Cannabinoid inhibition of mechanosensitive K+ channels
Abstract
Cannabidiol (CBD) is a prominent non-psychoactive small molecule produced by cannabis plants used clinically as an antiepileptic. Here, we show CBD and other cannabinoids are potent inhibitors of mechanosensitive two-pore domain K+ (K2P) channels, including TRAAK and TREK-1 that contribute to spike propagation in myelinated axons. Five TRAAK mutations that cause epilepsy or the neurodevelopmental syndrome FHEIG (facial dysmorphism, hypertrichosis, epilepsy, intellectual/developmental delay, and gingival overgrowth) retain sensitivity to cannabinoid inhibition. A cryo-EM structure reveals CBD binds in the intracellular cavity of TREK-1 to sterically block ion conduction. These results show that cannabinoids and endogenous lipids compete for a common binding site to inhibit channel activity, identify mechanosensitive K2Ps as potential physiological targets of CBD, and suggest cannabinoids could counter gain-of-function in TRAAK channelopathies.
License: CC BY 4.0 This work is licensed under a Creative Commons Attribution 4.0 International License, which allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use.
Article links: DOI: 10.1101/2024.12.09.627564 | PubMed: 39713384 | PMC: PMC11661169
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (8.0 MB)
Introduction
Phytocannabinoids are hydrophobic phenolic compounds produced by cannabis plants. Over one hundred phytocannabinoids have been identified, with delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD) among the most prevalent 1,2. THC is the primary psychoactive cannabinoid that underlies the euphoria-inducing effects of cannabis 1. The effects of THC are mainly mediated by partial agonism of CB1 and CB2 G-protein coupled receptors (EC50 ~ 50nM) 1. CBD is the predominant non-psychoactive cannabinoid and is an effective antiepileptic approved by regulatory agencies in multiple jurisdictions for the treatment of seizures in Lennox-Gastaut syndrome, Dravet’s syndrome, and tuberous sclerosis complex 3–8. In contrast to THC, the molecular basis for the antiepileptic effects of CBD is unknown 1,2. CBD is not a CB1 or CB2 receptor agonist; rather, it negatively modulates CB1 and GPR55 receptors (IC50 ~500 nM)9–11. Like other classic anticonvulsants, CBD inhibits voltage-gated sodium channels (IC50 ~2–10 μM) 12,13. However, the relevance of these targets is debated2, as CBD’s antiepileptic effects are observed in slice recordings at concentrations ≤100 nM and in treated patients at ~50–100 nM (as measured in plasma or estimated in brain tissue). CBD also modulates other ion channels, activating TRPV1–4 (EC50 ~1–4 μM), TRPA1 (EC50 110 nM) 14,15, and Kv7.2/7.3 (EC50 200 nM) 16,17, and inhibiting BK (IC50 280 nM) 18, but the importance of these channels to CBD pharmacology is similarly unverified.
TRAAK (TWIK-related arachidonic acid-activated K+ channel) is a mechanosensitive potassium-selective ion channel belonging to the two-pore domain (K2P) K+ ion channel family. TRAAK is localized to axon initial segments 19,20 and nodes of Ranvier 21,22, where it controls the resting membrane potential and facilitates action potential repolarization, thereby enabling high frequency spiking. Gain-of-function TRAAK mutations cause epileptic and neurodevelopmental disorders in humans, with four reported variants in FHEIG (facial dimorphism, hypertrichosis, epilepsy, intellectual disability, and gingival outgrowth) syndrome and one in Rolandic epilepsy 23–28.
TRAAK displays low leak activity under resting conditions and is highly activated by increasing membrane tension29,30. Conformational equilibrium between up and down states of transmembrane helix 4 (TM4) from each subunit of the dimeric channel underlies mechanical activation 30–32. At low tension, the TM4 down state predominates, creating membrane-facing fenestrations above TM4 that connect the channel cavity to the surrounding membrane inner leaflet. In this conformation, lipid acyl chains access the channel cavity through the open fenestrations to block conduction. Leak activity is due to rare spontaneous delipidation of the TM4 down state, resulting in brief openings. Increased membrane tension promotes upward movement of the TM4s due to changes in the shape of the channel that are energetically favored under tension. This movement of TM4 seals the lateral fenestrations, preventing lipid block, and results in longer duration and higher conductance mechanically-gated openings. Similar TM4 up and down states have been observed in the related mechanosensitive K2Ps TREK-1 and TREK-2, though gating in these channels also involves conformational changes in the K+-coordinating selectivity filter.
Given the physiological contribution of TRAAK and TREK-1 to action potential propagation, implication of TRAAK in epileptic disorders, and demonstrated channel inhibition by hydrophobic lipid acyl chains, we asked whether CBD modulates TRAAK—and related K2P channel—activity. We show that cannabinoids are potent blockers of mechanosensitive K2Ps and elucidate the mechanistic basis of channel inhibition.
Results
We first investigated cannabinoid inhibition of TRAAK channels in excised patches. CBD potently inhibited TRAAK, with effects evident at 1–10 nM, full inhibition at ~1 μM, and an IC50 ~ 145 nM (Fig. 1A–D). Preliminary experiments using plastic perfusion components resulted in lower apparent potency, similar to effects reported in a study of CBD activation of Kv7.2/7.3 channels and attributed to cannabinoid absorption to plastics 16. Final experiments were therefore performed with all glass components. We next asked if the related mechanosensitive K2Ps TREK-1 and TREK-2 were similarly cannabinoid-sensitive. All three channels were inhibited by CBD, but TREK-1 and TREK-2 were ~10-fold less sensitive than TRAAK (IC50s 1.5 and 1.3 μM, respectively, Fig. 1D–H). TREK-2 showed a steeper response profile and larger Hill-coefficient (1.3, 1.5, and 3.8 for TRAAK, TREK-1, and TREK-2, respectively), which suggests TREK-2 may have a unique CBD binding mode and/or stoichiometry.
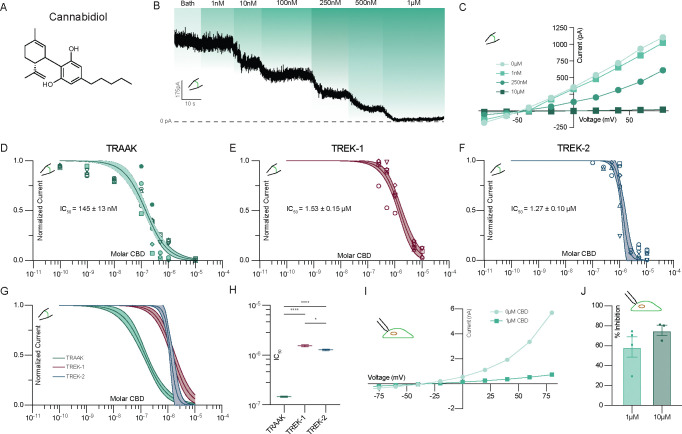
In excised inside-out patch experiments, CBD has direct access to the intracellular side of the channels (Fig. 1A–H). To test whether CBD can inhibit TRAAK from extracellular side, we conducted whole-cell recordings from cultured cells. TRAAK remained strongly inhibited by extracellular CBD (Fig. 1I,J). While conducting these experiments, we found bath perfusion readily activated TRAAK, likely due to shear forces that increase membrane tension 33,34. We used focal perfusion and low solution flow rates to minimize mechanical channel activation, but cannot exclude residual mechanical effects contributing to the lower apparent sensitivity of TRAAK to extracellular compared to intracellular CBD (Fig. 1D,J).
CBD consists of a bicyclic head group and hydrocarbon tail (Fig. 1A). To identify the chemical features of CBD important for channel inhibition, we evaluated several related phytocannabinoids with select structural differences. Cannabinol (CBN), an oxidized derivative of Δ9-THC, has with the same hydrocarbon tail as CBD but a different, tricyclic head structure (Figure 2A). The additional aromatization in CBN increases head planarity and eliminates rotational freedom of motion relative to CBD. CBN is ~33% more potent than CBD (IC50 107 nM), suggesting that the entropic cost of ordering the bicyclic head of CBD limits affinity. Cannabidiphorol (CBDP) and cannabidivarin (CBDV) share the same bicyclic head structure as CBD, but possess hydrocarbon tails with two additional or two fewer methylene units, respectively. These changes in hydrocarbon tail length have dramatic effects on potency. Compared to CBD, the more hydrophobic CBDP is ~10-fold more potent (IC50 17±3 nM) while the less hydrophobic CBDV is ~40-fold less potent (5.58 ± 0.34 μM, Fig. 2B–D). CBDV still fully inhibits TRAAK channels at high (20 μM) concentration. This demonstrates that increasing planarity or hydrophobicity among related cannabinoids increases inhibitor potency.
Gain-of-function TRAAK mutations have been identified in human disease 23–28 and functional studies of K2Ps 35,36. The mechanistic basis for channel activation by several of these mutations is known and involves stabilization of either leak- or mechanically gated-like open states 32. We evaluated five TRAAK gain-of-function mutants to better understand CBD’s mechanism of action and to explore the susceptibility of disease-causing channel variants to cannabinoid inhibition.
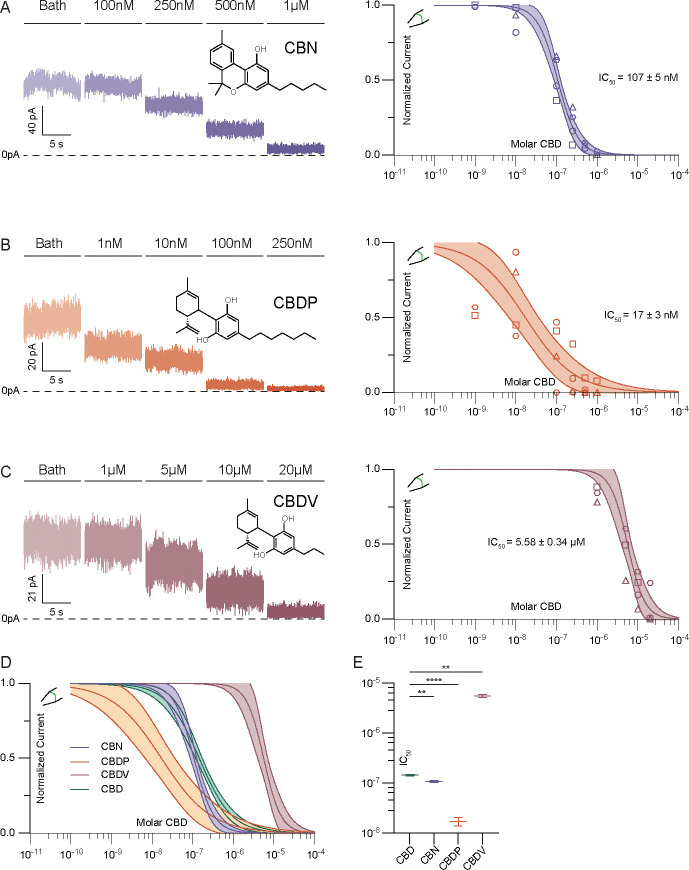
We first considered the FHEIG-inducing mutations TRAAKA270P and TRAAKA198E 23–25,28. These mutants are highly activated under low-tension basal conditions, with open probabilities well over 0.9, because they mimic the effect of high membrane tension on wild-type channels 32. Both mutations shift the conformational equilibrium from the TM4-down, lipid-blocked/leak open state to TM4-up, mechanically gated-like open conformations 32. We reasoned that if CBD inhibits TRAAK by competing with lipid acyl chains to block the channel cavity in the TM4-down state, these mutants will show diminished sensitivity to inhibition. Indeed, TRAAKA270P and TRAAKA198E were ~8- and ~4-fold less sensitive than wild-type TRAAK to CBD (IC50 = 1.12±0.04 μM and 496±26 nM, respectively, Fig. 3A,B, Fig. S1). For both mutants, high (10 μM) concentrations of CBD fully inhibited channel activity.
We next considered three mutants, TRAAKG165R, TRAAKG165E, and TRAAKP259L, recently identified in cases of FHEIG (G165R26, G165E27) and Rolandic epilepsy (P259L27). All three mutants retained high sensitivity to CBD (Figure 3C–E), with TRAAKG165E and TRAAKP259L statistically indistinguishable (IC50 = 162 ± 8 and 156 ± 8 nM, respectively) and TRAAKG165R only modestly reduced compared to wild-type (IC50 = 356±18 nM) (Fig. 3C–E, Fig. S1). This suggests these mutations activate TRAAK to a lesser degree or in a manner distinct from the FHEIG-causing mutations TRAAKA270P, TRAAKA198E.
Finally, we considered TRAAKG158D. TRAAKG158D is activated under basal conditions with an open probability ~0.7 and similar mutations at this site activate all vertebrate K2Ps 32,35. G158 points into the channel cavity towards the lipid binding site; introduction of the negatively charged aspartic acid electrostatically disfavors lipid block, shifting the conformational equilibrium towards a TM4-down, leak-like open state under basal conditions 32. We reasoned that if CBD, like lipids, binds in the channel cavity to block conduction, this mutant would be less sensitive to inhibition. The response of TRAAKG158D to CBD treatment was found to be different from wild-type TRAAK in two ways. First, the channel was ~7.1-fold less sensitive to CBD with an EC50 = 1.03 ± 0.22 μM (Fig. 3F, Fig. S1). Second, CBD showed lower efficacy against TRAAKG158D than wild-type TRAAK or other disease-causing variants, with inhibition plateauing at ~45% of initial current even at the highest CBD concentration tested (10 μM, Fig. 3E). These results suggest that (i) CBD has reduced affinity for TRAAKG158D due to electrosteric repulsion between the mutated residue and the cannabinoid and (ii) CBD only partially occludes its conduction pathway, potentially because of an altered CBD binding pose.
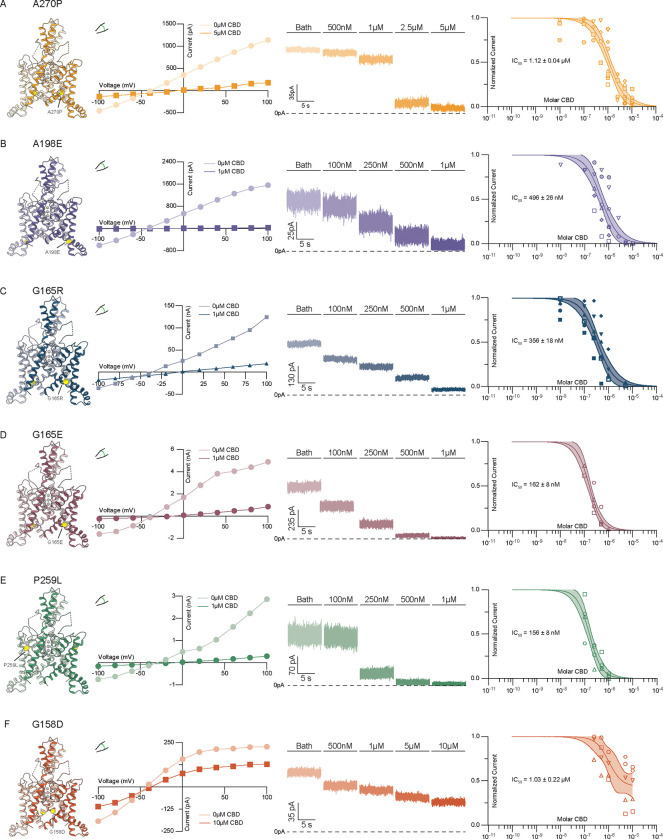
Data to this point are consistent with a model in which cannabinoids bind in the channel cavity to inhibit mechanosensitive K+ channels and preferentially bind TM4-down over TM4-up states. A prediction of this model is that increasing membrane tension will reduce CBD inhibition of TRAAK current by promoting TM4-up states. We tested this prediction by using methyl-beta-cyclodextrin (mβCD) to increase membrane tension in TRAAK-containing patches. mβCD has been shown to irreversibly increase tension and activate force-gated ion channels by sequestering cholesterol and lipids from the membrane 37,38. Fig. 4A shows an experiment in which TRAAK activity and CBD sensitivity were recorded before and after treatment with mβCD. Prior to mβCD treatment, 1 μM CBD treatment resulted in near complete (91 ± 3%) inhibition (Fig. 4A,B) that was reversible after washout. Subsequent mβCD treatment activated channels and activity remained elevated following mβCD washout, indicating stable high membrane tension in the patch. From this activated state, 1 μM CBD treatment resulted in only partial (43 ± 13 %) inhibition. Approximately 10-fold higher CBD concentrations were required to achieve a similar degree of inhibition to prior to mβCD treatment (Fig. 4A).
A second prediction of our model is that CBD inhibition will reduce mechanically activated TRAAK currents by preferentially binding to and stabilizing TM4-down conformations. To test this, we measured current elicited by steps of negative pressure before CBD application and after addition of 0.5, 1, and 10 μM CBD (Fig. 4C–E, Fig. S2). We found that as CBD concentration increased, more negative pressure was required to activate TRAAK to the same degree.
These results support our model for cavity binding and steric occlusion of the conduction path by CBD, but do not fully exclude an alternative explanation in which CBD inhibits TRAAK indirectly by decreasing basal tension or changing other membrane properties to promote closed channel states. We therefore asked if increasing concentrations of CBD alters membrane tension. In a patch recording, the Young-Laplace equation T = ΔPr/2 relates membrane tension (T) to the measurable pressure difference across the lipid bilayer (ΔP) and membrane radius of curvature (r). We measured changes in patch radius elicited by pressure steps before and after CBD treatment (Figure S3). We found that even 50 μM CBD, a concentration higher than that used in the other electrophysiology experiments herein, had no effect on membrane tension, consistent with direct inhibition of TRAAK and TREK channels by CBD.
We next pursued a structural approach to gain molecular insight into the basis for cannabinoid inhibition of mechanosensitive K+ channels. Cryo-EM structures of Danio rerio TREK-1 in apo, phosphatidylethanolamine(PE)-inhibited, and phosphatidic acid (PA)-activated states are known 39. Like human TREK-1 (Fig. 1E), we found Danio rerio TREK-1 is inhibited by CBD (IC50 = 11.1 ± 1.3 μM, Supplementary Fig. 4). We determined a cryo-EM structure of Danio rerio TREK-1 in the presence of 100 μM CBD (Fig. 5A–B, Figs. S5–6, Table S1). The structure was resolved to an overall resolution of 3.94 Å, though local resolution for most of the channel is substantially higher and map quality in regions discussed below is comparable to previously reported structures resolved to 2.8–3.5 Å 39 (Fig. S6).
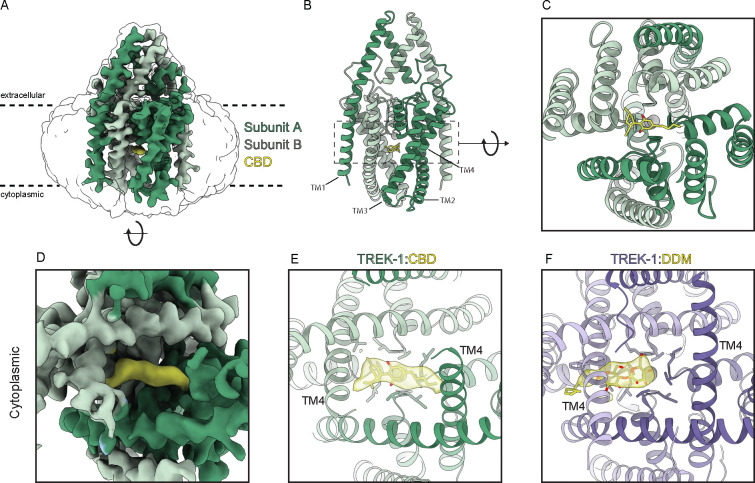
A strong, elongated density feature consistent with CBD is evident in the channel cavity directly underneath the selectivity filter (Fig. 5A–E). The density is best fit with the CBD aromatic ring centered under the filter and the terpene head group and hydrophobic tail projecting on either side towards the lateral fenestrations. We note this placement is speculative because the resolution is insufficient to unambiguously define CBD binding pose. Several lines of structural evidence support this model for CBD binding in the channel cavity. First, cavity density in the TREK-1:CBD structure is markedly different than that in the apo TREK-1 structure 39, which was prepared in the same way apart from the addition of CBD. In apo TREK-1, the cavity density is smaller and biased towards one subunit, extends into the surrounding micelle, and is consistent with a modeled dodecyl maltoside detergent (DDM) molecule (Fig. 5E,F). Second, CBD addition resulted in conformational changes to TM4 position. In the TREK-1:CBD structure both TM4s are down, while in apo TREK-1 one TM4 is up and one TM4 is down (Fig. 5E, Fig. S7). The symmetric TM4-down TREK-1:CBD conformation is more similar to the inhibited TREK-1:PE structure than the apo TREK-1 structure (Fig. S7). Third, the TREK-1:CBD structure shows differences in selectivity filter occupancy (Supplementary Fig. 8). In the presence of CBD, density for K+ is evident in sites 2 and 4. In contrast, K+ density is evident in sites 1–3 in apo TREK-1, 1–3 in TREK-1:POPE, and 1–4 in TREK-1:POPA structures 39. We conclude CBD inhibits TREK-1 by binding in the channel cavity to sterically block ion passage. Destabilization of K+ coordination in the selectivity filter may additionally contribute to channel inhibition.
Discussion
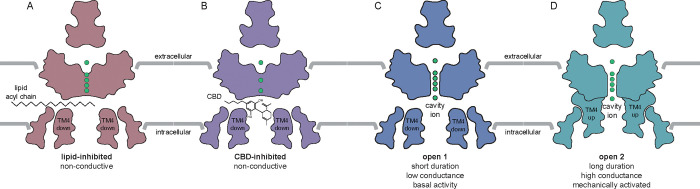
Data presented here support a model in which CBD inhibits mechanosensitive K+ channels by binding in the channel cavity to block ion conduction (Fig. 6). At low tension, TRAAK and TREK channels predominantly adopt TM4-down conformations that expose lateral fenestrations above TM4 to the surrounding lipid membrane 31,32. In this conformation, the channels are predominantly closed because lipid acyl chains access the cavity through lateral fenestrations and block ion conduction; spontaneous delipidation results in low leak activity. High tension promotes a TM4-up conformation that closes lateral fenestrations to prevent lipid block, resulting in a high conductance, open channel. We found increasing mechanical activation of TRAAK reduces CBD inhibition, while increasing CBD concentration reduces mechanically activated currents (Fig. 4). Mutations that promote TM4-up conformations decrease channel sensitivity to CBD (Fig. 3). The TREK-1:CBD structure shows both TM4s down, rather than one up and one down as in the apo TREK-1 structure 39. This is likely because CBD as modeled would sterically clash with TM4 residues if the helix adopted an up conformation. We conclude CBD preferentially binds to TM4-down channel conformations and effectively competes with abundant membrane lipids for an overlapping cavity site to block ion conduction. Whether CBD accesses the channel cavity through membrane-facing fenestrations, through the cytoplasm, or both, remains to be determined.
CBD modulates distantly-related ion channels in the voltage-gated ion channel superfamily, inhibiting Navs and BK 13,18 and activating TRPVs, TRPA1 14,15, and KCNQs 16. With an IC50 of 145 nM, TRAAK is among the highest-affinity CBD targets known, while TREK-1 and TREK-2 show roughly 10-fold lower potency similar to Navs. CBD is the highest affinity TRAAK inhibitor reported to date with ~3–5 fold higher affinity than RU-TRAAK-1 and −2 and greater than 10-fold higher affinity than nonspecific inhibitors ruthenium red and TKDC40,41.
Structures of TRPV2 15,42,43, Nav1.7 44, and KCNQ7.2/7.3 17 in complex with CBD have been reported. Notably, CBD binds all these channels in similar fenestration site(s) that connect channel cavities to the surrounding membrane. CBD binding within all four fenestrations of TRPV2 and KCNQ channels stabilizes their open states, while binding within one fenestration of Nav1.7 (together with a second CBD molecule binding to the IFM motif) stabilizes the inactivated state. In bacterial NavM, CBD binds deeper within all four fenestrations, partially entering the channel cavity 45. The mechanism of action of CBD on mechanosensitive K+ channels is fundamentally different. CBD does not bind within fenestrations, but rather fully accesses the channel cavity to bind directly underneath the selectivity filter (Fig. 5). Differences in these binding sites suggests structural insights could guide the design of cannabinoid derivatives with higher specificity to particular ion channels.
CBD inhibition of mechanosensitive K2Ps has potential clinical relevance that warrants further exploration. The molecular target of CBD that underlies its antiepileptic effects remains unknown, but TRAAK is a compelling candidate for several reasons. TRAAK activity controls spike propagation in myelinated neurons, where it localizes to axon initial segments 19,20 and nodes of Ranvier 21,22. In these specialized neuronal compartments, TRAAK colocalizes with Nav channels, the target of most classic anticonvulsants, and KCNQ channels, the target of the only approved K+ channel-specific antiepileptic. Inhibition of TRAAK or TREK-1 reduces Nav availability by preventing recovery from inactivation, thereby limiting spike velocity and frequency21,22, which could diminish the ectopic excitability characteristic of epileptic disorders. In addition, gain-of-function mutations in TRAAK cause epilepsy in humans, either in a form of Rolandic epilepsy or the neurodevelopmental disorder FHEIG 23–28. While some of these patients respond to anti-seizure medications including carbamazepine, oxcarbazepine, and valproate, others are refractory to treatment 26. There are currently no specific TRAAK inhibitors known. We show all known disease-causing variants (TRAAKG165E, TRAAKG165R, TRAAKP259L, TRAAKA270P, and TRAAKA198E) retain sensitivity to inhibition by CBD. These results suggest CBD or other cannabinoids could be pursued as a targeted treatment of TRAAK channelopathies in patients.
Methods
Electrophysiology
For patch clamp recording from Xenopus oocytes, genes encoding full-length Homo sapiens TRAAK (UniProt Q9NYG8–2), TREK-1 (UniProt O95069), and TREK-2 (UniProt P57789) were codon optimized for eukaryotic expression, synthesized (Genewiz), and cloned into a modified pGEMHE vector using Xho1 and EcoR1 restriction sites. The transcribed messages encode H. sapiens TRAAK amino acids 1–393, TREK-1 amino acids 1–426, or TREK-2 amino acids 1–538 with an additional three amino acids (SNS) at the C terminus. TRAAK mutants were introduced by polymerase chain reaction (PCR). Linearized DNA was transcribed in vitro using T7 polymerase. Complementary RNA (0.1 to 10 ng for TRAAK, TREK-1, TREK-2, and mutants) in 50 nL H2O was injected into Xenopus laevis oocytes extracted from anesthetized frogs. Currents were recorded at 25 °C from inside-out patches excised from oocytes 1 to 5 d after RNA injection. The pipette solution contained 15 mM KCl, 135 mM NaCl, 2 mM MgCl2, and 10 mM HEPES (pH = 7.4 with KOH) and the bath solution contained 150 mM KCl and 10 mM HEPES (pH = 7.1 with KOH). Borosilicate glass pipettes were pulled to 2–5 MΩ resistance.
For patch clamp recording from cultured cells, drTREK-1 or hsTRAAK genes were cloned into a modified pCEH vector to generate C-terminally EGFP-tagged constructs29. HEK293T were cultured in DMEM (Gibco, Thermo Fisher Scientific) with 10% FBS and 100 U ml−1 penicillin and 100 μg ml−1 streptomycin. Trypsinized cells were deposited on 12-mm glass coverslips in a six-well dish one to two days before transfection. For transfection, drTREK-1 or hsTRAAK plasmids were mixed with FuGENE 6 in OptiMEM at a 1:3 ratio with 1 μg DNA per well per construct and applied to cells in growth medium with antibiotics. Patching was conducted from 24 to 60 h post-transfection. Coverslips were placed in a perfusion chamber at room temperature in isotonic bath solution: 135 mM NaCl, 15 mM KCl, 1 mM CaCl2, 1 mM MgCl2, 10 mM HEPES, adjusted to a final pH of 7.5 with NaOH. Cells were chosen by a combination of the presence GFP fluorescence and cell morphology consistent with healthy interphase cells. Borosilicate glass pipettes were pulled to a resistance of 1.2–1.8 MΩ for patch electrophysiology and 1.8–2.8 MΩ for whole cell electrophysiology. Pipettes were filled with pipette solution: 150 mM KCL, 5 mM EGTA, 3 mM MgCl2, and 10 mM HEPES, adjusted to pH 7.5 with KOH.
Patches were evaluated with voltage-step protocols to confirm TRAAK expression and minimal non-specific leak based on reversal potential. Currents were recorded using an Axopatch 200B amplifier (Molecular Devices) at a bandwidth of 1 kHz and digitized with an Digidata 1550B (Molecular Devices) at 100–500 kHz using pClamp10.7 software. Pressure was applied with a second-generation high-speed pressure clamp device (HSPC-2-SB, ALA Scientific Instruments). Aggregated data were from patches from n ≥ 2 different cells on different days. No relevant differences were observed between patches from different cells. In an effort to minimize variability associated with differences in resting membrane tension, we targeted patches with low basal curvature and low resting channel activity and ensured measured pressure difference across the patch was as close to zero as possible at rest to prevent mechanical activation of channels. Data were analyzed using Clampfit 10.7, Excel, and Graphpad Prism software. For voltage families, the data were decimated 100x and plotted in Prism. Statistical analysis was conducted with Dunnett’s multiple comparisons test after an ordinary one-way ANOVA using Prism software.
To avoid cannabinoid absorption to plastic, cannabinoid solutions were made in glass vials and diluted by weight, perfusion was performed using glass tubing pulled from borosilicate glass Pasteur pipettes (Fisherbrand, Cat# 13–678-20C), and recordings were performed in glass chambers. Channel activity was continuously recorded during perfusion steps. Patches were exposed to each cannabinoid concentration until a stable current level was reached (0.5–2 minutes). CBD (Cayman Chemical cat#90080) was prepared as a 10mM stock solution in DMSO, CBN (Cayman Chemical cat#25495) was prepared as a 80mM stock solution in DMSO, CBDV (Phytolab Cat#85955) was prepared as a 10mM stock solution in DMSO, and CBDP (Cayman Chemical cat#33611) was purchased as a 2.9 mM stock solution in methanol. Cannabinoids were serially diluted in bath solution to achieve working concentrations of 50 μM to 10 nM. Bath volume in the recording chamber was minimized by suction and drug-containing solutions were delivered three times to ensure establishment of desired concentration in the recording chamber. Cannabinoids were applied from low to high concentration. The recording chamber and perfusion system were washed extensively with methanol after every trial.
The effects of cannabinoids on channel activity were quantified by the reduction in channel current evoked at 0 mV during a gap-free recording or during 200 ms long voltage families with steps ranging from −100 to +100 mV. Currents were normalized to those prior to cannabinoid application to facilitate comparisons across different patches and cells. Dose response curves were four-parametric Boltzmann fits to aggregated data. For voltage-step recordings, current was measured at 0mV from each recording. Data were aggregated for all recordings with and without drug present and were normalized to the baseline activity before drug administration. Comparisons between constructs and drug isoforms were conducted with ordinary one-way ANOVA and Tukey multiple-comparison tests.
Patch imaging and membrane tension calculation
A construct encoding EGFP fused to the CAAX-containing C-terminal tail of H. sapiens H-Ras (NP_005334 amino acids 170–189) through a GGRS linker was cloned into a pCS2+ vector using Gibson assembly. Linearized DNA was transcribed in vitro using T7 polymerase. 3–10 ng complementary RNA in 50 nL H2O was injected into Xenopus laevis oocytes. Excised patches were illuminated with an LED light engine (SpectraX, Lumencor) through a GFP filter (450/50 nm excitation, 506 nm dichroic mirror, 500 nm longpass emission filter) and water immersion objective lens (x60, NA1.0). Movies were recorded at 120 Hz with an infrared camera (IR-2000, DAGE-MTI). Images were preprocessed within FIJI (ImageJ). Image contrast was enhanced to facilitate analysis. Video files were loaded into FIJI and converted into a JPEG stack. Frames were time matched to stimuli by multiplying frame rate and time. Tension was calculated using python scripts as previously reported 30.
TREK-1 expression and purification
Danio rerio TREK-1 (UniProt Q9NYG8–2) was cloned for expression in Pichia pastoris as previously described 29 with modifications described here. The construct used for purification included an additional 26 amino acid N-terminal sequence from human TRAAK compared to Q9NYG8 that improved heterologous expression. The final construct is C-terminally truncated by 119 amino acids, incorporates two mutations to remove N-linked glycosylation sites (N104Q/N108Q), and is expressed as a C-terminal PreScission protease-cleavable EGFP-10x His fusion protein. As a result, there is an additional amino acid sequence of ‘‘SNSLEVLFQ’’ at the C terminus of the final purified protein after protease cleavage.
Starter cultures of recombinant Pichia were grown in YPD with 0.5 mg/mL Zeocin and grown at 30°C with shaking at 250 rpm overnight (12–14 h). Four 1L flasks of BMGY with 25ug/mL Zeocin were inoculated with 10mL of starter culture each. Cells grew for approximately 24 hours at 30°C at 250 rpm. 1L cell cultures were pelleted by centrifugation at 8000g for 10 minutes and resuspended in 1L BMMY with 25ug/mL Zeocin. Cells were harvested 40–60 h after induction with methanol and flash frozen in liquid N2. ~60 g of frozen Pichia cells expressing drTREK-1 were disrupted by milling (Retsch model MM301) 5 times for 3 min at 25 Hz. All subsequent purification steps were carried out at 4°C. Milled Pichia cells were thawed in 200 mL of Lysis Buffer containing 50 mM TRIS, 150 mM KCl, 1mM EDTA pH 8. Protease inhibitors (Final Concentrations: E64 (1 μM), pepstatin A (1 μg/mL), soy trypsin inhibitor (10 μg/mL), benzamidine (1 mM), aprotinin (1 μg/mL), leupeptin (1μg/mL), AEBSF (1mM), PMSF (1mM)), benzonase (10μL) and DNAse (10 μL) were added to the lysis buffer immediately before use. Cells were lysed by sonication and centrifuged at 150,000 × g for 45 minutes. The supernatant was discarded, and residual nucleic acid was removed from the top of the membrane pellet using DPBS. Membrane pellets were scooped into a Dounce homogenizer containing extraction buffer (50 mM TRIS, 150 mM KCl, 1 mM EDTA, 1.5% n-Dodecyl-β-D-Maltopyranoside (DDM, Anatrace, Maumee, OH), 0.3% cholesteryl hemisuccinate Tris salt (CHS, Anatrace, Maumee, OH) pH 8). A stock solution of 10% DDM, 2% CHS was dissolved and clarified by bath sonication in 200 mM HEPES pH 8 prior to addition to buffer to the indicated final concentration. Membrane pellets were then homogenized in extraction buffer and this mixture (150 mL final volume) was gently stirred at 4°C for 2 hours. The extraction mixture was centrifuged at 33,000 × g for 45 minutes and the supernatant, containing solubilized membrane protein, was bound to 4 mL of Sepharose resin coupled to anti-GFP nanobody for 2 hours at 4°C. The resin was then collected in a column and washed with 10 mL of buffer 1 (20 mM TRIS, 150 mM KCl, 1 mM EDTA, 0.025% DDM, 0.005% CHS, pH 8), 40 mL of buffer 2 (20 mM TRIS, 500 mM KCl, 1 mM EDTA, 0.025% DDM, 0.005% CHS, pH 8), and 10 mL of buffer 1. The resin was then resuspended in 6 mL of buffer 1 with 0.5 mg of PPX protease and rocked gently in the capped column overnight (~ 12–14 h). Cleaved TREK-1 was then eluted with an additional 12 mL of wash buffer, spin concentrated to ~1 mL with Amicon Ultra spin concentrator 100 kDa cutoff (Millipore) and loaded onto a Superose S200 increase column (GE Healthcare, Chicago, IL) on an NGC system (Bio-Rad, Hercules, CA) equilibrated in an elution buffer (20 mM TRIS, 150 mM KCl, 1 mM EDTA, 0.025% DDM, pH 8). Peak fractions containing TREK-1 protein were then collected and spin concentrated prior to sample freezing.
Cryo-electron microscopy
TREK-1 in DDM detergent was prepared at a final concentration of 3.1 (purification 1) or 4.4 (purification 2) mg/mL. CBD was spiked into the sample from a stock concentration of 10 mM in DMSO to a final concentration of 100 μM. The sample was incubated on ice for 30 minutes then clarified by a 10-minute 21,000 × g spin at 4°C prior to grid preparation. 3.4 μl of protein was applied to freshly glow discharged Holey Carbon, 300 mesh R 1.2/1.3 gold grids (C-flat, Electron Microscopy Sciences, USA (purification 1), Quantifoil, Großlöbichau, Germany (purification 2)) and plunge frozen in liquid ethane using a FEI Vitrobot Mark IV (ThermoFisher Scientific) set to 4°C, 100% humidity, 1 blot force, wait time of ~5 seconds, and 3 second blot time. Grids were clipped and stored in liquid nitrogen.
Both datasets were collected on a Titan Krios G3i electron micro-scope (Thermo Fisher) operated at 300 kV and equipped with a Gatan BioQuantum Imaging Filter with a slit width of 20 eV. Dose-fractionated images (~50 electrons per Å2 over 50 frames) were recorded on a K3 direct electron detector (Gatan) at a pixel size of 0.848 Å. 242 movies were collected in an 11 × 11 hole pattern with two targets per hole around a central hole position using image shift. Defocus was varied from −0.5 to −1.8 μm using SerialEM 46.
Motion correction was performed on 9,046 micrographs from collection 1 and 8,470 micrographs from collection 2 using patch motion correction in cryoSPARC v4.4.1 47. Contrast transfer function (CTF) parameters were fit with patch CTF 48. For collection 1, template-free auto-picking of particles was performed with a blob picker with a minimum particle size of 120 Å and a maximum particle size of 200 Å on 5,481 movies CTF fit to 5.0Å or better, yielding an initial set of 2,315,705 particles. These were extracted at a 300-pixel box size for two-dimensional (2D) classification. Iterative rounds of 2D classification yielded a set of 45,022 particles that were used to train a Topaz model. Topaz-trained particle-picking yielded 1,176,701 particles which were manually curated through iterative rounds of 2D classing and multiple multi-class Ab-initios in which particle subsets that yielded the best 3D structure were chosen. Final ab-initio and non-uniform refinements (C1, 4 extra passes, 15Å initial resolution) yielded a 5.14 Å map from 261,227 particles.
For collection 2, the topaz model from collection 1 was utilized to conduct topaz-trained particle picking on 7,968 movies CTF fit to 7Å or better. Topaz picking and extraction a box size of 300Å yielded an initial set of 1,673,778 particles. These particles were curated through iterative rounds of 2D classification to a subset of 129,260 particles. These particles were then re-extracted from 3729 micrographs CTF fit to between 2.4Å and 7Å for a subset of 92,936 particles that were then used to train a new topaz model on the 3729 micrographs CTF fit between 2.4Å and 7Å. The second round of Topaz training, picking, and extraction yielded 750,776 particles. These classes were manually curated through a single round of 2D classification before a 3-class Ab-initio. The class with the clearest protein density was chosen yielding a subset of 316,175 particles with clear protein density. These particles were subjected to iterative rounds of 2D and 3D classification. Final ab-initio and non-uniform refinements (C1, 4 extra passes, 15Å initial resolution) yielded a 5.86 Å map from 228,680 particles.
The 261,227 particles from collection 1 and 228,680 particles from collection 2 were then re-extracted at a 300Å box size and joined into one particle set. Particles too close to the edges of micrographs were discarded, resulting in a joint subset of 484,500 particles. These particles were curated over 3 rounds of 3D classification to remove remaining junk. An ab-initio reconstruction of the remaining 160,072 particles was performed to provide an initial volume and a subsequent non-uniform refinement (C1, 4 extra passes, 15Å initial resolution) which resulted in a map with a 4.31 Å overall resolution. This map was post-processed in cryoSPARC v4.4.1 and used for reference-based motion correction. The resulting ‘shiny’ particles were subject to one round of 2D classification before one 3-class 3D classification resulting in 117,795 particles. These particles were used to generate a new ab initio model. We generated a mask to refine this map in Chimera (UCSF) from the previously reported drTREK-1 structure 39. This mask was imported to cryoSPARC v4.4.1 and modified to a dilation radius of 7 pixels and a soft padding of 25 pixels. Local refinement (C1, 2 extra passes, 12Å initial resolution) of our volume with this mask resulted in a map with 4.15Å overall resolution. This map was utilized for a second round of reference-based motion correction. An ab initio and local refinement (C1, 2 extra passes, 12Å initial resolution) of the twice-polished 117,609 particles resulted in a final map of 3.94Å nominal resolution.
The final non-uniformed local refined and sharpened map from Cryosparc was used for modeling. An initial apo TREK-1 model (PDB: 8DE7) was rigid body fit to the density in Phenix 49. Model building was performed iteratively with manual adjustment in Coot 50, global real space refinement in Phenix 51, and geometry assessment in Molprobity52.
Supplementary Materials
References
- E. W. Bow, J. M. Rimoldi. The Structure–Function Relationships of Classical Cannabinoids: CB1/CB2 Modulation.. Perspect. Med. Chem., 2016
- K. M. Nelson. The Essential Medicinal Chemistry of Cannabidiol (CBD).. J. Med. Chem., 2020. [PubMed]
- I. Miller. Dose-Ranging Effect of Adjunctive Oral Cannabidiol vs Placebo on Convulsive Seizure Frequency in Dravet Syndrome.. JAMA Neurol., 2020. [PubMed]
- E. A. Thiele. Long-term cannabidiol treatment for seizures in patients with tuberous sclerosis complex: An open-label extension trial.. Epilepsia, 2022. [PubMed]
- O. Devinsky. Long-term cannabidiol treatment in patients with Dravet syndrome: An open-label extension trial.. Epilepsia, 2019. [PubMed]
- O. Devinsky. Effect of Cannabidiol on Drop Seizures in the Lennox–Gastaut Syndrome.. N. Engl. J. Med., 2018. [PubMed]
- O. Devinsky. Randomized, dose-ranging safety trial of cannabidiol in Dravet syndrome.. Neurology, 2018. [PubMed]
- O. Devinsky. Trial of Cannabidiol for Drug-Resistant Seizures in the Dravet Syndrome.. N. Engl. J. Med., 2017. [PubMed]
- E. Ryberg. The orphan receptor GPR55 is a novel cannabinoid receptor.. Br. J. Pharmacol., 2007. [PubMed]
- A. Straiker, M. Dvorakova, A. Zimmowitch, K. P. Mackie. Cannabidiol inhibits endocannabinoid signaling in autaptic hippocampal neurons.. Mol. Pharmacol., 2018
- E. C. Rosenberg. Cannabidiol modulates excitatory-inhibitory ratio to counter hippocampal hyperactivity.. Neuron, 2023. [PubMed]
- A. J. Hill. Voltage-gated sodium (NaV) channel blockade by plant cannabinoids does not confer anticonvulsant effects per se.. Neurosci. Lett., 2014. [PubMed]
- M.-R. Ghovanloo. Inhibitory effects of cannabidiol on voltage-dependent sodium currents.. J. Biol. Chem., 2018. [PubMed]
- C. Muller, P. Morales, P. H. Reggio. Cannabinoid Ligands Targeting TRP Channels.. Front. Mol. Neurosci., 2019. [PubMed]
- R. A. Pumroy. Molecular mechanism of TRPV2 channel modulation by cannabidiol.. eLife, 2019. [PubMed]
- H.-X. B. Zhang. Cannabidiol activates neuronal Kv7 channels.. eLife, 2022. [PubMed]
- D. Ma. Ligand activation mechanisms of human KCNQ2 channel.. Nat. Commun., 2023. [PubMed]
- J. Monat. Direct Inhibition of BK Channels by Cannabidiol, One of the Principal Therapeutic Cannabinoids Derived from Cannabis sativa.. J. Nat. Prod., 2024. [PubMed]
- V. Luque-Fernández. An ankyrin G–binding motif mediates TRAAK periodic localization at axon initial segments of hippocampal pyramidal neurons.. Proc. Natl. Acad. Sci., 2024. [PubMed]
- G. E Jr, Y Wu, Y Ogawa, X Ding, M. N Rasband. An evolutionarily conserved AnkyrinG-dependent motif clusters axonal K2P K+ channels.. J. Cell Biol., 2024. [PubMed]
- S. G. Brohawn. The mechanosensitive ion channel TRAAK is localized to the mammalian node of Ranvier.. eLife, 2019. [PubMed]
- H. Kanda. TREK-1 and TRAAK Are Principal K+ Channels at the Nodes of Ranvier for Rapid Action Potential Conduction on Mammalian Myelinated Afferent Nerves.. Neuron, 2019. [PubMed]
- C. K. Bauer. Mutations in KCNK4 that Affect Gating Cause a Recognizable Neurodevelopmental Syndrome.. American journal of human genetics, 2018. [PubMed]
- R. M. Elhossini. A recurrent KCNK4 variant in a dominant pedigree with hypertrichosis and gingival fibromatosis syndrome: Variable phenotypic expressivity and insights on patients’ dental management.. Am. J. Méd. Genet. Part A, 2024. [PubMed]
- P. Mariani. Hereditary gingival fibromatosis associated with the missense mutation of the KCNK4 gene.. Oral Surg., Oral Med., Oral Pathol. Oral Radiol., 2021. [PubMed]
- M. Krygier. The epilepsy phenotype of KCNK4-related neurodevelopmental disease.. Seizure: Eur. J. Epilepsy, 2024
- H.-J. Yan. Expanding the phenotypic spectrum of KCNK4: From syndromic neurodevelopmental disorder to rolandic epilepsy.. Front. Mol. Neurosci., 2023. [PubMed]
- R. Garg, J. Sullivan, D. Blair, R. Wallerstein. KCNK4-related channelopathy causing a neurodevelopmental syndrome.. BMJ Case Rep., 2023
- S. G. Brohawn, Z. Su, R. MacKinnon. Mechanosensitivity is mediated directly by the lipid membrane in TRAAK and TREK1 K+ channels.. Proc. Natl. Acad. Sci., 2014. [PubMed]
- B. Sorum, T. Docter, V. Panico, R. A. Rietmeijer, S. G. Brohawn. Tension activation of mechanosensitive two-pore domain K+ channels TRAAK, TREK-1, and TREK-2.. Nat. Commun., 2024. [PubMed]
- S. G. Brohawn, E. B. Campbell, R. MacKinnon. Physical mechanism for gating and mechanosensitivity of the human TRAAK K+ channel.. Nature, 2014. [PubMed]
- R. A. Rietmeijer, B. Sorum, B. Li, S. G. Brohawn. Physical basis for distinct basal and mechanically gated activity of the human K+ channel TRAAK.. Neuron (, 2021. [DOI]
- A. J. Patel. A mammalian two pore domain mechano-gated S-like K+ channel.. The EMBO journal, 1998. [PubMed]
- M. Lengyel, P. Enyedi, G. Czirják. Negative Influence by the Force: Mechanically Induced Hyperpolarization via K2P Background Potassium Channels.. Int J Mol Sci, 2021. [PubMed]
- I. B. Soussia. Mutation of a single residue promotes gating of vertebrate and invertebrate two-pore domain potassium channels.. Nat Commun, 2019. [PubMed]
- M. Lolicato, P. M. Riegelhaupt, C. Arrigoni, K. A. Clark, D. L. Minor. Transmembrane helix straightening and buckling underlies activation of mechanosensitive and thermosensitive K(2P) channels.. Neuron, 2014. [PubMed]
- Y. Zhang. Visualization of the mechanosensitive ion channel MscS under membrane tension.. Nature, 2021. [PubMed]
- C. D. Cox, Y. Zhang, Z. Zhou, T. Walz, B. Martinac. Cyclodextrins increase membrane tension and are universal activators of mechanosensitive channels.. Proc. Natl. Acad. Sci., 2021. [PubMed]
- P. A. M. Schmidpeter. Membrane phospholipids control gating of the mechanosensitive potassium leak channel TREK1.. Nat Commun, 2023. [PubMed]
- Z. Su, E. C. Brown, W. Wang, R. Mackinnon. Novel cell-free high-throughput screening method for pharmacological tools targeting K+ channels.. Proceedings of the National Academy of Sciences of the United States of America, 2016. [PubMed]
- A. M. Natale, P. E. Deal. Jr., D. L. M. Structural insights into the mechanisms and pharmacology of K2P potassium channels.. J Mol Biol, 2021. [DOI | PubMed]
- R. A. Pumroy. Structural insights into TRPV2 activation by small molecules.. Nat. Commun., 2022. [PubMed]
- A. Gochman. Cannabidiol sensitizes TRPV2 channels to activation by 2-APB.. eLife, 2023. [PubMed]
- J. Huang. Cannabidiol inhibits Nav channels through two distinct binding sites.. Nat. Commun., 2023. [PubMed]
- L. G. Sait. Cannabidiol interactions with voltage-gated sodium channels.. eLife, 2020. [PubMed]
- D. N. Mastronarde. Automated electron microscope tomography using robust prediction of specimen movements.. Journal of structural biology, 2005. [PubMed]
- A. Punjani, J. L. Rubinstein, D. J. Fleet, M. A. Brubaker. cryoSPARC: algorithms for rapid unsupervised cryo-EM structure determination.. Nature Methods, 2017. [PubMed]
- J. Zivanov, T. Nakane, S. H. W. Scheres. Estimation of high-order aberrations and anisotropic magnification from cryo-EM data sets in RELION −3.1.. IUCrJ, 2020
- P. V. Afonine. Real-space refinement in PHENIX for cryo-EM and crystallography.. Acta crystallographica. Section D, Structural biology, 2018. [PubMed]
- P. Emsley, B. Lohkamp, W. G. Scott, K. Cowtan. Features and development of Coot.. Acta crystallographica Section D, Biological crystallography, 2010. [PubMed]
- D. Liebschner. Macromolecular structure determination using X-rays, neutrons and electrons: recent developments in Phenix.. Acta crystallographica. Section D, Structural biology, 2019. [PubMed]
- C. J. Williams. MolProbity: More and better reference data for improved all-atom structure validation.. Protein science : a publication of the Protein Society, 2018. [PubMed]
