Cannabinoid therapeutics in orofacial pain management: a systematic review
Abstract
The objective of this paper was to investigate the published evidence regarding effects of cannabinoids (natural and synthetic) on post‐operative and/or out‐of‐office pain management in patients suffering from orofacial pain that presents in the dental setting. Three online databases (Ovid (MEDLINE), PubMed (MEDLINE), Scopus) were searched (July 2021). Additional studies were sought through grey literature searching (Cochrane Library Trials and ClinicalTrials.gov) and hand‐searching the reference lists of included articles. All studies that analysed cannabinoid products and pain management of conditions that present in the general or specialist dental setting in the English language were included. Of the five articles included, one reported a significant effect on temporomandibular disorder pain relief using a topical cannabidiol formulation compared to a placebo. Four articles reported no significant effects of cannabinoids for pain management across various orofacial pain conditions. Although one study reported a positive effect, insufficient evidence exists to support a tangible clinical benefit of cannabinoids in managing orofacial pain, further research is recommended to investigate the benefits of cannabinoids’ use. © 2022 Australian Dental Association.
Article type: Review Article
Keywords: Orofacial pain, analgesia, cannabis, cannabinoids, cannabidiol, marijuana
Affiliations: College of Medicine & Dentistry James Cook University Smithfield Queensland Australia; Australian Institute of Tropical Health and Medicine James Cook University Queensland Australia
License: © 2022 The Authors. Australian Dental Journal published by John Wiley & Sons Australia, Ltd on behalf of Australian Dental Association. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1111/adj.12934 | PubMed: 36082517 | PMC: PMC10087667
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.4 MB)
INTRODUCTION
Cannabis plants have been used for many millennia, as evidenced by their presence in excavated tombs dating back to the first millennium BCE.ref. adj12934-bib-0001, ref. adj12934-bib-0002 In recent years, the focus has shifted away from the commonly known euphoric effects of cannabis towards their therapeutic potential.ref. adj12934-bib-0003 Specifically, cannabinoids have shown promise due to their antinociceptive, antiemetic and anticonvulsant properties.ref. adj12934-bib-0004 As the medicinal use of cannabinoids continue to rise throughout the world, their application for potential dental therapeutic benefit is being explored.ref. adj12934-bib-0005
The Cannabis genus of flowering plants mainly comprises the sativa and indica species.ref. adj12934-bib-0005 The two notable pharmacological constituents, that is cannabinoids isolated from the plant are delta‐9‐tetrahydrocannabinol (THC) and cannabidiol (CBD).ref. adj12934-bib-0006 THC is principally responsible for the psychoactive effects and works via activation of the endocannabinoid system. Interacting with cannabinoid type 1 (CB1) and cannabinoid type 2 (CB2) receptors, THC subsequently modulates neurotransmitter release impacting nociception, immune function, appetite and mood.ref. adj12934-bib-0005, ref. adj12934-bib-0006, ref. adj12934-bib-0007, ref. adj12934-bib-0008 CBD is the non‐psychoactive component and with very low affinity for CB1 and CB2 receptors.ref. adj12934-bib-0006 It is thought that CBD inhibits anandamide degradation. Anandamide acts on the transient receptor potential vanilloid type 1 (TRPV1), consequently showing potent anticonvulsant, antiemetic and anti‐inflammatory properties.ref. adj12934-bib-0008, ref. adj12934-bib-0009 These three receptors that cannabinoids activate are found across the central and peripheral nervous system in many organs and tissues, including the dental pulp and the periodontium.ref. adj12934-bib-0005, ref. adj12934-bib-0010
Three groups of cannabinoids have been reported: ‘phytocannabinoids’ which are derived from the cannabis plant; ‘endocannabinoids’ which are naturally occurring cannabinoids synthesized in the human body that interact with cannabinoid receptors and synthetic cannabinoids.ref. adj12934-bib-0011 Synthetic cannabinoids agonize CB1 and CB2 receptors and typically have a greater affinity for the CB1 receptor than endocannabinoids.ref. adj12934-bib-0011
While cannabinoids have been legalized for medical use within the international community for some time, it was not until 29 February 2016 when changes to the Narcotic Drugs Amendment Act 2016, that the cultivation, production and distribution of medicinal marijuana were legalized in Australia. Prior to this change, Australia classed cannabinoids as a Schedule 9 Prohibited Substance.ref. adj12934-bib-0005, ref. adj12934-bib-0011, ref. adj12934-bib-0012 Currently, patients can access only two TGA‐approved medicinal cannabis products, these are CBD oil (‘Epidyolex’) for epilepsy and Nibiximols (‘Sativex’) spray on prescription from general practitioners or specialist medical practitioners according to state‐based legislation.ref. adj12934-bib-0013 A wide variety of unregistered cannabinoid products are also available which may also be accessed through the Therapeutic Goods Administration (TGA) under the Special Access Scheme (Category B) and the Authorised Prescriber Scheme.ref. adj12934-bib-0013 Not being included in the ARTG means the TGA has not assessed these products for safety, quality or effectiveness. How the dental practitioners may be able to access medicinal cannabinoids is yet to be defined at both state level and federal level.ref. adj12934-bib-0014 This is vital as the dentists would be the first point of contact for patients with oro‐facial pain.
In February 2021, the TGA changed the scheduling of low‐dose CBD oil (contain no more than 1% THC and used at a maximum dosage of 150 milligrams or less per day) from a Schedule 4 (S4) drug to a ‘Pharmacist Only Schedule 3 (S3) drug’.ref. adj12934-bib-0015 Despite this, there are currently no CBD oil products on the Australian market that fulfil the S3 requirements.ref. adj12934-bib-0016 Types of cannabinoid products available in Australia which are currently regulated as S4 (prescription only) and S8 drugs (controlled drugs) including floss/bud, oils, oral‐mucosal spray, liquid capsules and CBD patches, creams and gels.ref. adj12934-bib-0012 These products can be administered via several methods including oral‐mucosal spray, sublingual oil capsules or tablets, smoking, vaporization and trans‐dermal application.ref. adj12934-bib-0005, ref. adj12934-bib-0012
Nabiximol (Sativex®) and cannabidiol (Epidyolex®) are the only cannabinoids currently registered on the ARTG.ref. adj12934-bib-0017 Sativex® is an oral‐mucosal spray with a 1:1 THC and CBD ratio from cannabis extract used to treat spasticity in multiple sclerosis patients.ref. adj12934-bib-0013 Epidyolex® is a plant‐derived oil‐based formulation of CBD used to treat seizures associated with refractory childhood epilepsy, such as Dravet syndrome and Lennox‐Gastaut syndrome.ref. adj12934-bib-0013 These medicines contain differing THC and CBD ratios.ref. adj12934-bib-0013 The synthetic cannabinoids dronabinol (Marinol®, Syndros®) and nabilone (Cesamet®) are licensed for medicinal use in the USA but not currently available in Australia, although have been used in Australia in the past under the SAS scheme.ref. adj12934-bib-0005, ref. adj12934-bib-0017, ref. adj12934-bib-0018
Globally, there is growing acceptance, popularity and use of cannabinoid products for a range of health conditions, including orofacial pain.ref. adj12934-bib-0005 Treating patients with pain in the orofacial region is one of the prominent roles that dentists play.ref. adj12934-bib-0005 Orofacial pain in the dental setting can include acute pain (e.g. pulpitis, apical periodontitis, post‐operative surgical pain) or chronic pain (e.g. temporomandibular disorders (TMD), burning mouth syndrome (BMS), trigeminal neuralgia).ref. adj12934-bib-0005 Recent studies have been carried out using Cannabis sativa oil to manage patients diagnosed with primary BMS, to improve the quality of life (QOL) in patients with head and neck cancer (HNC) and via topical application of CBD to treat TMD.ref. adj12934-bib-0019, ref. adj12934-bib-0020, ref. adj12934-bib-0021, ref. adj12934-bib-0022 Hence, it would be very beneficial to review the current literature on the therapeutic use and effects of cannabinoids in the orofacial region.ref. adj12934-bib-0005 Consequently, the main objective of this systematic review was to explore the published evidence regarding effectiveness of cannabinoids in orofacial pain management in a dental setting.
METHODS
The systematic review was carried out in accordance with the Preferred Reporting Items for Systematic reviews and Meta‐Analyses (PRISMA) statement.ref. adj12934-bib-0023 The clinical question ‘Are cannabinoid therapeutics effective in (acute and chronic) orofacial pain management, when compared to other pharmacological or placebo treatments’? informed the PICO framework of the systematic review, where P (population) is humans; I (intervention) is cannabinoids (natural and synthetic); C (comparison) is other pharmacological treatments or placebos and O (outcome) is improved pain management.
Information sources and search strategy
An electronic database search using Ovid (MEDLINE), PubMed (MEDLINE) and Scopus was performed on 11 July 2021 to systematically retrieve articles. A grey literature search of ClinicalTrials.gov register and Cochrane Library Trials database was performed on the same date to search for eligible studies. No restrictions were implemented for the date or location of the publications. Databases were searched using different combinations of MeSH terms and keywords (Appendix 1). The ClinicalTrials.gov register was searched multiple times with one keyword change in each search due to user interface limitations. After record screening, citations within the included studies were also manually searched for eligible studies.
Selection process
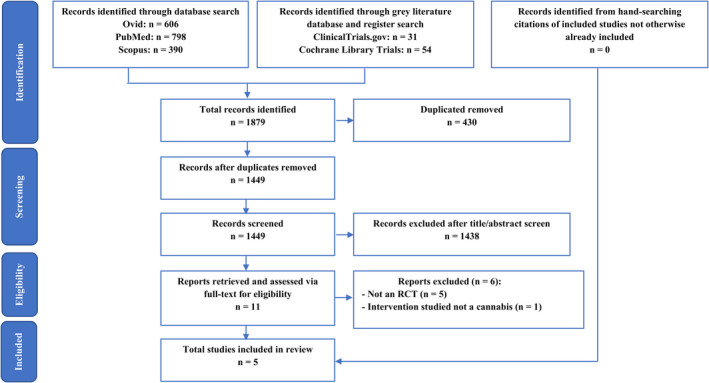
The selection process of this review was carried out as per the PRISMA flow diagram illustrated in Figure 1.ref. adj12934-bib-0023 EndNoteTM (Version 20, Clarivate Analytics) was used during the identification and screening stage to store and sort articles. Titles and abstracts of identified records were screened independently by four assessors. Conflicts in screening were discussed and resolved with mediators. Full‐text articles were then retrieved, and each report was examined by two independent assessors for eligibility for inclusion in the review. Any disagreements were arbitrated by a third independent assessor and senior researcher through discussion.
Eligibility criteria
Studies were eligible for inclusion if they satisfied the following inclusion criteria:
- In vivo randomized controlled trials (RCT) involving adult humans (>18 years); published or completed with results.
- Subjects with orofacial pain (acute or chronic) as diagnosed by a dentist or dental therapist in the general or specialist dental setting.
- Investigated cannabinoids and their effect on pain management.
Studies were excluded if they were not written in the English language, involved in vitro or animal studies, included subjects with orofacial pain along with other conditions in which cannabinoids may influence outcomes (e.g. epilepsy, multiple sclerosis), or involved use of cannabinoid‐receptor agonists synthesized from sources other than cannabis plants. Reviews, opinion papers, case series/reports, observational studies, conference abstracts and pilot studies were also excluded.
Data collation process
Data were extracted and compiled into a spreadsheet using a customized data form. A calibration process was used for six reviewers. Data were extracted independently from each included article by two different reviewers. Results and rationale were then reviewed by all six extractors and any disagreements were resolved by discussion and consensus was reached.
The following data items were extracted: author(s); year of publication; location of study; funding source, if identifiable; study design; sampling characteristics; measured outcome and methodology of measuring scale/device used; initial recording of measurement; follow‐up periods; adverse events and final outcomes.
Quality and risk of bias of included studies
Articles were assessed for Risk of Bias using Version 2 of the Cochrane risk‐of‐bias tool for randomized trials (RoB 2).ref. adj12934-bib-0024 RoB 2 has a fixed set of bias domains, each comprising a series of signalling questions aimed to elicit information about features of trial design, conduct and reporting. Based on answers to signalling questions, a proposed judgement of ‘low’/ ‘high’ risk of bias or expressing ‘some concerns’ was generated for each domain by an algorithm.ref. adj12934-bib-0024 Note that the first domain is study‐level, while all other domains are outcome‐level. Additional considerations were required when assessing risk of bias in randomized crossover trials, compared to individualized‐randomized parallel‐group trials.ref. adj12934-bib-0024 Each article was assessed independently by two assessors and findings were collated into a spreadsheet detailing each assessor’s name, their judgement and support for judgement. The evidence level of each included study was also confirmed as per the Oxford Centre for Evidence‐Based Medicine levels of evidence.ref. adj12934-bib-0025 Results and rationale were then reviewed by all five remaining assessors and any disagreements were resolved by discussion and consensus reached. For studies assessed with a high risk of bias, caution was used when interpreting results in the evidence synthesis.
RESULTS
The infographic of study selection (Figure 1) summarizes the discriminative and stepwise elimination of articles from 1879 total records down to five. The initial search of the literature was comprehensive in terminology, as well as database and grey literature navigation, yielding 1794 and 85 records respectively. After pooling of records, 430 duplicates were removed, leaving 1449 records to be screened. These records’ titles and abstracts were assessed against the inclusion and exclusion criteria (specified in methodology), which excluded 1438 studies from further appraisal as they did not meet the inclusion criteria. Full‐text articles of the remaining eleven records were retrieved and assessed for eligibility based on the inclusion criteria, from which five articles were deemed suitable for inclusion in this review. Specifically, five other reports were excluded as the study design was not an RCT, and one was excluded as the intervention was not a cannabinoid (Table 1). The study characteristics of included studies are presented in Tables 2 and 3.
Table 1: Summary of papers excluded and reasons for exclusion, after assessing full‐text
| Author | Year | Country | Objective of the study | Reason for exclusion |
|---|---|---|---|---|
| Elliot et al ref. adj12934-bib-0019 | 2016 | USA | To better understand why patients with a history of head and neck cancer treated with radiotherapy are using medical marijuana. | Study design not an RCT. |
| Gambino et al ref. adj12934-bib-0020 | 2021 | Italy | To evaluate the use of Cannabis sativa oil in the management of patients diagnosed with primary burning mouth syndrome. | Study design not an RCT. |
| Zhang et al ref. adj12934-bib-0021 | 2018 | Canada | To examine the differences in ‘Quality of Life’ and psychosocial outcomes between marijuana users and nonusers with newly diagnosed head and neck cancer. | Study design not an RCT. |
| Ware et al ref. adj12934-bib-0035 | 2002 | Canada | To determine medicinal use including dose size and frequency among patients with chronic non‐cancer pain and to describe the main symptoms for which relief was sought. | Study design not an RCT. |
| Phan et al ref. adj12934-bib-0036 | 2010 | Germany | To investigate tolerance and effectiveness of cream containing cannabinoid receptor agonist, N‐palmitoylethanolamine, on burning pain from postherpetic neuralgia. | Study design not an RCT. |
| Marini et al ref. adj12934-bib-0037 | 2012 | Italy | To compare the effect of palmitoylethanolamide versus ibuprofen (NSAID) for pain relief in temporomandibular joint osteoarthritis or arthralgia. | Did not investigate a cannabis constituent or synthetic derivative of cannabis; palmitoylethanolamide is an endocannabinoid‐like lipid mediator. |
Table 2: General characteristics of the included studies
| Author | Year | Location | Objective of the study | Funding source | Study design | Measurement for pain | Initial recording | Further follow‐up post intervention | Evidence levelref. adj12934-bib-0024 |
|---|---|---|---|---|---|---|---|---|---|
| Côté et al ref. adj12934-bib-0026 | 2016 | Canada | To compare the effects of nabilone versus placebo on the quality of life and side effects during radiotherapy for head and neck carcinomas | Canadian Institutes of Health Research; Fond de recherche en santé du Québec; ICN Valeant Pharmaceuticals | Randomized, double‐blind, placebo‐controlled trial | 10 cm Visual Analog Scale (VAS) | Before intervention | Every 7 days during intervention and 28 days after intervention | 2B |
| Kalliomäki et al ref. adj12934-bib-0027 | 2013 | USA | To evaluate the analgesic efficacy of AZD1940 in patients undergoing third molar surgical removal | AstraZeneca R&D | Randomized, double‐blind, double‐dummy, placebo‐controlled study | 10 cm Visual Analog Scale | Immediately after intervention | Every 20 minutes for the first 4 hours, every 60 minutes thereafter for 8 hours | 1B |
| Nitecka‐Buchta et al ref. adj12934-bib-0022 | 2019 | Poland | To evaluate the efficiency of the myorelaxant effect of CBD after the transdermal application in patients with myofascial pain | MedycynaCBD and Maciej Pawlowski for material support | Randomized, double‐blind, double‐arm, parallel‐group trial | 10 cm Visual Analog Scale | Before intervention | 14 days after intervention | 1B |
| Ostenfeld et al ref. adj12934-bib-0028 | 2011 | UK, Italy, Germany | To evaluate the postoperative analgesic efficacy of GW824166 in patients undergoing third molar tooth extraction | No funding stated | Randomized, double‐blind, placebo‐controlled study | 10 cm Visual Analog Scale, 4‐Point Categorical Verbal Rating Scale (VRS) | Before intervention and 1 hour post intervention | Every 15 minutes from 2 to 4 hours, then hourly at hour 5, 6, 7, 8, 9, 10 | 1B |
| Raft et al ref. adj12934-bib-0029 | 1977 | USA | To evaluate the effects of intravenous tetrahydrocannabinol in patients undergoing elective removal of four impacted third molars | National Institute of Dental Research; Division of Research Facilities and Resources | Randomized, double‐blind, placebo‐controlled crossover trial | Pain thresholds using:1. A strain‐gauge algometer that measured periosteal pain in gram pressure (g)2. TECA B2 EMG nerve stimulator that applied a square wave electrocutaneous stimulus to skin | Before intervention | At midpoint and 30 minutes post intervention, then at 24 hours and one month | 1B |
Table 3: Data extracted from the included articles and their major findings
| Author | Sample type | Sample size and grouping | Cannabinoid type, route and frequency of administration | Measured outcome | Reported findings |
|---|---|---|---|---|---|
| Côté et al ref. adj12934-bib-0026 | Patients aged 18‐80 years with squamous cell carcinoma of the head and/or neck being treated by radiotherapy or radiochemotherapy | Total: 56‐ Nabilone 0.5 mg: 28‐ Placebo: 28After drop‐outs‐ Nabilone 0.5 mg: 19‐ Placebo: 13 | Nabilone (synthetic cannabinoid) orally, 1 pill daily for first week, 2 pills daily for second week, maximum 4 pills daily from third week until end of radiotherapy | Difference in pain scores using VAS between groups | No significant difference between the 2 groups |
| Kalliomäki et al adj12934-bib-0027 | Patients aged 18‐45 Years with a Body Mass Index (BMI) between 18‐33 kg/mref. adj12934-bib-0002 and body weight between 50‐120 kg and scheduled for surgical removal of one impacted third molar | Total: 151‐ AZD1940 800 μg: 61‐ Placebo: 59‐ Naproxen 500 mg: 31 | AZD1940 (synthetic CB1 and CB2 receptor agonist) single oral dose | Difference in pain scores using VAS between groups | No significant difference between AZD1940 and placebo |
| Nitecka – Buchta et al ref. adj12934-bib-0022 | Patients aged 18‐60 years in good general health with temporomandibular disorder and presence of all teeth | Total: 60‐ Cannabidiol 20%: 30‐ Placebo: 30 | CBD transdermal formulation, topically twice daily for 14 days | Pain intensity changes using VAS between groups | Significant effect of CBD on average pain level and pain intensity compared to placebo |
| Ostenfeld et al ref. adj12934-bib-0028 | Patients aged 18‐50 in general good health and scheduled for surgical extraction of up to 4 third molar teeth | Total: 123‐ GW842166 100 mg pre‐operatively + placebo post‐operatively: 34‐ GW842166 800 mg pre‐operatively + placebo post‐operatively: 27‐ Ibuprofen 800 mg pre‐operatively + 400 mg post‐operatively: 31‐ Placebo pre‐ and post‐operatively: 31 | GW842166 (selective CB2 receptor agonist) single oral dose | Pain intensity changes in VAS and VRS between groups | No significant effect of GW842166 compared to placebo |
| Raft et al ref. adj12934-bib-0029 | Male patients aged 18‐28 years requiring elective removal of four impacted third molars | Total: 10‐ THC 0.22 mg/kg: 10‐ THC 0.44 mg/kg: 10‐ Diazepam 0.157 mg/kg: 10‐ Ringer’s lactate (placebo): 10 | THC single intravenous dose | Changes in pain detection and tolerance thresholds using strain‐gauge algometer and TECA B2 EMG nerve stimulator between interventions | No significant analgesic effect from THC compared to placebo; six subjects preferred placebo to low‐dose THC |
Risk of bias within included studies
The risk of bias within included studies was determined using the domain‐specific signalling questions in Appendix 2. Of the individually randomized, parallel‐group trials, Nitecka et al were judged to be of low risk of bias for all domains (Table 4).ref. adj12934-bib-0022 The overall risk‐of‐bias judgment for Côté et al’s study was high‐risk (Table 4).ref. adj12934-bib-0026 Studies by Kalliomäki et al and Ostenfeld et al raised some concerns, as there was risk of bias arising from the randomization process (Table 4).ref. adj12934-bib-0027, ref. adj12934-bib-0028 The study by Raft et al was a crossover trial and appraised using the appropriate RoB 2 tool and noted as raising some concerns of bias (Table 5).ref. adj12934-bib-0029
Table 4: Risk of bias assessment of individual‐randomized, parallel‐group trials according to the domains of the RoB 2 Toolref. adj12934-bib-0024
| Domains | ||||||
|---|---|---|---|---|---|---|
| Study | 1 | 2 | 3 | 4 | 5 | Overall |
| Côté et al ref. adj12934-bib-0026 | ||||||
| Kalliomäki et al ref. adj12934-bib-0027 | ||||||
| Nitecka‐Buchta et al ref. adj12934-bib-0022 | ||||||
| Ostenfeld et al ref. adj12934-bib-0028 | ||||||
Key: Low risk of bias, Some concerns, High risk of bias.
Table 5: Risk of bias assessment of Crossover trial according to the domains of the RoB 2 Toolref. adj12934-bib-0024
| Domains | |||||||
|---|---|---|---|---|---|---|---|
| Study | 1 | S | 2 | 3 | 4 | 5 | Overall |
| Raft et al ref. adj12934-bib-0029 | |||||||
Key: Low risk of bias, Some concerns, High risk of bias.
Effects of cannabinoids on pain relief
Post‐operative orofacial pain is often a challenging form of pain to manage as it can significantly affect quality of life and delay recovery of patients. Two studies included in this review compared synthetic cannabinoids to placebos and NSAIDs (naproxen and ibuprofen respectively) with their effect on pain management following third molar extractions.ref. adj12934-bib-0027, ref. adj12934-bib-0028 Côté et al also investigated a synthetic cannabinoid, nabilone, compared to placebo for HNC pain.ref. adj12934-bib-0026 These three studies investigated interventions that were administered orally, and none of the studies reported evidence that the synthetic cannabinoids were more effective than placebo for pain relief.ref. adj12934-bib-0026, ref. adj12934-bib-0027, ref. adj12934-bib-0028 Only study by Nitecka‐Buchta et al reported a statistically significant effect of CBD on pain relief compared to placebo.ref. adj12934-bib-0022 On the contrary, four other included studies found that the cannabinoid investigated did not produce a statistically significant pain relief when compared with placebo.ref. adj12934-bib-0026, ref. adj12934-bib-0027, ref. adj12934-bib-0028, ref. adj12934-bib-0029 In fact, Kalliomäki et al and Ostenfeld et al both found NSAIDs were more effective (statistically) than placebo across all endpoints.ref. adj12934-bib-0027, ref. adj12934-bib-0028 The remaining two studies investigated phytocannabinoids, THC and CBD.ref. adj12934-bib-0010 Raft et al investigated THC, the main psychoactive compound in cannabis, without CBD to determine if THC pain responses were due to a disruption of sensory coding or direct neural action on nociceptors.ref. adj12934-bib-0029 Ten subjects required removal of four impacted third molars, and each subject participated at four separate trials wherein a different premedicant was administered for each wisdom tooth over a standard 5‐minute period, after which dental surgery was commenced.ref. adj12934-bib-0029 Four different intravenous premedicants for third molar extractions: (1) THC (0.022 mg/kg body weight); (2) THC (0.044 mg/kg); (3) diazepam (0.157 mg/kg) and (4) placebo (Ringer’s lactate).ref. adj12934-bib-0029 There was no significant analgesic effect from THC compared to placebo, and six subjects preferred placebo to low‐dose THC (0.022 mg/kg).ref. adj12934-bib-0029 On the contrary, Nitecka‐Buchta et al reported statistically significant effects of CBD formulation on TMD pain relief compared to placebo, after 14 days of transdermal application over the masseter muscles.ref. adj12934-bib-0022 These findings may suggest that a topical route of administration of CBD is effective for pain relief as it acts on peripheral CB1/CB2 receptors with a higher local bioavailability; the bioavailability of oral CBD is limited due to digestive enzymes.ref. adj12934-bib-0022
All the included articles investigated patients’ pain changes after administering the cannabinoid either after a specified time or over multiple time periods (Tables 2 and 3). There were variations between studies in sampling and cannabinoid used. Côté et al studied patients suffering from squamous cell carcinoma of the head and/or neck area and reported no significant difference in pain scores between those who received oral nabilone and placebo groups.ref. adj12934-bib-0026 Kalliomäki et al studied patients undergoing third molar extraction and divided the treatment population into three groups placebo, naproxen and an experimental synthetic CB1 and CB2 receptor agonist (AZD1940) compared to placebo.ref. adj12934-bib-0027 No significant differences in pain scores were noted between the two groups; however, the patients who took naproxen, a nonsteroidal anti‐inflammatory drug (NSAID), had significantly reduced pain.ref. adj12934-bib-0027 Ostenfeld et al also studied patients requiring a third molar extraction and a synthetic CB2 agonist (GW842166) at two doses (100 mg and 800 mg), compared to placebo and ibuprofen (800mg preoperative and 400mg postoperatively).ref. adj12934-bib-0028 They found no statistically significant difference for pain relief between placebo and 100 mg GW842166.ref. adj12934-bib-0028 There was improved pain relief with 800mg GW842166 but it was statistically insignificant.ref. adj12934-bib-0028 Notably, ibuprofen was found to be better, both statistically and clinically,at providing pain relief when compared with placebo.ref. adj12934-bib-0028
Raft et al included ten patients, each requiring removal of four third molars.ref. adj12934-bib-0029 THC was administered intravenously, at doses of 0.022 mg/kg and 0.044 mg/kg, with intravenous diazepam and intravenous placebo as comparison groups.ref. adj12934-bib-0029 Raft et al recorded changes in pain thresholds using a strain‐gauge algometer applied to the glabellar eminence that measured periosteal pain in gram pressure and a TECA B2 EMG nerve stimulator that applied a square wave electro‐cutaneous stimulus to skin of the acromion process.ref. adj12934-bib-0029 Patients responded to two psychophysical thresholds: a pain detection level and a pain tolerance level at which the stimulus intensity could no longer be withstood.ref. adj12934-bib-0029 Notably, no significant difference was produced between THC and placebo for pain tolerance.ref. adj12934-bib-0029 Furthermore, no rationale was provided for choosing intravenous route of THC administration, a route which is unfortunately not an option for the broader dental community, and thus poses issues with access and direct implication to the general dental practice.
Nitecka‐Buchta et al carried out a study measuring the outcome of TMJ pain after transdermal application of a CBD cream to masseter muscles.ref. adj12934-bib-0022 It was observed that pain intensity measured on the VAS scale after 14 days of twice daily CBD cream (Group 1) application to masseter muscles was significantly decreased from VASI = 5.6 (SD = 1.38) on Day 0, to VASII = 1.67 (SD = 1.44).ref. adj12934-bib-0022 The average pain level of masseter muscles after the application of the placebo formulation (Group 2) changed from VASI = 5.10 (SD = 1.26) on Day 0 to VASII = 4.60 (SD = 1.58) on Day 14.ref. adj12934-bib-0022 The reduction in pain intensity in VAS scale was statistically significant in Group 1 (70.2% reduction) and was not significant in Group 2 (9.81% reduction).ref. adj12934-bib-0022
Safety profile, tolerance and adverse events associated with cannabinoids can vary significantly between patients based on various factors, including but not limited to, the type of cannabinoid used, dosage and frequency, duration of use and comorbidities that may need additional treatment. No significant difference in adverse effects such as nausea, sleep and mood changes, drowsiness, anxiety and xerostomia were reported in the study that compared Nabilone and placebo during radiotherapy for head and neck cancers.ref. adj12934-bib-0026 Some of the adverse events noted in clinical trial involving AZD1940, a synthetic CB1/CB2 receptor agonist included postural dizziness, nausea, hypotension and headache and notably presyncope or syncope episodes.ref. adj12934-bib-0027 However, all the reported events were classed as mild to moderate in intensity and effected less than 10% of participants.ref. adj12934-bib-0027 Similar patterns of mild to moderate adverse events were noted in another RCT involving GW842166 wherein headache, nausea, pyrexia and syncope pharyngolaryngeal pain were reported across the study groups.ref. adj12934-bib-0028 In the study that utilized transdermal cannabidiol application for TMD, the authors stated that no adverse effects were recorded.ref. adj12934-bib-0022, ref. adj12934-bib-0029 In the study using intravenous tetrahydrocannabinol, no subjects experienced true clinical psychosis, however anxiety and some dysphoria were noted on administration of THC (0.022mg/kg) in six subjects.ref. adj12934-bib-0029 In fact, one subject became so anxious after receiving THC (0.022 mg/kg) that surgery had to be terminated; however this subject used hashish for the previous 18 months while on active duty in Vietnam, and declared that THC recalled frightening wartime experiences.ref. adj12934-bib-0029 Overall, the adverse reactions occurring with these specific products trialled in the respective patient groups caused mild‐moderate adverse effects.
DISCUSSION
As cannabinoids become more widely accepted and legally available, more research is being conducted regarding its medicinal use, particularly in pain relief and management. This can be evidenced by a range of recently published papers including a systematic review of randomized controlled trials led by the International Association for the Study of Pain (IASP) Presidential Task Force on Cannabis and Cannabinoid Analgesia.ref. adj12934-bib-0030, ref. adj12934-bib-0031 The aforementioned paper evaluated evidence on efficacy and adverse events associated with cannabinoid use for a range of chronic pain management and concluded that a very low‐quality evidence exists supporting their efficacy and significant adverse events were reported with their usage on short term (<7 days) and longer term usage (>7 days).ref. adj12934-bib-0030, ref. adj12934-bib-0031
In a recent anonymous online cross‐sectional survey conducted in Australia 2 years after legal access was introduced (in 2016), less than 3% of the respondents (25 out of 1044) had obtained cannabinoids through legal prescription which was attributed to lack of awareness of access through medical practitioner.ref. adj12934-bib-0032 This paper and an earlier report by the same group highlight the need for further research within this expanding option in pain management.ref. adj12934-bib-0032, ref. adj12934-bib-0033 However, exponential growth in popularity of cannabinoid usage over the last 4 years, specifically in cancer patients has significantly increased awareness. Furthermore, limited scientific literature around its use in dentistry have been published possibly due to restricted access and lack of compelling evidence for its efficacy, as noted in this review.
In our review, five publications that evaluated the use of cannabinoids on pain management, with varying results, were identified and appraised. Although all the included studies in the analysis were human studies, variations in sample populations, gender differences in study population, type of cannabinoid, routes of administration, and outcome measurements contributed to the heterogeneity of included studies. This presents difficulties when attempting to draw direct comparisons between studies to formulate concise conclusions.
Effective orofacial pain management is often achieved using a combination of anti‐inflammatory, steroids and NSAIDs. Of the studies included in our review, statistically significant pain relief was noted for TMD pain upon topical application of a transdermal CBD formulation over the masseter muscles. Its benefit may be derived from its action on peripheral CB1/CB2 receptors with a higher local bioavailability by avoiding the gut.ref. adj12934-bib-0022 This approach presents a new avenue for chronic pain management that avoids the adverse effects of first‐pass metabolism following enteral administration, especially in cases where higher drug concentrations are required to achieve a sufficient systemic bioavailability.ref. adj12934-bib-0022 However, the concerns around the adequacy (or lack of) oral formulations of the synthetic cannabinoids investigated by three other included studies to impart analgesic effects still exist.ref. adj12934-bib-0026, ref. adj12934-bib-0027, ref. adj12934-bib-0028 Additionally, very low and suboptimal intravenous drug concentrations use was a potential limitation in the study involving THC.ref. adj12934-bib-0029
Methodology of recording outcome measures did not differ between studies, aside from Raft et al.ref. adj12934-bib-0029 Pain was measured and recorded utilizing the Visual Analog Scale by most studies.ref. adj12934-bib-0022, ref. adj12934-bib-0026, ref. adj12934-bib-0027, ref. adj12934-bib-0028As such, the quality of these studies in terms of outcome measurements were relatively equivalent despite the VAS score being a relatively blunt instrument. However, Raft et al recorded changes in pain detection and pain tolerance thresholds after drug administration using a cutaneous and pressure stimulus, as previous discussed.ref. adj12934-bib-0029 Studies included in this review treated different types of pain, which contributes to the variation between studies. Furthermore, four included studies reported pain intensity as their outcome measure whilst only Raft et al ref. adj12934-bib-0029 assessed pain tolerance. Three studies treated patients requiring third molar extractions and sought to manage the associated post‐operative pain.ref. adj12934-bib-0027, ref. adj12934-bib-0028, ref. adj12934-bib-0029 Côté et al included patients that were undergoing radiotherapy and/or chemotherapy for squamous cell carcinoma, while Nitecka‐Buctha et al studied patients that suffered from TMD pain.ref. adj12934-bib-0022, ref. adj12934-bib-0026 These differing patient types with varying causes of pain may result in different reported pain and relief, ultimately impacting measured outcomes and the ability to draw comparisons. For instance, the acuity and chronicity of pain differed between studies, as third molar extractions typically result in acute pain compared to TMD and cancer, which may have a chronic element of pain.ref. adj12934-bib-0022, ref. adj12934-bib-0027 Furthermore, pain can be a personal and subjective experience, mainly when anxiety, certain perceptions or past experiences are involved.ref. adj12934-bib-0034 Consequently, tailored research may need to be pursued for each of these types of pain and evaluate how different cannabinoids, pain perceptions and experiences affect pain relief.
The included studies had a range of limitations. The sample size in Côté et al study was relatively small (56 patients) and from a single centre.ref. adj12934-bib-0026 Twenty‐four participants dropped out, seemingly related to their health status, therefore, it is possible that the absence of outcome data depended on its true value (Domain 3, Table 4); this gives rise to loss‐to‐follow‐up bias.ref. adj12934-bib-0024 Additionally, there were some concerns in the randomization process as there was an unbalanced distribution of patients with advanced carcinoma, receiving combined modality treatments (radiochemotherapy) were unequally represented in both groups (Domain 1).ref. adj12934-bib-0024, ref. adj12934-bib-0026 This could explain higher dropouts from the placebo group, or alternatively there may have been less dropouts from the nabilone group due to the benefits of this intervention—neither conclusion can be drawn with certainty. Studies by Kalliomäki et al and Ostenfeld et al raise some concerns of bias as random component used in sequence generation, nor was the concealment of allocation sequence (Domain 1, Table 4) was included.ref. adj12934-bib-0027, ref. adj12934-bib-0028 Raft et al’s crossover trial raised some concerns of bias due to inadequacies of information surrounding period and carry‐over effects (Domain S, Table 5).ref. adj12934-bib-0024, ref. adj12934-bib-0029 Additionally, despite a homogenous sample that did not differ in their pre‐surgical anxiety ‘trait’ levels, one subject had to be excluded because they became so anxious after injection of THC (0.022 mg/kg) that surgery had to be terminated.ref. adj12934-bib-0029 However, missing outcome data from one participant in one intervention are quite small to have any significant difference on the estimated effect of intervention.ref. adj12934-bib-0029 In addition, the measurement of pain threshold /tolerance as an outcome instead of pain relief made it difficult to compare this study with others.
This paper sought to review RCTs of synthetic and natural cannabinoids. The inclusion criteria were expanded to include all these cannabinoids due to the known ethical barriers associated with investigating marijuana use in subjects. Moreover, the legality of recreational or medicinal marijuana widely varies per jurisdiction.ref. adj12934-bib-0005, ref. adj12934-bib-0013 As a result, none of the included studies investigated cannabinoid use by inhalation or sublingual administration. There are some non‐randomized studies investigating the benefits of marijuana for HNC patients, however, these subjects were mostly existing marijuana users.ref. adj12934-bib-0019, ref. adj12934-bib-0021 Hence, this review supports the recommendation that further studies are needed to investigate the benefits of cannabinoids in different formulations to inform future policy changes surrounding its use as a dental therapeutic. While four out of five studies included in this review failed to find significantly positive results, some studies that were excluded from this review (due to non‐RCT design) reported a positive association between cannabinoid use and pain relief. In a recent prospective, open‐label, single‐arm pilot study, Gambino et al evaluated the use of C. sativa oil in the management of patients diagnosed with primary BMS.ref. adj12934-bib-0020 The subjects showed a statistically significant improvement in the clinical remission of oral symptoms over time.ref. adj12934-bib-0020 Levels of anxiety and depression also demonstrated a statistically significant and favourable improvement with no reports of severe reactions.ref. adj12934-bib-0020 One‐third of the patients experienced adverse events; nevertheless, no subjects had to discontinue the treatment due to these adverse events, which were all transient.ref. adj12934-bib-0020 The most common adverse event reported was dizziness occurring in three out of 17 patients (17.6%), followed by headache (11.8%) and constipation (5.9%).ref. adj12934-bib-0020 Overall, the treatment was well‐tolerated and effective in patients similar to some studies included in this review.ref. adj12934-bib-0020
While Côté et al did not find nabilone to be effective at improving the QOL of HNC patients, other non‐randomized studies concluded that marijuana use did lead to pain relief in HNC patients.ref. adj12934-bib-0026 Elliot et al carried out a cross‐sectional study investigating why patients with a history of HNC treated with radiotherapy were using medical marijuana.ref. adj12934-bib-0019 Using four different QOL questionnaires in their sample of 15 patients, they found that medical marijuana subjectively assisted with pain management and other long‐term side effects of radiotherapy, including altered sense, appetite, depression, dysphagia, and xerostomia.ref. adj12934-bib-0019 Zhang et al, in a prospective cohort study, compared similar QOL measures in recreational marijuana users case‐matched with nonusers who were newly diagnosed with HNC and treated in a tertiary setting.ref. adj12934-bib-0021 They supported Elliot et al‘s findings, with statistically significantly lower pain scores in marijuana users (loose‐leaf marijuana) than nonusers.ref. adj12934-bib-0021 Similarly, statistically significant improvement was found for anxiety/depression, tiredness, drowsiness, appetite, and general well‐being measured using (Edmonton Symptom Assessment System (ESAS) score).ref. adj12934-bib-0021 However, Elliott et al’s study had a small sample size and both studies relied on samples of historical users of marijuana, which limits the external validity of the results.ref. adj12934-bib-0019, ref. adj12934-bib-0021 These factors may contribute to the differences in the randomized, double‐blind, placebo‐controlled HNC trial performed by Côté et al. Conversely, it should be acknowledged that other constituents and effect(s) of cannabinoids may be responsible for reduced pain perception, as compared to the synthetic cannabinoid studied by Côté et al which mainly agonizes CB1/CB2 receptors.ref. adj12934-bib-0006, ref. adj12934-bib-0026
CONCLUSIONS
The cannabinoids CBD and THC exhibit a wide spectrum of antinociceptive and anti‐inflammatory actions with a range of side effects.ref. adj12934-bib-0004, ref. adj12934-bib-0005, ref. adj12934-bib-0020 Theoretically, there is great potential to use cannabinoids in the management and treatment of orofacial pain, however, its use in healthcare remains controversial and in its infancy.ref. adj12934-bib-0005 Specifically, THC as an intravenously delivered cannabinoid has limited potential for use in dental setting due to dentists rarely using intravenous mode of medication delivery and risk of psychogenic effects. Generally, a low‐quality evidence supporting the use of cannabinoids to treat pain and inflammation exist, with a lack of consistent and compelling high‐quality evidence pertaining to its effectiveness in orofacial pain. Although one study in this review reports positive effects, insufficient evidence exists to support a tangible clinical benefit of natural and synthetic cannabinoids in managing orofacial pain, especially for drugs delivered into systemic circulation.ref. adj12934-bib-0022 Despite the one study demonstrating topical CBD may be beneficial for treating orofacial pain, further research is needed prior to its use. Future research in the form of rigorous randomized studies, including crossover RCTs to evaluate varying doses of topical and systemic cannabinoids and possible interactions with other medications is suggested. As the therapeutic benefits of natural and synthetic cannabinoids continue to evolve, dental professionals will need to be familiar with the potential indications for use and possible interactions in the practice setting in an era of constantly changing legislation.
REGISTRATION AND PROTOCOL
This review was carried out in accordance with the PRISMA guidelinesref. adj12934-bib-0023 and registered with PROSPERO 2022 Registration number CRD42022274854 available from: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42022274854
DISCLOSURE
Nothing to declare.
| Electronic Database | MeSH terms and keywords search |
|---|---|
| Ovid | exp Dentistry/ or exp Stomatognathic Diseases/ or exp Pharyngeal Diseases/ or exp neck/ or exp facial bones/ or exp jaw/ or pterygopalatine fossa/ or exp sphenoid bone/ or exp temporal bone/ or exp temporomandibular joint/ or exp stomatognathic system/ or exp pharynx/ or exp facial nerve/ or glossopharyngeal nerve/ or exp trigeminal nerve/ or "head and neck neoplasms"/ or exp otorhinolaryngologic neoplasms/ or exp maxillary sinus/ or exp Odontogenic Tumors/Cannabis/ or exp Cannabinoids/ or exp "Marijuana Use"/ or Medical Marijuana/ or Marijuana Abuse/ or exp Receptors, Cannabinoid/ or exp Endocannabinoids/exp Pain/ or Pain Management/ or exp "Anesthesia and Analgesia"/ or exp analgesics/ or exp muscle relaxants, central/ or exp Inflammation/ or exp Anti‐Inflammatory Agents/ or exp somatosensory disorders/ or exp Pain Perception/ or Pain Measurement/ or Nociceptors/(dent* or orofacial or "oral medicine" or "oral pathology" or "oral surgery" or temporomandibular or trigeminal or molar or tooth* or teeth or myofunctional or periodont* or endodont* or (osteonecrosis and jaw) or periapical or "radicular cyst" or "burning mouth" or "dry socket" or "alveolar osteitis" or stomatitis or "oral mucositis" or jaw or mandib* or maxilla* or masticat* or "oral mucosa" or "oral ulcers" or masseter or tongue or saliva* or pharyn* or (head and neck) or "alveolar bone" or temporalis or oropharyn* or labial* or lip or pulp or palate or palatal or odont*).mp. [mp=title, abstract, original title, name of substance word, subject heading word, floating sub‐heading word, keyword heading word, organism supplementary concept word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier, synonyms](cannab* or marijuana or hemp or nabilone or dronabinol or THC or tetrahydrocannabinol or CBD or nabiximol or sativex or nanabis).mp. [mp=title, abstract, original title, name of substance word, subject heading word, floating sub‐heading word, keyword heading word, organism supplementary concept word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier, synonyms](pain or analges* or anaesthe* or inflammat* or "muscle relaxant" or anti‐inflammatory or antiinflammatory or nocicept* or ache or antinocicept* or hyperalgesia or allodynia or neuropathic pain).mp. [mp=title, abstract, original title, name of substance word, subject heading word, floating sub‐heading word, keyword heading word, organism supplementary concept word, protocol supplementary concept word, rare disease supplementary concept word, unique identifier, synonyms]1 or 42 or 53 or 67 and 8 and 9 |
| PubMed | (cannab* or marijuana or "cbd" or "thc" or nabilone or hemp or nabiximol or sativex or dronabinol)AND (dent* or orofacial or tooth* or head or neck or odont* or teeth or jaw or stomatitis or trigeminal)AND (pain or analges* or anaesthe* or inflammat* or “muscle relaxant” or antiinflammatory or anti‐inflammatory or nocicept* or antinocicept* or hyperalgesia or allodynia or neuropathic pain) |
| Scopus | (dent* OR "third molar" OR "tooth extraction" OR pulp OR toothache OR "oral cancer" OR "burning mouth" OR "trigeminal neuralgia" OR "oral mucositis" OR "head and neck" OR temporomandibular OR odont* OR “dry socket” OR “alveolar osteitis” OR “oral surgery” OR stomatitis OR pharyn* OR jaw OR orofacial)AND (cannab* OR marijuana OR hemp OR dronabinol OR "illicit drug" OR cbd OR thc OR nabilone OR nabiximol OR sativex OR tetrahydrocannabinol)AND (pain OR analges* OR anaesthe* OR inflammat* OR “muscle relaxant” OR anti‐inflammatory OR antiinflammatory OR nocicept* OR "neuropathic pain" OR hyperalgesia OR allodynia) |
| ClinicalTrials.gov | Cannabis | Completed Studies | Studies with Results | insert keyword consecutively from list 1‐13 below |
| Cochrane Library Trials | (cannab* or marijuana or "cbd" or "thc" or nabilone or hemp or nabiximol or sativex or dronabinol)AND (dent* or orofacial or tooth* or head or neck or odont* or teeth or jaw or stomatitis or trigeminal or temporomandibular)AND (pain or analges* or anaesthe* or inflammat* or “muscle relaxant” or antiinflammatory or anti‐inflammatory or nocicept* or antinocicept* or hyperalgesia or allodynia or neuropathic pain) |
| Domain | Signalling questions for individual‐randomized, parallel‐group trials | Signalling questions for crossover trials |
|---|---|---|
| 1. Randomization process | ||
| 1.1 | Was the allocation sequence random? | |
| 1.2 | Was the allocation sequence concealed until participants were enrolled and assigned to interventions? | |
| 1.3 | Did baseline differences between intervention groups suggest a problem with the randomization process? | Did baseline differences between intervention groups at the start of the first period suggest a problem with the randomization process? |
| S. Bias arising from period and carry‐over effects | ||
| S1 | n/a | Was the number of participants allocated to each of the sequences equal or nearly equal? |
| S2 | n/a | If N/PN/NI to S.1: Were period effects accounted for in the analysis? |
| S3 | n/a | Was there sufficient time for any carryover effects to have disappeared before outcome assessment in the second period? |
| 2. Deviations from intended interventions | ||
| 2.1 | Were participants aware of their assigned intervention during the trial? | Were participants aware of their assigned intervention during each period of the trial? |
| 2.2 | Were carers and people delivering the interventions aware of participants’ assigned intervention during the trial? | Were carers and people delivering the interventions aware of participants’ assigned intervention during each period of the trial? |
| 2.3 | If Y/PY/NI to 2.1 or 2.2: Were there deviations from the intended intervention that arose because of the trial context? | |
| 2.4 | If Y/PY to 2.3: Were these deviations likely to have affected the outcome? | |
| 2.5 | If Y/PY/NI to 2.4: Were these deviations from intended intervention balanced between groups? | If Y/PY/NI to 2.3: Were these deviations from intended intervention balanced between interventions? |
| 2.6 | Was an appropriate analysis used to estimate the effect of assignment to intervention? | |
| 2.7 | If N/PN/NI to 2.6: Was there potential for a substantial impact (on the result) of the failure to analyse participants in the group to which they were randomized? | |
| 3. Missing outcome data | ||
| 3.1 | Were data for this outcome available for all, or nearly all, participants randomized? | |
| 3.2 | If N/PN/NI to 3.1: Is there evidence that the result was not biased by missing outcome data? | |
| 3.3 | If N/PN to 3.2: Could missingness in the outcome depend on its true value? | |
| 3.4 | If Y/PY/NI to 3.3: Is it likely that missingness in the outcome depended on its true value? | |
| 4. Measurement of the outcome | ||
| 4.1 | Was the method of measuring the outcome inappropriate? | |
| 4.2 | Could measurement or ascertainment of the outcome have differed between intervention groups? | Could measurement or ascertainment of the outcome have differed between interventions within each sequence? |
| 4.3 | If N/PN/NI to 4.1 and 4.2: Were outcome assessors aware of the intervention received by study participants? | |
| 4.4 | If Y/PY/NI to 4.3: Could assessment of the outcome have been influenced by knowledge of intervention received? | |
| 4.5 | If Y/PY/NI to 4.4: Is it likely that assessment of the outcome was influenced by knowledge of intervention received? | |
| 5. Selection of the reported result | ||
| 5.1 | Were the data that produced this result analysed in accordance with a pre‐specified analysis plan that was finalized before unblinded outcome data were available for analysis? | |
| 5.2 | Is the numerical result being assessed likely to have been selected, on the basis of the results, from multiple eligible outcome measurements (e.g. scales, definitions, time points) within the outcome domain? | |
| 5.3 | Is the numerical result being assessed likely to have been selected, on the basis of the results, from multiple eligible analyses of the data? | |
| 5.4 | n/a | Is a result based on data from both periods sought, but unavailable on the basis of carryover having been identified? |
References
- The origin and use of cannabis in Eastern Asia linguistic‐cultural implications.. Econ Bot, 1974
- Ancient cannabis burial shroud in a Central Eurasian cemetery.. Econ Bot, 2016
- Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders.. Epilepsia, 2014. [PubMed]
- The therapeutic potential of cannabis and cannabinoids.. Dtsch Ärztebl Int, 2012. [PubMed]
- Cannabis & orofacial pain: a systematic review.. Br J Oral Maxillofac Surg, 2021. [PubMed]
- Cannabis, a complex plant: different compounds and different effects on individuals.. Ther Adv Psychopharmacol, 2012. [PubMed]
- Cannabinoid receptors and the endocannabinoid system: signaling and function in the central nervous system.. Int J Mol Sci, 2018. [PubMed]
- Targeting CB2 receptors and the endocannabinoid system for the treatment of pain.. Brain Res Rev, 2009. [PubMed]
- Vanilloid TRPV1 receptor mediates the antihyperalgesic effect of the nonpsychoactive cannabinoid, cannabidiol, in a rat model of acute inflammation.. Br J Pharmacol, 2004. [PubMed]
- Differential TRPV1 and TRPV2 channel expression in dental pulp.. J Dent Res, 2011. [PubMed]
- 11 Department of Health Office of Medicinal Cannabis . Clinical guidance: for the use of medicinal cannabis products in Queensland. Brisbane: Queensland Health, 2018.
- 12 Australian Institute of Health and Welfare . Alcohol, tobacco & other drugs in Australia. Canberra: Australian Institute of Health and Welfare, 2022.
- Prescribing medicinal cannabis.. Australian prescriber, 2020. [PubMed]
- 14 (Queensland) ADA . Important changes to the medicines and poisons act. Bowen Hills: ADAQ, 2021.
- 15 Therapeutic Goods Administration . Over‐the‐counter access to low dose cannabidiol. Canberra: Therapeutic Goods Administration, 2020.
- 16 Australian Register of Therapeutic Goods. Canberra: Therapeutic Goods Administration, 2019.
- 17 Pharmaceutical Services . Prescriptions and dispensing. St Leonards: New South Wales Government Health, 2022.
- 18 Jung K , Piomelli D . Lipids: the endocannabinoid system. In: Jez J , ed. Encyclopedia of Biological Chemistry III. 3rd ed. Amsterdam: Elsevier, 2021:776–784.
- Medical marijuana use in head and neck squamous cell carcinoma patients treated with radiotherapy.. Support Care Cancer, 2016. [PubMed]
- Evaluating the suitability and potential efficiency of cannabis sativa oil for patients with primary burning mouth syndrome: a prospective, open‐label, single‐arm pilot study.. Pain Med, 2021. [PubMed]
- Association of marijuana use with psychosocial and quality of life outcomes among patients with head and neck cancer.. JAMA Otolaryngol Head Neck Surg, 2018. [PubMed]
- Myorelaxant effect of transdermal cannabidiol application in patients with TMD: a randomized, double‐blind trial.. J Clin Med, 2019. [PubMed]
- The PRISMA 2020 statement: an updated guideline for reporting systematic reviews.. Syst Rev, 2021. [PubMed]
- RoB 2: a revised tool for assessing risk of bias in randomised trials.. BMJ, 2019
- 25 Phillips B , Ball C , Sackett D , et al. Oxford Centre for Evidence‐Based Medicine: levels of evidence (March 2009). Oxford: Centre for Evidence‐Based Medicine, 2009.
- Improving quality of life with nabilone during radiotherapy treatments for head and neck cancers: a randomized double‐blind placebo‐controlled trial.. Ann Otol Rhinol Laryngol, 2016. [PubMed]
- Evaluation of the analgesic efficacy of AZD1940, a novel cannabinoid agonist, on post‐operative pain after lower third molar surgical removal.. Scand J Pain, 2013. [PubMed]
- A randomized, controlled study to investigate the analgesic efficacy of single doses of the cannabinoid receptor‐2 agonist GW842166, ibuprofen or placebo in patients with acute pain following third molar tooth extraction.. Clin J Pain, 2011. [PubMed]
- Effects of intravenous tetrahydrocannabinol on experimental and surgical pain. Psychological correlates of the analgesic response.. Clin Pharmacol Ther, 1977. [PubMed]
- Cannabinoids, cannabis, and cannabis‐based medicine for pain management: a systematic review of randomised controlled trials.. Pain, 2021. [PubMed]
- International Association for the Study of Pain Presidential Task Force on Cannabis and Cannabinoid Analgesia: research agenda on the use of cannabinoids, cannabis, and cannabis‐based medicines for pain management.. Pain, 2021. [PubMed]
- Medical cannabis use in the Australian community following introduction of legal access: the 2018–2019 Online Cross‐Sectional Cannabis as Medicine Survey (CAMS‐18).. Harm Reduction Journal, 2020. [PubMed]
- Medicinal cannabis in Australia, 2016: the Cannabis as Medicine Survey (CAMS‐16).. Med J Aust, 2018. [PubMed]
- Individual differences in the subjective experience of pain: new insights into mechanisms and models.. Headache, 2010. [PubMed]
- Cannabis use for chronic non‐cancer pain: results of a prospective survey.. Pain, 2003. [PubMed]
- Adjuvant topical therapy with a cannabinoid receptor agonist in facial postherpetic neuralgia.. J Dtsch Dermatol Ges, 2010. [PubMed]
- Palmitoylethanolamide versus a nonsteroidal anti‐inflammatory drug in the treatment of temporomandibular joint inflammatory pain.. J Orofac Pain, 2012. [PubMed]
