Cannabinoid use generalizes stress responses by altering the astrocyte plasticity through extracellular matrix signaling in the nucleus accumbens core
Abstract
The rising legal acceptance of cannabis and the high comorbidity between cannabis use disorder (CUD) and post-traumatic stress disorder (PTSD) highlight the importance of understanding how stress and cannabis influence the brain. We recently discovered that cannabis use promotes two PTSD-like symptoms: avoidance coping behaviors and the generalization of stress-coping responses to a neutral stimulus not previously linked to stress. To investigate the neuroadaptations behind these changes, we used in vivo zymography and confocal microscopy to examine how stress and cannabinoid use influence multipartite synaptic plasticity in the nucleus accumbens core (NAcore), including astroglial plasticity, Synapsin-I density, and matrix metalloproteinases (MMP-2,9) activity. For this purpose, rats were restraint stressed for 2h and simultaneously exposed to an odor that became the stress-conditioned stimulus (stress-CS). Three weeks later, rats self-administered cannabinoids (delta9-tetrahydrocannabinol + cannabidiol; THC + CBD) for 10 days, followed by 10 days of withdrawal. We then evaluated the effect of stress-CS or neutral odor (NS) on coping strategies in a defensive burying task. We demonstrated for the first time that THC + CBD generalized stress responses to the NS by causing astrocytes to retract from synapses and decreasing Synapsin-I density in the NAcore. Furthermore, cannabinoid use promoted avoidant coping behaviors in response to a stress-CS by triggering strong activation of MMP-2,9, driven largely by MMP-2, causing a re-association of astrocytes to synapses along with an increase of Synapsin-I density and astrocyte atrophy. However, these neuroadaptations only occurred in males. Overall, these findings highlight potential therapeutic targets like MMPs and astrocytes for treating co-occurring CUD/PTSD.
Affiliations: Medical University of South Carolina
License: CC BY 4.0 This work is licensed under a Creative Commons Attribution 4.0 International License, which allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use.
Article links: DOI: 10.21203/rs.3.rs-7254957/v1 | PubMed: 40894006 | PMC: PMC12393475
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (5.2 MB)
Introduction
For certain individuals, a single traumatic event can lead to trauma and stress-related disorders, including post-traumatic stress disorder (PTSD)[ref. 1]. Cannabis use disorder (CUD) is often comorbid with PTSD and is the most prevalent co-occurring condition among veterans diagnosed with CUD[ref. 2]. Patients with comorbid PTSD and CUD report higher PTSD symptom severity and poorer treatment outcomes relative to those without CUD[ref. 3, ref. 4]. Furthermore, recent longitudinal studies demonstrate that cannabis use is associated with higher PTSD symptom severity, including greater traumatic thought intrusions[ref. 5, ref. 6] and depressive symptoms[ref. 7]. Despite this evidence, PTSD is often listed as a qualified medical condition for the use of cannabis in the United States[ref. 8, ref. 9]. As of 2025, 31 states listed cannabis as a treatment for PTSD[ref. 10]. Consequently, the increasing use of cannabis for self-medication of PTSD and the comorbid PTSD and CUD underscores the need to identify the neurobiological mechanisms through which cannabis use alters and potentially exacerbates the symptoms associated with PTSD.
Using an stress cue-conditioned (stress-CS) animal model of PTSD, we recently demonstrated that an acute stress potentiates cannabis consumption in male rats[ref. 11]. Additionally, we discovered that cannabis use promotes two significant PTSD-like symptoms: the generalization of stress-coping responses to a neutral stimulus that is not produced by stress exposure in the absence of cannabis use, and engaging in avoidance coping behaviors when faced with a conditioned stress odor[ref. 11]. Importantly, these findings differ from those observed after cocaine and heroin self-administration, where rats exposed to a stress-CS engage in active coping behaviors, without generalizing to an odor not associated with acute stress experience[ref. 12–ref. 14]. Finally, the cannabis-induced behavioral changes were accompanied by reduced spine density in the nucleus accumbens core (NAcore) and a further decrease in spine head diameter after exposure to the stress-CS[ref. 11], indicating that cannabis may be altering how environmental stimuli elicit synaptic plasticity.
Nonneuronal components of the neuropil adjacent to synapses, including astroglia and extracellular matrix (ECM), form what is termed the multipartite synapse and function together with pre- and postsynapses to regulate synaptic plasticity[ref. 15, ref. 16]. For instance, astrocytes regulate the synaptic plasticity by insulating synapses, thereby providing them with metabolic support, gliotransmitters, and maintaining ion homeostasis[ref. 16]. In parallel, destabilizing the ECM via catalytic signaling induced by matrix metalloproteinases (MMPs) is associated with neuronal and astroglial plasticity. Both environmental stressors and addictive drugs alter ECM signaling and astroglial morphology and signaling, and contribute to the development of stress- and drug-related disorders[ref. 17]. However, the effects of cannabis use, either independently or in conjunction with stress, on the nonneuronal components of multipartite synapses remain unknown. The objective of this study was to assess whether the interaction between stress and cannabis use influences components of multipartite synapses, including astroglial plasticity, synapse density, and ECM catalytic signaling in the NAcore. For this purpose, following an acute restraint stress, astrocytes were labeled in rats that were trained to self-administer delta9-tetrahydrocannabinol + cannabidiol (THC + CBD)[ref. 18]. After assessing how cannabinoid use affected coping strategies through a defensive burying task (DBT), we employed confocal microscopy, in vivo zymography, and digital rendering techniques to quantify astrocyte morphology, the colocalization of astrocytes with the presynaptic marker Synapsin-I, and the gelatinolytic activity of MMP-2 and MMP-9 in the NAcore.
Methods
Animals
Male and female Long-Evans rats (~ 250 g; Charles River Laboratories) were double-housed according to sex on a 12-h light/dark cycle with food and water available ad libitum. Experiments occurred in the light cycle. All procedures received approval from the Animal Care and Use Committee of the Medical University of South Carolina and were conducted following National Institutes of Health guidelines.
Acute stress and odor pairing
Rats were placed in a restraining device for 2 hours and paired with an odor (lemon or sandalwood; Sun essential, Phoenix, AZ, USA) that became the stress-CS. Control rats were exposed to the same odor in the home cage for 2 hours (Sham-CS). The odor that was not stress- or sham-paired was used during the DBT as a neutral odor control (stress-NS). Two weeks after the stress procedure, rats received indwelling catheters as previously described[ref. 18].
Viral labeling
After catheter implantation, rats were implanted with bilateral guide cannulae into the NAcore (AP: 1.5mm, ML: ± 1.7mm, DV: −5.5mm). AAV5/GFAP-hM3dq-mCherry (University of Zurich) was injected into the left hemisphere (1.0 μl, 0.15 μl/min, 5 min diffusion) to label astrocytes. The microinjectors are 2mm longer than the guide cannulae to reach the target.
THC + CBD self-administration
Following surgery, rats underwent 5 days of vapor exposure to a THC + CBD mixture in a 10:1 ratio, as previously described[ref. 18]. Rats were then trained to self-administer THC + CBD on a fixed ratio 1 schedule, during 90 min sessions for 10 consecutive days. Active lever presses delivered 0.4 + 0.04 mg/kg THC + CBD and were paired with cues (light + tone) followed by a 20-s timeout. Another cohort of rats was trained to self-administer the vehicle (1% ethanol). Following self-administration, animals underwent 10 days of withdrawal to ensure the absence of THC metabolites during the DBT.
Defensive burying task
Coping strategies were quantified as previously described[ref. 11]. Briefly, bedding was placed in one half of the standard home cage, opposite the odor. Rats were placed facing away from the odor on the bedding side of the cage, and behavior was recorded for 15 min using Ethovision XT software (Leesburg, VA, USA). Active (burying) and avoidant (escape, immobility and grooming) coping mechanisms were quantified.
In vivo zymography
In vivo zymography was used to quantify MMP-2,9 activity as previously described in detail[ref. 19]. FITC-gelatin was microinjected into the right hemisphere (1.5μl, 0.5μl/min, 3 min diffusion) immediately before the DBT. Rats were then perfused with 4% PFA immediately after the DBT and brains were sliced at 100μm. Only slices containing the injection track and the anterior commissure on the same frame were analyzed (ImageJ; National Institute of Mental Health, Bethesda, MD). MMP-2 (444288, MilliporeSigma) and MMP-9 (444278, MilliporeSigma) inhibitors were dissolved in 1% DMSO and bilaterally microinjected before FITC-gelatin in a separate rat cohort as previously described[ref. 20].
Immunohistochemistry
Left hemisphere slices, containing the GFAP-mCherry virus, were permeabilized for 15 min in PBS with 0.2% Triton X-100 (PBST), then blocked with 2% PBST containing 5% normal goat serum. Rabbit anti-synapsin-I antibody (1:1000, ab64581, Abcam) was incubated for 72 hours at 4°C. Slices were washed with 0.2% PBST, then incubated with Alexa Fluor 405(1:1000, ab175652, Abcam) for 72 hours at 4°C.
Confocal microscopy
Astrocyte images were acquired using a Leica SP5 with a 63X oil-immersion objective lens at 16-bit resolution with a 1-μm step size. Images were then imported into Imaris software (version 9.7.0). The mCherry signal was used for 3-dimensional reconstruction of astrocytes. The astrocyte-Synapsin-I proximity was quantified by using each rendered astrocyte as a region of interest (ROI) for colocalization analysis with Synapsin-I. The colocalization channel displays the results as the percentage of the ROI overlapping with Synapsin-I puncta. To determine the Synapsin-I density, we normalized the thresholded signal intensity to the volume of the frame from which it was acquired. Imaging and analysis were performed in a blinded manner.
Statistics
Data were analyzed with GraphPad Prism 10. Behavioral and in vivo zymography data were analyzed using two-way ANOVAs with Tukey’s post hoc comparison. For astroglial measures, a D’Agostino & Pearson test was initially used to assess the normality of each group. The Kruskal-Wallis test with Dunn’s post hoc was used for non-normally distributed treatment groups. Coping strategies and astroglial distribution were analyzed with a multiple Chi-square followed by a Bonferroni correction.
Results
Acute stress potentiated THC + CBD use in male rats
Rats were subjected to acute restraint stress for 2 hours and simultaneously exposed to an odor. 3 weeks after the stress, rats were trained to self-administer THC + CBD or its vehicle for 10 days, followed by 10 days of withdrawal (Fig. 1A). In THC + CBD trained rats, stressed males pressed more on the active lever than sham rats (Fig. 1B). Stressed males consumed more THC + CBD than sham rats (Fig. 1C). Both stressed and sham rats showed equivalent discrimination between the active and inactive levers (Fig. 1D). No differences between stress and sham groups were observed in vehicle-trained rats (Fig. 1E–G).
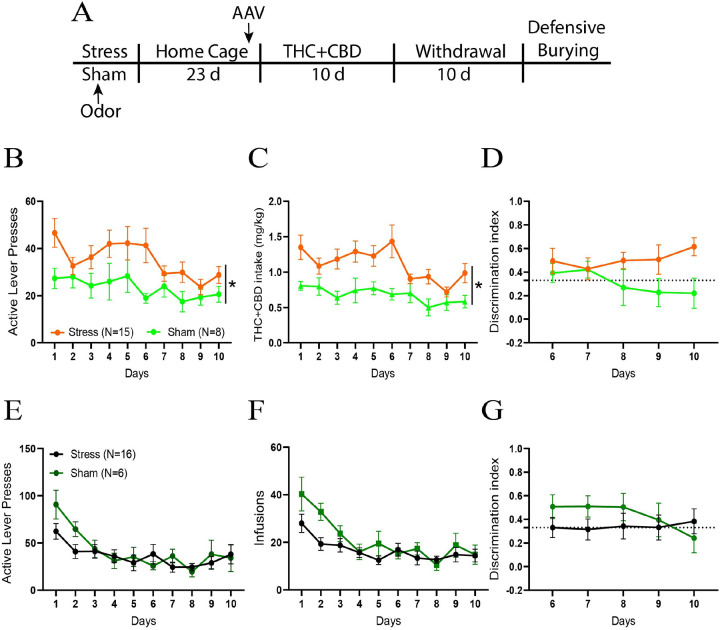
In female THC + CBD trained rats, no differences were measured between groups on the active lever presses and THC + CBD intake (Fig. S1A-B). However, stressed rats had a higher discrimination index than sham rats (Fig. S1C), indicating that the acute stress may increase the reinforcing value of the THC + CBD in females. No differences between stress and sham groups were observed in vehicle-trained rats (Fig. S1D-F).
THC + CBD promoted avoidant coping strategies in male rats
Following the withdrawal period, we used DBT to evaluate the effect of THC + CBD on active (burying) and avoidant (escape, immobility and grooming) coping strategies. To this end, stressed rats were exposed to either the stress-CS odor previously associated with the stressful event or a neutral odor stimulus (NS) for 15 min. Non-stressed rats were exposed to the odor previously associated with the sham experience. Regarding active coping, in vehicle-trained male rats, the stress-CS caused an increase in burying compared to the sham group. Conversely, in THC + CBD-trained rats, the stress-NS group exhibited greater burying than the sham group. Furthermore, compared to vehicle rats, THC + CBD self-administration elevated burying in the stress-NS group while decreasing it in the stress-CS group (Fig. 2A). Regarding avoidant coping strategies, we found that THC + CBD self-administration induced a strong escape behavior under all stress conditions compared to vehicle rats (Fig. 2B). Conversely, the percent time spent immobile was reduced after THC + CBD (Fig. 2C). Moreover, stress-CS significantly increased grooming in vehicle-trained rats, while both stressed groups (CS and NS) exhibited a higher grooming frequency than the sham group in THC + CBD-trained rats (Fig. 2D). When comparing the distribution of coping strategies between groups, we found no difference among the stress-NS and sham-CS groups in vehicle-trained rats (Fig. 2E), while the stress-CS promoted an active coping strategy by increasing burying behavior. In summary, Fig. 2 shows that THC + CBD withdrawal promoted avoidant coping in the sham group, while a neutral odor stimulus facilitated both active and avoidant coping in stressed rats. Also, exposure to the stress-CS after THC + CBD further changed stress behaviors compared to the sham and stress-CS veh groups. Combined, these data indicate that THC + CBD use caused male rats exposed to an earlier stressor to generalize stress responses to neutral stimuli and worsen stress responses to conditioned stimuli.
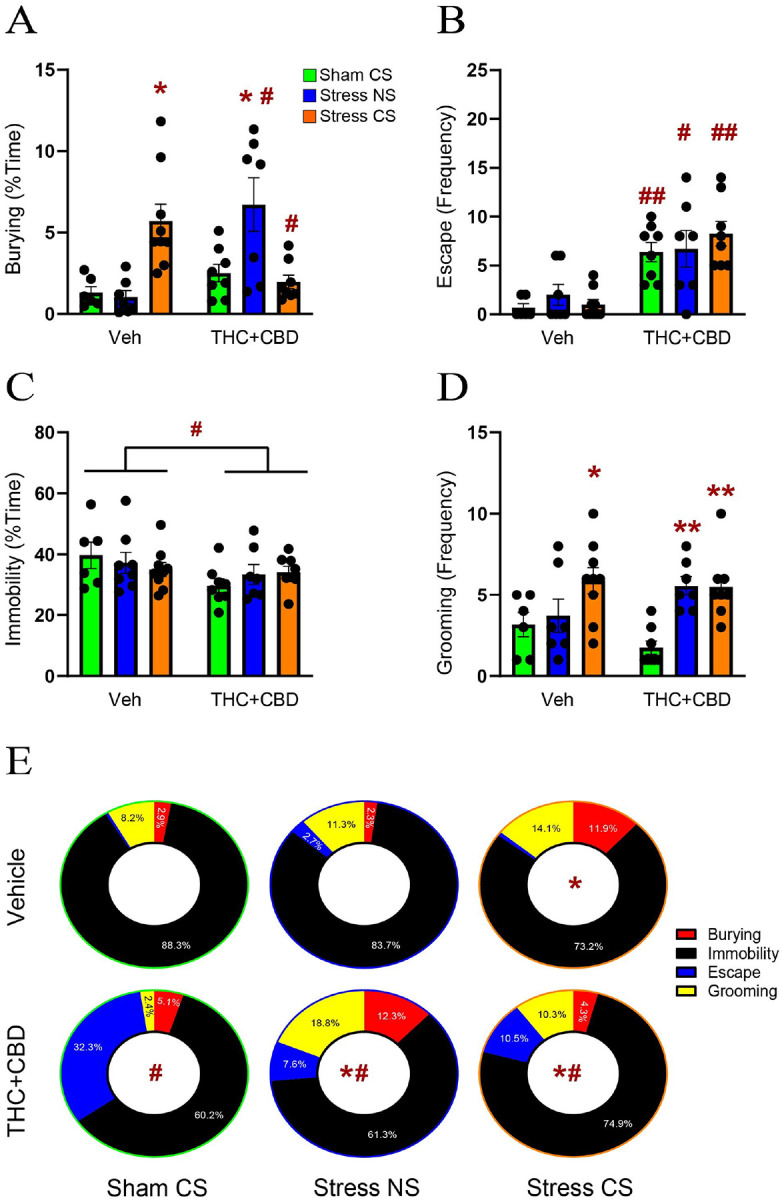
Surprisingly, in females, neither the stress nor the THC + CBD significantly affect coping strategies in the DBT (Fig. S2).
Withdrawal from cannabinoids reduced the synaptic proximity and promoted astrocytic insulation of synapses after the stress-CS exposure in male rats
To determine how stress and cannabinoids influence the plasticity of NAcore astrocytes, astroglia were labeled with a membrane-bound fluorescent reporter. Rats were perfused immediately after DBT, and NAcore slices labeled with the presynaptic marker Synapsin-I (Fig. 3A). To quantify proximity between astrocytes and synapses, each astrocyte was digitally rendered (Fig. 3B), and the co-labeling of the astrocyte surface with Synapsin-I was quantified (Fig. 3C). Consistent with our previous report[ref. 21], we found that acute stress (stress-NS) induced a constitutive reduction in astrocyte-Synapsin-I proximity, while exposure to the stress-CS resulted in a re-association between astrocytes and Synapsin-I in vehicle-trained male rats. Withdrawal from THC + CBD decreased astrocyte-Synapsin-I proximity in the sham group compared to vehicle rats. In stressed THC + CBD-trained rats, exposure to the NS further decreased the proximity between astrocytes and Synapsin-I relative to sham, while re-exposure to the stress-CS enhanced synaptic insulation by astroglia. Additionally, when compared to vehicle rats, the use of THC + CBD potentiated astrocytic insulation of synapses induced by the stress-CS (Fig. 3D, left). Because the cumulative frequency plots (Fig. 3D, middle) indicated different subpopulations of astrocytes, we divided the data into three subpopulations based on the values generating three equal populations in the sham-CS vehicle group. The multiple Chi2 analysis revealed that exposure to the stress-CS did not fully restore the astrocytic insulation of synapses in vehicle-trained rats (Fig. 3D, right). Indeed, the astrocytes having less co-registration with Synapsin-I constituted a larger population in the stress-CS group than in the sham-CS group.
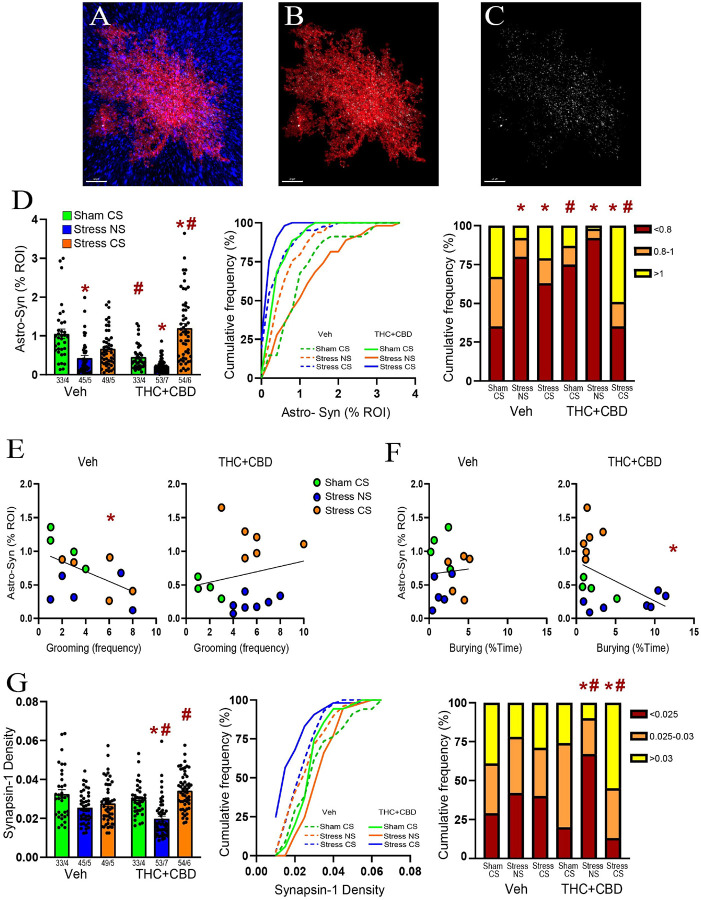
Astrocyte-Synapsin-I proximity negatively correlated with grooming in vehicle-trained males (Fig. 3E, left). This correlation disappeared after THC + CBD use (Fig. 3E, right). Furthermore, while no correlation existed between astrocyte-Synapsin-I proximity and burying in vehicle-trained rats (Fig. 3F, left), a negative correlation appeared after THC + CBD use (Fig. 3F, right). This illustrates that using cannabinoids alters the function of synaptic insulation in managing coping strategies.
In females, exposure to the stress-CS increased the astrocyte-Synapsin-I proximity in both vehicle- and THC + CBD-trained rats (Fig. S3A, left). Compared to vehicle rats, astrocytes near Synapsin-I were more abundant in the stress-NS group, while stress-CS exposure nearly eliminated astrocytes at mid-distance in THC + CBD females (Fig. S3A right). Moreover, in THC + CBD-trained females, astrocyte-Synapsin-I proximity correlated positively with burying behavior and negatively with grooming (Fig. S3B-C).
Combination of acute stress and cannabinoid use reduced Synapsin-I density in NAcore
Chronic stress is known to diminish the levels of synaptic markers, such as Synapsin-I[ref. 22]. We previously found that neither acute stress nor stress-CS exposure altered Synapsin-I density in the NAcore of male rats[ref. 21]. Nevertheless, the impact of acute stress followed by cannabinoid use on Synapsin-I remains unknown. Synapsin-I density was measured for each astrocyte digitally isolated. Similar to our prior report, Synapsin-I density remained unchanged in vehicle-trained males, regardless of the stress condition (Fig. 3G, left). Likewise, no difference was found between sham-CS groups. These findings suggest that the changes in astrocyte-Synapsin-I proximity observed in vehicle-trained rats and during withdrawal from THC + CBD result solely from astrocytic motility. However, the combination of acute stress and THC + CBD withdrawal lowered Synapsin-I density in stress-NS rats, while exposure to the stress-CS restored Synapsin-I density. The multiple Chi2 analysis indicated that astrocytes with high Synapsin-I density formed a larger population in the stress-CS group compared to the sham-CS group (Fig. 3G, right). Furthermore, THC + CBD increased Synapsin-I density in the stress-CS group compared to the vehicle rats. This indicates that the potentiation of synaptic insulation induced by the drug following the stress-CS exposure observed in Fig. 3D could result largely from increased neuronal synapses associated with astroglia.
Unlike males, the interaction between acute stress and cannabinoid use increased the Synapsin-I density in female NAcore (Fig. S3D, left). In stressed vehicle females, astrocytes with low Synapsin-I density increased compared to the sham-CS group. These effects are reversed after THC + CBD use (Fig. S3D, right).
Stress-CS induced astrocyte atrophy after cannabinoid use in male rats
Chronic stress causes astrocyte shrinkage in various brain areas[ref. 23]. Previously, we found that neither stress nor stress-CS influence astrocyte structure in the NAcore of males[ref. 21]. We replicated these negative data in vehicle males and demonstrated that after THC + CBD use the stress-CS reduced both the surface area and the volume of astrocytes (Fig. 4A–B left). Moreover, in Sham rats, THC + CBD use increased the subpopulation of astrocytes with smaller surface areas compared to Sham vehicle rats (Fig. 4A right). Additionally, in vehicle-trained rats, the stress-CS group exhibited a higher proportion of astrocytes with larger volumes than the sham group (Fig. 4B right).
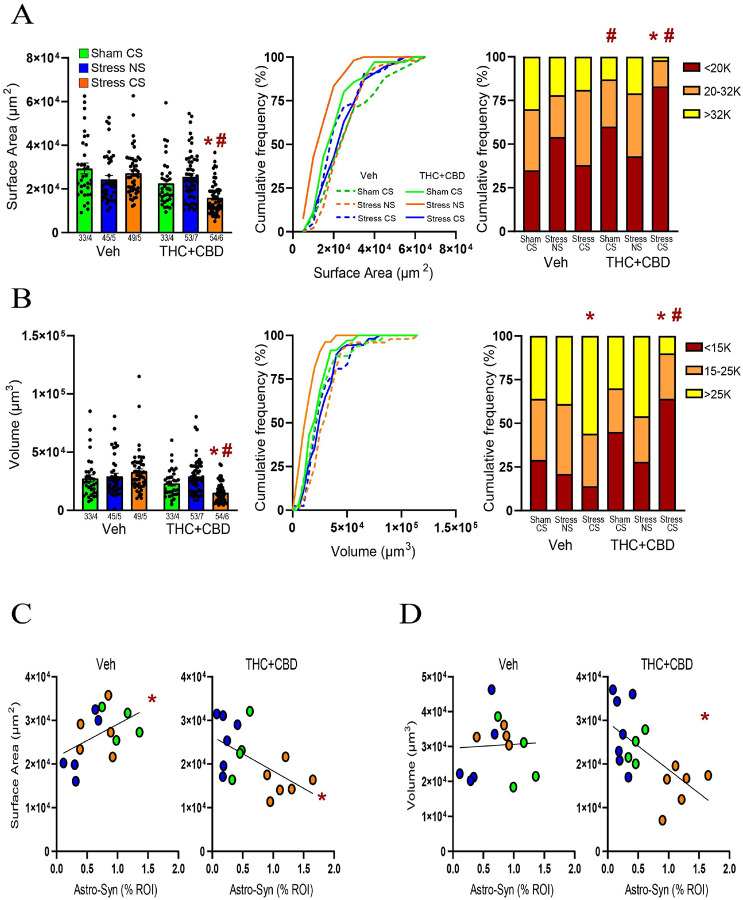
Cannabinoid use in males transformed a positive correlation between surface area and astrocyte-Synapsin-I proximity in vehicle-trained rats into a negative correlation (Fig. 4C). Additionally, in THC + CBD-trained rats, the volume showed a negative correlation with astrocyte-Synapsin-I proximity, unlike in vehicle rats (Fig. 4D). In summary, these data suggest that exposure to a stress-CS interacts with cannabinoid withdrawal to induce an increase in both astroglial coverage of synapses and astrocyte atrophy.
In females, the acute stress induced a constitutive decrease in astrocyte surface area and volume in both vehicle- and THC + CBD-trained rats. Re-exposure to stress-CS restored astrocyte morphology in vehicle females, but THC + CBD prevented this effect (Fig. S4A-B). Additionally, when comparing astrocyte morphology between sexes, THC + CBD withdrawal alone increased both the surface area and volume of astrocytes in females compared to males. (Fig. S4C-D).
MMP-2,9 activity is increased during withdrawal from cannabinoid use and is potentiated following the stress-CS exposure in male rats
MMP- 2 and – 9 are key components of the ECM signaling and multipartite plasticity [ref. 16, ref. 24]. Both addictive drugs and stress exposure alter the activity of MMP-2 and MMP-9[ref. 17]. Using in vivo zymography we quantified the gelatinolytic activity of MMP-2,9 during the DBT[ref. 19]. Withdrawal from THC + CBD increased NAcore MMP-2,9 activity regardless of the stress condition in males (Fig. 5B). Moreover, stress-CS exposure increased the MMP-2,9 activity in both vehicle and THC + CBD males. However, THC + CBD use potentiated the MMP-2,9 activity compared to vehicle rats.
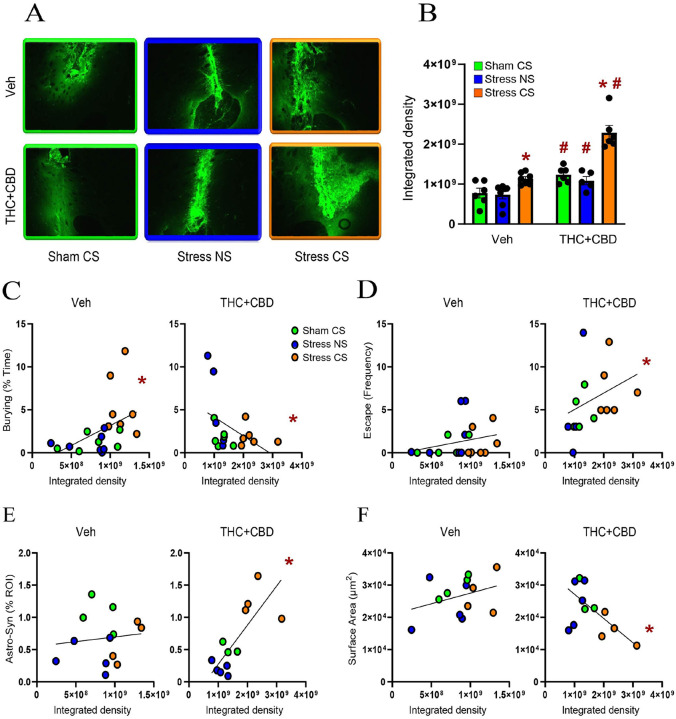
Cannabinoid use reversed a positive correlation between MMP-2,9 activity and burying behavior in vehicle-trained rats into a negative correlation (Fig. 5C). Moreover, MMP-2,9 activity was positively correlated with the escape behavior in THC + CBD-trained, not vehicle rats (Fig. 5D). These data indicate that cannabinoid use modifies how ECM signaling regulates coping strategies, specifically by inducing a shift from active to avoidant coping. Additionally, after cannabinoid use, MMP-2,9 activity positively correlated with astrocyte-Synapsin I proximity, while negatively correlating with astrocyte surface area (Fig. 5E–F), suggesting that ECM signaling regulates the astroglial plasticity.
Neither stress nor the THC + CBD alter the MMP-2,9 activity in females (Fig. S5A). However, similar to males, MMP-2,9 activity showed a positive correlation with astrocyte-Synapsin-I proximity and a negative correlation with astrocyte volume after cannabinoid use (Fig. S5B-C).
Both MMP-2 and MMP-9 regulated the astroglial coverage of synapses, while MMP-2 influenced the astrocyte morphology
To determine whether the changes in astroglial plasticity induced by the stress-CS in males are preceded by changes in the ECM catalytic signaling, we examined the effect of MMP-2 and MMP-9 inhibitors on stress-CS-induced astroglial plasticity. To this end, during THC + CBD withdrawal, another cohort of stressed males received bilateral microinjections of inhibitors (0.1 nmol) or their vehicle (1% DMSO) in NAcore, followed by a unilateral microinjection of the FITC-gelatin before stress-CS exposure (Fig. 6A). Compared to the DMSO microinjection, either MMP-2 or MMP-9 inhibitors altered the coping strategies. MMP-9 inhibitor notably reduced the burying behavior (Fig. 6B and S6). The coping strategies also significantly differ between the MMP-2 and MMP-9-treated groups (Fig. 6B), with the MMP-2 inhibitor significantly reducing grooming (Fig. 6B and S6). Interestingly, these alterations in stress-CS-induced behaviors were accompanied by only a nonsignificant reduction in gelatinolytic activity by either of the MMP inhibitors (Fig. 6C).
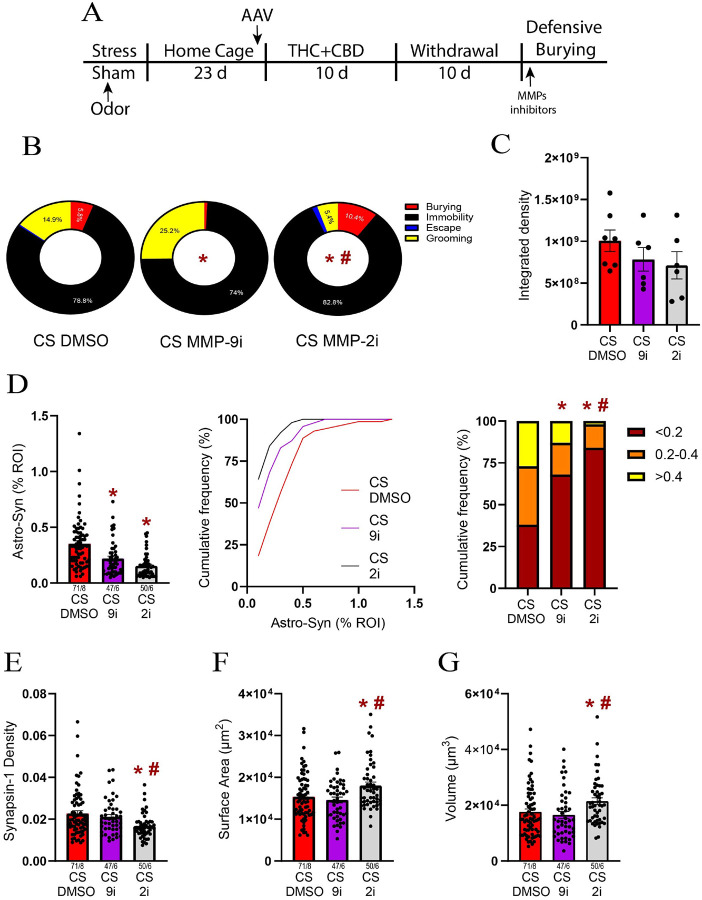
Both MMP-2 and − 9 inhibitors reduced the astroglial coverage of synapses (Fig. 6D left). Furthermore, the MMP-2 inhibitor nearly eliminated astrocytes found near Synapsin-I (Fig. 6D right). A likely contributor to the reduction in synaptic coverage by astroglia was markedly reduced Synapsin-I density after treatment with the MMP-2, but not MMP-9 inhibitor (Fig. 6E). Finally, the MMP-2 inhibitor reversed astrocyte atrophy by increasing both surface area and volume (Fig. 6F–G).
Discussion
We recently demonstrated that cannabis use, after a single traumatic event, promotes two PTSD-like symptoms: generalization of stress responses to neutral stimuli and avoidant coping strategies following conditioned stress exposure[ref. 11]. To explore the neuroadaptations behind these changes, we used in vivo zymography and confocal microscopy to assess how stress and cannabinoid use affect multipartite synaptic plasticity in the NAcore, a brain region linked to stress and addiction cues responding[ref. 2]. We made four primary discoveries: 1) cannabinoid withdrawal, without any traumatic event, reduced the astroglial coverage of synapses and increased MMP-2,9 signaling in the ECM, 2) a prior traumatic event reduced Synapsin-I density during cannabinoid withdrawal, 3) exposure to a stress-CS after cannabinoid use led to avoidant coping strategies by activating MMP-2,9 in the NAcore, resulting in the re-association of astrocytes with synapses and astrocyte atrophy, and 4) these multipartite adaptations by stress and cannabinoids occurred only in males.
THC + CBD disrupts coping strategies in males
In line with our previous report, we found that THC + CBD use generalized stress responses to a neutral stimulus in stressed rats, by inducing both active (burying) and avoidant (escape and grooming) coping strategies. Importantly, a neutral stimulus did not trigger stress responses in vehicle-trained rats, suggesting that the effects seen in THC + CBD-trained rats result from the interaction between acute stress and cannabinoid withdrawal. Additionally, we found that cannabinoid use induced notable escape behavior in non-stressed rats. Both preclinical and clinical studies support a contention that extensive and prolonged cannabis use is associated with more severe withdrawal symptoms[ref. 25–ref. 27] and our data indicate that cannabinoid use, regardless of previous traumatic experiences, generalizes stress responses to unconditioned stimuli. Since stress generalization is a hallmark of PTSD[ref. 1], these preclinical data support longitudinal clinical studies[ref. 5, ref. 6] indicating that cannabis use worsens PTSD symptoms. Furthermore, we previously demonstrated that a stress-CS is sufficient to reinstate cocaine, heroin, and alcohol seeking, reflecting an active coping strategy[ref. 12, ref. 14]. Accordingly, we found that the stress-CS induced burying behavior in vehicle-trained rats. However, after cannabinoid use, re-exposure to the stress-CS reduced burying while promoting escape and grooming behaviors. Since avoidance behavior is a key criterion for defining PTSD, and avoidance coping strategies are associated with increased activation of the stress axis[ref. 28], our findings suggest that cannabis use heightened the salience of the stress-CS and thereby worsened PTSD symptoms.
THC + CBD use alters how astroglial coverage regulates coping strategies in males
The synaptic insulation provided by astrocytes plays a crucial role in synaptic plasticity by supplying metabolic support, gliotransmitters, and maintaining ion homeostasis[ref. 16]. Withdrawal from cocaine, heroin, and methamphetamine self-administration reduced the proximity between astrocytes and synapses in NAcore[ref. 29–ref. 32]. Similarly, we demonstrated that withdrawal from THC + CBD reduced the astroglial coverage of synapses in NAcore, suggesting that this effect is shared by many classes of addictive drugs. Akin to withdrawal from addictive drugs, an acute restraint stress also induced enduring decreases in astroglial coverage in NAcore[ref. 21]. Consistently, we found that acute stress reduced astrocyte-Synapsin-I proximity in both vehicle- and THC + CBD-trained rats. Finally, we demonstrated that re-exposure to the stress-CS increased the astroglial coverage of synapses in both vehicle- and THC + CBD-trained rats. However, while the stress-CS only partially restored the astrocytic insulation of synapses in vehicle-trained rats, cannabinoid exposure potentiated the re-association of astrocytes to synapses induced by the stress-CS. In examining astroglial insulation related to coping strategies, we found a negative correlation between astrocytes-Synapsin-I proximity and grooming behavior in vehicle-trained rats. This implies that astroglial coverage may partly countermand the expression of avoidant coping behaviors, although this correlation vanished following THC + CBD usage. Conversely, in THC + CBD-trained males, the proximity of astrocytes to Synapsin-I was negatively correlated with burying behavior. Thus, THC + CBD inhibits active coping strategies and potentially exacerbates the salience of the stress-CS by promoting synaptic insulation by astrocytes. Overall, this finding suggests that stress-CS exposure during cannabinoid withdrawal alters how astroglial coverage in the NAcore influences coping strategies.
THC + CBD use following an acute stress reduces the Synapsin-I density in NAcore
Severe stressors are known to decrease the levels of synaptic markers, including Synapsin-I and PSD95, in several brain regions. For example, chronic unpredictable mild stress (CUMS) decreases the expression of both Synapsin-I and PSD95 in the prefrontal cortex and hippocampus[ref. 22, ref. 33] and a single prolonged stress (SPS) reduces Synapsin-I and PSD95 in the hippocampus[ref. 34]. In NAcore, the combination of SPS and foot shock reduces PSD95[ref. 35]. In contrast, we found that neither 2 hours of acute restraint stress, stress-CS exposure, nor withdrawal from THC + CBD without stress pre-exposure affected the Synapsin-I density in the NAcore[ref. 7]. Thus, the reduction seen in astroglial coverage produced by stress pre-exposure (stress-NS) or THC + CBD without stress pre-exposure are largely due to astrocytic motility not an overall reduction in synapses. In contrast, we found that both Synapsin-I and astroglial synaptic proximity were reduced in the group receiving acute restraint stress and cannabinoid use (Stress-NS). Thus, the retraction of astrocytes induced by cannabinoid withdrawal, along with the loss of synapses resulting from the interplay between stress and cannabinoid use, may contribute to how THC + CBD generalizes stress responses to a neutral stimulus.
THC + CBD use induces astrocyte atrophy during exposure to the stress-CS
It is well-documented that stress alters astrocyte morphology[ref. 23]. For example, chronic restraint stress reduced the process length, branching, and volume of astrocytes in the prefrontal cortex[ref. 36], and astrocyte volume in the amygdala[ref. 37]. CUMS also induced astrocyte atrophy in the prefrontal cortex and hippocampus[ref. 38, ref. 39]. Moreover, the SPS protocol led to astrocyte atrophy in the hippocampus[ref. 40], where astrocyte atrophy is linked with anxiety and depressive-like behavior. For instance, the astrocyte morphology negatively correlates with anxiety, while treatments restoring the astrocyte morphology alleviate symptoms of anxiety and depression in animal models[ref. 39–ref. 41]. In NAcore, neither acute restraint stress nor the stress-CS affect astroglial morphology[ref. 21].
In contrast with stress, the effects of THC on astrocyte morphology are less understood. In organotypic hippocampal slices, prolonged incubation with THC reduces astrocytic processes[ref. 42]. Astrocytic processes are also reduced in the amygdala of rats treated with high doses of THC during adolescence[ref. 43]. However, the effects of voluntary cannabinoid consumption on astrocyte structure were unknown. Here, we demonstrated that THC + CBD self-administration, with or without a prior stressful experience, did not affect measures of astrocyte morphology, including surface area and volume. However, re-exposure to the stress-CS triggered astrocyte atrophy in THC + CBD, not vehicle-trained rats. Surprisingly, we observed a negative correlation between astrocyte surface area and volume and synapse coverage, indicating that cannabinoid use and stress-CS triggered both astrocyte atrophy and increased proximity of astroglia to synapses.
THC + CBD use changes the function of MMP-2,9 in regulating coping strategies in males
The gelatinases MMP-2 and MMP-9 play a necessary role in activity-dependent multipartite synaptic plasticity[ref. 16]. For example, MMP-9 controls dendritic spine enlargement induced by long-term potentiation[ref. 44]. Thus, over the last 20 years, research has focused on the role of MMP-2,9 in various psychiatric disorders, including drug and stress-related disorders[ref. 17]. Our laboratory demonstrated that drug seeking induced by Pavlovian cues requires transient activation of MMP-9 in the NAcore, which leads to both spine head expansion and the insertion of AMPA receptors on D1-medium spiny neurons[ref. 20, ref. 45, ref. 46]. In parallel, withdrawal from cocaine or heroin self-administration constitutively increases the MMP-2 activity around D2-MSNs [ref. 20, ref. 47]. Concerning stress-related disorders, both acute and chronic restraint stress increase MMP-9 activity in the hippocampus[ref. 48, ref. 49]. We also found that acute restraint stress increases MMP-9 in NAcore[ref. 19]. However, the effect of stress-CS on the activity of MMP-2,9 was unknown. Given that both stress-CS and drug cues promote spine head expansion[ref. 11, ref. 45], we hypothesized that stress-CS would increase MMP-2,9 activity. As anticipated, MMP-2,9 activity was increased after exposure to stress-CS in vehicle-trained rats. We also demonstrated that THC + CBD withdrawal constitutively increased MMP-2,9 activity. The combination of stress pre-exposure and cannabinoid withdrawal (stress-NS) did not alter the basal activity of MMP-2,9. However, re-exposure to the stress-CS during cannabinoid withdrawal increased the MMP-2,9 activity. Cannabinoid use not only elevated MMP-2,9 activity but also contributed to a shift from active to avoidant coping behaviors. Thus, while MMP-2,9 activity positively correlated with burying in vehicle-trained rats, cannabinoid use reversed the positive correlation to a negative correlation with burying and revealed a positive correlation with escape behavior.
MMP-2 is a key regulator of the multipartite synaptic changes induced by the stress-CS following cannabinoid use.
Considering that stress-CS re-exposure after cannabinoid use strongly activated MMP-2,9 and simultaneously increased astroglial coverage and astrocyte atrophy, we hypothesized that these events are interconnected. Supporting our hypothesis, MMP-2,9 activity positively correlated with astroglial coverage and negatively with astrocyte surface area. To establish a causal relationship between ECM catalytic signaling and astroglial plasticity, we administered inhibitors of both gelatinases before the DBT. We found that inhibiting either MMP-2 and MMP-9 reduced astroglial coverage of synapses in NAcore, while only the MMP-2 inhibitor reduced Synapsin-I density and prevented astrocyte atrophy. Additionally, inhibiting MMP-2 decreased grooming while increasing burying, indicating that MMP-2 activation by stress-CS contributes to the switch from active to avoidant behaviors in THC + CBD withdrawn rats. In summary, these data constitute the first in vivo evidence linking the catalytic activities of MMP-2 and MMP-9 to astroglial plasticity and stress-associated behaviors.
Sexual dimorphism
Women are twice as likely to develop PTSD and exhibit greater vulnerability for anxiety-related disorders [ref. 51–ref. 53]. However, historically, preclinical studies on stress-related disorders mostly used males and focused primarily on freezing behavior[ref. 50]. Including females and assessing other stress responses introduced new insights into coping strategies[ref. 51, ref. 52]. Indeed, research demonstrates that male rats freeze more than females, while females display higher escape-like behaviors[ref. 50, ref. 53–ref. 55]. Accordingly, we found that females escape more than males across all stress conditions. Surprisingly, neither stress nor THC + CBD had a significant impact on coping strategies in females. This may be due to high variability we observed in stress responses between females. Although the estrous cycle is not involved in escape-like behavior [ref. 42], it would be interesting to evaluate its influence on other coping responses. Despite not affecting behaviors, stress and cannabinoid use changed multipartite plasticity in females. Unlike males, the number of synapses (Synapsin-I puncta) is increased in stressed rats following THC + CBD use. Regarding astroglial plasticity, cannabinoid withdrawal without stress pre-exposure increased both the surface area and volume of NAcore astrocytes in females compared to males. This result differs from cocaine, where prolonged withdrawal elicits NAcore astrocyte atrophy in males without affecting female astrocytes[ref. 29]. Furthermore, while acute restraint stress did not affect astrocyte morphology in males, it induced astrocyte atrophy in females. The opposite effect is observed after chronic stress, where CUMS reduces astrocyte morphology in males without affecting females[ref. 56]. Our findings contribute to research on sexual dimorphism in astroglial plasticity. More studies are needed to understand how stress and cannabis differently affect female and male multipartite plasticity.
Clinically relevant targets
This study identified neurobiological mechanisms behind PTSD-like symptoms after cannabinoid use in male rats, highlighting potential therapeutic targets such as MMPs and astrocytes for comorbid CUD/PTSD. Recent studies demonstrate elevated plasma levels of MMP-2 and MMP-9 in PTSD patients[ref. 57, ref. 58]. Moreover, the nonselective MMP inhibitor doxycycline disrupts the consolidation of cued fear memory in humans[ref. 59]. Additionally, a single dose of minocycline, a more specific MMP-9 inhibitor, administered before a contextual fear memory paradigm, reduced the consolidation of fear memory[ref. 60]. These clinical data suggest that MMP inhibition represents a potentially effective strategy to treat the comorbidity between PTSD and CUD. Another interesting strategy would be to target astroglial plasticity. For example, the antidepressant fluoxetine alleviates depressive-like symptoms, partly by reversing astrocyte atrophy caused by CUMS[ref. 39]. Moreover, in humans, antidepressant treatment reverses astroglial atrophy in the hippocampus[ref. 61]. Thus, exploring the impact of antidepressant treatment on astrocyte atrophy resulting from stress-CS after cannabis use would be intriguing.
References
- 1.Trauma and stressor-related discorders. Posttraumatic stress disorder., in Diagnostic and statistical manual of mental disorders (5th Ed), A.P. Association, Editor. 2013, American Psychiatric Association.
- M.O. Bonn-Miller, M.M. Bucossi, J.A. Trafton. The underdiagnosis of cannabis use disorders and other Axis-I disorders among military veterans within VHA.. Mil Med,, 2012. [PubMed]
- M.O. Bonn-Miller. The impact of posttraumatic stress disorder on cannabis quit success.. Am J Drug Alcohol Abuse,, 2015. [PubMed]
- M. Loflin, M. Earleywine, M. Bonn-Miller. Medicinal versus recreational cannabis use: Patterns of cannabis use, alcohol use, and cued-arousal among veterans who screen positive for PTSD.. Addict Behav,, 2017. [PubMed]
- N.A. Livingston. Longitudinal course of mental health symptoms among veterans with and without cannabis use disorder.. Psychol Addict Behav,, 2022. [PubMed]
- J. Metrik. Cannabis use and posttraumatic stress disorder: prospective evidence from a longitudinal study of veterans.. Psychol Med,, 2022. [PubMed]
- A. Murkar. Increased cannabis intake during the COVID-19 pandemic is associated with worsening of depression symptoms in people with PTSD.. BMC Psychiatry,, 2022. [PubMed]
- V. Kondev, N. Winters, S. Patel. Cannabis use and posttraumatic stress disorder comorbidity: Epidemiology, biology and the potential for novel treatment approaches.. Int Rev Neurobiol,, 2021. [PubMed]
- D.S. Hasin. US Epidemiology of Cannabis Use and Associated Problems.. Neuropsychopharmacology,, 2018. [PubMed]
- 10.PTSD and Medical Cannabis Programs. 2025 01/02/2025; Available from: https://www.mpp.org/issues/medical-marijuana/ptsd-medical-cannabis-programs/.
- R. Hodebourg. Cannabinoid use is enhanced by stress and changes conditioned stress responses.. Neuropsychopharmacology,, 2022. [PubMed]
- J.S. Carter, A.M. Kearns, C.M. Reichel. Complex Interactions Between Sex and Stress on Heroin Seeking.. Front Neurosci,, 2021. [PubMed]
- J.S. Carter. Long-term impact of acute restraint stress on heroin self-administration, reinstatement, and stress reactivity.. Psychopharmacology (Berl),, 2020. [PubMed]
- C. Garcia-Keller. N-Acetylcysteine treatment during acute stress prevents stress-induced augmentation of addictive drug use and relapse.. Addict Biol,, 2020. [PubMed]
- A. Dityatev, D.A. Rusakov. Molecular signals of plasticity at the tetrapartite synapse.. Current Opinion in Neurobiology,, 2011. [PubMed]
- R. Hodebourg. Nonneuronal contributions to synaptic function.. Neuron,, 2025
- R. Hodebourg, P.W. Kalivas, A. Kruyer. Extrasynaptic therapeutic targets in substance use and stress disorders.. Trends Pharmacol Sci,, 2022. [PubMed]
- S. Spencer. A Model of Delta(9)-Tetrahydrocannabinol Self-administration and Reinstatement That Alters Synaptic Plasticity in Nucleus Accumbens.. Biol Psychiatry,, 2018. [PubMed]
- R. Hodebourg. In Vivo Zymography: An Essential Activity Assay for Studying the Activity of Matrix Metalloproteinases in Specific Brain Cells.. Methods Mol Biol,, 2025. [PubMed]
- V.C. Chioma. Heroin Seeking and Extinction From Seeking Activate Matrix Metalloproteinases at Synapses on Distinct Subpopulations of Accumbens Cells.. Biol Psychiatry,, 2020
- C. Garcia-Keller. Behavioral and accumbens synaptic plasticity induced by cues associated with restraint stress.. Neuropsychopharmacology,, 2021
- X. Wang. High-frequency repetitive transcranial magnetic stimulation improves depressive-like behaviors in CUMS-induced rats by modulating astrocyte GLT-1 to reduce glutamate toxicity.. Journal of Affective Disorders,, 2024. [PubMed]
- Y. Bansal, S.A. Codeluppi, M. Banasr. Astroglial Dysfunctions in Mood Disorders and Rodent Stress Models: Consequences on Behavior and Potential as Treatment Target.. Int J Mol Sci,, 2024
- A. Beroun. MMPs in learning and memory and neuropsychiatric disorders.. Cell Mol Life Sci,, 2019. [PubMed]
- U. Bonnet, U.W. Preuss. The cannabis withdrawal syndrome: current insights.. Subst Abuse Rehabil,, 2017. [PubMed]
- J.P. Connor. Clinical management of cannabis withdrawal.. Addiction,, 2022. [PubMed]
- K.R. Trexler. Novel behavioral assays of spontaneous and precipitated THC withdrawal in mice.. Drug Alcohol Depend,, 2018. [PubMed]
- J.M. Koolhaas. Coping styles in animals: current status in behavior and stress-physiology.. Neuroscience & Biobehavioral Reviews,, 1999. [PubMed]
- R. Kim. Abstinence-Dependent Effects of Long-Access Cocaine Self-Administration on Nucleus Accumbens Astrocytes Are Observed in Male, But Not Female, Rats.. eNeuro,, 2022
- A. Kruyer. Heroin Cue-Evoked Astrocytic Structural Plasticity at Nucleus Accumbens Synapses Inhibits Heroin Seeking.. Biol Psychiatry,, 2019. [PubMed]
- M.D. Scofield. Cocaine Self-Administration and Extinction Leads to Reduced Glial Fibrillary Acidic Protein Expression and Morphometric Features of Astrocytes in the Nucleus Accumbens Core.. Biological Psychiatry,, 2016. [PubMed]
- B.M. Siemsen. Effects of Methamphetamine Self-Administration and Extinction on Astrocyte Structure and Function in the Nucleus Accumbens Core.. Neuroscience,, 2019. [PubMed]
- B. Ning. The rapid antidepressant effect of acupuncture on two animal models of depression by inhibiting M1-Ach receptors regulates synaptic plasticity in the prefrontal cortex.. Brain Res,, 2024. [PubMed]
- Y.-Y. Peng. Deletion of p75NTR rescues behavioral and cognitive dysfunction in SPS-induced PTSD mice through hippocampal PI3K/Akt/mTOR pathway.. International Journal of Biological Macromolecules,, 2025. [PubMed]
- L. Gou. (2R,6R)-hydroxynorketamine improves PTSD-associated behaviors and structural plasticity via modulating BDNF-mTOR signaling in the nucleus accumbens.. Journal of Affective Disorders,, 2023. [PubMed]
- R.J. Tynan. Chronic stress-induced disruption of the astrocyte network is driven by structural atrophy and not loss of astrocytes.. Acta Neuropathol,, 2013. [PubMed]
- S. Naskar, S. Chattarji. Stress Elicits Contrasting Effects on the Structure and Number of Astrocytes in the Amygdala versus Hippocampus.. eneuro,, 2019
- S. Aten. Chronic Stress Impairs the Structure and Function of Astrocyte Networks in an Animal Model of Depression.. Neurochem Res,, 2023. [PubMed]
- S.S. Lin. Electroacupuncture prevents astrocyte atrophy to alleviate depression.. Cell Death Dis,, 2023. [PubMed]
- L. Xia. FGF2 blocks PTSD symptoms via an astrocyte-based mechanism.. Behavioural Brain Research,, 2013. [PubMed]
- J.S. Soto. Astrocyte Gi-GPCR signaling corrects compulsive-like grooming and anxiety-related behaviors in Sapap3 knockout mice.. Neuron,, 2024. [PubMed]
- E. Landucci. Neuronal and Astrocytic Morphological Alterations Driven by Prolonged Exposure with Δ9-Tetrahydrocannabinol but Not Cannabidiol.. Toxics,, 2022
- J.-M.N. Ferland. Dose mediates the protracted effects of adolescent THC exposure on reward and stress reactivity in males relevant to perturbation of the basolateral amygdala transcriptome.. Molecular Psychiatry,, 2022
- X. b. Wang. Extracellular proteolysis by matrix metalloproteinase-9 drives dendritic spine enlargement and long-term potentiation coordinately.. Proceedings of the National Academy of Sciences,, 2008
- C. Garcia-Keller. Extracellular Matrix Signaling Through beta3 Integrin Mediates Cocaine Cue-Induced Transient Synaptic Plasticity and Relapse.. Biol Psychiatry,, 2019. [PubMed]
- C. Garcia-Keller. Relapse-Associated Transient Synaptic Potentiation Requires Integrin-Mediated Activation of Focal Adhesion Kinase and Cofilin in D1-Expressing Neurons.. J Neurosci,, 2020. [PubMed]
- A.C. Smith. Synaptic plasticity mediating cocaine relapse requires matrix metalloproteinases.. Nat Neurosci,, 2014. [PubMed]
- F.I. Aguayo. Matrix Metalloproteinase 9 Displays a Particular Time Response to Acute Stress: Variation in Its Levels and Activity Distribution in Rat Hippocampus.. ACS Chem Neurosci,, 2018. [PubMed]
- M.A. van der Kooij. Role for MMP-9 in stress-induced downregulation of nectin-3 in hippocampal CA1 and associated behavioural alterations.. Nat Commun,, 2014. [PubMed]
- J.R. Mitchell. Behavioral and neural correlates of diverse conditioned fear responses in male and female rats.. Neurobiol Stress,, 2024. [PubMed]
- A. Chu. A fear conditioned cue orchestrates a suite of behaviors in rats.. Elife,, 2024
- D. Dopfel. Individual variability in behavior and functional networks predicts vulnerability using an animal model of PTSD.. Nat Commun,, 2019. [PubMed]
- T.M. Gruene. Sexually divergent expression of active and passive conditioned fear responses in rats.. Elife,, 2015
- L. Colon. Sexual differentiation of contextual fear responses.. Learn Mem,, 2018. [PubMed]
- P.R. Zambetti, B.P. Schuessler, J.J. Kim. Sex Differences in Foraging Rats to Naturalistic Aerial Predator Stimuli.. iScience,, 2019. [PubMed]
- S.C. Woodburn, J.L. Bollinger, E.S. Wohleb. Synaptic and behavioral effects of chronic stress are linked to dynamic and sex-specific changes in microglia function and astrocyte dystrophy.. Neurobiol Stress,, 2021. [PubMed]
- M.V. Brahmajothi, M.B. Abou-Donia. PTSD Susceptibility and Challenges: Pathophysiological Consequences of Behavioral Symptoms.. Mil Med,, 2020. [PubMed]
- L. Wu, M. Jin. Clinical Significance of Serum MMP-9, S100-β and GFAP in Patients with Mental Disorders after Traumatic Brain Injury.. Actas Esp Psiquiatr,, 2025. [PubMed]
- D.R. Bach, A. Tzovara, J. Vunder. Blocking human fear memory with the matrix metalloproteinase inhibitor doxycycline.. Mol Psychiatry,, 2018. [PubMed]
- Y. Xia. Attenuating human fear memory retention with minocycline: a randomized placebo-controlled trial.. Transl Psychiatry,, 2024. [PubMed]
- J.A. Cobb. Density of GFAP-immunoreactive astrocytes is decreased in left hippocampi in major depressive disorder.. Neuroscience,, 2016. [PubMed]
