Analysis of Chemical Composition, Antioxidant Capacity, Acute Toxicity, and Antinociceptive Properties of Aqueous Extract of Origanum Majorana L.
Abstract
Pharmacological evaluation showed that AEOM significantly reduced pain in an animal model, suggesting potential analgesic properties. Acute toxicity studies indicated no adverse effects on kidney and liver function or blood parameters at doses up to 800 mg/kg. The analgesic effect is likely mediated by flavonoids in the extract, which may inhibit pain pathways. These findings suggest that O. majorana has promising therapeutic applications, particularly as a natural analgesic agent.
Article type: Research Article
Keywords: Phytochemical composition, Antioxidant, Toxicity, Antinociceptive
Affiliations: Laboratory of biochemistry Faculty of medicine and pharmacy University Cadi Ayyad Marrakech Morocco; Laboratory of Pharmacology Neurobiology and Behavior Faculty of Sciences Semlalia University Cadi Ayyad Marrakech Morocco; Laboratory of Physiopathology Genetic Molecular and Biotechnology Faculty of Sciences Aïn Chock Hassan II University Casablanca Morocco; Laboratory of Biological Engineering Faculty of Sciences and Technology Sultan Moulay Slimane University Beni Mellal 23000 Morocco; Department of Chemistry and Technologies of Drug Sapienza University P. le Aldo Moro 500185 Rome Italy
License: © 2024 The Author(s). Chemistry & Biodiversity published by Wiley-VHCA AG CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202401580 | PubMed: 39446705 | PMC: PMC11908776
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (656 KB)
Introduction
In recent decades, herbal remedies have gained global recognition due to their effectiveness, affordability, and limited side effects in the treatment of diseases. Natural products, particularly those derived from plants, play a key role as important sources of raw materials. These resources have led to the discovery of numerous bioactive molecules, offering various benefits in the fields of nutrition, cosmetics, and pharmaceuticals.ref. cbdv202401580-bib-0001
Morocco, known for its rich biodiversity, boasts an impressive variety of plant species. Among the 4,200 species and subspecies of vascular plants in the country, 800 are endemic.ref. cbdv202401580-bib-0002 This diversity underscores the value of Moroccan flora, which has long been exploited for medicinal purposes. As a result, research into medicinal plants has intensified, exploring their potential as sources of alternative remedies.ref. cbdv202401580-bib-0003 Among these, Origanum majorana L., known as sweet marjoram, is a medicinal plant of the Lamiaceae family, widely used in traditional medicine for its various therapeutic properties.[ref. cbdv202401580-bib-0004, ref. cbdv202401580-bib-0005] This plant, primarily distributed in Mediterranean regions, is rich in phytochemicals such as thymol, carvacrol, and various flavonoids, which explain its numerous biological properties.[ref. cbdv202401580-bib-0006, ref. cbdv202401580-bib-0007, ref. cbdv202401580-bib-0008, ref. cbdv202401580-bib-0009]
Origanum majorana, commonly used in traditional medicine, has therapeutic applications depending on the plant parts used. In Moroccan traditional medicine, the leaves are primarily utilized for their anti‐cooling, antipyretic, and antihypertensive properties, as well as for treating allergies, fever, flu, and respiratory infections[ref. cbdv202401580-bib-0010, ref. cbdv202401580-bib-0011, ref. cbdv202401580-bib-0016] Additionally, the leaves and stems are effective against rheumatism,[ref. cbdv202401580-bib-0011, ref. cbdv202401580-bib-0013] stomach pain, headache, cough, and insomnia.ref. cbdv202401580-bib-0014 The plant is also used in infusion form for calming, antispasmodic effects, and treating colds, fever, and headaches[ref. cbdv202401580-bib-0015, ref. cbdv202401580-bib-0016]
The extracts of O. majorana exhibit notable biological activities, including antibacterial properties against various pathogenic bacteria and antifungal effects against pathogenic fungi.ref. cbdv202401580-bib-0017 Furthermore, O. majorana possesses antiparasitic, antidiabetic, anti‐inflammatory, analgesic, antipyretic, hepatoprotective, antimutagenic, and gastrointestinal properties.[ref. cbdv202401580-bib-0018, ref. cbdv202401580-bib-0019, ref. cbdv202401580-bib-0020, ref. cbdv202401580-bib-0021, ref. cbdv202401580-bib-0022] In this context, our study aimed to experimentally evaluate the traditional uses of the aqueous extract of O. majorana on animal models. We have examined the chemical composition of this extract, its antioxidant properties, acute toxicity, and antinociceptive effects.
Results and Discussion
HPLC Ms/Ms Analysis
Chemical analysis of the aqueous extract of O. majorana, conducted via LC–MS/MS, has enabled the identification of a variety of phenolic compounds, as detailed in Table 1. Among them, gallic acid, caffeic acid, and chlorogenic acid stand out for their pronounced bioactive properties. Additionally, other minor compounds have been identified in this variety originating from the Azilal region.
Table 1: Annotated compounds from O. majorana aqueous extract using LC–MS/MS.
| PeakNo. | Proposed compound | Rt(min) | [M−H]–(m/z) | FragmentsMS/MS(m/z) |
|---|---|---|---|---|
| 1 | Gallic acid | 5.2 | 169.014 | 125, 79 |
| 2 | Caffeic acid | 8.5 | 179.034 | 135, 89 |
| 3 | Dihydroxy phenolic acid | 12.3 | 153.018 | 109, 81 |
| 4 | Chlorogenic acid | 15.7 | 353.087 | 191, 135 |
| 5 | Syringic acid | 18.6 | 197.045 | 153, 109 |
| 6 | Vanillic acid | 20.4 | 167.034 | 123, 95 |
| 7 | p‐Coumaric acid | 22.8 | 163.039 | 119, 93 |
| 8 | Ferulic acid | 25.3 | 193.050 | 149, 107 |
| 9 | Rosmarinic acid | 28.1 | 359.076 | 197, 161 |
| 10 | Trans‐2 Dihydroxycinnamic acid | 30.5 | 181.050 | 137, 91 |
| 11 | Cinnamic acid | 32.2 | 147.044 | 103, 79 |
Phytochemical Study
Our results showed significant and remarkable levels of total polyphenols (186.06±0.1 mg GAE/g), flavonoids (72.3±0.9 mg QE/g), and condensed tannins (4.49±0.08 mg CE/g) in AEOM leaves. (Table 2).
Table 2: Mean values of total polyphenol, flavonoids, and tannin contents in the extract.
| Total Polyphenol Contentmg GAE/g DM | Flavonoidsmg QE/g DM | Taninsmg CE/g DM | |
|---|---|---|---|
| Crude extracts (mg/g) | 186.06±0.1 | 72.3±0.9 | 4.49±0.08 |
GAE: gallic acid equivalent, QE: Qercetin equivalent, CE: Catechin equivalent, DM: dry matter.
Antioxidant Activity
AEOM exhibited antioxidant activity similar to that of a standard antioxidant, as evaluated through both the DPPH scavenging assay and the reducing power assay. The calculated IC50 value was 2.23±0.03 mg/mL in the DPPH assay, indicating its capability to convert the stable purple‐colored DPPH radical into the yellow‐colored DPPH‐H form (Table 3). Additionally, AEOM demonstrated reducing power, with an IC50 value of 1.9±0.01 mg/mL
Table 3: The antioxidant activity of the AEOM by DPPH and FRAP.
| Antioxidant Assay | Plant Extract(IC50=mg/mL) | Standard Antioxidant(IC50=mg/mL) | |
|---|---|---|---|
| Quercetin | BHT | ||
| DPPH | 2.23±0.1 | 0.1±0.0 | 0.2±0.0 |
| FRAP | 1.9±0.0 | 0.1±0.0 | 0.1±0.0 |
Data are mean±SEM.
Estimation of LD50
The oral administration of single doses (1000, 2000, and 5000 mg/kg body weight) of AEOM did not lead to any mortality among the treated animals during the fourteen‐day observation period. No signs of toxicity were observed, and no significant alterations in body weight or relative organ weight were found among the treated animals. These results indicated that the oral lethal dose 50 (LD50) of AEOM exceeded 5000 mg/kg body weight in mice.
Biochemical and Histological Analyses
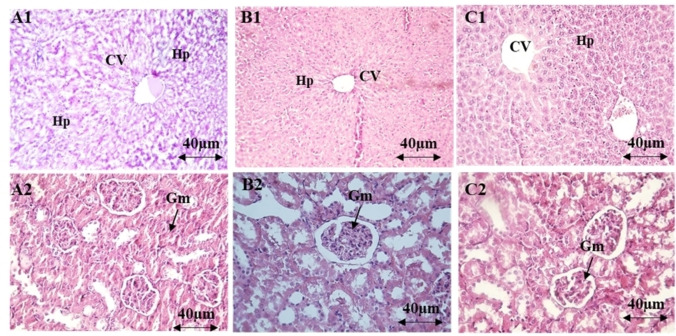
The administration of AEOM did not affect the levels of biochemical parameters (urea, creatinine, ASAT, ALAT) at any dose (Table 4). Microscopic examination of the kidney and liver tissues showed a normal appearance similar to that of the control group, indicating that no damaging changes or morphological disturbances were caused by the oral administration of AEOM (Figure 1).
Table 4: Biochemical parameters in the serum of mice treated with AEOM.
| Groups | Alanine transaminase[ALT (U/L)] | Aspartate aminotransferase[AST (U/L)] | Creatinine(mmol/L) | Urea(mmol/L) |
|---|---|---|---|---|
| Control | 54.22±1.61 | 134.22±1.70 | 30.23±1.14 | 9.32±1.01 |
| AEOM 1 g/kg | 50.61±1.52 | 138.24±4.83 | 32.61±2.31 | 12.91±1.54 |
| AEOM 2 g/kg | 52.74±2.30 | 136.21±1.92 | 31.10±2.81 | 12.90±1.60 |
| AEOM 5 g/kg | 51.42±2.10 | 138.30±4.20[a] | 36.50±2.80 | 13.81±1.50 |
Data are expressed as mean±SEM, [a] indicate p<0.01 compared with saline‐treated controls.
Antinociceptive Activity
Hot Plate Test
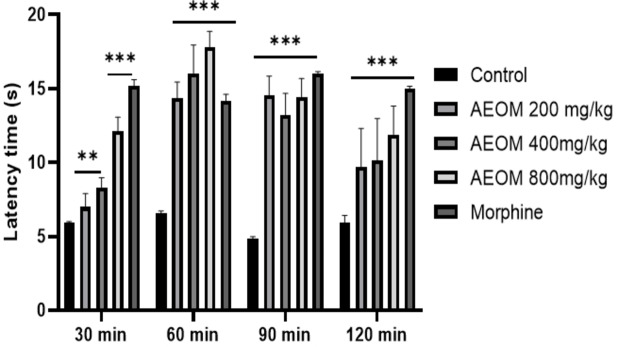
The results of the analgesic effects of the plant extract and the standard drug are illustrated in Figure 2. The antinociceptive activity results reveal that AEOM significantly increased (p<0.001) time latency at 30, 60, and 90 minutes compared to the control group. Similarly, the standard drug (morphine 10 mg/kg) significantly prolonged the time intervals in all cases compared to the control group.
Abdominal Writhing
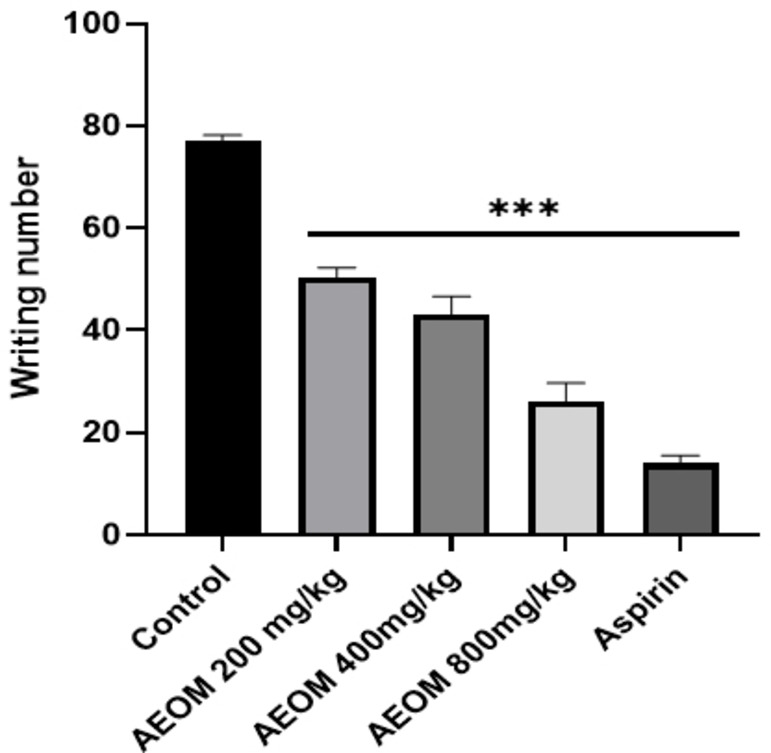
The application of AEOM at all dosage levels resulted in a significant reduction in the number of abdominal constrictions compared to the control group (Figure 3). This effect was more pronounced at higher doses (p<0.01) and was comparable to the effects observed in the morphine and aspirin groups, which served as positive controls.
Formalin Test
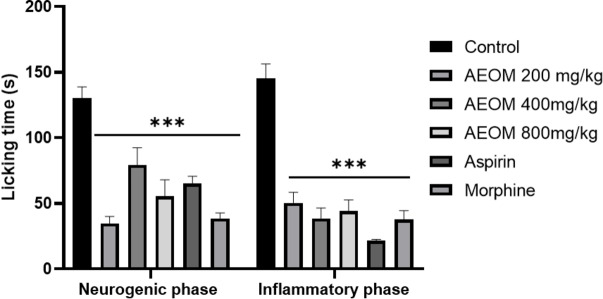
The formalin test results are shown in Figure 4. The extract exhibited notable analgesic effects by decreasing paw licking time during both the neurogenic (Phase 1) and inflammatory (Phase 2) phases, with the reduction being particularly evident in this last phase. Administration of AEOM at doses of 200, 400, and 800 mg/kg resulted in reduced paw licking time during both phases of the formalin trial. In comparison, the reference drug, aspirin, demonstrated greater efficacy in the second phase, while morphine, the standard drug, reduced licking time in both Phase 1 and Phase 2.
Phytotherapy, the medicinal use of plant‐derived compounds, has attracted substantial attention for its ability to alleviate a wide range of ailments affecting a significant portion of the global population.ref. cbdv202401580-bib-0023 Notably, this therapeutic approach targets critical symptoms such as pain and oxidative stress, which are commonly associated with numerous chronic and acute conditions.ref. cbdv202401580-bib-0024 Our study on O. majorana L. underlines the relevance of phytotherapy in this context. By examining the phytochemical composition and biological activities of O. majorana, we aim to elucidate its potential in mitigating these physiological challenges through its natural bioactive compounds.
The objective of this study was to evaluate the phytochemical composition, acute toxicity, antioxidant activity, and pharmacological effects of the aqueous extract of O. majorana L. (AEOM). The phytochemical analysis revealed significant amounts of total polyphenols, flavonoids, and condensed tannins, all known for their potential health benefits. AEOM exhibited notable antioxidant effects in both DPPH and reducing power tests. Additionally, the acute toxicity study revealed the safety of the extract up to a dose of 5000 mg/kg. HPLC analysis identified 11 secondary metabolites, with chlorogenic acid as the major component. The results of the pain tests demonstrated significant positive responses, thereby highlighting the effectiveness and promising potential of AEOM in modulating this complex physiological process.
Polyphenols are essential in determining how natural chemicals behave biologically. Phytochemical screening in this investigation indicated a significant amount of flavonoids, polyphenols and modest level of condensed tannins. We found numerous parallels and some discrepancies that may be attributed to both internal and extrinsic factors when we compared our results with those of other studies previously obtained for the same collected species but in various parts of the world.[ref. cbdv202401580-bib-0025, ref. cbdv202401580-bib-0026, ref. cbdv202401580-bib-0027] Studies carried out in Morocco have shown that there are small differences in the amounts of condensed tannins, flavonoids, and polyphenols. Variations may be attributed to variations in the plant organ, habitat, genotype, and kind of solvent employed in the extraction procedure.ref. cbdv202401580-bib-0028 Specifically, the effects of edaphic, climatic, and genetic factors.[ref. cbdv202401580-bib-0029, ref. cbdv202401580-bib-0030]
The investigation employed various methods to evaluate the antioxidant properties of the aqueous extract. Several established assays, including the DPPH radical scavenging assay and the FRAP assay, were used to assess the antioxidant capacity of the extract. The findings indicated that AEOM exhibited significant antioxidant activity by efficiently scavenging free radicals and reducing oxidative damage, as evidenced by its effectiveness in neutralizing the DPPH radicals and enhancing the FRAP values. The results of the antioxidant activity of AEOM showed strong anti‐DPPH activity, with an IC50 of 0.3 mg/mL, as well as significant reducing and iron‐chelating activities.ref. cbdv202401580-bib-0031 Similarly, the ethanolic extracts showed positive results in terms of antioxidant activity with IC50 values of 11.5 mg/mL for anti‐DPPH and 67.2 mg. Additionally, other studies, such as those conducted by Roby et al. (2013)ref. cbdv202401580-bib-0001 and Dhull et al. (2016ref. cbdv202401580-bib-0032 have also validated the antioxidant activity of O. majorana leaf extracts, demonstrating good activity in various testing techniques, such as the reduction of DPPH and ABTS free radicalsref. cbdv202401580-bib-0033 These results highlight the strong antioxidant profile of oregano compounds and their medical importance.ref. cbdv202401580-bib-0034
Additionally, the extract demonstrated a high total phenolic content, suggesting the presence of potent antioxidants in the plant material. As demonstrated by Nagendrappa,ref. cbdv202401580-bib-0035 phenolic compounds may directly contribute to the antioxidative effects. Flavonoids, known for their robust antioxidant activities in laboratory settings, have the ability to scavenge various reactive oxygen species (ROS) such as superoxide and nitric oxide radicals.ref. cbdv202401580-bib-0036 This investigation into the antioxidant potential of AEOM has uncovered compelling results, emphasizing its noteworthy antioxidant activity. The research aimed to explore the potential benefits of this extract in addressing oxidative stress, a factor known to contribute to various diseases and aging processes.ref. cbdv202401580-bib-0037
The acute toxicity study showed that oral administration of AEOM at single doses (1000, 2000, and 5000 mg/kg body weight) did not result in mortality in mice; the animals survived until the end of the study. Similarly, the growth and relative organ weight of the AEOM‐treated animals remained unchanged throughout the experimental period. Biochemical analysis revealed no significant changes in renal and liver function parameters, while hematological analysis showed no effects on on the levels of white blood cell, red blood cell, and platelets. Histopathological examination revealed no morphological abnormalities in kidney, liver, and spleen tissues. Therefore, the LD50 of AEOM was higher than 5000 mg/kg. According to Kennedy Jr. et al.,ref. cbdv202401580-bib-0038 substances with LD50 values greater than 5.0 g/kg when orally administered are generally considered practically non‐toxic. These findings are in line with previous studies demonstrating the safety of the aqueous extract of O. majorana aerial parts.ref. cbdv202401580-bib-0039
The hotplate test is among the most common tests to evaluate the activity of opioid compounds, which are centrally acting analgesics in several animal species. This test is notable for its tendency to respond to painful stimuli that pass through neural pathways. It represents a supra‐spinal organized pain response.[ref. cbdv202401580-bib-0040, ref. cbdv202401580-bib-0041] Based on our results, the aqueous extract of O. majorana showed anti‐nociceptive activity. These findings are in agreement with results of Fachini‐Queiroz et al.ref. cbdv202401580-bib-0042 that demonstrated that luteolin, thymol, carvacrol, and ursolic acid in the O. majorana extract would be responsible for the anti‐nociceptive activity of this plant.ref. cbdv202401580-bib-0043
On the other hand, the writhing test is applied to show peripheral anti‐nociceptive activity in mice. It is appropriate for differentiating between central and peripheral nociception.ref. cbdv202401580-bib-0044 The injection of acetic acid produces peritoneal inflammation, which triggers a response characterized by cramps.ref. cbdv202401580-bib-0045 Previous studies have shown that acetic acid indirectly induces the release of endogenous pain mediators (such as prostaglandins, kinins, histamine, etc.) that stimulate nociceptive neurons, which are sensitive to nonsteroidal anti‐inflammatory drugs and opioids.ref. cbdv202401580-bib-0046 Thus, the aqueous extract of O. majorana showed a significant decrease in the number of writhes compared to the control group and the ASA‐treated group. O. majorana, commonly known as marjoram, exhibits significant analgesic effects, primarily attributed to its aqueous extract, whose major compounds are gallic acid, caffeic acid, and chlorogenic acid, according to several studies. The analgesic properties of O. majorana are linked to multiple mechanisms, including the modulation of cholinergic, opioid, and dopaminergic systems, as well as the inhibition of N‐Methyl‐D‐aspartate (NMDA) receptors and the reduction of nitric oxide (NO) production.ref. cbdv202401580-bib-0047 These pathways contribute to the antinociceptive effects of these compounds, which correspond to the action of traditional non‐steroidal anti‐inflammatory drugs (NSAIDs) that inhibit the prostaglandin metabolic pathway, as noted by Okazaki et al.ref. cbdv202401580-bib-0048 who reported the antiplatelet activity of the methanol extract of O. majorana. Furthermore, the ability of O. majorana to modulate inflammatory responses by inhibiting pro‐inflammatory cytokines, such as IL‐1β, IL‐6, interferon (IFN)‐α, and TNF‐α, which are key players in the pathways of fever and pain, highlights its potential therapeutic applications in pain and inflammation management. The richness in polyphenols and flavonoids in the aqueous extract of O. majorana not only enhances its analgesic effects but also underscores the complex interaction of the plant with the mechanisms of the central nervous system, offering a promising natural alternative for pain relief.
Conclusions
Our investigation into the therapeutic potential of the Origanum majorana L. extract underscores the promising role of herbal medicine in addressing diverse health challenges, particularly those related to pain management and oxidative stress. Through rigorous analyses including phytochemical composition, acute toxicity evaluation, antioxidant activity, and pharmacological effects, we have revealed compelling insights. The abundant presence of polyphenols, flavonoids, and condensed tannins in the extract, as well as its robust antioxidant effects, indicate its potential in combating oxidative damage. Safety assessments up to a dose of 5000 mg/kg in acute toxicity studies align with previous safety profiles of similar extracts, affirming its suitability for therapeutic use. HPLC analysis identified key secondary metabolites like chlorogenic acid contributing to its bioactivity. Notably, our pain tests demonstrated significant anti‐nociceptive and peripheral analgesic activities, suggesting a promising avenue for pain management. These findings not only contribute to the growing body of evidence supporting the therapeutic efficacy of O. majorana L. extract but also highlight its potential applications in holistic healthcare approaches, paving the way for further research and clinical exploration in phytotherapy.
Experimental Section
Plant Samples
The leaves of O. majorana were collected in 2022 from Azilal, a region in the central Atlas Mountains of Morocco (31°57′41′′ N, 6°34′15′′ W). The plant’s authenticity was initially confirmed by botanist Professor A. Ouhammou and pharmacologist Professor A. Chait from the Faculty of Sciences at Semlalia, Cadi Ayyad University. The plants were then carefully preserved and recorded under voucher specimen MARK‐14381 in the herbarium of the Department of Biology, Faculty of Sciences Semlalia, Cadi Ayyad University, Marrakech, Morocco.
Extract Preparation
The process began with macerating the leaves of O. majorana in water, with continuous agitation for 12 hours. Afterward, the resulting liquid underwent centrifugation at 1200 rpm, followed by filtration and lyophilization using a Christ instrument. The resulting dry powder was then aseptically stored in amber bottles at 4 °C until needed for further use.
Phytochemical Study
Total Phenolic Content (TPC)
The total phenolic content (TPC) of the extract was measured using a modified Folin‐Ciocalteu method based on Singleton et al..ref. cbdv202401580-bib-0049 To begin the assessment, 0.4 mL of the diluted extract was mixed with 1.5 mL of Folin‐Ciocalteu reagent. Afterward, 1.6 mL of a 7.5 % sodium carbonate solution was added. The mixture was then kept in the dark at room temperature for 2 hours, and the absorbance was read at 765 nm. Gallic acid was used as the standard reference, and the results were expressed in milligrams of gallic acid equivalents per gram of dry weight (mg GAE/g DW).
Total Flavonoid Content (TFC)
The total flavonoid content (TFC) of the extract was determined following the method outlined by Zhishen et al..ref. cbdv202401580-bib-0050 Initially, 200 μL of the extract was mixed with 1 mL of distilled water. Subsequently, 60 μL of 5 % NaNO2 and 60 μL of 10 % AlCl3 were introduced into the solution. After a five‐minute incubation period, 400 μL of 1 M NaOH was added, and the absorbance was measured at 510 nm. The TFC was then expressed as milligrams of catechin equivalents per gram of dry matter (mg CE/g DM).
Total Condensed Tannins
To assess the condensed tannin content, the method outlined by Aitbaba et al.ref. cbdv202401580-bib-0051 was followed. In summary, 400 μL of the diluted samples were combined with 3 mL of a 4 % methanol vanillin solution and 1.5 mL of concentrated HCl. Following a 15 minute incubation period, the absorbance was measured at 500 nm. The condensed tannin content was then determined and expressed as milligrams of catechin equivalents per gram of dry matter (mg CE/g DM).
Antioxidant Activity
Radical Scavenging Activity (DPPH)
The antioxidant potential of the extract was assessed using the 2,2‐diphenyl‐1‐picrylhydrazyl (DPPH) assay, following the method described by Mansouri et al.ref. cbdv202401580-bib-0052 with modifications from Baslam et al..ref. cbdv202401580-bib-0053 In brief, 1.5 mL of a methanolic DPPH solution (6×10−5 M) was mixed with 60 μL of the extract at various concentrations (1, 2, 4, 6, and 8 mg/mL) of AEOM. The mixture was protected from light and left at room temperature for 30 minutes. After the incubation period, the absorbance was measured at 515 nm. A negative control was prepared by mixing 1.5 mL of the DPPH solution with 60 μL of methanol. Positive controls consisted of butylated hydroxytoluene (BHT) and quercetin. The percentage of inhibition was determined using the following formula:
Where A control represents the absorbance of the control, and A sample denotes the absorbance of the test compound.
The concentration of the sample that causes 50 % inhibition (IC50) was ascertained from a graph illustrating the percentages of inhibition plotted against the sample concentrations.
Ferric Reducing Ability Power (FRAP)
The FRAP test, as outlined by Oyaizu,ref. cbdv202401580-bib-0054 evaluates the inhibition of Fe (II)‐Ferrazine complex formation during sample incubation with ferrous iron. In detail, a mixture of 1 mL distilled water, 2.5 mL phosphate buffer (0.2 M, pH 6.6), and 2.5 mL potassium ferricyanide (K3[Fe(CN)6], 1 %) was combined with 0.5 mL of extract solutions at varying concentrations. Following a 30 minute incubation, 2.5 mL distilled water, 2.5 mL 10 %trichloroacetic acid, and 0.5 mL FeCl3 were introduced into the mixture. Absorbance was then measured at 700 nm. Quercetin and butylated hydroxytoluene (BHT) served as positive controls.
Animal Study
Animals
Adult male Swiss mice (25–35 g) were obtained from the animal care unit of the Faculty of Science Semlalia, Cadi Ayyad University, Marrakech, Morocco. These animals were housed under controlled conditions at a constant room temperature (22±2 °C) with a 12 hour light/12 hour dark cycle and had unrestricted access to food and water. All animal procedures strictly complied with the guidelines outlined in the European Council Directive (EU2010/63). The study was conducted ethically and received approval from the Council Committee of the Research Laboratory, Faculty of Science, Cadi Ayyad University of Marrakech. Every effort was made to minimize potential animal suffering.
Acute Toxicity
The limit test dose for the acute toxicity study (5000 mg/kg) was conducted in accordance with the guidelines of the Organization for Economic Cooperation and Development (OECD) (guideline n. 423). Four groups of mice were used, each comprising six animals. Three groups received oral administration of AEOM at doses of 1000, 2000, and 5000 mg/kg, respectively, while the remaining group (negative control) received distilled water. Administration was performed at a rate of 10 mL/kg. The mice were closely monitored for signs of toxicity and mortality during the first two hours following the administration of the extract.
Biochemical and Histological Analyses
On day 14, the animals underwent euthanasia via cervical dislocation to obtain blood samples for the assessment of potential changes in hematological and biochemical parameters. Organs, including the liver, spleen, and kidneys, were preserved in buffered formalin (10 %) and subsequently embedded in paraffin. Following this, 5 μm sections were obtained using a microtome and subjected to hematoxylin and eosin staining for microscopic examination.
Analgesic Tests
Hot Plate Test
The methodology previously employed by Okolo et al.ref. cbdv202401580-bib-0055 was implemented in this study. The animals were subjected to a test environment comprising a glass cylinder placed on a heated metal plate maintained at a temperature of 55±1 °C. The reaction time was gauged by measuring the latency to unpleasant responses such as licking, shaking one paw, and jumping. The treated mice received three oral doses of AEOM (200, 400, and 800 mg/kg), while the control group was orally administered 10 mL/kg of water. An intraperitoneal injection of 10 mg/kg of morphine was also administered. After treatment, latencies to nociceptive responses were measured at intervals of 30, 60, 90, and 120 minutes.
Abdominal Writhing Induced by Acetic Acid
The procedure outlined by Koster et al., 1959ref. cbdv202401580-bib-0043 was employed in this study. In summary, intra‐peritoneal injections of 0.6 % acetic acid (0.1 mL/10 g) were administered to mice. Thirty minutes before the injections, all experimental groups (5 mice per group) were treated with either the vehicle, aspirin (200 mg/kg), or the tree doses of the AEOM. Subsequently, individual cages were designated for each group to facilitate counting of abdominal spasms over a 30 minute period.
Paw Licking Induced by Formalin
The experiment adhered to the methodology described by Hunskaar & Hole,.ref. cbdv202401580-bib-0056 Mouse groups (n=5) were orally administered various doses of AEOM (250, 500, and 1000 mg/kg) 45 minutes before receiving a 20 μL injection of 2 % formalin (v/v in 0.9 % saline) into the subplantar area of the right hind paw. The control group received a vehicle (10 mL/kg of saline). The reference analgesic medications, morphine (10 mg/kg, i. p.) and aspirin (200 mg/kg, i. p.), were included. Following formalin injection in each mouse, the duration of paw licking was measured during two intervals: 0–5 min (neurogenic phase) and 15–30 min (inflammatory phase).
Statistical Analysis
Data from each measurement were presented as mean values±SEM (standard error of the mean). The analysis and presentation of results were conducted using SigmaPlot 12.0 software, employing one‐way analysis of variance (one‐way ANOVA). In cases where differences were deemed significant (p<0.05), post hoc analysis techniques were applied.
Funding
This study did not receive financial support.
Ethical Issue
Reducing the number of animals and suffering were done according to the recommendations of the European Council Directive (EU2010/63), and authorized by the council committee of the faculty of Sciences, Cadi Ayyad University Marrakech Morocco.
Author Contributions
M.A.El Amiri: Conceptualization, Data curation, Formal analysis, Writing‐original draft preparation, Validation; H. Kabdy: Data curation, Validation, Writing‐original draft preparation; A. Aitbaba: Data curation, Writing‐original draft preparation; J. Laadraoui: Formal analysis, Validation; A. Baslam: Formal analysis, Validation; R. Aboufatima: Conceptualization; L. El Yazouli: Formal analysis, Validation; S. Moubtakir: Formal analysis, Validation S. Garzoli: Writing‐review and editing, Supervision; A. Chait: Conceptualization, Writing‐review and editing, Supervision.
Conflict of Interests
Authors declare no conflict of interest.
References
- 1A. Aitbabaa, H. Kabdy, A. Baslam, A. Azraida, H. Aboufatima, R. El, L. Yazouli, A. Chait, Chemistry & Biodiversity, 2024, e202400228–e202400228.
- 2J. Bellakhdar, 1997, Contribution à l′étude de la pharmacopée traditionnelle au Maroc: la situation actuelle, les produits, les sources du savoir (enquête ethnopharmacologique de terrain réalisée de 1969à 1992) (Doctoral dissertation, Université Paul Verlaine-Metz).
- 3 M. E.-A. Farh , Y.-J. Kim , D.-C. Yang , J Ginseng Res 2018, 42(1), 9–15.29348716
- 4 J. H. Ietswaart , Leiden Botanical Series 1980, 4(1), 1–153.
- 5N. Vasudeva. Origanum majorana L.-Phyto-pharmacological review. 2015, https://nopr.niscpr.res.in/handle/123456789/33655.
- 6R. do S. B. Chaves, R. L. Martins, A. B. L. Rodrigues, E. de M. Rabelo, A. L. F. Farias, C. M. da C. V. Araújo, T. F. Sobral, A. K. R. Galardo, S. S. M. da S. de Almeida, bioRxiv 2019, 595900, DOI: 10.1101/595900.
- 7 L. Guerra-Boone , R. Alvarez-Román , R. Salazar-Aranda , A. Torres-Cirio , V. M. Rivas-Galindo , N. Waksman de Torres , G. González , L. A. Pérez-López , Pakistan journal of pharmaceutical sciences. 2015, 28.
- 8 N. Jelali , W. Dhifi , T. Chahed , B. Marzouk , J. Food Biochem. 2011, 35(5), 1443–1450, 10.1111/j.1745-4514.2010.00465.x.
- 9 K. P. Sefeer , K. Elumalai , Ann. Entomol. 2018, 36(02), 01–05.
- 10 F. Z. Ennacerie , F. R. Filali , E.-D. A. Najia Moukrad , International Journal of Advanced Research. 2017, 5(9), 1238–1247.
- 11 A. Alaoui , S. Laaribya , Nature & Technology. 2017, 9(2),15–24.
- 12 M. Elmastas , L. Ozturk , I. Gokce , R. Erenler , H. Y. Aboul-Enein , Anal. Lett. 2004, 37(9), 1859–1869.
- 13 S. Zougagh , A. Belghiti , T. Rochd , I. Zerdani , J. Mouslim , Nat Prod Bioprospect 2019, 9(1), 35–48. 10.1007/s13659-018-0194-6.30484065
- 14 T. Benali , A. Khabbach , A. Ennabili , K. Hammani , Moroccan Journal of Biology. 2017, 14(14), 1–14.
- 15 O. Benkhnigue , F. B. Akka , S. Salhi , M. Fadli , A. Douira , L. Zidane , J Anim Plant Sci. 2014, 23(1), 3539–3568.
- 16 M. El Hafian , N. Benlandini , H. Elyacoubi , L. Zidane , A. Rochdi , Journal of Applied Biosciences. 2014, 81, 7198–7213.
- 17 M. Ghourri , L. Zidane , A. Douira , International Journal of Biological and Chemical Sciences. 2013, 7(4), 1688–1700.
- 18 I. Demirtas , R. Erenler , M. Elmastas , A. Goktasoglu , Food Chem. 2013, 136(1), 34–40.23017389
- 19 H. A. Khatab , N. S. Elhaddad , British Biotechnology Journal. 2015, 8(1), 1–11.
- 20 G. Amor , L. Caputo , A. La Storia , V. De Feo , G. Mauriello , T. Fechtali , Molecules 2019, 24(22), 4021.31698834
- 21 C. Busatta , R. S. Vidal , A. S. Popiolski , A. J. Mossi , C. Dariva , M. R. A. Rodrigues , F. C. Corazza , M. L. Corazza , J. V. Oliveira , R. L. Cansian , Food Microbiol. 2008, 25(1), 207–211.17993397
- 22 H. Hajlaoui , H. Mighri , M. Aouni , N. Gharsallah , A. Kadri , Microbial pathogenesis. 2016, 95, 86–94.26997648
- 23 N. Chaachouay , L. Zidane , Drugs and Drug Candidates. 2024, 3(1), 184–207.
- 24C. Forni, F. Facchiano, M. Bartoli, S. Pieretti, A. Facchiano, D. D’Arcangelo, S. Norelli, G. Valle, R. Nisini, S. Beninati, BioMed research international, 2019, https://www.hindawi.com/journals/bmri/2019/8748253/abs/.
- 25 A. Benslama , S. Daci , L. Z. Nabti , H. Bendif , A. Harrar , European Journal of Biological Research. 2021, 11(4), 509–518.
- 26 S. B. Dhull , P. Kaur , S. S. Purewal , Resour.-Effic. Technol. 2016, 2(4), 168–174.
- 27 S. Duletić-Laušević , A. A. Aradski , S. Kolarević , B. Vuković-Gačić , M. Oaldje , J. Živković , K. Šavikin , P. D. Marin , J. Appl. Bot. Food Qual. 2018, 91, 126–134.
- 28 R. Erenler , B. Meral , O. Sen , M. Elmastas , A. Aydin , O. Eminagaoglu , G. Topcu , Pharm Biol 2017, 55(1), 1646–1653.28431483
- 29 K. Bouterfas , Z. Mehdadi , M. M. Elaoufi , A. Latreche , W. Benchiha , Ann. Pharm. Fr. 2016, 74(6), 453–462. https://www.sciencedirect.com/science/article/pii/S0003450916300608.27553439
- 30 A. Khadhri , I. Bouali , C. Aouadhi , M. C. Lagel , E. Masson , A. Pizzi , Biomed. Chromatogr. 2019, 33(11), e4665.31339581
- 31 S. B. Dhull , P. Kaur , S. S. Purewal , Resour.-Effic. Technol. 2016, 2(4), 168–174.
- 32 R. Erenler , O. Sen , H. Aksit , I. Demirtas , A. S. Yaglioglu , M. Elmastas , Telci , J. Sci. Food Agric. 2016, 96(3), 822–836.25721137
- 33 R. Kiani , A. Arzani , S. A. M. Mirmohammady Maibody , Front Plant Sci 2021, 12, 646221.33841475
- 34 G. Nagendrappa , Resonance. 2005, 10, 65–74.
- 35 B. Halliwell , Free Radical Biol. Med. 2009, 46(5), 531–542.19111608
- 36 D. Giustarini , I. Dalle-Donne , D. Tsikas , R. Rossi , Critical Reviews in Clinical Laboratory Sciences. 2009, 46(5–6), 241–281. 10.3109/10408360903142326.19958214
- 37 G. L. Kennedy Jr. , R. L. Ferenz , B. A. Burgess , J. Appl. Toxicol. 1986, 6(3), 145–148. 10.1002/jat.2550060302.3722708
- 38 A. Amaghnouje , H. Mechchate , I. Es-Safi , S. S. Boukhira , A. M. Aliqahtani , O. A. Noman , F. Nasr , Molecules 2020, 25(23), 5653.33266220
- 39 D. C. Chapman , J. Phys. Oceanogr. 1985, 15(8), 1060–1075.
- 40 F. J. Morales , S. Jiménez-Pérez , Food Chem. 2001, 72(1), 119–125.
- 41F. C. Fachini-Queiroz, R. Kummer, C. F. Estevao-Silva, M. D. de Barros Carvalho, J. M. Cunha, R. Grespan, C. A. Bersani-Amado, R. K. N. Cuman, Evidence-based complementary and alternative medicine: eCAM, 2012, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3418667/.
- 42 D. Le Bars , M. Gozariu , S. W. Cadden , Pharmacol Rev 2001, 53(4), 597–652.11734620
- 43 R. Koster , M. Anderson , E. De Beer , Federation Proceedings. 1959, 18, 412–417.
- 44 M. R. Sulaiman , Z. A. Zakaria , A. S. Bujarimin , M. N. Somchit , D. A. Israf , S. Moin , Pharm Biol 2008, 46(12), 838–845. 10.1080/13880200802366710.
- 45 D. M. Seoudi , A. M. Medhat , I. H. Hewedi , S. A. Osman , M. K. Mohamed , M. Arbid , J. Radiat. Res. Appl. Sci 2009, 2, 513–534.
- 46 K. Okazaki , S. Nakayama , K. Kawazoe , Y. Takaishi , Phytother. Res. 1998, 12(8), 603.
- 47 B. M. Peskar , M. Trautmann , P. Nowak , B. A. Peskar , Agents Actions 1991, 33, 240.1659153
- 48 E. K. Akkol , F. Göger , M. Koşar , K. H. C. Başer , Food Chem. 2008, 108(3), 942–949.26065756
- 49V. L. Singleton, R. Orthofer, R. M. Lamuela-Raventós, 1999, 299, 152–178.
- 50 J. Zhishen , T. Mengcheng , W. Jianming , Food Chem. 1999, 64(4), 555–559. 10.1016/S0308-8146(98)00102-2.
- 51 A. Aitbaba , Z. Sokar , A. Chait , Bangladesh Journal of Pharmacology. 2023, 18(2).
- 52 A. Mansouri , G. Embarek , E. Kokkalou , P. Kefalas , Food Chem. 2005, 89(3), 411–420. 10.1016/j.foodchem.2004.02.051.
- 53 A. Baslam , A. Aitbaba , R. Aboufatima , F. Agouram , S. Boussaa , A. Chait , M. Baslam , Horticulturae. 2023, 9(11), 10.3390/horticulturae9111196.
- 54 M. Oyaizu , The Japanese journal of nutrition and dietetics. 1986, 44(6), 307–315.
- 55 C. O. Okolo , P. B. Johnson , E. M. Abdurahman , I. Abdu-Aguye , I. M. Hussaini , J. Ethnopharmacol. 1995, 45(2), 125–129. 10.1016/0378-8741(94)01199-A.7776661
- 56 S. Hunskaar , K. Hole , Pain. 1987, 30(1), 103–114. 10.1016/0304-3959(87)900.3614974
