Influence of Drying Methods on the Phenolic Content and Antioxidant Properties of Crocus sativus L. Tepals and Leaves By‐Products From Meknes (Morocco)
Abstract
In recent years, much research has focused on the valorization of Crocus sativus by‐products as sources of bioactive compounds. This study was designed to investigate the impact of two different drying methods, freeze‐drying and air‐drying, on the phenolic content and antioxidant activity of leaves and tepals by‐products from C. sativus collected in Meknes, Morocco. Stigmas were included in the study to provide further information on this precious spice. The phenolic content of the hydroalcoholic extracts was determined spectrophotometrically and characterized by HPLC‐PDA/ESI‐MS analysis. The antioxidant properties were evaluated using different in vitro assays. Both in the DPPH and in the ferrous ion chelating activity assays, the extracts from freeze‐dried tepals (Cs‐TFD) and leaves (Cs‐LFD) displayed higher activity than those from air‐dried plant materials. Cs‐LFD showed the best scavenging activity, while superimposable chelating activity was observed for Cs‐TFD and Cs‐LFD. Finally, no toxicity was observed in the Artemia salina lethality bioassay.
Article type: Research Article
Keywords: antioxidant activity, by‐products, drying method, phenolics, saffron
Affiliations: Faculty of Sciences, Department of Biology Moulay Ismail University Meknes Morocco; Messina Institute of Technology c/o Department of Chemical, Biological, Pharmaceutical and Environmental Sciences, former Veterinary School University of Messina Messina Italy; Chromaleont s.r.l., c/o Messina Institute of Technology c/o Department of Chemical, Biological, Pharmaceutical and Environmental Sciences, former Veterinary School University of Messina Messina Italy; Department of Chemical, Biological, Pharmaceutical and Environmental Sciences University of Messina Messina Italy; Foundation “Prof. Antonio Imbesi” University of Messina Messina Italy
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202402967 | PubMed: 40062417 | PMC: PMC12270362
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (830 KB)
Introduction
Crocus sativus L. is an herbaceous perennial plant belonging to Iridaceae family. The corm is a subsoil organ providing vegetative propagation; it contains two or three apical buds and four to seven secondary buds. The apical buds sprout to produce leaves, a floral axis, and two or three daughter corms. The secondary buds produce a cauline axis and a clump of leaves [ref. 1]. The leaves develop from September to May, after which they begin to dry, corresponding to the beginning of the dormancy phase of the corms [ref. 2]. C. sativus has a subhysteranthous behavior, which means that the flowers can appear before, simultaneously, or after the leaves [ref. 1]. The flowers appear from October to the middle of November. They are composed of six violet tepals, three stamens, and a pistil, which contains an inferior ovary and a style ending with a red stigma divided into three filaments [ref. 3].
The name saffron is commonly applied to refer to both the plant and the dried stigmas, which represent the most precious spice in the world. The production of saffron spice generates enormous quantities of by‐products without any industrial application, which must thereby be discarded. It is estimated that saffron tepals and anthers represent about 90% of the whole flower’s fresh weight; to yield 1 kg of stigmas, 63 kg of floral by‐products, and 1500 kg of foliage are wasted, as well as hundreds of bulbs that are too small and/or have morphological or biological alterations [ref. 4, ref. 5, ref. 6]. How to reuse these unexploited by‐products has become a major issue for farmers and industries. In fact, from the perspective of the circular economy, the sustainable use of agrifood wastes and/or by‐products to produce value‐added products for potential applications in the cosmetic, pharmaceutical, or food industries represents a crucial goal. In the last decades, many researchers have focused their attention on the valorization of C. sativus by‐products [ref. 1].
Numerous studies have demonstrated diverse pharmacological effects of C. sativus, including antioxidant, anticancer, antidiabetic, antineurodegenerative, anti‐inflammatory, and antidepressant [ref. 7, ref. 8, ref. 9]. The bioactive potential of this species was attributed to several secondary metabolites belonging to the phytochemical classes of carotenoids and polyphenols. Stigmas are mainly rich in apocarotenoids such as crocin, crocetin, safranal, and picrocrocin. Tepals contain flavonoids, including different classes such as flavones, flavonols, and flavanones, anthocyanins, and lutein diesters [ref. 10, ref. 11]. Leaves are studied less compared to other plant parts. Their phytochemical composition comprises xanthones, flavonoids, isoflavonoids, and hydroxycinnamic acids [ref. 12].
Obtaining bioactive chemical components from plant sources depends on several factors, including the geographical location and the choice of the phenological stage of the plant, as well as the post‐harvest processes, mainly the methods applied for drying plant material, the extraction technique and the solvent [ref. 13, ref. 14, ref. 15, ref. 16, ref. 17].
A survey of recent literature revealed that saffron and C. sativus by‐products from some Moroccan regions such as Talioune, Oujda, and Boulmane have previously been investigated [ref. 18, ref. 19, ref. 20, ref. 21, ref. 22].
The present study was designed to evaluate the impact of the drying method on the phenolic content and the antioxidant activity of leaves and tepals by‐products from C. sativus collected in the locality of Ait Ouallal, Meknes, Morocco. The influence of drying methods on the phytochemical composition of C. sativus by‐products has been little investigated. Actually, few bibliographic data report studies on the effect of drying temperatures and air flows on the phenolic profile of floral bio‐residues composed by tepals, stamens, and styles; besides, research was carried out to compare the effect of different drying processes on the stability of anthocyanin extracts from tepals [ref. 6, ref. 23, ref. 24]. To our knowledge, similar studies have not been conducted previously on the leaves.
In this work, C. sativus leaves and tepals were subjected to two drying methods, namely freeze‐drying (FD) or lyophilization and air‐drying (AD); then, hydroalcoholic extracts were obtained from the plant material. The qualitative‐quantitative phenolic profile of C. sativus extracts was characterized by high‐performance liquid chromatography coupled to a photodiode array and electrospray ionization mass spectrometry (HPLC‐PDA/ESI‐MS) analysis. The antioxidant properties of the extracts were evaluated by different in vitro systems based on diverse mechanisms. In addition, the potential toxicity of C. sativus extracts was assessed by the Artemia salina lethality bioassay. Taking into account that the phytochemistry and biological potential of C. sativus from the Meknes region has not previously been investigated, it seemed interesting to extend the study to the stigmas as well.
Results and Discussion
Extraction Yields
The yields of C. sativus tepal, leaf, and stigma 70% MeOH extracts are reported in Table 1. The results showed that the yields varied depending on the plant part; higher extraction yields were found for stigma and tepal extracts, while the lowest yields were observed for leaf extracts.
TABLE 1: Yields of Crocus sativus tepal, leaf, and stigma hydroalcoholic extracts.
| Plant material | Extract | Yield % |
|---|---|---|
| Tepals freeze‐dried | Cs‐TFD | 46.75 |
| Tepals air‐dried | Cs‐TAD | 59.11 |
| Leaves freeze‐dried | Cs‐LFD | 28.05 |
| Leaves air‐dried | Cs‐LAD | 20.23 |
| Stigmas air‐dried | Cs‐SAD | 60.10 |
Note: The yields are referred to 100 g of dried plant material (dw).
Similarly to our results, in the work of Lahmass et al. carried out on ethanol extracts from different parts of C. sativus obtained by maceration for 24 h, stigma extract presented higher yield compared to leaf extract; nevertheless, these were far lower than those found in our study (28.76% and 4.3%) [ref. 20]. Our results agree with those previously reported by Ouahhoud et al. for extracts from C. sativus obtained by maceration for 24 h, three times, with 80% ethanol, showing higher extraction yields for tepals (69.45%) and stigma (64.57%) compared to that of leaf extract (31.19%) [ref. 18]. The results also agree with those of Benkerroum et al. for C. sativus tepal and stigma extracts obtained by maceration with 80% MeOH twice (45%–64% and 49.4%–66%, respectively) [ref. 25].
The comparison between the two drying methods highlighted different results for tepal and leaf extracts; in fact, for the tepal extracts, the yield of Cs‐TFD was slightly lower than that of Cs‐TAD, while for the leaf extracts Cs‐LFD and Cs‐LAD an inverse trend was observed.
Phytochemical Investigations
Determination of Total Phenolic Content
The total phenolic content (TPC) of tepal, leaf, and stigma hydroalcoholic extracts of C. sativus is reported in Table 2. The results highlighted significant differences between the TPC values in the different plant parts. Both the tepal extracts exhibited the highest phenolic content (65.94 ± 1.15 mg gallic acid equivalents [GAE]/g extract for Cs‐TFD and 57.01 ± 1.59 mg GAE/g extract for Cs‐TAD) compared to leaf and stigma extracts, which instead showed a quite similar quantity of total phenolics. The TPC of the extracts decreases in the following order: Cs‐TFD > Cs‐TAD > Cs‐SAD > Cs‐LFD > Cs‐LAD.
TABLE 2: Total phenolic content of Crocus sativus tepal, leaf, and stigma hydroalcoholic extracts.
| Crocus sativus extract | TPC (mg GAE/g extract) |
|---|---|
| Cs‐TFD | 65.94 ± 1.15cbdv202402967-tbl2-note-0001 |
| Cs‐TAD | 57.01 ± 1.59cbdv202402967-tbl2-note-0001 |
| Cs‐LFD | 41.26 ± 2.74cbdv202402967-tbl2-note-0001 |
| Cs‐LAD | 37.90 ± 0.57cbdv202402967-tbl2-note-0001 |
| Cs‐SAD | 42.88 ± 1.66cbdv202402967-tbl2-note-0001 |
Note: Values are expressed as the mean ± SD. Different superscript letters (a–d) indicate significant differences at p < 0.05 by Duncan’s test.
From the comparison of TPC values and the yields obtained for the extracts, it was highlighted that the stigma extract, exhibiting an extraction yield close to that of tepal extracts, showed a phenolic content comparable to that of the leaf extracts, whose extraction yields were approximately half and a third. Similar findings have been previously reported in other works [ref. 18, ref. 20].
Among the extracts obtained from C. sativus by‐products subjected to the two drying methods, a correspondence between extraction yields and TPC was observed only for leaf extracts, while for tepal extracts, greater quantities of phenolic compounds were extracted in Cs‐TFD, exhibiting a lower yield than Cs‐TAD.
Notably, the results of our investigations indicate that the drying method significantly affects the TPC of the plant material, FD being more effective than AD. Drying process conditions, such as temperature, air flow, and duration, can allow stability or alter the TPC of the plant material. Phenolic compounds might be degraded either by high temperature or by oxidation; therefore, a considerable amount of polyphenols might be lost during the drying process [ref. 6]. The favorable effect of the FD process is associated with the elimination of water from the plant material by sublimation rather than a long stay at room temperature or the use of high temperatures. This allows for less alteration of the physicochemical quality and the preservation of the active compounds [ref. 26].
The TPC highlighted in our extracts was found to be far higher than those reported in previous investigations. Lahmass et al. determined the TPC of ethanol extracts obtained by maceration at room temperature for 24 h from different air‐dried parts of C. sativus harvested in Oujda, located in the north‐east of Morocco, resulting in 23.32 µg GAE/mg extract 16.63 µg GAE/mg extract for leaf and stigma extract, respectively [ref. 20]. In another study, the TPC of ethanol extracts obtained from air‐dried leaves and stigmas of Indian C. sativus were 5.62 and 8.28 mg/g, respectively [ref. 27]. On the other hand, the amount of TPC of the MeOH extract obtained by maceration at 35°C for 24 h of dried tepals from C. sativus collected in Taliouine, Morocco, was 65.34 mg GAE/g extract, superimposable to our values [ref. 19]. Similar TPC values were also found by Ouahhoud et al. in 80% ethanol extracts obtained by maceration for 24 h, three times, of oven dried (37°C) tepals, leaves, and stigmas from C. sativus cultivated in Taliouine (64.66 µg GA eq/mg extract, 38.56 µg GA eq/mg extract, and 34.41 µg GA eq/mg extract, respectively) [ref. 18]. In a recent work published by Benkerroum et al., tepals and stigmas from C. sativus cultivated in Taliouine shade‐dried at room temperature and extracted by maceration with 80% MeOH contained phenolic compounds at the rate of 64.73 and 56.11 mg GAE/g extract [ref. 25].
It should be underlined that the comparison of our results with previous studies highlighted that the application of ultrasound at 50°C allowed efficient phenolic extraction in much shorter times than maceration.
Phenolic Characterization by HPLC‐PDA/ESI‐MS Analysis
Phenolic profile analyses were carried out using HPLC‐PDA/ESI‐MS (Figure 1). As listed in Table 3, a total of 31 phenolic compounds were detected according to λ max, retention times, mass spectrometry, and literature data. Compounds were mainly assigned to flavonoids, such as kaempferol, quercetin, and isorhamnetin derivatives, with different isomers present among tepals, leaves, and stigmas; while leaf extracts were the only ones in which the presence of different acetophenone derivatives was highlighted.
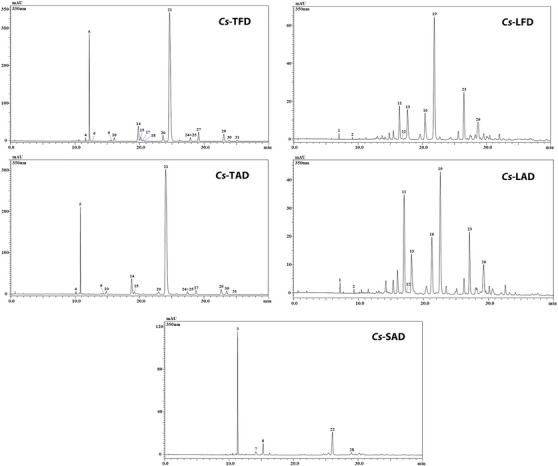
TABLE 3: Semi‐quantification of phenolic compounds in Crocus sativus tepal, leaf, and stigma hydroalcoholic extracts through LC‐PDA/ESI‐MS analysis.
| Peak | Compound | tR (min) | UV max (nm) | [M−H]− | Cs‐TFD | Cs‐TAD | Cs‐LFD | Cs‐LAD | Cs‐SAD | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Trihydroxy‐methoxyacetophenone‐diglucoside | 7.03 | 286, 335sh | 545cbdv202402967-tbl3-note-0002, 521, 197 | –— | — | x× | × | — | — |
| 2 | Unknown | 9.11 | 287, 335sh | 631cbdv202402967-tbl3-note-0002, 607, 197 | — | — | × | × | — | — |
| 3 | Kaempferol triglucoside | 11.33 | 265, 346 | 771, 609, 447, 285 | — | — | — | — | 5.02 ± 0.039 | [ref. 28] |
| 4 | Myricetin diglucosyl‐rhamnoside | 11.65 | 254, 351 | 787, 625, 463 | × | × | — | — | — | [ref. 22] |
| 5 | Kaempferol triglucoside | 12.23 | 265, 346 | 771, 609, 285 | 11.65 ± 0.003 | 7.39 ± 0.012 | — | — | — | [ref. 29] |
| 6 | Kaempferol rutinosyl‐hexoside | 12.74 | 264, 347 | 755 | × | — | — | — | — | [ref. 22] |
| 7 | Unknown | 14.14 | 250 | 375, 167 | — | — | — | — | × | — |
| 8 | Kaempferol triglucoside | 15.28 | 265, 341 | 771, 609, 447, 285 | — | — | — | — | 0.66 ± 0.007 | [ref. 28] |
| 9 | Kaempferol glucosyl‐(6″‐acetylgalactoside)‐hexoside | 15.62 | 265, 348 | 813, 651 | × | × | — | — | — | [ref. 22] |
| 10 | Kaempferol sophoroside | 16.08 | 264, 344 | 609, 447 | 0.81 ± 0.001 | 0.57 ± 0.003 | — | — | — | [ref. 22] |
| 11 | Quercetin triglucoside | 16.45 | 272, 334 | 787, 625, 463, 301 | — | — | 1.68 ± 0.033 | 4.20 ± 0.040 | — | — |
| 12 | 4′,6′‐Dihydroxy‐2′‐methoxyacetophenone 6′‐glucoside | 17.23 | 284, 320sh | 367cbdv202402967-tbl3-note-0002, 343, 181 | — | — | × | × | — | [ref. 30] |
| 13 | 2,3,4‐Trihydroxy‐6‐methoxyacetophenone‐3‐β‐d‐glucopyranoside | 17.71 | 285, 329sh | 383cbdv202402967-tbl3-note-0002, 359, 197 | — | — | × | × | — | [ref. 30] |
| 14 | Quercetin sophoroside | 19.81 | 255, 352 | 625, 463, 301 | 5.18 ± 0.018 | 4.74 ± 0.000 | — | — | — | [ref. 22] |
| 15 | Quercetin sophoroside | 20.19 | 264, 343 | 625, 463 | 1.18 ± 0.009 | 0.41 ± 0.002 | — | — | — | [ref. 22] |
| 16 | Isorhamnetin sophorosyl‐rhamnoside | 20.44 | 253, 351 | 785, 315 | — | — | 2.00 ± 0.004 | 2.74 ± 0.006 | — | [ref. 28] |
| 17 | Kaempferol sophoroside | 20.50 | 265, 339 | 609, 447 | 0.14 ± 0.003 | — | — | — | — | [ref. 22] |
| 18 | Luteolin (or kaempferol) malonyl‐dihexoside | 20.72 | 265, 347 | 695, 447 | × | — | — | — | — | — |
| 19 | 6‐Hydroxyluteolin 7‐glucoside | 21.87 | 268, 349 | 447 | — | — | 5.68 ± 0.007 | 3.43 ± 0.007 | — | [ref. 31] |
| 20 | Isorhamnetin sophoroside | 23.66 | 265, 343 | 639, 477, 315 | 2.11 ± 0.002 | 0.54 ± 0.007 | — | — | — | [ref. 22] |
| 21 | Kaempferol sophoroside | 24.67 | 265, 346 | 609, 447, 285 | 71.44 ± 0.060 | 63.84 ± 0.113 | — | — | — | [ref. 22] |
| 22 | Kaempferol sophoroside | 26.02 | 265, 347 | 609, 447, 285 | — | — | — | — | 2.57 ± 0.007 | [ref. 28] |
| 23 | Kaempferol 3,7,4′‐triglucoside | 26.50 | 271, 335 | 771 | — | — | 2.64 ± 0.011 | 2.26 ± 0.005 | — | [ref. 29] |
| 24 + 25 | Kaempferol rutinoside + quercetin hexoside | 27.87 | 264, 347 | 593, 463, 285 + 463, 301 | 0.97 ± 0.016 | 0.54 ± 0.008 | — | — | — | [ref. 22] |
| 26 | Trihydroxy‐methoxyacetophenone‐acetylglucopyranoside | 28.65 | 285, 329sh | 401, 359, 197 | — | — | × | × | — | — |
| 27 | Isorhamnetin rutinoside | 29.14 | 253, 353 | 623, 477, 315 | 2.48 ± 0.019 | 0.76 ± 0.000 | — | — | — | [ref. 22] |
| 28 | Vanillic acid | 29.29 | 287 | 169cbdv202402967-tbl3-note-0003 | — | — | — | — | × | [ref. 32] |
| 29 | Kaempferol glucoside | 33.05 | 265, 346 | 447, 285 | 2.53 ± 0.009 | 1.61 ± 0.008 | — | — | — | [ref. 22] |
| 30 | Kaempferol‐(6″‐acetyl‐glucoside)‐glucoside | 33.88 | 266, 347 | 651, 489, 285 | 0.17 ± 0.005 | 0.83 ± 0.009 | — | — | — | [ref. 22] |
| 31 | Isorhamnetin glucoside | 35.13 | 267, 347 | 477, 315 | 0.25 ± 0.007 | × | — | — | — | [ref. 22] |
Note: Quantification of phenolic compounds is reported in mg/g of dried extract ± SD (n = 3). ×: detected, but not quantified; sh: wavelength shoulder.
Detected as [M+Na]+.
Detected as [M+H]+.
In general, among the understudied extracts, tepal ones were the quali‐quantitatively richest, especially in terms of kaempferol derivatives. In particular, a total of 16 compounds were detected in Cs‐TFD and 13 in Cs‐TAD; for almost all compounds detected in tepal extracts, a greater amount of these was found in Cs‐TFD. Among the phenolic compounds identified, the kaempferol‐sophoroside isomer turned out to be the most abundant one in both tepal extracts (71.44 ± 0.060 and 63.84 ± 0.113 mg/g, for Cs‐TFD and Cs‐TAD, respectively), followed by kaempferol triglucoside (11.65 ± 0.003 and 7.39 ± 0.012 mg/g, for Cs‐TFD and Cs‐TAD, respectively).
In both leaf extracts, Cs‐LFD and Cs‐LAD, a superimposable qualitative profile was highlighted, with a total of nine compounds detected. Differently from tepal extracts, 6‐hydroxyluteolin 7‐glucoside was the most abundant compound in Cs‐LFD extract (5.68 ± 0.007 mg/g), while in Cs‐LAD was quercetin triglucoside (4.20 ± 0.040 mg/g).
Finally, the analysis of stigma extract Cs‐SAD led to the identification of five phenolic compounds, kaempferol triglucoside being the main component detected in the extract (5.02 ± 0.039 mg/g).
A. salina Leach Lethality Bioassay
The assessment of toxicity in medicinal plant extracts plays a crucial role in ensuring their safety and efficacy. The microcrustacean A. salina is widely used as a model organism for the preliminary estimation of the toxicity of plant extracts, as it offers a number of significant advantages such as ease of cultivation, cost‐effectiveness, rapid response, and reproducibility [ref. 33]. Within this perspective, the A. salina lethality bioassay was used for toxicity assessment of C. sativus tepal, leaf, and stigma extracts. Based on Clarkson’s toxicity criterion applied for the assessment of the degree of toxicity, the results obtained after 24 h of incubation showed no toxicity against brine shrimp larvae for all the extracts, with LC50 values over 1000 µg/mL [ref. 34].
Antioxidant Activity
Antioxidants are considered health‐supporting compounds due to their ability to protect biological systems from oxidative damage produced by reactive oxygen species (ROS). Plant‐derived antioxidants have received a great deal of attention because they play a pivotal role in the prevention and/or treatment of several disorders related to oxidative stress, such as cardiovascular, inflammatory, and neurodegenerative diseases, diabetes, cancers, as well as aging [ref. 5, ref. 35].
As is known, various methods based on different mechanisms should be used to estimate the antioxidant properties of plant extracts since numerous activities are involved in the process of oxidation inhibition by antioxidant compounds [ref. 36]. Antioxidants have been traditionally divided into two classes: primary or chain‐breaking antioxidants (mainly acting by ROS/RNS scavenging) and secondary or preventative antioxidants (usually acting by transition metal ion chelation) [ref. 37]. Depending on the mechanism of the chemical reactions involved, antioxidant assays are based on electron transfer (ET), hydrogen atom transfer (HAT), or chelation of transition metals. The ET mechanism involves a redox reaction with an oxidant as an indicator of the reaction endpoint. HAT‐based methods are generally composed of a synthetic free radical generator, an oxidizable molecular probe, and an antioxidant [ref. 38]. In addition to ET and HAT mechanisms, estimating the antioxidant capacity of an extract or compound of metal ion chelation can also be considered one of the most important mechanisms of action of secondary antioxidants. Metal chelators reduce the amount of available transition metals, thus decreasing the extent of hydroxyl radicals generated by the Fenton reaction and limiting metal ion‐induced lipid oxidation [ref. 36]. Recent investigations have demonstrated that transition metals such as Fe2+ and Cu2+ are responsible for the pathogenesis of several diseases, including neurodegenerative (Alzheimer’s and Parkinson’s) and cardiovascular diseases [ref. 38].
Among the most important groups of plant‐based antioxidants are phenolic compounds, which have one or more aromatic rings with one or more hydroxy groups; these compounds have attracted increasing attention due to their powerful antioxidant properties and their beneficial effects in the prevention of various oxidative stress‐related diseases [ref. 39]. Phenolic compounds can act as hydrogen or electron donors, capable of stabilizing unpaired electrons (radicals) and chelating transition metal ions, resulting from different conjugations and varying numbers of hydroxyl groups [ref. 40].
In the present work, to determine and compare the antioxidant efficacy of the hydroalcoholic extracts of C. sativus tepals and leaves by‐products subjected to different drying procedures, along with stigma extract, three in vitro tests were performed, namely the 2,2‐diphenyl‐1‐picrylhydrazyl (DPPH) and the reducing power assays to evaluate the primary antioxidant properties, and the ferrous ion chelating activity assay to assess the secondary antioxidant properties.
Regarding the DPPH test, the free radical scavenging activity of the extracts was determined by the capacity to reduce the stable free radical DPPH• to the non‐radical form DPPH‐H. This reduction, based on the dual mechanism of HAT and ET, is indicated by the color change of the solution from purple to yellow. The degree of discoloration shows the efficiency of the antioxidant activity of plant extracts [ref. 27].
The results of the DPPH test showed that the extracts exhibited radical scavenging activity in the range of concentrations tested (0.0625–2 mg/mL), which augments with the increase of extract concentration. As depicted in Figure 2, both the leaf extracts exhibited higher activity than those from tepals and stigmas, the latter displaying moderate scavenging activity only, as compared to the standard BHT. At the highest concentration tested, the activity of the extracts ranged between 17.9% (Cs‐SAD) and 93.2% (Cs‐LFD), the latter close to that of standard BHT (95.8%). Furthermore, the extracts obtained from freeze‐dried plant material, namely Cs‐TFD and Cs‐LFD, were shown to display greater scavenging activity compared to those from air‐dried plant material Cs‐TAD and Cs‐LAD. The IC50 values calculated for C. sativus extracts confirmed these results, demonstrating the best activity for Cs‐LFD (0.93 ± 0.05 mg/mL), and the lowest activity for Cs‐SAD (IC50 > 2 mg/mL) (Table 4).
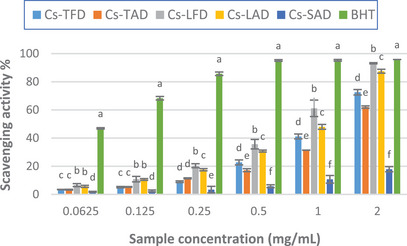
TABLE 4: DPPH radical scavenging activity, reducing power, and ferrous ion chelating activity of Crocus sativus tepal, leaf, and stigma hydroalcoholic extracts.
| Crocus sativus extract | DPPH scavenging activity, IC50 (mg/mL) | Reducing power (ASE/mL) | Chelating activity, IC50 (mg/mL) |
|---|---|---|---|
| Cs‐TFD | 1.34 ± 0.05cbdv202402967-tbl4-note-0001 | 18.17 ± 2.45cbdv202402967-tbl4-note-0001 | 0.42 ± 0.02cbdv202402967-tbl4-note-0001 |
| Cs‐TAD | 1.60 ± 0.01cbdv202402967-tbl4-note-0001 | 9.22 ± 0.52cbdv202402967-tbl4-note-0001 | 0.88 ± 0.19cbdv202402967-tbl4-note-0001 |
| Cs‐LFD | 0.93 ± 0.05cbdv202402967-tbl4-note-0001 | 12.27 ± 3.92cbdv202402967-tbl4-note-0001 | 0.44 ± 0.04cbdv202402967-tbl4-note-0001 |
| Cs‐LAD | 1.06 ± 0.01cbdv202402967-tbl4-note-0001 | 12.04 ± 1.05cbdv202402967-tbl4-note-0001 | 0.81 ± 0.06cbdv202402967-tbl4-note-0001 |
| Cs‐SAD | ND | 12.15 ± 2.84cbdv202402967-tbl4-note-0001 | 0.92 ± 0.06cbdv202402967-tbl4-note-0001 |
| Standard | BHT | BHT | EDTA |
| 0.06 ± 0.01cbdv202402967-tbl4-note-0001 | 1.44 ± 0.02cbdv202402967-tbl4-note-0001 | 0.02 ± 6.86E−05cbdv202402967-tbl4-note-0001 |
Note: The results are expressed as the mean ± SD (n = 3). Different superscript letters (a–e) within the same column indicate significant differences at p < 0.05 by Duncan’s test.
Our results agree with those previously reported in other studies, which showed good free radical scavenging activity for C. sativus leaf and tepal extracts and weak activity for the stigma extract [ref. 5, ref. 20, ref. 25]. Differently, in the study reported by Baba et al. stigma extracts exhibited the highest activity, whereas the leaf extracts resulted in much less active [ref. 27].
As demonstrated by both spectrophotometric determination and HPLC‐PDA/ESI‐MS analysis, C. sativus tepal extracts, Cs‐TFD and Cs‐TAD, are richer in phenolic compounds, mainly flavonoids such as kaempferol, quercetin, and isorhamnetin derivatives. Nonetheless, the best radical scavenging activity was observed for the leaf extracts; this could be explained by considering their phenolic profile, which was different, both qualitatively and qualitatively. Among the phenolics identified in Cs‐LFD, 6‐hydroxyluteolin 7‐glucoside resulted in the most abundant; it was found in greater amount than Cs‐LAD, while it was not detected in the other extracts. The good free radical scavenging properties of 6‐hydroxyluteolin 7‐glucoside have been previously reported; the presence of this compound could explain, almost in part, the observed results [ref. 41]. Furthermore, among detected phenolic compounds, the presence of four acetophenone derivatives was highlighted exclusively in Cs‐LFD and Cs‐LAD; nonetheless, previous studies have shown moderate to mild DPPH free radical scavenging properties for several acetophenone glycosides [ref. 42, ref. 43].
The reducing power of C. sativus extracts was evaluated by the potassium ferricyanide method, an ET‐based assay measuring the reduction of Fe(III) to Fe(II). Compounds with reduction potential react with potassium ferricyanide (Fe3+) to form potassium ferrocyanide (Fe2+), which subsequently reacts with ferric chloride to form a ferric–ferrous complex. The color of the solution changes from yellow to different shades of green and blue, having an absorption maximum of 700 nm [ref. 44]. An increase in absorbance is proportional to an increase in the reducing power [ref. 45, ref. 46].
The results of the assay, depicted in Figure 3 and Table 4, showed that all the extracts displayed moderate reducing power as compared to the standard. Among the extracts, Cs‐TAD exhibited the best activity, slightly higher than that of Cs‐TFD, with an absorbance value of 0.84 ± 0.10 at the maximum tested concentration (2 mg/mL), approximately three times lower than that of the standard BHT (2.40 ± 0.10), and an ASE/mL value of 9.22 ± 0.52.
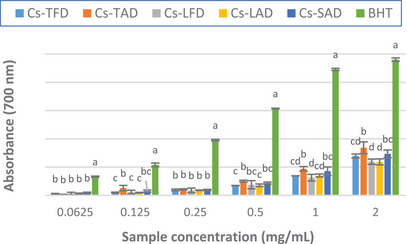
In agreement with our results, in the study of Baba et al. moderate reducing power was observed for all the C. sativus extracts tested, greater for the stigma extracts than the leaves ones [ref. 27]. Our results disagree with those previously reported by Lahmass et al., who showed good reducing power for C. sativus stigma extracts, much greater than those of the leaves [ref. 20].
The chelating properties of the extracts were assessed by determining the extent of Fe2+–ferrozine complex formation. Chelation of this metal ion with ferrozine leads to the formation of a red‐colored complex; in the presence of chelating agents, the formation of the complex is stopped, and the red color decreases. Measuring the rate of color reduction allows the estimation of the chelating activity [ref. 5].
The results of the assay showed that the extracts exhibited good chelating properties compared to the standard EDTA. As depicted in Figure 4, for almost all concentrations tested, a significant difference was observed between the two diverse drying methods; indeed, the activity of the extracts from freeze‐dried tepals and leaves was higher than those from air‐dried plant material, reaching approximately 85% for both extracts at the highest concentration tested. The IC50 values calculated for the extracts also confirmed these results; in fact, Cs‐TFD and Cs‐LFD showed the lowest IC50, which resulted in superimposable (0.42 ± 0.02 and 0.44 ± 0.04 mg/mL, respectively) and about half that of Cs‐TAD and Cs‐LAD (Table 4).
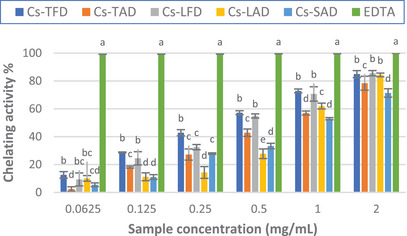
The stigma extract Cs‐SAD was found to display the lowest activity, reaching approximately 71% at the highest concentration tested; nonetheless, the IC50 calculated for the extract was close to that of Cs‐TAD and Cs‐LAD.
The obtained results do not agree with those previously reported by Ouahhoud et al., who found the best ferrous ion chelating activity for 80% ethanol extract of C. sativus tepals, followed by that of the stigmas, whereas the leaf extract resulted in the less active [ref. 18]. Our results also differ from those of Sánchez‐Vioque et al., who found low ferrous ion chelating activity for C. sativus leaf extract and negligible activity for the tepal extract [ref. 5].
Despite the different TPC, as well as the qualitative–quantitative phenolic profiles, both Cs‐TFD and Cs LFD showed comparable chelating activity, which was higher than that of the other extracts. Many studies have confirmed that flavonoids can behave as antioxidants because of their chelating properties [ref. 47]. The presence of 6‐hydroxyluteolin 7‐glucoside could partly explain the good chelating properties of Cs‐LFD; indeed, it has been previously demonstrated that 6‐hydroxyluteolin is an effective chelator of Fe2+ ions [ref. 48].
Overall, the results of antioxidant assays showed that the extracts from C. sativus tepals and leaves by‐products harvested from the Meknes region could be exploited as natural sources of phenolic antioxidants displaying good radical scavenging and Fe2+ chelating properties and that FD improves the recovery of antioxidant compounds, which are partially lost with the AD process. Taking into account the better antioxidant activity displayed by the leaf extracts, which showed a lower phenolic content than tepal ones, it cannot be ruled out that phenolics are not the only ones responsible for the highlighted antioxidant properties and that other polar antioxidant compounds belonging to different chemical classes may be contained in the phytocomplexes, such as carotenoids, present in both leaves and tepals of C. sativus, which are well‐known to be effective antioxidants, in particular, xanthophylls such as lutein, which are more polar than carotenes [ref. 11, ref. 25, ref. 49]. Polysaccharides, previously detected in C. sativus leaves and tepals, may also contribute to the observed activity [ref. 12, ref. 50, ref. 51].
The results of our investigations add information to previous studies on the influence of drying methods on the phenolic recovery from C. sativus floral by‐products and provide information on this topic relating to leaves for the first time [ref. 6, ref. 23, ref. 24]. These findings further support the studies aimed at the valorization of C. sativus by‐products, indicating freeze‐drying as a strategy to increase the production of natural antioxidants, which can be considered as active ingredients in food supplements, functional foods, and beverages in pharmaceutical preparations and cosmetic formulations. This could further improve the use and exploitation of these by‐products, becoming a valuable source of income while reducing biowaste, thus increasing the profitability and sustainability of saffron production.
Conclusions
In this work, the influence of the drying method on the phenolic content and the antioxidant activity of leaves and tepals by‐products from C. sativus collected in the locality of Ait Ouallal, Meknes, Morocco, was investigated. Since the plant growing in this area had not previously been studied, it seemed interesting to include the stigmas in our research, to provide additional information on this commercially precious spice. The comparative study carried out on the hydroalcoholic extracts of C. sativus tepals and leaves clearly showed that the use of FD allows the preserve higher quantities of antioxidant compounds, which are instead partly lost with the AD process. Overall, the results of the different antioxidant assays highlighted both primary and secondary antioxidant activity for the extracts, which, therefore, could be exploited as a valuable source of antiradical and iron‐chelating compounds. The good chelating properties of the extracts from freeze‐dried plant material deserve particular interest. At last, the A. salina lethality bioassay showed the absence of toxicity for all the extracts, indicating their potential safety.
The results of the present study provide further insights into the knowledge of efficient strategies for the recovery of natural antioxidant biomolecules from C. sativus tepals and leaves by‐products that may be applied in the food, pharmaceutical, and cosmeceutical industries.
Experimental Section
Chemicals and Reagents
LC/MS‐grade water (H2O), acetonitrile (ACN), quercetin 3‐O‐rhamnoside, luteolin‐7‐O‐glucoside, Folin–Ciocâlteu reagent, sodium carbonate, gallic acid, DPPH, butylated hydroxytoluene (BHT), potassium hexacyanoferrate(III), iron(III) chloride hexahydrate, l‐ascorbic acid, sodium phosphate monobasic monohydrate, potassium phosphate dibasic, trichloroacetic acid, iron(II) chloride, 3‐(2‐pyridyl)‐5,6‐diphenyl‐1,2,4‐triazine‐4′,4″‐disulfonic acid sodium salt (ferrozine), and ethylenediaminetetraacetic acid (EDTA) disodium salt dihydrate, were obtained from Merck Life Science (Merck KGaA, Darmstadt, Germany). Methanol (MeOH) was purchased from Omniascientific snc (Messina, Italy).
Plant Material
The tepals and stigmas of C. sativus L. were harvested in November 2022; the leaves were collected in February 2023 in the locality of Meknes, specifically in Ait Ouallal region (33°50′28.536″ N–5°35′43.361″ W). The plant taxonomic identification was performed by Prof. Ouafae Benkhnigue at the Scientific Institute of Rabat, Morocco. A voucher specimen under the number RAB114620 has been deposited in the herbarium of the University Mohammed V, Rabat, Morocco.
Drying Procedures
For tepals and leaves two different methods were utilized, namely FD and AD. For FD process, tepals and leaves were immediately frozen after collection and successively freeze‐dried by using a lyophilizer model Alpha 1‐4 LD plus (Christ, Germany) at −53°C; the AD process was performed indoors at room temperature for 1 week. The stigmas were air‐dried indoors for 4 days.
Extraction Procedure
The dried plant material was ground; then, 1 g of each sample was subjected to preventive maceration for 1 h at 25°C using a MeOH–H2O mixture (70:30 v/v), with a sample‐to‐solvent ratio of 1:10 (w/v). The extraction was performed using 70% MeOH (plant material‐to‐solvent ratio of 1:10 w/v) in an ultrasonic bath at 50°C for 15 min; the procedure was repeated three times. After filtration, the extraction solutions were combined and centrifuged at 3000 rpm for 10 min at room temperature; then, the supernatants were evaporated to dryness by high‐performance solvent evaporation system EZ‐2 Plus (Genevac Ltd., Ipswich, UK). The yields of the extracts, referred to 100 g of dried plant material, are reported in Table 1.
Phytochemical Investigations
Determination of TPC
The TPC of C. sativus tepal, leaf, and stigma hydroalcoholic extracts was estimated according to the colorimetric method described by Gao et al. [ref. 52]. Briefly, a volume of 0.1 mL of each sample solution was mixed with 0.2 mL of Folin–Ciocâlteu reagent; then, 2 mL of distilled water and 1 mL of 15% sodium carbonate were added. The mixture was kept for 2 h at room temperature in the dark; afterward, the absorbance was measured at 765 nm by UV‐1601 spectrophotometer (Shimadzu, Milan, Italy). For the quantitative estimation of total polyphenols, the calibration curve of gallic acid was used. The TPC was expressed as mg GAE/g of extract (dw) ± standard deviation (SD).
Phenolic Profile Characterization by HPLC‐PDA/ESI‐MS Analysis
The tepal, leaf, and stigma hydroalcoholic extracts of C. sativus were analyzed by HPLC‐PDA/ESI‐MS to characterize their phenolic profile [ref. 22, ref. 28–ref. 32].
Sample preparation: The dried extracts were redissolved in 70% MeOH (10 mg/mL). For the chromatographic separation, an injection volume of 2 µL was employed, and the analysis was performed in triplicate.
HPLC/MS analytical condition: Chromatographic analysis was accomplished by means of a Shimadzu HPLC system (Kyoto, Japan) equipped with a CBM‐20A controller, two LC‐20AD dual‐plunger parallel‐flow pumps, a DGU20A5R degasser, a CTO‐20AC column oven, a SIL‐30AC autosampler, an SPD‐M20A photodiode array detector, and an LCMS‐2020 single quadrupole mass spectrometer, with the employment of ESI source operated in negative and positive ionization modes. Chromatographic separations were carried out on Ascentis Express RP C18 columns (150 × 2.1 mm; 2.7 µm) (Merck Life Science, Merck KGaA, Darmstadt, Germany). The employed mobile phase was composed of two solvents: water (Solvent A) and ACN (Solvent B) both acidified with 0.1% of formic acid v/v. The flow rate was set at 0.5 mL/min, under gradient elution 0 min—0% B, 10 min—10% B, 20 min—11% B, 30 min—15% B, 50 min—18% B, 65 min—23% B, 70 min—100% B, 75 min—100% B. PDA was applied in the range of 190–400 nm and monitored at a wavelength of 350 nm (sampling frequency: 40, time constant: 0.080 s). MS conditions were as follows: scan range and the scan speed were set at a mass‐to‐charge ratio (m/z) 100–1600 and 7500 amu/s, respectively; event time: 0.3 s, nebulizing gas (N2) flow rate: 1.5 L/min, drying gas (N2) flow rate: 15 L/min, interface temperature: 350°C, heat block temperature: 300°C, DL temperature: 300°C, DL voltage: 1 V, interface voltage: M‐ 4.5 kV.
Standards: Calibration curves of two standards (quercetin 3‐O‐rhamnoside and luteolin‐7‐O‐glucoside) were employed for the quantification of the flavonoid content in sample extracts. Each analysis was performed in 6 repetitions. Data acquisition was performed by Shimadzu LabSolution software ver. 5.99. Quercetin 3‐O‐rhamnoside (0.1, 1, 10, 50, 100 ppm), y = 8054x + 27 465, R2 = 0.9997, LOD = 0.034, LOQ = 0.103; luteolin‐7‐O‐glucoside (0.1, 1, 10, 50, 100 ppm), y = 12 881x + 54 935, R2 = 0.9996, LOD = 0.016, LOQ = 0.048.
A. Salina Leach Lethality Bioassay
The toxicity of C. sativus tepal, leaf, and stigma hydroalcoholic extracts was investigated by performing the brine shrimp (A. salina Leach) lethality bioassay, according to the protocol previously reported by Meyer et al., with some modifications [ref. 53]. A. salina cysts were placed in a hatchery dish containing artificial seawater (32 g sea salt/L distilled water) and incubated for hatching under a 60 W lamp, at a temperature of 24°C–26°C. Twenty‐four hours after hatching, active nauplii free from eggshells were collected from the brighter portion of the hatchery dish. For the assay, ten brine shrimp larvae were placed in plates containing 5 mL of artificial seawater mixed with different amounts of the extracts appropriately dissolved to obtain final concentrations in the range 10–1000 µg/mL. After incubation at 24°C–26°C for 24 h, live larvae were counted; then, the median lethal concentration (LC50) values were estimated. Three replicates of each sample concentration were tested. To assess the toxicity level of the extracts, Clarkson’s toxicity criterion was used [ref. 34].
Antioxidant Activity
Free Radical Scavenging Activity
The DPPH assay was used to determine the free radical scavenging activity of C. sativus tepal, leaf, and stigma hydroalcoholic extracts, according to the method of Ohnishi et al. [ref. 54]. For each extract, concentrations ranging from 0.0625 to 2 mg/mL were prepared; a 0.5 mL aliquot of each sample solution was mixed with 3 mL of DPPH methanol solution (0.1 mM). After incubation of the mixture for 20 min in the dark at room temperature, the absorbance was read at 517 nm using a model UV‐1601 spectrophotometer (Shimadzu). For the control, 0.5 mL solvent was used instead of the sample solution. BHT was used as the reference standard. The results are expressed as scavenging activity (%). The mean 50% inhibitory concentration (IC50) values were also determined.
Reducing Power
The evaluation of Fe3+–Fe2+ transformation by the potassium ferricyanide method was used to assess the reducing power of C. sativus extracts [ref. 55]. For each extract, concentrations ranging from 0.0625 to 2 mg/mL were prepared. A mixture containing 1 mL of each sample solution, 2.5 mL of phosphate buffer (0.2 M, pH 6.6), and 2.5 mL of 1% potassium ferricyanide was prepared and incubated for 20 min at 50°C. After rapid cooling, 2.5 mL of 10% trichloroacetic acid was added; then, the mixture was centrifuged at 3000 rpm for 10 min. A volume of 2.5 mL of the supernatant was taken and mixed with 2.5 mL of distilled water and 0.5 mL of 0.1% ferric chloride. After 10 min incubation at room temperature and in the dark, the absorbance of the samples was measured at 700 nm. BHT and ascorbic acid were used as reference standards. The results are expressed as mean absorbance values ± SD and ascorbic acid equivalent (ASE/mL) ± SD.
Ferrous Ion (Fe2+) Chelating Activity
The Fe2+ chelating activity of C. sativus extracts was estimated according to the method reported by Kumar et al. [ref. 56]. For each extract, concentrations ranging from 0.0625 to 2 mg/mL were prepared. An aliquot of 1 mL of each sample solution was mixed with 0.5 mL methanol and 0.05 mL of ferrous chloride (2 mM). Then, 0.1 mL of ferrozine solution (5 mM) was added and the reaction mixture was incubated for 10 min in the dark at room temperature. Afterward, the absorbance of the samples was read at 562 nm by spectrophotometer. The EDTA was used as the reference standard. The results are expressed as the mean inhibition of the ferrozine–Fe2+ complex formation (%) ± SD and IC50 ± SD.
Statistical Analysis
The results of TPC determination and antioxidant assays were obtained from the average of three independent experiments. The data were analyzed by one‐way analysis of variance (ANOVA), followed by Duncan’s multiple range test (DMRT), using the software SPSS v.21. p < 0.05 were considered statistically significant.
Author Contributions
Soukaina Abou‐Wakil: Conceptualization, Investigation, Data curation, Writing – original draft. Francesco Cacciola: Conceptualization, Investigation, Data curation, Supervision, Writing – original draft, Writing – review and editing. Maria Fernanda Taviano: Conceptualization, Investigation, Data curation, Supervision, Writing – original draft, Writing – review and editing. Mohamed Rochd: Investigation, Data curation. Roberto Laganà Vinci: Investigation, Writing – original draft. Federica Davì: Investigation. Fatima Housti: Data curation,, Writing – review and editing. Luigi Mondello: Data curation, Writing – review and editing. Nicola Micale: Data curation, Writing – review and editing. Natalizia Miceli: Data curation, Writing – review and editing. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Saffron (Crocus sativus L.), the King of Spices: An Overview,”. Scientia Horticulturae, 2020
- Effect of Corm Size, Water Stress and Cultivation Conditions on Photosynthesis and Biomass Partitioning During the Vegetative Growth of Saffron (Crocus sativus L.),”. Industrial Crops and Products, 2012
- 3 G. L. Alonso , A. Zalacain , and M. Carmona , “Saffron,”in Handbook of Herbs and Spices, 2nd ed., ed. K.V. Peter (Woodhead Publishing, 2012), 469–498.
- Crocus sativus L. Stigmas and Byproducts: Qualitative Fingerprint, Antioxidant Potentials and Enzyme Inhibitory Activities,”. Food Research International, 2018. [PubMed]
- In Vitro Antioxidant and Metal Chelating Properties of Corm, Tepal and Leaf from Saffron (Crocus sativus L.),”. Industrial Crops and Products, 2012
- Preservation of Saffron Floral Bio‐Residues by Hot Air Convection,”. Food Chemistry, 2013. [PubMed]
- A Comprehensive Review of the Pharmacological Potential of Crocus sativus and Its Bioactive Apocarotenoids,”. Biomedicine & Pharmacotherapy, 2018. [PubMed]
- Anti‐inflammatory, Antioxidant, and Immunomodulatory Effects of Crocus sativus L. and Its Main Constituents,”. Phytotherapy Research, 2016. [PubMed]
- Main Chemical Compounds and Pharmacological Activities of Stigmas and Tepals of ‘Red Gold’ Saffron,”. Trends in Food Science & Technology, 2016
- Comprehensive Extraction and Chemical Characterization of Bioactive Compounds in Tepals of Crocus sativus L.,”. Molecules, 2023. [PubMed]
- Identification and Quantification of Flavonols, Anthocyanins and Lutein Diesters in Tepals of Crocus sativus by Ultra Performance Liquid Chromatography Coupled to Diode Array and Ion Trap Mass Spectrometry Detections,”. Industrial Crops and Products, 2013
- Pharmacological Potential and Chemical Composition of Crocus sativus Leaf Extracts,”. Molecules, 2022
- Review on Drying Methods for Herbal Plants,”. Materials Today: Proceedings, 2022
- Drying Methodology Effect on the Phenolic Content, Antioxidant Activity of Myrtus communis L. Leaves Ethanol Extracts and Soybean Oil Oxidative Stability,”. BMC Chemistry, 2021. [PubMed]
- Different Techniques for Extraction and Micro/Nanoencapsulation of Saffron Bioactive Ingredients,”. Trends in Food Science & Technology, 2019
- The Effects of Geographical Origin and Virus Infection on the Saffron (Crocus sativus L.) Quality,”. Food Chemistry, 2019. [PubMed]
- Quantification of Saffron (Crocus sativus L.) Metabolites Crocins, Picrocrocin and Safranal for Quality Determination of the Spice Grown Under Different Environmental Moroccan Conditions,”. Scientia Horticulturae, 2009
- Antioxidant Activity, Metal Chelating Ability and DNA Protective Effect of the Hydroethanolic Extracts of Crocus sativus Stigmas, Tepals and Leaves,”. Antioxidants, 2022. [PubMed]
- Chemical Characterization and Antioxidant Compounds of Flower Parts of Moroccan Crocus sativus L.,”. Journal of the Saudi Society of Agricultural Sciences, 2019
- Determination of Antioxidant Properties of Six By‐Products of Crocus sativus L. (Saffron) Plant Products,”. Waste and Biomass Valorization, 2017
- Identification of Compounds of Crocus sativus by GC‐MS and HPLC/UV‐ESI‐MS and Evaluation of Their Antioxidant, Antimicrobial, Anticoagulant, and Antidiabetic Properties,”. Pharmaceuticals, 2023. [PubMed]
- Flavonoid Composition and Antibacterial Properties of Crocus sativus L. Petal Extracts,”. Molecules, 2023
- Drying Kinetics of Saffron Floral Bio‐Residues,”. Italian Journal of Food Science, 2016
- Effect of Drying Processes on Stability of Anthocyanin Extracts From Saffron Petal,”. Evolving Trends in Engineering and Technology, 2014
- Stigmas and Petals of Crocus sativus L. (Taliouine, Morocco): Comparative Evaluation of Their Phenolic Compounds, Antioxidant, and Antibacterial Activities,”. Scientific World Journal, 2024
- Hot Air and Freeze‐Drying of High‐Value Foods: A Review,”. Journal of Food Engineering, 2001
- Phytochemical Analysis and Antioxidant Activity of Different Tissue Types of Crocus sativus and Oxidative Stress Alleviating Potential of Saffron Extract in Plants, Bacteria, and Yeast,”. South African Journal of Botany, 2015
- Predictive Multi Experiment Approach for the Determination of Conjugated Phenolic Compounds in Vegetal Matrices by Means of LC‐MS/MS,”. Molecules, 2022. [PubMed]
- Identification of the Flavonoid Fraction in Saffron Spice by LC/DAD/MS/MS: Comparative Study of Samples From Different Geographical Origins,”. Food Chemistry, 2007
- Phenolic Glucosides and a Gamma‐Lactone Glucoside From the Sprouts of Crocus sativus ,”. Planta Medica, 1999. [PubMed]
- 6‐Hydroxyflavones and Other Flavonoids of Crocus ,”. Zeitschrift für Naturforschung C, 1984
- Phytochemical Differentiation of Saffron (Crocus sativus L.) by High Resolution Mass Spectrometry Metabolomic Studies,”. Molecules, 2021. [PubMed]
- 33 L. Mendes Da Silva and F. J. Da Silva , “Bioassay With Artemia salina L.: A Gateway to Understanding the Toxicity of Medicinal Plant Extracts,” in Botânica, Ecologia e Sustentabilidade: Uma Perspectiva Multidisciplinar, ed. L. Mendes da Silva (Editora Científica Digital, 2023), 50–69.
- Toxicоlogical Evaluation of the Plant Products Using Brine Shrimp (Artemia salina L.) Model,”. Macedonian Pharmaceutical Bulletin, 2014
- Oxidative Stress: The Role of Antioxidant Phytochemicals in the Prevention and Treatment of Diseases,”. International Journal of Molecular Sciences, 2024. [PubMed]
- Analytical Methods Used in Determining Antioxidant Activity: A Review,”. International Journal of Molecular Sciences, 2021. [PubMed]
- Antioxidant Activity/Capacity Measurement. 1. Classification, Physicochemical Principles, Mechanisms, and Electron Transfer (ET)‐Based Assays,”. Journal of Agricultural and Food Chemistry, 2016. [PubMed]
- The Versatility of Antioxidant Assays in Food Science and Safety—Chemistry, Applications, Strengths, and Limitations,”. Antioxidants, 2020. [PubMed]
- Dietary Polyphenols and Their Role in Oxidative Stress‐Induced Human Diseases: Insights Into Protective Effects, Antioxidant Potentials and Mechanism of Action,”. Frontiers in Pharmacology, 2022
- Plant Phenolics: Extraction, Analysis and Their Antioxidant and Anticancer Properties,”. Molecules, 2010. [PubMed]
- Antioxidant Activities of Polyphenols From Sage (Salvia officinalis),”. Food Chemistry, 2001
- Antioxidant, Hypoglycemic and Molecular Docking Studies of Methanolic Extract, Fractions and Isolated Compounds From Aerial Parts of Cymbopogon citratus (DC.) Stapf,”. Molecules, 2022. [PubMed]
- Antioxidant Acetophenone Glycosides From the Roots of Euphorbia ebracteolata Hayata,”. Natural Product Research, 2017. [PubMed]
- Evaluation of Reducing Ability and Antioxidant Activity of Fruit Pomace Extracts by Spectrophotometric and Electrochemical Methods,”. Journal of Analytical Methods in Chemistry, 2020. [PubMed]
- Plant In Vitro Culture for the Production of Antioxidants—A Review,”. Biotechnology Advances, 2008. [PubMed]
- Phenolic Compounds and Antioxidant Activity From Saffron (Crocus sativus L.) Petal,”. Journal of Agricultural Science, 2012
- Flavonoids and Their Properties to Form Chelate Complexes,”. Biotechnology and Food Science, 2012
- A Novel HPLC‐Assisted Method for Investigation of the Fe2+‐Chelating Activity of Flavonoids and Plant Extracts,”. Molecules, 2014. [PubMed]
- Lutein and β‐Carotene Characterization in Free and Nanodispersion Forms in Terms of Antioxidant Activity and Cytotoxicity,”. Journal of Pharmaceutical Innovation, 2023
- Novel Insight Into Utilization of Flavonoid Glycosides and Biological Properties of Saffron (Crocus sativus L.) Flower Byproducts,”. Journal of Agricultural and Food Chemistry, 2020. [PubMed]
- Proteins, Polysaccharides and Their Derivatives as Macromolecular Antioxidant Supplements: A Review of In Vitro Screening Methods and Strategies,”. International Journal of Biological Macromolecules, 2023. [PubMed]
- Changes in Antioxidant Effects and Their Relationship to Phytonutrients in Fruits of Sea Buckthorn (Hippophae rhamnoides L.) During Maturation,”. Journal of Agricultural and Food Chemistry, 2000. [PubMed]
- Brine Shrimp: A Convenient General Bioassay for Active Plant Constituents,”. Planta Medica, 1982
- Inhibitory Effects of Chlorogenic Acids on Linoleic Acid Peroxidation and Haemolysis,”. Phytochemistry, 1994
- Studies on Products of Browning Reaction. Antioxidative Activities of Products of Browning Reaction Prepared From Glucosamine,”. Japanese Journal of Nutrition and Dietetics, 1986
- Evaluation of Antioxidant Properties of Elaeocarpus ganitrus Roxb. Leaves,”. Iranian Journal of Pharmaceutical Sciences, 2008
