Modulation of the endocannabinoid system reduces inflammatory signalling in canine mammary carcinoma cells
Abstract
Background:
Canine mammary carcinoma (CMC) is characterised by a chronic inflammatory microenvironment resembling human breast cancer; however, the upstream regulatory mechanisms driving this phenotype remain unclear. The endocannabinoid system (ECS) has emerged as a potential modulator of inflammation and tumour biology. This study investigated the role of the ECS in CMC and evaluated the anti‐inflammatory effects of cannabidiol (CBD).
Methods:
Primary cell cultures were established from surgically excised CMC tissues, with matched normal mammary epithelium used as controls. Basal mRNA expression of ECS‐related receptors (CB1, CB2, transient receptor potential vanilloid 1 [TRPV1], G‐protein‐coupled receptor 55 [GPR55] and peroxisome proliferator‐activated receptor alpha [PPAR‐α]) and inflammatory mediators (COX‐1, COX‐2, interleukin [IL]‐4, IL‐6, IL‐33, IL‐17A, tumour necrosis factor‐alpha [TNF‐α] and LCN2) was assessed by reverse transcription quantitative polymerase chain reaction. Cytokine secretion (IL‐6, IL‐8, TNF‐α and IL‐17A) was quantified by enzyme‐linked immunosorbent assay. Cell viability assays were performed to determine the 24‐h IC50 of CBD (32 µM), and sub‐cytotoxic concentrations (3, 10 and 20 µM) were subsequently applied for 24 h.
Results:
Canine mammary carcinoma‐derived cells exhibited significant overexpression of ECS receptors (CB1, CB2, TRPV1, GPR55 and PPAR‐α) compared to normal controls. These cells also showed increased secretion of pro‐inflammatory cytokines, including IL‐6, IL‐8, TNF‐α and IL‐17A. Treatment with CBD at 10–20 µM significantly downregulated key inflammatory genes, particularly COX‐2, IL‐6 and TNF‐α, and reduced corresponding cytokine release without compromising cell viability.
Conclusion:
The ECS is upregulated in CMC and appears to contribute to the inflammatory tumour microenvironment. Cannabidiol effectively attenuates this inflammatory phenotype at sub‐cytotoxic concentrations, supporting its potential as a therapeutic agent in CMC.
Article type: Research Article
Keywords: cannabidiol, dogs, endocannabinoid receptors, inflammation, mammary tumour, PPAR‐α
Affiliations: Department of Veterinary Science University of Messina Messina Italy; Department of Chemical Biological Pharmaceutical and Environmental Science University of Messina Messina Italy; Link University Rome Campus Italy Rome Italy
License: © 2026 The Author(s). Veterinary Record Open published by John Wiley & Sons Ltd on behalf of British Veterinary Association. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/vro2.70034 | PMC: PMC13128961
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (3.2 MB)
INTRODUCTION
The canine mammary gland is a tubuloalveolar structure comprising stroma, parenchyma, vessels, ducts and nerves.ref. vro270034-bib-0001 The posterior mammary glands are more frequently affected than the anterior glands and may present with one or more nodules.ref. vro270034-bib-0002 Breast tumours are among the most common cancers in dogs and account for approximately 25%‒30% of all the tumours recorded in dogs.ref. vro270034-bib-0003 Dogs can develop either benign or malignant tumours. Malignant tumours tend to be locally invasive, often fixed to the overlying skin or underlying tissues, and generally grow rapidly.ref. vro270034-bib-0004 In contrast, benign tumours are usually small, well circumscribed and slow growing.ref. vro270034-bib-0002, ref. vro270034-bib-0005 Metastases most commonly occur in regional lymph nodes and lungs, although the brain, bones and various abdominal organs can also be involved.ref. vro270034-bib-0006 Recent research has highlighted the roles of ovarian hormones and certain growth factors in the aetiology of breast tumours.ref. vro270034-bib-0006
Dogs are valuable models for studying malignancies such as lymphoma and mammary tumours because their cancers develop spontaneously and share biological features with human disease.ref. vro270034-bib-0007 Inflammation plays a crucial role in this process, and rather than combating tumourigenesis, it often potentiates it.ref. vro270034-bib-0008 Inflammation leads to cancer through two pathways: an intrinsic pathway driven by genetic changes (e.g., oncogene activation) and an extrinsic pathway caused by chronic inflammation at specific sites.ref. vro270034-bib-0009 Both pathways converge to create an inflammatory tumour microenvironment that promotes malignant transformation.ref. vro270034-bib-0010, ref. vro270034-bib-0011 Immune cells are continually recruited and activated, thereby perpetuating this inflammatory milieu and fostering tumour development.ref. vro270034-bib-0012 Beyond these well‐characterised mechanisms, recent studies have identified endogenous regulatory systems as key modulators of tumour progression and inflammation.
In particular, the endocannabinoid system (ECS) has emerged as a critical modulator of the chronic inflammatory milieu that drives tumour progression.ref. vro270034-bib-0013 Previous studies have indicated that endocannabinoid levels rise in proportion to disease severity in breast cancer, suggesting a direct link to tumour progression.ref. vro270034-bib-0014 The ECS comprises natural endocannabinoids and their receptors, CB1 and CB2.ref. vro270034-bib-0015 When endocannabinoids bind to these receptors, they activate G‐proteins that regulate several intracellular signalling pathways, including the MAPK/ERK, PI3K/Akt and cAMP‐dependent pathways.ref. vro270034-bib-0016 Modulation of these pathways can lead to reduced cell proliferation, induction of apoptosis and autophagy, inhibition of angiogenesis, and decreased cell migration and invasion, ultimately contributing to the suppression of tumour progression.ref. vro270034-bib-0017 Among the exogenous cannabinoids, cannabidiol (CBD) has garnered significant attention for its ability to interfere with pro‐tumourigenic inflammatory loops and inhibit cancer cell proliferation through interactions with multiple molecular targets, including CB1 and CB2 receptors, transient receptor potential vanilloid (TRPV) channels, G‐protein‐coupled receptor 55 (GPR55) and peroxisome proliferator‐activated receptors (PPARs).ref. vro270034-bib-0018 Cannabinoids can thus affect survival, proliferation, invasion and metastasis in tumour cells, exhibiting anti‐proliferative, pro‐apoptotic and anti‐angiogenic properties.ref. vro270034-bib-0019, ref. vro270034-bib-0020 Mechanistically, cannabinoids have been shown to induce autophagy and apoptosis, as well as to inhibit vascular endothelial growth factor. Moreover, the ECS exerts anti‐inflammatory and immunomodulatory effects, contributing to its multi‐level action.
Based on these insights, we aimed to investigate whether targeting the ECS using cannabinoids might induce cancer cell death.ref. vro270034-bib-0021 In addition to the classical cannabinoid receptors (CB1 and CB2), this study investigated the role of other ECS’s receptor, including the peroxisome proliferator‐activated receptor alpha (PPAR‐α). PPAR‐α is a ligand‐activated transcription factor widely recognised for its potent anti‐inflammatory properties and its crucial involvement in regulating cell cycle progression and apoptosis in various malignancies, including breast cancer.ref. vro270034-bib-0022, ref. vro270034-bib-0023 Notably, in oncological contexts, PPAR‐α is often co‐expressed or cross‐modulated with the ECS, suggesting a complex interplay that may influence tumour cell fate and the local inflammatory microenvironment.ref. vro270034-bib-0021, ref. vro270034-bib-0024, ref. vro270034-bib-0025, ref. vro270034-bib-0026 Based on these findings, we hypothesised that the ECS plays a key role in sustaining the aggressive inflammatory phenotype of canine mammary carcinoma (CMC), and that targeting this system with CBD may mitigate these pro‐tumourigenic processes. Consequently, this study aimed to characterise the expression of ECS receptors, including the involvement of ECS’s receptors such as PPAR‐α, and inflammatory markers in primary CMC cell cultures.
MATERIALS AND METHODS
Samples
Mammary gland lesions were surgically excised for therapeutic purposes from privately owned dogs at the Veterinary Teaching Hospital of Messina. Surplus tissue was donated to the study after informed consent was obtained from each owner. Three intact female dogs (Beagle, 6 years; Rottweiler, 10 years; and German Shepherd dog, 13 years) met the inclusion criteria: (i) primary tumour, (ii) no prior chemotherapy or anti‐inflammatory medication, and (iii) histological confirmation of mixed‐type carcinoma with focal comedocarcinoma (WHO classification, grade II), verified independently by two board‐certified pathologists. Immediately after excision, specimens were divided: one fragment was fixed in 10% neutral‐buffered formalin (<30 min post‐surgery) for histopathology, while a second fragment was placed on ice in Dulbecco’s phosphate‐buffered saline (PBS) with penicillin (100 IU/mL) and streptomycin (100 µg/mL) and processed for primary cell isolation within 2 h. Control tissue consisted of macroscopically normal mammary epithelium from the adjacent gland of the same dog; when unavailable, histologically normal tissue excised for unrelated reasons in age‐matched bitches was used. All procedures were conducted in accordance with the European Directive 2010/63/EU. Samples were anonymised and coded for downstream analyses.
Cannabidiol
Cannabidiol (C‐045‐1ML, Cerilliant Sigma‒Aldrich) was dissolved in methanol to prepare the stock solution (1.0 mg/mL). For all experimental applications, final dilutions were adjusted based on the tests performed. The prepared CBD was then stored at 4°C.
Cell isolation
Surgically excised tumour specimens were immediately placed on ice and trimmed of adipose tissue under aseptic conditions. Tissues were rinsed twice in cold PBS supplemented with antibiotics as previously described, then minced into ∼1 mm3 fragments with sterile scalpels. Tissue fragments were mechanically disaggregated by gentle pressing through a 70 µm cell strainer (Becton Dickinson Falcon) into ice‐cold Dulbecco’s modified Eagle medium (DMEM, Gibco). The suspension was centrifuged (300 ×g, 10 min, 4°C) and the pellet was resuspended in DMEM, supplemented with 10% fetal bovine serum, 2 mM L‐glutamine and antibiotics (100 IU/mL penicillin and 100 µg/mL streptomycin). Cells (1 × 105 cells/cm2) were seeded in 60 mm tissue‐culture dishes (Costar, Corning) and incubated at 37°C in a humidified 5% CO2 atmosphere. Medium was changed after 24 h to remove non‐adherent debris and thereafter every 48 h. Upon reaching 80%–90% confluence (≈5–7 days), cells were trypsinised (0.05% trypsin‒EDTA, 5 min, 37°C) and sub‐cultured at a 1:2 ratio; passage 5 cells were used for all downstream assays. Cell viability was 95% or more by trypan‐blue exclusion, and cultures were routinely screened for Mycoplasma contamination with a polymerase chain reaction (PCR)‐based kit (Eurofins).
Histological examination
Tissue fragments were fixed in 10% neutral‐buffered formalin for 24 h at room temperature, routinely processed, and embedded in paraffin. Serial 5 µm sections were cut on a rotary microtome (Leica Microsystems). Paraffin‐embedded tissue sections were first deparaffinised in xylene through three changes of 5 min each. Sections were then rehydrated through a graded ethanol series (100%, 95%, 70% and 50%), with 10 min in 100% and 95% ethanol, followed by 5 min in 70% and 50% ethanol, and finally rinsed in distilled water for 5 min.
Sections were stained with haematoxylin for 5 min and subsequently washed in running tap water until the excess stain was removed. The slides were then counterstained with eosin for 3 min.
After staining, sections were dehydrated through graded ethanol, including rapid washes followed by 95% ethanol for 1 min and two changes of 100% ethanol for 5 min each. Finally, sections were cleared in xylene for 15 min, mounted with a permanent mounting medium, and coverslipped. Histological evaluation was performed independently by two board‐certified veterinary pathologists using a Leica DM2500 microscope (Leica Microsystems).
Immunofluorescence
Cells (5 × 104 cells/well) were seeded directly onto the optically clear bottoms of standard 12‐well culture plates and grown to 70%–80% confluence. Monolayers were rinsed twice with PBS and fixed in 4% paraformaldehyde/PBS for 15 min at room temperature, permeabilised with 0.1% Triton X‐100 (5 min) and blocked for 1 h in PBS containing 5% bovine serum albumin. Primary antibodies—PPAR‐α (rabbit, ab8934, 1:200) or β‐tubulin‐Alexa Fluor 488 (mouse, MA5‐16308, 1:400) previously validated in dog ref. vro270034-bib-0027—were applied overnight at 4°C. After three PBS washes, wells were incubated for 1 h at room temperature with species‐specific secondary antibodies (goat anti‐rabbit‐Texas red both 1:500 #T‐2767, Thermo Fisher), counterstained with 4′,6‐diamidino‐2‐phenylindole (1 µg/mL, 5 min) and maintained in PBS for imaging. Fluorescence micrographs were captured in‐plate with an EVOS M5000 system (20× and 40× objectives) using identical exposure settings for all conditions.
Reverse transcription quantitative polymerase chain reaction
Total RNA was extracted from each experimental group with the RNeasy Mini Kit (Qiagen, Cat. #74104) according to the manufacturer’s protocols.ref. vro270034-bib-0028 After quantifying the RNA by nanodrop, we performed c‐DNA synthesis using the iScript cDNA Synthesis Kit (Bio‐Rad, Cat. #1708891) according to the manufacturer’s protocols.ref. vro270034-bib-0029 Polymerase chain reaction was subsequently performed using quantitative PCR (qPCR) was performed on a CFX96 Touch Real‐Time PCR System (Bio‐Rad) with SsoAdvanced Universal SYBR Green Supermix (Bio‐Rad, Cat. #1725271) or Quantitec SYBR Green PCR (Quiagen Cat. #204343). Commercially available validated primers for interleukin (IL)‐33 (QuantiTect, Cat. #QT00897092), LCN2 (Bio‐Rad, Cat. #qCfaCIP0002507), IL‐4 (Bio‐Rad, Cat. #qCfaCIP0015120), IL‐6 (Bio‐Rad, Cat. #10042976), COX1 (Bio‐Rad, Cat. #qCfaCEP0005352), COX2 (Bio‐Rad, Cat. #qCfaCEP0005353), tumour necrosis factor‐alpha (TNF‐α) (Bio‐Rad, Cat. #qCfaCEP0011393), PPAR‐α (Bio‐Rad, Cat. #qCfaCED0030703), TRPV1 (Bio‐Rad, Cat. #qCfaCEP0019089), GPR55 (Bio‐Rad, Cat. #qCfaCED0037760), glyceraldehyde‐3‐phosphate dehydrogenase (GAPDH) (Bio‐Rad, Cat. #qCfaCED0037760) and ACTG1 (Bio‐Rad, Cat. #qCfaCEP0016209) were used. The mRNA level fold change was determined using the 2‒∆∆Ct data analysis method.ref. vro270034-bib-0030 Normalisation was performed using GAPDH as an internal control. The stability of this reference gene in canine mammary gland specimens has been previously validated by Etschmann et al.,ref. vro270034-bib-0031 and its reliability was confirmed in our samples by the consistency of cycle threshold (Ct) values across experimental groups (mean Ct variation < 0.5).
Cell cycle analysis
After 24 h exposure to 33 µM CBD (IC50) or vehicle control, 1 × 106 cells were harvested and stained according to manufacturer’s protocols ref. vro270034-bib-0032 with FxCycle PI/RNase Staining Solution (Invitrogen Thermo Fisher, Cat. #F10797). Samples were acquired on an Attune NxT flow cytometer (Thermo Fisher Scientific).
Enzyme‐linked immunosorbent assay
DuoSet enzyme‐linked immunosorbent assay (ELISA) kits R&D System were used to evaluate cytokines concentration for TNF‐ɑ (Cat. #DY1507), IL‐8 (Cat. #DY1608), IL‐6 (Cat. #DY1609) and IL‐17A (Cat. #DY5848) following the manufacturer’s instructions.ref. vro270034-bib-0028
Statistics
Data represent the mean ± standard error of the mean from three independent biological replicates (n = 3 dogs), each assayed in technical triplicate to ensure experimental consistency. Statistical tests were performed in GraphPad Prism v10.0 (GraphPad Software). Normality of distribution was verified with the Shapiro–Wilk test. Student’s t‐test was employed for comparisons between two groups, while one‐way ANOVA analysis was used for comparisons involving more than two groups with a single variable, followed by Tukey’s test. Statistical significance was determined as a p‐value of less than 0.05.
RESULTS
Histological analysis
Histological examination shown in Figure 1 revealed pleomorphic, malignant epithelial cells associated with a benign myoepithelial component and metaplastic cartilage (which exhibit no atypia) supported by a fibrovascular stroma. Malignant epithelial cells were arranged in tubules; they showed hypochromic nuclei, large single nucleoli and eosinophilic cytoplasm with distinct margins. In some areas, neoplastic cells surrounded necrotic zones containing cellular debris and apoptotic bodies (comedonecrosis). The malignant epithelial cells at the periphery showed a pattern of growth solid or tubulopapillary, moderate anisokaryosis and mitotic activity than 8 per 2.37 mm. Based on the histological features highlighted, it was possible to make a diagnosis of carcinoma mixed type with areas of comedocarcinoma.ref. vro270034-bib-0033
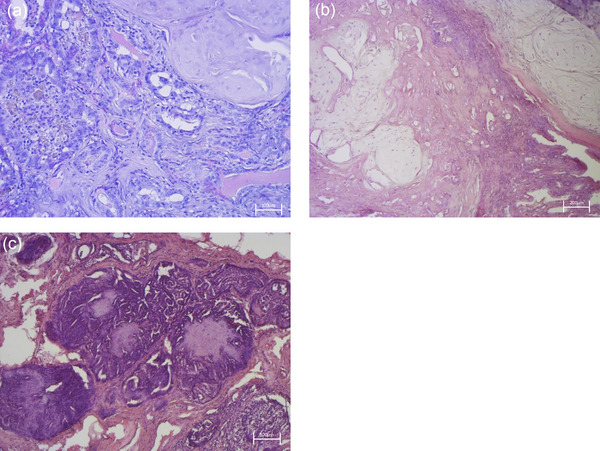
Microscopic morphology of canine mammary primary cell carcinoma
As shown in Figure 2, phase‐contrast microscopy revealed a heterogeneous population comprising spindle‐shaped and polygonal epithelial‐like cells (Figure 2a), consistent with the biphasic nature of the original tumour. This morphological heterogeneity persisted from the first sub‐culture (P1) through late passages (≥P6), while cells maintained stable, rapid proliferation. Immunofluorescence for β‐tubulin—a cytoskeletal marker linked to high‐grade mammary malignancies and showed intense cytoplasmic staining and highlighted mitotic spindles, confirming the high mitotic activity of the culture (Figure 2b‒d).ref. vro270034-bib-0025, ref. vro270034-bib-0034
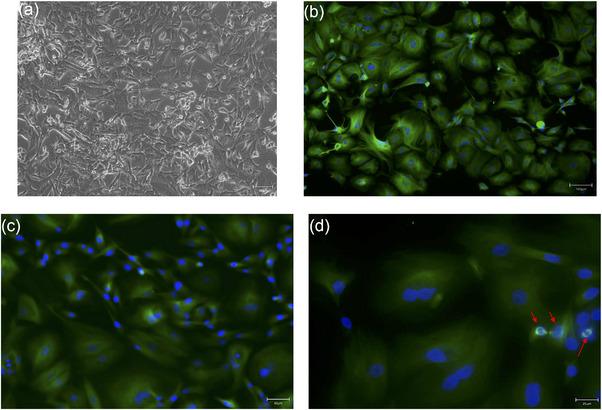
Basal inflammatory profile of primary CMC cell
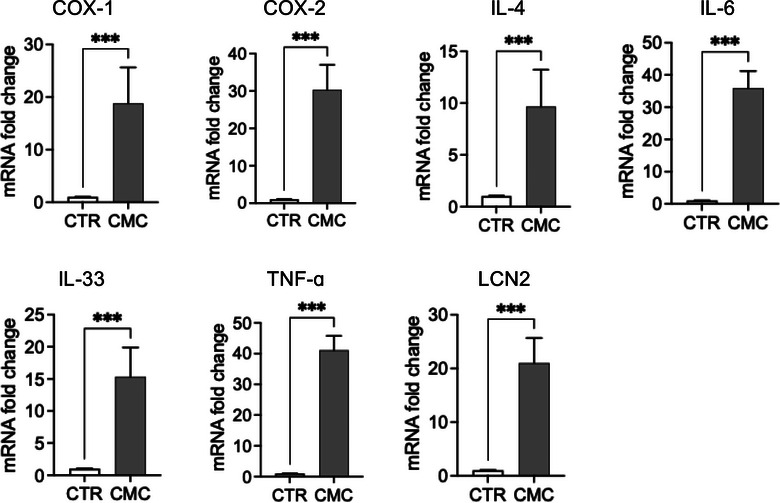
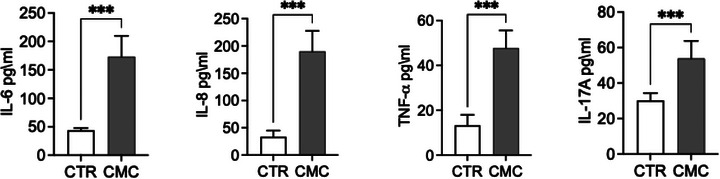
We evaluated the expression of genes involved in inflammatory and immune response pathways, including COX‐1, COX‐2, IL‐4, IL‐6, IL‐33, TNF‐α and LCN2 (Figure 3). Both the constitutive enzyme COX‐1 and its inducible isoform COX‐2 were significantly upregulated in CMC tissues, where they are associated with disease progression and the maintenance of a pro‐inflammatory state. Notably, while COX‐1 expression was significantly increased, COX‐2 exhibited an even greater upregulation, supporting the presence of an active inflammatory phenotype in this tumour type. To further characterise the inflammatory and immune landscape of CMC cells, we analysed the expression of key cytokines, including IL‐4, IL‐6, IL‐33, and TNF‐α, known to orchestrate tumour‐associated inflammation. Interleukin‐6 and TNF‐α, two major mediators of inflammatory signalling and immune modulation, were significantly elevated at the transcriptional level. In addition to these cytokines, IL‐4 recognised for its role in shaping the tumour microenvironment and influencing immune polarisation—was also significantly upregulated in tumour tissues. Furthermore, we observed markedly increased mRNA levels of IL‐33 and LCN2, both implicated in tumour progression through their involvement in immune cell recruitment as well as in the regulation of proliferation, migration and invasion of tumour and mesenchymal cells. Consistent with the transcriptional findings, analysis of culture supernatants revealed significantly higher concentrations of IL‐6, IL‐8, TNF‐α and IL‐17A in primary CMC cultures compared with control mammary epithelial cells (Figure 7a). These data confirm that CMC cells exhibit not only an activated inflammatory gene expression profile but also a corresponding pro‐inflammatory secretory phenotype (Figure 4).
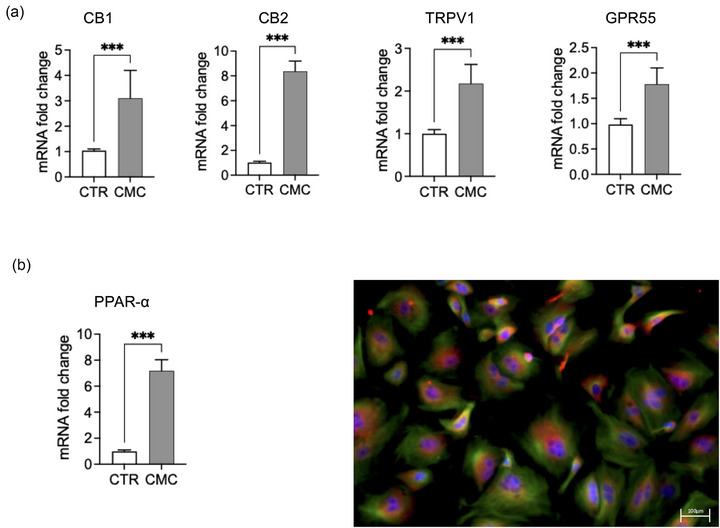
Expression of endocannabinoid‐related receptors
Gene expression analysis revealed a significant upregulation of multiple endocannabinoid‐related receptors in CMC cells compared with matched control mammary epithelium (Figure 5). mRNA levels of CNR1 (CB1) and CNR2 (CB2) were markedly increased in CMC samples. Similarly, the non‐classical cannabinoid‐related receptors TRPV1 and GPR55 showed significant overexpression relative to controls (Figure 5a). In addition, PPAR‐α mRNA expression was significantly elevated in CMC cells (Figure 5b). Immunofluorescence analysis confirmed PPAR‐α protein expression and demonstrated increased nuclear localisation in tumour cells compared to controls, supporting transcriptional activation of this pathway.
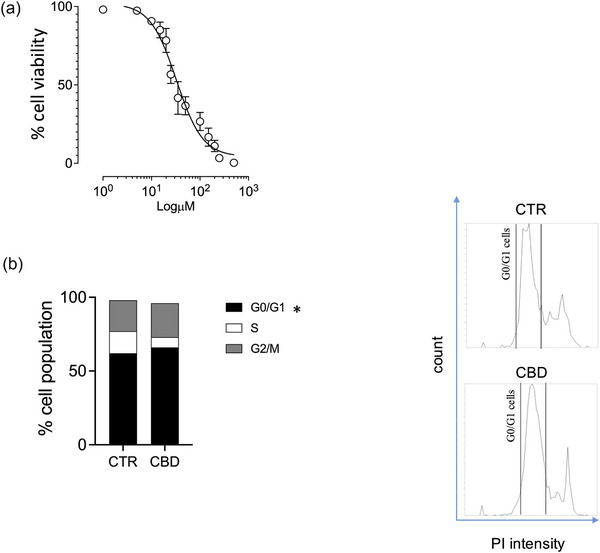
Anti‐proliferative effects of CBD: viability and cell cycle analysis
To characterise the antiproliferative activity of CBD, we first generated a 24‐h concentration–response curve, identifying an IC50 of 32.63 µM (Figure 6a). We then assessed cell cycle distribution after 24 h exposure to 33 µM CBD. Flow‐cytometric analysis of DNA content revealed a significant increase in the G0/G1 fraction compared with vehicle‐treated cells (Figure 6b), consistent with an antiproliferative effect at this concentration.
Cannabidiol attenuates inflammatory gene expression and cytokine production in primary CMC cells
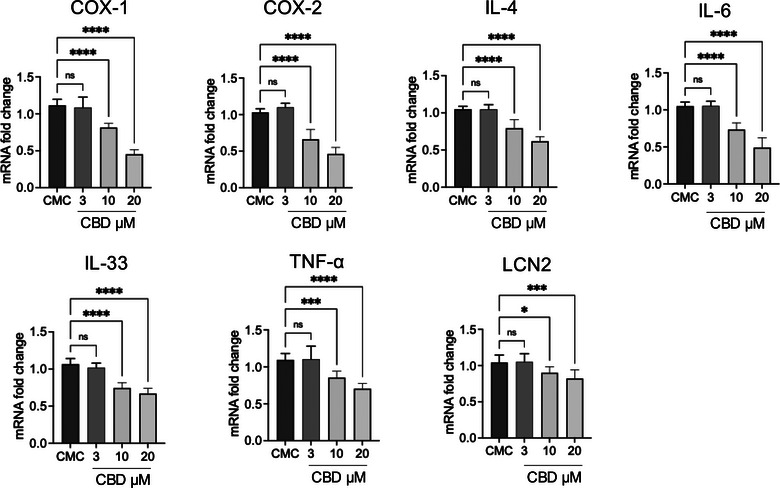
Treatment of primary CMC cultures with CBD resulted in a consistent, dose‐dependent modulation of inflammatory mediators at both the transcriptional and protein levels (Figures 7 and 8). At the mRNA level in Figure 7, exposure to 3 µM CBD did not produce significant changes compared to untreated CMC cells for most targets, indicating that this concentration is largely sub‐threshold for transcriptional modulation. In contrast, 10 µM CBD significantly reduced the expression of COX‐1, COX‐2, IL‐4, IL‐6, IL‐33, TNF‐α and LCN2. The highest concentration tested (20 µM) induced a further and more pronounced downregulation of all analysed genes, with COX‐2, IL‐6 and IL‐33 showing particularly marked reductions.
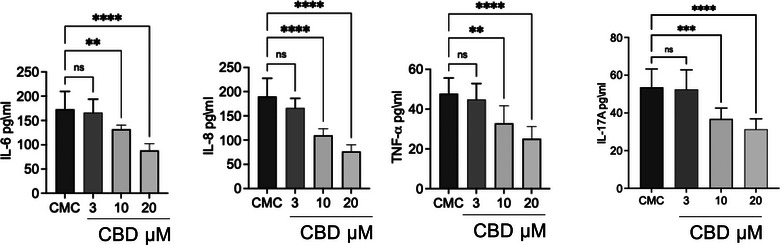
Figure 8, on the other hand, highlights the effect of CBD at different concentrations (3, 10 and 20 µM) on cytokine levels in CMC cells supernatant. Cannabidiol dose dependently reduced the levels of IL‐6, IL‐8 and TNF‐α compared to the CMC group. For IL‐17A, the reduction was visible but not statistically significant for concentrations at 3 and 10 µM, becoming significant only at 20 µM.
Consistent with the gene expression data, ELISA analysis of culture supernatants (Figure 8) revealed a dose‐dependent decrease in cytokine release. While 3 µM CBD did not significantly affect IL‐6, IL‐8, TNF‐α or IL‐17A levels, treatment with 10 µM significantly reduced IL‐6, IL‐8 and TNF‐α concentrations. The strongest inhibitory effect was observed at 20 µM, where all measured cytokines, including IL‐17A, were significantly decreased compared to untreated CMC cells. Notably, IL‐17A required the highest concentration to reach statistical significance, suggesting differential sensitivity among cytokine pathways to CBD modulation.
DISCUSSION
The ECS plays a crucial role in regulating various physiological functions in mammals.ref. vro270034-bib-0035 Furthermore, this system is implicated in several pathological states as it serves as an endogenous regulator for restoring both cellular and tissue homeostasis.ref. vro270034-bib-0036 In fact, its physiological effects are exploited as therapeutic targets in various diseases including cancer, neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease, chronic pain syndromes and inflammatory disorders.ref. vro270034-bib-0037, ref. vro270034-bib-0038, ref. vro270034-bib-0039, ref. vro270034-bib-0040
Recently, the ECS has garnered significant interest in tumourigenesis and cancer therapy.ref. vro270034-bib-0041 As is widely recognised, cancer is a complex disease involving various cellular responses and interactions, including dysregulated cell proliferation and apoptosis, angiogenesis, immune evasion and interactions with the tumour microenvironment.ref. vro270034-bib-0042 One key feature is increased proliferation and loss of cell cycle control, but inflammatory and immune responses also contribute significantly.ref. vro270034-bib-0043
Recent studies have focused on understanding the pathological processes underlying canine mammary tumours, as well as on identifying potential treatments or adjuncts to standard pharmacological approaches.ref. vro270034-bib-0044, ref. vro270034-bib-0045 In this context, cannabinoids attract great interest, not only for their well‐known anti‐inflammatory and immunomodulatory properties but also for their potential role in regulating tumour cell proliferation.ref. vro270034-bib-0046, ref. vro270034-bib-0047 Moreover, their relatively favourable pharmacological profile allows them to be used in combination with conventional chemotherapy, and in some cases, they exhibit synergistic effects, particularly in glioblastoma, breast cancer and pancreatic cancer models.ref. vro270034-bib-0048, ref. vro270034-bib-0049, ref. vro270034-bib-0050
In this study, we examined the role of the ECS in CMC using cells isolated from patients undergoing surgical resection. Histological examination enabled us to identify the specific type of mammary tumour, and the isolated tumour cells stabilised in vitro exhibited phenotypic features consistent with the original histological condition. We then analysed the gene expression of key inflammatory markers, such as COX‐1 and COX‐2, known to be fundamental in shaping the tumour microenvironment through immune cell recruitment and inflammatory responses. Both COX‐1 and COX‐2 were significantly upregulated in tumour tissues. These enzymes are known mediators of inflammation in both human and canine mammary cancers.ref. vro270034-bib-0051, ref. vro270034-bib-0052 Notably, in various tumour types, including breast cancer, COX‐2 has been linked to disease progression and often correlates with tumour invasiveness and aggressiveness in both humans and animals.ref. vro270034-bib-0053 Accordingly, the use of non‐steroidal anti‐inflammatory drugs, especially selective COX‐2 inhibitors, can have beneficial effects in such cancers.ref. vro270034-bib-0054
To study CBD’s anti‐inflammatory properties without the interference of significant cell death, dosages were selected based on preliminary cytotoxicity tests. By using concentrations corresponding to a 20% dilution of the CBD stock solution, we ensured that the effects observed on CMC cells were primarily due to biochemical signalling rather than reduced cell viability. Among the most significant pleiotropic pro‐inflammatory cytokines involved in breast cancer progression are IL‐6 and TNF‐α, both overexpressed in this setting.ref. vro270034-bib-0055 Interleukin‐4, a pleiotropic cytokine produced by T lymphocytes, acts on various cell types (T and B lymphocytes, monocytes, fibroblasts, endothelial cells and macrophages) and contributes to tumour progression and metastasis by promoting cell survival and proliferation, modulating immune responses towards a tumour‐permissive phenotype and activating nuclear factor‐kappa B (NF‐κB) signalling.ref. vro270034-bib-0056, ref. vro270034-bib-0057 Interleukin‐33, a member of the IL‐1 cytokine family, is constitutively expressed in the nuclei of epithelial, endothelial and fibroblast‐like cells.ref. vro270034-bib-0058 Under cellular stress, damage or necrosis, IL‐33 is released into the cytoplasm to function as an alarmin, binding its specific receptor ST2.ref. vro270034-bib-0059 Interleukin‐33 exhibits pleiotropic roles in inflammatory diseases, particularly in cancer.ref. vro270034-bib-0060 Its influence can be either pro‐ or anti‐tumourigenic, depending on tumour type, expression levels, bioactivity and the nature of the inflammatory environment.ref. vro270034-bib-0061 Experimental studies have demonstrated that IL‐33 can directly promote tumourigenesis by enhancing proliferation and colony formation in breast cancer cell lines.ref. vro270034-bib-0032 LCN2 also emerges as a key player, as it is expressed at high levels in carcinoma tissues.ref. vro270034-bib-0062 Over the past two decades, abnormal LCN2 expression has been linked to various pathological conditions, including inflammation and cancer in multiple organs.ref. vro270034-bib-0063 Such findings may prove useful in guiding pharmacological interventions in veterinary oncology and identifying new therapeutic targets. Previous studies have demonstrated that components of the ECS are dysregulated in various cancers and are associated with tumour progression, inflammation and patient prognosis.ref. vro270034-bib-0064, ref. vro270034-bib-0065, ref. vro270034-bib-0066 In this context, our study expands on these findings by characterising the expression of ECS‐related receptors, together with key inflammatory markers in primary CMC cells, thereby providing further insight into the interplay between ECS signalling and tumour‐associated inflammation.ref. vro270034-bib-0018, ref. vro270034-bib-0067
Previous research has indicated that cannabinoid receptors can modulate the growth and development of mammary tumours.ref. vro270034-bib-0068, ref. vro270034-bib-0069 Their expression may change within tumours due to both endogenous ECS regulation and interactions with exogenous cannabinoids.ref. vro270034-bib-0070 Indeed, in breast tumours, CB1 and CB2 receptor levels often differ from those in healthy tissues, influencing how tumour cells respond to cannabinoids.ref. vro270034-bib-0071 Because the ECS has shown anti‐cancer potential,ref. vro270034-bib-0072 we assessed CB1 and CB2 receptor expression in CMC cells. Our results revealed elevated CB1 and CB2 expression relative to healthy tissue controls. Although the present study focused on evaluating the global modulation of the ECS, it is important to consider that the ECS contains numerous receptors, including PPAR‐γ, which play a crucial role in mediating anti‐inflammatory effects, such as inhibition of pro‐inflammatory cytokine production, suppression of NF‐κB signalling and reduction of immune cell activation.ref. vro270034-bib-0073, ref. vro270034-bib-0074 The effect of CBD on this receptor has been highlighted in several studies.ref. vro270034-bib-0070, ref. vro270034-bib-0075 Its modulation by the CBD used in this study was confirmed through its upregulation in these tumour cells.ref. vro270034-bib-0076 Both our in vitro assays and molecular analyses confirmed higher levels of PPAR‐α in tumour cells.
The genes for TRPV1 and GPR55 are also increased in dogs with this disease.ref. vro270034-bib-0077 The overexpression of TRPV1 and GPR55 receptors reveals the involvement of alternative signalling pathways.ref. vro270034-bib-0026, ref. vro270034-bib-0070 Indeed, the TRPV1 receptor, although known for its role in pain perception, is increasingly recognised as a mediator of tumour growth and tissue invasion.ref. vro270034-bib-0078 In parallel, the GPR55 receptor has been widely demonstrated to be involved in tumour cell survival and metastasis.ref. vro270034-bib-0079
The spontaneous occurrence of CMC also highlights the relevance of this model in a comparative oncology context, as dogs share the same domestic environment and are exposed to similar environmental stressors and pollutants, including heavy metals, which have been associated with cancer development in highly industrialised areas.ref. vro270034-bib-0080, ref. vro270034-bib-0081, ref. vro270034-bib-0082, ref. vro270034-bib-0083 In this framework, the modulation of inflammatory and proliferative pathways observed in our study further supports the translational value of targeting the ECS.
Our results demonstrate that CBD exerts a significant antiproliferative effect on CMC cells, characterised by an IC50 of approximately 33 µM. This reduction in viability is closely related to a clear modulation of the cell cycle, with a significant accumulation of cells in the G0 phase following exposure to CBD.ref. vro270034-bib-0084 Regarding the pharmacological relevance of these concentrations, a frequently debated point in veterinary oncology, it is important to contextualise these findings. Although an IC50 of approximately 33 µM exceeds the peak plasma concentrations (Cmax) typically observed in dogs, which range from 0.1 to 2.5 µM according to Di Salvo et al.ref. vro270034-bib-0085 Cannabidiol’s high lipophilicity and its large apparent volume of distribution should be taken into account when considering the pharmacological relevance of the concentrations tested in vitro.ref. vro270034-bib-0086 Although the IC50 identified in our model exceeds the peak plasma concentrations reported in dogs, the physicochemical characteristics of CBD suggest that its distribution is not adequately reflected by circulating levels alone. At present, however, direct measurements of CBD concentrations within mammary tumour tissue are unavailable, and dedicated pharmacokinetic studies will be necessary to clarify this aspect.
Importantly, we observed that even at sub‐cytotoxic concentrations (3–10 µM), CBD significantly reduced inflammatory gene expression and cytokine secretion. This indicates that meaningful biological activity occurs at levels below those required to impair cell viability. The overexpression of ECS‐related receptors detected in CMC cells may contribute to an enhanced sensitivity to CBD, potentially creating a signalling context in which inflammatory and proliferative pathways are particularly amenable to modulation.ref. vro270034-bib-0087
These findings suggest that ECS upregulation in CMC could represent a therapeutically relevant vulnerability deserving further investigation. In turn, CBD may have therapeutic potential in the treatment of this tumour, acting as multi‐target modulator of the ECS. Although these preliminary results are promising, future studies should clarify the underlying mechanisms of action and evaluate the clinical applicability of these approaches.
LIMITATIONS
Although these results are encouraging, it is important to acknowledge some limitations. The research used primary cell cultures from a small cohort of three canines. A larger and more diverse sample is needed to accurately reflect the histological and molecular variability of CMC, despite the statistical significance of these findings. Furthermore, although the 2D in vitro model used here is crucial for mechanical characterisation, it cannot accurately mimic the intricate 3D structure of a solid tumour or the systemic immunological interactions found in a living human.ref. vro270034-bib-0088
To better understand the molecular interactions between the ECS and inflammatory mediators, future research should focus on more in‐depth mechanistic investigations. To evaluate the safety, pharmacokinetics and therapeutic efficacy of CBD as an adjuvant treatment, it will be crucial to move from in vitro models to controlled clinical trials in canine patients. Furthermore, it should be noted that while gene expression analysis provided robust data on the modulation of the ECS and inflammatory markers, validation at the protein level for receptors and COX enzymes remains a challenge. This is primarily due to the limited availability of high‐affinity antibodies specifically validated or cross‐reactive for canine targets.ref. vro270034-bib-0089 Future studies will benefit from the development of more specific proteomic tools for veterinary oncology. Our results demonstrate that CBD modulates inflammation in CMC cells, likely through a mechanism related to the ECS. Although further studies using specific inhibitors are needed to confirm these pathways, this study represents a crucial step in understanding the therapeutic potential of cannabinoids in canine patients.
CONCLUSIONS
This study provides evidence that CMC exhibits a significant overexpression of CB1, CB2, TRPV1, GPR55 and PPAR‐α, indicating significant upregulation of the ECS. Our data suggest that CMC cells actively modulate these signalling pathways to sustain a robust pro‐inflammatory microenvironment. We have demonstrated that this inflammatory cascade is effectively interrupted by CBD; at non‐cytotoxic levels (10–20 µM), CBD reduces cytokine release and silences key inflammatory genes without compromising overall cell survival. Furthermore, it has been shown that CBD therapy could prevent uncontrolled tumour cell proliferation by inducing cell cycle arrest in the G0/G1 phase.
These findings may also have relevant implications for human health, as CMC shares key molecular and pathological features with human breast cancer. Therefore, the modulation of ECS‐related pathways observed in this study may reflect conserved mechanisms that could be exploited for the development of novel anti‐inflammatory and anti‐tumour strategies in human oncology.
AUTHOR CONTRIBUTIONS
Methodology, formal analysis and writing—original draft: Gianluca Antonio Franco. Methodology and data acquisition: Ylenia Marino. Investigation: Claudia Rifici. Formal analysis: Roberta Fusco. Supervision and validation: Rosanna di Paola. Conceptualisation and supervision: Salvatore Cuzzocrea. Data curation and writing—review and editing: Giuseppe Catone and Cecilia Vullo. Conceptualisation, supervision and writing—original draft and review and editing: Enrico Gugliandolo.
CONFLICTS OF INTEREST
The authors declare they have no conflicts of interest.
FUNDING INFORMATION
None.
ETHICS STATEMENT
None.
References
- Development, anatomy, histology, lymphatic drainage, clinical features, and cell differentiation markers of canine mammary gland neoplasms.. Vet Pathol., 2011. [PubMed]
- Canine mammary gland tumors.. Vet Clin Small Anim Pract., 2003
- Epidemiological studies on canine mammary tumour and its relevance for breast cancer studies.. IOSR J Pharm., 2012
- The role of connective tissue proliferation in invasive growth of normal and malignant tissues: a review.. Br J Cancer., 1958. [PubMed]
- Canine mammary tumors: risk factors, prognosis and treatments. J Vet Adv. 2016;6:1291‒1300.
- Factors influencing canine mammary cancer development and postsurgical survival.. J Natl Cancer Inst., 1969. [PubMed]
- Canine tumors: a spontaneous animal model of human carcinogenesis.. Transl Res., 2012. [PubMed]
- A role for T‐lymphocytes in human breast cancer and in canine mammary tumors.. BioMed Res Int., 2014. [PubMed]
- Chronic inflammation in cancer development.. Front Immunol., 2012. [PubMed]
- Inflammation and cancer: a comparative view.. J Vet Intern Med., 2012. [PubMed]
- Cancer and inflammation: promise for biologic therapy.. J Immunother., 2010. [PubMed]
- Inflammation, microenvironment, and the immune system in cancer progression.. Curr Pharm Des., 2009. [PubMed]
- The therapeutic aspects of the endocannabinoid system (ECS) for cancer and their development: from nature to laboratory.. Curr Pharm Des., 2016. [PubMed]
- The use of cannabinoids as anticancer agents.. Prog Neuro‐Psychopharmacol Biol Psychiatr., 2016
- The endocannabinoid system and its modulation by phytocannabinoids.. Neurotherapeutics., 2015. [PubMed]
- CB1 and CB2 receptor‐mediated signalling: a focus on endocannabinoids.. Prostaglandins Leukot Essent Fatty Acids., 2002. [PubMed]
- Programmed cell death pathways in cancer: a review of apoptosis, autophagy and programmed necrosis.. Cell Prolif., 2012. [PubMed]
- Insights into the effects of the endocannabinoid system in cancer: a review.. Br J Pharmacol., 2018. [PubMed]
- Cannabinoids: a new hope for breast cancer therapy?. Cancer Treat Rev., 2012. [PubMed]
- Cannabinoids as therapeutic agents in cancer: current status and future implications.. Oncotarget., 2014. [PubMed]
- Kiupel M. Surgical pathology of tumors of domestic animals. Davis‐Thompson DVM Foundation; 2008.
- Peroxisome proliferator‐activated receptor‐alpha (PPARA) genetic polymorphisms and breast cancer risk: a Long Island ancillary study.. Carcinogenesis., 2008. [PubMed]
- Involvement of PPARα in the growth inhibitory effect of arachidonic acid on breast cancer cells.. Br J Nutr., 2008. [PubMed]
- In vitro comparative models for canine and human breast cancers.. Clujul Med., 2016. [PubMed]
- Complex mammary tumours in the female dog: a review.. J Dairy Res., 2005. [PubMed]
- Expression and functionality of TRPV1 receptor in human MCF‐7 and canine CF.41 cells.. Vet Comp Oncol., 2015. [PubMed]
- Immunohistochemical staining of peroxisome proliferator activated receptor alpha and gamma in normal, benign, and malignant canine mammary tissues.. Thai J Vet Med., 2018
- Environmental mycotoxins and brain health: protective role of bromelain against fumonisin B1 in SH‐SY5Y cells. Environ Toxicol. 2026:1‒13.
- Protective effect of snail secretion filtrate in an in vitro model of mastitis.. Vet Rec Open., 2026. [PubMed]
- Effects of the antagomiRs 15b and 200b on the altered healing pattern of diabetic mice.. Br J Pharmacol., 2018. [PubMed]
- Selection of reference genes for quantitative real‐time PCR analysis in canine mammary tumors using the GeNorm algorithm.. Vet Pathol., 2006. [PubMed]
- Quick cytogenetic screening of breeding bulls using flow cytometric sperm DNA histogram analysis.. Acta Vet Hung., 2016. [PubMed]
- Surgical pathology of tumors of domestic animals. In: Kiupel M, editor. Mammary tumors. Vol. 2. Davis‐Thompson DVM Foundation; 2019.
- In vitro comparative models for canine and human breast cancers.. Clujul Med., 2016. [PubMed]
- The endocannabinoid system of animals.. Animals., 2019. [PubMed]
- The role of the endocannabinoid system in the control of energy homeostasis.. Int J Obesity,, 2006
- Homeostatic changes of the endocannabinoid system in Parkinson’s disease.. Movement Disord., 2011. [PubMed]
- Effect of dietary fat on endocannabinoids and related mediators: consequences on energy homeostasis, inflammation and mood.. Mol Nutr Food Res., 2010. [PubMed]
- Role of the endocannabinoid system in the neuroendocrine responses to inflammation.. Curr Pharm Des., 2014. [PubMed]
- The endocannabinoid system in pain and inflammation: its relevance to rheumatic disease.. Eur J Rheumatol., 2017. [PubMed]
- The immune endocannabinoid system of the tumor microenvironment.. Int J Mol Sci., 2020. [PubMed]
- Emerging approaches to study cell–cell interactions in tumor microenvironment.. Oncotarget., 2019. [PubMed]
- The cell cycle: a review.. Vet Pathol., 1998. [PubMed]
- From conventional to precision therapy in canine mammary cancer: a comprehensive review.. Front Vet Sci., 2021. [PubMed]
- Canine mammary tumors: classification, biomarkers, traditional and personalized therapies.. Int J Mol Sci., 2024. [PubMed]
- Cannabidiol (CBD) in cancer management.. Cancers., 2022. [PubMed]
- Mechanisms of cannabidiol (CBD) in cancer treatment: a review.. Biology., 2022. [PubMed]
- Potential use of cannabinoids for the treatment of pancreatic cancer.. J Pancreatic Cancer,, 2019
- Synergistic anticancer activity of cannabinoids and terpenes against triple‐negative breast cancer resistance.. Int J Mol Sci., 2026. [PubMed]
- Cannabigerol is a potential therapeutic agent in a novel combined therapy for glioblastoma.. Cells., 2021. [PubMed]
- Expression of Cox‐1 and Cox‐2 in canine mammary tumours.. J Comp Pathol., 2007. [PubMed]
- A dual inhibitor of cyclooxygenase and 5‐lipoxygenase protects against kainic acid‐induced brain injury.. Neuromol Med., 2015
- The role of Cox‐2 expression in the prognosis of dogs with malignant mammary tumours.. Res Vet Sci., 2010. [PubMed]
- Usefulness of selective COX‐2 inhibitors as therapeutic agents against canine mammary tumors.. Oncol Rep., 2014. [PubMed]
- Prognostic relevance of inflammatory cytokines Il‐6 and TNF‐alpha in patients with breast cancer: a systematic review and meta‐analysis.. Curr Oncol., 2025. [PubMed]
- Proinflammatory and anti‐inflammatory cytokines mediated by NF‐κB factor as prognostic markers in mammary tumors.. Mediators Inflamm., 2016. [PubMed]
- Blockade of the JNK signalling as a rational therapeutic approach to modulate the early and late steps of the inflammatory cascade in polymicrobial sepsis.. Mediators Inflamm., 2015. [PubMed]
- Interleukin‐33 (IL‐33): a nuclear cytokine from the IL‐1 family.. Immunol Rev., 2018. [PubMed]
- The IL‐1‐like cytokine IL‐33 is constitutively expressed in the nucleus of endothelial cells and epithelial cells in vivo: a novel ‘alarmin’?. PloS One,, 2008. [PubMed]
- Role of IL‐33 in inflammation and disease.. J Inflamm., 2011
- The pleiotropic immunomodulatory functions of IL‐33 and its implications in tumor immunity.. Front Immunol., 2018. [PubMed]
- Gene expression profiling of spontaneously occurring canine mammary tumours: insight into gene networks and pathways linked to cancer pathogenesis.. PLoS One., 2018. [PubMed]
- Oncogenic and tumor‐suppressive roles of Lipocalin 2 (LCN2) in tumor progression.. Oncol Res., 2025. [PubMed]
- The interplay between cancer biology and the endocannabinoid system‐significance for cancer risk, prognosis and response to treatment.. Cancers., 2020. [PubMed]
- The interplay between the immune and the endocannabinoid systems in cancer.. Cells., 2021. [PubMed]
- The endocannabinoid system: a target for cancer treatment.. Int J Mol Sci., 2020. [PubMed]
- The interplay between cancer biology and the endocannabinoid system—significance for cancer risk, prognosis and response to treatment.. Cancers., 2020. [PubMed]
- Synthetic cannabinoid receptor agonists inhibit tumor growth and metastasis of breast cancer.. Mol Cancer Therap., 2009. [PubMed]
- Crosstalk between chemokine receptor CXCR4 and cannabinoid receptor CB2 in modulating breast cancer growth and invasion.. PloS One., 2011. [PubMed]
- The role of the endocannabinoid system in oncology and the potential use of cannabis derivatives for cancer management in companion animals.. Animals,, 2025. [PubMed]
- The structure–function relationships of classical cannabinoids: CB1/CB2 modulation.. Persp Med Chem., 2016
- Modulation of the endocannabinoid system as a potential anticancer strategy.. Front Pharmacol., 2019. [PubMed]
- Stimulation of nuclear receptor PPAR‐γ limits NF‐kB‐dependent inflammation in mouse cystic fibrosis biliary epithelium.. Hepatology., 2015. [PubMed]
- Mechanisms of anti‐inflammatory and neuroprotective actions of PPAR‐gamma agonists.. Front Biosci J Virtual Lib., 2008
- An update on PPAR activation by cannabinoids.. Br J Pharmacol., 2016. [PubMed]
- Expression of peroxisome proliferator‐activated receptor α, and PPARα regulated genes in spontaneously developed hepatocellular carcinomas in fatty acyl‐CoA oxidase null mice.. Int J Oncol., 2002. [PubMed]
- The cytotoxic effects of cannabidiol and cannabigerol on glioblastoma stem cells may mostly involve GPR55 and TRPV1 signalling.. Cancers., 2022. [PubMed]
- Expression and functionality of TRPV1 receptor in human and canine mammary cancer cells.. 2014
- 79 Laliberte S . Assessment of cannabinoid receptor expression and anti‐neoplastic effects of cannabidiol (CBD) in canine urothelial carcinoma cells. University of Guelph; 2022.
- Levels of heavy metals in adolescents living in the industrialised area of Milazzo‐Valle del Mela (Northern Sicily).. J Environ Public Health., 2014. [PubMed]
- Mortality and heavy metals environmental exposure: a study in dogs.. Front Vet Sci., 2024. [PubMed]
- 82 Pandey G Heavy metals toxicity in domestic animal. International E‐Publication. International Science Congress Association; 2013.
- Levels of heavy metals in liver and kidney of dogs from urban environment.. Open Vet J., 2012. [PubMed]
- Cannabinol inhibits cell growth and triggers cell cycle arrest and apoptosis in cancer cells.. Biocatal Agric Biotechnol., 2023
- Pharmacokinetics, efficacy, and safety of cannabidiol in dogs: an update of current knowledge.. Front Vet Sci., 2023. [PubMed]
- Critical aspects affecting cannabidiol oral bioavailability and metabolic elimination, and related clinical implications.. CNS Drugs., 2020. [PubMed]
- Expression of cannabinoid receptors in human osteoarthritic cartilage: implications for future therapies.. Cannabis Cannabinoid Res., 2016. [PubMed]
- Engineering cancer microenvironments for in vitro 3‐D tumor models.. Mater Today., 2015
- Development and pharmacokinetic assessment of a fully canine anti‐PD‐1 monoclonal antibody for comparative translational research in dogs with spontaneous tumors.. MAbs., 2023. [PubMed]
