Antibacterial phenolic compounds from the flowering plants of Asia and the Pacific: coming to the light
Abstract
Context:
The emergence of pan-resistant bacteria requires the development of new antibiotics and antibiotic potentiators.
Objective:
This review identifies antibacterial phenolic compounds that have been identified in Asian and Pacific Angiosperms from 1945 to 2023 and analyzes their strengths and spectra of activity, distributions, molecular masses, solubilities, modes of action, structures-activities, as well as their synergistic effects with antibiotics, toxicities, and clinical potential.
Methods:
All data in this review was compiled from Google Scholar, PubMed, Science Direct, Web of Science, and library search; other sources were excluded. We used the following combination of keywords: ‘Phenolic compound’, ‘Plants’, and ‘Antibacterial’. This produced 736 results. Each result was examined and articles that did not contain information relevant to the topic or coming from non-peer-reviewed journals were excluded. Each of the remaining 467 selected articles was read critically for the information that it contained.
Results:
Out of ∼350 antibacterial phenolic compounds identified, 44 were very strongly active, mainly targeting the cytoplasmic membrane of Gram-positive bacteria, and with a molecular mass between 200 and 400 g/mol. 2-Methoxy-7-methyljuglone, [6]-gingerol, anacardic acid, baicalin, vitexin, and malabaricone A and B have the potential to be developed as antibacterial leads.
Conclusions:
Angiosperms from Asia and the Pacific provide a rich source of natural products with the potential to be developed as leads for treating bacterial infections.
Article type: Review Article
Keywords: Angiosperms, antibiotics, Asia-Pacific, inflammation, superbugs
Affiliations: Department of Chemistry, Faculty of Science, Universiti Malaya, Kuala Lumpur, Malaysia; School of Allied Health Sciences, Walailak University, Nakhon Si Thammarat, Thailand; Department of Biotechnology, University of Development Alternative, Dhaka, Bangladesh; Faculty of Education and Teacher Training, Davao Oriental State University, Mati, Philippines; Faculty of Agriculture and Life Science, Davao Oriental State University, Mati, Philippines; Department of Microbiology, Sikkim Manipal University, Gangtok, India; Institute for Tropical Biology and Conservation, Universiti Malaysia Sabah, Kota Kinabalu, Malaysia; Natural Product Division, Forest Research Institute of Malaysia, Kepong, Malaysia; Department of Pharmaceutical Chemistry, College of Pharmacy, University of Ha’il, Ha’il, Saudi Arabia; School of Pharmacy, University of Nottingham Malaysia, Semenyih, Malaysia; School of Pharmacy, Hunan University of Chinese Medicine, Changsha, China
License: © 2024 The Author(s). Published by Informa UK Limited, trading as Taylor & Francis Group. CC BY 4.0 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. The terms on which this article has been published allow the posting of the Accepted Manuscript in a repository by the author(s) or with their consent.
Article links: DOI: 10.1080/13880209.2024.2407530 | PubMed: 39392281 | PMC: PMC11486068
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (7.7 MB)
Introduction
The resistance of bacteria to antibiotics has increased to the point that treating nosocomial infections in intensive care units has become difficult, and in some cases even impossible. The list of bacterial strains resistant to antibiotics continues to grow. The intrinsic mechanisms of resistance in Gram-negative bacteria to antibiotics include, at least in part, an outer lipopolysaccharides coat carrying a net negative charge which halts (partially) the entry of negatively charged molecules (Denyer and Maillard ref. 2002; van den Berg ref. 2010) as well as porins preventing the penetration of lipophilic molecules (Bauer et al. ref. 1988; van den Berg ref. 2010). Bacteria keep on exchanging genes (among other things via plasmid transfer) coding for efflux pumps, such as NorA (Sun et al. ref. 2020), TetK in Staphylococcus aureus (Macêdo et al. ref. 2022), and AcrAB in Escherichia coli (Kuete et al. ref. 2010). Other resistance mechanisms acquired through gene exchange include enzymes that inactivate antibiotics (β-lactamases) (Eumkeb et al. ref. 2010; Siriwong et al. ref. 2015) and structurally altered bacterial targets (Oyedemi et al. ref. 2016).
The mode of action of antibiotics, which target a specific bacterial macromolecule or enzyme, sooner or later leads to the inevitable development of resistance. Some superbugs have accumulated genes of resistance to almost all known antibiotics (Willyard ref. 2017). Examples are Mycobacterium tuberculosis (clinical isolate CIBIN 99) (Uc-Cachón et al. ref. 2014), methicillin-resistant S. aureus (MRSA) SCCmec III (Asghar ref. 2014) and USA300 strains (Carrel et al. ref. 2015), Stenotrophomonas maltophilia (Gordon and Wareham ref. 2010), and vancomycin-resistant enterococcus (VRE) (Tan, Hua, et al. ref. 2020). Acinetobacter baumannii, which began as a hospital commensal bacterium, has transformed over the last decades into a bacterium resistant to almost all antibiotics (Osterburg et al. ref. 2009). In 2016, the World Health Organization (WHO) listed carbapenem-resistant A. baumannii as first on the list of bacteria posing a threat to human health (Willyard ref. 2017). In 2019, Nichols described the case of a 48-year-old man succumbing to a pan-resistant A. baumannii following a lung transplant (Nichols ref. 2019). The number of bacterial strains on the verge of becoming resistant to all antibiotics continues to increase inexorably, and among them is E. coli O157:H7 posing the risk of incurable food poisoning (Haile et al. ref. 2022).
Identifying antibacterial molecules with chemical structures completely different from those of antibiotics currently in use and capable of evading or inhibiting bacterial resistance is an urgent necessity (Chusri et al. ref. 2009). There are numerous sources of antibacterial compounds in the living world, particularly in flowering plants. Flowering plants also called Angiosperms are organized into 11 major taxa or clades distributed into three groups: (i) basal Angiosperms (protomagnoliids, magnoliids, monocots, and eudicots), (ii) core Angiosperms (core eudicots, rosids, fabids, and malvids), and (iii) upper Angiosperms (asterids, lamiids, and campanulids) (Byng et al. ref. 2009). Within each clade, plants are grouped into different orders, families, genera, and species producing secondary metabolites, one of those roles is often to fight against bacterial infections. These antibacterial natural products are either present in plants before bacterial infection (phytoanticipins) or synthesized during bacterial infection (phytoalexins) (Van Etten et al. ref. 1994). Compared to antibiotics, they do not have a single bacterial target (Yuan et al. ref. 2021). Their antibacterial activity is in vitro qualitatively evaluated using paper discs or agar wells and quantified by calculating the minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC). When the MBC/MIC ratio is ≤4, the compounds are bactericidal whereas a MBC/MIC ≥ 8 indicates a bacteriostatic effect (Huang et al. ref. 2021). Phytoanticipins and phytoalexins can act synergistically with antibiotics, in which case the fractional inhibitory concentration index (FICI) is <0.5 (Miklasińska-Majdanik et al. ref. 2018). These antibacterial agents belong to three major phytochemical groups: alkaloids, terpenes, and phenolic compounds.
Phenolic compounds are structurally defined by a benzene ring substituted by at least one hydroxyl group. They are organized into two categories: non-flavonoids and flavonoids, and they occupy a major role in plant defense against bacteria (Weinstein and Albersheim ref. 1983). Several observations made since the 1940s indicate that phenolic compounds can escape acquired bacterial resistance. As early as 1945, Fogg and Lodge observed that Enterobacter aerogenes could not develop resistance to phenolic compounds (Fogg and Lodge ref. 1945). More recently, Chen et al. (ref. 2018) reported the almost impossibility of S. aureus to develop resistance against a phenolic compound identified from an Asian orchid used in traditional Chinese medicine. At the same time, there is growing evidence that phenolic compounds can weaken the resistance of bacteria to antibiotics and enhance the activity of antibiotics.
The hypothesis of using phenolic compounds as a source of antibacterial molecules to treat bacterial infections has been recently raised (Ecevit et al. ref. 2022; Sun and Shahrajabian ref. 2023). In this context, this comprehensive and scholarly evidence-based review aims to cover, organize, and correlate the data accumulated from 1945 to 2023 regarding the antibacterial phenolic compounds identified from flowering plants from Asia and the Pacific. This review covers the distribution, strength, influence of molecular mass and solubility, structure-activity, mechanisms of action, synergistic activity with antibiotics, toxicity, and clinical potential. This review provides a taxonomical, phytochemical, biomolecular, and physicochemical rationale to facilitate the discovery of leads for treating bacterial infections. All data in this review was compiled from Google Scholar, PubMed, Science Direct, Web of Science, and library search, other sources were excluded. We used the following combination of keywords: ‘Phenolic compound’, ‘Plants’, and ‘Antibacterial’. Each result was examined and articles that did not contain information relevant to the topic or coming from non-peer-reviewed journals were excluded. The remaining selected articles were read critically for the information that they contained.
Non-flavonoids
Hydroxycinnamic acid derivatives
They are derived from l-phenyl alanine and are found primarily in the monocots and upper Angiosperms (Figure 1). They have weak but broad-spectrum antibacterial activity. Examples are cinnamic acid (1) from Cinnamomum zeylanicum Bl. (Lauraceae, magnoliids) and p-coumaric acid (2) (Guzman ref. 2014). Caffeic acid (3) from Plantago major L. (Plantaginaceae, lamiids) was active against Pseudomonas aeruginosa (MIC: 31.3 μg/mL) (Perumal et al. ref. 2015; Kępa et al. ref. 2018).
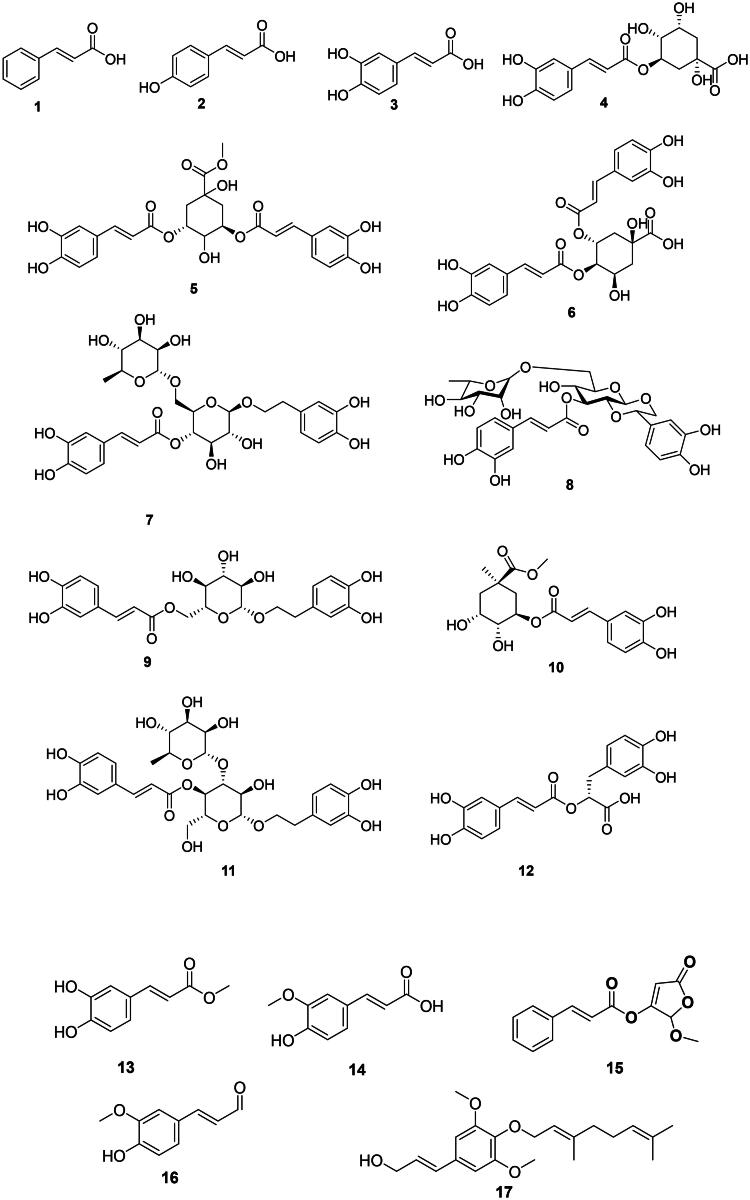
In upper Angiosperms, caffeic acid forms esters with quinic acid, such as chlorogenic acid (4) (Klebsiella pneumoniae) (Cai et al. ref. 2019), 3,5-di-O-caffeoylquinic acid (5), and 4,5-di-O-caffeoylquinic (6) from Lonicera japonica Thunb. (Caprifoliaceae, campanulids) which is a plant used in traditional Chinese medicine (Xiong et al. ref. 2013). Forsythiaside (7) from Forsythia suspensa (Thunb.) Vahl (Oleaceae, lamiids) inhibited the growth of E. coli, P. aeruginosa, and S. aureus with MIC values of 38.3, 38.3, and 76.6 µg/mL, respectively (Qu et al. ref. 2008). From this plant, lianqiaoxinoside B (8) was active against Bacillus dysenteriae (MIC: 36.7 µg/mL) (Kuang et al. ref. 2011). Other examples include calceolarioside B (9) from Sargentodoxa cuneata (Oliv.) Rehder and E.H. Wilson (Lardizabalaceae, eudicots) with S. aureus (MIC: 64 µg/mL), methyl 3-O-caffeoylquinate (10) with of S. aureus (MIC: 32 µg/mL) (Zeng et al. ref. 2015), and verbascoside (11) from Stachytarpheta indica (L.) Vahl (Verbenaceae, lamiids) with Enterococcus faecalis, Shigella sonnei (MIC: 31.2 µg/mL) (Nguyen et al. ref. 2018), and Staphylococcus sp. (MIC: 9.7 µg/mL) (Agampodi et al. ref. 2022).
The coupling of two caffeic acid units forms rosmarinic acid (12) in Rosmarinus officinalis L. (Lamiaceae, lamiids). Rosmarinic acid (12) has weak but broad-spectrum bactericidal activity (Abedini et al. ref. 2013). The esterification of caffeic acid forms methyl caffeate (13) in Solanum torvum Sw. (Solanaceae, lamiids) active against rifampicin-resistant M. tuberculosis (MIC: 8 μg/mL) (Balachandran et al. ref. 2012). Methoxylation of caffeic acid in position 3 gives ferulic acid (14) (Guzman ref. 2014) weakly bactericidal for S. aureus and E. coli (Cai et al. ref. 2019).
Other examples are 2-methoxy-2-butenolide-3-cinnamate (15) from Polygonum glabrum Willd. (Polygonaceae, malvids) (M. tuberculosis, MIC: 1.4 µg/mL) (Said et al. ref. 2015), coniferylaldehyde (16) from Ficus benghalensis L. (Moraceae, fabids) bactericidal for Streptococcus mutans (MIC/MBC: 62.5/62.5 µg/mL) (Meerungrueang and Panichayupakaranant ref. 2014), and nelumol A (17) from Toddalia asiatica (L.) Lam. (Rutaceae, malvids) (M. tuberculosis, MIC: 50 µg/mL) (Phatchana and Yenjai ref. 2014).
Phenylpropanoids
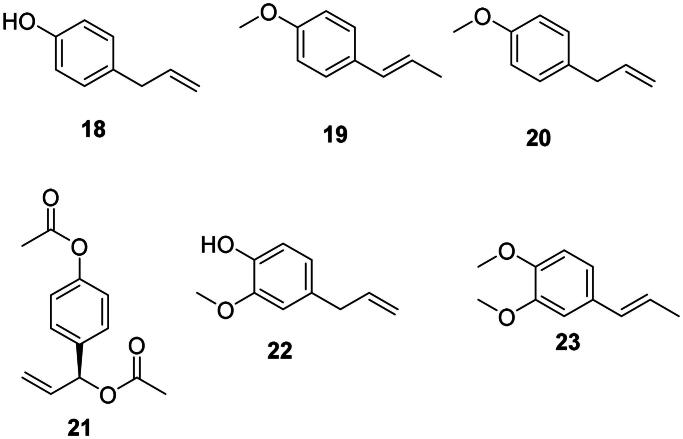
p-Coumaric acid (2) is the precursor of antibacterial phenylpropanoids (Yu and Jez ref. 2008) such as chavicol (18), anethole (19), and estragole (methyl chavicol) (20) (Atkinson ref. 2016) (Figure 2). Anethole (19) was weakly bactericidal against A. baumannii (Newberne et al. ref. 1999) and Bacillus cereus (MIC/MBC: 50/100 µg/mL) (Phanthong et al. ref. 2013). 1′-Acetoxychavicol acetate (21) from Alpinia galanga (L.) Willd. (Zingiberaceae, monocots) inhibited the growth of M. tuberculosis with a MIC value as low as 0.7 µg/mL (Warit et al. ref. 2017). Ferulic acid (14) is the precursor of eugenol (22) in Eugenia aromatica (L.) Baill. (Myrtaceae, malvids) active against B. cereus (MIC: 15.6 µg/mL), E. coli (MIC: 31.2 µg/mL) (Mohammed and Al-Bayati ref. 2009), and Vibrio parahaemolyticus (Ashrafudoulla et al. ref. 2020). Methylisoeugenol (23) from Daucus carota L (Apiaceae, campanulids) was effective against Campylobacter jejuni (Rossi et al. ref. 2007).
Coumarins
These chromene-2-ones originate from the ortho-hydroxylation and cyclization of cinnamic acid (1) (Shimizu ref. 2014) (Figure 3).
Figure 3. Coumarins.
Figure 4. Stilbenes.
Simple coumarins
One of the simplest coumarins with one hydroxyl group at carbon 7 is umbelliferone (24) in Acacia nilotica (L.) Willd. ex Delile (Fabaceae, fabids) (Singh et al. ref. 2010) bactericidal for S. mutans (MIC/MBC: 15.6/15.6 µg/mL) (Meerungrueang and Panichayupakaranant ref. 2014). Addition of a hydroxyl group at carbon 8 of umbelliferone (24) forms esculetin (25) in Viola prionantha Bunge (Violaceae, fabids) active against B. cereus (MIC: 50 µg/mL) (Xie et al. ref. 2004). Methoxylation of umbelliferone (24) in position 8 produces scopoletin (26) in Pelargonium sidoides (Geraniaceae, malvids) effective against Mycobacterium smegmatis (MIC: 7.8 µg/mL) (Mativandlela et al. ref. 2007) as well as S. aureus, Enterococcus faecium, and S. maltophilia (Buathong et al. ref. 2019). Scopoletin (26) and scoparone (27) from Canarium pentatinervium Miq. (Burseraceae, malvids) were bactericidal for S. aureus with MIC/MBC values of 25/50 and 50/100 µg/mL, respectively (Mogana et al. ref. 2020; Mfonku et al. ref. 2021). Other instances are fraxetin (28) from Fraxinus rhynchophylla Hance (Oleaceae, lamiids) (S. aureus) (Wang et al. ref. 2014), 7,8-dimethoxy-6-hydroxy-coumarin (29) from Haloxylon salicornicum (Moq.) Bunge ex Boiss. (Amaranthaceae, malvids) (M. tuberculosis, MIC: 100 µg/mL) (Bibi et al. ref. 2010), and euryacoumarin A (30) from Eurya chinensis R. Br. (Pentaphylacaceae, asterids) (Song et al. ref. 2017).
Furanocoumarins
Examples are bakuchicin (31) from Psoralea corylifolia L. (Fabaceae) (Khatune et al. ref. 2004) and isoimperatorin (32) from Prangos hulusii Şenol, Yıldırım & Seçmen (Apiaceae) (MRSA, MIC: 16 µg/mL) (Tan et al. ref. 2017). Isoimperatorin (32) is antimycobacterial (Guo et al. ref. 2014).
C-Prenylated coumarins
Antibacterial coumarins with an isoprene group at carbon 6 are found in the Rutaceae family. This is the case of desmethylsuberosine (33) in Feronia lucida Teijsm. & Binn. ex Scheff. (Rahman and Gray ref. 2002). Another example is ulopterol (34) from T. asiatica with Staphylococcus epidermidis (MIC: 15.6 µg/mL) and E. coli (MIC: 62.5 µg/mL) (Raj et al. ref. 2012). The ayurvedic medicinal plant Aegle marmelos (L.) Corrêa produces xanthoarnol (35) active against E. faecalis (MIC: 18.7 µg/mL) (Chakthong et al. ref. 2012). Examples of antimycobacterial coumarins with two isoprene groups are dentatin (36), nor-dentatin (37), and clausenidine (38) in Clausena excavata Burm.f. (Sunthitikawinsakul et al. ref. 2003).
O-Prenylated coumarins
They are common in the Rutaceae and Apiaceae families. Imperatonin (39) and marmin (40) from A. marmelos inhibited the growth of M. tuberculosis with IC50 values of 12.4 and 4.3 µg/mL, respectively (Chinchansure et al. ref. 2015). Similarly, 8-geranyloxy-5,7-dimethyloxycoumarin (41) from T. asiatica was antimycobacterial (Phatchana and Yenjai ref. 2014). In the Apiaceae family, Ferula pseudalliacea Rech.f. produces sanandajin (42) (S. aureus) (Dastan et al. ref. 2016).
3-Phenyl coumarins
These coumarins are found in the Fabaceae family. We can cite glycycoumarin (43) from Glycyrrhiza glabra L. with S. mutans (MIC: 12.5 µg/mL) (Demizu et al. ref. 1988) and Haemophilus influenzae (MIC: 25 µg/mL), as well as glycyrin (44) against H. influenzae and Moraxella catarrhalis (MIC: 25 µg/mL) (Tanaka et al. ref. 2001). Other examples are psoralidine (45) from P. corylifolia (Khatune et al. ref. 2004) and indicanin B (46) from Erythrina indica Lam. the latter active against S. aureus (MIC: 9.7 µg/mL) and M. smegmatis (MIC: 18.5 µg/mL) (Waffo et al. ref. 2000). Wedelolactone (47) from Eclipta alba (L.) Hassk. (Asteraceae, campanulids) inhibited the growth of S. aureus, Salmonella typhimurium, and S. epidermidis with MIC values of 20, 25, and 15 µg/mL, respectively (Dalal et al. ref. 2010).
4-Phenyl prenylated coumarins
These coumarins are prevalent in the fabids. Examples are calophyllolide (48), inophyllum C (49), and inophyllum E (50) from Calophyllum inophyllum L. (Calophyllaceae) (Yimdjo et al. ref. 2004) as well as mesuol (51) (MDR-S. aureus, MIC: 2 µg/mL) from Mesua ferrea L. (Calophyllaceae) (Verotta et al. ref. 2004). Cajanuslactone (52) from Cajanus cajan (L.) Huth (Fabaceae) was bactericidal for S. aureus (MIC/MBC: 31/125 µg/mL) (Kong et al. ref. 2010).
Benzocoumarins
Dendrobium nobile Lindl. (Orchidaceae, monocots), is an orchid used in traditional Chinese medicine that produces dendrocoumarin (53) which inhibited S. aureus, E. coli, Micrococcus tetragenus, Kocuria rhizophila, and B. cereus with MIC values of 2.5, 0.6, 5, 5 and 2.5 µg/mL, respectively (Zhou et al. ref. 2018). From this orchid, itolide A (54) was active against S. aureus, E. coli, M. tetragenus, K. rhizophila, and B. cereus with MIC values of 2.5, 1.2, 5, 10, and 1.2 µg/mL, respectively (Zhou et al. ref. 2018).
Isocoumarins
8-Hydroxy-6-methoxy-3-pentylisocoumarin (55) from Xylosma longifolia Clos (Salicaceae, fabids) (M. tuberculosis: 40.5 µg/mL) (Truong et al. ref. 2011).
Stilbenes
The condensation of one hydroxycinnamic acid unit with three malonyl-CoA units and decarboxylation gives rise to stilbenes (Abe ref. 2020; Valletta et al. ref. 2021). These are generally weakly antibacterial, but their spectrum of activity is broad (Mattio et al. ref. 2020) (Figure 4). Resveratrol (56) from Cassia grandis L.f. (Fabaceae) was bactericidal against S. aureus (MIC: 125/125 µg/mL), E. coli (MIC: 50/125 µg/mL) (Kusumaningtyas et al. ref. 2020), S. mutans (50/50 µg/mL), and S. sanguis (50/100 µg/mL) (Yim et al. ref. 2010). Resveratrol (56) was bacteriostatic for a panel of Gram-positive bacteria (Paulo et al. ref. 2010). Hydroxylation of resveratrol (56) at carbon 3′ forms piceatannol (57), which was bactericidal for S. aureus (MIC/MBC: 125/125 µg/mL) and E. coli (MIC/MBC: 100/125 µg/mL) (Kusumaningtyas et al. ref. 2020). Methoxylation of resveratrol (56) at positions 3 and 5 gives rise to pterostilbene (58), which was bactericidal for B. cereus (MIC: 25 µg/mL) (Shih et al. ref. 2021). Smiglastilbene (59) from Smilax glabra Roxb. (Smilacaceae, monocots) was weakly active against Gram-positive bacteria (Xu et al. ref. 2013). Rhaponticin (60) from Rheum rhaponticum L. (Polygonaceae, malvids) was weakly bactericidal against M. tuberculosis (MIC/MBC: 128/256 µg/mL) (Smolarz et al. ref. 2013). The methoxylation of stilbenes increases their antibacterial strength. This is observable with 3-hydroxy-5-methoxystilbene (61) from P. glabrum (M. tuberculosis, MIC: 3.3 µg/mL) (Said et al. ref. 2015). An increase in activity is also observed when stilbenes are prenylated as in the case of cajanin stilbene acid (62) from C. cajan with S. epidermidis (MIC/MBC: 13/100 µg/mL), S. aureus (MIC/MBC: 25/105 µg/mL), and Bacillus subtilis (MIC/MBC: 25/250 µg/mL) (Kong et al. ref. 2010). Cajanin stilbene acid (62) was active against VRE with a MIC as low as 1 µg/mL. Interestingly, intravenous administration of cajanin stilbene at a dose of 5 mg/kg per day for 7 days resulted in a 90% survival rate in rodents infected with VRE (Tan, Hua, et al. ref. 2020).
Dihydrostilbenes
Reduction of the Δ7,7′ double bond of stilbenes forms antibacterial dihydrostilbenes in monocots. We can cite for instance dihydropinosylvine (63) from Dioscorea batatas Decne. (Dioscoreaceae, Monocots) (Takasugi et al. ref. 1987) and the phytoalexin desmethylbatatasin IV (64) (P. aeruginosa, MIC: 10 µg/mL) (Fagboun et al. ref. 1987; Adesanya et al. ref. 1989). Bletilla striata (Thunb.) Rchb. F. (Orchidaceae) which is used in traditional Chinese medicine, produces batatasin III (65) as well as an unusual type of dihydrostilbenes with ethylbenzene groups, namely bulbocol (66), shanciguol (67), and shancigusine B (68) active against S. aureus with MIC values of 9, 7, and 3 µg/mL, respectively (Jiang et al. ref. 2019).
Oligostilbenes
Plants of the family Vitaceae (Rosids) and Dipterocarpaceae (Malvids) use resveratrol (56) to construct oligostilbenes active against Gram-positive bacteria. ɛ-Viniferin (69) from Vitis amurensis Rup. (Vitaceae) was bacteriostatic for MRSA (MIC: 50 µg/mL) (Basri et al. ref. 2014) and bactericidal for S. mutans and S. sanguis with MIC/MBC values of 25/50 and 50/50 µg/mL, respectively (Yim et al. ref. 2010). Other examples include balanocarpol (70), vaticanol B (71) (Sahidin et al. ref. 2017), and flexuosol A (72) from Dryobalanops lanceolata Burck (Dipterocarpaceae) (Wibowo et al. ref. 2011). We can also cite dehydro-δ-viniferine (73) from Dryobalanops rappa Becc (Wibowo et al. ref. 2022) bacteriostatic for S. aureus (MIC/MBC: 2/16 µg/mL) (Mattio et al. ref. 2019).
Miscellaneous
The prenylated stilbene derivative lakoochin A (74) from Artocarpus lakoocha Wall. ex Roxb. (Moraceae) inhibited the growth of M. tuberculosis (MIC: 12.5 µg/mL) (Puntumchai et al. ref. 2004).
Diarylheptanoids
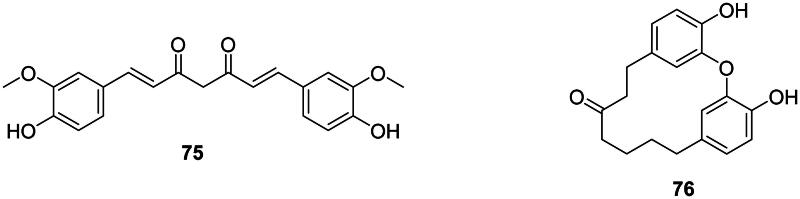
The addition of two units of p-coumaric acid (2) or ferulic acid (14) with one malonyl-CoA unit forms antibacterial diarylheptanoids (Abe ref. 2020) such as curcumin (75) in Curcuma longa L. (Zingiberaceae) (Gunes et al. ref. 2016). The cyclic diaryheptanoid engelhardione (76) from Engelhardia roxburghiana Wall. (Juglandaceae, fabids) inhibited the growth of M. tuberculosis with the MIC value of 2 µg/mL (Lin et al. ref. 2005) (Figure 5).
Lignans
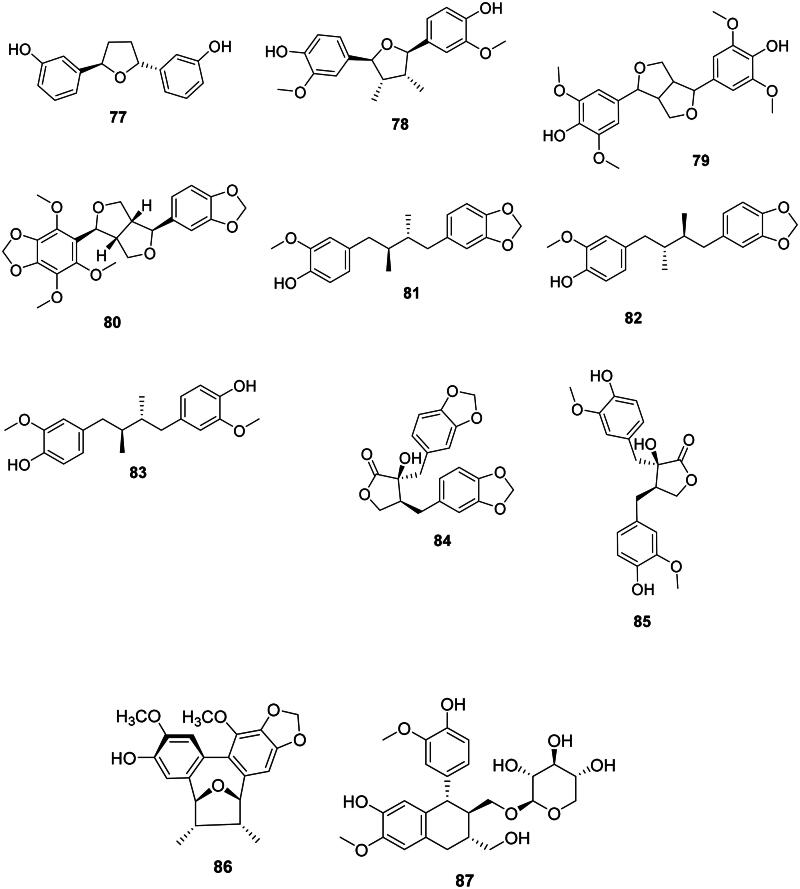
These phenolic compounds come from the coupling of two phenylpropanoid units between carbons 8 and 8′ (Lewis and Davin ref. 1999; Satake et al. ref. 2015) (Figure 6).
Tetrahydrofurans
Ammaniol (77) from Ammannia multiflora Roxb. (Lythraceae, malvid) inhibited the growth of M. tuberculosis (MIC: 25 µg/mL) (Upadhyay et al. ref. 2012). Nectandrine B (78) from Myristica fragrans Houtt. (Myristicaceae, magnoliids) was active against Pseudomonas syringae (IC50: 63 µg/mL) (Cho et al. ref. 2007).
Furanofurans
In upper Angiosperms, examples are syringaresinol (79) from Canthium horridum Bl. (Rubiaceae, lamiids) (Yang et al. ref. 2010) and ecbolin A (80) in Ecbolium viride (Forssk.) Alston (Acanthaceae, lamiids) (S. aureus, MIC: 7.8 µg/mL) (Cecilia et al. ref. 2012).
Dibenzylbutanes
Macelignan (81) from M. fragrans was bactericidal against S. mutans (MIC/MBC: 3.9/7.8 µg/mL) (Chung et al. ref. 2006). From this plant, erythro-austrobailignan-6 (82) and meso-dihydroguaiaretic acid (83) inhibited the growth of Agrobacterium tumefaciens with IC50 values of 17 and 23 µg/mL, respectively (Cho et al. ref. 2007). erythro-Austrobailignan-6 (82) was active against MRSA and MDR-M. tuberculosis with the MIC values of 50 µg/mL, respectively (Reyes-Melo et al. ref. 2017).
Dibenzylbutyrolactones
Examples are meridinol (84) in Lasia spinosa (L.) Thwaites (Araceae, monocots) (Hasan et al. ref. 2011) as well as (−)-nortrachelogenin (85) from Patrinia scabisifolia Link (Caprifoliaceae, campanulids), the latter being active against E. coli O157:H7 (Lee, Ji, et al. ref. 2016).
Dibenzocyclooctadienes
Manglisin B (86) from Manglietiastrum sinicum Y.W. Law (Magnoliaceae, magnoliids) (Ding et al. ref. 2014; Qiang et al. ref. 2022).
Aryltetralins
Schizandriside (87) from Acer truncatum Bunge (Sapindaceae, malvids) developed an inhibition zone against S. aureus (2 µg/disc) (Dong et al. ref. 2006; Shen et al. ref. 2022).
Neolignans
These lignans come from the coupling of two phenylpropanoid units between carbons other than 8 and 8′ (Teponno et al. ref. 2016) (Figure 7).
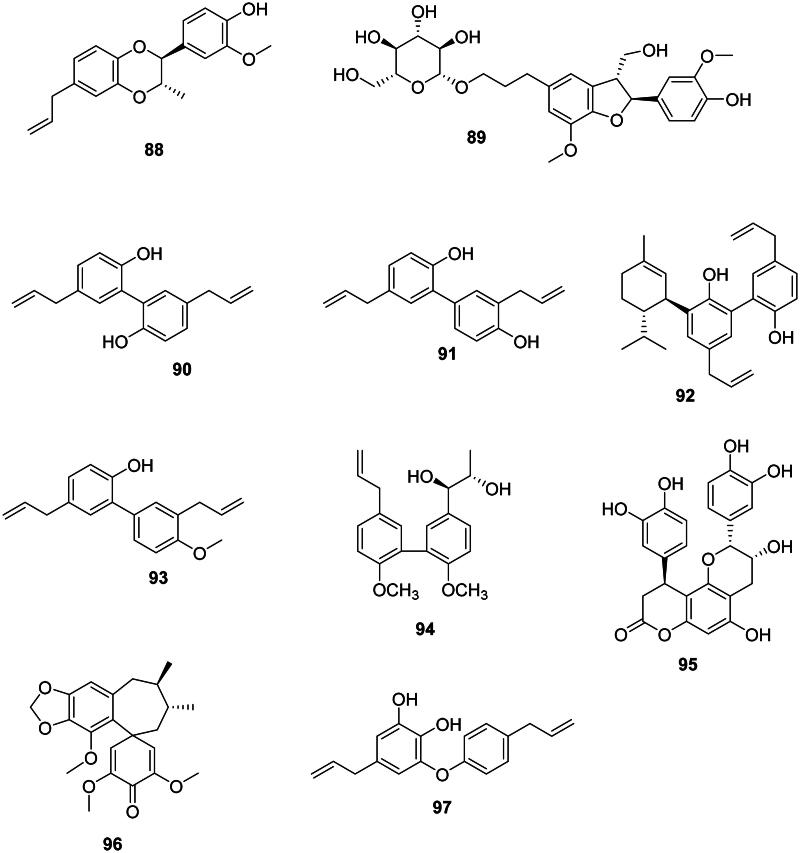
Benzodioxanes
Melaleucin A (88) from Melaleuca bracteata F. Muell. (Myrtaceae, malvids) inhibited the growth of MRSA (MIC: 8 µg/mL) (Li et al. ref. 2017).
Dihydrobenzofuran lignans
The 8-5′ coupling of two phenylpropanoid units (Wang, Wang, et al. ref. 2022) yields dihydrobenzofuran lignans which have broad-spectrum antibacterial effects. An example is glochidioboside (89) in the genus Glochidion J.R. Forst. & G. Forst. (Phyllanthaceae, fabids) which inhibited the growth of E. coli O157:H7 (Lee, Woo, et al. 2015).
Biphenyl lignans
The coupling of two phenylpropanoid units between carbons 3 and 3′ forms biphenyl lignans mostly found in the Magnoliaceae family. Examples are magnolol (90) and honokiol (91) in Magnolia officinalis Rehder & E.H. Wilson active against Gram-positive bacteria (Ho et al. ref. 2001). Honokiol (91) was bactericidal against S. aureus, B. subtilis, Propionibacterium acnes, and Propionibacterium granulosum with MIC/MBC values of 13.1/26.6, 8.2/16.7, 4.1/16.7, and 8.2/16.7 µg/mL, respectively (Kim et al. ref. 2010) as well as MRSA (MIC: 12.5 µg/mL) (Syu et al. ref. 2004). From M. officinalis, piperitylmagnolol (92) gave MIC values of 12.5, 6.2, 6.2, and 6.2 µg/mL with S. aureus, MRSA, E. faecalis, and VRE, respectively, and was bactericidal against VRE (Syu et al. ref. 2004). Other examples are 3,5′-diallyl-2′-hydroxy-4-methoxybiphenyl (93) from Magnolia grandiflora L. (Clark et al. ref. 1981). In the family Moraceae, an example is (7′R,8′S)-4,4′-dimethoxy-strebluslignanol (94) from Streblus asper Lour. (Moraceae) (Nie et al. ref. 2016).
Miscellaneous lignans
These antibacterial lignans are abundant in basal Angiosperms and include cinchonain Ib (95) from S. glabra (Xu et al. ref. 2013), manglisin A (96) from M. sinicum (Ding et al. ref. 2014), and the biphenyl ether lignan obovatol (97) from Magnolia obovata Aiton (Magnoliaceae) (Ito et al. ref. 1982).
Hydroxybenzoic acid derivatives
They derive from the shikimate pathway (Ossipov et al. ref. 2003) and have a broad spectrum of antibacterial activity (Figure 8). One of the simplest examples is 4-hydroxybenzoic acid (98) from Oryza sativa L. (Poaceae, monocots) (Cho et al. ref. 1998). Further addition of hydroxyl groups at positions 3 and 5 forms gallic acid (99) active against S. aureus, MRSA (MIC: 64 µg/mL), M. tuberculosis (MIC: 66.6 µg/mL) (Deng et al. ref. 2013), and S. epidermidis (Adesina et al. ref. 2000). Methyl gallate (100) from Rhus chinensis Mill. (Anacardiaceae, malvids) was active against P. aeruginosa and E. coli with MIC values of 12.5 and 25 µg/mL, respectively (Saxena et al. ref. 1994) as well as against S. aureus (MIC: 7.8 µg/mL) (Xu et al. ref. 2015), Vibrio cholerae (MIC: 30 µg/mL) (Sánchez et al. ref. 2013), Salmonella typhi (MIC: 3.9 µg/mL) (Choi et al. ref. 2014), and M. tuberculosis (MIC: 50 µg/mL) (Hernández-García et al. ref. 2019). Methyl gallate (100) was weakly bactericidal against Shigella dysenteriae (MIC/MBC: 128/256, µg/mL) (Acharyya et al. ref. 2015) and K. pneumoniae (MIC/MBC: 100/300 µg/mL (Li, Lin, et al. ref. 2016). From Rhus glabra L. (Anacardiaceae), 4-methoxy-3,5-dihydroxybenzoic acid (101) was broadly antibacterial (Saxena et al. ref. 1994).
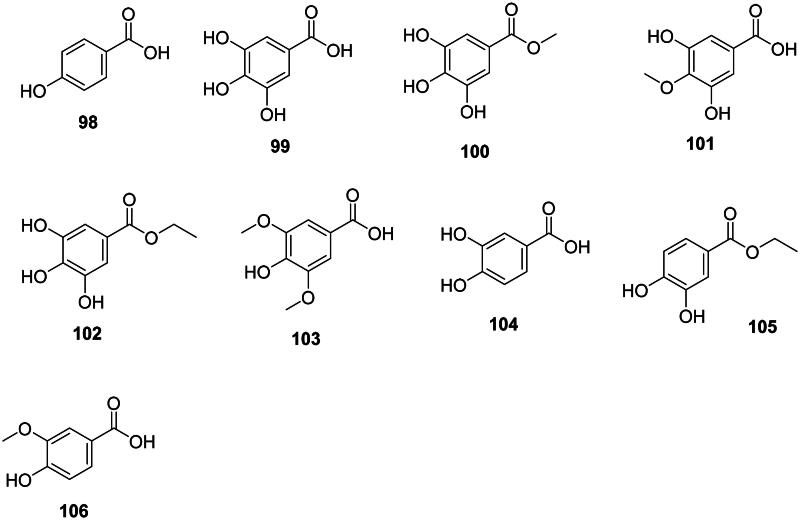
In the family Saxifragaceae (core eudicots), Saxifraga melanocentra Franch. produces ethyl gallate (102) which is bactericidal against K. pneumoniae (Li, Lin, et al. ref. 2016). Methoxylation of gallic acid (99) at positions 3 and 5 forms syringic acid (103) in Ardisia elliptica Thunb. (Myrsinaceae, asterids) active against S. typhimurium (MIC: 62.5 µg/mL) (Phadungkit and Luanratana ref. 2006).
Protocatechuic acid (104) (Metsämuuronen and Sirén ref. 2019) from Arbutus unedo L. (Ericaceae, asterids) was active against A. baumannii (Liu et al. ref. 2005), while protocatechuic acid ester (105) from Arachis hypogaea L. (Fabaceae) was weakly active against S. aureus (Miklasińska et al. ref. 2015). Protocatechuic acid (104) is the precursor of vanillic acid (106) (Metsämuuronen and Sirén ref. 2019) which was active against M. tuberculosis (MIC: 83.3 µg/mL) (Deng et al. ref. 2013).
Miscellaneous simple phenolic compounds
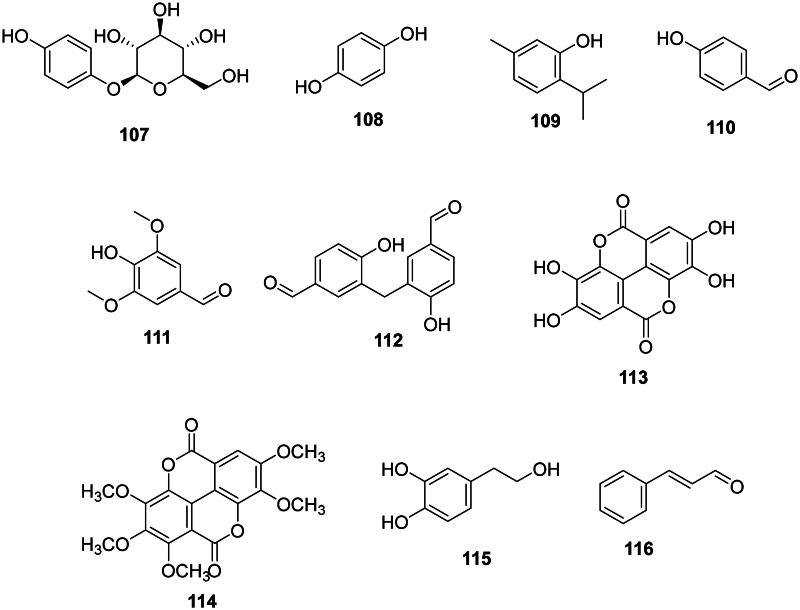
Examples are arbutin (107) and hydroquinone (108) from A. unedo (Jurica et al. ref. 2017) (Figure 9), thymol (109) from Thymus vulgaris L. (Lamiaceae) (E. coli, MIC: 8 µg/mL) (Xu et al. ref. 2008), 4-hydroxybenzaldehyde (110) from Alpinia conchigera Griff. (Zingiberaceae) (Aziz et al. ref. 2012), syringaldehyde (111) from Juglans regia L. (Juglandaceae) (Colaric et al. ref. 2005) (S. mutans, MIC/MBC: 62.5/62.5 µg/mL) (Meerungrueang and Panichayupakaranant ref. 2014), 3,3′-methylene-bis(4-hydroxybenzaldehyde) (112) from S. asper (B. subtilis, MIC: 27 µg/mL) (Nie et al. ref. 2016), ellagic acid (113) (Ghudhaib et al. ref. 2010), 3,3′,4,4′,5′-pentamethylcoruleoellagic acid (114) from Rhodamnia dumetorum (DC.) Merr. and L.M Perry (Haemophilus influenza, MIC: 9.3 µg/mL) (Lakornwong et al. ref. 2018; Munvera et al. ref. 2020), hydroxytyrosol (115) from S. cuneata (S. aureus, MIC: 2 µg/mL) (Zeng et al. ref. 2015), and cinnamaldehyde (116) in Cinnamomum cassia (L.) J. Presl (Lauraceae) (Firmino et al. ref. 2018).
Benzoquinones
Phenolic compounds in this group are generally strongly antibacterial (Figure 10). Examples are 2,6-dimethoxy-1,4-benzoquinone (117) from Ficus foveolata Pittier (Moraceae) bactericidal for S. mutans (MIC/MBC: 7.8/7.8 µg/mL) (Nishina et al. ref. 1991; Meerungrueang and Panichayupakaranant ref. 2014) and thymoquinone (118) from Nigella sativa L. (Ranunculaceae, eudicots) (Dey et al. ref. 2014) bactericidal for Listeria monocytogenes (MIC: 8/8 µg/mL) and S. aureus (MIC/MBC 8/16 µg/mL) (Chaieb et al. ref. 2011) and active against E. coli (Cetin-Karaca and Newman ref. 2015). Another example is abruquinone B (119) from Abrus precatorius L. (Fabaceae) (M. tuberculosis, MIC: 12.5 µg/mL) (Limmatvapirat et al. ref. 2004). Pulsaquinone (120) from Pulsatilla koreana (Yabe ex Nakai) Nakai ex T. Mori (Ranunculaceae) was active against P. acnes, B. subtilis, S. aureus, S. mutans, P. aeruginosa, and S. sonnei with the MIC values of 2, 2.7, 2, 2, 3.3, and 2 µg/mL, respectively (Cho et al. ref. 2009).
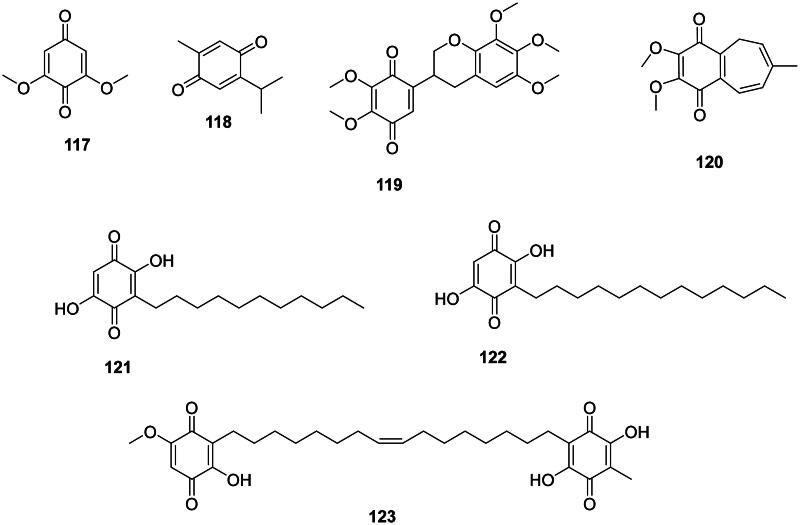
Plants of the Myrsinaceae family produce antibacterial benzoquinones substituted with long-chain alkyl groups, such as embelin (121) from Embelia ribes Burm.f. bactericidal for S. aureus (MIC/MBC: 20/75 µg/mL) (Chitra et al. ref. 2003; Radhakrishnan et al. ref. 2011), rapanone (122) in Ardisia crenata Sims (Podolak et al. ref. 2021), and ardisiaquinone B (123) from Ardisia sieboldii Miq. (Enterobacter aerogenes, MIC: 16 µg/mL) (Ogawa and Shinsaku ref. 1968; Omosa et al. ref. 2016).
1,4-Naphthoquinones
Simple 1,4-naphthoquinones
Simple 1,4-naphthoquinones originate from the polyketide or shikimate pathways (Widhalm and Rhodes ref. 2016). These are among the most potent known antibacterial compounds found in Angiosperms (Figure 11). An example is juglone (124) (Zmantar et al. ref. 2016), bacteriostatic against Streptococcus pyogenes (MIC/MBC: 1.5/100 µg/mL) (Macé et al. ref. 2017) and active against M. smegmatis (MIC: 0.7 µg/mL) (Clark et al. ref. 1990). 3-Methoxyjuglone (125) from E. roxburghiana was active against M. tuberculosis with a MIC as low as 0.2 µg/mL (Lin et al. ref. 2005). 2-Methoxy-1,4-naphthoquinone (126) (lawsone methyl ether) from Impatiens balsamina L. (Balsaminaceae, asterids) inhibited the growth of Aeromonas salmonicida and MRSA with MIC values of 2 and 15.6 μg/mL, respectively (Yang et al. ref. 2001).
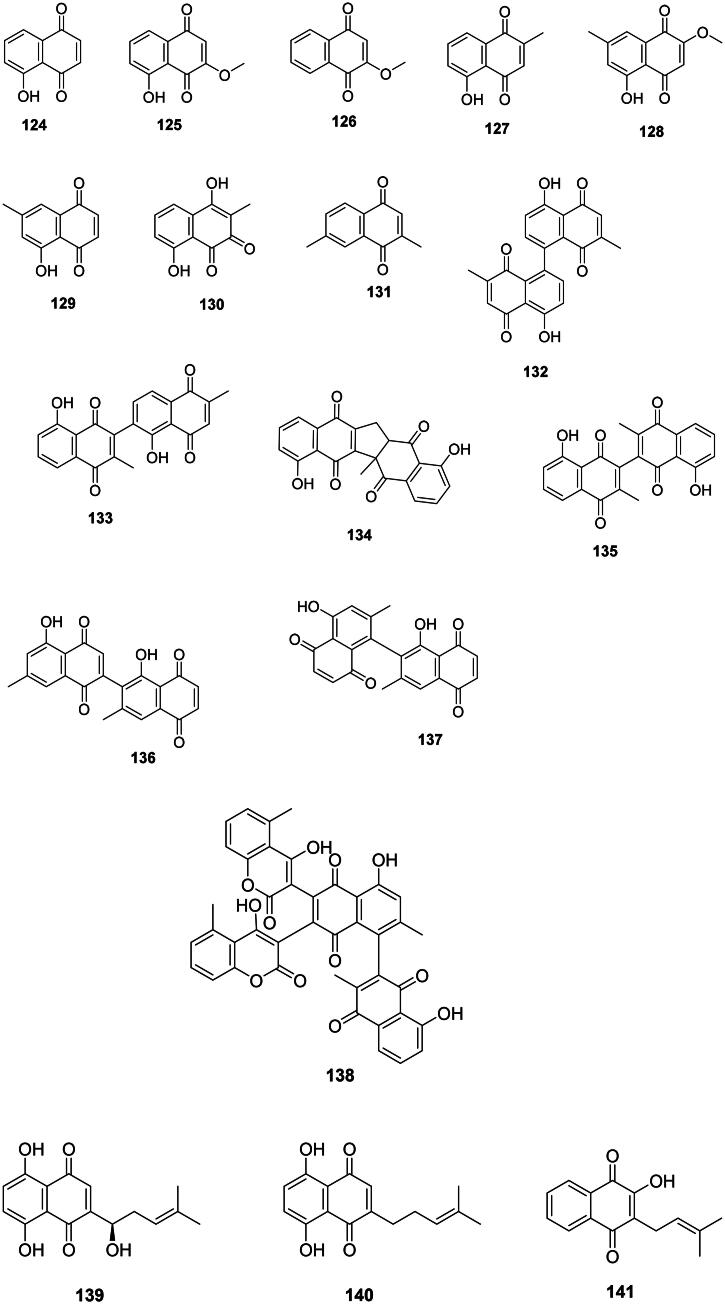
The condensation of one acetyl-CoA unit with five malonyl-CoA units forms naphthoquinones with strong antibacterial effects in the order Ericales (asterids). An example is plumbagin (127) from Diospyros kaki Thunb. (Ebenaceae) (Lee and Lee ref. 2008) active against S. epidermidis (MIC: 0.7 µg/mL) (Jeyachandran et al. ref. 2009), Neisseria gonorrhoae (MIC: 19.5 µg/mL) (Kuete et al. ref. 2009), Gardnerella vaginalis (Sobhani et al. ref. 2018), MDR-M. tuberculosis (MIC/MBC: 0.2/1.5 µg/mL), and bactericidal for Proteus vulgaris (MIC/MBC: 16/16 µg/mL) (Dey et al. ref. 2014). Plumbagin (127) was bactericidal for M. smegmatis and M. tuberculosis (MIC/MBC: 4.8/9.7 µg/mL) (Kuete et al. ref. 2009). From D. kaki, 2-methoxy-7-methyl juglone (128) (Gu et al. ref. 2004) inhibited the growth of M. tuberculosis with a MIC as low as 0.5 µg/mL (selectivity index: 30.2) (Mahapatra et al. ref. 2007). Diospyros maritima Bl. (Ebenaceae) yields 7-methyljuglone (129) active against M. tuberculosis (MIC: 0.5 µg/mL) (Bapela et al. ref. 2006) and bacteriostatic for M. smegmatis (MIC/MBC: 1.5/15.6 µg/mL) (McGaw et al. ref. 2008). D. maritima produces droserone (130) which inhibited the growth of pan-resistant Mycobacterium tuberculosis (clinical isolate CIBIN 99) with the MIC value of 25 µg/mL) (Uc-Cachón et al. ref. 2014). Chimaphilin (131) from Monenes uniflora L. (Ericaceae) was active against S. aureus (MIC: 25 µg/mL) (Saxena et al. ref. 1996) and M. tuberculosis (IC50: 5.4 µg/mL) (Li et al. ref. 2018).
1,4-Naphthoquinones oligomers
These phenolic compounds are produced from the oxydative coupling of naphthoquinones in the Ebenaceae (Figure 11). Examples of dimers of plumbagin (127) are maritinone (132), chitranone (133), zeylanone (134), and 3,3′-biplumbagin (135) in D. maritima (Gu et al. ref. 2004) which inhibited pan-resistant M. tuberculosis (clinical isolate CIBIN 99) with the MIC values of 3.1, 3.1, 12.5, and 3.1 µg/mL, respectively (Uc-Cachón et al. ref. 2014). Of note, the selectivity indexes of maritinone (132) and 3,3′-biplumbagin (135) were 74.3 and 194.1, respectively (Uc-Cachón et al. ref. 2014).
Diospyrin (136) formed from the coupling between a pair of 7-methyljuglone (129) is active against Corynebacterium dyphtheriae (MIC: 3.1 µg/mL) (Adeniyi et al. ref. 2000) and bacteriostatic for Mycobacterium bovis (MIC/MBC: 1.7/39 µg/mL) (McGaw et al. ref. 2008). Likewise, isodiospyrin (137) inhibited Streptococcus pneumoniae with a MIC value as low as 0.7 µg/mL (McGaw et al. ref. 2008). An example of 1,4-naphthoquinone dimer coupled with coumarins is diospyrone (138) active against MDR-K. pneumoniae and MDR-P. aeruginosa (Kuete et al. ref. 2009).
Prenylated 1,4-naphthoquinones
Lamiids and plants of the Boraginaceae family combine a 4-hydroxybenzoic acid unit with a geranyl group to form antibacterial prenylated 1,4-naphthoquinones with naphthazarin scaffolds (Rajbhandari et al. ref. 2007). This is the case of shikonin (139) and deoxyshikonin (140) from Lithospermum erythrorhizon Siebold & Zucc. used in traditional Chinese medicine (Brigham et al. ref. 1999). Shikonin (139) from Arnebia euchroma (Royle ex Benth.) I.M. Johnst. was bactericidal against MRSA (MIC/MBC: 6.2/12.5 µg/mL) (Shen et al. ref. 2002). In the Bignoniaceae family, an example is lapachol (141) from Oroxylum indicum (L.) Kurz (Bignoniaceae) (Ali et al. ref. 1998).
Anthraquinones
Simple anthraquinones
Angiosperms produce antibacterial anthraquinones from the polyketide or shikimate pathways (Figure 12). For example, chrysophanol (142) from R. rhaponticum, formed by the addition of one acetyl-CoA unit to seven malonyl-CoA units, was bactericidal against M. tuberculosis (64/128 µg/mL) (Smolarz et al. ref. 2013) and active against S. epidermidis (MIC: 31.2 µg/mL) (Coopoosamy and Magwa ref. 2006a). The oxidation of chrysophanol (142) at carbon 3 forms aloe-emodin (143) in R. rhaponticum (Coopoosamy and Magwa ref. 2006b; Alaadin et al. ref. 2007; Lee, Kang, et al. ref. 2010). Aloe-emodin (143) was active against MRSA (MIC: 2 µg/mL) (Hatano et al. ref. 1999; Alaadin et al. ref. 2007), S. mutans (MIC: 1.2 µg/mL) (Zheng et al. ref. 2011), and bactericidal for M. tuberculosis (MIC/MBC: 64/128 µg/mL) (Smolarz et al. ref. 2013). The oxidation of aloe-emodin (143) at carbon 3 forms rhein (144) in Rheum officinale Baill. (Polygonaceae) effective against MRSA (MIC: 15.6 µg/mL) (Joung et al. ref. 2012), Bacteroides fragilis (MIC: 1.5 µg/mL) (Cyong et al. ref. 1987), and Porphyromonas gingivalis (MIC: 2.5 µg/mL) (Azelmat et al. ref. 2015).
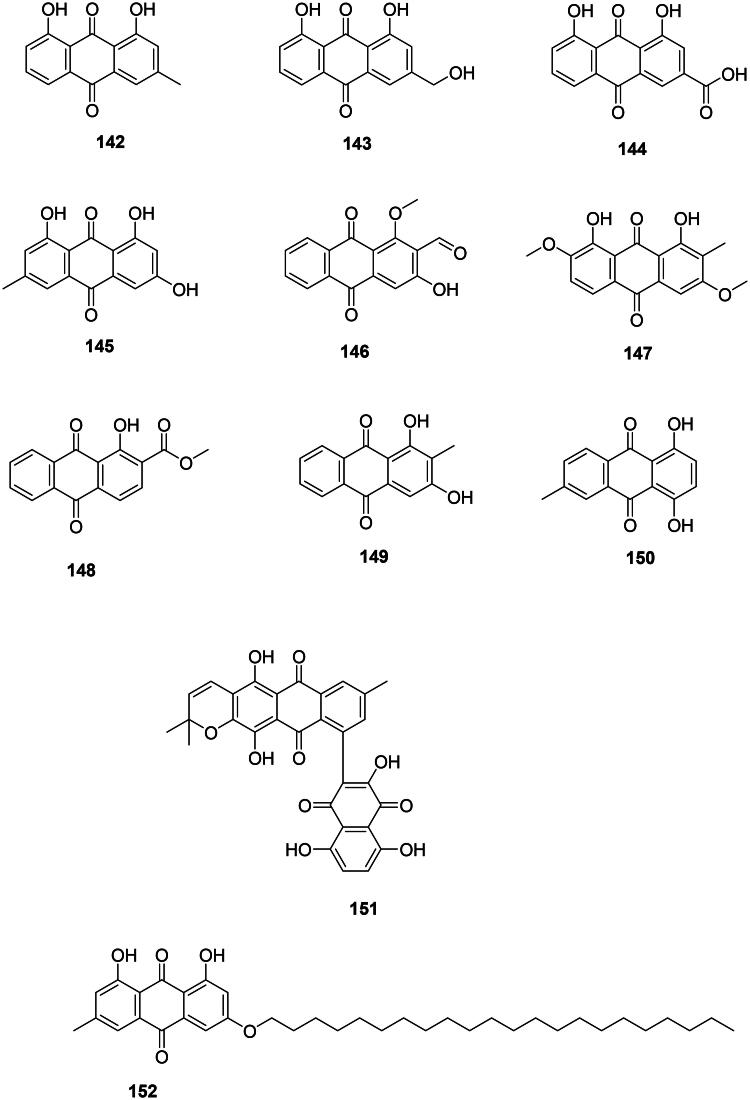
Another example is emodin (145) from Cassia alata L. (Fabaceae) with methicillin-sensitive S. aureus (MIC: 25 µg/mL) (Joung et al. ref. 2012), S. aureus (MIC: 8 µg/mL) (Yan et al. ref. 2017), MRSA (MIC: 1.5 µg/mL) (Promgool et al. ref. 2014), M. tuberculosis (MIC/MBC: 4/8 µg/mL) (Dey et al. ref. 2014), B. cereus (MIC/MBC: 8/8 µg/mL) (Dey et al. ref. 2014), and Haemophilus parasuis (MIC/MBC: 32/64 µg/mL) (Li, Song, et al. ref. 2016).
In the family Rubiaceae, examples are damnacanthal (146) from Morinda elliptica (Hook.f.) Ridl. (M. tuberculosis, MIC: 13 μg/mL) (Pollo et al. ref. 2020), 1,8-dihydroxy-2-methyl-3,7-dimethoxyanthraquinone (147) from Morinda angustifolia Roxb. (Xiang et al. ref. 2008), 1-hydroxy-2-methoxycarbonyl-anthraquinone (148) from Coptosapelta flavescens Korth. (MRSA, MIC: 16 µg/mL) (Kongyen et al. ref. 2014), and rubiadin (149) from Rubia tinctoria L. (S. aureus, MIC: 32 µg/mL) (Comini et al. ref. 2011).
6-Methyl-1,4-dihydroxyanthraquinone (150) from Tectona grandis L.f. (Verbenaceae) was bacteriostatic for Klebsiella aerogenes (16/128 µg/mL) (Bitchagno et al. ref. 2015).
Miscellaneous anthraquinones
T. grandis produces an unusual dimer of anthraquinone and naphthoquinone: tectograndone (151), bactericidal for E. coli (MIC/MBC: 32/128 µg/mL) (Bitchagno et al. ref. 2015). Revandchinone-3 (152) from Rheum emodi Wall. (Polygonaceae) inhibited the growth of a broad spectrum of bacteria (Babu et al. ref. 2003).
Tannins
Proanthocyanidins
The coupling of two catechin and or epigallocatechin units forms proanthocyanidins (Figure 13). These phytoanticipins are weakly antibacterial but their spectrum of activity is broad. An example is cinnamtanin B1 (153) from Vaccinium vitis-idaea L. (Ericaceae) against P. gingivalis and Prevotella intermedia (MIC: 100 µg/mL) (Ho et al. ref. 2001). Another illustration is (+)-epigallocatechin-(2β→O→7, 4β→8)-(+)-catechin (154) from Quercus ilex L (Fagaceae, fabids) (Karioti et al. ref. 2011). The antibacterial spectrum of proanthocyanidin oligomers and polymers is limited to Gram-positive bacteria as in ZP-CT-A from Zanthoxylum piperitum DC (Rutaceae) (MRSA, MIC: 128 µg/mL) (Kusuda et al. ref. 2006), theasinensin A. (155) and B (156) (MRSA, MIC: 64 µg/mL) (Hatano et al. ref. 2003), and proanthocyanidins from Diospyros kaki L. (Ebenaceae) (Wang et al. ref. 2020).
Figure 13. Tannins.
Gallotannins
The esterification of a glucose unit by several units of gallic acid (99) forms gallotannins (Ossipov et al. ref. 2003). These phytoanticipins are weakly antibacterial and include 1,2,3,4,6-penta-O-galloyl-β-d-glucose (157) from A. truncatum (S. aureus, MIC: 60 µg/mL) (Zhang et al. ref. 2008; Lin et al. ref. 2011) and tannic acid (158) from Alnus japonica (Thunb.) Steud. (Betulaceae, fabids) (Wu et al. ref. 2010).
Ellagitannins
Ellagitannins result from the coupling of two adjacent units of gallic acid (99) within gallotannins and generally have weak but broad-spectrum antibacterial activities (Figure 13) (Al-Harbi et al. ref. 2017). These tannins are found in fabids and include, for example, corilagin (159) and geraniin (160) from Acalypha wilkesiana Müll. Arg. (Euphorbiaceae) active against S. aureus with MIC values of 50 and 25 µg/mL, respectively (Adesina et al. ref. 2000). Corilagin (159) was active against E. coli (MIC: 62.5 µg/mL) (Li et al. ref. 2013) and geraniin (159) with Vibrio vulficus (MIC: 25 µg/mL) (Taguri et al. ref. 2006). In the malvids, examples are castalagin (161) from Terminalia catappa L. (Combretaceae) (Clostridium perfringens, MIC: 67 µg/mL) (Taguri et al. ref. 2006) and punicalagin (162) (S. aureus, MIC: 250 µg/mL) from Punica granatum L. (Lythraceae) (Xu et al. ref. 2017; Li et al. ref. 2020). The oligomeric ellagitannin isorugosin A from Liquidambar formosana Hance (Altingiaceae, core eudicots) was active against MRSA (Shimozu et al. ref. 2017).
Miscellaneous phenolic compounds
Long-chain alkyl phenols
These phenolic compounds originate from the polyketide pathway and are strongly active against Gram-positive bacteria (Sampietro et al. ref. 2013) (Figure 14). In the basal Angiosperms, magnoliids produce antibacterial alkylresorcinols, such as knerachelin B (163) from Knema furfuracea (Hook.f. and Thomson) Warb. (Myristicaceae) (S. aureus, MIC: 4 µg/mL) (Zahir et al. ref. 1993), malabaricone A (164) (S. aureus, MIC: 0.5 µg/mL, bactericidal, selectivity index ≥ 80), and malabaricone B (165) (MRSA, MIC: 0.5 µg/mL, selectivity index ≥ 80) (Sivadas et al. ref. 2023). Malabaricone B (165) inhibited the growth of VRE and MRSA with MIC values as low as 1 µg/mL and was bactericidal against MRSA (Sivadas et al. ref. 2023). Myristica fragrans produces malabaricone C (166) active against S. aureus (MIC: 4 µg/mL) (Orabi et al. ref. 1991). In monocots, Zingiber officinale Roscoe (Zingiberaceae) produces antibacterial alkyl catechols, such as [6]-gingerol (167), [10]-gingerol (168), and [12]-gingerol (169) (Hiserodt et al. ref. 1998; Park et al. ref. 2008).
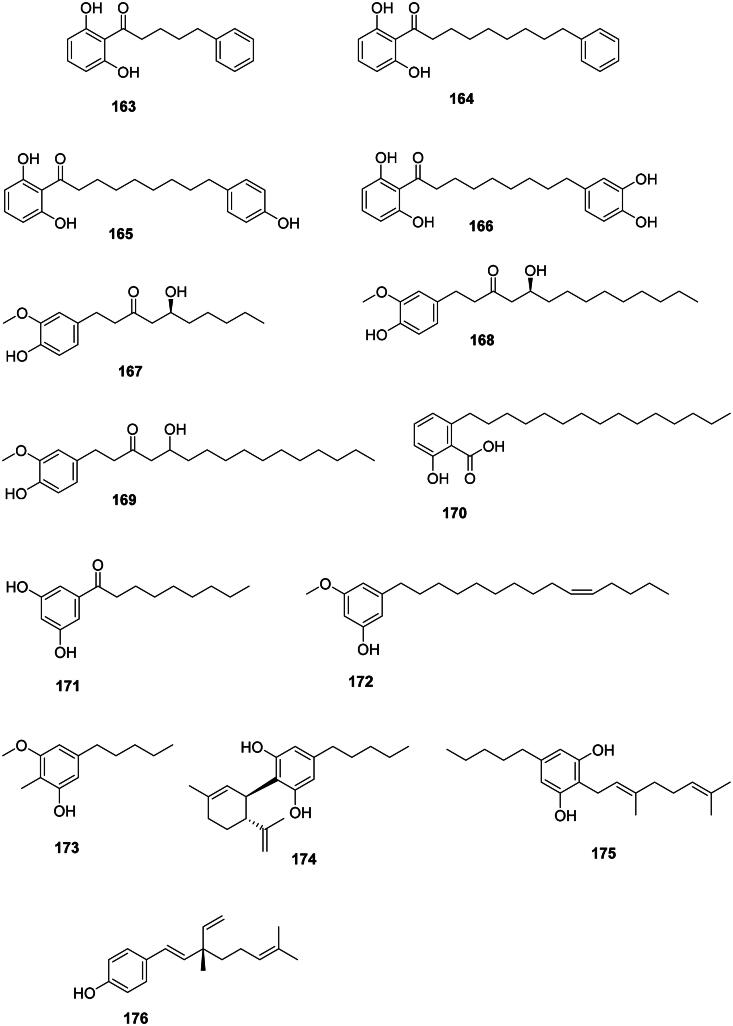
Anacardic acid (170) from Anacardium occidentale L. (Anacardiaceae) inhibited the growth of S. mutans and P. acnes with MIC values as low as 1.5 and 0.7 µg/mL, respectively (Kubo et al. ref. 1993). Other long-chain alkyl phenols of this kind have been identified in Semecarpus anacardium L.f. (Anacardiaceae) (Sundaram et al. ref. 2014).
In upper Angiosperms, Ardisia cornudentata Mez (Myrsinaceae) produces 1-(3,5-dihydroxyphenyl)nonan-1′-one (171), belamcandol (172), and 3-methoxy-2-methyl-5-pentylphenol (173) which inhibited the growth of M. tuberculosis with MIC values of 6, 33.8, and 2.5 µg/mL, respectively (Chang et al. ref. 2011). Cannabis sativa L (Cannabaceae, fabids) produce cannabidiol (174) and cannabigerol (175) active against S. aureus with MIC values as low as 0.5 and 1 µg/mL, respectively (Appendino et al. ref. 2008; Radwan et al. ref. 2009). Cannabidiol (174) inhibited the growth of E. faecium, M. catarrhalis, N. gonorrhoeae, Neisseria meningitidis, Legionella pneumophila, and A. baumannii with MIC values of 0.5, 1, 1, 0.2, 1, and 64 µg/mL, respectively (Blaskovich et al. ref. 2021). Cannabidiol (174) applied topically could treat MRSA-infected mice but was inactive when given orally (Blaskovich et al. ref. 2021). Aerva sanguinolenta (L.) Bl. (Amaranthaceae) produces bakuchiol (176) active against S. mutans with a MIC as low as 0.9 µg/mL (Rao et al. ref. 2012) as well as Mycobacterium aurum (MIC: 15. 9 µg/mL) (Newton et al. ref. 2002).
Prenylated phloroglucinols
Fabids, and to a lesser extent malvids, produce prenylated phloroglucinols active against Gram-positive bacteria (Figure 15). In the family Hypericaceae, examples are chinesin I (177) from Hypericum japonicum Thunb. (S. aureus, MIC: 3.1 µg/mL) (Nagai and Tada ref. 1987) as well as hyperjaponicol C (178) (Li et al. ref. 2018), and olympicin A (179) from Hypericum olympicum L. (Shiu et al. ref. 2012). Hypercalin A (180) and hypercalin B (181) from Hypericum acmosepalum N. Robson inhibited S. aureus (expressing NorA) with MIC values as low as 2 and 0.5 µg/mL, respectively (Osman et al. ref. 2012).
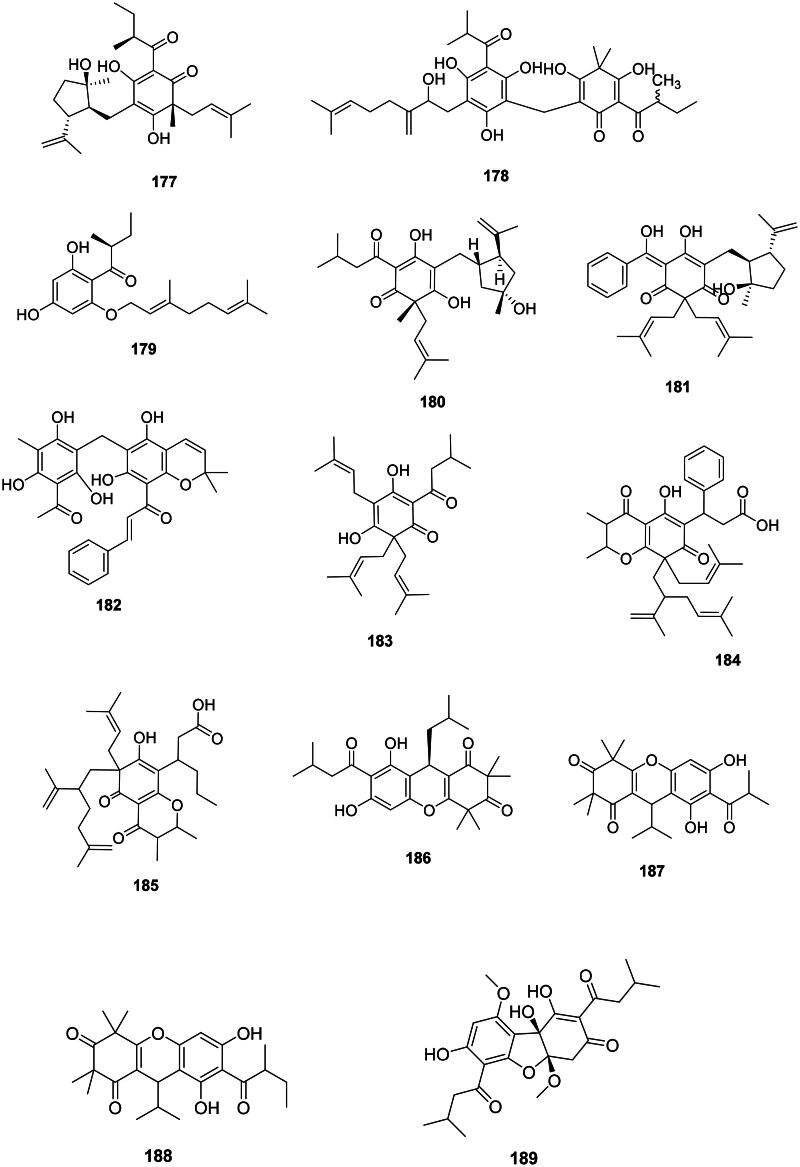
Other examples are, in the fabids, rottlerin (182) from Mallotus philippensis (Lam.) Müll. Arg. (Euphorbiaceae) (Pandey et al. ref. 2016), lupulone (183) from Humulus lupulus L. (Cannabaceae) (MRSA, MIC: 0.6 µg/mL) (Bocquet et al. ref. 2019), as well as calophynic acid (184) and brasiliensic acid (185) from C. inophyllum (Yimdjo et al. ref. 2004). In the family Myrtaceae, rhodomyrtone (186), isomyrtucommulone B (187), and myrciarone B (188) from Myrciaria dubia (Kunth) McVaugh inhibited B. subtilis with the MIC values of 0.7, 1.5, and 1.5 µg/mL, respectively (Kaneshima et al. ref. 2017). Callistemenonone A (189) from Callistemon viminalis (Sol. ex Gaertn.) G. Don was bactericidal for B. cereus (MIC/MBC: 5/20 µg/mL) (Xiang et al. ref. 2017).
Prenylated acetophenones
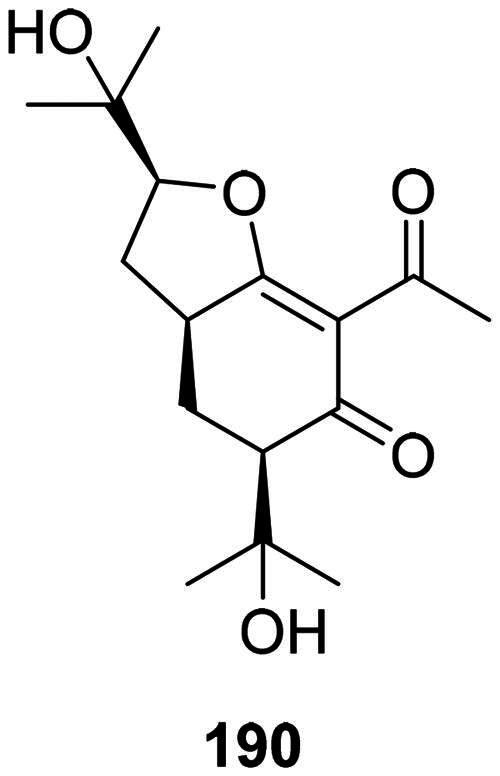
Meliviticine A (190) from Melicope viticina (Wall. ex Kurtz) T.G. Hartley (Rutaceae) inhibited the growth of MRSA, S. typhi, and P. aeruginosa (MIC: 50 µg/mL) (Li et al. ref. 2019) (Figure 16).
Prenylated benzophenones
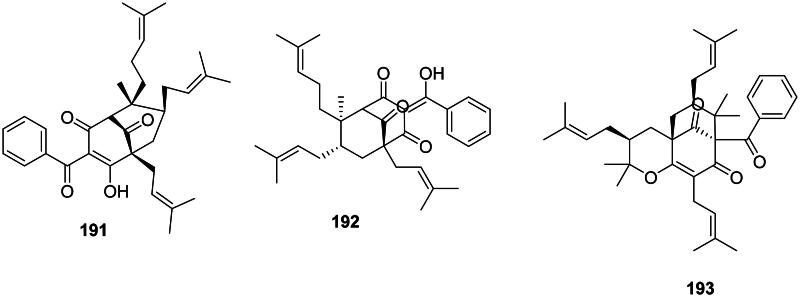
The addition of one benzoyl-CoA unit with three malonyl-CoA units and substitutions with dimethylallyl groups form prenylated benzophenones (Abe ref. 2020). They are active against Gram-positive bacteria and are found in the family Clusiaceae (fabids) (Figure 17). Cowanone (191) from Garcinia cowa Roxb. inhibited the growth of MRSA with a MIC value as low as 0.5 µg/mL (Trisuwan and Ritthiwigrom ref. 2012). Garcinia multiflora Champ. ex Benth. produces chamuangone (192) bactericidal for S. pyogenes (MIC/MBC: 7.8/31.2 µg/mL) (Sakunpak and Panichayupakaranant ref. 2012) as well as garcimultiflorone A (193) (Chen et al. ref. 2008).
Prenylated xanthones
Internal coupling of benzophenones forms a wide range of antibacterial prenylated xanthones in the Hypericaceae, Calophyllaceae, and Clusiaceae families (Figure 18). In the Hypericaceae family, examples are isocudraniaxanthone B (194), isojacareubine (195) bactericidal against MRSA (SCCmec III) (MIC/MBC: 4/16 µg/mL) (Zuo et al. ref. 2012), and cochinchinone A (196) (P. aeruginosa, MIC: 4.7 µg/mL) from Cratoxylum cochinchinense (Lour.) Bl. (Boonnak et al. ref. 2009). Other examples are gerontoxanthone I (197) and 9-hydroxycalabaxanthone (198) from Cratoxylum formosum (Jack) Benth. & Hook.f. ex Dyer (S. typhi, MIC: 1.1 µg/mL) (Boonsri et al. ref. 2006). Caloxanthone A (199) from C. inophyllum was active against S. aureus (Yimdjo et al. ref. 2004).
Figure 18. Prenylated xanthones.
Thai researchers have identified in plants of the Garcinia L. genus (Clusiaceae) a plethora of antibacterial xanthones that have often in common dimethylallyl groups at position 1 and or 7. This is the case for 12b-hydroxy-des-d-garcigerrin A (200) from Garcinia dulcis (Roxb.) Kurz bacteriostatic for MRSA (MIC/MBC: 4/>200 µg/mL) (Thepthong et al. ref. 2017). Garcinia mangostana L. produces α-mangostin (201) (Nguyen and Marquis ref. 2011) active against VRE (MIC: 3.1 µg/mL) (Sakagami et al. ref. 2005), B. subtilis (MIC: 0.5 µg/mL) (Auranwiwat et al. ref. 2014), S. typhimurium (Yahayu et al. ref. 2013), and P. acnes (MIC: 0.7 µg/mL) (Al-Massarani et al. ref. 2013; Ahmad et al. ref. 2019). α-Mangostin (201) and β-mangostin (202) inhibited M. tuberculosis (MIC: 6.2 µg/mL) as well as garcinone B (203) (MIC: 12.7 µg/mL) (Suksamrarn et al. ref. 2003). β-Mangostin (202) was active against B. cereus with a MIC value as low as 0.2 µg/mL (Auranwiwat et al. ref. 2014). γ-Mangostin (204) inhibited the growth of MRSA and VRE with MIC values of 3.1 and 6.2 µg/mL, respectively (Dharmaratne et al. ref. 2013). Garcinone C (205) was antileptospiral (Seesom et al. ref. 2013). We can also mention garcicowanone A (206), rubraxanthone (207), fuscaxanthone A (208), 9-hydroxycalabaxanthone (198), and garcinianone A (209) from G. cowa, active against B. subtilis with the MIC values of 0.2, 1, 8, 4, and 4 µg/mL, respectively (Trisuwan and Ritthiwigrom ref. 2012; Auranwiwat et al. ref. 2014). From this plant, cowanol (210), cowanin (211), and garciniacowone (212) inhibited S. aureus with MIC values of 2, 4, and 2 µg/mL, respectively. Cowanol (210) was effective against E. coli (MIC: 8 µg/mL) (Siridechakorn et al. ref. 2012) and yielded a MIC value of 2 µg/mL against MRSA (Siridechakorn et al. ref. 2012; Trisuwan and Ritthiwigrom ref. 2012). Other examples are nigrolineaxanthone F (213), brasilixanthone (214) (MRSA, MIC: 2 µg/mL) (Rukachaisirikul, Tadpetch, et al. ref. 2005), 3-hydroxyblancoxanthone (215) (B. cereus, MIC: 4 µg/mL), nigrolineaxanthone Q (216) (Micrococcus luteus, MIC: 8 µg/mL) (Raksat et al. ref. 2019), 8-desoxygartanin (217) (S. aureus, MIC: 16 µg/mL), ananixanthone (218) (S. aureus, MIC: 32 µg/mL), and nigrolineaxanthone N (219) (MRSA, MIC: 4 µg/mL) from Garcinia nigrolineata Planch. ex T. Anderson (Rukachaisirikul et al. ref. 2003). Garcinia scortechinii King produces nigrolineaxanthone G (220) and 6-deoxyjacareubin (221) (MRSA, MIC: 4 µg/mL) (Rukachaisirikul, Tadpetch, et al. ref. 2005).
The internal cyclization of dimethylallyl groups forms caged xanthones, such as moreollic acid (222) from Garcinia hanburyi Hook.f. (MRSA, MIC: 25 µg/mL), as well as morellic acid (223) (Sukpondma et al. ref. 2005) and gambogic acid (224) bactericidal for MRSA (USA300) with the MIC/MBC of 12.5/25 and 25/50 µg/mL, respectively (Chaiyakunvat et al. ref. 2016). From G. scortechinii, scortechinone B (225), C (226), and F (227) were active against S. aureus with the MIC of 2, 8, and 4 µg/mL, respectively (Rukachaisirikul, Phainuphong, et al. ref. 2005).
Non-prenylated and hydroxylated xanthones have milder activities as in 1,5,6-trihydroxyxanthone (228) from Garcinia succifolia Kurz (S. aureus, MIC: 64 µg/mL) (Duangsrisai et al. ref. 2014). In the family Moraceae, gerontoxanthone H (229) from Cudrania cochinchinensis (Lour.) Kudô & Masam. was active against B. cereus with the MIC of 1.5 μg/mL (Fukai et al. ref. 2004).
Chromanes and chromenes
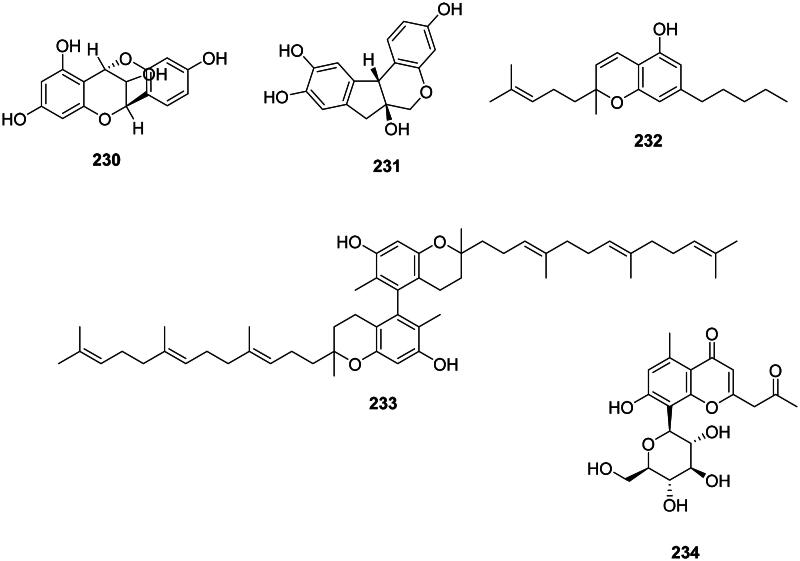
Plants in the fabids and malvids produce antibacterial chromanes and chromenes (Figure 19). For instance, cyanomaclurin (230) from Artocarpus heterophyllus Lam. (Moraceae) was bacteriostatic for S. mutans (Septama and Panichayupakaranant ref. 2015). Brasilin (231) from Caesalpinia sappan L. (Fabaceae) used in traditional Chinese medicine was active against MRSA, VRE, and MDR-Burkholderia cepacia (Xu and Lee ref. 2004) as well as S. pyogenes (MIC: 4 µg/mL) (Yin et al. ref. 2004). C. sativa produces cannabichromene (232) (S. aureus, MIC: 2 µg/mL) (Appendino et al. ref. 2008; Radwan et al. ref. 2009). The dimeric prenylated chromane garciniacowol (233) from G. cowa inhibited MRSA with the MIC of 2 µg/mL (Siridechakorn et al. ref. 2012). The chromane glycoside aloesin (234) in the genus Rumex L. (Polygonaceae) was active against M. tuberculosis (MIC: 2.8 µM) (Liang et al. ref. 2010).
Naphthalenols
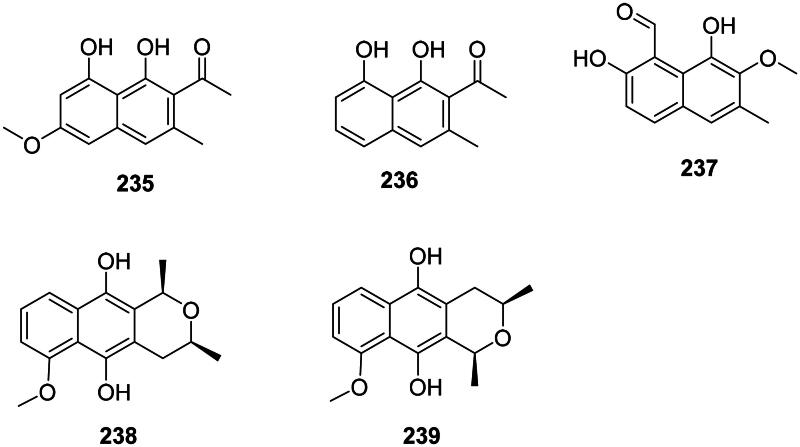
They are found in the malvids. Examples are torachrysone (235) in Rumex japonicus Houtt. (Polygonaceae) active against MRSA (MIC: 32 µg/mL) (Hatano et al. ref. 1999) and M. tuberculosis (Nishina et al. ref. 1993; Liang et al. ref. 2010), nepodin (236) from Rumex aquaticus L. (Polygonaceae) (Orbán-Gyapai ref. 2017), and hibicuslide C (237) from Hibiscus taiwanensis S.Y. Hu (Malvaceae) (MDR-P. aeruginosa) (Lee, Choi, et al. ref. 2016). In the monocots, Eleutherine bulbosa (Mill.) Urb. (Iridaceae) produces eleubosas A (238) and B (239) (E. coli, MIC: 12.5 µg/mL) (Jiang et al. ref. 2020) (Figure 20).
Phenanthrenes
Internal cyclization of stilbenes forms antibacterial phenanthrenes and biphenanthrenes in monocots (Chapatwala et al. ref. 1981) (Figure 21). Examples are 2,7-dihydroxy-4-methoxyphenanthrene (240) from Dioscorea bulbifera L. (Dioscoreaceae) (Kuete et al. ref. 2012) and blestriacin (241) from B. striata (MRSA, MIC: 2 μg/mL, bactericidal) (Chen et al. ref. 2018). Bletilla striata produces 4,7,7′-trimethoxy-9′,10′-dihydro(1,3′-biphenanthrene)-2,2′,5′-triol (242) (S. aureus, MIC: 8 µg/mL) as well as 4,8,4′,8′-tetramethoxy(1,1′-biphenanthrene)-2,7,2′,7′-tetrol (243) (S. aureus, bactericidal) (4,8,4′,8′-TBT) (Qian et al. ref. 2015). Arundina graminifolia (D. Don) Hochr (Orchidaceae) yields blestiarene A (244) and densiflorol B (245) bacteriostatic for S. aureus and E. coli (Zhang et al. ref. 2022).
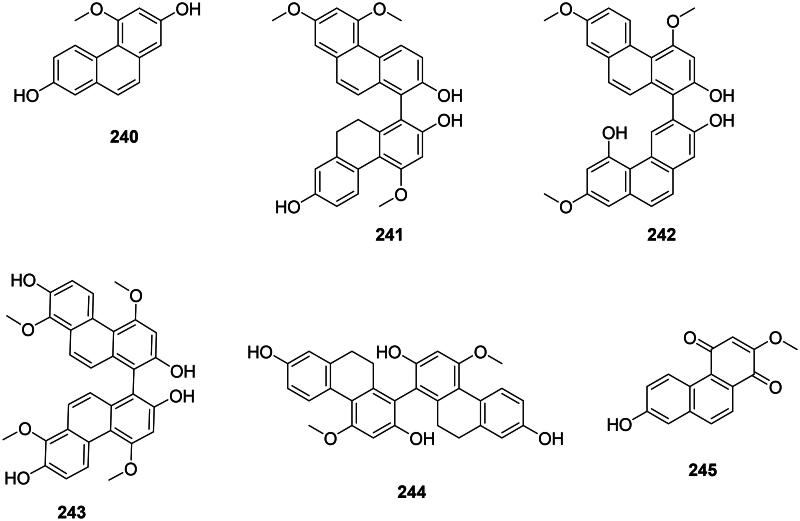
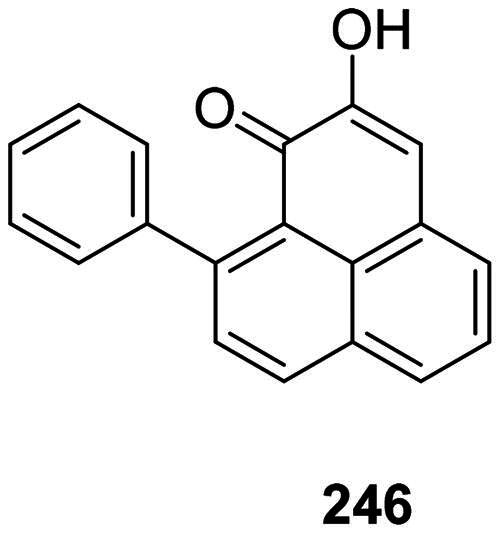
Phenylphenalenones
Diarylheptanoids serve in the monocots as precursors for the synthesis of phenylphenalenones phytoalexins, such as anigorufone (246) from Macropidia fuliginosa (Hook.) Druce (Haemodoraceae) (Brkljaca et al. ref. 2019) (Figure 22). Other antibacterial phenylphenalenones are found in the genus Musa L. (Musaceae) (Krishnamurthy et al. ref. 2023).
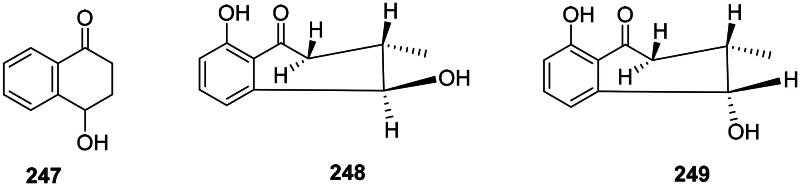
Tetralones
Examples are 4-hydroxy-1-tetralone (247) from E. roxburghiana (M. tuberculosis, MIC: 4 µg/mL) (Lin et al. ref. 2005; Wu et al. ref. 2012), l-epineo-isoshinanolone (248) (E. coli, MIC: 12.5 µg/mL), and neoiso-shinanolone (249) from Plumbago zeylanica L. (Plumbaginaceae, malvids) (Jetty et al. ref. 2010) (Figure 23).
Other non-flavonoids
They mostly occur in fabids (Figure 24). Examples are licocoumarone (250) (S. aureus, MIC: 6.2 µg/mL) from G. glabra (Demizu et al. ref. 1988), gancaonin I (251) (MRSA, MIC: 1.5 µg/mL) from Glycyrrhiza uralensis Fisch. ex DC. (Fabaceae) (Fukai et al. ref. 2002), and albanol B (252) from Morus alba L. (Moraceae) (S. typhimurium, MIC: 5 µg/mL) (Park et al. ref. 2003, Sohn et al. ref. 2004).
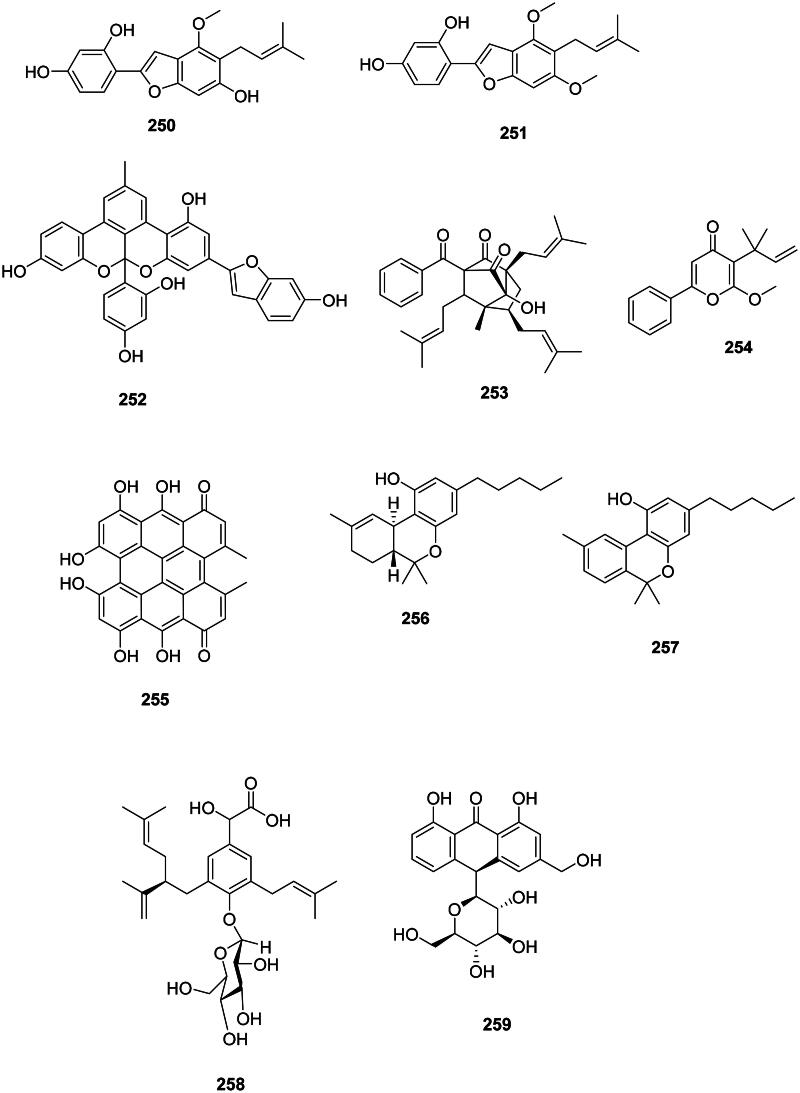
In the family Hypericaceae, examples are hypatulin A (253) from Hypericum patulum Thunb. (B. subtilis, MIC: 16 μg/mL) (Tanaka et al. ref. 2016), hyperenone A (254) from H. acmosepalum (S. aureus expressing NorA, MIC: 2 µg/mL) (Osman et al. ref. 2012), and hypericin (255) from Hypericum perforatum L. (Feyzioğlu et al. ref. 2013).
Δ9-Tetrahydrocannabinol (256) and cannabinol (257) from C. sativa are formed via the polyketide pathway (Abe ref. 2020) and were very strongly active against S. aureus (MIC: 1 µg/mL) (Appendino et al. ref. 2008; Radwan et al. ref. 2009). We can also cite harpulliaside A (258) from Harpullia pendula Planch. ex F. Muell. (Sapindaceae) (V. parahaemolyticus, MIC: 35 µg/mL) (Abdelkader et al. ref. 2016) as well as aloin A (259) from A. vera (Coopoosamy and Magwa ref. 2006b).
Flavonoids
Chalcones
The addition of one hydroxycinnamic acid unit with three malonyl-CoA units forms chalcones (Abe et al. ref. 2006; Wang et al. ref. 2023) (Figure 25). These 1,3-diphenyl-2-propen-1-ones occur in the fabids and malvids and are active against Gram-positive bacteria and mycobacteria. Examples are 2′,4′-dihydroxychalcone (260) from Muntingia calabura L. (Mutingiaceae, malvids) bactericidal for S. aureus (MIC/MBC: 50/100 µg/mL) (Sufian et al. ref. 2013) and butein (261) from Butea monosperma (Lam.) Taub. (Fabaceae) (M. tuberculosis, MIC: 12.5 µg/mL) (Chokchaisiri et al. ref. 2009).
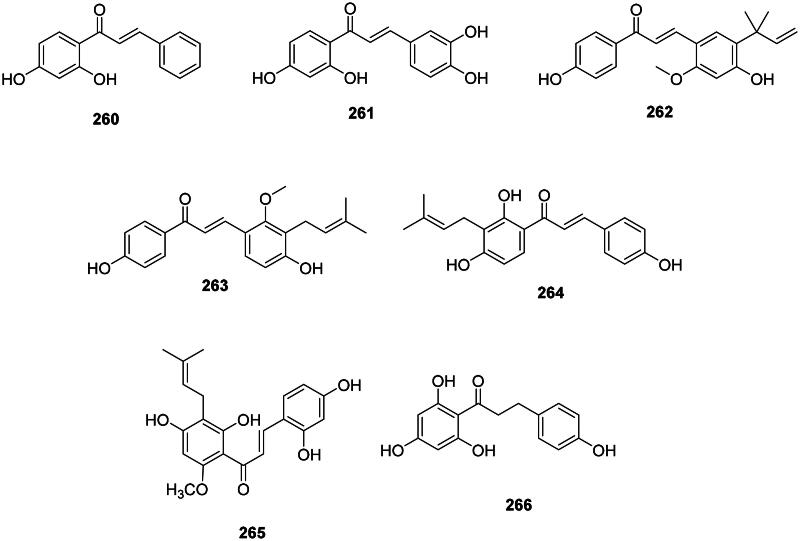
The prenylation of chalcones in the family Fabaceae enhances their antibacterial activity as in licochalcone A (262) from Glycyrrhiza inflata Batalin with S. aureus (MIC: 3 µg/mL) (Tsukiyama et al. ref. 2002), MRSA (MIC: 6.2 µg/mL), and P. gingivalis (MIC: 10 µg/mL) (Fukai et al. ref. 2002). Other examples are licochalcone C (263) (M. luteus, MIC: 6.2 µg/mL) (Haraguchi et al. ref. 1998) and isobavachalcone (264) from P. corylifolia (Yin et al. ref. 2004). Sophora flavescens Aiton is used in traditional Chinese medicine and produces 7,9,2′,4′-tetrahydroxy-8-isopentenyl-5-methoxychalcone (7,9,2′,4′-TIMC) (265) which was effective against MRSA (MIC: 0.9 µg/mL) and VRE (MIC: 7.8 µg/mL) (Lee, Kim, et al. ref. 2010).
The condensation of one dihydrohydroxycinnamic acid unit with three malonyl-CoA units forms antibacterial dihydrochalcones (Ibdah et al. ref. 2017) such as phloretin (266) from Malus domestica (Suckow) Borkh. (Rosaceae, fabids) (S. aureus, MIC: 7.8 µg/mL) (Barreca et al. ref. 2014).
Flavanones
Simple flavanones
These antibacterial 2-phenyl-2,3-dihydro-4H-chromen-4-ones come from the cyclization of chalcones in malvids and fabids (Shah and Smith ref. 2020) (Figure 26). Examples are artocarpanone (267) from A. heterophyllus bactericidal for E. coli (MIC/MBC: 3.9/7.8 µg/mL) (Septama and Panichayupakaranant ref. 2017), pinocembrin (268) from G. glabra (M. tuberculosis, MIC: 3.3 µg/mL) (Fukui et al. ref. 1988; Chou et al. ref. 2011), and naringenin (269) in the genus Citrus L. (Rutaceae) (S. pyogenes, MIC: 50 µg/mL) (Macé et al. ref. 2017).
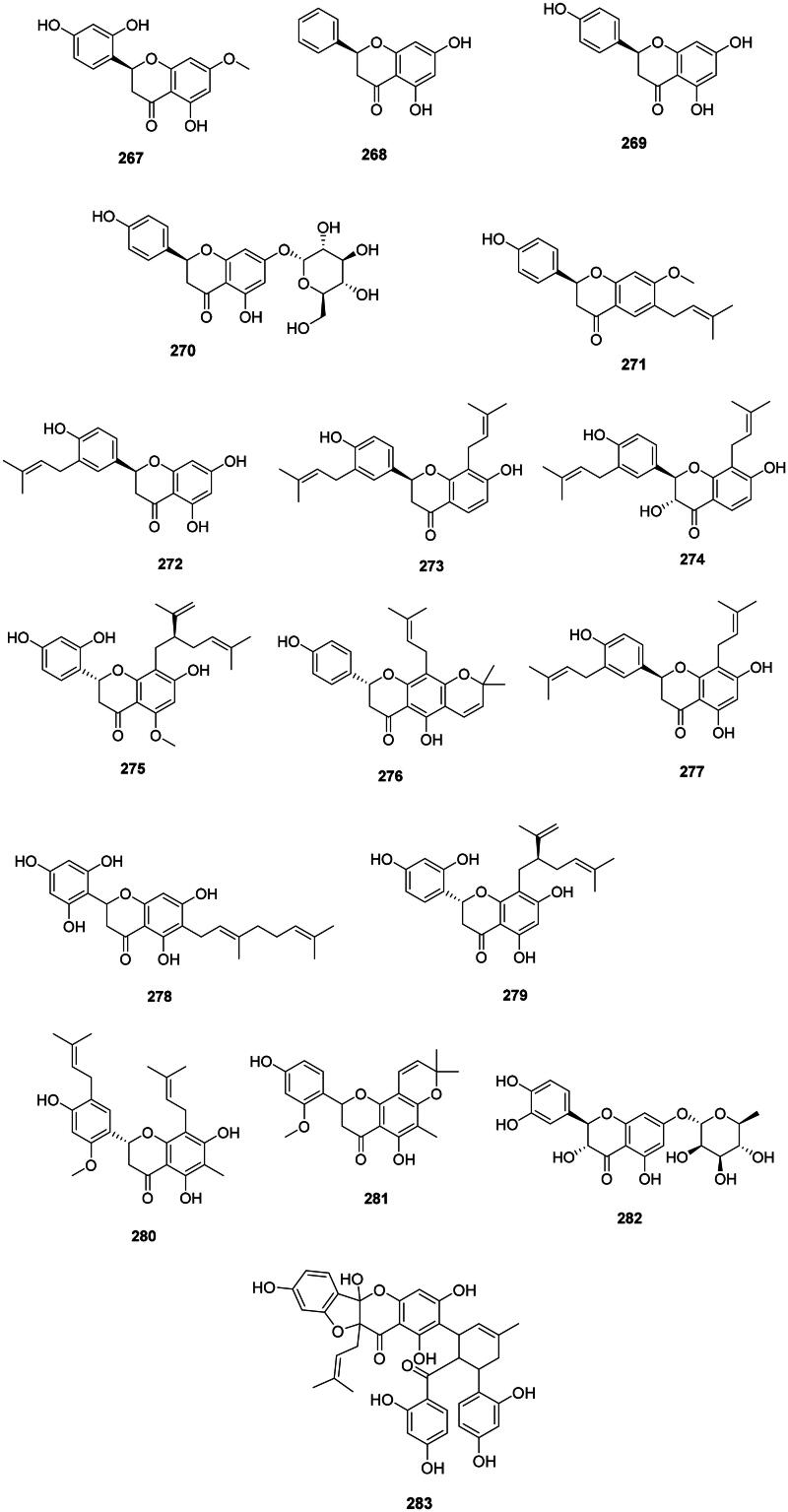
Glycosylation of naringenin (269) in position 8 forms prurin (270) in Acacia farnesiana Wall. (Fabaceae) active against MDR-M. tuberculosis (MIC: 50 µg/mL) and C. jejuni (MIC: 50 µg/mL) (Hernández-García et al. ref. 2019).
Prenylated flavanones
Plants of the Fabaceae family produce prenylated flavanones active against Gram-positive bacteria (Figure 26). Examples of flavanones with dimethylallyl moieties are bavachinin (271) from P. corylifolia (Yin et al. ref. 2004) and licoflavanone (272) in G. glabra (Fukui et al. ref. 1988). From this plant, glabrol (273) and 3-hydroxyglabrol (274) yielded the MIC values of 1.5 and 6.2 µg/mL against S. aureus, respectively (Mitscher et al. ref. 1980). Glabrol (273) was active against M. smegmatis (MIC: 1.5 µg/mL) (Mitscher et al. ref. 1980). Other instances are kurarinone (275) from S. flavescens (MRSA, MIC: 2 µg/mL) (Chen et al. ref. 2005), lupinifolin (276) from Derris reticulata Craib. (Mazimba et al. ref. 2012) (S. aureus, MIC: 8 µg/mL, bactericidal) (Yusook et al. ref. 2017), and euchrestaflavanone A (277) from Flemingia strobilifera (L.) W.T. Aiton (P. aeruginosa, MIC: 17 µg/mL) (Madan et al. ref. 2008).
An example of geranylated flavanone is sophoraflavanone D (278) from Echinosophora koreensis Nakai (Fabaceae) (E. coli MIC: 20 µg/mL) (Sohn et al. ref. 2004). A lavandulyl group enhances the antibacterial strength of flavanones as in sophoraflavanone G (279) from Sophora exigua Craib. (Fabaceae) with a MIC as low as 0.5 µg/mL against MRSA (Cha et al. ref. 2007, ref. 2009).
Plants of the Celastraceae family (fabids) produce antibacterial flavanones. Examples are (2S)-5,7,4′-trihydroxy-2′-methoxy-8,5′-di(3-methyl-2-butenyl)-6-methylflavanone (280) and (±)-5,4′-dihydroxy-2′-methoxy-6′,6″-dimethypyraro-(2″,3″:7,8)-6-methyflavanone (281) (MRSA, IC50: 2 µg/mL) from Tripterygium wilfordii Hook.f. used in traditional Chinese medicine (Chen et al. ref. 2017).
Flavanone-O-glycosides
Taxifolin-7-O-rhamnoside (282) from H. japonicum was bactericidal for MRSA (MIC/MBC: 32/64 µg/mL) (An et al. ref. 2011).
Miscellaneous
There are flavones with complex polyphenolic structures, such as sanggenon D (283) from Morus alba L. (Moraceae) active against S. epidermidis (MIC: 40 µg/mL) (Sohn et al. ref. 2004).
Isoflavans
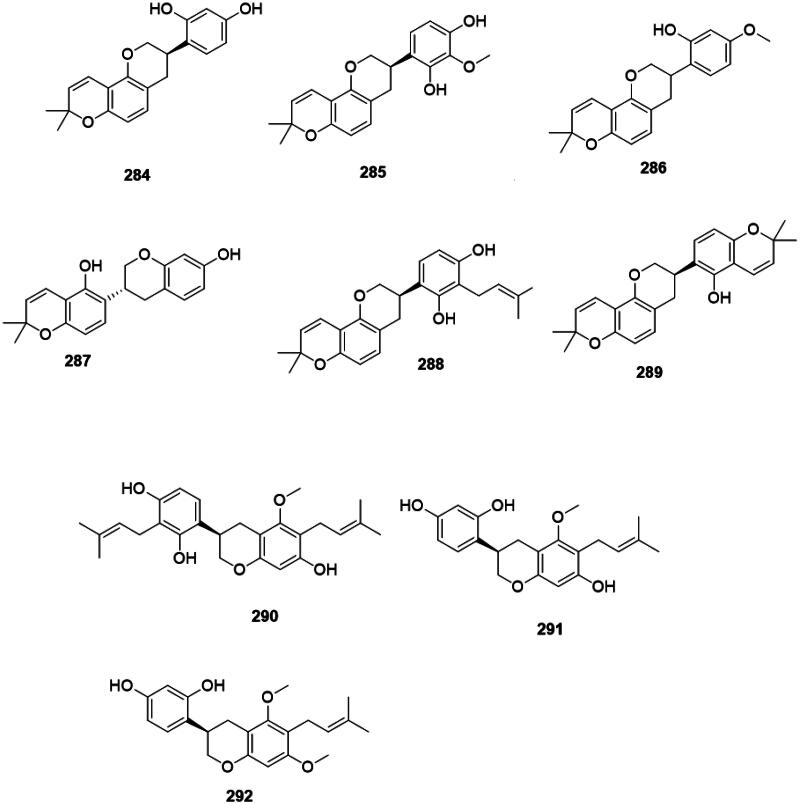
Plants of the Fabaceae produce antibacterial isoflavans (Pičmanová et al. ref. 2013) (Figure 27). Glabridin (284) from G. glabra inhibited the growth of S. aureus (MIC: 6.2 µg/mL) (Gupta et al. ref. 2008), MRSA (MIC: 12.5 µg/mL) (Fukai et al. ref. 2002), M. smegmatis (6.2 µg/mL) (Mitscher et al. ref. 1980), and P. gingivalis (MIC: 10 µg/mL) (Azelmat et al. ref. 2015). From G. glabra, other antibacterial isoflavans are 3′-methoxyglabridin (285), 4′-O-methylglabridin (286), phaseollinisoflavan (287), hispaglabridin A (288) (S. aureus, MIC: 3.1 µg/mL), and hispaglabridin B (289) (S. aureus, MIC: 6.2 µg/mL) (Mitscher et al. ref. 1980). Other examples include licoricidin (290) (MRSA, MIC: 3.1 µg/mL), glyasperin C (291) (E. faecium), and glyasperin D (292) (MRSA, MIC: 6.2 µg/mL) from G. uralensis (Fukai et al. ref. 2002; Gafner et al. ref. 2011; Eerdunbayaer et al. ref. 2014; Villinski et al. ref. 2014).
Isoflavanones
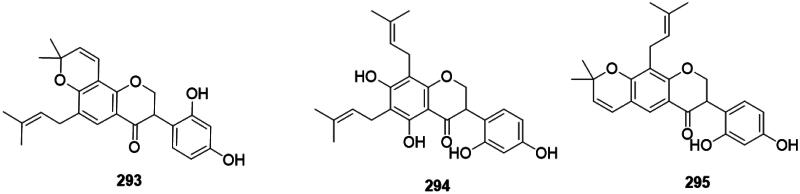
Plants of the Fabaceae family isomerize flavanones into isoflavanones active against Gram-positive bacteria (Pičmanová et al. ref. 2013). For example, Erythrina variegata L. (Fabaceae) produces orientanol F (293) (MRSA, MIC90: 12.5 µg/mL) (Tanaka et al. ref. 2002), orientanol E (294) (MRSA, MIC90/MBC90: 3.1/25 µg/mL, bacteriostatic) (Tanaka et al. ref. 2015), and bidwillon B (295) (MRSA, MIC: 3.1 µg/mL) (Sato et al. ref. 2003) (Figure 28).
Flavones
Simple flavones
The formation of a Δ2,3 double bond in flavanones forms flavones that generally have moderate antibacterial activities (Figure 29) (Zhao et al. ref. 2016). Flavones that have an unsubstituted C ring are common in upper Angiosperms. This is the case, for example, of chrysin (297) of Oroxylum indicum (L.) Kurz (Bignoniaceae, lamiids) (Ali et al. ref. 1998; Zhao et al. ref. 2022) and baicalein (298) (S. typhimurium, MIC: 64 μg/mL) in Scutellaria baicalensis Georgi (Lamiaceae, lamiids) used in traditional Chinese medicine (Yang et al. ref. 2000; Wu et al. ref. 2018). Another example is galangin (299) from Helichrysum aureonitens Sch. Bip. (Asteraceae, campanulids) which was active against 4-quinolone-resistant S. aureus (Cushnie and Lamb ref. 2006) and Mycobacterium phlei (MIC: 50 µg/mL) (Pomilio et al. ref. 1992).
Figure 29. Flavones.
Flavones with hydroxylated C rings are ubiquitous in Angiosperms and include notably apigenin (300) (K. pneumoniae, MIC: 25 µg/mL), luteolin (301) (P. aeruginosa, MIC: 25 µg/mL) (Sathiamoorthy et al. ref. 2007; Bustos et al. ref. 2018), kaempferol (302) (S. mutans, MIC: 32 µg/mL) (Yamada et al. ref. 1999), quercetin (303) (Salmonella enteridis, MIC: 15.6 µg/mL) (Phadungkit and Luanratana ref. 2006), robinetin (304) (Mori et al. ref. 1987), and myricetin (305) (Xu et al. ref. 2015).
Methoxylated flavones
Upper Angiosperms often produce antibacterial methoxylated flavones (Figure 29). We can cite for instance isorhamnetin (306) from A. elliptica (S. typhimurium, MIC: 15.6 µg/mL) (Phadungkit and Luanratana ref. 2006). Vitex negundo L. (Verbenaceae) produces penduletin (307) (MRSA, MIC: 10 µg/mL) (Sichaem et al. ref. 2021) and artemetin (308) (K. pneumoniae, MIC: 25 µg/mL) (Sathiamoorthy et al. ref. 2007). Other examples are found in the family Asteraceae (Murillo et al. ref. 2003).
Prenylated flavones
They are common in the family Moraceae and include papyriflavonol A (309) from Broussonetia papyrifera (L.) L’Hér. ex Vent. (S. typhimurium, MIC: 10 µg/mL), morusin (310) from Morus mongolica (Bureau) C.K. Schneid. (S. epidermidis, MIC: 20 µg/mL), kuwanon C (311) (S. typhimurium, MIC: 6.2 µg/mL) (Sohn et al. ref. 2004), kuwanon G (312) (S. mutans, MIC: 8 µg/mL) from M. alba (Park et al. ref. 2003), and artocarpin (313) from A. heterophyllus (S. mutans, MIC/MBC: 4.4/8.9 µM) (Septama and Panichayupakaranant ref. 2018) (Figure 29).
Flavone-O-glycosides
Examples are isocytisoside (314) from Aquilegia vulgaris L. (Ranunculaceae) (S. aureus, MIC: 15.6 µg/mL) (Bylka et al. ref. 2004), quercetin 7-O-glucoside (315) in Gossypium arboreum L. (Malvaceae) (Waage and Hedin ref. 1984) (Figure 29). Other illustrations are afzelin (316) and kaempferol-7-rhamnoside (317) from Bryophyllum pinnatum (Lam.) Oken (Crassulaceae, core eudicots) active against S. typhi with the MIC values of 2 and 1 µg/mL, respectively (Tatsimo et al. ref. 2012), luteoloside (318) from L. japonica (Xiong et al. ref. 2013), hyperoside (319) from H. perforatum (Pretorius et al. ref. 2003; Saçıcı and Yesilada ref. 2022), as well as taxifolin-7-O-α-l-rhamnopyranoside (320) (TLRP) (An et al. ref. 2011). Baicalin (321) from S. baicalensis inhibited the growth of S. typhimurium (MIC/MBC: 64/>128 μg/mL) (Wu et al. ref. 2018). Rutin (322) from Sophora japonica L. (Fabaceae) (Balbaa et al. ref. 1974) was active P. aeruginosa, A. baumannii, and S. aureus with the MIC values of 16, 8, and 4 µg/mL, respectively (Orhan et al. ref. 2010).
Flavone-C-glycosides
Apigenin-8-C-glucopyranoside (vitexin) (323) (Figure 29) from V. negundo inhibited the growth of Mycobacterium fortuitum (MIC: 30 µg/mL) (Aderogba et al. ref. 2019). It should be noted that vitexin (323), although weakly active in vitro against S. aureus, was nevertheless active in vivo against S. aureus with an additional inflammatory action (Das et al. ref. 2022).
Isoflavones
The isomerization and dehydrogenation of flavanones form isoflavones in the family Fabaceae (Artigot et al. ref. 2013) (Figure 30). These phytoalexins are often active against Gram-positive bacteria and include for instance biochanin A (324) from Cassia fistula L. (Sartorelli et al. ref. 2009) (S. pyogenes, MIC: 32 µg/mL) (Pohjala et al. ref. 2012; Hummelova et al. ref. 2015), santal (325) from Derris scandens (Roxb.) Benth. (MRSA, MIC: 2 µg/mL) (Mahabusarakam et al. ref. 2004), and formononetin (326) from Glycyrrhiza pallidiflora Maxim. (Kajiyama et al. ref. 1993; Mutai et al. ref. 2015). In the family Iridaceae (monocots), tectorigenin (327) from Belamcanda chinensis (L.) Redouté was active against S. aureus (MIC: 50 µg/mL) (Oh et al. ref. 2001) and MRSA (MIC: 125 µg/mL) (Joung et al. ref. 2014).
Figure 30. Isoflavones.
Prenylated isoflavones
They are prevalent in the Fabaceae family and are active against Gram-positive bacteria (Figure 30). Examples are licoisoflavone A (328) and B (329) from G. uralensis (MRSA, MIC: 32 µg/mL) (Wu et al. ref. 2019; Chen et al. ref. 2023), derrisisoflavone A (330) from D. scandens (MRSA, MIC: 4 µg/mL) (Mahabusarakam et al. ref. 2004), lupalbigenin (331) from G. dulcis (S. aureus, MIC: 4 µg/mL) (Deachathai et al. ref. 2005), 6,8-diisoprenyl-5,7,4′-trihydroxyisoflavone (332) (S. mutans, MIC: 2 µg/mL) (He et al. ref. 2006), 8-(γ,γ-dimethylallyl)-wighteone (333) from G. uralensis (MRSA, MIC: 8 µg/mL) (Hatano et al. ref. 2000; Eerdunbayaer et al. ref. 2014), and auriculasin (334) from Flemingia philippinensis Merr. & Rolfe with MDR-E. coli (MIC: 2 µg/mL) (Mohamed et al. ref. 2022).
Isoflavone glycosides
Genistin (335) from F. strobilifera inhibited the growth of S. epidermidis, S. aureus, MRSA, P. aeruginosa, and E. coli with the MIC of 31.2, 62.5, 34, 125, and 146 µg/mL, respectively (Madan et al. ref. 2008; Boutaghane et al. ref. 2019) (Figure 30).
Pterocarpans
The reduction and cyclization of isoflavones form pterocarpans in the family Fabaceae. These phytoalexins are strongly active against Gram-positive bacteria (Figure 30). Examples are glycinol (336) from Glycine max (L.) Merr. (Weinstein and Albersheim ref. 1983), orientanol C (337) from E. variegata (MRSA, MIC90:12.5 µg/mL), as well as orientanol B (338) (MIC: 3.1 µg/mL) (Tanaka et al. ref. 2002). From this plant, erycristagallin (339) was active against MRSA (MIC90: 6.2 µg/mL) (Tanaka et al. ref. 2002) and Actinomyces viscosum (MIC: 1.5 µg/mL) (Sato et al. ref. 2003). Glycyrrhizol A (340) and B (341) from G. uralensis inhibited S. mutans with the MIC values of 1 and 32 µg/mL, respectively (He et al. ref. 2006). Erybraedin A (342) from E. zeyheri was bacterostatic for VRE (MIC: 1.5 µg/mL) (Sato et al. ref. 2004).
Flavans
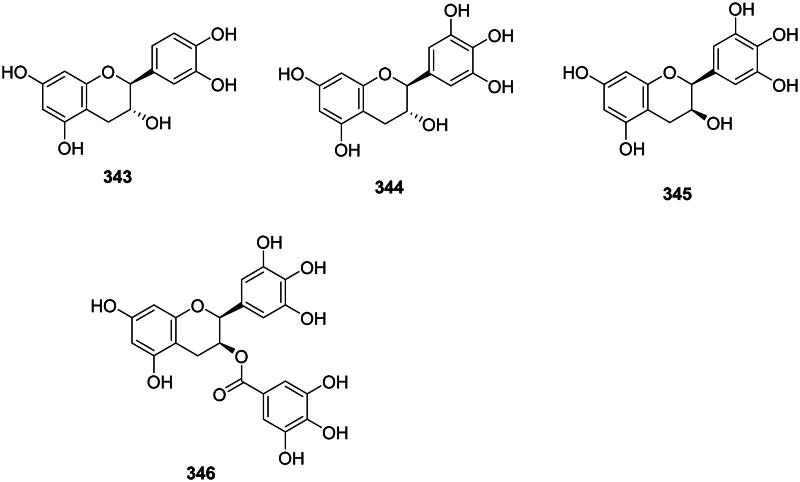
The reduction of flavanones forms antibacterial flavans (Cao et al. ref. 2020) (Figure 31). Examples are catechin (343) gallocatechin (344) (Pretorius et al. ref. 2003), and epigallocatechin (345) (M. smegmatis, MIC: 7.8 µg/mL) (Mativandlela et al. ref. 2007). (−)-Epigallocatechin 3-O-gallate (EGCG) (346) from Camellia sinensis (L.) Kuntze (Theaceae, asterids) inhibited the growth of C. jejuni (MIC: 8 μg/mL), N. gonorrhoea (MIC: 32 μg/mL), S. pneumoniae (MIC: 32 μg/mL) (Matsumoto et al. ref. 2012), MDR-A. baumannii (MIC: 78 μg/mL, bactericidal) (Osterburg et al. ref. 2009), and S. maltophilia (Navarro-Martínez et al. ref. 2005; Gordon and Wareham ref. 2010).
Antibacterial strength and spectrum of activity
Over the last decades, several MIC threshold values have been proposed to identify natural plant products with very strong antibacterial activity (Fabry et al. ref. 1998; Ríos and Recio ref. 2005), the latest being a MIC ≤ 4 µg/mL (Tankeo and Kuete ref. 2023). Since phenolic compounds often have low therapeutic indices and limited oral bioavailabilities (Serrano et al. ref. 2009; Velderrain-Rodríguez et al. ref. 2014) we recommend reducing this threshold value below 2 µg/mL.
Thus, out of ∼350 antibacterial phenolic compounds identified from 1945 to 2023 in Angiosperms from Asia and the Pacific, 44 are very strongly active (MIC < 2 µg/mL) (Table 1). To the extent that resistance thresholds must be considered for antibiotics, a MIC of <1 µg/mL could be used as a threshold for the possible clinical development of natural products. Plumbagin (127), isodiospyrin (137), malabaricone A (164), malabaricone B (165), anacardic acid (170), cannabidiol (174), bakuchiol (176), hypercalin B (181), lupulone (183), rhodomyrtone (186), cowanone (191), α-mangostin (201), β-mangostin (202), garcicowanone A (206), 7,9,2′,4′-tetrahydroxy-8-isopentenyl-5-methoxychalcone (265), and sophoraflavanone G (279) have a MIC < 1 µg/mL against Gram-positive bacteria. A MIC of <1 µg/mL was obtained with 1′-acetoxychavicol acetate (21), juglone (124), 3-methoxyjuglone (125), plumbagin (127), and 2-methoxy-7-methyljuglone (128) against mycobacteria. Dendrocoumarin (53) and cannabidiol (174) have a MIC < 1 µg/mL against Gram-negative bacteria.
Table 1.: Phenolic compounds with very strong antibacterial activity (MIC < 2 µg/mL).
| MM | LogD | PSA | FRB | Planar | G | HA | HD | S | MIC < 1 µg/mL | BS | BC | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Non-flavonoids | ||||||||||||
| Hydroxycinnamic acid derivatives | ||||||||||||
| 2-Methoxy-2-butenolide-3-cinnamate (15) | 260 | – | – | – | – | – | Mb | |||||
| Phenylpropanoids | ||||||||||||
| 1′-Acetoxychavicol acetate (21) | 234.2 | 2.3 | 53 | 6 | 4 | 0 | Mb | √ | ||||
| Coumarins | ||||||||||||
| Dendrocoumarin (53) | 244 | – | – | – | √ | – | – | G− | √ | |||
| Itolide A (54) | 244 | – | – | – | √ | – | – | G−/G+ | ||||
| Stilbenes | ||||||||||||
| Cajanin stilbene acid (62) | 338.4 | 5.8 | 67 | 6 | 2 | 4 | G+ | |||||
| 1,4-Naphthoquinones | ||||||||||||
| Juglone (124) | 174.1 | 0.9 | 54 | 0 | 1 | 3 | G+ | √ | ||||
| Mb | √ | |||||||||||
| 3-Methoxyjuglone (125) | – | – | – | – | √ | – | – | Mb | √ | |||
| Plumbagin (127) | 188.1 | 1.4 | 54 | 0 | √ | 3 | 1 | G+/Mb | √ | |||
| 2-Methoxy-7-methyl juglone (128) | 218.2 | – | 64 | 1 | √ | 4 | 1 | Mb | √ | |||
| 7-Methyljuglone (129) | 188.1 | 1.3 | 54 | 0 | √ | 3 | 1 | Mb | √ | |||
| Diospyrin (136) | 372 | 0.5 | 109 | 1 | √ | 6 | 2 | Mb | √ | |||
| Isodiospyrin (137) | 374 | 0.5 | 109 | 1 | √ | 6 | 2 | G+ | √ | |||
| Anthraquinones | ||||||||||||
| Aloe-emodin (143) | 270.2 | 1.2 | 95 | 1 | √ | 5 | 3 | G−/G+ | ||||
| Rhein (144) | 284.2 | −0.5 | 112 | 1 | √ | 6 | 3 | G− | ||||
| Emodin (145) | 270.2 | 1.7 | 0 | √ | 5 | 3 | G+ | |||||
| Long chain alkyl phenols | ||||||||||||
| Malabaricone A (164) | 326.4 | 5.6 | 58 | 10 | √ | 3 | 2 | G+ | √ | √ | ||
| Malabaricone B (165) | 328.4 | 4.5 | 78 | 10 | √ | 4 | 3 | G+ | √ | √ | ||
| Anacardic acid (170) | 348.5 | 6.4 | 58 | 10 | √ | 5 | 4 | G+ | √ | |||
| Cannabidiol (174) | 324.2 | 6.4 | 40 | 6 | √ | 2 | 2 | G+/G− | √ | |||
| Cannabigerol (175) | 316.4 | 6.7 | 40 | 9 | √ | 2 | 2 | G+ | ||||
| Bakuchiol (176) | 256.3 | 5.6 | 20 | 6 | √ | 1 | 1 | G+ | √ | |||
| Prenylated phloroglucinols | ||||||||||||
| Hypercalin B (181) | 518.7 | – | 94.8 | 8 | √ | 5 | 3 | G+ | √ | |||
| Lupulone (183) | 414.5 | 3.8 | 75 | 9 | √ | 4 | 4 | G+ | √ | |||
| Rhodomyrtone (186) | 442.5 | 5.3 | 101 | 5 | √ | √ | 2 | 6 | G+ | √ | ||
| Isomyrtucommulone B (187) | 415.2 | – | – | – | √ | √ | – | – | G+ | |||
| Myrciarone B (188) | 429.2 | – | – | – | √ | √ | – | – | G+ | |||
| Prenylated benzophenones | ||||||||||||
| Cowanone (191), | – | – | – | – | √ | – | – | G+ | √ | |||
| Prenylated xanthones | ||||||||||||
| Gerontoxanthone I (197) | 396.4 | 3.8 | 107 | 4 | √ | √ | 6 | 4 | G− | |||
| 9-Hydroxycalabaxanthone (198) | 426.5 | 4.4 | 85 | 3 | √ | √ | 6 | 2 | G− | |||
| α-Mangostin (201) | 410.4 | 4.2 | 96 | 5 | √ | √ | 6 | 3 | G+ | √ | ||
| β-Mangostin (202) | 424.4 | 4.6 | 85 | 6 | √ | √ | 6 | 2 | G+ | √ | ||
| Garcicowanone A (206) | 356.4 | 4.7 | 85.2 | 4 | √ | √ | 6 | 2 | G+ | √ | ||
| Rubraxanthone (207) | 410.1 | 4.1 | 96 | 6 | √ | √ | 6 | 3 | G+ | |||
| Gerontoxanthone H (229) | 380.4 | 4.6 | 87 | 4 | √ | √ | 5 | 3 | G+ | |||
| Other non-flavonoids | ||||||||||||
| Gancaonin I (251) | 354.3 | 4.1 | 72 | 5 | 5 | 2 | G+ | |||||
| Δ9-tetrahydrocannabinol (256) | 314.4 | 7.2 | 29 | 4 | √ | √ | 2 | 1 | G+ | |||
| Cannabinol (257) | 310.2 | 6.7 | 29 | 4 | √ | √ | 2 | 1 | G+ | |||
| Flavonoids | ||||||||||||
| Chalcones | ||||||||||||
| 7,9,2′,4′-TIMC (265) | – | – | – | – | √ | – | – | G+ | √ | |||
| Flavanones | ||||||||||||
| Glabrol | (273) | 392.4 | 5.7 | 67 | 5 | √ | 4 | 2 | G+/Mb | |||
| Sophoraflavanone G (279) | 424.4 | 5.3 | 107 | 6 | √ | 6 | 4 | G+ | √ | |||
| Flavones | ||||||||||||
| Kaempferol-7-rhamnoside (317) | 432.3 | −0.3 | 166 | 3 | 10 | 6 | G− | |||||
| Isoflavones | ||||||||||||
| Erycristagallin (339) | 390.4 | 6.3 | 63 | 4 | √ | √ | 4 | 2 | G+ | |||
| Glycyrrhizol A (340) | 420.1 | 6.3 | 72 | 5 | √ | √ | 5 | 5 | G+ | |||
| Erybraedin A (342) | 392.4 | 6 | 59 | 4 | √ | √ | 4 | 2 | G+ | |||
MM: molecular mass (g/mol); LogD: at pH 7.4; PSA: polar surface area (Ų); G: one or more long prenyl or long-chain alkyl group present; FRB: freely rotating bond; H: hydrogen bond acceptor; HD: hydrogen bond donor; S: spectrum; G+: Gram-positive; Mb: mycobacteria; BS: bacteriostatic; BC: bactericidal; –: not available.
Distribution of phenolic compounds with very strong antibacterial activity
Among those above 44 phenolic compounds, 33 originate from core Angiosperms, including 25 from fabids (Table 2). In the fabids, eight phenolic compounds come from the Fabaceae family, five from the Clusiaceae family, and three from the Hypericaceae family. The other major clades that produce such compounds are the core Angiosperms are the malvids and that fabids (which are sister groups) and the Asterids Fabids and Malvids produce prenylated phenolic compounds. Asterids produce naphthoquinones. No phenolic compounds with very strong antibacterial activity were identified among protomagnoliids, eudicots, rosids, lamiids, and campanulids.
Table 2.: Distribution of phenolics compound with very strong antibacterial activity (MIC < 2 µg/mL).
| Group | Clade | Order | Family | |
|---|---|---|---|---|
| Non-flavonoids | ||||
| Hydroxycinnamic acid derivatives | ||||
| 2-Methoxy-2-butenolide-3-cinnamate (15) | Core angiosperms | Malvids | Caryophyllales | Polygonaceae |
| Phenylpropanoids | ||||
| 1′-Acetoxychavicol acetate (21) | Basal angiosperms | Monocots | Zingiberales | Zingiberaceae |
| Coumarins | ||||
| Dendrocoumarin (53) | Basal angiosperms | Monocots | Asparagales | Orchidaceae |
| Itolide A (54) | Basal angiosperms | Monocots | Asparagales | Orchidaceae |
| Stilbenes | ||||
| Cajanin stilbene acid (62) | Core angiosperms | Fabids | Fabales | Fabaceae |
| 1,4-Napthoquinones | ||||
| Juglone (124) | Core angiosperms | Fabids | Fagales | Juglandaceae |
| 3-Methoxyjuglone (125) | Core angiosperms | Fabids | Fagales | Juglandaceae |
| Plumbagin (127) | Upper angiosperms | Asterids | Ericales | Ebenaceae |
| 2-Methoxy-7-methyl juglone (128) | Upper angiosperms | Asterids | Ericales | Ebenaceae |
| 7-Methyljuglone (129) | Upper angiosperms | Asterids | Ericales | Ebenaceae |
| Diospyrin (136) | Upper angiosperms | Asterids | Ericales | Ebenaceae |
| Isodiospyrin (137) | Upper angiosperms | Asterids | Ericales | Ebenaceae |
| Anthraquinones | ||||
| Aloe-emodin (143) | Core angiosperms | Malvids | Caryophyllales | Polygonaceae |
| Rhein (144) | Core angiosperms | Malvids | Caryophyllales | Polygonaceae |
| Emodin (145) | Core angiosperms | Malvids | Caryophyllales | Polygonaceae |
| Long chain alkyl phenols | ||||
| Malabaricone A (164) | Basal angiosperms | Magnoliids | Laurales | Myristicaceae |
| Malabaricone B (165) | Basal angiosperms | Magnoliids | Laurales | Myristicaceae |
| Anacardic acid (170) | Core angiosperms | Fabids | Sapindales | Anacardiaceae |
| Cannabidiol (174) | Core angiosperms | Fabids | Rosales | Cannabaceae |
| Cannabigerol (175) | Core angiosperms | Fabids | Rosales | Cannabaceae |
| Bakuchiol (176) | Core angiosperms | Malvids | Caryophyllales | Amaranthaceae |
| Prenylated phloroglucinols | ||||
| Hypercalin B (181) | Core angiosperms | Fabids | Malpighiales | Hypericaceae |
| Lupulone (183) | Core angiosperms | Fabids | Rosales | Cannabaceae |
| Rhodomyrtone (186) | Core angiosperms | Malvids | Myrtales | Myrtaceae |
| Isomyrtucommulone B (187) | Core angiosperms | Malvids | Myrtales | Myrtaceae |
| Myrciarone B (188) | Core angiosperms | Malvids | Myrtales | Myrtaceae |
| Prenylated benzophenones | ||||
| Cowanone (191) | Core angiosperms | Fabids | Malpighiales | Clusiaceae |
| Prenylated xanthones | ||||
| Gerontoxanthone I (197) | Core angiosperms | Fabids | Malpighiales | Hypericaceae |
| 9-Hydroxycalabaxanthone (198) | Core angiosperms | Fabids | Malpighiales | Hypericaceae |
| α-Mangostin (199) | Core angiosperms | Fabids | Malpighiales | Clusiaceae |
| β-Mangostin (201) | Core angiosperms | Fabids | Malpighiales | Clusiaceae |
| Garcicowanone A (206) | Core angiosperms | Fabids | Malpighiales | Clusiaceae |
| Rubraxanthone (207) | Core angiosperms | Fabids | Malpighiales | Clusiaceae |
| Gerontoxanthone H (229) | Core angiosperms | Fabids | Rosales | Moraceae |
| Others non-flavonoids | ||||
| Gancaonin I (251) | Core angiosperms | Fabids | Fabales | Fabaceae |
| Δ9-Tetrahydrocannabinol (256) | Core angiosperms | Fabids | Rosales | Cannabaceae |
| Cannabinol (257) | Core angiosperms | Fabids | Rosales | Cannabaceae |
| Flavonoids | ||||
| Chalcones | ||||
| 7,9,2′,4′-TIMC (265) | Core angiosperms | Fabids | Fabales | Fabaceae |
| Flavanones | ||||
| Glabrol (274) | Core angiosperms | Fabids | Fabales | Fabaceae |
| Sophoraflavanone G (280) | Core angiosperms | Fabids | Fabales | Fabaceae |
| Flavones | ||||
| Kaempferol-7-rhamnoside (318) | Core angiosperms | Core eudicots | Saxifragales | Crassulaceae |
| Isoflavones | ||||
| Erycristagallin (340) | Core angiosperms | Fabids | Fabales | Fabaceae |
| Glycyrrhizol A (341) | Core angiosperms | Fabids | Fabales | Fabaceae |
| Erybraedin A (342) | Core angiosperms | Fabids | Fabales | Fabaceae |
Influence of molecular mass on the antibacterial strength and spectrum of activity
The molecular mass of phenolic compounds can be tentatively classified as follows: <200 g/mol = low, 200–400 g/mol = moderate, and >400 g/mol = high. Consequently, phenolic compounds with very strong activity against Gram-positive bacteria, Gram-negative bacteria, or mycobacteria have on average a low, moderate, or high molecular mass (Table 1). These phenolic compounds have molecular masses ranging from 174.1 to 518.7 g/mol. None of the phenolic compounds with a MIC < 1 µg/mL against Gram-negative bacteria or mycobacteria have high molecular masses, probably due to porins’ molecular mass exclusion limit (Bauer et al. ref. 1988; Niederweis ref. 2003).
Influence of solubility on the antibacterial strength and spectrum of activity
The solubility of phenolic compounds can be tentatively classified as follows at pH 7.4: LogD <1 = hydrophilic, LogD between 1 and 5 = amphiphilic, and LogD > 5 = lipophilic. Consequently, phenolic compounds with very strong antibacterial activity are on average amphiphilic (Table 1). Phenolic compounds with a MIC < 1 µg/mL against Gram-positive bacteria are lipophilic or nearly lipophilic. Phenolic compounds with a MIC < 1 µg/mL against Gram-negative bacteria are hydrophilic, amphiphilic, or lipophilic. Lipophilic compounds do not pass through the porins of Gram-negative bacteria (Bauer et al. ref. 1988; van den Berg ref. 2010), suggesting a mechanism of action for lipophilic phenolic compounds involving the permeabilization of the outer membrane. Likewise, porins of mycobacteria likely prevent the entry of lipophilic phenolic compounds. The polar surface area of phenolic compounds with very strong antibacterial activity varies from 29 to 166 Å2.
Main mechanisms of action
Most phenolic compounds are bactericidal. Their two main mechanisms of action are the permeabilization of the cytoplasmic membrane and the destruction of the peptidoglycan wall (Table 3). Downstream effects of cytoplasmic membrane permeabilization include respiratory chain inhibition, change in cytoplasmic pH, generation of reactive oxygen species, and ultimately the inhibition of DNA, RNA, and protein synthesis (Booth ref. 1985; Kubo et al. ref. 2003; Yuan et al. ref. 2021). For mycobacteria, the principal mechanism of action is the inhibition of enzymes, such as DNA primase (Gajadeera et al. ref. 2015) or shikimate kinase (Pandey et al. ref. 2016). There does not appear to be a relationship between molecular mass or solubility with a specific bacterial target.
Table 3.: Mechanism of action.
| MM | LogD | Planar | G | Target | S | References | |
|---|---|---|---|---|---|---|---|
| Non-flavonoids | |||||||
| Hydroxycinnamic acid derivatives | |||||||
| Cinnamic acid (1) | 148.1 | −0.6 | Membrane | G+ | Cai et al. ref. 2019 | ||
| G− | Hemaiswarya and Doble ref. 2010 | ||||||
| Caffeic acid (3) | 180.1 | −1.7 | Membrane | G+ | Hemaiswarya and Doble ref. 2010 | ||
| G− | Hemaiswarya and Doble ref. 2010 | ||||||
| Chlorogenic acid (4) | 354.3 | −3.9 | Membrane | G+ | Cai et al. ref. 2019 | ||
| G− | Hemaiswarya and Doble ref. 2010 | ||||||
| Phenylpropanoids | |||||||
| Eugenol (22) | 164.2 | 2.4 | √ | Membrane | G− | Ashrafudoulla et al. ref. 2020 | |
| Coumarins | |||||||
| Fraxetin (28) | 208.1 | 0.2 | √ | Membrane | G+ | Wang et al. ref. 2014 | |
| DNA | G+ | Wang et al. ref. 2014 | |||||
| RNA | G+ | Wang et al. ref. 2014 | |||||
| Topoisomerases | G+ | Wang et al. ref. 2014 | |||||
| Stilbenes | |||||||
| Resveratrol (56) | 228.2 | 2.8 | DNA | G+ | Paulo et al. ref. 2010 | ||
| Pterostilbene (58) | 256.2 | 3.8 | DNA | G+ | Shih et al. ref. 2021 | ||
| Cajanin stilbene acid (62) | 338.4 | 5.8 | √ | Phosphotransferase | G+ | Tan, Hua, et al. ref. 2020 | |
| ɛ-Viniferin (69) | 454.4 | 4.6 | Membrane | G+ | Basri et al. ref. 2014 | ||
| Dehydro-δ-viniferin (73) | 451.1 | – | Membrane | G+ | Mattio et al. ref. 2019 | ||
| Lignans | |||||||
| (−)-Nortrachelogenin (85) | 374.3 | 1.1 | Membrane | G− | Lee, Ji, et al. ref. 2016 | ||
| Glochidioboside (89) | 522.5 | – | Membrane | G− | Lee, Woo, et al. 2015 | ||
| Magnolol (90) | 266.3 | 4.0 | Membrane | G+ | Liu et al. ref. 2014 | ||
| Honokiol (91) | 266.3 | 4.1 | Membrane | G+ | Liu et al. ref. 2014 | ||
| Hydroxybenzoic acid derivatives | |||||||
| Gallic acid (99) | 170.1 | −2.3 | Membrane | G− | Kang et al. ref. 2018 | ||
| Methyl gallate (100) | 184.1 | 1.3 | Membrane | G− | Acharyya et al. ref. 2015 | ||
| Ethyl gallate (102) | 198.1 | 1.4 | Peptidoglycan | G− | Li, Song, et al. ref. 2016 | ||
| Miscellaneous simple phenolic compounds | |||||||
| Hydroquinone (108) | 110.1 | 0.5 | Membrane | G+ | Jeyanthi et al. ref. 2021 | ||
| Membrane | G0 | Jeyanthi et al. ref. 2021 | |||||
| Peptidoglycan | G− | Ma et al. ref. 2019 | |||||
| Thymol (109) | 150.2 | 3.0 | Membrane | G− | Xu et al. ref. 2008 | ||
| Membrane | G+ | Yuan et al. ref. 2018 | |||||
| Ellagic acid (113) | 302.1 | −2.0 | √ | DNA gyrase | G− | Ohemeng et al. ref. 1993 | |
| Benzoquinones | |||||||
| Thymoquinone (118) | 164.2 | 1.9 | ATP synthase | G− | Ahmad et al. ref. 2015 | ||
| 1,4-Naphthoquinones | |||||||
| Plumbagin (127) | 188.1 | 1.4 | √ | DNA | G− | Farr et al. ref. 1985 | |
| Membrane | G− | Wang, Kong, et al. ref. 2022 | |||||
| Peptidoglycan | G+ | Periasamy et al. ref. 2019 | |||||
| 7-Methyljuglone (129) | 188.1 | 1.3 | √ | Mycothiol disulfide reductase | Mb | Mahapatra et al. ref. 2007 | |
| Shikonin (139) | 288.2 | 3.2 | √ | √ | Membrane | G+ | Lee, Lee, et al. ref. 2015 |
| Peptidoglycan | G+ | Lee, Lee, et al. ref. 2015 | |||||
| ATPase | G+ | Lee, Lee, et al. ref. 2015 | |||||
| Topoisomerases | Plyta et al. ref. 1998 | ||||||
| Anthraquinones | |||||||
| Aloe-emodin (143) | 270.2 | 1.7 | √ | DNA primase | Mb | Gajadeera et al. ref. 2015 | |
| Membrane | G+ | Li et al. ref. 2021 | |||||
| Emodin (145) | 270.2 | 1.7 | √ | Membrane | G− | Gajadeera et al. ref. 2015 | |
| DNA | G− | Zhang et al. ref. 2020 | |||||
| Tannins | |||||||
| Tannic acid (158) | 1701.1 | 3.1 | Peptidoglycan | G+ | Dong et al. ref. 2018 | ||
| Corilagin (159) | 634.4 | 0.9 | Membrane | G− | Li et al. ref. 2013 | ||
| Punicalagin (162) | 1084.7 | −4.4 | Membrane | G− | Li et al. ref. 2020 | ||
| ZP-CT-A | – | – | Membrane | G+ | Kusuda et al. ref. 2006 | ||
| Long chain alkyl phenols | |||||||
| Malabaricone A (164) | 328.4 | 4.5 | √ | Membrane | G+ | Sivadas et al. ref. 2023 | |
| Malabaricone B (165) | 326.4 | 5.6 | √ | Membrane | G+ | Sivadas et al. ref. 2023 | |
| Anacardic acid (170) | 348.5 | 6.4 | √ | Membrane | G+ | Kubo et al. ref. 2003 | |
| Cannabidiol (174) | 324.2 | 6.4 | √ | Membrane | G+ | Blaskovich et al. ref. 2021 | |
| Cannabigerol (175) | 316.4 | 6.7 | √ | Membrane | G+ | Aqawi et al. ref. 2021 | |
| Prenylated phloroglucinols | |||||||
| Rottlerin (182) | 516.5 | 5.8 | √ | Shikimate kinase | Mb | Pandey et al. ref. 2016 | |
| Rhodomyrtone (186) | 442.5 | 5.3 | √ | √ | Membrane | G+ | Saeloh et al. ref. 2018 |
| Peptidoglycan | G+ | Saeloh et al. ref. 2018 | |||||
| Callistemenonone A (189) | 461.1 | – | √ | √ | Membrane | G+ | Xiang et al. ref. 2017 |
| DNA | G+ | Saeloh et al. ref. 2018 | |||||
| Prenylated xanthones | |||||||
| Cochinchinone A (196) | 448.5 | 6.9 | √ | √ | Membrane | G− | Boonnak et al. ref. 2009 |
| α-Mangostin (201) | 410.4 | 4.2 | √ | √ | Respiratory chain | G+ | Nguyen and Marquis ref. 2011 |
| Membrane | G+ | Sivaranjani et al. ref. 2019 | |||||
| Chromanes and chromenes | |||||||
| Brasilin (231) | 286.2 | 1.6 | √ | DNA | G+ | Xu and Lee ref. 2004 | |
| Naphthalenols | |||||||
| Hibicuslide C (237) | 232.3 | – | √ | DNA | G− | Lee, Choi, et al. ref. 2016 | |
| Phenanthrenes | |||||||
| Blestriacin (241) | – | – | √ | Membrane | G+ | Chen et al. ref. 2018 | |
| 4, 8, 4′, 8′-TBT (243) | – | – | √ | Membrane | G+ | Huang et al. ref. 2021 | |
| Blestiarene A (244) | – | – | √ | Membrane | G+ | Zhang et al. ref. 2022 | |
| Densiflorol B (245) | – | √ | Membrane | G+ | Zhang et al. ref. 2022 | ||
| Other non-flavonoids | |||||||
| Hyperenone A (254) | 271.1 | – | √ | MurE ligase | Mb | Osman et al. ref. 2010 | |
| Flavonoids | |||||||
| Chalcones | |||||||
| Licochalcone A (262) | 338.3 | 4.3 | √ | Respiratory chain | G+ | Haraguchi et al. ref. 1998 | |
| Flavanones | |||||||
| Artocarpanone (267) | 302.2 | 2.3 | Membrane | G− | Septama and Panichayupakaranant ref. 2017 | ||
| Naringenin (269) | 272.2 | 2.2 | Membrane | G+ | Tsuchiya and Iinuma ref. 2000 | ||
| Glabrol (273) | 392.4 | 5.7 | √ | Membrane | G+ | Wu et al. ref. 2019 | |
| Lupinifolin (276) | 406.4 | 5.8 | √ | Membrane | G+ | Yusook et al. ref. 2017 | |
| Sophoraflavanone G (279) | 424.4 | 5.3 | √ | Membrane | G+ | Tsuchiya and Iinuma ref. 2000 | |
| Peptidoglycan | G+ | Kim and Kim ref. 2020 | |||||
| Isoflavanones | |||||||
| Bidwillon B (295) | – | – | √ | √ | DNA | G+ | Sato et al. ref. 2003 |
| Flavones | |||||||
| Galangin (299) | 270.2 | 1.5 | √ | Membrane | G+ | Cushnie and Lamb ref. 2005 | |
| DNA helicase | G− | Chen and Huang ref. 2011 | |||||
| Luteolin (301) | 286.2 | 1.1 | √ | Membrane | G+ | Siriwong et al. ref. 2015 | |
| Peptidoglycan | G+ | Siriwong et al. ref. 2015 | |||||
| DNA | G+ | Siriwong et al. ref. 2015 | |||||
| Quercetin (303) | 302.2 | 2.1 | √ | Membrane | G+ | Siriwong et al. ref. 2015 | |
| Peptidoglycan | G+ | Siriwong et al. ref. 2015 | |||||
| DNA | G+ | Siriwong et al. ref. 2015 | |||||
| Kuwanon G (312) | 692.2 | 5.1 | √ | √ | Membrane | G+ | Park et al. ref. 2003 |
| Artocarpin (313) | 436.3 | 4.5 | √ | √ | Membrane | G+ | Septama and Panichayupakaranant ref. 2018 |
| Membrane | G− | Septama et al. ref. 2022 | |||||
| Isoflavones | |||||||
| Tectorigenin (327) | 300.2 | 1.5 | √ | Membrane | G+ | Joung et al. ref. 2014 | |
| ATP-binding cassette | G+ | Joung et al. ref. 2014 | |||||
| Peptidoglycan | G+ | Joung et al. ref. 2014 | |||||
| Glycinol (336) | 272.2 | 1.4 | √ | Membrane | G− | Weinstein and Albersheim ref. 1983 | |
| DNA | G− | Weinstein and Albersheim ref. 1983 | |||||
| Isoflavans | |||||||
| Erycristagallin (339) | 390.4 | 6.3 | √ | √ | Membrane | G+ | Sato et al. ref. 2003 |
| DNA | G+ | Sato et al. ref. 2003 | |||||
| Flavans | |||||||
| EGCG (346) | 458.3 | 1.6 | Membrane | G− | Cao et al. ref. 2019 | ||
| DNA gyrase | G+ | Gradišar et al. ref. 2007 | |||||
| DNA gyrase | G− | Gradišar et al. ref. 2007 | |||||
| Dihydrofolate reductase | G− | Navarro-Martínez et al. ref. 2005 | |||||
MM: molecular mass (g/mol); LogD: at pH 7.4; G: prenyl or long-alkyl chain group; –: non-available; S: spectrum; G+: gram-positive; G−: gram-negative; Mb: mycobacteria.
Structure-activity
General observations
Phenolic compounds with a MIC < 2 µg/mL are, for the most part, not flavonoids (Table 1). These mainly include long-chain alkyl phenols, prenylated phloroglucinols, prenylated xanthones, and 1,4-naphthoquinones. Dimethylallyl, geranyl, farnesyl, lavandulyl, and long-chain alkyl groups tend to penetrate and remain in the hydrophobic region of the cytoplasmic membrane and, in doing so, render it permeable (Tsukiyama et al. ref. 2002; Appendino et al. ref. 2008; Kim et al. ref. 2014; Omosa et al. ref. 2016; Araya-Cloutier et al. ref. 2018; Yuan et al. ref. 2021) (Table 3). In addition, when these groups make phenolic compounds amphiphilic, they become surfactants (Xia et al. ref. 1995). Phenolic compounds that cross the porins of Gram-negative bacteria (hydrophilic or close to hydrophilic) reach the cytoplasmic membrane to destabilize it.
This is probably the case for (−)-nortrachelogenin (85) (Lee, Ji, et al. ref. 2016) and glochidioboside (89) with E. coli O157:H7 (Lee, Woo, et al. 2015). A restricted number of freely rotating chemical bonds and the presence of a planar skeleton (as in anthraquinones or naphthoquinones) favor the intercalation of phenolic compounds within bacterial DNA (Table 3) (Bhakta and Siva ref. 2012). Most phenolic compounds with a MIC < 1 µg/mL against mycobacteria are 1,4-naphthoquinones.
Non-flavonoid phenolic compounds
The presence of quinone scaffolds and an increase in hydrogen bond acceptors enhance the activity of non-flavonoid phenolic compounds (Table 1). Quinones, through the redox cycle (Lown ref. 1983) and in the presence of Cu2+ and O2, generate reactive oxygen species that destroy bacterial DNA (Sakihama et al. ref. 2002). This is the case for plumbagin (127) in E. coli (Farr et al. ref. 1985). Naphthoquinones and other planar phenolic compounds with adjacent hydroxyl and ketone groups chelate Zn2+ ions and, in doing so, inhibit zinc metalloenzymes, such as topoisomerase I (Tse-Dinh and Beran-Steed ref. 1988; Plyta et al. ref. 1998; Tesauro et al. ref. 2010). Catechol groups in tannins and other polyhydroxylated phenolic compounds chelate Fe3+ ions which are necessary for bacterial growth (Serrano et al. ref. 2009; Farha et al. ref. 2020). Similarly, pyrogallol groups chelate divalent cations that are essential for the stabilization of the negative charges in the central oligosaccharide chains of the outer layer of Gram-negative bacteria (Denyer and Maillard ref. 2002; Taguri et al. ref. 2006).
Certain functional groups readily and randomly form cross-links with bacterial macromolecules. For example, furan and pyrone moieties in furanocoumarins form covalent bonds with DNA and proteins (Dall’Acqua et al. ref. 1978; Bordin et al. ref. 1993; Wamer et al. ref. 1995). Similarly, α, β-unsaturated carbonyls in butenolides readily open to form covalent bonds with the nucleophilic groups (thiols) of proteins via hetero-Michael addition reactions (Jackson et al. ref. 2017). Benzoquinones form covalent bonds with proteins (Wipf and Jung ref. 1999; Hettegger et al. ref. 2021). Other examples include α-methylene γ-lactones that alkylate DNA and proteins (Gach and Janecka ref. 2014). Tannins randomly interact with surface proteins and the cytoplasmic membrane (Shimozu et al. ref. 2017; Tintino et al. ref. 2017; Wang et al. ref. 2020) and at high concentrations coagulate proteins (Serrano et al. ref. 2009). Thus, they often behave as invalid metabolic panaceas.
Another mechanism of action is the induction of apoptosis as in pterostilbene (58) with B. cereus (Shih et al. ref. 2021) and quercetin (303) (Li et al. ref. 2015). None of these compounds are known so far to interact with outer membrane protein A (OmpA) in A. baumannii (Nie et al. ref. 2020).
Flavonoids
Prenyl groups enhance the activity of flavonoids against Gram-positive bacteria (Matsumoto et al. ref. 2012; Yuan et al. ref. 2021), as in 7,9,2′,4′-tetrahydroxy-8-isopentenyl-5-methoxychalcone (265) and sophoraflavanone G (279) (Table 1). Sophoraflavanone G (279) is a surfactant that induced the lysis of E. faecium (Tsuchiya and Iinuma ref. 2000; Kim and Kim ref. 2020). Flavonoid glycosides are able to pass through porins and can therefore dissolve the cytoplasmic membrane of Gram-negative bacteria (Tagousop et al. ref. 2018). This could explain why the only flavonoid with a MIC < 2 against a Gram-negative bacterium is kaempferol-7-rhamnoside (317). No flavonoid showed very strong activity against mycobacteria.
Regarding the influence of the number and the position of hydroxyl groups on the antibacterial mechanism of flavonoids, a hydroxyl group at carbon 3 of ring A and hydroxylation of ring B at carbons 3′, 4′, or 5′ promote the intercalation of flavonoids into bacterial DNA (Bartoszewski and Króliczewski ref. 2019). It has also been observed that a hydroxyl group at carbon 3 (Hazni et al. ref. 2008), 5, 6, or 7 enhance antibacterial activity (Hummelova et al. ref. 2015). However, an increasing number of hydroxyl groups results in a decrease in activity (Mori et al. ref. 1987; Xu et al. ref. 2015). Flavonoids with catechol scaffolds or hydroxyl groups at carbon 3 or 5 and a ketone group at position 4 can chelate Fe3+ ions (Porfírio et al. ref. 2014; Jahanshahi et al. ref. 2022).
Synergistic activity with antibiotics
Weakening of intrinsic resistance
Phenolic compounds are primarily synergistic with β-lactam antibiotics against Gram-positive bacteria and, to a lesser extent, with aminoglycosides, tetracyclines, and fluoroquinolones (Table 4). There is no obvious association between the molecular mass or solubility of phenolic compounds and synergistic effects with specific classes of antibiotics. However, there appears to be a link between mechanism of action and synergy with a given class of antibiotic. For example, phenolic compounds targeting the cytoplasmic membrane or the peptidoglycan wall often work synergistically with antibiotics targeting the cytoplasmic membrane or the peptidoglycan wall (Table 4). The synergistic mechanism of action phenolic compounds with prenyl or long-chain alkyl groups relies, at least in part, on the destabilization of the cytoplasmic membrane (Tsukiyama et al. ref. 2002; Appendino et al. ref. 2008; Omosa et al. ref. 2016; Zabawa et al. ref. 2016).
Table 4.: Synergy with antibiotics.
| MM | LogD | Class of antibiotic | Target | Antibiotic | Bacteria | References | |
|---|---|---|---|---|---|---|---|
| Non-flavonoids | |||||||
| Hydroxycinnamic acid derivatives | |||||||
| Cinnamic acid (1) | 148.1 | −0.6 | Aminoglycoside | Ribosomes | Amikacin | S. aureus | Hemaiswarya and Doble ref. 2010 |
| Aminoglycoside | Ribosomes | Amikacin | E. coli | Hemaiswarya and Doble ref. 2010 | |||
| β-Lactam | Peptidoglycan | Ampicillin | S. aureus | Hemaiswarya and Doble ref. 2010 | |||
| β-Lactam | Peptidoglycan | Ampicillin | E. coli | Hemaiswarya and Doble ref. 2010 | |||
| β-Lactam | Peptidoglycan | Ciprofloxacin | S. aureus | Hemaiswarya and Doble ref. 2010 | |||
| β-Lactam | Peptidoglycan | Ciprofloxacin | E. coli | Hemaiswarya and Doble ref. 2010 | |||
| β-Lactam | Peptidoglycan | Ciprofloxacin | P. aeruginosa | Hemaiswarya and Doble ref. 2010 | |||
| Macrolide | Ribosomes | Erythromycin | S. aureus | Hemaiswarya and Doble ref. 2010 | |||
| Macrolide | Ribosomes | Erythromycin | E. coli | Hemaiswarya and Doble ref. 2010 | |||
| Glycopeptide | Ribosomes | Vancomycin | S. aureus | Hemaiswarya and Doble ref. 2010 | |||
| Glycopeptide | Peptidoglycan | Vancomycin | E. coli | Hemaiswarya and Doble ref. 2010 | |||
| Caffeic acid (3) | 180.1 | −1.7 | Fluoroquinolone | DNA | Norfloxacin | S. aureus | Lima et al. ref. 2016 |
| β-Lactam | Peptidoglycan | Imipenem | E. coli | Lima et al. ref. 2016 | |||
| Chlorogenic acid (4) | 354.3 | −3.9 | Fluoroquinolone | DNA | Levofloxacin | K. pneumoniae | Tan, Gao, et al. ref. 2020 |
| Macrolide | Ribosomes | Erythromycin | S. aureus | Kępa et al. ref. 2018 | |||
| Licosamide | Ribosomes | Clindamycin | S. aureus | Kępa et al. ref. 2018 | |||
| β-Lactam | Peptidoglycan | Cefoxitin | S. aureus | Kępa et al. ref. 2018 | |||
| Phenylpropanoids | |||||||
| Eugenol (22) | 164.2 | 2.4 | Aminoglycoside | Ribosomes | Streptomycin | L. monocytogenes | Liu et al. ref. 2015 |
| Aminoglycoside | Ribosomes | Streptomycin | S. typhimuriim | Liu et al. ref. 2015 | |||
| Coumarins | |||||||
| Isoimperatorin (32) | 270.2 | – | Ansamycin | DNA | Rifampicin | M. tuberculosis | Guo et al. ref. 2014 |
| Ethylenediamine | Wall | Ethambutol | M. tuberculosis | Guo et al. ref. 2014 | |||
| Hydrazide | Membrane | Isoniazid | M. tuberculosis | Guo et al. ref. 2014 | |||
| Stilbenes | |||||||
| Resveratrol (56) | 228.2 | 2.8 | Polypeptide | Membrane | Polymixin B | E. coli | Liu et al. ref. 2020 |
| Polypeptide | Membrane | Polymixin B | K. pneumoniae | Liu et al. ref. 2020 | |||
| Pterostilbene (58) | 256.2 | 3.8 | β-Lactam | Peptidoglycan | Oxacillin | MRSA | Ishak et al. ref. 2016 |
| Aminoglycoside | Ribosomes | Gentamycin | S. aureus | Lee et al. ref. 2017 | |||
| Aminoglycoside | Ribosomes | Gentamycin | P. aeruginosa | Lee et al. ref. 2017 | |||
| Aminoglycoside | Ribosomes | Gentamycin | E. coli | Lee et al. ref. 2017 | |||
| Cajanin stilbene acid (62) | 338.4 | 5.8 | Polypeptide | Membrane | Colistin | E. coli | Jia et al. ref. 2023 |
| ɛ-Viniferin (69) | 454.4 | 4.6 | Glycopeptide | Peptidoglycan | Vancomycin | MRSA | Basri et al. ref. 2014 |
| Lignans | |||||||
| Magnolol (90) | 266.3 | 4.0 | Aminoglycoside | Ribosomes | Amikacin | MRSA | Zuo et al. ref. 2015 |
| Fluoroquinolone | DNA | Levofloxacin | MRSA | Zuo et al. ref. 2015 | |||
| Phosphonic | Peptidoglycan | Fosfomycin | MRSA | Zuo et al. ref. 2015 | |||
| β-Lactam | Peptidoglycan | Piperacillin | MRSA | Zuo et al. ref. 2015 | |||
| Honokiol (91) | 266.3 | 4.1 | Aminoglycoside | Ribosomes | Amikacin | MRSA | Zuo et al. ref. 2015 |
| Fluoroquinolone | DNA | Levofloxacin | MRSA | Zuo et al. ref. 2015 | |||
| Phosphonic | Peptidoglycan | Fosfomycin | MRSA | Zuo et al. ref. 2015 | |||
| β-Lactam | Peptidoglycan | Piperacillin | MRSA | Zuo et al. ref. 2015 | |||
| Hydroxybenzoic acid derivatives | |||||||
| Gallic acid (99) | 170.1 | −2.3 | Sulfonamide | Folic acid | Sulfamethoxazole | P. aeruginosa | Jayaraman et al. ref. 2010 |
| Tetracycline | Ribosomes | Tetracycline | P. aeruginosa | Jayaraman et al. ref. 2010 | |||
| Methyl gallate (100) | 184.1 | 1.3 | Fluoroquinolone | DNA | Ciprofloxacin | Salmonella sp. | Choi et al. ref. 2008 |
| Protocatechuic acid (104) | 154.1 | −1.8 | Sulfonamide | Folic acid | Sulfamethoxazole | P. aeruginosa | Jayaraman et al. ref. 2010 |
| Miscellaneous simple phenolic compounds | |||||||
| Thymol (109) | 150.2 | 3.0 | Aminoglycoside | Ribosomes | Streptomycin | L. monocytogenes | Liu et al. ref. 2015 |
| Ellagic acid (113) | 302.1 | −2.0 | Tetracycline | Ribosomes | Tetracycline | E. coli | Jenic et al. ref. 2021 |
| Aminocoumarin | DNA | Novobiocin | A. baumannii | Chusri et al. ref. 2009 | |||
| Steroid | Ribosome | Fusidic acid | A. baumannii | Chusri et al. ref. 2009 | |||
| Ansamycin | DNA | Rifampicin | A. baumannii | Chusri et al. ref. 2009 | |||
| Cinnamaldehyde (116) | 132.1 | 1.7 | Aminoglycoside | Ribosomes | Streptomycin | L. monocytogenes | Liu et al. ref. 2015 |
| Aminoglycoside | Ribosomes | Streptomycin | S. typhimurium | Liu et al. ref. 2015 | |||
| Aminoglycoside | Ribosomes | Streptomycin | S. typhimurium | Liu et al. ref. 2015 | |||
| Benzoquinones | |||||||
| Thymoquinone (118) | 164.2 | 1.9 | Aminoglycoside | Ribosomes | Gentamicin | S. epidermidis | Liu et al. ref. 2015 |
| β-Lactam | Peptidoglycan | Penicillin | S. epidermidis | Dera et al. ref. 2021 | |||
| Fluoroquinolone | DNA | Ofloxacin | K. pneumoniae | Dera et al. ref. 2021 | |||
| Tetracycline | Ribosomes | Tetracycline | K. pneumoniae | Dera et al. ref. 2021 | |||
| β-Lactam | Peptidoglycan | Penicillin | K. pneumoniae | Dera et al. ref. 2021 | |||
| Quinolone | DNA | Nalidixic acid | K. pneumoniae | Dera et al. ref. 2021 | |||
| 1,4-Naphthoquinones | |||||||
| Plumbagin (124) | 188.1 | 1.4 | Aminoglycoside | Ribosomes | Gentamycin | K. pneumoniae | Chen et al. ref. 2020 |
| β-Lactam | Peptidoglycan | Oxacillin | S. aureus | Rondevaldova et al. ref. 2015 | |||
| polypeptide | Membrane | Colistin | P. aeruginosa | Wang, Wang, et al. ref. 2022 | |||
| Lawsone methyl ether (126) | 188.1 | 1.6 | β-Lactam | Peptidoglycan | Ampicillin | MRSA | Meah et al. ref. 2020 |
| 7-Methyljuglone (129) | 188.1 | 1.3 | Hydrazide | Membrane | Isoniazid | M. tuberculosis | Bapela et al. ref. 2006 |
| Ansamycin | DNA | Rifampicin | M. tuberculosis | Bapela et al. ref. 2006 | |||
| Shikonin (139) | 288.2 | 3.2 | Aminoglycoside | Ribosomes | Gentamycin | MRSA | Li et al. ref. 2022 |
| β-Lactam | Peptidoglycan | Amoxicillin | MRSA | Li et al. ref. 2022 | |||
| Aminoglycoside | Ribosome | Amikacin | MRSA | Li et al. ref. 2022 | |||
| Anthraquinones | |||||||
| Rhein (144) | 284.2 | −0.5 | β-Lactam | Peptidoglycan | Amoxicillin | MRSA | Joung et al. ref. 2012 |
| β-Lactam | Peptidoglycan | Ampicillin | MRSA | Joung et al. ref. 2012 | |||
| Nitroimidazole | DNA | Metronidazole | P. gingivalis | Azelmat et al. ref. 2015 | |||
| Emodin (145) | 270.2 | 1.7 | β-Lactam | Peptidoglycan | Ampicillin | MRSA | Lee, Kang, et al. ref. 2010 |
| β-Lactam | Peptidoglycan | Oxacillin | MRSA | Lee, Kang, et al. ref. 2010 | |||
| Tannins | |||||||
| Theasinensin A (155) | 914.7 | 2.8 | β-Lactam | Peptidoglycan | Oxacillin | MRSA | Hatano et al. ref. 2003 |
| β-Lactam | Peptidoglycan | Penicillin G | MRSA | Hatano et al. ref. 2003 | |||
| β-Lactam | Peptidoglycan | Ampicillin | MRSA | Hatano et al. ref. 2003 | |||
| Aminoglycoside | Ribosomes | Streptomycin | MRSA | Hatano et al. ref. 2003 | |||
| Corilagin (159) | 634.4 | 0.9 | β-Lactam | Peptidoglycan | Oxacillin | MRSA | Shimizu et al. ref. 2001 |
| Tannic acid (158) | 1701.1 | 3.1 | Aminocoumarin | DNA | Novobiocin | A. baumannii | Chusri et al. ref. 2009 |
| Steroid | Ribosomes | Fusidic acid | A. baumannii | Chusri et al. ref. 2009 | |||
| Ansamycin | DNA | Rifampicin | A. baumannii | Chusri et al. ref. 2009 | |||
| Punicalagin (162) | 1084.7 | −4.4 | β-Lactam | Peptidoglycan | Oxacillin | MRSA | Mun et al. ref. 2018 |
| ZP-CT-A | – | – | β-Lactam | Peptidoglycan | Oxacillin | MRSA | Kusuda et al. ref. 2006 |
| β-Lactam | Peptidoglycan | Penicillin G | MRSA | Kusuda et al. ref. 2006 | |||
| β-Lactam | Peptidoglycan | Ampicillin | MRSA | Kusuda et al. ref. 2006 | |||
| β-Lactam | Peptidoglycan | Cefmetazole | MRSA | Kusuda et al. ref. 2006 | |||
| Long chain alkyl phenols | |||||||
| Malabaricone B (165) | 328.4 | 4.5 | Aminoglycoside | Ribosomes | Gentamycin | S. aureus | Sivadas et al. ref. 2023 |
| Polypeptide | Membrane | Daptomycin | S. aureus | Sivadas et al. ref. 2023 | |||
| Anacardic acid (170) | 348.5 | 6.4 | β-Lactam | Peptidoglycan | Methicillin | MRSA | Muroi and Kubo ref. 1996 |
| Cannabidiol (174) | 324.2 | 6.4 | Polypeptide | Membrane | Polymixin B | A. baumannii | Hussein et al. ref. 2022 |
| Polypeptide | Membrane | Bacitracin | S. aureus | Wassmann et al. ref. 2020 | |||
| β-Lactam | peptidoglycan | Ampicillin | Gram-S. typhimurium | Gildea et al. ref. 2022 | |||
| Polypeptide | Membrane | Polymyxin B | S. typhimurium | Gildea et al. ref. 2022 | |||
| Prenylated xanthones | |||||||
| Isojacareubin (195) | 328.3 | 2.8 | β-Lactam | Peptidoglycan | Ampicillin | MRSA | Zuo et al. ref. 2012 |
| β-Lactam | Peptidoglycan | Ceftazidime | MRSA | Zuo et al. ref. 2012 | |||
| Fluoroquinolone | DNA | Levofloxacin | MRSA | Zuo et al. ref. 2012 | |||
| α-Mangostin (201) | 410.4 | 4.2 | Tetracycline | Ribosomes | Tetracycline | S. aureus | Ahmad et al. ref. 2019 |
| Macrolide | Ribosomes | Erythromycin | S. aureus | Ahmad et al. ref. 2019 | |||
| Licosamide | Ribosomes | Clindamycin | S. aureus | Ahmad et al. ref. 2019 | |||
| Aminoglycoside | Ribosomes | Gentamicin | VRE | Sakagami et al. ref. 2005 | |||
| Glycopeptide | Peptidoglycan | Vancomycin | MRSA | Sakagami et al. ref. 2005 | |||
| γ-Mangostin (204) | 396.4 | 3.8 | β-Lactam | Peptidoglycan | Penicillin G | L. interrogans | Seesom et al. ref. 2013 |
| Naphthalenols | |||||||
| Hibicuslide C (237) | 232.3 | – | Ansamycin | DNA | Rifampicin | P. aeruginosa | Lee, Choi, et al. ref. 2016 |
| Fluoroquinolone | DNA | – | P. aeruginosa | Lee, Choi, et al. ref. 2016 | |||
| Phenanthrenes | |||||||
| 4,8,4′,8′-TBT (243) | – | – | Glycopeptide | Peptidoglycan | Vancomycin | S. aureus | Huang et al. ref. 2021 |
| Macrolide | Ribosomes | Erythromycin | S. aureus | Huang et al. ref. 2021 | |||
| Flavonoids | |||||||
| Chalcones | |||||||
| Licochalcone A (262) | 338.3 | 4.3 | Nitroimidazole | DNA | Metronidazole | P. gingivalis | Azelmat et al. ref. 2015 |
| 7,9,2′,4′-TIMC (265) | – | – | β-Lactam | Peptidoglycan | Gentamicin | MRSA | Lee, Kim, et al. ref. 2010 |
| β-Lactam | Peptidoglycan | Ampicillin | MRSA | Lee, Kim, et al. ref. 2010 | |||
| β-Lactam | Peptidoglycan | Ampicillin | VRE | Lee, Kim, et al. ref. 2010 | |||
| Licosamide | Ribosome | Clindamycin | VRE | Lee, Kim, et al. ref. 2010 | |||
| Flavanones | |||||||
| Artocarpanone (267) | 302.2 | 2.3 | Fluoroquinolone | DNA | Norfloxacin | MRSA | Septama et al. ref. 2017 |
| Lupinifolin (276) | 406.4 | 5.8 | β-Lactam | Peptidoglycan | Ampicillin | MRSA | Rattanakiat et al. ref. 2021 |
| β-Lactam | Peptidoglycan | Cloxacillin | MRSA | Rattanakiat et al. ref. 2021 | |||
| Sophoraflavanone G (279) | 424.4 | 5.3 | β-Lactam | Peptidoglycan | Ampicillin | S. mutans | Cha et al. ref. 2007 |
| Fluoroquinolone | DNA | Norfloxacin | MRSA | Sun et al. ref. 2020 | |||
| Phosphonic | Peptigoglycan | Fosfomycin | MRSA | Sakagami et al. ref. 1998 | |||
| Glycopeptide | Peptidoglycan | Vancomycin | MRSA | Sakagami et al. ref. 1998 | |||
| Isoflavans | |||||||
| Glabridin (284) | 324.3 | 4.3 | Nitroimidazole | DNA | Metronidazole | P. gingivalis | Azelmat et al. ref. 2015 |
| Bidwillon B (295) | – | – | Monoxycarbolic acid | Ribosomes | Mupirocin | MRSA | Sato et al. ref. 2003 |
| Isoflavanones | |||||||
| Eryzerin C (296) | 422.5 | – | Glycopeptide | Peptidoglycan | Vancomycin | VRE | Sato et al. ref. 2004 |
| Flavones | |||||||
| Baicalein (298) | 270.2 | 1.6 | β-Lactam | Peptidoglycan | Ampicillin | S. suis | Lu et al. ref. 2021 |
| Galangin (299) | 270.2 | 1.5 | β-Lactam | Peptidoglycan | Ceftazidime | S. aureus | Eumkeb et al. ref. 2010 |
| β-Lactam | Peptidoglycan | Ampicillin | MRSA | Lu et al. ref. 2021 | |||
| Luteolin (301) | 286.2 | 1.1 | β-Lactam | Peptidoglycan | Ceftazidime | S. pyogenes | Siriwong et al. ref. 2015 |
| Quercetin (303) | 302.2 | 2.1 | β-Lactam | Peptidoglycan | Ceftazidime | S. pyogenes | Siriwong et al. ref. 2015 |
| Myricetin (305) | 318.2 | 1.5 | Sulfonamide | Folic acid | Sulfamethoxazole | P. aeruginosa | Jayaraman et al. ref. 2010 |
| β-Lactam | Peptidoglycan | Oxacillin | MRSA | Pinto et al. ref. 2020 | |||
| Nitroimidazole | DNA | Metronidazole | P. gingivalis | Azelmat et al. ref. 2015 | |||
| Morusin (310) | 420.2 | 4.7 | β-Lactam | Peptidoglycan | Oxacillin | MRSA | Aelenei et al. ref. 2020 |
| Kuwanon G (312) | 692.2 | 5.1 | β-Lactam | Peptidoglycan | Oxacillin | MRSA | Aelenei et al. ref. 2020 |
| Artocarpin (313) | 436.3 | 4.5 | Tetracycline | Ribosome | Tetracycline | P. aeruginosa | Septama et al. ref. 2022 |
| TLRP (320) | 450.4 | – | Fluoroquinolone | DNA | Levofloxacin | MRSA | An et al. ref. 2011 |
| β-Lactam | Peptidoglycan | Ceftazidime | MRSA | An et al. ref. 2011 | |||
| Isoflavones | |||||||
| Biochanin A (324) | 284.2 | 1.9 | Fluoroquinolone | DNA | Ciprofloxacin | S. aureus | Liu et al. ref. 2011 |
| Erybraedin A (342) | 392.4 | 6.0 | Glycopeptide | Peptidoglycan | Vancomycin | VRE | Sato et al. ref. 2004 |
| Flavans | |||||||
| EGCG (346) | 458.3 | 1.6 | Sulfonamide | Folic acid | Sulfamethoxazole | S. maltophilia | Navarro-Martínez et al. ref. 2005 |
–: non-available; LogD: at pH 7.4.
Antibiotics whose activity is increased against Gram-negative bacteria in the presence of phenolic products are often those that target the synthesis of ribosomes, DNA, or folic acid metabolism, as well as the lipopolysaccharide envelope. For example, polymyxin B, colistin, and other antibiotics whose functioning is based on the permeabilization of the outer envelope of lipopolysaccharides allow the penetration of phenolic compounds (amphiphilic or lipophilic) previously unable to cross porins. This mechanism could, at least in part, explain the synergy observed between polymyxin B and cajanin stilbene acid (62) in mice against E. coli (Jia et al. ref. 2023) and polymyxin B and cannabidiol (174) against polymyxin B-resistant A. baumannii (Hussein et al. ref. 2022). In addition, pyrogallol form complexes with divalent cations which are essential for the stabilization of the outer lipopolysaccharides coat of Gram-negative bacteria (Denyer and Maillard ref. 2002; Taguri et al. ref. 2006) and thus reduce the resistance of Gram-negative bacteria to antibiotics as in the case of ellagic acid (113) and tannic acid (158) (Andjelković et al. ref. 2006; Jayaraman et al. ref. 2010). Tannins, such as corilagin (159), work synergistically with β-lactam antibiotics against Gram-positive bacteria (Shimizu et al. ref. 2001).
Phenolic compounds are not only synergistic with antibiotics but also among each other. The question then arises as to whether the synergy of phenolic compounds would constitute an additional strategy developed by plants to keep phytopathogenic bacteria in check. For example, rhein (144) is synergistic with licochalcone A (262), glabridin (284), or myricetin (305) against P. gingivalis (Azelmat et al. ref. 2015).
Efflux pumps
Gram-negative bacteria resist antibacterial phenolic compounds by means of efflux pumps (Kuete et al. ref. 2010). Several phenolic compounds inhibit these pumps, such as gallic acid (99) and ellagic acid (113) with TetK in S. aureus (Macêdo et al. ref. 2022), juglone (124) (Zmantar et al. ref. 2016), and tannic acid (158) (Tintino et al. ref. 2017). Ellagic acid (113) and tannic acid (158) inhibited efflux pumps in A. baumannii (Chusri et al. ref. 2009). It appears that prenyl groups particularly farnesyl and lavandulyl groups, can bind to and inhibit voltage-gated Ca2+ channels (De Loof and Schoofs ref. 2019). The resulting decrease in the concentration of Ca2+ ions in the cytoplasm leads to the inhibition of Ca2+-dependent efflux pumps (Nava et al. ref. 2020). This mechanism likely explains why sophoraflavanone G (279) inhibited NorA in MRSA (Sun et al. ref. 2020). Some molecules used therapeutically that act on human neuroreceptors, such as reserpine are in vitro capable to inhibit bacterial efflux pumps (Piddock et al. ref. 2010; Sridevi et al. ref. 2017; Saber and Kandala ref. 2018). It raises the question of whether neuroactive phenolic products would be a potential group of pump inhibitors.
β-Lactamases
Benzoic acid derivatives (Jiamboonsri et al. ref. 2023), proanthocyanidin oligomers (Kusuda et al. ref. 2006), and flavones tend to inhibit β-lactamases (Siriwong et al. ref. 2015).
Escaping the development resistance
Antibacterial phenolic compounds that target the cytoplasmic membrane of Gram-positive bacteria evade bacterial resistance. This the case for instance of brasilin (231) with MRSA (Xu and Lee ref. 2004), sophoraflavanone G (279) with MRSA (Weng et al. ref. 2023), blestriacin (241) with S. aureus (Chen et al. ref. 2018), malabaricone B (165) with S. aureus (Sivadas et al. ref. 2023), glabrol (273) with MRSA (Wu et al. ref. 2019), as well as cannabidiol (174) with MRSA (Blaskovich et al. ref. 2021).
Toxicity
Unlike traditional antibiotics which come from bacteria or fungi, phenolic compounds from Angiosperms have the advantage of not having specific bacterial targets. It allows them to avoid the development of resistance but gives them a generally narrow therapeutic window. Most phenolic compounds have the disadvantage of often being toxic in vitro for mammalian cells at concentrations close to or similar to their MICs. This is the case for xanthones (Boonsri et al. ref. 2006; Mahabusarakam et al. ref. 2008; Yahayu et al. ref. 2013; Pattamadilok ref. 2016), lignans (Syu et al. ref. 2004; Manna et al. ref. 2015), naphthoquinones (Gu et al. ref. 2004; Yang et al. ref. 2019), coumarins (Phatchana and Yenjai ref. 2014), stilbene oligomers (Sahidin et al. ref. 2017), prenylated flavonoids (Sohn et al. ref. 2004), phenolic glycosides (Zeng et al. ref. 2015), and anthraquinones (Ali et al. ref. 2000). Planar phenolic compounds, such as anthraquinones can intercalate into DNA, inhibit topoisomerase II, and induce chromosomal damage in mammalian cells (Mueller and Stopper ref. 1999; Bhakta and Siva ref. 2012; Chakarov et al. ref. 2014). Examples of pan-assay interference compounds (PAINS) or invalid metabolic panacea (IMP) are curcumin (75) and tannins (Bisson et al. ref. 2016; Nelson et al. ref. 2017). It is therefore necessary to determine the selectivity indices of antibacterial phenolic compounds. An antibacterial phenolic compound with a selectivity index >10 merits further pharmacological examination (Tamargo et al. ref. 2015). This is the case for 2-methoxy-7-methyljuglone (128) (Gu et al. ref. 2004), maritinone (132), 3,3′-biplumbagin (135) (Uc-Cachón et al. ref. 2014), malabaricone A (164) (Orabi et al. ref. 1991), and malabaricone B (165) (Sivadas et al. ref. 2023). Phenolic products with unfavorable selectivity indexes can be used to synthesize less toxic antibacterial derivatives (Cham et al. ref. 2023).
Clinical potential
Several phenolic compounds identified in Asian and Pacific Angiosperms can combat bacterial infections in vivo. For example, ethyl gallate (100) administered orally at a dose of 50 mg/kg/day increased the survival rate of mice infected with S. typhimurium by more than 70% (Choi et al. ref. 2014). Anacardic acid (170) and glabrol (273) were active against MRSA and VRE in insects, respectively (Wu et al. ref. 2019; Saedtler et al. ref. 2020). Furthermore, a very interesting aspect of phenolic compounds pharmacology is that they can be both antibacterial and anti-inflammatory or even immunomodulatory. The inflammatory response during infections caused by Gram-negative bacteria is owed, at least in part, by lipopolysaccharides which induce the secretion of nitric oxide and cytokines. Antibacterial phenolic compounds like euryacoumarin A (30) inhibit lipopolysaccharide-induced nitric oxide production in RAW264.7 (IC50: 35.6 μM) (Song et al. ref. 2017). Vitexin (323) given at the dose of 400 µg/kg inhibited lipopolysaccharides-induced lung inflammation in mice (De Melo et al. ref. 2005). Vitexin (323) given parenterally at the dose of 60 mg/kg to mice infected with S. aureus attenuated the production of pro-inflammatory cytokines (Chen et al. ref. 2022). Baicalin (321) was able to protect mice against S. typhimurium infection, modulate bacterial virulence, and quell the host inflammatory response (Wu et al. ref. 2018). Another example is [6]-gingerol (167) which exerted both antibacterial and immunomodulatory activity against M. tuberculosis in mice (Bhaskar et al. ref. 2020).
Concerning the synergistic effect of antibacterial phenolic compounds with antibiotics in vivo, examples are chlorogenic acid (4) with levofloxacin against K. pneumoniae (Tan, Gao, et al. ref. 2020), plumbagin (127) with colistin against colistin-resistant P. aeruginosa (Wang, Kong, et al. ref. 2022), and sophoraflavanone G (279) with norfloxacin against effluxing antibiotic-resistant S. aureus (Sun et al. ref. 2020). Baicalein (298) was synergistic with ampicillin against Streptococcus suis (Lu et al. ref. 2021), with linezolid against S. aureus (Liu et al. ref. 2020), and with myricetin (305) with oxacillin against MRSA (Pinto et al. ref. 2020).
Concluding remarks
The knowledge accumulated over the last decades highlights that the phenolic compounds with very strong antibacterial activity identified in the Angiosperms of Asia and the Pacific mainly come from fabids, often carry isoprene or long-chain alkyl groups, are often planar, with a molecular mass ranging from ∼200 to 400 g/mol, and are often amphiphilic. These products are mainly active against Gram-positive bacteria, and primarily target the cytoplasmic membrane, thus avoiding the development of resistance. 2-Methoxy-7-methyljuglone (128), malabaricone A (164), and malabaricone B (165) (Sivadas et al. ref. 2023) with MIC values <1 µg/mL and selectivity indices >10 could potentially be developed as antibacterial agents as well as anacardic acid (170). Unlike commonly used antibiotics, some of these phenolic compounds are not only antibacterial or antibiotic potentiators but also anti-inflammatory or immunomodulators, such as [6]-gingerol (167), baicalin (321), and vitexin (323). The clinical development of antibiotics or antibiotic potentiators to treat pan-resistant bacteria from phenolic compounds from Asia and Pacific Angiosperms should come to light.
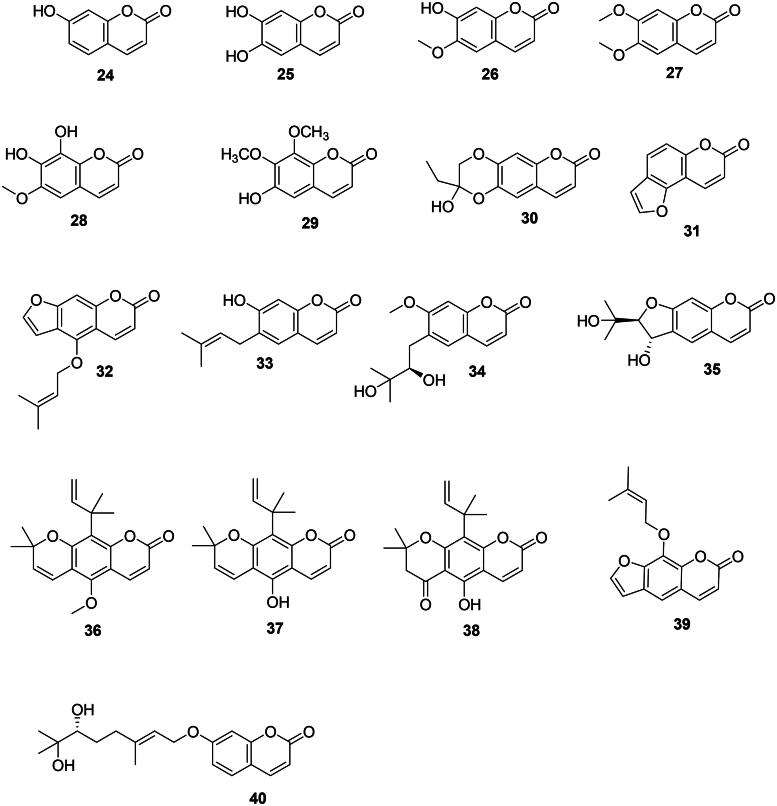
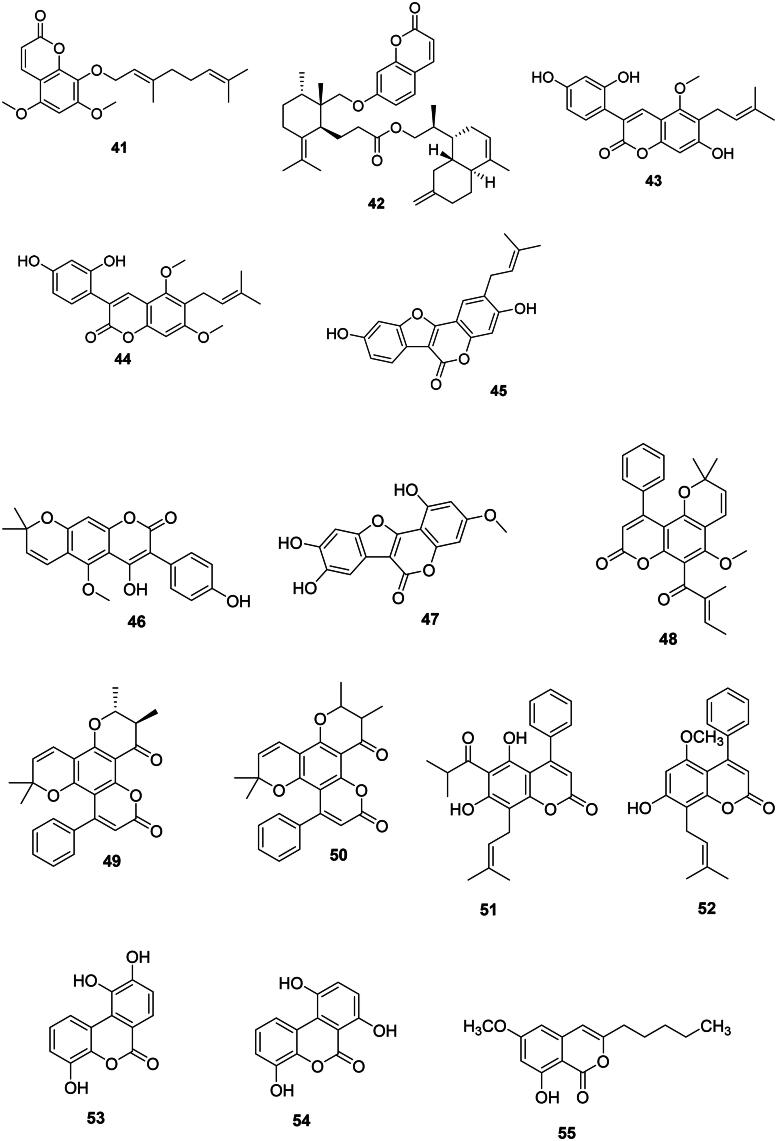
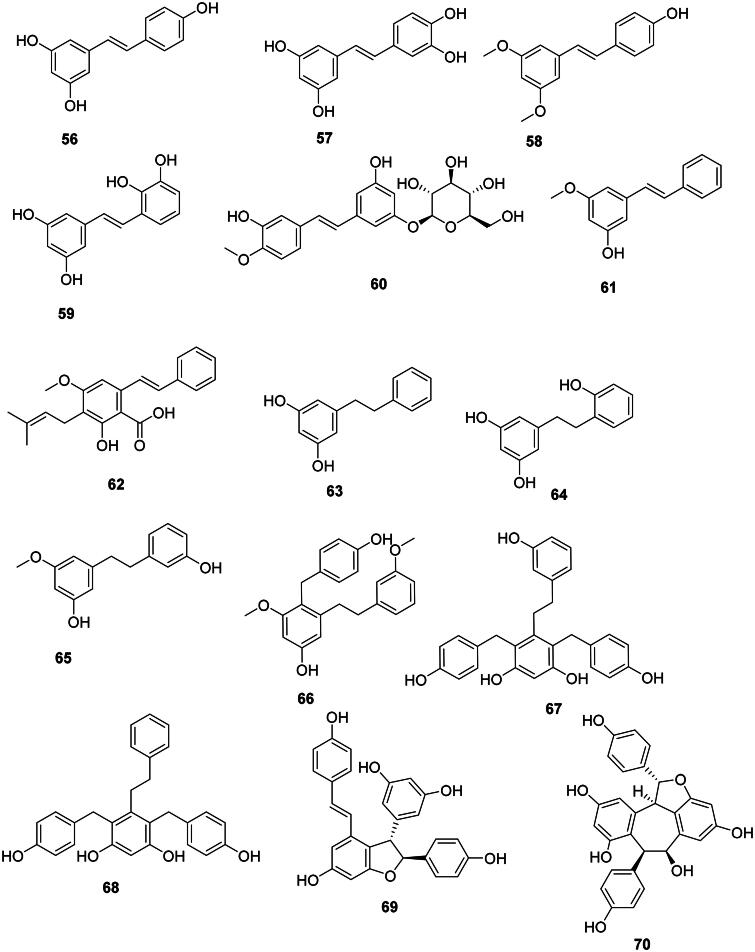
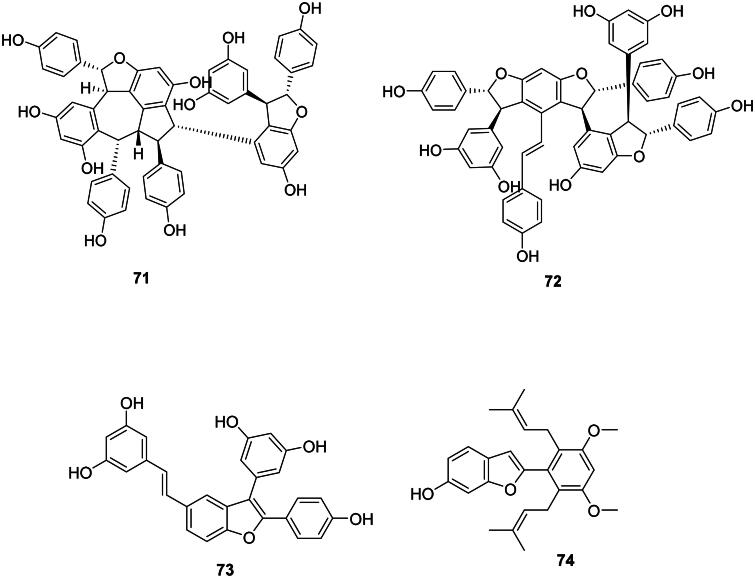
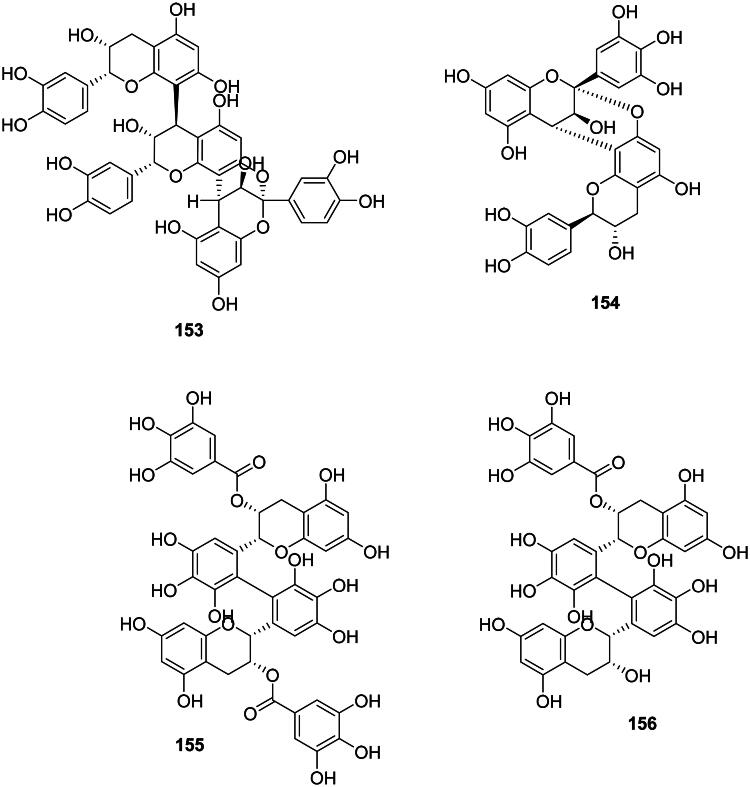
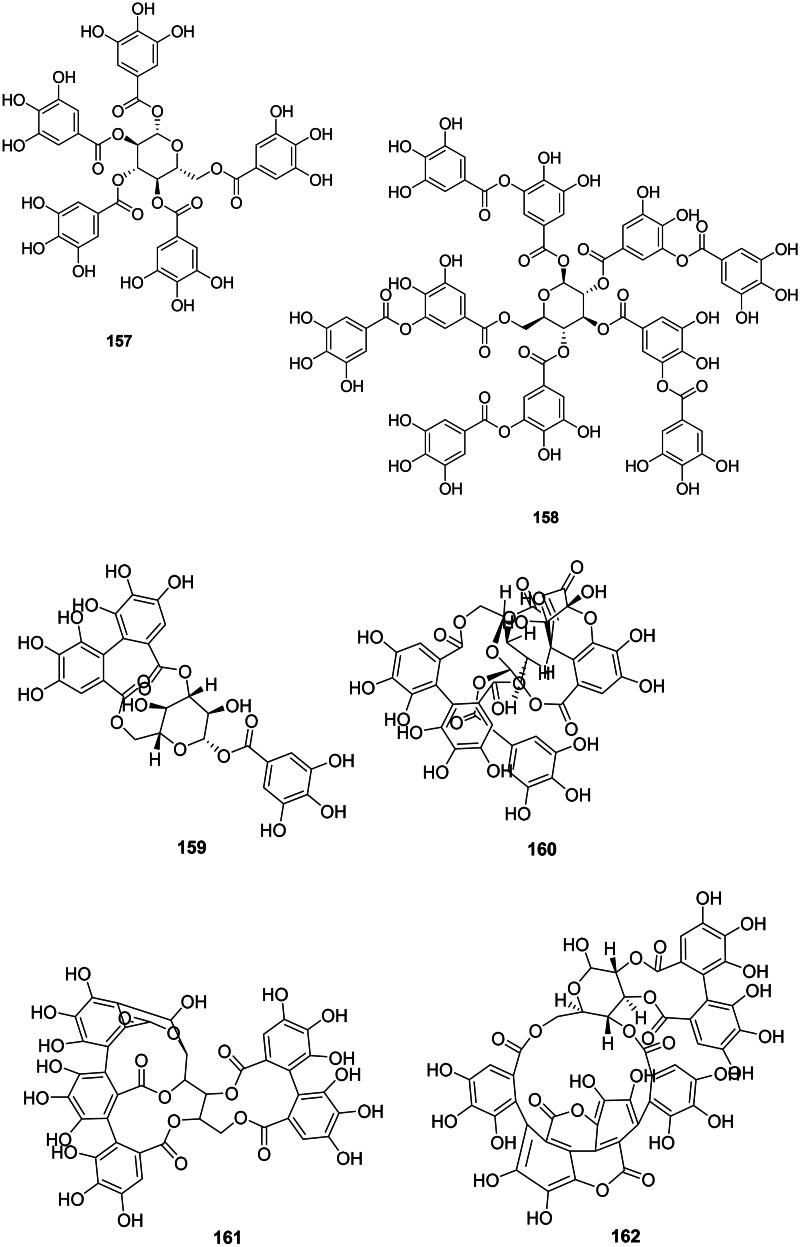
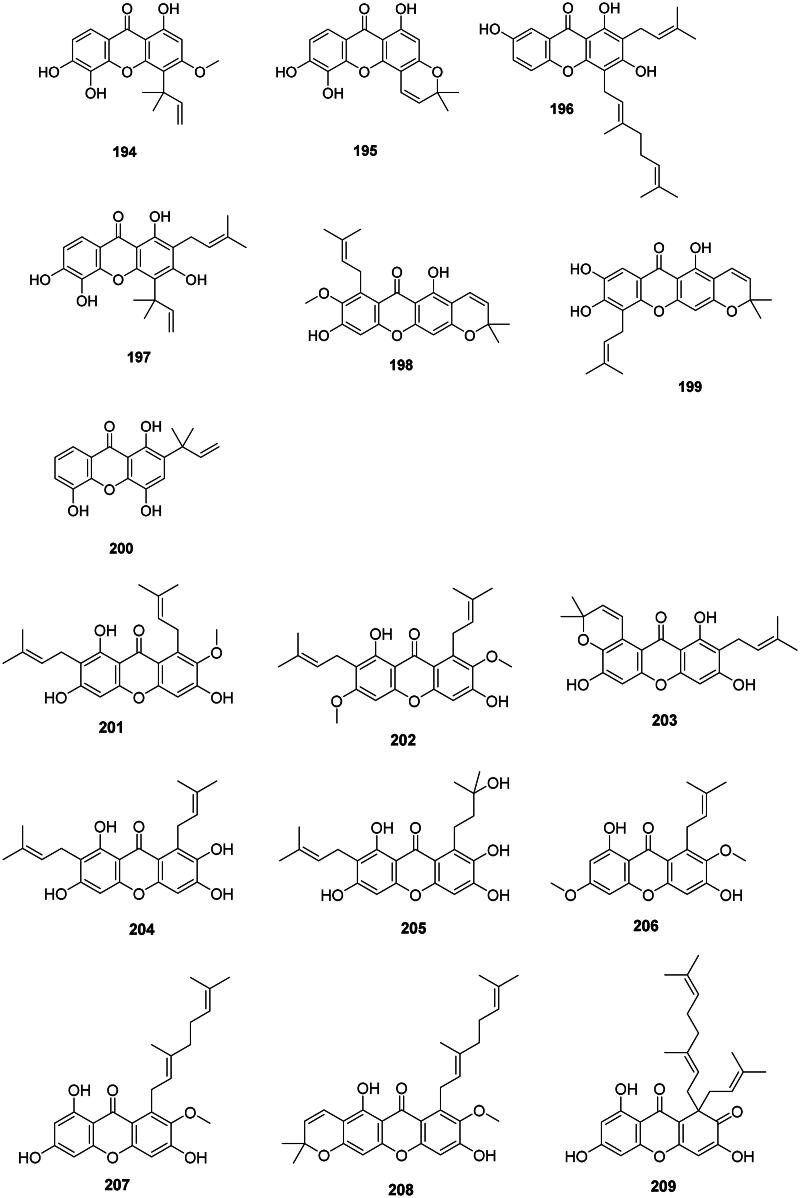
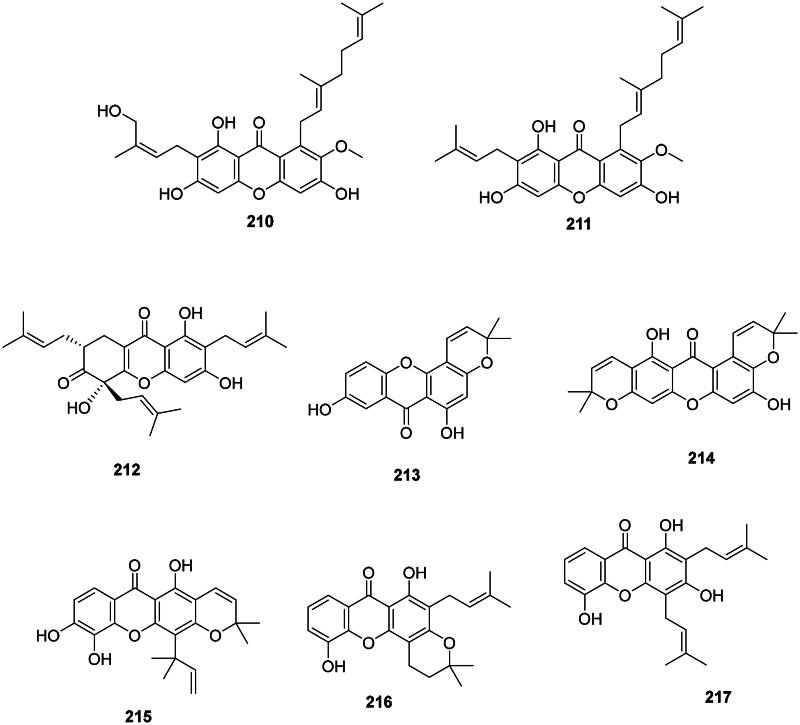
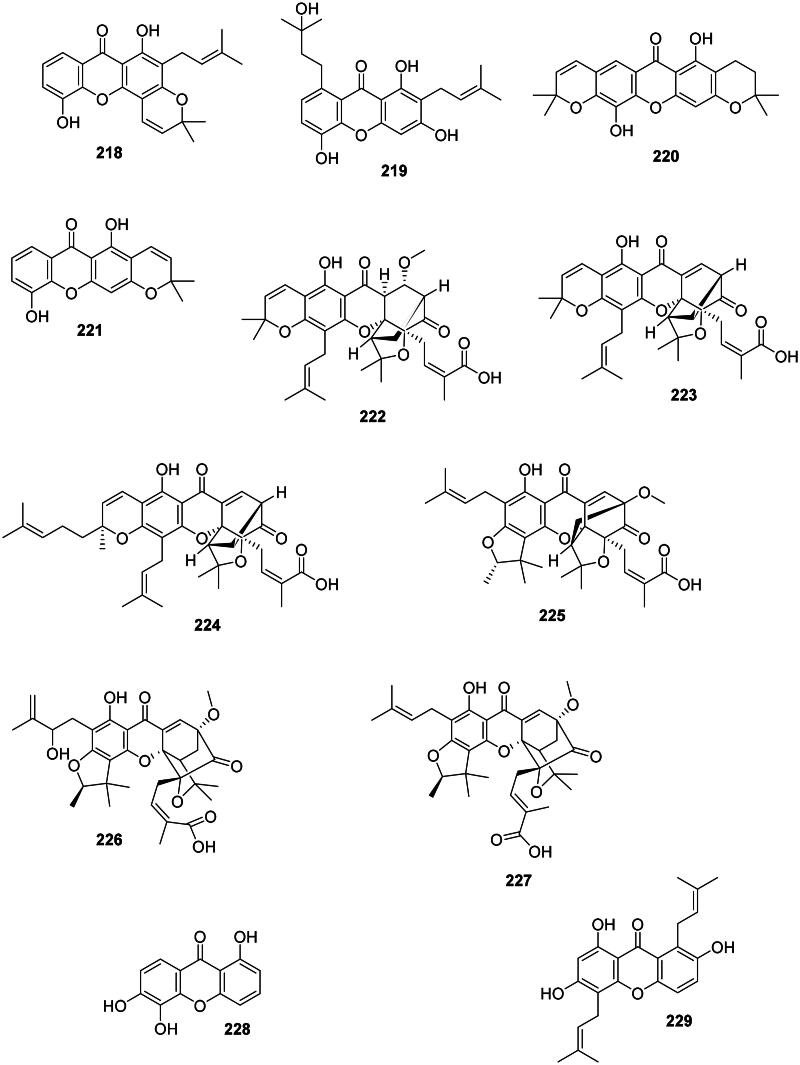
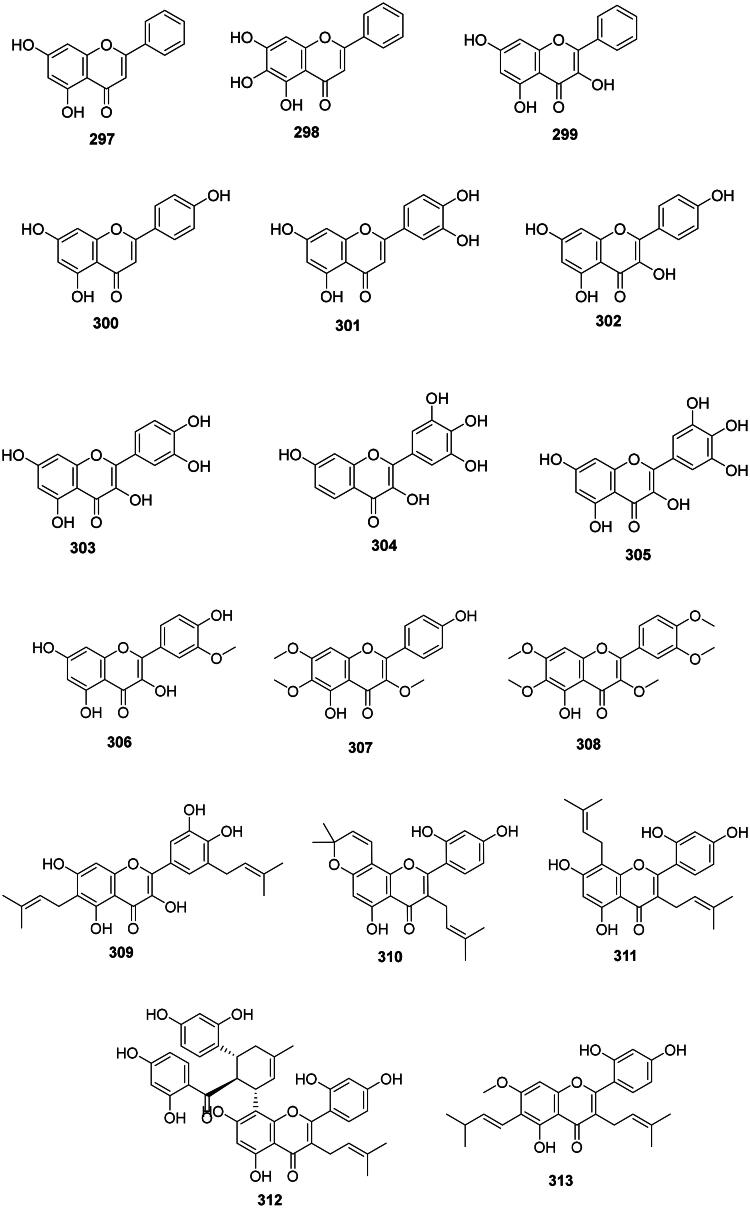
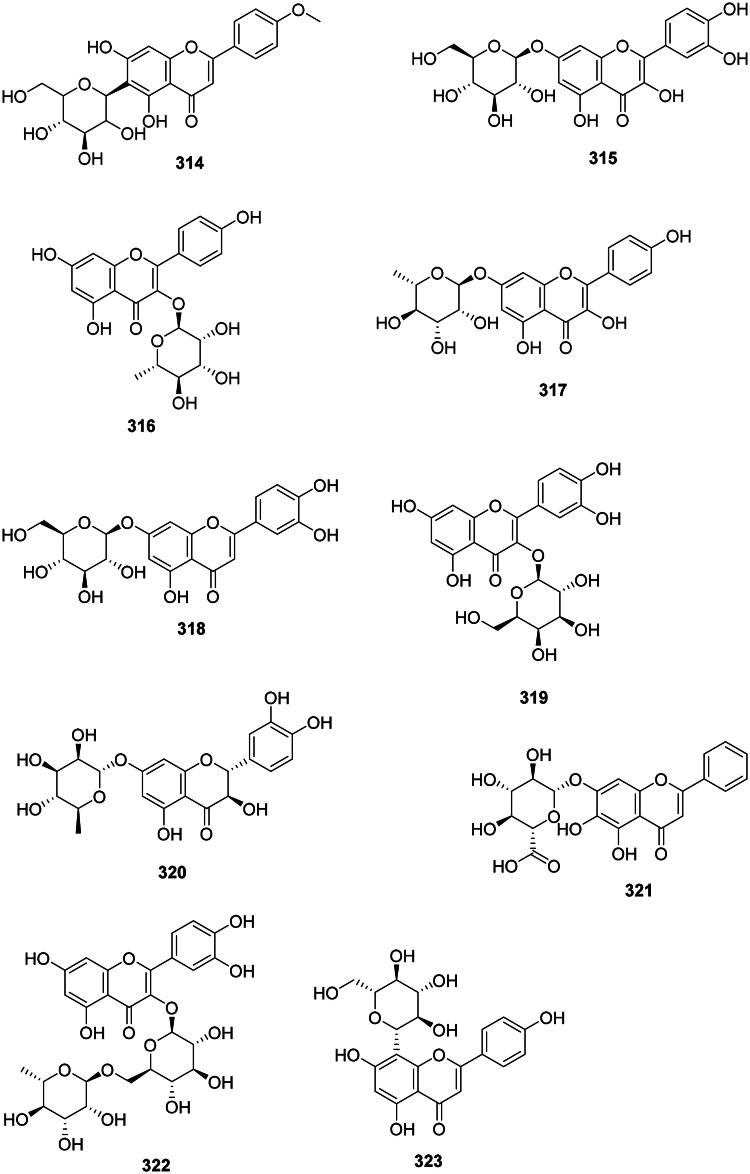
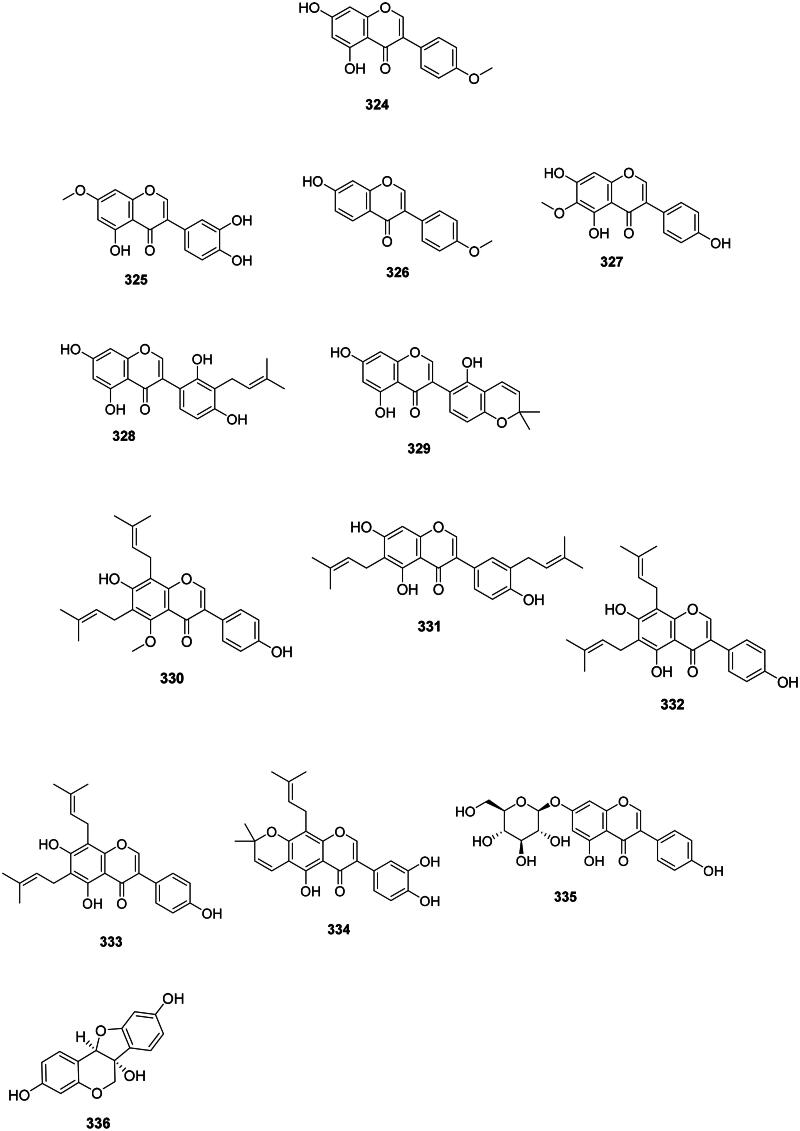
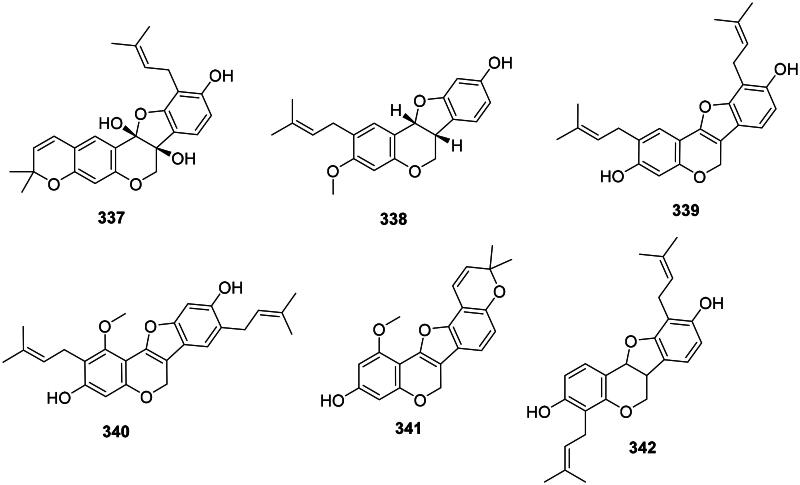
References
- Harpulliasides A and B: two new benzeneacetic acid derivatives from Harpullia pendula.. Phyochem Lett., 2016. [DOI]
- Engineered biosynthesis of plant polyketides: manipulation of chalcone synthase.. Org Lett., 2006. [DOI | PubMed]
- Biosynthesis of medicinally important plant metabolites by unusual type III polyketide synthases.. J Nat Med., 2020. [DOI | PubMed]
- Rosmarinic acid and its methyl ester as antimicrobial components of the hydromethanolic extract of Hyptis atrorubens Poit. (Lamiaceae).. eCAM., 2013. [DOI]
- Intracellular and membrane-damaging activities of methyl gallate from Terminalia chebula against multidrug-resistant Shigellas.. J Med Microbiol., 2015. [DOI | PubMed]
- Antibacterial activity of diospyrin, isodiospyrin and bisisodiospyrin from the root of Diospyros piscatoria (Gurke) (Ebenaceae).. Phytother Res., 2000. [DOI | PubMed]
- Bioactive constituents from Malvastrum coromandelianum (L.) Garcke leaf extracts.. S Afr J Bot., 2019. [DOI]
- Dihydrostilbene phytoalexins from Dioscorea bulbifera and D. dumentorum.. Phytochemistry., 1989. [DOI]
- Antimicrobial constituents of the leaves of Acalypha wilkesiana and Acalypha hispida.. Phytother Res., 2000. [DOI | PubMed]
- Prenylated phenolics as promising candidates for combination antibacterial therapy: Morusin and Kuwanon G.. Saudi Pharm J., 2020. [DOI | PubMed]
- Antibacterial and anti-inflammatory activity of extracts and major constituents derived from Stachytarpheta indica Linn. leaves and their potential implications for wound healing.. Appl Biochem Biotechnol., 2022. [DOI | PubMed]
- Synergistic effect of α-mangostin on antibacterial activity of tetracycline, erythromycin, and clindamycin against acne involved bacteria.. CHM., 2019. [DOI]
- Thymoquinone inhibits Escherichia coli ATP synthase and cell growth.. PLOS One., 2015. [DOI | PubMed]
- Antibacterial activity of the Iraqi Rheum ribes root.. Pharm Biol., 2007. [DOI]
- Antibacterial and anti-hemolytic activity of tannins from Pimenta dioica against methicillin resistant Staphylococcus aureus.. Bangladesh J Pharmacol., 2017. [DOI]
- Antiviral, cyototoxic and antimicrobial activities of anthraquinones from the roots of Morinda elliptica.. Pharm Biol., 2000. [DOI | PubMed]
- Antimicrobial and anti-inflammatory activities of extracts and constituents of Oroxylum indicum (L.) Vent.. Phytomedicine., 1998. [DOI | PubMed]
- Phytochemical, antimicrobial and antiprotozoal evaluation of Garcinia mangostana pericarp and α-mangostin, its major xanthone derivative.. Molecules., 2013. [DOI | PubMed]
- Antibacterial and synergy of a flavanonol rhamnoside with antibiotics against clinical isolates of methicillin-resistant Staphylococcus aureus (MRSA).. Phytomedicine., 2011. [DOI | PubMed]
- Iron-chelation properties of phenolic acids bearing catechol and galloyl groups.. Food Chem., 2006. [DOI]
- Antibacterial cannabinoids from Cannabis sativa: a structure-activity study.. J Nat Prod., 2008. [DOI | PubMed]
- Anti-bacterial properties of cannabigerol toward Streptococcus mutans.. Front Microbiol., 2021. [DOI | PubMed]
- Rapid membrane permeabilization of Listeria monocytogenes and Escherichia coli induced by antibacterial prenylated phenolic compounds from legumes.. Food Chem., 2018. [DOI | PubMed]
- Expression of key genes of the isoflavonoid pathway in hypocotyls and cotyledons during soybean seed maturation.. Crop Sci., 2013. [DOI]
- Molecular characterization of methicillin-resistant Staphylococcus aureus from tertiary care hospitals.. Oak J Med Sci., 2014
- Antibacterial and antibiofilm mechanism of eugenol against antibiotic resistance Vibrio parahaemolyticus.. Food Microbiol., 2020. [DOI | PubMed]
- Phenylpropenes: occurrence, distribution, and biosynthesis in fruit.. J Agric Food Chem., 2016. [DOI]
- Antibacterial tetraoxygenated xanthones from the immature fruits of Garcinia cowa.. Fitoterapia., 2014. [DOI | PubMed]
- The anthraquinone rhein exhibits synergistic antibacterial activity in association with metronidazole or natural compounds and attenuates virulence gene expression in Porphyromonas gingivalis.. Arch Oral Biol., 2015. [DOI | PubMed]
- Antimicrobial compounds from Alpinia conchigera.. J Ethnopharmacol., 2012. [DOI | PubMed]
- Antimicrobial constituents from the rhizomes of Rheum emodi.. Phytochemistry., 2003. [DOI | PubMed]
- Antimicrobial and antimycobacterial activities of methyl caffeate from Solanum torvum Swartz. fruit.. Indian J Microbiol., 2012. [DOI | PubMed]
- Total flavonoid and rutin content of the different organs of Sophora japonica L.. J AOAC Int., 1974. [DOI]
- Activity of 7-methyljuglone in combination with antituberculous drugs against Mycobacterium tuberculosis.. Phytomedicine., 2006. [DOI | PubMed]
- Biochemical and antimicrobial activity of phloretin and its glycosilated derivatives present in apple and kumquat.. Food Chem., 2014. [DOI | PubMed]
- Comprehensive review of antimicrobial activities of plant flavonoids.. Phytochem Rev., 2019. [DOI]
- Bacteriostatic antimicrobial combination: antagonistic interaction between epsilon-viniferin and vancomycin against methicillin-resistant Staphylococcus aureus.. Biomed Res Int., 2014. [DOI | PubMed]
- The pho-controlled outer membrane porin PhoE does not contain specific binding sites for phosphate or polyphosphates.. JBC., 1988. [DOI]
- Morindone, an anthraquinone, intercalates DNA sans toxicity: a spectroscopic and molecular modeling perspective.. Appl Biochem Biotechnol., 2012. [DOI | PubMed]
- [6]-Gingerol exhibits potent anti-mycobacterial and immunomodulatory activity against tuberculosis.. Int Immunopharmacol., 2020. [DOI | PubMed]
- In vitro antituberculosis activities of the constituents from Haloxylon salicornicum.. Bioorg Med Chem Lett., 2010. [DOI | PubMed]
- Can invalid bioactives undermine natural product-based drug discovery?. J Med Chem., 2016. [DOI | PubMed]
- Antibacterial activity of ethanolic extract and compounds from fruits of Tectona grandis (Verbenaceae).. BMC Complement Altern Med., 2015. [DOI | PubMed]
- The antimicrobial potential of cannabidiol.. Commun Biol., 2021. [DOI | PubMed]
- Phenolic compounds from Humulus lupulus as natural antimicrobial products: new weapons in the fight against methicillin resistant Staphylococcus aureus, Leishmania mexicana and Trypanosoma brucei strains.. Molecules., 2019. [DOI | PubMed]
- Anti-Pseudomonas aeruginosa xanthones from the resin and green fruits of Cratoxylum cochinchinense.. Tetrahedron., 2009. [DOI]
- Antibacterial and cytotoxic xanthones from the roots of Cratoxylum formosum.. Phytochemistry., 2006. [DOI | PubMed]
- Regulation of cytoplasmic pH in bacteria.. Microbiol Rev., 1985. [DOI | PubMed]
- Furocoumarin sensitization induces DNA‐protein cross‐links.. Photochem Photobiol., 1993. [DOI | PubMed]
- Chemical constituents of Genista numidica Spach aerial parts and their antimicrobial, antioxidant and antityrosinase activities.. Nat Prod Res., 2019. [DOI | PubMed]
- Cell-specific production and antimicrobial activity of naphthoquinones in roots of Lithospermum erythrorhizon.. Plant Physiol., 1999. [DOI | PubMed]
- Antimicrobial evaluation of the constituents from Macropidia fuliginosa (Hook.) Druce.. Nat Prod Commun., 2019
- Chemovariation and antibacterial activity of extracts and isolated compounds from species of Ixora and Greenea (Ixoroideae, Rubiaceae).. Peer J., 2019. [DOI | PubMed]
- Flavonoids as protective agents against oxidative stress induced by gentamicin in systemic circulation. Potent protective activity and microbial synergism of luteolin.. Food Chem Toxicol., 2018. [DOI | PubMed]
- Antimicrobial activity of isocytisoside and extracts of Aquilegia vulgaris L.. Lett Appl Microbiol., 2004. [DOI | PubMed]
- An update of the angiosperm phylogeny group classification for the orders and families of flowering plants: APG III.. Bot J Linn., 2009. [DOI]
- Antibacterial activity and mechanism of cinnamic acid and chlorogenic acid against Alicyclobacillus acidoterrestris vegetative cells in apple juice.. Int J of Food Sci Tech., 2019. [DOI]
- MYB transcription factors as regulators of secondary metabolism in plants.. Biology., 2020. [DOI | PubMed]
- Study on the mechanism of epigallocatechin gallate (EGCG) to the cell membrane of Escherichia coli.. Sci Adv Mater., 2019. [DOI]
- USA300 methicillin-resistant Staphylococcus aureus, United States, 2000–2013.. EID., 2015
- Ecbolin A: a bioactive compound from the roots of Ecbolium viride (Forssk.) Alston.. Asian J. Pharm. Clin. Res., 2012
- Antimicrobial efficacy of plant phenolic compounds against Salmonella and Escherichia coli.. Food Biosci., 2015. [DOI]
- Antibacterial activity of sophoraflavanone G from the roots of Sophora flavescens.. J Microbiol Biotechnol., 2007. [PubMed]
- Antibacterial activity of sophoraflavanone G from the roots of Sophora flavescens against methicillin‐resistant Staphylococcus aureus.. Phytother Res., 2009. [DOI | PubMed]
- Antibacterial activity of thymoquinone, an active principle of Nigella sativa and its potency to prevent bacterial biofilm formation.. BMC Complement Altern Med., 2011. [DOI | PubMed]
- Caged xanthones: potent inhibitors of global predominant MRSA USA300.. Bioorg Med Chem Lett., 2016. [DOI | PubMed]
- DNA damage and mutation. Types of DNA damage.. BioDiscovery., 2014
- Alkaloid and coumarins from the green fruits of Aegle marmelos.. Phytochemistry., 2012. [DOI | PubMed]
- Exploring the antibacterial potential of semisynthetic phytocannabinoid: tetrahydrocannabidiol (THCBD) as a potential antibacterial agent against sensitive and resistant strains of Staphylococcus aureus.. ACS Infect Dis., 2023. [DOI | PubMed]
- Antitubercular resorcinol analogs and benzenoid C-glucoside from the roots of Ardisia cornudentata.. Planta Med., 2011. [DOI | PubMed]
- Antimicrobial activity of juncusol, a novel 9-10-dihydrophenanthrene from the marsh plant Juncus roemerianus.. Life Sci., 1981. [DOI | PubMed]
- Phenanthrene antibiotic targets bacterial membranes and kills Staphylococcus aureus with a low propensity for resistance development.. Front Microbiol., 2018. [DOI | PubMed]
- Inhibition of Klebsiella pneumoniae DnaB helicase by the flavonol galangin.. Protein J., 2011. [DOI | PubMed]
- New polyisoprenyl benzophenone derivatives and antitubercular constituents from Garcinia multiflora.. Planta Med., 2008. [DOI]
- Inhibition of growth of Streptococcus mutans, methicillin-resistant Staphylococcus aureus, and vancomycin-resistant enterococci by kurarinone, a bioactive flavonoid from Sophora flavescens.. J Clin Microbiol., 2005. [DOI | PubMed]
- In vitro inhibition of human cytochrome P450 enzymes by licoisoflavone B from Glycyrrhiza uralensis Fisch. ex DC.. Toxicol Sci., 2023. [DOI | PubMed]
- Synergistic effect and mechanism of plumbagin with gentamicin against carbapenem-resistant Klebsiella pneumoniae.. Infect Drug Resist., 2020. [DOI | PubMed]
- Vitexin mitigates Staphylococcus aureus-induced mastitis via regulation of ROS/ER stress/NF-κB/MAPK pathway.. Oxid Med Cell Longev., 2022. [DOI]
- Prenylated flavonoids from the stems and roots of Tripterygium wilfordii.. Fitoterapia., 2017. [DOI | PubMed]
- Antimycobacterium activity of coumarins from fruit pulp of Aegle marmelos (L.) Correa.. JCS., 2015
- Antibacterial activity of embelin.. Fitoterapia., 2003. [DOI | PubMed]
- Isolation and antifungal activity of lignans from Myristica fragrans against various plant pathogenic fungi.. Pest Manag Sci., 2007. [DOI | PubMed]
- Antimicrobial activity of 4-hydroxybenzoic acid and trans 4-hydroxycinnamic acid isolated and identified from rice hull.. Biosci Biotechnol Biochem., 1998. [DOI | PubMed]
- Anti-acne activities of pulsaquinone, hydropulsaquinone, and structurally related 1,4-quinone derivatives.. Arch Pharm Res., 2009. [DOI | PubMed]
- In vitro activity of methyl gallate from Galla rhois alone and in combination with ciprofloxacin against clinical isolates of Salmonella.. J Microbiol Biotechnol., 2008. [PubMed]
- Methyl gallate from Galla rhois successfully controls clinical isolates of Salmonella infection in both in vitro and in vivo systems.. PLOS One., 2014. [DOI | PubMed]
- Bioactive flavonoids of the flowers of Butea monosperma.. Chem Pharm Bull., 2009. [DOI]
- New flavanones from the leaves of Cryptocarya chinensis and their antituberculosis activity.. Chem Biodivers., 2011. [DOI | PubMed]
- Anticariogenic activity of macelignan from Myristica fragrans (nutmeg) against Streptococcus mutans.. Phytomedicine., 2006. [DOI | PubMed]
- Enhancing antibiotic activity: a strategy to control Acinetobacter infections.. J Antimicrob Chemother., 2009. [DOI | PubMed]
- Antimicrobial activity of phenolic constituents of Magnolia grandiflora L.. J Pharm Sci., 1981. [DOI | PubMed]
- Antimicrobial activity of juglone.. Phytother Res., 1990. [DOI]
- Phenolic acids, syringaldehyde, and juglone in fruits of different cultivars of Juglans regia L.. J Agric Food Chem., 2005. [DOI | PubMed]
- Antibacterial activity of anthraquinone derivatives from Heterophyllaea pustulata (Rubiaceae).. J Photochem Photobiol B., 2011. [DOI | PubMed]
- Antibacterial activity of chrysophanol isolated from Aloe excelsa (Berger).. AJB., 2006a
- Antibacterial activity of aloe emodin and aloin A from Aloe excelsa.. AJB., 2006b
- Assessment of the antibacterial activity of galangin against 4-quinolone resistant strains of Staphylococcus aureus.. Phytomedicine., 2006. [DOI | PubMed]
- Detection of galangin-induced cytoplasmic membrane damage in Staphylococcus aureus by measuring potassium loss.. J Ethnopharmacol., 2005. [DOI | PubMed]
- Anti-Bacteroides fragilis substance from rhubarb.. J Ethnopharmacol., 1987. [DOI | PubMed]
- Phytochemical screening of methanolic extract and antibacterial activity of active principles of hepatoprotective herb, Eclipta alba.. Ethnobot Leaf., 2010
- Investigation on the dark interaction between furocoumarins and DNA.. Chem Biol Interact., 1978. [DOI | PubMed]
- Vitexin alters Staphylococcus aureus surface hydrophobicity to obstruct biofilm formation.. Microbiol Res., 2022. [DOI | PubMed]
- New coumarin derivatives from Ferula pseudalliacea with antibacterial activity.. Nat Prod Res., 2016. [DOI | PubMed]
- Mode of action of farnesol, the “noble unknown” in particular in Ca2+ homeostasis, and its juvenile hormone-esters in evolutionary retrospect.. Front Neurosci., 2019. [DOI | PubMed]
- C-glycosylflavones from the aerial parts of Eleusine indica inhibit LPS-induced mouse lung inflammation.. Planta Med., 2005. [DOI | PubMed]
- Phenolic compounds from the fruit of Garcinia dulcis.. Phytochemistry., 2005. [DOI | PubMed]
- Antioxidant and antimicrobial constituents of licorice: isolation and structure elucidation of a new benzofuran derivative.. Chem Pharm Bull., 1988. [DOI]
- Chemical constituents from the roots of Ranunculus ternatus and their inhibitory effects on Mycobacterium tuberculosis.. Molecules., 2013. [DOI | PubMed]
- Cellular impermeability and uptake of biocides and antibiotics in gram-negative bacteria.. J Appl Microbiol., 2002. [PubMed]
- Synergistic efficacies of thymoquinone and standard antibiotics against multi-drug resistant isolates.. Saudi Med J., 2021. [DOI | PubMed]
- Antitubercular and antibacterial activity of quinonoid natural products against multi‐drug resistant clinical isolates.. Phytother Res., 2014. [DOI | PubMed]
- Antibacterial activity of xanthones from Garcinia mangostana (L.) and their structure–activity relationship studies.. Nat Prod Res., 2013. [DOI | PubMed]
- Antimicrobial constituents of the mature carpels of Manglietiastrum sinicum.. J Nat Prod., 2014. [DOI | PubMed]
- Antimicrobial and anti-biofilm activity of tannic acid against Staphylococcus aureus.. Nat Prod Res., 2018. [DOI | PubMed]
- A new neolignan glycoside from the leaves of Acer truncatum.. Molecules., 2006. [DOI | PubMed]
- Antibacterial and EGFR-tyrosine kinase inhibitory activities of polyhydroxylated xanthones from Garcinia succifolia.. Molecules., 2014. [DOI | PubMed]
- Preventing microbial infections with natural phenolic compounds.. Future Pharmacol., 2022. [DOI]
- Structures of new phenolics isolated from licorice, and the effectiveness of licorice phenolics on vancomycin-resistant Enterococci.. Molecules., 2014. [DOI | PubMed]
- Reversing β-lactam antibiotic resistance of Staphylococcus aureus with galangin from Alpinia officinarum Hance and synergism with ceftazidime.. Phytomedicine., 2010. [DOI | PubMed]
- Antibacterial activity of East African medicinal plants.. J Ethnopharmacol., 1998. [DOI | PubMed]
- Dihydrostilbene phytoalexins from Dioscorea rotundata.. Phytochemistry., 1987. [DOI]
- Tannins as an alternative to antibiotics.. Food Biosci., 2020. [DOI]
- Toxicity and mutagenicity of plumbagin and the induction of a possible new DNA repair pathway in Escherichia coli.. J Bacteriol., 1985. [DOI | PubMed]
- Antibacterial effect of hypericin.. Afr J Microbiol Res., 2013
- Antibacterial and antibiofilm activities of Cinnamomum essential oil and cinnamaldehyde: antimicrobial activities.. Sci World J., 2018. [DOI]
- The mode of antibacterial action of phenols in relation to drug-fastness.. Trans Faraday Soc., 1945. [DOI]
- Antimicrobial activity of licorice flavonoids against methicillin-resistant Staphylococcus aureus.. Fitoterapia., 2002. [DOI | PubMed]
- Antimicrobial activity of hydrophobic xanthones from Cudrania cochinchinensis against Bacillus subtilis and methicillin‐resistant Staphylococcus aureus.. Chem Biodivers., 2004. [DOI | PubMed]
- Two antimicrobial flavanones from the leaves of Glycyrrhiza glabra.. Chem Pharm Bull., 1988. [DOI]
- α-Methylene-γ-lactones as a novel class of anti-leukemic agents.. Anticancer Agents Med Chem., 2014. [DOI | PubMed]
- Isoflavonoids and coumarins from Glycyrrhiza uralensis: antibacterial activity against oral pathogens and conversion of isoflavans into isoflavan-quinones during purification.. J Nat Prod., 2011. [DOI | PubMed]
- Antimycobacterial activity of DNA intercalator inhibitors of Mycobacterium tuberculosis primase DnaG.. J Antibiot., 2015. [DOI]
- Effect of ellagic acid on some types of pathogenic bacteria.. JNUS., 2010. [DOI]
- Cannabis sativa CBD extract exhibits synergy with broad-spectrum antibiotics against Salmonella enterica subs enterica serovar typhimurium.. Microorganisms., 2022. [DOI | PubMed]
- Antimicrobial activity of the green tea polyphenol (−)-epigallocatechin-3-gallate (EGCG) against clinical isolates of Stenotrophomonas maltophilia.. Int J Antimicrob Agents., 2010. [DOI | PubMed]
- Green tea catechins inhibit bacterial DNA gyrase by interaction with its ATP binding site.. J Med Chem., 2007. [DOI | PubMed]
- Cytotoxic and antimicrobial constituents of the bark of Diospyros maritima collected in two geographical locations in Indonesia.. J Nat Prod., 2004. [DOI | PubMed]
- Antibacterial effects of curcumin: an in vitro minimum inhibitory concentration study.. Toxicol Ind Health., 2016. [DOI | PubMed]
- In vitro activity of isoimperatorin, alone and in combination, against Mycobacterium tuberculosis.. Lett Appl Microbiol., 2014. [DOI | PubMed]
- Antimicrobial potential of Glycyrrhiza glabra roots.. J Ethnopharmacol., 2008. [DOI | PubMed]
- Natural cinnamic acids, synthetic derivatives and hybrids with antimicrobial activity.. Molecules., 2014. [DOI | PubMed]
- Prevalence, antibiogram, and multidrug-resistant profile of E. coli O157: H7 in retail raw beef in Addis Ababa, Ethiopia.. Front Vet Sci., 2022. [DOI | PubMed]
- Mode of antibacterial action of retrochalcones from Glycyrrhiza inflata.. Phytochemistry., 1998. [DOI | PubMed]
- Antimicrobial and cytotoxic activity from Lasia spinosa and isolated lignan.. Lat Am J Pharm., 2011
- Theasinensin A, a tea polyphenol formed from (−)-epigallocatechin gallate, suppresses antibiotic resistance of methicillin-resistant Staphylococcus aureus.. Planta Med., 2003. [DOI | PubMed]
- Phenolic constituents of licorice: structures of glicophenone and glicoisoflavanone, and effects of licorice phenolics on methicillin-resistant Staphylococcus aureus.. Chem Pharm Bull., 2000. [DOI]
- Phenolic constituents of Cassia seed and antibacterial effect of some naphthalenes and anthraquinones on methicillin-resistant Staphylococcus aureus.. Chem Pharm Bull., 1999. [DOI]
- Phytochemical constituents from Cassia alata with inhibition against methicillin-resistant Staphylococcus aureus (MRSA).. Planta Med., 2008. [DOI | PubMed]
- Antibacterial compounds from Glycyrrhiza uralensis.. J Nat Prod., 2006. [DOI | PubMed]
- Synergistic interaction of phenylpropanoids with antibiotics against bacteria.. J Med Microbiol., 2010. [DOI | PubMed]
- Chemical composition of Acacia farnesiana (L) wild fruits and its activity against Mycobacterium tuberculosis and dysentery bacteria.. J Ethnopharmacol., 2019. [DOI | PubMed]
- Strain-induced reactivity effects in the reaction of 2,5-dihydroxy-[1,4]-benzoquinone with diamine.. COC., 2021. [DOI]
- Isolation of 6-, 8-, and 10-gingerol from Ginger rhizome by HPLC and preliminary evaluation of inhibition of Mycobacterium avium and Mycobacterium tuberculosis.. J Agric Food Chem., 1998. [DOI]
- Antimicrobial activity of honokiol and magnolol from Magnolia officinalis.. Phytother Res., 2001. [DOI | PubMed]
- The antibacterial properties of 4,8,4′,8′-tetramethoxy (1,1′-biphenanthrene)-2,7,2′,7′-tetrol from fibrous roots of Bletilla striata.. Indian J Microbiol., 2021. [DOI | PubMed]
- The relationship between structure and in vitro antibacterial activity of selected isoflavones and their metabolites with special focus on antistaphylococcal effect of demethyltexasin.. Lett Appl Microbiol., 2015. [DOI | PubMed]
- Mechanisms underlying synergistic killing of polymyxin b in combination with cannabidiol against Acinetobacter baumannii: a metabolomic study.. Pharmaceutics., 2022. [DOI | PubMed]
- Biosynthetic pathway and metabolic engineering of plant dihydrochalcones.. J Agric Food Chem., 2017. [DOI | PubMed]
- Pterostilbene enhanced anti-methicillin resistant Staphylococcus aureus (MRSA) activity of oxacillin.. Am J Infect Dis., 2016. [DOI]
- Obovatol and obovatal, novel biphenyl ether lignans from the leaves of Magnolia obovata Thunb.. Chem Pharm Bull., 1982. [DOI]
- Covalent modifiers: a chemical perspective on the reactivity of α,β-unsaturated carbonyls with thiols via hetero-Michael addition reactions.. J Med Chem., 2017. [DOI | PubMed]
- Naringin is a promising natural compound for therapy of iron-overload disorders.. Braz J Pharm Sci., 2022
- Activity and interactions of antibiotic and phytochemical combinations against Pseudomonas aeruginosa in vitro.. Int J Biol Sci., 2010. [DOI | PubMed]
- Reversal of tetracycline resistance by cepharanthine, cinchonidine, ellagic acid and propyl gallate in a multi-drug resistant Escherichia coli.. Nat Prod Bioprospect., 2021. [DOI | PubMed]
- Antimicrobial activities of neo-and 1-epineo-isoshinanolones from Plumbago zeylanica roots.. Pharm Biol., 2010. [DOI | PubMed]
- Antibacterial activity of plumbagin and root extracts of Plumbago zeylanica L.. Acta Biol Crac Ser Bot., 2009
- Effect of naturally isolated hydroquinone in disturbing the cell membrane integrity of Pseudomonas aeruginosa MTCC 741 and Staphylococcus aureus MTCC 740.. Heliyon., 2021. [DOI | PubMed]
- Cajanin stilbene acid: a direct inhibitor of colistin resistance protein MCR-1 that restores the efficacy of polymyxin B against resistant Gram-negative bacteria.. Phytomedicine., 2023. [DOI | PubMed]
- Assessing the potential of gallic acid and methyl gallate to enhance the efficacy of β-lactam antibiotics against methicillin-resistant Staphylococcus aureus by targeting β-lactamase: in silico and in vitro studies.. Antibiotics., 2023. [DOI | PubMed]
- The chemical constituents from the active fractions of Eleutherine bulbosa with their antimicrobial activity.. Nat Prod Res., 2020. [DOI | PubMed]
- Antibacterial stilbenes from the tubers of Bletilla striata.. Fitoterapia., 2019. [DOI | PubMed]
- Synergistic effect of rhein in combination with ampicillin or oxacillin against methicillin-resistant Staphylococcus aureus.. Exp Ther Med., 2012. [DOI | PubMed]
- The antibacterial assay of tectorigenin with detergents or ATPase inhibitors against methicillin-resistant Staphylococcus aureus.. Evid Based Complement Alternat Med., 2014. [DOI | PubMed]
- Arbutin and its metabolite hydroquinone as the main factors in the antimicrobial effect of strawberry tree (Arbutus unedo L.) leaves.. J Herb Med., 2017. [DOI]
- Flavonoids and isoflavonoids of chemotaxonomic significance from Glycyrrhiza pallidiflora (Leguminosae).. Biochem Syst Ecol., 1993. [DOI]
- Antimicrobial constituents of peel and seeds of camu-camu (Myrciaria dubia).. Biosci Biotechnol Biochem., 2017. [DOI | PubMed]
- Antibacterial activity of gallic acid against Shigella flexneri and its effect on biofilm formation by repressing mdoH gene expression.. Food Control., 2018. [DOI]
- Antimicrobial properties of Quercus ilex L. proanthocyanidin dimers and simple phenolics: evaluation of their synergistic activity with conventional antimicrobials and prediction of their pharmacokinetic profile.. J Agric Food Chem., 2011. [DOI | PubMed]
- Antimicrobial potential of caffeic acid against Staphylococcus aureus clinical strains.. Biomed Res Int., 2018. [DOI | PubMed]
- Antibacterial compounds from the seeds of Psoralea corylifolia.. Fitoterapia., 2004. [DOI | PubMed]
- Antibacterial effect of sophoraflavanone G by destroying the cell wall of Enterococcus faecium.. J Appl Pharm Sci., 2020
- Nonallergenic urushiol derivatives inhibit the oxidation of unilamellar vesicles and of rat plasma induced by various radical generators.. Free Radic Biol Med., 2014. [DOI | PubMed]
- Synthesis and microbiological evaluation of honokiol derivatives as new antimicrobial agents.. Arch Pharm Res., 2010. [DOI | PubMed]
- Cajanuslactone, a new coumarin with anti-bacterial activity from pigeon pea [Cajanus cajan (L.) Mills] leaves.. Food Chem., 2010. [DOI]
- Anthraquinone and naphthoquinone derivatives from the roots of Coptosapelta flavescens.. Nat Prod Commun., 2014. [PubMed]
- Phenylphenalenone-type phytoalexins in banana (Musa species): a comprehensive review for new research directions.. Phytochem Rev., 2023. [DOI]
- Lianqiaoxinoside B, a novel caffeoyl phenylethanoid glycoside from Forsythia suspensa.. Molecules., 2011. [DOI | PubMed]
- Antibacterial activity of long-chain alcohols against Streptococcus mutans.. J Agric Food Chem., 1993. [DOI]
- Antibacterial action of anacardic acids against methicillin resistant Staphylococcus aureus (MRSA).. J Agric Food Chem., 2003. [DOI | PubMed]
- Antibacterial activities of the extracts, fractions and compounds from Dioscorea bulbifera.. BMC Complement Altern Med., 2012. [DOI | PubMed]
- Efflux pumps are involved in the defense of gram-negative bacteria against the natural products isobavachalcone and diospyrone.. Antimicrob Agents Chemother., 2010. [DOI | PubMed]
- Diospyrone, crassiflorone and plumbagin: three antimycobacterial and antigonorrhoeal naphthoquinones from two Diospyros.. Int J Antimicrob Agents., 2009. [DOI | PubMed]
- Polyphenolic constituent structures of Zanthoxylum piperitum fruit and the antibacterial effects of its polymeric procyanidin on methicillin-resistant Staphylococcus aureus.. Biosci Biotechnol Biochem., 2006. [DOI | PubMed]
- Two stilbenes from Indonesian Cassia grandis and their antibacterial activities.. Res J Chem Environ., 2020
- A new coruleoellagic acid derivative from stems of Rhodamnia dumetorum.. Nat Prod Res., 2018. [DOI | PubMed]
- Acaricidal activity and function of mite indicator using plumbagin and its derivatives from Diospyros kaki Thunb. roots (Ebenaceae).. J Microbiol Biotechnol., 2008. [PubMed]
- Antibacterial and synergistic activity of prenylated chalcone from the roots of Sophora flavescens.. J Korean Soc Appl Biol Chem., 2010. [DOI]
- Antibacterial activity of hibicuslide C on multidrug-resistant Pseudomonas aeruginosa isolates.. Curr Microbiol., 2016. [DOI | PubMed]
- Antibacterial mechanism of (−)-nortrachelogenin in Escherichia coli O157.. Curr Microbiol., 2016. [DOI | PubMed]
- Glochidioboside kills pathogenic bacteria by membrane perturbation.. Curr Microbiol., 2015. [DOI | PubMed]
- Bactericidal effect of pterostilbene alone and in combination with gentamicin against human pathogenic bacteria.. Molecules., 2017. [DOI | PubMed]
- Synergistic effect of emodin in combination with ampicillin or oxacillin against methicillin-resistant Staphylococcus aureus.. Pharm Biol., 2010. [DOI | PubMed]
- The mechanism underlying the antibacterial activity of shikonin against methicillin-resistant Staphylococcus aureus.. eCAM., 2015. [DOI]
- Lignans: biosynthesis and function.. Comprehensive Nat Prod Chem., 1999
- Antibacterial neolignans from the leaves of Melaleuca bracteata.. Fitoterapia., 2017. [DOI | PubMed]
- Punicalagin damages the membrane of Salmonella typhimurium.. J Food Prot., 2020. [DOI | PubMed]
- Antibacterial constituents of Fructus Chebulae immaturus and their mechanisms of action.. BMC Complement Altern Med., 2016. [DOI | PubMed]
- The antibacterial activity and action mechanism of emodin from Polygonum cuspidatum against Haemophilus parasuis vitro.. Microbiol Res., 2016. [DOI]
- Effect of corilagin on membrane permeability of Escherichia coli, Staphylococcus aureus and Candida albicans.. Phytother Res., 2013. [DOI | PubMed]
- Synergistic antibacterial activity with conventional antibiotics and mechanism of action of shikonin against methicillin-resistant Staphylococcus aureus.. Int J Mol Sci., 2022. [DOI | PubMed]
- Antibacterial activity and membrane-targeting mechanism of aloe-emodin against Staphylococcus epidermidis.. Front Microbiol., 2021. [DOI | PubMed]
- (±)-Meliviticines A and B: rearranged prenylated acetophenone derivatives from Melicope viticina and their antimicrobial activity.. Bioorg Chem., 2019. [DOI | PubMed]
- Antibacterial active compounds from Hypericum ascyron L. induce bacterial cell death through apoptosis pathway.. Eur J Med Chem., 2015. [DOI | PubMed]
- Antibacterial dimeric acylphloroglucinols from Hypericum japonicum.. J Nat Prod., 2018. [DOI | PubMed]
- Bioactive compounds from Rumex plants.. Phytochem Lett., 2010. [DOI]
- Antimicrobial and enhancement of the antibiotic activity by phenolic compounds: gallic acid, caffeic acid and pyrogallol.. Microb Pathog., 2016. [DOI | PubMed]
- Antitubercular and antiplasmodial constituents of Abrus precatorius.. Planta Med., 2004. [DOI | PubMed]
- Inhibitory effects of 1,2,3,4,6-penta-O-galloyl-β-d-glucopyranose on biofilm formation by Staphylococcus aureus.. Antimicrob Agents Chemother., 2011. [DOI | PubMed]
- Antitubercular constituents from the roots of Engelhardia roxburghiana.. Planta Med., 2005. [DOI | PubMed]
- In vitro synergy of biochanin A and ciprofloxacin against clinical isolates of Staphylococcus aureus.. Molecules., 2011. [DOI | PubMed]
- In vitro antibacterial activity of roselle calyx and protocatechuic acid.. Phytother Res., 2005. [DOI | PubMed]
- Resveratrol enhances the antimicrobial effect of polymyxin B on Klebsiella pneumoniae and Escherichia coli isolates with polymyxin B resistance.. BMC Microbiol., 2020. [DOI | PubMed]
- Synergy among thymol, eugenol, berberine, cinnamaldehyde and streptomycin against planktonic and biofilm‐associated food‐borne pathogens.. Lett Appl Microbiol., 2015. [DOI | PubMed]
- Antibacterial synergy between linezolid and baicalein against methicillin-resistant Staphylococcus aureus biofilm in vivo.. Microb Pathog., 2020. [DOI | PubMed]
- New mechanism of magnolol and honokiol from Magnolia officinalis against Staphylococcus aureus.. Nat Prod Commun., 2014. [PubMed]
- The mechanism of action of quinone antibiotics.. Mol Cell Biochem., 1983. [DOI | PubMed]
- Baicalein ameliorates Streptococcus suis-induced infection in vitro and in vivo.. Int J Mol Sci., 2021
- Antimicrobial mechanism of hydroquinone.. Appl Biochem Biotechnol., 2019. [DOI | PubMed]
- Anti-bacterial activity of phenolic compounds against Streptococcus pyogenes.. Medicines., 2017. [DOI | PubMed]
- Evaluation of ellagic acid and gallic acid as efflux pump inhibitors in strains of Staphylococcus aureus.. Biol Open., 2022
- A new flavanone from Flemingia strobilifera (Linn) R. Br. and its antimicrobial activity.. Trop J Pharm Res., 2008. [DOI]
- A benzil and isoflavone derivatives from Derris scandens Benth.. Phytochemistry., 2004. [DOI | PubMed]
- Antibacterial and cytotoxic xanthones from Cratoxylum cochinchinense.. Phytochem Lett., 2008. [DOI]
- Activity of 7-methyljuglone derivatives against Mycobacterium tuberculosis and as subversive substrates for mycothiol disulfide reductase.. Bioorg Med Chem., 2007. [DOI | PubMed]
- The variable chemotherapeutic response of malabaricone A in leukemic and solid tumor cell lines depends on the degree of redox imbalance.. Phytomedicine., 2015. [DOI | PubMed]
- Antitubercular activity of compounds from Pelargonium sidoides.. Pharm Biol., 2007. [DOI]
- Antibacterial and antifungal activities of new acylated derivatives of epigallocatechin gallate.. Front Microbiol., 2012. [DOI | PubMed]
- Stilbenoids: a natural arsenal against bacterial pathogens.. Antibiotics., 2020. [DOI | PubMed]
- Antimicrobial activity of resveratrol-derived monomers and dimers against foodborne pathogens.. Sci Re., 2019
- A flavanone and antimicrobial activities of the constituents of extracts from Mundulea sericea.. Nat Prod Res., 2012. [DOI | PubMed]
- Purified compounds and extracts from Euclea species with antimycobacterial activity against Mycobacterium bovis and fast-growing mycobacteria.. Biol Pharm Bull., 2008. [DOI | PubMed]
- Synergistic effect on anti‐methicillin‐resistant Staphylococcus aureus among combinations of α‐mangostin‐rich extract, lawsone methyl ether and ampicillin.. Lett Appl Microbiol., 2020. [DOI | PubMed]
- Antimicrobial activities of some Thai traditional medical longevity formulations from plants and antibacterial compounds from Ficus foveolata.. Pharm Biol., 2014. [DOI | PubMed]
- Bioactive phenolic compounds, metabolism and properties: a review on valuable chemical compounds in Scots pine and Norway spruce.. Phytochem Rev., 2019. [DOI]
- Isolation and characterization of antisalmonellal anthraquinones and coumarins from Morinda lucida Benth. (Rubiaceae).. Chem Pap., 2021. [DOI]
- Antibacterial activity of protocatechuic acid ethyl ester on Staphylococcus aureus clinical strains alone and in combination with antistaphylococcal drugs.. Molecules., 2015. [DOI | PubMed]
- Phenolic compounds diminish antibiotic resistance of Staphylococcus aureus clinical strains.. Int J Environ Res Public Health., 2018
- Antimicrobial agents from higher plants. Antimicrobial isoflavanoids and related substances from Glycyrrhiza glabra L. var. typica.. J Nat Prod., 1980. [DOI | PubMed]
- Antibacterial activities of the extracts, fractions and isolated compounds from Canarium patentinervium Miq. against bacterial clinical isolates.. BMC Complement Med Ther., 2020. [DOI | PubMed]
- Mechanistic study of the antibacterial potential of the prenylated flavonoid auriculasin against Escherichia coli.. Arch Pharm., 2022
- Isolation and identification of antibacterial compounds from Thymus kotschyanus aerial parts and Dianthus caryophyllus flower buds.. Phytomedicine., 2009. [DOI | PubMed]
- Antibacterial activity and mode of action of plant flavonoids against Proteus vulgaris and Staphylococcus aureus.. Phytochemistry., 1987. [DOI]
- Characterization of the genotoxicity of anthraquinones in mammalian cells.. Biochim Biophys Acta., 1999. [DOI | PubMed]
- Punicalagin suppresses methicillin resistance of Staphylococcus aureus to oxacillin.. J Pharmacol Sci., 2018. [DOI | PubMed]
- Chemical constituents from leaves and trunk bark of Rinorea oblongifolia (Violaceae).. Nat Prod Res., 2020. [DOI | PubMed]
- Antimycobacterial flavones from Haplopappus sonorensis.. Fitoterapia., 2003. [DOI | PubMed]
- Antibacterial activity of anacardic acid and totarol, alone and in combination with methicillin against methicillin resistant Staphylococcus aureus.. J Appl Bacteriol., 1996. [DOI | PubMed]
- Synthesis, antimycobacterial evaluation and pharmacophore modeling of analogues of the natural product formononetin.. Bioorg Med Chem Lett., 2015. [DOI | PubMed]
- Antimicrobial compounds, chinesin I and II from flowers of Hypericum chinense L.. Chem Lett., 1987. [DOI]
- Evidence of calcium signaling and modulation of the LmrS multidrug resistant efflux pump activity by Ca2+ ions in S. aureus.. Front Microbiol., 2020. [DOI | PubMed]
- Antifolate activity of epigallocatechin gallate against Stenotrophomonas maltophilia.. Antimicrob Agents Chemother., 2005. [DOI | PubMed]
- The essential medicinal chemistry of curcumin: mini perspective.. J Med Chem., 2017. [DOI | PubMed]
- The FEMA GRAS assessment of trans-anethole used as a flavouring substance.. FCT., 1999. [DOI]
- The evaluation of forty-three plant species for in vitro antimycobacterial activities; isolation of active constituents from Psoralea corylifolia and Sanguinaria canadensis.. J Ethnopharmacol., 2002. [DOI | PubMed]
- Antimicrobial constituents from leaves of Dolichandrone spathacea and their relevance to traditional use.. Planta Med., 2018
- Antimicrobial actions of α-mangostin against oral streptococci.. Can J Microbiol., 2011. [DOI | PubMed]
- Death from pan-resistant superbug.. Autops Case Rep., 2019. [DOI | PubMed]
- Outer membrane protein A (OmpA) as a potential therapeutic target for Acinetobacter baumannii infection.. J Biomed Sci., 2020. [DOI | PubMed]
- Antimicrobial lignans derived from the roots of Streblus asper.. Phytochem Lett., 2016. [DOI]
- Mycobacterial porins–new channel proteins in unique outer membranes.. Mol Microbiol., 2003. [DOI | PubMed]
- 2,6-Dimethoxy-p-benzoquinone as an antibacterial substance in the bark of Phyllostachys heterocycla var. pubescens, a species of thick-stemmed bamboo.. J Agric Food Chem., 1991. [DOI]
- Antimicrobial components, trachrysone and 2-methoxystypandrone, in Rumex japonicus Houtt.. J Agric Food Chem., 1993. [DOI]
- Hydroxybenzoquinones from Myrsinaceae plants. III. The structures of 2-hydroxy-5-methoxy-3-pentadecenylbenzoquinone and ardisiaquinones A, B and C from Ardisia spp.. Chem Pharm Bull., 1968. [DOI]
- Detection of antifungal activity in Belamcanda chinensis by a single-cell bioassay method and isolation of its active compound, tectorigenin.. Biosci Biotechnol Biochem., 2001. [DOI | PubMed]
- DNA gyrase inhibitory and antibacterial activity of some flavones.. Bioorg Med Chem Lett., 1993. [DOI]
- Antibacterial activities and structure–activity relationships of a panel of 48 compounds from Kenyan plants against multidrug resistant phenotypes.. SpringerPlus., 2016. [DOI | PubMed]
- Isolation and characterization of two antimicrobial agents from mace (Myristica fragrans).. J Nat Prod., 1991. [DOI | PubMed]
- Pharmacological screening of Polygonaceae species and isolation of biologically active compounds from Rumex aquaticus L. and Rumex thyrsiflorus Fingerh [doctoral dissertation].. 2017
- Antibacterial, antifungal, and antiviral activities of some flavonoids.. Microbiol Res., 2010. [DOI | PubMed]
- Antibacterial from Hypericum acmosepalum showing inhibition of ATP dependent MurE ligase from Mycobacterium tuberculosis.. Planta Med., 2010. [DOI]
- An antibacterial from Hypericum acmosepalum inhibits ATP-dependent MurE ligase from Mycobacterium tuberculosis.. Int J Antimicrob Agents., 2012. [DOI | PubMed]
- Gallic acid and hydrolysable tannins are formed in birch leaves from an intermediate compound of the shikimate pathway.. Biochem Syst Ecol., 2003. [DOI]
- Highly antibiotic‐resistant Acinetobacter baumannii clinical isolates are killed by the green tea polyphenol (−)‐epigallocatechin‐3‐gallate (EGCG).. Clin Microbiol Infect., 2009. [DOI | PubMed]
- Novel R-plasmid conjugal transfer inhibitory and antibacterial activities of phenolic compounds from Mallotus philippensis (Lam.).. J Glob Antimicrob Resist., 2016. [DOI | PubMed]
- Protein kinase C-δ inhibitor, rottlerin inhibits growth and survival of mycobacteria exclusively through shikimate kinase.. Biochem Biophys Res Commun., 2016. [DOI | PubMed]
- Kuwanon G: an antibacterial agent from the root bark of Morus alba against oral pathogens.. J Ethnopharmacol., 2003. [DOI | PubMed]
- Antibacterial activity of [10]‐gingerol and [12]‐gingerol from ginger rhizome against periodontal bacteria.. Phytother Res., 2008. [DOI | PubMed]
- Xanthones from Garcinia cowa flowers and their cytotoxicity.. Thai J Pharm Sci., 2016
- Antimicrobial activity and effects of resveratrol on human pathogenic bacteria.. World J Microbiol Biotechnol., 2010. [DOI]
- In vitro antibacterial activity of plumbagin from Plumbago zeylanica L. against methicillin‐resistant Staphylococcus aureus.. Lett Appl Microbiol., 2019. [DOI | PubMed]
- Anti-infective potential of caffeic acid and epicatechin 3-gallate isolated from methanol extract of Euphorbia hirta (L.) against Pseudomonas aeruginosa.. Nat Prod Res., 2015. [DOI | PubMed]
- Anti-Salmonella activity of constituents of Ardisia elliptica Thunb.. Nat Prod Res., 2006. [DOI | PubMed]
- Antibacterial activity of essential oils and their active components from Thai spices against foodborne pathogens.. Sci., 2013. [DOI]
- Cytotoxic coumarins from Toddalia asiatica.. Planta Med., 2014. [DOI | PubMed]
- Functional expression and subcellular localization of pea polymorphic isoflavone synthase CYP93C18.. Biologia Plant., 2013. [DOI]
- Natural and synthetic compounds such as trimethoprim behave as inhibitors of efflux in gram-negative bacteria.. J Antimicrob Chemother., 2010. [DOI | PubMed]
- The antivirulence compound myricetin possesses remarkable synergistic effect with antibacterials upon multidrug resistant Staphylococcus aureus.. Microb Pathog., 2020. [DOI | PubMed]
- Inhibition of topoisomerase I by naphthoquinone derivatives.. Bioorg Med Chem Lett., 1998. [DOI | PubMed]
- Bioactive benzoquinones content variability in red-berry and white-berry varieties of Ardisia crenata Sims. and assessment of cytotoxic activity.. Nat Prod Res., 2021. [DOI | PubMed]
- The isoflavone biochanin A inhibits the growth of the intracellular bacteria Chlamydia trachomatis and Chlamydia pneumoniae.. Planta Med., 2012. [DOI]
- Search for antimicrobial activity among fifty-two natural and synthetic compounds identifies anthraquinone and polyacetylene classes that inhibit Mycobacterium tuberculosis.. Front Microbiol., 2020. [DOI | PubMed]
- Antimicrobial constituents of Gomphrena martiana and Gomphrena boliviana.. J Ethnopharmacol., 1992. [DOI | PubMed]
- Electrochemical study of the increased antioxidant capacity of flavonoids through complexation with iron (II) ions.. Electrochim Acta., 2014. [DOI]
- Purification and identification of antibacterial compounds from Euclea crispa subs crispa (Ebenaceae) leaves.. S Afr J Bot., 2003. [DOI]
- Antibacterial and antioxidative compounds from Cassia alata Linn.. Songklanakarin J Sci Technol., 2014
- Lakoochins A and B, new antimycobacterial stilbene derivatives from Artocarpus lakoocha.. J Nat Prod., 2004. [DOI | PubMed]
- Antibacterial biphenanthrenes from the fibrous roots of Bletilla striata.. J Nat Prod., 2015. [DOI | PubMed]
- Identification, molecular cloning, and functional characterization of a coniferyl alcohol acyltransferase involved in the biosynthesis of dibenzocyclooctadiene lignans in Schisandra chinensis.. Front Plant Sci., 2022. [DOI | PubMed]
- Antioxidant and antibacterial activity of two compounds (forsythiaside and forsythin) from Forsythia suspensa.. J Pharm Pharmacol., 2008. [DOI | PubMed]
- A potential antibacterial agent embelin a natural benzoquinone extracted from Embelia ribes.. Biol Med., 2011
- Biologically active cannabinoids from high-potency Cannabis sativa.. J Nat Prod., 2009. [DOI | PubMed]
- Antimicrobial constituents from the stem bark of Feronia limonia.. Phytochemistry., 2002. [DOI | PubMed]
- Antimicrobial activity of ulopterol from Toddalia asiatica (L.) Lam.: a traditional medicinal plant.. J Ethnopharmacol., 2012. [DOI | PubMed]
- Antibacterial and antiviral naphthazarins from Maharanga bicolor.. Pharmazie., 2007. [PubMed]
- A tocotrienol quinone dimer and xanthones from the leaf extract of Garcinia nigrolineata.. Fitoterapia., 2019. [DOI | PubMed]
- Isolation and characterization of a potent antimicrobial compound from Aerva sanguinolenta Blume: an alternative source of bakuchiol.. J Pharm Res., 2012
- Synergistic activity of lupinifolin in combinations with antibiotics against Staphylococcus aureus.. Pak J Biol Sci., 2021. [DOI | PubMed]
- Meso-dihydroguaiaretic acid derivatives with antibacterial and antimycobacterial activity.. Bioorg Med Chem., 2017. [DOI | PubMed]
- Medicinal plants and antimicrobial activity.. J Ethnopharmacol., 2005. [DOI | PubMed]
- In vitro combinatory antimicrobial effect of plumbagin with oxacillin and tetracycline against Staphylococcus aureus.. Phytother Res., 2015. [DOI | PubMed]
- (E)-Methylisoeugenol and elemicin: antibacterial components of Daucus carota L. essential oil against Campylobacter jejuni.. J Agric Food Chem., 2007. [DOI | PubMed]
- Antibacterial xanthones from the leaves of Garcinia nigrolineata.. J Nat Prod., 2003. [DOI | PubMed]
- Antibacterial caged-tetraprenylated xanthones from the stem bark of Garcinia scortechinii.. Planta Med., 2005. [DOI | PubMed]
- Benzopyran, biphenyl, and tetraoxygenated xanthone derivatives from the twigs of Garcinia nigrolineata.. J Nat Prod., 2005. [DOI | PubMed]
- The inhibitory effect of fluphenazinedecanoate and caffeine on Staphylococcus aureus efflux pumps.. CRMB., 2018
- Development of new and validated HPTLC methods for the qualitative and quantitative analysis of hyperforin, hypericin and hyperoside contents in Hypericum species.. Phytochem Anal., 2022. [DOI | PubMed]
- Antibacterial anacardic acid derivatives.. ACS Infect Dis., 2020. [DOI | PubMed]
- The novel antibiotic rhodomyrtone traps membrane proteins in vesicles with increased fluidity.. PLOS Pathog., 2018. [DOI | PubMed]
- Antibacterial and cytotoxic potencies of stilbene oligomers from stem barks of baoti (Dryobalanops lanceolata) growing in Kendari, Indonesia.. Asian J Pharm Clin Res., 2017. [DOI]
- A new butenolide cinnamate and other biological active chemical constituents from Polygonum glabrum.. Nat Prod Res., 2015. [DOI | PubMed]
- Antibacterial activity of α-mangostin against vancomycin-resistant enterococci (VRE) and synergism with antibiotics.. Phytomedicine., 2005. [DOI | PubMed]
- Anti‐MRSA activity of sophoraflavanone G and synergism with other antibacterial agents.. Lett Appl Microbiol., 1998. [DOI | PubMed]
- Plant phenolic antioxidant and prooxidant activities: phenolics-induced oxidative damage mediated by metals in plants.. Toxicology., 2002. [DOI | PubMed]
- Antibacterial activity of Thai edible plants against gastrointestinal pathogenic bacteria and isolation of a new broad spectrum antibacterial polyisoprenylated benzophenone, chamuangone.. Food Chem., 2012. [DOI]
- Sampietro DA, Belizán MM, Apud GR, Juarez JH, Vattuone MA, Catalán CA. 2013. Alkylresorcinols: chemical properties, methods of analysis and potential uses in food, industry and plant protection. In: Céspedes CL, Seigler DS, Sampietro DA, editors. Natural antioxidants and biocides from wild medicinal plants. Wallingford: CABI; p. 148–166.
- Isolation, characterization and mode of antimicrobial action against Vibrio cholerae of methyl gallate from Acacia farnesiana.. J Appl Microbiol., 2013. [DOI | PubMed]
- Antiparasitic activity of biochanin A, an isolated isoflavone from fruits of Cassia fistula (Leguminosae).. Parasitol Res., 2009. [DOI | PubMed]
- Essences in metabolic engineering of lignan biosynthesis.. Metabolites., 2015. [DOI | PubMed]
- New antifungal flavonoid glycoside from Vitex negundo.. Bioorg Med Chem Lett., 2007. [DOI | PubMed]
- Antibacterial property of isoflavonoids from Erythrina variegata against cariogenic oral bacteria.. Phytomedicine., 2003. [DOI | PubMed]
- Antibacterial activity of phytochemicals from Erythrina zeyheri against vancomycin‐resistant Enterococci and their combinations with vancomycin.. Phytother Res., 2004. [DOI | PubMed]
- Chlorochimaphilin: a new antibiotic from Moneses uniflora.. J Nat Prod., 1996. [DOI | PubMed]
- Antimicrobial constituents of Rhus glabra.. J Ethnopharmacol., 1994. [DOI | PubMed]
- Antileptospiral activity of xanthones from Garcinia mangostana and synergy of gamma-mangostin with penicillin G.. BMC Complement Altern Med., 2013. [DOI | PubMed]
- Antibacterial assay-guided isolation of active compounds from Artocarpus heterophyllus heartwoods.. Pharm Biol., 2015. [DOI | PubMed]
- Antibacterial activity of artocarpanone from Artocarpus heterophyllus heartwoods against diarrheal pathogens and its mechanism of action on membrane permeability.. J Appl Pharm Sci., 2017
- Artocarpin isolated from Artocarpus heterophyllus heartwoods alters membrane permeability of Streptococcus mutans.. J Appl Pharma Sci., 2018
- A synergy interaction of artocarpin and tetracycline against Pseudomonas aeruginosa and its mechanism of action on membrane permeability.. Z Naturforsch C J Biosci., 2022. [DOI | PubMed]
- A synergistic effect of artocarpanone from Artocarpus heterophyllus L. (Moraceae) on the antibacterial activity of selected antibiotics and cell membrane permeability.. J Intercul Ethnopharmacol., 2017
- Tannins: current knowledge of food sources, intake, bioavailability and biological effects.. Mol Nutr Food Res., 2009. [PubMed]
- Flavonoids in agriculture: chemistry and roles in, biotic and abiotic stress responses, and microbial associations.. Agronomy., 2020
- Antimicrobial activities of naphthazarins from Arnebia euchroma.. J Nat Prod., 2002. [DOI | PubMed]
- Biosynthesis, total synthesis, and pharmacological activities of aryltetralin-type lignan podophyllotoxin and its derivatives.. Nat Prod Rep., 2022. [DOI | PubMed]
- Assessment of the antibacterial mechanism of pterostilbene against Bacillus cereus through apoptosis-like cell death and evaluation of its beneficial effects on the gut microbiota.. J Agric Food Chem., 2021. [DOI | PubMed]
- 2-Oxoglutarate-dependent dioxygenases in the biosynthesis of simple coumarins.. Front Plant Sci., 2014. [DOI | PubMed]
- Marked potentiation of activity of β-lactams against methicillin-resistant Staphylococcus aureus by corilagin.. Antimicrob Agents Chemother., 2001. [DOI | PubMed]
- Structures and antibacterial properties of isorugosins H–J, oligomeric ellagitannins from Liquidambar formosana with characteristic bridging groups between sugar moieties.. J Nat Prod., 2017. [DOI | PubMed]
- Antibacterial acylphloroglucinols from Hypericum olympicum.. J Nat Prod., 2012. [DOI | PubMed]
- A new labdane-type diterpenoid from the leaves of Vitex negundo L.. Nat Prod Res., 2021. [DOI | PubMed]
- Umbelliferone–an antioxidant from Acacia nilotica (L.) Willd. ex.. Del. Food Chem., 2010. [DOI]
- Antibacterial dihydrobenzopyran and xanthone derivatives from Garcinia cowa stem barks.. Fitoterapia., 2012. [DOI | PubMed]
- Synergy and mode of action of ceftazidime plus quercetin or luteolin on Streptococcus pyogenes.. Evid Based Complement Alternat Med., 2015. [DOI | PubMed]
- Naturally derived malabaricone B as a promising bactericidal candidate targeting multidrug-resistant Staphylococcus aureus also possess synergistic interactions with clinical antibiotics.. Antibiotics., 2023. [DOI | PubMed]
- Deciphering the antibacterial mode of action of alpha-mangostin on Staphylococcus epidermidis RP62A through an integrated transcriptomic and proteomic approach.. Front Microbiol., 2019. [DOI | PubMed]
- Antimycobacterial effect of extract and its components from Rheum rhaponticum.. J AOAC Int., 2013. [DOI | PubMed]
- Tracking leading anti-Candida compounds in plant samples; Plumbago europaea.. Iran J Microbiol., 2018. [PubMed]
- Antimicrobial and cytotoxic activity of 18 prenylated flavonoids from medicinal plants: Morus alba L, Morus mongolica Schneider, Broussonetia papyrifera (L.) Vent, Sophora flavescens Ait and Echinosophora koreensis Nakai.. Phytomedicine., 2004. [DOI | PubMed]
- Anti-inflammatory and antimicrobial coumarins from the stems of Eurya chinensis.. J Asian Nat Prod Res., 2017. [DOI | PubMed]
- Inhibitory effects of reserpine against efflux pump activity of antibiotic resistance bacteria.. Chem Biol Lett., 2017
- Isolation and identification of antibacterial and cytotoxic compounds from the leaves of Muntingia calabura L.. J Ethnopharmacol., 2013. [DOI | PubMed]
- Antibacterial caged-tetraprenylated xanthones from the fruits of Garcinia hanburyi.. Chem Pharm Bull., 2005. [DOI]
- Antimycobacterial activity of prenylated xanthones from the fruits of Garcinia mangostana.. Chem Pharm Bull., 2003. [DOI]
- Therapeutic potential of phenolic compounds in medicinal plants—natural health products for human health.. Molecules., 2023. [DOI]
- Synergism of sophoraflavanone G with norfloxacin against effluxing antibiotic-resistant Staphylococcus aureus.. Int J Antimicrob Agents., 2020. [DOI | PubMed]
- Isolation and characterization of catechol derivatives from Semecarpus anacardium seeds and their antibacterial potential in in vitro.. Biomed Prev Nutr., 2014. [DOI]
- Coumarins and carbazoles from Clausena excavata exhibited antimycobacterial and antifungal activities.. Planta Med., 2003. [DOI | PubMed]
- Antimicrobial and cytotoxic activities of neolignans from Magnolia officinalis.. Chem Biodivers., 2004. [DOI | PubMed]
- Antimicrobial activities of flavonoid glycosides from Graptophyllum grandulosum and their mechanism of antibacterial action.. BMC Complement Altern Med., 2018. [DOI | PubMed]
- Antibacterial spectrum of plant polyphenols and extracts depending upon hydroxyphenyl structure.. Biol Pharm Bull., 2006. [DOI | PubMed]
- Antifungal compounds from Dioscorea batatas inoculated with Pseudomonas cichorii.. Phytochemistry., 1987. [DOI]
- Narrow therapeutic index drugs: a clinical pharmacological consideration to flecainide.. Eur J Clin Pharmacol., 2015. [DOI | PubMed]
- Antibacterial activities of prenylated coumarins from the roots of Prangos hulusii.. Molecules., 2017. [DOI | PubMed]
- Synergistic effect of chlorogenic acid and levofloxacin against Klebsiella pneumonia infection in vitro and in vivo.. Sci Rep., 2020. [DOI | PubMed]
- Cajanin stilbene acid inhibited vancomycin-resistant enterococcus by inhibiting phosphotransferase system.. Front Pharmacol., 2020. [DOI | PubMed]
- Two new isoflavanones from the roots of Erythrina variegata.. Nat Prod Commun., 2015. [PubMed]
- Antibacterial activity of isoflavonoids from Erythrina variegata against methicillin‐resistant Staphylococcus aureus.. Lett Appl Microbiol., 2002. [DOI | PubMed]
- Hypatulins A and B, meroterpenes from Hypericum patulum.. Org Lett., 2016. [DOI | PubMed]
- Antibacterial compounds of licorice against upper airway respiratory tract pathogens.. J Nutr Sci Vitaminol., 2001. [DOI | PubMed]
- Tankeo SB, Kuete V. 2023. African plants acting on Pseudomonas aeruginosa: cut-off points for the antipseudomonal agents from plants. In: Kuete V, Jacquot JP, editors. Advances in botanical research. Cambridge (MA): Academic Press; p. 337–412.
- Antimicrobial and antioxidant activity of kaempferol rhamnoside derivatives from Bryophyllum pinnatum.. BMC Res Notes., 2012. [DOI | PubMed]
- Recent advances in research on lignans and neolignans.. Nat Prod Rep., 2016. [DOI | PubMed]
- Erybraedin C, a natural compound from the plant Bituminaria bituminosa, inhibits both the cleavage and relegation activities of human topoisomerase I.. Biochem J., 2010. [DOI | PubMed]
- Prenylated xanthones from the stem bark of Garcinia dulcis.. Phytochem Lett., 2017. [DOI]
- Tannic acid affects the phenotype of Staphylococcus aureus resistant to tetracycline and erythromycin by inhibition of efflux pumps.. Bioorg Chem., 2017. [DOI | PubMed]
- Benzophenone and xanthone derivatives from the inflorescences of Garcinia cowa.. Arch Pharm Res., 2012. [DOI | PubMed]
- Chemical constituents from Xylosma longifolia and their anti-tubercular activity.. Phytochem Lett., 2011. [DOI]
- Escherichia coli DNA topoisomerase I is a zinc metalloprotein with three repetitive zinc-binding domains.. JBC., 1988. [DOI]
- Reduction of membrane fluidity by antibacterial sophoraflavanone G from Sophora exigua.. Phytomedicine., 2000. [DOI | PubMed]
- Antibacterial activity of licochalcone A against spore-forming bacteria.. Antimicrob Agents Chemother., 2002. [DOI | PubMed]
- Naphthoquinones from Diospyros anisandra exhibit potent activity against pan-resistant first-line drugs Mycobacterium tuberculosis strains.. Pulm Pharmacol Ther., 2014. [DOI | PubMed]
- Bioenhancing and antimycobacterial agents from Ammannia multiflora.. Planta Med., 2012. [DOI | PubMed]
- Impact of environmental factors on stilbene biosynthesis.. Plants., 2021. [DOI | PubMed]
- Going forward laterally: transmembrane passage of hydrophobic molecules through protein channel walls.. ChemBioChem., 2010. [DOI | PubMed]
- Two classes of plant antibiotics: phytoalexins versus “phytoanticipins”.. Plant Cell., 1994. [DOI | PubMed]
- Phenolic compounds: their journey after intake.. Food Funct., 2014. [DOI | PubMed]
- 4-Alkyl-and 4-phenylcoumarins from Mesua ferrea as promising multidrug resistant antibacterials.. Phytochemistry., 2004. [DOI | PubMed]
- Pyrano-isoflavans from Glycyrrhiza uralensis with antibacterial activity against Streptococcus mutans and Porphyromonas gingivalis.. J Nat Prod., 2014. [DOI | PubMed]
- Biologically-active flavonoids from Gossypium arboreum.. Phytochemistry., 1984. [DOI]
- Indicanines B and C, two isoflavonoid derivatives from the root bark of Erythrina indica.. Phytochemistry., 2000. [DOI | PubMed]
- Furocoumarin‐photosensitized hydroxylation of guanosine in RNA and DNA.. Photochem Photobiol., 1995. [DOI | PubMed]
- Antibacterial mechanism of fraxetin against Staphylococcus aureus.. Mol Med Rep., 2014. [DOI | PubMed]
- Review of lignans from 2019 to 2021: newly reported compounds, diverse activities, structure-activity relationships and clinical applications.. Phytochemistry., 2022. [DOI | PubMed]
- Persimmon oligomeric proanthocyanidins exert antibacterial activity through damaging the cell membrane and disrupting the energy metabolism of Staphylococcus aureus.. ACS Food Sci Technol., 2020. [DOI]
- Isolation and biological activity of natural chalcones based on antibacterial mechanism classification.. Bioorg Med Chem., 2023. [DOI | PubMed]
- Plumbagin resurrect colistin susceptible against colistin-resistant Pseudomonas aeruginosa in vitro and in vivo.. Front Microbiol., 2022. [DOI | PubMed]
- In vitro activities of enantiopure and racemic 1′-acetoxychavicol acetate against clinical isolates of Mycobacterium tuberculosis.. Sci Pharm., 2017. [DOI | PubMed]
- Cannabidiol is an effective helper compound in combination with bacitracin to kill gram-positive bacteria.. Sci Rep., 2020. [DOI | PubMed]
- Host-pathogen interactions: XXIII. The mechanism of the antibacterial action of glycinol, a pterocarpan phytoalexin synthesized by soybeans.. Plant Physiol., 1983. [DOI | PubMed]
- Antimicrobial activities of lavandulylated flavonoids in Sophora flavences against methicillin-resistant Staphylococcus aureus via membrane disruption.. J Adv Res., 2023. [DOI | PubMed]
- Cytotoxic and antibacterial properties of resveratrol oligomers from the stem bark of Dryobalanops rappa.. Nat Prod J., 2022
- Oligostilbenoids from the stem bark of Dryobalanops aromatica.. Planta Med., 2011. [DOI]
- Biosynthesis and molecular actions of specialized 1,4-naphthoquinone natural products produced by horticultural plants.. Hortic Res., 2016. [DOI | PubMed]
- The drug-resistant bacteria that pose the greatest health threats.. Nature., 2017. [DOI | PubMed]
- Nucleophilic additions to 4,4-disubstituted 2,5-cyclohexadienones: can dipole effects control facial selectivity?. Chem Rev., 1999. [DOI | PubMed]
- Inhibitory effects on bacterial growth and b‐ketoacyl‐ACP reductase by different species of maple leaf extracts and tannic acid.. Phytother Res., 2010. [DOI | PubMed]
- Secondary metabolites from the stems of Engelhardia roxburghiana and their antitubercular activities.. Phytochemistry., 2012. [DOI | PubMed]
- Baicalin protects mice against Salmonella typhimurium infection via the modulation of both bacterial virulence and host response.. Phytomedicine., 2018. [DOI | PubMed]
- Antibacterial effect and mode of action of flavonoids from licorice against methicillin-resistant Staphylococcus aureus.. Front Microbiol., 2019. [DOI | PubMed]
- Structure-function relationship of acyl amino acid surfactants: surface activity and antimicrobial properties.. J Agric Food Chem., 1995. [DOI]
- Antimicrobial anthraquinones from Morinda angustifolia.. Fitoterapia., 2008. [DOI | PubMed]
- Callistemenonone A, a novel dearomatic dibenzofuran-type acylphloroglucinol with antimicrobial activity from Callistemon viminalis.. Sci Rep., 2017. [DOI | PubMed]
- Antibacterial activity of the Chinese traditional medicine, Zi Hua Di Ding.. Phytother Res., 2004. [DOI | PubMed]
- Screening and identification of the antibacterial bioactive compounds from Lonicera japonica Thunb. leaves.. Food Chem., 2013. [DOI | PubMed]
- The antibacterial principle of Caesalpina sappan.. Phytother Res., 2004. [DOI | PubMed]
- The antibacterial mechanism of carvacrol and thymol against Escherichia coli.. Lett Appl Microbiol., 2008. [DOI | PubMed]
- Chemical constituents from the rhizomes of Smilax glabra and their antimicrobial activity.. Molecules., 2013. [DOI | PubMed]
- New flavonoid glycosides from Sedum aizoon L.. Fitoterapia., 2015. [DOI | PubMed]
- Antimicrobial activity of punicalagin against Staphylococcus aureus and its effect on biofilm formation.. Foodborne Pathog Dis., 2017. [DOI | PubMed]
- Cytotoxic and antimicrobial xanthones from Cratoxylum arborescens (Guttiferae).. MJS., 2013. [DOI]
- Identification of kaempferol from the Leaves of Diospyros kaki and its antimicrobial activity against Streptococcus mutans.. Biocontrol Sci., 1999. [DOI]
- The effect of emodin on Staphylococcus aureus strains in planktonic form and biofilm formation in vitro.. Arch Microbiol., 2017. [DOI | PubMed]
- Chemical constituents and antimicrobial activities of Canthium horridum.. Nat Prod Commun., 2010. [PubMed]
- Study on the inhibitory activity, in vitro, of baicalein and baicalin against skin fungi and bacteria.. Zhong Yao Cai., 2000. [PubMed]
- Antimicrobial and cytotoxic juglones from the immature exocarps of Juglans mandshurica.. Nat Prod Res., 2019. [DOI | PubMed]
- Isolation of an antimicrobial compound from Impatiens balsamina L. using bioassay‐guided fractionation.. Phytother Res., 2001. [DOI | PubMed]
- The antimicrobial activity of compounds from the leaf and stem of Vitis amurensis against two oral pathogens.. Bioorg Med Chem Lett., 2010. [DOI | PubMed]
- Antimicrobial and cytotoxic agents from Calophyllum inophyllum.. Phytochemistry., 2004. [DOI | PubMed]
- Antibacterial prenylflavone derivatives from Psoralea corylifolia, and their structure–activity relationship study.. Bioorg Med Chem., 2004. [DOI | PubMed]
- Nature’s assembly line: biosynthesis of simple phenylpropanoids and polyketides.. Plant J., 2008. [DOI | PubMed]
- Antibacterial activity and mechanism of plant flavonoids to gram-positive bacteria predicted from their lipophilicities.. Sci Rep., 2021. [DOI | PubMed]
- Antibacterial mechanism of thymol to methicillin-resistant Staphylococcus aureus.. J South China Agric Univ., 2018
- Lupinifolin from Derris reticulata possesses bactericidal activity on Staphylococcus aureus by disrupting bacterial cell membrane.. J Nat Med., 2017. [DOI | PubMed]
- Treatment of gram-negative bacterial infections by potentiation of antibiotics.. Curr Opin Microbiol., 2016. [DOI | PubMed]
- Knerachelins A and B, antibacterial phenylacylphenols from Knema furfuracea.. J Nat Prod., 1993. [DOI | PubMed]
- Antimicrobial and cytotoxic phenolics and phenolic glycosides from Sargentodoxa cuneata.. Fitoterapia., 2015. [DOI | PubMed]
- The antibacterial efficacy of an aceraceous plant [Shantung maple (Acer truncatum Bunge)] may be related to inhibition of bacterial β‐oxoacyl‐acyl carrier protein reductase (FabG).. Biotechnol Appl Biochem., 2008. [DOI | PubMed]
- Comparative transcriptomic and proteomic analysis of the antibacterial activity of emodin on Aeromonas hydrophila.. Aquaculture., 2020. [DOI]
- Membrane-damage antibacterial mechanism of phenanthrene compounds from Arundina gramnifolia (D. Don) Hochr.. S Afr J Bot., 2022. [DOI]
- A specialized flavone biosynthetic pathway has evolved in the medicinal plant, Scutellaria baicalensis.. Sci Adv., 2016. [DOI | PubMed]
- In vitro and in vivo synergistic effect of chrysin in combination with colistin against Acinetobacter baumannii.. Front Microbiol., 2022. [DOI | PubMed]
- Antibacterial compounds from Siraitia grosvenorii leaves.. Nat Prod Res., 2011. [DOI | PubMed]
- Dendrocoumarin: a new benzocoumarin derivative from the stem of Dendrobium nobile.. Nat Prod Res., 2018. [DOI | PubMed]
- Use of juglone as antibacterial and potential efflux pump inhibitors in Staphylococcus aureus from the oral cavity.. Microb Pathog., 2016. [DOI | PubMed]
- Isojacareubin from the Chinese herb Hypericum japonicum: potent antibacterial and synergistic effects on clinical methicillin-resistant Staphylococcus aureus (MRSA).. Int J Mol Sci., 2012. [DOI | PubMed]
- In vitro synergism of magnolol and honokiol in combination with antibacterial agents against clinical isolates of methicillin-resistant Staphylococcus aureus (MRSA).. BMC Complement Altern Med., 2015. [DOI | PubMed]
