Nonconventional Techniques in Plant Alkaloid Extraction: A Decade of Progress (2014–2023)
Abstract
Plant metabolism encompasses primary and secondary pathways, with secondary metabolism yielding diverse natural products crucial for plant adaptation and ecosystem interactions. Among these products, plant‐derived alkaloids are bioactive compounds of significant pharmacological interest, traditionally extracted using conventional solvents known for their environmental and health hazards. Historically, alkaloid extraction relied on acid–base methods, which separate these compounds based on their solubility under different pH conditions. Since the 19th century, extraction techniques have evolved from traditional methods to modern approaches such as supercritical fluid extraction (SFE), microwave‐assisted extraction (MAE), ultrasound‐assisted extraction (UAE), and the use of ionic liquids (ILs) and deep eutectic solvents (DESs). These advanced methods optimize yield, reduce extraction times, and enhance sustainability by minimizing solvent use and environmental impact. This review explores the evolution of alkaloid extraction methodologies from 2014 to 2023, emphasizing eco‐friendly techniques. It discusses the principles and applications of SFE, MAE, UAE, ILs, and DESs in extracting alkaloids from plants, highlighting their pivotal role in modern natural product chemistry. These advancements underscore ongoing efforts to develop efficient and sustainable practices in alkaloid extraction, which is essential for pharmaceutical and medicinal applications.
Article type: Review Article
Keywords: alkaloids, green solvents, microwave extraction, plant species, supercritical fluid extraction
Affiliations: Programa de Pós‐Graduação em Biologia Química, Instituto de Ciências Ambientais Químicas e Farmacêuticas Universidade Federal de São Paulo Diadema Brazil; Instituto de Ciências Ambientais, Químicas e Farmacêuticas Universidade Federal de São Paulo Diadema Brazil; Departamento de Química, Instituto de Ciências Ambientais Químicas e Farmacêuticas, Universidade Federal de São Paulo Diadema Brazil
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202403225 | PubMed: 40117174 | PMC: PMC12351439
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.9 MB)
Introduction
Plant metabolism is divided into primary and secondary pathways. Primary metabolism includes essential processes like glycolysis and the Krebs cycle, which produce energy, reducing equivalents, and fundamental biomolecules (carbohydrates, proteins, lipids, and nucleic acids). In contrast, secondary metabolism involves compounds not directly linked to energy production or structural functions. These metabolites vary among plant species, contributing to their uniqueness [ref. 1].
Plant‐derived compounds play a crucial role in adapting of plants to their environments, enabling them to interact with diverse ecosystems effectively. These molecules, collectively known as natural products (NPs), enhance a species’ survival by serving various biological functions. For instance, they can act as antibiotics, antifungals, and antivirals, protecting plants from pathogens. In addition, they may exhibit anti‐germination properties or toxic activities toward other plants, serving as phytoalexins. Moreover, certain metabolites function as UV protectants, absorbing ultraviolet light and thereby preventing damage to leaves [ref. 2]. Secondary metabolites are typically classified based on their biosynthetic pathways. The main families of these molecules include phenolic compounds, terpenes/steroids, and alkaloids.
Alkaloids exhibit remarkable structural diversity and are synthesized through various biosynthetic pathways, being present in about 20% of known vascular plants [ref. 3]. According to the Dictionary of Natural Products database (https://dnp.chemnetbase.com/chemical/ChemicalSearch.xhtml?dswid=‐733), over 49 600 natural alkaloids—encompassing those found in both fungi and plants—have been identified and cataloged. This vast diversity highlights the significance of alkaloids in nature and their potential applications in various fields, particularly in drug discovery and development [ref. 4].
These NPs are low molecular weight compounds, typically characterized by their alkaline nature due to the presence of a heterocyclic ring containing a nitrogen atom, which contributes to their pharmacological activity. The term “alkaloid” was first introduced by W. Meissner, an apothecary from Halle, in 1819. He noted that these compounds resembled alkalis in appearance and thus named them alkaloids [ref. 5]. Friedrich Sertürner, an apothecary’s assistant from Westphalia, achieved the first isolation of morphine (1) Figure 1, a pivotal alkaloid, in 1805, marking a significant milestone in chemistry and pharmacology. Subsequently, pharmacists Pierre Joseph Pelletier and Joseph Benaimé Caventou utilized Sertürner’s methodology to isolate a diverse array of alkaloids between 1817 and 1821. Among these were notable compounds such as brucine (2), febrifuge (3), quinine (4), caffeine (5), and veratrine (6) [ref. 6].
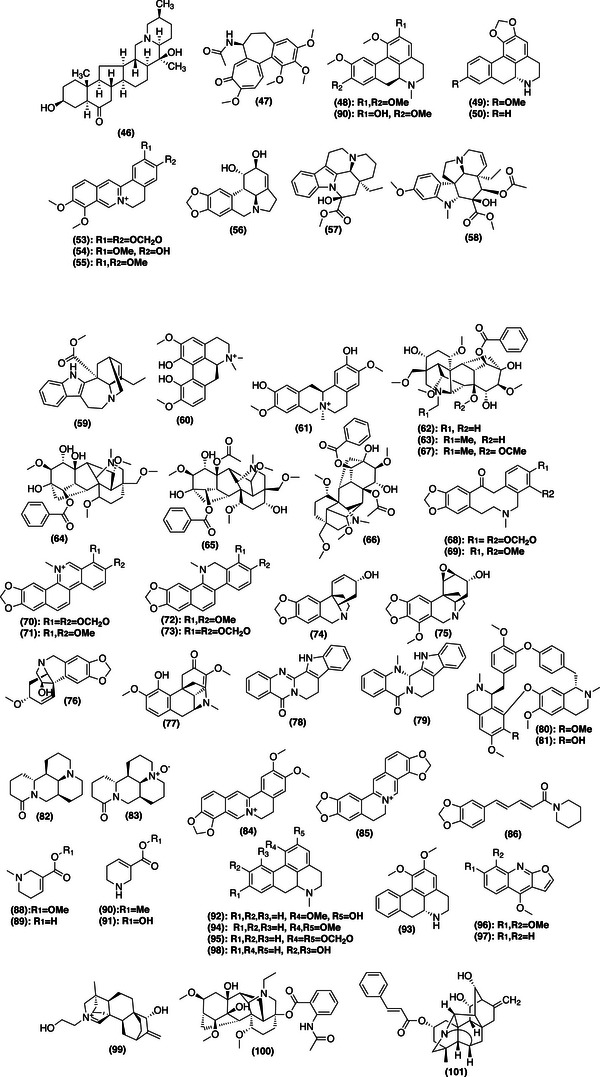
FIGURE 1Chemical structures of natural products isolated from plant material through diverse extraction techniques.
Alkaloids are classified into true alkaloids, pseudoalkaloids, or protoalkaloids based on their structural and biosynthetic features. True alkaloids are characterized by having nitrogen incorporated into a heterocyclic ring system, which originates from amino acid biosynthesis. Pseudoalkaloids, on the other hand, incorporate nitrogen from amino acids through transamination reactions. However, their carbon skeleton derives from pathways other than amino acid biosynthesis. Protoalkaloids originate from N‐methylation and decarboxylation reactions of amino acids. In these compounds, the nitrogen atom is not part of a heterocyclic system but is instead incorporated through methylation and subsequent decarboxylation processes. These classifications reflect the diverse biosynthetic pathways and structural characteristics that define alkaloids in nature [ref. 7, ref. 8, ref. 9, ref. 10].
The Vital Role of Alkaloids in Pharmacology
Plant extracts containing alkaloids have historically been employed empirically as medicines, poisons, and “magic potions.” Dating back to 4000 bc, Sumerians used opium for its sedative and analgesic effects. Similarly, civilizations in the Americas—including the Incas, Aztecs, Mayans, Olmecs, and Toltecs—contributed to medicinal knowledge by using alkaloid‐containing substances such as (4), ipecacuanha, and coca for various therapeutic purposes [ref. 11].
Before 1800, most drugs were prepared as tinctures, decoctions, or infusions using water‐based or water/ethanol plant extracts. While some of these preparations were highly effective or toxic, their precise chemical compositions remained unknown. However, during the 19th century, advancements in organic chemistry and chemical analysis enabled the isolation and characterization of several compounds from plants [ref. 12]. Following the purification of (1), Pelletier and Caventou achieved notable milestones by isolating strychnine (7) from the seeds of Asian Strychnos trees in 1818. They also successfully extracted (4) from Cinchona in 1820 [ref. 13, ref. 14].
In this scenario, the main plant families that biosynthesize alkaloids are Amaryllidaceae, Apocynaceae, Asteraceae, Berberidaceae, Boraginaceae, Elaeagnaceae, Erythroxylaceae, Fabaceae, Liliaceae, Loganiaceae, Menispermaceae, Papaveraceae, Ranunculaceae, Rubiaceae, Rutaceae, Solanaceae, and Zygophyllaceae [ref. 5]. However, alkaloids can also be found in other biological sources, indicating their widespread occurrence and significance in nature. The use of medicinal plants for treating various types of diseases has become increasingly popular in society. Since ancient times, plants have been used to ease pain and cure diseases long before the development of modern medicines [ref. 15].
Armand Séquin and Friedrich Sertürner were pioneers in the study and application of purified NPs [ref. 15], with one of their most important discoveries being morphine (1), an alkaloid known for its sedative effects and use in pain relief. Morphine is derived from opium, which comes from Papaver somniferum (the opium poppy) [ref. 15, ref. 16, ref. 17, ref. 18]. Their work led to the identification and isolation of other alkaloids from opium, including codeine (8), thebaine (9), and narcotine (10), which contributed to the creation of the opioid drug class, recognized for its powerful analgesic properties [ref. 15].
Alkaloids also play an essential role in the treatment of diseases like cancer. For example, vinblastine (11) and vincristine (12), extracted from Catharanthus roseus (a member of the Apocynaceae family), are used in cancer therapies [ref. 19, ref. 20]. Cocaine (13), derived from Erythroxylum coca, has been used as a local anesthetic since the 19th century [ref. 21], though its toxicity and addictive nature led to the search for safer alternatives, such as topacocaine (14), which offers similar effects with reduced toxicity.
In recent years, camptothecin (15), extracted from Camptotheca acuminata, has shown anti‐tumor properties by inhibiting topoisomerase I, a crucial enzyme in tumor cells [ref. 22]. In addition, Atropa belladonna yields atropine (16) and scopolamine (17), the latter being the main component of Buscopan, used for its analgesic and sedative properties in treating abdominal pain and cramps [ref. 23]. Ephedrine (18), extracted from Ephedra, is used for its sympathomimetic effects, though it can have adverse cardiovascular effects, and it is often used by athletes to enhance performance [ref. 24]. These historical examples highlight the significance of studying plant‐derived alkaloids due to their vast therapeutic potential. Despite some alkaloids’ toxicity, their contributions to medical therapies are invaluable [ref. 25].
Fundamentals of Classical Alkaloid Extraction From Plants: A Comprehensive Overview
Extraction plays a crucial role in separating and recovering bioactive compounds from plant material, thereby transforming the matrix into a sample suitable for analysis. Traditional techniques such as decoction, maceration, infusion, digestion, and percolation, known since the 11th century, represent some of the oldest employed methods. In the 18th century, the introduction of the “Soxhlet extraction” technique integrated aspects of digestion and decoction methods [ref. 26, ref. 27].
The limitations of traditional extraction methods prompted the development of numerous methods for extracting plant‐derived alkaloids [ref. 28]. Therefore, alkaloids are traditionally extracted from crude plant extracts using acid–base extraction methods. This method exploits the fact that alkaloids can exist in two forms: as ionic ammonium salts or as neutral free bases, depending on the pH conditions [ref. 29]. In plant extracts, these compounds are typically found as polar salts of organic acids, making them soluble in polar solvents such as methanol. Conversely, the free base forms of most alkaloids are insoluble in water but can be effectively extracted using nonpolar solvents such as benzene, dichloromethane, diethyl ether, chloroform, or ethyl acetate [ref. 30].
In recent decades, unconventional extraction methods, designed to be more environmentally friendly, have been introduced. These methods aim to reduce the use of synthetic and organic chemicals, decrease operating time, and improve both extract yield and quality. Examples include ultrasound‐assisted extraction (UAE), microwave‐assisted extraction (MAE), and supercritical fluid extraction (SFE) [ref. 31]. In addition, green solvents such as ionic liquids (ILs) and deep eutectic solvents (DESs) are being employed to replace traditional hazardous solvents widely used in industry [ref. 27].
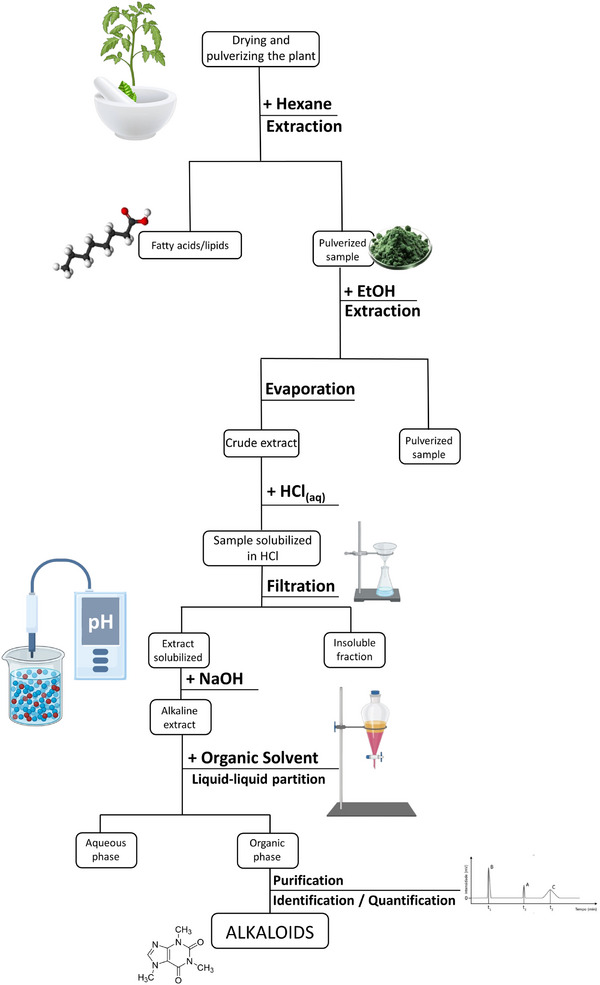
Acid–base extraction remains a core technique for isolating alkaloids from plants, which is crucial in both organic chemistry and pharmaceuticals [ref. 32]. This method, dating back to the early days of modern chemistry, stems from significant breakthroughs like morphine isolation [ref. 33], the extraction of quinine [ref. 34], and the discovery of harmaline (19) from Peganum harmala [ref. 35]. While efficient and versatile, the method has some drawbacks, such as the potential for compound degradation or artifact formation due to its multi‐step nature [ref. 36, ref. 37]. The process typically involves using nonpolar solvents to extract lipids, followed by polar solvents to extract alkaloids, which are then acidified and alkalinized to convert them into their free base form (Figure 2). Despite its effectiveness, further advancements are needed to improve the efficiency of acid–base extraction [ref. 38, ref. 39, ref. 40]. Based on this context, this review offers a comprehensive exploration of plant metabolism, with a particular emphasis on the critical roles of secondary metabolites. It focuses on plant‐derived alkaloids, a key class of NPs, highlighting their pharmacological importance and their impact on medicine, from early isolations to modern therapeutic agents. In addition, it covers advancements in extraction methods, emphasizing the shift toward modern techniques from 2014 to 2023.
Materials and Methods
To track and investigate the most recent methodologies for extracting plant‐derived alkaloids from 2014 to 2023, an extensive literature survey was performed using the following databases: SciFindern, Web of Science, Scopus, and PubMed. The selection of keywords aimed to refine and enrich our strategy, ensuring more objective and relevant results. The descriptors chosen for this survey are listed below, and the Boolean term “AND” was employed among the descriptors. The Rayyan platform (https://www.rayyan.ai/) efficiently assisted in the removal of duplicate data.
- “Microwave plant alkaloid extraction”
- “Supercritical fluid plant alkaloid extraction”
- “Ionic liquid plant alkaloid extraction”
- “Deep eutectic solvent plant alkaloid extraction.”
Results and Discussion
Modern extraction techniques, such as SFE, MAE, UAE, pressurized liquid extraction (PLE), and DESs, have revolutionized NP chemistry by enhancing efficiency and sustainability. These methods offer advantages over traditional techniques: SFE uses supercritical CO2 for high yields with minimal solvents, promoting green chemistry. MAE and UAE use energy waves to break plant cell walls, accelerating extraction and improving compound recovery. PLE employs high pressure and temperature to increase solvent penetration. DESs, biodegradable and nontoxic, effectively dissolve diverse NPs. Historically, SFE gained prominence in the 1980s, MAE in 1986, and UAE in the 20th century. ILs were explored in 1982, while DESs emerged in 2003, with natural deep eutectic solvents (NADESs) following in 2011 [ref. 41, ref. 42, ref. 43, ref. 44, ref. 45, ref. 46, ref. 47, ref. 48, ref. 49, ref. 50, ref. 51, ref. 52, ref. 53, ref. 54, ref. 55, ref. 56, ref. 57]. These innovations optimize phytochemical extraction while aligning with sustainability goals. The discussion will further explore their role in obtaining bioactive plant‐derived alkaloids.
Highlights and Trends of Recent Advancements in Extraction Techniques
Based on MAE strategies, initially, our survey identified a total of 201 articles across the searched databases. After applying exclusion criteria, we selected 12 articles that specifically detailed methodologies for obtaining alkaloids, primarily focusing on extracting bioactive alkaloids from plants. Most of these investigations aimed to enhance extraction efficiency through comparative methodologies, often employing microwave‐assisted or ultrasound‐assisted methods. These studies involved the extraction, isolation, and identification of 24 different alkaloids using MAE strategies. Some studies did not specify the identified alkaloids, while others focused on extracting multiple metabolites. Before conducting full‐scale extractions, optimization of extraction methods was performed using various parameters to identify the most efficient conditions.
In our database search on supercritical fluids, we consulted 17 articles that investigated methodologies using supercritical fluids to extract diverse classes of alkaloids. Most studies aimed to optimize methods and compare them with traditional techniques of alkaloid extraction. These studies identified and isolated 22 alkaloids, although specific identifications varied among publications, with some studies focusing on multiple alkaloid extractions. Optimization of extraction methods with varied parameters preceded the comprehensive extraction process to achieve optimal efficiency. For ILs extraction methods, we reviewed nine articles detailing methodologies for extracting different classes of alkaloids. Moreover, since 2017, we have observed a notable increase in the use of eutectic solvents for extracting plant‐derived alkaloids. A total of 21 studies have been published on this approach, covering the extraction of 44 alkaloids. Since the inception of using these solvents in alkaloid extraction, there has been at least one study involving DESs per year. Notably, although more prevalent overall, NADESs have demonstrated greater prominence since 2018.
To provide a clearer overview of the annual publication trends, Figure 3 shows the number of publications per year related to the various extraction methods discussed in this review. As the graph illustrates, in 2014, the only topics with publications were MAE and SFE. From 2015 onward, research interests diversified, with new topics like ILs being explored. In 2018, the focus shifted as NADES emerged, while other topics saw a decline in activity. Beginning in 2020, DES started gaining more attention, with NADES becoming particularly prominent in 2023.
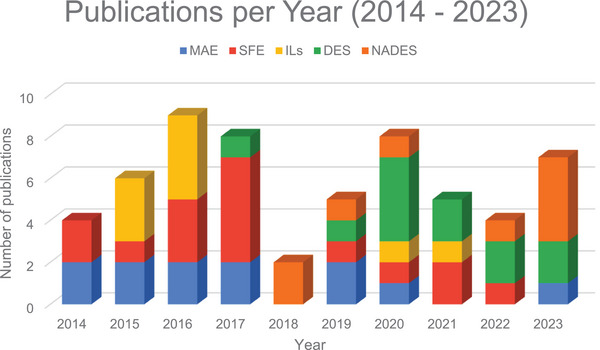
Over the years, publications on MAE have remained relatively steady, showing only slight fluctuations. Although there was a minor decline in 2021 and 2022, this trend reversed in 2023. SFE saw a gradual increase in interest, peaking in 2017, but the number of related publications declined afterward. ILs were a popular topic between 2015 and 2016, but interest dropped sharply in subsequent years, with only one publication in both 2020 and 2021. DES reached a peak in 2020, reflecting a growing interest during that period, but experienced a slight decline thereafter. This decline could be attributed to the significant growth in publications on NADES in 2023, which suggests a potential shift in focus and an increase in relevance or interest in this topic.
Overall, these trends reflect the dynamic nature of the field, with research interests evolving over time. New methodologies and solvents are continuously being explored and evaluated for their efficiency and applicability in extracting valuable plant‐derived alkaloids. The shift in focus suggests that while some topics gain prominence, others decline, highlighting the ongoing search for more effective and sustainable extraction methods.
Alkaloids Extraction Assisted by Microwave (MAE) and Ultrasound (UAE)
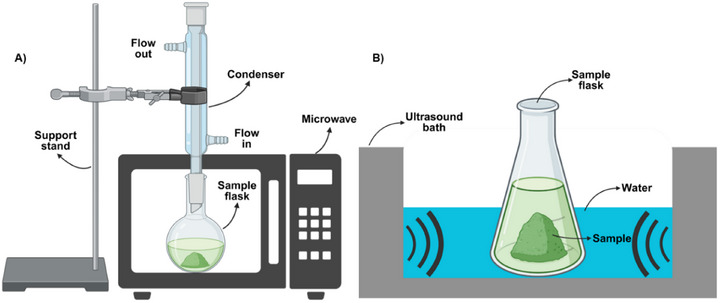
In the MAE method, electromagnetic radiation is utilized to enhance extraction kinetics and improve extract quality. Microwave energy, absorbed by polar compounds based on their dielectric constant, dissipates as heat, enabling targeted heating of specific metabolites in plant samples for more efficient extraction. Traditional methods such as solvent extraction, leaching, and hydrodistillation are often deemed inefficient and time‐consuming due to the degradation of active metabolites. Effective pre‐extraction and extraction procedures are critical for processing bioactive constituents from plants [ref. 58, ref. 59]. The food industry is actively seeking new extraction techniques to address safety concerns, environmental regulations, and the reduction of waste and energy consumption [ref. 60]. Modern methods like MAE and UAE have proven to be more efficient, cost‐effective, and faster compared to conventional solvent extraction methods. Emerging technologies such as UAE, microwave‐assisted techniques, and PLE offer sustainable and “green” solutions, yielding high‐purity extracts while reducing solvent and energy usage. In addition, UAE simplifies handling and processing, enhances product purity, and reduces solvent and energy requirements by operating at lower temperatures or eliminating the need for solvents [ref. 61, ref. 62]. A schematic illustration of MAE (a) and UAE (b) methods is shown in Figure 4.
Based on the studies by Desgrouas et al. [ref. 63] and Liu al. [ref. 64], both publications focused on optimizing the extraction of bisbenzylisoquinoline alkaloids from Stephania species using MAE strategies. Several parameters, such as power levels, solvents, solid–liquid ratios, temperatures, and extraction times to enhance the extraction of cepharanthine (20) from Stephania rotunda Lour. [ref. 63]. The authors identified ethanol–water (50:50, v/v) with a solid–liquid ratio of 1/20 (w/v), operated at 100 W for 15 min at 80°C, as the optimal conditions, achieving high efficiency and reducing extraction time compared to the UAE (Table 1). In contrast, focusing on the extraction and purification of the alkaloids (20), cycleanine (21), and isotetrandrine (22) from Stephania cepharantha, a MAE combined with solid‐phase extraction (SPE) (MAE‐SPE) approach was employed. The optimized method involved heating plant material with 0.01 mol/L HCl at 100 W and 60°C for 2 min (Table 1), followed by purification using a polymeric cation exchange‐SPE (PCX‐SPE) cartridge, providing specific yields for each compound, demonstrating the efficacy of the approach in extracting and purifying alkaloids from the species [ref. 64]. Both studies underscore the effectiveness of MAE in enhancing extraction efficiency while reducing environmental impact compared to traditional methods.
TABLE 1: Summary of study conditions for alkaloid extraction using microwave‐assisted extraction (MAE) and ultrasound‐assisted extraction (UAE) techniques.
| Target alkaloid(s) | Plant source | Technique | Optimized conditions | References |
|---|---|---|---|---|
| Cepharanthine (20) | Stephania rotunda Lour. | MAE | Ethanol–water (50:50, v/v); solid–liquid ratio 1:20 (w/v); 100 W; 15 min; 80°C | Desgrouas et al. [ref. 63] |
| Cycleanine (21), isotetrandrine (22), cepharanthine (20) | Stephania cepharantha | MAE‐SPE | 0.01 mol/L HCl; 100 W; 2 min; 60°C | Liu et al. [ref. 64] |
| Humantenidine (23), humantenmine (24), gelsemine (25), koumine (26) | Gelsemium elegans | UAE/MAE | Methanol; 3–10 min, 1–3 extraction cycles; 40°C–52°C | Li et al. [ref. 65] |
| Diverse alkaloids | Sophora flavescens | MAATPE | Ethanol/ammonium sulfate; 780 W; 5 min; 90°C | Zhang et al. [ref. 66] |
| Solasodine (27) | Solanum nigrum L. | MAATEPE | Ethanol, ammonium sulfate, water (28:16:56, w/w/w) | Sakti et al. [ref. 67] |
| Bianfugedine (28), menisporphine (29), 6‐O‐demethylmenisporphine (30), bainfugecine (31), dauriporphine (32), dauriporphinoline (33) | Menispermum dauricum | MAE | 70% ethanol; liquid–solid ratio 20 mL/g; 11 min; 60°C | Wei et al. [ref. 68] |
| Harmine (34), harmaline (19), vasicine (35) | Peganum harmala | MAE | 80% ethanol (v/v); 0.2 g plant material; 600 W; 8 min; 80°C | Shang et al. [ref. 69] |
| Camptothecin (15) | Nothapodytes nimmoniana | MAE | Methanol; solid–liquid ratio1:50 g/mL; 150 W; 40°C | Patil et al. [ref. 70] |
| Betacyanins | Gomphrena globose | MAE/UAE | Ethanol; solid‐to‐liquid ratio 5 g/L; 8 min; 60°C | Roriz et al. [ref. 71] |
| Ricinine (36) | Ricinus communis | MAE | 10% ethyl acetate in methanol; liquid–solid ratio 25 mL/g; 1000 W; 15 min; 175°C | Nebo et al. [ref. 72] |
| Vasicine (35), vasicinone (37) | Justicia adhatoda | MAE | Methanol; 1 g dried sample in 40 mL solvent; 1200 W; 6 min | Padhiari et al. [ref. 73] |
| Carpaine (38) | Carica papaya | MAE | 80% aqueous ethanol; 1 kg powdered leaves; 800 W; 20 min; 60°C | Mandour et al. [ref. 74] |
Li et al. [ref. 65] explored UAE/MAE methods to extract four indole alkaloids from Gelsemium elegans: humantenidine (23), humantenmine (24), gelsemine (25), and koumine (26). The optimized extraction parameters included solvent type, extraction time, temperature, liquid‐to‐solid ratio, and number of extraction cycles. Methanol was found to be the most effective solvent, yielding the highest concentrations of all alkaloids compared to acetonitrile, ethanol, and ethyl acetate. The study showed that increasing the extraction temperature from 40°C to 52°C, extending extraction time from 3 to 10 min, and performing 1–3 extraction cycles led to significant improvements in alkaloid yields under the optimized conditions [ref. 65].
Zhang et al. [ref. 66] introduced microwave‐assisted aqueous two‐phase extraction (MAATPE) as a novel method to extract alkaloids from Sophora flavescens. They combined MAE with aqueous two‐phase extraction (ATPE), using ethanol/ammonium sulfate as the solvent. Operating at 90°C and 780 W for 5 min, this approach yielded 63.78 ± 0.45 mg/g of alkaloids with a recovery rate of 92.09 ± 0.14%, showing significant improvement in yield and purity compared to traditional methods [ref. 66].
Building on this, Sakti et al. [ref. 67] also applied MAATPE to extract solasodine (27) from Solanum nigrum L. They optimized their method using ethanol, ammonium sulfate, and water (28:16:56, w/w/w) as the aqueous two‐phase system (ATPS), determined through response surface methodology (RSM). Their results demonstrated higher extraction yields and reduced extraction times compared to conventional approaches, highlighting the efficiency of MAATPE in extracting bioactive metabolites [ref. 67].
In addition, a method for the simultaneous quantification of six oxoisoaporphine alkaloids extracted from Menispermum dauricum: bianfugedine (28), menisporphine (29), 6‐O‐demethylmenisporphine (30), bainfugecine (31), dauriporphine (32), and dauriporphinoline (33). Optimal extraction conditions, determined through orthogonal experiments, involved using 70% ethanol as the solvent at 60°C for 11 min, with a solvent/solid ratio of 20 mL/g. This resulted in a total alkaloid content yield of 232.8 µg/g [ref. 68].
Three bioactive alkaloids—harmaline (19), harmine (34), and vasicine (35)—were simultaneously extracted and determined from P. harmala seeds using an efficient method. The extraction involved using 0.2 g of plant material with 6 mL of 80% ethanol (v/v) at 600 W and 80°C for 8 min. This process yielded 38.7 mg/g of (19), 17.3 mg/g of (34), and 5.9 mg/g of (35) [ref. 69].
The extraction of (15) from Nothapodytes nimmoniana was also investigated [ref. 70]. Solvents such as acetone, chloroform, ethanol, and methanol were used to optimize extraction conditions, including a solid‐to‐liquid ratio of 1:50 g/mL, 150 W microwave power, 40°C temperature, and particle size range of 0.84–0.42 mm. Methanol provided the highest extraction efficiency, yielding 3.349 ± 0.007 mg/g due to its favorable dielectric constant, viscosity, and polarity index. In conclusion, the extraction time was minimized to 2 min, and kinetic studies emphasized the substantial influence of stirring, power level, and temperature on extraction efficiency.
Roriz et al. [ref. 71] optimized the extraction of betacyanins from Gomphrena globosa using RSM with a combination of MAE and UAE. The optimal conditions—8 min extraction time, 60°C, 0% ethanol content, and a solid‐to‐liquid ratio of 5 g/L—yielded 39.6 ± 1.8 mg/g of betacyanins [ref. 71]. Another study was performed to extract ricinine (36) from Ricinus communis leaves. The optimal conditions—175°C, 1000 W, 15 min, 10% ethyl acetate in methanol, and a liquid‐to‐solid ratio of 25 mL/g—yielded approximately 1.5 mg/g of (36) per gram of dried leaves in 15 min [ref. 72].
In 2020, Padhiari et al. optimized a method to simultaneously quantify (35) and vasicinone (37) from different parts of Justicia adhatoda using MAE with methanol. They irradiated a 1 g dried sample in 40 mL of solvent at 1200 W for 6 min. This method outperformed maceration, ultrasonic‐assisted extraction, and heat reflux extraction in efficiency. Yields of (35) and (37) were highest in the leaf (6.39 and 3.60 mg/g, respectively), followed by the stem (4.00 and 1.89 mg/g) and the root (1.23 and 0.73 mg/g) [ref. 73]. A similar approach was used to enhance the extraction of carpaine (38) from Carica papaya leaves, resulting in an extraction efficiency of 18.75%. (38) was subsequently isolated as pure crystals from the leaf extracts, showcasing MAE’s ability to efficiently extract and purify specific compounds from plants [ref. 74].
MAE and UAE offer distinct advantages and considerations in extracting bioactive compounds, particularly plant‐derived alkaloids, MAE utilizes electromagnetic radiation to selectively “fish” compounds, speeding up extraction kinetics and enhancing yield and purity. This method reduces solvent use and energy consumption. However, MAE requires precise optimization of parameters like power levels and extraction time to avoid heat‐induced degradation of sensitive compounds. Conversely, UAE utilizes sound waves to disrupt plant cell structures, facilitating the release of intracellular compounds with minimal energy and time. Operating at lower temperatures, UAE preserves thermally sensitive compounds and ensures a safer process. Yet, it may necessitate longer extraction times and careful control of sonication to prevent sample degradation or emulsification. Both techniques significantly improve extraction efficiency and purity, advancing applications in pharmaceuticals, food, and cosmetics industries. Their sustainable and effective extraction capabilities make MAE and UAE valuable tools in modern extraction methodologies.
Alkaloids Extraction by Supercritical Fluid
SFE, particularly with CO2 as the solvent, offers significant advantages in the extraction of alkaloids. It utilizes CO2 above its critical temperature and pressure to achieve high selectivity and efficiency in extracting target compounds. SFE minimizes the use of organic solvents, making it suitable for pharmaceutical and research applications. However, SFE requires precise optimization of parameters such as temperature, pressure, and cosolvent use to achieve good yields and maintain compound stability. Compared to traditional methods like maceration and UAE, SFE often yields lower quantities but produces more purified extracts, thereby streamlining downstream purification processes.
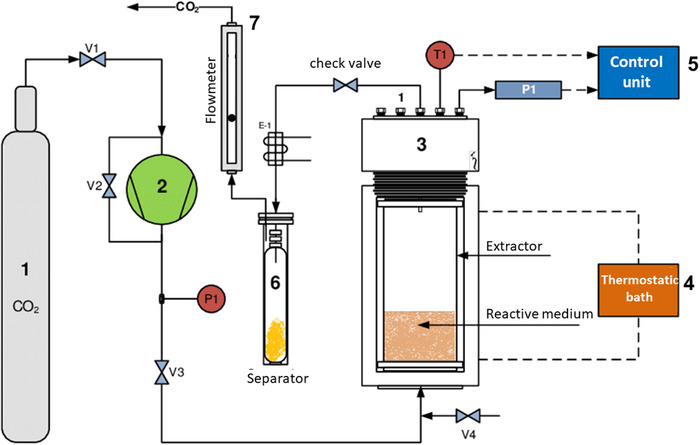
Several studies (Table 2) illustrate the application of SFE (scCO2) and other advanced techniques in extracting specific alkaloids from natural sources, showcasing their potential for efficient extraction and yield optimization in pharmaceutical and research applications. A schematic illustration of an extraction unit is shown in Figure 5.
TABLE 2: Summary of study conditions for alkaloid extraction using supercritical fluid extraction.
| Target alkaloid(s) | Plant source | Optimized conditions | References |
|---|---|---|---|
| Galanthamine (39) | Narcissus pseudonarcissus | 25% (v/v) NH4OH; 70°C; 220 bar; 3 h | Rachmaniah et al. [ref. 75] |
| Lycopodine (42) | Lycopodium clavatum | 300 bar; 40°C | Da Silva et al. [ref. 76] |
| Cyclopamine (43) | Veratrum californicum | SFE using copper coil setup yielded 0.66 mg/g | Turner et al. [ref. 77] |
| Chelidonine (44) | Chelidonium majus | Temperature, solvent density, and basified cosolvents varied; SFE followed by ESE | Gañán et al. [ref. 78] |
| Betacyanins | Hylocereus polyrhizus | scCO2 with 10% of cosolvents; 50°C; 25 MPa | Fathordoobady et al. [ref. 79] |
| Pentacyclic alkaloids | Uncaria tomentosa | Calvo et al. [ref. 80] | |
| Diverse alkaloids | Melocactus zehntneri | 300 bar; 35°C | Brandão et al. [ref. 81] |
| Diverse alkaloids | Fritillaria thunbergii | 90.3% ethanol; 61.3°C; 30.6 MPa; 2.9 h | Ruan et al. [ref. 82] |
| Colchicine (47) | Gloriosa superba | Liquid CO2 with 3% water as cosolvent; 60°C; 400 bar; 2 h | Balkrishna et al. [ref. 83] |
| Aporphine alkaloids | Annona cherimola | 100 bar; 75°C; 15% methanol as cosolvent | Galarce‐Bustos et al. [ref. 84] |
| Diverse alkaloids | Papaver bracteatum | 351 atm pressure; 90 mL modifier volume (9% v/w); 40°C; 15 min static, 55 min dynamic extraction | Salehi et al. [ref. 85] |
| Methylxanthines | Yerba mate leaves | Hegel et al. [ref. 86] | |
| Diverse alkaloids | Sophora moorcroftiana | 31 MPa pressure; 70°C; 2.7 h | Hu et al. [ref. 87] |
Rachmaniah et al. investigated the extraction of galanthamine (39) from Narcissus pseudonarcissus bulbs [ref. 75], focusing on optimizing extraction conditions such as particle size, CO2 density (temperature and pressure), flow rate, and plant material treatment. Ten alkaloids were identified in the scCO2 extracts, including (39), lycoramine (40), and norgalanthamine (41). The highest (39) yield (303 µg/g) was achieved at 70°C and 220 bar over a 3‐h period, using plant material with a particle size of 53–1000 µm moistened with 25% (v/v) of aqueous ammonia. Utilizing RSM, another study extracted lycopodine (42) from Lycopodium clavatum [ref. 76]. Optimal conditions were 300 bar and 40°C without pretreatment, resulting in 20.29% of (42). From Veratrum californicum, Turner et al. investigated various extraction techniques for obtaining cyclopamine (43) [ref. 77]. Eight methods were compared, including Soxhlet reflux, ethanol and benzene soaks, SFE, and MAE. The supercritical fluid method, using a copper coil setup, yielded 0.66 mg/g of (43), comparable to other methods but lower than that achieved with ethanol soak (8.03 mg/g).
Gañán et al. investigated both SFE and enhanced solvent extraction (ESE) methods for fractionating extracts from Chelidonium majus aerial and terrestrial parts [ref. 78]. They studied the effects of temperature, solvent density, and basified cosolvents on extraction yield, kinetics, and composition, comparing these methods with traditional approaches like Soxhlet and low‐pressure solvent extraction. Their findings highlighted SFE’s initial selectivity for chelidonine (44) rich extracts, followed by an ESE step to enhance overall yields, albeit with a dilution of alkaloid concentration. Fathordoobady et al. assessed the feasibility of SFE for extracting betacyanin pigments from Hylocereus polyrhizus, comparing it with conventional solvent extraction methods [ref. 79]. The SFE strategy achieved comparable betacyanin contents and extraction efficiencies while significantly reducing organic solvent usage, suggesting environmental benefits over traditional methods. The application of SFE using CO2 and ethanol as an entrainer to extract Uncaria tomentosa bark to obtain pentacyclic alkaloids with reduced tannin and antioxidant levels. The study emphasized SFE’s capability to selectively extract alkaloids when combined with ethanol, although it required substantial amounts of plant material for effective high‐pressure extraction [ref. 80].
Brandão et al. extracted alkaloids from the aerial parts of Melocactus zehntneri using supercritical CO2 [ref. 81]. They compared the global yield and alkaloid profile of SFE extracts with those obtained through conventional techniques, including maceration and UAE. SFE was conducted on both adult and young plant specimens in parallel experiments. The highest global yield was 0.68%, achieved at 300 bar and 35°C using adult plant material, followed by 0.52% from young material at the same temperature. For conventional methods, maceration showed the highest efficiency with a 14 ± 1% yield after 72 h, and slightly lower yields for 24 and 48 h (11 ± 2% and 12 ± 1%, respectively). UAE yielded 13 ± 1%, similar to maceration, indicating no additional benefit from ultrasonic waves for this material. Although SFE had a lower yield, it was the most selective for extracting alkaloids and also produced polysaccharide‐free extracts. This reduced the number of purification steps, making SFE a more sustainable technique due to the unique properties of scCO2. Similarly, Ruan et al. optimized SFE conditions for extracting alkaloids from Fritillaria thunbergii flowers, achieving high yields of total alkaloids, peimine (45), and peiminine (46). These studies demonstrate SFE’s efficacy and selectivity, emphasizing its potential for producing high‐quality extracts with fewer purification steps and greater environmental benefits [ref. 82].
Recent studies have continued to demonstrate the effectiveness of SFE [ref. 83]. Colchicine (47) was successfully extracted from Gloriosa superba seeds. The researchers processed 3 kg of seed powder containing 0.70% (47), using liquid CO2 with 3% water as the cosolvent. Conducted at 60°C for 2 h, with pressures ranging from 200 to 450 bar, the optimal yield of 93.6% was achieved at 400 bar, corresponding to 27% of (47). Galarce‐Bustos et al. optimized SFE to extract alkaloids from Annona cherimola, evaluating the extracts’ bioactivity profile [ref. 84]. Several aporphine alkaloids were identified, including S‐(+)‐glaucine (48), xylopine (49), and anonaine (50), and the optimal conditions are described in Table 2.
For the extraction of alkaloids from the organs of Papaver bracteatum L., Salehi et al. conducted an experimental design including a 2( n − 1) fractional factorial and central composite design (CCD) to evaluate and determine optimal conditions: 351 atm pressure, 90 mL modifier volume (9% v/w), 40°C oven temperature, and extraction times of 15 min static and 55 min dynamic. This study highlights SFE’s effectiveness in extracting specific alkaloids under optimized parameters [ref. 85].
The extraction of (5), theobromine (51), and theophylline (52) was also explored from dry yerba mate leaves [ref. 86]. The effects of cosolvents in both static and continuous loading methods were considered. Pure scCO2 demonstrated high selectivity for alkaloids but resulted in a low extraction yield of 0.39 g/kg of yerba mate. The addition of ethanol significantly increased the alkaloid content, reaching up to 5.0 g/kg with continuous loading. Using hydrated ethanol further enhanced the extraction yields, achieving 9.4 g/kg of alkaloids with continuous loading. The total amount of alkaloids was extracted from Sophora moorcroftiana, with the aim of mitigating issues associated with organic solvent residue and complex industrial production found in other extraction methods. Thus, parameters were systematically varied, including pressure, temperature, time, alkalization time, carrier ethanol concentration, and sample crushing time. Box–Behnken response surface analysis identified the optimal conditions (Table 2), resulting in an extraction yield of 68.88 µg of total alkaloids per gram of S. moorcroftiana [ref. 87].
IL Plant Alkaloid Extraction
ILs are a class of solvents known for their unique properties that exceed those classified as traditional. They possess negligible vapor pressure, are nonflammable, exhibit high thermal stability, and show low chemical reactivity, enhancing safety and environmental friendliness. Comprising organic cations paired with inorganic or organic anions, ILs remain liquid at room temperature, offering exceptional dissolving and extraction capabilities alongside a wide liquidus range. Their applications span synthesis, catalysis, electrochemistry, and analytical chemistry, demonstrating superior efficiency in extraction processes and reducing solvent usage while yielding higher outputs [ref. 88, ref. 89].
In solid–liquid extraction, ILs enhance interactions due to their ionic nature, robustly interacting with matrices to increase permeability and extraction efficiency. Their nonflammable and recyclable properties underline their sustainability as solvents, promising advancements in analytical methods and the extraction of active compounds from complex materials. The ability to tailor ILs further augments their potential for diverse applications across scientific and industrial domains [ref. 88]. Figure 6 illustrates three different methods of extraction using ILs: (A) aqueous solution extraction; (B) ATPSs; (C) UAE.
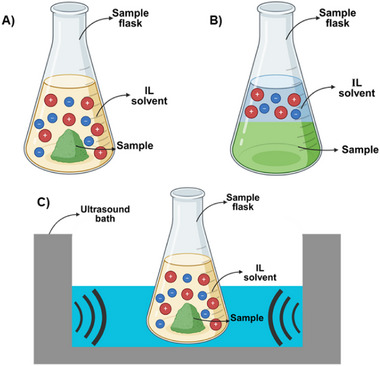
In this sense, after carefully reviewing publications, we found that 24 alkaloids were extracted, isolated, and identified using ILs as solvents (Table 3). Before extraction, comparative studies were conducted using different cations and anions to determine the most efficient solvent composition. In addition, the concentration of these ILs was evaluated, revealing that both lower and higher concentrations are less effective. Consequently, intermediate concentrations are typically favored. Furthermore, it was concluded that the structure of ILs significantly influences their physicochemical properties, thereby impacting the efficiency of alkaloid extractions.
TABLE 3: Summary of study conditions for alkaloid extraction using ionic liquids.
| Target alkaloid(s) | Plant source | Assistance technique | IL Composition | Optimized conditions | References |
|---|---|---|---|---|---|
| Berberine (53), jatrorrhizine (54), palmatine (55) | Phellodendron amurense | Ultrasound‐assisted extraction | [Bmim][Br] | 100 W ultrasonic power, 75 min, solid–liquid ratio 1:14 | Wang et al. [ref. 90] |
| S‐(+)‐glaucine (48) | Glaucium flavum | Aqueous solution extraction | [C4C1im][Ace] | 1 M IL‐aqueous solution, solid–liquid ratio 1:40, methanol comparison | Bogdanov et al. [ref. 91]; Keremedchieva et al. [ref. 92] |
| Galanthamine (39) | Leucojum aestivum | Infrared‐assisted extraction | [C4C1im]Cl | Optimized time and temperature | Svinyarov et al. [ref. 88] |
| Benzoylmesaconine (62), benzoylaconine (63), benzoylhypaconine (64), mesaconitine (65), hypaconitine (66), aconitine (67) | Aconitum carmichaeli | Aqueous two‐phase systems | [C6MIM]Br‐K2HPO4 | [CnMIM]Br (n = 4, 6, 8)‐K2HPO4, varying IL alkyl chain lengths, phase separation optimization | Wang et al. [ref. 93] |
| Lycorine (56), lycoramine (40), galanthamine (39) | Lycoris radiata | Infrared‐assisted extraction | [C4MIM]Cl | 0.5 M [C4MIM]Cl, 60% methanol, liquid‐to‐solid ratio 50:1, 10 min | Xu et al. [ref. 94] |
| Protopine (68), allocryptopine (69), sanguinarine (70), chelerythrine (71), dihydrochelerythrin, dihydrosanguinarine | Macleaya cordata | Not specified | [C6MIM][Br] | 100 µL IL, pH 4.0, 15 min UAE, 80°C | Li et al. [ref. 89] |
| Vincamine (57), vindoline (58), catharanthine (59) | Vinca minor, Catharanthus roseus | Not specified | [BMIm]Cl | Ultrasonic bath 50°C for 30 min | Koel et al. [ref. 95] |
| Berberine (53), palmatine (55), jatrorrhizine (54), magnoflorine (60), phellodendrine (61) | P. amurense | Ultrasound‐assisted extraction | [C4mim][OAc]/DMSO | On‐aqueous solvent system with DMSO, optimized viscosity, enhanced extraction yield | Peng et al. [ref. 96] |
In 2015, the use of ILs for alkaloid extraction saw notable advancements after three key studies. Wang et al. developed an extraction method for berberine (53), jatrorrhizine (54), and palmatine (55) from Phellodendron amurense Rupr. [ref. 90]. They systematically optimized extraction parameters, including varying IL compositions (different cations and anions), IL concentration, solid–liquid ratio, ultrasound power, and extraction time. Optimal conditions using [Bmim][Br] with 100 W ultrasonic power, 75 min extraction time, and a 1:14 solvent to raw material ratio yielded a high extraction efficiency of 106.7% (Table 3). Simultaneously, Bogdanov et al. and Keremedchieva et al. explored IL‐based extraction systems form (48) from Glaucium flavum Cr. [ref. 91, ref. 92]. Bogdanov et al. compared the efficiency of 1 M [C4C1im][Ace]‐aqueous solution versus methanol, highlighting the superior performance of the IL‐based system with an optimal solid–liquid ratio of 1:40 (wt/v) [ref. 91]. Keremedchieva et al. investigated the back‐extraction capabilities of various organic solvents and found that chloroform achieved quantitative recovery of (48) in a single step, resulting in product purities exceeding 95%. These first studies were important milestones because they demonstrated the effectiveness of ILs in improving both the efficiency and purity of plant‐derived alkaloids extraction [ref. 92].
Shortly thereafter, Svinyarov et al. explored aqueous solutions of various hydrophilic imidazolium‐, pyrrolidinium‐, and ammonium‐based ILs as extractants to efficiently obtain (39) from the aerial parts of Leucojum aestivum L. [ref. 88]. They explored several cations and anions, optimizing conditions like extraction time, temperature, and solvent concentration. The study established optimal parameters for quantitative extraction of (39), leading to a method using [C4C1im]Cl for preliminary extraction followed by high‐performance liquid chromatography (HPLC) quantification in plant material. In a study focused on extracting aconitum alkaloids from Aconitum carmichaeli Debx. using IL‐based ATPSs (IL‐ATPS), various parameters were optimized and compared with conventional techniques. Several ILs and inorganic salts were combined, and the findings indicated that [CnMIM]Br (n = 4, 6, 8)‐K2HPO4 systems formed clear and stable two‐phase systems, an ideal condition for extracting those alkaloids due to effective phase separation and suitable pH conditions [ref. 93]. Notably, extraction yields increased with the alkyl chain length of the ILs from butyl to hexyl.
The application of ILs in infrared‐assisted extraction (IRAE) of alkaloids from Lycoris radiata was conducted [ref. 94]. Parameters of the system IL‐IRAE were systematically optimized using ILs with different cations and anions, focusing on achieving high extraction yields of (39), (40), and lycorine (56). Best conditions included 0.5 M [C4MIM]Cl combined with 60% methanol, a liquid‐to‐solid ratio of 50:1 (mL/g), an extraction time of 10 min, and a 275‐W lamp. Similarly, Li et al. (2017) developed a green extraction method for alkaloids from Macleaya cordata, particularly focusing on 1‐hexyl‐3‐methylimidazolium ILs with various anions (Cl−, Br−, BF4−, PF6−). Most water‐miscible ILs were efficient for the procedure, with [C6MIM][Br] showing optimal results. The study showed that extraction was influenced by the anion type and the length of the alkyl chain in the IL cation. The extraction procedure was improved by dissolving varying amounts of [C6MIM][Br] in water, determining that 100 µL provided the highest recovery of target alkaloids [ref. 89].
Koel et al. evaluated five imidazolium‐based ILs and nine DESs for extracting indole alkaloids from the leaves of Vinca minor and C. roseus. They focused on extracting three specific alkaloids: vincamine (57) from V. minor, and vindoline (58) and catharanthine (59) from C. roseus. The efficiency of ILs and DESs varied depending on the plant and compound, but both showed potential for comparable or better yields than methanol. The IL [BMIm]Cl produced extraction yields similar to methanol, especially for C. roseus. Extraction yields were consistent, with standard deviations between 4% and 7%. Yields for (59) ranged from 60.2 to 329.9 µg/g, for (58) from 114.8 to 659.7 µg/g in C. roseus, and for (57) from 6.0 to 173.9 µg/g in V. minor. The study demonstrated the potential of ILs and DESs as effective solvents for alkaloid extraction, with specific selectivity for different compounds [ref. 95].
In a recent study, Peng et al. explored using ILs for dissolving lignocellulose, selecting a 1‐butyl‐3‐methylimidazolium acetate/dimethyl sulfoxide ([C4mim][OAc]/DMSO) nonaqueous solvent system as a green solvent for alkaloid extraction. This system effectively extracted (53), (54), (55), magnoflorine (60), and phellodendrine (61) from 0.1 g of P. amurense. The addition of DMSO reduced the viscosity of the IL and aided in dissolving cell walls. Compared to traditional methods like heating reflux extraction and maceration extraction, incorporating UAE technology increased the yield and quality of alkaloid active substances while reducing extraction time [ref. 96].
ILs have shown significant potential since their introduction for extracting alkaloids from plant materials, achieving high efficiencies while prioritizing environmental sustainability. Their versatility, customizable through variations in both cationic and anionic components, enhances their utility and allows for tailored optimization in specific applications. Table 3 provides a comprehensive summary of ILs used in referenced studies, detailing extracted alkaloids and their respective plant sources. Moreover, the literature suggests a shift from ILs to DESs in extraction methods, highlighting the considerable promise of both solvent systems. Thus, in the next topic, we will explore advancements in plant‐derived alkaloids using DESs.
DES Plant Alkaloid Extraction: DES and NADES
DESs and NADESs have emerged as green alternatives to conventional organic solvents for extraction processes. DESs are a class of liquids formed by mixing two or more solid components, typically prepared under agitation and maintained at 40°C–90°C. These components include a hydrogen bond acceptor (HBA) and a hydrogen bond donor (HBD), which, when combined, exhibit unique physicochemical properties such as higher biodegradability, thermal stability, and lower toxicity [ref. 97, ref. 98].
Common HBAs used in the preparation of DESs include choline chloride and l‐proline. These are combined in different molar ratios with HBDs, such as carboxylic acids, polyols, and alcohols. The choice of HBA and HBD components, as well as their molar ratios, can lead to more selective and specific extraction processes, as illustrated by Takla et al. [ref. 99]. Their study demonstrates how varying the combination of HBA and HBD (Figure 7) can effectively extract groups of alkaloids or specific alkaloids, highlighting the flexibility and efficiency of using DESs for extractions [ref. 100, ref. 101].
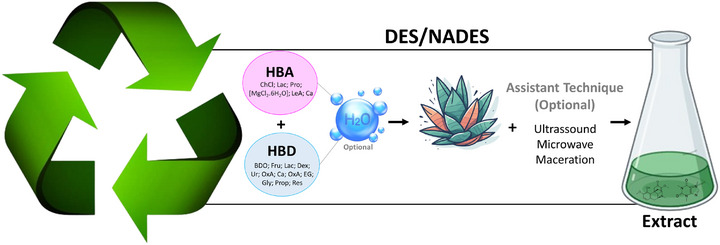
In addition, NADES are a subclass of DESs, consisting mainly of natural compounds such as sugars and amino acids. This subclassification is inspired by empirical observations that plants can use eutectic mixtures to survive under extreme conditions. Furthermore, the use of DES/NADES is often assisted by complementary techniques such as maceration, MAE and UAE to optimize the transfer of target compounds from the matrix to the solvent and to reduce the extraction time [ref. 102]. Scientific studies underscore the benefits of using DES and NADES over conventional solvents. These advantages include ease of preparation, targeted extraction capability through specific combinations of HBAs and HBDs, the ability to simultaneously extract both polar and nonpolar compounds, low cost of raw materials, and improved sustainability of the extraction methods [ref. 103].
In contrast to the benefits of using DES/NADES in alkaloid extractions, several challenges deserve attention. Chief among them is the high viscosity of these mixtures, which complicates their analysis using HPLC and limits their suitability for gas chromatography due to their low volatility. Moreover, the high viscosity can impede the efficiency of the extraction process. Other critical factors include temperature, solid/liquid ratio, and extraction time. Of particular concern is the water content in these mixtures, which, while potentially reducing viscosity, can also excessively increase polarity and interfere with hydrogen bonding interactions between HBAs and HBDs, thereby adversely affecting extraction efficiency.
The landmark study performed by Wang et al. introduced the use of DES for alkaloid extraction from Camellia sinensis [ref. 103]. They explored combinations of choline chloride with other components such as 1,4‐butanediol, ethylene glycol, various butanediols, hexanediol, and acids like malonic, lactic, and malic, as well as acetamide, methylurea, and urea. The primary alkaloids extracted were (5) and (51), with the combination yielding the highest amounts of these compounds identified as choline chloride: 1,4‐butanediol (1:3) with 50% water (Table 4). Moreover, the study highlighted that adding water in appropriate proportions to eutectic mixtures significantly mitigated the challenge of high viscosity, thereby improving extraction performance. Optimal yields for (5) and (51) were achieved when DESs were blended with approximately 40−60 wt% of water. In addition, the study compared various DES‐assisted extraction techniques, including mechanical extraction, ultrasound, and microwave methods, with mechanical extraction proving particularly efficient in extracting the target compounds [ref. 104].
TABLE 4: Summary of study conditions for alkaloid extraction using supercritical fluid extraction.
| Target alkaloid(s) | Plant Source | Part | Extraction for | Solvent composition | Assistance technique | References |
|---|---|---|---|---|---|---|
| Caffeine (5) (22.64 mg/g) and theobromine (51) (0.3573 mg/g) | Camellia sinensis | Leaves | DES | ChCl:BDO (1:3) with 50% H2O | Mechanochemical extraction, ultrasound, microwave | Wang et al. [ref. 103] |
| Lycorine (56) (1.26 mg/mL), crinine (74) (0.68 mg/mL) crinamine (76) (0.69 mg/mL) | Crinum bulbispermum | Bulbs | NADES | ChCl:Fru (5:2) with 20.57% H2O | Ultrasound | Shawky et al. [ref. 105] |
| Lycorine (56) (0.845 mg/mL), crinine (74) (0.563 mg/mL), and crinamine (76) (0.483 mg/mL) | Crinum powellii | Bulbs | NADES | ChCl:Fru (5:2) with 35% H2O | Ultrasound | Takla et al. [ref. 99] |
| Sinomenine (77) (2 g/100 g); magnoflorine (60) (1.5 g/100 g); berberine (53) (8 g/100 g); rutaecarpine (78) (0.06 g/100 g); evodiamine (79) (0.01 g/100 g); tetrandrine (80) (0.7 g/100 g); fangchinoline (81) (1.2 g/100 g); matrine (82) (0.01 g/100 g); ammothamnine (83) (2.6 g/100 g); epiberberine (84) (1.5 g/100 g); palmatine (55) (2 g/100 g); coptisine (85) (2.8 g/100 g) | Caulis sinomenii, Coptis chinensis, Stephania tetrandra, Tetradium ruticarpum, and Sophora flavescens | Plant material | DES | ChCl:Lac (1:2) with 30% H2O | Ultrasound | Jiang et al. [ref. 101] |
| Harmaline (19) (nd), piperine (86) for Larrea cuneifolia (33.13 ± 0.39 µg/mL), (86) for L. divaricata (44.84 ± 0.46 µg/mL); caffeine (5) (nd) | L. cuneifolia, Larrea divaricata, Thymus vulgaris, and Origanum vulgare | Plant material | NADES | Lac:Dex (5:1) with 15% H2O | Ultrasound | Espino et al. [ref. 106] |
| Caffeine (5) (CP 0.55 g/100 g; CH, reaching 0.10/100 g), theobromine (51) CH 0.55 g/100 g) | Coffee, cocoa, pod husk | Coffee pulp (CP), cocoa husk (CH), and pod husk (CPH) | DES | ChCl:Lac:H2O (1:2:1.5) | Heat stirring‐assisted extraction (HSE) or ultrasound‐probe assisted extraction (UPAE) | Ruesgas‐Ramon et al. [ref. 107] |
| Catharanthine (59) (60.2–329.9 µg/g), vincamine (57) (114.8–659.7 µg/g), vindoline (58) (6.0 and 173.9 µg/g) | Catharanthus roseus and Vinca minor | Leaves | DES | ChCl:Ur (1:1 and 1:2) with 25% H2O | Ultrasound | Koel et al. [ref. 95] |
| Caffeine (5) (1.599 mg/g) and theobromine (51) (5.004 mg/g) | Cocoa bean shell (CBS) | Husk | DES | ChCl:BDO (1:2) with 30% H2O | Ultrasound | Pavlović et al. [ref. 108] |
| Boldine (87) (2.36 mg/g) | Peumus boldus | Leaves | NADES | Pro:OxA (1:1) with 20% H2O | Ultrasound | Torres‐Vega et al. [ref. 110] |
| Berberine (53) (10.82 mg/g) and palmatine (55) (4.74 mg/g) | Phellodendri amurensis | Plant material | DES | ChCl:Ca (1:2) with 30% H2O | Ultrasound | Li et al. [ref. 109] |
| Arecoline (88) (34.07–35.86 mg/g), arecaine (89) (2.11–2.25 mg/g), guvacoline (90) (11.12–15.89 µg/g), and guvacine (91) (0.53–0.85 µg/g) | Areca catechu L. | Seeds | DES | Pro:EG (1:2) and Pro:Lac (1:2) | Ultrasound | Wang et al. [ref. 111] |
| Betalains (betacyanin and betaxanthins) (3.65–3.99 mg/g) | Beta vulgaris | Plant material | DES | [MgCl2·6H2O]:Ur (2:1) and (1:1) | Ultrasound | Hernández‐Aguirre et al. [ref. 112] |
| Protopine (68) (0.49 mg/g), chelidonine (44) (2.80 mg/g), berberine (53) (0.33 mg/g), chelerythrine (71) (2.31 mg/g), coptisine (85) (1.07 mg/g), and sanguinarine (70) (1.71 mg/g) | Chelidonium majus | Roots and shoots | NADES | MT, MC, and TC in different molar ratio | Ultrasound | Strzemski et al. [ref. 116] |
| O‐Nornuciferine (92) (0.069 g/100 g), N‐nornuciferine (93) (0.152 g/100 g), nuciferine (94) (0.334 g/100 g), and roemerine (95) (0.041 g/100 g) | Nelumbo nucifera | Leaves | DES | ChCl:Prop (1:4) with 30% H2O | Ultrasound | Liu et al. [ref. 113] |
| Skimmianine (96) (76.6 µg/g), dictamnine (97) (97.85 µg/g), and evodiamine (79) (531.67 µg/g) | Evodia lepta | Roots | DES | LeA:Gly (1.5:1) containing 30% K2HPO4 | Microwave with the exception of extractions with methanol mixtures, which were carried out at room temperature | Yu et al. [ref. 115] |
| Fangchinoline (81) (7.23 mg/g) and tetrandrine (80) (13.36 mg/g) | Stephania tetrandra | Roots | DES | ChCl:EG (1:2) with 20% H2O | Ultrasound | He et al. [ref. 54] |
| No specification, alkaloid total apomorphine derivatives (392.3 ± 8.0 µg ACE/mL) (ACE: apomorphine hydrochloride equivalent) | Nicotiana tabacum | Inflorescences | NADES | ChCl:Ur:H2O (1:2:1.5) | Maceration | Leal et al. [ref. 116] |
| Atisinium (99) (Method 1: 85.73 ± 4.48 mg/g), lappaconitine (100) (Method 1: 7.99 ± 0.11 mg/g), 2‐O‐cinnamoyl hetisine (101) (Method 2: 11.67 ± 0.15 mg/g) | Aconitum heterophyllum | Roots | NADES | Method 1—Lac:Gly (1:1) and Method 2—ChCl:Res (1:1) | Ultrasound | Sharma et al. [ref. 118] |
| No specification, alkaloid total apomorphine derivatives (1123.0 ± 7.0 µg ACE/mL) (ACE: apomorphine hydrochloride equivalent) | N. tabacum | Leaves | NADES | ChCl:Ur:H2O (1:2:1.5) | Maceration | Leal et al. [ref. 117] |
| Caffeine (5) (21.61 ± 0.95 mg/g) and theobromine (51) (2.02 ± 0.10 mg/g) | Ilex paraguariensis | Plant material | NADES | Ca:Gly:H2O (1:3:9) | Ultrasound | Cunha et al. [ref. 104] |
Note: Choline chloride (ChCl); 1,4‐butanediol (BDO); fructose (Fru); lactic acid (Lac); dextrose (Dex); water (H2O); citric acid (Ca); l‐proline (Pro); ethylene glycol (EG); magnesium chloride hexahydrate ([MgCl2·6H2O]); urea (Ur); glycerol (Gly); menthol–thymol (MT); menthol–camphor (MC); thymol–camphor (TC); propanediol (Prop); levulinic acid (LeA); resorcinol (Res); oxalic acid (OxA). Other combinations were tested, but only the most important of them are in this study.
In 2018, a noticeable trend toward the increased use of NADES over DES emerged. Two studies published that year focused on these solvents: one examining both types and another specifically exploring NADES. Both studies investigated the extraction of alkaloids such as (56), crinine (74), and crinamidine (75) from the plant species Crinum bulbispermum and C. powellii [ref. 99, ref. 105].
Shawky et al. used DES/NADES consisting of choline chloride and fructose in a 5:2 molar ratio, with varying water concentrations (7%–20.57% v/v), and ultrasound assistance to extract alkaloids from C. bulbispermum bulbs. They obtained concentrations of (56) (1.26 mg/mL), (74) (0.68 mg/mL), and crinamine (76) (0.69 mg/mL) [ref. 105]. Similarly, Takla et al. employed NADES with the same choline chloride and fructose molar ratio, but with 35% water and UAE to obtain alkaloids from C. powellii bulbs. They achieved concentrations of (56) (0.845 mg/mL), (74) (0.563 mg/mL), and (76) (0.483 mg/mL) [ref. 99]. These studies demonstrate the effectiveness of DES and NADES in extracting alkaloids, highlighting the role of water content and ultrasound in enhancing extraction efficiency. The consistent use of the choline chloride‐fructose system across both studies underscores its applicability and effectiveness in alkaloid extraction from different plant species.
In 2019, Jiang et al. employed DESs to extract alkaloids from several medicinal plants, including Caulis sinomenii, Coptis chinensis, Stephania tetrandra, Tetradium ruticarpum, and S. flavescens. They tested 75 different mixtures with varying molar ratios. Focusing on C. sinomenii, the study utilized a mixture of choline chloride and lactic acid (1:2), supplemented with 30% water and assisted by ultrasound. The extraction yielded the following alkaloids: Sinomenine (77) (2.0 g/100 g), (60) (1.5 g/100 g), (53) (8.0 g/100 g), rutaecarpine (78) (0.06 g/100 g), evodiamine (79) (0.01 g/100 g), tetrandrine (80) (0.7 g/100 g), fangchinoline (81) (1.2 g/100 g), matrine (82) (0.01 g/100 g), ammothamnine (83) (2.6 g/100 g), epiberberine (84) (1.5 g/100 g), (55) (2.0 g/100 g), and coptisine (85) (2.8 g/100 g) [ref. 101].
The extraction study of Larrea cuneifolia and L. divaricata focused on obtaining alkaloids like (5), (19), and piperine (86) using NADES [ref. 106]. The NADES composition used was lactic acid and dextrose (5:1), with the addition of 15% water (v/v), and ultrasound was employed to enhance extraction efficiency. Concentrations obtained for (86) were 33.13 ± 0.39 µg/mL in L. cuneifolia, and 44.84 ± 0.46 µg/mL in L. divaricata.
Ruesgas‐Ramon et al. applied the DES approach to extract alkaloids from coffee pulp (CP), cocoa husk (CH), and pod husk (CPH). Their DES formulations, featuring choline chloride, lactic acid, and water (1:2:1.5), were tested alongside variants containing betaine, glycerol, and 1,4‐butanediol. Comparative conventional ethanol‐based methods demonstrated slightly superior extraction efficiencies overall, with modest differences in yields across methods. The most effective combination (choline chloride acid) yielded 0.55 g/100 g of (5)from CP with ultrasound assistance, 0.10 g/100 g of (5) from CH with heat stirring‐assisted extraction, and 0.55 g/100 g of (51) from CH with ultrasound [ref. 107].
Pavlović et al. conducted a study on alkaloid extraction from cocoa bean shell (CBS) using a DES system composed of choline chloride and butan‐1,4‐diol (1:2) with 30% of water. The extraction process involved microwave heating, and 16 different mixtures with various molar ratios were tested. The main alkaloids extracted were (5) and (51), yielding 1.599 and 5.004 mg/g, respectively [ref. 108]. Various combinations of DESs were tested to extract alkaloids from C. roseus and V. minor leaves. These DESs included mixtures of choline chloride with urea in ratios (1:1 and 1:2), supplemented with 25% of water. In addition, other DES combinations evaluated were choline chloride:acetamide, choline chloride:urea, choline acetate:urea, choline chloride:glycerol, choline chloride:maltose, menthol:formic acid, menthol:acetic acid and menthol:lactic acid. Ultrasonic extraction was utilized to enhance efficiency. Results showed (59) concentrations ranging from 60.2 to 329.9 µg/g and (58) concentrations from 114.8 to 659.7 µg/g for C. roseus. (57) was extracted from V. minor with yields ranging from 6.0 to 173.9 µg/g [ref. 95].
Furthermore, the main alkaloids extracted from Phellodendri amurensis were (53) (10.82 mg/g) and (55) (4.74 mg/g) [ref. 109]. The study utilized a DES composed of choline chloride and citric acid (1:2 molar ratio) with 30% water for extraction, employing ultrasound as the processing method. Other combinations of choline chloride with citric acid, malic acid, levulinic acid, succinic acid, lactic acid, ethylene glycol, glycerol, sucrose, and xylitol in different molar ratios were also investigated. Torres‐Vega et al. used a NADES composed of l‐proline and oxalic acid (1:1) with 20% of water. In addition, they tested other combinations of choline chloride and l‐proline with 1,2‐propanediol, glycerol, lactic acid, levulinic acid, and citric acid in various molar ratios. The process was enhanced by UAE and MAE for the extraction of boldine (87) from the leaves of Peumus boldus, with a yield of 2.362 ± 0.055 mg/g [110].
Arecoline (88) (34.07–35.86 mg/g), arecaine (89) (2.11–2.25 mg/g), guvacoline (90) (11.12–15.89 µg/g), and guvacine (91) (0.53–0.85 µg/g) were the main alkaloids extracted from Areca catechu L. seeds [ref. 111]. The study utilized UAE and a DES composed of l‐proline and ethylene glycol (1:2) and l‐proline and lactic acid (1:2). However, other DES combinations were tested, varying the molar ratios (Table 4). All prepared DESs were diluted with 30% water (v/v) to optimize extraction.
Another study focused on the extraction of betalains, including betacyanins and betaxanthins, from Beta vulgaris. The extraction method employed was UAE using magnesium chloride hexahydrate [MgCl2·6H2O] (HBA) and urea (HBD), in two molar ratios: (2:1) and (1:1). This study aimed to investigate the efficiency of these DESs in extracting natural pigments. The results showed that betalains were extracted with yields ranging from 3.65 to 3.99 mg/g, depending on the molar ratio used [ref. 112].
Another study using DES in 2022 was developed by Liu et al. [ref. 113], who focused on the extraction of O‐nornuciferine (92), N‐nornuciferine (93), nuciferine (94), and roemerine (95) from lotus leaves (Nelumbo nucifera). Seventy different mixtures with varying molar ratios were tested using ultrasound assistance. The results showed extraction yields of 0.069 g/100 g for (92), 0.152 g/100 g for (93), 0.334 g/100 g for (94), and 0.041 g/100 g for (95) under the condition of choline chloride–propanediol (1:4) containing 30% water, and a solid–liquid ratio of 1:100 g/mL.
The extraction of (44), (53), (85), (68), (71), and (70) from C. majus was performed using UAE. In a study by Strzemski et al., solvents such as menthol–thymol (MT), menthol–camphor (MC), and thymol–camphor (TC) were used in various molar ratios. These ratios included MT (5:5 and 6:4), MC (6:4 and 7:3), and TC (4:6 and 5:5), with acidified water and methanol serving as control extractants. The extraction results showed yields of (68) (0.49 mg/g), (44) (2.80 mg/g), (53) (0.33 mg/g), (71) (2.31 mg/g), (85) (1.07 mg/g), and (70) (1.71 mg/g) under these experimental conditions [ref. 114].
In 2023, several studies focused on the obtention of bioactive alkaloids. Yu et al. investigated the extraction of (79), skimmianine (96), and dictamnine (97) from Evodia lepta roots using levulinic acid and glycerol (1.5:1) with 30% of K2HPO4 solution, achieving yields of 76.60 µg/g for (96), 97.85 µg/g for (97), and 531.67 µg/g for (79) under MAE conditions [ref. 115]. Concurrently, He et al. explored DES for extracting (81) and (80) from S. tetrandra roots, utilizing choline chloride‐glycol (1:2) with 20% water, and achieved yields of 7.23 mg/g for (81) and 13.36 mg/g for (80) using UAE [ref. 54]. In addition, Leal et al. [ref. 116, ref. 117] investigated NADES for extracting alkaloids from Nicotiana tabacum, obtaining 392.3 ± 8.0 µg ACE/mL of apomorphine hydrochloride (98) from inflorescences [ref. 117] and 1123.0 ± 7.0 µg ACE/mL from leaves [ref. 117] using choline chloride:urea:water (1:2:1.5). Sharma et al. also utilized NADES for extracting atisinium (99), lappaconitine (100), and 2‐O‐cinnamoyl hetisine (101) from Aconitum heterophyllum roots, achieving yields of 85.73 ± 4.48, 7.99 ± 0.11, and 11.67 ± 0.15 mg/g, respectively, under various DES compositions [ref. 118]. In addition, Fernández et al. investigated NADES for extracting (5) and (51) from Ilex paraguariensis using CGH (citric acid:glycerol:water), LGH (lactic acid:glucose:water), and ChGH (choline chloride:glycerol:water), yielding 21.61 ± 0.95 mg/g of (5) and 2.02 ± 0.10 mg/g of (51) under UAE conditions [ref. 119].
DESs have evolved significantly for alkaloid extraction from various plants. Techniques like ultrasound, mechanical extraction, and microwave assistance have further improved yields, with mechanical methods standing out for their robustness in extracting alkaloids. These developments underscore the ongoing innovation in solvent design and extraction techniques, paving the way for enhanced bioactive compound recovery and broader applications in pharmaceutical and agricultural industries.
Most Commonly Conditions Used in DES/NADES Systems
In the selected studies, various combinations of HBA and HBD were used with variations in molar ratios. In addition, in most cases, water was added to the system to reduce viscosity. In this context, as illustrated in Figure 8, it can be observed that in terms of HBA, the most used compounds are choline chloride, present in 56% of the studies, followed by l‐proline in 12%, and lactic acid and menthol, both with 8%. Other compounds include magnesium chloride hexahydrate, thymol, levulinic acid, and citric acid, each representing 6% of the studies.
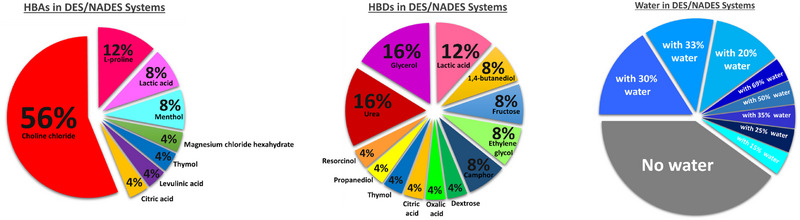
Among the most frequently used compounds, such as HBD, urea and glycerol stand out, each appearing in 16% of the studies, followed by lactic acid in 12%. Compounds such as 1,4‐butanediol, fructose, ethylene glycol, and camphor were used in 8% of the studies. Other compounds, including dextrose, oxalic acid, citric acid, thymol, propanediol, and resorcinol, were utilized in 4% of the studies.
Regarding the addition of water to the DES/NADES systems, 40% of the studies did not incorporate water into the eutectic solvent system. In the remaining studies, the proportion of added water varied between 15% and 69%. In 16% of the studies, 30% water was added; 12% of the studies used 20% water, and another 12% employed 33%. The remaining proportions include 69% of water (4% of the studies), 50% of water (4%), 35% of water (4%), 25% of water (4%), and 15% of water (4%).
Thus, it becomes evident that choline chloride is the most widely used component as an HBA, standing out in a significant number of studies. Similarly, urea and glycerol emerge as the most utilized HBDs, indicating their relevance in the processes analyzed. Furthermore, it can be observed that in the majority of studies using these compounds, there is a common practice of adding water to the system, with variations in the proportions employed. However, it is important to note that a considerable portion of the authors (40% of the studies) chose not to include water in the DES/NADES systems, preferring to maintain the system in its original eutectic form without dilution.
The inclusion of water in eutectic systems is influenced by various factors, including the desired properties, solubility, and stability of the target compounds. Adding water is often beneficial, particularly for extracting NPs, as it significantly improves extraction efficiency for polar substances. This is evident in the data from this survey, where 60% of studies utilized water in their systems. Water’s ability to lower the viscosity of DES/NADES mixtures enhances their manageability and increases the solubility of hydrophilic compounds, aiding in the extraction process. However, water’s presence is not always advantageous. It can negatively impact compounds prone to hydrolysis and, in some organic synthesis or separation processes, might affect the selectivity or efficiency of the procedure. Therefore, the decision to add water must be carefully considered, balancing the benefits of enhanced extraction efficiency and reduced viscosity against the potential downsides related to compound stability and process selectivity [ref. 120, ref. 121, ref. 122].
For a more comprehensive view, Table 5 correlates the advantages, disadvantages, main components of each solvent, and the most common uses of each complementary extraction technique.
TABLE 5: Comparison of advantages, disadvantages, composition of mixtures, and main uses of nonconventional and conventional solvents.
| MAE/UAE | IL | SFE | DES | NADES | Conventional solvents | |
|---|---|---|---|---|---|---|
| Advantages | Improve extraction efficiency, speed, and cost‐effectiveness while providing sustainable, high‐purity extracts with reduced solvent and energy consumption | Negligible vapor pressure, nonflammability, high thermal stability, low chemical reactivity, excellent solubility, extraction efficiency, a broad liquidus range, enhances interactions with solid matrices, increasing permeability, and reducing solvent consumption | Uses supercritical CO₂ for selective, efficient extraction while reducing organic solvent use | Ease of preparation, targeted extraction with HBA and HBD, capability to extract polar and nonpolar compounds simultaneously, low cost of raw materials, sustainability | Greater biocompatibility, environmental sustainability, low toxicity, renewable sources | Ease of preparation, targeted extraction with HBA and HBD, capability to extract polar and nonpolar compounds simultaneously, low cost of raw materials, sustainability |
| Disadvantages | Requires precise optimization and longer extraction times to prevent degradation | Concentration and IL structure influence | Requires precise optimization of parameters to achieve good yields and maintain compound stability | Variable biocompatibility potential depending on chosen components, high viscosity | Greater difficulty in preparing a stable eutectic mixture from natural compounds, high viscosity | High environmental toxicity |
| Main components of mixtures | Not applicable | Organic cations paired with inorganic or organic anions | CO2 as solvent | HBA and HBD, often synthetic compounds | Sugars, amino acids, organic acids | Derivatives of hydrocarbons and other synthetic compounds |
| Main uses | Targeted heating of specific metabolites in plant samples for more efficient extraction | Widely used in synthesis, catalysis, electrochemistry, and analytical chemistry | Ideal for pharmaceutical and research applications | Extraction of bioactive compounds, catalysis, materials synthesis | Extraction of natural products, biocatalysis, food preservation | Chemical and pharmaceutical industries, large‐scale extraction operations |
Conclusion and Perspectives
Modern techniques, such as SFE, MAE, and UAE, have revolutionized the field of NP chemistry, especially in extracting bioactive compounds like plant‐derived alkaloids. These strategies offer substantial advantages over traditional solvent extraction methods by significantly enhancing extraction efficiency, reducing processing times, and improving the purity of extracted compounds. Despite these advancements, further research is needed to standardize and optimize extraction conditions to maximize yield and ensure reproducibility across different plant species and alkaloid classes.
Looking forward, integrating these advanced extraction techniques with sustainable solvents, such as ILs, DESs, and NADESs, aligns with the United Nations’ Sustainable Development Goals (SDGs). These green solvents, derived from renewable resources, support SDG 12 (Responsible Consumption and Production) by promoting sustainable chemical use and reducing waste. They also contribute to SDG 13 (Climate Action) by lowering the carbon footprint associated with traditional organic solvents.
Future research should focus on developing novel DES and NADES formulations tailored for specific alkaloid classes and plant matrices, advancing SDG 9 (Industry, Innovation, and Infrastructure) through sustainable technology and process optimization. This approach will enhance solvation properties, improve extraction yield and purity, and minimize environmental impact.
Furthermore, integrating computational modeling and process intensification techniques can streamline extraction methods, further supporting SDG 9 and SDG 8 (Decent Work and Economic Growth) by boosting industrial efficiency and economic viability. Computational tools can predict solvent–solute interactions and guide the design of optimized extraction conditions, while process intensification improves throughput and scalability. These advancements make green extraction technologies practical for industrial‐scale applications in pharmaceuticals, cosmetics, and functional foods.
In conclusion, modern extraction techniques, coupled with sustainable solvents and advanced computational methods, represent the future of alkaloid extraction, offering efficient, eco‐friendly, and economically viable solutions for various industry segments. In addition, the integration of artificial intelligence (AI) and machine learning (ML) will enhance the optimization and scaling of alkaloid extraction, allowing for more efficient and targeted processes. These technologies will work alongside sustainable extraction methods, creating adaptive protocols that maximize yield and reduce waste. Furthermore, the adoption of circular economy principles and biorefinery concepts will enable the repurposing of waste materials from extraction, reducing environmental impact and promoting resource reuse. Together, these advancements will foster a more sustainable, efficient, and economically viable future for NP exploration, aligning with global sustainability goals.
Author Contributions
Thiago André Moura Veiga: writing – original draft preparation and editing, writing – review and editing, supervision, funding acquisition. Victor Menezes Sipoloni: writing – original draft preparation and editing, writing – original draft preparation, writing – review and editing. Milena Costa Bassicheto: writing – original draft preparation. Maria Vitória de Oliveira: writing – original draft preparation. Ana Beatriz dos Santos de Souza: writing – original draft preparation. Gabriella Melo Alves: writing – original draft preparation.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Plant Metabolomics: An Overview of the Role of Primary and Secondary Metabolites Against Different Environmental Stress Factors,”. Life, 2023. [DOI | PubMed]
- Production of Plant Secondary Metabolites in Plant Cell and Tissue Culture: The Example of Tabernaemontana and Aspidosperma Genera,”. Revista Brasileira de Farmacognosia, 2008. [DOI]
- Plant Alkaloids: Main Features, Toxicity, and Mechanisms of Action,”. Plant Toxins, 2015. [DOI]
- Dictionary of Natural Products,” accessed September 14, 2024,
- 5 T. Aniszewski , Alkaloids: Chemistry, Biology, Ecology, and Applications (Elsevier, 2015).
- 6 T. Aniszewski , Alkaloids‐Secrets of Life: Alkaloid Chemistry, Biological Significance, Applications and Ecological Role (Elsevier, 2007).
- 7 P. M. Dewick , Medicinal Natural Products: A Biosynthetic Approach, 3rd ed. (John Wiley & Sons, 2009).
- 8 J. Buckingham , K. H. Baggaley , A. D. Roberts , and L. F. Szabó , eds., Dictionary of Alkaloids, 2nd ed. (CRC Press, 2010).
- Biochemistry, Distribution and Taxonomic Relevance of Higher Plant Alkaloids,”. Phytochemistry, 1988. [DOI]
- Chromone and Flavonoid Alkaloids: Occurrence and Bioactivity,”. Molecules, 2011. [DOI | PubMed]
- Was Pereirina the First Alkaloid Isolated in Brazil?”. Revista Brasileira de Farmacognosia, 2009. [DOI]
- 12 E. Ravina , The Evolution of Drug Discovery: From Traditional Medicines to Modern Drugs (John Wiley & Sons, 2011).
- Strychnine as Target, Samarium Diiodide as Tool: A Personal Story,”. Chemical Record, 2015. [DOI | PubMed]
- Chemistry and Synthesis of Major Opium Alkaloids: A Comprehensive Review,”. Journal of the Iranian Chemical Society, 2021. [DOI]
- Os Produtos Naturais e a Química Medicinal Moderna,”. Química Nova, 2006. [DOI]
- Produtos Naturais Bioativos de Origem Vegetal e o Desenvolvimento de Fármacos,”. Química Nova, 1990. [DOI]
- Morphine and Codeine in Oral Fluid After Controlled Poppy Seed Administration,”. Drug Testing and Analysis, 2014. [DOI | PubMed]
- The Morphine Group. Part I. A Discussion of the Constitutional Problem,”. Journal of the Chemical Society, 1923. [DOI]
- Characterization of a New Potyvirus Causing Mosaic and Flower Variegation in Catharanthus roseus in Brazil,”. Scientia Agricola, 2011. [DOI]
- Anticancer Potential of Alkaloids: A Key Emphasis to Colchicine, Vinblastine, Vincristine, Vindesine, Vinorelbine and Vincamine,”. Cancer Cell International, 2022. [DOI | PubMed]
- 21 R. H. F. Manske , ed., The Alkaloids: Chemistry and Physiology (Academic Press, 1955).
- Camptothecin Delivery Methods,”. Pharmaceutical Research, 2002. [DOI | PubMed]
- A Simple Square‐Wave Voltammetric Method for the Determination of Scopolamine in Pharmaceuticals Using a Boron‐doped Diamond Electrode,”. Química Nova, 2014. [DOI]
- Infarto do Miocárdio em Atleta Jovem Associado ao Uso de Suplemento Dietético Rico em Efedrina,”. Arquivos Brasileiros De Cardiologia, 2006. [DOI]
- Natural Products as a Vital Source for the Discovery of Cancer Chemotherapeutic and Chemopreventive Agents,”. Medical Principles and Practice, 2016. [DOI | PubMed]
- A Critical Analysis of Extraction Techniques Used for Botanicals: Trends, Priorities, Industrial Uses and Optimization Strategies,”. TrAC Trends in Analytical Chemistry (, 2018
- New Horizons in the Extraction of Bioactive Compounds Using Deep Eutectic Solvents: A Review,”. Analytica Chimica Acta, 2017. [DOI | PubMed]
- Quinina: 470 Anos de História, Controvérsias e Desenvolvimento,”. Química Nova, 2009. [DOI]
- 29 P. Dey , A. Kundu , A. Kumar , M. Gupta , B. M. Lee , and T. Bhakta , “Analysis of Alkaloids (Indole Alkaloids, Isoquinoline Alkaloids, Tropane Alkaloids),” in Recent Advances in Natural Products Analysis, eds. A. S. Silva , S. F. Nabavi , M. Saeedi , and S. M. Nabavi (Elsevier, 2020), 505–567, 10.1016/B978-0-12-816455-6.00015-9.
- Two Faces of Alkaloids,”. Journal of Chemical Education, 2000. [DOI]
- Techniques for Extraction of Bioactive Compounds From Plant Materials: A Review,”. Journal of Food Engineering, 2013. [DOI]
- Anatomical Adaptations of Four Crassula Species to Water Availability,”. Bioscience Horizons: The International Journal of Student Research, 2011. [DOI]
- The Isolation of Morphine by Serturner,”. Indian Journal of Anaesthesia, 2016. [DOI | PubMed]
- Isolating Colchicine in 19th Century: An Old Drug Revisited,”. Current Pharmaceutical Design, 2018. [DOI | PubMed]
- Neuropharmacological Potentials of β‐Carboline Alkaloids for Neuropsychiatric Disorders,”. European Journal of Pharmacology, 2021. [DOI | PubMed]
- Versatile Acid Base Sustainable Solvent for Fast Extraction of Various Molecular Weight Chitin From Lobster Shell,”. Carbohydrate Polymers, 2018. [DOI | PubMed]
- Green Formulation for Synthetic Dye Extraction Using Synergistic Mixture of Acid‐Base Extractant,”. Separation and Purification Technology, 2019. [DOI]
- Atividade Antiproliferativa dos Extratos e da Fração Orgânica Obtidos das Folhas de Virola sebifera Aubl. (Myristicaceae),”. Revista Brasileira de Farmacognosia, 2007. [DOI]
- Visually Tracking Acid–Base Extractions Using Colorful Compounds,”. Journal of Chemical Education, 2020. [DOI]
- Acid–Base Reaction‐Based Dispersive Liquid–Liquid Microextraction Method for Extraction of Three Classes of Pesticides From Fruit Juice Samples,”. Journal of Chromatography A, 2016. [DOI | PubMed]
- Green Extraction of Natural Products: Concept and Principles,”. International Journal of Molecular Sciences, 2020. [DOI | PubMed]
- Extraction of Natural Substances With Supercritical Gases, 10. Communication: Qualitative and Quantitative Determination of Solubilities of Steroids in Supercritical Carbon Dioxide,”. Fette, Seifen, Anstrichmittel, 1984. [DOI]
- Supercritical Fluids in Separation Science—The Dreams, the Reality and the Future,”. Journal of Chromatography A, 1999. [DOI | PubMed]
- Sub‐and Supercritical Fluid Extraction of Functional Ingredients From Different Natural Sources: Plants, Food‐By‐Products, Algae and Microalgae: A Review,”. Food Chemistry, 2006. [DOI]
- Supercritical Fluid Extraction With Cosolvent of Alkaloids From Lupinus mutabilis Sweet and Comparison With Conventional Method,”. Journal of Food Process Engineering, 2021. [DOI]
- Microwave Extraction: A Novel Sample Preparation Method for Chromatography,”. Journal of Chromatography A, 1986. [DOI]
- 47 F. Chemat , G. Cravotto , eds., Microwave‐Assisted Extraction for Bioactive Compounds: Theory and Practice (Springer, 2012).
- Rapid and Green Analytical Method for the Determination of Quinoline Alkaloids From Cinchona succirubra Based on Microwave‐Integrated Extraction and Leaching (MIEL) Prior to High Performance Liquid Chromatography,”. International Journal of Molecular Sciences, 2011. [DOI | PubMed]
- Microwave‐Assisted Extraction of Bioactive Alkaloids From Stephania sinica ,”. Separation and Purification Technology, 2014. [DOI]
- Advances in Ultrasound‐Assisted Extraction of Bioactive Compounds From Cash Crops—A Review,”. Ultrasonics Sonochemistry, 2018. [DOI | PubMed]
- Application of Ultrasound as Clean Technology for Extraction of Specialized Metabolites From Stinging Nettle (Urtica dioica L.),”. Frontiers in Nutrition, 2022. [DOI | PubMed]
- An Overview on the Application of Ultrasound in Extraction, Separation and Purification of Plant Polysaccharides,”. Central European Journal of Chemistry, 2010. [DOI]
- Innovative Applications of Ionic Liquids as “Green” Engineering Liquids,”. Chemical Engineering Communications, 2006. [DOI]
- Effective Extraction of Bioactive Alkaloids From the Roots of Stephania tetrandra by Deep Eutectic Solvents‐Based Ultrasound‐Assisted Extraction,”. Journal of Chromatography A, 2023. [DOI | PubMed]
- The Most Potent Natural Pharmaceuticals, Cosmetics, and Food Ingredients Isolated From Plants With Deep Eutectic Solvents,”. Journal of Agricultural and Food Chemistry, 2023. [DOI | PubMed]
- Are Natural Deep Eutectic Solvents the Missing Link in Understanding Cellular Metabolism and Physiology?”. Plant Physiology, 2011. [DOI | PubMed]
- Green Solvents From Ionic Liquids and Deep Eutectic Solvents to Natural Deep Eutectic Solvents,”. Comptes Rendus Chimie, 2018. [DOI]
- Enhancement of Solasodine Extracted From Fruits of Solanum nigrum L. by Microwave‐Assisted Aqueous Two‐Phase Extraction and Analysis by High‐Performance Liquid Chromatography,”. Molecules, 2019. [DOI | PubMed]
- Methods of Extraction and Fractionation of Bio‐Active Components of Morinda coreia for Anti‐Tubercular and Anticancer Activities,”. International Journal of Pharmaceutical Sciences and Research, 2020. [DOI]
- Ultrasound‐Assisted and Microwave‐Assisted Extraction, GC‐MS Characterization and Antimicrobial Potential of Freeze‐Dried L. camara Flower,”. Journal of Pure and Applied Microbiology, 2022. [DOI]
- Optimized Microwave‐Assisted Extraction (MAE) of Alkaloids and Polyphenols From Berberis Roots Using Multiple‐Component Analysis,”. Scientific Reports, 2020. [DOI]
- Extraction of Bioactive Compounds From Senecio brasiliensis Using Emergent Technologies,”. 3 Biotech, 2021. [DOI]
- Rapid and Green Extraction, Assisted by Microwave and Ultrasound of Cepharanthine From Stephania rotunda Lour,”. Separation and Purification Technology, 2014. [DOI]
- Microwave‐Assisted Extraction Followed by Solid‐Phase Extraction for the Chromatographic Analysis of Alkaloids in Stephania cepharantha ,”. Journal of Chromatographic Science, 2016. [DOI | PubMed]
- Ultrasound/Microwave‐Assisted Extraction and Comparative Analysis of Bioactive/Toxic Indole Alkaloids in Different Medicinal Parts of Gelsemium elegans Benth by Ultra‐High Performance Liquid Chromatography With MS/MS,”. Journal of Separation Science, 2014. [DOI | PubMed]
- Simultaneous Extraction and Purification of Alkaloids From Sophora flavescens Ait. By Microwave‐Assisted Aqueous Two‐Phase Extraction With Ethanol/Ammonia Sulfate System,”. Separation and Purification Technology, 2015. [DOI]
- Microscopic Characters, Phytochemical Screening Focus on Alkaloid and Total Phenolic Content of Uncaria Gambir Roxb. and Uncaria sclerophylla Roxb. Leaves,”. Pharmacognosy Journal, 2019. [DOI]
- Microwave‐Assisted Extraction in Combination With HPLC‐UV for Quantitative Analysis of Six Bioactive Oxoisoaporphine Alkaloids in Menispermum dauricum DC,”. Biomedical Chromatography, 2015. [DOI | PubMed]
- Microwave‐Assisted Extraction of Three Bioactive Alkaloids From Peganum harmala L. and Their Acaricidal Activity Against Psoroptes cuniculi In Vitro,”. Journal of Ethnopharmacology, 2016. [DOI | PubMed]
- Microwave‐Assisted Process Intensification and Kinetic Modelling: Extraction of Camptothecin From Nothapodytes nimmoniana Plant,”. Industrial Crops and Products, 2017. [DOI]
- Modern Extraction Techniques Optimized to Extract Betacyanins From Gomphrena globosa L.,”. Industrial Crops and Products, 2017. [DOI]
- Microwave‐Assisted Extraction of Ricinine From Ricinus communis Leaves,”. Antioxidants, 2019. [DOI | PubMed]
- Simultaneous Quantification of Vasicine and Vasicinone in Different Parts of Justicia adhatoda Using High‐Performance Thin‐Layer Chromatography–Densitometry: Comparison of Different Extraction Techniques and Solvent Systems,”. Journal of Planar Chromatography—Modern TLC, 2020. [DOI]
- Carpaine: Multitarget Docking for Its Antiproliferative Potential on CHO‐K1 Human Ovarian Carcinoma,”. Egyptian Journal of Chemistry, 2023. [DOI]
- Environmentally Benign Supercritical CO2 Extraction of Galanthamine From Floricultural Crop Waste of Narcissus pseudonarcissus ,”. Journal of Supercritical Fluids, 2014. [DOI]
- Analysis of Supercritical Fluid Extraction of Lycopodine Using Response Surface Methodology and Process Mathematical Modeling,”. Chemical Engineering Research and Design, 2015. [DOI]
- Cyclopamine Bioactivity by Extraction Method From Veratrum californicum ,”. Bioorganic & Medicinal Chemistry, 2016. [DOI | PubMed]
- Alkaloids From Chelidonium majus L.: Fractionated Supercritical CO2 Extraction With Co‐Solvents,”. Separation and Purification Technology, 2016. [DOI]
- Effect of Solvent Type and Ratio on Betacyanins and Antioxidant Activity of Extracts From Hylocereus polyrhizus Flesh and Peel by Supercritical Fluid Extraction and Solvent Extraction,”. Food Chemistry, 2016. [DOI | PubMed]
- Controlling Concentration of Bioactive Components in Cat’s Claw‐Based Products With a Hybrid Separation Process,”. Journal of Supercritical Fluids, 2017. [DOI]
- Extraction of Bioactive Alkaloids From Melocactus zehntneri Using Supercritical Fluid,”. Journal of Supercritical Fluids, 2017. [DOI]
- Extraction of Total Alkaloids, Peimine and Peiminine From the Flower of Fritillaria thunbergii Miq Using Supercritical Carbon Dioxide,”. Journal of CO2 Utilization, 2017. [DOI]
- Colchicine: Isolation, LC‐MS QTOF Screening, and Anticancer Activity Study of Gloriosa superba Seeds,”. Molecules, 2019. [DOI | PubMed]
- Usage of Supercritical Fluid Techniques to Obtain Bioactive Alkaloid‐Rich Extracts From Cherimoya Peel and Leaves: Extract Profiles and Their Correlation With Antioxidant Properties and Acetylcholinesterase and α‐Glucosidase Inhibitory Activities,”. Food & Function, 2020. [DOI | PubMed]
- Micronization of Thebaine Extracted From Papaver bracteatum Lindl. Using Supercritical Fluid Technology,”. Journal of AOAC International, 2021. [DOI]
- Alkaloid‐Rich vs. Antioxidant‐Rich Yerba Mate (Ilex paraguariensis) Extracts: Exploiting the Selectivity of Supercritical CO2 Using Hydrated Ethanol as Co‐Solvent,”. Journal of Supercritical Fluids, 2021. [DOI]
- Optimization of Supercritical‐CO2 Extraction and Pharmacokinetics in SD Rats of Alkaloids From Sophora moorcroftiana Seed,”. Scientific Reports, 2022. [DOI | PubMed]
- Ionic Liquid‐Supported Solid–Liquid Extraction of Bioactive Alkaloids. IV. New HPLC Method for Quantitative Determination of Galantamine in Leucojum aestivum L. (Amaryllidaceae),”. Separation Science and Technology, 2016. [DOI]
- Rapid Determination of Alkaloids in Macleaya cordata Using Ionic Liquid Extraction Followed by Multiple Reaction Monitoring UPLC–MS/MS Analysis,”. Journal of Pharmaceutical and Biomedical Analysis, 2017. [DOI | PubMed]
- Ionic Liquid‐Aqueous Solution Ultrasonic‐assisted Extraction of Three Kinds of Alkaloids From Phellodendron amurense Rupr and Optimize Conditions Use Response Surface,”. Ultrasonics Sonochemistry, 2015. [DOI | PubMed]
- Ionic Liquid‐Supported Solid‐Liquid Extraction of Bioactive Alkaloids. III. Ionic Liquid Regeneration and Glaucine Recovery From Ionic Liquid‐Aqueous Crude Extract of Glaucium flavum Cr. (Papaveraceae),”. Separation and Purification Technology, 2015. [DOI]
- Ionic Liquid‐Based Aqueous Biphasic Systems—A Facile Approach for Ionic Liquid Regeneration From Crude Plant Extracts,”. Processes, 2015. [DOI]
- Green Determination of Aconitum Alkaloids in Aconitum carmichaeli (Fuzi) by an Ionic Liquid Aqueous Two‐phase System and Recovery of the Ionic Liquid Coupled With In Situ Liquid–liquid Microextraction,”. Analytical Methods, 2016. [DOI]
- Ionic‐Liquid‐Based Infrared‐Assisted Extraction (IL‐IRAE) Coupled With HPLC–MS: A Green and Convenient Tool for Determination of TCMs,”. Chromatographia, 2017. [DOI]
- Extraction of Bioactive Compounds From Catharanthus roseus and Vinca minor ,”. Separation and Purification Technology, 2020. [DOI]
- Development of a Novel Functionality for a Highly Efficient Imidazole‐Based Ionic Liquid Non‐Aqueous Solvent System for the Complete Extraction of Target Alkaloids From Phellodendron amurense Rupr. Under Ultrasound‐Assisted Conditions,”. Industrial Crops and Products, 2021. [DOI]
- Deep Eutectic Solvents: Syntheses, Properties and Applications,”. Chemical Society Reviews, 2012. [DOI | PubMed]
- Deep Eutectic Solvents in Membrane Science and Technology: Fundamental, Preparation, Application, and Future Perspective,”. Separation and Purification Technology, 2021. [DOI]
- Green Techniques in Comparison to Conventional Ones in the Extraction of Amaryllidaceae Alkaloids: Best Solvents Selection and Parameters Optimization,”. Journal of Chromatography A, 2018. [DOI | PubMed]
- 100 Y. V. D. Nageswar , K. Ramesh , and R. Katla , in “Deep Eutectic Solvent‐Mediated Organic Transformations,” in Ionic Liquids, Deep Eutectic Solvents, Crown Ethers, Fluorinated Solvents, Glycols and Glycerol, eds. C. Mukhopadhyay and B. Banerjee (De Gruyter, 2023): 107–144.
- Green and Efficient Extraction of Different Types of Bioactive Alkaloids Using Deep Eutectic Solvents,”. Microchemical Journal, 2019. [DOI]
- Advancing Green Extraction of Bioactive Compounds Using Deep Eutectic Solvent‐Based Ultrasound‐Assisted Matrix Solid‐Phase Dispersion: Application to UHPLC‐PAD Analysis of Alkaloids and Organic Acids in Coptidis rhizoma ,”. Talanta, 2024. [DOI | PubMed]
- Ecofriendly Mechanochemical Extraction of Bioactive Compounds From Plants With Deep Eutectic Solvents,”. ACS Sustainable Chemistry & Engineering, 2017. [DOI]
- Extraction Techniques With Deep Eutectic Solvents,”. TrAC Trends in Analytical Chemistry, 2018. [DOI]
- Evaluation of the Influence of Green Extraction Solvents on the Cytotoxic Activities of Crinum (Amaryllidaceae) Alkaloid Extracts Using In‐Vitro‐In‐Silico Approach,”. Journal of Ethnopharmacology, 2018. [DOI | PubMed]
- NADES‐Mediated Folk Plant Extracts as Novel Antifungal Agents Against Candida albicans ,”. Journal of Pharmaceutical and Biomedical Analysis, 2019. [DOI | PubMed]
- Biomolecules Extraction From Coffee and Cocoa By‐ and Co‐Products Using Deep Eutectic Solvents,”. Journal of the Science of Food and Agriculture, 2020. [DOI | PubMed]
- Green Extraction Methods for Active Compounds From Food Waste—Cocoa Bean Shell,”. Foods, 2020. [DOI | PubMed]
- Ultrasound‐assisted Extraction of Bioactive Alkaloids From Phellodendri amurensis Cortex Using Deep Eutectic Solvent Aqueous Solutions,”. New Journal of Chemistry, 2020. [DOI]
- Green Extraction of Alkaloids and Polyphenols From Peumus boldus Leaves With Natural Deep Eutectic Solvents and Profiling by HPLC‐PDA‐IT‐MS/MS and HPLC‐QTOF‐MS/MS,”. Plants, 2020. [DOI | PubMed]
- Arecanut (Areca catechu L.) Seed Extracts Extracted by Conventional and Eco‐Friendly Solvents: Relation Between Phytochemical Compositions and Biological Activities by Multivariate Analysis,”. Journal of Applied Research on Medicinal and Aromatic Plants, 2021. [DOI]
- Extraction and Stabilization of Betalains From Beetroot (Beta vulgaris) Wastes Using Deep Eutectic Solvents,”. Molecules, 2021. [DOI | PubMed]
- Evaluation of Green and Efficient Deep Eutectic Solvents as Media for Extracting Alkaloids From Lotus Leaf,”. Biomedical Chromatography, 2022. [DOI | PubMed]
- Effectiveness of Volatile Natural Deep Eutectic Solvents (VNADESs) for the Green Extraction of Chelidonium majus Isoquinoline Alkaloids,”. Molecules, 2022. [DOI | PubMed]
- A Deep Eutectic Solvent Binary‐Phase System for Alkaloid Extraction From Chinese Herb Evodia lepta Residue and Its Mechanism,”. Journal of Cleaner Production, 2023. [DOI]
- Morphological Characterization of Nicotiana tabacum Inflorescences and Chemical‐Functional Analysis of Extracts Obtained From Its Powder by Using Green Solvents (NaDESs),”. Plants, 2023. [DOI]
- Nicotiana tabacum Leaf Waste: Morphological Characterization and Chemical‐Functional Analysis of Extracts Obtained From Powder Leaves by Using Green Solvents,”. Molecules, 2023. [DOI | PubMed]
- NADES‐Based Selective Extraction of Bioactive Molecules: A Case Study With Commercially Important Himalayan Medicinal Plant Aconitum heterophyllum ,”. Sustainable Chemistry and Pharmacy, 2023. [DOI]
- Therapeutic Product Based on Yerba Mate Extract and Eutectic System by One Step Ultrasound Approach,”. Sustainable Chemistry and Pharmacy, 2023. [DOI]
- Solubility and Stability of Some Pharmaceuticals in Natural Deep Eutectic Solvents‐Based Formulations,”. Molecules, 2021. [DOI | PubMed]
- An Experimental and Theoretical Investigation of the Physicochemical Properties on Choline Chloride–Lactic Acid‐Based Natural Deep Eutectic Solvent (NADES),”. Journal of Molecular Liquids, 2019. [DOI]
- Which Is the Best Food Emerging Solvent: IL, DES, or NADES?,”. Trends in Food Science & Technology, 2019. [DOI]
