Phenolic Profile and Biological Evaluation of Salvia ceratophylloides Ard.: A Novel Source of Rosmarinic Acid
Abstract
Salvia ceratophylloides Ard. (Sc), a rare endemic species from Southern Italy, has been chemically unexplored. We report the first analysis of its polyphenolic profile, antioxidant potential, and preliminary toxicity, compared with the well‐studied Salvia officinalis L. (So). Plants were cultivated under identical Mediterranean conditions to minimize variability. Leaf hydroalcoholic extracts were examined for polyphenols (high‐performance liquid chromatography with photodiode array detection and electrospray ionization mass spectrometry [HPLC‐PDA/ESI‐MS]), antioxidant activity (2,2‐diphenyl‐1‐picrylhydrazyl [DPPH], reducing power, Fe2+ chelation), and toxicity (Artemia salina lethality bioassay). Although differing qualitatively, both species showed nearly identical total phenolic content (So 274.59 mg/g; Sc 274.27 mg/g). Notably, Sc was rich in rosmarinic acid and consistently exhibited superior antioxidant activity. It was also nontoxic against A. salina, in contrast to So (LC50 = 79.27 ± 11.62 µg/mL). These findings highlight Sc as a promising source of bioactive compounds and warrant further pharmacological and conservation studies.
Article type: Research Article
Keywords: antioxidant activity, chemical diversity, high‐performance liquid chromatography with photodiode array detection and electrospray ionization mass spectrometry [HPLC‐PDA/ESI‐MS], phenolic compounds
Affiliations: Department of Chemical, Biological, Pharmaceutical and Environmental Sciences University of Messina Messina Italy; Messina Institute of Technology c/o Department of Chemical, Biological, Pharmaceutical and Environmental Sciences University of Messina Messina Italy; Chromaleont s.r.l., c/o Messina Institute of Technology c/o Department of Chemical, Biological, Pharmaceutical and Environmental Sciences University of Messina Messina Italy
License: © 2026 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202503276 | PubMed: 41662584 | PMC: PMC12885525
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.0 MB)
Introduction
The excessive accumulation of free radicals in the human body is a key factor in the development of degenerative conditions such as chronic inflammation and neurodegenerative disorders [ref. 1, ref. 2]. Oxidative stress, which arises from an imbalance between reactive oxygen species (ROS) and the body’s antioxidant defense, compromises cellular integrity and normal physiological functions. In this context, plant‐derived polyphenols have attracted considerable attention due to their ability to counteract oxidative damage and to modulate biological processes, including inflammation, cell signaling, and apoptosis [ref. 3, ref. 4]. Due to their chemical diversity and multifunctional effects, polyphenols represent an important focus in natural product research. Consequently, the identification of polyphenol‐rich plant species and the evaluation of their chemical composition and biological activity remain central goals in pharmacognosy and phytochemistry.
The genus Salvia L. (Lamiaceae), comprising approximately 1000 species distributed across tropical and temperate regions, is among the most diverse genera within the family. In Italy, 20 species contribute significantly to national plant biodiversity. Many Salvia species are valued for their culinary, medicinal, and ornamental uses. Among these, Salvia officinalis L. (common sage, So) is the most extensively studied, with numerous studies reporting antioxidant, anti‐inflammatory, antibacterial, and anticancer properties [ref. 5, ref. 6]. Although early investigations primarily focused on its essential oils, increasing attention has been directed toward polar constituents, particularly phenolic acids and flavonoids, as key contributors to its biological activities [ref. 7, ref. 8]. In contrast, Salvia ceratophylloides Ard. (Sc) is a rare and endangered species, endemic to a restricted hilly area near Reggio Calabria in Southern Italy. Despite its ecological significance and distinctive morphological traits, this species has received little attention from a phytochemical and pharmacological perspective. To date, research has mainly addressed its ecophysiology [ref. 9, ref. 10] and volatile organic compound profile [ref. 11], leaving its phenolic composition and biological potential unexplored. The present study aims to fill this gap by providing the first detailed characterization of the polyphenolic profile, antioxidant activity, and preliminary safety evaluation of Sc. A comparative analysis with So, used as a reference species, was conducted under controlled cultivation conditions to minimize environmental variability. Beyond its potential biological relevance, Sc represents an important component of Southern Italy’s endemic flora. Therefore, the characterization of its bioactive compounds contributes not only to natural product research but also to the valorization and conservation of this underexplored species.
Results and Discussion
Morpho‐Physiological Measurements
According to literature data [ref. 9, ref. 10, ref. 11], Sc and So differed significantly in gas exchange and, consequently, in plant biomass (Table 1).
TABLE 1: Mean values ± standard deviation (SD) of total plant biomass (dry weight [DW] plant) recorded at the end of the experimental period (n = 5) and of stomatal conductance to water vapor (g L), transpiration rate (E L), and photosynthetic rate (An), recorded weekly from February to April in Salvia ceratophylloides (Sc) and Salvia officinalis (So).
| Sc | So | p value | |
|---|---|---|---|
| DW plant (g) | 3.62 ± 0.95 | 5.91 ± 0.73 | 0.002 |
| gL (mmol/m2/s) | 314.3 ± 57.2 | 393.9 ± 65.2 | 0.006 |
| EL (mmol/m2/s) | 2.44 ± 0.47 | 2.24 ± 0.53 | 0.373 |
| An (mmol/m2/s) | 4.21 ± 1.12 | 6.71 ± 1.88 | 0.001 |
Note: p values as resulted by a Student’s t‐test are reported.
In detail, despite being cultivated under identical optimal conditions, So showed higher stomatal conductance and photosynthetic rate than Sc, whereas both species showed comparable transpiration rates. Consequently, So accumulated significantly greater biomass than Sc by the end of the experimental period.
Determination of Polyphenolic Compounds by High‐Performance Liquid Chromatography With Photodiode Array Detection and Electrospray Ionization Mass Spectrometry [HPLC‐PDA/ESI‐MS]
The two Salvia species under investigation exhibited distinct qualitative and quantitative phenolic profiles. In total, 14 phenolic compounds were tentatively identified across both extracts: 13 in So and 5 in Sc. These included seven phenolic acids and seven flavonoids, as illustrated in Figure 1 and detailed in Table 2. Interestingly, despite significant differences in the extraction yields of Sc and So, approximately double in the former (42%) compared to the latter (23%), as well as in the composition and number of compounds, the polyphenolic amount was remarkably similar between the two species: 274.59 mg/g in So and 274.27 mg/g in Sc. In So extract, rosmarinic acid was the most abundant compound (142.00 ± 3.75 mg/g), followed by luteolin‐7‐O‐glucuronide (53.40 ± 3.67 mg/g). In contrast, Sc was characterized by higher level of rosmarinic acid (183.18 ± 7.54 mg/g), accompanied by caffeic acid derivative (58.99 ± 1.06 mg/g) as the second most abundant phenolic. This substantial presence of rosmarinic acid in Sc suggests that this rare, endemic species may serve as a valuable and novel natural source of this compound, with potential nutraceutical or pharmaceutical applications. To the best of our knowledge, this is the first report on the phenolic composition of the aerial parts of Sc. The identification of rosmarinic acid as its dominant phenolic compound is particularly noteworthy, especially considering that So, a widely studied member of the Lamiaceae family, is commonly recognized as one of the richest natural sources of rosmarinic acid. When compared with previously published data on other Lamiaceae species, the levels of rosmarinic acid found in Sc are especially promising. For instance, So has been reported to contain between 12.2 and 296 mg/kg of rosmarinic acid in infusion preparations [ref. 12]. In a separate study, Sharma et al. [ref. 13] reported concentrations ranging from 67.41 ± 2.03 to 18 821.33 ± 150.20 mg/kg in different extracts (Soxhlet methanol extract, and ultrasound water and methanol extracts) obtained from the leaves of cultivated So. These reports, considered within their respective analytical frameworks, indicate that Salvia species exhibit a marked capacity for rosmarinic acid accumulation. In this context, the high accumulation observed in Sc supports its classification as a particularly rich natural source of this bioactive compound. Phenolic compounds in both species were identified using HPLC‐PDA at 330 nm, based on the comparison of retention times, UV absorption spectra, and mass spectra, with those of available standards or literature data. The predominance of phenolic acids and flavonoids observed here is consistent with chromatographic investigations indicating that these compound classes constitute the principal bioactive fraction in Salvia spp. [ref. 14]. As expected, our analysis confirmed the presence of rosmarinic acid as the principal phenolic acid in So, consistent with numerous previous studies [ref. 1, ref. 12, ref. 15, ref. 16].
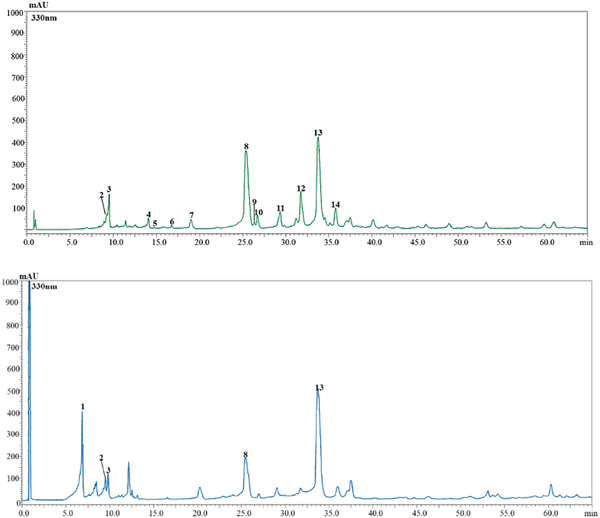
TABLE 2: Characterization of polyphenol compounds in Salvia officinalis (So) and Salvia ceratophylloides (Sc) leaf extracts through high‐performance liquid chromatography with photodiode array detection and electrospray ionization mass spectrometry (HPLC‐PDA/ESI‐MS) analysis.
| Peak no. | Name | tR (min) | UVmax | [M − H]− | Somg/g dry extract ± SD | Scmg/g dry extract ± SD | Standard employed for quantification | Refs. |
|---|---|---|---|---|---|---|---|---|
| 1 | Caffeic acid derivative | 6.8 | 326 | 415, 191 | — | 58.99 ± 1.06 | Caffeic acid | — |
| 2 | 1‐O‐Caffeolyl glucose | 9.3 | 298sh, 326 | 341 | 8.49 ± 0.47 | 4.29 ± 0.99 | Caffeic acid | [ref. 18] |
| 3 | Caffeic acid | 9.4 | 322 | 179 | 19.08 ± 0.87 | 11.02 ± 0.89 | Caffeic acid | [ref. 18] |
| 4 | Luteolin rutinoside | 14.1 | 270,335 | 593, 285 | 1.42 ± 0.08 | — | Luteolin‐7‐O‐glucoside | [ref. 17] |
| 5 | Lithospermic acid A | 14.8 | 327 | 537 | 0.19 ± 0.02 | — | Caffeic acid | [ref. 19] |
| 6 | Quercetin 3‐O‐glucuronide | 16.8 | 280sh,343 | 477, 301 | 0.75 ± 0.02 | — | Quercitrin | [ref. 19] |
| 7 | Apigenin 7‐O‐Allosyl‐(1‐>2)‐glucoside | 19.2 | 340 | 593, 269 | 1.80 ± 0.06 | — | Apigenin | [ref. 19] |
| 8 | Luteolin‐7‐O‐glucuronide | 25.6 | 253, 347 | 461,285 | 53.40 ± 3.67 | 16.79 ± 2.02 | Luteolin‐7‐O‐glucoside | [ref. 17] |
| 9 | Quercetin 3‐O‐glucuronide isomer | 26.5 | 343 | 477, 301 | 0.63 ± 0.02 | — | Quercitrin | [ref. 19] |
| 10 | Salvianolic acid C | 26.9 | 344 | 491 | 12.46 ± 0.26 | — | Salvianolic acid B | [ref. 19] |
| 11 | Chrysoeriol‐7‐O‐rutinoside | 29.3 | 276, 334 | 607, 299 | 5.99 ± 0.06 | — | Luteolin‐7‐O‐glucoside | [ref. 19] |
| 12 | Apigenin‐O‐pentoside | 32.0 | 335 | 445, 269 | 7.22 ± 0.32 | — | Apigenin | [ref. 19] |
| 13 | Rosmarinic acid | 34.1 | 328 | 359, 179 | 142.00 ± 3.75 | 183.18 ± 7.54 | Caffeic acid | [ref. 18] |
| 14 | Salvianolic acid k | 36.1 | 287, 323 | 555 | 21.16 ± 0.23 | — | Salvianolic acid B | [ref. 19] |
| Total | 274.59 | 274.27 | ||||||
Note: Values are expressed as the mean of mg/g of dried extract ± SD, sh: wavelength shoulder.
Qualitatively, our findings are largely consistent with those reported by Martins et al. [ref. 17], with a few exceptions. Compounds such as sagecoumarin and sagerinic acid, previously detected in So, were not found in our samples.
Notably, significant quantitative differences were also observed, which may be attributed to variations in extraction protocols, environmental growing conditions, or plant phenological stages at the time of sampling. These differences underscore the importance of standardized methods when comparing phytochemical profiles across studies.
Given the comparable phenolic content evidenced for Sc and So, the much higher amount in the extraction yield of Sc appears to be due to other polar compounds belonging to different chemical classes contained in the leaves of Sc, extracted with 70% ethanol, which paves the way for further studies aimed at obtaining a thorough characterization of the phytochemistry of this unexplored species.
Antioxidant Activity
Single‐electron transfer (SET) and hydrogen‐atom transfer (HAT) represent two major categories encompassing the primary chemical mechanisms underlying antioxidant activity. The SET mechanism involves the transfer of a single electron to reduce oxidant species. Conversely, the HAT mechanism consists of hydrogen atom donation to neutralize free radicals. Although many antioxidant reactions are characterized as following either SET or HAT pathways, these mechanisms can operate simultaneously. Consequently, the application of multiple complementary assays is recommended to obtain a comprehensive evaluation of the antioxidant properties of plant extracts [ref. 20]. In this study, the 2,2‐diphenyl‐1‐picrylhydrazyl (DPPH) test, which involves both HAT and SET mechanisms, and the reducing power assay, based on the SET mechanism, were used to evaluate the primary antioxidant activity of Sc and So extracts. In addition, ferrous ion chelating activity was used to evaluate the secondary antioxidant capacity. The results of the DPPH test are shown in Figure 2. Sc extract exhibited notable radical scavenging activity compared with the standard antioxidant butylated hydroxytoluene (BHT) and higher activity than So extract at the lower tested concentrations (0.0625–0.25 mg/mL). Both extracts displayed a similar concentration‐dependent trend, reaching maximum inhibition values of approximately 87% at 0.5 mg/mL. The strong ability of Sc to neutralize the stable free radical DPPH demonstrates its potential to mitigate oxidative stress effectively. The IC50 value confirmed the higher scavenging activity of the Sc extract compared with So, although both were less active than BHT (Table 3).
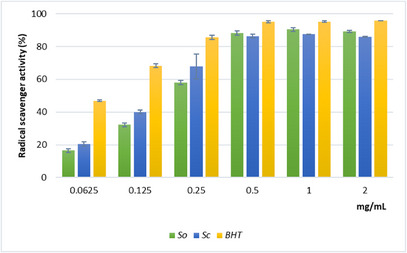
TABLE 3: Mean 50% inhibitory concentration (IC50) determined by 2,2‐diphenyl‐1‐picrylhydrazyl (DPPH) free radical scavenging activity and by Fe2+‐chelating activity assays, and ascorbic acid equivalents (ASE/mL) determined by the reducing power assay of leaf extracts from Salvia ceratophylloides (Sc) and Salvia officinalis (So).
| IC50, DPPH (mg/mL) | Reducing power (ASE/mL) | IC50, Fe2+ chelating activity (mg/mL) | |
|---|---|---|---|
| Sc | 0.174 ± 0.007a | 10.169 ± 0.508 | 0.471 ± 0.011a |
| So | 0.225 ± 0.007b | 9.174 ± 0.186 | >2 mg/mLb |
| Standard | BHT | BHT | EDTA |
| 0.07 ± 0.001 | 1.443 ± 0.021 | 0.0067 ± 0.0003 | |
| p value | <0.001 | 0.06 | <0.0001 |
Note: Data are expressed as the mean ± SD (n = 3). Standards: BHT for DPPH and reducing power; EDTA for Fe2+ chelation. Differences between Sc and So were evaluated using Student’s t‐test. Different letters within the same column indicate statistically significant differences (p < 0.05). The corresponding p values are also reported.
Abbreviations: BHT, butylated hydroxytoluene; EDTA, ethylenediaminetetraacetic acid disodium salt dihydrate.
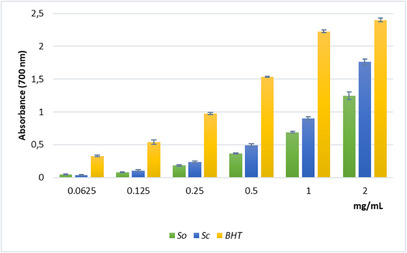
The reducing power assay (Figure 3), which evaluates the ability to interrupt radical chain reaction through Fe3+‐Fe2 + reduction, revealed a moderate but concentration‐dependent activity for the Sc extract, reaching an absorbance value of 1.8 at the highest concentration assayed. Nonetheless, the ascorbic acid equivalents per milliliter (ASE/mL) values calculated for the extracts were quite similar (Table 3).
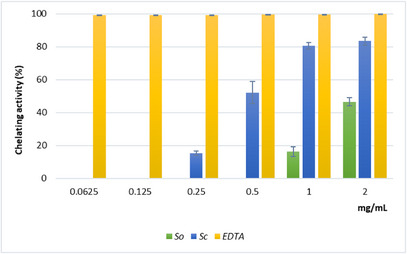
The results of the ferrous ion (Fe2+) chelating activity (Figure 4) further indicated a stronger secondary antioxidant capacity for the Sc extract compared with So. Chelating activity increased with concentration, reaching about 80% at 1 and 2 mg/mL for Sc. The IC50 value for Sc was much lower than that for So, although both extracts showed weaker chelating ability than ethylenediaminetetraacetic acid disodium salt dihydrate (EDTA) (Table 3).
Overall, these findings indicated that the Sc leaf extract possesses appreciable primary and secondary antioxidant activities under the experimental conditions adopted. These effects may be linked with its higher content of rosmarinic acid, together with caffeic acid and its derivatives, compounds whose antioxidant properties are well documented. Consistent with this interpretation, studies on other medicinal plants have shown that variation in antioxidant performance is largely attributable to differences in the abundance of highly active phenolic acids, particularly rosmarinic acid and structurally related derivatives [ref. 21]. Previous investigations have shown the effectiveness of rosmarinic acid in DPPH scavenging, reducing power, and chelation assays [ref. 22, ref. 23]. In any case, the possible additive or synergistic effects of the different flavonoid derivatives and phenolic acids contained in the phytocomplexes that may contribute to the observed antioxidant activities should also be taken into account. For example, moderate synergism has previously been demonstrated for a combination of rosmarinic acid and luteolin‐7‐O‐glucuronide in the DPPH assay [ref. 24]. However, the biological relevance of these antioxidant properties remains preliminary and requires further validation in cellular and in vivo models.
Artemia salina Lethality Bioassay
The toxicity of Sc and So extracts was preliminarily evaluated using the Artemia salina lethality bioassay, a widely adopted screening method for assessing the toxicity of plant extracts. This assay offers several advantages, including simplicity, low cost, reproducibility, and ease of implementation, making it suitable for early stage safety evaluation [ref. 25]. The results of the assay are shown in Figure 5. According to Clarkson’s toxicity criterion, the Sc extract was classified as non‐toxic to brine shrimp larvae, as the LC50 value was found to be over 1000 µg/mL after 24 h of exposure. In contrast, the So extract exhibited marked toxicity, with an LC50 value lower than 100 µg/mL, equal to 79.27 ± 11.62 µg/mL. No mortality was observed in the control groups.
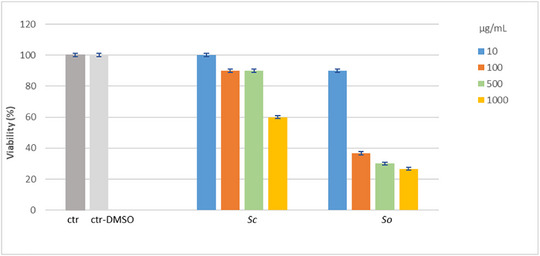
The marked toxicity highlighted for So is an unexpected result, because some literature data report the safety of So preparations. The lack of acute toxicity in rats was shown for an 80% ethanol extract obtained from leaves and flowers of So by maceration [ref. 26]. Another recent study demonstrated the absence of acute toxicity in mice for a 70% ethanol leaf extract obtained from leaves of So by 24‐h maceration [ref. 27]. Our disagreeing result could be explained by the selected extraction procedure. Ultrasound‐assisted extraction is an alternative and more efficient extraction method compared to conventional maceration, which can ensure improved recovery of bioactive components. It can be hypothesized that the use of this technique, in combination with a moderate temperature and using 70% ethanol as solvent, led to the extraction of phytoconstituents in high concentration, which may also include toxic compounds, such as the terpenes thujone and camphor [ref. 28]. Interestingly, the Sc extract obtained with the same procedure was found to be nontoxic; this finding would seem to indicate a lower content of toxic constituents in the leaves of Sc. The different toxicity of Sc and So extracts could be clarified by further phytochemical investigations.
Conclusion
This study provides the first phytochemical characterization and preliminary biological evaluation of the rare endemic Sc. Despite its restricted geographical distribution, this species was found to accumulate high levels of rosmarinic acid and to exhibit notable antioxidant capacity in a set of in vitro assays, together with an absence of toxicity in preliminary A. salina bioassay. In comparison, So displayed lower antioxidant performance and higher toxicity under the same experimental conditions.
Taken together, these findings suggest that Sc is a promising source of bioactive phenolic compounds. However, the biological effects reported here should be regarded as preliminary, as they are based exclusively on in vitro antioxidant assays and a simple toxicity screening model. Further investigations involving cellular systems and in vivo studies are necessary to substantiate its potential relevance for nutraceutical or pharmaceutical applications. Additionally, the results highlight the importance of conserving this underexplored endemic species as part of Italy’s phytochemical biodiversity [ref. 29].
Future research could also investigate the use of Sc extracts as natural antioxidant additives in biodegradable or other eco‐friendly packaging materials. The phenolic compounds may enhance the functional properties of such films, although this remains a preliminary hypothesis requiring experimental validation (e.g., [ref. 30, ref. 31]).
Experimental Section
Plant Material and Growth Conditions
Experiments were performed on 5‐month‐old plants of Salvia ceratophylloides Ard. and Salvia officinalis L. Seeds of both species, kindly provided by the Botanical Garden of the University of Messina, were soaked in distilled water for 24 h and sown in greenhouse trays in October 2023. One month after germination, individual seedlings were transplanted into 3.5 L plastic pots filled with a nutrient‐rich potting substrate formulated for vegetable cultivation (Comodo, Tercomposti S.P.A., Italy).
Plants were grown in a greenhouse at the Department of Chemical, Biological, Pharmaceutical and Environmental Sciences (ChiBioFarAm), University of Messina. The greenhouse, oriented northwest, received natural daylight supplemented with LED lamps (Led Spectrum Grow 6, I‐GROW s.r.l., Italy), providing a 16 h light/8 h dark photoperiod and a photosynthetic photon flux density of 450 µmol/m2/s. Temperature was maintained between 24°C during the day and 18°C at night.
In February 2024, plants were transferred to an open terrace at the ChiBioFarAm Department, where they were exposed to natural environmental conditions and regularly irrigated to maintain field capacity. Leaf sampling for chemical analyses was conducted in April 2024.
Morpho‐Physiological Measurements
To monitor plant physiological status throughout the experiment, stomatal conductance to water vapor (g L), transpiration rate (E L), and net photosynthetic rate (An) were measured weekly from February to April. Measurements were performed on at least five fully expanded leaves per species using a portable photosynthesis system (LCi Analyzer, ADC Bioscientific Ltd., Herts, UK).
At the end of the experimental period, plant biomass was assessed. Roots (carefully rinsed to remove soil), stems, leaves, and flowers were oven‐dried at 80°C for 3 days to determine their dry weight (DW).
Extraction Procedure
For the preparation of the extracts, the leaves of Sc and So were lyophilized, finely ground, and subjected to extraction using 70% ethanol (1:10 w/v). A preliminary maceration at 25°C was performed for 1 h, followed by ultrasonic‐assisted extraction at 50°C for 15 min, repeated twice.
The combined extracts were filtered and concentrated to dryness using a rotary evaporator. Extraction yields were calculated on the basis of 100 g of lyophilized plant material and amounted to 42% for Sc and 23% for So. The yield obtained for So agrees with that previously found by Glisic et al. [ref. 32] in the 70% ethanol leaf extract of So (1:12 w/v) obtained using ultrasound‐assisted extraction for 45 min at 40°C, resulting in approximately 20%.
Determination of Polyphenolic Compounds by HPLC‐PDA/ESI‐MS
The qualitative and quantitative characterization of the polyphenolic components of Sc and So extracts was performed using HPLC‐PDA/ESI‐MS.
The dried extracts were redissolved in 70% ethanol and filtered through a 0.45 µm Acrodisc nylon membrane filter (Merck Life Science, Merck KGaA, Darmstadt, Germany) before analysis. LC–MS grade solvents, including water, acetonitrile, and formic acid, along with analytical standards (caffeic acid, apigenin, salvianolic acid B, luteolin‐7‐O‐glucoside, and quercitrin), were also purchased from Merck Life Science, Merck KGaA, Darmstadt, Germany.
Chromatographic analysis was carried out using a Shimadzu HPLC system (Kyoto, Japan) equipped with a CBM‐20A controller, two LC‐30AD dual‐plunger parallel‐flow pumps, a DGU20A3R degasser, a CTO‐20AC column oven, a SIL‐30AC autosampler, an SPD‐M30A photodiode array (PDA) detector, and an LCMS‐2020 mass spectrometer. The system employed an ESI source operating in negative ionization mode. Separations were performed on an Ascentis Express C18 column (150 × 2.1 mm; 2.7 µm) obtained from Merck Life Science, Merck KGaA, Darmstadt, Germany [ref. 33].
The mobile phase consisted of water (solvent A) and acetonitrile (solvent B), both acidified with 0.1% formic acid (v/v), under the following gradient elution conditions: 0 min: 0% B—10 min: 10% B—20 min: 11% B—30 min: 15% B—50 min: 18% B—65 min: 23% B.
The flow rate was set at 0.5 mL/min, and the injection volume was 2 µL. PDA detection was applied over the 190–450 nm range and monitored at 330 nm (sampling frequency: 40 Hz, time constant: 0.050 s).
Mass spectrometry (MS) conditions were as follows: scan range: m/z 100–1200; scan speed: 7500 amu/s; event time: 0.3 s; nebulizing gas (N2) flow rate: 1.5 L/min; drying gas (N2) flow rate: 15 L/min; interface temperature: 350°C; heat block temperature: 300°C; desolvation line temperature: 300°C; desolvation line voltage: 1 V; interface voltage: −4.5 kV.
Quantification of the polyphenolic content in the extracts was based on calibration curves constructed for five standards: caffeic acid, apigenin, salvianolic acid B, luteolin‐7‐O‐glucoside, and quercitrin (Table 4). Data acquisition and processing were performed using Shimadzu LabSolution software version 5.97.
TABLE 4: Regression equations, correlation coefficients (R 2), LoD and LoQ, repeatability, and accuracy values for each reference material employed.
| Compounds name | Calibration curve | R2 | LoD | LoQ | Repeatability (%)100 mg/L | Accuracy (%)100 mg/L |
|---|---|---|---|---|---|---|
| Caffeic acid | y = 4068.7x + 19686 | 0.9996 | 0.053 | 0.161 | 0.25 | 100.30 |
| Apigenin | y = 22357x ‐ 1527.1 | 0.9999 | 0.037 | 0.111 | 0.26 | 100.50 |
| Salvianolic acid B | y = 3670.4x + 1415.2 | 0.9999 | 0.145 | 0.439 | 0.14 | 100.82 |
| Luteolin‐7‐O‐glucoside | y = 12881x + 54935 | 0.9995 | 0.048 | 0.016 | 0.31 | 99.58 |
| Quercitrin | y = 8054x + 27465 | 0.9997 | 0.034 | 0.103 | 0.12 | 99.85 |
Antioxidant Activity
To comprehensively assess the antioxidant capacity of the extracts, three in vitro assays were performed: (i) DPPH radical scavenging assay, (ii) reducing power assay, and (iii) ferrous ion chelating activity assay.
DPPH Radical Scavenging Assay
The DPPH test was carried out to estimate the free radical scavenging activity of Sc and So hydroalcoholic extracts, using the method reported by Ohnishi et al. [ref. 34]. The extracts were tested at concentrations ranging from 0.0625 to 2 mg/mL. For each sample, 0.5 mL of the extract solution was mixed with 3 mL of freshly prepared 0.1 mM DPPH methanolic solution. The mixture was incubated in the dark at room temperature for 20 min, after which the absorbance was recorded at 517 nm using a UV‐1601 spectrophotometer (Shimadzu). BHT was used as the reference standard. Results were expressed as the mean radical scavenging activity (%) ± standard deviation (SD) and the mean 50% inhibitory concentration (IC50) ± SD, derived from three independent experiments.
Reducing Power Assay
The reducing power of Sc and So extracts was determined using the Fe3+–Fe2+ transformation method, as described by Oyaizu et al. [ref. 35]. Extract concentrations ranged from 0.0625 to 2 mg/mL, with BHT and ascorbic acid serving as reference standards. To perform the assay, a reaction mixture was prepared by combining 2.5 mL of 0.2 M phosphate buffer (pH 6.6), 2.5 mL of 1% potassium ferricyanide, and 1 mL of the extract solution. The mixture was incubated at 50°C for 20 min and then rapidly cooled. Subsequently, 2.5 mL of 10% trichloroacetic acid was added, and the mixture was centrifuged at 3000 rpm for 10 min at 4°C. The supernatant (2.5 mL) was combined with 2.5 mL of distilled water and 0.5 mL of 0.1% ferric chloride. The reaction mixture was incubated in the dark at room temperature for 10 min, and the absorbance was measured at 700 nm. Results are presented as mean absorbance values ± SD and ASE/mL ± SD, based on three independent experiments.
Ferrous Ion (Fe2+) Chelating Activity Assay
The Fe2+ chelating activities of Sc and So extracts were determined spectrophotometrically by measuring the formation of the Fe2+‐ferrozine complex, following the protocol described by Kumar et al. [ref. 36]. Extract concentrations ranged from 0.0625 to 2 mg/mL, with EDTA as the reference standard. In this assay, 0.05 mL of 2 mM ferrous chloride solution was mixed with 1 mL of the extract solution and 0.5 mL of methanol. Then, 0.2 mL of 5 mM ferrozine solution was added, and the mixture was shaken vigorously before being incubated in the dark at room temperature for 10 min. The absorbance was recorded at 562 nm using a spectrophotometer. Results are reported as the mean inhibition of the ferrozine‐Fe2+ complex formation (%) ± SD and IC50 ± SD, based on the average of three independent experiments.
Artemia salina Leach (Brine Shrimp) Lethality Bioassay
The potential toxicity of Sc and So extracts was determined using the brine shrimp lethality bioassay [ref. 37]. Brine shrimp eggs were incubated under a 60 W lamp that provided direct light and warmth (24–26°C) in artificial seawater (33 g sea salt/L deionized water). To achieve final concentrations in the range of 10–1000 µg/mL, 10 brine shrimp larvae were incubated for 24 h at 24–26°C in artificial seawater mixed with varying volumes of the extracts dissolved in DMSO. Control groups included artificial seawater alone and artificial seawater added with 2% DMSO, corresponding to the highest solvent concentration used in the assay. After counting the number of surviving nauplii, GraphPad Prism 10 was used to calculate the median lethal concentration (LC50) values. The test was performed in triplicate. The extracts’ level of toxicity was evaluated using Clarkson’s toxicity criterion, which states that an extract is nontoxic if its LC50 is greater than 1000 µg/mL [ref. 38].
Statistical Analysis
Statistical analyses were performed using SigmaStat 12.0 (SPSS Inc., Chicago, IL, USA). Differences between species in g L, E L, An (n = 40), DW (five plants per species), and antioxidant assay results (three independent extracts per species, n = 3) were assessed using Student’s t‐test. Data are expressed as mean ± SD, and differences were considered statistically significant at p < 0.05.
Author Contributions
Douaa Bekkai: conceptualization, investigation, data curation, writing – original draft. Natalizia Miceli: investigation, data curation, writing – original draft. Maria Fernanda Taviano: investigation. Carmelo Coppolino: investigation. Francesco Cacciola: conceptualization, supervision. Luigi Mondello: conceptualization. Patrizia Trifilò: conceptualization, supervision.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Characterization and Quantification of Phenolic Compounds and Antioxidant Properties of Salvia Species Growing in Different Habitats,”. Industrial Crops and Products, 2013
- A Review of the Antioxidant Potential of Medicinal Plant Species,”. Food and Bioproducts Processing, 2011
- Oxidative Stress and Antioxidant Potential of One Hundred Medicinal Plants,”. Current Topics in Medicinal Chemistry, 2017. [PubMed]
- Polyphenols and Their Benefits: A Review,”. International Journal of Food Properties, 2017
- Bioactive Profile of Various Salvia officinalis L. Preparations,”. Plants, 2019. [PubMed]
- Effect of Salvia officinalis on Diabetic Patients,”. Journal of Renal Injury Prevention, 2013. [PubMed]
- Pharmacological Properties of Salvia officinalis and Its Components,”. Journal of Traditional and Complementary Medicine, 2017. [PubMed]
- Sage (Salvia officinalis L.): A Botanical Marvel With Versatile Pharmacological Properties and Sustainable Applications in Functional Foods,”. South African Journal of Botany, 2024
- Too Dry to Survive: Leaf Hydraulic Failure in Two Salvia Species Can be Predicted on the Basis of Water Content,”. Plant Physiology and Biochemistry, 2021. [PubMed]
- When Water Availability Is Low, Two Mediterranean Salvia Species Rely on Root Hydraulics,”. Plants, 2021. [PubMed]
- The Assessment and the Within‐Plant Variation of the Morpho‐Physiological Traits and VOCs Profile in Endemic and Rare Salvia ceratophylloides Ard. (Lamiaceae),”. Plants, 2021. [PubMed]
- Rapid UHPLC Determination of Polyphenols in Aqueous Infusions of Salvia officinalis L. (Sage Tea),”. Journal of Chromatography B, 2011
- Identification and Characterization of Polyphenols and Volatile Terpenoid Compounds in Different Extracts of Garden Sage (Salvia officinalis L.),”. Pharmacognosy Research, 2020
- Bioactive Chemical Composition and Pharmacological Insights Into Salvia Species,”. Frontiers in Molecular Biosciences, 2025. [PubMed]
- Variations in Essential Oil, Phenolic Compounds, and Antioxidant Activity of Tunisian Cultivated Salvia officinalis L,”. Journal of Agricultural and Food Chemistry, 2009. [PubMed]
- Comparison of Conventional and Ultrasound‐Assisted Extraction Techniques on Mass Fraction of Phenolic Compounds From Sage (Salvia officinalis L.),”. Chemical and Biochemical Engineering Quarterly, 2015
- Evaluation of Bioactive Properties and Phenolic Compounds in Different Extracts Prepared From Salvia officinalis L,”. Food Chemistry, 2015. [PubMed]
- Qualitative Analysis and Simultaneous Quantification of Phenolic Compounds in the Aerial Parts of Salvia miltiorrhiza by HPLC‐DAD and ESI/MS n,”. Phytochemical Analysis, 2011. [PubMed]
- Salvia officinalis and Lippia triphylla: Chemical Characterization and Evaluation of Antidepressant‐Like Activity,”. Journal of Pharmaceutical and Biomedical Analysis, 2021. [PubMed]
- Antioxidant Capacity Determination in Plants and Plant‐Derived Products: A Review,”. Oxidative Medicine and Cellular Longevity, 2016. [PubMed]
- Unlocking the Potential of Rosmarinic Acid: A Review on Extraction, Isolation, Quantification, Pharmacokinetics and Pharmacology,”. Phytomedicine Plus, 2025
- Antioxidant Activity of Rosmarinic Acid Extracted and Purified From Mentha piperita ,”. Archives of Razi Institute, 2021. [PubMed]
- The Role of Rosmarinic Acid in Cancer Prevention and Therapy: Mechanisms of Antioxidant and Anticancer Activity,”. Antioxidants, 2024. [PubMed]
- Antimicrobial and Antioxidant Properties of Four Lycopus Taxa and an Interaction Study of Their Major Compounds,”. Molecules (Basel, Switzerland), 2020. [PubMed]
- Artemia Species: An Important Tool to Screen General Toxicity Samples,”. Current Pharmaceutical Design, 2020. [PubMed]
- Phytochemical Screening, Acute Toxicity and Antidiabetic Activity of Ethanolic Extract of Salvia officinalis L. in Wistar Rat,”. Agriculturae Conspectus Scientificus, 2023
- Hydroethanolic Extract of Salvia officinalis L. Leaves Improves Memory and Alleviates Neuroinflammation in ICR Mice,”. Scientific World Journal, 2025. [PubMed]
- Determination of the Biologically Active Flavour Substances Thujone and Camphor in Foods and Medicines Containing Sage (Salvia officinalis L.),”. Chemistry Central Journal, 2011. [PubMed]
- Genetic Structure of Populations of Salvia ceratophylloides Endemic to Southern Calabria (Southern Italy),”. Heliyon, 2024. [PubMed]
- Polysaccharide‐Based Edible and Biodegradable Films From Eruca vesicaria Mucilage: Structural Characterization, Biocompatibility, and Environmental Impact,”. Food Measure, 2025
- Preparation and Properties of Novel Mucilage Composite Films Reinforced With Polydimethylsiloxane,”. Macromolecular Materials and Engineering, 2024
- The Combined Extraction of Sage (Salvia officinalis L.): Ultrasound Followed by Supercritical CO2 Extraction,”. Ultrasonics Sonochemistry, 2011. [PubMed]
- Differential Cadmium Responses in Two Salvia Species: Implications for Tolerance and Ecotoxicity,”. Plants, 2026
- Inhibitory Effects of Chlorogenic Acids on Linoleic Acid Peroxidation and Haemolysis,”. Phytochemistry, 1994
- Studies on Products of Browning Reaction. Antioxidative Activities of Products of Browning Reaction Prepared From Glucosamine,”. Japanese Journal of Nutrition and Dietetics, 1986
- Improved Survival in Multiple Myeloma and the Impact of Novel Therapies,”. Blood, 2008. [PubMed]
- Brine Shrimp: A Convenient General Bioassay for Active Plant Constituents,”. Planta Medica, 1982
- Toxicological Evaluation of the Plant Products Using Brine Shrimp (Artemia salina L.) Model,”. Macedonian Pharmaceutical Bulletin, 2014
