Poorly differentiated adenocarcinoma with signet-ring cells in duodenal papilla: a case report
Abstract
An 82-year-old woman with common bile duct (CBD) dilatation observed during routine ultrasonography was referred to our hospital. Preliminary blood tests revealed elevated levels of hepatobiliary enzymes. Computed tomography (CT) scan showed lower bile duct wall thickening and enhancement. Esophagogastroduodenoscopy revealed mildly swollen papilla of Vater, without ulceration. Endoscopic retrograde cholangiography demonstrated that the CBD was grossly dilated with a constriction in the lower part. The final diagnosis indicated poorly differentiated adenocarcinoma of duodenal papilla with signet-ring cells; pT3N0M0, stage IIA (Unio Internationalis Contra Cancrum, 7th edition), for which subtotal stomach-preserving pancreaticoduodenectomy (SSPPD) was performed. This case is quite rare, and the surgery resulted in a desirable outcome. The patient has been disease-free for 5 years since the surgery.
Article type: Case Report
Keywords: Duodenal papillary cholangiocarcinoma, Poorly differentiated adenocarcinoma, Signet-ring cell
Affiliations: 0004 0377 5215grid.413779.fDepartment of Surgery, Anjo Kosei Hospital, 28 Higashihirokute, Anjo-cho, Anjo, Aichi 446-8602 Japan
License: © The Author(s). 2017 CC BY 4.0 Open AccessThis article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Article links: DOI: 10.1186/s40792-017-0287-1 | PubMed: 28097624 | PMC: PMC5241256
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (944 KB)
Background
Most duodenal papillary carcinomas (DPCs) are well differentiated [ref. 1]. Poorly differentiated DPCs are rare and have unfavorable prognosis. Signet-ring cells in the duodenal papilla are an indication of poor prognosis. Here, we report a case of poorly differentiated DPC with signet-ring cells that had favorable outcome.
Case presentation
An 82-year-old woman with common bile duct (CBD) dilatation, as observed during routine ultrasonography, was referred to our hospital. She was asymptomatic. Laboratory test results were aspartate aminotransferase level, 278 IU/L; alanine aminotransferase level, 184 IU/L; alkaline phosphatase level, 1877 IU/L; total bilirubin level, 0.42 mg/dL; amylase level, 47 IU/L; and presence of routine inflammatory markers. Laboratory tumor marker levels, carcinoembryonic antigen, and carbohydrate antigen 19–9 levels were normal.
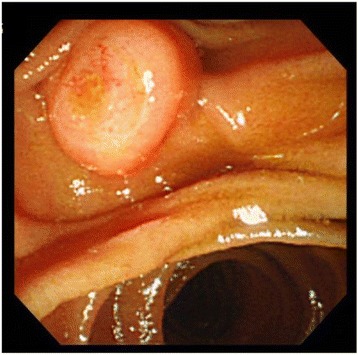
Esophagogastroduodenoscopy (EGD) revealed mildly swollen papilla of Vater, without any mucosal erosion (Fig. 1).
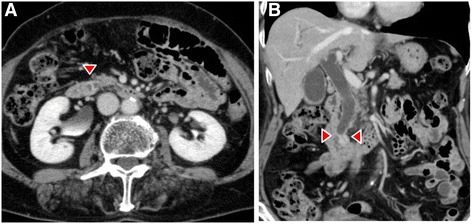
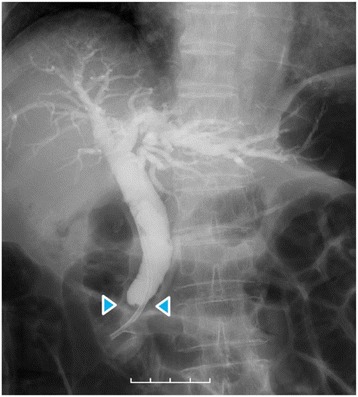
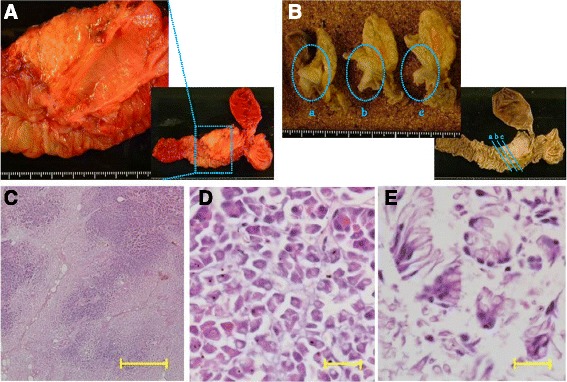
Dynamic computed tomography (CT) showed bile duct wall thickening that was enhanced in the lower part of the CBD (Fig. 2). Endoscopic retrograde cholangiography (ERC) demonstrated abrupt obstruction of the lower CBD (Fig. 3). Histological examination of biopsy specimens from the lower CBD showed adenocarcinoma with signet-ring cells. We diagnosed extrahepatic cholangiocarcinoma and performed subtotal stomach-preserving pancreaticoduodenectomy (SSPPD). The surgery lasted 348 min, and the total blood loss was 525 mL. Histopathology report suggested atypical epithelial cells exhibiting infiltrative growth, with fibrosis of the duodenal papilla (Fig. 4c). Tumor cells displayed intracytoplasmic mucus deposition, crescent-shaped nucleoli (Fig. 4d, e) extensions along the lower CBD, and invasion of pancreatic parenchyma (3 mm). AcbBd, exposed protruded type, 22 × 16 mm, por2/sig, pT3a, sci, INFc, ly1, v1, ne1, pN0, pHM0, pPM0, pEM0, PV0, A0, R0, pStage IIA according to the Japanese Classification on Cancer of the biliary tract [ref. 2] and pT3N0M0 stage IIA in accordance with Union for Internationatinal Cancer Control, 7th edition. The final diagnosis was poorly differentiated adenocarcinoma with signet-ring cell of DPC. The patient developed pancreatic fistula postoperatively (ISGPF grade B), with no other complications and was discharged on day 37 postsurgery. She has remained disease-free for 5 years since the surgery.
Discussions
DPC is a rare clinical entity, occurring in less than 6 per million people annually. It represents 0.2% of all gastrointestinal cancers and accounts for only 6% of all cancers developing in the periampullary region [ref. 3]. The Japanese Society of Hepato-Biliary-Pancreatic Surgery reported that the most common histological type of DPC is well-differentiated adenocarcinoma (36.3%), followed by papillary adenocarcinoma (27.6%) and moderately differentiated adenocarcinoma (25.0%). Poorly differentiated adenocarcinoma is rare and accounts for 5.5% of all DPC cases [ref. 4]. Signet-ring cells are also extremely uncommon histologic types at this site and arise mainly from the stomach. Signet-ring cell carcinomas (SRCC) are characterized by signet-ring cells with intracytoplasmic mucin occupying more than 50% of the tumor [ref. 5]. In our case, the proportion of signet-ring cells to whole carcinoma was about 10–15%, so we diagnosed poorly differentiated adenocarcinoma with signet-ring cells, and not SRCC. Furthermore, poorly differentiated DPCs are rare, and on a PubMed search using key words like Vater, poorly differentiated, and signet, only ten well-documented cases were found (Table 1) [ref. 6–ref. 15]. The 11 cases, including our case, consisted of three men and eight women with ages ranging from 43 to 83 years (mean 62 years). Jaundice was the most common symptom (54.5%). Two cases of DPC with jaundice survived for more than 5 years after surgery, while rapid metastasis [ref. 13] was observed in others, as was disseminating carcinomatosis without jaundice [ref. 6].
Table 1: Published cases of poorly differentiated adenocarcinoma in duodenal papilla with signet-ring cells
| Author | Year | Age (years) | Sex | Complaint | TNM stagea | Distant metastasis | Treatment | Histology | Follow-up (months) | Outcome |
|---|---|---|---|---|---|---|---|---|---|---|
| Nabeshima [ref. 5] | 2003 | 49 | M | Purpura (DICb) | T3NxM1 stage IV | Lung, bone marrow | Chemotherapy | Por/sig | 12 | Died |
| Eriguchi[ref. 6] | 2003 | 83 | M | Jaundice | T3N0M0 stage IIA | – | PD | Sig | 12 | Alive |
| Ramia [ref. 7] | 2004 | 67 | F | Jaundice | T2N0M0 stage IB | – | PD | Sig | 12 | Alive |
| Akatsu [ref. 8] | 2007 | 43 | F | Jaundice | T2N0M0 stage IB | – | PD | Sig | 90 | Alive |
| Bloomston [ref. 9] | 2006 | 58 | F | Jaundice | T2N0M0 stage IB | – | PD | Sig | 134 | Alive |
| Ishibashi [ref. 10] | 2009 | 59 | M | Abdominal pain | T3N0M0 stage IIA | – | PD | Sig | 18 | Died |
| Ogata [ref. 11] | 2010 | 42 | F | Jaundice | T4N1M0 stage III | – | SSPPD | Sig | 6 | Alive |
| Matsuoka [ref. 12] | 2013 | 61 | F | Left visual disturbance | TxNxM1 stage IV | Brain | Gross total resection of brain tumor | Sig | 3 | Died |
| Acharya [ref. 13] | 2013 | 78 | F | Jaundice | T3N0M0 stage IIA | – | PD | Sig | 6 | Alive |
| Wakasugi [ref. 14] | 2015 | 59 | F | Elevated transaminase | T3N1M1 stage IV | Paraaortic node | PD | Sig | 7 | Alive |
| Our case | 2016 | 82 | F | – | T3N0M0 stage IIA | – | SSPPD | Por/sig | 60 | Alive |
aInternational Union Against Cancer TNM classification
bDisseminated intravascular coagulation
The 5-year survival rate in patients with DPC after radical resection is 30–68% [ref. 16–ref. 19]. In most cases, the prognosis of DPC is better than biliary and pancreatic carcinomas. Owing to their anatomical location, ampullary tumors become clinically apparent earlier because of biliary or pancreatic duct occlusion. Since DPC is often diagnosed at an early stage, surgical resection has a higher probability of success. The average recurrence interval of DPC was 13–22 months [ref. 19, ref. 20], and recurrence includes liver metastasis, local recurrence, peritoneal metastasis, and bone metastasis [ref. 19–ref. 21]. The important factors affecting the prognosis are lymph node status, depth of tumor invasion, and degree of tumor differentiation [ref. 22, ref. 23]. Patients with pancreatic infiltration tend to have early recurrences [ref. 24]. Lymph node status is a significant predictive factor in liver metastasis, and a 5-year survival rate in patients with lymph node-positive status is 19.1% and of those with node-negative status is 63.7% [ref. 21]. Immunohistochemical staining patterns of cytokeratin and mucin allow further classification of SRCC to intestinal, pancreatobiliary, gastric, and mixed type. SRCC patients with intestinal type are favorable, and those with mixed type reveal poor prognosis.
In our case, although the tumor consisted of poorly differentiated adenocarcinoma with signet-ring cells and had infiltrated into the pancreatic parenchyma pT3, both of which indicating poor prognosis, the outcome was desirable. She was diagnosed with DPC incidentally in the health checkup before having jaundice; therefore, the ratio of signet-ring cells was relatively low. Similar reports are very few for DPC [ref. 25].
It is well known that surgical resection is the only curative treatment for DPC. No evidence-based chemotherapy regimens exist either for the treatment of unresectable cancers or for postoperative adjuvant therapy; therefore, we simply followed up the patient. Fortunately, the patient is doing well, without any signs of tumor recurrence since the last 5 years.
Conclusions
Here, we have presented favorable results in a case of poorly differentiated DPC with signet-ring cells. It is however important to study some more cases with similar outcomes to establish its characteristics.
References
- T Terada. Pathologic observations of the duodenum in 615 consecutive duodenal specimens in a single Japanese hospital: II. malignant lesions. Int J Clin Exp Pathol, 2012. [PubMed]
- 2.Japanese Society of Hepato-Biliary-Pancreatic Surgery. General Rules for Clinical and Pathological Studies on Cancer of the Biliary Tract (in Japanese), 6th ed. Tokyo: Kanahara shuppan; 2013. p. 31.
- JR Howe, DS Klimstra, RD Moccia, KC Conlon, MF Brennan. Factors predictive of survival in ampullary carcinoma. Ann Surg, 1998. [DOI | PubMed]
- T Nagakawa, M Kayahara. Tandogan chiryoseiseki ga oshieru tandogan no sindan to chiryo no arikata (in Japanese), 2005
- L Li, QH Chen, JD Sullivan, FU Breuer. Signet-ring cell carcinoma of the ampulla of Vater. Ann Clin Lab Sci, 2004. [PubMed]
- S Nabeshima, Y Kishihara, A Nabeshima, S Yamaga, M Kinjo, S Kashiwagi, J Hayashi. Poorly differentiated adenocarcinoma with signet-ring cells of the Vater’s ampulla, without jaundice but with disseminated carcinomatosis. Fukuoka Igaku Zasshi, 2003. [PubMed]
- N Eriguchi, S Aoyagi, A Jimi. Signet-ring cell carcinoma of the ampulla of Vater: report of a case. Surg Today, 2003. [DOI | PubMed]
- JM Ramia, A Mansilla, J Villar, K Muffak, D Garrote, JA Ferron. Signet-ring-cell carcinoma of the Vater’s ampulla. J Pancreas, 2004
- T Akatsu, K Aiura, S Takahashi, K Kameyama, M Kitajima, Y Kitagawa. Signet-ring cell carcinoma of the ampulla of Vater: report of a case. Surg Today, 2007. [DOI | PubMed]
- M Bloomston, M Walker, WL Frankel. Radical resection in signet ring carcinoma of the ampulla of Vater: report of an 11-year survivor. Am Surg, 2006. [PubMed]
- Y Ishibashi, Y Ito, K Omori, K Wakabayashi. Signet ring cell carcinoma of the ampulla of Vater. A case report. J Pancreas, 2009
- S Ogata, A Kimura, K Hatsuse, J Yamamoto, H Shimazaki, K Nakanishi, T Kawai. Poorly differentiated adenocarcinoma with signet-ring cell carcinoma of the extrahepatic bile duct in a 42-year-old Japanese female: a case report. Acta Med Okayama, 2010. [PubMed]
- G Matsuoka, H Waniguchi, S Suzuki, K Honda, Y Okada. A brain metastasis from ampullary carcinoma: a case report. No Shinkei Geka, 2013. [PubMed]
- MN Acharya, N Panagiotopoulous, P Cohen, R Ahmad, LR Jiao. Poorly-differentiated signet-ring cell carcinoma of the ampulla of Vater: report of a rare malignancy. J Pancreas, 2013
- M Wakasugi, M Tanemura, K Furukawa, M Murata, M Miyazaki, M Oshita, K Yoshida, H Yasuoka, H Akamatsu. Signet ring cell carcinoma of the ampulla of vater: report of a case and a review of the literature. Int J Surg Gase Rep, 2015. [DOI]
- CJ Yeo, JL Cameron, TA Sohn, KD Lillemoe, HA Pitt, MA Talamini, RH Hruban, SE Ord, PK Sauter, J Coleman, ML Zahurak, LB Grochow, RA Abrams. Six hundred fifty consecutive pancreaticoduodenectomies in the 1990s: pathology, complications, and outcomes. Ann Surg, 1997. [DOI | PubMed]
- TC Böttger, J Boddin, A Heintz, T Junginger. Clinicopathologic study for the assessment of resection for ampullary carcinoma. World J Surg, 1997. [DOI | PubMed]
- V Bettschart, MQ Rahman, FJ Engelken, Madhavan, RW Parks, OJ Garden. Presentation, treatment and outcome in patients with ampullary tumours. Br J Surg, 2004. [DOI | PubMed]
- T Todoroki, N Koike, Y Morishita, T Kawamoto, N Ohkohchi, J Shoda, Y Fukuda, H Takahashi. Patterns and predictors of failure after curative resections of carcinoma of the ampulla of Vater. Ann Surg Oncol, 2003. [DOI | PubMed]
- M Kayahara, T Nagakawa, T Ohta, H Kitagawa, I Miyazaki. Surgical strategy for carcinoma of the papilla of Vater on the basis of lymphatic spread and mode of recurrence. Surgery, 1997. [DOI | PubMed]
- HP Hsu, TM Yang, YH Hsieh, YS Shan, PW Lin. Predictors for patterns of failure after pancreaticoduodenectomy in ampullary cancer. Ann Surg Oncol, 2007. [DOI | PubMed]
- RD Kim, PS Kundhal, ID Mcgilvray, MS Cattral, B Taylor, B Langer, DR Grant, G Zogopoulos, SA Shah, PD Greig, S Gallinger. Predictors of failure after pancreaticoduodenectomy for ampullary carcinoma. J Am Coll Surg, 2006. [DOI | PubMed]
- MA Talamini, RC Moesinger, HA Pitt, TA Sohn, RH Hruban, KD Lillemoe, CJ Yeo, JL Cameron. Adenocarcinoma of the ampulla of Vater. A 28-year experience. Ann Surg, 1997. [DOI | PubMed]
- T Ajiki, H Onoyama, T Kamigaki, N Miyazaki, M Yamamoto, Y Saito. A clinicopathological study of resected cases of carcinoma of the papilla of Vater or lower bile duct. J Jpn Surg Assoc (in Japanese), 1994
- X Wen, W Wu, B Wang, H Yao, X Teng. Signet ring cell carcinoma of the ampulla of Vater: immunophenotype and differentiation. Oncol Lett, 2014. [PubMed]
