Exposome involvement in the development of acne vulgaris
Abstract
Acne vulgaris is one of the most prevalent chronic inflammatory skin diseases worldwide, characterized by marked clinical heterogeneity, fluctuating disease course, and strong sensitivity to environmental and lifestyle factors. The exposome, encompassing lifelong environmental, lifestyle, psychosocial, microbial, and intrinsic exposures, offers an integrative framework for re-conceptualizing acne as an environmentally modulated inflammatory disease. This review synthesizes external and internal exposomal drivers of acne, including pollution, radiation, climate and occupational factors, diet, smoking, cosmetics, psychosocial stress, and microbial ecosystems. We highlight the sebaceous gland as a central exposome sensor that integrates metabolic, immune, microbial, and neuroendocrine signals. Genetic susceptibility, epigenetic reprogramming, and non-coding RNA networks are key modifiers translating environmental exposures into persistent inflammatory and metabolic responses within the pilosebaceous unit. Importantly, adopting a health equity and social determinants of health (SDOH) perspective, we emphasize how structural and socioeconomic inequalities shape exposome burden, disease severity, and access to care. We propose that exposome-informed, low-cost, community-level prevention strategies, combined with evidence-based therapies, offer a pragmatic and equitable approach to acne management. Integrating molecular mechanisms with real-world and equity considerations, this framework advances understanding of acne pathophysiology and supports translation into more inclusive clinical practice.
Article type: Review Article
Keywords: acne vulgaris, environmental and lifestyle factors, exposome, health equity and inclusion, non-coding RNAs, sebaceous gland, skin of color, social determinants of health
Affiliations: Department of Dermatology-Venereology, School of Medicine, University of Patras, Patras, Greece; Department of Biochemistry, School of Medicine, University of Patras, Patras, Greece; Department of Dermatology, Venereology and Immunology, Universitaetsklinikum Ruppin-Brandenburg, Brandenburg Medical School Theodor Fontane and Faculty of Health Sciences Brandenburg, Neuruppin, Germany
License: Copyright © 2026 Grafanaki, Bakoli Sgourou, Maniatis and Zouboulis. CC BY 4.0 This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
Article links: DOI: 10.3389/fimmu.2026.1779036 | PMC: PMC13035778
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (2.4 MB)
Introduction
Acne vulgaris is among the most prevalent chronic inflammatory skin diseases worldwide, affecting both adolescents and adults and imposing a substantial psychosocial and quality-of-life burden (ref. 1–ref. 4). Its classical pathogenic model characterized by sebaceous hyperactivity, follicular hyperkeratinization, Cutibacterium acnes (C. acnes) colonization, and inflammation, remains foundational (ref. 5–ref. 7). However, it fails to fully explain the marked clinical heterogeneity, fluctuating disease course, and strong environmental sensitivity observed in patients with acne (ref. 8).
The sebaceous gland is now recognized as a highly dynamic neuro-immuno-endocrine organ, often referred to as the “brain of the skin,” capable of integrating endocrine, immune, microbial, and environmental signals (ref. 9, ref. 10). Sebocytes express pattern-recognition receptors, respond to metabolic and microbial cues, and actively participate in innate immunity via cytokines, lipid mediators, and antimicrobial peptides, positioning the pilosebaceous unit as a central sensor and effector of exposomal stress (ref. 11, ref. 12). Within the pilosebaceous unit, C. acnes functions as a commensal organism whose pathogenicity is not universal but instead depends on strain diversity, host immune context, and environmental stressors. Acne-associated C. acnes phylotypes preferentially activate Toll-like receptor-2 (TLR2), NLRP3 inflammasome signaling, and interleukin-1β (IL-1β) production in keratinocytes and sebocytes, thereby amplifying local inflammation and lesion progression (ref. 13–ref. 15).
The exposome, defined as the totality of environmental exposures from conception onward, provides an integrative framework for understanding acne as an environmentally modulated disease (ref. 16, ref. 17). The exposome encompasses all external and internal exposures encountered across the lifespan and their cumulative impact on health and disease (ref. 18). External environmental exposures (extrinsic exposome) interact with host biology to shape the internal exposome, driving molecular, cellular, and tissue-level alterations that contribute to the onset, progression, and exacerbation of chronic inflammatory disorders such as acne (ref. 19). Environmental pollution represents a key extrinsic exposomal factor. Particulate matter (PM) and chemical pollutants induce oxidative stress, inflammatory signaling, and dysregulated lipogenesis in sebocytes, often synergizing with C. acnes and contributing to acne exacerbation in urban areas (ref. 20–ref. 22). Repetitive exposomal insults may sustain subclinical inflammation and sebaceous gland priming, supporting the concept of acne as a continuously evolving lesion cycle rather than a series of isolated events (ref. 23, ref. 24).
Physical and lifestyle exposome factors further modulate the biology of acne. Ultraviolet radiation (UV) alters sebaceous lipid composition, induces oxidative stress, and modulates inflammatory pathways, challenging the historical view of UV exposure as uniformly beneficial (ref. 25). High-glycemic and lipogenic diets elevate insulin and insulin-like growth factor-1 (IGF-1) activating PI3K/Akt/mTOR while suppressing FOXO1, which promotes sebaceous lipogenesis and inflammation, with alcohol intake and specific fatty acids further amplifying these metabolic pathways (ref. 26–ref. 31). Conversely, bioactive dietary compounds include resveratrol (e.g. grapes, berries), lactoferrin (an iron-binding milk-derived protein), and tea-derived saponins (e.g. green tea, oolong tea) exert sebosuppressive and anti-inflammatory effects (ref. 32–ref. 37).
Psychosocial stress represents a critical yet often underestimated exposome domain, with catecholamines and neuroendocrine mediators influencing C. acnes behavior and amplifying inflammatory responses, while acne itself is strongly correlated with anxiety, depression, and reduced quality of life, reinforcing a bidirectional stress–disease cycle (ref. 4, ref. 18, ref. 19, ref. 38–ref. 40). Finally, the microbial exposome, mediated by distinct C. acnes phylotypes and their capacity to activate inflammasome pathways, interacts dynamically with environmental and lifestyle factors, further contributing to acne development and persistence (ref. 12, ref. 41).
In this review, we integrate these dimensions into an exposome-based model in which environmental, lifestyle, microbial, and intrinsic factors affect sebaceous gland biology and immune–metabolic crosstalk to determine acne development, severity, and persistence. From healthcare equity and inclusive lens, we integrate social determinants of health (SDOH) to support affordable, accessible and preventive acne care through community-level interventions that translate scientific advances into equitable health benefits for acne patients (f1).
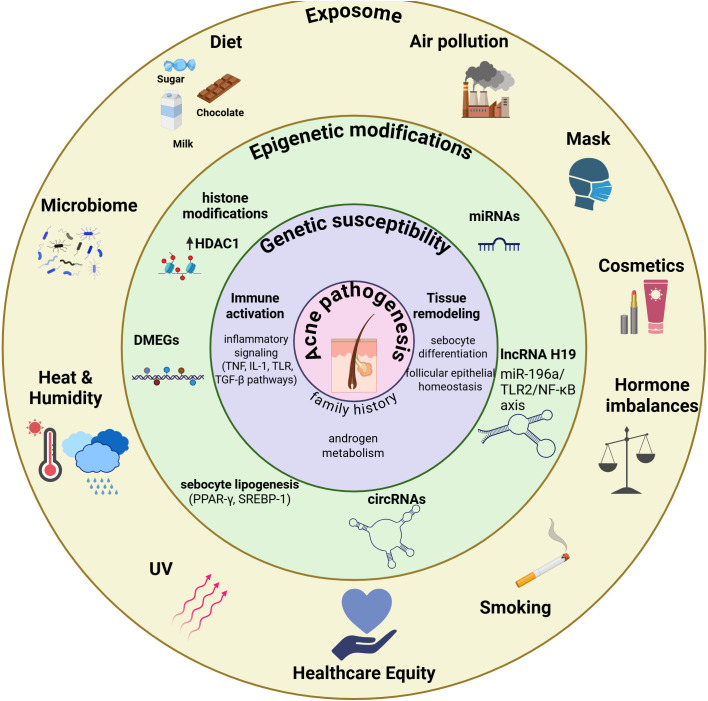
External exposome of acne vulgaris
Air pollution
Air pollution has emerged as a significant environmental contributor to acne vulgaris, acting through complex oxidative and inflammatory mechanisms that disturb skin homeostasis.
Population-based studies have linked exposure to air pollutants with an increased risk and severity of acne. In a Greek study, individuals working near power plants -exposed to higher levels of CO2, CO, SO2, NO2, and PM- had significantly greater odds of developing acne compared to those without such exposure (adjusted odds ratio [aOR] = 3.07). Moreover, severe NO2 exposure was associated with an even higher risk (aOR = 8.24) (ref. 42). Similarly, a large retrospective study demonstrated positive associations between PM2.5, PM10, and NO2 concentrations and outpatient visits for acne vulgaris, while SO2 showed inverse correlations (ref. 43).
PM is classified according to aerodynamic diameter, including PM10 (≤10 μm), PM2.5 (≤2.5 μm) and ultrafine particles (≤0.1 μm), with the smaller particles exhibiting greater skin penetration capacity (ref. 44, ref. 45). Air pollutants such as PM and nitrogen oxides promote acne, primarily through oxidative stress-mediated pathways. In synergy with ultraviolet radiation, pollutants generate reactive oxygen species (ROS), which induce lipid peroxidation of sebum components like squalene, leading to comedogenic byproducts that promote follicular hyperkeratinization, barrier disruption, and inflammatory cytokine release, which favor comedogenesis and inflammatory lesion formation (ref. 46, ref. 47).
Sebocytes are particularly susceptible to pollutant-induced stress. Experimental studies show that PM directly activates sebocytes and synergises with C. acnes to amplify inflammatory responses, lowering the inflammatory activation threshold within the pilosebaceous unit (ref. 48–ref. 50). A central molecular mediator linking pollution exposure to sebaceous and immune dysregulation is the aryl hydrocarbon receptor (AhR) (ref. 22). PM synergizes with C. acnes by amplifying TLR- and AhR-dependent signaling, increasing ROS generation, lowering inflammatory activation thresholds, and enhancing cytokine release (IL-1β, IL-6, IL-8), thus exacerbating the development of inflammatory acne lesions (ref. 49, ref. 51). C. acnes–induced AhR activation modulates inflammatory and barrier-related gene expression, suggesting that pollutant-derived AhR ligands exacerbate acne by amplifying microbe-driven immune signaling. Pollution-related psychosocial and physiological stress may further modulate acne severity. Stress-induced catecholamines influence C. acnes behavior by enhancing bacterial growth, biofilm formation, and virulence, while concurrently amplifying host inflammatory responses through neuroimmune interactions within the pilosebaceous unit (ref. 40, ref. 52, ref. 53).
Clinical studies in highly polluted urban environments, including Shanghai and Mexico City, revealed pollution-associated alterations in sebum composition, reduced vitamin E and squalene levels, and increased lipid peroxidation—biochemical changes strongly implicated in acne pathogenesis (ref. 47, ref. 54). An 8-week study in Chinese acne patients further demonstrated correlations between pollutant exposure, increased sebum secretion, and higher inflammatory and non-inflammatory lesion counts (ref. 54). Pollution-induced oxidative stress likely acts on a pre-existing vulnerable lipid milieu in acne-prone skin, characterized by oxidized squalene and reduced linoleic acid, further amplifying follicular hyperkeratinization and inflammation (ref. 55, ref. 56).
Climatic factors and ultraviolet radiation in acne pathophysiology
Climatic factors, including heat, humidity, and ultraviolet radiation (UVR), significantly influence acne pathophysiology. These environmental stressors contribute to seasonal flares and acne variants such as acne tropicana, acne majorca, or tropical acne (ref. 23, ref. 57–ref. 60).
Heat, humidity, and seasonal variation
Elevated temperature and humidity enhance sweat and sebum secretion, promote follicular occlusion, and favor the proliferation of C. acnes. These factors are particularly relevant in tropical climates, where acne exacerbations are common. Seasonal variation has been consistently documented: in one study of 452 patients, over 50% reported worsening acne during summer, while another cross-sectional study of 171 patients found that nearly half exhibited seasonal symptom variation, with 40% experiencing aggravation in warmer months (ref. 25, ref. 60–ref. 63). Chronic occupational or recreational outdoor exposure further increases acne prevalence, particularly when combined with pollution, traffic-related emissions, or industrial exposure, suggesting synergistic oxidative and inflammatory stressors (ref. 64).
Ultraviolet radiation
UVR exerts complex, wavelength-dependent effects on acne. Both UVA and UVB induce sebaceous gland hyperplasia, stratum corneum thickening, increased sebum secretion, and comedogenesis (ref. 62, ref. 63, ref. 65, ref. 66). UVR acts on keratinocytes, sebocytes, and immune cells to trigger the release of antimicrobial peptides (AMPs) and to activate innate immune pathways, while suppressing adaptive immunity. This immune shift alters the skin microbiome, facilitating C. acnes overcolonization UV-induced microbial damage generates pathogen-associated molecular patterns (PAMPs), interfering with UVR-induced immunosuppression, further amplifying local inflammation and acne flares (ref. 67, ref. 68).
The clinical response to sunlight is bidirectional. Moderate exposure may transiently improve inflammatory lesions, whereas excessive or chronic exposure typically worsens disease severity (ref. 17). Long-wavelength UVA1 (340–400 nm), visible light (VL) (~400 nm), and infrared radiation exhibit anti-inflammatory and antimicrobial properties, reducing C. acnes colonization without increasing sebum production or proinflammatory cytokines (ref. 69). These properties underpin their use in selected phototherapy protocols for acne (ref. 70–ref. 74). The effects of UVB radiation are dose- and exposure-dependent. While low or intermittent UVB exposure may exert immunosuppressive and antimicrobial effects, higher or cumulative doses promote keratinocyte hyperproliferation, increased sebum production, and pro-inflammatory cytokine release, resulting in acne exacerbation (ref. 63, ref. 75).
C. acnes produces endogenous porphyrins that are highly photosensitive. UV and VL excitation of these porphyrins generates cytotoxic ROS, contributing to bacterial killing and forming the basis for photodynamic and blue-light acne therapies (ref. 76, ref. 77). Interestingly, a large retrospective cohort of 19,939 Chinese college students reported a protective association with low-dose exposure to UVB, while identifying UVA as a potential acne risk factor, underscoring population-specific and environmental modifiers (ref. 25, ref. 75). Overall, UVR constitutes a complex physical exposome factor in acne. While short-term exposure may transiently suppress inflammation, cumulative or repeated exposure contributes to oxidative stress, dysregulated lipogenesis, altered lesion distribution, and post-inflammatory sequelae (ref. 25).
UV-induced ROS drive lipid peroxidation and DNA damage in sebocytes and keratinocytes, lowering the activation threshold of innate immune pathways. Redox imbalance activates inflammasome signaling, particularly the NLRP3 inflammasome, a key mediator of acne-associated inflammation (ref. 78, ref. 79). Pharmacological redox modulation can attenuate inflammasome activation. Notably, auranofin, an orally administered anti-rheumatic gold compound that inhibits thioredoxin reductase, reduces oxidative stress and suppresses NLRP3 inflammasome activation under oxidative stress conditions, which highlights its potential relevance in redox-driven inflammatory skin disease such as acne (ref. 80–ref. 82).
However, not all studies confirm a direct association between UVR and acne. A cross-sectional study found no statistically significant relationship between UV exposure and acne severity, nor associations with family history, diet, or age at menarche (ref. 83).
Ultraviolet radiation and skin of color
In individuals with skin of color (SOC), typically corresponding to Fitzpatrick skin phototypes IV-VI, the interaction between UVR, VL, and acne inflammation carries distinct clinical implications. While some SOC patients may experience transient improvement in inflammatory lesions during summer, others develop flares accompanied by post-inflammatory hyperpigmentation (PIH) or post-inflammatory erythema, particularly following excoriated or persistent inflammatory lesions (ref. 84, ref. 85).
VL and UVA1 play a critical role in PIH pathogenesis in SOC. VL activates Opsin-3 photoreceptors in phototypes III–V, triggering Ca2+ influx and MITF (microphthalmia-associated transcription factor) -dependent melanogenesis, which regulates the expression of tyrosinase and other pigment-related genes, leading to redistribution of melanin toward the upper epidermis (ref. 86). VL and UVA may act synergistically, amplifying pigmentary sequelae even in the absence of severe inflammation.
Photosensitizing acne treatments further complicate management in SOC. Isotretinoin frequently necessitates dose reduction or interruption due to photosensitivity (ref. 87, ref. 88). Tetracyclines, particularly doxycycline, are associated with significant phototoxicity: in a prospective study of 106 patients treated over two years, phototoxic reactions occurred in 20% receiving 150 mg/day and 40% receiving 200 mg/day, with a higher incidence in regions of intense solar exposure (ref. 89).
Finally, the widespread use of skin-lightening products in SOC populations poses additional risks. Female SOC users report high rates of adverse effects, including acne, rash, and melasma (ref. 90, ref. 91). Mercury-containing skin-whitening creams, still used globally, act as melanotoxins and can cause severe nodulocystic acne, along with renal and neurological toxicity following chronic exposure (ref. 92). Health education and stricter regulation of hazardous cosmetics are therefore critical components of acne prevention and management in SOC.
Sunbeds, photoprotection, and pigmentary sequelae
Despite well-documented risks, including photocarcinogenesis, photoaging, and PIH, some acne patients use sunbeds as self-treatment. Dermatologists should actively discourage this practice, particularly in patients receiving isotretinoin (ref. 93, ref. 94).
Daily photoprotection is essential for all acne patients, and particularly critical for SOC due to PIH vulnerability (ref. 60, ref. 95–ref. 97). Broad-spectrum sunscreens protect against barrier damage caused by UVR and pollution, thus preserving epidermal barrier integrity, reducing transepidermal water loss (TEWL), and providing anti-inflammatory, antioxidant, sebum-regulating, and antipollution benefits (ref. 5, ref. 98). However, cosmetic acceptability remains a key determinant of adherence; lightweight, non-greasy, matte formulations designed for oily skin improve compliance (ref. 99–ref. 101).
Protection against UVA1 and VL is best achieved with mineral filters such as titanium dioxide and zinc oxide, particularly when combined with iron oxides that are superior at blocking VL. Because UVA1 and VL contribute to PIH, sunscreens containing mineral UV filters and pigmentary iron oxides are strongly recommended (ref. 102, ref. 103), especially for SOC patients who are more prone to pigmentary sequelae (ref. 104–ref. 107). In a cross-sectional study (n = 208), acne clearance was the primary concern for light-skinned women, whereas PIH resolution was the top priority for over 40% of dark-skinned women, compared with only 8% of light-skinned participants (ref. 108).
Tinted sunscreens with iron oxides provide effective protection against UVA1 and VL while offering cosmetic camouflage, a rapid and inexpensive intervention shown to improve quality of life (ref. 109, ref. 110). Rigorous photoprotection is also mandatory following chemical peels and laser procedures for acne (ref. 111).
Occupational and lifestyle factors
Non-inflammatory acne, primarily characterized by comedonal lesions, may result from occupational or environmental exposures that induce follicular occlusion without significant inflammation. Several occupational settings and environmental agents are recognized for their potential to cause acne. In a Greek cohort, acne prevalence was significantly higher among bitumen workers, footwear artisans, and paint industry employees, demonstrating the established entities of coal tar acne and oil acne (ref. 112–ref. 114). These conditions are attributed to chronic exposure to tar derivatives, cutting oils, and related lipophilic compounds that induce mechanical obstruction of sebaceous ducts and promote comedone formation. Males appear more frequently affected, likely due to both higher occupational exposure and a greater baseline predisposition to acne (ref. 113, ref. 114). Although such exposures are less common today, it is suggested that insoluble organic compounds such as coal tar and crude oil interact with keratinous material within follicles, forming obstructive plugs (ref. 113).
Chloracne and related environmental acneiform eruptions
Chloracne is an acne variant caused by environmental pollutants and represents a chronic occupational acneiform dermatosis. It is caused by halogenated aromatic hydrocarbons like polychlorinated dibenzo-p-dioxins and polychlorinated biphenyls (PCBs) and typically manifests weeks to months after exposure (ref. 115). Histopathologically, it features keratinocyte hyperplasia, follicular hyperkeratosis, and cyst formation in the absence of significant inflammation (ref. 116).
A documented case of chloracne following accidental PCB leakage demonstrated clinical resolution with appropriate management, emphasizing the reversible nature of the condition when exposure is eliminated (ref. 117). Similarly, acneiform eruptions associated with polycyclic aromatic hydrocarbons (PAHs) from tobacco smoke suggest overlapping pathogenetic mechanisms between environmental pollutants and occupational acne (ref. 118).
Healthcare workers and PPE-related acne
During the COVID-19 pandemic, acne associated with prolonged face mask use (“maskne”) emerged as a common occupational skin condition among healthcare workers (ref. 119, ref. 120). Acne vulgaris and seborrheic dermatitis were among the most frequently reported conditions linked to personal protective equipment (PPE) use (ref. 121–ref. 125). Several observational studies in healthcare settings demonstrated a clear association between prolonged mask use and either new-onset acne or exacerbation of pre-existing acne, supporting the concept of PPE-related acne as a distinct occupational entity (ref. 120).
The pathogenesis involves the creation of a warm, humid microenvironment under face masks, which enhances sebum production and follicular occlusion (ref. 126). Mask-related factors, including material type, tight fit, duration of daily use, and repeated or prolonged reuse, have been consistently identified as contributors to acne risk among healthcare workers and other mask-wearing populations (ref. 122, ref. 127, ref. 128). In addition, friction and pressure at mask contact sites may induce mechanical acne, particularly over the cheeks, chin, and nasal bridge (ref. 129).
In addition, increased anxiety levels and psychological occupational stress among healthcare professionals during the pandemic may have exacerbated acne symptoms independently of mechanical or occlusive factors (ref. 130–ref. 132).
Smoking and acne vulgaris
The relationship between smoking and acne vulgaris remains controversial. Some investigations have indicated a protective role of smoking, including an online survey reporting a 30% lower acne prevalence among smokers (ref. 133), and others confirming an inverse association (ref. 134, ref. 135). Conversely, several studies identified smoking as an aggravating factor (ref. 136–ref. 138), while others found no significant relationship (ref. 139). A meta-analysis concluded that smoking is a risk factor for adult acne in Asian populations (ref. 140).
Postadolescent acne typically manifests as inflammatory lesions affecting the lower face and neck. However, a distinct comedonal postadolescent acne (CPAA) phenotype characterized by numerous micro- and macrocomedones with minimal inflammation, has been strongly linked to cigarette smoking and is now considered one of the most frequent types of smoking-associated adult acne (ref. 141).
Tobacco and cannabis consumption act as human-derived environmental pollutants capable of disrupting skin homeostasis and potentially acne pathogenesis (ref. 133, ref. 138, ref. 142). However, a definitive causal link between smoking and acne remains unproven. Cigarette smoke contains thousands of toxic compounds, including nicotine, carbon monoxide, tar, formaldehyde, hydrogen cyanide, ammonia, and heavy metals, which compromise skin integrity by increasing TEWL and promoting premature connective tissue damage (ref. 135). Smoking upregulates matrix metalloproteinases (MMP-1 and MMP-3), accelerates collagen and elastin degradation, suppresses collagen synthesis, impairs wound healing, and promotes elastosis, collectively weakening the pilosebaceous unit and facilitating acne development (ref. 143–ref. 145).
Mechanistic studies have showed that smoking induces acne pathogenesis through proinflammatory and oxidative pathways. Cigarette smoke increases interleukin-1α (IL-1α) expression, a key cytokine in comedogenesis, early acne lesion formation and follicular inflammation (ref. 135, ref. 138, ref. 141, ref. 146). Meantime, smoking generates excessive ROS, leading to lipid peroxidation, including oxidized squalene accumulation, which amplifies inflammatory signaling and acne severity (ref. 147). These oxidative mechanisms closely resemble those triggered by air pollutants such as PM2.5 and ozone, reinforcing the concept of a convergent oxidative acne exposome.
Additionally, cigarette smoke alters sebocyte lipid metabolism by downregulating scavenger receptor class B type 1 (SR-B1), a key regulator of sebaceous lipid uptake and homeostasis. Reduced SR-B1 expression results in qualitative sebum alterations, promoting follicular occlusion, microbial dysbiosis, and inflammation (ref. 148, ref. 149).
Cannabis and cannabinoids in acne vulgaris
The global use of medical cannabis products (MCPs), containing tetrahydrocannabinol (THC) and/or cannabidiol (CBD), has expanded although dermatologic evidence remains limited (ref. 150). Cannabinoids exert anti-inflammatory, antipruritic, anti-aging, and antineoplastic effects through interaction with the cutaneous endocannabinoid system (ECS), which regulates keratinocyte activity, immune balance, and sebaceous gland function (ref. 150, ref. 151).
Cannabidiol (CBD), the major non-psychoactive phytocannabinoid, exhibits sebostatic, antiproliferative, and anti-inflammatory activity. In human sebocytes and skin organ culture, CBD inhibited lipogenesis induced by arachidonic acid, linoleic acid, and testosterone, reduced proliferation via transient receptor potential vanilloid-4 (TRPV4) activation, and downregulated nuclear receptor interacting protein-1 (NRIP1) through inhibition of the ERK1/2 MAPK pathway. CBD also upregulated tribbles homolog 3 (TRIB3) via A2A adenosine receptor signaling, suppressing NF-κB–mediated inflammation (ref. 152). Comparative analyses revealed that cannabichromene (CBC) and THC exert sebostatic effects, whereas cannabigerol (CBG) and cannabigerovarin (CBGV) show pro-sebogenic properties (ref. 153). In a split-face clinical trial, a 3% cannabis seed extract cream applied twice daily for 12 weeks significantly reduced sebum production and erythema without adverse effects (ref. 154).
Hemp seed hexane extract (HSHE) has also shown antimicrobial and anti-inflammatory activity against C. acnes, downregulating iNOS, COX-2, IL-1β, and IL-8 expression, and suppressing NF-κB, MAPK, ERK, and JNK activation. HSHE further inhibited 5-lipoxygenase and MMP-9, promoted collagen synthesis, and modulated AMPK and AKT/FoxO1 pathways, further reducing inflammation and lipogenesis (ref. 155).
Despite increasing consumer use of cannabis-based skincare products, often without dermatologic supervision, epidemiologic data remain inconsistent. In a survey of 504 adults, 17.6% reported using over-the-counter cannabis formulations, primarily for acne (28.4%) and psoriasis (26.1%), often without dermatologic guidance (ref. 150). A French survey (>10,000 participants) associated regular cannabis use with higher acne prevalence (OR = 2.88; 95% CI: 1.55–5.37) (ref. 142), whereas subsequent international studies failed to confirm this, likely due to self-reporting bias and legal constraints (ref. 23, ref. 64, ref. 156).
Cosmetics and facial practices as chronic exposomal stressors in acne vulgaris
Cosmetic products constitute chronic, repetitive, low-dose exposomal exposures applied directly to acne-prone skin and have been increasingly implicated in acne exacerbation. Historically described as acne cosmetica, their acneigenic potential is primarily mediated through comedogenicity, defined as the induction of microcomedone formation within the follicular infundibulum, the earliest lesion of acne vulgaris (ref. 157). Comedogenic ingredients such as long-chain fatty acids, fatty alcohols, esters, lanolin derivatives, waxes, and occlusive oils, can promote microcomedone formation by altering follicular keratinization and sebum composition (ref. 158).
From an exposomal perspective, cosmetic-induced occlusion alters the biophysical and biochemical microenvironment of the follicle by reducing oxygen tension and modifying lipid composition, which favors early microcomedone formation (ref. 23, ref. 159, ref. 160). The acneigenic risk is not solely dependent on individual ingredients but also on formulation complexity, frequency of application, duration of contact, and cumulative exposure, reinforcing the exposome concept of dose and chronicity (ref. 23).
Acne vulgaris frequently persists into adulthood, particularly in women, with prevalence estimates of 12% in women and 3% in men over 25 years of age (ref. 161–ref. 164). Notably, 40% of adult patients report acne aggravation following cosmetic use, especially with skin-lightening products, underscoring cosmetics as clinically relevant acne-modifying exposures in adult populations (ref. 165). Conversely, gender of male, warm water for washing face, frequent sun protection, frequent use of moisturizing products and extended use of pad and phone were identified as independent risk factors for acne, while physical exercise and cosmetic usage were identified as independent protective factors. Moreover, participants who utilized a higher number of moisturizing products were associated with an increased risk of developing acne (ref. 166).
The epidermal barrier, composed primarily of ceramides, cholesterol and free fatty acids within the stratum corneum, regulates TEWL and protects against microbial and chemical insults (ref. 167, ref. 168). Skin surface pH is a critical determinant of barrier integrity and microbial homeostasis, and deviations from physiological acidity predispose to inflammatory dermatoses, including acne vulgaris (ref. 19, ref. 169, ref. 170). Modern cleansers for acne-prone skin are therefore formulated with acidic syndets and buffered with organic acids or salts (e.g., lactic or citric acid, sodium lactate or citrate) and frequently incorporate hydroxy acids (salicylic, glycolic, lactic, mandelic), as well as ascorbic, ferulic, and linoleic acids, to support keratinization control and barrier function (ref. 171). In contrast, many traditional soaps and shampoos remain alkaline (pH 9–10 and 6–7, respectively), a profile considered barrier-disruptive and suboptimal for acne-prone skin (ref. 172).
Behavioral facial practices constitute an important exposomal factor, besides the product composition. Layering multiple cosmetic products, prolonged or overnight wear, and inadequate removal increase contact time and increase acne risk (ref. 23, ref. 173). Additionally, mechanical and occlusive practices, including mask use, repetitive face touching, pressure from devices, or friction from accessories, contribute to acne mechanica, characterized by follicular injury, sweat retention, and localized inflammation (ref. 157). Case-control studies confirm increased odds of acne associated with powders, foundations, and cleansers containing comedogenic formulations, independent of hormonal or familial confounders (ref. 173). Conversely, data on modern non-comedogenic and barrier-supportive formulations are not inherently acneigenic, emphasizing the pivotal role of ingredient selection, formulation science, and exposure behavior (ref. 174–ref. 177).
Cosmeceuticals in acne and skin of color
Cosmeceuticals and dermocosmetics contain biologically active ingredients targeting key acne pathways, including sebogenesis, inflammation, follicular hyperkeratinization, oxidative stress, and epidermal barrier dysfunction. Growing evidence supports their use as adjunctive agents in mild acne, acne-prone skin, and maintenance therapy, improving lesion counts, tolerability, and patient-reported outcomes (ref. 178–ref. 181). Active ingredients such as niacinamide, azelaic acid, salicylic acid, antioxidants, and selected plant-derived extracts (Silybum marianum, Myrtus communis, Cirsium eriophorum) demonstrate anti-inflammatory, sebum-modulating, and barrier-supportive effects when appropriately formulated (ref. 180–ref. 182). Mechanistically, these bioactive ingredients work through distinct pathways. Salicylic acid induces desquamation via corneodesmosome disruption and cyclooxygenase inhibition (ref. 183, ref. 184). Concurrently, azelaic acid inhibits C. acnes, reduces keratinocyte hyperproliferation and mitochondrial oxidoreductase activity (ref. 185, ref. 186). Niacinamide complements these actions by enhancing ceramide synthesis, strengthens barrier integrity and inhibits pro-inflammatory cytokine release (ref. 187, ref. 188). Barrier-repair cosmeceuticals containing ceramides, glycerin, and panthenol improve tolerability of topical retinoids and benzoyl peroxide and enhance adherence (ref. 178, ref. 189).
Patients with SOC exhibit increased susceptibility to post-inflammatory hyperpigmentation, keloidal scarring, and irritant dermatitis. In this population, cosmeceuticals may mitigate acne-associated dyspigmentation through inflammation control and gentle pigment modulation; however, inappropriate use of comedogenic, fragranced, or irritating formulations may exacerbate both acne and hyperpigmentation (ref. 174, ref. 190, ref. 191). Aggressive or unregulated depigmenting products, frequently marketed to skin of color populations, may further induce irritant or contact dermatitis, worsening outcomes (ref. 179, ref. 192).
Taken together, cosmetics and cosmeceuticals represent important, yet modifiable components of the acne exposome. When appropriately formulated and used, cosmeceuticals serve as valuable adjuncts in mild acne, maintenance therapy, and supportive care alongside pharmacological treatments. However, they should not replace evidence-based medical therapies in moderate-to-severe disease, as their effects remain modest and misuse may delay effective intervention (ref. 179, ref. 193–ref. 195). Dermatologist-guided selection and patient education remain essential to optimize benefit and minimize harm across diverse skin types (ref. 178, ref. 192, ref. 195, ref. 196).
Nutrition and acne
Acne has often been described as a “Western disease” due to its high prevalence in Western societies, where lifestyle and dietary habits significantly differ from those in non-Western populations (ref. 197). The strong correlation between diet and acne suggests that certain dietary patterns, particularly those common in Western diets, may exacerbate acne by affecting insulin sensitivity and hormone levels (ref. 198). Mechanistic and clinical data consistently implicate Western dietary components – particularly dairy products, high-glycemic-load foods, and specific protein and fat sources – in acne pathophysiology through insulin/IGF-1–mTORC1–FoxO1–driven pathways (ref. 199–ref. 202).
Dairy products
Cow’s milk consumption has been consistently linked to the development of acne, with skim milk showing a stronger association than whole milk (ref. 139). This is attributed to milk-derived bioactive molecules, including insulin-like growth factor-1 (IGF-1) and other hormones that can trigger acne by activating the FoxO1 and mTORC1 pathways (ref. 203–ref. 205). Nutritional and metabolic exposome factors influence insulin/IGF-1–mTORC1 signaling, androgen bioavailability, oxidative stress, and innate immune activation (ref. 206). Collectively, these pathways promote sebocyte lipogenesis, follicular hyperkeratinization, and inflammasome-mediated IL-1β release, leading to microcomedone formation and sustaining inflammatory lesion development (ref. 207). Meta-analyses support a dose-dependent relationship between milk intake and increased acne risk, though the impact of different forms of dairy remains debated (ref. 201, ref. 208–ref. 210).
High-glycemic load diets
Diets high in glycemic load have been strongly associated with acne (ref. 198). High-glycemic foods cause rapid spikes in blood sugar levels, leading to increased insulin production and subsequent activation of sebaceous glands. Clinical studies have shown that individuals who adopt a low-glycemic-load diet experience significant improvements in acne symptoms, along with enhanced insulin sensitivity and weight loss (ref. 211, ref. 212). These clinical effects are supported by mechanistic evidence linking glycemic load to mTORC1 overactivation, androgen bioavailability, and inflammatory signaling within the pilosebaceous unit (ref. 199–ref. 202).
Omega-3 fatty acids and polyunsaturated fatty acids
Omega-3 polyunsaturated fatty acids (PUFAs), particularly eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) derived from fish and marine sources, are consistently associated with reduced acne incidence and severity, in contrast to Western diets rich in saturated fats and omega-6 PUFAs that increase acne risk (ref. 1, ref. 139, ref. 198). Lipid profiling studies demonstrate that acne patients exhibit significantly lower circulating and erythrocyte EPA levels, indicating a systemic pro-inflammatory lipid profile (ref. 213–ref. 215).
Omega-3 fatty acids exert pleiotropic anti-acne effects by attenuating IGF-1 signaling, suppressing mTORC1 activation, and inhibiting TLR-1/2–mediated inflammatory pathways and NLRP3 inflammasome activation within sebocytes and innate immune cells (ref. 216). They also shift eicosanoid production toward less inflammatory and pro-resolving lipid mediators, counterbalancing arachidonic acid–driven inflammation (ref. 217).
Randomized controlled trials demonstrate that oral omega-3 supplementation, alone or combined with gamma-linolenic acid, significantly reduces inflammatory lesion counts and cytokines such as IL-8 (ref. 218–ref. 220). Omega-3 fatty acids modulate the gut–skin axis by increasing the relative abundance of short-chain fatty acid (SCFA)–producing taxa, including Lachnospiraceae and Ruminococcaceae, while reducing pro-inflammatory Proteobacteria (ref. 221, ref. 222). This microbial shift enhances the production of anti-inflammatory metabolites such as butyrate and propionate, which exert systemic immunomodulatory and epigenetic effects on acne pathogenesis (ref. 223, ref. 224). Omega-3 supplementation may also mitigate isotretinoin-associated adverse effects, including xerotic cheilitis and mucocutaneous dryness and hypertriglyceridemia in metabolically predisposed individuals (ref. 225, ref. 226).
Experimental sebocyte models demonstrate that linoleic acid induces distinct transcriptional and lipidomic changes compared with saturated (palmitic acid) and omega-6 arachidonic acid exposure, underscoring fatty acid–specific effects on sebocyte inflammation and differentiation (ref. 227). Genetic and MR analyses support a causal link between circulating PUFA profiles, especially omega-3 levels, and acne susceptibility, providing population-level evidence that lipid metabolism is a contributory factor in acne pathogenesis (ref. 228, ref. 229).
Chocolate and whey protein
The relationship between chocolate consumption and acne remains controversial. While some studies found no significant correlation (ref. 139, ref. 216, ref. 230), others reported an association between chocolate intake and acne exacerbation. In an online survey, chocolate consumption increased acne likelihood by up to 30% (ref. 133), and pure cocoa ingestion or daily dark chocolate intake triggered flare-ups in acne-prone males (ref. 231, ref. 232). Proposed mechanisms include sugar- and leucine-driven activation of insulin/IGF-1–mTORC1 signaling, which may outweigh cocoa’s intrinsic antioxidant properties (ref. 199, ref. 201).
Whey protein supplementation has similarly been implicated in acne due to its high leucine content and potent stimulation of IGF-1 and mTORC1 pathways. Case reports describe acne flares in athletes using whey protein supplements (ref. 233–ref. 235). However, recent randomized controlled trials have not consistently demonstrated significant changes in acne lesion counts with whey protein use over six months (ref. 233, ref. 236, ref. 237). Thus, chocolate and whey protein may exacerbate acne in susceptible individuals, though interindividual variability and dietary context remain critical (ref. 199, ref. 201).
Sebaceous gland as an exposome sensor
Sebocyte intrinsic stress responses triggered by the exposome
Sebocytes are highly responsive stress-sensing cells that translate environmental and internal exposome signals into inflammatory, metabolic, and cell fate decisions (ref. 238, ref. 239). Exposure to physical, chemical, microbial, and metabolic stressors activates conserved danger-sensing pathways – including inflammasome signaling, autophagy, and regulated cell death – placing intrinsic sebocyte stress responses at the center of acne initiation, amplification, and chronicity (ref. 240, ref. 241). Within this context, hypoxia represents a fundamental metabolic stressor of the pilosebaceous unit. The pilosebaceous unit is intrinsically prone to hypoxia due to follicular occlusion, sebum accumulation, microbial overgrowth, and inflammatory edema, with external factors such as occlusive clothing, mechanical pressure, heat, and impaired microcirculation further aggravating hypoxic conditions (ref. 242). Hypoxia induces sebaceous lipogenesis via hypoxia-inducible factor (HIF)–dependent pathways, promoting lipid accumulation and creating an anaerobic microenvironment favouring proliferation of C. acnes (ref. 243, ref. 244).
Inflammasome activation and ion channels
Inflammasome activation represents a key molecular link between exposomal stress and sebocyte-driven inflammation. The NLRP3 inflammasome, activated by oxidative stress, mitochondrial dysfunction, and microbial signals, has emerged as a critical mediator of acne-associated inflammation. 5-Aminolevulinic acid photodynamic therapy (ALA-PDT), a treatment widely used in dermatology for inflammatory and neoplastic skin conditions, induces IL-1β secretion in human SZ95 sebocytes via NLRP3 inflammasome activation (ref. 245), demonstrating that oxidative and phototoxic stress can directly trigger innate immune signaling within sebocytes independent of immune cell infiltration. Given the ubiquity of UV- and photo-oxidative stress, these mechanisms are likely relevant beyond therapeutic settings.
Ion channel–mediated stress sensing has been implicated in sebocyte inflammation. TRPV3, a temperature- and chemical-sensitive cation channel, promotes sebocyte inflammation through transcriptional modulation of TLR2 (ref. 246). As TRPV3 can be activated by thermal stress and chemical irritants, this pathway provides a direct molecular link between environmental exposure and inflammatory signaling within the pilosebaceous unit.
Autophagy, ferroptosis, and sebocyte cell fate
Beyond inflammatory signaling, exposome-induced stress influences sebocyte cell fate through modulation of autophagy and regulated cell death pathways. Autophagy is a key homeostatic mechanism controlling lipid metabolism, organelle turnover, and stress adaptation in sebocytes. Clinical and experimental evidence indicates that autophagy activation improves skin barrier function and alleviates acne symptoms (ref. 247). Mechanistically, autophagy regulates sebaceous lipid production and contributes to the sebosuppressive effects of retinoic acid, one of the most effective systemic acne therapies, suggesting that retinoids may partially restore impaired stress-adaptive autophagic flux in acne-prone sebocytes (ref. 248).
Ferroptosis, an iron-dependent form of lipid peroxidation-driven cell death, has recently been implicated in sebaceous gland targeting strategies. Nanoparticle-based photothermal therapy induces sebaceous gland ferroptosis, leading to reduced sebaceous activity and clinical acne improvement (ref. 249).
Intrinsic exposome
Genetic susceptibility in acne vulgaris
The contribution of genetic factors to acne vulgaris is substantial and well supported by epidemiologic, familial, and genome-wide association studies (GWAS) (T1). Acne is now regarded as a complex polygenic disorder, with multiple susceptibility loci influencing sebaceous gland activity, androgen signaling, inflammation, follicular keratinization, and innate immunity. Comprehensive reviews of the genetic architecture of acne have identified numerous risk variants across diverse populations, confirming the strong heritable basis of the disease (ref. 250–ref. 252).
Table 1: Genetic determinants and related pathways implicated in acne vulgaris.
| Study type | Gene/locus | Biological pathway | Associated clinical phenotype |
|---|---|---|---|
| Twin studies | Multiple loci | Heritability 81-85% environment 15-19% | Acne development |
| GWAS | 1q41, 5q11.2, 11q13.1 | TGF−β signaling | Associated with severe acne |
| 1q24.2, 11p11.2 | Follicular development | Risk to severe acne | |
| GWAS meta−analysis | Wnt, TGF−β, ECM, | Hair follicle development, morphogenesis | Risk to severe acne |
| TNF (rs1800629) | Inflammatory signaling | Reduced risk of mild acne (A allele) | |
| CYP17A1 (rs743572) | Androgen biosynthesis | Reduced risk of severe acne (protective allele) | |
| FST (rs629725) | Follicular development, inflammation (TGF-β) | Modestly increased risk of acne (A allele) | |
| TLR4 (3′-UTR SNPs) | Inflammatory signaling | Acne susceptibility | |
| Mendelian randomization | Proteome → Genetic drug targets | Immune modulation, lipid metabolism, and sebocyte function | Acne treatment |
| Metabolite-linked loci | Systemic serum metabolites and inflammation | Acne risk |
Familial aggregation provides early evidence for genetic predisposition. A meta-analysis showed that individuals with affected first-degree relatives have more than a threefold increased risk of developing acne (OR 3.41; 95% CI 2.31–5.05) (ref. 139). Similarly, in a Chinese Han population, first-degree relatives of acne patients were 4.05 times more likely to develop the disease compared to relatives of healthy controls (ref. 253). In Lithuanian adolescents, family history ranked among the strongest predictors of acne, particularly when both parents were affected (OR 2.6), alongside metabolic and pubertal factors such as BMI >25, menarche in girls, and facial hair growth in boys (ref. 254).
Twin studies further quantify the genetic contribution. In female twin cohorts, additive genetic effects accounted for 81% (95% CI 73–87%) of acne variance, with environmental factors explaining the remaining 19% (ref. 255). Similar estimates were reported in large Australian twin studies, attributing up to 85% of acne susceptibility to genetic influences (ref. 256). These findings position acne among dermatologic diseases with one of the highest heritability estimates.
Large-scale GWAS have refined the genetic architecture of acne, identified multiple reproducible risk loci and reinforcing its polygenic nature. Genome-wide significant loci replicated across populations include 1q41, 5q11.2, and 11q13.1, regions enriched for genes involved in TGF-β signaling, as well as additional loci at 1q24.2 and 11p11.2 (ref. 257–ref. 259). Subsequent meta-analyses expanded the catalog of susceptibility loci, implicating pathways related to sebaceous gland biology, immune regulation, epidermal barrier function, and follicular development (ref. 260). A landmark GWAS meta-analysis demonstrated enrichment of acne risk loci in pathways governing hair follicle morphogenesis, extracellular matrix remodeling, and 249/TGF-β signaling, demonstrating the pilosebaceous unit as a genetically primed target organ (ref. 261). Aberrant activation of TGF-β and Wnt/β-catenin signaling contributes to acne scar formation through dysregulated fibroblast activity and extracellular matrix remodeling, while crosstalk between these pathways and retinoid signaling modulates epidermal differentiation, collagen turnover, and interindividual variability in therapeutic response (ref. 262–ref. 266).
Candidate gene studies and meta-analyses further support these associations. A systematic review encompassing 51 studies across Asian and Caucasian populations identified 60 acne-associated genes/loci with over 100 variants, many located in regulatory or coding regions (ref. 267). Frequently studied genes included TNF, interleukins, and cytochrome P450 family members. While many variants showed modest or nonsignificant pooled effects, specific alleles demonstrated population-level associations: the TNF rs1800629 A allele was associated with reduced risk of mild acne (pOR 0.60; 95% CI 0.33–0.86), the CYP17A1 rs743572 T allele with lower severe acne risk (pOR 0.59; 95% CI 0.40–0.79), and the FST rs629725 A allele with a modestly increased risk (pOR 1.19; 95% CI 1.14–1.23). Novel single-nucleotide polymorphisms (SNPs) in the 3′ untranslated region of TLR4 further implicate innate immune sensing in acne susceptibility (ref. 267).
Beyond risk prediction, Mendelian randomization (MR) approaches have begun to translate genetic findings into causal inference and therapeutic prioritization. Proteome-wide MR analyses identified genetically supported protein targets involved in immune modulation, lipid metabolism, and sebocyte function, highlighting potential novel druggable pathways for acne therapy (ref. 268). In parallel, MR analyses investigating circulating metabolites demonstrated causal relationships between specific serum metabolic profiles and acne risk, supporting a genetically mediated link between systemic metabolism, inflammation, and acne pathophysiology (ref. 269). Together, these data position acne genetics at the intersection of developmental biology, immunometabolism, and precision therapeutics.
Gene expression and pathway analyses provide functional validation of genetic risk. Inflammatory acne lesions exhibit increased expression of matrix metalloproteinases (MMP-1, MMP-3), IL-8, and antimicrobial peptides including human β-defensin-4 and granzyme B (ref. 270). Immunohistochemical studies confirm elevated β-defensin-2 expression in lesional and perilesional skin, with more modest changes in β-defensin-1 (ref. 271), reflecting enhanced innate immune activation and tissue remodeling. Although genetic susceptibility clearly influences acne risk, its relationship with disease severity is less consistent, suggesting that downstream regulatory mechanisms and environmental modifiers shape the clinical phenotype (ref. 1). Genetic risk establishes a primed pilosebaceous unit upon which exposomal and epigenetic modifiers act.
Epigenetic and post-transcriptional regulation in acne vulgaris
Epigenetic mechanisms constitute a critical interface between genetic susceptibility and environmental exposures in acne vulgaris, enabling sustained activation of inflammatory, metabolic, and immune pathways without alterations in DNA sequence (T2). Through histone modifications, non-coding RNAs, and DNA methylation changes, epigenetic regulation shapes sebocyte and keratinocyte responses to microbial, hormonal, metabolic, and environmental stressors, contributing to acne initiation, severity, phenotypic heterogeneity, and chronicity. Importantly, sebocytes function as highly plastic, epigenetically regulated endocrine–immune units rather than passive lipid-producing cells (ref. 239, ref. 240, ref. 272–ref. 274). Epigenetic dysregulation may also explain the mechanistic links between acne vulgaris, acne syndromes, mosaicism, and exposome-driven interindividual variability (ref. 275).
Table 2: Key epigenetic mechanisms implicated in acne vulgaris.
| Epigenetic mechanism | Key molecules | Sample | Molecular alterations | Clinical outcome |
|---|---|---|---|---|
| Histone acetylation | HDAC1 ↑ | Serum, Keratinocytes | Pro−inflammatory transcriptional bias | Acne severity |
| SCFAs (butyrate) by C. acnes | Sebocytes, Keratinocytes | HDAC inhibition | Chronic inflammatory dermatoses/acne vulgaris | |
| miRNAs | miR−146a−5p ↑, miR−143 ↑ | Acne lesions | Suppression of TNF, IL-6, and IL-8 (anti−inflammatory role) | Limits skin inflammation |
| miR-146a | Sebocytes (SZ95) & acne lesions | Regulates TLR1/2- and TLR4-mediated inflammation, proliferation, and lipid synthesis through GNG7 | Acne inflammation and lipid production | |
| miR−21, miR−155 | Acne lesions | Sustained cytokine release (pro−inflammatory role) | Severe acne symptoms | |
| miR−21, miR−150 | Plasma | Post-inflammatory remodeling and fibrosis | Acne severity with scarring | |
| lncRNA | H19 ↑ by C. acnes | Keratinocytes | Activation of miR-196a/TLR2/NF−κB axis | Inflammation within the pilosebaceous unit |
| circRNAs | circ_0105040 | Keratinocytes | Sponges miR−146a | C. acnes biofilm-induced inflammation |
| DNA methylation | DMEGs (PTPRC, CD86, ITGAM, CD8A, IL1B, TNF, TLR4, ITGB2, SPI1, LCP2, and STAT3) | Lymphocytes, Myeloid cells | Enhanced immune activation | Early-stage inflammatory acne lesions |
| ZDHHC20 | Acne lesions | Reduced protein palmitoylation and immune signaling | Reduced acne vulgaris risk |
Histone modifications and chromatin dynamics
Altered histone acetylation plays a central role in acne-associated inflammation and sebaceous gland hyperactivity. Elevated serum levels of histone deacetylase 1 (HDAC1) have been reported in acne patients, indicating a systemic epigenetic milieu favoring transcriptional repression of anti-inflammatory genes and amplification of pro-inflammatory signaling (ref. 276). HDAC-driven chromatin condensation influences cytokine expression, innate immune responses, and keratinocyte differentiation, correlating with disease severity. (f2).
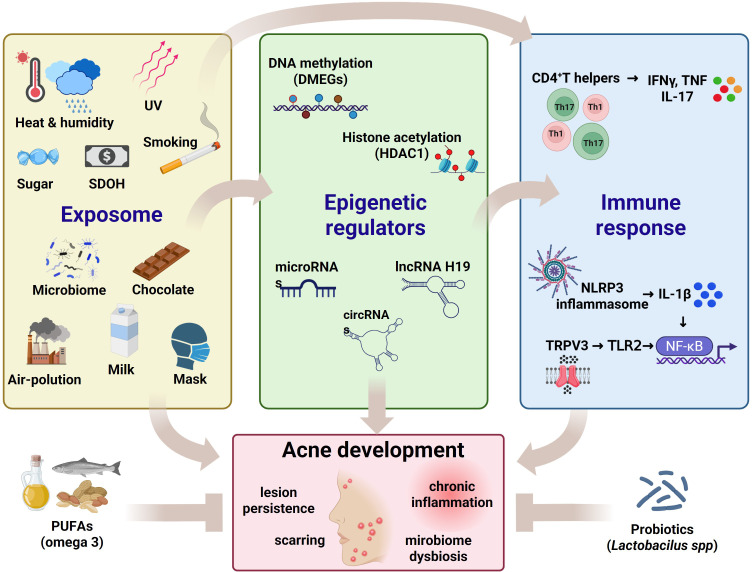
Pharmacological evidence further supports the functional relevance of histone regulation in acne. Minocycline suppresses sebocyte lipogenesis through inhibition of p300 histone acetyltransferase (HAT) activity in human SZ95 sebocytes, reducing acetylation at lipogenic gene promoters and downregulating lipid synthesis (ref. 277). These findings suggest that commonly used acne therapies may exert clinically meaningful epigenetic modulation on sebocyte metabolism, beyond antimicrobial activity.
Epigenetic control of sebocyte lipogenesis represents a core pathogenic axis. Key transcriptional regulators of lipid synthesis, including sterol regulatory element-binding protein-1 (SREBP-1) and peroxisome proliferator-activated receptor-γ (PPAR-γ), are tightly governed by chromatin accessibility and epigenetic co-regulators. In vivo and in vitro studies using synthetic decoy oligodeoxynucleotides targeting SREBP-1 and PPAR-γ demonstrated marked suppression of sebaceous lipogenesis and improvement of acne-like inflammation, confirming that sebocyte hyperactivity is epigenetically programmed rather than purely hormonally driven (ref. 278).
Metabolite-sensing G-protein–coupled receptors further link metabolic cues to epigenetic regulation. GPR119 has emerged as a regulator of sebocyte lipid synthesis, differentiation, and inflammatory signaling (ref. 279). By responding to lipid-derived metabolites, GPR119 likely integrates nutritional and metabolic exposome signals into epigenetically controlled transcriptional networks, reinforcing links between diet, metabolism, and acne.
Emerging evidence suggests that ion channels indirectly shape sebocyte immune and metabolic phenotypes via epigenetic mechanisms. The transient receptor potential melastatin 5 (TRPM5), a calcium-activated cation channel, has been implicated in sebocyte regulation; its antagonist triphenylphosphine oxide increases sebaceous lipogenesis and alters sebocyte immune profiles in a TRPM5-independent manner (ref. 280), implying downstream or off-target epigenetic effects on lipid and inflammatory gene expression. These findings highlight the complexity of sebocyte regulatory networks and support the concept of sebocytes as environmental sensors integrating ionic, microbial, and metabolic signals into epigenetically stabilized functional states.
DNA methylation alterations and immune programming
Acne lesions display distinct DNA methylation profiles compared with normal skin, particularly affecting genes involved in innate and adaptive immunity. Genome-wide methylation analyses identified 31,134 differentially methylated sites across 22 chromosomes and 770 differentially methylated and expressed genes (DMEGs), which are defined as genes exhibiting concurrent changes in DNA methylation status and transcriptional expression. Eleven key DMEGs—PTPRC, CD86, ITGAM, CD8A, IL1B, TNF, TLR4, ITGB2, SPI1, LCP2, and STAT3—are central to immune activation and inflammatory signaling in early acne lesions (ref. 281). Notably, methylation changes are stage-dependent, with the most pronounced alterations observed in early inflammatory acne, implicating DNA methylation as a driver of lesion initiation rather than late persistence.
Systemic epigenetic reprogramming is supported by peripheral blood analyses showing altered immune cell composition (reduced NK cells, increased neutrophils) and methylation changes in loci such as PDGFD, LOC105375130 (CARD11 signaling), and ARHGEF10, genes involved in NF-κB activation, cytoskeletal dynamics, migration, and apoptosis (ref. 282). Population-based studies further demonstrate DNA methylation–mediated genetic risk in young men with severe acne, indicating that epigenetic modifications amplify inherited susceptibility (ref. 283). Importantly, not all methylation changes are pathogenic; methylation at cg18095732 regulating ZDHHC20 expression has been associated with reduced acne risk, highlighting protective epigenetic adaptations (ref. 284).
Epigenetic alterations converge on macrophage- and T-cell–mediated inflammation. Macrophages engulf C. acnes (ref. 285) a and activate the NLRP3 inflammasome, leading to IL-1β release, a key initiating event in acne pathogenesis (ref. 286–ref. 289). Both IL1B and STAT3, prominent DMEGs, participate in this axis, resembling inflammatory signaling observed in atherosclerosis (ref. 290). Notably, C. acnes directly activates the NLRP3 inflammasome in human sebocytes, reinforcing the sebaceous gland as an active immunological organ (ref. 78, ref. 291).
Adaptive immunity further sustains inflammation. Acne lesions exhibit prominent infiltration of CD4+ Th1 and Th17 cells, correlating with lesion severity and chronicity (ref. 159, ref. 292, ref. 293). Epigenetically regulated genes such as CD8A, PTPRC, and ITGB2 underscore adaptive immune recruitment and persistence (ref. 294), supporting contemporary models of acne as an immune-mediated disease characterized by dysregulated innate–adaptive crosstalk (ref. 286, ref. 287). Finally, environmental and dietary exposures may modulate acne-associated epigenetic landscapes. Epigenetic dysregulation is increasingly recognized in inflammatory dermatoses, including rosacea (ref. 19) and hidradenitis suppurativa (HS), where methylation abnormalities affect genes involved in wound repair and keratinocyte proliferation (ref. 295–ref. 297). Given mechanistic overlaps between HS and acne (ref. 198, ref. 298), these observations reinforce the centrality of the epigenome-inflammation axis. Dietary factors particularly milk-derived miRNAs, that survive pasteurization, may further influence acne epigenetics by targeting p53 and DNMT1, potentially attenuating tumor suppressor function and DNA methylation pathways (ref. 299). Persistent milk consumption may therefore represent a nutritional epigenetic trigger exacerbating acne and other Western inflammatory diseases linked to p53/DNMT1 dysregulation.
Non-coding RNAs in acne
Epigenetic control via microRNAs (miRNAs), long non-coding RNAs (lncRNAs), and circular RNAs (circRNAs) plays a crucial role in acne pathophysiology by regulating post-transcriptional gene expression in immune cells, keratinocytes, and sebocytes.
Several miRNAs involved in immune regulation, oxidative stress, and metabolic signaling have been implicated in acne. Both acne vulgaris and hidradenitis suppurativa (HS) share deregulated miRNA profiles primarily governing innate immune pathways. Anti-inflammatory miRNAs such as miR-146a-5p and miR-143 are upregulated upon C. acnes biofilm exposure, attenuating IL-6, IL-8, and TNF production through targeting of TLR2 and NF-κB signaling. Conversely, pro-inflammatory miRNAs, including miR-21 and miR-155, are overexpressed in acne lesions, promoting cytokine release and sustaining inflammation (ref. 300). Additionally, miR-146a connects TLR-mediated inflammatory responses with sebocyte proliferation and indirectly regulates lipid production via GNG7. Following TLR1/2 and TLR4 stimulation, miR-146a is upregulated in sebocytes and in acne lesions, where it suppresses cytokine secretion (IL-8) and chemotaxis, enhances proliferation, and reduces apoptosis. Simultaneous downregulation of GNG7 appears to shift sebocyte activity from proliferation toward increased lipid synthesis (ref. 301).
Oxidative stress–related miRNAs are particularly enriched in severe acne. Altered expression of miRNAs involved in redox balance and inflammatory amplification has been documented, reinforcing the link between oxidative stress and epigenetic dysregulation in acne severity (ref. 302). In line with this, dysregulated expression of FoxO1, mTOR-related miRNAs (miR-21, miR-29b, miR-98) has been associated with metabolic syndrome features in acne patients, suggesting convergence between metabolic signaling and epigenetic control (ref. 303).
Comparative profiling of acne patients with and without scarring identified 88 differentially expressed miRNAs, with miR-223, miR-21, and miR-150 emerging as key regulators. Plasma levels of miR-21 and miR-150 were significantly elevated in patients with scarring, implicating these miRNAs in post-inflammatory remodeling and fibrosis (ref. 304). More recently, miR-191-5p has been associated with acne severity and disease progression, highlighting its potential role as a prognostic biomarker (ref. 305). In addition, serum exosomal miRNA profiling revealed a marked downregulation of hsa-miR-124-3p in severe acne, implicating impaired immune resolution and T-cell regulation in advanced disease stages (ref. 306).
LncRNAs fine-tune inflammatory signaling within the pilosebaceous unit. The lncRNA H19 is induced in keratinocytes following C. acnes exposure and facilitates inflammation via the miR-196a/TLR2/NF-κB axis reinforcing innate immune activation within the pilosebaceous unit (ref. 307). Dysregulated lncRNA networks have been identified in familial acne inversa associated with NCSTN mutations, suggesting shared epigenetic regulatory principles across acneiform disorders (ref. 308).
CircRNAs add an additional regulatory layer by acting as miRNA sponges. Multiple circRNAs are downregulated in acne lesions, including circRNA_0084927, circRNA_0001073, circRNA_0005941, circRNA_0086376, and circRNA_001816, suggesting potential loss of post-transcriptional regulatory control (ref. 309). Functional studies have demonstrated that hsa_circ_0105040 promotes C. acnes biofilm–induced inflammation by sequestering miR-146a, thereby amplifying inflammatory signaling in human keratinocytes (ref. 310). These findings position circRNAs as critical modulators linking microbial stimuli to sustained inflammatory responses in acne (ref. 311).
Microbiome-epigenome crosstalk in acne vulgaris
The microbial exposome, consisting of skin – gut microbiota and their bioactive products, acts as a central mediator linking external and internal exposures to host epigenetic plasticity. Rather than reflecting simple microbial overgrowth, acne arises from qualitative shifts in microbial communities, strain-level functional activity and their metabolites, which reprogram gene expression in sebocytes, keratinocytes, and immune cells through epigenetic mechanisms (T3).
Table 3: Skin and gut microbiome alterations in acne vulgaris.
| Microbiome site | Phylum/Species | Change in acne | Functional implication | Clinical correlation |
|---|---|---|---|---|
| Skin(Acne lesions) | C. acnes/A1 | Increased | Chronic inflammatory lesions and persistence | Chronic acne |
| Staphylococci | Increased | – | Acne severity | |
| S. epidermidis | Increased or decreased | Protective or competitive effects against C. acnes | Severity−dependent | |
| Malassezia spp. | Increased | Pro−inflammatory interaction | Inflammatory severity of acne | |
| Firmicutes ↑/Proteobacteria ↓ | Increased ratio | Barrier disruption | Acne severity | |
| Gut | Bacteroidetes ↑/Firmicutes ↓ | Increased ratio | Metabolic inflammation | Western−diet acne |
| Probiotics (Lactobacillus spp) | Decreased | Increased systemic inflammation and gut skin microbiota modulation | Acne severity |
Skin microbiome-epigenetic interactions
C. acnes, long considered the primary pathogen in acne, comprises multiple phylogenetic strains with distinct virulence and immunomodulatory profiles. Multilocus sequence typing studies revealed substantial regional and interpersonal heterogeneity, demonstrating dynamic interactions between Staphylococcus epidermidis (S. epidermidis) and yeasts such as Malassezia species within a complex follicular ecosystem (ref. 312–ref. 314). Importantly, C. acnes is an abundant commensal in both healthy and acne-prone skin, and acne development is linked to strain-level dysbiosis rather than bacterial overgrowth (ref. 315).
Phylotype IA1 is more prevalent in acne lesions, with SLST molecular typing confirming phylotype A1 as significantly more common in acne patients than in controls (ref. 316). However, its abundance does not consistently correlate with disease severity, underscoring the importance of microbial functional activity and host epigenetic responsiveness rather than microbial load alone (ref. 317, ref. 318). Biofilm formation within pilosebaceous follicles further stabilizes inflammatory niches and contributes to lesion persistence, especially during adolescence (ref. 319) (f3).
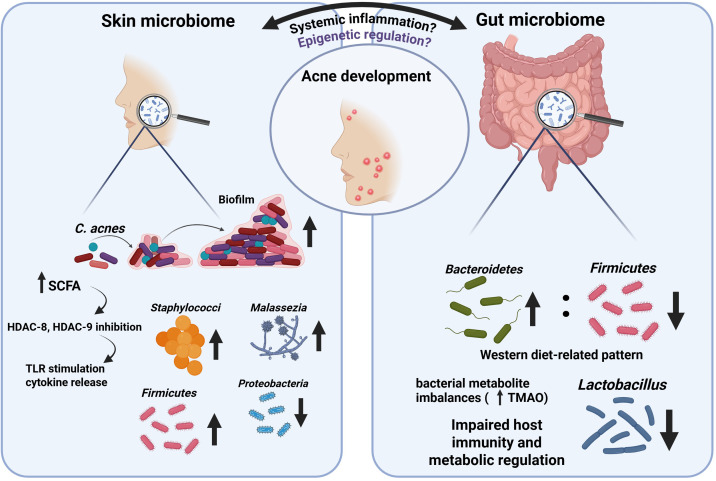
In this context, acne-associated C. acnes strains contribute to pathogenesis through the secretion of extracellular vesicles (EVs) which are nanoscale membranous structures that act as potent mediators of host–microbe communication. C. acnes–derived EVs induce acne-like phenotypes more rapidly and robustly than whole bacterial extracts (ref. 320). EVs activate myeloid cells and their capacity to engage both epithelial and immune compartments. Notably, EV composition varies across C. acnes phylotypes, conferring phylotype-specific immunomodulatory potential, with acne-associated strains producing EVs enriched in pro-inflammatory proteins and lipids that induce IL-1β, IL-6, TNF-α, CXCL1/5, and IL-17 (ref. 321, ref. 322). These findings reinforce the concept that acne-related dysbiosis reflects qualitative alterations in microbial secretomes, with downstream epigenetic consequences, rather than simple shifts in bacterial abundance.
Microbial metabolites provide a direct mechanistic bridge between the microbiome and the epigenome. Short-chain fatty acids (SCFAs), such as butyrate produced by C. acnes under anaerobic conditions, act as endogenous HDAC inhibitors and directly modulate keratinocyte and sebocyte transcriptional programs (ref. 323). Experimental inhibition of HDAC8 and HDAC9 enhances Toll-like receptor–mediated cytokine release in keratinocytes and sebocytes supporting the concept that microbial metabolites fine-tune cutaneous immune responses via epigenetic mechanisms (ref. 324, ref. 325). Disruption of microbiome–epigenome crosstalk predisposes to chronic inflammatory dermatoses, including acne vulgaris (ref. 326).
Acne skin shows increased Firmicutes and reduced Proteobacteria relative to healthy controls, with marked genus-level instability. A metagenomic study that identified 7,303 operational taxonomic units (OTUs), 3,833 were unique to acne and 1,011 to healthy controls. Acne-associated taxa include Lachnospiraceae, Clostridiales, Moraxellaceae, Prevotella, and Lactococcus garvieae, whereas Achromobacter, Stenotrophomonas, Porphyromonas, Prevotella, and Pseudomonas predominated in controls (ref. 327). Longitudinal studies during puberty indicate that these microbial shifts evolves dynamically alongside hormonal and sebaceous changes (ref. 328).
Microbial interactions further influence epigenetically regulated inflammation. S. epidermidis may exert protective or competitive effects against C. acnes, which belongs to phylum Actinobacteria, a dominant component of the healthy skin microbiome, while Malassezia species correlate positively with inflammatory severity (ref. 329, ref. 330). Clinically, reduced antibiotic efficacy in resistant C. acnes strains, indicates that therapeutic benefit derives from microbiome modulation rather than just antimicrobial action (ref. 331–ref. 333). In a clinical trial of 26 acne patients, both topical erythromycin and a dermocosmetic formulation reduced lesions over 28 days, yet their microbiological effects differed. Erythromycin reduced Actinobacteria, whereas the dermocosmetic decreased both Actinobacteria and Staphylococci, depicting the relevance of microbiome-preserving strategies (ref. 334).
Gut microbiome, epigenetic programming, and the gut–skin axis
Systemic microbial dysbiosis further amplifies acne-associated inflammation through the gut–skin axis. Acne patients exhibit higher gut microbial diversity, reduced Actinobacteria, and increased Proteobacteria (ref. 327, ref. 335). At the phylum level, decreased Firmicutes and increased Bacteroidetes result in an elevated Bacteroidetes/Firmicutes ratio, a microbial signature linked to Western dietary patterns and low-grade systemic inflammation (ref. 336). Within 38 differential taxa, Bacteroidetes, Bacteroidia, and Bacteroidales are enriched in acne. Depletion of beneficial taxa such as Clostridia, Clostridiales, Lachnospiraceae, and Ruminococcaceae, key producers of anti-inflammatory SCFAs, may reduce epigenetic repression of pro-inflammatory pathways in peripheral immune cells (ref. 337).
Recent Mendelian randomization studies provide causal evidence linking specific gut microbial taxa to acne susceptibility, supporting a contributory role of gut dysbiosis beyond confounding lifestyle or dietary factors (ref. 338, ref. 339). Additionally, altered microbial-derived metabolites further implicate systemic metabolic–epigenetic crosstalk. Elevated circulating trimethylamine N-oxide (TMAO) levels in acne patients suggest enhanced pro-inflammatory signaling and immune priming via gut microbiota–host metabolic interactions (ref. 340).
Comparative studies also suggest disease-specific gut–skin microbial signatures modulated by demographic and clinical factors. In a study of 8 acne and 19 rosacea patients with matched controls, cutaneous microbial composition was profiled using 16S rRNA gene sequencing of skin samples. The analysis revealed that Serratia marcescens and C. acnes were more abundant in rosacea lesions compared to acne. Interestingly, C. acnes was more prevalent in controls than in patient groups for both conditions, suggesting a potential protective commensal role (ref. 341). The C. acnes profile in papulopustular rosacea resembles that of acne more closely than non-inflammatory rosacea, indicating overlapping inflammatory microbiome–epigenetic circuits across these dermatoses (ref. 19).
Emerging research suggests that gut dysbiosis, or microbial imbalance, may influence acne through systemic inflammation. Probiotics, particularly those containing Lactobacillus strains, have shown promise in reducing acne severity by modulating gut and skin microbiota and lowering systemic inflammation (ref. 342). Dietary patterns are a key determinant of microbial composition. High-glycemic index and low-fiber Western diets deplete SCFA-producing taxa and enrich pro-inflammatory microbial profiles, promoting systemic inflammation and epigenetic immune reprogramming affecting acne. In this context, probiotic and prebiotic interventions are best considered as nutrition-linked biological modulation that restores the gut-skin axis, rather than as isolated biological therapeutic methods (ref. 312, ref. 343).
Microbiome-modulating therapies within the acne exposome
These data position acne as a disease of microbiome-conditioned epigenetic dysregulation, with direct implications for therapy. Probiotic-based interventions in acne vulgaris function as exposure-modifying strategies that modulate host–microbe interactions across dietary, pharmaceutical, and cosmetic domains (ref. 344, ref. 345). Oral probiotics, prebiotics, symbiotics, and postbiotics, particularly Lactobacillus and Bifidobacterium strains, can modestly but significantly reduce acne severity (ref. 346–ref. 349). They primarily act through the dietary and intrinsic exposome, modulating the gut–skin axis by restoring microbial diversity, attenuating systemic inflammation, improving epithelial barrier function, and influencing metabolic and immune signaling pathways implicated in acne pathophysiology, including IGF-1, mTORC1, and Th17 polarization (ref. 221, ref. 346, ref. 350, ref. 351). Randomized controlled trials further support strain-specific efficacy, with L. plantarum and L. paraplantarum demonstrating anti-inflammatory, antibacterial activity against C. acnes and improvements in clinical acne scores (ref. 347, ref. 352–ref. 357). Additionally, postbiotics and probiotic-derived metabolites as biologically active components exert anti-inflammatory and antibiofilm effects without the risks associated with live organisms (ref. 358–ref. 360). Novel strategies targeting C. acnes virulence, such as antibiofilm compounds and immune-enhancing approaches (e.g., anti-CAMP1 IgG), suggest a shift toward precision microbiome interventions rather than broad-spectrum antimicrobial suppression (ref. 361, ref. 362).
In contrast, topical probiotics and postbiotic formulations operate within the cutaneous and cosmetic exposome, directly shaping the local microbial ecosystem, suppressing C. acnes while preserving commensal balance (ref. 363–ref. 365).
Importantly, microbiome-directed strategies may also mitigate iatrogenic exposomal stressors, particularly antibiotic-induced dysbiosis and antimicrobial resistance, which are increasingly recognized as long-term modifiers of acne trajectory (ref. 366). Nevertheless, current clinical guidelines acknowledge probiotics primarily as adjunctive therapies, citing heterogeneity in formulations, dosing, and study design as barriers to formal recommendation (ref. 5, ref. 96).
Hormonal and endocrine exposome in acne vulgaris
Cutaneous steroidogenesis as an exposome-responsive system
Human skin is an active site of steroidogenesis, expressing the enzymatic machinery required for the synthesis and metabolism of glucocorticoids, androgens, estrogens, and steroid precursors (ref. 367). Sebaceous glands display robust expression of steroidogenic enzymes involved in androgen activation and inactivation, enabling local modulation of the hormonal microenvironment independently of circulating hormone levels (ref. 368).
This intracrine steroidogenic activity enables sebocytes to rapidly adapt to internal and external stressors, such as inflammation, metabolic changes, and environmental challenges (ref. 369). However, dysregulation of local steroidogenesis may amplify sebaceous lipogenesis and inflammatory signaling, contributing to acne development (ref. 367). Environmental stressors and lifestyle-related exposome factors -such as chronic stress, sleep disruption, and metabolic imbalance- may influence cutaneous steroidogenic pathways, indirectly modulating acne severity through endocrine mechanisms (ref. 370, ref. 371).
IGF-1 and metabolic hormones linking diet to sebocyte activation
Insulin-like growth factor 1 (IGF-1) represents a key metabolic hormone bridging dietary exposome factors and sebaceous gland activation (ref. 372). IGF-1 stimulates sebocyte proliferation, lipogenesis, and inflammation through activation of PI3K/Akt/FoxO1 and JAK2/STAT3 signaling pathways, pathways that are critically implicated in acne pathophysiology (ref. 30). Elevated IGF-1 signaling, commonly associated with high-glycemic diets, insulin resistance, and Western dietary patterns, promotes sebaceous gland hyperactivity and creates a pro-inflammatory microenvironment within the pilosebaceous unit (ref. 199, ref. 373).
Experimental inhibition of IGF-1–induced signaling attenuates lipid synthesis and inflammation in human sebocytes, supporting diet and metabolism as modifiable exposomal drivers of acne (ref. 30).
Retinoid signaling and endocrine modulation of sebocyte function
Retinoids exert their biological effects through retinoic acid receptors (RARs) and retinoid X receptors (RXRs), which function as ligand-activated transcription factors regulating sebocyte differentiation, lipid synthesis, and inflammatory responses (ref. 374). Isotretinoin acts indirectly on sebocytes, affecting gene expression, cell differentiation, and metabolic pathways rather than simply inducing sebocyte apoptosis (ref. 375). These indirect effects suggest that isotretinoin may modulate, restore dysregulated endocrine and metabolic signaling within the sebaceous gland, counteracting exposome-driven hormonal overstimulation (ref. 376, ref. 377).
Acne, obesity, and insulin resistance
Acne vulgaris and metabolic dysregulation seem to be closely associated. In adult males with acne, higher plasma glucose and insulin levels have been observed following oral glucose testing, a correlation not consistently seen in females, suggesting sex-specific metabolic patterns (ref. 378, ref. 379). Although body mass index (BMI), waist circumference, and visceral fat area tend to be higher in male acne patients, these differences do not always reach statistical significance (ref. 380).
Other investigations on acne patients have documented significantly elevated BMI, body fat percentage, and fat mass among acne patients, with obesity being prevalent. Notably, moderate-to-severe acne is associated with more pronounced metabolic alterations than mild disease, including hyperinsulinemia and metabolic syndrome features (ref. 379, ref. 381, ref. 382). Insulin resistance appears more prevalent among acne patients and may represent a predisposing metabolic factor contributing to disease development (ref. 383).
In pediatric and adolescent populations, early-onset prepubertal acne (ages 7–12) is significantly associated with higher BMI and increased obesity rates, particularly among females, with prevalence rising with age (ref. 384, ref. 385). Hence, metabolic dysregulation and insulin resistance may precede or accompany acne-onset early in life, potentially influencing disease persistence and severity.
Metabolic syndrome and acne severity
The presence of acne scars, often considered a surrogate marker of severe or chronic inflammatory disease, is disproportionately higher among patients with metabolic syndrome. This observation supports a bidirectional relationship between metabolic dysfunction and acne severity, where systemic insulin resistance and oxidative stress may exacerbate inflammatory acne, while chronic acne may reflect underlying metabolic imbalance (ref. 386). Furthermore, metabolic assessment and longitudinal follow-up in patients with moderate-to-severe acne may be useful for early detection and management of cardiometabolic comorbidities (ref. 380, ref. 387).
Multiple clinical and observational studies demonstrate higher BMI, central obesity, dyslipidemia, and impaired glucose metabolism among acne patients, with metabolic abnormalities being more pronounced in moderate-to-severe disease (ref. 387–ref. 390). Case series and cross-sectional studies further suggest that acne may represent an early cutaneous marker of systemic metabolic dysfunction, especially in adolescents and young adults (ref. 387, ref. 388).
At the molecular level, insulin resistance leads to compensatory hyperinsulinemia and enhanced IGF-1 signaling, resulting in mTORC1 overactivation. This promotes sebocyte proliferation, lipogenesis, and androgen synthesis while suppressing FoxO1-mediated transcriptional control, collectively driving follicular hyperkeratinization and inflammation characteristic of acne (ref. 391, ref. 392).
Social determinants, digital environments, and gender diversity in the acne exposome
The exposome of acne vulgaris extends beyond biological and environmental triggers and includes the social determinants of health (SDOH), the structural conditions shaping exposure, vulnerability, healthcare access, and outcomes across the lifespan. As the “causes of the causes,” SDOH such as socioeconomic position, education, neighborhood environment, healthcare infrastructure, and structural discrimination influence acne onset, severity, treatment trajectories, and psychosocial burden through upstream mechanisms rather than individual behaviors alone (ref. 393–ref. 395).
Barriers to dermatologic care such as cost, transportation limitations, rural residence, and specialist scarcity, delay diagnosis and treatment escalation, increasing the risk of chronic inflammation, scarring, and psychosocial burden (ref. 19, ref. 396, ref. 397). Inequitable access to effective therapies underscores persistent gaps in pharmacoequity, particularly for prescription topical agents, systemic antibiotics, and isotretinoin (ref. 398).
Limited access to professional care frequently results in reliance on over the counter (OTC) acne products and informal treatment strategies. Although OTC regimens may provide partial symptom control, prolonged unsupervised use, inappropriate combinations, and variable formulation quality can contribute to irritant dermatitis, suboptimal disease control, and treatment fatigue (ref. 106, ref. 399). Socioeconomic and regional disparities strongly shape OTC utilization, particularly in settings where prescription access is delayed or constrained, such as parts of Latin America and Asia (ref. 106, ref. 400). Within an exposome framework, self-directed acne care represents a structural consequence of healthcare inequities rather than a purely individual choice.
Digital and social media exposures
Social media platforms represent a pervasive informational and psychosocial layer of the acne exposome. Acne-related content on TikTok and other platforms shows high engagement but inconsistent medical accuracy, frequently promoting anecdotal or potentially harmful practices (ref. 400–ref. 403). Adolescents and adults with acne commonly report social media as a primary source of treatment information, influencing product choice, expectations, and adherence outside medical supervision (ref. 404–ref. 407).
Beyond information exposure, social media intensifies appearance-based comparison and stigma, influencing self-presentation, photo-sharing, and dating behaviors, particularly among women and younger individuals (ref. 408, ref. 409). Increased exposure to acne-related content is associated with higher appearance anxiety, psychological distress, and social withdrawal, positioning social media as a psychosocial stress amplifier (ref. 410–ref. 412).
Digital health tools, including teledermatology and online self-management platforms, may improve access and continuity of acne care. However, uneven digital literacy, broadband access, language availability, and device ownership risk reinforcing existing disparities and preferentially benefiting already advantaged populations (ref. 413–ref. 415). The digital divide thus constitutes a structural exposomal determinant of acne outcomes (ref. 416). Equity-oriented deployment is essential for digital acne interventions to avoid widening gaps (ref. 417).
Gender diversity, hormonal modulation, and minority stress
Sexual and gender minority populations experience distinct acne-related exposures at the intersection of biological, hormonal, psychosocial, and structural factors. LGBTQ+ individuals face higher rates of healthcare discrimination, delayed care, and mental health comorbidities, potentially exacerbating acne burden and limiting timely treatment (ref. 418, ref. 419).
In transgender and gender-diverse individuals, acne commonly emerges or worsens during gender-affirming hormone therapy (GAHT), particularly testosterone-based masculinizing regimens (ref. 420–ref. 422). A large retrospective matched cohort study demonstrated higher acne incidence and severity in transmasculine individuals initiating testosterone compared with cisgender controls, with the greatest risk during the first year and persistence over time. Increased rates of moderate-to-severe acne requiring systemic therapy were observed. Notably, transfeminine individuals receiving estradiol also showed higher acne incidence than matched cisgender men, indicating risk beyond androgen-dominant regimens (ref. 423).
Qualitative and mixed-methods studies highlight acne-related stigma, body image distress, and barriers to affirming dermatologic care in transgender and gender-diverse populations. Acne may interfere with gender expression and carry heterogeneous meanings during GAHT, positioning it as both a biological and psychosocial exposomal outcome within a minority stress framework (ref. 424).
Acne in this setting reflects interactions between endocrine modulation, sebaceous gland responsiveness, psychosocial stress, and structural barriers to care. Expert consensus supports anticipatory counseling before GAHT, routine assessment of acne severity and psychosocial burden, and integration of dermatologic care into multidisciplinary gender-affirming pathways (ref. 422, ref. 425, ref. 426).
Psychosocial burden as an exposomal outcome
Acne vulgaris is associated with substantial psychosocial morbidity, including anxiety, depression, low self-esteem, body dysmorphic symptoms, and suicidal ideation (ref. 427–ref. 432). Large-scale and longitudinal studies indicate that acne-related mental health comorbidity is globally prevalent and increasing over time (ref. 431). Psychological distress correlates only modestly with objective disease severity; patient-reported outcomes highlight the central role of perceived visibility, stigma, and social evaluation (ref. 428, ref. 432).
SDOH critically modifies these relationships. Socioeconomic disadvantage, delayed access to care, and structural marginalization increase the likelihood of prolonged disease duration and cumulative psychosocial harm (ref. 430, ref. 431, ref. 433). Racial and ethnic differences further demonstrate socially patterned acne burden, with greater concern regarding post-inflammatory hyperpigmentation and psychosocial impact independent of lesion severity (ref. 108).
Acne treatment and mental health interact bidirectionally. Clinical improvement generally enhances psychosocial well-being (ref. 432, ref. 434, ref. 435), whereas concerns regarding neuropsychiatric adverse effects, particularly with isotretinoin, need careful patient selection, monitoring, and contextualized risk communication, supported by recent EudraVigilance analyses (ref. 436). Resilience, social support, and adaptive coping strategies mitigate adverse outcomes, and adjunctive interventions such as cosmetic camouflage improve emotional well-being without altering disease biology (ref. 110, ref. 178, ref. 437).
Conclusions and future perspectives: towards an equity-oriented acne exposome framework
Acne vulgaris is a chronic, inflammatory disease shaped by cumulative interactions between external exposures, microbial ecosystems, and intrinsic interpatient molecular susceptibility. The exposome framework provides a coherent model to explain disease heterogeneity and variable treatment response, while shifting attention from isolated risk factors to cumulative and context-dependent determinants. Within this framework, the sebaceous gland functions as a central biological sensor of exposomal stress. Recurrent environmental and lifestyle-related exposures induce metabolic, inflammatory, and epigenetic reprogramming that contributes to disease persistence and differential responsiveness to therapy. These mechanisms offer a biological rationale for population-level disparities in acne outcomes (f1).
Beyond biological susceptibility, inequities in acne burden are strongly driven by structural barriers to affordable and continuous care. Socioeconomic disadvantage is associated with delayed access to dermatologic services, limited availability of evidence-based treatments due to cost, fragmented follow-up, and greater reliance on non-regulated skincare practices. Health policy analyses consistently demonstrate that such access-related barriers disproportionately affect low-income and underserved populations, reinforcing preventable chronicity and long-term sequelae (ref. 438, ref. 439).
Future research and policy efforts should therefore prioritize larger interventional studies and community-level exposomics approaches that integrate environmental exposure data, population health indicators, and healthcare access metrics. The value of scalable, cost-efficient exposome methodologies for identifying high-risk communities and informing targeted interventions without reliance on expensive individual-level profiling is evident (ref. 440, ref. 441). Such approaches align exposome science with public health surveillance and equity-oriented policy frameworks (ref. 19).
From a health systems perspective, an exposome-informed equity framework supports low-cost, high-impact interventions, including integration of simplified, evidence-based acne management pathways into primary care and community health services, improved access to affordable non-comedogenic skincare, and population-level mitigation of environmental exposures such as air pollution and occupational hazards. At a policy level, incorporating acne management into primary-care clinical guidelines and ensuring reimbursement for essential topical therapies and basic skincare products could represent a pragmatic, equity-oriented strategy to reduce disparities in acne outcomes. Furthermore, promotion of healthy, balanced diets and lifestyle behaviors through educational programs should be incorporated to support overall better wellbeing and long-term disease prevention.
In conclusion, aligning exposome research with healthcare policy and equity principles enables a shift toward more accessible, preventive, and sustainable acne care. Emphasizing affordability, access, and community-level intervention ensures that advances in exposome science translate into meaningful population health gains rather than exacerbating existing inequities in dermatologic care.
References
- AHS Heng, FT Chew. Systematic review of the epidemiology of acne vulgaris.. Sci Rep (, 2020. [DOI | PubMed]
- WCAM Witkam, SE Dal Belo, S Pourhamidi, E Raynaud, M Moreau, L Aguilar. The epidemiology of acne vulgaris in a multiethnic adolescent population from Rotterdam, the Netherlands: A cross-sectional study.. J Am Acad Dermatol (, 2024. [DOI | PubMed]
- HH Oon, DZJ Lim, JS Barbieri, B Dréno, CL Goh, AM Layton. A call for action to tackle the increasing global burden and challenges of acne.. J Eur Acad Dermatol Venereol. (, 2023. [DOI | PubMed]
- L Lukaviciute, R Ganceviciene, P Navickas, A Navickas, J Grigaitiene, CC Zouboulis. Anxiety, depression, and suicidal ideation amongst patients with facial dermatoses (Acne, rosacea, perioral dermatitis, and folliculitis) in Lithuania.. Dermatology. (, 2020. [DOI | PubMed]
- AL Zaenglein, AL Pathy, BJ Schlosser, A Alikhan, HE Baldwin, DS Berson. Guidelines of care for the management of acne vulgaris.. J Am Acad Dermatol (, 2016. [DOI | PubMed]
- CC Zouboulis. Acne and sebaceous gland function.. Clin Dermatol (, 2004. [DOI | PubMed]
- DL Guguluş, D Vâţă, IA Popescu, AI Pătraşcu, IA Halip, M Mocanu. The epidemiology of acne in the current era: trends and clinical implications.. Cosmetics. (, 2025. [DOI | PubMed]
- A Alexis, J Tan, M Rocha, D Kerob, A Demessant, F Ly. Is acne the same around the world?. J Clin Aesthet Dermatol (, 2024
- CC Zouboulis, GJ Yoshida, Y Wu, L Xia, MR Schneider. Sebaceous gland: Milestones of 30-year modelling research dedicated to the “brain of the skin.. Exp Dermatol (, 2020. [DOI | PubMed]
- CC Zouboulis. Endocrinology and immunology of acne: Two sides of the same coin.. Exp Dermatol (, 2020. [DOI | PubMed]
- CC Zouboulis, T Coenye, L He, K Kabashima, T Kobayashi, C Niemann. Sebaceous immunobiology – skin homeostasis, pathophysiology, coordination of innate immunity and inflammatory response and disease associations.. Front Immunol (, 2022. [DOI | PubMed]
- CC Zouboulis, MK Oeff, N Hiroi, E Makrantonaki, SR Bornstein. Involvement of pattern recognition receptors in the direct influence of bacterial components and standard antiacne compounds on human sebaceous gland cells.. Skin Pharmacol Physiol (, 2021. [DOI | PubMed]
- J Kim, MT Ochoa, SR Krutzik, O Takeuchi, S Uematsu, AJ Legaspi. Activation of toll-like receptor 2 in acne triggers inflammatory cytokine responses.. J Immunol (, 2002. [DOI | PubMed]
- AS Oliveira, C Gaspar, J Rolo, R Palmeira-de-Oliveira, JP Teixeira, J Martinez-de-Oliveira. Comparative efficacy of essential oils against Cutibacterium acnes: Effect upon strains from phylotypes with different virulence patterns.. Microb Pathog (, 2025. [DOI | PubMed]
- KJ Spittaels, K van Uytfanghe, CC Zouboulis, C Stove, A Crabbé, T Coenye. Porphyrins produced by acneic Cutibacterium acnes strains activate the inflammasome by inducing K+ leakage.. iScience. (, 2021. [DOI | PubMed]
- T Passeron, J Krutmann, ML Andersen, R Katta, CC Zouboulis. Clinical and biological impact of the exposome on the skin.. J Eur Acad Dermatol Venereology. (, 2020. [DOI | PubMed]
- CP Wild. The exposome: from concept to utility.. Int J Epidemiol. (, 2012. [DOI | PubMed]
- K Grafanaki, A Bania, EG Kaliatsi, E Vryzaki, Y Vasilopoulos, S Georgiou. The imprint of exposome on the development of atopic dermatitis across the lifespan: A narrative review.. J Clin Med (, 2023. [DOI | PubMed]
- K Grafanaki, D Bakoli Sgourou, A Maniatis, E Pasmatzi. The exposomal imprint on rosacea: More than skin deep.. J Eur Acad Dermatol Venereol (, 2025. [DOI | PubMed]
- J Krutmann, D Moyal, W Liu, S Kandahari, GS Lee, N Nopadon. Pollution and acne: is there a link?. Clin Cosmet Investig Dermatol (, 2017. [DOI | PubMed]
- JY Hong, YH Choi, YJ Roh, MK Lee, CC Zouboulis, KY Park. Effect of afzelin on inflammation and lipogenesis in particulate matter-stimulated Cacnes-treated SZ95 sebocytes.. Front Med (Lausanne). (, 2025. [DOI | PubMed]
- K Cao, G Chen, W Chen, X Hou, T Hu, L Lu. Formalin-killed Propionibacterium acnes activates the aryl hydrocarbon receptor and modifies differentiation of SZ95 sebocytes in vitro.. Eur J Dermatol (, 2021. [DOI | PubMed]
- B Dréno, V Bettoli, E Araviiskaia, M Sanchez Viera, A Bouloc. The influence of exposome on acne.. J Eur Acad Dermatol Venereol. (, 2018. [DOI | PubMed]
- CC Zouboulis, N Kollias. Acne lesions cycle.. J Eur Acad Dermatol Venereol. (, 2025. [DOI | PubMed]
- J Piquero-Casals, D Morgado-Carrasco, E Rozas-Muñoz, JF Mir-Bonafé, C Trullàs, E Jourdan. Sun exposure, a relevant exposome factor in acne patients and how photoprotection can improve outcomes.. J cosmetic Dermatol (, 2023. [DOI | PubMed]
- J Kleemann, J Cinatl, S Hoffmann, N Zöller, D Özistanbullu, CC Zouboulis. Alcohol promotes lipogenesis in sebocytes-implications for acne.. Cells. (, 2024. [DOI | PubMed]
- E Flori, A MastroFrancesco, M Ottaviani, M Maiellaro, CC Zouboulis, E Camera. Desaturation of sebaceous-type saturated fatty acids through the SCD1 and the FADS2 pathways impacts lipid neosynthesis and inflammatory response in sebocytes in culture.. Exp Dermatol (, 2023. [DOI | PubMed]
- AM Hossini, X Hou, T Exner, B Fauler, J Eberle, A Rabien. Free fatty acids induce lipid accumulation, autophagy, and apoptosis in human sebocytes.. Skin Pharmacol Physiol (, 2023. [DOI | PubMed]
- D Törőcsik, F Fazekas, S Póliska, A Gregus, EA Janka, K Dull. Epidermal growth factor modulates palmitic acid-induced inflammatory and lipid signaling pathways in SZ95 sebocytes.. Front Immunol (, 2021. [DOI | PubMed]
- C Cai, S Liu, Y Liu, S Huang, S Lu, F Liu. Paeoniflorin mitigates insulin-like growth factor 1-induced lipogenesis and inflammation in human sebocytes by inhibiting the PI3K/Akt/FoxO1 and JAK2/STAT3 signaling pathways.. Nat Prod Bioprospect. (, 2024. [DOI | PubMed]
- TM Smith, K Gilliland, GA Clawson, D Thiboutot. IGF-1 induces SREBP-1 expression and lipogenesis in SEB-1 sebocytes via activation of the phosphoinositide 3-kinase/Akt pathway.. J Invest Dermatol (, 2008. [DOI | PubMed]
- J Li, YC Huang, JM Deng, M Yu, CC Zouboulis, GL Wang. Tea (Camellia sinensis) Seed Saponins Act as Sebosuppression Agents via the AMPK/mTOR Pathway.. J Cosmet Dermatol (, 2025. [DOI | PubMed]
- YT Su, CC Zouboulis, W Cui, AP Zhang. Lactoferrin regulates sebogenesis and inflammation in SZ95 human sebocytes and mouse model of acne.. J Cosmet Dermatol (, 2023. [DOI | PubMed]
- Z Wei, G Chen, T Hu, X Mo, X Hou, K Cao. Resveratrol ameliorates lipid accumulation and inflammation in human SZ95 sebocytes via the AMPK signaling pathways in vitro.. J Dermatol Sci (, 2021. [DOI | PubMed]
- EJM Taylor, Y Yu, J Champer, J Kim. Resveratrol demonstrates antimicrobial effects against propionibacterium acnes in vitro.. Dermatol Ther (Heidelb) (, 2014. [DOI | PubMed]
- S Saric, M Notay, RK Sivamani. Green tea and other tea polyphenols: effects on sebum production and acne vulgaris.. Antioxidants (Basel). (, 2016. [DOI | PubMed]
- H Chan, G Chan, J Santos, K Dee, JK Co. A randomized, double-blind, placebo-controlled trial to determine the efficacy and safety of lactoferrin with vitamin E and zinc as an oral therapy for mild to moderate acne vulgaris.. Int J Dermatol (, 2017. [DOI | PubMed]
- ASM Morshed, T Noor, MA Uddin Ahmed, FS Mili, S Ikram, M Rahman. Understanding the impact of acne vulgaris and associated psychological distress on self-esteem and quality of life via regression modeling with CADI, DLQI, and WHOQoL.. Sci Rep (, 2023. [DOI | PubMed]
- SM Gallitano, DS Berson. How acne bumps cause the blues: the influence of acne vulgaris on self-esteem.. Int J Womens Dermatol (, 2018. [DOI | PubMed]
- V Borrel, P Thomas, C Catovic, PJ Racine, Y Konto-Ghiorghi, L Lefeuvre. Acne and stress: impact of catecholamines on cutibacterium acnes.. Front Med (Lausanne). (, 2019. [DOI | PubMed]
- KJ Spittaels, R Ongena, CC Zouboulis, A Crabbé, T Coenye. Cutibacterium acnes phylotype I and II strains interact differently with human skin cells.. Front Cell Infect Microbiol (, 2020. [DOI | PubMed]
- CE Haddad, NE Gerbaka, S Hallit, C Tabet. Association between exposure to ambient air pollution and occurrence of inflammatory acne in the adult population.. BMC Public Health (, 2021. [DOI | PubMed]
- W Liu, X Pan, A Vierkötter, Q Guo, X Wang, Q Wang. A time-series study of the effect of air pollution on outpatient visits for acne vulgaris in beijing.. Skin Pharmacol Physiol (, 2018. [DOI | PubMed]
- HJ Kim, IH Bae, ED Son, J Park, N Cha, HW Na. Transcriptome analysis of airborne PM2.5-induced detrimental effects on human keratinocytes.. Toxicol Lett (, 2017. [DOI | PubMed]
- BE Kim, J Kim, E Goleva, E Berdyshev, J Lee, KA Vang. Particulate matter causes skin barrier dysfunction.. JCI Insight (, 2021. [DOI | PubMed]
- A Ali, H Khan, R Bahadar, A Riaz, MHHB Asad. The impact of airborne pollution and exposure to solar ultraviolet radiation on skin: mechanistic and physiological insight.. Environ Sci pollut Res Int (, 2020. [DOI | PubMed]
- MA Lefebvre, DM Pham, B Boussouira, D Bernard, C Camus, QL Nguyen. Evaluation of the impact of urban pollution on the quality of skin: a multicentre study in Mexico.. Int J cosmetic science. (, 2015. [DOI | PubMed]
- DY Lee, K Yamasaki, J Rudsil, CC Zouboulis, GT Park, JM Yang. Sebocytes express functional cathelicidin antimicrobial peptides and can act to kill propionibacterium acnes.. J Invest Dermatol (, 2008. [DOI | PubMed]
- HH Noh, SH Shin, YJ Roh, NJ Moon, SJ Seo, KY Park. Particulate matter increases Cutibacterium acnes-induced inflammation in human epidermal keratinocytes via the TLR4/NF-κB pathway.. PloS One (, 2022. [DOI | PubMed]
- MH Kwack, NG Ha, WJ Lee. Dieckol inhibits the effects of particulate matter 10 on sebocytes, outer root sheath cells, and cutibacterium acnes-pretreated mice.. Ann Dermatol (, 2022. [DOI | PubMed]
- EY Lim. Kim GD. Particulate matter-induced emerging health effects associated with oxidative stress and inflammation.. Antioxidants (Basel). (, 2024. [DOI | PubMed]
- T Coenye, K Honraet, B Rossel, HJ Nelis. Biofilms in skin infections: Propionibacterium acnes and acne vulgaris (. 2008
- D Roosterman, T Goerge, SW Schneider, NW Bunnett, M Steinhoff. Neuronal control of skin function: the skin as a neuroimmunoendocrine organ.. Physiol Rev (, 2006. [DOI | PubMed]
- MA Lefebvre, DM Pham, B Boussouira, H Qiu, C Ye, X Long. Consequences of urban pollution upon skin statusA controlled study in Shanghai area.. Int J Cosmet Sci (, 2016. [DOI | PubMed]
- G Bocheva, RM Slominski, AT Slominski. Environmental air pollutants affecting skin functions with systemic implications.. Int J Mol Sci (, 2023. [DOI | PubMed]
- HS Han, J Seok, KY Park. Air pollution and skin diseases.. Ann Dermatol (, 2025. [DOI | PubMed]
- SB Tucker. Occupational tropical acne.. Cutis. (, 1983. [PubMed]
- G Plewig, AM Kligman. Acne tropicalis (Tropical acne), in:. ACNE and ROSACEA (, 2000
- M Gfesser, WI Worret. Seasonal variations in the severity of acne vulgaris.. Int J Dermatol (, 1996. [DOI | PubMed]
- K Sardana, RC Sharma, R Sarkar. Seasonal variation in acne vulgaris–myth or reality.. J Dermatol (, 2002. [DOI | PubMed]
- I Narang, K Sardana, R Bajpai, VK Garg. Seasonal aggravation of acne in summers and the effect of temperature and humidity in a study in a tropical setting.. J cosmetic Dermatol (, 2019. [DOI | PubMed]
- WJ Lee, SY Chae, HS Ryu, YH Jang, SJ Lee, DW Kim. Inflammatory Cytokine Expression and Sebum Production after Exposure of Cultured Human Sebocytes to Ultraviolet A Radiation and Light at Wavelengths of 650 nm and 830 nm.. Ann Dermatol (, 2015. [DOI | PubMed]
- WJ Lee, KH Park, MY Sohn, WC Lee, SJ Lee, DW Kim. Ultraviolet B irradiation increases the expression of inflammatory cytokines in cultured sebocytes.. J Dermatol (, 2013. [DOI | PubMed]
- B Dreno, J Shourick, D Kerob, A Bouloc, C Taïeb. The role of exposome in acne: results from an international patient survey.. J Eur Acad Dermatol VenereologyJEADV. (, 2020. [DOI | PubMed]
- TL Scott, PA Christian, MV Kesler, KM Donohue, B Shelton, K Wakamatsu. Pigment-independent cAMP-mediated epidermal thickening protects against cutaneous UV injury by keratinocyte proliferation.. Exp Dermatol (, 2012. [DOI | PubMed]
- DH Suh, TE Kwon, JI Youn. Changes of comedonal cytokines and sebum secretion after UV irradiation in acne patients.. Eur J Dermatol (, 2002
- V Patra, SN Byrne, P Wolf. The skin microbiome: is it affected by UV-induced immune suppression?. Front Microbiol (, 2016. [DOI | PubMed]
- S Letsiou, E Koldiri, A Beloukas, E Rallis, V Kefala. Deciphering the effects of different types of sunlight radiation on skin function: A review.. Cosmetics. (, 2024. [DOI | PubMed]
- J Xu, S Huang, Z Fu, W Zheng, W Luo, N Zhuang. Effects of light and laser therapies on the microecosystem of sebaceous glands in acne treatment.. Photodermatol Photoimmunol Photomed. (, 2025. [DOI | PubMed]
- MS Nestor. The use of photodynamic therapy for treatment of acne vulgaris.. Dermatol Clin (, 2007. [DOI | PubMed]
- C Wang, M Duan. Association among presentation, severity, and risk factors of acne vulgaris among Chinese college students: A cross-sectional study.. Med (Baltimore). (, 2025. [DOI | PubMed]
- Y Dong, G Zhou, J Chen, L Shen, Z Jianxin, Q Xu. A new LED device used for photodynamic therapy in treatment of moderate to severe acne vulgaris.. Photodiagnosis Photodyn Ther (, 2016. [DOI | PubMed]
- D Piccolo, D Kostaki, C Dianzani, G Crisman, C Conforti. Effective intense pulsed light protocol in the treatment of moderate to severe acne vulgaris of the chest and back.. J Clin Aesthet Dermatol (, 2022
- HH Kwon, JB Lee, JY Yoon, SY Park, HH Ryu, BM Park. The clinical and histological effect of home-use, combination blue-red LED phototherapy for mild-to-moderate acne vulgaris in Korean patients: a double-blind, randomized controlled trial.. Br J Dermatol (, 2013. [DOI | PubMed]
- P Chen, S Li, Y Xiao, B Zou, J Li, X Chen. Long-term exposure to low levels of ambient UVB are associated with a decreased risk of moderate-to-severe acne: A retrospective cohort study in college students.. Photodermatology photoimmunology photomedicine. (, 2023. [DOI | PubMed]
- Y Wang, W Zhu, M Shu, Y Jiang, RL Gallo, YT Liu. The response of human skin commensal bacteria as a reflection of UV radiation: UV-B decreases porphyrin production.. PloS One (, 2012. [DOI | PubMed]
- R Haag, O Gurow, M Mack, J Moisel, M Hessling. Enhancement and Limitations of Green-Spectrum Dual-Wavelength Irradiation in Porphyrin-Based Antimicrobial Strategies Targeting Cutibacterium acnes subsp. Elongatum.. Pharmaceutics. (, 2026. [DOI | PubMed]
- ZJ Li, DK Choi, KC Sohn, MS Seo, HE Lee, Y Lee. Propionibacterium acnes activates the NLRP3 inflammasome in human sebocytes.. J Invest Dermatol (, 2014. [DOI | PubMed]
- J Tschopp, K Schroder. NLRP3 inflammasome activation: The convergence of multiple signalling pathways on ROS production?. Nat Rev Immunol (, 2010. [DOI | PubMed]
- G Yang, SJ Lee, HC Kang, YY Cho, HS Lee, CC Zouboulis. Repurposing auranofin, an anti-rheumatic gold compound, to treat acne vulgaris by targeting the NLRP3 inflammasome.. Biomol Ther (Seoul). (, 2020. [DOI | PubMed]
- A Sahib, A Abu Raghif. Oxidative stress in acne vulgaris: an important therapeutic target.. J Mol Pathophysiology. (, 2013. [DOI | PubMed]
- HA Al-Shobaili. Oxidants and anti-oxidants status in acne vulgaris patients with varying severity.. Ann Clin Lab Sci (, 2014
- A Kaminsky, M Florez-White, E Bagatin, MI Arias. Large prospective study on adult acne in Latin America and the Iberian Peninsula: risk factors, demographics, and clinical characteristics. Int J Dermatol (, 2019. [DOI | PubMed]
- D Morgado-Carrasco, J Piquero-Casals, C Granger, C Trullàs, T Passeron. Melasma: The need for tailored photoprotection to improve clinical outcomes.. Photodermatol Photoimmunol Photomed. (, 2022. [DOI | PubMed]
- E Austin, AN Geisler, J Nguyen, I Kohli, I Hamzavi, HW Lim. Visible light. Part I: Properties and cutaneous effects of visible light.. J Am Acad Dermatol (, 2021. [DOI | PubMed]
- DD Lynn, T Umari, CA Dunnick, RP Dellavalle. The epidemiology of acne vulgaris in late adolescence.. Adolesc Health Med Ther (, 2016. [DOI | PubMed]
- V Bettoli, A Guerra-Tapia, MI Herane, J Piquero-Martín. Challenges and solutions in oral isotretinoin in acne: reflections on 35 years of experience.. Clin Cosmet Investig Dermatol (, 2019. [DOI | PubMed]
- JQ Del Rosso. Clinical relevance of skin barrier changes associated with the use of oral isotretinoin: the importance of barrier repair therapy in patient management.. J Drugs Dermatol (, 2013
- AM Layton, WJ Cunliffe. Phototoxic eruptions due to doxycycline–a dose-related phenomenon.. Clin Exp Dermatol (, 1993. [DOI | PubMed]
- JA Wehlie, SA İsse, MA Omar, SA Sheikh, Aİ Ali, TA Ali. Determinants of awareness of risks associated with skin lightening products among female dermatology patients in Mogadishu: a cross-sectional study.. BMC Womens Health (, 2025. [DOI | PubMed]
- MY Francis, E Minja, M Dennis, G Shayo. Skin lightening practice and its cutaneous effects among women in kinondoni municipality, dar es salaam, Tanzania.. J Cosmet Dermatol (, 2025. [DOI | PubMed]
- S Alsuwaidan, KN Nagshabandi, A Alajlan. Severe nodulocystic acne induced by mercury-containing skin-whitening cream: A case report and call for public awareness.. Case Rep Dermatol (, 2024. [DOI | PubMed]
- R Bali, A Ji-Xu, SJ Felton. The significant health threat from sunbed use as a self treatment in patients with acne.. Clin Exp Dermatol (, 2022. [DOI | PubMed]
- C Dessinioti, AJ Stratigos. An epidemiological update on indoor tanning and the risk of skin cancers.. Curr Oncol (, 2022. [DOI | PubMed]
- T Passeron, HW Lim, CL Goh, HY Kang, F Ly, A Morita. Photoprotection according to skin phototype and dermatoses: practical recommendations from an expert panel.. J Eur Acad Dermatol Venereol. (, 2021. [DOI | PubMed]
- RV Reynolds, H Yeung, CE Cheng, F Cook-Bolden, SR Desai, KM Druby. Guidelines of care for the management of acne vulgaris.. J Am Acad Dermatol (, 2024. [DOI | PubMed]
- CL Goh, Y Wu, B Welsh, MF Abad-Casintahan, CJ Tseng, J Sharad. Expert consensus on holistic skin care routine: Focus on acne, rosacea, atopic dermatitis, and sensitive skin syndrome.. J Cosmet Dermatol (, 2023. [DOI | PubMed]
- WP Bowe, LH Kircik. The importance of photoprotection and moisturization in treating acne vulgaris.. J Drugs Dermatol (, 2014
- SQ Wang, P Virmani, HW Lim. Consumer acceptability and compliance: the next frontier in sunscreen innovation.. Photodermatol Photoimmunol Photomed. (, 2016. [DOI | PubMed]
- AB Lyons, C Trullas, I Kohli, IH Hamzavi, HW Lim. Photoprotection beyond ultraviolet radiation: A review of tinted sunscreens.. J Am Acad Dermatol (, 2021. [DOI | PubMed]
- S Kalia. The evolution, safety, and public perception of sunscreens: A historical and contemporary review.. Photodermatol Photoimmunol Photomed. (, 2025. [DOI | PubMed]
- H Dumbuya, PE Grimes, S Lynch, K Ji, M Brahmachary, Q Zheng. Impact of iron-oxide containing formulations against visible light-induced skin pigmentation in skin of color individuals.. J Drugs Dermatol (, 2020. [DOI | PubMed]
- PE Grimes, J Paturi, Y Chen, J Wangari-Olivero, H Dumbuya, X Yan. Photoprotection efficacy of sun protection factor and iron oxide formulations in diverse skin with melasma and photodamage.. J Drugs Dermatol (, 2025. [DOI | PubMed]
- AF Alexis, H Woolery-Lloyd, K Williams, A Andriessen, VD Callender, S Kang. Racial/ethnic variations in acne: implications for treatment and skin care recommendations for acne patients with skin of color.. J Drugs Dermatol (, 2021. [DOI | PubMed]
- A Alexis, H Woolery-Lloyd, A Andriessen, S Kang, D Rodriguez, V Callender. Racial/ethnic variations in acne: A practical algorithm for treatment and maintenance, including skincare recommendations for skin of color patients with acne.. J Drugs Dermatol (, 2022. [PubMed]
- A Andriessen, AC Rodas Diaz, PC Gameros, O Macias, JR Neves, CG Gonzalez. Over the counter products for acne treatment and maintenance in latin america: A review of current clinical practice.. J Drugs Dermatol (, 2021. [DOI | PubMed]
- VD Callender, H Baldwin, FE Cook-Bolden, AF Alexis, L Stein Gold, E Guenin. Effects of topical retinoids on acne and post-inflammatory hyperpigmentation in patients with skin of color: A clinical review and implications for practice.. Am J Clin Dermatol (, 2022. [DOI | PubMed]
- VD Callender, AF Alexis, SR Daniels, AK Kawata, CT Burk, TK Wilcox. Racial differences in clinical characteristics, perceptions and behaviors, and psychosocial impact of adult female acne.. J Clin Aesthet Dermatol (, 2014
- W Liu, J Krutmann, Y Tian, C Granger, J Piquero-Casals, C Trullàs. Commentary: facial aesthetic dermatological procedures and photoprotection in chinese populations.. Dermatol Ther (Heidelb). (, 2023. [DOI | PubMed]
- LL Levy, JJ Emer. Emotional benefit of cosmetic camouflage in the treatment of facial skin conditions: personal experience and review.. Clin Cosmet Investig Dermatol (, 2012. [DOI | PubMed]
- DE Castillo, JE Keri. Chemical peels in the treatment of acne: patient selection and perspectives.. Clin Cosmet Investig Dermatol (, 2018. [DOI | PubMed]
- E Zorba, A Karpouzis, A Zorbas, T Bazas, S Zorbas, E Alexopoulos. Occupational dermatoses by type of work in Greece.. Saf Health Work. (, 2013. [DOI | PubMed]
- GA Moustafa, E Xanthopoulou, E Riza, A Linos. Skin disease after occupational dermal exposure to coal tar: a review of the scientific literature.. Int J Dermatol (, 2015. [DOI | PubMed]
- BB Adams, VB Chetty, DF Mutasim. Periorbital comedones and their relationship to pitch tar: a cross-sectional analysis and a review of the literature.. J Am Acad Dermatol (, 2000. [DOI | PubMed]
- Q Ju, CC Zouboulis, L Xia. Environmental pollution and acne: Chloracne.. Dermatoendocrinol. (, 2009. [DOI | PubMed]
- O Yamamoto, Y Tokura. Photocontact dermatitis and chloracne: two major occupational and environmental skin diseases induced by different actions of halogenated chemicals.. J Dermatol Sci (, 2003. [DOI | PubMed]
- LT Budnik, R Wegner, U Rogall, X Baur. Accidental exposure to polychlorinated biphenyls (PCB) in waste cargo after heavy seas.. Global waste transport as Source PCB exposure. Int Arch Occup Environ Health (, 2014. [DOI | PubMed]
- AT Patterson, FT Tian, DM Elston, BH Kaffenberger. Occluded cigarette smoke exposure causing localized chloracne-like comedones.. Dermatology. (, 2015. [DOI | PubMed]
- C Han, J Shi, Y Chen, Z Zhang. Increased flare of acne caused by long time mask wearing during COVID -19 pandemic among general population.. Dermatologic Ther (, 2020
- SK Rathi, JM Dsouza. Maskne: A new acne variant in covid-19 era.. Indian J Dermatol (, 2022. [DOI | PubMed]
- T Montero-Vilchez, C Cuenca-Barrales, A Martinez-Lopez, A Molina-Leyva, S Arias-Santiago. Skin adverse events related to personal protective equipment: a systematic review and meta-analysis.. J Eur Acad Dermatol Venereol. (, 2021. [DOI | PubMed]
- I Proietti, I Borrelli, N Skroza, PE Santoro, MR Gualano, N Bernardini. Adverse skin reactions to personal protective equipment during COVID-19 pandemic in Italian health care workers.. Dermatol Ther (, 2022. [DOI | PubMed]
- N Hamnerius, A Pontén, O Bergendorff, M Bruze, J Björk, C Svedman. Skin exposures, hand eczema and facial skin disease in healthcare workers during the COVID-19 pandemic: A cross-sectional study.. Acta Derm Venereol (, 2021. [DOI | PubMed]
- WYB Ho, LYC Tan, X Zhao, D Wang, HLJ Lim. Epidemiology of occupational dermatoses associated with personal protective equipment use in the COVID-19 pandemic: Risk factors and mitigation strategies for frontline health care workers.. JAAD Int (, 2022. [DOI | PubMed]
- S Cretu, M Dascalu, SR Georgescu, CM Salavastru. Personal protective equipment use and face acne in health care providers during the COVID-19 pandemic in Romania: A new occupational acne type?. J Eur Acad Dermatol Venereol (, 2022. [DOI | PubMed]
- S Abdali, J Yu. Occupational dermatoses related to personal protective equipment used during the COVID-19 pandemic.. Dermatologic clinics. (, 2021. [DOI | PubMed]
- L Techasatian, S Lebsing, R Uppala, W Thaowandee, J Chaiyarit, C Supakunpinyo. The effects of the face mask on the skin underneath: A prospective survey during the COVID-19 pandemic.. J Prim Care Community Health (, 2020. [DOI | PubMed]
- A Tunçer Vural. The development of acne vulgaris due to face masks during the pandemic, risk awareness and attitudes of a group of university students.. J Cosmetic Dermatol (, 2022. [DOI | PubMed]
- SP Raju, M Sachdev, N Khunger, N Madnani. Mask acne in skin of color: A significant dermatological condition amidst the COVID-19 pandemic.. J Clin Aesthet Dermatol (, 2022
- N Metin, Ç Turan, Z Utlu. Changes in dermatological complaints among healthcare professionals during the COVID-19 outbreak in Turkey.. Acta Dermatovenerol Alp Pannonica Adriat. (, 2020. [DOI | PubMed]
- A Dani, A Eseonu, K Bibee. Risk factors for the development of acne in healthcare workers during the COVID-19 pandemic.. Arch Dermatol Res (, 2023. [DOI | PubMed]
- R Elisheva. Adverse Effects of Prolonged Mask Use among Healthcare Professionals during COVID-19.. J Infect Dis Epidemiol (, 2020
- P Wolkenstein, A Machovcová, JC Szepietowski, D Tennstedt, S Veraldi, A Delarue. Acne prevalence and associations with lifestyle: a cross-sectional online survey of adolescents/young adults in 7 European countries.. J Eur Acad Dermatol VenereologyJEADV. (, 2018. [DOI | PubMed]
- S Rombouts, T Nijsten, J Lambert. Cigarette smoking and acne in adolescents: results from a cross-sectional study.. J Eur Acad Dermatol VenereologyJEADV. (, 2007. [DOI | PubMed]
- I Klaz, I Kochba, T Shohat, S Zarka, S Brenner. Severe acne vulgaris and tobacco smoking in young men.. J Invest Dermatol (, 2006. [DOI | PubMed]
- T Schäfer, A Nienhaus, D Vieluf, J Berger, J Ring. Epidemiology of acne in the general population: the risk of smoking.. Br J Dermatol (, 2001. [DOI | PubMed]
- AAT Chuh, V Zawar, WCW Wong, A Lee. The association of smoking and acne in men in Hong Kong and in India: a retrospective case-control study in primary care settings.. Clin Exp Dermatol (, 2004. [DOI | PubMed]
- B Capitanio, JL Sinagra, M Ottaviani, V Bordignon, A Amantea, M Picardo. Acne and smoking.. Dermato-endocrinology. (, 2009. [DOI | PubMed]
- AD Landro, S Cazzaniga, F Parazzini, V Ingordo, F Cusano, L Atzori. Family history, body mass index, selected dietary factors, menstrual history, and risk of moderate to severe acne in adolescents and young adults.. J Am Acad Dermatol (, 2012. [DOI | PubMed]
- JZ Zhang, F Xiang, SR Yu, D Luo, TT Li, XJ Kang. Association between acne and smoking: systematic review and meta-analysis of observational studies.. Chin Med J (, 2021. [DOI | PubMed]
- B Capitanio, JL Sinagra, V Bordignon, P Cordiali Fei, M Picardo, CC Zouboulis. Underestimated clinical features of postadolescent acne.. J Am Acad Dermatol (, 2010. [DOI | PubMed]
- P Wolkenstein, L Misery, JM Amici, R Maghia, S Branchoux, C Cazeau. Smoking and dietary factors associated with moderate-to-severe acne in French adolescents and young adults: results of a survey using a representative sample.. Dermatology. (, 2015. [DOI | PubMed]
- G Valacchi, C Sticozzi, A Pecorelli, F Cervellati, C Cervellati, E Maioli. Cutaneous responses to environmental stressors.. Ann N Y Acad Sci (, 2012. [DOI | PubMed]
- LN Jorgensen, F Kallehave, E Christensen, JE Siana, F Gottrup. Less collagen production in smokers.. Surgery. (, 1998. [DOI]
- M Just, M Ribera, E Monsó, JC Lorenzo, C Ferrándiz. Effect of smoking on skin elastic fibres: morphometric and immunohistochemical analysis.. Br J Dermatol (, 2007. [DOI | PubMed]
- YS Yang, HK Lim, KK Hong, MK Shin, JW Lee, SW Lee. Cigarette smoke-induced interleukin-1 alpha may be involved in the pathogenesis of adult acne.. Ann Dermatol (, 2014. [DOI | PubMed]
- M Ottaviani, T Alestas, E Flori, A MastroFrancesco, CC Zouboulis, M Picardo. Peroxidated squalene induces the production of inflammatory mediators in HaCaT keratinocytes: a possible role in acne vulgaris.. J Invest Dermatol (, 2006. [DOI | PubMed]
- I Crivellari, C Sticozzi, G Belmonte, XM Muresan, F Cervellati, A Pecorelli. SRB1 as a new redox target of cigarette smoke in human sebocytes.. Free Radic Biol Med (, 2017. [DOI | PubMed]
- C Sticozzi, G Belmonte, A Pecorelli, B Arezzini, C Gardi, E Maioli. Cigarette smoke affects keratinocytes SRB1 expression and localization via H2O2 production and HNE protein adducts formation.. PloS One (, 2012. [DOI | PubMed]
- S Yeroushalmi, D Nemirovsky, M Mamlouk, D Feldman, K Nelson, A Sparks. Consumer perspectives on and utilization of medical cannabis to treat dermatologic conditions.. J Drugs Dermatol (, 2022. [DOI | PubMed]
- T Sheriff, MJ Lin, D Dubin, H Khorasani. The potential role of cannabinoids in dermatology.. J Dermatolog Treat (, 2020. [DOI | PubMed]
- A Oláh, BI Tóth, I Borbíró, K Sugawara, AG Szöllõsi, G Czifra. Cannabidiol exerts sebostatic and antiinflammatory effects on human sebocytes.. J Clin Invest. (, 2014. [DOI | PubMed]
- A Oláh, A Markovics, J Szabó-Papp, PT Szabó, C Stott, CC Zouboulis. Differential effectiveness of selected non-psychotropic phytocannabinoids on human sebocyte functions implicates their introduction in dry/seborrhoeic skin and acne treatment.. Exp Dermatol (, 2016. [DOI | PubMed]
- A Ali, N Akhtar. The safety and efficacy of 3% Cannabis seeds extract cream for reduction of human cheek skin sebum and erythema content.. Pak J Pharm Sci (, 2015
- S Jin, MY Lee. The ameliorative effect of hemp seed hexane extracts on the Propionibacterium acnes-induced inflammation and lipogenesis in sebocytes.. PloS One (, 2018. [DOI | PubMed]
- DH Marks, A Friedman. The therapeutic potential of cannabinoids in dermatology.. Skin Ther Lett (, 2018
- AM Kligman, OH Mills. Acne cosmetica.. Arch Dermatol (, 1972. [DOI | PubMed]
- JE Fulton, SR Pay, JE Fulton. Comedogenicity of current therapeutic products, cosmetics, and ingredients in the rabbit ear.. J Am Acad Dermatol (, 1984. [DOI | PubMed]
- CC Zouboulis, E Jourdan, M Picardo. Acne is an inflammatory disease and alterations of sebum composition initiate acne lesions.. J Eur Acad Dermatol Venereol. (, 2014. [DOI | PubMed]
- JQ Del Rosso, L Kircik. The primary role of sebum in the pathophysiology of acne vulgaris and its therapeutic relevance in acne management.. J Dermatolog Treat (, 2024. [DOI | PubMed]
- DE Branisteanu, MP Toader, EA Porumb, IL Serban, AC Pinzariu, CI Branisteanu. Adult female acne: Clinical and therapeutic particularities (Review).. Exp Ther Med (, 2022. [DOI | PubMed]
- V Goulden, GI Stables, WJ Cunliffe. Prevalence of facial acne in adults.. J Am Acad Dermatol (, 1999
- AC Perkins, J Maglione, GG Hillebrand, K Miyamoto, AB Kimball. Acne vulgaris in women: prevalence across the life span.. J Womens Health (Larchmt). (, 2012. [DOI | PubMed]
- F Poli, B Dreno, M Verschoore. An epidemiological study of acne in female adults: results of a survey conducted in France.. J Eur Acad Dermatol Venereol. (, 2001. [DOI | PubMed]
- RM George, R Sridharan. Factors aggravating or precipitating acne in Indian adults: A hospital-based study of 110 cases.. Indian J Dermatol (, 2018. [DOI | PubMed]
- Q Xia, Z Wang, Y Tang, X Luan, T Deng, L Fan. Exploring the influencing factors on acne, melasma, and rosacea: A case-control study in China.. J cosmetic Dermatol (, 2024. [DOI | PubMed]
- D Mijaljica, JP Townley, F Spada, IP Harrison. The heterogeneity and complexity of skin surface lipids in human skin health and disease.. Prog Lipid Res (, 2024. [DOI | PubMed]
- LA Schachner, AF Alexis, A Andriessen, D Berson, M Gold, DJ Goldberg. Insights into acne and the skin barrier: Optimizing treatment regimens with ceramide-containing skincare.. J Cosmet Dermatol (, 2023. [DOI | PubMed]
- MH Schmid-Wendtner, HC Korting. The pH of the skin surface and its impact on the barrier function.. Skin Pharmacol Physiol (, 2006. [DOI | PubMed]
- K Grafanaki, C Antonatos, A Maniatis, A Petropoulou, E Vryzaki, Y Vasilopoulos. Intrinsic effects of exposome in atopic dermatitis: genomics, epigenomics and regulatory layers.. J Clin Med (, 2023. [DOI | PubMed]
- M Lukić, I Pantelić, SD Savić. Towards optimal pH of the skin and topical formulations: from the current state of the art to tailored products.. Cosmetics. (, 2021. [DOI | PubMed]
- J Tarun, J Susan, J Suria, VJ Susan, S Criton. Evaluation of pH of bathing soaps and shampoos for skin and hair care.. Indian J Dermatol (, 2014. [DOI | PubMed]
- K Choi, H Liu, Y Zhu, Z Jiang, S Lu. A case-control study exploring the association between cosmetic use and acne risk: implications for prevention and clinical practice.. Clin Cosmet Investig Dermatol (, 2025. [DOI | PubMed]
- D Thiboutot, AM Layton, I Traore, G Gontijo, P Troielli, Q Ju. International expert consensus recommendations for the use of dermocosmetics in acne.. J Eur Acad Dermatol Venereol. (, 2025. [DOI | PubMed]
- I Kurokawa, M Kobayashi, Y Nomura, M Abe, D Kerob, B Dreno. The role and benefits of dermocosmetics in acne management in Japan.. Dermatol Ther (Heidelb). (, 2023. [DOI | PubMed]
- JKL Tan, K Bhate. A global perspective on the epidemiology of acne.. Br J Dermatol (, 2015. [DOI | PubMed]
- HS Kim, JY Ko, DH Suh, HJ Ryu, E Baek, S Cho. Addressing the unmet need in acne management: A novel dermocosmetics guideline tailored to asian patient subgroups.. Cosmetics. (, 2024. [DOI | PubMed]
- G Monfrecola, S Cacciapuoti, C Capasso, M Delfino, G Fabbrocini. Tolerability and camouflaging effect of corrective makeup for acne: results of a clinical study of a novel face compact cream.. Clin Cosmet Investig Dermatol (, 2016. [DOI | PubMed]
- V Bettoli, C Coutanceau, V Georgescu. A real-life, international, observational study demonstrating the efficacy of A cosmetic emulsion in the supportive care of mild-to-moderate facial acne.. Clin Cosmet Investig Dermatol (, 2019. [DOI | PubMed]
- S Laneri, I Dini, A Tito, R Di Lorenzo, M Bimonte, A Tortora. Plant cell culture extract of Cirsium eriophorum with skin pore refiner activity by modulating sebum production and inflammatory response.. Phytother Res (, 2021. [DOI | PubMed]
- F Bageorgou, L Li, C Beausillon, A Stennevin, A Ortiz-Brugués, JH Saurat. Tolerability and effectiveness of a dermocosmetic product containing Silybum marianum fruit extract in adolescents and young adults with acne-prone skin: An international, phase IV, longitudinal study.. J Cosmet Dermatol (, 2023. [DOI | PubMed]
- E Bagatin, MD Thouvenin, A Bacquey, S Baradat, C Lauze, V Mengeaud. The usefulness of a dermocosmetic containing Myrtus communis extract and azelaic acid for maintenance phase of adult female acne: Results from a randomized exploratory investigator-blinded comparative study.. J Eur Acad Dermatol Venereol. (, 2023. [DOI | PubMed]
- XM Xu, L Sansores-Garcia, XM Chen, N Matijevic-Aleksic, M Du, KK Wu. Suppression of inducible cyclooxygenase 2 gene transcription by aspirin and sodium salicylate.. Proc Natl Acad Sci U S A. (, 1999. [DOI | PubMed]
- D Huccetogullari, ZW Luo, SY Lee. Metabolic engineering of microorganisms for production of aromatic compounds.. Microb Cell Fact. (, 2019. [DOI | PubMed]
- AG Petrovici, M Spennato, I Bîtcan, F Péter, L Cotarcă, A Todea. A comprehensive review of azelaic acid pharmacological properties, clinical applications, and innovative topical formulations.. Pharm (Basel). (, 2025. [DOI | PubMed]
- M Detmar, A Mayer-da-Silva, R Stadler, CE Orfanos. Effects of azelaic acid on proliferation and ultrastructure of mouse keratinocytes in vitro.. J Invest Dermatol (, 1989. [DOI | PubMed]
- C Marques, F Hadjab, A Porcello, K Lourenço, C Scaletta, P Abdel-Sayed. Mechanistic insights into the multiple functions of niacinamide: therapeutic implications and cosmeceutical applications in functional skincare products.. Antioxidants (Basel). (, 2024. [DOI | PubMed]
- O Tanno, Y Ota, N Kitamura, T Katsube, S Inoue. Nicotinamide increases biosynthesis of ceramides as well as other stratum corneum lipids to improve the epidermal permeability barrier.. Br J Dermatol (, 2000. [DOI | PubMed]
- A Khammari, D Kerob, AL Demessant, M Nioré, B Dréno. A dermocosmetic regimen is able to mitigate skin sensitivity induced by a retinoid-based fixed combination treatment for acne: Results of a randomized clinical trial.. J Cosmet Dermatol (, 2024. [DOI | PubMed]
- G Fabbrocini, M Saint Aroman. Cosmeceuticals based on Rhealba(®) Oat plantlet extract for the treatment of acne vulgaris.. J Eur Acad Dermatol Venereol. (, 2014. [DOI | PubMed]
- G Fabbrocini, AB Rossi, MD Thouvenin, C Peraud, V Mengeaud, A Bacquey. Fragility of epidermis: acne and post-procedure lesional skin.. J Eur Acad Dermatol Venereol. (, 2017. [DOI | PubMed]
- F Carballido, A Philippe, M Maitre, C Lauze, N Chanssard, L Garidou. A dermocosmetic product containing the sap of oat plantlets and Garcinia mangostana extract improves the clinical signs of acne.. J Eur Acad Dermatol Venereol. (, 2024. [DOI | PubMed]
- A Khammari, A Demessant-Flavigny, D Kerob, S Seité, B Dréno. A salicylic acid-based dermocosmetic is effective as an adjunct to benzoyl peroxide for mild to moderate acne and as monotherapy in maintenance post benzoyl peroxide.. J Drugs Dermatol (, 2023. [DOI | PubMed]
- T Tempark, A Shem, S Lueangarun. Efficacy of ceramides and niacinamide-containing moisturizer versus hydrophilic cream in combination with topical anti-acne treatment in mild to moderate acne vulgaris: A split face, double-blinded, randomized controlled trial.. J Cosmet Dermatol (, 2024. [DOI | PubMed]
- M Belmontesi. Integrating dermocosmetic therapy for acne: addressing severity levels in real-life experiences.. J Drugs Dermatol (, 2025. [DOI | PubMed]
- IBS Sitohang, L Norawati, SW Yenny, A Kusumawardani, S Murlistyarini, SV Setiawan. Effectiveness and safety of a dermocosmetic cream as an adjunct to adapalene for mild and moderate acne in Indonesia: results of a multicenter randomized controlled study.. Clin Cosmet Investig Dermatol (, 2024. [DOI | PubMed]
- L Cordain, S Lindeberg, M Hurtado, K Hill, SB Eaton, J Brand-Miller. Acne vulgaris: a disease of Western civilization.. Arch Dermatol (, 2002. [DOI | PubMed]
- A Khan, MW Chang. The role of nutrition in acne vulgaris and hidradenitis suppurativa.. Clinics Dermatol (, 2022. [DOI | PubMed]
- BC Melnik. Linking diet to acne metabolomics, inflammation, and comedogenesis: an update.. Clin Cosmet Investig Dermatol (, 2015. [DOI | PubMed]
- EA González-Mondragón, LDC Ganoza-Granados, ME Toledo-Bahena, AM Valencia-Herrera, MR Duarte-Abdala, KA Camargo-Sánchez. Acne and diet: a review of pathogenic mechanisms.. Bol Med Hosp Infant Mex. (, 2022. [DOI | PubMed]
- J Meixiong, C Ricco, C Vasavda, BK Ho. Diet and acne: A systematic review.. JAAD Int (, 2022. [DOI | PubMed]
- I Ryguła, W Pikiewicz, K Kaminiów. Impact of diet and nutrition in patients with acne vulgaris.. Nutrients. (, 2024. [DOI | PubMed]
- CA Adebamowo, D Spiegelman, FW Danby, AL Frazier, WC Willett, MD Holmes. High school dietary dairy intake and teenage acne.. J Am Acad Dermatol (, 2005. [DOI | PubMed]
- BC Melnik. Lifetime impact of cow’s milk on overactivation of mTORC1: from fetal to childhood overgrowth, acne, diabetes, cancers, and neurodegeneration.. Biomolecules (, 2021. [DOI | PubMed]
- BC Melnik. Western diet-induced imbalances of FoxO1 and mTORC1 signalling promote the sebofollicular inflammasomopathy acne vulgaris.. Exp Dermatol (, 2016. [DOI | PubMed]
- B Melnik. Dietary intervention in acne: Attenuation of increased mTORC1 signaling promoted by Western diet.. Dermatoendocrinol. (, 2012. [DOI | PubMed]
- S Li, MS Brown, JL Goldstein. Bifurcation of insulin signaling pathway in rat liver: mTORC1 required for stimulation of lipogenesis, but not inhibition of gluconeogenesis.. Proc Natl Acad Sci U S A. (, 2010. [DOI | PubMed]
- CR Juhl, HKM Bergholdt, IM Miller, GBE Jemec, JK Kanters, C Ellervik. Dairy intake and acne vulgaris: A systematic review and meta-analysis of 78,529 children, adolescents, and young adults.. Nutrients (, 2018. [DOI | PubMed]
- M Aghasi, M Golzarand, S Shab-Bidar, A Aminianfar, M Omidian, F Taheri. Dairy intake and acne development: A meta-analysis of observational studies.. Clin Nutr (Edinburgh Scotland). (, 2019. [DOI | PubMed]
- R Dai, W Hua, W Chen, L Xiong, L Li. The effect of milk consumption on acne: a meta-analysis of observational studies.. J Eur Acad Dermatol VenereologyJEADV. (, 2018. [DOI | PubMed]
- RN Smith, NJ Mann, A Braue, H Mäkeläinen, GA Varigos. A low-glycemic-load diet improves symptoms in acne vulgaris patients: a randomized controlled trial.. Am J Clin Nutr (, 2007. [DOI | PubMed]
- RN Smith, A Braue, GA Varigos, NJ Mann. The effect of a low glycemic load diet on acne vulgaris and the fatty acid composition of skin surface triglycerides.. J Dermatol science. (, 2008. [DOI | PubMed]
- K Cao, Y Liu, N Liang, X Shen, R Li, H Yin. Fatty acid profiling in facial sebum and erythrocytes from adult patients with moderate acne.. Front Physiol (, 2022. [DOI | PubMed]
- İ Aslan, F Özcan, T Karaarslan, E Kıraç, M Aslan. Decreased eicosapentaenoic acid levels in acne vulgaris reveals the presence of a proinflammatory state.. Prostaglandins Other Lipid Mediat. (, 2017. [DOI | PubMed]
- A Guertler, T Fiedler, D Lill, AC Kuna, A Volsky, J Wallmichrath. Deficit of omega-3 fatty acids in acne patients-A cross-sectional pilot study in a german cohort.. Life (Basel). (, 2024. [DOI | PubMed]
- C Conforti, M Agozzino, G Emendato, A Fai, F Fichera, GF Marangi. Acne and diet: a review.. Int J Dermatol (, 2022. [DOI | PubMed]
- A Balić, D Vlašić, B Marinović, ZB Mokos. Omega-3 versus omega-6 polyunsaturated fatty acids in the prevention and treatment of inflammatory skin diseases.. Int J Mol Sci (, 2020. [DOI | PubMed]
- G Khayef, J Young, B Burns-Whitmore, T Spalding. Effects of fish oil supplementation on inflammatory acne.. Lipids Health disease. (, 2012. [DOI | PubMed]
- JY Jung, HH Kwon, JS Hong, JY Yoon, MS Park, MY Jang. Effect of dietary supplementation with omega-3 fatty acid and gamma-linolenic acid on acne vulgaris: a randomised, double-blind, controlled trial.. Acta dermato-venereologica. (, 2014. [DOI | PubMed]
- TH Imam, J Wang, FS Khayat. Bariatric surgery in a patient on peritoneal dialysis.. Perit Dial Int (, 2013. [DOI | PubMed]
- P Sánchez-Pellicer, L Navarro-Moratalla, E Núñez-Delegido, B Ruzafa-Costas, J Agüera-Santos, V Navarro-López. Acne, microbiome, and probiotics: the gut-skin axis.. Microorganisms. (, 2022. [DOI | PubMed]
- X He, Z Zhang, H Jiang, H Luo, Q Gan, K Wei. Causal association of gut microbes and blood metabolites with acne identified through systematic mendelian randomization.. Sci Rep (, 2024. [DOI | PubMed]
- Y Huang, F Liu, J Lai, S Jiang, X Tan, L Chen. The adjuvant treatment role of ω-3 fatty acids by regulating gut microbiota positively in the acne vulgaris.. J Dermatolog Treat (, 2024. [DOI | PubMed]
- S Nambidi, NS Sennie, SB Kondaveeti, A Banerjee, S Pathak, AK Duttaroy. A review of short-Chain fatty acids in gut and skin: Possible implications in skin aging.. J Funct Foods. (, 2025. [DOI | PubMed]
- S Krishna, JP Okhovat, J Kim, CN Kim. Influence of ω-3 fatty acids on triglyceride levels in patients using isotretinoin.. JAMA Dermatol (, 2015. [DOI | PubMed]
- KY Park, EJ Ko, IS Kim, K Li, BJ Kim, SJ Seo. The effect of evening primrose oil for the prevention of xerotic cheilitis in acne patients being treated with isotretinoin: a pilot study.. Ann Dermatol (, 2014. [DOI | PubMed]
- D Kovács, E Camera, S Póliska, A Cavallo, M Maiellaro, K Dull. Linoleic acid induced changes in SZ95 sebocytes-comparison with palmitic acid and arachidonic acid.. Nutrients. (, 2023. [DOI | PubMed]
- BR Kim, G Kim, SP Jin, CW Choi, J Kim, H Park. Causal association between polyunsaturated fatty acids and acne: a two-sample Mendelian randomization study.. Br J Dermatol (, 2025. [DOI | PubMed]
- M Liu, S Diaz-Torres, BL Mitchell, D Toledo-Flores, P Gharhakhani, MA Simpson. The role of lipid metabolism in acne risk: integrating blood metabolite and genetic insights.. Skin Health Dis (, 2025. [DOI | PubMed]
- NB Silverberg. Whey protein precipitating moderate to severe acne flares in 5 teenaged athletes.. Cutis. (, 2012
- S Vongraviopap, P Asawanonda. Dark chocolate exacerbates acne.. Int J Dermatol (, 2016. [DOI | PubMed]
- C Caperton, S Block, M Viera, J Keri, B Berman. Double-blind, placebo-controlled study assessing the effect of chocolate consumption in subjects with a history of acne vulgaris.. J Clin aesthetic Dermatol (, 2014
- FP Cengiz, BC Cemil, N Emiroglu, AG Bahali, N Onsun. Acne located on the trunk, whey protein supplementation: Is there any association?. Health promotion Perspect (, 2017. [DOI | PubMed]
- BC Melnik. Evidence for acne-promoting effects of milk and other insulinotropic dairy products.. Nestle Nutr Workshop Ser Pediatr Program. (, 2011. [DOI | PubMed]
- T Simonart. Acne and whey protein supplementation among bodybuilders.. Dermatology. (, 2012. [DOI | PubMed]
- J Muhaidat, A Qablan, F Gharaibeh, GH Albataineh, N Abdo, D Alshiyab. The effect of whey protein supplements on acne vulgaris among male adolescents and young adults: A case-control study from north of Jordan.. Dermatol Res practice. (, 2024. [DOI | PubMed]
- K Sompochpruetikul, T Khongcharoensombat, Y Chongpison, W Rittirongwattana, P Asawanonda, N Noppakun. Whey protein and male acne: A double-blind, randomized controlled trial.. J Dermatol (, 2024. [DOI | PubMed]
- M Picardo, M Ottaviani, E Camera, A MastroFrancesco. Sebaceous gland lipids.. Dermatoendocrinol. (, 2009. [DOI | PubMed]
- MR Schneider, R Paus. Sebocytes, multifaceted epithelial cells: lipid production and holocrine secretion.. Int J Biochem Cell Biol (, 2010. [DOI | PubMed]
- M Schmidt, H Binder, MR Schneider. The metabolic underpinnings of sebaceous lipogenesis.. Commun Biol (, 2025. [DOI | PubMed]
- RW Clayton, K Göbel, CM Niessen, R Paus, M van Steensel M a., X Lim. Homeostasis of the sebaceous gland and mechanisms of acne pathogenesis.. Br J Dermatol (, 2019. [DOI | PubMed]
- FW Danby. Ductal hypoxia in acne: is it the missing link between comedogenesis and inflammation?. J Am Acad Dermatol (, 2014. [DOI | PubMed]
- Jy Park, Jy Jung, Hj Kim, B IH, K DY, L TR. Hypoxia leads to abnormal epidermal differentiation via HIF-independent pathways.. Biochem Biophys Res Commun (, 2016. [DOI | PubMed]
- K Choi, M Jin, CC Zouboulis, Y Lee. Increased lipid accumulation under hypoxia in SZ95 human sebocytes.. Dermatology. (, 2021. [DOI | PubMed]
- J Zhang, Y Chang, S Liu, J Tuo, Z Xu, J Ying. ALA-PDT promotes IL-1β secretion from human SZ95 sebocytes via activation of the NLRP3 inflammasome.. Photodiagnosis Photodyn Ther (, 2024. [DOI | PubMed]
- Z Wei, M Gao, Y Liu, R Zeng, J Liu, S Sun. TRPV3 promotes sebocyte inflammation via transcriptional modulating TLR2 in acne.. Biochim Biophys Acta Mol Basis Dis (, 2024. [DOI | PubMed]
- Y Lee, K Shin, KO Shin, S Yoon, J Jung, E Hwang. Topical application of autophagy-activating peptide improved skin barrier function and reduced acne symptoms in acne-prone skin.. J Cosmet Dermatol (, 2021. [DOI | PubMed]
- SH Seo, JY Jung, K Park, AM Hossini, CC Zouboulis, SE Lee. Autophagy regulates lipid production and contributes to the sebosuppressive effect of retinoic acid in human SZ95 sebocytes.. J Dermatol Sci (, 2020. [DOI | PubMed]
- R Wu, J Li, H Tian, D Song, T Zhao, Y Tian. Unveiling the mechanism of photothermal therapy in acne man-agement: targeting sebaceous gland ferroptosis via umbilical cord mesenchymal stem cell membrane-encapsulated Au-Ag-PDA.. Front Bioeng Biotechnol (, 2024. [DOI | PubMed]
- MAM Van Steensel. The genetics of acne.. Ann Hum Genet (, 2025. [DOI | PubMed]
- R Lichtenberger, MA Simpson, C Smith, J Barker, AA Navarini. Genetic architecture of acne vulgaris.. J Eur Acad Dermatol VenereologyJEADV. (, 2017. [DOI | PubMed]
- H Zhang, Z Zhang. Genetic variants associated with acne vulgaris.. Int J Gen Med (, 2023. [DOI | PubMed]
- SX Xu, HL Wang, X Fan, LD Sun, S Yang, PG Wang. The familial risk of acne vulgaris in Chinese Hans – a case-control study.. J Eur Acad Dermatol Venereology. (, 2007. [DOI | PubMed]
- J Karciauskiene, S Valiukeviciene, H Gollnick, A Stang. The prevalence and risk factors of adolescent acne among schoolchildren in Lithuania: a cross-sectional study.. J Eur Acad Dermatol Venereology. (, 2014. [DOI | PubMed]
- V Bataille, H Snieder, AJ MacGregor, P Sasieni, TD Spector. The influence of genetics and environmental factors in the pathogenesis of acne: a twin study of acne in women.. J Invest Dermatol (, 2002. [DOI | PubMed]
- A Mina-Vargas, L Colodro-Conde, K Grasby, G Zhu, S Gordon, SE Medland. Heritability and GWAS analyses of acne in Australian adolescent twins.. Twin Res Hum genetics Off J Int Soc Twin Stud (, 2017. [DOI | PubMed]
- L He, WJ Wu, JK Yang, H Cheng, XB Zuo, W Lai. Two new susceptibility loci 1q24.2 and 11p11.2 confer risk to severe acne.. Nat Commun (, 2014. [DOI | PubMed]
- AA Navarini, MA Simpson, M Weale, J Knight, I Carlavan, P Reiniche. Genome-wide association study identifies three novel susceptibility loci for severe Acne vulgaris.. Nat Commun (, 2014. [DOI | PubMed]
- BL Mitchell, JR Saklatvala, N Dand, FA Hagenbeek, X Li, JL Min. Genome-wide association meta-analysis identifies 29 new acne susceptibility loci.. Nat Commun (, 2022. [DOI | PubMed]
- M Teder-Laving, M Kals, A Reigo, R Ehin, T Objärtel, M Vaht. Genome-wide meta-analysis identifies novel loci conferring risk of acne vulgaris.. Eur J Hum Genet (, 2024. [DOI | PubMed]
- C Petridis, AA Navarini, N Dand, J Saklatvala, D Baudry, M Duckworth. Genome-wide meta-analysis implicates mediators of hair follicle development and morphogenesis in risk for severe acne.. Nat Commun (, 2018. [DOI | PubMed]
- A Vallée, Y Lecarpentier, R Guillevin, JN Vallée. Interactions between TGF-β1, canonical WNT/β-catenin pathway and PPAR γ in radiation-induced fibrosis.. Oncotarget. (, 2017. [DOI | PubMed]
- M Sato. Upregulation of the Wnt/beta-catenin pathway induced by transforming growth factor-beta in hypertrophic scars and keloids.. Acta Derm Venereol. (, 2006. [DOI | PubMed]
- G Katerina, M Alexandros, S Vasilina, P Efstathia, T Argyris. Fibrotic disease of the skin and lung: shared pathways, environmental drivers, and therapeutic opportunities in a changing climate (. 2025
- MM Ali, RM Porter, ML Gonzalez. Intense pulsed light enhances transforming growth factor beta1/Smad3 signaling in acne-prone skin.. J Cosmet Dermatol (, 2013. [DOI | PubMed]
- W Shang, AYQ Tan, MAM van Steensel, X Lim. Aberrant wnt signaling induces comedo-like changes in the murine upper hair follicle.. J Invest Dermatol (, 2022. [DOI | PubMed]
- AHS Heng, YH Say, YY Sio, YT Ng, FT Chew. Gene variants associated with acne vulgaris presentation and severity: a systematic review and meta-analysis.. BMC Med Genomics (, 2021. [DOI | PubMed]
- R Ju, Y Ying, Q Zhou, Y Cao. Exploring genetic drug targets in acne vulgaris: A comprehensive proteome-wide mendelian randomization study.. J Cosmet Dermatol (, 2024. [DOI | PubMed]
- X Wang, Y Wu, P Zhao, X Wang, W Wu, J Yang. The causal relationship between serum metabolites and acne vulgaris: a Mendelian randomization study.. Sci Rep (, 2024. [DOI | PubMed]
- NR Trivedi, KL Gilliland, W Zhao, W Liu, DM Thiboutot. Gene array expression profiling in acne lesions reveals marked upregulation of genes involved in inflammation and matrix remodeling.. J Invest Dermatol (, 2006. [DOI | PubMed]
- CM Chronnell, LR Ghali, RS Ali, AG Quinn, DB Holland, JJ Bull. Human beta defensin-1 and -2 expression in human pilosebaceous units: upregulation in acne vulgaris lesions.. J Invest Dermatol (, 2001. [DOI | PubMed]
- NA Veniaminova, YY Jia, AM Hartigan, TJ Huyge, SY Tsai, M Grachtchouk. Distinct mechanisms for sebaceous gland self-renewal and regeneration provide durability in response to injury.. Cell Rep (, 2023. [DOI | PubMed]
- J Burclaff, JC Mills. Plasticity of differentiated cells in wound repair and tumorigenesis, part II: skin and intestine.. Dis Model Mech (, 2018. [DOI | PubMed]
- G Donati, FM Watt. Stem cell heterogeneity and plasticity in epithelia.. Cell Stem Cell (, 2015. [DOI | PubMed]
- S Baroud, J Wu, CC Zouboulis. Acne syndromes and mosaicism.. Biomedicines. (, 2021. [DOI | PubMed]
- HA Abdelkader, I Amin, LA Rashed, M Samir, M Ezzat. Histone deacetylase 1 in patients with alopecia areata and acne vulgaris: An epigenetic alteration.. Australas J Dermatol (, 2022. [DOI | PubMed]
- HS Shin, CC Zouboulis, MK Kim, DH Lee, JH Chung. Minocycline suppresses lipogenesis via inhibition of p300 histone acetyltransferase activity in human SZ95 sebocytes.. J Eur Acad Dermatol Venereol. (, 2022. [DOI | PubMed]
- H Gu, HJ An, MG Gwon, S Bae, CC Zouboulis, KK Park. The effects of synthetic SREBP-1 and PPAR-γ Decoy oligodeoxynucleotide on acne-like disease in vivo and in vitro via lipogenic regulation.. Biomolecules. (, 2022. [DOI | PubMed]
- A Markovics, Á Angyal, KF Tóth, D Ádám, Z Pénzes, J Magi. GPR119 is a potent regulator of human sebocyte biology.. J Invest Dermatol (, 2020. [DOI | PubMed]
- D Ádám, J Arany, KF Tóth, O Pető, T Nyitrai, BI Tóth. The TRPM5 antagonist triphenylphosphine oxide increases sebaceous lipogenesis and modulates immune phenotype of human sebocytes in a TRPM5-independent manner.. Exp Dermatol (, 2025. [DOI | PubMed]
- L Liu, Y Xue, J Chen, Y Li, T Chen, X Pan. DNA methylation profiling and integrative multi-omics analysis of skin samples reveal important contribution of epigenetics and immune response in the pathogenesis of acne vulgaris.. Clin Immunol (Orlando Fla). (, 2023. [DOI | PubMed]
- H Wang, T Dang, J Feng, W Wu, L He, J Yang. Identification of differentially methylated genes for severe acne by genome-wide DNA methylation and gene expression analysis.. Epigenetics. (, 2023. [DOI | PubMed]
- Y Wu, Y Chen, B Chen, W Wu, J Yang. DNA methylation mediated genetic risk in severe acne in a young men population.. Front Med (Lausanne). (, 2023. [DOI | PubMed]
- K Gong, X Ji, S Wu, DD Zhao, M Zhu. DNA Methylation at cg18095732 Modulates ZDHHC20 Expression and Decreases Acne Vulgaris Risk.. Clin Cosmet Investig Dermatol (, 2025. [DOI | PubMed]
- M Toyoda, M Morohashi. Pathogenesis of acne.. Med Electron Microsc. (, 2001. [DOI | PubMed]
- E Firlej, W Kowalska, K Szymaszek, J Roliński, J Bartosińska. The role of skin immune system in acne.. J Clin Med (, 2022. [DOI | PubMed]
- Z Jin, Y Song, L He. A review of skin immune processes in acne.. Front Immunol (, 2023. [DOI | PubMed]
- M Qin, A Pirouz, MH Kim, SR Krutzik, HJ Garbán, J Kim. Propionibacterium acnes Induces IL-1β secretion via the NLRP3 inflammasome in human monocytes.. J Invest Dermatol (, 2014. [DOI | PubMed]
- M Mohamed, HAA Shehata, NF Fahmy, R Saleh. Evaluation of serum levels of interleukins 1-beta, 10 and 12 in patients with acne vulgaris.. J Cosmet Dermatol (, 2022. [DOI | PubMed]
- Y Xue, M Luo, X Hu, X Li, J Shen, W Zhu. Macrophages regulate vascular smooth muscle cell function during atherosclerosis progression through IL-1β/STAT3 signaling.. Commun Biol (, 2022. [DOI | PubMed]
- M Kistowska, S Gehrke, D Jankovic, K Kerl, A Fettelschoss, L Feldmeyer. IL-1β drives inflammatory responses to propionibacterium acnes in vitro and in vivo.. J Invest Dermatol (, 2014. [DOI | PubMed]
- AHT Jeremy, DB Holland, SG Roberts, KF Thomson, WJ Cunliffe. Inflammatory events are involved in acne lesion initiation.. J Invest Dermatol (, 2003. [DOI | PubMed]
- M Kistowska, B Meier, T Proust, L Feldmeyer, A Cozzio, T Kuendig. Propionibacterium acnes promotes Th17 and Th17/Th1 responses in acne patients.. J Invest Dermatol (, 2015. [DOI | PubMed]
- I Carlavan, B Bertino, M Rivier, P Martel, V Bourdes, M Motte. Atrophic scar formation in patients with acne involves long-acting immune responses with plasma cells and alteration of sebaceous glands.. Br J Dermatol (, 2018. [DOI | PubMed]
- U Radhakrishna, U Ratnamala, DD Jhala, N Vadsaria, M Patel, LV Uppala. Methylated miRNAs may serve as potential biomarkers and therapeutic targets for hidradenitis suppurativa.. J Eur Acad Dermatol Venereol. (, 2022. [DOI | PubMed]
- F Gibson, A Hanly, N Grbic, N Grunberg, M Wu, M Collard. Epigenetic dysregulation in autoimmune and inflammatory skin diseases.. Clin Rev Allergy Immunol (, 2022. [DOI | PubMed]
- Y Sawada, RL Gallo. Role of epigenetics in the regulation of immune functions of the skin.. J Invest Dermatol (, 2021. [DOI | PubMed]
- A Pink, F Anzengruber, AA Navarini. Acne and hidradenitis suppurativa.. Br J Dermatol (, 2018. [DOI | PubMed]
- BC Melnik. Milk disrupts p53 and DNMT1, the guardians of the genome: implications for acne vulgaris and prostate cancer.. Nutr Metab (Lond). (, 2017. [DOI | PubMed]
- F Borgia, L Peterle, P Custurone, M Vaccaro, G Pioggia, S Gangemi. MicroRNA cross-involvement in acne vulgaris and hidradenitis suppurativa: A literature review.. Int J Mol Sci (, 2022. [DOI | PubMed]
- K Dull, F Fazekas, D Deák, D Kovács, S Póliska, A Szegedi. miR-146a modulates TLR1/2 and 4 induced inflammation and links it with proliferation and lipid production via the indirect regulation of GNG7 in human SZ95 sebocytes.. Sci Rep (, 2021. [DOI | PubMed]
- B Caliş, FH Yerlikaya, A Ataseven, SA Temiz, DE Onmaz. Oxidative stress-related miRNAs in patients with severe acne vulgaris.. Indian J Dermatol (, 2022. [DOI | PubMed]
- N Akdağ, E Atli, D Zhuri, H Sezginer Güler, Y Gürsel Ürün. A Study of FoxO1, mTOR, miR-21, miR-29b, and miR-98 Expression Levels Regarding Metabolic Syndrome in Acne Vulgaris Patients.. Cureus (, 2024. [PubMed]
- W Ghumra, N Lee, H Whitehouse, R Bhutani, D Lagos, AM Layton. MicroRNAs as biomarkers of atrophic scarring in acne: a cross-sectional analysis of 41 patients.. Clin Exp Dermatol (, 2021. [DOI | PubMed]
- X Luo, T Chen, Yy Feng, Pm Zhou, B Kan, L Han. The association of miR-191-5p in acne vulgaris and its predictive value for disease progression.. Turkish J Biochem (, 2025
- S Zhang, Y Liang, M Xia, X Tian, Z Chen, L Lin. Serum exosomal microRNA profiling reveals a down-regulation of hsa-miR-124-3p in patients with severe acne.. Front Immunol (, 2025. [DOI | PubMed]
- S Yang, F Fang, X Yu, C Yang, X Zhang, L Wang. Knockdown of H19 inhibits the pathogenesis of acne vulgaris by targeting the miR-196a/TLR2/NF-κB axis.. Inflammation. (, 2020. [DOI | PubMed]
- Y He, W Wang, X Ma, Z Duan, B Wang, M Li. Discovery and potential functional characterization of long noncoding RNAs associated with familial acne inversa with NCSTN mutation.. Dermatology. (, 2024. [DOI | PubMed]
- J Liang, X Wu, S Sun, P Chen, X Liang, J Wang. Circular RNA expression profile analysis of severe acne by RNA-Seq and bioinformatics.. J Eur Acad Dermatol VenereologyJEADV. (, 2018. [DOI | PubMed]
- Y Liu, M Zhou, N Zheng, H Xu, X Chen, Z Duan. Hsa_circ_0105040 promotes Cutbacterium acnes biofilm induced inflammation via sponge miR-146a in human keratinocyte.. Int Immunopharmacol. (, 2024. [DOI | PubMed]
- AE El-Taweel, AE Al-Refae, M Abd-Ellateef, R Mohammad. Circular ribonucleic acid expression in patient with acne.. Egyptian J Med Res (, 2022. [DOI]
- A Niedźwiedzka, MP Micallef, M Biazzo, C Podrini. The role of the skin microbiome in acne: challenges and future therapeutic opportunities.. Int J Mol Sci (, 2024. [DOI | PubMed]
- E Barnard, B Shi, D Kang, N Craft, H Li. Author Correction: The balance of metagenomic elements shapes the skin microbiome in acne and health.. Sci Rep (, 2020. [DOI | PubMed]
- K Sripath, J Bangperng, P Chumnanpeun, T E-kobon. Unique genetic diversity of Propionibacterium acnes isolated from human facial skins in Thailand revealed by the multi-locus sequence typing scheme.. BMC Microbiol (, 2025. [DOI | PubMed]
- S Fitz-Gibbon, S Tomida, BH Chiu, L Nguyen, C Du, M Liu. Propionibacterium acnes strain populations in the human skin microbiome associated with acne.. J Invest Dermatol (, 2013. [DOI | PubMed]
- MA Dagnelie, S Corvec, M Saint-Jean, V Bourdès, JM Nguyen, A Khammari. Decrease in diversity of propionibacterium acnes phylotypes in patients with severe acne on the back.. Acta dermato-venereologica. (, 2018. [DOI | PubMed]
- C Paugam, S Corvec, M Saint-Jean, ML Moigne, A Khammari, A Boisrobert. Propionibacterium acnes phylotypes and acne severity: an observational prospective study.. J Eur Acad Dermatol VenereologyJEADV. (, 2017. [DOI | PubMed]
- K Nakase, N Hayashi, Y Akiyama, S Aoki, N Noguchi. Antimicrobial susceptibility and phylogenetic analysis of Propionibacterium acnes isolated from acne patients in Japan between 2013 and 2015.. J Dermatol (, 2017. [DOI | PubMed]
- I Cavallo, F Sivori, M Truglio, F De Maio, F Lucantoni, G Cardinali. Skin dysbiosis and Cutibacterium acnes biofilm in inflammatory acne lesions of adolescents.. Sci Rep (, 2022. [DOI | PubMed]
- EJ Choi, HG Lee, IH Bae, W Kim, J Park, TR Lee. Propionibacterium acnes-derived extracellular vesicles promote acne-like phenotypes in human epidermis.. J Invest Dermatol (, 2018. [DOI | PubMed]
- A Chudzik, P Migdał, M Paściak. Different cutibacterium acnes phylotypes release distinct extracellular vesicles.. Int J Mol Sci (, 2022. [DOI | PubMed]
- MP Cros, J Mir-Pedrol, L Toloza, N Knödlseder, J Maruotti, CC Zouboulis. New insights into the role of Cutibacterium acnes-derived extracellular vesicles in inflammatory skin disorders.. Sci Rep (, 2023. [DOI | PubMed]
- M Shu, Y Wang, J Yu, S Kuo, A Coda, Y Jiang. Fermentation of Propionibacterium acnes, a commensal bacterium in the human skin microbiome, as skin probiotics against methicillin-resistant Staphylococcus aureus.. PloS One (, 2013. [DOI | PubMed]
- JA Sanford, AM O’Neill, CC Zouboulis, RL Gallo. Short-chain fatty acids from cutibacterium acnes activate both a canonical and epigenetic inflammatory response in human sebocytes.. J Immunol (, 2019. [DOI | PubMed]
- JA Sanford, LJ Zhang, MR Williams, JA Gangoiti, CM Huang, RL Gallo. Inhibition of HDAC8 and HDAC9 by microbial short-chain fatty acids breaks immune tolerance of the epidermis to TLR ligands.. Sci Immunol (, 2016. [DOI | PubMed]
- K Szabó, F Balogh, D Romhányi, L Erdei, B Toldi, R Gyulai. Epigenetic regulatory processes involved in the establishment and maintenance of skin homeostasis-the role of microbiota.. Int J Mol Sci (, 2025. [DOI | PubMed]
- J Shi, JW Cheng, Q Zhang, ZX Hua, X Miao. Comparison of the skin microbiota of patients with acne vulgaris and healthy controls.. Ann palliative Med (, 2021. [DOI | PubMed]
- AM Schneider, ZT Nolan, K Banerjee, AR Paine, Z Cong, SL Gettle. Evolution of the facial skin microbiome during puberty in normal and acne skin.. J Eur Acad Dermatol Venereol. (, 2023. [DOI | PubMed]
- R Siddiqui, Z Makhlouf, NA Khan. The increasing importance of the gut microbiome in acne vulgaris.. Folia microbiologica. (, 2022. [DOI | PubMed]
- N Akaza, H Akamatsu, S Numata, S Yamada, A Yagami, S Nakata. Microorganisms inhabiting follicular contents of facial acne are not only Propionibacterium but also Malassezia spp.. J Dermatol (, 2016. [DOI | PubMed]
- B Dréno, S Pécastaings, S Corvec, S Veraldi, A Khammari, C Roques. Cutibacterium acnes (Propionibacterium acnes) and acne vulgaris: a brief look at the latest updates.. J Eur Acad Dermatol VenereologyJEADV. (, 2018. [DOI | PubMed]
- J McLaughlin, S Watterson, AM Layton, AJ Bjourson, E Barnard, A McDowell. Propionibacterium acnes and acne vulgaris: new insights from the integration of population genetic, multi-omic, biochemical and host-microbe studies.. Microorganisms (, 2019. [DOI | PubMed]
- J Buddenkotte, M Steinhoff. Recent advances in understanding and managing rosacea.. F1000Research. (, 2018. [DOI | PubMed]
- B Dreno, R Martin, D Moyal, JB Henley, A Khammari, S Seité. Skin microbiome and acne vulgaris: Staphylococcus, a new actor in acne.. Exp Dermatol (, 2017. [DOI | PubMed]
- HM Yan, HJ Zhao, DY Guo, PQ Zhu, CL Zhang, W Jiang. Gut microbiota alterations in moderate to severe acne vulgaris patients.. J Dermatol (, 2018. [DOI | PubMed]
- Y Deng, H Wang, J Zhou, Y Mou, G Wang, X Xiong. Patients with acne vulgaris have a distinct gut microbiota in comparison with healthy controls.. Acta Dermato Venereologica. (, 2018. [DOI | PubMed]
- B Dréno, MA Dagnelie, A Khammari, S Corvec. The skin microbiome: A new actor in inflammatory acne.. Am J Clin Dermatol (, 2020. [DOI | PubMed]
- X Ji, S Wu, D Zhao, Q Bai, Y Wang, K Gong. Revealing the impact of gut microbiota on acne through mendelian randomization analysis.. Clin Cosmet Investig Dermatol (, 2024. [DOI | PubMed]
- Q Cao, J Guo, S Chang, Z Huang, Q Luo. Gut microbiota and acne: A Mendelian randomization study.. Skin Res Technol (, 2023. [DOI | PubMed]
- EC Bal Bayazıtlı, GA Saraç, A Uzdoğan, O Acar, HB Karaman, A Külhan. Association between altered gut microbiota and acne vulgaris: A comparative study of trimethylamine N-oxide levels in patients and healthy controls.. Dermatol Pract Conceptual. (, 2025. [DOI | PubMed]
- KG Thompson, BM Rainer, C Antonescu, L Florea, EF Mongodin, S Kang. Comparison of the skin microbiota in acne and rosacea.. Exp Dermatol (, 2021. [DOI | PubMed]
- M Mohamed, A Ullah, R Hassan, M Hamza, I Mohamed, M Salam. The impact of probiotics on acne vulgaris: A meta-analysis of randomized controlled trials.. Cureus. (, 2025. [DOI | PubMed]
- A Ashkanani, G Ashkanani, M Yousef, M Rob, M Al-Marri, N Naseem. Microbiome and skin health: A systematic review of nutraceutical interventions, disease severity, inflammation, and gut microbiota.. Microorganisms. (, 2026. [DOI | PubMed]
- RA Hillier, R Gibson, T Maruthappu, K Whelan, EJ Prpa, HR Neill. Probiotics, prebiotics, and synbiotics as oral supplements for skin health, function, and disease throughout the life course: A scoping review.. Nutr Rev (, 2025. [DOI | PubMed]
- S Puebla-Barragan, G Reid. Probiotics in cosmetic and personal care products: trends and challenges.. Molecules. (, 2021. [DOI | PubMed]
- A Goodarzi, S Mozafarpoor, M Bodaghabadi, M Mohamadi. The potential of probiotics for treating acne vulgaris: A review of literature on acne and microbiota.. Dermatol Ther (, 2020. [DOI | PubMed]
- DK Lee, MJ Kim, JW Ham, HM An, MK Cha, SW Lee. In vitro evaluation of antibacterial activities and anti-inflammatory effects of Bifidobacterium spp.. addressing acne vulgaris. Arch Pharm Res (, 2012. [DOI | PubMed]
- F Deidda, A Amoruso, S Nicola, T Graziano, M Pane, L Mogna. New approach in acne therapy: A specific bacteriocin activity and a targeted anti IL-8 property in just 1 probiotic strain, the L. salivarius LS03.. J Clin Gastroenterol. (, 2018. [DOI | PubMed]
- F Rinaldi, L Marotta, A Mascolo, A Amoruso, M Pane, G Giuliani. Facial acne: A randomized, double-blind, placebo-controlled study on the clinical efficacy of a symbiotic dietary supplement.. Dermatol Ther (Heidelb). (, 2022. [DOI | PubMed]
- IAMP Sutema, IR Latarissa, IGAR Widowati, CR Sartika, NWE Ciptasari, K Lestari. Efficacy of probiotic supplements and topical applications in the treatment of acne: A scoping review of current results.. J Exp Pharmacol (, 2025. [DOI | PubMed]
- M Notay, N Foolad, AR Vaughn, RK Sivamani. Probiotics, prebiotics, and synbiotics for the treatment and prevention of adult dermatological diseases.. Am J Clin Dermatol (, 2017. [DOI | PubMed]
- L Liang, X Qi, X Jiang, T Chen, L Dong. Lactobacillus plantarum MH-301 as an effective adjuvant to isotretinoin in the treatment of acne vulgaris: a randomized and open-label trail.. Front Med (Lausanne). (, 2023. [DOI | PubMed]
- H Cui, C Guo, Q Wang, C Feng, Z Duan. A pilot study on the efficacy of topical lotion containing anti-acne postbiotic in subjects with mild -to -moderate acne.. Front Med (Lausanne). (, 2022. [DOI | PubMed]
- C Podrini, L Schramm, G Marianantoni, J Apolinarska, C McGuckin, N Forraz. Topical administration of lactiplantibacillus plantarum (SkinDuoTM) serum improves anti-acne properties.. Microorganisms. (, 2023. [DOI | PubMed]
- MJ Kim, KP Kim, E Choi, JH Yim, C Choi, HS Yun. Effects of lactobacillus plantarum CJLP55 on clinical improvement, skin condition and urine bacterial extracellular vesicles in patients with acne vulgaris: A randomized, double-blind, placebo-controlled study.. Nutrients. (, 2021. [DOI | PubMed]
- WH Tsai, CH Chou, YJ Chiang, CG Lin, CH Lee. Regulatory effects of Lactobacillus plantarum-GMNL6 on human skin health by improving skin microbiome.. Int J Med Sci (, 2021. [DOI | PubMed]
- H Cha, SK Kim, M Kook, TH Yi. Lactobacillus paraplantarum THG-G10 as a potential anti-acne agent with anti-bacterial and anti-inflammatory activities.. Anaerobe. (, 2020. [DOI | PubMed]
- I Dapkevicius, V Romualdo, AC Marques, CM Lopes, MH Amaral. Acne vulgaris topical therapies: application of probiotics as a new prevention strategy.. Cosmetics. (, 2023. [DOI | PubMed]
- FH Al-Ghazzewi, RF Tester. Effect of konjac glucomannan hydrolysates and probiotics on the growth of the skin bacterium Propionibacterium acnes in vitro.. Int J Cosmet Sci (, 2010. [DOI | PubMed]
- H Cui, C Feng, C Guo, Z Duan. Development of novel topical anti-acne cream containing postbiotics for mild-to-moderate acne: an observational study to evaluate its efficacy.. Indian J Dermatol (, 2022. [DOI | PubMed]
- MP Wei, H Yu, YH Guo, YL Cheng, YF Xie, WR Yao. Synergistic combination of Sapindoside A and B: A novel antibiofilm agent against Cutibacterium acnes.. Microbiol Res (, 2022. [DOI | PubMed]
- TT Min, K Choowongkomon, HH Htoo, P Nonejuie, D Haltrich, M Yamabhai. Anti-CAMP1 IgG promotes macrophage phagocytosis of Cutibacterium acnes type II.. Microbiol Res (, 2024. [DOI | PubMed]
- R Knackstedt, T Knackstedt, J Gatherwright. The role of topical probiotics in skin conditions: A systematic review of animal and human studies and implications for future therapies.. Exp Dermatol (, 2020. [DOI | PubMed]
- K França. Topical probiotics in dermatological therapy and skincare: A concise review.. Dermatol Ther (Heidelb). (, 2021. [DOI | PubMed]
- S Sathikulpakdee, S Kanokrungsee, P Vitheejongjaroen, N Kamanamool, M Udompataikul, M Taweechotipatr. Efficacy of probiotic-derived lotion from Lactobacillus paracasei MSMC 39–1 in mild to moderate acne vulgaris, randomized controlled trial.. J Cosmet Dermatol (, 2022. [DOI | PubMed]
- S Aghamohammad, M Rohani. Antibiotic resistance and the alternatives to conventional antibiotics: The role of probiotics and microbiota in combating antimicrobial resistance.. Microbiol Res (, 2023. [DOI | PubMed]
- A Slominski, B Zbytek, G Nikolakis, PR Manna, C Skobowiat, M Zmijewski. Steroidogenesis in the skin: implications for local immune functions.. J Steroid Biochem Mol Biol (, 2013. [DOI | PubMed]
- T Inoue, Y Miki, S Kakuo, A Hachiya, T Kitahara, S Aiba. Expression of steroidogenic enzymes in human sebaceous glands.. J Endocrinol (, 2014. [DOI | PubMed]
- M Fritsch, CE Orfanos, CC Zouboulis. Sebocytes are the key regulators of androgen homeostasis in human skin.. J Invest Dermatol (, 2001. [DOI | PubMed]
- A Chiu, SY Chon, AB Kimball. The response of skin disease to stress: changes in the severity of acne vulgaris as affected by examination stress.. Arch Dermatol (, 2003. [DOI | PubMed]
- M Samaniego, M Alonso, N Sohail, L Mostaghimi. Sleep disturbances and acne: A comprehensive review.. Dermatol Pract Concept. (, 2025. [DOI | PubMed]
- D Deplewski, RL Rosenfield. Role of hormones in pilosebaceous unit development.. Endocr Rev (, 2000. [DOI | PubMed]
- BC Melnik, G Schmitz. Role of insulin, insulin-like growth factor-1, hyperglycaemic food and milk consumption in the pathogenesis of acne vulgaris.. Exp Dermatol (, 2009. [DOI | PubMed]
- B Dréno, CC Zouboulis, J Tan, H Baldwin, JK Krishnaswamy, R Chavda. Role of retinoic acid receptor subtypes in the pathophysiology of acne.. J Drugs Dermatol (, 2023. [DOI | PubMed]
- D Kovács, K Hegyi, A Szegedi, D Deák, S Póliska, R Rühl. Isotretinoin is indirectly effective in sebocytes.. Br J Dermatol (, 2020. [DOI | PubMed]
- BC Melnik. Acne transcriptomics: fundamentals of acne pathogenesis and isotretinoin treatment.. Cells. (, 2023. [DOI | PubMed]
- CC Zouboulis, L Xia, H Akamatsu, H Seltmann, M Fritsch, S Hornemann. The human sebocyte culture model provides new insights into development and management of seborrhoea and acne.. Dermatology. (, 1998. [DOI | PubMed]
- N Emiroğlu, FP Cengiz, F Kemeriz. Insulin resistance in severe acne vulgaris.. Postepy Dermatol Alergol. (, 2015. [DOI | PubMed]
- M Nagpal, D De, S Handa, A Pal, N Sachdeva. Insulin resistance and metabolic syndrome in young men with acne.. JAMA Dermatol (, 2016. [DOI | PubMed]
- SP Sinikumpu, J Jokelainen, K Tasanen, L Huilaja. Cardiovascular and metabolic profile of subjects with acne in a cohort of middle-aged patients: A general population study of 1,932 subjects.. Acta Derm Venereol (, 2023. [DOI | PubMed]
- F Tamer. Do patients with acne tend to have increased body fat? Comparison of body composition analysis of patients with acne vulgaris and healthy individuals: a prospective case control study.. Arch Dermatol Res (, 2024. [DOI | PubMed]
- AF Bungau, AF Radu, SG Bungau, CM Vesa, DM Tit, LM Endres. Oxidative stress and metabolic syndrome in acne vulgaris: Pathogenetic connections and potential role of dietary supplements and phytochemicals.. BioMed Pharmacother. (, 2023. [DOI | PubMed]
- M Gruszczyńska, A Sadowska-Przytocka, W Szybiak, B Więckowska, K Lacka. Insulin resistance in patients with acne vulgaris.. Biomedicines. (, 2023. [DOI | PubMed]
- KE Rodriguez Baisi, AL Weaver, H Shakshouk, MM Tollefson. Acne incidence in preadolescents and association with increased body mass index: A population-based retrospective cohort study of 643 cases with age- and sex-matched community controls.. Pediatr Dermatol (, 2023. [DOI | PubMed]
- C Frénard, S Mansouri, S Corvec, A Boisrobert, A Khammari, B Dréno. Prepubertal acne: A retrospective study.. Int J Womens Dermatol (, 2021. [DOI | PubMed]
- LG Biagi, A Sañudo, E Bagatin. Severe acne and metabolic syndrome: A possible correlation.. Dermatology. (, 2019. [DOI | PubMed]
- LM Endres, AF Bungau, DM Tit, GS Bungau, A Radu, CC Diaconu. Acne vulgaris associated with metabolic syndrome: A three-case series highlighting pathophysiological links and therapeutic challenges.. Diagnostics (Basel). (, 2025. [DOI | PubMed]
- S Chandak, A Singh, B Madke, S Jawade, R Khandelwal. Acne vulgaris and metabolic syndrome: A possible association.. Cureus. (, 2022. [DOI | PubMed]
- I Podder, K Agarwal, A Anurag. Metabolic status, obesity, and quality of life in patients with acne vulgaris: A cross-sectional case-control study.. Indian J Dermatol (, 2021. [DOI | PubMed]
- M Zandi, E Behboudi, MR Shojaei, S Soltani, H Karami. Letter to the editor regarding “An overview on serology and molecular tests for COVID-19: an important challenge of the current century (doi: 10.22034/iji.2021.88660.1894.).. Iran J Immunol (, 2022. [DOI | PubMed]
- BC Melnik, CC Zouboulis. Potential role of FoxO1 and mTORC1 in the pathogenesis of Western diet-induced acne.. Exp Dermatol (, 2013. [DOI | PubMed]
- NF Agamia, DM Abdallah, O Sorour, B Mourad, DN Younan. Skin expression of mammalian target of rapamycin and forkhead box transcription factor O1, and serum insulin-like growth factor-1 in patients with acne vulgaris and their relationship with diet.. Br J Dermatol (, 2016. [DOI | PubMed]
- M Marmot, R Bell, TAJ Houweling, S Taylor. Closing the gap in a generation: health equity through action on the social determinants of health.. Lancet (, 2008. [DOI | PubMed]
- P Braveman. What are health disparities and health equity? We need to be clear.. Public Health Rep (, 2014. [DOI | PubMed]
- P Braveman, L Gottlieb. The social determinants of health: it’s time to consider the causes of the causes.. Public Health Rep (, 2014. [DOI | PubMed]
- N Douthit, S Kiv, T Dwolatzky, S Biswas. Exposing some important barriers to health care access in the rural USA.. Public Health (, 2015. [DOI | PubMed]
- ST Syed, BS Gerber, LK Sharp. Traveling towards disease: transportation barriers to health care access.. J Community Health (, 2013. [DOI | PubMed]
- UR Essien, SB Dusetzina, WF Gellad. A policy prescription for reducing health disparities-achieving pharmacoequity.. JAMA. (, 2021. [DOI | PubMed]
- E Sánchez-Sánchez, FL Fernández-Cerezo, J Díaz-Jimenez, M Rosety-Rodriguez, AJ Díaz, FJ Ordonez. Consumption of over-the-counter drugs: prevalence and type of drugs.. Int J Environ Res Public Health (, 2021. [DOI | PubMed]
- YT Liu, YW Wang, C Tu, JW Ren, J Huo, XJ Nan. Recommendations for managing adult acne and adolescent acne based on an epidemiological study conducted in China.. Sci Rep (, 2024. [DOI | PubMed]
- DX Zheng, AY Ning, MA Levoska, L Xiang, C Wong, JF Scott. Acne and social media: A cross-sectional study of content quality on TikTok.. Pediatr Dermatol (, 2021. [DOI | PubMed]
- B Irfan, I Yasin, A Yaqoob. Navigating digital dermatology: an analysis of acne-related content on tikTok.. Cureus. (, 2023. [DOI | PubMed]
- L Iyengar, S Saldanha, AH Chong. Acne: A thematic qualitative analysis of acne content on tikTok.. Australas J Dermatol (, 2025. [DOI | PubMed]
- A Yousaf, R Hagen, E Delaney, S Davis, Z Zinn. The influence of social media on acne treatment: A cross-sectional survey.. Pediatr Dermatol (, 2020. [DOI | PubMed]
- CF Finan, P Leon, L Midani, DA Hill, AC Smidt. Social media use among adolescents with acne: A cross-sectional survey study.. Pediatr Dermatol (, 2024. [DOI | PubMed]
- SS Ertekin, NS Salici, V Manav Bas, MG Karali, EZ Ergün, EB Avcı. Influence of social media and internet on treatment decisions in adult female acne patients: A cross-sectional survey study.. Dermatol Pract Concept. (, 2024. [DOI | PubMed]
- RK Bahaj, ZH Alsaggaf, MH Abduljabbar, JO Hariri. The influence of social media on the treatment of acne in Saudi Arabia.. Cureus. (, 2022. [DOI | PubMed]
- N Kluger, C Taieb, J Bedran, L Payraud, C Beausillon, A Sammain. Impact of acne on women’s social media and dating apps photo activity.. J Eur Acad Dermatol Venereol. (, 2025. [DOI | PubMed]
- M Jankowski, A Goroncy. Anatomical variants of acne differ in their impact on social perception.. J Eur Acad Dermatol Venereol. (, 2024. [DOI | PubMed]
- Z Zhang, M Zhou. The impact of social media information exposure on appearance anxiety in young acne patients: a moderated chain mediation model.. Front Psychol (, 2024. [DOI | PubMed]
- EA Rieder, A Andriessen, V Cutler, ME Gonzalez, JL Greenberg, P Lio. Dermatology in contemporary times: building awareness of social media’s association with adolescent skin disease and mental health.. J Drugs Dermatol (, 2023. [DOI | PubMed]
- JP Claudel, N Auffret, MT Leccia, F Poli, B Dréno. Acne from the young patient’s perspective.. J Eur Acad Dermatol Venereol. (, 2020. [DOI | PubMed]
- M Haimi. The tragic paradoxical effect of telemedicine on healthcare disparities- a time for redemption: a narrative review.. BMC Med Inform Decis Mak. (, 2023. [DOI | PubMed]
- CR Lyles, AE Sharma, JD Fields, Y Getachew, U Sarkar, L Zephyrin. Centering health equity in telemedicine.. Ann Fam Med (, 2022. [DOI | PubMed]
- CR Lyles, RM Wachter, U Sarkar. Focusing on digital health equity.. JAMA. (, 2021. [DOI | PubMed]
- SA Saeed, RM Masters. Disparities in health care and the digital divide.. Curr Psychiatry Rep (, 2021. [DOI | PubMed]
- R Essery, M Steele, S Easton, S Pollet, C Cairns, R LeFeuvre. Planning and optimizing a digital self-management support intervention: Acne Care Online.. Br J Health Psychol (, 2025. [DOI | PubMed]
- F Lagacé, K D’Aguanno, C Prosty, A Laverde-Saad, L Cattelan, L Ouchene. The role of sex and gender in dermatology – from pathogenesis to clinical implications.. J Cutan Med Surg (, 2023. [DOI | PubMed]
- L Ragmanauskaite, B Kahn, B Ly, H Yeung. Acne and the lesbian, gay, bisexual, or transgender teenager.. Dermatol Clin (, 2020. [DOI | PubMed]
- L Campos-Muñoz, D López-De Lara, ML Rodríguez-Rojo, A Conde-Taboada, E López-Bran. Transgender adolescents and acne: A cases series.. Pediatr Dermatol (, 2018. [DOI | PubMed]
- CC Motosko, GA Zakhem, MK Pomeranz, A Hazen. Acne: a side-effect of masculinizing hormonal therapy in transgender patients.. Br J Dermatol (, 2019. [DOI | PubMed]
- R Radi, S Gold, JP Acosta, J Barron, H Yeung. Treating acne in transgender persons receiving testosterone: A practical guide.. Am J Clin Dermatol (, 2022. [DOI | PubMed]
- CA Smith, O Kaabi, AK Manatunga, TL Lash, MJ Silverberg, D Getahun. Acne incidence and severity in transgender individuals (. 2026
- S Gold, M Siira, S Willner, C Alcid, SC Chen, V Tangpricha. Lived experience of acne and acne treatment in transgender patients.. JAMA Dermatol (, 2024. [DOI | PubMed]
- N Auffret, F Ballanger, MT Leccia, JP Claudel, B Dréno. Acne in transgender patients: disease management and treatment recommendations from a group of experts on acne.. Eur J Dermatol (, 2024. [DOI | PubMed]
- J Choe, A Shields, A Ferreira, S Gold, JW Gotschall, K Kamal. Isotretinoin for acne in transgender and gender-diverse individuals receiving masculinizing hormone therapy.. JAMA Dermatol (, 2024. [DOI | PubMed]
- C Hammill, T Vaillancourt. Acne and its association with internalizing problems.. Dermatol Rev (, 2023. [DOI | PubMed]
- LE Barnes, MM Levender, AB Fleischer, SR Feldman. Quality of life measures for acne patients.. Dermatol Clin (, 2012. [DOI | PubMed]
- A Mufaddel, AA Elnour, AA Omer, EH Alshora. Psychiatric comorbidity in patients with acne.. Open J Psychiatry (, 2017. [DOI]
- L Lukaviciute, P Navickas, A Navickas, J Grigaitiene, R Ganceviciene, CC Zouboulis. Quality of life, anxiety prevalence, depression symptomatology and suicidal ideation among acne patients in Lithuania.. J Eur Acad Dermatol Venereol. (, 2017. [DOI | PubMed]
- Y Chen, S Sun, H Yang, X Fei, Y Zhang, J Song. Global prevalence of mental health comorbidity in patients with acne: an analysis of trends from 1961 to 2023.. Clin Exp Dermatol (, 2025. [DOI | PubMed]
- AM Layton, V Bettoli, V Delore, E Puentes, JKL Tan. The burden of acne vulgaris on health-related quality of life and psychosocial well-being domains: A systematic review.. Am J Clin Dermatol (, 2025. [DOI | PubMed]
- S Samsudin, Z Zulkifli, NA Hassan, R Ismail, R Ramalingam. Psychological distress and its association with functional disability index among acne patients attending dermatology clinics in the kuantan tertiary hospitals.. MJMHS. (, 2024. [DOI]
- RK Andersen, D Bouazzi, C Erikstrup, KR Nielsen, KS Burgdorf, MT Bruun. The social and psychological impact of acne treatment: A cross-sectional study of blood donors.. J Cutan Med Surg (, 2022. [DOI | PubMed]
- K Grafanaki, S Georgiou, AJ Stratigos. Solidarity and voluntarism amid the COVID-19 pandemic: skin cancer screening for blood donors.. Dermatol Pract Concept. (, 2021. [DOI | PubMed]
- DV Szilagyi, DM Tit, CT Judea-Pusta, AF Radu, GS Bungau, A Radu. Real-world evidence of neuropsychiatric adverse reactions to isotretinoin: insights from eudravigilance (2005-2025).. Pharm (Basel). (, 2025. [DOI | PubMed]
- C Flinn, M Ungar, S Deschênes, F Nearchou. Resilience and trajectories of depressive symptoms and suicidal ideation in the context of exposure to acne: Findings from the Lifelines Cohort Study.. J Health Psychol (, 2025. [DOI | PubMed]
- A Murray, D O’Neill. Health-care equity–for all generations?. Lancet (, 2009. [DOI | PubMed]
- O O’Donnell. Health and health system effects on poverty: A narrative review of global evidence.. Health Policy. (, 2024. [DOI | PubMed]
- MK Chung, JS House, FS Akhtari, KC Makris, MA Langston, KT Islam. Decoding the exposome: data science methodologies and implications in exposome-wide association studies (ExWASs).. Exposome (, 2024. [DOI | PubMed]
- JA Stingone, AM Geller, DB Hood, KC Makris, CP Mouton, JC States. Community-level exposomics: a population-centered approach to address public health concerns.. Exposome (, 2023. [DOI | PubMed]
