Psychotomimetic compensation versus sensitization
Abstract
It is a paradox that psychotomimetic drugs can relieve symptoms that increase risk of and cooccur with psychosis, such as attention and motivational deficits (e.g., amphetamines), pain (e.g., cannabis) and symptoms of depression (e.g., psychedelics, dissociatives). We introduce the ideas of psychotomimetic compensation and psychotomimetic sensitization to explain this paradox. Psychotomimetic compensation refers to a short‐term stressor or drug‐induced compensation against stress that is facilitated by engagement of neurotransmitter/modulator systems (endocannabinoid, serotonergic, glutamatergic and dopaminergic) that mediate the effects of common psychotomimetic drugs. Psychotomimetic sensitization occurs after repeated exposure to stress and/or drugs and is evidenced by the gradual intensification and increase of psychotic‐like experiences over time. Theoretical and practical implications of this model are discussed.
Article type: Review Article
Keywords: psychedelic, psychosis, pychotomimetic, schizophrenia, sensitization, stress, substance use
Affiliations: Department of Human Development and Family Studies, School of Human Ecology University of Wisconsin‐Madison Madison Wisconsin USA; Department of Neurology and Psychiatry University of California San Francisco San Francisco California USA; Department of Psychiatry, School of Medicine and Public Health University of Wisconsin‐Madison Madison Wisconsin USA; Vail Health Behavioral Health Innovation Center Vail Colorado USA; Center for the Study of Human Health Emory University Atlanta Georgia USA; Department of Spiritual Health Emory University Woodruff Health Sciences Center Atlanta Georgia USA
License: © 2024 The Author(s). Pharmacology Research & Perspectives published by British Pharmacological Society and American Society for Pharmacology and Experimental Therapeutics and John Wiley & Sons Ltd. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/prp2.1217 | PubMed: 38923845 | PMC: PMC11194300
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (3.6 MB)
INTRODUCTION
Well known psychotomimetic agents include classic psychedelics such as mescaline, psilocybin and its active metabolite psilocin, and LSD ref. prp21217-bib-0001; cannabis and its psychoactive constituent Delta‐9‐Tetrahydrocannabinol (THC)ref. prp21217-bib-0002; dissociative anesthetics such as ketamine ref. prp21217-bib-0003; and frequent high doses of dopamine‐enhancing drugs like methamphetamine.ref. prp21217-bib-0004 The capacity for these drugs to elicit psychotomimetic “psychosis mimicking” statesref. prp21217-bib-0005 is supported by preclinical studies and animal models of psychosis,ref. prp21217-bib-0006 psychological tests and subjective reports in humans,ref. prp21217-bib-0007, ref. prp21217-bib-0008 as well as epidemiological data on substance use in populations who are at risk for and experience psychosis.ref. prp21217-bib-0009, ref. prp21217-bib-0010
However, the fact that psychotomimetics are used to treat pain,ref. prp21217-bib-0011 attention deficitsref. prp21217-bib-0012 and depressionref. prp21217-bib-0013 raises questions about the nature of psychotic symptoms and whether they reflect compensatory (albeit dysfunctional) responses to stress or homeostatic imbalance. It has been argued before that psychosis is a mere indicator of homeostatic imbalance, illness, or enduring social dysfunction,ref. prp21217-bib-0014 but this view does not explain how temporary psychotic‐like states elicited by psychotomimetics, or induced intentionally by ritual or ascetic practices, can be therapeutic or at least lead to short‐term increases in well‐being.ref. prp21217-bib-0015, ref. prp21217-bib-0016 Nor does it account for the fact that all rapid‐acting antidepressants (psychedelics, ketamine, sleep deprivation) are potent psychotomimetics.ref. prp21217-bib-0013 Or how, over time, an individual can become sensitized to a psychotomimetic stress response, in general or in response to specific cues (e.g., social threat), and thereby more prone to recurring and severe psychotic experiences.ref. prp21217-bib-0017, ref. prp21217-bib-0018, ref. prp21217-bib-0019, ref. prp21217-bib-0020, ref. prp21217-bib-0021 In all these cases we believe that the ideas of psychotomimetic compensation and sensitization provide a parsimonious and falsifiable explanation for these phenomena.
PSYCHOTOMIMETIC COMPENSATION
Arthur Koestler: You must admit that these drugs cause psychosis. A temporary psychosis… Would you say its therapeutic?
Timothy Leary: That’s what the effect should be called. TTP. INSTANT MYSTICISM. Temporary Therapeutic Psychosis.ref. prp21217-bib-0022 pg. 152
People use psychotomimetic drugs for various reasons, but one is to relieve symptoms of pain, anxiety and depression.ref. prp21217-bib-0023, ref. prp21217-bib-0024 It follows that psychotomimetic drugs may “mimic” an endogenous process of psychotomimetic compensation that relieves subjective symptoms of fatigue, pain, anxiety and depression. Indeed, psychotic symptoms (e.g., hallucinations) occur more frequently in contexts of chronic or high stress—for example, during fever,ref. prp21217-bib-0025 sleep deprivation,ref. prp21217-bib-0026 starvation,ref. prp21217-bib-0027 low oxygen conditionsref. prp21217-bib-0028 or socially stressful scenarios that elicit fear or shame.ref. prp21217-bib-0029 Psychotomimetic compensation—mirroring the effects of psychotomimetic drugs—may help normalize metabolic function,ref. prp21217-bib-0030, ref. prp21217-bib-0031 reduce inflammation,ref. prp21217-bib-0032 prevent brain damageref. prp21217-bib-0033 and ameliorate signs and symptoms of injury and illness such as lethargy, behavioral inhibition, social withdrawal, pain, low motivation, sadness and shame (Figure 1).
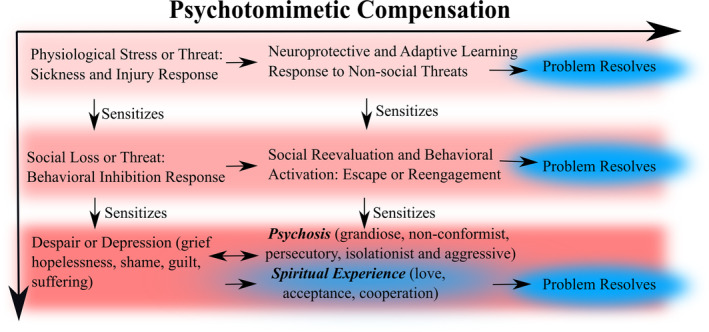
It might not be immediately obvious that a compensatory response to starvation or hypoxia could also compensate against depression, but from an evolutionary perspective, parsimonious explanations are generally preferable. We think psychotomimetic responses to physiological threats and non‐social environmental dangers have been evolutionarily co‐opted to protect against psychosocial threats. Similar explanations have been applied to depression‐like socioemotional responses (e.g., shame, grief, guilt) that appear to co‐opt more basic physiological responses to sickness and injury, such as sadness and lethargy.ref. prp21217-bib-0034 Within this framework, endogenous psychotomimetic signaling not only protects against immediate threat but also compensates against the deleterious effects of enduring sickness and injury responses (e.g., inflammation), social loss and threat, and depression (Figure 1).
PHENOMENOLOGY OF PSYCHOTOMIMETIC COMPENSATION
In order to understand how psychotic symptoms reflect compensatory processes, we must first acknowledge that psychosis reflects an abnormal, extreme, recurring and dysfunctional experiential state. In contrast, a less intense or time‐limited process of psychotomimetic compensation can elicit pleasant experiences that may or may not exhibit mild psychotic‐like or dissociative‐like features depending upon the intensity of stressor and subsequent compensation. For example, many individuals use saunas, exercise, fast or deprive themselves of sleep to increase feelings of wellbeing, and all these practices have antidepressant effects.ref. prp21217-bib-0035, ref. prp21217-bib-0036, ref. prp21217-bib-0037, ref. prp21217-bib-0038 The desired subjective effects of these practices often occur after an initial period of mild discomfort. In the case of exercise, an initial period of unpleasantness, pain, exhaustion, confusion, hunger or tiredness may give way to a “flow”,ref. prp21217-bib-0039 “runners high”ref. prp21217-bib-0040 or “second‐wind”ref. prp21217-bib-0041 type state in which one experiences the desired increase in body‐high, perceptual clarity, creativity, euphoria and energy.
As the duration and intensity of stress increase, the likelihood of florid psychotic and dissociative symptoms increases. One does not run for a minute and achieve a runner’s high, and a mild runner’s high is not likely to elicit the misperceptions and hallucinations that are more common during ultra‐long distance running.ref. prp21217-bib-0042, ref. prp21217-bib-0043 Likewise, the psychotomimetic effects of sleep deprivation become more psychotic‐like over time, evolving from distortions and hallucinations in the visual domain to include auditory hallucinations and delusional ideation.ref. prp21217-bib-0026 The idea that psychotic‐like experiences reflect compensatory processes, and not just dysfunction, is further supported by the fact that the mere threat of physical harm can elicit psychotomimetic compensation. Experiences of time‐expansion during car crashesref. prp21217-bib-0044 and dissociation in the face of other dangers are two examples of this phenomenon.
In the clinical arena, psychotomimetic compensation manifests as a shift away from depressogenic emotions and behaviors towards psychotogenic emotions and behaviors, which helps to explain both the causal relationships and phenomenological distinctions between depression and florid psychosis. Transient depression‐like states can be adaptive, serving as brakes to stop behavioral perseverance in the face of negative feedback such as criticism, social loss, or failure.ref. prp21217-bib-0045
Psychotomimetic compensation helps resolve depression‐like states by promoting reevaluation and reengagement (see Figure 1). This compensatory process appears pathogenically amplified in bipolar mood and psychotic disorders. For example, a sense of connection and meaning is typically lost in depressionref. prp21217-bib-0046 or lacking in premorbid stages of schizophrenia,ref. prp21217-bib-0047 while it is amplified in maniaref. prp21217-bib-0048 and first‐episode psychosis.ref. prp21217-bib-0047 Whereas depression is characterized by negative self‐evaluationsref. prp21217-bib-0049 and reduced self‐serving bias,ref. prp21217-bib-0050 delusional ideation and psychosis are associated with externalizing and self‐serving biases.ref. prp21217-bib-0051, ref. prp21217-bib-0052 By extension, and in accordance with a spectrum perspective on psychotomimetic compensation, a variety of psychotic‐like (e.g., conspiracy, apocalyptic or new‐age) beliefs might function as a schizotypal defense mechanism against uncertainty or social threat.ref. prp21217-bib-0053, ref. prp21217-bib-0054
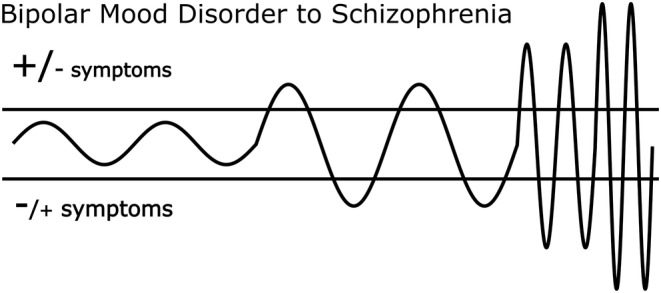
We realize that psychotic depression (PD) could be viewed as an exception to this proposed depression vs mania/psychosis dichotomy, and acknowledge that a lumping together of mania and psychosis may seem problematic, as oftentimes they do not occur together, and when they do cooccur, sometimes psychotic symptoms precede manic symptoms and sometimes manic symptoms precede psychosis. However, various features distinguish PD from psychotic disorders, such as intact insight,ref. prp21217-bib-0055, ref. prp21217-bib-0056 less positive symptoms and less thought disorder.ref. prp21217-bib-0057, ref. prp21217-bib-0058 These distinctions suggest that PD may represent, similar to the prodrome, remission or chronic phase of schizophrenia, a blunted form of psychosis—characterized predominantly by negative symptomsref. prp21217-bib-0058—which in turn may correspond to blunted dissociative or dopaminergic drive (see Figure 3), or in some cases an antecedent phase of illness.
Indeed, individuals with PD are at high risk of converting to schizophrenia and bipolar disorder.ref. prp21217-bib-0059, ref. prp21217-bib-0060 Psychosis is also generally more common and severe in mania than depression, and signs of mania such as increased activity and reduced sleep are common during acute psychotic episodes.ref. prp21217-bib-0061 Regarding the phenomenological relationship between chronic schizophrenia to bipolarity; frequent mood shifts and quick transitions between negative symptoms (associated with depression) and positive symptoms (associated with mania)ref. prp21217-bib-0058 can be observed in some cases of incipient and chronic psychosis.ref. prp21217-bib-0062
Studies have found that variation in positive beliefs about self predict paranoia,ref. prp21217-bib-0063 and that rapid symptom fluctuations predict violence, mediated by anger,ref. prp21217-bib-0064 in the context of first episode psychosis. John Custance,ref. prp21217-bib-0065 pseudonymous author of Wisdom, Folly and Madness, also perceived a similarity in the longer‐duration affective and cognitive shifts in individuals diagnosed with bipolar disorder and similar but more frequent shifts seen in some individuals diagnosed with schizophrenia; shifts which may occur on an hourly or even second by second basis (see alsoref. prp21217-bib-0066). Indeed, our psychotomimetic sensitization model suggests that over time psychotic experiences can become more frequent and severe (see Figure 2).
Quick transitions from negative (tired/distracted) to positive (emotional/visionary) states on the order of seconds to minutes, are also characteristic of the temporal structure of naturally occurring religious/spiritual experiences.ref. prp21217-bib-0067, ref. prp21217-bib-0068 And although illness, despair and grief do often precede these transitionsref. prp21217-bib-0069, ref. prp21217-bib-0070—it would be misleading to interpret religious/spiritual experiences—which are often rare, unique, life events—as signs of mental disorder (although recurring spiritual/religious experiences are also quite commonref. prp21217-bib-0068). Moreover, while we focus here on psychotomimetic compensation as a response to stress—it should be noted that psychotic‐like experiences, or altered states of consciousness (ASCs), also occur during transitions between sleep and wakefulness, and while there are interesting interactions to explore between stress responses and fluctuations in arousalref. prp21217-bib-0071, ref. prp21217-bib-0072—these questions fall beyond the purview of this piece.
PSYCHOPHARMACOLOGY OF PSYCHOTOMIMETIC COMPENSATION
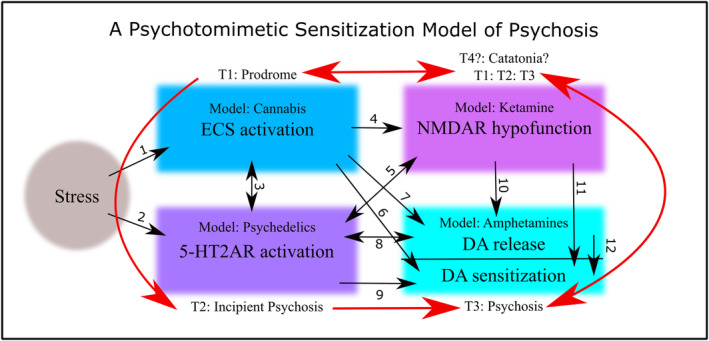
Endogenous psychotomimetic compensation is mediated by the same neurotransmitter/modulator systems engaged by psychotomimetic drugs such as cannabis, psychedelics, dissociative anesthetics, and stimulants (Figure 3). The endocannabinoid system (ECS), for example, modulates responses to homeostatic changes and various stressors,ref. prp21217-bib-0073, ref. prp21217-bib-0074 and mediates adaptations to social defeat.ref. prp21217-bib-0075, ref. prp21217-bib-0076, ref. prp21217-bib-0077, ref. prp21217-bib-0078, ref. prp21217-bib-0079 The ECS is also believed to be responsible for the euphoric runner’s high that occurs during high intensity endurance exercise.ref. prp21217-bib-0080, ref. prp21217-bib-0081
Various stressors also upregulate, sensitize, and activate 5‐HT2A receptors which can have hyperthermic, anti‐inflammatory, antinociceptive and adaptive learning effects (as reviewed in Ref. [ref. 82]; also see [ref. 83]). Preclinical trials show that partial antagonism of NMDA receptors can protect against ischemia,ref. prp21217-bib-0084 hypoglycemia,ref. prp21217-bib-0085 seizure,ref. prp21217-bib-0086 traumatic brain injury,ref. prp21217-bib-0087 or drug‐related toxicityref. prp21217-bib-0088 but timing matters. To be neuroprotective, N‐methyl‐d‐aspartate Receptor (NMDAR) antagonism must occur within a therapeutic time window surrounding the physiological insult.ref. prp21217-bib-0033, ref. prp21217-bib-0089, ref. prp21217-bib-0090
Common acute effects of cannabis, psychedelics, and disassociatives, as well as acute stressors such as exercise, fasting, hyperthermia and psychosocial stress, include a transient increase in brain‐derived neurotrophic factor (BDNF) which is implicated in neuroprotection and neuroplasticity.ref. prp21217-bib-0091, ref. prp21217-bib-0092, ref. prp21217-bib-0093, ref. prp21217-bib-0094, ref. prp21217-bib-0095, ref. prp21217-bib-0096, ref. prp21217-bib-0097 All psychotomimetics and various acute stressors also (typically) increase phasic dopamine release and subsequent DA sensitization, which is crucial to understanding how individuals are cross‐sensitized to psychosis via stress and/or drugs (see Figure 3; and below).
It is beyond the purview of this review to examine, in detail, all the compensatory functions and interactions between relevant neurotransmitter systems (Figure 3). So instead, we will focus on the compensatory potential of one exogenous psychotomimetic agent, classic serotonin (5‐HT) 2A receptor (5‐HT2AR) agonist psychedelics, and one endogenous psychotomimetic agent, kynurenic acid (KYNA).
CLASSIC PSYCHEDELIC COMPENSATION: 5‐HT2AR ACTIVATION
There are clear parallels between the temporal trajectory of the psychedelic experience and the trajectory of endogenous psychotomimetic compensation. Recall that a period of discomfort often precedes a mild dissociative, hypomanic or psychotic‐like “high” (e.g., runners high) in cases of intense physical stress (also see Figure 1). Similarly, a moderate or high dose of psilocybin containing mushrooms often first elicits feelings of nervousness, anxiety, confusion, nausea, being uncomfortable or lethargic.ref. prp21217-bib-0141 After this onset or “comeup” phase, a peak psychedelic experience occurs, followed by what is often described as pleasurable “comedown” and “afterglow”.ref. prp21217-bib-0141, ref. prp21217-bib-0142 Interestingly, the phenomenology of the psychedelic mushroom comeup maps reasonably well onto the illness‐like symptoms reported during fever, hypoglycemia, ketoacidosis, and panic attacks which include nausea, dizziness, confusion, rapid or deep breathing and anxiety.ref. prp21217-bib-0143, ref. prp21217-bib-0144, ref. prp21217-bib-0145 These symptoms are also evident in acute stress reactions and disorders caused by traumatic events.ref. prp21217-bib-0146
One explanation for these phenomenological similarities is that stress‐induced 5‐HT2AR activation functions, in part, to increase access to energy (e.g., stored glucose) and increase ATP synthesis and metabolismref. prp21217-bib-0031, ref. prp21217-bib-0147, ref. prp21217-bib-0148, ref. prp21217-bib-0149 in demanding circumstances, as well as to help buffer the organism against the excitotoxic effects of excessive energy demands or compromised metabolic function.ref. prp21217-bib-0031, ref. prp21217-bib-0150, ref. prp21217-bib-0151
Psychedelic activation of 5‐HT2ARs promotes mitochondrial biogenesis,ref. prp21217-bib-0030, ref. prp21217-bib-0152, ref. prp21217-bib-0153 resulting in increased expression of antioxidant enzymes, decreased cellular reactive oxygen species, and enhanced neuroprotection against excitotoxic and oxidative stressref. prp21217-bib-0030 (also see Ref. [ref. 31] for review). Consistent with these effects, the psychedelic agent N,N‐dimethyltryptamine (DMT) reduces infarct size and improves functional recovery following experimentally‐induced cerebral ischemia in rodents and psychedelics more generally are under development for stroke, as well as neurodegenerative conditions such as Alzheimer’s disease.ref. prp21217-bib-0154
In regard to how metabolic stress or energy deficits trigger a psychedelic‐like serotonergic response, it has been proposed that chemosensitive serotonin neurons in the brainstem are responsive to changes in pH,ref. prp21217-bib-0155, ref. prp21217-bib-0156 which is an expected consequence of intense exercise,ref. prp21217-bib-0157 hypoxia,ref. prp21217-bib-0158 inflammation,ref. prp21217-bib-0159 ketosisref. prp21217-bib-0160 and other states linked to psychotomimetic compensation such as near‐death experiences (NDEs). Some scholars point to endogenous psychedelic release during NDEs,ref. prp21217-bib-0161 which is also consistent with hypoxia‐induced rerouting of tryptophan metabolism towards increased tryptamine production,ref. prp21217-bib-0162 and the fact that NDEs occurring due to cardiac arrest are associated with hypoxia and increased end‐tidal carbon dioxide (CO2) levels.ref. prp21217-bib-0163
There is some debate as to the similarity of NDEs and psychedelic experiences,ref. prp21217-bib-0164, ref. prp21217-bib-0165 but the visionary and emotional characteristics and effects of these experiences are quite similarref. prp21217-bib-0166, ref. prp21217-bib-0167, ref. prp21217-bib-0168 as are their temporal trajectories. A substantial subset of distressing NDEs are characterized at first by panic and fear, but subsequently turn into positive experiences of calm, peace and love.ref. prp21217-bib-0169 A similar phenomenological transition from panic to peace is described in experiences of drowning.ref. prp21217-bib-0170 It is noteworthy in this regard that the 5‐HT2AR mediates CO2 induced arousal,ref. prp21217-bib-0171 and that CO2 inhalation elicits feelings of anxiety and panicref. prp21217-bib-0172, ref. prp21217-bib-0173 as well as (less‐well‐known) cathartic, pleasurable and visionary experiences.ref. prp21217-bib-0174, ref. prp21217-bib-0175 One of the possible negative consequences of psychedelic use is also an increase in anxiety and panic.ref. prp21217-bib-0176, ref. prp21217-bib-0177
Psychotomimetic responses to respiratory and metabolic stress map onto the symptoms and phenomenological trajectories of psychotic disorders as well. Panic attacks elicit mental imagery, strong emotions and a sensed loss of controlref. prp21217-bib-0144—subjective features that relate to psychedelic and incipient psychotic experiences. Indeed, anxiety and panic often precede first‐episode psychosis,ref. prp21217-bib-0178, ref. prp21217-bib-0179 and panic symptoms are common and severe in psychotic disorders.ref. prp21217-bib-0178 Additionally, both psychosis and mania are linked to low brain pHref. prp21217-bib-0180, ref. prp21217-bib-0181, ref. prp21217-bib-0182 (although see Ref. [ref. 183])—which, as mentioned above—may be a common trigger of psychotomimetic compensation, whether elicited by changes in respiration or metabolism. The putative links between respiratory and metabolic crises and psychotomimetic compensation could help explain some of the paradoxical links between respiratory, metabolic and psychotic disorders; for example, how it is that respiratory and metabolic disorders increase the risk of psychosis, while treating psychosis with antipsychotics further increases risk of respiratory and metabolic disorders.ref. prp21217-bib-0184, ref. prp21217-bib-0185
Christopher Palmerref. prp21217-bib-0151 convincingly argues that psychiatric disorders including schizophrenia and bipolar disorder are caused in part by compromised metabolic function, and it is possible that psychotic and manic symptoms result, in part, from an attempt to compensate against metabolic stress.ref. prp21217-bib-0186 Although in need of replication, an older study by Kitay and Altschuleref. prp21217-bib-0187 found that 40% of individuals hospitalized with psychosis had ketone levels—a primary alternative fuel sources for humans –above the upper limit seen in controls. Other studies have found increased lactate levels—another potent alternative fuel sourceref. prp21217-bib-0188—in schizophrenia and bipolar disorders.ref. prp21217-bib-0180, ref. prp21217-bib-0189
Although atypical antipsychotics (5‐HT2AR blockers) and mood stabilizers help treat the symptoms of psychosis and mania, they often contribute to metabolic dysfunction and related health issues such as diabetes and obesity.ref. prp21217-bib-0185, ref. prp21217-bib-0190 Conversely, preliminary data suggests ketogenic diets may be useful in treating psychiatric disorders.ref. prp21217-bib-0191, ref. prp21217-bib-0192, ref. prp21217-bib-0193 One explanation for the putative antipsychotic and antiepileptic effects of ketogenic diet is that, by providing access to a viable alternative fuel source, or by promoting stable increases in neuroprotective KYNA,ref. prp21217-bib-0194, ref. prp21217-bib-0195, ref. prp21217-bib-0196, ref. prp21217-bib-0197, ref. prp21217-bib-0198 the ketogenic diet dampens phasic psychotomimetic compensation mediated, for example, by activation of 5‐HT2ARs. It follows theoretically that it may be the bipolar fluctuations between depressive and manic symptoms, or negative and positive symptoms, or between abstinence and binge drug use, that contributes to bipolar, psychotic and substance abuse disorders.
Having touched upon psychotomimetic compensations to physiological stress, let us now turn towards the socioemotional trajectory of psychotomimetic compensation—as modeled by psychedelics. Psychedelics often elicit and then resolve stressful or challenging experiences, a process that is associated with long‐term increases in well‐being.ref. prp21217-bib-0199 Despite a dearth of research on the temporal trajectory of the psychedelic experience,ref. prp21217-bib-0141 descriptions of emotional breakthroughs,ref. prp21217-bib-0199 catharsis, revised life priorities,ref. prp21217-bib-0200 renewed senses of purpose, heroic doses and journeys, and death‐rebirth experiencesref. prp21217-bib-0201 all suggest that these experiences have the temporal structure of a “problem(T1)‐solving(T2) experience” that has been related to spiritual and acute psychotic experiences in the past.ref. prp21217-bib-0202
The transition from an uncomfortable psychedelic comeup to a relieving comedown also maps reasonably well onto relevant clinical transitions from depression to mania, and from psychotic prodrome to first‐episode psychosis.ref. prp21217-bib-0067 In subjective reports of transitions from depression/prodrome towards mania/psychosis individuals often first report feeling better, temporarily, but then experience increased distress associated with enduring hyperactive or psychotic experiences.ref. prp21217-bib-0047, ref. prp21217-bib-0203 We notice a similar distress when psychedelic mushroom experiences endure longer than hoped for, leading to exhaustion, further panic, or the inability to sleep or return to a comfortable baseline.ref. prp21217-bib-0141
One risk associated with the antidepressant use of psychotomimetic drugs, sleep deprivation, fasting or ketogenic diet is the possibility of triggering a switch towards mania and psychosis.ref. prp21217-bib-0204, ref. prp21217-bib-0205, ref. prp21217-bib-0206 There are plenty of examples, however, when compensatory switches—even ones that resemble mania or psychosis—do not lead to progressive dysregulation and dysfunction. The transition from initial shamanic sickness to shamanic death‐rebirth experience and shamanic electionref. prp21217-bib-0207 is one example, as are innumerable other types of salvific, spiritual or secular self‐transformative experiences.ref. prp21217-bib-0068, ref. prp21217-bib-0082
PSYCHOTOMIMETIC INTERACTIONS
The fact that we observe a switch from an aversive illness‐like state to a state of relief in the psychedelic mushroom experienceref. prp21217-bib-0141 suggests that interactions and downstream contributions from non‐serotonergic NT systems influence the trajectory of psychedelic experiences (see Figure 3 for references and reviews). Building on the ideas of Freedman,ref. prp21217-bib-0208 Marona‐Lewicka and colleaguesref. prp21217-bib-0209 suggest that there are two distinct temporal phases to the LSD experience, the first psychedelic and mediated by 5‐HT2AR activation and the second manic/paranoid and mediated by dopamine (DA) signaling.
Although LSD has more affinity for dopamine receptors than some other psychedelics like psilocybin, Vollenweider and colleagues,ref. prp21217-bib-0008 found that DA subtype 2 receptor blockade reduced oceanic boundlessness, increased dread of ego‐dissolution, and had no effect on visionary restructuralization in a group of healthy volunteers given psilocybin.ref. prp21217-bib-0008 In another study haloperidol (D2R antagonist) pretreatment led to increased death in rodents given LSD.ref. prp21217-bib-0210 Thus, it appears that dopamine signaling may play an important role in all psychedelic experiences, perhaps particularly in the transition from uncomfortable comeup (associated with rising 5‐HT2AR activation) towards peak experience, emotional breakthrough and pleasurable come‐down.
Similarly, regional alterations in glutamate are implicated in both negative and positive experiences of ego‐dissolution under psilocybin.ref. prp21217-bib-0211 It has recently been shown that concurrent use of lithium—with putative effects on DA and NMDAR neurotransmission—drastically increases risk of seizures and bad trips on psychedelics.ref. prp21217-bib-0212 Taken together, this all suggests that the positive effects of psychedelic 5‐HT2AR agonism are somewhat dependent upon interactions with other NT systems. Moreover, it is possible, perhaps even likely, that the psychotomimetic engagement of other NT systems (see Figure 2) exert counterbalancing effects on 5‐HT2AR agonism. For example, psychedelics increase body temperature,ref. prp21217-bib-0213 while cannabinoid receptor agonists,ref. prp21217-bib-0214 D2R agonistsref. prp21217-bib-0215 and NMDAR antagonists all reduce body temperature.ref. prp21217-bib-0216
The complex interplay of NT systems involved in psychotomimetic compensation (Figure 2), as alluded to by the limited number examples provided above, gives us a preliminary sense of why psychotic‐like experiences occur in contexts of stress and energy deficits, and why, although antipsychotics can help treat symptoms of psychosis, they can also contribute to weight gain,ref. prp21217-bib-0217 metabolic disorder,ref. prp21217-bib-0218, ref. prp21217-bib-0219 susceptibility to heat stressref. prp21217-bib-0220 and related life‐threatening conditions such as neuroleptic malignant syndrome.ref. prp21217-bib-0221
KYNURENIC ACID: ENDOGENOUS NMDAR ANTAGONISM
Elevated levels of glutamate can lead to excitotoxicity via NMDA receptor activation, Ca2+ influx, oxidative stress and mitochondrial dysfunction, a process which may be protected against, to some extent, by a compensatory increase in brain kynurenic acid (KYNA); an NMDA receptor antagonist.ref. prp21217-bib-0222 Animal models also show that KYNA increases energy expenditure and improves metabolic function.ref. prp21217-bib-0223
In line with our psychotomimetic compensation model, increased neurotoxic quinolinic acid (an NMDA receptor agonist) and decreased KYNA is seen in depression,ref. prp21217-bib-0224, ref. prp21217-bib-0225 while increased levels of KYNA is associated with mania and psychosisref. prp21217-bib-0226, ref. prp21217-bib-0227, ref. prp21217-bib-0228, ref. prp21217-bib-0229, ref. prp21217-bib-0230 (although see Ref. [ref. 231, ref. 232]). Stress‐induced increases in salivary KYNA are elevated in individuals diagnosed with schizophrenia,ref. prp21217-bib-0233, ref. prp21217-bib-0234 and increased levels of KYNA are associated with deficits in pre‐pulse inhibiton and audio‐sensory gating in animal models of schizophrenia (see Ref. [ref. 228] for review).
The belief that endogenous antagonism of NMDARs plays a role in the pathophysiology of schizophreniaref. prp21217-bib-0235 is supported by the fact that exogenous NMDAR antagonism, via ketamine for example, is a good pharmacological model of the negative and positive symptoms of psychosis.ref. prp21217-bib-0236 Ketamine, however, is also a rapid acting antidepressant.ref. prp21217-bib-0013 Other conventional antidepressants,ref. prp21217-bib-0237 and non‐pharmacological interventions for depression such as exercise,ref. prp21217-bib-0238 sleep deprivationref. prp21217-bib-0239, ref. prp21217-bib-0240 and fastingref. prp21217-bib-0241 also increase levels of KYNA. Although not known at the time of writing, a clear prediction is that, like ketamine,ref. prp21217-bib-0242 classic psychedelics (e.g., LSD, psilocybin) will also increase central nervous system levels of neuroprotective kynurenic acid (KYNA).
PSYCHOTOMIMETIC SENSITIZATION
Functional psychotic disorders like schizophrenia are increasingly conceived of as neurodevelopmental disorders, but accumulation of life stress or drug abuse is still very much implicated in their development.ref. prp21217-bib-0243 Figure 3 (see above) presents a unified (albeit simplified) model of how predisposed individuals can be sensitized to psychosis over‐time via recurrent stress, recurrent drug use or both, all via shared pathways. From a developmental perspective the idea of psychotomimetic sensitization fits observations that first episodes of psychosis develop much more slowly than recurrent episodes.ref. prp21217-bib-0244 The initial schizophrenia prodrome can last years, and manic psychoses often take years to manifest after the onset of depressive symptoms.ref. prp21217-bib-0059 In contrast, subsequent psychotic episodes emerge swiftly and with few warning signs.ref. prp21217-bib-0244
Psychotic disorders are highly heritable, but this fact does not contradict the model presented here, which highlights the etiological role played by stress and psychotomimetic drug exposure. Developmental vulnerability to psychosis in the absence of repeated drug use suggests an endogenous psychotomimetic process that is most likely facilitated by chronic or high‐dose stress during critical developmental windows.ref. prp21217-bib-0082, ref. prp21217-bib-0243 Prenatal and early life stress predisposes individuals to psychosis, and individuals with schizophrenia show increased dopamine release in response to stress, an effect which is mediated by the 5‐HT2AR ref. prp21217-bib-0245, ref. prp21217-bib-0246, ref. prp21217-bib-0247 and primed via activation of the ECS.ref. prp21217-bib-0129, ref. prp21217-bib-0130 Dopaminergic sensitization develops in response to and then eventually reinforces psychotomimetic signaling, producing a self‐perpetuating feedback loop.
A single administration of a dopamine‐enhancing drug does not typically induce the psychotic‐like experiences that are elicited by other psychotomimetics.ref. prp21217-bib-0248, ref. prp21217-bib-0249 Chronic and high doses of dopamine‐enhancing drugs do induce psychotic‐like phenomena, alter NMDA receptor functioning and expression,ref. prp21217-bib-0248, ref. prp21217-bib-0249, ref. prp21217-bib-0250 and increase 5‐HT2AR sensitivity and activity.ref. prp21217-bib-0251, ref. prp21217-bib-0252, ref. prp21217-bib-0253 Antagonizing 5‐HT2ARs in turn has antipsychotic effects in patients with schizophrenia and those receiving L‐DOPA for Parkinson’s disease.ref. prp21217-bib-0254, ref. prp21217-bib-0255 These synergisms begin to reveal how psychotic experiences emerge, are reinforced, and sensitized over time via the same processes implicated in learning and addiction and that are implicated in the psychotomimetic effects of psychedelics. What at first can be a benign or positive response to stress or drugs, can over‐time begin to resemble a dysfunctional disorder that is beyond the control of the individual.
Acknowledging that a susceptibility to psychosis develops over time; psychosis itself is much more likely to manifest when—borrowing terminology from psychedelics—a person’s set (mindset) and setting (social context) are negative.ref. prp21217-bib-0256 Indeed, negative mindsets are associated with mental phenomena seen in psychosis, including jumping to conclusions,ref. prp21217-bib-0257 delusions and hallucinations.ref. prp21217-bib-0258 In regard to social context, a threat to or loss of social status is also believed to make people more vulnerable schizophrenia.ref. prp21217-bib-0020, ref. prp21217-bib-0259, ref. prp21217-bib-0260, ref. prp21217-bib-0261, ref. prp21217-bib-0262 The same “set and setting” context‐dependency holds true for drug‐induced psychotic‐like experiences.
Experiences of childhood trauma increase psychotic symptoms elicited by amphetamines,ref. prp21217-bib-0263 neuroticism predicts challenging experiences with psychedelicsref. prp21217-bib-0264 and psychedelic use in emotionally alienating, non‐supportive environments increases the risk of problematic psychotic‐like (often paranoid) reactions. Thus, while we have been discussing sensitization to psychotomimetic compensation in the broader sense, there is a case to be made that specific symptoms of psychosis—e.g., derogatory auditory verbal hallucinations—reflect a sensitization to specific stimuli (e.g., social cues) and emotions (e.g., shame).ref. prp21217-bib-0029 Nevertheless, there are also noticeable trends that support a broader conception of psychotomimetic sensitization, such as a general convergence towards paranoia in emerging psychotic disorders, or the increased preponderance of auditory hallucinations and delusions with extended time awake in sleep deprivation models of psychosis.ref. prp21217-bib-0026
Therefore, while we agree with the consensus that psychedelic experiences tend to be positive when set and setting are positive, we note that a process of psychotomimetic sensitization could still increase susceptibility to psychotic‐like experiences in negative future contexts; with younger and more frequent poly‐substance users being at greater risk.
DO PSYCHEDELICS INCREASE THE RISK OF PSYCHOSIS?
It is beyond the purview of this piece to address risk factors associated with each commonly used psychotomimetic agent. However, links between cannabis use and psychosis have been firmly established.ref. prp21217-bib-0009, ref. prp21217-bib-0265, ref. prp21217-bib-0266 It is also generally accepted that chronic high‐dose methamphetamine use increases the risk of psychosis.ref. prp21217-bib-0267 Less attention has been directed at the risks associated with the use of subanesthetic doses of dissociative anesthetics such as ketamine and use of classic psychedelics.
A longitudinal study of 2588 adolescents in Munich, Germany found that use of psychedelics was positively associated with lifetime experience of psychotic symptoms.ref. prp21217-bib-0268 More recently, two large‐scale, population‐based studies report that prior year use of LSD predicts increased depression, suicidal ideation, and presence of any/serious mental health condition.ref. prp21217-bib-0268, ref. prp21217-bib-0269 Conversely, Krebs and Johansenref. prp21217-bib-0270 report that lifetime use of psychedelics is not an independent risk factor for mental health problems or psychotic symptoms when other risk factors are taken into account. However, the unadjusted data in Krebs and Johansenref. prp21217-bib-0270 shows double the rates of psychotic symptoms among people who have ever used psychedelics (see fig. 3 of Ref. [ref. 270, ref. 271]; 50). Survey data available to the public also show that individuals in the USA who have ever used hallucinogens (which according SAMSHA operationalization includes classic psychedelics, ecstasy/MDMA and dissociatives) are about twice as likely to have received inpatient mental health treatment within the last year prior to survey (data visualization available at https://pdas.samhsa.gov/#/).
Of course, this increased risk could be due to other confounding variables associated with psychedelic use, such as risk‐taking, or other substance use, and there is some debate over whether the use of other controlled substances, including cannabis, should be controlled for in analyses of risks associated with psychedelics.ref. prp21217-bib-0271, ref. prp21217-bib-0272 According to data from Krebs and Johansen,ref. prp21217-bib-0270 98% percent of people who had ever used psychedelics also tried cannabis. Although it has been argued that drawing conclusions from the very niche population that only uses psychedelics has limited generalizability, data from such a study might provide information essential for estimating the actual effect size of the direct risk from psychedelic use. However, given the known influences of set and setting on the psychedelic experience, and our proposed psychotomimetic sensitization model, which highlights relevant interactions between psychotomimetic drug effects, it may not always be appropriate to isolate the effects of psychedelics from contextual confounds, or to only report/highlight adjusted odds ratios. At the very least, caution needs to be taken when communicating information to the public. As Johnstadref. prp21217-bib-0272 suggests, widespread adoption of psychedelics on a society‐wide level could promote a scenario in which occasional psychedelic use increases quality of life in already well‐adjusted individuals and promotes dysfunction when used frequently or in more negative contexts.
Controlling for mental illness among users of psychedelics is also a debatable analytic strategy, particularly if we entertain the possibility that psychedelic use could increase susceptibility to psychosis, or that other mental health factors could influence the effects of psychedelic use. The adage that psychedelics only cause psychosis in vulnerable individuals avoids the issue, considering that the number of vulnerable individuals is far greater than the number of people who go on to develop a psychotic disorder.
Within the USA the most common ages to first use hallucinogens (16–18) and cannabis (15–18) precede the typical age of onset of schizophrenia‐spectrum disorders,ref. prp21217-bib-0273 supporting the possibility of a causal effect (data visualization available at https://pdas.samhsa.gov/#/). The number of people reporting psychotic or other adverse reactions to psychedelics is admittedly low,ref. prp21217-bib-0274 but may be underreported, and different methods of collecting data (e.g., internet survey, home interviews, patient data) might generate pictures of relative risk different from those obtained from carefully controlled and psychotherapeutically‐supported clinical trials. Interviews of individuals referred to early intervention services for first‐episode psychosis, for example, reveal rates of hallucinogen use that are much higher than that observed in the general population.ref. prp21217-bib-0275, ref. prp21217-bib-0276
A recent meta‐analysis suggests that the transition to schizophrenia occurs in 26% of hallucinogen‐induced psychoses, compared to 34% and 22% of psychoses induced by cannabis and amphetamines, respectively.ref. prp21217-bib-0010 These transition rates are comparable to transition rates from brief and atypical psychoses, and psychoses not otherwise specified (36%), but are higher than transition rates from other types of drug‐induced psychosis (e.g., from alcohol or opioids).ref. prp21217-bib-0010 However, other variables, such as age, may account for some of these differences.ref. prp21217-bib-0010 Kendler et al.ref. prp21217-bib-0277 found that the likelihood of conversion to schizophrenia does decrease with the age of first substance‐induced psychosis, and that the mean time to schizophrenia conversion after substance‐induced psychosis is 39 months. These observations roughly conform to Boutros et al.ref. prp21217-bib-0278 observations that peak substance related admissions to Connecticut (USA) state hospitals during the late 1960s and early 1970s preceded a sharp rise in psychotic and mood disorder diagnoses some five years later.
In summary, current evidence suggests that we should not prematurely dismiss the psychotogenic risks posed by psychedelics, particularly within populations most likely to use these agents outside of clinical contexts, both now and in all possible futures. The possibility of psychotomimetic compensation and sensitization provide additional reasons for being cautious when trying to assess independent risks associated with psychedelic use. Models of risk must be grounded in theory, and there are theoretical reasons to suspect that adverse life events, other substance use, and mental health factors will interact with psychedelic use in determining the risk of psychosis or other mental health outcomes.
Nomenclature of targets and ligands
Key protein targets and ligands in this article are hyperlinked to corresponding entries in http://www.guidetopharmacology.org, the common portal for data from the IUPHAR/BPS Guide to PHARMACOLOGY,ref. prp21217-bib-0284 and are permanently archived in the Concise Guide to PHARMACOLOGY 2019/20.ref. prp21217-bib-0285
CONCLUSIONS, IMPLICATIONS, AND LIMITATIONS
Psychedelics elicit positive psychological and spiritual experiences as well as experiences that resemble psychosis. We explain this apparent paradox by appealing to the process of psychotomimetic compensation, which is generally therapeutic at short durations and infrequent intervals but can become pathogenic when chronic or frequent via a process of psychotomimetic sensitization. This model is generally consistent with the hormetic effects of short‐duration stressorsref. prp21217-bib-0279 and the idea of canalization—the entrenchment and stabilization of thought and behavior—as applied to psychopathology.ref. prp21217-bib-0280 Use of psychedelics is associated with an increased risk of psychotic symptom development in the general population, although any attempt to isolate the risk ascribable to psychedelics alone poses theoretical problems. Individuals prone to psychosis are also more likely to use psychotomimetics,ref. prp21217-bib-0281, ref. prp21217-bib-0282 questioning the directionality of any association, although a compensatory use of psychotomimetics, as a form of coping, would be consistent with our framework. A few practical implications follow. First, we see no reason to prematurely dismiss a psychotomimetic perspective on psychedelics. This perspective, if approached in a balanced way, might help us understand and mitigate the risks associated with the therapeutic and recreational use of psychedelics, as well as to understand psychosis.
There are also limitations to our analysis, and potential risks associated with pathologizing and suppressing compensatory psychotomimetic processes. By highlighting the importance of the duration, frequency, and intensity of psychotomimetic experiences, we limit consideration of other context‐specific factors that influence the form and outcome of these experiences, such as controllability, aesthetics and social environment. In some cases, controlling the contexts in which psychotomimetic experiences occur will yield more benefits than trying to suppress the emergence of these experiences altogether, which could unintentionally favor the emergence of psychotic symptoms during periods of inattention, reduced control and decreased social support. Shamanic cures to psychotic‐like developmental crises are found all over the world in forager cultures with few class or political distinctions, and are achieved by gaining a mastery over (perceived) spirits that can otherwise persecute and destroy the individual.ref. prp21217-bib-0207 Shamanic rituals share many parallels with common and ideal “settings” for recreational and therapeutic use of psychedelics.ref. prp21217-bib-0201 Aesthetic manipulations (eyeshades, music, visualization techniques) favor the emergence of visual and multimodal as opposed to auditory hallucinations, and repetitive movements (dancing) and collective rituals contribute to feelings of positive emotion and control. Social environment and preparation might likewise be leveraged to promote prosocial as opposed to antisocial attitudes and beliefs, although addressing the relationship between values and mental health raises complex ethical and methodological hurdles.
Our presentation of psychotomimetic sensitization is further limited in two important ways. First, we do not address the importance of periodic motivational and attentional deficits in promoting the psychotomimetic sensitization process, which is consistent with how sensitization to drugs occurs, via periodic abstinence and binges.ref. prp21217-bib-0139 Second, mechanisms unrelated or only peripherally related to sensitization also contribute to psychotic disorder. An alternative explanation to our sensitization model would be that increased psychotomimetic compensation over time simply reflects increased brain or metabolic dysfunctionref. prp21217-bib-0014, ref. prp21217-bib-0151—although we cannot discount the possibility that such dysfunction would contribute to a process of sensitization—in fact we expect that it would. These considerations, along with those mentioned in the previous paragraph do not support the idea of a clear linear relationship between number of psychotomimetic experiences and pathology. It remains to be seen whether controlled engagement with psychotomimetic experiences, for example via psychospiritual practices or the therapeutically‐constrained use of psychedelics, could reduce symptoms of psychosis or the distress associated with these symptoms.ref. prp21217-bib-0283
In conclusion, psychotomimetic experiences are relatively normal, fall on a spectrum, and function as compensations against stressors of all sorts. It follows that they are neither abnormal nor miraculous experiences, and should not be categorially suppressed or promoted, but rather engaged with more neutrally in accordance with the specific needs of individuals and communities. The suitability of personal solutions to environmental challenges cannot and should not be reduced to simple, context‐independent, biocentric explanations.
AUTHOR CONTRIBUTIONS
AB and CLR wrote the manuscript. AB, CLR, and RCH revised the manuscript.
FUNDING INFORMATION
Funding was provided to CLR via the Mary Sue and Mike Shannon Distinguished Chair for Healthy Minds, Children and Families and to AB via an academic scholarship from Usona Institute. The funders had no role in study design; in the collection, analysis, and interpretation of data; in the writing of the report; or in the decision to submit the article for publication.
ETHICS STATEMENT
Ethics approval was not required for this manuscript.
References
- Psychedelics and schizophrenia.. Trends Neurosci., 2009. [PubMed]
- The psychotomimetic effects of intravenous delta‐9‐tetrahydrocannabinol in healthy individuals: implications for psychosis.. Neuropsychopharmacology., 2004. [PubMed]
- Subanesthetic effects of the noncompetitive NMDA antagonist, ketamine, in humans: psychotomimetic, perceptual, cognitive, and neuroendocrine responses.. Arch Gen Psychiatry., 1994. [PubMed]
- Dose‐related psychotic symptoms in chronic methamphetamine users: evidence from a prospective longitudinal study.. JAMA Psychiatry., 2013. [PubMed]
- A review of the clinical effects of psychotomimetic agents.. Ann N Y Acad Sci., 1957. [PubMed]
- An overview of animal models related to schizophrenia.. Can J Psychiatr., 2019
- The psychotomimetic states inventory (PSI): measuring psychotic‐type experiences from ketamine and cannabis.. Schizophr Res., 2008. [PubMed]
- Psilocybin induces schizophrenia‐like psychosis in humans via a serotonin‐2 agonist action.. Neuroreport., 1998. [PubMed]
- Cannabis and psychosis: recent epidemiological findings continuing the “causality debate”.. Am J Psychiatry., 2022. [PubMed]
- Transition of substance‐induced, brief, and atypical psychoses to schizophrenia: a systematic review and meta‐analysis.. Schizophr Bull., 2020. [PubMed]
- Efficacy and safety of therapeutic use of cannabis derivatives and their synthetic analogs: overview of systematic reviews.. Phytother Res., 2022. [PubMed]
- The pharmacology of amphetamine and methylphenidate: relevance to the neurobiology of attention‐deficit/hyperactivity disorder and other psychiatric comorbidities.. Neurosci Biobehav Rev., 2018. [PubMed]
- A continuum hypothesis of psychotomimetic rapid antidepressants.. Brain Neurosci Adv., 2021. [PubMed]
- Psychotic experiences as a health indicator: a provisional framework.. Int J Soc Psychiatry., 2022. [PubMed]
- Effects of naturalistic psychedelic use on depression, anxiety, and well‐being: associations with patterns of use, reported harms, and transformative mental states.. Front Psych., 2022
- Effects of extreme ritual practices on psychophysiological well‐being.. Curr Anthropol., 2019
- Paranoia, sensitization and social inference: findings from two large‐scale, multi‐round behavioural experiments.. R Soc Open Sci., 2020. [PubMed]
- Does the concept of “sensitization” provide a plausible mechanism for the putative link between the environment and schizophrenia?. Schizophr Bull., 2008. [PubMed]
- Behavioural sensitization to daily life stress in psychosis.. Psychol Med., 2005. [PubMed]
- Experimentally induced social threat increases paranoid thinking.. R Soc Open Sci., 2018. [PubMed]
- Psychosocial stress and psychosis. A review of the neurobiological mechanisms and the evidence for gene‐stress interaction.. Schizophr Bull., 2008. [PubMed]
- 22 Leary T . High priest. New American Library; 1968.
- Motives for the use of serotonergic psychedelics: a systematic review.. Drug Alcohol Rev., 2022. [PubMed]
- Cannabis for therapeutic purposes: patient characteristics, access, and reasons for use.. Int J Drug Policy., 2013. [PubMed]
- Fever and acute brief psychosis in urban and rural settings in north India.. Br J Psychiatry., 1999. [PubMed]
- Severe sleep deprivation causes hallucinations and a gradual progression toward psychosis with increasing time awake.. Front Psych., 2018
- Eating disorders and psychosis: seven hypotheses.. World journal of psychiatry., 2014. [PubMed]
- Isolated psychosis during exposure to very high and extreme altitude–characterisation of a new medical entity.. Psychol Med., 2018. [PubMed]
- Is shame hallucinogenic?. Front Psychol., 2017. [PubMed]
- Serotonin regulates mitochondrial biogenesis and function in rodent cortical neurons via the 5‐HT2A receptor and SIRT1–PGC‐1α axis.. Proc Natl Acad Sci., 2019. [PubMed]
- Effects of serotonergic psychedelics on mitochondria: transdiagnostic implications for mitochondria‐related pathologies.. J Psychopharmacol., 2023. [PubMed]
- Psychedelics as anti‐inflammatory agents.. Int Rev Psychiatry., 2018. [PubMed]
- Therapeutic time windows of compounds against NMDA receptors signaling pathways for ischemic stroke.. J Neurosci Res., 2021b. [PubMed]
- The evolutionary significance of depression in Pathogen Host Defense (PATHOS‐D).. Mol Psychiatry., 2013. [PubMed]
- Fasting interventions for stress, anxiety and depressive symptoms: a systematic review and meta‐analysis.. Nutrients., 2021. [PubMed]
- The impact of whole‐body hyperthermia interventions on mood and depression–are we ready for recommendations for clinical application?. Int J Hyperthermia., 2019
- Sleep deprivation as treatment for depression: systematic review and meta‐analysis.. Acta Psychiatr Scand., 2021. [PubMed]
- The role of exercise in the treatment of depression: biological underpinnings and clinical outcomes.. Mol Psychiatry., 2023. [PubMed]
- Flow states in exercise: a systematic review.. Psychol Sport Exerc., 2019
- The runner’s high revisited: a phenomenological analysis.. J Phenomenol Psychol., 2016
- The effect of external temperature on second wind.. American Journal of Physiology‐Legacy Content., 1926
- Misperceptions and hallucinatory experiences in ultra‐trailer, high‐altitude runners.. Riv Psichiatr., 2020. [PubMed]
- Visual hallucinations in 246‐km mountain ultra‐marathoners: an observational study.. Chin J Physiol., 2021. [PubMed]
- When seconds turn into minutes: time expansion experiences in altered states of consciousness.. J Humanist Psychol., 2022
- The bright side of being blue: depression as an adaptation for analyzing complex problems.. Psychol Rev., 2009. [PubMed]
- Understanding anhedonia: a qualitative study exploring loss of interest and pleasure in adolescent depression.. Eur Child Adolesc Psychiatry., 2020. [PubMed]
- The lived experience of psychosis: a bottom‐up review co‐written by experts by experience and academics.. World Psychiatry., 2022. [PubMed]
- Toward a refined phenomenology of mania: combining clinician‐assessment and self‐report in the French EPIMAN study.. J Affect Disord., 2001. [PubMed]
- Shame, guilt, and depressive symptoms: a meta‐analytic review.. Psychol Bull., 2011. [PubMed]
- Attributional style in depression: a meta‐analytic review.. J Pers Soc Psychol., 1986. [PubMed]
- Cognitive biases in individuals with psychotic‐like experiences: a systematic review and a meta‐analysis.. Schizophr Res., 2020. [PubMed]
- A comprehensive meta‐analysis of the self‐serving bias in schizophrenia spectrum disorders compared to non‐clinical subjects.. Neurosci Biobehav Rev., 2021. [PubMed]
- Personality and cognitive predictors of New Age practices and beliefs.. Personal Individ Differ., 2005
- Belief in conspiracy theories: the predictive role of schizotypy, Machiavellianism, and primary psychopathy.. PLoS One., 2019. [PubMed]
- Insight into illness in patients with mania, mixed mania, bipolar depression and major depression with psychotic features.. Bipolar Disord., 2002. [PubMed]
- The association of insight with psychotic symptoms, depression, and cognition in early psychosis: a 3‐year follow‐up.. Schizophr Res., 2007. [PubMed]
- Validity of subtyping psychotic depression: examination of phenomenology and demographic characteristics.. Am J Psychiatry., 1988. [PubMed]
- Phenomenology of first‐episode psychosis in schizophrenia, bipolar disorder, and unipolar depression: a comparative analysis.. Clin Schizophr Relat Psychoses., 2012. [PubMed]
- Diagnostic conversion from unipolar depression to bipolar disorder, schizophrenia, or schizoaffective disorder: a nationwide prospective 15‐year register study on 43 495 inpatients.. Bipolar Disord., 2020. [PubMed]
- Diagnostic shifts during the decade following first admission for psychosis.. Am J Psychiatry., 2011. [PubMed]
- The phenomenology of mania: a new look at some old patients.. Arch Gen Psychiatry., 1973. [PubMed]
- Moving from static to dynamic models of the onset of mental disorder: a review.. JAMA Psychiatry., 2017. [PubMed]
- The negative and positive self: a longitudinal study examining self‐esteem, paranoia and negative symptoms in individuals with first‐episode psychosis.. Early Interv Psychiatry., 2011. [PubMed]
- Capturing the ebb and flow of psychiatric symptoms with dynamical systems models.. Am J Psychiatry., 2009. [PubMed]
- 65 Custance J . Wisdom, madness, and folly: the philosophy of a lunatic. Pellegrini & Cudahy; 1952.
- 66 Podvoll EM . The seduction of madness: a revolutionary approach to the recovery at home. Century Press; 1991.
- The trajectory of psychedelic, spiritual, and psychotic experiences: implications for cognitive scientific perspectives on religion. [Under Review].. 2023b
- From suffering to salvation: making sense of religious experiences. [Manuscript in prep].. 2023c
- 69 Hardy AC . The spiritual nature of man: a study of contemporary religious experience. Clarendon; 1979.
- 70 Yaden DB , Newberg A . The varieties of spiritual experience: 21st century research and perspectives. Oxford University Press; 2022.
- Stress and sleep reactivity: a prospective investigation of the stress‐diathesis model of insomnia.. Sleep., 2014. [PubMed]
- The impact of stress on sleep: pathogenic sleep reactivity as a vulnerability to insomnia and circadian disorders.. J Sleep Res., 2018. [PubMed]
- Neurobiological interactions between stress and the endocannabinoid system.. Neuropsychopharmacology., 2016. [PubMed]
- The endocannabinoid system in modulating fear, anxiety, and stress.. Dialogues Clin Neurosci., 2022
- Cannabinoid receptor 1 signalling modulates stress susceptibility and microglial responses to chronic social defeat stress.. Transl Psychiatry., 2021a. [PubMed]
- Endocannabinoid‐mediated plasticity in nucleus accumbens controls vulnerability to anxiety after social defeat stress.. Cell Rep., 2016. [PubMed]
- Genetic dissection of the role of cannabinoid type‐1 receptors in the emotional consequences of repeated social stress in mice.. Neuropsychopharmacology., 2012. [PubMed]
- Adverse social experiences in adolescent rats result in enduring effects on social competence, pain sensitivity and endocannabinoid signaling.. Front Behav Neurosci., 2016. [PubMed]
- Endocannabinoid signaling in the control of social behavior.. Trends Neurosci., 2017. [PubMed]
- Wired to run: exercise‐induced endocannabinoid signaling in humans and cursorial mammals with implications for the ‘runner’s high’.. J Exp Biol., 2012. [PubMed]
- Exercise‐induced euphoria and anxiolysis do not depend on endogenous opioids in humans.. Psychoneuroendocrinology., 2021. [PubMed]
- Pivotal mental states.. J Psychopharmacol., 2021. [PubMed]
- Psilocybin induces acute and persisting alterations in immune status and the stress response in healthy volunteers.. medRxiv., 2022
- Excitotoxicity: still hammering the ischemic brain in 2020.. Front Neurosci., 2020. [PubMed]
- Regional neuroprotective effects of the NMDA receptor antagonist MK‐801 (dizocilpine) in hypoglycemic brain damage.. J Cereb Blood Flow Metab., 1990. [PubMed]
- Blockade of GluN2B‐containing NMDA receptors reduces short‐term brain damage induced by early‐life status epilepticus.. Neurotoxicology., 2019. [PubMed]
- Efficacy of prophylactic versus therapeutic administration of the NMDA receptor antagonist MK‐801 on the acute neurochemical response to a concussion in a rat model combining force and rotation.. J Neurosurg., 2021. [PubMed]
- 88 Collins SA . 3, 4‐methylenedioxy‐methamphetamine (MDMA)‐induced increases in hippocampal glutamate concentrations and its impact on the dentate gyrus (Doctoral dissertation). University of Toledo; 2015.
- Toward wisdom from failure: lessons from neuroprotective stroke trials and new therapeutic directions.. Stroke., 2002. [PubMed]
- Why did NMDA receptor antagonists fail clinical trials for stroke and traumatic brain injury?. Lancet Neurol., 2002. [PubMed]
- Preliminary evidence of cannabinoid effects on brain‐derived neurotrophic factor (BDNF) levels in humans.. Psychopharmacology., 2009. [PubMed]
- Repeated hyperthermia exposure increases circulating Brain Derived Neurotrophic Factor levels which is associated with improved quality of life, and reduced anxiety: a randomized controlled trial.. J Therm Biol., 2020. [PubMed]
- BDNF release is required for the behavioral actions of ketamine.. Int J Neuropsychopharmacol., 2015
- Acute psychosocial stress increases serum BDNF levels: an antagonistic relation to cortisol but no group differences after mental training.. Neuropsychopharmacology., 2019. [PubMed]
- Psychedelics promote structural and functional neural plasticity.. Cell Rep., 2018. [PubMed]
- Intermittent fasting and cognitive performance–targeting BDNF as potential strategy to optimise brain health.. Front Neuroendocrinol., 2022. [PubMed]
- Exercise and circulating BDNF: mechanisms of release and implications for the design of exercise interventions.. Appl Physiol Nutr Metab., 2018. [PubMed]
- Serotonin and brain function: a tale of two receptors.. J Psychopharmacol., 2017. [PubMed]
- Serotonin 2A receptors and cannabinoids.. Prog Brain Res., 2021. [PubMed]
- Endocannabinoid control of glutamate NMDA receptors: the therapeutic potential and consequences of dysfunction.. Oncotarget., 2016. [PubMed]
- Atypical but not typical antipsychotic drugs ameliorate phencyclidine‐induced emotional memory impairments in mice.. Eur Neuropsychopharmacol., 2019. [PubMed]
- LSD and DOB: interaction with 5‐HT2A receptors to inhibit NMDA receptor‐mediated transmission in the rat prefrontal cortex.. Eur J Neurosci., 1999. [PubMed]
- M100907, a selective 5‐HT2A receptor antagonist and a potential antipsychotic drug, facilitates N‐methyl‐D‐aspartate–receptor mediated neurotransmission in the rat medial prefrontal cortical neurons in vitro.. Neuropsychopharmacology., 1998. [PubMed]
- The 5‐HT2A receptor antagonist M100, 907 prevents extracellular glutamate rising in response to NMDA receptor blockade in the mPFC.. J Neurochem., 2004. [PubMed]
- Blockade of phencyclidine‐induced hyperlocomotion by olanzapine, clozapine and serotonin receptor subtype selective antagonists in mice.. Psychopharmacology., 1997. [PubMed]
- Reproducing the dopamine pathophysiology of schizophrenia and approaches to ameliorate it: a translational imaging study with ketamine.. Mol Psychiatry., 2021. [PubMed]
- Blockade of phencyclidine‐induced hyperlocomotion by clozapine and MDL 100,907 in rats reflects antagonism of 5‐HT2A receptors.. Eur J Pharmacol., 1995. [PubMed]
- Phencyclidine‐induced head‐twitch responses as 5‐HT2 receptor‐mediated behavior in rats.. Neurosci Lett., 1987. [PubMed]
- Pharmacological characterizations of memantine‐induced disruption of prepulse inhibition of the acoustic startle response in mice: involvement of dopamine D2 and 5‐HT2A receptors.. Behav Brain Res., 2011. [PubMed]
- 5‐HT2A receptor antagonists block MK‐801‐induced stereotypy and hyperlocomotion.. Eur J Pharmacol., 1998. [PubMed]
- Effects of 5‐hydroxytryptamine 2 receptor antagonism on the behavioral activation and immediate early gene expression induced by dizocilpine.. J Pharmacol Exp Ther., 1998. [PubMed]
- Combined serotonin (5‐HT) 1A agonism, 5‐HT2A and dopamine D2 receptor antagonism reproduces atypical antipsychotic drug effects on phencyclidine‐impaired novel object recognition in rats.. Behav Brain Res., 2015. [PubMed]
- Enhanced prefrontal serotonin 2A receptor signaling in the subchronic phencyclidine mouse model of schizophrenia.. J Neurosci Res., 2013. [PubMed]
- Clozapine acts as an agonist at serotonin 2A receptors to counter MK‐801‐induced behaviors through a βarrestin2‐independent activation of Akt.. Neuropsychopharmacology., 2014. [PubMed]
- Risperidone attenuates MK‐801‐induced hyperlocomotion in mice via the blockade of serotonin 5‐HT2A/2C receptors.. Eur J Pharmacol., 2007. [PubMed]
- The effects of Δ9‐tetrahydrocannabinol on the dopamine system.. Nature., 2016. [PubMed]
- Endocannabinoid modulation of dopamine neurotransmission.. Neuropharmacology., 2017. [PubMed]
- A brain on cannabinoids: the role of dopamine release in reward seeking and addiction.. Cold Spring Harb Perspect Med., 2021. [PubMed]
- Assessment of transient dopamine responses to smoked cannabis.. Drug Alcohol Depend., 2021. [PubMed]
- Endocannabinoid modulation of amphetamine sensitization is disrupted in a rodent model of lesion‐induced dopamine dysregulation.. Synapse., 2009. [PubMed]
- Prenatal THC exposure produces a hyperdopaminergic phenotype rescued by pregnenolone.. Nat Neurosci., 2019. [PubMed]
- Effects of adolescent Δ9‐tetrahydrocannabinol exposure on the behavioral effects of cocaine in adult Sprague–Dawley rats.. Exp Clin Psychopharmacol., 2019. [PubMed]
- Context‐specific reversal of cocaine sensitization by the CB1 cannabinoid receptor antagonist rimonabant.. Neuropsychopharmacology., 2008. [PubMed]
- Chronic Δ9‐tetrahydrocannabinol exposure induces a sensitization of dopamine D2/3 receptors in the mesoaccumbens and nigrostriatal systems.. Neuropsychopharmacology., 2012. [PubMed]
- Chronic (−)‐Δ9‐tetrahydrocannabinol treatment induces sensitization to the psychomotor effects of amphetamine in rats.. Eur J Pharmacol., 1999. [PubMed]
- Chronic treatment with Δ9‐tetrahydrocannabinol enhances the locomotor response to amphetamine and heroin. Implications for vulnerability to drug addiction.. Neuropharmacology., 2001. [PubMed]
- Cocaine‐induced endocannabinoid release modulates behavioral and neurochemical sensitization in mice.. Addict Biol., 2015. [PubMed]
- Behavioral sensitization to amphetamine follows chronic administration of the CB1 agonist WIN 55,212‐2 in Lewis rats.. Pharmacol Biochem Behav., 2002. [PubMed]
- Adolescent THC exposure causes enduring prefrontal cortical disruption of GABAergic inhibition and dysregulation of sub‐cortical dopamine function.. Sci Rep., 2017. [PubMed]
- Mesolimbic dopamine dysregulation as a signature of information processing deficits imposed by prenatal THC exposure.. Prog Neuro‐Psychopharmacol Biol Psychiatry., 2021
- The role of the CB1 cannabinoid receptor and its endogenous ligands, anandamide and 2‐arachidonoylglycerol, in amphetamine‐induced behavioural sensitization.. Behav Brain Res., 2008. [PubMed]
- Repeated but not acute treatment with∆ 9‐tetrahydrocannabinol disrupts prepulse inhibition of the acoustic startle: reversal by the dopamine D2/3 receptor antagonist haloperidol.. Eur Neuropsychopharmacol., 2014. [PubMed]
- Amphetamine effects on dopamine levels and behavior following cannabinoid exposure during adolescence.. Eur J Pharmacol., 2004. [PubMed]
- Stress‐induced dopamine response in subjects at clinical high risk for schizophrenia with and without concurrent cannabis use.. Neuropsychopharmacology., 2014. [PubMed]
- Serotonin 5‐HT2 receptor interactions with dopamine function: implications for therapeutics in cocaine use disorder.. Pharmacol Rev., 2015. [PubMed]
- The effects of ketamine on dopaminergic function: meta‐analysis and review of the implications for neuropsychiatric disorders.. Mol Psychiatry., 2018. [PubMed]
- Modulation of amphetamine‐induced striatal dopamine release by ketamine in humans: implications for schizophrenia.. Biol Psychiatry., 2000. [PubMed]
- Effects of MK‐801 on spontaneous and amphetamine‐stimulated dopamine release in striatum measured with in vivo microdialysis in awake rats.. Brain Res Bull., 1996. [PubMed]
- Dopamine ‘ups and downs’ in addiction revisited.. Trends Neurosci., 2021. [PubMed]
- On the relationship of first‐episode psychosis to the amphetamine‐sensitized state: a dopamine D2/3 receptor agonist radioligand study.. Transl Psychiatry., 2020. [PubMed]
- The temporal trajectory of the psychedelic mushroom experience mimics the narrative arc of the Hero’s Journey. [Manuscript in prep].. 2023a
- The psychedelic afterglow phenomenon: a systematic review of subacute effects of classic serotonergic psychedelics.. Ther Adv Psychopharmacol., 2023. [PubMed]
- Consumer reports of “keto flu” associated with the ketogenic diet.. Front Nutr., 2020
- The experience of panic attacks in adolescents: an interpretative phenomenological analysis study.. Emot Behav Diffic., 2021
- Diabetic ketoacidosis: clinical characteristics and precipitating factors.. Cureus., 2020
- Acute stress disorder.. Curr Opin Psychol., 2017. [PubMed]
- Positron emission tomography and fluorodeoxyglucose studies of metabolic hyperfrontality and psychopathology in the psilocybin model of psychosis.. Neuropsychopharmacology., 1997. [PubMed]
- Hyperglycemia induced by the 5‐HT receptor agonist, 5‐methoxytryptamine, in rats: involvement of the peripheral 5‐HT2A receptor.. Eur J Pharmacol., 1997. [PubMed]
- The involvement of the peripheral 5‐HT2A receptor in peripherally administered serotonin‐induced hyperglycemia in rats.. Life Sci., 1995. [PubMed]
- BDNF mediates adaptive brain and body responses to energetic challenges.. Trends Endocrinol Metab., 2014. [PubMed]
- 151 Palmer CM . Brain energy: a revolutionary breakthrough in understanding mental health—and improving treatment for anxiety, depression, OCD, PTSD, and more. Blackstone Publishing; 2022.
- 5‐HT2 receptor regulation of mitochondrial genes: unexpected pharmacological effects of agonists and antagonists.. J Pharmacol Exp Ther., 2016. [PubMed]
- 5‐hydroxytryptamine receptor stimulation of mitochondrial biogenesis.. J Pharmacol Exp Ther., 2010. [PubMed]
- N, N‐dimethyltryptamine reduces infarct size and improves functional recovery following transient focal brain ischemia in rats.. Exp Neurol., 2020. [PubMed]
- Serotonergic neurons as carbon dioxide sensors that maintain pH homeostasis.. Nat Rev Neurosci., 2004. [PubMed]
- 156 Teran FA , Richerson GB . 5‐HT neurons and central CO2 chemoreception. Handbook of behavioral neuroscience. Vol 31. Elsevier; 2020:377‐391.
- Blood and muscle pH after maximal exercise in man.. J Appl Physiol., 1972. [PubMed]
- 158 Swenson ER . Hypoxia and its acid–base consequences: from mountains to malignancy. Translation in Progress; 2016:301‐323.
- The acid–base balance and gender in Inflammation: a mini‐review.. Front Immunol., 2018. [PubMed]
- The link between brain acidosis, breathing and seizures: a novel mechanism of action for the ketogenic diet in a model of infantile spasms.. Brain Commun., 2021. [PubMed]
- Biosynthesis and extracellular concentrations of N, N‐dimethyltryptamine (DMT) in mammalian brain.. Sci Rep., 2019. [PubMed]
- Hypoxia routes tryptophan homeostasis towards increased tryptamine production.. Front Immunol., 2021. [PubMed]
- The effect of carbon dioxide on near‐death experiences in out‐of‐hospital cardiac arrest survivors: a prospective observational study.. Crit Care., 2010
- Neurochemical models of near‐death experiences: a large‐scale study based on the semantic similarity of written reports.. Conscious Cogn., 2019. [PubMed]
- This is your brain on death: a comparative analysis of a near‐death experience and subsequent 5‐Methoxy‐DMT experience.. Front Psychol., 2023
- Anomalous psychedelic experiences: at the neurochemical juncture of the humanistic and parapsychological.. J Humanist Psychol., 2022
- Comparison of psychedelic and near‐death or other non‐ordinary experiences in changing attitudes about death and dying.. PLoS One., 2022. [PubMed]
- DMT models the near‐death experience.. Front Psychol., 2018. [PubMed]
- 169 Greyson B , Bush NE . 14 distressing near‐death experiences. In: Bailey LW , Yates J , eds. The Near‐Death Experience. Routledge; 2013:207‐230.
- The experience of drowning.. Med Leg J., 2022. [PubMed]
- 5‐HT2A receptor activation is necessary for CO2‐induced arousal.. J Neurophysiol., 2015. [PubMed]
- Onset of spontaneous panic attacks: a prospective study of risk factors.. Psychosom Med., 2006. [PubMed]
- Characterization of a 7% carbon dioxide (CO2) inhalation paradigm to evoke anxiety symptoms in healthy subjects.. J Psychopharmacol., 2005. [PubMed]
- 174 Meduna LJ . Carbondioxide therapy: a neurophysiological treatment of nervous disorders. Charles C Thomas Publisher; 1950.
- Cognitive factors in carbon dioxide therapy.. J Psychosom Res., 1982. [PubMed]
- Associations between psychedelic use, abuse, and dependence and lifetime panic attack history in a representative sample.. J Anxiety Disord., 2007. [PubMed]
- Focusing on the negative: cases of long‐term negative psychological responses to psychedelics.. Sci Rep., 2023. [PubMed]
- Panic attacks and psychoticism.. Am J Psychiatry., 2004. [PubMed]
- The prodromal phase of first‐episode psychosis: past and current conceptualizations.. Schizophr Bull., 1996. [PubMed]
- Brain lactate and pH in schizophrenia and bipolar disorder: a systematic review of findings from magnetic resonance studies.. Neuropsychopharmacology., 2018. [PubMed]
- Decreased brain pH as a shared endophenotype of psychiatric disorders.. Neuropsychopharmacology., 2018. [PubMed]
- Decreased brain PH and pathophysiology in schizophrenia.. Int J Mol Sci., 2021. [PubMed]
- Evidence for altered energy metabolism, increased lactate, and decreased pH in schizophrenia brain: a focused review and meta‐analysis of human postmortem and magnetic resonance spectroscopy studies.. Schizophr Res., 2020. [PubMed]
- Effects of antipsychotics, antidepressants and mood stabilizers on risk for physical diseases in people with schizophrenia, depression and bipolar disorder.. World Psychiatry., 2015. [PubMed]
- Atypical antipsychotics‐induced metabolic syndrome and nonalcoholic fatty liver disease: a critical review.. Neuropsychiatr Dis Treat., 2019. [PubMed]
- Schizophrenia: a disorder of broken brain bioenergetics.. Mol Psychiatry., 2022. [PubMed]
- Blood ketone concentration in patients with mental and emotional disorders.. AMA Archives of Neurology & Psychiatry., 1952. [PubMed]
- Lactate: the ugly duckling of energy metabolism.. Nat Metab., 2020. [PubMed]
- Increased lactate levels and reduced pH in postmortem brains of schizophrenics: medication confounds.. J Neurosci Methods., 2008. [PubMed]
- Bipolar disorder and type 2 diabetes mellitus: a bidirectional relationship.. Eur J Psychiatry., 2022
- The ketogenic diet for refractory mental illness: a retrospective analysis of 31 inpatients.. Front Psych., 2022
- Ketogenic diet as a metabolic treatment for mental illness.. Curr Opin Endocrinol Diabetes Obes., 2020. [PubMed]
- Ketogenic diet for schizophrenia: clinical implication.. Curr Opin Psychiatry., 2019. [PubMed]
- A ketogenic diet substantially reshapes the human metabolome.. Clin Nutr., 2023. [PubMed]
- Decreased cerebrospinal fluid kynurenic acid in epileptic spasms: a biomarker of response to corticosteroids.. EBioMedicine., 2022. [PubMed]
- Changes in tryptophan and kynurenine pathway metabolites in the blood of children treated with ketogenic diet for refractory epilepsy.. Seizure., 2019. [PubMed]
- Ketogenic diet increases concentrations of kynurenic acid in discrete brain structures of young and adult rats.. J Neural Transm., 2012. [PubMed]
- Regulation of kynurenine metabolism by a ketogenic diet.. J Lipid Res., 2018. [PubMed]
- Emotional breakthrough and psychedelics: validation of the emotional breakthrough inventory.. J Psychopharmacol., 2019. [PubMed]
- Patient experiences of psilocybin‐assisted psychotherapy: an interpretative phenomenological analysis.. J Humanist Psychol., 2017
- The evolved psychology of psychedelic set and setting: inferences regarding the roles of shamanism and entheogenic ecopsychology.. Front Pharmacol., 2021. [PubMed]
- 202 Boisen AT . The exploration of the inner world. A study of mental disorder and religious experience; 1936.
- The stages of mania: a longitudinal analysis of the manic episode.. Arch Gen Psychiatry., 1973. [PubMed]
- Rate of switch from depression into mania after therapeutic sleep deprivation in bipolar depression.. Psychiatry Res., 1999. [PubMed]
- Triggers for acute mood episodes in bipolar disorder: a systematic review.. J Psychiatr Res., 2023. [PubMed]
- Affective switch associated with oral, low dose ketamine treatment in a patient with treatment resistant bipolar I depression. Case report and literature review.. Front Psychiatry., 2020. [PubMed]
- 207 Brouwer A , Winkelman MJ , Raison CL . Shamanism; psychopathology and psychotherapy. Religion, Brain and Behavior; 2023d [Accepted for Publication].
- 208 Freedman DX . LSD: the bridge from human to animal. In: Jacobs BL , ed. Hallucinogens: neurochemical, behavioral, and clinical perspectives. Raven Press; 1984:203‐226.
- Distinct temporal phases in the behavioral pharmacology of LSD: dopamine D 2 receptor‐mediated effects in the rat and implications for psychosis.. Psychopharmacology., 2005. [PubMed]
- Lysergic acid diethylamide antagonism by chlorpromazine, haloperidol, diazepam, and pentobarbital in the rabbit.. Toxicol Appl Pharmacol., 1977. [PubMed]
- Me, myself, bye: regional alterations in glutamate and the experience of ego dissolution with psilocybin.. Neuropsychopharmacology., 2020. [PubMed]
- Classic psychedelic coadministration with lithium, but not lamotrigine, is associated with seizures: an analysis of online psychedelic experience reports.. Pharmacopsychiatry., 2021. [PubMed]
- Direct comparison of the acute effects of lysergic acid diethylamide and psilocybin in a double‐blind placebo‐controlled study in healthy subjects.. Neuropsychopharmacology., 2022. [PubMed]
- Synthetic cannabinoids found in “spice” products alter body temperature and cardiovascular parameters in conscious male rats.. Drug Alcohol Depend., 2017. [PubMed]
- Activating dopamine D2 receptors reduces brown adipose tissue thermogenesis induced by psychological stress and by activation of the lateral habenula.. Sci Rep., 2019. [PubMed]
- Synergistic interaction between ketamine and magnesium in lowering body temperature in rats.. Physiol Behav., 2014. [PubMed]
- Effects of antidepressant and antipsychotic use on weight gain: a systematic review.. Obes Rev., 2019. [PubMed]
- Real‐world data on the adverse metabolic effects of second‐generation antipsychotics and their potential determinants in adult patients: a systematic review of population‐based studies.. Adv Ther., 2021. [PubMed]
- Antipsychotics promote metabolic disorders disrupting cellular lipid metabolism and trafficking.. Trends Endocrinol Metab., 2019. [PubMed]
- Heat intolerance in patients with chronic schizophrenia maintained with antipsychotic drugs.. Am J Psychiatry., 2000. [PubMed]
- Neuroleptic malignant syndrome.. Am J Psychiatry., 2007. [PubMed]
- Natural molecules and neuroprotection: kynurenic acid, pantethine and α‐lipoic acid.. Int J Mol Sci., 2021. [PubMed]
- Kynurenic acid and Gpr35 regulate adipose tissue energy homeostasis and inflammation.. Cell Metab., 2018. [PubMed]
- Kynurenine pathway in major depression: evidence of impaired neuroprotection.. J Affect Disord., 2007. [PubMed]
- Reduction of kynurenic acid to quinolinic acid ratio in both the depressed and remitted phases of major depressive disorder.. Brain Behav Immun., 2015. [PubMed]
- Increased plasma level of kynurenic acid in drug‐free patients with first‐episode schizophrenia compared to patients with chronic schizophrenia and healthy controls: preliminary data.. Nord J Psychiatry., 2022. [PubMed]
- Elevated salivary kynurenic acid levels related to enlarged choroid plexus and severity of clinical phenotypes in treatment‐resistant schizophrenia.. Brain Behav Immun., 2022b. [PubMed]
- Kynurenic acid in schizophrenia: a systematic review and meta‐analysis.. Schizophr Bull., 2017. [PubMed]
- Peripheral and central levels of kynurenic acid in bipolar disorder subjects and healthy controls.. Transl Psychiatry., 2019. [PubMed]
- Kynurenine pathway metabolites are associated with gray matter volume in subjects with schizophrenia.. Front Psych., 2022
- Decreased plasma concentrations of kynurenine and kynurenic acid in schizophrenia patients.. Eur Psychiatry., 2023
- Serum kynurenic acid is reduced in affective psychosis.. Transl Psychiatry., 2017. [PubMed]
- Stress‐induced increase in kynurenic acid as a potential biomarker for patients with schizophrenia and distress intolerance.. JAMA Psychiatry., 2014. [PubMed]
- Dysregulation of kynurenine metabolism is related to proinflammatory cytokines, attention, and prefrontal cortex volume in schizophrenia.. Mol Psychiatry., 2020. [PubMed]
- The origin of NMDA receptor hypofunction in schizophrenia.. Pharmacol Ther., 2020. [PubMed]
- Ketamine‐induced NMDA receptor hypofunction as a model of memory impairment and psychosis.. Neuropsychopharmacology., 1999. [PubMed]
- The relationship between plasma serotonin and kynurenine pathway metabolite levels and the treatment response to escitalopram and desvenlafaxine.. Brain Behav Immun., 2020. [PubMed]
- Kynurenic acid acts as a signaling molecule regulating energy expenditure and is closely associated with metabolic diseases.. Front Endocrinol., 2022
- Sex differences in hippocampal memory and kynurenic acid formation following acute sleep deprivation in rats.. Sci Rep., 2018. [PubMed]
- Acute sleep deprivation during pregnancy in rats: rapid elevation of placental and fetal inflammation and kynurenic acid.. Neurobiol Stress., 2020. [PubMed]
- Two‐day fasting affects kynurenine pathway with additional modulation of short‐term whole‐body cooling: a quasi‐randomised crossover trial.. Br J Nutr., 2023
- Antidepressant effect of repeated ketamine administration on kynurenine pathway metabolites in patients with unipolar and bipolar depression.. Brain Behav Immun., 2018. [PubMed]
- Schizophrenia: a developmental disorder with a risk of non‐specific but avoidable decline.. Schizophr Res., 2022. [PubMed]
- The nature of relapse in schizophrenia.. BMC Psychiatry., 2013. [PubMed]
- The activation of 5‐HT2A receptors in prefrontal cortex enhances dopaminergic activity.. J Neurochem., 2005. [PubMed]
- M100, 907, a selective 5‐HT2A antagonist, attenuates dopamine release in the rat medial prefrontal cortex.. Brain Res., 2001. [PubMed]
- Evidence for the preferential involvement of 5‐HT2A serotonin receptors in stress‐and drug‐induced dopamine release in the rat medial prefrontal cortex.. Neuropsychopharmacology., 2006. [PubMed]
- The methamphetamine‐associated psychosis spectrum: a clinically focused review.. Int J Ment Heal Addict., 2020
- Comparison of amphetamine psychosis and schizophrenia.. Br J Psychiatry., 1965. [PubMed]
- The experimental reproduction of amphetamine psychosis.. Arch Gen Psychiatry., 1973. [PubMed]
- Long‐lasting alterations in 5‐HT 2A receptor after a binge regimen of methamphetamine in mice.. Int J Neuropsychopharmacol., 2014. [PubMed]
- Methamphetamine‐induced sensitization includes a functional upregulation of ventral pallidal 5‐HT2A/2C receptors.. Synapse., 2008. [PubMed]
- Dopamine requires unique residues to signal via the serotonin 2A receptor.. Neuroscience., 2020. [PubMed]
- Pimavanserin for patients with Parkinson’s disease psychosis: a randomised, placebo‐controlled phase 3 trial.. Lancet., 2014. [PubMed]
- Pimavanserin, a serotonin2A receptor inverse agonist, for the treatment of Parkinson’s disease psychosis.. Neuropsychopharmacology., 2010. [PubMed]
- Set and setting, psychedelics and the placebo response: an extra‐pharmacological perspective on psychopharmacology.. J Psychopharmacol., 2016. [PubMed]
- The influence of positive affect on jumping to conclusions in delusional thinking.. Personal Individ Differ., 2011
- Temporal dynamics of suspiciousness and hallucinations in clinical high risk and first episode psychosis.. Psychiatry Res., 2020. [PubMed]
- Social defeat: risk factor for schizophrenia?. Br J Psychiatry., 2005. [PubMed]
- Hypothesis: social defeat is a risk factor for schizophrenia?. Br J Psychiatry., 2007
- The social defeat hypothesis of schizophrenia: an update.. Schizophr Bull., 2013. [PubMed]
- The social defeat hypothesis of schizophrenia: issues of measurement and reverse causality.. World Psychiatry., 2016. [PubMed]
- The relationship between childhood trauma, dopamine release and dexamphetamine‐induced positive psychotic symptoms: a [11C]‐(+)‐PHNO PET study.. Transl Psychiatry., 2019. [PubMed]
- Neuroticism is associated with challenging experiences with psilocybin mushrooms.. Pers Individ Dif., 2017. [PubMed]
- Consensus paper of the WFSBP task force on cannabis, cannabinoids and psychosis.. World J Biol Psychiatry., 2022. [PubMed]
- Cannabis use and psychosis: a review of reviews.. Eur Arch Psychiatry Clin Neurosci., 2020. [PubMed]
- A systematic review of risk factors for methamphetamine‐associated psychosis.. Australian & New Zealand Journal of Psychiatry., 2018. [PubMed]
- Associations between use of cocaine, amphetamines, or psychedelics and psychotic symptoms in a community sample.. Acta Psychiatr Scand., 2011. [PubMed]
- Mental health conditions and receipt of mental health care by illicit lysergic acid diethylamide (LSD) use status among young adults in the United States.. Addiction., 2022. [PubMed]
- Psychedelics and mental health: a population study.. PLoS One., 2013. [PubMed]
- The link between use of psychedelic drugs and mental health problems.. J Psychopharmacol., 2015. [PubMed]
- Who is the typical psychedelics user? Methodological challenges for research in psychedelics use and its consequences.. Nordic Stud Alcohol Drugs., 2021
- Age at onset of mental disorders worldwide: large‐scale meta‐analysis of 192 epidemiological studies.. Mol Psychiatry., 2022. [PubMed]
- Adverse reactions to psychedelic drugs. A review of the literature.. J Nerv Ment Dis., 1984. [PubMed]
- Substance use and abuse in first‐episode psychosis: prevalence before and after early intervention.. Schizophr Bull., 2007. [PubMed]
- Substance use in a population‐based clinic sample of people with first‐episode psychosis.. Br J Psychiatry., 2007. [PubMed]
- Prediction of onset of substance‐induced psychotic disorder and its progression to schizophrenia in a Swedish national sample.. Am J Psychiatry., 2019. [PubMed]
- Chronological association between increases in drug abuse and psychosis in Connecticut state hospitals.. J Neuropsychiatry Clin Neurosci., 1998. [PubMed]
- Hormesis defined.. Ageing Res Rev., 2008. [PubMed]
- Canalization and plasticity in psychopathology.. Neuropharmacology., 2023. [PubMed]
- Genetic vulnerability to schizophrenia is associated with cannabis use patterns during adolescence.. Drug Alcohol Depend., 2018. [PubMed]
- Genetic risk for schizophrenia is associated with substance use in emerging adulthood: an event‐level polygenic prediction model.. Psychol Med., 2019. [PubMed]
- Could psychedelic drugs have a role in the treatment of schizophrenia? Rationale and strategy for safe implementation.. Mol Psychiatry., 2023. [PubMed]
- The IUPHAR/BPS Guide to PHARMACOLOGY in 2019: updates and expansion to encompass the new guide to IMMUNOPHARMACOLOGY.. Nucleic Acids Res., 2018. [DOI | PubMed]
- The concise guide to PHARMACOLOGY 2019/20: introduction and other protein targets.. Br J Pharmacol., 2019. [PubMed]
