Chemical Characterization and Allelopathic Effects of Ambrosia psilostachya (Asteraceae)
Abstract
Ambrosia spp. have been introduced and distributed across different continents as an invasive alien plant species. The present study aimed to identify the chemical components of essential oil isolated from different parts of Ambrosia psilostachya and evaluate the phytotoxic effect of its aqueous extracts on the growth parameters of Phaseolus vulgaris L. (bean) and Arachis hypogaea L. (peanut) under greenhouse conditions. The chemical components of the essential oil of A. psilostachya were analyzed by gas chromatography‐mass spectrometry. Total 44 compounds, including isoaromadendrene epoxide (17.38 %), citral (15.13 %), caryophyllene (7.23 %), borneol (8.76%), (+)‐2‐bornanone (6.45 %), eucalyptol (4.61 %), and 22 compounds including endo‐borneol (22.76%), (+)‐2‐bornanone (8.08%), caryophyllene oxide (7.77%), citral (14.65%), cis‐verbenol (4.43%), and eucalyptol (4.72%) as the major components were identified and isolated in the shoots and roots, respectively. To evaluate the allelopathic effect of A. psilostachya on bean and peanut seedlings, the aqueous extracts were prepared at four concentrations (25%, 50%, 75%, and 100%). The extracts at the concentrations of 50% or higher had a significant inhibitory effect on yield, dry weight, height, and LAI in both species. The inhibitory activity of A. psilostachya may be attributed to its high content of isoaromadendrene epoxide, citral, caryophyllene, and caryophyllene oxide. This study highlights that the allelopathy effects of A. psilostachya are due to the action of its compounds, which should be analyzed in future studies, and an accurate evaluation of the allelopathy effects of this invasive plant requires comprehensive, long‐term studies in the environment.
Article type: Research Article
Keywords: allelopathy, essential oil, invasive plants, alien species, weeds
Affiliations: Department of Plant Production & Genetics Faculty of Agriculture University of Zanjan Zanjan Iran; Plant Protection Research Department Guilan Agricultural and Natural Resources Research and Education Center, Agricultural Research Education and Extension Organization Rasht Iran; Department of Agronomy and Plant Breeding Faculty of Agricultural Sciences University of Guilan Rasht Iran; Department of Biomedical Surgical and Dental Sciences University of Milan Milan Italy; National Interuniversity Consortium of Materials Science and Technology (INSTM) Firenze Italy
License: © 2025 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202502443 | PubMed: 41237115 | PMC: PMC12761355
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (634 KB)
Introduction
Invasive plants seriously threaten flora by competing or allelopathy into the environment [ref. 1]. Allelopathy is a phenomenon in which living or dead plants produce and release chemical compounds [ref. 2] that can affect the germination or growth of other species can alter their performance, and ultimately plant community structure. Indeed, allelopathy is a mechanism to facilitate the spread of invasive species by releasing allelochemicals such as alkaloids, flavonoids, terpenoids, phenolics, etc. [ref. 3, ref. 4]. Allelochemicals are released into the environment through root exudation, leaching, and decomposition from living or dead plants. The Ambrosia spp. represent one of the most invasive groups of weeds in the world, the invasion of them into field crops has caused concerns in many countries [ref. 5]. The effect of the allelopathic activities of Ambrosia spp. is documented in many literature [ref. 6, ref. 7, ref. 8]. According to [ref. 9] results, all extracts of Ambrosia artemisiifolia significantly reduced the germination of the Zea mays seeds. Similar findings have also been reported by [ref. 10] that A. artemisiifolia inhibits the early growth of tomato and lettuce, as well as the germination of weed species such as large crabgrass (Digitaria sanguinalis). Moreover, allelopathic impacts of Ambrosia spp. on the growth and germination of native species in invaded ranges demonstrate that allelopathy plays a potential role in the success of their invasion [ref. 11, ref. 12].
Ambrosia psilostachya (western ragweed), a member of the Asteraceae, is native to North America and has spread to America, Europe, and Asia [ref. 13]. The occurrence of the A. psilostachya in Iran was first reported in 2017 [ref. 14]. It is a noxious weed spread that can occur by seeds and rhizomes. Allelopathy effects due to the volatile materials of different parts of A. psilostachya have previously been reported by [ref. 15], but their attempts to identify its chemical components were unsuccessful. Although there are reports on identifying compounds from different species of Ambrosia, little knowledge has been reported about the chemical composition of A. psilostachia. A. psilostachya often colonizes coastal areas, dunes, sandy soils, rivers, and ruderal habitats [ref. 16], due to the high spread potential, it is reported in some south strip areas of the Caspian Sea (Guilan), Iran, where it has been considered an invasive weed in urban areas and fields [ref. 14, ref. 17]. Considering the vast invasion of this species in the world, this study aims to: (i) Identification of the chemical compositions of the essential oil extracted from A. psilostachya, and (ii) evaluate the potential allelopathic effects of A. psilostachya on crop plants was conducted. Phaseolus vulgaris L. is one of the most important legumes in the world, playing an important role in human nutrition, and Arachis hypogaea L., an annual legume, is a rich source of multivitamins, thiamine, folic acid, and tocopherols [ref. 18].
Materials and Methods
Collection and Sample Preparation
The study was conducted during 2020‐2021 in the laboratory and greenhouse of the University of Guilan and the University of Zanjan, Iran. Plant samples consisting of mature plants of A. psilostachya, at the vegetative stage, were harvested from infected fields in Guilan Province, Bandar Anzali, Iran, in August 2020 (36.6939°N, 48.4099°E). The plant was identified taxonomically by Dr.Somayeh Tokasi, assistant professor of weed Science (Plant Protection Research Department, Gilan Agricultural and Natural Resources Research and Education Center, Agricultural Research, Education and Extension Organization, AREEO, Rasht, Iran). A specimen was kept at Halophytes and C4 Research Laboratory, School of Biology, University of Tehran, Tehran. Fresh A. psilostachya plants were separated into leaves, shoots, and roots, and were used for essential oil extraction. To prepare the aqueous extract, part of the samples was diced into 1 cm long pieces and dried in the oven at 70°C for 48 h. Extracts were made by placing 100 g of dried material separately into 1000 mL of distilled water and leaving it at room temperature for 72 h.
Identification of Chemical Compounds of A. psilostachya
Essential Oil Isolation
Plant shoots and roots from air‐dried plant material were used separately for extraction. For each sample, the plant material (100 g) was placed in a 2000 mL distillation flask and 1.000 mL of distilled water was added [ref. 19]. The essential oils of each sample were obtained by water distillation using a Clevenger apparatus (Ildam, Ankara, Turkey) according to the European Pharmacopoeia method [ref. 20]. The oils were dried overusing anhydrous sodium sulfate until analysis. The oil yield was calculated according to the dry weight of plant materials and the amount of essential oils obtained.
Gas Chromatography‐Mass Spectrometry Analysis
Chemical analysis of the essential oil of A. psilostachya was obtained with gas chromatography‐mass spectrometry (GC‐MS) and Mass Hunter workstation software. Each sample was subjected to GC‐MS using a flame ionization detector (FID), and the column was a fused silica capillary DB‐5MS (5% phenylmethylpolysyloxane, 30 m × 0.25 mm; film thickness 0.25 µm; J&W Scientific Fisons, Folsom, CA). The temperature program used was as follows: 5 min at 60 then 4°C min−1 up to 220°C, then 11°C min−1 up to 280°C, held for 15 min. Helium was the carrier gas at a flow rate of 1 mL/min. The flow ratio was equal to 1/50. MS in the mass of 70 eV and 30–550 amu, and the ion source’s temperature was set to 230°C. According to the compounds known in the literature, their retention indices are compared by contrasting the same series of n‐alkanes on the HP‐5MS capillary column and stored in the spectrograph database by comparing their mass spectra with NIST05.LIB and NIST05s.LIB, as well as by CO injection of available reference compounds.
Phytotoxic Effect of the Aqueous Extracts of A. psilostachya
Preparation of Aqueous Extracts
The second experiment was to determine the phytotoxic activities of A. psilostachya extract on bean and peanut seedlings. For the preparation of the aqueous extract, the dried plant material was powdered by an electric blender. A stock solution was prepared by adding ground powder into distilled water and shaking for 48 h on a shaker. Finally, from a stock solution, four concentrations were then made.
Greenhouse Experiments
A greenhouse experiment was conducted from May through October 2021. The experiment was conducted in a completely randomized design, consisting of different concentrations of A. psilostachya extracts (0%, C0; 25%, C1; 50%, C2; 75% C3; 100% C4), with four replicates. In this experiment, seeds of bean and peanut were sterilized using Sodium hypochlorite (NaOCl) for 3 min to avoid contamination. Seeds were then washed with distilled water before planting. Experimental pots (18 × 18 × 25 cm) were filled with a sterilized mixture of arable soil. Ten seeds of each plant were sown separately in each pot and thinned after germination. Immediately after seeding the target plants, the pots were watered with different concentrations of eques extracts prepared from A. psilostachya. The pots were watered to maintain field capacity. To determine the growth traits, at final harvesting, the leaf area index (LAI), biomass dry weight, lengths of the shoot, and yield were recorded. Shoot height was measured from the soil level to the upper point of the terminal bud in millimeters using a ruler. After the oven drying of the samples at 60°C, yield and dry weight aboveground biomass were measured with an electronic balance (0.001 g accuracy). The total plant leaf area was measured by a digital planimeter, and LAI was calculated by dividing the total plant leaf area (A: dm2) by its ground area (P: dm2).
Statistical Analysis
Data were subjected to statistical analysis, and means were analyzed using the analysis of variance (ANOVA). The differences between the means were determined using the L.S.D. test at a significant level, p < 0.05. Statistical package Unistat (Unistat, Inc., USA) was used.
Results and Discussion
Identification of Chemical Compounds of A. psilostachya
GC/MS analysis of the essential oils of A. psilostachya allowed the identification of 44 compounds from aerial plant parts and 22 compounds from roots (Table 1). Our results revealed that the main constituents of A. psilostachya essential oil from shoot and leaves were isoaromadendrene epoxide (17.38 %), citral (15.13 %), caryophyllene (7.23 %), borneol (8.76%), (+)‐2‐bornanone (6.45 %), eucalyptol (4.61 %), whereas the major compounds from root were endo‐borneol 22.76%, (+)‐2‐bornanone 8.08%, caryophyllene oxide 7.77%, citral 14.65%, cis‐verbenol 4.43%, and eucalyptol 4.72% (Table 1). All other components were detected with content below 5%. The chemical structure of major compounds identified in the essential oil of the shoot and root of A. psilostachya is presented in Figure 1.
TABLE 1: Essential oil composition of Ambrosia psilostachya.
| Chemical component of the shoot | |||
|---|---|---|---|
| Chemical name | Formula | RT | AREA SUM% |
| Heptane | C7H16 | 5.42 | 0.26 |
| Camphene | C10H16 | 9.48 | 0.56 |
| Sulcatone | C8H14O | 9.94 | 0.3 |
| Eucalyptol | C10H18O | 26.52 | 4.61 |
| Sabinene hydrate | C10H18O | 11.74 | 0.69 |
| cis‐Verbenol | C10H16O | 11.91 | 0.18 |
| Caren‐4‐ol | C10H16O | 12.05 | 0.49 |
| trans‐Verbenol | C10H16O | 12.428 | 3.03 |
| (+)‐2‐Bornanone | C10H16O | 12.49 | 6.45 |
| Rosefuran epoxide | C10H14O2 | 12.71 | 0.47 |
| Borneol | C10H18O | 12.77 | 8.76 |
| Terpinen‐4‐ol | C10H18O | 12.87 | 1.5 |
| (‐)‐trans‐Isopiperitenol | C10H16O | 13.15 | 1.67 |
| Levoverbenone | C10H14O | 13.34 | 1.64 |
| Citral | C10H16O | 13.942 | 15.13 |
| Thymol | C10H14O | 14.158 | 2.47 |
| E‐Methylgeranate | C11H18O2 | 14.533 | 0.34 |
| Nerol acetate | C12H20O2 | 15.176 | 3.57 |
| Copaene | C15H24 | 15.398 | 0.69 |
| Caryophyllene | C15H24 | 15.958 | 7.23 |
| β‐Cedrene | C15H24 | 16.034 | 0.29 |
| 1,4,7,‐Cycloundecatriene, 1,5,9,9‐ tetramethyl‐, Z,Z,Z | C15H24 | 16.327 | 0.73 |
| trans‐. β‐Ionone | C13H20O | 16.511 | 0.48 |
| β‐cubebene | C15H24 | 16.6 | 0.41 |
| Cadinenes | C15H24 | 16.944 | 0.33 |
| Kessane | C15H26O | 17.141 | 0.18 |
| Caryophyllene oxide | C15H24O | 17.383 | 0.52 |
| Palustrol | C15H26O | 17.561 | 0.71 |
| Spatulenol | C15H24O | 17.631 | 4 |
| Bisabolene | C15H24O | 17.694 | 2.42 |
| Isoaromadendrene epoxide | C15H24O | 17.739 | 17.38 |
| Viridiflorol | C15H26O | 17.809 | 2.15 |
| Ledol | C15H26O | 17.91 | 0.73 |
| Humulene epoxide 2 | C15H24O | 17.987 | 1.99 |
| Benzoic acid | C16H30O4Si3 | 18.03 | 0.24 |
| Longipinane | C15H26 | 18.01 | 0.49 |
| Isospathulenol | C15H24O | 18.17 | 0.49 |
| 7R,8R‐8‐Hydroxy‐4‐isopropylidene‐7methylbicyclo[5.3.1]undec‐1‐ene | C15H24O | 18.21 | 3.85 |
| alpha‐Cadino | C15H26O | 18.324 | 0.58 |
| 4(15),5,10(14)‐Germacratrien‐1‐ol | C15H24O | 18.37 | 0.59 |
| Tricyclo[5.2.2.0(1,6)]undecan‐3‐ol, 2methylene‐6,8,8‐trimethyl | C15H24O | 18.52 | 0.27 |
| Longipinocarveol, trans‐ | C15H24O | 18.67 | 0.72 |
| Ylangenol | C15H24O | 19.25 | 0.18 |
| Hydroxy‐α‐muurolene | C15H24O | 19.4 | 0.22 |
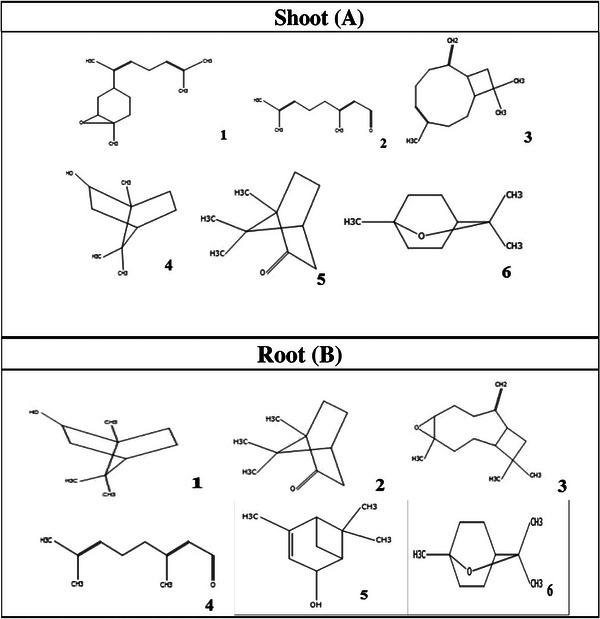
The identification of chemical components of A. psilostachya essential oil has been documented in some previous studies [ref. 21, ref. 22]. Our results showed that there were differences between the main components of the essential oil of A. psilostachya and those previously reported in A. trifida [ref. 23] and A. artemisiifolia [ref. 24], which belong to the same genus (Table 2). Interestingly, the chemical profile of A. psilostachya essential oil differed significantly in the present study, compared to previous reports. Sesquiterpenoids, flavonoids, terpenoids, steroids, and coumarins are the major compounds identified in Ambrosia spp. [ref. 25]. The main compounds of A. psilostachya essential oil obtained in the current study, such as the isoaromadendrene epoxide, citral, β‐caryophyllene, bornanone, eucalyptol, differed from the composition reported by [ref. 21], rich in germacrene, trans‐β‐ocimene, and β‐caryophyllene. Furthermore, it can be noted that the major essential oil components of the shoots of A. psilostachya were oxygenated sesquiterpenes and monoterpene aldehydes, whereas, in a similar study, the chemical compositions of the essential oils isolated from parts of A. psilostachya included sesquiterpene hydrocarbons and monoterpene hydrocarbons as the principal compounds [ref. 21]. The results obtained can be attributed to the fact that Ambrosia spp. is a very diverse genus of Asteraceae, presenting a high degree of morphological and chemical variability [ref. 26]. Different factors, including genetic, biotic, and abiotic factors, could influence the main constituents of essential oil, which may affect the quantity and quality of the plant’s secondary metabolites [ref. 27]. In fact, plants are forced to change the function and chemical composition of their essential oils to adapt to different environmental conditions [ref. 28]. Moreover, moisture and soil can also affect essential oil production and their quality and biological activity [ref. 29, ref. 30, ref. 31].
TABLE 2: The main composition (relative amount, %) of the essential oils from Ambrosia psilostachya, Ambrosia trifida, and Ambrosia artemisiifolia.
| A. trifida | A. artemisiifolia | A. psilostachya |
|---|---|---|
| Bornyl acetate (15.5%) | Germacrene D (24.1%) | Epoxide (17.38 %) |
| Borneol (8.5%) | Limonene (16.8%) | Citral (15.13 %) |
| Caryophyllene oxide (8.3%) | α‐pinene (8.0%) | Caryophyllene (7.23 %) |
| α‐Pinene (8.0%) | β‐Myrcene (7.4%) | (+)‐2‐Bornanone (6.45 %) |
| Germacrene D (6.3%) | Borneol (2.9%) | Borneol (8.76%) |
Phytotoxic Activities of Extracts of A. psilostachya
ANOVA analysis revealed significant (p ≤ 0.05) differences among mean squares of LAI, biomass dry weight, lengths of the shoot, and yield under various concentrations. The influence of A. psilostachya aqueous extracts on the target plants is shown in Figure 2A–D. Results showed that increasing the concentrations of A. psilostachya extracts caused a reduction in both bean and peanut growth traits.
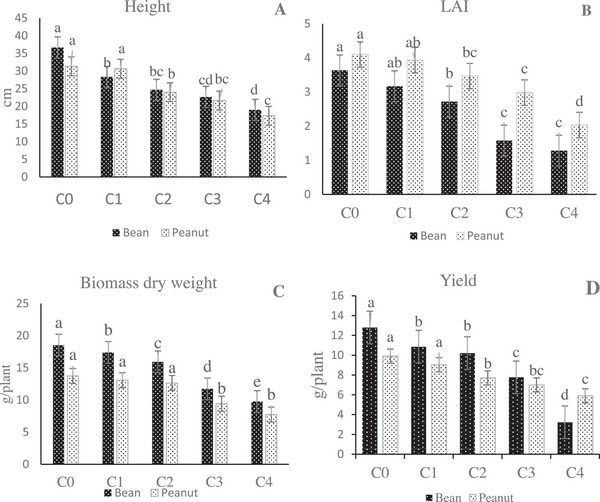
The application of aqueous extracts of A. psilostachya caused a considerable decrease in the height of bean plants at all concentrations. As the concentration of A. psilostachya aqueous extracts increased, the height of seedlings gradually decreased compared to the control plants. In peanut plants, there was no significant difference in the height between control plants and 25% concentration, but at higher concentrations of the aqueous extract, the height decreased. LAI showed variations in both crop plants. When compared with control (C0), lower concentrations (i.e., C1 and C2 treatments) of aqueous extracts had no significant effects on the LAI of both the crops, while the highest concentration (C4) inhibited LAI. In bean plants, the magnitude of the percent of decrease at different concentrations followed the order: C1 (12%) < C2 (25%) < C3 (56%) < C4 (64%) in LAI, respectively, compared to control, while in peanut plants, the percent of the decrease in LAI was C1 (4%) < C2 (15%) < C3 (27%) < C4 (50%). The results of biomass dry weight showed that the dry weight of the bean plants was significantly inhibited by all the concentrations of the aqueous extracts. In contrast, the biomass dry weight of the peanut plants was inhibited at a concentration of 75% and above. The aqueous extract of A. psilostachya significantly (p = 0.05) reduced the yield of crop species. At the concentration of 100%, the yield of beans and peanuts was reduced by nearly 60% and 40%, respectively. Compared to the control plants, the 25% concentration of aqueous extracts of A. psilostachya had no significant effect on the yield of peanut plants, but, in bean plants, this parameter showed a significant reduction at all concentrations.
Allelopathic activity of Ambrosia spp. has been documented on germination, seedling growth, and yield of crop species such as corn, maize, rice, wheat, and soybean [ref. 9, ref. 15, ref. 32], and extracts from different species of the genus Ambrosia exhibited significant inhibitory effects on other plants, especially crop seedlings [ref. 7, ref. 33]. In conformity with previous studies, the results of this study showed that the presence of A. psilostachya strongly affected the growth traits of test crop plants with a dose‐dependent trend, so that the increase in the concentration of A. psilostachya extract decreased growth traits of both bean and peanut plants. In previous literature, it was found that the presence of allelochemical compounds inhibited cytokine activity and decreased the height of plants [ref. 34, ref. 35], and, consistent with these studies, the height of both crop plants decreased with increasing the concentration of A. psilostachya extract. Results showed that LAI was also inhibited by A. psilostachya aqueous extract in the tested plants after treatment. Under stress conditions, due to a decrease in cell number and cell size, leaf area decreases [ref. 36]. The high concentrations of A. psilostachya extracts, by inhibition of LAI, led to a reduction in the growth of the tested plants. This reduction may be related to the inhibition of cell division and/or cell expansion due to the presence of chemical components in the extracts [ref. 37, ref. 38]. Furthermore, the allelopathic compounds can affect enzymatic activity, chlorophyll synthesis, cell viability, and organelle size [ref. 39, ref. 40]. These results suggest that the invasion of A. psilostachya in agricultural areas could severely affect crop production. In this study, the bean was more sensitive than the peanut to A. psilostachya aqueous extract, so that the yield of peanut plants was only reduced by 40% at the 100% concentration, while the yield of bean was reduced by 74% at the same concentration. Moreover, the result showed that the extract did not significantly inhibit the yield of peanut plants at low concentrations, which could suggest that the allelopathic compounds in A. psilostachya have a limited effect on this parameter in peanut. In other words, peanut plants were less affected by A. psilostachya extract concentration than beans. These findings are in agreement with the results of another study, which reported that different crops showed different sensitivity to the A. tirifida [ref. 7]. These results show that the response of plants to allelopathic interference of invasive species may differ from species to species. Under stressful situations presence of enzymes such as catalase, superoxide dismutase, and peroxidase regulates ROS accumulation and reduces cellular damage [ref. 41]. Therefore, plants show different tolerance to adverse conditions.
The observed inhibitory effects of A. psilostachya extracts could be attributed to the presence of allelochemical compounds, which have phytotoxic activity. Allelochemicals can inhibit the process of photosynthesis so that the plant growth and the biomass dry weight of the seedlings decrease [ref. 42]. The presence of stress in the form of allelochemical compounds could disrupt cell division and enlargement, thus inhibiting the growth of plants. The mode of action of many allelochemicals is the production of reactive oxygen species (ROS) and induction of oxidative stress [ref. 43]. The composition and major components of plant extracts play a significant role in their biological activity. Maximum inhibition of aqueous extracts of A. psilostachya could be due to the release of phytotoxins in the soil [ref. 44]. In this regard, many reports confirmed that Ambrosia spp. exhibited powerful phytotoxic effects on germination and seedling growth, such as lettuce, watermelon, corn, and tomato [ref. 9, ref. 45, ref. 46]. In line with a previous study, the extract of A. artemisiifolia decreased the dried weight of lettuce plants [ref. 10]. It was also reported that the aerial parts of A. trifida have the potential to produce harmful phytochemicals, which have an inhibitory effect on plant growth [ref. 23]. In the present study, the potent phytotoxic activity of the A. psilostachya could be ascribed to the presence of oxygenated compounds such as isoaromadendrene epoxide already reported in other plant species, including Artemisia spp. [ref. 47], Rosmarinus officinalis [ref. 48], and Cinnamomum camphora [ref. 49, ref. 50]. Reported that the antimicrobial activity of the plant components in part may be associated with the high percentage of isoaromadendrene epoxide, caryophyllene oxide, eudesmol, and aromadendrene epoxide. Therefore, due to the high concentration of isoaromadendrene epoxide, citral, caryophyllene, and caryophyllene oxide in this plant, the allelopathy activity of A. psilostachya could be associated with their presence.
Conclusions and Perspectives
To the best of our knowledge, this is the first report identifying the chemical components of A. psilostachya essential oil in Iran. The results of this study confirm that A. psilostachya has a strong phytotoxic potential on crop plants such as bean and peanut, so that its adverse effects increase with increased concentration. Based on the high concentration of isoaromadendrene epoxide, citral, caryophyllene, and caryophyllene oxide in essential oil and their allelopathic activity, the allelopathy of A. psilostachya essential oil could be associated with their presence. Due to the different sensitivity of crop plants to A. psilostachya essential oil, the results of this study could help choose crop plants when heavy A. psilostachya infestations occur. Finally, it should be noted that the results of this study could also promote the use of A. psilostachya as a source to manage weeds.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- 1 E. L. Rice , Allelopathy (Academic Press, 2012).
- Making Allelopathy Respectable,”. Science, 2003. [DOI | PubMed]
- 3 S. Latif , G. Chiapusio , and L. Weston , “Allelopathy and the Role of Allelochemicals in Plant Defence,” in Advances in Botanical Research, ed. G. Becard (Elsevier, 2017), 19–54.
- Allelopathic Plants 21. Mikania micrantha HBK,”. Allelopathy Journal, 2007
- Absence of Mutagenicity in Somatic and Germ Cells of Mice Submitted to Subchronic Treatment With an Extract of Croton cajucara Benth. (Euphorbiaceae),”. Genetics and Molecular Biology, 2006. [DOI]
- 6 H. Shabaitah , Allelopathic Effect of Plant Extract From Ambrosia artemisiifolia on Inhibition of Orobacnhe aegyptiaca Seed Germination and Plant Host Interaction (Palestine Technical University‐Kadoorie, 2022).
- Chemical Composition of Ambrosia trifida L. and Its Allelopathic Influence on Crops,”. Plants, 2021. [PubMed]
- Allelopathic Effect of Three Weed Species on the Germination and Seedling Growth of Tomato (Lycopersicon esculentum),”. International Journal for Science and Research‐Granthaalayah, 2022
- The Allelopathic, Cytotoxic and Genotoxic Effect of Ambrosia artemisiifolia on the Germination and Root Meristems of Zea mays,”. Caryologia, 2018. [DOI]
- Allelopathic Effects of Ambrosia artemisiifolia L. in the Invasive Process,”. Crop Protection, 2013. [DOI]
- Evidence for Allelopathy as a Mechanism of Community Composition Change by an Invasive Exotic Shrub, Chrysanthemoides Monilifera spp. Rotundata,”. Plant and Soil, 2009. [DOI]
- Allelopathic Interference of Ambrosia trifida With Wheat (Triticum aestivum),”. Agriculture, Ecosystems & Environment, 2007. [DOI]
- Heterogeneity in Ragweed Pollen Exposure Is Determined by Plant Composition at Small Spatial Scales ,”. Science of the Total Environment, 2014. [DOI | PubMed]
- First Report of Ambrosia Psilostachya From Iran: An Invasive Plant Species Establishing in Coastal Area of Gilan Province (N Iran),”. Plant Journal, 2017
- Possible Role of Ambrosia psilostachya on Pattern and Succession in Old‐fields,”. American Midland Naturalist (, 1971. [DOI]
- The Worldwide Spread, Success, and Impact of Ragweed (Ambrosia spp.),”. Critical Reviews in Plant Sciences, 2017. [DOI]
- Emerging Invasive Weeds in Iran: Occurrence, Ecological Impacts, and Sustainable Management,”. Plants, 2025. [DOI | PubMed]
- Biologically Active Components and Nutraceuticals in Peanuts and Related Products: Review,”. Food Reviews International, 2007. [DOI]
- Water Distillation Extraction of Essential Oil From Sideritis raeseri Herb,”. Environmental Processes, 2019. [DOI]
- 20 European Directorate for the Quality of Medicines & HealthCare , Guide to the Preparation, Use and Quality Assurance of Blood Components: Recommendation No. R (95) 15 (Manhattan Publishing Company, 1997).
- Chemical Composition and Allelopathic Activity of Parthenium hysterophorus and Ambrosia polystachya Weeds Essential Oils,”. American Journal of Plant Sciences, 2014
- The Structure of Ambrosiol. A New Sesquiterpene Lactone From Ambrosia psilostachya ,”. The Journal of Organic Chemistry, 1966. [DOI]
- Chemical Composition and Antimicrobial Activity of Ambrosia artemisiifolia L. essential Oil,”. Journal of Essential Oil Research, 2004. [DOI]
- Chemical Composition and Antimicrobial Activity of the Essential Oil From Ambrosia trifida L,”. Molecules (Basel, Switzerland), 2006. [DOI | PubMed]
- A Comprehensive Review of Secondary Metabolites of the Plant Genus Ambrosia ,”. Phytochemistry Reviews (, 2025
- Biochemistry and Species Problems in Ambrosia (Asteraceae‐Ambrosieae),”. Plant Systematics and Evolution, 1976. [DOI]
- Phytotoxicity and Cytotoxicity of Essential Oil From Leaves of Plectranthus Amboinicus, Carvacrol, and Thymol in Plant Bioassays,”. Journal of Agricultural and Food Chemistry, 2015. [DOI | PubMed]
- Factors Influencing Yield, Chemical Composition and Efficacy of Essential Oils,”. International Journal of Multidisciplinary Research and Growth Evaluation, 2024. [DOI]
- Optimum Stand Density for Maximum Essential Oil Yield in Commercial Fennel Crops,”. HortScience, 2006. [DOI]
- Diversity in the Composition of Monoterpenoids of Origanum microphyllum (Labiatae),”. Biochemical Systematics and Ecology, 2002. [DOI]
- Environmental Factors Affecting Chemical Variability of Essential Oils in Thymus piperella L,”. Biochemical Systematics and Ecology, 1998. [DOI]
- Allelopathic Effects of Plant Extracts on Common Ragweed (Ambrosia artemisiifolia L.),”. Journal of Plant Diseases and Plant Protection, 2008
- Influence of Ambrosia artemisiifolia extract on Germination and Growth of Amaranthus retroflexus and Zea Mays,”. Agricultural Science Research Journal, 2019
- Potensi Ekstrak Daun Gulma Babadotan (Ageratum conyzoides L.) Terhadap Perkecambahan Dan Pertumbuhan Paspalum Conjugatum Berg,”. Al‐Kauniyah: Jurnal Biologi, 2013
- Allelochemical Effect of Ageratum conyzoides L. Leaf Extract on Soybean [Glycine max (L.) Merr. cv Grobogan] Growth,”. Journal of Physics: Conference Series, 2018
- Effect of Water and Salt Stresses on the Growth, Gas Exchange and Water Relations in Argyranthemum coronopifolium Plants,”. Plant Science, 1998
- Effect of Soil Type on the Allelotoxic Activity of Medicago sativa L. residues in Vicia faba L. agroecosystems,”. Journal of Taibah University for Science, 2014
- Control of Parthenium hysterophorus L., by Aqueous Extracts of Allelopathic Grasses,”. Pakistan Journal of Botany, 2006
- Defensive Response of Radish Seedlings to the Oxidative Stress Arising From Phenolic Compounds in the Extract of Peppermint (Mentha× piperita L.),”. Scientia Horticulturae, 2017. [DOI]
- Phytotoxicity and Cytotoxicity of Citrus Aurantiifolia Essential Oil and Its Major Constituents: Limonene and Citral,”. Industrial Crops and Products, 2017. [DOI]
- Allelopathic Effects of Cinnamomum septentrionale Leaf Litter on Eucalyptus Grandis Saplings,”. Global Ecology and Conservation, 2020. [DOI]
- Potensi Alelopati Ekstrak Serasah Daun Mangga (Mangifera indica (L.)) Terhadap Pertumbuhan Gulma Rumput Grinting (Cynodon dactylon (L.)) Press.”. Jurnal Protobiont, 2015
- 43 A. Gniazdowska , U. Krasuska , O. Andrzejczak , and D. Soltys , “Allelopathic Compounds as Oxidative Stress Agents: YES or NO,” in Reactive Oxygen and Nitrogen Species Signaling and Communication in Plants, eds. K. Gupta and A. Igamberdiev (Springer, 2015): 155–176.
- Air‐drying Depresses Rates of Leaf Litter Decomposition,”. Soil Biology and Biochemistry, 1998. [DOI]
- Chemical Composition and Phytotoxicity of Essential Oil From Invasive Plant, Ambrosia artemisiifolia L,”. Ecotoxicology and Environmental Safety, 2021. [DOI | PubMed]
- Effects and Related Mechanisms of Allelopathy of Ambrosia trifida’s Pollens on Corn Pollens, Stigmas, and Yield,”. Journal of Agricultural and Food Chemistry, 2024. [DOI | PubMed]
- Volatile Oils Content of Some Species of Artemisia Growing Under Different Environmental Conditions and Its Effect on Germination of Seeds of Some Plants,”. IOSR Journal of Agriculture and Veterinary Science, 2018
- Gas Chromatography Mass Spectrum and Fourier‐transform Infrared Spectroscopy Analysis of Methanolic Extract of Rosmarinus oficinalis Leaves,”. Journal of Pharmacognosy and Phytotherapy, 2015
- Algicidal Properties of Extracts From Cinnamomum camphora Fresh Leaves and Their Main Compounds,”. Ecotoxicology and Environmental Safety, 2018. [DOI | PubMed]
- Chemical Composition of the Essential Oil of Carduncellus helenioides (Desf.) Hanelt From Algeria,”. Oriental Journal of Chemistry, 2016. [DOI]
