Structural changes of tubulin by interacting with Δ9-tetrahydrocannabinol: in-vitro and theoretical studies
Abstract
Background:
There is growing evidence of the contribution of microtubule dynamics to dendritic spine changes, synaptic plasticity, axonal transportation, and cell polarity. Besides, one of the well-studied effects of Cannabis on human behavior is memory disability. As Δ9-tetrahydrocannabinol (Δ9-THC) is the most pivotal chemical of Cannabis, we investigated the effect of Δ9-THC on microtubule dynamicity and the structural study of tubulin (microtubule monomer).
Results:
Our results show that Δ9-THC changes microtubule dynamicity compared to the control group. The turbidity assay results demonstrated that Δ9-THC reduces microtubule polymerization in a concentration-dependent manner. Circular Dichroism spectroscopy also studied the structural changes of the purified tubulin, which revealed significant changes in the secondary structure of the tubulin. Furthermore, Silico studies predicted one binding site for Δ9-THC on β-tubulin.
Conclusions:
We concluded that Δ9-THC could reduce the microtubule’s stability, which may conversely affect brain function by microtubule dynamic changes caused by secondary structural changes of tubulin and preventing tubulin-tubulin interaction.
Supplementary Information:
The online version contains supplementary material available at 10.1186/s12868-025-00957-5.
Article type: Research Article
Keywords: Microtubule dynamicity, Δ, Tubulin structure
Affiliations: https://ror.org/05vf56z40grid.46072.370000 0004 0612 7950Department of Biochemistry, Institute of Biochemistry and Biophysics (IBB), University of Tehran, Tehran, Iran; https://ror.org/03hh69c200000 0004 4651 6731Cinna Gen Medical Biotechnology Research Center, Alborz University of Medical Science, Karaj, Iran; https://ror.org/02twggb97grid.495554.c0000 0005 0272 3736Department of Microbial Biotechnology, Faculty of Biotechnology, Amol University of Special Modern Technologies, Amol, Iran; https://ror.org/02twggb97grid.495554.c0000 0005 0272 3736Faculty of Engineering Technology, Amol University of Special Modern Technologies, Amol, Iran
License: © The Author(s) 2025 CC BY 4.0 Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
Article links: DOI: 10.1186/s12868-025-00957-5 | PubMed: 40739185 | PMC: PMC12312531
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (1.3 MB)
Introduction
Microtubules are cytoskeletal filaments in eukaryotic cells, made up of α and β subunits [ref. 1], and are polarized polymeric tubes with dynamic ends [ref. 2]. Microtubules are essential for numerous aspects of the cell’s function. Cell morphogenesis, chromosome segregation, and intracellular vesicle transport are critical roles of microtubules [ref. 3, ref. 4].
Furthermore, microtubules are critical structural and functional connectivity structures across the neural network and are involved in signal transportation along the axons [ref. 5, ref. 6]. Dynamic microtubules are involved in dendritic spine changes and synaptic plasticity [ref. 7]. Microtubules’ motor proteins (dynein) are involved in the trafficking of intracellular subunits [ref. 8–ref. 10]. Different research groups have studied the structural changes that lead to the functional alteration of microtubules [ref. 11–ref. 13].
Δ9-Tetrahydrocannabinol (Δ9-THC) and cannabidiol (CBD), two major cannabinoids from Cannabis sativa, influence the cytoskeleton, particularly microtubules. Research on the direct interaction between the two ingredients and tubulin is limited. However, some studies have explored the effects of cannabinoids on the cytoskeleton, particularly microtubules composed of tubulin proteins. CBD has been observed to disrupt the organization of microtubules and microfilaments in PC12 cells, leading to a reduction in cytoskeletal integrity in a dose-dependent manner [ref. 14]. cannabinoid agonist affect acetylation of α-tubulin and gene expression in the prefrontal cortex of adult mice [ref. 15].
Δ9-THC is one of the most critical and psychoactive ingredients of Cannabis. It also impairs several aspects of cognitive functions, with the most robust effect on humans’ short-term, episodic, and working memory [ref. 16, ref. 17].
The negative effect of Δ9-THC on synaptic plasticity is well-accepted [ref. 18, ref. 19], and numerous studies have focused on this impairment’s signaling pathways, which are poorly understood [ref. 20–ref. 23]. Daniel Jimenez et al. reported that the activation of mouse astroglia type-1 cannabinoid receptors associated with mitochondrial membranes (mtCB1) disturbs glucose metabolism and lactate production in the brain. This leads to an alteration in neuronal functions [ref. 24]. Other results show that Δ9-THC-induced apoptosis in glioma cells may be independent of CB1 receptors and due to the THC-stimulated hydrolysis of sphingomyelin in glioma cells [ref. 25]. It has been shown that the long-term administration of Δ9-THC in laboratory animals impairs microtubule dynamicity [ref. 26].
According to previous studies on the direct role of microtubules in synaptic plasticity, cell structure, and the proven effects of Δ9-THC on the cell function, it can be presumed that Δ9-THC could exert its effect through changes in microtubule dynamicity along with other CB1 receptor-related mechanisms signaling pathway. To study this assumption, following our previous study of the systemic administration of Δ9-THC to rats [ref. 26], we examined the interaction between Δ9-THC and microtubule protein kinetically using a set of spectrophotometric techniques. Our results indicated that Δ9-THC has a binding site on tubulin and changes the dynamic behavior of the protein by inducing structural changes in the tubulin dimers.
Materials and methods
Chemicals
Δ9-THC (Cat# T4764) in methanol, prepared in a vehicle containing 4% DMSO and 96% saline (sterile saline 9%). Guanosine 5′-triphosphate (GTP, Cat# G0635), Piperazine-N, N′-bis (2-ethane sulfonic acid) (PIPES, Cat# P6757), EGTA (Cat# E3889), MgSO4 (Cat# E3889), Dimethyl sulfoxide (DMSO, Cat# D2650), and other chemicals were purchased from Merck.
Animals
Twenty-five adult Wistar rats, weighing 200–250 g, were purchased from the Institute of Biochemistry and Biophysics, University of Tehran, Tehran, Iran. The animals were kept under 12 h light/dark periods with access to standard food and water at the Neuroscience Research Center at the Institute of Biochemistry and Biophysics, University of Tehran, Tehran, Iran. The animals were anesthetized with an intraperitoneal injection of ketamine/xylazine (80 mg/kg and 10 mg/kg, respectively) as previously described [ref. 27]. The brain was quickly removed, and all brain tissue was fixed in a -80 °C for the next experiments, and the remaining carcasses were disposed of at the IBB animal care center.
Rat brain tubulin extraction and purification
Tubulin was purified from the rat brain in two cycles of polymerization and depolymerization [ref. 28, ref. 29]. For further purification, we applied cellulose phosphate column chromatography to remove MAPs for structural studies of the purified tubulins. The purity of the obtained fractions was determined by Coomassie blue staining of 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE).
Protein kinetics via turbidity measurement
The tubulin assembly assay was performed at 37 °C in the presence of a final concentration of 1 mM GTP solution in double-distilled water with a UV/visible spectrophotometer (at 350 nm wavelength), which was an adjustable temperature device [ref. 30]. To examine the effect of Δ9-THC on microtubule and tubulin assembly, the protein was pre-incubated with different concentrations of Δ9-THC for 2 min, and GTP was added to the solution to start the assay.
Kinetic parameters of the tubulin polymerization
We followed the polymerization assay at 350 nm, as mentioned above. Information about the nucleation, lag phase, and elongation obtained from the polymerization curve [ref. 31] was assessed. The nucleation phase was studied with two different parameters, including (i) the tenth time or “t1/10” which refers to the necessary time to produce 1/10 of the final amount of the polymer, and (ii) parameter p, known as the number of successive steps in the nucleation process. The parameter p was used for estimating the nucleus size, which can be characterized by plotting log A(t)/Amax against log(t) where A(t) is the absorbance at a given time and calculating the slope of the plot [ref. 32].
We also studied the elongation phase by plotting (1- At /Amax) against the time, which provides a first-order rate constant of elongation (kobs). To analyze the tubulin assembly, we incubated the tubulin purified by cellulose phosphate column chromatography. Purified tubulin (30 µM) was incubated with different concentrations of Δ9-THC (0–1 µM) to test the direct interaction of Δ9-THC with tubulin in the absence of MAPs.
Intrinsic fluorescence spectroscopy
The tendency of Δ9-THC to tubulins was determined by the intrinsic fluorescence of tubulins, which was detected in the presence of different concentrations (0–1 µM) of Δ9– THC. The emission of tubulins was detected at 295 nm excitation in the range of 300–500 nm [ref. 33]. The abovementioned experiments were carried out using the Cary Eclipse fluorescence spectrophotometer (Varian, Australia).
Circular dichroism spectroscopy (CD)
The CD spectroscopy assay was performed by a model 215 circular Dichroism (Aviv Biomedical, USA). In this procedure, the tubulin dimers were pre-incubated with different concentrations of Δ9-THC (0–1 µM). The far UV CD spectra were recorded from 190 to 260 nm using a 1 mm path length quartz cuvette. Moreover, the secondary structural changes in tubulin were analyzed by the CDNN program.
Molecular Docking studies
Molecular docking was performed using AutoDock tools version 1.5.6 with standard parameters. The crystal structure of tubulin was obtained from the Brookhaven Protein Data Bank (PDB entry 1JFF), and the ligand-free structure was used as the initial protein structure [ref. 34]. The ligand structure was obtained from Pub Chem (Pub Chem CID 16078). The grid box was defined (100_ 86 _74 Å) for a general pre-calculation of the interaction of Δ9-THC over the whole macromolecule, grid maps with a spacing of 0.375 Å in each direction, and the center of the grid was set to 19.17,0 and 0 Å. After recognizing the approximate binding site of Δ9-THC on tubulin, flexible docking of Δ9-THC over the β-tubulin binding site was performed. The grid box was defined as 40_40_40 Å, and the center of the grid was set to x = 3.641, y = -16.032, and z = 19.64. Images were created using Python Molecule Viewer (PMV), PyMol molecular viewer v.1.1, and the program Lig Plot v.1.0, which generates schematic 2-D representations of protein-ligand complexes from the PDB file input [ref. 35].
Statistical analysis
The analysis of the data obtained was performed using SPSS software version 16. One-way analysis of variance (ANOVA) was performed, and Tukey post hoc analysis was performed to compare the means of the experimental groups. All results are expressed as mean ± SEM, and differences were considered significant at P < 0.05, P < 0.01, and P < 0.001.
Results
Rat brain tubulin extraction and purification
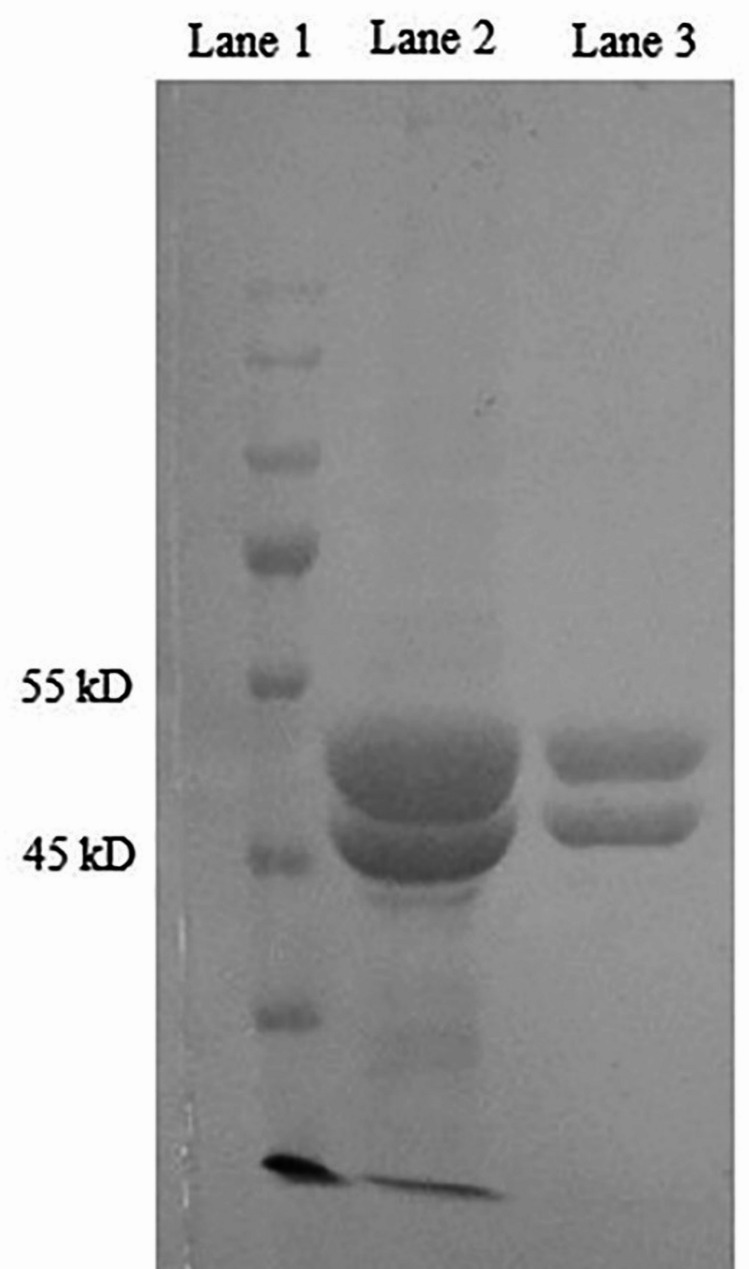
Tubulin dimers were obtained from two cycles of assembly and disassembly from the animals’ brain extracts, followed by a purification step with a phosphocellulose column (Fig. 1). MAP-free tubulin was successfully obtained, and a high level of purity was observed by sodium dodecyl sulfate-polyacrylamide gel (SDS-PAGE) stained by Coomassie Brilliant blue. As shown in Fig. 1, α-tubulin and β-tubulin are two separate narrow lines in lane 2.
Δ9-THC significantly decreased tubulin polymerization with a significant effect on nucleation and elongation
Turbidity measurements were conducted at 350 nm to characterize the effects of Δ9-THC on tubulin dynamicity (Fig. 2).
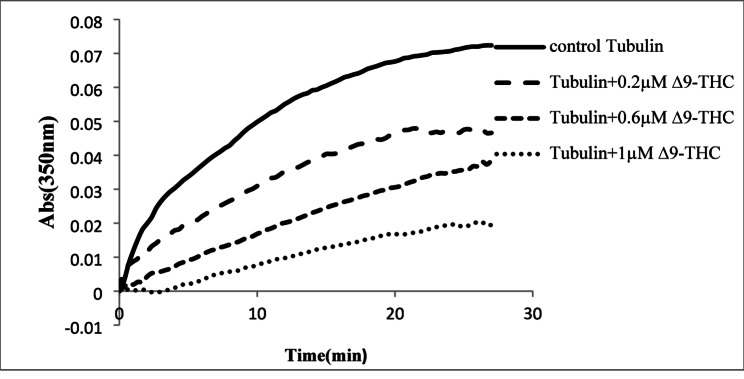
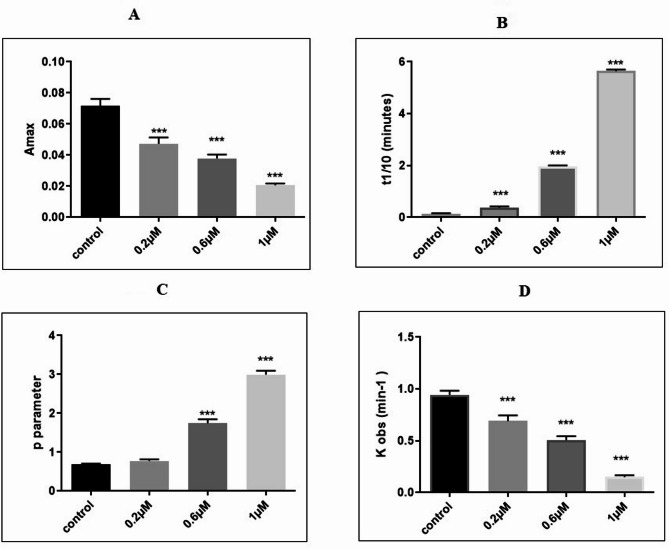
The kinetic parameters calculated from the polymerization curve are presented in Table 1. The maximum absorbance (Amax( at the steady state decreased significantly in a dose-dependent manner in the drug-treated samples as compared to the control (df = 11, F (3,8) = 119.1, p < 0.001, Fig. 3A). Δ9-THC significantly increased the tenth time of polymerization in all drug-treated tubulins (df = 11, F (3,8) = 114.56, p < 0.01 in 0.2 µM and p < 0.001 in 0.6, 1 µM Δ9-THC samples, Fig. 3B). Parameter p increased significantly in 0.6 and 1 Δ9-THC (df = 11, F (3, 8) = 61.3, p < 0.001, Fig. 3C), while kobs decreased in a dose-dependent manner in Δ9-THC samples (df = 11, F (3, 8) = 207, p < 0.001, Fig. 3D).
Table 1: The polymerization and purification of tubulin depict the influence of different concentrations of Δ9-THC (0.2 ΜM, 0.6 ΜM, and 1 µM) on four relevant parameters: maximum absorbance (Amax), half-life (t1/10), parameter P, and the observed rate constant (kobs). It is noteworthy that Amax is reduced by increasing concentrations of Δ9-THC, whereas both t1/10 and parameter P exhibit dose-dependent enhancement. In contrast, kobs fell off steeply with increasing Δ9-THC concentrations, reflecting a concentration-dependent Inhibition of the observed rate constant. This table gives a quantitative summary of the dose-response relation of Δ9-THC with the parameters measured, supplying information about its biochemical and kinetic action. *P < 0.05, **p < 0.01, ***p < 0.001; values represent the mean ± SEM
| Δ9-THC concentration | Amax | t1/10(minutes) | parameter p | kobs |
|---|---|---|---|---|
| Control | 0.071 ± 0.004 | 0.147 ± 0.016 | 0.68 ± 0.03 | 0.94 ± 0.04 |
| 0.2 µM Δ9-THC | 0.048 ± 0.004** | 0.37 ± 0.052* | 0.77 ± 0.04 | 0.69 ± 0.055** |
| 0.6 µM Δ9-THC | 0.037 ± 0.002** | 1.97 ± 0.03** | 1.74 ± 0.100*** | 0.506 ± 0.037** |
| 1 µM Δ9-THC | 0.020 ± 0.001*** | 5.65 ± 0.529** | 2.99 ± 0.11*** | 0.15 ± 0.016** |
Δ9-THC interacts with the tubulin dimer through one binding site
Our results from the fluorescence titration indicated that Δ9-THC has a quenching effect on tubulin. The fluorescence emission intensity of tubulin decreased rapidly in the presence of increasing concentrations of Δ9-THC, an interaction between tubulin and Δ9-THC as shown in Fig. 4A. The number of binding sites and the binding constant were calculated by plotting log [ (F0–F)/F] against log [Δ9-THC] (Eq. 1), where F0 and F are the fluorescence intensities before and after the addition of the ligand, Ka is the binding constant, and [Q] is the concentration of the quencher.
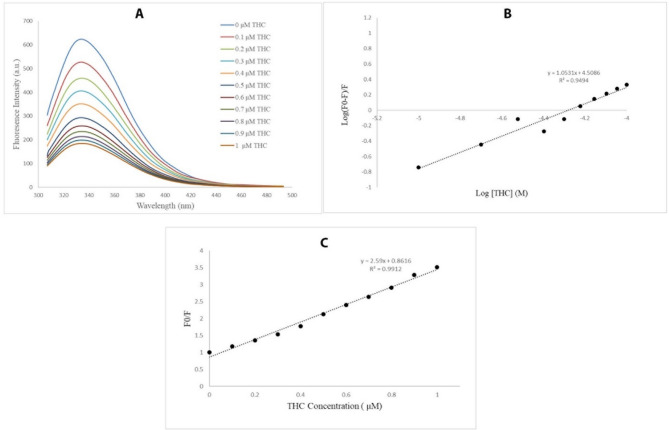
This plot demonstrated that one binding site exists for Δ9-THC on the tubulin dimer and the binding constant is equal to 3.16 × 104 L. mol− 1, indicating the tubulin’s high affinity for Δ9-THC as shown in Fig. 4B.
\[
\:Log\left(\frac{{F}_{0}-F}{F}\:\right)=Log{K}_{a}+nLog\left[Q\right]
\]
The Stern-Volmer equation (Eq. 2) was used to define the quenching constant. Kq, Ksv, and τ0 are the biomolecular quenching rate constants, the Stern–Volmer quenching constant, and the average lifetime of the biomolecule without the quencher (τ0 = 10− 8 s), respectively.
This plot indicated that the quenching constant was equal to 2.59 × 1014 L. s. mol− 1, suggesting a static interaction between Δ9-THC and tubulin (Fig. 4C). The fluorescence quenching mechanism is dynamic or static [ref. 36]. According to our data, the plot of Stern − Volmer is linear and the obtained Kq (2.59 × 1014 M− 1 S− 1) is larger than 2 × 1010 M− 1 S− 1 verifying that the main fluorescence quenching mechanism is likely static (Table 2).
Table 2: Biophysical parameters of Δ9-THC interaction with tubulin
| Ka (M− 1) | Ksv(M− 1) | Kq (M− 1 S− 1) | n |
|---|---|---|---|
| 3.16 × 104 | 2.59 × 106 | 2.59 × 1014 | 1.053 |
\[
\:\frac{{F}_{0}}{F}=1+{K}_{sv}\left[Q\right]=1+{K}_{q}{\tau}_{0}
\]
CD spectroscopy demonstrated a notably decreased α-helix in proportion to other secondary structures
The CD data showed alterations in the secondary structure of tubulin (Fig. 5; Table 3). Decreasing intensity and broadening of both the positive and the negative CD bands led to a shift in the x-axis toward higher wavelengths and exhibited a slight red shift (Fig. 5).
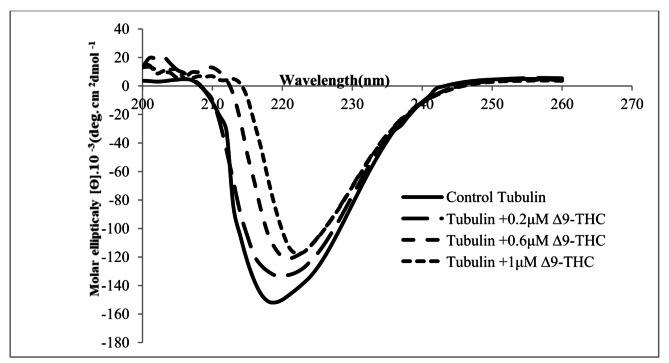
Table 3: Alterations of the tubulin secondary structures in the presence of different concentrations of Δ9-THC. The decrease in α-helix structure was significantly more in the drug-treated groups than in the control group. The increase in β-turn, β-antiparallel, β-parallel, and the random coil was significantly observed in the drug-treated groups compared with the control group. *p < 0.05, **p < 0.01, ***p < 0.001; Values represent the mean ± SEM
| Δ9-THC concentration | α-helix (%) | β-turn (%) | β-antiparallel (%) | β-parallel (%) | Random coil(%) |
|---|---|---|---|---|---|
| Control | 52.6 ± 1.447 | 12.27 ± 0.688 | 3.93 ± 0.497 | 5.2 ± 0.251 | 21.73 ± 0.933 |
| 0.2µM | 42.75 ± 1.824* | 14.77 ± 0.95 | 6.1 ± 0.264** | 6.76 ± 0.145** | 28.53 ± 0.78** |
| 0.6µM | 30.47 ± 0.913*** | 15.20 ± 1.035** | 8.13 ± 0.185*** | 8.76 ± 0.12*** | 31.97 ± 1.126** |
| 1µM | 24.02 ± 2.170*** | 19.07 ± 0.548** | 10.93 ± 0.348*** | 10.67 ± 0.176*** | 36.73 ± 1.146** |
The increase in β-sheet structure and decrease in α-helix coils were significantly observed in the drug-treated groups compared with the control sample (Table 3). α-helix secondary structure decreased significantly in Δ9-THC treated groups compared with the control (df = 11, F (3,8) = 58.9, p < 0.001). On the contrary, β-turn (df = 11, F (3,8) = 11.48, p = 0.0029), β-antiparallel (df = 11, F (3,8) = 75.12, p < 0.001), β-parallel (df = 11, F (3,8) = 174.1, p < 0.001), and random coil (df = 11, F (3,8) = 39.11, p < 0.001) increased significantly compared with the control group (Table 3).
Docking studies
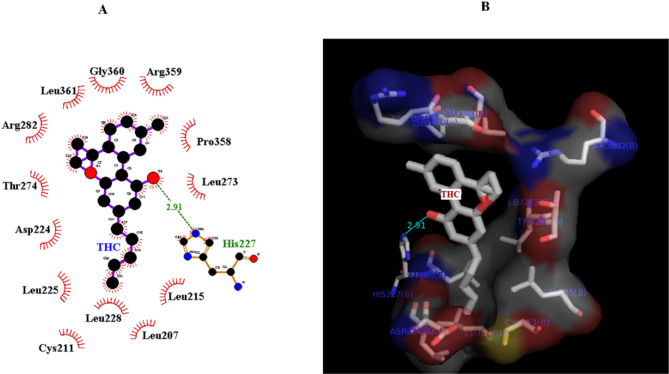
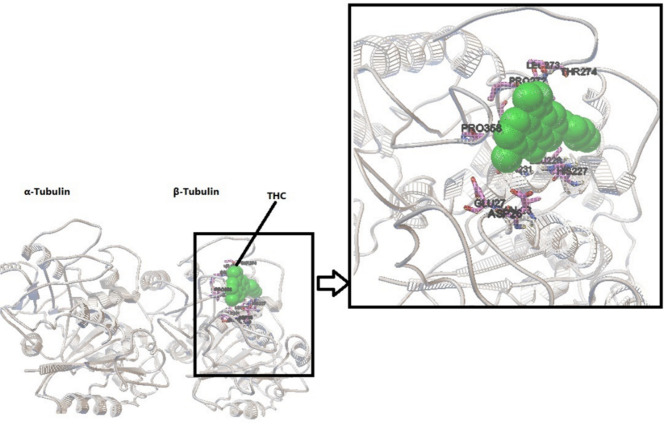
Molecular docking provides insight into the binding of Δ9-THC to tubulin. The conformer with the lowest energy in the cluster, with 20 members of 400 conformers, was selected. According to AutoDock calculations, the binding energy of this structure was − 7 kcal/mol. A hydrogen bond between the side chain of His 227 and the OH group of the cyclohexane ring of Δ9-THC is orienting the ligand molecule to occupy the binding site. The results show that van der Waals interactions, hydrogen bonds, and hydrophobic interactions play a significant role in the binding of Δ9-THC to a hydrophobic pocket in β-tubulin. Based on the modeling studies, the dominant interaction is hydrophobic. As shown in Figs. 6A and B, the hydrophobic site of β-tubulin interacts with Δ9-THC via Arg359, Pro358, leu228, cys211, leu215, leu225, leu273, Thr 274, Arg 282, leu 361, leu207, and Gly360. According to the first docking over the entire tubulin, there is no binding site on α-tubulin, and all the full binding sites are on β-tubulin (Fig. 7). Δ9-THC mainly binds close to the GTP binding site on β-tubulin.
Discussion
In this study, we proposed that Δ9-THC induces significant changes in the microtubule dynamicity resulting from the structural changes in the tubulin dimes.
Dynamic microtubules are involved in synaptic plasticity in the brain [ref. 7, ref. 37]. Therefore, microtubule dysfunction may affect synaptic plasticity and spatial memory. Uv-visible spectroscopy represented that Δ9-THC exerts its effect through changes in microtubule dynamicity as well as, and microtubule dysfunction may affect synaptic plasticity as one, among others, possible mechanisms.
Studying the kinetics of the polymerization assay provided interesting data, increasing the tenth times (t1/10) demonstrates a more extended lag phase in Δ9-THC incubated samples compared to the control group. This suggests that the necessary time for tubulin dimer assembly in nucleation increased by Δ9-THC. In addition, the p parameter increased by increasing the Δ9-THC concentration, implying that Δ9-THC disturbs the lag phase and nucleation, while the kobs value decreased strongly in a dose-dependent manner, showing that increasing Δ9-THC concentration has a critical role in reducing the elongation process of polymerization. Therefore, Δ9-THC suppresses elongation. It seems that Δ9-THC decelerates the nucleation and elongation process and reduces the final amount of the protein polymer.
The fluorescence intensity of the tubulin dimers decreased in the presence of Δ9-THC. As the Δ9-THC did not have any intrinsic fluorescence intensity, the fluorescence quenching seems to be the interactions between Δ9-THC and tubulin dimers, which is confirmed by previous studies [ref. 26]. The fluorescent studies and Stern-Volmer plot in this experiment show that there is a single stable site of Δ9-THC- tubulin interaction. The reduction of the fluorescence intensity originated from one of the static quenching mechanisms. Furthermore, the number of tubulin-binding sites (n) was calculated to be one. Three significant classes of tubulin-binding drugs have been identified: I; Colchicine binding ligands; ii, the Vinca domain drugs; and iii; Texans and Epothilone family. Vinca domain inhibitors, next to the nucleotide-binding region, inhibit polymerization by preventing nucleic acid exchange [ref. 38, ref. 39]. Our computer-based study demonstrated one binding site for Δ9-THC on tubulin, which is around the Vinca region and makes a hydrogen bond with His 227 and electrostatic and hydrophobic interactions with hydrophobic amino acids on β-tubulin and might inhibit the GDP-GTP exchange and prevent microtubule polymerization.
Δ9-THC is a hydrophobic structure that contains three cyclic structures: a phenol ring, a pyran ring, and a cyclohexane ring, causing rigidity in the tubulin’s structure and the alkyl moiety [ref. 40]. Adding Δ9-THC to the tubulin solution might induce the exposure of hydrophobic amino acids normally buried inside α- helix secondary structures.
Our CD spectroscopy results elucidated that the proportion of α-helix structures was reduced in Δ9-THC treated compared to the control. Additionally, the proportion of β-sheets and other structures was increased in Δ9-THC treated in comparison with the control. This result is notably similar to previous in vitro studies [ref. 26]. The addition of Δ9-THC to the protein solution induces the secondary structures of the protein to transit to β- sheet and random coil structures, aiming to expose more hydrophobic amino acids. Thus, we predict that Δ9-THC can bind to tubulin dimers through hydrophobic interactions.
Conclusions
In conclusion, our study reveals that Δ9-THC significantly disrupts microtubule dynamics by altering the structural properties of tubulin dimers, which may impair brain function. Δ9-THC extends the lag phase of tubulin assembly, reduces polymerization, and interacts with tubulin near the Vinca domain. Additionally, Δ9-THC induces a shift from α-helix to β-sheet and random coil structures, exposing the hydrophobic amino acids. These findings highlight the molecular mechanisms through which Δ9-THC affects neuronal functions. While this study provides insights into the direct interaction of Δ9-THC with tubulin, in vitro binding assays may not fully replicate cellular conditions. Our future studies will focus on cell culture studies to validate these findings.
Supplementary Materials
References
- EW Dent. Of microtubules and memory: implications for microtubule dynamics in dendrites and spines. Mol Biol Cell, 2017. [PubMed]
- A Akhmanova, MO Steinmetz. Microtubule minus-end regulation at a glance. J Cell Sci, 2019. [PubMed]
- A Akhmanova, MO Steinmetz. Control of microtubule organization and dynamics: two ends in the limelight. Nat Rev Mol Cell Biol, 2015. [PubMed]
- PJ Foster, S Fürthauer, MJ Shelley, DJ Needleman. From cytoskeletal assemblies to living materials. Curr Opin Cell Biol, 2019. [PubMed]
- P Verstraelen, JR Detrez, M Verschuuren, J Kuijlaars, R Nuydens, J-P Timmermans, WH De Vos. Dysregulation of microtubule stability impairs morphofunctional connectivity in primary neuronal networks. Front Cell Neurosci, 2017. [PubMed]
- S Maday, AE Twelvetrees, AJ Moughamian, EL Holzbaur. Axonal transport: cargo-specific mechanisms of motility and regulation. Neuron, 2014. [PubMed]
- J Jaworski, LC Kapitein, SM Gouveia, BR Dortland, PS Wulf, I Grigoriev, P Camera, SA Spangler, P Di Stefano, J Demmers. Dynamic microtubules regulate dendritic spine morphology and synaptic plasticity. Neuron, 2009. [PubMed]
- 8.Collingridge GL, Isaac JT, Wang YTJN. Receptor trafficking and synaptic plasticity. 2004, 5(12):952.
- V Anggono, RL Huganir. Regulation of AMPA receptor trafficking and synaptic plasticity. Curr Opin Neurobiol, 2012. [PubMed]
- TE Chater, Y Goda. The role of AMPA receptors in postsynaptic mechanisms of synaptic plasticity. Front Cell Neurosci, 2014. [PubMed]
- SA Yousefzadeh, M Jarah, GH Riazi. Tryptophan improves memory independent of its role as a serotonin precursor: potential involvement of microtubule proteins. J Mol Neurosci, 2020. [PubMed]
- D Atarod, G Eskandari-Sedighi, F Pazhoohi, SM Karimian, M Khajeloo, GH Riazi. Microtubule dynamicity is more important than stability in memory formation: an in vivo study. J Mol Neurosci, 2015. [PubMed]
- M Mohammadkhani, D Gholami, G Riazi. The effects of chronic morphine administration on Spatial memory and microtubule dynamicity in male mice’s brain. IBRO Neurosci Rep, 2024. [PubMed]
- SK Tahir, JE Trogadis, JK Stevens, AM Zimmerman. Cytoskeletal organization following cannabinoid treatment in undifferentiated and differentiated PC12 cells. Biochem Cell Biol, 1992. [PubMed]
- J Tomas-Roig, S Ramasamy, D Zbarsky, U Havemann-Reinecke, S Hoyer-Fender. Psychosocial stress and cannabinoid drugs affect acetylation of α-tubulin (K40) and gene expression in the prefrontal cortex of adult mice. PLoS ONE, 2022. [PubMed]
- M Ranganathan, DC D’Souza. The acute effects of cannabinoids on memory in humans: a review. Psychopharmacology, 2006. [PubMed]
- JM Sullivan. Cellular and molecular mechanisms underlying learning and memory impairments produced by cannabinoids. Learn Mem, 2000. [PubMed]
- LE Wise, AJ Thorpe, AH Lichtman. Hippocampal CB 1 receptors mediate the memory impairing effects of Δ 9-tetrahydrocannabinol. Neuropsychopharmacology, 2009. [PubMed]
- JR Clarke, JI Rossato, S Monteiro, LR Bevilaqua, I Izquierdo, M Cammarota. Posttraining activation of CB1 cannabinoid receptors in the CA1 region of the dorsal hippocampus impairs object recognition long-term memory. Neurobiol Learn Mem, 2008. [PubMed]
- E Puighermanal, G Marsicano, A Busquets-Garcia, B Lutz, R Maldonado, A Ozaita. Cannabinoid modulation of hippocampal long-term memory is mediated by mTOR signaling. Nat Neurosci, 2009. [PubMed]
- P Derkinderen, E Valjent, M Toutant, J-C Corvol, H Enslen, C Ledent, J Trzaskos, J Caboche, J-A Girault. Regulation of extracellular signal-regulated kinase by cannabinoids in hippocampus. J Neurosci, 2003. [PubMed]
- A Ozaita, E Puighermanal, R Maldonado. Regulation of PI3K/Akt/GSK-3 pathway by cannabinoids in the brain. J Neurochem, 2007. [PubMed]
- M Maroso, GG Szabo, HK Kim, A Alexander, AD Bui, S-H Lee, B Lutz, I Soltesz. Cannabinoid control of learning and memory through HCN channels. Neuron, 2016. [PubMed]
- D Jimenez-Blasco, A Busquets-Garcia, E Hebert-Chatelain, R Serrat, C Vicente-Gutierrez, C Ioannidou, P Gómez-Sotres, I Lopez-Fabuel, M Resch-Beusher, E Resel. Glucose metabolism links astroglial mitochondria to cannabinoid effects. Nature, 2020. [PubMed]
- C Sánchez, I Galve-Roperh, C Canova, P Brachet, M Guzmán. Δ9-Tetrahydrocannabinol induces apoptosis in C6 glioma cells. FEBS Lett, 1998. [PubMed]
- D Gholami, AR Noori, M Mohammadkhani, Z Emruzi, GH Riazi. The long-term effects of Δ9-tetrahydrocannabinol on microtubule dynamicity in rats. Arch Biochem Biophys, 2020. [PubMed]
- KH Choi, K Whisler, DL Graham, DW Self. Antisense-induced reduction in nucleus accumbens Cyclic AMP response element binding protein attenuates cocaine reinforcement. Neuroscience, 2006. [PubMed]
- 28.Miller HP, Wilson L. Preparation of microtubule protein and purified tubulin from bovine brain by cycles of assembly and disassembly and phosphocellulose chromatography. Methods in cell biology. Volume 95. edn.: Elsevier; 2010. pp. 2–15.
- D Gholami, G Riazi, R Fathi, M Sharafi, A Shahverdi. Comparison of polymerization and structural behavior of microtubules in rat brain and sperm affected by the extremely low-frequency electromagnetic field. BMC Mol Cell Biology, 2019
- ML Shelanski, F Gaskin, CR Cantor. Microtubule assembly in the absence of added nucleotides. Proc Natl Acad Sci U S A, 1973. [PubMed]
- KA Johnson, GG Borisy. Kinetic analysis of microtubule self-assembly in vitro. J Mol Biol, 1977. [PubMed]
- C Bonfils, N Bec, B Lacroix, M-C Harricane, C Larroque. Kinetic analysis of tubulin assembly in the presence of the microtubule-associated protein TOGp. J Biol Chem, 2007. [PubMed]
- G-G Chang, H-J Lee. Monitoring protein conformational changes by quenching of intrinsic fluorescence. J Biochem Biophys Methods, 1984. [PubMed]
- JM Word, SC Lovell, JS Richardson, DC Richardson. Asparagine and glutamine: using hydrogen atom contacts in the choice of side-chain amide orientation1. J Mol Biol, 1999. [PubMed]
- AC Wallace, RA Laskowski, JM Thornton. LIGPLOT: a program to generate schematic diagrams of protein-ligand interactions. Protein Eng, 1995. [PubMed]
- J Ding, L Yuan, L Gao, J Chen. Fluorescence quenching of a Rhodamine derivative: selectively sensing Cu2 + in acidic aqueous media. J Lumin, 2012
- EW Dent. Dynamic microtubules at the synapse. Curr Opin Neurobiol, 2020. [PubMed]
- A Cormier, M Marchand, RB Ravelli, M Knossow, Gigant, BJEr. Structural insight into the Inhibition of tubulin by vinca domain peptide ligands. EMBO Rep, 2008. [PubMed]
- A Mitra, D Sept. Localization of the antimitotic peptide and depsipeptide binding site on beta-tubulin. Biochemistry, 2004. [PubMed]
- RS Borges, J Batista, RB Viana, AC Baetas, E Orestes, MA Andrade, KM Honório, AB Da Silva. Understanding the molecular aspects of tetrahydrocannabinol and Cannabidiol as antioxidants. Molecules, 2013. [PubMed]
