Conservative Treatment of Sigmoid Diverticulum Perforation Secondary to Migrated Biliary Plastic Prostheses Inserted by Endoscopic Retrograde Cholangiopancreatography: A Case Report of an Unusual Adverse Event and Literature Review
Abstract
Distal migration of biliary plastic stents is rare. Although these stents are primarily used in the treatment of benign diseases of the biliopancreatic tract, their distal migration can lead to severe complications, such as perforation of any part of the digestive system. The authors report a case of sigmoid diverticulum perforation caused by the migration of a biliary plastic stent, which had been initially placed due to a failure to extract a common bile duct (CBD) stone. A review of similar cases in the literature was conducted, and the findings were analyzed in relation to the reported case. The search was performed in MEDLINE and the Cochrane Library, covering studies published between 1975 and 2025. Only studies describing the placement of biliary plastic stents during endoscopic retrograde cholangiopancreatography (ERCP) were included, while studies with incomplete data were excluded. This study highlights this rare and serious complication, which carries a high morbidity rate. Despite careful stent positioning during ERCP and periodic follow-up, this adverse event (AE) cannot always be prevented. Although distal stent migration with perforation can often be treated endoscopically, preoperative evaluation of the patient’s clinical condition and precise localization of the perforation is crucial for successful endoscopic stent removal, thus avoiding the need for surgery.
Article type: Case Report
Keywords: adverse events, biliary plastic prostheses, biliary stent, complication, intestinal, migrated stent, migration, perforation, stent, stents
License: Copyright © 2025, Orsini-Ardengh et al. CC BY 4.0 This is an open access article distributed under the terms of the Creative Commons Attribution License CC-BY 4.0., which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Article links: DOI: 10.7759/cureus.79042 | PubMed: 40099079 | PMC: PMC11913399
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (1.7 MB)
Introduction
The biliary plastic prostheses (PP), inserted during endoscopic retrograde cholangiopancreatography (ERCP), are used to treat benign and/or malignant biliary obstructions [ref. 1]. Common indications include bile leakage after cholecystectomy, failure to extract common bile duct (CBD) stones, post-cholecystectomy inflammatory biliary stenosis, post-liver transplant anastomosis biliary duct stricture (BDS), and pseudotumor chronic pancreatitis (PCP). On the malignant spectrum, pancreatic cancer (PC), cholangiocarcinoma, liver cancer, and hepatic metastases are among the conditions treated with PP [ref. 1,ref. 2].
Adverse events (AEs) occur in 5% of cases after PP insertion. These events are usually immediate and unrelated to the type of prostheses, which may include infections, hemorrhage, and pancreatitis. Late AEs include PP dysfunction, acute cholecystitis (AC), duodenal perforation, ulceration, and bleeding. Migration occurs in approximately 6% of PP cases, 1% of partially covered self-expandable metallic stents (SEMS), and 20% of fully covered SEMS. PP migration is more frequent in benign obstructive conditions, with endoscopic treatment being feasible in over 90% of cases with low morbidity [ref. 3,ref. 4].
Secondary perforation due to PP migration primarily affects the duodenum during insertion or late migration [ref. 5]. Other reported sites of perforation include the colon, distal ileum, liver, and pancreas. Diverticular disease of the colon increases the risk of secondary perforation from migrated PP [ref. 6,ref. 7]. Most cases require surgical procedures for PP removal, repair and suturing of the perforated organ(s), or resection of the affected colonic segments. However, the feasibility of endoscopic removal following distal migration of a PP with colon perforation depends on factors such as clinical stability, absence of peritoneal irritation, and availability of a surgical team for eligible patients [ref. 7,ref. 8]. A recent systematic review on this topic revealed that the global mortality rate was 17.4%, making this AE a concern regarding the treatment approach [ref. 9].
We report a case of sigmoid diverticular perforation caused by PP migration after the failed extraction of a giant choledocholithiasis. The clinical scenario of a perforated acute abdomen, the time between PP insertion and symptom onset, and the successful conservative treatment with PP removal via colonoscopy make this case noteworthy. Furthermore, the authors conducted a systematic review of reported cases of colon perforation caused by PP migration.
Case presentation
A 77-year-old female patient was admitted to the emergency room with abdominal pain localized in the left flank and iliac fossa, with no signs of peritoneal irritation, afebrile, and hemodynamically stable. Medical history revealed chronic diseases such as diabetes, hypertension, hypothyroidism, and obesity. The patient underwent cholecystectomy in 1990, due to calculous gallbladder disease. In 2009, she presented with mild jaundice with increased direct bilirubin levels (1.9 mg/dL), elevated alkaline phosphatase (777 U/L), gamma-glutamyl transpeptidase (1666.0 U/L), pyruvic transaminase (288 U/L), and oxaloacetic transaminase (264 U/L). At that time, abdominal ultrasonography (US) and magnetic resonance cholangiopancreatography (MR/MRCP) showed gallstones in the proximal third of the CBD with upstream ductal dilatation. On that occasion, an ERCP was performed to remove stones. Two years later she had a new episode of jaundice and underwent another ERCP, which showed saccular dilatation of the common hepatic duct with filling defects inside. Ductal screening with balloon extractor, stone removal, and placement of a polyethylene biliary stent (8.5 Fr x 9 cm) were performed.
The clinical history and physical examination supported the presumptive diagnosis of acute pancreatitis (AP). Computed tomography for emergency assessment showed a tubular structure with hyper attenuation, located in the lumen of the sigmoid. The upper end of the stent transfixed the colon and peritoneal membrane, reaching the left abdominal wall with no signs of peritonitis (Figure 1).
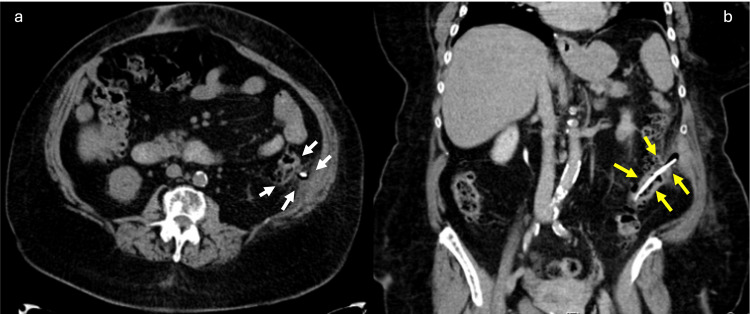
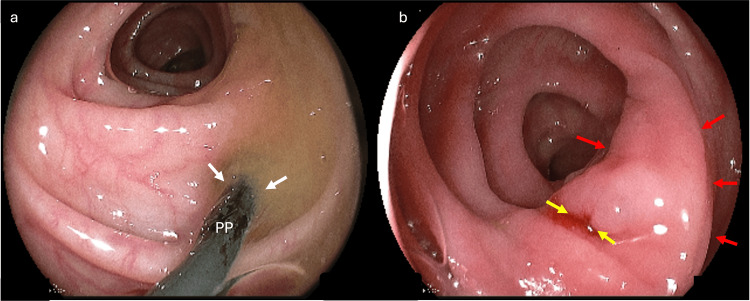
We opted for PP stent removal by colonoscopy despite the exuberant clinical setting of abdominal pain (Figure 2). The procedure was uneventful, with satisfactory patient recovery without surgical intervention. Intravenous ceftriaxone and metronidazole have been administered since her admission. She stayed in the hospital for three days after the prosthesis removal, with a good outcome and no pain complaints.
Literature review
Method
Population design: We included all patients who underwent ERCP with PP placement for the treatment of benign and/or malignant biliary and pancreatic diseases. Case reports with complete data regarding the occurrence and outcomes of perforation, as well as the therapeutic strategy employed, were selected.
Database and search strategy: The search was conducted in MEDLINE and the Cochrane Library for studies published between 1975 and 2025. Medical Subject Headings (MeSH) terminology and user-defined keywords commonly found in relevant articles were applied. The keywords included "(biliary plastic prostheses OR biliary stent OR stent OR stents) AND (migration OR migrated OR complication OR adverse events OR perforation, intestinal) AND (colon perforation OR colonic perforation OR sigmoid perforation OR caecum perforation OR appendiceal perforation OR diverticular impaction OR colovesical fistula)."
Only publications involving human subjects were included, and the bibliographies of relevant articles were reviewed to identify additional studies. We primarily identified case series related to this subject.
Data analysis and outcomes: Data was collected using a predesigned form (Excel spreadsheet) and considered for extraction when available in the text, tables, and/or figures, including information regarding treatment intent. The extracted data included the following categorical variables: epidemiological characteristics of the population (sex and age), indications and diagnoses (benign and malignant) for ERCP, performance of papillotomy, type of PP implanted (diameter and length), time between prosthesis placement and the onset of symptoms related to perforation, location of the perforation, and the therapeutic strategy adopted. Studies in which we did not find data we considered important, such as indication for ERCP, perforation site, and type of treatment administered, were excluded.
Results
Study population: This study included all case reports identified through the search strategy described in the previous subsection, in addition to the description of the current case. The results of the search strategy can be seen in Figure 3.
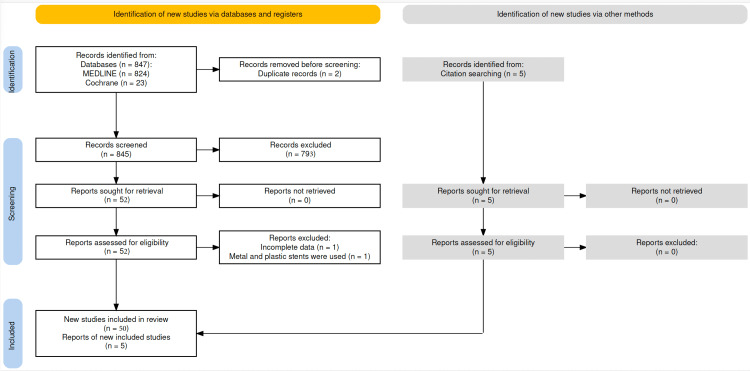
The search found 52 studies, of which two were excluded. One for describing the migration of a biliary pigtail PP associated with a biliary SEMS [ref. 10] and another for presenting incomplete data [ref. 11]. Another five reports were cited by other studies [ref. 12–ref. 16]. Therefore, 55 studies were identified, reporting a total of 58 patients, with Hogendorf et al. [ref. 15], Ruffolo et al. [ref. 17], and Virgilio et al. [ref. 18] reporting two cases each. Including the present case, 59 cases were analyzed [ref. 19]. The average age was 69.7 years (ranging from 26 to 94), with a higher prevalence in women: 40 out of 59 (68%) presented with colonic perforation due to the distal migration of the plastic biliary prosthesis inserted during ERCP. The clinical characteristics of the population are presented in Table 1.
Table 1: ABS, anastomotic bile stricture; AC, acute cholangitis; B, benign; BDS, bile duct strictures; BL, bile leakage; CBD, common bile duct; F, female; JDD, juxtapapillary duodenal diverticulum; LC, laparoscopic cholecystectomy; LT, liver transplantation; M, male; M, malign; N/A, not available; PC, pancreatic carcinoma; PCP, pseudotumoral chronic pancreatitis; PL, primary pancreatic lymphoma; SF, splenic flexure; PP, plastic prostheses
| N | Author, publication year | Gender | Age | Benign disease? | Type of lesion | Papilotomy (yes/no) | PP (diameter (F)) | PP (length (cm)) |
| 1 | Ruffolo TA et al., 1992 [ref. 17] | F | 72 | Yes | CBD stone | Yes | 10 | 12 |
| 2 | Ruffolo TA et al., 1992 [ref. 17] | F | 70 | Yes | CBD stones + AC | Yes | 7 | 5 |
| 3 | D’Costa H et al., 1994 [ref. 20] | M | 73 | No | PC | Yes | 10 | 12.5 |
| 4 | Schaafsma RJ et al., 1996 [ref. 21] | F | 77 | Yes | CBD stone | Yes | 7 | 10 |
| 5 | Baty V et al., 1996 [ref. 22] | F | 86 | No | PC | Yes | 10 | 10 |
| 6 | Lenzo NP and Garas G 1998 [ref. 23] | F | 85 | Yes | CBD stone | Yes | 10 | 7.5 |
| 7 | Ang BK et al., 1998 [ref. 24] | M | 71 | Yes | CBD stones + AC | Yes | 10 | 12 |
| 8 | Storkson RH et al., 2000 [ref. 25] | M | 86 | Yes | JDD + CBD dilatation | Yes | 7 | 5 |
| 9 | Figueras RG et al., 2001 [ref. 26] | M | 47 | Yes | PCP + BDS | Yes | 10 | 10 |
| 10 | Klein U et al., 2001 [ref. 27] | M | 70 | Yes | BDS after LC | Yes | 10 | 10 |
| 11 | Wilhelm A et al., 2003 [ref. 28] | F | 85 | Yes | CBD stone | Yes | 7 | 10 |
| 12 | Elliott M et al., 2003 [ref. 29] | F | 80 | Yes | CBD stone | Yes | 10 | 10 |
| 13 | Diller R et al., 2003 [ref. 30] | F | 58 | Yes | LT + ABS | Yes | 7 | 10 |
| 14 | Blake AM et al., 2004 [ref. 31] | F | 65 | Yes | CBD stone | Yes | N/A | N/A |
| 15 | Soto-Avila JJ et al., 2006 [ref. 32] | F | 47 | Yes | CBD stones + BDS | Yes | 7 | 10 |
| 16 | Anderson EM et al., 2007 [ref. 33] | F | 80 | Yes | CBD stone | Yes | 10 | 7 |
| 17 | Namdar T et al., 2007 [ref. 34] | F | 65 | Yes | BL after LC | No | 12 | 10 |
| 18 | Belyaev O et al., 2008 [ref. 35] | F | N/A | Yes | CBD stone | N/A | N/A | N/A |
| 19 | Aryal KR et al., 2008 [ref. 36] | F | 57 | Yes | BL after LC | Yes | 10 | 7 |
| 20 | Hunter K et al., 2010 [ref. 12] | F | 72 | Yes | BDS after LC | Yes | 10 | 10 |
| 21 | Bagul A et al., 2010 [ref. 37] | F | 79 | Yes | BDS after LC | Yes | 10 | 9 |
| 22 | Wagemakers S et al., 2011 [ref. 38] | F | 76 | Yes | CBD stone | Yes | N/A | N/A |
| 23 | Peter A et al., 2011 [ref. 39] | F | 69 | Yes | BDS after LC | Yes | 10 | 7 |
| 24 | Jafferbhoy SF et al., 2011 [ref. 40] | F | 82 | Yes | BL after LC | Yes | 7 | 7 |
| 25 | Lankisch TO et al., 2011 [ref. 41] | F | 65 | No | PC | Yes | 10 | 10 |
| 26 | Malgras B et al., 2011 [ref. 42] | M | 73 | No | PC | Yes | 10 | 5 |
| 27 | Alcaide N et al., 2012 [ref. 43] | M | 73 | Yes | CBD stones + BDS | Yes | 10 | 12 |
| 28 | Kittappa K et al., 2013 [ref. 44] | F | 58 | Yes | CBD stones + BDS | Yes | 10 | 12 |
| 29 | Jones M, et al., 2013 [ref. 45] | M | 66 | Yes | CBD stones + BDS | Yes | 10 | 8 |
| 30 | de Andres AB et al., 2013 [ref. 13] | M | 70 | Yes | CBD stone | Yes | 10 | 9 |
| 31 | Warnock JM et al., 2013 [ref. 14] | F | 77 | No | PL | Yes | 10 | 8 |
| 32 | Barut I and Tarhan OR, 2014 [ref. 46] | F | 26 | Yes | BDS after LC | Yes | 10 | 10 |
| 33 | Konstantinidis C et al., 2014 [ref. 47] | F | 69 | Yes | CBD stone | Yes | N/A | N/A |
| 34 | Virgilio E et al., 2015 [ref. 18] | F | N/A | Yes | CBD stone | N/A | N/A | N/A |
| 35 | Virgilio E et al., 2015 [ref. 18] | F | N/A | Yes | CBD stone | Yes | 10 | 12 |
| 36 | Mady RF et al., 2015 [ref. 48] | M | N/A | No | PC | N/A | N/A | N/A |
| 37 | Chittleborough TJ et al., 2016 [ref. 49] | M | 73 | Yes | CBD stones + AC | Yes | 10 | 5 |
| 38 | Chou, ND et al., 2017 [ref. 50] | F | 85 | Yes | CBD stone | N/A | N/A | N/A |
| 39 | Siaperas P et al., 2017 [ref. 51] | F | 75 | Yes | CBD stone | Yes | 10 | 7 |
| 40 | Hogendorf P et al., 2018 [ref. 15] | F | 76 | Yes | CBD stone | N/A | N/A | N/A |
| 41 | Hogendorf et al., 2018 [ref. 15] | M | 68 | No | PC | Yes | 7 | 10 |
| 42 | Riccardi, M et al., 2019 [ref. 52] | F | 79 | Yes | CBD stone | Yes | 10 | 10 |
| 43 | Hnaris K and Bechara R, 2019 [ref. 53] | F | 90 | Yes | Mirizzi’s syndrome | Yes | 10 | 10 |
| 44 | Marcos P et al., 2020 [ref. 54] | F | 65 | Yes | CBD stone | Yes | 10 | 5 |
| 45 | Tao Y and Long J, 2021 [ref. 55] | M | 54 | Yes | CBD stones + AC + AP | N/A | N/A | N/A |
| 46 | Pengemä P et al, 2021 [ref. 56] | F | 66 | Yes | PCP + BDS | Yes | 10 | 5 |
| 47 | Park TY et al., 2021 [ref. 57] | M | 74 | Yes | CBD stones + AC | Yes | 10 | 7 |
| 48 | Ong XZ and Leow Y, 2021 [ref. 16] | M | 57 | Yes | CBD stone | N/A | N/A | N/A |
| 49 | Yamaguchi D et al., 2022 [ref. 58] | F | 86 | Yes | CBD stones + AC | Yes | 7 | 7 |
| 50 | Kodia K et al., 2022 [ref. 59] | F | 60 | No | GC + CBD invasion | N/A | N/A | N/A |
| 51 | Kwong E et al., 2023 [ref. 60] | F | 94 | Yes | CBD stone | Yes | 10 | 7 |
| 52 | Rybinski F et al., 2023 [ref. 61] | M | 78 | Yes | CBD stones + AC + AP | Yes | 10 | 10 |
| 53 | Mohammadi Tofigh A et al., 2023 [ref. 62] | F | 65 | Yes | CBD stone | Yes | 10 | 10 |
| 54 | Berdugo Hurtado F et al., 2023 [ref. 63] | M | 50 | Yes | BDS after LC | Yes | 10 | 7 |
| 55 | He Y et al., 2024 [ref. 64] | M | 35 | Yes | CBD stone | Yes | 8.5 | 10 |
| 56 | Vergara-Fernándes O et al., 2024 [ref. 65] | M | 67 | No | PC | Yes | 10 | 8 |
| 57 | Swied MY et al., 2024 [ref. 66] | F | 54 | YES | BDS after LC | Yes | 7 | 10 |
| 58 | Beloy JB et al., 2024 [ref. 67] | F | 79 | Yes | CBD stones + AC | Yes | 8.5 | 7 |
| 59 | Ardengh AO et al., 2025 | F | 77 | Yes | CBD stone | Yes | 8.5 | 9 |
Indication for ERCP and sphincterotomy: Fifty out of 59 (85%) patients had benign findings involving the biliary and/or pancreatic ducts, while nine out of 59 (15%) had malignant diseases. All were treated with PP insertion during ERCP. Among the benign findings, the diagnoses included BDS (14), biliary fistula after laparoscopic cholecystectomy (3), choledocholithiasis (36), and a large juxtapapillary diverticulum with CBD dilation (1). Among the identified malignant diseases, 7/59 (12%) were cases of PC, 1/59 (2%) was a primary pancreatic lymphoma, and 1/59 (2%) was a gallbladder carcinoma with CBD invasion. Eleven out of 59 (22%) patients had CBD stricture after cholecystectomy (4/59 (7%) of whom had associated stones), 2/59 (3%) had distal bile duct stricture due to pseudotumoral chronic pancreatitis (PCP), and 1/59 (2%) had a bile duct-to-bile duct anastomosis stricture after liver transplantation. In 36/59 (61%) cases, the authors reported difficulty clearing the bile duct due to the presence of stones, leading to the implantation of PP in 36/59 (61%). Of these, 7/59 (12%) had traumatic bile duct stricture, and 8/59 (13.5%) had acute cholangitis (AC), in which 1/59 (2%) also presented with pancreatitis. Three out of 59 (5%) underwent ERCP with PP insertion due to bile leakage through the cystic duct after cholecystectomy. Sphincterotomy before PP insertion was described in 51/59 (86%) and was performed in 50/51 (98%) patients.
Diameter and length of PPs and migration time: Details regarding the characteristics of the PPs were described in 48/59 (81%) cases. A 10F PP was inserted in 34 out of 48 (71%) cases, a 7F in 10 out of 48 (21%), an 8.5F in three out of 48 (6%), and a 12F in one out of 48 (2%). The length of the PPs was reported as 5 cm, 7 cm, 7.5 cm, 8 cm, 9 cm, 10 cm, 12 cm, and 12.5 cm in 6/48 (13%), 10/48 (21%), 1/48 (2%), 3/48 (6%), 3/48 (6%), 19/48 (40%), 5/48 (10%), and 1/48 (2%) case, respectively. A 10F and 10 cm BPP was implanted in 11/48 (23%) of the cases.
The migration time of the PP was expressed in days and referred to the period between PP implantation during ERCP and the onset of symptoms due to perforation. This analysis was possible in 53/59 (90%) cases. The average time reported was 274.4 days (min. 4 days, max. 2920 days).
Perforation site: The perforation site was described in all reports. The appendix, caecum, ascending colon, sigmoid, and rectum were the sites where PPs were found via endoscopy and/or surgery in 2/59 (3.4%), 4/59 (6.8%), 10/59 (17%), 41/59 (69%), and 2/59 (3.4%) cases, respectively. The sigmoid was the most affected segment, and in 23/59 (39%) patients, the perforation occurred at a diverticulum where the PP had impacted. Overall, 24/59 (40.6%) patients had perforation and PP impaction at a diverticulum, of which 23/59 (39%) in the sigmoid colon and 1/59 (1.7%) in the ascending colon.
Major complications during and after perforation: Major complications during perforation included 3/59 (5.1%) pelvic abscesses: 1/59 (1.7%) during cecal perforation, 1/59 (1.7%) during sigmoid perforation, and 1/59 (1.7%) occurring days after endoscopic removal of the PP in the sigmoid colon. Other severe complications included fistula formation in 7/59 (11.8%) cases: 3/59 (5.1%) coloduodenal fistula with ascending colon perforation, 2/59 (3.4%) colovesical fistulas after sigmoid perforation, 1/59 (1.7%) colovaginal fistula after sigmoid perforation, and 1/42 (1.7%) colocutaneous fistula after ascending colon perforation, treated by manual removal of the PP. Additionally, double perforation occurred in 3/59 (5.1%) patients due to PP impaction in the sigmoid diverticula (2) and sigmoid (1).
Treatment: Treatment was surgical, endoscopic, combined (endoscopy+surgery), or manual in 42/59 (71.2%), 14/59 (23.7%), 2/59 (3.4%), and 1/59 (1.7%) cases, respectively. For the combined treatment, in 1/59 (2.3%) patients, the PP was removed endoscopically, but the presence of a pelvic abscess required surgical intervention to close the orifice. Another case involved endoscopic PP removal followed by the development of a pelvic abscess days later, which required surgical treatment. Among the 14 endoscopic treatments, in 4/16 (25%) cases, the endoscopic removal of the PP was followed by the placement of metallic clips to close the perforated diverticular orifice (Table 2).
Table 2: *After the endoscopic removal, the patient developed a diverticular perforation and required an emergency laparotomy with sigmoid resection. **The patient died after surgery. PP, plastic prostheses
| N | Author, publication year | Migration time (days) | Perforation (site) | Treatment |
| 1 | Ruffolo TA et al., 1992 [ref. 17] | 365 | Sigmoid diverticulum | Endoscopy removal |
| 2 | Ruffolo TA et al., 1992 [ref. 17] | 60 | Sigmoid diverticulum | Endoscopy removal |
| 3 | D’Costa H et al., 1994 [ref. 20] | 90 | Ascending colon | Surgery |
| 4 | Schaafsma RJ et al., 1996 [ref. 21] | 180 | Sigmoid diverticulum | Surgery |
| 5 | Baty V et al., 1996 [ref. 22] | 21 | Sigmoid diverticulum | Surgery |
| 6 | Lenzo NP and Garas G, 1998 [ref. 23] | 30 | Sigmoid diverticulum | Surgery |
| 7 | Ang BK et al., 1998 [ref. 24] | 1095 | Coloduodenal fistula (ascending colon) | Surgery |
| 8 | Storkson RH et al, 2000 [ref. 25] | 14 | Sigmoid | Surgery |
| 9 | Figueras RG et al., 2001 [ref. 26] | 14 | Colocutaneous fistula (ascending colon) | Manual removal |
| 10 | Klein U et al., 2001 [ref. 27] | 1095 | Sigmoid diverticulum | Surgery |
| 11 | Wilhelm A et al., 2003 [ref. 28] | 730 | Colovesical fistula (sigmoid) | Surgery |
| 12 | Elliott M et al., 2003 [ref. 29] | 120 | Sigmoid | Surgery |
| 13 | Diller R et al., 2003 [ref. 30] | 38 | Pelvic abscess (sigmoid diverticulum) | Endoscopy removal + surgery* |
| 14 | Blake AM et al., 2004 [ref. 31] | 90 | Colovaginal fistula (sigmoid diverticulum) | Surgery |
| 15 | Soto-Avila JJ et al., 2006 [ref. 32] | 540 | Colovesical fistula (sigmoid diverticulum) | Surgery |
| 16 | Anderson EM et al., 2007 [ref. 33] | 60 | Pelvic abscess (sigmoid) | Endoscopy removal |
| 17 | Namdar T et al., 2007 [ref. 34] | 540 | Rectum | Surgery |
| 18 | Belyaev O et al., 2008 [ref. 35] | 21 | Sigmoid diverticulum (double perforation) | Surgery |
| 19 | Aryal KR et al., 2008 [ref. 36] | 90 | Sigmoid diverticulum | Surgery |
| 20 | Hunter K et al., 2010 [ref. 12] | 2190 | Sigmoid diverticulum | Surgery |
| 21 | Bagul A et al., 2010 [ref. 37] | 30 | Sigmoid diverticulum | Endoscopy removal |
| 22 | Wagemakers S et al., 2011 [ref. 38] | 30 | Sigmoid | Surgery |
| 23 | Peter A et al., 2011 [ref. 39] | 150 | Sigmoid diverticulum | Surgery |
| 24 | Jafferbhoy SF et al., 2011 [ref. 40] | 90 | Sigmoid diverticulum | Endoscopy removal |
| 25 | Lankisch TO et al., 2011 [ref. 41] | 60 | Sigmoid | Surgery |
| 26 | Malgras B et al, 2011 [ref. 42] | 15 | Sigmoid diverticulum (double perforation) | Surgery |
| 27 | Alcaide N et al., 2012 [ref. 43] | 15 | Sigmoid diverticulum | Endoscopy removal |
| 28 | Kittappa K et al., 2013 [ref. 44] | 540 | Sigmoid diverticulum | Surgery |
| 29 | Jones M et al., 2013 [ref. 45] | 540 | Pelvic abscess (caecum) | Endoscopy removal + surgery |
| 30 | de Andres AB et al., 2013 [ref. 13] | 60 | Sigmoid diverticulum | Surgery |
| 31 | Warnock JM et al., 2013 [ref. 14] | 60 | Sigmoid diverticulum | Surgery ** |
| 32 | Barut I and Tarhan OR, 2014 [ref. 46] | 330 | Caecum | Surgery |
| 33 | Konstantinidis C et al., 2014 [ref. 47] | 60 | Sigmoid (double perforation) | Surgery |
| 34 | Virgilio E et al., 2015 [ref. 18] | N/A | Sigmoid | Surgery |
| 35 | Virgilio E et al., 2015 [ref. 18] | N/A | Sigmoid | Endoscopy removal |
| 36 | Mady RF et al., 2015 [ref. 48] | 30 | Sigmoid | Surgery |
| 37 | Chittleborough TJ et al., 2016 [ref. 49] | 30 | Sigmoid | Surgery |
| 38 | Chou ND et al., 2017 [ref. 50] | N/A | Sigmoid | Endoscopy removal + clip closure |
| 39 | Siaperas P et al., 2017 [ref. 51] | 180 | Sigmoid | Surgery |
| 40 | Hogendorf P et al., 2018 [ref. 15] | 180 | Sigmoid diverticulum | Surgery |
| 41 | Hogendorf et al., 2018 [ref. 15] | 30 | Rectum | Surgery |
| 42 | Riccardi M et al., 2019 [ref. 52] | 28 | Sigmoid | Surgery |
| 43 | Hnaris K and Bechara R, 2019 [ref. 53] | N/A | Ascending colon | Endoscopy removal + clip closure |
| 44 | Marcos P et al., 2020 [ref. 54] | 30 | Sigmoid | Surgery |
| 45 | Tao Y and Long J, 2021 [ref. 55] | 90 | Sigmoid | Surgery |
| 46 | Pengemä P et al., 2021 [ref. 56] | 4 | Appendix | Surgery |
| 47 | Park TY et al., 2021 [ref. 57] | 30 | Ascending colon diverticulum | Surgery |
| 48 | Ong XZ and Leow Y, 2021 [ref. 16] | 2920 | Caecum | Surgery |
| 49 | Yamaguchi D et al., 2022 [ref. 58] | 30 | Sigmoid diverticulum | Endoscopy removal + clip closure |
| 50 | Kodia K et al., 2022 [ref. 59] | 60 | Ascending colon | Surgery |
| 51 | Kwong E et al., 2023 [ref. 60] | N/A | Sigmoid | Endoscopy removal |
| 52 | Rybinski F et al., 2023 [ref. 61] | 30 | Ascending colon | Endoscopy removal + clip closure |
| 53 | Mohammadi Tofigh A et al., 2023 [ref. 62] | 1095 | Caecum | Surgery |
| 54 | Berdugo Hurtado F et al., 2023 [ref. 63] | 120 | Coloduodenal fistula (ascending colon) | Endoscopy removal |
| 55 | He Y et al., 2024 [ref. 64] | 60 | Coloduodenal fistula (ascending colon) | Surgery |
| 56 | Vergara-Fernándes O et al., 2024 [ref. 65] | 43 | Ascending colon | Surgery |
| 57 | Swied MY et al., 2024 [ref. 66] | N/A | Appendix | Surgery |
| 58 | Beloy JB et al., 2024 [ref. 67] | 11 | Sigmoid | Surgery |
| 59 | Ardengh AO et al., 2025 | 180 | Sigmoid diverticulum | Endoscopy removal |
Discussion
The insertion of a PP in the CBD is a well-established therapeutic strategy for the treatment of obstructive jaundice caused by benign and malignant diseases [ref. 1,ref. 2]. Complications are rare and may occur either during the insertion of the PP or subsequently [ref. 3]. Early complications include AP, infection, and bleeding. Inadvertent manipulation of the CBD and/or the main pancreatic duct during catheterization, sphincterotomy, or the use of contrast agents activates pancreatic coenzymes, triggering an inflammatory process that may lead from minimal to severe systemic complications [ref. 4]. Late complications related to the use of PP include dysfunction, obstruction, migration, AC, bleeding, intestinal obstruction, ulceration, and perforation of the small intestine or colon, as well as other organs, resulting in abscesses and/or fistulas [ref. 3–ref. 5], as observed in the patient of the current case report.
The PP migration occurs in approximately 6% of cases [ref. 2]. Some studies report incidence rates of proximal and distal migration of biliary and pancreatic PP ranging from 4.9% to 7.5% [ref. 2,ref. 3]. Distal migration of PP is more frequent in benign strictures compared to those caused by malignant diseases, similar to what occurred with our patient [ref. 2,ref. 3].
PP dysfunction is frequently delayed in diagnosis and is significant. The migration of the PP can occur proximally or distally. Obstruction of the PP is caused by "dirty" or thick bile, biliary sludge, bacterial biofilm, or tumor growth through or around the PP [ref. 2–ref. 4]. This complication is much more common in PP compared to SEMS, especially in smaller-diameter PP.
Some authors believe that adherence caused by tumor tissue decreases PP mobility within the CBD, thereby reducing the likelihood of migration. The insertion of PP reduces the local inflammatory process found in benign conditions, decreasing PP adherence and facilitating its displacement. Other authors have reported that malignant diseases more effectively anchor the PP, acting as a rack, causing its displacement, particularly toward the proximal portion of the biliary tract. The implantation of a single, short, and thick PP also favors displacement [ref. 3–ref. 6].
Endoscopic treatment of migrated PP is feasible in 90% of cases with low morbidity [ref. 2]. When follow-up imaging does not identify the PP in the CBD and migration toward the intestine is confirmed, PP retrieval depends on its location. Usually, the PP is excreted in feces without major issues. However, when it remains impacted for an extended period, it can lead to serious and potentially life-threatening complications. Most complications due to distal migration occur in the duodenum. In cases of partial migration involving a long PP, friction against the duodenal wall opposite to the papilla may result in ulceration, which can progress to perforation [ref. 8,ref. 18,ref. 49]. Abdominal diseases such as inflammatory bowel strictures, internal hernias, and diverticular disease of the colon are risk factors for PP impaction, increasing the risk of intestinal loop perforation, similar to the patient in the current case report [ref. 6,ref. 8]. Following distal migration, PP can be removed via colonoscopy, especially in the absence of colon perforation. However, in cases of potential perforation, a detailed assessment of the patient’s clinical condition and imaging findings guides the best treatment approach, whether endoscopic or surgical. The absence of peritonitis, sepsis, hemodynamic instability, and the involvement of a trained surgical team allows for the endoscopic removal of PP without subsequent complications, as demonstrated within the case report [ref. 17,ref. 18,ref. 30,ref. 33,ref. 37,ref. 40,ref. 43,ref. 45,ref. 50,ref. 53,ref. 58,ref. 60,ref. 61,ref. 63].
A review of the literature identified 58 cases of distally migrated PP with colon perforation [ref. 12–ref. 18,ref. 20–ref. 67]. Most patients were elderly, with a mean age of 69.7 years, and a higher prevalence in women (40/59 (68%)), as observed in our patient. Most cases involved benign diseases (50 out of 59 (85%)) affecting the biliary tract, while malignant diseases were present in nine out of 59 (15%) of cases. Choledocholithiasis (36/59 (61%)) was the most prevalent finding, similar to our case, followed by bile duct strictures (14/59 (23.7%)), biliary fistulas post-laparoscopic cholecystectomy (3/59 (6%)), and giant juxtapapillary diverticulum with CBD dilation (1/59 (2%)). PC, primary pancreatic lymphoma, and gallbladder carcinoma were identified in 7/59 (12%), 1/59 (2%), and 1/59 (2%) of cases, respectively.
Difficulty clearing the CBD due to calculi, leading to the option of a PP implant, was frequently reported in 23 patients found in the literature [ref. 22], similar to our case. As in our patient, the 10F, 10 cm PP was the most frequently implanted (18.6%) among all patients, indicating that large diameter and length do not prevent distal PP migration. Notably, the time for PP migration ranged from four days to eight years, with an average of 274.4 days (4-2920). Our patient presented symptoms 180 days after PP insertion via ERCP for the treatment of large CBD stones.
In most cases, the exact time of displacement, impaction, and intestinal perforation could not be estimated due to a lack of clinical follow-up. The preferred site for PP impaction was the sigmoid colon in 41/59 (69.4%), followed by the ascending colon in 10/59 (17%). Diverticular disease of the colon was the most significant risk factor, present in 23/59 (39%) and 1/59 (1.7%) of the sigmoid and ascending colon cases, respectively. These risk factors were also present in our patient.
Major complications included pelvic abscesses in 3/59 (5%) and fistulas in 7/59 (11.8%), such as colovesical in 2/59 (4.6%), coloduodenal in 3/59 (5%), colovaginal in 1/59 (1.7%), and colocutaneous in 1/59 (1.7%) fistulas. Most patients presented with abdominal pain; two exhibited urinary symptoms, including recurrent infections, pneumaturia, and dysuria [ref. 22,ref. 25]. Among these, one had fecal leakage into the vagina [ref. 23], one had cutaneous drainage of secretions [ref. 8], and another presented with osteoarticular symptoms due to a pelvic abscess, similar to what occurred in this case report. All were managed surgically.
Treatment included surgical, endoscopic, combined (endoscopy + surgery), and manual PP removal approaches in 42/59 (71.2%), 14/59 (23.7%), 2/59 (3.4%), and 1/59 (1.7%) cases, respectively. In four endoscopic cases (25%), metallic clips were placed to close the perforated diverticular orifice after PP removal. In our case, this approach was unnecessary as computed tomography already showed the area around the PP to be blocked. In addition, the mortality rate resulting from postoperative complications in this study was 1/59 (1.7%) [ref. 14]. This rate is below that described in the systematic review by Wilson et al, which was 17.4% [ref. 9].
Conclusions
This study describes a rare and serious complication with high morbidity following distal migration of a PP used in the treatment of giant choledocholithiasis. Careful positioning of the PP during ERCP at the time of insertion and periodic follow-up after its implantation did not prevent this complication. Although distal migration of the PP can lead to perforation, it can be treated endoscopically. However, preoperative assessment of the patient’s clinical condition and precise localization of the perforation are essential for the successful removal of the PP via endoscopy.
References
- JP AbiMansour, JA Martin. Biliary endoscopic retrograde cholangiopancreatography. Gastroenterol Clin North Am, 2024. [PubMed]
- JM Dumonceau, A Tringali, IS Papanikolaou. Endoscopic biliary stenting: indications, choice of stents, and results: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline – Updated October 2017. Endoscopy, 2018. [PubMed]
- MA Anderson, L Fisher, R Jain. Complications of ERCP. Gastrointest Endosc, 2012. [PubMed]
- L Leifeld, R Jakobs, T Frieling. Influence of specialization on primary success and complication rate in ERCP. Proposal to improve the quality of ERCP. Z Gastroenterol, 2024. [PubMed]
- M Yaprak, A Mesci, T Colak. Biliary stent migration with duodenal perforation. Eurasian J Med, 2008. [PubMed]
- M Arhan, B Odemiş, E Parlak, I Ertuğrul, O Başar. Migration of biliary plastic stents: experience of a tertiary center. Surg Endosc, 2009. [PubMed]
- GA Paspatis, V Papastergiou, A Mpitouli. Distal biliary stent migration in patients with irretrievable bile duct stones: long-term comparison between straight and double-pigtail stents. Dig Dis Sci, 2022. [PubMed]
- JF Johanson, MJ Schmalz, JE Geenen. Incidence and risk factors for biliary and pancreatic stent migration. Gastrointest Endosc, 1992. [PubMed]
- N Wilson, C Ezeani, A Ismail. Bowel perforation caused by biliary stent migration after ERCP: a systematic review. J Clin Gastroenterol, 2024
- Z Wilson, J Diab, S Putnis. Migratory biliary stent resulting in colonic perforation: a rare complication and review of literature. J Surg Case Rep, 2023
- Y Asghari, F Firuzpour. Distal sigmoid perforation secondary to migration of a biliary stent: a case report. Int J Surg Case Rep, 2025. [PubMed]
- K Hunter, T Siddiqui, Oo Komolafe, DCS Chong. Colonic perforation secondary to migrated biliary stent. Case report of an unusual complication, and literature review. Scottish Med J, 2010
- B de Andrés A, R Francisca Moreno, M Patricia Legido, J José Rabadán, R Juan Beltrán de Heredia. Perforación diverticular sigmoidea secundaria a la migración de endoprótesis biliar plástica. Rev Chil Cir (online), 2013
- JM Warnock, RJ Thompson. A potentially fatal complication of biliary stent migration. J Med Cases, 2013
- P Hogendorf, A Skulimowski, A Durczyński, J Strzelczyk. Migrated plastic biliary stent causing colon perforation: analysis of emergency admission and literature review. Post N Med, 2018
- X Ong, Y Leow. Cecal perforation secondary to migrated stent mimicking perforated appendicitis. SAS J Surg, 2021
- TA Ruffolo, GA Lehman, S Sherman, R Aycock, A Hayes. Biliary stent migration with colonic diverticular impaction. Gastrointest Endosc, 1992. [PubMed]
- E Virgilio, G Pascarella, CM Scandavini, B Frezza, T Bocchetti, G Balducci. Colonic perforations caused by migrated plastic biliary stents. Korean J Radiol, 2015. [PubMed]
- NR Haddaway, MJ Page, CC Pritchard, LA McGuinness. PRISMA2020: An R package and Shiny app for producing PRISMA 2020-compliant flow diagrams, with interactivity for optimised digital transparency and Open Synthesis. Campbell Syst Rev, 2022
- H D’Costa, E Toy, MJ Dennis, C Brown. Intestinal perforation—an unusual complication of endoscopic biliary stenting. Br J Radiol, 1994. [PubMed]
- RJ Schaafsma, P Spoelstra, J Pakan, K Huibregtse. Sigmoid perforation: a rare complication of a migrated biliary endoprosthesis. Endoscopy, 1996. [PubMed]
- V Baty, B Denis, MA Bigard, P Gaucher. Sigmoid diverticular perforation relating to the migration of a polyethylene endoprosthesis. Endoscopy, 1996. [PubMed]
- NP Lenzo, G Garas. Biliary stent migration with colonic diverticular perforation. Gastrointest Endosc, 1998. [PubMed]
- BK Ang, SB Wee, SP Kaushik, CH Low. Duodenal-colic fistula resulting from migration of a biliary stent: a case report. Gastrointest Endosc, 1998. [PubMed]
- RH Størkson, B Edwin, O Reiertsen, AE Faerden, O Sortland, AR Rosseland. Gut perforation caused by biliary endoprosthesis. Endoscopy, 2000. [PubMed]
- RG Figueiras, MO Echart, AG Figueiras, GP González. Colocutaneous fistula relating to the migration of a biliary stent. Eur J Gastroenterol Hepatol, 2001. [PubMed]
- U Klein, F Weiss, O Wittkugel. Migration of a biliary Tannenbaum stent with perforation of sigmoid diverticulum (German). Rofo, 2001. [PubMed]
- A Wilhelm, C Langer, G Zoeller, R Nustede, H Becker. Complex colovesicular fistula: a severe complication caused by biliary stent migration. Gastrointest Endosc, 2003. [PubMed]
- M Elliott, S Boland. Sigmoid colon perforation following a migrated biliary stent. ANZ J Surg, 2003. [PubMed]
- R Diller, N Senninger, G Kautz, D Tübergen. Stent migration necessitating surgical intervention. Surg Endosc, 2003. [PubMed]
- AM Blake, N Monga, EM Dunn. Biliary stent causing colovaginal fistula: case report. JSLS, 2004. [PubMed]
- JJ Soto-Ávila, ME Gómez-Wong, F Preciado-Hernández. Fístula colovesical secundaria a la migración de una endoprótesis biliar. Reporte de un caso. Cir Cir, 2006. [PubMed]
- EM Anderson, J Phillips-Hughes, R Chapman. Sigmoid colonic perforation and pelvic abscess complicating biliary stent migration. Abdom Imaging, 2007. [PubMed]
- T Namdar, AM Raffel, SA Topp. Complications and treatment of migrated biliary endoprostheses: a review of the literature. World J Gastroenterol, 2007. [PubMed]
- O Belyaev, CA Müller, W Uhl. Double sigmoid colon perforation by a migrated biliary stent. Acta Chirurgica Belgica, 2008. [PubMed]
- KR Aryal, DJ Sherlock. A case of colonic perforation from biliary stent. Endoscopy, 2008
- A Bagul, C Pollard, AR Dennison. A review of problems following insertion of biliary stents illustrated by an unusual complication. Ann R Coll Surg Engl, 2010
- S Wagemakers, M Ibelings. Enterovesicale fistel door gemigreerde galwegstent (Dutch). Ned Tijdschr Geneeskd, 2011
- A Peter, M Walker, K Shinil. Biliary stent migration presenting as transdiverticular sigmoid perforation. J Surg Case Rep, 2011
- SF Jafferbhoy, P Scriven, J Bannister, MH Shiwani, P Hurlstone. Endoscopic management of migrated biliary stent causing sigmoid perforation. BMJ Case Rep, 2011
- TO Lankisch, TA Alten, F Lehner, W Knitsch. Biliary stent migration with colonic perforation: a very rare complication and the lesson that should be learned from it. Gastrointest Endosc, 2011
- B Malgras, C Pierret, JP Tourtier, G Olagui, C Nizou, V Duverger. Double Sigmoid colon perforation due to migration of a biliary stent. J Visc Surg, 2011
- N Alcaide, S Lorenzo-Pelayo, MT Herranz-Bachiller, C de la Serna-Higuera, J Barrio, M Perez-Miranda. Sigmoid perforation caused by a migrated biliary stent and closed with clips. Endoscopy, 2012
- K Kittappa, K Maruthachalam, R Brookstein, S Debrah. Migrated biliary stent presenting as a sigmoid diverticulitis-case report. Indian J Surg, 2013
- M Jones, B George, J Jameson, G Garcea. Biliary stent migration causing perforation of the caecum and chronic abdominal pain. BMJ Case Rep, 2013
- I Barut, OR Tarhan. Cecum perforation due to biliary stent migration. Saudi Med J, 2014. [PubMed]
- C Konstantinidis, P Varsos, S Kympouris, S Volteas. Migrated biliary plastic stent causing double sigmoid colon perforation. J Surg Case Rep, 2014
- RF Mady, OS Niaz, MM Assal. Migrated biliary stent causing perforation of sigmoid colon and pelvic abscess. BMJ Case Rep, 2015
- TJ Chittleborough, S Mgaieth, B Kirkby, J Zakon. Remove the migrated stent: sigmoid colon perforation from migrated biliary stent. ANZ J Surg, 2016. [PubMed]
- ND Chou, RA Burbridge, PS Jowell. Colonic perforation secondary to retained biliary stent. Am J Gastroenterol, 2017
- P Siaperas, A Ioannidis, A Skarpas, A Angelopoulos, I Drikos, I Karanikas. A rare cause for Hartmann’s procedure due to biliary stent migration: a case report. Int J Surg Case Rep, 2017. [PubMed]
- M Riccardi, K Deters, F Jabbar. Sigmoid diverticulitis and perforation secondary to biliary stent migration. Case Rep Surg, 2019. [PubMed]
- K Hnaris, R Bechara. Removal of a migrated biliary stent perforating the colonic wall: a case description of endoscopic repair. Endoscopy, 2019
- P Marcos, G Capelão, C Atalaia-Martins, P Clara, L Eliseu, H Vasconcelos. Sigmoid perforation by a migrated plastic biliary stent. GE Port J Gastroenterol, 2020. [PubMed]
- Y Tao, J Long. Sigmoid colon perforation caused by migrated plastic biliary stents: a case report. Int J Colorectal Dis, 2021. [PubMed]
- P Pengermä, J Katunin, A Turunen, P Sileri, G Giarratano, A Palomäki, A Kechagias. Appendiceal perforation due to biliary stent migration in a neutropenic patient with lung cancer receiving chemotherapy: a case report. Mol Clin Oncol, 2021. [PubMed]
- TY Park, SW Hong, HC Oh, JH Do. Colonic diverticular perforation by a migrated biliary stent: a case report with literature review. Medicine (Baltimore), 2021
- D Yamaguchi, G Nagatsuma, A Jinnouchi. Successful endoscopic closure with an over-the-scope clip for sigmoid colon perforation due to bile duct stent migration. Clin J Gastroenterol, 2022. [PubMed]
- K Kodia, CT Huerta, Y Arora, C Wickham, AR Deshpande, N Paluvoi. Minimally invasive management of an ascending colonic perforation secondary to distal biliary stent migration: a multidisciplinary, novel laparoendoscopic approach. J Surg Case Rep, 2022
- E Kwong, K Jones, S Park, K Kwok. Successful nonoperative management of a sigmoid colon perforation caused by a migrated biliary stent. ACG Case Rep J, 2023
- F Rybinski, H Heinrich, M Zimmerli, S Kahl. The pierced colon: when biliary stents go the wrong way. ACG Case Rep J, 2023
- A Mohammadi Tofigh, H Tahmasbi, M Iranshahi, A Haghbin Toutounchi, H Khoshnoudi, SP Kouchak Hosseini. Cecal perforation induced by migrated biliary stent as a rare complication of ERCP: a case report and literature review. Case Rep Surg, 2023. [PubMed]
- F Berdugo Hurtado, A Ortiz Sánchez, B Zúñiga de Mora Figueroa, A Barrientos Delgado. Endoscopic rescue of a fistulizing biliary plastic stent. Rev Esp Enferm Dig, 2023. [PubMed]
- Y He, R Ye, Y Xie, X Deng, L Zhang. Endoscopic treatment of duodenocolic fistula caused by migrating biliary stents. Am J Case Rep, 2024
- O Vergara-Fernández, D Tueme de la Peña, J Canto-Losa. Plastic biliary stent migration as a cause of ascending colon perforation. Rev Esp Enferm Dig, 2024. [PubMed]
- MY Swied, Y Al Turk, M Maitar. Migrated plastic biliary stent causing cecal perforation. ACG Case Rep J, 2024
- JB Beloy, NP Lund, AM Van Hell, S Allamaneni. Case report: migratory biliary stent resulting in sigmoid colon perforation. J Surg Case Rep, 2024
