Gibberellic Acid (GA3): A Versatile Chiral Building Block for Syntheses of Pharmaceutical Agents
Abstract
Gibberellic acid (GA3), an ent‐kaurene tetracyclic diterpene, has been considered to be a chiral pool for the chemical transformation of significant heterocyclic compounds. This chiral pool continues to influence modern synthetic chemistry as an inexpensive and versatile starting material since it is widely applied in agriculture. This review focuses on the stereoselective syntheses of bioactive agents with pharmaceutical potency prepared from Gibberellic acid.
Article type: Review Article
Keywords: Gibberellins, Gibberellic acid (GA, Stereoselective syntheses, Pharmaceutical agents, Building block
Affiliations: Institute of Pharmaceutical Chemistry University of Szeged Eötvös utca 6 H-6720 Szeged Hungary; HUN-REN–SZTE Stereochemistry Research Group of the Hungarian Academy of Sciences Eötvös utca 6 H-6720 Szeged Hungary
License: © 2024 The Author(s). Chemistry & Biodiversity published by Wiley-VHCA AG CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202401823 | PubMed: 39307688 | PMC: PMC11741152
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (6.2 MB)
Introduction
Historical Perspective and Classification of GAs
The family of gibberellins (GAs), a group of phytohormones, regulate various stages of plant growth and development,ref. cbdv202401823-bib-0001 including stem and root expansion,ref. cbdv202401823-bib-0002 flowering[ref. cbdv202401823-bib-0003, ref. cbdv202401823-bib-0004] and seed germination.[ref. cbdv202401823-bib-0005, ref. cbdv202401823-bib-0006] The discovery of GAs in Japan in the 1930s can be linked back to the initiation of research on rice infection associated with specific symptoms, such as overgrowth of stems and insufficient seed production. These symptoms are mainly due to exertions of the fungus Gibberella fujikuroi (also known as Fusarium fujikuroi),[ref. cbdv202401823-bib-0007, ref. cbdv202401823-bib-0008] with the main component called gibberellin. Since then, numerous efforts have been employed to elucidate the biosynthetic origin of GA in fungi and later in plants, ultimately converging on geranylgeranyl diphosphate origin with some differences related to regulating enzymes.[ref. cbdv202401823-bib-0009, ref. cbdv202401823-bib-0010] In both routes, GA12‐aldehyde 4 (Figure 1) is formed and then it is converted to other GAs by the action of a group of specific enzymes.[ref. cbdv202401823-bib-0011, ref. cbdv202401823-bib-0012] Afterward, the mechanism of GAs stimulating growth expression was also investigated and could be referred to as inducing DELLA (aspartic acid–glutamic acid–leucine–leucine–alanine) proteins degradation.[ref. cbdv202401823-bib-0013, ref. cbdv202401823-bib-0014]
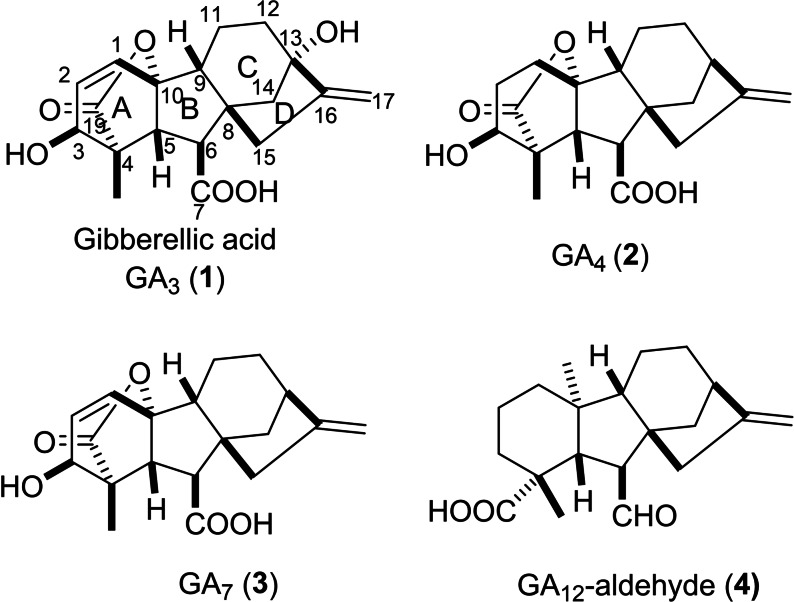
Based on structures and their biosynthetic route, GAs can be classified into two major groups: full diterpenoid skeleton compounds (C20‐GAs) and others with 19 carbon atoms (C19‐GAs such as GA3 1, GA4 2, and GA7 3) (see Figure 1) that response to main activities.15 Gibberellic acid (GA3, 1) is a tetracyclic dihydroxy lactone acid with a perhydrofluorene carbon skeleton with a diversity of functional groups (C1−C2 double bond, C10 γ‐lactone ring and C13‐OH).ref. cbdv202401823-bib-0016 In 1996 Perez et al. suggested the key role of C10 γ‐lactone ring in its biological activity, whereas the stereochemistry of its functional groups has been shown to be responsible for its instability under different conditions.[ref. cbdv202401823-bib-0016, ref. cbdv202401823-bib-0017]
GA3 Commercial Production
Since the discovery of GA3, many reports have highlighted its agro‐industrial applications such as seed germination,[ref. cbdv202401823-bib-0005, ref. cbdv202401823-bib-0018] stem elongation,ref. cbdv202401823-bib-0002 flowering,ref. cbdv202401823-bib-0019 and inclining fruit growth.ref. cbdv202401823-bib-0020 Based on these critical activities, many time‐ and cost‐saving and highly productive methods have been developed to produce it in commercial quantities. Among them, fermentation methods including solid‐state fermentation (SSF),ref. cbdv202401823-bib-0021 semi‐solid‐state fermentation (SSSF)ref. cbdv202401823-bib-0022 and submerged fermentation (SmF),ref. cbdv202401823-bib-0023 have been applied. The highest yield of GA3 was obtained by Oliveira et al. in 2017 using SSF and SSSF methods.ref. cbdv202401823-bib-0024 These methods, in particular SSF, allow the reuse of agricultural and industrial by‐products, that can be used as substrates for fermentation. As shown in Table 1, a wide range of substrates were employed to enhance the fermentation yields.
Table 1: Gibberellic acid production by fermentation using different substrates.
| Microorganism | Method | Substrate | Yield |
|---|---|---|---|
| Fusarium moniliforme | SSF | Citric pulp | 5.9 g/kg25 |
| Fusarium fujikuroi | SSSF | Citric pulp | 4.8 g/kg24 |
| Fusarium fujikuroi | SSF | Coffee husk | 492 mg/kg26 |
| Fusarium fujikuroi | SSF | Wheat bran | 6.8 g/kg27 |
| Fusarium proliferatum | SSF | Pigeon pea pods | 7.8 mg/g28 |
Besides agricultural applications, the structural diversity of GA3 has been attracting the attention of synthetic organic chemists not only from the viewpoint of target‐oriented synthesis, but also from the viewpoint of utilizing a new synthetic methodology developed by themselves. This review article focused on the stereoselective syntheses of agents with remarkable pharmaceutical potential, starting from GA3 (1).
Ring‐Distortion Strategy of GA3
Ring A and Carboxylic Group
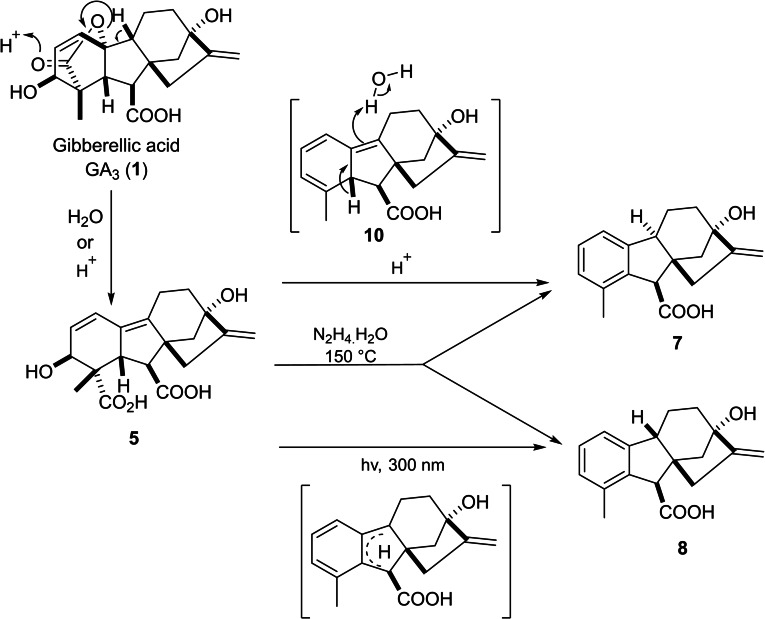
The wide diversity of GA3 functional groups has contributed to its high reactivity and low stability in both acidic and alkaline conditions. The main stereochemical aspect that facilitates the lactone elimination reaction is the oxygen end of the γ‐lactone ring as it is in allylic position to the C1– alkene moiety in a trans‐anti‐periplanar orientation concerning the tertiary hydrogen atom at C9. Subsequently, GA3 goes through lactone elimination, yielding gibberellenic acid even under neutral conditions.[ref. cbdv202401823-bib-0029, ref. cbdv202401823-bib-0030] In general, GA3 usually breaks down in aqueous solutions into gibberellenic acid 5, isogibberellic acid 6, allogibberic acid 7, epiallogibberic acid 8 and dehydroallogibberic acid 9 (shown in Figure 2) in different ratios based on reaction conditions (reaction time, pH and temperature).ref. cbdv202401823-bib-0031
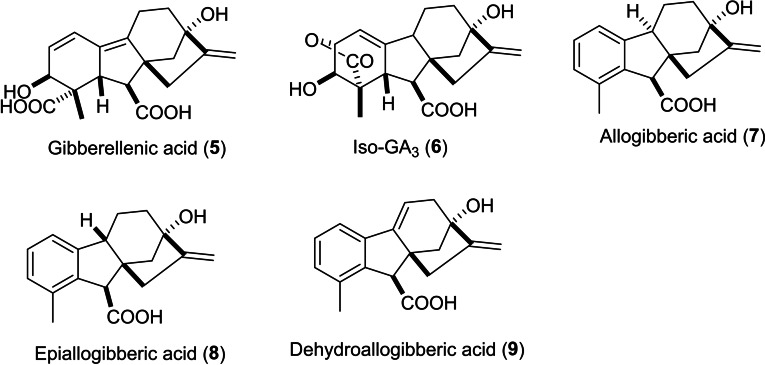
Pryce suggested the potential mechanism of this decomposition.ref. cbdv202401823-bib-0031 In this pathway, gibberellenic acid 5 goes through thermal decomposition into intermediate triene 10, which later yields the major product 9α‐H allogibberic acid 7 (with C9‐C10 trans‐fused ring system)ref. cbdv202401823-bib-0032 through a rearrangement based on solvent exchange. As the C9‐C10 cis‐fused ring system was critical for the synthesis of pharbinilic acid, the preparation of 9β‐H epiallogibberic 8 ref. cbdv202401823-bib-0033 was carried out by either photochemical transformation of the trieneref. cbdv202401823-bib-0034 or heating GA3 in the presence of hydrazine (Scheme 1).ref. cbdv202401823-bib-0035
Rings C and D
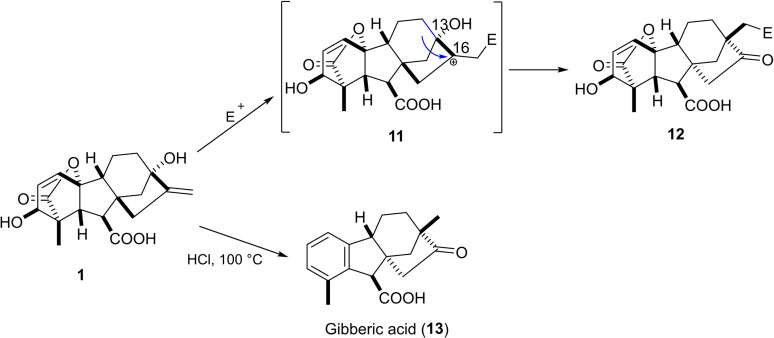
Various rearrangement reactions of rings C and D in GA3 were reported and could be explained by the electrophilic attack on the C16‐C17 alkene moiety and the presence of the allylic C13 hydroxy group.ref. cbdv202401823-bib-0036
Treatment of either GA3 or allogibberic acid with concentrated HCl under reflux led to the formation of ketone derivative 13 (gibberic acid).ref. cbdv202401823-bib-0037 Under these conditions, Wagner–Meerwein rearrangement took place, including the migration of C12‐C13 to C16 with the inversion of the configuration of the C/D rings (Scheme 2).ref. cbdv202401823-bib-0038
In the presence of DDQ, an oxidative rearrangement of C and D rings of methyl allogibberate yielded α‐keto‐ester 15 due to C13‐C16 migration to C12 (Scheme 3).ref. cbdv202401823-bib-0039
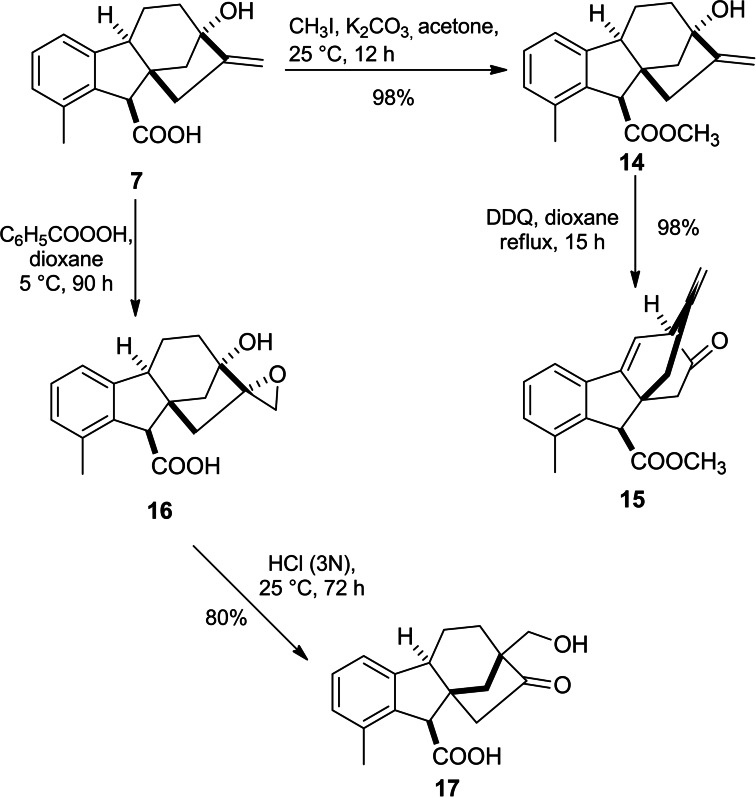
Hydroxymethyl ketone derivative 17 was successfully obtained through HCl‐mediated rearrangement of epoxide 16. In detail, this rearrangement took place with the involvement of a 6‐membered transition state followed by Wagner–Meerwein rearrangement to stabilize the carbonium ion (Scheme 3).ref. cbdv202401823-bib-0040
GA3‐Derived Pharmaceutical Agents
Toxicity Studies
As a consequence of the economic and vital applications of GA3 in the agriculture and industrial sectors, studies regarding its potential effects on mammalian systems have emerged. Kimura et al. intensely studied the subacute and subchronic toxicity of GA3.ref. cbdv202401823-bib-0041 In both cases, it was indicated that GA3 was asymptomatic and it did not induce any histological changes, where the highest tolerable dose in mice was 15 g/kg orally as defined by acute toxicity. Celik et al. investigated the potential changes in the antioxidant defence system in rats induced by GA3.ref. cbdv202401823-bib-0042 A high lipid peroxidation rate was detected in specific tissues accompanied by systematic toxicity in the spleen, stomach, heart, lungs and kidneys. Another analysis on the sub‐chronic toxicity of GA3 on hepatic functions in adult male albino rats was conducted by Hussein et al.ref. cbdv202401823-bib-0043 A remarkable hepatotoxicity was observed and could be explained by GA3‐mediated lipid peroxidation, which increased the MDA levels and thus induced apoptosis and Bcl‐2 overexpression. Troudi et al. also reported the potential neurotoxic effects of GA3 on pregnant rats in the advanced stages (daily 200 ppm GA3 in drinking water).ref. cbdv202401823-bib-0044 A significant neurotoxicity was observed and manifested by GA3‐induced blockage of cerebral and cerebellar AChE and enhanced lipid peroxidation.
Anticancer Activity
Since ent‐kaurene diterpenes are well‐known for their critical and various bioactivities including anticancer, antimicrobial and antifungal properties, many efforts have been employed to find GA3‐based derivatives with promising biological activities. These can be considered as lead compounds for medicinal chemistry. An investigation accomplished by Koehler in 2009 indicated that Gibberellic acid 1 and 9α‐H allogibberic acid 7 could modulate NF‐κB pathway activity and, consequently, they could be utilized for the therapy of several NF‐κB‐related diseases, for instance, cancer and autoimmune diseases.ref. cbdv202401823-bib-0045
Chen et al. introduced a synthetic route to GA3‐based α,β‐unsaturated diketone derivatives (Scheme 4).ref. cbdv202401823-bib-0046 In this case, the SeO2‐mediated allylic oxidation of both the corresponding methyl or benzyl ester 18 and 19 leads to the formation of diols 20 and 21, respectively. Oxidation of the secondary alcohol group in diols 20 and 21 with oxalyl chloride in the presence of triethylamine in DMSO (Swern oxidation) together with an exchange of the tertiary hydroxy group at C13 to chlorine was successfully performed to yield compounds 22 and 23, respectively. All designed compounds were evaluated against a panel of human cancer cell lines (HT29, A549, HepG2 and MKN28). MTT assay revealed that compounds 22 and 23 exhibited the most significant activity against human colon carcinoma cell line HT29 (IC50=2.9 and 4.5 μM, respectively). In the case of human gastric carcinoma cell line MKN28, compound 25a was found to have the most potent inhibitory activity with an IC50 value of 0.8 μM. Moreover, compound 22 showed a complete inhibition towards topoisomerase I at 8 μg mL−1 concentration. Furthermore, as reported by Zhang et al., the in vitro and in vivo antiangiogenic activities of compound 22 downregulated VEGF receptor signaling.ref. cbdv202401823-bib-0047
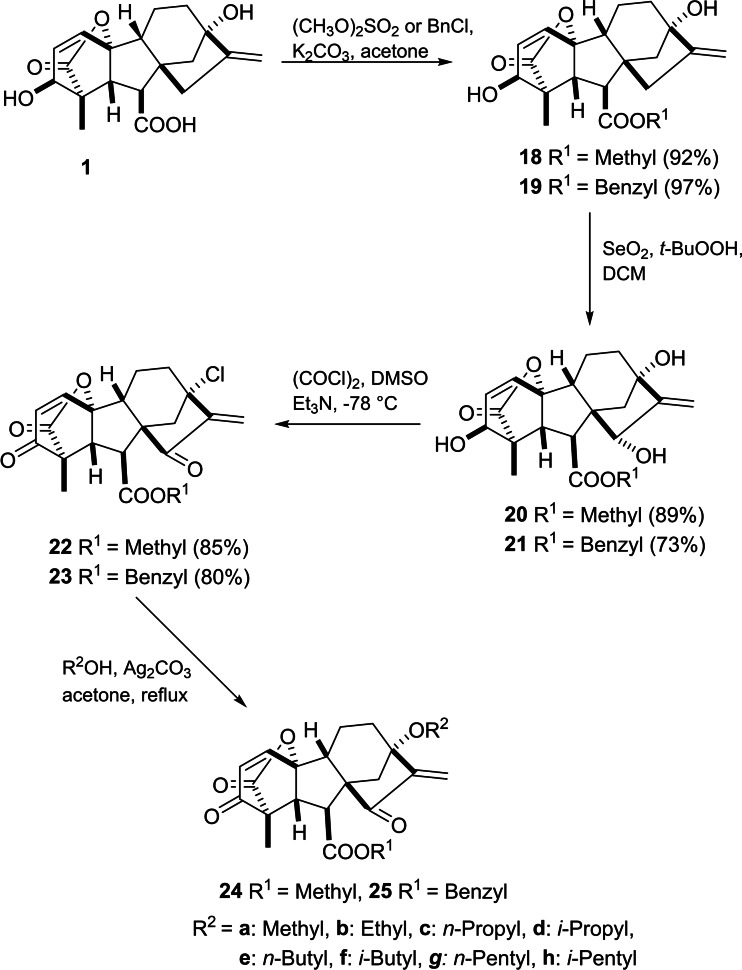
Following this procedure, an additional structural modification was reported by Jingbo et al. to obtain derivatives bearing an ethereal moiety at C13 (see Scheme 4) by converting the chlorine substituent into a variety of alkoxy groups in the presence of silver carbonate. This structural modification led to compound 25h, which is an efficient compound against MKN28 with a low IC50 value (0.21 μM).ref. cbdv202401823-bib-0048
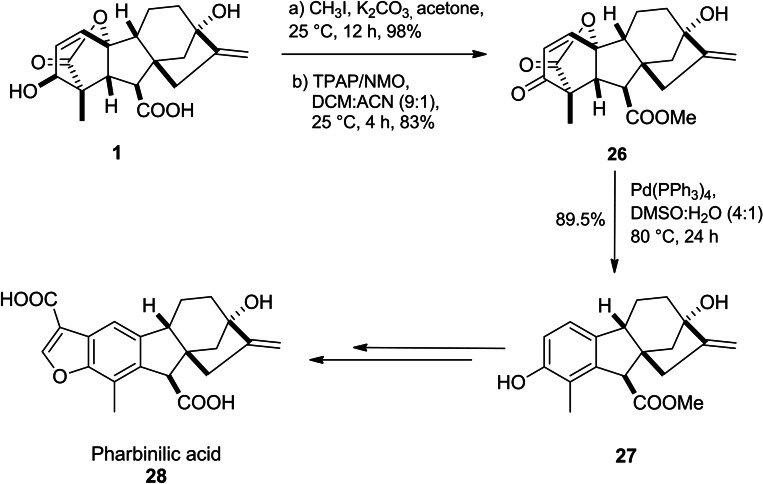
Pharbinillic acid 28, an epiallogibberic acid‐related compound, isolated from Pharbitis nil, exhibited cytotoxicity against several cancer cell lines (A549, SK‐OV‐3, SK‐MEL‐2 and HCT‐15) as well as modulated NF‐κB activity. Annand et al. reported a seven‐step synthetic pathway of pharbinilic acid starting from GA3.ref. cbdv202401823-bib-0036 The key intermediate, hydroxy epiallogibberic methyl ester 27, was obtained through Griffith–Ley oxidation (TPAP/NMO) of GA3 methyl ester to produce enone 26 and then it was subjected to Pd(PPh3)4‐mediated ring A aromatization to form target product 28 (Scheme 5). Further investigations have shown that enone 26 selectively inhibited nuclear translocation of IKKα and displayed selective cytotoxicity towards cancerous cell lines.ref. cbdv202401823-bib-0049
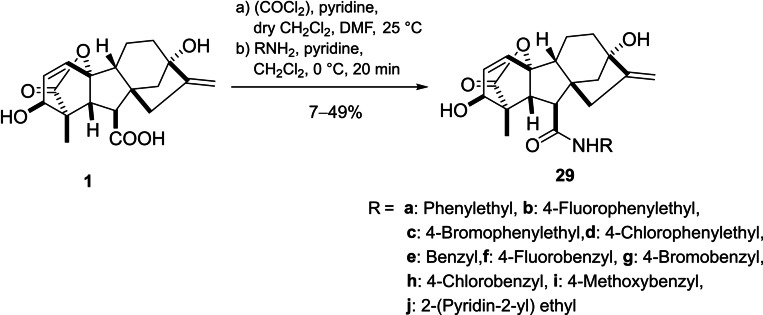
Egbewande et al. reported a parallel‐solution‐phase synthesis of GA3 amides (Scheme 6). The carboxylic group was activated by oxalyl chloride to yield the corresponding acyl chlorides, which were then coupled with the desired amines to form compounds 29. A considerable reduction in free cholesterol uptake in prostate cancer cells was observed upon treatment with compounds 29b‐c, 29g‐h and 29j.ref. cbdv202401823-bib-0050
A series of hydroxy epiallogibberic acids coupled with 1,2,3‐triazole bearing an α,β‐unsaturated ketone moiety was synthesized by Wu et al. (Scheme 7). In this protocol, methyl hydroxy epiallogibberate 27 was obtained by (Dess–Martin periodinane) DMP‐mediated oxidation of GA3 methyl ester. Phenol protection with TBSCl followed by allylic oxidation mediated with SeO2/t‐BuOOH and subsequent reduction of ester group with lithium aluminium hydride (LAH)‐based yielded compound 32. Next the primary alcohol group was tosylated and then it was treated with sodium azide to provide azide 33. The desired triazoles were obtained from compound 33 by a three‐step sequence including CuAAC azide–alkyne cycloaddition, Dess–Martin oxidation and deprotection. The in‐vitro evaluation confirmed the essential role of the α,β‐unsaturated ketone moiety in cytotoxicity against cancer cell lines, where compounds 34c, 34d and 34e exhibited potent cytotoxicity, which is 8 to 20 times stronger than that of reference cisplatin. Compound 34d was found to trigger cell cycle arrest in the S phase and promoted apoptosis in SMMC‐7721 cell lines.ref. cbdv202401823-bib-0051
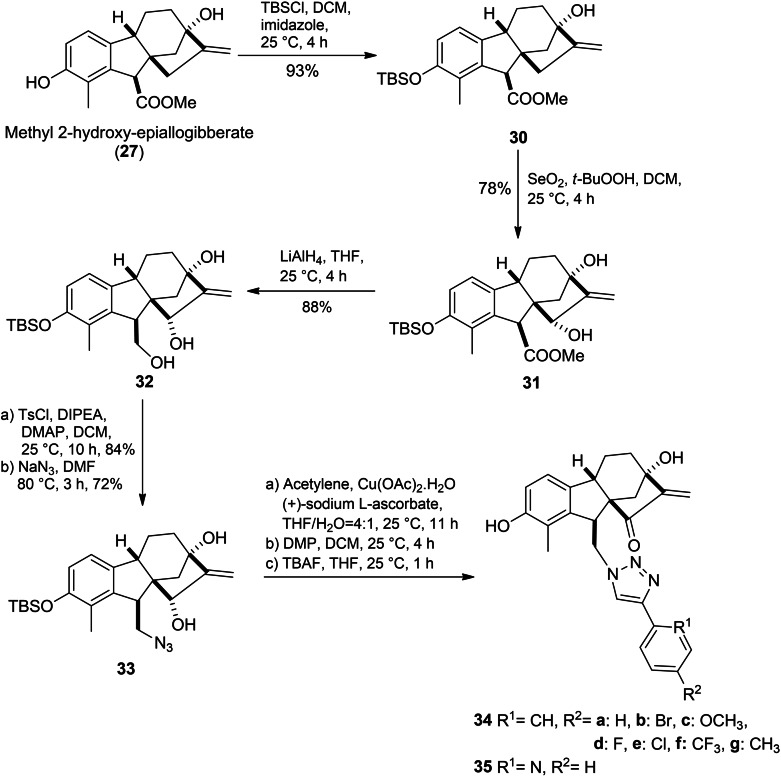
In order to further investigate potential GA3‐based antitumor derivatives, Zhu et al. designed and synthesized a library of allogibberic acid‐substituted benzyl esters 36 and their keto derivatives 37 through DDQ‐mediated rearrangement of the corresponding ester (Scheme 8A).
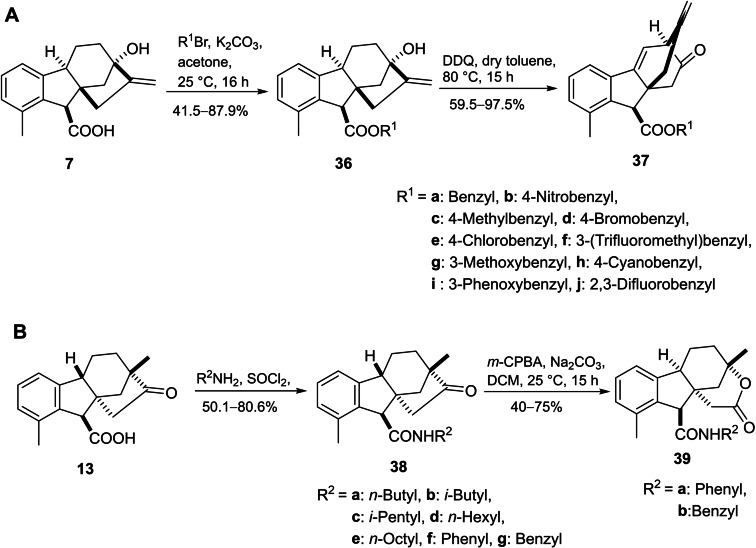
On the other hand, a series of gibberic carboxamides was also prepared by treatment of amides 38 under Baeyer–Villiger oxidation conditions (m‐CPBA) leading to the formation of corresponding lactones 39 (Scheme 8B). The cytotoxicity of the designed compounds varied among the cancer cell lines. Within the ester derivatives, compounds featuring the meta‐substituted benzyl group displayed greater activity than those with a para‐substituted benzyl substituent with more selectivity towards HL‐60. Considering the amides, compounds with linear saturated substituents showed the best activity, especially compound 38e, which had strong activity against all tested cell lines (HL‐60, MCF‐7, SW480 and NCI−H226).ref. cbdv202401823-bib-0052
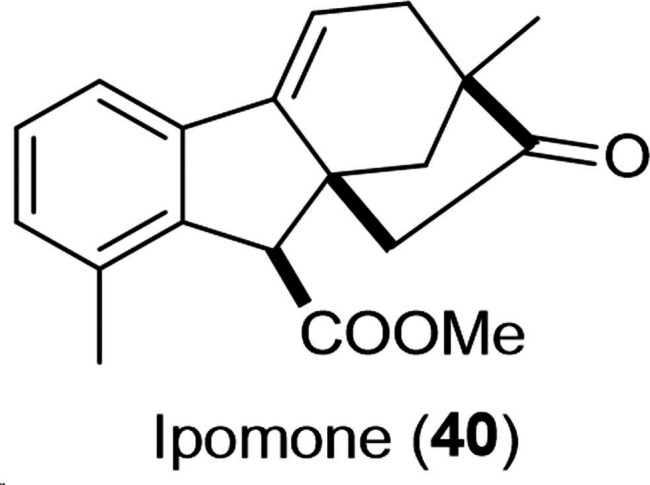
Recently, a new gibberic acid derivative, the methyl ester of dehydrogibberic acid (ipomone 40, shown in Figure 3), was successfully isolated and identified by Goel et al. from acidified hydroalcoholic extract of Ipomoea nil seeds.ref. cbdv202401823-bib-0053 The suggested mechanism of ipomone formation was the acid‐mediated rearrangement of dehydroallogibberic acid. Despite the low cytotoxicity of ipomone against cancer cell lines (IC50=34–86 μM), the western blot analysis indicated a dose‐dependent decline in the expression of caspase‐3 and PARP‐1 in lung cancer A549 cells approving that ipomone could stimulate apoptosis and autophagy in these cells.
Khdar et al. introduced the synthetic route of a library of aminodiols based on allogibberic acid (Scheme 9).ref. cbdv202401823-bib-0054 The methodology involved the esterification of allogibberic acid followed by stereospecific synthesis of epoxide and subsequent ring opening with various primary and secondary amines catalyzed by lithium perchlorate to produce aminodiols 43a.
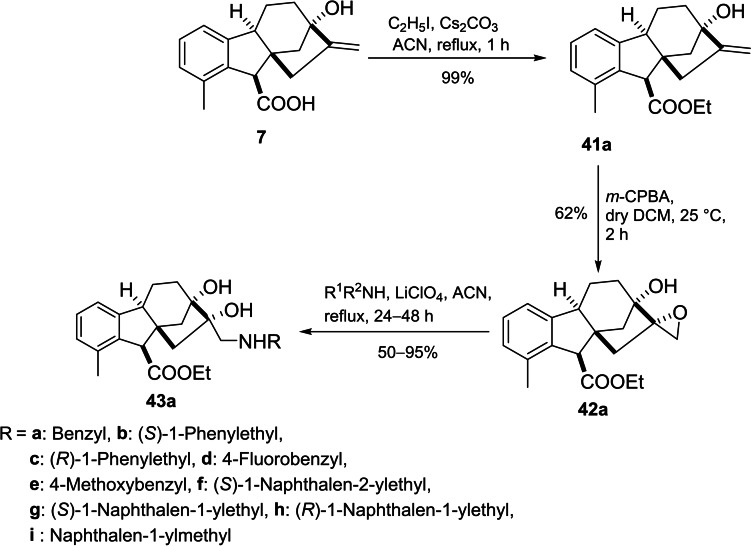
Considering another possibility, the isomerization of the ester group of 41a under alkaline conditions followed by epoxidation and then aminolysis of the resulting epoxide led to the formation of aminodiols 43b (Scheme 10).
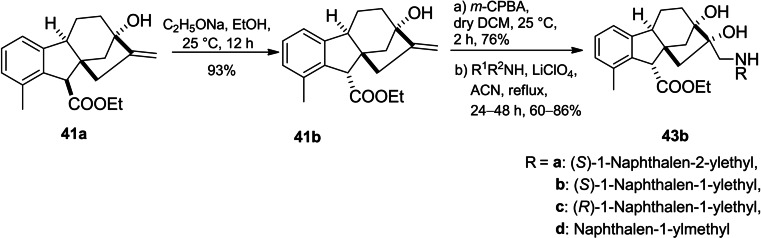
The MTT assay demonstrated that secondary aminodiols (especially naphtyl ethylamine derivatives with IC50=4–7 μM) possessed the most important antiproliferative activity against a panel of cancer cell lines (A2780, HeLa, SiHa and MDA‐MB‐231). In contrast, the highest cancer selectivity was shown by compound 43ag. Moreover, the importance of the R configuration of the ester group for antiproliferative activity was also indicated.ref. cbdv202401823-bib-0054
Continued interest in the modification of allogibberic acid, compound 44 was successfully prepared by Khdar et al. through BF3 ⋅ OEt2‐mediated rearrangement of epoxide 42 (Scheme 11).ref. cbdv202401823-bib-0055 This step produced the groundwork to design a library of 1,3‐aminoalcohols 46 by either reductive amination of the hydroxymethyl ketone 44 or reductive alkylation of primary aminoalcohol 45.
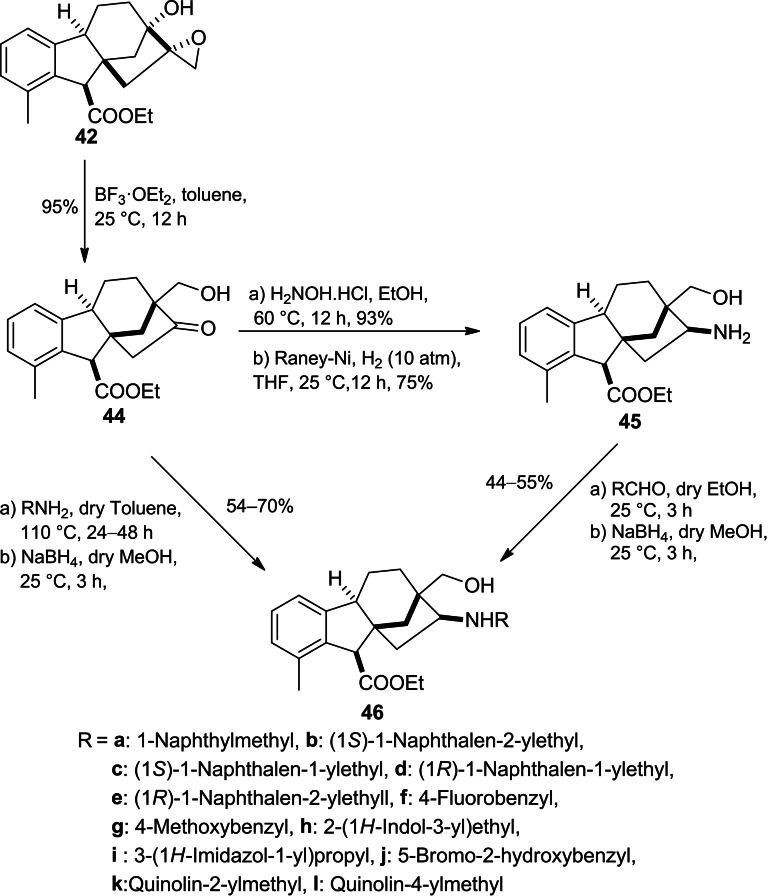
In addition, the corresponding regioisomers 49 were successfully synthesized in three steps including reduction of mesyl derivative 44 followed by azidation of the corresponding aldohol 47 and subsequent Pd‐catalysed hydrogenolysis of azide derivative 48. Furthermore, the exchange of the mesyl function in mesylate 47 with various primary amines or reductive alkylation of primary aminoalcohol 49 yielded the desired 1,3‐aminoalcohols 50 (Scheme 12).
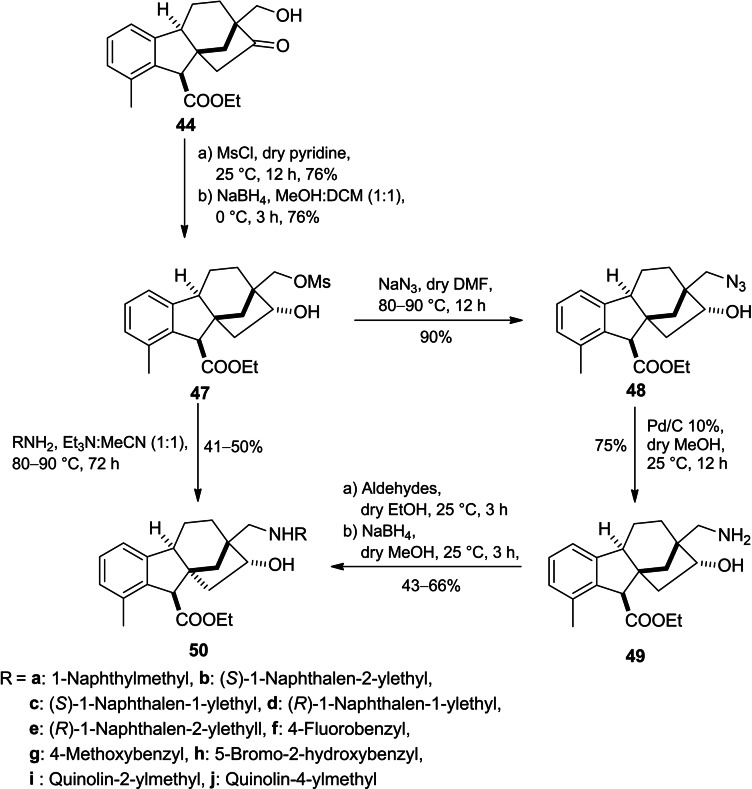
According to the MTT assay, a marked and substantial variation was discerned in the antiproliferative activity exhibited by the regioisomers. Substituted benzyl derivatives (50f, 50g and 50h) and indolyl derivative 46h showed potent activity with a cell line‐dependent effect. Moreover, they also displayed modest cancer selectivity with calculated IC50 values higher on NIH/3T3 fibroblast cells compared to tested cancer cell lines.
Based on the aforementioned structural modifications, the structural aspects responsible for the anticancer activity of gibberellic acid and allogibberic acid was summarized as shown in Figures 4 and 5. It is obvious that the presence of diketone at positions 3 and 15 of methyl or benzyl gibberrate along with the substitution of OH at position 13 with Cl, OMe, or O‐(i‐pentyl) is critical for promising anticancer activity. The combination of ring A aromatization and benzyl ester functionality gave rise to new promising derivatives. The introduction of the OH group at position 2 on the aromatic ring A and a ketone group at position 15, as well as the transformation of the carboxyl group into 4‐substituted 1,2,3‐triazole, showed a crucial role in the activity.
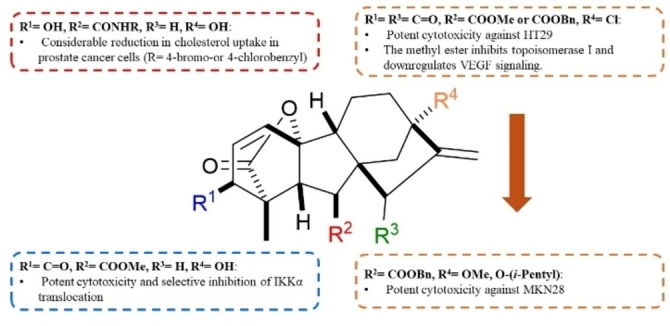
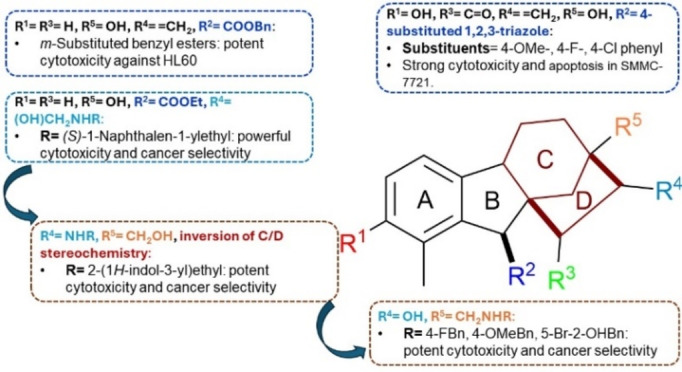
On the other hand, bearing ethyl allogibberate with 3‐amino‐1,2‐diol motif or with 1,3‐aminoalcohl moiety alongside inversion of rings C/D stereochemistry also displayed a significant influence on the activity.
Antimicrobial Activity
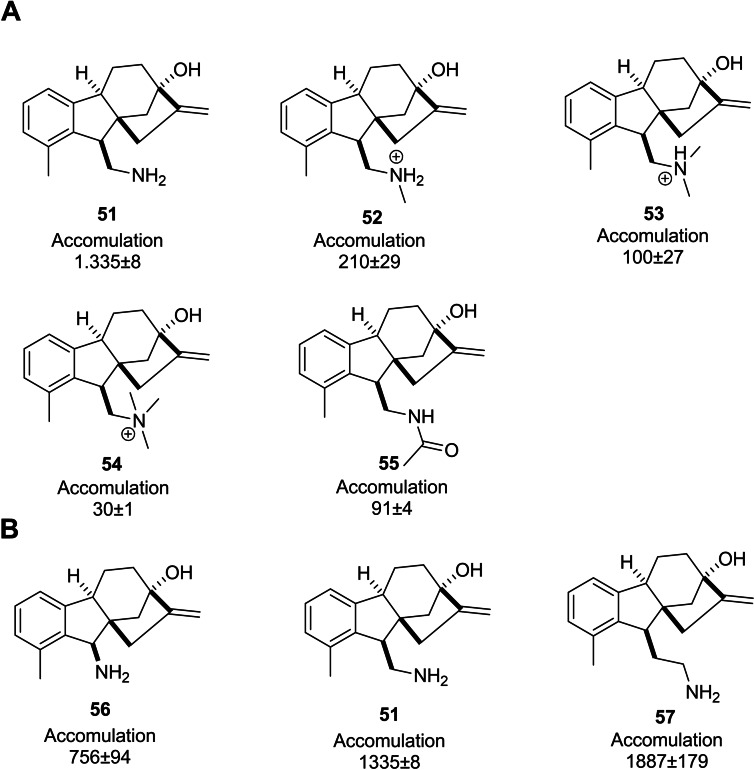
Richter et al. conducted a comprehensive investigation into the accumulation of a wide variety of small molecules in Gram‐negative bacteria, especially E. coli, aiming at highlighting the potential factors that affect accumulating compounds. This study established the high accumulation of primary amine derivatives of allogibberic acid in E. coli MG1655. Its monomethyl, dimethyl, trimethyl, and acetylated derivatives (Figure 6A), in turn, exhibited a dramatic decrease in accumulation. It was also concluded that when the primary amine functionality was more systematically separated from the condensed ring system, the compounds had more accumulation (Figure 6B).ref. cbdv202401823-bib-0056
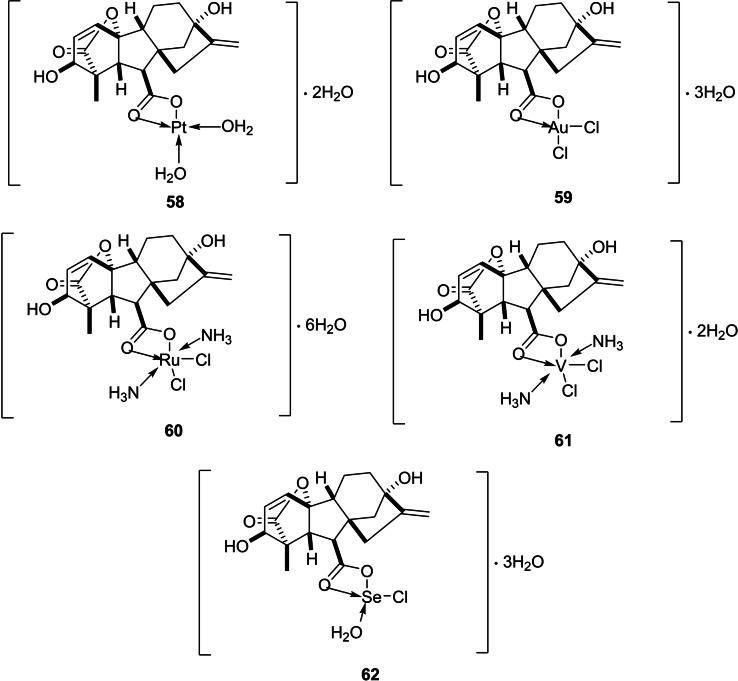
Another study was undertaken by El‐Sayed et al. with the objective of preparing and characterizing a group of GA3 complexes with Pt(II), Au(III), Ru(III), V(III), and Se(IV) ions (Figure 7). The study included biological evaluation as well. Reactions were conducted under neutral pH, in which the anionic form GA3 − was dominant. The desired complexes were obtained by the reaction of the ions with GA3 − at 65 °C. The Kirby‐Bauer disc diffusion method indicated that Au(III) and Se(IV) complexes displayed integral antimicrobial activity, particularly against E. coli and S. aureus. The cytotoxicity of GA3 against HepG2 and MCF‐7 cell lines, in turn, was remarkably increased when it was combined with Pt(II) and Au(III) ions.ref. cbdv202401823-bib-0057
In 2021, Toner et al. conducted an investigation on the antimicrobial effects of several diterpene phytohormones (gibberellins) against a group of phytopathogens and clinical pathogens.ref. cbdv202401823-bib-0058 The obtained results showed that GA3 1 failed to inhibit the growth of bacterial organisms of clinical significance to humans, while GA4 2, a gibberellin lacking C1−C2 double bond, and C13−OH, displayed a strong inhibition against all the tested clinical bacteria including Gram‐negative and Gram‐positive ones.
These results emphasize the importance of both the transformation of the carboxylic group into primary amine functionality and the formation of carboxylate complexes with several ions for antimicrobial activity.
Anti‐Inflammatory Activity
Since the ability of gibberellic acid 1 to modulate NF‐κB pathway activity was declared by Koehler in 2009,ref. cbdv202401823-bib-0045 the possible use of gibberellic acid to modulate inflammation response has been studied. Commencing from the standpoint that gibberellic acid can trigger A20‐like zinc finger proteins in plants,ref. cbdv202401823-bib-0059 Reihill et al. investigated the A20‐mediated anti‐inflammatory effect of gibberellic acid on airway epithelial cells.ref. cbdv202401823-bib-0060 The results showed that the higher levels of A20 mRNA and protein together with a considerable decrease in IL‐6 and IL‐8 release were observed in epithelial cells treated with gibberellic acid. This effect was regulated by GA3‐mediated induction of zinc finger protein A20 resulting in an incline in 1κBα levels and consequent decrease in NF‐κB expression. These findings suggest the possible use of gibberellic acid for chronic inflammation treatment. Recently, Xu et al. reported the role of gibberellic acid in reducing sepsis‐based neuroinflammation.ref. cbdv202401823-bib-0061 It was found that the incubation of microglial cells (the primary immune cells in the brain) with gibberellic acid resulted in overexpression of ZBTB16, an NF‐κB regulator, diminishing the LPS (Lipopolysaccharide)‐stimulated microglial M1 activation releasing anti‐inflammatory activity.
These investigations provide a strong foundation for the development of GA3‐based derivatives with promising anti‐inflammatory activity.
Other Activities
Besides the previously mentioned interesting bioactivities of gibberellic acid, other biological properties were reported in the literature. Kasamatsu et al. declared the successful use of gibberellic acid for the regeneration of salivary glands starting from adipose‐derived stem cells, where high levels of α‐amylase were detected in stem cells after 7 days of occupation with gibberellic acid.ref. cbdv202401823-bib-0062
On the other hand, the gibberellic acid hormonal effects on plants, including stem and root elongation and enhancement of flowering and fruit production, were extensively studied in the literature. The main mechanism that accounts for its actions is inducing DELLA protein degradation by binding GA3 with the DELLA receptor leading to conformational changes and subsequent degradation.[ref. cbdv202401823-bib-0008, ref. cbdv202401823-bib-0019, ref. cbdv202401823-bib-0063, ref. cbdv202401823-bib-0064, ref. cbdv202401823-bib-0065, ref. cbdv202401823-bib-0066]
Summary and Outlook
In conclusion, this review provides insights into the structural features underlying the potential biological activities of gibberellic acid and its derivatives, shedding light on the most promising derivatives with anticancer activity. Numerous structural modifications have been reported and a wide range of compounds bearing antiproliferative activity with diverse potencies and selectivities was reported. By critically evaluating existing pieces of information in the literature, it is proven that several functionalities, for instance, α,β‐unsaturated ketone, 1,2,3‐triazoles, meta‐substituted benzyl derivatives, amides, aminoalcohols and aminodiols, are crucial to the anticancer activity.
Future research should explore interdisciplinary methods to further elucidate the possible mechanisms of actions and potential targets. This could lead to more efficient structural modifications based on compound/target interactions. Moreover, a combination of the aforementioned structural modifications including aminodiols or aminoalcohols of meta‐substituted benzyl allogibberic acid esters, derivatives with substituted ring A and aminoalcohols, aminodiols or triazoles of pharbanillic acid, will allow additional evaluations. In the meantime, these derivatives should also be scanned for further biological activities (antifungal, antimicrobial and anti‐inflammatory activities).
What might the future application of GA3 look like? Owing to their low cost and structural diversity, GA3 could be extensively utilized by synthetic chemists in the total synthesis of natural products.ref. cbdv202401823-bib-0067 The functional groups make a variety of stereoselective transformations of GA3 possible, such as acylation or alkylation at the α‐position, while the double bond can be transformed by catalytic reduction, epoxidation, dihydroxylation, oxidative cleavage and ring contraction, to name a few possibilities.ref. cbdv202401823-bib-0068 In addition, the ent‐kaurene framework, having multiple asymmetric centres, will certainly play an important role as a promising starting material for natural product syntheses in the future.ref. cbdv202401823-bib-0069
Author Contributions
Z.A.K.: Writing – original draft preparation; T.M.L.: writing – review and editing; Z.S: supervision and editing, funding acquisition. All authors have read and agreed to the published version of the manuscript.
Conflict of Interests
The authors declare no conflict of interest.
Biographical Information
Zein Alabdeen Khdar was born in Latakia, Syria in 1990. He received his bachelor’s degree in pharmacy and pharmaceutical chemistry at Tishreen University, Syria in 2013. Then he obtained his MSc degree in drug design and quality control at Tishreen University in 2019. Since 2021, he has started his PhD in Pharmaceutical Sciences at the University of Szeged, Hungary. His research interests vary from the stereoselective synthesis of diterpene‐based aminoalcohols, aminodiols, and aminotriols to the In‐silico studies, including docking and ADMET analysis.
Biographical Information
Tam Minh Le was born in Vietnam in 1989. He received his MSc degree in Chemistry in Vietnam (2015) and his PhD in Pharmaceutical Science at the University of Szeged, Hungary (2020), where he continues to his research as a member of Hungarian Academy of Sciences in Institute of Pharmaceutical Chemistry until now. His research interests range from stereoselective synthesis of monoterpene‐based amino acid derivatives to the transformation of natural products, especially in the terpenoid field.
Biographical Information
Zsolt Szakonyi was born in 1969 in Szombathely, Hungary. He received his Master’s degree in Pharmacy in 1992. He undertook his PhD in synthetic organic chemistry in the group of Prof. Ferenc Fülöp and held his Ph.D. Degree at the University of Szeged (Hungary) in 1998. He joined the research group of Prof. Norbert De Kimpe at the University of Ghent, Belgium in period of 1999–2000 as a postdoctoral Research Fellow by FWO. He became full professor at the Institute of Pharmaceutical Chemistry, University of Szeged in 2019. His research interests centre on stereoselective synthesis of monoterpene‐ and diterpene‐based bi‐ and tridentate building blocks, beta‐amino acid derivatives, amino diols and saturated 1,3‐heterocycles with a special focus on homogeneous stereoselective catalysis.
References
- 1 B. B. Stowe , T. Yamaki , Annu. Rev. Plant Physiol. 1957, 8, 181–216.
- 2 J. Dayan , N. Voronin , F. Gong , T. Sun , P. Hedden , H. Fromm , R. Aloni , Plant Cell 2012, 24, 66–79.22253226
- 3 R. R. Sharma , R. Singh , Sci. Hortic. 2009, 119, 430–433.
- 4 N. Muñoz-Fambuena , C. Mesejo , M. C. González-Mas , D. J. Iglesias , E. Primo-Millo , M. Agustí , J. Plant Growth Regul. 2012, 31, 529–536.
- 5 S.-Y. Chen , S.-R. Kuo , C.-T. Chien , Tree Physiol. 2008, 28, 1431–1439.18595855
- 6 W. E. Finch-Savage , G. Leubner-Metzger , New Phytol. 2006, 171, 501–523.16866955
- 7 B. M. Bashyal , Indian Phytopathol. 2018, 71, 485–494.
- 8 L. N. Mander , Chem. Rev. 1992, 92, 573–612.
- 9 P. Hedden , A. L. Phillips , Trends Plant Sci. 2000, 5, 523–530.11120474
- 10 B. Tudzynski , Appl. Microbiol. Biotechnol. 2005, 66, 597–611.15578178
- 11 S. Yamaguchi , Annu. Rev. Plant Biol. 2008, 59, 225–251.18173378
- 12 C. Bömke , B. Tudzynski , Phytochemistry 2009, 70, 1876–1893.19560174
- 13 J. Peng , P. Carol , D. E. Richards , K. E. King , R. J. Cowling , G. P. Murphy , N. P. Harberd , Genes Dev. 1997, 11, 3194–3205.9389651
- 14 A. Dill , H.-S. Jung , T. Sun , Proc. Natl. Acad. Sci. 2001, 98, 14162–14167.11717468
- 15 D. Tarkowská , O. Novák , K. Floková , P. Tarkowski , V. Turečková , J. Grúz , J. Rolčík , M. Strnad , Planta 2014, 240, 55–76.24677098
- 16 B. E. Cross , J. Chem. Soc. Resumed 1954, 4670–4676.
- 17 F. J. Pérez , A. Vecchiola , M. Pinto , E. Agosin , Phytochemistry 1996, 41, 675–679.
- 18 Z. Li , Y. Gao , Y. Zhang , C. Lin , D. Gong , Y. Guan , J. Hu , Front. Plant Sci. 2018, 9, 1279.30356911
- 19 B. Dong , Y. Deng , H. Wang , R. Gao , G. Stephen , S. Chen , J. Jiang , F. Chen , Int. J. Mol. Sci. 2017, 18, 1259.28604637
- 20 L. Mariotti , P. Picciarelli , L. Lombardi , N. Ceccarelli , J. Plant Growth Regul. 2011, 30, 405–415.
- 21 R. R. Singhania , A. K. Patel , C. R. Soccol , A. Pandey , Biochem. Eng. J. 2009, 44, 13–18.
- 22 S. Selvaraj , V. R. Murty , J. Biotechnol. 2017, 12, 39–48.
- 23 E. M. Escamilla , S. L. Dendooven , I. P. Magaña , R. Parra S , M. De la Torre , J. Biotechnol. 2000, 76, 147–155.10656329
- 24 J. de Oliveira , C. Rodrigues , L. P. S. Vandenberghe , M. C. Câmara , N. Libardi , C. R. Soccol , BioMed Res. Int. 2017, 2017, e5191046.
- 25 C. Rodrigues , L. P. S. Vandenberghe , J. Teodoro , J. F. Oss , A. Pandey and C. R. Soccol , Braz. Arch. Biol. Technol. 2009, 52, 181–188.
- 26 C. M. M. Machado , C. R. Soccol , B. H. de Oliveira , A. Pandey , Appl. Biochem. Biotechnol. 2002, 102, 179–191.12396121
- 27 E. Agosin , M. Maureira , V. Biffani , F. Pérez , in Advances in Solid State Fermentation (Eds: S. Roussos , B. K. Lonsane , M. Raimbault , G. Viniegra-Gonzalez ), Springer Netherlands, Dordrecht 1997, 355–366.
- 28 D. Satpute , V. Sharma , K. Murarkar , M. Bhotmange , D. Dharmadhikari , Int. J. Environ. Pollut. 2010, 43, 201–213.
- 29 L. Kutschabsky , G. Adam , J. Chem. Soc. Perkin 1 1983, 1653.
- 30 J. R. Bearder , P. S. Kirkwood , J. MacMillan , J. Chem. Soc. Perkin 1 1981, 672.
- 31 R. J. Pryce , J. Chem. Soc. Perkin 1 1974, 1179–1184.
- 32 J. F. Grove , T. P. C. Mulholland , J. Chem. Soc. Resumed 1960, 3007–3022.
- 33 T. P. C. Mulholland , J. Chem. Soc. Resumed 1958, 2693–2701.
- 34 H. K. Al-Ekabi , G. A. W. Derwish , Can. J. Chem. 1984, 62, 1996–1998.
- 35 P. W. Brian , J. F. Grove , J. MacMillan , in Fortschritte der Chemie organischer Naturstoffe/Progress in the Chemistry of Organic Natural Products/Progrés Dans la Chimie des Substances Organiques Naturelles, (Ed: L. Zechmeister ), Springer, Vienna 1960, 350–433.
- 36 J. R. Annand , P. A. Bruno , A. K. Mapp , C. S. Schindler , Chem. Commun. 2015, 51, 8990–8993.
- 37 B. E. Cross , J. F. Grove , J. MacMillan , T. P. C. Mulholland , J. Chem. Soc. Resumed 1958, 2520–2536.
- 38 J. F. Grove , J. MacMillan , T. P. C. Mulholland , W. B. Turner , J. Chem. Soc. Resumed 1960, 3049–3057.
- 39 B. E. Cross , R. E. Markwell , J. Chem. Soc. Perkin 1 1973, 1476–1487.
- 40 K. Schreiber , G. Schneider , G. Sembdner , Tetrahedron 1968, 24, 73–78.
- 41 E. T. Kimura , P. R. Young , K. Staniszewski , J. Am. Pharm. Assoc. Sci. Ed 1959, 48, 127–129.
- 42 I. Celik , Y. Tuluce , I. Isik , J. Enzyme Inhib. Med. Chem. 2007, 22, 219–226.17518349
- 43 W. F. Hussein , F. Y. Farahat , M. A. Abass , A. S. Shehata , Life Science Journal 2011, 8, 373–383.
- 44 A. Troudi , H. Bouaziz , N. Soudani , I. Ben Amara , T. Boudawara , H. Touzani , B. Lyoussi , N. Zeghal , Exp. Toxicol. Pathol. Off. J. Ges. Toxikol. Pathol. 2012, 64, 583–590.
- 45World Intellectual Property Organization, WO2010062372A2, 2010.
- 46 J. Chen , Z. Sun , Y. Zhang , X. Zeng , C. Qing , J. Liu , L. Li , H. Zhang , Bioorg. Med. Chem. Lett. 2009, 19, 5496–5499.19679470
- 47 Y. Zhang , H. Zhang , J. Chen , H. Zhao , X. Zeng , H. Zhang , C. Qing , Invest. New Drugs 2012, 30, 8–16.20711631
- 48 J. Chen , Z. Sun , Y. Zhang , J. Chen , P. Li , X. Zeng , B. Yin , Y. Xie , C. Qing , H. Zhang , Chin. J. Org. Chem. 2014, 34, 788.
- 49 J. R. Annand , A. R. Henderson , K. S. Cole , A. J. Maurais , J. Becerra , Y. Liu , E. Weerapana , A. N. Koehler , A. K. Mapp , C. S. Schindler , ACS Med. Chem. Lett. 2020, 11, 1913–1918.33062173
- 50 F. A. Egbewande , M. C. Sadowski , C. Levrier , K. D. Tousignant , J. M. White , M. J. Coster , C. C. Nelson , R. A. Davis , J. Nat. Prod. 2018, 81, 838–845.29474071
- 51 M.-J. Wu , D.-M. Wu , J.-B. Chen , J.-F. Zhao , L. Gong , Y.-X. Gong , Y. Li , X.-D. Yang , H. Zhang , Bioorg. Med. Chem. Lett. 2018, 28, 2543–2549.29884535
- 52 S. Zhu , F. Luo , P.-H. Sun , Med. Chem. Res. 2020, 29, 1341–1354.
- 53 B. Goel , E. Chatterjee , B. Dey , N. Tripathi , N. Bhardwaj , A. Khattri , S. K. Guru , S. K. Jain , ACS Omega 2021, 6, 8253–8260.33817484
- 54 Z. A. Khdar , T. M. Le , Z. Schelz , I. Zupkó , Z. Szakonyi , Int. J. Mol. Sci. 2022, 23, 10366.36142293
- 55 Z. Alabdeen Khdar , T. Minh Le , Z. Schelz , I. Zupkó , Z. Szakonyi , RSC Med. Chem. DOI: 10.1039/D3MD00665D.
- 56 M. F. Richter , B. S. Drown , A. P. Riley , A. Garcia , T. Shirai , R. L. Svec , P. J. Hergenrother , Nature 2017, 545, 299–304.28489819
- 57 M. Y. El-Sayed , H. Fetooh , M. S. Refat , H. H. Eldaroti , A. M. A. Adam , H. A. Saad , J. Mol. Liq. 2019, 296, 111895.
- 58 P. Toner , D. Nelson , J. R. Rao , M. Ennis , J. E. Moore , B. Schock , Access Microbiol. DOI: 10.1099/acmi.0.000278.
- 59 Y. Liu , Y. Xu , J. Xiao , Q. Ma , D. Li , Z. Xue , K. Chong , J. Plant Physiol. 2011, 168, 1098–1105.21316795
- 60 J. A. Reihill , B. Malcomson , A. Bertelsen , S. Cheung , A. Czerwiec , R. Barsden , J. S. Elborn , H. Dürkop , B. Hirsch , M. Ennis , C. Kelly , B. C. Schock , Br. J. Pharmacol. 2016, 173, 778–789.26013851
- 61 H. Xu , Y.-Y. Lin , J.-J. Yu , N. Zhang , J.-M. Hu , J.-S. Qu , C.-M. Yuan , D.-Q. Chen , M. Liang , H. Cai , K. Zeng , Eur. J. Pharmacol. 2024, 976, 176665.38797312
- 62 A. Kasamatsu , M. Iyoda , K. Usukura , Y. Sakamoto , K. Ogawara , M. Shiiba , H. Tanzawa , K. Uzawa , Int. J. Mol. Med. 2012, 30, 243–247.22641429
- 63 S. H. Shah , S. Islam , F. Mohammad , M. H. Siddiqui , J. Plant Growth Regul. 2023, 42, 7352–7373.
- 64 A. Miceli , A. Moncada , L. Sabatino , F. Vetrano , Agronomy 2019, 9, 382.
- 65 P. W. Brian , J. R. Soc. Arts 1958, 106, 425–441.
- 66 P. V. Ammirato , Plant Physiol. 1977, 59, 579–586.16659897
- 67 R. A. Shenvi , ACS Cent. Sci. 2024, 10, 519–528.38559299
- 68 C. Ding , Y. Ding , H. Chen , J. Zhou , Stud. Nat. Prod. Chem. 2017, 54, 141–197, DOI: 10.1016/B978-0-444-63929-5.00005-X.
- 69 A. Kanwal , M. Bilal , N. Rasool , M. Zubair , S. A. A. Shah , Z. A. Zakaria , Pharmaceuticals (Basel) 2022, 15, 1392.36422521
