Brain Function Outcomes of Recent and Lifetime Cannabis Use
Abstract
Question:
Are recent cannabis use and lifetime cannabis use associated with differences in brain function during cognitive tasks?
Findings:
In this cross-sectional study of 1003 young adults, heavy lifetime cannabis use was associated with lower brain activation during a working memory task; this association remained after removing individuals with recent cannabis use. These results were not explained by differences in demographic variables, age at first cannabis use, alcohol use, or nicotine use.
Meaning:
These findings suggest that cannabis use is associated with short- and long-term brain function outcomes, especially during working memory tasks.
Affiliations: Department of Radiology, University of Colorado Anschutz Medical Campus, Aurora; Department of Psychiatry, University of Colorado Anschutz Medical Campus, Aurora; Department of Psychology, Colorado State University, Fort Collins; Laboratory of NeuroImaging, National Institute on Alcohol Abuse and Alcoholism, Bethesda, Maryland; Addictions Division, Centre for Addiction and Mental Health, Toronto, Ontario, Canada; Department of Pharmacology and Toxicology, University of Toronto, Toronto, Ontario, Canada; Division of Neurosciences and Clinical Translation, Department of Psychiatry, University of Toronto, Toronto, Ontario, Canada; Campbell Family Mental Health Research Institute, Centre for Addiction and Mental Health, Toronto, Ontario, Canada; Department of Psychological Clinical Science, University of Toronto Scarborough, Toronto, Ontario, Canada; Institute of Medical Science, University of Toronto, Toronto, Ontario, Canada; Institute for Mental Health Policy Research, Centre for Addiction and Mental Health, Toronto, Ontario, Canada; National Institute on Drug Abuse, Bethesda, Maryland
License: Copyright 2025 Gowin JL et al. JAMA Network Open. CC BY 4.0 This is an open access article distributed under the terms of the CC-BY License.
Article links: DOI: 10.1001/jamanetworkopen.2024.57069 | PubMed: 39874032 | PMC: PMC11775743
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (14 KB)
Introduction
As more states and countries have legalized the production and sale of cannabis for recreational and medical use,1 there has been an associated increase in the potency of cannabis products,2 cannabis use rates,3,4 and prevalence of cannabis use disorder.5 Greater accessibility of cannabis has also been associated with higher rates of cannabis-related motor vehicle crashes,6,7 and frequent cannabis use is associated with increased risk for hyperemesis syndrome8 and cardiovascular disease.9,10 Despite these negative effects, there is an increasing perception that cannabis is harmless.11 Thus, better understanding of recent and long-term effects of cannabis is critical for informing public health policies. Meta-analytic evidence indicates that short-term effects of cannabis include decreases in cognitive performance (eg, episodic verbal memory), but these reductions may not persist after 72 hours of abstinence.12 Given the cognitive effects of cannabis and the disruption of the endogenous cannabinoid system by tetrahydrocannabinol (THC),13,14 it may be that brain regions with high cannabinoid 1 (CB1) receptor density15 might be altered by cannabis. For example, there is evidence that cannabis use among adolescents is negatively associated with the thickness of the left prefrontal cortex (PFC) and right PFC and that the spatial pattern of cannabis-related cortical thinning is related to CB1 receptor density.16
Numerous brain imaging studies have examined the effects of cannabis on brain function. For example, relative to nonusers, frequent cannabis users showed a greater response to cannabis cues in the striatum and medial PFC, and activation of these regions correlated with cannabis craving.17 There may also be developmental interaction effects.18 For example, individuals with cannabis dependence, relative to matched control participants, showed greater functional connectivity density (ie, hyperconnectivity with surrounding regions) in the ventral striatum, and effects were more pronounced in individuals who began cannabis use earlier in life.19 Evidence has indicated that cannabis use reduces neural activation related to memory,20 executive function,21,22 emotion,23,24 reward processing,25 and social processing,26 but most of these previous studies had fewer than 30 participants with cannabis use history.20 Furthermore, whereas several efforts have successfully meta-analyzed the cognitive effects of cannabis across multiple domains,12,27 few have addressed the effects of cannabis use on brain function across multiple domains. It is also challenging to account for effects on multiple brain regions with an interpretable and clinically meaningful outcome, even though activation patterns of brain regions during tasks are not independent and, instead, are often highly correlated across regions. Evidence from a 2024 study suggests that brain analysis should consider features such as function, architectonics, connectivity, and topography.28 Such approaches, however, have seldom been applied to analysis of the effects of cannabis on brain function to help advance knowledge of the influence of history of use or recent use. Such work stands to improve understanding of how cannabis affects neural processing relevant to social, cognitive, and emotional function.
To address these knowledge gaps, we used data from the Human Connectome Project (HCP) for this study. The HCP has data across 7 tasks covering a range of brain functions. It also assesses lifetime cannabis use, cannabis dependence diagnosis (per Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition [DSM-IV] criteria), and age at first use and uses a urine toxicology screen at the time of scanning to assess for recent cannabis use. These data allowed us to disentangle outcomes associated with a lifetime history of cannabis use from those associated with recent use. The HCP dataset also allowed us to adjust for group differences between individuals with heavy, moderate, and no cannabis use, given that demographic and socioeconomic factors can influence brain function.29 We were also able to control for comorbid substance (eg, alcohol or nicotine) use, which is necessary to reduce the likelihood that any observed outcomes of cannabis use are actually attributable to use of other substances. Given that the largest effects of cannabis use are on learning, working memory,30 and verbal episodic memory,12 we hypothesized that cannabis would be associated with activation during working memory and language tasks, and that this association would be present for recent use and lifetime history of use.
Methods
Procedures for this cross-sectional study were approved by the Washington University Institutional Review Board. Participants provided written consent. We preregistered our analysis on the Open Science Framework repository.31 The study followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guideline.
Participants
The HCP study consisted of 1206 adults aged 22 to 37 years,32 of whom 1005 had functional magnetic resonance imaging (MRI) data. The goal of the study was to “chart the neural pathways that underlie brain function and behavior.”32 Participants were recruited to complete MRI scans at Washington University in Saint Louis, Missouri. The study sampled twins and their nontwin siblings, along with singleton community members.32 Sibling status, representing a biological relationship, was based on self-report and genotyping from blood or saliva samples. The study was launched in 2010, and scanning began in August 2012 and concluded in 2015.33 For this analysis, we accessed data from the 2017 preprocessed data release. Although we initially planned to examine the language task only, we decided to analyze all 7 functional MRI tasks to broaden our understanding of brain function outcomes of cannabis use.
Assessment of Cannabis Use
To assess recent use, participants provided urine samples on the day of scanning that were tested for the presence of cannabis metabolites. For analysis, we categorized individuals as having recent use if they had a positive THC result on the Accutest multidrug screen (Jant). A positive result is indicated if urine concentrations of the THC metabolite 11-Nor-Δ9-tetrahydrocannabinol-9-carboxylic acid (THC-COOH) exceed 50 ng/mL. This result typically indicates use in the past 10 days; however, very frequent users may have THC-COOH levels greater than 50 ng/mL for 1 month, or on some occasions several months, post use.34
To assess lifetime use, participants completed the Semi-Structured Assessment for the Genetics of Alcoholism (SSAGA).35 The interview assessed total lifetime number of cannabis uses on a Likert-rated scale (with response levels of never, 1-5, 6-10, 11-100, 101-999, and >1000). We categorized individuals as nonusers if they had used 10 or fewer times, as moderate users if they had used 11 to 999 times, and as heavy users if they had used 1000 or more times. The lifetime use variable was treated as an ordered factor (nonusers, 0; moderate users, 1; and heavy users, 2).
The SSAGA also assessed a diagnosis of cannabis dependence (lifetime) (per DSM-IV criteria) and age at first cannabis use (never or <14, 15-17, 18-20, or >21 years). We treated these as covariates.
Demographic and Clinical Assessment
Participants completed demographic screening to indicate age, sex, race, income, and education. Race was self-reported as American Indian or Alaska Native, Asian (including Native Hawaiian or Other Pacific Islander), Black or African American (hereinafter, Black), White, multiple races, or race unknown or not reported. These data were collected because sociocultural influences associated with race may also be associated with both cannabis use and brain activation patterns. We did not have sufficient data on ethnicity to analyze. Additional substance use variables were assessed with the SSAGA, including alcohol dependence diagnosis, and the Fagerström Test for Nicotine Dependence.36 To generate a single metric each for (1) alcohol use and (2) nicotine use that accounted for quantity and frequency, we created z score metrics as described previously.19 Episodic verbal memory was assessed with Form A of the Penn Word Memory Test.37 For crystallized intelligence, participants completed the National Institutes of Health Toolbox Picture Vocabulary Test,38 which assesses vocabulary knowledge and is associated with scholastic success39 and the “g” factor of intelligence.40
Brain Imaging Tasks
Seven tasks were used in this study, aiming to cover a broad range of behavioral processes.41 Tasks were chosen based on reliability of neural response and a well-characterized neurocognitive basis. The tasks examined neural response related to emotion, reward, motor function, working memory, language, relational or logical reasoning, and theory of mind or social information processing (task details are provided in the eMethods in Supplement 1). Neural response to the tasks has previously discriminated substance use history.42 For each task, we used the primary contrast, and we extracted activation levels from regions positively activated during the task (ie, regions engaged by the task, not regions that were deactivated by the task, such as nodes in the default mode network). Positive activation was defined as having significant activation (2-tailed P < .001), with most effect sizes (Cohen d) greater than 1.00 for the contrast (the task positive condition minus the control condition).43 We used the mean value of the activation levels across the listed regions for each task, so each participant had a single value representing the activation level for each task; the regions used are depicted in Figure 1. We chose to use a single value because (1) it reduced the number of outcomes for analysis, (2) it provided a more clinically interpretable metric, and (3) the activation levels across regions during a given task were not independent.44
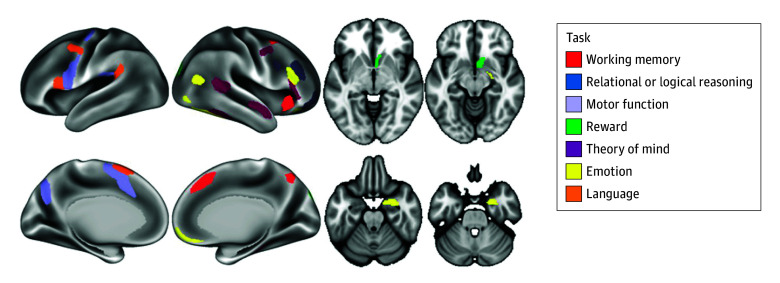
Imaging Acquisition and Processing
The HCP investigators used a 3T Connectome Skyra scanner (Siemens) to acquire imaging data. For parameters, see the data acquisition plan published previously.45 Preprocessing was conducted as part of the HCP minimally preprocessed dataset with the FMRIB Software Library (Oxford University).46
Statistical Analysis
Analytic Approach
Data were analyzed from January 31 to July 30, 2024. To examine participant characteristics, we compared nonusers, moderate users, and heavy users based on race, sex, age, education, income level, urine sample status, cannabis dependence diagnosis, age at first cannabis use, alcohol dependence diagnosis, nicotine dependence score, and alcohol z scores indicating quantity or frequency of use. We used the Wilcoxon rank sum test or the Fisher exact test to determine significance levels for the Table, and we report χ2 values in the Results. Statistical significance was set at a 2-tailed α ≤ .05.
Table. : Participant Characteristics by History of Cannabis Usea
| Characteristic | Nonuser (n = 736) | Moderate user (n = 179) | Heavy user (n = 88) | P value |
|---|---|---|---|---|
| Race | ||||
| American Indian or Alaska Native | 1 (0.1) | 0 | 1 (1.1) | <.001 |
| Asian (including Native Hawaiian or Other Pacific Islander) | 56 (7.6) | 6 (3.4) | 1 (1.1) | |
| Black or African American | 96 (13.0) | 22 (12.3) | 19 (21.6) | |
| White | 565 (76.8) | 137 (76.5) | 60 (68.2) | |
| Multiple races | 10 (1.4) | 9 (5.0) | 4 (4.5) | |
| Unknown or not reported | 8 (1.1) | 5 (2.8) | 3 (3.4) | |
| Sex | ||||
| Female | 432 (58.7) | 80 (44.7) | 21 (23.9) | <.001 |
| Male | 304 (41.3) | 99 (55.3) | 67 (76.1) | |
| Age, mean (SD), y | 28.8 (3.7) | 28.7 (3.8) | 28.3 (3.4) | .44 |
| Income, $ | ||||
| <50 000 | 335 (45.5) | 94 (52.5) | 58 (65.9) | .005 |
| ≥50 000 and <100 000 | 280 (38.0) | 56 (31.3) | 19 (21.6) | |
| ≥100 000 | 118 (16.0) | 28 (15.6) | 10 (11.4) | |
| Missing | 3 (0.4) | 1 (0.6) | 1 (1.1) | |
| Education | ||||
| High school diploma or GED | 126 (17.1) | 49 (27.4) | 22 (25.0) | <.001 |
| Less than high school diploma | 17 (2.3) | 5 (2.8) | 10 (11.4) | |
| Some college | 129 (17.5) | 36 (20.1) | 20 (22.7) | |
| Bachelor’s degree | 336 (45.7) | 68 (38.0) | 29 (33.0) | |
| Postgraduate degree | 128 (17.4) | 21 (11.7) | 7 (8.0) | |
| Cannabis dependencezoi241596t1n2 | 0 | 40 (22.3) | 53 (60.2) | <.001 |
| THC positive test result | 19 (2.6) | 37 (20.7) | 50 (56.8) | <.001 |
| Age at first cannabis use, y | ||||
| Never | 458 (62.2) | 0 | 0 | <.001 |
| <14 | 11 (1.5) | 17 (9.5) | 31 (35.2) | |
| 15-17 | 77 (10.5) | 86 (48.0) | 33 (37.5) | |
| 18-20 | 106 (14.4) | 58 (32.4) | 17 (19.3) | |
| ≥21 | 84 (11.4) | 18 (10.1) | 7 (8.0) | |
| Alcohol dependencezoi241596t1n2 | 29 (3.9) | 20 (11.2) | 11 (12.5) | <.001 |
| Nicotine dependencezoi241596t1n3 | ||||
| Median score (range) | 1.00 (0-6.00) | 1.00 (0-6.00) | 2.50 (0-6.00) | .02 |
| Nonsmoker | 627 (85.2) | 93 (52.0) | 26 (29.5) | |
| Alcohol z score, mean (SD)zoi241596t1n4 | −0.05 (0.53) | 0.17 (0.39) | 0.23 (0.46) | <.001 |
Unless specified otherwise, values are presented as No. (%) of participants.
Assessed using the Semi-Structured Assessment for the Genetics of Alcoholism using Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition criteria.
Score on the Fagerström Test for Nicotine Dependence.
Composite score representing frequency and quantity of alcohol consumption.
Abbreviations: GED, general educational development; THC, tetrahydrocannabinol.
For the primary analysis, we used 7 linear mixed-effects regression models (ie, 1 model for each of the 7 tasks). For each model, we used the lme4 package in R, version 4.4.0 (R Project for Statistical Computing), to assess associations of the tasks with (1) lifetime history of cannabis (linear and quadratic fit), (2) recent use, and (3) history of dependence. Lifetime history was coded as an ordered factor with 3 levels. Models were adjusted for effects of alcohol, nicotine, race, education, income, sex, and age at first cannabis use (reference: never used). Sibling status was assessed, and this nonindependence was accounted for by a random intercept of a single variable, MotherID, which was represented as a unique value for every biological family and coded as a factor. Given the 7 models, we performed false discovery rate correction for P values using the Benjamini-Hochberg method.
For graphical comparison, we calculated effect sizes (Cohen d) for activation during each task by (1) lifetime history of use (heavy users vs nonusers), (2) recent use (THC-positive vs THC-negative result), and (3) cannabis diagnosis (history vs no history of dependence). We report comparisons between nonusers and moderate users, as well as heavy users and moderate users, in eFigures 2 and 3 in Supplement 1.
Post Hoc and Sensitivity Analyses
For statistically significant results of brain activation, we conducted post hoc analysis to determine which regions were associated with the result. This involved running separate linear mixed-effects models as specified earlier for each brain region (eg, for the working memory task, the 4 regions included the anterior insula [AVI], the superior parietal lobule [7Pm], the medial PFC [8BM], and the dorsolateral PFC [i6-8]).
To examine how brain activation during the tasks are associated with cognitive performance, we fit the linear mixed-effects models described earlier for (1) accuracy during the working memory task, (2) performance on episodic verbal memory, and (3) accuracy during the theory of mind task. To confirm that outcomes associated with lifetime history of heavy use were not associated with recent cannabis use, we excluded all individuals who had a positive test result for THC and we reran the models for the working memory and theory of mind tasks. We assessed the assumption of no unmeasured confounders using the sensemakr package in R, whereby we fit a linear model as specified for our primary analyses and indicated that recent THC use was our benchmark covariate for confounding. This analysis indicates the level that an unmeasured confound would have to be associated with the primary independent variable (history of use) and outcome (activation) to reduce the estimate of the association to 0. We also examined correlation using Spearman rank-ordered correlation for activation during each of the 7 tasks, along with episodic verbal memory, crystallized intelligence, income, and education level (as an ordered factor). We conducted THC-by-sex interaction models for tasks that showed an association with recent use.
Results
Participant Characteristics
This study comprised 1003 adults (mean [SD] age, 28.7 [3.7] years), including 470 men (46.9%) and 533 women (53.1%). A total of 2 participants (0.1%) were American Indian or Alaska Native, 63 were Asian (6.3%), 137 were Black (13.7%), 762 were White (76.0%), and 23 (2.3%) were of multiple races; race was unknown or not reported for 16 participants (1.6%). Heavy lifetime users were more likely to be male, to have lower income, and to have lower levels of education than nonusers (χ22 = 41.7; P < .001) (Table). Heavy lifetime users were also more likely to have positive THC urine screens and a diagnosis of dependence relative to moderate users and nonusers (χ22 = 268.2; P < .001). Heavy lifetime users also had higher scores for nicotine dependence severity and alcohol use relative to those with negative THC test results as determined via the Wilcoxon rank sum test (W > 57 539; P < .001).
Brain Activation Outcomes of Cannabis Use
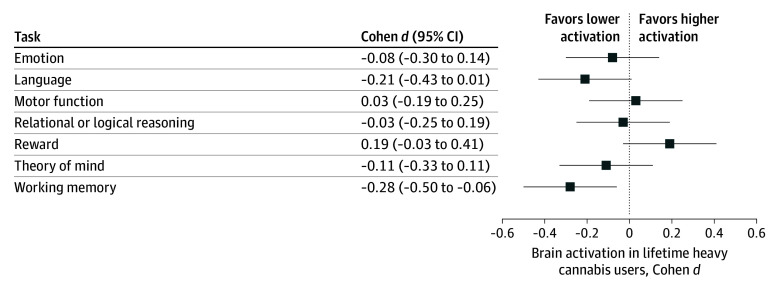
Although there were no linear associations between lifetime history of cannabis use and task activation after adjusting for associations with recent use, there were significant quadratic effects (β = −4.54 [95% CI, −7.55 to −1.54]; P = .003 and Benjamini-Hochberg adjusted P = .02), such that greater use was associated with lower activation (full statistical models are shown in eTables 1-7 in Supplement 1). The association between lifetime history of use and theory of mind task activation did not survive correction for multiple comparisons. A summary of effect sizes for brain activation among lifetime heavy users vs nonusers is illustrated in Figure 2; the largest effect size (Cohen d = −0.28 [95% CI, −0.50 to −0.06]; false discovery rate corrected P = .02) was observed for the working memory task. Central tendency by group is reported for each task in eTable 8 in Supplement 1.
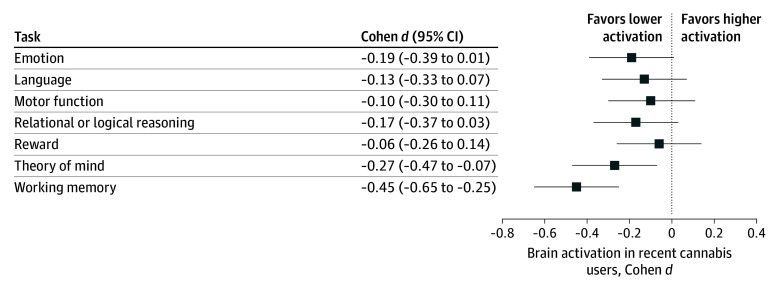
The comparison of individuals with positive vs negative THC test results revealed lower brain activation in recent cannabis users for the working memory and the theory of mind tasks. However, in the full model, the associations for the theory of mind task no longer remained after adjusting for race and education, because both of these variables were associated with a positive THC test result and also with task activation (eTables 10 and 11 in Supplement 1). After adjusting for associations with lifetime use, the working memory and motor tasks showed associations between recent cannabis use and activation levels but these did not survive false discovery rate correction. A summary of effect sizes for brain activation and positive vs negative urine samples is illustrated in Figure 3, and central tendency by group is reported in eTable 9 in Supplement 1. No task showed an association with diagnosis of cannabis dependence (eFigure 1 and eTable 12 in Supplement 1).
Post Hoc and Sensitivity Analyses
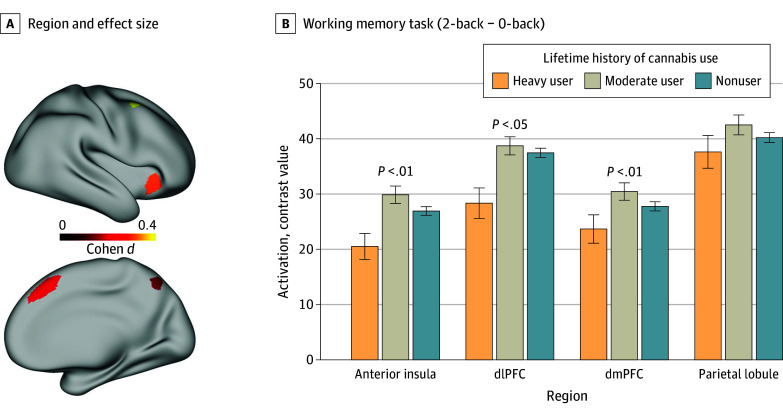
For the working memory task, the models for the anterior insula, medial PFC, and dorsolateral PFC showed associations with lifetime history of cannabis use (all t < −2.0; all P < .02), as shown in Figure 4 (full models are displayed in eTables 13-16 in Supplement 1). However, the model for the superior parietal lobule (t = −1.6; P = .10) did not.
When removing all individuals with a positive urine screen for THC, 38 individuals remained who had a lifetime history of heavy cannabis use, and the association with lifetime history of heavy use remained for working memory task brain activation (β = −3.66 [95% CI, −7.35 to 0.02]; P = .05). The analysis of no unmeasured confounders indicated that a variable could have 3 times the magnitude of association with history of use and brain activation, relative to recent use, and still not reduce the estimate of the primary association to 0, which seems unlikely.
For behavioral performance, recent cannabis use was associated with poorer performance on the working memory task, the episodic verbal memory task, and the theory of mind task (eTables 17-19 in Supplement 1). Lifetime history of heavy use was not associated with performance on these tasks.
Brain activation levels during the relational, theory of mind, and working memory tasks were correlated with crystallized intelligence, education, and scores on the verbal episodic memory task (ρ > 0.13; P < .001) (eFigure 4 in Supplement 1). There was no sex-by-THC interaction for working memory but there was for the motor task (t = −3.3; P = .001), such that women showed no association with THC (t = 1.88; P = .24) but men showed lower activation levels if they had a positive THC result (t = 3.17; P = .01) (eFigure 5 in Supplement 1).
Discussion
In this study, we characterized the association of recent cannabis use and heavy lifetime cannabis use with brain activation across 7 tasks. Heavy lifetime cannabis use was associated with lower brain activation only during the working memory task, and this association remained after excluding individuals with recent use. Brain activation levels during the working memory task were associated with verbal episodic memory performance, intelligence, and education, suggesting that they are meaningful indicators of cognitive function. Although recent cannabis use was associated with lower brain activation during the working memory and motor tasks, the association did not remain after false discovery rate correction. We observed an absence of an association of a dependence diagnosis with brain function, suggesting that factors associated with diagnosis, such as social and legal consequences, may be unrelated to brain function outcomes attributable to cannabis use. Thus, the diagnoses of dependence may be less relevant than recent or cumulative exposure to pharmacologically active components of cannabis (eg, THC).
In this study, lower brain activation during the working memory task in heavy cannabis users was most pronounced in the dorsolateral PFC, dorsomedial PFC, and anterior insula. These are regions that have a relatively high density of CB1 receptors and where receptor availability was found to be reduced in association with daily cannabis exposure.47 Similarly, rodent studies showed that THC exposure reduced the density and sensitivity of CB1 receptors in these brain regions,48 providing evidence that heavy cannabis use can cause neural adaption. Because THC can reduce CB1 density, this could provide a mechanism to explain findings that cannabis use is associated with lower cortical thickness in the dorsomedial PFC and dorsolateral PFC.16 The impact of these putative effects was observed on the working memory task in the current study. A previous study that examined the HCP data also showed that recent cannabis use was associated with lower activation during the working memory task in the anterior insula and middle frontal gyrus, and that their decreased activation mediated the association between cannabis use and poorer performance on an episodic memory task.49,50 Our results are consistent with these findings, although they suggest that heavy lifetime cannabis use among participants was associated with lower activation to a working memory task even after removing individuals with a positive urine screen at the time of testing to control for recent use. This finding also accords with evidence that heavy cannabis use alters brain activation in the absence of recent use51 and that acute THC administration reduces brain activation in brain regions involved in working memory.52
The association we observed between recent use and working memory task activation and performance suggests that abstaining from cannabis prior to cognitively demanding situations will likely help with performance. The exact duration of this period of abstinence is unclear, but studies suggest that residual cognitive effects of cannabis may remain for 2 to 4 weeks after abstinence.53,54 Furthermore, in heavy users, abstaining from cannabis may also lead to withdrawal symptoms, which may last for a week or more following cessation and could also affect performance.55 Our findings highlight the need to educate cannabis users about the consequences of recent and heavy lifetime cannabis use on cognitively demanding working memory tasks. Similarly, the association between heavy use and decreased brain function could motivate regular cannabis users to reduce their cannabis use and could encourage treatment. Further studies are required to determine guidance on the length of abstinence that may be necessary to improve cognitive performance.
We observed that recent cannabis use was associated with decreased behavioral accuracy in the theory of mind task with similar, albeit not statistically significant, brain activation outcomes for recent use and history of heavy cannabis use. Reduced brain activation to a theory of mind task was reported previously in cannabis users relative to healthy adults, and the study’s authors hypothesized that this could contribute to the increased risk of schizophrenia, a condition associated with profound deficits in theory of mind processes.56 Despite this evidence, few studies have investigated theory of mind–related activation in cannabis-using samples; to our knowledge, our study represents a relatively novel contribution. The deficits in theory of mind–related processing and working memory processing may suggest that THC exposure may affect overlapping neural mechanisms that could contribute to observed associations between THC and psychopathology. In our study, we also observed reduced activation in recent cannabis users, which could contribute to the emergence of acute psychoses observed during THC intoxication, particularly for high THC doses.57 For the motor task, we observed a significant interaction of sex with brain activation, such that men showed lower activation when they had a positive THC result but women showed no effect of THC. A 2022 review identified 18 studies that examined a sex-by-THC interaction effect58; although the majority of these studies showed no interaction, the few that did indicated that women experienced greater effects of cannabis than men.58 These effects included smaller orbitofrontal cortex and cerebellar59 volumes in women vs men with cannabis dependence.60 In addition, relative to nonusers, female (but not male) heavy cannabis users showed a blunted neural response to a stimulant challenge.61 Studies specifically designed and powered to assess the interaction of cannabis with sex throughout the lifespan are needed.
Limitations
This study has limitations. This was an uncontrolled, cross-sectional study, so the observed associations of cannabis with brain function outcomes should not be considered causal. Participants were young adults, so these findings may not generalize to other age groups. History of heavy cannabis use was defined as a lifetime history of greater than 1000 uses or a diagnosis of cannabis dependence, but the sample was recruited from the community, so it may represent a relatively low level of addiction severity. We lacked data to determine when the most recent use occurred or to quantify THC metabolite concentration. It is possible that the association of recent use with brain activation would have been larger in a study where use was determined to be closer to the scan time so that participants experience peak effects of THC during tasks (ie, 0.5-4 hours after use, depending on route of administration). The timing of heavy THC exposure is unknown; although age at first use was not statistically significant in our models, first use is a crude measure, and the timing of heavy use may still matter.16 We also lacked data on typical THC dose, potency, additional cannabis constituents (eg, cannabidiol), and route of cannabis administration. Finally, although the sample size was relatively large, some subgroups (eg, women with a positive urine sample) were small, limiting statistical power. Similarly, we could not examine other substance use (eg, opioids) due to low frequency, and we did not examine psychiatric comorbidities.
Conclusions
In this cross-sectional study of young adults, lifetime heavy cannabis use history was associated with lower brain activation related to working memory, with a small to medium effect size. Before adjustment for covariates and correction for multiple comparisons, recent and lifetime cannabis use were associated with poorer behavioral performance on the theory of mind task; therefore, theory of mind should be examined in future studies. Evidence supported that both recent and heavy lifetime cannabis use were associated with diminished brain activation and cognitive performance during working memory. These findings suggest that large, longitudinal studies are needed to assess the causality of cannabis use toward altering brain function and the duration over which these effects persist.
References
- Public health implications of legalising the production and sale of cannabis for medicinal and recreational use.. Lancet., 2019. [DOI | PubMed]
- A comprehensive review of cannabis potency in the United States in the last decade.. Biol Psychiatry Cogn Neurosci Neuroimaging., 2021. [DOI | PubMed]
- Public health impacts to date of the legalisation of medical and recreational cannabis use in the USA.. Neuropharmacology., 2021. [DOI | PubMed]
- Marijuana use and use disorders in adults in the USA, 2002-14: analysis of annual cross-sectional surveys.. Lancet Psychiatry., 2016. [DOI | PubMed]
- Chronic pain, cannabis legalisation, and cannabis use disorder among patients in the US Veterans Health Administration system, 2005 to 2019: a repeated, cross-sectional study.. Lancet Psychiatry., 2023. [DOI | PubMed]
- Trends in fatal motor vehicle crashes before and after marijuana commercialization in Colorado.. Drug Alcohol Depend., 2014. [DOI | PubMed]
- Cohort study of medical cannabis authorization and motor vehicle crash-related healthcare visits in 2014-2017 in Ontario, Canada.. Inj Epidemiol., 2021. [DOI | PubMed]
- Cannabinoid hyperemesis syndrome: prevalence and management in an era of cannabis legalization.. J Investig Med., 2024. [DOI | PubMed]
- Association of cannabis use with cardiovascular outcomes among US adults.. J Am Heart Assoc., 2024. [DOI | PubMed]
- Cannabis-related disorders and toxic effects.. N Engl J Med., 2023. [DOI | PubMed]
- Use of marijuana for medical purposes among adults in the United States.. JAMA., 2017. [DOI | PubMed]
- Association of cannabis with cognitive functioning in adolescents and young adults: a systematic review and meta-analysis.. JAMA Psychiatry., 2018. [DOI | PubMed]
- Endocannabinoid signaling in psychiatric disorders: a review of positron emission tomography studies.. Acta Pharmacol Sin., 2019. [DOI | PubMed]
- Cannabis addiction and the brain: a review.. J Neuroimmune Pharmacol., 2018. [DOI | PubMed]
- [18F]MK-9470, a positron emission tomography (PET) tracer for in vivo human PET brain imaging of the cannabinoid-1 receptor.. Proc Natl Acad Sci U S A., 2007. [DOI | PubMed]
- Association of cannabis use during adolescence with neurodevelopment.. JAMA Psychiatry., 2021. [DOI | PubMed]
- Patterns of brain function associated with cannabis cue-reactivity in regular cannabis users: a systematic review of fMRI studies.. Psychopharmacology (Berl)., 2021. [DOI | PubMed]
- The neurocognitive effects of cannabis across the lifespan.. Curr Behav Neurosci Rep., 2021. [DOI | PubMed]
- Subcortical local functional hyperconnectivity in cannabis dependence.. Biol Psychiatry Cogn Neurosci Neuroimaging., 2018. [DOI | PubMed]
- Residual effects of cannabis use in adolescent and adult brains: a meta-analysis of fMRI studies.. Neurosci Biobehav Rev., 2018. [DOI | PubMed]
- An evidence based review of acute and long-term effects of cannabis use on executive cognitive functions.. J Addict Med., 2011. [DOI | PubMed]
- Evidence on the acute and residual neurocognitive effects of cannabis use in adolescents and adults: a systematic meta-review of meta-analyses.. Addiction., 2022. [DOI | PubMed]
- Acute effects of delta-9-tetrahydrocannabinol, cannabidiol and their combination on facial emotion recognition: a randomised, double-blind, placebo-controlled study in cannabis users.. Eur Neuropsychopharmacol., 2015. [DOI | PubMed]
- Cannabis and emotion processing: a review of behavioral, physiological, and neural responses.. Exp Clin Psychopharmacol., 2023. [DOI | PubMed]
- The acute and non-acute effects of cannabis on reward processing: a systematic review.. Neurosci Biobehav Rev., 2021. [DOI | PubMed]
- Emotional processing deficits in chronic cannabis use: a replication and extension.. J Psychopharmacol., 2014. [DOI | PubMed]
- Neurocognitive consequences of chronic cannabis use: a systematic review and meta-analysis.. Neurosci Biobehav Rev., 2020. [DOI | PubMed]
- Principles of cortical areas and their implications for neuroimaging.. Neuron., 2024. [DOI | PubMed]
- Functional and structural brain correlates of socioeconomic status.. Cereb Cortex., 2020. [DOI | PubMed]
- A mini-review of relationships between cannabis use and neural foundations of reward processing, inhibitory control and working memory.. Front Psychiatry., 2021. [DOI | PubMed]
- 31Gowin JL, Manza P, Ellingson JM, Ross J, Karoly HC. Verbal memory and language processing in young adults who use cannabis: an analysis of the Human Connectome Project. Accessed January 3, 2024. https://osf.io/bv34e
- The WU-Minn Human Connectome Project: an overview.. Neuroimage., 2013. [DOI | PubMed]
- The Human Connectome Project: a retrospective.. Neuroimage., 2021. [DOI | PubMed]
- Detection time of drugs of abuse in urine.. Acta Clin Belg., 2000. [DOI | PubMed]
- A new, semi-structured psychiatric interview for use in genetic linkage studies: a report on the reliability of the SSAGA.. J Stud Alcohol., 1994. [DOI | PubMed]
- The Fagerström Test for Nicotine Dependence: a revision of the Fagerstrom Tolerance Questionnaire.. Br J Addict., 1991. [DOI | PubMed]
- Computerized neurocognitive scanning: I. Methodology and validation in healthy people.. Neuropsychopharmacology., 2001. [DOI | PubMed]
- Language measures of the NIH Toolbox Cognition Battery.. J Int Neuropsychol Soc., 2014. [DOI | PubMed]
- Relationship between language skills and academic achievement in first grade.. Percept Mot Skills., 2001. [DOI | PubMed]
- 40Cattell RB. Intelligence: Its Structure, Growth and Action. Elsevier; 1987.
- Function in the human connectome: task-fMRI and individual differences in behavior.. Neuroimage., 2013. [DOI | PubMed]
- Neuropsychosocial markers of binge drinking in young adults.. Mol Psychiatry., 2021. [DOI | PubMed]
- A multi-modal parcellation of human cerebral cortex.. Nature., 2016. [DOI | PubMed]
- Intrinsic and task-evoked network architectures of the human brain.. Neuron., 2014. [DOI | PubMed]
- The Human Connectome Project: a data acquisition perspective.. Neuroimage., 2012. [DOI | PubMed]
- FSL.. Neuroimage., 2012. [DOI | PubMed]
- Reversible and regionally selective downregulation of brain cannabinoid CB1 receptors in chronic daily cannabis smokers.. Mol Psychiatry., 2012. [DOI | PubMed]
- Sex, THC, and hormones: effects on density and sensitivity of CB1 cannabinoid receptors in rats.. Drug Alcohol Depend., 2019. [DOI | PubMed]
- Urinary tetrahydrocannabinol is associated with poorer working memory performance and alterations in associated brain activity.. Neuropsychopharmacology., 2019. [DOI | PubMed]
- Cannabis involvement and neuropsychological performance: findings from the Human Connectome Project.. J Psychiatry Neurosci., 2019. [DOI | PubMed]
- Long-term effects of frequent cannabis use on working memory and attention: an fMRI study.. Psychopharmacology (Berl)., 2006. [DOI | PubMed]
- Effects of Δ9-tetrahydrocannabinol on human working memory function.. Biol Psychiatry., 2012. [DOI | PubMed]
- Impact of 2 weeks of monitored abstinence on cognition in adolescent and young adult cannabis users.. J Int Neuropsychol Soc., 2020. [DOI | PubMed]
- One month of cannabis abstinence in adolescents and young adults is associated with improved memory.. J Clin Psychiatry., 2018. [DOI | PubMed]
- Clinical management of cannabis withdrawal.. Addiction., 2022. [DOI | PubMed]
- Alterations of theory of mind network activation in chronic cannabis users.. Schizophr Res., 2012. [DOI | PubMed]
- Cannabis and psychosis.. Psychiatr Clin North Am., 2023. [DOI | PubMed]
- Interaction of sex and cannabis in adult in vivo brain imaging studies: a systematic review.. Brain Neurosci Adv., 2022. [DOI | PubMed]
- Cannabis affects cerebellar volume and sleep differently in men and women.. Front Psychiatry., 2021. [DOI | PubMed]
- Orbitofrontal and caudate volumes in cannabis users: a multi-site mega-analysis comparing dependent versus non-dependent users.. Psychopharmacology (Berl)., 2017. [DOI | PubMed]
- Cannabis abusers show hypofrontality and blunted brain responses to a stimulant challenge in females but not in males.. Neuropsychopharmacology., 2016. [DOI | PubMed]
