Functional brain changes after alternative pharmacological interventions in posttraumatic stress disorder: A systematic review of clinical trials
Abstract
Background:
Posttraumatic stress disorder (PTSD) is a complex and heterogeneous mental health condition that can develop after exposure to a traumatic event. Clinical trials have used alternative pharmacological agents to treat PTSD, but their associated neural correlates remain unclear. The present systematic review aims to summarize the changes in brain function associated with the use of these alternative pharmacological agents in PTSD.
Methods:
Clinical trials using functional magnetic resonance imaging, either at rest or during the performance of tasks, were included if they compared the effects of alternative pharmacological agents between PTSD patients and either trauma‐exposed controls or never‐exposed healthy controls.
Results:
Sixteen studies were included, of which 11 used intranasal oxytocin, 2 used hydrocortisone, and 3 used delta‐9‐tetrahydrocannabinol (THC). Oxytocin administration was associated with the normalization of functional connectivity between the ventromedial prefrontal cortex and amygdala as well as enhanced the function of brain regions specifically involved in emotion processing (e.g., amygdala), working memory (e.g., dorsolateral prefrontal cortex), and reward (e.g., putamen). Hydrocortisone did not influence brain function at rest or during the performance of an autobiographical memory task, whereas THC was associated with the reduction of the amygdala and increased medial prefrontal cortex activation.
Conclusions:
This systematic review identified preliminary evidence for normalizing brain function after the use of alternative pharmacological agents. Importantly, sex‐specific differences were noted, in particular when using oxytocin, that will require further investigation.
Article type: Review Article
Keywords: functional magnetic resonance imaging, hydrocortisone, oxytocin, posttraumatic stress disorder
Affiliations: Department of Clinical Psychology, School of Medicine Shahid Beheshti University of Medical Science Tehran Iran; Faculty of Medicine Qazvin University of Medical Science Qazvin Iran; School of Medicine Shahid Beheshti University of Medical Science Tehran Iran; Department of Psychiatry Amsterdam University Medical Centers Location AMC, Amsterdam Public Health Amsterdam The Netherlands; ARQ National Psychotrauma Centre Diemen The Netherlands; NeuroRecovery Research Hub, School of Psychology The University of New South Wales (UNSW) Sydney Sydney New South Wales Australia; Neuroscience Research Australia Randwick New South Wales Australia
License: © 2023 The Authors. Brain and Behavior published by Wiley Periodicals LLC. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/brb3.3292 | PubMed: 37864378 | PMC: PMC10726808
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (621 KB)
INTRODUCTION
Posttraumatic stress disorder (PTSD) is a severe psychiatric disorder that can arise after exposure to a traumatic event. Although around 6% of all people exposed to a traumatic event will develop PTSD (Breslau, ref. 2001; Bromet et al., ref. 1998; Mclaughlin et al., ref. 2015), these rates are significantly higher (25%–35%) for assault victims, refugees, or combat veterans (Alexandra Kredlow et al., ref. 2022). Symptoms of PTSD include persistent re‐experiencing of the traumatic event, avoidance of trauma‐associated stimuli, negative thoughts or feelings, and increased arousal that persist for at least a month after trauma exposure (American Psychiatric Association, ref. 2013). Clinical manifestations and symptom intensity of PTSD are very heterogeneous (Regier et al., ref. 2013), possibly implying the involvement of multiple neurobiological systems (Albucher & Liberzon, ref. 2002).
Among the numerous theories proposed to explain PTSD, including the emotional processing to fear model (Foa & Kozak, ref. 1986), the dual‐representation theory (Brewin et al., ref. 1996), or the cognitive model of PTSD (Ehlers & Clark, ref. 2000), most neuroimaging studies have been based on the failure to inhibit fear model of PTSD (Liberzon & Sripada, ref. 2007; Rauch et al., ref. 2006). This model implies insufficient top–down regulation from the medial prefrontal cortex (mPFC) to the amygdala (Liberzon & Sripada, ref. 2007; Rauch et al., ref. 2006; Woodward et al., ref. 2006), leading to the hyperactivation of the amygdala. Changes in amygdala–mPFC connectivity can lead to failure to suppress the so‐called default mode network (Greicius & Menon, ref. 2004) during cognitive engagement (Selemon et al., ref. 2019). This aberrant top–down regulation can be normalized following successful psychological and/or pharmacological treatments (Quidé et al., ref. 2012). Anatomical results from the Enhancing NeuroImaging Genetics through Meta‐Analyses consortium indicate that smaller hippocampal and amygdala, in addition to smaller left and right lateral orbitofrontal gyri, were evident in people with current PTSD compared with trauma‐exposed control (TEC) subjects (Logue et al., ref. 2018; Wang et al., ref. 2021), although smaller hippocampal volume may be present prior to trauma exposure and could represent a risk factor to develop PTSD (Gilbertson et al., ref. 2002; Quidé et al., ref. 2018). Despite evidence for decreased mPFC activation following successful first‐line treatment, the effects of alternative treatments on amygdala activation or volume remain unclear (Manthey et al., ref. 2021; Quidé et al., ref. 2012). Other regions, such as the thalamus, the precuneus, and the occipital cortex, have also been inconsistently associated with PTSD and/or trauma exposure (Patel et al., ref. 2012; Quidé et al., ref. 2021; Sartory et al., ref. 2013).
Both psychological and pharmacological approaches have been recommended for treating PTSD. First‐line psychological interventions for PTSD include trauma‐focused cognitive behavioral therapy, although other recommended treatments, including eye movement desensitization and reprocessing, cognitive processing therapy, or prolonged exposure therapy, are also widely used (Asmundson et al., ref. 2019; Bisson & Olff, ref. 2021), whereas antidepressants such as selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants, or monoamine oxidase inhibitors have been recommended as first‐line pharmacological options (Albucher & Liberzon, ref. 2002). A meta‐analysis of pharmacological treatments indicates that sertraline, fluoxetine, paroxetine, prazosin, venlafaxine, quetiapine, and risperidone have a small positive effect with no evidence of superiority for one intervention over another (Hoskins et al., ref. 2021). The use of SSRIs has been linked with both changes in brain function and morphology in PTSD (Quidé et al., ref. 2012): Fluoxetine was found to normalize hyperactivity of the cerebellum, precuneus, and supplementary motor cortex (Fernandez et al., ref. 2001), whereas paroxetine was associated with increased hippocampal volume (Vermetten et al., ref. 2003) and increased frontal (orbitofrontal cortex, anterior cingulate cortex) function during trauma versus neutral script presentations (Fani et al., ref. 2011).
Evidence for the efficacy of current antidepressant approaches is limited, with about one third of PTSD patients still meeting criteria for PTSD after treatment (Hoskins et al., ref. 2021; Sijbrandij et al., ref. 2015). As such, alternative approaches have been proposed (De Kleine et al., ref. 2013). A popular approach is to use intranasal oxytocin: Oxytocin receptors are particularly present in key regions involved in PTSD, including the amygdala, hippocampus, parahippocampal gyrus, anterior cingulate cortex, hypothalamus, or olfactory bulb (Boccia et al., ref. 2013). Intranasal oxytocin administration in non‐trauma‐exposed healthy individuals modulates the activity of these brain regions and associated networks, reaching to the insula, nucleus accumbens, or the dorsomedial prefrontal cortex (Feng et al., ref. 2015; Olff et al., ref. 2010; Rilling et al., ref. 2014). By modulating the activity of these brain regions and networks, oxytocin can assist with fear regulation and reward processing (Harari‐Dahan & Bernstein, ref. 2014) by enhancing motivational salience and increasing the desire for social interaction (Yatzkar & Klein, ref. 2010). Similarly, cannabinoids have also been proposed to treat PTSD (Mayo et al., ref. 2022). The endocannabinoid system is essential in fear extinction (Hill et al., ref. 2018), learning, and memory (Ruehle et al., ref. 2012). Low dose of delta‐9‐tetrahydrocannabinol (THC), the psychoactive constituent of cannabis, significantly reduces amygdala reactivity to social cues of threat and fear inhibition tasks (Rabinak et al., ref. 2014, ref. 2020). More recently, ketamine has been proposed as a new option to treat chronic PTSD (Ragnhildstveit et al., ref. 2023). This non‐competitive antagonist of the N‐methyl‐d‐aspartate glutamate receptor can reverse the effects of trauma and chronic stress on the amygdala, hippocampus, and prefrontal cortex (Duman et al., ref. 2012), thus improving mood and fear learning by promoting neurogenesis, synaptogenesis, and cell proliferation (Clarke et al., ref. 2017; Duman et al., ref. 2012). Finally, the use of psychedelics, especially 3,4‐methylenedioxymethamphetamine (MDMA), has been used to augment the beneficial effects of psychotherapies in PTSD: MDMA is thought to reduce the fear response associated with re‐experiencing traumatic memories, to facilitate tolerable processing of traumatic content (memory reconsolidation, fear extinction) in patients with PTSD (Mithoefer et al., ref. 2011). These effects are thought to target the functional connectivity between the amygdala and hippocampus in healthy volunteers (Carhart‐Harris et al., ref. 2015) and in PTSD patients (Singleton et al., ref. 2022).
Other pharmacological agents have been proposed to prevent the development of PTSD, early following exposure to traumatic events. Administration of hydrocortisone, a synthetic form of cortisol, early following trauma exposure has a large effect in reducing the risk of PTSD (Sijbrandij et al., ref. 2015). Glucocorticoid receptors are widely expressed in the hippocampus, the amygdala, and the prefrontal cortex, to regulate the hypothalamic–pituitary–adrenal axis activity (Mcwen, ref. 2017; Smith & Vale, ref. 2006). Glucocorticoid administration may offer critical therapeutic advances (De Quervain et al., ref. 2003), by reducing the retrieval of traumatic memories and promote extinction and inhibitory fear learning (De Quervain et al., ref. 2009; Sijbrandij et al., ref. 2015), and can enhance the effects of prolonged exposure therapy (Yehuda et al., ref. 2015). Similarly to the use of glucocorticoids, the beta‐adrenergic blocker propranolol is used to prevent the development of PTSD by blocking the consolidation of fearful memories via the dorsal mPFC (dmPFC) and improving the learning of a safe context by the hippocampus (Kroes et al., ref. 2016). Although early evidence indicates that propranolol use is associated with decreased symptoms and increased activation in the right anterior cingulate cortex in response to fearful versus happy faces (Mahabir et al., ref. 2015), the overall efficacy of propranolol in preventing PTSD is unclear (Sijbrandij et al., ref. 2015).
Although evidence indicates that psychotherapy may upregulate the mPFC, leading to symptom reduction in PTSD patients (Manthey et al., ref. 2021), the neural correlates underlying successful alternative pharmacological intervention in PTSD remain unclear. Understanding the effects of these new treatment options on brain function is critical to extend existing models in order to consider the translational value of selected drugs or drug receptors into clinical therapy (Trist et al., ref. 2014) and to provide the best intervention options to people suffering from PTSD. The current study reviews the evidence resulting from clinical trials evaluating the changes in brain function following the use of these alternative pharmacological agents in PTSD patients.
MATERIALS AND METHODS
Search strategy
Adhering to the Preferred Reporting Items for Systematic Reviews and Meta‐Analyses (PRISMA) 2020 statement (Page et al., ref. 2021), the PubMed, Scopus, and Web of Science databases were searched for studies published before September 2022. The search was limited to clinical trials estimating the effects of alternative pharmacological agents other than first‐line pharmacological treatments (e.g., SSRIs), on resting‐state or task‐related functional magnetic resonance imaging (fMRI) outcomes. The search was performed on PubMed using the following MESH terms: stress disorders and posttraumatic and magnetic resonance imaging. The search was then restricted to Clinical Trials (search filters “article type: Clinical Trial”). Search on Web of Science and Scopus using the search terms MRI, fMRI, neuroimaging, magnetic resonance imaging crossed with therapeutic, treatment, intervention and again fully crossed with the terms posttraumatic stress disorder, PTSD. The study selection processes are outlined in the flow diagram (see Figure 1).
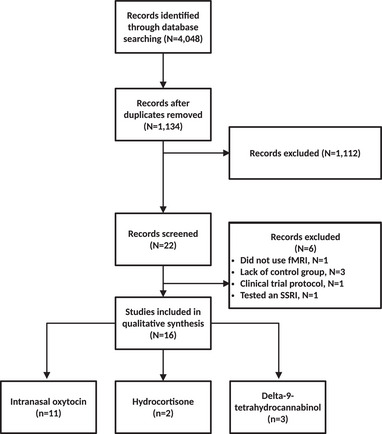
Study selection
Inclusion criteria included original articles on adult patients with PTSD, diagnosed based on either the criteria from the Diagnostic and Statistical Manual of Mental Disorders fourth or fifth editions (DSM‐IV, DSM‐IV‐TR, and DSM‐5) (APA, 1994, 2000, ref. 2013) or the International Statistical Classification of Diseases and Related Health Problems (ICD‐10) (World Health Organization, ref. 1992). Studies including a healthy control (HC) group and/or a group of TEC who did not develop PTSD following trauma were considered to determine both the unspecific (irrespective of the group) and specific effects of the studied pharmacological agents in PTSD patients relative to their control groups. Studies that were not clinical trials, studies that did not report fMRI as their outcome measures, studies with incomplete reporting, studies comparing two pharmacological interventions, studies not including an HC or TEC comparison groups, and studies published in other languages than English were excluded. Clinical trials allowing concomitant antidepressant use were excluded. Two reviewers independently screened the identified abstracts. Extracted data included the year of publication, sex, age, diagnosing tool, type of fMRI (resting state of task‐related fMRI), and the name of the task for task‐related fMRI studies.
Data extraction
Study characteristics were extracted, including sample size, diagnostic tools, type of treatment, and type of brain imaging (see Table 1). In addition, the relevant results from each study, including the main effects of the treatment of interest, the main effects of groups, and the effects of the group‐by‐treatment interactions on brain function, were extracted and are reported in Table 2. Other results within each study were not reported here if they were not related to the aims of the present study.
TABLE 1: Details of the included studies.
| Study | Sample size of patients (female) | Sample size of control group (female) Type of control group | Diagnostic tools | Type of treatment versus control (sham, placebo, other drug) | Type of fMRI |
|---|---|---|---|---|---|
| Intranasal oxytocin | |||||
| Nawjin et al. (ref. 2015) | 35 (14) | 37 (18)—TEC | CAPS‐MINI‐SCID | 40IU versus placebo | Monetary incentive delay |
| Koch et al. (ref. 2016) | 37 (16) | 40 (20)—TEC | CAPS‐MINI‐SCID | 40IU versus placebo | Resting‐state and emotional face‐matching, monetary and social incentive delay, reward tasks |
| Frijling et al. (ref. 2016) | 24 (15) | 20 (11)—HC | TSQ‐PDI‐CAPS‐MINI‐ETI | 40IU versus placebo | Resting‐state and Script‐driven imagery |
| Frijling et al. (ref. 2016) | 23 (14) | 18 (10)—HC | TSQ‐CAPS‐MINI | 40IU versus placebo | Emotional face‐matching task |
| Nawijn et al. (ref. 2015) | 35 (All) | 37 (All)—HC | CAPS‐MINI‐SCID | 40IU versus placebo | Social incentive delay task |
| Koch et al. (ref. 2018) | 36 (15) | 40 (20)—TEC | CAPS‐MINI‐SCID | 40IU versus placebo | Distraction task |
| Flanagan et al. (ref. 2018) | 16 (7) | 18 (11)—TEC | CTQ‐MINI‐SCID‐CAPS | 24IU versus placebo | Working memory task |
| Flanagan et al. (ref. 2019) | 17 (8) | 16 (10)‐TEC | CAPS‐SCID‐CTQ‐STAI | 24IU versus placebo | Facial affect recognition |
| Sippel et al. (ref. 2021) | 18 (12) | 16 (9)—TEC | CTQ‐CAPS‐SCID‐PDS | 24IU versus placebo | Facial affect recognition task |
| Crum et al. (ref. 2021) | 15 (7) | 16 (9)—TEC | CAPS‐PTDS‐MINI‐CTQ | 24IU versus placebo | Resting‐state |
| Hydrocortisone | |||||
| Metz et al. (ref. 2018) | 20 (All) | 40 (All)—HC | SCID‐PDS‐r‐CTQ‐ELS | 10 mg | Resting‐state fMRI |
| Metz et al. (ref. 2019) | 20 (All) | 40 (All)—HC | SCID‐PDS‐r‐ CTQ | 10 mg | Autobiographical memory test |
| Delta‐9‐tetrahydrocannabinol (THC) | |||||
| Rabinak et al. (ref. 2020) | 19 (14) | 52 (22)—HC, TEC | CAPS‐LEC‐5 | 7.5 mg versus placebo | Threat processing task |
| Pacitto et al. (ref. 2022) | 22 (15) | 30 (14)—TEC | CAPS‐LEC‐5 | 7.5 mg versus placebo | Emotion regulation task |
| Zabik et al. (ref. 2023) | 19 (13) | 52 (21)—HC, TEC | CAPS‐LEC‐5 | 7.5 mg versus placebo | Fear conditioning and extinction paradigm |
Abbreviations: CAPS, clinician‐administered PTSD scale; CTQ, child trauma questionnaire; ELS, early life stress; ETI, early trauma inventory; HAM‐D, Hamilton depression rating scale; HC, healthy controls; IU, international unit; LEC‐5, the life events checklist for DSM‐5; MADRS, Montgomery–Åsberg depression rating scale; MINI, mini‐international neuropsychiatric interview; MG, milligrams; PDI, peritraumatic distress inventory; PDS‐r, posttraumatic diagnostic scale; SCID, structured clinical interview for DSM; STAI, state‐trait anxiety inventory; TEC, trauma‐exposed controls; TSQ, trauma screening questionnaire.
TABLE 2: Effects of interest extracted from the included publications.
| Study | Effects of treatment of interest | Effects of group | Interaction group x treatment |
|---|---|---|---|
| Intranasal oxytocin | |||
| Crum et al. (ref. 2021) | ↓ Connectivity between DAN and VAN | – | – |
| Koch et al. (ref. 2016) | – | – | MalesOxytocin versus placebo (PTSD): ↑ right centromedial amygdala and left vmPFC connectivityMales PTSD versus TEC (placebo): ↓ right centromedial amygdala and left vmPFC connectivityFemalesOxytocin versus placebo (PTSD): ↓ right basolateral to bilateral dorsal anterior cingulate cortexPTSD versus TEC (placebo): ↑ right basolateral to bilateral dorsal anterior cingulate cortex |
| Frijling et al. (ref. 2016) | ↑ Connectivity between the amygdala and the insula ↓ connectivity between the amygdala and the ventro‐lateralPFC | – | Oxytocin: Trauma versus neutral script↓ connectivity between the amygdala and the left vmPFCPlacebo: Trauma versus neutral script↑ connectivity between the amygdala and the left vmPFC |
| Flanagan et al. (ref. 2018) | – | – | PTSD versus TEC (oxytocin)↑ connectivity between the dorsolateral prefrontal cortex and anterior cingulate increased in the 2‐back condition |
| Koch et al. (ref. 2016) | – | ‐ | Oxytocin versus placeboPTSD: ↓ amygdala reactivityHC: ↑ amygdala reactivity |
| Frijling et al. (ref. 2016) | – | – | Oxytocin versus placebo(PTSD)Fearful faces: ↑ right basolateral amygdala reactivityNeutral faces: ↑ left basolateral amygdala reactivity in women (not men) |
| Sippel et al. (ref. 2021) | – | – | Oxytocin versusplaceboPTSD: ↓ left amygdala and left anterior insula connectivity for femalesLeft amygdala‐ right anterior insula connectivity ↓ for females and ↑ for malesRight amygdala–right anterior insula connectivity for ↓ males and for ↑ females |
| Flanagan et al. (ref. 2019) | – | – | Oxytocin versus placeboPTSD: ROI: ↑ right amygdala and ↓ left amygdala to fearful faces; exploratory whole‐brain analyses: right occipital gyrus, right precentral/cingulate gyrus, left insula/inferior frontal gyrusTEC: ROI: ↑ left amygdala to fearful faces; exploratory whole‐brain analyses: bilateral fusiform gyrus |
| Nawijn et al. (ref. 2017) | Oxytocin versus placeboPTSD: ↑ right putamen activation and left insula in response to reward feedbackHC: ↓ right putamen activation and left insula in response to reward | ||
| Nawijn et al. (ref. 2016) | ↑ Left and right putamen, dorsal anterior cingulate cortex, and insula responses during reward and loss anticipation | ||
| Koch et al. (ref. 2016) | ↑ Left thalamus activation | OxytocinPTSD: ↑ connectivity between the left thalamus and the amygdala (males + females)TEC: ↑ connectivity between the left thalamus and the amygdalain males (not females) | |
| Hydrocortisone | |||
| Metz et al. (ref. 2018) | – | – | – |
| Metz et al. (ref. 2019) | – | – | – |
| Delta‐9‐tetrahydrocannabinol (THC) | |||
| a. Threat processing task | |||
| Rabinak et al. (ref. 2020) | – | – | THC versus placeboPTSD: ↓ amygdala activity and ↑ mPFC and ↓ amygdalostriatal and ↑ mPFC/rACC response to threatTEC: ↑ basolateral amygdalaHC: ↓ amygdalostriatal response to non‐threat |
| b. Emotion regulation task | |||
| Pacitto et al. (ref. 2022) | – | – | THC versus placeboPTSD: ↑ dmPFC activation during exposure to neutral images and ↑ cerebellar and posterior cingulate cortex (PCC)/precuneus activation during reappraisal↑ angular gyrus |
| c. Fear conditioning and extinction paradigm | |||
| Zabik et al. (ref. 2023) | – | – | THC versus placeboPTSD: ↑ vmPFC and amygdalaHC: ↑ vmPFC |
Abbreviations: HC, healthy controls; PFC, prefrontal cortex; PTSD, posttraumatic stress disorder; ROI, region‐of‐interest; TEC, trauma‐exposed controls; THC, delta‐9‐tetrahydrocannabinol; vmPFC, ventromedial prefrontal cortex.
Risk of bias assessment
Two authors (SL and AY) independently assessed the risks of bias using the Cochrane Collaboration’s Tool for Assessing Risk of Bias in Randomized Trials (RoB 2.0) (Sterne et al., ref. 2019). In addition to provide an overall bias estimation, this tool assesses five domains of potential bias for each individual study (selection of the reported result, measurement of the outcome, missing outcome data, deviation from intended interventions and randomization process) and ranks them as “low,” with “some concern” or “high.” In the event of discrepancy in bias assessment between reviewers, consensus was achieved through discussion.
RESULTS
A total of 4048 articles were identified. After removing duplicates, abstracts, and titles, 1134 papers were screened, from which 1112 articles were excluded. Twenty‐two articles were screened, with six records excluded because they did not satisfy the inclusion criteria (lack of control group, N = 3), tested the effects of an SSRI (paroxetine; N = 1), did not use fMRI (N = 1), and referred to a clinical trial protocol (N = 1). Sixteen articles were included in the review (see Figure 1 and Table 1); among them, 11 used intranasal oxytocin (3 resting‐state fMRI, 8 task‐related fMRI, from 4 distinct datasets), 2 used hydrocortisone (1 resting‐state fMRI, 1 task‐related fMRI, from one dataset), and 3 task‐related fMRI used THC (from one dataset). Effects of interventions, groups, and their interactions on brain function are reported in Table 2.
Oxytocin
Resting‐state fMRI
Koch et al. ref. 2016 used resting‐state fMRI to investigate the effects of intranasal oxytocin on the functional connectivity of the basolateral and centromedial amygdala in police officers with (n = 37) and without PTSD (n = 40) (Koch et al., ref. 2016). Following the administration of oxytocin, connectivity between the right centromedial amygdala and the left ventromedial prefrontal cortex (vmPFC) was normalized (enhanced) in male PTSD cases relative to placebo, reaching a similar level than the TEC group after oxytocin administration. Connectivity between these two regions was reduced in PTSD males compared to TEC males after placebo administration. There were effects of intervention or group on connectivity between the left vmPFC and the right centromedial amygdala in females. However, PTSD females showed reduced connectivity between the right basolateral to bilateral dorsal anterior cingulate cortex following oxytocin administration, compared to placebo (Koch et al., ref. 2016), whereas placebo was associated with enhanced connectivity between these regions in PTSD females, compared to TEC females. Compared to TEC (n = 16), childhood trauma survivors with PTSD (n = 15) showed reduced the strength of resting state connectivity between the dorsal (DAN; made of the precuneus, occipital cortex, parietal cortices, middle and superior frontal gyri, temporo‐occipital gyrus, and precentral gyrus) and ventral attentional networks (made of the supplemental motor area/superior frontal gyrus, bilateral superior and middle temporal gyrus, and bilateral inferior frontal gyrus) (Crum et al., ref. 2021). This pattern of connectivity was found increased after oxytocin administration in both groups.
Emotional processing
Four studies have investigated the effects of intranasal oxytocin administration while performing an emotional face‐matching task. In their cohort of police officers, Koch et al. examined the effects of oxytocin on amygdala reactivity toward emotional stimuli (Koch et al., ref. 2016). Compared to placebo (n = 40), oxytocin administration (n = 38) was associated with decreased amygdala reactivity in PTSD patients and enhanced amygdala reactivity in HCs. The intervention moderated the relationship between pre‐intervention anxiety scores and amygdala reactivity: This association between anxiety and amygdala reactivity was dampened after oxytocin administration, whereas it increased in the placebo group. In acute civilian PTSD patients, a single intranasal oxytocin administration (n = 23) showed enhanced right basolateral amygdala reactivity to fearful faces compared to the placebo group (n = 18). Results showed a significant intervention‐by‐sex interaction on amygdala reactivity to neutral faces, with women showing enhanced left amygdala reactivity to fearful stimuli after oxytocin administration (Frijling et al., ref. 2016).
In childhood trauma survivors with (n = 16) or without PTSD (n = 18), oxytocin administration was associated with reduced left amygdala–left anterior insula functional connectivity in response to fearful face presentation in females but not males, as well as reduced left amygdala–right anterior insula connectivity among females that was increased in males, and reduced right amygdala–right anterior insula connectivity in males that was increased in females (Sippel et al., ref. 2021). Finally, in the same sample, there was no effect of oxytocin on amygdala function, relative to placebo in the PTSD group. However, changes in left amygdala response to fearful face presentation were negatively associated with childhood trauma severity in individuals with PTSD (n = 17), whereas increased following oxytocin administration in the control group (n = 16). In addition, exploratory analyses outside of the amygdala indicated that the PTSD group showed larger patterns of activation in response to fearful faces in the right occipital gyrus, the right precentral/anterior cingulate gyrus, and the left insula‐inferior frontal gyrus (Flanagan et al., ref. 2019).
Social incentive delay task
Only one study investigated brain response to oxytocin when performing a social incentive delay task in police officers (Nawijn et al., ref. 2017). Compared to placebo (n = 36), intranasal oxytocin administration (n = 35) was associated with increased activation in the right putamen and left insula in response to reward feedback in the PTSD group, whereas decreased activation in these regions was evident in the control group.
Trauma‐related script (script‐driven imagery)
In a study of 44 civilians with acute PTSD (n = 24 received oxytocin and n = 20 received a placebo) recruited early following trauma exposure (scanning session within 11 days post trauma) (Frijling et al., ref. 2016), functional connectivity at rest between the (whole) amygdala and the left ventrolateral prefrontal cortex was weaker in response to a previously presented trauma‐related script compared to a neutral script following oxytocin administration, whereas this association was stronger in the placebo group. Irrespective of the script condition, the administration of oxytocin increased the strength of functional connectivity between the amygdala and the insula and decreased the strength of connectivity between the amygdala and the vmPFC (Frijling et al., ref. 2016).
Working memory
Only one study investigated the effects of oxytocin on brain function during the performance of an n‐back working memory task in people with childhood trauma‐related PTSD (n = 16) compared to childhood trauma TEC (n = 18) (Flanagan et al., ref. 2018). Compared to placebo, in the context of improved performance at the most difficult condition (2‐back), connectivity between the dmPFC and anterior cingulate cortex increased in the 2‐back condition among individuals with PTSD using oxytocin (relative to TEC) (Flanagan et al., ref. 2018).
Monetary incentive delay task
Only one study evaluated the effects of oxytocin on brain function during the performance of a monetary incentive delay task in police officers with (n = 35) or without PTSD (n = 37) (Nawijn et al., ref. 2016). Compared to placebo, oxytocin administration enhanced neural responses during reward and loss anticipation in both PTSD patients and controls in the left and right putamen, dorsal anterior cingulate cortex, and insula. Additionally, oxytocin relieved these anhedonia‐related neural reward deficits, suggesting that oxytocin may increase motivation and reward sensitivity.
Distraction task
The only study investigating the impact of oxytocin administration on brain response during the performance of a distraction task (Koch et al., ref. 2019) reported that former police officers showed enhanced left thalamus activation during the task after oxytocin administration compared to placebo, independently of being diagnosed (n = 37) or not with PTSD (n = 40). In addition, oxytocin administration was associated with increased left thalamus activity and task‐related functional connectivity with the amygdala in all PTSD patients and in male, but not female, TEC.
In summary, when processing emotional/traumatic stimuli, the administration of oxytocin was found to reduce amygdala reactivity, normalize mPFC–amygdala and amygdala–insula connectivities, and reduce the connectivity between ventral (affective) and dorsal (cognitive) attentional networks in PTSD. During the performance of (nonemotional) cognitive tasks, oxytocin administration was associated with increased activation and functional connectivity among regions critical for the cognitive processes studied: dorsolateral PFC and anterior cingulate cortex during the performance of a working memory task, putamen, dorsal anterior cingulate cortex, and insula during the performance of a reward, and thalamus during the performance of an attentional task. Importantly, sex differences have been observed.
Hydrocortisone
One group studied the effects of hydrocortisone on the brain function of females with PTSD (n = 20), borderline personality disorder (n = 18), and HCs (n = 40). There were no effects of hydrocortisone on patterns of resting‐state functional connectivity (Metz et al., ref. 2019) or brain function during the performance of an autobiographical memory task (Metz et al., ref. 2019).
Delta‐9‐tetrahydrocannabinol (THC)
Three studies from the same group, with sub‐samples of the same dataset, indicate that THC administration [HC (n = 25), TEC (n = 27), and PTSD (n = 19)] was associated with reduced amygdala reactivity and increased activation of the mPFC, along with amygdala‐mPFC functional connectivity during threat in adults with PTSD (Rabinak et al., ref. 2020). Relative to placebo (n = 26), administration of THC (n = 25) was associated with no overall changes in brain function during the performance of an emotion regulation task (independently of the condition) in people with PTSD (n = 21) compared to TEC (n = 30) (Pacitto et al., ref. 2022). However, in the PTSD group only, THC administration (n = 10), compared to placebo (n = 11), was associated with increased activation of the cerebellum when participants were asked to passively process neutral images (maintain neutral condition) only. Finally, while performing a fear conditioning and extinction paradigm, THC administration was associated with greater vmPFC activation in people with PTSD (n = 9) compared to TEC (n = 12), while greater activation of the hippocampus was evident during late (relative to early) extinction recall in the right hippocampus, independently of group (both PTSD and TEC) (Zabik et al., ref. 2023). Relative to placebo, THC administration was also associated with greater left amygdala activation during early presentations of the extinguished conditioned stimulus (relative to the unpaired conditioned and unconditioned stimuli) in PTSD.
Bias assessment
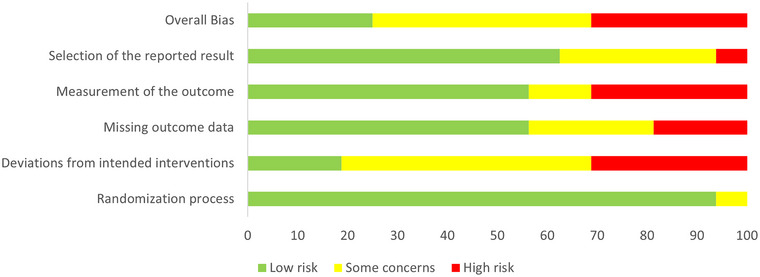
The overall quality assessment of the selected articles indicates that 25% of studies show low risk, 43.8% of studies included show some concerns, and 31.3% being considered at high risk (see Figure 2). Notably, the deviations from intended interventions were estimated being at 18.8% with low risk, 50% with some concerns, and 31.3% at high risk, whereas the selection of reported results was estimated to be at low risk for 62.5% of the included studies (31.3.2% with some concerns and 6.3% at high risk), and the measurement of the outcome and the missing outcome data were at low risk in 56.3% (12.5% with some concerns, 31.3% at high risk for the measurement of the outcome, 25% with some concerns, and 18.8% at high risk for the missing outcome). Finally, the randomization process was estimated being at low risk of bias in 92.8% of the included studies (6.3% with some concerns).
DISCUSSION
The present systematic review identified 16 clinical trials investigating the impacts of alternative pharmacological agents (oxytocin, hydrocortisone, and THC) on brain function in PTSD. Although these studies used various imaging paradigms (resting‐state fMRI and task‐related fMRI paradigms), results mostly converge toward similar functional changes in key regions involved in PTSD, including the amygdala, the medial prefrontal, dorsolateral prefrontal, anterior cingulate cortex, and parietal cortex. Sex‐specific effects were also reported, especially following the administration of oxytocin.
Functional brain changes after oxytocin administration
At rest, oxytocin administration was associated with changes in functional connectivity between frontal (e.g., vmPFC) and limbic and affective regions (e.g., amygdala). Normalization (increase) of the connectivity between the vmPFC and the amygdala, especially its centromedial nucleus, was evident in male, but not female, patients with PTSD (Koch et al., ref. 2016). This was further evident following exposure to a trauma‐related script (Frijling et al., ref. 2016). Based on the amygdalo‐centric model of PTSD (Rauch et al., ref. 2006), this increased connectivity may reflect increased top–down prefrontal control over the fear response. Interestingly, oxytocin administration was also associated with reduced connectivity strength between dorsal (mostly cognitive) and ventral (mostly affective) attentional networks (Crum et al., ref. 2021), suggesting that this pharmacological agent may help reduce focus on affective inner thoughts.
During the performance of cognitive tasks, oxytocin administration was associated with changes in activation in brain regions critical for the tasks studied. During the performance of a working memory task, oxytocin administration was associated with increased connectivity between the left dorsolateral prefrontal and anterior cingulate cortices, with greater effects in people diagnosed with PTSD compared to TEC (Flanagan et al., ref. 2018). The direction of effect observed between these two regions was not consistent with normalization of the aberrant patterns of connectivity in PTSD, suggesting that oxytocin‐related enhanced recruitment of this circuit may reflect some compensatory mechanisms to adequately perform the task and may not necessarily reflect increased cognitive efficiency or performance monitoring. During the performance of emotional tasks, results were inconsistent: Administration of oxytocin (compared to placebo) was associated with decreased amygdala activity in a group of police officers (Koch et al., ref. 2016), while associated with increased reactivity of the right basolateral amygdala in a group of acute civilian PTSD patients (Frijling et al., ref. 2016). This discrepancy may be related to the fact that police officers may have been repeatedly exposed to more stressful, if not traumatic events, than the group of acute PTSD patients. Sensitization of the stress system to repeated stress exposure might impact the baseline levels of amygdala reactivity and influence the way oxytocin modulates it. This explanation remains speculative, and future studies including groups of PTSD patients with different levels of basal stress and trauma exposure may help understanding how oxytocin administration may differently influence emotional processing. This is critical to provide the most efficient intervention to people suffering from PTSD.
Other studies have reported enhanced activation in striatal regions following oxytocin administration when performing rewarding tasks, such as social or monetary incentive delay task (Nawijn et al., ref. 2016, 2017). Although oxytocin was associated with increased putamen, anterior cingulate and insular activation independently of the group during reward and loss anticipation, increased putamen and insular activations were specifically observed in PTSD, and decreased in HCs, when social reward feedback was sought after. These findings may indicate that reward evaluation in PTSD may be facilitated by oxytocin administration enhancing motivational processing. However, the impact of oxytocin on brain function during these tasks is highly sensitive to sex, baseline levels of oxytocin system function, and potentially the type of traumatic event and chronicity of PTSD (Olff et al., ref. 2013). Finally, when performing a distraction task (Koch et al., ref. 2019), oxytocin administration was found to modulate thalamic activation, and connectivity with the amygdala, suggesting that oxytocin administration can enhance neural emotional control in PTSD. As suggested by the authors, intranasal oxytocin administration could be used as an enhancer for the changes in brain function produced by first‐line psychological approach for PTSD, such as cognitive‐behavioral therapy. This will need to be further tested in future trials. If these effects are confirmed, the association of oxytocin and CBT may provide important new avenues for the treatment of PTSD.
Functional changes after hydrocortisone administration
Effects of other pharmacological agents in PTSD are less evident. For instance, hydrocortisone administration was not associated with any changes in brain function, either at rest or during the performance of an autobiographical memory task (Metz et al., ref. 2019). The relatively small sample size of these studies may have limited the power to detect more subtle effects. It may also be the case that hydrocortisone may have long‐term effects, not captured in the present trials. Importantly, only two clinical trials using hydrocortisone have been published and met the inclusion criteria for the present systematic review. This strongly limits our understanding of the benefits of hydrocortisone as an alternative therapeutic approach or for PTSD. It would be important to replicate and confirm the lack of effects in larger populations, potentially in groups of patients exposed to different types of trauma. Larger samples allowing the investigation of sex‐specific effects of hydrocortisone on brain function are also warranted, as sex‐specific effects of PTSD on the stress system, especially on cortisol production and regulation, are now well established (Meewisse et al., ref. 2007).
Functional changes after delta‐9‐tetrahydrocannabinol (THC) administration
From the limited number of studies included in this systematic review, the administration of THC was associated with reduced amygdala reactivity to threat and enhanced cortico‐limbic activation during extinction learning (Rabinak et al., ref. 2020; Zabik et al., ref. 2023). According to the authors, these results indicate that the administration of an acute low dose of THC may help facilitate successful emotional regulation and self‐referential processing (Pacitto et al., ref. 2022). In addition, stronger top–down regulation from the vmPFC toward the amygdala during extinction learning in people with PTSD, relative to TEC (Zabik et al., ref. 2023), indicates that THC administration may enhance successful fear reduction and successful extinction learning (Milad & Quirk, ref. 2002). As noted, the limited number of studies, using the same dataset, prevents a clear understanding of the therapeutic effects of THC in PTSD. However, early evidence suggests that a low dose of THC may contribute to the fear of extinction if administered early posttrauma exposure. The effects of higher doses of THC or combinations of THC and varied levels of cannabidiol remain unclear and understudied. Future clinical trials are needed to better determine if THC, in conjunction with other cannabinoids or alone, at low or higher dose, in combination with psychotherapies or alone, may be an ideal approach to prevent the development of PTSD.
Limitations
This systematic review has several limitations. First, the present systematic review only focused on fMRI studies, excluding alternative methods to investigate changes in brain function (e.g., arterial spin labeling, positron emission tomography, single‐photon emission computerized tomography, and electroencephalography) or morphology. Although out of the scope of this systematic review, we cannot completely rule out that the use of some of these alternative pharmacological agents may have induced subtle morphological changes during the course of the presented clinical trials. In addition, the interpretations of the findings mentioned above are limited by the number of studies included and the heterogeneous research methods used, including the types of tasks, as well as the way drugs were administered (e.g., intranasal vs. oral administration). Other recent studies using ketamine (e.g., [Carhart‐Harris et al., ref. 2014; Norbury et al., ref. 2021], psychedelics [Singleton et al., ref. 2022], propranolol [Mahabir et al., ref. 2015], tolcapone [an inhibitor of the degradation of dopamine by catechol‐O‐methyl transferase] [Westphal et al., ref. 2021] or aprepitant [a neurokini‐1 receptor antagonist] [Kwako et al., ref. 2015]) were not included as they did not compared patients with PTSD to groups of TEC or HCs. Furthermore, differences in symptoms severity, trauma types, and age of trauma onset were not considered. The results reported here suggest strong sex‐specific effects of oxytocin, and not all included studies attempted to investigate these effects. Finally, the included studies were, in general, of small sample sizes, limiting their replicability and generalizability.
CONCLUSIONS
Despite the heterogeneity of methods and pharmacological agents used, the present systematic review of clinical trials has identified preliminary evidence for the normalization of brain function following the administration of pharmacological agents. Oxytocin and THC administration were overall associated with normalized amygdala function and amygdala–mPFC connectivity, as well as critical regions for cognitive processes studied (e.g., dmPFC in working memory, putamen in reward, hippocampus in fear condition, and extinction), whereas hydrocortisone was associated with no significant functional brain changes. Interpretation of the results from this systematic review needs to be cautious due to the limited number of clinical trials of small sample sizes included. Further clinical trials are necessary to better understand the neurobiological mechanisms and effects of these and other alternative pharmacological agents on brain function and morphology, alone or in combination with other approaches (e.g., psychotherapies), to benefit people suffering from PTSD.
AUTHOR CONTRIBUTIONS
Shahab Lotfinia: Conceptualization; data curation; formal analysis; investigation; methodology; project administration; resources and writing—original draft. Amin Afshar: Data curation; formal analysis; investigation; resources; writing—review and editing. Aram Yaseri: Data curation; formal analysis; investigation; resources; writing—review and editing. Miranda Olff: Methodology; project administration; supervision; writing—review and editing. Yann Quidé: Investigation; methodology; supervision; writing—review and editing.
CONFLICT OF INTEREST STATEMENT
The authors declare that they have no conflicts of interest.
FUNDING INFORMATION
No funding was received.
PEER REVIEW
The peer review history for this article is available at https://publons.com/publon/10.1002/brb3.3292.
References
- Psychopharmacological treatment in PTSD: A critical review.. Journal of Psychiatric Research,, 2002. [DOI | PubMed]
- Prefrontal cortex, amygdala, and threat processing: Implications for PTSD.. Neuropsychopharmacology,, 2022. [DOI | PubMed]
- American Psychiatric Association (APA) . (1994). Diagnostic and statistical manual of mental disorders (4th ed.). American Psychiatric Association.
- American Psychiatric Association (APA) . (2000). Diagnostic and statistical manual of mental disorders (4th ed.). American Psychiatric Association.
- American Psychiatric Association(APA) . (2013). Diagnostic and statistical manual of mental disorders: DSM‐5™ (5th ed.). American Psychiatric Association.
- A meta‐analytic review of cognitive processing therapy for adults with posttraumatic stress disorder.. Cognitive Behaviour Therapy,, 2019. [DOI | PubMed]
- Prevention and treatment of PTSD: The current evidence base.. European Journal of Psychotraumatology,, 2021. [DOI | PubMed]
- Immunohistochemical localization of oxytocin receptors in human brain.. Neuroscience,, 2013. [DOI | PubMed]
- The epidemiology of posttraumatic stress disorder: What is the extent of the problem?. Journal of Clinical Psychiatry,, 2001
- A dual representation theory of posttraumatic stress disorder.. Psychological Review,, 1996. [DOI | PubMed]
- Risk factors for DSM‐III‐R posttraumatic stress disorder: Findings from the national comorbidity survey.. American Journal of Epidemiology,, 1998. [DOI | PubMed]
- The effects of acutely administered 3,4‐methylenedioxymethamphetamine on spontaneous brain function in healthy volunteers measured with arterial spin labeling and blood oxygen level‐dependent resting state functional connectivity.. Biological Psychiatry,, 2015. [DOI | PubMed]
- The effect of acutely administered MDMA on subjective and BOLD‐fMRI responses to favourite and worst autobiographical memories.. The International Journal of Neuropsychopharmacology,, 2014. [DOI | PubMed]
- Ketamine modulates hippocampal neurogenesis and pro‐inflammatory cytokines but not stressor induced neurochemical changes.. Neuropharmacology,, 2017. [DOI | PubMed]
- Oxytocin, PTSD, and sexual abuse are associated with attention network intrinsic functional connectivity.. Psychiatry Research: Neuroimaging,, 2021. [DOI | PubMed]
- Pharmacological enhancement of exposure‐based treatment in PTSD: A qualitative review.. European Journal of Psychotraumatology,, 2013
- Glucocorticoids and the regulation of memory in health and disease.. Frontiers in Neuroendocrinology,, 2009. [PubMed]
- Glucocorticoid‐induced impairment of declarative memory retrieval is associated with reduced blood flow in the medial temporal lobe.. European Journal of Neuroscience,, 2003. [PubMed]
- Signaling pathways underlying the rapid antidepressant actions of ketamine.. Neuropharmacology,, 2012. [DOI | PubMed]
- A cognitive model of posttraumatic stress disorder.. Behaviour Research and Therapy,, 2000. [DOI | PubMed]
- Increased neural response to trauma scripts in posttraumatic stress disorder following paroxetine treatment: A pilot study.. Neuroscience Letters,, 2011. [DOI | PubMed]
- Oxytocin and vasopressin effects on the neural response to social cooperation are modulated by sex in humans.. Brain Imaging and Behavior,, 2015. [DOI | PubMed]
- Brain function in a patient with torture related post‐traumatic stress disorder before and after fluoxetine treatment: A positron emission tomography provocation study.. Neuroscience Letters,, 2001. [DOI | PubMed]
- Effects of oxytocin on working memory and executive control system connectivity in posttraumatic stress disorder.. Experimental and Clinical Psychopharmacology,, 2018. [DOI | PubMed]
- Impact of oxytocin on the neural correlates of fearful face processing in PTSD related to childhood trauma.. European Journal of Psychotraumatology,, 2019. [DOI | PubMed]
- Emotional processing of fear: Exposure to corrective information.. Psychological Bulletin,, 1986. [DOI | PubMed]
- Intranasal oxytocin affects amygdala functional connectivity after trauma script‐driven imagery in distressed recently trauma‐exposed individuals.. Neuropsychopharmacology,, 2016a. [DOI | PubMed]
- Effects of intranasal oxytocin on amygdala reactivity to emotional faces in recently trauma‐exposed individuals.. Social Cognitive and Affective Neuroscience,, 2016b. [DOI | PubMed]
- Smaller hippocampal volume predicts pathologic vulnerability to psychological trauma.. Nature Neuroscience,, 2002. [DOI | PubMed]
- Default‐mode activity during a passive sensory task: Uncoupled from deactivation but impacting activation.. Journal of Cognitive Neuroscience,, 2004. [DOI | PubMed]
- A general approach‐avoidance hypothesis of oxytocin: Accounting for social and non‐social effects of oxytocin.. Neuroscience and Biobehavioral Reviews,, 2014. [DOI | PubMed]
- Integrating endocannabinoid signaling and cannabinoids into the biology and treatment of posttraumatic stress disorder.. Neuropsychopharmacology,, 2018. [DOI | PubMed]
- Pharmacological therapy for post‐traumatic stress disorder: A systematic review and meta‐analysis of monotherapy, augmentation and head‐to‐head approaches.. European Journal of Psychotraumatology,, 2021. [DOI | PubMed]
- Effects of intranasal oxytocin on distraction as emotion regulation strategy in patients with post‐traumatic stress disorder.. European Neuropsychopharmacology,, 2019. [PubMed]
- Intranasal oxytocin administration dampens amygdala reactivity towards emotional faces in male and female PTSD patients.. Neuropsychopharmacology,, 2016a. [DOI | PubMed]
- Intranasal oxytocin normalizes amygdala functional connectivity in posttraumatic stress disorder.. Neuropsychopharmacology,, 2016b. [DOI | PubMed]
- Effects of intranasal oxytocin on distraction as emotion regulation strategy in patients with post‐traumatic stress disorder.. Eur Neuropsychopharmacol,, 2018. [PubMed]
- How administration of the beta‐blocker propranolol before extinction can prevent the return of fear.. Neuropsychopharmacology,, 2016. [DOI | PubMed]
- The neurokinin‐1 receptor antagonist aprepitant in co‐morbid alcohol dependence and posttraumatic stress disorder: A human experimental study.. Psychopharmacology,, 2015. [DOI | PubMed]
- The functional neuroanatomy of PTSD: A critical review.. Progress in Brain Research,, 2007. [DOI]
- Smaller hippocampal volume in posttraumatic stress disorder: A multisite ENIGMA‐PGC study: Subcortical volumetry results from posttraumatic stress disorder consortia.. Biological Psychiatry,, 2018. [DOI | PubMed]
- Emotional face processing in post‐traumatic stress disorder after reconsolidation impairment using propranolol: A pilot fMRI study.. Journal of Anxiety Disorders,, 2015. [DOI | PubMed]
- Does trauma‐focused psychotherapy change the brain? A systematic review of neural correlates of therapeutic gains in PTSD.. European Journal of Psychotraumatology,, 2021. [DOI | PubMed]
- Targeting the endocannabinoid system in the treatment of posttraumatic stress disorder: A promising case of preclinical‐clinical translation?. Biological Psychiatry,, 2022. [DOI | PubMed]
- Neurobiological and systemic effects of chronic stress.. Chronic Stress (Thousand Oaks),, 2017. [PubMed]
- Subthreshold posttraumatic stress disorder in the world health organization world mental health surveys.. Biological Psychiatry,, 2015. [DOI | PubMed]
- Cortisol and post‐traumatic stress disorder in adults: Systematic review and meta‐analysis.. British Journal of Psychiatry,, 2007. [DOI]
- Effects of hydrocortisone on autobiographical memory retrieval in patients with posttraumatic stress disorder and borderline personality disorder: The role of childhood trauma.. Neuropsychopharmacology,, 2019. [DOI | PubMed]
- Resting‐state functional connectivity after hydrocortisone administration in patients with post‐traumatic stress disorder and borderline personality disorder.. European Neuropsychopharmacology,, 2019. [DOI | PubMed]
- Resting‐state functional connectivity after hydrocortisone administration in patients with post‐traumatic stress disorder and borderline personality disorder.. Eur Neuropsychopharmacol,, 2018
- Neurons in medial prefrontal cortex signal memory for fear extinction.. Nature,, 2002. [DOI | PubMed]
- The safety and efficacy of +/−3,4‐methylenedioxymethamphetamine‐assisted psychotherapy in subjects with chronic, treatment‐resistant posttraumatic stress disorder: The first randomized controlled pilot study.. Journal of Psychopharmacology,, 2011. [DOI | PubMed]
- Intranasal oxytocin enhances neural processing of monetary reward and loss in post‐traumatic stress disorder and traumatized controls.. Psychoneuroendocrinology,, 2016. [DOI | PubMed]
- Intranasal oxytocin increases neural responses to social reward in post‐traumatic stress disorder.. Social Cognitive and Affective Neuroscience,, 2017. [DOI | PubMed]
- Intranasal oxytocin enhances neural processing of monetary reward and loss in post‐traumatic stress disorder and traumatized controls.. Psychoneuroendocrinology,, 2015
- Neuroimaging correlates and predictors of response to repeated‐dose intravenous ketamine in PTSD: Preliminary evidence.. Neuropsychopharmacology,, 2021. [DOI | PubMed]
- The role of oxytocin in social bonding, stress regulation and mental health: An update on the moderating effects of context and interindividual differences.. Psychoneuroendocrinology,, 2013. [DOI | PubMed]
- A psychobiological rationale for oxytocin in the treatment of posttraumatic stress disorder.. CNS Spectrums,, 2010. [DOI | PubMed]
- Cannabinoid modulation of brain activation during volitional regulation of negative affect in trauma‐exposed adults.. Neuropharmacology,, 2022. [DOI | PubMed]
- The PRISMA 2020 statement: An updated guideline for reporting systematic reviews.. The BMJ,, 2021. [DOI | PubMed]
- Neurocircuitry models of posttraumatic stress disorder and beyond: A meta‐analysis of functional neuroimaging studies.. Neuroscience and Biobehavioral Reviews,, 2012. [DOI | PubMed]
- Smaller hippocampal volume following sexual assault in women is associated with post‐traumatic stress disorder.. Acta Psychiatrica Scandinavica,, 2018. [DOI | PubMed]
- Differences between effects of psychological versus pharmacological treatments on functional and morphological brain alterations in anxiety disorders and major depressive disorder: A systematic review.. Neuroscience and Biobehavioral Reviews,, 2012. [DOI | PubMed]
- Aberrant intrinsic connectivity in women victims of sexual assault.. Brain Imaging and Behavior,, 2021. [DOI | PubMed]
- Cannabinoid modulation of prefrontal‐limbic activation during fear extinction learning and recall in humans.. Neurobiology of Learning and Memory,, 2014. [DOI | PubMed]
- Cannabinoid modulation of corticolimbic activation to threat in trauma‐exposed adults: A preliminary study.. Psychopharmacology,, 2020. [DOI | PubMed]
- The potential of ketamine for posttraumatic stress disorder: A review of clinical evidence.. Therapeutic Advances in Psychopharmacology,, 2023. [DOI | PubMed]
- Neurocircuitry models of posttraumatic stress disorder and extinction: Human neuroimaging research—Past, present, and future.. Biological Psychiatry,, 2006. [DOI | PubMed]
- The DSM‐5: Classification and criteria changes.. World Psychiatry,, 2013. [DOI | PubMed]
- Sex differences in the neural and behavioral response to intranasal oxytocin and vasopressin during human social interaction.. Psychoneuroendocrinology,, 2014. [DOI | PubMed]
- The endocannabinoid system in anxiety, fear memory and habituation.. Journal of Psychopharmacology,, 2012. [DOI | PubMed]
- In search of the trauma memory: A meta‐analysis of functional neuroimaging studies of symptom provocation in posttraumatic stress disorder (PTSD).. PLoS ONE,, 2013. [DOI | PubMed]
- Frontal lobe circuitry in posttraumatic stress disorder.. Chronic Stress (Thousand Oaks),, 2019. [PubMed]
- Pharmacological prevention of post‐traumatic stress disorder and acute stress disorder: A systematic review and meta‐analysis.. The Lancet Psychiatry,, 2015. [DOI | PubMed]
- Altered brain activity and functional connectivity after MDMA‐assisted therapy for post‐traumatic stress disorder.. Front Psychiatry,, 2022. [PubMed]
- Effects of intranasal oxytocin on threat‐and reward‐related functional connectivity in men and women with and without childhood abuse‐related PTSD.. Psychiatry Research: Neuroimaging,, 2021. [DOI | PubMed]
- The role of the hypothalamic‐pituitary‐adrenal axis in neuroendocrine responses to stress.. Dialogues in Clinical Neuroscience,, 2006. [DOI | PubMed]
- RoB 2: A revised tool for assessing risk of bias in randomised trials.. The BMJ,, 2019. [DOI | PubMed]
- Clinical pharmacology in neuroscience drug discovery: Quo vadis?. Current Opinion in Pharmacology,, 2014. [DOI | PubMed]
- Long‐term treatment with paroxetine increases verbal declarative memory and hippocampal volume in posttraumatic stress disorder.. Biological Psychiatry,, 2003. [DOI | PubMed]
- Cortical volume abnormalities in posttraumatic stress disorder: An ENIGMA‐psychiatric genomics consortium PTSD workgroup mega‐analysis.. Molecular Psychiatry,, 2021. [DOI | PubMed]
- Working memory, cortical dopamine tone, and frontoparietal brain recruitment in post‐traumatic stress disorder: A randomized controlled trial.. Translational Psychiatry,, 2021. [DOI | PubMed]
- Decreased anterior cingulate volume in combat‐related PTSD.. Biological Psychiatry,, 2006. [DOI | PubMed]
- World Health Organization . (1992). The ICD‐10 classification of mental and behavioural disorders: Clinical descriptions and diagnostic guidelines. World Health Organization.
- P.3.026 Intranasal oxytocin in patients with post traumatic stress disorder: A single dose, pilot double blind crossover study.. European Neuropsychopharmacology,, 2010. [DOI]
- Cortisol augmentation of a psychological treatment for warfighters with posttraumatic stress disorder: Randomized trial showing improved treatment retention and outcome.. Psychoneuroendocrinology,, 2015. [DOI | PubMed]
- Cannabinoid modulation of corticolimbic activation during extinction learning and fear renewal in adults with posttraumatic stress disorder.. Neurobiology of Learning and Memory,, 2023. [DOI | PubMed]
