Alzheimer’s Disease: Exploring the Landscape of Cognitive Decline
Abstract
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder characterized by cognitive decline, memory loss, and impaired daily functioning. The pathology of AD is marked by the accumulation of amyloid beta plaques and tau protein tangles in the brain, along with neuroinflammation and synaptic dysfunction. Genetic factors, such as mutations in APP, PSEN1, and PSEN2 genes, as well as the APOE ε4 allele, contribute to increased risk of acquiring AD. Currently available treatments provide symptomatic relief but do not halt disease progression. Research efforts are focused on developing disease-modifying therapies that target the underlying pathological mechanisms of AD. Advances in identification and validation of reliable biomarkers for AD hold great promise for enhancing early diagnosis, monitoring disease progression, and assessing treatment response in clinical practice in effort to alleviate the burden of this devastating disease. In this paper, we analyze data from the CAS Content Collection to summarize the research progress in Alzheimer’s disease. We examine the publication landscape in effort to provide insights into current knowledge advances and developments. We also review the most discussed and emerging concepts and assess the strategies to combat the disease. We explore the genetic risk factors, pharmacological targets, and comorbid diseases. Finally, we inspect clinical applications of products against AD with their development pipelines and efforts for drug repurposing. The objective of this review is to provide a broad overview of the evolving landscape of current knowledge regarding AD, to outline challenges, and to evaluate growth opportunities to further efforts in combating the disease.
Article type: Review Article
Keywords: Alzheimer’s disease, pathogenesis, aging, amyloid beta plaques, tau protein tangles, protein aggregation, biomarker
License: © 2024 The Authors. Published by American Chemical Society CC BY 4.0 Permits the broadest form of re-use including for commercial purposes, provided that author attribution and integrity are maintained (https://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1021/acschemneuro.4c00339 | PubMed: 39392435 | PMC: PMC11587518
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (13.8 MB)
Introduction
Alzheimer’s disease (AD) is a progressive neurological disorder characterized by cognitive decline, memory loss, and changes in behavior and personality, severe enough to strongly interfere with daily life and activities. Alzheimer’s typically begins slowly and worsens over time, eventually leading to severe impairment in memory, reasoning, judgment, and language skills.1−6 It is ultimately a fatal form of dementia and substantially shortens life expectancy.7,8
Alzheimer’s disease primarily affects neurons, the brain cells responsible for transmitting information throughout the brain and nervous system. The disease is associated with abnormal deposits of proteins in the brain, specifically β-amyloid plaques and tau tangles. These deposits disrupt communication between neurons, a hallmark of the disease.9 Advanced age is the greatest risk factor for Alzheimer’s disease, with the majority of cases occurring in individuals over 65. Other risk factors include genetics, family history, and certain lifestyle factors such as cardiovascular health and education level.10 Diagnosing AD conclusively involves identifying the hallmark pathological features of the disease, extracellular amyloid-beta plaques and intracellular neurofibrillary tangles composed of hyperphosphorylated tau protein.11,12 Diagnosis is typically based on a combination of medical history, cognitive assessments, neurological exams, and ruling out other possible causes of symptoms.13 While there is no cure for AD, there are medications and nondrug interventions that can help slow and manage symptoms along with improve quality of life for patients.
AD can be emotionally and physically challenging for both individuals with the disease and their caregivers. Supportive services such as counseling, support groups, and respite care can be valuable resources for managing the impact of the disease on daily life.14 Research into AD is intense, with efforts focused on understanding its underlying causes, developing more effective treatments, and ultimately finding a cure. Early detection and intervention are important for maximizing treatment effectiveness and improving outcomes for patients affected by the disease.
In this paper, we overview the research progress in Alzheimer’s disease by analyzing data from the CAS Content Collection.15 The CAS Content Collection is the largest human-curated collection of published scientific information, supporting comprehensive quantitative analysis of global research across parameters including time, geography, scientific discipline, application, disease, chemical composition, etc. Covering scientific literature published around the world over the past 150 years in more than 50 languages, the CAS Content Collection encompasses data and discoveries published in more than 50,000 scientific journals and by over 100 patent offices. A major advantage provided by the CAS Content Collection is that, along with the standard reference information, it also provides human curated data on major substances and concepts explored in the scientific publications. The CAS REGISTRY,16 the authoritative source for information on more than 250 million unique organic and inorganic substances and 70 million protein and nucleic acid sequences, is part of the CAS Content Collection. The CAS Content Collection is broadly accessible through CAS solutions including CAS SciFinder17 and CAS STNext.18
Here, we examine the publication landscape in the area in effort to provide insights into current knowledge advances and developments. We review the most discussed and emerging concepts and assess the strategies to combat the disease. We explore the genetic risk factors, pharmacological targets, and comorbid diseases. Finally, we inspect clinical applications of products against AD with their development pipelines and efforts for drug repurposing. The objective of this review is to provide a broad overview of the evolving landscape of current knowledge regarding AD, to outline challenges, and evaluate growth opportunities to further efforts in combating the disease. The merit of the article stems from the extensive, wide-ranging coverage of the most up-to-date scientific information, allowing unique, unmatched breadth of landscape analysis and in-depth insights.
Overview of Alzheimer’s Disease
Prevalence and Impact
The prevalence and impact of AD are significant and continue to grow as populations age. According to the World Health Organization (WHO), around 50 million people worldwide have dementia, and AD accounts for 60–70% of cases. The prevalence of Alzheimer’s increases with age, and the majority of cases occur in individuals over 65 years old. As life expectancy increases globally, the number of people living with AD is expected to rise substantially in the coming decades.19,20
AD has a profound impact on individuals, families, and society as a whole. Individuals with Alzheimer’s experience a progressive decline in cognitive function, memory loss, and changes in behavior and personality, leading to a loss of independence and ability to perform daily tasks. Caregivers, typically family members, bear a significant burden in providing care and support for individuals with AD, often leading to emotional, physical, and financial strain. The economic impact of AD is substantial, including direct medical costs for diagnosis, treatment, and long-term care, as well as indirect costs associated with lost productivity and caregiver burden.
Pathogenesis
The pathogenesis of AD involves a complex interplay of genetic, environmental, and age-related factors, leading to progressive neurodegeneration and cognitive decline.21 One of the defining features of AD is the accumulation of β-amyloid protein fragments in the brain, leading to the formation of insoluble plaques.22,23 β-Amyloid is produced from the cleavage of a larger protein called amyloid precursor protein (APP) by enzymes known as β-secretase and γ-secretase.24 The C99 domain of the amyloid precursor protein (APP) and the amyloid-β protein precursor intracellular domain (AICD) are both involved in the amyloidogenic pathway, which is associated with Alzheimer’s disease. Abnormal processing of APP, along with impaired clearance of β-amyloid from the brain, results in the accumulation of β-amyloid plaques, which are toxic to neurons and disrupt synaptic function (Figure A).
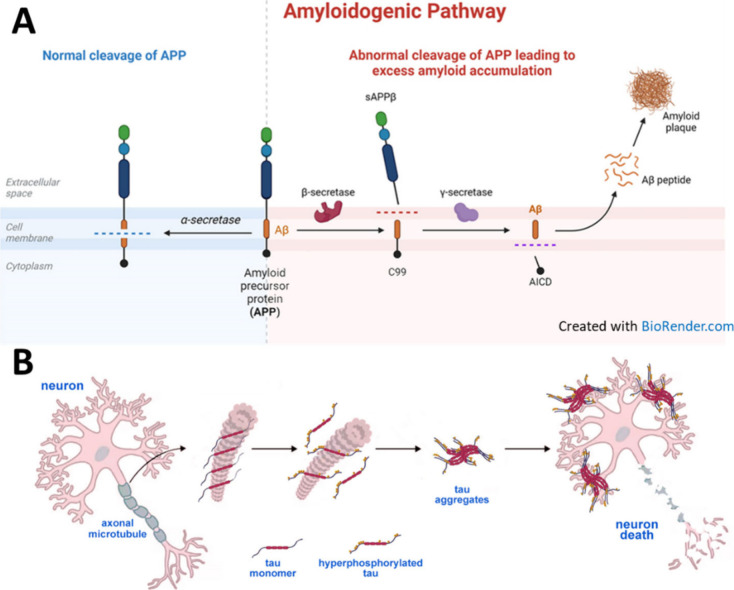
Another characteristic feature of AD is the abnormal accumulation of tau protein in the form of neurofibrillary tangles within neurons.25−27 Tau protein plays a crucial role in stabilizing microtubules, which are essential for maintaining the structure and function of neurons. In AD, tau protein becomes hyperphosphorylated, leading to its misfolding and aggregation into neurofibrillary tangles (Figure B). These tangles interfere with intracellular transport and contribute to neuronal dysfunction and cell death.
The accumulation of β-amyloid plaques and tau protein tangles disrupts normal neuronal function, leading to synaptic dysfunction and impaired neurotransmission.28 As the disease progresses, neurons become increasingly vulnerable to damage and eventually undergo cell death, resulting in widespread neuronal loss, particularly in brain regions critical for memory and cognitive function, such as the hippocampus and cerebral cortex.29
The structural elucidation of amyloid beta (Aβ) plaques and misfolded tau proteins has significantly advanced our understanding of AD pathology.11,30−33 Emerging evidence suggests that AD-related brain changes may result from a complex interplay among abnormal tau and beta-amyloid proteins. Recent advancements in cryo-EM and solid-state NMR have allowed high-resolution imaging of Aβ fibrils.32,34−36 This has revealed detailed arrangements of β-sheets and how they stack to form fibrils. Different strains of Aβ fibrils, known as polymorphs, have been identified. These polymorphs may influence the disease’s progression and severity.37,38 Dual Inhibitors of amyloid-β and tau aggregation with amyloid-β disaggregating properties were recently suggested as new multifunctional ligands that would affect different stages of AD pathogenesis.31 Tauvid has been approved by the US FDA for PET imaging tau pathology in AD.39
Neuroinflammation, characterized by the activation of microglia and astrocytes, plays a significant role in the pathogenesis of AD. Chronic inflammation in the brain exacerbates neuronal damage and contributes to disease progression by releasing pro-inflammatory cytokines, reactive oxygen species, and other neurotoxic molecules.40,41
Vascular dysfunction and impaired cerebral blood flow are common features of AD and may contribute to neuronal damage and cognitive decline. Chronic cerebral hypoperfusion, resulting from vascular pathology, can exacerbate β-amyloid deposition and tau pathology and increase the risk of cognitive impairment.42,43
While most cases of AD are sporadic, a small percentage are inherited in an autosomal dominant fashion, known as familial Alzheimer’s disease (FAD).44 FAD typically manifests as early-onset Alzheimer’s disease (EOAD), meaning symptoms often appear before the age of 65, sometimes as early as in a person’s 30s or 40s. Mutations in genes such as amyloid precursor protein (APP), presenilin 1 (PSEN1), and presenilin 2 (PSEN2) are associated with early-onset FAD, leading to increased production or altered processing of β-amyloid.45 The apolipoprotein E (APOE) ε4 allele is the strongest genetic risk factor for late-onset Alzheimer’s disease (LOAD) and is associated with increased β-amyloid deposition and enhanced risk of developing AD.
Stress, depression, and anxiety are increasingly recognized as significant risk factors for AD.46−48 Chronic stress leads to prolonged elevated levels of cortisol, a stress hormone.49 High cortisol levels can damage the hippocampus, a critical brain area for memory and learning, which is often one of the first regions affected in AD. Stress also triggers an inflammatory response, which can lead to chronic neuroinflammation.50 This inflammation can contribute to neuronal damage and the progression of AD.51,52 Stress has been associated with an increase in amyloid-beta production and tau protein hyperphosphorylation, both of which are hallmark features of AD pathology.53,54
Depression is linked to a reduction in hippocampal volume.55,56 Since the hippocampus is crucial for memory formation, its atrophy can predispose individuals to cognitive decline and AD. Depression often involves imbalances in neurotransmitters such as serotonin, norepinephrine, and dopamine.57 These imbalances can affect cognitive function and increase the risk of AD. Depression is also associated with increased levels of pro-inflammatory cytokines.58 Chronic inflammation can exacerbate neurodegeneration and amyloid plaque formation.59,60
Similar to stress, anxiety leads to dysregulated cortisol production, which can have detrimental effects on brain structures involved in memory and cognition.61 Anxiety can result in changes to synaptic plasticity and neurotransmitter release, impairing cognitive processes and potentially accelerating the onset of AD.62,63 Anxiety also increases oxidative stress, leading to cellular damage in the brain. Oxidative damage is a known contributor to the pathogenesis of AD.64,65
These conditions often co-occur and can exacerbate each other, creating a vicious cycle that heightens the risk for Alzheimer’s disease. For instance, chronic stress can lead to depression, and depression can increase anxiety, all of which collectively increase AD risk46 through shared and overlapping pathways: (i) the hypothalamic-pituitary-adrenal (HPA) axis, which regulates stress responses, can become dysregulated in all three conditions, leading to sustained high levels of stress hormones;66 (ii) stress, depression, and anxiety negatively impact neuroplasticity, the brain’s ability to adapt and reorganize, which is vital for maintaining cognitive function;67,68 (iii) all three conditions can lead to chronic neuroinflammation, which plays a critical role in the neurodegenerative processes underlying AD.69,70
Risk factors for cardiovascular disease, such as hypertension, high cholesterol, diabetes, and obesity, are also associated with an increased risk of AD.71−74 Regular physical exercise has been shown to have a protective effect against AD and cognitive decline.67,75 Some dietary patterns, such as the Mediterranean diet, rich in fruits, vegetables, whole grains, fish, and healthy fats, may lower the risk of AD.76−78 Higher levels of education and engagement in mentally stimulating activities throughout life may help reduce the risk of developing AD.79,80
Some environmental factors, such as air pollution, heavy metal exposure, and certain toxins, have been implicated as potential risk factors for AD, although more research is needed to fully understand their impact.81,82
Genetic Background
The genetic background of Alzheimer’s disease (AD) encompasses both rare familial forms and more common late-onset forms.83−85
Familial Alzheimer’s disease (FAD) represents a small percentage (∼5–10%85−87) of all AD cases and is inherited in an autosomal dominant pattern, meaning that an affected individual has a 50% chance of passing the mutated gene to their offspring.88,89 Mutations in three genes have been identified as causative for FAD: (i) Amyloid precursor protein (APP): mutations in the APP gene, located on chromosome 21, tend to inhibit cleavage by α-secretase and facilitate preferential cleavage by β-secretase, which leads to increased production or altered processing of β-amyloid, resulting in the accumulation of amyloid plaques in the brain; (ii) Presenilin 1 (PSEN1): mutations in the PSEN1 gene, located on chromosome 14, are the most common cause of FAD. PSEN1 is a component of the γ-secretase complex involved in the cleavage of amyloid precursor protein (APP), and mutations in PSEN1 enhance cleavage by γ-secretase and lead to increased production of β-amyloid; (iii) Presenilin 2 (PSEN2): mutations in the PSEN2 gene, located on chromosome 1, are less common but can also cause FAD. Like PSEN1, PSEN2 is involved in the processing of APP, enhanced cleavage by γ-secretase, and the production of β-amyloid.24,90−92
Late-onset Alzheimer’s disease (LOAD) is the most common form of AD and typically occurs after age of 65.93,94 While LOAD has a strong genetic component, it is influenced by multiple genetic and environmental factors. The strongest genetic risk factor for LOAD is the apolipoprotein E (APOE) gene, located on chromosome 19. The APOE gene has three common alleles: ε2, ε3, and ε4. The ε4 allele of APOE (APOE4) is associated with an increased risk of developing AD and a younger age of onset, at 65.1, with a narrow 95% prediction interval.95 Individuals who inherit one copy of the APOE ε4 allele have an increased risk, while those who inherit two copies (homozygous) have an even higher risk, estimated to be 8 to 12 times greater than those with no ε4 alleles.84,96 Recent study has concluded that APOE4 homozygotes represent in fact a distinct genetic form of AD.95,97
In addition to APOE, several other genetic variants have been identified as risk factors for LOAD through genome-wide association studies. These include genes involved in inflammation, cholesterol metabolism, immune response, and synaptic function. While these genetic risk factors increase susceptibility to AD, they do not guarantee that an individual will develop the condition. Environmental factors and gene-environment interactions also play a significant role in disease risk.93,94,98,99
Symptoms and Progression
AD progresses gradually over time, and the symptoms can vary from person to person. One of the most common early signs is difficulty remembering recent events, conversations, or information. Individuals may have trouble with tasks that require planning, decision-making, and problem-solving. They may become disoriented, especially in unfamiliar environments, and have difficulty following directions or understanding the passage of time. They may have trouble finding the right words or understanding spoken or written language. Changes in mood, such as depression, anxiety, or apathy, and shifts in personality traits may occur.100−104
As AD progresses, memory loss becomes more severe and may include forgetting the names of close family members or important personal information. Language difficulties may worsen, making it increasingly difficult to engage in conversations or express thoughts. Individuals may have difficulty making decisions or solving problems, and they may exhibit poor judgment in everyday situations. Behavioral symptoms such as agitation, aggression, wandering, or social withdrawal may become more pronounced. In the later stages of AD, individuals may experience physical symptoms such as difficulty swallowing, walking, or performing basic self-care tasks. As the disease progresses, individuals become increasingly dependent on others for assistance with activities of daily living.8,104,105
In the advanced stages of AD, individuals may lose the ability to communicate verbally, recognize loved ones, or control movement. They may require round-the-clock care in a residential facility or at home with the assistance of caregivers. Physical complications such as infections, falls, and malnutrition become more common, contributing to further decline in health.104−106
Diagnosis
Diagnosing AD involves a comprehensive assessment by healthcare professionals, including medical history, physical examination, cognitive assessments, and imaging tests. Cognitive tests are used to evaluate memory, attention, language, problem-solving, and other cognitive functions. These assessments may include standardized tests such as the Mini-Mental State Examination (MMSE) or the Montreal Cognitive Assessment (MoCA).107−109 The healthcare provider may also administer more in-depth neuropsychological testing to assess specific cognitive domains and identify patterns of impairment.
A neurological examination may be performed to assess for signs of neurological dysfunction, such as abnormal reflexes, muscle weakness, or coordination problems. Imaging tests such as magnetic resonance imaging (MRI) or positron emission tomography (PET) scans may be used to rule out other possible causes of cognitive impairment, such as stroke, tumor, or hydrocephalus.110 These imaging studies can also help visualize changes in the brain associated with AD, such as atrophy (shrinkage) of brain regions involved in memory and cognition or the presence of β-amyloid plaques and tau tangles. Blood tests may be performed to rule out other medical conditions that can cause cognitive impairment, such as thyroid dysfunction, vitamin deficiencies, or infections.110 Cerebrospinal fluid (CSF) analysis may be considered in some cases to measure levels of β-amyloid and tau proteins, which can be indicative of AD pathology. In addition, noninvasive diagnostic tests are being considered as a part to the comprehensive AD assessment. One such test, PrecivityAD2 is a clinical care assay that measures both plasma amyloid beta and tau peptide concentrations.111,112 Other assays utilizing not only blood, but saliva and urine are currently being researched.113,114
Diagnosis of AD is typically based on clinical criteria established by organizations such as the National Institute on Aging and the Alzheimer’s Association (NIA-AA) or the International Working Group (IWG) for AD.115−117 These criteria consider the presence and pattern of cognitive symptoms, the progression of symptoms over time, and the exclusion of other possible causes of dementia.
Treatment and Management
Treatment and management of AD aim to alleviate symptoms, slow down the progression of the disease, and improve the quality of life for individuals affected by the condition. While there is currently no cure for AD, various interventions can help manage symptoms and support overall well-being.118−121
Medications such as donepezil (Aricept), rivastigmine (Exelon), and galantamine (Razadyne) are cholinesterase inhibitors commonly prescribed to treat cognitive symptoms associated with AD.122 These drugs work by increasing levels of acetylcholine, a neurotransmitter involved in memory and learning, in the brain.123,124 Memantine (Namenda) is another medication approved for the treatment of AD.125 It works by regulating the activity of glutamate receptors, another neurotransmitter involved in learning and memory.126,127 These medications may help delay the decline in cognitive function, behavior, and daily functioning in some individuals with AD. Selective serotonin reuptake inhibitors (SSRIs) are primarily known for their use in treating depression and anxiety disorders. However, they have also been investigated for their potential benefits in managing symptoms of AD.128,129 SSRIs work by increasing the levels of serotonin in the brain by inhibiting its reuptake into the presynaptic cell, making more serotonin available to bind to the postsynaptic receptor.130,131 Commonly used SSRIs in AD include citalopram, sertraline, fluoxetine and escitalopram.130−133 These SSRIs are also used to manage depression and anxiety in AD, although their specific effects on AD-related symptoms are less well-documented.134
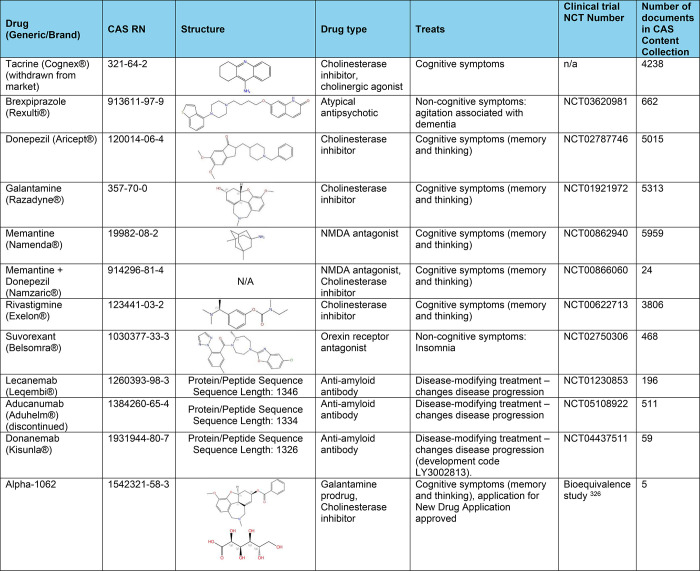
US FDA has approved a few antiamyloid-beta monoclonal antibodies for the treatment of Alzheimer’s disease.135−137 These therapies aim to reduce amyloid-beta plaques in the brain, which are thought to play a crucial role in the progression of AD. These monoclonal antibody drugs include: (i) Aducanumab (Aduhelm),138 targeting aggregated forms of amyloid-beta, including soluble oligomers and insoluble fibrils–by binding to these aggregates, it promotes their clearance from the brain (planned to be discontinued in November 2024, for “reprioritization” of its resources to focus on other Alzheimer’s disease treatments139); (ii) Lecanemab (Leqembi)140 that preferentially binds to soluble amyloid-beta protofibrils, facilitating their clearance from the brain–this action helps reduce amyloid plaque buildup and is thought to slow the progression of AD; (iii) Donanemab,141 which targets a specific modified form of amyloid-beta called N 3pG, which is found in amyloid plaques–by binding to this form of amyloid-beta, donanemab promotes the clearance of amyloid plaques from the brain. The approval of these antiamyloid-beta monoclonal antibodies marks a significant milestone in AD treatment.142 These therapies offer hope for slowing disease progression by targeting one of its key pathological features. However, the clinical benefits, optimal use, and safety profiles of these treatments continue to be areas of active research and discussion in the medical community. For more information on AD medications, see Table 1 and Figure in the Landscape analysis section further in the text.
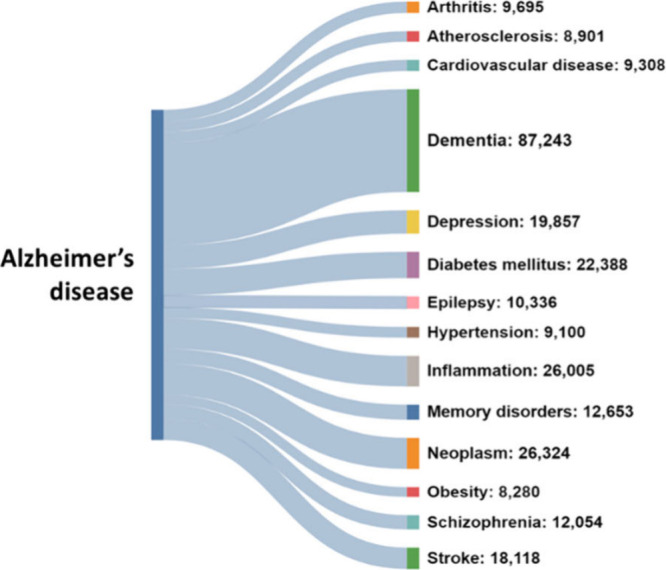
Table 1: FDA Approved Medications That Mitigate Symptoms of Alzheimer’s Dementia or Change the Disease Progression120,124,323−326
Engaging in mentally stimulating activities may help maintain cognitive function and slow down cognitive decline. Regular physical activity has been shown to have beneficial effects on cognition and overall health in individuals with AD. Eating a balanced diet can support overall brain health and well-being. Addressing sleep disturbances and establishing healthy sleep habits can help improve cognitive function and mood in individuals with AD.143−145
Behavioral interventions such as cognitive-behavioral therapy (CBT) or behavior modification techniques may help manage challenging behaviors such as agitation, aggression, or wandering.146−148 Counseling, support groups, and other psychosocial interventions can provide emotional support, education, and coping strategies for individuals with AD and their caregivers.149,150
Landscape Analysis of Alzheimer’s Disease Research
Journal Publication and Patent Trends (CAS Content Collection Data)
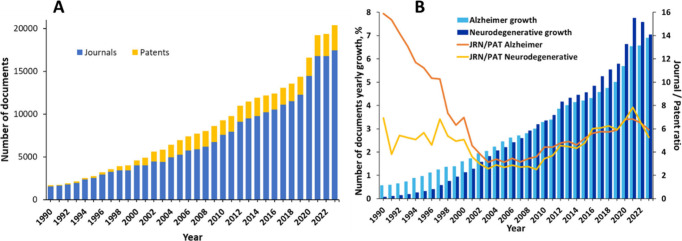
Our search in the CAS Content Collection15 for AD-related documents (search term: Alzheimer* in Title, Abstract, Keywords, and Concept fields) retrieved over 300,000 scientific documents (mainly journal articles and patents), including over 250,000 articles in scientific journals and nearly 50,000 patents. There has been a steady growth of these documents over the last three decades, with an >30% increase in the last three years (2021–2023) (Figure A). The journal articles largely dominate, showcasing the intense research in the area. The relative growth in the number of documents related to the AD as well as the journal/patent number ratio virtually coincides with that for the overall class of neurodegenerative diseases during the last two decades (Figure B) which is understandable since AD is by far the most prevalent and most widely explored disease in that class of diseases. The high journal/patent number ratio with regards to the AD research in the 1990s reflect the initial period of knowledge accumulation preceding the subsequent opportunities for commercialization.
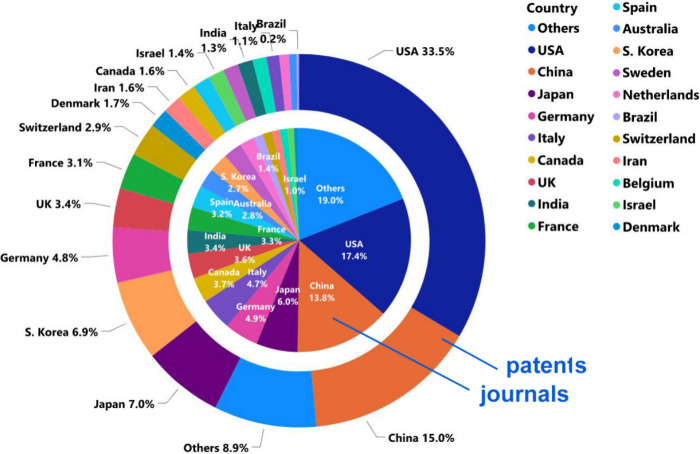
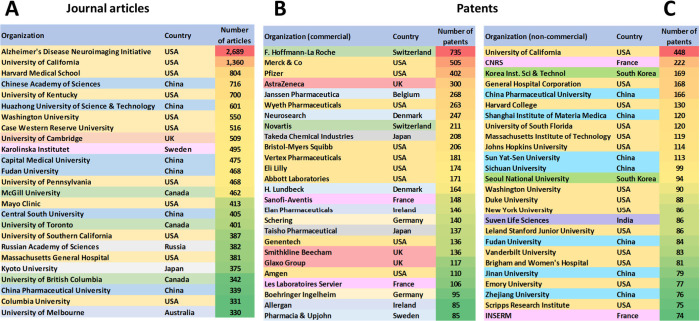
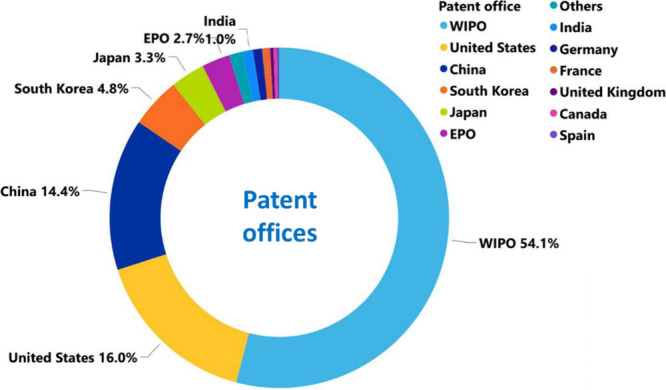
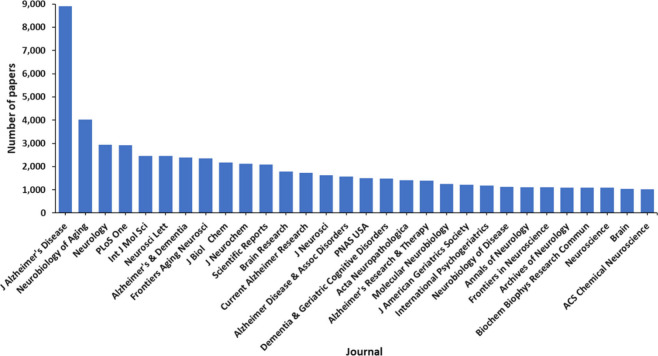
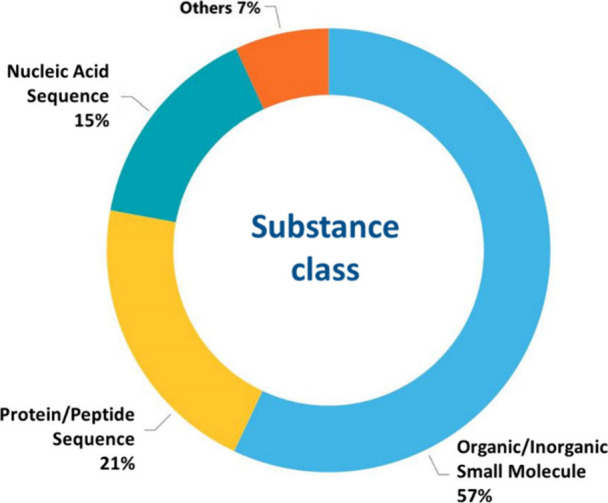
USA, China, Japan, Germany, and South Korea are the leaders with respect to the number of published journal articles and patents related to AD research (Figure ). The Alzheimer’s Disease Neuroimaging Initiative, University of California, Harvard Medical School, and the Chinese Academy of Sciences are the leaders with respect to the number of published journal articles related to the AD (Figure A). Patenting activity is dominated by corporate players as compared to academics (Figure B,C). F. Hoffmann-La Roche, Merck, Pfizer, and AstraZeneca have the highest number of patent applications among the companies (Figure B), while the University of California, the Centre national de la recherche scientifique (CNRS, France), and the Korea Institute of Science and Technology lead among the noncommercial organizations (Figure C). The most patent applications have been filed at the World Intellectual Property Organization (WIPO) followed by the US and China patent offices (Figure ). Journal of Alzheimer’s Disease is a distinct leader with respect to the number of published articles related to Alzheimer’s disease research, followed by Neurobiology of Aging, Neurology, and PLoS One (Figure ). With respect to the substance classes explored in the AD-related documents in CAS Content Collection, the largest part belong to the organic and inorganic small molecules (Figure ).
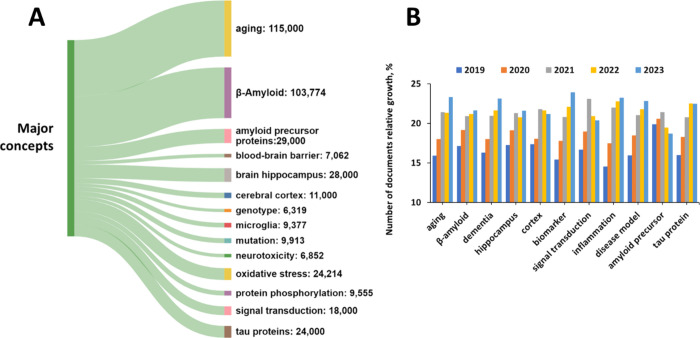
Aging and β-Amyloid Are the Most Widely Explored Concepts in AD Research
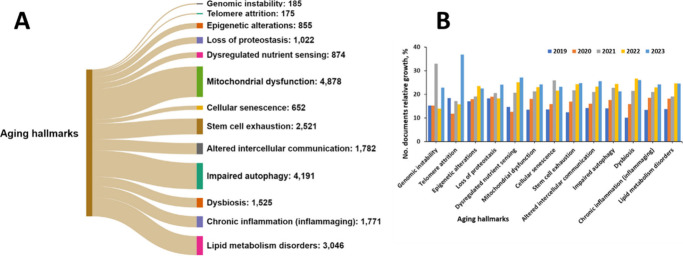
Utilizing the key advantage of the CAS Content Collection in providing information on major substances and concepts explored in the scientific publications as identified by subject matter experts (SME), we were able to identify most frequently discussed concepts within the Alzheimer’s disease-related document subset of the database, as well as to perform cross-concept searches in order to identify concept co-occurrence in the documents. The distribution of the Alzheimer’s disease-related concepts in the published documents (journals and patents) and their annual trends are presented in Figure . Aging and β-amyloid are the most widely explored concepts (Figure A). This finding is well rationalized. Indeed, aging is a leading risk factor for Alzheimer disease.20,151,152 Biological processes altered with aging, which have been implicated in AD. It has been explicated that the pathogenesis of AD and other neurodegenerative diseases is associated with the major hallmarks of aging: genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, mitochondrial dysfunction, cellular senescence, deregulated nutrient sensing, stem cell exhaustion and altered intercellular communication.153−156
According to the existing hypotheses, the accumulation of toxic amyloid β-protein (Aβ) in the central nervous system is the major pathophysiological feature and the basis of AD.157−159 Extracellular abnormalities of Aβ levels in the brain may result in the accumulation of Aβ, forming a structure rich in β-sheet structures. Upon forming oligomers, they recombine into fibrils to form amyloid plaques (Figure ).
Biomarkers Are the Fastest Growing Concept Related to AD
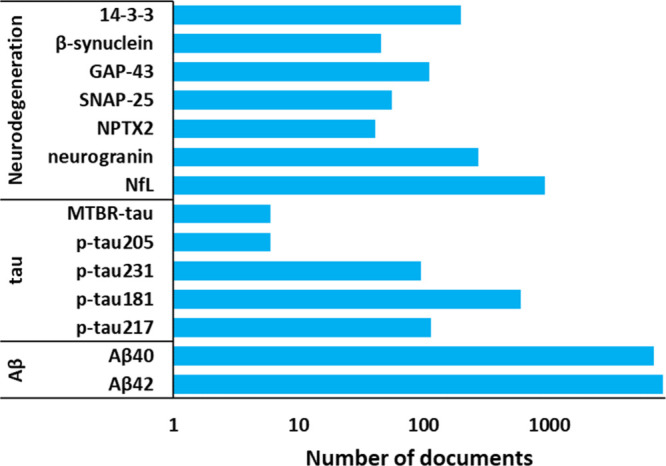
Figure B illustrates the annual trends in some key concepts during the last five years (2019–2023). The biomarker concept appears as the fastest growing one. Biomarkers are measurable indicators or characteristics that can provide information about the presence, progression, or risk of developing the disease.160,161 They play a crucial role in diagnosing, monitoring disease progression, assessing treatment response, and facilitating research into the underlying mechanisms of disease. For example, the levels of an amyloid-beta peptide, Aβ42, a fragment of amyloid-beta protein, in the cerebrospinal fluid (CSF) are typically decreased in individuals with AD compared to healthy individuals.162−165 Aβ42 is known to aggregate and form plaques in the brain, a hallmark pathology of Alzheimer’s. CSF levels of tau proteins, particularly phosphorylated tau, are elevated in AD patients.164−166 Tau proteins are involved in microtubule stabilization, and their abnormal phosphorylation leads to neurofibrillary tangle formation, another pathological feature of AD.25,167
Overall, biomarkers from cerebrospinal fluid (CSF) and from blood plasma including amyloid peptides and phosphorylated tau are used as AD biomarkers in clinic.168−171 The procedure for detection of CSF-based biomarkers is intrusive, it requires a lumbar puncture. In contrast, blood tests for AD biomarkers offer the potential for noninvasive, cost-effective screening and monitoring of disease progression, hence, blood-based biomarkers are highly preferred and are currently being actively researched and developed.172−174 These include measures of amyloid-beta, tau, neurofilament light chain, and other proteins associated with AD pathology (Figure ). Noteworthy, the p-tau217 blood test, for which US FDA granted Breakthrough Device designation in March 2024,175 has been reported to exhibit high diagnostic accuracy for identifying AD among individuals with cognitive symptoms.176 Genetic variants associated with AD, such as mutations in the amyloid precursor protein, presenilin 1 (PSEN1), and presenilin 2 (PSEN2) genes, can serve as biomarkers for increased risk of developing familial forms of AD.45,177 The apolipoprotein E (APOE) gene, particularly the ε4 allele, is a well-established genetic risk factor for late-onset AD. APOE genotyping can help identify individuals at higher risk of developing AD.178,179
Positron emission tomography (PET) imaging using radiotracers that bind to amyloid plaques allows for the visualization and quantification of amyloid deposition in the brain.180−182 Amyloid PET scans can help confirm the presence of AD pathology in individuals with cognitive impairment. PET imaging using radiotracers specific to tau aggregates enables the visualization of neurofibrillary tangles in the brain, providing insights into the spread and distribution of tau pathology in AD. Magnetic resonance imaging (MRI) techniques can detect structural changes, such as hippocampal atrophy and cortical thinning, as well as functional alterations, such as changes in brain connectivity, associated with AD.183−185
Biomarkers of neuroinflammation, such as markers of microglial activation and inflammatory cytokines, are being investigated for their potential role in AD pathogenesis and progression.186−188 Biomarkers of neuronal injury and degeneration, such as neurofilament light chain (NFL) levels in CSF or blood, can indicate ongoing neurodegenerative processes in AD.189,190 The identification and validation of reliable biomarkers for AD hold great promise for improving early diagnosis, monitoring disease progression, and assessing treatment response in clinical practice. Ongoing research efforts continue to advance our understanding of Alzheimer’s biomarkers and their clinical utility in the management of this devastating neurodegenerative disorder. Exemplary AD biomarkers are illustrated in Figure .
Association of Alzheimer’s Disease with Other Diseases
A large assortment of comorbid diseases is associated with AD.191 There is evidence that chronic diseases, including diabetes, cardiovascular disease, depression, and inflammatory bowel disease, may be associated with enhanced risk of AD. Disruption in certain shared biological pathways has been suggested as the underlying mechanism for the relationship between AD and these diseases. Particularly, inflammation is a common dysregulated pathway shared by most of the diseases associated with AD. We examined the co-occurrence of certain diseases with AD as reflected by the co-occurrence of the respective concepts in the documents of the CAS Content Collection (Figure A). Dementia and inflammation are between the expected co-occurrences. With respect to cancer, inverse occurrence of cancer and Alzheimer disease has been reported: patients with dominant cancer had a 43% lower risk of developing AD, and those with prevalent AD had a 69% lower risk of being diagnosed with cancer.192−194 Stroke has been reported to be associated with AD among elderly patients. The relation is strong in the presence of certain vascular risk factors.195,196 Recently, there is accumulating evidence demonstrating that hyperglycemia is a potential risk factor for the development of cognitive impairment or AD.197,198 It has been reported that AD patients have a high risk of developing certain types of epilepsy and subclinical epileptiform activity.199
Pharmaceutical Targets and Genetic Risk Factors
There are multiple potential pharmaceutical targets for the treatment of AD, as currently documented in the Common Alzheimer’s Disease Research Ontology (CADRO),200 including amyloid beta (Aβ) peptide, tau protein, apolipoprotein E4 (APOE4), lipids, lipoprotein and neurotransmitter receptors, enzymes, and many other regulators, and multitarget interventions.201 Amyloid beta and tau proteins are the major undisputed pharmacological targets for disease modifying therapies of AD (Figure A, lower panel). The fastest growing target according to the CAS data is the transactive response DNA binding protein of 43 kDa (TDP-43), a nuclear protein involved in the regulation of gene expression (Figure A, upper panel). Cytoplasmic inclusion bodies comprising phosphorylated and truncated forms of TDP-43 have been found in multiple AD cases, as well as in other proteinopathies including amyotrophic lateral sclerosis, frontotemporal dementia. TDP-43 deposits have been also found in neurons with neurofibrillary tangles. There is emerging evidence that TDP-43 may spread in a prion-like manner, which means it could propagate from cell to cell, potentially contributing to the progression of neurodegenerative diseases like AD.202 The most common genetic risk factor for AD, apolipoprotein E4 (APOE4), is associated with increased frequency of TDP-43 pathology.203−206
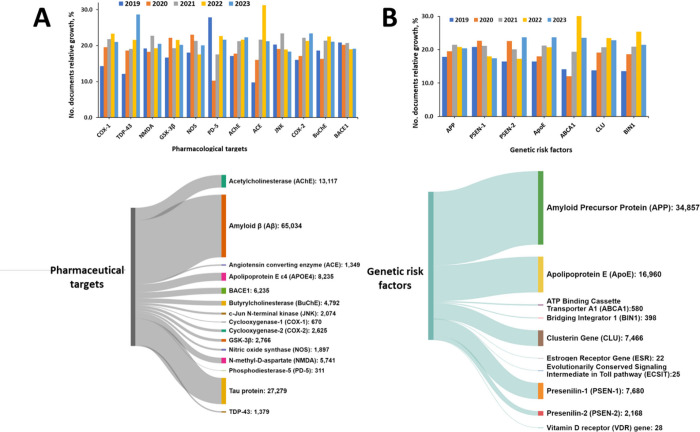
Angiotensin-converting enzyme (ACE) is another pharmacological target of AD exhibiting rapid growth in the number of publications included in the CAS Content Collection (Figure A, upper panel). Indeed, among the Aβ degrading enzymes such as neprilysin, insulin-degrading enzyme, endothelin-converting enzyme, and ACE, the last one is the most commonly targeted enzyme by inhibitors, mainly because it plays central role in the regulation of blood pressure and hypertension.207,208 However, genetic, pathological and biochemical studies have associated ACE with AD.209 ACE has been shown able to convert neurotoxic β-amyloid protein Aβ42 to Aβ40. Aβ42 is believed to play a causative role in the development of AD, whereas Aβ40 exhibits neuroprotective activities against Aβ42 aggregation as well as against metal-induced oxidative damage.210
Genetic factors play a major role in the development of AD. In the late-onset type of AD, which applies to 95% of cases, the underlying etiology is supposedly caused by a combination of genetic and environmental factors in the approximate ratio of 70:30, respectively.211 In the rare cases of early-onset AD, the disease etiology is allegedly almost exclusively genetic.212−216 Hence, genetics seem to play a major role in all types of AD.
Mutations in the dominant genes including Amyloid Precursor Protein (APP), Presenilin-1 (PSEN-1), Presenilin-2 (PSEN-2), apolipoprotein E (ApoE), and others, are associated with AD.83,217 From 30 discovered mutations in the APP gene, 25 have been found related to AD and causing accumulation of Aβ.218,219 PSEN1 and PSEN2 genes are also the autosomal dominant form of early-onset AD. Mutation in PSEN1 gene is more frequent, with more than 200 mutations, while in the PSEN2 gene only a rare form with less than 40 mutations was identified.45,220 Apolipoprotein E (ApoE), especially ApoEε4 plays an important role in Aβ deposition as a senile plaque. It has been found to cause cerebral amyloid angiopathy, known as a marker for AD.221 ApoEε4 is also related to vascular damage in the brain, leading to AD pathogenesis.222 Adenosine triphosphate (ATP)-binding cassette transporter A1 (ABCA1) is known to regulate the stability of ApoE lipidation. Mutation in the ABCA1 gene has been reported to result accumulation of cholesterol in tissues, and AD pathogenesis.222 In contrast to PSEN1, PSEN2, and APP mutations, Clusterin (CLU) and Bridging Integrator 1 (BIN1) genes are risk factors for late-onset AD.223−225
According to CAS Content Collection, the largest number of documents associate APP, ApoE, PSEN-1, and CLU genes with AD pathogenesis (Figure B, lower panel). ABCA1, CLU, BIN1, and ApoE are the risk factors exhibiting the fastest growth in the number of documents in the last five years (2019–2023) (Figure B, upper panel).
We further considered the correlation between the genetic risk factors of the AD and the pharmacological targets. The co-occurrence of genetic factors and pharmacological targets in AD highlights the intricate interplay between genetics and potential treatments. Understanding this correlation can provide insights into the underlying mechanisms of the disease and guide drug development efforts. Likely intersections of genetic factors and pharmacological targets include: (i) precision medicine: genetic profiling can help tailor treatments based on individual risk factors, for example, patients with APOE ε4 might benefit more from certain amyloid-targeting therapies;84,226 (ii) APP and secretase inhibitors: understanding mutations in APP and presenilin genes has led to the development of secretase inhibitors to reduce amyloid-beta production;218,227−229 (iii) TREM2 and inflammation modulation: genetic studies linking TREM2 to AD have spurred interest in developing drugs that modulate microglial activity to reduce neuroinflammation;230−232 (iv) combination therapies: considering the multifactorial nature of AD, combination therapies targeting both amyloid-beta and tau, along with anti-inflammatory and neuroprotective strategies, may be more effective.185,233,234
Indeed, many pharmacological approaches for treating AD target the pathological hallmarks of the disease:
- Drugs targeting amyloid-beta include β-secretase inhibitors, γ-secretase inhibitors, and monoclonal antibodies that bind to amyloid-beta and promote its clearance.235,236
- Drugs targeting tau protein include tau aggregation inhibitors, tau kinase inhibitors, and immunotherapies aimed at reducing tau pathology.237−240
- Anti-inflammatory drugs target neuroinflammation, which is implicated in Alzheimer’s disease progression.59,241,242
- Neuroprotective agents aim to preserve neuronal function and viability, potentially slowing disease progression.243−245
In effort to the get insight into the correlation between the genetic risk factors of AD and the pharmacological targets, we searched the documents of the CAS Content Collection related to AD for co-occurrences of certain major concepts related to the genetic risk factors and to the pharmacological targets within the AD-related document pool of the CAS Content Collection. The results are illustrated in Figure . APP co-occurs in documents with all target proteins, but with highest frequency with BACE1 and PD-4; ApoE most frequently co-occurs with TDP-43 and ACE, and PSEN-1–most frequently with GSK-3β and JNK.
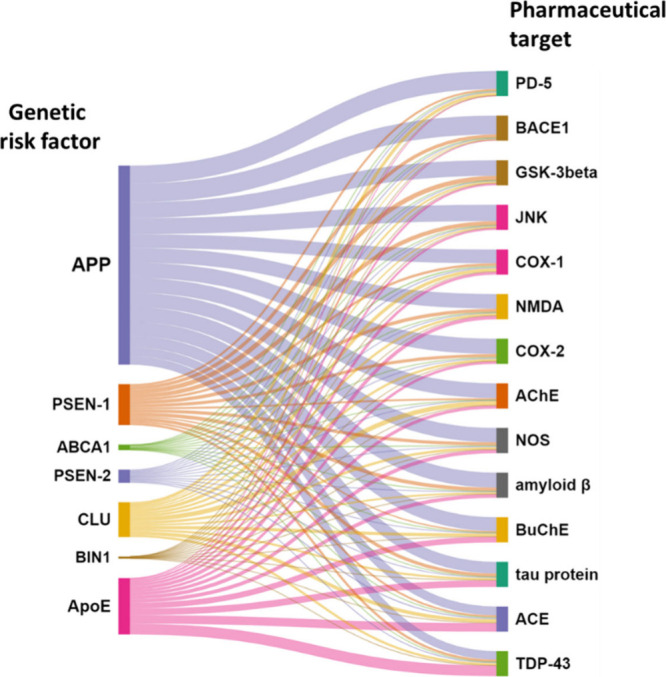
- The correlation between APP and BACE1 is critical in the context of AD. APP is a transmembrane protein that, when cleaved by BACE1, produces beta-amyloid peptides. These peptides can aggregate to form amyloid plaques, a hallmark of AD. High levels of BACE1 activity can increase the production of Aβ, thus promoting plaque formation. Changes in the expression or activity of either APP or BACE1 can significantly affect Aβ production. Studies have shown that upregulation of BACE1 is observed in AD patients, which correlates with increased Aβ production. There is also evidence that oxidative stress and neuroinflammation can upregulate BACE1 expression and activity. Targeting BACE1 to reduce its activity is a potential therapeutic approach to lower Aβ production.246−248
- The correlation between APP and PD-4 in AD is an emerging area of research, albeit less explored than the correlation between APP and BACE1. Potential correlation include: (i) cAMP signaling: altered cAMP signaling has been implicated in neurodegenerative diseases, including AD.249 PD-4 role in cAMP degradation suggests that it could impact processes relevant to AD pathology;250 (ii) Neuroinflammation: elevated PD-4 activity can lead to reduced cAMP levels, potentially contributing to neuroinflammation.251 Neuroinflammation is a known factor in Alzheimer’s disease progression; (iii) Memory and cognitive function: PD-4 inhibitors have been studied for their potential to improve cognitive function by increasing cAMP levels, which could be beneficial in AD.252,253 Targeting PD-4 to modulate cAMP levels offers a potential therapeutic strategy. By inhibiting PD-4, it may be possible to reduce neuroinflammation and improve cognitive function in AD patients.254
- The correlation between ApoE and TDP-43 in AD is a significant topic as both proteins are implicated in neurodegenerative processes. It involves complex interactions between genetic risk factors, protein aggregation, and neuroinflammatory processes. ApoE4 appears to influence TDP-43 pathology, potentially exacerbating the overall disease pathology and cognitive decline.203,255 Further research is needed to fully understand the mechanisms underlying this interaction and to explore potential therapeutic strategies targeting both ApoE4 and TDP-43 in AD.
- The correlation between ApoE and ACE in AD involves their roles in neurodegenerative processes, vascular health, and inflammation. Both proteins have been implicated in the disease, with ApoE being a major genetic risk factor84 and ACE being involved in amyloid-beta metabolism and blood pressure regulation.256
- The correlation between PSEN-1 and GSK-3β in AD is significant due to their roles in amyloid-beta production and tau phosphorylation, respectively. Both proteins are implicated in key pathways that contribute to the pathogenesis of AD. PSEN-1 mutations lead to increased amyloid-beta, which can activate GSK-3β and exacerbate tau pathology.257,258
- The correlation between PSEN-1 and JNK in AD involves their roles in cellular signaling, amyloid-beta production, and neurodegeneration. Both PSEN-1 and JNK are implicated in pathways that contribute to AD pathology, particularly in relation to amyloid-beta accumulation, tau phosphorylation, and neuronal apoptosis.28,246,257 Understanding this interaction provides insights into the molecular mechanisms of AD and highlights potential therapeutic targets for managing both amyloid and tau-related aspects of the disease.
Role of Primary Aging Hallmarks in Alzheimer’s Disease
Aging is characterized with a time-dependent gradual accumulation of cell damage and continual physiological functional decline. As such, it is also the most profound risk factor for many diseases. Neurodegenerative diseases, such as Alzheimer’s, Parkinson’s, and Huntington’s diseases, as well as sensorial disfunctions all increase considerably upon aging.259−262 With the growth in aging population and increasing burden of health care for people with age-related diseases, including AD, intense efforts have been put forth to understand and prevent the effects of aging. The major cellular and molecular hallmarks of aging have been identified and their relationships to age-related diseases, and especially to AD as one of the most common neurodegenerative disorders, have been explored.263
The hallmarks of aging include a variety of interrelated molecular and cellular mechanisms that act jointly to manage the aging process.264 Aging has been characterized as a progressive degeneration accompanied by processes like stem cell exhaustion, extracellular matrix modifications, cellular senescence, tissue inflammation, and metabolic dysfunction.153,156 These cellular and tissue modifications reflect inherent molecular deviations in mitochondria, epigenetics, DNA maintenance, proteostasis, intercellular interactions, and nutrient sensing, which cause genomic instability and impairment, including telomere dysfunction.153,156
DNA damage markers have been found in brain regions of AD patients, indicating that DNA damage may be an important pathological cause of AD, particularly in late-onset AD cases.265−267 Genomic instability impacts the expression of the genes linked to mitochondrial and metabolic dysfunction, altered proteostasis, and age-related inflammation (inflammaging), which are intrinsically involved in aging, and supports the view that DNA damage could be the root of aging and AD.268,269 Hence, targeting DNA damage or other aging hallmarks offers an approach to developing treatments to combat age-related diseases, including AD.268
Telomeres shorten with age, and some 50 nucleotides are lost upon each cell cycle. Neurons, as postmitotic cells, are not expected to have their telomeres shortened, yet they may still accumulate DNA damage, causing cellular senescence or even apoptosis.270,271 Neural stem cells, in contrast, are proliferative and affected by aging, so telomere maintenance is vital for their viability and self-renewal potential, while telomere shortening may trigger cognitive impairment.272 Epigenetic mechanisms are known to contribute directly to aging and aging-related diseases.273 It has been shown that epigenetics play a key role in maintaining genome integrity and regulating gene expression, and its dysfunction is closely related to AD pathogenesis.274
DNA methylation refers to the attachment of a methyl group to the DNA chain and is considered one of the aging hallmarks.153 Abnormal DNA methylation has been associated with many AD susceptibility genes, including amyloid precursor protein (APP), β- and γ-secretases, apolipoprotein E, to mention a few.263,275,276 As an organism ages, the proteome, like the genome, is easily damaged, so loss of proteostasis come up another hallmark of aging.153 At the early and advanced stages of AD, alteration of protein synthesis pathways, including nuclear chaperones, ribosomal proteins, and elongation factors, has been detected in the frontal cortex and hippocampus.277,278 Furthermore, proteins associated with various other biological processes are also aberrantly downregulated due to genomic instability in the AD brain.279,280
A search in the CAS Content Collection by SciFinder17 for co-occurrence of AD and the aging hallmarks term that AD is most often discussed in the context of mitochondrial dysfunction, impaired autophagy, and lipid metabolism disorders (Figure A). Indeed, mitochondrial dysfunction has been found related to virtually all the associated AD pathologies, including accumulation of plaques and tangles in the hippocampal and cortical neurons of the brain, abnormal microvasculature, interneuron miscommunication, enhanced β-amyloid production, elevated inflammatory response, advanced production of reactive oxygen species, impaired brain metabolism, tau hyperphosphorylation, and disruption of acetylcholine signaling.281−289
Autophagy is an evolutionarily conserved lysosome-dependent cellular pathway closely related to modulation of protein metabolism, by way of which damaged organelles and misfolded proteins are degraded and recycled to preserve protein homeostasis. Accumulating evidence has revealed that impaired autophagy critically contributes to AD pathogenesis.290,291 Immature autophagosome accumulation and dystrophic neurites have been detected in the AD patients brains.291 Furthermore, the expression of certain autophagy-related proteins has been reported to be downregulated in AD brains.292−294 Increasing evidence has recently indicated that dysfunctional autophagy is not just correlated with AD pathologies, but is possibly a causative factor for AD development. Various AD risk genes, including PSEN1, PICALM, CLU, TREM2 have been shown to modulate autophagy. Therefore, promoting autophagy to augment the elimination of misfolded proteins is recommended to be an opportunity for AD therapy.290
Though the abnormal accumulation of lipids has been described in the very first report of AD neuropathology,295,296 it has not been until recently that lipid homeostasis impairment has become a focus of AD research.297 Lipidomic and metabolomic studies have consistently exhibited alterations in the levels of various lipid classes emerging in early stages of AD brains. Multidimensional interactions between lipid metabolism and key AD pathogenic pathways including amyloidogenesis, bioenergetic deficit, oxidative stress, neuroinflammation, and myelin degeneration have been revealed.297
With respect to the relative growth in the number of documents correlating AD with various aging hallmarks, telomere attrition exhibits the highest growth, followed by the dysregulated nutrient sensing (Figure B). Indeed, the research of telomere biology is one of the most invested areas of research in AD prevention and treatment, due to its involvement in many age-related diseases.298,299 The presence of shorter telomeres in multiple somatic samples from AD patients, especially in leukocytes, has been reported.300 It has been suggested that the telomere shortening in peripheral leukocytes might be explained by certain known AD features on a molecular level such as high inflammatory cytokine levels and oxidative stress.301−303
Deregulated nutrient sensing is increasingly thought to play a role in the pathophysiology of neurodegenerative diseases such as AD.304−306 Nutrient sensing is increasingly emerging both as a key modulator of neurogenesis, and, through mTOR, of the autophagic process.307,308 The extensive role that deregulated nutrient sensing may play in dementia offers a possible therapeutic pathway, as many nutrient sensing-modulating therapeutics already exist.309
Approved Drugs
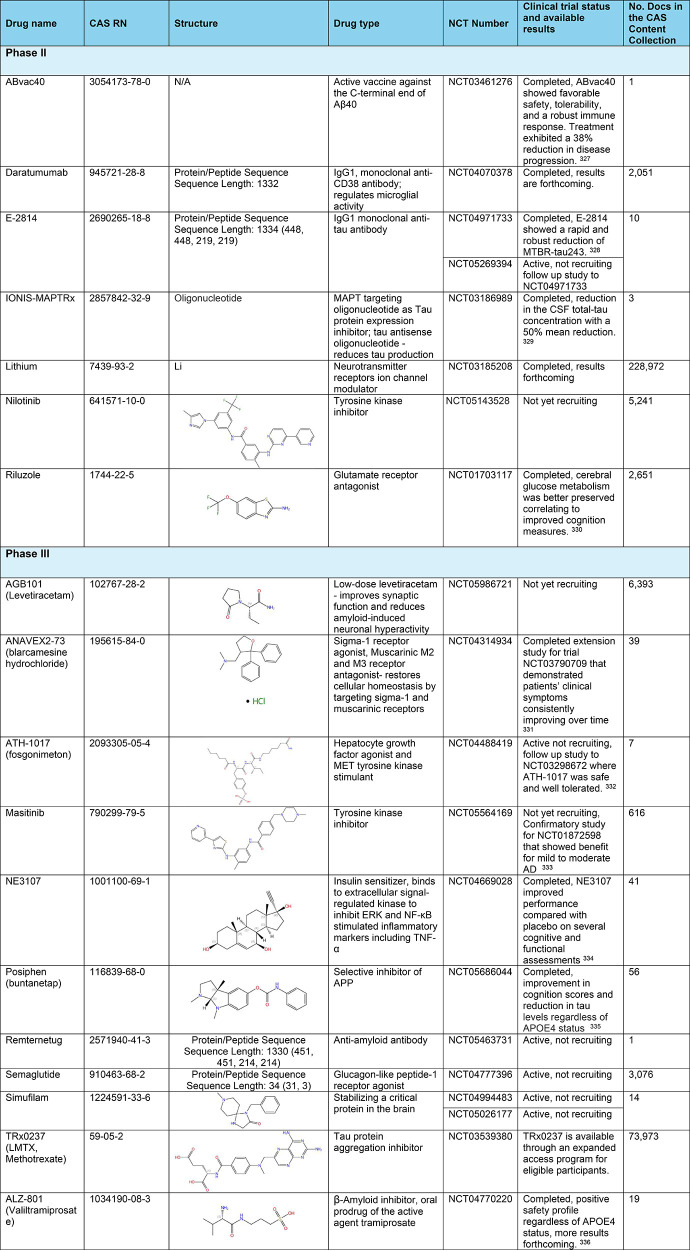
Several prescription drugs have been approved by the US FDA for Alzheimer’s disease to help either manage the symptoms of or to treat the disease progression (Table 1), plus some in late phases of clinical trials (Table 2). The majority of the FDA-approved drugs work best for people in the early stages of Alzheimer’s disease. There are currently no known interventions that will cure Alzheimer’s disease.
Table 2: Exemplary Clinical Trials Phase II and III of Anti-Alzheimer Therapeutics327−336
Cholinesterase inhibitors are prescribed to treat symptoms associated with memory, thinking, language, judgment and other thinking processes. These medications decrease the breakdown of acetylcholine, a chemical messenger important for memory and learning. These drugs support communication between nerve cells.120,124Glutamate regulators are prescribed to improve memory, attention, thinking, language and the ability to perform simple tasks. This type of drug works regulate the activity of glutamate, a chemical messenger that helps the brain process information.120,124,310Cholinesterase inhibitor + glutamate regulator is a type of drug for combination therapy.120,124Orexin receptor antagonist inhibits the activity of orexin, a type of neurotransmitter involved in the sleep–wake cycle.120,124Atypical antipsychotics are drugs that target the serotonin and dopamine chemical pathways in the brain. They are mainly used to treat schizophrenia and bipolar disorder and as add-on therapies for depressive disorders. These medications are also used to treat dementia-related behaviors.120,124Antiamyloids are the recent hope and promise in AD treatment.142,311 They work by attaching to and removing a protein that accumulates into plaques, β-amyloid, from the brain. These monoclonal antibody drugs target β-amyloid at a different stage of plaque formation (Figure ). However, the results of the ongoing clinical trials are still controversial, and for the time being the benefits of these drugs seem to be harder to quantify than potential harms.312,313
- Donepezil (Aricept) – approved to treat all stages of AD
- Rivastigmine (Exelon) – approved for mild-to-moderate AD
- Galantamine (Razadyne) – approved for mild-to-moderate AD
- Alpha-1062 (galantamine prodrug) – FDA New Drug Application acceptance for mild-to-moderate AD
- Memantine (Namenda) – approved for moderate-to-severe AD. It is an antagonist of the N-methyl-d-aspartate receptor (NMDA) subtype of glutamate receptor. By blocking the NMDA receptors, it helps regulate the activity of glutamate, preventing the overstimulation of these receptors and thereby protecting neurons from excitotoxicity.125
- Donepezil + Memantine (Namzaric) – approved for moderate-to-severe AD.
- Suvorexant (Belsomra) – approved for treatment of insomnia, effective for patients with mild to moderate AD.
- Brexpiprazole (Rexulti) – approved for treatment of agitation associated with dementia due to AD.
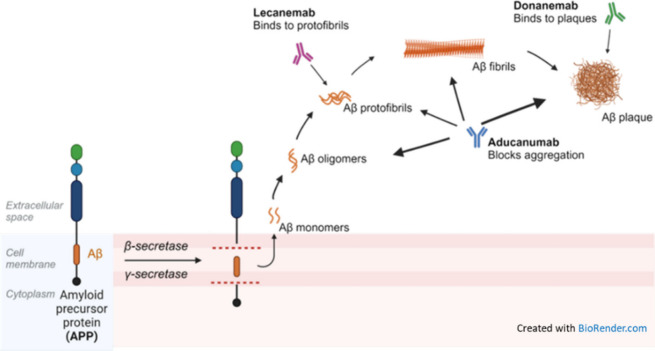
- Aducanumab (Aduhelm) is an antiamyloid antibody, which received accelerated FDA approval for treatment of early AD in 2021.314 It has been the first medication to demonstrate that removing β-amyloid from the brain reduces cognitive and functional decline. Aducanumab is being discontinued by its manufacturer, Biogen. The company stated that this decision is not related to any safety or efficacy concerns, but rather to reallocating resources to other Alzheimer’s disease treatments.315 In fact, the approval of aducanumab was controversial due to mixed results in clinical trials regarding its efficacy in slowing cognitive decline, as well as regulatory and advisory criticisms, economic challenges, and limited market uptake.
- Lecanemab (Leqembi) is a humanized antiamyloid IgG1 monoclonal antibody that binds with high affinity to Aβ soluble protofibrils, received traditional FDA approval for the treatment of early AD.316 It is the second therapy to demonstrate that removing β-amyloid from the brain reduces cognitive and functional decline in patients living with early AD and the first to show clinical benefits.317,318 Latest data have shown that lecanemab reduces markers of amyloid in early Alzheimer’s disease and results in moderately lower decline on measures of cognition and function than placebo at 18 months.319 However, its use comes with notable risks and limitations, including side effects like Amyloid-Related Imaging Abnormalities (ARIA), high costs, and uncertain long-term efficacy. Longer trials are warranted to determine the efficacy and safety of lecanemab in early Alzheimer’s disease.319
- Donanemab (Kisunla) is an antiamyloid antibody, approved by the FDA for the treatment of early AD in patients with mild cognitive impairment or mild dementia.320,321 Application for full FDA approval initially took place in 2023 with the FDA granting final approval July 2024.322 It targets β-amyloid plaque, reducing the excess protein reducing cognitive and functional decline.320 As with lecanemab, donanemab has safety concerns including ARIA especially among patients who are ApoE ε4 homozygotes, high costs, and its limitation to the treatment of patients with early disease activity. Continued clinical trials are needed to assess and monitor both safety and efficacy.
Drugs in Clinical Trials
Clinical trials researching the treatment of Alzheimer’s disease are explored in this section to gain an overall view of the past and current state of clinical development. Over 2200 clinical trials have been registered on clinicaltrials.gov over the last 20 years for Alzheimer’s diseases, reinforcing a strong interest in clinical development of treatments for this devastating disease. Figure . shows an increasing oscillating curve starting at around 40 clinical trials and raising to around 200 clinical trials per year, between the years 2003 to 2023.
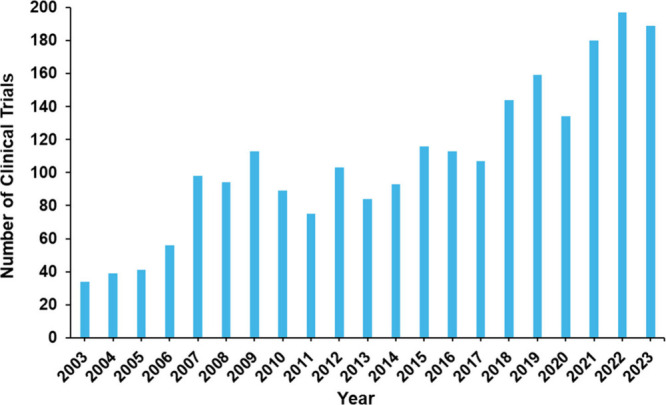
Analysis of Alzheimer’s disease therapeutic clinical trials reveals that around 41% of all trials are not phased (Figure A), including trials such as ones testing medical devices or behavioral interventions. The phase that contains the next largest group of trials is Phase I and Phase II studies, researching the safety and efficacy of newer anti-Alzheimer’s disease agents. Over half of all clinical trials in the past 10 years have been completed (Figure B). The status with the next largest group of trials is the recruiting status which is encouraging as new clinical trials are created and carried out to research the treatment of Alzheimer’s disease, offering hope to patients worldwide.
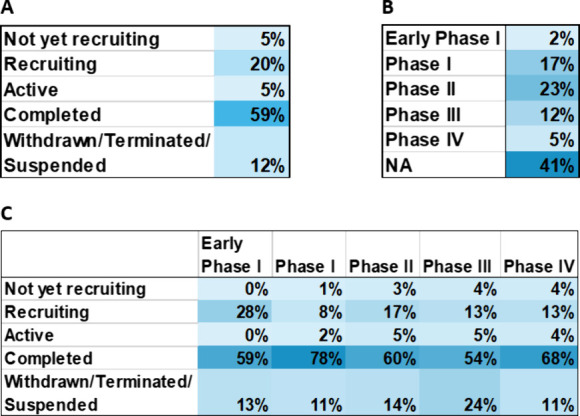
Exemplary Phase II and Phase III clinical trials and their studied drugs are highlighted in in Table 2 along with their CAS RN, structure, drug type, and number of publications in the CAS Content Collection.
Drug Repurposing
Since there is no cure for AD, drug repurposing studies have been intensely searching to identify existing drugs that could be repositioned to treat AD.337−342 As pharmaceutical development process is both time-consuming and costly, drug repurposing provides a chance to accelerate it by exploring the AD-related effects of agents approved for other disorders. These drugs have established safety profiles, pharmacokinetic description, formulations, dosages, and manufacturing procedures. Recently, in silico pharmacology has been widely applied and various computer applications including machine learning and artificial intelligence approaches have been explored in identifying potential drugs for repurposing to AD.343−346 Drug repurposing has already been attempted in AD with various methodologies applied and several clinical trials are currently evaluating drug-repurposing candidates for AD.337,347−349 Fifty widely discussed drugs for AD repurposing337−349 are summarized in Table 3.
Table 3: Exemplary Drugs Commonly Considered for Repurposing to AD, along with the Number of Documents in the CAS Content Collection Related to Them
| drug | CAS RN | no. docs | drug type |
|---|---|---|---|
| Lithium | 7439-93-2 | 958 | psychiatric |
| Metformin | 657-24-9 | 741 | antidiabetic |
| Risperidone | 106266-06-2 | 704 | antipsychotic |
| Nicotinamide | 98-92-0 | 693 | vitamin |
| Atorvastatin | 134523-00-5 | 574 | cardiovascular |
| Tamoxifen | 10540-29-1 | 503 | anticancer |
| Clozapine | 5786-21-0 | 437 | antipsychotic |
| Pioglitazone | 111025-46-8 | 436 | antidiabetic |
| Tacrolimus | 104987-11-3 | 416 | immunologic |
| Aripiprazole | 129722-12-9 | 332 | antipsychotic |
| Dronabinol | 1972-08-3 | 326 | appetite stimulant, antiemetic |
| Riluzole | 1744-22-5 | 306 | neurologic |
| Verapamil | 52-53-9 | 305 | Ca-channel blocker, antihypertensive |
| Venlafaxine | 93413-69-5 | 291 | psychiatric |
| Escitalopram | 128196-01-0 | 268 | psychiatric |
| Bromocriptine | 25614-03-3 | 260 | dopamine agonist |
| Bupropion | 34911-55-2 | 257 | antidepressant |
| Liraglutide | 204656-20-2 | 253 | antidiabetic |
| Levetiracetam | 102767-28-2 | 246 | Neurologic, antiepileptic |
| Methylphenidate | 113-45-1 | 231 | psychiatric |
| Mirtazapine | 85650-52-8 | 230 | psychiatric |
| Raloxifene | 84449-90-1 | 229 | estrogen receptor modulator |
| Thalidomide | 50-35-1 | 225 | immunomodulator |
| Vorinostat | 149647-78-9 | 215 | anticancer |
| Losartan | 114798-26-4 | 188 | cardiovascular |
| Leuprolide | 53714-56-0 | 188 | hormonal |
| Sunitinib | 557795-19-4 | 181 | anticancer |
| Lenalidomide | 191732-72-6 | 176 | anticancer, hematologic |
| Nilotinib | 641571-10-0 | 176 | anticancer |
| Amlodipine | 88150-42-9 | 173 | cardiovascular |
| Brexanolone | 516-54-1 | 169 | psychiatric |
| Zidovudine | 30516-87-1 | 159 | HIV antiviral |
| Telmisartan | 144701-48-4 | 150 | cardiovascular |
| Zolpidem | 82626-48-0 | 148 | neurologic |
| Prazosin | 19216-56-9 | 133 | cardiovascular |
| Cilostazol | 73963-72-1 | 113 | hematologic |
| Candesartan | 139481-59-7 | 108 | cardiovascular |
| Allopurinol | 315-30-0 | 105 | uric acid reducer |
| Deferiprone | 30652-11-0 | 100 | hematologic |
| Salsalate | 552-94-3 | 98 | anti-inflammatory |
| Montelukast | 158966-92-8 | 95 | anti-inflammatory |
| Vandetanib | 443913-73-3 | 92 | anticancer |
| Efavirenz | 154598-52-4 | 92 | HIV antiviral |
| Perindopril | 82834-16-0 | 87 | cardiovascular |
| Zopiclone | 43200-80-2 | 78 | neurologic |
| Valacyclovir | 124832-26-4 | 76 | antiviral |
| Dabigatran | 211914-51-1 | 69 | hematologic |
| Sodium phenylbutyrate | 1716-12-7 | 61 | anticancer, cystic fibrosis |
| Dapagliflozin | 461432-26-8 | 60 | antidiabetic |
We identified over 700 documents in CAS Content Collection discussing drug repurposing for AD. The number of AD drug repurposing publications increased dramatically over the past decade–from 13 documents in 2013 to 147 in 2023 (Figure B). The distribution of the considered potential repurposed drug classes with respect to their original disease targets according to the CAS Content Collection is illustrated in Figure A. Predictably, drugs for repurposing to AD are most often searched within the pool of nervous system agents. Anticancer drugs, metabolic agents, and immunomodulators are also between the hopeful drugs for repurposing to AD (Figure A).
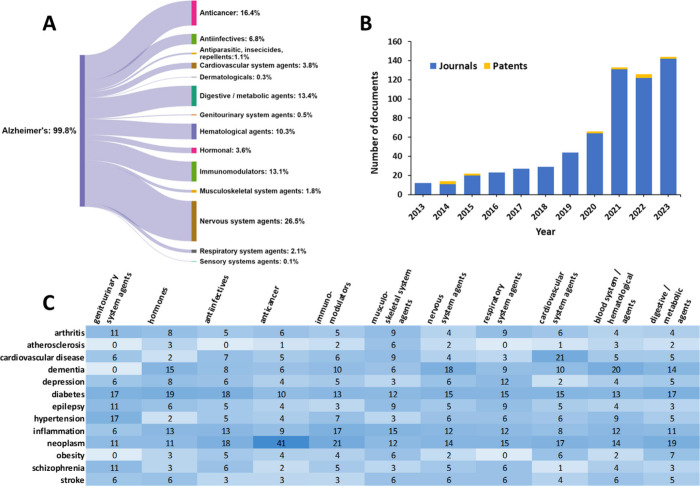
We further examined the correlation between diseases that co-occur with AD and the exploration of drug repurposing. The correlation between the concepts related to diseases co-occurring with AD and the exploring classes of drugs for repurposing to AD is illustrated in Figure C. In addition to certain obvious correlations such as cancers (neoplasm) – anticancer agents and cardiovascular diseases–cardiovascular system agents, there are some worth mentioning:
Dementia—Hematological agents
Studies have implicated vascular disorders as risk factors for dementia. Biological and epidemiological evidence have been reported concerning the role of certain hematologic factors such as homocysteine, cholesterol, fatty acids, antioxidants, and C-reactive protein in dementia.350 Abnormal (low or high) levels of hemoglobin have been associated with an enhanced risk of dementia, including AD, which may be associated with differences in white matter integrity.351 Association has been reported between blood leukocyte counts and increased risk of AD.352
Diabetes—Hormones
An imbalance in sex hormones has been identified as having a critical impact on type 2 diabetes. Androgens have a noteworthy sex-dimorphic association with type 2 diabetes, since hyperandrogenism in females and hypogonadism in males are risk factors for type 2 diabetes. Thus, treatments aimed at correcting these sex hormone imbalance may prevent the development of type 2 diabetes or help in its treatment.353
Inflammation—Immunomodulators
Inflammation is a central part of autoimmune diseases, which are caused by dysregulation of the immune system, involving imbalance between pro-inflammatory vs anti-inflammatory mediators.354
Neoplasm—Immunomodulators
Immunotherapy is treatment that uses your body’s own immune system to help fight cancer. Many immunotherapeutic agents operate by activating an efficient antitumor response or reversing tumor-mediated immunosuppression through modulation of significant regulatory pathways.355
Since AD is a complex neurodegenerative disorder, individuals with AD often have other health conditions or comorbidities. Some of these comorbidities may share underlying biological pathways or mechanisms with AD. By examining diseases that commonly co-occur with Alzheimer’s, researchers may identify potential candidate drugs for repurposing. Overall, understanding the relationship between comorbidities of AD and the potential for drug repurposing can provide valuable insights into the development of new treatments or therapeutic strategies for this devastating condition. Exploration of drug repurposing for AD involves leveraging the shared pathophysiological mechanisms and existing drug libraries of comorbid conditions, employing a combination of biological and computational approaches, and conducting rigorous clinical trials to validate the efficacy and safety of repurposed drugs.
Future Directions of Research
Research on Alzheimer’s disease is a dynamic field, with ongoing efforts aimed at understanding the underlying causes of the disease, developing effective treatments, and ultimately finding a cure. Understanding the molecular and cellular mechanisms underlying AD, including the role of β-amyloid plaques, tau protein tangles, neuroinflammation, and synaptic dysfunction, is a major focus of research. Advances in techniques such as imaging, genetics, and molecular biology are providing insights into the early stages of AD and potential targets for intervention.
Identifying reliable biomarkers for AD, including blood-based markers, cerebrospinal fluid markers, and imaging biomarkers, is crucial for early detection, accurate diagnosis, and monitoring disease progression. Recently, the first Alzheimer’s treatments targeting disease pathophysiology became available to patients and accelerated this need. Research is ongoing to develop new and more sensitive biomarkers that can detect AD pathology in its earliest stages, even before symptoms appear, when treatments are most effective.113,356 Noninvasive diagnostic tests are showing great promise in detecting β-amyloid and tau protein biomarkers equivalent or superior to FDA-approved CSF tests in the detection of AD pathology.111 These diagnostic assays, more easily accessible, will help alleviate the bottleneck and unmet needs of healthcare professionals and patients in evaluating them for AD.
Drug development efforts are focused on targeting various aspects of AD pathology, including reducing β-amyloid production or aggregation, inhibiting tau protein accumulation, modulating neuroinflammation, and promoting synaptic function and neuronal survival. Immunotherapy approaches, such as monoclonal antibodies targeting β-amyloid or tau proteins, are being investigated in clinical trials as potential disease-modifying treatments. Combination therapies targeting multiple pathways involved in AD pathogenesis are also being explored.
Advances in genetics and personalized medicine are leading to a better understanding of individual differences in AD risk, progression, and response to treatment. Precision medicine approaches aim to tailor treatments and interventions to the specific genetic and biological characteristics of each individual, potentially improving treatment efficacy and minimizing side effects.
Research continues to explore the efficacy of nonpharmacological interventions, such as cognitive training, physical exercise, diet, and lifestyle modifications, in reducing the risk of AD and improving cognitive function and overall brain health. Multimodal interventions combining various lifestyle factors, such as diet, exercise, cognitive stimulation, and social engagement, are being studied for their potential synergistic effects on brain health.
The analysis of large-scale data sets, including genetic data, clinical data, imaging data, and digital biomarkers, using advanced computational techniques such as machine learning and artificial intelligence, holds promise for uncovering new insights into AD and predicting disease progression. Collaborative initiatives such as the AD Neuroimaging Initiative (ADNI)357 and the Global Alzheimer’s Association Interactive Network (GAAIN)358 facilitate data sharing and collaboration among researchers worldwide.
While significant challenges remain in the fight against AD, sustained efforts in research, advocacy, and care have the potential to improve outcomes for individuals affected by the disease and advance our collective efforts toward a future without Alzheimer’s.
References
- E. Veljkovic, W. Xia, B. Phillips, E. T. Wong, J. Ho, A. Oviedo, J. Hoeng, M. Peitsch, E. Veljkovic, W. Xia, B. Phillips, E. T. Wong, J. Ho, A. Oviedo, J. Hoeng, M. Peitsch. Chapter 2 – Alzheimer’s Disease. In. Nicotine and Other Tobacco Compounds in Neurodegenerative and Psychiatric Diseases;, 2018
- A. V. Savonenko, T. Melnikova, T. Li, D. L. Price, P. C. Wong, M. J. Zigmond, L. P. Rowland, J. T. Coyle. Chapter 21 – Alzheimer Disease. In. Neurobiology of Brain Disorders;, 2015
- A. E. Budson, P. R. Solomon, A. E. Budson, P. R. Solomon. Chapter 3 – Alzheimer’s disease. In. Memory Loss;, 2011
- J. G. F. Boughey, N. R. Graff-Radford, A. H. V. Schapira, E. Byrne, S. DiMauro, R. S. J. Frackowiak, R. T. Johnson, Y. Mizuno, M. A. Samuels, S. D. Silberstein, Z. K. Wszolek. CHAPTER 65 – ALZHEIMER’S DISEASE. In. Neurology and Clinical Neuroscience;, 2007
- M. J. Murray, C. F. Murray, J. L. Atlee. Chapter 118 – Alzheimer’s Disease. In. Complications in Anesthesia (, 2007
- R. S. Doody, R. T. Johnson, J. W. Griffin, J. C. McArthur. Alzheimer’s Disease. In. Current Therapy in Neurologic Disease (, 2006
- 2023 Alzheimer’s disease facts and figures.. Alzheimer’s Dementia, 2023
- Alzheimer’s Disease Fact Sheet.
- What Happens to the Brain in Alzheimer’s Disease?
- R. A. Armstrong. Risk factors for Alzheimer’s disease.. Folia Neuropathol, 2019. [DOI | PubMed]
- M. P. Murphy, H. LeVine. Alzheimer’s disease and the amyloid-beta peptide.. J. Alzheimers Dis, 2010. [DOI | PubMed]
- M. Liu, T. Dexheimer, D. Sui, S. Hovde, X. Deng, R. Kwok, D. A. Bochar, M.-H. Kuo. Hyperphosphorylated tau aggregation and cytotoxicity modulators screen identified prescription drugs linked to Alzheimer’s disease and cognitive functions.. Sci. Rep., 2020. [DOI | PubMed]
- Medical Tests for Diagnosing Alzheimer’s.
- I. Ciccone. A Caregiver’s Perspective on Embracing Love for Patients With Alzheimer Disease.
- CAS Content Collection.
- CAS REGISTRY.
- SciFinder-n.
- CAS STNext.
- Dementia.. 2023
- What Causes Alzheimer’s Disease?
- Alzheimer’s disease.
- J. Hardy, D. J. Selkoe. The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics.. Science (New York, N.Y.), 2002. [DOI | PubMed]
- J. A. Hardy, G. A. Higgins. Alzheimer’s disease: the amyloid cascade hypothesis.. Science (New York, N.Y.), 1992. [DOI | PubMed]
- C. Patterson, J. W. Feightner, A. Garcia, G.-Y. R. Hsiung, C. MacKnight, A. D. Sadovnick. Diagnosis and treatment of dementia: 1. Risk assessment and primary prevention of Alzheimer disease.. Canadian Medical Association Journal, 2008. [DOI | PubMed]
- K. Iqbal, F. Liu, C. X. Gong, I. Grundke-Iqbal. Tau in Alzheimer disease and related tauopathies.. Curr. Alzheimer Res., 2010. [DOI | PubMed]
- H. Braak, K. Del Tredici, K. Kompoliti, L. V. Metman. Neurofibrillary Tangles. In. Encyclopedia of Movement Disorders;, 2010
- S. A. Youssef, J. L. Ram, P. M. Conn. Chapter 66 – Pathology of Brain Aging and Animal Models of Neurodegenerative Diseases. In. Conn’s Handbook of Models for Human Aging (, 2018
- R. Rajmohan, P. H. Reddy. Amyloid-Beta and Phosphorylated Tau Accumulations Cause Abnormalities at Synapses of Alzheimer’s disease Neurons.. J. Alzheimers Dis, 2017. [DOI | PubMed]
- H. Chi, H. Y. Chang, T. K. Sang. Neuronal Cell Death Mechanisms in Major Neurodegenerative Diseases.. Int. J. Mol. Sci., 2018. [DOI | PubMed]
- G. S. Bloom. Amyloid-β and Tau: The Trigger and Bullet in Alzheimer Disease Pathogenesis.. JAMA Neurology, 2014. [DOI | PubMed]
- A. Pasieka, D. Panek, N. Szałaj, A. Espargaró, A. Więckowska, B. Malawska, R. Sabaté, M. Bajda. Dual Inhibitors of Amyloid-β and Tau Aggregation with Amyloid-β Disaggregating Properties: Extended In Cellulo, In Silico, and Kinetic Studies of Multifunctional Anti-Alzheimer’s Agents.. ACS Chem. Neurosci., 2021. [DOI | PubMed]
- R. Diaz-Espinoza. Recent High-Resolution Structures of Amyloids Involved in Neurodegenerative Diseases.. Front Aging Neurosci, 2021. [DOI | PubMed]
- B. Chen, B. Marquez-Nostra, E. Belitzky, T. Toyonaga, J. Tong, Y. Huang, Z. Cai. PET Imaging in Animal Models of Alzheimer’s Disease.. Front Neurosci, 2022. [DOI | PubMed]
- M. A. G. Gilbert, N. Fatima, J. Jenkins, T. J. O’Sullivan, A. Schertel, Y. Halfon, M. Wilkinson, T. H. J. Morrema, M. Geibel, R. J. Read. CryoET of β-amyloid and tau within postmortem Alzheimer’s disease brain.. Nature, 2024. [DOI | PubMed]
- M. Zielinski, F. S. Peralta Reyes, L. Gremer, S. Schemmert, B. Frieg, L. U. Schäfer, A. Willuweit, L. Donner, M. Elvers, L. N. G. Nilsson. Cryo-EM of Aβ fibrils from mouse models find tg-APPArcSwe fibrils resemble those found in patients with sporadic Alzheimer’s disease.. Nature Neuroscience, 2023. [DOI | PubMed]
- K. Sharma, F. Stockert, J. Shenoy, M. Berbon, M. B. Abdul-Shukkoor, B. Habenstein, A. Loquet, M. Schmidt, M. Fändrich. Cryo-EM observation of the amyloid key structure of polymorphic TDP-43 amyloid fibrils.. Nat. Commun., 2024. [DOI | PubMed]
- N. Rodina, S. Hornung, R. Sarkar, S. Suladze, C. Peters, P. W. N. Schmid, Z. Niu, M. Haslbeck, J. Buchner, A. Kapurniotu. Modulation of Alzheimer’s Disease Aβ40 Fibril Polymorphism by the Small Heat Shock Protein αB-Crystallin.. J. Am. Chem. Soc., 2024. [DOI | PubMed]
- A. A. Bashit, P. Nepal, T. Connors, D. H. Oakley, B. T. Hyman, L. Yang, L. Makowski. Mapping the Spatial Distribution of Fibrillar Polymorphs in Human Brain Tissue.. Front Neurosci, 2022. [DOI | PubMed]
- C. Jie, V. Treyer, R. Schibli, L. Mu. Tauvid: The First FDA-Approved PET Tracer for Imaging Tau Pathology in Alzheimer’s Disease.. Pharmaceuticals, 2021. [DOI | PubMed]
- Y. Cai, J. Liu, B. Wang, M. Sun, H. Yang. Microglia in the Neuroinflammatory Pathogenesis of Alzheimer’s Disease and Related Therapeutic Targets.. Front Immunol, 2022. [DOI | PubMed]
- H. S. Kwon, S.-H. Koh. Neuroinflammation in neurodegenerative disorders: the roles of microglia and astrocytes.. Translational Neurodegeneration, 2020. [DOI | PubMed]
- A. Badimon, D. Torrente, E. H. Norris. Vascular Dysfunction in Alzheimer’s Disease: Alterations in the Plasma Contact and Fibrinolytic Systems.. Int. J. Mol. Sci., 2023. [DOI | PubMed]
- K. Govindpani, L. G. McNamara, N. R. Smith, C. Vinnakota, H. J. Waldvogel, R. L. Faull, A. Kwakowsky. Vascular Dysfunction in Alzheimer’s Disease: A Prelude to the Pathological Process or a Consequence of It?.. J. Clin. Med., 2019. [DOI | PubMed]
- Y. P. Tang, E. S. Gershon. Genetic studies in Alzheimer’s disease.. Dialogues Clin Neurosci, 2003. [DOI | PubMed]
- H. M. Lanoiselée, G. Nicolas, D. Wallon, A. Rovelet-Lecrux, M. Lacour, S. Rousseau, A. C. Richard, F. Pasquier, A. Rollin-Sillaire, O. Martinaud. APP, PSEN1, and PSEN2 mutations in early-onset Alzheimer disease: A genetic screening study of familial and sporadic cases.. PLoS Med., 2017. [DOI | PubMed]
- J. Wallensten, G. Ljunggren, A. Nager, C. Wachtler, N. Bogdanovic, P. Petrovic, A. C. Carlsson. Stress, depression, and risk of dementia – a cohort study in the total population between 18 and 65 years old in Region Stockholm.. Alzheimers Res. Ther, 2023. [DOI | PubMed]
- R. Botto, N. Callai, A. Cermelli, L. Causarano, I. Rainero. Anxiety and depression in Alzheimer’s disease: a systematic review of pathogenetic mechanisms and relation to cognitive decline.. Neurol Sci., 2022. [DOI | PubMed]
- S. Li, C. Wang, W. Wang, J. Tan. Trait anxiety, a personality risk factor associated with Alzheimer’s Disease.. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 2021. [DOI | PubMed]
- Cortisol and stress: What is the connection?
- M. A. Calcia, D. R. Bonsall, P. S. Bloomfield, S. Selvaraj, T. Barichello, O. D. Howes. Stress and neuroinflammation: a systematic review of the effects of stress on microglia and the implications for mental illness.. Psychopharmacology (Berl), 2016. [DOI | PubMed]
- J. Xie, L. Van Hoecke, R. E. Vandenbroucke. The Impact of Systemic Inflammation on Alzheimer’s Disease Pathology.. Front. Immunol., 2022. [DOI | PubMed]
- H. Akiyama, S. Barger, S. Barnum, B. Bradt, J. Bauer, G. M. Cole, N. R. Cooper, P. Eikelenboom, M. Emmerling, B. L. Fiebich. Inflammation and Alzheimer’s disease.. Neurobiol Aging, 2000. [DOI | PubMed]
- M. Saeedi, A. Rashidy-Pour. Association between chronic stress and Alzheimer’s disease: Therapeutic effects of Saffron.. Biomedicine & Pharmacotherapy, 2021. [DOI | PubMed]
- P. Rawat, U. Sehar, J. Bisht, A. Selman, J. Culberson, P. H. Reddy. Phosphorylated Tau in Alzheimer’s Disease and Other Tauopathies.. International journal of molecular sciences, 2022. [DOI | PubMed]
- J. D. Bremner, M. Narayan, E. R. Anderson, L. H. Staib, H. L. Miller, D. S. Charney. Hippocampal volume reduction in major depression.. Am. J. Psychiatry, 2000. [DOI | PubMed]
- Y. I. Sheline. Depression and the hippocampus: cause or effect?.. Biol. Psychiatry, 2011. [DOI | PubMed]
- Clinical Depression (Major Depressive Disorder).
- S. W. Jeon, Y. K. Kim. Neuroinflammation and cytokine abnormality in major depression: Cause or consequence in that illness?.. World J. Psychiatry, 2016. [DOI | PubMed]
- W. Zhang, D. Xiao, Q. Mao, H. Xia. Role of neuroinflammation in neurodegeneration development.. Signal Transduction and Targeted Therapy, 2023. [DOI | PubMed]
- J. W. Kinney, S. M. Bemiller, A. S. Murtishaw, A. M. Leisgang, A. M. Salazar, B. T. Lamb. Inflammation as a central mechanism in Alzheimer’s disease.. Alzheimer’s Dementia: Transl. Res. Clin. Interventions, 2018. [DOI]
- J. Cloyd. Cortisol and Anxiety: Understanding the Connection.
- L. G. Appelbaum, M. A. Shenasa, L. Stolz, Z. Daskalakis. Synaptic plasticity and mental health: methods, challenges and opportunities.. Neuropsychopharmacology, 2023. [DOI | PubMed]
- C. E. Lyons, A. Bartolomucci. Stress and Alzheimer’s disease: A senescence link?.. Neurosci Biobehav Rev., 2020. [DOI | PubMed]
- G. Juszczyk, J. Mikulska, K. Kasperek, D. Pietrzak, W. Mrozek, M. Herbet. Chronic Stress and Oxidative Stress as Common Factors of the Pathogenesis of Depression and Alzheimer’s Disease: The Role of Antioxidants in Prevention and Treatment.. Antioxidants, 2021. [DOI | PubMed]
- A. D. G. Fedoce, F. Ferreira, R. G. Bota, V. Bonet-Costa, P. Y. Sun, K. J. A. Davies. The role of oxidative stress in anxiety disorder: cause or consequence?.. Free Radic Res., 2018. [DOI | PubMed]
- Hypothalamic-Pituitary-Adrenal (HPA) Axis.
- J. E. Ahlskog, Y. E. Geda, N. R. Graff-Radford, R. C. Petersen. Physical exercise as a preventive or disease-modifying treatment of dementia and brain aging.. Mayo Clin Proc., 2011. [DOI | PubMed]
- R. B. Price, R. Duman. Neuroplasticity in cognitive and psychological mechanisms of depression: an integrative model.. Mol. Psychiatry, 2020. [DOI | PubMed]
- B. Guo, M. Zhang, W. Hao, Y. Wang, T. Zhang, C. Liu. Neuroinflammation mechanisms of neuromodulation therapies for anxiety and depression.. Translational Psychiatry, 2023. [DOI | PubMed]
- E. Won, Y. K. Kim. Neuroinflammation-Associated Alterations of the Brain as Potential Neural Biomarkers in Anxiety Disorders.. Int. J. Mol. Sci., 2020. [DOI | PubMed]
- V. Patel, P. Edison. Cardiometabolic risk factors and neurodegeneration: a review of the mechanisms underlying diabetes, obesity and hypertension in Alzheimer’s disease.. Journal of Neurology, Neurosurgery & Psychiatry, 2024. [DOI | PubMed]
- M. Kivipelto, T. Ngandu, L. Fratiglioni, M. Viitanen, I. Kåreholt, B. Winblad, E.-L. Helkala, J. Tuomilehto, H. Soininen, A. Nissinen. Obesity and Vascular Risk Factors at Midlife and the Risk of Dementia and Alzheimer Disease.. Archives of Neurology, 2005. [DOI | PubMed]
- R. F. Gottesman, A. L. Schneider, Y. Zhou, J. Coresh, E. Green, N. Gupta, D. S. Knopman, A. Mintz, A. Rahmim, A. R. Sharrett. Association Between Midlife Vascular Risk Factors and Estimated Brain Amyloid Deposition.. Jama, 2017. [DOI | PubMed]
- R. A. A. de Heus, D. L. K. de Jong, M. L. Sanders, G. J. van Spijker, M. H. Oudegeest-Sander, M. T. Hopman, B. A. Lawlor, M. G. M. Olde Rikkert, J. Claassen. Dynamic Regulation of Cerebral Blood Flow in Patients With Alzheimer Disease.. Hypertension, 2018. [DOI | PubMed]
- P. Iso-Markku, U. M. Kujala, K. Knittle, J. Polet, E. Vuoksimaa, K. Waller. Physical activity as a protective factor for dementia and Alzheimer’s disease: systematic review, meta-analysis and quality assessment of cohort and case–control studies.. British Journal of Sports Medicine, 2022. [DOI | PubMed]
- K. Lang. Eating more plants and less meat may reduce risk of Alzheimer’s disease.
- MIND and Mediterranean diets linked to fewer signs of Alzheimer’s brain pathology.
- S. Kheirouri, F. Valiei, A.-A. Taheraghdam. Association of plant-rich dietary patterns of mediterranean and MIND with risk of alzheimer disease.. Human Nutrition & Metabolism, 2024. [DOI]
- R. S. Wilson, C. F. Mendes de Leon, L. L. Barnes, J. A. Schneider, J. L. Bienias, D. A. Evans, D. A. Bennett. Participation in Cognitively Stimulating Activities and Risk of Incident Alzheimer Disease.. JAMA, 2002. [DOI | PubMed]
- Reading, writing, and playing games delay Alzheimer’s by 5 years.
- M. Chin-Chan, J. Navarro-Yepes, B. Quintanilla-Vega. Environmental pollutants as risk factors for neurodegenerative disorders: Alzheimer and Parkinson diseases.. Front. Cell. Neurosci., 2015. [DOI]
- M. Yegambaram, B. Manivannan, T. G. Beach, R. U. Halden. Role of environmental contaminants in the etiology of Alzheimer’s disease: a review.. Curr. Alzheimer Res., 2015. [DOI | PubMed]
- M. Khanahmadi, D. D. Farhud, M. Malmir. Genetic of Alzheimer’s Disease: A Narrative Review Article.. Iran J. Public Health, 2015. [PubMed]
- Alzheimer’s Disease Genetics Fact Sheet.
- R. E. Tanzi. The genetics of Alzheimer disease.. Cold Spring Harb Perspect Med., 2012. [DOI | PubMed]
- P. Johns, P. Johns. Chapter 12 – Dementia. In. Clinical Neuroscience;, 2014
- R. B. Maccioni, J. P. Muñoz, L. Barbeito. The Molecular Bases of Alzheimer’s Disease and Other Neurodegenerative Disorders.. Archives of Medical Research, 2001. [DOI | PubMed]
- G. M. Martin, J. E. Birren. Premature Aging. In. Encyclopedia of Gerontology (, 2007
- M. Canevelli, P. Piscopo, G. Talarico, N. Vanacore, A. Blasimme, A. Crestini, G. Tosto, F. Troili, G. L. Lenzi, A. Confaloni. Familial Alzheimer’s disease sustained by presenilin 2 mutations: Systematic review of literature and genotype–phenotype correlation.. Neuroscience & Biobehavioral Reviews, 2014. [DOI | PubMed]
- Familial Alzheimer’s Disease.
- Alzheimer’s Disease Genetics Fact Sheet.
- Rare types of dementia caused by genetic changes.
- G. D. Rabinovici. Late-onset Alzheimer Disease.. Continuum (Minneap Minn), 2019. [DOI | PubMed]
- C. Reitz, E. Rogaeva, G. W. Beecham. Late-onset vs nonmendelian early-onset Alzheimer disease.. Neurology Genetics, 2020. [DOI | PubMed]
- J. Fortea, J. Pegueroles, D. Alcolea, O. Belbin, O. Dols-Icardo, L. Vaqué-Alcázar, L. Videla, J. D. Gispert, M. Suárez-Calvet, S. C. Johnson. APOE4 homozygozity represents a distinct genetic form of Alzheimer’s disease.. Nat. Med., 2024. [DOI | PubMed]
- INHERITING TWO COPIES OF APOE4 LINKED TO RISK OF ALZHEIMER’S AT A YOUNGER AGE, STUDY SUGGESTS.
- Study defines major genetic form of Alzheimer’s disease.
- M. P. Mattson. Late-onset dementia: a mosaic of prototypical pathologies modifiable by diet and lifestyle.. npj Aging Mech. Dis., 2015. [DOI | PubMed]
- A. G. Efthymiou, A. M. Goate. Late onset Alzheimer’s disease genetics implicates microglial pathways in disease risk.. Mol. Neurodegener., 2017. [DOI | PubMed]
- C. Duyckaerts, B. Delatour, M.-C. Potier. Classification and basic pathology of Alzheimer disease.. Acta Neuropathologica, 2009. [DOI | PubMed]
- D. M. Holtzman, J. C. Morris, A. M. Goate. Alzheimer’s disease: the challenge of the second century.. Sci. Transl. Med., 2011. [DOI]
- S. Takeda. Progression of Alzheimer’s disease.. tau propagation, and its modifiable risk factors. Neurosci Res., 2019. [DOI | PubMed]
- Stages of Alzheimer’s.
- Alzheimer’s stages: How the disease progresses.
- What are the stages of Alzheimer’s disease?
- Stages of Alzheimer’s Disease.
- N. Ciesielska, R. Sokołowski, E. Mazur, M. Podhorecka, A. Polak-Szabela, K. Kędziora-Kornatowska. Is the Montreal Cognitive Assessment (MoCA) test better suited than the Mini-Mental State Examination (MMSE) in mild cognitive impairment (MCI) detection among people aged over 60? Meta-analysis.. Psychiatr Pol, 2016. [DOI | PubMed]
- X. Jia, Z. Wang, F. Huang, C. Su, W. Du, H. Jiang, H. Wang, J. Wang, F. Wang, W. Su. A comparison of the Mini-Mental State Examination (MMSE) with the Montreal Cognitive Assessment (MoCA) for mild cognitive impairment screening in Chinese middle-aged and older population: a cross-sectional study.. BMC Psychiatry, 2021. [DOI | PubMed]
- J. S. Fasnacht, A. S. Wueest, M. Berres, A. E. Thomann, S. Krumm, K. Gutbrod, L. A. Steiner, N. Goettel, A. U. Monsch. Conversion between the Montreal Cognitive Assessment and the Mini-Mental Status Examination.. Journal of the American Geriatrics Society, 2023. [DOI | PubMed]
- Neurological Diagnostic Tests and Procedures.
- N. R. Barthélemy, G. Salvadó, S. E. Schindler, Y. He, S. Janelidze, L. E. Collij, B. Saef, R. L. Henson, C. D. Chen, B. A. Gordon. Highly accurate blood test for Alzheimer’s disease is similar or superior to clinical cerebrospinal fluid tests.. Nature Medicine, 2024. [DOI]
- J. Henderson. C2N Diagnostics Releases the PrecivityAD2 Blood Test for Clinical Care, A Robust Assay with High Concordance to Amyloid PET and CSF.
- D. Shea, E. Colasurdo, A. Smith, C. Paschall, S. Jayadev, C. D. Keene, D. Galasko, A. Ko, G. Li, E. Peskind. SOBA: Development and testing of a soluble oligomer binding assay for detection of amyloidogenic toxic oligomers.. Proc. Natl. Acad. Sci. U. S. A., 2022. [DOI | PubMed]
- T. A. Bodily, A. Ramanathan, S. Wei, A. Karkisaval, N. Bhatt, C. Jerez, M. A. Haque, A. Ramil, P. Heda, Y. Wang. In pursuit of degenerative brain disease diagnosis: Dementia biomarkers detected by DNA aptamer-attached portable graphene biosensor.. Proc. Natl. Acad. Sci. U. S. A., 2023. [DOI | PubMed]
- J. L. Cummings, B. Dubois, J. L. Molinuevo, P. Scheltens. International Work Group criteria for the diagnosis of Alzheimer disease.. Med. Clin North Am., 2013. [DOI | PubMed]
- National Institute on Aging and Alzheimer’s Association Lead Effort to Update Diagnostic Criteria for Alzheimer’s Disease.
- Alzheimer’s and dementia.
- K. G. Yiannopoulou, S. G. Papageorgiou. Current and Future Treatments in Alzheimer Disease: An Update.. J. Cent Nerv Syst. Dis, 2020. [DOI | PubMed]
- M. Saljoughian. Alzheimer’s Disease and Current Treatment.. U.S. Pharmacist, 2023
- How Is Alzheimer’s Disease Treated?
- Treatments for Alzheimer’s.
- Alzheimer’s Disease Treatments.
- G. T. Grossberg. Cholinesterase inhibitors for the treatment of Alzheimer’s disease:: getting on and staying on.. Curr. Ther Res. Clin Exp, 2003. [DOI | PubMed]
- Medications for Memory, Cognition and Dementia-Related Behaviors.
- B. C. Tang, Y. T. Wang, J. Ren. Basic information about memantine and its treatment of Alzheimer’s disease and other clinical applications.. Ibrain, 2023. [DOI | PubMed]
- J. Korabecny, F. Zemek, O. Soukup, K. Spilovska, K. Musilek, D. Jun, E. Nepovimova, K. Kuca, R. Atta ur, M. I. Choudhary. Chapter 1 – Pharmacotherapy of Alzheimer’s Disease: Current State and Future Perspectives. In. Drug Design and Discovery in Alzheimer’s Disease;, 2014
- U. Rashid, F. L. Ansari, R. Atta ur, M. I. Choudhary. Chapter 2 – Challenges in Designing Therapeutic Agents for Treating Alzheimer’s Disease-from Serendipity to Rationality. In. Drug Design and Discovery in Alzheimer’s Disease;, 2014
- Y. Xie, P.-P. Liu, Y.-J. Lian, H.-B. Liu, J.-S. Kang. The effect of selective serotonin reuptake inhibitors on cognitive function in patients with Alzheimer’s disease and vascular dementia: focusing on fluoxetine with long follow-up periods.. Signal Transduction Targeted Ther., 2019. [DOI]
- B. Mdawar, E. Ghossoub, R. Khoury. Selective serotonin reuptake inhibitors and Alzheimer’s disease.. Neural Regener. Res., 2020. [DOI]
- J. P.-C. Chang, A. Zamparelli, M. A. Nettis, C. M. Pariante, S. Della Sala. Antidepressant Drugs: Mechanisms of Action and Side Effects. In. Encyclopedia of Behavioral Neuroscience,, 2022
- Selective serotonin reuptake inhibitors (SSRIs).
- Commonly prescribed antidepressants and how they work.
- K. Heinzerling, P. M. Miller. Chapter 48 – Medication Development for Amphetamine Dependence. In. Interventions for Addiction,, 2013
- A. S. Correia, N. Vale. Antidepressants in Alzheimer’s Disease: A Focus on the Role of Mirtazapine.. Pharmaceuticals, 2021. [DOI | PubMed]
- J. Cummings, A. M. L. Osse, D. Cammann, J. Powell, J. Chen. Anti-Amyloid Monoclonal Antibodies for the Treatment of Alzheimer’s Disease. BioDrugs: clinical immunotherapeutics.. biopharmaceuticals and gene therapy, 2024. [DOI]
- Monoclonal Antibodies Directed Against Amyloid for the Treatment of Alzheimer’s Disease.
- W. Wu, Y. Ji, Z. Wang, X. Wu, J. Li, F. Gu, Z. Chen, Z. Wang. The FDA-approved anti-amyloid-β monoclonal antibodies for the treatment of Alzheimer’s disease: a systematic review and meta-analysis of randomized controlled trials.. Eur. J. Med. Res., 2023. [DOI | PubMed]
- I. S. Padda, M. Parmar. Aducanumab.
- Aducanumab to Be Discontinued as an Alzheimer’s Treatment.
- Lecanemab Approved for Treatment of Early Alzheimer’s Disease.
- Donanemab.
- Z. Zhao, Y. Liu, S. Ruan, Y. Hu. Current Anti-Amyloid-β Therapy for Alzheimer’s Disease Treatment: From Clinical Research to Nanomedicine.. Int. J. Nanomedicine, 2023. [DOI | PubMed]
- What Are Stimulating Activities for People with Alzheimer’s?
- S. Gottlieb. Mental activity may help prevent dementia.. BMJ, 2003. [DOI]
- S. A. Schultz, J. Larson, J. Oh, R. Koscik, M. N. Dowling, C. L. Gallagher, C. M. Carlsson, H. A. Rowley, B. B. Bendlin, S. Asthana. Participation in cognitively-stimulating activities is associated with brain structure and cognitive function in preclinical Alzheimer’s disease.. Brain Imaging Behav, 2015. [DOI | PubMed]
- S. Forstmeier, A. Maercker, E. Savaskan, T. Roth. Cognitive behavioural treatment for mild Alzheimer’s patients and their caregivers (CBTAC): study protocol for a randomized controlled trial.. Trials, 2015. [DOI | PubMed]
- L. D. Reid, F. E. Avens, A. A. Walf. Cognitive behavioral therapy (CBT) for preventing Alzheimer’s disease.. Behav Brain Res., 2017. [DOI | PubMed]
- A. M. González-Martín, A. Aibar Almazán, Y. Rivas Campo, N. Rodríguez Sobrino, Y. Castellote Caballero. Addressing depression in older adults with Alzheimer’s through cognitive behavioral therapy: systematic review and meta-analysis.. Front. Aging Neurosci., 2023. [DOI]
- B. Patel, M. Perera, J. Pendleton, A. Richman, B. Majumdar. Psychosocial interventions for dementia: from evidence to practice.. Advances in Psychiatric Treatment, 2014. [DOI]
- E. Howe. Key psychosocial interventions for Alzheimer’s disease: an update.. Psychiatry (Edgmont), 2008
- R. Guerreiro, J. Bras. The age factor in Alzheimer’s disease.. Genome Med., 2015. [DOI | PubMed]
- Y. Hou, X. Dan, M. Babbar, Y. Wei, S. G. Hasselbalch, D. L. Croteau, V. A. Bohr. Ageing as a risk factor for neurodegenerative disease.. Nature Reviews Neurology, 2019. [DOI | PubMed]
- C. López-Otín, M. A. Blasco, L. Partridge, M. Serrano, G. Kroemer. The hallmarks of aging.. Cell, 2013. [DOI | PubMed]
- C. López-Otín, M. A. Blasco, L. Partridge, M. Serrano, G. Kroemer. Hallmarks of aging: An expanding universe.. Cell, 2023. [DOI | PubMed]
- M. P. Mattson, T. V. Arumugam. Hallmarks of Brain Aging: Adaptive and Pathological Modification by Metabolic States.. Cell Metab, 2018. [DOI | PubMed]
- R. Tenchov, J. M. Sasso, X. Wang, Q. A. Zhou. Aging Hallmarks and Progression and Age-Related Diseases: A Landscape View of Research Advancement.. ACS Chem. Neurosci., 2024. [DOI | PubMed]
- H. Hillen. The Beta Amyloid Dysfunction (BAD) Hypothesis for Alzheimer’s Disease.. Front. Neurosci., 2019. [DOI | PubMed]
- G. G. Glenner, C. W. Wong. Alzheimer’s disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein.. Biochem. Biophys. Res. Commun., 1984. [DOI | PubMed]
- C. L. Masters, G. Simms, N. A. Weinman, G. Multhaup, B. L. McDonald, K. Beyreuther. Amyloid plaque core protein in Alzheimer disease and Down syndrome.. Proc. Natl. Acad. Sci. U. S. A., 1985. [DOI | PubMed]
- A. De Gramont, S. Watson, L. M. Ellis, J. Rodón, J. Tabernero, A. De Gramont, S. R. Hamilton. Pragmatic issues in biomarker evaluation for targeted therapies in cancer.. Nature reviews Clinical oncology, 2015. [DOI]
- A. Godfrey, B. Vandendriessche, J. P. Bakker, C. Fitzer-Attas, N. Gujar, M. Hobbs, Q. Liu, C. A. Northcott, V. Parks, W. A. Wood. Fit-for-purpose biometric monitoring technologies: leveraging the laboratory biomarker experience.. Clinical and translational science, 2021. [DOI | PubMed]
- K. B. Shanthi, S. Krishnan, P. Rani. A systematic review and meta-analysis of plasma amyloid 1–42 and tau as biomarkers for Alzheimer’s disease.. SAGE Open Med., 2015. [DOI | PubMed]
- C. E. Teunissen, M. J. Chiu, C. C. Yang, S. Y. Yang, P. Scheltens, H. Zetterberg, K. Blennow. Plasma Amyloid-β (Aβ42) Correlates with Cerebrospinal Fluid Aβ42 in Alzheimer’s Disease.. J. Alzheimers Dis, 2018. [DOI | PubMed]
- J. C. Lee, S. J. Kim, S. Hong, Y. Kim. Diagnosis of Alzheimer’s disease utilizing amyloid and tau as fluid biomarkers.. Exp. Mol. Med., 2019. [DOI]
- T. Tapiola, I. Alafuzoff, S.-K. Herukka, L. Parkkinen, P. Hartikainen, H. Soininen, T. Pirttilä. Cerebrospinal Fluid β-Amyloid 42 and Tau Proteins as Biomarkers of Alzheimer-Type Pathologic Changes in the Brain.. Archives of Neurology, 2009. [DOI | PubMed]
- P. J. Visser, L. M. Reus, J. Gobom, I. Jansen, E. Dicks, S. J. van der Lee, M. Tsolaki, F. R. J. Verhey, J. Popp, P. Martinez-Lage. Cerebrospinal fluid tau levels are associated with abnormal neuronal plasticity markers in Alzheimer’s disease.. Mol. Neurodegener., 2022. [DOI | PubMed]
- R. Medeiros, D. Baglietto-Vargas, F. M. LaFerla. The role of tau in Alzheimer’s disease and related disorders.. CNS Neurosci Ther, 2011. [DOI | PubMed]
- K. Blennow, H. Hampel, M. Weiner, H. Zetterberg. Cerebrospinal fluid and plasma biomarkers in Alzheimer disease.. Nat. Rev. Neurol, 2010. [DOI | PubMed]
- A. J. Aschenbrenner, Y. Li, R. L. Henson, K. Volluz, J. Hassenstab, P. Verghese, T. West, M. R. Meyer, K. M. Kirmess, A. M. Fagan. Comparison of plasma and CSF biomarkers in predicting cognitive decline.. Ann. Clin Transl Neurol, 2022. [DOI | PubMed]
- L. S. Honig, M. S. Kang, A. J. Lee, D. Reyes-Dumeyer, A. Piriz, B. Soriano, Y. Franco, Z. D. Coronado, P. Recio, D. R. Mejía. Evaluation of Plasma Biomarkers for A/T/N Classification of Alzheimer Disease Among Adults of Caribbean Hispanic Ethnicity.. JAMA Network Open, 2023. [DOI | PubMed]
- N. J. Ashton, A. Puig-Pijoan, M. Milà-Alomà, A. Fernández-Lebrero, G. García-Escobar, F. González-Ortiz, P. R. Kac, W. S. Brum, A. L. Benedet, J. Lantero-Rodriguez. Plasma and CSF biomarkers in a memory clinic: Head-to-head comparison of phosphorylated tau immunoassays.. Alzheimer’s Dementia, 2023. [DOI]
- P. K. Mandal, J. C. Maroon, A. Garg, N. K. Arora, R. Bansal, A. Kaushik, A. Samkaria, G. Kumaran, Y. Arora. Blood Biomarkers in Alzheimer’s Disease.. ACS Chem. Neurosci., 2023. [DOI | PubMed]
- O. Hansson, K. Blennow, H. Zetterberg, J. Dage. Blood biomarkers for Alzheimer’s disease in clinical practice and trials.. Nature Aging, 2023. [DOI | PubMed]
- D. Angioni, J. Delrieu, O. Hansson, H. Fillit, P. Aisen, J. Cummings, J. R. Sims, J. B. Braunstein, M. Sabbagh, T. Bittner. Blood Biomarkers from Research Use to Clinical Practice: What Must Be Done? A Report from the EU/US CTAD Task Force.. J. Prev. Alzheimer’s Dis, 2022. [DOI | PubMed]
- P. Inacio. Biomarker test to ID Alzheimer’s wins FDA breakthrough designation.
- S. Palmqvist, P. Tideman, N. Mattsson-Carlgren, S. E. Schindler, R. Smith, R. Ossenkoppele, S. Calling, T. West, M. Monane, P. B. Verghese. Blood Biomarkers to Detect Alzheimer Disease in Primary Care and Secondary Care.. JAMA., 2024. [DOI]
- V. V. Giau, E. Bagyinszky, Y. S. Yang, Y. C. Youn, S. S. A. An, S. Y. Kim. Genetic analyses of early-onset Alzheimer’s disease using next generation sequencing.. Sci. Rep., 2019. [DOI | PubMed]
- B. R. Troutwine, L. Hamid, C. R. Lysaker, T. A. Strope, H. M. Wilkins. Apolipoprotein E and Alzheimer’s disease.. Acta Pharmaceutica Sinica B, 2022. [DOI | PubMed]
- A.-C. Raulin, S. V. Doss, Z. A. Trottier, T. C. Ikezu, G. Bu, C.-C. Liu. ApoE in Alzheimer’s disease: pathophysiology and therapeutic strategies.. Molecular Neurodegeneration, 2022. [DOI | PubMed]
- H. G. Pemberton, L. E. Collij, F. Heeman, A. Bollack, M. Shekari, G. Salvadó, I. L. Alves, D. V. Garcia, M. Battle, C. Buckley. Quantification of amyloid PET for future clinical use: a state-of-the-art review.. Eur. J. Nucl. Med. Mol. Imaging, 2022. [DOI | PubMed]
- C. Maschio, R. Ni. Amyloid and Tau Positron Emission Tomography Imaging in Alzheimer’s Disease and Other Tauopathies.. Front Aging Neurosci, 2022. [DOI | PubMed]
- W. Bao, F. Xie, C. Zuo, Y. Guan, Y. H. Huang. PET Neuroimaging of Alzheimer’s Disease: Radiotracers and Their Utility in Clinical Research.. Front. Aging Neurosci., 2021. [DOI | PubMed]
- A. Chandra, G. Dervenoulas, M. Politis. Magnetic resonance imaging in Alzheimer’s disease and mild cognitive impairment.. J. Neurol, 2019. [DOI | PubMed]
- G. Lombardi, G. Crescioli, E. Cavedo, E. Lucenteforte, G. Casazza, A. G. Bellatorre, C. Lista, G. Costantino, G. Frisoni, G. Virgili. Structural magnetic resonance imaging for the early diagnosis of dementia due to Alzheimer’s disease in people with mild cognitive impairment.. Cochrane Database Syst. Rev., 2020. [DOI | PubMed]
- P. Vemuri, C. R. Jack. Role of structural MRI in Alzheimer’s disease.. Alzheimer’s Res. Ther., 2010. [DOI | PubMed]
- S. Janelidze, N. Mattsson, E. Stomrud, O. Lindberg, S. Palmqvist, H. Zetterberg, K. Blennow, O. Hansson. CSF biomarkers of neuroinflammation and cerebrovascular dysfunction in early Alzheimer disease.. Neurology, 2018. [DOI | PubMed]
- F. Angiulli, E. Conti, C. P. Zoia, F. Da Re, I. Appollonio, C. Ferrarese, L. Tremolizzo. Blood-Based Biomarkers of Neuroinflammation in Alzheimer’s Disease: A Central Role for Periphery?.. Diagnostics, 2021. [DOI | PubMed]
- A. Rauf, H. Badoni, T. Abu-Izneid, A. Olatunde, M. M. Rahman, S. Painuli, P. Semwal, P. Wilairatana, M. S. Mubarak. Neuroinflammatory Markers: Key Indicators in the Pathology of Neurodegenerative Diseases.. Molecules, 2022. [DOI | PubMed]
- Y. Jung, J. S. Damoiseaux. The potential of blood neurofilament light as a marker of neurodegeneration for Alzheimer’s disease.. Brain, 2024. [DOI | PubMed]
- G. Giacomucci, S. Mazzeo, S. Bagnoli, A. Ingannato, D. Leccese, V. Berti, S. Padiglioni, G. Galdo, C. Ferrari, S. Sorbi. Plasma neurofilament light chain as a biomarker of Alzheimer’s disease in Subjective Cognitive Decline and Mild Cognitive Impairment.. J. Neurol, 2022. [DOI | PubMed]
- J. A. Santiago, J. A. Potashkin. The Impact of Disease Comorbidities in Alzheimer’s Disease.. Front Aging Neurosci, 2021. [DOI | PubMed]
- J. A. Driver, A. Beiser, R. Au, B. E. Kreger, G. L. Splansky, T. Kurth, D. P. Kiel, K. P. Lu, S. Seshadri, P. A. Wolf. Inverse association between cancer and Alzheimer’s disease: results from the Framingham Heart Study.. BMJ., 2012. [DOI | PubMed]
- C. M. Roe, A. Fitzpatrick, C. Xiong, W. Sieh, L. Kuller, J. Miller, M. Williams, R. Kopan, M. I. Behrens, J. Morris. Cancer linked to Alzheimer disease but not vascular dementia.. Neurology, 2010. [DOI | PubMed]
- A. Zabłocka, W. Kazana, M. Sochocka, B. Stańczykiewicz, M. Janusz, J. Leszek, B. Orzechowska. Inverse Correlation Between Alzheimer’s Disease and Cancer: Short Overview.. Mol. Neurobiol, 2021. [DOI | PubMed]
- L. S. Honig, M.-X. Tang, S. Albert, R. Costa, J. Luchsinger, J. Manly, Y. Stern, R. Mayeux. Stroke and the Risk of Alzheimer Disease.. Archives of Neurology, 2003. [DOI | PubMed]
- J. Pinho, M. Quintas-Neves, I. Dogan, K. Reetz, A. Reich, A. S. Costa. Incident stroke in patients with Alzheimer’s disease: systematic review and meta-analysis.. Sci. Rep., 2021. [DOI | PubMed]
- C. A. Rojas-Carranza, R. H. Bustos-Cruz, C. J. Pino-Pinzon, Y. V. Ariza-Marquez, R. M. Gomez-Bello, M. Canadas-Garre. Diabetes-Related Neurological Implications and Pharmacogenomics.. Curr. Pharm. Des, 2018. [DOI | PubMed]
- H. J. Lee, H. I. Seo, H. Y. Cha, Y. J. Yang, S. H. Kwon, S. J. Yang. Diabetes and Alzheimer’s Disease: Mechanisms and Nutritional Aspects.. Clin Nutr Res., 2018. [DOI | PubMed]
- I. Zawar, J. Kapur. Does Alzheimer’s disease with mesial temporal lobe epilepsy represent a distinct disease subtype?.. Alzheimer’s Dementia, 2023. [DOI]
- CADRO: Common Alzheimer’s Disease Research Ontology.
- C. Liggins, H. M. Snyder, N. Silverberg, S. Petanceska, L. M. Refolo, L. Ryan, M. C. Carrillo. International Alzheimer’s Disease Research Portfolio (IADRP) aims to capture global Alzheimer’s disease research funding.. Alzheimers Dement, 2014. [DOI | PubMed]
- P. Smethurst, J. Newcombe, C. Troakes, R. Simone, Y. R. Chen, R. Patani, K. Sidle. In vitro prion-like behaviour of TDP-43 in ALS.. Neurobiol Dis, 2016. [DOI | PubMed]
- A. Meneses, S. Koga, J. O’Leary, D. W. Dickson, G. Bu, N. Zhao. TDP-43 Pathology in Alzheimer’s Disease.. Mol. Neurodegener., 2021. [DOI | PubMed]
- L. R. Hayes, P. Kalab. Emerging Therapies and Novel Targets for TDP-43 Proteinopathy in ALS/FTD.. Neurotherapeutics, 2022. [DOI | PubMed]
- A. Babazadeh, S. L. Rayner, A. Lee, R. S. Chung. TDP-43 as a therapeutic target in neurodegenerative diseases: Focusing on motor neuron disease and frontotemporal dementia.. Ageing Research Reviews, 2023. [DOI | PubMed]
- K. L. Youmans, B. Wolozin. TDP-43: A new player on the AD field?.. Exp. Neurol., 2012. [DOI | PubMed]
- Angiotensin-converting enzyme (ACE) inhibitors.
- High Blood Pressure and ACE Inhibitors.
- K. Zou, M. Michikawa. Angiotensin-converting enzyme as a potential target for treatment of Alzheimer’s disease: inhibition or activation?.. Rev. Neurosci, 2008. [DOI | PubMed]
- S. Liu, F. Ando, Y. Fujita, J. Liu, T. Maeda, X. Shen, K. Kikuchi, A. Matsumoto, M. Yokomori, C. Tanabe-Fujimura. A clinical dose of angiotensin-converting enzyme (ACE) inhibitor and heterozygous ACE deletion exacerbate Alzheimer’s disease pathology in mice.. J. Biol. Chem., 2019. [DOI | PubMed]
- J. Dorszewska, M. Prendecki, A. Oczkowska, M. Dezor, W. Kozubski. Molecular basis of familial and sporadic Alzheimer’s disease.. Current Alzheimer Research, 2016. [DOI | PubMed]
- A. I. Jarmolowicz, H.-Y. Chen, P. K. Panegyres. The patterns of inheritance in early-onset dementia: Alzheimer’s disease and frontotemporal dementia.. Am. J. Alzheimer’s Dis., 2015. [DOI]
- D. Campion, C. Dumanchin, D. Hannequin, B. Dubois, S. Belliard, M. Puel, C. Thomas-Anterion, A. Michon, C. Martin, F. Charbonnier. Early-onset autosomal dominant Alzheimer disease: prevalence genetic heterogeneity, and mutation spectrum.. Am. J. Hum. Genet., 1999. [DOI | PubMed]
- C. M. van Duijn, P. de Knijff, M. Cruts, A. Wehnert, L. M. Havekes, A. Hofman, C. V. Broeckhoven. Apolipoprotein E4 allele in a population–based study of early–onset Alzheimer’s disease.. Nature genetics, 1994. [DOI | PubMed]
- T. S. Wingo, J. J. Lah, A. I. Levey, D. J. Cutler. Autosomal recessive causes likely in early-onset Alzheimer disease.. Archives of neurology, 2012. [DOI | PubMed]
- Z. Breijyeh, R. Karaman. Comprehensive Review on Alzheimer’s Disease: Causes and Treatment.. Molecules, 2020. [DOI | PubMed]
- C. Van Cauwenberghe, C. Van Broeckhoven, K. Sleegers. The genetic landscape of Alzheimer disease: clinical implications and perspectives.. Genet Med., 2016. [DOI | PubMed]
- N. M. Li, K. F. Liu, Y. J. Qiu, H. H. Zhang, H. Nakanishi, H. Qing. Mutations of beta-amyloid precursor protein alter the consequence of Alzheimer’s disease pathogenesis.. Neural Regen Res., 2019. [DOI | PubMed]
- J. TCW, A. M. Goate. Genetics of β-Amyloid Precursor Protein in Alzheimer’s Disease.. Cold Spring Harbor Perspect. Med., 2017. [DOI]
- Y. Cai, S. S. An, S. Kim. Mutations in presenilin 2 and its implications in Alzheimer’s disease and other dementia-associated disorders.. Clin Interv Aging, 2015. [DOI | PubMed]
- C. C. Liu, T. Kanekiyo, H. Xu, G. Bu. Apolipoprotein E and Alzheimer disease: risk mechanisms and therapy.. Nat. Rev. Neurol, 2013. [DOI | PubMed]
- L. T. Nordestgaard, A. Tybjærg-Hansen, B. G. Nordestgaard, R. Frikke-Schmidt. Loss-of-function mutation in ABCA1 and risk of Alzheimer’s disease and cerebrovascular disease.. Alzheimers Dement, 2015. [DOI | PubMed]
- E. M. Foster, A. Dangla-Valls, S. Lovestone, E. M. Ribe, N. J. Buckley. Clusterin in Alzheimer’s Disease: Mechanisms Genetics, and Lessons From Other Pathologies.. Front. Neurosci, 2019. [DOI | PubMed]
- C. J. Holler, P. R. Davis, T. L. Beckett, T. L. Platt, R. L. Webb, E. Head, M. P. Murphy. Bridging integrator 1 (BIN1) protein expression increases in the Alzheimer’s disease brain and correlates with neurofibrillary tangle pathology.. J. Alzheimers Dis, 2014. [DOI | PubMed]
- R. J. Andrew, P. De Rossi, P. Nguyen, H. R. Kowalski, A. J. Recupero, T. Guerbette, S. V. Krause, R. C. Rice, L. Laury-Kleintop, S. L. Wagner. Reduction of the expression of the late-onset Alzheimer’s disease (AD) risk-factor BIN1 does not affect amyloid pathology in an AD mouse model.. J. Biol. Chem., 2019. [DOI | PubMed]
- Y. Yamazaki, M. M. Painter, G. Bu, T. Kanekiyo. Apolipoprotein E as a Therapeutic Target in Alzheimer’s Disease: A Review of Basic Research and Clinical Evidence.. CNS Drugs, 2016. [DOI | PubMed]
- A. Capell, H. Steiner, H. Romig, S. Keck, M. Baader, M. G. Grim, R. Baumeister, C. Haass. Presenilin-1 differentially facilitates endoproteolysis of the beta-amyloid precursor protein and Notch.. Nat. Cell Biol., 2000. [DOI | PubMed]
- A. Petit, F. Bihel, C. Alvès da Costa, O. Pourquié, F. Checler, J. L. Kraus. New protease inhibitors prevent gamma-secretase-mediated production of Abeta40/42 without affecting Notch cleavage.. Nat. Cell Biol., 2001. [DOI | PubMed]
- M. S. Wolfe. γ-Secretase and Presenilin.
- Y. Li, H. Xu, H. Wang, K. Yang, J. Luan, S. Wang. TREM2: Potential therapeutic targeting of microglia for Alzheimer’s disease.. Biomedicine & Pharmacotherapy, 2023. [DOI | PubMed]
- M. Gratuze, C. E. G. Leyns, D. M. Holtzman. New insights into the role of TREM2 in Alzheimer’s disease.. Mol. Neurodegener, 2018. [DOI | PubMed]
- D. Melchiorri, S. Merlo, B. Micallef, J.-J. Borg, F. Dráfi. Alzheimer’s disease and neuroinflammation: will new drugs in clinical trials pave the way to a multi-target therapy?.. Frontiers in Pharmacology, 2023. [DOI | PubMed]
- J. L. Cummings, G. Tong, C. Ballard. Treatment Combinations for Alzheimer’s Disease: Current and Future Pharmacotherapy Options.. J. Alzheimers Dis, 2019. [DOI | PubMed]
- A. Wang, P. Das, R. C. Switzer, T. E. Golde, J. L. Jankowsky. Robust amyloid clearance in a mouse model of Alzheimer’s disease provides novel insights into the mechanism of amyloid-beta immunotherapy.. J. Neurosci., 2011. [DOI | PubMed]
- D. Schenk, G. S. Basi, M. N. Pangalos. Treatment strategies targeting amyloid β-protein.. Cold Spring Harb Perspect Med., 2012. [DOI | PubMed]
- D. A. Loeffler. Antibody-Mediated Clearance of Brain Amyloid-β: Mechanisms of Action Effects of Natural and Monoclonal Anti-Aβ Antibodies, and Downstream Effects.. J. Alzheimers Dis Rep, 2023. [DOI | PubMed]
- E. E. Congdon, E. M. Sigurdsson. Tau-targeting therapies for Alzheimer disease.. Nat. Rev. Neurol, 2018. [DOI | PubMed]
- W. H. Zheng, S. Bastianetto, F. Mennicken, W. Ma, S. Kar. Amyloid beta peptide induces tau phosphorylation and loss of cholinergic neurons in rat primary septal cultures.. Neuroscience, 2002. [DOI | PubMed]
- J. E. Swatton, L. A. Sellers, R. L. Faull, A. Holland, S. Iritani, S. Bahn. Increased MAP kinase activity in Alzheimer’s and Down syndrome but not in schizophrenia human brain.. Eur. J. Neurosci, 2004. [DOI | PubMed]
- Y. Zhang, H. Chen, R. Li, K. Sterling, W. Song. Amyloid β-based therapy for Alzheimer’s disease: challenges successes and future.. Signal Transduction and Targeted Therapy, 2023. [DOI | PubMed]
- P. Liu, Y. Wang, Y. Sun, G. Peng. Neuroinflammation as a Potential Therapeutic Target in Alzheimer’s Disease.. Clin Interv Aging, 2022. [DOI | PubMed]
- A. Ardura-Fabregat, E. Boddeke, A. Boza-Serrano, S. Brioschi, S. Castro-Gomez, K. Ceyzériat, C. Dansokho, T. Dierkes, G. Gelders, M. T. Heneka. Targeting Neuroinflammation to Treat Alzheimer’s Disease.. CNS Drugs, 2017. [DOI | PubMed]
- K. Barber, P. Mendonca, K. F. A. Soliman. The Neuroprotective Effects and Therapeutic Potential of the Chalcone Cardamonin for Alzheimer’s Disease.. Brain Sci., 2023. [DOI | PubMed]
- X. Chen, S. Jiang, R. Wang, X. Bao, Y. Li. Neural Stem Cells in the Treatment of Alzheimer’s Disease: Current Status Challenges, and Future Prospects.. J. Alzheimers Dis, 2023. [DOI | PubMed]
- J. Li, A. Haj Ebrahimi, A. B. Ali. Advances in Therapeutics to Alleviate Cognitive Decline and Neuropsychiatric Symptoms of Alzheimer’s Disease.. International journal of molecular sciences, 2024. [DOI | PubMed]
- H. Hampel, J. Hardy, K. Blennow, C. Chen, G. Perry, S. H. Kim, V. L. Villemagne, P. Aisen, M. Vendruscolo, T. Iwatsubo. The Amyloid-β Pathway in Alzheimer’s Disease.. Molecular Psychiatry, 2021. [DOI | PubMed]
- C. Cervellati, G. Valacchi, G. Zuliani. BACE1 role in Alzheimer’s disease and other dementias: from the theory to the practice.. Neural Regeneration Research, 2021. [DOI | PubMed]
- G. Bahn, J.-S. Park, U. J. Yun, Y. J. Lee, Y. Choi, J. S. Park, S. H. Baek, B. Y. Choi, Y. S. Cho, H. K. Kim. NRF2/ARE pathway negatively regulates BACE1 expression and ameliorates cognitive deficits in mouse Alzheimer’s models.. Proc. Natl. Acad. Sci. U. S. A., 2019. [DOI | PubMed]
- C. Martínez-Cué, N. Rueda. Signalling Pathways Implicated in Alzheimer’s Disease Neurodegeneration in Individuals with and without Down Syndrome.. Int. J. Mol. Sci., 2020. [DOI | PubMed]
- A. J. Tibbo, G. S. Tejeda, G. S. Baillie. Understanding PDE4’s function in Alzheimer’s disease; a target for novel therapeutic approaches.. Biochem. Soc. Trans., 2019. [DOI | PubMed]
- L. Ponsaerts, L. Alders, M. Schepers, R. M. W. de Oliveira, J. Prickaerts, T. Vanmierlo, A. Bronckaers. Neuroinflammation in Ischemic Stroke: Inhibition of cAMP-Specific Phosphodiesterases (PDEs) to the Rescue.. Biomedicines, 2021. [DOI | PubMed]
- Y. Wu, Z. Li, Y.-Y. Huang, D. Wu, H.-B. Luo. Novel Phosphodiesterase Inhibitors for Cognitive Improvement in Alzheimer’s Disease.. J. Med. Chem., 2018. [DOI | PubMed]
- R. Ricciarelli, C. Brullo, J. Prickaerts, O. Arancio, C. Villa, C. Rebosio, E. Calcagno, M. Balbi, B. T. J. van Hagen, E. K. Argyrousi. Memory-enhancing effects of GEBR-32a. a new PDE4D inhibitor holding promise for the treatment of Alzheimer’s disease.. Sci. Rep., 2017. [DOI | PubMed]
- J. Kim, J. M. Basak, D. M. Holtzman. The role of apolipoprotein E in Alzheimer’s disease.. Neuron, 2009. [DOI | PubMed]
- Y. Yamazaki, N. Zhao, T. R. Caulfield, C. C. Liu, G. Bu. Apolipoprotein E and Alzheimer disease: pathobiology and targeting strategies.. Nat. Rev. Neurol, 2019. [DOI | PubMed]
- A. R. Gomez, H. R. Byun, S. Wu, A. G. Muhammad, J. Ikbariyeh, J. Chen, A. Muro, L. Li, K. E. Bernstein, R. Ainsworth. Angiotensin Converting Enzyme (ACE) expression in microglia reduces amyloid β deposition and neurodegeneration by increasing SYK signaling and endolysosomal trafficking.. bioRxiv, April 28,, 2024. [DOI]
- J. Sinsky, K. Pichlerova, J. Hanes. Tau Protein Interaction Partners and Their Roles in Alzheimer’s Disease and Other Tauopathies.. Int. J. Mol. Sci., 2021. [DOI | PubMed]
- P. H. Reddy. Amyloid beta-induced glycogen synthase kinase 3β phosphorylated VDAC1 in Alzheimer’s disease: implications for synaptic dysfunction and neuronal damage.. Biochim. Biophys. Acta, 2013. [DOI | PubMed]
- Y. Hou, X. Dan, M. Babbar, Y. Wei, S. G. Hasselbalch, D. L. Croteau, V. A. Bohr. Ageing as a risk factor for neurodegenerative disease.. Nat. Rev. Neurol., 2019. [DOI | PubMed]
- M. R. Bowl, S. J. Dawson. Age-Related Hearing Loss.. Cold Spring Harb. Perspect. Med., 2019. [DOI | PubMed]
- A. L. Pelletier, L. Rojas-Roldan, J. Coffin. Vision Loss in Older Adults.. Am. Fam. Physician, 2016. [PubMed]
- Q. Alle, E. Le Borgne, O. Milhavet, J. M. Lemaitre. Reprogramming: Emerging Strategies to Rejuvenate Aging Cells and Tissues.. Int. J. Mol. Sci., 2021. [DOI | PubMed]
- J. Zhao, J. Huai. Role of primary aging hallmarks in Alzheimeŕs disease.. Theranostics, 2023. [DOI | PubMed]
- D. Chakravarti, K. A. LaBella, R. A. DePinho. Telomeres: history. health, and hallmarks of aging.. Cell, 2021. [DOI | PubMed]
- F. Coppedè, L. Migliore. DNA damage and repair in Alzheimer’s disease.. Curr. Alzheimer Res., 2009. [DOI | PubMed]
- X. Lin, A. Kapoor, Y. Gu, M. J. Chow, J. Peng, K. Zhao, D. Tang. Contributions of DNA Damage to Alzheimer’s Disease.. Int. J. Mol. Sci., 2020. [DOI | PubMed]
- Y. Hou, H. Song, D. L. Croteau, M. Akbari, V. A. Bohr. Genome instability in Alzheimer disease.. Mech Ageing Dev, 2017. [DOI | PubMed]
- B. Schumacher, J. Pothof, J. Vijg, J. H. J. Hoeijmakers. The central role of DNA damage in the ageing process.. Nature, 2021. [DOI | PubMed]
- M. Yousefzadeh, C. Henpita, R. Vyas, C. Soto-Palma, P. Robbins, L. Niedernhofer. DNA damage-how and why we age?.. eLife, 2021. [DOI | PubMed]
- F. Rossiello, U. Herbig, M. P. Longhese, M. Fumagalli, F. d’Adda di Fagagna. Irreparable telomeric DNA damage and persistent DDR signalling as a shared causative mechanism of cellular senescence and ageing.. Curr. Opin Genet Dev, 2014. [DOI | PubMed]
- S. Victorelli, J. F. Passos. Telomeres and Cell Senescence – Size Matters Not.. EBioMedicine, 2017. [DOI | PubMed]
- A. Anitha, I. Thanseem, M. M. Vasu, V. Viswambharan, S. A. Poovathinal. Telomeres in neurological disorders.. Adv. Clin Chem., 2019. [DOI | PubMed]
- D. Saul, R. L. Kosinsky. Epigenetics of Aging and Aging-Associated Diseases.. Int. J. Mol. Sci., 2021. [DOI | PubMed]
- A. C. Tecalco-Cruz, J. O. Ramírez-Jarquín, M. E. Alvarez-Sánchez, J. Zepeda-Cervantes. Epigenetic basis of Alzheimer disease.. World J. Biol. Chem., 2020. [DOI | PubMed]
- X. Wei, L. Zhang, Y. Zeng. DNA methylation in Alzheimer’s disease: In brain and peripheral blood.. Mech Ageing Dev, 2020. [DOI | PubMed]
- R. L. West, J. M. Lee, L. E. Maroun. Hypomethylation of the amyloid precursor protein gene in the brain of an Alzheimer’s disease patient.. J. Mol. Neurosci, 1995. [DOI | PubMed]
- K. Hernández-Ortega, P. Garcia-Esparcia, L. Gil, J. J. Lucas, I. Ferrer. Altered Machinery of Protein Synthesis in Alzheimer’s: From the Nucleolus to the Ribosome.. Brain Pathol, 2016. [DOI | PubMed]
- P. Garcia-Esparcia, G. Sideris-Lampretsas, K. Hernandez-Ortega, O. Grau-Rivera, T. Sklaviadis, E. Gelpi, I. Ferrer. Altered mechanisms of protein synthesis in frontal cortex in Alzheimer disease and a mouse model.. Am. J. Neurodegener Dis, 2017. [PubMed]
- X. Wang, M. L. Michaelis, E. K. Michaelis. Functional genomics of brain aging and Alzheimer’s disease: focus on selective neuronal vulnerability.. Curr. Genomics, 2010. [DOI | PubMed]
- M. Wang, P. Roussos, A. McKenzie, X. Zhou, Y. Kajiwara, K. J. Brennand, G. C. De Luca, J. F. Crary, P. Casaccia, J. D. Buxbaum. Integrative network analysis of nineteen brain regions identifies molecular signatures and networks underlying selective regional vulnerability to Alzheimer’s disease.. Genome Med., 2016. [DOI | PubMed]
- S. Bhatia, R. Rawal, P. Sharma, T. Singh, M. Singh, V. Singh. Mitochondrial Dysfunction in Alzheimer’s Disease: Opportunities for Drug Development.. Curr. Neuropharmacol, 2022. [DOI | PubMed]
- K. Takuma, S. S. Yan, D. M. Stern, K. Yamada. Mitochondrial dysfunction endoplasmic reticulum stress, and apoptosis in Alzheimer’s disease.. J. Pharmacol Sci., 2005. [DOI | PubMed]
- P. Picone, D. Nuzzo, L. Caruana, V. Scafidi, M. Di Carlo. Mitochondrial dysfunction: different routes to Alzheimer’s disease therapy.. Oxid Med. Cell Longev, 2014. [DOI | PubMed]
- I. W. Weidling, R. H. Swerdlow. Mitochondria in Alzheimer’s disease and their potential role in Alzheimer’s proteostasis.. Exp. Neurol., 2020. [DOI | PubMed]
- H. Atamna, W. H. Frey. Mechanisms of mitochondrial dysfunction and energy deficiency in Alzheimer’s disease.. Mitochondrion, 2007. [DOI | PubMed]
- M. P. Singulani, C. P. M. Pereira, A. F. F. Ferreira, P. C. Garcia, G. D. Ferrari, L. C. Alberici, L. R. Britto. Impairment of PGC-1α-mediated mitochondrial biogenesis precedes mitochondrial dysfunction and Alzheimer’s pathology in the 3xTg mouse model of Alzheimer’s disease.. Exp Gerontol, 2020. [DOI | PubMed]
- P. Bubber, V. Haroutunian, G. Fisch, J. P. Blass, G. E. Gibson. Mitochondrial abnormalities in Alzheimer brain: mechanistic implications.. Ann. Neurol., 2005. [DOI | PubMed]
- J. X. Chen, S. D. Yan. Amyloid-beta-induced mitochondrial dysfunction.. J. Alzheimers Dis, 2007. [DOI | PubMed]
- J. M. Perez Ortiz, R. H. Swerdlow. Mitochondrial dysfunction in Alzheimer’s disease: Role in pathogenesis and novel therapeutic opportunities.. Br. J. Pharmacol., 2019. [DOI | PubMed]
- Z. Zhang, X. Yang, Y.-Q. Song, J. Tu. Autophagy in Alzheimer’s disease pathogenesis: Therapeutic potential and future perspectives.. Ageing Research Reviews, 2021. [DOI | PubMed]
- R. A. Nixon, J. Wegiel, A. Kumar, W. H. Yu, C. Peterhoff, A. Cataldo, A. M. Cuervo. Extensive involvement of autophagy in Alzheimer disease: an immuno-electron microscopy study.. Journal of Neuropathology & Experimental Neurology, 2005. [DOI | PubMed]
- B. L. Heckmann, B. J. Teubner, E. Boada-Romero, B. Tummers, C. Guy, P. Fitzgerald, U. Mayer, S. Carding, S. S. Zakharenko, T. Wileman. Noncanonical function of an autophagy protein prevents spontaneous Alzheimer’s disease.. Sci. Adv., 2020. [DOI | PubMed]
- V. Lachance, Q. Wang, E. Sweet, I. Choi, C.-Z. Cai, X.-X. Zhuang, Y. Zhang, J. L. Jiang, R. D. Blitzer, O. Bozdagi-Gunal. Autophagy protein NRBF2 has reduced expression in Alzheimer’s brains and modulates memory and amyloid-beta homeostasis in mice.. Mol. Neurodegener., 2019. [DOI | PubMed]
- F. Pickford, E. Masliah, M. Britschgi, K. Lucin, R. Narasimhan, P. A. Jaeger, S. Small, B. Spencer, E. Rockenstein, B. Levine. The autophagy-related protein beclin 1 shows reduced expression in early Alzheimer disease and regulates amyloid β accumulation in mice.. J. Clin. Invest., 2008. [DOI | PubMed]
- E. Drouin, G. Drouin. The first report of Alzheimer’s disease.. Lancet Neurology, 2017. [DOI]
- R. A. Stelzmann, H. Norman Schnitzlein, F. Reed Murtagh. An English translation of Alzheimer’s 1907 paper. ″Uber eine eigenartige Erkankung der Hirnrinde″.. Clin Anat, 1995. [DOI | PubMed]
- F. Yin. Lipid metabolism and Alzheimer’s disease: clinical evidence. mechanistic link and therapeutic promise.. FEBS journal, 2023. [DOI | PubMed]
- K. Gao, C. Wei, J. Zhu, X. Wang, G. Chen, Y. Luo, D. Zhang, W. Yue, H. Yu. Exploring the Causal Pathway From Telomere Length to Alzheimer’s Disease: An Update Mendelian Randomization Study.. Front. Psychiatry, 2019. [DOI | PubMed]
- M. Herrmann, I. Pusceddu, W. März, W. Herrmann. Telomere biology and age-related diseases.. Clin Chem. Lab Med., 2018. [DOI | PubMed]
- D. A. Forero, Y. González-Giraldo, C. López-Quintero, L. J. Castro-Vega, G. E. Barreto, G. Perry. Meta-analysis of Telomere Length in Alzheimer’s Disease.. J. Gerontol A Biol. Sci. Med. Sci., 2016. [DOI | PubMed]
- E. Eitan, E. R. Hutchison, M. P. Mattson. Telomere shortening in neurological disorders: an abundance of unanswered questions.. Trends Neurosci, 2014. [DOI | PubMed]
- D. Scarabino, E. Broggio, G. Gambina, R. M. Corbo. Leukocyte telomere length in mild cognitive impairment and Alzheimer’s disease patients.. Exp Gerontol, 2017. [DOI | PubMed]
- E. R. Mahoney, L. Dumitrescu, M. Seto, K. N. H. Nudelman, R. F. Buckley, K. A. Gifford, A. J. Saykin, A. J. Jefferson, T. J. Hohman. Telomere length associations with cognition depend on Alzheimer’s disease biomarkers.. Alzheimers Dement (N Y), 2019. [DOI | PubMed]
- K. Fluegge. A model of lipid dysregulation and altered nutrient status in Alzheimer’s disease.. Alzheimer’s Dementia: Transl. Res. Clin. Interventions, 2019. [DOI]
- G. Y. Liu, D. M. Sabatini. mTOR at the nexus of nutrition. growth, ageing and disease.. Nat. Rev. Mol. Cell Biol., 2020. [DOI | PubMed]
- M. A. Shafei, M. Harris, M. E. Conway. Divergent metabolic regulation of autophagy and mTORC1—early events in Alzheimer’s disease?.. Front. Aging Neurosci., 2017. [DOI | PubMed]
- M. Fidaleo, V. Cavallucci, G. Pani. Nutrients. neurogenesis and brain ageing: From disease mechanisms to therapeutic opportunities.. Biochemical pharmacology, 2017. [DOI | PubMed]
- J. B. Jahrling, R.-M. Laberge. Age-related neurodegeneration prevention through mTOR inhibition: potential mechanisms and remaining questions.. Curr. Trends Med. Chem., 2015. [DOI]
- G. Rena, D. G. Hardie, E. R. Pearson. The mechanisms of action of metformin.. Diabetologia, 2017. [DOI | PubMed]
- FDA-Approved Treatments For Alzheimer’s.
- Anti-Amyloid Monoclonal Antibody Drugs Continue to Show Promise Treating Alzheimer Disease.
- M. Davey. Alzheimer’s ‘breakthrough’ stalls: why a much-hyped drug is facing approval delays.
- I. A. Scott. Monoclonal antibodies for treating early Alzheimer disease—a commentary on recent ‘positive’ trials.. Age Ageing, 2024. [DOI | PubMed]
- FDA Grants Accelerated Approval for Alzheimer’s Drug.
- Biogen to Realign Resources for Alzheimer’s Disease Franchise.
- FDA Grants Accelerated Approval for Alzheimer’s Disease Treatment.
- J. R. Sims, J. A. Zimmer, C. D. Evans, M. Lu, P. Ardayfio, J. Sparks, A. M. Wessels, S. Shcherbinin, H. Wang, E. S. Monkul Nery. Donanemab in Early Symptomatic Alzheimer Disease: The TRAILBLAZER-ALZ 2 Randomized Clinical Trial.. JAMA, 2023. [DOI | PubMed]
- LECANEMAB CONFIRMATORY PHASE 3 CLARITY AD STUDY MET PRIMARY ENDPOINT, SHOWING HIGHLY STATISTICALLY SIGNIFICANT REDUCTION OF CLINICAL DECLINE IN LARGE GLOBAL CLINICAL STUDY OF 1,795 PARTICIPANTS WITH EARLY ALZHEIMER’S DISEASE.
- C. H. van Dyck, C. J. Swanson, P. Aisen, R. J. Bateman, C. Chen, M. Gee, M. Kanekiyo, D. Li, L. Reyderman, S. Cohen. Lecanemab in Early Alzheimer’s Disease.. N. Engl. J. Med., 2023. [DOI | PubMed]
- M. A. Mintun, A. C. Lo, C. Duggan Evans, A. M. Wessels, P. A. Ardayfio, S. W. Andersen, S. Shcherbinin, J. Sparks, J. R. Sims, M. Brys. Donanemab in Early Alzheimer’s Disease.. New England Journal of Medicine, 2021. [DOI | PubMed]
- J. M. Long, D. M. Holtzman. Alzheimer Disease: An Update on Pathobiology and Treatment Strategies.. Cell, 2019. [DOI | PubMed]
- FDA approves treatment for adults with Alzheimer’s disease.
- Alzheimer’s: Drugs help manage symptoms.
- Treatment Alzheimer’s disease.
- Medications for Alzheimer’s Disease.
- Alpha Cognition Announces Positive Topline Results from Bioequivalence Study with ALPHA-1062 in Development for Alzheimer’s Disease.
- E. Molina, S. Castillo, A. M. Lacosta, J. Allué, N. Fandos, J. Romero, M. Montañés, L. Sarasa, J. Terencio, M. Boada. Update Phase 2 Study of ABVAC40, an active vaccine anti-AΒ40 in patients with mild cognitive impairment or very mild Alzheimer’s Disease.. Poster Presentation at 14th Clinical Trials on Alzheimer’s Disease (CTAD) Conference Boston, MA, November 9–12,, 2021
- J. Zhou, S. Rawal, T. Yagi, K. R. Wildsmith, E. Takahashi, K. Horie, N. R. Barthélemy, J. Aluri, B. Lalovic, P. Boyd. E2814: an anti-tau therapy engages its CNS target and affects the downstream tangle-specific biomarker MTBR-tau243 in Dominantly Inherited Alzheimer’s Disease.. Alzheimer’s Dementia, 2023. [DOI]
- C. J. Mummery, A. Börjesson-Hanson, D. J. Blackburn, E. G. B. Vijverberg, P. P. De Deyn, S. Ducharme, M. Jonsson, A. Schneider, J. O. Rinne, A. C. Ludolph. Tau-targeting antisense oligonucleotide MAPTRx in mild Alzheimer’s disease: a phase 1b. randomized, placebo-controlled trial.. Nature Medicine, 2023. [DOI]
- D. C. Matthews, X. Mao, K. Dowd, D. Tsakanikas, C. S. Jiang, C. Meuser, R. D. Andrews, A. S. Lukic, J. Lee, N. Hampilos. Riluzole. a glutamate modulator, slows cerebral glucose metabolism decline in patients with Alzheimer’s disease.. Brain, 2021. [DOI | PubMed]
- ANAVEX®2–73 (Blarcamesine) Shows Clinical Benefit in Long-Term 48Week Phase 2 Extension Study in PDD.
- X. Hua, K. Church, W. Walker, P. L’Hostis, G. Viardot, P. Danjou, S. Hendrix, H. J. Moebius. Safety. Tolerability, Pharmacokinetics, and Pharmacodynamics of the Positive Modulator of HGF/MET, Fosgonimeton, in Healthy Volunteers and Subjects with Alzheimer’s Disease: Randomized, Placebo-Controlled, Double-Blind, Phase I Clinical Trial.. J. Alzheimer’s Dis., 2022. [DOI | PubMed]
- B. Dubois, J. López-Arrieta, S. Lipschitz, T. Doskas, L. Spiru, S. Moroz, O. Venger, P. Vermersch, A. Moussy, C. D. Mansfield. Masitinib for mild-to-moderate Alzheimer’s disease: results from a randomized. placebo-controlled, phase 3, clinical trial.. Alzheimer’s Res. Ther., 2023. [DOI | PubMed]
- BioVie Announces Efficacy Data from Phase 3 Trial of NE3107 in Patients with Mild to Moderate Alzheimer’s Disease.
- Improvements in Cognition, Alzheimer Disease Biomarkers Linked to Novel Small Molecule Drug.
- Valiltramiprosate/ALZ-801.
- M. E. Grabowska, A. Huang, Z. Wen, B. Li, W. Q. Wei. Drug repurposing for Alzheimer’s disease from 2012–2022-a 10-year literature review.. Front. Pharmacol, 2023. [DOI | PubMed]
- Advanced Drug Repurposing and Combination Therapy Development.
- A. K. K. Das, D. Sharma, L. Sharma. Drug repurposing strategy for treating Alzheimer’s disease.. Alzheimer’s Dementia, 2021. [DOI]
- J. Bauzon, G. Lee, J. Cummings. Repurposed agents in the Alzheimer’s disease drug development pipeline.. Alzheimer’s Res. Ther., 2020. [DOI | PubMed]
- C. Anderson, M. Bucholc, P. L. McClean, S.-D. Zhang. The Potential of a Stratified Approach to Drug Repurposing in Alzheimer’s Disease.. Biomolecules, 2024. [DOI]
- L. C. Norins. Repurposing Licensed Drugs for Use Against Alzheimer’s Disease.. J. Alzheimers Dis, 2021. [DOI | PubMed]
- Y. Xu, J. Kong, P. Hu. Computational Drug Repurposing for Alzheimer’s Disease Using Risk Genes From GWAS and Single-Cell RNA Sequencing Studies.. Front. Pharmacol., 2021. [DOI | PubMed]
- S. Rodriguez, C. Hug, P. Todorov, N. Moret, S. A. Boswell, K. Evans, G. Zhou, N. T. Johnson, B. T. Hyman, P. K. Sorger. Machine learning identifies candidates for drug repurposing in Alzheimer’s disease.. Nat. Commun., 2021. [DOI | PubMed]
- C. Yan, M. E. Grabowska, A. L. Dickson, B. Li, Z. Wen, D. M. Roden, C. Michael Stein, P. J. Embí, J. F. Peterson, Q. Feng. Leveraging generative AI to prioritize drug repurposing candidates for Alzheimer’s disease with real-world clinical validation.. npj Digital Medicine, 2024. [DOI | PubMed]
- K. Savva, M. Zachariou, M. M. Bourdakou, N. Dietis, G. M. Spyrou. Network-based stage-specific drug repurposing for Alzheimer’s disease.. Computational and Structural Biotechnology Journal, 2022. [DOI | PubMed]
- C. Ballard. A Placebo Controlled, Randomized Double-blind Parallel Group 12-month Trial of Fasudil for the Treatment of Mild Cognitive Impairment Due to Alzheimer’s Disease, Nested within a Multi-Arm Phase 2 Clinical Trial Platform.
- N. Kumar, A. Gahlawat, R. N. Kumar, Y. P. Singh, G. Modi, P. Garg. Drug repurposing for Alzheimer’s disease: in silico and in vitro investigation of FDA-approved drugs as acetylcholinesterase inhibitors.. J. Biomol. Struct. Dyn., 2022. [DOI | PubMed]
- S. Y. Lee, M.-Y. Song, D. Kim, C. Park, D. K. Park, D. G. Kim, J. S. Yoo, Y. H. Kim. A proteotranscriptomic-based computational drug-repositioning method for Alzheimer’s disease.. Front. Pharmacol., 2020. [DOI]
- M. M. Mielke, P. P. Zandi. Hematologic risk factors of vascular disease and their relation to dementia.. Dement Geriatr Cogn Disord, 2006. [DOI | PubMed]
- F. J. Wolters, H. I. Zonneveld, S. Licher, L. G. M. Cremers, M. K. Ikram, P. J. Koudstaal, M. W. Vernooij, M. A. Ikram. Hemoglobin and anemia in relation to dementia risk and accompanying changes on brain MRI.. Neurology, 2019. [DOI | PubMed]
- J. Luo, J. Q. Thomassen, B. G. Nordestgaard, A. Tybjærg-Hansen, R. Frikke-Schmidt. Blood Leukocyte Counts in Alzheimer Disease.. JAMA Network Open, 2022. [DOI | PubMed]
- A. Gambineri, C. Pelusi. Sex hormones, obesity, and type 2 diabetes: is there a link?.. Endocr. Connect., 2019. [DOI | PubMed]
- K. D. Moudgil, S. H. Venkatesha. The Anti-Inflammatory and Immunomodulatory Activities of Natural Products to Control Autoimmune Inflammation.. Int. J. Mol. Sci., 2023. [DOI]
- B. Oliver, E. Jackson, H. Soliman, M. Singh, M. Salnikova. Chapter 17 – Immunostimulators and Immunomodulators in Cancer Treatment. In. Novel Approaches and Strategies for Biologics, Vaccines and Cancer Therapies;, 2015
- C. A. DeMarshall, J. Viviano, S. Emrani, U. Thayasivam, G. A. Godsey, A. Sarkar, B. Belinka, D. J. Libon, R. G. Nagele. Early Detection of Alzheimer’s Disease-Related Pathology Using a Multi-Disease Diagnostic Platform Employing Autoantibodies as Blood-Based Biomarkers.. J. Alzheimers Dis, 2023. [DOI | PubMed]
- Alzheimer’s Disease Neuroimaging Initiative.
- The Global Alzheimer’s Association Interactive Network.
