Physiological functions and pharmacological targeting of transient receptor potential channels
Abstract
Transient receptor potential (TRP) channels represent an extensive and diverse protein family fulfilling salient roles as versatile cellular sensors and effectors. The pivotal role of TRP and related ion channels in sensory processes has been well documented. Over the last few years, a new concept has emerged that TRP proteins control an exceptionally broad spectrum of homeostatic physiological functions such as maintenance of body temperature, blood pressure, transmitter release from neurons, mineral and energy homeostasis, and reproduction. This notion is further supported by more than 20 hereditary human diseases in areas as diverse as neurology, cardiology, hematology, pulmonology, nephrology, dermatology, and urology. Most TRP channel-related human disorders impinge on development, metabolism, and other homeostatic functions. The remarkable diversity of pathologies caused by TRP channel dysfunction underscores these proteins’ broad spectrum of roles in vivo. Here, we provide a comprehensive overview of our progress in the identification, characterization, and clinical relevance of pharmacological agents targeting mammalian TRP channels.
Significance Statement:
Accumulating evidence links transient receptor potential (TRP) channels to various human diseases and highlights TRPs as the most appealing pharmacological targets. The review provides an overview of this quickly developing research area, focusing on identified pharmacological modulators of mammalian TRP channels.
Affiliations: Walther-Straub Institute of Pharmacology and Toxicology, Ludwig Maximilian University of Munich, Munich, Germany; Immunology, Infection and Pandemic Research IIP, Fraunhofer Institute for Translational Medicine and Pharmacology ITMP, Frankfurt, Germany; Department of Pharmacology, University of Oxford, Oxford, United Kingdom; Rudolf-Boehm Institute of Pharmacology and Toxicology, Leipzig University, Leipzig, Germany; Renal Division, Department of Medicine, Medical Center, Faculty of Medicine, University of Freiburg, Freiburg, Germany; Institute of Pharmacy, Clinical Pharmacy, University of Regensburg, Regensburg, Germany; Comprehensive Pneumology Center, a member of the German Center for Lung Research (DZL), Munich, Germany
License: © 2025 The Author(s) CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.pharmr.2025.100089 | PubMed: 41118703 | PMC: PMC12799467
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (7.9 MB)
Introduction
The transient receptor potential (TRP) gene superfamily represents a large, evolutionarily conserved group of ion channels initially identified in a mutant strain of Drosophila melanogaster that displayed an abnormal response to light illumination.ref. bib1 The mutant flies exhibited a rapid decay in the light-induced electroretinogram response of photoreceptor cells, referred to as transient receptor potential, to distinguish it from the sustained receptor potential observed in wild-type (WT) flies.ref. 2., ref. 3., ref. 4. Subsequent genetic and molecular investigations unveiled a new type of ion channel, now known as the TRP channel.ref. bib5
A systematic search for homologous proteins led to the discovery of TRP channels across a diverse array of eukaryotes, including algae, fungi, choanoflagellates, invertebrates, and mammals.ref. bib6 In humans, 27 TRP proteins are known, which are subdivided into 6 families (Table 1): canonical TRP channels (TRPCs), vanilloid receptor and related TRP channels (TRPVs), melastatin-like TRP channels (TRPMs), ankyrin domain-enriched TRP channels (TRPAs), mucolipin-related TRP channels (TRPMLs), and polycystic kidney disease-related TRP proteins (TRPPs).ref. bib7,ref. bib8
Table 1: Nomenclature, structural data, and expression profiles of TRP channels
| Gene | Protein and UniProt Entya | Structures in PDBb | Expression Pattern |
|---|---|---|---|
| TRPCs | |||
| TRPC1 | TRPC1 (TRP1); P48995 | only as TRPC1/4 heteromer: 8WPL, 8WPM | ubiquitous |
| TRPC2 | TRPC2 (TRP2) pseudogene in humans and Old-World monkeys | n.d. | VNO of rodents and other macrosmatic mammals |
| TRPC3 | TRPC3 (TRP3); Q13507 | 7DXB, 7DXC, 7DXD, 7DXE, 6DJR, 6CUD, 5ZBG | brain, heart, lung, blood vessels prostate, placenta, kidney, testis |
| TRPC4 | TRPC4 (TRP4, CCE1); Q9UBN4 | 7B0J, 6G1K, 7B05, 7B0S, 7B16, 6JZO, 5Z96, 8WPN, 7B1G; as TRPC1/4 heteromer: 8WPL, 8WPM | high levels in brain and placenta, lower levels in heart, pancreas, kidney, endothelium |
| TRPC5 | TRPC5 (TRP5, CCE2); Q9UL62 | 7E4T, 7D4P, 7D4Q, 7WDB, 7X6C, 8GVW, 7X6I, 8GVX, 6AEI, 6YSN | high levels in brain, lower levels in kidney, blood vessels, liver, stomach |
| TRPC6 | TRPC6 (TRP6); Q9Y210 | 7DXF, 7DXG, 6UZB, 6UZA, 5YX9, 7A6U | placenta, lung, blood vessels, spleen, ovary, small intestine, neutrophils, podocytes |
| TRPC7 | TRPC7 (TRP7); Q9HCX4 | n.d. | hypophysis, kidney, heart, lung, blood vessel, eye, spleen, testis |
| TRPVs | |||
| TRPV1 | TRPV1 (VR1, OTRPC1); Q8NER1 | 5IRX, 5IRZ, 5IS0, 7L2H, 7L2I, 7L2J, 7L2K, 7L2L, 7L2M, 7L2N, 7L2O, 7L2P, 7L2R, 7L2S, 7L2T, 7L2U, 7L2V, 7L2W, 7L2X, 7LP9, 7LPA, 7LPB, 7LPC, 7LPD, 7LPE, 7LQY, 7LQZ, 7LR0, 7MZ5, 7MZ6, 7MZ7, 7MZ9, 7MZA, 7MZB, 7MZC, 7MZD, 7MZE, 7RQU, 7RQV, 7RQW, 7RQX, 7RQY, 7RQZ, 8GF8, 8GF9, 8GFA, 8JQR, 8T0C, 8T0E, 8T0Y, 8T10, 8T3L, 8T3M, 8U2Z, 8U30, 8U3A, 8U3C, 8U3J, 8U3L, 8U43, 8U4D, 8X94 | small-to medium diameter DRG and trigeminal ganglion sensory neurons, brain neurons, astrocytes and microglia |
| TRPV2 | TRPV2 (VRL-1, OTRPC2); Q9Y5S1 | 6OO3, 6OO4, 6OO5, 6OO7, 7XEM, 7XEO, 7XER, 7XEU, 7XEV, 7XEW, 7YEP, 6BWJ, 6BWM, 5AN8, 8SLX, 8SLY, 8FFL, 8FFM, 8FFN, 8FFQ, 5HI9, 6BO4, 6BO5, 6U84, 6U86, 6U88, 6U8A, 7N0M, 7N0N, 7T37, 7T38, 7ZJD, 7ZJE, 7ZJG, 7ZJH, 7ZJI, 9B3U, 9B3V, 9B3W, 9B3X, 9B3Y, 9B3Z, 8EKP, 8EKQ, 8EKR, 8EKS | medium-to-large diameter DRG and trigeminal ganglion sensory neurons, various immune cell types, red blood cells, neurons, microglial cells, melanocytes, vascular smooth muscle cells, urothelium |
| TRPV3 | TRPV3 (VRL3, oTRPC3); Q8NET8 | Tetrameric6DVW, 6DVY, 6DVZ, 6MHO, 6MHS, 6MHV, 6MHW, 6MHX, 6OT2, 6OT5, 6PVL, 6PVM, 6PVN, 6PVO, 6PVP, 6PVQ, 6LGP, 6UW4, 6UW6, 6UW8, 6UW9, 7MIJ, 7MIK, 7MIL, 7MIM, 7MIN, 7MIO, 7RAS, 7RAU, 7UGG, 7XJ0, 7XJ1, 7XJ2, 7XJ3, 8GKA, 8V6K, 8V6L, 8V6M, 8V6N, 8V6O, 9JDM, 9JE5, 9JEE, 9JEF, 9JEG, 9BKU | keratinocytes, oral gingival and epithelial cells, glandular cells and enterocytes in the small and large intestine |
| Pentameric8GKG, 9DIJ | |||
| TRPV4 | TRPV4 (TRP12, VRL-2, oTRPC4); Q9HBA0 | 8T1B, 8T1C, 8T1D, 8T1E, 8T1F, 8FC7, 8FC8, 8FC9, 8FCA, 8FCB, 8J1B, 8J1D, 8J1F, 8J1H, 8JKM, 8JU5, 8JU6, 8JVI, 8JVJ | Ubiquitous in vascular endothelial cells, pancreatic, tongue and salivary gland exocrine epithelial cells, epithelial cells in kidney tubules, bronchial, tracheal and fallopian tube ciliated cells, skin keratinocytes and melanocytes, macrophages, hepatic Kupffer cells, placentar trophoblast, and decidual cells |
| TRPV5 | TRPV5 (ECaC, ECaC1, CAT2, OTRPC3); Q9NQA5 | 6B5V, 6DMR, 6DMU, 6DMW, 6O1N, 6O1P, 6O1U, 6O20, 6PBE, 6PBF, 7T6J, 7T6K, 7T6L, 7T6M, 7T6N, 7T6O, 7T6P, 7T6Q, 7T6R, 8FFO, 8FHH, 8FHI, 8TF2, 8TF3, 8TF4, 8FFL, 8FFM, 8FFN, 8FFQ | DCT and collecting duct of the kidney, pancreas, small and large intestine, prostate gland, testis, brain, bone osteoclasts, and placenta |
| TRPV6 | TRPV6 (CaT1, ECaC2, OTRPC3); Q9H1D0 | 5IWK, 5IWP, 5IWR, 5IWT, 5WO6, 5WO7, 5WO8, 5WO9, 5WOA, 6BO8, 6BO9, 6BOA, 6BOB, 6D7O, 6D7P, 6D7Q, 6D7S, 6D7T, 6D7V, 6D7X, 6E2F, 6E2G, 7D2K, 7K4A, 7K4B, 7K4C, 7K4D, 7K4E, 7K4F, 7S88, 7S89, 7S8B, 7S8C, 8FOA, 8FOB, 8SP8, 9CUH, 9CUI, 9CUJ, 9CUK | Small intestine, glandular cells of the salivary gland, pancreas, prostate, thyroid, bronchiae, placenta, testis, epididymis, endometrium, stomach, caecum, main olfactory epithelium |
| TRPMs | |||
| TRPM1 | TRPM1 (Melastatin, MLSN1, LTRPC1); Q7Z4N2 | n.d. | melanocytes, retinal ON bipolar cells |
| TRPM2 | TRPM2 (LTRPC2); O94759 | 6MIX, 6MIZ, 6MJ2, 6PUO, 6PUR, 6PUU, 6PUS, 7VQ1, 8E6Q, 8E6T, 8E6R, 8E6S, 8E6U | ubiquitous; high levels in brain and immune cells |
| TRPM3 | TRPM3 (MLSN2, LTRPC3, TRPM3α2); Q9HCF6 | 8ED7, 8ED8, 8ED9, 8DDR, 8DDS, 8DDT, 8DDX, 8DDQ, 8DDU, 8DDV, 8DDW, 9B2A, 9B29, 9B28 | DRG sensory neurons, brain, kidney, pancreatic β-cells, placenta, testis |
| TRPM4 | TRPM4 (LTRPC4, TRPM4B); Q8TD43 | 9B93, 6BQV, 9B90, 6BCO, 6BCQ, 9B92, 9B94, 6BCL, 5WP6, 6BWI, 8RCR, 8RCU, 8RD9, 9B8W, 9B8Y, 6BCJ, 6BQR | ubiquitous; high levels in brain, heart, immune cells, and pancreatic β-cells |
| TRPM5 | TRPM5 (MTR1, LTRPC5); Q9NZQ8 | 8SLE, 8SL6, 8SL8, 8SLA, 8SLI, 8SLP, 8SLQ, 8SLW | type II taste receptor cells, tuft cells, olfactory epithelium, and pancreatic β-cells |
| TRPM6 | TRPM6 (ChaK2, Channel-kinase 2); Q9BX84 | n.d. | kidney, intestine, placenta, lung, testis |
| TRPM7 | TRPM7 (LTRPC7, TRP-PLIK, ChaK1, Channel-kinase 1, MagNum, MIC); Q96QT4 | 5ZX5, 6BWF, 6BWD, 8SI2, 8SI3, 8SIA, 8SI7, 8SI5, 8SI6, 8SI4, 8SI8, 8W2L; kinase domain: 1IAH, 1IA9, 1IAJ | ubiquitous |
| TRPM8 | TRPM8 (Trp-p8, CMR1, Cold receptor 1); Q7Z2W7 | 8BDC, 8E4L, 8E4M, 8E4N, 8E4O, 8E4P, 9B6D, 9B6E, 9B6F, 9B6G, 9B6H, 9B6J, 9B6K, 7WRA, 7RWB, 7WRC, 7WRD, 7WRE, 7WRF | DRG and TG sensory neurons, brain, prostate, pancreatic β-cells, placenta, testis |
| TRPAs | |||
| TRPA1 | TRPA1 (ANKTM1, TRPN1); O75762 | 3J9P, 6PQO, 6PQP, 6PQQ, 6V9V, 6V9W, 6V9X, 6V9Y, 6X2J, 6WJ5, 7JUP, 7OR0, 7OR1, 9MOE | DRG, trigeminal and vagal ganglia, enterochromaffin cells, astrocytes, Schwann cells, bronchial, alveolar, renal and urothelial epithelial cells, keratinocytes, melanocytes, cardiac fibroblasts, pancreatic β-cells, enterochromaffin cells, T-cells, pancreatic and colon cancer, neuroblastoma, glioblastoma |
| TRPMLs | |||
| TRPML1 | TRPML1 (MCOLN1, Mucolipin1); Q9GZU1 | 7SQ7, 7SQ8, 7SQ9, 5WPQ, 5WPT, 5WPV, 7MGL, 9CBZ, 9CBZ, 9CBZ, 5YDZ, 5YE2, 9CC2, 5YE5, 9EKT, 7SQ6, 9EKS, 9EKU, 6E7P, 6E7Y, 6E7Z, 5YE1, 9EKV, 5WJ5, 5WJ9, 9HJ6, 9HJ8, 9HL3, 9HlL4, 9HL6, 9HL8, 9HLA, 9HLB, 9HLC, 9HLD | ubiquitous |
| TRPML2 | TRPML2 (MCOLN2, Mucolipin2); Q8IZK6 | 7DYS, 9EKW, 9EKX, 9EKY, 9EKZ, 9EKO, 8EL1, 6HRS, 6HRR | thymus, spleen, kidney, trachea, liver, lung, colon, testis, thyroid, B- and T-cells, macrophages, dendritic cells |
| TRPML3 | TRPML3 (MCOLN3, Mucolipin3); Q8TDD5 | 6AYG, 6AYE, 6AYF | hair cells of the inner ear, organ of corti, utricle, stria vascularis, alveolar macrophages, skin melanocytes, neonatal enterocytes, kidney, lung, olfactory bulb (sensory neurons), nasal cavity, thymus, colon, trachea, several glands (parathyroid, salivary, adrenal, pituitary), testis, ovary |
| TRPP channels | |||
| PKD2 | TRPP2 (polycystin-2, PC2, PKD2); Q13563 | 8HK7, 8K3S, 6D1W, 9DLI, 9DWQ, 5K47, 5MKE, 5MKF, 5T4D, 6T9N, 6T9O, 6WB8; as PKD1-PKD2 heteromer: 6A70 | ubiquitous; high levels in the kidney, brain, heart |
| PKD2L1 | TRPP3 (polycystin L, PKD2L1); Q9P0L9 | 5Z1W, 6DU8; as PKD1L3-CTD-PKD2L1 heteromer: 7D7E, 7D7F | brain, taste receptor cells, kidney, lung |
| PKD2L2 | TRPP5 (PKD2L2); Q9NZM6 | n.d. | testis and brain, low levels in the kidney, liver, heart, lung |
n.d., not determined.
All TRP channels maintain a notable structural similarity in their channel-pore forming domains, which include 6 membrane-spanning helices and a short stretch of hydrophobic residues between the fifth and sixth transmembrane (TM) segments, often called the pore helix (PH). After the sixth helix, TRPCs, TRPVs, and TRPMs also feature a highly conserved segment known as the TRP domain. TRPPs and TRPMLs exhibit a more distinct topology of the channel segment because they include a long loop linking the first 2 TM helices and lack the TRP domain. Besides this, TRP channels display significant structural heterogeneity in their large N- and C-terminal domains. TRP proteins are assembled in tetramers, implying that 4 subunits contribute to a common membrane-spanning channel pore (Fig. 1).
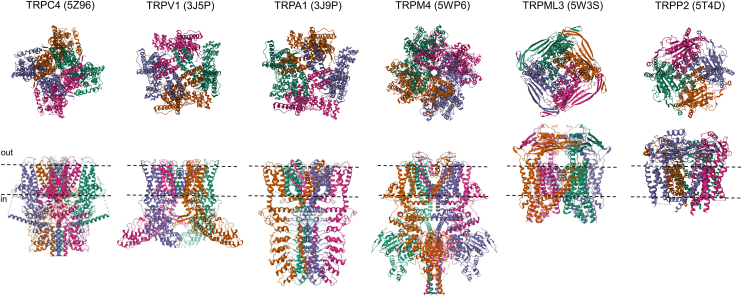
Over the past decade, significant progress has been made in single-particle cryogenic electron microscopy (cryo-EM) analysis of TRP channels, resulting in more than 400 structural models that encompass nearly all vertebrate TRP proteins (Table 1). The structural data obtained enable a detailed examination of the 3-dimensional (3D) arrangement of channel subunits at the atomic level and elucidate the roles of certain amino acids in the tetrameric assembly of TRP channels (Fig. 1). Undoubtedly, these results provide a foundation for structure-function analysis of TRP channels, including insights into regulatory mechanisms, the functional effects of pathogenic mutations, and structure-based drug design.
Apart from considerable structural variability (Fig. 1), TRP channels also display fascinating diversity in functional characteristics, subcellular distribution, expression patterns, and physiological roles (Table 1).ref. bib9,ref. bib10 The pivotal role of TRP and related ion channels in sensory processes has been highlighted by the 2021 Nobel Prize in Physiology or Medicine, awarded to David Julius and Ardem Patapoutian.ref. bib11 Clinical studies and experiments on preclinical disease models revealed the prominent role of TRP proteins in human health and disease.ref. bib12 Accordingly, TRP proteins have been identified as the most appealing pharmacological targets.ref. bib13,ref. bib14 This review provides an up-to-date assessment of TRP channels, emphasizing our progress in developing pharmacological agents that allow selective modulation of mammalian TRP channels in diverse pathophysiological settings.
TRPCs
TRPC gene family
The TRPC gene family in mammals consists of 7 members (Table 1): TRPC1–7. Notably, TRPC2 is a pseudogene in humans, as well as in Old World monkeys and apes (Catarrhini).ref. bib15,ref. bib16 Based on amino acid sequence similarity, the TRPC family is divided into 4 subgroups: TRPC1, TRPC2, TRPC3/6/7, and TRPC4/5 (Fig. 2A).
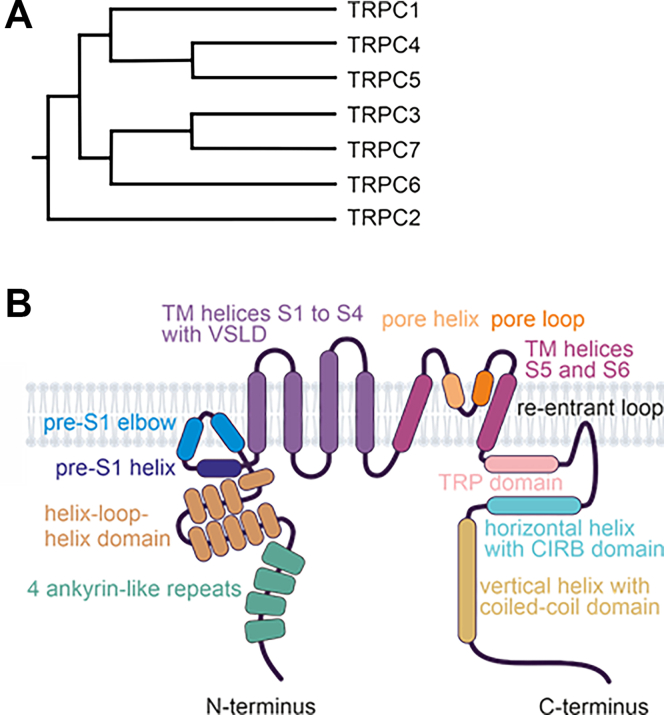
Dysregulation of specific TRPCs has been implicated in various disease states, including pulmonary and renal diseases, as well as neurological disorders. Despite recent advances in the development of potent and selective TRPC modulators, substantial clinical benefits have yet to be realized. This underscores the need for further research to fully elucidate the role of TRPCs in health and disease.
Domain topology, assembly, and functional characteristics of individual TRPCs
Domain topology of TRPCs
In 2018, the first high-resolution 3D structures of TRPCs were resolved using single-particle cryo-EM technology. Since then, several 3D structures of TRPC3, TRPC4, TRPC5, and TRPC6 channels, and TRPC1/4 heteromers have been identified, with resolutions ranging from 2.4 Å to 4.4 Å. These structures were obtained in the presence and absence of inhibitors, activators, and Ca2+, or in complex with interacting proteins such as calmodulin (CaM) or Gαi protein subunits.ref. 17., ref. 18., ref. 19., ref. 20., ref. 21., ref. 22., ref. 23., ref. 24., ref. 25., ref. 26., ref. 27., ref. 28., ref. 29., ref. 30. Table 1 summarizes the available Protein Data Bank (PDB) entries. Studies have revealed that all TRPCs exhibit a tetrameric structure, with each channel subunit adopting a Y-shaped arrangement, giving the tetramers rotational symmetry. The 4 monomers collectively form the central channel pore, which is permeable to both monovalent and divalent cations. Each channel monomer comprises 6 TM domains (TMDs) (S1–S6), formed by α-helices, with cytosolic N- and C-termini. The general structural features of TRPCs are depicted in Fig. 2B.
The N-terminus of TRPCs contains 4 ankyrin-like repeats, conserved across other TRP channel families such as TRPMs, TRPVs, and TRPA1.ref. bib31 These repeats play critical roles in protein or cytoskeleton interactionsref. bib32,ref. bib33 and stabilize the distal cytoplasmic portions of the C-terminus.ref. bib29,ref. bib34,ref. bib35 The first ankyrin-like repeat appears crucial for channel homo- or heterotetramerization,ref. bib36 as it interacts with a connecting helix – the rib helix – and the C-terminus of the adjacent monomer, potentially stabilizing the tetramer.ref. bib20 Notably, the 3D structure of TRPC5 in complex with the Gαi3 protein subunit has revealed direct interaction between Gαi3 and the ankyrin-like repeat domains 1 and 2.ref. bib27
TRPC4 and TRPC5 channels feature 7 helices, while TRPC3 and TRPC6 contain 9 helices of varying lengths, connected by loops (helix-loop-helix domain) located at the proximal N-terminus. This domain interacts with the C-terminal TRP domain, a conserved structure that follows the S6 TMD. Adjacent to this domain lies the pre-S1 elbow, a membrane-associated structure consisting of 2 helices. The following pre-S1 helix itself is parallel to the cytoplasmic membrane surface.
The TM helices S1–S4 form a Y-shaped “shank” and a voltage-sensing-like domain (VSLD) that harbors binding sites for various channel modulators. The second Y-shaped "leg" is composed of the TM helices S5 and S6, connected via the PH and pore loop. The ion-conducting pathway is primarily formed by S5, S6, and the pore domain, which includes the PH, turret, and loop. The pore walls are constructed of 4 pore loops and their corresponding S6 helices, with the extracellular pore region carrying a negative charge. TRPC5 has one additional negatively charged amino acid compared with TRPC4,ref. bib23 potentially explaining the higher single-channel conductance of TRPC5.ref. bib37 Mutations in this extracellular pore region alter channel properties, underscoring its role in ion selectivity and conductivity.ref. bib23,ref. bib38
The leucine-phenylalanine-tryptophan (LFW) motif in the PH, along with upstream residues, stabilizes the pore domain by interacting with the S5 domain of the same monomer and the S6 domain of an adjacent monomer. The ion selectivity filter is formed by phenylalanine and glycine residues, which are located 2 amino acids downstream of the LFW motif at the end of the PH and at the beginning of the pore loop, with their carbonyl oxygens interacting electrostatically with permeating cations, thereby forming the ion selectivity filter.ref. bib28 Below the selectivity filter lies a central, presumably water-filled, pore cavity formed by the S6 helix, with constriction sites at the cytoplasmic end. The narrowest part of the ion-conducting pore, the lower gate, is usually formed by 3 amino acids. These constriction sites, spaced by 3 amino acids each, consist of valine, histidine, and glutamine in TRPC1ref. bib30; leucine and isoleucineref. bib17 or leucine, isoleucine, and phenylalanineref. bib19 in TRPC3; isoleucine, asparagine, and glutamine in TRPC4ref. bib22 and TRPC5ref. bib23; and leucine, isoleucine, and phenylalanineref. bib28 or the adjacent amino acids isoleucine, asparagine, and glutamineref. bib18 in TRPC6. Interestingly, it was suggested that, depending on the selected inhibitor, the lower gate of TRPC6 is formed by isoleucine, asparagine, and glutamine.ref. bib19
Another conserved structure in TRPCs is the helical TRP domain, located proximally to the intracellular C-terminus. It includes the TRP box, which contains the amino acid motif glutamate-tryptophan-lysine-phenylalanine-alanine-arginine (EWKFAR). This motif stabilizes the cytoplasmic proximal section of the channel. The tryptophan and phenylalanine residues of the TRP box, along with tyrosine and lysine residues of the TRP helix, interact with the N-terminal proximal helices (helix-loop-helix domain), the intracellular loop between S4 and S5, and the cytoplasmic ends of the S1 and S4 helices. Consequently, the TRP domain forms a stabilizing contact surface between the TM regions and the proximal cytoplasmic sections of the channel.
Although the precise function of the TRP box remains unclear, growing evidence suggests that it plays a critical role in channel-gating regulation. For instance, glycine at position 503 in TRPC4 affects gating, as a substitution for serine results in uncontrolled channel opening.ref. bib39 Glycine 503 interacts with tryptophan 635 in the TRP domain, stabilizing the S6 segment that constitutes the lower gate.ref. bib20 Additionally, the TRP domain may interact with phosphoinositol-4,5-bisphosphate (PIP2), as observed in other TRP channels like TRPM8.ref. 40., ref. 41., ref. 42. In TRPC6, PIP2 binding likely occurs between the distal TRP box and the pre-S1 helix.ref. bib43 A lysine-to-alanine substitution at position 771 in the TRP box potentiates TRPC6 currents, altering the channel state. In TRPC3, PIP2 binds at the intersection of the pre-S1 helix and the S1 TM helix, inducing conformational changes via the re-entrant loop and TRP domain.ref. bib44 Altogether, the TRP domain is highly conserved within the TRP channel superfamily and is crucial for structural stabilization, gating regulation, and cofactor interactions.
Following the TRP domain, a loop containing 2 glutamates (in TRPC4 and TRPC6), 2 aspartates (in TRPC1 and TRPC3), or an aspartate-glutamate combination (in TRPC5 and TRPC7) extends into the cytoplasmic membrane layer. This loop interacts with the pre-S1 elbow of the N-terminus and the cytoplasmic end of the S1 helix. Although not resolved in all 3D structures, it is presumed to be a common feature of TRPCs.
TRPC4 and TRPC5 possess a unique extracellular disulfide bond between 2 cysteines near the S5 PH linker, which may play a role in redox sensing.ref. bib22,ref. bib45 Additionally, TRPC3 and TRPC6 differ from TRPC4 and TRPC5 in the length of their S3 helices on the extracellular side, which are approximately 4 helical turns longer.
A nearly parallel helix, slightly sloping toward the cytoplasmic membrane, has been variously termed “CH1”,ref. bib18 “horizontal helix”,ref. bib29 or “CTD rib helix”.ref. bib17 In TRPC4 and TRPC5, it is referred to as the “connecting helix”ref. bib22,ref. bib23 or “Rib helix + CaM 1,4,5-trisphosphate (IP3) receptor binding (CIRB) domain”.ref. bib20 The CIRB domain, present in all TRPCs, begins before the horizontal helix and encompasses most of it. The 3D structure of TRPC4 in complex with CaM reveals an interaction between 1 channel monomer and 1 CaM protein at the CIRB domain.ref. bib21 Interestingly, CaM binding depends on calcium concentration: at low calcium levels, IP3 receptor binding is favored, while at high calcium levels, CaM preferentially binds. CaM stabilizes the inactive channel state, whereas IP3 receptor binding promotes activation.ref. bib21,ref. bib46,ref. bib47
At the distal end of the C-terminus, a perpendicular helix forms another conserved feature, variously called “CH2”,ref. bib18 “CTD pole helix”,ref. bib17 “vertical helix”,ref. bib29 “coiled-coil (CC) domain”,ref. bib22,ref. bib23 or “C-term helix”ref. bib20 in TRPC4 and TRPC5. This helix exhibits a heptahedron-like pattern, and 4 such helices assemble to form a central, vertically extending tunnel in the distal cytoplasmic region. Together with the ankyrin repeat domains (ARDs), this structure is critical for tetrameric assembly.
TRPCs can be regulated by lipids, though the precise mechanism of lipid regulation remains incompletely understood. Recently, 2 distinct lipid-binding sites have been identified. Lipid binding site 1 is located in the inner leaflet of the VSLD, while lipid binding site 2 is situated in the pore region between the pore loop and the S6 helix of an adjacent channel monomer. In the apo states of TRPC5ref. 23., ref. 24., ref. 25. and TRPC4,ref. bib20,ref. bib22 a lipid has been found in the pore region, interacting with the phenylalanine and tryptophan residues of the LFW motif. This lipid has been identified as ceramide-1-phosphate, phosphatidic acid,ref. bib23,ref. bib25 or diacylglycerol (DAG).ref. bib24,ref. bib48 Mutations in lipid binding site 2 have been shown to affect the DAG sensitivity of TRPC3 and TRPC6 channels,ref. bib28,ref. bib49,ref. bib50 highlighting this region’s critical role in regulating channel activity.
In the 3D structure of TRPC6, a lipid identified as phosphatidylcholine was found in lipid binding site 2, interacting with the phenylalanine and tryptophan residues of the LFW motif.ref. bib28 Interestingly, this lipid was shifted upward toward the extracellular side and rotated vertically compared with the lipids found in the TRPC4 and TRPC5 structures.
At lipid binding site 1, cholesterol hemisuccinate—introduced during protein purification—was detected. However, in the TRPC3 structure, a phospholipid was identified at this site,ref. bib17 which may represent an endogenous lipid that binds to the channel. Additionally, the 3D structures of TRPC4 in both their ligand-bound and apo statesref. bib21 suggest the presence of a Ca2+ ion in the VSLD domain. This ion appears to stabilize ligand binding in the VSLD through a bridging water molecule. Similar cation densities have been observed in the TRPC4 and TRPC5 structures.ref. bib22,ref. bib23 Deleting the Ca2+ binding site in TRPC5 abolished channel activation by elevated extracellular Ca2+ concentrations.ref. bib24
To date, high-resolution structures of TRPCs in their open state are still lacking, and significant portions of the C-terminal region remain unresolved. However, comparisons of TRPC6 3D structures in the apo and activator-bound states suggest that channel opening involves critical movements of the TM helices.ref. bib28 Upon activation, the S6 helix of TRPC6 relaxes and moves downward, widening the restriction point at the lower gate, which is formed by leucine, isoleucine, and phenylalanine residues. This movement is accompanied by a downward bending of the S5 helix. Conversely, Vinayagam et alref. bib21 propose a different mechanism, in which channel activation involves movements of the VSLD and the TRP box.
Assembly of TRPCs
TRPCs can assemble into either homotetrameric or heterotetrameric channel complexes. However, TRPC1 alone may not form functional homotetrameric channels, at least in overexpression systems. Instead, it can form heterotetrameric channel complexes with other TRPC subunitsref. bib30,ref. 51., ref. 52., ref. 53., ref. 54., ref. 55., ref. 56. and even with subunits from other TRP channel families, such as TRPP2,ref. bib57 TRPV4,ref. bib58 and TRPV6.ref. bib59
In neurons, TRPC1 can heterotetramerize with TRPC4 and TRPC5 to form TRPC1/4/5 channels.ref. bib54,ref. bib55,ref. 60., ref. 61., ref. 62. Within these heterotetramers, TRPC1 alters the permeation properties and reduces calcium permeability.ref. bib51,ref. 63., ref. 64., ref. 65. Additionally, TRPC1 plays a regulatory role as part of a multiprotein complex that includes stromal interaction molecule 1 (STIM1) and the calcium release-activated calcium channel protein Orai, both of which are involved in store-operated calcium entry (SOCE).ref. 66., ref. 67., ref. 68. Heterotetrameric complexes can also form between TRPC3, TRPC6, and TRPC7 subunits,ref. bib60,ref. bib69,ref. bib70 as well as between TRPC3 and TRPC4.ref. bib71 TRPC2 channels, which are highly expressed in the vomeronasal organs (VNOs) of rodentsref. bib72 but are pseudogenes in humans, are more likely to exist as homomeric channels. In the brain, TRPC3, TRPC6, and TRPC7 preferentially form homomeric channels.ref. bib62 Assumably, the expression of TRPCs as homomeric or heteromeric channels might vary between different cells and tissues.
Functional characteristics of individual TRPCs
TRPCs are widely recognized as nonselective, calcium-permeable, and receptor-operated cation channels. These channels are activated downstream of phospholipase C (PLC) following the activation of Gq/11 protein-coupled receptors or receptor tyrosine kinases.ref. bib5,ref. bib73 PLC activation leads to the cleavage of PIP2 into the second messengers, inositol IP3 and DAG. IP3 promotes calcium release from intracellular stores, increasing the free intracellular calcium concentration, while DAG directly activates TRPCs, facilitating sodium and calcium influx and triggering cellular effects. All TRPCs can be directly activated by DAG,ref. 74., ref. 75., ref. 76., ref. 77., ref. 78. suggesting that DAG serves as an endogenous activator. However, the DAG sensitivity of TRPC4 and TRPC5 channels is tightly regulated and requires dephosphorylation of a threonine residue in the C-terminal postsynaptic density protein, Drosophila disc large tumor suppressor, and zonula occludens-1 protein (PDZ)-binding motif. This motif is unique to TRPC4 and TRPC5 channels and allows for the replacement of Na+/H+ exchanger regulatory factor (NHERF) 1 and 2 adapter proteins, which are essential for DAG sensitivity.ref. 77., ref. 78., ref. 79.
In the case of TRPC3, the use of the photoswitchable DAG derivative, OptoDArG, revealed that DAG might activate the channel through a fenestration involving a conserved glycine residue behind the channel’s selectivity filter.ref. bib49 However, the precise mechanism underlying lipid sensing remains to be fully elucidated. IP3 and IP3 receptors also modulate TRPC function. IP3 can directly activate TRPC7 channels,ref. bib80 while IP3 receptors activate TRPC3 and TRPC5 channels.ref. bib47,ref. bib81 This interaction occurs via the C-terminal CIRB motif of TRPCs, where IP3 receptor binding competes with CaM binding.ref. bib21,ref. bib46,ref. bib47,ref. bib82,ref. bib83 The binding of IP3 receptors establishes an active channel state, while CaM binding promotes an inactive state.
TRPC activity is further influenced by junctate, a TM protein expressed in the endoplasmic reticulum (ER) membrane that interacts with IP3 receptors.ref. 84., ref. 85., ref. 86. Junctate serves as a calcium-sensing structural component of Orai and STIM1 within the ER membrane at ER-plasma membrane junctions.ref. bib87 Notably, junctate enhances the formation of ER-plasma membrane junctions containing TRPC3 and IP3 receptors,ref. bib86 which may represent a mechanism by which IP3 receptors and TRPCs contribute to SOCE.
A store-operated activation mechanism has been proposed for TRPCs based on observations that the depletion of intracellular calcium stores triggers calcium influx through the plasma membrane.ref. bib88 This phenomenon is associated with the highly Ca2+-selective calcium release-activated current.ref. bib89 However, this current does not share the nonselective characteristics of TRPC currents. Despite this, TRPC1 has been suggested to function as a store-operated channel, either alone or in complex with Orai.ref. 90., ref. 91., ref. 92., ref. 93. Additionally, TRPC1 may also be activated via a receptor-dependent mechanism that involves store depletion, effectively integrating both activation pathways.ref. bib94
Currently, it is widely accepted that Orai and STIM proteins are the primary molecular components of store-operated calcium influx.ref. 95., ref. 96., ref. 97., ref. 98., ref. 99. STIM serves as a calcium sensor in the ER membrane and activates Orai proteins, which form the channel pore. Evidence suggests that TRPCs, particularly TRPC1, may interact with STIM and/or Orai, modulating SOCE.ref. bib100,ref. bib101 Nevertheless, while Orai is essential for calcium influx following store depletion, TRPCs are not strictly required, as demonstrated in studies using mice lacking all 7 TRPC genes.ref. bib102
The membrane lipid PIP2 also modulates TRPC function. PIP2, as a substrate of PLC, plays a role in receptor-operated signaling pathways but can also act as a second messenger that regulates cellular processes, potentially influencing ion channel activity.ref. bib103,ref. bib104 Interestingly, the effects of PIP2 vary depending on the patch-clamp configuration used. In inside-out patches, heterologously expressed TRPC5, TRPC3, TRPC6, and TRPC7 channels are activated by PIP2,ref. bib105,ref. bib106 whereas endogenously expressed TRPC6 channels are inhibited.ref. 107., ref. 108., ref. 109. Whole-cell patch-clamp recordings of heterologously overexpressed TRPC5 channels show that PIP2 depletion activates TRPC5,ref. bib77,ref. bib106 while PIP2 application through the patch pipette inhibits the channel. TRPC4 channels are similarly inhibited by PIP2 in an isoform-specific manner, with PIP2 binding to the C-terminal region stabilizing the inactive channel state.ref. bib110 Furthermore, the intracellular application of PIP2 reduces TRPC5 desensitization following receptor activation.ref. bib111
In overexpression systems, PIP2 depletion induces a conformational change in the TRPC5 C-terminal region, causing the dissociation of NHERF and conferring direct sensitivity to DAG.ref. bib77 This NHERF dissociation also occurs following protein kinase C (PKC) inhibition or threonine mutation in the C-terminal PDZ-binding motif of TRPC4/5 channels.ref. bib77 Similar PKC-related modulation of DAG sensitivity has been reported,ref. bib78,ref. bib79 suggesting a regulatory role of PIP2 in TRPC function. Notably, PIP2 application through the patch pipette enhances DAG-induced TRPC5 currents after PKC inhibition.ref. bib78
A PIP2-binding site has been proposed for TRPC5 near the linker regions between the S2 and S3 helices, the S4 and S5 helices, the TRP helix, and the helix-loop-helix domain.ref. bib27 The intracellular application of PIP2 increases the open probability of TRPC5 channels.ref. bib27 Moreover, trivalent cations and DAG allosterically modulate PIP2 binding to TRPC5, underscoring PIP2 as a critical factor in channel activation and inactivation.ref. bib78 Additionally, PIP2 binding to TRPC5 is enhanced by the interaction with Gαi protein subunits, making TRPC5 more readily open in the cell membrane.ref. bib27
Lipid regulation of TRPC3 channels may require an interplay between PIP2 and DAG.ref. bib112 Cleavage of PIP2 by PLC generates DAG, which can bind to lipid-binding site 2 within the channel pore, while PIP2 interacts with lipid-binding site 1 near the VSLD. This interaction inhibits TRPC3 channel opening, regulating DAG’s access to lipid-binding site 2.ref. bib112 Furthermore, it has been proposed that PIP2 modulates the ionic selectivity of the TRPC3 pore following receptor stimulation, and that in PIP2-rich membrane domains, TRPC3 may be recruited to ER-plasma membrane junctions, suggesting an interaction between TRPC3 and STIM1 to regulate calcium influx.ref. bib112
In TRPC6 channels, amino acid substitutions in the PIP2-binding site at the pre-S1 helixref. bib43 reduce receptor- and 1-oleoyl-2-acetyl-sn-glycerol–induced TRPC6 currents, indicating that PIP2 binding enhances channel activity. However, substituting lysine with glutamine in the distal TRP box reverses this effect, potentiating TRPC6 currents at low PIP2 concentrations.ref. bib43 This highlights the critical role of the C-terminus in PIP2-mediated regulation of TRPCs. Despite these findings, the lipid regulation of TRPCs, including the precise roles of PIP2 and DAG in channel activation, remains incompletely understood.
Cleavage of PIP2 by PLC also produces protons at the cytoplasmic side of the plasma membrane, causing localized acidification.ref. bib113 This acidification may influence TRPC4 channel activity by sensitizing PLCδ1 to calcium, leading to its activation and the potentiation of TRPC4 currents.ref. bib114 The signaling pathway for TRPC activation is complex, involving multiple components whose interplay is not yet fully elucidated. Additionally, extracellular protonsref. bib115 and trivalent cations, such as lanthanum and gadolinium,ref. bib38,ref. bib116 can potentiate TRPC4 and TRPC5 currents. Interestingly, TRPC4 and TRPC5 channels can be activated not only downstream of Gq/11 protein but also via Gi/o protein-coupled receptor activation. For example, Gi/o protein-coupled receptor activation triggers TRPC4 channel opening through PLCδ1 activation.ref. bib114 However, TRPC4 activation by Gi/o protein-coupled receptors may also occur independently of PLC, relying instead on a direct interaction with Gαi proteins.ref. bib117 Similarly, TRPC5 channels are activated by Gαi proteins.ref. bib118
Recent structural analysesref. bib27 have revealed that Gαi proteins directly interact with TRPC5 channels via the N-terminal ankyrin-like repeat domains 1 and 2, leading to channel activation in the presence of PIP2. Direct interactions between G proteins and ion channels have so far been well established only for G protein-activated inwardly rectifying potassium channels, where Gβγ subunits directly bind to the channel.ref. bib119 It has also been proposed that TRPC5 channels are activated downstream of Gs protein-coupled receptor stimulation through a cAMP-mediated intracellular calcium release.ref. bib120
The free intracellular calcium concentration also plays a critical role in regulating TRPC activity. Increasing free intracellular calcium levels above 300 nM activates TRPC4 and TRPC5 channels.ref. bib37,ref. bib121 Consequently, calcium release from the ER following receptor activation can induce TRPC4 and TRPC5 channel opening, further elevating free intracellular calcium levels and enhancing sodium and calcium influx. Simultaneously, depletion of intracellular calcium stores activates Orai channels, contributing to an additional increase in free intracellular calcium. Free intracellular calcium concentrations of approximately 1 μM were shown to potentiate receptor-operated TRPC5 channels.ref. bib122 Even higher concentrations, with an EC50 of around 12 μM, are required to activate the short isoform TRPC4β.ref. bib114 However, as noted earlier, elevated calcium levels also promote CaM binding,ref. bib21 which restricts the mobility of the TRP helix and locks the channel in its closed state.ref. bib21
TRPC4 and TRPC5 currents can also be potentiated by increasing extracellular calcium concentrations to 10 mM.ref. bib122 TRPC6 channels are similarly sensitive to free intracellular calcium levels, but their activation is primarily mediated through CaM-dependent kinase II phosphorylation.ref. bib80,ref. bib123 Elevated free intracellular calcium concentrations can also promote the translocation of TRPCs to the plasma membrane.ref. bib124 Additionally, higher extracellular calcium levels increase TRPC6 currents.ref. bib80
However, extracellular calcium concentrations exceeding physiological levels (≥3 mM) inhibit TRPC6 channel activity, while TRPC7 currents are inhibited by extracellular calcium even at micromolar concentrations.ref. bib80 TRPC3 currents are similarly suppressed by extracellular calcium.ref. bib125 Altogether, calcium exerts both stimulatory and inhibitory effects on different TRPCs, allowing for precise fine-tuning and regulation of channel function.
Expression pattern and primary physiological roles of TRPCs
An overview of the expression profile of TRPCs is provided in Table 1. TRPC1 is ubiquitously expressed across various tissues.ref. bib126,ref. bib127 It forms heterotetrameric channels with other TRPC protein subunits,ref. 51., ref. 52., ref. 53., ref. 54. and even with other TRP proteins, such as TRPP2,ref. bib128 TRPV4,ref. bib58 and TRPV6.ref. bib59 These interactions alter biophysical propertiesref. bib54 and reduce calcium permeability.ref. bib51,ref. bib59,ref. 63., ref. 64., ref. 65.
TRPC1 is highly expressed in neurons, where it plays roles in axonal chemotaxis,ref. bib129 mediates the slow excitatory postsynaptic potential induced by metabotropic glutamate receptors in Purkinje cells,ref. bib130 and provides neuroprotection against exogenous neurotoxins.ref. bib131 Additionally, TRPC1 enhances the differentiation of hippocampal neuronsref. bib132 and promotes the proliferation of neuronal progenitor cells in the hippocampusref. bib133 and cochlear spiral ganglion.ref. bib134 These findings suggest that TRPC1 is involved in neuronal homeostasis and might play a role in neurodegeneration.ref. bib135
In nonneuronal tissues, calcium influx via TRPC1 in mandibular salivary gland cells enhances salivary secretion.ref. bib136,ref. bib137 In the cardiovascular system, TRPC1 promotes proliferation,ref. bib65 influences vascular tone, and is upregulated in smooth muscle and cardiac myocytes after stenosis, leading to enhanced proliferation.ref. bib138,ref. bib139 This suggests a potential role for TRPC1 in cardiac dysfunction.ref. bib140 TRPC1 also regulates vascular function, inducing vasoconstriction.ref. 141., ref. 142., ref. 143., ref. 144. However, endothelial TRPC1 promotes vasodilation.ref. 145., ref. 146., ref. 147. TRPC1/4 heteromers regulate endothelial permeability in the lungs,ref. bib148 and TRPC1 contributes to pulmonary hypertension,ref. bib149,ref. bib150 immune system regulation,ref. bib151 cancer progression,ref. bib152 and endocrine functions, such as parathyroid hormone secretion.ref. bib153
TRPC2 channelsref. bib75 are functionally expressed in most macrosmatic species, such as fish and mammals.ref. bib154 However, in microsmatic species, such as hominids and Old World monkeys, TRPC2 is nonfunctional and has evolved into a pseudogene.ref. bib15,ref. bib16 This loss is associated with the degeneration of the VNO,ref. bib155 where TRPC2 channels are essential for pheromone perception.ref. bib72,ref. bib156
In the VNO, TRPC2 is crucial for pheromone-driven behaviors. TRPC2 deficiency results in impaired pheromone perception, sex-typical (sex is defined as the biological classification of individuals as male or female) brain changes,ref. bib157 and altered social behaviors, including reduced aggression and impaired olfactory sex recognition.ref. bib158,ref. bib159 TRPC2-deficient males may exhibit male–male mating behavior,ref. bib159 which is accompanied by reduced dopamine levels in dopaminergic neurons.ref. bib160 Similarly, TRPC2-deficient females may display male-typical sexual behavior.ref. bib161
Outside the VNO, TRPC2 channels have diverse functions. In thyroid cells, TRPC2 may increase thyroid-stimulating hormone receptor expression, reduce thyroglobulin maturation, influence free intracellular calcium and iodide homeostasis, and reduce thyroid cell proliferation.ref. bib162 In spermatozoa, TRPC2 channels enhance the acrosome reaction,ref. bib163 facilitating penetration of the oocyte.ref. bib163 In erythrocytes, TRPC2 channels have been implicated in oxidative stress-induced hemolytic anemia.ref. bib164 Expression in the testis was also demonstrated.ref. bib165
TRPC3 channels are highly expressed in the central nervous system (CNS),ref. bib166 with particularly prominent expression in the pituitary gland and Purkinje cells of the cerebellum.ref. 167., ref. 168., ref. 169. TRPC3 is also expressed in the cardiovascular system, notably in the heartref. bib170 and lungs of patients with pulmonary arterial hypertension.ref. bib171,ref. bib172
TRPC3 can form heterotetrameric channel complexes with TRPC6 and TRPC7,ref. bib60,ref. bib70 as well as with TRPC1ref. bib51,ref. bib56 and TRPC4ref. bib71 protein subunits. Mice deficient in the TRPC3 gene exhibit abnormal extrapyramidal coordination deficits, which are attributed to the absence of TRPC3-mediated calcium influx in Purkinje cells.ref. bib167 A point mutation in the TRPC3 gene (T635A), which leads to increased channel activity,ref. bib173 results in an ataxic phenotype. This mutant mouse line is referred to as "moonwalker."ref. bib174 In these mice, increased TRPC3 activity causes impaired differentiation of Purkinje cells during early postnatal development and extensive degeneration during late postnatal development.ref. bib174
In addition to Purkinje cells, TRPC3 is expressed in unipolar brush cells, which are excitatory interneurons in the cerebellum.ref. bib175,ref. bib176 These unipolar brush cells undergo significant degeneration within the first 4 weeks of postnatal development in moonwalker mice.ref. bib177
TRPC3 channels are also expressed in the kidneyref. bib178 and blood vessels. They are found in the endothelium of the low-pressure system, such as umbilical veinsref. bib179 and pulmonary arteries,ref. bib180 as well as in the high-pressure system, including afferent arterioles of the kidneyref. bib181 and coronary arteries.ref. bib182 Notably, TRPC3 channels are highly expressed in the endothelium of high-pressure arteries, where they are implicated in hypertensionref. bib181 and arteriosclerosis.ref. bib183
TRPC3 channels are also expressed in smooth muscle cells of resistance arteriesref. bib184,ref. bib185 and cardiomyocytes.ref. 186., ref. 187., ref. 188., ref. 189., ref. 190. They may promote cardiac hypertrophyref. bib188,ref. bib190 and fibrosis.ref. bib191 Additionally, TRPC3 channels have endocrine functions, enhancing insulin secretion,ref. bib192 and are involved in cancer, such as ovarian and breast cancer.ref. bib193,ref. bib194 TRPC3 also influences tumor energy metabolismref. bib195 by enhancing mitochondrial calcium uptake.ref. bib196,ref. bib197 This mitochondrial mechanism demonstrates that TRPC3 plays important roles both in the plasma membrane and intracellularly.ref. bib198
TRPC4 and TRPC5 channels are expressed in multiple tissues and organs, including the brain,ref. bib199 kidney,ref. bib200,ref. bib201 and vascular system.ref. bib202 TRPC4 is highly expressed in the endothelium,ref. bib203 where it regulates endothelial calcium homeostasis. In the CNS, TRPC4 and TRPC5 channels are significantly expressed and involved in neuroplasticity. TRPC4 is present in corticolimbic regionsref. bib199 and midbrain dopaminergic neurons in the ventral tegmental area and substantia nigra.ref. bib204
In peripheral sensory and spinal cord neurons, TRPC4 and TRPC5 contribute to pain, inflammation, and itch.ref. 205., ref. 206., ref. 207. TRPC1/4/5 heteromers, particularly TRPC4, are implicated in neurodegenerationref. bib208 and play a role in morphine tolerance and hyperalgesia.ref. bib209
TRPC5 is primarily expressed in the brain,ref. bib168,ref. bib199,ref. bib210,ref. bib211 where it is associated with neurite growth, neurotransmission, and learning.ref. bib61,ref. 212., ref. 213., ref. 214. In the amygdala, TRPC4 and TRPC5 mediate strong fear responses to aversive stimuliref. bib214,ref. bib215 and are involved in peripheral-induced neuropathic pain syndromes.ref. bib216 Notably, TRPC5 activation reduces mechanically induced neuropathic pain.ref. bib217
Beyond pain, TRPC5 influences metabolismref. bib218,ref. bib219 and contributes to epileptic activity. In hippocampal CA1 neurons, TRPC5 causes constant membrane depolarizations, known as plateau potentials,ref. bib220 which occur during epileptic seizures.ref. bib221,ref. bib222 TRPC5-deficient mice exhibit fewer epileptic seizures.ref. bib61 Additionally, TRPC5 inhibits hippocampal neurite length and growth cone morphology.ref. bib213
TRPC5 is expressed in adrenal chromaffin cells and plays a crucial role in adrenaline secretion, which is essential for preventing hypoglycemia.ref. bib223,ref. bib224 TRPC5 also contributes to angiogenesisref. bib225 and acts as a cold sensorref. bib226 in the heterologous overexpression system,ref. bib227,ref. bib228 the peripheral nervous system,ref. bib227 and in odontoblasts.ref. bib229
TRPC5 and TRPC6 channels exhibit opposing effects on the actin cytoskeleton of podocytes and fibroblasts. Receptor-operated TRPC5 activation leads to the loss of actin stress fibers, resulting in a motile and noncontractile phenotype,ref. bib230 which characterizes podocyte damage and contributes to proteinuria and kidney disease.ref. 231., ref. 232., ref. 233. In contrast, TRPC6 activation promotes the formation of actin stress fibers, establishing a contractile and nonmotile phenotype.ref. bib230
TRPC5 channels may also play a role in cancer by enhancing angiogenesis.ref. bib234,ref. bib235 Furthermore, TRPC5 contributes to chemotherapy-induced multidrug resistance in tumor cells by increasing the expression of ATP-binding cassette subfamily B member 1 transporters, also known as P-glycoprotein 1 or multidrug resistance protein 1.ref. 234., ref. 235., ref. 236. TRPC5 additionally functions as a pressure sensor in aortic baroreceptors, stabilizing blood pressure,ref. bib237 and mediates endothelium-dependent contraction of carotid arteries.ref. bib238,ref. bib239
TRPC6 channels are highly expressed in the vasculature, lungs, brain, placenta, spleen, ovaries, small intestine, neutrophils, and podocyte foot processes.ref. bib240 In neurons, TRPC6 channels increase endocannabinoid synthesis,ref. bib241 promote dendrite growth,ref. bib242,ref. bib243 and support neuronal plasticity.ref. bib243 TRPC6 is also expressed in extrinsic fibers innervating the intrinsic cardiac ganglia,ref. bib244 olfactory epithelium neurons,ref. bib245 retinal ganglion cells,ref. bib246 and various brain regions, including the cortex, hippocampus, substantia nigra, and cerebellum.ref. bib247
In the vascular system, TRPC6 channels mediate vasoconstrictionref. bib248 and promote vascular smooth muscle cell proliferation.ref. bib249,ref. bib250 TRPC6 was proposed to be a direct mechanosensorref. bib251 mediating myogenic vasoconstriction. However, TRPC6 rather acts as a mechanotransducer with indirect mechanosensitivity.ref. bib252,ref. bib253 Nevertheless, TRPC6 may contribute to ultrasound neuromodulation in the brainref. bib254 and is proangiogenic.ref. bib255,ref. bib256 Low extracellular pH activates TRPC6, inhibiting platelet aggregation,ref. bib257 while oxidants, like hydrogen peroxide, not only activate TRPC6 but also increase its membrane expression.ref. bib258
In the kidney, TRPC6 is expressed in glomeruli, tubular cells, and podocytes.ref. bib259 Mutations in the TRPC6 gene result in podocyte damage and are associated with focal segmental glomerulosclerosis (FSGS), a chronic kidney disease leading to end-stage renal failure.ref. bib260,ref. bib261 Although the pathomechanism remains unclear, TRPC6 channels in podocyte foot processes, which form the slit diaphragm, are crucial for maintaining calcium homeostasis.
Beyond renal functions, TRPC6 has neuronal roles, including involvement in neurodegeneration and Alzheimer’s disease, and is highly expressed in several cancers.ref. bib262 In the lungs, TRPC6 is found in airway smooth muscle cells,ref. bib263,ref. bib264 epithelial cells,ref. bib264 and endothelial cells.ref. bib265 TRPC6 contributes to hypoxic pulmonary vasoconstriction,ref. bib263 lung ischemia-reperfusion (IR)-induced edema,ref. bib265 and lung fibrosis.ref. bib266 In the heart, TRPC6-mediated zinc influx enhances myocardial contractility, suggesting its potential as a therapeutic target for heart failure.ref. bib267,ref. bib268 TRPC6, along with TRPC1, TRPC3, and TRPC5, also plays roles in the immune system and phagocytosis.ref. bib269
TRPC7 channels are the least studied TRPCs. They are expressed in the CNS, hypophysis, kidneys,ref. bib168 heart, lungs,ref. bib270 endothelium,ref. bib271 vasculature,ref. bib108,ref. bib272,ref. bib273 eyes, spleen, and testis.ref. bib76 TRPC7 activation has been linked to an increased breathing rate,ref. bib274 and may contribute to enhanced proliferation in autosomal dominant polycystic kidney disease (ADPKD).ref. bib275 Cardiac TRPC7 channels are implicated in arrhythmiasref. bib276 and myocardial apoptosis.ref. bib277 TRPC7 may also play a role in the pupillary light reflex,ref. bib278 although this remains controversial.ref. bib279
TRPC7 and TRPC6 channels are both involved in phototransduction in retinal ganglion cells, where they are activated downstream of the photosensitive Gq protein-coupled receptor melanopsin, leading to PLCβ4-induced TRPC6/7 activation and cAMP formation.ref. bib278 High TRPC7 expression is associated with the progression of hepatocellular carcinomaref. bib280,ref. bib281 and lung adenocarcinoma.ref. bib282
In summary, TRPCs play vital roles in the regulation of calcium homeostasis and are involved in vascular, neuronal, and kidney functions, sensory transduction, as well as cell migration and proliferation.
Human diseases associated with TRPCs
Surprisingly, the global knockout (KO) of all TRPCs results in viable mice that are fertile.ref. bib102,ref. bib283,ref. bib284 However, a multitude of animal models suggests that TRPCs, in particular but not exclusively, may underlie or aggravate different human diseases through their excessive activity.
A gain-of-function (GOF) mutation in the TRPC3 gene (R762H) is associated with a rare case of autosomal dominant adult-onset spinocerebellar ataxia type 41 (OMIM 616410). Overexpression of this variant in murine neuroblastoma cells leads to neuronal cell death, presumably caused by an increased open probability of the channel,ref. bib285 thereby resembling the phenotype of the mutated channel in the so-called “moonwalker” mouse.ref. bib174
TRPC5 is frequently discussed as a potential therapeutic target for treating kidney diseases, anxiety, and depression.ref. bib214,ref. bib286 In 2014, Mignon-Ravix et alref. bib287 described a loss-of-function (LOF) mutation in the TRPC5 gene associated with X-linked intellectual disabilities (OMIM 300982). Subsequently, additional missense variants in TRPC5, resulting in either constitutively open or nonfunctional channels, were linked to cases of intellectual disabilities, anxiety, and autism.ref. bib288,ref. bib289 Moreover, TRPC5 variants are associated with severe childhood-onset obesity, suggesting a potential role for TRPC5 in the regulation of food intake.ref. bib288
TRPC6 dysfunction, resulting from gene mutations or upregulation of its expression, is best understood in the context of pulmonary and renal diseases. In the kidney, several GOF mutations in the TRPC6 gene are closely linked to an autosomal-dominant form of FSGS2 (OMIM 603965),ref. bib290,ref. bib291 a rare progressive disease that ultimately leads to kidney failure due to progressive scarring of the glomeruli. Notably, LOF mutations in the TRPC6 channel cause a similar phenotype, particularly in juvenile forms of the disease.ref. bib292 Furthermore, an increased TRPC6 expression compared with healthy individuals was observed in podocytes of patients with diabetic kidney disease.ref. bib293 In the lung, studies using mouse models suggest that TRPC6 is essential for the regulation of hypoxia-mediated pulmonary vasoconstriction and pulmonary hypertension.ref. bib263,ref. bib294 In humans, a single-nucleotide polymorphism (SNP) in the TRPC6 promoter region, which leads to elevated basal TRPC6 expression, is associated with an increased risk of idiopathic pulmonary hypertension.ref. bib171,ref. bib295 Subsequently, Pousada et alref. bib296 identified 3 more TRPC6 SNPs in the 5′-untranslated region of the TRPC6 gene that were significantly more common in a cohort of patients with idiopathic pulmonary hypertension compared with the control group. Several mouse models of heart disease suggest an important role for TRPC3 and TRPC6 channels in the development of cardiac hypertrophy.ref. bib317,ref. bib298 Relating thereto, a study recently demonstrated an association between elevated TRPC6 expression and a higher risk of heart failure after chemotherapy with the cardiotoxic drug doxorubicin.ref. bib299
For TRPC1, TRPC4, and TRPC7 channels, only weak links between human pathologies and channel dysfunction have been reported to date.
Pharmacological modulators of TRPCs
In recent years, the availability of pharmacological modulators of TRPCs has substantially advanced from drugs acting on a range of TRPC isoforms to compounds acting more selectively on distinct TRPC isoforms, with some exceptions; most of the published TRPC blockers still do not sufficiently discriminate between TRPC1/4/5 or TRPC3/6/7. However, combining high-resolution cryo-EM with mutagenesis approaches has recently led to a much better understanding of how drugs modulate TRPC activity, which may facilitate the identification of selective and potent TRPC modulators in the future. Table 2ref. bib18,ref. bib28,ref. bib49,ref. bib201,ref. bib317,ref. bib298,ref. 300., ref. 301., ref. 302., ref. 303., ref. 304., ref. 305., ref. 306., ref. 307., ref. 308., ref. 309., ref. 310., ref. 311., ref. 312., ref. 313., ref. 314., ref. 315., ref. 316., ref. 317., ref. 318., ref. 319., ref. 320., ref. 321., ref. 322., ref. 323., ref. 324., ref. 325., ref. 326., ref. 327., ref. 328., ref. 329., ref. 330., ref. 331., ref. 332., ref. 333. provides an overview of TRPC modulators.
Table 2: Pharmacological modulators of TRPCs.
| Name (PubChem CIDa) | Effect | References |
|---|---|---|
| TRPC1,4,5 | ||
| Inhibitors | ||
| ML204 (230710) | Inhibition mTRPC4 IC50 = 2.9 μM; mTRPC5 IC50 = 10 μM | ref. bib300 |
| AC-1903 (667146) | Inhibition TRPC5 IC50 = 14.7 μM | ref. bib201 |
| Clemizole (2782) | Inhibition mTRPC5 IC50 = 1–1.3 μM; mTRPC4 IC50 = 6 μM | ref. bib301 |
| Duloxetine (60835) | Inhibition hTRPC5 IC50 = 0.54 μM | ref. bib302 |
| Pico145 (85473438) | Inhibition hTRPC4 IC50 = 0.35 nM; hTRPC5 IC50 = 1.3 nM | ref. bib303 |
| HC-070 (85473309) | Inhibition hTRPC4 and hTRPC5 IC50 = 0.35–3.4 nM | ref. bib304 |
| GFB-8438 (138471783) | Inhibition hTRPC5 IC50 = 0.18 μM; hTRPC4 IC50 = 0.29 μM | ref. bib305 |
| Activators | ||
| Riluzole (5070) | Activation mTRPC5 IC50 = 9.2 μM | ref. bib306 |
| Methylprednisolone (6741) | Activation mTRPC5 EC50 = 12 μM | ref. bib307 |
| BTD (46369355) | Activation mTRPC5 EC50 = 1.4 μM | ref. bib307 |
| (–)-Englerin A (46242512) | Activation hTRPC5 EC50 = 7.6 nM; hTRPC4 IC50 = 11.2 nM | ref. bib308 |
| AM237 (90403462) | Activation hTRPC5 EC50 = 15–20 nM | ref. bib309 |
| GFB-887 (N/A) | Inhibition TRPC5, in clinical trials | ref. bib310 |
| BI 1358894 (N/A) | Inhibition TRPC4; TRPC5, in clinical trials | ref. bib311 |
| TRPC3,6,7 | ||
| Inhibitors | ||
| Pyr3 (56964346) | Inhibition mTRPC3 IC50 = 0.7 μM | ref. bib312 |
| Pyr10 (53475435) | Inhibition TRPC3 IC50 = 0.72 μM | ref. bib313 |
| Compound 20 (C20, JW-65) (162659202) | Inhibition hTRPC3 IC50 = 0.37 μM | ref. bib314 |
| 60a (N/A) | Inhibition hTRPC3 IC50 = 90 nM | ref. bib315 |
| GSK2833503A (71818575) | Inhibition rTRPC6 IC50 = 3 nM; rTRPC3 IC50 = 21 nM | ref. bib317,ref. bib316 |
| GSK2332255B (71818573) | Inhibition rTRPC6 IC50 = 4 nM; rTRPC3 IC50 = 5 nM | ref. bib317,ref. bib316 |
| SAR7334 (53378752) | Inhibition hTRPC6 IC50 = 9.5 nM; hTRPC3 IC50 = 282 nM; TRPC7 IC50 = 226 nM | ref. bib318 |
| AM-1473 (167993650) | Inhibition hTRPC6 IC50 = 0.2 nM | ref. bib28 |
| Larixyl acetate (11957828) | Inhibition hTRPC6 IC50 = 0.58 μM; hTRPC3 IC50 = 6.83 μM | ref. bib319 |
| SH045 (134611888) | Inhibition hTRPC6 IC50 = 62 nM; hTRPC3 IC50 = 0.84 μM | ref. bib320 |
| BI 749327 (138377580) | Inhibition mTRPC6 IC50 = 13 nM; orally bioavailable | ref. bib298 |
| BTDM (162423070) | Inhibition hTRPC6 IC50 = 10 nM | ref. bib18 |
| DS88790512 (138319685) | Inhibition hTRPC6 IC50 = 11 nM; orally bioavailable | ref. bib321 |
| BI 764198 (138674835) | Inhibition hTRPC6, in clinical trials | ref. bib322 |
| Activators | ||
| PPZ1 (6462584), PPZ2 (6465626) | Activation mTRPC3/6/7 nonselective | ref. bib323 |
| GSK1702934A (16376051) | activation hTRPC3 EC50 = 80 nM; hTRPC6 EC50 = 440 nM | ref. bib324 |
| Compound 4n (N/A) | Activation hTRPC3 EC50 = 20 nM; mTRPC7 EC50 = 90 nM μM; mTRPC6 EC50 = 1.39 μM | ref. bib325 |
| Artemisinin (68827) | Activation hTRPC3 EC50 = 30–50 μM | ref. bib326 |
| AM-0883 (145997911) | Activation hTRPC6 EC50 = 46 nM | ref. bib28 |
| M085 (N/A) | Activation hTRPC6, mTRPC6 EC50 = 3.8 μM | ref. bib327 |
| C20 (N/A) | Positive allosteric modulator TRPC6 | ref. bib328 |
| PhoDAG-1 (121225613)PhoDAG-3 (121225610) | Photoswitchable activator hTRPC6, mTRPC6; mTRPC2 | ref. 328., ref. 329., ref. 330. |
| OptoDArG (131954527) | Photoswitchable activator hTRPC3; mTRPC6 | ref. bib49,ref. bib331 |
| OptoBI-1 (146018968) | Photoswitchable activator hTRPC3; mTRPC6, hTRPC6, hTRPC7 | ref. bib331,ref. bib332 |
| BTDAzo (N/A) | Photoswitchable activator mTRPC5 | ref. bib333 |
| dfdc-OptoBI-1 (N/A) | Photoswitchable activator mTRPC6 | ref. bib334 |
a PubChem Compound Identification number. N/A – not available.
Inhibitors of TRPC1/4/5 channels
The first identified inhibitors of TRPC4 and TRPC5 channels discriminated poorly between the 2 isoforms and were of low potency. ML204 inhibits TRPC4 channels (IC50 = 2.9 μM) with a 3-fold preference over TRPC5 (IC50 = 10 μM) and a 19-fold selectivity over TRPC6.ref. bib300 In a transgenic rat model of FSGS with podocyte-specific overexpression of the angiotensin II AT1 receptor, intraperitoneal application of ML204 suppressed proteinuria and prevented podocyte loss.ref. bib201 AC-1903, which inhibits TRPC5 less potently (IC50 = 14.7 μM) but does not inhibit TRPC4 or TRPC6 channels, was also effective in the transgenic rat model mentioned above and in a model of hypertension-induced FSGS (Dahl salt-sensitive rats), reducing proteinuria and protecting podocytes.ref. bib201 However, the pathogenic role of TRPC5 in podocytes was recently called into question.ref. bib335
The antihistamine clemizole displays a 6-fold preference for TRPC5 (IC50 = 1–1.3 μM) over TRPC4 (IC50 = 6 μM).ref. bib301 Cryo-EM has revealed the binding site of clemizole, which is located within the VSLD of TRPC5.ref. bib24 Duloxetine, an antidepressant that is also effective in the treatment of neuropathic pain, inhibits TRPC5 channels (IC50 = 0.54 μM) by fitting into the same binding pocket.ref. bib302
In comparison, the xanthine-based compound Pico145 (HC-068) is considerably more potent, inhibiting TRPC1/4/5 channels with an IC50 of 1.3 nM and 0.35 nM for TRPC5 and TRPC4, respectively.ref. bib303 Its close analog, HC-070, blocks homo- and heteromeric TRPC4/5 channels with IC50 values between 0.3 and 3.4 nM.ref. bib303,ref. bib304 The cryo-EM structure of the human homomeric TRPC5 channels in the presence of Pico145 identified the binding of the drug to lipid binding site 2 between individual TRPC5 subunits, displacing a lipid upon binding of the drug.ref. bib25 This binding site, which is highly conserved within the TRPC family,ref. bib25 was also determined for the Pico145-bound TRPC1/4 heteromer,ref. bib30 and further confirmed by Song et alref. bib24 for the binding of HC-070 to TRPC5. HC-070 is effective in animal models of neurological diseases, as oral administration in mice allows the compound to cross the blood-brain barrier, exerting antidepressant and anxiolytic effects.ref. bib304 Moreover, intraperitoneal administration of HC-070 reverses cognitive and motor deficits in rat models of Parkinson’s disease.ref. bib336,ref. bib337
Screening of a 400,000-compound library and subsequent hit optimization led to the discovery of several pyridazinone-based inhibitors, with GFB-8438 being the most promising regarding its physicochemical properties. GFB-8438 inhibits TRPC5 (IC50 = 0.18 μM) with a similar potency to TRPC4 channels (IC50 = 0.29 μM).ref. bib305 Cryo-EM studies performed on TRPC4 homomers demonstrated the binding of GFB-8438 and closely related compounds, GFB-9289 and GFB-8749, to the VSLD of TRPC4.ref. bib21 In the deoxycorticosterone acetate-salt rat model of hypertension and renal inflammation, GFB-8438 exerts nephroprotective effects, evident by reduced protein and albumin concentrations in the urine.ref. bib305
Inhibitors of TRPC3/6/7 channels
TRPC3 is, at least within the TRPC family, selectively inhibited by pyrazole compounds Pyr3 and Pyr10 (IC50 = ∼0.7 μM).ref. bib312,ref. bib313 In vivo, Pyr3 reduces cardiac hypertrophy and transition to heart failure in mice subjected to pressure overload,ref. bib312 whereas Pyr10-mediated TRPC3 inhibition alleviates systemic inflammatory responses in mice after treatment with lipopolysaccharide.ref. bib197 However, both drugs also inhibit ORAI1 channels, obscuring the attribution of their beneficial effects to individual channel blockage.ref. bib313 Structural optimization of Pyr3 results in the development of compound C20 (JW-65), a derivative with increased metabolic stability and low toxicity, which retains similar potency for TRPC3 inhibition (IC50 = 0.37 μM), while exhibiting improved selectivity over ORAI1 channels.ref. bib314 Based on the same lead structure, compound 60a, with a 4-fold improvement in potency, was later synthesized.ref. bib315
The aminothiazole GSK2833503A (GSK503A) potently inhibits TRPC3 and TRPC6 with a higher selectivity for TRPC6 (IC50 = 3 nM) over TRPC3 (IC50 = 21 nM), whereas GSK2332255B (GSK255B) inhibits both TRPC3 and TRPC6 with a similar potency (IC50 = 3–4 nM).ref. bib317,ref. bib316 Both drugs reduce hypertrophy and fibrosis in a model of cardiac hypertrophy, possibly by acting on both channels.ref. bib317
SAR7334 was identified through a pharmacophore-guided design of aminoindanol derivatives based on the broad TRP channel blocker SKF96365. SAR7334 predominantly inhibits TRPC6 but also TRPC3 and TRPC7 channels with IC50 values of 9.5, 282, and 226 nM, respectively, and suppresses hypoxic pulmonary vasoconstriction in explanted mouse lungs exposed to hypoxic conditions.ref. bib318 Based on SAR7334, the most potent and selective TRPC6 inhibitor to date, AM-1473, was developed (IC50 = 0.2 nM),ref. bib28 which binds to a pocket formed by the cytoplasmic portions of S1–S4 and the TRP helix.ref. bib28
Larixyl acetate, a diterpenoid from larch resin, primarily inhibits TRPC6 channels with a 10-fold selectivity for TRPC6 (IC50 = 0.6 μM) over TRPC3. It effectively prevents acute hypoxia-induced vasoconstriction in isolated lungs from miceref. bib319 and offers protection against pressure overload-induced heart failure.ref. bib338 Subsequent structural optimization yielded the methylcarbamate derivative SH045 with an improved potency for TRPC6 (IC50 = 62 nM). SH045 reduced edema in an animal model of lung IRref. bib320 and ameliorated renal fibrosis in obese mice after unilateral ureteral obstruction.ref. bib339
BI 749327 is an orally bioavailable TRPC6 blocker (IC50 = 13 nM) with high selectivity.ref. bib298 Due to its favorable physicochemical properties, the compound has been tested in several animal models. The administration of BI 749327 improved heart function and reduced fibrosis in mice subjected to pressure overload, and reduced renal fibrosis in a renal injury model.ref. bib298 Moreover, in a mouse model of severe Duchenne muscular dystrophy, TRPC6 inhibition by BI 749327, starting from day P3, improved skeletal and cardiac muscle function and survival in mice.ref. bib340
Other highly potent TRPC6 inhibitors include the high-affinity TRPC6 inhibitor BTDM (IC50 = 10 nM), which binds at the interface between the pore and VSLD,ref. bib18 and orally bioavailable DS88790512 (IC50 = 11 nM).ref. bib321 However, neither of these compounds has been tested in vivo so far.
Activators of TRPC4/5 channels
Riluzole, which is the only Food and Drug Administration (FDA)-approved drug to treat amyotrophic lateral sclerosis, activates TRPC5 with low potency (EC50 = 9.2 μM) but is, at least within the TRPC family, specific for TRPC5.ref. bib306 A screening approach by Beckmann et alref. bib307 identified methylprednisolone (EC50 = 12 μM) and the benzothiadiazine derivative (BTD) (EC50 = 1.4 μM) as novel TRPC5 agonists. Notably, BTD alleviated mechanical allodynia in diabetic peripheral neuropathic rats, presumably via the downregulation of TRPC5 expression and anti-inflammatory and antiapoptotic effects of BTD.ref. bib217
(–)-Englerin A, derived from the bark of the Phyllanthus engleri tree, displays the highest potency and efficacy and activates both TRPC4 and TRPC5 in low nanomolar concentrations (EC50 = 11.2 nM and 7.6 nM for TRPC4 and TRPC5, respectively).ref. bib308 However, (–)-englerin A is lethal in rodents when administered at concentrations near those required to activate TRPC4, likely due to excessive TRPC4 activation leading to pulmonary edema.ref. bib341
Following up on the structure of the TRPC1/4/5 blocker Pico145, Minard et alref. bib309 recently synthesized the analog AM237, which potently activates homomeric TRPC5 (EC50 = 15–20 nM) but not heteromeric TRPC1/5, TRPC4/5, or homomeric TRPC4 channels.
Activators of TRPC3/6/7 channels
Small molecules that activate TRPC3 channels include piperazine-derived compoundsref. bib323 PPZ1 and PPZ2, which do not discriminate well between TRPC3/6/7 and GSK1702934A, a potent TRPC3/6 activator (EC50 = 80 and 440 nM for TRPC3 and 6, respectively).ref. bib324 Qu et alref. bib325 developed a series of pyrazolopyrimidine-derived TRPC3/6/7 agonists with a preference for TRPC3, of which compound 4n was the most potent (EC50 = 20 nM). The antimalarial drug artemisinin activates TRPC3 with low potency (EC50 = 30–50 μM) but with a high preference for TRPC3 over TRPC6 and TRPC7.ref. bib326
The TRPC6 activator AM-0883 is highly potent (EC50 = 46 nM) with a binding site between the PH and the S6 helix of the adjacent subunit,ref. bib28 which is similar to that of HC-70 and Pico145 in the TRPC1/4/5 channels. The same binding site is targeted by structurally distinct TRPC6 activators M-085 (EC50 = 3.8 μM) and GSK1702934A.ref. bib327 In addition to TRPC6 channel activators, the substance C20 was identified, which acts as a positive allosteric modulator, enabling TRPC6 current increases in the presence of 1-oleoyl-2-acetyl-sn-glycerol.ref. bib328
Optical control of TRPCs
Recently, compounds have been developed for the precise optical control of TRPCs. These photoswitches are generated by linking a light-sensitive azobenzene moiety to a known channel modulator, enabling light of distinct wavelengths to switch the compound’s activity on and off. Photoswitchable DAGs, such as PhoDAGref. bib329 and OptoDArG,ref. bib49 are used to rapidly activate DAG-sensitive TRPCs: photoswitchable DAGs are switched on to their active cis-isomer upon exposure to 370 nm UVA light and off through trans-isomerization at 460 nM. In the cis configuration, PhoDAG1 and the more membrane-permeant PhoDAG3 activate TRPC2 in mouse vomeronasal sensory neurons.ref. bib330,ref. bib342 Cis-PhoDAG1 also activates heterologously expressed TRPC6 channels.ref. bib330,ref. bib331,ref. bib342 Another photoswitchable DAG, OptoDArG, features 2 photoswitchable azobenzene moieties and is active in the cis-form at 365 nm and inactive at 430 nm. OptoDArG enables optical control of TRPC2, TRPC3, and TRPC6 channels upon photoisomerization.ref. bib49,ref. bib331,ref. bib342
Based on the TRPC3/6 agonist GSK1702934A, Opto-BI-1 was developed to enable optical control of TRPC3 channels in human vascular endothelial cells and mouse hippocampal neurons,ref. bib332 as well as the precise control of TRPC6 channel function.ref. bib331 More recently, BTD served as a starting point for the generation of the photoswitchable TRPC5 agonist BTDAzo, which can control TRPC5 channels in isolated cells and mouse brain slices (EC50 = 1.5 μM).ref. bib333 In the future, it will be fascinating to explore whether photoswitchable TRPC modulators can also be applied in vivo. A crucial step toward the in vivo application of photopharmaceuticals is the development of red-light switchable compounds, such as the recently developed dfdc-OptoBI-1.ref. bib334 Red light is nonphototoxic and offers greater tissue penetration, making it particularly suitable for biomedical applications.
Ongoing or completed clinical trials with TRPCs as therapeutic targets
To date, only a few clinical trials have been initiated that use small molecules targeting TRPCs. Considering their prominent role in lung and kidney diseases, TRPC5 and TRPC6 have emerged as the most promising therapeutic targets. The TRPC5 inhibitor GFB-887 is well tolerated in healthy patients (phase 1 study; NCT03970122).ref. bib310 It was further tested in patients with FSGS (NCT04950114) and those suffering from diabetic nephropathy or FSGS (NCT04387448) to evaluate the possible beneficial effect of GFB-887 on kidney function. However, both studies were terminated due to business reasons, and no results have been published to date.
The TRPC4/5 channel inhibitor BI 1358894 has recently been explored as a potential treatment for psychiatric disorders, including depression and anxiety. Its safety, tolerability, and pharmacokinetics were demonstrated in 2 phase 1 studies (NCT03210272 and NCT03754959).ref. bib343 However, phase 2 trials assessing the efficacy of BI 1358894 in patients with major depression (NCT04423757), post-traumatic stress (NCT05103657), and borderline personality disorders (NCT04566601) did not show efficacy of the drug.ref. bib344,ref. bib345 Nonetheless, the outcome of another phase 2 trial (NCT04521478) investigating its efficacy in patients with major depression who showed an inadequate response to standard antidepressants is still awaited.
The TRPC6 inhibitor BI 764198 was well tolerated in 4 phase 1 studies (NCT03854552, NCT04102462, NCT04656288, and NCT04176536) and is currently being investigated in individuals with FSGS in a phase 2 trial (NCT05213624).ref. bib346 In another phase 2 trial (NCT04604184), the same drug failed to reduce the risk and/or severity of acute respiratory distress syndrome during the course of the COVID-19 disease.ref. bib322 Additionally, an observational study (NCT05507879) is currently exploring whether TRPC6 variants can predict chemotherapy-related cardiomyopathy and heart failure in breast cancer patients.
TRPVs
TRPV gene family
The TRPV gene family consists of 6 distinct members: TRPV1–6 (Table 1), which can be categorized into 2 main subgroups based on their homology and functional characteristics: the thermosensitive channels TRPV1–4, which are nonselective for monovalent cations, and the Ca2+-selective channels TRPV5 and TRPV6 (reviewed in Vennekens et alref. bib347) (Fig. 3A).
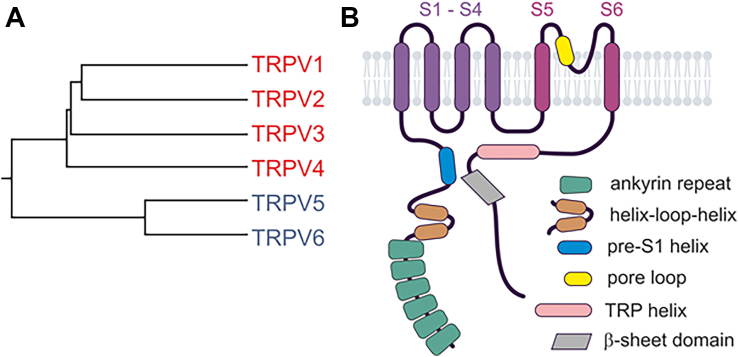
TRPVs, especially TRPV1, have been extensively studied and have emerged as promising drug targets for treating various human diseases. In this chapter, we will provide an overview of the key advancements in understanding TRPV characteristics and their roles in health and disease. We will introduce and discuss modulators of TRPVs and their applicability in animal and human disease models. However, due to their abundance, the focus will be on compounds that have already been well validated and are characterized by relatively high specificity and potency.
Domain topology, assembly, and functional characteristics of TRPVs
Domain topology of TRPVs
Cryo-EM and crystallographic studies have provided structures for all homotetrameric mammalian TRPV complexes, including numerous structures in their ligand-bound or CaM-bound states. Overall, the available homotetrameric TRPV structures display a rotationally symmetric subunit assembly with protein moieties mostly extending into the cytoplasmic space and only minor extracellular bulges formed by short loops that connect the TM-spanning segments S1–S6 and the pore loop, which is intercalated between S5 and S6 (Fig. 3B).
Several expert reviews have elaborated on common or distinct structural features of specific TRPV isoforms and their contribution to the regulatory and biophysical properties of the respective channels.ref. 347., ref. 348., ref. 349., ref. 350., ref. 351., ref. 352., ref. 353., ref. 354., ref. 355., ref. 356., ref. 357. Table 1 summarizes the available PDB entries for TRPVs in their apo or ligand-bound states.
The intracellular N- and C-termini of TRPVs are joined by TMDs that are organized in a similar fashion as in TRPCs. A bundle composed of the first 4 TM-spanning helices forms a VSLD, which connects via the α-helical S4–S5 linker to a second fold, consisting of S5, a re-entrant short pore loop, and S6. The N-termini of TRPVs contain an ARD with 6 consecutive ankyrin repeats that, in some but not all TRPV isotypes, engage in contact with neighboring subunits. The ARD is followed by 2 α-helices that are referred to as the helix-loop-helix linker domain and a pre-S1 α-helix.
Forming a sharp turn, the S6 of the TMD is connected to the cytosolic C-terminus by a TRP domain, which contains an α-helix that is oriented parallel to the plasma membrane, and intimately contacts the S4–S5 linker as well as the pre-S1 helix, and is supposed to undergo a rotational movement during channel opening.ref. bib359,ref. bib360 Following the TRP helix, a β-sheet–containing domain engages in additional interactions with the N-terminus of the same channel subunit.
Structural motifs within the permeation pathway include a cone-shaped vestibule at the outer mouth of the pore, which exposes negatively charged amino acids and may attract cations, engage in salt bridges with neighboring subunits, or become protonated under acidic extracellular conditions, eg, in inflamed tissues.ref. bib361,ref. bib362 Notably, the strong electronegativity of the vestibule of TRPV6 has been proposed to resemble that of Drosophila Orai, thereby providing a common mechanism for divalent-selective permeation.ref. bib363
The selectivity filter of the TRPV1–4 subgroup shares an I-G-M/L-G-D/E motif, whereas the Ca2+-selective TRPV5 and TRPV6 channels display a distinct L-T-V/I-I-D amino acid sequence. The latter is located in the second part of the pore loop, which is centrally positioned and kinks back from a short PH to form an outward-pointing vertical stack of amino acids that narrows down the entry pathway and coordinates influxing cations in 1 or 2 sites before releasing them to another coordination site located within an inner cavity that leads to the lower gate.ref. bib364,ref. bib365 In all human TRPVs, the outer vestibule narrows down to the selectivity filter with an aspartic acid (or glutamic acid in TRPV2), possibly expelling anions, and representing a first landing platform-like coordination site for influxing cations in the upper part of the selectivity filter. Since neutralization of this anionic amino acid in TRPV1 or TRPV4 not only reduces the permeability of divalent cations, but also lowers the potency of ruthenium red-mediated channel block, it has been recognized early as part of the binding site of the polycationic open pore blocker,ref. bib366,ref. bib367 a concept that has been confirmed by structural analyses.ref. bib368 In the Ca2+-selective isoforms TRPV5 and TRPV6, the upper coordination site may bind divalent cations more tightly, thereby causing longer occupancy times and contributing to repulsive forces between stacked divalent cations that would allow a “knock-on” mechanism of Ca2+-selective permeation.ref. bib365 The second coordination site, formed in the central and lower parts of the selectivity filter, opens toward an inner cavity that is flanked by residues and backbone carbonyls within S6. Owing to their inverted teepee-like helix bundling and crossing, the S6 segments constrict the pore diameter to form the inner gate. The opening of the inner gate involves reorientation within the S6, characterized by a partial α– to π-helical rearrangement, which allows rotation of the lower part of S6.ref. bib362,ref. bib369 Depending on the channel isoform and the applied activators, TRPVs can adopt several open states, some of which feature a pore radius of more than 2 Å, allowing the permeation of large organic cations, such as NMDG+, YoPro-1, MEQ+, or QX-314.ref. 369., ref. 370., ref. 371., ref. 372. Like in TRPCs, the VSLD of TRPVs can harbor lipids in positions that, in some cases, overlap with ligand binding sites.ref. bib353,ref. bib369,ref. 373., ref. 374., ref. 375., ref. 376.
Assembly of TRPV complexes
Structural, biophysical, and functional analyses have demonstrated that all TRPV isoforms are capable of forming homomeric complexes, yielding functionally active cation channels. Heteromeric TRPV assemblies can form between the closely related isoforms TRPV5 and TRPV6, but also between TRPV1 and TRPV2.ref. 377., ref. 378., ref. 379. Other studies found a more promiscuous pattern of heteromerization between the heat-sensitive TRPV1–4 subunits,ref. 380., ref. 381., ref. 382. or heteromeric complexes across different TRP channel families, such as TRPV1 and TRPA1,ref. bib384 TRPV2 and TRPP2,ref. bib385 TRPV4 and TRPC1ref. bib145,ref. bib386 or TRPC1/TRPP2,ref. bib387 TRPV5 and TRPML3,ref. bib388 or TRPV6 and TRPC1.ref. bib59 At present, the relevance of heteromeric TRPV complexes is not yet sufficiently understood, and heteromer-specific pharmacological tools are lacking.
As an exception to the rule that TRP channels assemble as tetramers, a small fraction of purified and reconstituted TRPV3 channel subunits has been shown to transiently engage in a noncanonical pentameric assembly when studied by high-speed atomic force microscopy under specific stimulation conditions, and its properties as a dilated pore conformation have been proposed.ref. bib389 The proof that electrophysiological single channel recordings actually show currents through a pentameric TRPV3 complex is, however, lacking, and the experimental settings have been critically commented.ref. bib390
Finally, assemblies between TRPVs and auxiliary subunits or temporary interaction partners may contribute to the regulation of channel activity, plasma membrane targeting, internalization, or degradation. The plasma membrane stability and lifetime of TRPV1 have been found to be positively modulated by physical interaction with the toll-like receptor 4,ref. bib391 and by the interaction with Kvβ1, a non–pore-forming subunit of voltage-gated potassium channels.ref. bib392 Functional interactions between TRPV1, TRPV3, and TRPV4 with associated anoctamin 1, a Ca2+-activated Cl– channel, have been found to enhance capsaicin-evoked nociception, promote wound healing, secretion from exocrine glands, and release of vasodilatory factors from endothelial cells.ref. bib393 In keratinocytes, TRPV3 has been shown to form a complex with the EGF receptor, which is associated with a mutual augmentation of functional activities.ref. bib394 Another interaction of TRPV3 with TMEM79 was demonstrated to decrease the plasma membrane abundance of TRPV3 by promoting its degradation.ref. bib395 Likewise, trafficking of TRPV2 and TRPV4 has been proposed to be regulated by their respective interactomes, as recently reviewed.ref. bib396 TRPV4 physically interacts with the small GTPase RhoA, which dampens the TRPV4 activity unless disease-related mutations in either TRPV4 or RhoA prevent their assembly.ref. bib397,ref. bib398
Physical interactions of TRPV5 channels involve the catalytic processing by the extracellular β-glucuronidase klotho,ref. bib399 and an intracellular interaction with the Ca2+-buffering protein calbindin-D(28K).ref. bib400 A serine-threonine kinase with-no-lysine 4-dependent forward trafficking from the Golgi apparatus to the plasma membrane has been described as a result of fibroblast growth factor-23 signaling.ref. bib401 The apical plasma membrane trafficking of TRPV5 may be further stabilized by interactions with the multi-PDZ domain protein NHERF2,ref. bib402 and by a second noncatalytical function of the soluble extracellular domain of α-klotho to connect TRPV5 with the membrane protein galectin-1.ref. bib403 Both TRPV5 and TRPV6 have been shown to interact with Rab11a, which targets the channels to the plasma membrane,ref. bib404 where they might be concentrated in the apical membrane of polarized cells by interacting with the PDZ domain-bearing scaffolding protein NHERF4.ref. bib405 Finally, a physical interaction of TRPV6 with the protein tyrosine phosphatase PTP1B has been shown to remove a Src-mediated tyrosine phosphorylation, thereby dampening the channel activity when studied in a heterologous expression system.ref. bib406
In the future, more studies of TRPV interactomes, applying quantitatively accurate and unbiased methodologies like those recently presentedref. bib62 for TRPC1, may provide additional hypotheses to unravel functionally relevant macromolecular assemblies involving TRPVs.
Functional characteristics of TRPV complexes
Based on sequence homology and functional properties, TRPVs can be subdivided into 2 subgroups. The TRPV1–4 subgroup forms warmth- or heat-activated, Ca2+-permeable, but poorly selective (pCa/pNa = 2–10) cation channels that typically share an outwardly rectifying current voltage relationship. By contrast, TRPV5 and TRPV6 form Ca2+-selective channels that are at least, to some degree, spontaneously active, but not activated by heat, and give rise to inwardly rectifying ionic currents.
The founding member, TRPV1, is a prototypical temperature sensor that is further sensitized by inflammatory mediators, chemical ligands, or low extracellular pH. The heat- or capsaicin-induced gating mechanism of TRPV1 is characterized by a uniquely large shift of its voltage-dependent activation curve from nonphysiological positive potentials to more negative potentials.ref. bib407 Since large shifts in half-maximally activating membrane potentials are a common feature within thermally activated TRP channels, including TRPV3, TRPM4, and TRPM8, and since activating ligands can cause similar shifts in these channels, an atypical voltage sensor with a small gating charge has been proposed to integrate various inputs by shifting the window of voltage-dependent gating into the range of physiological resting membrane potentials.ref. bib408
The temperature thresholds of heterologously expressed human TRPV1–4 channels observed at membrane potentials of –60 mV to –100 mV in quiescent cells scale between mild warmth of 23–39 °C for TRPV3 and TRPV4,ref. bib409,ref. bib410 to moderate heat of above 43 °C for TRPV1.ref. bib411 Other than the rat or mouse TRPV2 orthologs, which are activated at noxious hot temperatures of >52 °C, human TRPV2 has been found to be heat-insensitive.ref. bib412,ref. bib413 Notably, TRPV1 orthologs that are isolated from species adapted to lower or higher temperatures can display corresponding changes in temperature-dependent channel gating, with higher temperature thresholds found in camel or ground squirrel TRPV1,ref. bib414 while TRPV1 in amphibians or zebrafish is tuned to lower temperatures.ref. 414., ref. 415., ref. 416. These thresholds shall not be considered as absolute values because they are voltage-dependent and modulated by ligands, second messengers, or during repeated activation cycles. Conditions that mimic inflammation, such as stimulation of PLC- and PKC-coupling bradykinin receptors or cAMP-elevating prostaglandin receptors or extracellular acidification, lower the temperature threshold for TRPV1 activation. Similarly, capsaicinref. bib418 or piperine,ref. bib419 the pungent ingredients of chili pepper and black pepper, respectively, ethanol,ref. bib420 or certain spiderref. bib421,ref. bib422 and scorpionref. bib423,ref. bib424 venoms shift the activation threshold to temperatures that are well below physiological values. Finally, anandamide and structurally related endovanilloids act as activators or positive modulators of TRPV1 channel activity.ref. bib425 Hence, TRPV1 is a polymodal sensor that integrates physiological, pathophysiological, and alimentary or toxic stimuli. Similar changes in temperature thresholds have been reported for repeatedly activated TRPV3 with and without additional sensitization by cholesterol supplementation.ref. bib409,ref. bib426,ref. bib427
Since local temperatures are elevated by inflammation-associated hyperemia, TRPV1 strongly contributes to constant pain sensation and confers a major component of thermal inflammatory hyperalgesia. The underlying mechanisms have been studied in impressive detail. Mutagenesis studies have revealed that protonation of E600 in the loop that links S5 with the pore loop is the most likely candidate to initiate TRPV1 sensitization in tissue acidosis.ref. bib361 In sensory neurons, TRPV1 modulation via stimulation of G protein-coupled receptors (GPCRs) can either enhance or mitigate thermal or capsaicin-induced responses. While Gq-coupled B1 bradykinin and EP1 prostaglandin receptors, as well as Gs-coupled EP4 or IP prostaglandin receptors, sensitize TRPV1 to lower temperatures,ref. 427., ref. 428., ref. 429. Gi-coupled μ opioidref. bib431 or GABAB1 receptorsref. bib432 dampen the TRPV1 channel activity.
When strongly activated for longer time periods, TRPV1–4 channels tend to change their permeation properties, allowing penetration of organic cations. This behavior has been referred to as pore dilation, as recently reviewed.ref. bib433 It is of pharmacological interest that large pore diameters found in TRPV1 allow the penetration of cationic tool compounds and drugs, such as the organic cation NMDG+, the quinolinium-based chloride indicator dyes MEQ+ and MQAE+, the DNA stain YoPro-1, and even local anaesthetics.ref. bib370,ref. bib372,ref. bib434 While some observations and conclusions may be restricted to prolonged activation of strongly overexpressed channels in heterologous expression systems, leading to unwanted changes in intracellular cation concentrations,ref. bib435 the development of large TRPV1 pore diameters that allow the permeation of organic cations has been confirmed by structural biology approaches.ref. bib362,ref. bib436
In stark contrast to TRPV1–4, TRPV5 and TRPV6 are highly selective for Ca2+ with pCa/pNa > 100 for both channels,ref. bib437,ref. bib438 indicating their specialized function in cellular Ca2+ transport. However, under divalent-free conditions, they become permeant to monovalent cations,ref. bib437,ref. bib439 such as Na+ and K+. The regulation of TRPV5 and TRPV6 activity involves various intracellular signaling pathways and extracellular factors, whereby PIP2 and extracellular Ca2+ play a decisive role. TRPV5 and TRPV6 are constitutively active in the presence of PIP2, which stabilizes the channel in its open configuration.ref. bib440,ref. bib441 Both channels are also sensitive to extracellular pH, with protons acting as potent inhibitors,ref. 441., ref. 442., ref. 443. and recently, cryo-EM structures revealed that the proton-dependent block of TRPV5 is caused by a disruption of the PIP2 binding pocket, thereby preventing PIP2 binding to TRPV5.ref. bib445
TRPV5 and TRPV6 undergo rapid inactivation in the presence of high intracellular Ca2+ concentrations, which allows both proteins to dynamically adjust the Ca2+ content of the cell, thereby preventing excessive Ca2+ influx and maintaining cellular calcium homeostasis. This inactivation is mainly caused by the binding of Ca2+-CaM to the channel protein.ref. bib446,ref. bib447 Cryo-EM studies of TRPV5 and TRPV6, together with Ca2+-CaM, have revealed that upon binding of Ca2+-CaM to the C-terminal regions of the channel, it sterically inhibits the ion-conducting pore.ref. 447., ref. 448., ref. 449. The basal activity of both channels is, therefore, largely determined by the interplay between PIP2-dependent activation and Ca2+-CaM–dependent inactivation.ref. bib354,ref. bib451
In electrophysiological recordings, the current voltage curve of TRPV5 and TRPV6 displays a marked inward rectification, which is a hallmark of Ca2+ channels and can be partly attributed to the inhibition by intracellular Mg2+ via a mechanism that has yet to be clarified.ref. bib358,ref. bib440,ref. bib452 In conclusion, the biophysical properties of TRPV5 and TRPV6, including their high Ca2+ selectivity, constitutive activity, and regulation by CaM and calciotropic hormones, underscore their importance in maintaining cellular and organismic calcium homeostasis.
Expression pattern and primary physiological roles of TRPVs
Numerous studies have investigated the mRNA and protein expression of TRPVs in a variety of species using different methodologies, which have sometimes yielded inconsistent findings. This chapter focuses on TRPV expression in human tissues and includes data from the Human Protein Atlas and single-cell transcriptomic analyses.ref. bib453,ref. bib454 An overview of the expression profile of TRPVs is provided in Table 1.
The most prominent expression of TRPV1 is found in nociceptive neurons whose somata are localized in the dorsal root ganglia (DRG) and in the trigeminal ganglion.ref. 454., ref. 455., ref. 456. The fine nerve endings of their dendrites reach the entire skin, the oropharyngeal mucosa, and other internal organs, such as the urinary bladder. Compared with strongly myelinated sensory neurons that confer touch sensitivity, the TRPV1-expressing nociceptive neurons typically have a small or medium diameter and feature either poorly myelinated Aδ fibers or unmyelinated C fibers. In the case of DRG neurons, they terminate in the substantia gelatinosa within the dorsal horn of the spinal cord, where they are connected to the second neuron of the pain pathway and the spinothalamic tract via excitatory glutamatergic synapses. The primary afferent function of TRPV1 channels is to confer heat perception, thermal nociception, and the pungent or “hot” sensation of various alimentary spices. In diseased states that trigger inflammation, TRPV1 can become strongly sensitized and chiefly mediates inflammatory thermal hyperalgesia and constant pain sensations.ref. 457., ref. 458., ref. 459.
Importantly, TRPV1-expressing nociceptive neurons also exert a pseudo-efferent function by releasing the strongly vasodilatory calcitonin gene-related peptide and the inflammation-mimicking peptide substance P from free nerve endings, which contribute to thermoregulation by enhancing cutaneous blood flow and passive heat dissipation.ref. bib425,ref. bib461 Since TRPV1 is activated by warmth or moderate heat, this feedback mechanism is ideally suited to maintain body temperature within a narrow range while not yet losing significant amounts of water and electrolytes, which would be the consequence of sweating. Notably, most TRPV1 inhibitors also disrupt this thermoregulatory function. Consequently, the adverse effects of analgesic TRPV1-targeting drugs not only include burning or scalding injuries, but also a significant elevation of body temperature.ref. bib460,ref. bib462,ref. bib463 In the brain, TRPV1 expression is found in neurons, astrocytes, and microglia.ref. bib464
TRPV2 has initially been identified in DRG neurons as well, but the TRPV2-positive neurons are larger in diameter and poorly overlap with the population of TRPV1-expressing neurons.ref. bib412 Later, the expression of TRPV2 has been found to be much more widespread, with the strongest expression in various immune cells, including macrophages, monocytes, neutrophils, T lymphocytes, mast cells, and dendritic cells.ref. 464., ref. 465., ref. 466. In the CNS, TRPV2 is also strongly expressed in a wide variety of excitatory or inhibitory neurons and in the microglia. According to single-cell transcriptomic analyses, an abundant expression of TRPV2 is found in tissue-resident immune cells, such as lung macrophages, placental Hofbauer cells, as well as nonimmune cells, such as cutaneous melanocytes, vascular smooth muscle cells, the urothelium, and red blood cells.ref. bib454,ref. bib468,ref. bib469
Despite its high abundance in various cell types, the primary function of TRPV2 is still poorly understood. No obvious thermal or mechanical nociceptive sensory phenotype has been detected in mice lacking TRPV2 expression.ref. bib470 In agreement with the strong TRPV2 expression in cell types of the innate and adaptive immune system, phenotypes are more prominent upon immunological challenges. In macrophages, TRPV2 activity is critical for efficient cell migration, phagocytosis, and bacterial clearance.ref. bib471,ref. bib472 Similarly, TRPV2-deficient mice displayed attenuated B-cell responses and antibody formation upon immunization.ref. bib473
The most prominent site of TRPV3 expression is found in basal and suprabasal cutaneous keratinocytes, as well as in epithelial cells of the hair follicles.ref. bib426,ref. bib474 TRPV3 expression has also been detected in sensory DRG and trigeminal ganglion neurons,ref. bib409,ref. bib475 but based on KO mouse models, the functional role of TRPV3 as a primary sensor for warmth or heat perception has been controversial.ref. bib476,ref. bib477 Since TRPV3, like TRPV4, contributes to warmth-induced ionic currents in keratinocytes, a functional link to sensory neurons may involve the formation or release of paracrine factors such as ATP, prostaglandin E2, nitric oxide, or transforming growth factor-α to transmit the signals to sensory neurons.ref. bib394,ref. 477., ref. 478., ref. 479., ref. 480., ref. 481. TRPV3 expression has also been demonstrated in epithelial tissues of the oral cavity, in glandular cells of the small intestine, and in enterocytes of the small and large intestine.ref. bib454,ref. bib483,ref. bib484 In the CNS, a moderate TRPV3 expression is found dispersed over neuronal and glial cells.
The primary function of TRPV3 is best established in the development and maintenance of intact skin architecture. As GOF mutations in TRPV3 cause hair loss and mutilating keratoderma (see below), and since TRPV3-deficient mice display wavy hairs, curly whiskers, and a partially defective skin barrier,ref. bib394,ref. bib474 undisturbed TRPV3 activity appears indispensable for the proper development of the skin and skin appendages. Consistently, TRPV3 activity has been shown to promote keratinocyte proliferation and migration in vitro, and may therefore support wound healing.ref. bib483,ref. bib485,ref. bib486
Among the heat-sensitive TRPV1–4 channels, the expression of TRPV4 is most widespread. Initially, its expression has been detected in the kidney, lung, trachea, liver, spleen, brain, prostate, and placenta.ref. 486., ref. 487., ref. 488., ref. 489. At the cellular level, TRPV4 is strongly expressed in many human epithelial, glandular, and endothelial cell types, such as in exocrine epithelial cells of the salivary and pancreatic glands, in tracheal, bronchial, and fallopian tube ciliated epithelial cells, in epithelial cells of the choroid plexus, in tubular epithelia of the kidney, in female breasts, in tracheal and tongue glandular cells, in placental trophoblast and decidual cells, in vascular endothelial cells, and in skin keratinocytes and melanocytes, only to name a few.ref. bib453,ref. bib454 High levels of TRPV4 expression have also been found in tissue-resident macrophages, including hepatic Kupffer cells.ref. bib454
In line with the widely distributed expression of TRPV4, manifold primary functions of TRPV4 have been identified. A common motif of some of them is based on the indirect activation of TRPV4 by hypotonic stress, causing the conversion of arachidonic acid to epoxyeicosatrienoic acids that, in turn, activate TRPV4.ref. bib491,ref. bib492 In glandular and exocrine cells, the activation of Ca2+ influx through TRPV4 seems to initiate a secondary opening of anoctamin 1, a Ca2+-regulated chloride channel, to initiate fluid secretion,ref. bib493,ref. bib494 while acute pharmacological activation of TRPV4 in vascular endothelial cells mediates the formation of nitric oxide and triggers microvascular leakage, causing circulatory collapse.ref. bib495 Under more physiological conditions, shear stress can activate endothelial TRPV4 channels, thereby triggering vasodilation and outgrowth of collateral vessels.ref. bib24,ref. bib496,ref. bib497 Finally, TRPV4 plays an important role in development, as pathogenic GOF mutations in human TRPV4 are linked to congenital skeletal and neuromuscular disorders.ref. bib498
TRPV5 is mainly expressed in the apical membrane compartment of epithelial cells of the kidney, distal convoluted tubules (DCTs), and collecting ducts.ref. bib442 In human tissues, TRPV5 transcripts have also been detected in the pancreas, duodenum, jejunum, colon, placenta, prostate gland, testis, brain, and bone osteoclasts.ref. bib499,ref. bib500 Vitamin D-response elements have been identified in the TRPV5 promoter, and TRPV5 protein expression was found to correlate with the expression of other vitamin D receptor target genes in rat kidneys.ref. bib501 Other studies found that TRPV5 expression in mice appeared to be regulatedref. bib502 by Ca2+ rather than by 1,25-dihydroxyvitamin D3 or that TRPV5 expression is also regulatedref. bib503 by estrogens. In DCTs obtained from the kidneys of transgenic reporter mice that express enhanced GFP under the control of a TRPV6 promoter, 1,25-dihydroxyvitamin D3– and parathyroid hormone-dependent transcriptional regulation of TRPV5 was detected.ref. bib504 In the same study, TRPV5 deficiency was shown to strongly impede transepithelial Ca2+ transport, which also represents the primary function of TRPV5. TRPV5 deficiency is associated with severe renal Ca2+ wasting, highlighting the seminal role of TRPV5 in renal Ca2+ reabsorption and bone mineralization.ref. bib505,ref. bib506
Compared with TRPV5, the expression of TRPV6 in mice is more widespread and mostly found in extrarenal tissues and organs. It includes the Ca2+-absorbing epithelia in the small intestine, exocrine and endocrine epithelia of the salivary gland, pancreas, and prostate gland, as well as subsets of epithelial cells in the thyroid, stomach, duodenum, caecum, epididymis, endometrium, placenta, and mucus-secreting epithelia in the main olfactory epithelium and the bronchiae.ref. 506., ref. 507., ref. 508. In human tissues, a similar TRPV6 expression pattern has been found.ref. bib454,ref. 509., ref. 510., ref. 511. Like TRPV5, TRPV6 expression is regulated in a 1,25-dihydroxyvitamin D3-dependent fashion.ref. bib513,ref. bib514 In addition, TRPV6 expression has been shownref. bib502,ref. bib515,ref. bib516 to be upregulated by estrogens and dietary Ca2+.
Notably, TRPV6 expression in polarized epithelia strongly overlaps with that of the vitamin D receptor and other 1,25-dihydroxyvitamin D3-regulated proteins that are involved in transepithelial Ca2+ transport, such as the Ca2+-buffering calbindins D(9k) and D(28k), as well as the plasma membrane calcium ATPase.ref. bib511,ref. bib517 Accordingly, the primary function of TRPV6 is to transport Ca2+ across epithelial barriers. Important transport routes include the 1,25-dihydroxyvitamin D3-dependent regulation of Ca2+ resorption in the small intestine,ref. bib518 fetal Ca2+ supply via placental Ca2+ transport,ref. bib519 and maintenance of fertility by lowering the Ca2+ concentration in the seminal fluid.ref. bib520
Human diseases associated with TRPVs
Although variants in the TRPV1 gene have been identified, they are rare and not commonly associated with human diseases. Katz et alref. bib521 reported the phenotypes of 2 individuals carrying a homozygous missense mutation in the ARD of the channel. This mutation, which leads to a complete loss of TRPV1 activity, causes an elevated heat-pain tolerance and a higher cold-pain threshold. Another study linked 2 independently identified TRPV1 missense variants in individuals to a high risk of malignant hyperthermia.ref. bib522 Other SNPs in TRPV1 were associated with nocturnal, usual, and chronic cough.ref. bib523
An altered TRPV2 expression is mainly associated with the development and progression of several solid tumors and hematological malignancies, as reviewed recently.ref. bib467,ref. bib524 For instance, in triple-negative breast cancer (TNBC), TRPV2 expression correlates with recurrence-free survival of TNBC patients, opening up the possibility that TRPV2 activation, for example, by cannabidiol, might be beneficial as an adjuvant therapy in TNBC.ref. bib525 A similar observation was made in patients suffering from glioblastoma, where TRPV2 expression decreased with disease progression.ref. bib526 In contrast, a higher TRPV2 expression was associated with worse outcomes in multiple myeloma,ref. bib527 prostate cancer,ref. bib528 and gastric carcinoma.ref. bib529
Pathologies arising from TRPV3 dysfunction mainly affect the skin and are strongly associated with itch. The clearest link exists between Olmsted syndrome (OLMS1, OMIM 614594), a rare congenital disorder, and GOF mutations in TRPV3, as demonstrated in a series of clinical reports.ref. bib474 Olmsted syndrome is characterized by palmoplantar keratoderma and periorificial hyperkeratosis, accompanied by severe pruritus and, in extreme cases, spontaneous amputation of fingers or toes. An elevated expression of TRPV3 is also linked to atopic dermatitisref. bib530,ref. bib531 and psoriasis,ref. bib532 inflammatory skin conditions, in which TRPV3 activation might contribute to chronic pruritus. Furthermore, patients with itching scars from burn injuries display increased TRPV3 expression in the epidermis of the affected areas. The role of TRPV3 in pruritus is further highlighted by the fact that topical application of the TRPV3 activator carvacrol causes itching in burn scars.ref. bib533,ref. bib534
Autosomal dominant TRPV4 disorders are primarily associated with skeletal dysplasias or motor function disorders, though phenotypic overlap occurs. In skeletal dysplasia, affected individuals mainly present with brachydactyly, short stature, and progressive scoliosis,ref. bib535 but individual manifestations and severity vary among individuals. More than 50 different TRPV4 mutations have been identified so far,ref. bib536 distributed widely across the gene with a clustering of mutations in the region between TM5 and TM6. Most mutations lead to overactive TRPV4 channels, as seen in autosomal dominant brachyolmia type 3 (OMIM 113500),ref. bib537 metatropic dysplasia (OMIM 156530),ref. bib538 and spondylometaphyseal Kozlowski type dysplasia (OMIM 1842522).ref. bib539 However, some reported mutations also result in a reduced availability of TRPV4 at the plasma membrane, for example, in familiar digital arthropathy-brachydactyly (OMIM 606835).ref. bib540 A TRPV4 mutation with a trafficking defect has also been observed in hereditary motor and sensory neuropathy type IIC (OMIM 606071), also known as Charcot-Marie-Tooth disease type 2C, a neuromuscular disorder mainly characterized by progressive peripheral neuropathy, as well as in congenital distal spinal muscular atrophy (OMIM 600175) and scapuloperoneal spinal muscular atrophy (OMIM 606071).ref. bib541,ref. bib542
Recently, a pathogenic homozygous missense mutation in TRPV5 (V598M) was identified that causes a LOF phenotype associated with a novel form of autosomal recessive hypercalciuria and calcium wasting. The mutation, which affects the TRP helix region, results in protein misfolding and a complete loss of TRPV5-mediated calcium uptake upon overexpression in human embryonic kidney (HEK) 293 cells.ref. bib506
Dysregulation of TRPV6 activity by mutations or abnormal expression levels is linked to several human diseases. Homozygous or compound heterozygous mutations in TRPV6 have been identified in individuals suffering from transient neonatal hyperparathyroidism (OMIM 618188), a condition associated with fetal skeletal abnormalities. Some of the mutations cause TRPV6 trafficking deficits or partial loss of function, which is believed to reduce calcium transport across the placenta, followed by an impaired fetal bone mineralization.ref. bib543,ref. bib544 Functionally deficient TRPV6 variants are also associated with hereditary and familial pancreatitis.ref. bib545,ref. bib546 Moreover, in recent years, several studies have attributed TRPV6 as an oncochannel in cancers of epithelial origin.ref. bib547 In most malignancies, an elevated TRPV6 expression correlates with a more aggressive form of the disease and a higher risk for metastasis, possibly contributing to a poorer prognosis in prostate cancer,ref. 547., ref. 548., ref. 549. breast cancer,ref. bib551,ref. bib552 ovarian cancer,ref. bib553 and pancreatic cancer.ref. bib554 However, additional research is needed to fully understand the mechanisms by which the putative oncochannels TRPV6 and TRPV2 may influence cancer progression and to explore the potential of pharmacological modulation – whether activation or inhibition – as a therapeutic strategy for controlling tumor growth and metastasis in specific cancer types.
Pharmacological modulators of TRPVs
Apart from TRPV1 and, to a lesser extent, TRPV4, the availability of specific and potent TRPV modulators remains limited. While currently available modulators provide valuable tools, their limitations regarding specificity, potency, and toxicity when applied in vivo underscore the need for the development of novel compounds. Table 3ref. bib454,ref. bib455,ref. bib472,ref. bib485,ref. 554., ref. 555., ref. 556., ref. 557., ref. 558., ref. 559., ref. 560., ref. 561., ref. 562., ref. 563., ref. 564., ref. 565., ref. 566., ref. 567., ref. 568., ref. 569., ref. 570., ref. 571., ref. 572., ref. 573., ref. 574., ref. 575., ref. 576., ref. 577., ref. 578., ref. 579., ref. 580., ref. 581., ref. 582., ref. 583., ref. 584., ref. 585., ref. 586., ref. 587., ref. 588., ref. 589., ref. 590., ref. 591., ref. 592., ref. 593., ref. 594., ref. 595., ref. 596., ref. 597., ref. 598., ref. 599., ref. 600., ref. 601., ref. 602. provides an overview of TRPV modulators.
Table 3: Pharmacological modulators of TRPVs.
| Name (PubChem CIDa) | Effect | References |
|---|---|---|
| TRPV1 | ||
| Selected TRPV1 inhibitors | ||
| SB-705498 (9910486) | Inhibition hTRPV1 IC50 = 3–6 nM; in clinical trials | ref. bib555 |
| AMG 517 (16007367) | Inhibition hTRPV1 IC50 = 0.9 nM; in clinical trials | ref. bib556 |
| A-1165442 (46191567) | Inhibition hTRPV1 IC50 = 9 nM for capsaicin activation, partial block of H+ activation | ref. bib557 |
| A-1165901 (171378652) | Inhibition hTRPV1 IC50 = 19 nM for capsaicin activation, potentiation of H+ activation | ref. bib558 |
| AMG8562 (56603667) | Inhibition hTRPV1 IC50 = 1.8 nM for capsaicin activation; IC50 > 10 μM for heat activation, potentiates H+ activation | ref. bib559 |
| NEO6860 (N/A) | Inhibition hTRPV1 IC50 = 41.5 nM for capsaicin activation; IC50 > 4 μM for heat activation; in clinical trials | ref. bib560 |
| PAC-14028 (asivatrep) (56649347) | Inhibition rTRPV1 IC50 = 55 nM; topical application; in clinical trials | ref. bib561 |
| Selected TRPV1 activators | ||
| Capsaicin (1548943) | Activation rTRPV1 EC50 = 0.7 μM; hTRPV1 EC50 = 31.6 nM; in clinical trials | ref. bib455,ref. bib562 |
| Resiniferatoxin (5702546) | Activation rTRPV1 EC50 = 39.1 nM; hTRPV1 EC50 = 4 nM; in clinical trials | ref. bib455,ref. bib562 |
| Anandamide (5281969) | Activation hTRPV1 EC50 = 1.3 μM | ref. bib563 |
| CA-008 (vocacapsaicin) (121349852) | Prodrug of trans-capsaicin; in clinical trials | ref. bib564 |
| TRPV2 | ||
| Inhibitors | ||
| Tranilast (5282230) | Inhibition hTRPV2, mTRPV2 IC50 approx. 10 μM; in clinical trials | ref. bib565 |
| Lumin (23305342) | Inhibition mTRPV2 IC50 = 5 μM | ref. bib565 |
| Valdecoxib (119607) | Inhibition rTRPV2 IC50 = 10 μM | ref. bib566 |
| Monanchomycalin B (102489008) | Inhibition mTRPV2 IC50 = 2.8 μM; hTRPV3 IC50 = 3.2 μM | ref. bib567 |
| B304-1 (N/A) | Partial inhibition mTRPV2 IC50 = 22.2 μM | ref. bib568 |
| B304-2 (N/A) | Partial Inhibition mTRPV2 IC50 = 3.7 μM | ref. bib568 |
| Piperlongumine (637858) | Inhibition hTRPV2 IC50 = 4.6 μM | ref. bib569 |
| SET2 (155541857) | Inhibition mTRPV2 IC50 = 0.5 μM | ref. bib570 |
| IV2-1 (N/A) | Inhibition rTRPV2 IC50 = 6.3 μM | ref. bib472 |
| Activators | ||
| Probenecid (4911) | Activation; in clinical trials | ref. bib571 |
| Cannabidiol (644019) | Activation | ref. bib572 |
| TRPV3 | ||
| Inhibitors | ||
| Citrusinine-II (10016895) | Inhibition mTRPV3 IC50 = 12.4 μM | ref. bib573 |
| Isochlorogenic acid A (6474310) | Inhibition hTRPV3 IC50 = 2.7 μM | ref. bib574 |
| Isochlorogenic acid B (5281780) | Inhibition hTRPV3 IC50 = 0.9 μM | ref. bib574 |
| Osthole (10228) | Inhibition hTRPV3 IC50 = 37 μM | ref. bib575 |
| Forsythoside B (23928102) | Inhibition hTRPV3 IC50 = 6.7 μM | ref. bib576 |
| Verbascoside (5281800) | Inhibition hTRPV3 IC50 = 14 μM | ref. bib577 |
| Alpha-mangostin (5281650) | Inhibition hTRPV2 IC50 = 77 nM; hTRPV2 GOF mutant IC50 approx. 2 μM | ref. bib578 |
| Compound 74a (155184122) | Inhibition hTRPV3 IC50 = 0.38 μM | ref. bib579 |
| Trpvicin (122589101) | Inhibition hTRPV3 IC50 = 0.38 μM; blocks G573S GOF mutant IC50 = 0.66 μM | ref. bib580 |
| Local anesthetics (bupivacaine, mepivacaine, lidocaine, ropivacaine) | Inhibition hTRPV3 low potency (0.17–2 mM) | ref. bib581 |
| Dyclonine (3180) | Inhibition mTRPV3 IC50 = 3.2 μM | ref. bib582 |
| Flopropione (3362) | Inhibition hTRPV3 IC50 = 18 μM | ref. bib583 |
| GRC15300 (N/A) | Inhibition of TRPV3; in clinical trials | Reviewed inref. bib584 |
| Activators | ||
| Naturally occurring monoterpenes (thymol, carvacrol, camphor) | Activation of low potency | ref. bib585 |
| Incensole acetate (73755086) | Activation mTRPV3 EC50 = 16 μM | ref. bib586 |
| Tetrahydrocannabivarin (93147) | Activation rTRPV3 EC50 = 6.1 μM | ref. bib587 |
| KS0365 (N/A) | Activation mTRPV3 EC50 = 5.1 μM (cholesterol-enriched cells) | ref. bib485 |
| TRPV4 | ||
| Inhibitors | ||
| GSK2193874 (53464483) | Inhibition rTRPV4 IC50 = 2 nM; hTRPV4 IC50 = 40 nM | ref. bib588 |
| GSK2798745 (71227359) | Inhibition hTRPV4 IC50 = 1.8 nM; in clinical trials | ref. bib589 |
| HC-067047 (2742550) | Inhibition hTRPV4 IC50 = 48 nM; rTRPV4 IC50 = 133 nM; mTRPV4 IC50 = 17 nM | ref. bib590 |
| RN-1734 (3601086) | Inhibition hTRPV4 IC50 = 2.3 μM | ref. bib591 |
| RN-9893 (121513880) | Inhibition hTRPV4 IC50 = 0.42 μM; rTRPV4 IC50 = 0.66 μM; mTRPV4 IC50 =0.32 μM | ref. bib592 |
| Activators | ||
| GSK1016790A (23630211) | Activation hTRPV4 EC50 = 2 nM; mTRPV4 EC50=2.1 nM | ref. bib593 |
| 36-HCl (N/A) | Activation hTRPV4 EC50 = 60 nM | ref. bib594 |
| RN-1747 (5068295) | Activation hTRPV4 EC50 = 0.77 μM; m/rTRPV4 EC50 = 4 μM | ref. bib591 |
| Curcumin (969516) | Activation is low potency, poor selectivity | ref. bib595 |
| Puerarin (5281807) | Activation is low potency | ref. bib596 |
| TRPV5,6 | ||
| Inhibitors | ||
| Miconazole (4189) | Inhibition, TRPV6 > TRPV5; active >100 μM | ref. bib597 |
| Econazole (3198) | Inhibition, TRPV6 > TRPV5; active >100 μM | ref. bib597 |
| ZINC17988990 (27791261) | Inhibition rbTRPV5 IC50 = 0.11 μM; hTRPV5 IC50 = 0.18 μM | ref. bib598 |
| Compound 3 (N/A) | Inhibition TRPV6 IC50 = 90 μM; TRPV5 IC50 = 503 μM | ref. bib597 |
| cis-22a (169553405) | Inhibition hTRPV6 IC50 = 0.32 μM | ref. bib599 |
| 3OG (N/A) | Inhibition hTRPV6 IC50 = 83 nM; hTRPV5 IC50 = 531 nM | ref. bib600 |
| SOR-C13 (121596688) | Inhibition hTRPV6 IC50 = 14 nM; in clinical trials | ref. bib601 |
| SOR-C27 (N/A) | Inhibition TRPV6 IC50 = 64 nM | ref. bib601 |
| Tetrahydrocannabivarin (93147) | Inhibition rTRPV5 IC50= 4.8 μM; mTRPV6 IC50 = 9.4 μM | ref. bib602 |
| Compound 9e (N/A) | Photoswitchable inhibitor TRPV6 | ref. bib603 |
a PubChem Compound Identification number. N/A – not available.
TRPV1
Over the past 20 years, numerous TRPV1-modulating compounds, inhibitors, and activators have been introduced, primarily with the intention of treating diverse pain conditions. As a highly druggable target, TRPV1 has attracted considerable research interest, resulting in an abundance of selective and potent modulators. They will be only briefly summarized here, and we refer to current reviews for more detailed information.ref. bib604
First-generation TRPV1 inhibitors, such as SB-705498 or AMG 517,ref. bib555,ref. bib556 are polymodal antagonists of TRPV1 that block activation by capsaicin, protons, and heat, as reviewed by Garami et al.ref. bib463 However, many of these compounds cause hyperthermia in vivo and reduce the perception of noxious heat, resulting in burn injuries (reviewed by Romanovsky et alref. bib462). To address these issues, second-generation TRPV1 inhibitors were developed, which target TRPV1 depending on the mode of channel activation. For instance, A-1165442 blocks capsaicin and heat-evoked TRPV1 responses with minimal effects on H+-activated TRPV1 and does not significantly change core body temperature in rats.ref. bib557 Other drugs, such as A-1165901 and AMG8562,ref. bib558,ref. bib559 block TRPV1 activation by capsaicin while potentiating H+ activation. Interestingly, they cause hypothermia in mice. NEO6860 is specific only for vanilloid activation of TRPV1 and leaves H+ activation unaffected.ref. bib560 It also does not alter body temperature.ref. bib605
The prototypical TRPV1 activator is capsaicin.ref. bib454 Other naturally occurring TRPV1 activators include the superagonist resiniferatoxin,ref. bib562 arachidonic acid metabolites such as anandamide,ref. bib563 and several venom peptides, as reviewed by Hwang et al.ref. bib606 Capsaicin is therapeutically relevant in the treatment of neuropathic pain conditions due to its ability to desensitize and ultimately cause ablation and defunctionalization of TRPV1-expressing pain-conducting fibers after prolonged application, such as through topical capsaicin patches, as recently reviewed by Alalami et al.ref. bib607
TRPV2
Compared with TRPV1, the development of TRPV2-modulating compounds has been much less the focus of research. While specific and potent activators of TRPV2 are still lacking, some progress has been made regarding TRPV2 inhibitors. Iwata et alref. bib565 identified several TRPV2 inhibitors, including the antiallergic drug tranilast (IC50 = 10 μM) and the cyanine dye lumin (IC50 = 5 μM). Lumin acts as a general immunostimulant and exerts cardioprotective effects in a hamster model of dilated cardiomyopathy (δ-sarcoglycan-deficient hamster).ref. bib565 The administration of tranilast prevents cardiac dysfunction in a mouse cardiomyopathy model (dystrophin-utrophin double KO)ref. bib608 and suppresses fibrosis progression in a mouse model of nonalcoholic steatohepatitis.ref. bib609 However, apart from its action on TRPV2, tranilast also exerts pleiotropic effects on other targets in immune cells, fibroblasts, the cardiovascular system, and tumor cells, as reviewed by Darakhshan and Pour,ref. bib610 and it needs to be further confirmed to what extent the beneficial effects of tranilast and lumin in cardiac disease models depend on TRPV2 inhibition. Valdecoxib, a cyclooxygenase-2 inhibitor withdrawn from the market due to its unfavorable cardiovascular side effects, blocks rat TRPV2 channels with moderate potency (IC50 = 10 μM) but not TRPV1, TRPV3, and TRPV4 channels.ref. bib566 Monanchomycalin B, an alkaloid isolated from the marine sponge Monanchora pulchra, only poorly discriminates between TRPV1, TRPV2, and TRPV3 channels (IC50 = 6.0, 2.8, and 3.2 μM, respectively).ref. bib567 Other natural compounds that inhibit TRPV2 include coumarin derivative enantiomers from the roots of the orange jasmine Murraya exotica, B304-1 and B304-2, which partially inhibitref. bib568 TRPV2 channels (IC50 = 22.2 and 3.7 μM, respectively) but not TRPV1, TRPV3, or TRPV4 channels, as well as piperlongumine, an alkaloid from the long pepper Piper longum. Piperlongumine selectively inhibits human TRPV2 (IC50 = 4.6 μM) and reduces tumor sizes when applied to a murine glioblastoma model.ref. bib569 However, due to low solubility, the compound has to be encapsulated in β-cyclodextrin and applied to an implantable dextran-dendrimer hydrogel scaffold.
Synthetic TRPV2 inhibitors include SET2 (IC50 = 0.5 μM)ref. bib570 and IV2-1 (IC50 = 6.3 μM),ref. bib472 which do not affect TRPV1, TRPV3, and TRPV4 channels. However, neither compound has been tested in TRPV2-relevant disease models yet.
Regarding TRPV2 activation, particularly human TRPV2, has proven difficult to activate without inducing cytotoxic effects at the concentrations of the drugs required for robust activation. Currently, probenecid and cannabinoids, or a combination of both, are primarily used for in vitro studies.ref. bib413,ref. bib571,ref. bib572,ref. bib611
TRPV3
Various natural compounds isolated from plants inhibit TRPV3 channels, though most of them are only moderately potent. Nonetheless, some of them have demonstrated efficacy in vivo, particularly in mouse models of acute and chronic itch. Citrusinine II, derived from the small evergreen tree Atalantia monophylla, inhibits TRPV3, albeit with a relatively low potency (IC50 = 12.4 μM). It suppresses itch in mouse models of both acute and chronic pruritus when administered subcutaneously.ref. bib573 Naturally occurring isochlorogenic acid A (IC50 = 2.7 μM) and B (IC50 = 0.9 μM), active ingredients of the herb Achillea alpina, inhibit TRPV3 and reduce ear swelling and chronic pruritus in mouse models of topical carvacrol treatment.ref. bib574,ref. bib612 The coumarin osthole, isolated from Cnidium monnieri (IC50 = 37 μM for hTRPV3)—a plant used in traditional Chinese medicine—attenuates dry skin itch and histamine-dependent itch.ref. bib575 Subsequent studies by the same group have demonstrated the efficacy of the TRPV3 inhibitors forsythoside B, which is found in a number of plants of the mint order (IC50 = 6.7 μM),ref. bib576 and plant-derived verbascoside (IC50 = 14 μM) in similar disease models.ref. bib577 More recently, α-mangostin from the mangosteen plant was identified as a highly potent inhibitor of WT TRPV3 (IC50 = 77 nM) and TRPV3 GOF mutants (G573S and G573C) (IC50 ∼2 μM).ref. bib578
In addition to naturally occurring substances, several chemically synthesized TRPV3 inhibitors were developed. Optimization of primary hits regarding absorption, distribution, metabolism, and excretion properties has led to the discovery of compound 74a (IC50 = 0.38 μM) with favorable drug-like properties and efficacy in mouse models of neuropathic and central pain.ref. bib579 Another compound, Trpvicin (IC50 = 0.38 μM), stabilizes both WT TRPV3 and a GOF mutant (G573S) in their closed conformations, effectively inhibiting hair loss in a mouse model carrying the G568V mutation, relieving symptoms of chronic and acute itch.ref. bib580 Some local anesthetics, which are sometimes used to treat pruritus and pain, have also shown efficacy in inhibiting TRPV3 channels, although with low potency. Bupivacaine, mepivacaine, lidocaine, and ropivacaine inhibit TRPV3 with IC50 values ranging from 170 μM to 2.5 mM.ref. bib581 Dyclonine, a clinically used anesthetic, acts at least 2 orders of magnitude more potently on TRPV3 channels (IC50 = 3.2 μM) than on TRPV1, TRPV2, TRPM8, and TRPA1 and relieves carvacrol-induced scratching in mice.ref. bib582 Flopropione, an antispasmic agent, also blocks TRPV3 channels (IC50 = 18 μM) and alleviates symptoms in mouse models of skin inflammation induced by skin sensitizers.ref. bib583
Naturally occurring monoterpenes, such as thymol, carvacrol, or camphor, activate TRPV3 but with poor potencies.ref. bib585 A screening of Boswellia extracts for bioactive components identified the diterpene incensole acetate (EC50 = 16 μM) as a novel TRPV3 activator. It exerts antidepressant and anxiolytic effects in WT but not in TRPV3-deficient mice, suggesting that these effects are indeed mediated via TRPV3 activation.ref. bib586 Tetrahydrocannabivarin, a nonpsychoactive analogue of tetrahydrocannabinol, stimulates TRPV3 channels (EC50 = 6.1 μM) by binding to the vanilloid site but also activates several other TRP channels.ref. bib377,ref. bib587 More recently, the synthetic compound KS0365 was identified, showing 3-fold greater potency than 2-aminoethoxydiphenyl borate (EC50 = 5.1 μM, calculated in cholesterol-enriched cells) in activating TRPV3 without affecting TRPV2 channels.ref. bib485
TRPV4
Significant progress has been made to improve the pharmacology of TRPV4 channels, as several pharmaceutical companies have set out to develop novel TRPV4 modulators. Subsequently, their efficacies have been demonstrated in different mouse models of diseases.
In terms of antagonists, highly potent and selective drugs are now available. Thorneloe et alref. bib588 reported that the orally available TRPV4 antagonist GSK2193874 (IC50 = 2 nM for rTRPV4 and 40 nM for hTRPV4) was beneficial in mouse models of pulmonary edema. GSK2193874 is highly specific for TRPV4, demonstrating selectivity across more than 200 tested targets. The compound GSK2798745 (IC50 = 1.8 nM) resulted from a lead optimization process and demonstrated efficacy in a rat model of pulmonary edema.ref. bib589 In rats, cyclophosphamide-induced cystitis was inhibited by Hydra’s HC-067047 (IC50 values were 48 nM for hTRPV4, 133 nM for rTRPV4, and 17 nM for mTRPV4).ref. bib590 Renovis Pharma also identified several TRPV4-targeting modulators, including both activators and inhibitors. RN-1734 inhibited TRPV4 (IC50 = 2.3 μM) with moderate potency.ref. bib591 Later, the same groupref. bib592 introduced orally bioavailable RN-9893 with an improved potency (IC50 values of 0.42 μM, 0.66 μM, and 0.32 μM for human, rat, and mouse TRPV4 receptors, respectively) and high specificity for TRPV4.
Several selective activators of TRPV4 are available. GlaxoSmithKline’s GSK1016790A is highly potent (EC50 = 2 nM for hTRPV4) and selective for TRPV4. Systemic administration of GSK1016790A in animals causes a severe drop in blood pressure up to circulatory collapse and death, highlighting the role of TRPV4 in the regulation of vascular tone and vasodilation.ref. bib495,ref. bib593 Recently, a novel TRPV4 agonist was discovered (EC50 = 60 nM), which is suitable for in vivo application. The quinazolin-4(3H)-one derivative 36-HCl suppressed the progression of osteoarthritis in a rat model of surgically induced osteoarthritis (meniscal tear model) through intra-articular application.ref. bib594 Renovis Pharma introduced the piperazine RN-1747 with EC50 values of 0.77 and 4 μM for hTRPV4 and mTRPV4/rTRPV4, respectively.ref. bib591
Naturally occurring TRPV4 activators include curcumin and puerarin, although both compounds only show low potencies and are, in the case of curcumin, only poorly selective for TRPV4.ref. bib595,ref. bib596
TRPV5 and TRPV6 channels
Several compounds block TRPV5 and TRPV6 channels. Initially, their potency was low, and most of them did not discriminate well between the 2 isoforms. Miconazole and econazole demonstrate approximately 2-fold higher activity for TRPV6 than for TRPV5 but require high concentrations (>100 μM) for effective channel blockade.ref. bib597 Cryo-EM studies of TRPV6 in complex with econazole revealed binding to the periphery of the channel, where econazole replaced a lipid.ref. bib613,ref. bib614
Structure-based virtual screening has further advanced the identification of TRPV5-selective compounds. By virtually screening the econazole binding pocket using a database of 12 million compounds, 3 novel TRPV5 inhibitors were identified, including ZINC17988990, which selectively inhibits rabbit human TRPV5 but not human TRPV6 (IC50 = 0.11 and 0.18 μM, respectively).ref. bib598
Based on the lead compound TH-1177,ref. bib615 Landowski et alref. bib597 introduced the weakly potent compound 3 with a 5-fold selectivity for TRPV6 (IC50 = 90 μM) over TRPV5 (IC50 = 503 μM). Subsequent efforts have led to the development of cis-22a (IC50 = 0.32 μM) through ligand-based virtual screening, which exerts a 7-fold selectivity for TRPV5 compared with TRPV6.ref. bib599 However, cis-22a is not suitable for in vivo studies due to its low stability against microsomal degradation. Chemical modification of cis-22a resulted in the discovery of 3OG with a higher potency for TRPV6 inhibition (IC50 = 83 nM) and improved microsomal stability.ref. bib600 Cryo-EM, X-ray crystallography, and mutagenesis studies identified 2 types of binding sites for cis-22a in the TM region: one overlaps with lipid binding site 2 and the other is located at the intracellular pore entry site, which also serves as a binding region for Ca2+-CaM.ref. bib616,ref. bib617
Several naturally occurring compounds inhibit TRPV6 channels. SOR-C13 and SOR-C27, 2 short peptides derived from sorcidin, a paralytic venom of the shrew Blarina brevicauda, block TRPV6 with IC50 values of 14 and 64 nM, respectively. In mice, these peptides were used to detect TRPV6-overexpressing tumorsref. bib601 and reduced tumor growth in a xenograft model.ref. bib553 Tetrahydrocannabivarin blocks both TRPV5 and TRPV6 channels (IC50 = 4.8 μM and 9.4 μM, respectively) by binding to a site at the interface between the channel’s pore and the surrounding membrane.ref. bib602,ref. bib618
Photoswitchable inhibitors of TRPVs
Recently, Cunha et alref. bib603 developed a photoswitchable TRPV6 inhibitor based on the chemical structure of a previously reported TRPV6 inhibitor by introducing a phenyldiazo group to the molecule. Compound 9e rapidly switches by illumination with UVA light from the almost ineffective E-isomer to the inhibitory Z-isomer (IC50 = 1.7 μM).ref. bib599,ref. bib603
Ongoing or completed clinical trials with TRPVs as therapeutic targets
With respect to clinical trials, TRPV1 is by far the most intensely studied member of the TRPV family. According to the ClinicalTrials.gov database, nearly 100 studies have targeted TRPV1 for various conditions, with a focus on asthma and cough, inflammatory skin diseases, and, in particular, various pain conditions. However, due to hyperthermia and an increased likelihood of burn injuries associated with TRPV1 inhibition, many first-generation TRPV1 antagonists were withdrawn from clinical trials or did not progress further.ref. bib604 Mode-specific second-generation TRPV1 inhibitors, such as NEO6860 (NCT02337543), do not affect heat and pH activation of TRPV1 and provide a better safety profile.ref. bib560 However, NEO6860 did not demonstrate superior efficacy compared with placebo in a phase 2 trial to treat knee osteoarthritis (NCT02712957).ref. bib605 Topical TRPV1 antagonists are well tolerated and are under investigation for the treatment of inflammatory skin diseases, such as atopic dermatitis (PAC-14028, asivatrep; NCT02583022, NCT02757729, and NCT02965118), where they show promising effects.ref. bib619 Another approach involves the desensitization of TRPV1 channels, which is used in therapeutic approaches such as the use of capsaicin-containing creams for the treatment of moderate pain or the intravesical instillation of capsaicin or resiniferatoxin for an overactive bladder. This strategy is also being explored in trials investigating capsaicin formulations or the TRPV1 agonist CA-008 (vocacapsaicin) for the management of chronic pain conditions, as reviewed by Iftinca et al.ref. bib620
Compared with TRPV1, far fewer studies have evaluated the efficacy of compounds targeting other members of the TRPV family. Two drug repurposing studies have evaluated the use of the nonspecific TRPV2 activator probenecid, an FDA-approved drug to treat gout and hyperuricemia. In a small phase 4 study (NCT03965351) involving patients with functionally univentricular (Fontan) circulation, probenecid improved cardiac function compared with placebo.ref. bib621 Another phase 2 study, involving 20 patients, investigated probenecid as a positive inotropic agent for the treatment of heart failure (NCT01814319) and demonstrated a better cardiac function.ref. bib622 However, given the nonspecific action of probenecid, further confirmation is required to determine whether TRPV2 activation underpins its potential cardiac benefits. The TRPV2 inhibitor tranilast is currently being studied in a phase 1/2 study of patients suffering from advanced esophageal cancer for its efficacy when combined with traditional chemotherapy (jRCTs051190076).ref. bib623
Although preclinical studies suggest a role for TRPV3 in skin diseases, to date, only the TRPV3 inhibitor, GRC15300 (SAR292833), by Glenmark Pharmaceuticals has progressed to clinical trials for targeting osteoarthritis and neuropathic pain. However, the drug failed to meet its primary endpoint in a phase 2 trial in 2013 (NCT01463397).
Alongside TRPV1, TRPV4-selective modulators have achieved notable clinical progress within the TRPV family. GSK2798745, a highly potent TRPV4 inhibitor developed by GlaxoSmithKline,ref. bib589 has entered several early-phase clinical trials. No safety issues or serious side effects were observed in a phase 1 study (NCT02119260).ref. bib624 However, in 2017, a phase 2a study of heart failure patients (NCT02497937) failed to demonstrate significant effects of TRPV4 inhibition on pulmonary gas diffusion as an indicator of lung congestion.ref. bib625 In 2019, a combined phase 1/2 study (NCT03372603) assessed the effect of the same molecule on chronic cough, but the study was terminated due to a lack of efficacy. GSK2798745 was also unable to reduce alveolar barrier disruption in a model of lipopolysaccharide-induced acute lung injury in another phase 1 trial (NCT03511105),ref. bib1328 and the study was terminated due to a low probability of achieving a positive outcome of the primary endpoint. A recently completed phase 1 study (NCT04292912), evaluating GSK2798745 in patients with diabetic macular edema, has yet to publish results. Additionally, in 2023, an observational study started, monitoring the natural history of neuropathic pain in patients with confirmed genetic mutations in the TRPV4 gene (NCT05600764).
TRPV6, due to its overexpression in many solid tumors, is considered to comprise a novel target for anticancer therapy.ref. bib601 In 2015, the safety and tolerability of SOR-C13 from Sorcimed Biopharma were demonstrated in a phase 1 study (NCT01578564) involving patients with advanced solid tumors, with some experiencing antitumor effects of the drug.ref. bib626 These findings were followed up in another recently completed phase 1 trial (NCT03784677), but no results have been published yet. The FDA has granted SOR-C13 an orphan drug designation for advanced ovarian and pancreatic cancer. CBP-1008, by Coherent Biopharma, is a bispecific ligand-drug conjugate targeting folate receptor α and TRPV6 linked to the cytostatic monomethyl auristatin E. Treatment with CBP-1008 is currently evaluated in an ongoing phase 1 trial (NCT04740398) for advanced solid tumors.
TRPMs
TRPM gene family
The founding member of the TRPM gene subfamily was identified as transcripts enriched in melanomas and, therefore, named melastatin (now TRPM1; Table 1).ref. bib5,ref. bib7 The human TRPM gene family consists of 8 members. Based on amino acid similarity, TRPM proteins form 2 phylogenetic groups, TRPM1/3/6/7 and TRPM2/8/4/5, which can be further subdivided into 4 pairs of homologous channels: TRPM1/3, TRPM2/8, TRPM4/5, and TRPM6/7 (Fig. 4A).ref. bib5,ref. bib7,ref. bib627,ref. bib628 The structural organization and key biophysical characteristics were found to be conserved within pairs; however, with some exceptions.
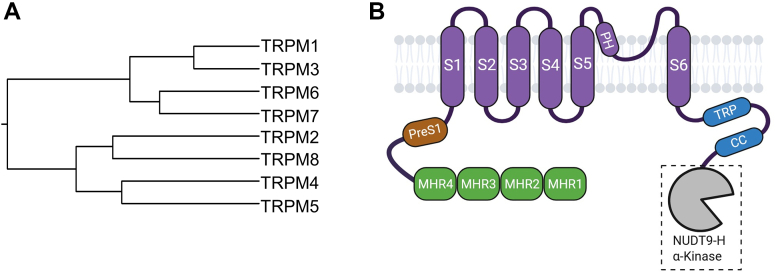
Domain topology, assembly, and functional characteristics of individual TRPMs
Domain topology and channel assembly
The domain organization of TRPMs is illustrated in Fig. 4B. The large N-terminus of TRPMs, comprising ∼70% of the total protein sequence, is unique among ion channels and subdivided into 4 melastatin homology regions (MHR1–4). The membrane-spanning segment of TRPMs, similar to other TRP channels, contains a small amphiphilic domain (pre-S1 helix) and 6 TM helices (S1–S6). The region between S5 and S6 forms a short PH and a pore-forming loop. The S6 helix is linked to a highly conserved TRP helix and a CC domain. TRPM proteins function as tetrameric channel complexes, with 4 S5–S6 segments forming a common membrane-spanning channel pore (Fig. 4B).
TRPMs function as homotetramers—4 subunits assemble to form a channel pore. In addition, the closely related TRPM1 and TRPM3 proteins, as well as TRPM6 and TRPM7, can form TRPM1/3 and TRPM6/7 heterotetrameric channels.ref. 629., ref. 630., ref. 631., ref. 632., ref. 633. Cryo-EM was successfully used to address high-resolution structures of TRPM2,ref. 634., ref. 635., ref. 636., ref. 637., ref. 638., ref. 639., ref. 640. TRPM3,ref. 641., ref. 642., ref. 643. TRPM4,ref. 644., ref. 645., ref. 646., ref. 647., ref. 648. TRPM5,ref. bib649,ref. bib650 TRPM7,ref. 651., ref. 652., ref. 653. and TRPM8.ref. bib40,ref. bib41,ref. 654., ref. 655., ref. 656. These findings provided new mechanistic insights into structure-function relationships of TRPMs.ref. bib657,ref. bib658
Three TRPMs contain additional C-terminal segments. TRPM2 contains the nudix hydrolase 9 homology (NUDT9-H) domain (Fig. 4B).ref. bib659,ref. bib660 NUDT9 proteins cleave ADP-ribose (ADPR) into AMP and ribose-5-phosphate.ref. bib661 NUDT9-H of human TRPM2 binds ADPR but does not exhibit enzymatic activity, while invertebrate TRPM2 proteins retain the capability to cleave ADPR.ref. bib640,ref. bib662
The C-terminal domains of TRPM6 and TRPM7 encode α-type kinase domains (Fig. 4B).ref. 663., ref. 664., ref. 665. α-Kinases are a group of atypical serine/threonine protein kinases with low primary sequence similarity to conventional protein kinases.ref. bib666,ref. bib667 C-terminal regions in other TRPM proteins do not contain enzymatic domains (Fig. 4B).
Alternative mRNA splicing creates additional diversity among TRPMs. TRPM1 is expressed as a “long” active variant and as a “short” transcript that lacks the sequence encoding the TM channel segment.ref. bib668 Alternative mRNA processing of TRPM2 results in truncated channel versions with different functional characteristics.ref. 669., ref. 670., ref. 671. Alternative splicing of the pore-coding sequence in TRPM3 produces channels with distinct cation selectivity.ref. bib672 TRPM4 is expressed as TRPM4a and TRPM4b variants with low and high channel activity, respectively.ref. bib673,ref. bib674 Alternative splicing of TRPM6 and TRPM7 creates isoforms encoding the N-terminal segments directly fused to the α-kinase domains.ref. bib629,ref. bib675
Functional characteristics
TRPMs have been extensively investigated, and despite the overall structural similarity, they differ significantly in functional characteristics and cellular roles.ref. bib657,ref. bib658 The phylogenetic group of TRPM1, 3, 6, and 7 (Fig. 4A) represents channels that are highly permeable to divalent cations, including Zn2+, Mg2+, and Ca2+, and are regulated by PIP2 and intracellular Mg2+.
TRPM1, the founding member of the TRPM subfamily, forms a constitutively active channel highly expressed in melanocytes and the retina.ref. bib633,ref. bib676 The constitutive activity of TRPM1 can be further stimulated by the neurosteroid pregnenolone sulfate (PS) and inhibited by extracellular Zn2+ or intracellular Mg2+ ions.ref. bib633 As mentioned above, TRPM3 produces several alternatively spliced variants,ref. bib677,ref. bib678 including TRPM3α1 and TRPM3α2. TRPM3α1 contains a longer pore-forming segment between S5 and S6 and is highly permeable to Na+ ions.ref. bib672 TRPM3α2 contains a shorter pore-forming sequence and is characterized by a high permeability to divalent cations.ref. bib672,ref. bib679 In addition, the S1–S4 regions of TRPM3α2 contains a noncanonical ion permeation mechanism called “omega” Na+ currents.ref. bib680ref. bib906 TRPM3α2 is the most studied channel variant referred to herein as TRPM3. TRPM3 is negatively regulated by intracellular Mg2+ and PIP2 depletion and stimulated by PS and other steroids.ref. bib672,ref. 682., ref. 683.–ref. bib681 TRPM3 is modulated by osmolality and D–erythro-sphingosine.ref. bib682,ref. bib683 Cryo-EM structures have demonstrated that PS activates TRPM3 through a site at the outer region of the channel pore formed by the PH and S1.ref. bib642 The TRPM3 channel is activated by heat, underlying the temperature responses of the dorsal root and trigeminal ganglia neurons.ref. bib684,ref. bib685 Stimulation of receptors coupled to Gi and Go causes inhibition of the channel through the direct assembly of the Gβγ subunits with TRPM3.ref. bib641,ref. bib686,ref. bib687
TRPM6 and TRPM7 are homologous bifunctional proteins containing TM channel segments fused to cytosolic kinase domains and, therefore, are frequently named channel kinases.ref. bib5,ref. bib7,ref. bib627 Thus, one TRPM6 or TRPM7 tetramer will form a typical TRP channel unit linked to 4 cytosolic kinase domains.ref. bib5,ref. bib7,ref. bib627 Furthermore, recent proteomic studies demonstrated that ARL15, PPT4A1–3, and CNNM1–4 proteins coassemble to such channel-kinase complexes.ref. bib688,ref. bib689 Channel complexes formed by TRPM6 and TRPM7 are highly permeable to divalent cations and are negatively regulated by cytoplasmic Mg2+ and Mg·ATP as well as membrane levels of PIP2.ref. bib664,ref. bib665,ref. bib675,ref. 693., ref. 694., ref. 695., ref. 696., ref. 697., ref. 698., ref. 699., ref. 700., ref. 701. The cryo-EM structures of the truncated TRPM7 have been resolved in the closed and open states.ref. 651., ref. 652., ref. 653. The solved structures are consistent with the idea that the lower channel gate contains the regulatory Mg2+ binding site.ref. bib699
The α-kinase domains of TRPM6 and TRPM7 display low amino acid sequence homology to conventional serine/threonine kinases.ref. bib667 However, the crystal structure of the TRPM7 kinase reveals considerable structural similarity to other protein kinases.ref. bib666 The mass spectrometry approach identified multiple autophosphorylation sites mainly located in a serine/threonine-rich region situated upstream of the kinase domain of TRPM7.ref. bib688,ref. bib700,ref. bib701 Similar to TRPM7 kinase, TRPM6 kinase can phosphorylate its own serine/threonine residues.ref. bib700 In immune cells, the TRPM7 kinase domain can be cleaved from the channel domain by caspases upon Fas-receptor stimulation.ref. bib702,ref. bib703 Other studies have reported that cleaved TRPM6 and TRPM7 kinases are detected in the cell nucleus, where they can phosphorylate histones.ref. bib703,ref. bib704 Currently, the known phosphorylation substrates of TRPM6 and TRPM7 kinases comprise ∼20 proteins with varied subcellular locations and functional roles.ref. 708., ref. 709., ref. 710., ref. 711., ref. 712., ref. 713., ref. 714., ref. 715., ref. 716. Overall, such functional diversity makes it challenging to develop a unified model of the cellular roles of the TRPM6 and TRPM7 kinase domains.
The TRPM2, 4, 5, and 8 phylogenetic group (Fig. 4A) comprises channels with diverse functional characteristics. The TRPM2 and TRPM8 channels are permeable to divalent and monovalent cations and are often called nonselective cation channels or Ca2+-permeable cation channels. By contrast, TRPM4 and TRPM5 are impermeable to Ca2+ and are thus frequently called monovalent cation-selective channels.
TRPM2 was recognized as an unusual TRP channel due to the presence of the C-terminal NUDT9-H domain. TRPM2 is directly activated by intracellular ADPR.ref. bib659,ref. 717., ref. 718., ref. 719., ref. 720., ref. 721., ref. 722., ref. 723. Recently, cryo-EM analysis of TRPM2 has identified 2 ADPR-binding pockets located in the MHR1/2 and NUDT9-H domains.ref. 634., ref. 635., ref. 636., ref. 637., ref. 638., ref. 639. Both sites play a role in the opening of the TRPM2 channel. However, the interaction of ADPR with MHR1/2 underpins the prime regulatory mechanism in the human TRPM2 channel.ref. 634., ref. 635., ref. 636., ref. 637., ref. 638., ref. 639. In addition, intracellular Ca2+ and membrane PIP2 were identified as crucial physiological ligands of TRPM2 required for channel opening by ADPR.ref. bib662,ref. 724., ref. 725., ref. 726., ref. 727., ref. 728. The Ca2+ binding site is formed by acidic side chains of residues located in the S2 and S3 helices and the TRP domain of TRPM2.ref. 634., ref. 635., ref. 636., ref. 637., ref. 638., ref. 639. PIP2 is found in a cavity often called a “vanilloid binding pocket.”ref. bib634 TRPM2 is positively regulated by warm temperatures (>35 °C).ref. 729., ref. 730., ref. 731., ref. 732. The structural basis for the temperature sensitivity of TRPM2 remains puzzling. The TRPM2 channel was also suggested as a cellular redox sensor because it is activated by peroxides, like H2O2, or other agents that produce reactive oxygen species (ROS).ref. bib730 However, ROS act indirectly on TRPM2, likely due to the elevation of intracellular ADPR.ref. bib718,ref. bib719,ref. bib723,ref. bib731,ref. bib732
TRPM8 shares significant structural homology with TRPM2. However, the C-terminal segment of TRPM8 lacks NUDT9-H. TRPM8 is activated by cold (<23–28 °C) and chemical agents evoking a sensation of coolness, including menthol and icilin.ref. bib14,ref. bib406,ref. bib733,ref. bib734 TRPM8 is a voltage-dependent channel.ref. bib407,ref. bib735 Analogously to TRPM2, the channel activity of TRPM8 is critically dependentref. bib42,ref. bib736 on PIP2 and intracellular Ca2+. The depletion of PIP2 prevents channel opening by pharmacological compounds and cold,ref. bib42,ref. 739., ref. 740., ref. 741. whereas Ca2+ is required for TRPM8 activation by icilin.ref. bib40,ref. bib656 Cryo-EM studies of TRPM8 have identified the binding sites of Ca2+, PIP2, and cooling agonists.ref. bib40,ref. bib41,ref. 654., ref. 655., ref. 656.,ref. bib739 The location of Ca2+ and PIP2 binding sites in TRPM8 parallels the corresponding sites in TRPM2.ref. bib40,ref. bib41,ref. 654., ref. 655., ref. 656.,ref. bib739 Cooling agents acoltremon (WS-12) and icilin interact with a binding pocket formed by the S1–S4 helices and the TRP domain of TRPM8, frequently called the VSLD.ref. bib40,ref. bib41,ref. 654., ref. 655., ref. 656.,ref. bib739,ref. bib740 However, the structural basis of the effects evoked by voltage and temperature on TRPM8 has not yet been established.ref. bib739,ref. bib741,ref. bib742
TRPM4 and TRPM5 display functional characteristics that are unique among TRP channels—both channels are selective for monovalent cations and are activated upon increases in cytosolic Ca2+ levels.ref. bib673,ref. bib674,ref. 746., ref. 747., ref. 748. Two Ca2+-binding pockets are found in TRPM4 and TRPM5.ref. 644., ref. 645., ref. 646., ref. 647., ref. 648., ref. 649., ref. 650. One site is evolutionarily conserved, and, like in TRPM2 and TRPM8, it is formed by negatively charged residues in the TM domains of TRPM4 and TRPM5.ref. 648., ref. 649., ref. 650. This site is primarily responsible for the Ca2+-dependent opening of the TRPM4 and TRPM5 channels.ref. 648., ref. 649., ref. 650. Another Ca2+-binding site is unique to TRPM proteins and is located at the interface of the MHR1/2 and MHR3/4 cytosolic domains.ref. 648., ref. 649., ref. 650. In TRPM4, the interaction of Ca2+ with the cytosolic domain regulates a complex conformational temperature transition of the channel.ref. bib648 In TRPM5, the binding of Ca2+ to this site affects the structural dynamics of the N-terminal domain, which subsequently regulates the voltage- and Ca2+-dependent opening of the channel.ref. bib649,ref. bib650 In addition, TRPM4 and TRPM5 are regulated by PIP2.ref. bib673,ref. bib674,ref. 746., ref. 747., ref. 748., ref. 749. ATP is a negative modulator of TRPM4.ref. bib746,ref. bib747 ATP binds to TRPM4 through a site located at the interface of the MHR1/2 domains, and this interaction is temperature-dependent.ref. bib644 Importantly, ATP does not act on TRPM5.ref. bib649,ref. bib746,ref. bib747 TRPM5 was found to be a heat-sensitive channel that contributes to the temperature-dependent reception of chemical stimuli by taste receptor cells.ref. bib748,ref. bib749
Expression pattern and primary physiological roles
TRPM1 and TRPM3
Initially, TRPM1 was identified as a transcript enriched in human melanomas and was suggested to be a potential tumor suppressor.ref. bib750,ref. bib751 However, the particular function of TRPM1 in melanomas and skin melanocytes remains unclear. The pathophysiological role of TRPM1 has been extensively investigated in the context of ON-bipolar neurons, which form synapses with rod, cone, and horizontal cells in the retina,ref. bib676,ref. 755., ref. 756., ref. 757. because LOF mutations in the human TRPM1 gene cause congenital stationary night blindness.ref. 757., ref. 758., ref. 759., ref. 760., ref. 761. In the dark, rod cells secrete glutamate, which activates metabotropic glutamate receptor 6 (mGluR6) in postsynaptic ON-bipolar neurons. mGluR6 is a Go protein-coupled receptor, and its activation leads to the release of Gα(o) and Gβγ. Both subunits directly interact and deactivate the TRPM1 channel.ref. bib676,ref. 762., ref. 763., ref. 764. Exposure to light blocks the release of glutamate and inactivates mGluR6, leading to the opening of the TRPM1 channel and depolarization of ON-bipolar neurons.ref. bib676,ref. 762., ref. 763., ref. 764. Importantly, stationary night blindness was developed by Trpm1 KO mice, confirming the monogenic basis of the disease.ref. bib753
TRPM3 is highly expressed in nociceptive neurons, pancreatic β cells, the brain, and several other tissues.ref. bib677 In pancreatic islets, TRPM3 functions as an ionotropic steroid receptor responsible for PS-induced Ca2+ influx in β cells, leading to enhanced insulin secretion.ref. bib762 In addition, TRPM3 can underlie PS-stimulated Zn2+ uptake in β cells, the crucial factor for insulin-containing dense core vesicles.ref. bib679 PIP2 is directly associated with TRPM3.ref. bib641,ref. bib763,ref. bib764 In insulinoma cells, stimulation of Gq protein-coupled receptors leads to the stimulation of PLC, depletion of its substrate PIP2, and inhibition of TRPM3.ref. bib763 As mentioned above, activation of GPCRs leads to the inhibition of TRPM3 through the direct association of Gβγ with the channel.ref. bib641,ref. bib686,ref. bib687 This regulatory mechanism contributes to the antinociceptive effects of μ opioid receptor agonists in DRG neurons.ref. bib686 The TRPM3 channel is required for the temperature sensitivity of DRG and trigeminal ganglia neurons.ref. bib684,ref. bib685,ref. 766., ref. 767., ref. 768. Trpm3 KO mice exhibited diminished sensitivity to noxious heat and reduced inflammatory heat hyperalgesia.ref. bib684,ref. bib685 Consequently, TRPM3 was suggested as a new analgesic drug target.ref. bib768
TRPM6 and TRPM7
TRPM7 is a ubiquitously expressed channel. Endogenous Mg2+-regulated TRPM7 currents have been found in virtually all primary isolated cells and stable cell lines examined, supporting the notion that TRPM7 is a versatile channel that plays a housekeeping cellular role.ref. bib675,ref. bib691,ref. bib699,ref. 770., ref. 771., ref. 772., ref. 773., ref. 774., ref. 775. To this end, independent evidenceref. bib690,ref. bib691,ref. 776., ref. 777., ref. 778. supports the concept that the TRPM7 channel represents the principal route for the cellular uptake of divalent cations, especially Mg2+. In line with this assumption, the genetic disruption or pharmacological inhibition of TRPM7 causes cell cycle arrest.ref. bib690,ref. bib691,ref. bib775 Besides the homeostatic control of cellular Mg2+ and Zn2+ contents, TRPM7 is recognized as a Ca2+ channel shaping Ca2+-dependent cellular pathwaysref. 779., ref. 780., ref. 781., ref. 782. and a vesicular Zn2+ release channel.ref. bib692 In this context, tissue-specific ablation of Trpm7 in mice was used to elucidate the role of TRPM7 in the systemic balance of divalent cations. Unexpectedly, kidney-restricted deletion of Trpm7 in mice did not cause apparent changes in the development, physical appearance, and biochemical characteristics of biological fluids.ref. bib690 In another mouse strain, Trpm7 was inactivated in enterocytes throughout the whole intestine, including the colon.ref. bib690 Newborn mutants displayed growth failure and 100% mortality before weaning. Moreover, Trpm7-deficient pups displayed low Zn2+, Mg2+, and Ca2+ levels in serum, urine, and bones. Nutritional Zn2+ and Mg2+ supplementation of breastfeeding females extended the lifespan of mutant pups. These findingsref. bib690 support the concept that intestinal TRPM7 operates as a master regulator of the body’s balance of Zn2+, Mg2+, and Ca2+.
Transgenic mouse models have been extensively used to investigate the role of TRPM7 in prenatal development (reviewed by Chubanov et alref. bib665,ref. bib782). Among other exciting findings, it was demonstrated that TRPM7 is indispensable for early embryo development.ref. bib783,ref. bib784 Also, TRPM7 is abundantly expressed in gametes.ref. 784., ref. 785., ref. 786., ref. 787. KO of Trpm7 reduced Mg2+ and Zn2+ levels in oocytes and 4-cell embryos, leading to arrested embryonic development at the blastocyst stage.ref. bib785 Interestingly, Mg2+ but not Zn2+ supplementation rescues the arrest of Trpm7-deficient zygotes.ref. bib785 These findings correlate well with the in vitro examination of mouse embryonic stem cells and embryonic trophoblast stem cells, demonstrating that the loss of TRPM7 function leads to Mg2+-dependent proliferation arrest.ref. bib775,ref. bib787 In other studies, conditional mutagenesis of Trpm7 at different embryonic stages has demonstrated that TRPM7 is indispensable for organogenesis of the kidney, heart, CNS, and immune organs.ref. 789., ref. 790., ref. 791.
Unlike the ubiquitously present TRPM7 channel, the expression of TRPM6 is limited to transporting epithelial cells of the placenta, kidneys, and intestine.ref. bib629,ref. bib787,ref. bib791,ref. bib792 The necessity for epithelial cells to express both TRPM6 and TRPM7 remains a topic of debate.ref. bib663,ref. bib793 Some studies suggest that the TRPM6 channel represents the close functional homolog of TRPM7 and that both proteins operate independently.ref. bib632,ref. bib794,ref. bib795 An alternative view is that TRPM6 assembles with TRPM7 in heteromeric channels, which are less susceptible to metabolic negative control by cytosolic Mg·ATP.ref. 629., ref. 630., ref. 631.,ref. bib787,ref. bib796
TRPM6 was found to be highly expressed in the DCT segment of the kidney and in enterocytes of the gastrointestinal tract.ref. bib791,ref. bib792 Consequently, a comparative examination of mice with global versus kidney- or intestine-specific deletions of Trpm6 was conducted.ref. bib787 Mice lacking Trpm6 in the whole body or specifically in the intestine exhibited severe hypomagnesemia and depletion of Mg2+ in bones due to impaired intestinal uptake of Mg2+ ions.ref. bib787,ref. bib797 Dietary Mg2+ supplementation fully normalized the biochemical and physiological characteristics of Trpm6-deficient mice.ref. bib787 In contrast, 2 independent mouse strains with a kidney-specific KO of Trpm6 displayed little or no impact on serum Mg2+ levels of mutant mice.ref. bib787,ref. bib798 These findings aligned with experimentsref. bib690 involving kidney- versus intestine-specific deletions of Trpm7. Hence, the traditional kidney-centric view on the organismal balance of divalent cations needs some adjustment.
TRPM2 and TRPM8
TRPM2 is a ubiquitously expressed channel implicated in many physiological processes, including insulin secretion by pancreatic β cells, Ca2+ signaling in immune cells, and body temperature sensation by somatosensory and hypothalamic neurons.ref. bib718,ref. bib719,ref. 729., ref. 730., ref. 731.,ref. 800., ref. 801., ref. 802., ref. 803., ref. 804., ref. 805., ref. 806., ref. 807., ref. 808., ref. 809., ref. 810., ref. 811., ref. 812., ref. 813., ref. 814., ref. 815., ref. 816., ref. 817., ref. 818., ref. 819. TRPM2 is implicated in pathophysiological conditions linked to excessive ROS production, for instance, inflammation, neurodegenerative disorders, and IR injury.ref. bib807,ref. bib808,ref. bib817,ref. 820., ref. 821., ref. 822., ref. 823., ref. 824., ref. 825., ref. 826., ref. 827., ref. 828., ref. 829., ref. 830., ref. 831. IR injury is characterized by increased tissue levels of ROS leading to Ca2+ overload, cell death, and inflammatory processes.ref. bib828 In this context, pharmacological inhibition of TRPM2 was suggested as a new strategy for treating IR injury.ref. bib827
The physiological role of TRPM8 was investigated in genetic mouse models.ref. bib831,ref. bib832 Trpm8 KO mice showed behavioral deficiency after exposure to cold temperatures.ref. 834., ref. 835., ref. 836., ref. 837. TRPM8 is defined as the principal mediator of acute and inflammatory pain and irritation-induced reflexes.ref. 834., ref. 835., ref. 836., ref. 837., ref. 838., ref. 839., ref. 840., ref. 841., ref. 842., ref. 843., ref. 844., ref. 845., ref. 846. TRPM8 is abundantly expressed in the nerve endings of DRG neurons innervating the urinary bladder and contributes to symptoms of urinary urgency and other bladder reflexes.ref. bib846,ref. bib847 Consequently, TRPM8 has been proposed as a new target for the treatment of pain, cancer, and other disorders.
TRPM4 and TRPM5
As TRPM4 and TRPM5 are impermeable to divalent cations, including Ca2+, their activation leads to Na+ influx and depolarization of the plasma membrane.ref. bib674 In electrically nonexcitable cells, the opening of TRPM4 and TRPM5 reduces the driving force for Ca2+ entry through Ca2+-permeable channels.ref. bib749,ref. 849., ref. 850., ref. 851., ref. 852. In excitable cells, like cardiomyocytes and neurons, depolarization of the cell membrane opens voltage-activated Ca2+ channels.ref. bib749,ref. 849., ref. 850., ref. 851., ref. 852. TRPM4 is a ubiquitously expressed channel, and its role in shaping cellular responses to external stimuli is well documented in diverse immune and endocrine cells, cardiomyocytes, and neurons.ref. bib749,ref. 849., ref. 850., ref. 851., ref. 852.
TRPM5 is highly expressed in type II taste receptor cells, mediating responses to sweet, amino acids, and bitter compounds.ref. bib852,ref. bib853 In taste cells, the activation of GPCRs leads to PLCβ2-evoked release of Ca2+ from intracellular stores and the opening of the TRPM5 channel.ref. 852., ref. 853., ref. 854., ref. 855. The activation of TRPM5 causes membrane depolarization, the opening of voltage-gated Ca2+ channels, and consequently, the Ca2+-dependent release of the transmitter ATP.ref. 852., ref. 853., ref. 854., ref. 855. Accordingly, deletion of Trpm5 in mice impaired taste reception.ref. bib748,ref. bib853 TRPM5 was found to be a heat-sensitive channel, and this characteristic contributes to the temperature-dependent reception of chemical stimuli by taste receptor cells in the tongue.ref. bib748,ref. bib749
In addition, TRPM5 was identified as a prime transduction channel in chemosensory tuft cells, also known as brush cells.ref. 856., ref. 857., ref. 858., ref. 859., ref. 860., ref. 861., ref. 862., ref. 863., ref. 864., ref. 865., ref. 866., ref. 867. Tuft cells are solitary epithelial cells containing apical “brush-like” microvilli that are present in many internal organs, including the respiratory system, thymus, gall bladder, urethra, and gastrointestinal tract.ref. bib867,ref. bib868 Tuft cells are crucial players in type 2 immune responses because they can detect pathogenic helminths, bacteria, and viruses.ref. bib857,ref. 867., ref. 868., ref. 869., ref. 870., ref. 871., ref. 872., ref. 873., ref. 874., ref. 875., ref. 876., ref. 877. Upon activation, tuft cells release leukotrienes, acetylcholine, interleukin-25, and ATP, mobilizing tissue-resident immune cells and other protective responses.ref. bib857,ref. 867., ref. 868., ref. 869., ref. 870., ref. 871., ref. 872., ref. 873., ref. 874., ref. 875., ref. 876., ref. 877., ref. 878.
Human diseases associated with TRPMs
Gene association studies revealed the causal role of TRPMs in several human disorders. Thus, LOF mutations in the human TRPM1 gene cause congenital stationary night blindness (type 1C), leading to impaired mGluR6/Go/TRPM1 signaling in ON-bipolar neurons in the retina.ref. 757., ref. 758., ref. 759., ref. 760., ref. 761.
De novo heterozygous point mutations in TRPM3 have been identified in patients with developmental and epileptic encephalopathy (DEE).ref. bib878,ref. bib879 DEE is a group of chronic encephalopathies characterized by epilepsy and intellectual disability.ref. bib878 Electrophysiological analysis of TRPM3 revealed that DEE-associated mutations represent GOF mutations.ref. 881., ref. 882., ref. 883. Pharmacological inhibition of TRPM3 by primidone has been demonstrated as a potential treatment for DEE patients.ref. 880., ref. 881., ref. 882., ref. 883., ref. 884.
LOF mutations in the human TRPM6 gene give rise to a disorder known as primary hypomagnesemia type 1, intestinal (HOMG1).ref. bib648,ref. 885., ref. 886., ref. 887. HOMG1 patients are typically infants presenting with generalized convulsions, muscle spasms, and very low blood levels of Mg2+ and Ca2+. Supplementation with high doses of Mg2+ in patients relieves hypomagnesemia and all other symptoms, including hypocalcemia.ref. bib791,ref. bib792 Therefore, this disorder is frequently called primary hypomagnesemia with secondary hypocalcemia.ref. bib791,ref. bib792,ref. bib887 Clinical assessment of the first HOMG1 patients revealed that hypomagnesemia developed due to defective intestinal Mg2+ uptake.ref. 885., ref. 886., ref. 887. In follow-up studies, renal leak of Mg2+ was also detected in Mg2+-supplemented HOMG1 individuals.ref. bib791,ref. bib792,ref. bib887
Missense mutations in TRPM7 have been linked to stillbirth.ref. bib888 Stillbirth is defined as the loss of a fetus after 22 weeks of gestation during pregnancy.ref. bib889 Worldwide, the stillbirth rate is ∼14 cases per 1000 births, and the etiology of this disease remains poorly understood.ref. bib889 Recently,ref. bib888 sequencing of tissue samples from affected fetuses revealed heterozygous nonsynonymous variants in TRPM7. Upon heterologous expression, introducing 2 mutations in TRPM7 caused a reduction in channel activity, whereas 2 other substitutions led to proteasomal degradation of TRPM7.ref. bib889 However, the exact physiological process impaired by these mutations in TRPM7 has not been established yet.
A new form of macrothrombocytopenia has been linkedref. bib890 to missense substitutions in TRPM7. Macrothrombocytopenia is a group of disorders characterized by abnormally large platelets due to their impaired formation in megakaryocytes.ref. bib890 The affected patients were heterozygous for LOF point mutations in TRPM7 and displayed reduced Mg2+ levels in platelets.ref. bib890 Notably, a mouse strain with conditional megakaryocyte-restricted Trpm7 KO also developed macrothrombocytopenia.ref. bib890
Trigeminal neuralgia is a human disease defined by severe facial pain.ref. bib891 Whole-exome sequencing identified 1 patient heterozygous for the A931T mutation affecting the S3 helix of TRPM7.ref. bib891 Electrophysiological analysis of the A931T TRPM7 channel variant revealed atypical “omega” Na+ currents.ref. bib891 Hence, it was proposed that these “omega” currents depolarize trigeminal ganglion neurons, causing pain in trigeminal neuralgia patients.ref. bib891
Recently, mutations in TRPM7 have been linked to an autosomal dominant variant of hypomagnesemia (low serum concentrations of Mg2+).ref. 893., ref. 894., ref. 895. The affected patients were heterozygous for LOF point mutations in TRPM7. Apart from hypomagnesemia, the patients displayed other less prominent symptoms, including episodes of hypocalcemia (low serum concentrations of Ca2+), seizures, and muscle cramps. In addition, some individuals suffered from migraine, autism, and developmental delays, mainly affecting speech and motor skills. Notably, supplementation with high doses of Mg2+ in patients could only partially normalize serum concentrations of Mg2+ and incompletely ameliorate other symptoms.ref. bib892,ref. bib893
GOF and LOF point mutations in the human TRPM4 gene have been linked to different forms of cardiac conduction defects, including progressive familial heart block type I,ref. bib895,ref. bib896 Brugada syndrome,ref. 898., ref. 899., ref. 900., ref. 901. right-bundle branch block, atrioventricular block, and complete heart block.ref. 902., ref. 903., ref. 904. However, it remains puzzling why either reduced or increased TRPM4 activity leads to different forms of cardiac conduction defects.
Pharmacological modulators of TRPMs
As outlined above, TRPMs critically contribute to diverse physiological processes and are considered prospective drug targets for the treatment of human diseases.ref. bib657,ref. bib658 Consequently, numerous studies have been conducted to identify small organic compounds suitable for the pharmacological regulation of TRPMs in cultured cells and animal disease models.ref. bib657,ref. bib658 Herein, we summarize the key developments in these research areas and discuss the identified pharmacological modulators of TRPMs. However, the present chapter will not cover the effects of nonspecific channel inhibitors (eg, ruthenium red and 2-aminoethoxydiphenyl borate) or compounds incompletely characterized in terms of their potency and efficacy.
TRPM1 and TRPM3
The pharmacological toolkit for TRPM1 has not yet been developed. TRPV1 agonists, capsaicin and anandamide, were used to activate endogenous TRPM1 currents in ON-bipolar cells.ref. bib754 The response of ON-bipolar cells to capsaicin was blocked by the TRPV1 inhibitor capsazepine.ref. bib754 Similarly, an antibiotic agent, voriconazole, was suggested to inhibit capsaicin-evoked TRPM1 currents in ON-bipolar cells.ref. bib904 However, evidence of the direct action of these compounds on the TRPM1 channel and the pharmacological characteristics of such interactions (eg, IC50) remains to be seen.
Several synthetic compounds positively regulate TRPM3 channel activity, including CIM0216, clotrimazole, and nifedipine (Table 4).ref. bib906,ref. 682., ref. 906., ref. 907., ref. 908., ref. 909. CIM0216 was determined to be the most potent activator of the TRPM3 channel.ref. bib905 The antifungal agent clotrimazole causes potentiation of the TRPM3 channel, as this compound does not affect basal or heat-activated TRPM3 currents but robustly stimulates TRPM3 upon coapplication with PS.ref. bib906 In addition, several potent inhibitors of TRPM3 have been identified. Thus, the FDA-approved drugs diclofenac, maprotiline, and primidone were found to be potent inhibitors of PS-induced TRPM3 activity (Table 4).ref. bib907 Notably, primidone could attenuate thermal nociception in animals.ref. bib907 Recently,ref. bib642 cryo-EM structures of TRPM3 were addressed in complex with PS, primidone, and CIM0216. While PS interacts with TRPM3 through a site at the outer region of the channel pore, primidone, nifedipine, and CIM0216 bind to TRPM3 within the cavity between the S1–S4 segments and the TRP domain.ref. bib642,ref. bib643 Another study demonstrated that 2 fruit flavanones, naringenin and hesperetin, and the spiny restharrow derivative, ononetin, are potent inhibitors of TRPM3 (Table 4).ref. bib908 Follow-up hit optimization experiments uncovered fruit flavanones isosakuranetin and liquiritigenin, displaying improved potency in the block of TRPM3 currents (Table 4).ref. bib909 Moreover, isosakuranetin and hesperetin were capable of reducing the sensitivity of mice to noxious heat and PS-induced pain.ref. bib909
Table 4: Pharmacological modulators of TRPM3
| Name (PubChem CIDa) | Effect | References |
|---|---|---|
| CIM0216 (42887770) | Activation, EC50 = 0.77 μM | ref. bib905 |
| Clotrimazole (2812) | Potentiation, EC50 = 20 nM | ref. bib906 |
| Nifedipine (4485) | Activation, EC50 = 30–32 μM | ref. bib762 |
| Diclofenac (3033) | Inhibition, IC50 = 6.2 μM | ref. bib907 |
| Maprotiline (4011) | Inhibition, IC50 = 1.3 μM | ref. bib907 |
| Primidone (4909) | Inhibition, IC50 = 0.6 μM | ref. bib907 |
| Naringenin (439246) | Inhibition, IC50 = 0.5 μM | ref. bib908 |
| Hesperetin (72281) | Inhibition, IC50 = 2.0 μM | ref. bib908 |
| Ononetin (259632) | Inhibition, IC50 = 0.3 μM | ref. bib908 |
| Isosakuranetin (160481) | Inhibition, IC50 = 50 nM | ref. bib909 |
| Liquiritigenin (114829) | Inhibition, IC50 = 0.5 μM | ref. bib909 |
a PubChem Compound Identification number.
TRPM6 and TRPM7
Several small molecules have been defined as negative regulators of the TRPM7 channel.ref. bib664,ref. 910., ref. 911., ref. 912. A significant fraction of these agents represent polyspecific channel blockers, incompletely characterized compounds, or low-potency antagonists of TRPM7.ref. bib664,ref. bib910,ref. bib911 However, NS8593, waixenicin A, FTY720, VER155008, CCT128930, cannabigerolic acid, and cannabidivarin were found to be potent inhibitors of TRPM7 currents with IC50 values in the low micromolar to nanomolar range (Table 5).ref. 913., ref. 914., ref. 915., ref. 916., ref. 917., ref. 918., ref. 919., ref. 920., ref. 921., ref. 922. Noteworthy, waixenicin A, VER155008, CCT128930, and cannabigerolic acid selectively suppressed the TRPM7 channel and displayed no effects on the homologous TRPM6 channel.ref. bib652,ref. bib653,ref. bib914,ref. bib915,ref. bib919 In contrast, NS8593 and FTY720 inhibited both channels, TRPM6 and TRPM7.ref. bib652,ref. bib653,ref. bib915,ref. bib917 NS8593, waixenicin A, and FTY720 were the most extensively used to map the cellular roles of the TRPM7 channel in different physiological and pathophysiological settings, including animal models of human diseases, such as tissue fibrosis, metabolic, cardiovascular, and immune disorders, and treatment of tumors, inflammation, and aortic aneurysm.ref. bib712,ref. bib781,ref. 923., ref. 924., ref. 925., ref. 926., ref. 927., ref. 928., ref. 929., ref. 930., ref. 931., ref. 932., ref. 933., ref. 934., ref. 935.
Table 5: Pharmacological modulators of TRPM6 and TRPM7
| Name (PubChem CIDa) | Effect | References |
|---|---|---|
| TRPM7 | ||
| NS8593 (71311765) | Channel inhibition, IC50 = 1.6 μMb (3.9 μMc) | ref. bib913 |
| Waixenicin A (73755210) | Channel inhibition, IC50 = 7.0 μMb (16 nMc) | ref. bib914 |
| FTY720 (107969) | Channel inhibition, IC50 = 0.72 μM | ref. bib917 |
| VER155008 (25195348) | Channel inhibition, IC50 = 0.11 μM | ref. bib915 |
| CCT128930 (17751819) | Channel inhibition, IC50 = 0.86 μMb (0.63 μMc) | ref. bib916 |
| Cannabigerolic acid (CBDA) (6449999) | Channel inhibition, IC50 = 1.8 μM | ref. bib919 |
| Cannabidivarin (CBDV) (11601669) | Channel inhibition, IC50 = 3.4 μM | ref. bib919 |
| Naltriben (5486827) | Channel activation, EC50 = 21 μM | ref. bib920 |
| Mibefradil (60663) | Channel activation, EC50 = 53 μM | ref. bib921 |
| TG100-115 (10427712) | Kinase inhibition, IC50 = 1.07 μM | ref. bib922 |
| TRPM6 | ||
| Iloperidone (71360) | Channel inhibition, IC50 = 0.73 μM | ref. bib915 |
| Ifenprodil (3689) | Channel inhibition, IC50 = 3.33 μM | ref. bib915 |
a PubChem Compound Identification number.
Recently,ref. bib652,ref. bib653 cryo-EM structures of TRPM7 were solved in complex with NS8593, VER155008, and CCT128930. All 3 inhibitors bind to the same site in TRPM7, located on the cytoplasmic side of the membrane at the interface of the S3, S4, and S5 helices and the TRP domain. This ligand-binding pocket in TRPM7 is called a vanilloid-like site because the homologous cavity in the TRPV1 channel has been previously defined as a vanilloid regulatory site.ref. bib652,ref. bib653 However, whether waixenicin A and FTY720 bind to the vanilloid-like site of TRPM7 or act through an alternative mechanism remains to be examined.
A set of small molecules serving as TRPM7 channel agonists has been identified.ref. bib920,ref. bib921 Among them, naltriben and mibefradil have been characterized in detail (Table 5). Both agents can potently activate TRPM7 currents without depletion of intracellular Mg2+, indicating that both compounds act as true agonists of the TRPM7 channel.ref. bib920,ref. bib921 Consequently, many studies employed naltriben and mibefradil, frequently in combination with TRPM7 inhibitors, to examine the role of this channel in different cellular processes.ref. bib664,ref. 910., ref. 911., ref. 912. More recently, the cryo-EM structure of TRPM7 was solved in the open state in complex with naltriben.ref. bib653 A comparison of the closed and open naltriben-bound structures of TRPM7 uncovered particular conformational rearrangements associated with agonist-induced activation of the TRPM7 channel. Naltriben-binding pockets (4 sites per tetramer) were found at the intersubunit interface, formed by the MHR4/pre-S1 helix of one subunit and the MHR4 domain of the neighboring subunit. Intriguingly, this ligand-binding site has not been identified in TRPMs before.ref. bib653
The selective pharmacological modulators of TRPM7 kinase remain to be identified. Currently, only 1 compound, TG100-115 (Table 5), is known as an inhibitor of TRPM7 kinase activity, but this molecule also inactivates TRPM6 kinase.ref. bib630,ref. bib688,ref. bib922
In contrast to TRPM7, the pharmacological profile of TRPM6 is less established. Recently, 2 structurally unrelated compounds, iloperidone and ifenprodil, were defined as potent inhibitors of the TRPM6 channel (Table 5).ref. bib915 Notably, both reagents showed no impact on the TRPM7 channel.ref. bib915 As mentioned above, NS8593 and FTY720 can suppress TRPM6 currents.ref. bib652,ref. bib653,ref. bib915,ref. bib917 Hence, the available pharmacological toolkit enables selective or combined targeting of TRPM6 and TRPM7 in physiological conditions or preclinical experimental models, for instance, in patient-derived primary cells.
TRPM2 and TRPM8
H2O2– and ADPR-evoked TRPM2 currents can be blocked by several synthetic and natural compounds, including N-(p-amylcinnamoyl)anthranilic acid, tyrphostin AG 490 (AG490), clotrimazole, JNJ-28583113, scalaradial, and 2,3-dihydroquinazolin-4(1H)-one derivative D9 (Table 6).ref. 936., ref. 937., ref. 938., ref. 939., ref. 940., ref. 941., ref. 942., ref. 943., ref. 944., ref. 945., ref. 946., ref. 947., ref. 948., ref. 949., ref. 950., ref. 951., ref. 952., ref. 953., ref. 954., ref. 955., ref. 956. The generation of synthetic analogs of ADPR represents another strategy to target TRPM2. Thus, 8-phenyl-2′-deoxy-ADPR was found to be a potent inhibitor of TRPM2 currents.ref. bib942 Two other synthesized ADPR analogs with substitutions in the pyrophosphate segment of the nucleotide (compounds 7i and 8a) displayed considerable potency and selectivity in the suppression of TRPM2 (Table 6).ref. bib943 TatM2NX is a cell-permeable peptide designed to interact with ADRP binding in TRPM2.ref. bib944 In electrophysiological experiments, TatM2NX was found to be a potent inhibitor of the TRPM2 channel (Table 6).ref. bib944 Despite outstanding progress in structural assessment of TRPM2 channels from different species,ref. 634., ref. 635., ref. 636., ref. 637., ref. 638., ref. 639. the molecular basis underpinning the inhibitory effect of the ligands mentioned above remains unknown. Also, it is worth noting that pharmacological compounds acting as agonists of the TRPM2 channel have not yet been identified.
Table 6: Selected examples of pharmacological modulators of TRPM2 and TRPM8.
| Name (PubChem CIDa) | Effect | References |
|---|---|---|
| TRPM2 | ||
| N-(p-Amylcinnamoyl)anthranilic acid (ACA) (5353376) | Inhibition, IC50 = 1.7 μM | ref. bib936 |
| AG490 (5328779) | Inhibition, IC50 = 0.4 μM | ref. bib937 |
| Clotrimazole (2812) | Inhibition, IC50 = ∼1 μM | ref. bib938 |
| JNJ-28583113 (164628567) | Inhibition, IC50 = 0.13 μM | ref. bib939 |
| Scalaradial (21637538) | Inhibition, IC50 = 0.21 μM | ref. bib940 |
| 2,3-dihydroquinazolin-4(1H)-one derivative D9c (N/A) | Inhibition, IC50 = 3.7 μM | ref. bib941 |
| 8-phenyl-2′-deoxy-ADPR (compound 86c) (N/A) | Inhibition, IC50 = 3 μM | ref. bib942 |
| ADPR analogues 7ic and 8ac (N/A) | Inhibition, IC50 = ∼5 μM | ref. bib943 |
| TatM2NX (154699439) | Inhibition, IC50 = 0.40 μM | ref. bib944 |
| TRPM8 | ||
| (–)-Menthol (16666) | Activation, EC50 = 48 μM | ref. bib945 |
| Icilin (161930) | Activation, EC50 = 0.36 μM | ref. bib946 |
| Acoltremon (WS-12) (11266244) | Activation, EC50 = 0.19 μM | ref. bib947 |
| Azo-menthol (N/A) | Activation, EC50 = 4.4 μM | ref. bib948 |
| (+)-Sesamin (72307) | Inhibition, IC50 = 9.8 μM | ref. bib949 |
| Hispidulin (5281628) | Inhibition, IC50 = 1.7 μM | ref. bib950 |
| Oroxylin A (5320315) | Inhibition, IC50 = 9.7 μM | ref. bib950 |
| AMTB (16095383) | Inhibition, IC50 = ∼1 μM | ref. bib951 |
| M8-B (69316632) | Inhibition, IC50 = 7.8 nM | ref. bib952 |
| TC-I 2000b (compound 87c) (57326210) | Inhibition, IC50 = 36 nM | ref. bib953 |
| AMG 333 (71144018) | Inhibition, IC50 = 13 nM | ref. bib954 |
| RQ-00203078 (49783953) | Inhibition, IC50 = 8.3 nM | ref. bib955 |
| TC-I 2014b (compound 5c) (135883253) | Inhibition, IC50 = 3 nM | ref. bib956 |
a PubChem Compound Identification number. N/A – not available.
TRPM8 has been proposed as a new target for the treatment of pain, and consequently, a very comprehensive collection of TRPM8 modulators has been developed. TRPM8 agonists like menthol, icilin, and WS-12 are broadly used to explore the pharmacological potential of this channel (Table 6).ref. 945., ref. 946., ref. 947. In addition to menthol, other natural products with menthol-like cooling effects are defined as activators of TRPM8, including camphor, rotundifolone, eucalyptol, and borneol.ref. bib957 However, these compounds affect TRPM8 at a high micromolar range of concentrations and elicit multiple effects on other proteins.ref. bib957,ref. bib958 The structures of such cooling agents serve as blueprints for designing dozens of synthetic agents with EC50 values in the nanomolar range.ref. bib957,ref. bib958 These synthetic substances have been predominantly documented in patents from pharmaceutical companies and await further validation.ref. bib957,ref. bib958 Recently, the first photoswitchable TRPM8 activator, azo-menthol, has been developed, which enables optical regulation of TRPM8 currents with UV and blue light (Table 6).ref. bib948
Screening natural products led to the discovery of TRPM8 inhibitors, such as sesamin, hispidulin, and oroxylin A (Table 6).ref. bib949,ref. bib950 In addition, a series of synthetic TRPM8 antagonists have been identified, for instance, AMTB, M8-B, TC-I 2000, AMG 333, RQ-00203078, and TC-I 2014 (Table 6).ref. 951., ref. 952., ref. 953., ref. 954., ref. 955., ref. 956.,ref. bib959 AMTB and TC-I 2014 were used in the cryo-EM analysis of TRPM8, and the resolved structures revealed that, analogously to agonists WS-12 and icilin, both inhibitors interact with a ligand-binding site formed by residues of the S1–S4 segments of TRPM8.ref. bib40,ref. bib654 Finally, it is worth noting that pharmaceutical companies have synthesized several potent inhibitors of TRPM8, but similar to the situation with TRPM8 activators, the functional impacts of these entities on TRPM8 are only briefly reported in patents.ref. bib957,ref. bib958
TRPM4 and TRPM5
In initial studies, several polyspecific channel blockers were used to inhibit TRPM4 currents, for instance, 9-phenanthrol and MPB-104 (Table 7).ref. bib649,ref. 960., ref. 961., ref. 962., ref. 963., ref. 964., ref. 965., ref. 966., ref. 967., ref. 968., ref. 969., ref. 970. However, these agents displayed a low potency toward TRPM4. Subsequently, more potent TRPM4 inhibitors were identified, such as 4-chloro-2-{[2-(2-chlorophenoxy)acetyl]amino} benzoic acid, 4-chloro-2-[2-(naphthalen-1-yloxy)acetamido] benzoic acid, and meclofenamate (Table 7).ref. bib962,ref. bib963,ref. bib971 In mice, meclofenamate inhibited the Ca2+ overload-induced background current in ventricular cardiomyocytes and suppressed catecholaminergic polymorphic ventricular tachycardia-associated arrhythmias in a TRPM4-dependent manner.ref. bib963 U73122 was found to be a potent activator of TRPM4, which can stimulate TRPM4 currents in the absence of intracellular Ca2+ (Table 7).ref. bib964 Another compound, a 3,5-bis(trifluoromethyl)pyrazole derivative (YM-58483), is defined as a potentiator (or enhancer) of the TRPM4 channel because the degree of TRPM4 activation is dependent on the presence of intracellular Ca2+ (Table 7).ref. bib965
Table 7: Pharmacological modulators of TRPM4 and TRPM5
| Name (PubChem CIDa) | Effect | References |
|---|---|---|
| TRPM4 | ||
| 9-Phenanthrol (10229) | Inhibition, IC50 = 17–23 μM | ref. bib960 |
| MPB-104 (11738767) | Inhibition, IC50 = 11–24 μM | ref. bib960 |
| 4-Chloro-2-{[2-(2-chlorophenoxy)acetyl]amino} benzoic acid (CBAb, compound 5c) (2264067) | Inhibition, IC50 = 1.8 μM | ref. bib962 |
| 4-Chloro-2-[2-(naphthalen-1-yloxy)acetamido] benzoic acid (NBAb, compound 6c) (1295523) | Inhibition, IC50 = 0.2 μM | ref. bib962 |
| Meclofenamate (4038) | Inhibition, IC50 = 3.4 μM | ref. bib963 |
| U73122 (104794) | Activation, EC50 = 0.44 μM | ref. bib964 |
| 3,5-Bis(trifluoromethyl)pyrazole derivative BTP2 (YM-58483) (2455) | Potentiation, EC50 = 8–500 nM | ref. bib965 |
| Necrocide 1 (NC1) (49783440) | Activation, EC50 = 0.31 μM | ref. bib966 |
| TRPM5 | ||
| Triphenylphosphine oxide (TPPOb) (13097) | Inhibition, IC50 = 12 μM | ref. bib967 |
| NDNAb (674882) | Inhibition, IC50 = 2.4 nM | ref. bib649 |
| Stevioside (442089) | Potentiation, EC50 = 690 nM | ref. bib968 |
| Benzo[d]isothiazole derivatives 61c, 64c (164611814, 164619590) | Activation, EC50 = 8–44 nM | ref. bib969 |
| Tetrahydroisoquinoline derivative 39c (167993652) | Activation, EC50 = 80 nM | ref. bib970 |
a PubChem Compound Identification number.
Recently, a small molecule, necrocide 1 (NC1), was identified as a potent activator of human TRPM4 but not mouse TRPM4 (Table 7).ref. bib966 Interestingly, upon activation of TRPM4, NC1 induces necrotic cell death because of Na+ overload.ref. bib966 Despite significant progress in cryo-EM analysis of TRPM4,ref. 644., ref. 645., ref. 646., ref. 647., ref. 648. the structural basis for the inhibitory and stimulatory effects of the ligands mentioned above remains unknown.
Several pharmacological agents, such as flufenamic acid, clotrimazole, and quinine, were found suitable for inhibiting the TRPM5 channel.ref. bib747,ref. bib972 However, these compounds were active in the high micromolar range and capable of suppressing TRPM4.ref. bib747,ref. bib972 Subsequently, triphenylphosphine oxide demonstrated improved selectivity and potency toward TRPM5, whereas N′-(3,4-dimethoxybenzylidene)-2-(naphthalen-1-yl)acetohydrazide (NDNA) represents the most potent inhibitor of TRPM5 currents (Table 7).ref. bib649,ref. bib967 Cryo-EM analysis demonstrated that NDNA binds to a cleft between the S1–S4 segment and the S5–S6 helices, known as the vanilloid binding site in TRPVs, stabilizing the channel in a closed conformation.ref. bib649 Recently, NC1 was identified as a compound that induces necrotic cell death through direct activation of the TRPM4 channel through the NDNA-binding site (Table 7).ref. bib966
Several natural compounds are applicable for the positive regulation of TRPM5 (Table 7). Steviol glycosides, such as stevioside, potentiate the Ca2+-dependent activity of the TRPM5 channel and are thus defined as potentiators of TRPM5.ref. bib968 In other studies, high-throughput screening and lead optimization strategies suggested several synthetic compounds to act as potent TRPM5 agonists.ref. bib969,ref. bib970 Among several benzo[d]isothiazole derivatives, 2 molecules (referred to as compounds 61 and 64) activated the TRPM5 channel with EC50 values in the nanomolar range.ref. bib969 A series of tetrahydroisoquinoline-based molecules (ie, compound 39) stimulated TRPM5 with an EC50 of 0.1–10 μM (Table 7).ref. bib970 However, a more comprehensive biophysical assessment is needed to conclude that these ligands open the TRPM5 channel in a Ca2+– and voltage-independent fashion.
Ongoing or completed clinical trials with TRPMs
According to the ClinicalTrials.gov database,ref. bib973 TRPM3 is the subject of a clinical trial (NCT05275751) aimed at examining whether the redundant functions of TRPV1, TRPA1, and TRPM3 observed in mice regarding heat perception are also applicable to humans. Another trial (NCT03252834) is examining whether the genetic variants of TRPM2 represent biomarkers for chemotherapy-induced abnormal thermal sensation in cancer patients. Early treatment of cerebral edema and intracranial pressure is crucial for improving outcomes. The study NCT06017635 investigates whether TRPM4 expression levels can serve as a diagnostic marker for cerebral edema in children. It is well documented that early mobilization of patients in the surgical intensive care unit improves outcomes. The trial NCT01363102 examines whether genetic polymorphisms in TRPM6 and other genes are linked to sleep quality and muscle strength, and whether these associations relate to early mobilization in surgical patients. The project NCT04229992 examines the association between SNPs in TRPM7, dietary intake of calcium and magnesium, and the risk of developing colorectal cancer.
Several ongoing clinical trials assess TRPM8 as a therapeutic target for various pathophysiological conditions. The trial NCT01408446 investigates the impact of the TRPM8 agonist menthol (Table 6) on the prevention of prehypertension and mild hypertension. This trial aims to assess the effects of dietary menthol on blood pressure and metabolic parameters. The study NCT05935280 aims to determine whether TRPM8 contributes to cold pain perception in humans. Cold pain will be experimentally induced by injecting a cooling solution (3 °C) into the skin, along with TRPM8 inhibitors to assess their effects. Experiments with animals indicated that activating TRPM8, which is expressed in the dermal tissue of the limbs, using menthol is beneficial for stroke recovery. The investigation NCT05877079 aims to examine the impact of such treatment on patients with acute ischemic stroke. The trials NCT04711044, NCT04554888, NCT04515056, and NCT03943407 evaluate the effects of menthol on itch induced by histamine, cowhage, and papain. The project NCT03610386 examines the effect of menthoxypropanediol, a derivative of menthol, on pruritus in atopic dermatitis (eczema) using biopsies from patients with atopic dermatitis. Applying menthol topically increases resting energy expenditure, likely by activating brown adipose tissue. The aim of the study NCT07030725 is to determine whether applying menthol to the front of the thorax will boost thermogenesis through brown adipose tissue activation and enhanced blood flow in skeletal muscles. The project NCT01565070 examines whether menthol can alleviate symptoms associated with knee osteoarthritis, thereby reducing immobility and isolation in older adults.
Recently, the FDA approved WS-12, a TRPM8 agonist (Table 6), for the treatment of symptoms associated with dry eye disease.ref. bib974 WS-12 stimulates corneal nerves to promote natural tear production and has been found beneficial in 40% of patients over 90 days of treatment.ref. bib974
On this background, we anticipate that the recently developed modulators of TRPMs will enable the design of new clinical trials in the near future.
TRPAs
TRPA gene family
In humans, TRPA1 is the only member of the ankyrin-repeat TRP channel subfamily (Table 1). It is a polymodal irritant sensor that is expressed in nociceptive neurons and some nonneuronal cell types. Its marked promiscuity to be activated by a plethora of natural products, drugs, and drug-like compounds sets it apart from most other TRP channels and often results in covalent modification or indirect mechanisms, which include the formation of ROS or oxidized membrane lipids.
Domain topology, assembly, and functional characteristics of TRPA1
Domain topology and channel assembly
A prominent and name-giving property of TRPA1 is its extended N-terminal ARD, featuring approximately 16 (14–18, depending on the species) consecutive ankyrin-like folds that consist of about 33 amino acids each. This N-terminal ARD makes up most of the intracellular volume of the channel protein. The 3D structure of mammalian TRPA1 has been elucidated by cryo-EM, first at a rather low resolution of about 16 Å,ref. bib975 and more recently, at a resolution of 4 Åref. bib976 or ∼3 Å,ref. bib297 which allowed for a more reliable reconstruction. While the first 11 ankyrin repeats appear as a concave crescent-like density that extends away from the central symmetry axis, the ankyrin repeats 12–16 closely surround a central bundle of the 4 C-terminal located α-helical structures, which engage in CC helices with their respective neighbors. A prominent regulatory site is located in the linker region between the ARD and the first TM segment S1 region. This linker, also referred to as the coupling domain, contains cysteine (C621 and C641) and lysine (K710) residues that surround a binding pocket and can be covalently modified by electrophilic TRPA1-activating drugs.ref. bib297
Like other TRP channels, the TMD can be subdivided into a VSLD comprising the first 4 TM-spanning segments S1–S4, a helical S4–S5 linker, a pore-forming fold that consists of TM helix S5, the re-entrant pore loop, and a tilted S6 helix, which strongly constricts the pore in its closed conformation. The pore loop features 2 short helical segments that position a string of 3 glutamate residues (E920, E924, and E930) to generate a negatively charged surface in and around the extracellular pore mouth. The most centrally positioned E920, together with D915, is a key feature of the selectivity filter that constricts the pore and divides the permeation pathway into an outer vestibule and an inner cavity.ref. bib977 At present, structural data on the open channel state of TRPA1 are still lacking. Hypothetical models postulate a rotation of S6 that repositions the strongly constricting hydrophobic amino acids I957 and V961 away from the central axis, which then may be flanked by E966 and open to form a hydrophilic cation-conducting pore.ref. bib978 The TMD is followed by a TRP-like domain and the aforementioned C-terminal CC-forming domain.
Finally, the TMD features grooves and clefts that allow the noncovalent binding of allosteric TRPA1-modulating drugs. An intersubunit cleft between S4 and the S4–S5 linker of one subunit, and S5 and S6 of the neighboring subunit, has been demonstrated to adopt GNE551, a noncovalent TRPA1 activator.ref. bib979 The TRPA1 antagonist A-967079 most likely binds to a pocket formed in the upper part of the TMD flanked by S5, S6, and the first PH.ref. bib976 Figure 5 provides a graphical illustration of the domain topology of TRPA1.
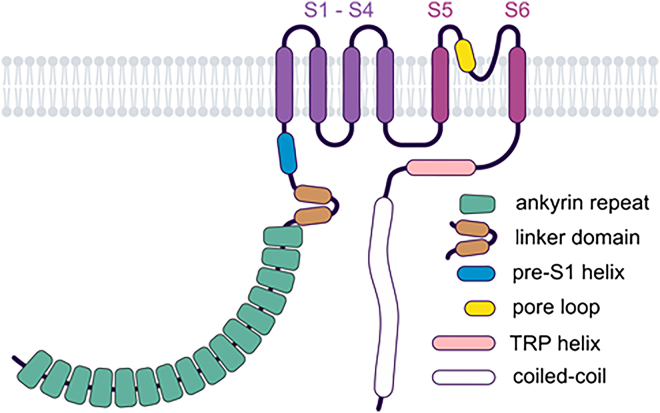
All structural biology data on recombinantly expressed TRPA1 channel complexes confirm the expected homotetrameric conformation with rotational symmetry (see PDB entries provided in Table 1). Evidence of heteromeric channel assemblies composed of TRPA1 and TRPV1 subunits was obtained through coimmunoprecipitation analyses, Förster resonance energy transfer, single-channel current properties, and atomic force microscopy of heterologously expressed concatemers.ref. bib384,ref. bib980 The extent and physiological, pathophysiological, and pharmacological relevance of a possible heteromeric assembly have not yet been clearly defined. Moreover, the complexes may either result from a heterotetrameric assembly, as suggested by Fischer et al,ref. bib384 or from functional interaction of side-by-side assemblies of homotetrameric complexes that may interact directly via a bridging protein,ref. bib981 such as TMEM100, or by an A kinase-anchoring protein (AKAP79/150) as a scaffold.ref. bib982
Functional characteristics of TRPA1 channel complexes
Due to its responsiveness to numerous compounds and conditions and its strong expression in nociceptive or chemosensory afferent neurons, there is a broad consensus that TRPA1 acts as a polymodal irritant sensor. A plethora of input queues converge toward TRPA1 activation, including the covalent binding of cysteine-reactive or other electrophilic activators,ref. bib983,ref. bib984 the indirect effects of compounds that induce lipid peroxidation, such as the formation of 4-hydroxynonenal,ref. bib985 ultraviolet light, or phototoxic substances that can generate ROS upon illumination,ref. bib986,ref. bib987 ROS themselves,ref. bib988 as well as nonreactive TRPA1 activators, some of which are summarized below. In addition, TRPA1 activity can be further potentiated by intra- and extracellular Ca2+ ions,ref. 990., ref. 991., ref. 992. or via GPCRs that sense inflammatory mediators.ref. bib992,ref. bib993
Initial findings suggesting that TRPA1 may sense noxious cold temperaturesref. bib994 were soon challenged,ref. bib995 and the results strongly relied on the investigated species.ref. bib996,ref. bib997 Genetic as well as pharmacological evidence has even attributed TRPA1 to contribute to a heat-sensing pathway.ref. bib765 Human TRPA1 seems to be both cold- and heat-sensitive, and structural determinants for thermal activation have been identified.ref. bib998
With respect to its biophysical properties, TRPA1 mediates poorly selective cation currents that show a marked outward rectification when recorded in the presence of physiological concentrations of divalent cations.ref. bib977,ref. bib990 Depending on the applied activator, TRPA1 inward currents exhibit a slight and variable 2- to 8-fold preference for divalent versus monovalent cations.ref. bib999,ref. bib1000 Likewise, unitary current amplitudes strongly depend on the composition of bath and pipette solutions and the applied agonists. In isotonic solutions, they range between 48 pS for inward currents measured in the presenceref. bib999 of 5 mM intracellular Mg2+ and 251 pS in symmetrical Cs+ solutionsref. bib977 containing only 0.1 mM Mg2+. The smaller, nonflickering unitary currents in the presence of divalent cations that can permeate through the TRPA1 pore are consistent with the assumption that divalent cations act as permeating blockers.ref. bib1001 Applying charge carriers of different sizes, a pore diameter of at least 8.2 Å has been determined,ref. bib977 and the pore may further dilate up to 13.8 Å when activated by mustard oil.ref. bib999 Accordingly,ref. 1003., ref. 1004., ref. 1005. TRPA1 prolonged activation by a variety of reactive or nonreactive agonists allows permeation of the organic cations Yo-Pro-1 or NMDG+, a phenomenon that has been attributed to pore dilation. Alternatively, pore dilation might be mimicked by changed intracellular ion concentrations.ref. bib435
Expression pattern and primary physiological roles of TRPA1
Owing to its primary role as an irritant sensor, a strong expression of TRPA1 is found in sensory afferent neurons whose cell bodies are located in the dorsal root and trigeminal ganglia.ref. bib994,ref. bib1005 In these sensory neurons, TRPA1 expression strongly overlaps with that of TRPV1, qualifying them as nociceptors. Likewise, TRPA1 expression in enterochromaffin cells confers chemosensation and is coupled to the release of serotonin to regulate intestinal motility.ref. bib1006 A moderate or low expression of TRPA1 is found in many other tissues and cell types, including various types of cancer, as recently reviewed,ref. bib1007 and summarized in Table 1.
In general, chemosensation is governed by unpleasant smell, bitter taste, and pain, causing avoidance behaviors and controlling protective reflexes that limit exposure to potentially harmful substances. Being expressed in nociceptive neurons and cells that can confer cough or vomiting reflexes, TRPA1 is strategically positioned to recognize irritants and other xenobiotics, preventing further ingestion. Owing to species-dependent thermal TRPA1 activation, nocifensive responses may include the avoidance of potentially noxious hot or cold temperatures.
Human diseases associated with the TRPA1 channel
A rare disease-causing mutation in human TRPA1 gives rise to familial episodic pain syndrome (OMIM 615040). A single family with 21 affected members in 4 generations has been identified to carry an N885S missense mutation in S4, leading to a GOF variant of TRPA1 with 5-fold increased inward currents at normal resting potential, loss of outward rectification, and a shift in cinnamaldehyde (100 μM)-induced, voltage-dependent activation of about 56 mV, causing higher channel activity at the physiological range of membrane potentials.ref. bib1008 Epigenome-wide analyses of DNA methylation in twins with discordant sensitivity to heat-induced pain revealed that an increased pain sensitivity correlated with the demethylation of the TRPA1 promoter, possibly resulting in an enhanced channel expression in nociceptive neurons.ref. bib1009
Besides the direct contributions of TRPA1 in pain perception and neuralgia, numerous physiological and pathophysiological responses have been described to involve TRPA1-mediated processes. A prominent theme is the role of TRPA1 in pulmonary and intestinal vagal afferents to sense irritants, thereby contributing to protective reflexes like cough and emesis or vomiting, but also in exaggerated responses, such as bronchial or visceral hypersensitivity, allergic responses, and chronic obstructive pulmonary disease (COPD), or its contribution to the development of various skin diseases, IR damage, migraine, and other forms of headache, as recently reviewed.ref. bib1010,ref. bib1011 TRPA1 activation in pulmonary fibroblasts has been shown to prevent the transition into myofibroblasts and may thus protect from the development of lung fibrosis.ref. bib1012,ref. bib1013 In addition, TRPA1 expression in the vascular endothelium may be exploited to treat cardiovascular diseases,ref. bib1014 and aberrant TRPA1 expression in various malignancies has been proposed to enable cancer cells to sense and cope with conditions of oxidative stress.ref. bib1015
Pharmacological modulators of the TRPA1 channel
TRPA1-activating compounds
Besides the role of TRPA1 in direct or indirect responses to noxious cold or hot temperatures, TRPA1 may be regarded as a broadly specific irritant sensor. Owing to its unique sensitivity toward cysteine-modifying compounds and ROS, TRPA1 is activated by a plethora of chemical stimuli. They may be grouped into (1) pungent tastants and spices, (2) oxidants and phototoxic compounds, (3) approved drugs, and (4) specifically developed TRPA1-selective activators. Table 8ref. bib979,ref. bib983,ref. bib985,ref. 1017., ref. 1018., ref. 1019., ref. 1020., ref. 1021., ref. 1022., ref. 1023., ref. 1024., ref. 1025., ref. 1026., ref. 1027., ref. 1028., ref. 1029., ref. 1030., ref. 1031., ref. 1032., ref. 1033., ref. 1034., ref. 1035., ref. 1036., ref. 1037., ref. 1038., ref. 1039., ref. 1040. summarizes some of the most prominent TRPA1-activating compounds, as well as TRPA1 inhibitors or blockers.
Table 8: Pharmacological modulators of TRPA1.
| Name (PubChem CIDa) | Effect | References |
|---|---|---|
| JT010 (18524489) | Channel activation, EC50 = 0.65 nM | ref. bib1016 |
| PF-4840154 (53380803) | Channel activation, EC50 = 23 nM | ref. bib1017 |
| GNE551 (2135890) | Channel activation, EC50 = 254 nM | ref. bib979 |
| Dibenzoxazepine (9213) | Channel activation, EC50 = 63 nM | ref. bib1018 |
| Morphanthridine (10878016) | Channel activation, EC50 = 83 nM | ref. bib1018 |
| Acrolein (7847) | Channel activation, EC50 = 5 μM | ref. bib1016 |
| Allyl isothiocyanate (5971) | Channel activation, EC50 = 11-64.5 μM | ref. bib983,ref. bib1019 |
| Allicin (65036) | Channel activation, EC50 = 7.5 μM) | ref. bib1020 |
| Thymol (6989) | Channel activation, EC50 = 127 μM | ref. bib1021 |
| Menthol (16666) | Channel activation, EC50 = 95 μM | ref. bib1022 |
| Isoflurane (3763) | Channel activation, EC50 = 180 μM | ref. bib1023 |
| Apomorphine (6005) | Channel activation, EC50 = 7.1 μM | ref. bib1024 |
| Auranofin (16667669) | Channel activation, EC50 = 1 μM | ref. bib1025 |
| Isovelleral (37839) | Channel activation, EC50 = 0.5 μM | ref. bib1026 |
| Flufenamic acid (3371) | Channel activation, EC50 = 147 μM | ref. bib1027 |
| Clopidogrel (60606) | Channel activation, EC50 = 5.4 μM | ref. bib1028 |
| Ticlopidine (5472) | Channel activation, EC50 = 7.2 μM | ref. bib1028 |
| Nicotine (89594) | Channel activation, EC50 = 17 μM | ref. bib1019 |
| 2-Iodoacetamide (3727) | Channel activation, EC50 = 357 μM | ref. bib1029 |
| 2-Methylsulfonothioyloxyethanamine (MTSEA) (53443082) | Channel activation, EC50 = 1.58 mM | ref. bib1029 |
| 4-Hydroxynonenal (5283344) | Channel activation, EC50 = 13-27 μM | ref. bib985,ref. bib1030 |
| HC-030031 (1150897) | Channel inhibition, IC50 = 0.7-6.2 μM | ref. bib1031 |
| A-967079 (42641861) | Channel inhibition, IC50 = 51 nM | ref. bib1032 |
| AP18 (9584673) | Channel inhibition, IC50 = 3.1 μM | ref. bib1033 |
| LY3526318 (118961431) | Channel inhibition, IC50 = 13.5 nM | ref. bib1034 |
| AM-0902 (73297271) | Channel inhibition, IC50 = 131 nM | ref. bib1035 |
| BAY-390 (155539293) | Channel inhibition, IC50 = 16 nM | ref. bib1036 |
| GDC-0334 (122490062) | Channel inhibition, IC50 = 1.7 nM | ref. bib1037 |
| Ruthenium red (656819) | Channel block, IC50 < 10 μM | ref. bib1031,ref. bib1038,ref. bib1039 |
N/A, not available.
Pungent tastants, spices, and natural products
Soon after the initial characterization of TRPA1, its activation by mustard oil, cinnamon oil, ginger, and others was recognized.ref. bib1005,ref. bib1040 Allicin, the spicy and unstable ingredient of garlic, strongly activates TRPA1, whereas its heat-derived conversion products, diallyl mono-, di-, and trisulfide, less strongly and/or less potently act on the channel.ref. bib1041 Other natural compounds that cause TRPA1 opening include menthol, thymol, and nicotine.ref. bib1019,ref. bib1021,ref. bib1022,ref. bib1042 One should note that most compounds require concentrations of 10–300 μM to elicit strong effects on TRPA1, and some of them (menthol, cinnamaldehyde, nicotine, and camphor) exert bimodal effects with a current inhibition when applied at even higher concentrations.ref. bib1022,ref. bib1043
ROS, peroxidation products, and cysteine-modifying compounds
In chemosensory neurons, TRPA1 is a prominent molecular substrate that decodes ROS or chemical oxidants either directly or via the formation of peroxidation products of membrane lipids. Effective oxidants include hydrogen peroxide, hypochlorite,ref. bib1044 and cysteine-modifying compounds, such as 2-methylsulfonothioyloxyethanamine or iodoacetamide.ref. bib1029 While several cysteine-modifying compounds can covalently bind to TRPA1, ROS, ultraviolet light, or visible light in the presence of photosensitizing compoundsref. bib986 are likely to act in an indirect fashion, eg, by peroxidation products of membrane lipids, such as 4-hydroxy-2-nonenal, 4-oxo-nonenal, and 4-hydroxyhexenal or oxidized phospholipids.ref. bib985,ref. bib1030,ref. bib1045,ref. bib1046
Approved drugs
Since several FDA-approved drugs or drug metabolites are capable of activating TRPA1, stimulation of chemosensory neurons and vagal afferents may contribute to adverse responses to the respective drugs. Acrolein, an irritating and highly electrophilic metabolite of cyclophosphamide, activates the TRPA1 channel.ref. bib995 The pungent smell of the TRPA1-activating volatile anesthetics isoflurane and desflurane limits their application during the induction of general anesthesia and may be linked to TRPA1 activation.ref. bib1023 Stinging pain during photodynamic therapy may be caused by ROS that indirectly activate TRPA1.ref. bib1047 Other TRPA1-activating drugs include several fenamates, apomorphine, auranofin, ticlopidine, and clopidogrel.ref. bib1024,ref. bib1025,ref. bib1027,ref. bib1028 In most cases, therapeutic plasma concentrations are much lower than the concentrations required to activate TRPA1. Nonetheless, high local concentrations of TRPA1-activating drugs may contribute to adverse gastrointestinal effects during oral application.
Selective, highly potent, and photoswitchable TRPA1 activators
To gain a deeper insight into TRPA1-mediated functions and to further validate TRPA1 as a potential drug target, highly potent and selective TRPA1 activators are eagerly sought. Any kind of electrophilic activator would be highly prone to exerting off-target effects. Therefore, electrophilic TRPA1 activators such as JT010 may be regarded as second-generation tool compounds despite their increased potency.ref. bib1016 The nonelectrophilic peptide TRPA1 activators, PF-4840154 or GNE551, are more drug-like and may provide some advantages as activating compounds in screening approaches.ref. bib979,ref. bib1017 Interestingly, GNE551, as well as a pain-inducing plasma membrane-permeable scorpion toxin WaTx, induce TRPA1 activation modes that differ from that induced by allyl isothiocyanate, with a lack of current inactivation and longer single-channel opening events, respectively.ref. bib1048 By identifying TRPA1-activating compounds with reversibly photoswitchable azobenzene or azopyrazole moieties, optical control of zebrafish or human TRPA1 activity has been achieved.ref. bib1049,ref. bib1050
TRPA1 inhibitors
Like in many other TRP channels, inward currents through TRPA1 are blocked by ruthenium red, a polycationic compound, which presumably plugs into the outer entrance of the pore and obliterates the permeation pathway in a voltage-dependent fashion.ref. bib1038 The first TRPA1-selective inhibitor developed by Hydra Biosciences, HC-030031, acts in a voltage-independent fashion and has been demonstrated to counteract formalin-induced pain upon intraperitoneal application in rats.ref. bib1031 Two inhibitory compounds, AP18 and A967079, share a styrene pharmacophore and are structurally related to the activating compound cinnamaldehyde. While the inhibitory potency of AP18 lies in the micromolar range, A967079 reaches a nanomolar potency, especially with regard to inhibition of human TRPA1.ref. bib1033,ref. bib1051
In a preclinical study, using a guinea pig model of chronic cough, intraperitoneally applied GRC 17536 (60–100 mg/kg) was more effective in suppressing citric acid-induced cough responses than dextrometorphan (50 mg/kg) as a comparator drug.ref. bib1052 In the patent literature, more drug-like TRPA1-targeting compounds with low to mid-nanomolar inhibitory potency have been proposed by Bayer (WO2021233752A1), MSD (WO2011043954A1), Hofmann La-Roche (WO2019182925A1 and WO2018029288A1), Genentech (WO2018162607A1), Orion Pharma (WO2014053694), and Eli Lilly (WO2019152465A1), some of which share striking similarities with GRC 17536 or LY3526318. Bayer has decided to make one of these compounds, BAY-390, publicly available as an orally bioavailable and CNS-penetrant drug that inhibits rat and human TRPA1 with comparable potency and may facilitate further validation of TRPA1 as a potential pharmacological target in a variety of preclinical disease models.ref. bib1036
Ongoing or completed clinical trials with the TRPA1 channel as a therapeutic target
Owing to its irritant-sensing properties, TRPA1 has gained considerable interest as a pharmacological target in diseased conditions to control symptoms such as pain, itch, cough, or neurogenic inflammation, including migraine.
Among the first completed phase 1 and 2 trials, Glenmark Pharmaceuticals Ltd has focused on the compound GRC 17536, a thienopyrimidinedione derivative, as either a systemically applied or inhaled TRPA1 antagonist. A combined phase 1/2 trial tested the safety, tolerability, and efficacy of inhaled GRC 17536 in patients with mild allergic asthma. While inhaled GRC 17536 met the safety endpoints in single and repeated application regimes, the drug failed to reach the primary efficacy endpoint to reduce the drop of the 1-second forced expiratory volume (FEV1) after an allergen challenge (Eudra CT: 2012-002567-99). In a cohort of elderly patients suffering from chronic cough, inhaled GRC 17536 again failed to prove effective (Eudra CT: 2013-002728-17). In a third study, GRC 17536 (250 mg) or placebo was administered twice daily orally for 28 days to diabetic patients suffering from peripheral neuropathic pain. After 4 weeks of treatment, a significant decline in the average pain intensity was not achieved as the primary endpoint for all participants in the subgroup with moderate to severe pain (Eudra-CT: 2012-002320-33). No other studies with GRC 17536 have since been reported, implying that further development of this candidate may have been stopped.
More recently, Eli Lilly has embarked on another series of clinical trials, aiming at validating the purin-based compound LY3526318 as a potential analgesic drug in osteoarthritis (NCT05080660), low back pain (NCT05086289), or diabetic neuropathy (NCT05177094). According to information provided via clinicaltrials.gov, all 3 studies have been completed in 2022. As of the time of writing, results remain undisclosed, and a possible progression into phase 3 trials has not been announced yet. In oropharyngeal dysphagia, various TRP channel activators have been probed for possible relief and reconstitution of safe swallowing. Notably, activators of TRPV1 (capsaicin and piperine) or TRPA1 (cinnamaldehyde and citral) provided the best results, highlighting a possible use of TRPA1-targeting sensory stimulants in deglutition disorders.ref. bib1053
TRPMLs
Introduction
The TRP channels TRPML1, TRPML2, and TRPML3 (also called MCOLN1–3 or mucolipin1–3) are Ca2+-permeable, nonselective cation channels expressed in early endosomes, late endosomes, recycling endosomes, and lysosomes. TRPMLs are like the other TRP channels, 6 TMD proteins. They form tetramers, with the channel pore between TMD5 and 6. Since 2016, numerous structures have been determined for all 3 TRPML members using either X-ray crystallography or cryo-EM (see “Domain topology, assembly, and functional characteristics of TRPMLs”).ref. 1055., ref. 1056., ref. 1057., ref. 1058., ref. 1059., ref. 1060., ref. 1061.
In contrast to TRPML1, which is ubiquitously expressed, TRPML2 is predominantly found in immune cells, while TRPML3 is found in immune cells, other specialized cells, such as melanocytes or hair cells of the inner ear, and endocrine glands and secretory cells, as recently shown by a whole-body analysis of TRPML3 expression in a GFP-reporter mouse model.ref. bib1061 TRPMLs mediate cation flux from endosomes and lysosomes, sense endolysosomal pH, regulate membrane potential across endolysosomal membranes, regulate trafficking, exocytosis, endocytosis/phagocytosis, and autophagy in the endolysosomal system, and participate in lysosomal biogenesis, cell membrane repair, cell migration, and nutrient sensing (see “Expression pattern and primary physiological roles of TRPMLs”).
LOF mutations in the human TRPML1 gene (>50 deletions, point mutations, in-frame deletions, early stop mutations, and other types of mutations) cause the neurodegenerative lysosomal storage disorder mucolipidosis type IV (MLIV) in humans. MLIV is characterized by psychomotor abnormalities, corneal clouding, retinal degeneration, and achlorhydria, which results in an increase in blood gastrin levels, iron deficiency due to an absence of acid secretion in the stomach, and endolysosomal accumulation of macromolecules, lipids, and heavy metals like zinc and iron in endolysosomes throughout the body.ref. 1063., ref. 1064., ref. 1065., ref. 1066., ref. 1067., ref. 1068. In human TRPML2, an SNP, which is common in certain African populations, results in the TRPML2 variant TRPML2K370Q. TRPML2K370Q reportedly disrupts the ability of the channel protein to enhance viral infections, raising the possibility of altered susceptibility to certain viral infections in homozygous carriers of this and possibly other TRPML2 polymorphisms.ref. bib1068 Mutations in TRPML3, TRPML3,A419P and TRPML3I362T/A419P are GOF mutations causing deafness and circling behavior in mice (Varitint-waddler mutants).ref. 1070., ref. 1071., ref. 1072., ref. 1073., ref. 1074. Equivalent mutations in the human isoform likewise result in strong GOF.ref. bib1069 The discovery of humans homozygous for early stop codon variants of TRPML2 and TRPML3 (TRPML2K329∗ and TRPML3R390∗) argues that LOF of either TRPML2 or TRPML3 is not lethal.ref. bib1074 Nothing is, however, known about the pathophysiological features or disease susceptibility of individuals carrying these mutations. In addition to the aforementioned pathologies, roles of TRPMLs in cancer, lung disease, cardiovascular and kidney disease, Alzheimer’s and Parkinson’s disease (PD), inflammation and immunity, osteoclast function and bone remodeling, muscular dystrophy, and intestinal pathology have been suggested (see “Human diseases associated with TRPMLs and mouse models”).
Endogenous activators of TRPMLs are the phosphoinositides phosphatidylinositol 3,5-bisphosphate (PI[3,5]P2) and phosphatidylinositol 3-phosphate (PI3P), the latter being a demonstratedref. bib1075 agonist of TRPML3, while PI(3,5)P2 activates all 3 isoforms. PIKfyve, a FYVE finger-containing phosphoinositide kinase, catalyzes the conversion from PI3P to PI(3,5)P2, the latter being predominantly found on late endosomal/lysosomal membranes, while PI3P is found on early endosomal and autophagosomal membranes.ref. bib1075,ref. bib1076 Besides the discovery of endogenous ligands, in over 10 years, a plethora of small-molecule agonists and a number of antagonists have been identified, mostly through high-throughput screenings, and were subsequently functionally characterized (see “Pharmacology of TRPMLs”).
TRPML1 as a drug target, specifically TRPML1 activation to treat, eg, lysosomal storage disorders and other neurodegenerative diseases, has gained much attention in recent years. Thus, according to publicly available information, Calporta Therapeutics, acquired by Merck in 2019, has developed preclinical stage TRPML1 agonists for potential treatment of Niemann-Pick C disease (NPC) and other lysosomal storage diseases, as well as amyotrophic lateral sclerosis, Alzheimer’s disease, and PD. Caraway Therapeutics, bought by Merck in 2023, has developed, with support from the Michael J. Fox Foundation, TRPML1 agonists for GBA-PD treatment. And Casma Therapeutics has likewise developed TRPML1 agonists according to the Alzheimer’s Drug Discovery Foundation (see “TRPMLs as therapeutic targets”).
In sum, we will discuss here the current knowledge of TRPMLs from structural aspects to function and physiology, including pathophysiology, and potential therapeutic applications, including currently available pharmacological tools to modulate TRPML channel activity.
Domain topology, assembly, and functional characteristics of TRPMLs
TRPMLs have, like other TRP channels, long been postulated to comprise 6 TMDs with a pore (P) loop between TMD5 and 6, and the functional pore being formed by tetrameric assembly.ref. bib1077 Structural evidence available since 2016 eventually confirmed these predictions.ref. 1055., ref. 1056., ref. 1057., ref. 1058., ref. 1059., ref. 1060., ref. 1061. The TRPMLs together with the TRPPs differ from the rest of the TRP channels due to the presence of a large extracellular/luminal loop between TMD1 and 2. Structural analysis revealed that the 4 luminal linker domains form a square-shaped canopy with a central opening above the channel pore.ref. bib1054 The canopy in TRPMLs forms a cap-like structure and acts as a highly negative electrostatic trap or sink, which facilitates ion selection by favourably attracting divalent Ca2+ ions, limiting the access of monovalent cations to the filter, thereby reducing the permeation of monovalent ions.ref. bib1059 All TRPMLs are activated by PI(3,5)P2, and several amino acids have been identified either by functional assays (eg, endolysosomal patch-clamp) or in structural studies to affect PI(3,5)P2 binding, eg, K55, R61, K65, R318, and R322 in TRPML1 (Fig. 6).ref. bib1054,ref. bib1056,ref. bib1066,ref. 1069., ref. 1070., ref. 1071., ref. 1072., ref. 1073., ref. 1074., ref. 1075.,ref. 1079., ref. 1080., ref. 1081., ref. 1082., ref. 1083., ref. 1084. Two additional amino acids in TRPML1, Y355 and R403 were postulated to be involved in PI(3,5)P2 activation. Thus, the phosphate group of PI(3,5)P2 induces Y355 to form a π-cation interaction with R403, moving the TMD4–5 linker, resulting in an allosteric activation of the channel.ref. bib1078
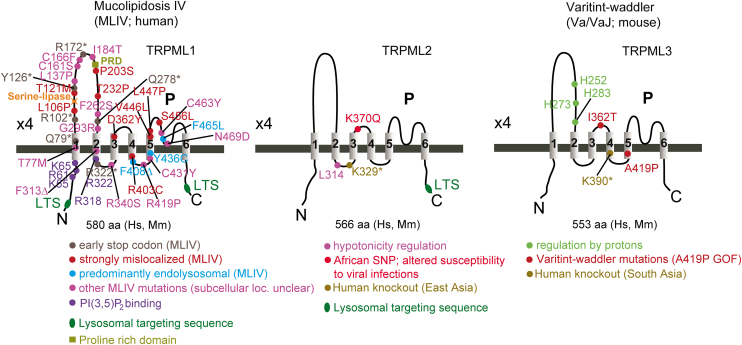
Notably, R403K is an MLIV-causing mutation in human TRPML1 (Fig. 6) that cannot be significantly activated by PI(3,5)P2 anymore. By contrast, the small-molecule agonists ML-SA1 and MK6-83 (Table 9)ref. bib1061,ref. bib1064,ref. bib1066,ref. bib1068,ref. bib1069,ref. 1080., ref. 1081., ref. 1082.,ref. 1085., ref. 1086., ref. 1087., ref. 1088., ref. 1089., ref. 1090., ref. 1091., ref. 1092., ref. 1093., ref. 1094., ref. 1095., ref. 1096., ref. 1097., ref. 1098., ref. 1099., ref. 1100., ref. 1101., ref. 1102., ref. 1103. can still activate this mutant, albeit to a much lesser extent than WT.ref. bib1066,ref. bib1078 In humans, >50 MLIV caused by homozygous TRPML1 mutations or heterozygous combinations of mutations has been identified. The most common mutation is MCOLN1IVS3-2A>G, a splicing mutation (AJ major),ref. bib1103,ref. bib1104 followed by AJ minor (MCOLN1511del6434 or MCOLN1Ex1-7del), both resulting in the loss of a functional gene product. Other mutations comprise single point mutations (either correctly localized in lysosomes (blue), substantially mislocalized (red), or of unknown subcellular localization; marked in pink), in-frame deletions (F313Δ, F408Δ), and early stop mutations (Q79∗, R102∗, Y126∗, R172∗, Q278∗, and R322∗). In addition, small deletions or insertions leading to frame shifts, splicing mutations and complex rearrangements can also occur (see Krogsaeter et alref. bib1079; “Expression pattern and primary physiological roles of TRPMLs”; Fig. 6). While most of the point mutations are severely mislocalized, some retain their lysosomal localization and at least residual activity,ref. bib1066 eg, F408Δ and F465L. Y436C, however, despite being correctly expressed in lysosomes, was shownref. bib1066 to be neither activated by PI(3,5)P2 nor the synthetic agonist MK6-83. This is in accordance with data provided by Schmiege et al,ref. bib1058 demonstrating that Y436A is not activatable by the synthetic ligand ML-SA1, as this amino acid, Y436, together with I468, F428, C429, C432, Y436, F465, F505, F513, Y499, and Y507 forms a hydrophobic cavity accommodating the agonist ML-SA1. Like Y436A, F465A reportedly cannot be activated by ML-SA1 in whole-cell patch clamp experiments using a plasma membrane redirected TRPML1 channel.ref. bib1058 By contrast, Chen et alref. bib1066 reported that F465L can still be activated with the synthetic small-molecule agonist of TRPML1, MK6-83, in endolysosomal patch-clamp experiments. Furthermore, Chen et alref. bib1066 found that mutation F465L has lost its pH sensitivity, ie, pH 4.6, in the lysosomal lumen and cannot further increase channel activity compared with pH 7.2, as seen typically in WT TRPML1.ref. bib1066,ref. bib1095 In contrast to TRPML1, which shows increasing activity with decreasing (ie, more acidic) luminal pH, TRPML3 activity increases with increasing pH, ie, from luminal 4.6 to 6.8 or 7.2 (Fig. 7).ref. bib1083 Since a pH of 6.8 is common in early endosomes, TRPML3 would naturally be more active in early rather than late endosomes or lysosomes. Indeed, functionally TRPML3 seems largely silent in endogenous acidic lysosomes and only becomes active if the luminal pH of the lysosomes rises, eg, under pathogenic conditions (see Miao et alref. bib1105 and “Expression pattern and primary physiological roles of TRPMLs”).
Table 9: Summary of TRPML channel characteristics
| Name | TRPML1 | TRPML2 | TRPML3 |
|---|---|---|---|
| Synonyms | MCOLN1, Mucolipin1 | MCOLN2, Mucolipin2 | MCOLN3, Mucolipin3 |
| Length (aa) Hs | 580 | 566 | 553 |
| Length (aa) Mm | 580 | 566 (538; isoform 2) | 553 |
| Seq motifs | Serine lipase; Lysosomal targeting seq. (N- and C-terminal); Proline rich domain (PRD) | Lysosomal targeting seq. (LTS; N-terminal in Mm) | N.D. |
| Localization | Late endosomes (LE)/lysosomes (LY); LRO? | Rab4+ and Rab11+ recycling endosomes (RE); early endosomes (EE)ref. bib1080,ref. bib1081; LE/LYref. bib1081; LRO?; PM (in-vitro/OE) | EE; LE/LY; not in RE; melanosomes? PM (in-vitro/OE); phagophore during autophagy |
| Tissue distribution | Ubiquitous | Thymus, spleen, kidney, trachea, liver, lung, colon, testis, thyroid, B- and T-cells, macrophages, dendritic cells | Hair cells of the inner ear, organ of corti, utricle, stria vascularis, lung (alveolar macrophages), (skin) melanocytes, (neonatal) enterocytes, kidney, lung, olfactory bulb (sensory neurons), nasal cavity, thymus, colon, trachea, several glands (parathyroid, salivary, adrenal, pituitary), testes, ovariesref. bib1061,ref. bib1069,ref. 1085., ref. 1086., ref. 1087. |
| Activators | PI(3,5)P2 (endogenous)ML1-SA1 (EVP-169) = isoform selectiveref. bib1087Others:ML-SA1 (not isoform selective)ref. bib1088SF-22, SF-51 (not isoform selective)ref. bib1088MK6-83 (EC50, 0.11 μM (patch-clamp))ref. bib1066ML-SA3 (isoform selectivity unclear), ML-SA5 (isoform selectivity unclear)ref. bib1089Rapamycinref. bib1090NAADP? | PI(3,5)P2 (endogenous)ML2-SA1 (EVP-22) = isoform selective (EC50, 1.2 μM (Ca2+ imaging))ref. bib1081Others:ML-SA1 (not isoform selective)SF-21; SF-41; SF-81 (not isoform selective)ref. bib1064Rapamycinref. bib1090(+)-trans-ML-SI3 = TRPML2 agonist (see section on inhibitors) | PI3P, PI(3,5)P2 (endogenous)ML3-SA1 (EVP-77; mouse isoform selective; EC50, 9 μM (Ca2+ imaging)ref. bib1087EVP-21 (human isoform selective; EC50, 4.3 μM (Ca2+ imaging)ref. bib1091Others:ML-SA1 (not isoform selective)SF-11; SN-1; SF-21; SF-22; SF-31; SF-23; SF-41; SF-51; SF-32; SF-24; SF-33; SN-2; SF61; SF-71; SF-81ref. bib1084 |
| Inhibitors | PIP2 (endogenous)ML-SI1 (not isoform selective; stereochemistry of the active isomer not elucidated; dependent on activator)ML-SI2 (structure not published)(–/+)-trans-ML-SI3 (not isoform selective)ref. bib1092; racemic trans-isomer commercially available; both enantiomers available by enantioselective synthesisref. bib1093EDME (isoform-selective; IC50, 0.6 μM (Ca2+ imaging) and 0.2 μM (patch-clamp))ref. bib1094PRU-10, PRU-12 (EDME derivatives; isoform selective; IC50, 0.4 and 0.3 μM (Ca2+ imaging))ref. bib1094High luminal pHSphingomyelins (SMs) | PIP2 (endogenous)ML-SI1, (–)-trans-ML-SI3 (not isoform selective; (+)-trans-ML-SI3 = TRPML2 agonist!)ref. bib1092Low luminal pH | P)P2 (endogenous)(–)-trans-ML-SI3 (effect weaker than for TRPML1 and TRPML2)ref. bib1092No other synthetic small molecule blockers currently availableLow luminal pH and high luminal Na+ |
| Regulators | Acidic luminal pH increases activityref. bib1066,ref. bib1095 | Acidic luminal pH reduces activityref. bib1081 | Low luminal Na+ potentiates activationAcidic luminal pH reduces activityref. bib1096 |
| Disease mutations or polymorphisms associated with a phenotype | MLIV is associated with mutations in HsTRPML1; symptoms include severe psychomotor retardation, retinal degeneration, corneal clouding, achlorhydria, elevated serum gastrin levels, iron deficiency, (lipid) storage bodies in almost every cell type (>50 MLIV causing homozygous mutations or heterozygous combinations of mutations identified) | TRPML2K370Q disrupts the ability of the channel protein to enhance viral infections, raising the possibility of altered susceptibility to certain viral infections in homozygous carriers of this and possibly other TRPML2 polymorphismsref. bib1068 | Deafness, circling behavior, head bobbing and coat color dilution is associated with mutations in MmTRPML3 (Varitint-waddler mutations TRPML3A419P (Va) and TRPML3A419P/I362T (VaJ)) |
| (Disease-associated)GOF mutants | V432P (Hs, Mm) | A425P (Hs); A396P (Mm, isoform 2) | Va (A419P) and VaJ (A419P/I362T) (Hs, Mm) |
| (Disease-associated)LOF mutants | IVS3-2A>G (AJ major), Ex1-7del (AJ minor), T77M, Q79∗, R102∗, L106P, T121M, Y126∗, L137P, C161S, C166F, R172∗, I184T, P203S, T232P, F262S, Q278∗, G293R, F313Δ, R322∗, R340S, D362Y, R403C, F408Δ, R419P, C431Y, Y436C, V446L, L447P, S456L, C463Y, F465L, N469D, small deletions or insertions leading to frame shifts, splicing mutations and complex rearrangements (see also Krogsaeter et al.ref. bib1079 and Fig. 6) | K329∗ (homozygous; Hs) | K390∗ (homozygous; Hs) |
| KO mouse models | KO mice display enlarged vacuoles, psychomotor defects, retinal degeneration, impairments in basal and histamine-stimulated gastric acid secretion,ref. bib1097 impaired myelination and reduced brain ferric iron,ref. bib1098 early-onset muscular dystrophyref. bib1099TRPML1/3 co-deficiency causes accelerated endolysosomal vacuolation of enterocytes and failure-to-thrive from birth to weaningref. bib1100 | KO mice display defects in inflammatory mediator release, in particular CCL2 (MCP-1)ref. bib1081,ref. bib1101 | KO mice display no auditory or vestibular phenotype and no coat color dilutionref. bib1085; Two different KO mouse models (Mcoln3tm1.2Hels and Mcoln3tm1.1Jga) show an increased susceptibility to develop emphysema/COPD and increased MMP12 levels in broncho-alveolar fluid and in the supernatant of cultured alveolar macrophagesref. bib1087TRPML1/3 co-deficiency causes accelerated endolysosomal vacuolation of enterocytes and failure-to-thrive from birth to weaningref. bib1100 |
| Functions | Lysosomal exocytosis; regulates autophagy (TFEB, calcineurin, CaMKKβ/VPS34); role in sorting/transport in late endocytic pathway; regulation of lysosomal lipid and cholesterol trafficking; ROS sensor in lysosomes; endolysosomal cation/heavy metal (iron, zinc) homeostasis; role in gastric acid secretion; regulation of lysosomal motility; plasma membrane repair; phagocytosis; endolysosomal pH regulation?; vesicle fusion, fission?; NAADP receptor? | Osmo-/mechanosensation in RE; EE/RE trafficking; endolysosomal cation homeostasis; vesicle fusion, fission?; endolysosomal pH regulation? | Endocytosis, macropinocytosis (MMPs); regulates autophagy; EE trafficking; endo-lysosomal cation homeostasis; senses lysosome neutralization by pathogens to trigger their expulsion; vesicle fusion, fission? endolysosomal pH regulation? |
| Interacting proteins | ALG2ref. bib1102; TRPML2, TRPML3, TPC1?, TPC2?; LAPTMs; Hsp40; Hsc70 | TRPML1, TRPML3, Hsc70? | TRPML1, TRPML2, GATE16, TPC1?, TPC2?, Hsc70? |
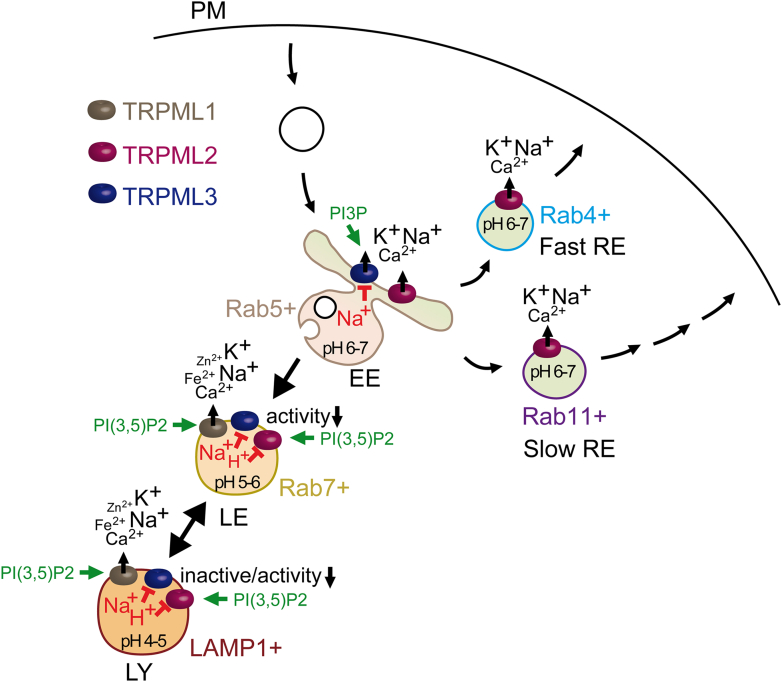
A further feature of TRPML3 is its sensitivity to high luminal Na+ levels (Fig. 7). The lower the Na+ concentration in the lumen of TRPML3-expressing endolysosomes, the higher its activity. This effect seems to be dependent on E361, as mutation E361A has an increased activity in high Na+-containing luminal solution.ref. bib1064 While E361 is located in the luminal loop between TMD3 and 4, amino acids H252, H272, and H283, which are involved in pH sensing and pH-dependent TRPML3 activity regulation, are located in the large luminal loop between TMD1 and 2. The effect of luminal pH on TRPML2 is similar to that of TRPML3. TRPML2 activity, stimulated with either PI(3,5)P2 or the TRPML2 isoform-selective agonist ML2-SA1, increases with increasing (ie, less acidic) luminal pH.ref. bib1081 This is in accordance with TRPML2 being also expressed in less acidic vesicles, in particular Rab11+ and Rab4+ recycling endosomes (Table 9),ref. bib1081 while the expression of TRPML1 is strictly limited to late endosomes/lysosomes. A special feature of TRPML2, in contrast to TRPML1 and TRPML3, is its sensitivity to osmo-/mechano-stimulation.ref. bib1080 The point mutation L314R within the TRPML2 PI(3,5)P2 binding pocket abolishes osmo-/mechanosensitivity and slows down the fast recycling pathway, while activation with ML2-SA1 is preserved. Introducing leucine residues at positions corresponding to L314 in TRPML1 (ie, R322L) or TRPML3 (ie, R309L) was not sufficient to induce osmo-/mechanosensitivity in TRPML1 or TRPML3.ref. bib1080 A role of TRPML2 in fast recycling processes and the secretion of inflammatory mediators, such as CCL2 (MCP-1), has been postulated.ref. bib1081,ref. bib1101
In contrast to TRPML2, for both TRPML1 and TRPML3, roles in autophagy have been demonstrated.ref. bib1075,ref. bib1106,ref. bib1107 Medina et alref. bib1106 showed that TRPML1-mediated Ca2+ signaling regulates autophagy through transcription factor EB (TFEB) and calcineurin. TFEB is a well established master gene for lysosomal biogenesis, driving the expression of autophagy and lysosomal genes.ref. bib1108 TRPML1 controls both TFEB activity and TFEB downstream transcriptional targets.ref. bib1106,ref. bib1109 In addition, Scotto Rosato et alref. bib1109 showed that during starvation, TRPML1 links lysosomal Ca2+ release to autophagosome biogenesis through the activation of the CaMKKβ/VPS34 pathway, a mechanism which is independent of the TFEB transcriptional program and involves activation of the CaMKKβ and the AMP-activated protein kinase AMPK. Furthermore, ROS can induce autophagy via TRPML1. An increase in mitochondrial ROS levels or exogenous oxidants directly stimulates TRPML1-mediated Ca2+ release from lysosomes, thus triggering calcineurin-dependent TFEB nuclear translocation.ref. bib1106,ref. bib1110 Furthermore, a role for TRPML1 in the fusion of autophagic vesicles with lysosomes has been postulated.ref. bib1111,ref. bib1112 On the other hand, Cao et alref. bib1113 claimed that TRPML1 is required for the recovery of enlarged lysosomes and proposed a role for TRPML1 in lysosomal fission. Despite these controversies, the role of TRPML1 in autophagy modulation via multiple pathways is well established.
Less evidence is available for the exact role of TRPML3 in autophagy, but recent results by Kim et alref. bib1075 showed that TRPML3 activation increases autophagy while TRPML3 inhibition suppresses it. Kim et alref. bib1075 identified PI3P as a physiological activator of TRPML3 to release Ca2+ from the phagophore, thus promoting autophagy. The possibility that TRPML3 modulates autophagy independently from TRPML1 gained considerable momentum with these new results.
Besides being an autophagy regulator, regulation of lysosomal exocytosis is another well established role of TRPML1.ref. bib1099,ref. 1115., ref. 1116., ref. 1117. Upon TRPML1 stimulation, lysosomes move to the plasma membrane (visualized, eg, by LAMP1 translocation). Lysosomes can fuse directly with the plasma membrane and release their content into the extracellular space. Increase in local Ca2+ seems to trigger the fusion process,ref. bib1117,ref. bib1118 and the concept that the lysosome releases Ca2+ by itself via TRPML1 for this process has gained much interest.ref. bib1095,ref. bib1099,ref. bib1114,ref. bib1115,ref. 1120., ref. 1121., ref. 1122., ref. 1123., ref. 1124. Lysosomes containing GOF mutants of TRPML1 can also undergo uncontrolled lysosomal exocytosis.ref. bib1124 By contrast, in TRPML1 KO mice, Park et alref. bib1125 found that fusion of lysosomes with secretory organelles leads to uncontrolled exocytosis. How these findings can be reconciled with the accumulating evidence that TRPML1 is a positive regulator of lysosomal exocytosis remains unresolved.
There is also evidence for a role of TRPML3 in lysosomal exocytosis under certain conditions, ie, when the lumen of the lysosome gets neutralized and TRPML3 becomes more active. Miao et alref. bib1105 showed that TRPML3 is required for bacterial expulsion, specifically for uropathogenic Escherichia coli (UPEC) release from infected cells, through lysosomal exocytosis. UPEC, after infecting bladder epithelial cells, are targeted for degradation by the autophagic machinery. However, UPEC escapes degradation by increasing the lysosomal pH. TRPML3 activity is triggered by an increase in pH, and UPEC can be exocytosed; nevertheless, it is also a potentially important protective mechanism in other infectious diseases.
Expression pattern and primary physiological roles of TRPMLs
TRPML1 is ubiquitously expressed. TRPML2 is predominantly found in immune cells, including macrophages, dendritic cells, and B and T cells (for recent reviews see Chao et alref. bib1126 and Spix et alref. bib1127; Table 9). TRPML3 is likewise found to be expressed in immune cells, eg, in alveolar macrophages in the lung (Table 9).ref. bib1086 TRPML3 is also found in skin and inner ear melanocytes, hair cells of the inner ear, olfactory sensory neurons, principal cells of the collecting duct in the kidney, and in endocrine glands and secretory cells, as recently shown by a whole-body analysis of TRPML3 expression in a GFP-reporter mouse model (Table 9).ref. bib1061,ref. bib1128 Thus, TRPML3 was found to be expressed in the adenohypophysis of the pituitary gland, the cortex of the adrenal gland, the parathyroid gland (presumably in chief cells), and testes (presumably in spermatozoa). For comparison with human expression data, please refer to Grimm et alref. bib1128 or the Human Protein Atlas.ref. bib453
Human diseases associated with TRPMLs and mouse models
LOF mutations in TRPML1 lead to progressive neurodegeneration in humans, as discussed above. Mutations also affect the eye and stomach. Patients suffer, eg, from corneal clouding, retinal degeneration, achlorhydria, and iron deficiency resulting from an absence of acid secretion in the stomach. However, TRPML1 is ubiquitously expressed, and endolysosomal accumulation of macromolecules, lipids, heavy metal ions like Zn2+ and Fe2+, and probably others in endolysosomes throughout the body of MLIV patients must be assumed.ref. 1063., ref. 1064., ref. 1065., ref. 1066., ref. 1067., ref. 1068. In addition to brain defects, potential defects due to lack or dysfunction of TRPML1 in the kidney have been proposed.ref. 1130., ref. 1131., ref. 1132., ref. 1133., ref. 1134. Other organs may also be affected, eg, liver, as MLIV patient fibroblasts were shown to accumulate cholesterolref. bib1116 or spleen, where loss of TRPML1 results in defective red blood cell clearance by macrophages.ref. bib1114
MLIV goes along with a strongly reduced quality of life and overall life expectancy. Currently, no curative treatment is available, although treatment of several LOF mutations with small-molecule TRPML1 agonists has been proposed.ref. bib1066 Most promising candidates for such an approach would be patients with mutations in TRPML1 that do not result in strong mislocalization of the protein or mutations that have a loss of PI(3,5)P2 sensitivity, while synthetic small-molecule agonists are still able to activate the channel, with the prerequisite that they must retain some lysosomal localization.
Another proposed strategy is the replacement of the TRPML1 function by enhancing the activity of an alternative Ca2+-permeable cation channel in endolysosomes, eg, 2-pore channel 2 (TPC2; see Prat Castro et alref. bib1134 and Scotto Rosato et alref. bib1116). TRPML1 is permeable for Na+, K+, and Ca2+ but also for Zn2+, Fe2+, and other metal ions. While activation by TPC2 could potentially rescue the functions associated with Na+/Ca2+ permeability, the accumulation of metal ions in the lysosomal lumen, such as Zn2+ or Fe2+, may require additional strategies. While TRPML1 LOF mutations or KO result in severe disease in human and mouse (the murine MLIV phenotype is very similar to the human phenotype),ref. bib1098,ref. bib1135,ref. bib1136 no disease-causing LOF or KO phenotypes for TRPML2 or TRPML3 are known in humans. Apparently, homozygous mutations in TRPML2 and TRPML3, resulting in an early stop (TRPML2K329∗ and TRPML3R390∗) in humans, are not lethal (Table 9).ref. bib1074 Early stop mutations or complete loss of TRPML1 in humans are likewise not lethal, but result in severe phenotypes. Whether homozygous carriers of TRPML2K329∗ and TRPML3,R390∗ identified in East and South Asia, are severely ill or have other health disadvantages, is not known. Several heterozygous carriers were also identified in North Borneo (Dusun people).
A homozygous mutation in TRPML2, common in certain African populations, is TRPML2K370Q. TRPML2K370Q reportedly disrupts the ability of the channel protein to enhance viral infections, raising the possibility of an altered susceptibility to certain viral infections in homozygous carriers of this and possibly other TRPML2 polymorphisms.ref. bib1068
Disease-causing mutations for TRPML3 have been described only in mice. The GOF mutations, A419P (Va) and A419/P/I362T (VaJ), cause the Varitint-waddler phenotype characterized by circling behavior, deafness, and coat color dilution.ref. 1070., ref. 1071., ref. 1072., ref. 1073., ref. 1074.,ref. bib1082,ref. bib1083 Introducing A419P or A419P/I362T mutations into WT human TRPML3 likewise result in GOF effects.ref. bib1069 The Varitint-waddler phenotype can be rescued by overexpression of plasma membrane ATPase, suggesting that cytosolic Ca2+ overload due to the TRPML3 GOF mutation A419P is causative for the observed effects.ref. bib1073 The additional mutation of I362T in VaJ results in a slightly less severe phenotype, which nevertheless shows similar Ca2+ overload as well as circling behavior, deafness, and coat color dilution.ref. bib1069,ref. bib1082 The reason for VaJ being milder remains unclear. Of note, however, I362T is located next to E361, which was reported to affect the Na+ sensitivity of TRPML3, increasing channel activity.ref. bib1064
Besides diseases associated directly with mutations in TRPMLs, KO mouse models and other functional studies have revealed additional potential roles of TRPMLs in physiology and pathophysiology, from immune cell function and cancer to gastrointestinal, kidney, cardiovascular, neurodegenerative, lung, and infectious diseases.
TRPML1 activity is strongly reduced with increasing pH; hence, a backup channel getting engaged when TRPML1 is blocked, eg, as in the case of bladder epithelial cell infection with UPEC, which neutralize the lysosomal lumen to evade degradation, seems an elegant solution.ref. bib1105 TRPML3 can fulfill that function as it gets activated when the pH in the lysosomal lumen increases, while under normal lysosomal pH conditions, it would be largely silent. Other indications for roles of TRPMLs in infectious diseases relate to TRPML1, but increasingly also to TRPML2 and TRPML3. Thus, TRPML1 was shown to play a role in Helicobacter pylori infection,ref. bib1137 where virulence factor vacuolating cytotoxin A promotes more severe disease development and gastric colonization. Virulence factor vacuolating cytotoxin A targets TRPML1 to disrupt endolysosomal trafficking and autophagy, an effect that could be reversed by direct activation of TRPML1 with a small-molecule agonist, leading to the clearance of intracellular bacteria. Furthermore, Khan et alref. bib1138 reported on the role of TRPML1 in cooperation with the big-conductance Ca2+ activated K+ channel (BK) in HIV infection and proposed that TRPML1/BK coactivation leads to an enhanced acidification of endolysosomes, resulting in an increased degradation of Tat protein, which facilitates HIV replication. TRPML2 was postulated to enhance viral infections, eg, yellow fever virus, influenza A virus, and equine arteritis virus infections, and the human TRPML2 variant, TRPML2K370Q, discussed before, reportedly shows a LOF phenotype with respect to viral enhancement.ref. bib1068 Quite to the contrary, Gibbs et alref. bib1139 found that TRPML2 acts also as an inward rectifying Mg2+ channel on endolysosomes and can thus deprive Salmonella Typhi of Mg2+, restricting growth.
Xu et al. recently showed that suppressing either TRPML3 or BK helps bacterial survival, whereas increasing either TRPML3 or BK favors bacterial clearance.ref. bib1140
Hence, in sum, it is currently claimed that activation of TRPML1 and TRPML3 would be beneficial in treating certain infectious diseases, while in the case of TRPML2, it remains unclear whether activation or inhibition may be more beneficial. This may, of course, also depend on the type of infection.
Generally, all 3 TRPMLs are expressed in a range of immune cells, including different types of macrophages, natural killer cells, dendritic cells, B and T cells, microglia, and astrocytes (Table 9).ref. bib1126,ref. bib1127,ref. 1142., ref. 1143., ref. 1144. In the mouse lung, TRPML3 is almost exclusively expressed in alveolar macrophagesref. bib1087 and in 2 independent KO mouse models (Mcoln3tm1.2Hels and Mcoln3tm1.1Jga), an increased susceptibility to develop an emphysema-like phenotype was found. KO mice showed differences in lung function and histological parameters such as elastance and compliance, or the mean linear chord length (mean free distance in the air spaces), pointing to an emphysema-like phenotype compared with WT mice, which was further and more strongly exacerbated in KO mice compared with WT mice after elastase or tobacco smoke treatment. In broncho-alveolar fluid and the supernatant of cultured alveolar macrophages, increased levels of matrix metalloproteinase 12 (macrophage elastase) were detected, a known risk factor for emphysema and COPD development.ref. bib1087,ref. bib1144 The authors further found that the relative TRPML3 expression was increased in samples from human smokers with COPD compared with healthy smokers. TRPML3 expression was also increased in smokers compared with nonsmokers. This was interpreted as being a potential compensatory mechanism to increase the uptake of excess matrix metalloproteinase 12 and potentially other MMPs with the help of TRPML3.
TRPML1, in addition, was shown to play an important role in the gastrointestinal tract. Thus, Sahoo et alref. bib1067 found that TRPML1 overexpression or activation in mouse parietal cells induced gastric acid secretion, while TRPML1 inhibitors blocked it. This is in accordance with human MLIV patients who are reportedly achlorhydric. Mechanistically, TRPML1 was found to play a role in gastric acid secretion in parietal cells by regulating the trafficking and exocytosis of H+/K+-ATPase-rich tubulovesicles after histamine stimulation.ref. bib1067 Chandra et alref. bib1097 found that Trpml1 KO mice have significant impairments in basal and histamine-stimulated gastric acid secretion.
There are also reports of progressive renal failure in MLIV patients, and blockade of TRPML1 was found to suppress the interaction of lysosomes and multivesicular bodies, leading to increased exosome release from mouse podocytes.ref. bib1145 In addition, Nakamura et alref. bib1129 suggested a role of lipidated LC3 interacting with TRPML1 to release lysosomal Ca2+ essential for TFEB activation during kidney injury and lysosomal damage response.ref. bib1132
In 2 recent works published by the same group,ref. bib1146,ref. bib1147 it is claimed that inhibition of TRPML1 has a protective role in myocardial ischemia/reperfusion injury. Mechanistically, this was attributed to a restoration of impaired cardiomyocyte autophagy by blocking TRPML1, which gets activated by ROS elevation, following myocardial ischemia/reperfusion injury. Activated TRPML1, releasing lysosomal Zn2+, reportedly blocks autophagic flux in cardiomyocytes by disrupting the fusion between autophagosomes and lysosomes. This is a surprising finding, as TRPML1, according to the vast majority of publications, is believed to promote autophagy rather than inhibit it. Thus, a large body of evidence suggests that TRPML1 promotes autophagy through activation of TFEB, mediated by lysosomal Ca2+ release.ref. bib1106,ref. bib1107,ref. bib1110,ref. bib1111,ref. bib1148,ref. bib1149 TRPML1 is also much less permeable to Zn2+ compared with Ca2+, hence a dominant effect of Ca2+ on autophagy would be expected.
Several lines of evidence point to a role of TRPMLs in different types of cancer, eg, breast cancer, melanoma, or gliomaref. bib1094,ref. 1151., ref. 1152., ref. 1153., ref. 1154., ref. 1155., ref. 1156., ref. 1157., ref. 1158. (for recent reviews, see ref. bib1123,ref. bib1152,ref. 1159., ref. 1160., ref. 1161.). The loss or inhibition of TRPMLs reduces, eg, cancer cell migration and invasion, and roles of TRPMLs in TFEB-mediated gene transcription and lysosomal exocytosis promoting invasiveness and drug resistance in cancer cells, cancer cell nutrient sensing, and antitumor immunity have been proposed. Collectively, the data suggest that TRPMLs, in particular TRPML1, stimulate oncogenesis by enhancing survival, growth, invasiveness, and mitochondrial activity of cancer cells.ref. bib1159
Two recent papersref. bib1161,ref. bib1162 are challenging this view. Xing et alref. bib1162 claim that TRPML1 activation inhibits autophagy (similar worksref. bib1146,ref. bib1163 discussed above) and that this autophagy inhibition suppresses cancer (melanoma) metastasis. Similarly, Du et alref. bib1161 suggest that TRPML1 small molecule activation induces Zn2+ release mediated cell death in metastatic melanoma, emphasizing that instead of inhibition, activation of TRPML1 may be beneficial in treating metastasis formation in cancer, at least in melanoma.
Due to the neurodegenerative phenotype in MLIV disease and several studies showing TRPML1 activation to rescue lysosomal storage and neurodegenerative disease phenotypes, TRPML1 appears to be a promising novel drug target for the treatment of such diseases. We will, therefore, in the following chapter, focus on this topic and discuss it in more detail after a brief discussion of currently available pharmacological tools to modulate TRPMLs.
Pharmacology of TRPMLs
It was already mentioned that the currently known endogenous activators of TRPMLs are the phosphoinositides PI(3,5)P2 (agonist for all 3 TRPMLs) and PI3P (agonistref. bib1075 for TRPML3), while PIP2 inhibits TRPMLs (Table 9). How about lipophilic small molecule modulators of TRPMLs? In addition to Table 9 presented here, a comprehensive and detailed overview of the currently available pharmacology for TRPMLs has been published recently by Rautenberg et alref. bib1091 Of note, isoform-selective activators for all 3 TRPMLs have become available in recently: ML1-SA1 (EVP-169)ref. bib1087 for human/mouse TRPML1, ML2-SA1 (EVP-22)ref. bib1081 for human/mouse TRPML2, EVP-21 for human TRPML3, and ML3-SA1 (EVP-77)ref. bib1087,ref. bib1164 for mouse TRPML3. ML1-SA1 is structurally related to ML-SA1 published previouslyref. bib1065 (Fig. 8). ML2-SA1 is a derivative of the previously publishedref. bib1086 structure SN-2; likewise, ML3-SA1 (EVP-77) and EVP-21 are derived from SN-2 (Fig. 8). In contrast to these isoform-selective agonists, ML-SA1 and MK6-83 are not isoform-selective TRPML channel agonists.ref. bib1066,ref. bib1067
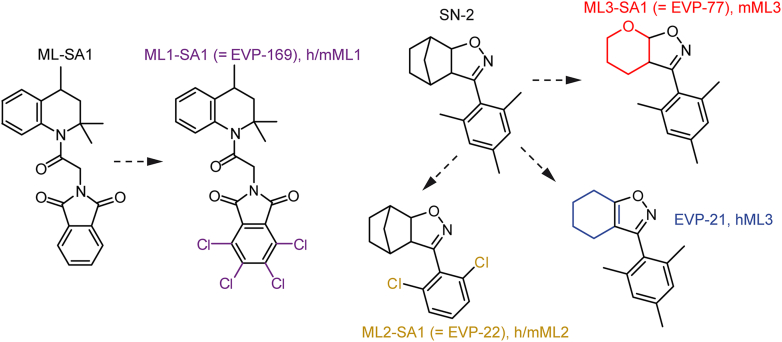
Regarding inhibitors of TRPMLs, there are currently only isoform-selective antagonists available for TRPML1: EDME (17β-estradiol methyl ether)ref. bib1094 and its derivatives PRU-10 and PRU-12, the latter 2 showing reduced efficacy at the estrogen receptor α compared with EDME.ref. bib1094 Other available inhibitorsref. bib1165 are ML-SI1 and ML-SI3. From the original publication, it remains, however, unclear which of the stereoisomers of ML-SI1 and ML-SI3 are functionally active. ML-SI1 has 4 different stereoisomers, and currently, only racemic mixtures are commercially available.ref. bib1092 One commercial product sold as a TRPML1 inhibitor is GW405833. This compound differs from the published structureref. bib1165 as it is not based on an indoline moiety (for details see Rautenberg et alref. bib1091). Importantly,ref. bib1092,ref. bib1164 this compound is inactive on TRPML1. Nevertheless, GW405833 was used at least in 2 publications erroneously as a TRPML1 inhibitor.ref. bib1122,ref. bib1166 In addition, GW405833 is also sold as a selective cannabinoid CB2 receptor agonist. Of note, ML-SI1 in the structure as published by Wang et al,ref. bib1165 blocks both TRPML1 and TRPML2 with equal potency.ref. bib1092,ref. bib1164
Commercially available ML-SI3 is a racemic mixture of trans-enantiomers.ref. bib1092 Separation of the trans-enantiomers of ML-SI3 revealed that the (–)-enantiomer is an inhibitor of all 3 TRPMLs, while the (+)-enantiomer inhibits TRPML1, but activates TRPML2 and TRPML3 (Table 9).ref. bib1092 The commercially available racemic mixture also activates TRPML2 but blocks TRPML1 and TRPML3.ref. bib1092
TRPMLs as therapeutic targets
That loss of TRPML1 leads to severe neurodegeneration (MLIV) is undisputable. But does activation of TRPML1 ameliorate lysosomal storage and neurodegenerative disease phenotypes? In 2012, Shen et alref. bib1065 claimed that abnormal lipid accumulation (cholesterol, sphingolipids, sphingomyelin) in Niemann-Pick type C1 patient cells can be reversed by TRPML1 activation. It was also shownref. bib1065,ref. bib1134 that sphingomyelin can directly block TRPML1. FIG4 (encoding Sac3 protein) deficiency, which causes a rare peripheral neuropathy with severe motor deficits called Charcot-Marie-Tooth type 4J, results in decreased levels of both PI5P and the endogenous agonist of TRPML1, PI(3,5)P2. Zou et alref. bib1167 showed that TRPML1 activation rescues the abnormal lysosomal storage in FIG4-deficient cells and in ex vivo nervous tissue. Rescue effects of TRPML1 activation were recently confirmed for 2 subtypes of demyelinating Charcot-Marie-Tooth disease in an independent study.ref. bib1168 Amelioration of lysosomal storage in Niemann-Pick type A and Fabry disease, as well as Niemann-Pick type C1 disease, was shown to depend on the BK that forms a physical and functional coupling with TRPML1. Importantly, Ca2+ release via TRPML1 activates BK, which in turn facilitates further Ca2+ release, enhancing membrane trafficking and lysosomal exocytosis.ref. bib1169,ref. bib1170
Tsunemi et alref. bib1121 found that increased lysosomal exocytosis by TRPML1 activation protects human inducible pluripotent stem cell (iPSC)-derived dopaminergic neurons in a model of familial PD from α−synuclein toxicity, caused by mutations in ATP13A2 (CLN12). Another link between PD and TRPML1 has recently been established by Sasazawa et alref. bib1171 reporting that acrolein, an aldehyde that is significantly elevated in PD patient serum, enhances autophagy via a newly discovered JIP4-TRPML1-ALG2 pathway.
In APP/PS1 double transgenic mice and hippocampal neurons with AD-like alterations, Zhang et alref. bib1172 found that overexpression of TRPML1 played a neuroprotective role in AD by ameliorating cognitive function and attenuating cognitive impairments. Xu et al.ref. bib1122 reported that TFEB regulates, in a TRPML1-dependent manner, the lysosomal exocytosis of tau and that TFEB loss exacerbates tau pathology and spreading. In another recent study, Somogyi et alref. bib1173 show that dysfunction of TRPML1 is associated with abnormalities in the endolysosomal system in AD and APOE-ε4 iPSC-derived neurons. Inhibition of PIKfyve, the key enzyme in the production of the TRPML1 agonist PI(3,5)P2, recapitulated these results, while effects could be reverted or reduced by the TRPML agonist ML-SA1.
Other examples for TRPML1 activation providing potential therapeutic benefit in neurodegenerative or lysosomal storage diseases are HIV gp120-related lysosomal storage, where TRPML1 activation cleared amyloid β (Aβ) from lysosomal compartments in neuronsref. bib1174, and data suggesting TRPML1 activation to promote autophagy, facilitating the clearance of accumulated α-synuclein in both in vitro and in vivo models of MPP+/MPTP-induced Parkinson’s disease.ref. bib1175
In contrast to TRPML1, TRPML2 expression is largely absent from the human brain, while TRPML3 appears to be expressed to some extent in the hippocampus, cerebral cortex, and hypothalamus.ref. bib1079 Importantly, TRPML3 activity increases with increasing pH, suggesting that even under conditions of increased lysosomal pH, as often observed in lysosomal storage and neurodegenerative diseases, TRPML3 would still be active to drive lysosomal exocytosis similar to TPC2, the activity of which does likewise not depend on the luminal pH.ref. bib1105,ref. bib1116,ref. bib1134
Currently, it is unclear how much lysosomal Ca2+ release would be beneficial and how it can be finely controlled so that it is, on the one hand, sufficient enough to promote lysosomal exocytosis and autophagy, while on the other hand avoiding potential cytotoxicity due to cytosolic Ca2+ overload. The possibility of a hyperactive TRPML1 under certain disease conditionsref. bib1176,ref. bib1177 and the controversies around this possibility have been discussed recently.ref. bib1079,ref. bib1134 In addition, Zn2+, Fe2+, and other heavy metal ions, which may be released alongside Ca2+ from lysosomes after TRPML1 activation, may pose a risk for increased cytotoxicity.ref. bib1178
In sum, despite compelling and increasing evidence for a beneficial effect of TRPML1 activation in clearing lysosomal storage and promoting lysosomal exocytosis and autophagy, more empirical evidence is needed, as well as safety and chronic dosing studies. It also remains unclear if a defect in lysosomal acidification can be corrected by TRPML1 activation, although some evidence suggests this.ref. bib1138,ref. bib1174
TRPP channels
Introduction
TRP polycystin (TRPP) channels are Ca2+-permeable nonselective cation channels with conserved roles in biological processes such as tubular morphogenesis and left-right patterning of organs in vertebrates.ref. bib1179,ref. bib1180 TRPP channels are regarded as the most ancient subfamily of TRP channels, with orthologs in organisms ranging from yeast to mammals.ref. bib6,ref. bib1181 The founding member of the TRPP channels, TRPP2, was discovered as the PKD2 gene product mutated in ADPKD.ref. bib1182 TRPP channels form homotetrameric complexes and heterotetrameric protein assemblies with other TRP channels and polycystin-1 (PC1) family members. The physiological importance of the heteromeric PC1-TRPP2 receptor-channel complex is underscored by the fact that mutations in the PKD1 gene, encoding PC1, also cause polycystic kidney disease.ref. bib1183 Since the discovery of the genes encoding the founding members of the polycystins, PKD1 and PKD2, 6 additional family members have been identifiedref. 1185., ref. 1186., ref. 1187., ref. 1188., ref. 1189., ref. 1190. based on sequence and structural homology: PKD1L1, PKD1L2, PKD1L3, PKDREJ, PKD2L1, and PKD2L2. The PKD2-like genes encode TRPP channels, whereas the PKD1-like genes encode PC1 family proteins that assemble with TRPP channels in a modular fashion. TRPP channels are found in various tissues and regulate calcium signaling in primary cilia and other cellular compartments. Recent advances in determining the structure and function of TRPP channels in homo- and heterotetrameric complexes have provided first insights into the structural basis for channel gating and ion permeation. Pharmacological modulators of TRPP channels are still scarce.
Domain topology, assembly, and functional characteristics of individual TRPP channels
The nomenclature of TRPP ion channels is ambiguous. Initial publications named the gene products of PKD1 and PKD2 PC1 and polycystin-2. The founding member of the TRPP subfamily, polycystin-2 (PC2), was later classified as TRPP2, and this designation is commonly used in the literature. The latter designation of TRPP2 as TRPP1 has not been widely adopted and has caused confusion, as it has been used for both PC1 and polycystin-2. We therefore advocate for the following TRP nomenclature: 1) only bona fide ion channels with 6 TMDs should be named TRPP channels, and 2) PC1-like proteins with 11 TMDs should be referred to with their gene names until a unified protein nomenclature exists (Table 1). Because of ambiguity, TRPP1 and TRPP4 should not be used.
Domain topology and assembly
Like all TRP channels, TRPP family members have 6 TMDs (S1–S6) and intracellular amino- and carboxy-termini (Fig. 9A). The TM segments S1–S4 form a voltage-sensor domain and the segments S5-S6 constitute the pore domain. A characteristic feature of TRPP ion channels is the large extracellular loop between S1 and S2, consisting of more than 200 amino acids in TRPP2, TRPP3, and TRPP5, respectively. This extracellular loop of TRPP2 contains 5 conserved asparagine-linked glycosylation sites (N299, N305, N328, N362, and N375), which are required for efficient TRPP2 biogenesis and stability.ref. bib1190 The carboxy-terminal region of TRPP2 comprises motifs involved in channel regulation, assembly, and trafficking, including a Ca2+-binding EF hand, a CC domain, and an ER retention motif with an acidic amino acid cluster (Fig. 9A).ref. 1192., ref. 1193., ref. 1194., ref. 1195., ref. 1196., ref. 1197., ref. 1198., ref. 1199. The EF hand has been implicated in Ca2+-dependent regulation of TRPP2.ref. bib1197,ref. bib1198 A more recent study, however, questions the hypothesis that Ca2+ occupancy of the TRPP2 EF hand is responsible for the regulation of channel activity.ref. bib1199 The acidic cluster is involved in protein trafficking, whereas the 2 CC domains contribute to homo- and heteromerization of TRPP2 subunits.ref. 1193., ref. 1194., ref. 1195.,ref. bib1196,ref. bib1198
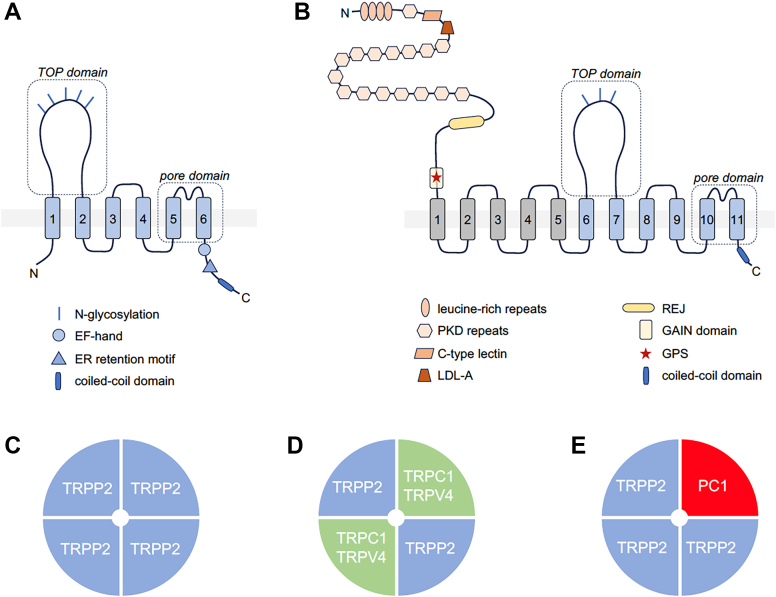
TRPP2 contains several predicted and validated phosphorylation sites, some of which have been studied in more detail (S76, S801, S812, and S829).ref. bib1191,ref. bib1196,ref. 1201., ref. 1202., ref. 1203., ref. 1204., ref. 1205., ref. 1206., ref. 1207. Glycogen synthase kinase 3 (GSK3) was shown to phosphorylate serine 76 to promote redistribution of TRPP2 from the plasma membrane to intracellular compartments.ref. bib1203 Phosphorylation of serine 801 is increased by epidermal growth factor stimulation and appears to be protein kinase D-dependent.ref. bib1204 Phosphorylation of serine 812 by PKC K2 contributes to regulation of TRPP2 trafficking and ion channel activity.ref. bib1196,ref. bib1200,ref. bib1206 Serine 829 phosphorylation by aurora A or protein kinase A has been reported to modulate ion channel function.ref. bib1202,ref. bib1205 It should be noted, however, that the functional importance of these TRPP2 modifications has been difficult to evaluate as overexpressed TRPP2 is trapped in the ER, impeding the electrophysiological analysis at the plasma membrane (see below). The overall domain topology of TRPP3 and TRPP5 resembles TRPP2. While motifs such as C-terminal CC domains and phosphorylation sites have also been predicted in these channels, there is still much less information compared with TRPP2.ref. bib1207,ref. bib1208 TRPP3 channel function has been shown to be regulated by palmitoylation and phosphorylation at the cytoplasmic N-terminal domain (at cysteine 39 and threonine 39, respectively).ref. bib1209 Furthermore,ref. bib1210 TRPP3 was found to be regulated by cAMP signaling via a cluster of phosphorylation sites at S682, S685, and S686.
Homotetrameric TRPP channel complexes
Recently, the first 3D structures of TRPP2 and TRPP3 have been resolved using single-particle cryo-EM.ref. 1212., ref. 1213., ref. 1214., ref. 1215., ref. 1216. These studies show that TRPP channels are assembled as homotetrameric complexes with each subunit comprising a voltage-sensor domain (S1–S4), a tetragonal opening for polycystins (TOP) domain, formed by the extracellular loop between S1 and S2, and a pore domain (S5–S6), jointly formed by the 4 subunits. The individual channel subunits interface through their TOP and pore domains, suggesting a role of these subunit interactions in homotetrameric assembly. The structural arrangement of the voltage-sensor and pore domains in TRPP2 and TRPP3 provides mechanistic insights into voltage-dependent gating.
The voltage-sensor domains of the respective TRPP subunits connect to the pore domains of neighboring subunits via an S4-S5 linker helix. This S4–S5 linker helix is thought to communicate the activation state of the voltage-sensor domain to control gating of the pore domain. The S4 segment of TRPP2 and TRPP3 contains 2 positive gating charges, which are thought to move outward in response to membrane depolarization.ref. bib1212,ref. bib1216 This outward movement may be coupled to the opening of the pore via lateral displacement.
The TOP domain extends from the S1 and S2 helices on the extracellular side of the voltage-sensor domain. This domain is not found in TRPCs, but is similar to a corresponding domain of TRPMLs.ref. bib1054,ref. bib1059 The TOP domain is composed of 5 β strands and 2 α-helices and forms extracellular contacts with the extracellular loop between S3 and S4, suggesting a functional connection to the voltage-sensor domain. In support of this notion, ADPKD-causing missense mutations in this domain can significantly shift the voltage dependence of TRPP2 opening.ref. bib1217
The pore domain constitutes the ion-conductive pathway and the selectivity filter. TRPP2 and TRPP3 are Ca2+-permeable nonselective cation channels. TRPP3 is more selective for Ca2+ than TRPP2, probably because the selectivity filter of TRPP3 harbors a second ring of negatively charged aspartate residues that is not present in TRPP2.ref. bib1218 Structural and functional studies suggest that TRPP channels have multiple gates, with the lower gate in the S6 segment being mobilized by uncoiling its secondary helical structure.ref. bib1213 The upper gate is thought to be within the selectivity filter and might be involved in channel inactivation.ref. 1214., ref. 1215., ref. 1216. Asparagine 533 in the outer pore loop of mouse TRPP3 was shown to be essential for its voltage-dependent inactivation.ref. bib1219
The carboxy-terminal domains have not been structurally resolved in the reported TRPP2 and TRPP3 structures. However, the structures of isolated fragments containing the EF hand or the CC domain have been determined.ref. bib1198,ref. 1221., ref. 1222., ref. 1223. The isolated CC domain of TRPP2 forms trimers, which appears to contradict the homotetrameric assembly of whole TRPP channels.ref. bib1223 Yet, the trimeric assembly of the isolated CC domains may be explained by the fact that heteromultimeric assemblies of TRPP channels with PC1 family proteins occur at a 3:1 stoichiometry (see below). While the precise role of the coiled-coil motif for TRPP channel assembly and structure is not known, it appears to be important for channel function, since truncating mutations that delete the CC motif of TRPP2 cause ADPKD (The ADPKD Mutation Database, https://pkdb.mayo.edu/variants).
Heteromeric TRPP channel complexes
TRPP2 has been shown to interact with several TRP channels (TRPC1, TRPC3, TRPC4, TRPC5, TRPC7, and TRPV4) in heterologous expression systems.ref. bib1224 There are currently no 3D structures of these heteromeric assemblies, but atomic force studies proposed a 2:2 stoichiometry with an alternating subunit arrangement for TRPP2/TRPC1 and TRPP2/TRPV4 heterotetramers, respectively.ref. bib57,ref. bib128,ref. bib385,ref. bib1225 It has been proposed that the channel properties of these heteromeric TRPP2 complexes are modulated by the TRP subunit composition, adapting the functional properties of TRPP2 to tissue-specific roles, including mechano- and thermosensation.ref. bib1225 However, there is still limited information concerning the physiological role of most of these heteromeric TRPP2 complexes in vivo.
Heteromeric complexes formed by TRPP channels with members of the PC1 family have been shown to play an essential role in biological processes such as tubular morphogenesis and establishment of left-right asymmetry.ref. bib1179 Mutations in the genes encoding PC1 and TRPP2 cause polycystic kidney disease in humans and model organisms (see “Expression pattern and primary physiological roles of TRPP channels” and “Human diseases associated with TRPP channels”). Both proteins interact to form a receptor-ion channel complex.ref. bib1226,ref. bib1227 PC1 and related family members (PKD1L1, PKD1L2, PKD1L3, and PKDREJ) are rather large proteins (210–520 kDa) with 11 TMDs (S1–S11). The 6 carboxy-terminal TMDs of PC1 (S6–S11) share high sequence homology with TRPP2. Despite this homology, PC1 is not an ion channel itself, but may contribute to the pore domain of heteromeric complexes with TRPP2. Members of the PC1 family have a large extracellular N-terminal domain, which is thought to be involved in the sensing of mechanical or chemical cues.ref. 1229., ref. 1230., ref. 1231., ref. 1232. This domain contains multiple motifs suggesting interaction with cell matrix or extracellular proteins (Fig. 9B).ref. bib1232,ref. bib1233 PC1 has the largest extracellular domain with 3074 amino acids, followed by PKD1L1 (1784 amino acids), PKD1L2 (1344 amino acids), PKD1L3 (1083 amino acids), and PKD1REJ (1184 amino acids). The PC1 N-terminal domain contains multiple motifs, including leucine-rich repeats, 15 PKD repeats, an low-density lipoprotein A–related motif, a C-type lectin domain, and a receptor for egg jelly module (Fig. 9B). Interestingly, PC1 shows similarities to the adhesion class GPCRs (adhesion GPCRs). A common feature of PC1 and adhesion GPCRs is a GPCR autoproteolysis-inducing domain and autoproteolytic cleavage of the extracellular amino-terminus at a G protein-receptor-coupled proteolytic site.ref. 1235., ref. 1236., ref. 1237. Activation of adhesion GPCRs through a tethered agonist has been proposed to involve a stalk region preceding the first TMD.ref. bib1237 Recent studiesref. bib1238,ref. bib1239 suggest that a similar mechanism may apply for the activation of PC1.
The 3D structures of the heteromeric PC1-TRPP2 complex and the PKD1L3-TRPP3 complex were determined using cryo-EM, revealing a 1:3 stoichiometry, which had already been proposed in earlier studies.ref. 1241., ref. 1242., ref. 1243.
Owing to the sequence homology to TRPP2, the S6–S11 TMDs of PC1 are arranged with similar symmetry to TRPP2 subunits within the heteromeric structure.ref. bib1211,ref. bib1213,ref. bib1215,ref. bib1241 The same holds true for the highly homologous TOP domains of PC1.ref. bib1241 In PC1 and PKD1L3, the TOP domains extend from the extracellular S6–S7 loop (Fig. 9B). In contrast to homomeric TRPP2 and TRPP3 channels which have symmetric channel selectivity filters, the pore domain of heteromeric PC1-TRPP2 channels is asymmetric due to the contribution of the S10 and S11 segments of PC1-related subunits.ref. bib1240,ref. bib1241 Based on the structure of the pore domain, the cation selectivity of these heteromeric channels is predicted to be distinct from homomeric TRPP2 channels, because the PC1 pore loop lacks the aspartate residues found in TRPP2. This prediction is supported by electrophysiological experiments (see below). In the published structure of the PC1-TRPP2 complex, 3 positively charged residues in the pore lining S11 of PC1 (R4100, R4107, and H4111) plug the ion permeation pathway. It has been speculated that lateral displacement S11 of PC1, possibly coupled to conformational changes in distant parts of the complex, may gate the PC1-TRPP2 heteromeric channel.ref. bib1180 Future studies of the structure of the heteromeric PC1-TRPP2 complex in the open state, ideally with a bound activating ligand, are required to unravel its gating mechanism.
The structure of the PKD1L3-TRPP3 complex was determined in a closed and in a Ca2+-bound open state.ref. bib1240 Two Ca2+-binding sites that are probably involved in gating the channel complex were identified. In the closed state, the PKD1L3-TRPP3 complex is blocked by K2069 from PKD1L3, which appears to plug the ion permeation pathway in the absence of Ca2+. At high Ca2+ concentrations, K2069 of PKD1L3 is displaced by the Ca2+ ion coordinated by the D523 side chain of TRPP3 and main chain carbonyls of both TRPP3 and PKD1L3. The second Ca2+ binding site is in the extracellular cleft of the voltage sensor domain within the third TRPP3 subunit of the heteromeric complex. Electrophysiological experiments support the hypothesis that Ca2+ binding of the voltage sensor domain of TRPP3 is responsible for Ca2+-dependent activation.ref. bib1240 In summary, the structures of the heteromeric TRPP channel complexes have provided mechanistic insights into ion permeation and gating. It should be noted that all structures of the heteromeric TRPP complexes have been determined using truncated forms of the PC1-related subunits. In the heteromeric PC1-TRPP2 structure, PC1 was missing the extracellular N-terminal domain and the intracellular C-terminus. In the PKD1L3-TRPP3 structure, PKD1L3 was missing its N-terminal extracellular domain and the first 5 TMDs (S1–S5).ref. bib1240,ref. bib1241
Functional characteristics of individual TRPP channels
TRPP channels are Ca2+-permeable nonselective cation channels. Their biophysical properties are modulated by differential assemblies with members of the PC1 family. Here, we summarize the functional properties of individual homotetrameric and heteromeric TRPP channel complexes.
TRPP2
TRPP2 function has been studied in the plasma membrane, in the ER, and in primary cilia. The functional analysis of TRPP2 in the plasma membrane has proven difficult because heterologously expressed TRPP2 in mammalian cell lines localizes mostly, if not exclusively, in the ER.ref. bib1191,ref. bib1196,ref. bib1197,ref. bib1243 Despite earlier studies reporting TRPP2 currents after heterologous expression in different cell types,ref. bib1244 many later studies failed to record increased whole cell currents after overexpression of WT TRPP2 (with or without coexpression of PC1).ref. bib1196,ref. bib1213 Earlier functional studies of TRPP2 are reviewed elsewhere.ref. bib1244 In the ER, TRPP2 operates as a Ca2+ release channel, and different mechanisms have been proposed on how this may affect Ca2+ signaling and ER Ca2+ homeostasis. One study showed that TRPP2-mediated Ca2+ release decreases the ER concentration, thereby regulating the sensitivity of cells to apoptotic stimuli.ref. bib1245 Another study proposed that TRPP2 amplifies ER Ca2+ release via Ca2+-dependent activation of TRPP2,ref. bib1197 whereas others reported increased Ca2+ release from the ER through direct association with the inositol trisphosphate receptor.ref. bib1246 Reconstitution of TRPP2 proteins isolated from the ER was used to record single-channel currents and Ca2+ regulation of the channel.ref. bib1197,ref. bib1206
Recent progress in the electrophysiological characterization of TRPP2 channels has been achieved through 2 methodological breakthroughs: (1) direct electrophysiological recordings from primary cilia,ref. bib1247,ref. bib1248 and (2) GOF mutations in TRPP2 enabling the electrophysiological characterization in the plasma membrane.ref. bib1249
TRPP2 localizes to the membrane of primary cilia.ref. bib1250 Patch-clamp recordings from cilia showed that endogenous and heterologous TRPP2 channels have a cation permeability profile of K+ > Na+ >> Ca2+ with a single channel conductance of 139 pS (in the presence of K+). TRPP2 has a 10-fold higher permeability for Na+ than for Ca2+ ions.ref. bib1248,ref. bib1251,ref. bib1252 Despite the relatively low Ca2+ selectivity, opening of TRPP2 channels can trigger Ca2+ signals in cilia and other cellular compartments due to the huge Ca2+ concentration gradient with an extracellular concentration that is 10,000 times higher than the intracellular Ca2+ concentration. Together with the negative membrane potential, this provides a big electrochemical driving force for Ca2+ to enter cells. TRPP2 is voltage-dependent with an outwardly rectifying current-voltage relationship. This voltage dependence is modulated through the intracellular Ca2+ concentration.ref. bib1199,ref. bib1248 Furthermore, TRPP2 whole cell cation currents at the plasma membrane could be recorded in Xenopus oocytes over-expressing a TRPP2 F604P GOF mutant, which has enabled functional studies of TRPP2 at the plasma membraneref. bib1249 and studies of disease-associated missense mutations in the pore loop of TRPP2 that alter its channel function.ref. bib1253
In addition to homomeric complexes, TRPP2 forms heteromeric complexes with members of the PC1 family, which modulate its functional properties. The PC1-TRPP2 channel complex has been studied the most because of its involvement in ADPKD (see “Expression pattern and primary physiological roles of TRPP channels”). Despite intense research efforts, many functional features of this channel complex remain poorly understood. This can be explained by the fact that heterologous expression of PC1 together with TRPP2 does not give rise to constitutively active channels in the plasma membrane.ref. bib1252,ref. bib1254 Initial studies reporting increased whole cell currents upon co-expression of PC1 and TRPP2 in the plasma membraneref. bib1195,ref. bib1255 could not be reproduced by others.ref. bib1252,ref. bib1254
Two recent studies have provided insights into the channel function of the PC1-TRPP2 complex. In the first study in Xenopus oocytes, co-expression of PC1 with TRPP2 harboring 2 GOF mutations (L677A/N681A) resulted in altered ion selectivity, with greater Ca2+ permeability compared with the TRPP2 mutant channels alone, suggesting a contribution of PC1 to the selectivity filter.ref. bib1256 In a second study, the TRPP2 F604P GOF mutant was coexpressed with PC1 containing a strong N-terminal signal peptide to increase plasma membrane trafficking.ref. bib1254 Kidney epithelial cells coexpressing these constructs showed constitutive outwardly rectifying ion currents, whereas coexpression of WT PC1 and TRPP2 produced no currents. Interestingly, the C-type lectin domain from the PC1 N-terminus was used as a soluble activator of the PC1-TRPP2 F604P complex, suggesting that extracellular ligands binding to the complex can modulate channel activity.ref. bib1254 Furthermore, it was shown that cilia-enriched oxysterol 7β,27-dihydroxycholesterol is required for TRPP2 ion channel activation.ref. bib1257 The key takeaway from these studies is that heteromeric PC1-TRPP2 channels without GOF mutations appear to be constitutively closed, and active mutant channels in the heteromeric complex are more Ca2+-permeable than homomeric TRPP2 channels. The identification of the physiological activation mechanism of the PC1-TRPP2 complex remains one of the most important future challenges, because it will enable the study of the biophysical properties of the native WT complex and downstream signaling pathways, which may be dysregulated in ADPKD.
The PKD1L1-TRPP2 complex is required for the establishment of left-right organ asymmetry (see “Expression pattern and primary physiological roles of TRPP channels”).ref. 1259., ref. 1260., ref. 1261. Cilia-mediated asymmetric Ca2+ signals in the embryonic node have been shown to result in asymmetric gene expression to establish left-right asymmetry.ref. bib1258,ref. bib1259,ref. bib1261,ref. bib1262 Genetic data from humans, mice, and zebrafish implicate the PKD1L1-TRPP2 complex in the generation of these asymmetric Ca2+ signals.ref. 1259., ref. 1260., ref. 1261.,ref. bib1263 However, there are no direct measurements of PKD1L1-TRPP2 channels in the embryonic node to date. Future work will have to determine the biophysical properties of this complex and its activation mechanism.
Heteromeric TRPP channel complexes with PKDREJ have been proposed to play a role in fertilization. PKDREJ-TRPP2 and PKDREJ-TRPP3 co-immunoprecipitate when over-expressed in HEK293 cells.ref. bib1264 To date, there are no functional channel data of these heteromeric complexes.
In summary, the modular assembly of TRPP2 with different members of the PC1 family appears to enable tissue-specific functions that are tuned to specific physiological requirements, eg, responsiveness to different, yet to be identified, ligands that activate the respective heteromeric complexes.
TRPP3
Unlike TRPP2, ion currents from homomeric TRPP3 channels can be measured from the plasma membrane when heterologously expressed.ref. bib1207 TRPP3 is an outwardly rectifying nonselective cation channel which conducts mono- and divalent cations.ref. bib1213 TRPP3 is more Ca2+-selective than TRPP2 with a Ca2+ permeability that is 15 times higher than that for Na+, probably because of an additional aspartate (D525) in the selectivity filter.ref. bib1218 TRPP3 has properties of voltage-dependent channels, such as voltage-dependent inactivation and tail currents after membrane repolarization.ref. bib1216,ref. bib1219 Similar to TRPP2, TRPP3 activity is modulatedref. bib1218 by intracellular Ca2+. In heterologous expression systems (Xenopus oocytes and mammalian cells), TRPP3 has been shown to be activated by acidic and alkaline extracellular pH,ref. bib1265,ref. bib1266 and has been proposed to play a role in sour taste transduction (see “Expression pattern and primary physiological roles of TRPP channels”).
Heteromeric PKD1L1-TRPP3 channels have been shown to regulate the ciliary Ca2+ concentration.ref. bib1267 Endogenous PKD1L1-TRPP3 channels have been measured directly by patch-clamping of primary cilia in fibroblasts and retinal pigment epithelial cells. These currents recorded from cilia were activatedref. bib1247 by ATP and blocked by Gd3+. High membrane pressure increased the open probability of heteromeric PKD1L1-TRPP3 channels, but there is currently no data suggesting a direct role of this heteromeric complex in ciliary mechanotransduction. Since homomeric TRPP3 channels can be measured at the plasma membrane and in cilia, the contribution of the PKD1L1 subunit to the functional pore can be determined by comparing the permeation properties of the homomeric and heteromeric channels.ref. bib1213,ref. bib1218,ref. bib1247 The single-channel conductance of homomeric TRPP3 channels is larger than the conductance of heteromeric PKD1L1-TRPP3 channels (156 pS versus 96 pS, respectively; with Na+ as charge carrier). The Ca2+ selectivity over Na+ of monomeric TRPP3 channels is higher than that of PKD1L1-TRPP3 channels (15- versus 6-fold, respectively). These alterations of the biophysical properties of the heteromeric complex are likely explained by the contribution of the PKD1L1 pore domain (S10-S11) to the selectivity filter of the heteromeric PKD1L1-TRPP3 complex. The precise structural features determining these biophysical properties remain to be investigated.
Co-expression of PKD1L3 and TRPP3 in HEK293 cells, Xenopus oocytes, and HEK cells results in ion currents that are activated by extracellular Ca2+ and pH changes (acidic and alkaline).ref. bib1268,ref. bib1269 PKD1L3-TRPP3 operates as nonselective cation channel with preference for Ca2+ over Na+ (PCa2+/PNa+ ≈ 11). Interestingly, the pH- or Ca2+-activated currents have no voltage dependence.ref. bib1270,ref. bib1271 Since the regulation by Ca2+ and pH is also observed in homomeric TRPP3 channels, it is difficult to distinguish whether this regulation is a feature of homo- or heteromeric channels in an overexpression system. Taken together, PKD1L3 and TRPP3 form complexes, but their functional features and their physiological relevance require further investigation.
TRPP5
The biophysical properties and the physiological function of TRPP5 channels are the least well characterized of the TRPP channels. It has been reported that overexpression of TRPP5 in HEK293 cells produces single-channel currents with a conductance of 25 pS that are not voltage-sensitive.ref. bib1264,ref. bib1272 There are no reports of endogenous TRPP5 currents.
Expression pattern and primary physiological roles of TRPP channels
Expression pattern
The genes encoding TRPP channels are expressed in many organs in vertebrates. Transcriptome analyses have detected mRNA of TRPP channels in nearly all human and mouse tissues. Targeted studies focusing on individual TRPP channels have confirmed and expanded these findings: PKD2 and PKD2L1 transcripts are present in numerous fetal and adult tissues, including the heart, brain, lungs, spleen, testes, ovaries, and kidneys.ref. bib1182,ref. bib1187,ref. bib1188,ref. bib1208,ref. bib1271,ref. bib1273 PKD2L2 expression appears to be more restricted to the brain and testis.ref. bib1184,ref. bib1188 Splice variants of TRPP channels have been identified, but their functional properties are currently unknown.ref. bib1182,ref. bib1184,ref. bib1187,ref. bib1188
PKD2 expression is modulated by post-transcriptional regulation. The RNA-binding protein bicaudal C (BICC1) and the microRNA group 17 (miR-17) have been reported to regulate TRPP2 expression levels in opposite directions.ref. 1275., ref. 1276., ref. 1277. miR-17 has been shown to repress Pkd2 expression by binding on the 3′-untranslated region of Pkd2 mRNA. This may be of physiological relevance since overexpression of miR-17 in the kidneys of transgenic mice leads to kidney cysts. In contrast, loss of miR-17 reduced cyst growth in a mouse model with polycystic kidney disease caused by Kif3a KO.ref. bib1274 Conversely, BICC1 enhances Pkd2 mRNA stability and translation efficiency.ref. bib1276 Loss of Bicc1 results in cystic kidneys in model organisms and antagonizes the repressive activity of miR-17.ref. bib1276 Since miR-17 targets many genes, including several genes associated with cystic kidney disease, it remains to be determined whether the effects of miR-17 on kidney cysts are caused solely by the reduction of TRPP2 expression.
TRPP2 and PC1 have also been studied in invertebrate model organisms, which provided fundamental biological insights such as the discovery that the polycystins localize in primary cilia.ref. 1278., ref. 1279., ref. 1280., ref. 1281., ref. 1282., ref. 1283. In Caenorhabditis elegans and Drosophila melanogaster, Pkd2 expression is restricted to ciliated cells, namely male-specific sensory neurons and spermatozoa, respectively.ref. bib1277,ref. bib1282 In the meantime, a convergence of additional findings from mammalian model organisms suggests that defective ciliary signaling plays an important role in the pathogenesis of polycystic kidney disease and related disorders, which are now collectively called ciliopathies.ref. bib1283 In addition to primary cilia, PC1 and TRPP2 have been found in the ER and the lateral membrane.ref. bib1243,ref. bib1284 More recently, fragments of PC1 have also been detected in mitochondria.ref. 1286., ref. 1287., ref. 1288.
The physiologically relevant cellular localization of the PC1-TRPP2 complex is debated, but considerable evidence suggests that this channel complex functions in primary cilia or in the plasma membrane. However, a function in other membranes, for example, in the ER, cannot be ruled out and requires further studies. Trafficking of PC1 and TRPP2 to the plasma membrane or cilia appears to be interdependent, supporting the hypothesis that the heteromeric complex, rather than homomeric assemblies of each subunit, is the functionally relevant channel complex at these locations.ref. bib1288,ref. bib1289 However, it has also been reportedref. bib1290 that TRPP2 traffics to cilia without PC1. In addition to PC1, multiple other proteins have been shown to be involved in the trafficking of TRPP2 to cilia and the plasma membrane.ref. bib1196,ref. bib1243,ref. bib1291,ref. bib1292
The identification of specific cell types expressing TRPP proteins in vivo has been challenging. While TRPP2-specific antibodies enable detection of TRPP2 by Western blot in the kidney, in the heart, and in other organs, the unequivocal detection of the cellular and subcellular distribution of TRPP2 protein in vivo has been hampered by low expression levels and the lack of antibodies with sufficient sensitivity and specificity for immunohistochemical and immunofluorescence studies. The same applies to TRPP3 and TRPP5. In the future, this limitation may be overcome by the introduction of epitope tags to endogenously expressed TRPP channels,ref. bib1286 or by generating TRPP-reporter alleles to detect cell types expressing these channels using combinatorial genetic approaches, which have been successfully employed to detect the expression of other TRP channels in specific cell types.ref. bib1293
Primary physiological roles of TRPP channels
The physiological roles of TRPP channels have been studied in several model organisms with a focus on TRPP2 function because of its relevance in human disease.
TRPP2
The primary physiological roles of TRPP2 in vertebrates are the regulation of tubular morphogenesis and the establishment of organ left-right asymmetry.ref. bib1179,ref. bib1294 The importance of TRPP2 in controlling the morphology of epithelial tubules was first recognized when PKD2 was cloned as the second gene mutated in ADPKD patients.ref. bib1182 The requirement of TRPP2 for the formation of properly shaped tubules and for left-right patterning was later confirmed in mouse and zebrafish.ref. 1296., ref. 1297., ref. 1298., ref. 1299. Loss of TRPP2 results in polycystic kidney disease in mice,ref. bib1298 and pronephric cysts in zebrafish larvae.ref. bib1296 Constitutive Pkd2 KO mice develop cystic kidneys, edema, and hemorrhage and die in utero around midgestation.ref. 1298., ref. 1299., ref. 1300. Kidney cysts originate from dilatations along all nephron segments mimicking cyst formation in human ADPKD, with the notable exception that cyst formation in ADPKD is focal due to loss of heterozygosity in individual tubule cells, whereas PKD mouse models display much more widespread cyst formation due to Pkd2 inactivation in all tubular epithelial cells. A mouse model with a Pkd2 allele prone to spontaneous recombination (Pkd2WS25) mimics the loss of heterozygosity through somatic mutations in ADPKD and shows focal cyst formation similar to the human disease.ref. bib1298 Conditional inactivation of Pkd2 in specific cell types prevents embryonic lethality and enables studies of organ-specific functions of TRPP2.ref. bib1299 Mutations in PKD1 in humans or KO of Pkd1 in mice also cause polycystic kidney disease.ref. bib1300 Multiple lines of evidence, including the similarity of KO phenotypes of Pkd1 and Pkd2, the coassembly of PC1 and TRPP2 in a heteromeric complex, and their interdependence of trafficking to cilia, support the notion that the PC1-TRPP2 complex rather than homomeric assemblies of these proteins are critical for the proper regulation of tubular shape (Fig. 10A). How Ca2+ signals triggered by this complex control the shape of epithelial tubes remains to be determined.
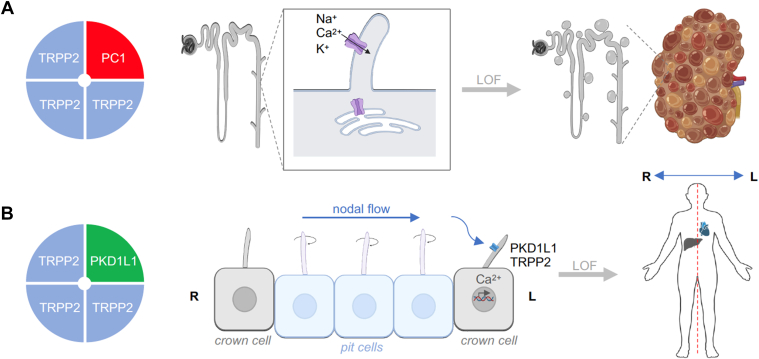
A second important physiological function of TRPP2 is its role in the establishment of left-right asymmetry. Loss of TRPP2 in mice and zebrafish causes left-right asymmetry defects that have been shown to be caused by impaired TRPP2-mediated Ca2+ signaling in the embryonic node.ref. bib1260,ref. 1296., ref. 1297., ref. 1298. Left-right asymmetry is controlled by Ca2+-dependent asymmetric gene expression in cells on one side of the embryonic node, a transient concave structure in the midline of the postgastrulation embryo.ref. bib1301 The cells in the embryonic node are ciliated. So-called pit cells in the embryonic node generate fluid flow directed toward the left side by the beating of motile cilia. Perinodal crown cells possess immotile primary cilia, which are required to sense the fluid flow to trigger intracellular Ca2+ signals (Fig. 10B).ref. bib1260,ref. bib1301 It is still debated whether the signal or stimulus sensed by these cilia is mechanical or chemical.ref. bib1260,ref. bib1261,ref. 1296., ref. 1297., ref. 1298.,ref. bib1302 Notably, Pkd1 KO mice do not display left-right asymmetry defects despite otherwise extensive phenotypic similarities to Pkd2 KO mice. However, loss of PKD1L1 causes left-right asymmetry defects in humans and model organisms.ref. bib1258,ref. bib1259,ref. bib1263 Since Ca2+ transients in perinodal crown cells require both TRPP2 and PKD1L1, and both channel subunits localize to primary cilia, PKD1L1-TRPP2 heteromeric channels probably function as sensors of nodal flow in the embryo (Fig. 10B).
TRPP2 channels are broadly expressed and likely have additional physiological functions. This is supported by the embryonic lethality of constitutive Pkd2 KO mice, which is not caused by the kidney phenotype.ref. bib1298,ref. bib1299 Instead, the embryonic lethality in Pkd2 KO mice appears to be caused by vascular defects in the placenta.ref. bib1299 TRPP2 and PC1 appear to have a role in vascular integrity, since loss of function of both proteins causes cardiovascular phenotypes, ranging from cardiac valve defects to aneurysms and abnormal vascular permeability in KO mice.ref. bib1299,ref. bib1303,ref. bib1304 Extrarenal manifestations of ADPKD patients support a role of PC1 and TRPP2 in the cardiovascular system.ref. bib1305 In cardiomyocytes, TRPP2 has been reported to regulate Ca2+ release through ryanodine receptors through direct association.ref. bib1306 A recent study showed that natriuretic peptide production requires TRPP2 in the heart, and loss of this pathway may contribute to the development of hypertension in ADPKD.ref. bib1307 Studies of TRPP2 in the regulation of vascular tone are contradictory. In arterial smooth muscle cells, TRPP2 is proposed to contribute to systemic blood pressure and to the myogenic response in cerebral arteries through vasoconstriction.ref. bib1308,ref. bib1309 In vascular endothelial cells, TRPP2 was reported to mediate vasodilation through activation of nitric oxide synthase.ref. bib1310 In addition, TRPP2 and Filamin-A have been proposed to regulate pressure sensing in mouse vascular smooth muscle cells, by fine-tuning stretch-activated channels to adapt the vascular myogenic response.ref. bib1311 These seemingly paradoxical functions of TRPP2 in the vasculature might be explained by differences in specific locations and cell types within the vasculature. Further studies are required for a comprehensive understanding of TRPP2 function in the vasculature and other organs.
TRPP3
The physiological functions of TRPP3 are much less well understood. The phenotypes of TRPP3 (Pkd2l1) KO models suggest functions in the CNS, cardiomyocytes, and early development. Pkd2l1 KO mice show hippocampal and thalamo-cortical hyperexcitability with increased susceptibility to seizures.ref. bib1312 Like TRPP2, TRPP3 localizes to primary cilia. TRPP3 channel activity has been measured in neurons contacting the subependymal cerebrospinal fluid. These neurons have protrusions with a primary cilium that extends into the central canal, where it is thought to sense mechanical or chemical signals from the cerebrospinal fluid.ref. bib1273,ref. bib1313,ref. bib1314 In zebrafish, related neurons contacting the cerebrospinal fluid were shown to be mechanosensitive cells. The detection of cerebrospinal fluid flow through these neurons was shown to require mechanosensitive TRPP3 channels.ref. bib1314
The organismal function of heteromeric PKD1L1-TRPP3 channels remains poorly understood. PKD1L1-TRPP3 channels have been reportedref. bib1267 to control the Ca2+ concentration in cilia and to regulate Hedgehog-dependent transcription of glioma-associated oncogene homolog 1. The physiological consequences of these cellular events in vivo remain to be determined.
Heteromeric PKD1L3-TRPP3 channels have been proposed as a candidate sour taste receptor in gustatory cells.ref. bib1271,ref. bib1315 TRPP3 is expressed in some gustatory type III cells, and acid-evoked Ca2+ responses and optogenetic activation of these cells support a role of these cells in sour taste perception.ref. bib1269,ref. bib1315,ref. bib1316 However, the role of the PKD1L3-TRPP3 in sour taste transduction is controversial. Mice with genetic ablation of TRPP3-expressing cells were shown to be completely devoid of acid responses in electrophysiology recordings to sour stimuli, supporting a role of these cells in sour taste reception.ref. bib1315 Based on these and additional results showing acid activation of the complex, PKD1L3-TRPP3 channels were proposed to form the sour taste receptor.ref. bib1269 Subsequent studies in PKD1L3-deficient mice, however, showed normal sour taste responsiveness in behavioral and electrophysiological experiments.ref. bib1317
TRPP5
Mouse TRPP5 mRNA and protein expression have been reported in spermatocytes and spermatids, but its role in male reproduction or other physiological functions has not been studied yet.ref. bib1318
Human diseases associated with TRPP channels
TRPP2
TRPP2 was first identified as the gene product of PKD2, the second causative gene for ADPKD.ref. bib1182 Mutations in PKD2 account for ∼15% of ADPKD cases, mutations in PKD1 for ∼80%, and a few additional genes for the remaining 5%.ref. bib1183,ref. bib1319 ADPKD is by far the most common genetic cause of kidney failure and affects ∼1/1000 individuals in the general population.ref. bib1319,ref. bib1320 The disease is characterized by polycystic kidneys, with cyst development starting in the fetus and continuing through a patient’s lifetime. Continuous development and growth of cysts compresses the remaining tubules. In the majority of patients, this results in reduced kidney function and ultimately kidney failure. The clinical course of ADPKD is highly variable, but ∼50% of patients have kidney failure by 60 years of age.ref. bib1320 Multiple extra-renal clinical manifestations, including liver cysts, pancreas cysts, intracranial aneurysms, and cardiac valvular disease, show that ADPKD is a systemic disorder.ref. bib1319,ref. bib1320 These extrarenal clinical manifestations point to functions of TRPP2 and PC1 in multiple organs, which are continuing to be studied in conditional mouse models.
Hundreds of unique ADPKD mutations have been identified, which are spread across PKD1 and PKD2 without obvious mutational hotspots (The ADPKD Mutation Database, https://pkdb.mayo.edu/variants). Patients with PKD1 mutations tend to have more severe disease compared with those with PKD2 mutations, and truncating mutations usually result in a more severe phenotype than nontruncating missense mutations.ref. bib1321 There is significant inter- and intrafamilial variability in ADPKD symptoms even in patients with the same germline mutation. This suggests the existence of genetic, environmental, and epigenetic modifiers of ADPKD.
Each human kidney has about 1,000,000 nephrons. However, cysts develop only in a very small fraction (1%–5%) of nephrons. The focal nature of cyst formation in ADPKD can be explained by the 2-hit model.ref. bib1322,ref. bib1323 According to this model, a germline mutation (first hit) and a somatic mutation (second hit) in the normal allele are required for cyst formation in ADPKD. The loss of heterozygosity in kidney cells leads to the complete loss of functional polycystin proteins, which causes focal cyst formation. Thus, even though the mode of inheritance of ADPKD is dominant, the process of cyst formation is recessive at the cellular level. Multiple lines of evidence ranging from genetic analyses of cyst epithelia in patients to mouse models support the 2-hit model.ref. bib1298,ref. bib1323,ref. bib1324
TRPP3
To date, no variants in PKD2L1, the gene encoding TRPP3, have been associated with human disease.
TRPP5
Like for TRPP3, there are no reports of human disease associated with variants in TRPP5.
Pharmacological modulators of TRPP channels
There is very limited information on pharmacological modulators of TRPP channels. No validated specific activators or blockers of TRPP2 are available because of the difficulty of measuring TRPP2 channel activity in heterologous expression systems. A recent study showed that some TRPML agonists (MK6-83, ML2-SA1, SF-21, SF-22, SF-23, SF-24, SF-31, SF-32, SF-33, SF-41, SF-71, SN-2, and rapamycin) inhibit the activity of TRPP2 with a F604P GOF mutation at high concentrations (see “Pharmacological modulators of TRPP channels”). Two of these TRPML agonists, ML-SA1 and SF-51, further activate the TRPP2 F604P channel, but not WT TRPP2, at low concentrations and inactivate it at higher concentrations.ref. bib1325 TRPP3 is activated by acidic pH (see above), and is blocked by flufenamic acid at rather high concentrations (0.5 mM).ref. bib1207 Furthermore, TRPP3 has been shown to be inhibited by amiloride, phenamil, benzamil, and 5-(N-ethyl-N-isopropyl)amiloride with an order of potency of phenamil > benzamil > 5-(N-ethyl-N-isopropyl)amiloride > amiloride, with IC50 values of 0.14, 1.1, 10.5, and 143 μM, respectively.ref. bib1326 There is still only one study reporting TRPP5 channel measurements,ref. bib1272 and no pharmacological modulators of TRPP5 are available to date.
Ongoing or completed clinical trials with TRPP channels as therapeutic targets
There are no ongoing or completed clinical trials with TRPP channels as therapeutic targets. The majority of ADPKD is caused by mutations in PKD1, which led to the hypothesis that pharmacological activation of TRPP2 might mitigate the disease. However, there are no validated pharmacological activators of WT TRPP2 to date, and it remains to be determined whether pharmacological activation of TRPP2 can compensate for the loss of PC1, which is thought to be an essential subunit of the heteromeric PC1-TRPP2 complex. The development of ivacaftor and related drugs for the treatment of cystic fibrosis has shown the efficacy of potentiators and correctors of mutated ion channels harboring missense mutations.ref. bib1327 It is conceivable that similar approaches might be applicable for ADPKD.
Conclusions and outlook
Recent experiments using animal disease models and human genetic studies have linked TRP channels to various pathophysiological processes, highlighting their broad therapeutic potential. Moreover, significant progress has been made in developing potent pharmacological agents targeting TRP channels in conjunction with electrophysiological and structural analysis of these proteins, which provides the mechanistic basis for innovative treatments of a wide array of human disorders. Despite the relevance of TRP channels as pivotal therapeutic targets for the treatment of human diseases, the clinical modulation of TRP channels has turned out to be more challenging than initially anticipated. The following 3 preclinical issues deserve future attention. (1) There still is a pressing need to further our understanding of the pathophysiological role of TRP channels, their exact contribution to cellular, tissue, and organismal homeostasis and dysfunction, including activation mechanisms in a native environment and reliable tissue expression with high spatial resolution. (2) Unwanted side effects, as noted in clinical trials, may arise from either off-target or off-tissue interactions of drug candidates. Recent advances in molecular approaches, such as single-particle cryo-EM, in combination with AI-guided computational methods, will refine the development of modality-specific and activity-dependent modulators, which can be validated through in-depth biophysical analyses. To limit off-tissue side effects, the direct local or topical application of TRP channel modulators appears to be an appropriate strategy. Long-term toxicity of topically applied chemical probes and drug candidates can be averted by controlled local inactivation of compounds, for instance, by introducing photoswitches. (3) To foster clinical translation, reliable and robust preclinical disease models must be developed, including genetically modified mouse models, in vitro human-derived organoids, and engineered human tissue cultured in biomimetic chambers. In this regard, progress in cellular reprogramming of iPSCs holds the promise of providing relevant preclinical models for early validation of TRP channel modulators.
Conflict of interest
Thomas Gudermann functions as an Associate Editor of Pharmacological Reviews. All other authors declare no conflicts of interest.
References
- D.J. Cosens, A. Manning. Abnormal electroretinogram from a Drosophila mutant. Nature, 1969. [DOI | PubMed]
- C. Montell, G.M. Rubin. Molecular characterization of the Drosophila trp locus: a putative integral membrane protein required for phototransduction. Neuron, 1989. [DOI | PubMed]
- F. Wong, E.L. Schaefer, B.C. Roop, J.N. LaMendola, D. Johnson-Seaton, D. Shao. Proper function of the Drosophila trp gene product during pupal development is important for normal visual transduction in the adult. Neuron, 1989. [DOI | PubMed]
- B. Minke, C. Wu, W.L. Pak. Induction of photoreceptor voltage noise in the dark in Drosophila mutant. Nature, 1975. [DOI | PubMed]
- C. Montell. The TRP superfamily of cation channels. Sci STKE, 2005. [DOI]
- K. Venkatachalam, C. Montell. TRP channels. Annu Rev Biochem, 2007. [DOI | PubMed]
- C. Montell, L. Birnbaumer, V. Flockerzi. A unified nomenclature for the superfamily of TRP cation channels. Mol Cell, 2002. [DOI | PubMed]
- D.E. Clapham. SnapShot: mammalian TRP channels. Cell, 2007. [DOI | PubMed]
- M. Gees, G. Owsianik, B. Nilius, T. Voets. TRP channels. Compr Physiol, 2012. [DOI | PubMed]
- D.E. Clapham. TRP channels as cellular sensors. Nature, 2003. [DOI | PubMed]
- H. Ledford, E. Callaway. Medicine Nobel goes to scientists who discovered biology of senses. Nature, 2021. [DOI]
- L. Yue, H. Xu. TRP channels in health and disease at a glance. J Cell Sci, 2021. [DOI]
- M.M. Diver, J.V. Lin King, D. Julius, Y. Cheng. Sensory TRP channels in three dimensions. Annu Rev Biochem, 2022. [DOI | PubMed]
- A. Dhaka, V. Viswanath, A. Patapoutian. Trp ion channels and temperature sensation. Annu Rev Neurosci, 2006. [DOI | PubMed]
- E.R. Liman, H. Innan. Relaxed selective pressure on an essential component of pheromone transduction in primate evolution. Proc Natl Acad Sci U S A, 2003. [DOI | PubMed]
- J. Zhang, D.M. Webb. Evolutionary deterioration of the vomeronasal pheromone transduction pathway in catarrhine primates. Proc Natl Acad Sci U S A, 2003. [DOI | PubMed]
- C. Fan, W. Choi, W. Sun, J. Du, W. Lü. Structure of the human lipid-gated cation channel TRPC3. eLife, 2018. [DOI]
- Q. Tang, W. Guo, L. Zheng. Structure of the receptor-activated human TRPC6 and TRPC3 ion channels. Cell Res, 2018. [DOI | PubMed]
- W. Guo, Q. Tang, M. Wei, Y. Kang, J.X. Wu, L. Chen. Structural mechanism of human TRPC3 and TRPC6 channel regulation by their intracellular calcium-binding sites. Neuron, 2022. [DOI | PubMed]
- D. Vinayagam, T. Mager, A. Apelbaum. Electron cryo-microscopy structure of the canonical TRPC4 ion channel. eLife, 2018. [DOI]
- D. Vinayagam, D. Quentin, J. Yu-Strzelczyk. Structural basis of TRPC4 regulation by calmodulin and pharmacological agents. eLife, 2020. [DOI]
- J. Duan, J. Li, B. Zeng. Structure of the mouse TRPC4 ion channel. Nat Commun, 2018. [DOI | PubMed]
- J. Duan, J. Li, G.L. Chen. Cryo-EM structure of TRPC5 at 2.8-A resolution reveals unique and conserved structural elements essential for channel function. Sci Adv, 2019. [DOI]
- K. Song, M. Wei, W. Guo. Structural basis for human TRPC5 channel inhibition by two distinct inhibitors. eLife, 2021. [DOI]
- D.J. Wright, K.J. Simmons, R.M. Johnson, D.J. Beech, S.P. Muench, R.S. Bon. Human TRPC5 structures reveal interaction of a xanthine-based TRPC1/4/5 inhibitor with a conserved lipid binding site. Commun Biol, 2020. [DOI | PubMed]
- Y. Yang, M. Wei, L. Chen. Structural identification of riluzole-binding site on human TRPC5. Cell Discov, 2022. [DOI | PubMed]
- J. Won, J. Kim, H. Jeong. Molecular architecture of the Gαi-bound TRPC5 ion channel. Nat Commun, 2023. [DOI | PubMed]
- Y. Bai, X. Yu, H. Chen. Structural basis for pharmacological modulation of the TRPC6 channel. eLife, 2020. [DOI]
- C.M. Azumaya, F. Sierra-Valdez, J.F. Cordero-Morales, T. Nakagawa. Cryo-EM structure of the cytoplasmic domain of murine transient receptor potential cation channel subfamily C member 6 (TRPC6). J Biol Chem, 2018. [DOI | PubMed]
- J. Won, J. Kim, J. Kim. Cryo-EM structure of the heteromeric TRPC1/TRPC4 channel. Nat Struct Mol Biol, 2025. [DOI | PubMed]
- K.E. Huffer, A.A. Aleksandrova, A. Jara-Oseguera, L.R. Forrest, K.J. Swartz. Global alignment and assessment of TRP channel transmembrane domain structures to explore functional mechanisms. eLife, 2020. [DOI]
- G. Vazquez, B.J. Wedel, O. Aziz, M. Trebak, J.W. Putney. The mammalian TRPC cation channels. Biochim Biophys Acta, 2004. [DOI | PubMed]
- M.P. Lussier, S. Cayouette, P.K. Lepage. MxA, a member of the dynamin superfamily, interacts with the ankyrin-like repeat domain of TRPC. J Biol Chem, 2005. [DOI | PubMed]
- R. Schindl, C. Romanin. Assembly domains in TRP channels. Biochem Soc Trans, 2007. [DOI | PubMed]
- R. Gaudet. A primer on ankyrin repeat function in TRP channels and beyond. Mol Biosyst, 2008. [DOI | PubMed]
- R. Schindl, I. Frischauf, H. Kahr. The first ankyrin-like repeat is the minimum indispensable key structure for functional assembly of homo- and heteromeric TRPC4/TRPC5 channels. Cell Calcium, 2008. [DOI | PubMed]
- M. Schaefer, T.D. Plant, A.G. Obukhov, T. Hofmann, T. Gudermann, G. Schultz. Receptor-mediated regulation of the nonselective cation channels TRPC4 and TRPC5. J Biol Chem, 2000. [DOI | PubMed]
- S. Jung, A. Muhle, M. Schaefer, R. Strotmann, G. Schultz, T.D. Plant. Lanthanides potentiate TRPC5 currents by an action at extracellular sites close to the pore mouth. J Biol Chem, 2003. [DOI | PubMed]
- A. Beck, T. Speicher, C. Stoerger. Conserved gating elements in TRPC4 and TRPC5 channels. J Biol Chem, 2013. [DOI | PubMed]
- Y. Yin, S.C. Le, A.L. Hsu, M.J. Borgnia, H. Yang, S.Y. Lee. Structural basis of cooling agent and lipid sensing by the cold-activated TRPM8 channel. Science, 2019. [DOI]
- Y. Yin, M. Wu, L. Zubcevic, W.F. Borschel, G.C. Lander, S.Y. Lee. Structure of the cold- and menthol-sensing ion channel TRPM8. Science, 2018. [DOI | PubMed]
- T. Rohacs, C.M. Lopes, I. Michailidis, D.E. Logothetis. PI(4,5)P2 regulates the activation and desensitization of TRPM8 channels through the TRP domain. Nat Neurosci, 2005. [DOI | PubMed]
- M.X. Mori, R. Okada, R. Sakaguchi. Critical contributions of pre-S1 shoulder and distal TRP box in DAG-activated TRPC6 channel by PIP(2) regulation. Sci Rep, 2022. [DOI]
- A. Clarke, J. Skerjanz, M.A. Gsell. PIP2 modulates TRPC3 activity via TRP helix and S4-S5 linker. Nat Commun, 2024. [DOI | PubMed]
- S.Z. Xu, P. Sukumar, F. Zeng. TRPC channel activation by extracellular thioredoxin. Nature, 2008. [DOI | PubMed]
- M. Berridge. Conformational coupling: a physiological calcium entry mechanism. Sci STKE, 2004. [DOI]
- H. Kanki, M. Kinoshita, A. Akaike, M. Satoh, Y. Mori, S. Kaneko. Activation of inositol 1,4,5-trisphosphate receptor is essential for the opening of mouse TRP5 channels. Mol Pharmacol, 2001. [DOI | PubMed]
- U. Storch, M. Mederos Y Schnitzler, T.T. Gudermann. A greasy business: identification of a diacylglycerol binding site in human TRPC5 channels by cryo-EM. Cell Calcium, 2021. [DOI]
- M. Lichtenegger, O. Tiapko, B. Svobodova. An optically controlled probe identifies lipid-gating fenestrations within the TRPC3 channel. Nat Chem Biol, 2018. [DOI | PubMed]
- H. Erkan-Candag, A. Clarke, O. Tiapko, M.A. Gsell, T. Stockner, K. Groschner. Diacylglycerols interact with the L2 lipidation site in TRPC3 to induce a sensitized channel state. EMBO Rep, 2022. [DOI]
- U. Storch, A.L. Forst, M. Philipp, T. Gudermann, M. Mederos y Schnitzler. Transient receptor potential channel 1 (TRPC1) reduces calcium permeability in heteromeric channel complexes. J Biol Chem, 2012. [DOI | PubMed]
- B. Lintschinger, M. Balzer-Geldsetzer, T. Baskaran. Coassembly of Trp1 and Trp3 proteins generates diacylglycerol- and Ca2+-sensitive cation channels. J Biol Chem, 2000. [DOI | PubMed]
- S. Sours-Brothers, M. Ding, S. Graham, R. Ma. Interaction between TRPC1/TRPC4 assembly and STIM1 contributes to store-operated Ca2+ entry in mesangial cells. Exp Biol Med (Maywood), 2009. [DOI | PubMed]
- C. Strubing, G. Krapivinsky, L. Krapivinsky, D.E. Clapham. TRPC1 and TRPC5 form a novel cation channel in mammalian brain. Neuron, 2001. [DOI | PubMed]
- C. Strubing, G. Krapivinsky, L. Krapivinsky, D.E. Clapham. Formation of novel TRPC channels by complex subunit interactions in embryonic brain. J Biol Chem, 2003. [DOI | PubMed]
- X. Liu, B.C. Bandyopadhyay, B.B. Singh, K. Groschner, I.S. Ambudkar. Molecular analysis of a store-operated and 2-acetyl-sn-glycerol-sensitive non-selective cation channel. Heteromeric assembly of TRPC1-TRPC3. J Biol Chem, 2005. [DOI | PubMed]
- T. Kobori, G.D. Smith, R. Sandford, J.M. Edwardson. The transient receptor potential channels TRPP2 and TRPC1 form a heterotetramer with a 2: 2 stoichiometry and an alternating subunit arrangement. J Biol Chem, 2009. [DOI | PubMed]
- X. Ma, S. Qiu, J. Luo. Functional role of vanilloid transient receptor potential 4-canonical transient receptor potential 1 complex in flow-induced Ca2+ influx. Arterioscler Thromb Vasc Biol, 2010. [DOI | PubMed]
- R. Schindl, R. Fritsch, I. Jardin. Canonical transient receptor potential (TRPC) 1 acts as a negative regulator for vanilloid TRPV6-mediated Ca2+ influx. J Biol Chem, 2012. [DOI | PubMed]
- T. Hofmann, M. Schaefer, G. Schultz, T. Gudermann. Subunit composition of mammalian transient receptor potential channels in living cells. Proc Natl Acad Sci U S A, 2002. [DOI | PubMed]
- K.D. Phelan, U.T. Shwe, J. Abramowitz. Canonical transient receptor channel 5 (TRPC5) and TRPC1/4 contribute to seizure and excitotoxicity by distinct cellular mechanisms. Mol Pharmacol, 2013. [DOI | PubMed]
- A. Kollewe, Y. Schwarz, K. Oleinikov. Subunit composition, molecular environment, and activation of native TRPC channels encoded by their interactomes. Neuron, 2022. [DOI | PubMed]
- N. Medic, A. Desai, A. Olivera. Knockout of the Trpc1 gene reveals that TRPC1 can promote recovery from anaphylaxis by negatively regulating mast cell TNF-alpha production. Cell Calcium, 2013. [DOI | PubMed]
- J. Kim, M. Kwak, J.P. Jeon. Isoform- and receptor-specific channel property of canonical transient receptor potential (TRPC)1/4 channels. Pflugers Arch, 2014. [DOI | PubMed]
- Y. Eraç, Ç. Sellİ, M. Tosun. TRPC1 ion channel gene regulates store-operated calcium entryand proliferation in human aortic smooth muscle cells. Turk J Biol, 2016. [DOI]
- K.P. Lee, S. Choi, J.H. Hong. Molecular determinants mediating gating of Transient Receptor Potential Canonical (TRPC) channels by stromal interaction molecule 1 (STIM1). J Biol Chem, 2014. [DOI | PubMed]
- K.T. Cheng, X. Liu, H.L. Ong, W. Swaim, I.S. Ambudkar. Local Ca2+ entry via Orai1 regulates plasma membrane recruitment of TRPC1 and controls cytosolic Ca2+ signals required for specific cell functions. PLoS Biol, 2011. [DOI]
- E.C. Ong, V. Nesin, C.L. Long. A TRPC1 protein-dependent pathway regulates osteoclast formation and function. J Biol Chem, 2013. [DOI | PubMed]
- M. Trebak, G. Vazquez, G.S. Bird, J.W. Putney. The TRPC3/6/7 subfamily of cation channels. Cell Calcium, 2003. [DOI | PubMed]
- M. Goel, W.G. Sinkins, W.P. Schilling. Selective association of TRPC channel subunits in rat brain synaptosomes. J Biol Chem, 2002. [DOI | PubMed]
- M. Poteser, A. Graziani, C. Rosker. TRPC3 and TRPC4 associate to form a redox-sensitive cation channel. Evidence for expression of native TRPC3-TRPC4 heteromeric channels in endothelial cells. J Biol Chem, 2006. [DOI | PubMed]
- E.R. Liman, D.P. Corey, C. Dulac. TRP2: a candidate tranduction channel for mammalian pheromone sensory signaling. Proc Natl Acad Sci U S A, 1999. [DOI | PubMed]
- M. Trebak. Canonical transient receptor potential channels in disease: targets for novel drug therapy?. Drug Discov Today, 2006. [DOI | PubMed]
- T. Hofmann, A.G. Obukhov, M. Schaefer, C. Harteneck, T. Gudermann, G. Schultz. Direct activation of human TRPC6 and TRPC3 channels by diacylglycerol. Nature, 1999. [DOI | PubMed]
- P. Lucas, K. Ukhanov, T. Leinders-Zufall, F. Zufall. A diacylglycerol-gated cation channel in vomeronasal neuron dendrites is impaired in TRPC2 mutant mice: mechanism of pheromone transduction. Neuron, 2003. [DOI | PubMed]
- T. Okada, R. Inoue, K. Yamazaki. Molecular and functional characterization of a novel mouse transient receptor potential protein homologue TRP7. Ca(2+)-permeable cation channel that is constitutively activated and enhanced by stimulation of G protein-coupled receptor. J Biol Chem, 1999. [DOI | PubMed]
- U. Storch, A.L. Forst, F. Pardatscher. Dynamic NHERF interaction with TRPC4/5 proteins is required for channel gating by diacylglycerol. Proc Natl Acad Sci U S A, 2017. [DOI | PubMed]
- M. Ningoo, L.D. Plant, A. Greka, D.E. Logothetis. PIP2 regulation of TRPC5 channel activation and desensitization. J Biol Chem, 2021. [DOI]
- K. Venkatachalam, F. Zheng, D.L. Gill. Regulation of canonical transient receptor potential (TRPC) channel function by diacylglycerol and protein kinase C. J Biol Chem, 2003. [DOI | PubMed]
- J. Shi, E. Mori, Y. Mori. Multiple regulation by calcium of murine homologues of transient receptor potential proteins TRPC6 and TRPC7 expressed in HEK293 cells. J Physiol, 2004. [DOI | PubMed]
- K. Kiselyov, X. Xu, G. Mozhayeva. Functional interaction between InsP3 receptors and store-operated Htrp3 channels. Nature, 1998. [DOI | PubMed]
- J. Tang, Y. Lin, Z. Zhang, S. Tikunova, L. Birnbaumer, M.X. Zhu. Identification of common binding sites for calmodulin and inositol 1,4,5-trisphosphate receptors on the carboxyl termini of trp channels. J Biol Chem, 2001. [DOI | PubMed]
- M.X. Zhu, J. Tang. TRPC channel interactions with calmodulin and IP3 receptors. Novartis Found Symp, 2004. [PubMed]
- S. Stamboulian, M.J. Moutin, S. Treves. Junctate, an inositol 1,4,5-triphosphate receptor associated protein, is present in rodent sperm and binds TRPC2 and TRPC5 but not TRPC1 channels. Dev Biol, 2005. [DOI | PubMed]
- S. Treves, C. Franzini-Armstrong, L. Moccagatta. Junctate is a key element in calcium entry induced by activation of InsP3 receptors and/or calcium store depletion. J Cell Biol, 2004. [DOI | PubMed]
- S. Treves, M. Vukcevic, J. Griesser, C.F. Armstrong, M.X. Zhu, F. Zorzato. Agonist-activated Ca2+ influx occurs at stable plasma membrane and endoplasmic reticulum junctions. J Cell Sci, 2010. [DOI | PubMed]
- S. Srikanth, M. Jew, K.D. Kim, M.K. Yee, J. Abramson, Y. Gwack. Junctate is a Ca2+-sensing structural component of Orai1 and stromal interaction molecule 1 (STIM1). Proc Natl Acad Sci U S A, 2012. [DOI | PubMed]
- J.W. Putney. A model for receptor-regulated calcium entry. Cell Calcium, 1986. [DOI | PubMed]
- M. Hoth, R. Penner. Depletion of intracellular calcium stores activates a calcium current in mast cells. Nature, 1992. [DOI | PubMed]
- I. Jardin, G.M. Salido, J.A. Rosado. Role of lipid rafts in the interaction between hTRPC1, Orai1 and STIM1. Channels, 2008. [DOI | PubMed]
- M.S. Kim, W. Zeng, J.P. Yuan, D.M. Shin, P.F. Worley, S. Muallem. Native store-operated Ca2+ influx requires the channel function of Orai1 and TRPC1. J Biol Chem, 2009. [DOI | PubMed]
- M. Gueguinou, T. Harnois, D. Crottes. SK3/TRPC1/Orai1 complex regulates SOCE-dependent colon cancer cell migration: a novel opportunity to modulate anti-EGFR mAb action by the alkyl-lipid Ohmline. Oncotarget, 2016. [DOI | PubMed]
- I.S. Ambudkar, L.B. de Souza, H.L. Ong. TRPC1, Orai1, and STIM1 in SOCE: friends in tight spaces. Cell Calcium, 2017. [DOI | PubMed]
- J. Shi, F. Miralles, L. Birnbaumer, W.A. Large, A.P. Albert. Store depletion induces Gαq-mediated PLCβ1 activity to stimulate TRPC1 channels in vascular smooth muscle cells. FASEB J, 2016. [DOI | PubMed]
- J. Roos, P.J. DiGregorio, A.V. Yeromin. STIM1, an essential and conserved component of store-operated Ca2+ channel function. J Cell Biol, 2005. [DOI | PubMed]
- J. Liou, M.L. Kim, W.D. Heo. STIM is a Ca2+ sensor essential for Ca2+-store-depletion-triggered Ca2+ influx. Curr Biol, 2005. [DOI | PubMed]
- R.M. Luik, M.M. Wu, J. Buchanan, R.S. Lewis. The elementary unit of store-operated Ca2+ entry: local activation of CRAC channels by STIM1 at ER-plasma membrane junctions. J Cell Biol, 2006. [DOI | PubMed]
- A.V. Yeromin, S.L. Zhang, W. Jiang, Y. Yu, O. Safrina, M.D. Cahalan. Molecular identification of the CRAC channel by altered ion selectivity in a mutant of Orai. Nature, 2006. [DOI | PubMed]
- M. Vig, A. Beck, J.M. Billingsley. CRACM1 multimers form the ion-selective pore of the CRAC channel. Curr Biol, 2006. [DOI | PubMed]
- J.J. Lopez, I. Jardin, J. Sanchez-Collado, G.M. Salido, T. Smani, J.A. Rosado. TRPC channels in the SOCE scenario. Cells, 2020. [DOI | PubMed]
- G.S. Bird, J.W. Putney. Calcium Entry Channels in Non-excitable Cells, 2018
- S. Susperreguy, M. Yamashita, C.-I. Choi. Genetic evidence against involvement of TRPC proteins in SOCE, ROCE, and CRAC channel function. Proc Natl Acad Sci U S A, 2024. [DOI]
- T. Rohacs. Phosphoinositide regulation of non-canonical transient receptor potential channels. Cell Calcium, 2009. [DOI | PubMed]
- D.E. Logothetis, V.I. Petrou, M. Zhang. Phosphoinositide control of membrane protein function: a frontier led by studies on ion channels. Annu Rev Physiol, 2015. [DOI | PubMed]
- L. Lemonnier, M. Trebak, J.W. Putney. Complex regulation of the TRPC3, 6 and 7 channel subfamily by diacylglycerol and phosphatidylinositol-4,5-bisphosphate. Cell Calcium, 2008. [DOI | PubMed]
- M. Trebak, L. Lemonnier, W.I. DeHaven, B.J. Wedel, G.S. Bird, J.W. Putney. Complex functions of phosphatidylinositol 4,5-bisphosphate in regulation of TRPC5 cation channels. Pflugers Arch, 2009. [DOI | PubMed]
- A.P. Albert, S.N. Saleh, W.A. Large. Inhibition of native TRPC6 channel activity by phosphatidylinositol 4,5-bisphosphate in mesenteric artery myocytes. J Physiol, 2008. [DOI | PubMed]
- M. Ju, J. Shi, S.N. Saleh. Albert AP, Large WA. Ins(1,4,5)P3 interacts with PIP2 to regulate activation of TRPC6/C7 channels by diacylglycerol in native vascular myocytes. J Physiol, 2010. [DOI | PubMed]
- J. Shi, M. Ju, J. Abramowitz, W.A. Large, L. Birnbaumer, A.P. Albert. TRPC1 proteins confer PKC and phosphoinositol activation on native heteromeric TRPC1/C5 channels in vascular smooth muscle: comparative study of wild-type and TRPC1−/− mice. FASEB J, 2012. [DOI | PubMed]
- K. Otsuguro, J. Tang, Y. Tang. Isoform-specific inhibition of TRPC4 channel by phosphatidylinositol 4,5-bisphosphate. J Biol Chem, 2008. [DOI | PubMed]
- B.J. Kim, M.T. Kim, J.H. Jeon, S.J. Kim, I. So. Involvement of phosphatidylinositol 4,5-bisphosphate in the desensitization of canonical transient receptor potential 5. Biol Pharm Bull, 2008. [DOI | PubMed]
- H. Liu, W.Y. Lin, S.R. Leibow, A.J. Morateck, M. Ahuja, S. Muallem. TRPC3 channel gating by lipids requires localization at the ER/PM junctions defined by STIM1. J Cell Biol, 2022. [DOI]
- J. Huang, C.H. Liu, S.A. Hughes, M. Postma, C.J. Schwiening, R.C. Hardie. Activation of TRP channels by protons and phosphoinositide depletion in Drosophila photoreceptors. Curr Biol, 2010. [DOI | PubMed]
- D.P. Thakur, J.B. Tian, J. Jeon. Critical roles of Gi/o proteins and phospholipase C-δ1 in the activation of receptor-operated TRPC4 channels. Proc Natl Acad Sci U S A, 2016. [DOI | PubMed]
- M. Semtner, M. Schaefer, O. Pinkenburg, T.D. Plant. Potentiation of TRPC5 by protons. J Biol Chem, 2007. [DOI | PubMed]
- A.G. Obukhov, M.C. Nowycky. TRPC5 channels undergo changes in gating properties during the activation-deactivation cycle. J Cell Physiol, 2008. [DOI | PubMed]
- J.P. Jeon, K.P. Lee, E.J. Park. The specific activation of TRPC4 by Gi protein subtype. Biochem Biophys Res Commun, 2008. [DOI | PubMed]
- J.P. Jeon, C. Hong, E.J. Park. Selective Gαi subunits as novel direct activators of transient receptor potential canonical (TRPC)4 and TRPC5 channels. J Biol Chem, 2012. [DOI | PubMed]
- M.R. Whorton, R. MacKinnon. X-ray structure of the mammalian GIRK2-βγ G-protein complex. Nature, 2013. [DOI | PubMed]
- C. Hong, J. Kim, J.P. Jeon. Gs cascade regulates canonical transient receptor potential 5 (TRPC5) through cAMP mediated intracellular Ca2+ release and ion channel trafficking. Biochem Biophys Res Commun, 2012. [DOI | PubMed]
- S.A. Gross, G.A. Guzman, U. Wissenbach. TRPC5 is a Ca2+-activated channel functionally coupled to Ca2+-selective ion channels. J Biol Chem, 2009. [DOI | PubMed]
- N.T. Blair, J.S. Kaczmarek, D.E. Clapham. Intracellular calcium strongly potentiates agonist-activated TRPC5 channels. J Gen Physiol, 2009. [DOI | PubMed]
- J. Shi, N. Geshi, S. Takahashi. Molecular determinants for cardiovascular TRPC6 channel regulation by Ca2+/calmodulin-dependent kinase II. J Physiol, 2013. [DOI | PubMed]
- V.J. Bezzerides, I.S. Ramsey, S. Kotecha, A. Greka, D.E. Clapham. Rapid vesicular translocation and insertion of TRP channels. Nat Cell Biol, 2004. [DOI | PubMed]
- B. Thyagarajan, M. Poteser, C. Romanin, H. Kahr, M.X. Zhu, K. Groschner. Expression of Trp3 determines sensitivity of capacitative Ca2+ entry to nitric oxide and mitochondrial Ca2+ handling: evidence for a role of Trp3 as a subunit of capacitative Ca2+ entry channels. J Biol Chem, 2001. [DOI | PubMed]
- P.D. Wes, J. Chevesich, A. Jeromin, C. Rosenberg, G. Stetten, C. Montell. TRPC1, a human homolog of a Drosophila store-operated channel. Proc Natl Acad Sci U S A, 1995. [DOI | PubMed]
- X. Zhu, P.B. Chu, M. Peyton, L. Birnbaumer. Molecular cloning of a widely expressed human homologue for the Drosophila trp gene. FEBS Lett, 1995. [DOI | PubMed]
- L. Tsiokas, T. Arnould, C. Zhu, E. Kim, G. Walz, V.P. Sukhatme. Specific association of the gene product of PKD2 with the TRPC1 channel. Proc Natl Acad Sci U S A, 1999. [DOI | PubMed]
- G.X. Wang, M.M. Poo. Requirement of TRPC channels in netrin-1-induced chemotropic turning of nerve growth cones. Nature, 2005. [DOI | PubMed]
- S.J. Kim, Y.S. Kim, J.P. Yuan, R.S. Petralia, P.F. Worley, D.J. Linden. Activation of the TRPC1 cation channel by metabotropic glutamate receptor mGluR1. Nature, 2003. [DOI | PubMed]
- S. Bollimuntha, B.B. Singh, S. Shavali, S.K. Sharma, M. Ebadi. TRPC1-mediated inhibition of 1-methyl-4-phenylpyridinium ion neurotoxicity in human SH-SY5Y neuroblastoma cells. J Biol Chem, 2005. [DOI | PubMed]
- X. Wu, T.K. Zagranichnaya, G.T. Gurda, E.M. Eves, M.L. Villereal. A TRPC1/TRPC3-mediated increase in store-operated calcium entry is required for differentiation of H19-7 hippocampal neuronal cells. J Biol Chem, 2004. [DOI | PubMed]
- M. Li, C. Chen, Z. Zhou, S. Xu, Z. Yu. A TRPC1-mediated increase in store-operated Ca2+ entry is required for the proliferation of adult hippocampal neural progenitor cells. Cell Calcium, 2012. [DOI | PubMed]
- H.C. Chen, C.H. Wang, C.P. Shih. TRPC1 is required for survival and proliferation of cochlear spiral ganglion stem/progenitor cells. Int J Pediatr Otorhinolaryngol, 2015. [DOI | PubMed]
- J. Skerjanz, L. Bauernhofer, K. Lenk. TRPC1: the housekeeper of the hippocampus. Cell Calcium, 2024. [DOI]
- X. Liu, K.T. Cheng, B.C. Bandyopadhyay. Attenuation of store-operated Ca2+ current impairs salivary gland fluid secretion in TRPC1(-/-) mice. Proc Natl Acad Sci U S A, 2007. [DOI | PubMed]
- Y. Sun, L. Birnbaumer, B.B. Singh. TRPC1 regulates calcium-activated chloride channels in salivary gland cells. J Cell Physiol, 2015. [DOI | PubMed]
- B. Kumar, K. Dreja, S. Shah. Upregulated TRPC1 channel in vascular injury in vivo and its role in human neointimal hyperplasia. Circ Res, 2006. [DOI | PubMed]
- T. Ohba, H. Watanabe, M. Murakami. Upregulation of TRPC1 in the development of cardiac hypertrophy. J Mol Cell Cardiol, 2007. [DOI | PubMed]
- N. Tang, W. Tian, G.-Y. Ma. TRPC channels blockade abolishes endotoxemic cardiac dysfunction by hampering intracellular inflammation and Ca2+ leakage. Nat Commun, 2022. [DOI | PubMed]
- N. Kunichika, Y. Yu, C.V. Remillard, O. Platoshyn, S. Zhang, J.X. Yuan. Overexpression of TRPC1 enhances pulmonary vasoconstriction induced by capacitative Ca2+ entry. Am J Physiol Lung Cell Mol Physiol, 2004. [DOI | PubMed]
- M.J. Lin, G.P. Leung, W.M. Zhang. Chronic hypoxia-induced upregulation of store-operated and receptor-operated Ca2+ channels in pulmonary arterial smooth muscle cells: a novel mechanism of hypoxic pulmonary hypertension. Circ Res, 2004. [DOI | PubMed]
- A. Bergdahl, M.F. Gomez, A.K. Wihlborg. Plasticity of TRPC expression in arterial smooth muscle: correlation with store-operated Ca2+ entry. Am J Physiol Cell Physiol, 2005. [DOI | PubMed]
- S.E. Wolfle, M.F. Navarro-Gonzalez, T.H. Grayson, C. Stricker, C.E. Hill. Involvement of nonselective cation channels in the depolarisation initiating vasomotion. Clin Exp Pharmacol Physiol, 2010. [DOI | PubMed]
- H.Z.E. Greenberg, S.R.E. Carlton-Carew, D.M. Khan. Heteromeric TRPV4/TRPC1 channels mediate calcium-sensing receptor-induced nitric oxide production and vasorelaxation in rabbit mesenteric arteries. Vasc Pharmacol, 2017. [DOI]
- M.Y. Kochukov, A. Balasubramanian, R.C. Noel, S.P. Marrelli. Role of TRPC1 and TRPC3 channels in contraction and relaxation of mouse thoracic aorta. J Vasc Res, 2013. [DOI | PubMed]
- Y.Y. Qu, L.M. Wang, H. Zhong. TRPC1 stimulates calcium-sensing receptor-induced store-operated Ca2+ entry and nitric oxide production in endothelial cells. Mol Med Rep, 2017. [DOI | PubMed]
- M. Zergane, W.M. Kuebler, L. Michalick. Heteromeric TRP channels in lung inflammation. Cells, 2021. [DOI | PubMed]
- M. Malczyk, C. Veith, B. Fuchs. Classical transient receptor potential channel 1 in hypoxia-induced pulmonary hypertension. Am J Respir Crit Care Med, 2013. [DOI | PubMed]
- K. Malkmus, M. Brosien, F. Knoepp. Deletion of classical transient receptor potential 1, 3 and 6 alters pulmonary vasoconstriction in chronic hypoxia-induced pulmonary hypertension in mice. Front Physiol, 2022. [DOI]
- V. Nascimento Da Conceicao, Y. Sun, K. Ramachandran. Resolving macrophage polarization through distinct Ca(2+) entry channel that maintains intracellular signaling and mitochondrial bioenergetics. iScience, 2021. [DOI]
- O.M. Elzamzamy, R. Penner, L.A. Hazlehurst. The role of TRPC1 in modulating cancer progression. Cells, 2020. [DOI | PubMed]
- M. Onopiuk, B. Eby, V. Nesin. Control of PTH secretion by the TRPC1 ion channel. JCI Insight, 2020. [DOI]
- E.R. Liman, C. Dulac. TRP Ion Channel Function in Sensory Transduction and Cellular Signaling Cascades, 2007
- N. Hecker, U. Lächele, H. Stuckas, P. Giere, M. Hiller. Convergent vomeronasal system reduction in mammals coincides with convergent losses of calcium signalling and odorant-degrading genes. Mol Ecol, 2019. [DOI | PubMed]
- T. Hofmann, M. Schaefer, G. Schultz, T. Gudermann. Cloning, expression and subcellular localization of two novel splice variants of mouse transient receptor potential channel 2. Biochem J, 2000. [DOI | PubMed]
- D.R. Pfau, S. Baribeau, F. Brown, N. Khetarpal, S. Marc Breedlove, C.L. Jordan. Loss of TRPC2 function in mice alters sex differences in brain regions regulating social behaviors. J Comp Neurol, 2023. [DOI | PubMed]
- B.G. Leypold, C.R. Yu, T. Leinders-Zufall, M.M. Kim, F. Zufall, R. Axel. Altered sexual and social behaviors in trp2 mutant mice. Proc Natl Acad Sci U S A, 2002. [DOI | PubMed]
- L. Stowers, T.E. Holy, M. Meister, C. Dulac, G. Koentges. Loss of sex discrimination and male-male aggression in mice deficient for TRP2. Science, 2002. [DOI | PubMed]
- Y. Beny-Shefer, N. Zilkha, Y. Lavi-Avnon. Nucleus accumbens dopamine signaling regulates sexual preference for females in male mice. Cell Rep, 2017. [DOI | PubMed]
- T. Kimchi, J. Xu, C. Dulac. A functional circuit underlying male sexual behaviour in the female mouse brain. Nature, 2007. [DOI | PubMed]
- K. Tornquist, P. Sukumaran, K. Kemppainen, C. Lof, T. Viitanen. Canonical transient receptor potential channel 2 (TRPC2): old name-new games. Importance in regulating of rat thyroid cell physiology. Pflugers Arch, 2014. [DOI | PubMed]
- M.K. Jungnickel, H. Marrero, L. Birnbaumer, J.R. Lemos, H.M. Florman. Trp2 regulates entry of Ca2+ into mouse sperm triggered by egg ZP3. Nat Cell Biol, 2001. [DOI | PubMed]
- I. Hirschler-Laszkiewicz, W. Zhang, K. Keefer. Trpc2 depletion protects red blood cells from oxidative stress-induced hemolysis. Exp Hematol, 2012. [DOI | PubMed]
- B. Vannier, M. Peyton, G. Boulay. Mouse trp2, the homologue of the human trpc2 pseudogene, encodes mTrp2, a store depletion-activated capacitative Ca2+ entry channel. Proc Natl Acad Sci U S A, 1999. [DOI | PubMed]
- X. Zhu, M. Jiang, M. Peyton. TRP, a novel mammalian gene family essential for agonist-activated capacitative Ca2+ entry. Cell, 1996. [DOI | PubMed]
- J. Hartmann, E. Dragicevic, H. Adelsberger. TRPC3 channels are required for synaptic transmission and motor coordination. Neuron, 2008. [DOI | PubMed]
- A. Riccio, A.D. Medhurst, C. Mattei. mRNA distribution analysis of human TRPC family in CNS and peripheral tissues. Brain Res Mol Brain Res, 2002. [DOI | PubMed]
- B. Wu, F.G.C. Blot, A.B. Wong. TRPC3 is a major contributor to functional heterogeneity of cerebellar Purkinje cells. eLife, 2019. [DOI]
- M. Goel, C.D. Zuo, W.G. Sinkins, W.P. Schilling. TRPC3 channels colocalize with Na+/Ca2+ exchanger and Na + pump in axial component of transverse-axial tubular system of rat ventricle. Am J Physiol Heart Circ Physiol, 2007. [DOI | PubMed]
- Y. Yu, I. Fantozzi, C.V. Remillard. Enhanced expression of transient receptor potential channels in idiopathic pulmonary arterial hypertension. Proc Natl Acad Sci U S A, 2004. [DOI | PubMed]
- B. Masson, A. Saint-Martin Willer, M. Dutheil. Contribution of transient receptor potential canonical channels in human and experimental pulmonary arterial hypertension. Am J Physiol Lung Cell Mol Physiol, 2023. [DOI | PubMed]
- S.M. Hanson, M.S. Sansom, E.B. Becker. Modeling suggests TRPC3 hydrogen bonding and not phosphorylation contributes to the ataxia phenotype of the moonwalker mouse. Biochemistry, 2015. [DOI | PubMed]
- E.B.E. Becker, P.L. Oliver, M.D. Glitsch. A point mutation in TRPC3 causes abnormal Purkinje cell development and cerebellar ataxia in moonwalker mice. Proc Natl Acad Sci U S A, 2009. [DOI | PubMed]
- M.R. Dino, F.H. Willard, E. Mugnaini. Distribution of unipolar brush cells and other calretinin immunoreactive components in the mammalian cerebellar cortex. J Neurocytol, 1999. [DOI | PubMed]
- M.R. Dino, R.J. Schuerger, Y. Liu, N.T. Slater, E. Mugnaini. Unipolar brush cell: a potential feedforward excitatory interneuron of the cerebellum. Neuroscience, 2000. [DOI | PubMed]
- G. Sekerkova, J.A. Kim, M.J. Nigro. Early onset of ataxia in moonwalker mice is accompanied by complete ablation of type II unipolar brush cells and Purkinje cell dysfunction. J Neurosci, 2013. [DOI | PubMed]
- M. Goel, W.G. Sinkins, C.D. Zuo, U. Hopfer, W.P. Schilling. Vasopressin-induced membrane trafficking of TRPC3 and AQP2 channels in cells of the rat renal collecting duct. Am J Physiol Renal Physiol, 2007. [DOI | PubMed]
- K. Groschner, S. Hingel, B. Lintschinger. Trp proteins form store-operated cation channels in human vascular endothelial cells. FEBS Lett, 1998. [DOI | PubMed]
- M. Kamouchi, S. Philipp, V. Flockerzi. Properties of heterologously expressed hTRP3 channels in bovine pulmonary artery endothelial cells. J Physiol, 1999. [DOI | PubMed]
- F. Thilo, C. Loddenkemper, E. Berg, W. Zidek, M. Tepel. Increased TRPC3 expression in vascular endothelium of patients with malignant hypertension. Mod Pathol, 2009. [DOI | PubMed]
- K. Smedlund, G. Vazquez. Involvement of native TRPC3 proteins in ATP-dependent expression of VCAM-1 and monocyte adherence in coronary artery endothelial cells. Arterioscler Thromb Vasc Biol, 2008. [DOI | PubMed]
- K.B. Smedlund, L. Birnbaumer, G. Vazquez. Increased size and cellularity of advanced atherosclerotic lesions in mice with endothelial overexpression of the human TRPC3 channel. Proc Natl Acad Sci U S A, 2015. [DOI | PubMed]
- Q. Xi, A. Adebiyi, G. Zhao. IP3 constricts cerebral arteries via IP3 receptor-mediated TRPC3 channel activation and independently of sarcoplasmic reticulum Ca2+ release. Circ Res, 2008. [DOI | PubMed]
- H.W. Park, J.Y. Kim, S.K. Choi. Serine-threonine kinase with-no-lysine 4 (WNK4) controls blood pressure via transient receptor potential canonical 3 (TRPC3) in the vasculature. Proc Natl Acad Sci U S A, 2011. [DOI | PubMed]
- H. Nakayama, B.J. Wilkin, I. Bodi, J.D. Molkentin. Calcineurin-dependent cardiomyopathy is activated by TRPC in the adult mouse heart. FASEB J, 2006. [DOI | PubMed]
- X. Wu, P. Eder, B. Chang, J.D. Molkentin. TRPC channels are necessary mediators of pathologic cardiac hypertrophy. Proc Natl Acad Sci U S A, 2010. [DOI | PubMed]
- J.W. Han, Y.H. Lee, S.I. Yoen. Resistance to pathologic cardiac hypertrophy and reduced expression of CaV1.2 in Trpc3-depleted mice. Mol Cell Biochem, 2016. [DOI | PubMed]
- N. Onohara, M. Nishida, R. Inoue. TRPC3 and TRPC6 are essential for angiotensin II-induced cardiac hypertrophy. EMBO J, 2006. [DOI | PubMed]
- M. Poteser, H. Schleifer, M. Lichtenegger. PKC-dependent coupling of calcium permeation through transient receptor potential canonical 3 (TRPC3) to calcineurin signaling in HL-1 myocytes. Proc Natl Acad Sci USA, 2011. [DOI | PubMed]
- T. Numaga-Tomita, S. Oda, T. Shimauchi, A. Nishimura, S. Mangmool, M. Nishida. TRPC3 Channels in cardiac fibrosis. Front Cardiovasc Med, 2017. [DOI | PubMed]
- G. Rached, Y. Saliba, D. Maddah. TRPC3 regulates islet beta-cell insulin secretion. Adv Sci (Weinh), 2023. [DOI]
- S. Li, K. Jiang, J. Li. Estrogen enhances the proliferation and migration of ovarian cancer cells by activating transient receptor potential channel C3. J Ovarian Res, 2020. [DOI | PubMed]
- N. Hirata, S. Yamada, S. Yanagida. Lysophosphatidic acid promotes the expansion of cancer stem cells via TRPC3 channels in triple-negative breast cancer. Int J Mol Sci, 2022. [DOI | PubMed]
- V. Farfariello, D.V. Gordienko, L. Mesilmany. TRPC3 shapes the ER-mitochondria Ca2+ transfer characterizing tumour-promoting senescence. Nat Commun, 2022. [DOI | PubMed]
- S. Feng, H. Li, Y. Tai. Canonical transient receptor potential 3 channels regulate mitochondrial calcium uptake. Proc Natl Acad Sci U S A, 2013. [DOI | PubMed]
- J. Casas, C. Meana, J.R. López-López, J. Balsinde, M.A. Balboa. Lipin-1-derived diacylglycerol activates intracellular TRPC3 which is critical for inflammatory signaling. Cell Mol Life Sci, 2021. [DOI | PubMed]
- B. Wang, S. Xiong, S. Lin. Enhanced mitochondrial transient receptor potential channel, canonical type 3-mediated calcium handling in the vasculature from hypertensive rats. J Am Heart Assoc, 2017. [DOI]
- M.A. Fowler, K. Sidiropoulou, E.D. Ozkan, C.W. Phillips, D.C. Cooper. Corticolimbic expression of TRPC4 and TRPC5 channels in the rodent brain. PLoS One, 2007. [DOI]
- S. Sours, J. Du, S. Chu, M. Ding, X.J. Zhou, R. Ma. Expression of canonical transient receptor potential (TRPC) proteins in human glomerular mesangial cells. Am J Physiol Renal Physiol, 2006. [DOI | PubMed]
- Y. Zhou, P. Castonguay, E.H. Sidhom. A small-molecule inhibitor of TRPC5 ion channels suppresses progressive kidney disease in animal models. Science, 2017. [DOI | PubMed]
- J.C. Gonzalez-Cobos, M. Trebak. TRPC channels in smooth muscle cells. Front Biosci (Landmark Ed), 2010. [DOI | PubMed]
- S. Negri, P. Faris, R. Berra-Romani, G. Guerra, F. Moccia. Endothelial transient receptor potential channels and vascular remodeling: extracellular Ca2+ entry for angiogenesis, arteriogenesis and vasculogenesis. Front Physiol, 2019. [DOI | PubMed]
- W.D. Klipec, K.R. Burrow, C. O’Neill. Loss of the trpc4 gene is associated with a reduction in cocaine self-administration and reduced spontaneous ventral tegmental area dopamine neuronal activity, without deficits in learning for natural rewards. Behav Brain Res, 2016. [DOI | PubMed]
- Z.C. Sun, S.B. Ma, W.G. Chu, D. Jia, C. Luo. Canonical transient receptor potential (TRPC) channels in nociception and pathological pain. Neural Plast, 2020. [DOI]
- C.F. Cohen, A.S. Prudente, T. Berta, S.H. Lee. Transient receptor potential Channel 4 small-molecule inhibition alleviates migraine-like behavior in mice. Front Mol Neurosci, 2021. [DOI]
- S.H. Lee, R. Tonello, Y. Choi, S.J. Jung, T. Berta. Sensory neuron–expressed TRPC4 is a target for the relief of psoriasiform itch and skin inflammation in mice. J Invest Dermatol, 2020. [DOI | PubMed]
- J. Jeon, F. Bu, G. Sun. Contribution of TRPC channels in neuronal excitotoxicity associated with neurodegenerative disease and ischemic stroke. Front Cell Dev Biol, 2020. [DOI]
- W.-G. Chu, F.-D. Wang, Z.-C. Sun. TRPC1/4/5 channels contribute to morphine-induced analgesic tolerance and hyperalgesia by enhancing spinal synaptic potentiation and structural plasticity. FASEB J, 2020. [DOI | PubMed]
- T. Okada, S. Shimizu, M. Wakamori. Molecular cloning and functional characterization of a novel receptor-activated TRP Ca2+ channel from mouse brain. J Biol Chem, 1998. [DOI | PubMed]
- S. Philipp, J. Hambrecht, L. Braslavski. A novel capacitative calcium entry channel expressed in excitable cells. EMBO J, 1998. [DOI | PubMed]
- J. Bröker-Lai, A. Kollewe, B. Schindeldecker. Heteromeric channels formed by TRPC1, TRPC4 and TRPC5 define hippocampal synaptic transmission and working memory. EMBO J, 2017. [DOI | PubMed]
- A. Greka, B. Navarro, E. Oancea, A. Duggan, D.E. Clapham. TRPC5 is a regulator of hippocampal neurite length and growth cone morphology. Nat Neurosci, 2003. [DOI | PubMed]
- A. Riccio, Y. Li, J. Moon. Essential role for TRPC5 in amygdala function and fear-related behavior. Cell, 2009. [DOI | PubMed]
- A. Riccio, Y. Li, E. Tsvetkov. Decreased anxiety-like behavior and Gαq/11-dependent responses in the amygdala of mice lacking TRPC4 channels. J Neurosci, 2014. [DOI | PubMed]
- H. Wei, B. Sagalajev, M.A. Yüzer, A. Koivisto, A. Pertovaara. Regulation of neuropathic pain behavior by amygdaloid TRPC4/C5 channels. Neurosci Lett, 2015. [DOI | PubMed]
- P. Adhya, B. Vaidya, S.S. Sharma. BTD: a TRPC5 activator ameliorates mechanical allodynia in diabetic peripheral neuropathic rats by modulating TRPC5-CAMKII-ERK pathway. Neurochem Int, 2023. [DOI]
- P. Khare, J. Chand, A. Ptakova. The TRPC5 receptor as pharmacological target for pain and metabolic disease. Pharmacol Ther, 2024. [DOI]
- A.P. Koivisto, T. Voets, M.J. Iadarola, A. Szallasi. Targeting TRP channels for pain relief: a review of current evidence from bench to bedside. Curr Opin Pharmacol, 2024. [DOI]
- C. Tai, D.J. Hines, H.B. Choi, B.A. MacVicar. Plasma membrane insertion of TRPC5 channels contributes to the cholinergic plateau potential in hippocampal CA1 pyramidal neurons. Hippocampus, 2011. [DOI | PubMed]
- M.A. Dichter, G.F. Ayala. Cellular mechanisms of epilepsy: a status report. Science, 1987. [DOI | PubMed]
- D.D. Fraser, B.A. MacVicar. Cholinergic-dependent plateau potential in hippocampal CA1 pyramidal neurons. J Neurosci, 1996. [DOI | PubMed]
- J. Bröker-Lai, J. Rego Terol, C. Richter. TRPC5 controls the adrenaline-mediated counter regulation of hypoglycemia. EMBO J, 2024. [DOI | PubMed]
- E. Carbone. TRPC5: a new entry to the chromaffin cell’s palette of ion channels that control adrenal response to hypoglycemia. EMBO J, 2024. [DOI | PubMed]
- Y. Zhu, M. Gao, T. Zhou. The TRPC5 channel regulates angiogenesis and promotes recovery from ischemic injury in mice. J Biol Chem, 2019. [DOI | PubMed]
- A. Ptakova, V. Vlachova. Thermosensing ability of TRPC5: current knowledge and unsettled questions. J Physiol Sci, 2024. [DOI | PubMed]
- K. Zimmermann, J.K. Lennerz, A. Hein. Transient receptor potential cation channel, subfamily C, member 5 (TRPC5) is a cold-transducer in the peripheral nervous system. Proc Natl Acad Sci U S A, 2011. [DOI | PubMed]
- A. Ptakova, M. Mitro, L. Zimova, V. Vlachova. Cellular context determines primary characteristics of human TRPC5 as a cold-activated channel. J Cell Physiol, 2022. [DOI | PubMed]
- L. Bernal, P. Sotelo-Hitschfeld, C. König. Odontoblast TRPC5 channels signal cold pain in teeth. Sci Adv, 2021. [DOI]
- D. Tian, S.M.P. Jacobo, D. Billing. Antagonistic regulation of actin dynamics and cell motility by TRPC5 and TRPC6 channels. Sci Signal, 2010. [DOI]
- C. Faul, K. Asanuma, E. Yanagida-Asanuma, K. Kim, P. Mundel. Actin up: regulation of podocyte structure and function by components of the actin cytoskeleton. Trends Cell Biol, 2007. [DOI | PubMed]
- T. Takeda, T. McQuistan, R.A. Orlando, M.G. Farquhar. Loss of glomerular foot processes is associated with uncoupling of podocalyxin from the actin cytoskeleton. J Clin Invest, 2001. [DOI | PubMed]
- K. Asanuma, E. Yanagida-Asanuma, C. Faul, Y. Tomino, K. Kim, P. Mundel. Synaptopodin orchestrates actin organization and cell motility via regulation of RhoA signalling. Nat Cell Biol, 2006. [DOI | PubMed]
- X. Ma, Y. Cai, D. He. Transient receptor potential channel TRPC5 is essential for P-glycoprotein induction in drug-resistant cancer cells. Proc Natl Acad Sci U S A, 2012. [DOI | PubMed]
- X. Ma, Z. Chen, D. Hua. Essential role for TrpC5-containing extracellular vesicles in breast cancer with chemotherapeutic resistance. Proc Natl Acad Sci U S A, 2014. [DOI | PubMed]
- T. Wang, Z. Chen, Y. Zhu. Inhibition of transient receptor potential channel 5 reverses 5-fluorouracil resistance in human colorectal cancer cells. J Biol Chem, 2015. [DOI | PubMed]
- O.C. Lau, B. Shen, C.O. Wong. TRPC5 channels participate in pressure-sensing in aortic baroreceptors. Nat Commun, 2016. [DOI]
- C. Liang, Y. Zhang, D. Zhuo. Endothelial cell transient receptor potential channel C5 (TRPC5) is essential for endothelium-dependent contraction in mouse carotid arteries. Biochem Pharmacol, 2019. [DOI | PubMed]
- Y. Chu, S. Wang, Y. Zhu, F. Yu, K. Zhang, X. Ma. TRPC5 mediates endothelium-dependent contraction in the carotid artery of diet-induced obese mice. Hypertens Res, 2022. [DOI | PubMed]
- B. Nilius, G. Owsianik. The transient receptor potential family of ion channels. Genome Biol, 2011. [DOI | PubMed]
- T.K. Bardell, E.L. Barker. Activation of TRPC6 channels promotes endocannabinoid biosynthesis in neuronal CAD cells. Neurochem Int, 2010. [DOI | PubMed]
- Y. Tai, S. Feng, R. Ge. TRPC6 channels promote dendritic growth via the CaMKIV-CREB pathway. J Cell Sci, 2008. [DOI | PubMed]
- J.H. Heiser, A.M. Schuwald, G. Sillani, L. Ye, W.E. Müller, K. Leuner. TRPC6 channel-mediated neurite outgrowth in PC12 cells and hippocampal neurons involves activation of RAS/MEK/ERK, PI3K, and CAMKIV signaling. J Neurochem, 2013. [DOI | PubMed]
- M.A. Calupca, S.A. Locknar, R.L. Parsons. TRPC6 immunoreactivity is colocalized with neuronal nitric oxide synthase in extrinsic fibers innervating guinea pig intrinsic cardiac ganglia. J Comp Neurol, 2002. [DOI | PubMed]
- R. Elsaesser, G. Montani, R. Tirindelli, J. Paysan. Phosphatidyl-inositide signalling proteins in a novel class of sensory cells in the mammalian olfactory epithelium. Eur J Neurosci, 2005. [DOI | PubMed]
- E.J. Warren, C.N. Allen, R.L. Brown, D.W. Robinson. The light-activated signaling pathway in SCN-projecting rat retinal ganglion cells. Eur J Neurosci, 2006. [DOI | PubMed]
- Y. Sun, P. Sukumaran, B.C. Bandyopadhyay, B.B. Singh. Physiological function and characterization of TRPCs in neurons. Cells, 2014. [DOI | PubMed]
- A.J. Hill, J.M. Hinton, H. Cheng. A TRPC-like non-selective cation current activated by alpha 1-adrenoceptors in rat mesenteric artery smooth muscle cells. Cell Calcium, 2006. [DOI | PubMed]
- Q. Wang, D. Wang, G. Yan, L. Sun, C. Tang. TRPC6 is required for hypoxia-induced basal intracellular calcium concentration elevation, and for the proliferation and migration of rat distal pulmonary venous smooth muscle cells. Mol Med Rep, 2016. [DOI | PubMed]
- Y. Yu, M. Sweeney, S. Zhang. PDGF stimulates pulmonary vascular smooth muscle cell proliferation by upregulating TRPC6 expression. Am J Physiol Cell Physiol, 2003. [DOI | PubMed]
- M.A. Spassova, T. Hewavitharana, W. Xu, J. Soboloff, D.L. Gill. A common mechanism underlies stretch activation and receptor activation of TRPC6 channels. Proc Natl Acad Sci U S A, 2006. [DOI | PubMed]
- M. Mederos y Schnitzler, U. Storch, S. Meibers. Gq-coupled receptors as mechanosensors mediating myogenic vasoconstriction. EMBO J, 2008. [DOI | PubMed]
- C.D. Cox, K. Poole, B. Martinac. Re-evaluating TRP channel mechanosensitivity. Trends Biochem Sci, 2024. [DOI | PubMed]
- Y. Matsushita, K. Yoshida, M. Yoshiya. TRPC6 is a mechanosensitive channel essential for ultrasound neuromodulation in the mammalian brain. Proc Natl Acad Sci U S A, 2024. [DOI]
- R. Ge, Y. Tai, Y. Sun. Critical role of TRPC6 channels in VEGF-mediated angiogenesis. Cancer Lett, 2009. [DOI | PubMed]
- M.A. Hamdollah Zadeh, C.A. Glass, A. Magnussen, J.C. Hancox, D.O. Bates. VEGF-mediated elevated intracellular calcium and angiogenesis in human microvascular endothelial cells in vitro are inhibited by dominant negative TRPC6. Microcirculation, 2008. [DOI | PubMed]
- A. Berna-Erro, L. Albarran, N. Dionisio. The canonical transient receptor potential 6 (TRPC6) channel is sensitive to extracellular pH in mouse platelets. Blood Cells Mol Dis, 2014. [DOI | PubMed]
- S. Graham, M. Ding, Y. Ding. Canonical transient receptor potential 6 (TRPC6), a redox-regulated cation channel. J Biol Chem, 2010. [DOI | PubMed]
- M. Goel, W.G. Sinkins, C.D. Zuo, M. Estacion, W.P. Schilling. Identification and localization of TRPC channels in the rat kidney. Am J Physiol Renal Physiol, 2006. [DOI | PubMed]
- X. Liu, X. Yao, S.Y. Tsang. Post-translational modification and natural mutation of TRPC channels. Cells, 2020. [DOI | PubMed]
- A. Staruschenko, R. Ma, O. Palygin, S.E. Dryer. Ion channels and channelopathies in glomeruli. Physiol Rev, 2023. [DOI | PubMed]
- C.N. Englisch, F. Paulsen, T. Tschernig. TRPC Channels in the physiology and pathophysiology of the renal tubular system: what do we know?. Int J Mol Sci, 2022. [DOI | PubMed]
- N. Weissmann, A. Dietrich, B. Fuchs. Classical transient receptor potential channel 6 (TRPC6) is essential for hypoxic pulmonary vasoconstriction and alveolar gas exchange. Proc Natl Acad Sci U S A, 2006. [DOI | PubMed]
- R.L. Corteling, S. Li, J. Giddings, J. Westwick, C. Poll, I.P. Hall. Expression of transient receptor potential C6 and related transient receptor potential family members in human airway smooth muscle and lung tissue. Am J Respir Cell Mol Biol, 2004. [DOI | PubMed]
- N. Weissmann, A. Sydykov, H. Kalwa. Activation of TRPC6 channels is essential for lung ischaemia-reperfusion induced oedema in mice. Nat Commun, 2012. [DOI | PubMed]
- M. Ratnasingham, P. Bradding, K.M. Roach. The role of TRP channels in lung fibrosis: mechanisms and therapeutic potential. Int J Biochem Cell Biol, 2025. [DOI]
- S. Oda, K. Nishiyama, Y. Furumoto. Myocardial TRPC6-mediated Zn2+ influx induces beneficial positive inotropy through β-adrenoceptors. Nat Commun, 2022. [DOI | PubMed]
- A. Dietrich. Is Zn2+ the new Ca2+ for TRPC6 channels in the myocardium?. Cell Calcium, 2023. [DOI]
- M.S. Alavi, V. Soheili, A. Roohbakhsh. The role of transient receptor potential (TRP) channels in phagocytosis: a comprehensive review. Eur J Pharmacol, 2024. [DOI]
- Y. Jang, Y. Lee, S.M. Kim, Y.D. Yang, J. Jung, U. Oh. Quantitative analysis of TRP channel genes in mouse organs. Arch Pharm Res, 2012. [DOI | PubMed]
- H. Yip, W.Y. Chan, P.C. Leung. Expression of TRPC homologs in endothelial cells and smooth muscle layers of human arteries. Histochem Cell Biol, 2004. [DOI | PubMed]
- R.L. Walker, J.R. Hume, B. Horowitz. Differential expression and alternative splicing of TRP channel genes in smooth muscles. Am J Physiol Cell Physiol, 2001. [DOI | PubMed]
- Y. Maruyama, Y. Nakanishi, E.J. Walsh, D.P. Wilson, D.G. Welsh, W.C. Cole. Heteromultimeric TRPC6-TRPC7 channels contribute to arginine vasopressin-induced cation current of A7r5 vascular smooth muscle cells. Circ Res, 2006. [DOI | PubMed]
- F. Ben-Mabrouk, A.K. Tryba. Substance P modulation of TRPC3/7 channels improves respiratory rhythm regularity and ICAN-dependent pacemaker activity. Eur J Neurosci, 2010. [DOI | PubMed]
- K. Miyagi, S. Kiyonaka, K. Yamada. A pathogenic C terminus-truncated polycystin-2 mutant enhances receptor-activated Ca2+ entry via association with TRPC3 and TRPC7. J Biol Chem, 2009. [DOI | PubMed]
- J. Alvarez, A. Coulombe, O. Cazorla. ATP/UTP activate cation-permeable channels with TRPC3/7 properties in rat cardiomyocytes. Am J Physiol Heart Circ Physiol, 2008. [DOI | PubMed]
- S. Satoh, H. Tanaka, Y. Ueda. Transient receptor potential (TRP) protein 7 acts as a G protein-activated Ca2+ channel mediating angiotensin II-induced myocardial apoptosis. Mol Cell Biochem, 2007. [DOI | PubMed]
- T. Xue, M.T. Do, A. Riccio. Melanopsin signalling in mammalian iris and retina. Nature, 2011. [DOI | PubMed]
- C.E. Perez-Leighton, T.M. Schmidt, J. Abramowitz, L. Birnbaumer, P. Kofuji. Intrinsic phototransduction persists in melanopsin-expressing ganglion cells lacking diacylglycerol-sensitive TRPC subunits. Eur J Neurosci, 2011. [DOI | PubMed]
- X. Zhang, Z. Li, H. Nie. The IGF2BP2-lncRNA TRPC7-AS1 axis promotes hepatocellular carcinoma cell proliferation and invasion. Cell Signal, 2024. [DOI]
- S. Zhu, H. Ye, X. Xu. Involvement of TRPC7-AS1 expression in hepatitis B virus-related hepatocellular carcinoma. J Oncol, 2021. [DOI]
- J.L. Liang, M.H. Tsai, Y.C. Hsieh. TRPC7 facilitates cell growth and migration by regulating intracellular Ca2+ mobilization in lung adenocarcinoma cells. Oncol Lett, 2023. [DOI | PubMed]
- J.E. Camacho Londoño, A. Marx, A.E. Kraft. Angiotensin-II-evoked Ca2+ entry in murine cardiac fibroblasts does not depend on TRPC channels. Cells, 2020. [DOI | PubMed]
- K. Formoso, S. Susperreguy, M. Freichel, L. Birnbaumer. RNA-seq analysis reveals TRPC genes to impact an unexpected number of metabolic and regulatory pathways. Sci Rep, 2020. [DOI | PubMed]
- B.L. Fogel, S.M. Hanson, E.B.E. Becker. Do mutations in the murine ataxia gene TRPC3 cause cerebellar ataxia in humans?. Mov Disord, 2015. [DOI | PubMed]
- T. Schaldecker, S. Kim, C. Tarabanis. Inhibition of the TRPC5 ion channel protects the kidney filter. J Clin Invest, 2013. [DOI | PubMed]
- C. Mignon-Ravix, P. Cacciagli, N. Choucair. Intragenic rearrangements in X-linked intellectual deficiency: results of a-CGH in a series of 54 patients and identification of TRPC5 and KLHL15 as potential XLID genes. Am J Med Genet A, 2014. [DOI | PubMed]
- Y. Li, T.M. Cacciottolo, N. Yin. Loss of transient receptor potential channel 5 causes obesity and postpartum depression. Cell, 2024. [DOI | PubMed]
- E. Leitão, C. Schröder, I. Parenti. Systematic analysis and prediction of genes associated with monogenic disorders on human chromosome X. Nat Commun, 2022. [DOI | PubMed]
- M.P. Winn, P.J. Conlon, K.L. Lynn. A mutation in the TRPC6 cation channel causes familial focal segmental glomerulosclerosis. Science, 2005. [DOI | PubMed]
- J. Reiser, K.R. Polu, C.C. Möller. TRPC6 is a glomerular slit diaphragm-associated channel required for normal renal function. Nat Genet, 2005. [DOI | PubMed]
- M. Riehle, A.K. Büscher, B.O. Gohlke. TRPC6 G757D loss-of-function mutation associates with FSGS. J Am Soc Nephrol, 2016. [DOI | PubMed]
- A. Staruschenko, D. Spires, O. Palygin. Role of TRPC6 in progression of diabetic kidney disease. Curr Hypertens Rep, 2019. [DOI | PubMed]
- N. Urban, K. Hill, L. Wang, W.M. Kuebler, M. Schaefer. Novel pharmacological TRPC inhibitors block hypoxia-induced vasoconstriction. Cell Calcium, 2012. [DOI | PubMed]
- Y. Yu, S.H. Keller, C.V. Remillard. A functional single-nucleotide polymorphism in the TRPC6 gene promoter associated with idiopathic pulmonary arterial hypertension. Circulation, 2009. [DOI | PubMed]
- G. Pousada, A. Baloira, D. Valverde. Molecular and clinical analysis of TRPC6 and AGTR1 genes in patients with pulmonary arterial hypertension. Orphanet J Rare Dis, 2015. [DOI | PubMed]
- K. Seo, P.P. Rainer, V. Shalkey Hahn. Combined TRPC3 and TRPC6 blockade by selective small-molecule or genetic deletion inhibits pathological cardiac hypertrophy. Proc Natl Acad Sci U S A, 2014. [DOI | PubMed]
- B.L. Lin, D. Matera, J.F. Doerner. In vivo selective inhibition of TRPC6 by antagonist BI 749327 ameliorates fibrosis and dysfunction in cardiac and renal disease. Proc Natl Acad Sci U S A, 2019. [DOI | PubMed]
- N. Norton, J.E. Crook, L. Wang. Association of genetic variants at TRPC6 with chemotherapy-related heart failure. Front Cardiovasc Med, 2020. [DOI | PubMed]
- M. Miller, J. Shi, Y. Zhu. Identification of ML204, a novel potent antagonist that selectively modulates native TRPC4/C5 ion channels. J Biol Chem, 2011. [DOI | PubMed]
- J.M. Richter, M. Schaefer, K. Hill. Clemizole hydrochloride is a novel and potent inhibitor of transient receptor potential channel TRPC5. Mol Pharmacol, 2014. [DOI | PubMed]
- L. Zimova, A. Ptakova, M. Mitro, J. Krusek, V. Vlachova. Activity dependent inhibition of TRPC1/4/5 channels by duloxetine involves voltage sensor-like domain. Biomed Pharmacother, 2022. [DOI]
- H.N. Rubaiy, M.J. Ludlow, M. Henrot. Picomolar, selective, and subtype-specific small-molecule inhibition of TRPC1/4/5 channels. J Biol Chem, 2017. [DOI | PubMed]
- S. Just, B.L. Chenard, A. Ceci. Treatment with HC-070, a potent inhibitor of TRPC4 and TRPC5, leads to anxiolytic and antidepressant effects in mice. PLoS One, 2018. [DOI]
- M. Yu, M.W. Ledeboer, M. Daniels. Discovery of a potent and selective TRPC5 inhibitor, efficacious in a focal segmental glomerulosclerosis model. ACS Med Chem Lett, 2019. [DOI | PubMed]
- J.M. Richter, M. Schaefer, K. Hill. Riluzole activates TRPC5 channels independently of PLC activity. Br J Pharmacol, 2014. [DOI | PubMed]
- H. Beckmann, J. Richter, K. Hill, N. Urban, H. Lemoine, M. Schaefer. A benzothiadiazine derivative and methylprednisolone are novel and selective activators of transient receptor potential canonical 5 (TRPC5) channels. Cell Calcium, 2017. [DOI | PubMed]
- Y. Akbulut, H.J. Gaunt, K. Muraki. (-)-Englerin A is a potent and selective activator of TRPC4 and TRPC5 calcium channels. Angew Chem Int Ed Engl, 2015. [DOI | PubMed]
- A. Minard, C.C. Bauer, E. Chuntharpursat-Bon. Potent, selective, and subunit-dependent activation of TRPC5 channels by a xanthine derivative. Br J Pharmacol, 2019. [DOI | PubMed]
- L. Walsh, J.F. Reilly, C. Cornwall. Safety and efficacy of GFB-887, a TRPC5 channel inhibitor, in patients with focal segmental glomerulosclerosis, treatment-resistant minimal change disease, or diabetic nephropathy: TRACTION-2 trial design. Kidney Int Rep, 2021. [DOI | PubMed]
- S. Grimm, C. Keicher, C. Paret. The effects of transient receptor potential cation channel inhibition by BI 1358894 on cortico-limbic brain reactivity to negative emotional stimuli in major depressive disorder. Eur Neuropsychopharmacol, 2022. [DOI | PubMed]
- S. Kiyonaka, K. Kato, M. Nishida. Selective and direct inhibition of TRPC3 channels underlies biological activities of a pyrazole compound. Proc Natl Acad Sci U S A, 2009. [DOI | PubMed]
- H. Schleifer, B. Doleschal, M. Lichtenegger. Novel pyrazole compounds for pharmacological discrimination between receptor-operated and store-operated Ca(2+) entry pathways. Br J Pharmacol, 2012. [DOI | PubMed]
- S. Zhang, L.O. Romero, S. Deng. Discovery of a highly selective and potent TRPC3 inhibitor with high metabolic stability and low toxicity. ACS Med Chem Lett, 2021. [DOI | PubMed]
- J. Wang, S. Zhang, V.K. Boda. Discovery of a potent and selective TRPC3 antagonist with neuroprotective effects. Bioorg Med Chem, 2025. [DOI]
- D.G. Washburn, D.A. Holt, J. Dodson. The discovery of potent blockers of the canonical transient receptor channels, TRPC3 and TRPC6, based on an anilino-thiazole pharmacophore. Bioorg Med Chem Lett, 2013. [DOI | PubMed]
- T. Maier, M. Follmann, G. Hessler. Discovery and pharmacological characterization of a novel potent inhibitor of diacylglycerol-sensitive TRPC cation channels. Br J Pharmacol, 2015. [DOI | PubMed]
- N. Urban, L. Wang, S. Kwiek, J. Rademann, W.M. Kuebler, M. Schaefer. Identification and validation of Larixyl acetate as a potent TRPC6 inhibitor. Mol Pharmacol, 2016. [DOI | PubMed]
- S. Häfner, F. Burg, M. Kannler. A (+)-larixol congener with high affinity and subtype selectivity toward TRPC6. ChemMedChem, 2018. [DOI | PubMed]
- K. Motoyama, T. Nagata, J. Kobayashi. Discovery of a bicyclo[4.3.0]nonane derivative DS88790512 as a potent, selective, and orally bioavailable blocker of transient receptor potential canonical 6 (TRPC6). Bioorg Med Chem Lett, 2018. [DOI | PubMed]
- L.B. Ware, N. Soleymanlou, D.F. McAuley. TRPC6 inhibitor (BI 764198) to reduce risk and severity of ARDS due to COVID-19: a phase II randomised controlled trial. Thorax, 2023. [DOI | PubMed]
- S. Sawamura, M. Hatano, Y. Takada. Screening of transient receptor potential canonical channel activators identifies novel neurotrophic piperazine compounds. Mol Pharmacol, 2016. [DOI | PubMed]
- GGd de la Cruz, B. Svobodova, M. Lichtenegger, O. Tiapko, K. Groschner, T. Glasnov. Intensified microwave-assisted N-acylation procedure – Synthesis and activity evaluation of TRPC3 channel agonists with a 1,3-Dihydro-2H-benzo[d]imidazol-2-one core. Synlett, 2017. [DOI | PubMed]
- C. Qu, M. Ding, Y. Zhu. Pyrazolopyrimidines as potent stimulators for transient receptor potential canonical 3/6/7 channels. J Med Chem, 2017. [DOI | PubMed]
- N. Urban, M. Schaefer. Direct activation of TRPC3 channels by the antimalarial agent artemisinin. Cells, 2020. [DOI | PubMed]
- P.-L. Yang, X.-H. Li, J. Wang. GSK1702934A and M085 directly activate TRPC6 via a mechanism of stimulating the extracellular cavity formed by the pore helix and transmembrane helix S6. J Biol Chem, 2021. [DOI]
- S. Häfner, N. Urban, M. Schaefer. Discovery and characterization of a positive allosteric modulator of transient receptor potential canonical 6 (TRPC6) channels. Cell Calcium, 2019. [DOI | PubMed]
- J.A. Frank, D.A. Yushchenko, D.J. Hodson. Photoswitchable diacylglycerols enable optical control of protein kinase C. Nat Chem Biol, 2016. [DOI | PubMed]
- T. Leinders-Zufall, U. Storch, K. Bleymehl. PhoDAGs enable optical control of diacylglycerol-sensitive transient receptor potential channels. Cell Chem Biol, 2018. [DOI | PubMed]
- M. Keck, C. Hermann, K. Lützel. Photoswitchable TRPC6 channel activators evoke distinct channel kinetics reflecting different gating behaviors. iScience, 2024. [DOI]
- O. Tiapko, N. Shrestha, S. Lindinger. Lipid-independent control of endothelial and neuronal TRPC3 channels by light. Chem Sci, 2019. [DOI | PubMed]
- M. Müller, K. Niemeyer, N. Urban. BTDAzo: a photoswitchable TRPC5 channel activator. Angew Chem Int Ed Engl, 2022. [DOI]
- K. Lützel, H. Laqua, M.B. Sathian. A platform for the development of highly red-shifted azobenzene-based optical tools. Angew Chem Int Ed Engl, 2025. [DOI]
- O.K. Polat, E. Isaeva, Y.R. Sudhini. The small GTPase regulatory protein Rac1 drives podocyte injury independent of cationic channel protein TRPC5. Kidney Int, 2023. [DOI | PubMed]
- B. Vaidya, I. Roy, S.S. Sharma. Neuroprotective potential of HC070, a potent TRPC5 channel inhibitor in Parkinson’s disease models: a behavioral and mechanistic study. ACS Chem Neurosci, 2022. [DOI | PubMed]
- B. Vaidya, P. Gupta, J.K. Laha, I. Roy, S.S. Sharma. Amelioration of Parkinson’s disease by pharmacological inhibition and knockdown of redox sensitive TRPC5 channels: focus on mitochondrial health. Life Sci, 2023. [DOI]
- M. Jia, W. Liu, K. Zhang. Larixyl acetate, a TRPC6 inhibitor, attenuates pressure overload-induced heart failure in mice. Mol Med Rep, 2024. [DOI | PubMed]
- Z. Zheng, Y. Xu, U. Krügel. In vivo inhibition of TRPC6 by SH045 attenuates renal fibrosis in a New Zealand obese (NZO) mouse model of metabolic syndrome. Int J Mol Sci, 2022. [DOI | PubMed]
- B.L. Lin, J.Y. Shin, W.P. Jeffreys. Pharmacological TRPC6 inhibition improves survival and muscle function in mice with Duchenne muscular dystrophy. JCI Insight, 2022. [DOI]
- C. Carson, P. Raman, J. Tullai. Englerin A agonizes the TRPC4/C5 cation channels to inhibit tumor cell line proliferation. PLoS One, 2015. [DOI]
- T. Leinders-Zufall, U. Storch, M. Mederos Y Schnitzler. A diacylglycerol photoswitching protocol for studying TRPC channel functions in mammalian cells and tissue slices. Star Protoc, 2021. [DOI]
- R. Fuertig, M. Goettel, L. Herich, J. Hoefler, S.T. Wiebe, V. Sharma. Effects of single and multiple ascending doses of BI 1358894 in healthy male volunteers on safety, tolerability and pharmacokinetics: two phase I partially randomised studies. CNS Drugs, 2023. [DOI | PubMed]
- J.B. Dwyer, C. Schmahl, M. Makinodan. Efficacy and safety of BI 1358894 in patients with borderline personality disorder: results of a phase 2 randomized, placebo-controlled, parallel group dose-ranging trial. J Clin Psychiatry, 2025. [DOI]
- C. Reist, P. Li, T. Le Nguyen, S.D. Süssmuth. Safety of BI 1358894 in patients with major depressive disorder: results and learnings from a phase II randomized decentralized clinical trial. Clin Transl Sci, 2024. [DOI]
- H. Trachtman, M. Kretzler, H.E. Desmond, W. Choi, R.C. Manuel, N. Soleymanlou. TRPC6 inhibitor BI 764198 in focal segmental glomerulosclerosis: phase 2 study design. Kidney Int Rep, 2023. [DOI | PubMed]
- R. Vennekens, G. Owsianik, B. Nilius. Vanilloid transient receptor potential cation channels: an overview. Curr Pharm Des, 2008. [DOI | PubMed]
- K. Castillo, I. Diaz-Franulic, J. Canan, F. Gonzalez-Nilo, R. Latorre. Thermally activated TRP channels: molecular sensors for temperature detection. Phys Biol, 2018. [DOI]
- R. Sánchez-Hernández, M. Benítez-Angeles, A.M. Hernández-Vega, T. Rosenbaum. Recent advances on the structure and the function relationships of the TRPV4 ion channel. Channels, 2024. [DOI]
- P. Yuan. Structural biology of thermoTRPV channels. Cell Calcium, 2019. [DOI]
- M. Benítez-Angeles, S.L. Morales-Lázaro, E. Juárez-González, T. Rosenbaum. TRPV1: structure, endogenous agonists, and mechanisms. Int J Mol Sci, 2020. [DOI | PubMed]
- M.K. van Goor, L. De Jager, Y. Cheng, J. van der Wijst. High-resolution structures of transient receptor potential vanilloid channels: unveiling a functionally diverse group of ion channels. Protein Sci, 2020. [DOI | PubMed]
- M.V. Yelshanskaya, K.D. Nadezhdin, M.G. Kurnikova, A.I. Sobolevsky. Structure and function of the calcium-selective TRP channel TRPV6. J Physiol, 2021. [DOI | PubMed]
- T. Rohacs, E.C. Fluck, JJd De Jesús-Pérez, V.Y. Moiseenkova-Bell. What structures did, and did not, reveal about the function of the epithelial Ca2+ channels TRPV5 and TRPV6. Cell Calcium, 2022. [DOI]
- M.V. Yelshanskaya, A.I. Sobolevsky. Ligand-binding sites in vanilloid-subtype TRP channels. Front Pharmacol, 2022. [DOI]
- A.P. Kalinovskii, L.L. Utkina, Y.V. Korolkova, Y.A. Andreev. TRPV, Ion C. TRPV3 ion channel: from gene to pharmacology. Int J Mol Sci, 2023. [DOI | PubMed]
- J. Lei, M. Tominaga. Unlocking the therapeutic potential of TRPV3: insights into thermosensation, channel modulation, and skin homeostasis involving TRPV3. Bioessays, 2024. [DOI]
- A. Neuberger, A.I. Sobolevsky. Molecular pharmacology of the onco-TRP channel TRPV6. Channels, 2023. [DOI]
- M. Liao, E. Cao, D. Julius, Y. Cheng. Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature, 2013. [DOI | PubMed]
- A.K. Singh, L.L. McGoldrick, A.I. Sobolevsky. Structure and gating mechanism of the transient receptor potential channel TRPV3. Nat Struct Mol Biol, 2018. [DOI | PubMed]
- S.E. Jordt, M. Tominaga, D. Julius. Acid potentiation of the capsaicin receptor determined by a key extracellular site. Proc Natl Acad Sci U S A, 2000. [DOI | PubMed]
- K. Zhang, D. Julius, Y. Cheng. Structural snapshots of TRPV1 reveal mechanism of polymodal functionality. Cell, 2021. [DOI | PubMed]
- 362.Saotome K, Singh AK, Sobolevsky AI. Calcium entry channels in non-excitable cells: determining the crystal structure of TRPV6. 2018.
- K. Saotome, A.K. Singh, M.V. Yelshanskaya, A.I. Sobolevsky. Crystal structure of the epithelial calcium channel TRPV6. Nature, 2016. [DOI | PubMed]
- C.M. Ives, N.J. Thomson, U. Zachariae. A cooperative knock-on mechanism underpins Ca2+-selective cation permeation in TRPV channels. J Gen Physiol, 2023. [DOI]
- C. García-Martínez, C. Morenilla-Palao, R. Planells-Cases, J.M. Merino, A. Ferrer-Montiel. Identification of an aspartic residue in the P-loop of the vanilloid receptor that modulates pore properties. J Biol Chem, 2000. [DOI | PubMed]
- T. Voets, J. Prenen, J. Vriens. Molecular determinants of permeation through the cation channel TRPV4. J Biol Chem, 2002. [DOI | PubMed]
- R.A. Pumroy, JJd De Jesús-Pérez, A.D. Protopopova. Molecular details of ruthenium red pore block in TRPV channels. EMBO Rep, 2024. [DOI | PubMed]
- L.L. McGoldrick, A.K. Singh, K. Saotome. Opening of the human epithelial calcium channel TRPV6. Nature, 2018. [DOI | PubMed]
- N. Hellwig, T.D. Plant, W. Janson, M. Schäfer, G. Schultz, M. Schaefer. TRPV1 acts as proton channel to induce acidification in nociceptive neurons. J Biol Chem, 2004. [DOI | PubMed]
- M.-K. Chung, A.D. Güler, M.J. Caterina. Biphasic currents evoked by chemical or thermal activation of the heat-gated ion channel, TRPV3. J Biol Chem, 2005. [DOI | PubMed]
- A.M. Binshtok, B.P. Bean, C.J. Woolf. Inhibition of nociceptors by TRPV1-mediated entry of impermeant sodium channel blockers. Nature, 2007. [DOI | PubMed]
- L. Zubcevic, S. Le, H. Yang, S.-Y. Lee. Conformational plasticity in the selectivity filter of the TRPV2 ion channel. Nat Struct Mol Biol, 2018. [DOI | PubMed]
- Y. Gao, E. Cao, D. Julius, Y. Cheng. TRPV1 structures in nanodiscs reveal mechanisms of ligand and lipid action. Nature, 2016. [DOI | PubMed]
- Z. Deng, G. Maksaev, M. Rau. Gating of human TRPV3 in a lipid bilayer. Nat Struct Mol Biol, 2020. [DOI | PubMed]
- R.A. Pumroy, A.D. Protopopova, T.C. Fricke. Structural insights into TRPV2 activation by small molecules. Nat Commun, 2022. [DOI | PubMed]
- K.D. Nadezhdin, A. Neuberger, L.S. Khosrof. TRPV3 activation by different agonists accompanied by lipid dissociation from the vanilloid site. Sci Adv, 2024. [DOI]
- J.G.J. Hoenderop, T. Voets, S. Hoefs. Homo- and heterotetrameric architecture of the epithelial Ca2+ channels TRPV5 and TRPV6. EMBO J, 2003. [DOI | PubMed]
- N. Hellwig, N. Albrecht, C. Harteneck, G. Schultz, M. Schaefer. Homo- and heteromeric assembly of TRPV channel subunits. J Cell Sci, 2005. [DOI | PubMed]
- A.R. Rutter, Q.-P. Ma, M. Leveridge, T.P. Bonnert. Heteromerization and colocalization of TrpV1 and TrpV2 in mammalian cell lines and rat dorsal root ganglia. NeuroReport, 2005. [DOI | PubMed]
- W. Cheng, F. Yang, C.L. Takanishi, J. Zheng. Thermosensitive TRPV channel subunits Coassemble into heteromeric channels with intermediate conductance and gating properties. J Gen Physiol, 2007. [DOI | PubMed]
- W. Cheng, F. Yang, S. Liu. Heteromeric heat-sensitive transient receptor potential channels exhibit distinct temperature and chemical response. J Biol Chem, 2012. [DOI | PubMed]
- F. Hu, X. Cao, C. Niu, K. Wang. Coassembly of warm temperature-sensitive transient receptor potential vanilloid (TRPV) 3 and TRPV4 channel complexes with distinct functional properties. Mol Pharmacol, 2022. [DOI | PubMed]
- M.J.M. Fischer, D. Balasuriya, P. Jeggle. Direct evidence for functional TRPV1/TRPA1 heteromers. Pflugers Arch, 2014. [DOI | PubMed]
- A.P. Stewart, G.D. Smith, R.N. Sandford, J.M. Edwardson. Atomic force microscopy reveals the alternating subunit arrangement of the TRPP2-TRPV4 heterotetramer. Biophys J, 2010. [DOI | PubMed]
- X. Ma, K.-T. Cheng, C.-O. Wong. Heteromeric TRPV4-C1 channels contribute to store-operated Ca(2+) entry in vascular endothelial cells. Cell Calcium, 2011. [DOI | PubMed]
- J. Du, X. Ma, B. Shen, Y. Huang, L. Birnbaumer, X. Yao. TRPV4, TRPC1, and TRPP2 assemble to form a flow-sensitive heteromeric channel. FASEB J, 2014. [DOI | PubMed]
- Z. Guo, C. Grimm, L. Becker, A.J. Ricci, S. Heller. A novel ion channel formed by interaction of TRPML3 with TRPV5. PLoS One, 2013. [DOI]
- S. Lansky, J.M. Betancourt, J. Zhang. A pentameric TRPV3 channel with a dilated pore. Nature, 2023. [DOI | PubMed]
- A. Neuberger, A.I. Sobolevsky. Pentameric TRPV3: an artifact or a clue to channel function?. Cell Calcium, 2023. [DOI]
- H. Min, W.-H. Cho, H. Lee. Association of TRPV1 and TLR4 through the TIR domain potentiates TRPV1 activity by blocking activation-induced desensitization. Mol Pain, 2018. [DOI]
- Y. Wang, X. Mo, C. Ping. Site-specific contacts enable distinct modes of TRPV1 regulation by the potassium channel Kvβ1 subunit. J Biol Chem, 2020. [DOI | PubMed]
- Y. Takayama, M. Tominaga. Interaction between TRP channels and anoctamins. Cell Calcium, 2024. [DOI]
- X. Cheng, J. Jin, L. Hu. TRP channel regulates EGFR signaling in hair morphogenesis and skin barrier formation. Cell, 2010. [DOI | PubMed]
- J. Lei, R.U. Yoshimoto, T. Matsui, M. Amagai, M.A. Kido, M. Tominaga. Involvement of skin TRPV3 in temperature detection regulated by TMEM79 in mice. Nat Commun, 2023. [DOI | PubMed]
- P. Doñate-Macián, J. Enrich-Bengoa, I.R. Dégano, D.G. Quintana, A. Perálvarez-Marín. Trafficking of stretch-regulated TRPV2 and TRPV4 channels inferred through Interactomics. Biomolecules, 2019. [DOI | PubMed]
- D.H. Kwon, F. Zhang, B.A. McCray. TRPV4-Rho GTPase complex structures reveal mechanisms of gating and disease. Nat Commun, 2023. [DOI | PubMed]
- K.D. Nadezhdin, I.A. Talyzina, A. Parthasarathy, A. Neuberger, D.X. Zhang, A.I. Sobolevsky. Structure of human TRPV4 in complex with GTPase RhoA. Nat Commun, 2023. [DOI | PubMed]
- Q. Chang, S. Hoefs, A.W. van der Kemp, C.N. Topala, R.J. Bindels, J.G. Hoenderop. The beta-glucuronidase klotho hydrolyzes and activates the TRPV5 channel. Science, 2005. [DOI | PubMed]
- T.T. Lambers, F. Mahieu, E. Oancea. Calbindin-D28K dynamically controls TRPV5-mediated Ca2+ transport. EMBO J, 2006. [DOI | PubMed]
- O. Andrukhova, A. Smorodchenko, M. Egerbacher. FGF23 promotes renal calcium reabsorption through the TRPV5 channel. EMBO J, 2014. [DOI | PubMed]
- M. Palmada, S. Poppendieck, H.M. Embark. Requirement of PDZ domains for the stimulation of the epithelial Ca2+ channel TRPV5 by the NHE regulating factor NHERF2 and the serum and glucocorticoid inducible kinase SGK1. Cell Physiol Biochem, 2005. [DOI | PubMed]
- J. Lee, K.D. Ju, H.J. Kim. Soluble α-klotho anchors TRPV5 to the distal tubular cell membrane independent of FGFR1 by binding TRPV5 and galectin-1 simultaneously. Am J Physiol Renal Physiol, 2021. [DOI | PubMed]
- S.F.J. van de Graaf, Q. Chang, A.R. Mensenkamp, J.G.J. Hoenderop, R.J.M. Bindels. Direct interaction with Rab11a targets the epithelial Ca2+ channels TRPV5 and TRPV6 to the plasma membrane. Mol Cell Biol, 2006. [DOI | PubMed]
- S.F.J. van de Graaf, J.G.J. Hoenderop, A.W.C.M. van der Kemp, S.M. Gisler, R.J.M. Bindels. Interaction of the epithelial Ca2+ channels TRPV5 and TRPV6 with the intestine- and kidney-enriched PDZ protein NHERF4. Pflugers Arch, 2006. [DOI | PubMed]
- L. Sternfeld, E. Krause, A. Schmid. Tyrosine phosphatase PTP1B interacts with TRPV6 in vivo and plays a role in TRPV6-mediated calcium influx in HEK293 cells. Cell Signal, 2005. [DOI | PubMed]
- T. Voets, G. Droogmans, U. Wissenbach, A. Janssens, V. Flockerzi, B. Nilius. The principle of temperature-dependent gating in cold- and heat-sensitive TRP channels. Nature, 2004. [DOI | PubMed]
- B. Nilius, K. Talavera, G. Owsianik, J. Prenen, G. Droogmans, T. Voets. Gating of TRP channels: a voltage connection?. J Physiol, 2005. [DOI | PubMed]
- H. Xu, I.S. Ramsey, S.A. Kotecha. TRPV3 is a calcium-permeable temperature-sensitive cation channel. Nature, 2002. [DOI | PubMed]
- H. Sawai, K. Kohda, T. Kurahashi. The effect of temporal adaptation to different temperatures and osmolarities on heat response of TRPV4 in cultured cells. J Therm Biol, 2019. [DOI]
- P. Hayes, H.J. Meadows, M.J. Gunthorpe. Cloning and functional expression of a human orthologue of rat vanilloid receptor-1. Pain, 2000. [DOI | PubMed]
- M.J. Caterina, T.A. Rosen, M. Tominaga, A.J. Brake, D. Julius. A capsaicin-receptor homologue with a high threshold for noxious heat. Nature, 1999. [DOI | PubMed]
- M.P. Neeper, Y. Liu, T.L. Hutchinson, Y. Wang, C.M. Flores, N. Qin. Activation properties of heterologously expressed mammalian TRPV2: evidence for species dependence. J Biol Chem, 2007. [DOI | PubMed]
- W.J. Laursen, E.R. Schneider, D.K. Merriman, S.N. Bagriantsev, E.O. Gracheva. Low-cost functional plasticity of TRPV1 supports heat tolerance in squirrels and camels. Proc Natl Acad Sci U S A, 2016. [DOI | PubMed]
- E.O. Gracheva, J.F. Cordero-Morales, J.A. González-Carcacía. Ganglion-specific splicing of TRPV1 underlies infrared sensation in vampire bats. Nature, 2011. [DOI | PubMed]
- M. Ohkita, S. Saito, T. Imagawa, K. Takahashi, M. Tominaga, T. Ohta. Molecular cloning and functional characterization of Xenopus tropicalis frog transient receptor potential vanilloid 1 reveal its functional evolution for heat, acid, and capsaicin sensitivities in terrestrial vertebrates. J Biol Chem, 2012. [DOI | PubMed]
- S. Hori, M. Tateyama, T. Shirai, Y. Kubo, O. Saitoh. Two single-point mutations in ankyrin Repeat one drastically change the threshold temperature of TRPV1. Nat Commun, 2023. [DOI | PubMed]
- M. Tominaga, M.J. Caterina, A.B. Malmberg. The cloned capsaicin receptor integrates multiple pain-producing stimuli. Neuron, 1998. [DOI | PubMed]
- F.N. McNamara, A. Randall, M.J. Gunthorpe. Effects of piperine, the pungent component of black pepper, at the human vanilloid receptor (TRPV1). Br J Pharmacol, 2005. [DOI | PubMed]
- M. Trevisani, D. Smart, M.J. Gunthorpe. Ethanol elicits and potentiates nociceptor responses via the vanilloid receptor-1. Nat Neurosci, 2002. [DOI | PubMed]
- J. Siemens, S. Zhou, R. Piskorowski. Spider toxins activate the capsaicin receptor to produce inflammatory pain. Nature, 2006. [DOI | PubMed]
- C.J. Bohlen, A. Priel, S. Zhou, D. King, J. Siemens, D. Julius. A bivalent tarantula toxin activates the capsaicin receptor, TRPV1, by targeting the outer pore domain. Cell, 2010. [DOI | PubMed]
- S.K. Singh, S.B. Deshpande. Nociceptive vascular reflexes evoked by scorpion venom modulate cardiorespiratory parameters involving vanilloid receptor 1 in anaesthetised rats. Neurosci Lett, 2009. [DOI | PubMed]
- M.A. Hakim, W. Jiang, L. Luo. Scorpion toxin, BmP01, induces pain by targeting TRPV1 channel. Toxins, 2015. [DOI | PubMed]
- P.M. Zygmunt, J. Petersson, D.A. Andersson. Vanilloid receptors on sensory nerves mediate the vasodilator action of anandamide. Nature, 1999. [DOI | PubMed]
- A.M. Peier, A.J. Reeve, D.A. Andersson. A heat-sensitive TRP channel expressed in keratinocytes. Science, 2002. [DOI | PubMed]
- A.S. Klein, A. Tannert, M. Schaefer. Cholesterol sensitises the transient receptor potential channel TRPV3 to lower temperatures and activator concentrations. Cell Calcium, 2014. [DOI | PubMed]
- L.S. Premkumar, G.P. Ahern. Induction of vanilloid receptor channel activity by protein kinase C. Nature, 2000. [DOI | PubMed]
- G. Bhave, W. Zhu, H. Wang, D.J. Brasier, G.S. Oxford, R.W. Gereau. cAMP-dependent protein kinase regulates desensitization of the capsaicin receptor (VR1) by direct phosphorylation. Neuron, 2002. [DOI | PubMed]
- T. Moriyama, T. Higashi, K. Togashi. Sensitization of TRPV1 by EP1 and IP reveals peripheral nociceptive mechanism of prostaglandins. Mol Pain, 2005. [DOI | PubMed]
- I. Vetter, B.D. Wyse, G.R. Monteith, S.J. Roberts-Thomson, P.J. Cabot. The mu opioid agonist morphine modulates potentiation of capsaicin-evoked TRPV1 responses through a cyclic AMP-dependent protein kinase A pathway. Mol Pain, 2006. [DOI | PubMed]
- C. Hanack, M. Moroni, W.C. Lima. GABA blocks pathological but not acute TRPV1 pain signals. Cell, 2015. [DOI | PubMed]
- L.G.B. Ferreira, R.X. Faria. TRPing on the pore phenomenon: what do we know about transient receptor potential ion channel-related pore dilation up to now?. J Bioenerg Biomembr, 2016. [DOI | PubMed]
- M.-K. Chung, A.D. Güler, M.J. Caterina. TRPV1 shows dynamic ionic selectivity during agonist stimulation. Nat Neurosci, 2008. [DOI | PubMed]
- M. Li, G.E.S. Toombes, S.D. Silberberg, K.J. Swartz. Physical basis of apparent pore dilation of ATP-activated P2X receptor channels. Nat Neurosci, 2015. [DOI | PubMed]
- D.H. Kwon, F. Zhang, J.G. Fedor, Y. Suo, S.-Y. Lee. Vanilloid-dependent TRPV1 opening trajectory from cryoEM ensemble analysis. Nat Commun, 2022. [DOI | PubMed]
- R. Vennekens, J.G. Hoenderop, J. Prenen. Permeation and gating properties of the novel epithelial Ca(2+) channel. J Biol Chem, 2000. [DOI | PubMed]
- L. Yue, J.B. Peng, M.A. Hediger, D.E. Clapham. CaT1 manifests the pore properties of the calcium-release-activated calcium channel. Nature, 2001. [DOI | PubMed]
- B. Nilius, R. Vennekens, J. Prenen, J.G. Hoenderop, R.J. Bindels, G. Droogmans. Whole-cell and single channel monovalent cation currents through the novel rabbit epithelial Ca2+ channel ECaC. J Physiol, 2000. [DOI | PubMed]
- J. Lee, S.-K. Cha, T.-J. Sun, C.-L. Huang. PIP2 activates TRPV5 and releases its inhibition by intracellular Mg2+. J Gen Physiol, 2005. [DOI | PubMed]
- B. Thyagarajan, V. Lukacs, T. Rohacs. Hydrolysis of phosphatidylinositol 4,5-bisphosphate mediates calcium-induced inactivation of TRPV6 channels. J Biol Chem, 2008. [DOI | PubMed]
- J.G. Hoenderop, A.W. van der Kemp, A. Hartog. Molecular identification of the apical Ca2+ channel in 1, 25-dihydroxyvitamin D3-responsive epithelia. J Biol Chem, 1999. [DOI | PubMed]
- R. Vennekens, J. Prenen, J.G. Hoenderop, R.J. Bindels, G. Droogmans, B. Nilius. Modulation of the epithelial Ca2+ channel ECaC by extracellular pH. Pflugers Arch, 2001. [DOI | PubMed]
- B.-I. Yeh, Y.K. Kim, W. Jabbar, C.-L. Huang. Conformational changes of pore helix coupled to gating of TRPV5 by protons. EMBO J, 2005. [DOI | PubMed]
- E.C. Fluck, A.T. Yazici, T. Rohacs, V.Y. Moiseenkova-Bell. Structural basis of TRPV5 regulation by physiological and pathophysiological modulators. Cell Rep, 2022. [DOI]
- Td de Groot, N.V. Kovalevskaya, S. Verkaart. Molecular mechanisms of calmodulin action on TRPV5 and modulation by parathyroid hormone. Mol Cell Biol, 2011. [DOI | PubMed]
- N.V. Kovalevskaya, F.M. Bokhovchuk, G.W. Vuister. The TRPV5/6 calcium channels contain multiple calmodulin binding sites with differential binding properties. J Struct Funct Genomics, 2012. [DOI | PubMed]
- T.E.T. Hughes, R.A. Pumroy, A.T. Yazici. Structural insights on TRPV5 gating by endogenous modulators. Nat Commun, 2018. [DOI | PubMed]
- A.K. Singh, L.L. McGoldrick, E.C. Twomey, A.I. Sobolevsky. Mechanism of calmodulin inactivation of the calcium-selective TRP channel TRPV6. Sci Adv, 2018. [DOI]
- S. Dang, M.K. van Goor, D. Asarnow. Structural insight into TRPV5 channel function and modulation. Proc Natl Acad Sci U S A, 2019. [DOI | PubMed]
- C. Cao, E. Zakharian, I. Borbiro, T. Rohacs. Interplay between calmodulin and phosphatidylinositol 4,5-bisphosphate in Ca2+-induced inactivation of transient receptor potential vanilloid 6 channels. J Biol Chem, 2013. [DOI | PubMed]
- T. Voets, A. Janssens, J. Prenen, G. Droogmans, B. Nilius. Mg2+-dependent gating and strong inward rectification of the cation channel TRPV6. J Gen Physiol, 2003. [DOI | PubMed]
- M. Uhlén, L. Fagerberg, B.M. Hallström. Proteomics. Tissue-based map of the human proteome. Science, 2015. [DOI]
- M. Karlsson, C. Zhang, L. Méar. A single-cell type transcriptomics map of human tissues. Sci Adv, 2021. [DOI]
- M.J. Caterina, M.A. Schumacher, M. Tominaga, T.A. Rosen, J.D. Levine, D. Julius. The capsaicin receptor: a heat-activated ion channel in the pain pathway. Nature, 1997. [DOI | PubMed]
- D.N. Cortright, M. Crandall, J.F. Sanchez, T. Zou, J.E. Krause, G. White. The tissue distribution and functional characterization of human VR1. Biochem Biophys Res Commun, 2001. [DOI | PubMed]
- M. Hou, R. Uddman, J. Tajti, M. Kanje, L. Edvinsson. Capsaicin receptor immunoreactivity in the human trigeminal ganglion. Neurosci Lett, 2002. [DOI | PubMed]
- J.B. Davis, J. Gray, M.J. Gunthorpe. Vanilloid receptor-1 is essential for inflammatory thermal hyperalgesia. Nature, 2000. [DOI | PubMed]
- H.H. Chuang, E.D. Prescott, H. Kong. Bradykinin and nerve growth factor release the capsaicin receptor from PtdIns(4,5)P2-mediated inhibition. Nature, 2001. [DOI | PubMed]
- C. Moore, R. Gupta, S.-E. Jordt, Y. Chen, W.B. Liedtke. Regulation of pain and itch by TRP channels. Neurosci Bull, 2018. [DOI | PubMed]
- M. Tognetto, S. Amadesi, S. Harrison. Anandamide excites central terminals of dorsal root ganglion neurons via vanilloid receptor-1 activation. J Neurosci, 2001. [DOI | PubMed]
- A.A. Romanovsky, M.C. Almeida, A. Garami. The transient receptor potential vanilloid-1 channel in thermoregulation: a thermosensor it is not. Pharmacol Rev, 2009. [DOI | PubMed]
- A. Garami, Y.P. Shimansky, Z. Rumbus. Hyperthermia induced by transient receptor potential vanilloid-1 (TRPV1) antagonists in human clinical trials: insights from mathematical modeling and meta-analysis. Pharmacol Ther, 2020. [DOI]
- E. Zhang, P. Liao. Brain transient receptor potential channels and stroke. J Neurosci Res, 2015. [DOI | PubMed]
- C.I. Saunders, D.A. Kunde, A. Crawford, D.P. Geraghty. Expression of transient receptor potential vanilloid 1 (TRPV1) and 2 (TRPV2) in human peripheral blood. Mol Immunol, 2007. [DOI | PubMed]
- I. Kojima, M. Nagasawa. TRPV2. Handb Exp Pharmacol, 2014. [DOI | PubMed]
- G. Santoni, C. Amantini, F. Maggi. The TRPV2 cation channels: from urothelial cancer invasiveness to glioblastoma multiforme interactome signature. Lab Invest, 2020. [DOI | PubMed]
- W. Everaerts, J. Vriens, G. Owsianik. Functional characterization of transient receptor potential channels in mouse urothelial cells. Am J Physiol Renal Physiol, 2010. [DOI | PubMed]
- A. Belkacemi, C.F. Trost, R. Tinschert. The TRPV2 channel mediates Ca2+ influx and the Δ9-THC-dependent decrease in osmotic fragility in red blood cells. Haematologica, 2021. [DOI | PubMed]
- U. Park, N. Vastani, Y. Guan, S.N. Raja, M. Koltzenburg, M.J. Caterina. TRP vanilloid 2 knock-out mice are susceptible to perinatal lethality but display normal thermal and mechanical nociception. J Neurosci, 2011. [DOI | PubMed]
- T.M. Link, U. Park, B.M. Vonakis, D.M. Raben, M.J. Soloski, M.J. Caterina. TRPV2 has a pivotal role in macrophage particle binding and phagocytosis. Nat Immunol, 2010. [DOI | PubMed]
- R. Raudszus, A. Paulig, N. Urban. Pharmacological inhibition of TRPV2 attenuates phagocytosis and lipopolysaccharide-induced migration of primary macrophages. Br J Pharmacol, 2023. [DOI | PubMed]
- C. Li, M. Zhao, X. Liu. Ion channel TRPV2 is critical in enhancing B cell activation and function. J Exp Med, 2024. [DOI]
- Z. Lin, Q. Chen, M. Lee. Exome sequencing reveals mutations in TRPV3 as a cause of Olmsted syndrome. Am J Hum Genet, 2012. [DOI | PubMed]
- G.D. Smith, M.J. Gunthorpe, R.E. Kelsell. TRPV3 is a temperature-sensitive vanilloid receptor-like protein. Nature, 2002. [DOI | PubMed]
- A. Moqrich, S.W. Hwang, T.J. Earley. Impaired thermosensation in mice lacking TRPV3, a heat and camphor sensor in the skin. Science, 2005. [DOI | PubMed]
- S.M. Huang, X. Li, Y. Yu, J. Wang, M.J. Caterina. TRPV3 and TRPV4 ion channels are not major contributors to mouse heat sensation. Mol Pain, 2011. [DOI | PubMed]
- M.-K. Chung, H. Lee, A. Mizuno, M. Suzuki, M.J. Caterina. TRPV3 and TRPV4 mediate warmth-evoked currents in primary mouse keratinocytes. J Biol Chem, 2004. [DOI | PubMed]
- S.M. Huang, H. Lee, M.-K. Chung. Overexpressed transient receptor potential vanilloid 3 ion channels in skin keratinocytes modulate pain sensitivity via prostaglandin E2. J Neurosci, 2008. [DOI | PubMed]
- S. Mandadi, T. Sokabe, K. Shibasaki. TRPV3 in keratinocytes transmits temperature information to sensory neurons via ATP. Pflugers Arch, 2009. [DOI | PubMed]
- T. Miyamoto, M.J. Petrus, A.E. Dubin, A. Patapoutian. TRPV3 regulates nitric oxide synthase-independent nitric oxide synthesis in the skin. Nat Commun, 2011. [DOI | PubMed]
- P. Yang, M.X. Zhu. TRPV3. Handb Exp Pharmacol, 2014. [DOI | PubMed]
- R. Aijima, B. Wang, T. Takao. The thermosensitive TRPV3 channel contributes to rapid wound healing in oral epithelia. FASEB J, 2015. [DOI | PubMed]
- M. Bischof, S. Olthoff, C. Glas, O. Thorn-Seshold, M. Schaefer, K. Hill. TRPV3 endogenously expressed in murine colonic epithelial cells is inhibited by the novel TRPV3 blocker 26E01. Cell Calcium, 2020. [DOI]
- M. Maier, S. Olthoff, K. Hill. KS0365, a novel activator of the transient receptor potential vanilloid 3 (TRPV3) channel, accelerates keratinocyte migration. Br J Pharmacol, 2022. [DOI | PubMed]
- Y. Yamanoi, J. Lei, Y. Takayama, S. Hosogi, Y. Marunaka, M. Tominaga. TRPV3-ANO1 interaction positively regulates wound healing in keratinocytes. Commun Biol, 2023. [DOI | PubMed]
- W. Liedtke, Y. Choe, M.A. Martí-Renom. Vanilloid receptor-related osmotically activated channel (VR-OAC), a candidate vertebrate osmoreceptor. Cell, 2000. [DOI | PubMed]
- R. Strotmann, C. Harteneck, K. Nunnenmacher, G. Schultz, T.D. Plant. OTRPC4, a nonselective cation channel that confers sensitivity to extracellular osmolarity. Nat Cell Biol, 2000. [DOI | PubMed]
- U. Wissenbach, M. Bödding, M. Freichel, V. Flockerzi. Trp12, a novel Trp related protein from kidney. FEBS Lett, 2000. [DOI | PubMed]
- N.S. Delany, M. Hurle, P. Facer. Identification and characterization of a novel human vanilloid receptor-like protein, VRL-2. Physiol Genomics, 2001. [DOI | PubMed]
- H. Watanabe, J. Vriens, J. Prenen, G. Droogmans, T. Voets, B. Nilius. Anandamide and arachidonic acid use epoxyeicosatrienoic acids to activate TRPV4 channels. Nature, 2003. [DOI | PubMed]
- J. Vriens, G. Owsianik, B. Fisslthaler. Modulation of the Ca2 permeable cation channel TRPV4 by cytochrome P450 epoxygenases in vascular endothelium. Circ Res, 2005. [DOI | PubMed]
- S. Derouiche, Y. Takayama, M. Murakami, M. Tominaga. TRPV4 heats up ANO1-dependent exocrine gland fluid secretion. FASEB J, 2018. [DOI | PubMed]
- M. Kashio, S. Derouiche, R.U. Yoshimoto. Involvement of TRPV4 in temperature-dependent perspiration in mice. eLife, 2024. [DOI]
- R.N. Willette, W. Bao, S. Nerurkar. Systemic activation of the transient receptor potential vanilloid subtype 4 channel causes endothelial failure and circulatory collapse: part 2. J Pharmacol Exp Ther, 2008. [DOI | PubMed]
- R. Köhler, W.-T. Heyken, P. Heinau. Evidence for a functional role of endothelial transient receptor potential V4 in shear stress-induced vasodilatation. Arterioscler Thromb Vasc Biol, 2006. [DOI | PubMed]
- C. Troidl, K. Troidl, W. Schierling. Trpv4 induces collateral vessel growth during regeneration of the arterial circulation. J Cell Mol Med, 2009. [DOI | PubMed]
- S.H. Berth, L. Vo, D.H. Kwon. Combined clinical, structural, and cellular studies discriminate pathogenic and benign TRPV4 variants. Brain, 2025. [DOI | PubMed]
- D. Müller, J.G. Hoenderop, I.C. Meij. Molecular cloning, tissue distribution, and chromosomal mapping of the human epithelial Ca2+ channel (ECAC1). Genomics, 2000. [DOI | PubMed]
- B.C.J. van der Eerden, J.G.J. Hoenderop, TJd de Vries. The epithelial Ca2+ channel TRPV5 is essential for proper osteoclastic bone resorption. Proc Natl Acad Sci U S A, 2005. [DOI | PubMed]
- J.G.J. Hoenderop, D. Müller, A.W.C.M. van der Kemp. Calcitriol controls the epithelial calcium channel in kidney. J Am Soc Nephrol, 2001. [DOI | PubMed]
- K. Weber, R.G. Erben, A. Rump, J. Adamski. Gene structure and regulation of the murine epithelial calcium channels ECaC1 and 2. Biochem Biophys Res Commun, 2001. [DOI | PubMed]
- M. van Abel, J.G.J. Hoenderop, O. Dardenne. 1,25-dihydroxyvitamin D(3)-independent stimulatory effect of estrogen on the expression of ECaC1 in the kidney. J Am Soc Nephrol, 2002. [DOI | PubMed]
- E.A.E. van der Hagen, M. Lavrijsen, F. van Zeeland. Coordinated regulation of TRPV5-mediated Ca2+ transport in primary distal convolution cultures. Pflugers Arch, 2014. [DOI | PubMed]
- J.G.J. Hoenderop, J.P.T.M. van Leeuwen, B.C.J. van der Eerden. Renal Ca2+ wasting, hyperabsorption, and reduced bone thickness in mice lacking TRPV5. J Clin Invest, 2003. [DOI | PubMed]
- N. Guleray Lafci, M. van Goor, S. Cetinkaya. Decreased calcium permeability caused by biallelic TRPV5 mutation leads to autosomal recessive renal calcium-wasting hypercalciuria. Eur J Hum Genet, 2024. [DOI | PubMed]
- D. Hirnet, J. Olausson, C. Fecher-Trost. The TRPV6 gene, cDNA and protein. Cell Calcium, 2003. [DOI | PubMed]
- Y. Suzuki, C.S. Kovacs, H. Takanaga, J.-B. Peng, C.P. Landowski, M.A. Hediger. Calcium channel TRPV6 is involved in murine maternal-fetal calcium transport. J Bone Miner Res, 2008. [DOI | PubMed]
- P. Wartenberg, F. Lux, K. Busch. A TRPV6 expression atlas for the mouse. Cell Calcium, 2021. [DOI]
- J.B. Peng, X.Z. Chen, U.V. Berger. Human calcium transport protein CaT1. Biochem Biophys Res Commun, 2000. [DOI | PubMed]
- N.F. Barley, A. Howard, D. O’Callaghan, S. Legon, J.R. Walters. Epithelial calcium transporter expression in human duodenum. Am J Physiol Gastrointest Liver Physiol, 2001. [DOI | PubMed]
- J.B. Peng, E.M. Brown, M.A. Hediger. Structural conservation of the genes encoding CaT1, CaT2, and related cation channels. Genomics, 2001. [DOI | PubMed]
- M. van Abel, J.G.J. Hoenderop, A.W.C.M. van der Kemp, J.P.T.M. van Leeuwen, R.J.M. Bindels. Regulation of the epithelial Ca2+ channels in small intestine as studied by quantitative mRNA detection. Am J Physiol Gastrointest Liver Physiol, 2003. [DOI | PubMed]
- M.B. Meyer, M. Watanuki, S. Kim, N.K. Shevde, J.W. Pike. The human transient receptor potential vanilloid type 6 distal promoter contains multiple vitamin D receptor binding sites that mediate activation by 1,25-dihydroxyvitamin D3 in intestinal cells. Mol Endocrinol, 2006. [DOI | PubMed]
- S.J. van Cromphaut, M. Dewerchin, J.G. Hoenderop. Duodenal calcium absorption in vitamin D receptor-knockout mice: functional and molecular aspects. Proc Natl Acad Sci U S A, 2001. [DOI | PubMed]
- S.J. van Cromphaut, K. Rummens, I. Stockmans. Intestinal calcium transporter genes are upregulated by estrogens and the reproductive cycle through vitamin D receptor-independent mechanisms. J Bone Miner Res, 2003. [DOI | PubMed]
- J.B. Peng, X.Z. Chen, U.V. Berger, P.M. Vassilev, E.M. Brown, M.A. Hediger. A rat kidney-specific calcium transporter in the distal nephron. J Biol Chem, 2000. [DOI | PubMed]
- J.G.J. Hoenderop, B. Nilius, R.J.M. Bindels. Calcium absorption across epithelia. Physiol Rev, 2005. [DOI | PubMed]
- C. Fecher-Trost, F. Lux, K.-M. Busch. Maternal transient receptor potential vanilloid 6 (Trpv6) is involved in offspring bone development. J Bone Miner Res, 2019. [DOI | PubMed]
- P. Weissgerber, U. Kriebs, V. Tsvilovskyy. Male fertility depends on Ca2+ absorption by TRPV6 in epididymal epithelia. Sci Signal, 2011. [DOI]
- B. Katz, R. Zaguri, S. Edvardson. Nociception and pain in humans lacking a functional TRPV1 channel. J Clin Invest, 2023. [DOI]
- F. Vanden Abeele, S. Lotteau, S. Ducreux. TRPV1 variants impair intracellular Ca2+ signaling and may confer susceptibility to malignant hyperthermia. Genet Med, 2019. [DOI | PubMed]
- L.A.M. Smit, M. Kogevinas, J.M. Antó. Transient receptor potential genes, smoking, occupational exposures and cough in adults. Respir Res, 2012. [DOI | PubMed]
- K.S. Siveen, P.B. Nizamuddin, S. Uddin. TRPV2: a cancer biomarker and potential therapeutic target. Dis Markers, 2020. [DOI]
- M. Elbaz, D. Ahirwar, Z. Xiaoli. TRPV2 is a novel biomarker and therapeutic target in triple negative breast cancer. Oncotarget, 2018. [DOI | PubMed]
- M. Nabissi, M.B. Morelli, C. Amantini. TRPV2 channel negatively controls glioma cell proliferation and resistance to Fas-induced apoptosis in ERK-dependent manner. Carcinogenesis, 2010. [DOI | PubMed]
- S. Fabris, K. Todoerti, L. Mosca. Molecular and transcriptional characterization of the novel 17p11.2-p12 amplicon in multiple myeloma. Genes Chromosomes Cancer, 2007. [DOI | PubMed]
- M. Monet, V. Lehen’kyi, F. Gackiere. Role of cationic channel TRPV2 in promoting prostate cancer migration and progression to androgen resistance. Cancer Res, 2010. [DOI | PubMed]
- P. Zoppoli, G. Calice, S. Laurino. TRPV2 calcium channel gene expression and outcomes in gastric cancer patients: a clinically relevant association. J Clin Med, 2019. [DOI | PubMed]
- Y. Qu, G. Wang, X. Sun, K. Wang. Inhibition of the warm temperature-activated Ca2+-permeable transient receptor potential vanilloid TRPV3 channel attenuates atopic dermatitis. Mol Pharmacol, 2019. [DOI | PubMed]
- C. Larkin, W. Chen, I.L. Szabó. Novel insights into the TRPV3-mediated itch in atopic dermatitis. J Allergy Clin Immunol, 2021. [DOI | PubMed]
- S.S. Özcan, G. Gürel, M. Çakır. Gene expression profiles of transient receptor potential (TRP) channels in the peripheral blood mononuclear cells of psoriasis patients. Hum Exp Toxicol, 2021. [DOI | PubMed]
- Y.S. Yang, S.I. Cho, M.G. Choi. Increased expression of three types of transient receptor potential channels (TRPA1, TRPV4 and TRPV3) in burn scars with post-burn pruritus. Acta Derm Venerol, 2015. [DOI | PubMed]
- H.O. Kim, K. Jin Cheol, K. Yu Gyeong, K. In Suk. Itching caused by TRPV3 (transient receptor potential Vanilloid-3) activator application to skin of burn patients. Med (Kaunas Lith), 2020. [DOI]
- E. Andreucci, S. Aftimos, M. Alcausin. TRPV4 related skeletal dysplasias: a phenotypic spectrum highlighted byclinical, radiographic, and molecular studies in 21 new families. Orphanet J Rare Dis, 2011. [DOI | PubMed]
- G. Nishimura, E. Lausch, R. Savarirayan. TRPV4-associated skeletal dysplasias. Am J Med Genet C Semin Med Genet, 2012. [DOI | PubMed]
- M.J. Rock, J. Prenen, V.A. Funari. Gain-of-function mutations in TRPV4 cause autosomal dominant brachyolmia. Nat Genet, 2008. [DOI | PubMed]
- N. Camacho, D. Krakow, S. Johnykutty. Dominant TRPV4 mutations in nonlethal and lethal metatropic dysplasia. Am J Med Genet A, 2010. [DOI | PubMed]
- D. Krakow, J. Vriens, N. Camacho. Mutations in the gene encoding the calcium-permeable ion channel TRPV4 produce spondylometaphyseal dysplasia, Kozlowski type and metatropic dysplasia. Am J Hum Genet, 2009. [DOI | PubMed]
- S.R. Lamandé, Y. Yuan, I.L. Gresshoff. Mutations in TRPV4 cause an inherited arthropathy of hands and feet. Nat Genet, 2011. [DOI | PubMed]
- M. Auer-Grumbach, A. Olschewski, L. Papić. Alterations in the ankyrin domain of TRPV4 cause congenital distal SMA, scapuloperoneal SMA and HMSN2C. Nat Genet, 2010. [DOI | PubMed]
- G. Landouré, A.A. Zdebik, T.L. Martinez. Mutations in TRPV4 cause Charcot-Marie-Tooth disease type 2C. Nat Genet, 2010. [DOI | PubMed]
- Y. Suzuki, D. Chitayat, H. Sawada. TRPV6 variants interfere with maternal-fetal calcium transport through the placenta and cause transient neonatal hyperparathyroidism. Am J Hum Genet, 2018. [DOI | PubMed]
- Y. Suzuki, H. Sawada, T. Tokumasu. Novel TRPV6 mutations in the spectrum of transient neonatal hyperparathyroidism. J Physiol Sci, 2020. [DOI | PubMed]
- A. Masamune, H. Kotani, F.L. Sörgel. Variants that affect function of calcium channel TRPV6 are associated with early-onset chronic pancreatitis. Gastroenterology, 2020. [DOI | PubMed]
- S. Hamada, E. Masson, J.-M. Chen. Functionally deficient TRPV6 variants contribute to hereditary and familial chronic pancreatitis. Hum Mutat, 2022. [DOI | PubMed]
- V. Lehen’kyi, N. Prevarskaya. Oncogenic TRP channels. Adv Exp Med Biol, 2011. [DOI | PubMed]
- J.B. Peng, L. Zhuang, U.V. Berger. CaT1 expression correlates with tumor grade in prostate cancer. Biochem Biophys Res Commun, 2001. [DOI | PubMed]
- T. Fixemer, U. Wissenbach, V. Flockerzi, H. Bonkhoff. Expression of the Ca2+-selective cation channel TRPV6 in human prostate cancer: a novel prognostic marker for tumor progression. Oncogene, 2003. [DOI | PubMed]
- U. Wissenbach, B. Niemeyer, N. Himmerkus, T. Fixemer, H. Bonkhoff, V. Flockerzi. TRPV6 and prostate cancer: cancer growth beyond the prostate correlates with increased TRPV6 Ca2+ channel expression. Biochem Biophys Res Commun, 2004. [DOI | PubMed]
- K.A. Bolanz, M.A. Hediger, C.P. Landowski. The role of TRPV6 in breast carcinogenesis. Mol Cancer Ther, 2008. [DOI | PubMed]
- I. Dhennin-Duthille, M. Gautier, M. Faouzi. High expression of transient receptor potential channels in human breast cancer epithelial cells and tissues: correlation with pathological parameters. Cell Physiol Biochem, 2011. [DOI | PubMed]
- H. Xue, Y. Wang, T.J. MacCormack. Inhibition of Transient Receptor Potential vanilloid 6 channel, elevated in human ovarian cancers, reduces tumour growth in a xenograft model. J Cancer, 2018. [DOI | PubMed]
- G. Mesquita, A. Haustrate, A. Mihalache. TRPV6 channel is involved in pancreatic ductal adenocarcinoma aggressiveness and resistance to chemotherapeutics. Cancers, 2023. [DOI | PubMed]
- M.J. Gunthorpe, S.L. Hannan, D. Smart. Characterization of SB-705498, a potent and selective vanilloid receptor-1 (VR1/TRPV1) antagonist that inhibits the capsaicin-, acid-, and heat-mediated activation of the receptor. J Pharmacol Exp Ther, 2007. [DOI | PubMed]
- E.M. Doherty, C. Fotsch, A.W. Bannon. Novel vanilloid receptor-1 antagonists: 2. Structure-activity relationships of 4-oxopyrimidines leading to the selection of a clinical candidate. J Med Chem, 2007. [DOI | PubMed]
- E.A. Voight, A.R. Gomtsyan, J.F. Daanen. Discovery of (R)-1-(7-chloro-2,2-bis(fluoromethyl)chroman-4-yl)-3-(3-methylisoquinolin-5-yl)urea (A-1165442): a temperature-neutral transient receptor potential vanilloid-1 (TRPV1) antagonist with analgesic efficacy. J Med Chem, 2014. [DOI | PubMed]
- A. Garami, E. Pakai, H.A. McDonald. TRPV1 antagonists that cause hypothermia, instead of hyperthermia, in rodents: compounds’ pharmacological profiles, in vivo targets, thermoeffectors recruited and implications for drug development. Acta Physiol, 2018. [DOI]
- S.G. Lehto, R. Tamir, H. Deng. Antihyperalgesic effects of (R,E)-N-(2-hydroxy-2,3-dihydro-1H-inden-4-yl)-3-(2-(piperidin-1-yl)-4-(trifluoromethyl)phenyl)-acrylamide (AMG8562), a novel transient receptor potential vanilloid type 1 modulator that does not cause hyperthermia in rats. J Pharmacol Exp Ther, 2008. [DOI | PubMed]
- W. Brown, R.L. Leff, A. Griffin. Safety, pharmacokinetics, and pharmacodynamics study in healthy subjects of oral NEO6860, a modality selective transient receptor potential vanilloid Subtype 1 antagonist. J Pain, 2017. [DOI | PubMed]
- J.W. Yun, J.A. Seo, Y.S. Jeong. TRPV1 antagonist can suppress the atopic dermatitis-like symptoms by accelerating skin barrier recovery. J Dermatol Sci, 2011. [DOI | PubMed]
- D. Smart, J.C. Jerman, M.J. Gunthorpe. Characterisation using FLIPR of human vanilloid VR1 receptor pharmacology. Eur J Pharmacol, 2001. [DOI | PubMed]
- D. Smart, M.J. Gunthorpe, J.C. Jerman. The endogenous lipid anandamide is a full agonist at the human vanilloid receptor (hVR1). Br J Pharmacol, 2000. [DOI | PubMed]
- T. Knotts, K. Mease, L. Sangameswaran, M. Felx, S. Kramer, J. Donovan. Pharmacokinetics and local tissue response to local instillation of vocacapsaicin, a novel capsaicin prodrug, in rat and rabbit osteotomy models. J Orthop Res, 2022. [DOI | PubMed]
- Y. Iwata, Y. Katayama, Y. Okuno, S. Wakabayashi. Novel inhibitor candidates of TRPV2 prevent damage of dystrophic myocytes and ameliorate against dilated cardiomyopathy in a hamster model. Oncotarget, 2018. [DOI | PubMed]
- Y. Bluhm, R. Raudszus, A. Wagner, N. Urban, M. Schaefer, K. Hill. Valdecoxib blocks rat TRPV2 channels. Eur J Pharmacol, 2022. [DOI]
- Y. Korolkova, T. Makarieva, K. Tabakmakher. Marine cyclic guanidine alkaloids Monanchomycalin B and Urupocidin A act as inhibitors of TRPV1, TRPV2 and TRPV3, but not TRPA1 receptors. Mar Drugs, 2017. [DOI]
- Q. Zhou, Y. Shi, H. Qi. Identification of two natural coumarin enantiomers for selective inhibition of TRPV2 channels. FASEB J, 2020. [DOI | PubMed]
- J. Conde, R.A. Pumroy, C. Baker. Allosteric antagonist modulation of TRPV2 by piperlongumine impairs glioblastoma progression. ACS Cent Sci, 2021. [DOI | PubMed]
- H. Chai, X. Cheng, B. Zhou. Structure-based discovery of a subtype-selective inhibitor targeting a transient receptor potential vanilloid channel. J Med Chem, 2019. [DOI | PubMed]
- S. Bang, K.Y. Kim, S. Yoo, S.H. Lee, S.W. Hwang. Transient receptor potential V2 expressed in sensory neurons is activated by probenecid. Neurosci Lett, 2007. [DOI | PubMed]
- N. Qin, M.P. Neeper, Y. Liu, T.L. Hutchinson, M.L. Lubin, C.M. Flores. TRPV2 is activated by cannabidiol and mediates CGRP release in cultured rat dorsal root ganglion neurons. J Neurosci, 2008. [DOI | PubMed]
- Y. Han, A. Luo, P.M. Kamau. A plant-derived TRPV3 inhibitor suppresses pain and itch. Br J Pharmacol, 2021. [DOI | PubMed]
- H. Qi, Y. Shi, H. Wu, C. Niu, X. Sun, K. Wang. Inhibition of temperature-sensitive TRPV3 channel by two natural isochlorogenic acid isomers for alleviation of dermatitis and chronic pruritus. Acta Pharm Sin B, 2022. [DOI | PubMed]
- X.-Y. Sun, L.-L. Sun, H. Qi. Antipruritic effect of natural coumarin osthole through selective inhibition of thermosensitive TRPV3 channel in the skin. Mol Pharmacol, 2018. [DOI | PubMed]
- H. Zhang, X. Sun, H. Qi. Pharmacological inhibition of the temperature-sensitive and Ca2+-permeable transient receptor potential vanilloid TRPV3 channel by natural Forsythoside B attenuates pruritus and cytotoxicity of keratinocytes. J Pharmacol Exp Ther, 2019. [DOI | PubMed]
- X. Sun, H. Qi, H. Wu, Y. Qu, K. Wang. Anti-pruritic and anti-inflammatory effects of natural verbascoside through selective inhibition of temperature-sensitive Ca2+-permeable TRPV3 channel. J Dermatol Sci, 2020. [DOI | PubMed]
- T.H. Dang, J.Y. Kim, H.J. Kim, B.J. Kim, W.K. Kim, J.H. Nam. Alpha-mangostin: a potent inhibitor of TRPV3 and pro-inflammatory cytokine secretion in keratinocytes. Int J Mol Sci, 2023. [DOI]
- A. Gomtsyan, R.G. Schmidt, E.K. Bayburt. Synthesis and pharmacology of (Pyridin-2-yl)methanol derivatives as novel and selective transient receptor potential vanilloid 3 antagonists. J Med Chem, 2016. [DOI | PubMed]
- J. Fan, L. Hu, Z. Yue. Structural basis of TRPV3 inhibition by an antagonist. Nat Chem Biol, 2023. [DOI | PubMed]
- R. Horishita, Y. Ogata, R. Fukui. Local anesthetics inhibit transient receptor potential vanilloid Subtype 3 channel function in Xenopus oocytes. Anesth Analg, 2021. [DOI | PubMed]
- Q. Liu, J. Wang, X. Wei. Therapeutic inhibition of keratinocyte TRPV3 sensory channel by local anesthetic dyclonine. eLife, 2021. [DOI]
- Y. Xu, Y. Qu, C. Zhang. Selective inhibition of overactive warmth-sensitive Ca2+-permeable TRPV3 channels by antispasmodic agent flopropione for alleviation of skin inflammation. J Biol Chem, 2024. [DOI]
- L.M. Broad, A.J. Mogg, E. Eberle, M. Tolley, D.L. Li, K.L. Knopp. TRPV3 in drug development. Pharmaceuticals, 2016. [DOI | PubMed]
- A.K. Vogt-Eisele, K. Weber, M.A. Sherkheli. Monoterpenoid agonists of TRPV3. Br J Pharmacol, 2007. [DOI | PubMed]
- A. Moussaieff, N. Rimmerman, T. Bregman. Incensole acetate, an incense component, elicits psychoactivity by activating TRPV3 channels in the brain. FASEB J, 2008. [DOI | PubMed]
- L. De Petrocellis, A. Ligresti, A.S. Moriello. Effects of cannabinoids and cannabinoid-enriched cannabis extracts on TRP channels and endocannabinoid metabolic enzymes. Br J Pharmacol, 2011. [DOI | PubMed]
- K.S. Thorneloe, M. Cheung, W. Bao. An orally active TRPV4 channel blocker prevents and resolves pulmonary edema induced by heart failure. Sci Transl Med, 2012. [DOI]
- C.A. Brooks, L.S. Barton, D.J. Behm. Discovery of GSK2798745: a clinical candidate for inhibition of transient receptor potential vanilloid 4 (TRPV4). ACS Med Chem Lett, 2019. [DOI | PubMed]
- W. Everaerts, X. Zhen, D. Ghosh. Inhibition of the cation channel TRPV4 improves bladder function in mice and rats with cyclophosphamide-induced cystitis. Proc Natl Acad Sci U S A, 2010. [DOI | PubMed]
- F. Vincent, A. Acevedo, M.T. Nguyen. Identification and characterization of novel TRPV4 modulators. Biochem Biophys Res Commun, 2009. [DOI | PubMed]
- Z.-L. Wei, M.T. Nguyen, D.J.R. O’Mahony. Identification of orally-bioavailable antagonists of the TRPV4 ion-channel. Bioorg Med Chem Lett, 2015. [DOI | PubMed]
- K.S. Thorneloe, A.C. Sulpizio, Z. Lin. N-((1 S)-1-{[4-((2 S)-2-{[(2, 4-dichlorophenyl) sulfonyl] amino}-3-hydroxypropanoyl)-1-piperazinyl] carbonyl}-3-methylbutyl)-1-benzothiophene-2-carboxamide (GSK1016790A), a novel and potent transient receptor potential vanilloid 4 channel agonist induces urinary bladder contraction and hyperactivity: part I. J Pharmacol Exp Ther, 2008. [DOI | PubMed]
- M. Atobe, T. Nagami, S. Muramatsu. Discovery of novel transient receptor potential vanilloid 4 (TRPV4) agonists as regulators of chondrogenic differentiation: identification of Quinazolin-4(3H)-ones and in vivo studies on a surgically induced rat model of osteoarthritis. J Med Chem, 2019. [DOI | PubMed]
- J. Shao, J. Han, Y. Zhu. Curcumin induces endothelium-dependent relaxation by activating endothelial TRPV4 channels. J Cardiovasc Transl Res, 2019. [DOI | PubMed]
- T. Zhou, Z. Wang, M. Guo. Puerarin induces mouse mesenteric vasodilation and ameliorates hypertension involving endothelial TRPV4 channels. Food Funct, 2020. [DOI | PubMed]
- C.P. Landowski, K.A. Bolanz, Y. Suzuki, M.A. Hediger. Chemical inhibitors of the calcium entry channel TRPV6. Pharm Res, 2011. [DOI | PubMed]
- T.E. Hughes, J.S. Del Rosario, A. Kapoor. Structure-based characterization of novel TRPV5 inhibitors. eLife, 2019. [DOI]
- C. Simonin, M. Awale, M. Brand. Optimization of TRPV6 calcium channel inhibitors using a 3D ligand-based virtual screening method. Angew Chem Int Ed Engl, 2015. [DOI | PubMed]
- M.R. Cunha, R. Bhardwaj, A.L. Carrel. Natural product inspired optimization of a selective TRPV6 calcium channel inhibitor. RSC Med Chem, 2020. [DOI | PubMed]
- C.V. Bowen, D. DeBay, H.S. Ewart. In vivo detection of human TRPV6-rich tumors with anti-cancer peptides derived from soricidin. PLoS One, 2013. [DOI]
- A. Janssens, C. Silvestri, A. Martella, J.M. Vanoevelen, V. Di Marzo, T. Voets. Δ9-tetrahydrocannabivarin impairs epithelial calcium transport through inhibition of TRPV5 and TRPV6. Pharmacol Res, 2018. [DOI | PubMed]
- M.R. Cunha, R. Bhardwaj, S. Lindinger. Photoswitchable inhibitor of the calcium channel TRPV6. ACS Med Chem Lett, 2019. [DOI | PubMed]
- D. Bamps, J. Vriens, J. De Hoon, T. Voets. TRP channel cooperation for nociception: therapeutic opportunities. Annu Rev Pharmacol Toxicol, 2021. [DOI | PubMed]
- P. Arsenault, D. Chiche, W. Brown. NEO6860, modality-selective TRPV1 antagonist: a randomized, controlled, proof-of-concept trial in patients with osteoarthritis knee pain. PAIN Rep, 2018. [DOI]
- S.-M. Hwang, Y.-Y. Jo, C.F. Cohen, Y.-H. Kim, T. Berta, C.-K. Park. Venom peptide toxins targeting the outer pore region of transient receptor potential vanilloid 1 in pain: implications for analgesic drug development. Int J Mol Sci, 2022. [DOI | PubMed]
- K. Alalami, J. Goff, H. Grimson. Does topical capsaicin affect the central nervous system in neuropathic pain? A narrative review. Pharmaceuticals, 2024. [DOI | PubMed]
- Y. Iwata, T. Matsumura. Blockade of TRPV2 is a novel therapy for cardiomyopathy in muscular dystrophy. Int J Mol Sci, 2019. [DOI | PubMed]
- T. Nagata, S. Shakado, E. Yamauchi. Tranilast inhibits TRPV2 and suppresses fibrosis progression and weight gain in a NASH model mouse. Anticancer Res, 2024. [DOI | PubMed]
- S. Darakhshan, A.B. Pour. Tranilast: a review of its therapeutic applications. Pharmacol Res, 2015. [DOI | PubMed]
- L. Zhang, C. Simonsen, L. Zimova. Cannabinoid non-cannabidiol site modulation of TRPV2 structure and function. Nat Commun, 2022. [DOI | PubMed]
- S.-W. Sun, R.-R. Wang, X.-Y. Sun. Identification of transient receptor potential vanilloid 3 antagonists from Achillea alpina L. and separation by liquid-liquid-refining extraction and high-speed counter-current chromatography. Molecules, 2020. [DOI | PubMed]
- A. Neuberger, K.D. Nadezhdin, A.I. Sobolevsky. Structural mechanisms of TRPV6 inhibition by ruthenium red and econazole. Nat Commun, 2021. [DOI | PubMed]
- JJd De Jesús-Pérez, M. Gabrielle, S. Raheem, E.C. Fluck, T. Rohacs, V.Y. Moiseenkova-Bell. Structural mechanism of TRPV5 inhibition by econazole. Structure, 2024. [DOI | PubMed]
- D.M. Haverstick, T.N. Heady, T.L. Macdonald, L.S. Gray. Inhibition of human prostate cancer proliferation in vitro and in a mouse model by a compound synthesized to block Ca2+ entry. Cancer Res, 2000. [PubMed]
- R. Bhardwaj, S. Lindinger, A. Neuberger. Inactivation-mimicking block of the epithelial calcium channel TRPV6. Sci Adv, 2020. [DOI]
- C. Humer, S. Lindinger, A.L. Carrel, C. Romanin, C. Höglinger. TRPV6 Regulation by Cis-22a and cholesterol. Biomolecules, 2022. [DOI | PubMed]
- A. Neuberger, Y.A. Trofimov, M.V. Yelshanskaya. Molecular pathway and structural mechanism of human oncochannel TRPV6 inhibition by the phytocannabinoid tetrahydrocannabivarin. Nat Commun, 2023. [DOI | PubMed]
- K.P. Lee, M.V. Koshelev. Upcoming topical TRPV1 anti-pruritic compounds. Dermatol Online J, 2020. [DOI]
- M. Iftinca, M. Defaye, C. Altier. TRPV1-targeted drugs in development for human pain conditions. Drugs, 2021. [DOI | PubMed]
- J. Rubinstein, J.G. Woo, A.M. Garcia. Probenecid improves cardiac function in subjects with a Fontan circulation and augments cardiomyocyte calcium homeostasis. Pediatr Cardiol, 2020. [DOI | PubMed]
- N. Robbins, M. Gilbert, M. Kumar. Probenecid improves cardiac function in patients with heart failure with reduced ejection fraction in vivo and cardiomyocyte calcium sensitivity in vitro. J Am Heart Assoc, 2018. [DOI]
- A. Shiozaki, M. Kudou, H. Fujiwara. Clinical safety and efficacy of neoadjuvant combination chemotherapy of tranilast in advanced esophageal squamous cell carcinoma: Phase I/II study (TNAC). Medicine, 2020. [DOI]
- N. Goyal, P. Skrdla, R. Schroyer. Clinical pharmacokinetics, safety, and tolerability of a novel, first-in-class TRPV4 ion channel inhibitor, GSK2798745, in healthy and heart failure subjects. Am J Cardiovasc Drugs, 2019. [DOI | PubMed]
- G.M. Stewart, B.D. Johnson, D.L. Sprecher. Targeting pulmonary capillary permeability to reduce lung congestion in heart failure: a randomized, controlled pilot trial. Eur J Heart Fail, 2020. [DOI | PubMed]
- S. Mole, A. Harry, A. Fowler. Investigating the effect of TRPV4 inhibition on pulmonary-vascular barrier permeability following segmental endotoxin challenge. Pulm Pharmacol Ther, 2020. [DOI | PubMed]
- S. Fu, H. Hirte, S. Welch. First-in-human phase I study of SOR-C13, a TRPV6 calcium channel inhibitor, in patients with advanced solid tumors. Investig New Drugs, 2017. [DOI | PubMed]
- D.E. Clapham, C. Montell, G. Schultz, D. Julius. International Union of Pharmacology. XLIII. Compendium of voltage-gated ion channels: transient receptor potential channels. Pharmacol Rev, 2003. [DOI | PubMed]
- A. Fleig, R. Penner. The TRPM ion channel subfamily: molecular, biophysical and functional features. Trends Pharmacol Sci, 2004. [DOI | PubMed]
- V. Chubanov, S. Waldegger, Y. Mederos, M. Schnitzler. Disruption of TRPM6/TRPM7 complex formation by a mutation in the TRPM6 gene causes hypomagnesemia with secondary hypocalcemia. Proc Natl Acad Sci U S A, 2004. [DOI | PubMed]
- S. Ferioli, S. Zierler, J. Zaißerer, J. Schredelseker, T. Gudermann, V. Chubanov. TRPM6 and TRPM7 differentially contribute to the relief of heteromeric TRPM6/7 channels from inhibition by cytosolic Mg2+ and Mg·ATP. Sci Rep, 2017. [DOI | PubMed]
- Z. Zhang, H. Yu, J. Huang. The TRPM6 kinase domain determines the Mg·ATP sensitivity of TRPM7/M6 heteromeric ion channels. J Biol Chem, 2014. [DOI | PubMed]
- M. Li, J. Jiang, L. Yue. Functional characterization of homo- and heteromeric channel kinases TRPM6 and TRPM7. J Gen Physiol, 2006. [DOI | PubMed]
- S. Lambert, A. Drews, O. Rizun. Transient receptor potential melastatin 1 (TRPM1) is an ion-conducting plasma membrane channel inhibited by zinc ions. J Biol Chem, 2011. [DOI | PubMed]
- Z. Zhang, B. Tóth, A. Szollosi, J. Chen, L. Csanády. Structure of a TRPM2 channel in complex with Ca2+ explains unique gating regulation. eLife, 2018. [DOI]
- Y. Yin, M. Wu, A.L. Hsu. Visualizing structural transitions of ligand-dependent gating of the TRPM2 channel. Nat Commun, 2019. [DOI | PubMed]
- Y. Huang, P.A. Winkler, W. Sun, W. Lü, J. Du. Architecture of the TRPM2 channel and its activation mechanism by ADP-ribose and calcium. Nature, 2018. [DOI | PubMed]
- Y. Huang, B. Roth, W. Lü, J. Du. Ligand recognition and gating mechanism through three ligand-binding sites of human TRPM2 channel. eLife, 2019. [DOI]
- L. Wang, T.M. Fu, Y. Zhou, S. Xia, A. Greka, H. Wu. Structures and gating mechanism of human TRPM2. Science, 2018. [DOI]
- X. Yu, Y. Xie, X. Zhang. Structural and functional basis of the selectivity filter as a gate in human TRPM2 channel. Cell Rep, 2021. [DOI]
- Y. Huang, S. Kumar, J. Lee, W. Lü, J. Du. Coupling enzymatic activity and gating in an ancient TRPM chanzyme and its molecular evolution. Nat Struct Mol Biol, 2024. [DOI | PubMed]
- C. Zhao, R. MacKinnon. Structural and functional analyses of a GPCR-inhibited ion channel TRPM3. Neuron, 2023. [DOI | PubMed]
- Y. Yin, C.G. Park, S. Feng. Molecular basis of neurosteroid and anticonvulsant regulation of TRPM3. Nat Struct Mol Biol, 2025. [DOI | PubMed]
- S. Kumar, F. Jin, S.J. Park. Convergent agonist and heat activation of nociceptor TRPM3. bioRxiv, 2025. [DOI]
- J. Guo, J. She, W. Zeng, Q. Chen, X.C. Bai, Y. Jiang. Structures of the calcium-activated, non-selective cation channel TRPM4. Nature, 2017. [DOI | PubMed]
- H.E. Autzen, A.G. Myasnikov, M.G. Campbell, D. Asarnow, D. Julius, Y. Cheng. Structure of the human TRPM4 ion channel in a lipid nanodisc. Science, 2018. [DOI | PubMed]
- P.A. Winkler, Y. Huang, W. Sun, J. Du, W. Lü. Electron cryo-microscopy structure of a human TRPM4 channel. Nature, 2017. [DOI | PubMed]
- J. Duan, Z. Li, J. Li. Structure of full-length human TRPM4. Proc Natl Acad Sci U S A, 2018. [DOI | PubMed]
- J. Hu, S.J. Park, T. Walter. Physiological temperature drives TRPM4 ligand recognition and gating. Nature, 2024. [DOI | PubMed]
- Z. Ruan, E. Haley, I.J. Orozco. Structures of the TRPM5 channel elucidate mechanisms of activation and inhibition. Nat Struct Mol Biol, 2021. [DOI | PubMed]
- S. Karuppan, L.G. Schrag, C.M. Pastrano, A. Jara-Oseguera, L. Zubcevic. Structural dynamics at cytosolic interprotomer interfaces control gating of a mammalian TRPM5 channel. Proc Natl Acad Sci U S A, 2024. [DOI]
- J. Duan, Z. Li, J. Li. Structure of the mammalian TRPM7, a magnesium channel required during embryonic development. Proc Natl Acad Sci U S A, 2018. [DOI | PubMed]
- K.D. Nadezhdin, L. Correia, A. Shalygin. Structural basis of selective TRPM7 inhibition by the anticancer agent CCT128930. Cell Rep, 2024. [DOI]
- K.D. Nadezhdin, L. Correia, C. Narangoda. Structural mechanisms of TRPM7 activation and inhibition. Nat Commun, 2023. [DOI | PubMed]
- M.M. Diver, Y. Cheng, D. Julius. Structural insights into TRPM8 inhibition and desensitization. Science, 2019. [DOI | PubMed]
- C. Zhao, Y. Xie, L. Xu. Structures of a mammalian TRPM8 in closed state. Nat Commun, 2022. [DOI | PubMed]
- Y. Yin, F. Zhang, S. Feng. Activation mechanism of the mouse cold-sensing TRPM8 channel by cooling agonist and PIP2. Science, 2022. [DOI]
- Y. Huang, R. Fliegert, A.H. Guse, W. Lü, J. Du. A structural overview of the ion channels of the TRPM family. Cell Calcium, 2020. [DOI]
- Y. Chen, X. Zhang, T. Yang. Emerging structural biology of TRPM subfamily channels. Cell Calcium, 2019. [DOI | PubMed]
- A.L. Perraud, A. Fleig, C.A. Dunn. ADP-ribose gating of the calcium-permeable LTRPC2 channel revealed by Nudix motif homology. Nature, 2001. [DOI | PubMed]
- B.W. Shen, A.L. Perraud, A. Scharenberg, B.L. Stoddard. The crystal structure and mutational analysis of human NUDT9. J Mol Biol, 2003. [DOI | PubMed]
- A.S. Mildvan, Z. Xia, H.F. Azurmendi. Structures and mechanisms of Nudix hydrolases. Arch Biochem Biophys, 2005. [DOI | PubMed]
- A. Szollosi. Two decades of evolution of our understanding of the transient receptor potential melastatin 2 (TRPM2) cation channel. Life, 2021. [DOI | PubMed]
- V. Chubanov, T. Gudermann. Trpm. TRPM6. Handb Exp Pharmacol, 2014. [DOI | PubMed]
- A. Fleig, V. Chubanov. TRPM7. Handb Exp Pharmacol, 2014. [DOI | PubMed]
- V. Chubanov, L. Mittermeier, T. Gudermann. Role of kinase-coupled TRP channels in mineral homeostasis. Pharmacol Ther, 2018. [DOI | PubMed]
- H. Yamaguchi, M. Matsushita, A.C. Nairn, J. Kuriyan. Crystal structure of the atypical protein kinase domain of a TRP channel with phosphotransferase activity. Mol Cell, 2001. [DOI | PubMed]
- A.G. Ryazanov, M.D. Ward, C.E. Mendola. Identification of a new class of protein kinases represented by eukaryotic elongation factor-2 kinase. Proc Natl Acad Sci U S A, 1997. [DOI | PubMed]
- X.Z. Xu, F. Moebius, D.L. Gill, C. Montell. Regulation of melastatin, a TRP-related protein, through interaction with a cytoplasmic isoform. Proc Natl Acad Sci U S A, 2001. [DOI | PubMed]
- E. Wehage, J. Eisfeld, I. Heiner, E. Jüngling, C. Zitt, A. Lückhoff. Activation of the cation channel long transient receptor potential channel 2 (LTRPC2) by hydrogen peroxide. A splice variant reveals a mode of activation independent of ADP-ribose. J Biol Chem, 2002. [DOI | PubMed]
- W. Zhang, X. Chu, Q. Tong. A novel TRPM2 isoform inhibits calcium influx and susceptibility to cell death. J Biol Chem, 2003. [DOI | PubMed]
- T. Uemura, J. Kudoh, S. Noda, S. Kanba, N. Shimizu. Characterization of human and mouse TRPM2 genes: identification of a novel N-terminal truncated protein specifically expressed in human striatum. Biochem Biophys Res Commun, 2005. [DOI | PubMed]
- J. Oberwinkler, A. Lis, K.M. Giehl, V. Flockerzi, S.E. Philipp. Alternative splicing switches the divalent cation selectivity of TRPM3 channels. J Biol Chem, 2005. [DOI | PubMed]
- T. Hofmann, V. Chubanov, T. Gudermann, C. Montell. TRPM5 is a voltage-modulated and Ca(2+)-activated monovalent selective cation channel. Curr Biol, 2003. [DOI | PubMed]
- P. Launay, A. Fleig, A.L. Perraud, A.M. Scharenberg, R. Penner, J.P. Kinet. TRPM4 is a Ca2+-activated nonselective cation channel mediating cell membrane depolarization. Cell, 2002. [DOI | PubMed]
- L.W. Runnels, L. Yue, D.E. Clapham. The TRPM7 channel is inactivated by PIP(2) hydrolysis. Nat Cell Biol, 2002. [DOI | PubMed]
- F.M. Schneider, F. Mohr, M. Behrendt, J. Oberwinkler. Properties and functions of TRPM1 channels in the dendritic tips of retinal ON-bipolar cells. Eur J Cell Biol, 2015. [DOI | PubMed]
- J. Oberwinkler, S.E. Phillipp. TRPM3. Handb Exp Pharmacol, 2007. [DOI]
- K. Held, V.D. Aloi, A.C.N. Freitas. Pharmacological properties of TRPM3 isoforms are determined by the length of the pore loop. Br J Pharmacol, 2022. [DOI | PubMed]
- T.F.J. Wagner, A. Drews, S. Loch. TRPM3 channels provide a regulated influx pathway for zinc in pancreatic beta cells. Pflugers Arch, 2010. [DOI | PubMed]
- K. Held, F. Gruss, V.D. Aloi. Mutations in the voltage-sensing domain affect the alternative ion permeation pathway in the TRPM3 channel. J Physiol, 2018. [DOI | PubMed]
- J. Vriens, K. Held, A. Janssens. Opening of an alternative ion permeation pathway in a nociceptor TRP channel. Nat Chem Biol, 2014. [DOI | PubMed]
- T.F.J. Wagner, S. Loch, S. Lambert. Transient receptor potential M3 channels are ionotropic steroid receptors in pancreatic beta cells. Nat Cell Biol, 2008. [DOI | PubMed]
- B.I. Tóth, M. Konrad, D. Ghosh. Regulation of the transient receptor potential channel TRPM3 by phosphoinositides. J Gen Physiol, 2015. [DOI | PubMed]
- E. Persoons, S. Kerselaers, T. Voets, J. Vriens, K. Held. Partial agonistic actions of sex hormone steroids on TRPM3 function. Int J Mol Sci, 2021. [DOI]
- C. Grimm, R. Kraft, G. Schultz, C. Harteneck. Activation of the melastatin-related cation channel TRPM3 by D-erythro-sphingosine. Mol Pharmacol, 2005. [DOI | PubMed]
- C. Grimm, R. Kraft, S. Sauerbruch, G. Schultz, C. Harteneck. Molecular and functional characterization of the melastatin-related cation channel TRPM3. J Biol Chem, 2003. [DOI | PubMed]
- J. Vriens, G. Owsianik, T. Hofmann. TRPM3 is a nociceptor channel involved in the detection of noxious heat. Neuron, 2011. [DOI | PubMed]
- M. Mulier, N. Van Ranst, N. Corthout. Upregulation of TRPM3 in nociceptors innervating inflamed tissue. eLife, 2020. [DOI]
- S. Dembla, M. Behrendt, F. Mohr. Anti-nociceptive action of peripheral mu-opioid receptors by G-beta-gamma protein-mediated inhibition of TRPM3 channels. eLife, 2017. [DOI]
- M. Behrendt, F. Gruss, R. Enzeroth. The structural basis for an on-off switch controlling Gbetagamma-mediated inhibition of TRPM3 channels. Proc Natl Acad Sci U S A, 2020. [DOI | PubMed]
- A. Kollewe, V. Chubanov, F.T. Tseung. The molecular appearance of native TRPM7 channel complexes identified by high-resolution proteomics. eLife, 2021. [DOI]
- Z. Bai, J. Feng, G.A.C. Franken. CNNM proteins selectively bind to the TRPM7 channel to stimulate divalent cation entry into cells. PLoS Biol, 2021. [DOI]
- L. Mittermeier, L. Demirkhanyan, B. Stadlbauer. TRPM7 is the central gatekeeper of intestinal mineral absorption essential for postnatal survival. Proc Natl Acad Sci U S A, 2019. [DOI | PubMed]
- C. Schmitz, A.L. Perraud, C.O. Johnson. Regulation of vertebrate cellular Mg2+ homeostasis by TRPM7. Cell, 2003. [DOI | PubMed]
- S.A. Abiria, G. Krapivinsky, R. Sah. TRPM7 senses oxidative stress to release Zn2+ from unique intracellular vesicles. Proc Natl Acad Sci U S A, 2017. [DOI | PubMed]
- M.K. Monteilh-Zoller, M.C. Hermosura, M.J.S. Nadler, A.M. Scharenberg, R. Penner, A. Fleig. TRPM7 provides an ion channel mechanism for cellular entry of trace metal ions. J Gen Physiol, 2003. [DOI | PubMed]
- M. Faouzi, T. Kilch, F.D. Horgen, A. Fleig, R. Penner. The TRPM7 channel kinase regulates store-operated calcium entry. J Physiol, 2017. [DOI | PubMed]
- C. Bates-Withers, R. Sah, D.E. Clapham. TRPM7, the Mg(2+) inhibited channel and kinase. Adv Exp Med Biol, 2011. [DOI | PubMed]
- P. Demeuse, R. Penner, A. Fleig. TRPM7 channel is regulated by magnesium nucleotides via its kinase domain. J Gen Physiol, 2006. [DOI | PubMed]
- J.A. Kozak, M. Matsushita, A.C. Nairn, M.D. Cahalan. Charge screening by internal pH and polyvalent cations as a mechanism for activation, inhibition, and rundown of TRPM7/MIC channels. J Gen Physiol, 2005. [DOI | PubMed]
- J. Xie, B. Sun, J. Du. Phosphatidylinositol 4,5-bisphosphate (PIP(2)) controls magnesium gatekeeper TRPM6 activity. Sci Rep, 2011. [DOI | PubMed]
- E. Schmidt, C. Narangoda, W. Nörenberg. Structural mechanism of TRPM7 channel regulation by intracellular magnesium. Cell Mol Life Sci, 2022. [DOI | PubMed]
- K. Clark, J. Middelbeek, N.A. Morrice, C.G. Figdor, E. Lasonder, F.N. van Leeuwen. Massive autophosphorylation of the Ser/Thr-rich domain controls protein kinase activity of TRPM6 and TRPM7. PLoS One, 2008. [DOI]
- M. Matsushita, J.A. Kozak, Y. Shimizu. Channel function is dissociated from the intrinsic kinase activity and autophosphorylation of TRPM7/ChaK1. J Biol Chem, 2005. [DOI | PubMed]
- B.N. Desai, G. Krapivinsky, B. Navarro. Cleavage of TRPM7 releases the kinase domain from the ion channel and regulates its participation in Fas-induced apoptosis. Dev Cell, 2012. [DOI | PubMed]
- G. Krapivinsky, L. Krapivinsky, Y. Manasian, D.E. Clapham. The TRPM7 chanzyme is cleaved to release a chromatin-modifying kinase. Cell, 2014. [DOI | PubMed]
- G. Krapivinsky, L. Krapivinsky, N.E. Renthal, A. Santa-Cruz, Y. Manasian, D.E. Clapham. Histone phosphorylation by TRPM6’s cleaved kinase attenuates adjacent arginine methylation to regulate gene expression. Proc Natl Acad Sci U S A, 2017. [DOI | PubMed]
- K. Brandao, F. Deason-Towne, X. Zhao, A.L. Perraud, C. Schmitz. TRPM6 kinase activity regulates TRPM7 trafficking and inhibits cellular growth under hypomagnesic conditions. Cell Mol Life Sci, 2014. [DOI | PubMed]
- M.V. Dorovkov, A.G. Ryazanov. Phosphorylation of annexin I by TRPM7 channel-kinase. J Biol Chem, 2004. [DOI | PubMed]
- K. Clark, J. Middelbeek, E. Lasonder. TRPM7 regulates myosin IIA filament stability and protein localization by heavy chain phosphorylation. J Mol Biol, 2008. [DOI | PubMed]
- A.L. Perraud, X. Zhao, A.G. Ryazanov, C. Schmitz. The channel-kinase TRPM7 regulates phosphorylation of the translational factor eEF2 via eEF2-k. Cell Signal, 2011. [DOI | PubMed]
- M.V. Dorovkov, S.N. Beznosov, S. Shah, L. Kotlianskaia, A.S. Kostiukova. [Effect of mutations imitating the phosphorylation by TRPM7 kinase on the function of the N-terminal domain of tropomodulin]. Biofizika, 2008. [DOI | PubMed]
- F. Deason-Towne, A.L. Perraud, C. Schmitz. Identification of Ser/Thr phosphorylation sites in the C2-domain of phospholipase C γ2 (PLCγ2) using TRPM7-kinase. Cell Signal, 2012. [DOI | PubMed]
- A. Romagnani, V. Vettore, T. Rezzonico-Jost. TRPM7 kinase activity is essential for T cell colonization and alloreactivity in the gut. Nat Commun, 2017. [DOI | PubMed]
- S. Voringer, L. Schreyer, W. Nadolni. Inhibition of TRPM7 blocks MRTF/SRF-dependent transcriptional and tumorigenic activity. Oncogene, 2020. [DOI | PubMed]
- K. Ogata, T. Tsumuraya, K. Oka. The crucial role of the TRPM7 kinase domain in the early stage of amelogenesis. Sci Rep, 2017. [DOI]
- Y. Sano, K. Inamura, A. Miyake. Immunocyte Ca2+ influx system mediated by LTRPC2. Science, 2001. [DOI | PubMed]
- M. Kolisek, A. Beck, A. Fleig, R. Penner. Cyclic ADP-ribose and hydrogen peroxide synergize with ADP-ribose in the activation of TRPM2 channels. Mol Cell, 2005. [DOI | PubMed]
- A. Beck, M. Kolisek, L.A. Bagley, A. Fleig, R. Penner. Nicotinic acid adenine dinucleotide phosphate and cyclic ADP-ribose regulate TRPM2 channels in T lymphocytes. FASEB J, 2006. [DOI | PubMed]
- I. Lange, R. Penner, A. Fleig, A. Beck. Synergistic regulation of endogenous TRPM2 channels by adenine dinucleotides in primary human neutrophils. Cell Calcium, 2008. [DOI | PubMed]
- A. Sumoza-Toledo, R. Penner. TRPM2: a multifunctional ion channel for calcium signalling. J Physiol, 2011. [DOI | PubMed]
- M. Faouzi, R. Penner. TRPM2. Handb Exp Pharmacol, 2014. [DOI | PubMed]
- P. Yu, Z. Liu, X. Yu. Direct gating of the TRPM2 channel by cADPR via specific interactions with the ADPR binding pocket. Cell Rep, 2019. [DOI | PubMed]
- D. McHugh, R. Flemming, S.Z. Xu, A.L. Perraud, D.J. Beech. Critical intracellular Ca2+ dependence of transient receptor potential melastatin 2 (TRPM2) cation channel activation. J Biol Chem, 2003. [DOI | PubMed]
- J. Starkus, A. Beck, A. Fleig, R. Penner. Regulation of TRPM2 by extra- and intracellular calcium. J Gen Physiol, 2007. [DOI | PubMed]
- B. Tóth, L. Csanády. Identification of direct and indirect effectors of the transient receptor potential melastatin 2 (TRPM2) cation channel. J Biol Chem, 2010. [DOI | PubMed]
- L. Csanády, B. Törocsik. Four Ca2+ ions activate TRPM2 channels by binding in deep crevices near the pore but intracellularly of the gate. J Gen Physiol, 2009. [DOI | PubMed]
- B. Tóth, L. Csanády. Pore collapse underlies irreversible inactivation of TRPM2 cation channel currents. Proc Natl Acad Sci USA, 2012. [DOI | PubMed]
- K. Song, H. Wang, G.B. Kamm. The TRPM2 channel is a hypothalamic heat sensor that limits fever and can drive hypothermia. Science, 2016. [DOI | PubMed]
- C.H. Tan, P.A. McNaughton. The TRPM2 ion channel is required for sensitivity to warmth. Nature, 2016. [DOI | PubMed]
- K. Togashi, Y. Hara, T. Tominaga. TRPM2 activation by cyclic ADP-ribose at body temperature is involved in insulin secretion. EMBO J, 2006. [DOI | PubMed]
- Á. Bartók, L. Csanády. Dual amplification strategy turns TRPM2 channels into supersensitive central heat detectors. Proc Natl Acad Sci U S A, 2022. [DOI]
- Y. Hara, M. Wakamori, M. Ishii. LTRPC2 Ca2+-permeable channel activated by changes in redox status confers susceptibility to cell death. Mol Cell, 2002. [DOI | PubMed]
- A.L. Perraud, C.L. Takanishi, B. Shen. Accumulation of free ADP-ribose from mitochondria mediates oxidative stress-induced gating of TRPM2 cation channels. J Biol Chem, 2005. [DOI | PubMed]
- O. Grubisha, L.A. Rafty, C.L. Takanishi. Metabolite of SIR2 reaction modulates TRPM2 ion channel. J Biol Chem, 2006. [DOI | PubMed]
- S. Fuessel, D. Sickert, A. Meye. Multiple tumor marker analyses (PSA, hK2, PSCA, trp-p8) in primary prostate cancers using quantitative RT-PCR. Int J Oncol, 2003. [DOI | PubMed]
- L. Tsavaler, M.H. Shapero, S. Morkowski, R. Laus. Trp-p8, a novel prostate-specific gene, is up-regulated in prostate cancer and other malignancies and shares high homology with transient receptor potential calcium channel proteins. Cancer Res, 2001. [PubMed]
- S. Brauchi, P. Orio, R. Latorre. Clues to understanding cold sensation: thermodynamics and electrophysiological analysis of the cold receptor TRPM8. Proc Natl Acad Sci U S A, 2004. [DOI | PubMed]
- Y. Yudin, V. Lukacs, C. Cao, T. Rohacs. Decrease in phosphatidylinositol 4,5-bisphosphate levels mediates desensitization of the cold sensor TRPM8 channels. J Physiol, 2011. [DOI | PubMed]
- L. Liu, Y. Yudin, J. Nagwekar, C. Kang, N. Shirokova, T. Rohacs. Gαq sensitizes TRPM8 to inhibition by PI(4,5)P2 depletion upon receptor activation. J Neurosci, 2019. [DOI | PubMed]
- L. Liu, T. Rohacs. Regulation of the cold-sensing TRPM8 channels by phosphoinositides and G(q)-coupled receptors. Channels, 2020. [DOI | PubMed]
- Y. Yin, S.Y. Lee. Current view of ligand and lipid recognition by the menthol receptor TRPM8. Trends Biochem Sci, 2020. [DOI | PubMed]
- L. Xu, Y. Han, X. Chen. Molecular mechanisms underlying menthol binding and activation of TRPM8 ion channel. Nat Commun, 2020. [DOI | PubMed]
- X. Lu, Z. Yao, Y. Wang. The acquisition of cold sensitivity during TRPM8 ion channel evolution. Proc Natl Acad Sci U S A, 2022. [DOI]
- T. Voets, G. Owsianik, B. Nilius. TRPM8. Handb Exp Pharmacol, 2007. [DOI]
- D. Prawitt, M.K. Monteilh-Zoller, L. Brixel. TRPM5 is a transient Ca2+-activated cation channel responding to rapid changes in [Ca2+]i. Proc Natl Acad Sci U S A, 2003. [DOI | PubMed]
- D. Liu, E.R. Liman. Intracellular Ca2+ and the phospholipid PIP2 regulate the taste transduction ion channel TRPM5. Proc Natl Acad Sci U S A, 2003. [DOI | PubMed]
- B. Nilius, J. Prenen, G. Droogmans. Voltage dependence of the Ca2+-activated cation channel TRPM4. J Biol Chem, 2003. [DOI | PubMed]
- B. Nilius, J. Prenen, J. Tang. Regulation of the Ca2+ sensitivity of the nonselective cation channel TRPM4. J Biol Chem, 2005. [DOI | PubMed]
- N.D. Ullrich, T. Voets, J. Prenen. Comparison of functional properties of the Ca2+-activated cation channels TRPM4 and TRPM5 from mice. Cell Calcium, 2005. [DOI | PubMed]
- K. Talavera, K. Yasumatsu, T. Voets. Heat activation of TRPM5 underlies thermal sensitivity of sweet taste. Nature, 2005. [DOI | PubMed]
- R. Vennekens, B. Nilius. Insights into TRPM4 function, regulation and physiological role. Handb Exp Pharmacol, 2007. [DOI]
- L.M. Duncan, J. Deeds, J. Hunter. Down-regulation of the novel gene melastatin correlates with potential for melanoma metastasis. Cancer Res, 1998. [PubMed]
- S. Irie, T. Furukawa. TRPM1. Handb Exp Pharmacol, 2014. [DOI | PubMed]
- E. Oancea, J. Vriens, S. Brauchi, J. Jun, I. Splawski, D.E. Clapham. TRPM1 forms ion channels associated with melanin content in melanocytes. Sci Signal, 2009. [DOI]
- C.W. Morgans, J. Zhang, B.G. Jeffrey. TRPM1 is required for the depolarizing light response in retinal ON-bipolar cells. Proc Natl Acad Sci U S A, 2009. [DOI | PubMed]
- Y. Shen, J.A. Heimel, M. Kamermans, N.S. Peachey, R.G. Gregg, S. Nawy. A transient receptor potential-like channel mediates synaptic transmission in rod bipolar cells. J Neurosci, 2009. [DOI | PubMed]
- R.R. Bellone, S.A. Brooks, L. Sandmeyer. Differential gene expression of TRPM1, the potential cause of congenital stationary night blindness and coat spotting patterns (LP) in the Appaloosa horse (Equus caballus). Genetics, 2008. [DOI | PubMed]
- M.M. van Genderen, M.M.C. Bijveld, Y.B. Claassen. Mutations in TRPM1 are a common cause of complete congenital stationary night blindness. Am J Hum Genet, 2009. [DOI | PubMed]
- I. Audo, S. Kohl, B.P. Leroy. TRPM1 is mutated in patients with autosomal-recessive complete congenital stationary night blindness. Am J Hum Genet, 2009. [DOI | PubMed]
- Z. Li, P.I. Sergouniotis, M. Michaelides. Recessive mutations of the gene TRPM1 abrogate ON bipolar cell function and cause complete congenital stationary night blindness in humans. Am J Hum Genet, 2009. [DOI | PubMed]
- C. Iosifidis, J. Liu, T. Gale. Clinical and genetic findings in TRPM1-related congenital stationary night blindness. Acta Ophthalmol, 2022. [DOI | PubMed]
- Y. Shen, M.A.F. Rampino, R.C. Carroll, S. Nawy. G-protein-mediated inhibition of the Trp channel TRPM1 requires the Gβγ dimer. Proc Natl Acad Sci U S A, 2012. [DOI | PubMed]
- Y. Xu, C. Orlandi, Y. Cao. The TRPM1 channel in ON-bipolar cells is gated by both the α and the βγ subunits of the G-protein Go. Sci Rep, 2016. [DOI]
- S. Zhao, V. Carnevale, M. Gabrielle, E. Gianti, T. Rohacs. Computational and functional studies of the PI(4,5)P2 binding site of the TRPM3 ion channel reveal interactions with other regulators. J Biol Chem, 2022. [DOI]
- I. Vandewauw, K. De Clercq, M. Mulier. A TRP channel trio mediates acute noxious heat sensing. Nature, 2018. [DOI | PubMed]
- M. Vanneste, M. Mulier, A.C. Nogueira Freitas. TRPM3 is expressed in afferent bladder neurons and is upregulated during bladder inflammation. Int J Mol Sci, 2021. [DOI | PubMed]
- M. Zhao, L. Liu, Z. Chen. Upregulation of transient receptor potential cation channel subfamily M member-3 in bladder afferents is involved in chronic pain in cyclophosphamide-induced cystitis. Pain, 2022. [DOI | PubMed]
- J. Vriens, T. Voets. Sensing the heat with TRPM3. Pflugers Arch, 2018. [DOI | PubMed]
- H.H. Kerschbaum, M.D. Cahalan. Single-channel recording of a store-operated Ca2+ channel in Jurkat T lymphocytes. Science, 1999. [DOI | PubMed]
- M. Prakriya, R.S. Lewis. Separation and characterization of currents through store-operated CRAC channels and Mg2+-inhibited cation (MIC) channels. J Gen Physiol, 2002. [DOI | PubMed]
- J.A. Kozak, H.H. Kerschbaum, M.D. Cahalan. Distinct properties of CRAC and MIC channels in RBL cells. J Gen Physiol, 2002. [DOI | PubMed]
- M.J. Nadler, M.C. Hermosura, K. Inabe. LTRPC7 is a Mg.ATP-regulated divalent cation channel required for cell viability. Nature, 2001. [DOI | PubMed]
- D.E. Clapham, L.W. Runnels, C. Strübing. The TRP ion channel family. Nat Rev Neurosci, 2001. [DOI | PubMed]
- L.V. Ryazanova, M.V. Dorovkov, A. Ansari, A.G. Ryazanov. Characterization of the protein kinase activity of TRPM7/ChaK1, a protein kinase fused to the transient receptor potential ion channel. J Biol Chem, 2004. [DOI | PubMed]
- L.V. Ryazanova, L.J. Rondon, S. Zierler. TRPM7 is essential for Mg(2+) homeostasis in mammals. Nat Commun, 2010. [DOI | PubMed]
- G. Ardestani, A. Mehregan, A. Fleig, F.D. Horgen, I. Carvacho, R.A. Fissore. Divalent cation influx and calcium homeostasis in germinal vesicle mouse oocytes. Cell Calcium, 2020. [DOI]
- M. Tashiro, H. Inoue, M. Konishi. Modulation of Mg2+ influx and cytoplasmic free Mg2+ concentration in rat ventricular myocytes. J Physiol Sci, 2019. [DOI | PubMed]
- L.W. Runnels, L. Yue, D.E. Clapham. TRP-PLIK, a bifunctional protein with kinase and ion channel activities. Science, 2001. [DOI | PubMed]
- M.S. Schappe, M.E. Stremska, G.W. Busey. Efferocytosis requires periphagosomal Ca2+-signaling and TRPM7-mediated electrical activity. Nat Commun, 2022. [DOI | PubMed]
- M.S. Schappe, K. Szteyn, M.E. Stremska. Chanzyme TRPM7 mediates the Ca2+ influx essential for lipopolysaccharide-induced toll-like receptor 4 endocytosis and macrophage activation. Immunity, 2018. [DOI | PubMed]
- S.K. Mendu, M.E. Stremska, M.S. Schappe. Targeting the ion channel TRPM7 promotes the thymic development of regulatory T cells by promoting IL-2 signaling. Sci Signal, 2020. [DOI]
- V. Chubanov, M. Köttgen, R.M. Touyz, T. Gudermann. TRPM channels in health and disease. Nat Rev Nephrol, 2024. [DOI | PubMed]
- I. Carvacho, G. Ardestani, H.C. Lee, K. McGarvey, R.A. Fissore, K. Lykke-Hartmann. TRPM7-like channels are functionally expressed in oocytes and modulate post-fertilization embryo development in mouse. Sci Rep, 2016. [DOI]
- M.L. Bernhardt, P. Stein, I. Carvacho. TRPM7 and CaV3.2 channels mediate Ca(2+) influx required for egg activation at fertilization. Proc Natl Acad Sci U S A, 2018. [DOI | PubMed]
- N. Gupta, C. Soriano-Úbeda, P. Stein. Essential role of Mg(2+) in mouse preimplantation embryo development revealed by TRPM7 chanzyme-deficient gametes. Cell Rep, 2023. [DOI]
- A. Schütz, C. Richter, P. Weissgerber. Trophectoderm cell failure leads to peri-implantation lethality in Trpm7-deficient mouse embryos. Cell Rep, 2021. [DOI]
- V. Chubanov, S. Ferioli, A. Wisnowsky. Epithelial magnesium transport by TRPM6 is essential for prenatal development and adult survival. eLife, 2016. [DOI]
- J. Jin, B.N. Desai, B. Navarro, A. Donovan, N.C. Andrews, D.E. Clapham. Deletion of Trpm7 disrupts embryonic development and thymopoiesis without altering Mg2+ homeostasis. Science, 2008. [DOI | PubMed]
- J. Jin, L.J. Wu, J. Jun. The channel kinase, TRPM7, is required for early embryonic development. Proc Natl Acad Sci U S A, 2012. [DOI | PubMed]
- R. Sah, P. Mesirca, X. Mason. The timing of myocardial Trpm7 deletion during cardiogenesis variably disrupts adult ventricular function, conduction and repolarization. Circulation, 2013. [DOI | PubMed]
- K.P. Schlingmann, S. Weber, M. Peters. Hypomagnesemia with secondary hypocalcemia is caused by mutations in TRPM6, a new member of the TRPM gene family. Nat Genet, 2002. [DOI | PubMed]
- R.Y. Walder, D. Landau, P. Meyer. Mutation of TRPM6 causes familial hypomagnesemia with secondary hypocalcemia. Nat Genet, 2002. [DOI | PubMed]
- J. van der Wijst, R.J.M. Bindels, J.G.J. Hoenderop. Mg2+ homeostasis: the balancing act of TRPM6. Curr Opin Nephrol Hypertens, 2014. [DOI | PubMed]
- T. Voets, B. Nilius, S. Hoefs. TRPM6 forms the Mg2+ influx channel involved in intestinal and renal Mg2+ absorption. J Biol Chem, 2004. [DOI | PubMed]
- G. Cao, J.G.J. Hoenderop, R.J.M. Bindels. Insight into the molecular regulation of the epithelial magnesium channel TRPM6. Curr Opin Nephrol Hypertens, 2008. [DOI | PubMed]
- Z. Zhang, M. Faouzi, J. Huang. N-Myc-induced up-regulation of TRPM6/TRPM7 channels promotes neuroblastoma cell proliferation. Oncotarget, 2014. [DOI | PubMed]
- A. Adella, L.M.M. Gommers, C. Bos, P.A. Leermakers, J.H.F. de Baaij, J.G.J. Hoenderop. Characterization of intestine-specific TRPM6 knockout C57BL/6 J mice: effects of short-term omeprazole treatment. Pflugers Arch, 2025. [DOI | PubMed]
- Y. Funato, D. Yamazaki, D. Okuzaki, N. Yamamoto, H. Miki. Importance of the renal ion channel TRPM6 in the circadian secretion of renin to raise blood pressure. Nat Commun, 2021. [DOI | PubMed]
- S. Yamamoto, S. Shimizu, S. Kiyonaka. TRPM2-mediated Ca2+influx induces chemokine production in monocytes that aggravates inflammatory neutrophil infiltration. Nat Med, 2008. [DOI | PubMed]
- A. Di, X.P. Gao, F. Qian. The redox-sensitive cation channel TRPM2 modulates phagocyte ROS production and inflammation. Nat Immunol, 2011. [DOI | PubMed]
- A. Sumoza-Toledo, I. Lange, H. Cortado. Dendritic cell maturation and chemotaxis is regulated by TRPM2-mediated lysosomal Ca2+ release. FASEB J, 2011. [DOI | PubMed]
- F.H. Robledo-Avila, J.D. Ruiz-Rosado, K.L. Brockman, S. Partida-Sánchez. The TRPM2 ion channel regulates inflammatory functions of neutrophils during Listeria monocytogenes infection. Front Immunol, 2020. [DOI | PubMed]
- H. Knowles, J.W. Heizer, Y. Li. Transient Receptor Potential melastatin 2 (TRPM2) ion channel is required for innate immunity against Listeria monocytogenes. Proc Natl Acad Sci U S A, 2011. [DOI | PubMed]
- K. Uchida, K. Dezaki, B. Damdindorj. Lack of TRPM2 impaired insulin secretion and glucose metabolisms in mice. Diabetes, 2011. [DOI | PubMed]
- N. Melzer, G. Hicking, K. Göbel, H. Wiendl. TRPM2 cation channels modulate T cell effector functions and contribute to autoimmune CNS inflammation. PLoS One, 2012. [DOI]
- M. Kashio, T. Sokabe, K. Shintaku. Redox signal-mediated sensitization of transient receptor potential melastatin 2 (TRPM2) to temperature affects macrophage functions. Proc Natl Acad Sci U S A, 2012. [DOI | PubMed]
- K. Haraguchi, A. Kawamoto, K. Isami. TRPM2 contributes to inflammatory and neuropathic pain through the aggravation of pronociceptive inflammatory responses in mice. J Neurosci, 2012. [DOI | PubMed]
- V.G. Ostapchenko, M. Chen, M.S. Guzman. The transient receptor potential melastatin 2 (TRPM2) channel contributes to beta-amyloid oligomer-related neurotoxicity and memory impairment. J Neurosci, 2015. [DOI | PubMed]
- M. Kashio, M. Tominaga. Redox signal-mediated enhancement of the temperature sensitivity of transient receptor potential melastatin 2 (TRPM2) elevates glucose-induced insulin secretion from pancreatic islets. J Biol Chem, 2015. [DOI | PubMed]
- N.E. Hoffman, B.A. Miller, J. Wang. Ca2+ entry via Trpm2 is essential for cardiac myocyte bioenergetics maintenance. Am J Physiol Heart Circ Physiol, 2015. [DOI | PubMed]
- P.T. Manna, T.S. Munsey, N. Abuarab. TRPM2-mediated intracellular Zn2+ release triggers pancreatic beta-cell death. Biochem J, 2015. [DOI | PubMed]
- A. Di, T. Kiya, H. Gong, X. Gao, A.B. Malik. Role of the phagosomal redox-sensitive TRP channel TRPM2 in regulating bactericidal activity of macrophages. J Cell Sci, 2017. [DOI | PubMed]
- J. Miyanohara, M. Kakae, K. Nagayasu. TRPM2 channel aggravates CNS inflammation and cognitive impairment via activation of microglia in chronic cerebral hypoperfusion. J Neurosci, 2018. [DOI | PubMed]
- S. Alawieyah Syed Mortadza, J.A. Sim, V.E. Neubrand, L.H. Jiang. A critical role of TRPM2 channel in Aβ42 -induced microglial activation and generation of tumor necrosis factor-α. Glia, 2018. [DOI | PubMed]
- M. Tsutsui, R. Hirase, S. Miyamura. TRPM2 exacerbates central nervous system inflammation in experimental autoimmune encephalomyelitis by increasing production of CXCL2 chemokines. J Neurosci, 2018. [DOI | PubMed]
- R. Paricio-Montesinos, F. Schwaller, A. Udhayachandran. The sensory coding of warm perception. Neuron, 2020. [DOI | PubMed]
- P. Zong, J. Feng, Z. Yue. Functional coupling of TRPM2 and extrasynaptic NMDARs exacerbates excitotoxicity in ischemic brain injury. Neuron, 2022. [DOI | PubMed]
- G. Wang, L. Cao, X. Liu. Oxidant sensing by TRPM2 inhibits neutrophil migration and mitigates inflammation. Dev Cell, 2016. [DOI | PubMed]
- B.A. Miller, J. Wang, I. Hirschler-Laszkiewicz. The second member of transient receptor potential-melastatin channel family protects hearts from ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol, 2013. [DOI | PubMed]
- I. Alim, L. Teves, R. Li, Y. Mori, M. Tymianski. Modulation of NMDAR subunit expression by TRPM2 channels regulates neuronal vulnerability to ischemic cell death. J Neurosci, 2013. [DOI | PubMed]
- B.A. Miller, N.E. Hoffman, S. Merali. TRPM2 channels protect against cardiac ischemia-reperfusion injury: role of mitochondria. J Biol Chem, 2014. [DOI | PubMed]
- G. Gao, W. Wang, R.K. Tadagavadi. TRPM2 mediates ischemic kidney injury and oxidant stress through RAC1. J Clin Invest, 2014. [DOI | PubMed]
- M. Mittal, N. Urao, C.M. Hecquet. Novel role of reactive oxygen species-activated Trp melastatin channel-2 in mediating angiogenesis and postischemic neovascularization. Arterioscler Thromb Vasc Biol, 2015. [DOI | PubMed]
- Y. Jang, S.H. Lee, B. Lee. TRPM2, a susceptibility gene for bipolar disorder, regulates glycogen synthase Kinase-3 activity in the brain. J Neurosci, 2015. [DOI | PubMed]
- M. Kakae, J. Miyanohara, M. Morishima. Pathophysiological role of TRPM2 in age-related cognitive impairment in mice. Neuroscience, 2019. [DOI | PubMed]
- S.Y. Ko, S.E. Wang, H.K. Lee. Transient receptor potential melastatin 2 governs stress-induced depressive-like behaviors. Proc Natl Acad Sci U S A, 2019. [DOI | PubMed]
- E. Eraslan, A. Tanyeli, E. Polat, E. Polat. 8-Br-cADPR, a TRPM2 ion channel antagonist, inhibits renal ischemia-reperfusion injury. J Cell Physiol, 2019. [DOI | PubMed]
- K.Y. Zhan, P.L. Yu, C.H. Liu, J.H. Luo, W. Yang. Detrimental or beneficial: the role of TRPM2 in ischemia/reperfusion injury. Acta Pharmacol Sin, 2016. [DOI | PubMed]
- X. Hu, L. Wu, X. Liu. Deficiency of ROS-activated TRPM2 channel protects neurons from cerebral ischemia-reperfusion injury through upregulating autophagy. Oxid Med Cell Longev, 2021. [DOI]
- H. Khanahmad, S.M. Mirbod, F. Karimi. Pathological mechanisms induced by TRPM2 ion channels activation in renal ischemia-reperfusion injury. Mol Biol Rep, 2022. [DOI | PubMed]
- L. Almaraz, J.A. Manenschijn, E. de la Peña, F. Viana. TRPM8. Handb Exp Pharmacol, 2014. [DOI | PubMed]
- Y. Liu, R. Mikrani, Y. He. TRPM8 channels: a review of distribution and clinical role. Eur J Pharmacol, 2020. [DOI]
- W.M. Knowlton, A. Bifolck-Fisher, D.M. Bautista, D.D. McKemy. TRPM8, but not TRPA1, is required for neural and behavioral responses to acute noxious cold temperatures and cold-mimetics in vivo. Pain, 2010. [DOI | PubMed]
- D.M. Bautista, J. Siemens, J.M. Glazer. The menthol receptor TRPM8 is the principal detector of environmental cold. Nature, 2007. [DOI | PubMed]
- A. Dhaka, A.N. Murray, J. Mathur, T.J. Earley, M.J. Petrus, A. Patapoutian. TRPM8 is required for cold sensation in mice. Neuron, 2007. [DOI | PubMed]
- R.W. Colburn, M.L. Lubin, D.J. Stone. Attenuated cold sensitivity in TRPM8 null mice. Neuron, 2007. [DOI | PubMed]
- B. Liu, L. Fan, S. Balakrishna, A. Sui, J.B. Morris, S.E. Jordt. TRPM8 is the principal mediator of menthol-induced analgesia of acute and inflammatory pain. Pain, 2013. [DOI | PubMed]
- I. Alcalde, A. Íñigo-Portugués, O. González-González. Morphological and functional changes in TRPM8-expressing corneal cold thermoreceptor neurons during aging and their impact on tearing in mice. J Comp Neurol, 2018. [DOI | PubMed]
- A.D. Weyer, S.G. Lehto. Development of TRPM8 antagonists to treat chronic pain and Migraine. Pharmaceuticals, 2017. [DOI | PubMed]
- A. Parra, R. Madrid, D. Echevarria. Ocular surface wetness is regulated by TRPM8-dependent cold thermoreceptors of the cornea. Nat Med, 2010. [DOI | PubMed]
- T. Quallo, N. Vastani, E. Horridge. TRPM8 is a neuronal osmosensor that regulates eye blinking in mice. Nat Commun, 2015. [DOI | PubMed]
- R. Ramachandran, E. Hyun, L. Zhao. TRPM8 activation attenuates inflammatory responses in mouse models of colitis. Proc Natl Acad Sci U S A, 2013. [DOI | PubMed]
- G. Shapovalov, D. Gkika, M. Devilliers. Opiates modulate thermosensation by internalizing cold receptor TRPM8. Cell Rep, 2013. [DOI | PubMed]
- W.M. Knowlton, R. Palkar, E.K. Lippoldt. A sensory-labeled line for cold: TRPM8-expressing sensory neurons define the cellular basis for cold, cold pain, and cooling-mediated analgesia. J Neurosci, 2013. [DOI | PubMed]
- N. Milenkovic, W.J. Zhao, J. Walcher. A somatosensory circuit for cooling perception in mice. Nat Neurosci, 2014. [DOI | PubMed]
- P. Uvin, J. Franken, S. Pinto. Essential role of transient receptor potential M8 (TRPM8) in a model of acute cold-induced urinary urgency. Eur Urol, 2015. [DOI | PubMed]
- W. Everaerts, T. Gevaert, B. Nilius, D. De Ridder. On the origin of bladder sensing: Tr(i)ps in urology. Neurourol Urodyn, 2008. [DOI | PubMed]
- I. Mathar, G. Jacobs, M. Kecskes, A. Menigoz, K. Philippaert, R. Vennekens. TRPM4. Handb Exp Pharmacol, 2014. [DOI | PubMed]
- E.R. Liman. TRP Ion Channel Function in Sensory Transduction and Cellular Signaling Cascades, 2007
- R. Guinamard, L. Sallé, C. Simard. The non-selective monovalent cationic channels TRPM4 and TRPM5. Adv Exp Med Biol, 2011. [DOI | PubMed]
- E.R. Liman. TRPM5. Handb Exp Pharmacol, 2014. [DOI | PubMed]
- C.A. Pérez, L. Huang, M. Rong. A transient receptor potential channel expressed in taste receptor cells. Nat Neurosci, 2002. [DOI | PubMed]
- Y. Zhang, M.A. Hoon, J. Chandrashekar. Coding of sweet, bitter, and umami tastes: different receptor cells sharing similar signaling pathways. Cell, 2003. [DOI | PubMed]
- E.R. Liman. TRPM5 and taste transduction. Handb Exp Pharmacol, 2007. [DOI]
- S. Kaske, G. Krasteva, P. König. TRPM5, a taste-signaling transient receptor potential ion-channel, is a ubiquitous signaling component in chemosensory cells. BMC Neurosci, 2007. [DOI | PubMed]
- M. Keshavarz, S. Faraj Tabrizi, A.L. Ruppert. Cysteinyl leukotrienes and acetylcholine are biliary tuft cell cotransmitters. Sci Immunol, 2022. [DOI]
- A. Perniss, S. Liu, B. Boonen. Chemosensory cell-derived acetylcholine drives tracheal mucociliary clearance in response to virulence-associated formyl peptides. Immunity, 2020. [DOI | PubMed]
- S. Wiederhold, T. Papadakis, V. Chubanov, T. Gudermann, G. Krasteva-Christ, W. Kummer. A novel cholinergic epithelial cell with chemosensory traits in the murine conjunctiva. Int Immunopharmacol, 2015. [DOI | PubMed]
- A. Soultanova, A. Voigt, V. Chubanov. Cholinergic chemosensory cells of the thymic medulla express the bitter receptor Tas2r131. Int Immunopharmacol, 2015. [DOI | PubMed]
- K. Deckmann, G. Krasteva-Christ, A. Rafiq. Cholinergic urethral brush cells are widespread throughout placental mammals. Int Immunopharmacol, 2015. [DOI | PubMed]
- G. Krasteva-Christ, A. Soultanova, B. Schütz. Identification of cholinergic chemosensory cells in mouse tracheal and laryngeal glandular ducts. Int Immunopharmacol, 2015. [DOI | PubMed]
- B. Schütz, I. Jurastow, S. Bader. Chemical coding and chemosensory properties of cholinergic brush cells in the mouse gastrointestinal and biliary tract. Front Physiol, 2015. [DOI | PubMed]
- A.R. Panneck, A. Rafiq, B. Schütz. Cholinergic epithelial cell with chemosensory traits in murine thymic medulla. Cell Tissue Res, 2014. [DOI | PubMed]
- K. Deckmann, K. Filipski, G. Krasteva-Christ. Bitter triggers acetylcholine release from polymodal urethral chemosensory cells and bladder reflexes. Proc Natl Acad Sci U S A, 2014. [DOI | PubMed]
- G. Krasteva, P. Hartmann, T. Papadakis. Cholinergic chemosensory cells in the auditory tube. Histochem Cell Biol, 2012. [DOI | PubMed]
- M.I. Hollenhorst, R. Nandigama, S.B. Evers. Bitter taste signaling in tracheal epithelial brush cells elicits innate immune responses to bacterial infection. J Clin Invest, 2022. [DOI]
- M.E. Kotas, C.E. O’Leary, R.M. Locksley. Tuft cells: context- and tissue-specific programming for a conserved cell lineage. Annu Rev Pathol Mech Dis, 2023. [DOI]
- C. Schneider, C.E. O’Leary, R.M. Locksley. Regulation of immune responses by tuft cells. Nat Rev Immunol, 2019. [DOI | PubMed]
- C. Schneider, C.E. O’Leary, J. von Moltke. A metabolite-triggered tuft cell-ILC2 circuit drives small intestinal remodeling. Cell, 2018. [DOI | PubMed]
- J. von Moltke, M. Ji, H.E. Liang, R.M. Locksley. Tuft-cell-derived IL-25 regulates an intestinal ILC2-epithelial response circuit. Nature, 2016. [DOI | PubMed]
- F. Gerbe, E. Sidot, D.J. Smyth. Intestinal epithelial tuft cells initiate type 2 mucosal immunity to helminth parasites. Nature, 2016. [DOI | PubMed]
- M.R. Howitt, S. Lavoie, M. Michaud. Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science, 2016. [DOI | PubMed]
- M.S. Nadjsombati, J.W. McGinty, M.R. Lyons-Cohen. Detection of succinate by intestinal tuft cells triggers a Type 2 innate immune circuit. Immunity, 2018. [DOI | PubMed]
- W. Lei, W. Ren, M. Ohmoto. Activation of intestinal tuft cell-expressed Sucnr1 triggers type 2 immunity in the mouse small intestine. Proc Natl Acad Sci U S A, 2018. [DOI | PubMed]
- C. Bomidi, M. Robertson, C. Coarfa, M.K. Estes, S.E. Blutt. Single-cell sequencing of rotavirus-infected intestinal epithelium reveals cell-type specific epithelial repair and tuft cell infection. Proc Natl Acad Sci U S A, 2021. [DOI]
- G. Krasteva, B.J. Canning, P. Hartmann. Cholinergic chemosensory cells in the trachea regulate breathing. Proc Natl Acad Sci U S A, 2011. [DOI | PubMed]
- N. Abdel Wadood, M.I. Hollenhorst, M.I. Elhawy. Tracheal tuft cells release ATP and link innate to adaptive immunity in pneumonia. Nat Commun, 2025. [DOI | PubMed]
- D.A. Dyment, P.A. Terhal, C.F. Rustad. De novo substitutions of TRPM3 cause intellectual disability and epilepsy. Eur J Hum Genet, 2019. [DOI | PubMed]
- R. Roelens, A.N.F. Peigneur, T. Voets, J. Vriens. Neurodevelopmental disorders caused by variants in TRPM3. Biochim Biophys Acta Mol Cell Res, 2024. [DOI]
- E. Van Hoeymissen, K. Held, A.C. Nogueira Freitas, A. Janssens, T. Voets, J. Vriens. Gain of channel function and modified gating properties in TRPM3 mutants causing intellectual disability and epilepsy. eLife, 2020. [DOI]
- L. Burglen, E. Van Hoeymissen, L. Qebibo. Gain-of-function variants in the ion channel gene TRPM3 underlie a spectrum of neurodevelopmental disorders. eLife, 2023. [DOI]
- M.A. Lines, P. Goldenberg, A. Wong. Phenotypic spectrum of the recurrent TRPM3 p.(Val837Met) substitution in seven individuals with global developmental delay and hypotonia. Am J Med Genet A, 2022. [DOI | PubMed]
- L.L. Becker, D. Horn, F. Boschann. Primidone improves symptoms in TRPM3-linked developmental and epileptic encephalopathy with spike-and-wave activation in sleep. Epilepsia, 2023. [DOI | PubMed]
- M. Friedman, G. Hatcher, L. Watson. Primary hypomagnesaemia with secondary hypocalcaemia in an infant. Lancet, 1967. [DOI | PubMed]
- P.J. Milla, P.J. Aggett, O.H. Wolff, J.T. Harries. Studies in primary hypomagnesaemia: evidence for defective carrier-mediated small intestinal transport of magnesium. Gut, 1979. [DOI | PubMed]
- J.C. Woodard, P.D. Webster, A.A. Carr. Primary hypomagnesemia with secondary hypocalcemia, diarrhea and insensitivity to parathyroid hormone. Am J Dig Dis, 1972. [DOI | PubMed]
- K.P. Schlingmann, M.C. Sassen, S. Weber. Novel TRPM6 mutations in 21 families with primary hypomagnesemia and secondary hypocalcemia. J Am Soc Nephrol, 2005. [DOI | PubMed]
- J.H. Cartwright, Q. Aziz, S.C. Harmer, S. Thayyil, A. Tinker, P.B. Munroe. Genetic variants in TRPM7 associated with unexplained stillbirth modify ion channel function. Hum Mol Genet, 2020. [DOI | PubMed]
- L. Hug, D. You, H. Blencowe. Global, regional, and national estimates and trends in stillbirths from 2000 to 2019: a systematic assessment. Lancet, 2021. [DOI | PubMed]
- S. Stritt, P. Nurden, R. Favier. Defects in TRPM7 channel function deregulate thrombopoiesis through altered cellular Mg(2+) homeostasis and cytoskeletal architecture. Nat Commun, 2016. [DOI]
- R. Gualdani, P. Gailly, J.H. Yuan. A TRPM7 mutation linked to familial trigeminal neuralgia: omega current and hyperexcitability of trigeminal ganglion neurons. Proc Natl Acad Sci U S A, 2022. [DOI]
- R. Vargas-Poussou, F. Claverie-Martin, C. Prot-Bertoye. Possible role for rare TRPM7 variants in patients with hypomagnesaemia with secondary hypocalcaemia. Nephrol Dial Transplant, 2023. [DOI | PubMed]
- M. Lei, P. Wang, H. Li. Case Report: recurrent Hemiplegic Migraine Attacks Accompanied by Intractable Hypomagnesemia due to a de novo TRPM7 Gene Variant. Front Pediatr, 2022. [DOI]
- W. Bosman, K.M. Butler, C.A. Chang. Pathogenic heterozygous TRPM7 variants and hypomagnesemia with developmental delay. Clin Kidney J, 2024. [DOI]
- M. Kruse, E. Schulze-Bahr, V. Corfield. Impaired endocytosis of the ion channel TRPM4 is associated with human progressive familial heart block type I. J Clin Invest, 2009. [DOI | PubMed]
- X. Daumy, M.Y. Amarouch, P. Lindenbaum. Targeted resequencing identifies TRPM4 as a major gene predisposing to progressive familial heart block type I. Int J Cardiol, 2016. [DOI | PubMed]
- H. Liu, S. Chatel, C. Simard. Molecular genetics and functional anomalies in a series of 248 Brugada cases with 11 mutations in the TRPM4 channel. PLoS One, 2013. [DOI]
- A. Janin, F. Bessière, T. Georgescu, V. Chanavat, P. Chevalier, G. Millat. TRPM4 mutations to cause autosomal recessive and not autosomal dominant Brugada type 1 syndrome. Eur J Med Genet, 2019. [DOI]
- F. Gualandi, F. Zaraket, M. Malagù. Mutation load of multiple ion channel gene mutations in Brugada syndrome. Cardiology, 2017. [DOI | PubMed]
- K.H.C. Li, S. Lee, C. Yin. Brugada syndrome: a comprehensive review of pathophysiological mechanisms and risk stratification strategies. Int J Cardiol Heart Vasc, 2020. [DOI]
- N. Syam, S. Chatel, L.C. Ozhathil. Variants of transient receptor potential melastatin Member 4 in childhood atrioventricular block. J Am Heart Assoc, 2016. [DOI]
- B. Bianchi, L.C. Ozhathil, A. Medeiros-Domingo, M.H. Gollob, H. Abriel. Four TRPM4 cation channel mutations found in cardiac conduction diseases lead to altered protein stability. Front Physiol, 2018. [DOI | PubMed]
- B. Stallmeyer, S. Zumhagen, I. Denjoy. Mutational spectrum in the Ca(2+)–activated cation channel gene TRPM4 in patients with cardiac conductance disturbances. Hum Mutat, 2012. [DOI | PubMed]
- W.H. Xiong, R.L. Brown, B. Reed, N.S. Burke, R.M. Duvoisin, C.W. Morgans. Voriconazole, an antifungal triazol that causes visual side effects, is an inhibitor of TRPM1 and TRPM3 channels. Invest Ophthalmol Vis Sci, 2015. [DOI | PubMed]
- K. Held, T. Kichko, K. De Clercq. Activation of TRPM3 by a potent synthetic ligand reveals a role in peptide release. Proc Natl Acad Sci U S A, 2015. [DOI | PubMed]
- U. Krügel, I. Straub, H. Beckmann, M. Schaefer. Primidone inhibits TRPM3 and attenuates thermal nociception in vivo. Pain, 2017. [DOI | PubMed]
- I. Straub, F. Mohr, J. Stab. Citrus fruit and Fabacea secondary metabolites potently and selectively block TRPM3. Br J Pharmacol, 2013. [DOI | PubMed]
- I. Straub, U. Krügel, F. Mohr. Flavanones that selectively inhibit TRPM3 attenuate thermal nociception in vivo. Mol Pharmacol, 2013. [DOI | PubMed]
- V. Chubanov, T. Gudermann. Mapping TRPM7 function by NS8593. Int J Mol Sci, 2020. [DOI | PubMed]
- V. Chubanov, S. Ferioli, T. Gudermann. Assessment of TRPM7 functions by drug-like small molecules. Cell Calcium, 2017. [DOI | PubMed]
- H.S. Sun, F.D. Horgen, D. Romo. Waixenicin A, a marine-derived TRPM7 inhibitor: a promising CNS drug lead. Acta Pharmacol Sin, 2020. [DOI | PubMed]
- V. Chubanov, Y. Mederos, M. Schnitzler, M. Meißner. Natural and synthetic modulators of SK (K(ca)2) potassium channels inhibit magnesium-dependent activity of the kinase-coupled cation channel TRPM7. Br J Pharmacol, 2012. [DOI | PubMed]
- S. Zierler, G. Yao, Z. Zhang. Waixenicin A inhibits cell proliferation through magnesium-dependent block of transient receptor potential melastatin 7 (TRPM7) channels. J Biol Chem, 2011. [DOI | PubMed]
- A. Rössig, K. Hill, W. Nörenberg. Pharmacological agents selectively acting on the channel moieties of TRPM6 and TRPM7. Cell Calcium, 2022. [DOI]
- Z. Guan, X. Chen, S. Fang, Y. Ji, Z. Gao, Y. Zheng. CCT128930 is a novel and potent antagonist of TRPM7 channel. Biochem Biophys Res Commun, 2021. [DOI | PubMed]
- X. Qin, Z. Yue, B. Sun. Sphingosine and FTY720 are potent inhibitors of the transient receptor potential melastatin 7 (TRPM7) channels. Br J Pharmacol, 2013. [DOI | PubMed]
- S. Suzuki, A. Fleig, R. Penner. CBGA ameliorates inflammation and fibrosis in nephropathy. Sci Rep, 2023. [DOI | PubMed]
- S. Suzuki, C. Wakano, M.K. Monteilh-Zoller, A.J. Cullen, A. Fleig, R. Penner. Cannabigerolic acid (CBGA) inhibits the TRPM7 ion channel through its kinase domain. Function, 2024. [DOI]
- T. Hofmann, S. Schäfer, M. Linseisen, L. Sytik, T. Gudermann, V. Chubanov. Activation of TRPM7 channels by small molecules under physiological conditions. Pflugers Arch, 2014. [DOI | PubMed]
- S. Schäfer, S. Ferioli, T. Hofmann, S. Zierler, T. Gudermann, V. Chubanov. Mibefradil represents a new class of benzimidazole TRPM7 channel agonists. Pflugers Arch, 2016. [DOI | PubMed]
- C. Song, Y. Bae, J. Jun. Identification of TG100-115 as a new and potent TRPM7 kinase inhibitor, which suppresses breast cancer cell migration and invasion. Biochim Biophys Acta Gen Subj, 2017. [DOI | PubMed]
- G.W. Busey, M.C. Manjegowda, T. Huang. Analogs of FTY720 inhibit TRPM7 but not S1PRs and exert multimodal anti-inflammatory effects. J Gen Physiol, 2024. [DOI]
- F. Alatrag, M. Amoni, R. Kelly-Laubscher, A. Gwanyanya. Cardioprotective effect of fingolimod against calcium paradox-induced myocardial injury in the isolated rat heart. Can J Physiol Pharmacol, 2022. [DOI | PubMed]
- M.K. Shin, R. Mitrut, C. Gu. Pharmacological and genetic blockade of Trpm7 in the carotid body treats obesity-induced hypertension. Hypertension, 2021. [DOI | PubMed]
- X. Sisquella, T. Nebl, J.K. Thompson. Plasmodium falciparum ligand binding to erythrocytes induce alterations in deformability essential for invasion. eLife, 2017. [DOI]
- A. Khalil, T. Shekh-Ahmad, S. Kovac. Drugs acting at TRPM7 channels inhibit seizure-like activity. Epilepsia Open, 2023. [DOI | PubMed]
- E. Turlova, R. Wong, B. Xu. TRPM7 mediates neuronal cell death upstream of calcium/calmodulin-dependent protein kinase II and calcineurin mechanism in neonatal hypoxic-ischemic brain injury. Transl Stroke Res, 2021. [DOI | PubMed]
- R. Wong, H. Gong, R. Alanazi. Inhibition of TRPM7 with waixenicin A reduces glioblastoma cellular functions. Cell Calcium, 2020. [DOI]
- S. Suzuki, R. Penner, A. Fleig. TRPM7 contributes to progressive nephropathy. Sci Rep, 2020. [DOI | PubMed]
- Q. Li, X. Lei, H. Liu. Transient receptor potential melastatin 7 aggravates necrotizing enterocolitis by promoting an inflammatory response in children. Transl Pediatr, 2022. [DOI | PubMed]
- X. Wang, M. Wang, T.T. Zhu. The TRPM7 chanzyme in smooth muscle cells drives abdominal aortic aneurysm in mice. Nat CardioVasc Res, 2025. [DOI | PubMed]
- P. Zong, C.X. Li, J. Feng. TRPM7 channel activity promotes the pathogenesis of abdominal aortic aneurysms. Nat CardioVasc Res, 2025. [DOI | PubMed]
- L. Correia, A. Shalygin, A. Erbacher, J. Zaisserer, T. Gudermann, V. Chubanov. TRPM7 underlies cadmium cytotoxicity in pulmonary cells. Arch Toxicol, 2025. [DOI | PubMed]
- M. Egawa, E. Schmücker, C. Grimm, T. Gudermann, V. Chubanov. Expression profiling identified TRPM7 and HER2 as potential targets for the combined treatment of cancer cells. Cells, 2024. [DOI | PubMed]
- R. Kraft, C. Grimm, H. Frenzel, C. Harteneck. Inhibition of TRPM2 cation channels by N-(p-amylcinnamoyl)anthranilic acid. Br J Pharmacol, 2006. [DOI | PubMed]
- S. Shimizu, R. Yonezawa, T. Hagiwara. Inhibitory effects of AG490 on H2O2-induced TRPM2-mediated Ca(2+) entry. Eur J Pharmacol, 2014. [DOI | PubMed]
- K. Hill, S. McNulty, A.D. Randall. Inhibition of TRPM2 channels by the antifungal agents clotrimazole and econazole. Naunyn Schmiedebergs Arch Pharmacol, 2004. [DOI | PubMed]
- L. Fourgeaud, C. Dvorak, M. Faouzi. Pharmacology of JNJ-28583113: a novel TRPM2 antagonist. Eur J Pharmacol, 2019. [DOI | PubMed]
- J.G. Starkus, P. Poerzgen, K. Layugan. Scalaradial is a potent inhibitor of transient receptor potential melastatin 2 (TRPM2) ion channels. J Nat Prod, 2017. [DOI | PubMed]
- H. Zhang, H. Liu, X. Luo. Design, synthesis and biological activities of 2,3-dihydroquinazolin-4(1H)-one derivatives as TRPM2 inhibitors. Eur J Med Chem, 2018. [DOI | PubMed]
- C. Moreau, T. Kirchberger, J.M. Swarbrick. Structure-activity relationship of adenosine 5′-diphosphoribose at the transient receptor potential melastatin 2 (TRPM2) channel: rational design of antagonists. J Med Chem, 2013. [DOI | PubMed]
- X. Luo, M. Li, K. Zhan. Selective inhibition of TRPM2 channel by two novel synthesized ADPR analogues. Chem Biol Drug Des, 2018. [DOI | PubMed]
- I. Cruz-Torres, D.S. Backos, P.S. Herson. Characterization and optimization of the novel transient receptor potential melastatin 2 antagonist tatM2NX. Mol Pharmacol, 2020. [DOI | PubMed]
- M. Bandell, A.E. Dubin, M.J. Petrus. High-throughput random mutagenesis screen reveals TRPM8 residues specifically required for activation by menthol. Nat Neurosci, 2006. [DOI | PubMed]
- D.D. McKemy, W.M. Neuhausser, D. Julius. Identification of a cold receptor reveals a general role for TRP channels in thermosensation. Nature, 2002. [DOI | PubMed]
- M. Bödding, U. Wissenbach, V. Flockerzi. Characterisation of TRPM8 as a pharmacophore receptor. Cell Calcium, 2007. [DOI | PubMed]
- J. Becker, C.S. Ellerkmann, H. Schmelzer. Optical control of TRPM8 channels with photoswitchable menthol. Angew Chem Int Ed Engl, 2025. [DOI]
- Y. Sui, S. Li, Y. Zhao. Identification of a natural compound, sesamin, as a novel TRPM8 antagonist with inhibitory effects on prostate adenocarcinoma. Fitoterapia, 2020. [DOI]
- S. Sanechika, C. Shimobori, K. Ohbuchi. Identification of herbal components as TRPA1 agonists and TRPM8 antagonists. J Nat Med, 2021. [DOI | PubMed]
- E.S.R. Lashinger, M.S. Steiginga, J.P. Hieble. AMTB, a TRPM8 channel blocker: evidence in rats for activity in overactive bladder and painful bladder syndrome. Am J Physiol Renal Physiol, 2008. [DOI | PubMed]
- M.C. Almeida, T. Hew-Butler, R.N. Soriano. Pharmacological blockade of the cold receptor TRPM8 attenuates autonomic and behavioral cold defenses and decreases deep body temperature. J Neurosci, 2012. [DOI | PubMed]
- N.A. Tamayo, Y. Bo, V. Gore. Fused piperidines as a novel class of potent and orally available transient receptor potential melastatin type 8 (TRPM8) antagonists. J Med Chem, 2012. [DOI | PubMed]
- D.B. Horne, K. Biswas, J. Brown. Discovery of TRPM8 antagonist ( S)-6-(((3-fluoro-4-(trifluoromethoxy)phenyl)(3-fluoropyridin-2-yl)methyl)carbamoyl)nicotinic acid (AMG 333), a clinical candidate for the treatment of Migraine. J Med Chem, 2018. [DOI | PubMed]
- M. Ohmi, Y. Shishido, T. Inoue. Identification of a novel 2-pyridyl-benzensulfonamide derivative, RQ-00203078, as a selective and orally active TRPM8 antagonist. Bioorg Med Chem Lett, 2014. [DOI | PubMed]
- D.J. Parks, W.H. Parsons, R.W. Colburn. Design and optimization of benzimidazole-containing transient receptor potential melastatin 8 (TRPM8) antagonists. J Med Chem, 2011. [DOI | PubMed]
- R. González-Muñiz, M.A. Bonache, C. Martín-Escura, I. Gómez-Monterrey. Recent progress in TRPM8 modulation: an update. Int J Mol Sci, 2019. [DOI | PubMed]
- C. Izquierdo, M. Martín-Martínez, I. Gómez-Monterrey, R. González-Muñiz. TRPM8 channels: advances in structural studies and pharmacological modulation. Int J Mol Sci, 2021. [DOI | PubMed]
- Y. Okamoto, T. Ohkubo, T. Ikebe, J. Yamazaki. Blockade of TRPM8 activity reduces the invasion potential of oral squamous carcinoma cell lines. Int J Oncol, 2012. [DOI | PubMed]
- T. Grand, M. Demion, C. Norez. 9-phenanthrol inhibits human TRPM4 but not TRPM5 cationic channels. Br J Pharmacol, 2008. [DOI | PubMed]
- Z.M. Kovács, C. Dienes, T. Hézső. Pharmacological modulation and (patho)physiological roles of TRPM4 channel-part 1: modulation of TRPM4. Pharmaceuticals, 2022. [DOI | PubMed]
- L.C. Ozhathil, C. Delalande, B. Bianchi. Identification of potent and selective small molecule inhibitors of the cation channel TRPM4. Br J Pharmacol, 2018. [DOI | PubMed]
- F. Vandewiele, A. Pironet, G. Jacobs. TRPM4 inhibition by meclofenamate suppresses Ca2+-dependent triggered arrhythmias. Eur Heart J, 2022. [DOI | PubMed]
- M.G. Leitner, N. Michel, M. Behrendt. Direct modulation of TRPM4 and TRPM3 channels by the phospholipase C inhibitor U73122. Br J Pharmacol, 2016. [DOI | PubMed]
- R. Takezawa, H. Cheng, A. Beck. A pyrazole derivative potently inhibits lymphocyte Ca2+ influx and cytokine production by facilitating transient receptor potential melastatin 4 channel activity. Mol Pharmacol, 2006. [DOI | PubMed]
- W. Fu, J. Wang, T. Li. Persistent activation of TRPM4 triggers necrotic cell death characterized by sodium overload. Nat Chem Biol, 2025. [DOI | PubMed]
- R.K. Palmer, K. Atwal, I. Bakaj. Triphenylphosphine oxide is a potent and selective inhibitor of the transient receptor potential melastatin-5 ion channel. Assay Drug Dev Technol, 2010. [DOI | PubMed]
- K. Philippaert, A. Pironet, M. Mesuere. Steviol glycosides enhance pancreatic beta-cell function and taste sensation by potentiation of TRPM5 channel activity. Nat Commun, 2017. [DOI]
- A. Barilli, L. Aldegheri, F. Bianchi. From high-throughput screening to target validation: Benzo [d] isothiazoles as potent and selective agonists of human transient receptor potential cation channel subfamily M Member 5 possessing in vivo gastrointestinal prokinetic activity in rodents. J Med Chem, 2021. [DOI | PubMed]
- M. Sabat, L.F. Raveglia, L. Aldegheri. The discovery of (1R, 3R)-1-(3-chloro-5-fluorophenyl)-3-(hydroxymethyl)-1,2,3,4-tetrahydroisoquinoline-6-carbonitrile, a potent and selective agonist of human transient receptor potential cation channel subfamily m member 5 (TRPM5) and evaluation of as a potential gastrointestinal prokinetic agent. Bioorg Med Chem, 2022. [DOI]
- P. Arullampalam, B. Preti, D. Ross-Kaschitza, M. Lochner, J.S. Rougier, H. Abriel. Species-specific effects of cation channel TRPM4 small-molecule inhibitors. Front Pharmacol, 2021. [DOI]
- K. Talavera, K. Yasumatsu, R. Yoshida. The taste transduction channel TRPM5 is a locus for bitter-sweet taste interactions. FASEB J, 2008. [DOI | PubMed]
- D.A. Zarin, T. Tse, R.J. Williams, R.M. Califf, N.C. Ide. The ClinicalTrials.gov results database – update and key issues. N Engl J Med, 2011. [DOI | PubMed]
- Y. Zhou, W. Zhang, F. Sun. Acoltremon: the first TRPM8 agonist approved for the treatment of dry eye disease. Drug Discov Ther, 2025. [DOI | PubMed]
- T.L. Cvetkov, K.W. Huynh, M.R. Cohen, V.Y. Moiseenkova-Bell. Molecular architecture and subunit organization of TRPA1 ion channel revealed by electron microscopy. J Biol Chem, 2011. [DOI | PubMed]
- C.E. Paulsen, J.-P. Armache, Y. Gao, Y. Cheng, D. Julius. Structure of the TRPA1 ion channel suggests regulatory mechanisms. Nature, 2015. [DOI | PubMed]
- Y. Suo, Z. Wang, L. Zubcevic. Structural insights into electrophile irritant sensing by the human TRPA1 channel. Neuron, 2020. [DOI | PubMed]
- A.P. Christensen, N. Akyuz, D.P. Corey. The outer pore and selectivity filter of TRPA1. PLoS One, 2016. [DOI]
- K.A. Woll, K.A. Skinner, E. Gianti. Sites contributing to TRPA1 activation by the anesthetic propofol identified by photoaffinity labeling. Biophys J, 2017. [DOI | PubMed]
- C. Liu, R. Reese, S. Vu. A non-covalent ligand reveals biased agonism of the TRPA1 ion channel. Neuron, 2021. [DOI | PubMed]
- A. Staruschenko, N.A. Jeske, A.N. Akopian. Contribution of TRPV1-TRPA1 interaction to the single channel properties of the TRPA1 channel. J Biol Chem, 2010. [DOI | PubMed]
- H.-J. Weng, K.N. Patel, N.A. Jeske. Tmem100 is a regulator of TRPA1-TRPV1 complex and contributes to persistent pain. Neuron, 2015. [DOI | PubMed]
- L. Zimova, K. Barvikova, L. Macikova. Proximal C-terminus serves as a signaling hub for TRPA1 channel regulation via its interacting molecules and supramolecular complexes. Front Physiol, 2020. [DOI | PubMed]
- A. Hinman, H.-H. Chuang, D.M. Bautista, D. Julius. TRP channel activation by reversible covalent modification. Proc Natl Acad Sci USA, 2006. [DOI | PubMed]
- B.Z. Zsidó, R. Börzsei, E. Pintér, C. Hetényi. Prerequisite binding modes determine the dynamics of action of covalent agonists of ion channel TRPA1. Pharmaceuticals, 2021. [DOI | PubMed]
- M. Trevisani, J. Siemens, S. Materazzi. 4-hydroxynonenal, an endogenous aldehyde, causes pain and neurogenic inflammation through activation of the irritant receptor TRPA1. Proc Natl Acad Sci U S A, 2007. [DOI | PubMed]
- K. Hill, M. Schaefer. Ultraviolet light and photosensitising agents activate TRPA1 via generation of oxidative stress. Cell Calcium, 2009. [DOI | PubMed]
- N.W. Bellono, L.G. Kammel, A.L. Zimmerman, E. Oancea. UV light phototransduction activates transient receptor potential A1 ion channels in human melanocytes. Proc Natl Acad Sci U S A, 2013. [DOI | PubMed]
- N. Nishio, W. Taniguchi, Y.K. Sugimura. Reactive oxygen species enhance excitatory synaptic transmission in rat spinal dorsal horn neurons by activating TRPA1 and TRPV1 channels. Neuroscience, 2013. [DOI | PubMed]
- S. Zurborg, B. Yurgionas, J.A. Jira, O. Caspani, P.A. Heppenstall. Direct activation of the ion channel TRPA1 by Ca2+. Nat Neurosci, 2007. [DOI | PubMed]
- Y.Y. Wang, R.B. Chang, H.N. Waters, D.D. McKemy, E.R. Liman. The nociceptor ion channel TRPA1 is potentiated and inactivated by permeating calcium ions. J Biol Chem, 2008. [DOI | PubMed]
- J.F. Doerner, G. Gisselmann, H. Hatt, C.H. Wetzel. Transient receptor potential channel A1 is directly gated by calcium ions. J Biol Chem, 2007. [DOI | PubMed]
- R.Z. Hill, T. Morita, R.B. Brem, D.M. Bautista. S1PR3 mediates itch and pain via distinct TRP channel-dependent pathways. J Neurosci, 2018. [DOI | PubMed]
- S.A. Maher, E.D. Dubuis, M.G. Belvisi. G-protein coupled receptors regulating cough. Curr Opin Pharmacol, 2011. [DOI | PubMed]
- G.M. Story, A.M. Peier, A.J. Reeve. ANKTM1, a TRP-like channel expressed in nociceptive neurons, is activated by cold temperatures. Cell, 2003. [DOI | PubMed]
- D.M. Bautista, S.-E. Jordt, T. Nikai. TRPA1 mediates the inflammatory actions of environmental irritants and proalgesic agents. Cell, 2006. [DOI | PubMed]
- J. Chen, D. Kang, J. Xu. Species differences and molecular determinant of TRPA1 cold sensitivity. Nat Commun, 2013. [DOI | PubMed]
- H. Zhang, C. Wang, K. Zhang. The role of TRPA1 channels in thermosensation. Cell Insight, 2022. [DOI]
- L. Moparthi, V. Sinica, V.K. Moparthi. The human TRPA1 intrinsic cold and heat sensitivity involves separate channel structures beyond the N-ARD domain. Nat Commun, 2022. [DOI | PubMed]
- Y. Karashima, J. Prenen, K. Talavera, A. Janssens, T. Voets, B. Nilius. Agonist-induced changes in Ca(2+) permeation through the nociceptor cation channel TRPA1. Biophys J, 2010. [DOI | PubMed]
- Y.V. Bobkov, E.A. Corey, B.W. Ache. The pore properties of human nociceptor channel TRPA1 evaluated in single channel recordings. Biochim Biophys Acta, 2011. [DOI | PubMed]
- A. Alam, N. Shi, Y. Jiang. Structural insight into Ca2+ specificity in tetrameric cation channels. Proc Natl Acad Sci U S A, 2007. [DOI | PubMed]
- J. Chen, D. Kim, B.R. Bianchi. Pore dilation occurs in TRPA1 but not in TRPM8 channels. Mol Pain, 2009. [DOI | PubMed]
- T.G. Banke, S.R. Chaplan, A.D. Wickenden. Dynamic changes in the TRPA1 selectivity filter lead to progressive but reversible pore dilation. Am J Physiol Cell Physiol, 2010. [DOI | PubMed]
- T.G. Banke. The dilated TRPA1 channel pore state is blocked by amiloride and analogues. Brain Res, 2011. [DOI | PubMed]
- S.-E. Jordt, D.M. Bautista, H.-H. Chuang. Mustard oils and cannabinoids excite sensory nerve fibres through the TRP channel ANKTM1. Nature, 2004. [DOI | PubMed]
- K. Nozawa, E. Kawabata-Shoda, H. Doihara. TRPA1 regulates gastrointestinal motility through serotonin release from enterochromaffin cells. Proc Natl Acad Sci U S A, 2009. [DOI | PubMed]
- J. Li, H. Zhang, Q. Du. Research progress on TRPA1 in diseases. J Membr Biol, 2023. [DOI | PubMed]
- B. Kremeyer, F. Lopera, J.J. Cox. A gain-of-function mutation in TRPA1 causes familial episodic pain syndrome. Neuron, 2010. [DOI | PubMed]
- J.T. Bell, A.K. Loomis, L.M. Butcher. Differential methylation of the TRPA1 promoter in pain sensitivity. Nat Commun, 2014. [DOI | PubMed]
- K. Talavera, J.B. Startek, J. Alvarez-Collazo. Mammalian transient receptor potential TRPA1 channels: from structure to disease. Physiol Rev, 2020. [DOI | PubMed]
- Z. Hu, Y. Zhang, W. Yu. Transient receptor potential ankyrin 1 (TRPA1) modulators: recent update and future perspective. Eur J Med Chem, 2023. [DOI]
- J.M.G. Yap, T. Ueda, Y. Kanemitsu. AITC inhibits fibroblast-myofibroblast transition via TRPA1-independent MAPK and NRF2/HO-1 pathways and reverses corticosteroids insensitivity in human lung fibroblasts. Respir Res, 2021. [DOI | PubMed]
- F. Geiger, S. Zeitlmayr, C.A. Staab-Weijnitz. An inhibitory function of TRPA1 channels in TGF-β1-driven fibroblast-to-myofibroblast differentiation. Am J Respir Cell Mol Biol, 2023. [DOI | PubMed]
- S. Gao, K.K. Kaudimba, S. Guo. Transient receptor potential ankyrin Type-1 channels as a potential target for the treatment of cardiovascular diseases. Front Physiol, 2020. [DOI | PubMed]
- F. Moccia, D. Montagna. Transient receptor potential ankyrin 1 (TRPA1) channel as a sensor of oxidative stress in cancer cells. Cells, 2023. [DOI | PubMed]
- J. Takaya, K. Mio, T. Shiraishi. A potent and site-selective agonist of TRPA1. J Am Chem Soc, 2015. [DOI | PubMed]
- T. Ryckmans, A.A. Aubdool, J.V. Bodkin. Design and pharmacological evaluation of PF-4840154, a non-electrophilic reference agonist of the TrpA1 channel. Bioorg Med Chem Lett, 2011. [DOI | PubMed]
- B. Brône, P.J. Peeters, R. Marrannes. Tear gasses CN, CR, and CS are potent activators of the human TRPA1 receptor. Toxicol Appl Pharmacol, 2008. [DOI | PubMed]
- K. Talavera, M. Gees, Y. Karashima. Nicotine activates the chemosensory cation channel TRPA1. Nat Neurosci, 2009. [DOI | PubMed]
- D.M. Bautista, P. Movahed, A. Hinman. Pungent products from garlic activate the sensory ion channel TRPA1. Proc Natl Acad Sci U S A, 2005. [DOI | PubMed]
- S.P. Lee, M.T. Buber, Q. Yang. Thymol and related alkyl phenols activate the hTRPA1 channel. Br J Pharmacol, 2008. [DOI | PubMed]
- Y. Karashima, N. Damann, J. Prenen. Bimodal action of menthol on the transient receptor potential channel TRPA1. J Neurosci, 2007. [DOI | PubMed]
- J.A. Matta, P.M. Cornett, R.L. Miyares, K. Abe, N. Sahibzada, G.P. Ahern. General anesthetics activate a nociceptive ion channel to enhance pain and inflammation. Proc Natl Acad Sci U S A, 2008. [DOI | PubMed]
- A. Schulze, B. Oehler, N. Urban, M. Schaefer, K. Hill. Apomorphine is a bimodal modulator of TRPA1 channels. Mol Pharmacol, 2013. [DOI | PubMed]
- N. Hatano, H. Suzuki, Y. Muraki, K. Muraki. Stimulation of human TRPA1 channels by clinical concentrations of the antirheumatic drug auranofin. Am J Physiol Cell Physiol, 2013. [DOI | PubMed]
- J. Escalera, C.A. von Hehn, B.F. Bessac, M. Sivula, S.E. Jordt. TRPA1 mediates the noxious effects of natural sesquiterpene deterrents. J Biol Chem, 2008. [DOI | PubMed]
- H. Hu, J. Tian, Y. Zhu. Activation of TRPA1 channels by fenamate nonsteroidal anti-inflammatory drugs. Pflugers Arch Eur J Physiol, 2010. [DOI | PubMed]
- A. Schulze, P. Hartung, M. Schaefer, K. Hill. Transient receptor potential ankyrin 1 (TRPA1) channel activation by the thienopyridine-type drugs ticlopidine, clopidogrel, and prasugrel. Cell Calcium, 2014. [DOI | PubMed]
- L.J. Macpherson, A.E. Dubin, M.J. Evans. Noxious compounds activate TRPA1 ion channels through covalent modification of cysteines. Nature, 2007. [DOI | PubMed]
- L.J. Macpherson, B. Xiao, K.Y. Kwan. An ion channel essential for sensing chemical damage. J Neurosci, 2007. [DOI | PubMed]
- C.R. McNamara, J. Mandel-Brehm, D.M. Bautista. TRPA1 mediates formalin-induced pain. Proc Natl Acad Sci U S A, 2007. [DOI | PubMed]
- J. Chen, S.K. Joshi, S. DiDomenico. Selective blockade of TRPA1 channel attenuates pathological pain without altering noxious cold sensation or body temperature regulation. Pain, 2011. [DOI | PubMed]
- M. Petrus, A.M. Peier, M. Bandell. A role of TRPA1 in mechanical hyperalgesia is revealed by pharmacological inhibition. Mol Pain, 2007. [DOI | PubMed]
- L.M. Broad, J.G. Suico, P.K. Turner. Preclinical and clinical evaluation of a novel TRPA1 antagonist LY3526318. Pain, 2025. [DOI | PubMed]
- L.B. Schenkel, P.R. Olivieri, A.A. Boezio. Optimization of a novel quinazolinone-based series of transient receptor potential A1 (TRPA1) antagonists demonstrating potent in vivo activity. J Med Chem, 2016. [DOI | PubMed]
- S. Mesch, D. Walter, A. Laux-Biehlmann. Discovery of BAY-390, a selective CNS penetrant chemical probe as transient receptor potential ankyrin 1 (TRPA1) antagonist. J Med Chem, 2023. [DOI | PubMed]
- A. Balestrini, V. Joseph, M. Dourado. A TRPA1 inhibitor suppresses neurogenic inflammation and airway contraction for asthma treatment. J Exp Med, 2021. [DOI]
- H. Xu, N.T. Blair, D.E. Clapham. Camphor activates and strongly desensitizes the transient receptor potential vanilloid subtype 1 channel in a vanilloid-independent mechanism. J Neurosci, 2005. [DOI | PubMed]
- K. Nagata, A. Duggan, G. Kumar, J. García-Añoveros. Nociceptor and hair cell transducer properties of TRPA1, a channel for pain and hearing. J Neurosci, 2005. [DOI | PubMed]
- M. Bandell, G.M. Story, S.W. Hwang. Noxious cold ion channel TRPA1 is activated by pungent compounds and bradykinin. Neuron, 2004. [DOI | PubMed]
- L.J. Macpherson, B.H. Geierstanger, V. Viswanath. The pungency of garlic: activation of TRPA1 and TRPV1 in response to allicin. Curr Biol CB, 2005. [DOI | PubMed]
- B. Xiao, A.E. Dubin, B. Bursulaya, V. Viswanath, T.J. Jegla, A. Patapoutian. Identification of transmembrane domain 5 as a critical molecular determinant of menthol sensitivity in mammalian TRPA1 channels. J Neurosci, 2008. [DOI | PubMed]
- Y.A. Alpizar, M. Gees, A. Sanchez. Bimodal effects of cinnamaldehyde and camphor on mouse TRPA1. Pflugers Arch Eur J Physiol, 2013. [DOI | PubMed]
- B.F. Bessac, M. Sivula, H. CAv, J. Escalera, L. Cohn, S.-E. Jordt. TRPA1 is a major oxidant sensor in murine airway sensory neurons. J Clin Invest, 2008. [DOI | PubMed]
- D.A. Andersson, C. Gentry, S. Moss, S. Bevan. Transient receptor potential A1 is a sensory receptor for multiple products of oxidative stress. J Neurosci, 2008. [DOI | PubMed]
- B. Oehler, K. Kistner, C. Martin. Inflammatory pain control by blocking oxidized phospholipid-mediated TRP channel activation. Sci Rep, 2017. [DOI | PubMed]
- A. Babes, S.K. Sauer, L. Moparthi. Photosensitization in porphyrias and photodynamic therapy involves TRPA1 and TRPV1. J Neurosci, 2016. [DOI | PubMed]
- J.V. Lin King, J.J. Emrick, M.J.S. Kelly. A cell-penetrating scorpion toxin enables mode-specific modulation of TRPA1 and pain. Cell, 2019. [DOI | PubMed]
- P.-Y. Lam, A.R. Thawani, E. Balderas. TRPswitch — a step function chemo-optogenetic ligand for the vertebrate TRPA1 channel. J Am Chem Soc, 2020. [DOI | PubMed]
- Z. Qiao, J. Luo, Y.-Q. Tang. Photosensitive and photoswitchable TRPA1 agonists optically control pain through channel desensitization. J Med Chem, 2021. [DOI | PubMed]
- S. McGaraughty, K.L. Chu, R.J. Perner, S. Didomenico, M.E. Kort, P.R. Kym. TRPA1 modulation of spontaneous and mechanically evoked firing of spinal neurons in uninjured, osteoarthritic, and inflamed rats. Mol Pain, 2010. [DOI | PubMed]
- I. Mukhopadhyay, A. Kulkarni, S. Aranake. Transient receptor potential ankyrin 1 receptor activation in vitro and in vivo by pro-tussive agents: GRC 17536 as a promising anti-tussive therapeutic. PLoS One, 2014. [DOI]
- N. Tomsen, O. Ortega, D. Alvarez-Berdugo, L. Rofes, P. Clavé. A comparative study on the effect of acute pharyngeal stimulation with TRP agonists on the biomechanics and neurophysiology of swallow response in patients with oropharyngeal dysphagia. Int J Mol Sci, 2022. [DOI]
- Q. Chen, J. She, W. Zeng. Structure of mammalian endolysosomal TRPML1 channel in nanodiscs. Nature, 2017. [DOI | PubMed]
- M. Fine, X. Li. A structural overview of TRPML1 and the TRPML family. Handb Exp Pharmacol, 2023. [DOI | PubMed]
- M. Hirschi, M.A. Herzik, J. Wie. Cryo-electron microscopy structure of the lysosomal calcium-permeable channel TRPML3. Nature, 2017. [DOI | PubMed]
- M. Li, W.K. Zhang, N.M. Benvin. Structural basis of dual Ca2+/pH regulation of the endolysosomal TRPML1 channel. Nat Struct Mol Biol, 2017. [DOI | PubMed]
- P. Schmiege, M. Fine, G. Blobel, X. Li. Human TRPML1 channel structures in open and closed conformations. Nature, 2017. [DOI | PubMed]
- X. Song, J. Li, M. Tian. Cryo-EM structure of mouse TRPML2 in lipid nanodiscs. J Biol Chem, 2022. [DOI]
- X. Zhou, M. Li, D. Su. Cryo-EM structures of the human endolysosomal TRPML3 channel in three distinct states. Nat Struct Mol Biol, 2017. [DOI | PubMed]
- B. Spix, A.J. Castiglioni, N.N. Remis. Whole-body analysis of TRPML3 (MCOLN3) expression using a GFP-reporter mouse model reveals widespread expression in secretory cells and endocrine glands. PLoS One, 2022. [DOI]
- B. Venugopal, M.F. Browning, C. Curcio-Morelli. Neurologic, gastric, and opthalmologic pathologies in a murine model of mucolipidosis type IV. Am J Hum Genet, 2007. [DOI | PubMed]
- M.C. Micsenyi, K. Dobrenis, G. Stephney. Neuropathology of the Mcoln1(-/-) knockout mouse model of mucolipidosis type IV. J Neuropathol Exp Neurol, 2009. [DOI | PubMed]
- C. Grimm, S. Jörs, Z. Guo, A.G. Obukhov, S. Heller. Constitutive activity of TRPML2 and TRPML3 channels versus activation by low extracellular sodium and small molecules. J Biol Chem, 2012. [DOI | PubMed]
- D. Shen, X. Wang, X. Li. Lipid storage disorders block lysosomal trafficking by inhibiting a TRP channel and lysosomal calcium release. Nat Commun, 2012. [DOI | PubMed]
- C.-C. Chen, M. Keller, M. Hess. A small molecule restores function to TRPML1 mutant isoforms responsible for mucolipidosis type IV. Nat Commun, 2014. [DOI | PubMed]
- N. Sahoo, M. Gu, X. Zhang. Gastric acid secretion from parietal cells is mediated by a Ca2+ efflux channel in the tubulovesicle. Dev Cell, 2017. [DOI | PubMed]
- N. Rinkenberger, J.W. Schoggins. Mucolipin-2 cation channel increases trafficking efficiency of endocytosed viruses. mBio, 2018. [DOI | PubMed]
- C. Grimm, M.P. Cuajungco, A.F.J. van Aken. A helix-breaking mutation in TRPML3 leads to constitutive activity underlying deafness in the varitint-waddler mouse. Proc Natl Acad Sci U S A, 2007. [DOI | PubMed]
- H.J. Kim, Q. Li, S. Tjon-Kon-Sang, I. So, K. Kiselyov, S. Muallem. Gain-of-function mutation in TRPML3 causes the mouse Varitint-Waddler phenotype. J Biol Chem, 2007. [DOI | PubMed]
- H. Xu, M. Delling, L. Li, X. Dong, D.E. Clapham. Activating mutation in a mucolipin transient receptor potential channel leads to melanocyte loss in varitint-waddler mice. Proc Natl Acad Sci U S A, 2007. [DOI | PubMed]
- K. Nagata, L. Zheng, T. Madathany, A.J. Castiglioni, J.R. Bartles, J. García-Añoveros. The varitint-waddler (Va) deafness mutation in TRPML3 generates constitutive, inward rectifying currents and causes cell degeneration. Proc Natl Acad Sci U S A, 2008. [DOI | PubMed]
- C. Grimm, S. Jörs, S. Heller. Life and death of sensory hair cells expressing constitutively active TRPML3. J Biol Chem, 2009. [DOI | PubMed]
- J. Böck, E. Krogsaeter, M. Passon. Human genome diversity data reveal that L564P is the predominant TPC2 variant and a prerequisite for the blond hair associated M484L gain-of-function effect. PLoS Genet, 2021. [DOI]
- S.W. Kim, M.K. Kim, S. Hong. The intracellular Ca2+ channel TRPML3 is a PI3P effector that regulates autophagosome biogenesis. Proc Natl Acad Sci U S A, 2022. [DOI]
- Y. Posor, W. Jang, V. Haucke. Phosphoinositides as membrane organizers. Nat Rev Mol Cell Biol, 2022. [DOI | PubMed]
- C. Harteneck, T.D. Plant, G. Schultz. From worm to man: three subfamilies of TRP channels. Trends Neurosci, 2000. [DOI | PubMed]
- M. Fine, P. Schmiege, X. Li. Structural basis for PtdInsP2-mediated human TRPML1 regulation. Nat Commun, 2018. [DOI | PubMed]
- E. Krogsaeter, A.S. Rosato, C. Grimm. TRPMLs and TPCs: targets for lysosomal storage and neurodegenerative disease therapy?. Cell Calcium, 2022. [DOI]
- C.-C. Chen, E. Krogsaeter, E.S. Butz. TRPML2 is an osmo/mechanosensitive cation channel in endolysosomal organelles. Sci Adv, 2020. [DOI]
- E. Plesch, C.-C. Chen, E. Butz. Selective agonist of TRPML2 reveals direct role in chemokine release from innate immune cells. eLife, 2018. [DOI]
- F. Di Palma, I.A. Belyantseva, H.J. Kim, T.F. Vogt, B. Kachar, K. Noben-Trauth. Mutations in Mcoln3 associated with deafness and pigmentation defects in varitint-waddler (Va) mice. Proc Natl Acad Sci U S A, 2002. [DOI | PubMed]
- H.J. Kim, Q. Li, S. Tjon-Kon-Sang. A novel mode of TRPML3 regulation by extracytosolic pH absent in the varitint-waddler phenotype. EMBO J, 2008. [DOI | PubMed]
- C. Grimm, S. Jörs, S.A. Saldanha. Small molecule activators of TRPML3. Chem Biol, 2010. [DOI | PubMed]
- S. Jörs, C. Grimm, L. Becker, S. Heller. Genetic inactivation of Trpml3 does not lead to hearing and vestibular impairment in mice. PLoS One, 2010. [DOI]
- C. Grimm, L.M. Holdt, C.C. Chen. High susceptibility to fatty liver disease in two-pore channel 2-deficient mice. Nat Commun, 2014. [DOI | PubMed]
- B. Spix, E.S. Butz, C.-C. Chen. Lung emphysema and impaired macrophage elastase clearance in mucolipin 3 deficient mice. Nat Commun, 2022. [DOI | PubMed]
- D. Shen. 2012
- L. Yu, X. Zhang, Y. Yang. Small-molecule activation of lysosomal TRP channels ameliorates Duchenne muscular dystrophy in mouse models. Sci Adv, 2020. [DOI]
- X. Zhang, W. Chen, P. Li. Agonist-specific voltage-dependent gating of lysosomal two-pore Na+ channels. eLife, 2019. [DOI]
- S. Rautenberg, M. Keller, C. Leser, C.-C. Chen, F. Bracher, C. Grimm. Endolysosomal Voltage-Dependent Cation Channels, 2022
- C. Leser, M. Keller, S. Gerndt. Chemical and pharmacological characterization of the TRPML calcium channel blockers ML-SI1 and ML-SI3. Eur J Med Chem, 2021. [DOI]
- K. Kriegler, C. Leser, P. Mayer, F. Bracher. Effective chiral pool synthesis of both enantiomers of the TRPML inhibitor trans-ML-SI3. Arch Pharm (Weinheim), 2022. [DOI]
- P. Rühl, A.S. Rosato, N. Urban. Estradiol analogs attenuate autophagy, cell migration and invasion by direct and selective inhibition of TRPML1, independent of estrogen receptors. Sci Rep, 2021. [DOI | PubMed]
- X.-P. Dong, X. Cheng, E. Mills. The type IV mucolipidosis-associated protein TRPML1 is an endolysosomal iron release channel. Nature, 2008. [DOI | PubMed]
- C.-C. Chen, C. Cang, S. Fenske. Patch-clamp technique to characterize ion channels in enlarged individual endolysosomes. Nat Protoc, 2017. [DOI | PubMed]
- M. Chandra, H. Zhou, Q. Li, S. Muallem, S.L. Hofmann, A.A. Soyombo. A role for the Ca2+ channel TRPML1 in gastric acid secretion, based on analysis of knockout mice. Gastroenterology, 2011. [DOI | PubMed]
- Y. Grishchuk, K.A. Peña, J. Coblentz. Impaired myelination and reduced brain ferric iron in the mouse model of mucolipidosis IV. Dis Models Mech, 2015. [DOI]
- X. Cheng, X. Zhang, Q. Gao. The intracellular Ca2+ channel MCOLN1 is required for sarcolemma repair to prevent muscular dystrophy. Nat Med, 2014. [DOI | PubMed]
- N.N. Remis, T. Wiwatpanit, A.J. Castiglioni, E.N. Flores, J.A. Cantú, J. García-Añoveros. Mucolipin co-deficiency causes accelerated endolysosomal vacuolation of enterocytes and failure-to-thrive from birth to weaning. PLoS Genet, 2014. [DOI]
- L. Sun, Y. Hua, S. Vergarajauregui, H.I. Diab, R. Puertollano. Novel role of TRPML2 in the regulation of the innate immune response. J Immunol, 2015. [DOI | PubMed]
- X. Li, N. Rydzewski, A. Hider. A molecular mechanism to regulate lysosome motility for lysosome positioning and tubulation. Nat Cell Biol, 2016. [DOI | PubMed]
- R. Bargal, N. Avidan, T. Olender. Mucolipidosis type IV: novel MCOLN1 mutations in Jewish and non-Jewish patients and the frequency of the disease in the Ashkenazi Jewish population. Hum Mutat, 2001. [DOI | PubMed]
- L. Edelmann, J. Dong, R.J. Desnick, R. Kornreich. Carrier screening for mucolipidosis type IV in the American Ashkenazi Jewish population. Am J Hum Genet, 2002. [DOI | PubMed]
- Y. Miao, G. Li, X. Zhang, H. Xu, S.N. Abraham. A TRP channel senses lysosome neutralization by pathogens to trigger their expulsion. Cell, 2015. [DOI | PubMed]
- D.L. Medina, S. Di Paola, I. Peluso. Lysosomal calcium signalling regulates autophagy through calcineurin and TFEB. Nat Cell Biol, 2015. [DOI | PubMed]
- A. Scotto Rosato, S. Montefusco, C. Soldati. TRPML1 links lysosomal calcium to autophagosome biogenesis through the activation of the CaMKKβ/VPS34 pathway. Nat Commun, 2019. [DOI | PubMed]
- C. Settembre, C. Di Malta, V.A. Polito. TFEB links autophagy to lysosomal biogenesis. Science, 2011. [DOI | PubMed]
- M. Palmieri, S. Impey, H. Kang. Characterization of the CLEAR network reveals an integrated control of cellular clearance pathways. Hum Mol Genet, 2011. [DOI | PubMed]
- X. Zhang, X. Cheng, L. Yu. MCOLN1 is a ROS sensor in lysosomes that regulates autophagy. Nat Commun, 2016. [DOI]
- R.M. Dayam, A. Saric, R.E. Shilliday, R.J. Botelho. The phosphoinositide-gated lysosomal Ca(2+) channel, TRPML1, is required for Phagosome maturation. Traffic (Copenhagen Den), 2015. [DOI]
- N. Zhang, Z. Zhang, I. Ozden, S. Ding. Imaging mitochondrial Ca2+ uptake in astrocytes and neurons using genetically encoded Ca2+ indicators (GECIs). J Vis Exp, 2022. [DOI]
- Q. Cao, Y. Yang, X.Z. Zhong, X.-P. Dong. The lysosomal Ca2+ release channel TRPML1 regulates lysosome size by activating calmodulin. J Biol Chem, 2017. [DOI | PubMed]
- M. Samie, X. Wang, X. Zhang. A TRP channel in the lysosome regulates large particle phagocytosis via focal exocytosis. Dev Cell, 2013. [DOI | PubMed]
- S. Di Paola, D.L. Medina. TRPML1-/TFEB-dependent regulation of lysosomal exocytosis. Methods Mol Biol (Clifton NJ), 2019. [DOI]
- A. Scotto Rosato, E.K. Krogsaeter, D. Jaślan. TPC2 rescues lysosomal storage in mucolipidosis type IV, Niemann-Pick type C1, and Batten disease. EMBO Mol Med, 2022. [DOI]
- A. Rodríguez, P. Webster, J. Ortego, N.W. Andrews. Lysosomes behave as Ca2+-regulated exocytic vesicles in fibroblasts and epithelial cells. J Cell Biol, 1997. [DOI | PubMed]
- J.P. Luzio, P.R. Pryor, N.A. Bright. Lysosomes: fusion and function. Nat Rev Mol Cell Biol, 2007. [DOI | PubMed]
- J.M. LaPlante, J. Falardeau, M. Sun. Identification and characterization of the single channel function of human mucolipin-1 implicated in mucolipidosis type IV, a disorder affecting the lysosomal pathway. FEBS Lett, 2002. [DOI | PubMed]
- D.L. Medina, A. Fraldi, V. Bouche. Transcriptional activation of lysosomal exocytosis promotes cellular clearance. Dev Cell, 2011. [DOI | PubMed]
- T. Tsunemi, T. Perez-Rosello, Y. Ishiguro. Increased lysosomal exocytosis induced by lysosomal Ca2+ channel agonists protects human dopaminergic neurons from α-synuclein toxicity. J Neurosci, 2019. [DOI | PubMed]
- Y. Xu, S. Du, J.A. Marsh. TFEB regulates lysosomal exocytosis of tau and its loss of function exacerbates tau pathology and spreading. Mol Psychiatry, 2021. [DOI | PubMed]
- M. Xu, X.-P. Dong. Endolysosomal TRPMLs in Cancer. Biomolecules, 2021. [DOI | PubMed]
- X.-P. Dong, X. Wang, D. Shen. Activating mutations of the TRPML1 channel revealed by proline-scanning mutagenesis. J Biol Chem, 2009. [DOI | PubMed]
- S. Park, M. Ahuja, M.S. Kim. Fusion of lysosomes with secretory organelles leads to uncontrolled exocytosis in the lysosomal storage disease mucolipidosis type IV. EMBO Rep, 2016. [DOI | PubMed]
- Y.-K. Chao, S.-Y. Chang, C. Grimm. Endo-lysosomal cation channels and infectious diseases. Rev Physiol Biochem Pharmacol, 2023. [DOI | PubMed]
- B. Spix, Y.-K. Chao, C. Abrahamian, C.-C. Chen, C. Grimm. TRPML. TRPML Cation channels in inflammation and immunity. Front Immunol, 2020. [DOI | PubMed]
- C. Grimm, M. Barthmes, C. Wahl-Schott. Mammalian Transient Receptor Potential (TRP) Cation Channels, 2014
- S. Nakamura, S. Shigeyama, S. Minami. LC3 lipidation is essential for TFEB activation during the lysosomal damage response to kidney injury. Nat Cell Biol, 2020. [DOI | PubMed]
- A. Galione, L.C. Davis, A.J. Morgan. A cellular protection racket: how lysosomal Ca2+ fluxes prevent kidney injury. Cell Calcium, 2021. [DOI]
- G. Li, P.-L. Li. Lysosomal TRPML1 channel: implications in cardiovascular and kidney diseases. Adv Exp Med Biol, 2021. [DOI | PubMed]
- G. Li, D. Huang, N. Li, J.K. Ritter, P.-L. Li. Regulation of TRPML1 channel activity and inflammatory exosome release by endogenously produced reactive oxygen species in mouse podocytes. Redox Biol, 2021. [DOI]
- G. Li, D. Huang, Y. Zou. Impaired autophagic flux and dedifferentiation in podocytes lacking Asah1 gene: role of lysosomal TRPML1 channel. Biochim Biophys Acta Mol Cell Res, 2023. [DOI]
- S. Prat Castro, V. Kudrina, D. Jaślan, J. Böck, A. Scotto Rosato, C. Grimm. Neurodegenerative lysosomal storage disorders: TPC2 comes to the rescue!. Cells, 2022. [DOI | PubMed]
- M. Mepyans, L. Andrzejczuk, J. Sosa. Early evidence of delayed oligodendrocyte maturation in the mouse model of mucolipidosis type IV. Dis Models Mech, 2020. [DOI]
- S. DeRosa, M. Salani, S. Smith. MCOLN1 gene therapy corrects neurologic dysfunction in the mouse model of mucolipidosis IV. Hum Mol Genet, 2021. [DOI | PubMed]
- M.I. Capurro, L.K. Greenfield, A. Prashar. VacA generates a protective intracellular reservoir for Helicobacter pylori that is eliminated by activation of the lysosomal calcium channel TRPML1. Nat Microbiol, 2019. [DOI | PubMed]
- N. Khan, K.L. Lakpa, P.W. Halcrow. BK channels regulate extracellular Tat-mediated HIV-1 LTR transactivation. Sci Rep, 2019. [DOI]
- K.D. Gibbs, L. Wang, Z. Yang. Human variation impacting MCOLN2 restricts Salmonella Typhi replication by magnesium deprivation. Cell Genomics, 2023. [DOI]
- M. Xu, X.Z. Zhong, P. Huang. TRPML3/BK complex promotes autophagy and bacterial clearance by providing a positive feedback regulation of mTOR via PI3P. Proc Natl Acad Sci U S A, 2023. [DOI]
- M.P. Cuajungco, J. Silva, A. Habibi, J.A. Valadez. The mucolipin-2 (TRPML2) ion channel: a tissue-specific protein crucial to normal cell function. Pflugers Arch Eur J Physiol, 2016. [DOI | PubMed]
- J.P. Goodridge, B. Jacobs, M.L. Saetersmoen. Remodeling of secretory lysosomes during education tunes functional potential in NK cells. Nat Commun, 2019. [DOI | PubMed]
- P. Steiner, E. Arlt, I. Boekhoff, T. Gudermann, S. Zierler. TPC functions in the immune system. Handb Exp Pharmacol, 2023. [DOI | PubMed]
- B. Spix, A. Jeridi, M. Ansari, A.Ö. Yildirim, H.B. Schiller, C. Grimm. Endolysosomal cation channels and lung disease. Cells, 2022. [DOI | PubMed]
- G. Li, D. Huang, J. Hong, O.M. Bhat, X. Yuan, P.-L. Li. Control of lysosomal TRPML1 channel activity and exosome release by acid ceramidase in mouse podocytes. Am J Physiol Cell Physiol, 2019. [DOI | PubMed]
- Z. Sui, M.-M. Wang, Y. Xing, J. Qi, W. Wang. Targeting MCOLN1/TRPML1 channels to protect against ischemia-reperfusion injury by restoring the inhibited autophagic flux in cardiomyocytes. Autophagy, 2022. [DOI | PubMed]
- Y. Xing, Z. Sui, Y. Liu. Blunting TRPML1 channels protects myocardial ischemia/reperfusion injury by restoring impaired cardiomyocyte autophagy. Basic Res Cardiol, 2022. [DOI | PubMed]
- D.L. Medina. Lysosomal calcium and autophagy. Int Rev Cell Mol Biol, 2021. [DOI | PubMed]
- L. Zhang, Y. Fang, X. Cheng, Y. Lian, H. Xu. Interaction between TRPML1 and p62 in regulating autophagosome-lysosome fusion and impeding neuroaxonal dystrophy in Alzheimer’s disease. Oxid Med Cell Longev, 2022. [DOI]
- M.B. Morelli, M. Nabissi, C. Amantini. Overexpression of transient receptor potential mucolipin-2 ion channels in gliomas: role in tumor growth and progression. Oncotarget, 2016. [DOI | PubMed]
- C. Grimm, K. Bartel, A.M. Vollmar, M. Biel. Endolysosomal cation channels and cancer-A link with great potential. Pharmaceuticals (Basel, Switzerland), 2018. [DOI | PubMed]
- J. Jung, K.J. Cho, A.K. Naji. HRAS-driven cancer cells are vulnerable to TRPML1 inhibition. EMBO Rep, 2019. [DOI]
- J. Jung, K. Venkatachalam. TRPML1 and RAS-driven cancers – Exploring a link with great therapeutic potential. Channels, 2019. [DOI | PubMed]
- J. Jung, K. Venkatachalam. TRPing the homeostatic alarm – Melanoma cells are selectively vulnerable to TRPML1 deletion. Cell Calcium, 2019. [DOI]
- S.Y. Kasitinon, U. Eskiocak, M. Martin. TRPML1 promotes protein homeostasis in melanoma cells by negatively regulating MAPK and mTORC1 signaling. Cell Rep, 2019. [DOI | PubMed]
- M.B. Morelli, C. Amantini, D. Tomassoni, M. Nabissi, A. Arcella, G. Santoni. Transient receptor potential mucolipin-1 channels in glioblastoma: role in Patient’s survival. Cancers, 2019. [DOI | PubMed]
- M. Xu, S. Almasi, Y. Yang. The lysosomal TRPML1 channel regulates triple negative breast cancer development by promoting mTORC1 and purinergic signaling pathways. Cell Calcium, 2019. [DOI | PubMed]
- P. Faris, M. Shekha, D. Montagna, G. Guerra, F. Moccia. Endolysosomal Ca2+ signalling and cancer hallmarks: two-pore channels on the move, TRPML1 lags behind. Cancers, 2018. [DOI | PubMed]
- Y. Yang, X. Zhai. TRPML1-emerging roles in cancer. Cells, 2020. [DOI | PubMed]
- C. Abrahamian, C. Grimm. Endolysosomal cation channels and MITF in melanocytes and melanoma. Biomolecules, 2021. [DOI | PubMed]
- W. Du, M. Gu, M. Hu. Lysosomal Zn2+ release triggers rapid, mitochondria-mediated, non-apoptotic cell death in metastatic melanoma. Cell Rep, 2021. [DOI]
- H. Xing, J. Tan, Y. Miao, Y. Lv, Q. Zhang. Crosstalk between exosomes and autophagy: a review of molecular mechanisms and therapies. J Cell Mol Med, 2021. [DOI | PubMed]
- Y. Xing, X. Wei, Y. Liu. Autophagy inhibition mediated by MCOLN1/TRPML1 suppresses cancer metastasis via regulating a ROS-driven TP53/p53 pathway. Autophagy, 2022. [DOI | PubMed]
- S. Rautenberg, M. Keller, C. Leser, C.-C. Chen, F. Bracher, C. Grimm. Expanding the toolbox: novel modulators of endolysosomal cation channels. Handb Exp Pharmacol, 2023. [DOI | PubMed]
- W. Wang, Q. Gao, M. Yang. Up-regulation of lysosomal TRPML1 channels is essential for lysosomal adaptation to nutrient starvation. Proc Natl Acad Sci U S A, 2015. [DOI | PubMed]
- S.W. Kim, D.H. Kim, K.S. Park. Palmitoylation controls trafficking of the intracellular Ca2+ channel MCOLN3/TRPML3 to regulate autophagy. Autophagy, 2019. [DOI | PubMed]
- J. Zou, B. Hu, S. Arpag. Reactivation of lysosomal Ca2+ efflux rescues abnormal lysosomal storage in FIG4-deficient cells. J Neurosci, 2015. [DOI | PubMed]
- J.R. Edgar, A.K. Ho, M. Laurá. A dysfunctional endolysosomal pathway common to two sub-types of demyelinating Charcot-Marie-Tooth disease. Acta Neuropathol Commun, 2020. [DOI | PubMed]
- Q. Cao, X.Z. Zhong, Y. Zou, Z. Zhang, L. Toro, X.-P. Dong. BK. BK Channels alleviate lysosomal storage diseases by providing positive feedback regulation of lysosomal Ca2+ release. Dev Cell, 2015. [DOI | PubMed]
- X.Z. Zhong, X. Sun, Q. Cao, G. Dong, R. Schiffmann. BK channel agonist represents a potential therapeutic approach for lysosomal storage diseases. Sci Rep, 2016. [DOI]
- Y. Sasazawa, S. Souma, N. Furuya. Oxidative stress-induced phosphorylation of JIP4 regulates lysosomal positioning in coordination with TRPML1 and ALG2. EMBO J, 2022. [DOI]
- L. Zhang, Y. Fang, X. Zhao. miR-204 silencing reduces mitochondrial autophagy and ROS production in a murine AD model via the TRPML1-activated STAT3 pathway. Mol Ther Nucleic Acids, 2021. [DOI | PubMed]
- A. Somogyi, E.D. Kirkham, E. Lloyd-Evans. The synthetic TRPML1 agonist, ML-SA1, rescues Alzheimer-related alterations of the endosomal-autophagic-lysosomal system. J Cell Sci, 2023. [DOI]
- M. Bae, N. Patel, H. Xu. Activation of TRPML1 clears intraneuronal Aβ in preclinical models of HIV infection. J Neurosci, 2014. [DOI | PubMed]
- L.-K. Wu, S. Agarwal, C.-H. Kuo. Artemisia Leaf Extract protects against neuron toxicity by TRPML1 activation and promoting autophagy/mitophagy clearance in both in vitro and in vivo models of MPP+/MPTP-induced Parkinson’s disease. Phytomed Int J Phytother Phytopharmacol, 2022. [DOI]
- K.P. Lee, A.V. Nair, C. Grimm. A helix-breaking mutation in the epithelial Ca(2+) channel TRPV5 leads to reduced Ca(2+)-dependent inactivation. Cell Calcium, 2010. [DOI | PubMed]
- P.P.Y. Lie, L. Yoo, C.N. Goulbourne. Axonal transport of late endosomes and amphisomes is selectively modulated by local Ca2+ efflux and disrupted by PSEN1 loss of function. Sci Adv, 2022. [DOI]
- B. Fernández, P. Olmedo, F. Gil. Iron-induced cytotoxicity mediated by endolysosomal TRPML1 channels is reverted by TFEB. Cell Death Dis, 2022. [DOI | PubMed]
- T. Busch, M. Kottgen, A. Hofherr. TRPP2 ion channels: critical regulators of organ morphogenesis in health and disease. Cell Calcium, 2017. [DOI | PubMed]
- O. Esarte Palomero, M. Larmore, P.G. DeCaen. Polycystin channel complexes. Annu Rev Physiol, 2023. [DOI | PubMed]
- C.P. Palmer, E. Aydar, M.B.A. Djamgoz. A microbial TRP-like polycystic-kidney-disease-related ion channel gene. Biochem J, 2005. [DOI | PubMed]
- T. Mochizuki, G. Wu, T. Hayashi. PKD2, a gene for polycystic kidney disease that encodes an integral membrane protein. Science, 1996. [DOI | PubMed]
- Polycystic kidney disease: the complete structure of the PKD1 gene and its protein. The International Polycystic Kidney Disease Consortium. Cell, 1995. [DOI | PubMed]
- L. Guo, T.H. Schreiber, S. Weremowicz, C.C. Morton, C. Lee, J. Zhou. Identification and characterization of a novel polycystin family member, polycystin-L2, in mouse and human: sequence, expression, alternative splicing, and chromosomal localization. Genomics, 2000. [DOI | PubMed]
- J. Hughes, C.J. Ward, R. Aspinwall, R. Butler, P.C. Harris. Identification of a human homologue of the sea urchin receptor for egg jelly: a polycystic kidney disease-like protein. Hum Mol Genet, 1999. [DOI | PubMed]
- A. Li, X. Tian, S.W. Sung, S. Somlo. Identification of two novel polycystic kidney disease-1-like genes in human and mouse genomes. Genomics, 2003. [DOI | PubMed]
- H. Nomura, A.E. Turco, Y. Pei. Identification of PKDL, a novel polycystic kidney disease 2-like gene whose murine homologue is deleted in mice with kidney and retinal defects. J Biol Chem, 1998. [DOI | PubMed]
- B. Veldhuisen, L. Spruit, H.G. Dauwerse, M.H. Breuning, D.J. Peters. Genes homologous to the autosomal dominant polycystic kidney disease genes (PKD1 and PKD2). Eur J Hum Genet, 1999. [DOI | PubMed]
- G. Wu, T. Hayashi, J.H. Park. Identification of PKD2L, a human PKD2-related gene: tissue-specific expression and mapping to chromosome 10q25. Genomics, 1998. [DOI | PubMed]
- A. Hofherr, C. Wagner, S. Fedeles, S. Somlo, M. Köttgen. N-glycosylation determines the abundance of the transient receptor potential channel TRPP2. J Biol Chem, 2014. [DOI | PubMed]
- Y. Cai, Y. Maeda, A. Cedzich. Identification and characterization of polycystin-2, the PKD2 gene product. J Biol Chem, 1999. [DOI | PubMed]
- A. Celić, E.T. Petri, B. Demeler, B.E. Ehrlich, T.J. Boggon. Domain mapping of the polycystin-2 C-terminal tail using de novo molecular modeling and biophysical analysis. J Biol Chem, 2008. [DOI | PubMed]
- A.S. Ćelić, E.T. Petri, J. Benbow, M.E. Hodsdon, B.E. Ehrlich, T.J. Boggon. Calcium-induced conformational changes in C-terminal tail of polycystin-2 are necessary for channel gating. J Biol Chem, 2012. [DOI | PubMed]
- A. Giamarchi, S. Feng, L. Rodat-Despoix. A polycystin-2 (TRPP2) dimerization domain essential for the function of heteromeric polycystin complexes. EMBO J, 2010. [DOI | PubMed]
- K. Hanaoka, F. Qian, A. Boletta. Co-assembly of polycystin-1 and −2 produces unique cation-permeable currents. Nature, 2000. [DOI | PubMed]
- M. Köttgen, T. Benzing, T. Simmen. Trafficking of TRPP2 by PACS proteins represents a novel mechanism of ion channel regulation. EMBO J, 2005. [DOI | PubMed]
- P. Koulen, Y. Cai, L. Geng. Polycystin-2 is an intracellular calcium release channel. Nat Cell Biol, 2002. [DOI | PubMed]
- E.T. Petri, A. Celic, S.D. Kennedy, B.E. Ehrlich, T.J. Boggon, M.E. Hodsdon. Structure of the EF-hand domain of polycystin-2 suggests a mechanism for Ca2+-dependent regulation of polycystin-2 channel activity. Proc Natl Acad Sci U S A, 2010. [DOI | PubMed]
- T.N. Vien, L.C.T. Ng, J.M. Smith. Disrupting polycystin-2 EF hand Ca(2+) affinity does not alter channel function or contribute to polycystic kidney disease. J Cell Sci, 2020. [DOI]
- X. Fu, Y. Wang, N. Schetle. The subcellular localization of TRPP2 modulates its function. J Am Soc Nephrol, 2008. [DOI | PubMed]
- X. Li, Y. Luo, P.G. Starremans, C.A. McNamara, Y. Pei, J. Zhou. Polycystin-1 and polycystin-2 regulate the cell cycle through the helix-loop-helix inhibitor Id2. Nat Cell Biol, 2005. [DOI | PubMed]
- O.V. Plotnikova, E.N. Pugacheva, E.A. Golemis. Aurora A kinase activity influences calcium signaling in kidney cells. J Cell Biol, 2011. [DOI | PubMed]
- A.J. Streets, D.J. Moon, M.E. Kane, T. Obara, A.C. Ong. Identification of an N-terminal glycogen synthase kinase 3 phosphorylation site which regulates the functional localization of polycystin-2 in vivo and in vitro. Hum Mol Genet, 2006. [DOI | PubMed]
- A.J. Streets, A.J. Needham, S.K. Gill, A.C. Ong. Protein kinase D-mediated phosphorylation of polycystin-2 (TRPP2) is essential for its effects on cell growth and calcium channel activity. Mol Biol Cell, 2010. [DOI | PubMed]
- A.J. Streets, O. Wessely, D.J. Peters, A.C. Ong. Hyperphosphorylation of polycystin-2 at a critical residue in disease reveals an essential role for polycystin-1-regulated dephosphorylation. Hum Mol Genet, 2013. [DOI | PubMed]
- Y. Cai, G. Anyatonwu, D. Okuhara. Calcium dependence of polycystin-2 channel activity is modulated by phosphorylation at Ser812. J Biol Chem, 2004. [DOI | PubMed]
- X.Z. Chen, P.M. Vassilev, N. Basora. Polycystin-L is a calcium-regulated cation channel permeable to calcium ions. Nature, 1999. [DOI | PubMed]
- M. Murakami, T. Ohba, F. Xu. Genomic organization and functional analysis of murine PKD2L1. J Biol Chem, 2005. [DOI | PubMed]
- W. Zheng, J. Yang, E. Beauchamp. Regulation of TRPP3 channel function by N-terminal domain palmitoylation and phosphorylation. J Biol Chem, 2016. [DOI | PubMed]
- E.Y.J. Park, M. Kwak, K. Ha, I. So. Identification of clustered phosphorylation sites in PKD2L1: how PKD2L1 channel activation is regulated by cyclic adenosine monophosphate signaling pathway. Pflugers Arch, 2018. [DOI | PubMed]
- M. Grieben, A.C. Pike, C.A. Shintre. Structure of the polycystic kidney disease TRP channel Polycystin-2 (PC2). Nat Struct Mol Biol, 2017. [DOI | PubMed]
- R.E. Hulse, Z. Li, R.K. Huang, J. Zhang, D.E. Clapham. Cryo-EM structure of the polycystin 2-l1 ion channel. eLife, 2018. [DOI]
- P.S. Shen, X. Yang, P.G. DeCaen. The structure of the polycystic kidney disease channel PKD2 in lipid nanodiscs. Cell, 2016. [DOI | PubMed]
- Q. Su, F. Hu, Y. Liu. Cryo-EM structure of the polycystic kidney disease-like channel PKD2L1. Nat Commun, 2018. [DOI | PubMed]
- M. Wilkes, M.G. Madej, L. Kreuter. Molecular insights into lipid-assisted Ca(2+) regulation of the TRP channel Polycystin-2. Nat Struct Mol Biol, 2017. [DOI | PubMed]
- T. Numata, K. Tsumoto, K. Yamada. Integrative approach with electrophysiological and theoretical methods reveals a new role of S4 positively charged residues in PKD2L1 channel voltage-sensing. Sci Rep, 2017. [DOI | PubMed]
- T.N. Vien, J. Wang, L.C.T. Ng, E. Cao, P.G. DeCaen. Molecular dysregulation of ciliary polycystin-2 channels caused by variants in the TOP domain. Proc Natl Acad Sci U S A, 2020. [DOI | PubMed]
- P.G. DeCaen, X. Liu, S. Abiria, D.E. Clapham. Atypical calcium regulation of the PKD2-L1 polycystin ion channel. eLife, 2016. [DOI]
- T. Shimizu, T. Higuchi, T. Toba. The asparagine 533 residue in the outer pore loop region of the mouse PKD2L1 channel is essential for its voltage-dependent inactivation. FEBS Open Bio, 2017. [DOI]
- M.D. Allen, S. Qamar, M.K. Vadivelu, R.N. Sandford, M. Bycroft. A high-resolution structure of the EF-hand domain of human polycystin-2. Protein Sci, 2014. [DOI | PubMed]
- K.L. Molland, L.N. Paul, D.A. Yernool. Crystal structure and characterization of coiled-coil domain of the transient receptor potential channel PKD2L1. Biochim Biophys Acta, 2012. [DOI | PubMed]
- J. Zhu, Y. Yu, M.H. Ulbrich. Structural model of the TRPP2/PKD1 C-terminal coiled-coil complex produced by a combined computational and experimental approach. Proc Natl Acad Sci U S A, 2011. [DOI | PubMed]
- Y. Yang, C. Keeler, I.Y. Kuo, E.J. Lolis, B.E. Ehrlich, M.E. Hodsdon. Oligomerization of the polycystin-2 C-terminal tail and effects on its Ca2+-binding properties. J Biol Chem, 2015. [DOI | PubMed]
- M. Semmo, M. Kottgen, A. Hofherr. The TRPP subfamily and polycystin-1 proteins. Handb Exp Pharmacol, 2014. [DOI | PubMed]
- M. Kottgen, B. Buchholz, M.A. Garcia-Gonzalez. TRPP2 and TRPV4 form a polymodal sensory channel complex. J Cell Biol, 2008. [DOI | PubMed]
- F. Qian, F.J. Germino, Y. Cai, X. Zhang, S. Somlo, G.G. Germino. PKD1 interacts with PKD2 through a probable coiled-coil domain. Nat Genet, 1997. [DOI | PubMed]
- L. Tsiokas, E. Kim, T. Arnould, V.P. Sukhatme, G. Walz. Homo- and heterodimeric interactions between the gene products of PKD1 and PKD2. Proc Natl Acad Sci U S A, 1997. [DOI | PubMed]
- A. Hofherr, M. Kottgen. Polycystic kidney disease: Cilia and mechanosensation revisited. Nat Rev Nephrol, 2016. [DOI | PubMed]
- S. Kim, H. Nie, V. Nesin. The polycystin complex mediates Wnt/Ca(2+) signalling. Nat Cell Biol, 2016. [DOI | PubMed]
- S.M. Nauli, F.J. Alenghat, Y. Luo. Polycystins 1 and 2 mediate mechanosensation in the primary cilium of kidney cells. Nat Genet, 2003. [DOI | PubMed]
- F. Qian, W. Wei, G. Germino, A. Oberhauser. The nanomechanics of polycystin-1 extracellular region. J Biol Chem, 2005. [DOI | PubMed]
- O. Ibraghimov-Beskrovnaya, N.O. Bukanov, L.C. Donohue, W.R. Dackowski, K.W. Klinger, G.M. Landes. Strong homophilic interactions of the Ig-like domains of polycystin-1, the protein product of an autosomal dominant polycystic kidney disease gene, PKD1. Hum Mol Genet, 2000. [DOI | PubMed]
- A.J. Streets, L.J. Newby, M.J. O’Hare, N.O. Bukanov, O. Ibraghimov-Beskrovnaya, A.C. Ong. Functional analysis of PKD1 transgenic lines reveals a direct role for polycystin-1 in mediating cell-cell adhesion. J Am Soc Nephrol, 2003. [DOI | PubMed]
- T. Langenhan, G. Aust, J. Hamann. Sticky signaling–adhesion class G protein-coupled receptors take the stage. Sci Signal, 2013. [DOI]
- F. Qian, A. Boletta, A.K. Bhunia. Cleavage of polycystin-1 requires the receptor for egg jelly domain and is disrupted by human autosomal-dominant polycystic kidney disease 1-associated mutations. Proc Natl Acad Sci U S A, 2002. [DOI | PubMed]
- W. Wei, K. Hackmann, H. Xu, G. Germino, F. Qian. Characterization of cis-autoproteolysis of polycystin-1, the product of human polycystic kidney disease 1 gene. J Biol Chem, 2007. [DOI | PubMed]
- X. Qu, N. Qiu, M. Wang. Structural basis of tethered agonism of the adhesion GPCRs ADGRD1 and ADGRF1. Nature, 2022. [DOI | PubMed]
- S. Pawnikar, B.S. Magenheimer, E.N. Munoz, R.L. Maser, Y. Miao. Mechanism of tethered agonist-mediated signaling by polycystin-1. Proc Natl Acad Sci U S A, 2022. [DOI]
- S. Pawnikar, B.S. Magenheimer, K. Joshi. Activation of polycystin-1 signaling by binding of stalk-derived peptide agonists. eLife, 2024. [DOI]
- Q. Su, M. Chen, Y. Wang. Structural basis for Ca(2+) activation of the heteromeric PKD1L3/PKD2L1 channel. Nat Commun, 2021. [DOI | PubMed]
- Q. Su, F. Hu, X. Ge. Structure of the human PKD1-PKD2 complex. Science, 2018. [DOI]
- Y. Yu, M.H. Ulbrich, M.H. Li. Structural and molecular basis of the assembly of the TRPP2/PKD1 complex. Proc Natl Acad Sci U S A, 2009. [DOI | PubMed]
- M. Kottgen, G. Walz. Subcellular localization and trafficking of polycystins. Pflugers Arch, 2005. [DOI | PubMed]
- H.F. Cantiello. Regulation of calcium signaling by polycystin-2. Am J Physiol Renal Physiol, 2004. [DOI | PubMed]
- T. Wegierski, D. Steffl, C. Kopp. TRPP2 channels regulate apoptosis through the Ca2+ concentration in the endoplasmic reticulum. EMBO J, 2009. [DOI | PubMed]
- Y. Li, J.M. Wright, F. Qian, G.G. Germino, W.B. Guggino. Polycystin 2 interacts with type I inositol 1,4,5-trisphosphate receptor to modulate intracellular Ca2+ signaling. J Biol Chem, 2005. [DOI | PubMed]
- P.G. DeCaen, M. Delling, T.N. Vien, D.E. Clapham. Direct recording and molecular identification of the calcium channel of primary cilia. Nature, 2013. [DOI | PubMed]
- S.J. Kleene, N.K. Kleene. The native TRPP2-dependent channel of murine renal primary cilia. Am J Physiol Renal Physiol, 2017. [DOI | PubMed]
- M. Arif Pavel, C. Lv, C. Ng. Function and regulation of TRPP2 ion channel revealed by a gain-of-function mutant. Proc Natl Acad Sci U S A, 2016. [DOI | PubMed]
- G.J. Pazour, J.T. San Agustin, J.A. Follit, J.L. Rosenbaum, G.B. Witman. Polycystin-2 localizes to kidney cilia and the ciliary level is elevated in orpk mice with polycystic kidney disease. Curr Biol, 2002. [DOI | PubMed]
- S.J. Kleene, N.K. Kleene. Inward Ca(2+) current through the polycystin-2-dependent channels of renal primary cilia. Am J Physiol Renal Physiol, 2021. [DOI | PubMed]
- X. Liu, T. Vien, J. Duan, S.H. Sheu, P.G. DeCaen, D.E. Clapham. Polycystin-2 is an essential ion channel subunit in the primary cilium of the renal collecting duct epithelium. eLife, 2018. [DOI]
- T. Staudner, L. Geiges, J. Khamseekaew, F. Sure, C. Korbmacher, A.V. Ilyaskin. Disease-associated missense mutations in the pore loop of polycystin-2 alter its ion channel function in a heterologous expression system. J Biol Chem, 2024. [DOI]
- K. Ha, M. Nobuhara, Q. Wang. The heteromeric PC-1/PC-2 polycystin complex is activated by the PC-1 N-terminus. eLife, 2020. [DOI]
- P. Delmas, S.M. Nauli, X. Li. Gating of the polycystin ion channel signaling complex in neurons and kidney cells. FASEB J, 2004. [DOI | PubMed]
- Z. Wang, C. Ng, X. Liu. The ion channel function of polycystin-1 in the polycystin-1/polycystin-2 complex. EMBO Rep, 2019. [DOI]
- K. Ha, N. Mundt-Machado, P. Bisignano. Cilia-enriched oxysterol 7β,27-DHC is required for polycystin ion channel activation. Nat Commun, 2024. [DOI | PubMed]
- S. Field, K.L. Riley, D.T. Grimes. Pkd1l1 establishes left-right asymmetry and physically interacts with Pkd2. Development, 2011. [DOI | PubMed]
- K. Kamura, D. Kobayashi, Y. Uehara. Pkd1l1 complexes with Pkd2 on motile cilia and functions to establish the left-right axis. Development, 2011. [DOI | PubMed]
- S. Yoshiba, H. Shiratori, I.Y. Kuo. Cilia at the node of mouse embryos sense fluid flow for left-right determination via Pkd2. Science, 2012. [DOI | PubMed]
- S. Yuan, L. Zhao, M. Brueckner, Z. Sun. Intraciliary calcium oscillations initiate vertebrate left-right asymmetry. Curr Biol, 2015. [DOI | PubMed]
- D. Takao, T. Nemoto, T. Abe. Asymmetric distribution of dynamic calcium signals in the node of mouse embryo during left-right axis formation. Dev Biol, 2013. [DOI | PubMed]
- F. Vetrini, L.C. D’Alessandro, Z.C. Akdemir. Bi-allelic Mutations in PKD1L1 Are Associated with Laterality Defects in Humans. Am J Hum Genet, 2016. [DOI | PubMed]
- K.A. Sutton, M.K. Jungnickel, C.J. Ward, P.C. Harris, H.M. Florman. Functional characterization of PKDREJ, a male germ cell-restricted polycystin. J Cell Physiol, 2006. [DOI | PubMed]
- T. Shimizu, T. Higuchi, T. Fujii, B. Nilius, H. Sakai. Bimodal effect of alkalization on the polycystin transient receptor potential channel, PKD2L1. Pflugers Arch, 2011. [DOI | PubMed]
- T. Shimizu, A. Janssens, T. Voets, B. Nilius. Regulation of the murine TRPP3 channel by voltage, pH, and changes in cell volume. Pflugers Arch, 2009. [DOI | PubMed]
- M. Delling, P.G. DeCaen, J.F. Doerner, S. Febvay, D.E. Clapham. Primary cilia are specialized calcium signalling organelles. Nature, 2013. [DOI | PubMed]
- P. Chen, J.Z. Wu, J. Zhao. PKD2L1/PKD1L3 channel complex with an alkali-activated mechanism and calcium-dependent inactivation. Eur Biophys J, 2015. [DOI | PubMed]
- H. Kawaguchi, A. Yamanaka, K. Uchida. Activation of polycystic kidney disease-2-like 1 (PKD2L1)-PKD1L3 complex by acid in mouse taste cells. J Biol Chem, 2010. [DOI | PubMed]
- C. Fujimoto, Y. Ishimaru, Y. Katano. The single pore residue Asp523 in PKD2L1 determines Ca2+ permeation of the PKD1L3/PKD2L1 complex. Biochem Biophys Res Commun, 2011. [DOI | PubMed]
- Y. Ishimaru, H. Inada, M. Kubota, H. Zhuang, M. Tominaga, H. Matsunami. Transient receptor potential family members PKD1L3 and PKD2L1 form a candidate sour taste receptor. Proc Natl Acad Sci USA, 2006. [DOI | PubMed]
- Y. Xiao, X. Lv, G. Cao. Overexpression of Trpp5 contributes to cell proliferation and apoptosis probably through involving calcium homeostasis. Mol Cell Biochem, 2010. [DOI | PubMed]
- A. Orts-Del’immagine, N. Wanaverbecq, C. Tardivel, V. Tillement, M. Dallaporta, J. Trouslard. Properties of subependymal cerebrospinal fluid contacting neurones in the dorsal vagal complex of the mouse brainstem. J Physiol, 2012. [DOI | PubMed]
- V. Patel, D. Williams, S. Hajarnis. miR-17∼92 miRNA cluster promotes kidney cyst growth in polycystic kidney disease. Proc Natl Acad Sci USA, 2013. [DOI | PubMed]
- H. Sun, Q.W. Li, X.Y. Lv. MicroRNA-17 post-transcriptionally regulates polycystic kidney disease-2 gene and promotes cell proliferation. Mol Biol Rep, 2010. [DOI | PubMed]
- U. Tran, L. Zakin, A. Schweickert. The RNA-binding protein bicaudal C regulates polycystin 2 in the kidney by antagonizing miR-17 activity. Development, 2010. [DOI | PubMed]
- M.M. Barr, J. DeModena, D. Braun, C.Q. Nguyen, D.H. Hall, P.W. Sternberg. The Caenorhabditis elegans autosomal dominant polycystic kidney disease gene homologs lov-1 and pkd-2 act in the same pathway. Curr Biol, 2001. [DOI | PubMed]
- M.M. Barr, P.W. Sternberg. A polycystic kidney-disease gene homologue required for male mating behaviour in C. elegans. Nature, 1999. [DOI | PubMed]
- Z. Gao, D.M. Ruden, X. Lu. PKD2 cation channel is required for directional sperm movement and male fertility. Curr Biol, 2003. [DOI | PubMed]
- A. Hofherr, C. Seger, F. Fitzpatrick. The mitochondrial transporter SLC25A25 links ciliary TRPP2 signaling and cellular metabolism. PLoS Biol, 2018. [DOI]
- M. Kottgen, A. Hofherr, W. Li. Drosophila sperm swim backwards in the female reproductive tract and are activated via TRPP2 ion channels. PLoS One, 2011. [DOI]
- T.J. Watnick, Y. Jin, E. Matunis, M.J. Kernan, C. Montell. A flagellar polycystin-2 homolog required for male fertility in Drosophila. Curr Biol, 2003. [DOI | PubMed]
- B.K. Yoder. Role of primary cilia in the pathogenesis of polycystic kidney disease. J Am Soc Nephrol, 2007. [DOI | PubMed]
- A. Hofherr, M. Kottgen. TRPP channels and polycystins. Adv Exp Med Biol, 2011. [DOI | PubMed]
- C.C. Lin, M. Kurashige, Y. Liu. A cleavage product of Polycystin-1 is a mitochondrial matrix protein that affects mitochondria morphology and function when heterologously expressed. Sci Rep, 2018. [DOI | PubMed]
- C.C. Lin, L.F. Menezes, J. Qiu. In vivo Polycystin-1 interactome using a novel Pkd1 knock-in mouse model. PLoS One, 2023. [DOI]
- L. Onuchic, V. Padovano, G. Schena. The C-terminal tail of polycystin-1 suppresses cystic disease in a mitochondrial enzyme-dependent fashion. Nat Commun, 2023. [DOI | PubMed]
- Y. Cai, S.V. Fedeles, K. Dong. Altered trafficking and stability of polycystins underlie polycystic kidney disease. J Clin Invest, 2014. [DOI | PubMed]
- X. Su, M. Wu, G. Yao. Regulation of polycystin-1 ciliary trafficking by motifs at its C-terminus and polycystin-2 but not by cleavage at the GPS site. J Cell Sci, 2015. [DOI | PubMed]
- L. Geng, D. Okuhara, Z. Yu. Polycystin-2 traffics to cilia independently of polycystin-1 by using an N-terminal RVxP motif. J Cell Sci, 2006. [DOI | PubMed]
- H. Hoffmeister, K. Babinger, S. Gurster. Polycystin-2 takes different routes to the somatic and ciliary plasma membrane. J Cell Biol, 2011. [DOI | PubMed]
- C.K. Rezi, M.G. Aslanyan, G.D. Diwan. DLG1 functions upstream of SDCCAG3 and IFT20 to control ciliary targeting of polycystin-2. EMBO Rep, 2024. [DOI | PubMed]
- A. Wyatt, P. Wartenberg, M. Candlish, G. Krasteva-Christ, V. Flockerzi, U. Boehm. Genetic strategies to analyze primary TRP channel-expressing cells in mice. Cell Calcium, 2017. [DOI | PubMed]
- M. Kottgen. TRPP2 and autosomal dominant polycystic kidney disease. Biochim Biophys Acta, 2007. [DOI | PubMed]
- B.W. Bisgrove, B.S. Snarr, A. Emrazian, H.J. Yost. Polaris and Polycystin-2 in dorsal forerunner cells and Kupffer’s vesicle are required for specification of the zebrafish left-right axis. Dev Biol, 2005. [DOI | PubMed]
- T. Obara, S. Mangos, Y. Liu. Polycystin-2 immunolocalization and function in zebrafish. J Am Soc Nephrol, 2006. [DOI | PubMed]
- P. Pennekamp, C. Karcher, A. Fischer. The ion channel polycystin-2 is required for left-right axis determination in mice. Curr Biol, 2002. [DOI | PubMed]
- G. Wu, V. D’Agati, Y. Cai. Somatic inactivation of Pkd2 results in polycystic kidney disease. Cell, 1998. [DOI | PubMed]
- M.A. Garcia-Gonzalez, P. Outeda, Q. Zhou. Pkd1 and Pkd2 are required for normal placental development. PLoS One, 2010. [DOI]
- W. Lu, B. Peissel, H. Babakhanlou. Perinatal lethality with kidney and pancreas defects in mice with a targetted Pkd1 mutation. Nat Genet, 1997. [DOI | PubMed]
- J. McGrath, S. Somlo, S. Makova, X. Tian, M. Brueckner. Two populations of node monocilia initiate left-right asymmetry in the mouse. Cell, 2003. [DOI | PubMed]
- T.A. Katoh, T. Omori, K. Mizuno. Immotile cilia mechanically sense the direction of fluid flow for left-right determination. Science, 2023. [DOI | PubMed]
- C. Boulter, S. Mulroy, S. Webb, S. Fleming, K. Brindle, R. Sandford. Cardiovascular, skeletal, and renal defects in mice with a targeted disruption of the Pkd1 gene. Proc Natl Acad Sci USA, 2001. [DOI | PubMed]
- K. Kim, I. Drummond, O. Ibraghimov-Beskrovnaya, K. Klinger, M.A. Arnaout. Polycystin 1 is required for the structural integrity of blood vessels. Proc Natl Acad Sci USA, 2000. [DOI | PubMed]
- I.Y. Kuo, A.B. Chapman. Polycystins, ADPKD, and cardiovascular disease. Kidney Int Rep, 2020. [DOI | PubMed]
- G.I. Anyatonwu, M. Estrada, X. Tian, S. Somlo, B.E. Ehrlich. Regulation of ryanodine receptor-dependent calcium signaling by polycystin-2. Proc Natl Acad Sci U S A, 2007. [DOI | PubMed]
- K.M. Márquez-Nogueras, B. Elliott, P. Thuo. Cardiac localized polycystin-2 in the natriuretic peptide signaling pathway and hypertension. J Am Soc Nephrol, 2025. [DOI | PubMed]
- S. Bulley, C. Fernandez-Pena, R. Hasan. Arterial smooth muscle cell PKD2 (TRPP1) channels regulate systemic blood pressure. eLife, 2018. [DOI]
- D. Narayanan, S. Bulley, M.D. Leo. Smooth muscle cell transient receptor potential polycystin-2 (TRPP2) channels contribute to the myogenic response in cerebral arteries. J Physiol, 2013. [DOI | PubMed]
- C.E. MacKay, M.D. Leo, C. Fernandez-Pena. Intravascular flow stimulates PKD2 (polycystin-2) channels in endothelial cells to reduce blood pressure. eLife, 2020. [DOI]
- R. Sharif-Naeini, J.H. Folgering, D. Bichet. Polycystin-1 and -2 dosage regulates pressure sensing. Cell, 2009. [DOI | PubMed]
- G. Yao, C. Luo, M. Harvey. Disruption of polycystin-L causes hippocampal and thalamocortical hyperexcitability. Hum Mol Genet, 2016. [DOI | PubMed]
- A. Orts-Del’Immagine, R. Seddik, F. Tell. A single polycystic kidney disease 2-like 1 channel opening acts as a spike generator in cerebrospinal fluid-contacting neurons of adult mouse brainstem. Neuropharmacology, 2016. [DOI | PubMed]
- J.R. Sternberg, A.E. Prendergast, L. Brosse. Pkd2l1 is required for mechanoception in cerebrospinal fluid-contacting neurons and maintenance of spine curvature. Nat Commun, 2018. [DOI | PubMed]
- A.L. Huang, X. Chen, M.A. Hoon. The cells and logic for mammalian sour taste detection. Nature, 2006. [DOI | PubMed]
- C.E. Wilson, A. Vandenbeuch, S.C. Kinnamon. Physiological and behavioral responses to optogenetic stimulation of PKD2L1(+) type III taste cells. eNeuro, 2019. [DOI]
- N. Horio, R. Yoshida, K. Yasumatsu. Sour taste responses in mice lacking PKD channels. PLoS One, 2011. [DOI]
- Y. Chen, Z. Zhang, X.Y. Lv. Expression of Pkd2l2 in testis is implicated in spermatogenesis. Biol Pharm Bull, 2008. [DOI | PubMed]
- E. Cornec-Le Gall, A. Alam, R.D. Perrone. Autosomal dominant polycystic kidney disease. Lancet, 2019. [DOI | PubMed]
- C. Bergmann, L.M. Guay-Woodford, P.C. Harris, S. Horie, D.J.M. Peters, V.E. Torres. Polycystic kidney disease. Nat Rev Dis Primers, 2018. [DOI | PubMed]
- E. Cornec-Le Gall, M.P. Audrezet, J.M. Chen. Type of PKD1 mutation influences renal outcome in ADPKD. J Am Soc Nephrol, 2013. [DOI | PubMed]
- F. Qian, G.G. Germino. "Mistakes happen": somatic mutation and disease. Am J Hum Genet, 1997. [DOI | PubMed]
- F. Qian, T.J. Watnick, L.F. Onuchic, G.G. Germino. The molecular basis of focal cyst formation in human autosomal dominant polycystic kidney disease type I. Cell, 1996. [DOI | PubMed]
- Z. Zhang, H. Bai, J. Blumenfeld. Detection of PKD1 and PKD2 somatic variants in autosomal dominant polycystic kidney cyst epithelial cells by whole-genome sequencing. J Am Soc Nephrol, 2021. [DOI | PubMed]
- Z. Wang, M. Chen, Q. Su. Molecular and structural basis of the dual regulation of the polycystin-2 ion channel by small-molecule ligands. Proc Natl Acad Sci USA, 2024. [DOI]
- X.Q. Dai, A. Ramji, Y. Liu, Q. Li, E. Karpinski, X.Z. Chen. Inhibition of TRPP3 channel by amiloride and analogs. Mol Pharmacol, 2007. [DOI | PubMed]
- P.B. Davis, U. Yasothan, P. Kirkpatrick. Ivacaftor. Nat Rev Drug Discov, 2012. [DOI | PubMed]
