Complementary medicines used in ulcerative colitis and unintended interactions with cytochrome P450-dependent drug-metabolizing enzymes
Abstract
Ulcerative colitis (UC) is an idiopathic, chronic inflammatory disease with multiple genetic and a variety of environmental risk factors. Although current drugs significantly aid in controlling the disease, many people have led to the application of complementary therapies due to the common belief that they are natural and safe, as well as due to the consideration of the side effect of current drugs. Curcumin, cannabinoids, wheatgrass, Boswellia, wormwood and Aloe vera are among the most commonly used complementary medicines in UC. However, these treatments may have adverse and toxic effects due to unintended interactions with drugs or drug-metabolizing enzymes such as cytochrome P450s; thus, being ignorant of these interactions might cause deleterious effects with severe consequences. In addition, the lack of complete and controlled long-term studies with the use of these complementary medicines regarding drug metabolism pose additional risk and unsafety. Thus, this review aims to give an overview of the potential interactions of drug-metabolizing enzymes with the complementary botanical medicines used in UC, drawing attention to possible adverse effects.
Article type: Review Article
Keywords: Cytochrome P450, CYPs, ulcerative colitis, complementary medicines, herbal products, adverse effect
Affiliations: Department of Molecular Biology and Genetics, Faculty of Life and Natural Sciences, Abdullah Gül University, Kayseri, Turkey; Department of Biology, Faculty of Arts and Sciences, Pamukkale University, Denizli, Turkey
License: © TÜBİTAK CC BY 4.0 This work is licensed under a Creative Commons Attribution 4.0 International License.
Article links: DOI: 10.55730/1300-0144.5482 | PubMed: 36422483 | PMC: PMC10395683
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.2 MB)
1. Introduction
Inflammatory bowel diseases (IBDs) are idiopathic inflammatory disorders that have a chronic course characterized by exacerbation and remission throughout an individual’s lifetime. It is thought that a genetic predisposition as well as environmental factors play a role in the manifestation of these diseases. IBDs are classified into two types: ulcerative colitis (UC) and Crohn’s disease (CD) [ref. 1, ref. 2]. UC and CD are distinguished by the sections of the digestive system affected and by phases of relapses and remission, causing symptoms that are somewhat different between the two diseases [ref. 3–ref. 5]. UC is still an idiopathic inflammatory disease of the colonic mucosa with an unknown aetiology that is more prevalent globally than CD. Interaction between genetic and environmental factors, immune dysfunction, intestinal flora dysbiosis, and impaired intestinal mucosal barrier function are currently the leading causes of IBD. UC, with an accelerating global incidence, has an unfavourable influence on patients’ health as well as their emotional, social and occupational life [ref. 2].
Although the burden of the disease is increasing, therapy for UC is challenging due to the absence of clearly defined medical treatments to manage periods of recurrence and remission with long-term consequences. However, drugs such as 5-aminosalicylate, azathioprine, 6-mercaptopurine, cyclosporine, and antitumour necrosis factor monoclonal antibodies have been utilized to treat UC [ref. 6,ref. 7]. The main focus of UC treatment is to improve quality of life with minimum steroid exposure while also alleviating symptoms and mucosal inflammation. Annual direct medical costs per patient in the United States ranged from $6217 to $11,477 and from €8949 to €10,395 in Europe, posing a significant financial burden on patients, families, and governments [ref. 8–ref. 11]. Thus, the use of complementary and alternative medicine (CAM) among UC patients is popular, with herbal therapies being the most commonly utilized form of treatment. It is estimated that 28.9% of the US population use one or more natural medicines on a regular basis, with herbal products accounting for 9.6%–12.1% of these. Various herbal products used in the clinic have been found in studies to be useful in the treatment of UC [ref. 12–ref. 18].
The microsomal cytochrome P450 system is a product of the CYP superfamily of genes, and it is a key electron transport chain in the membrane of the endoplasmic reticulum of the cell. These enzymes are critical in the oxidative activation, inactivation, and excretion of the majority of xenobiotics and drugs, as well as in controlling the degree of their toxicity, among other functions [ref. 19–ref. 24]. Cytochrome P450 catalyses the oxidation of lipophilic compounds by introducing one atom of molecular oxygen into the substrate, transforming the latter into either less toxic or completely harmless hydrophilic metabolites that may be excreted from the body. The electron travels from NADPH or NADH via a flavoprotein cytochrome P450 reductase or a cytochrome b5 reductase to distinct isoforms of cytochrome P450 (CYP450s) during oxidative metabolism in the microsomal microenvironment involving the cytochrome P4S0 system (Figure 1) [ref. 25–ref. 27]. Many endobiotic and xenobiotic oxidation events are facilitated by cytochrome P450s, which are the terminal oxidase components of an electron transport system found in the endoplasmic reticulum and responsible for many of these reactions.
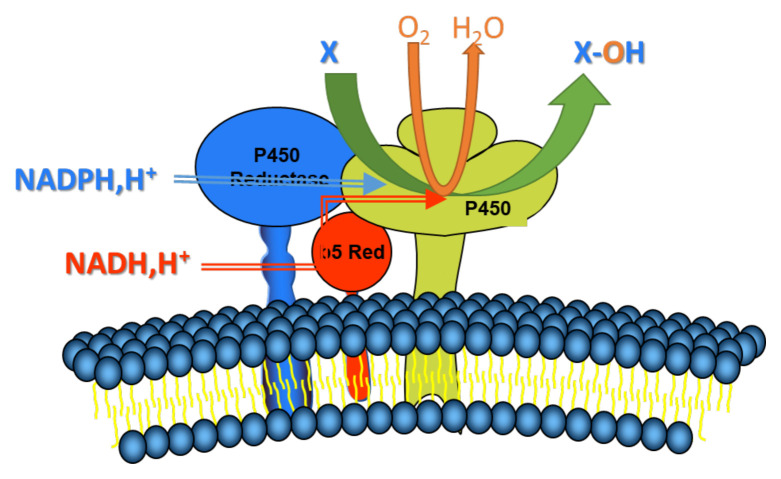
Humans express a total of 57 CYP proteins; of these, seven are found in mitochondria, where they catalyse particular oxidation events associated with steroid metabolism. Only six genes —CYP1A2, CYP2C9, CYP2B6, CYP2C19, CYP2D6, and CYP3A4— make substantial contributions to drug clearance out of the remaining 50 genes studied. It has been estimated that approximately 60 % of all drug-related material provided to patients is cleared by these six CYPs. The CYP3A subfamily is responsible for metabolizing approximately half of the medicines that have been cleared by CYPs [ref. 28–ref. 30]. The role of some critical CYP isoforms in the pathogenesis or aetiology of ulcerative colitis concerns gene polymorphisms [ref. 31]. In addition, their significance in metabolism concerning ulcerative colitis in patients has been reported [ref. 32]. Plant-derived phytochemicals are also substrates for various subtypes of the CYP450 family and can induce or reduce the activity of these enzymes; thus, the concurrent use of phytochemicals and conventional drugs can cause an increase or decrease in serum levels of both substances via induction or inhibition of CYP450 activity (Figure 2).
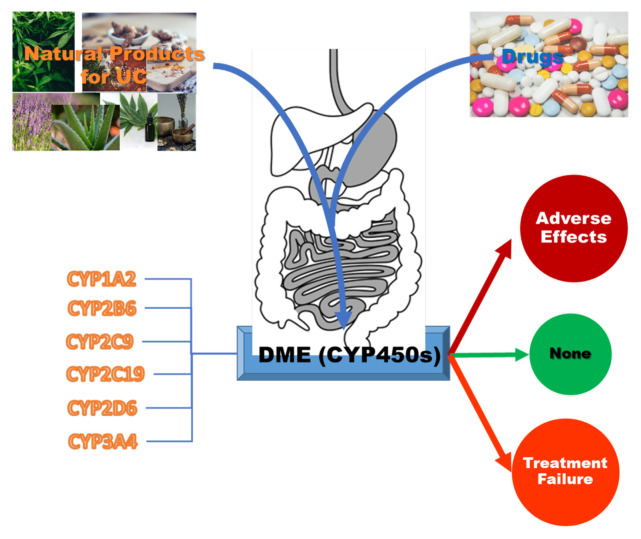
A computerized search of PubMed, Web of Science, and Scopus for randomized controlled trials involving ulcerative colitis was conducted to identify the natural plants used by patients to treat ulcerative colitis. The unfavourable effects of these natural products on cytochrome P450-mediated drug metabolism were reviewed in this review without addressing the therapeutic effectiveness of these natural compounds. A promising choice for curing ulcerative colitis are natural plants and products; however, they are not as safe as the public believes and may lead to serious consequences in both short-term and long-term usage. The individual plants are discussed in detail in alphabetical order below.
1.1. Aloe vera
Aloe vera is a plant that belongs to the Liliaceae family and is extensively used in traditional medicine worldwide. Aloe vera is considered the most widely studied and commercialized plant species. It has been used as a folk remedy for the treatment of a wide range of diseases, including skin problems and gastrointestinal disorders [ref. 33,ref. 34]. The mucilaginous extract obtained from the meaty leaves has antiinflammatory effects and is used by certain physicians to treat patients with ulcerative colitis [ref. 16,ref. 34,ref. 35]. Several constituents from various phytochemical classes, such as alkaloids, anthrones, chromones, flavonoids, glyocoproteins, naphthalenes and pyrones, have been isolated from different aloe species [ref. 36–ref. 38]. It is still not yet completely clear which compounds are responsible for the various observed pharmacological properties [ref. 35,ref. 39,ref. 40]. In addition, some components, such as anthraquinones, can have harmful effects, such as genotoxic, mutagenic, and tumour-promoting effects. Further studies need to be conducted to more accurately define the activities of each component [ref. 41,ref. 42]. Aloe vera is considered a somewhat safe remedy in alternative and complementary medicines (ACM); however, liver toxicity has been reported by several scientists [ref. 34,ref. 43,ref. 44]. Cui and coworkers [ref. 45] showed that aloe vera polysaccharide supplementation significantly downregulated the gene expression, protein synthesis and enzyme activities of hepatic CYP1A2 and 3A4 proteins. Similarly, aloe vera juice inhibited the activity of recombinant human CYP3A4 and CYP2D6 enzymes, which are expressed by the metabolism of testosterone and dextromethorphan, respectively [ref. 46,ref. 47]. There is therapeutic significance to this mechanistic suppression of the CYP3A4 and CYP2D6 enzymes since CYP3A4 and CYP2D6 are the most important enzymes in Phase-I drug responses, accounting for approximately 60%–75% of all phase-I drug reactions [ref. 48,ref. 49]. In addition, large differences in CYP2D6 metabolism have been observed as a result of genetic polymorphisms in this enzyme. Interference with drug metabolism is indicated by the reported inhibition values, which may have consequences for human drug metabolism as well [ref. 46].
It has been observed that aloe vera contains 94.8 mg per kilogram of dry matter of quercetin [ref. 50]. Quercetin, a flavanol, reduced the CYP2C9-mediated 4-methylhydroxylation of tolbutamide and the CYP3A4-mediated 6-hydroxylation of testosterone at micromolar concentrations, suggesting that it may interact with conventional medications that are metabolized by CYP2C9 and CYP3A4 [ref. 51,ref. 52].
Furthermore, anthraquinones are the most important type of components in aloe species, including aloeemodin, rhein, emodin, chrysophanol, physcion and their glucosides [ref. 37,ref. 53]. Studies have demonstrated that several common herbs, particularly those having high concentrations of anthraquinones and naphthoquinone, have significant inhibitory effects against xenobiotic metabolism. Anthraquinones also exhibit high inhibitory effects against the phase I and phase II metabolism of xenobiotics. The underlying mechanism for this activity might be the strong inhibition of the enzymes CYP1A2, 1A1, and SULT1, which are involved in the metabolism of not only foreign xenobiotics but also endogenous chemicals such as melatonin. Similar inhibitions were further confirmed for recombinant human CYP1A1, 1A2, and 2B1, which were inhibited by emodin with IC50 values of 12.25, 0.67, and 14.89, respectively [ref. 54]. More potent inhibitions were also reported [ref. 55]. Emodin potently inhibited the activities of recombinant human CYP1A2 (IC50 0.67–7.62 M) and CYP3A4 (IC50 10 M), as well as in human microsomes. More interestingly, this significant inhibition was confirmed in primary human hepatocytes, indicating that it is a general phenomenon [ref. 56]. More importantly, recent studies have shown that anthraquinones and their metabolites cause DNA damage [ref. 57]. In conclusion, aloe vera, which contains high concentrations of anthraquinones, has the potential to significantly block drug metabolism as well as melatonin metabolism.
Rhein is another anthraquinone metabolite that is abundantly present in aloe vera and other plants [ref. 58]. Rhein inhibited the activity of the enzymes CYP1A2, CYP2C9, CYP2D6, CYP2E1, and CYP3A, with Ki values ranging from 10 to 30 μmol in rat liver microsomes, suggesting the possible interaction between rhein and other concomitantly administered drugs [ref. 59]. Moreover, aloe vera significantly inhibited the levels of both cytochrome P450 and b5 while inducing cytochrome P450 and b5 reductases in a dose-dependent manner [ref. 60]. Thus, aloe vera affects not only CYPs but also the reductase component of the microsomal electron transport system. This observation may be further indicative of interference with oxidative drug metabolism.
In addition, aloe-emodin inhibited the levels of N-acetylation of sulfamethoxazole and p-aminosalicylate, as well as the expression of NAT1 mRNA and protein in A375 cells. S2 cells [ref. 61]. Inhibition was further shown to be dose-dependent using the common substrate 2-aminofluorene, which was transformed by NATs to 2-acetylaminoflourene [ref. 62]. The findings were validated by the use of a cDNA microarray and a PCR assay to determine gene expression levels. The results demonstrated that aloe-emodin suppresses NAT activity in human melanoma cells by downregulating the production of the NAT gene and protein.
In conclusion, aloe and its phytochemical ingredients have been shown to inhibit all six main CYP families, which are responsible for drug metabolism. Additionally, they seem to block NAT enzymes, which are involved in the metabolism of mesalamine, which is one of the most widely prescribed medications for ulcerative colitis patients. These findings highlight the need for more research on aloe vera, such as hazardous chemical discovery, mechanism-based suppression of CYP activities, and toxicokinetic interactions in both metabolically competent cell and animal models. As a result, aloe is a natural herbal substance that is far from being safe for human use, as suggested in the literature [34,ref. 45,ref. 46]. When using aloe vera and its products, special attention should be given to the administration of medications, and the advice of a physician should be followed. Otherwise, unavoidable adverse interactions may result as well as catastrophic health consequences.
1.2. Ananas comosus
Pineapple (Ananas comosus) is a tropical plant whose edible fruit is one of the most important tropical fruits in the international trade market [ref. 63,ref. 64]. Tropical fruits such as pineapple juice and fruit have long been used as traditional treatments to cure digestion system discomforts [ref. 65]. In addition to polyphenols and fibres, bromelain is a combination of proteolytic enzymes and the active component found in the pineapple plant A. comosus. Bromelain, a cysteine protease, has been shown to be effective in a variety of therapeutic areas due to its antiinflammatory and anticancer properties, as well as its capacity to promote apoptotic cell death [ref. 66]. It possesses antiinflammatory actions and benefits in patients with UC and has been the topic of several investigations [ref. 67,ref. 68]. The antiinflammatory effect of bromelain is reported to be related to protease activity [ref. 69,ref. 70]. Bromelain has been shown to be beneficial both clinically and endoscopically in people with mild UC in clinical investigations [ref. 64,ref. 71].
There are no extensive studies on the drug interactions or interactions with CYPs of pineapple or bromelain. Pineapple fruit juices were implicated in DIs in vitro due to their inhibitory effects on CYP2C9 or CYP3A4. Hidaka and colleagues investigated the effect of bromelain on diclofenac 40-hydroxylase activity, presuming that bromelain is a CYP2C9 inhibitor candidate. At a final dosage of 50 mg/mL, the addition of bromelain resulted in almost total suppression of CYP2C9 activity. The inhibition was concentration dependent, and the IC50 value was found to be 1.2 mg/mL. These findings imply that bromelain is a powerful inhibitor of CYP2C9 and is connected to the inhibitory effect of pineapple juice. CYP2C9 is responsible for the metabolic clearance of several drugs, including warfarin and phenytoin, within a restricted therapeutic plasma concentration range [ref. 72]. Pineapple fruit juice, due to its high bromelain content, was found to have the most pronounced inhibitory properties on CYP2C9 compared to other fruit juices. The effect of the inhibition was proportionally dependent on the increase in the amount of pineapple juice. Moreover, starfruit juice was found to be a very potent inhibitor of CYP3A4 compared to grapefruit juice; specifically, an assay of midazolam 1-hydroxylase activity of human CYP3A showed that residual activity of midazolam 1-hydroxylase (%) with starfruit juice was only 0.1 ± 0.0, compared to the 14.7 ± 0.5 for grapefruit juice. It would be interesting to study these inhibitory activities in vivo to determine the clinical relevance of tropical fruit juices on DI [ref. 73].
In another study, pineapple juice increased celecoxib and montelukast systemic exposure [ref. 74]. Celecoxib is a powerful selective cyclooxygenase-2 (COX-2) inhibitor used to treat inflammatory diseases such as osteoarthritis and rheumatoid arthritis [ref. 75]. It is extensively metabolized in the liver, mostly by CYP2C9, and inhibits CYP2D6, although it is not a substrate for this metabolic enzyme [ref. 76]. These findings corroborated prior reported findings that pineapple juice had an inhibiting impact on CYP2C9.
However, another study examined the potential protective effects of fruit or vegetable juices against the genotoxicity of heterocyclic aromatic amines (HAAs), which were activated by human xenobiotic-metabolizing enzymes produced in immortal mammalian cells, and the results are very promising. They employed genetically modified metabolically active lung fibroblasts of Chinese hamster V79 cells that included human CYP1A2, NAT2*4 and SULT1A1*1, which were obtained from hamster V79 [ref. 77,ref. 78]. Despite the fact that pineapple juices provided only marginal protection against HAA genotoxicity, the results provided evidence of the pineapple juices’ inhibitory potential on the CYP1A2 and NAT2*4 enzymes. To better understand these connections, further in-depth research should be conducted.
Thus, the inhibitory impact on CYP2C9 is thought to be the major mechanism underlying pineapple juice interactions, while minor interplay with CYP1A2 and NAT should also be considered. It is reasonable to conclude that pineapple is the safest herbal product to consume among all others; nonetheless, even slight fluctuations in plasma concentrations owing to pineapple interactions may be harmful to individuals taking drugs that are metabolized by the enzyme CYP2C9.
1.3. Andrographis paniculata
Andrographis paniculata, a member of the plant family Acanthaceae, is one of the most prominent medicinal herbs in the world and has been used for centuries in the treatment of a wide range of diseases throughout Asia, America, and Africa. In addition, it contains a number of photochemical compounds with distinct and fascinating biological features. Randomized, double-blinded and multicentre studies investigating the A. paniculata extract have suggested similar efficacy to mesalamine for patients with mild to moderate ulcerative colitis [ref. 15,ref. 79–ref. 82].
Studies have reported that Andrographis paniculata extract and the major component andrographolide greatly alter both the expression and activity of important P450 isoforms, such as CYP1A1, CYP1A2, CYP2A4, CYP2B9, CYP2B10, CYP2C9, CYP2E1 and CYP3A4, in human hepatocytes [ref. 83–ref. 90]. There was a time-dependent increase in the mRNA expression of all major CYP isoforms, e.g., CYP1A2, CYP2C9, CYP2E1 and CYP3A4, in human hepatocytes treated with andrographolide. Thus, a risk assessment of A. paniculata should be conducted, since the expression of genes, especially drug metabolism-related genes, might be affected. Moreover, Andrographis paniculata extract (APE) inhibited the activities of CYP1A2 and CYP2C in rat and human liver microsomes, but this inhibition was not confirmed in vivo [ref. 91–ref. 94]. Furthermore, the metabolism of theophylline by CYP1A2 was inhibited significantly with APE [ref. 95].
Muthiah and colleagues [ref. 96] investigated the effects of several herbs used widely in the southeast, including AP, on the activity of cytochrome P450 2C8 (CYP2C8), an underappreciated human hepatic enzyme involved in drug metabolism. The effects were assessed using amodiaquine N-demethylase activity, which is an in vitro CYP2C8 activity marker. AP had a low IC50 and Ki of 14.8 and 6.26 g/mL, respectively. To the best of our knowledge, this is the first report of AP-induced CYP2C8 suppression. However, the low level of inhibition shown in this investigation suggests that clinically relevant interactions between AP and CYP2C8 substrates are unlikely. The enzyme CYP2C8 is principally responsible for the metabolism of cerivastatin, paclitaxel, rosiglitazone, troglitazone, amodiaquine and chloroquine. Concurrent AP intake may be relevant in drug-herb interactions in vivo through CYP2C8 inhibition. Moreover, interactions between AP and human cytochrome P450 2C19 (CYP2C19), a key hepatic cytochrome P450 isoform implicated in the metabolism of several therapeutic medications, have been studied. Mixed-type inhibition of CYP2C19 was seen in the AP ethanol extract, with moderate Ki values, suggesting that the inhibitors in the extract may bind to both the enzyme and the enzyme–substrate complex, with the same or different affinities [ref. 97]. As a result, even if the interaction between AP and CYP2C19 substrates (such as omeprazole, proguanil, barbiturates, citalopram, and diazepam) is minimal, caution should be used when AP is provided concurrently with these drugs.
Aqueous extracts of Andrographis paniculata at a concentration of 100 g/mL significantly inhibited CYP3A4 (>85%). This result might be attributed to andrographolide, which had good fitting scores in the docking experiment, as well as the presence of various phenolic compounds in the studied extracts [ref. 89]. In addition, by using the fluorogenic CYP assay, Andrographis paniculata and its phytochemicals, such as andrographolide, bacoside A, and asiaticoside, were investigated for their ability to inhibit pooled microsomal CYP450s as well as human recombinant CYP3A4, CYP2D6, CYP2C9, and CYP1A2 enzymes, followed by the determination of the inhibitory concentration (IC50). The most potent inhibitory action was shown by A. paniculata against CYP3A4 and CYP2D6 [ref. 93,ref. 94,ref. 98]. Furthermore, both andrographolide and 14-deoxy-11,12-didehydroandrographolide have been shown to inhibit CYP1A2, CYP2D6, and CYP3A4 expression in HepG2 cells [ref. 87].
As a result, several different types of CYP isoforms have been observed to interact with AP when tested in vitro. It was discovered that this herb inhibited CYP1A2, CYP2C9, and CYP3A4 more than CYP2D6, CYP2E1, and CYP2B isoforms, indicating that this plant had a distinct inhibitory pattern.
1.4. Artemisia absinthium [wormwood
Wormwood, or Artemisia absinthium L., is an important perennial shrubby medicinal plant native to Asia, the Middle East, Europe, and North Africa [ref. 99]. A. absinthium is one of the most significant herbs with a variety of pharmacological properties, including antiinflammatory, antibacterial, antiviral, hypoglycaemic, and hepatoprotective effects, wound healing, and cardiovascular disease prevention [ref. 100–ref. 104]. Despite concerns about the study’s quality due to nonrandomization, insufficient blinding, the short observational duration, and failure to record adverse effects, randomized controlled studies as well as animal model studies proved the impact of wormwood in IBD [ref. 105]. In an open-label study, twenty patients were received either conventional therapy alone or extra Artemisia absinthium for six weeks [ref. 106]. After 6 weeks, patients in the intervention group had a substantial reduction in disease activity and depression, as well as an improvement in quality of life. Within the control group, no significant changes were discovered. Tumour necrosis factor alpha (TNF-α) levels were also observed to be significantly lower in the intervention group. In another study, patients who were administered wormwood had similar benefits, including reduced intensity of symptoms, enhanced quality of life, decreased depression, and greater well-being [ref. 15,ref. 107].
Wormwood contains a variety of phytochemicals, including kaempferol, quercetin, and coumarin, which have been demonstrated to inhibit either cytochrome P450 isoform 3A4 or 2B6, which are responsible for the metabolism of its main ingredient artemisinin in humans [ref. 104,ref. 108,ref. 109]. Artemisinin is mostly an antimalarial drug metabolized by CYP2B6 in the liver, with little contribution from CYP3A4 to produce therapeutically inactive metabolites [ref. 110]. Furthermore, it was discovered that artemisinin suppressed human microsomal CYP2B6 activity but not CYP3A4 activity [ref. 111]. Furthermore, Xing and colleagues [ref. 112] demonstrated that artemisinin medicines are possible inducers of P450 enzymes, with the most inducible enzymes being CYP2B6 and CYP3A4, which are thought to be the key enzymes implicated in artemisinin drug autoinduction. Artemisinin drugs also exhibited mild inhibition of CYP2B6 but not CYP3A4. In addition, artemisinin was reported to inhibit CYP2B6 in vitro through a partly mixed manner of inhibition in another investigation. Artemisinin’s mechanism was neither time-dependent nor NADPH-dependent, suggesting a reversible manner of inhibition. However, the projected degree of CYP2B6 inhibition in vivo by both artemether and artemisinin was suggested to be moderate [ref. 113]. Thus, wormwood may endanger conventional medications processed by CYP2B6 and CYP3A while also enhancing artemisinin bioavailability.
Eupatilin and jaceosidin are pharmacologically active flavones discovered in Artemisia species. Although they are not reported in A. absinthium, similar methoxylated flavones have been reported [ref. 114,ref. 115]. Eupatilin and jaceosidin Eupatilin and jaceosidin were reported to inhibit CYP1A2-catalysed phenacetin O-deethylation with IC50 values of 9.4 μM and 5.3 μM, respectively, as well as CYP2C9-catalysed diclofenac 4-hydroxylation with IC50 values of 4.1 μM and 10.2 μM, respectively. They were also shown to have a considerable inhibitory effect on CYP2C19-catalysed [S-mephenytoin 4′-hydroxylation, CYP2D6-catalysed bufuralol 1′-hydroxylation, and CYP2C8-catalysed amodiaquine N-deethylation. A kinetic study of human liver microsomes suggested that eupatilin and jaceosidin are competitive inhibitors of CYP1A2 and a mixed-type inhibitor of CYP2C9 [ref. 116]. At the highest dose tested (100 μM), eupastilin and jaceosidin demonstrated no inhibition of CYP2A6-catalysed coumarin 7-hydroxylation and CYP3A4-catalysed midazolam 1′-hydroxylation. These in vitro findings imply that wormwood should be investigated further for possible pharmacokinetic drug interactions in vivo owing to CYP1A2 and CYP2C9 suppression.
The modulation of cytochrome P450 activities of Artemisia plant extracts (water or ethanolic) on human cells, as well as the antiinflammatory activity, have been researched by pioneering researchers [ref. 117,ref. 118]. There was a substantial decrease in CYP3A4 activity in all Artemisia plant samples (A. annua, A. afra, A. abrotanum, A. absintium, A. apiacea, A. herba alba, and A. pontica) tested. The amount of CYP3A4 inhibition observed with all Artemisia samples was unexpectedly large, reaching up to 6 times the levels observed in ketoconazol or diluted grapefruit juice. Because grapefruit juice has a reputation for being the most powerful CYP3A4 inhibitor derived from a plant, this is shocking to learn that all Artemisia samples exhibit this high level of CYP3A4 inhibition.
In conclusion, the DDI prediction indicated that Artemisia might result in both induction and inhibition. Additionally, combination medicines (including artemisinin) cleared by CYP2B6 and CYP3A4 and, to a lesser extent, CYP1A2 and CYP2C9 may need a dosage adjustment to sustain treatment or prevent adverse effects. In addition, considering its well-known potential toxic effects [ref. 119] highlighted by the known neurotoxicity of thujones, its utilization should be restricted.
1.5. Boswellia serrata
The Boswellia serrata tree is a moderately large deciduous tree that is native to India, the Middle East, and Northern Africa. When the paper-thin bark of the Boswellia tree is peeled back, sticky oleoresin is revealed. Extracts of this oleoresin have been utilized in Ayurvedic medicine to treat inflammatory diseases and gastrointestinal issues for its antiinflammatory, antiseptic, astringent, and stimulating action [ref. 120–ref. 122]. Studies have shown that patients with chronic colitis who were treated with Boswellia gum resin showed decreased intensity of symptoms and better quality of life [ref. 123,ref. 124]. Clinical, haematological and biochemical parameters exhibited declining trends with complete resolution of ulcers after 6 weeks of treatment [ref. 120,ref. 121,ref. 125,ref. 126].
A type of Ayurvedic medicine, BV-9238, a proprietary formulation of Withania somnifera, Boswellia serrata, Zingiber officinale, and Curcuma longa, was investigated for its toxic effects and cytochrome P450 enzyme inhibition activity [ref. 127. Human recombinant CYP1A2, CYP2C19, CYP2C9, CYP2D6, CYP2E1, and CYP3A4 isomers were tested using 3-cyano-7-ethoxycoumarin as a substrate, while the CYP3A4 isomer employed 7-benzyloxy-4-(trifluoromethyl)-coumarin. Although no particular prototype substrate was used at different doses, CYP2C9 and CYP3A4 were the most susceptible to inhibition, with 35% and 19% inhibition, respectively, at a concentration of 0.5 g/mL of BV-9238, while CYP4502E1, CYP450 2D6, and CYP450 2C9 activities were the least sensitive. Similarly, total boswellic acids were shown to be effective inhibitors, while additional components in Boswellia serrata extract play an important role [ref. 127].
The inhibition of the applied CYP enzymes 1A2/2C8/2C9/2C19/2D6 and 3A4 by the frankincense extracts (Boswellia carteri, Boswellia frereana, Boswellia sacra and Boswellia serrata) was determined using LC/LC/ESI-MS. The boswellic acids 11-keto-β-boswellic acid and acetyl-11-keto-β-boswellic acid showed the highest inhibitory activities for the CYP enzymes 2C8/2C9 and 3A4, with IC50 values in the range of 5 to 10 μM. Since the gum resin of B. frereana also showed a comparable strong inhibition of the applied CYP enzymes, the boswellic acids are obviously not primarily responsible for the potent inhibitory activity of the frankincense extracts. Therefore, phytoconstituents other than boswellic acid and its metabolites exert P450 inhibitory actions. Since herbal extracts of B. serrata are widely used for the treatment of inflammatory diseases, further in vitro and in vivo studies are needed to clarify the clinical relevance of these findings [ref. 128].
The authors provided case reports of suppression of metabolic pathways (CYP2C19, CYP3A4, and CYP2C9) [ref. 129]. Incidences of an interaction between the herb Boswellia serrata and the anticoagulant warfarin have been recorded. Because it inhibits lipoxygenase and interacts with COX-1, it is possible that boswellic acid is the source of the interaction between boswellia and warfarin. Furthermore, boswellia has been shown to inhibit the isoenzymes CYP2C19, CYP3A4 and CYP2C9, which are responsible for the metabolism of warfarin, hence increasing its anticoagulant efficacy [ref. 122,ref. 130,ref. 131]. According to current research, the use of boswellia in patients who have achieved warfarin stabilization is not recommended. Given the estimated number of prescriptions (14,632,370) in the United States in 2019, the boswellic acid interaction is highly critical. Furthermore, Boswellia serrata extract as well as boswellic acids, 11-keto-β-boswellic acid, and acetyl-11-keto-β-boswellic were identified as an additional class of potent P-glycoprotein (Pgp) inhibitors based on the modulation of Pgp-mediated efflux. Thus, it also affects drug transport in the gastrointestinal tract as well as drug metabolism [ref. 132].
It became apparent that Boswellia extracts and frankincense contain several phytochemicals that modulate CYP2C19, CYP3A4 and CYP2C9 as well as Pgp, suggesting unavoidable adverse interactions that may pose serious health consequences. It is obvious that more research is needed to obtain definitive conclusions about Boswellia toxicities in the future.
1.6. Cannabis sativa
Cannabis sativa, usually known as Indian hemp, is an herbaceous annual plant that has been grown mostly in Central Asia (India and China) since antiquity [ref. 133]. C. sativa has been used for millennia as a source of fibre, food, oil, and medicine, as well as for recreational and religious purposes [ref. 134]. It includes many chemically active chemicals, including cannabinoids, terpenoids, flavonoids, and alkaloids [ref. 135,ref. 136]. Δ9-Tetrahydrocannabinol (THC) and cannabidiol (CBD) are the most important phytocannabinoids. Clinical investigations have shown that cannabis has beneficial effects in the gut, as well as the capacity to decrease inflammation, discomfort, and hypermotility, and that it may be utilized in the treatment of ulcerative colitis [ref. 124,ref. 137–ref. 143].
Numerous in vitro and in vivo investigations suggest that cannabis may impact P450 isoenzymes to alter drug metabolism. The comprehensive analysis implicated the role of P450 in the metabolism of various exogenous cannabinoids, including tetrahydrocannabinol (THC; CYP2C9, 2J2; 3A4), cannabidiol (CBD; CYP2C19, 2J2; 3A4), and cannabinol (CBN; CYP2C9, 2J2, 3A4) [ref. 144–ref. 148]. Cannabinoid inhibition or induction of CYP (e.g., THC as a CYP1A2 inducer and CBD as a 3A4 inhibitor) may influence the metabolism of various medications metabolized by these CYPs [ref. 136,ref. 144,ref. 148–ref. 153]. However, the applicability of experimental findings in cells or animals to humans has yet to be proven, and specific clinical trials are required to validate these interactions. The effects of cannabis and its major phytoconstituents are listed as follows:
Cannabis, particularly CBD, has a strong inhibitory effect on CYP1A2 activity. Competitive inhibition of 7-ethoxyresorufin O-deethylase activity catalysed by recombinant hCYP1A2 was observed in the presence of these cannabinoids. CBD was shown to be the most effective inhibitor of CYP1A activity. CBN was also efficient at decreasing the activity of CYP1A2. However, THC was a less powerful inhibitor of CYP1 activity and less selective against CYP1 inhibition than CBD and CBN [ref. 154]. In contrast, marijuana inhalation increased the expression of the CYP1A2 enzyme [ref. 155]. Thus, the route of administration of cannabis is important in exerting selective actions on CYP1A family members. In addition, it was discovered that caffeine metabolism mediated by CYP1A2 was minimally affected in the presence of cannabinoids [ref. 153].
THC, CBN, and CBD were found to be highly effective inhibitors of the CYP2A6 and CYP2C9 enzymes but a weaker inhibitor of CYP2B6. THC and CBN both displayed mechanism-based inhibition of the enzyme CYP2A6. The cannabinoids THC, CBD, and CBN suppressed the coumarin 7-hydroxylase activity of recombinant CYP2A6 in a noncompetitive manner, with apparent Ki at approximately 30–40 μM. THC, CBD, and CBN inhibited recombinant CYP2B6 7-dependent benzoxyresorufin O-debenzylase activity in a mixed fashion in a dose-dependent manner, with very small Ki values at approximately 2 μM, suggesting stronger inhibitory action on CYP2B6 than CYP2A6 [ref. 156]. Similarly, another study demonstrated that the majority of the cannabinoids tested were shown to decrease CYP2B6 activity to a limited extent [ref. 153]. In the presence of THC, CBN, CBD, and CBC, the apparent Vmax of CYP2J2-dependent AEA metabolism was lowered to 20%–45% of that of the uninhibited control enzyme [ref. 145].
A competitive inhibition study of CYP3A revealed that THC, CBD, and CBN were all effective inhibitors, with CBD being the most potent at a concentration comparable to that found in typical cannabis inhalation. CBD was shown to be the most powerful inhibitor of CYP3A4 and CYP3A5 among the three main cannabinoids tested, and it was found to be competitive. The IC50 values for THC or CBN for CYP3A4 or CYP3A5 were quite high. In addition, CYP3A7 activity was suppressed to a comparable level by these cannabinoids but in a mixed manner [ref. 155,ref. 157]. Moreover, CBD competitively prevented the synthesis of 2 alpha-hydroxy-testosterone and 16 alpha-hydroxy-testosterone, which was catalysed by a reconstituted system containing purified hepatic cytochrome P-450. However, it had no effect on the production of androstenedione (androst-4-ene-3,17-dione) or 7 alpha-OH-testosterone. According to the results of the kinetic analyses, CBD had a distinct inhibitory profile for testosterone oxidation in comparison to the profile of SKF 525-A. However, partial suppression of the CYP3A4-dependent metabolism of nifedipine and testosterone has been observed in certain studies [ref. 153].
CYP2C9 was found to be inhibited by major cannabinoids, with the inhibitory effect of CBD being dependent on the substrates used in the experiments. Most of the cannabinoids reduced the CYP2C9-mediated metabolism of tolbutamide by more than 50%, and as a result, they were further investigated. The inhibitory concentrations (IC50) of CBD, CBDV, cannabigerolic acid and THCA varied between 2.5 and 6.4 μM [ref. 153]. Cannabinol (CBN) was the only cannabinoid shown to have no effect on the activity of the CYP2C9 enzyme. Hepatocytes and recombinant CYP2C19 were shown to have decreased (S)-mephenytoin 4′-hydroxylase activity when exposed to CBD in a concentration-dependent mixed manner. Additionally, CBD reduced the activities of recombinant CYP2C19, which catalysed omeprazole 5-hydroxylase and 3-O-methylfluorescein O-demethylase [ref. 153,ref. 155,ref. 158–ref. 160]. It is possible that cannabis usage will inhibit CYP2C9 to a degree that is clinically meaningful for people on warfarin, a very common drug used to reduce the risk of clotting [ref. 147]. Thus, it is highly recommending that such practices be strictly prohibited and that clinical drug-drug interaction research be conducted to determine the impact of marijuana on CYP2C9 activity. Similar to CYP2C9, THC, CBD, and CBN were found to inhibit CYP2D6, with CBD being the most potent when administered at a higher concentration than that found in typical cannabis consumption [ref. 146]. All of these major cannabinoids inhibited the activities of recombinant CYP2D6 and pooled human liver microsomes in a concentration-dependent manner, with CBD demonstrating the greatest inhibitory potency [ref. 161]. However, it has been shown that dextromethorphan O-demethylase activities are only partially inhibited [ref. 153].
In summary, the effects of cannabis and its phytoconstituents on CYPs are undeniable. There is enough evidence to draw firm judgement on the safety of cannabis or cannabidiol in people with UC. To obtain clearer findings on the safety of cannabis usage, studies with greater methodological quality and a larger number of participants with long-term follow-up are necessary. Patients should be told that the use of cannabis and cannabinoids may result in DDIs that have an impact on the effectiveness, tolerability, and safety of their medication.
1.7. Curcuma longa
Turmeric (Curcuma longa L.) is a perennial herb that is a member of the Zingiberaceae family of plants. It is one of the most important Indian spices and natural colouring agents due to its rhizomes. From ancient times to the present, this plant has been widely used for a variety of purposes, including for therapeutic purposes in foods [ref. 162–ref. 166]. Turmeric is also one of the most popular dietary supplements in the world [ref. 167]. Curcuma spp. has yielded over 427 chemical compounds, all of which have been isolated and identified. This genus has a high concentration of flavonoids, tannins, anthocyanin, phenolic compounds, oil, organic acids, and inorganic compounds, among other things. Curcumin, one of the most important active components in Curcuma, is known for its powerful antiinflammatory and antioxidant properties. Furthermore, pharmacological research has shown that Curcuma has a broad variety of actions, including hepatoprotective, antifungal, antihypertensive, and neuroprotective properties [ref. 162, ref. 168–ref. 174]. It has been reported that curcumin-loaded nanocarriers significantly improve CUR’s antiinflammatory properties, enhancing its overall therapeutic efficacy [ref. 175]. Curcumin supplementation demonstrated some additional therapeutic efficacy in patients with UC when taken in conjunction with antiinflammatory medicines [ref. 176–ref. 179]. Conversely, conflicting findings indicated that low-dose oral curcumin was unsuccessful in producing remission in mild to moderate instances of UC [ref. 180]. Thus, curcumin seems to be a potential therapy for sustaining remission in people with quiescent, mild to moderate UC, but further research on curcumin is needed to support the present results.
Curcumin is a competitive inhibitor of CYP1A2 with an IC50 ranging from 40–100 μM according to in vitro testing involving human CYP enzymes produced in the E. coli cell membrane or liver microsomes (Appiah-Opong et al., 2007; Bamba et al., 2011). Likewise, other curcuminoids were found to inhibit CYP1A2, which accounts for approximately 13% of human liver cytochromes, and biotransform arylamine drugs such as theophylline and lidocaine [ref. 127,ref. 181–ref. 186]. However, human liver CYP1A2-dependent phenacetin O-deethylation activity has been reported to remain unaltered with curcumonol [ref. 187].
Curcumin at concentrations ranging from 0.9 to 100 μM was shown to be a competitive inhibitor of the CYP2B6 enzyme, a member of the CYP2 family that plays an important role in chemotherapeutic drug metabolism. For example, cyclophosphamide [ref. 181,ref. 182,ref. 188–ref. 191] Curcuma extract reduced CYP2C19 activity as a noncompetitive inhibitor with an IC50 of 4.3−7.4 μM; CYP2C19 is an isoform involved in the biotransformation of approximately 20% of the medications on the market [ref. 183,ref. 187,ref. 189,ref. 192–ref. 195].
In human epithelial colorectal adenocarcinoma cells, turmeric extracts were found to inhibit the activity of the intestinal CYP3A4 isoform by approximately 50% [ref. 196]. Pure curcumin also induced 30%–40% inhibition of the CYP3A4 isoenzyme in Caco-2 cells, similar to the results obtained with curcumin extract [ref. 189,ref. 196,ref. 197]. Curcumin was shown to be a competitive inhibitor of the CYP3A4 isoform. In addition, other in vitro investigations of CYP isoforms have revealed the inhibitory effects of curcumin and other curcuminoids [ref. 127 ref. 184,ref. 186,ref. 187,ref. 190,ref. 192,ref. 198,ref. 199]. Several additional studies utilizing intestinal human colon cancer cell lines, human hepatocytes, and HepG2 cells likewise failed to find any significant effects of curcumin on the mRNA expression of CYP3A4 [ref. 181,ref. 200–ref. 202]. The CYP3A4 isoform is the most notable CYP in that it is the primary drug-metabolizing enzyme in liver and intestinal tissue, catalysing the metabolism of half of all commercially available pharmaceuticals [ref. 203–ref. 205]. In addition to its significant role in first-pass metabolism, it plays a significant role in the metabolism of endogenous substrates such as cholesterol and bile acids [ref. 206,ref. 207]. Thus, well-designed clinical studies in healthy volunteers as well as patients are required to obtain an acceptable level of evidence required to establish curcumin-CYP3A4 interactions in humans.
The effect of curcumonoids on CYP enzymes (e.g., CYP17A1, CYP19A1, and CYP21A2) involved in the steroidogenic pathway was also investigated in another research study. Both the 17-hydroxylase and 17,20-lyase activities of CYP17A1 and the aromatase activity of CYP19A1 were considerably inhibited [ref. 191,ref. 208]. Analogously, curcumin extract was shown to strongly inhibit 5aR1, an enzyme implicated in the NADPH-dependent reduction of steroids [ref. 209]. Therefore, although they are not involved in drug metabolism, studies have shown that curcuma and curcuminoids may induce suppression of steroid metabolism as well as perturbation of the related processes. A critical concern in the use of turmeric is the inaccurate categorization of Curcuma species, which occurs as a result of the large number of closely related species. Because of this, Curcuma species (Zingiberaceae) have been misclassified, resulting in unintended human exposure to phytochemicals that alter CYP expression and activity [ref. 210]. Additionally, it was reported that NAT activity in human A549 cells and cytosols was suppressed by curcumin in a dose-dependent manner; NAT is responsible for the metabolism of 5-ASA to inactive metabolites. The results also demonstrated that NAT1 mRNA expression was inhibited and decreased by curcumin.
As a result, curcuma and its main constituent curcumin interact with the drug-metabolizing enzymes CYP1A2, 2B6, 2C9 and 3A4, NAT, CYP17A1 and 19A1. It affects the pharmacokinetics of medications taken concurrently in conjunction with it, despite its general public safety perception.
1.8. Germinated barley foodstuff/wheat grass juice
Germinated barley foodstuff (GBF), which is composed of dietary fibre and glutamine-rich protein that serves as a probiotic, has been shown to lessen the recurrence rate and clinical reduction in disease activity in patients [ref. 211–ref. 214]. On the basis of its high water-holding capacity and ability to control microbiota, it has been hypothesized that GBF may play a significant role in the prolongation of remission and therapy in UC [ref. 16,ref. 212,ref. 215–ref. 217]. Similarly, administration of wheat grass juice resulted in considerably reduced disease activity, decreased rectal bleeding, and decreased stomach discomfort. Because this inquiry was intended to be a pilot study, the findings may serve as a foundation for more clinical studies in the future with larger sample numbers, healthy control groups, and higher dosages of GBF and WGJ [ref. 15,ref. 218].
There have been no reported interactions between Hordeum vulgare and Triticum aestivum with any of the P450 isoforms that are involved in the metabolism of drugs. As a result, they are considered to be the safest alternatives to conventional drugs for the treatment of UC.
1.9. Indigo naturalis
Indigo naturalis (IN) is an herbal medication made from the leaves and stems of plants such as Indigofera tinctoria, Strobilanthes cusia, and Polygonum tinctorium. It is used to treat a variety of diseases, including UC. IN is an herbal medicine that is quality controlled in China if it contains more than 2% indigo and 0.13% indirubin, which is the legal limit. IN that originates in Fujian is often considered to be of the best quality. In randomized, placebo-controlled clinical trials, IN was shown to be useful in the treatment of ulcerative colitis [ref. 219–ref. 224]. Unfortunately, even though there is evidence that IN may be beneficial in the treatment of UC, there are many unknowns, including the effective components and therapeutic targets that remain to be discovered. A recent study applied a network pharmacology method to anticipate the molecular mechanism that underlies the action and the effective components of IN in the treatment of UC in general [ref. 225].
There are numerous unknowns about both the efficacy of IN for UC and the interaction of IN with drug metabolizing enzymes. The data that are currently available, even if limited, indicate that IN may offer a considerable threat when used in conjunction with currently utilized pharmaceuticals [ref. 224,ref. 226]. CYP1A1 expression in colon tissue from six patients was found to have increased by a factor of 12.557 after 8 weeks of IN treatment [ref. 224]. However, although CYP1A1 is involved in the metabolism of a few drugs, such as theophylline, it is mostly engaged in the metabolic activation of aromatic hydrocarbons into potentially carcinogenic compounds [ref. 227,ref. 228]. Furthermore, both indigo and indirubin were discovered to be AhR ligands, implying that the expression of genes controlled by AhR, including CYP1A2, is changed [ref. 229–ref. 231]. In addition, indirubin activated the transcription of the CYP3A4 gene in HepG2 cells via the activation of the PXR gene. More in-depth investigations into the molecular mechanism of indirubin-induced CYP3A4 activation may provide vital insights into the interactions between herbs and drugs as well as drug-drug interactions [ref. 232]. Another study sought to determine the possible effects of Realgar-Indigo naturalis (RIN) on the activities of four CYP450 isozymes, specifically the activities of CYP1A2, CYP2C11, CYP2E1, and CYP3A1/2 in rats. The findings revealed that RIN has the ability to considerably suppress CYP1A2 enzyme activity while simultaneously increasing CYP2C11 enzyme activity. Unexpectedly, the RIN high dosage group showed considerable inhibition of CYP3A1/2 enzyme activity, with various dosages exhibiting a good dose-dependent response [ref. 233].
Thus, all these limited findings of IN suggested that drugs administered in conjunction with IN may need special consideration in terms of drug-drug interactions, and further detailed studies are required.
1.10. Miscellaneous
A number of other herbal products exhibiting antiinflammatory properties are reportedly used in the treatment of UC, and various preclinical and clinical studies are currently available. Among the most notable plants that have been utilized are Bletilla striata, Capparis ovata, Commiphora wightii, Cynara scolymus, Fumaria officinalis, Glycyrrhiza glabra, Glycyrrhiza uralensis, Hypericum perforatum, Mentha piperita, Momordica charantia, Oenothera biennis, Plantago ovata, Plantago psyllium, Potentilla erecta, Sanguisorba officinalis, Sophorae flavescentis, Sorbus domestica, Strobilanthes cusia, and Withania somnifera [ref. 13,ref. 15,ref. 16,ref. 220,ref. 234–ref. 251]. However, due to methodological flaws, heterogeneity in the definition of diagnostic criteria, and the absence of placebo control and blinded measurement of subjective conclusions, the majority of trials associating herbal medicines with conventional therapies have not provided significant evidence to support the use of herbal medicines. In addition, it is unfortunate that there are no detailed studies on the occurrence of acute or chronic side effects and an understanding of more subtle and potentially long-term consequences involving DME interactions. There is a paucity of credible evidence on the effectiveness and safety of the majority of these plants in UC patients. Therefore, the exact mechanisms of action of these herbal products and their interaction with the various cytochrome P-450 isoforms remain a mystery at this time.
2. Conclusion
The most notable characteristics of traditional herbal medicines are undoubtedly their multicomponent and multieffect natures. Overall, the clinical relevance of the CYP system is well known in drug-drug interactions as well as herbal medicine-drug interactions because various xenobiotics, including those found in traditional herbal medicines, can influence their activity and thus have an impact on systemic drug exposure, i.e. efficacy and toxicity. As shown by the studies examined in this review, herbal medicines used to treat ulcerative colitis are not exempt from this phenomenon (Table).
Table: An overview how the herbal products used to treat ulcerative colitis affect major drug-metabolising enzymes.
| Herbal Product | Major drug metabolising CYPs | Others | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1A2 | 2B6 | 2C9 | 2C19 | 2D6 | 3A4 | 1A1 | 2B1/9 | 2E1 | NAT | |
| Aloe vera | ↓↓↓ | − | ↓↓↓ | − | ↓↓ | ↓↓↓ | ↓↓ | ↓ | ↓↓↓ | ↓↓ |
| Ananas comosus | ↓ | − | ↓↓↓ | − | − | ↓↓↓ | − | − | − | ↓ |
| Andrographis paniculata | ↓↓↓ | − | ↓↓ | ↓↓ | ↓ | ↓↓↓ | ↓ | ↓ | ↓↓ | − |
| Artemisia absinthium | ↓ | ↓↓↓ | ↓ | ↓↓ | ↓↓ | ↓↓↓ | − | − | − | − |
| Boswellia serrata | − | − | ↓↓ | ↓↓ | − | ↓↓ | − | − | − | − |
| Cannabis sativa | ↑/↓↓ | ↓ | ↓↓↓ | ↓↓ | ↓↓ | ↓↓↓ | − | − | − | − |
| Curcuma longa | ↓↓ | ↓↓ | − | ↓↓↓ | − | ↓↓↓ | − | − | − | ↓↓↓ |
| GBF/WGJ | − | − | − | − | − | − | − | − | − | − |
| Indigo naturalis | ↓ | − | − | − | − | ↑↑/↓↓↓ | ↑↑↑↑ | − | ↑ | − |
−: no data, one-arrow: weak, two-arrow: moderate; three-arrow: strong; ↑: increase; ↓: decrease.
In conclusion, despite their possible antiinflammatory properties, herbal products should not be prescribed due to their interactions with drug-metabolizing enzymes and adverse events, according to the canonical primum nonnocere (first, do no harm) medical concern. Moreover, considering the polymorphisms in human CYP genes, which largely alter the function of the enzymes, the prediction of adverse effects is almost impossible. Thus, physicians should encourage their patients to declare their use of complementary medicines to avoid significant consequences that may result from concurrent use of conventional pharmaceuticals.
References
- E Szigethy, L McLafferty, A Goyal. Inflammatory bowel disease. Child and Adolescent Psychiatric Clinics of North America, 2010. [DOI | PubMed]
- T Kobayashi, B Siegmund, C Le Berre, SC Wei, M Ferrante, B Shen, CN Bernstein, S Danese, L Peyrin-Biroulet, T Hibi. Ulcerative colitis. Nature Reviews Disease Primers, 2020. [DOI | PubMed]
- SB Hanauer. Inflammatory bowel disease: epidemiology, pathogenesis, and therapeutic opportunities. Inflammatory Bowel Diseases, 2006. [DOI | PubMed]
- YZ Zhang, YY Li. Inflammatory bowel disease: pathogenesis. World Journal of Gastroenterology, 2014. [DOI | PubMed]
- JT Chang. Pathophysiology of Inflammatory Bowel Diseases. The New England Journal of Medicine, 2020. [DOI | PubMed]
- M Harbord, R Eliakim, D Bettenworth, K Karmiris, K Katsanos, U Kopylov, T Kucharzik, T Molnar, T Raine, S Sebastian, HT de Sousa, A Dignass, F Carbonnel, Cs European, O Colitis. Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 2: Current Management. Journal of Crohn’s and Colitis, 2017. [DOI | PubMed]
- K Matsuoka, T Kobayashi, F Ueno, T Matsui, F Hirai, N Inoue, J Kato, K Kobayashi, K Kobayashi, K Koganei, R Kunisaki, S Motoya, M Nagahori, H Nakase, F Omata, M Saruta, T Watanabe, T Tanaka, T Kanai, Y Noguchi, KI Takahashi, K Watanabe, T Hibi, Y Suzuki, M Watanabe, K Sugano, T Shimosegawa. Evidence-based clinical practice guidelines for inflammatory bowel disease. Journal of Gastroenterology, 2018. [DOI | PubMed]
- RD Cohen, AP Yu, EQ Wu, J Xie, PM Mulani, J Chao. Systematic review: the costs of ulcerative colitis in Western countries. Alimentary Pharmacology and Therapeutics, 2010. [DOI | PubMed]
- KD Null, Y Xu, MK Pasquale, C Su, A Marren, J Harnett, J Mardekian, A Manuchehri, P Healey. Ulcerative Colitis Treatment Patterns and Cost of Care. Value Health, 2017. [DOI | PubMed]
- KT Park, OG Ehrlich, JI Allen, P Meadows, EM Szigethy, K Henrichsen, SC Kim, RC Lawton, SM Murphy, M Regueiro, DT Rubin, NM Engel-Nitz, CA Heller. The Cost of Inflammatory Bowel Disease: An Initiative From the Crohn’s & Colitis Foundation. Inflammatory Bowel Diseases, 2020. [DOI | PubMed]
- S Alulis, K Vadstrup, J Olsen, TR Jorgensen, N Qvist, P Munkholm, A Borsi. The cost burden of Crohn’s disease and ulcerative colitis depending on biologic treatment status – a Danish register-based study. BMC Health Services Research, 2021. [DOI | PubMed]
- RJ Hilsden, MJ Verhoef, H Rasmussen, A Porcino, JC DeBruyn. Use of complementary and alternative medicine by patients with inflammatory bowel disease. Inflammatory Bowel Diseases, 2011. [DOI | PubMed]
- F Ke, PK Yadav, LZ Ju. Herbal medicine in the treatment of ulcerative colitis. Saudi Journal of Gastroenterology, 2012. [DOI | PubMed]
- SC Ng, YT Lam, KK Tsoi, FK Chan, JJ Sung, JC Wu. Systematic review: the efficacy of herbal therapy in inflammatory bowel disease. Alimentary Pharmacology and Therapeutics, 2013. [DOI | PubMed]
- J Langhorst, H Wulfert, R Lauche, P Klose, H Cramer, GJ Dobos, J Korzenik. Systematic review of complementary and alternative medicine treatments in inflammatory bowel diseases. Journal of Crohn’s and Colitis, 2015. [DOI | PubMed]
- EK Akkol, B Karpuz, E Sobarzo-Sanchez, H Khan. A phytopharmacological overview of medicinal plants used for prophylactic and treatment of colitis. Food and Chemical Toxicology, 2020. [DOI | PubMed]
- V Dubey, S Singh. Alternative Herbal Medicines For Prevention And Treatment Of Ulcerative Colitis. European Journal of Molecular and Clinical Medicine, 2020
- S Picardo, M Altuwaijri, SM Devlin, CH Seow. Complementary and alternative medications in the management of inflammatory bowel disease. Therapeutic Advances in Gastroenterology, 2020. [DOI | PubMed]
- EC Miller. Some current perspectives on chemical carcinogenesis in humans and experimental animals: Presidential Address. Cancer Research, 1978. [PubMed]
- FP Guengerich. Roles of cytochrome P-450 enzymes in chemical carcinogenesis and cancer chemotherapy. Cancer Research, 1988. [PubMed]
- FP Guengerich, DH Kim. In vitro inhibition of dihydropyridine oxidation and aflatoxin B1 activation in human liver microsomes by naringenin and other flavonoids. Carcinogenesis, 1990. [DOI | PubMed]
- E Arinc, A Sen. Effects of in vivo benzene treatment on cytochrome-p450 and mixed-function oxidase activities of gilthead seabream (Sparus-aurata) liver-microsomes. Comparative Biochemistry and Physiology Part C: Toxicology and Pharmacology, 1993. [DOI]
- E Arinc, A Sen. In-vivo effects of the anesthetic, benzocaine, on liver microsomal cytochrome-P450 and mixed-function oxidase activities of gilthead seabream (Sparus-aurata). Comparative Biochemistry and Physiology Part C: Pharmacology, Toxicology and Endocrinology, 1994. [DOI]
- M Ozkarsli, H Sevim, A Sen. In vivo effects of Urtica urens (dwarf nettle) on the expression of CYP1A in control and 3-methylcholanthrene-exposed rats. Xenobiotica, 2008. [DOI | PubMed]
- GG Gibson, P Skett. Pharmacological and toxicological aspects of drug metabolism. Introduction to Drug Metabolism, 1986
- A Sen, E Arinc. Purification and Characterization of Cytochrome P450 Reductase from Liver Microsomes of Feral Leaping Mullet (Liza saliens). Journal of Biochemical and Molecular Toxicology, 1998. [DOI | PubMed]
- FP Guengerich, N Meanwell, GI Georg, S Wang. Introduction: Drug Metabolism and Toxicology Special Issue. Journal of Medicinal Chemistry, 2020. [DOI | PubMed]
- DR Nelson. The cytochrome p450 homepage. Human Genomics, 2009. [DOI | PubMed]
- T Omura. Structural diversity of cytochrome P450 enzyme system. Journal of Biochemistry, 2010. [DOI | PubMed]
- A Gaedigk, M Ingelman-Sundberg, NA Miller, JS Leeder, M Whirl-Carrillo, TE Klein, C PharmVar Steering. The Pharmacogene Variation (PharmVar) Consortium: Incorporation of the Human Cytochrome P450 (CYP) Allele Nomenclature Database. Clinical Pharmacology and Therapeutics, 2018. [DOI | PubMed]
- O Buyukgoze, N Osmanoglu, S Arslan, A Sen. Association of the CYP1A1*2A, GSTT1 null, GSTM1 null, mEPHX*3, and XRCC1–399 genetic polymorphisms with ulcerative colitis. International Journal of Colorectal Disease, 2013. [DOI | PubMed]
- A Sen, H Stark. Role of cytochrome P450 polymorphisms and functions in development of ulcerative colitis. World Journal of Gastroenterology, 2019. [DOI | PubMed]
- MH Radha, NP Laxmipriya. Evaluation of biological properties and clinical effectiveness of Aloe vera: A systematic review. Journal of Traditional and Complementary Medicine, 2015. [DOI | PubMed]
- M Akaberi, Z Sobhani, B Javadi, A Sahebkar, SA Emami. Therapeutic effects of Aloe spp. in traditional and modern medicine: A review. Biomedicine and Pharmacotherapy, 2016. [DOI | PubMed]
- S Bendjedid, S Lekmine, A Tadjine, R Djelloul, C Bensouici. Analysis of phytochemical constituents, antibacterial, antioxidant, photoprotective activities and cytotoxic effect of leaves extracts and fractions of Aloe vera. Biocatalysis and Agricultural Biotechnology, 2021. [DOI]
- L Lucini, M Pellizzoni, R Pellegrino, GP Molinari, G Colla. Phytochemical constituents and in vitro radical scavenging activity of different Aloe species. Food Chemistry, 2015. [DOI | PubMed]
- YN Sun, W Li, SY Yang, JS Kang, JY Ma, YH Kim. Isolation and identification of chromone and pyrone constituents from Aloe and their anti-inflammatory activities. Journal of Functional Foods, 2016. [DOI]
- PT Mpiana, KT Ngbolua, DST Tshibangu, JT Kilembe, BZ Gbolo, DT Mwanangombo, CL Inkoto, EM Lengbiye, CM Mbadiko, A Matondo, GN Bongo, DD Tshilanda. Identification of potential inhibitors of SARS-CoV-2 main protease from Aloe vera compounds: A molecular docking study. Chemical Physics Letters, 2020. [DOI | PubMed]
- SO Amoo, AO Aremu, J Van Staden. Unraveling the medicinal potential of South African Aloe species. Journal of Ethnopharmacology, 2014. [DOI | PubMed]
- M Cardarelli, Y Rouphael, M Pellizzoni, G Colla, L Lucini. Profile of bioactive secondary metabolites and antioxidant capacity of leaf exudates from eighteen Aloe species. Industrial Crops and Products, 2017. [DOI]
- SO Muller, I Eckert, WK Lutz, H Stopper. Genotoxicity of the laxative drug components emodin, aloe-emodin and danthron in mammalian cells: topoisomerase II mediated?. Mutation Research, 1996. [DOI | PubMed]
- S Choi, M-H Chung. A review on the relationship between aloe vera components and their biologic effects. Seminars in Integrative Medicine, 2003. [DOI]
- HN Yang, DJ Kim, YM Kim, BH Kim, KM Sohn, MJ Choi, YH Choi. Aloe-induced toxic hepatitis. Journal of Korean Medical Science, 2010. [DOI | PubMed]
- R Kumar, AK Singh, A Gupta, A Bishayee, AK Pandey. Therapeutic potential of Aloe vera-A miracle gift of nature. Phytomedicine, 2019. [DOI | PubMed]
- Y Cui, Y Cheng, Y Guo, Y Xie, W Yao, W Zhang, H Qian. Evaluating the hepatoprotective efficacy of Aloe vera polysaccharides against subchronic exposure of aflatoxins B 1. Journal of the Taiwan Institute of Chemical Engineers, 2017. [DOI]
- A Djuv, OG Nilsen. Aloe vera juice: IC(5)(0) and dual mechanistic inhibition of CYP3A4 and CYP2D6. Phytotherapy Research, 2012. [DOI | PubMed]
- MS Yang, CP Yu, CY Huang, PL Chao, SP Lin, YC Hou. Aloe activated P-glycoprotein and CYP 3A: a study on the serum kinetics of aloe and its interaction with cyclosporine in rats. Food and Function, 2017. [DOI | PubMed]
- M Ingelman-Sundberg. Genetic polymorphisms of cytochrome P450 2D6 (CYP2D6): clinical consequences, evolutionary aspects and functional diversity. The Pharmacogenomics Journal, 2005. [DOI | PubMed]
- T Lynch, A Price. The effect of cytochrome P450 metabolism on drug response, interactions, and adverse effects. American Family Physician, 2007. [PubMed]
- B Sultana, F Anwar. Flavonols (kaempeferol, quercetin, myricetin) contents of selected fruits, vegetables and medicinal plants. Food Chemistry, 2008. [DOI | PubMed]
- N He, T Edeki. The inhibitory effects of herbal components on CYP2C9 and CYP3A4 catalytic activities in human liver microsomes. American Journal of Therapeutics, 2004. [DOI | PubMed]
- P Gougis, M Hilmi, A Geraud, O Mir, C Funck-Brentano. Potential cytochrome P450-mediated pharmacokinetic interactions between herbs, food, and dietary supplements and cancer treatments. Critical Reviews in Oncology/Hematology, 2021. [DOI | PubMed]
- YX Zhang, JS Li, WW Peng, X Liu, GM Yang, LH Chen, BC Cai. Comparative pharmacokinetics of aloe-emodin, rhein and emodin determined by liquid chromatography-mass spectrometry after oral administration of a rhubarb peony decoction and rhubarb extract to rats. Pharmazie, 2013. [PubMed]
- J Sridhar, J Liu, M Foroozesh, CL Klein Stevens. Inhibition of cytochrome p450 enzymes by quinones and anthraquinones. Chemical Research in Toxicology, 2012. [DOI | PubMed]
- Y Liu, MST Mapa, RL Sprando. Anthraquinones inhibit cytochromes P450 enzyme activity in silico and in vitro. Journal of Applied Toxicology, 2021. [DOI | PubMed]
- W Jiang, X Tian, Y Wang, Z Sun, P Dong, C Wang, X Huo, B Zhang, S Huang, S Deng, X Wang, X Ma. The natural anthraquinones from Rheum palmatum induced the metabolic disorder of melatonin by inhibiting human CYP and SULT enzymes. Toxicology Letters, 2016. [DOI | PubMed]
- X Wang, X Xin, Y Sun, L Zou, H Li, Y Zhao, R Li, Y Peng, J Zheng. Chemical Reactivity of Aloe-Emodin and Its Hydroxylation Metabolites to Thiols. Chemical Research in Toxicology, 2019. [DOI | PubMed]
- J Fei, Y Peng, H Tan, X Chen, J Yang, J Li. Study on the electrochemical behavior and differential pulse voltammetric determination of rhein using a nanoparticle composite film-modified electrode. Bioelectrochemistry, 2007. [DOI | PubMed]
- JC Tang, H Yang, XY Song, XH Song, SL Yan, JQ Shao, TL Zhang, JN Zhang. Inhibition of cytochrome P450 enzymes by rhein in rat liver microsomes. Phytotherapy Research, 2009. [DOI | PubMed]
- RP Singh, S Dhanalakshmi, AR Rao. Chemomodulatory action of Aloe vera on the profiles of enzymes associated with carcinogen metabolism and antioxidant status regulation in mice. Phytomedicine, 2000. [DOI | PubMed]
- SY Lin, JH Yang, TC Hsia, JH Lee, TH Chiu, YH Wei, JG Chung. Effect of inhibition of aloe-emodin on N-acetyltransferase activity and gene expression in human malignant melanoma cells (A375.S2). Melanoma Research, 2005. [DOI | PubMed]
- JG Chung, YC Li, YM Lee, JP Lin, KC Cheng, WC Chang. Aloe-emodin inhibited N-acetylation and DNA adduct of 2-aminofluorene and arylamine N-acetyltransferase gene expression in mouse leukemia L 1210 cells. Leukemia Research, 2003. [DOI | PubMed]
- A Hassan, Z Othman, J Siriphanich. Pineapple (Ananas comosus L Merr). Postharvest Biology and Technology of Tropical and Subtropical Fruits, 2011
- RPD Nascimento, A Moya, A Machado, MV Geraldi, P Diez-Echave, T Vezza, J Galvez, CBB Cazarin, MR Marostica Junior. Review on the potential application of non-phenolic compounds from native Latin American food byproducts in inflammatory bowel diseases. Food Research International, 2021. [DOI | PubMed]
- AB Thomson, M Keelan, A Thiesen, MT Clandinin, M Ropeleski, GE Wild. Small bowel review: normal physiology part 1. Digestive Diseases and Sciences, 2001. [DOI | PubMed]
- LC de Lencastre Novaes, AF Jozala, AM Lopes, V de Carvalho Santos-Ebinuma, PG Mazzola, A Pessoa Junior. Stability, purification, and applications of bromelain: A review. Biotechnology Progress, 2016. [DOI | PubMed]
- AD Blackwood, J Salter, PW Dettmar, MF Chaplin. Dietary fibre, physicochemical properties and their relationship to health. The Journal of the Royal Society for the Promotion of Health, 2000. [DOI | PubMed]
- LP Hale, PK Greer, CT Trinh, MR Gottfried. Treatment with oral bromelain decreases colonic inflammation in the IL-10-deficient murine model of inflammatory bowel disease. Clinical Immunology, 2005. [DOI | PubMed]
- K Bhui, S Prasad, J George, Y Shukla. Bromelain inhibits COX-2 expression by blocking the activation of MAPK regulated NF-kappa B against skin tumor-initiation triggering mitochondrial death pathway. Cancer Letters, 2009. [DOI | PubMed]
- Z Zhou, L Wang, P Feng, L Yin, C Wang, S Zhi, J Dong, J Wang, Y Lin, D Chen, Y Xiong, J Peng. Inhibition of Epithelial TNF-alpha Receptors by Purified Fruit Bromelain Ameliorates Intestinal Inflammation and Barrier Dysfunction in Colitis. Frontiers in Immunology, 2017. [DOI | PubMed]
- KY Li, JL Wang, JP Wei, SY Gao, YY Zhang, LT Wang, G Liu. Fecal microbiota in pouchitis and ulcerative colitis. World Journal of Gastroenterology, 2016. [DOI | PubMed]
- M Hidaka, M Nagata, Y Kawano, H Sekiya, H Kai, K Yamasaki, M Okumura, K Arimori. Inhibitory effects of fruit juices on cytochrome P450 2C9 activity in vitro. Bioscience, Biotechnology, and Biochemistry, 2008. [DOI | PubMed]
- Z Petric, I Zuntar, P Putnik, D Bursac Kovacevic. Food-Drug Interactions with Fruit Juices. Foods, 2020. [DOI | PubMed]
- SA Helmy, HM El-Bedaiwy, SM El-Masry. Effect of pineapple juice on the pharmacokinetics of celecoxib and montelukast in humans. Therapeutic Delivery, 2020. [DOI | PubMed]
- SK Paulson, MB Vaughn, SM Jessen, Y Lawal, CJ Gresk, B Yan, TJ Maziasz, CS Cook, A Karim. Pharmacokinetics of celecoxib after oral administration in dogs and humans: effect of food and site of absorption. Journal of Pharmacology and Experimental Therapeutics, 2001. [PubMed]
- U Werner, D Werner, T Rau, MF Fromm, B Hinz, K Brune. Celecoxib inhibits metabolism of cytochrome P450 2D6 substrate metoprolol in humans. Clinical Pharmacology and Therapeutics, 2003. [DOI | PubMed]
- Y Yanagawa, M Sawada, T Deguchi, FJ Gonzalez, T Kamataki. Stable expression of human CYP1A2 and N-acetyltransferases in Chinese hamster CHL cells: mutagenic activation of 2-amino-3-methylimidazo4,5-fquinoline and 2-amino-3,8-dimethylimidazo4,5-fquinoxaline. Cancer Research, 1994. [PubMed]
- KL Platt, R Edenharder, S Aderhold, E Muckel, H Glatt. Fruits and vegetables protect against the genotoxicity of heterocyclic aromatic amines activated by human xenobiotic-metabolizing enzymes expressed in immortal mammalian cells. Mutation Research/Genetic Toxicology and Environmental Mutagenesis, 2010. [DOI | PubMed]
- T Tang, SR Targan, ZS Li, C Xu, VS Byers, WJ Sandborn. Randomised clinical trial: herbal extract HMPL-004 in active ulcerative colitis – a double-blind comparison with sustained release mesalazine. Alimentary Pharmacology and Therapeutics, 2011. [DOI | PubMed]
- WJ Sandborn, SR Targan, VS Byers, DA Rutty, H Mu, X Zhang, T Tang. Andrographis paniculata extract (HMPL-004) for active ulcerative colitis. The American Journal of Gastroenterology, 2013. [DOI | PubMed]
- L Zhang, N Cao, Y Wang, Y Wang, C Wu, X Cheng, C Wang. Improvement of Oxazolone-Induced Ulcerative Colitis in Rats Using Andrographolide. Molecules, 2019. [DOI | PubMed]
- V Vetvicka, L Vannucci. Biological properties of andrographolide, an active ingredient of Andrographis Paniculata: a narrative review. Annals of Translational Medicine, 2021. [DOI | PubMed]
- J George, B Goodwin, C Liddle, M Tapner, GC Farrell. Time-dependent expression of cytochrome P450 genes in primary cultures of well-differentiated human hepatocytes. Journal of Laboratory and Clinical Medicine, 1997. [DOI | PubMed]
- C Liddle, BJ Goodwin, J George, M Tapner, GC Farrell. Separate and interactive regulation of cytochrome P450 3A4 by triiodothyronine, dexamethasone, and growth hormone in cultured hepatocytes. The Journal of Clinical Endocrinology and Metabolism, 1998. [DOI | PubMed]
- E LeCluyse, A Madan, G Hamilton, K Carroll, R DeHaan, A Parkinson. Expression and regulation of cytochrome P450 enzymes in primary cultures of human hepatocytes. Journal of Biochemical and Molecular Toxicology, 2000. [DOI | PubMed]
- W Chatuphonprasert, K Jarukamjorn, S Kondo, N Nemoto. Synergistic increases of metabolism and oxidation-reduction genes on their expression after combined treatment with a CYP1A inducer and andrographolide. Chemico-Biological Interactions, 2009. [DOI | PubMed]
- JP Ooi, M Kuroyanagi, SF Sulaiman, TS Muhammad, ML Tan. Andrographolide and 14-deoxy-11, 12-didehydroandrographolide inhibit cytochrome P450s in HepG2 hepatoma cells. Life Sciences, 2011. [DOI | PubMed]
- ML Tan, LE Lim. The effects of Andrographis paniculata (Burm.f.) Nees extract and diterpenoids on the CYP450 isoforms’ activities, a review of possible herb-drug interaction risks. Drug and Chemical Toxicology, 2015. [DOI | PubMed]
- ML Ashour, FS Youssef, HA Gad, M Wink. Inhibition of Cytochrome P450 (CYP3A4) Activity by Extracts from 57 Plants Used in Traditional Chinese Medicine (TCM). Pharmacognosy Magazine, 2017. [DOI | PubMed]
- CC Yen, YT Liu, YJ Lin, YC Yang, CC Chen, HT Yao, HW Chen, CK Lii. Bioavailability of the diterpenoid 14-deoxy-11,12-didehydroandrographolide in rats and up-regulation of hepatic drug-metabolizing enzyme and drug transporter expression. Phytomedicine, 2019. [DOI | PubMed]
- K Jarukamjorn, K Don-in, C Makejaruskul, T Laha, S Daodee, P Pearaksa, BO Sripanidkulchai. Impact of Andrographis paniculata crude extract on mouse hepatic cytochrome P450 enzymes. Journal of Ethnopharmacology, 2006. [DOI | PubMed]
- A Jaruchotikamol, K Jarukamjorn, W Sirisangtrakul, T Sakuma, Y Kawasaki, N Nemoto. Strong synergistic induction of CYP1A1 expression by andrographolide plus typical CYP1A inducers in mouse hepatocytes. Toxicology and Applied Pharmacology, 2007. [DOI | PubMed]
- D Pekthong, H Martin, C Abadie, A Bonet, B Heyd, G Mantion, L Richert. Differential inhibition of rat and human hepatic cytochrome P450 by Andrographis paniculata extract and andrographolide. Journal of Ethnopharmacology, 2008. [DOI | PubMed]
- D Pekthong, N Blanchard, C Abadie, A Bonet, B Heyd, G Mantion, A Berthelot, L Richert, H Martin. Effects of Andrographis paniculata extract and Andrographolide on hepatic cytochrome P450 mRNA expression and monooxygenase activities after in vivo administration to rats and in vitro in rat and human hepatocyte cultures. Chemico-Biological Interactions, 2009. [DOI | PubMed]
- CF Chien, YT Wu, WC Lee, LC Lin, TH Tsai. Herb-drug interaction of Andrographis paniculata extract and andrographolide on the pharmacokinetics of theophylline in rats. Chemico-Biological Interactions, 2010. [DOI | PubMed]
- YD Muthiah, CE Ong, SA Sulaiman, R Ismail. Inhibition of Human Cytochrome P450 2c8-catalyzed Amodiaquine N-desethylation: Effect of Five Traditionally and Commonly Used Herbs. Pharmacognosy Research, 2016. [DOI | PubMed]
- Y Pan, BA Abd-Rashid, Z Ismail, R Ismail, JW Mak, PC Pook, HM Er, CE Ong. In vitro modulatory effects of Andrographis paniculata, Centella asiatica and Orthosiphon stamineus on cytochrome P450 2C19 (CYP2C19). Journal of Ethnopharmacology, 2011. [DOI | PubMed]
- A Kar, S Pandit, K Mukherjee, S Bahadur, PK Mukherjee. Safety assessment of selected medicinal food plants used in Ayurveda through CYP450 enzyme inhibition study. J Journal of the Science of Food and Agriculture, 2017. [DOI | PubMed]
- M Nigam, M Atanassova, AP Mishra, R Pezzani, HP Devkota, S Plygun, B Salehi, WN Setzer, J Sharifi-Rad. Bioactive Compounds and Health Benefits ofArtemisiaSpecies. Natural Product Communications, 2019. [DOI]
- AH Gilani, KH Janbaz. Preventive and curative effects of Artemisia absinthium on acetaminophen and CCl4-induced hepatotoxicity. General Pharmacology, 1995. [DOI | PubMed]
- RK Joshi. Volatile composition and antimicrobial activity of the essential oil of Artemisia absinthium growing in Western Ghats region of North West Karnataka, India. Pharmaceutical Biology, 2013. [DOI | PubMed]
- KS Chung, HE Choi, JS Shin, EJ Cho, YW Cho, JH Choi, NI Baek, KT Lee. Chemopreventive effects of standardized ethanol extract from the aerial parts of Artemisia princeps Pampanini cv. Sajabal via NF-kappaB inactivation on colitis-associated colon tumorigenesis in mice. Food and Chemical Toxicology, 2015. [DOI | PubMed]
- J Ahamad, SR Mir, S Amin. A Pharmacognostic Review on Artemisia Absinthium. International Research Journal of Pharmacy, 2019. [DOI]
- GE Batiha, A Olatunde, A El-Mleeh, HF Hetta, S Al-Rejaie, S Alghamdi, M Zahoor, A Magdy Beshbishy, T Murata, A Zaragoza-Bastida, N Rivero-Perez. Bioactive Compounds, Pharmacological Actions, and Pharmacokinetics of Wormwood (Artemisia absinthium). Antibiotics (Basel), 2020. [DOI | PubMed]
- SC Yan, YJ Wang, YJ Li, WY Cai, XG Weng, Q Li, Y Chen, Q Yang, XX Zhu. Dihydroartemisinin Regulates the Th/Treg Balance by Inducing Activated CD4+ T cell Apoptosis via Heme Oxygenase-1 Induction in Mouse Models of Inflammatory Bowel Disease. Molecules, 2019. [DOI | PubMed]
- S Gunther, P Fagone, G Jalce, AG Atanasov, C Guignabert, F Nicoletti. Role of MIF and D-DT in immune-inflammatory, autoimmune, and chronic respiratory diseases: from pathogenic factors to therapeutic targets. Drug Discovery Today, 2019. [DOI | PubMed]
- B Omer, S Krebs, H Omer, TO Noor. Steroid-sparing effect of wormwood (Artemisia absinthium) in Crohn’s disease: a double-blind placebo-controlled study. Phytomedicine, 2007. [DOI | PubMed]
- D Lopes-Lutz, DS Alviano, CS Alviano, PP Kolodziejczyk. Screening of chemical composition, antimicrobial and antioxidant activities of Artemisia essential oils. Phytochemistry, 2008. [DOI | PubMed]
- M Nigam, M Atanassova, AP Mishra, R Pezzani, HP Devkota, S Plygun, B Salehi, WN Setzer, J Sharifi-Rad. Bioactive Compounds and Health Benefits ofArtemisiaSpecies. Natural Product Communications, 2019. [DOI]
- M Govindarajan, G Benelli. Artemisia absinthium-borne compounds as novel larvicides: effectiveness against six mosquito vectors and acute toxicity on non-target aquatic organisms. Parasitology Research, 2016. [DOI | PubMed]
- MR Desrosiers, A Mittelman, PJ Weathers. Dried Leaf Artemisia Annua Improves Bioavailability of Artemisinin via Cytochrome P450 Inhibition and Enhances Artemisinin Efficacy Downstream. Biomolecules, 2020. [DOI | PubMed]
- J Xing, BJ Kirby, D Whittington, Y Wan, DR Goodlett. Evaluation of P450 inhibition and induction by artemisinin antimalarials in human liver microsomes and primary human hepatocytes. Drug Metabolism and Disposition, 2012. [DOI | PubMed]
- T Ericsson, C Masimirembwa, A Abelo, M Ashton. The evaluation of CYP2B6 inhibition by artemisinin antimalarials in recombinant enzymes and human liver microsomes. Drug Metabolism Letters, 2012. [DOI | PubMed]
- SRM Ibrahim, JM Badr, KAE Sayed, DTA Youssef. A New Cytotoxic Sesquiterpene and Three Anti-inflammatory Flavonoids from Egyptian Tanacetum Santolinoides. Natural Product Communications, 2019. [DOI]
- A Szopa, J Pajor, P Klin, A Rzepiela, HO Elansary, FA Al-Mana, MA Mattar, H Ekiert. Artemisia absinthium L-Importance in the History of Medicine, the Latest Advances in Phytochemistry and Therapeutical, 2020. [DOI | PubMed]
- HY Ji, SY Kim, DK Kim, JH Jeong, HS Lee. Effects of eupatilin and jaceosidin on cytochrome p450 enzyme activities in human liver microsomes. Molecules, 2010. [DOI | PubMed]
- P Melillo de Magalhaes, I Dupont, A Hendrickx, A Joly, T Raas, S Dessy, T Sergent, YJ Schneider. Anti-inflammatory effect and modulation of cytochrome P450 activities by Artemisia annua tea infusions in human intestinal Caco-2 cells. Food Chemistry, 2012. [DOI | PubMed]
- AF Attah, AA Fagbemi, O Olubiyi, H Dada-Adegbola, A Oluwadotun, A Elujoba, CP Babalola. Therapeutic Potentials of Antiviral Plants Used in Traditional African Medicine With COVID-19 in Focus: A Nigerian Perspective. Frontiers in Pharmacology, 2021. [DOI | PubMed]
- NS Radulovic, MS Gencic, NM Stojanovic, PJ Randjelovic, ZZ Stojanovic-Radic, NI Stojiljkovic. Toxic essential oils. Part V: Behaviour modulating and toxic properties of thujones and thujone-containing essential oils of Salvia officinalis L., Artemisia absinthium L., Thuja occidentalis L. and Tanacetum vulgare L. Food and Chemical Toxicology, 2017. [DOI | PubMed]
- M Abdel-Tawab, O Werz, M Schubert-Zsilavecz. Boswellia serrata: an overall assessment of in vitro, preclinical, pharmacokinetic and clinical data. Clinical Pharmacokinetics, 2011. [DOI | PubMed]
- HP Ammon. Boswellic Acids and Their Role in Chronic Inflammatory Diseases. Advances in Experimental Medicine and Biology, 2016. [DOI | PubMed]
- T Efferth, F Oesch. Anti-inflammatory and anti-cancer activities of frankincense: Targets, treatments and toxicities. Semin Cancer Biol, 2020. [DOI | PubMed]
- I Gupta, A Parihar, P Malhotra, S Gupta, R Ludtke, H Safayhi, HP Ammon. Effects of gum resin of Boswellia serrata in patients with chronic colitis. Planta Medica, 2001. [DOI | PubMed]
- M Gupta, N Singh, M Gulati, R Gupta, K Sudhakar, B Kapoor. Herbal bioactives in treatment of inflammation: An overview. South African Journal of Botany, 2021. [DOI]
- RM Hartmann, HS Fillmann, MI Martins, L Meurer, NP Marroni. Boswellia serrata has beneficial anti-inflammatory and antioxidant properties in a model of experimental colitis. Phytotherapy Research, 2014. [DOI | PubMed]
- G Holleran, F Scaldaferri, A Gasbarrini, D Curro. Herbal medicinal products for inflammatory bowel disease: A focus on those assessed in double-blind randomised controlled trials. Phytotherapy Research, 2020. [DOI | PubMed]
- D Dey, S Chaskar, N Athavale, D Chitre. Acute and chronic toxicity, cytochrome p450 enzyme inhibition, and HERG channel blockade studies with a polyherbal, ayurvedic formulation for inflammation. BioMed Research International, 2015. [DOI | PubMed]
- A Frank, M Unger. Analysis of frankincense from various Boswellia species with inhibitory activity on human drug metabolising cytochrome P450 enzymes using liquid chromatography mass spectrometry after automated on-line extraction. Journal of Chromatography A, 2006. [DOI | PubMed]
- A Koeberle, A Henkel, M Verhoff, L Tausch, S Konig, D Fischer, N Kather, S Seitz, M Paul, J Jauch, O Werz. Triterpene Acids from Frankincense and Semi-Synthetic Derivatives That Inhibit 5-Lipoxygenase and Cathepsin G. Molecules, 2018. [DOI | PubMed]
- A Paoletti, E Gallo, S Benemei, M Vietri, F Lapi, R Volpi, F Menniti-Ippolito, L Gori, A Mugelli, F Firenzuoli, A Vannacci. Interactions between Natural Health Products and Oral Anticoagulants: Spontaneous Reports in the Italian Surveillance System of Natural Health Products. Evidence-Based Complementary and Alternative Medicine, 2011. [DOI | PubMed]
- N Milic, N Milosevic, S Golocorbin Kon, T Bozic, L Abenavoli, F Borrelli. Warfarin interactions with medicinal herbs. Natural Product Communications, 2014. [PubMed]
- CC Weber, K Reising, WE Muller, M Schubert-Zsilavecz, M Abdel-Tawab. Modulation of Pgp function by boswellic acids. Planta Medica, 2006. [DOI | PubMed]
- MA ElSohly, MM Radwan, W Gul, S Chandra, A Galal. Phytochemistry of Cannabis sativa L. Phytocannabinoids: Unraveling the Complex Chemistry and Pharmacology of Cannabis sativa, 2017
- SA Bonini, M Premoli, S Tambaro, A Kumar, G Maccarinelli, M Memo, A Mastinu. Cannabis sativa: A comprehensive ethnopharmacological review of a medicinal plant with a long history. Journal of Ethnopharmacology, 2018. [DOI | PubMed]
- CM Andre, JF Hausman, G Guerriero. Cannabis sativa: The Plant of the Thousand and One Molecules. Frontiers in Plant Sciences, 2016. [DOI | PubMed]
- P Alves, C Amaral, N Teixeira, G Correia-da-Silva. Cannabis sativa: Much more beyond Delta(9)-tetrahydrocannabinol. Pharmacological Research, 2020. [DOI | PubMed]
- E Tibirica. The multiple functions of the endocannabinoid system: a focus on the regulation of food intake. Diabetology and Metabolic Syndrome, 2010. [DOI | PubMed]
- R Schicho, M Storr. Cannabis finds its way into treatment of Crohn’s disease. Pharmacology, 2014. [DOI | PubMed]
- W Ahmed, S Katz. Therapeutic Use of Cannabis in Inflammatory Bowel Disease. Gastroenterology and Hepatology (N Y), 2016. [PubMed]
- PM Irving, T Iqbal, C Nwokolo, S Subramanian, S Bloom, N Prasad, A Hart, C Murray, JO Lindsay, A Taylor, R Barron, S Wright. A Randomized, Double-blind, Placebo-controlled, Parallel-group, Pilot Study of Cannabidiol-rich Botanical Extract in the Symptomatic Treatment of Ulcerative Colitis. Inflammatory Bowel Diseases, 2018. [DOI | PubMed]
- TS Kafil, TM Nguyen, JK MacDonald, N Chande. Cannabis for the treatment of ulcerative colitis. The Cochrane Database of Systematic Reviews, 2018. [DOI | PubMed]
- ACA Carvalho, GA Souza, SV Marqui, EL Guiguer, AC Araujo, CJ Rubira, RA Goulart, UAP Flato, P Bueno, RL Buchaim, SM Barbalho. Cannabis and Canabidinoids on the Inflammatory Bowel Diseases: Going Beyond Misuse. International Journal of Molecular Sciences, 2020. [DOI | PubMed]
- A Perisetti, AH Rimu, SA Khan, P Bansal, H Goyal. Role of cannabis in inflammatory bowel diseases. Annals of Gastroenterology, 2020. [DOI | PubMed]
- O Zendulka, G Dovrtelova, K Noskova, M Turjap, A Sulcova, L Hanus, J Jurica. Cannabinoids and Cytochrome P450 Interactions. Current Drug Metabolism, 2016. [DOI | PubMed]
- WR Arnold, AT Weigle, A Das. Cross-talk of cannabinoid and endocannabinoid metabolism is mediated via human cardiac CYP2J2. Journal of Inorganic Biochemistry, 2018. [DOI | PubMed]
- BC Foster, H Abramovici, CS Harris. Cannabis and Cannabinoids: Kinetics and Interactions. The American Journal of Medicine, 2019. [DOI | PubMed]
- J Greger, V Bates, L Mechtler, F Gengo. A Review of Cannabis and Interactions With Anticoagulant and Antiplatelet Agents. The Journal of Clinical Pharmacology, 2020. [DOI | PubMed]
- D Amaral Silva, DW Pate, RD Clark, NM Davies, AOS El-Kadi, R Lobenberg. Phytocannabinoid drug-drug interactions and their clinical implications. Pharmacology and Therapeutics, 2020. [DOI | PubMed]
- SM Stout, NM Cimino. Exogenous cannabinoids as substrates, inhibitors, and inducers of human drug metabolizing enzymes: a systematic review. Drug Metabolism Reviews, 2014. [DOI | PubMed]
- CJ Lucas, P Galettis, J Schneider. The pharmacokinetics and the pharmacodynamics of cannabinoids. Br The Journal of Clinical Pharmacology, 2018. [DOI | PubMed]
- MA Alsherbiny, CG Li. Medicinal Cannabis-Potential Drug Interactions. Medicines (Basel), 2018. [DOI | PubMed]
- M Vazquez, N Guevara, C Maldonado, PC Guido, P Schaiquevich. Potential Pharmacokinetic Drug-Drug Interactions between Cannabinoids and Drugs Used for Chronic Pain. BioMed Research International, 2020. [DOI | PubMed]
- PT Doohan, LD Oldfield, JC Arnold, LL Anderson. Cannabinoid Interactions with Cytochrome P450 Drug Metabolism: a Full-Spectrum Characterization. The American Association of Pharmaceutical Scientists Journal, 2021. [DOI | PubMed]
- S Yamaori, M Kushihara, I Yamamoto, K Watanabe. Characterization of major phytocannabinoids, cannabidiol and cannabinol, as isoform-selective and potent inhibitors of human CYP1 enzymes. Biochemical Pharmacology, 2010. [DOI | PubMed]
- C Rong, NE Carmona, YL Lee, RM Ragguett, Z Pan, JD Rosenblat, M Subramaniapillai, M Shekotikhina, F Almatham, A Alageel, R Mansur, RC Ho, RS McIntyre. Drug-drug interactions as a result of co-administering Delta(9)-THC and CBD with other psychotropic agents. Expert Opinion on Drug Safety, 2018. [DOI | PubMed]
- S Yamaori, C Maeda, I Yamamoto, K Watanabe. Differential inhibition of human cytochrome P450 2A6 and 2B6 by major phytocannabinoids. Forensic Toxicology, 2011. [DOI]
- S Yamaori, J Ebisawa, Y Okushima, I Yamamoto, K Watanabe. Potent inhibition of human cytochrome P450 3A isoforms by cannabidiol: role of phenolic hydroxyl groups in the resorcinol moiety. Life Sciences, 2011. [DOI | PubMed]
- R Jiang, S Yamaori, Y Okamoto, I Yamamoto, K Watanabe. Cannabidiol is a potent inhibitor of the catalytic activity of cytochrome P450 2C19. Drug Metabolism and Pharmacokinetics, 2013. [DOI | PubMed]
- S Yamaori, K Koeda, M Kushihara, Y Hada, I Yamamoto, K Watanabe. Comparison in the in vitro inhibitory effects of major phytocannabinoids and polycyclic aromatic hydrocarbons contained in marijuana smoke on cytochrome P450 2C9 activity. Drug Metabolism and Pharmacokinetics, 2012. [DOI | PubMed]
- P Damkier, D Lassen, MMH Christensen, KG Madsen, M Hellfritzsch, A Pottegard. Interaction between warfarin and cannabis. Basic and Clinical Pharmacology and Toxicology, 2019. [DOI | PubMed]
- S Yamaori, Y Okamoto, I Yamamoto, K Watanabe. Cannabidiol, a major phytocannabinoid, as a potent atypical inhibitor for CYP2D6. Drug Metabolism and Disposition, 2011. [DOI | PubMed]
- HP Ammon, MA Wahl. Pharmacology of Curcuma longa. Planta Medica, 1991. [DOI | PubMed]
- BB Aggarwal, C Sundaram, N Malani, H Ichikawa. Curcumin: the Indian solid gold. Advances in Experimental Medicine and Biology, 2007. [DOI | PubMed]
- BB Aggarwal, KB Harikumar. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. The International Journal of Biochemistry and Cell Biology, 2009. [DOI | PubMed]
- AB Kunnumakkara, D Bordoloi, G Padmavathi, J Monisha, NK Roy, S Prasad, BB Aggarwal. Curcumin, the golden nutraceutical: multitargeting for multiple chronic diseases. British Journal of Pharmacology, 2017. [DOI | PubMed]
- B Kocaadam, N Sanlier. Curcumin, an active component of turmeric (Curcuma longa), and its effects on health. Critical Reviews in Food Science and Nutrition, 2017. [DOI | PubMed]
- R Andrew, AA Izzo. Principles of pharmacological research of nutraceuticals. British Journal of Pharmacology, 2017. [DOI | PubMed]
- A Goel, AB Kunnumakkara, BB Aggarwal. Curcumin as “Curecumin”: from kitchen to clinic. Biochemical Pharmacology, 2008. [DOI | PubMed]
- NS Dosoky, WN Setzer. Chemical Composition and Biological Activities of Essential Oils of Curcuma Species Nutrients, 2018. [DOI | PubMed]
- V Soleimani, A Sahebkar, H Hosseinzadeh. Turmeric (Curcuma longa) and its major constituent (curcumin) as nontoxic and safe substances: Review. Phytotherapy Research, 2018. [DOI | PubMed]
- Z Ayati, M Ramezani, MS Amiri, AT Moghadam, H Rahimi, A Abdollahzade, A Sahebkar, SA Emami. Ethnobotany, Phytochemistry and Traditional Uses of Curcuma spp. and Pharmacological Profile of Two Important Species (C. longa and C. zedoaria): A Review. Current Pharmaceutical Design, 2019. [DOI | PubMed]
- J Sharifi-Rad, YE Rayess, AA Rizk, C Sadaka, R Zgheib, W Zam, S Sestito, S Rapposelli, K Neffe-Skocinska, D Zielinska, B Salehi, WN Setzer, NS Dosoky, Y Taheri, M El Beyrouthy, M Martorell, EA Ostrander, HAR Suleria, WC Cho, A Maroyi, N Martins. Turmeric and Its Major Compound Curcumin on Health: Bioactive Effects and Safety Profiles for Food, Pharmaceutical, Biotechnological and Medicinal Applications. Frontiers in Pharmacology, 2020. [DOI | PubMed]
- RS Ahmad, MB Hussain, MT Sultan, MS Arshad, M Waheed, MA Shariati, S Plygun, MH Hashempur. Biochemistry, Safety, Pharmacological Activities, and Clinical Applications of Turmeric: A Mechanistic Review. Evidence-Based Complementary and Alternative Medicine, 2020. [DOI | PubMed]
- ME Abd El-Hack, MT El-Saadony, AA Swelum, M Arif, MM Abo Ghanima, M Shukry, A Noreldin, AE Taha, KA El-Tarabily. Curcumin, the active substance of turmeric: its effects on health and ways to improve its bioavailability. Journal of the Science of Food and Agriculture, 2021. [DOI | PubMed]
- C Quispe, N Cruz-Martins, ML Manca, M Manconi, O Sytar, N Hudz, M Shanaida, M Kumar, Y Taheri, M Martorell, J Sharifi-Rad, G Pintus, WC Cho. Nano-Derived Therapeutic Formulations with Curcumin in Inflammation-Related Diseases. Oxidative Medicine and Cellular Longevity, 2021. [DOI | PubMed]
- PR Holt, S Katz, R Kirshoff. Curcumin therapy in inflammatory bowel disease: a pilot study. Digestive Diseases and Sciences, 2005. [DOI | PubMed]
- H Hanai, T Iida, K Takeuchi, F Watanabe, Y Maruyama, A Andoh, T Tsujikawa, Y Fujiyama, K Mitsuyama, M Sata, M Yamada, Y Iwaoka, K Kanke, H Hiraishi, K Hirayama, H Arai, S Yoshii, M Uchijima, T Nagata, Y Koide. Curcumin maintenance therapy for ulcerative colitis: randomized, multicenter, double-blind, placebo-controlled trial. Clinical and Gastroenterology & Hepatology, 2006. [DOI | PubMed]
- V Singla, V Pratap Mouli, SK Garg, T Rai, BN Choudhury, P Verma, R Deb, V Tiwari, S Rohatgi, R Dhingra, S Kedia, PK Sharma, G Makharia, V Ahuja. Induction with NCB-02 (curcumin) enema for mild-to-moderate distal ulcerative colitis – a randomized, placebo-controlled, pilot study. Journal of Crohn’s and Colitis, 2014. [DOI | PubMed]
- A Lang, N Salomon, JC Wu, U Kopylov, A Lahat, O Har-Noy, JY Ching, PK Cheong, B Avidan, D Gamus, I Kaimakliotis, R Eliakim, SC Ng, S Ben-Horin. Curcumin in Combination With Mesalamine Induces Remission in Patients With Mild-to-Moderate Ulcerative Colitis in a Randomized Controlled Trial. Clinical Gastroenterology and Hepatology, 2015. [DOI | PubMed]
- S Kedia, V Bhatia, S Thareja, S Garg, VP Mouli, S Bopanna, V Tiwari, G Makharia, V Ahuja. Low dose oral curcumin is not effective in induction of remission in mild to moderate ulcerative colitis: Results from a randomized double blind placebo controlled trial. World J Gastrointest Pharmacology and Therapeutics, 2017. [DOI | PubMed]
- RJ Price, MP Scott, AM Giddings, DG Walters, RH Stierum, C Meredith, BG Lake. Effect of butylated hydroxytoluene, curcumin, propyl gallate and thiabendazole on cytochrome P450 forms in cultured human hepatocytes. Xenobiotica, 2008. [DOI | PubMed]
- LP Volak, S Ghirmai, JR Cashman, MH Court. Curcuminoids inhibit multiple human cytochromes P450, UDP-glucuronosyltransferase, and sulfotransferase enzymes, whereas piperine is a relatively selective CYP3A4 inhibitor. Drug Metabolism and Disposition, 2008. [DOI | PubMed]
- AC Liu, LX Zhao, J Xing, T Liu, FY Du, HX Lou. Pre-treatment with curcumin enhances plasma concentrations of losartan and its metabolite EXP3174 in rats. Biological and Pharmaceutical Bulletin, 2012. [DOI | PubMed]
- XF Koe, TS Tengku Muhammad, AS Chong, HA Wahab, ML Tan. Cytochrome P450 induction properties of food and herbal-derived compounds using a novel multiplex RT-qPCR in vitro assay, a drug-food interaction prediction tool. Food Science & Nutrition, 2014. [DOI | PubMed]
- VB Liju, K Jeena, R Kuttan. Chemopreventive activity of turmeric essential oil and possible mechanisms of action. Asian Pacific Journal of Cancer Prevention, 2014. [DOI | PubMed]
- R Bahramsoltani, R Rahimi, MH Farzaei. Pharmacokinetic interactions of curcuminoids with conventional drugs: A review. Journal of Ethnopharmacology, 2017. [DOI | PubMed]
- DX Sun, ZZ Fang, YY Zhang, YF Cao, L Yang, J Yin. Inhibitory effects of curcumenol on human liver cytochrome P450 enzymes. Phytotherapy Research, 2010. [DOI | PubMed]
- H Jinno, T Tanaka-Kagawa, A Ohno, Y Makino, E Matsushima, N Hanioka, M Ando. Functional characterization of cytochrome P450 2B6 allelic variants. Drug Metabolism and Disposition, 2003. [DOI | PubMed]
- R Appiah-Opong, JN Commandeur, B van Vugt-Lussenburg, NP Vermeulen. Inhibition of human recombinant cytochrome P450s by curcumin and curcumin decomposition products. Toxicology, 2007. [DOI | PubMed]
- SB Kim, SS Cho, HJ Cho, IS Yoon. Modulation of Hepatic Cytochrome P450 Enzymes by Curcumin and its Pharmacokinetic Consequences in Sprague-dawley Rats. Pharmacognosy Magazine, 2015. [DOI | PubMed]
- P Rodriguez Castano, S Parween, AV Pandey. Bioactivity of Curcumin on the Cytochrome P450 Enzymes of the Steroidogenic Pathway. International Journal of Molecular Sciences, 2019. [DOI | PubMed]
- Y Bamba, YS Yun, A Kunugi, H Inoue. Compounds isolated from Curcuma aromatica Salisb. inhibit human P450 enzymes. Journal of Natural Medicines, 2011. [DOI | PubMed]
- Q Liu, DS Dang, YF Chen, M Yan, GB Shi, QC Zhao. The influence of omeprazole on platelet inhibition of clopidogrel in various CYP2C19 mutant alleles. Genetic Testing and Molecular Biomarkers, 2012. [DOI | PubMed]
- MV Holmes, P Perel, T Shah, AD Hingorani, JP Casas. CYP2C19 genotype, clopidogrel metabolism, platelet function, and cardiovascular events: a systematic review and meta-analysis. The Journal of the American Medical Association, 2011. [DOI | PubMed]
- YB Jarrar, SJ Lee. Molecular functionality of CYP2C9 polymorphisms and their influence on drug therapy. Drug Metabolism and Drug Interactions, 2014. [DOI | PubMed]
- XL Hou, K Takahashi, N Kinoshita, F Qiu, K Tanaka, K Komatsu, K Takahashi, J Azuma. Possible inhibitory mechanism of Curcuma drugs on CYP3A4 in 1alpha,25 dihydroxyvitamin D3 treated Caco-2 cells. International Journal of Pharmaceutics, 2007. [DOI | PubMed]
- W Zhang, LY Lim. Effects of spice constituents on P-glycoprotein-mediated transport and CYP3A4-mediated metabolism in vitro. Drug Metabolism and Disposition, 2008. [DOI | PubMed]
- YW Hsieh, CY Huang, SY Yang, YH Peng, CP Yu, PD Chao, YC Hou. Oral intake of curcumin markedly activated CYP 3A4: in vivo and ex-vivo studies. Scientific Reports, 2014. [DOI | PubMed]
- M Mohajeri, B Behnam, AFG Cicero, A Sahebkar. Protective effects of curcumin against aflatoxicosis: A comprehensive review. Journal of Cellular Physiology, 2018. [DOI | PubMed]
- JL Raucy. Regulation of CYP3A4 expression in human hepatocytes by pharmaceuticals and natural products. Drug Metabolism and Disposition, 2003. [DOI | PubMed]
- A Graber-Maier, KB Buter, J Aeschlimann, C Bittel, M Kreuter, J Drewe, H Gutmann. Effects of Curcuma extracts and curcuminoids on expression of P-glycoprotein and cytochrome P450 3A4 in the intestinal cell culture model LS180. Planta Medica, 2010. [DOI | PubMed]
- TC Seah, YL Tay, HK Tan, TS Muhammad, HA Wahab, ML Tan. Determination of CYP3A4 Inducing Properties of Compounds Using a Laboratory-Developed Cell-Based Assay. International Journal of Toxicology, 2015. [DOI | PubMed]
- SJ Lee, OR Hedstrom, K Fischer, JL Wang-Buhler, A Sen, I Cok, DR Buhler. Immunohistochemical localization and differential expression of cytochrome P450 3A27 in the gastrointestinal tract of rainbow trout. Toxicology and Applied Pharmacology, 2001. [DOI | PubMed]
- HH Agus, P Tekin, M Bayav, A Semiz, A Sen. Drug Interaction Potential of the Seed Extract of Urtica urens L. (Dwarf Nettle). Phytotherapy Research, 2009. [DOI | PubMed]
- D Wang, W Sadee. CYP3A4 intronic SNP rs35599367 (CYP3A4*22) alters RNA splicing. Pharmacogenetics and Genomics, 2016. [DOI | PubMed]
- M Kato, K Chiba, A Hisaka, M Ishigami, M Kayama, N Mizuno, Y Nagata, S Takakuwa, Y Tsukamoto, K Ueda, H Kusuhara, K Ito, Y Sugiyama. The intestinal first-pass metabolism of substrates of CYP3A4 and P-glycoprotein-quantitative analysis based on information from the literature. Drug Metabolism and Pharmacokinetics, 2003. [DOI | PubMed]
- J Chen, Z Huang, Z Chen, M Yuan, Y Liu, Y Zhu. The effect of carbon on surface quality of solid-state-sintered silicon carbide as optical materials. Materials Characterization, 2014. [DOI]
- G Celik, H Akca, A Sen. Investigation of aromotase inhibition by several dietary vegetables in human non-small cell lung cancer cell lines. Turkish Journal of Biochemistry, 2013. [DOI | PubMed]
- J Srivilai, K Rabgay, N Khorana, N Waranuch, N Nuengchamnong, K Ingkaninan. A new label-free screen for steroid 5alpha-reductase inhibitors using LC-MS. Steroids, 2016. [DOI | PubMed]
- P Pimkaew, J Kublbeck, A Petsalo, J Jukka, A Suksamrarn, R Juvonen, S Auriola, P Piyachaturawat, P Honkakoski. Interactions of sesquiterpenes zederone and germacrone with the human cytochrome P450 system. Toxicology in Vitro, 2013. [DOI | PubMed]
- Y Araki, A Andoh, Y Fujiyama, O Kanauchi, K Takenaka, A Higuchi, T Bamba. Germinated barley foodstuff exhibits different adsorption properties for hydrophilic versus hydrophobic bile acids. Digestion, 2001. [DOI | PubMed]
- T Bamba, O Kanauchi, A Andoh, Y Fujiyama. A new prebiotic from germinated barley for nutraceutical treatment of ulcerative colitis. Journal of Gastroenterology and Hepatology, 2002. [DOI | PubMed]
- O Kanauchi, T Suga, M Tochihara, T Hibi, M Naganuma, T Homma, H Asakura, H Nakano, K Takahama, Y Fujiyama, A Andoh, T Shimoyama, N Hida, K Haruma, H Koga, K Mitsuyama, M Sata, M Fukuda, A Kojima, T Bamba. Treatment of ulcerative colitis by feeding with germinated barley foodstuff: first report of a multicenter open control trial. Journal of Gastroenterology, 2002. [DOI | PubMed]
- H Hanai, O Kanauchi, K Mitsuyama, A Andoh, K Takeuchi, I Takayuki, Y Araki, Y Fujiyama, A Toyonaga, M Sata, A Kojima, M Fukuda, T Bamba. Germinated barley foodstuff prolongs remission in patients with ulcerative colitis. International Journal of Molecular Medicine, 2004. [PubMed]
- K Mitsuyama, T Saiki, O Kanauchi, T Iwanaga, N Tomiyasu, T Nishiyama, H Tateishi, A Shirachi, M Ide, A Suzuki, K Noguchi, H Ikeda, A Toyonaga, M Sata. Treatment of ulcerative colitis with germinated barley foodstuff feeding: a pilot study. Alimentary Pharmacology and Therapeutics, 1998. [DOI | PubMed]
- O Kanauchi, T Oshima, A Andoh, M Shioya, K Mitsuyama. Germinated barley foodstuff ameliorates inflammation in mice with colitis through modulation of mucosal immune system. Scandian Journal of Gastroenterology, 2008. [DOI | PubMed]
- A Triantafyllidi, T Xanthos, A Papalois, JK Triantafillidis. Herbal and plant therapy in patients with inflammatory bowel disease. Annals of Gastroenterology, 2015. [PubMed]
- E Ben-Arye, E Goldin, D Wengrower, A Stamper, R Kohn, E Berry. Wheat grass juice in the treatment of active distal ulcerative colitis: a randomized double-blind placebo-controlled trial. Scandian Journal of Gastroenterology, 2002. [DOI | PubMed]
- H Suzuki, T Kaneko, Y Mizokami, T Narasaka, S Endo, H Matsui, A Yanaka, A Hirayama, I Hyodo. Therapeutic efficacy of the Qing Dai in patients with intractable ulcerative colitis. World Journal of Gastroenterology, 2013. [DOI | PubMed]
- F Zhang, Y Li, F Xu, Y Chu, W Zhao. Comparison of Xilei-san, a Chinese herbal medicine, and dexamethasone in mild/moderate ulcerative proctitis: a double-blind randomized clinical trial. Journal of Alternative and Complementary Medicine, 2013. [DOI | PubMed]
- S Sugimoto, M Naganuma, H Kiyohara, M Arai, K Ono, K Mori, K Saigusa, K Nanki, K Takeshita, T Takeshita, M Mutaguchi, S Mizuno, R Bessho, Y Nakazato, T Hisamatsu, N Inoue, H Ogata, Y Iwao, T Kanai. Clinical Efficacy and Safety of Oral Qing-Dai in Patients with Ulcerative Colitis: A Single-Center Open-Label Prospective Study. Digestion, 2016. [DOI | PubMed]
- M Naganuma, S Sugimoto, K Mitsuyama, T Kobayashi, N Yoshimura, H Ohi, S Tanaka, A Andoh, N Ohmiya, K Saigusa, T Yamamoto, Y Morohoshi, H Ichikawa, K Matsuoka, T Hisamatsu, K Watanabe, S Mizuno, W Suda, M Hattori, S Fukuda, A Hirayama, T Abe, M Watanabe, T Hibi, Y Suzuki, T Kanai, IS Group. Efficacy of Indigo Naturalis in a Multicenter Randomized Controlled Trial of Patients With Ulcerative Colitis. Gastroenterology, 2018. [DOI | PubMed]
- M Naganuma. Treatment with indigo naturalis for inflammatory bowel disease and other immune diseases. Immunological Medicine, 2019. [DOI | PubMed]
- JP Saiki, JOL Andreasson, KV Grimes, LR Frumkin, E Sanjines, MG Davidson, KT Park, B Limketkai. Treatment-refractory ulcerative colitis responsive to indigo naturalis. BMJ Open Gastroenterology, 2021. [DOI | PubMed]
- S Gu, Y Xue, Y Gao, S Shen, Y Zhang, K Chen, S Xue, J Pan, Y Tang, H Zhu, H Wu, D Dou. Mechanisms of indigo naturalis on treating ulcerative colitis explored by GEO gene chips combined with network pharmacology and molecular docking. Scientific Reports, 2020. [DOI | PubMed]
- S Kondo, T Araki, Y Okita, A Yamamoto, Y Hamada, M Katsurahara, N Horiki, M Nakamura, T Shimoyama, T Yamamoto, Y Takei, M Kusunoki. Colitis with wall thickening and edematous changes during oral administration of the powdered form of Qing-dai in patients with ulcerative colitis: a report of two cases. Clinical Journal of Gastroenterology, 2018. [DOI | PubMed]
- J Mimura, M Ema, K Sogawa, Y Fujii-Kuriyama. Identification of a novel mechanism of regulation of Ah (dioxin) receptor function. Genes and Development, 1999. [DOI | PubMed]
- A Sen, E Arinc. Further immunochemical and biocatalytic characterization of CYP1A1 from feral leaping mullet liver (Liza saliens) microsomes. Comparative Biochemistry and Physiology Part C: Toxicology and Pharmacology, 2000. [PubMed]
- BC Spink, MM Hussain, BH Katz, L Eisele, DC Spink. Transient induction of cytochromes P450 1A1 and 1B1 in MCF-7 human breast cancer cells by indirubin. Biochemical Pharmacology, 2003. [DOI | PubMed]
- J Adachi, Y Mori, S Matsui, T Matsuda. Comparison of gene expression patterns between 2,3,7,8-tetrachlorodibenzo-p-dioxin and a natural arylhydrocarbon receptor ligand, indirubin. Toxicological Sciences, 2004. [DOI | PubMed]
- M Sinz, S Kim, S Ferguson, E LeCluyse. Evaluating and Predicting Human Cytochrome P450 Enzyme Induction. Pharmaceutical Sciences Encyclopedia, 2010
- T Kumagai, Y Aratsu, R Sugawara, T Sasaki, S Miyairi, K Nagata. Indirubin, a component of Ban-Lan-Gen, activates CYP3A4 gene transcription through the human pregnane X receptor. Drug Metabolism and Pharmacokinetics, 2016. [DOI | PubMed]
- H-H Xu, Z-C Ma, Q-L Shi, S-H Yang, L Jiang, X-M Chen, Y Gao. Synergistic effect and different toxicities of adjuvant components of Realgar–Indigo Naturalis formula. Chinese Herbal Medicines, 2018. [DOI]
- SM Greenfield, AT Green, JP Teare, AP Jenkins, NA Punchard, CC Ainley, RP Thompson. A randomized controlled study of evening primrose oil and fish oil in ulcerative colitis. Alimentary Pharmacology and Therapeutics, 1993. [DOI | PubMed]
- B Brinkhaus, C Hentschel, C Von Keudell, G Schindler, M Lindner, H Stutzer, R Kohnen, SN Willich, W Lehmacher, EG Hahn. Herbal medicine with curcuma and fumitory in the treatment of irritable bowel syndrome: a randomized, placebo-controlled, double-blind clinical trial. Scandian Journal of Gastroenterology, 2005. [DOI | PubMed]
- F Emendorfer, F Emendorfer, F Bellato, VF Noldin, V Cechinel-Filho, RA Yunes, F Delle Monache, AM Cardozo. Antispasmodic activity of fractions and cynaropicrin from Cynara scolymus on guinea-pig ileum. Biological and Pharmaceutical Bulletin, 2005. [DOI | PubMed]
- R Huber, AV Ditfurth, F Amann, C Guthlin, M Rostock, R Trittler, K Kummerer, I Merfort. Tormentil for active ulcerative colitis: an open-label, dose-escalating study. Journal of Clinical Gastroenterology, 2007. [DOI | PubMed]
- JM Kim, HW Kang, MY Cha, D Yoo, N Kim, IK Kim, J Ku, S Kim, SH Ma, HC Jung, IS Song, JS Kim. Novel guggulsterone derivative GG-52 inhibits NF-kappaB signaling in intestinal epithelial cells and attenuates acute murine colitis. Laboratory Investigation, 2010. [DOI | PubMed]
- H Wan, Y Chen. Effects of antidepressive treatment of Saint John’s wort extract related to autonomic nervous function in women with irritable bowel syndrome. Int The International Journal of Psychiatry in Medicine, 2010. [DOI | PubMed]
- T Kudo, S Okamura, Y Zhang, T Masuo, M Mori. Topical application of glycyrrhizin preparation ameliorates experimentally induced colitis in rats. World Journal of Gastroenterology, 2011. [DOI | PubMed]
- Y Gong, Q Zha, L Li, Y Liu, B Yang, L Liu, A Lu, Y Lin, M Jiang. Efficacy and safety of Fufangkushen colon-coated capsule in the treatment of ulcerative colitis compared with mesalazine: a double-blinded and randomized study. Journal of Ethnopharmacology, 2012. [DOI | PubMed]
- K Fukunaga, Y Ohda, N Hida, M Iimuro, Y Yokoyama, K Kamikozuru, K Nagase, S Nakamura, H Miwa, T Matsumoto. Placebo controlled evaluation of Xilei San, a herbal preparation in patients with intractable ulcerative proctitis. Journal of Gastroenterology and Hepatology, 2012. [DOI | PubMed]
- R Khanna, JK MacDonald, BG Levesque. Peppermint oil for the treatment of irritable bowel syndrome: a systematic review and meta-analysis. Journal of Clinical Gastroenterology, 2014. [DOI | PubMed]
- S Karakurt, A Semiz, G Celik, AM Gencler-Ozkan, A Sen, O Adali. Contribution of ellagic acid on the antioxidant potential of medicinal plant Epilobium hirsutum. Nutrition and Cancer, 2016. [DOI | PubMed]
- O Ozgun-Acar, G Celik-Turgut, I Gazioglu, U Kolak, S Ozbal, BU Ergur, S Arslan, A Sen, G Topcu. Capparis ovata treatment suppresses inflammatory cytokine expression and ameliorates experimental allergic encephalomyelitis model of multiple sclerosis in C57BL/6 mice. Journal of Neuroimmunology, 2016. [DOI | PubMed]
- GM Sebepos-Rogers, DS Rampton. Herbs and Inflammatory Bowel Disease. Gastroenterology Clinics of North America, 2017. [DOI | PubMed]
- I Gazioglu, S Semen, OO Acar, U Kolak, A Sen, G Topcu. Triterpenoids and steroids isolated from Anatolian Capparis ovata and their activity on the expression of inflammatory cytokines. Pharmaceutical Biology, 2020. [DOI | PubMed]
- A Semiz, O Ozgun Acar, H Cetin, G Semiz, A Sen. Suppression of Inflammatory Cytokines Expression with Bitter Melon (Momordica Charantia) in TNBS-instigated Ulcerative Colitis. Journal of Translational Internal Medicine, 2020. [DOI | PubMed]
- I Suntar, CK Cevik, AO Ceribasi, A Gokbulut. Healing effects of Cornus mas L. in experimentally induced ulcerative colitis in rats: From ethnobotany to pharmacology. Journal of Ethnopharmacology, 2020. [DOI | PubMed]
- H Yu, TN Li, Q Ran, QW Huang, J Wang. Strobilanthes cusia (Nees) Kuntze, a multifunctional traditional Chinese medicinal plant, and its herbal medicines: A comprehensive review. Journal of Ethnopharmacology, 2021. [DOI | PubMed]
- X Zhu, Y Yang, W Gao, B Jiang, L Shi. Capparis spinosa Alleviates DSS-Induced Ulcerative Colitis via Regulation of the Gut Microbiota and Oxidative Stress. Evidence-Based Complementary and Alternative Medicine, 2021. [DOI | PubMed]
