Understanding the role of oxylipins in Cannabis to enhance cannabinoid production
Abstract
Phytocannabinoids are medically important specialized defense compounds that are sparsely distributed among plants, yet Cannabis sativa can synthesize unprecedented amounts of these compounds within highly specialized surface cell factories known as glandular trichomes. The control mechanisms that allow for this high level of productivity are poorly understood at the molecular level, although increasing evidence supports the role of oxylipin metabolism in phytocannabinoid production. Oxylipins are a large class of lipid-based oxygenated biological signaling molecules. Although some oxylipins are known to participate in plant defense, roles for the majority of the ca. 600 plant oxylipins are largely unknown. In this review, we examine oxylipin gene expression within glandular trichomes and identify key oxylipin genes that determine the fate of common lipid precursors. Mechanisms by which oxylipins may be interacting with phytocannabinoid metabolism, as well as specialized plant metabolism more broadly, are discussed and a model summarizing these contributions proposed.
Article type: Review Article
Keywords: glandular trichomes, green leaf volatiles, jasmonates, lipoxygenase, oxylipins, specialized metabolites
Affiliations: Australian Research Council Research Hub for Medicinal Agriculture, Department of Ecological Plant and Animal Sciences, School of Agriculture, Biomedicine and Environment, La Trobe University, Bundoora, VIC, Australia; La Trobe Institute of Sustainable Agriculture and Food, Department of Ecological Plant and Animal Sciences, School of Agriculture, Biomedicine and Environment, La Trobe University, Bundoora, VIC, Australia; Australian Research Council Research Hub for Protected Cropping, Department of Ecological Plant and Animal Sciences, School of Agriculture, Biomedicine and Environment, La Trobe University, Bundoora, VIC, Australia; Australian Research Council Research Hub for Sustainable Crop Protection, Department of Ecological Plant and Animal Sciences, School of Agriculture, Biomedicine and Environment, La Trobe University, Bundoora, VIC, Australia
License: Copyright © 2025 Senevirathne, Gendall, Johnson and Welling CC BY 4.0 This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
Article links: DOI: 10.3389/fpls.2025.1568548 | PubMed: 40343123 | PMC: PMC12058684
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (677 KB)
Introduction
Cannabis sativa L. (Cannabis) is a chemically complex plant that has a repertoire of more than 500 diverse molecules that span several classes of specialized metabolites, including a large class of medically important isoprenylated resorcinol aliphatic polyketides known as phytocannabinoids (PCs). PCs serve as the active pharmaceutical ingredients in medicines used as adjunct therapies for drug-resistant epilepsies (Epidiolex) and multiple sclerosis (Sativex), as well as nausea, pain, and loss of appetite (Dronabinol) (ref. Lichtman et al., 2018; ref. Thiele et al., 2018). In plants, PCs play a critical role in defense responses, including protection from UV radiation, herbivorous insects, and microbial pathogens, but the exact role of PCs in Cannabis is poorly understood (ref. Tanney et al., 2021). While many of the enzymes responsible for producing these metabolites have been elucidated, the control mechanisms governing their synthesis remain underexplored (ref. Stout et al., 2012; ref. Welling et al., 2023). Increasing evidence suggests that PC production may be mediated by oxylipins (ref. Bailey, 2019; ref. Booth et al., 2020; ref. Burgel et al., 2020; ref. Apicella et al., 2022; ref. Garrido et al., 2022; ref. Welling et al., 2023; T1).
Table 1: Effect of JAs on PC production and glandular trichome density in Cannabis.
| Cannabis variety | MeJA/JA treatment | Effect on PC production/trichome density | Changes in gene expression | Reference |
|---|---|---|---|---|
| Medicinal (Skunk) | 100 μM jasmonic acid/0.3 mM MeJA applied to cell cultures | No detectable level of Δ9-THC | No detectable change in THCAS | ref. Flores-Sanchez et al., 2009 |
| Industrial hemp (Santhica) | 0.03, 0.1, 0.3, 1, and 3 mM jasmonic acid applied to 15-day old seedlings | Not reported | Higher expression of LOX2 and 4-coumarate-CoA ligase-like 7 | ref. Behr et al., 2018 |
| Industrial hemp (Cherry, Cherry Blossom, Canada) | 44.9 mg/L MeJA applied as foliar and roots as separate treatments for 7weeks old plants | A significant increase in CBDA (>2-fold) in foliar applications in all three varietiesA decrease in Δ9-THC in and increase in total PC content | Not reported | ref. Bailey, 2019 |
| Medicinal (White Tangy Haze) | 0, 100, 500, and 1000 μM total plant spray MeJA after 2 weeks of flower development | A significant increase inΔ9-THC in 1000 μM at week 1 (>0.1-fold) and week 4 (>0.07-fold) | Not reported | ref. Apicella et al., 2022 |
| Medicinal (Beatriz) | 0, 0.1, 1.0, and 10 mM MeJA, 8 applications (once a week) after shifting to reproductive photoperiod | An increase in Δ9-THCA (>0.08-fold) and CBDA (>0.15-fold) in response to 0.1 mMA decrease in Δ9-THCA and CBDA in response to 10 mM | No significant change in the expression of AOC-1, AOC-2, PT4, THCAS, and CBDAS | ref. Garrido et al., 2022 |
| Not reported | 150 μM MeJA applied to 4-week-old plants | Not reported | Significant increase in expression of CsLOX1, CsLOX2, CsLOX5, CsLOX8, CsLOX10, CsLOX11, CsLOX12, CsLOX3, CsLOX17 2 hours and CsLOX1, CsLOX2, CsLOX5, CsLOX10, CsLOX11, CsLOX12, CsLOX16, 24 hours post application | ref. Fayaz et al., 2023 |
| Industrial hemp (MW6-15) | 0, 1, 7.5, and 15 mM MeJA, foliar spray on 14th days after transitioning into reproductive photoperiod, after initiation of terminal flowering and 7 and 14-days post first application | A significant increase (~1-fold) in total cannabinoids, Δ9-THCA, THCVA CBDA, CBDVA, and CBCA (> 1-fold) in response to 15 mM | JMT responsive to 7.5 and 15 mM MeJA in the leaf and inflorescence. LOX-L and HPL highly expressed in the leaf and inflorescence | ref. Welling et al., 2023 |
| Hemp (Hot Blond) | 100, 200, and 400 μM, foliar spray on 0 and 21 days after transitioning to reproductive photoperiod | A significant increase in glandular trichome density in calyx,A significant increase in CBD (>0.1-fold in both leaves and inflorescence) and Δ9-THC (>0.3-fold in leaves, >0.1-fold in inflorescence) in response to 100 mM | Not reported | ref. Hahm et al., 2024 |
| CANN97 | 1 µM MeJA applied 5 times at 3-day intervals to 18-day old plants | A significant increase in leaf sessile glandular trichome density and non-glandular trichome density | Significant increase in the expression of CsJAZ1, CsJAZ2, CsJAZ3, CsMYC1, CsMYC4, CsCOI1 | ref. Huang et al., 2024 |
| High CBD | 100 μM jasmonic acid as foliar spray, for consecutive 8 weeks after shifting to flowering photoperiod | No significant effect on overall glandular trichome density in bractsA decrease in Δ9-THCA, and CBDA but significant increase (> 0.2-fold) in Δ9-THC and CBD after 7 and 8 weeks in flowering | Not reported | ref. Oultram et al., 2024 |
| Not reported | Adventitious roots incubated in 0, 50, 100, 150, and 200 μM MeJA for 2 days | A significant increase (>0.5-fold) in CBD in 200 µM | Not reported | ref. Wang Y, et al., 2024 |
Oxylipins are structurally and functionally diverse, lipid-based biological signaling molecules that participate in plant immunity (ref. Blée, 2002; ref. Christensen et al., 2016; ref. Ameye et al., 2018; ref. Wasternack and Feussner, 2018; ref. Toporkova et al., 2024; f1). Oxylipins play vital roles in regulating biological functions in plants including growth and development, innate and induced defense mechanisms as response to diverse environmental elicitors and as secondary messengers activating defense compounds (ref. McGarry et al., 2024; ref. Wang et al., 2024; ref. Yokoyama et al., 2025). They are produced by the activity of lipoxygenases (LOXs) and various enzymes of the cytochrome P450 CYP74 family, such as allene oxide synthase (AOS) and hydroperoxide lyase (HPL) (ref. Song and Brash, 1991; ref. Howe et al., 2000; ref. Wasternack and Feussner, 2018). Jasmonates (JAs), including jasmonate-isoleucine (JA-Ile), jasmonic acid, and its methylated form methyl jasmonate (MeJA) are oxylipin-derived phytohormones formed from the LOX-AOS cascade (ref. Wasternack and Hause, 2013; ref. Wasternack and Feussner, 2018; ref. Ghorbel et al., 2021).
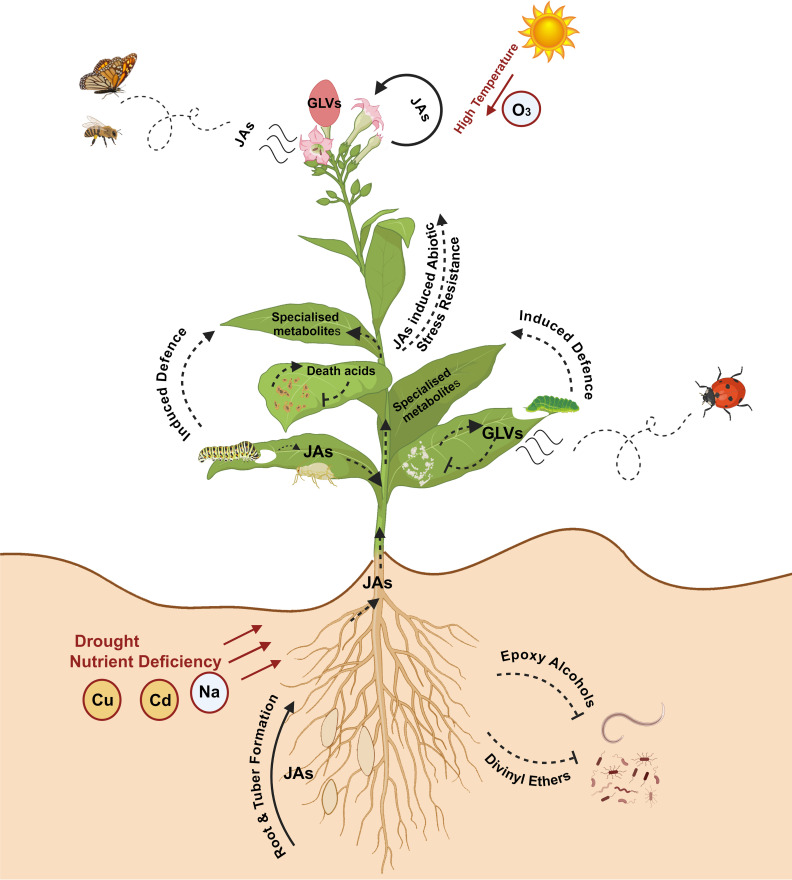
JAs induce specialized metabolism in diverse plant lineages, including Solanum lycopersicum (tomato) and Lavandula spp. (lavender), while also regulating the development of glandular trichomes and flowers where PCs are concentrated (ref. Boughton et al., 2005; ref. Chen et al., 2018; ref. Dong et al., 2022a; ref. Kianersi et al., 2022; ref. Yan et al., 2022). More recently, JAs have been shown to enhance the levels of PCs, although the intricate mechanism(s) driving this increase in PC production is not fully understood (T1). There is also an emerging hypothesis that green leaf volatiles (GLVs) formed via the LOX-HPL branch of oxylipin metabolism may provide substrate for the synthesis of hexanoic acid, the polyketide starter unit for PC production, although only limited evidence has been reported supporting this interaction (ref. Stout et al., 2012; ref. Welling et al., 2023). There has been considerable gene expansion in the LOX family in Cannabis compared with other species, and expression of some LOXs have been localized to the glandular trichomes where PCs are synthesized, suggesting that these may have acquired specialized roles in trichome biology and PC metabolism (ref. Livingston et al., 2020; ref. Borrego et al., 2023; ref. Fayaz et al., 2023; ref. Welling et al., 2023). Collectively, these data serve as evidence for the distinct yet incompletely understood role of the oxylipin pathway in PC production. In this review, we use a combination of phylogenetic and transcriptomic analyses to investigate the mechanisms by which oxylipins may influence PC production in Cannabis and propose a model highlighting these potential interactions.
Cannabis: a versatile and underutilized crop
Origin, classification and uses
Cannabis is a predominantly dioecious, herbaceous annual plant in the Cannabaceae family that originated in central Asia and now has a broad distribution (ref. Kovalchuk et al., 2020). This genus comprises only one species but exhibits high polymorphism, with subspecies: C. sativa ssp. sativa, C. sativa ssp. indica, and C. sativa ssp. rudelaris characterized by varying chemical and morphological features (ref. Small, 2015; ref. Hazekamp et al., 2016). Although the plant is known for its intoxicating properties, it has been used over millennia for medicine, fiber, and oil extraction (ref. Omare et al., 2021; ref. Shakil et al., 2021). Current applications also extend to phytoremediation, functional foods, and as ornamental plants (ref. Frassinetti et al., 2018; ref. Wu et al., 2021; ref. Hesami et al., 2022b). Classification of Cannabis has often been based on its end use, with recreational drug types typically referred to as ‘marijuana’ and fiber or seed types ‘industrial hemp’. Plants can also be classified into chemical phenotypes (chemotypes) based on their proportions of Δ9-tetrahydrocannabinol (Δ9-THC) or cannabidiol (CBD) PCs, or on the level of Δ9-THC, the principal intoxicant found in Cannabis (ref. Jin et al., 2021; ref. Hesami et al., 2022a; ref. Salamone et al., 2022).
Despite the multiple end uses of Cannabis, research on this plant has been constrained over several decades due to its narcotic status, which has limited germplasm exchange and the application of modern genetic improvement strategies (ref. Welling et al., 2016). Recent reforms to the cultivation and use of Cannabis by many legislators have enabled strong growth in licit Cannabis markets and these are projected to increase rapidly in the coming decades (ref. de Brito Siqueira et al., 2023). Similarly, legislative reform has allowed for an increase in Cannabis research, with recent efforts focusing on establishing genomic resources for Cannabis and improving understanding on factors driving PC yield, such as flowering behavior as well as trichome initiation and hormonal regulation (ref. Livingston et al., 2020; ref. Hurgobin et al., 2021; ref. Livingston et al., 2021; ref. Apicella et al., 2022; ref. Toth et al., 2022; ref. Sands et al., 2023; ref. Steel et al., 2023; ref. Dowling et al., 2024; ref. Huang et al., 2024).
PCs: important medicinal compounds
Extensive research has been conducted on the pharmacology of the two major PCs Δ9-THC and CBD, both of which have modulatory effects on the human endocannabinoid system (ECS). Δ9-THC is a partial agonist of type 1 G protein-coupled cannabinoid receptors (ref. Tagen and Klumpers, 2022). These receptors have important roles in regulating memory, mood, sleep, appetite, inflammation, and pain sensation, and their activation has been shown to have analgesic, neuroprotective, and anti-nausea effects (ref. Mechoulam and Parker, 2013). In contrast, CBD is also thought to modulate the ECS via a less understood mechanism involving other ECS targets and has been reported to counteract the intoxicating properties of Δ9-THC (ref. Devinsky et al., 2014). Medicines containing Δ9-THC and CBD are currently used in the treatment of pediatric epilepsies (e.g., Dravet syndrome and Lennox-Gastaut syndrome) and to alleviate side-effects of chemotherapy, with many other PCs, such as the varinoid PCs cannabidivarin (CBDV; GWP42006) and tetrahydrocannabivarin (THCV; GWP42004), at various levels of drug development (ref. Chanda et al., 2019; ref. Urits et al., 2019; ref. Britch et al., 2021). Other non-intoxicating and low abundant or minor PCs, including cannabichromene (CBC), cannabigerol (CBG) and their carboxylated analogues, cannabichromenic acid (CBCA) and cannabigerolic acid (CBGA), are also considered promising therapeutics (ref. Borrelli et al., 2013; ref. Anderson et al., 2021; ref. Cabrera et al., 2021).
Glandular trichomes are multicellular structures that synthesize, secrete, and store enormous amounts of PCs
Cannabis produces glandular as well as non-glandular trichomes on the surface of leaves and other parts of the plant body (ref. Livingston et al., 2020; ref. Xie et al., 2023). However, it is the capitate stalked glandular trichomes on modified leaves (perigonal bracts) surrounding female flowers that are the primary site for PC biosynthesis (f2). These trichomes are multicellular organs, consisting of a stalk, a disc of secretory cells, and a large globular head containing an extracellular cavity formed by cell wall delamination that stores the resinous compounds (ref. Hammond and Mahlberg, 1973; ref. Livingston et al., 2020; f2). These specific trichome morphotypes can produce substantial amounts of PCs, with these compounds contributing up to 40% of their dry weight (w/w) (ref. Livingston et al., 2020). Capitate stalked glandular trichome initiation and development are coordinated with the onset of flowering and likely influenced by several phytohormones, including gibberellins, cytokinins, and JAs, as have been established in numerous plant species (ref. Pattanaik et al., 2014; ref. Li et al., 2021; ref. Huang et al., 2024).
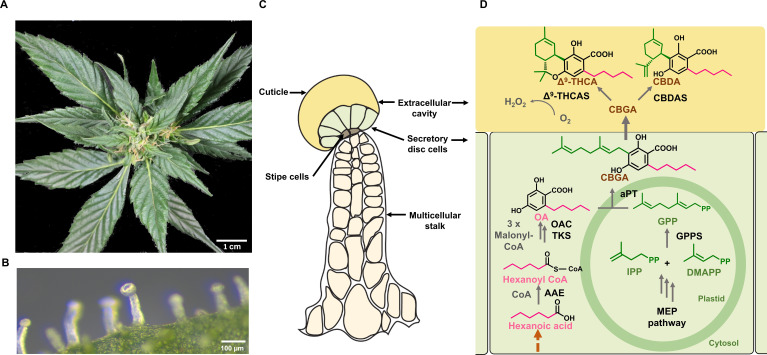
The biosynthesis of PCs traverses across several organelles within the secretory disc cells of glandular trichomes (f2). The precursors of PCs, olivetolic acid (OA), and geranyl pyrophosphate (GPP) are synthesized by the polyketide pathway in the cytosol and the methylerythritol 4-phosphate (MEP) pathway in plastids, while the final step occurs in the extracellular cavity (ref. Livingston et al., 2022; f2). OA is formed by a type III polyketide synthase (tetraketide synthase; TKS), which produces a tetraketide intermediate, while the accessory protein, OA cyclase (OAC) catalyzes a C2-C7 aldol condensation (ref. Gagne et al., 2012). Together, these enzymes form OA using three molecules of malonyl-CoA and an activated fatty acid, hexanoyl-CoA (ref. Stout et al., 2012). Prenylation of OA with GPP produce the first PC, CBGA, a step catalyzed by an aromatic prenyltransferase, CBGA synthase (CBGAS) (ref. Luo et al., 2019; ref. Sands et al., 2023). CBGA is then transported to the extracellular cavity and converted to Δ9– THCA and CBDA by Δ9– THCA and CBDA synthases, respectively (ref. Taura et al., 1996; ref. Sirikantaramas et al., 2005). This compartmentalization is thought to be attributed to the cytotoxicity of PCs and their bi-product H2O2 which is formed at a molar ratio of 1:1 during the oxidative cyclisation of the isoprenoid residue of CBGA (ref. Sirikantaramas et al., 2004, ref. 2005; f2). The acidic PCs are then non-enzymatically decarboxylated over time or when subjected to heat, forming the bioactive neutral PCs, such as Δ9-THC and CBD (ref. Sirikantaramas and Taura, 2017).
The origin of PC fatty acid precursors is unresolved
A key unanswered question in PC biosynthesis is the origin of the fatty acid hexanoic acid, which, once activated, serves as the C6 carbon starter unit for OA (ref. Welling et al., 2023; f2). Several metabolic pathways could synthesize hexanoic acid within the secretory disc cells of capitate stalked trichomes. In the trichome glands of Petunia, C6-C8 straight-chain acyl acids are formed from branched-chain amino acid catabolism and α-ketoacid elongation (ref. Kroumova et al., 1994). This pathway requires the activity of four proteins, yet transcripts for these genes have not been reported in Cannabis trichomes (ref. Kroumova et al., 1994; ref. Marks et al., 2009). A second pathway involves de novo fatty acid synthesis. This is supported by trichome-specific expressed sequence tags for a 3-oxoacyl- [acyl-carrier-protein (ACP)] reductase (fabG), as well as the association of this gene with changes in PC composition in a genome-wide association study (ref. Marks et al., 2009; ref. Welling et al., 2020). While C6-specific thioesterases required to terminate fatty acid elongation have not been recovered from trichome cells, this enzyme could reduce the β-keto group following the first two condensation reactions of acetyl CoA, forming hexanoic acid, and, instead of leaving the condensing enzyme, participate in additional reactions with malonyl CoA (ref. Horper and Marner, 1996; ref. Stout et al., 2012). Contrary to this hypothesis is the functional validation of a cytosolic-localized hexanoic acid-specific AAE (hexanoyl-CoA synthetase) which is highly expressed in Cannabis trichome secretory disc cells (ref. Stout et al., 2012).
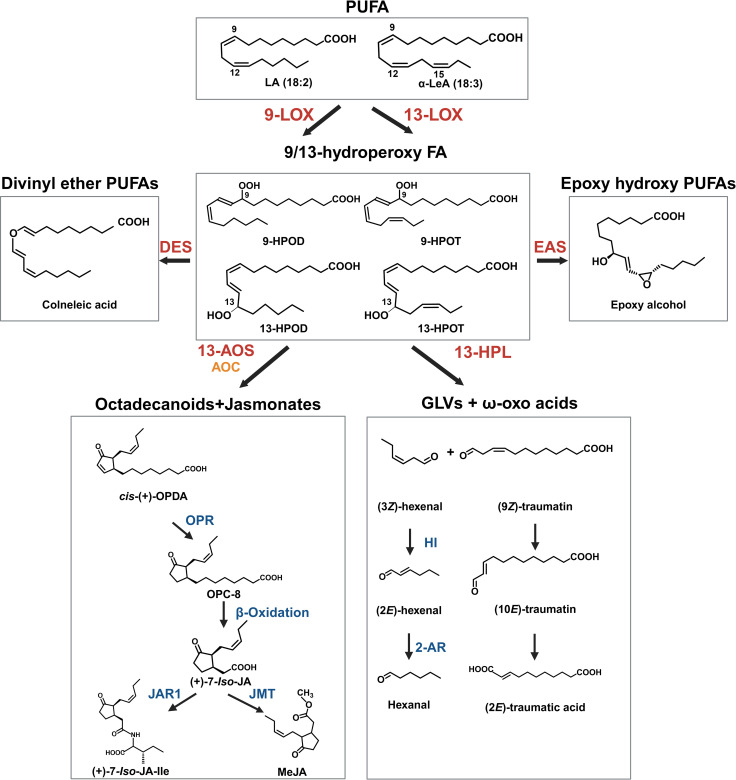
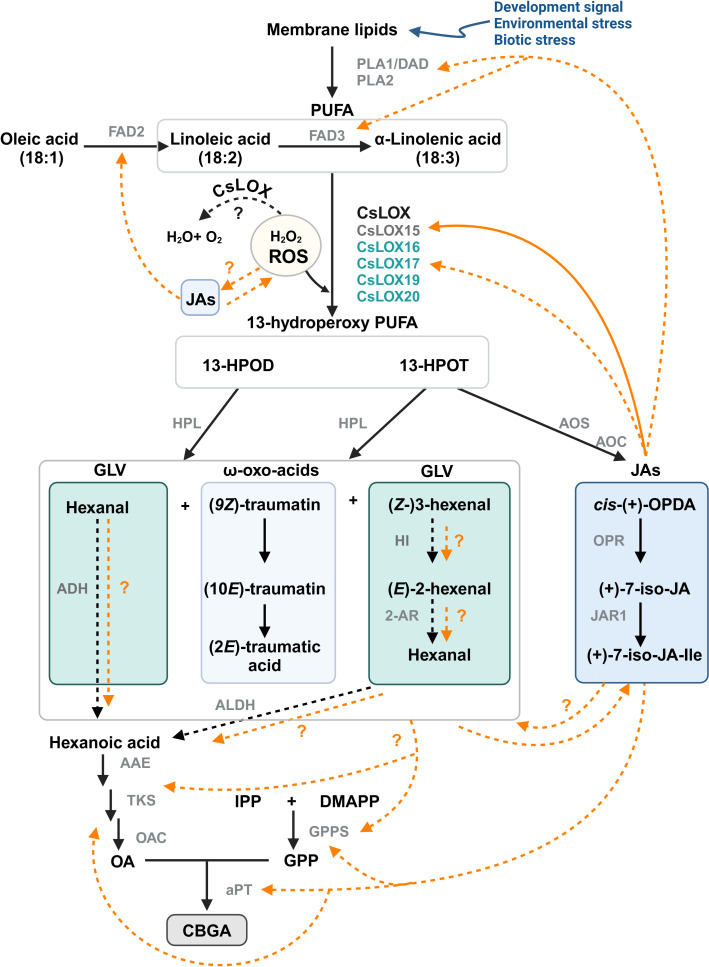
A third hypothetical pathway could involve an oxylipin-based origin (ref. Stout et al., 2012; ref. Welling et al., 2023). This would involve the degradation of polyunsaturated fatty acids (PUFAs), such as linoleic or linolenic acid, and the activity of LOX to form hydroperoxy PUFAs that can be cleaved into C6 aldehydes and progenitors of hexanoic acid (ref. Nakashima et al., 2013; ref. Welling et al., 2023; f3). Genes encoding desaturases, plastid-localized LOXs, and HPL that participate in these reactions are highly expressed in Cannabis trichomes (ref. Stout et al., 2012; ref. Balcke et al., 2017), while the secondary cleavage product of LOX/HPL, α-oxo acid 12-oxo-(10E)-dodecenoic acid ((10E)-traumatin) (f3), has recently been correlated with PC content in Cannabis inflorescences following treatment with the oxylipin phytohormone MeJA (ref. Welling et al., 2023; T1). C18 PUFAs and their oxylipin derivatives are also highly abundant in type VI stalked glandular trichomes of Solanum spp. and genes encoding acyl-hydrolyzing GDSL-type lipases that can release free C18 fatty acid substrates from galactolipids have been identified in Cannabis quantitative trait loci (QTL) for PC content (ref. Cropano et al., 2022). The following sections provide a detailed discussion of oxylipins in Cannabis and highlight key areas of research that could be used to understand the interaction between oxylipins and PC production.
Oxylipin pathway and metabolism
Key metabolic branch points determine the fate of oxylipin molecules and their biological functions
In plants, oxylipins are generated through the oxidation of PUFAs. PUFA oxidation can occur enzymatically by dioxygenases or nonenzymatic by the activity of free radicals (e.g., singlet oxygen (1O2)) (ref. Brash, 1999; ref. Zoeller et al., 2012; f3). The resulting hydroperoxides are highly reactive and act as key substrates in oxylipin metabolism, with the fate of oxylipin molecules decided by several enzymatic branch points including (1) the previously described HPL branch that forms C6 aldehydes, alcohols and acyl esters (collectively known as GLVs) concomitant with 12-oxo-(9Z)-dodecenoic acids ((9Z)-traumatin), (2) the JAs-forming AOS pathway, (3) the divinyl ether synthase (DES) pathway that forms divinyl ethers, and (4) the epoxy alcohol synthase/peroxygenase pathways that produce epoxy hydroxy PUFAs (ref. Wasternack and Hause, 2013; ref. Christensen et al., 2015, ref. 2016; f3). Oxylipins within each branch can have distinct roles (ref. Schlotzhauer et al., 1996; f3). For example, divinyl ethers and epoxy alcohols act as defense compounds in roots and leaves (ref. Blée, 1998; ref. Hamberg, 1999; ref. Weber et al., 1999; ref. Hamberg, 2004; ref. Sanadhya et al., 2021; ref. Gorina et al., 2022; ref. Toporkova et al., 2024; f1). The HPL and AOS branches are among the most extensively researched, with AOS-derived JAs having broad roles as signaling molecules, in reproduction, plant growth, and development (ref. Scala et al., 2013; ref. Ghasemi Pirbalouti et al., 2014; ref. Ameye et al., 2018; ref. Raza et al., 2021; f1, f3).
LOX serves as the critical juncture dictating oxylipin fate
LOXs, such as linoleate 13S-lipoxygenase (EC 1.13.11.12), serve as primary enzymes that mediate the fate of lipid precursors (f3). These are monomeric non-heme iron (Fe)-containing dioxygenases widely distributed in plants that convert PUFAs into hydroperoxides in a two-step reaction, involving the reduction of Fe3+ to Fe2+ by proton-coupled electron transfer, followed by oxygen insertion (ref. Egmond et al., 1973; ref. Goldsmith et al., 2002). LOXs are categorized into two main functional classes based on the carbon position they catalyze. For the 9-LOX and 13-LOX this is at the 9th and 13th position from the carbonyl carbon in the fatty acid, respectively, and can also be distinguished as type I and type II based on subcellular localization (f3). In addition, different LOX paralogs with the same regiospecificity can control different branches of the oxylipin pathway but are spatially separated. For example, in Zea mays, the JA-producing 13-LOX ZmLOX8 is localized to the chloroplast, while a GLV-producing 13-LOX ZmLOX10 is localized to non-photosynthetic plastids (ref. Christensen et al., 2013). While often associated with defense process, impaired LOX function has resulted in changes in plant reproduction, fruit ripening and root development (ref. Kolomiets et al., 2001; ref. Fortes et al., 2004; ref. Liu and Han, 2010; ref. Caldelari et al., 2011; ref. Vogt et al., 2013; ref. Liu et al., 2020).
Phylogenetic insights and structural features of Cannabis LOX proteins
Phylogenetic analysis of the Cannabis LOX protein family reveals distinct clades that may have specific metabolic functions
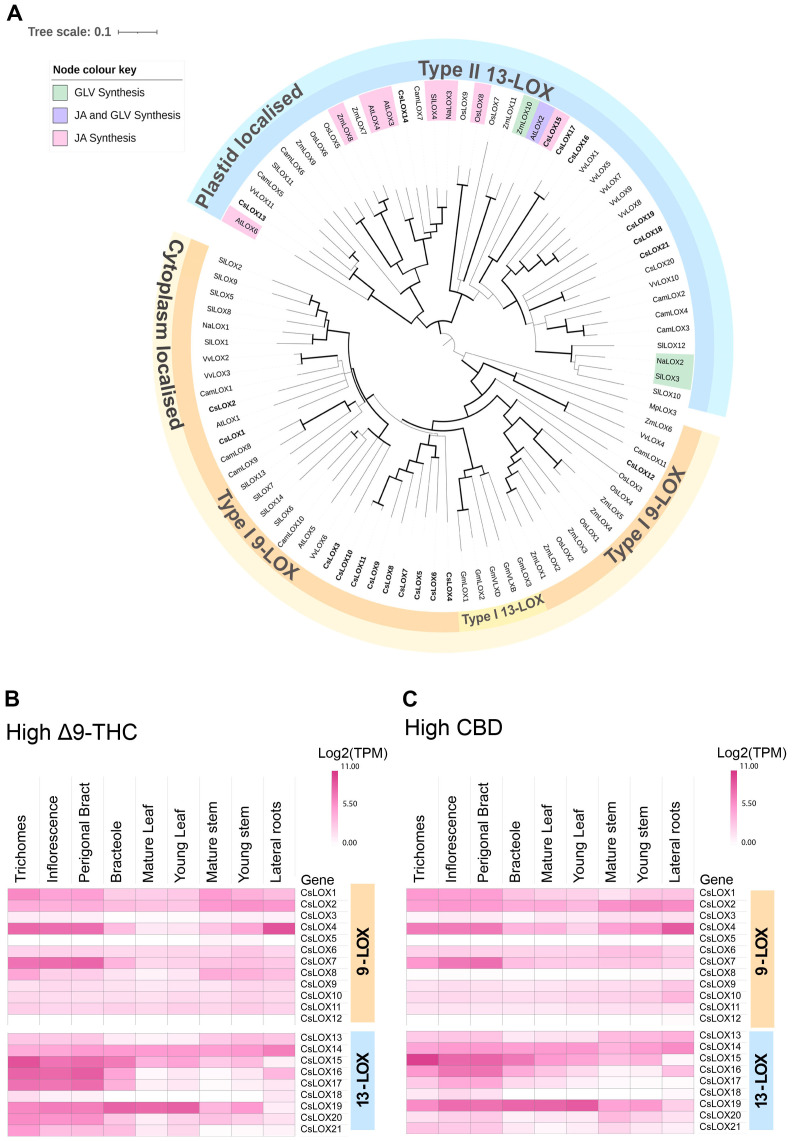
To explore the diversity of LOX gene family in Cannabis, a phylogenetic tree was constructed using protein sequences predicted from the Cannabis var. CBDRx genome [NCBI GenBank Accession: GCA_900626175.2] (Supplementary Table S1). Of the twenty-one Cannabis LOXs, all had the PLAT/LH2 domain (IPR001024) at the N terminus except CsLOX20 (T2, Supplementary Table S1). They also contained the C-terminal domain IPR013819 and were classified as LOX super family (IPR000907) members, which is consistent with authentic LOX proteins (ref. Chen et al., 2015; ref. Song et al., 2016). Twelve were clustered with the type-I cytosolic-localized 9-LOXs, while nine were clustered with the type-II plastidial-localized 13-LOX sub-class (f4). Within the 13-LOX sub-group, CsLOX13 and CsLOX14 form a clade with LOX orthologs associated with JA synthesis, while CsLOX15-CsLOX17 form a distinct clade that shows homology with AtLOX2, a dual functioning ortholog involved in both JA and GLV synthesis (ref. Chen et al., 2004; ref. Allmann et al., 2010; ref. Chauvin et al., 2013; ref. Christensen et al., 2013; ref. Shen et al., 2014; ref. Mochizuki et al., 2016; ref. He et al., 2020; f4).
Table 2: LOC IDs of Cannabis 21 LOXs and different nomenclature used in other studies.
| Nomenclature1 | LOC ID | AA length | Major domains | Annotation | Nomenclature of genes used by different authors | |||
|---|---|---|---|---|---|---|---|---|
| Protein family IPR000907 | PLAT/LH2 (IPR001024) | C Terminal (IPR013819) | ref. Fayaz et al. (2023) | ref. Welling et al. (2023) | ||||
| CsLOX1 | LOC115719608 | 860 | 21-860 | 18-161 | 163-860 | probable linoleate 9S-lipoxygenase 5 | CsLOX8 | |
| CsLOX2 | LOC115718693 | 955 | 116-955 | 113-256 | 258-955 | probable linoleate 9S-lipoxygenase 5 | CsLOX4 | |
| CsLOX3 | LOC115724062 | 873 | 32-873 | 29-172 | 174-873 | probable linoleate 9S-lipoxygenase 5 | CsLOX18 | |
| CsLOX4 | LOC115709296 | 859 | 18-859 | 15-160 | 162-869 | probable linoleate 9S-lipoxygenase 5 | CsLOX1 | |
| CsLOX5 | LOC115720291 | 848 | 34-848 | 5-149 | 151-848 | probable linoleate 9S-lipoxygenase 5 | CsLOX5 | |
| CsLOX6 | LOC115721132 | 859 | 42-859 | 18-160 | 162-859 | probable linoleate 9S-lipoxygenase 5 | CsLOX6 | |
| CsLOX7 | LOC115719336 | 871 | 29-871 | 21-163 | 165-871 | probable linoleate 9S-lipoxygenase 5 | CsLOX7 | |
| CsLOX8 | LOC115722276 | 869 | 51-869 | 19-161 | 163-869 | probable linoleate 9S-lipoxygenase 5 | CsLOX21 | |
| CsLOX9 | LOC115721268 | 875 | 52-875 | 25-167 | 169-875 | probable linoleate 9S-lipoxygenase 5 | CsLOX3 | |
| CsLOX10 | LOC115722275 | 868 | 45-868 | 18-160 | 162-868 | probable linoleate 9S-lipoxygenase 5 | CsLOX20 | |
| CsLOX11 | LOC115723988 | 868 | 45-868 | 18-160 | 162-868 | probable linoleate 9S-lipoxygenase 5 | CsLOX19 | |
| CsLOX12 | LOC115718785 | 707 | 1-707 | 1-51 | 54-707 | linoleate 9S-lipoxygenase 1 | CsLOX2 | |
| CsLOX13 | LOC115712696 | 935 | 129-935 | 54-236 | 238-935 | lipoxygenase 6, chloroplastic | CsLOX17 | LOX |
| CsLOX14 | LOC115707105 | 931 | 125-931 | 97-236 | 238-931 | linoleate 13S-lipoxygenase 3-1, chloroplastic | CsLOX1 | |
| CsLOX15 | LOC115719612 | 928 | 114-928 | 80-227 | 229-928 | linoleate 13S-lipoxygenase 2-1, chloroplastic | CsLOX13 | LOX-L |
| CsLOX16 | LOC115719614 | 926 | 105-926 | 78-219 | 221-926 | lipoxygenase 2, chloroplastic | CsLOX14 | |
| CsLOX17 | LOC115720530 | 926 | 105-926 | 78-219 | 221-926 | lipoxygenase 2, chloroplastic | CsLOX15 | |
| CsLOX18 | LOC115719613 | 929 | 121-929 | 92-232 | 234-929 | linoleate 13S-lipoxygenase 2-1, chloroplastic | CsLOX9 | |
| CsLOX19 | LOC115719615 | 922 | 105-922 | 92-228 | 230-922 | linoleate 13S-lipoxygenase 2-1, chloroplastic | CsLOX12 | |
| CsLOX20 | LOC115719616 | 716 | 22-716 | – | 23-716 | linoleate 13S-lipoxygenase 2-1, chloroplastic | CsLOX11 | |
| CsLOX21 | LOC115719617 | 906 | 83-906 | 75-212 | 214-906 | linoleate 13S-lipoxygenase 2-1, chloroplastic | CsLOX10 | |
1Borrego et al., 2023 LOX nomenclature.
Enhanced expression of Cannabis LOX in trichomes and floral tissues
We hypothesized that LOXs which have high expression in PC producing tissues, such as trichomes, perigonal bracts and inflorescences may have important roles in PC production. For example, some 9-LOX isoforms are exclusively expressed in roots where they participate in the synthesis of defense compounds (e.g., etherolenic acid, colnelenic acid) against root pathogens (ref. Grechkin et al., 1997; ref. Sanadhya et al., 2021). CsLOX7, CsLOX15, CsLOX16 and CsLOX17 consistently showed high levels of expression in the PC-producing tissues across two medicinal cannabis chemotypes, while CsLOX15 was predominantly expressed in the trichomes (f4), indicating its potential involvement in trichome biochemistry. The recent heterologous expression of CsLOX15 in Nicotiana attenuata and N. benthamiana plants indicate that this paralog encodes a functional enzyme, although activity was assessed using a single substrate, linoleic acid (ref. Fayaz et al., 2023). Elucidating the molecular context of CsLOX15 in its native environment will be critical to disentangle its precise role within Cannabis glandular trichomes and subsequent impact on PC production.
CsLOX15 structure – conserved residues in catalytic region
To further understand the structure and function of CsLOX15 and other Cannabis LOXs, we compared the predicted structures of these Cannabis proteins with the crystal structure from the Glycine max (soybean) GmLOX1. As expected, many of the functional residues were conserved across the CsLOXs, with minor exceptions (ref. Meyer et al., 2008; ref. Offenbacher et al., 2017; ref. Ruddat et al., 2004; ref. Li et al., 2018) (Supplementary Table S2, Supplementary Figure S1). The predicted 3D structure of CsLOX15 shows the β-sheet rich N-terminal domain as well as the α-helix rich long carboxylic domain within the catalytic site, which facilitates electron transfer and the insertion of oxygen (f5, Supplementary Figure S1). The predicted 3D structure of CsLOX15, however, indicates that the side chain of W585 near the Fe ligand interacts with residue H584 (GmLOX1: H499) (f5). This may reduce space around Fe ligand by distorting the octahedral geometry, which could impact electron transfer (ref. Steczko et al., 1992; ref. Minor et al., 1996; ref. Tomchick et al., 2001; ref. Liavonchanka and Feussner, 2006; f5). We also observed that the N780 residue (GmLOX1: N694 involved in Fe binding) was positioned further away from the Fe ligand which could further weaken contact and increase flexibility (f5), Modification of N694H in GmLOX1 reduced flexibility due to stronger H bonding in the coordination sphere and resulted in reduced catalytic activity (ref. Holman et al., 1998; ref. Schenk et al., 2003; ref. Segraves et al., 2006). The application of electron paramagnetic resonance (EPR) and magnetic circular dichroism (MCD) hydrogen deuterium exchange mass spectrometry will further aid in resolving the 3D structure and catalytic center of LOXs in non-model species such as Cannabis (ref. Holman et al., 1998; ref. Hu et al., 2019; ref. Gaffney, 2020).
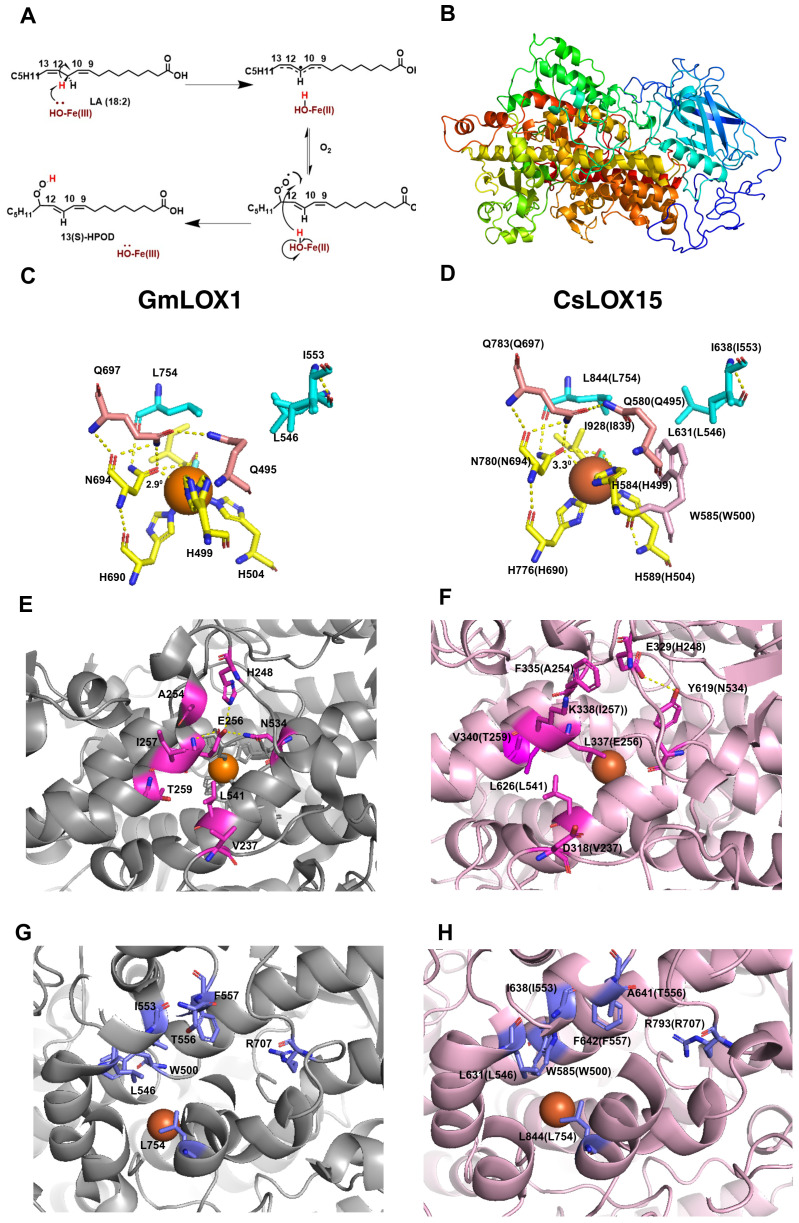
Analysis of the in silico predicted 3D structure of CsLOX15 also indicates changes in the substrate entry pocket which could potentially impact substrate specificity. Strong H bonds from residues H248-E256-N534 at the entrance of the substrate binding site of GmLOX1 have been partially lost in CsLOX15 (f5) (ref. Skrzypczak-Jankun et al., 2001). Replacement of these with hydrophobic residues leucine and threonine as seen in CsLOX15 may alter the H bond network and expand the PUFA entry point (ref. Youn et al., 2006). Replacement of V340 with the bulkier T259 residue (GmLOX1) also appears to have expanded the entry pocket (f5). Substrate orientation may also be affected in CsLOX15. For example, there was less distance between A641 (GmLOX1: T556) and R797 (GmLOX1: R707) within the substrate cavity which could potentially affect pocket volume, substrate entry and orientation (ref. Hornung et al., 1999; ref. Hughes et al., 2001; ref. Hershelman et al., 2019; f5). Understanding the impact of these changes is complicated by the fact that there are no crystal structures of plant LOXs bound with linoleic acid or α-linolenic acid (ref. Knapp and Klinman, 2003; ref. Newie et al., 2016). Using in-silico tools such as Autodock to model the interaction between fatty acid and LOX proteins would provide deeper insights into substrate-enzyme interactions (ref. Naimuzzaman et al., 2025).
Variations in the predicted tertiary structure of CsLOX15 compared to GmLOX1 suggest potential for differences in catalytic activity that might allow CsLOX15 to catalyze reactions utilizing numerous fatty acid substrates. As Cannabis glandular trichome disc cells are proposed to have non-photosynthetic chloroplasts, the role of CsLOX15 may facilitate the mobilization of storage lipids to provide carbon for producing defense compounds (ref. Livingston et al., 2022). To meet the carbon demand, trichomes reliant on the supply of photosynthates from source organs, may be partially dependent on locally stored non-structural carbohydrates to maintain carbon supply (ref. Huang et al., 2020). Hence, trichome-specific LOXs could be providing an alternate carbon source through hydrolysis of storage lipids.
The cytochrome P450 enzymatic step may represent an important branch point to study the effects of oxylipins on PC production
Both HPL and AOS are homologous cytochrome P450 enzymes that do not require molecular oxygen or the reducing power of NADPH reductase (ref. Li et al., 2008). Structural analysis of the A. thaliana AOS protein shows that one point mutation in the catalytic site converts AOS to HPL (ref. Li et al., 2008). While both enzymes compete for the same substrate (e.g. hydroperoxy fatty acids), competition can be controlled through temporal regulation, with changes in the time of expression observed in HPL- or AOS-specific LOXs in N. attenuata, or through spatial discrimination, as described previously in Z. mays and tomato (ref. Froehlich et al., 2001; ref. Allmann et al., 2010). Silencing HPL in soybean, rice, and potato impaired GLV production while increasing JAs synthesis, suggesting crosstalk between these pathways (ref. Vancanneyt et al., 2001; ref. Tong et al., 2012; ref. Wang et al., 2020). As Cannabis is predicted to contain only a single copy of HPL (CYP74B) and AOS (CYP74A) (ref. Borrego et al., 2023), silencing CsHPL or CsAOS2 would be beneficial to determine compensatory mechanisms from loss of function of either gene and subsequent impact on PC production. To elucidate sequential steps up and downstream of these CYP74 enzymes, protein-protein interactions could also be explored by chemical cross-linking coupled with mass spectrometry (MS)-cleavable tag analysis (ref. Piersimoni et al., 2021).
JAs and other oxylipins in PC production
The precise mechanism(s) by which JAs mediate PC production remains unresolved
There are several mechanisms by which JAs such as MeJA may increase PC content (T1). For example, through glandular trichome induction, increasing inflorescence compactness by reducing internode length, and/or the spacing of repeating phytomer units that host these specialized structures (ref. Spitzer-Rimon et al., 2019; ref. Welling et al., 2023; ref. Huang et al., 2024). The complexity of Cannabis floral architecture makes measuring JA responses challenging. Consequently, there have only been limited attempts to quantify trichome density on floral leaves (e.g., perigonal bracts/calyxes), and there are mixed reports of the effectiveness of MeJA to induce trichomes on the foliar fan leaves (ref. Welling et al., 2023; ref. Hahm et al., 2024; ref. Huang et al., 2024). Promisingly, a nearly two-fold increase in trichome density of calyxes was observed in 100 µM MeJA treated plants (ref. Hahm et al., 2024). However, trichome imaging was limited to five calyxes per plant, which may not be representative, and density measurements were calculated based on a 6 x 4 mm leaf surface area (ref. Hahm et al., 2024). The application of deep learning models and high spatial resolution imaging technologies could be used to accurately measure changes in floral architecture following exogenous JA exposure (ref. Huang et al., 2024; ref. Matsumoto et al., 2024).
JAs may also directly increase PC content by inducing PC gene expression and synthesis. To date, no significant changes in the expression of PC-related genes have been reported following a MeJA treatment (T1). However, spatiotemporal analysis of PC and precursor pathway genes within target tissues, such as the trichome secretory cells, following JA exposure are lacking (ref. Garrido et al., 2022; ref. Welling et al., 2023). JA related molecular responses are often associated with JA pathway genes (LOX, AOS, AOC, JAZ (JASMONATE-ZIM-DOMAIN), COI1 (CORONATINE INSENSITIVE 1) interacting with transcription factors like MYC2 and basic helix–loop–helix (bHLH)148, and a similar model is predicted for Cannabis (ref. Schuurink and Tissier, 2020; ref. Wang et al., 2020; ref. Han et al., 2022; ref. Song et al., 2022; ref. Xie et al., 2023; ref. Huang et al., 2024). The regulatory functions of JA signaling are typically initiated by the synthesis of JA-Ile, which promotes formation of the SCFCOI1–JAZ co-receptor complex (ref. Hickman et al., 2017). Degradation of JAZ repressor proteins by SCFCOI1 results in the release of JAZ-mediated transcription factors, including MYC2, which activates the expression of JA-responsive genes by targeting their promoters (ref. Hickman et al., 2017).
In Cannabis, the application of exogenous MeJA has increased the expression of JA-responsive genes like JAZ and COI1 and the key regulator of these genes, CsMYC4 (ref. Huang et al., 2024). This suggests that CsMYC4 may have a similar function to MYC orthologs in tomato and L. angustifolia, both of which influence glandular trichome size and density (ref. Xu et al., 2018; ref. Dong et al., 2022b; ref. Huang et al., 2024). Members of the WRKY gene family, which have roles in glandular trichome formation in Artemisia annua, have also been shown to be responsive to MeJA in Cannabis (ref. Xie et al., 2021; ref. Rashid et al., 2023), indicating that a much larger network of transcription factors may be contributing to JA-mediated trichome formation. To fully understand these complexes, it will be necessary to comprehensively reconstruct the gene regulatory networks that form in response to JA exposure in Cannabis.
Mechanisms by which oxylipins may be interacting with PC production
Despite significant progress towards understanding the influence of oxylipin metabolism on PC production (T1), there are multiple avenues by which these lipid molecules may be driving metabolite production (f6). The following interactions are proposed: (i) JAs could induce PC production through transcription regulatory mechanism, either indirectly through JA-induced responses, or directly by inducing the biosynthesis pathways. Similarly, HPL derived oxo acids and GLVs could interact with JAs and induce JA mediated responses or be directly involved as signal molecules inducing PCs, (ii) oxylipin-induced cleavage of fatty acids may form aldehydes that act as a direct carbon source for polyketide and PC synthesis, and (iii) oxylipins could be involved in the detoxification of reactive oxygen species (ROS) in the microenvironment of glandular trichomes (ref. Balcke et al., 2017), as LOXs consume H2O2, the biproduct of PC synthesis (ref. Taura et al., 1996; ref. Sirikantaramas et al., 2005). ROS could also be generated in response to JAs and in return JAs can modulate ROS homeostasis (ref. Denness et al., 2011; ref. Corpas et al., 2015; ref. Demiwal et al., 2024; ref. Zhao et al., 2025). The interplay between ROS and JAs may lead to complex signaling pathways that could activate specialized metabolism in Cannabis. Additionally, JA signaling is closely aligned with the regulation of other phytohormones, including ethylene and salicylic acid, all of which can affect LOX expression and PC production (ref. Yamamoto et al., 2020; ref. Li et al., 2021; ref. Jeyasri et al., 2023; ref. Rodrigues Magalhães et al., 2023).
Conclusion
Despite being a multi-use crop with huge potential as a source for food, fiber and medicine, research on Cannabis has been constrained for more than 50 years. There exists a substantial knowledge gap on molecular drivers responsible for the diverse array of chemicals synthesized by Cannabis glandular trichomes. Cannabis has an extensive number of LOXs, and several are highly expressed in trichomes and are responsive to JAs (ref. Fayaz et al., 2023). We identified the trichome-specific CsLOX15, which together with CsLOX16 and CsLOX17, forms a distinct phylogenetic clade. The use of chemical inhibitors and gene silencing approaches, such as virus-induced gene-silencing (ref. Schachtsiek et al., 2019; ref. Alter et al., 2022), targeting these LOXs and other key branch points of oxylipin biosynthesis will be essential in understanding the contribution of oxylipins to PC production. Structure-function analysis of candidate LOXs by site-directed mutagenesis may also prove useful in understanding sequence variation among trichome-specific LOXs (ref. Farmer et al., 1994; ref. Li et al., 2020).
Determining the influence of oxylipin products among different trichomes types or tissues would also be interesting avenues for future research. However, due to high reactivity and volatile nature of these compounds, challenges exist in examining these in their native form. The use of spatial omics analyses of glandular trichomes and adjacent tissues may also help to resolve the contextual molecular components of these highly specialized cell types and aid in understanding of the molecular interactions between oxylipin and PC pathways. To trace the fate of oxylipin products, it will be necessary to use isotopic tracers to label PUFA precursors. Disentangling these interactions could provide important knowledge on the partitioning of carbon resources between primary and specialized metabolism and offer new opportunities for the biotechnological enhancement of Cannabis. These innovations could ultimately facilitate the development of elite chemical phenotypes for both industrial and medicinal applications.
References
- S. Allmann, R. Halitschke, R. C. Schuurink, I. T. Baldwin. Oxylipin channelling in Nicotiana attenuata: lipoxygenase 2 supplies substrates for green leaf volatile production.. Plant Cell Environ., 2010. [DOI | PubMed]
- H. Alter, R. Peer, A. Dombrovsky, M. Flaishman, B. Spitzer-Rimon. Tobacco rattle vrus as a tool for rapid reverse-genetics screens and analysis of gene function in Cannabis sativa L.. Plants, 2022. [DOI | PubMed]
- M. Ameye, S. Allmann, J. Verwaeren, G. Smagghe, G. Haesaert, R. C. Schuurink. Green leaf volatile production by plants: a meta-analysis.. New Phytol., 2018. [DOI | PubMed]
- L. L. Anderson, A. Ametovski, J. L. Luo, D. Everett-Morgan, I. S. McGregor, S. D. Banister. Cannabichromene, related phytocannabinoids, and 5-fluoro-cannabichromene have anticonvulsant properties in a mouse model of Dravet syndrome.. ACS Chem. Neurosci., 2021. [DOI | PubMed]
- P. V. Apicella, L. B. Sands, Y. Ma, G. A. Berkowitz. Delineating genetic regulation of cannabinoid biosynthesis during female flower development in.. Cannabis sativa. Plant Direct, 2022. [DOI | PubMed]
- R. Bailey. The effect of elicitor stimulation on cannabinoid production by industrial hemp (Cannabis sativa) varieties in a hydroponic system (, 2019
- G. U. Balcke, S. Bennewitz, N. Bergau, B. Athmer, A. Henning, P. Majovsky. Multi-omics of tomato glandular trichomes reveals distinct features of central carbon metabolism supporting high productivity of specialized metabolites.. Plant Cell, 2017. [DOI | PubMed]
- M. Behr, S. Lutts, J. F. Hausman, G. Guerriero. Jasmonic acid to boost secondary growth in hemp hypocotyl.. Planta, 2018. [DOI | PubMed]
- E. Blée. Biosynthesis of phytooxylipins: the peroxygenase pathway.. Lipid/Fett, 1998. [DOI]
- E. Blée. Impact of phyto-oxylipins in plant defense.. Trends Plant Sci., 2002. [DOI | PubMed]
- J. K. Booth, M. M. S. Yuen, S. Jancsik, L. L. Madilao, J. E. Page, J. Bohlmann. Terpene synthases and terpene variation in Cannabis sativa .. Plant Physiol., 2020. [DOI | PubMed]
- E. J. Borrego, M. Robertson, J. Taylor, Z. Schultzhaus, E. M. Espinoza. Oxylipin biosynthetic gene families of Cannabis sativa .. PLoS One, 2023. [DOI | PubMed]
- F. Borrelli, I. Fasolino, B. Romano, R. Capasso, F. Maiello, D. Coppola. Beneficial effect of the non-psychotropic plant cannabinoid cannabigerol on experimental inflammatory bowel disease.. Biochem. Pharmacol., 2013. [DOI | PubMed]
- A. J. Boughton, K. Hoover, G. W. Felton. Methyl jasmonate application induces increased densities of glandular trichomes on tomato, Lycopersicon esculentum .. J. Chem. Ecol., 2005. [DOI | PubMed]
- A. R. Brash. Lipoxygenases: occurrence, functions, catalysis, and acquisition of substrate.. J. Biol. Chem., 1999. [DOI | PubMed]
- S. C. Britch, S. Babalonis, S. L. Walsh. Cannabidiol: pharmacology and therapeutic targets.. Psychopharmacol., 2021. [DOI]
- L. Burgel, J. Hartung, D. Schibano, S. Graeff-Hönninger. Impact of different phytohormones on morphology, yield and cannabinoid content of Cannabis sativa L.. Plants, 2020. [DOI | PubMed]
- C. L. R. Cabrera, S. Keir-Rudman, N. Horniman, N. Clarkson, C. Page. The anti-inflammatory effects of cannabidiol and cannabigerol alone, and in combination.. Pulm. Pharmacol. Ther., 2021. [DOI | PubMed]
- D. Caldelari, G. Wang, E. E. Farmer, X. Dong. Arabidopsis lox3 lox4 double mutants are male sterile and defective in global proliferative arrest.. Plant Mol. Biol., 2011. [DOI | PubMed]
- D. Chanda, D. Neumann, J. F. Glatz. The endocannabinoid system: overview of an emerging multi-faceted therapeutic target.. Prostaglandins Leukot. Essent. Fatty Acids, 2019. [DOI | PubMed]
- A. Chauvin, D. Caldelari, J. L. Wolfender, E. E. Farmer. Four 13-lipoxygenases contribute to rapid jasmonate synthesis in wounded Arabidopsis thaliana leaves: a role for lipoxygenase 6 in responses to long-distance wound signals.. New Phytol., 2013. [DOI | PubMed]
- G. Chen, R. Hackett, D. Walker, A. Taylor, Z. Lin, D. Grierson. Identification of a specific isoform of tomato lipoxygenase (TomloxC) involved in the generation of fatty acid-derived flavor compounds.. Plant Physiol., 2004. [DOI | PubMed]
- G. Chen, P. G. L. Klinkhamer, R. Escobar-Bravo, K. A. Leiss. Type VI glandular trichome density and their derived volatiles are differently induced by jasmonic acid in developing and fully developed tomato leaves: implications for thrips resistance.. Plant Sci., 2018. [DOI | PubMed]
- Z. Chen, X. Chen, H. Yan, W. Li, Y. Li, R. Cai. The lipoxygenase gene family in poplar: identification, classification, and expression in response to MeJA treatment.. PLoS One, 2015. [DOI | PubMed]
- S. A. Christensen, A. Huffaker, C. T. Hunter, H. T. Alborn, E. A. Schmelz. A maize death acid, 10-oxo-11-phytoenoic acid, is the predominant cyclopentenone signal present during multiple stress and developmental conditions.. Plant Signal. Behav., 2016. [DOI | PubMed]
- S. A. Christensen, A. Huffaker, F. Kaplan, J. Sims, S. Ziemann, G. Doehlemann. Maize death acids, 9-lipoxygenase-derived cyclopente(a)nones, display activity as cytotoxic phytoalexins and transcriptional mediators.. Proc. Natl. Acad. Sci. U.S.A., 2015. [DOI | PubMed]
- S. A. Christensen, A. Nemchenko, E. Borrego, I. Murray, I. S. Sobhy, L. Bosak. The maize lipoxygenase, ZmLOX10, mediates green leaf volatile, jasmonate and herbivore-induced plant volatile production for defense against insect attack.. Plant J., 2013. [DOI | PubMed]
- F. J. Corpas, D. K. Gupta, J. M. Palma. Production sites of reactive oxygen species (ROS) in organelles from plant cells,” in. Reactive Oxygen Species and Oxidative Damage in Plants Under Stress. Eds., 2015. [DOI]
- C. Cropano, D.Á. Carrera, G. M. George, L. Katsir, M. M. Vogt, M. E. Ruckle. Quantitative trait loci (QTLs) associated with a high-varin trait in Cannabis. WO2022180532A1. Available online at:, 2022
- A. L. de Brito Siqueira, P. V. Cremasco, J. O. Bahú, A. P. da Silva, L. R. M. de Andrade, P. G. González. Phytocannabinoids: Pharmacological effects, biomedical applications, and worldwide prospection.. J. Tradit. Complement. Med., 2023. [DOI]
- P. Demiwal, S. U. Nabi, J. I. Mir, M. K. Verma, S. R. Yadav, P. Roy. Methyl jasmonate improves resistance in scab-susceptible Red Delicious apple by altering ROS homeostasis and enhancing phenylpropanoid biosynthesis.. Plant Physiol. Biochem., 2024. [DOI | PubMed]
- L. Denness, J. F. McKenna, C. Segonzac, A. Wormit, P. Madhou, M. Bennett. Cell wall damage-induced lignin biosynthesis is regulated by a reactive oxygen species-and jasmonic acid-dependent process in Arabidopsis.. Plant Physiol., 2011. [DOI | PubMed]
- O. Devinsky, M. R. Cilio, H. Cross, J. Fernandez-Ruiz, J. French, C. Hill. Cannabidiol: pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders.. Epilepsia, 2014. [DOI | PubMed]
- Y. Dong, J. Li, W. Zhang, H. Bai, H. Li, L. Shi. Exogenous application of methyl jasmonate affects the emissions of volatile compounds in lavender (Lavandula angustifolia).. Plant Physiol. Biochem., 2022. [DOI | PubMed]
- Y. Dong, W. Zhang, J. Li, D. Wang, H. Bai, H. Li. The transcription factor LaMYC4 from lavender regulates volatile Terpenoid biosynthesis.. BMC Plant Biol., 2022. [DOI | PubMed]
- C. A. Dowling, J. Shi, J. A. Toth, M. A. Quade, L. B. Smart, P. F. McCabe. A FLOWERING LOCUS T ortholog is associated with photoperiod-insensitive flowering in hemp (Cannabis sativa L.).. Plant J., 2024. [DOI | PubMed]
- M. Egmond, G. A. Veldink, J. F. G. Vliegenthart, J. Boldingh. C-11 H-abstraction from linoieic acid, the rate-limiting step in lipoxygenase catalysis.. Biophys. Res. Commun., 1973. [DOI]
- M. Fayaz, M. Kundan, U. Gani, P. Sharma, M. A. Wajid, K. Katoch. Identification of Lipoxygenase gene repertoire of Cannabis sativa and functional characterization of CsLOX13 gene.. Plant Sci., 2023. [DOI | PubMed]
- E. E. Farmer, D. Caldelari, G. Pearce, M. Walker-Simmons, C. A. Ryan. Diethyldithiocarbamic acid inhibits the octadecanoid signaling pathway for the wound induction of proteinase inhibitors in tomato leaves.. Plant Physiol., 1994. [DOI]
- I. J. Flores-Sanchez, J. Pec, J. Fei, Y. H. Choi, J. Dusek, R. Verpoorte. Elicitation studies in cell suspension cultures of Cannabis sativa L.. J. Biotechnol., 2009. [DOI | PubMed]
- A. M. Fortes, M. J. Coronado, P. S. Testillano, M. D. C. Risueño, M. S. Pais. Expression of lipoxygenase during organogenic nodule formation from hop internodes.. J. Histochem. Cytochem., 2004. [DOI | PubMed]
- S. Frassinetti, E. Moccia, L. Caltavuturo, M. Gabriele, V. Longo, L. Bellani. Nutraceutical potential of hemp (Cannabis sativa L.) seeds and sprouts.. Food Chem., 2018. [DOI | PubMed]
- J. E. Froehlich, A. Itoh, G. A. Howe. Tomato allene oxide synthase and fatty acid hydroperoxide lyase, two cytochrome P450s involved in oxylipin metabolism, are targeted to different membranes of chloroplast envelope.. Plant Physiol., 2001. [DOI | PubMed]
- B. J. Gaffney. EPR spectroscopic studies of lipoxygenases.. Chem. Asian J., 2020. [DOI | PubMed]
- S. J. Gagne, J. M. Stout, E. Liu, Z. Boubakir, S. M. Clark, J. E. Page. Identification of olivetolic acid cyclase from Cannabis sativa reveals a unique catalytic route to plant polyketides.. Proc. Natl. Acad. Sci. U.S.A., 2012. [DOI | PubMed]
- J. Garrido, S. Rico, C. Corral, C. Sanchez, N. Vidal, J. J. Martinez-Quesada. Exogenous application of stress-related signaling molecules affect growth and cannabinoid accumulation in medical cannabis (Cannabis sativa L.).. Front. Plant Sci., 2022. [DOI]
- A. Ghasemi Pirbalouti, S. E. Sajjadi, K. Parang. A review (research and patents) on jasmonic acid and its derivatives.. Arch. Pharm. (Weinheim)., 2014. [DOI | PubMed]
- M. Ghorbel, F. Brini, A. Sharma, M. Landi. Role of jasmonic acid in plants: the molecular point of view.. Plant Cell Rep., 2021. [DOI | PubMed]
- C. R. Goldsmith, R. T. Jonas, T. D. P. Stack. C– H bond activation by a ferric methoxide complex: modeling the rate-determining step in the mechanism of lipoxygenase.. J. Am. Chem. Soc, 2002. [DOI | PubMed]
- S. S. Gorina, L. S. Mukhtarova, T. M. Iljina, Y. Y. Toporkova, A. N. Grechkin. Detection of divinyl ether synthase CYP74H2 biosynthesizing (11Z)-etheroleic and (1′Z)-colnelenic acids in asparagus (Asparagus officinalis L.).. Phytochemistry, 2022. [DOI | PubMed]
- A. N. Grechkin, A. V. Ilyasov, M. Hamberg. On the mechanism of biosynthesis of divinyl ether oxylipins by enzyme from garlic bulbs.. Eur. J. Biochem., 1997. [DOI | PubMed]
- S. Hahm, Y. Lee, K. Lee, J. Park. Optimization of cannabinoid production in hemp through methyl jasmonate application in a vertical farming system.. Horticulturae, 2024. [DOI]
- M. Hamberg. An epoxy alcohol synthase pathway in higher plants: biosynthesis of antifungal trihydroxy oxylipins in leaves of potato.. Lipids, 1999. [DOI | PubMed]
- M. Hamberg. Isolation and structures of two divinyl ether fatty acids from Clematis vitalba .. Lipids, 2004. [DOI | PubMed]
- C. T. Hammond, P. G. Mahlberg. Morphology of glandular hairs of Cannabis sativa from scanning electron microscopy.. Am. J. Bot., 1973. [DOI]
- G. Han, Y. Li, Z. Yang, C. Wang, Y. Zhang, B. Wang. Molecular mechanisms of plant trichome development.. Front. Plant Sci., 2022. [DOI]
- A. Hazekamp, K. Tejkalová, S. Papadimitriou. Cannabis: from cultivar to chemovar II—a metabolomics approach to cannabis classification.. Cannabis Cannabinoid Res., 2016. [DOI]
- Y. He, E. J. Borrego, Z. Gorman, P. C. Huang, M. V. Kolomiets. Relative contribution of LOX10, green leaf volatiles and JA to wound-induced local and systemic oxylipin and hormone signature in Zea mays (maize).. Phytochemistry, 2020. [DOI | PubMed]
- D. Hershelman, K. M. Kahler, M. J. Price, I. Lu, Y. Fu, P. A. Plumeri. Oxygenation reactions catalyzed by the F557V mutant of soybean lipoxygenase-1: evidence for two orientations of substrate binding.. Arch. Biochem. Biophys., 2019. [DOI | PubMed]
- M. Hesami, M. Pepe, A. Baiton, A. M. P. Jones. Current status and future prospects in cannabinoid production through in vitro culture and synthetic biology.. Biotechnol. Adv., 2022. [DOI]
- M. Hesami, M. Pepe, A. Baiton, S. A. Salami, A. M. P. Jones. New insight into ornamental applications of cannabis: Perspectives and challenges.. Plants, 2022. [DOI | PubMed]
- R. Hickman, M. C. Van Verk, A. J. Van Dijken, M. P. Mendes, I. A. Vroegop-Vos, L. Caarls. Architecture and dynamics of the jasmonic acid gene regulatory network.. Plant Cell., 2017. [DOI | PubMed]
- T. R. Holman, J. Zhou, E. I. Solomon. Spectroscopic and functional characterization of a ligand coordination mutant of soybean lipoxygenase-1: first coordination sphere analogue of human 15-lipoxygenase.. J. Am. Chem. Soc., 1998. [DOI]
- E. Hornung, M. Walther, H. Kühn, I. Feussner. Conversion of cucumber linoleate 13-lipoxygenase to a 9-lipoxygenating species by site-directed mutagenesis.. Proc. Natl. Acad. Sci. U.S.A., 1999. [DOI | PubMed]
- W. Horper, F. J. Marner. Biosynthesis of primin and miconidin and its derivatives.. Phytochemistry, 1996. [DOI]
- G. A. Howe, G. I. Lee, A. Itoh, L. Li, A. E. DeRocher. Cytochrome P450-dependent metabolism of oxylipins in tomato. Cloning and expression of allene oxide synthase and fatty acid hydroperoxide lyase.. Plant Physiol., 2000. [DOI | PubMed]
- S. Hu, A. R. Offenbacher, E. M. Thompson, C. L. Gee, J. Wilcoxen, C. A. Carr. Biophysical characterization of a disabled double mutant of soybean lipoxygenase: the “undoing” of precise substrate positioning relative to metal cofactor and an identified dynamical network.. J. Am. Chem. Soc., 2019. [DOI | PubMed]
- X. Huang, W. Chen, Y. Zhao, J. Chen, Y. Ouyang, M. Li. Deep learning-based quantification and transcriptomic profiling reveal a methyl jasmonate-mediated glandular trichome formation pathway in Cannabis sativa .. Plant J., 2024. [DOI]
- J. Huang, A. Rücker, A. Schmidt, G. Gleixner, J. Gershenzon, S. Trumbore. Production of constitutive and induced secondary metabolites is coordinated with growth and storage in Norway spruce saplings.. Tree Physiol., 2020. [DOI | PubMed]
- R. Hughes, D. M. Lawson, A. R. Hornostaj, S. A. Fairhurst, R. Casey. Mutagenesis and modelling of linoleate-binding to pea seed lipoxygenase.. Eur. J. Biochem., 2001. [DOI | PubMed]
- B. Hurgobin, M. Tamiru-Oli, M. T. Welling, M. S. Doblin, A. Bacic, J. Whelan. Recent advances in Cannabis sativa genomics research.. New Phytol., 2021. [DOI | PubMed]
- R. Jeyasri, P. Muthuramalingam, K. Karthick, H. Shin, S. H. Choi, M. Ramesh. Methyl jasmonate and salicylic acid as powerful elicitors for enhancing the production of secondary metabolites in medicinal plants: an updated review.. Plant Cell Tissue Organ Cult., 2023. [DOI | PubMed]
- D. Jin, P. Henry, J. Shan, J. Chen. Identification of chemotypic markers in three chemotype categories of cannabis using secondary metabolites profiled in inflorescences, leaves, stem bark, and roots.. Front. Plant Sci., 2021. [DOI]
- R. Jost, O. Berkowitz, A. Pegg, B. Hurgobin, M. Tamiru-Oli, M. T. Welling. Sink strength, nutrient allocation, cannabinoid yield, and associated transcript profiles vary in two drug-type Cannabis chemovars.. J. Exp. Bot., 2025. [DOI | PubMed]
- L. A. Kelley, S. Mezulis, C. M. Yates, M. N. Wass, M. J. E. Sternberg. The Phyre2 web portal for protein modeling, prediction and analysis.. Nat. Protoc., 2015. [DOI | PubMed]
- F. Kianersi, D. A. Azarm, A. Pour-Aboughadareh, P. Poczai. Change in secondary metabolites and expression pattern of key rosmarinic acid related genes in Iranian lemon balm (Melissa officinalis L.) ecotypes using methyl jasmonate treatments.. Molecules, 2022. [DOI | PubMed]
- M. J. Knapp, J. P. Klinman. Kinetic studies of oxygen reactivity in soybean lipoxygenase-1.. Biochemistry, 2003. [DOI | PubMed]
- M. V. Kolomiets, D. J. Hannapel, H. Chen, M. Tymeson, R. J. Gladon. Lipoxygenase is involved in the control of potato tuber development.. Plant Cell, 2001. [DOI | PubMed]
- I. Kovalchuk, M. Pellino, P. Rigault, R. Van Velzen, J. Ebersbach, J. R. Ashnest. The genomics of Cannabis and its close relatives.. Annu. Rev. Plant Biol., 2020. [DOI | PubMed]
- A. B. Kroumova, Z. Xie, G. J. Wagner. A pathway for the biosynthesis of straight and branched, odd-and even-length, medium-chain fatty acids in plants.. Proc. Natl. Acad. Sci. U.S.A., 1994. [DOI | PubMed]
- I. Letunic, P. Bork. Interactive Tree Of Life (iTOL): an online tool for phylogenetic tree display and annotation.. Bioinformatics, 2007. [DOI | PubMed]
- L. Li, Z. Chang, Z. Pan, Z. Q. Fu, X. Wang. Modes of heme binding and substrate access for cytochrome P450 CYP74A revealed by crystal structures of allene oxide synthase.. Proc. Natl. Acad. Sci. U.S.A., 2008. [DOI | PubMed]
- P. Li, A. V. Soudackov, S. Hammes-Schiffer. Impact of mutations on the binding pocket of soybean lipoxygenase: implications for proton-coupled electron transfer.. J. Phys. Chem. Lett., 2018. [DOI | PubMed]
- J. Li, X. Wang, R. Jiang, B. Dong, S. Fang, Q. Li. Phytohormone-based regulation of trichome development.. Front. Plant Sci., 2021. [DOI]
- Y. Li, L. Qiu, Q. Zhang, X. Zhuansun, H. Li, X. Chen. Exogenous sodium diethyldithiocarbamate, a Jasmonic acid biosynthesis inhibitor, induced resistance to powdery mildew in wheat.. Plant Direct., 2020. [DOI | PubMed]
- A. Liavonchanka, I. Feussner. Lipoxygenases: occurrence, functions and catalysis.. J. Plant Physiol., 2006. [DOI | PubMed]
- A. H. Lichtman, E. A. Lux, R. McQuade, S. Rossetti, R. Sanchez, W. Sun. Results of a double-blind, randomized, placebo-controlled study of nabiximols oromucosal spray as an adjunctive therapy in advanced cancer patients with chronic uncontrolled pain.. J. Pain Symptom Manage., 2018. [DOI | PubMed]
- S. Liu, B. Han. Differential expression pattern of an acidic 9/13-lipoxygenase in flower opening and senescence and in leaf response to phloem feeders in the tea plant.. BMC Plant Biol., 2010. [DOI | PubMed]
- J. Liu, Y. Zhou, J. Li, F. Wang, Y. Yang. Comprehensive genomic characterization and expression analysis of the lipoxygenase gene family in watermelon under hormonal treatments.. Agriculture, 2020. [DOI]
- S. J. Livingston, E. J. Bae, F. Unda, M. G. Hahn, S. D. Mansfield, J. E. Page. Cannabis glandular trichome cell walls undergo remodeling to store specialized metabolites.. Plant Cell Physiol., 2021. [DOI | PubMed]
- S. J. Livingston, T. D. Quilichini, J. K. Booth, D. C. Wong, K. H. Rensing, J. Laflamme-Yonkman. Cannabis glandular trichomes alter morphology and metabolite content during flower maturation.. Plant J., 2020. [DOI | PubMed]
- S. J. Livingston, K. H. Rensing, J. E. Page, A. L. Samuels. A polarized supercell produces specialized metabolites in cannabis trichomes.. Curr. Biol., 2022. [DOI | PubMed]
- X. Luo, M. A. Reiter, L. d’Espaux, J. Wong, C. M. Denby, A. Lechner. Complete biosynthesis of cannabinoids and their unnatural analogues in yeast.. Nature, 2019. [DOI | PubMed]
- M. D. Marks, L. Tian, J. P. Wenger, S. N. Omburo, W. Soto-Fuentes, J. He. Identification of candidate genes affecting Δ9-tetrahydrocannabinol biosynthesis in Cannabis sativa .. J. Exp. Bot., 2009. [DOI | PubMed]
- S. Matsumoto, Y. Utsumi, T. Kozuka, M. Iwamura, T. Nakai, D. Yamauchi. CT image-based 3D inflorescence estimation of Chrysanthemum seticuspe .. Front. Plant Sci., 2024. [DOI]
- R. C. McGarry, Y.-T. Lin, H. Kaur, H. Higgs, O. Arias-Gaguancela, B. G. Ayre. Disrupted oxylipin biosynthesis mitigates pathogen infections and pest infestations in cotton (Gossypium hirsutum).. J. Exp. Bot., 2024. [DOI | PubMed]
- R. Mechoulam, L. A. Parker. The endocannabinoid system and the brain.. Annu. Rev. Psychol., 2013. [DOI | PubMed]
- M. P. Meyer, D. R. Tomchick, J. P. Klinman. Enzyme structure and dynamics affect hydrogen tunneling: The impact of a remote side chain (I553) in soybean lipoxygenase-1.. Proc. Natl. Acad. Sci. U.S.A., 2008. [DOI | PubMed]
- W. Minor, J. Steczko, B. Stec, Z. Otwinowski, J. T. Bolin, R. Walter. Crystal structure of soybean lipoxygenase L-1 at 1.4 Å resolution.. Biochemistry, 1996. [DOI | PubMed]
- S. Mochizuki, K. Sugimoto, T. Koeduka, K. Matsui. Arabidopsis lipoxygenase 2 is essential for formation of green leaf volatiles and five-carbon volatiles.. FEBS Lett., 2016. [DOI | PubMed]
- M. Naimuzzaman, M. M. Hasan, A. Kumer, A. Y. Hossin, M. Harun-Ur-Rashid, S. K. Roy. Computational and In silico study of novel fungicides against combating root rot, gray mold, fusarium wilt, and cereal rust.. PloS One, 2025. [DOI | PubMed]
- A. Nakashima, S. H. von Reuss, H. Tasaka, M. Nomura, S. Mochizuki, Y. Iijima. Traumatin-and dinortraumatin-containing galactolipids in Arabidopsis: their formation in tissue-disrupted leaves as counterparts of green leaf volatiles.. J. Biol. Chem., 2013. [DOI | PubMed]
- J. Newie, A. Andreou, P. Neumann, O. Einsle, I. Feussner, R. Ficner. Crystal structure of a lipoxygenase from Cyanothece sp. may reveal novel features for substrate acquisition.. J. Lipid Res., 2016. [PubMed]
- A. R. Offenbacher, S. Hu, E. M. Poss, C. A. Carr, A. D. Scouras, D. M. Prigozhin. Hydrogen–deuterium exchange of lipoxygenase uncovers a relationship between distal, solvent exposed protein motions and the thermal activation barrier for catalytic proton-coupled electron tunneling.. ACS Cent. Sci., 2017. [DOI | PubMed]
- M. O. Omare, J. K. Kibet, J. K. Cherutoi, F. O. Kengara. Current trends in the use of Cannabis sativa: beyond recreational and medicinal applications.. Open Access Libr. J., 2021. [DOI]
- J. M. J. Oultram, J. L. Pegler, A. L. Eamens, R. Gordon, D. J. Korbie, C. P. L. Grof. Exogenously applied gibberellic acid alters cannabinoid profile in Cannabis sativa L.. Agronomy, 2024. [DOI]
- S. Pattanaik, B. Patra, S. K. Singh, L. Yuan. An overview of the gene regulatory network controlling trichome development in the model plant, Arabidopsis .. Front. Plant Sci., 2014. [DOI]
- L. Piersimoni, P. L. Kastritis, C. Arlt, A. Sinz. Cross-linking mass spectrometry for investigating protein conformations and protein–protein interactions─ a method for all seasons.. Chem. Rev., 2021. [DOI | PubMed]
- A. Rashid, M. Khajuria, V. Ali, S. Faiz, S. Jamwal, D. Vyas. Genome-wide identification and expression profiling of WRKY family suggest their potential role in cannabinoid regulation in Cannabis sativa L.. Ind. Crops Prod., 2023. [DOI]
- A. Raza, S. Charagh, Z. Zahid, M. S. Mubarik, R. Javed, M. H. Siddiqui. Jasmonic acid: a key frontier in conferring abiotic stress tolerance in plants.. Plant Cell Rep., 2021. [DOI | PubMed]
- H. C. Rodrigues Magalhães, E. G. Alves Filho, S. L. Rivero Meza, A. Oliveira, D. S. Garruti, E. Purgatto. Effect of methyl jasmonate on the biosynthesis of volatile compounds associated with the ripening of grape tomato fruits.. J. Agric. Food Chem., 2023. [DOI | PubMed]
- V. C. Ruddat, R. Mogul, I. Chorny, C. Chen, N. Perrin, S. Whitman. Tryptophan 500 and arginine 707 define product and substrate active site binding in soybean lipoxygenase-1.. Biochemistry, 2004. [DOI | PubMed]
- S. Salamone, L. Waltl, A. Pompignan, G. Grassi, G. Chianese, A. Koeberle. Phytochemical characterization of Cannabis sativa L. chemotype V reveals three new dihydrophenanthrenoids that favorably reprogram lipid mediator biosynthesis in macrophages.. Plants, 2022. [DOI | PubMed]
- P. Sanadhya, A. Kumar, P. Bucki, N. Fitoussi, M. Carmeli-Weissberg, M. Borenstein. Tomato divinyl ether-biosynthesis pathway is implicated in modulating of root-knot nematode Meloidogyne javanica’s parasitic ability.. Front. Plant Sci., 2021. [DOI]
- L. B. Sands, S. R. Haiden, Y. Ma, G. A. Berkowitz. Hormonal control of promoter activities of Cannabis sativa prenyltransferase 1 and 4 and salicylic acid mediated regulation of cannabinoid biosynthesis.. Sci. Rep., 2023. [DOI | PubMed]
- A. Scala, S. Allmann, R. Mirabella, M. A. Haring, R. C. Schuurink. Green leaf volatiles: a plant’s multifunctional weapon against herbivores and pathogens.. Int. J. Mol. Sci., 2013. [DOI | PubMed]
- J. Schachtsiek, T. Hussain, K. Azzouhri, O. Kayser, F. Stehle. Virus-induced gene silencing (VIGS) in Cannabis sativa L.. Plant Methods, 2019. [DOI | PubMed]
- G. Schenk, M. L. Neidig, J. Zhou, T. R. Holman, E. I. Solomon. Spectroscopic characterization of soybean lipoxygenase-1 mutants: the role of second coordination sphere residues in the regulation of enzyme activity.. Biochemistry, 2003. [DOI | PubMed]
- W. S. Schlotzhauer, S. D. Pair, R. J. Horvat. Volatile constituents from the flowers of Japanese honeysuckle (Lonicera japonica).. J. Agric. Food Chem., 1996. [DOI]
- R. Schuurink, A. Tissier. Glandular trichomes: micro-organs with model status?. New Phytol., 2020. [DOI | PubMed]
- E. N. Segraves, M. Chruszcz, M. L. Neidig, V. Ruddat, J. Zhou, A. T. Wecksler. Kinetic, spectroscopic, and structural investigations of the soybean lipoxygenase-1 first-coordination sphere mutant, Asn694Gly.. Biochemistry, 2006. [DOI | PubMed]
- S. S. M. Shakil, M. Gowan, K. Hughes, M. N. K. Azam, M. N. Ahmed. A narrative review of the ethnomedicinal usage of Cannabis sativa Linnaeus as traditional phytomedicine by folk medicine practitioners of Bangladesh.. J. Cannabis Res., 2021. [DOI | PubMed]
- J. Shen, D. Tieman, J. B. Jones, M. G. Taylor, E. Schmelz, A. Huffaker. A 13-lipoxygenase, TomloxC, is essential for synthesis of C5 flavour volatiles in tomato.. J. Exp. Bot., 2014. [DOI | PubMed]
- F. Sievers, D. G. Higgins. The Clustal Omega Multiple Alignment Package.” in Multiple Sequence Alignment, ed.. Methods Mol. Biol., 2021. [DOI]
- S. Sirikantaramas, S. Morimoto, Y. Shoyama, Y. Ishikawa, Y. Wada, Y. Shoyama. The gene controlling marijuana psychoactivity: molecular cloning and heterologous expression of Δ1-tetrahydrocannabinolic acid synthase from Cannabis sativa L.. J. Biol. Chem., 2004. [DOI | PubMed]
- S. Sirikantaramas, F. Taura. Cannabinoids: biosynthesis and biotechnological applications,” in. Cannabis sativa L. – Botany and Biotechnology . Eds., 2017. [DOI]
- S. Sirikantaramas, F. Taura, Y. Tanaka, Y. Ishikawa, S. Morimoto, Y. Shoyama. Tetrahydrocannabinolic acid synthase, the enzyme controlling marijuana psychoactivity, is secreted into the storage cavity of the glandular trichomes.. Plant Cell Physiol., 2005. [DOI | PubMed]
- E. Skrzypczak-Jankun, R. A. Bross, R. T. Carroll, W. R. Dunham, M. O. Funk. Three-dimensional structure of a purple lipoxygenase.. J. Am. Chem. Soc, 2001. [DOI | PubMed]
- E. Small. Evolution and classification of Cannabis sativa (marijuana, hemp) in relation to human utilization.. Bot. Rev., 2015. [DOI]
- W. C. Song, A. R. Brash. Purification of an allene oxide synthase and identification of the enzyme as a cytochrome P-450.. Science, 1991. [DOI | PubMed]
- C. Song, Y. Cao, J. Dai, G. Li, M. A. Manzoor, C. Chen. The multifaceted roles of MYC2 in plants: toward transcriptional reprogramming and stress tolerance by jasmonate signaling.. Front.Plant Sci., 2022. [DOI]
- H. Song, P. Wang, C. Li, S. Han, J. Lopez-Baltazar, X. Zhang. Identification of lipoxygenase (LOX) genes from legumes and their responses in wild type and cultivated peanut upon Aspergillus flavus infection.. Sci. Rep., 2016. [DOI | PubMed]
- B. Spitzer-Rimon, S. Duchin, N. Bernstein, R. Kamenetsky. Architecture and florogenesis in female Cannabis sativa plants.. Front. Plant Sci., 2019. [DOI]
- J. Steczko, G. P. Donoho, J. C. Clemens, J. E. Dixon, B. Axelrod. Conserved histidine residues in soybean lipoxygenase: functional consequences of their replacement.. Biochemistry, 1992. [DOI | PubMed]
- L. Steel, M. Welling, N. Ristevski, K. Johnson, A. Gendall. Comparative genomics of flowering behavior in Cannabis sativa .. Front. Plant Sci., 2023. [DOI | PubMed]
- J. M. Stout, Z. Boubakir, S. J. Ambrose, R. W. Purves, J. E. Page. The hexanoyl-CoA precursor for cannabinoid biosynthesis is formed by an acyl-activating enzyme in Cannabis sativa trichomes.. Plant J., 2012. [DOI | PubMed]
- M. Tagen, L. E. Klumpers. Review of delta-8-tetrahydrocannabinol (Δ8-THC): Comparative pharmacology with Δ9-THC.. Br. J. Pharmacol., 2022. [DOI | PubMed]
- C. A. Tanney, R. Backer, A. Geitmann, D. L. Smith. Cannabis glandular trichomes: A cellular metabolite factory.. Front. Plant Sci., 2021. [DOI]
- F. Taura, S. Morimoto, Y. Shoyama. Purification and characterization of cannabidiolic-acid synthase from Cannabis sativa L.: Biochemical analysis of a novel enzyme that catalyzes the oxidocyclization of cannabigerolic acid to cannabidiolic acid.. J. Biol. Chem., 1996. [DOI | PubMed]
- E. A. Thiele, E. D. Marsh, J. A. French, M. Mazurkiewicz-Beldzinska, S. R. Benbadis, C. Joshi. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): a randomised, double-blind, placebo-controlled phase 3 trial.. Lancet, 2018. [DOI | PubMed]
- D. R. Tomchick, P. Phan, M. Cymborowski, W. Minor, T. R. Holman. Structural and functional characterization of second-coordination sphere mutants of soybean lipoxygenase-1.. Biochemistry, 2001. [DOI | PubMed]
- X. Tong, J. Qi, X. Zhu, B. Mao, L. Zeng, B. Wang. The rice hydroperoxide lyase OsHPL3 functions in defense responses by modulating the oxylipin pathway.. Plant J., 2012. [DOI | PubMed]
- Y. Y. Toporkova, E. O. Smirnova, S. S. Gorina. Epoxyalcohol synthase branch of lipoxygenase cascade.. Curr. Issues Mol. Biol., 2024. [DOI | PubMed]
- J. A. Toth, G. M. Stack, C. H. Carlson, L. B. Smart. Identification and mapping of major-effect flowering time loci Autoflower1 and Early1 in Cannabis sativa L.. Front. Plant Sci., 2022. [DOI]
- I. Urits, M. Borchart, M. Hasegawa, J. Kochanski, V. Orhurhu, O. Viswanath. An update of current cannabis-based pharmaceuticals in pain medicine.. Pain Ther., 2019. [DOI | PubMed]
- G. Vancanneyt, C. Sanz, T. Farmaki, M. Paneque, F. Ortego, P. Castañera. Hydroperoxide lyase depletion in transgenic potato plants leads to an increase in aphid performance.. Proc. Natl. Acad. Sci. U.S.A., 2001. [DOI | PubMed]
- J. Vogt, D. Schiller, D. Ulrich, W. Schwab, F. Dunemann. Identification of lipoxygenase (LOX) genes putatively involved in fruit flavour formation in apple (Malus domestica).. Tree Genet. Genomes, 2013. [DOI]
- Y. Wang, M. Liu, D. Ge, J. Akhter-Bhat, Y. Li, J. Kong. Hydroperoxide lyase modulates defense response and confers lesion-mimic leaf phenotype in soybean (Glycine max (L.) Merr.).. Plant J., 2020. [DOI | PubMed]
- J. Wang, L. Song, X. Gong, J. Xu, M. Li. Functions of jasmonic acid in plant regulation and response to abiotic stress.. Int. J. Mol. Sci., 2020. [DOI | PubMed]
- Y. Wang, D. Wang, S. Li, Y. Zhai, Y. Zhao, F. Zhong. Methyl jasmonate and salicylic acid enhance the total flavonoid, phenolics, and cannabidiol contents of Cannabis sativa L. adventitious roots.. Plant Cell Tissue Organ Cult., 2024. [DOI]
- X. Wang, Y. Zeng, L. Li, R. Fan, Y. Yan, L. Guo. Unraveling jasmonate-mediated plant immunity against green peach aphid (Myzus persicae) in peach: an integrated transcriptomic and RNA sequencing approach.. Plant Growth Regul., 2024. [DOI]
- C. Wasternack, I. Feussner. The oxylipin pathways: biochemistry and function.. Annu. Rev. Plant Biol., 2018. [DOI | PubMed]
- C. Wasternack, B. Hause. Jasmonates: biosynthesis, perception, signal transduction and action in plant stress response, growth and development. An update to the 2007 review in Annals of Botany.. Ann. Bot., 2013. [DOI | PubMed]
- H. Weber, A. Chételat, D. Caldelari, E. E. Farmer. Divinyl ether fatty acid synthesis in late blight–diseased potato leaves.. Plant Cell, 1999. [DOI | PubMed]
- M. T. Welling, M. A. Deseo, M. O’Brien, J. Clifton, A. Bacic, M. S. Doblin. Metabolomic analysis of methyl jasmonate treatment on phytocannabinoid production in Cannabis sativa .. Front. Plant Sci., 2023. [DOI]
- M. T. Welling, L. Liu, T. Kretzschmar, R. Mauleon, O. Ansari, G. J. King. An extreme-phenotype genome-wide association study identifies candidate cannabinoid pathway genes in Cannabis .. Sci. Rep., 2020. [DOI | PubMed]
- M. T. Welling, T. Shapter, T. J. Rose, L. Liu, R. Stanger, G. J. King. A belated green revolution for Cannabis: virtual genetic resources to fast-track cultivar development.. Front. Plant Sci., 2016. [DOI]
- Y. Wu, H. X. Trejo, G. Chen, S. Li. Phytoremediation of contaminants of emerging concern from soil with industrial hemp (Cannabis sativa L.): a review.. Environ. Dev. Sustain., 2021. [DOI]
- Z. Xie, Y. Mi, L. Kong, M. Gao, S. Chen, W. Chen. Cannabis sativa: origin and history, glandular trichome development, and cannabinoid biosynthesis.. Horticult. Res., 2023. [DOI]
- L. Xie, T. Yan, L. Li, M. Chen, Y. Ma, X. Hao. The WRKY transcription factor AaGSW2 promotes glandular trichome initiation in Artemisia annua .. J. Exp. Bot., 2021. [DOI | PubMed]
- J. Xu, Z. O. van Herwijnen, D. B. Dräger, C. Sui, M. A. Haring, R. C. Schuurink. SlMYC1 regulates type VI glandular trichome formation and terpene biosynthesis in tomato glandular cells.. Plant Cell, 2018. [DOI | PubMed]
- F. Yamamoto, F. Iwanaga, A. Al-Busaidi, N. Yamanaka. Roles of ethylene, jasmonic acid, and salicylic acid and their interactions in frankincense resin production in Boswellia sacra Flueck. trees.. Sci. Rep., 2020. [DOI | PubMed]
- X. Yan, L. Cui, X. Liu, Y. Cui, Z. Wang, H. Zhang. NbJAZ3 is required for jasmonate-meditated glandular trichome development in Nicotiana benthamiana .. Physiol. Plant., 2022. [DOI | PubMed]
- M. Yokoyama, T. Kurusu, H. Ohno, O. Ifuku, R. Harada, Y. Tada. Oxylipin KODA enhances the early growth of rice (Oryza sativa L.) under low-temperature stress at night to simulate a natural temperature condition.. Plant Biotechnol., 2025. [DOI]
- B. Youn, G. E. Sellhorn, R. J. Mirchel, B. J. Gaffney, H. D. Grimes, C. Kang. Crystal structures of vegetative soybean lipoxygenase VLX-B and VLX-D, and comparisons with seed isoforms LOX-1 and LOX-3.. Proteins, 2006. [DOI | PubMed]
- Y. Zhao, Y. Wu, X. Zhang, X. Zhu, Y. Hou, J. Chen. Methyl jasmonate attenuates chilling injury of prune fruit by maintaining ROS homeostasis and regulating GABA metabolism and energy status.. Postharvest Biol. Technol., 2025. [DOI]
- M. Zoeller, N. Stingl, M. Krischke, A. Fekete, F. Waller, S. Berger. Lipid profiling of the Arabidopsis hypersensitive response reveals specific lipid peroxidation and fragmentation processes: biogenesis of pimelic and azelaic acid.. Plant Physiol., 2012. [DOI | PubMed]
