Mechanisms underlying anxiety in Rett Syndrome: Translational insights from preclinical findings
Abstract
The role of serotonin in mechanisms of anxiety in Rett syndrome appears complex.
Mecp2 mutation alters HPA axis function but this is understudied in Rett syndrome.
Rett syndrome may inform understanding of anxiety in those with early life stress.
Article type: Review Article
Keywords: Rett syndrome, Anxiety, Epigenetics, Translational research
License: © 2022 The Authors CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.nsa.2022.100109 | PubMed: 40656225 | PMC: PMC12244188
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (3.0 MB)
Nomenclature:
MECP2
is the gene in humans
Mecp2
is the murine gene
MeCP2
is the protein in mouse and humans
Introduction
Rett syndrome is the second most common cause of severe learning disability in females (occurring in approximately 1:10,000 females). Babies born with Rett syndrome initially develop apparently normally, however, at the age of 6–18 months, development falters and subsequently regresses (Box 1). Rett syndrome has multiple comorbid conditions affecting overlapping physiological systems. Despite this heterogeneity, anxiety is a common feature amongst the comorbidities of Rett syndrome (ref. Gold et al., 2017). The mechanisms underlying anxiety are the focus of this review as further study may lead to new insights into the pathophysiology of anxiety.
Box 1
Stages of Rett syndrome (defined by Hanefeld, 1985) (Gold et al., 2017; Zhou et al., 2017)
| Stage (name) | Age | Clinical features |
| Stage I (‘stagnation’) | 6–18 months | Development slows and gain of new skills stops. |
| Stage II (‘regression’) | 1–4 years | Rapid loss of skills that have been gained, including communication, social skills and motor skills. Development of stereotypic hand-movements. Other common features that develop are seizures, disordered breathing (with hyperventilation and apneas), gastrointestinal complications and anxiety. |
| Stage III (‘pseudostationary’) | 2–10 years | There is a stabilisation and some skills that were lost in the regression stage may be regained. |
| Stage IV (‘motor deterioration’) | Can last for decades | There is a decrease in mobility, with muscle weakness or spasticity. Cognition and language skills do not alter. |
Alt-text: Box 1
The challenge of identifying anxiety in individuals with severe intellectual disability has been recognised. Two validated questionnaires have been developed for identifying presumed anxiety-related behaviours in Rett Syndrome: the Rett Syndrome Behaviour Questionnaire (RSBQ) and the Anxiety, Depression and Mood Scale (ADAMS) (ref. Esbensen et al., 2003; ref. Mount et al., 2002). These questionnaires rely on carer observation. The RBSQ was validated by comparing results in individuals with Rett syndrome and those with other severe intellectual disability; control for physical impairment was also included (ref. Mount et al., 2002). Anxiety symptoms in Rett syndrome appear similar to generalised anxiety and social anxiety as they include worsening of hyperventilation, inconsolable crying, trembling in the absence of fearful situations and withdrawal from social contact (ref. Buchanan et al., 2019).
While the diagnosis of Rett syndrome is made using clinical features, it is usually caused by mutations on chromosome Xq28 in the gene encoding methyl-CpG-binding protein 2 (MECP2). MeCP2 is found in most tissues, but is most abundant in the central nervous system (CNS) (ref. Shahbazian et al., 2002). A key role of MeCP2 is to bind to methylated DNA and it can both activate and repress gene expression, therefore acting as an epigenetic regulator. The effects of MeCP2 are cell-type and region specific and the level of MeCP2 within a cell is critical to the function of the cell. MeCP2 expression increases in the early months of life and the timing of expression in particular brain regions correlates with the maturation of the CNS (a time when sensory-driven neural activity shapes the CNS circuitry) (ref. Zimmermann et al., 2015). In Rett syndrome there is mosaicism (only the maternal or paternal X chromosome will be expressed in any one cell in the body) of MECP2 gene expression and this leads to variability in phenotype (ref. Gold et al., 2017). Further variations in expression of the condition are likely influenced by the numerous mutations in MECP2; to date 518 likely pathogenic mutations have been identified.
A review of autonomic dysregulation in individuals with Rett syndrome has highlighted the complexity of treatment (ref. Singh and Santosh, 2018). Treatments for anxiety disorders in neurotypical individuals are often suboptimal. This is further complicated in neurodivergent individuals in whom the mechanisms and treatments for anxiety are poorly understood. Symptoms suggestive of anxiety are found more commonly in Rett syndrome compared to individuals with other severe intellectual disability (ref. Mount et al., 2002) suggesting the mechanisms of anxiety may be entwined with the disorder. Greater understanding of anxiety mechanisms in Rett syndrome could facilitate development of more specific treatments and improve our mechanistic understanding of anxiety disorders in general.
A number of preclinical models of Rett syndrome have been developed in mice; many of these show similar features to patients with Rett syndrome, including anxiety-like behaviour, irregular breathing, motor impairment and seizures (ref. Katz et al., 2012). This review focuses on the preclinical evidence base. It highlights the heterogeneity in the mutations being studied, the study designs and the need for standardisation of behavioural assays. Despite these limitations, these models provide a system for the study of the cellular and molecular mechanisms of MeCP2 dysfunction and associated alterations in anxiety-like behaviour. The discussion draws together findings from pre-clinical studies with clinical observations and suggests future directions for Rett syndrome translational research, and postulates that there may be broader implications for other disorders of MeCP2 dysfunction where anxiety is a feature.
Methods
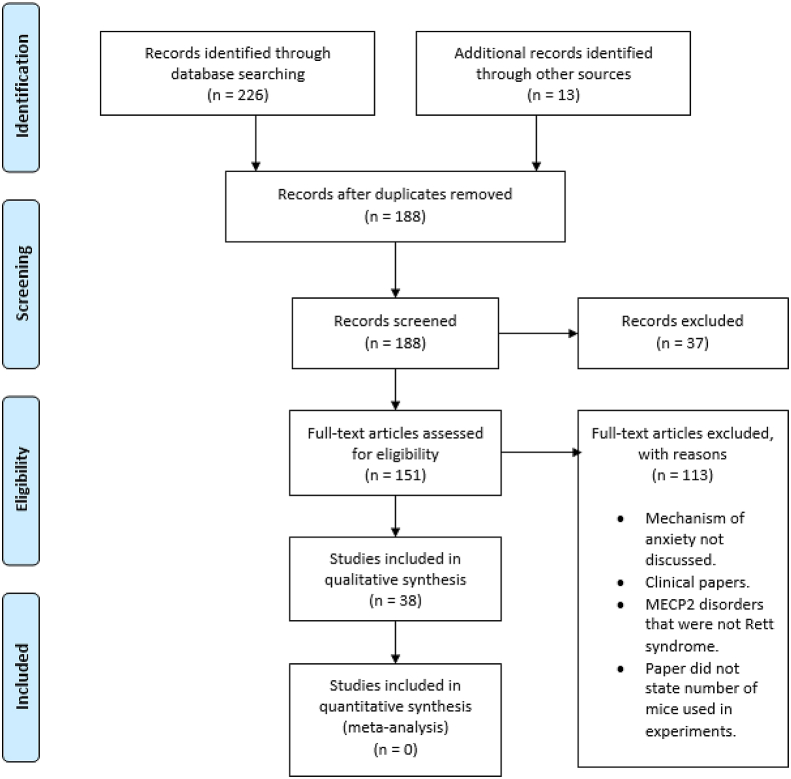
A systematic review was carried out using the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines (ref. Moher et al., 2009). Records were searched through PubMed, MEDLINE and Embase. The following search terms were used [Rett syndrome] AND [anxiety]. Searching was completed on February 3, 2022. Fig. 1 illustrates the selection process. All included studies made statements that animal experiments complied with ethical guidelines, except for ref. McGill et al., 2006, ref. Orefice et al., 2016 and ref. Wither et al., 2013 (ref. McGill et al., 2006; ref. Orefice et al., 2016; ref. Wither et al., 2013), where statements were not explicitly made. All included studies were published in peer-reviewed journals.
Results
Overview
Anxiety is postulated to develop when there are disruptions in the interconnected macro- and micro-circuits that process stimuli detected from the external world. As such, the first stage in the anxiety macrocircuit involves the sensory systems. Sensory signals are interpreted as potentially threatening through the interconnected activity (via neuronal projections between regions) of the amygdala, bed nucleus of the stria terminalis, ventral hippocampus and prefrontal cortex. Evaluation, where the interpreted signals are regulated, then occurs. This involves the medial prefrontal cortex, ventral tegmental area, nucleus accumbens, ventral pallidum, and hypothalamus. Following coordinated processing of a threat stimulus, a response is initiated (ref. Calhoon and Tye, 2015). Microcircuits function within each region of the anxiety macrocircuit and involve short-range connections between neuronal subtypes and associated interactions with glial cells, the blood vessel endothelium and surrounding specialised extracellular matrix (perineuronal net). The account of the preclinical studies has been structured to highlight the effect of MeCP2 loss in anxiety macrocircuit regions and within cellular subtypes on anxiety-like behaviour and/or physiological response (illustrated within Fig. 2) and subsequently summarises potential underlying molecular mechanisms. At present, there is limited literature examining macro- and micro-circuitry connections, which is briefly described in section 3.6. However, an effect of MeCP2 loss in CNS regions and cell subtypes on anxiety behaviour may suggest an impact on the anxiety circuitry, albeit with significant limitation on interpretation.
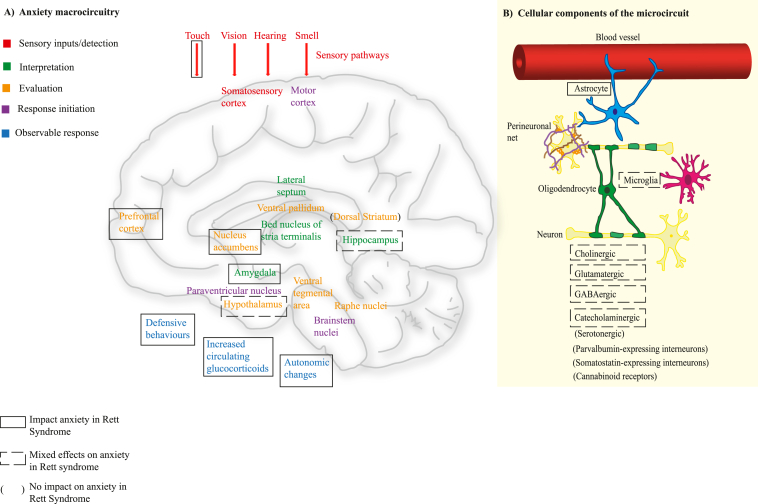
The tests of anxiety-like behaviours are described within Box 2. Generation of the mouse models relies on mutation of the Mecp2 alleles. In keeping with the variability of mutations identified in people with Rett syndrome, the mouse models involve varying degrees of severity of mutation – null alleles, large mutations and point mutations. For clarity, male Rett syndrome mice are referred to as ‘Mecp2 null’ and female Rett syndrome mice are referred to as ‘Mecp2+/−‘. Most earlier Rett syndrome studies used male mice, although this is changing with the recognition of the importance of studying both sexes and the obvious importance of using female mice given that in humans, females are affected. The reason that male mice had been predominantly used is because Mecp2 mutation causes a less severe phenotype in mice than humans and the male mice show a more pronounced phenotype than females. In addition, females have a more variable phenotype than males, due to mosaicism (ref. Ribeiro and MacDonald, 2020). Table 1 documents studies that correlate molecular changes with anxiety behaviour; included studies are arranged alphabetically on the basis of the first author(s). This table details experimental conditions and enables changes in anxiety behaviour to be identified within and between studies, as well as highlighting whether male and/or female mice were used. Outcomes of the studies within the table are summarised in the text. The sections documenting MeCP2 loss in macrocircuit regions and cell subtypes have been structured to summarise where this was associated with a change/no change/mixed effects on anxiety behaviour. The description ‘mixed effects’ was determined by there being inconsistent changes between the different behavioural tests within a study. The impacts of MeCP2 loss within anxiety macrocircuit regions and cell subtypes are summarised within Fig. 2. Details of the mutation used in each study have been documented within Table 2.
Table 1: ↑ increased ‘anxiety’ compared to control, ↓ decreased ‘anxiety’ compared to control, ↔ no significant difference compared to control, – no result). ‘/’ is used where more than one measure was used and indicated differing outcome: for example, in ref. Adachi et al., (2009), in the open field test there was no significant difference in time in the centre, but there was a significant difference in ratio of time in centre over time in the periphery. Flox mice show 50% reduction in MeCP2 expression. Some studies used this as a control, so a potential change in anxiety behaviour may be under-represented by this (Samaco, 2008).
| STUDY [REFERENCE] | MECP2 MUTATION (AND EXPERIMENTAL CONDITIONS, WHERE RELEVANT) | MOUSE GENDER | DIFFERENCE IN ANXIETY WITH MECP2 MUTATION±EXPERIMENTAL CONDITION COMPARED TO CONTROL (WITHOUT ADDITIONAL EXPERIMENTAL CONDITION, UNLESS OTHERWISE STATED) | |||
|---|---|---|---|---|---|---|
| Elevated plus maze | Light-dark assay | Open field test | Elevated zero maze | |||
| ref. Abellán-Álvaro et al., 2021 | Mecp2+/− mice | Female | ↓ | – | ↔ | – |
| Mecp2+/− mice that were separated from their mother. | Female | ↓ | – | ↔ | – | |
| ref. Adachi et al., 2009∗ control for genetic carrier system used instead of wildtype | Specific deletion of Mecp2 from the basolateral amygdala in mice | Male | ↑ | – | ↑/↔ | – |
| ref. Bittolo et al., 2016 | Mecp2 null mice treated with (control solution), | Male | ↓/↔ | – | – | – |
| Mecp2 null mice treated with mirtazapine. | Male | ↓/↔ | – | – | – | |
| Mecp2 null mice treated with desipramine. | Male | ↓/↔ | – | – | – | |
| ref. Castro et al., 2014 | Mecp2 null mice. | Male | ↓ | – | – | – |
| Mecp2 null mice with intraperitoneal rhIGF-1 treatment. | Male | ↔ | – | – | – | |
| ref. Chao et al., 2010 | Specific deletion of Mecp2 in GABAergic neurons. | Male | ↔/↑ | ↔ | ↑ | – |
| ref. Chin et al., 2019 | Mice with specific deletion of Mecp2 in excitatory neurons of the mouse forebrain. | Male | – | – | ↑ | – |
| Mice with specific deletion of Mecp2 in excitatory neurons of the forebrain, with choline supplementation. | Male | – | – | ↔ | – | |
| ref. De Filippis et al., 2014 | Mecp2 null mice. | Male | – | ↑/↓/↔ | – | – |
| Mecp2 null mice with intraperitoneal injection of a 5-HT7R selective agonist. | Male | – | ↔ | – | – | |
| ref. Derecki et al., 2012 | MECP2+/− mice | Female | – | – | ↑ | – |
| Mecp2+/− mice that were irradiated to kill resident microglia and then given bone marrow transplant from wildtype mice. | Female | – | – | ↔ | – | |
| ref. Flores Gutiérrez et al., 2020 | Mecp2+/− mice treated with control solution. | Female | ↓ | ↔/↑ | ↔ | – |
| Mecp2+/− mice with trimmed whiskers treated with control solution. | Female | ↔ | – | – | – | |
| Mecp2+/− mice (untrimmed whiskers) treated with mirtazapine | Female | ↔ | – | – | – | |
| ref. Fyffe et al., 2008 | Selective deletion of Mecp2 in Sim-1 expressing neurons, with associated reduction in MeCP2 expression in the paraventricular, supraoptic, and posterior hypothalamic nuclei, and in the nucleus of the lateral olfactory tract of the amygdala. | Male | – | ↔ | ↑ | – |
| ref. Gemelli et al., 2006 | Selective deletion of Mecp2 in the forebrain (prefrontal cortex, striatum, nucleus accumbens, hippocampus and amygdala) | Male | ↑ | – | ↑ | – |
| ref. Goffin et al., 2014∗loxp control used instead of wildtype | Specific deletion of Mecp2 in forebrain glutamatergic neurons and glial cells. | Male | – | – | ↔ | ↔ |
| ref. Hao et al., 2015 | Mecp2+/− mice exposed to deep brain stimulation (DBS). | Female | – | ↓ | ↓ | – |
| Mecp2+/− mice exposed to sham DBS. | Female | – | ↓ | ↓ | – | |
| Mecp2+/− mice exposed to no DBS/sham DBS. | Female | – | ↔ | ↔ | – | |
| ref. He et al., 2014 | Deletion of Mecp2 in parvalbumin-expressing interneurons | Male | ↔ | ↔ | ↔ | – |
| ref. Ito-Ishida et al., 2015 | Deletion of Mecp2 in parvalbumin-expressing interneurons. | Male | ↔ | ↔ | – | – |
| Deletion of Mecp2 in somatostatin-expressing interneurons | Male | ↔ | ↔ | – | – | |
| ref. Kerr et al., 2010 | Mecp2 null mice, 129/SvJ background, standard housed. | Male | ↓/↔ | – | – | – |
| Mecp2 null mice, 129/SvJ background, with environmental enrichment. | Male | ↔ | – | – | – | |
| Mecp2 null mice, 129/SvJ and C57BL/6J mixed background, standard housed. | Male | ↓/↔ | – | – | – | |
| Mecp2 null mice, 129/SvJ and C57BL/6J mixed background, with environmental enrichment. | Male | ↔ | – | – | – | |
| ref. Kondo et al., 2016 | Mecp2+/− mice, standard housed. | Female | ↓ | – | – | – |
| MECP2+/− mice with environmental enrichment. | Female | ↔ | – | – | – | |
| ref. Lang et al., 2012 | Mecp2 null mice. | Male | – | ↓ | ↓ | – |
| Mecp2 null mice with Mecp2 rescue in catecholaminergic neurons. | Male | – | ↓ (but significantly ↑ compared to null) | ↓ (but significantly ↑ compared to null) | – | |
| ref. Lioy et al., 2011∗ compared to control for genetic carrier system instead of wildtype | Mecp2 null mice. | Male | – | – | ↑ | ↓ |
| Mecp2 null mice with selective reintroduction of Mecp2 in astrocytes. | Male | – | – | ↑(but significantly ↓ compared to null) | ↓(but significantly ↑ compared to null) | |
| ref. Mantis et al., 2009 | Mecp2 null mice with standard diet. | Male | – | ↑ | ↔ | – |
| Mecp2 null mice with restricted ketogenic diet. | Male | – | ↑/↔ | ↔/↓/↑ | – | |
| Mecp2 null mice with caloric restriction. | Male | – | ↔ | ↔/↓ | – | |
| ref. Mellios et al., 2014 | Mecp2+/− mice. | Female | – | – | ↑ | – |
| Mecp2+/− mice with clenbuterol treatment. | Female | – | – | ↑ (but ↓ compared to MECP2+/− mice without clenbuterol) | – | |
| ref. Meng et al., 2016 | Mecp2 null mice. | Male | ↓ | ↑ | – | – |
| Mice with specific deletion of Mecp2 in glutamatergic neurons. | Male | ↓ | ↑ | – | – | |
| Mecp2 null mice that had specific reintroduction of Mecp2 in glutamatergic neurons. | Male | ↔ | ↔ | – | – | |
| Mecp2+/− mice, age 10 weeks | Female | ↓ | – | – | – | |
| Mecp2+/− mice with specific reintroduction of Mecp2 in glutamatergic neurons, age 10 weeks. | Female | ↔ | – | – | – | |
| Mecp2+/− mice, age 30 weeks | Female | ↔ | – | – | – | |
| Mecp2+/− mice with specific reintroduction of Mecp2 in glutamatergic neurons, age 30 weeks. | Female | ↔ | – | – | – | |
| ref. Mossner et al., 2020∗flox mice used as control instead of wildtype | Deletion of Mecp2 in VIP-expressing interneurons | Male | ↔ | – | ↔ | – |
| Deletion of Mecp2 in parvalbumin-expressing interneurons. | Male | ↔ | – | ↔ | – | |
| Deletion of Mecp2 in somatostatin-expressing interneurons | Male | ↔ | – | ↔ | – | |
| Deletion of Mecp2 in VIP-, parvalbumin- and somatostatin-expressing interneurons | Male | ↔ | – | ↔ | – | |
| ref. Orefice et al., 2016∗controls – wildtype and mice lacking the genetic carrier system – paper was not fully clear about which controls were used in the individual experiments. | Deletion of Mecp2 in dorsal root ganglion (DRG) and trigeminal somatosensory neurons | Male | ↑ | – | ↑ | – |
| Female | – | – | ↑ | – | ||
| Deletion of Mecp2 in DRG during adult life (as opposed to early life) | Male | ↔ | – | ↔ | – | |
| Female | – | – | ↔ | – | ||
| Mecp2 null mice | Male | ↑ | – | ↑ | – | |
| Mecp2 null mice with specific reintroduction of Mecp2 in peripheral sensory neurons | Male | ↔ | – | ↔ | – | |
| ref. Ricceri et al., 2011 | Mecp2 null mice | Male | – | – | ↑ | – |
| Mecp2 null mice with choline supplementation (behavioural tests at postnatal day 60). | Male | – | – | ↔ | – | |
| ref. Samaco et al., 2009 | Specific deletion of Mecp2 in Th-expressing DA/NE neurons. | Male | – | ↔ | – | – |
| Specific deletion of Mecp2 in PC12 ets factor 1 (PET1)-expressing serotonergic neurons | Male | – | ↔ | – | – | |
| Mecp2+/− mice. | Female | – | ↓ | ↓ | – | |
| Mecp2+/− mice with Mecp2 rescue in catecholaminergic neurons. | Female | – | ↔ | ↔ | – | |
| ref. Su et al., 2015 | Specific deletion of Mecp2 in the striatum. | Male | – | – | ↔ | ↔Compared to flox, not wildtype |
| Mecp2 null mice. | Male | – | – | ↑ | – | |
| Mecp2 null mice with specific re-expression of Mecp2 in the striatum. | Male | – | – | ↑ | – | |
| Mecp2+/− mice. | Female | – | – | ↔ | – | |
| Mecp2+/− mice with specific re-expression of Mecp2 in the striatum. | Female | – | – | ↔ | – | |
| ref. Ure et al., 2016 | Mecp2 null mice. | Male | ↓ | ↑ | – | – |
| Mecp2 null mice with specific reintroduction of Mecp2 into GABAergic neurons. | Male | ↔ | ↔ | – | – | |
| ref. Vigli et al., 2021 | Mecp2 null mice with intraperitoneal injection of saline. | Male | – | ↑ | – | – |
| Mecp2 null mice with intraperitoneal injection of cannabidiolic acid (CBDA) | Male | – | ↑ | – | – | |
| ref. Wither et al., 2013 | Mecp2+/− mice (since Mecp2+/− mice is an X-linked gene, X inactivation means that there can be differing levels of MECP2 in particular brain regions). For this study there was no control. The degree of anxiety behaviour was correlated with MeCP2 expression in various brain regions – results are expressed as anxiety level with lower MeCP2 expression compared to higher MeCP2 expression (akin to wildtype) in each brain region. | |||||
| Hippocampus | Female | – | ↓ | ↓ | – | |
| Cortex | Female | – | ↔ | ↔ | – | |
| Cerebellum | Female | – | ↔ | ↔ | – | |
| Spinal cord | Female | – | ↔ | ↔ | – | |
| ref. Zhang et al., 2014 | Mecp2 null mice | Male | – | ↑ | – | – |
| Mecp2 null mice exposed to deep-brain magnetic stimulation for 5 months. | Male | – | ↔ | – | – | |
| ref. Zhang et al., 2016 | Specific deletion of Mecp2 in neurons positive for choline acetyltransferase (ChAT, required for synthesis of acetylcholine). | Male | ↓/↔ | ↓∗ comparison made with control for the genetic carrier system, not wildtype | ↔∗ ↓ compared to control for the genetic carrier system | ↓∗ comparison made with control for the genetic carrier system, not wildtype |
| Specific deletion of Mecp2 in neurons positive for ChAT, but with re-expression of Mecp2 in the cholinergic neurons of the basal forebrain. | Male | ↔∗ comparison made with control for the genetic carrier system, not wildtype | – | – | – | |
| Specific deletion of Mecp2 in neurons positive for ChAT, but with re-expression of Mecp2 in the cholinergic neurons of the caudate/putamen | Male | ↓∗ comparison made with control for the genetic carrier system, not wildtype | – | – | – | |
| Selective activation of α7 nicotinic acetylcholine receptors in the hippocampus in the CA1 region of the hippocampus in mice with specific deletion of Mecp2 in neurons positive for ChAT. | Male | ↓ compared to control for Cre genetic carrier system, but↑ compared to mice with specific deletion of MECP2 in neurons positive for ChAT (without selective activation of α7 nicotinic acetylcholine receptors) | – | – | ↔ | |
| ref. Zhou et al., 2017 | Mecp2 null mice. | Male | ↓ | – | ↑ | – |
| Mecp2 null mice with selective reintroduction of Mecp2 into cholinergic neurons. | Male | ↓ | – | ↔ | – | |
Table 2: Analysis of study validity.
| Study | Is there evidence of a power calculation? | Can the number of mice used in experiments be clearly identified? | Is age of animals stated? | Was randomisation performed? | Evidence blinding was used? | What was the source of the animals? (details of genetic models found within appendix) | Model of MECP2 mutant mice | Mouse strain used | How were data from behavioural experiments captured? |
|---|---|---|---|---|---|---|---|---|---|
| ref. Abellán-Álvaro et al., 2021 | No | Yes | Yes | No | No | Wildtype and Mecp2+/− mice obtained from The Jackson laboratory and crossed in house. | Mecp2tm1.1Bird/J | Mecp2+/− mice were 129P2(C) and C57BL6 mixed background. Wildtype mice were C57BL/6J background. | Video tracking |
| Adachi et al., 2009 | No | Yes | Yes | No | No | Not clear. | Mecp2tm1.1Jae | All mice were 129/BALB/c background that was backcrossed with C57BL/6 for 10 generations. | Video tracking |
| ref. Bittolo et al., 2016 | No | Yes | Yes | No | No | Mecp2 null mice were obtained from The Jackson Laboratory and backcrossed on mice from Charles River. The origin of wildtype mice was not clearly stated. | Mecp2tm1.1Bird | Mecp2 null mice were C57BL/6 and 129P2(C) mixed background and backcrossed on C57BL/6. Wildtype were C57BL/6J background. | Video tracking |
| ref. Castro et al., 2014 | No | Yes | Yes | No | No | Genetic cross of mouse lines from The Jackson Laboratory. | Mecp2tm1.1Bird | All mice were C57BL/6 and 129P2 mixed background, that was backcrossed with C57BL/6J. | Video tracking |
| ref. Chao et al., 2010 | No | Yes | Yes | No | No | Mice heterozygous for Mecp2 mutation were obtained from Dr Adrian Bird. The source of other animals is not clearly stated. | Mecp2tm1Bird | The following strains appear to have been used (methods difficult to interpret due to multiple cross references): Viaat-Cre were FVB background, Viaat- Mecp2-/y mice and wildtype controls were mixed FVB, C57BL/6 and S6SvEvTac strains, and flox ‘controls’ were S6SvEvTac background. | Hand-held computer |
| Chin et al., 2018 | No | Yes | Yes | No | No | Genetic cross of mouse lines from The Jackson Laboratory. The origin of wildtype mice was not clearly stated. | Mecp2tm1Bird | Mecp2flox+/y; CaMKIIα-Cre mice were C57BL/6 and 129P2 mixed background and backcrossed to C57BL/6. Background not clearly stated for wildtype mice. | Automated system |
| ref. De Filippis et al., 2013 | No | Yes | Yes | No | No | Mecp2 null mice were from The Jackson Laboratory. The origin of wildtype mice was not clearly stated. | Mecp2tm1Hzo | Mecp2 null mice were C57BL/6J and 129S mixed background and backcrossed to C57BL/6J for at least 12 generations. Background not clearly stated for wildtype mice. | Video tracking |
| ref. De Filippis et al., 2014 | No | Yes for behavioural experiments; not clear for immunohistochemistry or Western blot. | Yes | No | No | Mecp2 null mice were obtained from The Jackson Laboratory. The origin of wildtype mice was not clearly stated. | Mecp2tm1Hzo | Mecp2 null mice were C57BL/6J and 129S mixed background and backcrossed to C57BL/6J for at least 12 generations. Background not clearly stated for wildtype mice. | Video tracking |
| ref. Derecki et al., 2012 | No | Yes | Yes | No | No | Mecp2+/− mice were obtained from The Jackson Laboratory. The origin of wildtype mice was not clearly stated. | The models of Mecp2+/− used in irradiation experiments was not clearly stated. | Not clearly stated for Mecp2+/− or wildtype mice. | Video tracking |
| ref. Flores Gutiérrez et al., 2020 | Yes | Yes | Yes | Yes | Yes | Wildtype and Mecp2+/− mice obtained from The Jackson laboratory and crossed in house. | Mecp2tm1.1Bird/J | Mecp2+/− mice were 129P2(C) and C57BL6 mixed background. Wildtype mice were C57BL/6J background. | Video tracking |
| ref. Fyffe et al., 2008 | No | Yes | Yes | No | No | Gifts from Adrian Bird, Brad Lowell and Joel Elmquist. | Presumed to be Mecp2tm1Bird | Mixed 129SvEv and FVB background for all mice. | Computer-operated digiscan optical animal activity system (open field test). Hand held computer (light-dark assay). |
| ref. Gambino et al., 2010 | No | Yes (number of cells stated for electrophysiology experiments) | Age range stated | No | No | Mecp2 null mice were obtained from The Jackson Laboratory. The origin of wildtype mice was not clearly stated. | Mecp2tm1Hzo | Mecp2 null mice were C57BL/6J and 129S mixed background and were backcrossed to C57BL/6J. Background not clearly stated for wildtype mice. | Not relevant |
| ref. Gemelli et al., 2006 | No | Yes | Yes | No | Yes | Mecp2 mutation created in house | Presumed to be Mecp2tm1.1Jae | All mice were mixed 129, BALBC mixed background and backcrossed to C57BL/6. | Video tracking |
| ref. Goffin et al., 2014 | No | Yes | Yes | Samples were pseudo-randomised | Yes | Genetic cross of mouse lines from The Jackson Laboratory | Presumed to be Mecp2tm1.1Jae | All mice were backcrossed on C57BL/6, but the original backgrounds were not clearly stated. | Video tracking (elevated zero maze) and beam breaks (open field test) |
| ref. Hao et al., 2015 | No | Yes | Yes | No | Yes | Not stated | Not stated | Mecp2+/− mice were mixed FVB and 129 background. Background not clearly stated for wildtype mice. | Video tracking |
| ref. He et al., 2014 | No | yes | Yes | No | Yes | Mouse lines were obtained from The Jackson Laboratory. The origin of wildtype mice was not clearly stated. | Mecp2tm1bird | PV-MECP2-/y, Mecp2flox/y and PV-Cre mice were mixed 129P2 and C57BL/6J background and backcrossed to C57BL/6J. The background of wildtype mice was not clearly stated. | Video tracking (open field test and elevated plus maze). Method of data capture was not stated for the light-dark assay |
| ref. Ito-Ishida et al., 2015 | No | Yes | Yes | No | No | PV-Cre and SOM-Cre mice were obtained from The Jackson Laboratory. The origin of other mice was not clearly stated. | Mecp2tm1Bird | PV- Mecp2-/y mice and flox ‘controls’ were mixed 129S6SvEvTac and C57BL/6 background. PV-Cre mice were C57BL/6 background. Som-Cre were mixed 129S4Sv and C57BL/6 background. SOM- Mecp2-/y mice were mixed 129S4Sv and C57BL/6 and 129S6SvEV background. Background not clearly stated for wildtype mice. | Video tracking |
| ref. Kerr et al., 2010 | No | Yes for qRT-PRC; no for behavioural experiments | Yes | No | No | Mecp2 null mice were obtained from Adrian Bird laboratory, University of Edinburgh and The Jackson Laboratory. The origin of wildtype mice was not clearly stated. | Mecp2tm1.1Bird | Mecp2 null mice were either 129S1/SvImJ background or mixed 129/SvJ and C57BL/6J background. Background not clearly stated for wildtype mice. | Human observer |
| Kondo 2016 | No | Yes | Yes | No | No | Not clearly stated | Mecp2tm1Tam | All mice were 129sv and C57BL/6 mixed background. | Video tracking |
| ref. Lang et al., 2012 | No | Yes | Yes | No | No | TH-cre mice were a gift from Dr Joseph Savitt. Mecp2 null mice were from The Jackson Laboratory and genetic crosses were performed in house. The origin of wildtype mice was not clearly stated. | Mecp2tm2Bird | Mecp2tm2Bird is originally mixed 129P2 and C57BL/6J background and these mice were backcrossed on C57BL/6J. The original background of TH-cre is unknown; this line was backcrossed on C57BL/6. Background not clearly stated for wildtype mice. | Video tracking |
| ref. Lioy et al., 2011 | No | Yes | Yes | No | No | Mecp2tm2Bird were from the Jackson laboratory. The origin of hGFAPcreT2 mice was unclear. | Mecp2tm2Bird | hGFAPcreT2 mice were on a mixed FVB/N and C57BL/6 background and backcrossed on C57BL/6 for at least 8 generations. Mecp2Stop/y mice were mixed 129P2 and C57BL/6 background and backcrossed on C57BL/6J. | Video tracking |
| ref. Mantis et al., 2009 | No | Yes | Yes | No | No | Mecp2 null mice were originally from The Jackson Laboratory and then maintained in-house. The origin of wildtype mice was not clearly stated. | Mecp2tm1Hzo | Mecp2 null mice were C576L/6J and 129S mixed background and backcrossed to C57BL/6J for at least 12 generations. Background not clearly stated for wildtype mice. | Automated system (open field test). Unclear method of data capture for light-dark assay. |
| ref. McGill et al., 2006 | No | Yes | Yes for ‘anxiety’ behavioural tests, but no for other tests/analyses | No | No | Mecp2 null mice were presumably created in-house, since author Huda Zoghbi donated the mutation to The Jackson Laboratory. The origin of wildtype mice was not clearly stated | Mecp2tm1Hzo | Mecp2 null mice were C576L/6J and 129SvEv mixed background and backcrossed to C57BL/6J for at least 12 generations. Background not clearly stated for wildtype mice. | Digiscan optical animal activity system (open field).Human observer (elevated plus maze and light-dark assay). |
| ref. Mellios et al., 2014 | No | Yes | Age range stated | No | No | Not stated | Mecp2tm1Bird | All mice were mixed 129P2 and C57BL/6 background and backcrossed on C57BL/6. | Data capture method was not stated. |
| ref. Meng et al., 2016 | No | Yes | Yes | No | Yes for behavioural experiments; not clearly stated for electrophysiology experiments. | Vglut2-Cre+/− knock-in line was a gift from Dr. Brad Lowell; references were given regarding the other mouse lines, but their origin was not clearly stated. | Mecp2tm1.1Bird and Mecp2tm2Bird | Mecp2LSL/y and Mecp2+/LSL mice were mixed C57BL6 and 129P2, backcrossed on 129S6SvEv. Vglut2-Cre+/− mice were >99% FVB strain. Mecp2flox+/y was mixed C57BL6 and 129P2 background. The remaining mice were created from crosses of the above. Background not clearly stated for wildtype mice. | Automated system (open field test and light-dark assay). Video tracking (elevated plus maze) |
| ref. Mossner et al., 2020 | No | Yes | Yes | No | Yes | All mouse lines were obtained from The Jackson Laboratory and crosses were performed in house. | Mecp2tm1Bird/J | Mecp2flox+/y;Dlx5/6-Cre mice were mixed CD1, C57BL/6 and 129P2 background.SOM-Mecp2-/y mice were mixed 129S4Sv and C57BL/6 background.PV-Mecp2-/y mice mixed C57BL/6 and 129P2 backgroundVip-Mecp2-/y mice were mixed 129S4Sv and C57BL/6 background.Flox mice (used as ‘controls’) were mixed C57BL/6 and 129P2 background. | Video tracking |
| ref. Nuber et al., 2005 | No | Yes | No; authors chose to classify according to symptoms rather than age. | No | No | Not clear from methods, although the Mecp2 null mice are presumed to have been created in house since the authorship includes Adrian Bird. | Presumed to be Mecp2tm1Bird | Mecp2 null mice presumed to be mixed C57BL6 and 129P2 background. Background not clearly stated for wildtype mice. | N/A |
| ref. Orefice et al., 2016 | No | Yes for behavioural experiments; no for immunohistochemistry or electrophysiology | Yes | No | Yes | Mecp2-/y and Mecp2R306C mice were obtained from Michael Greenberg, Harvard Medical School (also obtainable through The Jackson Laboratory). Mecp2f/y, Mecp2Stop and Emx1Cre mice were obtained from The Jackson laboratory. Cdx2Cre mice were obtained from Eric Fearon. AdvillinCre mice were obtained from Fan Wang, Duke University. AdvillinCreERT2 mice were obtained from John Wood, University College London. | Mecp2tm1.1Bird, Mecp2tm2Bird and Mecp2R306 | Male and female mice were all of mixed C57BL/6J and 129/SvEv mixed backgrounds, except for Mecp2 null and Mecp2R306 mice, which were on a C57BL/6J background. | Video tracking. |
| ref. Ren et al., 2012 | No | Yes | No | No | No | All mice were obtained from the Mouse Regional Resource Centre. | Mecp2tm1.1Jae | All mice were 129 and C57BL/6 mixed background. | N/A |
| ref. Ricceri et al., 2011 | No | Yes | Yes | No | Yes | Mecp2 null mice were from The Jackson Laboratory. The origin of wildtype mice was not clearly stated. | Mecp2tm1Hzo | Mecp2 null mice were of mixed C57BL/6J and 129SvEv background and backcrossed to C57BL/6J for at least 12 generations. Background not clearly stated for wildtype mice. | Video tracking. |
| ref. Samaco et al., 2009 | No | Yes | Age range stated | No | No | PET1-Cre mouse line was provided by Evan Deneris. The origin of other animals was not clearly stated, although author Huda Zoghbi is known to have created the Mecp2tm1Hzo model, therefore Mecp2 null mice have likely been produced in-house. | Not stated | Mecp2-/y and their wildtype control – genetic background not clearly stated.TH-Cre animals were FVB/N background. PET1-Cre animals were C57BL/6 background. Mecp2flox/+ females of 129S6/SvEv background were bred with the Cre animals. TH-Cre; Mecp2flox/y mice were therefore mixed FVB/N and 129S6/SvEv background and PET1-Cre; Mecp2flox/y mice were mixed C57BL/6 and 129S6/SvEv background. The background of Mecp2flox/y and wildtype controls was not clearly stated. | Digiscan optical animal activity system (open field test). Handheld computer (light-dark assay) |
| ref. Su et al., 2015 | No | Yes | Yes | No | No | Dlx5/6-Cre mice and Mecp2Stop/+ mice were from The Jackson Laboratory. Mecp2flox/+mice were from the Mutant Mouse Regional Resource Centre at UC Davis. All mice were backcrossed on C57BL/6 mice from the National Laboratory Animal Centre, Taiwan. The origin of wildtype mice was not clearly stated | Mecp2tm1.1Jae and Mecp2tm2Bird | Dlx5/6-Cre mice were CD1 strain.Mecp2flox/+ mice were 129S4/SvJae, BALB/c and C57BL/6 mixed background. Mecp2Stop/y mice were mixed C57BL/6 and 129P2 background. All mice were backcrossed to C57BL/6 for at least 10 generations. Background not clearly stated for wildtype mice. | Video capture. |
| ref. Ure et al., 2016 | No | Yes | No | No | Yes | Source not clearly stated. | Mecp2tm2Bird | Mecp2lox−Stop/y mice were 129S6SvEvTac background, Viaat-Cre mice were FVB background and Viaat-Cre; Mecp2lox−Stop/y mice were mixed 129S6SvEvTac and FVB background. Background not clearly stated for wildtype mice. | Automated system (open field test and light-dark assay). Video tracking (elevated plus maze). |
| ref. Vigli et al., 2021 | No | Yes | Yes | No | Yes | Mecp2 mutant and wildtype mice were obtained from The Jackson Laboratory. | Mecp2tm1Hzo | All mice were mixed C57BL/6 and 129S background. | Video tracking |
| ref. Wither et al., 2013 | No | Yes | Age range stated | No | No | The Jackson laboratory. | Mecp2tm1.1Bird and Mecp2tm2Bird | Mixed C57BL/6J and 129P2 background, backcrossed to C57BL/6. | Automated system (open field test). Human tracking (light-dark assay). |
| ref. Zhang et at 2014 | No | Yes | Yes | No | No | Mecp2 null mice were obtained from The Jackson Laboratory and wildtype mice appear to have been obtained from SLAC laboratory Animal. | Mecp2tm1Hzo | Mecp2 null mice were C57BL/6 and 129S mixed background, backcrossed to C57BL/6J for at least 12 generations. Wildtype controls appear to have been C57BL/6, although this is not explicitly stated. | Video tracking. |
| ref. Zhang et al., 2016 | No | Yes | The age-ranges were clearly stated, except for the age of animals where Western blot was performed. | No | Yes | Mice were obtained from The Jackson Laboratory and crosses performed in house. The origin of wildtype mice was not clearly stated. | Mecp2tm1Bird | Mecp2flox/- mice were mixed C57BL/6J and 129P2 mixed background. Chat-IRES Cre mice were mixed C57BL/6J and 129S6 background. The paper notes that all mice were C57BL/6J strain and therefore the above mice were presumably backcrossed to C57BL/6J. The strain of wildtype mice was not explicitly stated. | The behaviour in the elevated zero maze was videotaped and the data capture method for the light-dark assay, open field test and elevated plus maze was not stated. |
| ref. Zhou et al., 2017 | No | Yes | Yes | No | Yes | All mice were obtained from The Jackson Laboratory and crosses performed in house. The wildtype mice were created as part of the genetic cross. | Mecp2tm2Bird | Mecp2Stop/y mice were mixed C57BL/6J and 129P2 mixed background. Chat-IRES Cre mice were mixed C57BL/6J and 129S6 background. Both strains were backcrossed to C57BL/6J. | Automated system (open field test).Video tracking (elevated plus maze). |
Box 2
Description of unconditioned tests of anxiety
These four tests exploit mice’s innate avoidance of heights, light and open spaces, which conflicts with their spontaneous exploration of novel surroundings.
Open field – uses a chamber that is novel to the mouse. It has lines to demarcate the floor space. This enables activity to be quantified. A mouse is placed in the centre of the chamber and allowed to explore freely. Each line crossing is counted as a unit of activity. The amount of time spent in the centre versus periphery is scored. Greater activity and greater time spent in the centre both indicate lesser anxiety. Defecation, rearing and grooming behaviour can also be used as measures of anxiety (the more frequent these behaviours, the greater the level of anxiety). Total distance travelled/total activity was not included as an anxiety measure in this review since Rett syndrome affects motor activity, providing a confounding reason for altered total activity.
Light-dark assay – employs an environment that is novel to the mouse. It contains a larger light and smaller dark compartment. A mouse is placed in the light compartment and allowed to explore. The number of transitions and time spent in each compartment are recorded. The greater the number of transitions and the greater the amount of time spent in the light, the less anxious the mouse. Risk assessments – when mice adopt an extended posture before crossing from dark to light compartments – may also be used as a measure of anxiety (increased risk assessment by the mouse indicate higher levels of anxiety).
Elevated Plus maze – this consists of four arms extending from a central platform to make a plus shape. Two arms are walled and two are open (bright and exposed area); the central platform is also open. The plus maze is elevated above the ground. The mouse is released onto the central platform and the amount of time spent in the open and closed arms is assessed; more time in the open arm indicates less anxiety. The number of risk assessments made before moving from closed to open arms can be quantified; more risk assessments indicate greater anxiety.
Elevated zero maze – a variation of the elevated plus maze without the central starting platform – this is thought to be an advantage as mice often stay here for some time and interpretation of this behaviour is difficult. The elevated zero maze is circular and consists of alternating open and closed arcs (ref. Bailey and Crawley, 2009).
Other tests were employed within the studies, for example the startle response, but these have been excluded from the description in the results as the four tests above are considered the most effective tests of anxiety, as opposed to fear or a measure of social behaviour (which is defined as a separate feature of Rett syndrome). These four tests are also the most frequently used anxiety measures within the studies and aided comparability between studies. A novelty suppressed feeding test was used in one paper (ref. Kondo et al., 2016), but excluded as it is considered a less effective test of anxiety.
N.B. There is debate regarding ‘anthropomorphisation’ of pre-clinical animal models of anxiety behaviour. We refer to ‘anxiety’-related behaviours in mice as ‘anxiety’ throughout the review but recognise the marked simplification of this approach.
Alt-text: Box 2
Circuitry regions and cellular subtypes where loss of MeCP2 altered anxiety behaviour
Macrocircuitry regions
Loss of MeCP2 in the peripheral sensory ganglia has been associated with increased anxiety behaviour. Mice with this mutation had enhanced sensitivity to tactile stimuli when compared to controls. It is important to note that mice would have a greater anxiety response to touch than humans. However, the authors comment that other studies have shown that humans who have had sensory deprivation in early life have increased anxiety (ref. Orefice et al., 2016). Deletion of Mecp2 in the amygdala was associated with an increase in anxiety behaviour in male mice (ref. Adachi et al., 2009; ref. Gemelli et al., 2006). There is evidence for deletion of Mecp2 in the nucleus accumbens and prefrontal cortex being associated with increased anxiety (Mecp2 was deleted from multiple anatomic regions, therefore the relevance of individual regions is not clear) (ref. Gemelli et al., 2006).
Cellular subtypes
Selective reintroduction of Mecp2 in astrocytes of Mecp2 null mice was associated with anxiety behaviour being more typical of behaviours of control mice (although interpretation is complex as the null mice showed mixed anxiety behavioural changes compared to control) (ref. Lioy et al., 2011).
Circuitry regions and cellular subtypes where loss of MeCP2 had mixed effects on anxiety behaviour
Macrocircuitry regions
Lower MeCP2 expression in the hippocampus in Mecp2+/− mice was associated with lesser anxiety behaviour. However, expression of MeCP2 was not measured in anxiety-associated brain regions other than the hippocampus, making interpretation of these results difficult (ref. Wither et al., 2013). In contrast when Mecp2 was removed from the forebrain (inclusive of hippocampus), there was an increase in anxiety behaviour (ref. Gemelli et al., 2006). Sim-1 is a gene regulatory element required for the development of neurons in the paraventricular, supraoptic, and posterior hypothalamic nuclei, and in the nucleus of the lateral olfactory tract of the amygdala. Deletion of Mecp2 in Sim1-expressing neurons led to a decrease in MeCP2 expression in these regions. This did not result in a reliable difference in ‘anxiety’ behaviour compared to controls (ref. Fyffe et al., 2008).
Cellular subtypes
Altered MeCP2 function in cholinergic (ref. Zhou et al., 2017; ref. Zhang et al., 2016), glutamatergic (ref. Meng et al., 2016; ref. Goffin et al., 2014) and GABAergic (ref. Chao et al., 2010; ref. Ure et al., 2016) neurons resulted in a variable pattern of changes in anxiety behaviour. Mice with specific removal of Mecp2 from tyrosine hydroxylase-expressing dopaminergic (DA)/noradrenergic (NA) neurons showed no difference in anxiety behaviour compared to controls (ref. Samaco et al., 2009). Rescue of Mecp2 in catecholaminergic neurons normalised anxiety behaviour in male and female Mecp2-deficient mice (ref. Lang et al., 2012). However, there was no significant effect on anxiety behaviour when Mecp2 was rescued in the dorsal striatum of Mecp2 deficient male and female mice (ref. Su et al., 2015). Overall, this implies that if MeCP2 function in DA/NA neurons does affect anxiety behaviour, that this occurs via a specific subtype of catecholaminergic neuron and/or particular brain region.
In adult mice, microglial cells develop within the bone marrow and migrate to the brain (in addition to slow proliferation of microglia within the CNS). Mecp2+/− mice were irradiated to kill resident microglia and then given a bone marrow transplant from wildtype mice. Behavioural testing indicated that transplantation (intended to give rise to normal microglia) resulted in anxiety behaviour reducing to wildtype levels (ref. Derecki et al., 2012). A later study used Mecp2 null mice and replicated these methods. Anxiety behaviour was not explored, but other symptoms were not reversed through introduction of wildtype microglia, suggesting that anxiety behaviour may also not have been altered (ref. Wang et al., 2015).
Circuitry regions and cellular subtypes where loss of MeCP2 did not alter anxiety behaviour
Cellular subtypes
Specific loss of MeCP2 in GABAergic interneurons – parvalbumin, somatostatin, vasoactive intestinal peptide (separate or combined) – was not associated with a change in anxiety behaviour (ref. Ito-Ishida et al., 2015; ref. He et al., 2014; ref. Mossner et al., 2020).
Mice in which Mecp2 was deleted in PET1-expressing serotonergic neurons showed no difference in anxiety behaviour compared to controls. (ref. Samaco et al., 2009). Mecp2 null mice were found to have lower 5-HT7R (serotonin receptor) density in the cortex and hippocampus compared to wildtype. Intraperitoneal injection of LP-211 (a 5-HT7R selective agonist) in Mecp2 null mice was associated with increased cortical 5-HT7R density, but had no clear effect on anxiety behaviour (ref. De Filippis et al., 2014). Serotonin is a key neurotransmitter in anxiety disorders, so this apparent lack of effect on anxiety behaviour is of note. It has been shown that MeCP2 is involved in enhancing transcription of 5-HT1AR (ref. Philippe et al., 2018) and so it is possible that either MeCP2 does not modulate 5-HT7 production or that a greater concentration of 5-HT has less impact if there is lower receptor expression. In addition, studies have shown that administration of fluoxetine (a selective serotonin reuptake inhibitor, SSRI) resulted in raised MeCP2 levels in GABAergic neurons (ref. Cassel et al., 2006) and increased MeCP2 expression in the prefrontal cortex but not the CA3 region of the hippocampus (ref. Villani et al., 2021), highlighting that MeCP2 function within serotonergic neurons may be different to the effects of serotonin itself.
Cannabidiolic acid administration did not reduce anxiety in Mecp2 null mice (ref. Vigli et al., 2021). This contrasts with an earlier study that reported cannabidivardin improved symptoms (anxiety behaviour not assessed) in Mecp2 null mice (ref. Vigli et al., 2018), so it is possible that cannabinoid receptors may be relevant to anxiety in Rett syndrome.
Treatment studies that may provide further insights into these mixed effects on anxiety behaviour
Electrophysiological analysis of isolated cortical cell membranes of Mecp2 null mice showed reduced GABA and glutamate currents relative to those from Mecp2 null mice that had been administered mirtazapine prior to analysis. However, administration of mirtazapine and desipramine to Mecp2 null mice did not result in a clear change in anxiety behaviour (ref. Bittolo et al., 2016). Mecp2+/− mice initially showed a reduction in anxiety compared to wildtype and anxiety apparently increased with mirtazapine. However, this was subsequently attributed to increased whisker sensitivity altering the behaviour of the mice in the closed arms of the assay. Albeit whisker (sensory) sensitivity may be relevant to anxiety, in line with earlier findings (ref. Orefice et al., 2016)). There was a reduction in expression of parvalbumin positive neurons (fast-firing GABAergic interneurons) in the barrel cortex and basolateral amygdala in Mecp2+/− mice; expression was increased in the barrel cortex but not the amygdala following mirtazapine treatment, suggesting a possible mechanism for the increased sensitivity (ref. Flores Gutiérrez et al., 2020).
The mechanism of action of deep-brain magnetic stimulation (DMS) is largely unknown, though it may modulate the balance of excitatory and inhibitory circuits. Treatment with DMS for 5 months showed some reduction in anxiety behaviour of Mecp2 null mice to wildtype levels (ref. Zhang et al., 2014). Deep brain stimulation (DBS) also appeared to reduce anxiety behaviour in Mecp2+/− mice. However, the same effect was found with sham DBS, indicating handling and exposure as the reason for decreased anxiety (ref. Hao et al., 2015).
Molecular changes that potentially associate with the anxiety behavioural changes/physiologic response
MeCP2 effects on circuitry connections and molecular changes that are cell type and region specific
Loss of MeCP2 in the peripheral sensory ganglia may cause loss of the β3 unit of the GABAA receptor in these regions, resulting in increased peripheral nerve conduction, which may act as a mechanism for the increased anxiety behaviour (ref. Orefice et al., 2016). Excitatory projections from the cortex and thalamus to the lateral nucleus of the amygdala (LA) are involved in fear acquisition. MeCP2 specifically affected the functioning of the cortico-LA pathway (not the thalamo-LA pathway) through effects on stabilisation of AMPA receptors and synaptic elimination and maturation, highlighting a potential mechanism of anxiety in Rett syndrome, since there is an overlap between fear and anxiety circuitry (ref. Gambino et al., 2010). (ref. Gambino et al., 2010).
Loss of MeCP2 in cholinergic neurons in the basal forebrain led to decreased expression of choline acetyltransferase (ChAT). This was associated with reduced excitability of the cholinergic neurons, leading to downstream increased excitability of hippocampal pyramidal neurons. According to anxiety behavioural changes, it appears that these effects of MeCP2 were pathway-specific (ref. Zhang et al., 2016). By turn, choline supplementation significantly increased ChAT activity in Mecp2 null and wildtype mice, and reduced Mecp2 null anxiety levels to those of wildtype mice (ref. Ricceri et al., 2011). Choline supplementation in mice with specific loss of MeCP2 in excitatory neurons of the forebrain was associated with decreased anxiety and increased neurite length, number of dendritic branches and dendritic spine density, in the basal forebrain suggesting further molecular changes resulting from MeCP2 function in cholinergic neurons (ref. Chin et al., 2019).
Tyrosine hydroxylase (Th) and tryptophan hydroxylase 2 (Tph2) are rate-limiting enzymes in the production of DA/NA and 5-HT, respectively. MeCP2 may be necessary for their production as MeCP2 was found to bind to the Th and Tph2 promoters and there were reduced RNA levels for Th and Tph2 in Mecp2 null mice relative to wildtypes. Again this appears to be region-specific, since the expression of Th was lower in the locus coeruleus, midbrain containing substantia nigra and ventral tegmental area, but not the medulla, in Mecp2 null mice relative to wildtypes and Tph2 expression was lower in the hindbrain containing raphe nucleus B1-3, but not in the hindbrain region containing raphe nucleus B4-9, in Mecp2 null mice relative to wildtypes. These changes in enzyme expression were associated with reduced levels of monoamines (ref. Samaco et al., 2009).
Influence on cell signalling pathways
IGF-1 and BDNF both act via the phosphoinositide 3-kinase (PI3K)/Protein kinase B (Akt) pathway and the extracellular signal-regulated kinase (ERK) pathways. Lower serum IGF-1 (ref. Mellios et al., 2014) and lower hippocampal BDNF expression (ref. Kondo et al., 2016) were found in Mecp2+/− mice compared to wildtype mice. Increasing the serum IGF-1 levels (via clenbuterol treatment, a beta adrenergic agonist (ref. Mellios et al., 2014)) and environmental enrichment (ref. Kondo et al., 2016) resulted in anxiety behaviour returning to wildtype levels. Intraperitoneal rhIGF-1 treatment resulted in an increase of anxiety behaviour of MeCP2 null mice to wildtype levels (ref. Castro et al., 2014). Overall, these results indicate that modulation of IGF-1 and BDNF expression by MeCP2 may contribute to anxiety behaviour.
Possible changes to metabolic activity of CNS cells
Alterations in access to food in Mecp2 null mice suggested an improvement in anxiety. Mice fed a ketogenic diet or standard diet with caloric restriction had reduced anxiety compared to those given standard diet. However, the mice on restricted diets were more active as they persistently searched for food, wildtype mice on a similar restricted diet also showed a decrease in anxiety behaviour. This shows little evidence for the effects of cellular metabolic activity on anxiety behaviour or evidence that MeCP2 might affect metabolic activity (ref. Mantis et al., 2009).
Altered HPA axis function
mRNA for FKBP5 (a negative feedback regulator of the glucocorticoid receptor) and serum and glucocorticoid-inducible kinase (Sgk) levels were upregulated in whole-brain samples from Mecp2 null mice compared to wildtype, however there was no difference in levels in the basolateral amygdala, suggesting MeCP2’s modulation of their expression is region-specific (ref. Adachi et al., 2009; ref. Nuber et al., 2005). Mecp2 null mice in environmentally enriched housing showed increased mRNA expression of Sgk1in the hypothalamus and cortex that was associated with anxiety returning to wildtype levels, identifying a possible role for Sgk1 regulation in anxiety. (ref. Kerr et al., 2010).
MeCP2 regulation of corticotropin releasing hormone (CRH) expression differs between brain regions and between male and female mice: there was elevated CRH in the paraventricular nucleus of the hypothalamus, central amygdala and the bed nucleus of the stria terminalis (ref. McGill et al., 2006) of Mecp2 null compared to wildtype mice, whilst Mecp2+/− mice had lower expression of CRH mRNA in the hypothalamus than wildtype mice (ref. Kondo et al., 2016). CRH mRNA expression in the basolateral amygdala was not significantly different in mice without MeCP2 in the amygdala, compared to controls (ref. Adachi et al., 2009). The HPA axis was normal in Mecp2 null mice and so increased CRH expression (in certain regions) was identified as a key source of HPA axis dysfunction in Mecp2 null mice (ref. McGill et al., 2006). However, CRH1 antagonist administration significantly reduced anxiety behaviour of wildtype, but not Mecp2 null mice, whilst under restraint stress. Wildtype and Mecp2 null mice showed an irregular breathing pattern when restrained using a small restraint chamber, indicating a possible anxiety response. CRHR1 antagonist administration resulted in a closer to normal breathing pattern in the wildtype mice in the small chamber but notably affected breathing in Mecp2 null mice in both chambers (ref. Ren et al., 2012).
Physiological responses to stress, assessed through serum corticosterone levels were equivalent in Mecp2 null and wildtype mice at baseline and following 5–15 min of restraint stress (ref. McGill et al., 2006; ref. Nuber et al., 2005). However, this response altered after 30–60 min of restraint, at which point higher serum corticosterone was measured in Mecp2 null mice (ref. McGill et al., 2006; ref. Ren et al., 2012). Mecp2 null mice showed a greater increase in anxiety behaviour when moved from a large to a small chamber (restraint stress) compared to wildtype mice (ref. Ren et al., 2012). Environmental enrichment is associated with a reduction in corticosterone levels in Mecp2+/− mice and anxiety behaviour being similar to wildtype (ref. Kondo et al., 2016).
Environmental changes overlap with/modify the effects of genetic loss of MeCP2
Environmental enrichment normalised some anxiety behaviours in Mecp2 null mice. Behavioural testing was performed in two mouse strains: 129/SvJ and mixed 129/SvJ and C57BL/6J background. 129/SvJ mice are known to perform poorly in the elevated plus maze (ref. Kerr et al., 2010). Environmental enrichment normalised anxiety in Mecp2+/− mice (ref. Kondo et al., 2016). Environmentally enriched Mecp2 null mice had significantly lower mRNA expression for synaptophysin and post-synaptic density-95 protein in the hypothalamus, and lower mRNA expression for syntaxin 1A and synaptotagmin in the cortex, when compared to standard housed Mecp2 null mice (ref. Kerr et al., 2010).
Maternally-derived cortisol was shown not to affect anxiety behaviour of Mecp2 null and wildtype pups, or their response to restraint stress. Despite this, changes were found in mineralocorticoid receptor (MR) mRNA expression, which was increased in both genotypes in mice with mothers who drank cortisol solution; this effect was more marked in the Mecp2 null mice. Glucocorticoid receptor (GR) mRNA expression was increased in Mecp2 null mice with mothers who drank cortisol solution compared to Mecp2 null mice without cortisol supplementation. (ref. De Filippis et al., 2013).
Behavioural testing of Mecp2+/− and wildtype mice separated from their mothers (intended to induce early life stress) indicated that Mecp2+/− mice were less anxious and that maternal separation did not alter this. However, when exposed to a more anxiogenic paradigm – elevated plus maze under bright-light conditions – maternal separation resulted in both wildtype and Mecp2+/− mice being less anxious than their non-separated counterparts. Although maternal separation is usually used to induce early life stress, the authors noted that Mecp2+/− mothers (mothers of both wildtype and Mecp2+/− offspring in this study) tend to be neglectful and cannibalistic and C57BL/6 mice (the background in this study) also tend to be neglectful. Therefore, separation may have enabled the mice to become more resilient than the mice who stayed with the mothers. (ref. Abellán-Álvaro et al., 2021).
Discussion
Summary of findings
Many of the included studies illustrated effects on anxiety behaviour through use of mouse models with specific loss/reintroduction of Mecp2 from specific brain regions or cell subtypes. Loss/reintroduction of Mecp2 to some anxiety macrocircuit regions was associated with a change in anxiety behaviour, but for the majority of studies with loss/reintroduction of Mecp2 from particular cell subtypes, there were mixed effects on anxiety behaviour. Fig. 2, panel A summarises the brain regions identified as contributing/not contributing to anxiety in models of Rett syndrome and maps them onto the regions currently recognised to have a role in anxiety processing (ref. Calhoon and Tye, 2015). There is evidence (ref. Orefice et al., 2016) that the anxiety response could be modulated as early as the sensory detection stage. Fig. 2, panel B summarises the cell types identified as contributing/not contributing to anxiety in models of Rett syndrome. The mixed results found within some studies (ref. Zhou et al., 2017; ref. Zhang et al., 2016; ref. Chao et al., 2010; ref. Ure et al., 2016; ref. Samaco et al., 2009; ref. Lang et al., 2012; ref. Su et al., 2015) may reflect: variability in the validity of the anxiety behavioural tests (discussed in detail in section 4.2); the specificity of MeCP2 function within particular cell types; the fact that removal of Mecp2 from a cell subtype throughout the brain may mask the effects of that cell in a specific microcircuit; or may arise from the complexity of anxiety microcircuits (making a change to one neuronal or neurotransmitter subtype may not cause a definitive change to the overall circuit). If this last aspect were the case, there would be implications for anxiolytic drug treatments, which at present tend to target single or only few neurotransmitters. Inflammation is increasingly regarded as a contributor to mental disorders. Although the relevance of MeCP2 function in microglia is unclear (ref. Derecki et al., 2012; ref. Wang et al., 2015), there is evidence for an altered systemic inflammatory profile and for altered microglial activation (which can lead to increased release of proinflammatory molecules) in Rett syndrome (ref. Jin et al., 2017) suggesting possible routes for neuroinflammation contributing to anxiety symptoms.
Loss of MeCP2 in certain brain regions has been associated with reduced neurotransmitter (ref. Orefice et al., 2016; ref. Zhang et al., 2016; ref. Samaco et al., 2009; ref. Ricceri et al., 2011) and growth factor expression (ref. Kondo et al., 2016; ref. Mellios et al., 2014; ref. Castro et al., 2014), loss of neurotransmitter receptor subunits (ref. Orefice et al., 2016), reduced stabilisation of AMPA receptors (ref. Gambino et al., 2010), and alterations to synapses (ref. Gambino et al., 2010).
There is evidence for MeCP2 loss having an impact on regulation of the HPA axis, and mechanisms are beginning to be understood (ref. McGill et al., 2006; ref. Adachi et al., 2009; ref. Nuber et al., 2005; ref. Kerr et al., 2010; ref. Ren et al., 2012). There is evidence for increased FKBP5 mRNA expression in whole brains of null mice and CRH expression may be altered, although this appears to vary between brain regions and between male and female mice (ref. McGill et al., 2006; ref. Kondo et al., 2016; ref. Nuber et al., 2005). Mecp2 null mice show elevated serum corticosterone levels at a delayed timepoint following restraint stress (ref. McGill et al., 2006; ref. Ren et al., 2012).
Validity of the pre-clinical studies
Mecp2 mouse models have reasonable construct validity, as the majority of humans with Rett syndrome have MECP2 mutations. However, most mouse models are null for Mecp2; this is representative of large deletions in humans, which account for only 10% of cases. Although mouse models with conditional deletion of the Mecp2 gene are useful in understanding function of MeCP2 in individual cell subtypes, they do not recapitulate Rett syndrome, since they do not represent the global loss of MeCP2 and therefore have limited translational value. In models where there is conditional deletion of the Mecp2 gene in the targeted region, it is notable that they do not always result in complete loss of MeCP2 expression in that region (ref. Adachi et al., 2009; ref. Gemelli et al., 2006).
Only 11/38 of the studies included female mice (all found within Table 1), whereas Rett syndrome almost exclusively affects girls (ref. Katz et al., 2012). This is of particular significance in the study of anxiety disorders, since the prevalence of anxiety disorders in the general population is twice as high in women compared to men (ref. Bandelow and Michaelis, 2015) and sex-dependent effects on anxiety and stress-vulnerability have been found in Mecp2-deficient mice (ref. Philippe et al., 2018; ref. Cosentino et al., 2021).
As shown in Table 2, Mecp2 mouse models have differing genetic backgrounds and have been obtained from different sources. This makes comparison between studies and reproducibility more difficult. Although it has been shown that certain mouse models may show increased anxiety behaviour compared to others, there is no clear pattern amongst the studies reviewed in this paper (ref. Samaco et al., 2013). The effect of environmental enrichment on anxiety in mice with Mecp2 mutations emphasises that standardised animal husbandry practices are important in securing the reproducibility of results (ref. Katz et al., 2012; ref. Kondo et al., 2016; ref. Kerr et al., 2010) and the maternal separation study indicates that maternal temperament may affect anxiety (ref. Abellán-Álvaro et al., 2021).
There is debate as to the validity of mouse anxiety behavioural experiments. The behavioural experiments discussed in this paper best approximate to generalised anxiety disorder and specific phobias. These tests are imperfect, but there is no ‘gold standard’ anxiety behavioural experiment. Tests similar to the elevated plus maze and open field test have been performed in humans and behaviour within these tests is comparable between mouse and human. However, further work is needed to understand if the neurobiological anxiety mechanisms are comparable between mouse and human in these paradigms (ref. Bach, 2022). One key limitation of mouse behavioural experiments in the context of Rett syndrome mouse models is the motor impairment component of the phenotype. As anxiety behavioural experiments all rely on locomotion, this presents a clear a problem in interpretation. Measures such as ultrasonic vocalisation may be a useful alternative that does not rely on motor function (ref. Mun et al., 2015).
Although decreased time in the open arm and increased time in the closed arm of the elevated plus maze are typically interpreted as signs of increased anxiety in mice, it has been shown that female Mecp2tm1.1Bird mice avoid the closed arms due to whisker hypersensitivity, rather than true preference for the open arm (ref. Flores Gutiérrez et al., 2020). This potentially affects the interpretation of three of the papers involving female mice (ref. Kondo et al., 2016; ref. Meng et al., 2016; ref. Abellán-Álvaro et al., 2021). It would be important to clarify further whether this response is found across different Mecp2 mutations, mouse genders and with loss of MeCP2 from specific cells/anatomical regions. It also suggests that the open field test and light-dark assay may be preferable tests for quantifying anxiety behaviour in Rett syndrome mouse models, compared to the elevated plus maze or elevated zero maze (ref. Lioy et al., 2011; ref. Ure et al., 2016; ref. Flores Gutiérrez et al., 2020).
A diverse number of behavioural tests and variety of behavioural measures have been used in the studies within the review. For example, some studies measured latency to enter the open area of the elevated plus maze and others measured the time spent in the open area. This is a further factor that makes comparison between studies difficult. Physiological measures, such as heart rate may be considered as a more objective and translatable measure of anxiety response and it has been shown that physiological changes occur when mice are in the elevated plus maze (ref. Okonogi et al., 2018).
Anxiety mechanisms identified within human studies
Neurotransmitters and their metabolites
Prior to the identification of MECP2 mutation as the main cause of Rett syndrome, early research into the pathology of the syndrome identified that reduction of levels of biogenic amines and their metabolites was a common feature; however biogenic amines were not correlated with anxiety levels (ref. He et al., 2014; ref. Roux and Villard, 2010). Individuals with the pArg168X mutation (associated with more severe overall symptoms of Rett syndrome) had significantly lower cerebrospinal fluid (CSF) 5-hydroxy indole acetic acid (5-HIAA) levels than individuals with the pArg133Cys (associated with less severe global impairment) mutation or controls (ref. Samaco et al., 2009). RSBQ and ADAMS anxiety scores have significant (or trend towards) inverse correlation with level of global impairment (ref. Barnes et al., 2015; ref. Anderson et al., 2014; ref. Robertson et al., 2006). These findings suggest that lower CSF 5-HIAA levels are associated with lower anxiety levels in Rett syndrome (ref. Samaco et al., 2009; ref. Barnes et al., 2015). A study has not yet been done exploring the efficacy of SSRIs in individuals with Rett syndrome, however as part of the Rett Syndrome Natural History Study, parents have reported that they observed improvement in anxiety symptoms with SSRI treatment (ref. Buchanan et al., 2022).
HPA axis
Despite there being a number of pre-clinical studies suggesting a role for MeCP2 affecting HPA axis function, there have been few clinical studies exploring this. Total free urinary cortisol/24 h correlates well with free plasma cortisol levels (ref. El-Farhan et al., 2017). A study involving 10 females with Rett syndrome (stage III-IV) compared to age- and sex-matched controls showed no significant difference in total urinary cortisol/24 h (ref. Motil et al., 2006). In healthy individuals there is a 90% reduction in evening cortisol compared to morning concentration. An exploratory study of 30 individuals with Rett syndrome identified that in individuals with mutations leading to less severe impairment, the diurnal variation was more pronounced (closer to normal) than individuals with mutations leading to more severe global impairment (ref. Byiers et al., 2020). Further study, including correlation of cortisol with RSBQ, will be needed to understand the impact of MeCP2 on the HPA axis.
Growth factors
Although preclinical studies suggest a role for IGF-1 in anxiety behaviour, clinical trials for IGF-1 have produced a complex picture, discussed by ref. Singh and Santosh (2018). IGF-1 treatment is due to be studied further in a Phase 3 trial (ref. Neul et al., 2022). A post-mortem histology study suggested that BDNF expression is region-specific and that levels were higher in the cerebellum in Rett syndrome patients (ref. Pejhan et al., 2020a) although a study by the same group using ELISA (quantifying protein levels) suggested BDNF levels were lower in all regions (ref. Pejhan et al., 2020b); more detailed study of BDNF and IGF-1 expression in anxiety regions may be helpful for understanding its mechanism in Rett syndrome.
Gut-brain axis
Gastrointestinal symptoms are a common feature of Rett syndrome. Differences in the gut microbiome have been reported between Rett syndrome patients, but these did not correlate with differences in anxiety symptoms (as measured by the Gastrointestinal Health Questionnaire, validated tool for use in Rett syndrome patients), however it is possible that confounding factors affect interpretation of this correlation (ref. Thapa et al., 2021).
Sex-hormone influence
Fifteen out of twenty-one young women with Rett syndrome reported that premenstrual syndrome was a problem, and eight women described anxiety as being one of the premenstrual symptoms, suggesting their mood was modulated by hormonal changes (ref. Hamilton et al., 2012).
Potential environmental influence
Hospital Anxiety and Depression Scales (HADS) (ref. Zigmond and Snaith, 1983) were completed by mothers of individuals with Rett syndrome who were found to have greater anxiety, but not depression, than a British normative sample. Siblings of those with Rett syndrome had similar emotion regulation (according to the Strengths and Difficulties Questionnaire) (ref. Goodman, 1997) to the normal population. An increased severity of the RSBQ score (in the child with Rett syndrome) was correlated with a higher level of maternal stress and anxiety (ref. Cianfaglione et al., 2015). Unlike in preclinical studies, environmental enrichment did not alter anxiety symptoms (ref. Downs et al., 2018).
Summary
When combined with findings from a previous review of autonomic dysregulation in Rett syndrome (ref. Singh and Santosh, 2018), the findings described within the current review indicate that there are multiple potential mechanisms contributing to anxiety in Rett syndrome. As identified from the preclinical studies, further study is needed to understand the precise mechanisms of serotonin in contributing to anxiety in Rett syndrome. It appears that certain MECP2 mutations are associated with greater anxiety symptoms.
Epigenetics
Within rett syndrome
Although Rett syndrome is a genetic disorder, the mutation of MECP2 has epigenetic consequences, which are complex (ref. Marano et al., 2021). This MeCP2 dysfunction can interact with other epigenetic mechanisms; for example there may be differing levels of DNA methylation between individuals with Rett syndrome, adding another layer to the way MeCP2 can function (ref. Marano et al., 2021). Pre-clinical and clinical studies within this paper highlight potential environmental influences (with likely epigenetic effect) which may contribute to anxiety in Rett syndrome (ref. De Filippis et al., 2013; ref. Abellán-Álvaro et al., 2021; ref. Cianfaglione et al., 2015).
Rett syndrome may inform understanding of the epigenetics of anxiety
Epigenetic modulators, including MeCP2, are increasingly recognised as the mechanism by which development (and later life changes) can be affected by environment. Early life stress is a key environmental influence that predisposes individuals to mental health disorders, including anxiety disorders (ref. Gottschalk and Domschke, 2016; ref. Bearer and Mulligan, 2018; ref. Schiele et al., 2020). Preclinical studies have demonstrated different mechanisms linking reduced MeCP2 function with early life stress (ref. Murgatroyd et al., 2009; ref. Lewis et al., 2016) and these are depicted in Fig. 3. This mechanism is of interest since it suggests mechanistic overlap with Rett syndrome and that downstream cellular and molecular anxiety mechanisms of Rett syndrome could also be relevant to those with anxiety and a history of early life stress. Further, MECP2 mutation and epigenetic modification leading to altered MECP2 gene expression is reported in patients with autism spectrum condition (ASC) and schizophrenia (ref. Qiu, 2018; ref. Ciernia and LaSalle, 2016; ref. Richetto and Meyer, 2021). In a preclinical model, specific methylation of the Mecp2 promotor in the hippocampus was sufficient to reduce Mecp2 expression and induce most ASC-like behaviours (ref. Lu et al., 2020). ASC and schizophrenia have been associated with aberrant neuronal circuitry and anxiety (ref. Qiu, 2018).
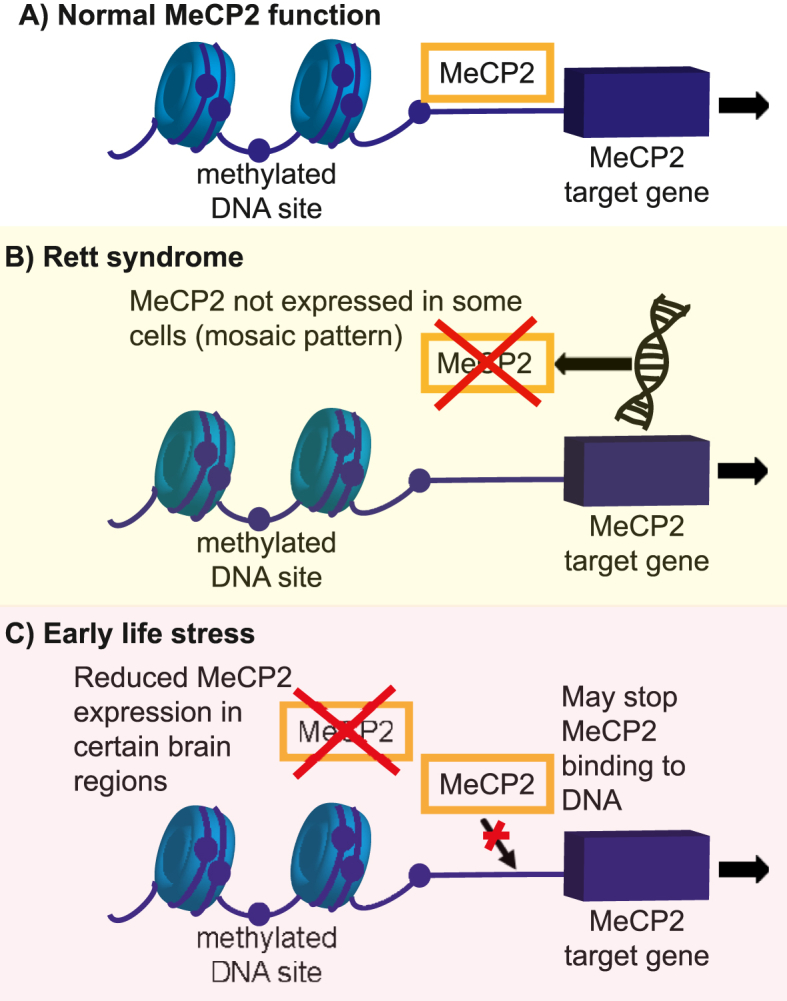
Epigenetic mechanisms of anxiety have previously been reviewed (ref. Gottschalk and Domschke, 2016) and the importance of developing biomarkers for the epigenetic effects of stress to enable individualised treatment has been emphasised (ref. Gottschalk et al., 2020). Peripheral biomarkers (often blood and saliva) can only provide limited understanding of epigenetic changes within the CNS. Rett syndrome models may provide a method to develop epigenetic biomarkers – such as blood, saliva, and possibly cerebrospinal fluid – that may correlate with CNS mechanisms and anxiety symptoms. This could be used to enhance understanding of anxiety in those exposed to early life stress or with ASC, in addition to guiding anxiety treatment in Rett syndrome (ref. Bearer and Mulligan, 2018).
Translation to therapeutic applications
Mecp2/MECP2 mutations result in comparable effects in mice and humans. This increases the likelihood that biological mechanisms are conserved between species and that these may have value as translational models for developing targeted drug treatments.
At present drug treatments for anxiety disorders are largely limited to SSRIs and serotonin and noradrenaline reuptake inhibitors (SNRIs). Understanding the molecular and cell- and region-specific effects of MeCP2 loss will be important in developing effective treatments for anxiety in Rett syndrome. The mechanisms identified in this review highlight the potential breadth of targets, for example the sensory system, a wider range of neurotransmitters and regulation of the HPA axis. The current range of investigations do not allow us to identify reliable potential treatment targets, but potentially compounds targeting astrocytic functions may be relevant (ref. Lioy et al., 2011). Results from pre-clinical and clinical studies seem to suggest that only specific serotonergic agents may be effective (ref. Samaco et al., 2009; ref. De Filippis et al., 2014; ref. Barnes et al., 2015).
Given the potential overlapping mechanism of Rett syndrome and early life stress, treatments for anxiety in Rett syndrome may also be effective in those with anxiety and a history of early life stress.
Anxiety in patients with ASC is often difficult to treat but there have been few studies of the efficacy of pharmacotherapy. The current evidence from studies of children and adolescents is insufficient to support treatment with SSRIs (ref. Stepanova et al., 2017). This suggests the need for an alternative innovative approach to treating anxiety symptoms and disorders, possibly linked to MeCP2 and its isoforms (ref. Good et al., 2021).
As well as anxiety, disordered breathing is a common feature in Rett syndrome. Although anxiety can worsen respiratory function, it is possible that altered respiratory function can exacerbate anxiety (ref. Ren et al., 2012). Within the field of anxiety research, the 7.5% CO2 inhalation experimental medicine model is well established. In this model humans inhale 7.5% CO2 over 20 min, which induces subjective and autonomic responses and neurocognitive changes that are comparable to the features of generalised anxiety disorder. The mechanisms are only partially understood (ref. Baldwin et al., 2017). Given that carbon dioxide/acid sensing is involved in breathing and the CO2 anxiety model, this prompts consideration of an overlap between anxiety and breathing mechanisms in Rett syndrome, for example via similar cellular or molecular sensing mechanisms. Exploration of these factors may extend knowledge of contributing mechanisms in the CO2 model and so inform understanding of overall mechanisms of anxiety.
Conclusion and next steps
Rett syndrome is a complex disorder with multiple intertwining cellular and systems abnormalities. The combined findings of pre-clinical and clinical studies show that progress is being made towards improved understanding of anxiety mechanisms in Rett syndrome.
Future steps for pre-clinical work will need to include greater use of female mice within studies, clarification of whether whisker sensitivity (affecting interpretation of behaviour in the elevated plus maze) is found across Rett syndrome models, inclusion of tests that are not affected by motor impairment (such as ultrasonic vocalisation) and inclusion of physiological markers (corticosterone and heart rate) that are quantifiable and translatable. Standardisation of anxiety tests would enable improved comparison between studies. Given that environmental factors may modify anxiety, standard environment should be used as well as consideration of maternal temperament. In terms of added mechanistic understanding, it will be helpful to develop understanding about the effects of Mecp2 mutation on specific anxiety-circuitry connections.
Given the importance of anxiety as a symptom within Rett syndrome, clinical studies should increasingly aim to include anxiety measures, including RSBQ. The HPA axis in individuals with Rett syndrome is an area that also requires increased study.
Improved understanding of anxiety mechanisms in Rett syndrome (which may include development of biomarkers to identify the mechanisms present within an individual, given the heterogenous nature of the syndrome) could enable more effective treatments to be developed. Importantly, the known MECP2 mutation means that there is the potential for translation from mouse models to human therapeutic applications. Improved understanding of potential overlapping mechanisms between Rett syndrome, autism and early life stress may allow more effective anxiolytic treatments to be developed across these disorders.
Funding
Bethan Impey worked on this review whilst in position as an NIHR funded Academic Clinical Fellow.
Declaration of competing interest
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: BI has a small number of stocks and shares in GlaxoSmithKline. TAN has no conflicts of interest to declare. DSB is Medical Patron of Anxiety UK, and President-Elect of the British Association for Psychopharmacology. He receives an editor fee from Wiley, for his editorship of Human Psychopharmacology. He was an unpaid consultant for Idorsia in July 2020.
References
- M. Abellán-Álvaro, O. Stork, C. Agustín-Pavón, M. Santos. MeCP2 haplodeficiency and early-life stress interaction on anxiety-like behavior in adolescent female mice. J. Neurodev. Disord., 2021. [DOI | PubMed]
- M. Adachi, A.E. Autry, H.E. Covington, L.M. Monteggia. MeCP2-mediated transcription repression in the basolateral amygdala may underlie heightened anxiety in a mouse model of Rett syndrome. J. Neurosci., 2009. [DOI | PubMed]
- A. Anderson, K. Wong, P. Jacoby, J. Downs, H. Leonard. Twenty years of surveillance in Rett syndrome: what does this tell us?. Orphanet J. Rare Dis., 2014. [DOI | PubMed]
- D.R. Bach. Cross-species anxiety tests in psychiatry: pitfalls and promises. Mol. Psychiatr., 2022
- K. Bailey, J. Crawley. 2009
- D.S. Baldwin, R. Hou, R. Gordon, N.T. Huneke, M. Garner. Pharmacotherapy in generalized anxiety disorder: novel experimental medicine models and emerging drug targets. CNS Drugs, 2017. [DOI | PubMed]
- B. Bandelow, S. Michaelis. Epidemiology of anxiety disorders in the 21st century. Dialogues Clin. Neurosci., 2015. [DOI | PubMed]
- K.V. Barnes, F.R. Coughlin, H.M. O’Leary, N. Bruck, G.A. Bazin, E.B. Beinecke. Anxiety-like behavior in Rett syndrome: characteristics and assessment by anxiety scales. J. Neurodev. Disord., 2015. [DOI | PubMed]
- E.L. Bearer, B.S. Mulligan. Epigenetic changes associated with early life experiences: saliva, a biospecimen for dna methylation signatures. Curr. Genom., 2018. [DOI]
- T. Bittolo, C.A. Raminelli, C. Deiana, G. Baj, V. Vaghi, S. Ferrazzo. Pharmacological treatment with mirtazapine rescues cortical atrophy and respiratory deficits in MeCP2 null mice. Sci. Rep., 2016. [DOI]
- C.B. Buchanan, J.L. Stallworth, A.E. Scott, D.G. Glaze, J.B. Lane, S.A. Skinner. Behavioral profiles in Rett syndrome: data from the natural history study. Brain Dev., 2019. [DOI | PubMed]
- C.B. Buchanan, J.L. Stallworth, A.E. Joy, R.E. Dixon, A.E. Scott, A.A. Beisang. Anxiety-like behavior and anxiolytic treatment in the Rett syndrome natural history study. J. Neurodev. Disord., 2022. [PubMed]
- B.J. Byiers, A. Payen, T. Feyma, A. Panoskaltsis-Mortari, M.J. Ehrhardt, F.J. Symons. Associations among diurnal salivary cortisol patterns, medication use, and behavioral phenotype features in a community sample of rett syndrome. Am. J. Intellect. Dev. Disabil., 2020. [DOI | PubMed]
- G.G. Calhoon, K.M. Tye. Resolving the neural circuits of anxiety. Nat. Neurosci., 2015. [DOI | PubMed]
- S. Cassel, D. Carouge, C. Gensburger, P. Anglard, C. Burgun, J.-B. Dietrich. Fluoxetine and cocaine induce the epigenetic factors MeCP2 and MBD1 in adult rat brain. Mol. Pharmacol., 2006. [DOI | PubMed]
- J. Castro, R.I. Garcia, S. Kwok, A. Banerjee, J. Petravicz, J. Woodson. Functional recovery with recombinant human IGF1 treatment in a mouse model of Rett Syndrome. Proc. Natl. Acad. Sci. USA, 2014. [DOI | PubMed]
- H.-T. Chao, H. Chen, R.C. Samaco, M. Xue, M. Chahrour, J. Yoo. Dysfunction in GABA signalling mediates autism-like stereotypies and Rett syndrome phenotypes. Nature, 2010. [DOI | PubMed]
- E.W. Chin, W.M. Lim, D. Ma, F.J. Rosales, E.L. Goh. Choline rescues behavioural deficits in a mouse model of Rett syndrome by modulating neuronal plasticity. Mol. Neurobiol., 2019. [DOI | PubMed]
- R. Cianfaglione, R.P. Hastings, D. Felce, A. Clarke, M.P. Kerr. Psychological well-being of mothers and siblings in families of girls and women with Rett syndrome. J. Autism Dev. Disord., 2015. [DOI | PubMed]
- A.V. Ciernia, J. LaSalle. The landscape of DNA methylation amid a perfect storm of autism aetiologies. Nat. Rev. Neurosci., 2016. [PubMed]
- L. Cosentino, F. Bellia, N. Pavoncello, D. Vigli, C. D’Addario, B. De Filippis. Methyl-CpG binding protein 2 dysfunction provides stress vulnerability with sex-and zygosity-dependent outcomes. Eur. J. Neurosci., 2021. [DOI]
- B. De Filippis, L. Ricceri, A. Fuso, G. Laviola. Neonatal exposure to low dose corticosterone persistently modulates hippocampal mineralocorticoid receptor expression and improves locomotor/exploratory behaviour in a mouse model of Rett syndrome. Neuropharmacology, 2013. [DOI | PubMed]
- B. De Filippis, P. Nativio, A. Fabbri, L. Ricceri, W. Adriani, E. Lacivita. Pharmacological stimulation of the brain serotonin receptor 7 as a novel therapeutic approach for Rett syndrome. Neuropsychopharmacology, 2014. [DOI | PubMed]
- N.C. Derecki, J.C. Cronk, Z. Lu, E. Xu, S.B. Abbott, P.G. Guyenet. Wild-type microglia arrest pathology in a mouse model of Rett syndrome. Nature, 2012. [DOI | PubMed]
- J. Downs, J. Rodger, C. Li, X. Tan, N. Hu, K. Wong. Environmental enrichment intervention for Rett syndrome: an individually randomised stepped wedge trial. Orphanet J. Rare Dis., 2018. [DOI | PubMed]
- N. El-Farhan, D.A. Rees, C. Evans. Measuring cortisol in serum, urine and saliva–are our assays good enough?. Ann. Clin. Biochem., 2017. [DOI | PubMed]
- A.J. Esbensen, J. Rojahn, M.G. Aman, S. Ruedrich. Reliability and validity of an assessment instrument for anxiety, depression, and mood among individuals with mental retardation. J. Autism Dev. Disord., 2003. [DOI | PubMed]
- J. Flores Gutiérrez, C. De Felice, G. Natali, S. Leoncini, C. Signorini, J. Hayek. Protective role of mirtazapine in adult female Mecp2+/− mice and patients with Rett syndrome. J. Neurodev. Disord., 2020. [PubMed]
- S.L. Fyffe, J.L. Neul, R.C. Samaco, H.-T. Chao, S. Ben-Shachar, P. Moretti. Deletion of Mecp2 in Sim1-expressing neurons reveals a critical role for MeCP2 in feeding behavior, aggression, and the response to stress. Neuron, 2008. [DOI | PubMed]
- F. Gambino, M. Khelfaoui, B. Poulain, T. Bienvenu, J. Chelly, Y. Humeau. Synaptic maturation at cortical projections to the lateral amygdala in a mouse model of Rett syndrome. PLoS One, 2010. [DOI]
- T. Gemelli, O. Berton, E.D. Nelson, L.I. Perrotti, R. Jaenisch, L.M. Monteggia. Postnatal loss of methyl-CpG binding protein 2 in the forebrain is sufficient to mediate behavioral aspects of Rett syndrome in mice. Biol. Psychiatr., 2006. [DOI]
- D. Goffin, E.S. Brodkin, J.A. Blendy, S.J. Siegel, Z. Zhou. Cellular origins of auditory event-related potential deficits in Rett syndrome. Nat. Neurosci., 2014. [DOI | PubMed]
- W.A. Gold, R. Krishnarajy, C. Ellaway, J. Christodoulou. Rett syndrome: a genetic update and clinical review focusing on comorbidities. ACS Chem. Neurosci., 2017. [DOI]
- K.V. Good, J.B. Vincent, J. Ausió. MeCP2: the genetic driver of Rett syndrome epigenetics. Front. Genet., 2021. [DOI]
- R. Goodman. The Strengths and Difficulties questionnaire: a research note. JCPP (J. Child Psychol. Psychiatry), 1997. [DOI | PubMed]
- M.G. Gottschalk, K. Domschke. Novel developments in genetic and epigenetic mechanisms of anxiety. Curr. Opin. Psychiatr., 2016. [DOI]
- M.G. Gottschalk, K. Domschke, M.A. Schiele. Epigenetics underlying susceptibility and resilience relating to daily life stress, work stress, and socioeconomic status. Front. Psychiatr., 2020. [DOI]
- A. Hamilton, M.P. Marshal, G.S. Sucato, P.J. Murray. Rett syndrome and menstruation. J. Pediatr. Adolesc. Gynecol., 2012. [DOI | PubMed]
- S. Hao, B. Tang, Z. Wu, K. Ure, Y. Sun, H. Tao. Forniceal deep brain stimulation rescues hippocampal memory in Rett syndrome mice. Nature, 2015. [DOI | PubMed]
- L-j He, N. Liu, T-l Cheng, X-j Chen, Y-d Li, Y-s Shu. Conditional deletion of Mecp2 in parvalbumin-expressing GABAergic cells results in the absence of critical period plasticity. Nat. Commun., 2014. [DOI]
- A. Ito-Ishida, K. Ure, H. Chen, J.W. Swann, H.Y. Zoghbi. Loss of MeCP2 in parvalbumin-and somatostatin-expressing neurons in mice leads to distinct Rett syndrome-like phenotypes. Neuron, 2015. [DOI | PubMed]
- X.-R. Jin, X.-S. Chen, L. Xiao. MeCP2 deficiency in neuroglia: new progress in the pathogenesis of Rett syndrome. Front. Mol. Neurosci., 2017. [DOI | PubMed]
- D.M. Katz, J.E. Berger-Sweeney, J.H. Eubanks, M.J. Justice, J.L. Neul, L. Pozzo-Miller. Preclinical research in Rett syndrome: setting the foundation for translational success. Dis Model Mech, 2012. [DOI | PubMed]
- B. Kerr, P.A. Silva, K. Walz, J.I. Young. Unconventional transcriptional response to environmental enrichment in a mouse model of Rett syndrome. PLoS One, 2010. [DOI]
- M.A. Kondo, L.J. Gray, G.J. Pelka, S.K. Leang, J. Christodoulou, P.P. Tam. Affective dysfunction in a mouse model of Rett syndrome: therapeutic effects of environmental stimulation and physical activity. Dev Neurobiol, 2016. [DOI | PubMed]
- M. Lang, R.G. Wither, J.M. Brotchie, C. Wu, L. Zhang, J.H. Eubanks. Selective preservation of MeCP2 in catecholaminergic cells is sufficient to improve the behavioral phenotype of male and female Mecp2-deficient mice. Hum. Mol. Genet., 2012. [DOI | PubMed]
- C.R. Lewis, R.M. Bastle, T.B. Manning, S.M. Himes, P. Fennig, P.R. Conrad. Interactions between early life stress, nucleus accumbens MeCP2 expression, and methamphetamine self-administration in male rats. Neuropsychopharmacology, 2016. [PubMed]
- D.T. Lioy, S.K. Garg, C.E. Monaghan, J. Raber, K.D. Foust, B.K. Kaspar. A role for glia in the progression of Rett’s syndrome. Nature, 2011. [DOI | PubMed]
- Z. Lu, Z. Liu, W. Mao, X. Wang, X. Zheng, S. Chen. Locus-specific DNA methylation of Mecp2 promoter leads to autism-like phenotypes in mice. Cell Death Dis., 2020. [PubMed]
- J.G. Mantis, C.L. Fritz, J. Marsh, S.C. Heinrichs, T.N. Seyfried. Improvement in motor and exploratory behavior in Rett syndrome mice with restricted ketogenic and standard diets. Epilepsy Behav., 2009. [DOI | PubMed]
- D. Marano, S. Fioriniello, M. D’Esposito, F. Della Ragione. Transcriptomic and epigenomic landscape in rett syndrome. Biomolecules, 2021. [DOI | PubMed]
- B.E. McGill, S.F. Bundle, M.B. Yaylaoglu, J.P. Carson, C. Thaller, H.Y. Zoghbi. Enhanced anxiety and stress-induced corticosterone release are associated with increased Crh expression in a mouse model of Rett syndrome. Proc. Natl. Acad. Sci. USA, 2006. [DOI | PubMed]
- N. Mellios, J. Woodson, R.I. Garcia, B. Crawford, J. Sharma, S.D. Sheridan. β2-Adrenergic receptor agonist ameliorates phenotypes and corrects microRNA-mediated IGF1 deficits in a mouse model of Rett syndrome. Proc. Natl. Acad. Sci. USA, 2014. [DOI | PubMed]
- X. Meng, W. Wang, H. Lu, L-j He, W. Chen, E.S. Chao. Manipulations of MeCP2 in glutamatergic neurons highlight their contributions to Rett and other neurological disorders. Elife, 2016. [DOI]
- D. Moher, A. Liberati, J. Tetzlaff, D.G. Altman, P. Group∗. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann. Intern. Med., 2009. [DOI | PubMed]
- J.M. Mossner, R. Batista-Brito, R. Pant, J.A. Cardin. Developmental loss of MeCP2 from VIP interneurons impairs cortical function and behavior. Elife, 2020. [DOI]
- K.J. Motil, R.J. Schultz, S. Abrams, K.J. Ellis, D.G. Glaze. Fractional calcium absorption is increased in girls with Rett syndrome. J. Pediatr. Gastroenterol. Nutr., 2006. [DOI | PubMed]
- R.H. Mount, T. Charman, R.P. Hastings, S. Reilly, H. Cass. The rett syndrome behaviour questionnaire (RSBQ): refining the behavioural phenotype of rett syndrome. JCPP (J. Child Psychol. Psychiatry), 2002. [DOI | PubMed]
- H. Mun, T. Lipina, J. Roder. Ultrasonic vocalizations in mice during exploratory behavior are context-dependent. 9: 316. Front. Behav. Neurosci., 2015. [DOI]
- C. Murgatroyd, A.V. Patchev, Y. Wu, V. Micale, Y. Bockmühl, D. Fischer. Dynamic DNA methylation programs persistent adverse effects of early-life stress. Nat. Neurosci., 2009. [DOI | PubMed]
- J.L. Neul, A.K. Percy, T.A. Benke, E.M. Berry-Kravis, D.G. Glaze, S.U. Peters. Design and outcome measures of LAVENDER, a phase 3 study of trofinetide for Rett syndrome. Contemp. Clin. Trials, 2022. [DOI | PubMed]
- U.A. Nuber, S. Kriaucionis, T.C. Roloff, J. Guy, J. Selfridge, C. Steinhoff. Up-regulation of glucocorticoid-regulated genes in a mouse model of Rett syndrome. Hum. Mol. Genet., 2005. [DOI | PubMed]
- T. Okonogi, R. Nakayama, T. Sasaki, Y. Ikegaya. Characterization of peripheral activity states and cortical local field potentials of mice in an elevated plus maze test. Front. Behav. Neurosci., 2018. [DOI | PubMed]
- L.L. Orefice, A.L. Zimmerman, A.M. Chirila, S.J. Sleboda, J.P. Head, D.D. Ginty. Peripheral mechanosensory neuron dysfunction underlies tactile and behavioral deficits in mouse models of ASDs. Cell, 2016. [DOI | PubMed]
- S. Pejhan, V.M. Siu, L. Ang, M. Del Bigio, M. Rastegar. Differential brain region-specific expression of MeCP2 and BDNF in Rett Syndrome patients: a distinct grey-white matter variation. Neuropathol. Appl. Neurobiol., 2020. [DOI | PubMed]
- S. Pejhan, M.R. Del Bigio, M. Rastegar. The MeCP2E1/E2-BDNF-miR132 homeostasis regulatory network is region-dependent in the human brain and is impaired in rett syndrome patients. Front. Cell Dev. Biol., 2020. [DOI | PubMed]
- T.J. Philippe, F. Vahid-Ansari, Z.R. Donaldson, B. Le François, A. Zahrai, V. Turcotte-Cardin. Loss of MeCP2 in adult 5-HT neurons induces 5-HT1A autoreceptors, with opposite sex-dependent anxiety and depression phenotypes. Sci. Rep., 2018. [DOI | PubMed]
- Z. Qiu. Deciphering MECP2-associated disorders: disrupted circuits and the hope for repair. Curr. Opin. Neurobiol., 2018. [DOI | PubMed]
- J. Ren, X. Ding, G.D. Funk, J.J. Greer. Anxiety-related mechanisms of respiratory dysfunction in a mouse model of Rett syndrome. J. Neurosci., 2012. [DOI | PubMed]
- M.C. Ribeiro, J.L. MacDonald. Sex differences in Mecp2-mutant Rett syndrome model mice and the impact of cellular mosaicism in phenotype development. Brain Res., 2020
- L. Ricceri, B. De Filippis, A. Fuso, G. Laviola. Cholinergic hypofunction in MeCP2-308 mice: beneficial neurobehavioural effects of neonatal choline supplementation. Behav. Brain Res., 2011. [DOI | PubMed]
- J. Richetto, U. Meyer. Epigenetic modifications in schizophrenia and related disorders: molecular scars of environmental exposures and source of phenotypic variability. Biol. Psychiatr., 2021
- L. Robertson, S.E. Hall, P. Jacoby, C. Ellaway, Nd Klerk, H. Leonard. The association between behavior and genotype in Rett syndrome using the Australian Rett Syndrome Database. Am J Med Genet B Neuropsychiatr Genet, 2006. [DOI]
- J.-C. Roux, L. Villard. Biogenic amines in Rett syndrome: the usual suspects. Behav. Genet., 2010. [DOI | PubMed]
- R.C. Samaco, C. Mandel-Brehm, H.-T. Chao, C.S. Ward, S.L. Fyffe-Maricich, J. Ren. Loss of MeCP2 in aminergic neurons causes cell-autonomous defects in neurotransmitter synthesis and specific behavioral abnormalities. Proc. Natl. Acad. Sci. USA, 2009. [DOI | PubMed]
- R.C. Samaco, C.M. McGraw, C.S. Ward, Y. Sun, J.L. Neul, H.Y. Zoghbi. Female Mecp2+/− mice display robust behavioral deficits on two different genetic backgrounds providing a framework for pre-clinical studies. Hum. Mol. Genet., 2013. [DOI | PubMed]
- M.A. Schiele, M.G. Gottschalk, K. Domschke. The applied implications of epigenetics in anxiety, affective and stress-related disorders-A review and synthesis on psychosocial stress, psychotherapy and prevention. Clin. Psychol. Rev., 2020
- M.D. Shahbazian, B. Antalffy, D.L. Armstrong, H.Y. Zoghbi. Insight into Rett syndrome: MeCP2 levels display tissue-and cell-specific differences and correlate with neuronal maturation. Hum. Mol. Genet., 2002. [DOI | PubMed]
- J. Singh, P. Santosh. Key issues in Rett syndrome: emotional, behavioural and autonomic dysregulation (EBAD)-a target for clinical trials. Orphanet J. Rare Dis., 2018. [DOI | PubMed]
- E. Stepanova, S. Dowling, M. Phelps, R.L. Findling. Pharmacotherapy of emotional and behavioral symptoms associated with autism spectrum disorder in children and adolescents. Dialogues Clin. Neurosci., 2017. [DOI | PubMed]
- S.-H. Su, F.-C. Kao, Y.-B. Huang, W. Liao. MeCP2 in the rostral striatum maintains local dopamine content critical for psychomotor control. J. Neurosci., 2015. [DOI | PubMed]
- S. Thapa, A. Venkatachalam, N. Khan, M. Naqvi, M. Balderas, J.K. Runge. Assessment of the gut bacterial microbiome and metabolome of girls and women with Rett Syndrome. PLoS One, 2021. [DOI]
- K. Ure, H. Lu, W. Wang, A. Ito-Ishida, Z. Wu, L-j He. Restoration of Mecp2 expression in GABAergic neurons is sufficient to rescue multiple disease features in a mouse model of Rett syndrome. Elife, 2016. [DOI]
- D. Vigli, L. Cosentino, C. Raggi, G. Laviola, M. Woolley-Roberts, B. De Filippis. Chronic treatment with the phytocannabinoid Cannabidivarin (CBDV) rescues behavioural alterations and brain atrophy in a mouse model of Rett syndrome. Neuropharmacology, 2018. [DOI | PubMed]
- D. Vigli, L. Cosentino, M. Pellas, B. De Filippis. Chronic treatment with cannabidiolic acid (CBDA) reduces thermal pain sensitivity in male mice and rescues the hyperalgesia in a mouse model of rett syndrome. Neuroscience, 2021. [DOI | PubMed]
- C. Villani, M. Carli, A.M. Castaldo, G. Sacchetti, R.W. Invernizzi. Fluoxetine increases brain MeCP2 immuno-positive cells in a female Mecp2 heterozygous mouse model of Rett syndrome through endogenous serotonin. Sci. Rep., 2021. [DOI | PubMed]
- J. Wang, J.E. Wegener, T.-W. Huang, S. Sripathy, H. De Jesus-Cortes, P. Xu. Wild-type microglia do not reverse pathology in mouse models of Rett syndrome. Nature, 2015. [DOI | PubMed]
- R.G. Wither, M. Lang, L. Zhang, J.H. Eubanks. Regional MeCP2 expression levels in the female MeCP2-deficient mouse brain correlate with specific behavioral impairments. Exp. Neurol., 2013. [DOI | PubMed]
- Y. Zhang, R.-R. Mao, Z.-F. Chen, M. Tian, D.-L. Tong, Z.-R. Gao. Deep-brain magnetic stimulation promotes adult hippocampal neurogenesis and alleviates stress-related behaviors in mouse models for neuropsychiatric disorders. Mol. Brain, 2014. [DOI | PubMed]
- Y. Zhang, S.-X. Cao, P. Sun, H.-Y. He, C.-H. Yang, X.-J. Chen. Loss of MeCP2 in cholinergic neurons causes part of RTT-like phenotypes via α7 receptor in hippocampus. Cell Res., 2016. [DOI | PubMed]
- H. Zhou, W. Wu, Y. Zhang, H. He, Z. Yuan, Z. Zhu. Selective preservation of cholinergic MeCP2 rescues specific Rett-syndrome-like phenotypes in MeCP2stop mice. Behav. Brain Res., 2017. [DOI | PubMed]
- A.S. Zigmond, R.P. Snaith. The hospital anxiety and depression scale. Acta Psychiatr. Scand., 1983. [DOI | PubMed]
- C.A. Zimmermann, A. Hoffmann, F. Raabe, D. Spengler. Role of mecp2 in experience-dependent epigenetic programming. Genes, 2015. [DOI | PubMed]
