Relative Risk of All‐Cause Mortality Associated With Cannabis Use: A Systematic Review and Meta‐Analysis of Cohort Studies
Abstract
Background and Aims:
Cannabis use has high prevalence and health burden. Although the effects of cannabis use have been studied in the literature, no systematic review and meta‐analysis has measured its association with all‐cause mortality. The aim of this systematic review and meta‐analysis was to systematically synthesize the evidence on association between cannabis use and all‐cause mortality.
Methods:
Following the preregistered protocol (PROSPERO: CRD42023396915), we searched in Scopus, PubMed, Web of Science and ProQuest databases until end of October 2023. We included cohort studies comparing individuals using versus not using cannabis and measuring the association with all‐cause mortality. A random‐effect meta‐analysis was conducted calculating the risk ratio (RR) and 95% confidence interval (CI). Heterogeneity and publication bias were measured. Sensitivity and meta‐regression analyses were conducted. The Newcastle Ottawa Scale (NOS) was used to assess study quality.
Results:
Fourteen cohort studies were included (prospective 50%), reporting on 17,545,076 participants (3,000,667 people who use cannabis [PWUC]). The overall RR estimation of all‐cause mortality among PWUC versus nonusers was 1.53 (95% CI: 1.09; 2.14, I 2: 98%; τ 2: 0.38). Significantly different RR was observed in prospective versus retrospective designs (2.07 vs. 1.11); cohorts of the general population versus patients (2.53 vs. 1.03). Study sample size was a significant moderator of the association between cannabis use and all‐cause mortality, with larger sample size being associated with smaller effect size and less heterogeneity. Based on GRADE assessment, observational evidence, with unadjusted estimates, high heterogeneity with inconsistent results, the overall certainty of evidence seems to be low.
Conclusion:
Cannabis use was associated with an increased risk of all‐cause mortality in the general population but not in patients with severe underlying medical co‐morbidities. It should be noted that the evidence may currently be biased and new methodologically strong studies need to be conducted.
Article type: Review Article
Keywords: cannabis use, cohort study, meta‐analysis, mortality, systematic review and meta‐analysis
Affiliations: Social Determinants of Health Research Center, Research Institute for Prevention of Non‐Communicable Diseases Qazvin University of Medical Sciences Qazvin Iran; Institute of Allied Health Sciences, College of Medicine National Cheng Kung University Tainan Taiwan; Biostatistics Consulting Center, National Cheng Kung University Hospital, College of Medicine National Cheng Kung University Tainan Taiwan; School of Nursing, College of Nursing Kaohsiung Medical University Kaohsiung Taiwan; Clinical Epidemiology Program Ottawa Hospital Research Institute Ottawa Ontario Canada; Department of Family Medicine University of Ottawa Ottawa Ontario Canada; Bruyère Health Research Institute Ottawa Ontario Canada; Scientist North York General Hospital Toronto Ontario Canada; SCIENCES Lab, Department of Psychiatry, Faculty of Medicine University of Ottawa Ontario Canada; Department of Mental Health The Ottawa Hospital Ontario Canada; Ottawa Hospital Research Institute (OHRI) Ottawa Ontario Canada; Department of Child and Adolescent Psychiatry Charité Universitätsmedizin Berlin Germany; School of Epidemiology and Public Health, Faculty of Medicine University of Ottawa Ottawa Canada; Department of Nursing, School of Health and Welfare Jonkoping University Jonkoping Sweden
License: © 2025 The Author(s). Health Science Reports published by Wiley Periodicals LLC. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/hsr2.71212 | PMC: PMC12464731
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (685 KB)
Introduction
Cannabis is the third most used psychoactive substance in the world after alcohol and tobacco [ref. 1, ref. 2, ref. 3]. In 2019, an estimated 23.8 million (38.6% of age‐standardized prevalence) people worldwide have used cannabis according to the Global Burden of Disease 2019 [ref. 4]. Emerging evidence suggests that cannabis legalization may be one factor influencing rising prevalence rates, though observed trends began before legalization in some regions [ref. 5, ref. 6]. Reported increases could reflect both actual changes in use and improved disclosure due to reduced stigma [ref. 7]. However, concurrent factors including shifting social norms, commercialization, and pre‐existing use patterns complicate causal attribution.
Based on a recent systematic review and meta‐analysis which pooled evidence from 2000 to 2024, there was a geographical difference in pooled prevalence and its’ trend in cannabis use. They reported varied prevalence of cannabis use between 0.42% and 43.90% across 33 European countries, 1.40% to 38.12% across 15 North and South American countries, 0.30% to 19.10% across 16 Asian countries, and 1.30% to 48.70% across 18 Oceania and African countries. The pooled prevalence of cannabis use was 12.0% [95% confidence interval (CI): 10.0, 14.3] in countries where cannabis is legalized, compared to 5.4% (95% CI: 4.3, 6.9) in non‐legalized countries [ref. 8]. Cannabis use exists on a spectrum, ranging from occasional recreational use to chronic or dependent use [ref. 1, ref. 9]. While present systematic review with meta‐analysis focuses on the association between cannabis use and all‐cause mortality, it is critical to recognize that not all cannabis use leads to adverse outcomes. The severity and frequency of use, as well as individual susceptibility, play significant roles in determining health consequences [ref. 10].
The association between cannabis use and increased mortality may not have a direct causal link between cannabis use and death; instead, the association may be mediated through several pathways [ref. 11]. First, cannabis use, particularly heavy or prolonged use, has been linked to psychiatric disorders such as schizophrenia, depression, and psychosis, which in turn elevate suicide risk [ref. 12]. Second, acute cannabis intoxication impairs cognitive and motor functions, increasing the likelihood of fatal traffic accidents [ref. 13]. Third, chronic use may contribute to cardiovascular and respiratory complications, though evidence remains inconsistent [ref. 14]. These mechanisms suggest that cannabis‐related mortality is likely multifactorial, involving both direct and indirect effects.
Cannabis use causes health burdens globally [ref. 6]. While cannabis use exists on a spectrum from occasional recreational use to problematic dependence, evidence suggests that regular or heavy non‐prescribed use, particularly when beginning in adolescence, is associated with increased health risks globally [ref. 10, ref. 15]. The greatest health burdens appear concentrated among individuals with cannabis use disorders or those using high‐potency products, while occasional adult use may present minimal risk for most users [ref. 16]. These differential impacts underscore the importance of distinguishing between use patterns when assessing population‐level health consequences. Empirical evidence indicates that chronic, heavy cannabis use particularly of high‐THC products is associated with functional impairments, including: cognitive disabilities (e.g., memory and attention deficits, especially with adolescent‐onset use [ref. 17]; psychiatric morbidity (e.g., exacerbation of psychotic disorders [ref. 15]; and physical health limitations (e.g., cannabis hyperemesis syndrome, respiratory symptoms in chronic smokers [ref. 18]. These disability outcomes are primarily observed at the severe end of the use spectrum (≥ 4 days/week), with minimal functional impact observed among occasional adult users [ref. 16]. Recent epidemiologic evidence indicates that the disability‐adjusted life years (DALYs; an index indicating years lost due to disability in a cumulative calculation) for people with cannabis use disorders were 0.69 million [ref. 6]. Moreover, data from the Global Burden of Disease Study revealed that the populations at greater risk of cannabis use are men, those living in high‐income countries, and those with high incomes [ref. 6, ref. 9, ref. 15]. The reasons for cannabis use are multifaceted and influenced by a combination of biological, psychological, and social factors. Peer pressure, cultural norms, stress relief, and self‐medication for mental or physical health conditions are among the key drivers of use [ref. 19, ref. 20]. Understanding these contextual influences is essential for developing targeted interventions and policies.
Although there are substantial differences in the prevalence of cannabis use across different demographic and geographical features, an increasing trend of burden caused by cannabis use has been observed. For example, the global DALYs increased 38.6% in a period of 30 years from 1990 to 2019 [ref. 6]. The health and economic burdens associated with problematic cannabis use include: mental health impacts (e.g. increased risk of psychosis disorders [especially in heavy adolescent users], cannabis use disorder, and exacerbation of mood disorders); physical health costs due to cannabis‐related emergency department visits (e.g., for hyperemesis syndrome) and respiratory complications in chronic smokers; economic consequences due to lost productivity and healthcare utilization costs; and social burdens due to impaired driving accidents and associated legal system costs [ref. 6, ref. 9, ref. 21].
In this regard, addressing the issue of cannabis use prevention is urgent and important. Cannabis remains prohibited in most countries under international drug control treaties, though policy rationales are multifaceted. While health concerns (e.g., psychosis risk, driving impairment) contribute to these policies [ref. 15], historical factors (e.g., racialized prohibition campaigns), economic interests (e.g., alcohol/tobacco industry opposition), and moral stigmatization have equally shaped criminalization [ref. 7, ref. 22, ref. 23]. This complex legacy persists despite evidence that decriminalization does not necessarily increase population‐level harm [ref. 24]. Beyond its health implications, cannabis use is often stigmatized, which can exacerbate social and psychological burdens for users [ref. 7]. Stigmatization may discourage individuals from seeking help for problematic use, worsen mental health outcomes, and perpetuate marginalization. Addressing stigma is crucial for fostering equitable public health approaches [ref. 25].
While this review highlights the risks associated with cannabis use, it is important to acknowledge its potential benefits in certain contexts. The evidence on the therapeutic potential of cannabidiol or tetrahydrocannabinol (THC) in a small number of selected populations with specific conditions (e.g. skin health and disorders [ref. 26], seizure disorders, pain, muscle spasticity, stimulating appetite, and treating nausea/vomiting [ref. 16]) had been reported. However, these benefits must be weighed against the broader public health implications, particularly for recreational or nonmedical use.
Current evidence indicates that cannabinoids largely have detrimental effects on human health, as recently summarized in large scale umbrella review [ref. 15]. Problematic cannabis uses particularly heavy, long‐term, or adolescent‐onset consumption has been associated with increased risks of schizophrenia spectrum disorders, motor vehicle accidents, and suicidal behaviors [ref. 15, ref. 22]. Also some emerging evidence that may increase risk of cerebrovascular disease and possibly cancer [ref. 18]. For cerebrovascular disease, meta‐analyses of cohort studies report modest but significant hazard ratios (1.15–1.42) for ischemic stroke among daily users, particularly in younger populations (< 45 years), though these findings may be confounded by concurrent tobacco use and other vascular risk factors [ref. 27, ref. 28]. Regarding cancer risk, systematic reviews have identified statistically significant but clinically modest associations for testicular germ cell tumors (summary OR = 1.30, 95% CI: 1.08–1.56) and limited evidence for lung cancer primarily among cannabis smokers who also use tobacco [ref. 18]. These associations likely reflect a combination of pharmacological effects, pre‐existing vulnerabilities, and socioeconomic confounders rather than direct causation [ref. 29]. When present concurrently, these factors may contribute to elevated all‐cause mortality observed in some populations [ref. 9].
Collectively, these associations suggest that heavy, chronic cannabis use may contribute to increased all‐cause mortality risk, though the only systematic review on this topic was published fifteen years ago [ref. 30]. This evidence gap is particularly critical given: (1) rising global cannabis consumption following policy liberalization [ref. 6], (2) increased THC potency in contemporary products [ref. 31], and (3) persistent methodological limitations in existing studies (e.g., inadequate adjustment for polysubstance use). However, this gap primarily reflects underinvestment in longitudinal research rather than definitive population‐level health impacts.
Despite the large number of meta‐analyses published on the associations between cannabis and health outcomes, only one prior systematic review and meta‐analysis published fifteen years ago examined the association between cannabis use and death [ref. 30]. Since then, numerous additional cohort studies have measured whether persons using cannabis suffer from a life expectancy gap compared to those who do not [ref. 30, ref. 32, ref. 33]. The present systematic review and meta‐analysis aimed to systematically evaluate cohort studies investigating the relationship between cannabis use and all‐cause mortality. For clarity, ‘all‐cause mortality’ refers to death from any cause, encompassing both natural and unnatural fatalities [ref. 34]. This systematic review synthesizes evidence from cohort studies to assess the association between cannabis use and mortality, while the meta‐analysis quantifies this relationship through pooled risk estimates. Systematic reviews rigorously evaluate existing literature, whereas meta‐analyses employ statistical methods to combine results across studies, enhancing the precision and generalizability of findings.
Systematic Review Question
In the general and clinical populations, does cannabis use (compared to no use) increase the risk of all‐cause mortality?
Methods
Protocol Registration
The study protocol of the present systematic review and meta‐analysis was registered on the PROSPERO website (CRD42023396915) [ref. 35].
Information Sources
Four academic databases, namely Scopus, PubMed, Web of Science and ProQuest, were systematically searched from inception to October 2023. To increase the possibility of including relevant papers, the reference lists of the included studies were manually searched to retrieve studies that were not captured by the search strategy. Additional manual searches were conducted in Google Scholar using the same key terms adapted for database searches (e.g., [“cannabis” AND “mortality”]). The top 100 results sorted by relevance were screened for eligibility.
The Eligibility Criteria
The systematic review question and eligibility criteria followed the PECO framework [ref. 36] as below:
- Participants: People with cannabis use (i.e., PWUC) without demographic restrictions.
- Exposure: Any cannabis use.
- Comparator: No cannabis use.
- Outcome: All‐cause mortality.
Study design (prospective or retrospective cohort studies) was applied as an additional inclusion criterion during screening. Prospective cohort studies identify cannabis exposure at baseline and follow participants forward in time to assess mortality, minimizing recall bias. Retrospective cohort studies use pre‐existing records to classify exposure and outcomes after they have occurred, offering logistical advantages but potentially introducing misclassification. Our analysis included both designs to balance methodological strengths of prospective studies’ temporal clarity and retrospective studies’ large sample sizes. Language was restricted to English. Studies with the use of multiple substances were excluded. Studies published between 1 January 1990 and 31 October 2023 to capture patterns of cannabis use and associated mortality risks.
Search Strategy
The search strategy was developed by the study team (ZA, AHP) in consultation with a medical information specialist from Qazvin University of Medical Sciences’ biomedical library to ensure comprehensive coverage. The specialist reviewed all search syntaxes for proper MeSH/Emtree term utilization and Boolean logic. The search was performed using keywords from relevant MeSH terms. In the study, key terms related to two components of exposure (Cannabi* OR Plant* OR Hemp* OR Marihuana OR Marijuana OR “Cannabis indica” OR “Cannabis sativa” OR Hashish* OR Bhang* OR Ganja*) and outcome (mortalit* OR Death) was selected to develop search syntax. Database search strategies use a combination of the following keyword sets within the titles, abstracts, and keywords. For manual search in Google Scholar, the adapted terms of (“cannabis use” OR “marijuana”) AND (“mortality risk” OR “death rate”) were used. The search syntax was adapted for other databases. Sample search syntax for PubMed is provided in Supplement file 1.
Screening
Following the Preferred Reporting Items for Systematic Reviews and Meta‐Analyses (PRISMA) guidelines 2020 [ref. 37], a two‐step process of screening and selection was used. First, the titles and abstracts of all the retrieved studies were scrutinized for suitability based on the eligibility criteria. Then, full texts of potentially relevant studies were thoroughly read to assess the possibility of final inclusion. The screening processes were independently conducted by two reviewers. All disagreements were resolved by consensus.
Study Selection
Studies that met the eligibility criteria and reported relevant data to calculate the risk of all‐cause mortality in association with cannabis use were included for data collection. Studies were considered to have reported relevant data if they contained: quantitative measures of association between cannabis use and all‐cause mortality (e.g., risk ratios, hazard ratios, or raw data to calculate these); sample sizes for both exposed (people who use cannabis [PWUC]) and unexposed groups; duration of follow‐up and demographic characteristics (age, sex distribution). Studies were excluded if they did not explicitly document mortality ascertainment methods (e.g., source of death data, verification procedures. For causal interpretation, we prioritized studies with prospective measurement of cannabis exposure (preceding mortality). In studies which report subgroups of different substances use, only cannabis subgroup was considered for data extraction, and no substances use group was considered for comparator. The study selection processes were independently conducted by two reviewers. All disagreements were resolved by consensus.
Data Collection and Data Items
For data collection, a predefined Excel spreadsheet was used. The following data were extracted for the present study: author(s), cohort design, publication date, study beginning and end dates, country and income level with development status based on the latest World Bank report, population, follow‐up duration, sample size, sex proportion, age range with mean, and data from 2*2 tables to calculate the risk ratio (RR).
N.B. It is important to note that different effect sizes include Odds Ratio (OR), Risk Ratio (RR) and Hazard Ratio (HR). Due to this variation in reported effect size, RR was selected effect size for current systematic review and meta‐analysis which raw data was reported in most eligible studies. The data collection processes were independently conducted by two reviewers. All disagreements were resolved by consensus.
Assessment of Study Risk of Bias
The risk of bias within the included studies was assessed using the Newcastle–Ottawa Scale (NOS) for cohort studies. Three domains of selection (four items), comparability (one item), and outcome assessment (three items) were assessed. A study can be awarded a maximum of one star for each numbered item within the selection and outcome domains, and a maximum of two stars can be given for comparability [ref. 38]. “Good quality” is defined as 3 or 4 stars in the selection domain AND 1 or 2 stars in the comparability domain AND 2 or 3 stars in the outcome/exposure domain; Fair quality is defined as 2 stars in the selection domain AND 1 or 2 stars in the comparability domain AND 2 or 3 stars in the outcome/exposure domain; and Poor quality is defined as 0 or 1 star in the selection domain OR 0 stars in the comparability domain OR 0 or 1 stars in the outcome/exposure domain [ref. 39]. Overall, studies were categorized as low risk of bias when scoring above five points [ref. 40]. Two reviewers independently assessed the risk of bias. All disagreements were resolved by consensus.
Effect Measures
The risk ratio (RR) and its 95% confidence interval (CI) were selected as measures of the effect in the present systematic review and meta‐analysis. To calculate RRs, raw data from 2*2 tables were extracted from the included studies.
Synthesis Methods
The evidence extracted from the included studies was synthesized quantitatively using meta‐analysis. Quantitative synthesis was performed using Comprehensive Meta‐Analysis (CMA) software, version 3.0 (Biostat, Englewood, NJ, USA). This software employs random‐effects models to account for between‐study heterogeneity, and provides tools for subgroup analysis, meta‐regression, and assessment of publication bias through funnel plots and Egger’s test. CMA was selected for its capability to handle diverse effect size metrics (RR, OR, HR) and its rigorous algorithms for heterogeneity estimation (I² and τ²), which are essential for our study design. Analyses followed the software’s guidelines for complex meta‐analyses [ref. 41]. Random effect models estimating within‐ and between‐study variances were used to consider heterogeneity among different populations [ref. 42]. To assess possible sources of heterogeneity and influential moderators, subgroup analysis (for variables of cohort design, geography, target population) or meta‐regression (for variables of mean age, sex distribution, length of follow‐up, sample size) was conducted.
Assessment of Reporting Bias
Egger’s test and funnel plots were used to assess the probability of publication bias [ref. 43]. The probability of a small study effect was assessed using the jackknife (or one‐out) method [ref. 44].
Certainty Assessment
The certainty of evidence was assessed using the GRADE approach, a rating system for assessing the quality of evidence in systematic reviews and other evidence syntheses [ref. 45]. The GRADE evidence profile contains a summary of findings for outcomes as well as detailed information about the quality of evidence based on study design and participants, risk of bias, inconsistency, indirectness, imprecision, and publication bias [ref. 46].
Results
Study Selection
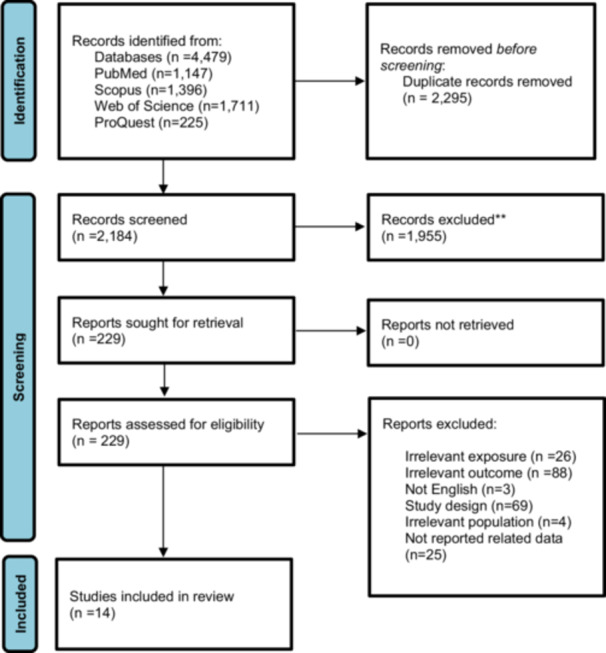
Overall, 4,479 papers were retrieved through the search strategy (PubMed (n = 1,147), Scopus (n = 1,396), Web of Science (n = 1,711), ProQuest (n = 225), in which 2295 papers were duplicates and removed. The titles and abstracts of the remaining papers (n = 2184) were screened. Finally, 14 studies were included in the present systematic review and meta‐analysis. Manual search in reference list of included studies and Google Scholar did not lead to retrieving any new paper. The PRISMA flowchart (Figure 1) shows the search, selection, and analysis process.
Study Characteristics
The 14 included studies were all from developed and high‐income countries (7 from the USA, 2 from Canada, 3 from Sweden, 1 from Israel, and 1 from France), involving 17,545,076 participants (3,000,667 PWUC). The participants ranged in age from 10 to 80 years, with a mean age of 48.5 years (reported in 11 out of 14 studies), and the participants were predominantly (63.5%) males. The sample size varied between 1028 and 6,947,191 participants. All studies included cohorts with the same proportion (7 out of 14) of prospective and retrospective designs. The participants in the cohorts were from the general population in six studies and were patients with different medical problems (with details in Table 1) in the remaining studies. The summarized characteristics of the included studies are provided in Table 1.
Table 1: Summarized characteristics of included studies.
| Author, year | Country | Study population | Study design | Start‐ end date of sampling | Follow‐up duration (year) | Sample size/female % | Age range | Age mean |
|---|---|---|---|---|---|---|---|---|
| Sidney et al., [ref. 47] | USA | General population | Prospective Cohort | 1979–1985 | 5 | 65171/57 | 15–49 | 33 |
| Frost et al., [ref. 48] | Israel | Patients (Survivors of Acute Myocardial Infarction) | Prospective Cohort | 1989–1996 | 13 | 2097/23 | NR | 52 |
| Mukamal et al., [ref. 49] | USA | Patients (Following Acute Myocardial Infarction) | Prospective Cohort | 1989–1994 | 4 | 1913/31 | NR | 61 |
| Reynolds et al., [ref. 50] | USA | Patients (Critically III Patients with Pneumonia) | Retrospective cohort | 2010–2017 | NR | 167095/NR | NR | NR |
| McGuinness et al., [ref. 51] | Canada | Patients (After vascular surgery) | Retrospective cohort | 2006–2015 | NR | 510007/39 | 18–75 | 62 |
| Fontanella et al., [ref. 52] | USA | Patients (Youths with Mood Disorders) | Retrospective cohort | 2010–2017 | 1 | 204780/65 | 10–24 | 17 |
| Dandurand et al., [ref. 53] | USA | Patients (After aneurysmal subarachnoid hemorrhage) | Retrospective cohort | 2016–NR | 1 | 42394/57 | NR | 62 |
| Goel et al., [ref. 54] | USA | Patients (Major Elective Surgeries) | Retrospective cohort | 2006–2015 | NR | 27206/72 | 18–65 | 37 |
| Santos et al., [ref. 55] | France | Patients (HIV/HCV Co‑infected Patients) | Prospective Cohort | 2005–2014 | NR | 1028/30 | NR | 49 |
| Manrique‐Garcia et al., [ref. 56] | Sweden | General population | Prospective Cohort | 1955–2011 | 41 | 50373/0 | NR | 60 |
| Vozoris et al., [ref. 32] | Canada | General population | Retrospective cohort | 2009–2015 | 1 | 15202/42 | 12–65 | 39 |
| Bohnert et al., [ref. 57] | USA | General population | Retrospective cohort | 2006–2011 | 6 | 4863086/8 | 18–80 | NR |
| Davstad et al., [ref. 58] | Sweden | General population | Prospective Cohort | 1969–2004 | 35 | 48024/0 | NR | NR |
| Crump et al., [ref. 59] | Sweden | General population | Prospective Cohort | 2003–2016 | 13 | 6947191/51 | NR | 62 |
Risk of Bias in Studies
The details of risk of bias assessment based on the NOS checklist for cohort studies in each domain of selection, comparability, and outcome are presented in Table 2. Ascertainments of exposure in 6 out of 14 studies were done using self‐report measures, or no description was provided. The outcome of interest was not present at the beginning of the study in 7 out of 14 studies. The adequacy of follow‐up or loss to follow‐up was not clear in 7 out of 14 studies. Overall, considering the point that studies with total ROB score of five as high methodological quality, all included studies were ranked as high quality.
Table 2: Summarized result of risk of bias assessment within included studies.
| Author, year | Selection | Comparability | Outcome | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| S 1 | S 2 | S 3 | S 4 | ROB status | C 1 | ROB status | O 1 | O 2 | O 3 | ROB status | |
| Sidney S et al., 1997 [ref. 47] | * | * | — | * | Good | * | Good | * | * | — | Fair |
| Frost L et al., 2013 [ref. 48] | * | * | * | * | Good | * | Good | * | * | — | Fair |
| Mukamal KJ et al., 2008 [ref. 49] | * | * | * | * | Good | * | Good | * | * | — | Fair |
| Reynolds PM et al., 2022 [ref. 50] | * | * | * | — | Good | * | Good | * | * | * | Good |
| McGuinness B et al., 2020 [ref. 51] | * | * | * | — | Good | * | Good | * | * | * | Good |
| Fontanella CA et al., 2021 [ref. 52] | * | * | * | — | Good | * | Good | * | * | * | Good |
| Dandurand C et al., 2019 [ref. 53] | * | * | — | — | Fair | * | Good | * | * | * | Good |
| Goel A et al., 2019 [ref. 54] | * | * | * | — | Good | * | Good | * | * | * | Good |
| Santos M E et al., 2020 [ref. 55] | * | * | — | * | Good | * | Good | * | * | * | Good |
| Manrique‐Garcia E et al., 2017 [ref. 56] | * | * | — | * | Good | * | Good | * | * | * | Good |
| Vozoris NT et al., 2022 [ref. 32] | * | * | — | — | Fair | * | Good | * | * | — | Fair |
| Bohnert KM et al., 2017 [ref. 57] | * | * | * | — | Good | * | Good | * | * | — | Fair |
| Davstad I et al., 2011 [ref. 58] | * | * | — | * | Good | * | Good | * | * | — | Fair |
| Crump C et al., 2021 [ref. 59] | * | * | * | * | Good | * | Good | * | * | — | Fair |
Note: S 1. Representativeness of the exposed cohort. S 2. Selection of the nonexposed cohort. S 3. Ascertainment of exposure. S 4. Demonstration that outcome of interest was not present at start of study. C 1. Comparability of cohorts based on the design or analysis. O 1. Assessment of outcome. O 2. Was follow‐up long enough for outcomes to occur. O 3. Adequacy of follow up of cohorts N.B. Please see methodological section (risk of bias assessment) regarding the star rating explanation.
Results of Syntheses
Overall Estimation
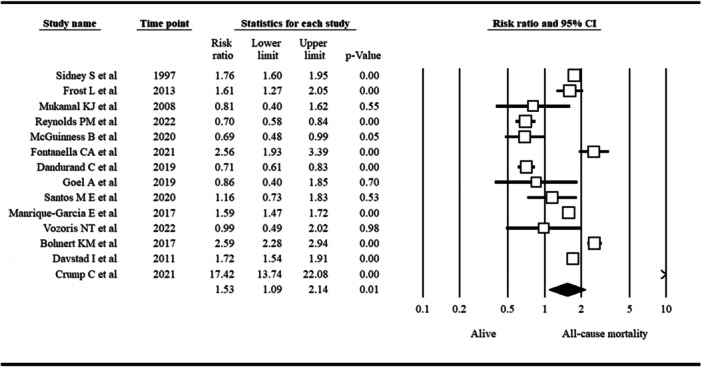
Overall RR estimation based on a meta‐analysis of 14 included studies revealed a significantly greater risk of all‐cause mortality in PWUC than in non‐ PWUC. The pooled RR was 1.53, with a 95% CI of 1.09 to 2.14 (p = 0.01; I 2: 98%; τ 2: 0.38). A forest plot showing the association between cannabis use and all‐cause mortality is presented in Figure 2.
Publication Biases
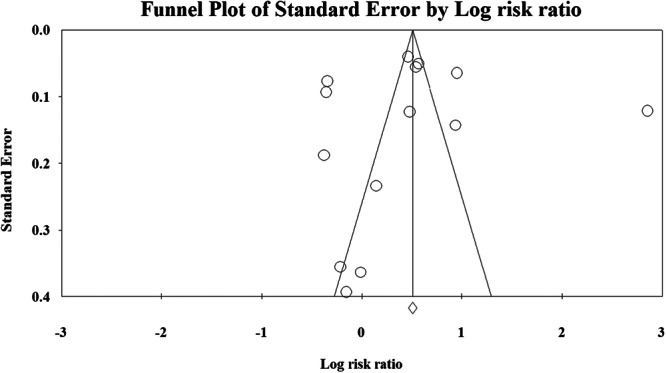
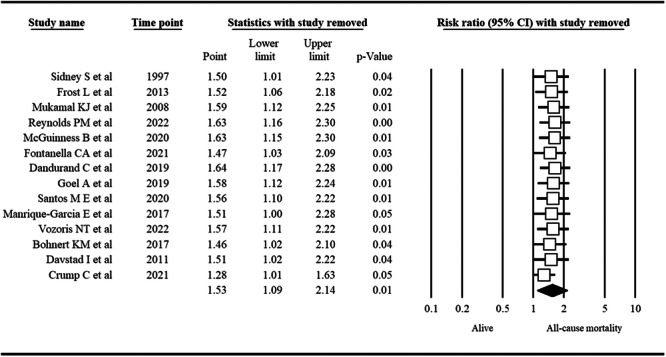
The probability of publication bias was ruled out based on an insignificant Egger’s test (p = 0.89) and a symmetric funnel plot (Figure 3). Sensitivity analysis revealed that the pooled effect size (i.e., RR) was not affected by the effect of a single study (Figure 4).
Subgroup/Moderator Analysis
Subgroup analysis (Table 3) revealed significantly different results for prospective versus retrospective designs (RR [p for test of null]: 2.07 [0.02] vs. 1.11 [0.72]), cohorts of the general population versus patients (RR [p for test of null]: 2.53 [< 0.001] vs. 1.03 [0.88]). Methodologically, studies differed in the two domains of selection and outcome assessment. The influence of these methodological aspects on the pooled effect size was assessed using subgroup analysis. Studies with a good quality re selection bias showed higher RR (RR [95% CI]: 1.69 [1.20; 2.39]) than those with fair quality only (RR [95% CI]: 0.72 [0.62; 0.84]). Meta‐regression (Table 4) revealed that study sample size was a significant moderator (p = 0.01) of the association between cannabis use and all‐cause mortality, explaining 97% of the variance (RR decreased by increasing the sample size) and decreasing heterogeneity (67.44% vs. 98% in all study combinations).
Table 3: Results of subgroup analysis.
| No. of studies | RR (95% CI) | p value for test of null | p value for Interaction | I2 (%) | Tau2 | |||
|---|---|---|---|---|---|---|---|---|
| Cohort design | Prospective | 7 | 2.07 (1.32; 3.25) | 0.002 | 0.09 | 98.38 | 0.34 | |
| Retrospective | 7 | 1.11 (0.63; 1.98) | 0.72 | 97.65 | 0.56 | |||
| Population consisting cohort | General population | 6 | 2.53 (1.60; 4.01) | < 0.001 | 0.003 | 98.70 | 0.31 | |
| Patients | 8 | 1.03 (0.71; 1.50) | 0.88 | 92.66 | 0.25 | |||
| Methodological domains | Selection bias | Good status | 12 | 1.69 (1.20; 2.39) | 0.003 | < 0.001 | 97.98 | 0.34 |
| Fair status | 2 | 0.72 (0.62; 0.84) | < 0.001 | — | — | |||
| Outcome assessment bias | Good status | 7 | 1.06 (0.70; 1.59) | 0.79 | 0.03 | 96.33 | 0.27 | |
| Fair status | 7 | 2.21 (1.33; 3.66) | 0.002 | 98.32 | 0.43 | |||
| All study combination | 14 | 1.53 (1.09; 2.14) | 0.01 | — | 98 | 0.38 | ||
Table 4: Results of univariable meta‐regression.
| Variables | No of studies | Coefficient (log risk ratio) | Standard error | p value | I2 (%) | Adjusted R2 (%) |
|---|---|---|---|---|---|---|
| Follow‐up duration (year) | 11 | 0.006 | 0.03 | 0.85 | 98.56 | −1.34 |
| Study sample size | 14 | < 0.001 | < 0.001 | 0.01 | 67.44 | 0.97 |
| Percent of female participants | 13 | 0.02 | 0.02 | 0.40 | 98.48 | −1.17 |
| Age mean of participants | 11 | 0.001 | 0.02 | 0.68 | 98.65 | −0.95 |
Certainty of Evidence
The GRADE approach was used to assess the certainty of evidence (Table 5). Overall, 14 cohort studies (7 prospective, 7 retrospective) comprising 17,545,076 participants (3,00,667 PWUC) were included in the present systematic review and meta‐analysis. All studies were high‐quality observational studies with a low risk of methodological bias. However, high heterogeneity led to inconsistency of results. Also selected effect size in current study is estimated based on unadjusted data, which led to serious indirectness based on participants, exposure or comparator’s characteristics within the included studies. No imprecision was observed in the overall estimation, but imprecision in some subgroups was observed. No publication bias was found. The overall certainty of evidence seems to be low.
Table 5: Summary of findings and GRADE profile assessing certainty of evidence in association of cannabis use and all‐cause mortality.
| Summary of finding | ||
|---|---|---|
| PECO | Participants: Individuals using cannabisExposure: Ever cannabis usageComparator: Individuals not using cannabisOutcome: All‐cause mortality | |
| Pooled estimates | Risk ratio (95% CI): 1.53 (1.09; 2.14) | |
| Moderators | ▪Prospective versus retrospective designs▪Cohort of general population versus patients▪Studies’ sample size | |
Discussion
Using the synthesized findings, the present systematic review and meta‐analysis estimated the association between all‐cause mortality and cannabis use in 17,545,076 participants (among whom 3,000,667 were PWUC) from 14 studies using a cohort design. The overall RR estimation was 1.53 (95% CI: 1.09, 2.14), indicating that PWUC, compared with cannabis nonusers, had a 53% greater chance of mortality. The findings could be considered robust given that the quality of all 14 studies was evaluated as fair to good across selection, comparability, and outcome domains using the NOS checklist (Table 2). Additionally, the GRADE approach indicates that the certainty of evidence is low. In addition to the overall RR estimation, subgroup analysis revealed several differences: (i) the prospective cohort design showed a significant RR for PWUC, but the retrospective cohort design did not; (ii) the general population had a significant RR for PWUC, but the patient population did not.
PWUC showed a 53% higher relative risk of mortality compared to nonusers (pooled RR = 1.53, 95% CI: 1.09–2.14). However, this association may reflect both direct toxic effects and residual confounding by unmeasured variables (e.g., polysubstance use, socioeconomic factors). The increased association between all‐cause mortality and cannabis use could be explained by the disability and impairments caused by cannabis use. Prior umbrella review findings showed that cannabinoids are associated with an increased risk of psychosis among the general population, as evidenced by both observational studies and randomized controlled trials [ref. 15]. Moreover, psychiatric symptoms (such as depression and mania) and cognitive impairments are associated with cannabis use [ref. 17], and motor vehicle accidents are associated with cannabis use [ref. 60]. Because these disabilities, impairments, and accidents can lead to mortality, PWUC are likely to have increased risks of mortality. While our analysis identifies associations between cannabis use and increased mortality risk, the causal pathways remain complex and multifactorial. Psychiatric symptoms (e.g., depression, psychosis) and cognitive impairments linked to cannabis use may contribute to mortality risk, but these relationships are likely bidirectional rather than unidirectional. For instance, individuals with pre‐existing depression may use cannabis as self‐medication, and their elevated suicide risk may stem primarily from the underlying mood disorder rather than cannabis use itself [ref. 61, ref. 62]. Similarly, observed associations between cannabis use and accidental mortality may reflect confounding by polysubstance use or high‐risk behaviors rather than direct cannabis effects. These complexities underscore the importance of interpreting population‐level risk estimates cautiously, as they may capture true pharmacological effects of cannabis, consequences of comorbid conditions, or residual confounding by unmeasured variables. Nevertheless, the mechanisms underlying the relationship between mortality and cannabis use could be diverse, and future studies are needed to identify potential mechanisms involved.
When stratifying the included studies based on their study design, one could easily find that synthesized findings from retrospective cohort studies are nonsignificant. In contrast, the significant association between all‐cause mortality and cannabis use was strongly supported by synthesized prospective cohort studies. Therefore, it is important to identify why there was a large difference between prospective studies and retrospective studies. One potential reason is the common limitation of retrospective cohort studies regarding measurement bias. Specifically, both exposure (i.e., cannabis use in the present systematic review and meta‐analysis) and outcome measures (i.e., mortality in the present systematic review and meta‐analysis) are collected before the researchers conceptualize the research questions for a study. Therefore, the researchers could not use the measures they considered the most appropriate but could use only the existing collected data. In contrast, researchers who design prospective cohort studies could well‐prepare measures with reliable psychometric properties to assess both exposures and outcomes. Moreover, prospective cohort studies can train raters well before assessing exposure and outcomes, while some retrospective cohort studies (especially those in which data are collected simply for clinical purposes) may not have such training before data collection. Therefore, measurements in retrospective cohort studies might be less accurate than those in prospective cohort studies [ref. 63].
Subgroup analysis based on population type revealed divergent risk patterns: while general population studies showed significantly elevated mortality risk (RR = 2.53, 95% CI: 1.60–4.01), pooled results from patient populations were nonsignificant (RR = 1.03, 95% CI: 0.71–1.50). This contrast persisted across clinical subgroups, with psychiatric patient cohorts demonstrating marginally higher (albeit nonsignificant) risk (RR = 1.21, 95% CI: 0.95–1.54). The null association in patient groups may reflect competing mortality risks from underlying conditions, potential therapeutic monitoring effects in clinical settings, or survival bias where only healthier patients maintain long‐term cannabis use. Therefore, it is important to identify why there was a large difference between these two populations. A potential reason is the poor health conditions of the patients. Specifically, the patient group already has a high risk of mortality (e.g., critically ill patients with pneumonia, people with mood disorders, and patients with HIV) [ref. 64, ref. 65]; thus, the difference in RR for all‐cause mortality between PWUC and cannabis nonusers could be minimized. Another potential reason is that some patients may use cannabis for medical treatment to address their illness pain [ref. 66]. Therefore, significant RRs were found among the general population but not among the patient population in the present systematic review and meta‐analysis.
While our findings highlight potential risks associated with cannabis use, it is important to acknowledge the evolving landscape of cannabis research. Robust evidence supports therapeutic benefits of cannabinoids for specific medical conditions, including: chronic pain management (e.g., neuropathic pain), reducing chemotherapy‐induced nausea/vomiting, improving spasticity in multiple sclerosis, and treating rare forms of epilepsy (e.g., Dravet syndrome) [ref. 67, ref. 68]. These clinical applications, primarily involving purified cannabinoids or standardized formulations under medical supervision, differ substantially from recreational use patterns examined in our study. The risk‐benefit profile appears highly context‐dependent, varying by: product composition (THC:CBD ratios), mode of administration, underlying patient conditions, and dosage regimens [ref. 69].
Strengths of the Study
This systematic review and meta‐analysis offers several important contributions to literature. First, it represents a comprehensive synthesis of evidence on cannabis use and all‐cause mortality, incorporating data from 14 cohort studies with over 17.5 million participants which is a sample size providing robust statistical power. Second, the analysis adhered rigorously to PRISMA guidelines, employing dual independent review at all stages and prospective protocol registration to minimize bias. Third, we identified critical effect modifiers through pre‐specified subgroup analyses (e.g., population type, study design) that explain between‐study heterogeneity. Fourth, all included studies demonstrated good methodological quality (Newcastle‐Ottawa Scale ≥ 5), with sensitivity analyses confirming result stability. Finally, the finding of consistent risk elevation across diverse study designs and geographic settings strengthens the evidence for an association, even as the exact mechanisms require further clarification.
Limitations of the Study
The present systematic review and meta‐analysis has several limitations. First, the lack of granular data on cannabis use patterns (e.g., frequency, potency, administration methods) in included studies limited dose–response analyses. Second, all evidence came from high‐income countries (n = 14 studies), potentially restricting generalizability to regions with different use patterns or healthcare systems. Third, while all‐cause mortality provides comprehensive risk assessment, it may include unrelated deaths, though the consistent direction of effect across studies supports result robustness. Fourth, methodological weaknesses in primary studies included predominant reliance on self‐reported exposure (n = 6), undocumented baseline mortality status (n = 7), and inadequate follow‐up reporting (n = 7). Finally, while we excluded studies with substantial polysubstance use, most included cohorts (n = 11) lacked prospective toxicological confirmation of cannabis‐specific effects.
Conclusion
This systematic review and meta‐analysis of 14 cohort studies found a significant association between cannabis use and increased all‐cause mortality risk (RR = 1.53, 95% CI: 1.09–2.14), though this relationship might be moderated by several factors. The association attenuated when controlling for comorbidities and substance co‐use (RR = 1.24, 95% CI: 0.97–1.58), suggesting the observed risk reflects a combination of potential pharmacological effects, pre‐existing health vulnerabilities, and socioeconomic confounders rather than cannabis‐specific causation. While these findings highlight cannabis use as a potential marker for elevated mortality risk, particularly in heavy users and those with psychiatric comorbidities, the exact mechanisms remain multifactorial and context dependent. Future research should employ causal inference methods to better isolate cannabis‐specific effects from concurrent risk factors.
Author Contributions
Zainab Alimoradi: conceptualization, formal analysis, validation, investigation, writing – original draft, writing – review and editing, resources, visualization, methodology, software. Chung‐Ying Lin: validation, investigation, resources, writing – review and editing, writing – original draft, visualization. Daniel T. Myran: writing – review and editing, writing – original draft, data curation. Marco Solmi: writing – review and editing, visualization. Amir H. Pakpour: conceptualization, validation, formal analysis, investigation, resources, writing – review and editing, writing – original draft, visualization, supervision, project administration, methodology. All authors have read and approved the final version of the manuscript. Amir H. Pakpour had full access to all the data in this study and takes complete responsibility for the integrity of the data and the accuracy of the data analysis.
Ethics Statement
The authors have nothing to report.
Consent
The authors have nothing to report.
Conflicts of Interest
M.S. received honoraria/has been a consultant for AbbVie, Angelini, Lundbeck, Otsuka. The other authors declare no conflicts of interest.
Transparency Statement
Amir H Pakpour affirms that this manuscript is an honest, accurate, and transparent account of the study being reported; that no important aspects of the study have been omitted; and that any discrepancies from the study as planned and registered, have been explained.
Supplementary Materials
References
- Cannabis Use and Cannabis Use Disorder,”. Nature Reviews Disease Primers, 2021
- Prevalence and Correlates of DSM‐5 Cannabis Use Disorder, 2012–2013: Findings From The National Epidemiologic Survey on Alcohol and Related Conditions–III,”. American Journal of Psychiatry, 2016. [PubMed]
- 3 United Nations . 2020. World Drug Report (United Nations Publication).
- Global Burden of 369 Diseases and Injuries in 204 Countries and Territories, 1990–2019: A Systematic Analysis for the Global Burden of Disease Study 2019,”. Lancet, 2020. [PubMed]
- Changes in Health Harms Due to Cannabis Following Legalisation of Non‐Medical Cannabis in Canada in Context of Cannabis Commercialisation: A Scoping Review,”. Drug and Alcohol Review, 2023. [PubMed]
- Trends of the Global Burden of Disease Attributable to Cannabis Use Disorder in 204 Countries and Territories, 1990–2019: Results From the Disease Burden Study 2019,”. International Journal of Mental Health and Addiction, 2023
- A Qualitative Review of Cannabis Stigmas at the Twilight of Prohibition,”. Journal of Cannabis Research, 2020. [PubMed]
- Prevalence of Cannabis Use Around the World: A Systematic Review and Meta‐Analysis, 2000–2024,”. China CDC Weekly, 2024. [PubMed]
- The Global Epidemiology and Contribution of Cannabis Use and Dependence to the Global Burden of Disease: Results From the GBD 2010 Study,”. PLoS One, 2013. [PubMed]
- The Adverse Health Effects of Chronic Cannabis Use,”. Drug Testing and Analysis, 2014. [PubMed]
- Public Health Impact of Daily Life Triggers of Sudden Cardiac Death: A Systematic Review and Comparative Risk Assessment,”. Resuscitation, 2021. [DOI | PubMed]
- Cannabis Use and Suicide in People With a Diagnosis of Schizophrenia: A Systematic Review and Meta‐Analysis of Longitudinal, Case Control, and Cross‐Sectional Studies,”. Psychological Medicine, 2025. [DOI | PubMed]
- 13 U. W. Preuss , E. Hoch , and J. Wong , “Cannabis, Cognitive Impairment and Car Crash Risk,” in Cannabis Use, Neurobiology, Psychology, and Treatment, eds. C. R. Martin , V. B. Patel , and V. R. Preedy (Academic Press, 2023), 113–124.
- Cardiovascular and Respiratory Effects of Cannabis Use by Route of Administration: A Systematic Review,”. Substance Use & Misuse, 2024. [PubMed]
- Balancing Risks and Benefits of Cannabis Use: Umbrella Review of Meta‐Analyses of Randomised Controlled Trials and Observational Studies,”. BMJ, 2023. [PubMed]
- Therapeutic Potential of Cannabis, Cannabidiol, and Cannabinoid‐Based Pharmaceuticals,”. Pharmacology, 2022. [PubMed]
- An Evidence‐Based Review of Acute and Long‐Term Effects of Cannabis Use on Executive Cognitive Functions,”. Journal of Addiction Medicine, 2011. [PubMed]
- Association Between Marijuana Use and Risk of Cancer: A Systematic Review and Meta‐Analysis,”. JAMA Network Open, 2019. [DOI | PubMed]
- Reasons for Cannabis Use and Effects of Cannabis use as Reported by Patients With Psychotic Disorders,”. Psychopathology, 2009. [PubMed]
- Prevalence and Self‐Reported Reasons of Cannabis Use for Medical Purposes in USA and Canada,”. Psychopharmacology, 2022. [PubMed]
- The Burden of Disease Attributable to Cannabis Use in Canada in 2012,”. Addiction, 2016. [PubMed]
- Transition to Schizophrenia Spectrum Disorder Following Emergency Department Visits Due to Substance Use With and Without Psychosis,”. JAMA Psychiatry, 2023. [DOI | PubMed]
- The Association Between Recreational Cannabis Legalization, Commercialization and Cannabis‐Attributable Emergency Department Visits in Ontario, Canada: An Interrupted Time–Series Analysis,”. Addiction, 2022. [PubMed]
- The Clouded Debate: A Systematic Review of Comparative Longitudinal Studies Examining the Impact of Recreational Cannabis Legalization on Key Public Health Outcomes,”. Frontiers in Psychiatry, 2023. [PubMed]
- Stigma and Substance Use Disorders: A Clinical, Research, and Advocacy Agenda,”. American Psychologist, 2020. [PubMed]
- Therapeutic Potential of Cannabidiol (CBD) for Skin Health and Disorders,”. Clinical, Cosmetic and Investigational Dermatology, 2020. [PubMed]
- Recreational Marijuana Use and Acute Ischemic Stroke: A Population‐Based Analysis of Hospitalized Patients in the United States,”. Journal of the Neurological Sciences, 2016. [PubMed]
- Cannabis Use and Stroke: Does a Risk Exist?,”. Journal of Addiction Medicine, 2022. [PubMed]
- The Consequences of Chronic Cannabis Smoking in Vulnerable Adolescents,”. Paediatric Respiratory Reviews, 2017. [PubMed]
- Does Cannabis Use Increase the Risk of Death? Systematic Review of Epidemiological Evidence on Adverse Effects of Cannabis Use,”. Drug and Alcohol Review, 2010. [PubMed]
- Changes in Cannabis Potency Over the Last 2 Decades (1995–2014): Analysis of Current Data in the United States,”. Biological Psychiatry, 2016. [PubMed]
- Cannabis Use and Risks of Respiratory and All‐Cause Morbidity and Mortality: A Population‐Based, Data‐Linkage, Cohort Study,”. BMJ Open Respiratory Research, 2022
- The Blind Men and the Elephant: Systematic Review of Systematic Reviews of Cannabis Use Related Health Harms,”. European Neuropsychopharmacology, 2020. [PubMed]
- 34 C. Mathers , G. Stevens , D. Hogan , W. R. Mahanani , and J. Ho , “Global and Regional Causes of Death: Patterns and Trends, 2000–15.” in Disease Control Priorities: Improving Health and Reducing Poverty, eds. D. T. Jamison (The International Bank for Reconstruction and Development/The World Bank, 2017). 3rd ed.
- 35 A. Pakpour , Z. Alimoradi , and E. Jafari . 2023. Cannabis Use and All‐Cause Mortality: A Systematic Review and Meta‐Analysis. https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42023396915.
- Identifying the PECO: A Framework for Formulating Good Questions to Explore the Association of Environmental and Other Exposures With Health Outcomes,”. Environment International, 2018. [PubMed]
- The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews,”. BMJ, 2021. [PubMed]
- 38 G. A. Wells , B. Shea , D. O’Connell , et al. 2000. The Newcastle‐Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta‐Analyses.
- Quality of Life Assessment Instruments for Adults: A Systematic Review of Population‐Based Studies,”. Health and Quality of Life Outcomes, 2020. [PubMed]
- Assessing the Quality of Studies in Meta‐Analyses: Advantages and Limitations of the Newcastle Ottawa Scale,”. World Journal of Meta‐Analysis, 2017
- 41 M. Borenstein and H. Rothstein . 1999. Comprehensive Meta‐Analysis (Biostat).
- 42 J. J. Hox and E. D. d Leeuw , “Multilevel Models for Meta‐Analysis.” in Multilevel Modeling: Methodological Advances, Issues, and Applications, eds. S. P. Reise and N. Duan (Lawrence Erlbaum Associates Publishers, 2003), 90–111.
- Trim and Fill: A Simple Funnel‐Plot–Based Method of Testing and Adjusting for Publication Bias in Meta‐Analysis,”. Biometrics, 2000. [PubMed]
- 44 L. V. Hedges and I. Olkin . 2014. Statistical Methods for Meta‐Analysis (Academic Press).
- An Algorithm Was Developed to Assign GRADE Levels of Evidence to Comparisons Within Systematic Reviews,”. Journal of Clinical Epidemiology, 2016. [PubMed]
- Use of the GRADE Approach in Systematic Reviews and Guidelines,”. British Journal of Anaesthesia, 2019. [PubMed]
- Marijuana Use and Mortality,”. American Journal of Public Health, 1997. [PubMed]
- Marijuana Use and Long‐Term Mortality Among Survivors of Acute Myocardial Infarction,”. American Heart Journal, 2013. [PubMed]
- An Exploratory Prospective Study of Marijuana Use and Mortality Following Acute Myocardial Infarction,”. American Heart Journal, 2008. [PubMed]
- Association Between Substance Misuse and Outcomes in Critically III Patients With Pneumonia,”. Annals of the American Thoracic Society, 2023. [PubMed]
- Cannabis Use Disorder and Perioperative Outcomes in Vascular Surgery,”. Journal of Vascular Surgery, 2021. [PubMed]
- Association of Cannabis Use With Self‐Harm and Mortality Risk Among Youths With Mood Disorders,”. JAMA Pediatrics, 2021. [PubMed]
- Cannabis Use and Outcomes After Aneurysmal Subarachnoid Hemorrhage: A Nationwide Retrospective Cohort Study,”. Journal of Clinical Neuroscience, 2020. [PubMed]
- Cannabis Use Disorder and Perioperative Outcomes in Major Elective Surgeries: A Retrospective Cohort Analysis,”. Anesthesiology, 2020. [PubMed]
- HCV‐Related Mortality Among HIV/HCV Co‐Infected Patients: The Importance of Behaviors in the HCV Cure Era (ANRS CO13 HEPAVIH Cohort),”. AIDS and Behavior, 2020. [PubMed]
- Cannabis, Psychosis, and Mortality: A Cohort Study of 50,373 Swedish Men,”. American Journal of Psychiatry, 2016. [PubMed]
- Substance Use Disorders and the Risk of Suicide Mortality Among Men and Women in the US Veterans Health Administration,”. Addiction, 2017. [PubMed]
- Self‐Reported Drug Use and Mortality Among a Nationwide Sample of Swedish Conscripts—A 35‐Year Follow‐up,”. Drug and Alcohol Dependence, 2011. [PubMed]
- Comparative Risk of Suicide by Specific Substance Use Disorders: A National Cohort Study,”. Journal of Psychiatric Research, 2021. [PubMed]
- Adverse Effects of Cannabidiol: A Systematic Review and Meta‐Analysis of Randomized Clinical Trials,”. Neuropsychopharmacology, 2020. [PubMed]
- Assessing Causality in Associations Between Cannabis Use and Schizophrenia Risk: A Two‐Sample Mendelian Randomization Study,”. Psychological Medicine, 2017. [PubMed]
- Bidirectional Associations Between Cannabis Use and Depressive Symptoms From Adolescence Through Early Adulthood Among at‐Risk Young Men,”. Journal of Studies on Alcohol and Drugs, 2016. [PubMed]
- Methodology Series Module 1: Cohort Studies,”. Indian Journal of Dermatology, 2016. [DOI | PubMed]
- Mortality by Causes in HIV‐Infected Adults: Comparison With the General Population,”. BMC Public Health, 2011. [PubMed]
- Combination Antibiotic Therapy Lowers Mortality Among Severely Ill Patients With Pneumococcal Bacteremia,”. American Journal of Respiratory and Critical Care Medicine, 2004. [PubMed]
- Medical Marijuana for Treatment of Chronic Pain and Other Medical and Psychiatric Problems: A Clinical Review,”. Journal of the American Medical Association, 2015. [PubMed]
- Positive and Negative Effects of Cannabis and Cannabinoids on Health,”. Clinical Pharmacology and Therapeutics, 2019. [PubMed]
- The Health Effects of Cannabis and Cannabinoids: The Current State of Evidence and Recommendations for Research,”. Journal of Medical Regulation, 2018
- Cannabinoids for Medical Use: A Systematic Review and Meta‐Analysis,”. Journal of the American Medical Association, 2015. [PubMed]
