Impact of prenatal exposure to delta 9‐tetrahydrocannabinol and cannabidiol on birth size and postnatal growth trajectories
Abstract
Background:
Prenatal exposure to cannabis (or more specifically, delta 9‐tetrahydrocannabinol [Δ9‐THC]) has been consistently linked to low birthweight. Animal models further show that Δ9‐THC is associated with rapid postnatal growth. Whether this association is modified by breastfeeding is unknown.
Methods:
In this exploratory study, we followed 128 mother–child pairs through 3 years. Urinary Δ9‐THC and cannabidiol (CBD) were measured mid‐gestation. Generalized linear models estimated the associations between Δ9‐THC and neonatal body composition. A mixed‐effects model estimated the association between Δ9‐THC and body mass index (BMI) z‐score trajectories. Interaction was assessed by a three‐way product term (Δ9‐THC × breastmilk months × age).
Results:
Fifteen children (12%) had Δ9‐THC exposure; three had concomitant CBD exposure. Prenatal exposure to Δ9‐THC alone was associated with lower fat mass (−95 g, 95% confidence interval [CI]: −174, −14) and neonatal adiposity (−2.1%; 95% CI: −4.2, −0.4) followed by rapid postnatal growth (0.42 increase in BMI z‐score per square root year; 95% CI: 0.12, 0.72). Breastfeeding modified this association (p = 0.04), such that growth was similar for those breastfed for 5 months whereas a shorter duration of breastfeeding was associated with 1.1 higher BMI z‐score at 3 years (95% CI: 0.21, 2.05).
Conclusions:
Our study suggests that prenatal exposure to Δ9‐THC may alter early‐life growth. Breastfeeding may stabilize rapid postnatal growth, but the impact of lactational exposure requires further investigation.
Article type: Research Article
Keywords: adiposity, BMI, breastfeeding, cannabidiol, cannabis, delta‐9‐tetrahydrocannabinol, foetal growth restriction, growth trajectory, postnatal weight gain
Affiliations: Department of Epidemiology Colorado School of Public Health Aurora Colorado USA; Lifecourse Epidemiology of Adiposity and Diabetes (LEAD) Center University of Colorado Anschutz Medical Campus Aurora Colorado USA; Department of Pediatrics, School of Medicine University of Colorado Anschutz Medical Campus Aurora Colorado USA; Department of Implementation Science Wake Forest University School of Medicine Winston‐Salem North Carolina USA; Department of Health Promotion and Behavioral Sciences The University of Texas Health Science Center Austin Texas USA; Department of Obstetrics and Gynecology School of Medicine, University of Colorado Anschutz Medical Campus Aurora Colorado USA; Department of Anesthesiology University of Colorado School of Medicine Colorado Aurora USA
License: © 2024 The Author(s). Pediatric Obesity published by John Wiley & Sons Ltd on behalf of World Obesity Federation. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1111/ijpo.13187 | PubMed: 39681476 | PMC: PMC11669837
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (593 KB)
INTRODUCTION
Over the past few decades, there has been a major shift in legality and attitudes towards cannabis use. As of 2024, cannabis is legal for recreational use in several countries (including Uruguay, Canada, South Africa, and Mexico) and 23 of 50 states in the United States. As cannabis becomes more socially acceptable, it is important to study the health effects, particularly in pregnancy.
Human epidemiologic studies have linked cannabis use during pregnancy with foetal growth restrictionref. ijpo13187-bib-0001, ref. ijpo13187-bib-0002 and low birthweight.ref. ijpo13187-bib-0003, ref. ijpo13187-bib-0004, ref. ijpo13187-bib-0005, ref. ijpo13187-bib-0006, ref. ijpo13187-bib-0007, ref. ijpo13187-bib-0008, ref. ijpo13187-bib-0009, ref. ijpo13187-bib-0010, ref. ijpo13187-bib-0011 However, since these studies relied on self‐report, it is unclear whether a specific component of cannabis is driving this association. Cannabis contains many cannabinoids, with delta‐Δ9‐tetrahydrocannabinol (Δ9‐THC) and cannabidiol (CBD) being the most common. Both Δ9‐THC and CBD interact with the endocannabinoid system, a complex signaling system involved in metabolism and glucose homeostasis. Δ9‐THC directly interacts with cannabinoid 1 (CB1) and cannabinoid 2 (CB2) receptors.ref. ijpo13187-bib-0012 Conversely, CBD indirectly interacts with CB1 and CB2 receptors, and may counteract some of the effects of Δ9‐THC.ref. ijpo13187-bib-0012 As such, murine models report that Δ9‐THC exposure decreases birthweight,ref. ijpo13187-bib-0013, ref. ijpo13187-bib-0014, ref. ijpo13187-bib-0015, ref. ijpo13187-bib-0016, ref. ijpo13187-bib-0017 whereas CBD may have no impact on birthweightref. ijpo13187-bib-0018 or may slightly increase it.ref. ijpo13187-bib-0019 However, whether Δ9‐THC and CBD act synergistically or antagonistically is unknown, as no human or animal studies have examined their individual or joint effects. This may have public health relevance, given the dramatic changes in the potency and ratio of Δ9‐THC:CBD in commercial cannabis products in recent years.ref. ijpo13187-bib-0020
Prenatal exposure to Δ9‐THC or CBD may also influence postnatal growth. Animal models show that prenatal exposure to Δ9‐THC is associated with rapid weight gain in infancy,ref. ijpo13187-bib-0013, ref. ijpo13187-bib-0014, ref. ijpo13187-bib-0015 whereas the effects of CBD on postnatal growth are less clear.ref. ijpo13187-bib-0021, ref. ijpo13187-bib-0022 One epidemiologic study reported that co‐exposure to tobacco and cannabis was associated with a rapid increase in BMI trajectory from birth to middle childhood.ref. ijpo13187-bib-0023 However, the impact of prenatal exposure to Δ9‐THC on postnatal growth trajectories has yet to be examined in longitudinal cohorts.
Clinicians use growth trajectories to make recommendations for stabilizing child growth. One such recommendation is to encourage mothers to exclusively breastfeed for about 6 months.ref. ijpo13187-bib-0024 A longer duration of breastfeeding is associated with healthier growth in infancyref. ijpo13187-bib-0025 and has been shown to protect against obesogenic effects of environmental exposures.ref. ijpo13187-bib-0026 However, these benefits may be negated by cannabinoid exposure via breast milk. Thus, it is critical to examine whether breastfeeding may impact the relationship between prenatal exposure to cannabis and postnatal growth.
To address the important research gaps, we assessed the associations between prenatal exposure to Δ9‐THC with birth size and postnatal growth trajectories through age 3 years. We hypothesized that prenatal exposure to Δ9‐THC, particularly in the absence of CBD, would be associated with lower neonatal adiposity followed by rapid growth in the first 3 years of life. We further hypothesized that a longer duration of exclusive breastfeeding would temper rapid postnatal growth among Δ9‐THC‐exposed offspring.
METHODS
We leveraged a racially and ethnically diverse Colorado‐based cohort: the Healthy Start study. Healthy Start began in 2010 as a study to better understand how overnutrition in pregnancy impacts obesity and cardiometabolic health of the offspring (NCT02273297). Pregnant women were recruited from the outpatient obstetrics clinics at the University of Colorado Hospital prior to 24 weeks of gestation. Women were excluded from this study if they were expecting multiple births or had pre‐existing diabetes, asthma, cancer, or psychiatric illness. Enrolled pregnant women were invited to participate in visits during pregnancy, at delivery, and after their child was born.
This exploratory study was conducted among a convenience subsample of 199 participants with available maternal urine collected at ~27 weeks gestation (the sample size was further reduced to 128 participants for the adiposity analysis and 140 participants for the trajectory analysis after exluding participants with missing data). The goal of this study was to investigate the impact of prenatal exposure to cannabis on neonatal body composition and BMI growth trajectories through 3 years of age.
Adiposity measures
At the delivery visit, neonatal fat mass and fat‐free mass were measured via whole body air displacement plethysmography (PEA POD, COSMED, Rome, Italy) within 72 h of delivery. The PEA POD system employs densitometric techniques to measure total body mass and two compartments in the offspring: fat mass (adipose tissue) and fat‐free mass. Neonatal adiposity (fat mass percentage) was calculated as a proportion of the fat mass divided by total mass. Fat mass and fat‐free mass were conducted twice. A third exam was conducted if fat mass percentage differed by >2%. The mean of the two closest measures was used in analyses.
Growth measures
Childhood weight, recumbent length (generally until 24 months), and standing height (generally after 24 months) were abstracted from medical records. These measurements were generally recorded at well‐child visits, which are recommended at 1, 2, 4, 6, 12, 18, 24, 30, and 36 months of age. BMI was calculated by dividing weight in kilograms by height in meters squared.
Prenatal exposure to cannabinoids
We measured Δ9‐THC, CBD, and its major metabolites in maternal urine collected at ~27 weeks gestation. Samples were analysed by iC42 Clinical Research and Development (Aurora, CO, USA), using a validated, specific, and highly sensitive liquid chromatography–tandem mass spectrometry (LC–MS/MS) assay.ref. ijpo13187-bib-0027 The LC–MS/MS system consisted of a 1200 high‐performance liquid chromatography system (Agilent Technologies, Palo Alto, CA, USA) and an API5000 tandem mass spectrometer (Sciex, Concord, ON, Canada) connected via an atmospheric pressure chemical ionization source run in the positive mode. The lower limits of quantification (LOQs) ranged from 0.39 ng/mL (for Δ9‐THC) to 7.82 ng/mL (for Δ9‐THC‐C‐gluc) (see Table 2).
TABLE 2: Summary of urinary cannabinoids and cannabinoid metabolitesijpo13187-note-0005 measured in maternal urine collected at ~27 weeks gestation, n = 128.
| Compound | Δ9‐THC | Δ9‐THC‐gluc | Δ9‐THC‐COOH | Δ9‐THC‐C‐gluc | CBD |
|---|---|---|---|---|---|
| Limit of quantification (ng/mL) | 0.39 | 1.56 | 0.78 | 7.82 | 0.78 |
| Limit of detection (ng/mL) | 0.20 | 0.78 | 0.39 | 3.91 | 0.39 |
| Minimum (ng/mL) | 0.3 | 0.8 | 0.4 | 4.5 | 0.5 |
| Maximum (ng/mL) | 0.9 | 24.1 | 20.1 | 1091.3 | 0.8 |
| Number of positive | 5 | 7 | 12 | 10 | 3 |
| Number of negative | 123 | 121 | 116 | 118 | 125 |
Abbreviations: Δ9‐THC, delta 9‐tetrahydrocannabinol; BMI, body mass index; CBD, cannabidiol.
No positive results for 11OH‐Δ9‐THC, CBD‐gluc, 6α‐OH‐CBD, 6β‐OH‐CBD, 7‐OH‐CBD, and CBD‐COOH.
Prenatal exposure to Δ9‐THC, alone or in combination with CBD, was categorized two ways. First, we characterized exposure to Δ9‐THC as not exposed (Δ9‐THC and its metabolites were below the LOD) and exposed (Δ9‐THC or metabolites exceeded the LOD). Second, we characterized exposure to Δ9‐THC alone as: not exposed (Δ9‐THC and/or its metabolites were not detected in the absence of CBD or its metabolites) and exposed (Δ9‐THC or its metabolites were detected, but CBD or its metabolites were not detected).
Covariates
Data on maternal education, household income, race, and ethnicity were collected through research questionnaires. Maternal age at delivery was calculated from delivery date and maternal date of birth. Maternal height was measured using a stadiometer at the first research visit. Pre‐pregnancy weight was obtained from medical records or from questionnaires completed at enrolment. Gestational weight gain was calculated as the difference between the last available weight measurement during pregnancy and the pre‐pregnancy weight. Maternal psychiatric disorders (non‐specified) and gestational diabetes status were obtained from medical records. Cotinine was measured in urine samples collected at 27 weeks gestation via solid‐phase competitive ELISA (Calbiotech Inc., El Cajon, CA, USA), with a sensitivity of 1 ng/mL. Prenatal exposure to tobacco was dichotomized as exposed (cotinine ≥ the limit of detection [LOD]; 0.05 ng/mL) and not exposed (cotinine < LOD). Mothers were asked to report the number of adults in the household (including themselves) who were regular smokers when their child was 5 months of age. Responses to this question ranged from 0 to 6. Self‐report of household cigarette smokers was dichotomized as no household smokers or any household smokers. Breastfeeding for at least 5 months was ascertained by asking mothers whether they were currently breastfeeding at the 5‐month visit.
Statistical analysis
Generalized linear models were used to estimate the association between prenatal exposure to Δ9‐THC and birthweight, fat mass, fat‐free mass, and adiposity. Covariates were selected by using a directed acyclic diagram. Our base models adjusted for maternal age, household income, gravidity, gestational age at urine collection, gestational diabetes, maternal psychiatric disorder, pre‐pregnancy BMI, gestational weight gain, offspring sex, gestational age at birth, and child race and ethnicity. We additionally adjusted for prenatal exposure to tobacco because co‐use of tobacco and cannabis is commonref. ijpo13187-bib-0028 and tobacco is independently associated with child growth.ref. ijpo13187-bib-0029 We further included birth length as a precision variable, given that neonatal body composition may vary by birth length. We present adjusted beta coefficients and means with corresponding 95% confidence intervals (CIs) for all models.
A mixed‐effects regression model examined the longitudinal association between prenatal exposure to Δ9‐THC with postnatal growth trajectories (1–36 months). We modeled both BMI trajectories and BMI z‐score trajectories to leverage the advantages of each approach. BMI z‐score trajectories allow for comparison of the child’s BMI relative to peers over time. This is particularly useful for paediatric populations, because boys and girls follow different growth trajectories.ref. ijpo13187-bib-0030 On the other hand, BMI trajectories describe the child’s BMI change over timeref. ijpo13187-bib-0031 while also allowing for a simpler interpretation. Based on the deviance information criteria, a square root transformation of age yielded the best fitting trajectory. We used Wald tests with Kenward–Roger degrees of freedom and an unstructured covariance matrix. To test for interaction by breastfeeding, we introduced a three‐way product term (Δ9‐THC × breastfeeding × the square root of age) and lower order product terms in a mixed‐effects regression model. The mixed‐effects regression models adjusted for all covariates listed above, as well as self‐report of household smokers at age 5 months.
All statistical analyses were conducted using Stata, Version 14.2 (StataCorp LP, College Station, TX, USA). An alpha level of 0.05 was used to determine statistical significance for all analyses, including interactions.
RESULTS
Of the 1410 participants initially enrolled in Healthy Start, 199 participants had available urine samples for Δ9‐THC/CBD analysis. The adiposity analytic sample was 128 participants, after excluding 49 participants who did not undergo the PEA POD assessment and 22 participants with missing covariate data. The trajectory analytic sample was 140 participants, after excluding 8 with insufficient length/height and weight measurements and 51 participants with missing covariate data. As compared to the overall cohort (n = 1410), mothers in the adiposity analytic sample (n = 128) were slightly older, had higher incomes, and were less likely to have prenatal exposure to tobacco (Table 1).
TABLE 1: Characteristics of eligible mothers and children in the Healthy Start study.
| Prenatal exposure to Δ9‐THCijpo13187-note-0003 | ||||
|---|---|---|---|---|
| All (n = 128) | Not exposed (n = 113) | Exposed (n = 15) | p‐value | |
| Mother characteristics | ||||
| Age (years) | 30 ± 6 | 30.3 ± 5.5 | 26.3 ± 5.4 | p < 0.01 |
| Gravidity | 1.4 ± 1.4 | 1.4 ± 1.4 | 1.5 ± 1.5 | p = 0.80 |
| Pre‐pregnancy BMI (kg/m2) | 24.8 ± 4.6 | 24.8 ± 4.7 | 24.8 ± 4.4 | p = 0.98 |
| Gestational weight gain (kg) | 14.8 ± 5.2 | 14.3 ± 4.9 | 18.1 ± 6.2 | p < 0.01 |
| Gestational diabetes (yes/borderline) | 17 (13%) | 15 (13%) | 2 (13%) | p = 0.96 |
| Household income ≥$70 000 | 56 (44%) | 53 (47%) | 3 (20%) | p = 0.11 |
| Some college education | 98 (77%) | 91 (81%) | 7 (47%) | p = 0.01 |
| Prenatal exposure to tobacco (maternal cotinine ≥ LOD) | 19 (15%) | 9 (8%) | 10 (67%) | p < 0.01 |
| Gestational age at urine collection (weeks) | 26.5 ± 2.7 | 26.6 ± 2.7 | 25.8 ± 2.9 | p = 0.71 |
| Race and ethnicity | ||||
| Hispanic | 33 (26%) | 31 (27%) | 2 (13%) | |
| Non‐Hispanic black | 13 (10%) | 7 (6%) | 6 (40%) | |
| Asian | 11 (9%) | 9 (8%) | 2 (13%) | |
| Pacific Islander | ||||
| American Indian or Alaska Native | ||||
| Non‐Hispanic white | 71 (56%) | 66 (58%) | 5 (33%) | p < 0.01 |
| Child characteristics | ||||
| Female | 73 (57%) | 62 (55%) | 11 (73%) | p = 0.18 |
| Gestational age at birth (weeks) | 39.5 ± 1.2 | 39.5 ± 1.2 | 39.4 ± 1.2 | p = 0.76 |
| Breastfeeding at age 5 months (yes) | ||||
| Household smokers at 5 months (any) | 12 (10%) | 9 (8%) | 3 (21%) | p = 0.13 |
Note: Continuous variables are expressed as means ± standard deviation. Independent samples t‐tests were used to examine the differences in means by cannabis categories. Categorical variables are expressed as proportions of column totals. Chi‐square tests were used to examine differences in proportions by cannabis categories.
Abbreviations: Δ9‐THC, delta 9‐tetrahydrocannabinol; BMI, body mass index; LOD, limit of detection.
The categories of were as follows: not exposed (Δ9‐THC below the LOD) and exposed (Δ9‐THC exceeded the LOD).
Within the adiposity analytic sample (n = 128), over half (51%) of the children were identified as non‐Hispanic White, 31% identified as Hispanic, 9% identified as non‐Hispanic Black, 2% identified as Asian, 2% identified as American Indian or Alaska native, and 6% identified as being multiracial. Most of the women had some college education (77%) and many had a household income ≥$70 000 (44%).
In the adiposity analytic sample (n = 128), 12% (n = 15) had detectable Δ9‐THC metabolites at ~27 weeks gestation (Table 2). Of these, three participants had concomitant CBD exposure. Pregnant people with Δ9‐THC exposure tended to be younger with less education and higher gestational weight gain. Δ9‐THC‐exposed children were breastfed for a shorter duration, were more likely to have prenatal and postnatal exposures to tobacco and were more likely to identify as Hispanic or non‐Hispanic White (Table 1). There were no differences in gravidity, pre‐pregnancy BMI, gestational diabetes, gestational age at urine collection, gestational age at birth, or offspring sex.
Neonates with prenatal exposure to Δ9‐THC alone had lower fat mass (−95 g; 95% CI: −174, −14; p = 0.05) and adiposity (−2.1%; 95% CI: −4.2, −0.04, p = 0.04), as compared to non‐exposed neonates (Table 3). However, these associations were slightly attenuated following adjustment for prenatal exposure to tobacco (Model 3) and further attenuated following adjustment for birth length (Model 4). There was no difference in birthweight or neonatal fat‐free mass according to prenatal exposure to Δ9‐THC.
TABLE 3: Adjustedijpo13187-note-0007 means and mean differences for the associations between prenatal exposure to Δ9‐THC and neonatal body composition, n = 128.
| Adjusted means and beta coefficients | |||||
|---|---|---|---|---|---|
| Δ9‐THC categories | n | Birth weight (g) | Fat‐free mass (g) | Fat mass (g) | Adiposity (% fat mass) |
| Model 1: Δ9‐THC and CBDijpo13187-note-0008 | |||||
| Not exposed | 113 | 3333 (3264, 3401) | 2905 (2859, 2953) | 317 (288, 346) | 9.5% (8.8, 10.3) |
| Exposed | 15 | −160 (−376, 56), p = 0.15 | −110 (−262, 41), p = 0.15 | −73 (−145, 0), p = 0.05 | −1.5 (−3.5, 0.3), p = 0.09 |
| Model 2: Δ9‐THC aloneijpo13187-note-0009 | |||||
| Not exposed | 116 | 3343 (3267, 3403) | 2895 (2847, 2945) | 317 (289, 346) | 9.8% (8.9, 10.6) |
| Exposed | 12 | −216 (−458, 27), p = 0.08 | −157 (−331, 17), p = 0.08 | −95 (−174, −14), p = 0.02 | −2.1 (−4.2, −0.04), p = 0.04 |
| Model 3: Δ9‐THC aloneijpo13187-note-0010 | |||||
| Not exposed | 116 | 3331 (3263, 3399) | 2891 (2843, 2938) | 317 (287, 347) | 9.6% (8.8, 10.3) |
| Exposed | 12 | −176 (−445, 92), p = 0.20 | −101 (−283, 81), p = 0.28 | −68 (−152, 16), p = 0.11 | −2.3 (−4.8, 0.03), p = 0.08 |
| Model 4: Δ9‐THC aloneijpo13187-note-0011 | |||||
| Not exposed | 116 | 3320 (3257, 3384) | 2882 (2839, 2924) | 314 (286, 342) | 9.5% (8.7, 10.2) |
| Exposed | 12 | −62 (−339, 216), p = 0.66 | −4 (−194, 185), p = 0.96 | −44 (−132, 44), p = 0.33 | −1.3 (−3.9, 1.2), p = 0.31 |
Abbreviations: Δ9‐THC, delta 9‐tetrahydrocannabinol; CBD, cannabidiol; CI, confidence interval; LOD, limit of detection.
Models 1 and 2 adjusted for maternal age (years), household income (≥$70 000, <$70 000 or missing/declined to answer), maternal education (High school degree or less, some college), maternal race/ethnicity (Hispanic, non‐Hispanic black, non‐Hispanic other, and non‐Hispanic white), gravidity, gestational diabetes (no, borderline/yes), age at urine collection (weeks), pre‐pregnancy body mass index (kg/m2), gestational weight gain (kg), offspring sex, and gestational age at birth (weeks).
The categories were as follows: not exposed (Δ9‐THC and its metabolites below the LOD) and exposed (Δ9‐THC and/or any of its metabolites exceeded the LOD).
The categories were as follows: not exposed (Δ9‐THC was not detected in the absence of CBD) and exposed (Δ9‐THC alone was detected, without CBD).
Model 3 adjusted for prenatal exposure to tobacco (cotinine measured in maternal urine at 27 weeks gestation<LOD, cotinine>LOD), in addition to all other variables in Model 2.
Model 4 adjusted for birth length (cm), in addition to all other variables in Model 3.
Compared to children with no exposure, children with prenatal exposure to Δ9‐THC experienced a more rapid BMI z‐score trajectory (Figure 1; 0.42 increase in BMI z‐score per year; 95% CI: 0.12, 0.72). This association was independent of prenatal or postnatal exposure to tobacco. A similar but non‐significant pattern was observed for BMI trajectories (Figure S1).
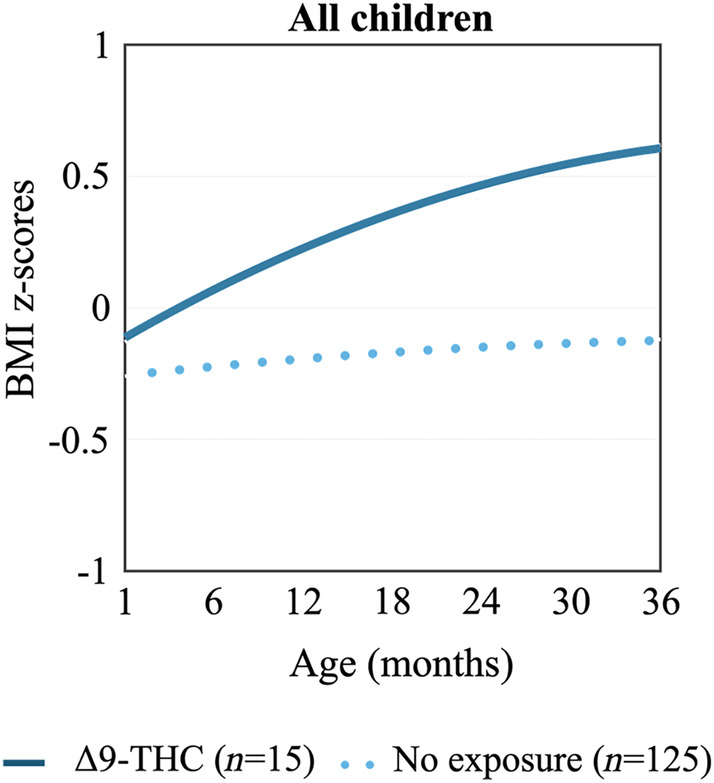
Breastfeeding modified the association between prenatal exposure to Δ9‐THC and postnatal BMI z‐score trajectories (Figure 2; p for three‐way interaction = 0.04). For example, Δ9‐THC did not appear to influence growth among those breastfed for 5 months whereas a shorter duration of breastfeeding was associated with 1.1 higher BMI z‐score at 36 months (95% CI: 0.21, 2.05).
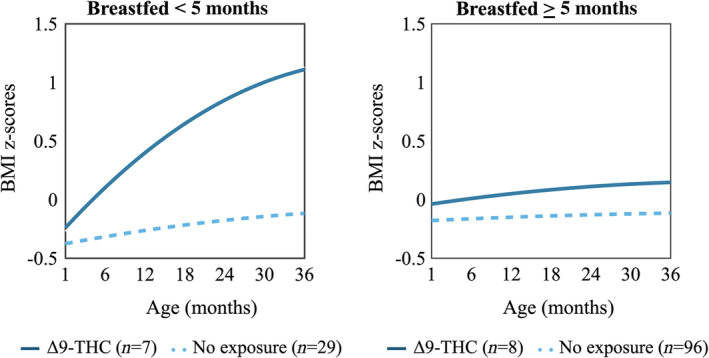
Similarly, breastfeeding modified the association between Δ9‐THC and postnatal BMI trajectories, but the interaction p‐value was not statistically significant (Figure S2).
DISCUSSION
Our results suggest that prenatal exposure to Δ9‐THC, particularly in the absence of CBD, is associated with a smaller size at birth followed by rapid postnatal growth. Our findings further suggest that the association between prenatal exposure to Δ9‐THC and postnatal growth trajectories is modified by breastfeeding, such that the longer the offspring was breastfed, the less prenatal exposure to Δ9‐THC impacted postnatal growth.
Prenatal exposure to Δ9‐THC, in the absence of CBD, influenced birth size. We speculate that this may be due to the distinct effects of the cannabinoids. Consistent with our pilot study, animal models have demonstrated that prenatal exposure to Δ9‐THC is associated with lower birthweight followed by rapid postnatal growth.ref. ijpo13187-bib-0013, ref. ijpo13187-bib-0014 Conversely, the effect of CBD on offspring growth is not clear, with animal models showing little to no impact on birthweightref. ijpo13187-bib-0018, ref. ijpo13187-bib-0019 or its effects on postnatal growth appear to be sex‐specific.ref. ijpo13187-bib-0021, ref. ijpo13187-bib-0022 The biological effects of Δ9‐THC and CBD on fetal growth may involve activation of CB1 and CB2 receptors, which influences brain, pancreas, and adipose tissue development of the fetus. Since Δ9‐THC has a higher affinity for CB1 and CB2 receptors than CBD, this could explain the seemingly stronger effects of Δ9‐THC on fetal growth we observed in this study. However, new evidence has emerged to suggest that Δ9‐THC and CBD interact with other non‐canonical receptors, such as inhibiting placental growth factor (PlGF) and vascular endothelial growth factor (VEGF) signaling pathwaysref. ijpo13187-bib-0032, ref. ijpo13187-bib-0033 (contributing to insufficient placental angiogenesis, a known driver of fetal growth restriction).ref. ijpo13187-bib-0034 While our study is a first step in understanding the independent effects of Δ9‐THC and CBD, considerable research is needed to address the paucity of data surrounding prenatal exposure to CBD, in the absence of Δ9‐THC, on child growth.
Our study results further suggest that Δ9‐THC‐exposed offspring grew more rapidly in early life. While some catch up growth is expected, over‐compensatory growth is generally a result of intrauterine growth restriction and increases the risk of metabolic diseases later in life.ref. ijpo13187-bib-0035 Findings from this study further suggest that a longer duration of breastfeeding may serve as a potential strategy for the normalizing growth among cannabis‐exposed offspring. Breastfeeding has numerous well‐documented benefits for both mother and infant. Breastfeeding may improve satiety, reduce problematic feeding behaviours, and/or provide cannabis‐exposed offspring with bioactive factors that have the potential to regulate growth.ref. ijpo13187-bib-0036 As such, any duration of exclusive breastfeeding tends to lower the future risk for obesity.ref. ijpo13187-bib-0037 However, these effects may be negated if the lactating person uses cannabis. Δ9‐THC accumulates in breastmilk and may be present up to 6 weeks following use.ref. ijpo13187-bib-0038, ref. ijpo13187-bib-0039 Furthermore, cannabinoids may alter lipid profiles of breastmilk.ref. ijpo13187-bib-0040 While our results suggest that a longer duration of breastfeeding may protect against rapid postnatal growth, we did not specifically measure cannabinoids in breastmilk, as breastmilk was not collected in our study. Therefore, future studies are needed to quantify exposure via breastmilk and to determine the extent to which lactational exposure to Δ9‐THC impacts growth.
Tobacco is commonly used with cannabis ref. ijpo13187-bib-0028 which makes it difficult to disentangle the effects of each exposure on child growth. While most studies report that the associations between prenatal exposure to cannabis and low birthweight are independent of tobacco,ref. ijpo13187-bib-0001, ref. ijpo13187-bib-0003, ref. ijpo13187-bib-0004, ref. ijpo13187-bib-0005, ref. ijpo13187-bib-0006, ref. ijpo13187-bib-0008 some studies report null associations after adjustment for tobacco.ref. ijpo13187-bib-0007, ref. ijpo13187-bib-0009 In our regression models, the associations between prenatal exposure to cannabis and birth size were attenuated following adjustment for tobacco. This may be due to tobacco explaining some of the same variance (as evidenced by the effect estimates moving towards the null) or a reduction in model precision (as evidenced by the widening of the confidence intervals). By contrast, in our mixed‐effects models, adjustment for prenatal and postnatal exposure to tobacco did not impact the results. This may suggest that prenatal exposure to tobacco may be less impactful on BMI trajectories as children age. Tobacco may also be an effect modifier. However, we lacked the sample size to explore the interaction between tobacco and cannabis. Prospective cohorts with sufficiently large subgroups of children with objective assessment of both cannabis and tobacco exposure during pregnancy are needed to understand the impact of polysubstance use on child growth.
Our study has some limitations. First, our study is limited by the cannabis exposure assessment. First, cannabinoids were measured only once at 27 weeks gestation. The urinary half‐life of Δ9‐THC‐metabolites is relatively short, ranging from 20 h (for light, infrequent users) to 10 days (for heavy, frequent use).ref. ijpo13187-bib-0041 Thus, our one‐time measure may fail to capture exposures early in pregnancy (when many people are just learning of their pregnancies) or during late gestation (when the majority of offspring adipose tissue growth occursref. ijpo13187-bib-0042) but more likely to capture heavy, frequent use. If the exposure misclassification were differential with respect to our outcome of interest, this may have biased the results away from the null. Second, cannabinoid concentrations may vary by urinary dilution between people. This is often overcome by correcting for urinary creatinine. However, creatinine was not measured as part of this study. Nevertheless, we believe that this represents a minor limitation to our approach since the goal of our study was to compare offspring with and without Δ9‐THC exposure rather than quantify a dose–response relationship. Finally, as described, we did not capture postnatal exposure, thus it is unclear if there are developmental windows when the child is more susceptible to cannabis.
Second, we lacked information on some important confounders. For instance, the use of alcohol and other substances during pregnancy have been linked to foetal growth restriction and may be used along with cannabis.ref. ijpo13187-bib-0043 Not adjusting for these confounders may have biased the results away from the null. Furthermore, although we adjusted for many relevant covariates, there is potential for residual confounding. In fact, it has been suggested that the associations between prenatal exposure to cannabis on offspring health outcomes may be explained by shared family‐based social and lifestyle factors, rather than by direct fetal programming.ref. ijpo13187-bib-0044 However, given the consistency of animal modelsref. ijpo13187-bib-0013, ref. ijpo13187-bib-0014, ref. ijpo13187-bib-0015, ref. ijpo13187-bib-0016, ref. ijpo13187-bib-0017 and epidemiologic studies,ref. ijpo13187-bib-0001, ref. ijpo13187-bib-0002, ref. ijpo13187-bib-0003, ref. ijpo13187-bib-0004, ref. ijpo13187-bib-0005, ref. ijpo13187-bib-0006, ref. ijpo13187-bib-0007, ref. ijpo13187-bib-0008, ref. ijpo13187-bib-0009, ref. ijpo13187-bib-0010 we conclude that prenatal exposure to Δ9‐THC is likely to be an important risk factor for altered prenatal and postnatal growth of the offspring.
Finally, our approach is limited by the small sample size and potential for selection bias for those who completed the follow‐up visits. Our analytic sample included mothers who were slightly older, had higher household incomes, were less likely to smoke cigarettes, and were more likely to have female offspring.
CONCLUSIONS
Over the past few decades, there has been a major shift in legality and attitudes towards cannabis use during pregnancy. Concurrently, there has also been a dramatic increase in the potency of cannabis.ref. ijpo13187-bib-0020 These trends are worrisome, as evidence suggests that cannabis use in pregnancy may contribute to adverse health effects in the offspring. Here, we found that prenatal exposure to Δ9‐THC, without evidence of CBD exposure, was associated with fetal growth restriction followed by rapid growth in early life. These effect estimates were non‐trivial. Δ9‐THC exposure was associated with a 216‐g reduction in birthweight and a 2.1% reduction in neonatal adiposity, which is comparable to the effect sizes of other known risk factors for low birthweight, such as malnutritionref. ijpo13187-bib-0045 or smokingref. ijpo13187-bib-0046 during pregnancy. A longer duration of exclusive breastfeeding may stabilize rapid growth among Δ9‐THC‐exposed offspring, but the impact of lactational exposure to Δ9‐THC requires further investigation. Considering the increasing use and acceptance of cannabis, these findings highlight the need for public health messaging surrounding cannabis use during pregnancy and while breastfeeding.
AUTHOR CONTRIBUTIONS
B.F.M. initiated the cannabinoid analysis for this study, analysed the data, and wrote each draft of the manuscript. C.S., J.K., and U.C. performed the cannabinoid analyses and made substantial contributions to the interpretation of the cannabinoid exposure assessment. provided substantial feedback on the interpretation of these measures. N.T.M., W.P., K.A.S., E.T.H., A.T.H., E.M.W., K.E.B., E.J.S., A.L.B.S., G.K., and D.D. provided critical feedback on the clinical and mechanistic interpretation of the findings.
FUNDING INFORMATION
This work was supported by the National Institutes of Health (R01DK076648, UH3OD023248, R01ES022934, R00ES028711, UL1TR003167, R21DA061104).
CONFLICT OF INTEREST STATEMENT
The authors declare no conflicts of interest.
CONSENT TO PARTICIPATE
Informed consent was obtained prior to participation in the parent study, which includes analysis of stored biospecimens.
Supplementary Materials
References
- Intrauterine cannabis exposure affects fetal growth trajectories: the generation R study.. J Am Acad Child Adolesc Psychiatry., 2009. [PubMed]
- Marijuana impairs growth in mid‐gestation fetuses.. Neurotoxicol Teratol., 2005. [PubMed]
- Cannabis use during the perinatal period in a state with legalized recreational and medical marijuana: the association between maternal characteristics, breastfeeding patterns, and neonatal outcomes.. J Pediatr., 2018. [PubMed]
- Association between maternal cannabis use and birth outcomes: an observational study.. BMC Pregnancy Childbirth., 2020. [PubMed]
- The deleterious effects of cannabis during pregnancy on neonatal outcomes.. Med J Aust., 2020. [PubMed]
- Cannabis use during pregnancy and risk of adverse birth outcomes: a longitudinal cohort study.. Eur Addict Res., 2021. [PubMed]
- Maternal use of cannabis and pregnancy outcome.. BJOG., 2002. [PubMed]
- Effect of marijuana use in pregnancy on fetal growth.. Am J Epidemiol., 1986. [PubMed]
- The association of marijuana use with outcome of pregnancy.. Am J Public Health., 1983. [PubMed]
- Impact of pregnancy marijuana use on birth outcomes: results from two matched population‐based cohorts.. J Perinatol., 2020. [PubMed]
- Effects of maternal marijuana and cocaine use on fetal growth.. N Engl J Med., 1989. [PubMed]
- 12 Pertwee RG . The diverse CB1 and CB2 receptor pharmacology of three plant cannabinoids: delta9‐tetrahydrocannabinol, cannabidiol and delta9‐tetrahydrocannabivarin. Br J Pharmacol. 2008;153(2):199‐215.
- Δ9‐tetrahydrocannabinol exposure during rat pregnancy leads to symmetrical fetal growth restriction and labyrinth‐specific vascular defects in the placenta.. Sci Rep., 2020. [PubMed]
- Maternal exposure to Δ9‐tetrahydrocannabinol impairs female offspring glucose homeostasis and endocrine pancreatic development in the rat.. Reprod Toxicol., 2020. [PubMed]
- In utero exposure to Δ9‐tetrahydrocannabinol leads to postnatal catch‐up growth and dysmetabolism in the adult rat liver.. Int J Mol Sci., 2021
- Exposure to Δ9‐tetrahydrocannabinol during rat pregnancy leads to impaired cardiac dysfunction in postnatal life.. Pediatr Res., 2021. [PubMed]
- Maternal Delta‐9‐tetrahydrocannabinol exposure induces abnormalities of the developing heart in mice.. Cannabis Cannabinoid Res., 2022. [PubMed]
- Gestational exposure to cannabidiol leads to glucose intolerance in 3‐month‐old male offspring.. J Endocrinol., 2024
- Maternal administration of cannabidiol promotes an anti‐inflammatory effect on the intestinal wall in a gastroschisis rat model.. Braz J Med Biol Res., 2018. [PubMed]
- Changes in cannabis potency over the last 2 decades (1995–2014): analysis of current data in the United States.. Biol Psychiatry., 2016. [PubMed]
- In utero exposure to cannabidiol disrupts select early‐life behaviors in a sex‐specific manner.. Transl Psychiatry., 2022. [PubMed]
- Developmental cannabidiol exposure increases anxiety and modifies genome‐wide brain DNA methylation in adult female mice.. Clin Epigenetics., 2021. [PubMed]
- Prenatal tobacco and cannabis co‐exposure and offspring obesity development from birth to mid‐childhood.. Pediatr Obes., 2023. [PubMed]
- 24 American Academy of Pediatrics Committee on Nutrition . Pediatric Nutrition Handbook. 6th ed. American Academy of Pediatrics; 2008.
- Growth of breast‐fed and formula‐fed infants from 0 to 18 months: the DARLING study.. Pediatrics., 1992. [PubMed]
- Protective effect of breastfeeding on the adverse health effects induced by air pollution: current evidence and possible mechanisms.. Int J Environ Res Public Health., 2019. [PubMed]
- Simultaneous quantification of 17 cannabinoids bY LC‐MS‐MS in human plasma.. J Anal Toxicol., 2021
- Marijuana and tobacco co‐use among a nationally representative sample of US pregnant and non‐pregnant women: 2005–2014 National Survey on Drug Use and Health findings.. Drug Alcohol Depend., 2017. [PubMed]
- Maternal smoking during pregnancy and child overweight: systematic review and meta‐analysis.. Int J Obes., 2008
- Sex‐specific differences in fetal and infant growth patterns: a prospective population‐based cohort study.. Biol Sex Differ., 2016. [PubMed]
- Body mass index trajectories in the first 5 years and associated antenatal factors.. Front Pediatr., 2021. [PubMed]
- 32 Blázquez C, González‐Feria L, Alvarez L, Haro A, Casanova ML, Guzmán M . Cannabinoids inhibit the vascular endothelial growth factor pathway in gliomas. Cancer Res. 2004;64(16):5617‐5623.
- 33 Derkinderen P, Valjent E, Toutant M, Corvol JC, Enslen H, Ledent C, Trzaskos J, Caboche J, Girault JA . Regulation of extracellular signal‐regulated kinase by cannabinoids in hippocampus. J Neurosci. 2003;23(6):2371‐2382.
- 34 Malamitsi‐Puchner A, Boutsikou T, Economou E, Sarandakou A, Makrakis E, Hassiakos D, Creatsas G . Vascular endothelial growth factor and placenta growth factor in intrauterine growth‐restricted fetuses and neonates. Mediators Inflamm. 2005;2005(5):293‐297.
- Being big or growing fast: systematic review of size and growth in infancy and later obesity.. BMJ., 2005. [PubMed]
- Mechanisms underlying the association between breastfeeding and obesity.. Int J Pediatr Obes., 2009. [PubMed]
- Relationship between exclusive breastfeeding and lower risk of childhood obesity: a narrative review of published evidence.. Clin Med Insights Pediatr., 2017. [PubMed]
- Marijuana use by breastfeeding mothers and cannabinoid concentrations in breast milk.. Pediatrics., 2018
- Persistence of Δ‐9‐tetrahydrocannabinol in human breast milk.. JAMA Pediatr., 2021. [PubMed]
- Cannabinoids accumulate in mouse breast milk and differentially regulate lipid composition and lipid signaling molecules involved in infant development.. BBA Adv., 2022
- Human cannabinoid pharmacokinetics.. Chem Biodivers., 2007. [PubMed]
- Adipose tissue and fetal programming.. Diabetologia., 2012. [PubMed]
- Alcohol, tobacco, cocaine, and marijuana use: relative contributions to preterm delivery and fetal growth restriction.. Subst Abus., 2014. [PubMed]
- Foetal tobacco and cannabis exposure, body fat and cardio‐metabolic health in childhood.. Pediatr Obes., 2021. [PubMed]
- Maternal diet quality in pregnancy and neonatal adiposity: the Healthy Start Study.. Int J Obes., 2005
- Fetal exposure to maternal active and secondhand smoking with offspring early‐life growth in the Healthy Start study.. Int J Obes (Lond)., 2019. [PubMed]
