Modulating Growth Factor Receptor Signaling to Promote Corneal Epithelial Homeostasis
Abstract
The corneal epithelium is the first anatomical barrier between the environment and the cornea; it is critical for proper light refraction onto the retina and prevents pathogens (e.g., bacteria, viruses) from entering the immune-privileged eye. Trauma to the highly innervated corneal epithelium is extremely painful and if not resolved quickly or properly, can lead to infection and ultimately blindness. The healthy eye produces its own growth factors and is continuously bathed in tear fluid that contains these proteins and other nutrients to maintain the rapid turnover and homeostasis of the ocular surface. In this article, we review the roles of growth factors in corneal epithelial homeostasis and regeneration and some of the limitations to their use therapeutically.
Article type: Review Article
Keywords: cornea, corneal epithelium, growth factor, growth factor receptor, c-Met, wound healing, corneal nerves
Affiliations: Department of Pharmacology and Toxicology, University of Louisville, Louisville, KY 40202, USA; Department of Ophthalmology and Vision Sciences, University of Louisville, Louisville, KY 40202, USA
License: © 2023 by the authors. CC BY 4.0 Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.3390/cells12232730 | PubMed: 38067157 | PMC: PMC10706396
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (3.0 MB)
1. Introduction
The corneal epithelium is the first anatomical barrier between the environment and the eye. Its structure is critical for proper light refraction onto the retina. The epithelial layer prevents pathogens (e.g., bacteria, viruses) from entering the immune-privileged eye and causing inflammation and scattering of light [ref. 1]. The corneal epithelium is the most densely innervated tissue in the body [ref. 2], so trauma is exceptionally painful. The healthy eye is continuously bathed in tear fluid containing proteins that are necessary for the rapid turnover and maintenance of the ocular surface [ref. 3]. Wounds to the epithelium promote the upregulation of growth factors in the tear fluid [ref. 4], exhibiting their importance in corneal wound healing. If wounds to the epithelium are not resolved quickly or properly, they can lead to infection, fibrosis, and ultimately blindness. In this article, we review the roles of growth factors in corneal epithelial homeostasis and regeneration as potential therapies and discuss some limitations to their use.
2. Corneal Epithelium
2.1. Anatomical Structure
The human cornea is comprised of 3 cellular layers separated by two distinct collagenous interfaces (Figure 1). The epithelium lies most anterior in the tissue, separated from the stroma by Bowman’s Layer. The stroma lies anterior to Descemet’s membrane, which separates it from the endothelium, the most posterior cellular layer. The endothelium interfaces with the aqueous humor and allows nutrients to diffuse to other corneal cells. It also regulates the hydration of the stroma, which is crucial for visual clarity [ref. 5].
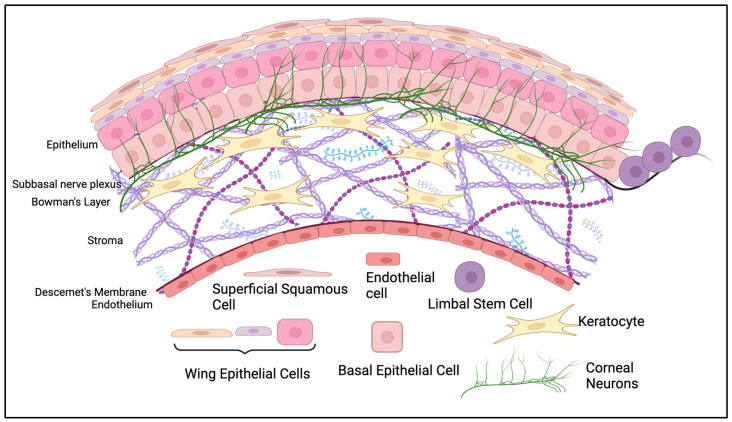
The epithelium is made of 5–7 layers of epithelial cells [ref. 1]. Corneal epithelial cells arise from limbal stem cells, which migrate to the central cornea and differentiate into basal cells [ref. 6]. As basal cells age, they differentiate and migrate anteriorly to the surface, where eventually they will reach their squamous form and shed [ref. 6]. This is a very rapid process, with the turnover of the corneal surface occurring every ~10 days [ref. 7,ref. 8]. This is advantageous for the restoration of the epithelial surface if it is ever damaged by trauma, surgery, disease, or drug side effects. The current model of corneal epithelial homeostasis is the XYZ hypothesis, where movement of limbal stem cells towards the central cornea (X), plus basal cell proliferation and differentiation in a vertical direction (Y), equals the loss of superficial squamous cells from the epithelial surface (Z) [ref. 6,ref. 9].
2.2. Tear Fluid
Because the cornea must remain transparent, there are no blood vessels to bring nutrients to the cells that reside there; rather, epithelial cells rely on the tear fluid and the aqueous humor to bathe and supply oxygen [ref. 10,ref. 11]. Epithelial cells facilitate uptake of nutrients via microvilli on its superficial layer, which allow for greater surface area interaction between the cornea and tear fluid [ref. 12]. Along with supplying the necessary molecules to maintain corneal homeostasis, the tear fluid also serves as the first protective layer of the ocular surface. It forms the barrier between the epithelium and the external environment.
In healthy eyes, the tear fluid consists of three phases. The most anterior phase is the lipid phase, or meibum, which originates in the meibomian glands located in the upper and lower eyelids. The meibum consists of different lipids and mucins that function to ensure an even spread of tear film over the surface of the eye and prevent evaporation of the aqueous layer [ref. 13]. The middle aqueous phase is created in and secreted from the lacrimal glands, which reside in the anterior lateral orbit above each eye. The aqueous layer contains mucins, electrolytes, antioxidants, and protein-tlike growth factors that contribute to the homeostasis of the ocular surface [ref. 12]. Lastly, the innermost mucin layer is secreted by the goblet cells of the conjunctiva [ref. 1,ref. 4,ref. 14,ref. 15,ref. 16].
2.3. Corneal Nerves
Between the epithelial cell layers is an intricate network of neurons that make the cornea the most densely innervated tissue in the body, with 7000 nociceptors per mm2 [ref. 2]. Corneal nerves vary in function, but the majority are sensory nerves that respond to touch, changes in temperature, and pain [ref. 17]. There are other types of nerves present in the epithelium as well, like peripheral nerves that branch from the superior cervical ganglion and supply sympathetic innervation [ref. 18,ref. 19,ref. 20]. Evidence of parasympathetic innervation in humans from the ciliary ganglion is limited, though it has been found in some animal models [ref. 2,ref. 19].
Most corneal epithelial nerves branch from the trigeminal nerve and enter the cornea radially at the stromal level. Intraepithelial corneal nerve endings (ICNs) branch upwards from the subbasal plexus, which resides beneath the epithelium but above the stroma (Figure 1) [ref. 2]. Many synapse through the lacrimal nucleus in the pons and connect to the facial nerve [ref. 21], which in turn activates the meibomian and lacrimal glands to promote production and secretion of tears [ref. 22,ref. 23]. Other outcomes of ICN activation include blinking to remove foreign objects, watering of the eye, and wound healing [ref. 2]. The increase in the production of growth factors after stimulation contributes greatly to the healing response of the cornea.
The ICNs are intimately connected with the corneal epithelial cells and are critical for their health. Because the cornea is transparent for proper light passage, ICNs shed their myelin sheaths and use the basal epithelial cells as their Schwann cell surrogates [ref. 24] (though new techniques have discovered evidence of non-myelinating Schwann cells present in the cornea [ref. 25,ref. 26,ref. 27]). Further, when primary corneal epithelial cells and trigeminal neurons are co-cultured, neurite outgrowth is increased [ref. 28]. Both cell types release factors that act on the other to help maintain homeostasis of the corneal surface. Nerve health directly impacts the health of the ocular surface.
3. Growth Factors and Their Receptors
The maintenance of a healthy cornea is driven by growth factor receptor signaling. These soluble proteins bind to their cognate receptor to induce biochemical changes inside the cell to alter its biology. For growth factors to exert their activity, their cognate receptor must be expressed on their target cells at a sufficient density to activate intracellular effectors. Although growth factors have biological roles in embryogenesis, organogenesis, and angiogenesis (and more), in mature and healthy ocular tissues they primarily contribute to maintaining homeostasis [ref. 29,ref. 30,ref. 31].
3.1. Receptor Expression
Many families of growth factors and their receptors are expressed in the corneal epithelium, including the platelet-derived growth factor receptor family (PDGFR), vascular endothelial growth receptor factor family (VEGFR), epidermal growth receptor factor family (EGFR), fibroblast growth factor receptor family (FGFR), insulin-like growth factor receptor family (IGFR), and hepatocyte growth factor receptor family (HGFR, or c-Met) (Table 1). When stimulated in the corneal epithelium, these receptors promote cell proliferation, migration, and differentiation to aid in corneal re-epithelialization.
Table 1: Growth factor receptor biology and negative regulators in the corneal epithelium.
| Receptor | Factor | Effects on Corneal Wound Healing | Regulation by CBL |
|---|---|---|---|
| c-Met | Hepatocyte Growth Factor (HGF) | ||
| EGFR | Epidermal Growth Factor (EGF) | ||
| Heparin binding-EGF (HB-EGF) | |||
| Betacellulin (BTC) | |||
| Transforming Growth Factor-α (TGF-α) | |||
| KGFR | Keratinocyte Growth Factor (KGF) | ||
| FGFR | Fibroblast Growth Factor (FGF) | ||
| IGF-1R; IGF-2R | Insulin Growth Factor (IGF)Insulin | ||
| PDGF-αR & PDGF-βR | Platelet Derived Growth Factor (PDGF) | ||
| VEGFR 2 | Vascular Endothelial Growth Factor (VEGF) | ||
| TrkA | Nerve Growth Factor (NGF) | ||
| TrkB | Brain derived neurotrophic factor (BDNF) | ||
| TGF-βR | Transforming Growth Factor-β (TGF-β1,2,3) | ||
| RET/GFR-α | Glial cell line-derivedneurotrophicfactor (GDNF) |
3.2. Growth Factor Expression
The growth factors that stimulate these receptors can originate from the tear fluid, the corneal nerves, and the epithelial cells themselves. They can also be synthesized in fibroblast-like cells (i.e., keratocytes), which secrete them to act in a paracrine manner on the epithelial cells nearby (Figure 2). Many growth factors are expressed in basal tears, but their levels are dynamic based on the stresses to the eye.
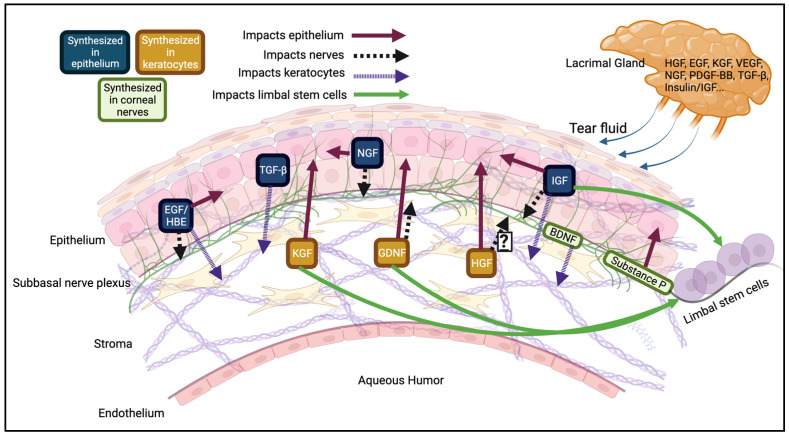
In mice, the wounding of the cornea increases growth factor presence in the aqueous phase of tear fluid. This delivers a higher concentration to the epithelium, promoting cellular proliferation, migration, and differentiation to promote re-epithelialization [ref. 32,ref. 33,ref. 34]. For example, human tear hepatocyte growth factor (HGF) mRNA levels increase ~1400% from the pre- to post- photorefractive or phototherapeutic keratectomy state [ref. 32]. The release of growth and neurotrophic factors in the tear fluid in vivo are, in part, controlled by the corneal nerves [ref. 35,ref. 36]. Nerves release neurotrophic factors/neuromodulators [i.e., substance P, calcitonin gene-related peptide (CGRP), acetylcholine, and vasoactive intestinal peptide (VIP)] that maintain the ocular surface [ref. 36]. Substance P and CGRP specifically have been shown to aid in corneal epithelial homeostasis and turnover [ref. 36].
Endogenous growth factors can be synthesized in a pro-form and associate with the cell membrane until they are processed by matrix metalloproteinases (MMPs) or a disintegrin and metalloproteases (ADAMs) [ref. 37,ref. 38]. The released soluble form is then able to bind and activate its cognate receptor. In in vitro corneal epithelial cell scratch wounds, MMP activity is increased, allowing for greater HB-EGF (heparin-binding EGF) processing and cleavage, resulting in the activation of the EGFR [ref. 39,ref. 40,ref. 41,ref. 42].
3.3. Receptor Mechanism of Action
Growth factor receptors have comparable mechanisms of action across the different families. Most growth factor receptors are embedded in the cell membrane in a monomeric form (others, like the insulin receptor, are already present in a dimerized form [ref. 43]).
Upon ligand binding, they undergo a conformational change and dimerize, and the intracellular kinase domains are brought close enough together to auto/trans-phosphorylate each other. Some receptors have ligand-stimulated tyrosine kinase activity [receptor tyrosine kinases (RTKs), i.e., c-Met and EGFR] and others have ligand-stimulated serine-threonine kinase activity [i.e., transforming growth factor-β (TGF-β) receptor]. Receptor phosphorylation is the unifying feature that allows for the docking and activation of downstream effectors [i.e., mitogen activated protein kinase (MAPK), phosphoinositide 3-kinase (PI3-K), signal transducer and activator of transcription 3 (STAT3), etc.]. Effectors translate the binding of extracellular ligands into intracellular biochemical signaling. The cell response is determined by how long individual effectors are active [ref. 116]. The contributions from each effector is governed by the spatial and temporal regulation of the receptor [ref. 116].
Alternative Signaling Mechanisms
Growth factor receptors interact with many cell surface molecules including mucins, plexins, integrins, other receptor tyrosine kinases (RTKs), and transient receptor potential (TRP) channels. Of these, TRP channels are particularly important regulators of the corneal epithelium, partly due to their expression in intraepithelial corneal nerve endings. In addition, TRP channels are present on the epithelial surface [ref. 117]. While they have their own roles in corneal homeostasis, the activation of these channels can lead to the stimulation of growth factor receptors: the treatment of human corneal epithelial cells (HCECs) with capsaicin, a potent agonist of the TRP vanilloid 1 (TRPV1) channel, induces the shedding of HB-EGF and activation of the EGFR [ref. 118,ref. 119]. There is evidence in other cell types (liver cancer cell lines, human prostate cancer lines, renal tubular cell lines) in which treatment with HGF increases the expression and activity of TRPC6, which contributes to cell proliferation [ref. 120]. Further, TRPV1′s Ca2+ influx channel activity is necessary for HGF-induced migration in HepG2 cells [ref. 121], so TRPV1 activation may be required in corneal epithelial cell motility as well.
3.4. Growth Factor Alterations in Disease States
The importance of growth factors can be seen by their varying levels in pathological conditions. Tear dysfunction—pathologies of tear fluid composition and make-up—is a leading cause of corneal epithelial disease and irritation [ref. 122]. The 2017 Dry Eye Workshop II (DEWS II) within the Tear Film and Ocular Surface society (TFOS) defined different types of Dry Eye Disease (DED). Within each of these subcategories (i.e., Sjögrens syndrome, aqueous deficient dry eye, meibomian gland dysfunction) the EGF concentration in the tears differs [ref. 123,ref. 124]. It remains to be determined whether changes in EGF levels are a cause or an effect of DED. DED also presents with upregulated MMPs [ref. 125,ref. 126], reflecting the eye’s need for more growth factor. The MMP inhibitor GM 6001 prevents wound-dependent EGFR activation, presenting as a delay in healing time, consistent with a block in the processing of HB-EGF [ref. 127].
Many disorders, including neurotrophic keratitis (NK), DED, and diabetes present with decreased corneal innervation. Less sensitivity to external stimuli and/or decreased connectivity with the lacrimal gland manifests as lowered tear production and secretion. This in turn decreases growth factor delivery to the cornea and manifests as delayed corneal wound healing [ref. 36,ref. 128,ref. 129,ref. 130]. Neuromodulators like substance P, CGRP, and NGF are downregulated in DED [ref. 131]. However, research is still necessary to determine if the lack of growth factor is causing the pathology or if the disease is causing the lack of growth factor.
There is ample literature discussing the changes in growth factors [ref. 132] in other corneal disorders like keratoconus [ref. 133], diabetes [ref. 133,ref. 134], bullous keratopathy [ref. 133], ocular rosacea [ref. 135], inflammatory surface diseases [ref. 136], and pseudomonas aeruginosa keratitis [ref. 137,ref. 138]. There are also genetic dimorphisms that impact corneal health. Many corneal dystrophies are manifested through single nucleotide polymorphisms (SNPs) in growth factor genes like TGF-β [ref. 139] and HGF [ref. 140] (though this has only been seen in certain demographics and not others [ref. 141,ref. 142]).
4. Growth Factor-Mediated Corneal Epithelial Restoration
4.1. The Role of Growth Factors for Corneal Epithelial Homeostasis and Restoration
The first evidence of growth factors playing a role in ocular biology came with the discovery of epidermal growth factor (EGF) in 1962 by Stanley Cohen [ref. 143]. His seminal discovery showed that addition of EGF accelerated the eyelid opening in newborn mice. Thirty years later, work by Zieske et al. demonstrated that introduction of EGF to debrided mouse corneas accelerated the rate of wound healing [ref. 144].
Other early studies demonstrated that FGF1 mRNA is upregulated 6-48h following corneal epithelial burning with a steady return to basal over 6 days [ref. 145]. This transient increase in ligand coupled with the presence of receptors on the cell surface point towards the use of exogenous growth factors as a natural way to enhance restoration of the epithelial surface.
4.2. Opportunities for the Use of Growth Factors
Due to the rapid turnover of the corneal epithelium, superficial scratches heal within 24–72 h in healthy individuals [ref. 146]. However, individuals who have diseases like diabetes [ref. 147,ref. 148,ref. 149] or are taking RTK inhibitors as anti-cancer therapy [ref. 150,ref. 151,ref. 152] often present with recurrent corneal erosions, leading to discomfort and potential loss of vision. Additionally, those who undergo corneal transplants or LASIK surgeries stand to benefit from pharmacologic agents that could help accelerate the healing process, alleviate pain associated with damaged corneal epithelium, aid in nerve regeneration, and prevent possible infection. Such agents also have the potential to help individuals undergoing limbal stem cell transplants by accelerating the restoration of the epithelial layer. Finally, compounds that accelerate the EGFR-mediated responses that promote corneal epithelial homeostasis will further our understanding of corneal epithelial biology as well as help in the development of the epithelial layers of artificial, bioengineered corneas.
4.2.1. Current FDA-Approved Growth Factor Therapies: Amniotic Membrane
Currently, there are limited options available to accelerate corneal epithelial wound healing. Amniotic membrane (AM) has a long history in tissue restoration including in regeneration of skin [ref. 153,ref. 154,ref. 155], in the dental clinic [ref. 156], as well as in ocular surface healing [ref. 157]. AM also has potent anti-inflammatory effects [ref. 158,ref. 159] and is used to prevent infection following surgery or burns [ref. 160,ref. 161]. AM is derived from placental tissue and consists of a single layer of epithelial cells, a basement membrane, an avascular matrix of connective tissue, and many pro-regenerative biomolecules, including EGF, HGF, FGF, and multiple cytokines [ref. 162,ref. 163].
The first use of AM on the ocular surface Is credited to de Rötth in 1940 [ref. 164], and since then, it has been used extensively in research and in the clinic. However, AM is limited in comfortability for the patient, how it is preserved, the variability of sources that it comes from, the types of corneal wounds that it can aid, and it does not address the underlying pathology [ref. 165,ref. 166,ref. 167,ref. 168]. However, it is still used in the clinic and is efficacious for healing some wounds [ref. 169], though the effects of the growth factors contained in AM have not been well-tested. Another promising option that has yet to be FDA-approved is the use of hydro- or collagenous gels that slowly release growth factors over time [ref. 170,ref. 171], much like AM. Further research is necessary to optimize these products for broader clinical use.
4.2.2. Current FDA-Approved Growth Factor Therapies: Recombinant Growth Factors
Corneal epithelial wounding insults both the epithelial cells and the corneal nerves. There is ample evidence that corneal nerves can regenerate after injury [ref. 111,ref. 128,ref. 172,ref. 173]. Part of this ability is due to epithelial cells and keratocytes releasing growth factors that act on corneal nerves including EGF, NGF, BDNF, and GDNF [ref. 99]. Topical treatment with growth factors like NGF and vascular endothelial growth factor (VEGF) can aid in corneal nerve regeneration and corneal healing [ref. 91,ref. 173]. Treatment to mouse corneal wounds with a dopamine receptor (D1 and D2) agonist increased the levels of NGF present in the cornea and promoted both nerve regeneration and re-epithelialization [ref. 174]. Further, combination therapy of IGF-1 and a substance P derivative, FGLM amide (phenylalanine-glycine-leucine-methionine amide), can aid in the healing of persistent corneal epithelial defects in patients with NK [ref. 84,ref. 175].
The first FDA-approved topical biologic medication in ophthalmology is Cenegermin/Oxervate®, a recombinant human NGF protein, specifically for NK [ref. 173,ref. 176,ref. 177], which is fitting as NGF was the first discovered growth factor in 1952 by Rita Levi-Montalcini and Stanley Cohen [ref. 178]. Cenegermin resolves roughly 72% of NK cases [ref. 179], but its use in other types of ocular perturbations (i.e., ulcerations, keratitis, surface wounds) is not well studied. Further, the use of this drug can present as a challenge to some patients, as it can be costly, difficult to prepare for self-administration, and has an intensive dosing regimen [ref. 180,ref. 181]. Additionally, there are often issues with patient compliance, as eye drops can cause pain, burning, and blurry vision after use [ref. 180].
There are other FDA-approved growth factor-based treatments including Regranex, which is a recombinant PDGF-BB ointment approved for healing diabetic ulcers. However, it is currently not recommended to use on any other types of wounds due to lack of clinical data and testing [ref. 182]. There are also some topical recombinant EGF formulations approved in countries outside of the USA for dermal applications [ref. 183,ref. 184,ref. 185] which may be useful for ocular surface wounds.
5. Emerging Opportunities
Given the available therapies, why would we need to continue the search? Many of these formulations are made up for a particular subset of patients—like Cenegermin which mainly aids in NK. But, as stated previously, it only aids in about 72% of patients. Further therapies are required to help in preventing corneal blindness due to imbalances in corneal epithelial homeostasis.
5.1. EGF: The Catalyst of Corneal Homeostasis Studies
A number of other growth factors have been considered to promote corneal re-epithelialization. Chief among these is topical EGF. Numerous corneal epithelial debridement models show that EGFR activity is both necessary (EGFR inhibitors prevent restoration) and sufficient (treatment with EGF accelerates restoration) for re-epithelialization [ref. 58,ref. 186]. However, in the clinic, the efficacy of topical EGF on corneal epithelial damage is dependent on the type of the wound. It has shown promise with traumatic ulcers [ref. 187,ref. 188], surface abrasions/lesions [ref. 59], and chemotherapy-induced erosions [ref. 151,ref. 189]; however, it has limited therapeutic benefit to patients with HSV-derived ulcers, bullous keratopathies, or penetrating keratoplasty [ref. 59,ref. 190,ref. 191].
It has been suggested that the sustained release of EGF is required for optimal EGFR signaling in the corneal epithelium [ref. 192]. This is likely due to “receptor desensitization” [ref. 193,ref. 194] which describes the peak in receptor activity (phosphorylation) following ligand stimulation that attenuates with time. This phenomenon of desensitization has been clearly described both in vitro and in vivo [ref. 195,ref. 196,ref. 197]. Molecular mechanisms include the dephosphorylation of the receptor by phosphatases, internalization of the receptor from the plasma membrane, and post-translational modifications (i.e., ubiquitin) that target the receptor for degradation. Receptor desensitization is a common feature of many receptors that are involved in corneal re-epithelialization and listed in Table 1 [ref. 198]. Under normal physiologic conditions, cellular mechanisms like receptor desensitization are critical for maintaining tissue homeostasis and preventing corneal hyperplasia [ref. 56,ref. 199]. However, under wounded conditions, these mechanisms of receptor inactivation slow the restoration process.
5.2. CBL-Mediated Desensitization of RTKs
Many RTKs are negatively regulated by the universal process of receptor ubiquitylation. Ubiquitylation limits receptor signaling, thus it is possible that their contribution to corneal epithelial homeostasis and wound healing is not fully recognized due to desensitization.
Antagonizing receptor ubiquitylation has emerged as a viable method for preventing receptor desensitization, thus sustaining receptor signaling. Ubiquitylation is involved in the endocytic trafficking of activated receptors. Ubiquitylated receptors bind to the endosomal sorting complexes required for transport (ESCRT) machinery on the limiting membrane of the late endosome and internalize into the intraluminal vesicles (ILV) of the late endosome. These late endosomes fuse with the lysosome and transfer the ILVs and its receptor cargo for degradation (Figure 3) [ref. 200].
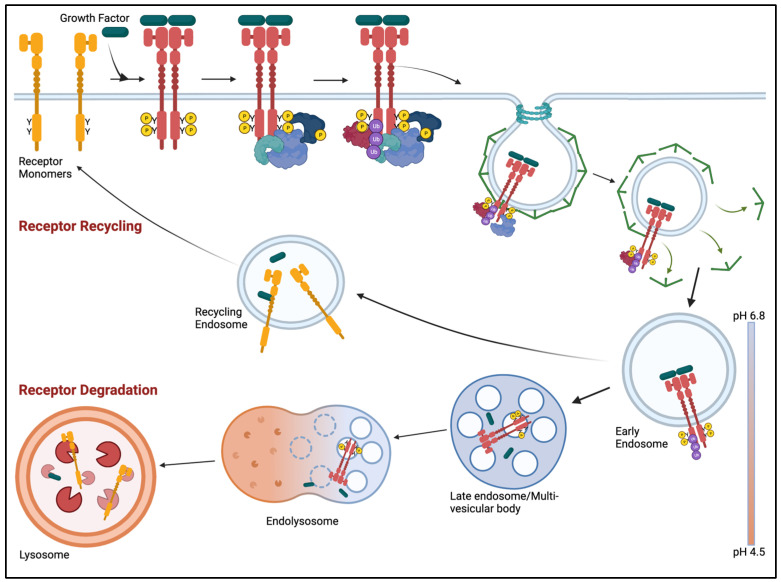
The ubiquitylation proteasome system (UPS) selectively regulates the balance of proteins in the cell. Proteins are conjugated with the 76-amino acid protein ubiquitin (Ub) via a series of enzymatic reactions involving the E1, E2, and E3 Ub ligase machinery that targets proteins for proteasomal or lysosomal degradation [ref. 201]. There are two E1 activating enzymes that transfer Ub to one of 38 E2-conjugating enzymes. There are several hundred E3 ligases that mediate the final step of Ub transfer to its specific target. E3 ligases are good pharmacologic targets because only liganded receptors are phosphorylated and have the requisite phosphotyrosine for ubiquitin transfer [ref. 45].
Protein ubiquitylation is counter-regulated by deubiquitylating enzymes (DUBs). Several studies highlight the role of E3 ligases in growth factor receptor desensitization. Two of the primary E3 ligases that regulate many RTKs in the corneal epithelium are c-Cbl and Cbl-b. These E3 ligases have roles in other facets of membrane trafficking (i.e., autophagy), which are independent of its E3 ligase activity and have only been observed in cancer cells [ref. 202]. The major role of c-Cbl and Cbl-b is downregulation of cell surface receptors.
Evidence to support ubiquitylation as a target comes from studies with the EGFR. The knockdown and/or knockout of c-Cbl decreased EGFR ubiquitylation and increased the rate of corneal epithelial in vitro wound healing [ref. 58,ref. 65]. Indirect pharmacologic inhibition of ubiquitylation via PP1 (Src inhibitor) resulted in faster in vitro and in vivo corneal re-epithelialization [ref. 58]. As indicated in Table 1, multiple receptors are regulated by CBL proteins. Antagonizing CBL activity may be a more universal approach to sustain receptor signaling in the corneal epithelium.
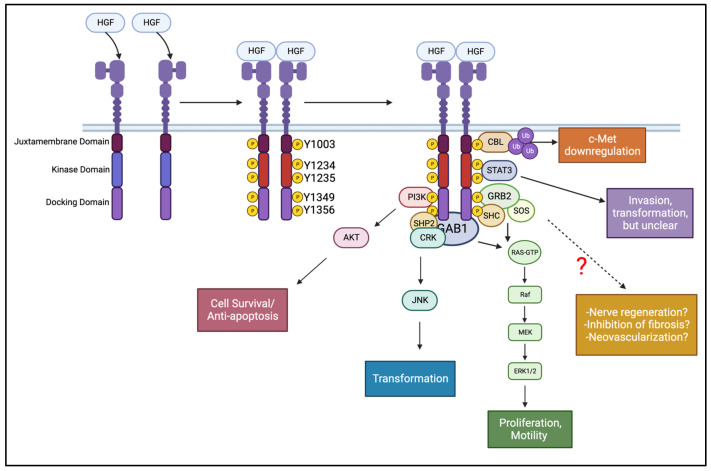
An additional example is the c-Met receptor. For instance, activation of c-Met by HGF promotes corneal epithelial restoration [ref. 44,ref. 45], but treatment with HGF at supraphysiologic concentrations can limit the therapeutic benefit of the growth factor [ref. 207]. Corneal epithelial cells deficient in c-Cbl and Cbl-b demonstrated slowed HGF-driven c-Met trafficking, which resulted in enhanced receptor and effector signaling. The greater magnitude and duration of c-Met phosphorylation in these knockout cells potentiated in vitro wound healing rates 2-fold [ref. 45].
5.3. HGF: The Multi-Faceted Growth Factor
Although re-epithelialization is the critical first step in corneal wound healing, other aspects such as chronic inflammation and nerve regeneration need to be considered. Additional roles for growth factors receptors include mitogenic [ref. 44], anti-fibrotic [ref. 47], anti-angiogenic [ref. 54], and neurotrophic [ref. 208] effects. While most growth factors and their receptors can aid in re-epithelialization, the unique features of the HGF:c-Met signaling axis fulfills many roles within the healing process.
5.3.1. Inhibition of Fibrosis
Inflammation, in the early stages of healing, is beneficial. Damaged epithelial cells continuously release growth factors like TGF-β and PDGF into the stroma [ref. 34,ref. 61,ref. 209]. When TGF-β and other cytokines are present in high concentrations in the stroma, they differentiate keratocytes into corneal fibroblasts and then to mature myofibroblasts [ref. 77,ref. 210,ref. 211,ref. 212,ref. 213,ref. 214,ref. 215]. Mature myofibroblasts are opaque and in chronic wound healing scenarios, they persistently release high levels of disordered extracellular matrix. This clouds the stromal layer of the cornea and impairs vision [ref. 77,ref. 216,ref. 217]. The stromal response to injury will not fully terminate unless the epithelial basement membrane reforms, so the healing of the epithelial layer is crucial for full ocular restoration.
The balance between anti-inflammatory (i.e., HGF and EGF) and pro-inflammatory (TGF-β) signaling mediators determine the extent of tissue damage. TGF-β levels rise as injuries become chronic, leading to fibrosis [ref. 218,ref. 219]. HGF can inhibit TGF-β production by upregulating Smad7 [ref. 47,ref. 220,ref. 221], which in turn prevents myofibroblasts from maturing [ref. 77,ref. 222,ref. 223]. Activation of the EGFR can promote nuclear factor κβ (NF-κβ) activity, which inhibits TGF-β signaling [ref. 224]. Further, HGF can suppress pro-inflammatory cytokines IL-1, IL-6, and IL-18 that are released from macrophages [ref. 225]. Lastly, combination gene therapy with HGF and bone morphogenic protein 7 (BMP7) decreased corneal fibrosis following in vivo rabbit corneal alkali burn [ref. 226,ref. 227].
5.3.2. Corneal Neovascularization
Another clinical complication in corneal wound healing is neo-vascularization. The absence of blood and lymphatic vessels in the cornea keeps it transparent and allows light to pass through and refract on the retina. Under normal conditions, the protein Notch1 suppresses VEGF expression in the cornea. When Notch1 activity decreases or when VEGFR is hyperactivated, neovascularization branches from the blood vessels of the limbal vascular plexus [ref. 235]. The downregulation of TGF-β in an in vivo burn model decreases not only infiltrating inflammatory cells and disordered ECM, but also the formation of new vessels [ref. 236].
HGF has been implicated as a regulator of angiogenesis, but there is no unifying model. In some tissues, HGF is a potent angiogenic factor, particularly in the retina [ref. 237,ref. 238,ref. 239]. For the anterior eye, some studies indicate that the inhibition of HGF via siRNA prevents VEGF-dependent corneal neovascularization, but this was accompanied by decreased epithelial proliferation and increased incidence of apoptosis [ref. 240]. Conversely, implantation of pellets into the mouse cornea that contain an HGF derivative, H-RN, also prevented VEGF-driven angiogenesis [ref. 52,ref. 55]. Together, these findings suggest a role for HGF in regulating corneal neovascularization, but further investigation is needed.
5.3.3. Neuro-Regeneration
Restoring the corneal epithelium provides a critical barrier to foreign agents and limits the chances of infection. However, without neuronal restoration, the eye lacks the necessary sensitivity it needs to avoid recurrent erosions and surface shedding.
c-Met has roles in in vivo [ref. 49], ex vivo [ref. 241], and in vitro [ref. 51,ref. 242] neuronal growth models. In aging mice, decreases in c-Met activation parallel the loss of nerve regeneration [ref. 50]. c-Met has also been implicated in the formation of new synapses [ref. 48]. However, there is no literature surrounding c-Met and HGF involvement in intraepithelial corneal nerve communication and wound healing, which is a gap in the field that needs to be addressed.
6. Conclusions
Damage to the corneal epithelium is a component of almost all corneal injuries. In addition to intense pain, perturbation to the epithelial layer makes the eye susceptible to infection and potentially loss of vision. Growth factor receptors have a central role in corneal epithelial homeostasis and regeneration, however, due to our incomplete understanding of their signaling, they have not yet reached their full potential as a therapy. Critical next steps include identifying the most efficacious mediators of corneal regeneration and, perhaps most importantly, identifying limitations to their use. Uncovering the fundamental mechanisms of how these proteins work will serve as a foundation for developing new therapies to treat the millions of individuals affected each year by corneal perturbations.
References
- P. Scott. Scott’s Anatomy of the Eye and Orbit, 2019
- L.J. Müller, C.F. Marfurt, F. Kruse, T.M.T. Tervo. Corneal nerves: Structure, contents and function. Exp. Eye Res., 2003. [DOI | PubMed]
- S. Koh, S.K. Rao, S.P. Srinivas, L. Tong, A.L. Young. Evaluation of ocular surface and tear function—A review of current approaches for dry eye. Indian J. Ophthalmol., 2022. [DOI | PubMed]
- B. Klenkler, H. Sheardown, L. Jones. Growth factors in the tear film: Role in tissue maintenance, wound healing, and ocular pathology. Ocul. Surf., 2007. [DOI | PubMed]
- J.A. Bonanno. Molecular mechanisms underlying the corneal endothelial pump. Exp. Eye Res., 2012. [DOI | PubMed]
- R.A. Thoft, J. Friend. The X, Y, Z hypothesis of corneal epithelial maintenance. Investig. Ophthalmol. Vis. Sci., 1983. [PubMed]
- C. Hanna, D.S. Bicknell, J.E. O’Brien. Cell Turnover in the Adult Human Eye. Arch. Ophthalmol., 1961. [DOI | PubMed]
- A. Haddad. Renewal of the rabbit corneal epithelium as investigated by autoradiography after intravitreal injection of 3H-thymidine. Cornea, 2000. [DOI | PubMed]
- R.A. Thoft, L.A. Wiley, N. Sundarraj. The multipotential cells of the limbus. Eye, 1989. [DOI | PubMed]
- P.E. Ludwig, M.J. Lopez, K.E. Sevensma. Anatomy, Head and Neck, Eye Cornea. StatPearls, 2023
- M. Yazdani. Tear film lipid layer and corneal oxygenation: A new function?. Eye, 2023. [DOI | PubMed]
- R.W. Beuerman, L. Pedroza. Ultrastructure of the human cornea. Microsc. Res. Tech., 1996. [DOI | PubMed]
- P.J. Driver, M.A. Lemp. Meibomian gland dysfunction. Surv. Ophthalmol., 1996. [DOI | PubMed]
- R.A. Sack, L. Conradi, D. Krumholz, A. Beaton, S. Sathe, C. Morris. Membrane array characterization of 80 chemokines, cytokines, and growth factors in open- and closed-eye tears: Angiogenin and other defense system constituents. Investig. Ophthalmol. Vis. Sci., 2005. [DOI | PubMed]
- J.M. Tiffany. Tears in health and disease. Eye, 2003. [DOI | PubMed]
- M. Rolando, M. Zierhut. The ocular surface and tear film and their dysfunction in dry eye disease. Surv. Ophthalmol., 2001. [DOI | PubMed]
- A. Rózsa, R.W. Beuerman. Density and Organization of Free Nerve Endings in the Corneal Epithelium of the Rabbit. Pain, 1982. [DOI | PubMed]
- C.F. Marfurt, R.E. Kingsley, S.E. Echtenkamp. Sensory and sympathetic innervation of the mammalian cornea. A retrograde tracing study. Investig. Ophthalmol. Vis. Sci., 1989. [PubMed]
- C.F. Marfurt, M.A. Jones, K. Thrasher. Parasympathetic Innervation of the Rat Cornea. Exp. Eye Res., 1998. [DOI | PubMed]
- C.F. Marfurt. Sympathetic innervation of the rat cornea as demonstrated by the retrograde and anterograde transport of horseradish peroxidase–wheat germ agglutinin. J. Comp. Neurol., 1988. [DOI | PubMed]
- C.F. Marfurt. Corneal Nerves: Anatomy. Encyclopedia of the Eye, 2010
- D.A. Dartt. Neural regulation of lacrimal gland secretory processes: Relevance in dry eye diseases. Prog. Retin. Eye Res., 2009. [DOI | PubMed]
- M. Labetoulle, C. Baudouin, M. Calonge, J. Merayo-Lloves, K.G. Boboridis, Y.A. Akova, P. Aragona, G. Geerling, E.M. Messmer, J. Benitez-Del-Castillo. Role of corneal nerves in ocular surface homeostasis and disease. Acta Ophthalmol., 2019. [DOI | PubMed]
- M.A. Stepp, G. Tadvalkar, R. Hakh, S. Pal-Ghosh. Corneal epithelial cells function as surrogate Schwann cells for their sensory nerves. Glia, 2017. [DOI | PubMed]
- P. Bargagna-Mohan, G. Schultz, B. Rheaume, E.F. Trakhtenberg, P. Robson, S. Pal-Ghosh, M.A. Stepp, K.S. Given, W.B. Macklin, R. Mohan. Corneal nonmyelinating Schwann cells illuminated by single-cell transcriptomics and visualized by protein biomarkers. J. Neurosci. Res., 2021. [DOI | PubMed]
- K. Mirmoeini, K. Tajdaran, J. Zhang, T. Gordon, A. Ali, D.R. Kaplan, K. Feinberg, G.H. Borschel. Schwann Cells Are Key Regulators of Corneal Epithelial Renewal. Investig. Ophthalmol. Vis. Sci., 2023. [DOI | PubMed]
- K. Feinberg, K. Tajdaran, K. Mirmoeini, S.C. Daeschler, M.A. Henriquez, K.E. Stevens, C.M. Mulenga, A. Hussain, P. Hamrah, A. Ali. The Role of Sensory Innervation in Homeostatic and Injury-Induced Corneal Epithelial Renewal. Int. J. Mol. Sci., 2023. [DOI | PubMed]
- B.S. Kowtharapu, T. Stahnke, A. Wree, R.F. Guthoff, O. Stachs. Corneal epithelial and neuronal interactions: Role in wound healing. Exp. Eye Res., 2014. [DOI | PubMed]
- F.S. Yu, J. Yin, K. Xu, J. Huang. Growth factors and corneal epithelial wound healing. Brain Res. Bull., 2010. [DOI | PubMed]
- A. Bukowiecki, D. Hos, C. Cursiefen, S.A. Eming. Wound-Healing Studies in Cornea and Skin: Parallels, Differences and Opportunities. Int. J. Mol. Sci., 2017. [DOI | PubMed]
- J. Imanishi, K. Kamiyama, I. Iguchi, M. Kita, C. Sotozono, S. Kinoshita. Growth factors: Importance in wound healing and maintenance of transparency of the cornea. Prog. Retin. Eye Res., 2000. [DOI | PubMed]
- T. Tervo, M. Vesaluoma, G.L. Bennett, R. Schwall, M. Helena, Q. Liang, S.E. Wilson. Tear hepatocyte growth factor (HGF) availability increases markedly after excimer laser surface ablation. Exp. Eye Res., 1997. [DOI | PubMed]
- S.E. Wilson, Q. Liang, W.J. Kim. Lacrimal gland HGF, KGF, and EGF mRNA levels increase after corneal epithelial wounding. Investig. Ophthalmol. Vis. Sci., 1999. [PubMed]
- S.E. Wilson, S.A. Lloyd. Epidermal growth factor and its receptor, basic fibroblast growth factor, transforming growth factor beta-1, and interleukin-1 alpha messenger RNA production in human corneal endothelial cells. Investig. Ophthalmol. Vis. Sci., 1991. [PubMed]
- B.S. Shaheen, M. Bakir, S. Jain. Corneal nerves in health and disease. Surv. Ophthalmol., 2014. [DOI | PubMed]
- L. Mastropasqua, G. Massaro-Giordano, M. Nubile, M. Sacchetti. Understanding the Pathogenesis of Neurotrophic Keratitis: The Role of Corneal Nerves. J. Cell. Physiol., 2017. [DOI | PubMed]
- J.H. Chang, Y.H. Huang, C.M. Cunningham, K.Y. Han, M. Chang, M. Seiki, Z. Zhou, D.T. Azar. Matrix metalloproteinase 14 modulates signal transduction and angiogenesis in the cornea. Surv. Ophthalmol., 2016. [DOI | PubMed]
- O. Zbodakova, K. Chalupsky, L. Sarnova, P. Kasparek, M. Jirouskova, M. Gregor, R. Sedlacek. ADAM10 and ADAM17 regulate EGFR, c-Met and TNF RI signalling in liver regeneration and fibrosis. Sci. Rep., 2021. [DOI | PubMed]
- J. Yin, X.Y. Fu-Shin. ERK1/2 mediate wounding-and G-protein-coupled receptor ligands-induced EGFR activation via regulating ADAM17 and HB-EGF shedding. Investig. Ophthalmol. Vis. Sci., 2009. [DOI | PubMed]
- B. Mulholland, S.J. Tuft, P.T. Khaw. Matrix metalloproteinase distribution during early corneal wound healing. Eye, 2005. [DOI | PubMed]
- K.-P. Xu, Y. Ding, J. Ling, Z. Dong, F.-S.X. Yu. Wound-Induced HB-EGF Ectodomain Shedding and EGFR Activation in Corneal Epithelial Cells. Investig. Ophthalmol. Vis. Sci., 2004. [DOI | PubMed]
- J.A. Breyer, S. Cohen. The epidermal growth factor precursor isolated from murine kidney membranes. Chemical characterization and biological properties. J. Biol. Chem., 1990. [DOI | PubMed]
- T. Gutmann, K.H. Kim, M. Grzybek, T. Walz, Ü. Coskun. Visualization of ligand-induced transmembrane signaling in the full-length human insulin receptor. J. Cell Biol., 2018. [DOI | PubMed]
- M. Omoto, K. Suri, A. Amouzegar, M. Li, K.R. Katikireddy, S.K. Mittal, S.K. Chauhan. Hepatocyte Growth Factor Suppresses Inflammation and Promotes Epithelium Repair in Corneal Injury. Mol. Ther., 2017. [DOI | PubMed]
- K. Tarvestad-Laise, B.P. Ceresa. Knockout of c-Cbl/Cbl-b slows c-Met trafficking resulting in enhanced signaling in corneal epithelial cells. J. Biol. Chem., 2023. [DOI | PubMed]
- M. Takahashi, S. Ota, T. Shimada, E. Hamada, T. Kawabe, T. Okudaira, M. Matsumura, N. Kaneko, A. Terano, T. Nakamura. Hepatocyte growth factor is the most potent endogenous stimulant of rabbit gastric epithelial cell proliferation and migration in primary culture. J. Clin. Investig., 1995. [DOI | PubMed]
- M.N. Shukla, J.L. Rose, R. Ray, K.L. Lathrop, A. Ray, P. Ray. Hepatocyte growth factor inhibits epithelial to myofibroblast transition in lung cells via Smad7. Am. J. Respir. Cell Mol. Biol., 2009. [DOI | PubMed]
- Z. Xie, K.L. Eagleson, H.H. Wu, P. Levitt. Hepatocyte Growth Factor Modulates MET Receptor Tyrosine Kinase and beta-Catenin Functional Interactions to Enhance Synapse Formation. eNeuro, 2016. [DOI | PubMed]
- N. Hashimoto, H. Yamanaka, T. Fukuoka, Y. Dai, K. Obata, T. Mashimo, K. Noguchi. Expression of HGF and cMet in the peripheral nervous system of adult rats following sciatic nerve injury. Clin. Neurosci. Neuropathol., 2001. [DOI]
- N. Lee, S. Kim, J. Lee. Impairment of peripheral nerve regeneration by insufficient activation of the HGF/c-Met/c-Jun pathway in aged mice. Heliyon, 2022. [DOI | PubMed]
- M. Hamanoue, N. Takemoto, K. Matsumoto, T. Nakamura, K. Nakajima, S. Kohsaka. Neurotrophic effect of hepatocyte growth factor on central nervous system neurons in vitro. J. Neurosci. Res., 1996. [DOI | PubMed]
- Y. Xu, H. Zhao, Y. Zheng, Q. Gu, J. Ma, X. Xu. A novel antiangiogenic peptide derived from hepatocyte growth factor inhibits neovascularization in vitro and in vivo. Mol. Vis., 2010. [PubMed]
- X. Xin, S. Yang, G. Ingle, C. Zlot, L. Rangell, J. Kowalski, R. Schwall, N. Ferrara, M.E. Gerritsen. Hepatocyte Growth Factor Enhances Vascular Endothelial Growth Factor-Induced Angiogenesis in Vitro and in Vivo. Am. J. Pathol., 2001. [DOI | PubMed]
- A.A. Ucuzian, A.A. Gassman, A.T. East, H.P. Greisler. Molecular mediators of angiogenesis. J. Burn Care Res., 2010. [DOI | PubMed]
- Y. Sun, L. Su, Z. Wang, Y. Xu, X. Xu. H-RN, a peptide derived from hepatocyte growth factor, inhibits corneal neovascularization by inducing endothelial apoptosis and arresting the cell cycle. BMC Cell Biol., 2013. [DOI | PubMed]
- P. Peschard, T.M. Fournier, L. Lamorte, M.A. Naujokas, H. Band, W.Y. Langdon, M. Park. Mutation of the c-Cbl TKB domain binding site on the Met receptor tyrosine kinase converts it into a transforming protein. Mol. Cell, 2001. [DOI | PubMed]
- P. Peschard, N. Ishiyama, T. Lin, S. Lipkowitz, M. Park. A conserved DpYR motif in the juxtamembrane domain of the Met receptor family forms an atypical c-Cbl/Cbl-b tyrosine kinase binding domain binding site required for suppression of oncogenic activation. J. Biol. Chem., 2004. [DOI | PubMed]
- J.S. Rush, M.A. Boeving, W.L. Berry, B.P. Ceresa. Antagonizing c-Cbl enhances EGFR-dependent corneal epithelial homeostasis. Investig. Ophthalmol. Vis. Sci., 2014. [DOI | PubMed]
- S. Daniele, L. Frati, C. Fiore, G. Santoni. The effect of the epidermal growth factor (EGF) on the corneal epithelium in humans. Albrecht Von Graefes Arch. Klin. Exp. Ophthalmol., 1979. [DOI | PubMed]
- H. Watanabe, Y. Ohashi, S. Kinoshita, R. Manabe, K. Ohshiden. Distribution of epidermal growth factor in rat ocular and periocular tissues. Graefe’s Arch. Clin. Exp. Ophthalmol., 1993. [DOI | PubMed]
- S.E. Wilson, Y.G. He, S.A. Lloyd. EGF, EGF receptor, basic FGF, TGF beta-1, and IL-1 alpha mRNA in human corneal epithelial cells and stromal fibroblasts. Investig. Ophthalmol. Vis. Sci., 1992. [PubMed]
- H.M. Boisjoly, C. Laplante, S.F. Bernatchez, C. Salesse, M. Giasson, M.-C. Joly. Effects of EGF, IL-1 and their combination on in vitro corneal epithelial wound closure and cell chemotaxis. Exp. Eye Res., 1993. [DOI | PubMed]
- C.R. Savage, S. Cohen. Proliferation of corneal epithelium induced by epidermal growth factor. Exp. Eye Res., 1973. [DOI | PubMed]
- W. Tao, G.I. Liou, X. Wu, T.O. Abney, P.S. Reinach. ETB and epidermal growth factor receptor stimulation of wound closure in bovine corneal epithelial cells. Investig. Ophthalmol. Vis. Sci., 1995. [PubMed]
- B.L.M. Crotchett, B.P. Ceresa. Knockout of c-Cbl slows EGFR endocytic trafficking and enhances EGFR signaling despite incompletely blocking receptor ubiquitylation. Pharmacol. Res. Perspect., 2021. [DOI | PubMed]
- I. Pinilla-Macua, A. Sorkin. Cbl and Cbl-b independently regulate EGFR through distinct receptor interaction modes. Mol. Biol. Cell, 2023. [DOI | PubMed]
- R. Tang, W.Y. Langdon, J. Zhang. Negative regulation of receptor tyrosine kinases by ubiquitination: Key roles of the Cbl family of E3 ubiquitin ligases. Front. Endocrinol., 2022. [DOI | PubMed]
- J.L. Peterson, E.D. Phelps, M.A. Doll, S. Schaal, B.P. Ceresa. The role of endogenous epidermal growth factor receptor ligands in mediating corneal epithelial homeostasis. Investig. Ophthalmol. Vis. Sci., 2014. [DOI | PubMed]
- M.A. Tolino, E.R. Block, J.K. Klarlund. Brief treatment with heparin-binding EGF-like growth factor, but not with EGF, is sufficient to accelerate epithelial wound healing. Biochim. Biophys. Acta BBA Gen. Subj., 2011. [DOI]
- W.-Y. Jeong, H.-Y. Yoo, C.-W. Kim. β-cellulin promotes the proliferation of corneal epithelial stem cells through the phosphorylation of erk1/2. Biochem. Biophys. Res. Commun., 2018. [DOI | PubMed]
- J.L. McClintock, B.P. Ceresa. Transforming growth factor-{alpha} enhances corneal epithelial cell migration by promoting EGFR recycling. Investig. Ophthalmol. Vis. Sci., 2010. [DOI | PubMed]
- C. Sotozono, T. Inatomi, M. Nakamura, S. Kinoshita. Keratinocyte growth factor accelerates corneal epithelial wound healing in vivo. Investig. Ophthalmol. Vis. Sci., 1995. [PubMed]
- C. Sotozono, S. Kinoshita, M. Kita, J. Imanishi. Paracrine role of keratinocyte growth factor in rabbit corneal epithelial cell growth. Exp. Eye Res., 1994. [DOI | PubMed]
- G. Chandrasekher, A.H. Kakazu, H.E. Bazan. HGF- and KGF-induced activation of PI-3K/p70 s6 kinase pathway in corneal epithelial cells: Its relevance in wound healing. Exp. Eye Res., 2001. [DOI | PubMed]
- F. Belleudi, L. Leone, V. Nobili, S. Raffa, F. Francescangeli, M. Maggio, S. Morrone, C. Marchese, M.R. Torrisi. Keratinocyte Growth Factor Receptor Ligands Target the Receptor to Different Intracellular Pathways. Traffic, 2007. [DOI | PubMed]
- P. Rieck, M. Assouline, M. Savoldelli, C. Hartmann, C. Jacob, Y. Pouliquen, Y. Courtois. Recombinant human basic fibroblast growth factor (Rh-bFGF) in three different wound models in rabbits: Corneal wound healing effect and pharmacology. Exp. Eye Res., 1992. [DOI | PubMed]
- J.V. Jester, P.A. Barry-Lane, H.D. Cavanagh, W.M. Petroll. Induction of alpha-smooth muscle actin expression and myofibroblast transformation in cultured corneal keratocytes. Cornea, 1996. [DOI | PubMed]
- J.V. Jester, J. Huang, W.M. Petroll, H.D. Cavanagh. TGFβ induced myofibroblast differentiation of rabbit keratocytes requires synergistic TGFβ, PDGF and integrin signaling. Exp. Eye Res., 2002. [DOI | PubMed]
- S.E. Wilson, J.J. Liu, R.R. Mohan. Stromal-epithelial interactions in the cornea. Prog. Retin. Eye Res., 1999. [DOI | PubMed]
- Y. Shakiba, K. Mansouri, D. Arshadi, N. Rezaei. Corneal neovascularization: Molecular events and therapeutic options. Recent Pat. Inflamm. Allergy Drug Discov., 2009. [DOI | PubMed]
- A. Wong, B. Lamothe, A. Lee, J. Schlessinger, I. Lax. FRS2α attenuates FGF receptor signaling by Grb2-mediated recruitment of the ubiquitin ligase Cbl. Proc. Natl. Acad. Sci. USA, 2002. [DOI | PubMed]
- W.L. Stuard, R. Titone, D.M. Robertson. The IGF/Insulin-IGFBP Axis in Corneal Development, Wound Healing, and Disease. Front. Endocrinol., 2020. [DOI]
- P. Trosan, E. Svobodova, M. Chudickova, M. Krulova, A. Zajicova, V. Holan. The key role of insulin-like growth factor I in limbal stem cell differentiation and the corneal wound-healing process. Stem Cells Dev., 2012. [DOI | PubMed]
- M. Nakamura, T. Nagano, T.-i. Chikama, T. Nishida. Up-Regulation of Phosphorylation of Focal Adhesion Kinase and Paxillin by Combination of Substance P and IGF-1 in SV-40 Transformed Human Corneal Epithelial Cells. Biochem. Biophys. Res. Commun., 1998. [DOI | PubMed]
- L.J. Shanley, C.D. McCaig, J.V. Forrester, M. Zhao. Insulin, not leptin, promotes in vitro cell migration to heal monolayer wounds in human corneal epithelium. Investig. Ophthalmol. Vis. Sci., 2004. [DOI | PubMed]
- J. Lyu, K.-S. Lee, C.-K. Joo. Transactivation of EGFR mediates insulin-stimulated ERK1/2 activation and enhanced cell migration in human corneal epithelial cells. Mol. Vis., 2006. [PubMed]
- B. Sehat, S. Andersson, L. Girnita, O. Larsson. Identification of c-Cbl as a new ligase for insulin-like growth factor-I receptor with distinct roles from Mdm2 in receptor ubiquitination and endocytosis. Cancer Res., 2008. [DOI | PubMed]
- H. Li, L. Xu, C. Li, L. Zhao, Y. Ma, H. Zheng, Z. Li, Y. Zhang, R. Wang, Y. Liu. Ubiquitin ligase Cbl-b represses IGF-I-induced epithelial mesenchymal transition via ZEB2 and microRNA-200c regulation in gastric cancer cells. Mol. Cancer, 2014. [DOI | PubMed]
- K. Kamiyama, I. Iguchi, X. Wang, J. Imanishi. Effects of PDGF on the migration of rabbit corneal fibroblasts and epithelial cells. Cornea, 1998. [DOI | PubMed]
- C. Rorsman, M. Tsioumpekou, C.-H. Heldin, J. Lennartsson. The ubiquitin ligases c-Cbl and Cbl-b negatively regulate platelet-derived growth factor (PDGF) BB-induced chemotaxis by affecting PDGF receptor β (PDGFRβ) internalization and signaling. J. Biol. Chem., 2016. [DOI | PubMed]
- Z. Pan, S. Fukuoka, N. Karagianni, V.H. Guaiquil, M.I. Rosenblatt. Vascular endothelial growth factor promotes anatomical and functional recovery of injured peripheral nerves in the avascular cornea. FASEB J., 2013. [DOI | PubMed]
- Z. Li, A.R. Burns, L. Han, R.E. Rumbaut, C.W. Smith. IL-17 and VEGF Are Necessary for Efficient Corneal Nerve Regeneration. Am. J. Pathol., 2011. [DOI | PubMed]
- A.J. Singh, R.D. Meyer, G. Navruzbekov, R. Shelke, L. Duan, H. Band, S.E. Leeman, N. Rahimi. A critical role for the E3-ligase activity of c-Cbl in VEGFR-2-mediated PLCγ1 activation and angiogenesis. Proc. Natl. Acad. Sci. USA, 2007. [DOI | PubMed]
- T. Blanco-Mezquita, C. Martinez-Garcia, R. Proença, J.D. Zieske, S. Bonini, A. Lambiase, J. Merayo-Lloves. Nerve growth factor promotes corneal epithelial migration by enhancing expression of matrix metalloprotease-9. Investig. Ophthalmol. Vis. Sci., 2013. [DOI | PubMed]
- E.D. Fixman, T.M. Fournier, D.M. Kamikura, M.A. Naujokas, M. Park. Pathways Downstream of Shc and Grb2 Are Required for Cell Transformation by the Tpr-Met Oncoprotein. J. Biol. Chem., 1996. [DOI | PubMed]
- E. Pedrotti, E. Bonacci, C. Chierego, A. De Gregorio, T. Cozzini, T. Brighenti, G. Caldarella, G. Pastore, A. Fasolo, G. Marchini. Eight months follow-up of corneal nerves and sensitivity after treatment with cenegermin for neurotrophic keratopathy. Orphanet J. Rare Dis., 2022. [DOI | PubMed]
- Y. Takahashi, N. Shimokawa, S. Esmaeili-Mahani, A. Morita, H. Masuda, T. Iwasaki, J. Tamura, K. Haglund, N. Koibuchi. Ligand-induced downregulation of TrkA is partly regulated through ubiquitination by Cbl. FEBS Lett., 2011. [DOI | PubMed]
- H.-K. Chen, Y.-Z. Li, A.-N. Ge, Y.-B. Zhu, S.-J. Wu, X. Bai, H.-H. Bai, Y.-N. Liu. Cbl-b modulated TrkA ubiquitination and function in the dorsal root ganglion of mice. Eur. J. Pharmacol., 2022. [DOI | PubMed]
- L. You, F.E. Kruse, H.E. Völcker. Neurotrophic Factors in the Human Cornea. Investig. Ophthalmol. Vis. Sci., 2000. [PubMed]
- J.H. McCarty, S.C. Feinstein. The TrkB receptor tyrosine kinase regulates cellular proliferation via signal transduction pathways involving SHC, PLCgamma, and CBL. J. Recept. Signal Transduct. Res., 1999. [DOI | PubMed]
- C. Pandya, A. Kutiyanawalla, G. Turecki, A. Pillai. Glucocorticoid regulates TrkB protein levels via c-Cbl dependent ubiquitination: A decrease in c-Cbl mRNA in the prefrontal cortex of suicide subjects. Psychoneuroendocrinology, 2014. [DOI | PubMed]
- S. Pancholi, A. Tullo, A. Khaliq, D. Foreman, M. Boulton. The effects of growth factors and conditioned media on the proliferation of human corneal epithelial cells and keratocytes. Graefe’s Arch. Clin. Exp. Ophthalmol., 1998. [DOI | PubMed]
- M. Haber, Z. Cao, N. Panjwani, D. Bedenice, W.W. Li, P.J. Provost. Effects of growth factors (EGF, PDGF-BB and TGF-β1) on cultured equine epithelial cells and keratocytes: Implications for wound healing. Vet. Ophthalmol., 2003. [DOI | PubMed]
- H. Mishima, M. Nakamura, J. Murakami, T. Nishida, T. Otori. Transforming growth factor-β modulates effects of epidermal growth factor on corneal epithelial cells. Curr. Eye Res., 1992. [DOI | PubMed]
- Y. Honma, K. Nishida, C. Sotozono, S. Kinoshita. Effect of transforming growth factor-β1 and-β2 onin vitroRabbit corneal epithelial cell proliferation promoted by epidermal growth factor, keratinocyte growth factor, or hepatocyte growth factor. Exp. Eye Res., 1997. [DOI | PubMed]
- J.V. Jester, J. Ho-Chang. Modulation of cultured corneal keratocyte phenotype by growth factors/cytokines control in vitro contractility and extracellular matrix contraction. Exp. Eye Res., 2003. [DOI | PubMed]
- T. MøSller-Pedersen, H.D. Cavanagh, W.M. Petroll, J.V. Jester. Neutralizing antibody to TGFβ modulates stromal fibrosis but not regression of photoablative effect following PRK. Curr. Eye Res., 1998. [DOI | PubMed]
- D. Karamichos, A. Hutcheon, J. Zieske. Reversal of fibrosis by TGF-β3 in a 3D in vitro model. Exp. Eye Res., 2014. [DOI | PubMed]
- W. Zuo, F. Huang, Y.J. Chiang, M. Li, J. Du, Y. Ding, T. Zhang, H.W. Lee, L.S. Jeong, Y. Chen. c-Cbl-Mediated Neddylation Antagonizes Ubiquitination and Degradation of the TGF-β Type II Receptor. Mol. Cell, 2013. [DOI | PubMed]
- E.A. Wohlfert, L. Gorelik, R. Mittler, R.A. Flavell, R.B. Clark. Cutting edge: Deficiency in the E3 ubiquitin ligase Cbl-b results in a multifunctional defect in T cell TGF-β sensitivity in vitro and in vivo. J. Immunol., 2006. [DOI | PubMed]
- G. Di, X. Qi, X. Zhao, S. Zhang, P. Danielson, Q. Zhou. Corneal Epithelium-Derived Neurotrophic Factors Promote Nerve Regeneration. Investig. Ophthalmol. Vis. Sci., 2017. [DOI | PubMed]
- L. You, S. Ebner, F.E. Kruse. Glial Cell–Derived Neurotrophic Factor (GDNF)–Induced Migration and Signal Transduction in Corneal Epithelial Cells. Investig. Ophthalmol. Vis. Sci., 2001. [PubMed]
- F. Bian, H. Qi, P. Ma, L. Zhang, K.-C. Yoon, S.C. Pflugfelder, D.-Q. Li. An Immunoprotective Privilege of Corneal Epithelial Stem Cells Against Th17 Inflammatory Stress by Producing Glial Cell-Derived Neurotrophic Factor. Stem Cells, 2010. [DOI | PubMed]
- B.D. Hyndman, M.J.F. Crupi, S. Peng, L.N. Bone, A.N. Rekab, E.Y. Lian, S.M. Wagner, C.N. Antonescu, L.M. Mulligan. Differential recruitment of E3 ubiquitin ligase complexes regulates RET isoform internalization. J. Cell Sci., 2017. [DOI | PubMed]
- S.C. Kales, M.M. Nau, A.S. Merchant, S. Lipkowitz. Enigma prevents Cbl-c-mediated ubiquitination and degradation of RETMEN2A. PLoS ONE, 2014. [DOI | PubMed]
- Z. Wang, H. Yang, F. Zhang, Z. Pan, J. Capó-Aponte, P.S. Reinach. Dependence of EGF-Induced Increases in Corneal Epithelial Proliferation and Migration on GSK-3 Inactivation. Investig. Ophthalmol. Vis. Sci., 2009. [DOI | PubMed]
- P.S. Reinach, S. Mergler, Y. Okada, S. Saika. Ocular transient receptor potential channel function in health and disease. BMC Ophthalmol., 2015. [DOI | PubMed]
- H. Yang, Z. Wang, J.E. Capó-Aponte, F. Zhang, Z. Pan, P.S. Reinach. Epidermal growth factor receptor transactivation by the cannabinoid receptor (CB1) and transient receptor potential vanilloid 1 (TRPV1) induces differential responses in corneal epithelial cells. Exp. Eye Res., 2010. [DOI | PubMed]
- Y. Okada, T. Sumioka, P.S. Reinach, M. Miyajima, S. Saika. Roles of Epithelial and Mesenchymal TRP Channels in Mediating Inflammatory Fibrosis. Front. Immunol., 2021. [DOI | PubMed]
- J. Song, Y. Wang, X. Li, Y. Shen, M. Yin, Y. Guo, L. Diao, Y. Liu, D. Yue. Critical role of TRPC6 channels in the development of human renal cell carcinoma. Mol. Biol. Rep., 2013. [DOI | PubMed]
- J. Waning, J. Vriens, G. Owsianik, L. Stüwe, S. Mally, A. Fabian, C. Frippiat, B. Nilius, A. Schwab. A novel function of capsaicin-sensitive TRPV1 channels: Involvement in cell migration. Cell Calcium, 2007. [DOI | PubMed]
- S.C. Pflugfelder. Tear dysfunction and the cornea: LXVIII Edward Jackson Memorial Lecture. Am. J. Ophthalmol., 2011. [DOI | PubMed]
- M.D.P. Willcox, P. Argüeso, G.A. Georgiev, J.M. Holopainen, G.W. Laurie, T.J. Millar, E.B. Papas, J.P. Rolland, T.A. Schmidt, U. Stahl. TFOS DEWS II Tear Film Report. Ocul. Surf., 2017. [DOI | PubMed]
- H. Lam, L. Bleiden, C.S. de Paiva, W. Farley, M.E. Stern, S.C. Pflugfelder. Tear cytokine profiles in dysfunctional tear syndrome. Am. J. Ophthalmol., 2009. [DOI | PubMed]
- C. García-López, M. Rodríguez-Calvo-de-Mora, D. Borroni, J.M. Sánchez-González, V. Romano, C. Rocha-de-Lossada. The role of matrix metalloproteinases in infectious corneal ulcers. Surv. Ophthalmol., 2023. [DOI | PubMed]
- M. Choi, Y.M. Park, B.Y. Ko. Comparative Evaluation of Matrix Metalloproteinase-9 Immunoassay and Tear Osmolarity Measurement for Diagnosing Severity of Dry Eye Disease. Korean J. Ophthalmol., 2023. [DOI | PubMed]
- E.R. Block, J.K. Klarlund. Wounding sheets of epithelial cells activates the epidermal growth factor receptor through distinct short- and long-range mechanisms. Mol. Biol. Cell, 2008. [DOI | PubMed]
- M.A. Stepp, S. Pal-Ghosh, G. Tadvalkar, A. Williams, S.C. Pflugfelder, C.S. de Paiva. Reduced intraepithelial corneal nerve density and sensitivity accompany desiccating stress and aging in C57BL/6 mice. Exp. Eye Res., 2018. [DOI | PubMed]
- M. Sacchetti, A. Lambiase. Neurotrophic factors and corneal nerve regeneration. Neural Regen. Res., 2017. [DOI | PubMed]
- M. Sacchetti, A. Lambiase. Diagnosis and management of neurotrophic keratitis. Clin. Ophthalmol., 2014. [DOI | PubMed]
- A. Lambiase, A. Micera, M. Sacchetti, M. Cortes, F. Mantelli, S. Bonini. Alterations of Tear Neuromediators in Dry Eye Disease. Arch. Ophthalmol., 2011. [DOI | PubMed]
- G.B. van Setten, T. Tervo, L. Viinikka, J. Perheentupa, A. Tarkkanen. Epidermal growth factor in human tear fluid: A minireview. Int. Ophthalmol., 1991. [DOI | PubMed]
- M. Saghizadeh, M. Chwa, A. Aoki, B. Lin, A. Pirouzmanesh, D.J. Brown, A.V. Ljubimov, M.C. Kenney. Altered Expression of Growth Factors and Cytokines in Keratoconus, Bullous Keratopathy and Diabetic Human Corneas. Exp. Eye Res., 2001. [DOI | PubMed]
- M. Saghizadeh, A.A. Kramerov, J. Tajbakhsh, A.M. Aoki, C. Wang, N.N. Chai, J.Y. Ljubimova, T. Sasaki, G. Sosne, M.R. Carlson. Proteinase and growth factor alterations revealed by gene microarray analysis of human diabetic corneas. Investig. Ophthalmol. Vis. Sci., 2005. [DOI | PubMed]
- K. Barton, D.C. Monroy, A. Nava, S.C. Pflugfelder. Inflammatory cytokines in the tears of patients with ocular rosacea. Ophthalmology, 1997. [DOI | PubMed]
- A. Thakur, M. Willcox. Cytokine and lipid inflammatory mediator profile of human tears during contact lens associated inflammatory diseases. Exp. Eye Res., 1998. [DOI | PubMed]
- X. Jiang, S.A. McClellan, R. Barrett, M. Foldenauer, L.D. Hazlett. HGF signaling impacts severity of Pseudomonas aeruginosa keratitis. Investig. Ophthalmol. Vis. Sci., 2014. [DOI | PubMed]
- X. Jiang, S.A. McClellan, R.P. Barrett, E.A. Berger, Y. Zhang, L.D. Hazlett. VIP and growth factors in the infected cornea. Investig. Ophthalmol. Vis. Sci., 2011. [DOI | PubMed]
- M. Salman, A. Verma, V.K. Singh, J. Jaffet, S. Chaurasia, D.K. Sahel, M. Ramappa, V. Singh. New Frontier in the Management of Corneal Dystrophies: Basics, Development, and Challenges in Corneal Gene Therapy and Gene Editing. Asia Pac. J. Ophthalmol., 2022. [DOI | PubMed]
- K.P. Burdon, S. Macgregor, Y. Bykhovskaya, S. Javadiyan, X. Li, K.J. Laurie, D. Muszynska, R. Lindsay, J. Lechner, T. Haritunians. Association of Polymorphisms in the Hepatocyte Growth Factor Gene Promoter with Keratoconus. Investig. Ophthalmol. Vis. Sci., 2011. [DOI | PubMed]
- S.E.M. Lucas, T. Zhou, N.B. Blackburn, R.A. Mills, J. Ellis, P. Leo, E. Souzeau, B. Ridge, J.C. Charlesworth, R. Lindsay. Rare, potentially pathogenic variants in 21 keratoconus candidate genes are not enriched in cases in a large Australian cohort of European descent. PLoS ONE, 2018. [DOI | PubMed]
- F.K. Alswailmi. Role of HGF polymorphisms in the development of keratoconus in South Asian population. Electron. J. Gen. Med., 2023. [DOI]
- S. Cohen. Isolation of a mouse submaxillary gland protein accelerating incisor eruption and eyelid opening in the new-born animal. J. Biol. Chem., 1962. [DOI | PubMed]
- J.D. Zieske, M. Wasson. Regional variation in distribution of EGF receptor in developing and adult corneal epithelium. J. Cell Sci., 1993. [DOI | PubMed]
- I. Dabin, Y. Courtois. Acidic fibroblast growth factor overexpression in corneal epithelial wound healing. Growth Factors, 1991. [DOI | PubMed]
- M.A. Stepp, J.D. Zieske, V. Trinkaus-Randall, B.M. Kyne, S. Pal-Ghosh, G. Tadvalkar, A. Pajoohesh-Ganji. Wounding the cornea to learn how it heals. Exp. Eye Res., 2014. [DOI | PubMed]
- J. Friend, Y. Ishii, R.A. Thoft. Corneal epithelial changes in diabetic rats. Ophthalmic Res., 1982. [DOI | PubMed]
- Y. Kaji. Prevention of diabetic keratopathy. Br. J. Ophthalmol., 2005. [DOI | PubMed]
- A.V. Ljubimov. Diabetic complications in the cornea. Vis. Res., 2017. [DOI | PubMed]
- Y.C. Liu, J.H. Francis, D.H. Abramson. Ocular Side Effects of Systemically Administered Chemotherapy. 2023
- C.G. Foerster, C. Cursiefen, F.E. Kruse. Persisting corneal erosion under cetuximab (Erbitux) treatment (epidermal growth factor receptor antibody). Cornea, 2008. [DOI | PubMed]
- G.E. Wood, H. Hockings, D.M. Hilton, S. Kermorgant. The role of MET in chemotherapy resistance. Oncogene, 2021. [DOI | PubMed]
- S.V. Murphy, A. Skardal, R.A. Nelson Jr, K. Sunnon, T. Reid, C. Clouse, N.D. Kock, J. Jackson, S. Soker, A. Atala. Amnion membrane hydrogel and amnion membrane powder accelerate wound healing in a full thickness porcine skin wound model. Stem Cells Transl. Med., 2020. [DOI | PubMed]
- S. Puyana, S. Ruiz, A. Elkbuli, E. Bernal, M. McKenney, R. Lim, M. Askari, H. Mir. Using Dehydrated Amniotic Membrane Skin Substitute in Facial Burns: Is There a Outcome Difference Between Adult and Pediatric Patients?. J. Craniofacial Surg., 2020. [DOI]
- B. Farhadihosseinabadi, M. Farahani, T. Tayebi, A. Jafari, F. Biniazan, K. Modaresifar, H. Moravvej, S. Bahrami, H. Redl, L. Tayebi. Amniotic membrane and its epithelial and mesenchymal stem cells as an appropriate source for skin tissue engineering and regenerative medicine. Artif. Cells Nanomed. Biotechnol., 2018. [DOI | PubMed]
- R. Mohan, A. Bajaj, M. Gundappa. Human Amnion Membrane: Potential Applications in Oral and Periodontal Field. J. Int. Soc. Prev. Community Dent., 2017. [DOI | PubMed]
- F. Dadkhah Tehrani, A. Firouzeh, I. Shabani, A. Shabani. A Review on Modifications of Amniotic Membrane for Biomedical Applications. Front. Bioeng. Biotechnol., 2021. [DOI | PubMed]
- R.A. Hortensius, J.H. Ebens, M.J. Dewey, B.A.C. Harley. Incorporation of the Amniotic Membrane as an Immunomodulatory Design Element in Collagen Scaffolds for Tendon Repair. ACS Biomater. Sci. Eng., 2018. [DOI | PubMed]
- H. He, W. Li, S.-Y. Chen, S. Zhang, Y.-T. Chen, Y. Hayashida, Y.-T. Zhu, S.C. Tseng. Suppression of activation and induction of apoptosis in RAW264. 7 cells by amniotic membrane extract. Investig. Ophthalmol. Vis. Sci., 2008. [DOI | PubMed]
- A. Horn, J. Saller, D. Cuttica, S. Neufeld. Review of Use of Amniotic Membrane Allograft in Total Ankle Replacements. Foot Ankle Orthop., 2019. [DOI]
- P.R. Tandel, D. Surati, S.R. Patel. Study of human amniotic membrane as a biological dressing in burn wounds. IJSS J. Surg., 2018
- T.J. Koob, J.J. Lim, N. Zabek, M. Massee. Cytokines in single layer amnion allografts compared to multilayer amnion/chorion allografts for wound healing. J. Biomed. Mater. Res. Part B Appl. Biomater., 2015. [DOI]
- N. Koizumi, T. Inatomi, C. Sotozono, N.J. Fullwood, A.J. Quantock, S. Kinoshita. Growth factor mRNA and protein in preserved human amniotic membrane. Curr. Eye Res., 2000. [DOI | PubMed]
- A. De Rötth. Plastic repair of conjunctival defects with fetal membranes. Arch. Ophthalmol., 1940. [DOI]
- B.C. Silveira, A.P. Ribeiro, F.D. Pizzinatto, P.M. Lobo, H.R. Miranda, N. de Assis Pereira. Effects of commercial amniotic membrane extract on the re-epithelialization time and the early expression of matrix metalloproteinase-9 in cats with experimentally induced corneal ulcers. Vet. Ophthalmol., 2023. [DOI | PubMed]
- S.-H. Lee, S.C.G. Tseng. Amniotic Membrane Transplantation for Persistent Epithelial Defects With Ulceration. Am. J. Ophthalmol., 1997. [DOI | PubMed]
- D.H. Dang, K.M. Riaz, D. Karamichos. Treatment of Non-Infectious Corneal Injury: Review of Diagnostic Agents, Therapeutic Medications, and Future Targets. Drugs, 2022. [DOI | PubMed]
- M.S. Murri, M. Moshirfar, O.C. Birdsong, Y.C. Ronquillo, Y. Ding, P.C. Hoopes. Amniotic membrane extract and eye drops: A review of literature and clinical application. Clin. Ophthalmol., 2018. [DOI | PubMed]
- A. Walkden. Amniotic Membrane Transplantation in Ophthalmology: An Updated Perspective. Clin. Ophthalmol., 2020. [DOI | PubMed]
- M.N. Soykan, B. Altug, H. Bas, H. Ghorbanpoor, H. Avci, S. Eroglu, S. Butun Sengel, A. Eker Sariboyaci, S. Gunes Bagis, O. Uysal. Developing a Novel Platelet-Rich Plasma-Laden Bioadhesive Hydrogel Contact Lens for the Treatment of Ocular Surface Chemical Injuries. Macromol. Biosci., 2023. [DOI | PubMed]
- C. Jumelle, E.S. Sani, Y. Taketani, A. Yung, F. Gantin, S.K. Chauhan, N. Annabi, R. Dana. Growth factor-eluting hydrogels for management of corneal defects. Mater. Sci. Eng. C Mater. Biol. Appl., 2021. [DOI | PubMed]
- S. Pal-Ghosh, A. Pajoohesh-Ganji, G. Tadvalkar, B.M. Kyne, X. Guo, J.D. Zieske, M.A. Stepp. Topical Mitomycin-C enhances subbasal nerve regeneration and reduces erosion frequency in the debridement wounded mouse cornea. Exp. Eye Res., 2016. [DOI | PubMed]
- S.C. Pflugfelder, M. Massaro-Giordano, V.L. Perez, P. Hamrah, S.X. Deng, L. Espandar, C.S. Foster, J. Affeldt, J.A. Seedor, N.A. Afshari. Topical Recombinant Human Nerve Growth Factor (Cenegermin) for Neurotrophic Keratopathy: A Multicenter Randomized Vehicle-Controlled Pivotal Trial. Ophthalmology, 2020. [DOI | PubMed]
- X. Zhang, S. Muddana, S.R. Kumar, J.N. Burton, P. Labroo, J. Shea, P. Stocking, C. Siegl, B. Archer, J. Agarwal. Topical Pergolide Enhance Corneal Nerve Regrowth Following Induced Corneal Abrasion. Investig. Ophthalmol. Vis. Sci., 2020. [DOI]
- T. Nishida, T.-i. Chikama, N. Morishige, R. Yanai, N. Yamada, J. Saito. Persistent Epithelial Defects Due to Neurotrophic Keratopathy Treated with a Substance P-Derived Peptide and Insulin-Like Growth Factor 1. Jpn. J. Ophthalmol., 2007. [DOI | PubMed]
- A. Bruscolini, M. Marenco, G.M. Albanese, A. Lambiase, M. Sacchetti. Long-term clinical efficacy of topical treatment with recombinant human nerve growth factor in neurotrophic keratopathy: A novel cure for a rare degenerative corneal disease?. Orphanet J. Rare Dis., 2022. [DOI | PubMed]
- E.D. Deeks, Y.N. Lamb. Cenegermin: A Review in Neurotrophic Keratitis. Drugs, 2020. [DOI | PubMed]
- L. Aloe. Rita Levi-Montalcini: The discovery of nerve growth factor and modern neurobiology. Trends Cell Biol., 2004. [DOI | PubMed]
- N. Fleeman, J. Mahon, S. Nevitt, R. Duarte, A. Boland, E. Kotas, Y. Dundar, J. McEntee, S. Ahmad. Cenegermin for Treating Neurotrophic Keratitis: An Evidence Review Group Perspective of a NICE Single Technology Appraisal. Pharmacoecon. Open, 2019. [DOI | PubMed]
- B.S. Adams, A.R. Patel. Cenegermin. StatPearls, 2023
- H. Sheha, S. Tighe, O. Hashem, Y. Hayashida. Update On Cenegermin Eye Drops In The Treatment Of Neurotrophic Keratitis. Clin. Ophthalmol., 2019. [DOI | PubMed]
- M. Chen, C. Chang, B. Levian, D.T. Woodley, W. Li. Why Are There So Few FDA-Approved Therapeutics for Wound Healing?. Int. J. Mol. Sci., 2023. [DOI | PubMed]
- V.K. Mohan. Recombinant human epidermal growth factor (REGEN-D™ 150): Effect on healing of diabetic foot ulcers. Diabetes Res. Clin. Pract., 2007. [DOI | PubMed]
- J. Berlanga-Acosta, J. Fernández-Montequín, C. Valdés-Pérez, W. Savigne-Gutiérrez, Y. Mendoza-Marí, A. García-Ojalvo, V. Falcón-Cama, D. García del Barco-Herrera, M. Fernández-Mayola, H. Pérez-Saad. Diabetic Foot Ulcers and Epidermal Growth Factor: Revisiting the Local Delivery Route for a Successful Outcome. BioMed Res. Int., 2017. [DOI | PubMed]
- H.L. Tuyet, T.T. Nguyen Quynh, H. Vo Hoang Minh, D.N. Thi Bich, T. Do Dinh, D. Le Tan, H.L. Van, T. Le Huy, H. Doan Huu, T.N. Tran Trong. The efficacy and safety of epidermal growth factor in treatment of diabetic foot ulcers: The preliminary results. Int. Wound J., 2009. [DOI | PubMed]
- J.D. Zieske, H. Takahashi, A.E.K. Hutcheon, A.C. Dalbone. Activation of Epidermal Growth Factor Receptor during Corneal Epithelial Migration. Investig. Ophthalmol. Vis. Sci., 2000. [PubMed]
- C. Scardovi, G. De Felice, A. Gazzaniga. Epidermal growth factor in the topical treatment of traumatic corneal ulcers. Ophthalmologica, 1993. [DOI | PubMed]
- J.C. Pastor, M. Calonge. Epidermal Growth Factor and Corneal Wound Healing: A Multicenter Study. Cornea, 1992. [DOI | PubMed]
- E. Ibrahim, W.H. Dean, N. Price, A. Gomaa, G. Ayre, S. Guglani, A. Sallam. Perforating corneal ulceration in a patient with lung metastatic adenocarcinoma treated with gefitinib: A case report. Case Rep. Ophthalmol. Med., 2012. [DOI | PubMed]
- M. Dellaert, T. Casey, S. Wiffen, J. Gordon, P. Johnson, A. Geerards, W. Rijneveld, L. Remeijer, P. Mulder, W. Beekhuis. Influence of topical human epidermal growth factor on postkeratoplasty re-epithelialisation. Br. J. Ophthalmol., 1997. [DOI | PubMed]
- A.S. Kandarakis, C. Page, H.E. Kaufman. The effect of epidermal growth factor on epithelial healing after penetrating keratoplasty in human eyes. Am. J. Ophthalmol., 1984. [DOI | PubMed]
- A. Buckley, J.M. Davidson, C.D. Kamerath, T.B. Wolt, S.C. Woodward. Sustained release of epidermal growth factor accelerates wound repair. Proc. Natl. Acad. Sci. USA, 1985. [DOI | PubMed]
- M.R. Bristow, F.L. Anderson, J.D. Port, L. Skerl, R.E. Hershberger, P. Larrabee, J.B. O’Connell, D.G. Renlund, K. Volkman, J. Murray. Differences in beta-adrenergic neuroeffector mechanisms in ischemic versus idiopathic dilated cardiomyopathy. Circulation, 1991. [DOI | PubMed]
- M.R. Bristow, R. Ginsburg, W. Minobe, R.S. Cubicciotti, W.S. Sageman, K. Lurie, M.E. Billingham, D.C. Harrison, E.B. Stinson. Decreased Catecholamine Sensitivity and β-Adrenergic-Receptor Density in Failing Human Hearts. N. Engl. J. Med., 1982. [DOI | PubMed]
- F. Ledda, G. Paratcha. Negative Regulation of Receptor Tyrosine Kinase (RTK) Signaling: A Developing Field. Biomark. Insights, 2007. [DOI | PubMed]
- C.L. Neben, M. Lo, N. Jura, O.D. Klein. Feedback regulation of RTK signaling in development. Dev. Biol., 2019. [DOI | PubMed]
- L.K. Goh, A. Sorkin. Endocytosis of receptor tyrosine kinases. Cold Spring Harb. Perspect. Biol., 2013. [DOI | PubMed]
- G. Swaminathan, A.Y. Tsygankov. The Cbl family proteins: Ring leaders in regulation of cell signaling. J. Cell. Physiol., 2006. [DOI | PubMed]
- P. Peschard, M. Park. Escape from Cbl-mediated downregulation. Cancer Cell, 2003. [DOI | PubMed]
- G. Viticchie, P.A.J. Muller. c-Met and Other Cell Surface Molecules: Interaction, Activation and Functional Consequences. Biomedicines, 2015. [DOI | PubMed]
- N.M. Weathington, R.K. Mallampalli. Emerging therapies targeting the ubiquitin proteasome system in cancer. J. Clin. Investig., 2014. [DOI | PubMed]
- C.H. Chang, K. Bijian, D. Qiu, J. Su, A. Saad, M.S. Dahabieh, W.H. Miller, M.A. Alaoui-Jamali. Endosomal sorting and c-Cbl targeting of paxillin to autophagosomes regulate cell-matrix adhesion turnover in human breast cancer cells. Oncotarget, 2017. [DOI | PubMed]
- N. Li, M. Lorinczi, K. Ireton, L.A. Elferink. Specific Grb2-mediated Interactions Regulate Clathrin-dependent Endocytosis of the cMet-tyrosine Kinase. J. Biol. Chem., 2007. [DOI | PubMed]
- A.M. van der Bliek, T.E. Redelmeier, H. Damke, E.J. Tisdale, E.M. Meyerowitz, S.L. Schmid. Mutations in human dynamin block an intermediate stage in coated vesicle formation. J. Cell Biol., 1993. [DOI | PubMed]
- A.M. van der Bliek, E.M. Meyerowitz. Dynamin-like protein encoded by the Drosophila shibire gene associated with vesicular traffic. Nature, 1991. [DOI | PubMed]
- J.S. Herskovits, C.C. Burgess, R.A. Obar, R.B. Vallee. Effects of mutant rat dynamin on endocytosis. J. Cell Biol., 1993. [DOI | PubMed]
- R.C. de Oliveira, S. Murillo, P. Saikia, S.E. Wilson. The Efficacy of Topical HGF on Corneal Fibrosis and Epithelial Healing after Scar-Producing PRK Injury in Rabbits. Transl. Vis. Sci. Technol., 2020. [DOI]
- H. Funakoshi, T. Nakamura. Hepatocyte growth factor (HGF): Neurotrophic functions and therapeutic implications for neuronal injury/diseases. Curr. Signal Transduct. Ther., 2011. [DOI]
- K. Nishida, C. Sotozono, W. Adachi, S. Yamamoto, N. Yokoi, S. Kinoshita. Transforming growth factor-beta 1, -beta 2 and -beta 3 mRNA expression in human cornea. Curr. Eye Res., 1995. [DOI | PubMed]
- B.M. Stramer, J.D. Zieske, J.C. Jung, J.S. Austin, M.E. Fini. Molecular mechanisms controlling the fibrotic repair phenotype in cornea: Implications for surgical outcomes. Investig. Ophthalmol. Vis. Sci., 2003. [DOI | PubMed]
- S.K. Masur, H.S. Dewal, T.T. Dinh, I. Erenburg, S. Petridou. Myofibroblasts differentiate from fibroblasts when plated at low density. Proc. Natl. Acad. Sci. USA, 1996. [DOI | PubMed]
- K. Nakamura, D. Kurosaka, M. Yoshino, T. Oshima, H. Kurosaka. Injured corneal epithelial cells promote myodifferentiation of corneal fibroblasts. Investig. Ophthalmol. Vis. Sci., 2002. [PubMed]
- H. Kaur, S.S. Chaurasia, F.W. de Medeiros, V. Agrawal, M.Q. Salomao, N. Singh, B.K. Ambati, S.E. Wilson. Corneal stroma PDGF blockade and myofibroblast development. Exp. Eye Res., 2009. [DOI | PubMed]
- H. Kaur, S.S. Chaurasia, V. Agrawal, C. Suto, S.E. Wilson. Corneal myofibroblast viability: Opposing effects of IL-1 and TGF β1. Exp. Eye Res., 2009. [DOI | PubMed]
- V. Singh, F.L. Barbosa, A.A. Torricelli, M.R. Santhiago, S.E. Wilson. Transforming growth factor β and platelet-derived growth factor modulation of myofibroblast development from corneal fibroblasts in vitro. Exp. Eye Res., 2014. [DOI | PubMed]
- S.E. Wilson. Corneal wound healing. Exp. Eye Res., 2020. [DOI | PubMed]
- J.V. Jester, T. Moller-Pedersen, J. Huang, C.M. Sax, W.T. Kays, H.D. Cavangh, W.M. Petroll, J. Piatigorsky. The cellular basis of corneal transparency: Evidence for ‘corneal crystallins’. J. Cell Sci., 1999. [DOI | PubMed]
- S. Mizuno, K. Matsumoto, T. Kurosawa, Y. Mizuno-Horikawa, T. Nakamura. Reciprocal balance of hepatocyte growth factor and transforming growth factor-beta 1 in renal fibrosis in mice. Kidney Int., 2000. [DOI | PubMed]
- T. Nakamura, S. Mizuno. The discovery of hepatocyte growth factor (HGF) and its significance for cell biology, life sciences and clinical medicine. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci., 2010. [DOI | PubMed]
- T. Inoue, H. Okada, T. Kobayashi, Y. Watanabe, Y. Kanno, J.B. Kopp, T. Nishida, M. Takigawa, M. Ueno, T. Nakamura. Hepatocyte growth factor counteracts transforming growth factor-beta1, through attenuation of connective tissue growth factor induction, and prevents renal fibrogenesis in 5/6 nephrectomized mice. FASEB J., 2003. [DOI | PubMed]
- K. Iekushi, Y. Taniyama, J. Azuma, F. Sanada, H. Kusunoki, T. Yokoi, N. Koibuchi, K. Okayama, H. Rakugi, R. Morishita. Hepatocyte growth factor attenuates renal fibrosis through TGF-β1 suppression by apoptosis of myofibroblasts. J. Hypertens., 2010. [DOI | PubMed]
- M. Ishizaki, G. Zhu, T. Haseba, S.S. Shafer, W.W. Kao. Expression of collagen I, smooth muscle alpha-actin, and vimentin during the healing of alkali-burned and lacerated corneas. Investig. Ophthalmol. Vis. Sci., 1993. [PubMed]
- H. Kurosaka, D. Kurosaka, K. Kato, Y. Mashima, Y. Tanaka. Transforming growth factor-beta 1 promotes contraction of collagen gel by bovine corneal fibroblasts through differentiation of myofibroblasts. Investig. Ophthalmol. Vis. Sci., 1998. [PubMed]
- N.S. Nagaraj, P.K. Datta. Targeting the transforming growth factor-beta signaling pathway in human cancer. Expert Opin. Investig. Drugs, 2010. [DOI]
- M. Kamimoto, S. Mizuno, K. Matsumoto, T. Nakamura. Hepatocyte growth factor prevents multiple organ injuries in endotoxemic mice through a heme oxygenase-1-dependent mechanism. Biochem. Biophys. Res. Commun., 2009. [DOI | PubMed]
- S. Gupta, N.R. Sinha, L.M. Martin, L.M. Keele, P.R. Sinha, J.T. Rodier, J.R. Landreneau, N.P. Hesemann, R.R. Mohan. Long-Term Safety and Tolerability of BMP7 and HGF Gene Overexpression in Rabbit Cornea. Transl. Vis. Sci. Technol., 2021. [DOI | PubMed]
- S. Gupta, M.K. Fink, A. Ghosh, R. Tripathi, P.R. Sinha, A. Sharma, N.P. Hesemann, S.S. Chaurasia, E.A. Giuliano, R.R. Mohan. Novel Combination BMP7 and HGF Gene Therapy Instigates Selective Myofibroblast Apoptosis and Reduces Corneal Haze In Vivo. Investig. Ophthalmol. Vis. Sci., 2018. [DOI | PubMed]
- E. Uchikawa, Z. Chen, G.Y. Xiao, X. Zhang, X.C. Bai. Structural basis of the activation of c-MET receptor. Nat. Commun., 2021. [DOI | PubMed]
- R. Paumelle, D. Tulashe, Z. Kherrouche, S. Plaza, C. Leroy, S. Reveneau, B. Vandenbunder, V. Fafeur. Hepatocyte growth factor/scatter factor activates the ETS1 transcription factor by a RAS-RAF-MEK-ERK signaling pathway. Oncogene, 2002. [DOI | PubMed]
- A. Kakazu, G. Chandrasekher, H.E. Bazan. HGF protects corneal epithelial cells from apoptosis by the PI-3K/Akt-1/Bad- but not the ERK1/2-mediated signaling pathway. Investig. Ophthalmol. Vis. Sci., 2004. [DOI | PubMed]
- G.A. Rodrigues, M. Park, J. Schlessinger. Activation of the JNK pathway is essential for transformation by the Met oncogene. EMBO J., 1997. [DOI | PubMed]
- M. Garcia-Guzman, F. Dolfi, K. Zeh, K. Vuori. Met-induced JNK activation is mediated by the adapter protein Crk and correlates with the Gab1—Crk signaling complex formation. Oncogene, 1999. [DOI | PubMed]
- Y.-W. Zhang, L.-M. Wang, R. Jove, G.F. Vande Woude. Requirement of Stat3 signaling for HGF/SF-Met mediated tumorigenesis. Oncogene, 2002. [DOI | PubMed]
- Z.A. Syed, W. Yin, K. Hughes, J.N. Gill, R. Shi, J.L. Clifford. HGF/c-met/Stat3 signaling during skin tumor cell invasion: Indications for a positive feedback loop. BMC Cancer, 2011. [DOI | PubMed]
- S. Biswas, M. Shafiquzzaman, G. Yu, P. Li, Q. Yu, P. Zhao, B. Li, J. Li. Notch1 signaling in keratocytes maintains corneal transparency by suppressing VEGF expression. Stem Cell Rep., 2022. [DOI]
- T. Sakamoto, H. Ueno, K. Sonoda, T. Hisatomi, K. Shimizu, H. Ohashi, H. Inomata. Blockade of TGF-β by in vivo gene transfer of a soluble TGF-β type II receptor in the muscle inhibits corneal opacification, edema and angiogenesis. Gene Ther., 2000. [DOI | PubMed]
- E.S. Colombo, G. Menicucci, P.G. McGuire, A. Das. Hepatocyte growth factor/scatter factor promotes retinal angiogenesis through increased urokinase expression. Investig. Ophthalmol. Vis. Sci., 2007. [DOI | PubMed]
- M. Boulton. A role for hepatocyte growth factor in diabetic retinopathy?. 1999
- M. Ramos-Lopez, T. Hirose, K. Lashkari. Hepatocyte growth factor receptor is selectively activated in the vascular compartment of stage 5 retinopathy of prematurity. Investig. Ophthalmol. Vis. Sci., 2003
- M. He, T. Han, Y. Wang, Y.H. Wu, W.S. Qin, L.Z. Du, C.Q. Zhao. Effects of HGF and KGF gene silencing on vascular endothelial growth factor and its receptors in rat ultraviolet radiation-induced corneal neovascularization. Int. J. Mol. Med., 2019. [DOI | PubMed]
- N. Lee, S.H. Lee, J. Lee, M.Y. Lee, J. Lim, S. Kim, S. Kim. Hepatocyte growth factor is necessary for efficient outgrowth of injured peripheral axons in in vitro culture system and in vivo nerve crush mouse model. Biochem. Biophys. Rep., 2021. [DOI | PubMed]
- S. Honda, M. Kagoshima, A. Wanaka, M. Tohyama, K. Matsumoto, T. Nakamura. Localization and functional coupling of HGF and c-Met/HGF receptor in rat brain: Implication as neurotrophic factor. Brain Res. Mol. Brain Res., 1995. [DOI | PubMed]
