Colorectal Cancer by Targeting ITGB3. Cell. Physiol. Biochem., 2016. [DOI | PubMed]
- J. Yang, S. Rao, R. Cao, S. Xiao, X. Cui, L. Ye. miR-30a-5p suppresses lung squamous cell carcinoma via ATG5-mediated autophagy. Aging, 2021. [DOI | PubMed]
- Y. Li, Y. Li, D. Chen, L. Jin, Z. Su, J. Liu, H. Duan, X. Li, Z. Qi, M. Shi. miR30a5p in the tumorigenesis of renal cell carcinoma: A tumor suppressive microRNA. Mol. Med. Rep., 2016. [DOI | PubMed]
- Y.L. Xie, Y.J. Yang, C. Tang, H.J. Sheng, Y. Jiang, K. Han, L.J. Ding. Estrogen combined with progesterone decreases cell proliferation and inhibits the expression of Bcl-2 via microRNA let-7a and miR-34b in ovarian cancer cells. Clin. Transl. Oncol., 2014. [DOI | PubMed]
- C. Bao, L. Guo. MicroRNA-148a-3p inhibits cancer progression and is a novel screening biomarker for gastric cancer. J. Clin. Lab. Anal., 2020. [DOI | PubMed]
- J. Luo, Y. Hou, W. Ma, M. Xie, Y. Jin, L. Xu, C. Li, Y. Wang, J. Chen, W. Chen. A novel mechanism underlying alcohol dehydrogenase expression: Hsa-miR-148a-3p promotes ADH4 expression via an AGO1-dependent manner in control and ethanol-exposed hepatic cells. Biochem. Pharmacol., 2021. [DOI | PubMed]
- H. Kim, Y. Ko, H. Park, H. Zhang, Y. Jeong, Y. Kim, M. Noh, S. Park, Y.M. Kim, Y.G. Kwon. MicroRNA-148a/b-3p regulates angiogenesis by targeting neuropilin-1 in endothelial cells. Exp. Mol. Med., 2019. [DOI]
- W. Miao, N. Li, B. Gu, G. Yi, Z. Su, H. Cheng. MiR-27b-3p suppresses glioma development via targeting YAP1. Biochem. Cell Biol., 2020. [DOI | PubMed]
- Y. Sun, T. Xu, Y.W. Cao, X.Q. Ding. Antitumor effect of miR-27b-3p on lung cancer cells via targeting Fzd7. Eur. Rev. Med. Pharmacol. Sci., 2017. [PubMed]
- Y. Chen, G. Chen, B. Zhang, C. Liu, Y. Yu, Y. Jin. miR-27b-3p suppresses cell proliferation, migration and invasion by targeting LIMK1 in colorectal cancer. Int. J. Clin. Exp. Pathol., 2017. [PubMed]
- X. Rong, D. Ge, D. Shen, X. Chen, X. Wang, L. Zhang, C. Jia, J. Zeng, Y. He, H. Qiu. miR-27b Suppresses Endothelial Cell Proliferation and Migration by Targeting Smad7 in Kawasaki Disease. Cell. Physiol. Biochem., 2018. [DOI | PubMed]
- W. Peng, S. Zhu, X. Li, J. Weng, S. Chen. miR-27b-3p Suppressed Osteogenic Differentiation of Maxillary Sinus Membrane Stem Cells by Targeting Sp7. Implant Dent., 2017. [DOI | PubMed]
- Y. Li, H. Zhang, Y. Dong, Y. Fan, Y. Li, C. Zhao, C. Wang, J. Liu, X. Li, M. Dong. MiR-146b-5p functions as a suppressor miRNA and prognosis predictor in non-small cell lung cancer. J. Cancer, 2017. [DOI | PubMed]
- Y. Li, Y. Wang, L. Yu, C. Sun, D. Cheng, S. Yu, Q. Wang, Y. Yan, C. Kang, S. Jin. miR-146b-5p inhibits glioma migration and invasion by targeting MMP16. Cancer Lett., 2013. [DOI | PubMed]
- A.I. Garcia, M. Buisson, P. Bertrand, R. Rimokh, E. Rouleau, B.S. Lopez, R. Lidereau, I. Mikaelian, S. Mazoyer. Down-regulation of BRCA1 expression by miR-146a and miR-146b-5p in triple negative sporadic breast cancers. EMBO Mol. Med., 2011. [DOI | PubMed]
- M. Kirchmeyer, F.A. Servais, M. Hamdorf, P.V. Nazarov, A. Ginolhac, R. Halder, L. Vallar, M. Glanemann, C. Rubie, F. Lammert. Cytokine-mediated modulation of the hepatic miRNome: miR-146b-5p is an IL-6-inducible miRNA with multiple targets. J. Leukoc. Biol., 2018. [DOI | PubMed]
- L. Han, Y. Zhou, R. Zhang, K. Wu, Y. Lu, Y. Li, R. Duan, Y. Yao, D. Zhu, Y. Jia. MicroRNA Let-7f-5p Promotes Bone Marrow Mesenchymal Stem Cells Survival by Targeting Caspase-3 in Alzheimer Disease Model. Front. Neurosci., 2018. [DOI | PubMed]
- Y. Yang, Y. Liu, N. Xie, L. Shao, H. Sun, Y. Wei, Y. Sun, P. Wang, Y. Yan, S. Xie. Anticancer roles of let-7f-1-3p in non-small cell lung cancer via direct targeting of integrin beta1. Exp. Med., 2021. [DOI]
- L. Du, X. Tao, X. Shen. Human umbilical cord mesenchymal stem cell-derived exosomes inhibit migration and invasion of breast cancer cells via miR-21-5p/ZNF367 pathway. Breast Cancer, 2021. [DOI | PubMed]
- Q. He, A. Ye, W. Ye, X. Liao, G. Qin, Y. Xu, Y. Yin, H. Luo, M. Yi, L. Xian. Cancer-secreted exosomal miR-21-5p induces angiogenesis and vascular permeability by targeting KRIT1. Cell Death Dis., 2021. [DOI | PubMed]
- C. De Santi, S. Vencken, J. Blake, B. Haase, V. Benes, F. Gemignani, S. Landi, C.M. Greene. Identification of MiR-21-5p as a Functional Regulator of Mesothelin Expression Using MicroRNA Capture Affinity Coupled with Next Generation Sequencing. PLoS ONE, 2017. [DOI | PubMed]
- L. Zhang, L. Yu, Y. Liu, S. Wang, Z. Hou, J. Zhou. miR-21-5p promotes cell proliferation by targeting BCL11B in Thp-1 cells. Oncol. Lett., 2021. [DOI | PubMed]
- L. Yan, R. Cao, Y. Liu, L. Wang, B. Pan, X. Lv, H. Jiao, Q. Zhuang, X. Sun, R. Xiao. MiR-21-5p Links Epithelial-Mesenchymal Transition Phenotype with Stem-Like Cell Signatures via AKT Signaling in Keloid Keratinocytes. Sci. Rep., 2016. [DOI | PubMed]
- X.B. Lv, X. Zhang, L. Deng, L. Jiang, W. Meng, Z. Lu, X. Wang. MiR-92a mediates AZD6244 induced apoptosis and G1-phase arrest of lymphoma cells by targeting Bim. Cell Biol. Int., 2014. [DOI | PubMed]
- M. Li, X. Guan, Y. Sun, J. Mi, X. Shu, F. Liu, C. Li. miR-92a family and their target genes in tumorigenesis and metastasis. Exp. Cell Res., 2014. [DOI | PubMed]
- N. Kalinina, G. Klink, E. Glukhanyuk, T. Lopatina, A. Efimenko, Z. Akopyan, V. Tkachuk. miR-92a regulates angiogenic activity of adipose-derived mesenchymal stromal cells. Exp. Cell Res., 2015. [DOI | PubMed]
- Y. Wang, H. Wang, C. Zhang, C. Zhang, H. Yang, R. Gao, Z. Tong. Plasma Hsa-miR-92a-3p in correlation with lipocalin-2 is associated with sepsis-induced coagulopathy. BMC Infect. Dis., 2020. [DOI | PubMed]
- G. Mao, Z. Zhang, Z. Huang, W. Chen, G. Huang, F. Meng, Z. Zhang, Y. Kang. MicroRNA-92a-3p regulates the expression of cartilage-specific genes by directly targeting histone deacetylase 2 in chondrogenesis and degradation. Osteoarthr. Cartil., 2017. [DOI]
- G. Mao, P. Wu, Z. Zhang, Z. Zhang, W. Liao, Y. Li, Y. Kang. MicroRNA-92a-3p Regulates Aggrecanase-1 and Aggrecanase-2 Expression in Chondrogenesis and IL-1beta-Induced Catabolism in Human Articular Chondrocytes. Cell. Physiol. Biochem., 2017. [DOI | PubMed]
- Y. Xu, L. Shen, F. Li, J. Yang, X. Wan, M. Ouyang. microRNA-16-5p-containing exosomes derived from bone marrow-derived mesenchymal stem cells inhibit proliferation, migration, and invasion, while promoting apoptosis of colorectal cancer cells by downregulating ITGA2. J. Cell. Physiol., 2019. [DOI | PubMed]
- H. Zhang, K. Yang, T. Ren, Y. Huang, X. Tang, W. Guo. miR-16-5p inhibits chordoma cell proliferation, invasion and metastasis by targeting Smad3. Cell Death Dis., 2018. [DOI | PubMed]
- S. Chava, C.P. Reynolds, A.S. Pathania, S. Gorantla, L.Y. Poluektova, D.W. Coulter, S.C. Gupta, M.K. Pandey, K.B. Challagundla. miR-15a-5p, miR-15b-5p, and miR-16-5p inhibit tumor progression by directly targeting MYCN in neuroblastoma. Mol. Oncol., 2020. [DOI | PubMed]
- L. Ruan, X. Qian. MiR-16-5p inhibits breast cancer by reducing AKT3 to restrain NF-kappaB pathway. Biosci. Rep., 2019. [DOI | PubMed]
- M. Calderon-Dominguez, A. Mangas, T. Belmonte, M. Quezada-Feijoo, M. Ramos, R. Toro. Ischemic dilated cardiomyopathy pathophysiology through microRNA-16-5p. Rev. Esp. Cardiol., 2021. [DOI | PubMed]
- F. Wang, A. Mao, J. Tang, Q. Zhang, J. Yan, Y. Wang, C. Di, L. Gan, C. Sun, H. Zhang. microRNA-16-5p enhances radiosensitivity through modulating Cyclin D1/E1-pRb-E2F1 pathway in prostate cancer cells. J. Cell. Physiol., 2019. [DOI | PubMed]
- N. Zhang, W.W. Li, C.M. Lv, Y.W. Gao, X.L. Liu, L. Zhao. miR-16-5p and miR-19b-3p prevent amyloid beta-induced injury by targeting BACE1 in SH-SY5Y cells. Neuroreport, 2020. [DOI | PubMed]
- L.X. Dong, H.L. Bao, Y.Y. Zhang, Y. Liu, G.W. Zhang, F.M. An. MicroRNA-16-5p/BTG2 axis affects neurological function, autophagy and apoptosis of hippocampal neurons in Alzheimer’s disease. Brain Res. Bull., 2021. [DOI | PubMed]
- L. Li, J. Jia, X. Liu, S. Yang, S. Ye, W. Yang, Y. Zhang. MicroRNA-16-5p Controls Development of Osteoarthritis by Targeting SMAD3 in Chondrocytes. Curr. Pharm. Des., 2015. [DOI | PubMed]
- Q. Yao, Y. Xing, Z. Wang, J. Liang, Q. Lin, M. Huang, Y. Chen, B. Lin, X. Xu, W. Chen. MiR-16-5p suppresses myofibroblast activation in systemic sclerosis by inhibiting NOTCH signaling. Aging, 2020. [DOI | PubMed]
- B. Cai, M. Ma, B. Chen, Z. Li, B.A. Abdalla, Q. Nie, X. Zhang. MiR-16-5p targets SESN1 to regulate the p53 signaling pathway, affecting myoblast proliferation and apoptosis, and is involved in myoblast differentiation. Cell Death Dis., 2018. [DOI | PubMed]
- T. Yu, X. You, H. Zhou, W. He, Z. Li, B. Li, J. Xia, H. Zhu, Y. Zhao, G. Yu. MiR-16-5p regulates postmenopausal osteoporosis by directly targeting VEGFA. Aging, 2020. [DOI | PubMed]
- L.S. Normann, M.H. Haugen, M.R. Aure, V.N. Kristensen, G.M. Maelandsmo, K.K. Sahlberg. miR-101-5p Acts as a Tumor Suppressor in HER2-Positive Breast Cancer Cells and Improves Targeted Therapy. Breast Cancer (Dove Med. Press), 2022. [DOI | PubMed]
- S. Yan, X. Shan, K. Chen, Y. Liu, G. Yu, Q. Chen, T. Zeng, L. Zhu, H. Dang, F. Chen. LINC00052/miR-101-3p axis inhibits cell proliferation and metastasis by targeting SOX9 in hepatocellular carcinoma. Gene, 2018. [DOI | PubMed]
- Y. Sheng, S. Ding, K. Chen, J. Chen, S. Wang, C. Zou, J. Zhang, Y. Cao, A. Huang, H. Tang. Functional analysis of miR-101-3p and Rap1b involved in hepatitis B virus-related hepatocellular carcinoma pathogenesis. Biochem. Cell Biol., 2014. [DOI | PubMed]
- L. Li, M.Y. Shao, S.C. Zou, Z.F. Xiao, Z.C. Chen. MiR-101-3p inhibits EMT to attenuate proliferation and metastasis in glioblastoma by targeting TRIM44. J. Neurooncol., 2019. [DOI | PubMed]
- C. Wang, B. Liu. miR-101-3p induces autophagy in endometrial carcinoma cells by targeting EZH2. Arch. Gynecol. Obstet., 2018. [DOI | PubMed]
- X. Guo, M. Chen, L. Cao, Y. Hu, X. Li, Q. Zhang, Y. Ren, X. Wu, Z. Meng, K. Xu. Cancer-Associated Fibroblasts Promote Migration and Invasion of Non-Small Cell Lung Cancer Cells via miR-101-3p Mediated VEGFA Secretion and AKT/eNOS Pathway. Front. Cell Dev. Biol., 2021. [DOI | PubMed]
- Y. Dong, Y. Gao, T. Xie, H. Liu, X. Zhan, Y. Xu. miR-101-3p Serves as a Tumor Suppressor for Renal Cell Carcinoma and Inhibits Its Invasion and Metastasis by Targeting EZH2. Biomed Res. Int., 2021. [DOI | PubMed]
- C. Luo, P.R. Merz, Y. Chen, E. Dickes, A. Pscherer, D. Schadendorf, S.B. Eichmuller. MiR-101 inhibits melanoma cell invasion and proliferation by targeting MITF and EZH2. Cancer Lett., 2013. [DOI | PubMed]
- M. Ziemann, S.C. Lim, Y. Kang, S. Samuel, I.L. Sanchez, M. Gantier, D. Stojanovski, M. McKenzie. MicroRNA-101-3p Modulates Mitochondrial Metabolism via the Regulation of Complex II Assembly. J. Mol. Biol., 2022. [DOI | PubMed]
- Q. Chen, X. Li, L. Kong, Q. Xu, Z. Wang, Q. Lv. miR-101-3p induces vascular endothelial cell dysfunction by targeting tet methylcytosine dioxygenase 2. Acta Biochim. Biophys. Sin., 2020. [DOI | PubMed]
- E. Kaneda-Ikeda, T. Iwata, N. Mizuno, T. Nagahara, M. Kajiya, K. Ouhara, M. Yoshioka, S. Ishida, H. Kawaguchi, H. Kurihara. Regulation of osteogenesis via miR-101-3p in mesenchymal stem cells by human gingival fibroblasts. J. Bone Miner. Metab., 2020. [DOI | PubMed]
- Y. He, X. Chen, Y. Yu, J. Li, Q. Hu, C. Xue, J. Chen, S. Shen, Y. Luo, F. Ren. LDHA is a direct target of miR-30d-5p and contributes to aggressive progression of gallbladder carcinoma. Mol. Carcinog., 2018. [DOI | PubMed]
- T. Bjornetro, K.R. Redalen, S. Meltzer, N.S. Thusyanthan, R. Samiappan, C. Jegerschold, K.R. Handeland, A.H. Ree. An experimental strategy unveiling exosomal microRNAs 486-5p, 181a-5p and 30d-5p from hypoxic tumour cells as circulating indicators of high-risk rectal cancer. J. Extracell. Vesicles, 2019. [DOI | PubMed]
- H. Jacob, L. Stanisavljevic, K.E. Storli, K.E. Hestetun, O. Dahl, M.P. Myklebust. Identification of a sixteen-microRNA signature as prognostic biomarker for stage II and III colon cancer. Oncotarget, 2017. [DOI | PubMed]
- Y. Song, C. Song, S. Yang. Tumor-Suppressive Function of miR-30d-5p in Prostate Cancer Cell Proliferation and Migration by Targeting NT5E. Cancer Biother. Radiopharm., 2018. [DOI | PubMed]
- G. Wang, B. Feng, Y. Niu, J. Wu, Y. Yang, S. Shen, Y. Guo, J. Liang, W. Guo, Z. Dong. A novel long noncoding RNA, LOC440173, promotes the progression of esophageal squamous cell carcinoma by modulating the miR-30d-5p/HDAC9 axis and the epithelial-mesenchymal transition. Mol. Carcinog., 2020. [DOI | PubMed]
- L. Liang, Z. Yang, Q. Deng, Y. Jiang, Y. Cheng, Y. Sun, L. Li. miR-30d-5p suppresses proliferation and autophagy by targeting ATG5 in renal cell carcinoma. FEBS Open Bio, 2021. [DOI]
- H. Zheng, B. Ding, K. Xue, J. Yu, W. Lou. Construction of a lncRNA/pseudogene-hsa-miR-30d-5p-GJA1 regulatory network related to metastasis of pancreatic cancer. Genomics, 2021. [DOI | PubMed]
- J. Zhang, Y. Xu, H. Liu, Z. Pan. MicroRNAs in ovarian follicular atresia and granulosa cell apoptosis. Reprod. Biol. Endocrinol., 2019. [DOI | PubMed]
- C. Hu, Z. Kang, L. Guo, F. Qu, R. Qu. The Role of LINC00284 in the Development of Thyroid Cancer via Its Regulation of the MicroRNA-30d-5p-Mediated ADAM12/Notch Axis. Front. Oncol., 2021. [DOI | PubMed]
- Y. Qi, Y. Hou, L. Qi. miR-30d-5p represses the proliferation, migration, and invasion of lung squamous cell carcinoma via targeting DBF4. J. Environ. Sci. Health C Toxicol. Carcinog., 2021. [DOI | PubMed]
- C. Li, Y. Yin, X. Liu, X. Xi, W. Xue, Y. Qu. Non-small cell lung cancer associated microRNA expression signature: Integrated bioinformatics analysis, validation and clinical significance. Oncotarget, 2017. [DOI | PubMed]
- S.M. Hosseini, B.M. Soltani, M. Tavallaei, S.J. Mowla, E. Tafsiri, A. Bagheri, H.R.K. Khorshid. Clinically Significant Dysregulation of hsa-miR-30d-5p and hsa-let-7b Expression in Patients with Surgically Resected Non-Small Cell Lung Cancer. Avicenna J. Med. Biotechnol., 2018. [PubMed]
- D. Chen, W. Guo, Z. Qiu, Q. Wang, Y. Li, L. Liang, L. Liu, S. Huang, Y. Zhao, X. He. MicroRNA-30d-5p inhibits tumour cell proliferation and motility by directly targeting CCNE2 in non-small cell lung cancer. Cancer Lett., 2015. [DOI | PubMed]
- B. Krist, U. Florczyk, K. Pietraszek-Gremplewicz, A. Jozkowicz, J. Dulak. The Role of miR-378a in Metabolism, Angiogenesis, and Muscle Biology. Int. J. Endocrinol., 2015. [DOI | PubMed]
- X.B. Guo, X.C. Zhang, P. Chen, L.M. Ma, Z.Q. Shen. miR378a3p inhibits cellular proliferation and migration in glioblastoma multiforme by targeting tetraspanin 17. Oncol. Rep., 2019. [DOI | PubMed]
- B. Zhu, J.J. Chen, Y. Feng, J.L. Yang, H. Huang, W.Y. Chung, Y.L. Hu, W.J. Xue. DNMT1-induced miR-378a-3p silencing promotes angiogenesis via the NF-kappaB signaling pathway by targeting TRAF1 in hepatocellular carcinoma. J. Exp. Clin. Cancer Res., 2021. [DOI | PubMed]
- X. Chu, W. Zheng, J. Wang, J. Zhang, Y. Pan, C. Shao. CDK6 inhibition targeted by miR-378a-3p protects against intestinal injury induced by ionizing radiation. Biochem. Biophys. Res. Commun., 2020. [DOI | PubMed]
- W. Soonthornchai, P. Tangtanatakul, K. Meesilpavikkai, V. Dalm, P. Kueanjinda, J. Wongpiyabovorn. MicroRNA-378a-3p is overexpressed in psoriasis and modulates cell cycle arrest in keratinocytes via targeting BMP2 gene. Sci. Rep., 2021. [DOI | PubMed]
- Y. Zheng, Z. Yang, C. Jin, C. Chen, N. Wu. hsa-miR-191-5p inhibits replication of human immunodeficiency virus type 1 by downregulating the expression of NUP50. Arch. Virol., 2021. [DOI | PubMed]
- Y. Zhang, L. Zhou, Z. Zhang, F. Ren, L. Chen, Z. Lan. miR10a5p inhibits osteogenic differentiation of bone marrowderived mesenchymal stem cells. Mol. Med. Rep., 2020. [DOI | PubMed]
- H. Vaher, T. Runnel, E. Urgard, A. Aab, G. Carreras Badosa, J. Maslovskaja, K. Abram, L. Raam, B. Kaldvee, T. Annilo. miR-10a-5p is increased in atopic dermatitis and has capacity to inhibit keratinocyte proliferation. Allergy, 2019. [DOI | PubMed]
- S. Bin, L. Xin, Z. Lin, Z. Jinhua, G. Rui, Z. Xiang. Targeting miR-10a-5p/IL-6R axis for reducing IL-6-induced cartilage cell ferroptosis. Exp. Mol. Pathol., 2021. [DOI | PubMed]
- Q. Zhang, J. Su, W. Kong, Z. Fang, Y. Li, Z. Huang, J. Wen, Y. Wang. Roles of miR-10a-5p and miR-103a-3p, Regulators of BDNF Expression in Follicular Fluid, in the Outcomes of IVF-ET. Front. Endocrinol., 2021. [DOI]
- T. Arai, A. Okato, S. Kojima, T. Idichi, K. Koshizuka, A. Kurozumi, M. Kato, K. Yamazaki, Y. Ishida, Y. Naya. Regulation of spindle and kinetochore-associated protein 1 by antitumor miR-10a-5p in renal cell carcinoma. Cancer Sci., 2017. [DOI | PubMed]
- S. Zhang, T. Al-Maghout, H. Cao, L. Pelzl, M.S. Salker, M. Veldhoen, A. Cheng, F. Lang, Y. Singh. Gut Bacterial Metabolite Urolithin A (UA) Mitigates Ca2+ Entry in T Cells by Regulating miR-10a-5p. Front. Immunol., 2019. [DOI | PubMed]
- J. Zhu, S. Du, J. Zhang, G. Huang, L. Dong, E. Ren, D. Liu. microRNA-10a-5p from gastric cancer cell-derived exosomes enhances viability and migration of human umbilical vein endothelial cells by targeting zinc finger MYND-type containing 11. Bioengineered, 2022. [DOI | PubMed]
- T.W. Liu, F. Liu, J. Kang. Let-7b-5p is involved in the response of endoplasmic reticulum stress in acute pulmonary embolism through upregulating the expression of stress-associated endoplasmic reticulum protein 1. IUBMB Life, 2020. [DOI | PubMed]
- S. Aday, I. Hazan-Halevy, A. Chamorro-Jorganes, M. Anwar, M. Goldsmith, N. Beazley-Long, S. Sahoo, N. Dogra, W. Sweaad, F. Catapano. Bioinspired artificial exosomes based on lipid nanoparticles carrying let-7b-5p promote angiogenesis in vitro and in vivo. Mol. Ther., 2021. [DOI | PubMed]
- D.S. Wu, C.F. Zheng, Y.P. Zeng, J. Lin, J.L. Chen, S.J. Shi. [Hsa-let-7b-5p Inhibits Proliferation of Human Leukemia THP-1 Cells via FTO/m(6)A/MYC Signaling Pathway]. Zhongguo Shi Yan Xue Ye Xue Za Zhi, 2020. [DOI | PubMed]
- N. Shakespear, M. Ogura, J. Yamaki, Y. Homma. Astrocyte-Derived Exosomal microRNA miR-200a-3p Prevents MPP(+)-Induced Apoptotic Cell Death Through Down-Regulation of MKK4. Neurochem. Res., 2020. [DOI | PubMed]
- R. Wang, H. Bao, S. Zhang, R. Li, L. Chen, Y. Zhu. miR-186-5p Promotes Apoptosis by Targeting IGF-1 in SH-SY5Y OGD/R Model. Int. J. Biol. Sci., 2018. [DOI | PubMed]
- Z. Bayat, Z. Ghaemi, M. Behmanesh, B.M. Soltani. Hsa-miR-186-5p regulates TGFbeta signaling pathway through expression suppression of SMAD6 and SMAD7 genes in colorectal cancer. Biol. Chem., 2021. [DOI | PubMed]
- W. Zhang, H. Yang, Z. Wang, Y. Wu, J. Wang, G. Duan, Q. Guo, Y. Zhang. miR-320a/SP1 negative reciprocal interaction contributes to cell growth and invasion in colorectal cancer. Cancer Cell Int., 2021. [DOI | PubMed]
- H. Zhao, T. Dong, H. Zhou, L. Wang, A. Huang, B. Feng, Y. Quan, R. Jin, W. Zhang, J. Sun. miR-320a suppresses colorectal cancer progression by targeting Rac1. Carcinogenesis, 2014. [DOI | PubMed]
- T. Guo, Y. Feng, Q. Liu, X. Yang, T. Jiang, Y. Chen, Q. Zhang. MicroRNA-320a suppresses in GBM patients and modulates glioma cell functions by targeting IGF-1R. Tumor Biol., 2014. [DOI]
- Y. Wang, J. Zeng, J. Pan, X. Geng, L. Li, J. Wu, P. Song, Y. Wang, J. Liu, L. Wang. MiR-320a inhibits gastric carcinoma by targeting activity in the FoxM1-P27KIP1 axis. Oncotarget, 2016. [DOI | PubMed]
- P. Boldt, B. Knechtle, P. Nikolaidis, C. Lechleitner, G. Wirnitzer, C. Leitzmann, T. Rosemann, K. Wirnitzer. Quality of life of female and male vegetarian and vegan endurance runners compared to omnivores-results from the NURMI study (step 2). J. Int. Soc. Sports Nutr., 2018. [DOI | PubMed]
- Z. Xishan, L. Ziying, D. Jing, L. Gang. MicroRNA-320a acts as a tumor suppressor by targeting BCR/ABL oncogene in chronic myeloid leukemia. Sci. Rep., 2015. [DOI | PubMed]
- C. Pisanu, E. Merkouri Papadima, C. Melis, D. Congiu, A. Loizedda, N. Orru, S. Calza, S. Orru, C. Carcassi, G. Severino. Whole Genome Expression Analyses of miRNAs and mRNAs Suggest the Involvement of miR-320a and miR-155-3p and their Targeted Genes in Lithium Response in Bipolar Disorder. Int. J. Mol. Sci., 2019. [DOI]
- Y. Li, J. Huang, C. Hu, J. Zhou, D. Xu, Y. Hou, C. Wu, J. Zhao, M. Li, X. Zeng. MicroRNA-320a: An important regulator in the fibrotic process in interstitial lung disease of systemic sclerosis. Arthritis Res., 2021. [DOI]
- Y. Lu, D. Wu, J. Wang, Y. Li, X. Chai, Q. Kang. miR-320a regulates cell proliferation and apoptosis in multiple myeloma by targeting pre-B-cell leukemia transcription factor 3. Biochem. Biophys. Res. Commun., 2016. [DOI | PubMed]
- S.P. Mittal, J. Mathai, A.P. Kulkarni, J.K. Pal, S. Chattopadhyay. miR-320a regulates erythroid differentiation through MAR binding protein SMAR1. Int. J. Biochem. Cell Biol., 2013. [DOI | PubMed]
- S. Jo, G. Xu, G. Jing, J. Chen, A. Shalev. Human Glucagon Expression Is under the Control of miR-320a. Endocrinology, 2021. [DOI | PubMed]
- R.P. Berkhout, R. Keijser, S. Repping, C.B. Lambalk, G.B. Afink, S. Mastenbroek, G. Hamer. High-quality human preimplantation embryos stimulate endometrial stromal cell migration via secretion of microRNA hsa-miR-320a. Hum. Reprod., 2020. [DOI | PubMed]
- D. Dahlmans, A. Houzelle, P. Andreux, J.A. Jorgensen, X. Wang, L.J. de Windt, P. Schrauwen, J. Auwerx, J. Hoeks. An unbiased silencing screen in muscle cells identifies miR-320a, miR-150, miR-196b, and miR-34c as regulators of skeletal muscle mitochondrial metabolism. Mol. Metab., 2017. [DOI | PubMed]
- F.J. Li, C.L. Zhang, X.J. Luo, J. Peng, T.L. Yang. Involvement of the MiR-181b-5p/HMGB1 Pathway in Ang II-induced Phenotypic Transformation of Smooth Muscle Cells in Hypertension. Aging Dis., 2019. [DOI | PubMed]
- M. Di Marco, S. Veschi, P. Lanuti, A. Ramassone, S. Pacillo, S. Pagotto, F. Pepe, J.N. George-William, C. Curcio, M. Marchisio. Enhanced Expression of miR-181b in B Cells of CLL Improves the Anti-Tumor Cytotoxic T Cell Response. Cancers, 2021. [DOI]
- J. Miao, Y. Zhu, L. Xu, X. Huang, X. Zhou. miR181b5p inhibits trophoblast cell migration and invasion through targeting S1PR1 in multiple abnormal trophoblast invasionrelated events. Mol. Med. Rep., 2020. [DOI | PubMed]
- A. Graham, J. Holbert, W.B. Nothnick. miR-181b-5p Modulates Cell Migratory Proteins, Tissue Inhibitor of Metalloproteinase 3, and Annexin A2 During In Vitro Decidualization in a Human Endometrial Stromal Cell Line. Reprod. Sci., 2017. [DOI | PubMed]
- L. Gong, P. Yang, L. Hu, C. Zhang. MiR-181b suppresses the progression of epilepsy by regulation of lncRNA ZNF883. Am. J. Transl. Res., 2020. [PubMed]
- K. Wu, J. Huang, T. Xu, Z. Ye, F. Jin, N. Li, B. Lv. MicroRNA-181b blocks gensenoside Rg3-mediated tumor suppression of gallbladder carcinoma by promoting autophagy flux via CREBRF/CREB3 pathway. Am. J. Transl. Res., 2019. [PubMed]
- C. Dieter, T.S. Assmann, A.R. Costa, L.H. Canani, B.M. de Souza, A.C. Bauer, D. Crispim. MiR-30e-5p and MiR-15a-5p Expressions in Plasma and Urine of Type 1 Diabetic Patients with Diabetic Kidney Disease. Front. Genet., 2019. [DOI | PubMed]
- X. Liu, Q. Li, L. Sun, L. Chen, Y. Li, B. Huang, Y. Liu, C. Jiang. miR-30e-5p Regulates Autophagy and Apoptosis by Targeting Beclin1 Involved in Contrast-induced Acute Kidney Injury. Curr. Med. Chem., 2021. [DOI | PubMed]
- G. Xu, J. Cai, L. Wang, L. Jiang, J. Huang, R. Hu, F. Ding. MicroRNA-30e-5p suppresses non-small cell lung cancer tumorigenesis by regulating USP22-mediated Sirt1/JAK/STAT3 signaling. Exp. Cell Res., 2018. [DOI | PubMed]
- S. Zhang, G. Li, C. Liu, S. Lu, Q. Jing, X. Chen, H. Zheng, H. Ma, D. Zhang, S. Ren. miR-30e-5p represses angiogenesis and metastasis by directly targeting AEG-1 in squamous cell carcinoma of the head and neck. Cancer Sci., 2020. [DOI | PubMed]
- R. Mishra, S. Bhattacharya, B.S. Rawat, A. Kumar, A. Kumar, K. Niraj, A. Chande, P. Gandhi, D. Khetan, A. Aggarwal. MicroRNA-30e-5p has an Integrated Role in the Regulation of the Innate Immune Response during Virus Infection and Systemic Lupus Erythematosus. iScience, 2020. [DOI | PubMed]
- S. Laudato, N. Patil, M.L. Abba, J.H. Leupold, A. Benner, T. Gaiser, A. Marx, H. Allgayer. P53-induced miR-30e-5p inhibits colorectal cancer invasion and metastasis by targeting ITGA6 and ITGB1. Int. J. Cancer, 2017. [DOI | PubMed]
- J. Ge, L. Mao, W. Xu, W. Fang, N. Wang, D. Ye, Z. Dong, H. Guan, C. Guan. miR-103a-3p Suppresses Cell Proliferation and Invasion by Targeting Tumor Protein D52 in Prostate Cancer. J. Investig. Surg., 2021. [DOI | PubMed]
- J. Zhang, Q. Lu, H. Pang, M. Zhang, W. Wei. MiR-103a-3p aggravates renal cell carcinoma by targeting TMEM33. Am. J. Transl. Res., 2021. [PubMed]
- A. Fasihi, B. MSoltani, A. Atashi, S. Nasiri. Introduction of hsa-miR-103a and hsa-miR-1827 and hsa-miR-137 as new regulators of Wnt signaling pathway and their relation to colorectal carcinoma. J. Cell. Biochem., 2018. [DOI | PubMed]
- P. Moskwa, F.M. Buffa, Y. Pan, R. Panchakshari, P. Gottipati, R.J. Muschel, J. Beech, R. Kulshrestha, K. Abdelmohsen, D.M. Weinstock. miR-182-mediated downregulation of BRCA1 impacts DNA repair and sensitivity to PARP inhibitors. Mol. Cell, 2011. [DOI | PubMed]
- Y. Jin, Z.L. Zhang, Y. Huang, K.N. Zhang, B. Xiong. MiR-182-5p inhibited proliferation and metastasis of colorectal cancer by targeting MTDH. Eur. Rev. Med. Pharmacol. Sci., 2019. [DOI | PubMed]
- X. Xu, J. Wu, S. Li, Z. Hu, X. Xu, Y. Zhu, Z. Liang, X. Wang, Y. Lin, Y. Mao. Downregulation of microRNA-182-5p contributes to renal cell carcinoma proliferation via activating the AKT/FOXO3a signaling pathway. Mol. Cancer, 2014. [DOI | PubMed]
- F. Wang, D. Wu, Z. Xu, J. Chen, J. Zhang, X. Li, S. Chen, F. He, J. Xu, L. Su. miR-182-5p affects human bladder cancer cell proliferation, migration and invasion through regulating Cofilin 1. Cancer Cell Int., 2019. [DOI | PubMed]
- M.F. Souza, I.M.S. Colus, A.S. Fonseca, V.C. Antunes, D. Kumar, L.R. Cavalli. MiR-182-5p Modulates Prostate Cancer Aggressive Phenotypes by Targeting EMT Associated Pathways. Biomolecules, 2022. [DOI | PubMed]
- A.E. Roser, L. Caldi Gomes, R. Halder, G. Jain, F. Maass, L. Tonges, L. Tatenhorst, M. Bahr, A. Fischer, P. Lingor. miR-182-5p and miR-183-5p Act as GDNF Mimics in Dopaminergic Midbrain Neurons. Mol. Nucleic Acids, 2018. [DOI | PubMed]
- P. Kulkarni, P. Dasgupta, N.S. Bhat, V. Shahryari, M. Shiina, Y. Hashimoto, S. Majid, G. Deng, S. Saini, Z.L. Tabatabai. Elevated miR-182-5p Associates with Renal Cancer Cell Mitotic Arrest through Diminished MALAT-1 Expression. Mol. Cancer Res., 2018. [DOI | PubMed]
- D. Jury, I. Daugaard, K.J. Sanders, L.L. Hansen, D. Agalliu, I.M. Pedersen. miR-151a enhances Slug dependent angiogenesis. Oncotarget, 2020. [DOI | PubMed]
- P. Tornero-Esteban, L. Rodriguez-Rodriguez, L. Abasolo, M. Tome, P. Lopez-Romero, E. Herranz, M.A. Gonzalez, F. Marco, E. Moro, B. Fernandez-Gutierrez. Signature of microRNA expression during osteogenic differentiation of bone marrow MSCs reveals a putative role of miR-335-5p in osteoarthritis. BMC Musculoskelet. Disord., 2015. [DOI | PubMed]
- A. Sandoval-Borquez, I. Polakovicova, N. Carrasco-Veliz, L. Lobos-Gonzalez, I. Riquelme, G. Carrasco-Avino, C. Bizama, E. Norero, G.I. Owen, J.C. Roa. MicroRNA-335-5p is a potential suppressor of metastasis and invasion in gastric cancer. Clin. Epigenet., 2017. [DOI | PubMed]
- W. Zhao, Y. Zhao, L. Chen, Y. Sun, S. Fan. miR-335-5p Inhibits Progression of Uterine Leiomyoma by Targeting ARGLU1. Comput. Math. Methods Med., 2022. [DOI | PubMed]
- G. Suyal, P. Pandey, A. Saraya, R. Sharma. Tumour suppressor role of microRNA-335-5p in esophageal squamous cell carcinoma by targeting TTK (Mps1). Exp. Mol. Pathol., 2022. [DOI | PubMed]
- M. Kay, B.M. Soltani, F.H. Aghdaei, H. Ansari, H. Baharvand. Hsa-miR-335 regulates cardiac mesoderm and progenitor cell differentiation. Stem Cell Res., 2019. [DOI]
- Y. Jin, D. Yu, W.H. Tolleson, B. Knox, Y. Wang, S. Chen, Z. Ren, H. Deng, Y. Guo, B. Ning. MicroRNA hsa-miR-25-3p suppresses the expression and drug induction of CYP2B6 in human hepatocytes. Biochem. Pharmacol., 2016. [DOI | PubMed]
- C. Yu, D. Wu, C. Zhao, C. Wu. CircRNA TGFBR2/MiR-25-3p/TWIST1 axis regulates osteoblast differentiation of human aortic valve interstitial cells. J. Bone Miner. Metab., 2021. [DOI | PubMed]
- W.Q. Jia, J.W. Zhu, C.Y. Yang, J. Ma, T.Y. Pu, G.Q. Han, M.M. Zou, R.X. Xu. Verbascoside inhibits progression of glioblastoma cells by promoting Let-7g-5p and down-regulating HMGA2 via Wnt/beta-catenin signalling blockade. J. Cell. Mol. Med., 2020. [DOI | PubMed]
- P. Yang, M. Zhang, X. Wang, A.L. Xu, M. Shen, B. Jiang, X. Zhou, L. Zhou. MicroRNA let-7g-5p alleviates murine collagen-induced arthritis by inhibiting Th17 cell differentiation. Biochem. Pharmacol., 2020. [DOI | PubMed]
- J. Zhang, H. Zhang, Y. Qin, C. Chen, J. Yang, N. Song, M. Gu. MicroRNA-200c-3p/ZEB2 loop plays a crucial role in the tumor progression of prostate carcinoma. Ann. Transl. Med., 2019. [DOI | PubMed]
- N. Maolakuerban, B. Azhati, H. Tusong, A. Abula, A. Yasheng, A. Xireyazidan. MiR-200c-3p inhibits cell migration and invasion of clear cell renal cell carcinoma via regulating SLC6A1. Cancer Biol., 2018. [DOI]
- E. Anastasiadou, E. Messina, T. Sanavia, L. Mundo, F. Farinella, S. Lazzi, F. Megiorni, S. Ceccarelli, P. Pontecorvi, F. Marampon. MiR-200c-3p Contrasts PD-L1 Induction by Combinatorial Therapies and Slows Proliferation of Epithelial Ovarian Cancer through Downregulation of beta-Catenin and c-Myc. Cells, 2021. [DOI | PubMed]
- G. Obeng, E.J. Park, M.G. Appiah, E. Kawamoto, A. Gaowa, M. Shimaoka. miRNA-200c-3p targets talin-1 to regulate integrin-mediated cell adhesion. Sci. Rep., 2021. [DOI | PubMed]
- R. Raue, A.C. Frank, D.C. Fuhrmann, P. de la Cruz-Ojeda, S. Rosser, R. Bauer, G. Cardamone, A. Weigert, S.N. Syed, T. Schmid. MicroRNA-200c Attenuates the Tumor-Infiltrating Capacity of Macrophages. Biology, 2022. [DOI | PubMed]
- J.M. Cao, G.Z. Li, M. Han, H.L. Xu, K.M. Huang. MiR-30c-5p suppresses migration, invasion and epithelial to mesenchymal transition of gastric cancer via targeting MTA1. Biomed. Pharmacol., 2017. [DOI]
- Y. Wang, X. Pei, P. Xu, Z. Tan, Z. Zhu, G. Zhang, Z. Jiang, Z. Deng. E2F7, regulated by miR30c, inhibits apoptosis and promotes cell cycle of prostate cancer cells. Oncol. Rep., 2020. [DOI | PubMed]
- Y.F. Zou, W.T. Liao, Z.J. Fu, Q. Zhao, Y.X. Chen, W. Zhang. MicroRNA-30c-5p ameliorates hypoxia-reoxygenation-induced tubular epithelial cell injury via HIF1alpha stabilization by targeting SOCS3. Oncotarget, 2017. [DOI | PubMed]
- A. Zietzer, M.R. Hosen, H. Wang, P.R. Goody, M. Sylvester, E. Latz, G. Nickenig, N. Werner, F. Jansen. The RNA-binding protein hnRNPU regulates the sorting of microRNA-30c-5p into large extracellular vesicles. J. Extracell. Vesicles, 2020. [DOI | PubMed]
- S. Bartoszewska, K. Kochan, A. Piotrowski, W. Kamysz, R.J. Ochocka, J.F. Collawn, R. Bartoszewski. The hypoxia-inducible miR-429 regulates hypoxia-inducible factor-1alpha expression in human endothelial cells through a negative feedback loop. FASEB J., 2015. [DOI | PubMed]
- L. Zhang, Q. Liu, Q. Mu, D. Zhou, H. Li, B. Zhang, C. Yin. MiR-429 suppresses proliferation and invasion of breast cancer via inhibiting the Wnt/beta-catenin signaling pathway. Thorac. Cancer, 2020. [DOI | PubMed]
- Y. Deng, F. Luan, L. Zeng, Y. Zhang, K. Ma. MiR-429 suppresses the progression and metastasis of osteosarcoma by targeting ZEB1. EXCLI J., 2017. [DOI | PubMed]
- D. Samantarrai, B. Mallick. miR-429 inhibits metastasis by targeting KIAA0101 in Soft Tissue Sarcoma. Exp. Cell Res., 2017. [DOI | PubMed]
- J.Y. Fan, Y.J. Fan, X.L. Wang, H. Xie, H.J. Gao, Y. Zhang, M. Liu, H. Tang. miR-429 is involved in regulation of NF-kappaBactivity by targeting IKKbeta and suppresses oncogenic activity in cervical cancer cells. FEBS Lett., 2017. [DOI | PubMed]
- M. Zhang, B.B. Dong, M. Lu, M.J. Zheng, H. Chen, J.Z. Ding, A.M. Xu, Y.H. Xu. miR-429 functions as a tumor suppressor by targeting FSCN1 in gastric cancer cells. Onco Targets Ther., 2016. [DOI | PubMed]
- Y. Liang, Z.J. Yu, M. Liu, H.M. Liu, J.Z. Zhang, T. Xiong, Y.Y. Tang, Z.P. Huang. hsamiR429 targets CBX8 to promote cell apoptosis in diffuse large Bcell lymphoma. Mol. Med. Rep., 2021. [DOI | PubMed]
- Y. Wang, M. Li, W. Zang, Y. Ma, N. Wang, P. Li, T. Wang, G. Zhao. MiR-429 up-regulation induces apoptosis and suppresses invasion by targeting Bcl-2 and SP-1 in esophageal carcinoma. Cell. Oncol., 2013. [DOI]
- C. Guo, D. Zhao, Q. Zhang, S. Liu, M.Z. Sun. miR-429 suppresses tumor migration and invasion by targeting CRKL in hepatocellular carcinoma via inhibiting Raf/MEK/ERK pathway and epithelial-mesenchymal transition. Sci. Rep., 2018. [DOI | PubMed]
- H. Xue, G.Y. Tian. MiR-429 regulates the metastasis and EMT of HCC cells through targeting RAB23. Arch. Biochem. Biophys., 2018. [DOI | PubMed]
- H. Dong, X. Hao, B. Cui, M. Guo. MiR-429 suppresses glioblastoma multiforme by targeting SOX2. Cell Biochem. Funct., 2017. [DOI | PubMed]
- Z. Wang, Z. Zhu, Z. Lin, Y. Luo, Z. Liang, C. Zhang, J. Chen, P. Peng. miR-429 suppresses cell proliferation, migration and invasion in nasopharyngeal carcinoma by downregulation of TLN1. Cancer Cell Int., 2019. [DOI | PubMed]
- D. Liu, L. Song, Z. Dai, H. Guan, H. Kang, Y. Zhang, W. Yan, X. Zhao, S. Zhang. MiR-429 suppresses neurotrophin-3 to alleviate perineural invasion of pancreatic cancer. Biochem. Biophys. Res. Commun., 2018. [DOI | PubMed]
- G. Wu, H. Zheng, J. Xu, Y. Guo, G. Zheng, C. Ma, S. Hao, X. Liu, H. Chen, S. Wei. miR-429 suppresses cell growth and induces apoptosis of human thyroid cancer cell by targeting ZEB1. Artif. Cells Nanomed. Biotechnol., 2019. [DOI | PubMed]
- W. Lei, Y.E. Liu, Y. Zheng, L. Qu. MiR-429 inhibits oral squamous cell carcinoma growth by targeting ZEB1. Med. Sci. Monit., 2015. [DOI | PubMed]
- M. Qiu, Z. Liang, L. Chen, G. Tan, K. Wang, L. Liu, J. Liu, H. Chen. MicroRNA-429 suppresses cell proliferation, epithelial-mesenchymal transition, and metastasis by direct targeting of BMI1 and E2F3 in renal cell carcinoma. Urol. Oncol., 2015. [DOI]
- X. Zhang, X. Yu, Z. Zhao, Z. Yuan, P. Ma, Z. Ye, L. Guo, S. Xu, L. Xu, T. Liu. MicroRNA-429 inhibits bone metastasis in breast cancer by regulating CrkL and MMP-9. Bone, 2020. [DOI | PubMed]
- A. Janaszak-Jasiecka, S. Bartoszewska, K. Kochan, A. Piotrowski, L. Kalinowski, W. Kamysz, R.J. Ochocka, R. Bartoszewski, J.F. Collawn. miR-429 regulates the transition between Hypoxia-Inducible Factor (HIF)1A and HIF3A expression in human endothelial cells. Sci. Rep., 2016. [DOI | PubMed]
- E. Zacharewicz, M. Kalanon, R.M. Murphy, A.P. Russell, S. Lamon. MicroRNA-99b-5p downregulates protein synthesis in human primary myotubes. Am. J. Physiol. Cell Physiol., 2020. [DOI | PubMed]
- Y.J. Li, Y. Wang, Y.Y. Wang. MicroRNA-99b suppresses human cervical cancer cell activity by inhibiting the PI3K/AKT/mTOR signaling pathway. J. Cell. Physiol., 2019. [DOI | PubMed]
- D. Yu, B. Green, W.H. Tolleson, Y. Jin, N. Mei, Y. Guo, H. Deng, I. Pogribny, B. Ning. MicroRNA hsa-miR-29a-3p modulates CYP2C19 in human liver cells. Biochem. Pharmacol., 2015. [DOI | PubMed]
- J.H. Ma, X. Bu, J.J. Wang, Y.X. Xie. MicroRNA-29-3p Regulates Hepatocellular Carcinoma Progression Through NF-kappaB Pathway. Clin. Lab., 2019. [DOI | PubMed]
- Y.H. Huang, Y.L. Yang, F.S. Wang. The Role of miR-29a in the Regulation, Function, and Signaling of Liver Fibrosis. Int. J. Mol. Sci., 2018. [DOI | PubMed]
- Y.L. Yang, Y.H. Chang, C.J. Li, Y.H. Huang, M.C. Tsai, P.Y. Chu, H.Y. Lin. New Insights into the Role of miR-29a in Hepatocellular Carcinoma: Implications in Mechanisms and Theragnostics. J. Pers. Med., 2021. [DOI | PubMed]
- Z. Zhao, L. Wang, W. Song, H. Cui, G. Chen, F. Qiao, J. Hu, R. Zhou, H. Fan. Reduced miR-29a-3p expression is linked to the cell proliferation and cell migration in gastric cancer. World J. Surg. Oncol., 2015. [DOI | PubMed]
- H. He, N. Wang, X. Yi, C. Tang, D. Wang. Long non-coding RNA H19 regulates E2F1 expression by competitively sponging endogenous miR-29a-3p in clear cell renal cell carcinoma. Cell Biosci., 2017. [DOI | PubMed]
- X. Deng, X. Chu, P. Wang, X. Ma, C. Wei, C. Sun, J. Yang, Y. Li. MicroRNA-29a-3p Reduces TNFalpha-Induced Endothelial Dysfunction by Targeting Tumor Necrosis Factor Receptor 1. Mol. Nucleic Acids, 2019. [DOI | PubMed]
- X. Chen, X. Li, C. Wei, C. Zhao, S. Wang, J. Gao. High expression of SETDB1 mediated by miR-29a-3p associates with poor prognosis and immune invasion in breast invasive carcinoma. Transl. Cancer Res., 2021. [DOI | PubMed]
- R. Zhou, S. Ren, C. Li, X. Zhang, W. Zhang. miR-29a is a potential protective factor for fibrogenesis in gluteal muscle contracture. Physiol. Res., 2020. [DOI | PubMed]
- F. Liu, Z.F. Wang, F.F. Liu, J.Z. Xu, Q. Liu, J. Lan. MicroRNA-29a-3p regulates osteoblast differentiation and peri-implant osseointegration in a rat model of hyperlipidemia by modulating Frizzled 4 expression. Hua Xi Kou Qiang Yi Xue Za Zhi, 2019. [DOI | PubMed]
- B. Lv, Z. Liu, S. Wang, F. Liu, X. Yang, J. Hou, Z. Hou, B. Chen. MiR-29a promotes intestinal epithelial apoptosis in ulcerative colitis by down-regulating Mcl-1. Int. J. Clin. Exp. Pathol., 2014. [PubMed]
- M.E. Widlansky, D.M. Jensen, J. Wang, Y. Liu, A.M. Geurts, A.J. Kriegel, P. Liu, R. Ying, G. Zhang, M. Casati. miR-29 contributes to normal endothelial function and can restore it in cardiometabolic disorders. EMBO Mol. Med., 2018. [DOI | PubMed]
- C. Glantschnig, M. Koenen, M. Gil-Lozano, M. Karbiener, I. Pickrahn, J. Williams-Dautovich, R. Patel, C.L. Cummins, M. Giroud, G. Hartleben. A miR-29a-driven negative feedback loop regulates peripheral glucocorticoid receptor signaling. FASEB J., 2019. [DOI | PubMed]
- P.Y. He, W.K. Yip, B.L. Chai, B.Y. Chai, M.F. Jabar, N. Dusa, N. Mohtarrudin, H.F. Seow. Inhibition of cell migration and invasion by miR29a3p in a colorectal cancer cell line through suppression of CDC42BPA mRNA expression. Oncol. Rep., 2017. [DOI | PubMed]
- Y. Liu, N. Duan, S. Duan. MiR-29a Inhibits Glioma Tumorigenesis through a Negative Feedback Loop of TRAF4/Akt Signaling. Biomed Res. Int., 2018. [DOI | PubMed]
- I. Fukumoto, N. Kikkawa, R. Matsushita, M. Kato, A. Kurozumi, R. Nishikawa, Y. Goto, K. Koshizuka, T. Hanazawa, H. Enokida. Tumor-suppressive microRNAs (miR-26a/b, miR-29a/b/c and miR-218) concertedly suppressed metastasis-promoting LOXL2 in head and neck squamous cell carcinoma. J. Hum. Genet., 2016. [DOI | PubMed]
- J. Li, X. Wan, W. Qiang, T. Li, W. Huang, S. Huang, D. Wu, Y. Li. MiR-29a suppresses prostate cell proliferation and induces apoptosis via KDM5B protein regulation. Int. J. Clin. Exp. Med., 2015. [PubMed]
- Y. Ma, Y. Sun. miR-29a-3p inhibits growth, proliferation, and invasion of papillary thyroid carcinoma by suppressing NF-kappaB signaling via direct targeting of OTUB2. Cancer Manag. Res., 2019. [DOI | PubMed]
- Y. Yan, C. Du, X. Duan, X. Yao, J. Wan, Z. Jiang, Z. Qin, W. Li, L. Pan, Z. Gu. Inhibiting collagen I production and tumor cell colonization in the lung via miR-29a-3p loading of exosome-/liposome-based nanovesicles. Acta Pharm. Sin. B, 2022. [DOI | PubMed]
- Y. Chen, W. Zhang, L. Yan, P. Zheng, J. Li. miR-29a-3p directly targets Smad nuclear interacting protein 1 and inhibits the migration and proliferation of cervical cancer HeLa cells. PeerJ, 2020. [DOI | PubMed]
- A. Geng, L. Luo, F. Ren, L. Zhang, H. Zhou, X. Gao. miR-29a-3p inhibits endometrial cancer cell proliferation, migration and invasion by targeting VEGFA/CD C42/PAK1. BMC Cancer, 2021. [DOI | PubMed]
- C. Jiang, F. Liu, S. Xiao, L. He, W. Wu, Q. Zhao. miR-29a-3p enhances the radiosensitivity of oral squamous cell carcinoma cells by inhibiting ADAM12. Eur. J. Histochem., 2021. [DOI | PubMed]
- D. Yu, W.H. Tolleson, B. Knox, Y. Jin, L. Guo, Y. Guo, S.A. Kadlubar, B. Ning. Modulation of ALDH5A1 and SLC22A7 by microRNA hsa-miR-29a-3p in human liver cells. Biochem. Pharmacol., 2015. [DOI | PubMed]
- Z. Liu, X. Wei, A. Zhang, C. Li, J. Bai, J. Dong. Long non-coding RNA HNF1A-AS1 functioned as an oncogene and autophagy promoter in hepatocellular carcinoma through sponging hsa-miR-30b-5p. Biochem. Biophys. Res. Commun., 2016. [DOI | PubMed]
- Q. Zhang, X.F. Ma, M.Z. Dong, J. Tan, J. Zhang, L.K. Zhuang, S.S. Liu, Y.N. Xin. MiR-30b-5p regulates the lipid metabolism by targeting PPARGC1A in Huh-7 cell line. Lipids Health Dis., 2020. [DOI | PubMed]
- C.C. Woo, W. Liu, X.Y. Lin, R. Dorajoo, K.W. Lee, A.M. Richards, C.N. Lee, T. Wongsurawat, I. Nookaew, V. Sorokin. The interaction between 30b-5p miRNA and MBNL1 mRNA is involved in vascular smooth muscle cell differentiation in patients with coronary atherosclerosis. Int. J. Mol. Sci., 2019. [DOI]
- F. Li, Q. Chen, X. Song, L. Zhou, J. Zhang. MiR-30b Is Involved in the Homocysteine-Induced Apoptosis in Human Coronary Artery Endothelial Cells by Regulating the Expression of Caspase 3. Int. J. Mol. Sci., 2015. [DOI | PubMed]
- Y. Luo, F. Zhou, X. Wu, Y. Li, B. Ye. miR-30b-5p inhibits osteoblast differentiation through targeting BCL6. Cell Cycle, 2022. [DOI | PubMed]
- C. Xu, Z. He, C. Lin, Z. Shen. MiR-30b-5p inhibits proliferation and promotes apoptosis of medulloblastoma cells via targeting MYB proto-oncogene like 2 (MYBL2). J. Investig. Med., 2020. [DOI]
- G. Park, B. Son, J. Kang, S. Lee, J. Jeon, J.H. Kim, G.R. Yi, H. Youn, C. Moon, S.Y. Nam. LDR-Induced miR-30a and miR-30b Target the PAI-1 Pathway to Control Adverse Effects of NSCLC Radiotherapy. Mol. Ther., 2019. [DOI | PubMed]
- H. Zhang, Y. He, J.X. Wang, M.H. Chen, J.J. Xu, M.H. Jiang, Y.L. Feng, Y.F. Gu. miR-30-5p-mediated ferroptosis of trophoblasts is implicated in the pathogenesis of preeclampsia. Redox Biol., 2020. [DOI | PubMed]
- S.F. Wang-Renault, S. Boudaoud, G. Nocturne, E. Roche, N. Sigrist, C. Daviaud, A. Bugge Tinggaard, V. Renault, J.F. Deleuze, X. Mariette. Deregulation of microRNA expression in purified T and B lymphocytes from patients with primary Sjogren’s syndrome. Ann. Rheum. Dis., 2018. [DOI | PubMed]
- A.I. Osip’yants, E.N. Knyazev, A.V. Galatenko, K.M. Nyushko, V.V. Galatenko, M.Y. Shkurnikov, B.Y. Alekseev. Changes in the Level of Circulating hsa-miR-297 and hsa-miR-19b-3p miRNA Are Associated with Generalization of Prostate Cancer. Bull. Exp. Biol. Med., 2017. [DOI | PubMed]
- Y. Ma, S. Tian, S. He, Q. Chen, Z. Wang, X. Xiao, L. Fu, X. Lei. The mechanism of action of FXR1P-related miR-19b-3p in SH-SY5Y. Gene, 2016. [DOI | PubMed]
- L. Ye, L.R. Morse, S.P. Falci, J.K. Olson, M. Shrivastava, N. Nguyen, C. Linnman, K.L. Troy, R.A. Battaglino. hsa-MiR-19a-3p and hsa-MiR-19b-3p Are Associated with Spinal Cord Injury-Induced Neuropathic Pain: Findings from a Genome-Wide MicroRNA Expression Profiling Screen. Neurotrauma Rep., 2021. [DOI | PubMed]
- L.E. Mullany, J.S. Herrick, L.C. Sakoda, W. Samowitz, J.R. Stevens, R.K. Wolff, M.L. Slattery. miRNA involvement in cell cycle regulation in colorectal cancer cases. Genes Cancer, 2018. [DOI | PubMed]
- F. Wu, J.Y. Wang, W. Chao, C. Sims, R.A. Kozar. miR-19b targets pulmonary endothelial syndecan-1 following hemorrhagic shock. Sci. Rep., 2020. [DOI | PubMed]
- D.A. Rivas, F. Peng, T. Benard, A.S. Ramos da Silva, R.A. Fielding, L.M. Margolis. miR-19b-3p is associated with a diametric response to resistance exercise in older adults and regulates skeletal muscle anabolism via PTEN inhibition. Am. J. Physiol. Cell Physiol., 2021. [DOI | PubMed]
- C. Guo, H. Pengli, C. Keru, L. Mingchuang, Z. Qingsong, C. Zheng, D. Hanhua, Q. Yuejun, L. Jing. Molecular mechanism of miR-19b-3p regulating apoptosis of medullary thyroid cancer cells by targeting MTUS1. China Oncol., 2021. [DOI]
- L. Cui, W. Du, N. Xu, J. Dong, B. Xia, J. Ma, R. Yan, L. Wang, F. Feng. Impact of MicroRNAs in Interaction with Environmental Factors on Autism Spectrum Disorder: An Exploratory Pilot Study. Front. Psychiatry, 2021. [DOI | PubMed]
- K. Li, X. Ya, X. Duan, Y. Li, X. Lin. miRNA-19b-3p Stimulates Cardiomyocyte Apoptosis Induced by Myocardial Ischemia Reperfusion via Downregulating PTEN. Dis. Markers, 2021. [DOI | PubMed]
- F. Hassiotou, A.R. Hepworth, T.M. Williams, A.-J. Twigger, S. Perrella, C.T. Lai, L. Filgueira, D.T. Geddes, P.E. Hartmann. Breastmilk cell and fat contents respond similarly to removal of breastmilk by the infant. PLoS ONE, 2013. [DOI | PubMed]
- S. Khan, A.R. Hepworth, D.K. Prime, C.T. Lai, N.J. Trengove, P.E. Hartmann. Variation in fat, lactose, and protein composition in breast milk over 24 hours: Associations with infant feeding patterns. J. Hum. Lact., 2013. [DOI | PubMed]
- F. Hassiotou, D.T. Geddes, P.E. Hartmann. Cells in human milk: State of the science. J. Hum. Lact., 2013. [DOI | PubMed]
- M. Jiang, X. Sang, Z. Hong. Beyond nutrients: Food-derived microRNAs provide cross-kingdom regulation. Bioessays, 2012. [DOI | PubMed]
- J.D. Arroyo, J.R. Chevillet, E.M. Kroh, I.K. Ruf, C.C. Pritchard, D.F. Gibson, P.S. Mitchell, C.F. Bennett, E.L. Pogosova-Agadjanyan, D.L. Stirewalt. Argonaute2 complexes carry a population of circulating microRNAs independent of vesicles in human plasma. Proc. Natl. Acad. Sci. USA, 2011. [DOI | PubMed]
- L. Ferraro, M. Ravo, G. Nassa, R. Tarallo, M.R. De Filippo, G. Giurato, F. Cirillo, C. Stellato, S. Silvestro, C. Cantarella. Effects of oestrogen on microRNA expression in hormone-responsive breast cancer cells. Horm. Cancer, 2012. [DOI | PubMed]
- D.R. Cochrane, N.S. Spoelstra, J.K. Richer. The role of miRNAs in progesterone action. Mol. Cell. Endocrinol., 2012. [DOI | PubMed]
- J. Zhang, F. Zhang, X. Didelot, K.D. Bruce, F.R. Cagampang, M. Vatish, M. Hanson, H. Lehnert, A. Ceriello, C.D. Byrne. Maternal high fat diet during pregnancy and lactation alters hepatic expression of insulin like growth factor-2 and key microRNAs in the adult offspring. BMC Genom., 2009. [DOI]
- S.R. Dall, I.L. Boyd. Evolution of mammals: Lactation helps mothers to cope with unreliable food supplies. Proc. R. Soc. Lond. Ser. B Biol. Sci., 2004. [DOI]
- K.M. Aagaard-Tillery, K. Grove, J. Bishop, X. Ke, Q. Fu, R. McKnight, R.H. Lane. Developmental origins of disease and determinants of chromatin structure: Maternal diet modifies the primate fetal epigenome. J. Mol. Endocrinol., 2008. [DOI | PubMed]
- M. Suter, P. Bocock, L. Showalter, M. Hu, C. Shope, R. McKnight, K. Grove, R. Lane, K. Aagaard-Tillery. Epigenomics: Maternal high-fat diet exposure in utero disrupts peripheral circadian gene expression in nonhuman primates. FASEB J., 2011. [DOI | PubMed]
- T. Saarela, J. Kokkonen, M. Koivisto. Macronutrient and energy contents of human milk fractions during the first six months of lactation. Acta Paediatr., 2005. [DOI | PubMed]
- Y. Huang, X.J. Shen, Q. Zou, S.P. Wang, S.M. Tang, G.Z. Zhang. Biological functions of microRNAs: A review. J. Physiol. Biochem., 2011. [DOI | PubMed]
- L.F. Lu, A. Liston. MicroRNA in the immune system, microRNA as an immune system. Immunology, 2009. [DOI | PubMed]
- A.J. Asirvatham, W.J. Magner, T.B. Tomasi. miRNA regulation of cytokine genes. Cytokine, 2009. [DOI | PubMed]
- T.-Y. Ha. The Role of MicroRNAs in Regulatory T Cells and in the Immune Response. Immune Netw., 2011. [DOI | PubMed]
- C. Liu, N. Li, G. Liu. The Role of MicroRNAs in Regulatory T Cells. J. Immunol. Res., 2020. [DOI | PubMed]
- E. Vigorito, K.L. Perks, C. Abreu-Goodger, S. Bunting, Z. Xiang, S. Kohlhaas, P.P. Das, E.A. Miska, A. Rodriguez, A. Bradley. microRNA-155 regulates the generation of immunoglobulin class-switched plasma cells. Immunity, 2007. [DOI | PubMed]
- M. Coffre, D. Benhamou, D. Rieß, L. Blumenberg, V. Snetkova, M.J. Hines, T. Chakraborty, S. Bajwa, K. Jensen, M.M.W. Chong. miRNAs Are Essential for the Regulation of the PI3K/AKT/FOXO Pathway and Receptor Editing during B Cell Maturation. Cell Rep., 2016. [DOI | PubMed]
- C. Xiao, D. Nemazee, A. Gonzalez-Martin. MicroRNA control of B cell tolerance, autoimmunity and cancer. Semin Cancer Biol., 2020. [DOI | PubMed]
- F. Li, Y. Huang, Y.Y. Huang, Y.S. Kuang, Y.J. Wei, L. Xiang, X.J. Zhang, Z.C. Jia, S. Jiang, J.Y. Li. MicroRNA-146a promotes IgE class switch in B cells via upregulating 14-3-3σ expression. Mol. Immunol., 2017. [DOI | PubMed]
- L.C. Edelstein, P.F. Bray. MicroRNAs in platelet production and activation. Blood, 2011. [DOI | PubMed]
- P. Czajka, A. Fitas, D. Jakubik, C. Eyileten, A. Gasecka, Z. Wicik, J.M. Siller-Matula, K.J. Filipiak, M. Postula. MicroRNA as Potential Biomarkers of Platelet Function on Antiplatelet Therapy: A Review. Front. Physiol., 2021. [DOI | PubMed]
- O. Sonmez, M. Sonmez. Role of platelets in immune system and inflammation. Porto Biomed. J., 2017. [DOI | PubMed]
- H.L. Anderson, I.E. Brodsky, N.S. Mangalmurti. The Evolving Erythrocyte: Red Blood Cells as Modulators of Innate Immunity. J. Immunol., 2018. [DOI | PubMed]
- N. Rivkin, E. Chapnik, A. Mildner, G. Barshtein, Z. Porat, E. Kartvelishvily, T. Dadosh, Y. Birger, G. Amir, S. Yedgar. Erythrocyte survival is controlled by microRNA-142. Haematologica, 2017. [DOI | PubMed]
- M.A. Lindsay. microRNAs and the immune response. Trends Immunol., 2008. [DOI | PubMed]
- A. Ventura, A.G. Young, M.M. Winslow, L. Lintault, A. Meissner, S.J. Erkeland, J. Newman, R.T. Bronson, D. Crowley, J.R. Stone. Targeted deletion reveals essential and overlapping functions of the miR-17 through 92 family of miRNA clusters. Cell, 2008. [DOI | PubMed]
- P. Landgraf, M. Rusu, R. Sheridan, A. Sewer, N. Iovino, A. Aravin, S. Pfeffer, A. Rice, A.O. Kamphorst, M. Landthaler. A mammalian microRNA expression atlas based on small RNA library sequencing. Cell, 2007. [DOI | PubMed]
- Q. Jing, S. Huang, S. Guth, T. Zarubin, A. Motoyama, J. Chen, F. Di Padova, S.C. Lin, H. Gram, J. Han. Involvement of microRNA in AU-rich element-mediated mRNA instability. Cell, 2005. [DOI | PubMed]
- R.M. O’Connell, K.D. Taganov, M.P. Boldin, G. Cheng, D. Baltimore. MicroRNA-155 is induced during the macrophage inflammatory response. Proc. Natl. Acad. Sci. USA, 2007. [DOI | PubMed]
- S. Altorki. Milk-Kinship in Arab Society: An Unexplored Problem in the Ethnography of Marriage. Ethnology, 1980. [DOI]
- S.B. Koralov, S.A. Muljo, G.R. Galler, A. Krek, T. Chakraborty, C. Kanellopoulou, K. Jensen, B.S. Cobb, M. Merkenschlager, N. Rajewsky. Dicer ablation affects antibody diversity and cell survival in the B lymphocyte lineage. Cell, 2008. [DOI | PubMed]
- A. Gaziel-Sovran, M.F. Segura, R. Di Micco, M.K. Collins, D. Hanniford, E. Vega-Saenz de Miera, J.F. Rakus, J.F. Dankert, S. Shang, R.S. Kerbel. miR-30b/30d regulation of GalNAc transferases enhances invasion and immunosuppression during metastasis. Cancer Cell, 2011. [DOI | PubMed]
- A.-B. Stittrich, C. Haftmann, E. Sgouroudis, A.A. Kühl, A.N. Hegazy, I. Panse, R. Riedel, M. Flossdorf, J. Dong, F. Fuhrmann. The microRNA miR-182 is induced by IL-2 and promotes clonal expansion of activated helper T lymphocytes. Nat. Immunol., 2010. [DOI | PubMed]
- A. Navarro, A. Gaya, A. Martinez, A. Urbano-Ispizua, A. Pons, O. Balague, B. Gel, P. Abrisqueta, A. Lopez-Guillermo, R. Artells. MicroRNA expression profiling in classic Hodgkin lymphoma. Blood, 2008. [DOI | PubMed]
- L. Vian, M. Di Carlo, E. Pelosi, F. Fazi, S. Santoro, A.M. Cerio, A. Boe, V. Rotilio, M. Billi, S. Racanicchi. Transcriptional fine-tuning of microRNA-223 levels directs lineage choice of human hematopoietic progenitors. Cell Death Differ., 2014. [DOI | PubMed]
- M. Perri, M. Lucente, R. Cannataro, I.F. De Luca, L. Gallelli, G. Moro, G. De Sarro, M.C. Caroleo, E. Cione. Variation in Immune-Related microRNAs Profile in Human Milk Amongst Lactating Women. Microrna, 2018. [DOI | PubMed]
- M.P. Roffel, K.R. Bracke, I.H. Heijink, T. Maes. miR-223: A Key Regulator in the Innate Immune Response in Asthma and COPD. Front. Med., 2020. [DOI]
- J. Dong, Y. Liu, W. Liao, R. Liu, P. Shi, L. Wang. miRNA-223 is a potential diagnostic and prognostic marker for osteosarcoma. J. Bone Oncol., 2016. [DOI | PubMed]
- J.B. Johnnidis, M.H. Harris, R.T. Wheeler, S. Stehling-Sun, M.H. Lam, O. Kirak, T.R. Brummelkamp, M.D. Fleming, F.D. Camargo. Regulation of progenitor cell proliferation and granulocyte function by microRNA-223. Nature, 2008. [DOI | PubMed]
- V.G. de Yebenes, L. Belver, D.G. Pisano, S. Gonzalez, A. Villasante, C. Croce, L. He, A.R. Ramiro. miR-181b negatively regulates activation-induced cytidine deaminase in B cells. J. Exp. Med., 2008. [DOI | PubMed]
- S.R. Quinn, N.E. Mangan, B.E. Caffrey, M.P. Gantier, B.R. Williams, P.J. Hertzog, C.E. McCoy, L.A. O’Neill. The role of Ets2 transcription factor in the induction of microRNA-155 (miR-155) by lipopolysaccharide and its targeting by interleukin-10. J Biol. Chem., 2014. [DOI | PubMed]
- B. Zhou, S. Wang, C. Mayr, D.P. Bartel, H.F. Lodish. miR-150, a microRNA expressed in mature B and T cells, blocks early B cell development when expressed prematurely. Proc. Natl. Acad. Sci. USA, 2007. [DOI | PubMed]
- C. Xiao, D.P. Calado, G. Galler, T.H. Thai, H.C. Patterson, J. Wang, N. Rajewsky, T.P. Bender, K. Rajewsky. MiR-150 controls B cell differentiation by targeting the transcription factor c-Myb. Cell, 2007. [DOI | PubMed]
- C. Xiao, L. Srinivasan, D.P. Calado, H.C. Patterson, B. Zhang, J. Wang, J.M. Henderson, J.L. Kutok, K. Rajewsky. Lymphoproliferative disease and autoimmunity in mice with increased miR-17-92 expression in lymphocytes. Nat. Immunol., 2008. [DOI | PubMed]
- C.Z. Chen, L. Li, H.F. Lodish, D.P. Bartel. MicroRNAs modulate hematopoietic lineage differentiation. Science, 2004. [DOI | PubMed]
- A. van den Berg, B.J. Kroesen, K. Kooistra, D. de Jong, J. Briggs, T. Blokzijl, S. Jacobs, J. Kluiver, A. Diepstra, E. Maggio. High expression of B-cell receptor inducible gene BIC in all subtypes of Hodgkin lymphoma. Genes Chromosomes Cancer, 2003. [DOI | PubMed]
- M. Metzler, M. Wilda, K. Busch, S. Viehmann, A. Borkhardt. High expression of precursor microRNA-155/BIC RNA in children with Burkitt lymphoma. Genes Chromosomes Cancer, 2004. [DOI | PubMed]
- S. Costinean, N. Zanesi, Y. Pekarsky, E. Tili, S. Volinia, N. Heerema, C.M. Croce. Pre-B cell proliferation and lymphoblastic leukemia/high-grade lymphoma in E(mu)-miR155 transgenic mice. Proc. Natl. Acad. Sci. USA, 2006. [DOI | PubMed]
- Y.S. Lee, K. Nakahara, J.W. Pham, K. Kim, Z. He, E.J. Sontheimer, R.W. Carthew. Distinct roles for Drosophila Dicer-1 and Dicer-2 in the siRNA/miRNA silencing pathways. Cell, 2004. [DOI | PubMed]
- B.S. Cobb, T.B. Nesterova, E. Thompson, A. Hertweck, E. O’Connor, J. Godwin, C.B. Wilson, N. Brockdorff, A.G. Fisher, S.T. Smale. T cell lineage choice and differentiation in the absence of the RNase III enzyme Dicer. J. Exp. Med., 2005. [DOI | PubMed]
- S.A. Muljo, K.M. Ansel, C. Kanellopoulou, D.M. Livingston, A. Rao, K. Rajewsky. Aberrant T cell differentiation in the absence of Dicer. J. Exp. Med., 2005. [DOI | PubMed]
- Q.J. Li, J. Chau, P.J. Ebert, G. Sylvester, H. Min, G. Liu, R. Braich, M. Manoharan, J. Soutschek, P. Skare. miR-181a is an intrinsic modulator of T cell sensitivity and selection. Cell, 2007. [DOI | PubMed]
- D. Yu, A.H. Tan, X. Hu, V. Athanasopoulos, N. Simpson, D.G. Silva, A. Hutloff, K.M. Giles, P.J. Leedman, K.P. Lam. Roquin represses autoimmunity by limiting inducible T-cell co-stimulator messenger RNA. Nature, 2007. [DOI | PubMed]
- L.F. Lu, T.H. Thai, D.P. Calado, A. Chaudhry, M. Kubo, K. Tanaka, G.B. Loeb, H. Lee, A. Yoshimura, K. Rajewsky. Foxp3-dependent microRNA155 confers competitive fitness to regulatory T cells by targeting SOCS1 protein. Immunity, 2009. [DOI | PubMed]
- F. Fazi, A. Rosa, A. Fatica, V. Gelmetti, M.L. De Marchis, C. Nervi, I. Bozzoni. A minicircuitry comprised of microRNA-223 and transcription factors NFI-A and C/EBPalpha regulates human granulopoiesis. Cell, 2005. [DOI | PubMed]
- E. Tili, J.J. Michaille, A. Cimino, S. Costinean, C.D. Dumitru, B. Adair, M. Fabbri, H. Alder, C.G. Liu, G.A. Calin. Modulation of miR-155 and miR-125b levels following lipopolysaccharide/TNF-alpha stimulation and their possible roles in regulating the response to endotoxin shock. J. Immunol., 2007. [DOI | PubMed]
- K.D. Taganov, M.P. Boldin, K.-J. Chang, D. Baltimore. NF-κB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc. Natl. Acad. Sci. USA, 2006. [DOI | PubMed]
- J. Mathieu, H. Ruohola-Baker. Regulation of stem cell populations by microRNAs. Transcriptional and Translational Regulation of Stem Cells, 2013
- X. Zhou, X. Li, M. Wu. miRNAs reshape immunity and inflammatory responses in bacterial infection. Signal Transduct. Target. Ther., 2018. [DOI | PubMed]
- F. Ma, S. Xu, X. Liu, Q. Zhang, X. Xu, M. Liu, M. Hua, N. Li, H. Yao, X. Cao. The microRNA miR-29 controls innate and adaptive immune responses to intracellular bacterial infection by targeting interferon-gamma. Nat. Immunol., 2011. [DOI | PubMed]
- H. Hemmi, O. Takeuchi, T. Kawai, T. Kaisho, S. Sato, H. Sanjo, M. Matsumoto, K. Hoshino, H. Wagner, K. Takeda. A Toll-like receptor recognizes bacterial DNA. Nature, 2000. [DOI | PubMed]
- A. Rolls, R. Shechter, A. London, Y. Ziv, A. Ronen, R. Levy, M. Schwartz. Toll-like receptors modulate adult hippocampal neurogenesis. Nat. Cell Biol., 2007. [DOI | PubMed]
- J.K. Dowling, A. Mansell. Toll-like receptors: The swiss army knife of immunity and vaccine development. Clin. Transl. Immunol., 2016. [DOI]
- C. Chen, S. Schaffert, R. Fragoso, C. Loh. Regulation of immune responses and tolerance: The microRNA perspective. Immunol. Rev., 2013. [DOI | PubMed]
- K.M. Pauley, S. Cha, E.K. Chan. MicroRNA in autoimmunity and autoimmune diseases. J. Autoimmun., 2009. [DOI | PubMed]
- T.X. Lu, A. Munitz, M.E. Rothenberg. MicroRNA-21 is up-regulated in allergic airway inflammation and regulates IL-12p35 expression. J. Immunol., 2009. [DOI | PubMed]
- W.H. Oddy. The long-term effects of breast-feeding on asthma and atopic disease. Breast-Feeding: Early Influences on Later Health, 2009
- C. Dupont, D.R. Armant, C.A. Brenner. Epigenetics: Definition, mechanisms and clinical perspective. Semin. Reprod. Med., 2009. [DOI | PubMed]
- R.D. Hotchkiss. The quantitative separation of purines, pyrimidines, and nucleosides by paper chromatography. J. Biol. Chem., 1948. [DOI | PubMed]
- R.L. Sinsheimer. The action of pancreatic deoxyribonuclease. II. Isomeric dinucleotides. J. Biol. Chem., 1955. [DOI | PubMed]
- X. Nan, H.H. Ng, C.A. Johnson, C.D. Laherty, B.M. Turner, R.N. Eisenman, A. Bird. Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature, 1998. [DOI | PubMed]
- C.R. Clapier, B.R. Cairns. The biology of chromatin remodeling complexes. Annu. Rev. Biochem., 2009. [DOI | PubMed]
- A.R. Isles, L.S. Wilkinson. Epigenetics: What is it and why is it important to mental disease?. Br. Med. Bull., 2008. [DOI | PubMed]
- H.J. Lipps, J. Postberg, D.A. Jackson, A. Silahtaroglu, J. Stenvang. MicroRNAs, epigenetics and disease. Essays Biochem., 2010. [DOI | PubMed]
- A. Ahmad, K.R. Ginnebaugh, S. Yin, A. Bollig-Fischer, K.B. Reddy, F.H. Sarkar. Functional role of miR-10b in tamoxifen resistance of ER-positive breast cancer cells through down-regulation of HDAC4. BMC Cancer, 2015. [DOI | PubMed]
- R.A. El-Awady, F. Hersi, H. Al-Tunaiji, E.M. Saleh, A.-H.A. Abdel-Wahab, A. Al Homssi, M. Suhail, A. El-Serafi, T. Al-Tel. Epigenetics and miRNA as predictive markers and targets for lung cancer chemotherapy. Cancer Biol. Ther., 2015. [DOI | PubMed]
- F. Memari, Z. Joneidi, B. Taheri, S.F. Aval, A. Roointan, N. Zarghami. Epigenetics and Epi-miRNAs: Potential markers/therapeutics in leukemia. Biomed. Pharmacol., 2018. [DOI]
- R. Benetti, S. Gonzalo, I. Jaco, P. Munoz, S. Gonzalez, S. Schoeftner, E. Murchison, T. Andl, T. Chen, P. Klatt. A mammalian microRNA cluster controls DNA methylation and telomere recombination via Rbl2-dependent regulation of DNA methyltransferases. Nat. Struct. Mol. Biol., 2008. [DOI | PubMed]
- B. Khraiwesh, M.A. Arif, G.I. Seumel, S. Ossowski, D. Weigel, R. Reski, W. Frank. Transcriptional control of gene expression by microRNAs. Cell, 2010. [DOI | PubMed]
- L. Tuddenham, G. Wheeler, S. Ntounia-Fousara, J. Waters, M.K. Hajihosseini, I. Clark, T. Dalmay. The cartilage specific microRNA-140 targets histone deacetylase 4 in mouse cells. FEBS Lett., 2006. [DOI | PubMed]
- J.-F. Chen, E.M. Mandel, J.M. Thomson, Q. Wu, T.E. Callis, S.M. Hammond, F.L. Conlon, D.-Z. Wang. The role of microRNA-1 and microRNA-133 in skeletal muscle proliferation and differentiation. Nat. Genet., 2006. [DOI | PubMed]
- A.H. Williams, G. Valdez, V. Moresi, X. Qi, J. McAnally, J.L. Elliott, R. Bassel-Duby, J.R. Sanes, E.N. Olson. MicroRNA-206 delays ALS progression and promotes regeneration of neuromuscular synapses in mice. Science, 2009. [DOI | PubMed]
- J.C. Chuang, P.A. Jones. Epigenetics and MicroRNAs. Pediatric Res., 2007. [DOI | PubMed]
- H. Denis, O. Van Grembergen, B. Delatte, S. Dedeurwaerder, P. Putmans, E. Calonne, F. Rothé, C. Sotiriou, F. Fuks, R. Deplus. MicroRNAs regulate KDM5 histone demethylases in breast cancer cells. Mol. Biosyst., 2016. [DOI | PubMed]
- M. Bianchi, A. Renzini, S. Adamo, V. Moresi. Coordinated Actions of MicroRNAs with other Epigenetic Factors Regulate Skeletal Muscle Development and Adaptation. Int. J. Mol. Sci., 2017. [DOI]
- S. Meng, H. Zhou, Z. Feng, Z. Xu, Y. Tang, M. Wu. Epigenetics in Neurodevelopment: Emerging Role of Circular RNA. Front Cell Neurosci., 2019. [DOI | PubMed]
- M. Magistri, M.A. Faghihi, G. St Laurent, C. Wahlestedt. Regulation of chromatin structure by long noncoding RNAs: Focus on natural antisense transcripts. Trends Genet, 2012. [DOI | PubMed]
- M.A.I. Al-Hatamleh, E.N.S. EAR, J.C. Boer, K. Ferji, J.L. Six, X. Chen, E. Elkord, M. Plebanski, R. Mohamud. Synergistic Effects of Nanomedicine Targeting TNFR2 and DNA Demethylation Inhibitor-An Opportunity for Cancer Treatment. Cells, 2019. [DOI]
- N. Celarain, J. Tomas-Roig. Aberrant DNA methylation profile exacerbates inflammation and neurodegeneration in multiple sclerosis patients. J. Neuroinflamm., 2020. [DOI]
- S. Wang, W. Wu, F.X. Claret. Mutual regulation of microRNAs and DNA methylation in human cancers. Epigenetics, 2017. [DOI | PubMed]
- M. Busslinger, A. Tarakhovsky. Epigenetic control of immunity. Cold Spring Harb. Perspect. Biol., 2014. [DOI | PubMed]
- L. Bezu, A.W. Chuang, P. Liu, G. Kroemer, O. Kepp. Immunological Effects of Epigenetic Modifiers. Cancers, 2019. [DOI]
- K. Hirahara, G. Vahedi, K. Ghoreschi, X.P. Yang, S. Nakayamada, Y. Kanno, J.J. O’Shea, A. Laurence. Helper T-cell differentiation and plasticity: Insights from epigenetics. Immunology, 2011. [DOI | PubMed]
- T.M. Aune, P.L. Collins, S.P. Collier, M.A. Henderson, S. Chang. Epigenetic Activation and Silencing of the Gene that Encodes IFN-gamma. Front. Immunol., 2013. [DOI | PubMed]
- A.M. Chomyk, C. Volsko, A. Tripathi, S.A. Deckard, B.D. Trapp, R.J. Fox, R. Dutta. DNA methylation in demyelinated multiple sclerosis hippocampus. Sci. Rep., 2017. [DOI | PubMed]
- J.M. Greer, P.A. McCombe. The role of epigenetic mechanisms and processes in autoimmune disorders. Biologics, 2012. [DOI | PubMed]
- S. Lovinsky-Desir, R.L. Miller. Epigenetics, asthma, and allergic diseases: A review of the latest advancements. Curr. Allergy Asthma Rep., 2012. [DOI | PubMed]
- W. Cookson. The alliance of genes and environment in asthma and allergy. Nature, 1999. [DOI | PubMed]
- E.G. Barrett. Maternal influence in the transmission of asthma susceptibility. Pulm. Pharmacol., 2008. [DOI]
- B. Suarez-Alvarez, A. Baragano Raneros, F. Ortega, C. Lopez-Larrea. Epigenetic modulation of the immune function: A potential target for tolerance. Epigenetics, 2013. [DOI | PubMed]
- M. Ehrlich, M.A. Gama-Sosa, L.H. Huang, R.M. Midgett, K.C. Kuo, R.A. McCune, C. Gehrke. Amount and distribution of 5-methylcytosine in human DNA from different types of tissues or cells. Nucleic Acids Res., 1982. [DOI | PubMed]
- K.L. Tucker. Methylated cytosine and the brain: A new base for neuroscience. Neuron, 2001. [DOI | PubMed]
- H. Cedar, Y. Bergman. Linking DNA methylation and histone modification: Patterns and paradigms. Nat. Rev. Genet., 2009. [DOI | PubMed]
- R.A. Hlady, S. Novakova, J. Opavska, D. Klinkebiel, S.L. Peters, J. Bies, J. Hannah, J. Iqbal, K.M. Anderson, H.M. Siebler. Loss of Dnmt3b function upregulates the tumor modifier Ment and accelerates mouse lymphomagenesis. J. Clin. Investig., 2012. [DOI | PubMed]
- S.A. Tammen, S. Friso, S.W. Choi. Epigenetics: The link between nature and nurture. Mol. Asp. Med., 2013. [DOI]
- D. You, E. Nilsson, D.E. Tenen, A. Lyubetskaya, J.C. Lo, R. Jiang, J. Deng, B.A. Dawes, A. Vaag, C. Ling. Dnmt3a is an epigenetic mediator of adipose insulin resistance. Elife, 2017. [DOI]
- Y. Hatazawa, Y. Ono, Y. Hirose, S. Kanai, N.L. Fujii, S. Machida, I. Nishino, T. Shimizu, M. Okano, Y. Kamei. Reduced Dnmt3a increases Gdf5 expression with suppressed satellite cell differentiation and impaired skeletal muscle regeneration. FASEB J., 2018. [DOI | PubMed]
- H. Kutay, C. Klepper, B. Wang, S.H. Hsu, J. Datta, L. Yu, X. Zhang, S. Majumder, T. Motiwala, N. Khan. Reduced susceptibility of DNA methyltransferase 1 hypomorphic (Dnmt1N/+) mice to hepatic steatosis upon feeding liquid alcohol diet. PLoS ONE, 2012. [DOI | PubMed]
- B.C. Melnik, G. Schmitz. Milk’s Role as an Epigenetic Regulator in Health and Disease. Diseases, 2017. [DOI]
- U. Smyczynska, M.A. Bartlomiejczyk, M.M. Stanczak, P. Sztromwasser, A. Wesolowska, O. Barbarska, E. Pawlikowska, W. Fendler. Impact of processing method on donated human breast milk microRNA content. PLoS ONE, 2020. [DOI | PubMed]
- B.C. Melnik, W. Stremmel, R. Weiskirchen, S.M. John, G. Schmitz. Exosome-Derived MicroRNAs of Human Milk and Their Effects on Infant Health and Development. Biomolecules, 2021. [DOI | PubMed]
- V. Fernandez Vallone, M. Leprovots, D. Ribatallada-Soriano, R. Gerbier, A. Lefort, F. Libert, G. Vassart, M.I. Garcia. LGR5 controls extracellular matrix production by stem cells in the developing intestine. EMBO Rep., 2020. [DOI | PubMed]
- R.J. Kusuma, S. Manca, T. Friemel, S. Sukreet, C. Nguyen, J. Zempleni. Human vascular endothelial cells transport foreign exosomes from cow’s milk by endocytosis. Am. J. Physiol. Cell Physiol., 2016. [DOI | PubMed]
- B.C. Melnik, S.M. John, G. Schmitz. Milk is not just food but most likely a genetic transfection system activating mTORC1 signaling for postnatal growth. Nutr. J., 2013. [DOI | PubMed]
- B.C. Melnik, F. Kakulas, D.T. Geddes, P.E. Hartmann, S.M. John, P. Carrera-Bastos, L. Cordain, G. Schmitz. Milk miRNAs: Simple nutrients or systemic functional regulators?. Nutr. Metab., 2016. [DOI]
- H. Cao, L. Wang, B. Chen, P. Zheng, Y. He, Y. Ding, Y. Deng, X. Lu, X. Guo, Y. Zhang. DNA Demethylation Upregulated Nrf2 Expression in Alzheimer’s Disease Cellular Model. Front. Aging Neurosci., 2015. [DOI | PubMed]
- M. Fabbri, R. Garzon, A. Cimmino, Z. Liu, N. Zanesi, E. Callegari, S. Liu, H. Alder, S. Costinean, C. Fernandez-Cymering. MicroRNA-29 family reverts aberrant methylation in lung cancer by targeting DNA methyltransferases 3A and 3B. Proc. Natl. Acad. Sci. USA, 2007. [DOI | PubMed]
- A. Kuroda, T.A. Rauch, I. Todorov, H.T. Ku, I.H. Al-Abdullah, F. Kandeel, Y. Mullen, G.P. Pfeifer, K. Ferreri. Insulin gene expression is regulated by DNA methylation. PLoS ONE, 2009. [DOI | PubMed]
- L.Q. Qin, K. He, J.Y. Xu. Milk consumption and circulating insulin-like growth factor-I level: A systematic literature review. Int. J. Food. Sci. Nutr., 2009. [DOI | PubMed]
- M. Ouni, Y. Gunes, M.-P. Belot, A.-L. Castell, D. Fradin, P. Bougnères. The IGF1 P2 promoter is an epigenetic QTL for circulating IGF1 and human growth. Clin. Epigenet., 2015. [DOI]
- S. Palacios-Ortega, M. Varela-Guruceaga, F.I. Milagro, J.A. Martinez, C. de Miguel. Expression of Caveolin 1 is enhanced by DNA demethylation during adipocyte differentiation. status of insulin signaling. PLoS ONE, 2014. [DOI | PubMed]
- M. Braud, D.A. Magee, S.D. Park, T.S. Sonstegard, S.M. Waters, D.E. MacHugh, C. Spillane. Genome-Wide microRNA Binding Site Variation between Extinct Wild Aurochs and Modern Cattle Identifies Candidate microRNA-Regulated Domestication Genes. Front. Genet., 2017. [DOI | PubMed]
- V. Labrie, O.J. Buske, E. Oh, R. Jeremian, C. Ptak, G. Gasiunas, A. Maleckas, R. Petereit, A. Zvirbliene, K. Adamonis. Lactase nonpersistence is directed by DNA-variation-dependent epigenetic aging. Nat. Struct. Mol. Biol., 2016. [DOI | PubMed]
- G. Jia, Y. Fu, X. Zhao, Q. Dai, G. Zheng, Y. Yang, C. Yi, T. Lindahl, T. Pan, Y.G. Yang. N6-methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nat. Chem. Biol., 2011. [DOI | PubMed]
- N. Auphan, J.A. DiDonato, C. Rosette, A. Helmberg, M. Karin. Immunosuppression by glucocorticoids: Inhibition of NF-kappa B activity through induction of I kappa B synthesis. Science, 1995. [DOI | PubMed]
- R.I. Scheinman, P.C. Cogswell, A.K. Lofquist, A.S. Baldwin. Role of transcriptional activation of I kappa B alpha in mediation of immunosuppression by glucocorticoids. Science, 1995. [DOI | PubMed]
- W.Y. Almawi, O.K. Melemedjian. Negative regulation of nuclear factor-kappaB activation and function by glucocorticoids. J. Mol. Endocrinol., 2002. [DOI | PubMed]
- S.-Q. Mao, S.M. Cuesta, D. Tannahill, S. Balasubramanian. Genome-wide DNA Methylation Signatures Are Determined by DNMT3A/B Sequence Preferences. Biochemistry, 2020. [DOI | PubMed]
- L. Sun, L. Huang, P. Nguyen, K.S. Bisht, G. Bar-Sela, A.S. Ho, C.M. Bradbury, W. Yu, H. Cui, S. Lee. DNA methyltransferase 1 and 3B activate BAG-1 expression via recruitment of CTCFL/BORIS and modulation of promoter histone methylation. Cancer Res., 2008. [DOI | PubMed]
- R. Deplus, C. Brenner, W.A. Burgers, P. Putmans, T. Kouzarides, Y. de Launoit, F. Fuks. Dnmt3L is a transcriptional repressor that recruits histone deacetylase. Nucleic Acids Res., 2002. [DOI | PubMed]
- E. Viré, C. Brenner, R. Deplus, L. Blanchon, M. Fraga, C. Didelot, L. Morey, A. Van Eynde, D. Bernard, J.-M. Vanderwinden. The Polycomb group protein EZH2 directly controls DNA methylation. Nature, 2006. [DOI | PubMed]
- J.Z. Tan, Y. Yan, X.X. Wang, Y. Jiang, H.E. Xu. EZH2: Biology, disease, and structure-based drug discovery. Acta Pharmacol. Sin., 2014. [DOI | PubMed]
- B. Lee, M.T. Muller. SUMOylation enhances DNA methyltransferase 1 activity. Biochem. J., 2009. [DOI | PubMed]
- V.K.C. Ponnaluri, P.O. Esteve, C.I. Ruse, S. Pradhan. S-adenosylhomocysteine Hydrolase Participates in DNA Methylation Inheritance. J. Mol. Biol., 2018. [DOI | PubMed]
- Y. Zhu, H. Lu, D. Zhang, M. Li, X. Sun, L. Wan, D. Yu, Y. Tian, H. Jin, A. Lin. Integrated analyses of multi-omics reveal global patterns of methylation and hydroxymethylation and screen the tumor suppressive roles of HADHB in colorectal cancer. Clin. Epigenet., 2018. [DOI]
- S.A. Obermann-Borst, P.H.C. Eilers, E.W. Tobi, F.H. de Jong, P.E. Slagboom, B.T. Heijmans, R.P.M. Steegers-Theunissen. Duration of breastfeeding and gender are associated with methylation of the LEPTIN gene in very young children. Pediatric Res., 2013. [DOI]
- M.H. Tao, C. Marian, P.G. Shields, N. Potischman, J. Nie, S.S. Krishnan, D.L. Berry, B.V. Kallakury, C. Ambrosone, S.B. Edge. Exposures in early life: Associations with DNA promoter methylation in breast tumors. J. Dev. Orig. Health Dis., 2013. [DOI | PubMed]
- A. Rossnerova, E. Tulupova, N. Tabashidze, J. Schmuczerova, M. Dostal, P. Rossner, H. Gmuender, R.J. Sram. Factors affecting the 27K DNA methylation pattern in asthmatic and healthy children from locations with various environments. Mutat. Res., 2013. [DOI]
- N. Soto-Ramirez, S.H. Arshad, J.W. Holloway, H. Zhang, E. Schauberger, S. Ewart, V. Patil, W. Karmaus. The interaction of genetic variants and DNA methylation of the interleukin-4 receptor gene increase the risk of asthma at age 18 years. Clin. Epigenet., 2013. [DOI]
- O.Y. Naumova, V.V. Odintsova, I.A. Arincina, S.Y. Rychkov, R.J. Muhamedrahimov, Y.V. Shneider, A.N. Grosheva, O.V. Zhukova, E.L. Grigorenko. A Study of the Association between Breastfeeding and DNA Methylation in Peripheral Blood Cells of Infants. Russ. J. Genet., 2019. [DOI]
- F.P. Hartwig, C. Loret de Mola, N.M. Davies, C.G. Victora, C.L. Relton. Breastfeeding effects on DNA methylation in the offspring: A systematic literature review. PLoS ONE, 2017. [DOI | PubMed]
- W.B. Sherwood, V. Bion, G.A. Lockett, A.H. Ziyab, N. Soto-Ramírez, N. Mukherjee, R.J. Kurukulaaratchy, S. Ewart, H. Zhang, S.H. Arshad. Duration of breastfeeding is associated with leptin (LEP) DNA methylation profiles and BMI in 10-year-old children. Clin. Epigenet., 2019. [DOI | PubMed]
- S. Pauwels, L. Symons, E.L. Vanautgaerden, M. Ghosh, R.C. Duca, B. Bekaert, K. Freson, I. Huybrechts, S.A.S. Langie, G. Koppen. The Influence of the Duration of Breastfeeding on the Infant’s Metabolic Epigenome. Nutrients, 2019. [DOI | PubMed]
- W.B. Sherwood, D.M. Kothalawala, L. Kadalayil, S. Ewart, H. Zhang, W. Karmaus, S.H. Arshad, J.W. Holloway, F.I. Rezwan. Epigenome-Wide Association Study Reveals Duration of Breastfeeding Is Associated with Epigenetic Differences in Children. Int. J. Environ. Res. Public Health, 2020. [DOI]
- N. Acevedo, B. Alashkar Alhamwe, L. Caraballo, M. Ding, A. Ferrante, H. Garn, J. Garssen, C.S. Hii, J. Irvine, K. Llinas-Caballero. Perinatal and Early-Life Nutrition, Epigenetics, and Allergy. Nutrients, 2021. [DOI | PubMed]
- X. Hao, Y. Wang, F. Ren, S. Zhu, Y. Ren, B. Jia, Y.P. Li, Y. Shi, Z. Chang. SNX25 regulates TGF-β signaling by enhancing the receptor degradation. Cell. Signal., 2011. [DOI | PubMed]
- W.H. Oddy, F. Rosales. A systematic review of the importance of milk TGF-beta on immunological outcomes in the infant and young child. Pediatric Allergy Immunol. Off. Publ. Eur. Soc. Pediatric Allergy Immunol., 2010. [DOI]
- B. Tirado-Rodriguez, E. Ortega, P. Segura-Medina, S. Huerta-Yepez. TGF- β: An important mediator of allergic disease and a molecule with dual activity in cancer development. J. Immunol. Res., 2014. [DOI | PubMed]
- L. Briollais, D. Rustand, C. Allard, Y. Wu, J. Xu, S.G. Rajan, M.F. Hivert, M. Doyon, L. Bouchard, P.O. McGowan. DNA methylation mediates the association between breastfeeding and early-life growth trajectories. Clin. Epigenet., 2021. [DOI]
- A.J. Simpkin, M. Suderman, T.R. Gaunt, O. Lyttleton, W.L. McArdle, S.M. Ring, K. Tilling, G. Davey Smith, C.L. Relton. Longitudinal analysis of DNA methylation associated with birth weight and gestational age. Hum. Mol. Genet., 2015. [DOI | PubMed]
- P.D. Fransquet, D. Hutchinson, C.A. Olsson, J. Wilson, S. Allsop, J. Najman, E. Elliott, R.P. Mattick, R. Saffery, J. Ryan. Perinatal maternal alcohol consumption and methylation of the dopamine receptor DRD4 in the offspring: The Triple B study. Environ. Epigenet., 2016. [DOI | PubMed]
- C.W. Genna. Epigenetics, Methylation, and Breastfeeding. Clin. Lact., 2018. [DOI]
- Y. Mallisetty, N. Mukherjee, Y. Jiang, S. Chen, S. Ewart, S.H. Arshad, J.W. Holloway, H. Zhang, W. Karmaus. Epigenome-Wide Association of Infant Feeding and Changes in DNA Methylation from Birth to 10 Years. Nutrients, 2020. [DOI]
- V.V. Odintsova, F.A. Hagenbeek, M. Suderman, D. Caramaschi, C.E.M. van Beijsterveldt, N.A. Kallsen, E.A. Ehli, G.E. Davies, G.T. Sukhikh, V. Fanos. DNA Methylation Signatures of Breastfeeding in Buccal Cells Collected in Mid-Childhood. Nutrients, 2019. [DOI]
- M.M. Hatmal, N.N. Issa, W. Alshaer, H.J. Al-Ameer, O. Abuyaman, R. Tayyem, N.S. Hijjawi. Association of Breastfeeding Duration with Susceptibility to Allergy, Influenza, and Methylation Status of TLR1 Gene. Medicina, 2019. [DOI]
- F.P. Hartwig, G. Davey Smith, A.J. Simpkin, C.G. Victora, C.L. Relton, D. Caramaschi. Association between Breastfeeding and DNA Methylation over the Life Course: Findings from the Avon Longitudinal Study of Parents and Children (ALSPAC). Nutrients, 2020. [DOI]
- R.M. Taylor, R. Smith, C.E. Collins, D. Mossman, M.W. Wong-Brown, E.C. Chan, T.J. Evans, J.R. Attia, N. Buckley, K. Drysdale. Global DNA methylation and cognitive and behavioral outcomes at 4 years of age: A cross-sectional study. Brain Behav., 2020. [DOI | PubMed]
- J.K.d. Castilhos, P.D.B. Campagnolo, S.d. Almeida, M.R. Vitolo, V.S. Mattevi. Impact of maternal dietary counseling in the first year of life on DNA methylation in a cohort of children. Genet. Mol. Biol., 2021. [DOI | PubMed]
- C.M. Anderson, S.L. Gillespie, D.K. Thiele, J.L. Ralph, J.E. Ohm. Effects of Maternal Vitamin D Supplementation on the Maternal and Infant Epigenome. Breastfeed. Med., 2018. [DOI | PubMed]
- O. Chatterjee, K. Patil, A. Sahu, L. Gopalakrishnan, P. Mol, J. Advani, S. Mukherjee, R. Christopher, T.S. Prasad. An overview of the oxytocin-oxytocin receptor signaling network. J. Cell Commun. Signal., 2016. [DOI | PubMed]
- M.C. Neville, S.M. Anderson, J.L. McManaman, T.M. Badger, M. Bunik, N. Contractor, T. Crume, D. Dabelea, S.M. Donovan, N. Forman. Lactation and neonatal nutrition: Defining and refining the critical questions. J. Mammary Gland Biol. Neoplasia, 2012. [DOI | PubMed]
- S. Baumgartner, D. Martin, C. Hagios, R. Chiquet-Ehrismann. Tenm, a Drosophila gene related to tenascin, is a new pair-rule gene. EMBO J., 1994. [DOI | PubMed]
- T. Oohashi, X.H. Zhou, K. Feng, B. Richter, M. Morgelin, M.T. Perez, W.D. Su, R. Chiquet-Ehrismann, U. Rauch, R. Fassler. Mouse ten-m/Odz is a new family of dimeric type II transmembrane proteins expressed in many tissues. J. Cell Biol., 1999. [DOI | PubMed]
- L. Zheng, Y. Michelson, V. Freger, Z. Avraham, K.J. Venken, H.J. Bellen, M.J. Justice, R. Wides. Drosophila Ten-m and filamin affect motor neuron growth cone guidance. PLoS ONE, 2011. [DOI | PubMed]
- Y. Chen, F. Siegel, S. Kipschull, B. Haas, H. Frohlich, G. Meister, A. Pfeifer. miR-155 regulates differentiation of brown and beige adipocytes via a bistable circuit. Nat. Commun., 2013. [DOI | PubMed]
- X. Chen, C. Gao, H. Li, L. Huang, Q. Sun, Y. Dong, C. Tian, S. Gao, H. Dong, D. Guan. Identification and characterization of microRNAs in raw milk during different periods of lactation, commercial fluid, and powdered milk products. Cell Res., 2010. [DOI | PubMed]
- A. He, L. Zhu, N. Gupta, Y. Chang, F. Fang. Overexpression of micro ribonucleic acid 29, highly up-regulated in diabetic rats, leads to insulin resistance in 3T3-L1 adipocytes. Mol. Endocrinol., 2007. [DOI | PubMed]
- M.A. Dehwah, A. Xu, Q. Huang. MicroRNAs and type 2 diabetes/obesity. J Genet Genom., 2012. [DOI]
- F. Hassiotou, D.T. Geddes. Programming of appetite control during breastfeeding as a preventative strategy against the obesity epidemic. J. Hum. Lact., 2014. [DOI | PubMed]
- F. Meng, R. Henson, H. Wehbe-Janek, K. Ghoshal, S.T. Jacob, T. Patel. MicroRNA-21 regulates expression of the PTEN tumor suppressor gene in human hepatocellular cancer. Gastroenterology, 2007. [DOI | PubMed]
- F. Olivieri, L. Spazzafumo, G. Santini, R. Lazzarini, M.C. Albertini, M.R. Rippo, R. Galeazzi, A.M. Abbatecola, F. Marcheselli, D. Monti. Age-related differences in the expression of circulating microRNAs: miR-21 as a new circulating marker of inflammaging. Mech. Ageing Dev., 2012. [DOI | PubMed]
- Y.J. Kim, S.J. Hwang, Y.C. Bae, J.S. Jung. MiR-21 regulates adipogenic differentiation through the modulation of TGF-beta signaling in mesenchymal stem cells derived from human adipose tissue. Stem Cells, 2009. [DOI | PubMed]
- S.M. Hammond. MicroRNAs as tumor suppressors. Nat. Genet., 2007. [DOI | PubMed]
- S.M. Johnson, H. Grosshans, J. Shingara, M. Byrom, R. Jarvis, A. Cheng, E. Labourier, K.L. Reinert, D. Brown, F.J. Slack. RAS is regulated by the let-7 microRNA family. Cell, 2005. [DOI | PubMed]
