Serotonergic Modulators in Alzheimer’s Disease: A Hope in the Hopeless Condition
Abstract
Alzheimer’s disease (AD) is the main cause of dementia worldwide. AD is a progressive brain neurodegenerative disease due to genetic and environmental factors that induce a progressive accumulation of intracellular hyperphosphorylated tau protein and extracellular amyloid protein (Aβ). However, anti‐AD medications cannot reverse the fundamental AD neuropathology due to amyloid plaques and related oxidative stress and inflammatory reactions. Thus, targeting other pathways might be reasonable in the management of AD. The serotonin (5‐HT) neurotransmitter plays a crucial role in preventing neurodegeneration and related oxidative stress and inflammatory reactions. In addition, the serotonergic system is highly dysregulated in many neurodegenerative diseases, including AD. Deregulation of serotonin synthesis and its receptors is involved in the pathogenesis of AD. Therefore, this review aims to discuss how the serotonergic system is affected in AD, and how 5‐HT modulators can reverse AD neuropathology and alleviate the associated neuropsychiatric disorders in AD patients.
Article type: Review Article
Keywords: Alzheimer’s disease, serotonin, 5‐HT receptors
Affiliations: Clinical Nutrition Department College of Applied Medical Sciences Umm Al‐Qura University Makkah Saudi Arabia; Department of Clinical Pharmacology and Medicine College of Medicine Mustansiriyah University Baghdad Iraq; Jabir Ibn Hayyan Medical University Kufa Iraq; Department of Biochemistry and Molecular Biology Faculty of Pharmacy Egyptian Russian University Cairo Egypt; University Centre for Research & Development Chandigarh University Chandigarh‐Ludhiana Highway Mohali Punjab India; Department of Research & Development Funogen Athens Greece; University Hospital Witten‐Herdecke University of Witten‐Herdecke Wuppertal Germany; Department of Pharmacology and Therapeutics Faculty of Veterinary Medicine Damanhour University Damanhour Egypt
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202403401 | PubMed: 40167033 | PMC: PMC12351446
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (713 KB)
Introduction
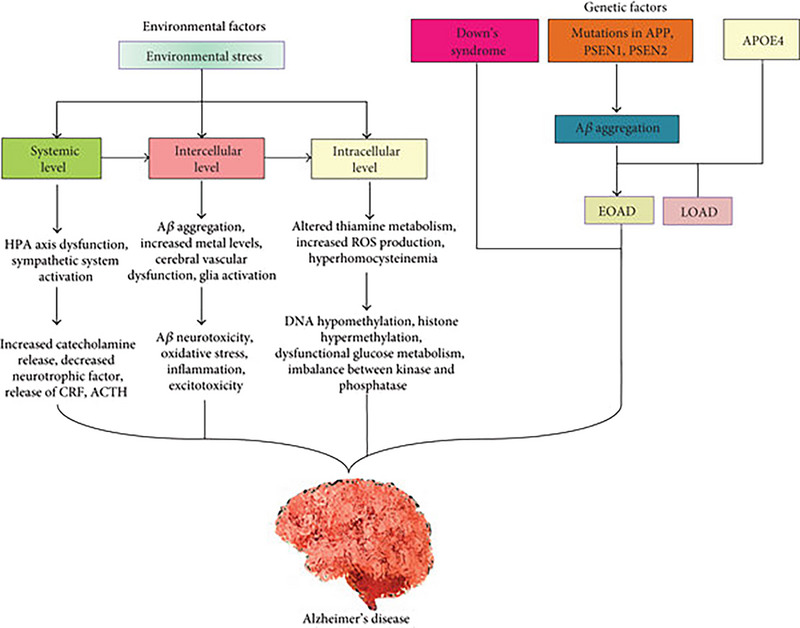
Alzheimer’s disease (AD) is considered the main cause of dementia in the elderly population around the world [ref. 1]. AD is a progressive brain neurodegenerative disease due to genetic and environmental factors that induce a progressive accumulation of intracellular hyperphosphorylated tau protein and extracellular amyloid protein (Aβ) [ref. 2] (Figure 1). The advanced deposition of hyperphosphorylated tau protein promotes the formation of neurofibrillary tangles (NFTs); however, the extracellular deposition of Aβ induces the formation of amyloid plaques. Both NFTs and amyloid plaques trigger direct neurodegeneration or indirectly by activating the release of reactive oxygen species (ROS) and proinflammatory cytokines, which induce neuronal apoptosis [ref. 3, ref. 4]. A majority of AD cases manifest as a late‐onset sporadic form, but genetically, the disease is divided into familial cases and sporadic cases. The familial form is due to mutations in three major genes (amyloid precursor protein [APP] gene, presenilin1 [PSEN1] gene, and presenilin 2 [PSEN2] gene). Familial AD, which represents 5%–10% of all AD cases, is linked with the development and progression of early‐onset AD (EOAD). However, sporadic AD, the most common type, represents 90% of all AD cases and contributes to the development of late‐onset AD (LOAD) [ref. 5, ref. 6]. Familial AD is mainly related to the overproduction of Aβ; however, sporadic AD is mainly related to the defect in the clearance of Aβ [ref. 7, ref. 8]. Interestingly, apolipoprotein A4 (ApoE4), which is involved in the regulation of brain cholesterol metabolism, is involved in the pathogenesis of EOAD and LOAD [ref. 9]. These neuropathological disorders involved in AD neuropathology distort synaptic plasticity and the release of different neurotransmitters [ref. 10]. Particularly, cholinergic neurons in the prefrontal cortex and hippocampus are mainly affected in AD, resulting in cognitive impairment and memory dysfunction [ref. 11]. Therefore, restoration of cholinergic neurotransmission by cholinergic agonists such as tacrine and donepezil could be effective in the management of AD [ref. 12]. Tacrine is a drug used in the treatment of AD as a cognitive enhancer and inhibitor of the enzyme acetylcholinesterase (AChE). However, its clinical application has been restricted due to its poor therapeutic efficacy and high prevalence of detrimental effects [ref. 12]. However, these anti‐AD medications cannot reverse the fundamental AD neuropathology due to NFTs, amyloid plaques, and related oxidative stress and inflammatory reactions [ref. 13]. Thus, targeting other pathways might be sensible in the management of AD, principally in the alleviation of NFTs and amyloid plaques and related reactions.
The serotonin (5‐HT) neurotransmitter plays a crucial role in preventing neurodegeneration and related oxidative stress and inflammatory reactions [ref. 14]. In addition, the serotonergic system is highly dysregulated in many neurodegenerative diseases, including AD [ref. 15]. Impairment of serotonin synthesis and the expression of 5‐HT receptors are implicated in the pathogenesis of AD [ref. 16]. Therefore, this review aims to discuss and explain how the serotonergic system is affected in AD, and how 5‐HT receptor agonists can reverse AD neuropathology and alleviate the associated neuropsychiatric disorders in AD patients.
Serotonin System: An Overview
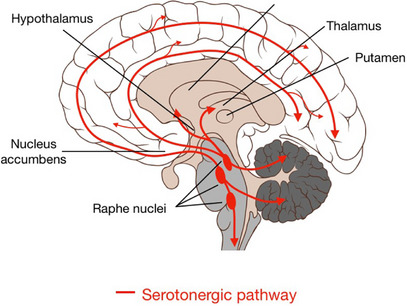
Serotonin or 5‐hydroxytryptamine (5‐HT) is a well‐known monoamine neurotransmitter involved in diverse biological functions [ref. 17]. 5‐HT was initially discovered in 1948 by Maurice M. Rapport and his coworkers at the Cleveland Clinic as a vasoconstrictor substance known as serotonin [ref. 18]. 5‐HT is involved in the regulation of mood, memory, learning, and sexual function [ref. 19]. In addition, 5‐HT has many peripheral functions such as vasoconstriction and platelet aggregation. Approximately 90% of 5‐HT in the human body is produced by enterochromaffin cells of the gastrointestinal tract, 8% by platelets and 2% in the central nervous system (CNS) [ref. 20, ref. 21]. 5‐HT in the CNS is mainly located in the raphe nuclei, which are located in the reticular formation of the brainstem [ref. 22] (Figure 2). 5‐HT from enterochromaffin cells is released into the circulation, where it is actively uptaken by platelets [ref. 23]. Under physiological conditions, released 5‐HT from platelets induces vasodilation by inducing the release of nitric oxide (NO) and inhibiting the release of norepinephrine [ref. 24]. However, during endothelial injury, platelet 5‐HT triggers vasoconstriction by contracting vascular smooth muscles [ref. 25]. Therefore, 5‐HT dysregulation is implicated in the pathogenesis of hypertension and atherosclerosis [ref. 26].
Of note, 5‐HT is synthesized from the amino acid tryptophan by tryptophan hydroxylase, which forms l‐5‐hydroxytryptophan, which is converted by aromatic l‐amino acid decarboxylase to 5‐HT [ref. 27]. In addition, 5‐HT hydroxylase catalyzes 5‐HT neurotransmitter synthesis in the CNS and the periphery [ref. 27]. 5‐HT is metabolized by monoamine oxidase (MAO) to 5‐hydroxyindolacetic acid (5‐HIAA) or stored by presynaptic vesicles [ref. 28]. The released 5‐HT in response to the action potential induces excitatory effects via activation of post‐synaptic 5‐HT receptors and controls the release of 5‐HT through activation of presynaptic receptors [ref. 29]. Furthermore, 5‐HT in the synaptic cleft is uptaken by special monoamine transporters into the presynaptic neurons [ref. 30].
5‐HT acts by activating 5‐HT receptors (Table 1), which are expressed on neurons and other cells. There are seven types of 5‐HT receptors (5‐HT1–5‐HT7), all are G‐protein coupled receptors except 5‐HT3 which is a ligand ion channel [ref. 29, ref. 31, ref. 32]. Importantly, the 5‐HT system is a target site for several drugs such as antidepressants, anxiolytics, and antipsychotics [ref. 33].
TABLE 1: List of 5‐HT receptors involved in AD disorders.
| 5‐HT receptor subtype | Role in Alzheimer’s disease | Pathophysiological implications | Potential therapeutic targets | References |
|---|---|---|---|---|
| 5‐HT1A | [ref. 116, ref. 117, ref. 118] | |||
| 5‐HT1B | [ref. 119, ref. 120] | |||
| 5‐HT1D | [ref. 121, ref. 122] | |||
| 5‐HT1F | [ref. 123, ref. 124, ref. 125] | |||
| 5‐HT2A | [ref. 126, ref. 127] | |||
| 5‐HT2B | [ref. 128, ref. 129, ref. 130] | |||
| 5‐HT3 | [ref. 131, ref. 132, ref. 133] | |||
| 5‐HT4 | [ref. 134, ref. 135, ref. 136] | |||
| 5‐HT5A | [ref. 137, ref. 138] | |||
| 5‐HT6 | [ref. 139, ref. 140, ref. 141] | |||
| 5‐HT7 | [ref. 142, ref. 143, ref. 144] |
Role of 5‐HT in AD
Mounting evidence indicates that 5‐HT can regulate neuronal proliferation, differentiation, migration, and apoptosis [ref. 34]. In addition, the serotonergic system regulates the production and clearance of misfolded proteins, including tau protein and Aβ [ref. 34, ref. 35]. It has been shown that 5‐HT is reduced and associated with synaptic dysfunction in AD [ref. 36]. Therefore, 5‐HT, as it improves memory and cognitive functions, is regarded as a therapeutic target in the management of AD [ref. 37]. A case‐control study observed that the hippocampal density of 5‐HT1 was reduced by 24% in patients with mild cognitive impairment, and by 49% in AD patients compared to healthy controls [ref. 38]. Consistently, a previous case‐control study illustrated that 5‐HT‐producing neurons in the raphe nuclei were significantly reduced compared to healthy controls. Importantly, injury of raphe nuclei and loss of 5‐HT‐producing neurons are correlated with the severity of symptoms in AD patients [ref. 39].
It has been revealed that a reduction in the expression of 5‐HT receptors is correlated with the development of depression in AD patients [ref. 40]. Of note, polymorphisms of 5‐HT2A and 5‐HT2C augment the risk of depression fivefold in patients with LOAD compared to healthy controls [ref. 41]. Thus, restoration of serotonergic signaling by selective serotonin‐reuptake inhibitors (SSRIs) could improve depressive symptoms and mitigate AD neuropathology [ref. 42].
The underlying cause for the reduction of 5‐HT levels and the expression of the 5‐HT receptor is related to AD neuropathology. It has been confirmed that Aβ and associated oxidative stress and neuroinflammation augment the expression of serotonin transporters (SERTs) in transgenic mice [ref. 43, ref. 44]. SERTs decrease the concentration of 5‐HT in the extracellular and synaptic cleft. Noristani et al. [ref. 45] found that higher concentrations of Aβ and tau protein promote the expression of SERTs in the hippocampus of transgenic mice with EOAD. However, in the LOAD with progressive neurodegeneration, the expression of SERTs is extremely reduced [ref. 46]. Furthermore, chronic low‐grade inflammation and the release of proinflammatory cytokines due to microglia activation in AD promote the development of neuroinflammation [ref. 47]. Interestingly, proinflammatory cytokines, mainly IL‐1β and TNF‐α, promote the expression of SERTs [ref. 48]. Therefore, tau protein, Aβ, and associated inflammation might be a possible mechanism for the deregulation of serotonergic neurotransmission in AD.
Activation of the 5‐HT system attenuates the aggregation of Aβ in the brain. Administration of antidepressant SSRIs reduces Aβ in brain interstitial fluid by 25%. However, chronic treatments with SSRIs decrease Aβ in the brain interstitial fluid by 50%. Similarly, direct administration of 5‐HT into the hippocampus prevents the production and accumulation of Aβ in transgenic mouse models. Consistently, findings from retrospective studies illustrated that prolonged use of SSRIs > 5 years reduced brain Aβ load measured by positron‐emitting tomography in depressed patients compared to SSRI nonusers [ref. 42, ref. 48–ref. 52]. However, there are contradictory findings regarding the effects of SSRIs on AD. For example, von Linstow et al. [ref. 53] observed that 5‐HT augmentation therapy by the SSRI escitalopram has minimal effects on Aβ levels in early‐stage AD‐like disease in mice. Remarkably, levels of insoluble Aβ40 increased in the neocortex but not in the hippocampus of SSRI‐treated mice compared with those treated with vehicle control, but they were unaffected in the hippocampus [ref. 53]. This finding suggests that modulation of the 5‐HTergic system has either no effect or increases Aβ neuropathology. Conversely, an experimental study conducted by Sheline et al. [ref. 54] observed that a single ip injection of escitalopram (5 mg/kg/day) reduced the levels of Aβ40 in the hippocampal interstitial fluid of APP/PS1 mice. Furthermore, chronic treatment with the SSRI paroxetine did not mitigate Aβ pathology and Aβ plaque‐induced microgliosis in the hippocampus of APP transgenic mice [ref. 55]. In addition, 3 months of 5‐HT depletion did not significantly impact the Aβ plaque load or Aβ42/Aβ40 ratio in APP transgenic mice. Thus, SSRIs treatment may be ineffective in the management of AD. Importantly, despite the fact that paroxetine occupies > 80% of serotonin transporters, it had no effect on the Aβ plaque load, number and size of plaques, and the Aβ plaque‐induced increases in microglial numbers in the dentate gyrus of APP transgenic mice [ref. 55]. These findings propose that factors other than dosing should be identified to clarify any discrepancies between different SSRI drugs.
These verdicts indicated that deregulation of 5‐HT synthesis and serotonergic neurotransmission is implicated in the pathogenesis of AD.
Dysregulation of 5‐HT Receptors in AD
Different studies highlighted that dysregulation of 5‐HT receptors is involved in AD neuropathology and related symptoms. In particular, selective dysregulation of 5‐HT receptors is implicated in AD and the progression of symptom severity. For example, 5‐HT1A is mainly expressed in the presynaptic neurons of the hippocampus that control cognitive and memory functions. 5‐HT1A inhibits the release of 5‐HT; therefore, pharmacological inhibition of this receptor can improve cognitive function in AD [ref. 56]. It has been shown that the hippocampal expression of 5‐HT1A is highly reduced in the early stages of AD patients compared to healthy controls [ref. 57]. Therefore, modulating the functional activity of 5‐HT1A could be an effective therapeutic strategy in the management of AD (Table 1).
Similarly, 5‐HT1B, which controls the release of 5‐HT, is reduced by the effect of APP in transgenic mice. However, the expression of SERTs and MAO are augmented, leading to the reduction of 5‐HT in the AD [ref. 58]. Furthermore, 5‐HT1B has a potent anti‐inflammatory effect, and 5‐HT1B agonists could be effective in reducing neuroinflammation in AD. 5‐HT1B agonists prevent the release of proinflammatory cytokines and the inflammatory signaling pathway ERK1/2, which mediate Aβ‐induced neurotoxicity in AD [ref. 59]. Recently, 5‐HT2B mRNA expression was widely diverse in adult microglia and was higher compared to other cortical cell subtypes. The density of 5‐HT2B was low and overall reduced in transgenic mice compared to wild‐type mice [ref. 60]. Thus, Aβ in AD reduces the affinity of 5‐HT2B binding to 5‐HT, which advocates biased agonists, rather than antagonists, might be useful for AD patients. Also, the expression of 5‐HT1D, 5‐HT1E, and 5‐HT1F is reduced in the hippocampus of AD [ref. 59, ref. 61].
Moreover, 5‐HT2A, which regulates the release of dopamine and glutamate in the prefrontal cortex, is extremely dysregulated and is associated with the alteration of dopaminergic neurotransmission in AD (Table 1) [ref. 62]. Lu et al. [ref. 63] observed that overexpression of the 5‐HT2A receptor promotes AD neuropathology. Consistently, inhibition of the 5‐HT2A receptor by the antihistamine desloratidine attenuates AD neuropathology by inhibiting neuroinflammation in transgenic mice [ref. 63]. In addition, 5‐HT2A receptor inverse agonist pimavanserin reduces Aβ‐induced neuropathology [ref. 64]. Moreover, the 5‐HT2B receptor, which has a neurotoxic effect, is involved in AD neuropathology by increasing APP processing and the production of brain Aβ. Remarkably, the expression of 5‐HT2B receptors has been reported to be augmented in AD patients compared to healthy controls [ref. 65]. In addition, the 5‐HT2C receptor, which reduces dopamine release in the mesolimbic cortex and increases the release of acetylcholine in the prefrontal cortex, is highly dysregulated in the pathogenesis of AD [ref. 66]. Activation of serotonergic neurotransmission by 5‐HT2C receptor agonists mitigates cognitive impairment in the AD model [ref. 67].
However, the expression of 5‐HT3 receptors in neurons promotes the production of Aβ in AD [ref. 68], suggesting a neurotoxic effect of this receptor. Consistently, administration of 5‐HT3 receptor antagonist for 8 weeks attenuates AD neuropathology in AD [ref. 68]. In addition, overexpression of 5‐HT3 receptors in the hippocampal GABA interneurons is correlated with cognitive dysfunction and behavioral disorders in AD [ref. 69] (Table 1). Moreover, the 5‐HT4 receptor has a neuroprotective effect against AD neuropathology by reversing the Aβ neurotoxic effect. Indeed, the 5‐HT4 receptor agonist usmarapride attenuates cognitive decline in the AD model [ref. 70] (Table 1). Findings from a preclinical study revealed that 5‐HT4 agonists reduced Aβ load by activating the non‐amyloidogenic pathway, which produces the neuroprotective soluble APPα instead of Aβ [ref. 43]. Likewise, 5‐HT4 receptor agonist prucalopride suppresses tauopathy in PS19 transgenic mice [ref. 71]. Therefore, activation of the 5‐HT4 receptor could be an important rational protocol for the future treatment of AD [ref. 70, ref. 71] (Table 1). In addition, 5‐HT5 is involved in memory consolidation and has a neuroprotective effect against AD neuropathology by modulating inflammatory signaling pathways (Table 1) [ref. 72]. Of interest is that 5‐HT6 has a complex function in the regulation of memory and cognitive function by controlling the release of acetylcholine and glutamate in AD [ref. 49]. 5‐HT6 receptor density is reduced in the cerebral cortex of AD patients and associated neuropsychiatric symptoms [ref. 50, ref. 51]. Importantly, 5‐HT6 inhibits the release of acetylcholine. Thus, 5‐HT6 antagonists have been reported by clinical trials to be effective in the management of AD [ref. 52]
Moreover, the 5‐HT7 receptor, which regulates cognitive function, is upregulated and associated with the development of psychotic symptoms in AD patients [ref. 49]. Thus, 5‐HT7 receptor antagonists may be effective in the management of neuropsychiatric symptoms in AD patients. Conversely, the 5‐HT7 receptor, which enhances hippocampal long‐term potentiation, can improve cognition in AD. A preclinical study revealed that activation of 5‐HT7 receptors improves cognitive function and long‐term potentiation in AD by inhibiting neuronal apoptosis in the hippocampus [ref. 73] (Table 1). Hence, the potential effect of the 5‐HT7 receptor seems controversial and needs to be verified by future studies.
Modulation of 5‐HT Neurotransmission in AD
It has been shown that SSRIs such as fluoxetine and sertraline, which are commonly used in the management of depression, have neuroprotective effects against neuropsychiatric disorders in AD [ref. 74, ref. 75]. Of interest, depression augments AD risk due to the development of neuroinflammation, oxidative stress, and neuronal apoptosis [ref. 76]. A longitudinal study observed that the incidence of AD was higher among patients with severe depression compared to nondepressed patients [ref. 76]. Therefore, chronic depression is regarded as a potential risk factor for the development and progression of AD [ref. 77]. Thus, early management of depression by antidepressant agents, including SSRIs could be an effective preventive measure against the development of AD (Table 2).
TABLE 2: List of 5‐HT modulators involved in AD treatment.
| Drug class | Mechanism | Efficacy on cognition | Efficacy on behavioral symptoms | Examples | References |
|---|---|---|---|---|---|
| Selective serotonin reuptake inhibitor (SSRIs) | Inhibit serotonin reuptake; increase synaptic serotonin levels | Mixed results; some improvement via depression alleviation | Effective for managing depression; mixed effects on anxiety | Citalopram, escitalopram, fluoxetine, sertraline | [ref. 145, ref. 146] |
| Serotonin agonists | Activate specific serotonin receptors (e.g., 5‐HT4 and 5‐HT6 | Potential for cognitive enhancement; ongoing studies | May improve behavioral symptoms through enhanced neurotransmission | RS 67333 (5‐HT4 agonist), SB 399885 (5‐HT6 agonist) | [ref. 147, ref. 148] |
| Serotonin antagonists | Block specific serotonin receptors (e.g., 5‐HT2A, 5‐HT7) | Limited evidence; potential for improvement | Effective for reducing psychotic symptoms; may alleviate anxiety | Clozapine (5‐HT2A antagonists), SB 699551 (5‐HT7 antagonists) | [ref. 149, ref. 150] |
Many studies confirmed that SSRIs reduce AD neuropathology by decreasing the production and the deposition of Aβ and hyperphosphorylated tau protein [ref. 78, ref. 79]. Fluoxetine and citalopram reduce Aβ production within 24 h of their administration in animal models. This finding was confirmed by the direct administration of 5‐HT into the hippocampus but not by other non‐SSRIs such as tianeptine [ref. 79]. In addition, fluoxetine reduces the production of Aβ40 and Aβ42, inhibits astrocyte activation and the development of neuroinflammation, improves synaptic plasticity by activating hippocampal neurogenesis, and attenuates the formation of NFTs. These findings indicated that elevated brain 5‐HT by SSRIs can alleviate AD neuropathology and associated cognitive decline. Citalopram reduces the early formation of Aβ rather than clearing the preexisting one in a dose‐dependent effect [ref. 80]. As well, citalopram reduces the production of the neurotoxic Aβ by activating the non‐amyloidogenic pathway through stimulation of α‐secretase [ref. 79]. Furthermore, citalopram improves synaptic plasticity and augments long‐term potentiation in animal models [ref. 81]. Besides, citalopram inhibits microglia and reduces the development of neuroinflammation, a hallmark of AD [ref. 82, ref. 83]. Conversely, SSRI paroxetine failed to decrease amyloid plaque and tauopathy in AD, which might be due to the differential occupancy of brain SERTs [ref. 84, ref. 85]. Likewise, an SSRI escitalopram, did not reduce brain amyloidosis but can reduce hippocampal tauopathy [ref. 86]. These preclinical studies suggest differential effects of SSRIs on AD neuropathology (Table 2).
Besides, clinical studies highlighted the potential effects of SSRIs against the development of AD. A cohort study found that SSRIs delay the conversion of mild cognitive impairment to symptomatic AD [ref. 87]. A longitudinal study showed that prolonged use of SSRIs, mainly sertraline, reduced AD significantly compared to SSRIs nonusers and other antidepressant users [ref. 88]. Of interest, SSRIs delays the onset of dementia in patients with Down syndrome [ref. 89]. It has been established that treatment for 2 years with SSRIs attenuates cortical atrophy and amyloid burden in patients with AD or mild cognitive impairment [ref. 90]. However, Bartels et al. [ref. 87] found that CSF Aβ1–42 levels were not affected by the effects of SSRIs, signifying that SSRIs have anti‐AD effects by another pathway not involved in Aβ neuropathology. Conversely, many clinical studies confirmed that SSRIs reduced CSF Aβ1–42 levels in patients with AD or mild cognitive impairment [ref. 91, ref. 92]. A systematic review and meta‐analysis revealed that SSRI fluoxetine has an ameliorative effect against cognitive dysfunction and AD development in patients with chronic depression [ref. 93].
Role of 5‐HT Agonists and Antagonists in AD
It has been illustrated that 5‐HT agonists, mainly of 5‐HT4 and 5‐HT6 receptors, can enhance cholinergic neurotransmission and promote non‐amyloidogenic processing of APP. Activation of 5‐HT4 receptors has been linked to improved memory and cognitive functions and reduced Aβ levels in animal models of AD [ref. 94, ref. 95]. Ongoing clinical trials evaluating the efficacy of 5‐HT agonists suggest that enhancing the 5‐HT signaling by 5‐HT agonists may improve the behavioral symptoms of AD patients [ref. 96] (Table 2).On the other hand, 5‐HT antagonists which block the action of 5‐HT at specific receptors (like 5‐HT2A and 5‐HT7) can alleviate psychotic symptoms and improve mood stability in AD patients by reducing neuroinflammation associated with AD pathology [ref. 97]. These receptors are implicated in mood regulation and cognitive processes. Furthermore, antagonizing 5‐HT7 receptors reduces anxiety and depression while potentially improving cognitive function. However, antagonists of 5‐HT2A receptors reduce hallucinations and delusions in AD patients. In addition, limited evidence suggests that 5‐HT7 antagonists could enhance cognitive performance; however, more research is needed to establish their efficacy in AD and associated neuropsychiatric disorders [ref. 97, ref. 98].
Buspirone
Buspirone is a partial agonist of the post‐synaptic 5‐HT1A receptor and antagonist of presynaptic dopamine D2–D4 that is used in the management of generalized anxiety disorders [ref. 98]. It has been revealed that buspirone improves cognitive impairment by activating 5‐HT1A receptors and increases dopamine release in the prefrontal cortex [ref. 99]. Similarly, buspirone attenuates MK‐801‐induced cognitive impairment by antagonizing D3 receptors in mice [ref. 100]. The cognitive enhancer effect of buspirone is mediated by activating the release of 5‐HT and dopamine and enhancing hippocampal neurogenesis [ref. 101, ref. 102]. In addition, 5‐HT1A receptor agonists enhance brain energy metabolism by antagonizing NMDA receptors in rats [ref. 103]. Furthermore, buspirone improves cognitive function in patients with schizophrenia [ref. 104]. Therefore, buspirone was suggested to be effective in the management of anxiety and cognitive impairment in AD patients [ref. 105]. Consistently, a retrospective study illustrated that buspirone mitigates neuropsychiatric disorders in patients with dementia, mainly AD and mixed dementia [ref. 106]. In addition, buspirone enhances adult hippocampal neurogenesis [ref. 107, ref. 108], which is highly distorted in AD. Besides, buspirone attenuates rotenone‐induced Parkinson’s disease (PD) by inhibiting neuroinflammation [ref. 109], a hallmark of AD neuropathology. However, both 5‐HT1A receptor agonists and antagonists can reduce hippocampal oxidative stress in AD [ref. 110]. This remarkable dual effect of 5‐HT1A receptor agonists and antagonists is related to the dynamic alterations of 5‐HT1A receptors in the different stages of AD [ref. 57]. Therefore, buspirone could be effective in treating AD by restoring serotonergic neurotransmission.
Trazodone
Trazodone is a mixed 5‐HT receptor agonist and antagonist, 5‐HT reuptake inhibitor, adrenergic receptor antagonist, and weak H1 receptor antagonist. In particular, trazodone is partial agonist of 5‐HT1A and antagonist of 5‐HT2A and 5‐HT2B. Trazodone is effective in the management of depression and anxiety disorders [ref. 111]. Trazodone is effective in treating sleep disturbance in AD patients [ref. 112]. A prospective study revealed that the use of trazodone for 6 months improves disinhibition and irritability in AD patients but does not enhance cognitive impairment [ref. 113]. Long‐term effect of trazodone improves cognitive decline in AD patients [ref. 114]. Moreover, the neuroprotective effect of trazodone against AD neuropathology is related to the inhibition of tau protein hyperphosphorylation in AD [ref. 115]. Therefore, trazodone, by increasing the 5‐HT neurotransmission and improving synaptic plasticity, could be effective in the management of AD.
Therefore, SSRIs and 5‐HT modulators have differential effects on AD neuropathology and associated neuropsychiatric disorders (Table 2).
Conclusions
5‐HT neurotransmission is deregulated and associated with the development of cognitive impairment in AD. In addition, dysregulation of 5‐HT receptors adversely affects the development and progression of AD by affecting the processing of Aβ and hyperphosphorylation of tau protein. Interestingly, selective agonists and antagonists of 5‐HT receptors are a promising therapeutic strategy in the management of AD and related oxidative stress and neuroinflammation. Particularly, SSRIs and 5‐HT modulators have neuroprotective effects against AD neuropathology. However, the long‐term effects of SSRIs and serotonin modulators on AD risk need to be verified by clinical trials and large‐scale clinical studies.
Author Contributions
Najlaa Hamed Almohmadi and Hayder M. Al‐kuraishy: conceptualization, data collection, and writing of the manuscript. Ali I. Al‐Gareeb: conceptualization, data collection, and writing of the manuscript, writing, supervision and editing of the manuscript. Ali K. Albuhadily: conceptualization, data collection, and writing of the manuscript. Morkoss M. Fakhry: writing, supervision and editing of the manuscript, graphical abstract. Athanasios Alexiou and Marios Papadakis: conceptualization, data collection, and writing of the manuscript. Gaber El‐Saber Batiha: writing, supervision and editing of the manuscript. All authors read and approved the final version of the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- A Potential Link Between Visceral Obesity and Risk of Alzheimer’s Disease,”. Neurochemical Research, 2023. [PubMed]
- Insights on Benzodiazepines’ Potential in Alzheimer’s Disease,”. Life Sciences, 2023. [PubMed]
- Amyloid‐β and Heart Failure in Alzheimer’s Disease: The New Vistas,”. Frontiers in Medicine, 2025. [PubMed]
- Primary Hypothyroidism and Alzheimer’s Disease: A Tale of Two,”. Cellular and Molecular Neurobiology, 2023. [PubMed]
- Long‐Term Use of Metformin and Alzheimer’s Disease: Beneficial or Detrimental Effects,”. Inflammopharmacology, 2023. [PubMed]
- An Aging, Pathology Burden, and Glial Senescence Build‐Up Hypothesis for Late Onset Alzheimer’s Disease,”. Nature Communications, 2023
- Neprilysin Inhibitors and Risk of Alzheimer’s Disease: A Future Perspective,”. Journal of Cellular and Molecular Medicine, 2024. [PubMed]
- Statins Use in Alzheimer Disease: Bane or Boon From Frantic Search and Narrative Review,”. Brain Sciences, 2022. [PubMed]
- The Potential Role of Human Islet Amyloid Polypeptide in Type 2 Diabetes Mellitus and Alzheimer’s Diseases,”. Diabetology & Metabolic Syndrome, 2023. [PubMed]
- Hypoglycemia and Alzheimer Disease Risk: The Possible Role of Dasiglucagon,”. Cellular and Molecular Neurobiology, 2024. [PubMed]
- The Cholinergic System, the Adrenergic System and the Neuropathology of Alzheimer’s Disease,”. International Journal of Molecular Sciences, 2021. [PubMed]
- Reimagining Cholinergic Therapy for Alzheimer’s Disease,”. Brain, 2022. [PubMed]
- Pathological Mechanisms and Therapeutic Strategies for Alzheimer’s Disease,”. Neural Regeneration Research, 2022. [PubMed]
- Central Nervous System Effects of 5‐HT7 Receptors: A Potential target for Neurodegenerative Diseases,”. Molecular Medicine, 2022. [PubMed]
- Highlighting Immune System and Stress in Major Depressive Disorder, Parkinson’s, and Alzheimer’s Diseases, With a Connection With Serotonin,”. International Journal of Molecular Sciences, 2021. [PubMed]
- Serotonin Receptors as a Potential Target in the Treatment of Alzheimer’s Disease,”. Biochemistry, 2023 Dec. [PubMed]
- The Mechanism of Secretion and Metabolism of Gut‐Derived 5‐Hydroxytryptamine,”. International Journal of Molecular Sciences, 2021. [PubMed]
- Serotonin,”. Current Biology, 2023. [PubMed]
- The Roles of Serotonin in Neuropsychiatric Disorders,”. Cellular and Molecular Neurobiology, 2022. [PubMed]
- Important Functions of Serotonin in the Gastrointestinal Tract,”. International Journal of Health, New Technologies and Social Work, 2023
- 21 M. Bellini , M. Fornai , P. U. Satta , et al., “The Role of Serotonin and Its Pathways in Gastrointestinal Disorders,” in The Complex Interplay Between Gut‐Brain, Gut‐Liver, and Liver‐Brain Axes , ed. C. Stasi (Academic Press, 2021), 67–94.
- Anatomical and Neurochemical Organization of the Serotonergic System in the Mammalian Brain and in Particular the Involvement of the Dorsal Raphe Nucleus in Relation to Neurological Diseases,”. Progress in Brain Research, 2021. [PubMed]
- Role of Brain Renin–Angiotensin System in Depression: A New Perspective,”. CNS Neuroscience & Therapeutics, 2024. [PubMed]
- Depression and Type 2 Diabetes: A Causal Relationship and Mechanistic Pathway,”. Diabetes, Obesity and Metabolism, 2024
- 25 P. M. Vanhoutte , “Serotonin: A Forgotten Signal From the Blood,” in Handbook of Behavioral Neuroscience, ed. C. P. Müller and K. A. Cunningham (Elsevier, 2020), 393–409.
- Antidepressants and Type 2 Diabetes: Highways to Knowns and Unknowns,”. Diabetology & Metabolic Syndrome, 2023. [PubMed]
- Construction of Cell Factory Capable of Efficiently Converting l‐Tryptophan Into 5‐Hydroxytryptamine,”. Microbial Cell Factories, 2022. [PubMed]
- Serotonin/5‐Hydroxytryptamine (5‐HT) Physiology,”. Southern African Journal of Anaesthesia and Analgesia, 2020
- Central 5‐HT Receptors and Their Function; Present and Future,”. Neuropharmacology, 2020. [PubMed]
- The Role of Serotonin in Synaptic Plasticity: Detailed Characterisation of the Effect of Serotonin on Long‐Term Potentiation Across the Hippocampus” (PhD thesis,. 2022
- 5‐HT Receptors and Temperature Homeostasis,”. Biomolecules, 2021. [PubMed]
- The Implication of 5‐HT Receptor Family Members in Aggression, Depression and Suicide: Similarity and Difference,”. International Journal of Molecular Sciences, 2022. [PubMed]
- Serotonin Receptor Activation Leads to Neurite Outgrowth and Neuronal Survival,”. Molecular Brain Research, 2005. [PubMed]
- Serotonin Receptor 2A Activation Promotes Evolutionarily Relevant Basal Progenitor Proliferation in the Developing Neocortex,”. Neuron, 2020. [PubMed]
- Serotonin Receptors as a Potential Target in the Treatment of Alzheimer’s Disease,”. Biochemistry, 2023. [PubMed]
- Serotonin: A New Hope in Alzheimer’s Disease?,”. ACS Chemical Neuroscience, 2015. [PubMed]
- Role of Serotonin in Alzheimer’s Disease,”. CNS Drugs, 2011. [PubMed]
- Serotonin 1A Receptors in the Living Brain of Alzheimer’s Disease Patients,”. Proceedings of the National Academy of Sciences of the United States of America, 2006. [PubMed]
- Brain Stem Serotonin‐Synthesizing Neurons in Alzheimer’s Disease: A Clinicopathological Correlation,”. Acta Neuropathologica, 1992. [PubMed]
- Serotonin in Depression and Alzheimer’s Disease: Focus on SSRI’s Beneficial Effects,”. Ageing Research Reviews (, 2024. [PubMed]
- Depression in Alzheimer’s Disease: The Effect of Serotonin Receptor Gene Variation,”. American Journal of Medical Genetics Part B: Neuropsychiatric Genetics, 2003
- Serotonin Signaling is Associated With Lower Amyloid‐β Levels and Plaques in Transgenic Mice and Humans,”. Proceedings of the National Academy of Sciences of the United States of America, 2011. [PubMed]
- 5‐HT4 Receptor Agonist Mediated Enhancement of Cognitive Function In Vivo and Amyloid Precursor Protein Processing In Vitro: A Pharmacodynamic and Pharmacokinetic Assessment,”. Neuropharmacology, 2011. [PubMed]
- Neuroinflammation and Amyloid‐beta 40 are Associated With Reduced Serotonin Transporter (SERT) Activity in a Transgenic Model of Familial Alzheimer’s Disease,”. Alzheimer’s Research & Therapy, 2019
- Increased Hippocampal CA1 Density of Serotonergic Terminals in a Triple Transgenic Mouse Model of Alzheimer’s Disease: An Ultrastructural Study,”. Cell Death & Disease, 2011. [PubMed]
- Ascending Monoaminergic Systems Alterations in Alzheimer’s Disease. Translating Basic Science Into Clinical Care,”. Neuroscience and Biobehavioral Reviews, 2013. [PubMed]
- Inflammasome NLRP3 Potentially Links Obesity‐Associated Low‐Grade Systemic Inflammation and Insulin Resistance With Alzheimer’s Disease,”. International Journal of Molecular Sciences, 2021. [PubMed]
- Interleukin‐1 Receptor Activation by Systemic Lipopolysaccharide Induces Behavioral Despair Linked to MAPK Regulation of CNS Serotonin Transporters,”. Neuropsychopharmacology, 2010. [PubMed]
- 5‐HT7 Receptors in Alzheimer’s Disease,”. Neurochemistry International, 2021. [PubMed]
- Differential Involvement of 5‐HT1B/1D and 5‐HT6 Receptors in Cognitive and Non‐Cognitive Symptoms in Alzheimer’s Disease,”. Neuropsychopharmacology, 2004. [PubMed]
- Involvement of an Altered 5‐HT6 Receptor Function in Behavioral Symptoms of Alzheimer’s Disease,”. Journal of Alzheimer’s Disease, 2008
- 5‐HT6 Receptor Antagonists as Novel Cognitive Enhancing Agents for Alzheimer’s Disease,”. Neurotherapeutics, 2008. [PubMed]
- Serotonin Augmentation Therapy by Escitalopram has Minimal Effects on Amyloid‐β Levels in Early‐Stage Alzheimer’s‐Like Disease in Mice,”. Alzheimer’s Research & Therapy, 2017
- Reply to Comment on “An Antidepressant Decreases CSF Aβ Production in Healthy Individuals and in Transgenic AD Mice”,”. Science Translational Medicine, 2014. [DOI]
- Efficacy of Chronic Paroxetine Treatment in Mitigating Amyloid Pathology and Microgliosis in APPSWE/PS1ΔE9 Transgenic Mice,”. Journal of Alzheimer’s Disease, 2022
- Agonist and Antagonist Bind Differently to 5‐HT1A Receptors During Alzheimer’s Disease: A Post‐Mortem Study With PET Radiopharmaceuticals,”. Neuropharmacology, 2016. [PubMed]
- Hippocampal 5‐HT1A Receptor Expression Changes in Prodromal Stages of Alzheimer’s Disease: Beneficial or Deleterious?,”. Neuropharmacology, 2017. [PubMed]
- 5‐HT1B and Other Related Serotonergic Proteins Are Altered in APPswe Mutation,”. Neuroscience Letters, 2015. [PubMed]
- Distribution of 5‐HT 1E Receptors in the Mammalian Brain and Cerebral Vasculature: An Immunohistochemical and Pharmacological Study,”. British Journal of Pharmacology, 2012. [PubMed]
- Serotonin‐2B Receptor (5‐HT2BR) Expression and Binding in the Brain of APPswe/PS1dE9 Transgenic Mice and in Alzheimer’s Disease Brain Tissue,”. Neuroscience Letters, 2025. [PubMed]
- The Role of Hippocampal 5‐HT1D and 5‐HT1F Receptors on Learning and Memory in Rats,”. Naunyn‐Schmiedeberg’s Archives of Pharmacology, 2023. [PubMed]
- Alterations in Dopamine System and in Its Connectivity With Serotonin in a Rat Model of Alzheimer’s Disease,”. Brain Communications, 2021. [PubMed]
- Antiallergic Drug Desloratadine as a Selective Antagonist of 5HT 2A Receptor Ameliorates Pathology of Alzheimer’s Disease Model Mice by Improving Microglial Dysfunction,”. Aging Cell, 2021. [PubMed]
- Pimavanserin, a 5HT2A Receptor Inverse Agonist, Rapidly Suppresses Aβ Production and Related Pathology in a Mouse Model of Alzheimer’s Disease,”. Journal of Neurochemistry, 2021. [PubMed]
- The 5HT2b Receptor in Alzheimer’s Disease: Increased Levels in Patient Brains and Antagonist Attenuation of Amyloid and Tau Induced Dysfunction,”. Journal of Alzheimer’s Disease, 2024
- Dual 5‐HT2A and 5‐HT2C Receptor Inverse Agonist That Affords In Vivo Antipsychotic Efficacy With Minimal hERG Inhibition for the Treatment of Dementia‐Related Psychosis,”. Journal of Medicinal Chemistry, 2024. [PubMed]
- Serotonin 5‐HT2C Receptor Agonism in Presymptomatic APP/PS1 Mice Rescues Seizure‐Induced Premature Mortality Perhaps We Just Plainly State What Is Happening,”. Alzheimer’s & Dementia, 2024
- Inhibiting 5‐Hydroxytryptamine Receptor 3 Alleviates Pathological Changes of a Mouse Model of Alzheimer’s Disease,”. Neural Regeneration Research, 2023. [PubMed]
- Amygdala Neuronal Dyshomeostasis via 5‐HT Receptors Mediates Mood and Cognitive Defects in Alzheimer’s Disease,”. Aging Cell, 2024. [PubMed]
- Rational Design of Novel Benzisoxazole Derivatives With Acetylcholinesterase Inhibitory and Serotoninergic 5‐HT4 Receptors Activities for the Treatment of Alzheimer’s Disease,”. Scientific Reports, 2020. [PubMed]
- 5‐HT4 Receptor Agonists Treatment Reduces Tau Pathology and Behavioral Deficit in the PS19 Mouse Model of Tauopathy,”. Frontiers in Cellular Neuroscience, 2024. [PubMed]
- Amyloid β Oligomers Disrupt Piriform Cortical Output via a Serotonergic Pathway,”. Neurobiology of Aging, 2023. [PubMed]
- The Effects of the 5‐HT7 Receptor on Hippocampal Long‐Term Potentiation and Apoptosis in a Rat Model of Alzheimer’s Disease,”. Brain Research Bulletin, 2017. [PubMed]
- Potential Cognitive Enhancing and Disease Modification Effects of SSRIs for Alzheimer’s Disease,”. Neuropsychiatric Disease and Treatment, 2007. [PubMed]
- Depression and Type 2 Diabetes: A Causal Relationship and Mechanistic Pathway,”. Diabetes, Obesity and Metabolism, 2024
- Depression and Incident Alzheimer Disease: The Impact of Disease Severity,”. American Journal of Geriatric Psychiatry, 2015
- Depression is a Risk Factor for Alzheimer Disease‐Review,”. Research Journal of Pharmacy and Technology, 2015
- Selective Serotonin Reuptake Inhibitors and Alzheimer’s Disease,”. Neural Regeneration Research, 2020. [PubMed]
- Serotonin Signaling is Associated With Lower Amyloid‐β Levels and Plaques in Transgenic Mice and Humans,”. Proceedings of the National Academy of Sciences of the United States of America, 2011. [PubMed]
- An Antidepressant Decreases CSF Aβ Production in Healthy Individuals and in Transgenic AD Mice,”. Science Translational Medicine, 2014
- Citalopram Ameliorates Impairments in Spatial Memory and Synaptic Plasticity in Female 3xTgAD Mice,”. BioMed Research International, 2017. [PubMed]
- Citalopram Restores Short‐Term Memory Deficit and Non‐Cognitive Behaviors in APP/PS1 Mice While Halting the Advance of Alzheimer’s Disease‐Like Pathology,”. Neuropharmacology, 2018. [PubMed]
- Regulation of Astrocyte Pathology by Fluoxetine Prevents the Deterioration of Alzheimer Phenotypes in an APP/PS1 Mouse Model,”. Glia, 2016. [PubMed]
- Long‐Term Ameliorative Effects of the Antidepressant Fluoxetine Exposure on Cognitive Deficits in 3 × TgAD Mice,”. Molecular Neurobiology, 2017. [PubMed]
- Established Amyloid‐β Pathology is Unaffected by Chronic Treatment With the Selective Serotonin Reuptake Inhibitor Paroxetine,”. Alzheimer’s & Dementia, 2018
- Escitalopram Attenuates β‐Amyloid‐Induced Tau Hyperphosphorylation in Primary Hippocampal Neurons Through the 5‐HT1A Receptor Mediated Akt/GSK‐3β Pathway,”. Oncotarget, 2016. [PubMed]
- Impact of SSRI Therapy on Risk of Conversion From Mild Cognitive Impairment to Alzheimer’s Dementia in Individuals With Previous Depression,”. American Journal of Psychiatry, 2018. [PubMed]
- Decreasing Hazards of Alzheimer’s Disease With the Use of Antidepressants: Mitigating the Risk of Depression and Apolipoprotein E,”. International Journal of Geriatric Psychiatry, 2018. [PubMed]
- Effects of Antidepressants on Longevity and Dementia Onset Among Adults With Down Syndrome,”. Journal of Clinical Psychiatry, 2014. [PubMed]
- Serotonin Selective Reuptake Inhibitor Treatment Improves Cognition and Grey Matter Atrophy but not Amyloid Burden During Two‐Year Follow‐Up in Mild Cognitive Impairment and Alzheimer’s Disease Patients With Depressive Symptoms,”. Journal of Alzheimer’s Disease, 2018
- The Effect of Citalopram Treatment on Amyloid‐β Precursor Protein Processing and Oxidative Stress in Human hNSC‐Derived Neurons,”. Translational Psychiatry, 2022. [PubMed]
- Antidepressants in Alzheimer’s Disease: A Focus on the Role of Mirtazapine,”. Pharmaceuticals, 2021. [PubMed]
- Emerging Therapeutic Potential of Fluoxetine on Cognitive Decline in Alzheimer’s Disease: Systematic Review,”. International Journal of Molecular Sciences, 2024. [PubMed]
- An Expanded Narrative Review of Neurotransmitters on Alzheimer’s Disease: The Role of Therapeutic Interventions on Neurotransmission,”. Molecular Neurobiology, 2025. [PubMed]
- Multi‐Target‐Directed Ligands Affecting Serotonergic Neurotransmission for Alzheimer’s Disease Therapy: Advances in Chemical and Biological Research,”. Current Medicinal Chemistry, 2017
- Serotonin 5‐HT 6 Receptor Antagonists in Alzheimer’s Disease: Therapeutic Rationale and Current Development Status,”. CNS Drugs, 2017. [PubMed]
- Serotonin Degeneration and Amyloid‐β Deposition in Mild Cognitive Impairment: Relationship to Cognitive Deficits,”. Journal of Alzheimer’s Disease, 2023
- Assessing the Efficacy and Safety of Combined Buspirone and Venlafaxine Treatment in Late‐Life Depression Accompanied by Cognitive Impairment: A Randomized Controlled Trial,”. General Hospital Psychiatry, 2024. [PubMed]
- Improving Cognition in Schizophrenia With Antipsychotics That Elicit Neurogenesis Through 5‐HT1A Receptor Activation,”. Neurobiology of Learning and Memory, 2014. [PubMed]
- Buspirone Counteracts MK‐801‐Induced Schizophrenia‐Like Phenotypes Through Dopamine D3 Receptor Blockade,”. Frontiers in Pharmacology, 2017. [PubMed]
- Dose Related Effects of Buspirone on Pain, Learning/Memory and Food Intake,”. Regulatory Toxicology and Pharmacology, 2018. [PubMed]
- Chronic Treatment With the 5‐HT1A Receptor Partial Agonist Tandospirone Increases Hippocampal Neurogenesis,”. Neurology and Therapy, 2014. [PubMed]
- Tandospirone, a 5‐HT1A Partial Agonist, Ameliorates Aberrant Lactate Production in the Prefrontal Cortex of Rats Exposed to Blockade of N‐Methy‐d‐Aspartate Receptors; Toward the Therapeutics of Cognitive Impairment of Schizophrenia,”. Frontiers in Behavioral Neuroscience, 2014. [PubMed]
- Augmentation Therapy With Serotonin1A Receptor Partial Agonists on Neurocognitive Function in Schizophrenia: A Systematic Review and Meta‐Analysis,”. Schizophrenia Research: Cognition, 2023. [PubMed]
- Buspirone in Alzheimer’s Disease,”. Expert Review of Neurotherapeutics, 2003. [PubMed]
- Buspirone for the Treatment of Dementia With Behavioral Disturbance,”. International Psychogeriatrics, 2017. [PubMed]
- An Exploratory Study of Combination Buspirone and Melatonin SR in Major Depressive Disorder (MDD): A Possible Role for Neurogenesis in Drug Discovery,”. Journal of Psychiatric Research, 2012. [PubMed]
- The Partial 5‐HT1A Receptor Agonist Buspirone Enhances Neurogenesis in the Opossum (Monodelphis domestica),”. European Neuropsychopharmacology, 2009. [PubMed]
- The Anxiolytic Drug Buspirone Prevents Rotenone‐Induced Toxicity in a Mouse Model of Parkinson’s Disease,”. International Journal of Molecular Sciences, 2022. [PubMed]
- Protective Effects of 5‐HT1A Receptor Antagonist and 5‐HT2A Receptor Agonist on the Biochemical and Histological Features in a Rat Model of Alzheimer’s Disease,”. Journal of Chemical Neuroanatomy, 2019. [PubMed]
- Trazodone: A Multifunctional Antidepressant. Evaluation of Its Properties and Real‐World Use,”. Journal of Gerontology and Geriatrics, 2021
- Trazodone Improves Sleep Parameters in Alzheimer Disease Patients: A randomized, Double‐Blind, and Placebo‐Controlled Study,”. American Journal of Geriatric Psychiatry, 2014
- Trazodone for Alzheimer’s Disease: A naturalistic Follow‐Up Study,”. Archives of Gerontology and Geriatrics, 2008. [PubMed]
- The Effects of Trazodone on Human Cognition: A Systematic Review,”. European Journal of Clinical Pharmacology, 2021. [PubMed]
- The Antidepressant Drug; Trazodone Inhibits Tau Amyloidogenesis: Prospects for Prophylaxis and Treatment of AD,”. Archives of Biochemistry and Biophysics, 2020. [PubMed]
- Role of Receptors in Relation to Plaques and Tangles in Alzheimer’s Disease Pathology,”. International Journal of Molecular Sciences, 2021. [PubMed]
- Anxiety and Alzheimer’s Disease Pathogenesis: Focus on 5‐HT and CRF Systems in 3xTg‐AD and TgF344‐AD Animal Models,”. Frontiers in Aging Neuroscience, 2023. [PubMed]
- Serotonergic Dysfunction May Mediate the Relationship Between Alcohol Consumption and Alzheimer’s Disease,”. Pharmacological Research, 2024. [PubMed]
- Glycogen Synthase Kinase‐3 Beta (GSK‐3β) Signaling: Implications for Parkinson’s Disease,”. Pharmacological Research, 2015. [PubMed]
- The Role of Glycogen Synthase Kinase 3 Beta in Neurodegenerative Diseases,”. Frontiers in Molecular Neuroscience, 2023. [PubMed]
- The Role of Hippocampal 5‐HT1D and 5‐HT1F Receptors on Learning and Memory in Rats,”. Naunyn‐Schmiedeberg’s Archives of Pharmacology, 2023. [PubMed]
- 5‐HT1D Receptors Inhibit the Monosynaptic Stretch Reflex by Modulating C‐Fiber Activity,”. Journal of Neurophysiology, 2019. [PubMed]
- Dysautonomia in the Pathogenesis of Migraine,”. Expert Review of Neurotherapeutics, 2018. [PubMed]
- Migraine: From Pathophysiology to Treatment,”. Journal of Neurology, 2023. [PubMed]
- Revisiting Migraine: The Evolving Pathophysiology and the Expanding Management Armamentarium,”. Cureus, 2023. [PubMed]
- Zinc and Central Nervous System Disorders,”. Nutrients, 2023. [PubMed]
- Strategies for Treatment of Disease‐Associated Dementia Beyond Alzheimer’s Disease: An Update,”. Current Neuropharmacology, 2022
- Implication of 5‐HT2B Receptors in the Serotonin Syndrome,”. Neuropharmacology, 2011. [PubMed]
- Developmentally Regulated Serotonin 5‐HT 2B Receptors,”. International Journal of Developmental Neuroscience, 2001. [PubMed]
- Serotonin and the 5‐HT2B Receptor in the Development of Enteric Neurons,”. Journal of Neuroscience, 2000. [PubMed]
- Role of 5‐HT3 Receptors in the Antidepressant Response,”. Pharmaceuticals, 2011
- 132 S. M. Mesoy and S. C. R. Lummis , “5‐HT3 Receptors,” in Textbook of Ion Channels Volume II: Properties, Function, and Pharmacology of the Superfamilies, ed. J. Zheng and M. C. Trudeau (CRC Press, 2023), 265–278.
- 5‐HT3 Receptors” (. 2016
- 5‐HT4 Receptors Constitutively Promote the Non‐Amyloidogenic Pathway of APP Cleavage and Interact With ADAM10,”. ACS Chemical Neuroscience, 2013. [PubMed]
- The Serotonin 5‐HT4 Receptor and the Amyloid Precursor Protein Processing,”. Experimental Gerontology, 2003. [PubMed]
- Chronic 5‐HT4 Receptor Activation Decreases Aβ Production and Deposition in hAPP/PS1 Mice,”. Neurobiology of Aging, 2013. [PubMed]
- Evaluation of Association of Common Variants in HTR1A and HTR5A With Schizophrenia and Executive Function,”. Scientific Reports, 2016. [PubMed]
- 5‐ht5A Receptors as a Therapeutic Target,”. Pharmacology & Therapeutics, 2006. [PubMed]
- 5‐HT6 Receptors: A Novel Target for Cognitive Enhancement,”. Pharmacology & Therapeutics, 2005. [PubMed]
- Differential Involvement of 5‐HT1B/1D and 5‐HT6 Receptors in Cognitive and Non‐Cognitive Symptoms in Alzheimer’s Disease,”. Neuropsychopharmacology, 2003
- Management of Dementia‐Related Psychosis, Agitation and Aggression: A Review of the Pharmacology and Clinical Effects of Potential Drug Candidates,”. CNS Drugs, 2020. [PubMed]
- The Role of Neurotransmitter Receptors in Antipsychotic Medication Efficacy for Alzheimer’s‐Related Psychosis,”. Egyptian Journal of Neurology, Psychiatry and Neurosurgery, 2024
- Chemical Update on the Potential for Serotonin 5‐HT6 and 5‐HT7 Receptor Agents in the Treatment of Alzheimer’s Disease,”. Bioorganic & Medicinal Chemistry Letters, 2021. [PubMed]
- 5‐HT7 Receptors in Alzheimer’s Disease,”. Neurochemistry International, 2021. [PubMed]
- Unraveling the Serotonergic Mechanism of Stress‐Related Anxiety: Focus on Co‐Treatment With Resveratrol and Selective Serotonin Reuptake Inhibitors,”. Biomedicines, 2024
- Cognitive Behavioral Therapy Combined With Selective Serotonin Reuptake Inhibitors for Premature Ejaculation: A Systematic Review and Meta‐Analysis,”
- 147 J. Tanveer , A. Baig , R. Rubeen , et al., “Unlocking the Mysteries: Serotonin Receptor Networks Explored,” in Serotonin—Neurotransmitter Horm Brain, Bowels Blood, K. Fatima‐Shad (IntechOpen, 2024).
- 5‐HT4 Receptor Agonist Effects on Functional Connectivity in the Human Brain: Implications for Procognitive Action,”. Biological Psychiatry: Cognitive Neuroscience and Neuroimaging, 2023. [PubMed]
- Receptors Involved in Mental Disorders and the Use of Clozapine, Chlorpromazine, Olanzapine, and Aripiprazole to Treat Mental Disorders,”. Pharmaceuticals, 2023
- Obstructive Sleep Apnea and Serotoninergic Signalling Pathway: Pathomechanism and Therapeutic Potential,”. International Journal of Molecular Sciences, 2024
- Dissecting Sex‐Related Cognition between Alzheimer’s Disease and Diabetes: From Molecular Mechanisms to Potential Therapeutic Strategies.”. Oxidative Medicine and Cellular Longevity, 2021. [PubMed]
- Molecular imaging of the human emotion circuit.”. Social and Affective Neuroscience of Everyday Human Interaction (, 2020
