Challenges in the management of adenocarcinoma of ampulla of Vater in pregnancy: A case report and review of literature
Abstract
Ampullary adenocarcinoma is a rare entity during pregnancy.
Diagnosing ampullary adenocarcinoma in pregnancy can be quite challenging, as the symptoms may overlap with the physiological changes of pregnancy. Furthermore, the diagnostic modalities’ invasiveness & the associated radiation exposure that might harm the fetus.
The best treatment modality for resectable tumors is in the form of surgery (i.e.; pancreaticoduodenectomy) that is possible during pregnancy. Yet challenging as its important to choose the appropriate time of surgery to insure the viability of the fetus without risking the progression of the disease.
Another challenge that might be encountered is intra-operatively, due to the bulkiness of the uterus which makes such a procedure even more difficult to preform safely & successfully.
Article type: Case Report
Keywords: Ampulla of Vater, Adenocarcinoma, Jaundice, Pregnancy, Pancreaticoduodenectomy
Affiliations: Imam Abdulrahman Bin Faisal University, Dammam, Saudi Arabia; Department of General Surgery, King Fahad Specialist Hospital, Dammam, Saudi Arabia
License: © 2019 The Authors CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.ijscr.2019.06.044 | PubMed: 31306902 | PMC: PMC6626975
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (945 KB)
Introduction
This work has been reported in line with the SCARE criteria [ref. 1].
Ampullary adenocarcinoma is a malignant tumor originating from the ampulla of Vater and it accounts for 0.5% of all gastrointestinal (GI) malignancies [ref. 2]. Ampullary adenocarcinoma is rare entity during pregnancy [ref. 3]. The best treatment modality for resectable tumors providing the best outcome is a complete surgical resection in the form of pancreaticoduodenectomy (Whipple procedure) [ref. 4]. Here, we report a case of 22 years old pregnant female at 28th week of gestation and its challenges in diagnosis and management & a review of literature.
Case presentation
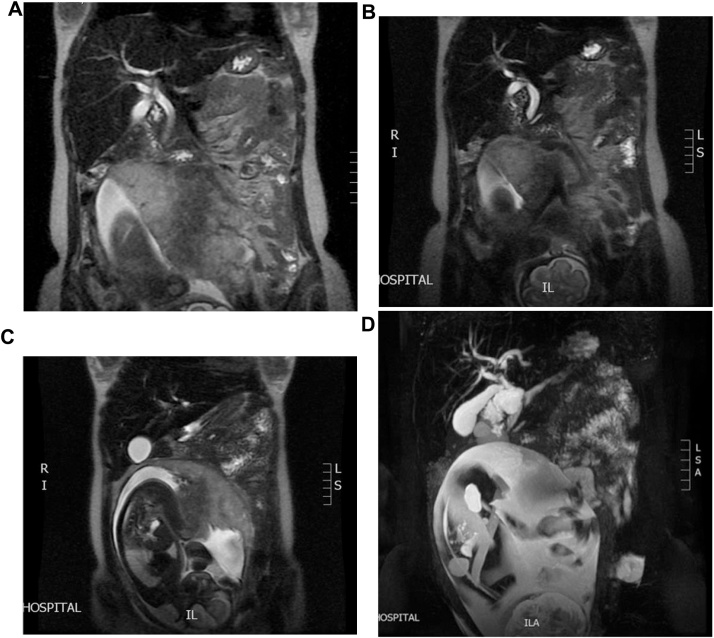
We report a case of a 22 years old pregnant female medically free, referred to us from her Obstetrician when she was complaining of abdominal pain and jaundice. Abdominal US confirmed a single intrauterine pregnancy at 28th week of gestation with appropriate growth for date and showed dilated intrahepatic ducts otherwise it was inconclusive due to the gravid uterus (Fig. 1). Her blood investigations showed a picture of cholestatic jaundice, and all other labs were within normal. So, we decided to proceed with a Magnetic resonance cholangiopancreatography (MRCP) which showed dilatation of both of the CBD (measuring 0.9 cm) & pancreatic duct, as well as an ampullary mass measuring 2 cm (Fig. 2). Later on, Endoscopic retrograde cholangiopancreatography (ERCP) with shielding of the abdomen to protect the fetus from radiation revealed an ampullary and distal CBD strictures. A punch biopsy was taken & the CBD was stented. The histopathology came as invasive adenocarcinoma & full metastatic work up was done and did not reveal any metastatic lesions. So, surgery was the best available option with the best possible outcome but we were reluctant to delay the surgery to ascertain the viability of the fetus. At 34th week of gestation induction of labor was done, both mother and the baby did well and were discharged home on 2nd day postpartum. The mother was readmitted one week later & full body CT scan repeated & there was no vascular invasion or distant metastasis.
Therefore, we proceeded with pancreaticoduodenectomy. A laparotomy incision was done, intraoperative examination of the abdomen revealed; a palpable mass at the ampulla of Vater and the stent was felt in the CBD and duodenum, a bulky uterus as the patient was still in the postpartum period. There was no vascular invasion, peritoneal deposits or any other distant metastasis. For the pancreatojejunostomy anastomosis, a two-layer end-to-side duct-to-mucosa approach was adopted. The pancreatic duct was stented to divert the pancreatic secretions away from the anastomosis. Then the hepaticojejunostomy was done in an end-to-side fashion followed by the gastrojejunostomy.
The Patient had uneventful postoperative course and was discharged 1 week after her surgery. Histopathology came as poorly differentiated invasive adenocarcinoma of ampulla of Vater with negative resection margins. Three out of thirteen lymph nodes revealed metastatic involvement so she received six cycles of adjuvant chemotherapy which she tolerated well. Upon 6 years follow up, computed tomography (CT) and positron emission tomography (PET) scans were normal with no evidence of recurrence.
Discussion
Patients with periampullary tumors can present with wide variety of gastrointestinal symptoms. The most commonly is obstructive jaundice 2ry to CBD obstruction. In other cases, they can present with biliary colic along with constitutional symptoms especially in adenocarcinoma compared to other types of GI malignancies [ref. 5].
We reviewed published articles concerning periampullary & pancreatic malignant tumors that has been diagnosed during pregnancy & confirmed by histopathology in the English literature. To the best of our knowledge, only 41 cases have been reported (Table 1) and our case is the 42nd. Porcel et al. published the first case of adenocarcinoma of ampulla of Vater in pregnant woman at 3rd trimester in 1992 [ref. 6]. Upon our review (N = 41), the common age of presentation that was found in the literature was during the 30 s in contrast to our case, where the patient presented to us at the age of 22 years old. The most common histopathological type was found to be adenocarcinoma (18 cases), followed by mucinous cystic neoplasm (16 cases), neuroendocrine neoplasms (3 cases), Solid-pseudopapillary neoplasms (3 cases) and 1 case had anaplastic.
Table 1: Literature review of 41 cases.
| Author | Histology | Patient age | Clinical presentation | Gestation at presentation (trimester/ weeks) | Diagnosis method | Gestation for Surgery (trimester/ weeks) | Complications |
|---|---|---|---|---|---|---|---|
| Smithers et al. [ref. 12] | MCN | – | – | 1st (7) | – | 1 st (8) | Tumor ruptured |
| Baiocchi et al. [ref. 13] | MCN | – | – | 3rd | – | Postpartum | None reported |
| Porcel et al. [ref. 6] | Adenocarcinoma | 43 | epigastric pain,Upper lumbar backache, nausea and vomiting. | 3rd (28) | Aspiration cytology | – | severe pre-eclampsia, HELLP syndrome, left axillar venous thrombosis, Pancreatic metastasisMother death 35 days post-partum |
| Olsen et al. [ref. 14] | MCN | – | – | 1st (6) | – | 2nd (18) | None reported |
| Simchuk et al. [ref. 15] | Adenocarcinoma | – | – | 2nd (16) | – | 2nd (16) | metastases |
| Sciscione et al. [ref. 16] | Neuroendocrine | 37 | Incidentally US finding (pancreatic mass) | 2nd (19) | – | 2nd (20) | Fetal death |
| Blackbourne et al. [ref. 17] | Adenocarcinoma | 32 | back pain, nausea, emesis, and dark urine | 2nd (14) | Intraoperative FNA | 2nd (17) | No complications |
| Ganepola et al. [ref. 18] | MCN | 37 | Abdominal pain | 1st (4) | Frozen section | 2nd (23) | No complications |
| Lopez-Tomassetti et al. [ref. 19] | MCN | 26 | abdominalpain and hyperemesis | 2nd (20) | Histological analysis of resected mass | 2nd (20) | No complications |
| Kato et al. [ref. 20] | MCN | 33 | abdominal distention | 2nd (15) | MRI- Intraoperative ultrasound | 2nd (20) | Pre-op IUGR |
| Marinoni et al. [ref. 8] | Adenocarcinoma | 38 | Epigastric pain | 3rd (27) | Biopsy by ERCP | Postpartum | Mother death 50 days post-partum |
| Lin Lin Su et al. [ref. 21] | Adenocarcinoma | 37 | intermittent epigastric pain | 2nd (22) | Ultrasound-guided liver biopsyconfirmed metastatic poorly differentiated adenocarcinoma,Consistent with a pancreatic primary. | 2nd | Termination of pregnancy |
| Ishikawa et al. [ref. 22] | MCN | 33 | Epigastric mass | 2nd (17) | MRI | Postpartum | No complications |
| Al Adnani et al. [ref. 23] | Adenocarcinoma | 27 | Prenatal care: HTN, fallen fetal growth | 3rd (30) | Pancreatic biopsy | – | IUGR, placental metastasis,Mother death 3 moths postpartum |
| Herring et al. [ref. 24] | MCN | 34 | Accidental finding of abdominal mass during routine follow up | 1st (3) | Histological analysis of resected mass | 2nd (17) | No complications |
| Ozden et al. [ref. 25] | MCN | 32 | Epigastric pain | 3rd (36) | Histological analysis of resected mass | 3rd (36) | Tumor rupture |
| Wiseman et al. [ref. 26] | MCN | 32 | LUQ pain with palpable mass | 2nd (15) | MRI- US guided drainage | 2nd (16) | Pre-op Intractable nausea |
| Hakamada et al. [ref. 27] | Anaplastic | 38 | Incidentally US finding | 1st trimester, 1st pregnancy | MRI- US | 2nd trimester, 2nd pregnancy | Intractable nausea, upper GI bleed (pre-op), local recurrence (post-op) |
| Ikuta et al. [ref. 28] | MCN | 30 | Left hypochondrial pain | 1st (10) | CT, – US guided drainage | Post-abortion | Missed abortion |
| Kamphues et al. [ref. 29] | Neuroendocrine | 32 | Arterial HTN | 2nd (19) | US, MRI | 2nd (19) | Splenic vein thrombosis renal artery compression (intra-op) |
| Kamphues et al. [ref. 29] | Neuroendocrine | 35 | Vomiting, weight loss | 2nd (16) | Needle biopsy | 2nd (18) | No complications |
| Kakoza et al. [ref. 11] | Adenocarcinoma | 40 | Epigastric pain, nausea, vomiting | 2nd (24) | Duodenal mucosal biopsy | Postpartum | Liver metastasis, mother death 6 months post-op |
| Asciutti et al. [ref. 30] | MCN | Epigastric pain + mass | 2nd (23) | – | Postpartum | Pancreatitis | |
| Onuma et al. [ref. 31] | Adenocarcinoma | 32 | frequent uterinecontractions | 3rd (30) | CT, Histological analysis of resected mass | 3rd (34) | Pre-op Gastric perforation |
| Naganuma et al. [ref. 32] | MCN | 32 | threat of premature labor | 3rd (33) | CT, Histological analysis of resected mass | 3rd (34) | Tumor rupture, local recurrence 6 months pos-op |
| Perera et al. [ref. 7] | Adenocarcinoma | 25 | Epigastric pain, nausea,One episode of emesis | 2nd (20) | MRCP, ERCP | No surgery, underwent chemotherapy | Mother Death |
| Lubner et al. [ref. 33] | Adenocarcinoma | 37 | nausea, vomiting,back pain, acholia, and dark colored urine | 2nd (16) | EUS with FNA | 2nd (18) | Death 12 months post diagnosis |
| Marci et al. [ref. 34] | Adenocarcinoma | 36 | Epigastric pain, vomiting, weight loss | 3rd (35) | US, CT | Postpartum | (PRE-OP) Acute renal failure |
| Feng et al. [ref. 35] | SPNs | 26 | Incidentally abdominal mass by US | 2nd (14) | US, MRI, needle biopsy | 2nd (14) | 3rd postoperative day pancreatic fistula occurred |
| Boyd et al. [ref. 36] | MCN | 21 | abdominal distention and fullness | 1st (10) | CT, MRI | 2nd (20) | No complications |
| Boyd et al. [ref. 36] | Adenocarcinoma | 29 | Emesis, epigastric pain | 3rd (37) | US, CT | Postpartum | 2 weeks post-op PE, iliac & femoral veins thrombosis, DIC, multiorgan system failure & death. |
| Boyd et al. [ref. 36] | Adenocarcinoma | 37 | right upper back pain, nausea and vomiting, alcoholic stools, and dark urine | 2nd (17) | US, ERCP, FNA | 2nd (19) | 4 months post-op liver metastasis, 1-year post-op death. |
| Liu et al. [ref. 37] | Adenocarcinoma | 31 | weight loss and progressive, positional dyspnea | 3rd trimester | US, CT, PET scan | 3rd (34) | Ovarian cyst rupture,(post-op) Pleural effusion, ascites |
| Tsuda et al. [ref. 38] | MCN | 28 | Referred as case of abdominal tumor | 1st (9) | US, MRI, | 2nd (18) | Mild glucose intolerance post-op. |
| Tica et al. [ref. 39] | MCN | 27 | abnormal sonogram | 3rd (29) | MRI | postpartum | No complications |
| Huang et al. [ref. 40] | SPNs | 29 | Epigastric pain, backache, nausea, and vomiting | 2nd (19) | US, MRI | 2nd (19) | Tumor rupture |
| MacDonald et al. [ref. 41] | SPNs | 23 | Incidentally abdominal mass by US | 2nd (14) | US, MRI | 2nd (18) | No complications |
| Labarca-Acosta et al. [ref. 42] | Adenocarcinoma | 35 | Vomiting, pain in the left epigastrium and hypochondrium, general weakness and weight loss. | 16 weeks | Fine-needle biopsy | – | Maternal death |
| Aker et al. [ref. 43] | Adenocarcinoma | 27 | Right upper quadrant pain, nausea,and vomiting. | 2nd (26) | ascites cytology | Post-partum | Fetal deathMetastasis to placenta, hepatic, supraclavicular lymph node |
| Davis et al. [ref. 44] | Adenocarcinoma | 34 | abdominal pain and failure to gain weight appropriately in pregnancy. | 2nd (26) | Pathologyon fine-needle aspiration of the pancreatic head mass confirmedpancreatic adenocarcinoma | – | Death 4 months after C-section |
| Aynioglu et al. [ref. 45] | Adenocarcinoma | 36 | recurrent severe abdominal pain radiating to the back, jaundice,nausea, and vomiting. | 3rd (28) | Histopathological analysis | Post-partum | No complication |
MCN = mucinous cystic neoplasm; HELLP = hemolysis& elevated liver enzymes level and low platelet; IUGR = intrauterine growth restriction; SPNs = Solid-pseudo papillary neoplasms.
In case of pregnancy, diagnosing of ampullary adenocarcinoma can be challenging. The way it does that is by making alarming symptoms of malignancy through the physiological changes of pregnancy. As jaundice can develop frequently in the 3rd trimester due to benign intrahepatic cholestasis, which can be misleading initially as in our case. Furthermore, other symptoms that adenocarcinoma of ampulla of Vater cause like abdominal discomfort, nausea & vomiting also can happen in normal pregnancy. Another challenge is that the gravid uterus that decreases the sensitivity of non-invasive imaging modalities [ref. 5]. The reported cases demonstrate a variety of gestational ages at presentation, which indicated a clinical difficulty in diagnosis. Unlike our case, most of the reported cases presented at 2nd trimester.
There are different radiological modalities that can aid in obtaining a confirmatory diagnosis and staging of ampullary carcinoma but not all of these can be used freely during pregnancy such as ERCP or CT scan, due to the risk of radiation exposure to the fetus [ref. 7]. The main diagnostic modalities for pregnant women are ultrasonography and MRCP. The ultrasonography is mainly used to identify the presence of biliary dilation & obstruction while MRCP can visualize the mass [ref. 8].
In our case, the MRCP revealed an ampullary mass measuring 2 cm with no vascular involvement. ERCP is used during pregnancy for diagnosis by obtaining a biopsy, stenting for biliary draining and to prepare the patient for surgery or if the tumor is not operable. To minimize the risk radiation exposure to the fetus the patient should have a lead shield in place as we did in our case. Biochemically, an elevated carbohydrate antigen 19-9 (CA 19-9) level may help to guide the diagnosis towards an ampullary adenocarcinoma [ref. 4]. Nevertheless, US and MRCP were the main diagnostic modalities reported in the literature.
Management wise, surgical resection is usually done by a Whipple procedure (Pancreaticoduodenectomy) followed by an adjuvant therapy is the standard of care for early stage disease. Such surgery carries a risk of many complications. One important complication to consider is pancreatic fistula or leak with a reported incidence ranging from 2 to 40% [ref. 9].
To decrease the risk of such complication, intraoperatively a pancreatic duct stent can be used. Adaptation of this technique to protect the anastomotic site & decrease the chance of any leak have led good to outcome of such procedure in some centers [ref. 10].
Surgical intervention is challenging in pregnant patients, the appropriate time to intervene depends on the gestational age at the time of diagnosis and the stage of the disease. The challenges that might be encountered in first trimester, is the risk of spontaneous abortion and the best strategy is to abort the pregnancy. While in the second trimester induced delivery can’t be applied as gestational age is not compatible with life and it is the most dangerous time to put the patient on chemotherapy. In the third trimester, the large size of the uterus is another challenge especially intraoperatively [ref. 11].
In our patient, the obstetrician induced labor at week 34 of gestation & Whipple procedure was done afterwards and the major challenge was a presence of a bulky uterus during the surgery.
Conclusion
Obtaining a diagnosis of ampullary adenocarcinoma in pregnant patients in their third trimester & managing them can be challenging due to; 1) the overlapping symptoms between it & the physiological changes of pregnancy, 2) the limitation of using the appropriate diagnostic modalities in order to avoid radiation exposure to the fetus, 3) the intraoperative technical difficulties 2ry to the gravid uterus. Yet, a delayed viable delivery followed by a definitive surgery in the form of pancreaticoduodenectomy is still achievable with a multi-disciplinary approach & good perioperative preparation in early stage disease.
Conflicts of interest
None.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Ethical approval
Case reports are exempted from ethical approval according to our institution policies.
Consent
Written informed consent was obtained from the patient for publication of this case report.
Author contribution
Abdullah Saleh AlQattan: study design, data collection, writing the paper, reviewing and editing the case report.
Feras Ahmed Alkuwaiti: study design, data collection, writing the paper.
Elham Saleh Alghusnah: study concept, reviewing article, correction and editing of the case report.
Shoukat Ahmad Bojal: study concept, reviewing article, correction and editing of the case report.
Mohammed Saad Alqahtani: study concept, reviewing the final manuscript of the case report, final approval.
Registration of research studies
N/A.
Guarantor
Dr. Mohammed Saad Alqahtani.
Provenance and peer review
Not commissioned, externally peer-reviewed.
References
- R.A. Agha, M.R. Borrelli, R. Farwana, K. Koshy, A. Fowler, D.P. Orgill, For the SCARE Group. The SCARE 2018 statement: updating consensus Surgical CAse REport (SCARE) guidelines. Int. J. Surg., 2018. [PubMed]
- Surveillance, Epidemiology, and End Results (SEER). 2019
- Paredes G. Reaño. Pancreatoduodenectomy for ampullary adenocarcinoma and re-intervention for ganglinonar recurrency. Rev. Gastroenterol. Peru, 2014. [PubMed]
- J. Askew, S. Connor. Review of the investigation and surgical management of resectable ampullary adenocarcinoma. HPB, 2013. [PubMed]
- O. Haddad, G. Porcu-Buisson, R. Sakr, B. Guidicelli, Y.P. Letreut, M. Gamerre. Diagnosis and management of adenocarcinoma of the ampulla of Vater during pregnancy. Eur. J. Obstet. Gynecol. Reprod. Biol., 2005. [PubMed]
- J.M. Porcel, J. Ordi, L. Castells, I. Farran. Probable pancreatic cancer in a pre-eclamptic patient. Eur. J. Obstet. Gynecol. Reprod. Biol., 1992. [PubMed]
- D. Perera, R. Kandavar, E. Palacios. Pancreatic adenocarcinoma presenting as acute pancreatitis during pregnancy: clinical and radiologic manifestations. J. State Med. Soc., 2011
- E. Marinoni, T. Di Netta, L. Caramanico, B. Tomei, M. Moscarini, R. Di Iorio. Metastatic pancreatic cancer in late pregnancy: a case report and review of the literature. J. Matern. Fetal Neonatal. Med., 2006. [PubMed]
- Stylianos Katsaragakis. A new pancreaticojejunostomy technique: a battle against postoperative pancreatic fistula. World J. Gastroenterol.: WJG, 2013. [PubMed]
- Giorgio Romano. Whipple’s pancreaticoduodenectomy: surgical technique and perioperative clinical outcomes in a single center. Int. J. Surg., 2015. [PubMed]
- R.M. Kakoza, C.M. Vollmer, K.E. Stuart, T. Takoudes, D.W. Hanto. Pancreatic adenocarcinoma in the pregnant patient: a case report and literature review. J. Gastrointest. Surg., 2009. [PubMed]
- B.M. Smithers, C. Welch, P. Goodall. Cystadenocarcinoma of the pancreas presenting in pregnancy. Br. J. Surg., 1986
- C. Baiocchi, G. Landonio, M. Majno, E. Minola, F. Scanzi, E. Ghislandi. Pancreatic cystadenocarcinoma and pregnancy: a case report. Tumori, 1990. [PubMed]
- M.E. Olsen, M.S. Greer, T.A. Feintuch. Pancreatic mucinous cystadenoma during pregnancy. Am. J. Gynecol. Health, 1993. [PubMed]
- E.J. Simchuk, J.P. Welch, R. Orlando. Antepartum diagnosis of pancreatic carcinoma: a case report. Conn. Med., 1995. [PubMed]
- A.C. Sciscione, J.B. Villeneuve, H.A. Pitt, T.R. Johnson. Surgery for pancreatic tumors during pregnancy: a case report and review of the literature. Am. J. Perinatol., 1996. [PubMed]
- L.H. Blackbourne, R.S. Jones, C.J. Catalano, J.C. Iezzoni, F.J. Bourgeois. Pancreatic adenocarcinoma in the pregnant patient: case report and review of the literature. Cancer, 1997. [PubMed]
- G.A. Ganepola, A.Y. Gritsman, N. Asimakopulos, A. Yiengpruksawan. Are pancreatic tumors hormone dependent? A case report of unusual, rapidly growing pancreatic tumor during pregnancy, its possible relationship to female sex hormones, and review of the literature. Am. Surg., 1999. [PubMed]
- E.M. Lopez-Tomassetti Fernandez, A. Martin Malagon, I. Arteaga Gonzalez, J.R. Muniz Montes, H. Diaz Luis, F. Gonzalez Hermoso, A. Carrillo Pallares. Mucinous cystic neoplasm of the pancreas during pregnancy: the importance of proper management. J. Hepatobiliary Surg., 2005
- M. Kato, K. Kubota, J. Kita. Huge mucinous cystadenoma of the pancreas developing during pregnancy: a case report. Pancreas, 2005. [PubMed]
- L. Lin Su, A. Biswas, A. Wee, W. Sufyan. Placental metastases from pancreatic adenocarcinoma in pregnancy. Acta Obstet. Gynecol. Scand., 2006. [PubMed]
- K. Ishikawa, T. Hirashita, H. Kinoshita, M. Kitano, S. Matsuo, T. Matsumata, S. Kitano. Large mucinous cystadenoma of the pancreas during pregnancy: report of a case. Surg. Today, 2007. [PubMed]
- M. Al-Adnani, L. Kiho, I. Scheimberg. Maternal pancreatic carcinoma metastatic to the placenta: a case report and literature review. Pediatr. Dev. Pathol., 2007. [PubMed]
- A.A. Herring, M.B. Graubard, S.I. Gan, S.D. Schwaitzberg. Mucinous cystadenocarcinoma of the pancreas during pregnancy. Pancreas, 2007. [PubMed]
- S. Ozden, B. Haliloglu, E. Ilter, F.T. Akin, A. Kebudi, O. Peker. An extremely rare cause of acute abdomen in pregnancy: ruptured pancreatic mucinous cystadenocarcinoma. Pancreas, 2007. [PubMed]
- J.E. Wiseman, M. Yamamoto, T.D. Nguyen, J. Bonadio, D.K. Imagawa. Cystic pancreatic neoplasm in pregnancy: a case report and review of the literature. Arch. Surg., 2008. [PubMed]
- K. Hakamada, T. Miura, A. Kimura, M. Nara, Y. Toyoki, S. Narumi, M. Sasak. Anaplastic carcinoma associated with a mucinous cystic neoplasm of the pancreas during pregnancy: report of a case and a review of the literature. World J. Gastroenterol., 2008. [PubMed]
- S. Ikuta, T. Aihara, C. Yasui. Large mucinous cystic neoplasm of the pancreas associated with pregnancy. World J. Gastroenterol., 2008. [PubMed]
- C.H. Kamphues, C. Rocken, P. Neuhaus, U.P. Neumann. Non-functioning, malignant pancreatic neuroendocrine tumour (PNET): a rare entity during pregnancy. Langenbecks Arch. Surg., 2009. [PubMed]
- S. Asciutti, T.T. Kanninen, G. Clerici, E. Nardi, D. Castellani, G.C. DIR, C. Clerici. Acute pancreatitis with a mucinous cystoadenoma of the pancreas in pregnancy. Anticancer Res., 2010. [PubMed]
- T. Onuma, Y. Yoshida, T. Yamamoto, F. Kotsuji. Diagnosis and management of pancreatic carcinoma during pregnancy. Obstet. Gynecol., 2010. [PubMed]
- S. Naganuma, K. Honda, S. Noriki. Ruptured mucinous cystic neoplasm with an associated invasive carcinoma of pancreatic head in a pregnant woman: report of a case and review of literature. Pathol. Int., 2011. [PubMed]
- S. Lubner. A 37-year-old pregnant woman with pancreatic adeno-carcinoma treated with surgery and adjuvant chemotherapy: a case report and literature review. J. Gastrointest. Oncol., 2011. [PubMed]
- R. Marci. Pancreatic cancer with liver metastases in a pregnant patient: case report and review of the literature. Clin. Exp. Obstet. Gynaecol., 2012
- Ji Feng Feng. Solid pseudopapillary tumor of the pancreas in a pregnant woman. Acta Gastro-Enterol. Belg., 2011
- C.A. Boyd, J. Benarroch-Gampel, G. Kilic, E.J. Kruse, S.M. Weber, T.S. Riall. Pancreatic neoplasms in pregnancy: diagnosis, complications, and management. J. Gastrointest. Surg., 2012. [PubMed]
- Stephen V. Liu. Metastatic pancreatic cancer during pregnancy presenting as pseudo-Meigs’ syndrome. Gynecol. Surg., 2012
- Hiroyuki Tsuda. Mucinous cystic neoplasm of the pancreas with severe dysplasia during pregnancy: case report and review of the literature. Taiwan. J. Obstet. Gynecol., 2012. [PubMed]
- Andrei Adrian Tica. Large pancreatic mucinous cystic neoplasm during pregnancy: what should be done?. Gynecol. Obstet. Invest., 2013. [PubMed]
- Shih-Chiang Huang. Spontaneous rupture of solid pseudopapillary neoplasm of the pancreas during pregnancy. Obstet. Gynecol., 2013. [PubMed]
- Frank MacDonald. Surgical therapy of a large pancreatic solid-pseudopapillary neoplasm during pregnancy. BMJ Case Rep., 2014
- M. Labarca-Acosta, E. Reyna-Villasmil, J. Aragón-Charris, J. Santos-Bolívar. Pancreatic adenocarcinoma during pregnancy. Cir. Esp. (Engl. Ed.), 2015
- S.Ş Aker, D.C. Katlan, T. Yüce, F. Söylemez. Placental, hepatic, and supraclavicular lymph node metastasis in pancreatic adenocarcinoma during pregnancy: a case report. Turk. J. Obstet. Gynecol., 2016. [PubMed]
- J. Davis, S. Bashir, H. Wubneh, M.L. Borum. Metastatic pancreatic adenocarcinoma during pregnancy. ACG Case Rep. J., 2016
- Öner Aynioglu. 2017
