Metal Ion Signaling in Biomedicine
Abstract
Complex multicellular organisms are composed of distinct tissues involving specialized cells that can perform specific functions, making such life forms possible. Species are defined by their genomes, and differences between individuals within a given species directly result from variations in their genetic codes. While genetic alterations can give rise to disease-causing acquisitions of distinct cell identities, it is now well-established that biochemical imbalances within a cell can also lead to cellular dysfunction and diseases. Specifically, nongenetic chemical events orchestrate cell metabolism and transcriptional programs that govern functional cell identity. Thus, imbalances in cell signaling, which broadly defines the conversion of extracellular signals into intracellular biochemical changes, can also contribute to the acquisition of diseased cell states. Metal ions exhibit unique chemical properties that can be exploited by the cell. For instance, metal ions maintain the ionic balance within the cell, coordinate amino acid residues or nucleobases altering folding and function of biomolecules, or directly catalyze specific chemical reactions. Thus, metals are essential cell signaling effectors in normal physiology and disease. Deciphering metal ion signaling is a challenging endeavor that can illuminate pathways to be targeted for therapeutic intervention. Here, we review key cellular processes where metal ions play essential roles and describe how targeting metal ion signaling pathways has been instrumental to dissecting the biochemistry of the cell and how this has led to the development of effective therapeutic strategies.
Affiliations: †Institut Curie, CNRS, INSERM, PSL Research University, 75005 Paris, France; ‡Université Paris-Saclay, UVSQ, 78180 Montigny-le-Bretonneux, France
License: © 2025 The Authors. Published by American Chemical Society CC BY 4.0 Permits the broadest form of re-use including for commercial purposes, provided that author attribution and integrity are maintained (https://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1021/acs.chemrev.4c00577 | PubMed: 39746035 | PMC: PMC11758815
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (22.7 MB)
Introduction
In 1941, Beadle and Tatum reported the discovery that genes control biochemical events in the cell.1 Shortly thereafter, Pauling and co-workers elucidated the molecular basis of sickle cell anemia2 (Figure ). Back then, it was anticipated that mutations in genomic DNA could lead to disease-causing dysfunctional gene products from which emerged the concept of “molecular disease” and the science of “molecular medicine” (Figure ). These discoveries were made without prior knowledge of the double helical structure of DNA, which was reported only a few years later by Franklin, Crick, Watson, and others3,4 (Figure ). Seventy years later, molecular editing of this genetic defect using CRISPR/Cas technology corrected sickle cell anemia in vivo5−8 (Figure ). In 1962, Gurdon demonstrated that terminally differentiated cell nuclei can give rise to healthy organisms using nuclear transplantation, which illustrated that cell differentiation, acquisition of a specific phenotype by a cell, does not necessarily involve gene restriction9 (Figure ). Almost half a century later, Yamanaka identified the key transcription factors (TFs) essential to produce pluripotent stem cells from adult fibroblasts10 (Figure ). These studies represent a real conceptual rupture, illuminating that cells can adopt different states or identities independently of genetic alterations, a biological process broadly termed cell plasticity or cell-state transitioning.
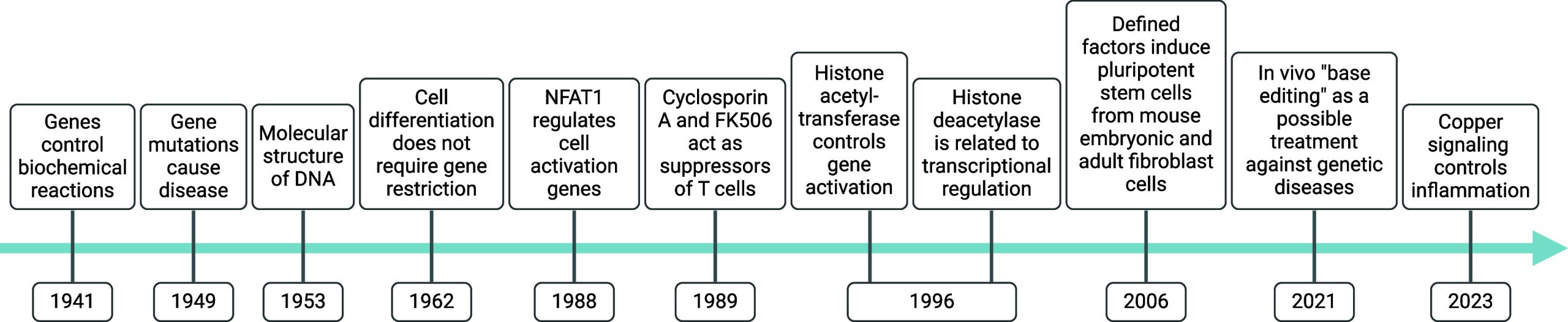
This challenges the view that cancer cells acquire resistance to therapy exclusively through genetic mutations. Indeed, cancer cells can exploit mechanisms similar to that found in development,11 allowing them to shift between highly proliferative to less proliferative yet more invasive states, to become refractory to current standard-of-care therapies, including antiproliferative drugs, and to promote cancer metastases.12 While gene mutations contribute to tumorigenesis, the capacity of a cell to adapt quickly to its environment and to adopt distinct states independently of genetic alterations drives drug tolerance. Similarly, immune cells can rapidly respond to the presence of pathogens and activate the appropriate clearance mechanisms by adopting distinct phenotypes, for instance, that of inflammatory macrophages.13 The acquisition of distinct cell states marks many biological and pathophysiological processes other than cancer, such as wound healing and inflammation. To this end, cells have evolved molecular mechanisms allowing rapid and reversible adaptation, which involves the capacity of a cell to convert extracellular physical, chemical, or biochemical signals into metabolic and/or transcriptional changes. In 1996, Allis and Schreiber independently reported the discovery of histone acetyltransferases (HATs) and histone deacetylases (HDACs), respectively, two distinct classes of enzymes, which through chemical modification of histone proteins impact DNA-related processes including gene expression14,15 (Figure ). These findings led to the concept that chromatin acts as a signaling platform rather than a mere structural element required for DNA compaction, and that different chromatin states shape transcriptional profiles and cell identity.16,17 Understanding the molecular basis underlying the control of cell states is of paramount importance, as this enables the development of therapeutic strategies to correct potential biochemical imbalances. Cell signaling encompasses the entire biochemical chain of events transducing external cellular stimuli to metabolic and transcriptional changes, enabling cells to acquire distinct physical, chemical, and biological properties.
Often considered to be trace elements, metals play central roles in cell signaling by balancing negative charges, by enabling cation exchange/transport (e.g., proton, sodium, potassium), as inducers of protein and nucleic acid folding, promoting or inactivating functions by metalloallostery, or as catalysts of chemical reactions (e.g., iron, copper) (Figure ). Here, we broadly define the action of metal ions in promoting signal transduction within a cell as “metal ion signaling”. The capacity of a given metal to promote specific biochemical processes within a cell is inherently linked to its position in the periodic table, that is, its intrinsic electron configuration (Figure a). Early on, Tsien recognized the critical role of calcium in developmental biology, identifying rapid increase of intracellular calcium levels in Xenopus laevis embryos upon T cell activation and during the cell cycle.18−21 It was in the late 1980s that Schreiber and Crabtree truly pioneered the field of cell signaling with the discovery of the first membrane-to-nucleus cell-signaling pathway involving metal ions and showing that calcium promotes specific transcriptional programs in T cells22−26 (Figure ). Metals can readily coordinate protein and nucleic acid residues, inducing conformational changes, thereby enabling activity. Other metals susceptible to readily accept (e.g., redox activity, Lewis acid catalysis) or release electrons can, on the contrary, directly catalyze specific chemical reactions, defining these metals as “enabling catalysts” as opposed to “facilitating cofactors”.27 For example, copper can catalyze the activation of hydrogen peroxide and oxidation of nicotinamide adenine dinucleotide hydride (NADH), which promotes metabolic and epigenetic programming of inflammatory macrophages13 (Figure ). Notably, it was shown that the content of specific metals varies in immune and cancer cells undergoing cell-state transitions and that some of these metals orchestrate the acquisition of a distinct cell identity13 (Figure b,c).
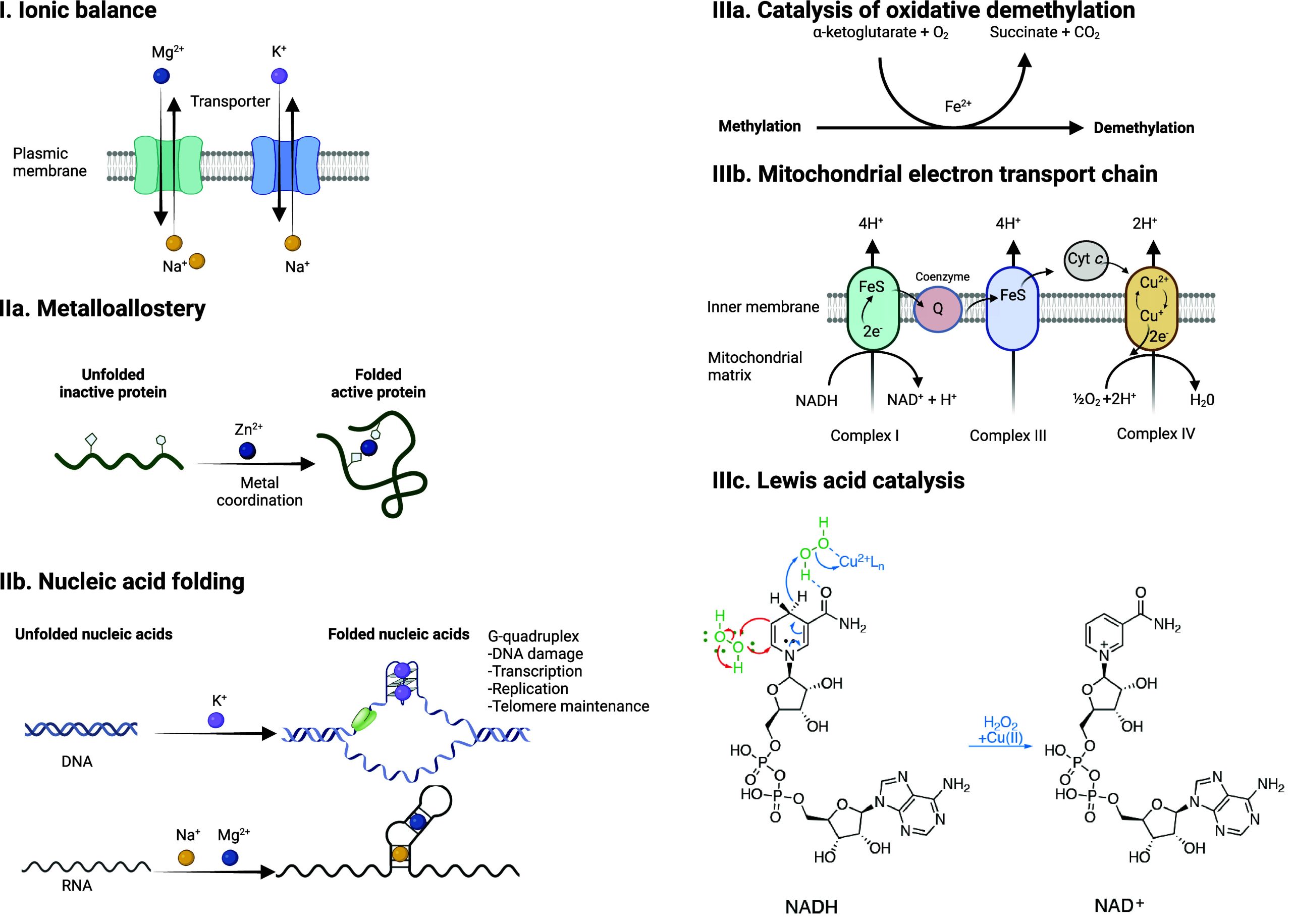
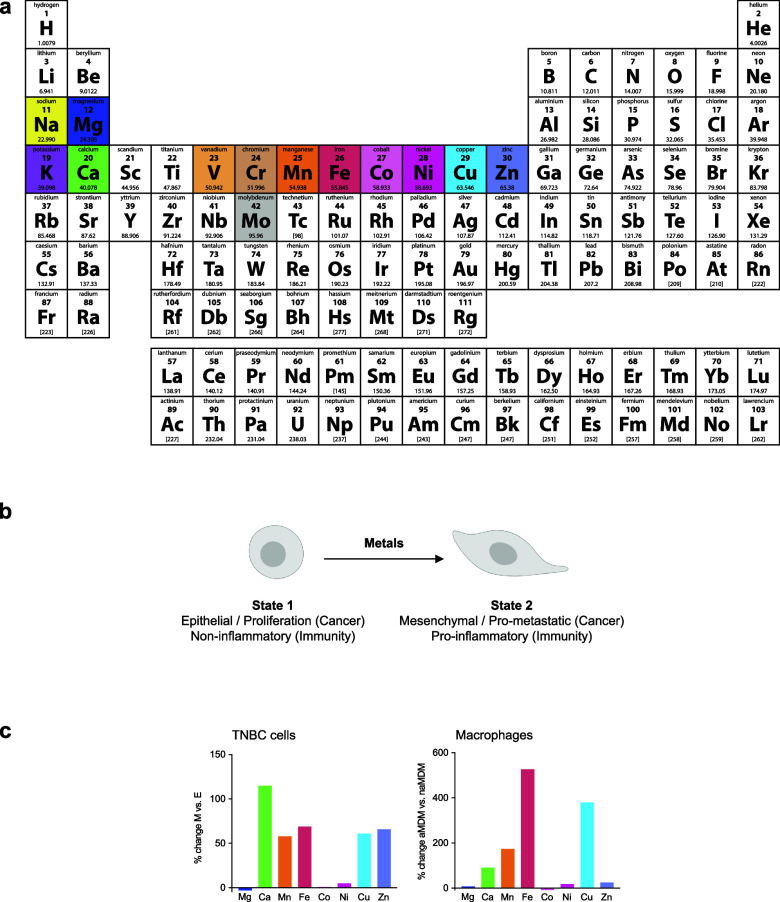
The field of bioinorganic cell biology has gained momentum with the development of new techniques to investigate metal contents of a cell, their subcellular localizations, and oxidation states. These include organelle-selective and oxidation state-specific reporters coupled to high-resolution fluorescence microscopy,28−32 quantitative inductively coupled plasma-mass spectrometry (ICP-MS)33 and nanoscale secondary ion mass spectrometry-based imaging.34 Cell signaling can go awry and cause disease. Thus, understanding the intricacies of cellular metal ion homeostasis is critical for the development of drugs to rebalance these processes. Biologically active small molecules have been instrumental in dissecting and manipulating the biochemistry of the cell, providing the means to identify druggable targets and to develop new medicines.35,36 While TFs, kinases, and other classes of proteins such as chromatin readers have long been thought to be undruggable by small molecules, phenotype-based drug target identification together with structure-based rational design have proven to be powerful approaches to identify previously uncharted targets and to elucidate mechanisms of action (MoA) and cell signaling cascades.37−43 While manipulating metal ion homeostasis with chelators and ionophores can confer therapeutic benefits44 in diseases characterized by a toxic metal ion imbalance, including myelodysplastic syndrome, β-thalassemia, Menkes disease (MD), and Wilson’s disease (WD), fine-tuning metal ion signaling is until now less common and the rational design of metal targeting compounds remains a challenging endeavor. Nevertheless, one may adopt the optimistic view previously crafted by the Nobel Peace Prize laureate Nelson Mandela and adapted here in the context of metal ion signaling: “It is undruggable until someone drugs it”.
In this review, we discuss the roles of metal ions in the biology of the cell and document how cells take up, distribute, store, and export metals. Then, we provide details as to how metals are exploited for their charge balance effects, information storage (supramolecular properties),45 and chemical reactivity within the cell (Figure ). Then we detail how these properties fuel signal transduction within the cell. Finally, we describe examples of metal ion signaling networks that have been targeted with therapeutic value, exploiting metals as direct targets or alternatively drugging other biomolecules upstream or downstream of key metal ion signaling cascades. While we do not provide an exhaustive list of biologically active small molecules targeting metal ion signaling, we have listed key examples of biologically active chemical substances impacting metal ion signaling in Tables 1–13, illustrating the power of chemistry to dissect and manipulate biological processes for basic research purposes and biomedicine.
Alkali Metal Ion Signaling
Regulation of Sodium and Potassium Homeostasis
Sodium and potassium are abundant in cellular systems. Both alkali metals are closely linked in their cellular regulation and function. For instance, some transporters are coupled antiporters or symporters of both metals as depicted Figure . Sodium is imported into cells through some cellular metal ion transporters coupled to various ion channels, including the voltage-gated sodium channel (VGSC), the epithelial sodium channel (ENaC), the acid-sensing ion channel (ASIC), the N-methyl-d-aspartate receptor (NMDAR), the purinergic P2X receptor 7 (P2X7R), and the sodium leak channel nonselective (NALCN), which are differentially expressed in distinct tissues46,47 and of which ENaC is the most well-characterized to date48 (Figure ). Ion channels are membrane proteins that form pores and allow ions to pass through. Sodium can also be taken up by transporters, which are proteins that pump ions via a membrane, often using energy, for instance, from the hydrolysis of adenosine triphosphate (ATP). Symporters transport different types of ions in the same direction, whereas antiporters transport different types of ions in opposite directions over a lipid membrane. Various antiporters have been documented, which include the sodium/calcium exchanger NCX and sodium/proton exchanger NHE1. Various symporters that import sodium have also been described, including amino acid symporters, the sodium/bicarbonate symporter NCBn1, NKCC, which imports sodium, potassium, and chloride, the sodium/glucose symporter SGLT, and the sodium/iodide symporter NIS.49,50 Other members of the NHE family are involved in the transport of sodium across membranes of different cell organelles, including NHE7 and NHE8 in the membrane of the Golgi apparatus and NHE in the membrane of lysosomes. NHE is also situated in the membrane of mitochondria, and mitochondrial sodium/calcium exchanger NCLX regulates mitochondrial sodium import and calcium export. Cellular export of sodium is coupled to potassium with the sodium/potassium antiporter, linking the cellular levels of these two alkali metals together. Other cation channels that transport sodium out of lysosomes have been identified, namely, two-pore channel (TPC) and transient receptor potential mucolipin (TRPML). Potassium is one of the most abundant cations in the extracellular matrix (ECM) and regulation of metal homeostasis between the ECM and the cell is essential to maintain cellular functions.51,52 Potassium can be imported into cells by the sodium/potassium ATPase53,54 or the symporter NKCC, which imports potassium, sodium, and chloride into cells46 (Figure ). Potassium export from cells on the contrary is mediated by potassium channels, including Kv1.3, Kv10.1, and Kv11.1 and calcium-activated potassium channel K.Ca3.1. Potassium transport into mitochondria is also regulated by potassium channels, including mitoKv1.3, mitoKATP, and Kir, whereas the potassium/proton exchanger (KHE) mediates export of potassium from this organelle. The transmembrane protein 175 (TMEM175) has been described as being able to import and export potassium from and into lysosomes. However, it was later reported to act as a proton-activated proton channel. Thus, its contribution to lysosomal potassium homeostasis should be revisited.55−57 In addition, voltage-gated potassium channels, such as Kv2, mediate potassium import into the endoplasmic reticulum (ER).
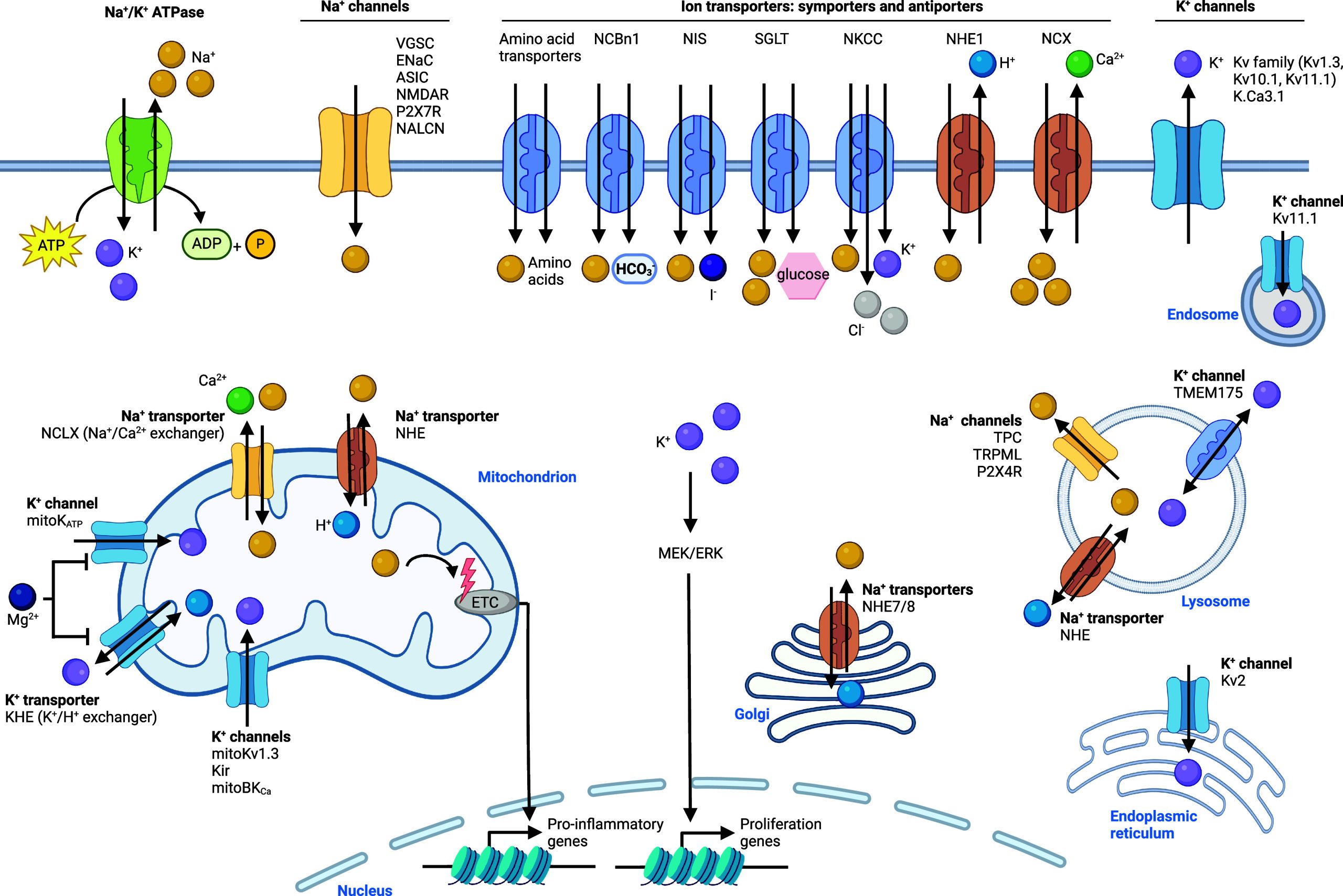
Cellular Functions of Sodium and Potassium
The presence of the NCX antiporter at the cell membrane makes sodium a regulator of intracellular calcium levels. In addition, the sodium/proton antiporter NHE1 plays a crucial role in the regulation of cellular pH, and thus sodium can act as a regulator of intracellular pH.58 Importantly, proteins of the NHE family are also integral members of the mitochondrial membrane and responsible for the generation of a proton gradient, which involves pumping protons against their electrochemical gradient across the inner mitochondrial membrane. This process is essential for the production of ATP in eukaryotic cells, and thus, sodium also plays a pivotal role in energy generation via oxidative phosphorylation in the electron transport chain (ETC).59 High sodium levels and redistribution of sodium in mitochondria impact the ETC, which leads to expression of inflammatory genes in immune cells.60 Thus, sodium is a key regulator of cell metabolism. Sodium also acts as an allosteric regulator of protein function, as exemplified by the opioid receptors, which are a type of G protein-coupled receptor (GPCR), whose folding and signaling activity are influenced by sodium.61 Furthermore, sodium, potassium, and magnesium cations impact folding of RNAs and resulting functions.62 In particular, alkali metals facilitate the folding of G-quadruplex (G4) DNAs and RNAs.63−65 G4 are believed to be involved in a plethora of cellular functions and biological effects,65−68 including gene transcription and translation, DNA replication, genome instability, and telomere maintenance.69,70 With its unique structure, chromatin operates as a complex signaling platform.17 In this context, G4 and other nucleic acid structures represent cell signaling elements. Therefore, alkali metals directly impact cell signaling, affecting the stability and integrity of the nucleic acid structures. In nerve cells, sodium plays a crucial role for the transmission of action potential, where rapid sodium influx and potassium efflux generate potentials across cell membranes.71 Sodium influx into astrocytes is mainly regulated by glutamate transporters, whereas efflux is mediated by the sodium/potassium ATPase.53,54 Mitochondrial potassium channels also exhibit a cytoprotective function, although the underlying signaling pathways are not clear and require further investigation.72 In addition, voltage-gated potassium channels influence the capacity of the plasma membrane to regulate potassium concentrations and thus cell volume via osmotic pressure.73 This can affect cell cycle progression, cell proliferation, and ultimately cell death mechanisms including apoptosis.74,75 Furthermore, potassium has been shown to activate the mitogen-activated protein kinase kinase/extracellular-signal-regulated kinase (MEK/ERK) pathway, impacting the expression of genes implicated in cell proliferation.76−78 Using DNA nanodevices that can detect sodium79 or potassium80 with subcellular resolution, it was recently shown that sodium and potassium gradients exist across cell membranes. This work led to the discovery that Kv11.1 is an endosomal potassium channel.
Sodium and Potassium Signaling and Diseases
Alteration of sodium homeostasis has been reported in various pathophysiological contexts including cancer, ischemia-reperfusion injury, and cardiovascular diseases.81,82 Hyper- or hypokalemia can arise in patients where potassium homeostasis is perturbed at the cellular and/or organismal levels.83 Hyperkalemia can lead to thrombocytosis, hemolysis, and high white cell counts, whereas hypokalemia can cause muscle weakness and cramps and lead to arrhythmia.
Sodium and Potassium Signaling in Cancer
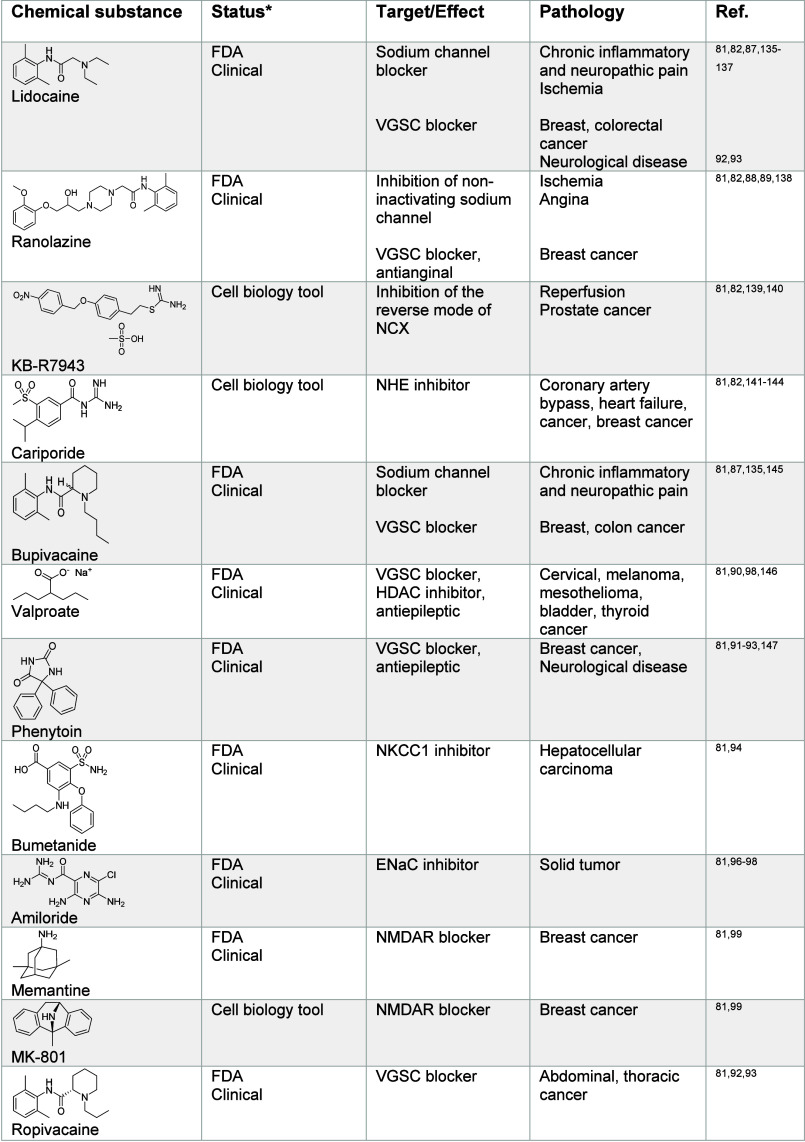
Solid tumors have been shown to contain increased levels of sodium. It has been proposed that sodium ions control osmolarity in the cancer microenvironment, potentially impacting cell metabolism and immune function.81 Since sodium and ATP production are coupled via the mitochondrial membrane potential, these changes in sodium levels go hand in hand with metabolic alterations. This can directly impact signaling pathways that require phosphorylation events, including Kirsten rat sarcoma virus (K-ras) and mitogen-activated protein kinase (MAPK) signaling.84 In addition, since sodium transport is regulated by various antiporters, any changes in sodium homeostasis also impact calcium, glucose, and magnesium levels as well as pH, affecting other cellular processes.50 Increased levels of VGSC channels have been observed in breast cancer cells with higher metastatic potential and in breast cancer metastases.85,86 This suggests that increased sodium levels in cancer cells are correlated with the acquisition of the metastatic phenotype. Tetrodotoxin (Table 1) is a toxic natural product found in pufferfish. It has been shown to inhibit VGSC channels and to impact cancer cell growth.85,87 Ranolazine, another VGSC inhibitor, has also been shown to be effective against metastatic breast cancer.88,89 The VGSC inhibitor valproate has been selected for the treatment of cervical cancer.90 The VGSC inhibitor phenytoin91 inhibits breast cancer growth in preclinical models, and other inhibitors against these classes of sodium channels are under development for the treatment of cancer, including ropivacaine,81,92,93 carbamazepine,87,92,93 lamotrigine,92,93 fosphenytoin,92,93 and others (Table 1). These examples highlight that targeting voltage-gated sodium channels may be exploited for the treatment of various cancers, including metastatic disease. Additional research is required to dissect the effect of VGSC on cell signaling in metastatic disease, specifically to better characterize the role of sodium in the acquisition of distinct states of cancer cells. This may illuminate drug effects and reveal how to more effectively exploit sodium signaling in clinical settings. Another class of sodium transporters, NKCC1, has been shown to be upregulated in metastatic hepatocellular carcinoma (HCC),94 and the inhibitor bumetanide has been demonstrated to impact tumor growth and metastases in preclinical models of HCC.81,94 The sodium-dependent glucose transporters SGLT enable the cellular uptake of glucose, representing an alternative mechanism to the canonical pathway implicating glucose transporters (GLUTs). SGLT transporters are highly expressed in pancreatic and prostate adenocarcinomas, and SGLT2 inhibitors such as canagliflozin (Table 1), which is clinically approved for the treatment of diabetes, showed reduction of pancreatic cancer in xenograft models.95 Amiloride is a blocker of ENaC channels and has been shown to inhibit growth of solid tumors.81,96−98 Additionally, NMDAR blockers including memantine and MK-801 have been shown to reduce growth of breast cancer in human xenografts.81,99 Together, these studies on sodium receptors illustrate how cellular homeostasis of alkali metals is dysregulated in cancer and how targeting specific channels or transporters can be exploited for therapeutic benefits. Small molecules provide the means to dissect metal ion signaling in great detail. Understanding how sodium impacts cancer cell fate and contributes to metastasis and drug-tolerance requires further investigation.
Table 1: Regulators of Sodium Signaling81,82,85,87−89,91−99,135−154
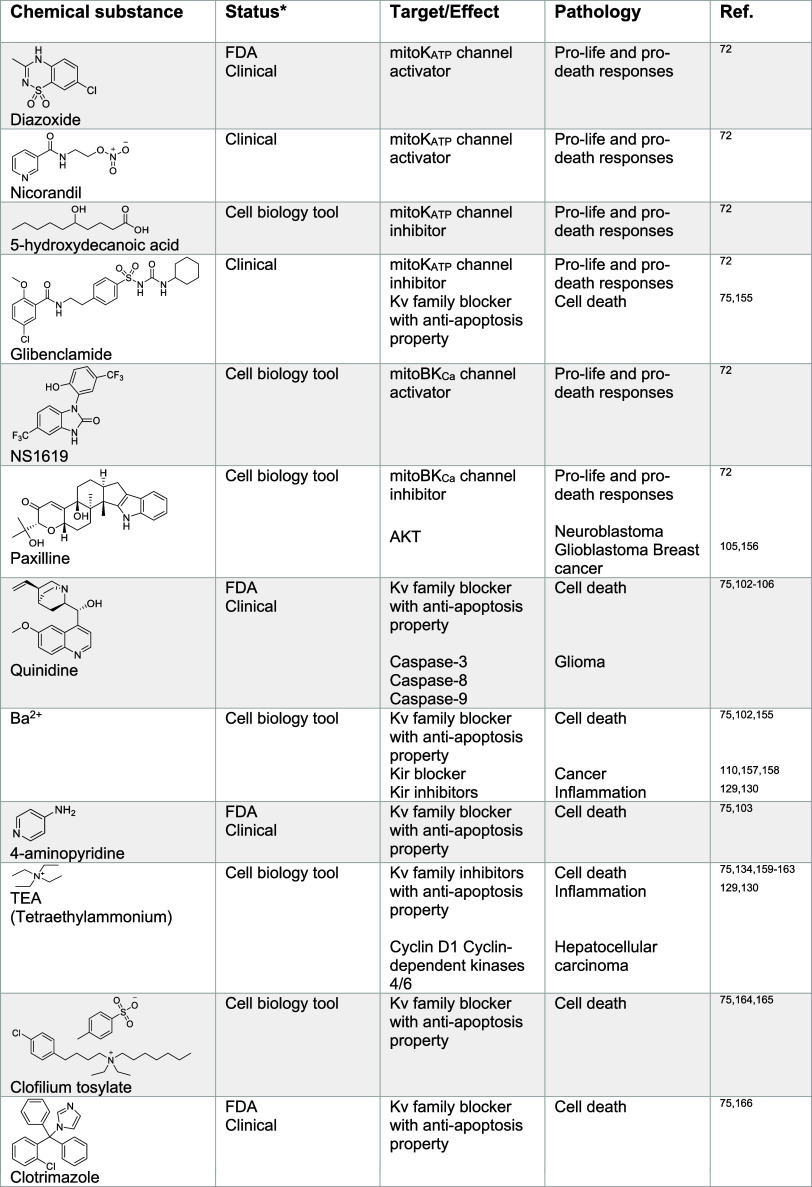
Potassium has been reported to be overabundant in the tumor microenvironment.100 It was argued that elevated potassium concentrations lead to perturbations in the electrochemical gradient required for the uptake of nutrients in T cells. This can lead to a reduction in histone acetylation at genes required for T cell effector function. This, in turn, was found to improve T cell multipotency and tumor clearance capacity. In another study, it was shown that distinct cancer types exhibit a reduced expression of the potassium channel Kv1.5 at the cell membrane.101 These cells could be sensitized to apoptosis by dichloroacetate, which causes an increased level of expression of Kv1.5. This promoted hyperpolarization of the cell and an inhibition of voltage-dependent entry of calcium, causing decreased glycolysis and increased mitochondrial respiration. Potassium channels have garnered a great deal of interest in cancer research because a dysregulation of these channels has been observed in many cancers, supporting a causal role of potassium in cancer progression, suggesting that targeting potassium signaling, or more broadly cellular potassium homeostasis, can be exploited for therapeutic intervention. For instance, small molecules such as quinidine and quinine (Table 2), block potassium channels with pro-apoptotic properties in glioma cells.75,102−106 TRAM-34 targets calcium-activated potassium channels and is under development for the treatment of glioblastoma.107−109 Dequalinium and amiodarone target potassium channels in breast cancer cells105,110−112 and mitochondrial Kv1.3 channel inhibitors like clofazimine or margatoxin represent promising therapeutic candidates for the treatment of lymphoma and lung adenocarcinoma.110,113,114 In addition, the Kv and Kir family blocker cisapride is in development for the treatment of gastric cancer.110,115 The antibiotic erythromycin also inhibits these channels110,116,117 and is under investigation for the treatment of cancer, autoimmune, and neurodegenerative disorders (for a list of sodium and potassium channel inhibitors, see Table 2). These examples highlight how targeting potassium channels and related signaling networks could provide powerful therapeutic opportunities in cancer. Furthermore, G4 structures, stabilized by potassium in the inner cavity composed of guanine residues are atypical chromatin targets that may provide the basis for drug design.118,119 For instance, the small molecule pyridostatin (Table 2) has been shown to target G4 in gene bodies and at telomeres, leading to transcription and replication-dependent DNA damage, activating a DNA damage signaling response and apoptosis in cancer cells.69,120,121 Pyridostatin was used in many laboratory settings122−124 to challenge the existence and dissect the biology of G4 nucleic acids in the cell, lending strong support to the idea that such structures, whose dynamic folding relies on alkali metals, can be exploited beyond academic research.
Table 2: Regulators of Potassium Signaling72,75,102−117,120,121,129−134,155−180
Sodium and Potassium Signaling in Immunity and Inflammation
The role of sodium in immune cell activation is complex. Concentrations of sodium chloride were found to be elevated in human and mouse skin infections. In a model of bacterial skin infection by Leishmania major, high levels of sodium chloride were found to lead to (p38/MAPK)-dependent nuclear factor of activated T cells 5 (NFAT5) signaling activation and epigenetic alterations in inflammatory macrophages.125 Explicit roles of sodium in this context remain incompletely understood and require further efforts. ERK1 and ERK2 activation was also observed in inflammatory macrophages under high sodium level conditions.126 In contrast, acquisition of the anti-inflammatory state of macrophages was inhibited under high sodium concentrations, reducing serine/threonine protein kinase (AKT) and mammalian target of rapamycin (mTOR) signaling.127 Given the complexity of mechanisms underlying activation of macrophages in vivo128 and that macrophage populations can shift between states, the contributions of sodium on macrophage plasticity in vivo requires additional investigation. Potassium has also been documented to play a role in inflammation since blocking potassium channels has been shown to attenuate inflammation.129−134 The role of potassium channels in inflammatory settings suggests that targeting key channels with barium ions, tetraethylammonium, iberiotoxin, as well as the FDA-approved drug quinine129−134 (Table 2) may be exploited in clinical settings to control immune responses and reduce inflammation.
Alkaline Earth Metal Ion Signaling
Magnesium Signaling
Regulation of Magnesium Homeostasis
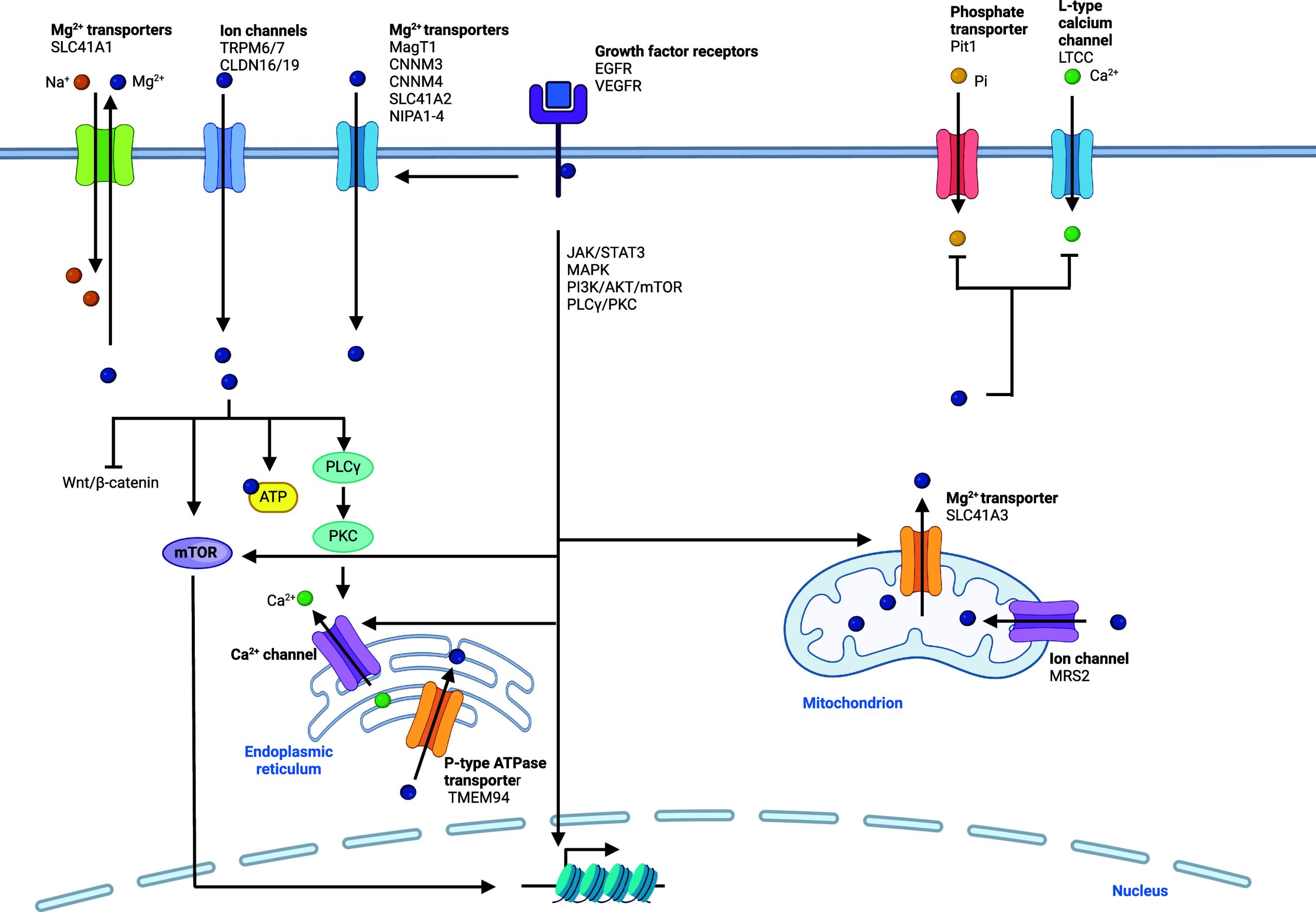
In the cell, magnesium is found as a +2 oxidation state, as both a free and bound metal ion, and it is the second most abundant intracellular metal after potassium. Magnesium is imported into the cell through ion channels and transporters. Notably, magnesium channels and transporters include transient receptor potential melastatin 6/7 (TRPM6/7)181,182 and the magnesium transporter MagT1183 (Figure ). Since TRPM7 also enables transport of zinc and calcium, strategies designed to target these receptors potentially affect intracellular levels of other metals.184 SLC41A2 is also a cell membrane specific magnesium transporter,185 and a proton/magnesium exchanger was found to play a major role in cellular magnesium import.186 In renal epithelial cells, claudin-16 (CLDN16) and CLDN19 are involved in divalent metal import, including magnesium.187 Magnesium transporters responsible for transport into and out of cell organelles have been described. These include mitochondrial magnesium channel MRS2188,189 and mitochondrial magnesium export protein SLC41A3.190 Magnesium is also found abundantly in the ER, and TMEM94 has been described as an ER-specific magnesium transporter.191
Cellular Functions of Magnesium
Magnesium plays key roles in the cell as a regulator of metabolic processes. It affects the activity of a large number of proteins, exerting an activity by directly binding to substrates, such as ATP,192 binding to the active sites of enzymes, or acting by means of metalloallostery, promoting enzyme complex formation. By doing so, magnesium controls the function of various enzymes and ion channels. It can also promote the folding of nucleic acids, impacting on functions. It plays a role in many fundamental biological processes including DNA replication, transcription and RNA translation.193 Indeed, magnesium stabilizes DNA and RNA structures and many enzymes involving nucleic acid processing require magnesium as a cofactor.194 Magnesium can act as a potent antagonist of calcium channels, such as L-type calcium channel (LTCC), and thus, magnesium levels can impact smooth muscle function, where fast calcium release and subsequent calcium binding to calmodulin regulates smooth muscle contraction.195,196 This exemplifies how a metal can influence the biological effect of another in this setting by directly controlling the influx of other metals via specific cellular transporters. In muscle cells, magnesium supplementation has been shown to activate mTOR signaling.197 It was argued that excess magnesium could promote the formation of magnesium–ATP complexes, thereby promoting phosphorylation cascades and stimulating mTOR signaling. Since other signaling pathways require phosphorylation and magnesium is essential for the activity of kinases, magnesium can stimulate other signaling cascades, including Janus kinase/signal transducer and activator of transcription (JAK/STAT) and MAPK.198,199 This might also explain how magnesium can promote osteogenic differentiation of mesenchymal stem cells.198
Magnesium Signaling and Diseases
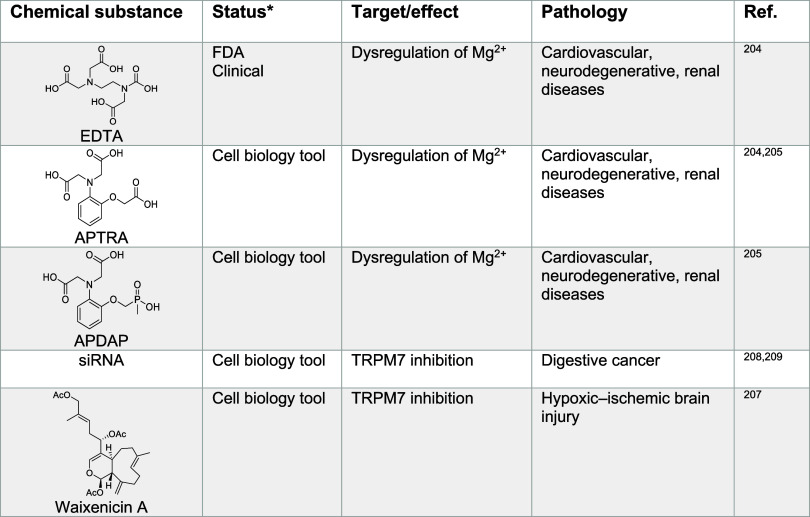
Magnesium levels are altered in several human disease settings, including cancer, cardiovascular diseases, neurological disorders, renal disorders, and diabetes.200,201 In particular, many cardiovascular defects are associated with reduced cellular magnesium influx and increased intracellular calcium levels. Thus, magnesium supplementation can actually alleviate some of the associated symptoms.202,203 Small molecules that chelate magnesium have been developed as laboratory tools to study cardiovascular, neurodegenerative, and renal diseases. These include ethylenediaminetetraacetic acid (EDTA), aminophenol triacetic acid (APTRA)202 and aminophenol-N,N-diacetate-O-methylene-methylphosphinate (APDAP)203 (Table 3), although some of these compounds can adversely alter metal homeostasis more broadly, lacking specificity for magnesium binding.
Magnesium Signaling in Cancer
In cancer, the contribution and subsequent effects of magnesium are also complex. Although an increase in magnesium levels has been reported in neoplastic cells, it remains unclear whether this effect is due to alterations of calcium levels and signaling or even that of other metals. It has been suggested that increased magnesium concentrations provide cancer cells with a metabolic advantage, in particular as it is needed for ATP production.206 Various studies have illustrated the value of targeting magnesium import into cells, for instance by using blockers of TRPM7, such as waixenicin A207 (Table 3). This strategy holds great promise for the clinical management of cancer.208,209 In addition, since magnesium is crucial for kinase function, signaling pathways like MAPK, JAK/STAT, and others can be impacted by changes in magnesium homeostasis.184 Indeed, several growth factors such as epithelial growth factor receptor (EGFR) and vascular endothelial growth factor receptor (VEGFR) can induce signaling pathways involving effector kinases downstream in the signaling cascade, including JAK/STAT3, MAPK, phosphatidylinositol-3 kinase (PI3K)/AKT/mTOR, and phospholipase C γ/protein kinase C (PLCγ/PKC).184 Thus, it will be important to better characterize the effect of small-molecule modulators of magnesium homeostasis in cancer.
Table 3: Regulators of Magnesium Signaling204,205,207−209
Magnesium Signaling in Immunity and Inflammation
Functional magnesium signaling has been documented in immune cells.210,211 For instance, mutations in MagT1 have been reported in several diseases, including an X chromosome-linked immunodeficiency characterized by CD4+ lymphopenia and in defective T cell activation. In particular, the latter setting hinted toward a direct effect of magnesium impacting cell states. In this case, T cell receptor signaling has been shown to be impaired upon changes in the intracellular magnesium levels. This leads to alteration of PLCγ1 function and reduction of inositol triphosphate (IP3) synthesis, impacting calcium signaling. Another report documented that lymphocyte function-associated antigen 1 (LFA-1) requires magnesium to adopt its active conformation to mediate signaling in T cells, which in turn increases calcium flux, leading to perturbation of cell signaling downstream.212 In neurons, γ-aminobutyric acid type A (GABAA) receptor signaling triggers release of magnesium from mitochondria, which impacts signaling pathways downstream, involving mainly calcium-dependent signaling, including ERK, cyclic-AMP responsive element-binding protein (CREB), and mTOR.213 Patients with non alcoholic steatohepatitis (NASH) are characterized by dysregulated magnesium levels in liver cells due to increased expression of the magnesium transporter CNNM4.214 This opens the possibility of targeting this transporter to potentially control this inflammatory disease. Interestingly, acetaminophen overdose causes liver inflammation, magnesium dysregulation, and CNNM4 overexpression.215 Taken together, targeting CNNM4 and magnesium balance in the liver might constitute a therapeutic route to treat an array of inflammatory liver diseases and disorders.
Calcium Signaling
Regulation of Calcium Homeostasis
Calcium ions are found in a +2 oxidation state. Calcium is abundant in the cell, and cytosolic concentrations as a free ion are low, being mostly bound to biomolecules. Calcium levels within organelles are tightly controlled.32,216−218 Several mechanisms regulating cellular calcium levels have been identified, involving cellular import and export proteins.219−222 Mechanisms of calcium uptake are diverse, the most thoroughly characterized are ion channels,223 including voltage-gated and ligand-gated channels as well as calcium release-activated calcium modulator/stromal interaction molecule ORAI/STIM (Figure ). ORAI/STIM consists of the pore-forming proteins ORAI 1 to 3 and STIM 1 and 2.224 Calcium can bind to and activate the family of calcium-sensing receptors (CaSR),225 which are GPCR that regulate calcium levels in the blood. Calcium uptake has been shown to be sensitive to poisoning and inactivation by other metals,226 including magnesium, cobalt, nickel, cadmium, and manganese ions. These observations support the notion that levels of distinct metals can impact calcium signaling.227 Besides lipid membrane channels that can transport calcium, other studies have shown that calcium can also be taken up by endocytosis,228 and calcium release from late endosomes and lysosomes is pH-dependent. For example, it has been shown that calcium can be taken up via the plasma membrane glycoprotein cluster of differentiation 44 (CD44)/hyaluronan metal endocytosis pathway in cells undergoing cell state transitions, specifically in macrophages acquiring a pro-inflammatory cell state13 and, indeed, calcium signaling is crucial for macrophage activation and function.229
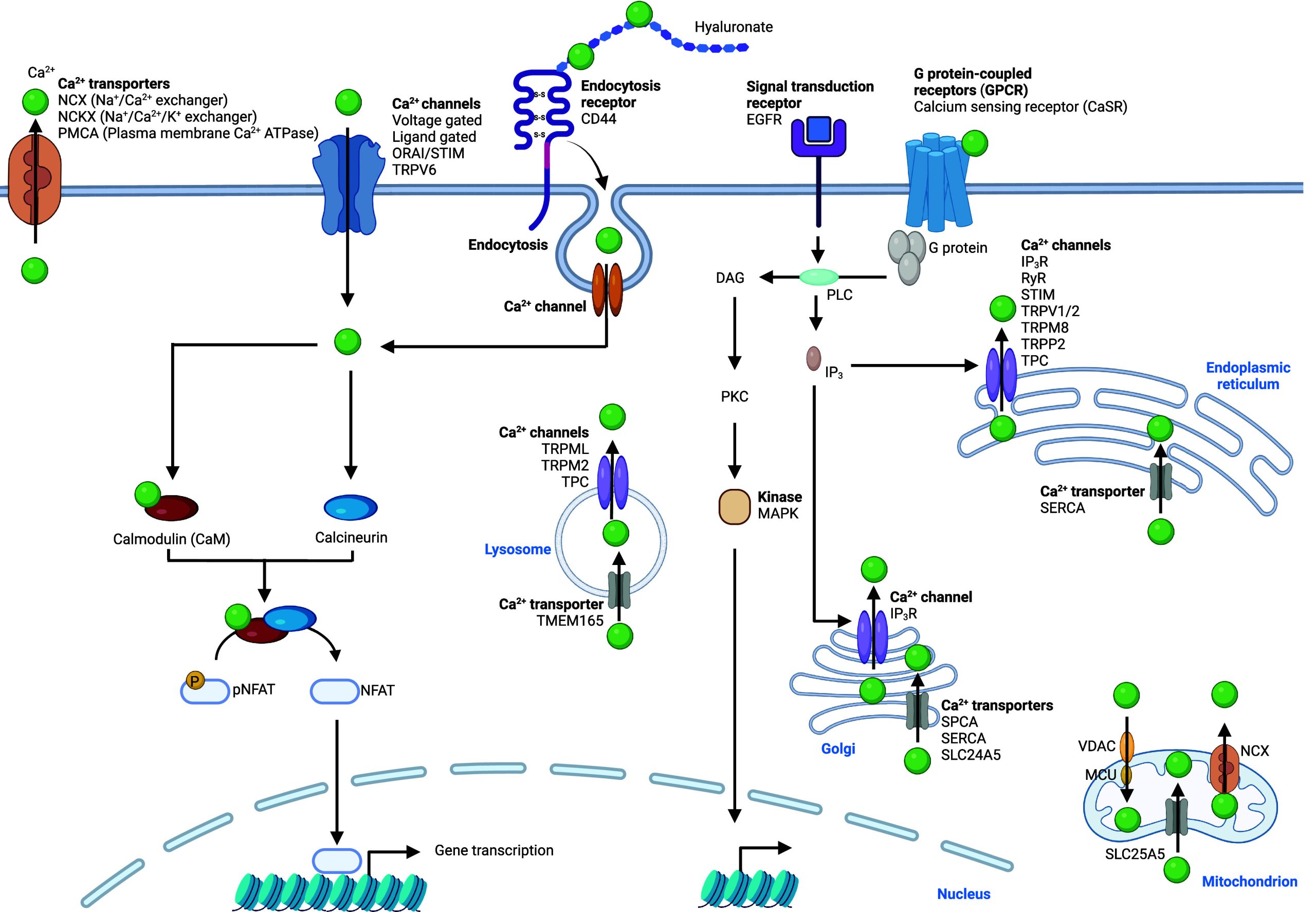
Complex machineries have been documented to be responsible for the release of calcium from cell organelles. Calcium is apparently not translocated from the endolysosomal compartment via divalent metal transporter 1 (DMT1).230 Other ion channels purportedly play that role. Using a calcium-dependent fluorescent pH-dependent reporter molecule (CalipHluor), ATPase cation transporting 13A2 (ATP13A2) has been shown to affect intracellular calcium levels231 and to facilitate lysosomal calcium import.218 Given its role in polyamine transport, it remains to be elucidated how this calcium transport is facilitated.232 The calcium/proton exchanger CAX was identified and suggested to be implicated in translocation of calcium into lysosomes.233 Recently, transmembrane protein 165 (TMEM165) has been identified as an importer of calcium into lysosomes.234 In addition to these importers, calcium has been shown to be exported from this compartment by the calcium channels TRPML, TRPM2, and TPC.219−222 This network of proteins highlights the importance of lysosomal calcium homeostasis for cells and provides the means to manipulate lysosomal calcium pools for cellular functions. In the cell, IP3 is a key signaling molecule that regulates calcium homeostasis (Figure ). IP3 can bind to the IP3 receptor (IP3R), which is mainly situated at the membrane of the ER and the Golgi apparatus, where it causes the release of calcium ions into the cytosol. Ryanodine receptor (RyR) represents another class of receptors mostly located at the membrane of the ER or the sarcoplasmic reticulum (SR) in cardiomyocytes. These receptors can also release calcium into the cytosol upon stimuli, defining a positive feedback mechanism that allows rapid calcium build-up for muscle contraction. Other calcium channels that export this metal ion from the ER include STIM, transient receptor potential vanilloid type 1 (TRPV1), TRPM8, transient receptor potential polycystin-2 (TRPP2), and TPC (Figure ).219−222 The calcium transporter sarcoplasmic/endoplasmic reticulum calcium ATPase (SERCA) has been reported to translocate calcium into the ER and the SR in muscle cells.235 SERCA as well as secretory pathway calcium ATPase (SPCA) are transporters that import calcium into the Golgi apparatus.236 SLC25A5 can also transport calcium into mitochondria and SLC24A5 to the Golgi apparatus.237 It has been shown that mitochondrial calcium uniporter (MCU) interacts with voltage-dependent anion channel (VDAC) to mediate calcium transport to mitochondria,238 and the calcium transporter NCX can export this metal from mitochondria. Calcium export from cells is mediated by different transporters, including the antiporters NCX, NCKX, and the calcium ATPase PMCA.223 Taken together, this complex network of proteins advocates for a prevalent role of calcium signaling, where levels in the cytosol and in different organelles are tightly controlled in a cell type-specific manner.
Cellular Functions of Calcium
Calcium plays a key role in smooth muscle contraction. It binds to calmodulin in the cytosol, which in turn activates myosin light chain kinases.195,196 Calcium is generally released quickly via RyR and IP3R from the ER and the SR in specialized cells.239,240 Calmodulin is a key calcium-dependent signaling protein whose activation occurs upon binding to calcium. Activated calmodulin can then form complexes with various proteins to relay signals, including to TFs,241 such as CREB.242 The regulation of this TF takes place via calcium/calmodulin-dependent protein kinases.243 Importantly, the ER and the SR store intracellular calcium, whose dysregulated homeostasis has been reported in various diseases, including diabetes, cardiovascular diseases, and cancers.244 Given that changes in cell states can rely on alterations of calcium signaling, it is conceivable that controlling calcium levels can provide control over the acquisition of a distinct cell identity. Pioneering work illuminated calcineurin and NFAT22 signaling, a pathway that connects the cell membrane to the nucleus, which is activated by calcium and plays a central role in development and T cell activation. Calcium regulates the function of calcineurin,245 a phosphatase that promotes translocation of NFAT to the nucleus, thereby activating specific transcriptional programs.25 Calcium plays a pivotal role during the development and maturation of neurons and ultimately the formation of neural networks. The underlying gene expression programs of these cells are controlled, at least partly, by calcium via activation of specific signaling pathways.246,247 MAPK signaling involves ERK, and interactions between ERK and other proteins are regulated by calcium.248 Another study demonstrated that calcium concentrations changed during the early steps of embryonic stem-cell-derived neural precursor development. These changes were linked to a functional calcium response network and alterations in RyR receptor expression during different stages of development.249 Interestingly, oscillations of calcium spikes were observed during neuronal development.250
Calcium Signaling and Diseases
Calcium Signaling in Cancer
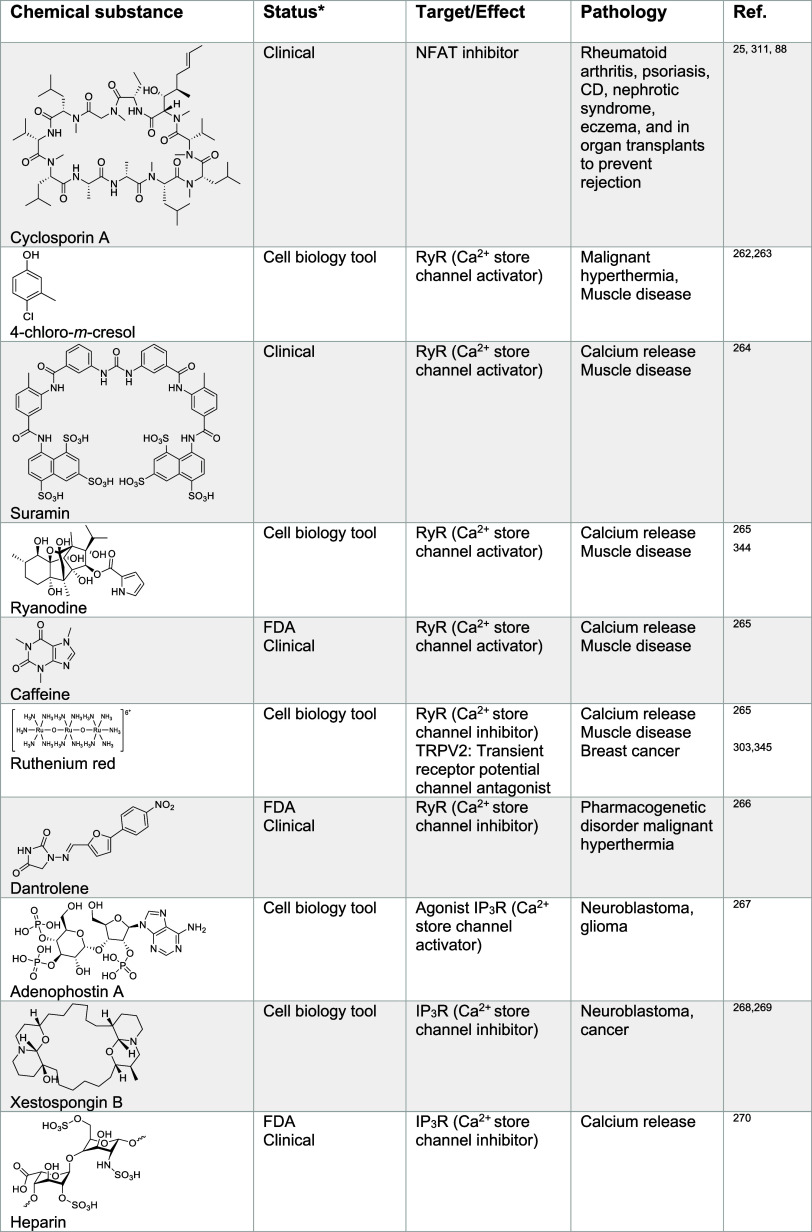
Specific GPCR are located in the cell membranes of particular cell types and can sense extracellular calcium levels. These GPCR are CaSR that are essentially found in neuronal cells. These receptors mediate important cellular signaling functions,251 relaying signals by means of induced conformational changes upon calcium binding in the extracellular matrix. For instance, it has been proposed that neural cell plasticity is strongly controlled by extracellular calcium signaling rather than intracellular calcium levels.252,253 CaSR dysregulations have been associated with cardiovascular diseases and cancer.254 In colorectal cancer, CaSR have been associated with antitumorigenic properties. In this context, receptor agonists represent interesting candidates for the treatment of colorectal cancer.255 Indeed, elevated intake of dietary calcium has been associated with reduced risk of colon cancer.256 In general, calcium levels and calcium signaling are altered in cancer cells.257 Signaling via these CaSR can determine cell fate, which makes them attractive potential therapeutic targets. These receptors are not solely activated by calcium, but also by molecules such as polyamines like spermine and the aminoglycoside antibiotic neomycin,258−260 providing a starting point for the development of new anticancer strategies. Given the effect of spermine on calcium and potassium channels (Tables 2 and 4), this opens the question of the specificity of this molecule and potentially others. Targeting calcium signaling in cancer, in particular specific calcium channels, has gained momentum with the development of several promising small molecules with therapeutic potential261 (Table 4). For instance, 4-chloro-m-cresol, suramin, and ryanodine were shown to activate RyR,262−265 whereas dantrolene and ruthenium red can inhibit these channels.265,266 IP3R activators, such as adenophostin A can promote calcium release from the ER.267 Conversely, IP3R inhibitors such as xestospongin B268,269 or heparin270 can block calcium translocation into the cytosol. Interestingly, xestospongin B has been shown to trigger apoptosis in neuroblastoma cells in vitro.268 Apoptosis has been linked to alterations of calcium homeostasis,271 potentially involving the B-cell lymphoma 2 (Bcl-2) protein regulating calcium fluxes272 and inducing ER stress. Dysregulation of ER calcium levels have been reported in cancer cells where calcium might be involved in malignant transformations, making small molecules modulators of calcium channels activity valuable cell biology tools and potential therapeutics in oncology.273 Other small molecule activators and inhibitors of calcium channels have been identified, including compounds targeting ORAI/STIM,274−280 voltage gated channels281−288 and others (Table 4). Notable ORAI/STIM inhibitors include N-methylnitrendipine (MRS-1844) and N-propargylnitrendipine (MRS-1845) have proven to be effective against lung cancer cells and leukemia.276,277,289 1-(5-Chloronaphthalen-1-yl)sulfonyl-1,4-diazepane (ML-9) has reached clinical development for the treatment of prostate cancer.275,276,290−292 The Cav3.1 to Cav3.3 inhibitor amlodipine is being developed for the treatment of epidermoid carcinoma.287 Interestingly, inhibitors against lysosomal calcium channels are also being developed, including the FDA-approved antifungal drug clotrimazole.293 Although imidazole-containing antifungal agents such as clotrimazole and econazole inhibit ergosterol biosynthesis in fungi leading to cell wall damage in these organisms, they can readily coordinate transition metals such as calcium.294 More research is required to study the mechanism of action (MoA) of ion channel inhibition by these compounds. TRPM8 inhibitors, including cannabigerol295,296 and M8-B296,297 are being studied for the treatment of lymphoma, lung cancer, breast cancer, and prostate cancer. In addition, the TRPV1 inhibitor capsaicin298−302 is in developement for the treatment of renal carcinoma, and the TRPV2 inhibitor probenecid303 has shown promising clinical effects against glioblastoma and bladder cancer. The synthetic peptides and TRPV6 inhibitors soricidin and SOR-C13304 have been shown to exhibit promising results in vitro for the potential treatments of ovarian, prostate, and brain cancers. Finally, the small molecule thapsigargin305 and the artificial peptide G202306 have been reported to inhibit SERCA for the treatment against several cancers, including prostate, colon, breast, and cervical cancers. Given the complexity of calcium transport and the plethora of calcium channels present in cells, with differential levels of expression in different tissues, targeting calcium transport selectively in tumors is a challenging endeavor and should be exploited taking into account tissue-specific features.307 The role of inflammation in tumorigenesis308 and the fact that calcium signaling plays a role in the control of cell plasticity in immunity and cancer raises the prospect of targeting calcium homeostasis for the clinical management of cancer.
Table 4: Regulators of Calcium Signaling24,25,88,258,260−270,274−306,311,312,314−317,322−325,330,341,344−397
Calcium Signaling in Immunity and Inflammation
Calcium signaling regulates NFAT transcriptional programs and T cell activation.25 Additionally, calcium levels were found to be elevated in inflammatory macrophages13 and lysosomal calcium trafficking has been shown to regulate dendritic cell migration,309,310 further supporting the central role of calcium in immunity. The small molecules FK-506 and cyclosporin A (Table 4) form complexes with distinct immunophilins, including FK-506-binding protein and cyclosporin-binding cyclophilin, respectively, susceptible to inhibit the calcium- and calmodulin-dependent phosphatase calcineurin. This in turn prevents NFAT dephosphorylation and translocation to the nucleus inhibiting associated transcriptional programs underpinning activated T cells.25,311−313 Since NFAT signaling plays key roles in immune cell activation, the small molecule FK-506 has been exploited for the treatment of eczema and psoriasis, whereas the related natural macrolide rapamycin is used to treat lymphangioleiomyomatosis,314−316 a rare systemic disease that results in the destruction of lung tissues. These compounds are also used in the context of organ transplants to prevent immune responses and organ rejection.317−320 Calcineurin has been shown to play a role in macrophages.321 Since calcium levels directly control the NFAT phosphorylation status, manipulating calcium levels can provide control over cell plasticity in immune cells, with major implications in settings such as autoimmune diseases and acute inflammation. Small molecules that can interfere with calcium signaling, either acting through direct calcium binding or via the targeting of other calcium-signaling effectors, including verapamil,281 ghrelin,288 mibefradil,322 2-[2-oxo-2-(2,2,4-trimethyl-3,4-dihydroquinolin-1-yl)ethyl]isoindole-1,3-dione (ML-SA1),323 cinacalcet,324 and others, are listed in Table 4.
Calcium oxalate can induce inflammation in the kidney via a signaling that triggers secretion of interleukin-1β (IL-1β).325 Studies on phagocytes showed that the calcium binding proteins S100 are markers of inflammation, potentially linking inflammatory genes and ultimately the expression of inflammatory proteins to calcium levels.326 RyR are also involved in T cell activation via hematological and neurological expressed 1-like protein (HN1L).327 The so-called calcium microdomains form at very early stages of T cell activation and in neuronal cell signaling. These domains enable the entry of calcium into the cytosol and are key elements of calcium signaling.328 In T cells, the two cation channels P2X4 and P2X7 are involved in calcium signaling, controlling calcium flux and T cell activation.329 Furthermore, nicotinic acid adenine dinucleotide phosphate (NAADP) is structurally related to IP3, which can bind to and open calcium channels inside the cell. The small molecule trans-Ned 19 (Table 4), an antagonist of NAADP-signaling, has been reported to influence T cell plasticity in a mouse model of intestinal inflammation, opening up potential therapeutic opportunities for the treatment of inflammation, including inflammatory bowel disease.330 Taken together, this body of work suggests that modulating calcium uptake and signaling in T cells and other immune cells can have a profound effect on cell plasticity and can be exploited therapeutically in various disease settings by targeting calcium homeostasis regulator proteins, downstream signaling effectors, or calcium itself.
Calcium Signaling in Neurological Disorders
Calcium is an important signaling effector connecting neurons in synapses via voltage-gated calcium channels among others.331 It is involved in the transmission of the depolarizing signal in synapses and thus plays a key role in information exchange between neurons.332−335 In addition, calcium has been shown to regulate gene expression, including genes that encode for neurotransmitter receptors in neurons by stimulating CREB-mediated transcription.336 This indicates that calcium plays a dual role, influencing action potentials and controlling the underlying expression of neuronal signal transduction transmitters. Specific neurons, such as GABAergic neurons express high levels of the calcium-binding protein parvalbumin, which acts as a calcium buffer.337 Cells can modulate signaling via proteins that sequester calcium ions. Changes in calcium levels have been observed in aging nervous systems and correlations with neurodegenerative diseases have been documented.338,339 Variations of calcium channel expression levels have been observed at the cell membrane, mitochondria and the ER during aging and in neurological disorders, including Alzheimer’s disease (AD) and Huntington’s disease (HD).340 In a cellular model, it was shown that mutated versions of the protein Huntingtin can increase calcium flux in cells.341 ER-mediated calcium signaling is a key determinant during neural cell plasticity affecting their dendrites. In addition, changes of calcium homeostasis of the ER have also been observed in AD and a functional link remains to be elucidated.342,343 Penfluridol has been successfully used to block T-type voltage-gated calcium channels in academic research, providing new insights for the development of drugs for clinical applications.284 The related T-type voltage-gated calcium channel inhibitor pimozide (Table 4) has been developed for the treatment of neurological disorders including epilepsy.284
d-Block Metal Ion Signaling
Manganese Signaling
Regulation of Manganese Homeostasis
Manganese is found in the +2 and +3 oxidation states in the cell and can participate in redox reactions. Concentrations of manganese in cells are tightly regulated, and manganese overload can lead to a poisoning condition known as manganism.398 This metal exhibits neurotoxic effects upon excessive exposure, and it has been associated with neurodegenerative diseases such as AD and Parkinson’s disease (PD).399−401 Several cellular uptake mechanisms have been described (Figure ). Ion channels enabling manganese transport through lipid membranes have been reported, including ZRT/IRT-like protein 8 (ZIP8), SLC39A14, calcium channels, and dopamine transporter 1 (DAT1) among others.402−406 Furthermore, it has been shown that manganese can be taken up by CD44/hyaluronan in macrophages,13 and also forms a complex with transferrin (Tf) that can be internalized by transferrin receptor (TfR1) via a mechanism akin to that involving iron and apotransferrin.407−409 Upon endocytosis and reduction of manganese(III), manganese(II) can be translocated from late endosomes into the cytosol by DMT1.402 Since DMT1 can transport manganese(II), it can also potentially mediate the import of manganese directly into the cell when localized at the plasma membrane, as reported for enterocytes. How manganese(III) is reduced in endosomes remains poorly understood. It is conceivable that metal reductases such as 6-transmembrane epithelial antigen of prostate (STEAP) mediate this reaction.408 Alternatively, since manganese(III) can easily capture electrons when subjected to the reducing environment of the cell,410 notably in the presence of glutathione (GSH), specific metal reductases may not be involved.
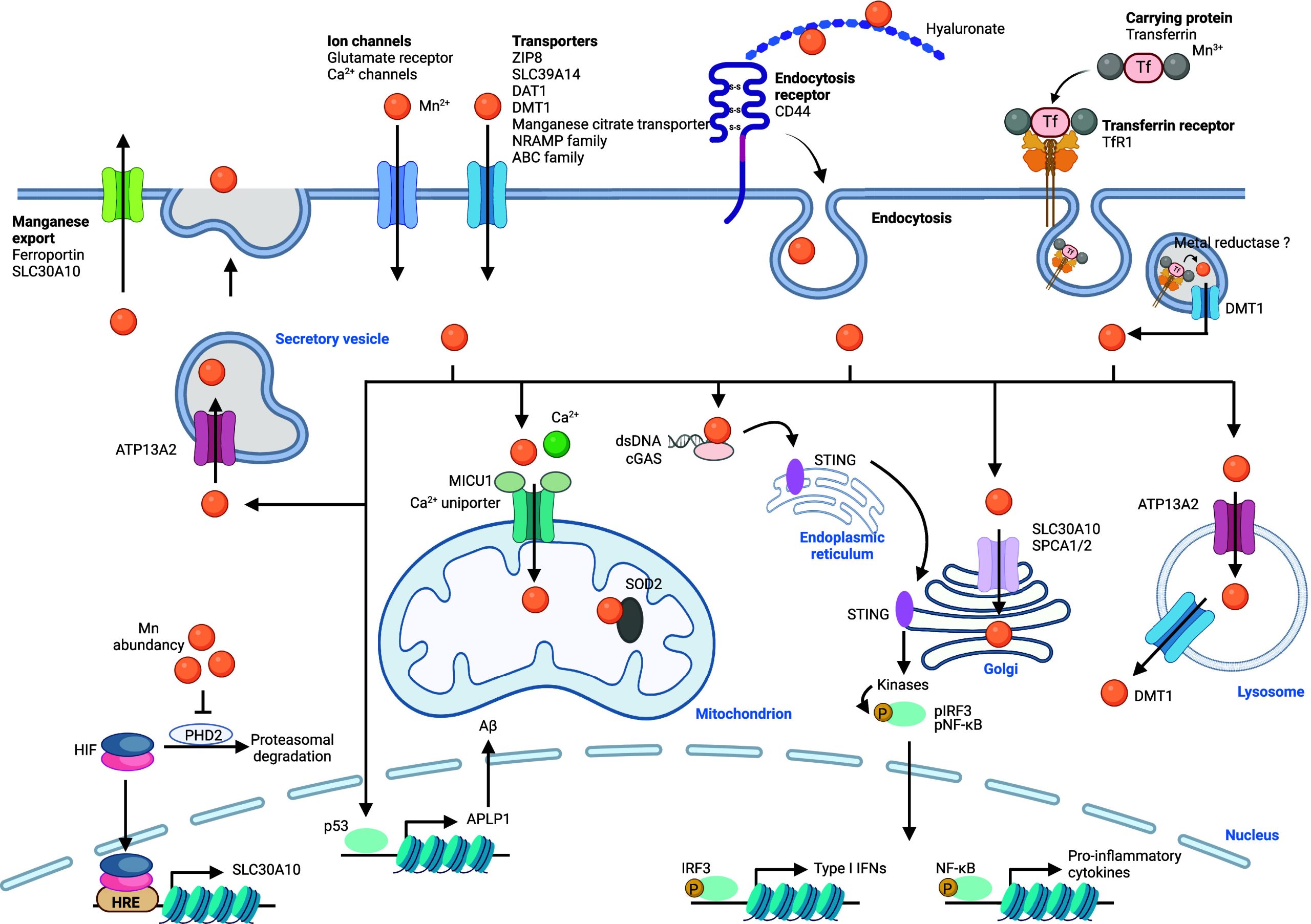
Manganese is transported into various organelles within the cell via specialized transporters including SLC30A10 and SPCA1/2 that drive manganese to the Golgi apparatus, and ATP13A2, which regulates manganese translocation into lysosomes and excretory vesicles411 (Figure ). The calcium uniporter situated in mitochondrial membranes can transport both calcium and manganese into the mitochondria. Interestingly, mitochondrial calcium uptake 1 protein (MICU1) interacts with the calcium uniporter to control its ability to translocate divalent metals. MICU1 contains a metal binding site, and calcium binding has been shown to elicit a conformational change of MICU1, which enables transport of divalent metals by the calcium uniporter.412 Manganese, however, can also interact with the metal binding site of MICU1, but it does not cause the same conformational change, leading to an inactivation of the calcium uniporter. Thus, when excess calcium is present, both calcium and manganese can be transported into mitochondria via this transporter, whereas excess manganese inhibits metal transport.413 SLC30A10 can also localize at the cell membrane and mediate the export of manganese. Interestingly, the iron export protein ferroportin has also been shown to control the cellular export of manganese.414 The expression of SLC30A10 is controlled by hypoxia-inducible factors (HIFs), and its expression is increased upon elevated manganese levels to prevent poisoning by this metal.415 Recent work has documented that proline hydroxylase 2 (PHD2) can be inactivated by manganese replacing reactive iron(II) when manganese levels are high. Since PHD enzymes control proline hydroxylation of HIFs, which leads to HIF degradation, inactivation of these enzymes leads to elevated HIF and SLC30A10 levels. Thus, PHD2 could be defined as an intracellular manganese sensor.416 Manganese homeostasis has traditionally been difficult to dissect due to the lack of adequate biological or chemical tools. Recently, cell-permeable fluorescent manganese(II)-specific probes have been developed to dissect manganese homeostasis,417 identifying the Golgi apparatus as a manganese storage site.418
Cellular Functions of Manganese
In mitochondria, manganese is part of the active site of the mitochondrial enzyme superoxide dismutase 2 (SOD2).419 Both the +2 and +3 oxidation states of the metal are involved in the disproportionation of superoxide radical.420 Manganese has been found to be upregulated in mitochondria of inflammatory macrophages.13 The production of hydrogen peroxide by SOD2 was found to be crucial for the interconversion of NADH into nicotinamide adenine dinucleotide (NAD+), enabling the production of metabolites required for the epigenetic regulation of transcriptional programs underlying acquisition of a pro-inflammatory phenotype. cGAS is a cytosolic DNA sensor that plays a crucial role in host defense against viral infections or genotoxic stress.421 Interestingly, manganese can activate monomeric cGAS, which positively couples with double-stranded (ds) DNA-dependent activation, as manganese(II) enhances the detection sensitivity of cGAS for dsDNA, thereby promoting its enzymatic activity.422 In addition, manganese(II) can activate cGAS independently of DNA.423 Activation of STING in turn recruits kinases, which phosphorylate the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and interferon regulatory factor 3 (IRF3), regulating the expression of inflammatory genes in immune cells.
Manganese Signaling and Diseases
Manganese Signaling in Cancer
Acquisition of a pro-metastatic cancer cell state has been shown to require a metabolic reprogramming similar to that found in immune cells for activation,13 supporting a role of manganese in cancer metastasis.424 Interestingly, increases of manganese levels are associated with metastatic potential in tumors.425 SOD2 mimetics, such as the manganese complex-forming small molecule M40403 (Table 5), have been shown to exhibit anticancer activity as well as effects on immune responses.426 SOD2-dependent increase of hydrogen peroxide has been shown to promote ERK1/2 signaling and transcriptional changes impacting metalloproteinase expression.427 Interestingly, iron can substitute manganese in SOD2 under iron-high and manganese-low conditions. This was shown to alter SOD2 activity, breaking down hydrogen peroxide and causing the production of reactive oxygen species (ROS) instead.428 Thus, alterations in the manganese/iron balance can profoundly impact the biology of the cell. cGAS-STING is also involved in immune sensing in the context of cancer.429 For instance, it was reported that manganese enhances cancer detection by the innate immune system.430 Indeed, manganese-loaded nanoparticles enhance cGAS-STING activation, which holds great potential for cancer immunotherapy.431 The cytokine interleukin-2 (IL-2) is used to treat malignant melanoma and metastatic renal cell carcinoma. However, IL-2 treatment can also cause hypertension, which limits its use. The SOD mimetic M40403 can reduce hypotension caused by IL-2, allowing for an increase of IL-2 administration in mice and enhancing its anticancer activity.426
Manganese Signaling in Immunity and Inflammation
Manganese increases the innate immune response in lipopolysaccharide-induced sepsis.432 Since manganese levels can affect the efficacy of the cGAS-STING pathway, manganese plays an integral role in host defense against viral infections.433 Targeting manganese signaling represents a potential therapeutic strategy in inflammatory diseases, aging-related inflammation, and neurodegeneration.421,434 Manipulating manganese homeostasis is a tractable strategy to control the innate immune response in infectious diseases and cancer.421,429 In line with these studies, in vitro experiments have revealed that manganese treatment leads to an increase of the expression of genes involved in the innate immune response, which is consistent with the role of manganese in the maintenance of NAD+ and the epigenetic control of transcriptional programs underlying inflammation.13,435
Manganese Signaling in Neurological Disorders
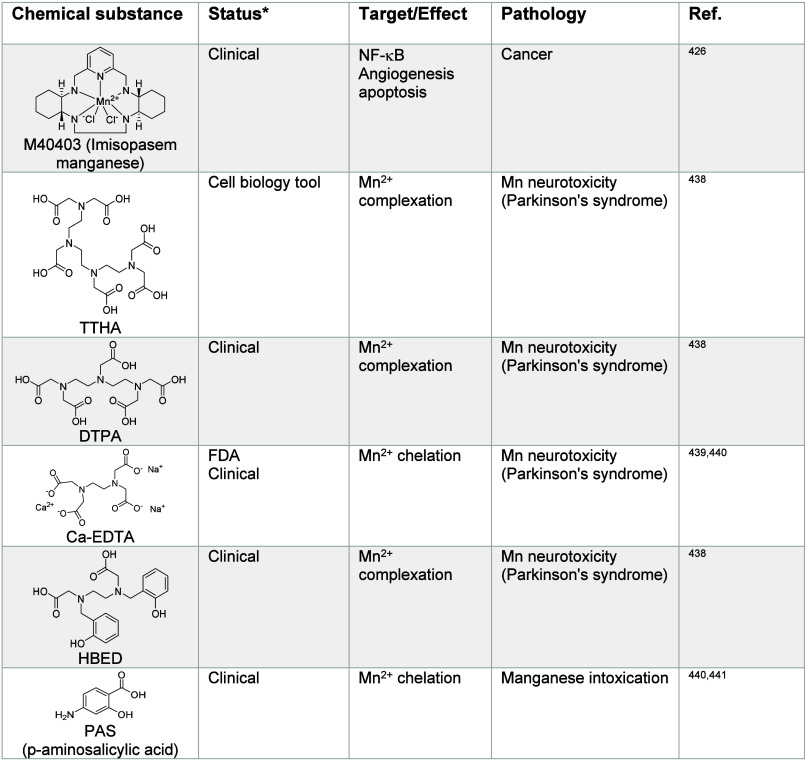
It has been shown that mutations of the cellular manganese importer SLC39A14 can lead to impairment of manganese transport and childhood-onset parkinsonism-dystonia,436 highlighting the role of manganese in brain development and neuronal plasticity. Upon increased exposure to manganese during development, manganese can accumulate in the basal ganglia and cause manganism, conferring a PD-like syndrome.437 Manganese chelators, such as triethylenetetramine hexaacetic acid (TTHA), DTPA and N,N′-di(2-hydroxybenzyl)ethylenediamine-N,N′-diacetic acid monohydrochloride hydrate (HBED)438,439 have been studied to treat manganism and manganese-induced neurotoxicity (Table 5). However, these small molecules can also chelate other metal ions and cause side effects.
Table 5: Regulators of Manganese Signaling426,438−441
Iron Signaling
Regulation of Iron Homeostasis
The regulation of cellular iron homeostasis is complex.442 In the cell, iron is mainly found as +2 and +3 oxidation states as well as a crystalline form of iron oxide. Unlike alkali and alkaline earth metal ions, iron is redox active under physiological conditions. It can form complexes with proteins, such as Tf and ferritin, glycans, such as hyaluronan, or is found as a labile iron pool, i.e., free species. Cellular iron homeostasis has been thoroughly documented with the discoveries of import, traffic, storage, and export mechanisms, where iron is involved in the regulation of many biological processes442,443 (Figure ). Cellular iron uptake can be mediated by several mechanisms, including the Tf/TfR1 mechanism. Tf binds to two iron(III) ions (holo-transferrin) and this complex becomes a competent binding partner of TfR1, which is internalized via a clathrin-dependent endocytosis mechanism.444 TfR1 then recycles back to the cell surface. Iron can also be imported into cells via transporters, such as ZIP14, ZIP8, or DMT1. For example, DMT1 is found on the cell membrane of enterocytes, where it mediates transport of iron into the cell. Tf-mediated cellular iron import has long been considered to be the main mechanism of iron endocytosis. The literature, however, refers to two distinct pools of iron in the blood: a pool that is Tf bound and another that is not transferrin-bound. In cancer and immune cells acquiring a distinct identity, i.e., cell-state transition,445 CD44 mediates endocytosis of iron(III)-bound hyaluronan,446 via a clathrin-independent endocytosis mechanism.447 There is a prevalence of metal binders in blood, plasma, and the extracellular space. The chemical composition of diseased tissues has been shown to be distinct from that of healthy tissues, for example, with the abundance of hyaluronan in the stroma of pancreatic tumors and in the lung of severe COVID-19 patients,448,449 which argues for the prevalence of CD44-mediated iron uptake over other mechanisms, and this plays a role in the control of cell identity by iron in cancer and inflammation.446,450,451
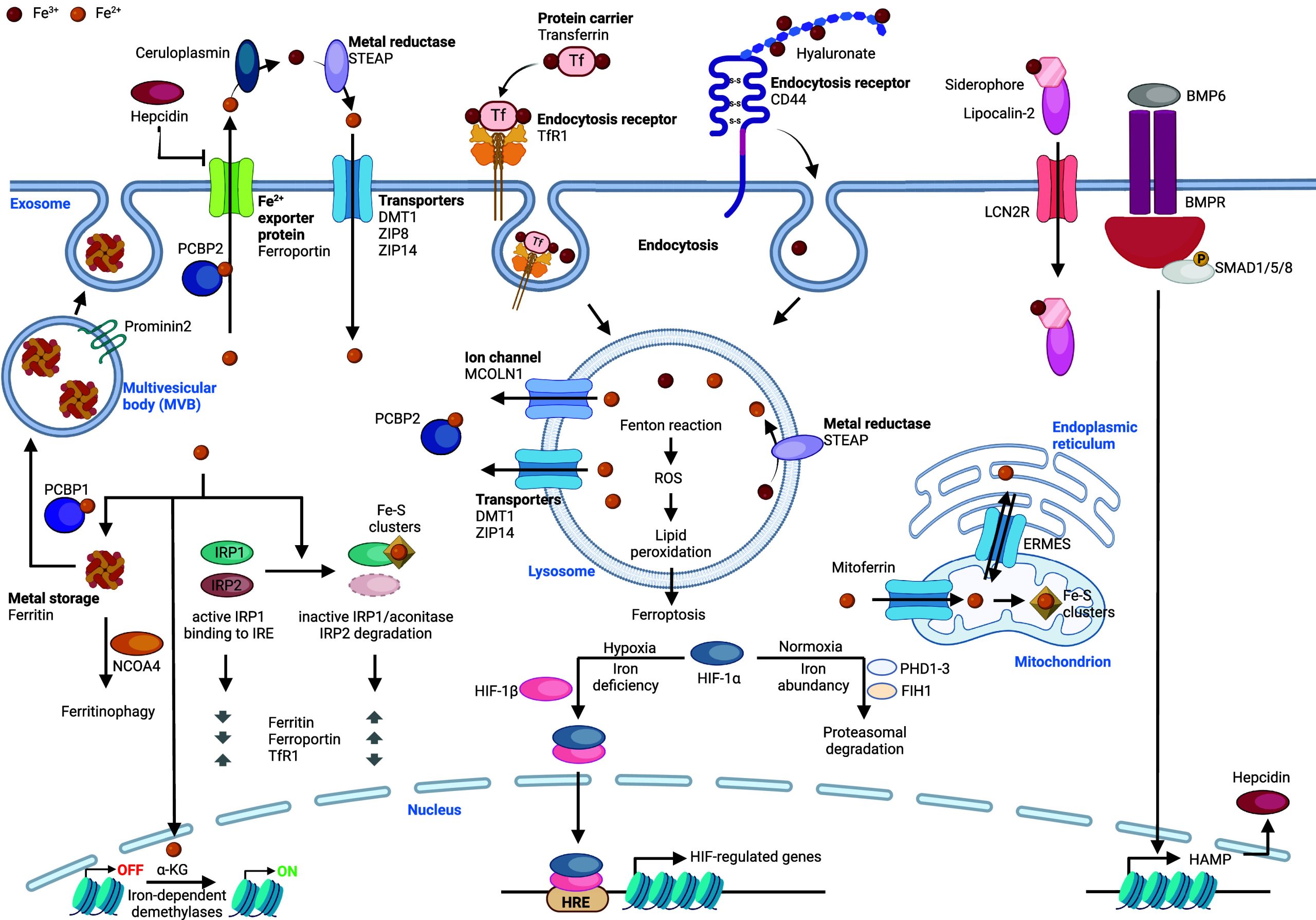
Importantly, intracellular iron regulates the function of iron regulatory protein 1 (IRP1) and IRP2, which are RNA-binding proteins.452 IRP1 is the cytosolic isoform of the mitochondrial aconitase. Iron regulates the binding of IRPs to specific RNA stem-loop structures, called iron-responsive element (IRE), to the 5′- and 3′-untranslated regions (UTRs) of various mRNAs including those encoding TfR1 and ferritin. Interestingly, IRP1 can form an iron–sulfur cluster that prevents its binding to IREs impacting translation. For example, when iron levels are low, IRP1 can bind to multiple IREs in the 3′-UTR of TfR1, stabilizing the mRNA transcript and promoting protein biosynthesis. In contrast, binding of IRP1 to the 5′-UTR of ferritin inhibits translation.453 Thus, while expression of TfR1 is reduced upon increase of cellular iron, expression of the iron storage protein ferritin is increased upon iron uptake.454,455 This iron signaling mechanism allows the cell to maintain low basal levels of iron, reducing endocytosis and increasing storage to avoid oxidative stress when the iron demand in the cell is moderate. In contrast, CD44 expression is unlocked by iron at the epigenetic and transcriptional levels, allowing cells to transiently upregulate the iron content when the demand is higher, for instance to enable rapid acquisition of a distinct cell state independently of genetic alterations, such as epithelial-to-mesenchymal transition (EMT) in cancer446 (Figure ).
Upon endocytosis, iron(III) is reduced by specific metal reductases, including the STEAP family of proteins.456 Iron(II) is then exported from the lumen to the cytosol via DMT1, where it is transferred to the iron chaperone PCBP1 and PCBP2.457 Iron is stored in the cell, forming a complex supramolecular structure with ferritin. Ferritin can form a multimeric complex composed of 24 subunits of ferritin heavy chain (FTH1) and ferritin light chain (FTL).458 FTH1 exhibits intrinsic ferroxidase activity, allowing oxidation and storage of iron as a crystalline redox-inactive form in the inner core of the ferritin multimer. Iron can be released from ferritin through ferritinophagy, a mechanism that targets ferritin for degradation.459 In this process, ferritin shuttles to autophagosomes via nuclear receptor coactivator 4 (NCOA4),460 where it is targeted for proteolysis in autolysosomes. NCOA4 is a key regulator of the available iron in the cell. Thus, interfering with ferritinophagy can impact DNA replication and induce cell proliferation defects as well as other processes reliant on iron chemistry.461
Regulation of the subcellular distribution of iron in the cell is critical and remains incompletely understood. For example, mitoferrins have been shown to mediate transport of iron to the mitochondria.462 Iron trafficking to the ER is more complex, as it requires the endoplasmic reticulum mitochondria encounter structure (ERMES), directly linking the ER with the mitochondria for iron exchange.463 Transport and regulation to and from other organelles are less documented. For instance, it is not yet clear how iron shuttles to the cell nucleus, a cellular compartment where iron controls key processes including epigenetic regulation and transcription.
Iron export from the cell is mediated via ferroportin,464 which receives iron from PCBP2.465 Interestingly, ferroportin can be blocked extracellularly by the hormone hepcidin,466,467 providing another level of control of cellular iron levels. Hepcidin itself is regulated by bone morphogenetic protein 6 (BMP6), which is also regulated by iron levels via the TF nuclear factor erythroid 2-related factor 2 (NRF2).468 Furthermore, iron-containing ferritin can be exported from cells.469 In line with this, Prominin2 has been implicated in the regulation of lipid dynamics and can promote the formation of ferritin-containing multivesicular bodies (MVBs) and exosomes to export cellular iron from mammary epithelial and breast carcinoma cells. This adaptative mechanism has been shown to protect cells against ferroptosis.470
Cellular Functions of Iron
Iron is a major functional component of hemoglobin, the metalloprotein in erythrocytes that enables oxygen transport in most animals.471 Heme comprises a complex of iron(II) tetracoordinated within a tetrapyrrole ring. Hemoglobin can reversibly bind to molecular oxygen and carbon dioxide in a pH-dependent manner. Thus, with the appropriate chemical environment, iron can fix and transport oxygen from the lungs to other tissues, enabling cellular respiration and transporting carbon dioxide back from cells for clearance.
Hypoxia-inducible transcription factors (HIFs) are TFs that respond to cellular oxygen levels.472,473 Interestingly, many gene targets of HIF are involved in the regulation of iron homeostasis.474 HIF is regulated by iron-dependent proline dioxygenase proteins that use iron and molecular oxygen to promote HIF hydroxylation.475,476 This enables binding of von Hippel–Lindau tumor suppressor (pVHL), which exhibits E3 ubiquitin ligase activity, leading to ubiquitination and proteasomal degradation of HIF. Interestingly, iron chelation and treatment with cobalt(II) ions inhibit pVHL binding and mimic hypoxia. Under hypoxic conditions, HIF is stable and regulates a specific transcriptional program.477,478 Thus, iron can act as a transcriptional repressor. Interestingly, besides its function in oxygen transport, heme itself can act as a cell signaling molecule,479 impacting on the activity of TFs480−482 and kinases.483 Cellular import and export of heme are regulated by a complex protein network.484
Iron is required for many other cellular and physiological processes, including erythropoiesis, immune function, mitochondrial respiration, nucleotide biosynthesis, DNA repair, DNA replication, and telomere maintenance.485,486 For instance, iron is a key component of iron–sulfur clusters found in proteins of the ETC and can act as a cofactor of specific enzymes in the cell. In mitochondria, iron controls cell metabolism, notably with the production of key metabolites required for the control of the epigenome and cell identity.446,487 This includes α-ketoglutarate (α-KG), a metabolite produced downstream of the iron–sulfur cluster-containing aconitase, an enzyme of the Krebs cycle that uses iron as a cofactor to mediate its activity. Specific demethylases of histones and nucleic acids exploit α-KG and molecular oxygen as cosubstrates of demethylation. Iron acts as a rate-limiting catalyst of these demethylases, which include histone demethylases of the Jumonji family, ten-11 translocation (TET) enzymes, fat mass and obesity-associated protein (FTO)488 and AlkB homologue 5489 that target methylcytosine and N6-methyladenosine, respectively.490−493 Thus, in contrast to its role as suppressor of HIF-related transcriptional programs, iron also mediates cell signaling by unlocking other transcriptional programs underlying epigenetic and epitranscriptomic control of cell identity in cancer and immunity.446
Iron Signaling and Diseases
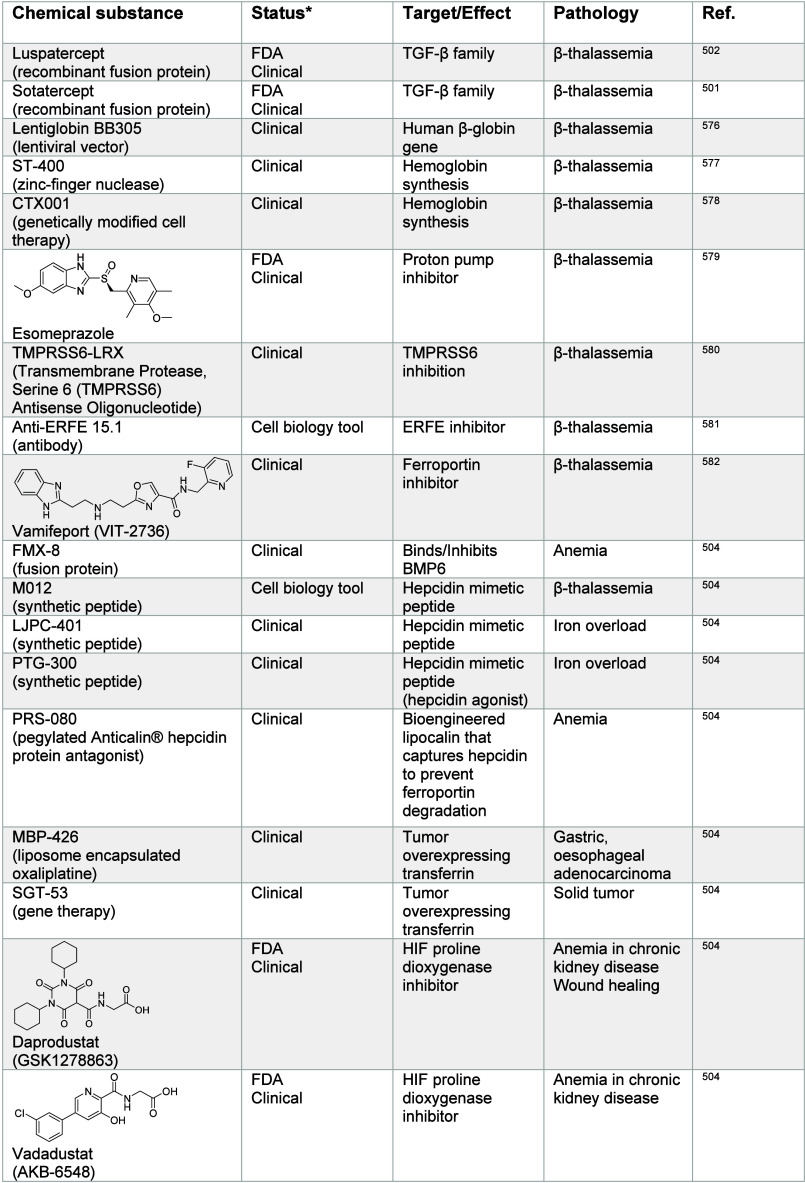
Imbalances in iron homeostasis can cause diseases.494 Iron-overload diseases, such as hereditary hemochromatosis, are characterized by tissue damage and fibrosis, which can lead to organ failure.495 In contrast, iron-deficiencies, also called anemia, can lead to poor oxygenation of tissues.496 Several strategies have been established to treat anemia-related diseases including supplementation with iron salts. Moreover, thalassemias make up a group of genetic diseases characterized by the production of abnormal hemoglobin. The most frequent is β-thalassemia,497−500 where mutations of β-globin have been reported. Standard-of-care treatments include regular blood transfusions. Alternative therapeutic strategies that can restore or improve hemoglobin function have been explored.501−503 Luspatercept is a recombinant fusion protein derived from activin receptor type II B that can bind to transforming growth factor (TGF) and reduce the suppressor of mothers against decapentaplegic (SMAD) signaling. This in turn leads to improved erythroid cell maturation.500 Accumulation of pulmonary iron has been associated with lung dysfunction. In contrast, HIF dysregulation has been observed in anemia caused by chronic kidney disease, which can be treated with HIF proline dioxygenase inhibitors such as daprodustat, vadadustat, molidustat, and desidustat (Table 6).504
Table 6: Regulators of Iron Signaling501,502,504,511,514,516,518,519,530,531,540−542,547,548,551−553,566−568,573−610
The tight regulation of free iron in the blood also plays a role in bacterial infection. Indeed, bacteria can secrete siderophores to sequester iron, which is essential for their growth. These include the plague-causing pathogen Yersinia pestis, among others.505 In humans, macrophages secrete lipocalin-2 (LCN2) upon stimulation with Toll-like receptors (TLR), which sequesters bacterial siderophores, limiting iron supply to bacteria.506
Iron Signaling in Cancer
Changes in iron metabolism fuel cancer progression on several levels, including cell proliferation, cancer cell invasion, and metastasis formation, as well as cancer recurrence and drug resistance.507,508 Iron is required for fundamental cellular processes by healthy cells, cancer cells, and importantly also by other cells comprising the tumor environment, such as cancer-associated fibroblasts, immune cells, endothelial cells, pericytes, and others. Thus, different cell types may compete for iron uptake. For instance, in renal cell carcinoma, it has been shown that macrophages of the tumor microenvironment secrete and supply iron to cancer cells.509 This is reminiscent of metabolic competition in the tumor microenvironment that drives cancer progression.510 Thus, the iron supply to the tumor impacts cancer progression.
Increase of cellular iron characterizes cancer cells in the drug-tolerant persister (DTP) state, which has been linked to cancer metastasis, relapse, and poor clinical outcomes.511−513 This feature has previously been exploited in preclinical models of cancer refractory to standard-of-care treatments, where small molecule-induced iron retention in lysosomes leads to a buildup of ROS in lysosomes, causing membrane lipid oxidation and ferroptosis.511,514 These studies highlight a potential therapeutic strategy for the clinical management of cancer.515 The natural product salinomycin has been identified as a potent eradicator of breast cancer stem cells (CSCs).516 Since salinomycin can act as a ionophore capable of transporting alkali metals across lipid bilayers,517 a MoA involving sodium/potassium transport had been evoked. However, the more potent synthetic derivative ironomycin was found to be effective against CSCs at concentrations that did not substantially alter alkali metal levels, indicating that other mechanisms may be at play in this context.511 Notably, it was found that the MoA of this class of compounds involves direct binding to iron(II) in lysosomes, interfering with iron translocation from the lumen to the cytosol and causing oxidative damage to lipid membranes in this organelle.511,518 In support to this MoA, DMT1 inhibitors, such as substituted pyrazoles and benzylisothioureas (Table 6), can block iron transport from lysosomes, leading to an increase of lysosomal iron and inducing death in cancer cells that had undergone an EMT program, a cell transformation that can confer stem-like properties and tolerance to standard-of-care treatments.519 The endolysosomal compartment plays a central role in the regulation of cellular iron homeostasis.520 Thus, lysosomal iron can be exploited for small molecule intervention. In line with this, iron-activatable prodrugs with a propensity to accumulate in lysosomes were designed. It was shown that chimeras of salinomycin or the related natural product narasin and the organic endoperoxide artesunate (Table 6), which can be readily cleaved upon exposure to iron in lysosomes, induces oxidative stress and ferroptosis.514 Increased iron trafficking was also reported in glioblastoma cancer stem-like cells512 and CSCs of ovarian cancer,513 thus putting forward the idea that other cancer types that are notoriously difficult to treat may be eradicated by directly manipulating cellular iron homeostasis.521 Melanoma cell differentiation is also linked to increased ferroptosis susceptibility,522 further supporting the key notion that cell plasticity requires iron, which confers vulnerability to oxidative stress in cancer.
Cancer and immune cells can undergo cell-state transitions via epigenetic reprogramming. To promote the activity of iron-dependent demethylases, it was found that cells upregulate mechanisms of iron import.13,446 In these settings, CD44-mediated iron endocytosis prevails over the canonical TfR1 pathway, which enables an increase of iron supply, catalyzing epigenetic changes underlying the acquisition of another cell state. Importantly, CD44 expression is not negatively regulated by iron, unlike TfR1, making it possible for cells to transiently increase the iron load. Following Le Chatelier’s principle,523 cells must increase the reagents available at specific chromatin loci to compete against the activity of methyltransferases capable of promoting hypermethylation of key histone and nucleic acid residues, thereby switching off the expression of specific genes and preventing cell state transitions. Changes of cell states underpin many biological processes, such as EMT11,445 and noradrenergic-to-mesenchymal transition in cancers,524 contributing to cancer metastases and the acquisition of a DTP cancer cell state. Other processes involving cell-state transitions include the activation of immune cells,13 including macrophage polarization.525 CD44 has been associated with these processes and is a reliable marker of cell plasticity, playing a central role in metal import and thus cell signaling. Targeting CD44 for therapeutic purposes may represent an interesting strategy in this context.526 CD44 expression is transcriptionally repressed at the epigenetic level by the histone 3 lysine 9 dimethyl (H3K9me2) mark.446 It was shown in triple negative breast cancer cells acquiring a mesenchymal cell state that the expression of CD44 is unlocked by the iron-dependent demethylase plant homeodomain finger protein 8 (PHF8), preferentially targeting H3K9me2.446 These studies show that the cell has evolved complementary iron endocytosis regulatory mechanisms enabling maintenance of functional iron levels, which are lower at the basal cell state, as well as an alternative mechanism required to increase the iron load to promote acquisition of a distinct identity. Thus, iron signaling allows for rapid and reversible cell adaptation that is best exemplified by the capacity of cancer cells to acquire drug-resistant states upon treatment with the standard-of-care and macrophages that rapidly adopt specific properties upon exposure to pathogens for clearance. The regulation of epigenomes defines the cell identity. It relies on complex mechanisms, including the well-orchestrated activities of demethylases527 and methyltransferases among others that target specific histone residues.528 In addition, nonhistone substrates are also targets for iron-dependent demethylases, including DNA and RNA, which can impact the transcriptome, proteome, and cell identity.491,529 Inhibitors of iron-dependent histone demethylases, such as GSK-J4 (Table 6), have been developed to inhibit specific demethylases that catalyze the demethylation of key histone marks.530,531 The iron-dependent histone lysine demethylase 3B (KDM3B) has been described as a sensor for intracellular iron levels that regulate mammalian target of rapamycin complex 1 (mTORC1) via H3K9me2, thus iron also impacts mTOR signaling.532 Manipulating RNA methylation has not yet been exploited with proven therapeutic benefits in clinical settings. Nevertheless, delineating how cellular iron homeostasis globally impacts cell signaling and the acquisition of distinct cell properties holds great promise for therapeutic intervention, despite its complexity. Intriguingly, since cells undergoing cell state transitions require increased levels of iron. The increased amount of this redox-active metal confers vulnerability to oxidative stress and thus to ferroptosis.511,533,534 Cells have developed systems to circumvent undesired chemistry mediated by redox-active iron, including glutathione peroxidase 4 (GPX4) and ferroptosis suppressor protein 1 (FSP1) as well as other lipid membrane repair mechanisms.535−539 Small molecule activators (for instance erastin540 or RSL3541 listed in Table 6) or inhibitors (for instance, liproxstatin-1542) of ferroptosis represent promising therapeutic strategies in diseases where iron has been implicated, including cancer and neurodegenerative diseases among others.543,544 NRF2 is upregulated under conditions of oxidative stress, which regulates the expression of genes that encode proteins involved in the management of oxidative stress.545 Interestingly, inhibition of NRF2 has been shown to lead to increased vulnerability to ferroptosis in ovarian cancer.546 Manipulating NRF2 activity may thus be exploited to sensitize cancer cells to other ferroptosis-inducing small molecules.
Previous work has shown that the iron chelators deferoxamine (DFO) and Dp44 mT can inhibit EMT.547 N-myc downstream regulated 1 (NDRG1) was specifically upregulated in breast cancer cells treated with DFO and 2-hydroxy-1-naphthylaldehyde isonicotinoyl hydrazone (AS8351) (Table 6), and high expression of this gene is associated with reduced tumor growth and metastatic suppression.548 It has been shown that TGF-β/SMAD and wingless/integrated (Wnt) signaling pathways were involved in the regulation of EMT proteins. Taken together, this suggests that targeting iron homeostasis or proteins of signaling pathways involved could be exploited in cancer. Other signaling pathways activated in cancer have been shown to be influenced by iron, for instance the MAPK/ERK pathway in neuroblastoma,549 where iron-dependent production of ROS impacts calcium signaling by activating a calcium channel.
Interestingly, LCN2 is expressed by cancer cells in the cerebrospinal fluid upon stimulation by macrophages.550 It was argued that these cancer cells can outcompete macrophages for iron uptake, promoting cancer growth. Iron chelation therapy with DFO (Table 6) was shown to inhibit cancer cell proliferation. Thus, targeting iron or LCN2 represent promising therapeutic strategies in this context. Indeed, DFO is used in anticancer therapy, for instance in hepatocellular carcinoma.551 Other iron chelators are also used as anticancer treatments, including deferasirox (DFX)552,553 (Table 6). However, DFX has been shown to localize in mitochondria, whereas DFO has been detected in lysosomes554 and the cell nucleus.446,554 Thus, therapeutic and off-target effects resulting from iron chelation are not expected to be identical. Additional investigation is needed to delineate the activity of iron chelators in disease-relevant settings. Importantly, about 40% of cancer patients are undergoing anemia, and clinical management can involve iron supplementation.555 Little is known about the adverse effect of iron supply on cancer progression, which requires further investigation.
Since HIF-1 has been linked to the regulation of glucose metabolism, angiogenesis, and cancer cell invasion, HIF signaling, which depends on iron homeostasis and levels of molecular oxygen, has been considered a potential target for cancer therapy.556 Indeed, many cancers show dysregulated HIF activity. Targeting HIF or HIF-signaling regulators, such as HIF proline dioxygenase,504 represents an interesting axis for the development of anticancer strategies.557
Iron Signaling in Immunity and Inflammation
Increase of iron is associated with macrophage activation in inflammatory settings.13 Notably, the iron homeostasis regulators IRP1 and IRP2 play a critical role in neutrophil development, enabling neutrophil differentiation into fully mature neutrophils.558 Similarly, iron controls the fate of hematopoietic stem cells acting at the chromatin level, which is molecularly reminiscent of mechanisms underlying cell-state transitions in cancer.559 COVID-19 patients are characterized by inflammation of lung tissues, anemia, altered expression of iron-homeostasis genes, low serum iron, and inefficient oxygen transport.560 Activation of macrophages in settings of acute inflammation has been shown to involve CD44-mediated iron uptake and iron-dependent epigenetic programming of the inflammatory cell state.13 Like in cancer cells, immune cells including dendritic cells and macrophages, upregulate iron uptake to promote the activity of iron-dependent demethylases, and unlock the expression of pro-inflammatory genes.13,446 The role of iron in the control of cell states in immunity is also supported by a study showing that a specific histone 3 lysine 27 trimethyl (H3K27me3)-specific demethylase is involved in macrophage activation.530 Increased iron was shown to be associated with increased Wnt signaling in cell culture.561 The protein pirin has been shown to control NF-κB signaling upon binding to ferric iron.562 Iron(III) is susceptible to changing the conformation of pirin, enabling binding to NF-κB. The therapeutic benefits gained by interfering with these processes require further examination. DFO and Dp44 mT (Table 6) treatments also influence c-Jun N-terminal kinase (JNK) and p38 MAPK signaling pathways, leading to cell cycle arrest and apoptosis.563 Iron has also been shown to accumulate in senescent cells, which contribute to the fibrotic disease.564 In addition, nonalcoholic fatty liver disease and steatohepatitis are both characterized by iron overload in hepatic stellate cells and iron-deficient hepatocytes. Both conditions can lead to liver fibrosis.565 Taken together, these studies suggest that a therapy targeting iron or rebalancing iron distribution between cell types could prevent fibrosis or mitigate its effect.
Iron Signaling in Neurological Disorders
Friedreich’s ataxia is a neurological disorder, where a deficiency of frataxin protein leads to aberrant iron–sulfur cluster biosynthesis, thereby impacting other cellular processes. Iron chelation using deferiprone (Table 6) has shown some clinical success, yet it remains currently unknown where in the cell this small molecule exerts its activity and mechanistic insights remain limited.566−568 Finally, iron imbalances have been reported extensively in neurological diseases, including in AD and PD as well as brain aging.569−572 However, the exact etiology of these diseases and the defects induced by iron in this context are not fully understood. Interestingly, the organoselenium small molecule ebselen can inhibit DMT1573 (Table 6) and has been selected for preclinical trials in the context of AD.519,574,575
Copper Signaling
Regulation of Copper Homeostasis
Copper is found as +1 and +2 oxidation states in the cell. Like iron, copper can participate in redox reactions under physiological conditions. In comparison with iron, cellular copper levels are about 100-fold13 lower in cells. In humans, heme, which is composed of an iron(II) core center, is the major component of hemoglobin, allowing for oxygen fixation and transport to cells. However, some invertebrates, including horseshoe crabs, have developed copper-based hemocyanins that play a similar role.611 Several mechanisms of cellular copper uptake have been documented612 (Figure ). The copper transporter 1 (Ctr1)613 has been described as a selective copper uptake protein, which takes up copper(I) ions. This is made possible by the reduction of copper(II) by metal reductases, such as those belonging to the STEAP family at the cell surface.456 Additionally, in endothelial cells, DMT1 localizes at the cell surface and enables the uptake of copper(II).614 In macrophages acquiring a pro-inflammatory phenotype, it has been shown that increase of copper(II) results from the up-regulation of CD44, which enables endocytosis of hyaluronan-bound metals.13 Export of copper(II) from endolysosomes to the cytosol was proposed to involve Ctr2615 and potentially DMT1. Although DMT1 translocates copper(II), the copper species that is transported by Ctr2 has not been well documented. STEAP enzymes are present in lysosomes, and it is conceivable that STEAP4 mediates the reduction of copper as it is emerging as an important regulator for copper homeostasis in the cell.616,617 In addition, SLC46A3 was identified as a lysosomal copper transporter.618 Copper transport and export mechanisms involve copper-transporting ATPase 1/2 (ATP7A/B). These ATPases tightly control cytosolic copper levels,619,620 which is supported by structural data.621 Cellular export of copper by ATP7B has been described to occur by exocytosis.622 In addition, these ATPases can transport copper to and from the Golgi apparatus. Other proteins have been implicated in the regulation of the ATP7A/B localization in the cell. For instance, pleckstrin homology domain containing family A 5 (PLEKHA5), PLEKHA6, and PLEKHA7 recruit PDZ domain-containing protein 11 (PDZD11) to distinct parts of the plasma membrane to promote the anterograde transport of copper by ATP7A to the cell periphery.623 Furthermore, SLC25A3 is a phosphate transporter found in the inner mitochondrial membrane that transports copper via the mitochondrial membrane.624 Copper can be sequestered and stored in the cell by metallothioneins (MTs), such as MT1, MT2, and MT3.625 MTs can also coordinate other metals, such as zinc, with some metal preferences. For example, MT3 has been shown to exhibit the highest affinity for copper within this class of proteins.626 Thus, balancing the expression of different MTs provides cells with the ability to control the levels of copper and zinc in the cytosol. Interestingly, copper(II) can catalyze the oxidation of GSH to glutathione disulfide (GSSG).627 The rate of copper-catalyzed oxidation of GSH to GSSG has been reported to be much lower than that of copper(II) complex formation with GSH, meaning that complexes between copper(II) and GSH can potentially exist in the cell before oxidation to GSSG occurs. In addition, GSH can form complexes with copper(I) to form tetranuclear [Cu4(GS)6] complexes, limiting free aquacopper(I) in the cell.628 Taken together, GSH can limit the concentrations of free copper(I) and copper(II) in the cell. Importantly, copper(I)/copper(II) redox cycling and ROS formation are influenced mainly by GSH and cysteine levels in the presence of hydrogen peroxide, suggesting that GSH and cysteine are important for buffering copper toxicity in the cell.629 Copper ions may also act as a catalyst for other chemical reactions in the cell in the presence of GSH, notably in mitochondria.630 Since copper(II) can participate and promote redox reactions in the cell, specific chaperones have evolved to regulate the pool of available chemically reactive copper. For example, specific chaperones including antioxidant 1 copper chaperone (ATOX1), also called Hah1,631 regulate copper(I) distribution in the cell, deliver the metal to copper-dependent enzymes and transfer it to ATP7A/B.632−634 The copper chaperone for superoxide dismutase (CCS)635 transfers the metal to SOD1, which unlike SOD2 predominantly localizes in the cytosol and contains a copper–zinc binuclear catalytic site responsible for the disproportionation of superoxide radicals. However, the role of zinc was not found to be critical for the enzymatic activity of superoxide dismutase under acidic conditions.636
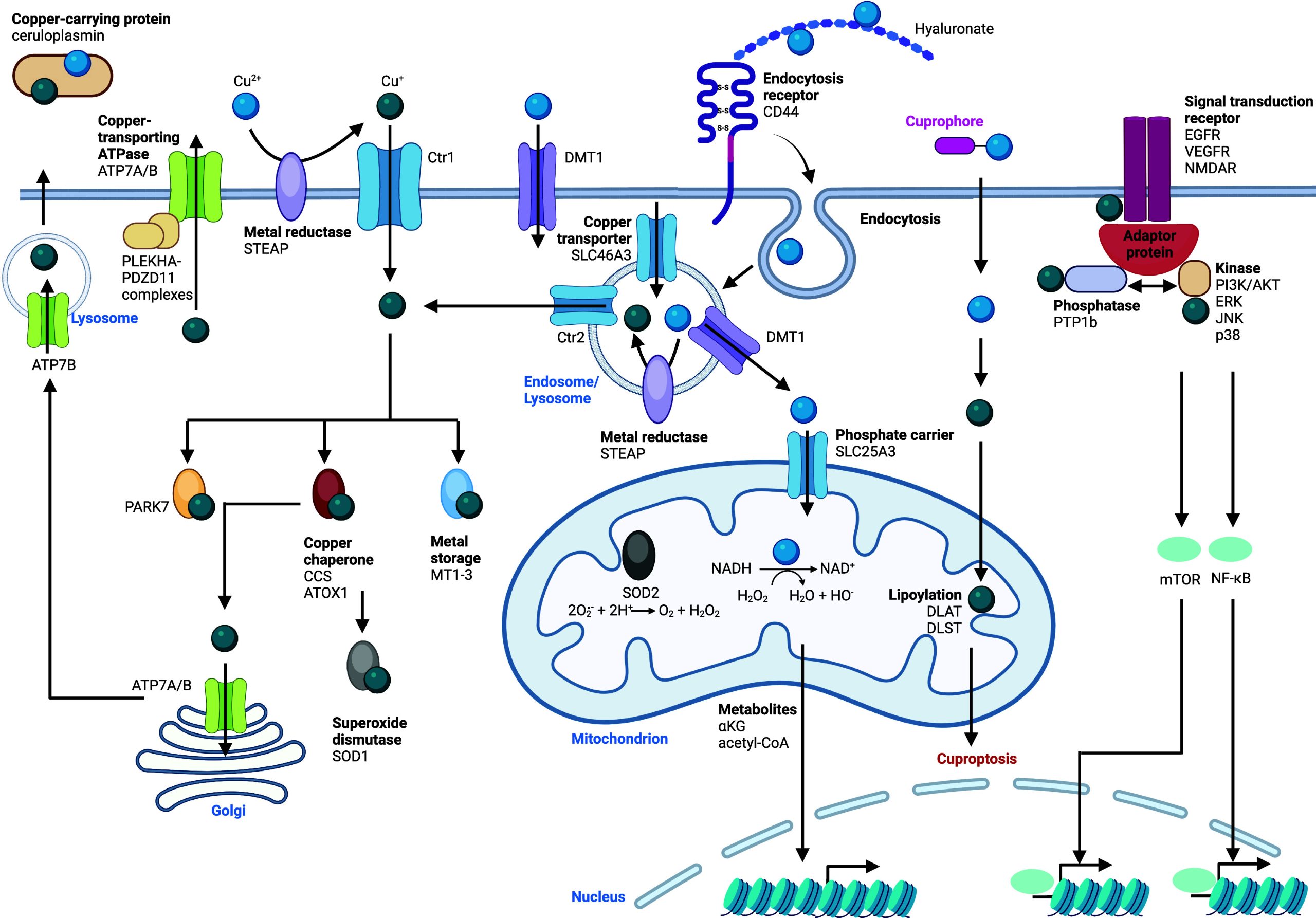
Cellular Functions of Copper
In mitochondria, copper is essential for several processes. It is involved in cuproproteins, enabling electrons to shuttle through the ETC. It plays an integral role in cytochrome c oxidase (also termed complex IV or COX), where three copper ions are coordinated inside the protein complex.637,638 This complex, which resides in the inner mitochondrial membrane, is the last enzyme of the ETC,639 which produces an electrochemical proton gradient that drives the synthesis of ATP. Mechanistically, copper is delivered to cytochrome c oxidase assembly protein (Sco1) by the copper chaperone COX17.640,641 Then, the copper chaperones Sco1 and Sco2 deliver copper to cytochrome c oxidase.642,643 Copper is also required for the activity of the Parkinson disease protein 7 (PARK7), which detects oxidative stress and resides in mitochondria, the nucleus, the ER, and the cytosol.644
In mitochondria, copper(II) has been detected in inflammatory macrophages and found to act as a Lewis acid, activating hydrogen peroxide and catalyzing the oxidation of NADH into NAD+ (see Figure IIIc). This chemical reaction has been shown to drive metabolic and epigenetic programming of inflammation in macrophages.13 Interestingly, activation of other immune cells including dendritic cells, and acquisition of a pro-metastatic DTP cancer cell state, have been shown to be driven by a similar copper-dependent chain of event.13 Whether this reaction is assisted by a protein or solely relies on free copper(II) remains unknown. Given that imidazole further increases the rate of copper-catalyzed NAD(H) redox cycling, it is conceivable that this reaction is assisted by a mitochondrial protein. In addition, copper is involved in several cell signaling pathways, acting as a component of several proteins, including kinases, phosphatases, and adapter proteins,645 impacting JNK,646 NF-κB,646−648 mTOR, and MAPK signaling pathways.649,650 In these cases, copper acts by means of metalloallostery promoting functional enzyme folding.651 For instance, copper can activate MAPK signaling, whereas disruption of the cellular copper import was found to reduce kinase activity. The MAPK kinase MEK1 binds two copper ions with high affinity, enhancing its catalytic activity.652 The copper chaperone CCS facilitates transfer of copper to MEK1 and MEK2, enhancing kinase activity.653 In addition, the copper chaperone ATOX1 is required in MAPK signaling in melanoma cells.654 Another study showed that casein kinase 2 (CK2) also requires copper for its catalytic activity.655 UNC51-like kinase-1 (ULK1) and ULK2 act downstream of mTOR and are activated by copper, with implications in adenocarcinoma.656 Copper can also directly activate PI3K/AKT signaling,657 however, explicit mechanisms at work remain incompletely understood. These studies support a functional role for copper in signaling pathways activated in cancer and inflammation. Designing selective inhibitors of these proteins or hijacking copper represents an appealing therapeutic angle. Copper overload in the cell has been linked to increased aggregate formation between lipoylated proteins via disulfide bond formation catalyzed by copper, predominantly occurring in mitochondria. Elesclomol (Table 7) has been shown to act as a cuprophore increasing intracellular copper levels, promoting this phenotype and leading to a form of cell death called cuproptosis.658 Finally, copper has also been reported to play a role in lipolysis by inhibiting the activity of the cyclic adenosine monophosphate (cAMP)-degrading phosphodiesterase 3B (PDE3B) via direct binding to the top cysteine residues of the enzyme in a mouse model of dysregulated copper homeostasis.659 Manipulating copper homeostasis may therefore be exploited for therapeutic intervention.
Copper Signaling and Diseases
Copper overload diseases with genetic background have been documented.660 MD661 is an X-chromosome linked recessive disorder with mutations in ATP7A. WD662 is an autosomal recessive disorder linked to mutations in ATP7B.663−666 MD is characterized by copper deficiency in specific tissues, leading to growth and developmental defects, whereas copper buildup is found in WD primarily in brain and liver, impacting behavior and causing digestive disorders. These diseases highlight the central role of cellular copper homeostasis in human physiology. MD is clinically managed by copper supplementation, including subcutaneous copper injections,667 elesclomol and L-DOPS (Droxidopa)668 (Table 7) treatment has been explored to rebalance copper distribution. In addition, metal chelators can be used to alter copper availability,669 and WD is treated using copper chelators, such as d-penicillamine, tetrathiomolybdate (TTM), trientine,649 and ATN-224,670 among others647,649 (Table 7).
Table 7: Regulators of Copper Signaling13,592,646−650,658,668,670,671,676,677,684,686−688,691,693,696−702,703704705−713,793,794
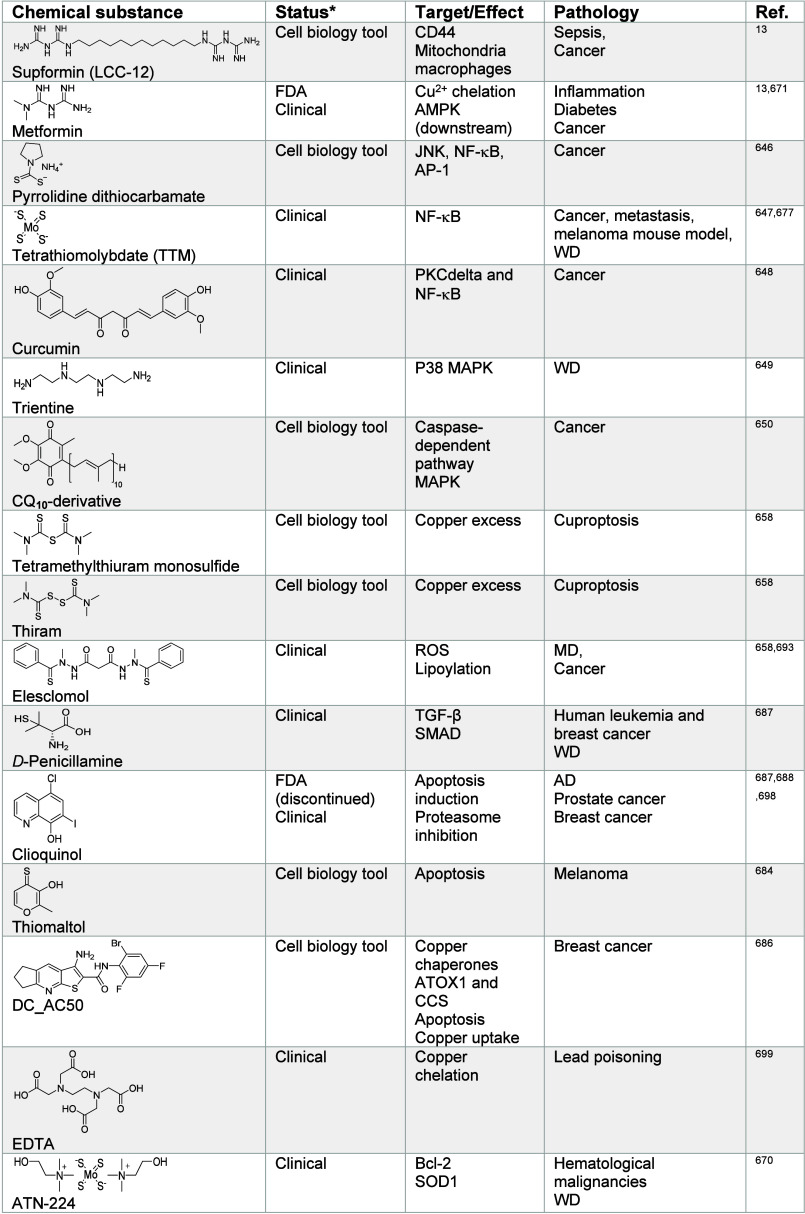
Copper Signaling in Immunity and Inflammation
Copper levels have been reported to be increased in inflamed malignant tissues and a STEAP4-IL17-dependent signaling axis has been documented in this context.616 Pro-inflammatory macrophages and cancer cells acquiring a pro-metastatic drug tolerant mesenchymal state are characterized by upregulation of CD44, increase of copper uptake, and a higher load of mitochondrial copper(II). Selective inactivation of mitochondrial copper(II) with small molecules such as metformin, identified by phenotypic screen, and the rationally designed dimer of biguanides supformin (LCC-12) (Table 7), were found to interfere with NAD(H) redox cycling and to antagonize specific epigenetic and transcriptional programs, notably those underlying inflammation.13,487 Interestingly, the basic pH of the mitochondrial matrix (i.e., proton gradient), but not mitochondrial copper(II), was found to drive the accumulation of biguanides in mitochondria, indicating that it is the organelle physiology that defines the subcellular localization of these biologically active compounds as opposed to the mechanistic target (i.e., copper(II)), emphasizing the value of phenotypic screen and target identification studies. Remarkably, copper(I) chelators and the copper chelator trientine, which accumulates in the nucleus, were found to be biologically inactive against pro-inflammatory macrophages. Pharmacological inactivation of mitochondrial copper(II) blocked immune cell activation, reducing acute inflammation in disease-relevant settings, and interfered with the acquisition of a DTP cancer cell state.13,671 Targeting mitochondrial copper(II) represents a powerful strategy to control the fate of immune cells for the clinical management of acute inflammation. It is not yet clear whether chronic forms of inflammation and autoimmune diseases are reliant on similar mechanisms and whether such a therapy can be exploited in these contexts. In COVID-19 patients, cytokine release syndrome characterizes the most severe cases.672 Remarkably, patients with diabetes undergoing metformin (Table 7) treatment exhibited less severe symptoms of COVID-19.673 Furthermore, COVID-19 patients were found to upregulate hyaluronan biosynthesis in the lung,448,449 supporting the contention that CD44/hyaluronan-mediated copper uptake drives immune cell activation in the context of acute inflammation. Thus, mitochondrial copper(II) inactivation with small molecules, such as supformin (Table 7) holds great promise for the clinical management of acute inflammation and sepsis.
Copper Signaling in Cancer
Copper has been implicated in cancer progression674 and copper chelators are being developed to specifically target cancer cells.675 Although such approaches are appealing, additional mechanistic rationales are needed to explain why specific copper chelators can target a cell niche in cancer. Mitochondrial copper(II) has been shown to regulate EMT and acquisition of a DTP cancer cell state.13 It is not clear whether controlling cancer cell identity through mitochondrial copper(II) inactivation can prevent metastasis in vivo or resensitize cancer cells to standard-of-care antiproliferative drugs. Nevertheless, compounds such as supformin (Table 7) represent valuable tools to perturb and dissect the biology of cancer cells that may be exploited for therapeutic purposes. An interesting study showed that the copper(II) chelator tetraethylenepentamine (TEPA) (Table 7) reduced the EMT signature in triple-negative breast cancers in mice and lead to a reduction of lung metastasis in this model.676 Whether TEPA chelates copper outside or inside of the cell and its subcellular distribution remain to be elucidated. The role of copper in MAPK signaling has direct implications in cancer, particularly in melanoma and thyroid cancers, where V600E mutations of v-raf murine sarcoma viral oncogene homologue B1 (BRAF) have been observed. BRAF(V600E) phosphorylates MEK1/2, which can then phosphorylate the ERK1/2 kinases, thereby activating MAPK signaling. The copper chelator tetrathiomolybdate (TTM) (Table 7), which is used for the treatment of WD, has been shown to reduce melanoma growth in mouse models.677,678 BRAF can be directly targeted and inhibited by vemurafenib (Table 7), and the cytostatic activities of copper chelators have been shown to contribute to their antineoplastic properties.677 Interestingly, pharmacological inhibition of Bcl-2 showed synergistic effects with TTM in BRAF(V600E) melanoma, inducing apoptosis.679 It will be important to dissect which copper-dependent pathway is impaired in these settings. In another study, TTM administration was found to reduce ceruloplasmin levels and the number of VEGFR2+ endothelial progenitor cells in the tumor microenvironment in triple-negative breast cancer patients with favorable survival enhancement.680 In addition, the same study showed reduced lung metastasis in preclinical mouse models of triple-negative breast cancer. Taken together, this work illustrates the value of copper chelation therapy and how it can be used in combination with other anticancer drugs for therapeutic benefits. Disulfiram (Table 7) has been reported to effectively kill human glioblastoma cell lines.681 In combination with copper, disulfiram has been shown to be effective against CSCs. It caused increased ROS production, subsequent inhibition of aldehyde dehydrogenase (ALDH) and NF-κB signaling.681 Interestingly, docetaxel-resistant prostate cancer cells were shown to express increased levels of ATP7B. Interestingly, these cells were resensitized to docetaxel upon treatment with disulfiram and copper.682 These promising findings warrant further examination.683 The ionophore thiomaltol (Table 7) has also been shown to exhibit promising cytotoxic effects in melanoma cells in combination with copper supplementation.684 The small molecule pyrrolidine dithiocarbamate646 (Table 7) is a copper chelator that was shown to affect NF-κB, JNK, and activating protein-1 (AP-1) signaling in various cancer cell lines, providing the basis for new therapeutic strategies. In addition, the copper chaperone ATOX1 has been identified as a potential prognosis marker in estrogen receptor-positive subtypes and also for early stage breast cancers.685 Additional work is needed to understand the functional role of copper homeostasis in this context to enable the development of copper-based therapeutic strategies. To this end inhibitors of copper-trafficking proteins such as 3-amino-N-(2-bromo-4,6-difluorophenyl)-6,7-dihydro-5H-cyclopenta[b]thieno[3,2-e]pyridine-2-carboxamide (DC_AC50) and its derivatives, which inhibit ATOX1 and CCS, were found to inhibit cancer cell proliferation686 (Table 7). The ionophore clioquinol687,688 (Table 7) has been shown to exhibit promising activity against prostate cancer in mice.689 However, this compound can form complexes with zinc and copper, making it challenging to assign an explicit MoA in this context.690 A screen for small molecule inhibitors of adenocarcinoma cell proliferation identified SOD1 as a promising target with the discovery of the SOD1 inhibitor lung cancer screen 1 (LCS-1)691 (Table 7). Furthermore, specific inhibitors against SOD1, such as LD94692 and LD100692 (Table 7), have also been reported to directly bind to copper in the active site of the enzyme.692 In addition, elesclomol has been selected to undergo clinical trials for the treatment of melanoma and may hold potential as a new anticancer therapeutic.658,693 Although elesclomol has been reported to induce cuproptosis, causing aggregation of lipoylated proteins via disulfide bond formation, it is not clear how elesclomol exerts its activity under these conditions.658 Investigating copper levels and oxidation states in specific cell organelles, as well as quantifying GSH levels is warranted to illuminate how manipulating copper homeostasis could be exploited for therapeutic benefits.694,695 Recent work has shown that copper-depleting nanoparticles can sequester copper, reducing tumor growth in mice.696,697 Such nanoparticles thus provide an interesting starting point to develop anticancer therapies based on copper depletion.
Zinc Signaling
Regulation of Zinc Homeostasis
Zinc is found as a +2 oxidation state in the cell, where it is generally considered a redox inactive metal. Yet, interactions with cysteines can confer a redox activity to zinc ions in the cell.714,715 Zinc is taken up into the cell by specific importers such as ZIP1–6, ZIP8, ZIP10, ZIP14, and TRPM7716−718 (Figure ). Other ion channels that have been characterized for calcium import as well as DMT1 and Ctr1 have also been reported to regulate the uptake of zinc ions.719,720 This illustrates the lack of selectivity of these channels and transporters for a given metal. Several organelle-specific zinc transporters have been documented for the import of zinc into mitochondria, the Golgi apparatus, the ER, the endolysosomal compartment and the nucleus, all belonging to the zinc transporter (ZnT) family of proteins717,718 (Figure ). Importantly, ZnT1 and ZnT10 have been described to reside in the cell membrane, where they can mediate the export of zinc from the cell. Interestingly, ZnT1 has been described as a zinc/calcium antiporter, supporting the notion that zinc can directly contribute to the control of calcium levels in the cell.721 Intracellular zinc levels can also be controlled by storage proteins including MTs,625,722,723 from which the metal can be released in a manner that is dependent on the reducing environment of the cell.715 It has also been reported that zinc ions can bind to and induce a conformational change of ZIP4, leading to endocytosis of the complex.724 Transporters for zinc into and from various cell organelles have been identified, including ZnT2/4 for zinc import into the endolysosomal compartment and ZIP3/8 for zinc translocation from these organelles (Figure ). ZnT5/6/7 transport zinc into the Golgi apparatus and ZIP7/9/11/13 were reported to export zinc from the Golgi apparatus. For the ER, ZnT5/6/7 have been characterized as zinc importers and ZIP7/9/13 as exporters. ZnT2/9 have been shown to mediate zinc uptake into mitochondria, whereas ZnT1 can import zinc into the nucleus and ZIP7 translocates the metal from the nucleus to the cytosol. Zinc is a key component of metal response element transcription factor-1 (MTF-1), which controls the transcription of ZnT1 and MTs, forming a positive feedback loop that regulates zinc levels in cells.725 Little is known about cellular zinc chaperones. A family of COG0523 zinc chaperone proteins conserved across vertebrates, which was named zinc-regulated GTPase metalloprotein activator (ZNG1) family, has been reported.726 ZNG1 proteins transfer zinc to methionine aminopeptidase 1 (METAP1) and ZNG1 knockdown resulted in the impairment of cellular zinc homeostasis and mitochondrial dysfunction.
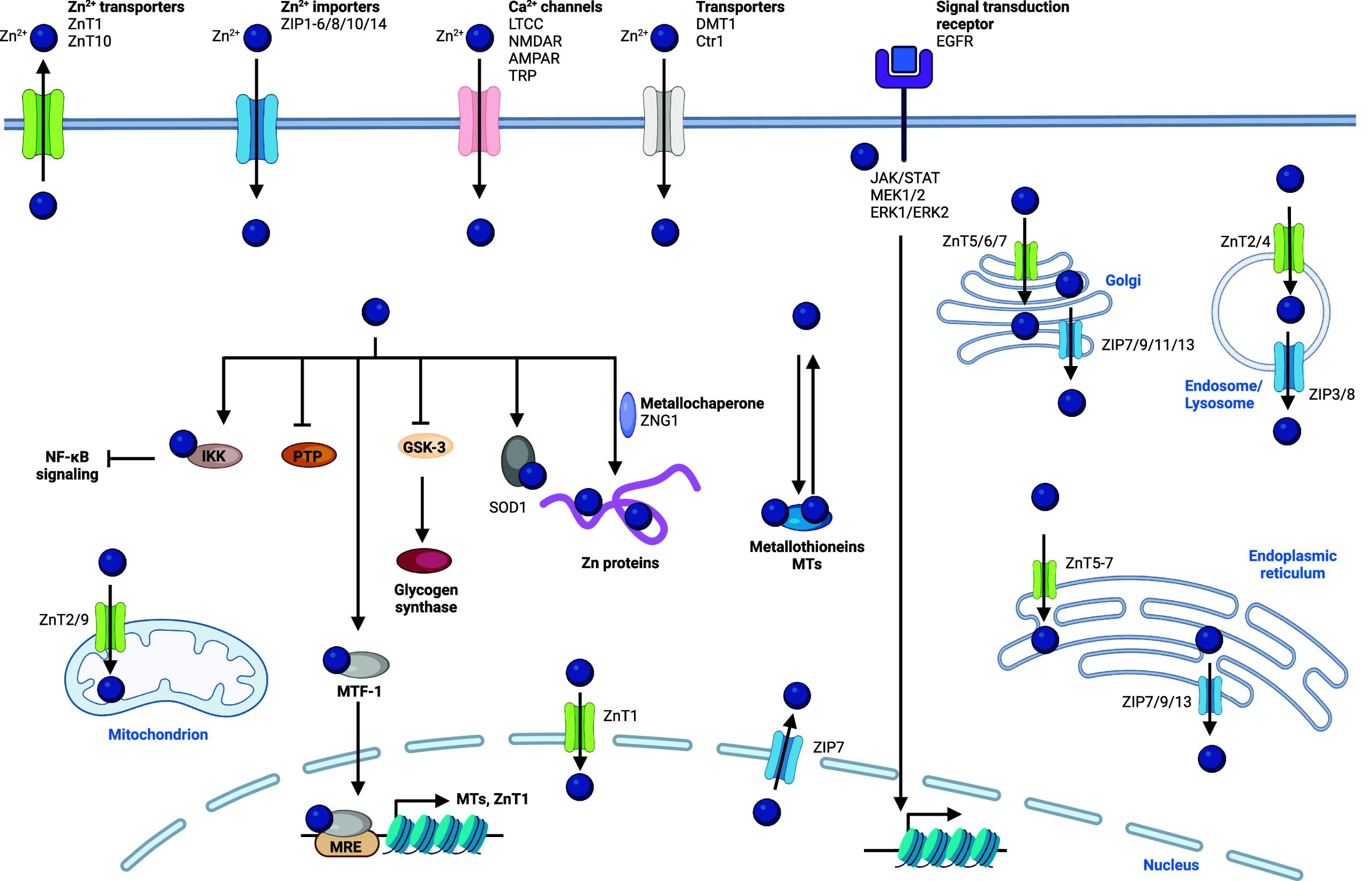
Cellular Functions of Zinc
The general high affinity for zinc binding to its biological substrates confers this metal cell signaling capacity by competing out other metals from biomolecules. For example, zinc has been shown to influence calcium homeostasis.727,728 Zinc is involved in cell proliferation and growth, being an important cofactor of TFs, potentially also acting through hormonal regulation of cell proliferation.729 Zinc is an important cofactor of zinc-finger proteins (ZNFs), constituting a large group of proteins.730 Roles of these proteins are widespread, including transcriptional regulation via ZNFs that are TFs, DNA repair, and signal transduction among others. Zinc is thus a crucial component of proteins that interact with nucleic acids. Zinc is important for the activity of many TFs, notably those involved in TGF-β and JAK/STAT signaling.730−732 The importance of ZIP6 in STAT signaling, which involves zinc, has been documented.733,734 Zinc and vanadium can also activate EGFR signaling and the Ras-dependent activation of MEK1/2 and ERK1/2 signaling by affecting the phosphorylation status of several components of the signaling cascade,735 although the exact mechanism orchestrating this is yet to be elucidated. Evidence for a role of zinc signaling in disease has been reported in cancer and inflammation.736 Interestingly, zinc has been shown to directly inhibit IκB kinase β (IKKβ) activity upon binding to a specific site located in the kinase domain of the protein,737 leading to a downregulation of NF-κB signaling. Similar effects of zinc were observed for protein kinase Cδ,738 indicating that zinc controls the activity of various protein kinases in the cell, affecting a multitude of pathways. Zinc has also been shown to impact stem cell pluripotency, shaping cell fate.739 A study showed that zinc is required for human embryonal development during egg activation, where a so-called zinc spark has been observed.740 Interestingly, intracellular zinc chelation led to a switch from a meiotic cell into a mitotic cell, suggesting that zinc is required for epigenetic control. Importantly, zinc is found in the active site of HDACs.741,742 Histone acetylation plays an important role in the epigenetic control of gene expression and can determine cell fate decisions. Taken together, these results indicate that zinc is involved in the control of cell identity. Zinc is also a component of SOD1, which is mainly found in the cytosol, and thus may play a role as a regulator of radical superoxide levels and hydrogen peroxide levels in the cell, potentially impacting oxidative stress and other ROS-dependent cell signaling pathways.
Zinc Signaling and Diseases
Zinc Signaling in Cancer
Zinc is widely implicated in transcriptional activation via ZNFs743 and in various signaling pathways in cancer.744 For example, the zinc finger protein p66β is a coactivator of the zinc finger transcription factor Snail, which plays crucial roles in the regulation of cell plasticity during development and in cancer.745 Cancer cell plasticity can give rise to a cell state refractory to standard-of-care treatments, constituting a drug-tolerant persister state. Interestingly, a study showed that KS10076 (Table 8), which can chelate copper, zinc, iron, and manganese, can eradicate ALDH1+ CSC subpopulations, characterized by a drug-tolerant cell state.746 The lack of metal specificity, however, limits mechanistic insights and raises the question of whether a specific role of zinc dominates this phenotype or whether altering homeostasis of other metals impairs specific cancer cell subpopulations. The putative role of zinc in cancer has prompted interest for the targeting of ZIP and ZnT family of transporters.717 ZIP7 plays a role in zinc transport from the ER, the Golgi apparatus, and the nucleus to the cytosol. Interestingly, protein kinase CK2 can trigger cytosolic zinc signaling by phosphorylation of ZIP7. Since tyrosine kinase signaling is dysregulated in various cancers, targeting ZIP7 selectively in cancer may confer therapeutic benefits.747 Small molecules such as 2-dimethylamino-4,5,6,7-tetrabromo-1H-benzimidazole (DMAT) and 4,5,6,7-tetrabromobenzotriazole (TBB)748 (Table 8) have been shown to reduce the proliferation of breast cancer cells, whereas the ZIP7 inhibitor NVS-ZP7-4 was found to be effective against T cell acute lymphoblastic leukemia.749 In addition, CK2 inhibitors like CX-4945748,750 (Table 8) can interfere with ZIP7 phosphorylation and have been developed for the treatment of estrogen receptor-positive breast cancer.748,750 Other anticancer strategies include targeting ZIP5,751 targeting ZIP6 with antibody conjugates,752−754 as well as other small molecules interfering with signaling pathways implicating ZIP9 and ZIP10 (Table 8). For instance, dutasteride755 (Table 8) has been developed for the treatment of bladder cancer, whereas bicalutamide756 is used for the treatment of melanoma and GSK690693757 (Table 8) for the treatment of osteosarcoma. The zinc chelator N,N,N′,N′-tetrakis(2-pyridinylmethyl)-1,2-ethanediamine (TPEN) (Table 8) has been shown to reduce the cell proliferation of breast and pancreatic cancer cells, suggesting that zinc is required for the maintenance of a highly proliferating cell state. Although not a suitable drug candidate, this study further validates zinc chelation as a possible anticancer therapy to target highly proliferating cells.758−761 Zinc chelation has also been investigated for therapeutic applications in other cancers, including small molecules (Table 8) such as clioquinol762−764 for prostate cancer, diethylenetriaminepentaacetic acid (DTPA) for breast cancer,758 and disulfiram for melanoma and liver cancer.765,766 Furthermore, zinc is required for the activity of several DNA repair/checkpoint proteins, for instance, apyrimidinic endonuclease, aprataxin PNK-like factor, and checkpoint protein with FHA and RING domains,767,768 protecting cells against DNA damage. Thus, imbalances in zinc homeostasis can impair these protective mechanisms and lead to genome instability with a potential impact on tumorigenesis.767 Since zinc is a crucial catalytic component of HDACs, changes in zinc homeostasis can potentially impact histone acetylation. Many HDAC inhibitors targeting the metal in the active site have been developed for therapeutic use, including vorinostat for cutaneous T cell lymphoma, panobinostat for multiple myeloma, and belinostat (Table 8) for the treatment of peripheral T cell lymphoma.769−772 However, HDAC inhibitors have not been used successfully against solid tumors. It has been reported that HDAC inhibitors can promote metastasis of solid tumors such as breast cancer.773,774 This can be explained by the fact that cell state transitions underlying the metastatic phenotypes require alterations of histone acetylation.11,445,775 Thus, HDAC inhibitors may adversely perturb epigenetic programs, regulating cancer cell proliferation and metastasis.
Table 8: Regulators of Zinc Signaling15,729,733,746,748−766,769−772,783,787−826
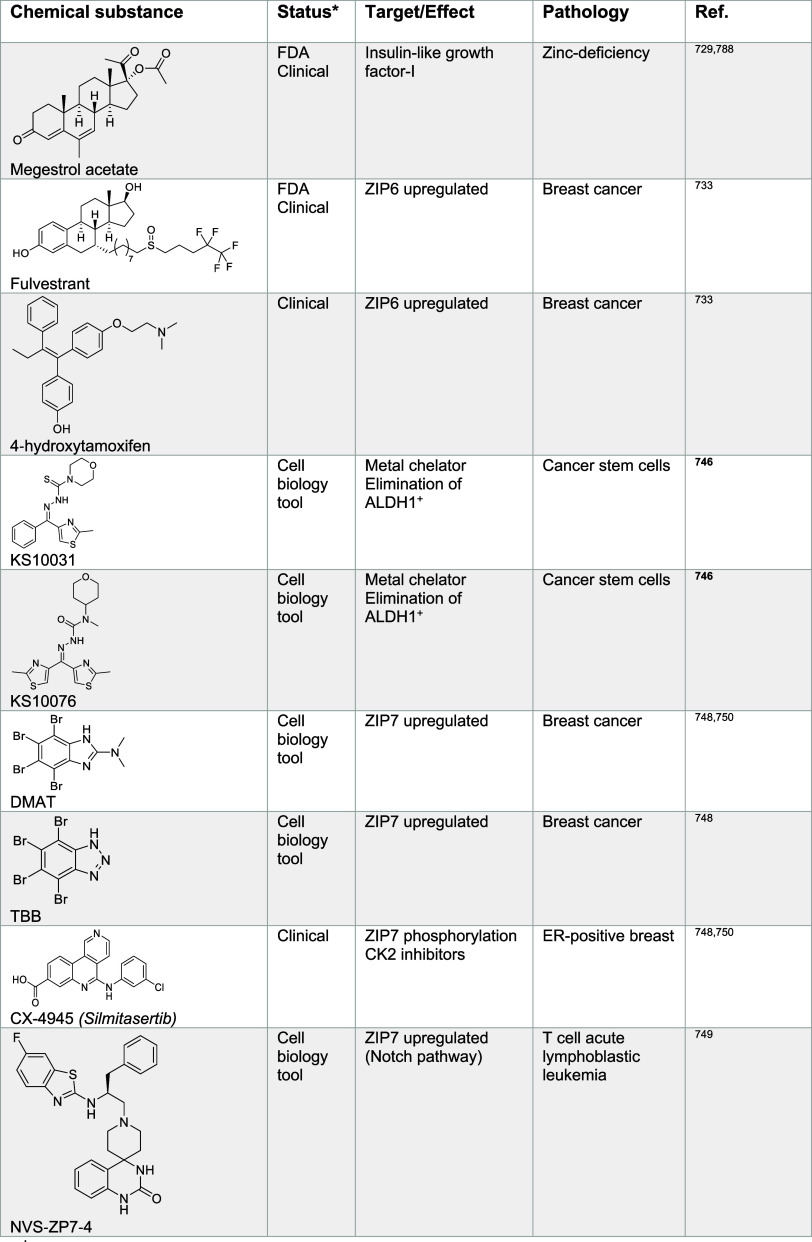
Zinc Signaling in Immunity and Inflammation
Zinc plays a crucial role in immunity.776 It is involved in ZNFs, which are important for immune cell function, and this includes several TFs.777−780 Furthermore, zinc transporters have been shown to be involved in innate immunity.719 Zinc generally represses NF-κB signaling, which plays important roles in immune cell activation.781 The inhibitory effect of zinc on NF-κB can potentially impact the activation of several types of immune cells. Thus, zinc supplementation or chelation could be exploited in specific pathological settings to modulate immune responses.737 Importantly, zinc signaling has been shown to be crucial for T and B cell activation.782 Interestingly, it has been shown that zinc can be supplied not only from extracellular sources, but also intracellular zinc is released from storage sites in immune cells during immune cell activation. For instance, MTs can stimulate zinc(II) release and activate CD4+ differentiation, which suggests that intracellular zinc reservoirs can be exploited to regulate cell state transitions.783 Interestingly, the small molecule 2,2′-dithiodipyridine (Table 8) can induce the release of zinc from MT, leading to increased CD4+ activation.783 Indeed, regulation of zinc homeostasis by MTs has been proposed to play a critical role in immune cell plasticity.722,784 Furthermore, free intracellular zinc has been described as a key determinant of immune cell plasticity in virtually all types of immune cells.785 Interestingly, upon T cell stimulation by cytokines, increases of zinc and calcium levels have been reported.786 Further investigation is needed to delineate the precise effect of zinc on cell signaling and cell plasticity in this context to be able to establish selective small molecule regulators of these processes. Zinc has been shown to exacerbate inflammation in viral liver disease. In this context, zinc has been shown to interfere with cytokine IFN-λ3 binding to interferon lambda receptor 1 (IFNLR1), which decreased antiviral activity and leads to increased viral replication in cells infected with influenza or hepatitis C virus.787 Thus, zinc chelation represents an interesting strategy to target chronic diseases linked to viral infection.
Cell Signaling of Other d-Block Metals (Cr, V, Mo, Co, Ni)
Other d-block metals including chromium, vanadium, cobalt, nickel, and molybdenum have been reported to play a role in the biology of the cell. In comparison to other metals, little is known about cellular homeostasis, roles in cell signaling, and implications in diseases. In this section, we document the biology of these metals and how this knowledge may be exploited for basic research and medical applications.
Regulation of Cellular Homeostasis
Mechanisms enabling the cellular uptake of chromium and vanadium have not been thoroughly documented. It may involve transporters previously described to regulate ion transport.827,828 Chromium has been reported to be taken up into the cell by Tf and TfR1-mediated endocytosis.829 In the cell, chromium and vanadium can coexist in various oxidation states. In bacteria, cobalt uptake, storage, and efflux have been documented in detail, but little is known in eukaryotic cells.830 Cobalt has been suggested to be taken up via divalent cation channels, which are not selective for a specific metal. In neurons, the same channels can mediate the uptake of cobalt, manganese, and calcium831 (Figure ). Cobalt is also taken up as vitamin B12 (also known as cobalamin, a coordination complex of cobalt) via the transcobalamin receptor.832,833 How cobalt is exported from cells is unknown. In plants, ferroportin has been reported to play that role,834 suggesting that this may also take place in human cells. Nickel is found in the cells as a +2 oxidation state, and regulation of its cellular uptake has not been documented in great detail. Nickel can be internalized via DMT1 and calcium channels835−838 (Figure ). In addition, artificial nickel particles can be taken up by macropinocytosis.839 How nickel is exported from the cell remains poorly understood. Molybdenum is taken up into the cell via ATP-dependent active transporters.840 Once in the cell, it can form a complex with pterin, termed molybdenum cofactor (Moco).841,842 Importantly, the biosynthesis of Moco requires iron, copper, and ATP. As a result, molybdenum homeostasis and its impact on cellular function are inherently linked to iron and copper metabolism.843
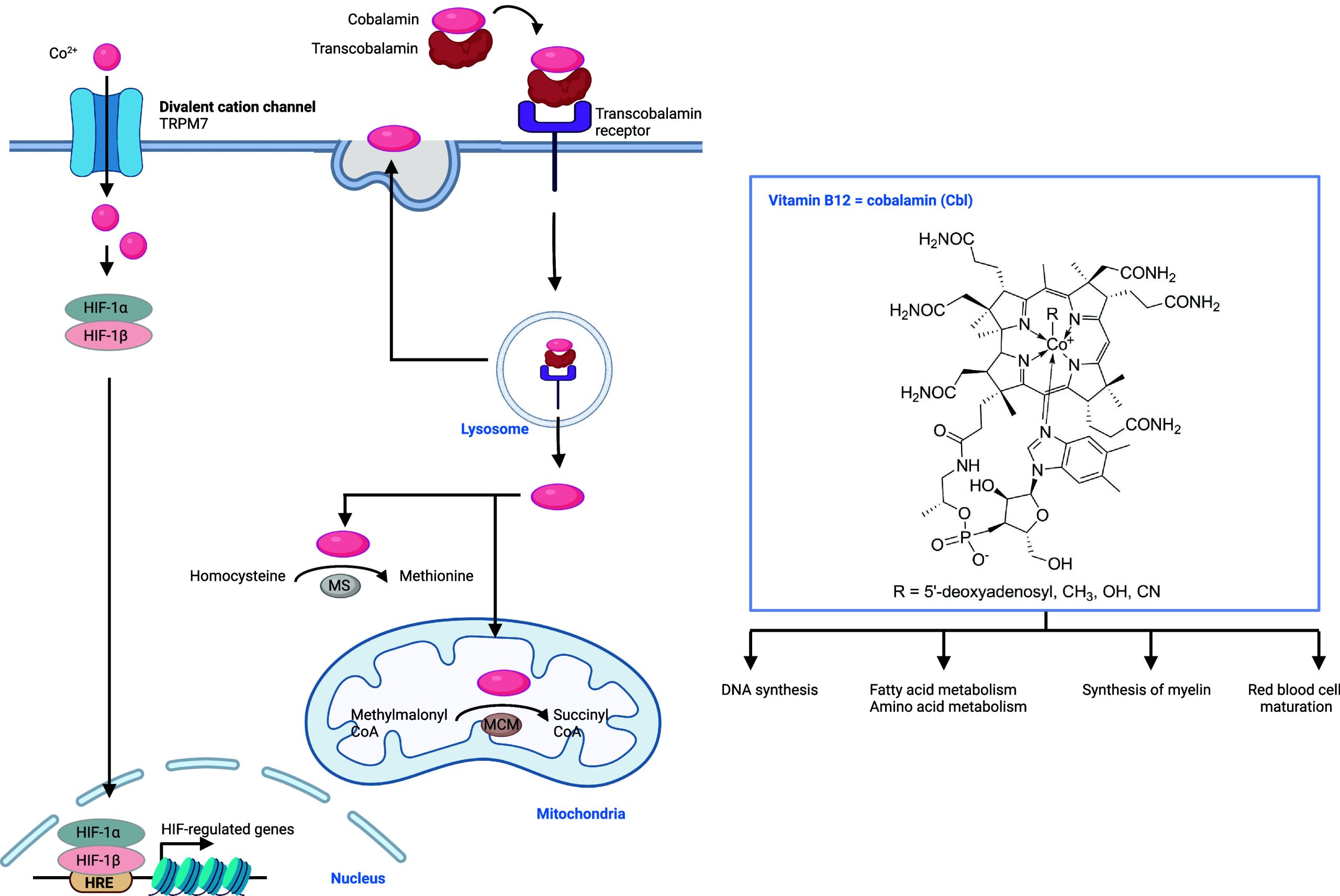
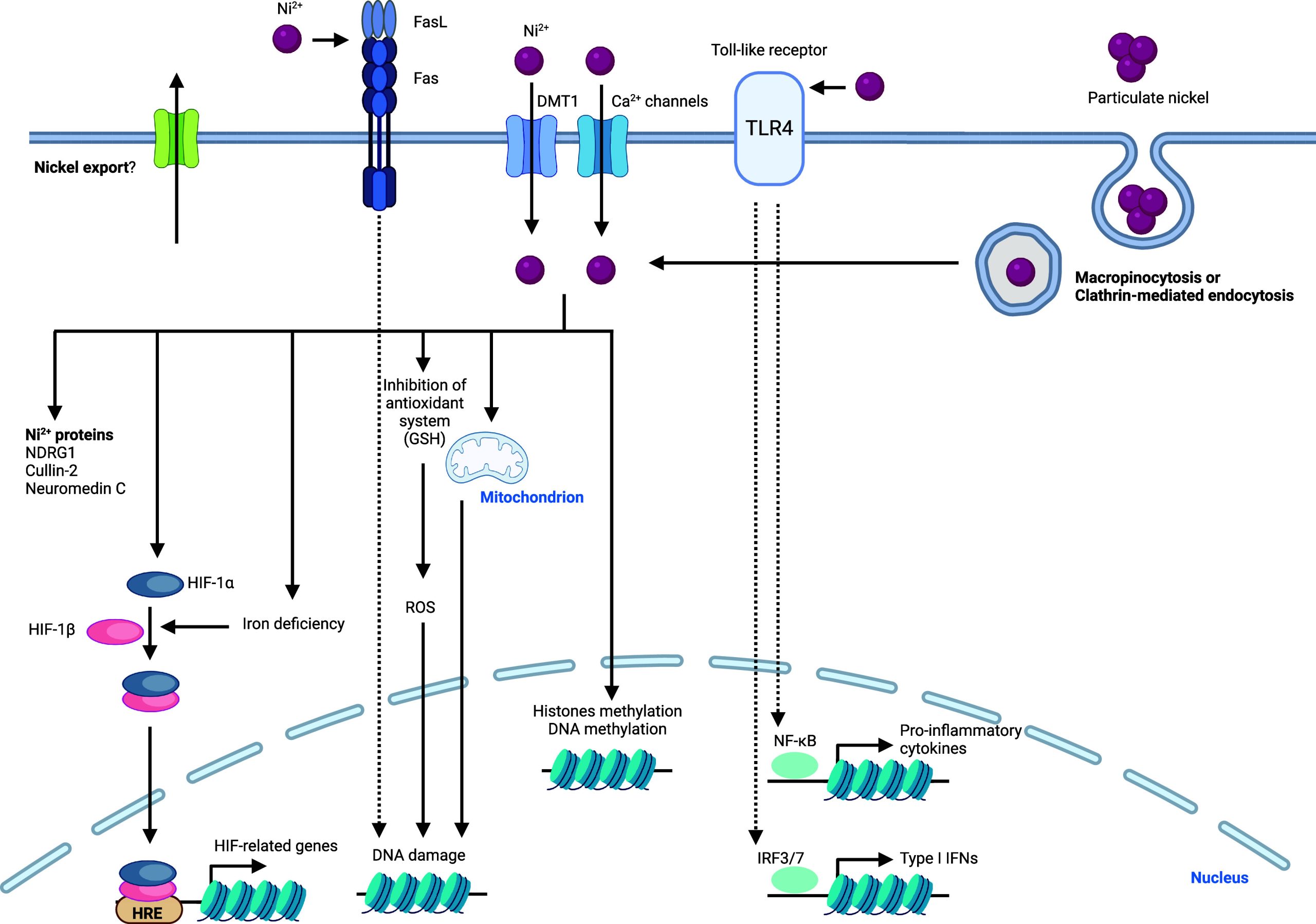
Cellular Functions of Other d-Block Metals
Vanadium can affect fatty acid metabolism, glycogen biosynthesis, and blood sugar levels.828,844 Although vanadium-dependent enzymes have been identified in bacteria, fungi, and plants, such as haloperoxidases and nitrogenases, vanadium-dependent enzymes have not been identified in higher eukaryotes to date.828,845 Chromium plays a role in carbohydrate, lipid, and protein biosynthesis846 (Figure ). This has been proposed to occur via chromodulin, a small polypeptide that can coordinate four chromium ions.847 Insulin-binding to the insulin receptor has been suggested to stimulate TfR1-mediated uptake of chromium, leading to internalization of chromium, allowing for sufficient amount of this metal to be imported such that four chromium ions can be coordinated by apochromodulin to form active holochromodulin.848,849 Holochromodulin then binds to the insulin receptor β subunit, which results in the activation of receptor tyrosine kinase and amplification of the insulin signal. Cobalt is part of the structure of vitamin B12, which is required for the activity of enzymes implicated in DNA biosynthesis, fatty acid metabolism, amino acid metabolism, synthesis of myelin, and maturation of red blood cells. Vitamin B12 is synthesized by some archaea and bacteria via a complex biosynthesis pathway that includes several cobalt chaperones.850 Cobalt is also required for the function of other noncorrin-cobalt-containing enzymes, including glucose isomerase, methylmalonyl-CoA carboxytransferase, aldehyde decarbonylase, and others.851 Thus, cobalt is involved in many metabolic processes. Similarly, nickel is incorporated into several nickel-dependent proteins, such as NDRG1, cullin-2, and neuromedin C. Nickel can induce DNA damage by inhibiting the antioxidant system, leading to decreased activity of catalase and SOD, and by promoting mitochondrial damage, causing an increase in mitochondrial 8-hydroxyguanin. Nickel stabilizes transcription factor HIF-1α and decreases cellular iron levels. Consequently HIF-1α and HIF-1β are translocated into the nucleus where they form a heterodimer, bind to HRE, and function as a TF.472,473 Since iron regulates HIF degradation,852 it is conceivable that nickel competes out iron from proline dioxygenases and consequently impacts HIF-1 regulation. Molybdenum-containing proteins have been identified, including mitochondrial amidoxime reductase, sulfite oxidase, xanthine oxidoreductase, and aldehyde oxidase.841,853 Two different types of cofactors that contain molybdenum have been reported, those containing iron and molybdenum on one hand and pterin-based molybdenum cofactors on the other hand.843
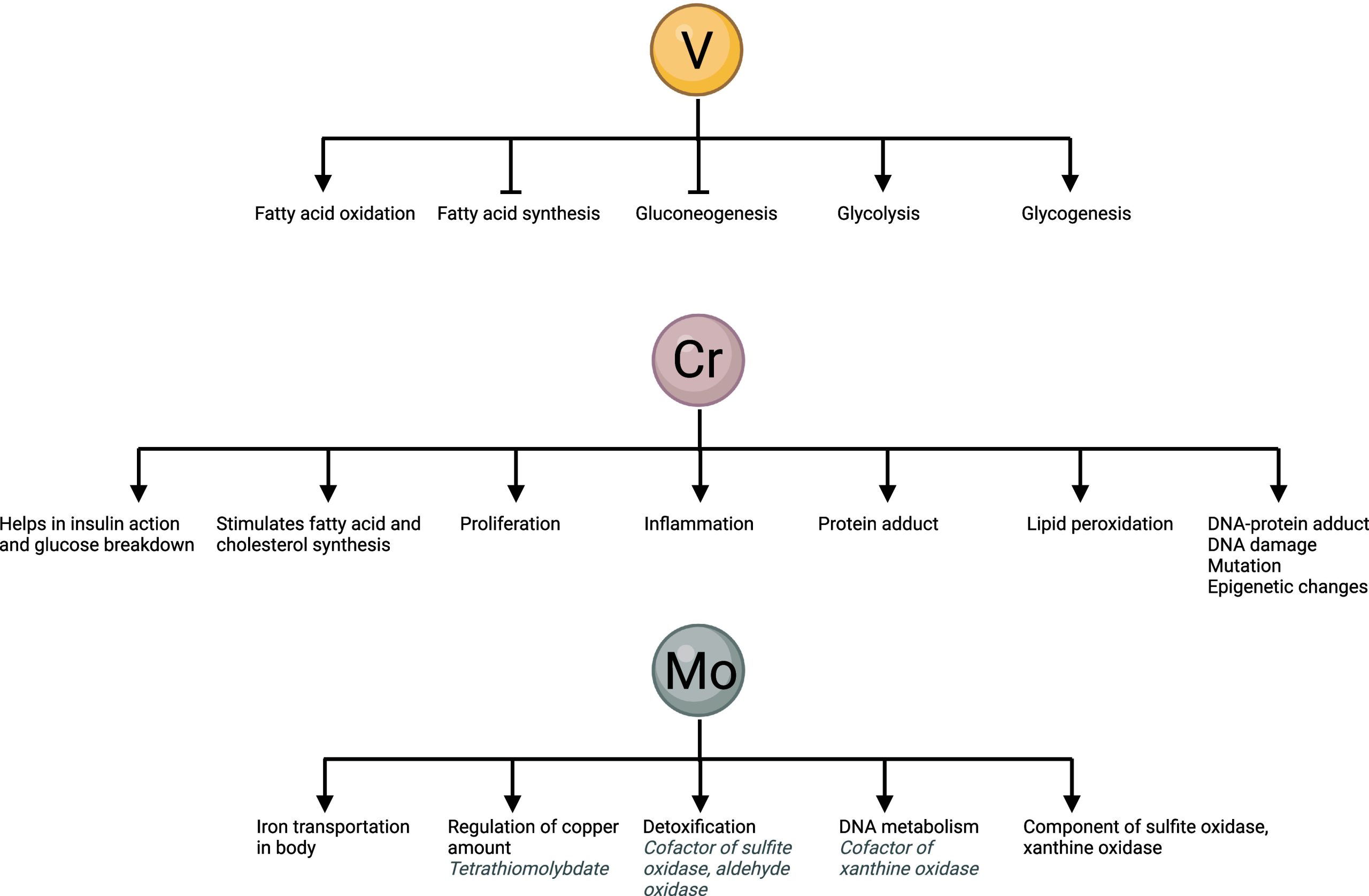
Other d-Block Metal Ion Signaling and Diseases
Other d-Block Metal Ion Signaling in Cancer
Vanadium has been shown to impact diabetes. Owing to the well-established increased glucose uptake that fuel cancer cell proliferation, it is conceivable that vanadium may also impact cancer.854 Thus, small molecules regulators of vanadium homeostasis and function may be exploited in this context.855 Vanadium, chromium, and nickel have also been described to be carcinogenic metals, and some evidence has emerged that these metals stimulate NF-κB signaling and cell cycle progression in cancer cells, although mechanisms at work remain poorly understood.856 High levels of vanadium and chromium have also been reported to increase the risk of colorectal cancer, lacking a mechanistic rationale.857 For example, it is not clear whether normal homeostasis and cell signaling involving these or other metals are altered upon overload with vanadium and chromium or whether this overload directly promotes undesired reactions contributing to the disease such as oxidative stress, altered mitochondrial metabolism or mutations in genomic DNA. In addition, higher levels of vanadium and chromium as well as other heavy metals have been identified in nonsmall cell lung cancer, suggesting that these metals could play a role in the etiology of the disease and/or contribute to cancer progression.858 Vanadium-containing compounds can inhibit several signaling pathways in cancer cells that include phosphorylation reactions in the signaling cascade, in particular MAPK signaling and PI3K/AKT signaling.859 Since these pathways can be upregulated in cancer cells, vanadium-containing compounds (Table 11) might find therapeutic applications. Chromium can cause DNA damage with interesting cytotoxic properties in cancer cells.860 This could potentially be exploited in cancer research and requires further studies. However, chromium itself has been reported to be carcinogenic, especially at higher exposures.861 Importantly, high concentrations of chromium and nickel in electronic nicotine delivery systems have been associated with increased lung cancer risk.862 Cobalt has been reported to stimulate PI3K/AKT signaling in oral squamous carcinoma cells.863 Nickel has been shown to have many effects on signaling in cancer cells.864 It can poison iron-dependent demethylases by replacing the redox-active iron catalyst, inactivating specific iron-dependent enzymes.865 Since these enzymes are involved in epigenetic regulation, nickel directly impacts the acquisition of distinct states of cancer cells. Nickel itself has also been shown to be carcinogenic and a driver of cancer progression.836
Other d-Block Metal Ion Signaling in Immunity and Inflammation
Vanadium-containing drugs, so-called vanodrugs,866 have been shown to exhibit an effect on inflammation by affecting NF-κB and TLR signaling. Although, whether these drugs impact vanadium homeostasis and cellular functions remain unclear and explicit MoA are yet to be elucidated.867 Treatment of macrophages with vanadate resulted in stimulation of NF-κB and JNK signaling, suggesting a pro-inflammatory effect of vanadate and highlighting an effect on the acquisition of a pro-inflammatory cell states of macrophages.868 In another study, the pro-inflammatory effect of vanadate has been demonstrated in bronchoalveolar lavage cells in clinical settings.869 Kinases, such as cytosolic protein tyrosine kinase and others,870 were shown to be inhibited by vanadium and vanadium-containing compounds, in particular vanadate- and polyoxovanadate-containing compounds (Table 11), which can thus broadly affect cell signaling pathways involving phosphorylation.871 Because of the effect of vanadium on phosphatases, vanadate has a positive effect on T cell activation via changes in tyrosine phosphorylation.872 Thus, interference with signaling in immune cells by vanadium can provide control over cell states. Chromium can affect T and B lymphocyte function, macrophage function, and alter cytokine production, impacting immune responses.873 Chromium can inhibit the phagocytotic function of macrophages.874 Whether specific signaling pathways impact cell states requires further investigation. Cobalt ions can activate TLR4 generating an inflammatory response.875 This can be attenuated in vitro by the small molecule CLI-095, which can have an effect on soft tissue necrosis and osteolysis875 (Table 9). A pro-inflammatory response upon exposure to cobalt ions has also been observed in other cell types, including neutrophils, monocytes, and epithelial cells of the gut and kidney.876 Indeed, cobalt has been reported to induce an inflammatory state in macrophages, but underlying mechanisms driving cell state transitions require further investigation.877 Nickel has been shown to act as a pro-inflammatory signaling element. Interestingly, zinc can inhibit nickel uptake and attenuate nickel-induced inflammation in THP-1 cells.835 Moreover, nickel promotes inflammatory responses by activating TLR4. The allergic and inflammatory reaction to nickel in some individuals has been explained with differential activation of specialized CD8+ cells.878,879 It is conceivable that nickel contributes to signaling pathways activating these cells, which require further studies. Nickel chelation has been successfully employed to treat chronic dermatosis, including compounds such as disulfiram, EDTA, and clioquinol880 (Table 10).
Table 9: Regulators of Cobalt Signaling875,890
Table 10: Regulators of Nickel Signaling835,836,880,907−913
Table 11: Regulators of Vanadium Signaling881−886,890−893,914−916,917918919−923,
Other d-Block Metal Ion Signaling in Other Diseases
Small molecules including EDTA, 2,3-disulfanylpropanol, and the clinically approved copper chelator d-penicillamine have been shown to reduce cellular vanadium levels881−886 (Table 11). Vanadate can also replace phosphate in cells, and the effect of vanadium on insulin metabolism has encouraged research in this area.887 Vanadium-containing compounds can also mimic insulin888,889 and therefore exert a positive effect in diabetes. Such compounds include vanadyl sulfate (VOSO4), bis(maltolato)oxovanadium(IV) (BMOV), and vanadyl-methylpicolinate complex (VO-MPA) among others890−893 (Table 11). These effects might also be translated to other clinical settings including cardiovascular diseases.894 The effect of chromium on amplifying insulin signaling also represents a promising line of research for the treatment of diabetes.895−897 Other small molecules that affect insulin-resistance in vitro including chromium complexes have been documented897−899 (Table 12) and represent an interesting starting point for the development of therapeutic strategies. Mutations in leucine-rich repeat kinase 2 (LRRK2) have been associated with the progression of PD. The cobalt-containing vitamin B12 is an allosteric inhibitor of LRRK2 and has been shown to reduce neurotoxicity in rodent models of PD900 (Table 9). In contrast, cobalt ions have been shown to be neurotoxic901 and to promote PD, presumably involving the production of ROS. However, explicit mechanisms are not fully understood.902 Nickel has been reported to drive cardiovascular and kidney diseases and lung fibrosis.903 The exact mechanisms underlying these effects remain to be characterized. Deficiency of the molybdenum cofactor Moco is found in a rare genetic disease that leads to early development defects and contributes to neurodegeneration.904 A treatment based on precursor Z/cPMP/fosdenopterin (Table 13) has been used for therapeutic intervention.842,905,906
Table 12: Regulators of Chromium Signaling924−931
Table 13: Regulators of Molybdenum Signaling841,905,906,932,933
Remarks
Metals exhibit a diverse range of chemistries including Lewis acid catalysis, redox, and supramolecular properties. The cell has evolved around these chemistries to mediate signal transduction while dealing with potentially detrimental reactions inherent to the reactivity of metals. This notion is best exemplified by the higher iron load required for cells to promote epigenetic reprogramming via oxidative demethylation of histone proteins, which concomitantly drives oxidation of lipids and confers vulnerability to ferroptosis.515 Thus, cells have developed machineries to repair oxidized membranes, enabling them to exploit iron to adapt to their environment.
Here, we have discussed the essential roles of metals in the biology of the cell. We have documented current knowledge on mechanisms regulating cellular metal homeostasis, including how metals are internalized by, distributed and stored within, and exported outside of the cell. We have described the multifaceted chemical roles of metals in transducing information inside the cell in normal physiology and how this regulation can contribute to diseases, in particular by promoting the acquisition of distinct cell properties and states. Based on their electron configuration, which defines their position in the periodic table, metals are found in the cell as crystalline forms or positively charged ions, having a propensity to act as counterions to balance charges in the cell, to promote conformational changes of biomolecules acting as cofactors that modulate function, or to mediate otherwise inefficient chemical reactions operating as catalysts.
Unlike organic biomolecules, such as proteins, nucleic acids, lipids, and glycans, metal ions are not synthesized in the cell but acquired from external sources such as nutrients. These species are smaller in size, conferring faster diffusion rates within the cell compared to biopolymers and exist in various oxidation states, being found either as free or bound species,216 making these chemical entities difficult to track within and outside of the cell. The literature clearly shows that multiple import and export mechanisms exist for metal ions and that these mechanisms are often not specific for a given metal. Furthermore, distinct metals can exhibit redundant properties. Thus, manipulating a given metal selectively in the cell to investigate its role in disease-relevant settings remains a challenging endeavor. Nature itself, which relies on fundamental principles of physical chemistry, has not been able to achieve complete selectivity for specific metals, which raises the question of whether we, the scientists, can invent new strategies and develop smarter chemistries to achieve selective control over metals in the cell. In other words, can we chemically target a given metal without affecting the biology of others? Can we manipulate a single function of a given metal selectively?
Our knowledge of the biology of metals is still in its infancy. For example, iron has been shown to fuel the activity of enzymes in the nucleus to promote epigenetic reprogramming and cell adaptation,446 yet it remains unknown how iron traffics to the nucleus where chromatin is localized. Copper(II) has been shown to catalyze NAD(H) redox cycling in mitochondria, enabling activation of immune cells in acute inflammation settings and to regulate the acquisition of a pro-metastatic state of cancer cells,13 but whether this is assisted by a protein stabilizing the transition state of this copper-catalyzed reaction remains unknown. Future efforts should be dedicated to identifying missing pieces in how cells control metal homeostasis. To this end, developing new probes and technologies to map out sites of action of metals in the cell, in an oxidation-state-specific manner, will be essential to our understanding of their roles in cell signaling. This may reveal new features that can be exploited for the development of small molecule modulators of cell signaling, either through direct metal targeting or alternatively acting upstream or downstream on associated signaling effectors. Another layer of complexity for the development of drugs involves our capacity to target specific tissues and specific cell types, in particular, the cells exhibiting a diseased state or more generally cells contributing to the disease or a pathological condition. How metal homeostasis is regulated at the level of specific tissues and between distinct cell types within a given tissue is complex and not fully understood, raising again the challenge of how to selectively target a metal ion signaling axis in a given cell type or tissue.
Discovery-driven approaches have been instrumental in delineating metal ion signaling pathways and the identification of related druggable targets. For example, Schreiber and Crabtree have illuminated calcium signaling partly by elucidating the MoA of complex biologically active small molecules, namely FK-506 and cyclosporin A,22−24,26 which has prompted interest into the development of small molecule-proximity inducers and given rise to the field of molecular glues.934 Notably, these compounds (Table 4) inhibit T cell activation by forming complexes with distinct immunophilins that bind to and inactivate the calcium- and calmodulin-dependent phosphatase calcineurin.22−24,26 These classes of compounds are potent immunosuppressors that have been used for the clinical management of organ transplants and other immune diseases. Importantly, calcium signaling is inhibited independently of direct calcium ion targeting. Similarly, studying the MoA of trichostatin A and trapoxin A (Table 8), Schreiber and co-workers have identified mammalian HDACs, contributing to the rise of epigenetics.15 Directly targeting zinc in the active site of HDACs using hydroxamic acid-containing small molecules has been shown to alter chromatin, impacting transcriptional programs and tumor progression. This pioneering work promoted the development of new anticancer therapeutics, demonstrating the druggable nature of this class of enzymes and raising the question of whether a given HDAC can be targeted selectively and at specific genomic loci.826,935 A similar rationale has been applied for the design of iron-dependent demethylase inhibitors, which can inactivate iron in the enzyme active site, with little specificity toward a given demethylase.531,936 Nevertheless, these studies emphasize the druggable nature of metalloenzymes targeting the metal active site. While targeting of free metal ions has been documented, notably with the use of the deferoxamine (Table 6) for the treatment of β-thalassemia and myelodysplastic syndrome, little is known about the sites of action of iron chelators in the cell and how, mechanistically, depleting iron confers therapeutic benefits with manageable off-target effects. In contrast, a small molecule screen coupled to molecular editing yielded small molecules, such as ironomycin, that directly target iron(II) in endolysosomes of DTP cancer cells and trigger ferroptosis in preclinical models of metastatic cancers.511,514,516 This work identified lysosomal iron as a druggable target in difficult-to-treat cancers. In another study, a phenotypic screen informed the design of the metformin dimer supformin capable of selective mitochondrial copper(II) targeting, antagonizing macrophage activation and preventing sepsis in preclinical models of acute inflammation.13 This work identified mitochondrial copper(II) as a driver of inflammation and a druggable target.
These studies have provided solid evidence that direct targeting of specific metals with small molecules, whose increased uptake is causal to the disease, can be achieved and confer therapeutic benefits. Hypothesis-driven approaches together with the rise of artificial intelligence and machine learning may represent an effective route forward, potentially enabling faster and more systematic production of biologically active small molecules and metal ion signaling drugs.937,938 It is not clear, however, whether these strategies can surpass discovery-based phenotypic approaches.36 Small molecules have illuminated cell signaling pathways involving metals and demonstrated that some of these processes can be targeted with therapeutic value. While no general rule can be established at this point and much remains to be learned in this area, it is fair to say that metal ion signaling regulators are undruggable until someone drugs them.
References
- E. L. Tatum, G. W. Beadle. Genetic Control of Biochemical Reactions in Neurospora: An ″Aminobenzoicless″ Mutant.. Proc. Natl. Acad. Sci. U.S.A., 1942. [DOI | PubMed]
- L. Pauling, H. A. Itano. Sickle cell anemia a molecular disease.. Science, 1949. [DOI | PubMed]
- R. E. Franklin, R. G. Gosling. Molecular configuration in sodium thymonucleate.. Nature, 1953. [DOI | PubMed]
- J. D. Watson, F. H. Crick. Molecular structure of nucleic acids; a structure for deoxyribose nucleic acid.. Nature, 1953. [DOI | PubMed]
- F. J. Mojica, C. Diez-Villasenor, J. Garcia-Martinez, E. Soria. Intervening sequences of regularly spaced prokaryotic repeats derive from foreign genetic elements.. J. Mol. Evol., 2005. [DOI | PubMed]
- M. Jinek, K. Chylinski, I. Fonfara, M. Hauer, J. A. Doudna, E. Charpentier. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity.. Science, 2012. [DOI | PubMed]
- L. Cong, F. A. Ran, D. Cox, S. Lin, R. Barretto, N. Habib, P. D. Hsu, X. Wu, W. Jiang, L. A. Marraffini. Multiplex genome engineering using CRISPR/Cas systems.. Science, 2013. [DOI | PubMed]
- G. A. Newby, J. S. Yen, K. J. Woodard, T. Mayuranathan, C. R. Lazzarotto, Y. Li, H. Sheppard-Tillman, S. N. Porter, Y. Yao, K. Mayberry. Base editing of haematopoietic stem cells rescues sickle cell disease in mice.. Nature, 2021. [DOI | PubMed]
- J. B. Gurdon. The developmental capacity of nuclei taken from intestinal epithelium cells of feeding tadpoles.. J. Embryol. Exp. Morphol., 1962. [DOI | PubMed]
- K. Takahashi, S. Yamanaka. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors.. Cell, 2006. [DOI | PubMed]
- M. A. Nieto, R. Y. Huang, R. A. Jackson, J. P. Thiery. Emt: 2016.. Cell, 2016. [DOI | PubMed]
- A. Perez-Gonzalez, K. Bevant, C. Blanpain. Cancer cell plasticity during tumor progression, metastasis and response to therapy.. Nat. Cancer, 2023. [DOI | PubMed]
- S. Solier, S. Müller, T. Cañeque, A. Versini, A. Mansart, F. Sindikubwabo, L. Baron, L. Emam, P. Gestraud, G. D. Pantos. A druggable copper-signalling pathway that drives inflammation.. Nature, 2023. [DOI | PubMed]
- J. E. Brownell, J. Zhou, T. Ranalli, R. Kobayashi, D. G. Edmondson, S. Y. Roth, C. D. Allis. Tetrahymena histone acetyltransferase A: a homolog to yeast Gcn5p linking histone acetylation to gene activation.. Cell, 1996. [DOI | PubMed]
- J. Taunton, C. A. Hassig, S. L. Schreiber. A mammalian histone deacetylase related to the yeast transcriptional regulator Rpd3p.. Science, 1996. [DOI | PubMed]
- T. Jenuwein, C. D. Allis. Translating the histone code.. Science, 2001. [DOI | PubMed]
- S. L. Schreiber, B. E. Bernstein. Signaling network model of chromatin.. Cell, 2002. [DOI | PubMed]
- E. Marban, T. J. Rink, R. W. Tsien, R. Y. Tsien. Free calcium in heart muscle at rest and during contraction measured with Ca2+ -sensitive microelectrodes.. Nature, 1980. [DOI | PubMed]
- T. J. Rink, R. Y. Tsien, A. E. Warner. Free calcium in Xenopus embryos measured with ion-selective microelectrodes.. Nature, 1980. [DOI | PubMed]
- R. Y. Tsien, T. Pozzan, T. J. Rink. T-cell mitogens cause early changes in cytoplasmic free Ca2+ and membrane potential in lymphocytes.. Nature, 1982. [DOI | PubMed]
- M. Poenie, J. Alderton, R. Steinhardt, R. Tsien. Calcium rises abruptly and briefly throughout the cell at the onset of anaphase.. Science, 1986. [DOI | PubMed]
- J. P. Shaw, P. J. Utz, D. B. Durand, J. J. Toole, E. A. Emmel, G. R. Crabtree. Identification of a putative regulator of early T cell activation genes.. Science, 1988. [DOI | PubMed]
- G. R. Crabtree. Contingent genetic regulatory events in T lymphocyte activation.. Science, 1989. [DOI | PubMed]
- M. W. Harding, A. Galat, D. E. Uehling, S. L. Schreiber. A receptor for the immunosuppressant FK506 is a cis-trans peptidyl-prolyl isomerase.. Nature, 1989. [DOI | PubMed]
- J. Liu, J. D. Farmer, W. S. Lane, J. Friedman, I. Weissman, S. L. Schreiber. Calcineurin is a common target of cyclophilin-cyclosporin A and FKBP-FK506 complexes.. Cell, 1991. [DOI | PubMed]
- S. L. Schreiber, G. R. Crabtree. The mechanism of action of cyclosporin A and FK506.. Immunol. Today, 1992. [DOI | PubMed]
- C. J. Chang. Searching for harmony in transition-metal signaling.. Nat. Chem. Biol., 2015. [DOI | PubMed]
- K. M. Dean, Y. Qin, A. E. Palmer. Visualizing metal ions in cells: an overview of analytical techniques, approaches, and probes.. Biochim. Biophys; Acta, 2012. [DOI | PubMed]
- M. K. Goshisht, N. Tripathi, G. K. Patra, M. Chaskar. Organelle-targeting ratiometric fluorescent probes: design principles, detection mechanisms, bio-applications, and challenges.. Chem. Sci., 2023. [DOI | PubMed]
- D. W. Domaille, E. L. Que, C. J. Chang. Synthetic fluorescent sensors for studying the cell biology of metals.. Nat. Chem. Biol., 2008. [DOI | PubMed]
- K. L. Haas, K. J. Franz. Application of metal coordination chemistry to explore and manipulate cell biology.. Chem. Rev., 2009. [DOI | PubMed]
- J. Tinker, P. Anees, Y. Krishnan. Quantitative Chemical Imaging of Organelles.. Acc. Chem. Res., 2024. [DOI | PubMed]
- S. C. Wilschefski, M. R. Baxter. Inductively Coupled Plasma Mass Spectrometry: Introduction to Analytical Aspects.. Clin. Biochem. Rev., 2019. [DOI | PubMed]
- F. Gyngard, M. L. Steinhauser. Biological explorations with nanoscale secondary ion mass spectrometry.. J. Anal. At. Spectrom, 2019. [DOI | PubMed]
- E. J. Brown, M. W. Albers, T. B. Shin, K. Ichikawa, C. T. Keith, W. S. Lane, S. L. Schreiber. A mammalian protein targeted by G1-arresting rapamycin-receptor complex.. Nature, 1994. [DOI | PubMed]
- S. L. Schreiber. Target-oriented and diversity-oriented organic synthesis in drug discovery.. Science, 2000. [DOI | PubMed]
- G. Fenteany, R. F. Standaert, W. S. Lane, S. Choi, E. J. Corey, S. L. Schreiber. Inhibition of proteasome activities and subunit-specific amino-terminal threonine modification by lactacystin.. Science, 1995. [DOI | PubMed]
- T. U. Mayer, T. M. Kapoor, S. J. Haggarty, R. W. King, S. L. Schreiber, T. J. Mitchison. Small molecule inhibitor of mitotic spindle bipolarity identified in a phenotype-based screen.. Science, 1999. [DOI | PubMed]
- A. C. Bishop, J. A. Ubersax, D. T. Petsch, D. P. Matheos, N. S. Gray, J. Blethrow, E. Shimizu, J. Z. Tsien, P. G. Schultz, M. D. Rose. A chemical switch for inhibitor-sensitive alleles of any protein kinase.. Nature, 2000. [DOI | PubMed]
- N. S. Hegde, D. A. Sanders, R. Rodriguez, S. Balasubramanian. The transcription factor FOXM1 is a cellular target of the natural product thiostrepton.. Nat. Chem., 2011. [DOI | PubMed]
- R. E. Moellering, M. Cornejo, T. N. Davis, C. Del Bianco, J. C. Aster, S. C. Blacklow, A. L. Kung, D. G. Gilliland, G. L. Verdine, J. E. Bradner. Direct inhibition of the NOTCH transcription factor complex.. Nature, 2009. [DOI | PubMed]
- A. Nakagawa, C. E. Adams, Y. Huang, S. R. Hamarneh, W. Liu, K. N. Von Alt, M. Mino-Kenudson, R. A. Hodin, K. D. Lillemoe, C. Fernandez-Del Castillo, A. L. Warshaw, A. S. Liss. Selective and reversible suppression of intestinal stem cell differentiation by pharmacological inhibition of BET bromodomains.. Sci. Rep., 2016. [DOI | PubMed]
- M. A. Dawson, R. K. Prinjha, A. Dittmann, G. Giotopoulos, M. Bantscheff, W. I. Chan, S. C. Robson, C. W. Chung, C. Hopf, M. M. Savitski. Inhibition of BET recruitment to chromatin as an effective treatment for MLL-fusion leukaemia.. Nature, 2011. [DOI | PubMed]
- E. W. Hunsaker, K. J. Franz. Emerging Opportunities To Manipulate Metal Trafficking for Therapeutic Benefit.. Inorg. Chem., 2019. [DOI | PubMed]
- J. M. Lehn. Toward self-organization and complex matter.. Science, 2002. [DOI | PubMed]
- M. J. Clausen, H. Poulsen. Sodium/Potassium homeostasis in the cell.. Met. Ions Life Sci., 2013. [DOI | PubMed]
- D. Shao, K. Okuse, M. B. Djamgoz. Protein-protein interactions involving voltage-gated sodium channels: Post-translational regulation, intracellular trafficking and functional expression.. Int. J. Biochem. Cell Biol., 2009. [DOI | PubMed]
- D. Pearce. SGK1 regulation of epithelial sodium transport.. Cell Physiol. Biochem., 2003. [DOI | PubMed]
- T. Kogai, K. Taki, G. A. Brent. Enhancement of sodium/iodide symporter expression in thyroid and breast cancer.. Endocr. Relat. Cancer, 2006. [DOI | PubMed]
- K. B. Gagnon, E. Delpire. Sodium Transporters in Human Health and Disease.. Front. Physiol., 2021. [DOI | PubMed]
- B. F. Palmer. Regulation of Potassium Homeostasis.. Clin. J. Am. Soc. Nephrol., 2015. [DOI | PubMed]
- M. L. Gumz, L. Rabinowitz, C. S. Wingo. An Integrated View of Potassium Homeostasis.. N. Engl. J. Med., 2015. [DOI | PubMed]
- C. R. Rose, A. Verkhratsky. Principles of sodium homeostasis and sodium signalling in astroglia.. Glia, 2016. [DOI | PubMed]
- C. R. Rose, A. Verkhratsky. Sodium homeostasis and signalling: The core and the hub of astrocyte function.. Cell Calcium, 2024. [DOI | PubMed]
- M. Hu, P. Li, C. Wang, X. Feng, Q. Geng, W. Chen, M. Marthi, W. Zhang, C. Gao, W. Reid. Parkinson’s disease-risk protein TMEM175 is a proton-activated proton channel in lysosomes.. Cell, 2022. [DOI | PubMed]
- J. Zhang, W. Zeng, Y. Han, W. R. Lee, J. Liou, Y. Jiang. Lysosomal LAMP proteins regulate lysosomal pH by direct inhibition of the TMEM175 channel.. Mol. Cell, 2023. [DOI | PubMed]
- W. Zheng, C. Shen, L. Wang, S. Rawson, W. J. Xie, C. Nist-Lund, J. Wu, Z. Shen, S. Xia, J. R. Holt, H. Wu, T.-M. Fu. pH regulates potassium conductance and drives a constitutive proton current in human TMEM175.. Sci. Adv., 2022. [DOI | PubMed]
- L. Counillon, Y. Bouret, I. Marchiq, J. Pouyssegur. Na(+)/H(+) antiporter (NHE1) and lactate/H(+) symporters (MCTs) in pH homeostasis and cancer metabolism.. Biochim. Biophys. Acta, 2016. [DOI | PubMed]
- M. Giacomello, A. Pyakurel, C. Glytsou, L. Scorrano. The cell biology of mitochondrial membrane dynamics.. Nat. Rev. Mol. Cell Biol., 2020. [DOI | PubMed]
- S. Geisberger, H. Bartolomaeus, P. Neubert, R. Willebrand, C. Zasada, T. Bartolomaeus, V. McParland, D. Swinnen, A. Geuzens, A. Maifeld. Salt Transiently Inhibits Mitochondrial Energetics in Mononuclear Phagocytes.. Circulation, 2021. [DOI | PubMed]
- G. Fenalti, P. M. Giguere, V. Katritch, X. P. Huang, A. A. Thompson, V. Cherezov, B. L. Roth, R. C. Stevens. Molecular control of delta-opioid receptor signalling.. Nature, 2014. [DOI | PubMed]
- Z. J. Tan, S. J. Chen. Salt contribution to RNA tertiary structure folding stability.. Biophys. J., 2011. [DOI | PubMed]
- A. Ambrus, D. Chen, J. Dai, T. Bialis, R. A. Jones, D. Yang. Human telomeric sequence forms a hybrid-type intramolecular G-quadruplex structure with mixed parallel/antiparallel strands in potassium solution.. Nucleic Acids Res., 2006. [DOI | PubMed]
- D. Varshney, J. Spiegel, K. Zyner, D. Tannahill, S. Balasubramanian. The regulation and functions of DNA and RNA G-quadruplexes.. Nat. Rev. Mol. Cell Biol., 2020. [DOI | PubMed]
- J. Spiegel, S. Adhikari, S. Balasubramanian. The Structure and Function of DNA G-Quadruplexes.. Trends Chem., 2020. [DOI | PubMed]
- H. J. Lipps, D. Rhodes. G-quadruplex structures: in vivo evidence and function.. Trends Cell Biol., 2009. [DOI | PubMed]
- D. Rhodes, H. J. Lipps. G-quadruplexes and their regulatory roles in biology.. Nucleic Acids Res., 2015. [DOI | PubMed]
- S. Müller, S. Kumari, R. Rodriguez, S. Balasubramanian. Small-molecule-mediated G-quadruplex isolation from human cells.. Nat. Chem., 2010. [DOI | PubMed]
- R. Rodriguez, K. M. Miller, J. V. Forment, C. R. Bradshaw, M. Nikan, S. Britton, T. Oelschlaegel, B. Xhemalce, S. Balasubramanian, S. P. Jackson. Small-molecule-induced DNA damage identifies alternative DNA structures in human genes.. Nat. Chem. Biol., 2012. [DOI | PubMed]
- S. Solier, S. Müller, R. Rodriguez. Whole-genome mapping of small-molecule targets for cancer medicine.. Curr. Opin. Chem. Biol., 2020. [DOI | PubMed]
- B. P. Bean. The action potential in mammalian central neurons.. Nat. Rev. Neurosci., 2007. [DOI | PubMed]
- D. Rotko, W. S. Kunz, A. Szewczyk, B. Kulawiak. Signaling pathways targeting mitochondrial potassium channels.. Int. J. Biochem. Cell Biol., 2020. [DOI | PubMed]
- L. Y. Jan, Y. N. Jan. Voltage-gated potassium channels and the diversity of electrical signalling.. J. Physiol., 2012. [DOI | PubMed]
- M. Bachmann, W. Li, M. J. Edwards, S. A. Ahmad, S. Patel, I. Szabo, E. Gulbins. Voltage-Gated Potassium Channels as Regulators of Cell Death.. Front. Cell Dev. Biol., 2020. [DOI | PubMed]
- S. P. Yu. Regulation and critical role of potassium homeostasis in apoptosis.. Prog. Neurobiol., 2003. [DOI | PubMed]
- D. Urrego, A. P. Tomczak, F. Zahed, W. Stuhmer, L. A. Pardo. Potassium channels in cell cycle and cell proliferation.. Philos. Trans. R. Soc. London B. Biol. Sci., 2014. [DOI | PubMed]
- S. Tahara, K. Fukuda, H. Kodama, T. Kato, S. Miyoshi, S. Ogawa. Potassium channel blocker activates extracellular signal-regulated kinases through Pyk2 and epidermal growth factor receptor in rat cardiomyocytes.. J. Am. Coll. Cardiol., 2001. [DOI | PubMed]
- J. A. McCubrey, L. S. Steelman, W. H. Chappell, S. L. Abrams, E. W. Wong, F. Chang, B. Lehmann, D. M. Terrian, M. Milella, A. Tafuri. Roles of the Raf/MEK/ERK pathway in cell growth, malignant transformation and drug resistance.. Biochim. Biophys. Acta, 2007. [DOI | PubMed]
- J. Zou, K. Mitra, P. Anees, D. Oettinger, J. R. Ramirez, A. T. Veetil, P. D. Gupta, R. Rao, J. J. Smith, P. Kratsios. A DNA nanodevice for mapping sodium at single-organelle resolution.. Nat. Biotechnol., 2024. [DOI | PubMed]
- P. Anees, A. Saminathan, E. R. Rozmus, A. Di, A. B. Malik, B. P. Delisle, Y. Krishnan. Detecting organelle-specific activity of potassium channels with a DNA nanodevice.. Nat. Biotechnol., 2024. [DOI | PubMed]
- T. K. Leslie, A. D. James, F. Zaccagna, J. T. Grist, S. Deen, A. Kennerley, F. Riemer, J. D. Kaggie, F. A. Gallagher, F. J. Gilbert. Sodium homeostasis in the tumour microenvironment.. Biochim. Biophys. Acta Rev. Cancer, 2019. [DOI | PubMed]
- E. Murphy, D. A. Eisner. Regulation of intracellular and mitochondrial sodium in health and disease.. Circ. Res., 2009. [DOI | PubMed]
- F. J. Gennari. Disorders of potassium homeostasis. Hypokalemia and hyperkalemia.. Crit. Care Clin., 2002. [DOI | PubMed]
- Y. Zhou, C. O. Wong, K. J. Cho, D. van der Hoeven, H. Liang, D. P. Thakur, J. Luo, M. Babic, K. E. Zinsmaier, M. X. Zhu. SIGNAL TRANSDUCTION. Membrane potential modulates plasma membrane phospholipid dynamics and K-Ras signaling.. Science, 2015. [DOI | PubMed]
- S. P. Fraser, J. K. Diss, A. M. Chioni, M. E. Mycielska, H. Pan, R. F. Yamaci, F. Pani, Z. Siwy, M. Krasowska, Z. Grzywna. Voltage-gated sodium channel expression and potentiation of human breast cancer metastasis.. Clin. Cancer Res., 2005. [DOI | PubMed]
- W. J. Brackenbury. Voltage-gated sodium channels and metastatic disease.. Channels, 2012. [DOI | PubMed]
- D. J. Kyle, V. I. Ilyin. Sodium channel blockers.. J. Med. Chem., 2007. [DOI | PubMed]
- M. B. A. Djamgoz. Ranolazine: a potential anti-metastatic drug targeting voltage-gated sodium channels.. Br. J. Cancer, 2024. [DOI | PubMed]
- V. Driffort, L. Gillet, E. Bon, S. Marionneau-Lambot, T. Oullier, V. Joulin, C. Collin, J. C. Pages, M. L. Jourdan, S. Chevalier, P. Bougnoux, J.-Y. Le Guennec, P. Besson, S. Roger. Ranolazine inhibits NaV1.5-mediated breast cancer cell invasiveness and lung colonization.. Mol. Cancer, 2014. [DOI | PubMed]
- J. Coronel, L. Cetina, I. Pacheco, C. Trejo-Becerril, A. Gonzalez-Fierro, E. de la Cruz-Hernandez, E. Perez-Cardenas, L. Taja-Chayeb, D. Arias-Bofill, M. Candelaria, S. Vidal, A. Dueñas-González. A double-blind, placebo-controlled, randomized phase III trial of chemotherapy plus epigenetic therapy with hydralazine valproate for advanced cervical cancer.. Preliminary results. Med. Oncol., 2011. [DOI]
- M. Nelson, M. Yang, A. A. Dowle, J. R. Thomas, W. J. Brackenbury. The sodium channel-blocking antiepileptic drug phenytoin inhibits breast tumour growth and metastasis.. Mol. Cancer, 2015. [DOI | PubMed]
- M. Mantegazza, G. Curia, G. Biagini, D. S. Ragsdale, M. Avoli. Voltage-gated sodium channels as therapeutic targets in epilepsy and other neurological disorders.. Lancet Neurol., 2010. [DOI | PubMed]
- J. J. Clare, S. N. Tate, M. Nobbs, M. A. Romanos. Voltage-gated sodium channels as therapeutic targets.. Drug Discovery Today, 2000. [DOI | PubMed]
- Y. Zhou, W. Sun, N. Chen, C. Xu, X. Wang, K. Dong, B. Zhang, J. Zhang, N. Hao, A. Sun. Discovery of NKCC1 as a potential therapeutic target to inhibit hepatocellular carcinoma cell growth and metastasis.. Oncotarget, 2017. [DOI | PubMed]
- C. Scafoglio, B. A. Hirayama, V. Kepe, J. Liu, C. Ghezzi, N. Satyamurthy, N. A. Moatamed, J. Huang, H. Koepsell, J. R. Barrio, E. M. Wright. Functional expression of sodium-glucose transporters in cancer.. Proc. Natl. Acad. Sci. U.S.A., 2015. [DOI | PubMed]
- H. Matthews, M. Ranson, M. J. Kelso. Anti-tumour/metastasis effects of the potassium-sparing diuretic amiloride: an orally active anti-cancer drug waiting for its call-of-duty?.. Int. J. Cancer, 2011. [DOI | PubMed]
- T. R. Kleyman, E. J. Cragoe. Cation transport probes: the amiloride series.. Methods Enzymol., 1990. [DOI | PubMed]
- T. R. Kleyman, E. J. Cragoe. Amiloride and its analogs as tools in the study of ion transport.. J. Membr. Biol., 1988. [DOI | PubMed]
- W. G. North, G. Gao, V. A. Memoli, R. H. Pang, L. Lynch. Breast cancer expresses functional NMDA receptors.. Breast Cancer Res. Treat., 2010. [DOI | PubMed]
- S. K. Vodnala, R. Eil, R. J. Kishton, M. Sukumar, T. N. Yamamoto, N.-H. Ha, P.-H. Lee, M. Shin, S. J. Patel, Z. Yu, D. C. Palmer, M. J. Kruhlak, X. Liu, J. W. Locasale, J. Huang, R. Roychoudhuri, T. Finkel, C. A. Klebanoff, N. P. Restifo. T cell stemness and dysfunction in tumors are triggered by a common mechanism.. Science, 2019. [DOI | PubMed]
- S. Bonnet, S. L. Archer, J. Allalunis-Turner, A. Haromy, C. Beaulieu, R. Thompson, C. T. Lee, G. D. Lopaschuk, L. Puttagunta, S. Bonnet. A mitochondria-K+ channel axis is suppressed in cancer and its normalization promotes apoptosis and inhibits cancer growth.. Cancer Cell, 2007. [DOI | PubMed]
- H. H. Nietsch, M. W. Roe, J. F. Fiekers, A. L. Moore, S. D. Lidofsky. Activation of potassium and chloride channels by tumor necrosis factor alpha. Role in liver cell death.. J. Biol. Chem., 2000. [DOI | PubMed]
- F. Beauvais, L. Michel, L. Dubertret. Human eosinophils in culture undergo a striking and rapid shrinkage during apoptosis. Role of K+ channels.. J. Leukoc. Biol., 1995. [DOI | PubMed]
- F. Gantner, S. Uhlig, A. Wendel. Quinine inhibits release of tumor necrosis factor, apoptosis, necrosis and mortality in a murine model of septic liver failure.. Eur. J. Pharmacol., 1995. [DOI | PubMed]
- C. Xia, C. Liu, S. Ren, Y. Cai, Q. Zhang, C. Xia. Potassium channels, tumorigenesis and targeted drugs.. Biomed. Pharmacother., 2023. [DOI | PubMed]
- Q. Ru, X. Tian, M. S. Pi, L. Chen, K. Yue, Q. Xiong, B. M. Ma, C. Y. Li. Voltage-gated K+ channel blocker quinidine inhibits proliferation and induces apoptosis by regulating expression of microRNAs in human glioma U87-MG cells.. Int. J. Oncol., 2015. [DOI | PubMed]
- X. Huang, L. Y. Jan. Targeting potassium channels in cancer.. J. Cell Biol., 2014. [DOI | PubMed]
- G. D’Alessandro, M. Catalano, M. Sciaccaluga, G. Chece, R. Cipriani, M. Rosito, A. Grimaldi, C. Lauro, G. Cantore, A. Santoro. KCa3.1 channels are involved in the infiltrative behavior of glioblastoma in vivo.. Cell Death. Dis., 2013. [DOI | PubMed]
- P. Song, Y. Du, W. Song, H. Chen, Z. Xuan, L. Zhao, J. Chen, J. Chen, D. Guo, C. Jin. KCa3.1 as an Effective Target for Inhibition of Growth and Progression of Intrahepatic Cholangiocarcinoma.. J. Cancer, 2017. [DOI | PubMed]
- N. Comes, A. Serrano-Albarras, J. Capera, C. Serrano-Novillo, E. Condom, S. Ramon y Cajal, J. C. Ferreres, A. Felipe. Involvement of potassium channels in the progression of cancer to a more malignant phenotype.. Biochim. Biophys. Acta, 2015. [DOI | PubMed]
- M. Abdul, A. Santo, N. Hoosein. Activity of potassium channel-blockers in breast cancer.. Anticancer Res., 2003. [PubMed]
- S. L. Payne, P. Ram, D. H. Srinivasan, T. T. Le, M. Levin, M. J. Oudin. Potassium channel-driven bioelectric signalling regulates metastasis in triple-negative breast cancer.. EBioMedicine, 2022. [DOI | PubMed]
- E. Gulbins, N. Sassi, H. Grassme, M. Zoratti, I. Szabo. Role of Kv1.3 mitochondrial potassium channel in apoptotic signalling in lymphocytes.. Biochim. Biophys. Acta, 2010. [DOI | PubMed]
- S. H. Jang, S. Y. Choi, P. D. Ryu, S. Y. Lee. Anti-proliferative effect of Kv1.3 blockers in A549 human lung adenocarcinoma in vitro and in vivo.. Eur. J. Pharmacol., 2011. [DOI | PubMed]
- X. D. Shao, K. C. Wu, Z. M. Hao, L. Hong, J. Zhang, D. M. Fan. The potent inhibitory effects of cisapride, a specific blocker for human ether-a-go-go-related gene (HERG) channel, on gastric cancer cells.. Cancer Biol. Ther., 2005. [DOI | PubMed]
- H. Wulff, N. A. Castle, L. A. Pardo. Voltage-gated potassium channels as therapeutic targets.. Nat. Rev. Drug Discovery, 2009. [DOI | PubMed]
- M. D’Amico, L. Gasparoli, A. Arcangeli. Potassium channels: novel emerging biomarkers and targets for therapy in cancer.. Recent Pat. Anticancer Drug Discovery, 2012. [DOI]
- L. H. Hurley, R. T. Wheelhouse, D. Sun, S. M. Kerwin, M. Salazar, O. Y. Fedoroff, F. X. Han, H. Han, E. Izbicka, D. D. Von Hoff. G-quadruplexes as targets for drug design.. Pharmacol. Ther., 2000. [DOI | PubMed]
- H. Han, L. H. Hurley. G-quadruplex DNA: a potential target for anti-cancer drug design.. Trends Pharmacol. Sci., 2000. [DOI | PubMed]
- R. Rodriguez, S. Müller, J. A. Yeoman, C. Trentesaux, J. F. Riou, S. Balasubramanian. A novel small molecule that alters shelterin integrity and triggers a DNA-damage response at telomeres.. J. Am. Chem. Soc., 2008. [DOI | PubMed]
- R. Hänsel-Hertsch, A. Simeone, A. Shea, W. W. I. Hui, K. G. Zyner, G. Marsico, O. M. Rueda, A. Bruna, A. Martin, X. Zhang. Landscape of G-quadruplex DNA structural regions in breast cancer.. Nat. Genet., 2020. [DOI | PubMed]
- Z. Yu, J. Spiegel, L. Melidis, W. W. I. Hui, X. Zhang, A. Radzevicius, S. Balasubramanian. Chem-map profiles drug binding to chromatin in cells.. Nat. Biotechnol., 2023. [DOI | PubMed]
- X. Zhang, J. Spiegel, S. Martinez Cuesta, S. Adhikari, S. Balasubramanian. Chemical profiling of DNA G-quadruplex-interacting proteins in live cells.. Nat. Chem., 2021. [DOI | PubMed]
- M. Olivieri, T. Cho, A. Alvarez-Quilon, K. Li, M. J. Schellenberg, M. Zimmermann, N. Hustedt, S. E. Rossi, S. Adam, H. Melo. A Genetic Map of the Response to DNA Damage in Human Cells.. Cell, 2020. [DOI | PubMed]
- J. Jantsch, V. Schatz, D. Friedrich, A. Schroder, C. Kopp, I. Siegert, A. Maronna, D. Wendelborn, P. Linz, K. J. Binger. Cutaneous Na+ storage strengthens the antimicrobial barrier function of the skin and boosts macrophage-driven host defense.. Cell Metab., 2015. [DOI | PubMed]
- W. C. Zhang, X. J. Zheng, L. J. Du, J. Y. Sun, Z. X. Shen, C. Shi, S. Sun, Z. Zhang, X. Q. Chen, M. Qin. High salt primes a specific activation state of macrophages, M(Na).. Cell Res., 2015. [DOI | PubMed]
- K. J. Binger, M. Gebhardt, M. Heinig, C. Rintisch, A. Schroeder, W. Neuhofer, K. Hilgers, A. Manzel, C. Schwartz, M. Kleinewietfeld. High salt reduces the activation of IL-4- and IL-13-stimulated macrophages.. J. Clin. Invest., 2015. [DOI | PubMed]
- D. M. Mosser, J. P. Edwards. Exploring the full spectrum of macrophage activation.. Nat. Rev. Immunol., 2008. [DOI | PubMed]
- C. C. Shieh, M. Coghlan, J. P. Sullivan, M. Gopalakrishnan. Potassium channels: molecular defects, diseases, and therapeutic opportunities.. Pharmacol. Rev., 2000. [PubMed]
- A. Di, S. Xiong, Z. Ye, R. K. S. Malireddi, S. Kometani, M. Zhong, M. Mittal, Z. Hong, T. D. Kanneganti, J. Rehman. The TWIK2 Potassium Efflux Channel in Macrophages Mediates NLRP3 Inflammasome-Induced Inflammation.. Immunity, 2018. [DOI | PubMed]
- N. Li, L. Liu, G. Li, M. Xia, C. Du, Z. Zheng. The role of BKCa in endometrial cancer HEC-1-B cell proliferation and migration.. Gene, 2018. [DOI | PubMed]
- F. Maqoud, A. Curci, R. Scala, A. Pannunzio, F. Campanella, M. Coluccia, G. Passantino, N. Zizzo, D. Tricarico. Cell Cycle Regulation by Ca(2+)-Activated K(+) (BK) Channels Modulators in SH-SY5Y Neuroblastoma Cells.. Int. J. Mol. Sci., 2018. [DOI | PubMed]
- P. Rosa, L. Sforna, S. Carlomagno, G. Mangino, M. Miscusi, M. Pessia, F. Franciolini, A. Calogero, L. Catacuzzeno. Overexpression of Large-Conductance Calcium-Activated Potassium Channels in Human Glioblastoma Stem-Like Cells and Their Role in Cell Migration.. J. Cell Physiol., 2017. [DOI | PubMed]
- Y. He, Y. Lin, F. He, L. Shao, W. Ma, F. He. Role for calcium-activated potassium channels (BK) in migration control of human hepatocellular carcinoma cells.. J. Cell Mol. Med., 2021. [DOI | PubMed]
- R. Amir, C. E. Argoff, G. J. Bennett, T. R. Cummins, M. E. Durieux, P. Gerner, M. S. Gold, F. Porreca, G. R. Strichartz. The role of sodium channels in chronic inflammatory and neuropathic pain.. J. Pain, 2006. [DOI]
- M. C. Haigney, E. G. Lakatta, M. D. Stern, H. S. Silverman. Sodium channel blockade reduces hypoxic sodium loading and sodium-dependent calcium loading.. Circulation, 1994. [DOI | PubMed]
- A. S. Terkawi, M. E. Durieux, A. Gottschalk, D. Brenin, M. Tiouririne. Effect of intravenous lidocaine on postoperative recovery of patients undergoing mastectomy: a double-blind, placebo-controlled randomized trial.. Reg. Anesth. Pain Med., 2014. [DOI | PubMed]
- G. Hasenfuss, L. S. Maier. Mechanism of action of the new anti-ischemia drug ranolazine.. Clin. Res. Cardiol., 2008. [DOI | PubMed]
- J. Kimura, T. Watano, M. Kawahara, E. Sakai, J. Yatabe. Direction-independent block of bi-directional Na+/Ca2+ exchange current by KB-R7943 in guinea-pig cardiac myocytes.. Br. J. Pharmacol., 1999. [DOI | PubMed]
- Z. Long, B. Chen, Q. Liu, J. Zhao, Z. Yang, X. Dong, L. Xia, S. Huang, X. Hu, B. Song. The reverse-mode NCX1 activity inhibitor KB-R7943 promotes prostate cancer cell death by activating the JNK pathway and blocking autophagic flux.. Oncotarget, 2106. [DOI]
- S. W. Boyce, C. Bartels, R. Bolli, B. Chaitman, J. C. Chen, E. Chi, A. Jessel, D. Kereiakes, J. Knight, L. Thulin. Impact of sodium-hydrogen exchange inhibition by cariporide on death or myocardial infarction in high-risk CABG surgery patients: results of the CABG surgery cohort of the GUARDIAN study.. J. Thorac. Cardiovasc. Surg., 2003. [DOI | PubMed]
- A. Baartscheer, C. A. Schumacher, M. M. van Borren, C. N. Belterman, R. Coronel, J. W. Fiolet. Increased Na+/H+-exchange activity is the cause of increased [Na+]i and underlies disturbed calcium handling in the rabbit pressure and volume overload heart failure model.. Cardiovasc. Res., 2003. [DOI | PubMed]
- R. M. Mentzer, C. Bartels, R. Bolli, S. Boyce, G. D. Buckberg, B. Chaitman, A. Haverich, J. Knight, P. Menasche, M. L. Myers. Sodium-hydrogen exchange inhibition by cariporide to reduce the risk of ischemic cardiac events in patients undergoing coronary artery bypass grafting: results of the EXPEDITION study.. Ann. Thorac. Surg., 2008. [DOI | PubMed]
- A. D. James, T. K. Leslie, J. D. Kaggie, L. Wiggins, L. Patten, J. Murphy O’Duinn, S. Langer, M. C. Labarthe, F. Riemer, G. Baxter. Sodium accumulation in breast cancer predicts malignancy and treatment response.. Br. J. Cancer, 2022. [DOI | PubMed]
- R. Christopherson, K. E. James, M. Tableman, P. Marshall, F. E. Johnson. Long-term survival after colon cancer surgery: a variation associated with choice of anesthesia.. Anesth. Analg., 2008. [DOI | PubMed]
- T. Berghmans, J. J. Lafitte, A. Scherpereel, L. Ameye, M. Paesmans, A. P. Meert, B. Colinet, C. Tulippe, L. Willems, N. Leclercq, J.-P. Sculler. VAC chemotherapy with valproic acid for refractory/relapsing small cell lung cancer: a phase II study.. ERJ. Open Res., 2015. [DOI | PubMed]
- M. Yang, D. J. Kozminski, L. A. Wold, R. Modak, J. D. Calhoun, L. L. Isom, W. J. Brackenbury. Therapeutic potential for phenytoin: targeting Na(v)1.5 sodium channels to reduce migration and invasion in metastatic breast cancer.. Breast Cancer Res. Treat., 2012. [DOI | PubMed]
- A. Scherpereel, T. Berghmans, J. J. Lafitte, B. Colinet, M. Richez, Y. Bonduelle, A. P. Meert, X. Dhalluin, N. Leclercq, M. Paesmans. Valproate-doxorubicin: promising therapy for progressing mesothelioma. A phase II study.. Eur. Respir. J., 2011. [DOI | PubMed]
- J. Lin, J. H. Hoffman-Censits, D. Duffy, I. Chervoneva, D. Kilpatrick, B. Kennedy, E. J. Trabulsi, C. D. Lallas, L. G. Gomella, T. Force, W. K. Kelly. A pilot phase II Study of digoxin in patients with recurrent prostate cancer as evident by a rising PSA.. J. Clin. Oncol., 2013. [DOI]
- C. Lee, L. V. Hryshko. SEA0400: a novel sodium-calcium exchange inhibitor with cardioprotective properties.. Cardiovasc. Drug Rev., 2004. [DOI | PubMed]
- M. A. Khan, P. Gerner, G. Kuo Wang. Amitriptyline for prolonged cutaneous analgesia in the rat.. Anesthesiology, 2002. [DOI | PubMed]
- V. I. Ilyin, J. D. Pomonis, G. T. Whiteside, J. E. Harrison, M. S. Pearson, L. Mark, P. I. Turchin, S. Gottshall, R. B. Carter, P. Nguyen. Pharmacology of 2-[4-(4-chloro-2-fluorophenoxy)phenyl]-pyrimidine-4-carboxamide: a potent, broad-spectrum state-dependent sodium channel blocker for treating pain states.. J. Pharmacol. Exp. Ther., 2006. [DOI | PubMed]
- R. M. Brochu, I. E. Dick, J. W. Tarpley, E. McGowan, D. Gunner, J. Herrington, P. P. Shao, D. Ok, C. Li, W. H. Parsons. Block of peripheral nerve sodium channels selectively inhibits features of neuropathic pain in rats.. Mol. Pharmacol., 2006. [DOI | PubMed]
- P. Weidmann, P. Ferrari. Central role of sodium in hypertension in diabetic subjects.. Diabetes Care, 1991. [DOI | PubMed]
- W. B. Reeves, S. V. Shah. Activation of potassium channels contributes to hypoxic injury in proximal tubules.. J. Clin. Invest., 1994. [DOI | PubMed]
- A. Khatun, M. Fujimoto, H. Kito, S. Niwa, T. Suzuki, S. Ohya. Down-Regulation of Ca(2+)-Activated K(+) Channel K(Ca)1.1 in Human Breast Cancer MDA-MB-453 Cells Treated with Vitamin D Receptor Agonists.. Int. J. Mol. Sci., 2016. [DOI | PubMed]
- S. Rajan, E. Wischmeyer, G. Xin Liu, R. Preisig-Muller, J. Daut, A. Karschin, C. Derst. TASK-3, a novel tandem pore domain acid-sensitive K+ channel. An extracellular histiding as pH sensor.. J. Biol. Chem., 2000. [DOI | PubMed]
- Y. Kim, H. Bang, D. Kim. TASK-3, a new member of the tandem pore K(+) channel family.. J. Biol. Chem., 2000. [DOI | PubMed]
- S. P. Yu, C. H. Yeh, S. L. Sensi, B. J. Gwag, L. M. Canzoniero, Z. S. Farhangrazi, H. S. Ying, M. Tian, L. L. Dugan, D. W. Choi. Mediation of neuronal apoptosis by enhancement of outward potassium current.. Science, 1997. [DOI | PubMed]
- S. P. Yu, Z. S. Farhangrazi, H. S. Ying, C. H. Yeh, D. W. Choi. Enhancement of outward potassium current may participate in beta-amyloid peptide-induced cortical neuronal death.. Neurobiol. Dis., 1998. [DOI | PubMed]
- L. V. Colom, M. E. Diaz, D. R. Beers, A. Neely, W. J. Xie, S. H. Appel. Role of potassium channels in amyloid-induced cell death.. J. Neurochem., 1998. [DOI | PubMed]
- B. McLaughlin, S. Pal, M. P. Tran, A. A. Parsons, F. C. Barone, J. A. Erhardt, E. Aizenman. p38 activation is required upstream of potassium current enhancement and caspase cleavage in thiol oxidant-induced neuronal apoptosis.. J. Neurosci., 2001. [DOI | PubMed]
- S. Krick, O. Platoshyn, M. Sweeney, H. Kim, J. X. Yuan. Activation of K+ channels induces apoptosis in vascular smooth muscle cells.. Am. J. Physiol. Cell Physiol., 2001. [DOI | PubMed]
- S. P. Yu, C. H. Yeh, F. Gottron, X. Wang, M. C. Grabb, D. W. Choi. Role of the outward delayed rectifier K+ current in ceramide-induced caspase activation and apoptosis in cultured cortical neurons.. J. Neurochem., 1999. [DOI | PubMed]
- M. Manikkam, Y. Li, B. M. Mitchell, D. E. Mason, L. C. Freeman. Potassium channel antagonists influence porcine granulosa cell proliferation, differentiation, and apoptosis.. Biol. Reprod., 2002. [DOI | PubMed]
- J. I. Elliott, C. F. Higgins. IKCa1 activity is required for cell shrinkage, phosphatidylserine translocation and death in T lymphocyte apoptosis.. EMBO Rep., 2003. [DOI | PubMed]
- C. Yan, F. Li, Y. Zhang, Y. Li, M. Li, F. Wang, G. Zhang, Y. Li, B. Li, X. Zhao. Effects of As2O3 and Resveratrol on the Proliferation and Apoptosis of Colon Cancer Cells and the hERG-mediated Potential Mechanisms.. Curr. Pharm. Des., 2019. [DOI | PubMed]
- W. Xiong, H. Fan, Q. Zeng, Z. Deng, G. Li, W. Lu, B. Zhang, S. Lai, X. Chen, X. Xu. The in vitro anticancer effects of FS48 from salivary glands of Xenopsylla cheopis on NCI-H460 cells via its blockage of voltage-gated K(+) channels.. Acta Pharm., 2023. [DOI | PubMed]
- A. Teisseyre, M. Chmielarz, A. Uryga, K. Sroda-Pomianek, A. Palko-Labuz. Co-Application of Statin and Flavonoids as an Effective Strategy to Reduce the Activity of Voltage-Gated Potassium Channels Kv1.3 and Induce Apoptosis in Human Leukemic T Cell Line Jurkat.. Molecules, 2022. [DOI | PubMed]
- X. Wang, Y. Chen, H. Liu, S. Guo, Y. Hu, Y. Zhan, H. An. A novel anti-cancer mechanism of Nutlin-3 through downregulation of Eag1 channel and PI3K/AKT pathway.. Biochem. Biophys. Res. Commun., 2019. [DOI | PubMed]
- G. Petroni, G. Bagni, J. Iorio, C. Duranti, T. Lottini, M. Stefanini, G. Kragol, A. Becchetti, A. Arcangeli. Clarithromycin inhibits autophagy in colorectal cancer by regulating the hERG1 potassium channel interaction with PI3K.. Cell Death Dis., 2020. [DOI | PubMed]
- B. R. Downie, A. Sanchez, H. Knotgen, C. Contreras-Jurado, M. Gymnopoulos, C. Weber, W. Stuhmer, L. A. Pardo. Eag1 expression interferes with hypoxia homeostasis and induces angiogenesis in tumors.. J. Biol. Chem., 2008. [DOI | PubMed]
- R. E. Garcia-Ferreiro, D. Kerschensteiner, F. Major, F. Monje, W. Stuhmer, L. A. Pardo. Mechanism of block of hEag1 K+ channels by imipramine and astemizole.. J. Gen. Physiol., 2004. [DOI | PubMed]
- S. Pillozzi, M. Masselli, E. De Lorenzo, B. Accordi, E. Cilia, O. Crociani, A. Amedei, M. Veltroni, M. D’Amico, G. Basso. Chemotherapy resistance in acute lymphoblastic leukemia requires hERG1 channels and is overcome by hERG1 blockers.. Blood, 2011. [DOI | PubMed]
- S. G. Meuth, T. Budde, T. Kanyshkova, T. Broicher, T. Munsch, H. C. Pape. Contribution of TWIK-related acid-sensitive K+ channel 1 (TASK1) and TASK3 channels to the control of activity modes in thalamocortical neurons.. J. Neurosci., 2003. [DOI | PubMed]
- B. Musset, S. G. Meuth, G. X. Liu, C. Derst, S. Wegner, H. C. Pape, T. Budde, R. Preisig-Muller, J. Daut. Effects of divalent cations and spermine on the K+ channel TASK-3 and on the outward current in thalamic neurons.. J. Physiol., 2006. [DOI | PubMed]
- M. A. Katsnelson, L. G. Rucker, H. M. Russo, G. R. Dubyak. K+ efflux agonists induce NLRP3 inflammasome activation independently of Ca2+ signaling.. J. Immunol., 2015. [DOI | PubMed]
- A. A. Goda, A. B. Siddique, M. Mohyeldin, N. M. Ayoub, K. A. El Sayed. The Maxi-K (BK) Channel Antagonist Penitrem A as a Novel Breast Cancer-Targeted Therapeutic.. Mar. Drugs, 2018. [DOI | PubMed]
- H. Lee, O. B. Kwon, J. E. Lee, Y. H. Jeon, D. S. Lee, S. H. Min, J. W. Kim. Repositioning Trimebutine Maleate as a Cancer Treatment Targeting Ovarian Cancer Stem Cells.. Cells, 2021. [DOI | PubMed]
- D. Fukushiro-Lopes, A. D. Hegel, A. Russo, V. Senyuk, M. Liotta, G. C. Beeson, C. C. Beeson, J. Burdette, R. K. Potkul, S. Gentile. Repurposing Kir6/SUR2 Channel Activator Minoxidil to Arrests Growth of Gynecologic Cancers.. Front Pharmacol., 2020. [DOI | PubMed]
- K. P. Schlingmann, S. Weber, M. Peters, L. Niemann Nejsum, H. Vitzthum, K. Klingel, M. Kratz, E. Haddad, E. Ristoff, D. Dinour. Hypomagnesemia with secondary hypocalcemia is caused by mutations in TRPM6, a new member of the TRPM gene family.. Nat. Genet., 2002. [DOI | PubMed]
- M. J. Nadler, M. C. Hermosura, K. Inabe, A. L. Perraud, Q. Zhu, A. J. Stokes, T. Kurosaki, J. P. Kinet, R. Penner, A. M. Scharenberg. LTRPC7 is a Mg.ATP-regulated divalent cation channel required for cell viability.. Nature, 2001. [DOI | PubMed]
- A. Goytain, G. A. Quamme. Identification and characterization of a novel mammalian Mg2+ transporter with channel-like properties.. BMC Genomics, 2005. [DOI | PubMed]
- Z. G. Zou, F. J. Rios, A. C. Montezano, R. M. Touyz. TRPM7, Magnesium, and Signaling.. Int. J. Mol. Sci., 2019. [DOI | PubMed]
- J. Sahni, B. Nelson, A. M. Scharenberg. SLC41A2 encodes a plasma-membrane Mg2+ transporter.. Biochem. J., 2007. [DOI | PubMed]
- O. Shaul, D. W. Hilgemann, J. de-Almeida-Engler, M. Van Montagu, D. Inze, G. Galili. Cloning and characterization of a novel Mg(2+)/H(+) exchanger.. EMBO J., 1999. [DOI | PubMed]
- P. J. Kausalya, S. Amasheh, D. Gunzel, H. Wurps, D. Muller, M. Fromm, W. Hunziker. Disease-associated mutations affect intracellular traffic and paracellular Mg2+ transport function of Claudin-16.. J. Clin. Invest., 2006. [DOI | PubMed]
- D. M. Bui, J. Gregan, E. Jarosch, A. Ragnini, R. J. Schweyen. The bacterial magnesium transporter CorA can functionally substitute for its putative homologue Mrs2p in the yeast inner mitochondrial membrane.. J. Biol. Chem., 1999. [DOI | PubMed]
- G. Zsurka, J. Gregan, R. J. Schweyen. The human mitochondrial Mrs2 protein functionally substitutes for its yeast homologue, a candidate magnesium transporter.. Genomics, 2001. [DOI | PubMed]
- L. Mastrototaro, A. Smorodchenko, J. R. Aschenbach, M. Kolisek, G. Sponder. Solute carrier 41A3 encodes for a mitochondrial Mg(2+) efflux system.. Sci. Rep., 2016. [DOI | PubMed]
- N. Vishnu, M. Venkatesan, T. R. Madaris, M. K. Venkateswaran, K. Stanley, K. Ramachandran, A. Chidambaram, A. K. Madesh, W. Yang, J. Nair. ERMA (TMEM94) is a P-type ATPase transporter for Mg(2+) uptake in the endoplasmic reticulum.. Mol. Cell, 2024. [DOI | PubMed]
- E. B. Babsky, P. F. Minajev. Combined action upon muscle of adenosine triphosphate, acetylcholine and potassium, calcium and magnesium ions.. Nature, 1946. [DOI]
- W. B. Vernon. The role of magnesium in nucleic-acid and protein metabolism.. Magnesium, 1988. [PubMed]
- A. Hartwig. Role of magnesium in genomic stability.. Mutat. Res., 2001. [DOI | PubMed]
- R. S. Adelstein, J. R. Sellers. Effects of calcium on vascular smooth muscle contraction.. Am. J. Cardiol., 1987. [DOI]
- D. C. Hill-Eubanks, M. E. Werner, T. J. Heppner, M. T. Nelson. Calcium signaling in smooth muscle.. Cold Spring Harb. Perspect. Biol., 2011. [DOI | PubMed]
- Y. Liu, Q. Wang, Z. Zhang, R. Fu, T. Zhou, C. Long, T. He, D. Yang, Z. Li, S. Peng. Magnesium supplementation enhances mTOR signalling to facilitate myogenic differentiation and improve aged muscle performance.. Bone, 2021. [DOI | PubMed]
- H. Qi, Y. Liu, L. Wu, S. Ni, J. Sun, J. Xue, Q. Liu, X. Ni, W. Fan. MicroRNA-16, via FGF2 Regulation of the ERK/MAPK Pathway, Is Involved in the Magnesium-Promoted Osteogenic Differentiation of Mesenchymal Stem Cells.. Oxid. Med. Cell Longev., 2020. [DOI | PubMed]
- R. Morris, N. J. Kershaw, J. J. Babon. The molecular details of cytokine signaling via the JAK/STAT pathway.. Protein Sci., 2018. [DOI | PubMed]
- J. H. de Baaij, J. G. Hoenderop, R. J. Bindels. Magnesium in man: implications for health and disease.. Physiol. Rev., 2015. [DOI | PubMed]
- C. G. Musso. Magnesium metabolism in health and disease.. Int. Urol. Nephrol., 2009. [DOI | PubMed]
- S. Chakraborti, T. Chakraborti, M. Mandal, A. Mandal, S. Das, S. Ghosh. Protective role of magnesium in cardiovascular diseases: a review.. Mol. Cell. Biochem., 2002. [DOI | PubMed]
- N. H. J. Leenders, M. G. Vervloet. Magnesium: A Magic Bullet for Cardiovascular Disease in Chronic Kidney Disease?.. Nutrients, 2019. [DOI | PubMed]
- B. Raju, E. Murphy, L. A. Levy, R. D. Hall, R. E. London. A fluorescent indicator for measuring cytosolic free magnesium.. Am. J. Physiol., 1989. [DOI | PubMed]
- E. R. H. Walter, M. A. Fox, D. Parker, J. A. G. Williams. Enhanced selectivity for Mg(2+) with a phosphinate-based chelate: APDAP versus APTRA.. Dalton. Trans., 2018. [DOI | PubMed]
- S. Castiglioni, J. A. Maier. Magnesium and cancer: a dangerous liason.. Magnes. Res., 2011. [DOI]
- H. S. Sun, F. D. Horgen, D. Romo, K. G. Hull, S. A. Kiledal, A. Fleig, Z. P. Feng. Waixenicin A, a marine-derived TRPM7 inhibitor: a promising CNS drug lead.. Acta Pharmacol. Sin., 2020. [DOI | PubMed]
- V. Trapani, D. Arduini, A. Cittadini, F. I. Wolf. From magnesium to magnesium transporters in cancer: TRPM7, a novel signature in tumour development.. Magnes. Res., 2013. [DOI | PubMed]
- J. Auwercx, P. Rybarczyk, P. Kischel, I. Dhennin-Duthille, D. Chatelain, H. Sevestre, I. Van Seuningen, H. Ouadid-Ahidouch, N. Jonckheere, M. Gautier. Mg(2+) Transporters in Digestive Cancers.. Nutrients, 2021. [DOI | PubMed]
- B. Chaigne-Delalande, M. J. Lenardo. Divalent cation signaling in immune cells.. Trends Immunol., 2014. [DOI | PubMed]
- F. Y. Li, B. Chaigne-Delalande, C. Kanellopoulou, J. C. Davis, H. F. Matthews, D. C. Douek, J. I. Cohen, G. Uzel, H. C. Su, M. J. Lenardo. Second messenger role for Mg2+ revealed by human T-cell immunodeficiency.. Nature, 2011. [DOI | PubMed]
- J. Lotscher, A.-A. Marti i Lindez, N. Kirchhammer, E. Cribioli, G. M. P. Giordano Attianese, M. P. Trefny, M. Lenz, S. I. Rothschild, P. Strati, M. Kunzli, C. Lotter, S. H. Schenk, P. Dehio, J. Loliger, L. Litzler, D. Schreiner, V. Koch, N. Page, D. Lee, J. Grahlert, D. Kuzmin, A.-V. Burgener, D. Merkler, M. Pless, M. L. Balmer, W. Reith, J. Huwyler, M. Irving, C. G. King, A. Zippelius, C. Hess. Magnesium sensing via LFA-1 regulates CD8(+) T cell effector function.. Cell, 2022. [DOI | PubMed]
- R. Yamanaka, Y. Shindo, K. Hotta, K. Suzuki, K. Oka. GABA-Induced Intracellular Mg(2+) Mobilization Integrates and Coordinates Cellular Information Processing for the Maturation of Neural Networks.. Curr. Biol., 2018. [DOI | PubMed]
- J. Simon, N. Goikoetxea-Usandizaga, M. Serrano-Macia, D. Fernandez-Ramos, D. Saenz de Urturi, J. J. Gruskos, P. Fernandez-Tussy, S. Lachiondo-Ortega, I. Gonzalez-Recio, R. Rodriguez-Agudo, V. Gutierrez-de-Juan, B. Rodriguez-Iruretagoyena, M. Varela-Rey, P. Gimenez-Mascarell, M. Mercado-Gomez, B. Gomez-Santos, C. Fernandez-Rodriguez, F. Lopitz-Otsoa, M. Bizkarguenaga, S. Dames, U. Schaeper, F. Martin, G. Sabio, P. Iruzubieta, J. Crespo, P. Aspichueta, K. H.-Y. Chu, D. Buccella, C. Martin, T. C. Delgado, L. A. Martinez-Cruz, M. L. Martinez-Chantar. Magnesium accumulation upon cyclin M4 silencing activates microsomal triglyceride transfer protein improving NASH.. J. Hepatol., 2021. [DOI | PubMed]
- I. Gonzalez-Recio, J. Simon, N. Goikoetxea-Usandizaga, M. Serrano-Macia, M. Mercado-Gomez, R. Rodriguez-Agudo, S. Lachiondo-Ortega, C. Gil-Pitarch, C. Fernandez-Rodriguez, D. Castellana, M. U. Latasa, L. Abecia, J. Anguita, T. C. Delgado, P. Iruzubieta, J. Crespo, S. Hardy, P. D. Petrov, R. Jover, M. A. Avila, C. Martin, U. Schaeper, M. L. Tremblay, J. W. Dear, S. Masson, M. V. McCain, H. L. Reeves, R. J. Andrade, M. I. Lucena, D. Buccella, L. A. Martinez-Cruz, M. L Martinez-Chantar. Restoring cellular magnesium balance through Cyclin M4 protects against acetaminophen-induced liver damage.. Nat. Commun., 2022. [DOI | PubMed]
- K. Jomova, M. Makova, S. Y. Alomar, S. H. Alwasel, E. Nepovimova, K. Kuca, C. J. Rhodes, M. Valko. Essential metals in health and disease.. Chem. Biol. Interact., 2022. [DOI | PubMed]
- M. Zajac, S. Modi, Y. Krishnan. The evolution of organellar calcium mapping technologies.. Cell Calcium, 2022. [DOI | PubMed]
- N. Narayanaswamy, K. Chakraborty, A. Saminathan, E. Zeichner, K. Leung, J. Devany, Y. Krishnan. A pH-correctable, DNA-based fluorescent reporter for organellar calcium.. Nat. Methods, 2019. [DOI | PubMed]
- D. E. Clapham. Calcium signaling.. Cell, 2007. [DOI | PubMed]
- M. D. Bootman. Calcium signaling.. Cold Spring Harb. Perspect. Biol., 2012. [DOI | PubMed]
- M. D. Bootman, G. Bultynck. Fundamentals of Cellular Calcium Signaling: A Primer.. Cold Spring Harb. Perspect. Biol., 2020. [DOI | PubMed]
- G. Csordas, G. Hajnoczky. Plasticity of mitochondrial calcium signaling.. J. Biol. Chem., 2003. [DOI | PubMed]
- M. J. Berridge, M. D. Bootman, H. L. Roderick. Calcium signalling: dynamics, homeostasis and remodelling.. Nat. Rev. Mol. Cell Biol., 2003. [DOI | PubMed]
- H. N. Rubaiy. ORAI Calcium Channels: Regulation, Function, Pharmacology, and Therapeutic Targets.. Pharmaceuticals, 2023. [DOI | PubMed]
- S. Ling, P. Shi, S. Liu, X. Meng, Y. Zhou, W. Sun, S. Chang, X. Zhang, L. Zhang, C. Shi. Structural mechanism of cooperative activation of the human calcium-sensing receptor by Ca(2+) ions and L-tryptophan.. Cell Res., 2021. [DOI | PubMed]
- H. Reuter. Calcium channel modulation by neurotransmitters, enzymes and drugs.. Nature, 1983. [DOI | PubMed]
- A. Rossi, L. Manzo, S. Orrenius, M. Vahter, P. Nicotera. Modifications of cell signalling in the cytotoxicity of metals.. Pharmacol. Toxicol., 1991. [DOI | PubMed]
- J. V. Gerasimenko, A. V. Tepikin, O. H. Petersen, O. V. Gerasimenko. Calcium uptake via endocytosis with rapid release from acidifying endosomes.. Curr. Biol., 1998. [DOI | PubMed]
- B. N. Desai, N. Leitinger. Purinergic and calcium signaling in macrophage function and plasticity.. Front. Immunol., 2014. [DOI | PubMed]
- A. Shawki, B. Mackenzie. Interaction of calcium with the human divalent metal-ion transporter-1.. Biochem. Biophys. Res. Commun., 2010. [DOI | PubMed]
- D. Ramonet, A. Podhajska, K. Stafa, S. Sonnay, A. Trancikova, E. Tsika, O. Pletnikova, J. C. Troncoso, L. Glauser, D. J. Moore. PARK9-associated ATP13A2 localizes to intracellular acidic vesicles and regulates cation homeostasis and neuronal integrity.. Hum. Mol. Genet., 2012. [DOI | PubMed]
- S. I. Sim, S. von Bulow, G. Hummer, E. Park. Structural basis of polyamine transport by human ATP13A2 (PARK9).. Mol. Cell, 2021. [DOI | PubMed]
- M. Melchionda, J. K. Pittman, R. Mayor, S. Patel. Ca2+/H+ exchange by acidic organelles regulates cell migration in vivo.. J. Cell Biol., 2016. [DOI | PubMed]
- M. Zajac, S. Mukherjee, P. Anees, D. Oettinger, K. Henn, J. Srikumar, J. Zou, A. Saminathan, Y. Krishnan. A mechanism of lysosomal calcium entry.. Sci. Adv., 2024. [DOI | PubMed]
- J. O. Primeau, G. P. Armanious, M. E. Fisher, H. S. Young. The SarcoEndoplasmic Reticulum Calcium ATPase.. Subcell. Biochem., 2018. [DOI | PubMed]
- I. Vandecaetsbeek, P. Vangheluwe, L. Raeymaekers, F. Wuytack, J. Vanoevelen. The Ca2+ pumps of the endoplasmic reticulum and Golgi apparatus.. Cold Spring Harb. Perspect. Biol., 2011. [DOI | PubMed]
- R. S. Ginger, S. E. Askew, R. M. Ogborne, S. Wilson, D. Ferdinando, T. Dadd, A. M. Smith, S. Kazi, R. T. Szerencsei, R. J. Winkfein. SLC24A5 encodes a trans-Golgi network protein with potassium-dependent sodium-calcium exchange activity that regulates human epidermal melanogenesis.. J. Biol. Chem., 2008. [DOI | PubMed]
- V. Shoshan-Barmatz, S. De. Mitochondrial VDAC, the Na(+)/Ca(2+) Exchanger, and the Ca(2+) Uniporter in Ca(2+) Dynamics and Signaling.. Adv. Exp. Med. Biol., 2017. [DOI | PubMed]
- D. S. Luciani, K. S. Gwiazda, T. L. Yang, T. B. Kalynyak, Y. Bychkivska, M. H. Frey, K. D. Jeffrey, A. V. Sampaio, T. M. Underhill, J. D. Johnson. Roles of IP3R and RyR Ca2+ channels in endoplasmic reticulum stress and beta-cell death.. Diabetes, 2009. [DOI | PubMed]
- C. W. Taylor, S. C. Tovey. IP(3) receptors: toward understanding their activation.. Cold Spring Harb. Perspect. Biol., 2010. [DOI | PubMed]
- D. Chin, A. R. Means. Calmodulin: a prototypical calcium sensor.. Trends Cell Biol., 2000. [DOI | PubMed]
- M. Sheng, M. A. Thompson, M. E. Greenberg. CREB: a Ca(2+)-regulated transcription factor phosphorylated by calmodulin-dependent kinases.. Science, 1991. [DOI | PubMed]
- P. Sun, H. Enslen, P. S. Myung, R. A. Maurer. Differential activation of CREB by Ca2+/calmodulin-dependent protein kinases type II and type IV involves phosphorylation of a site that negatively regulates activity.. Genes Dev., 1994. [DOI | PubMed]
- D. Mekahli, G. Bultynck, J. B. Parys, H. De Smedt, L. Missiaen. Endoplasmic-reticulum calcium depletion and disease.. Cold Spring Harb. Perspect. Biol., 2011. [DOI | PubMed]
- N. A. Clipstone, G. R. Crabtree. Identification of calcineurin as a key signalling enzyme in T-lymphocyte activation.. Nature, 1992. [DOI | PubMed]
- J. S. Wiegert, H. Bading. Activity-dependent calcium signaling and ERK-MAP kinases in neurons: a link to structural plasticity of the nucleus and gene transcription regulation.. Cell Calcium, 2011. [DOI | PubMed]
- N. Agell, O. Bachs, N. Rocamora, P. Villalonga. Modulation of the Ras/Raf/MEK/ERK pathway by Ca(2+), and calmodulin.. Cell. Signal., 2002. [DOI | PubMed]
- D. Chuderland, R. Seger. Calcium regulates ERK signaling by modulating its protein-protein interactions.. Commun. Integr. Biol., 2008. [DOI | PubMed]
- O. Forostyak, N. Romanyuk, A. Verkhratsky, E. Sykova, G. Dayanithi. Plasticity of calcium signaling cascades in human embryonic stem cell-derived neural precursors.. Stem Cells Dev., 2013. [DOI | PubMed]
- N. C. Spitzer, E. Olson, X. Gu. Spontaneous calcium transients regulate neuronal plasticity in developing neurons.. J. Neurobiol., 1995. [DOI | PubMed]
- L. D’Souza-Li. The calcium-sensing receptor and related diseases.. Arq. Bras. Endocrinol. Metabol., 2006. [DOI | PubMed]
- M. Schonewille, A. E. Girasole, P. Rostaing, C. Mailhes-Hamon, A. Ayon, A. B. Nelson, A. Triller, M. Casado, C. I. De Zeeuw, G. Bouvier. NMDARs in granule cells contribute to parallel fiber-Purkinje cell synaptic plasticity and motor learning.. Proc. Natl. Acad. Sci. U.S.A., 2021. [DOI | PubMed]
- C. Piochon, C. Levenes, H. K. Titley, C. Hansel. The calcium sensor, rather than the route of calcium entry, defines cerebellar plasticity pathways.. Proc. Natl. Acad. Sci. U.S.A., 2022. [DOI | PubMed]
- S. Tennakoon, A. Aggarwal, E. Kallay. The calcium-sensing receptor and the hallmarks of cancer.. Biochim. Biophys. Acta, 2016. [DOI | PubMed]
- L. Iamartino, T. Elajnaf, E. Kallay, M. Schepelmann. Calcium-sensing receptor in colorectal inflammation and cancer: Current insights and future perspectives.. World J. Gastroenterol., 2018. [DOI | PubMed]
- C. Han, A. Shin, J. Lee, J. Lee, J. W. Park, J. H. Oh, J. Kim. Dietary calcium intake and the risk of colorectal cancer: a case control study.. BMC Cancer, 2015. [DOI | PubMed]
- T. A. Stewart, K. T. Yapa, G. R. Monteith. Altered calcium signaling in cancer cells.. Biochim. Biophys. Acta, 2015. [DOI | PubMed]
- S. J. Quinn, C. P. Ye, R. Diaz, O. Kifor, M. Bai, P. Vassilev, E. Brown. The Ca2+-sensing receptor: a target for polyamines.. Am. J. Physiol. Cell Physiol., 1997. [DOI]
- G. Molostvov, S. Fletcher, R. Bland, D. Zehnder. Extracellular calcium-sensing receptor mediated signalling is involved in human vascular smooth muscle cell proliferation and apoptosis.. Cell Physiol. Biochem., 2008. [DOI | PubMed]
- E. M. Brown, R. Butters, C. Katz, O. Kifor. Neomycin mimics the effects of high extracellular calcium concentrations on parathyroid function in dispersed bovine parathyroid cells.. Endocrinology, 1991. [DOI | PubMed]
- C. Cui, R. Merritt, L. Fu, Z. Pan. Targeting calcium signaling in cancer therapy.. Acta Pharm. Sin. B, 2017. [DOI | PubMed]
- A. Herrmann-Frank, M. Richter, S. Sarkozi, U. Mohr, F. Lehmann-Horn. 4-Chloro-m-cresol, a potent and specific activator of the skeletal muscle ryanodine receptor.. Biochim. Biophys. Acta, 1996. [DOI | PubMed]
- F. Zorzato, E. Scutari, V. Tegazzin, E. Clementi, S. Treves. Chlorocresol: an activator of ryanodine receptor-mediated Ca2+ release.. Mol. Pharmacol., 1993. [PubMed]
- M. Hohenegger, M. Matyash, K. Poussu, A. Herrmann-Frank, S. Sarkozi, F. Lehmann-Horn, M. Freissmuth. Activation of the skeletal muscle ryanodine receptor by suramin and suramin analogs.. Mol. Pharmacol., 1996. [PubMed]
- B. E. Ehrlich, E. Kaftan, S. Bezprozvannaya, I. Bezprozvanny. The pharmacology of intracellular Ca(2+)-release channels.. Trends Pharmacol. Sci., 1994. [DOI | PubMed]
- F. Zhao, P. Li, S. R. Chen, C. F. Louis, B. R. Fruen. Dantrolene inhibition of ryanodine receptor Ca2+ release channels. Molecular mechanism and isoform selectivity.. J. Biol. Chem., 2001. [DOI | PubMed]
- M. Takahashi, K. Tanzawa, S. Takahashi. Adenophostins, newly discovered metabolites of Penicillium brevicompactum, act as potent agonists of the inositol 1,4,5-trisphosphate receptor.. J. Biol. Chem., 1994. [DOI | PubMed]
- E. Jaimovich, C. Mattei, J. L. Liberona, C. Cardenas, M. Estrada, J. Barbier, C. Debitus, D. Laurent, J. Molgo. Xestospongin B, a competitive inhibitor of IP3-mediated Ca2+ signalling in cultured rat myotubes, isolated myonuclei, and neuroblastoma (NG108–15) cells.. FEBS Lett., 2005. [DOI | PubMed]
- C. Cardenas, M. Muller, A. McNeal, A. Lovy, F. Jana, G. Bustos, F. Urra, N. Smith, J. Molgo, J. A. Diehl. Selective Vulnerability of Cancer Cells by Inhibition of Ca(2+) Transfer from Endoplasmic Reticulum to Mitochondria.. Cell Rep., 2016. [DOI | PubMed]
- F. O’Rourke, M. B. Feinstein. The inositol 1,4,5-trisphosphate receptor binding sites of platelet membranes. pH-dependency, inhibition by polymeric sulphates, and the possible presence of arginine at the binding site.. Biochem. J., 1990. [DOI | PubMed]
- S. Orrenius, B. Zhivotovsky, P. Nicotera. Regulation of cell death: the calcium-apoptosis link.. Nat. Rev. Mol. Cell Biol., 2003. [DOI | PubMed]
- W. T. Chiu, H. A. Chang, Y. H. Lin, Y. S. Lin, H. T. Chang, H. H. Lin, S. C. Huang, M. J. Tang, M. R. Shen. Bcl(−)2 regulates store-operated Ca(2+) entry to modulate ER stress-induced apoptosis.. Cell Death Discovery, 2018. [DOI]
- G. R. Monteith, N. Prevarskaya, S. J. Roberts-Thomson. The calcium-cancer signalling nexus.. Nat. Rev. Cancer, 2017. [DOI]
- H. Zhu, H. Zhang, F. Jin, M. Fang, M. Huang, C. S. Yang, T. Chen, L. Fu, Z. Pan. Elevated Orai1 expression mediates tumor-promoting intracellular Ca2+ oscillations in human esophageal squamous cell carcinoma.. Oncotarget, 2014. [DOI | PubMed]
- R. K. Motiani, M. C. Hyzinski-Garcia, X. Zhang, M. M. Henkel, I. F. Abdullaev, Y. H. Kuo, K. Matrougui, A. A. Mongin, M. Trebak. STIM1 and Orai1 mediate CRAC channel activity and are essential for human glioblastoma invasion.. Pflugers Arch., 2013. [DOI | PubMed]
- B. A. McNally, A. Somasundaram, M. Yamashita, M. Prakriya. Gated regulation of CRAC channel ion selectivity by STIM1.. Nature, 2012. [DOI | PubMed]
- A. V. Yeromin, S. L. Zhang, W. Jiang, Y. Yu, O. Safrina, M. D. Cahalan. Molecular identification of the CRAC channel by altered ion selectivity in a mutant of Orai.. Nature, 2006. [DOI | PubMed]
- B. Sampath, K. Sankaranarayanan. Glu106 targeted inhibitors of ORAI1 as potential Ca(2+) release-activated Ca(2+) (CRAC) channel blockers – molecular modeling and docking studies.. J. Recept. Signal. Transduct. Res., 2016. [DOI | PubMed]
- I. Derler, R. Schindl, R. Fritsch, P. Heftberger, M. C. Riedl, M. Begg, D. House, C. Romanin. The action of selective CRAC channel blockers is affected by the Orai pore geometry.. Cell Calcium, 2013. [DOI | PubMed]
- A. M. Sadaghiani, S. M. Lee, J. I. Odegaard, D. B. Leveson-Gower, O. M. McPherson, P. Novick, M. R. Kim, A. N. Koehler, R. Negrin, R. E. Dolmetsch. Identification of Orai1 channel inhibitors by using minimal functional domains to screen small molecule microarrays.. Chem. Biol., 2014. [DOI | PubMed]
- W. A. Catterall, E. Perez-Reyes, T. P. Snutch, J. Striessnig. International Union of Pharmacology. XLVIII. Nomenclature and structure-function relationships of voltage-gated calcium channels.. Pharmacol. Rev., 2005. [DOI | PubMed]
- G. Franckowiak, M. Bechem, M. Schramm, G. Thomas. The optical isomers of the 1,4-dihydropyridine BAY K 8644 show opposite effects on Ca channels.. Eur. J. Pharmacol., 1985. [DOI | PubMed]
- I. M. Mintz, V. J. Venema, K. M. Swiderek, T. D. Lee, B. P. Bean, M. E. Adams. P-type calcium channels blocked by the spider toxin omega-Aga-IVA.. Nature, 1992. [DOI | PubMed]
- C. M. Santi, F. S. Cayabyab, K. G. Sutton, J. E. McRory, J. Mezeyova, K. S. Hamming, D. Parker, A. Stea, T. P. Snutch. Differential inhibition of T-type calcium channels by neuroleptics.. J. Neurosci., 2002. [DOI | PubMed]
- H. K. Rim, H. W. Lee, I. S. Choi, J. Y. Park, H. W. Choi, J. H. Choi, Y. W. Cho, J. Y. Lee, K. T. Lee. T-type Ca2+ channel blocker, KYS05047 induces G1 phase cell cycle arrest by decreasing intracellular Ca2+ levels in human lung adenocarcinoma A549 cells.. Bioorg. Med. Chem. Lett., 2012. [DOI | PubMed]
- K. H. Kim, D. Kim, J. Y. Park, H. J. Jung, Y. H. Cho, H. K. Kim, J. Han, K. Y. Choi, H. J. Kwon. NNC 55–0396, a T-type Ca2+ channel inhibitor, inhibits angiogenesis via suppression of hypoxia-inducible factor-1alpha signal transduction.. J. Mol. Med., 2015. [DOI | PubMed]
- J. Yoshida, T. Ishibashi, M. Nishio. G1 cell cycle arrest by amlodipine, a dihydropyridine Ca2+ channel blocker, in human epidermoid carcinoma A431 cells.. Biochem. Pharmacol., 2007. [DOI | PubMed]
- N. Diaz-Lezama, M. Hernandez-Elvira, A. Sandoval, A. Monroy, R. Felix, E. Monjaraz. Ghrelin inhibits proliferation and increases T-type Ca2+ channel expression in PC-3 human prostate carcinoma cells.. Biochem. Biophys. Res. Commun., 2010. [DOI | PubMed]
- J. L. Harper, C. S. Camerini-Otero, A. H. Li, S. A. Kim, K. A. Jacobson, J. W. Daly. Dihydropyridines as inhibitors of capacitative calcium entry in leukemic HL-60 cells.. Biochem. Pharmacol., 2003. [DOI | PubMed]
- J. T. Smyth, W. I. Dehaven, G. S. Bird, J. W. Putney. Ca2+-store-dependent and -independent reversal of Stim1 localization and function.. J. Cell Sci., 2008. [DOI | PubMed]
- A. Kondratskyi, M. Yassine, C. Slomianny, K. Kondratska, D. Gordienko, E. Dewailly, V. Lehen’kyi, R. Skryma, N. Prevarskaya. Identification of ML-9 as a lysosomotropic agent targeting autophagy and cell death.. Cell Death Dis., 2014. [DOI | PubMed]
- J. Y. Wang, J. Sun, M. Y. Huang, Y. S. Wang, M. F. Hou, Y. Sun, H. He, N. Krishna, S. J. Chiu, S. Lin. STIM1 overexpression promotes colorectal cancer progression, cell motility and COX-2 expression.. Oncogene, 2015. [DOI | PubMed]
- K. Hill, S. McNulty, A. D. Randall. Inhibition of TRPM2 channels by the antifungal agents clotrimazole and econazole.. Naunyn. Schmiedebergs Arch. Pharmacol., 2004. [DOI | PubMed]
- S. N. Wu, H. F. Li, C. R. Jan, A. Y. Shen. Inhibition of Ca2+-activated K+ current by clotrimazole in rat anterior pituitary GH3 cells.. Neuropharmacology, 1999. [DOI | PubMed]
- L. De Petrocellis, A. Ligresti, A. S. Moriello, M. Allara, T. Bisogno, S. Petrosino, C. G. Stott, V. Di Marzo. Effects of cannabinoids and cannabinoid-enriched Cannabis extracts on TRP channels and endocannabinoid metabolic enzymes.. Br. J. Pharmacol., 2011. [DOI | PubMed]
- N. S. Yee. Roles of TRPM8 Ion Channels in Cancer: Proliferation, Survival, and Invasion.. Cancers, 2015. [DOI | PubMed]
- M. C. Almeida, T. Hew-Butler, R. N. Soriano, S. Rao, W. Wang, J. Wang, N. Tamayo, D. L. Oliveira, T. B. Nucci, P. Aryal. Pharmacological blockade of the cold receptor TRPM8 attenuates autonomic and behavioral cold defenses and decreases deep body temperature.. J. Neurosci., 2012. [DOI | PubMed]
- G. R. Seabrook, K. G. Sutton, W. Jarolimek, G. J. Hollingworth, S. Teague, J. Webb, N. Clark, S. Boyce, J. Kerby, Z. Ali. Functional properties of the high-affinity TRPV1 (VR1) vanilloid receptor antagonist (4-hydroxy-5-iodo-3-methoxyphenylacetate ester) iodo-resiniferatoxin.. J. Pharmacol. Exp. Ther., 2002. [DOI | PubMed]
- M. Tominaga, M. J. Caterina, A. B. Malmberg, T. A. Rosen, H. Gilbert, K. Skinner, B. E. Raumann, A. I. Basbaum, D. Julius. The cloned capsaicin receptor integrates multiple pain-producing stimuli.. Neuron, 1998. [DOI | PubMed]
- F. Yang, X. Xiao, W. Cheng, W. Yang, P. Yu, Z. Song, V. Yarov-Yarovoy, J. Zheng. Structural mechanism underlying capsaicin binding and activation of the TRPV1 ion channel.. Nat. Chem. Biol., 2015. [DOI | PubMed]
- T. Liu, G. Wang, H. Tao, Z. Yang, Y. Wang, Z. Meng, R. Cao, Y. Xiao, X. Wang, J. Zhou. Capsaicin mediates caspases activation and induces apoptosis through P38 and JNK MAPK pathways in human renal carcinoma.. BMC Cancer, 2016. [DOI | PubMed]
- P. R. de Jong, N. Takahashi, A. R. Harris, J. Lee, S. Bertin, J. Jeffries, M. Jung, J. Duong, A. I. Triano, J. Lee. Ion channel TRPV1-dependent activation of PTP1B suppresses EGFR-associated intestinal tumorigenesis.. J. Clin. Invest., 2014. [DOI | PubMed]
- A. Peralvarez-Marin, P. Donate-Macian, R. Gaudet. What do we know about the transient receptor potential vanilloid 2 (TRPV2) ion channel?.. FEBS J., 2013. [DOI | PubMed]
- C. V. Bowen, D. DeBay, H. S. Ewart, P. Gallant, S. Gormley, T. T. Ilenchuk, U. Iqbal, T. Lutes, M. Martina, G. Mealing. In vivo detection of human TRPV6-rich tumors with anti-cancer peptides derived from soricidin.. PLoS One, 2013. [DOI | PubMed]
- O. Thastrup, P. J. Cullen, B. K. Drobak, M. R. Hanley, A. P. Dawson. Thapsigargin, a tumor promoter, discharges intracellular Ca2+ stores by specific inhibition of the endoplasmic reticulum Ca2(+)-ATPase.. Proc. Natl. Acad. Sci. U.S.A., 1990. [DOI | PubMed]
- S. R. Denmeade, A. M. Mhaka, D. M. Rosen, W. N. Brennen, S. Dalrymple, I. Dach, C. Olesen, B. Gurel, A. M. DeMarzo, G. Wilding, M. A. Carducci, C. A. Dionne, J. V. Møller, P. Nissen, S. B.øg. Christensen, J. T. Isaacs. Engineering a prostate-specific membrane antigen-activated tumor endothelial cell prodrug for cancer therapy.. Sci. Transl. Med., 2012. [DOI]
- G. R. Monteith, D. McAndrew, H. M. Faddy, S. J. Roberts-Thomson. Calcium and cancer: targeting Ca2+ transport.. Nat. Rev. Cancer, 2007. [DOI | PubMed]
- L. M. Coussens, Z. Werb. Inflammation and cancer.. Nature, 2002. [DOI | PubMed]
- S. Maschalidi, P. Nunes-Hasler, C. R Nascimento, I. Sallent, V. Lannoy, M. Garfa-Traore, N. Cagnard, F. E. Sepulveda, P. Vargas, A.-M. Lennon-Dumenil, P. van Endert, T. Capiod, N. Demaurex, G. Darrasse-Jeze, B. Manoury. UNC93B1 interacts with the calcium sensor STIM1 for efficient antigen cross-presentation in dendritic cells.. Nat. Commun., 2017. [DOI | PubMed]
- M. Bretou, P. J. Saez, D. Sanseau, M. Maurin, D. Lankar, M. Chabaud, C. Spampanato, O. Malbec, L. Barbier, S. Muallem, P. Maiuri, A. Ballabio, J. Helft, M. Piel, P. Vargas, A.-M. Lennon-Dumenil. Lysosome signaling controls the migration of dendritic cells.. Sci. Immunol., 2017. [DOI | PubMed]
- E. A. Emmel, C. L. Verweij, D. B. Durand, K. M. Higgins, E. Lacy, G. R. Crabtree. Cyclosporin A specifically inhibits function of nuclear proteins involved in T cell activation.. Science, 1989. [DOI | PubMed]
- W. M. Flanagan, B. Corthesy, R. J. Bram, G. R. Crabtree. Nuclear association of a T-cell transcription factor blocked by FK-506 and cyclosporin A.. Nature, 1991. [DOI | PubMed]
- G. R. Crabtree, E. N. Olson. NFAT signaling: choreographing the social lives of cells.. Cell, 2002. [DOI | PubMed]
- Y. Kida. Efficacy and safety of sirolimus in lymphangioleiomyomatosis.. N. Engl. J. Med., 2011. [DOI]
- C. D. Burger. Efficacy and safety of sirolimus in lymphangioleiomyomatosis.. N. Engl. J. Med., 2011. [DOI]
- F. X. McCormack, Y. Inoue, J. Moss, L. G. Singer, C. Strange, K. Nakata, A. F. Barker, J. T. Chapman, M. L. Brantly, J. M. Stocks. Efficacy and safety of sirolimus in lymphangioleiomyomatosis.. N. Engl. J. Med., 2011. [DOI | PubMed]
- J. J. Augustine, K. A. Bodziak, D. E. Hricik. Use of sirolimus in solid organ transplantation.. Drugs, 2007. [DOI | PubMed]
- H.-Y. Wen, J. Wang, S.-X. Zhang, J. Luo, X.-C. Zhao, C. Zhang, C.-H. Wang, F.-Y. Hu, X.-J. Zheng, T. Cheng, H.-Q. Niu, G.-Y. Liu, W.-X. Yang, N.-N. Yu, J.-L. Ru, Q.-X. Chen, X.-C. Lu, P.-F. He, C. Gao, X.-F. Li. Low-Dose Sirolimus Immunoregulation Therapy in Patients with Active Rheumatoid Arthritis: A 24-Week Follow-Up of the Randomized, Open-Label.. Parallel-Controlled Trial. J. Immunol. Res., 2019. [DOI | PubMed]
- P. J. Miach. Cyclosporin A in organ transplantation.. Med. J. Aust., 1986. [DOI | PubMed]
- S. M. Flechner. Cyclosporine: a new and promising immunosuppressive agent.. Urol. Clin. North Am., 1983. [DOI | PubMed]
- C. Jennings, B. Kusler, P. P. Jones. Calcineurin inactivation leads to decreased responsiveness to LPS in macrophages and dendritic cells and protects against LPS-induced toxicity in vivo.. Innate Immun., 2009. [DOI | PubMed]
- R. L. Martin, J. H. Lee, L. L. Cribbs, E. Perez-Reyes, D. A. Hanck. Mibefradil block of cloned T-type calcium channels.. J. Pharmacol. Exp. Ther., 2000. [PubMed]
- W. Wang, Q. Gao, M. Yang, X. Zhang, L. Yu, M. Lawas, X. Li, M. Bryant-Genevier, N. T. Southall, J. Marugan, M. Ferrer, H. Xu. Up-regulation of lysosomal TRPML1 channels is essential for lysosomal adaptation to nutrient starvation.. Proc. Natl. Acad. Sci. U.S.A., 2015. [DOI | PubMed]
- E. F. Nemeth, D. Shoback. Calcimimetic and calcilytic drugs for treating bone and mineral-related disorders.. Best Pract. Res. Clin. Endocrinol. Metab., 2013. [DOI | PubMed]
- S. R. Mulay, O. P. Kulkarni, K. V. Rupanagudi, A. Migliorini, M. N. Darisipudi, A. Vilaysane, D. Muruve, Y. Shi, F. Munro, H. Liapis. Calcium oxalate crystals induce renal inflammation by NLRP3-mediated IL-1beta secretion.. J. Clin. Invest., 2013. [DOI | PubMed]
- D. Foell, M. Frosch, C. Sorg, J. Roth. Phagocyte-specific calcium-binding S100 proteins as clinical laboratory markers of inflammation.. Clin. Chim. Acta, 2004. [DOI | PubMed]
- H. G. Roggenkamp, I. Khansahib, L. C. Hernandez, Y. Zhang, D. Lodygin, A. Kruger, F. Gu, F. Mockl, A. Lohndorf, V. Wolters, D. Woike, A. Rosche, A. Bauche, D. Schetelig, R. Werner, H. Schluter, A. V. Failla, C. Meier, R. Fliegert, T. F. Walseth, A. Flugel, B.-P. Diercks, A. H. Guse. HN1L/JPT2: A signaling protein that connects NAADP generation to Ca(2+) microdomain formation.. Sci. Signal, 2021. [DOI | PubMed]
- M. J. Berridge. Calcium microdomains: organization and function.. Cell Calcium, 2006. [DOI | PubMed]
- V. J. Brock, I. M. A. Wolf, M. Er-Lukowiak, N. Lory, T. Stahler, L.-M. Woelk, H.-W. Mittrucker, C. E. Muller, F. Koch-Nolte, B. Rissiek, R. Werner, A. H. Guse, B.-P. Diercks. P2X4 and P2X7 are essential players in basal T cell activity and Ca(2+) signaling milliseconds after T cell activation.. Sci. Adv., 2022. [DOI | PubMed]
- M. Nawrocki, N. Lory, T. Bedke, F. Stumme, B. P. Diercks, A. H. Guse, C. Meier, N. Gagliani, H. W. Mittrucker, S. Huber. Trans-Ned 19-Mediated Antagonism of Nicotinic Acid Adenine Nucleotide-Mediated Calcium Signaling Regulates Th17 Cell Plasticity in Mice.. Cells, 2021. [DOI | PubMed]
- W. A. Catterall. Voltage-gated calcium channels.. Cold Spring Harb. Perspect. Biol., 2011. [DOI | PubMed]
- E. Bahar, H. Kim, H. Yoon. ER Stress-Mediated Signaling: Action Potential and Ca(2+) as Key Players.. Int. J. Mol. Sci., 2016. [DOI | PubMed]
- M. J. Berridge. Neuronal calcium signaling.. Neuron, 1998. [DOI | PubMed]
- M. J. Berridge. Unlocking the secrets of cell signaling.. Annu. Rev. Physiol., 2005. [DOI | PubMed]
- M. J. Berridge, P. Lipp, M. D. Bootman. The versatility and universality of calcium signalling.. Nat. Rev. Mol. Cell Biol., 2000. [DOI | PubMed]
- H. Bading. Transcription-dependent neuronal plasticity: The nuclear calcium hypothesis.. Eur. J. Biochem., 2000. [DOI | PubMed]
- O. Caillard, H. Moreno, B. Schwaller, I. Llano, M. R. Celio, A. Marty. Role of the calcium-binding protein parvalbumin in short-term synaptic plasticity.. Proc. Natl. Acad. Sci. U.S.A., 2000. [DOI | PubMed]
- G. E. Gibson, C. Peterson. Calcium and the aging nervous system.. Neurobiol. Aging, 1987. [DOI | PubMed]
- G. Zündorf, G. Reiser. Calcium dysregulation and homeostasis of neural calcium in the molecular mechanisms of neurodegenerative diseases provide multiple targets for neuroprotection.. Antioxid. Redox. Signal, 2011. [DOI | PubMed]
- M. P. Mattson. Calcium and neurodegeneration.. Aging Cell, 2007. [DOI | PubMed]
- V. A. Vigont, O. A. Zimina, L. N. Glushankova, J. A. Kolobkova, M. A. Ryazantseva, G. N. Mozhayeva, E. V. Kaznacheyeva. STIM1 Protein Activates Store-Operated Calcium Channels in Cellular Model of Huntington’s Disease.. Acta Naturae, 2014. [DOI | PubMed]
- J. K. O’Hare, K. C. Gonzalez, S. A. Herrlinger, Y. Hirabayashi, V. L. Hewitt, H. Blockus, M. Szoboszlay, S. V. Rolotti, T. C. Geiller, A. Negrean, V. Chelur, F. Polleux, A. Losonczy. Compartment-specific tuning of dendritic feature selectivity by intracellular Ca(2+) release.. Science, 2022. [DOI | PubMed]
- M. P. Mattson, F. M. LaFerla, S. L. Chan, M. A. Leissring, P. N. Shepel, J. D. Geiger. Calcium signaling in the ER: its role in neuronal plasticity and neurodegenerative disorders.. Trends Neurosci., 2000. [DOI | PubMed]
- I. Zimanyi, E. Buck, J. J. Abramson, M. M. Mack, I. N. Pessah. Ryanodine induces persistent inactivation of the Ca2+ release channel from skeletal muscle sarcoplasmic reticulum.. Mol. Pharmacol., 1992. [PubMed]
- V. Juvin, A. Penna, J. Chemin, Y. L. Lin, F. A. Rassendren. Pharmacological characterization and molecular determinants of the activation of transient receptor potential V2 channel orthologs by 2-aminoethoxydiphenyl borate.. Mol. Pharmacol., 2007. [DOI | PubMed]
- F. G. Perabo, A. Wirger, S. Kamp, H. Lindner, D. H. Schmidt, S. C. Muller, E. C. Kohn. Carboxyamido-triazole (CAI), a signal transduction inhibitor induces growth inhibition and apoptosis in bladder cancer cells by modulation of Bcl-2.. Anticancer Res., 2004. [PubMed]
- J. P. Dutcher, L. Leon, J. Manola, D. M. Friedland, B. Roth, G. Wilding. Phase II study of carboxyamidotriazole in patients with advanced renal cell carcinoma refractory to immunotherapy: E4896, an Eastern Cooperative Oncology Group Study.. Cancer, 2005. [DOI | PubMed]
- T. W. Moody, J. Chiles, E. Moody, G. J. Sieczkiewicz, E. C. Kohn. CAI inhibits the growth of small cell lung cancer cells.. Lung Cancer, 2003. [DOI | PubMed]
- B. Costa, G. Giagnoni, C. Franke, A. E. Trovato, M. Colleoni. Vanilloid TRPV1 receptor mediates the antihyperalgesic effect of the nonpsychoactive cannabinoid, cannabidiol, in a rat model of acute inflammation.. Br. J. Pharmacol., 2004. [DOI | PubMed]
- G. Velasco, S. Hernandez-Tiedra, D. Davila, M. Lorente. The use of cannabinoids as anticancer agents.. Prog. Neuropsychopharmacol. Biol. Psychiatry, 2016. [DOI | PubMed]
- P. Wahl, C. Foged, S. Tullin, C. Thomsen. Iodo-resiniferatoxin, a new potent vanilloid receptor antagonist.. Mol. Pharmacol., 2001. [DOI]
- C. B. Gonzales, N. B. Kirma, J. J. De La Chapa, R. Chen, M. A. Henry, S. Luo, K. M. Hargreaves. Vanilloids induce oral cancer apoptosis independent of TRPV1.. Oral. Oncol., 2014. [DOI | PubMed]
- Y. Zhu, Y. Lu, C. Qu, M. Miller, J. Tian, D. P. Thakur, J. Zhu, Z. Deng, X. Hu, M. Wu. Identification and optimization of 2-aminobenzimidazole derivatives as novel inhibitors of TRPC4 and TRPC5 channels.. Br. J. Pharmacol., 2015. [DOI | PubMed]
- B. E. Bierer, P. K. Somers, T. J. Wandless, S. J. Burakoff, S. L. Schreiber. Probing immunosuppressant action with a nonnatural immunophilin ligand.. Science, 1990. [DOI | PubMed]
- R. Kraft, C. Grimm, H. Frenzel, C. Harteneck. Inhibition of TRPM2 cation channels by N-(p-amylcinnamoyl)anthranilic acid.. Br. J. Pharmacol., 2006. [DOI | PubMed]
- B. Beck, G. Bidaux, A. Bavencoffe, L. Lemonnier, S. Thebault, Y. Shuba, G. Barrit, R. Skryma, N. Prevarskaya. Prospects for prostate cancer imaging and therapy using high-affinity TRPM8 activators.. Cell Calcium, 2007. [DOI | PubMed]
- A. Weil, S. E. Moore, N. J. Waite, A. Randall, M. J. Gunthorpe. Conservation of functional and pharmacological properties in the distantly related temperature sensors TRVP1 and TRPM8.. Mol. Pharmacol., 2005. [DOI | PubMed]
- H. J. Behrendt, T. Germann, C. Gillen, H. Hatt, R. Jostock. Characterization of the mouse cold-menthol receptor TRPM8 and vanilloid receptor type-1 VR1 using a fluorometric imaging plate reader (FLIPR) assay.. Br. J. Pharmacol., 2004. [DOI | PubMed]
- A. M. Peier, A. Moqrich, A. C. Hergarden, A. J. Reeve, D. A. Andersson, G. M. Story, T. J. Earley, I. Dragoni, P. McIntyre, S. Bevan. A TRP channel that senses cold stimuli and menthol.. Cell, 2002. [DOI | PubMed]
- Y. Liu, N. Qin. TRPM8 in health and disease: cold sensing and beyond.. Adv. Exp. Med. Biol., 2011. [DOI | PubMed]
- A. Tolcher, A. Patnaik, K. Papadopoulos, T. Mays, T. Stephan, D. J. Humble, M. W. Frohlich, R. B. Sims. 376 Preliminary results from a Phase 1 study of D-3263 HCl, a TRPM8 calcium channel agonist, in patients with advanced cancer.. Eur. J. Cancer Supp., 2010. [DOI]
- C. C. Chen, M. Keller, M. Hess, R. Schiffmann, N. Urban, A. Wolfgardt, M. Schaefer, F. Bracher, M. Biel, C. Wahl-Schott. A small molecule restores function to TRPML1 mutant isoforms responsible for mucolipidosis type IV.. Nat. Commun., 2014. [DOI | PubMed]
- Z. Olah, K. Josvay, L. Pecze, T. Letoha, N. Babai, D. Budai, F. Otvos, S. Szalma, C. Vizler. Anti-calmodulins and tricyclic adjuvants in pain therapy block the TRPV1 channel.. PLoS One, 2007. [DOI | PubMed]
- D. Smart, M. J. Gunthorpe, J. C. Jerman, S. Nasir, J. Gray, A. I. Muir, J. K. Chambers, A. D. Randall, J. B. Davis. The endogenous lipid anandamide is a full agonist at the human vanilloid receptor (hVR1).. Br. J. Pharmacol., 2000. [DOI | PubMed]
- A. Szallasi, P. M. Blumberg. Resiniferatoxin, a phorbol-related diterpene, acts as an ultrapotent analog of capsaicin, the irritant constituent in red pepper.. Neuroscience, 1989. [DOI | PubMed]
- L. Nie, Y. Oishi, I. Doi, H. Shibata, I. Kojima. Inhibition of proliferation of MCF-7 breast cancer cells by a blocker of Ca(2+)-permeable channel.. Cell Calcium, 1997. [DOI | PubMed]
- M. K. Chung, A. D. Guler, M. J. Caterina. Biphasic currents evoked by chemical or thermal activation of the heat-gated ion channel, TRPV3.. J. Biol. Chem., 2005. [DOI | PubMed]
- A. Moqrich, S. W. Hwang, T. J. Earley, M. J. Petrus, A. N. Murray, K. S. Spencer, M. Andahazy, G. M. Story, A. Patapoutian. Impaired thermosensation in mice lacking TRPV3, a heat and camphor sensor in the skin.. Science, 2005. [DOI | PubMed]
- H. Xu, N. T. Blair, D. E. Clapham. Camphor activates and strongly desensitizes the transient receptor potential vanilloid subtype 1 channel in a vanilloid-independent mechanism.. J. Neurosci., 2005. [DOI | PubMed]
- H. Xu, M. Delling, J. C. Jun, D. E. Clapham. Oregano, thyme and clove-derived flavors and skin sensitizers activate specific TRP channels.. Nat. Neurosci., 2006. [DOI | PubMed]
- P. L. Smith, K. N. Maloney, R. G. Pothen, J. Clardy, D. E. Clapham. Bisandrographolide from Andrographis paniculata activates TRPV4 channels.. J. Biol. Chem., 2006. [DOI | PubMed]
- R. K. Adapala, R. J. Thoppil, K. Ghosh, H. C. Cappelli, A. C. Dudley, S. Paruchuri, V. Keshamouni, M. Klagsbrun, J. G. Meszaros, W. M. Chilian. Activation of mechanosensitive ion channel TRPV4 normalizes tumor vasculature and improves cancer therapy.. Oncogene, 2016. [DOI | PubMed]
- K. S. Thorneloe, A. C. Sulpizio, Z. Lin, D. J. Figueroa, A. K. Clouse, G. P. McCafferty, T. P. Chendrimada, E. S. Lashinger, E. Gordon, L. Evans. N-((1S)-1-[4-((2S)-2-[(2,4-dichlorophenyl)sulfonyl]amino-3-hydroxypropanoyl)-1-piperazinyl]carbonyl-3-methylbutyl)-1-benzothiophene-2-carboxamide (GSK1016790A), a novel and potent transient receptor potential vanilloid 4 channel agonist induces urinary bladder contraction and hyperactivity: Part I.. J. Pharmacol. Exp. Ther., 2008. [DOI | PubMed]
- K. S. Thorneloe, W. Bao, H. Alsaid, M.-Y. Jian, M. Costell, K. Maniscalco, A. Olzinski, E. Gordon, I. Lozinskaya, L. Elefante. Abstract 13510: Discovery of Orally Active Transient Receptor Potential Vanilloid 4 (TRPV4) Blockers for the Treatment of Pulmonary Edema in Heart Failure.. Circulation, 2018
- Z. L. Wei, M. T. Nguyen, D. J. O’Mahony, A. Acevedo, S. Zipfel, Q. Zhang, L. Liu, M. Dourado, C. Chi, V. Yip. Identification of orally-bioavailable antagonists of the TRPV4 ion-channel.. Bioorg. Med. Chem. Lett., 2015. [DOI | PubMed]
- L. P. He, T. Hewavitharana, J. Soboloff, M. A. Spassova, D. L. Gill. A functional link between store-operated and TRPC channels revealed by the 3,5-bis(trifluoromethyl)pyrazole derivative, BTP2.. J. Biol. Chem., 2005. [DOI | PubMed]
- R. Takezawa, H. Cheng, A. Beck, J. Ishikawa, P. Launay, H. Kubota, J. P. Kinet, A. Fleig, T. Yamada, R. Penner. A pyrazole derivative potently inhibits lymphocyte Ca2+ influx and cytokine production by facilitating transient receptor potential melastatin 4 channel activity.. Mol. Pharmacol., 2006. [DOI | PubMed]
- C. P. Landowski, K. A. Bolanz, Y. Suzuki, M. A. Hediger. Chemical inhibitors of the calcium entry channel TRPV6.. Pharm. Res., 2011. [DOI | PubMed]
- D. M. Haverstick, T. N. Heady, T. L. Macdonald, L. S. Gray. Inhibition of human prostate cancer proliferation in vitro and in a mouse model by a compound synthesized to block Ca2+ entry.. Cancer Res., 2000. [PubMed]
- R. Dasari, A. De Carvalho, D. C. Medellin, K. N. Middleton, F. Hague, M. N. Volmar, L. V. Frolova, M. F. Rossato, J. J. De La Chapa, N. F. Dybdal-Hargreaves. Wittig derivatization of sesquiterpenoid polygodial leads to cytostatic agents with activity against drug resistant cancer cells and capable of pyrrolylation of primary amines.. Eur. J. Med. Chem., 2015. [DOI | PubMed]
- E. Shigetomi, X. Tong, K. Y. Kwan, D. P. Corey, B. S. Khakh. TRPA1 channels regulate astrocyte resting calcium and inhibitory synapse efficacy through GAT-3.. Nat. Neurosci., 2012. [DOI]
- M. E. Holmes, J. Chaudhary, A. K. Grover. Mechanism of action of the novel plasma membrane Ca(2+)-pump inhibitor caloxin.. Cell Calcium, 2003. [DOI | PubMed]
- J. Chaudhary, M. Walia, J. Matharu, E. Escher, A. K. Grover. Caloxin: a novel plasma membrane Ca2+ pump inhibitor.. Am. J. Physiol. Cell Physiol., 2001. [DOI | PubMed]
- A. Muscella, N. Calabriso, C. Vetrugno, F. P. Fanizzi, S. A. De Pascali, C. Storelli, S. Marsigliante. The platinum (II) complex [Pt(O,O’-acac)(gamma-acac)(DMS)] alters the intracellular calcium homeostasis in MCF-7 breast cancer cells.. Biochem. Pharmacol., 2011. [DOI | PubMed]
- D. E. Goeger, R. T. Riley, J. W. Dorner, R. J. Cole. Cyclopiazonic acid inhibition of the Ca2+-transport ATPase in rat skeletal muscle sarcoplasmic reticulum vesicles.. Biochem. Pharmacol., 1988. [DOI | PubMed]
- S. Yao, D. Gallenkamp, K. Wolfel, B. Luke, M. Schindler, J. Scherkenbeck. Synthesis and SERCA activities of structurally simplified cyclopiazonic acid analogues.. Bioorg. Med. Chem., 2011. [DOI | PubMed]
- A. Bergamo, A. Masi, M. A. Jakupec, B. K. Keppler, G. Sava. Inhibitory Effects of the Ruthenium Complex KP1019 in Models of Mammary Cancer Cell Migration and Invasion.. Met. Based Drugs, 2009. [DOI | PubMed]
- V. K. Wong, T. Li, B. Y. Law, E. D. Ma, N. C. Yip, F. Michelangeli, C. K. Law, M. M. Zhang, K. Y. Lam, P. L. Chan. Saikosaponin-d, a novel SERCA inhibitor, induces autophagic cell death in apoptosis-defective cells.. Cell Death Dis., 2013. [DOI | PubMed]
- B. Y. Law, M. Wang, D. L. Ma, F. Al-Mousa, F. Michelangeli, S. H. Cheng, M. H. Ng, K. F. To, A. Y. Mok, R. Y. Ko. Alisol B, a novel inhibitor of the sarcoplasmic/endoplasmic reticulum Ca(2+) ATPase pump, induces autophagy, endoplasmic reticulum stress, and apoptosis.. Mol. Cancer Ther., 2010. [DOI | PubMed]
- B. Yang, M. Zhang, J. Gao, J. Li, L. Fan, G. Xiang, X. Wang, X. Wang, X. Wu, Y. Sun. Small molecule RL71 targets SERCA2 at a novel site in the treatment of human colorectal cancer.. Oncotarget, 2015. [DOI | PubMed]
- J. A. Hwang, M. K. Hwang, Y. Jang, E. J. Lee, J. E. Kim, M. H. Oh, D. J. Shin, S. Lim, G. Ji, U. Oh. 20-O-beta-d-glucopyranosyl-20(S)-protopanaxadiol, a metabolite of ginseng, inhibits colon cancer growth by targeting TRPC channel-mediated calcium influx.. J. Nutr. Biochem., 2013. [DOI | PubMed]
- Y. Akbulut, H. J. Gaunt, K. Muraki, M. J. Ludlow, M. S. Amer, A. Bruns, N. S. Vasudev, L. Radtke, M. Willot, S. Hahn. (−)-Englerin A is a potent and selective activator of TRPC4 and TRPC5 calcium channels.. Angew. Chem., Int. Ed., 2015. [DOI]
- C. Carson, P. Raman, J. Tullai, L. Xu, M. Henault, E. Thomas, S. Yeola, J. Lao, M. McPate, J. M. Verkuyl. Englerin A Agonizes the TRPC4/C5 Cation Channels to Inhibit Tumor Cell Line Proliferation.. PLoS One, 2015. [DOI | PubMed]
- M. Miller, J. Shi, Y. Zhu, M. Kustov, J. B. Tian, A. Stevens, M. Wu, J. Xu, S. Long, P. Yang. Identification of ML204, a novel potent antagonist that selectively modulates native TRPC4/C5 ion channels.. J. Biol. Chem., 2011. [DOI | PubMed]
- K. Seo, P. P. Rainer, V. Shalkey Hahn, D. I. Lee, S. H. Jo, A. Andersen, T. Liu, X. Xu, R. N. Willette, J. J. Lepore. Combined TRPC3 and TRPC6 blockade by selective small-molecule or genetic deletion inhibits pathological cardiac hypertrophy.. Proc. Natl. Acad. Sci. U.S.A., 2014. [DOI | PubMed]
- E. Madan, R. Gogna, B. Keppler, U. Pati. p53 increases intra-cellular calcium release by transcriptional regulation of calcium channel TRPC6 in GaQ3-treated cancer cells.. PLoS One, 2013. [DOI | PubMed]
- A. Aggarwal, J. Hobaus, S. Tennakoon, M. Prinz-Wohlgenannt, J. Graca, S. A. Price, P. Heffeter, W. Berger, S. Baumgartner-Parzer, E. Kallay. Active vitamin D potentiates the anti-neoplastic effects of calcium in the colon: A cross talk through the calcium-sensing receptor.. J. Steroid. Biochem. Mol. Biol., 2016. [DOI | PubMed]
- J. Couper. Sur les effets du peroxyde de manganèse.. J. Chim. Méd. Pharm. Tox., 1837
- T. V. Peres, M. R. Schettinger, P. Chen, F. Carvalho, D. S. Avila, A. B. Bowman, M. Aschner. Manganese-induced neurotoxicity: a review of its behavioral consequences and neuroprotective strategies.. BMC Pharmacol. Toxicol., 2016. [DOI | PubMed]
- P. Dusek, P. M. Roos, T. Litwin, S. A. Schneider, T. P. Flaten, J. Aaseth. The neurotoxicity of iron, copper and manganese in Parkinson’s and Wilson’s diseases.. J. Trace Elem. Med. Biol., 2015. [DOI | PubMed]
- A. C. Martins, P. Morcillo, O. M. Ijomone, V. Venkataramani, F. E. Harrison, E. Lee, A. B. Bowman, M. Aschner. New Insights on the Role of Manganese in Alzheimer’s Disease and Parkinson’s Disease.. Int. J. Environ. Res. Public Health, 2019. [DOI | PubMed]
- C. Au, A. Benedetto, M. Aschner. Manganese transport in eukaryotes: the role of DMT1.. Neurotoxicology, 2008. [DOI | PubMed]
- V. C. Culotta, M. Yang, M. D. Hall. Manganese transport and trafficking: lessons learned from Saccharomyces cerevisiae.. Eukaryot. Cell, 2005. [DOI | PubMed]
- J. Roth, S. Ponzoni, M. Aschner. Manganese homeostasis and transport.. Met. Ions Life Sci., 2013. [DOI | PubMed]
- H. Fujishiro, T. Kambe. Manganese transport in mammals by zinc transporter family proteins, ZNT and ZIP.. J. Pharmacol. Sci., 2022. [DOI | PubMed]
- B. L. Steimle, F. M. Smith, D. J. Kosman. The solute carriers ZIP8 and ZIP14 regulate manganese accumulation in brain microvascular endothelial cells and control brain manganese levels.. J. Biol. Chem., 2019. [DOI | PubMed]
- Q. Ye, J. E. Park, K. Gugnani, S. Betharia, A. Pino-Figueroa, J. Kim. Influence of iron metabolism on manganese transport and toxicity.. Metallomics, 2017. [DOI | PubMed]
- T. E. Gunter, B. Gerstner, K. K. Gunter, J. Malecki, R. Gelein, W. M. Valentine, M. Aschner, D. I. Yule. Manganese transport via the transferrin mechanism.. Neurotoxicology, 2013. [DOI | PubMed]
- K. Abe, Y. Chiba, Y. Nishida. Facile uptake of manganese(III) by apotransferrin: possible origin of manganism.. Z. Naturforsch. C.J. Biosci., 2008. [DOI | PubMed]
- T. A. Zordan, L. G. Hepler. Thermochemistry and oxidation potentials of manganese and its compounds.. Chem. Rev., 1968. [DOI]
- J. Tan, T. Zhang, L. Jiang, J. Chi, D. Hu, Q. Pan, D. Wang, Z. Zhang. Regulation of intracellular manganese homeostasis by Kufor-Rakeb syndrome-associated ATP13A2 protein.. J. Biol. Chem., 2011. [DOI | PubMed]
- C. E. Gavin, K. K. Gunter, T. E. Gunter. Manganese and calcium transport in mitochondria: implications for manganese toxicity.. Neurotoxicology, 1999. [PubMed]
- K. J. Kamer, Y. Sancak, Y. Fomina, J. D. Meisel, D. Chaudhuri, Z. Grabarek, V. K. Mootha. MICU1 imparts the mitochondrial uniporter with the ability to discriminate between Ca(2+) and Mn(2+).. Proc. Natl. Acad. Sci. U.S.A., 2018. [DOI | PubMed]
- Z. Yin, H. Jiang, E. S. Lee, M. Ni, K. M. Erikson, D. Milatovic, A. B. Bowman, M. Aschner. Ferroportin is a manganese-responsive protein that decreases manganese cytotoxicity and accumulation.. J. Neurochem., 2010. [DOI | PubMed]
- C. Liu, T. Jursa, M. Aschner, D. R. Smith, S. Mukhopadhyay. Up-regulation of the manganese transporter SLC30A10 by hypoxia-inducible factors defines a homeostatic response to manganese toxicity.. Proc. Natl. Acad. Sci. U.S.A., 2021. [DOI | PubMed]
- K. C. Gurol, T. Jursa, E. J. Cho, W. Fast, K. N. Dalby, D. R. Smith, S. Mukhopadhyay. PHD2 enzyme is an intracellular manganese sensor that initiates the homeostatic response against elevated manganese.. Proc. Natl. Acad. Sci. U.S.A., 2024. [DOI | PubMed]
- S. Kahali, S. K. Das, R. Kumar, K. Gupta, R. Kundu, B. Bhattacharya, A. Nath, R. Venkatramani, A. Datta. A water-soluble, cell-permeable Mn(ii) sensor enables visualization of manganese dynamics in live mammalian cells.. Chem. Sci., 2024. [DOI | PubMed]
- S. Das, A. Carmona, K. Khatua, F. Porcaro, A. Somogyi, R. Ortega, A. Datta. Manganese Mapping Using a Fluorescent Mn(2+) Sensor and Nanosynchrotron X-ray Fluorescence Reveals the Role of the Golgi Apparatus as a Manganese Storage Site.. Inorg. Chem., 2019. [DOI | PubMed]
- A. K. Holley, V. Bakthavatchalu, J. M. Velez-Roman, D. K. St Clair. Manganese superoxide dismutase: guardian of the powerhouse.. Int. J. Mol. Sci., 2011. [DOI | PubMed]
- J. Azadmanesh, W. E. Lutz, L. Coates, K. L. Weiss, G. E. O. Borgstahl. Direct detection of coupled proton and electron transfers in human manganese superoxide dismutase.. Nat. Commun., 2021. [DOI | PubMed]
- A. Decout, J. D. Katz, S. Venkatraman, A. Ablasser. The cGAS-STING pathway as a therapeutic target in inflammatory diseases.. Nat. Rev. Immunol., 2021. [DOI | PubMed]
- R. M. Hooy, G. Massaccesi, K. E. Rousseau, M. A. Chattergoon, J. Sohn. Allosteric coupling between Mn2+ and dsDNA controls the catalytic efficiency and fidelity of cGAS.. Nucleic Acids Res., 2020. [DOI | PubMed]
- Z. Zhao, Z. Ma, B. Wang, Y. Guan, X. D. Su, Z. Jiang. Mn(2+) Directly Activates cGAS and Structural Analysis Suggests Mn(2+) Induces a Noncanonical Catalytic Synthesis of 2’3′-cGAMP.. Cell Rep., 2020. [DOI | PubMed]
- N. Hempel, P. M. Carrico, J. A. Melendez. Manganese superoxide dismutase (Sod2) and redox-control of signaling events that drive metastasis.. Anticancer Agents Med. Chem., 2011. [DOI | PubMed]
- M. P. Stelling, M. A. Soares, S. C. Cardoso, J. M. Motta, J. C. de Abreu, M. J. M. Antunes, V. G. de Freitas, J. A. Moraes, M. T. L. Castelo-Branco, C. A. Perez, M. S. G. Pavao. Manganese systemic distribution is modulated in vivo during tumor progression and affects tumor cell migration and invasion in vitro.. Sci. Rep., 2021. [DOI | PubMed]
- W. E. Samlowski, R. Petersen, S. Cuzzocrea, H. Macarthur, D. Burton, J. R. McGregor, D. Salvemini. A nonpeptidyl mimic of superoxide dismutase, M40403, inhibits dose-limiting hypotension associated with interleukin-2 and increases its antitumor effects.. Nat. Med., 2003. [DOI | PubMed]
- A. C. Ranganathan, K. K. Nelson, A. M. Rodriguez, K. H. Kim, G. B. Tower, J. L. Rutter, C. E. Brinckerhoff, T. T. Huang, C. J. Epstein, J. J. Jeffrey. Manganese superoxide dismutase signals matrix metalloproteinase expression via H2O2-dependent ERK1/2 activation.. J. Biol. Chem., 2001. [DOI | PubMed]
- D. Ganini, J. H. Santos, M. G. Bonini, R. P. Mason. Switch of Mitochondrial Superoxide Dismutase into a Prooxidant Peroxidase in Manganese-Deficient Cells and Mice.. Cell Chem. Biol., 2018. [DOI | PubMed]
- N. Samson, A. Ablasser. The cGAS-STING pathway and cancer.. Nat. Cancer, 2022. [DOI | PubMed]
- M. Lv, M. Chen, R. Zhang, W. Zhang, C. Wang, Y. Zhang, X. Wei, Y. Guan, J. Liu, K. Feng. Manganese is critical for antitumor immune responses via cGAS-STING and improves the efficacy of clinical immunotherapy.. Cell Res., 2020. [DOI | PubMed]
- X. Sun, Y. Zhang, J. Li, K. S. Park, K. Han, X. Zhou, Y. Xu, J. Nam, J. Xu, X. Shi. Amplifying STING activation by cyclic dinucleotide-manganese particles for local and systemic cancer metalloimmunotherapy.. Nat. Nanotechnol., 2021. [DOI | PubMed]
- Y. Gu, J. Tang, F. Zhang, Y. Qu, M. Zhao, M. Li, Z. Xie, X. Wang, L. Song, Z. Jiang. Manganese potentiates lipopolysaccharide-induced innate immune responses and septic shock.. Int. J. Biol. Macromol., 2023. [DOI | PubMed]
- C. Wang, Y. Guan, M. Lv, R. Zhang, Z. Guo, X. Wei, X. Du, J. Yang, T. Li, Y. Wan. Manganese Increases the Sensitivity of the cGAS-STING Pathway for Double-Stranded DNA and Is Required for the Host Defense against DNA Viruses.. Immunity, 2018. [DOI | PubMed]
- M. F. Gulen, N. Samson, A. Keller, M. Schwabenland, C. Liu, S. Gluck, V. V. Thacker, L. Favre, B. Mangeat, L. J. Kroese. cGAS-STING drives ageing-related inflammation and neurodegeneration.. Nature, 2023. [DOI | PubMed]
- H. Sui, Q. Chen, J. Yang, S. Srirattanapirom, T. Imamichi. Manganese enhances DNA- or RNA-mediated innate immune response by inducing phosphorylation of TANK-binding kinase 1.. iScience, 2022. [DOI | PubMed]
- K. Tuschl, E. Meyer, L. E. Valdivia, N. Zhao, C. Dadswell, A. Abdul-Sada, C. Y. Hung, M. A. Simpson, W. K. Chong, T. S. Jacques, R. L. Woltjer, S. Eaton, A. Gregory, L. Sanford, E. Kara, H. Houlden, S. M. Cuno, H. Prokisch, L. Valletta, V. Tiranti, R. Younis, E. R. Maher, J. Spencer, A. Straatman-Iwanowska, P. Gissen, L. A. M. Selim, G. Pintos-Morell, W. Coroleu-Lletget, S. S. Mohammad, S. Yoganathan, R. C. Dale, M. Thomas, J. Rihel, O. A. Bodamer, C. A. Enns, S. J. Hayflick, P. T. Clayton, P. B. Mills, M. A. Kurian, S. W. Wilson. Mutations in SLC39A14 disrupt manganese homeostasis and cause childhood-onset parkinsonism-dystonia.. Nat. Commun., 2016. [DOI | PubMed]
- G. F. Kwakye, M. M. Paoliello, S. Mukhopadhyay, A. B. Bowman, M. Aschner. Manganese-Induced Parkinsonism and Parkinson’s Disease: Shared and Distinguishable Features.. Int. J. Environ. Res. Public Health, 2015. [DOI | PubMed]
- P. Missy, M. C. Lanhers, Y. Grignon, M. Joyeux, D. Burnel. In vitro and in vivo studies on chelation of manganese.. Hum. Exp. Toxicol., 2000. [DOI | PubMed]
- E. Herrero Hernandez, G. Discalzi, C. Valentini, F. Venturi, A. Chio, C. Carmellino, L. Rossi, A. Sacchetti, E. Pira. Follow-up of patients affected by manganese-induced Parkinsonism after treatment with CaNa2EDTA.. Neurotoxicology, 2006. [DOI | PubMed]
- J. O. Aaseth, V. M. Nurchi. Chelation Combination-A Strategy to Mitigate the Neurotoxicity of Manganese, Iron, and Copper?.. Biomolecules, 2022. [DOI | PubMed]
- W. Zheng, Y. M. Jiang, Y. Zhang, W. Jiang, X. Wang, D. M. Cowan. Chelation therapy of manganese intoxication with para-aminosalicylic acid (PAS) in Sprague-Dawley rats.. Neurotoxicology, 2009. [DOI | PubMed]
- B. Galy, M. Conrad, M. Muckenthaler. Mechanisms controlling cellular and systemic iron homeostasis.. Nat. Rev. Mol. Cell Biol., 2024. [DOI | PubMed]
- N. C. Andrews. Forging a field: the golden age of iron biology.. Blood, 2008. [DOI | PubMed]
- C. Harding, J. Heuser, P. Stahl. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes.. J. Cell Biol., 1983. [DOI | PubMed]
- T. Brabletz, R. Kalluri, M. A. Nieto, R. A. Weinberg. EMT in cancer.. Nat. Rev. Cancer, 2018. [DOI | PubMed]
- S. Müller, F. Sindikubwabo, T. Cañeque, A. Lafon, A. Versini, B. Lombard, D. Loew, T. D. Wu, C. Ginestier, E. Charafe-Jauffret. CD44 regulates epigenetic plasticity by mediating iron endocytosis.. Nat. Chem., 2020. [DOI | PubMed]
- C. A. Eyster, J. D. Higginson, R. Huebner, N. Porat-Shliom, R. Weigert, W. W. Wu, R. F. Shen, J. G. Donaldson. Discovery of new cargo proteins that enter cells through clathrin-independent endocytosis.. Traffic, 2009. [DOI | PubMed]
- K. A. Queisser, R. A. Mellema, E. A. Middleton, I. Portier, B. K. Manne, F. Denorme, E. J. Beswick, M. T. Rondina, R. A. Campbell, A. C. Petrey. COVID-19 generates hyaluronan fragments that directly induce endothelial barrier dysfunction.. JCI Insight, 2021. [DOI | PubMed]
- H. W. Barnes, S. Demirdjian, N. L. Haddock, G. Kaber, H. A. Martinez, N. Nagy, H. Karmouty-Quintana, P. L. Bollyky. Hyaluronan in the pathogenesis of acute and post-acute COVID-19 infection.. Matrix. Biol., 2023. [DOI | PubMed]
- N. Sato, S. Kohi, K. Hirata, M. Goggins. Role of hyaluronan in pancreatic cancer biology and therapy: Once again in the spotlight.. Cancer Sci., 2016. [DOI | PubMed]
- M. A. Jacobetz, D. S. Chan, A. Neesse, T. E. Bapiro, N. Cook, K. K. Frese, C. Feig, T. Nakagawa, M. E. Caldwell, H. I. Zecchini. Hyaluronan impairs vascular function and drug delivery in a mouse model of pancreatic cancer.. Gut, 2013. [DOI | PubMed]
- M. W. Hentze, M. U. Muckenthaler, N. C. Andrews. Balancing acts: molecular control of mammalian iron metabolism.. Cell, 2004. [DOI | PubMed]
- J. Ma, S. Haldar, M. A. Khan, S. D. Sharma, W. C. Merrick, E. C. Theil, D. J. Goss. Fe2+ binds iron responsive element-RNA, selectively changing protein-binding affinities and regulating mRNA repression and activation.. Proc. Natl. Acad. Sci. U.S.A., 2012. [DOI | PubMed]
- P. Ponka, C. Beaumont, D. R. Richardson. Function and regulation of transferrin and ferritin.. Semin. Hematol., 1998. [PubMed]
- M. W. Hentze, L. C. Kuhn. Molecular control of vertebrate iron metabolism: mRNA-based regulatory circuits operated by iron, nitric oxide, and oxidative stress.. Proc. Natl. Acad. Sci. U.S.A., 1996. [DOI | PubMed]
- R. S. Ohgami, D. R. Campagna, A. McDonald, M. D. Fleming. The Steap proteins are metalloreductases.. Blood, 2006. [DOI | PubMed]
- I. Yanatori, Y. Yasui, M. Tabuchi, F. Kishi. Chaperone protein involved in transmembrane transport of iron.. Biochem. J., 2014. [DOI | PubMed]
- M. Plays, S. Müller, R. Rodriguez. Chemistry and biology of ferritin.. Metallomics, 2021. [DOI | PubMed]
- J. Wang, N. Wu, M. Peng, L. Oyang, X. Jiang, Q. Peng, Y. Zhou, Z. He, Q. Liao. Ferritinophagy: research advance and clinical significance in cancers.. Cell Death. Discovery, 2023. [DOI | PubMed]
- J. D. Mancias, X. Wang, S. P. Gygi, J. W. Harper, A. C. Kimmelman. Quantitative proteomics identifies NCOA4 as the cargo receptor mediating ferritinophagy.. Nature, 2014. [DOI | PubMed]
- G. Federico, F. Carrillo, F. Dapporto, M. Chiariello, M. Santoro, R. Bellelli, F. Carlomagno. NCOA4 links iron bioavailability to DNA metabolism.. Cell Rep., 2022. [DOI | PubMed]
- G. C. Shaw, J. J. Cope, L. Li, K. Corson, C. Hersey, G. E. Ackermann, B. Gwynn, A. J. Lambert, R. A. Wingert, D. Traver. Mitoferrin is essential for erythroid iron assimilation.. Nature, 2006. [DOI | PubMed]
- Y. Xue, S. Schmollinger, N. Attar, O. A. Campos, M. Vogelauer, M. F. Carey, S. S. Merchant, S. K. Kurdistani. Endoplasmic reticulum-mitochondria junction is required for iron homeostasis.. J. Biol. Chem., 2017. [DOI | PubMed]
- A. Donovan, A. Brownlie, Y. Zhou, J. Shepard, S. J. Pratt, J. Moynihan, B. H. Paw, A. Drejer, B. Barut, A. Zapata. Positional cloning of zebrafish ferroportin1 identifies a conserved vertebrate iron exporter.. Nature, 2000. [DOI | PubMed]
- I. Yanatori, D. R. Richardson, K. Imada, F. Kishi. Iron Export through the Transporter Ferroportin 1 Is Modulated by the Iron Chaperone PCBP2.. J. Biol. Chem., 2016. [DOI | PubMed]
- C. Pigeon, G. Ilyin, B. Courselaud, P. Leroyer, B. Turlin, P. Brissot, O. Loreal. A new mouse liver-specific gene, encoding a protein homologous to human antimicrobial peptide hepcidin, is overexpressed during iron overload.. J. Biol. Chem., 2001. [DOI | PubMed]
- G. Nicolas, M. Bennoun, I. Devaux, C. Beaumont, B. Grandchamp, A. Kahn, S. Vaulont. Lack of hepcidin gene expression and severe tissue iron overload in upstream stimulatory factor 2 (USF2) knockout mice.. Proc. Natl. Acad. Sci. U.S.A., 2001. [DOI | PubMed]
- A. L. Fisher, C. Y. Wang, Y. Xu, S. Phillips, J. A. Paulo, B. Malachowska, X. Xiao, W. Fendler, J. D. Mancias, J. L. Babitt. Quantitative proteomics and RNA-sequencing of mouse liver endothelial cells identify novel regulators of BMP6 by iron.. iScience, 2023. [DOI | PubMed]
- M. Truman-Rosentsvit, D. Berenbaum, L. Spektor, L. A. Cohen, S. Belizowsky-Moshe, L. Lifshitz, J. Ma, W. Li, E. Kesselman, I. Abutbul-Ionita. Ferritin is secreted via 2 distinct nonclassical vesicular pathways.. Blood, 2018. [DOI | PubMed]
- C. W. Brown, J. J. Amante, P. Chhoy, A. L. Elaimy, H. Liu, L. J. Zhu, C. E. Baer, S. J. Dixon, A. M. Mercurio. Prominin2 Drives Ferroptosis Resistance by Stimulating Iron Export.. Dev. Cell, 2019. [DOI | PubMed]
- A. R. Reddi, I. Hamza. Heme Mobilization in Animals: A Metallolipid’s Journey.. Acc. Chem. Res., 2016. [DOI | PubMed]
- G. L. Wang, B. H. Jiang, E. A. Rue, G. L. Semenza. Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension.. Proc. Natl. Acad. Sci. U.S.A., 1995. [DOI | PubMed]
- G. L. Wang, G. L. Semenza. Purification and characterization of hypoxia-inducible factor 1.. J. Biol. Chem., 1995. [DOI | PubMed]
- C. Peyssonnaux, V. Nizet, R. S. Johnson. Role of the hypoxia inducible factors HIF in iron metabolism.. Cell Cycle, 2008. [DOI | PubMed]
- P. H. Maxwell, M. S. Wiesener, G. W. Chang, S. C. Clifford, E. C. Vaux, M. E. Cockman, C. C. Wykoff, C. W. Pugh, E. R. Maher, P. J. Ratcliffe. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis.. Nature, 1999. [DOI | PubMed]
- M. Ivan, K. Kondo, H. Yang, W. Kim, J. Valiando, M. Ohh, A. Salic, J. M. Asara, W. S. Lane, W. G. Kaelin. HIFalpha targeted for VHL-mediated destruction by proline hydroxylation: implications for O2 sensing.. Science, 2001. [DOI | PubMed]
- P. J. Ratcliffe, J. F. O’Rourke, P. H. Maxwell, C. W. Pugh. Oxygen sensing, hypoxia-inducible factor-1 and the regulation of mammalian gene expression.. J. Exp. Biol., 1998. [DOI | PubMed]
- G. L. Semenza. Hypoxia-inducible factors in physiology and medicine.. Cell, 2012. [DOI | PubMed]
- S. M. Mense, L. Zhang. Heme: a versatile signaling molecule controlling the activities of diverse regulators ranging from transcription factors to MAP kinases.. Cell Res., 2006. [DOI | PubMed]
- S. Hira, T. Tomita, T. Matsui, K. Igarashi, M. Ikeda-Saito. Bach1, a heme-dependent transcription factor, reveals presence of multiple heme binding sites with distinct coordination structure.. IUBMB Life, 2007. [DOI | PubMed]
- K. Ogawa, J. Sun, S. Taketani, O. Nakajima, C. Nishitani, S. Sassa, N. Hayashi, M. Yamamoto, S. Shibahara, H. Fujita. Heme mediates derepression of Maf recognition element through direct binding to transcription repressor Bach1.. EMBO J., 2001. [DOI | PubMed]
- R. Liao, Y. Zheng, X. Liu, Y. Zhang, G. Seim, N. Tanimura, G. M. Wilson, P. Hematti, J. J. Coon, J. Fan. Discovering How Heme Controls Genome Function Through Heme-omics.. Cell Rep., 2020. [DOI | PubMed]
- R. S. Ranu, I. M. London. Regulation of protein synthesis in rabbit reticulocyte lysates: purification and initial characterization of the cyclic 3′:5′-AMP independent protein kinase of the heme-regulated translational inhibitor.. Proc. Natl. Acad. Sci. U.S.A., 1976. [DOI | PubMed]
- R. K. Donegan, C. M. Moore, D. A. Hanna, A. R. Reddi. Handling heme: The mechanisms underlying the movement of heme within and between cells.. Free Radic. Biol. Med., 2019. [DOI | PubMed]
- W. Xu, T. Barrientos, N. C. Andrews. Iron and copper in mitochondrial diseases.. Cell Metab., 2013. [DOI | PubMed]
- A. J. Schwartz, J. W. Goyert, S. Solanki, S. A. Kerk, B. Chen, C. Castillo, P. P. Hsu, B. T. Do, R. Singhal, M. K. Dame. Hepcidin sequesters iron to sustain nucleotide metabolism and mitochondrial function in colorectal cancer epithelial cells.. Nat. Metab., 2021. [DOI | PubMed]
- S. Müller, T. Cañeque, S. Solier, R. Rodriguez. Copper and iron orchestrate cell-state transitions in cancer and immunity.. Trends Cell Biol., 2024. [DOI]
- T. Gerken, C. A. Girard, Y. C. Tung, C. J. Webby, V. Saudek, K. S. Hewitson, G. S. Yeo, M. A. McDonough, S. Cunliffe, L. A. McNeill. The obesity-associated FTO gene encodes a 2-oxoglutarate-dependent nucleic acid demethylase.. Science, 2007. [DOI | PubMed]
- G. Zheng, J. A. Dahl, Y. Niu, P. Fedorcsak, C. M. Huang, C. J. Li, C. B. Vagbo, Y. Shi, W. L. Wang, S. H. Song. ALKBH5 is a mammalian RNA demethylase that impacts RNA metabolism and mouse fertility.. Mol. Cell, 2013. [DOI | PubMed]
- R. J. Klose, E. M. Kallin, Y. Zhang. JmjC-domain-containing proteins and histone demethylation.. Nat. Rev. Genet., 2006. [DOI | PubMed]
- S. M. Kooistra, K. Helin. Molecular mechanisms and potential functions of histone demethylases.. Nat. Rev. Mol. Cell Biol., 2012. [DOI | PubMed]
- X. Jiang, B. Liu, Z. Nie, L. Duan, Q. Xiong, Z. Jin, C. Yang, Y. Chen. The role of m6A modification in the biological functions and diseases.. Signal Transduct. Target Ther., 2021. [DOI | PubMed]
- W. A. Pastor, L. Aravind, A. Rao. TETonic shift: biological roles of TET proteins in DNA demethylation and transcription.. Nat. Rev. Mol. Cell Biol., 2013. [DOI | PubMed]
- I. De Domenico, D. McVey Ward, J. Kaplan. Regulation of iron acquisition and storage: consequences for iron-linked disorders.. Nat. Rev. Mol. Cell Biol., 2008. [DOI | PubMed]
- P. Brissot, A. Pietrangelo, P. C. Adams, B. de Graaff, C. E. McLaren, O. Loreal. Haemochromatosis.. Nat. Rev. Dis. Primers, 2018. [DOI | PubMed]
- S. R. Pasricha, J. Tye-Din, M. U. Muckenthaler, D. W. Swinkels. Iron deficiency.. Lancet, 2021. [DOI | PubMed]
- R. Galanello, R. Origa. Beta-thalassemia.. Orphanet. J. Rare. Dis., 2010. [DOI | PubMed]
- A. Cao, R. Galanello. Beta-thalassemia.. Genet. Med., 2010. [DOI | PubMed]
- R. Origa. beta-Thalassemia.. Genet. Med., 2017. [DOI | PubMed]
- A. T. Taher, K. M. Musallam, M. D. Cappellini. beta-Thalassemias.. N. Engl. J. Med., 2021. [DOI | PubMed]
- M. D. Cappellini, J. Porter, R. Origa, G. L. Forni, E. Voskaridou, F. Galacteros, A. T. Taher, J. B. Arlet, J. A. Ribeil, M. Garbowski. Sotatercept, a novel transforming growth factor beta ligand trap, improves anemia in beta-thalassemia: a phase II, open-label, dose-finding study.. Haematologica, 2019. [DOI | PubMed]
- M. D. Cappellini, V. Viprakasit, A. T. Taher, P. Georgiev, K. H. M. Kuo, T. Coates, E. Voskaridou, H. K. Liew, I. Pazgal-Kobrowski, G. L. Forni. A Phase 3 Trial of Luspatercept in Patients with Transfusion-Dependent beta-Thalassemia.. N. Engl. J. Med., 2020. [DOI | PubMed]
- L. Naldini. Genetic engineering of hematopoiesis: current stage of clinical translation and future perspectives.. EMBO Mol. Med., 2019. [DOI | PubMed]
- B. J. Crielaard, T. Lammers, S. Rivella. Targeting iron metabolism in drug discovery and delivery.. Nat. Rev. Drug Discovery, 2017. [DOI | PubMed]
- T. Chaaban, Y. Mohsen, Z. Ezzeddine, G. Ghssein. Overview of Yersinia pestis Metallophores: Yersiniabactin and Yersinopine.. Biology (Basel), 2023. [DOI | PubMed]
- T. H. Flo, K. D. Smith, S. Sato, D. J. Rodriguez, M. A. Holmes, R. K. Strong, S. Akira, A. Aderem. Lipocalin 2 mediates an innate immune response to bacterial infection by sequestrating iron.. Nature, 2004. [DOI | PubMed]
- S. V. Torti, F. M. Torti. Iron: The cancer connection.. Mol. Aspects Med., 2020. [DOI | PubMed]
- E. Tomat. Targeting iron to contrast cancer progression.. Curr. Opin. Chem. Biol., 2023. [DOI | PubMed]
- M. Schnetz, J. K. Meier, C. Rehwald, C. Mertens, A. Urbschat, E. Tomat, E. A. Akam, P. Baer, F. C. Roos, B. Brune, M. Jung. The Disturbed Iron Phenotype of Tumor Cells and Macrophages in Renal Cell Carcinoma Influences Tumor Growth.. Cancers, 2020. [DOI | PubMed]
- C. H. Chang, J. Qiu, D. O’Sullivan, M. D. Buck, T. Noguchi, J. D. Curtis, Q. Chen, M. Gindin, M. M. Gubin, G. J. van der Windt. Metabolic Competition in the Tumor Microenvironment Is a Driver of Cancer Progression.. Cell, 2015. [DOI | PubMed]
- T. T. Mai, A. Hamai, A. Hienzsch, T. Cañeque, S. Müller, J. Wicinski, O. Cabaud, C. Leroy, A. David, V. Acevedo. Salinomycin kills cancer stem cells by sequestering iron in lysosomes.. Nat. Chem., 2017. [DOI | PubMed]
- D. L. Schonberg, T. E. Miller, Q. Wu, W. A. Flavahan, N. K. Das, J. S. Hale, C. G. Hubert, S. C. Mack, A. M. Jarrar, R. T. Karl. Preferential Iron Trafficking Characterizes Glioblastoma Stem-like Cells.. Cancer Cell, 2015. [DOI | PubMed]
- D. Basuli, L. Tesfay, Z. Deng, B. Paul, Y. Yamamoto, G. Ning, W. Xian, F. McKeon, M. Lynch, C. P. Crum. Iron addiction: a novel therapeutic target in ovarian cancer.. Oncogene, 2017. [DOI | PubMed]
- M. Antoszczak, S. Müller, T. Cañeque, L. Colombeau, N. Dusetti, P. Santofimia-Castano, C. Gaillet, A. Puisieux, J. L. Iovanna, R. Rodriguez. Iron-Sensitive Prodrugs That Trigger Active Ferroptosis in Drug-Tolerant Pancreatic Cancer Cells.. J. Am. Chem. Soc., 2022. [DOI | PubMed]
- R. Rodriguez, S. L. Schreiber, M. Conrad. Persister cancer cells: Iron addiction and vulnerability to ferroptosis.. Mol. Cell, 2022. [DOI | PubMed]
- P. B. Gupta, T. T. Onder, G. Jiang, K. Tao, C. Kuperwasser, R. A. Weinberg, E. S. Lander. Identification of selective inhibitors of cancer stem cells by high-throughput screening.. Cell, 2009. [DOI | PubMed]
- E. F. Paulus, M. Kurz, H. Matter, L. Vértesy. Solid-State and Solution Structure of the Salinomycin–Sodium Complex: Stabilization of Different Conformers for an Ionophore in Different Environments.. J. Am. Chem. .Soc., 1998. [DOI]
- A. Versini, L. Colombeau, A. Hienzsch, C. Gaillet, P. Retailleau, S. Debieu, S. Muller, T. Caneque, R. Rodriguez. Salinomycin Derivatives Kill Breast Cancer Stem Cells by Lysosomal Iron Targeting.. Chemistry, 2020. [DOI | PubMed]
- A. L. Turcu, A. Versini, N. Khene, C. Gaillet, T. Caneque, S. Muller, R. Rodriguez. DMT1 Inhibitors Kill Cancer Stem Cells by Blocking Lysosomal Iron Translocation.. Chemistry, 2020. [DOI | PubMed]
- F. Rizzollo, S. More, P. Vangheluwe, P. Agostinis. The lysosome as a master regulator of iron metabolism.. Trends Biochem. Sci., 2021. [DOI | PubMed]
- C. Legendre, E. Garcion. Iron metabolism: a double-edged sword in the resistance of glioblastoma to therapies.. Trends Endocrinol. Metab., 2015. [DOI | PubMed]
- J. Tsoi, L. Robert, K. Paraiso, C. Galvan, K. M. Sheu, J. Lay, D. J. L. Wong, M. Atefi, R. Shirazi, X. Wang. Multi-stage Differentiation Defines Melanoma Subtypes with Differential Vulnerability to Drug-Induced Iron-Dependent Oxidative Stress.. Cancer Cell, 2018. [DOI | PubMed]
- H. Le Chatelier. Recherches Expérimentales et Théoriques sur les Équilibres Chimiques;, 1888
- C. Thirant, A. Peltier, S. Durand, A. Kramdi, C. Louis-Brennetot, C. Pierre-Eugene, M. Gautier, A. Costa, A. Grelier, S. Zaidi, N. Gruel, I. Jimenez, E. Lapouble, G. Pierron, D. Sitbon, H. J. Brisse, A. Gauthier, P. Freneaux, S. Grossetete, L. G. Baudrin, V. Raynal, S. Baulande, A. Bellini, J. Bhalshankar, A. M. Carcaboso, B. Geoerger, H. Rohrer, D. Surdez, V. Boeva, G. Schleiermacher, O. Delattre, I. Janoueix-Lerosey. Reversible transitions between noradrenergic and mesenchymal tumor identities define cell plasticity in neuroblastoma.. Nat. Commun., 2023. [DOI | PubMed]
- Y. Xia, Y. Li, X. Wu, Q. Zhang, S. Chen, X. Ma, M. Yu. Ironing Out the Details: How Iron Orchestrates Macrophage Polarization.. Front. Immunol., 2021. [DOI | PubMed]
- C. W. Menke-van der Houven van Oordt, C. Gomez-Roca, C. van Herpen, A. L. Coveler, D. Mahalingam, H. M. W. Verheul, W. T. A. van der Graaf, R. Christen, D. Ruttinger, S. Weigand, M. A. Cannarile, F. Heil, M. Brewster, A.-C. Walz, T. K. Nayak, E. Guarin, V. Meresse, C. Le Tourneau. First-in-human phase I clinical trial of RG7356, an anti-CD44 humanized antibody, in patients with advanced, CD44-expressing solid tumors.. Oncotarget, 2016. [DOI | PubMed]
- J. A. Losman, P. Koivunen, W. G. Kaelin. 2-Oxoglutarate-dependent dioxygenases in cancer.. Nat. Rev. Cancer, 2020. [DOI | PubMed]
- G. Millan-Zambrano, A. Burton, A. J. Bannister, R. Schneider. Histone post-translational modifications – cause and consequence of genome function.. Nat. Rev. Genet., 2022. [DOI | PubMed]
- Y. Fu, D. Dominissini, G. Rechavi, C. He. Gene expression regulation mediated through reversible m(6)A RNA methylation.. Nat. Rev. Genet., 2014. [DOI | PubMed]
- L. Kruidenier, C. W. Chung, Z. Cheng, J. Liddle, K. Che, G. Joberty, M. Bantscheff, C. Bountra, A. Bridges, H. Diallo. A selective jumonji H3K27 demethylase inhibitor modulates the proinflammatory macrophage response.. Nature, 2012. [DOI | PubMed]
- B. Heinemann, J. M. Nielsen, H. R. Hudlebusch, M. J. Lees, D. V. Larsen, T. Boesen, M. Labelle, L. O. Gerlach, P. Birk, K. Helin. Inhibition of demethylases by GSK-J1/J4.. Nature, 2014. [DOI | PubMed]
- J. S. Shapiro, H. C. Chang, Y. Tatekoshi, Z. Zhao, Z. S. Waxali, B. J. Hong, H. Chen, J. A. Geier, E. T. Bartom, A. De Jesus. Iron drives anabolic metabolism through active histone demethylation and mTORC1.. Nat. Cell Biol., 2023. [DOI | PubMed]
- S. J. Dixon, K. M. Lemberg, M. R. Lamprecht, R. Skouta, E. M. Zaitsev, C. E. Gleason, D. N. Patel, A. J. Bauer, A. M. Cantley, W. S. Yang. Ferroptosis: an iron-dependent form of nonapoptotic cell death.. Cell, 2012. [DOI | PubMed]
- S. J. Dixon, J. A. Olzmann. The cell biology of ferroptosis.. Nat. Rev. Mol. Cell Biol., 2024. [DOI | PubMed]
- S. Doll, F. P. Freitas, R. Shah, M. Aldrovandi, M. C. da Silva, I. Ingold, A. Goya Grocin, T. N. Xavier da Silva, E. Panzilius, C. H. Scheel. FSP1 is a glutathione-independent ferroptosis suppressor.. Nature, 2019. [DOI | PubMed]
- A. Seiler, M. Schneider, H. Forster, S. Roth, E. K. Wirth, C. Culmsee, N. Plesnila, E. Kremmer, O. Radmark, W. Wurst. Glutathione peroxidase 4 senses and translates oxidative stress into 12/15-lipoxygenase dependent- and AIF-mediated cell death.. Cell Metab., 2008. [DOI | PubMed]
- V. S. Viswanathan, M. J. Ryan, H. D. Dhruv, S. Gill, O. M. Eichhoff, B. Seashore-Ludlow, S. D. Kaffenberger, J. K. Eaton, K. Shimada, A. J. Aguirre. Dependency of a therapy-resistant state of cancer cells on a lipid peroxidase pathway.. Nature, 2017. [DOI | PubMed]
- C. Mao, X. Liu, Y. Zhang, G. Lei, Y. Yan, H. Lee, P. Koppula, S. Wu, L. Zhuang, B. Fang. DHODH-mediated ferroptosis defence is a targetable vulnerability in cancer.. Nature, 2021. [DOI | PubMed]
- K. Bersuker, J. M. Hendricks, Z. Li, L. Magtanong, B. Ford, P. H. Tang, M. A. Roberts, B. Tong, T. J. Maimone, R. Zoncu. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis.. Nature, 2019. [DOI | PubMed]
- S. J Dixon, D. N Patel, M. Welsch, R. Skouta, E. D Lee, M. Hayano, A. G Thomas, C. E Gleason, N. P Tatonetti, B. S Slusher, B. R Stockwell. Pharmacological inhibition of cystine-glutamate exchange induces endoplasmic reticulum stress and ferroptosis.. Elife, 2014. [DOI | PubMed]
- X. Sui, R. Zhang, S. Liu, T. Duan, L. Zhai, M. Zhang, X. Han, Y. Xiang, X. Huang, H. Lin, T. Xie. RSL3 Drives Ferroptosis Through GPX4 Inactivation and ROS Production in Colorectal Cancer.. Front. Pharmacol., 2018. [DOI | PubMed]
- J. P. Friedmann Angeli, M. Schneider, B. Proneth, Y. Y. Tyurina, V. A. Tyurin, V. J. Hammond, N. Herbach, M. Aichler, A. Walch, E. Eggenhofer. Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice.. Nat. Cell Biol., 2014. [DOI | PubMed]
- B. Hassannia, P. Vandenabeele, T. Vanden Berghe. Targeting Ferroptosis to Iron Out Cancer.. Cancer Cell, 2019. [DOI | PubMed]
- H. F. Yan, T. Zou, Q. Z. Tuo, S. Xu, H. Li, A. A. Belaidi, P. Lei. Ferroptosis: mechanisms and links with diseases.. Signal Transduct. Target Ther., 2021. [DOI | PubMed]
- Q. Ma. Role of nrf2 in oxidative stress and toxicity.. Annu. Rev. Pharmacol. Toxicol., 2013. [DOI | PubMed]
- A. Anandhan, M. Dodson, A. Shakya, J. Chen, P. Liu, Y. Wei, H. Tan, Q. Wang, Z. Jiang, K. Yang, J. G. Garcia, S. K. Chambers, E. Chapman, A. Ooi, Y. Yang-Hartwich, B. R. Stockwell, D. D. Zhang. NRF2 controls iron homeostasis and ferroptosis through HERC2 and VAMP8.. Sci. Adv., 2023. [DOI | PubMed]
- Z. Chen, D. Zhang, F. Yue, M. Zheng, Z. Kovacevic, D. R. Richardson. The iron chelators Dp44mT and DFO inhibit TGF-beta-induced epithelial-mesenchymal transition via up-regulation of N-Myc downstream-regulated gene 1 (NDRG1).. J. Biol. Chem., 2012. [DOI | PubMed]
- N. T. Le, D. R. Richardson. Iron chelators with high antiproliferative activity up-regulate the expression of a growth inhibitory and metastasis suppressor gene: a link between iron metabolism and proliferation.. Blood, 2004. [DOI | PubMed]
- P. Muñoz, G. Zavala, K. Castillo, P. Aguirre, C. Hidalgo, M. T. Nuñez. Effect of iron on the activation of the MAPK/ERK pathway in PC12 neuroblastoma cells.. Biol. Res., 2006. [DOI | PubMed]
- Y. Chi, J. Remsik, V. Kiseliovas, C. Derderian, U. Sener, M. Alghader, F. Saadeh, K. Nikishina, T. Bale, C. Iacobuzio-Donahue. Cancer cells deploy lipocalin-2 to collect limiting iron in leptomeningeal metastasis.. Science, 2020. [DOI | PubMed]
- T. Yamasaki, S. Terai, I. Sakaida. Deferoxamine for advanced hepatocellular carcinoma.. N. Engl. J. Med., 2011. [DOI | PubMed]
- G. Y. Lui, P. Obeidy, S. J. Ford, C. Tselepis, D. M. Sharp, P. J. Jansson, D. S. Kalinowski, Z. Kovacevic, D. B. Lovejoy, D. R. Richardson. The iron chelator, deferasirox, as a novel strategy for cancer treatment: oral activity against human lung tumor xenografts and molecular mechanism of action.. Mol. Pharmacol., 2013. [DOI | PubMed]
- T. Fukushima, H. Kawabata, T. Nakamura, H. Iwao, A. Nakajima, M. Miki, T. Sakai, T. Sawaki, Y. Fujita, M. Tanaka. Iron chelation therapy with deferasirox induced complete remission in a patient with chemotherapy-resistant acute monocytic leukemia.. Anticancer Res., 2011. [PubMed]
- R. Rodriguez, T. Caneque, L. Baron, S. Müller, A. Carmona, L. Colombeau, A. Versini, M. Sabatier, J. Sampaio, E. Mishima. Activation of lysosomal iron triggers ferroptosis in cancer.. Res. Sq., 2024. [DOI]
- C. M. Chaparro, P. S. Suchdev. Anemia epidemiology, pathophysiology, and etiology in low- and middle-income countries.. Ann. N.Y. Acad. Sci., 2019. [DOI | PubMed]
- G. L. Semenza. Targeting HIF-1 for cancer therapy.. Nat. Rev. Cancer, 2003. [DOI | PubMed]
- S. V. Torti, F. M. Torti. Iron and cancer: more ore to be mined.. Nat. Rev. Cancer, 2013. [DOI | PubMed]
- M. Bonadonna, S. Altamura, E. Tybl, G. Palais, M. Qatato, M. Polycarpou-Schwarz, M. Schneider, C. Kalk, W. Rudiger, A. Ertl, N. Anstee, R. Bogeska, D. Helm, M. D. Milsom, B. Galy. Iron regulatory protein (IRP)-mediated iron homeostasis is critical for neutrophil development and differentiation in the bone marrow.. Sci. Adv., 2022. [DOI | PubMed]
- Y. R. Kao, J. Chen, R. Kumari, A. Ng, A. Zintiridou, M. Tatiparthy, Y. Ma, M. M. Aivalioti, D. Moulik, S. Sundaravel. An iron rheostat controls hematopoietic stem cell fate.. Cell Stem Cell, 2024. [DOI | PubMed]
- A. L. Hanson, M. P. Mule, H. Ruffieux, F. Mescia, L. Bergamaschi, V. S. Pelly, L. Turner, P. Kotagiri, B. Gottgens, C. Hess, N. Gleadall, J. R. Bradley, J. A. Nathan, P. A. Lyons, H. Drakesmith, K. G. C. Smith. Iron dysregulation and inflammatory stress erythropoiesis associates with long-term outcome of COVID-19.. Nat. Immunol., 2024. [DOI | PubMed]
- M. J. Brookes, J. Boult, K. Roberts, B. T. Cooper, N. A. Hotchin, G. Matthews, T. Iqbal, C. Tselepis. A role for iron in Wnt signalling.. Oncogene, 2008. [DOI | PubMed]
- F. Liu, I. Rehmani, S. Esaki, R. Fu, L. Chen, V. de Serrano, A. Liu. Pirin is an iron-dependent redox regulator of NF-kappaB.. Proc. Natl. Acad. Sci. U.S.A., 2013. [DOI | PubMed]
- Y. Yu, D. R. Richardson. Cellular iron depletion stimulates the JNK and p38 MAPK signaling transduction pathways, dissociation of ASK1-thioredoxin, and activation of ASK1.. J. Biol. Chem., 2011. [DOI | PubMed]
- M. Maus, V. Lopez-Polo, L. Mateo, M. Lafarga, M. Aguilera, E. De Lama, K. Meyer, A. Sola, C. Lopez-Martinez, I. Lopez-Alonso. Iron accumulation drives fibrosis, senescence and the senescence-associated secretory phenotype.. Nat. Metab., 2023. [DOI | PubMed]
- H. Gao, Z. Jin, G. Bandyopadhyay, G. Wang, D. Zhang, K. C. E. Rocha, X. Liu, H. Zhao, T. Kisseleva, D. A. Brenner. Aberrant iron distribution via hepatocyte-stellate cell axis drives liver lipogenesis and fibrosis.. Cell Metab., 2022. [DOI | PubMed]
- R. C. Hider, A. V. Hoffbrand. The Role of Deferiprone in Iron Chelation.. N. Engl. J. Med., 2018. [DOI | PubMed]
- M. Pandolfo, L. Hausmann. Deferiprone for the treatment of Friedreich’s ataxia.. J. Neurochem., 2013. [DOI | PubMed]
- M. Pandolfo, J. Arpa, M. B. Delatycki, K. H. Le Quan Sang, C. Mariotti, A. Munnich, I. Sanz-Gallego, G. Tai, M. A. Tarnopolsky, F. Taroni. Deferiprone in Friedreich ataxia: a 6-month randomized controlled trial.. Ann. Neurol., 2014. [DOI | PubMed]
- S. Levi, M. Ripamonti, A. S. Moro, A. Cozzi. Iron imbalance in neurodegeneration.. Mol. Psychiatry., 2024. [DOI | PubMed]
- R. J. Ward, F. A. Zucca, J. H. Duyn, R. R. Crichton, L. Zecca. The role of iron in brain ageing and neurodegenerative disorders.. Lancet Neurol., 2014. [DOI | PubMed]
- T. A. Rouault. Iron metabolism in the CNS: implications for neurodegenerative diseases.. Nat. Rev. Neurosci., 2013. [DOI | PubMed]
- E. Stephenson, N. Nathoo, Y. Mahjoub, J. F. Dunn, V. W. Yong. Iron in multiple sclerosis: roles in neurodegeneration and repair.. Nat. Rev. Neurol., 2014. [DOI | PubMed]
- L. Xie, W. Zheng, N. Xin, J. W. Xie, T. Wang, Z. Y. Wang. Ebselen inhibits iron-induced tau phosphorylation by attenuating DMT1 up-regulation and cellular iron uptake.. Neurochem. Int., 2012. [DOI | PubMed]
- Y. Xie, Y. Tan, Y. Zheng, X. Du, Q. Liu. Ebselen ameliorates beta-amyloid pathology, tau pathology, and cognitive impairment in triple-transgenic Alzheimer’s disease mice.. J. Biol. Inorg. Chem., 2017. [DOI | PubMed]
- L. Xie, D. Yu, J. Hu, Y. Fang, Z. Zou, Y. Gu, D. Li. DMT1 inhibitor ebselen inhibits iron-induced amyloidogenic APP processing.. Int. J. Clin. Exp. Med., 2018
- F. Boulad, X. Wang, J. Qu, C. Taylor, L. Ferro, G. Karponi, S. Bartido, P. Giardina, G. Heller, S. E. Prockop. Safe mobilization of CD34+ cells in adults with beta-thalassemia and validation of effective globin gene transfer for clinical investigation.. Blood, 2014. [DOI | PubMed]
- M. C. Holmes, A. Reik, E. J. Rebar, J. C. Miller, Y. Zhou, L. Zhang, P. Li, S. Vaidya. A Potential Therapy for Beta-Thalassemia (ST-400) and Sickle Cell Disease (BIVV003).. Blood, 2017. [DOI]
- E. de Dreuzy, K. Bhukhai, P. Leboulch, E. Payen. Current and future alternative therapies for beta-thalassemia major.. Biomed. J., 2016. [DOI | PubMed]
- A. van Vuren, J. L. Kerkhoffs, S. Schols, A. Rijneveld, E. Nur, D. Peereboom, Y. Gandon, P. Welsing, R. van Wijk, R. Schutgens. Proton pump inhibition for secondary hemochromatosis in hereditary anemia: a phase III placebo-controlled randomized cross-over clinical trial.. Am. J. Hematol., 2022. [DOI | PubMed]
- M. McCaleb, J. Lickliter, A. Dibble, E. Schneider, M. Aghajan, S. Guo, S. Hughes, R. Geary, B. P. Monia. Transmembrane Protease, Serine 6 (TMPRSS6) Antisense Oligonucleotide (IONIS-TMPRSS6-LRX) Reduces Plasma Iron Levels of Healthy Volunteers in a Phase 1 Clinical Study.. Blood, 2018. [DOI]
- J. Arezes, N. Foy, K. McHugh, D. Quinkert, S. Benard, A. Sawant, J. N. Frost, A. E. Armitage, S. R. Pasricha, P. J. Lim. Antibodies against the erythroferrone N-terminal domain prevent hepcidin suppression and ameliorate murine thalassemia.. Blood, 2020. [DOI | PubMed]
- F. Richard, J. J. van Lier, B. Roubert, T. Haboubi, U. M. Gohring, F. Durrenberger. Oral ferroportin inhibitor VIT-2763: First-in-human, phase 1 study in healthy volunteers.. Am. J. Hematol., 2020. [DOI | PubMed]
- M. Sheetz, P. Barrington, S. Callies, P. H. Berg, J. McColm, T. Marbury, B. Decker, G. L. Dyas, S. M. E. Truhlar, R. Benschop. Targeting the hepcidin-ferroportin pathway in anaemia of chronic kidney disease.. Br. J. Clin. Pharmacol., 2019. [DOI | PubMed]
- M. Antoszczak, S. Muller, L. Colombeau, T. Caneque, R. Rodriguez. Rapid Access to Ironomycin Derivatives by Click Chemistry.. ACS Org. Inorg. Au., 2022. [DOI | PubMed]
- S. Garciaz, A. A. Guirguis, S. Muller, F. C. Brown, Y. C. Chan, A. Motazedian, C. L. Rowe, J. A. Kuzich, K. L. Chan, K. Tran. Pharmacologic Reduction of Mitochondrial Iron Triggers a Noncanonical BAX/BAK-Dependent Cell Death.. Cancer Discovery, 2022. [DOI | PubMed]
- D. Czerwonka, S. Muller, T. Caneque, L. Colombeau, A. Huczynski, M. Antoszczak, R. Rodriguez. Expeditive Synthesis of Potent C20-epi-Amino Derivatives of Salinomycin against Cancer Stem-Like Cells.. ACS Org. Inorg. Au., 2022. [DOI | PubMed]
- A. Versini, L. Saier, F. Sindikubwabo, S. Müller, T. Cañeque, R. Rodriguez. Chemical biology of salinomycin.. Tetrahedron, 2018. [DOI]
- D. R. Richardson, K. Milnes. The potential of iron chelators of the pyridoxal isonicotinoyl hydrazone class as effective antiproliferative agents II: the mechanism of action of ligands derived from salicylaldehyde benzoyl hydrazone and 2-hydroxy-1-naphthylaldehyde benzoyl hydrazone.. Blood, 1997. [DOI | PubMed]
- S. Song, T. Christova, S. Perusini, S. Alizadeh, R. Y. Bao, B. W. Miller, R. Hurren, Y. Jitkova, M. Gronda, M. Isaac. Wnt inhibitor screen reveals iron dependence of beta-catenin signaling in cancers.. Cancer Res., 2011. [DOI | PubMed]
- Y. Eberhard, S. P. McDermott, X. Wang, M. Gronda, A. Venugopal, T. E. Wood, R. Hurren, A. Datti, R. A. Batey, J. Wrana. Chelation of intracellular iron with the antifungal agent ciclopirox olamine induces cell death in leukemia and myeloma cells.. Blood, 2009. [DOI | PubMed]
- H. Zhou, T. Shen, Y. Luo, L. Liu, W. Chen, B. Xu, X. Han, J. Pang, C. A. Rivera, S. Huang. The antitumor activity of the fungicide ciclopirox.. Int. J. Cancer, 2010. [DOI | PubMed]
- R. D. Abeysinghe, B. T. Greene, R. Haynes, M. C. Willingham, J. Turner, R. P. Planalp, M. W. Brechbiel, F. M. Torti, S. V. Torti. p53-independent apoptosis mediated by tachpyridine, an anti-cancer iron chelator.. Carcinogenesis, 2001. [DOI | PubMed]
- S. V. Torti, F. M. Torti, S. P. Whitman, M. W. Brechbiel, G. Park, R. P. Planalp. Tumor cell cytotoxicity of a novel metal chelator.. Blood, 1998. [DOI | PubMed]
- B. T. Greene, J. Thorburn, M. C. Willingham, A. Thorburn, R. P. Planalp, M. W. Brechbiel, J. Jennings-Gee, J. t. Wilkinson, F. M. Torti, S. V. Torti. Activation of caspase pathways during iron chelator-mediated apoptosis.. J. Biol. Chem., 2002. [DOI | PubMed]
- J. Turner, C. Koumenis, T. E. Kute, R. P. Planalp, M. W. Brechbiel, D. Beardsley, B. Cody, K. D. Brown, F. M. Torti, S. V. Torti. Tachpyridine, a metal chelator, induces G2 cell-cycle arrest, activates checkpoint kinases, and sensitizes cells to ionizing radiation.. Blood, 2005. [DOI | PubMed]
- Z. Kovacevic, S. Chikhani, D. B. Lovejoy, D. R. Richardson. Novel thiosemicarbazone iron chelators induce up-regulation and phosphorylation of the metastasis suppressor N-myc down-stream regulated gene 1: a new strategy for the treatment of pancreatic cancer.. Mol. Pharmacol., 2011. [DOI | PubMed]
- Y. Yu, Y. S. Rahmanto, D. R. Richardson. Bp44mT: an orally active iron chelator of the thiosemicarbazone class with potent anti-tumour efficacy.. Br. J. Pharmacol., 2012. [DOI | PubMed]
- Q. Ba, M. Hao, H. Huang, J. Hou, S. Ge, Z. Zhang, J. Yin, R. Chu, H. Jiang, F. Wang. Iron deprivation suppresses hepatocellular carcinoma growth in experimental studies.. Clin. Cancer Res., 2011. [DOI | PubMed]
- Y. Yen, K. Margolin, J. Doroshow, M. Fishman, B. Johnson, C. Clairmont, D. Sullivan, M. Sznol. A phase I trial of 3-aminopyridine-2-carboxaldehyde thiosemicarbazone in combination with gemcitabine for patients with advanced cancer.. Cancer Chemother. Pharmacol., 2004. [DOI | PubMed]
- J. J. Knox, S. J. Hotte, C. Kollmannsberger, E. Winquist, B. Fisher, E. A. Eisenhauer. Phase II study of Triapine in patients with metastatic renal cell carcinoma: a trial of the National Cancer Institute of Canada Clinical Trials Group (NCIC IND.161).. Invest. New Drugs, 2007. [DOI | PubMed]
- B. Ma, B. C. Goh, E. H. Tan, K. C. Lam, R. Soo, S. S. Leong, L. Z. Wang, F. Mo, A. T. Chan, B. Zee. A multicenter phase II trial of 3-aminopyridine-2-carboxaldehyde thiosemicarbazone (3-AP, Triapine) and gemcitabine in advanced non-small-cell lung cancer with pharmacokinetic evaluation using peripheral blood mononuclear cells.. Invest. New Drugs, 2008. [DOI | PubMed]
- E. Liu, Z. Li, Y. Zhang, K. Chen. Hepcidin Induces M1Macrophage Polarization in Monocytes or THP-1 Derived Macrophages.. Iran. J. Immunol., 2019. [DOI | PubMed]
- L. Renders, K. Budde, C. Rosenberger, R. van Swelm, D. Swinkels, F. Dellanna, W. Feuerer, M. Wen, C. Erley, B. Bader. First-in-human Phase I studies of PRS-080#22, a hepcidin antagonist, in healthy volunteers and patients with chronic kidney disease undergoing hemodialysis.. PLoS One, 2019. [DOI | PubMed]
- S. Vadhan-Raj, R. Abonour, J. W. Goldman, D. A. Smith, C. A. Slapak, R. L. Ilaria, R. V. Tiu, X. Wang, S. Callies, J. Cox, J. L. Tuttle, Y.-K. Lau, E. J. Roeland. A first-in-human phase 1 study of a hepcidin monoclonal antibody, LY2787106, in cancer-associated anemia.. J. Hematol. Oncol., 2017. [DOI | PubMed]
- Y. M. Shah, T. Matsubara, S. Ito, S. H. Yim, F. J. Gonzalez. Intestinal hypoxia-inducible transcription factors are essential for iron absorption following iron deficiency.. Cell Metab., 2009. [DOI | PubMed]
- N. K. Das, A. J. Schwartz, G. Barthel, N. Inohara, Q. Liu, A. Sankar, D. R. Hill, X. Ma, O. Lamberg, M. K. Schnizlein. Microbial Metabolite Signaling Is Required for Systemic Iron Homeostasis.. Cell Metab., 2020. [DOI | PubMed]
- S. Portsmouth, D. van Veenhuyzen, R. Echols, M. Machida, J. C. A. Ferreira, M. Ariyasu, P. Tenke, T. D. Nagata. Cefiderocol versus imipenem-cilastatin for the treatment of complicated urinary tract infections caused by Gram-negative uropathogens: a phase 2, randomised, double-blind, non-inferiority trial.. Lancet Infect. Dis., 2018. [DOI | PubMed]
- J. Vadolas, G. Z. Ng, K. Kysenius, P. J. Crouch, S. Dames, M. Eisermann, T. Nualkaew, S. Vilcassim, U. Schaeper, G. Grigoriadis. SLN124, a GalNac-siRNA targeting transmembrane serine protease 6, in combination with deferiprone therapy reduces ineffective erythropoiesis and hepatic iron-overload in a mouse model of beta-thalassaemia.. Br. J. Hamaetol., 2021. [DOI]
- S. Altamura, U. Schaeper, S. Dames, K. Loffler, M. Eisermann, C. Frauendorf, K. Mudder, J. Neves, M. U. Muckenthaler. SLN124, a GalNAc-siRNA Conjugate Targeting TMPRSS6, Efficiently Prevents Iron Overload in Hereditary Haemochromatosis Type 1.. Hemasphere, 2019. [DOI | PubMed]
- S. Y. Lee. Temozolomide resistance in glioblastoma multiforme.. Genes Dis, 2016. [DOI | PubMed]
- H. Decker, N. Hellmann, E. Jaenicke, B. Lieb, U. Meissner, J. Markl. Minireview: Recent progress in hemocyanin research.. Integr. Comp. Biol., 2007. [DOI | PubMed]
- B. E. Kim, T. Nevitt, D. J. Thiele. Mechanisms for copper acquisition, distribution and regulation.. Nat. Chem. Biol., 2008. [DOI | PubMed]
- A. Dancis, D. S. Yuan, D. Haile, C. Askwith, D. Eide, C. Moehle, J. Kaplan, R. D. Klausner. Molecular characterization of a copper transport protein in S. cerevisiae: an unexpected role for copper in iron transport.. Cell, 1994. [DOI | PubMed]
- C. Lin, Z. Zhang, T. Wang, C. Chen, Y. James Kang. Copper uptake by DMT1: a compensatory mechanism for CTR1 deficiency in human umbilical vein endothelial cells.. Metallomics, 2015. [DOI | PubMed]
- P. V. van den Berghe, D. E. Folmer, H. E. Malingre, E. van Beurden, A. E. Klomp, B. van de Sluis, M. Merkx, R. Berger, L. W. Klomp. Human copper transporter 2 is localized in late endosomes and lysosomes and facilitates cellular copper uptake.. Biochem. J., 2007. [DOI | PubMed]
- Y. Liao, J. Zhao, K. Bulek, F. Tang, X. Chen, G. Cai, S. Jia, P. L. Fox, E. Huang, T. T. Pizarro, M. F. Kalady, M. W. Jackson, S. Bao, G. C. Sen, G. R. Stark, C. J. Chang, X. Li. Inflammation mobilizes copper metabolism to promote colon tumorigenesis via an IL-17-STEAP4-XIAP axis.. Nat. Commun., 2020. [DOI | PubMed]
- R. T. Scarl, C. M. Lawrence, H. M. Gordon, C. S. Nunemaker. STEAP4: its emerging role in metabolism and homeostasis of cellular iron and copper.. J. Endocrinol., 2017. [DOI | PubMed]
- J.-H. Kim, T. Matsubara, J. Lee, C. Fenollar-Ferrer, K. Han, D. Kim, S. Jia, C. J. Chang, H. Yang, T. Nagano, K. W. Krausz, S.-H. Yim, F. J. Gonzalez. Lysosomal SLC46A3 modulates hepatic cytosolic copper homeostasis.. Nat. Commun., 2021. [DOI | PubMed]
- S. Lutsenko, N. L. Barnes, M. Y. Bartee, O. Y. Dmitriev. Function and regulation of human copper-transporting ATPases.. Physiol. Rev., 2007. [DOI | PubMed]
- R. Purohit, M. O. Ross, S. Batelu, A. Kusowski, T. L. Stemmler, B. M. Hoffman, A. C. Rosenzweig. Cu(+)-specific CopB transporter: Revising P(1B)-type ATPase classification.. Proc. Natl. Acad. Sci. U.S.A., 2018. [DOI | PubMed]
- Z. Guo, F. Oradd, V. Bagenholm, C. Grønberg, J. F. Ma, P. Ott, Y. Wang, M. Andersson, P. A. Pedersen, K. Wang, P. Gourdon. Diverse roles of the metal binding domains and transport mechanism of copper transporting P-type ATPases.. Nat. Commun., 2024. [DOI | PubMed]
- E. V. Polishchuk, M. Concilli, S. Iacobacci, G. Chesi, N. Pastore, P. Piccolo, S. Paladino, D. Baldantoni, S. C.D. van IJzendoorn, J. Chan, C. J. Chang, A. Amoresano, F. Pane, P. Pucci, A. Tarallo, G. Parenti, N. Brunetti-Pierri, C. Settembre, A. Ballabio, R. S. Polishchuk. Wilson disease protein ATP7B utilizes lysosomal exocytosis to maintain copper homeostasis.. Dev. Cell, 2014. [DOI | PubMed]
- S. Sluysmans, I. Mean, T. Xiao, A. Boukhatemi, F. Ferreira, L. Jond, A. Mutero, C. J. Chang, S. Citi. PLEKHA5, PLEKHA6, and PLEKHA7 bind to PDZD11 to target the Menkes ATPase ATP7A to the cell periphery and regulate copper homeostasis.. Mol. Biol. Cell, 2021. [DOI | PubMed]
- A. Boulet, K. E. Vest, M. K. Maynard, M. G. Gammon, A. C. Russell, A. T. Mathews, S. E. Cole, X. Zhu, C. B. Phillips, J. Q. Kwong. The mammalian phosphate carrier SLC25A3 is a mitochondrial copper transporter required for cytochrome c oxidase biogenesis.. J. Biol. Chem., 2018. [DOI | PubMed]
- J. Calvo, H. Jung, G. Meloni. Copper metallothioneins.. IUBMB Life, 2017. [DOI | PubMed]
- J. S. Calvo, V. M. Lopez, G. Meloni. Non-coordinative metal selectivity bias in human metallothioneins metal-thiolate clusters.. Metallomics, 2018. [DOI | PubMed]
- K. Ngamchuea, C. Batchelor-McAuley, R. G. Compton. The Copper(II)-Catalyzed Oxidation of Glutathione.. Chemistry, 2016. [DOI | PubMed]
- M. T. Morgan, L. A. H. Nguyen, H. L. Hancock, C. J. Fahrni. Glutathione limits aquacopper(I) to sub-femtomolar concentrations through cooperative assembly of a tetranuclear cluster.. J. Biol. Chem., 2017. [DOI | PubMed]
- E. Falcone, F. Stellato, B. Vileno, M. Bouraguba, V. Lebrun, M. Ilbert, S. Morante, P. Faller. Revisiting the pro-oxidant activity of copper: interplay of ascorbate, cysteine, and glutathione.. Metallomics, 2023. [DOI | PubMed]
- M. Mari, A. Morales, A. Colell, C. Garcia-Ruiz, J. C. Fernandez-Checa. Mitochondrial glutathione, a key survival antioxidant.. Antioxid. Redox Signal, 2009. [DOI | PubMed]
- S. J. Lin, V. C. Culotta. The ATX1 gene of Saccharomyces cerevisiae encodes a small metal homeostasis factor that protects cells against reactive oxygen toxicity.. Proc. Natl. Acad. Sci. U.S.A., 1995. [DOI | PubMed]
- A. K. Boal, A. C. Rosenzweig. Structural biology of copper trafficking.. Chem. Rev., 2009. [DOI | PubMed]
- A. K. Wernimont, D. L. Huffman, A. L. Lamb, T. V. O’Halloran, A. C. Rosenzweig. Structural basis for copper transfer by the metallochaperone for the Menkes/Wilson disease proteins.. Nat. Struct. Biol., 2000. [DOI | PubMed]
- I. H. Hung, R. L. Casareno, G. Labesse, F. S. Mathews, J. D. Gitlin. HAH1 is a copper-binding protein with distinct amino acid residues mediating copper homeostasis and antioxidant defense.. J. Biol. Chem., 1998. [DOI | PubMed]
- V. C. Culotta, L. W. Klomp, J. Strain, R. L. Casareno, B. Krems, J. D. Gitlin. The copper chaperone for superoxide dismutase.. J. Biol. Chem., 1997. [DOI | PubMed]
- M. W. Pantoliano, J. S. Valentine, A. R. Burger, S. J. Lippard. A pH-dependent superoxide dismutase activity for zinc-free bovine erythrocuprein. Reexamination of the role of zinc in the holoprotein.. J. Inorg. Biochem., 1982. [DOI | PubMed]
- T. Tsukihara, H. Aoyama, E. Yamashita, T. Tomizaki, H. Yamaguchi, K. Shinzawa-Itoh, R. Nakashima, R. Yaono, S. Yoshikawa. Structures of metal sites of oxidized bovine heart cytochrome c oxidase at 2.8 A.. Science, 1995. [DOI | PubMed]
- D. Horn, A. Barrientos. Mitochondrial copper metabolism and delivery to cytochrome c oxidase.. IUBMB Life, 2008. [DOI | PubMed]
- B. Kadenbach, M. Huttemann. The subunit composition and function of mammalian cytochrome c oxidase.. Mitochondrion, 2015. [DOI | PubMed]
- G. Inesi. Molecular features of copper binding proteins involved in copper homeostasis.. IUBMB Life, 2017. [DOI | PubMed]
- D. M. Glerum, A. Shtanko, A. Tzagoloff. Characterization of COX17, a yeast gene involved in copper metabolism and assembly of cytochrome oxidase.. J. Biol. Chem., 1996. [DOI | PubMed]
- C. J. Hlynialuk, B. Ling, Z. N. Baker, P. A. Cobine, L. D. Yu, A. Boulet, T. Wai, A. Hossain, A. M. El Zawily, P. J. McFie. The Mitochondrial Metallochaperone SCO1 Is Required to Sustain Expression of the High-Affinity Copper Transporter CTR1 and Preserve Copper Homeostasis.. Cell Rep., 2015. [DOI | PubMed]
- M. N. Morgada, L. A. Abriata, C. Cefaro, K. Gajda, L. Banci, A. J. Vila. Loop recognition and copper-mediated disulfide reduction underpin metal site assembly of CuA in human cytochrome oxidase.. Proc. Natl. Acad. Sci. U.S.A., 2015. [DOI | PubMed]
- B. Bjorkblom, A. Adilbayeva, J. Maple-Grodem, D. Piston, M. Okvist, X. M. Xu, C. Brede, J. P. Larsen, S. G. M?ller. Parkinson disease protein DJ-1 binds metals and protects against metal-induced cytotoxicity.. J. Biol. Chem., 2013. [DOI | PubMed]
- A. Grubman, A. R. White. Copper as a key regulator of cell signalling pathways.. Expert. Rev. Mol. Med., 2014. [DOI | PubMed]
- S. H. Chen, J. K. Lin, Y. C. Liang, M. H. Pan, S. H. Liu, S. Y. Lin-Shiau. Involvement of activating transcription factors JNK, NF-kappaB, and AP-1 in apoptosis induced by pyrrolidine dithiocarbamate/Cu complex.. Eur. J. Pharmacol., 2008. [DOI | PubMed]
- Q. Pan, L. W. Bao, S. D. Merajver. Tetrathiomolybdate inhibits angiogenesis and metastasis through suppression of the NFkappaB signaling cascade.. Mol. Cancer Res., 2003. [PubMed]
- A. Kunwar, H. Narang, K. I. Priyadarsini, M. Krishna, R. Pandey, K. B. Sainis. Delayed activation of PKCdelta and NFkappaB and higher radioprotection in splenic lymphocytes by copper (II)-Curcumin (1:1) complex as compared to curcumin.. J. Cell Biochem., 2007. [DOI | PubMed]
- S. Kadowaki, D. Endoh, T. Okui, M. Hayashi. Trientine, a copper-chelating agent, induced apoptosis in murine fibrosarcoma cells by activation of the p38 MAPK pathway.. J. Vet. Med. Sci., 2009. [DOI | PubMed]
- H. L. Chen, C. Y. Chang, H. T. Lee, H. H. Lin, P. J. Lu, C. N. Yang, C. W. Shiau, A. Y. Shaw. Synthesis and pharmacological exploitation of clioquinol-derived copper-binding apoptosis inducers triggering reactive oxygen species generation and MAPK pathway activation.. Bioorg. Med. Chem., 2009. [DOI | PubMed]
- V. N. Pham, C. J. Chang. Metalloallostery and Transition Metal Signaling: Bioinorganic Copper Chemistry Beyond Active Sites.. Angew. Chem., Int. Ed., 2023. [DOI]
- M. L. Turski, D. C. Brady, H. J. Kim, B. E. Kim, Y. Nose, C. M. Counter, D. R. Winge, D. J. Thiele. A novel role for copper in Ras/mitogen-activated protein kinase signaling.. Mol. Cell. Biol., 2012. [DOI | PubMed]
- M. Grasso, G. J. Bond, Y. J. Kim, S. Boyd, M. Matson Dzebo, S. Valenzuela, T. Tsang, N. A. Schibrowsky, K. B. Alwan, N. J. Blackburn. The copper chaperone CCS facilitates copper binding to MEK1/2 to promote kinase activation.. J. Biol. Chem., 2021. [DOI | PubMed]
- Y. J. Kim, G. J. Bond, T. Tsang, J. M. Posimo, L. Busino, D. C. Brady. Copper chaperone ATOX1 is required for MAPK signaling and growth in BRAF mutation-positive melanoma.. Metallomics, 2019. [DOI | PubMed]
- J. E. Chojnowski, R. Li, T. Tsang, F. H. Alfaran, A. Dick, S. Cocklin, D. C. Brady, T. I. Strochlic. Copper Modulates the Catalytic Activity of Protein Kinase CK2.. Front. Mol. Biosci., 2022. [DOI | PubMed]
- T. Tsang, J. M. Posimo, A. A. Gudiel, M. Cicchini, D. M. Feldser, D. C. Brady. Copper is an essential regulator of the autophagic kinases ULK1/2 to drive lung adenocarcinoma.. Nat. Cell Biol., 2020. [DOI | PubMed]
- E. A. Ostrakhovitch, M. R. Lordnejad, F. Schliess, H. Sies, L. O. Klotz. Copper ions strongly activate the phosphoinositide-3-kinase/Akt pathway independent of the generation of reactive oxygen species.. Arch. Biochem. Biophys., 2002. [DOI | PubMed]
- P. Tsvetkov, S. Coy, B. Petrova, M. Dreishpoon, A. Verma, M. Abdusamad, J. Rossen, L. Joesch-Cohen, R. Humeidi, R. D. Spangler. Copper induces cell death by targeting lipoylated TCA cycle proteins.. Science, 2022. [DOI | PubMed]
- L. Krishnamoorthy, J. A. Cotruvo, J. Chan, H. Kaluarachchi, A. Muchenditsi, V. S. Pendyala, S. Jia, A. T. Aron, C. M. Ackerman, M. N. Wal. Copper regulates cyclic-AMP-dependent lipolysis.. Nat. Chem. Biol., 2016. [DOI | PubMed]
- L. Chen, J. Min, F. Wang. Copper homeostasis and cuproptosis in health and disease.. Signal Transduct Target Ther., 2022. [DOI | PubMed]
- J. H. Menkes, M. Alter, G. K. Steigleder, D. R. Weakley, J. H. Sung. A sex-linked recessive disorder with retardation of growth, peculiar hair, and focal cerebral and cerebellar degeneration.. Pediatrics, 1962. [DOI | PubMed]
- S. A. K. Wilson. Progressive lenticular degeneration: a familial nervous disease associated with cirrhosis of the liver.. Brain, 1912. [DOI]
- A. Czlonkowska, T. Litwin, P. Dusek, P. Ferenci, S. Lutsenko, V. Medici, J. K. Rybakowski, K. H. Weiss, M. L. Schilsky. Wilson disease.. Nat. Rev. Dis. Primers, 2018. [DOI | PubMed]
- R. E. Tanzi, K. Petrukhin, I. Chernov, J. L. Pellequer, W. Wasco, B. Ross, D. M. Romano, E. Parano, L. Pavone, L. M. Brzustowicz. The Wilson disease gene is a copper transporting ATPase with homology to the Menkes disease gene.. Nat. Genet., 1993. [DOI | PubMed]
- K. Petrukhin, S. G. Fischer, M. Pirastu, R. E. Tanzi, I. Chernov, M. Devoto, L. M. Brzustowicz, E. Cayanis, E. Vitale, J. J. Russo. Mapping, cloning and genetic characterization of the region containing the Wilson disease gene.. Nat. Genet., 1993. [DOI | PubMed]
- P. C. Bull, G. R. Thomas, J. M. Rommens, J. R. Forbes, D. W. Cox. The Wilson disease gene is a putative copper transporting P-type ATPase similar to the Menkes gene.. Nat. Genet., 1993. [DOI | PubMed]
- L. M. Guthrie, S. Soma, S. Yuan, A. Silva, M. Zulkifli, T. C. Snavely, H. F. Greene, E. Nunez, B. Lynch, C. De Ville. Elesclomol alleviates Menkes pathology and mortality by escorting Cu to cuproenzymes in mice.. Science, 2020. [DOI | PubMed]
- A. Donsante, P. Sullivan, D. S. Goldstein, L. R. Brinster, S. G. Kaler. L-threo-dihydroxyphenylserine corrects neurochemical abnormalities in a Menkes disease mouse model.. Ann. Neurol., 2013. [DOI | PubMed]
- M. E. Helsel, K. J. Franz. Pharmacological activity of metal binding agents that alter copper bioavailability.. Dalton Trans., 2015. [DOI | PubMed]
- K. Lee, M. M. Briehl, A. P. Mazar, I. Batinic-Haberle, J. S. Reboucas, B. Glinsmann-Gibson, L. M. Rimsza, M. E. Tome. The copper chelator ATN-224 induces peroxynitrite-dependent cell death in hematological malignancies.. Free Radic. Biol. Med., 2013. [DOI | PubMed]
- S. Müller, A. Versini, F. Sindikubwabo, G. Belthier, S. Niyomchon, J. Pannequin, L. Grimaud, T. Cañeque, R. Rodriguez. Metformin reveals a mitochondrial copper addiction of mesenchymal cancer cells.. PLoS One, 2018. [DOI | PubMed]
- J. B. Moore, C. H. June. Cytokine release syndrome in severe COVID-19.. Science, 2020. [DOI | PubMed]
- A. B. Crouse, T. Grimes, P. Li, M. Might, F. Ovalle, A. Shalev. Metformin Use Is Associated With Reduced Mortality in a Diverse Population With COVID-19 and Diabetes.. Front. Endocrinol., 2021. [DOI]
- E. J. Ge, A. I. Bush, A. Casini, P. A. Cobine, J. R. Cross, G. M. DeNicola, Q. P. Dou, K. J. Franz, V. M. Gohil, S. Gupta. Connecting copper and cancer: from transition metal signalling to metalloplasia.. Nat. Rev. Cancer, 2022. [DOI | PubMed]
- A. Steinbrueck, A. C. Sedgwick, J. T. Brewster, K. C. Yan, Y. Shang, D. M. Knoll, G. I. Vargas-Zuniga, X. P. He, H. Tian, J. L. Sessler. Transition metal chelators, pro-chelators, and ionophores as small molecule cancer chemotherapeutic agents.. Chem. Soc. Rev., 2020. [DOI | PubMed]
- E. M. Poursani, D. Mercatelli, P. Raninga, J. L. Bell, F. Saletta, F. V. Kohane, D. P. Neumann, Y. Zheng, J. R. C. Rouaen, T. R. Jue, F. T. Michniewicz, P. Schadel, E. Kasiou, M. Tsoli, G. Cirillo, S. Waters, T. Shai-Hee, R. Cazzoli, M. Brettle, I. Slapetova, M. Kasherman, R. Whan, F. Souza-Fonseca-Guimaraes, L. Vahdat, D. Ziegler, J. G. Lock, F. M. Giorgi, K. Khanna, O. Vittorio. Copper chelation suppresses epithelial-mesenchymal transition by inhibition of canonical and non-canonical TGF-beta signaling pathways in cancer.. Cell Biosci., 2023. [DOI | PubMed]
- D. C. Brady, M. S. Crowe, M. L. Turski, G. A. Hobbs, X. Yao, A. Chaikuad, S. Knapp, K. Xiao, S. L. Campbell, D. J. Thiele. Copper is required for oncogenic BRAF signalling and tumorigenesis.. Nature, 2014. [DOI | PubMed]
- S. Sammons, D. Brady, L. Vahdat, A. K. Salama. Copper suppression as cancer therapy: the rationale for copper chelating agents in BRAF(V600) mutated melanoma.. Melanoma Manag., 2016. [DOI | PubMed]
- Y. J. Kim, T. Tsang, G. R. Anderson, J. M. Posimo, D. C. Brady. Inhibition of BCL2 Family Members Increases the Efficacy of Copper Chelation in BRAF(V600E)-Driven Melanoma.. Cancer Res., 2020. [DOI | PubMed]
- N. Chan, A. Willis, N. Kornhauser, M. M. Ward, S. B. Lee, E. Nackos, B. R. Seo, E. Chuang, T. Cigler, A. Moore. Influencing the Tumor Microenvironment: A Phase II Study of Copper Depletion Using Tetrathiomolybdate in Patients with Breast Cancer at High Risk for Recurrence and in Preclinical Models of Lung Metastases.. Clin. Cancer Res., 2017. [DOI | PubMed]
- P. Liu, S. Brown, T. Goktug, P. Channathodiyil, V. Kannappan, J. P. Hugnot, P. O. Guichet, X. Bian, A. L. Armesilla, J. L. Darling. Cytotoxic effect of disulfiram/copper on human glioblastoma cell lines and ALDH-positive cancer-stem-like cells.. Br. J. Cancer, 2012. [DOI | PubMed]
- L. Song, V. Nguyen, J. Xie, S. Jia, C. J. Chang, E. Uchio, X. Zi. ATPase Copper Transporting Beta (ATP7B) Is a Novel Target for Improving the Therapeutic Efficacy of Docetaxel by Disulfiram/Copper in Human Prostate Cancer.. Mol. Cancer Ther., 2024. [DOI | PubMed]
- H. Li, J. Wang, C. Wu, L. Wang, Z. S. Chen, W. Cui. The combination of disulfiram and copper for cancer treatment.. Drug Discovery Today, 2020. [DOI | PubMed]
- O. Scrivner, L. Dao, M K. Newell-Rogers, B. Shahandeh, F. L Meyskens, S. K. Kozawa, F. Liu-Smith, G. Plascencia-Villa, M. Jose-Yacaman, S. Jia, C. J Chang, P. J Farmer. The ionophore thiomaltol induces rapid lysosomal accumulation of copper and apoptosis in melanoma.. Metallomics, 2022. [DOI | PubMed]
- S. Blockhuys, D. C. Brady, P. Wittung-Stafshede. Evaluation of copper chaperone ATOX1 as prognostic biomarker in breast cancer.. Breast Cancer, 2020. [DOI | PubMed]
- J. Wang, C. Luo, C. Shan, Q. You, J. Lu, S. Elf, Y. Zhou, Y. Wen, J. L. Vinkenborg, J. Fan. Inhibition of human copper trafficking by a small molecule significantly attenuates cancer cell proliferation.. Nat. Chem., 2015. [DOI | PubMed]
- D. Chen, Q. C. Cui, H. Yang, R. A. Barrea, F. H. Sarkar, S. Sheng, B. Yan, G. P. Reddy, Q. P. Dou. Clioquinol, a therapeutic agent for Alzheimer’s disease, has proteasome-inhibitory, androgen receptor-suppressing, apoptosis-inducing, and antitumor activities in human prostate cancer cells and xenografts.. Cancer Res., 2007. [DOI | PubMed]
- K. G. Daniel, D. Chen, S. Orlu, Q. C. Cui, F. R. Miller, Q. P. Dou. Clioquinol and pyrrolidine dithiocarbamate complex with copper to form proteasome inhibitors and apoptosis inducers in human breast cancer cells.. Breast Cancer Res., 2005. [DOI]
- L. C. Costello, R. B. Franklin. A comprehensive review of the role of zinc in normal prostate function and metabolism; and its implications in prostate cancer.. Arch. Biochem. Biophys., 2016. [DOI | PubMed]
- M. Di Vaira, C. Bazzicalupi, P. Orioli, L. Messori, B. Bruni, P. Zatta. Clioquinol, a drug for Alzheimer’s disease specifically interfering with brain metal metabolism: structural characterization of its zinc(II) and copper(II) complexes.. Inorg. Chem., 2004. [DOI | PubMed]
- R. Somwar, H. Erdjument-Bromage, E. Larsson, D. Shum, W. W. Lockwood, G. Yang, C. Sander, O. Ouerfelli, P. J. Tempst, H. Djaballah. Superoxide dismutase 1 (SOD1) is a target for a small molecule identified in a screen for inhibitors of the growth of lung adenocarcinoma cell lines.. Proc. Natl. Acad. Sci. U.S.A., 2011. [DOI | PubMed]
- X. Dong, Z. Zhang, J. Zhao, J. Lei, Y. Chen, X. Li, H. Chen, J. Tian, D. Zhang, C. Liu. The rational design of specific SOD1 inhibitors via copper coordination and their application in ROS signaling research.. Chem. Sci., 2016. [DOI | PubMed]
- J. R. Kirshner, S. He, V. Balasubramanyam, J. Kepros, C. Y. Yang, M. Zhang, Z. Du, J. Barsoum, J. Bertin. Elesclomol induces cancer cell apoptosis through oxidative stress.. Mol. Cancer Ther., 2008. [DOI | PubMed]
- R. Franco, J. A. Cidlowski. Apoptosis and glutathione: beyond an antioxidant.. Cell Death Differ., 2009. [DOI | PubMed]
- C. Y. Chung, J. M. Posimo, S. Lee, T. Tsang, J. M. Davis, D. C. Brady, C. J. Chang. Activity-based ratiometric FRET probe reveals oncogene-driven changes in labile copper pools induced by altered glutathione metabolism.. Proc. Natl. Acad. Sci. U.S.A., 2019. [DOI | PubMed]
- J. Bonet-Aleta, M. Encinas-Gimenez, M. Oi, A. T. Pezacki, V. Sebastian, A. de Martino, A. Martin-Pardillos, P. Martin-Duque, J. L. Hueso, C. J. Chang. Nanomedicine Targeting Cuproplasia in Cancer: Labile Copper Sequestration Using Polydopamine Particles Blocks Tumor Growth In Vivo through Altering Metabolism and Redox Homeostasis.. ACS Appl. Mater. Interfaces, 2024. [DOI | PubMed]
- L. Cui, A. M. Gouw, E. L. LaGory, S. Guo, N. Attarwala, Y. Tang, J. Qi, Y. S. Chen, Z. Gao, K. M. Casey. Mitochondrial copper depletion suppresses triple-negative breast cancer in mice.. Nat. Biotechnol., 2021. [DOI | PubMed]
- R. A. Cherny, C. S. Atwood, M. E. Xilinas, D. N. Gray, W. D. Jones, C. A. McLean, K. J. Barnham, I. Volitakis, F. W. Fraser, Y. Kim. Treatment with a copper-zinc chelator markedly and rapidly inhibits beta-amyloid accumulation in Alzheimer’s disease transgenic mice.. Neuron, 2001. [DOI | PubMed]
- M. K. Lawson, M. Valko, M. T. D. Cronin, K. Jomová. Chelators in Iron and Copper Toxicity.. Curr. Pharm. Rep., 2016. [DOI]
- W. Small, A. Molteni, Y. T. Kim, J. M. Taylor, C. H. Ts’ao, W. F. Ward. Mechanism of captopril toxicity to a human mammary ductal carcinoma cell line in the presence of copper.. Breast Cancer Res. Treat., 1999. [DOI | PubMed]
- F. Xie, F. Peng. Anti-Prostate Cancer Activity of 8-Hydroxyquinoline-2-Carboxaldehyde-Thiosemicarbazide Copper Complexes by Fluorescent Microscopic Imaging.. J. Fluoresc., 2017. [DOI | PubMed]
- F. Bisceglie, M. Baldini, M. Belicchi-Ferrari, E. Buluggiu, M. Careri, G. Pelosi, S. Pinelli, P. Tarasconi. Metal complexes of retinoid derivatives with antiproliferative activity: synthesis, characterization and DNA interaction studies.. Eur. J. Med. Chem., 2007. [DOI | PubMed]
- C. Fan, H. Su, J. Zhao, B. Zhao, S. Zhang, J. Miao. A novel copper complex of salicylaldehyde pyrazole hydrazone induces apoptosis through up-regulating integrin beta4 in H322 lung carcinoma cells.. Eur. J. Med. Chem., 2010. [DOI | PubMed]
- F. Wang, P. Jiao, M. Qi, M. Frezza, Q. P. Dou, B. Yan. Turning tumor-promoting copper into an anti-cancer weapon via high-throughput chemistry.. Curr. Med. Chem., 2010. [DOI | PubMed]
- S. C. Drew. Chelator PBT2 Forms a Ternary Cu(2+) Complex with beta-Amyloid That Has High Stability but Low Specificity.. Int. J. Mol. Sci., 2023. [DOI | PubMed]
- K. L. Summers, G. Roseman, K. M. Schilling, N. V. Dolgova, M. J. Pushie, D. Sokaras, T. Kroll, H. H. Harris, G. L. Millhauser, I. J. Pickering. Alzheimer’s Drug PBT2 Interacts with the Amyloid beta 1–42 Peptide Differently than Other 8-Hydroxyquinoline Chelating Drugs.. Inorg. Chem., 2022. [DOI | PubMed]
- A. Shrivastav, N. K. Singh, P. Tripathi, T. George, J. R. Dimmock, R. K. Sharma. Copper(II) and manganese(III) complexes of N’-[(2-hydroxy phenyl) carbonothioyl] pyridine-2-carbohydrazide: novel therapeutic agents for cancer.. Biochimie, 2006. [DOI | PubMed]
- E. W. Ainscough, A. M. Brodie, W. A. Denny, G. J. Finlay, S. A. Gothe, J. D. Ranford. Cytotoxicity of salicylaldehyde benzoylhydrazone analogs and their transition metal complexes: quantitative structure-activity relationships.. J. Inorg. Biochem., 1999. [DOI | PubMed]
- V. Prachayasittikul, S. Prachayasittikul, S. Ruchirawat, V. Prachayasittikul. 8-Hydroxyquinolines: a review of their metal chelating properties and medicinal applications.. Drug Des. Devel. Ther., 2013. [DOI]
- A. Chakraborty, P. Kumar, K. Ghosh, P. Roy. Evaluation of a Schiff base copper complex compound as potent anticancer molecule with multiple targets of action.. Eur. J. Pharmacol., 2010. [DOI | PubMed]
- W. J. Lian, X. T. Wang, C. Z. Xie, H. Tian, X. Q. Song, H. T. Pan, X. Qiao, J. Y. Xu. Mixed-ligand copper(ii) Schiff base complexes: the role of the co-ligand in DNA binding, DNA cleavage, protein binding and cytotoxicity.. Dalton Trans., 2016. [DOI | PubMed]
- D. Tang, G. Kroemer, R. Kang. Targeting cuproplasia and cuproptosis in cancer.. Nat. Rev. Clin. Oncol., 2024. [DOI | PubMed]
- Z. Pan, L. Huang, Y. Gan, Y. Xia, W. Yu. The Molecular Mechanisms of Cuproptosis and Small-Molecule Drug Design in Diabetes Mellitus.. Molecules, 2024. [DOI | PubMed]
- W. Maret. The redox biology of redox-inert zinc ions.. Free Radic. Biol. Med., 2019. [DOI | PubMed]
- W. Maret, B. L. Vallee. Thiolate ligands in metallothionein confer redox activity on zinc clusters.. Proc. Natl. Acad. Sci. U.S.A., 1998. [DOI | PubMed]
- M. Murakami, T. Hirano. Intracellular zinc homeostasis and zinc signaling.. Cancer Sci., 2008. [DOI | PubMed]
- E. Bafaro, Y. Liu, Y. Xu, R. E. Dempski. The emerging role of zinc transporters in cellular homeostasis and cancer.. Signal Transduct. Target Ther., 2017. [DOI | PubMed]
- J. Jeong, D. J. Eide. The SLC39 family of zinc transporters.. Mol. Aspects Med., 2013. [DOI | PubMed]
- S. Feske, H. Wulff, E. Y. Skolnik. Ion channels in innate and adaptive immunity.. Annu. Rev. Immunol., 2015. [DOI | PubMed]
- A. Espinoza, S. Le Blanc, M. Olivares, F. Pizarro, M. Ruz, M. Arredondo. Iron, copper, and zinc transport: inhibition of divalent metal transporter 1 (DMT1) and human copper transporter 1 (hCTR1) by shRNA.. Biol. Trace Elem. Res., 2012. [DOI | PubMed]
- C. Sun, B. He, Y. Gao, X. Wang, X. Liu, L. Sun. Structural insights into the calcium-coupled zinc export of human ZnT1.. Sci. Adv., 2024. [DOI | PubMed]
- E. Mocchegiani, R. Giacconi, E. Muti, C. Cipriano, L. Costarelli, S. Tesei, N. Gasparini, M. Malavolta. Zinc-bound metallothioneins and immune plasticity: lessons from very old mice and humans.. Immun. Ageing, 2007. [DOI | PubMed]
- A. K. Baltaci, K. Yuce, R. Mogulkoc. Zinc Metabolism and Metallothioneins.. Biol. Trace Elem. Res., 2018. [DOI | PubMed]
- C. Zhang, D. Sui, T. Zhang, J. Hu. Molecular Basis of Zinc-Dependent Endocytosis of Human ZIP4 Transceptor.. Cell Rep., 2020. [DOI | PubMed]
- D. Beyersmann, H. Haase. Functions of zinc in signaling, proliferation and differentiation of mammalian cells.. Biometals, 2001. [DOI | PubMed]
- A. Weiss, C. C. Murdoch, K. A. Edmonds, M. R. Jordan, A. J. Monteith, Y. R. Perera, A. M. Rodriguez Nassif, A. M. Petoletti, W. N. Beavers, M. J. Munneke. Zn-regulated GTPase metalloprotein activator 1 modulates vertebrate zinc homeostasis.. Cell, 2022. [DOI | PubMed]
- W. Maret. Zinc in Cellular Regulation: The Nature and Significance of ″Zinc Signals″.. Int. J. Mol. Sci., 2017. [DOI | PubMed]
- W. Maret. Crosstalk of the group IIa and IIb metals calcium and zinc in cellular signaling.. Proc. Natl. Acad. Sci. U.S.A., 2001. [DOI | PubMed]
- R. S. MacDonald. The role of zinc in growth and cell proliferation.. J. Nutr., 2000. [DOI | PubMed]
- M. Cassandri, A. Smirnov, F. Novelli, C. Pitolli, M. Agostini, M. Malewicz, G. Melino, G. Raschella. Zinc-finger proteins in health and disease.. Cell Death Discovery, 2017. [DOI | PubMed]
- M. Bendellaa, P. Lelievre, J. L. Coll, L. Sancey, A. Deniaud, B. Busser. Roles of zinc in cancers: From altered metabolism to therapeutic applications.. Int. J. Cancer, 2024. [DOI | PubMed]
- S. Supasai, L. Aimo, A. M. Adamo, G. G. Mackenzie, P. I. Oteiza. Zinc deficiency affects the STAT1/3 signaling pathways in part through redox-mediated mechanisms.. Redox Biol., 2017. [DOI | PubMed]
- C. Hogstrand, P. Kille, M. L. Ackland, S. Hiscox, K. M. Taylor. A mechanism for epithelial-mesenchymal transition and anoikis resistance in breast cancer triggered by zinc channel ZIP6 and STAT3 (signal transducer and activator of transcription 3).. Biochem. J., 2013. [DOI | PubMed]
- S. Yamasaki, K. Sakata-Sogawa, A. Hasegawa, T. Suzuki, K. Kabu, E. Sato, T. Kurosaki, S. Yamashita, M. Tokunaga, K. Nishida. Zinc is a novel intracellular second messenger.. J. Cell Biol., 2007. [DOI | PubMed]
- W. Wu, I. Jaspers, W. Zhang, L. M. Graves, J. M. Samet. Role of Ras in metal-induced EGF receptor signaling and NF-kappaB activation in human airway epithelial cells.. Am. J. Physiol. Lung Cell Mol. Physiol., 2002. [DOI | PubMed]
- B. Chen, P. Yu, W. N. Chan, F. Xie, Y. Zhang, L. Liang, K. T. Leung, K. W. Lo, J. Yu, G. M. K. Tse, W. Kang, K. F. To. Cellular zinc metabolism and zinc signaling: from biological functions to diseases and therapeutic targets.. Signal Transduct. Target Ther., 2024. [DOI | PubMed]
- M. J. Liu, S. Bao, M. Galvez-Peralta, C. J. Pyle, A. C. Rudawsky, R. E. Pavlovicz, D. W. Killilea, C. Li, D. W. Nebert, M. D. Wewers. ZIP8 regulates host defense through zinc-mediated inhibition of NF-kappaB.. Cell Rep., 2013. [DOI | PubMed]
- K. G. Slepchenko, J. M. Holub, Y. V. Li. Intracellular zinc increase affects phosphorylation state and subcellular localization of protein kinase C delta (delta).. Cell Signal, 2018. [DOI | PubMed]
- E. Z. Sim, T. Enomoto, N. Shiraki, N. Furuta, S. Kashio, T. Kambe, T. Tsuyama, A. Arakawa, H. Ozawa, M. Yokoyama. Methionine metabolism regulates pluripotent stem cell pluripotency and differentiation through zinc mobilization.. Cell Rep., 2022. [DOI | PubMed]
- F. E. Duncan, E. L. Que, N. Zhang, E. C. Feinberg, T. V. O’Halloran, T. K. Woodruff. The zinc spark is an inorganic signature of human egg activation.. Sci. Rep, 2016. [DOI | PubMed]
- E. Seto, M. Yoshida. Erasers of histone acetylation: the histone deacetylase enzymes.. Cold Spring Harb. Perspect. Biol., 2014. [DOI | PubMed]
- P. Gallinari, S. D. Marco, P. Jones, M. Pallaoro, C. Steinkuhler. HDACs, histone deacetylation and gene transcription: from molecular biology to cancer therapeutics.. Cell Res., 2007. [DOI | PubMed]
- J. Jen, Y. C. Wang. Zinc finger proteins in cancer progression.. J. Biomed. Sci., 2016. [DOI | PubMed]
- J. Wang, H. Zhao, Z. Xu, X. Cheng. Zinc dysregulation in cancers and its potential as a therapeutic target.. Cancer Biol. Med., 2020. [DOI | PubMed]
- X. Zou, L. Ma, Y. Zhang, Q. Zhang, C. Xu, D. Zhang, Y. Chu, J. Zhang, M. Li, H. Zhang, J. Wang, C. Peng, G. Wei, Y. Wu, Z. Hou, H. Jia. GATA zinc finger protein p66beta promotes breast cancer cell migration by acting as a co-activator of Snail.. Cell Death Dis., 2023. [DOI | PubMed]
- J. Kim, A. Park, J. Hwang, X. Zhao, J. Kwak, H. W. Kim, M. Ku, J. Yang, T. I. Kim, K. S. Jeong. KS10076, a chelator for redox-active metal ions, induces ROS-mediated STAT3 degradation in autophagic cell death and eliminates ALDH1(+) stem cells.. Cell Rep., 2022. [DOI | PubMed]
- C. Hogstrand, P. Kille, R. I. Nicholson, K. M. Taylor. Zinc transporters and cancer: a potential role for ZIP7 as a hub for tyrosine kinase activation.. Trends Mol. Med., 2009. [DOI | PubMed]
- K. M. Taylor, S. Hiscox, R. I. Nicholson, C. Hogstrand, P. Kille. Protein kinase CK2 triggers cytosolic zinc signaling pathways by phosphorylation of zinc channel ZIP7.. Sci. Signal, 2012. [DOI | PubMed]
- E. Nolin, S. Gans, L. Llamas, S. Bandyopadhyay, S. M. Brittain, P. Bernasconi-Elias, K. P. Carter, J. J. Loureiro, J. R. Thomas, M. Schirle. Discovery of a ZIP7 inhibitor from a Notch pathway screen.. Nat. Chem. Biol., 2019. [DOI | PubMed]
- S. Ziliotto, O. Ogle, K. M. Taylor. Targeting Zin(II) Signalling to Prevent Cancer.. Met. Ions. Life Sci., 2018. [DOI]
- J. Jin, Y. Guo, X. Dong, J. Liu, Y. He. Methylation-associated silencing of miR-193b improves the radiotherapy sensitivity of esophageal cancer cells by targeting cyclin D1 in areas with zinc deficiency.. Radiother. Oncol., 2020. [DOI | PubMed]
- T. Nimmanon, S. Ziliotto, O. Ogle, A. Burt, J. M. W. Gee, G. K. Andrews, P. Kille, C. Hogstrand, W. Maret, K. M. Taylor. The ZIP6/ZIP10 heteromer is essential for the zinc-mediated trigger of mitosis.. Cell. Mol. Life Sci., 2021. [DOI | PubMed]
- R. Barroso-Sousa, S. M. Tolaney. Clinical Development of New Antibody-Drug Conjugates in Breast Cancer: To Infinity and Beyond.. BioDrugs, 2021. [DOI | PubMed]
- R. Saravanan, V. Balasubramanian, S. S. Swaroop Balamurugan, I. Ezhil, Z. Afnaan, J. John, S. Sundaram, S. Gouthaman, S. B. Pakala, S. K. Rayala. Zinc transporter LIV1: A promising cell surface target for triple negative breast cancer.. J. Cell Physiol., 2022. [DOI | PubMed]
- J. Seok, H. J. Kwak, Y. Kwak, M. Lee, K. S. Park, A. Kim, S. G. Cho. Anti-oncogenic effects of dutasteride, a dual 5-alpha reductase inhibitor and a drug for benign prostate hyperplasia, in bladder cancer.. J. Transl. Med., 2023. [DOI | PubMed]
- C. Aguirre-Portoles, R. Payne, A. Trautz, J. K. Foskett, C. A. Natale, J. T. Seykora, T. W. Ridky. ZIP9 Is a Druggable Determinant of Sex Differences in Melanoma.. Cancer Res., 2021. [DOI | PubMed]
- H. Li, X. Shen, M. Ma, W. Liu, W. Yang, P. Wang, Z. Cai, R. Mi, Y. Lu, J. Zhuang, Y. Jiang, Y. Song, Y. Wu, H. Shen. ZIP10 drives osteosarcoma proliferation and chemoresistance through ITGA10-mediated activation of the PI3K/AKT pathway.. J. Exp. Clin. Cancer Res., 2021. [DOI | PubMed]
- M. Hashemi, S. Ghavami, M. Eshraghi, E. P. Booy, M. Los. Cytotoxic effects of intra and extracellular zinc chelation on human breast cancer cells.. Eur. J. Pharmacol., 2007. [DOI | PubMed]
- M. Donadelli, E. Dalla Pozza, C. Costanzo, M. T. Scupoli, A. Scarpa, M. Palmieri. Zinc depletion efficiently inhibits pancreatic cancer cell growth by increasing the ratio of antiproliferative/proliferative genes.. J. Cell Biochem., 2008. [DOI | PubMed]
- M. Donadelli, E. Dalla Pozza, M. T. Scupoli, C. Costanzo, A. Scarpa, M. Palmieri. Intracellular zinc increase inhibits p53(−/−) pancreatic adenocarcinoma cell growth by ROS/AIF-mediated apoptosis.. Biochim. Biophys. Acta, 2009. [DOI | PubMed]
- Z. Yu, Z. Yu, Z. Chen, L. Yang, M. Ma, S. Lu, C. Wang, C. Teng, Y. Nie. Zinc chelator TPEN induces pancreatic cancer cell death through causing oxidative stress and inhibiting cell autophagy.. J. Cell Physiol., 2019. [DOI | PubMed]
- B. Regland, W. Lehmann, I. Abedini, K. Blennow, M. Jonsson, I. Karlsson, M. Sjogren, A. Wallin, M. Xilinas, C. G. Gottfries. Treatment of Alzheimer’s disease with clioquinol.. Dement. Geriatr. Cogn. Disord., 2001. [DOI | PubMed]
- C. W. Ritchie, A. I. Bush, A. Mackinnon, S. Macfarlane, M. Mastwyk, L. MacGregor, L. Kiers, R. Cherny, Q. X. Li, A. Tammer. Metal-protein attenuation with iodochlorhydroxyquin (clioquinol) targeting Abeta amyloid deposition and toxicity in Alzheimer disease: a pilot phase 2 clinical trial.. Arch. Neurol., 2003. [DOI | PubMed]
- L. C. Costello, R. B. Franklin. Cytotoxic/tumor suppressor role of zinc for the treatment of cancer: an enigma and an opportunity.. Expert Rev. Anticancer Ther., 2012. [DOI | PubMed]
- S. M. Schmitt, M. Frezza, Q. P. Dou. New applications of old metal-binding drugs in the treatment of human cancer.. Front. Biosci., 2012. [DOI]
- S. S. Brar, C. Grigg, K. S. Wilson, W. D. Holder, D. Dreau, C. Austin, M. Foster, A. J. Ghio, A. R. Whorton, G. W. Stowell. Disulfiram inhibits activating transcription factor/cyclic AMP-responsive element binding protein and human melanoma growth in a metal-dependent manner in vitro, in mice and in a patient with metastatic disease.. Mol. Cancer Ther., 2004. [DOI | PubMed]
- E. Ho. Zinc deficiency, DNA damage and cancer risk.. J. Nutr. Biochem., 2004. [DOI | PubMed]
- I. Ahel, D. Ahel, T. Matsusaka, A. J. Clark, J. Pines, S. J. Boulton, S. C. West. Poly(ADP-ribose)-binding zinc finger motifs in DNA repair/checkpoint proteins.. Nature, 2008. [DOI | PubMed]
- J. E. Bolden, M. J. Peart, R. W. Johnstone. Anticancer activities of histone deacetylase inhibitors.. Nat. Rev. Drug Discovery, 2006. [DOI | PubMed]
- E. N. Libby, P. S. Becker, N. Burwick, D. J. Green, L. Holmberg, W. I. Bensinger. Panobinostat: a review of trial results and future prospects in multiple myeloma.. Expert Rev. Hematol., 2015. [DOI | PubMed]
- A. C. West, R. W. Johnstone. New and emerging HDAC inhibitors for cancer treatment.. J. Clin. Invest., 2014. [DOI | PubMed]
- Y. Li, E. Seto. HDACs and HDAC Inhibitors in Cancer Development and Therapy.. Cold Spring Harb. Perspect Med., 2016. [DOI | PubMed]
- Z. Hu, F. Wei, Y. Su, Y. Wang, Y. Shen, Y. Fang, J. Ding, Y. Chen. Histone deacetylase inhibitors promote breast cancer metastasis by elevating NEDD9 expression.. Signal. Transduct. Target Ther., 2023. [DOI | PubMed]
- M. E. Clements, L. Holtslander, J. R. Johnson, R. W. Johnson. Select HDAC Inhibitors Enhance Osteolysis and Bone Metastasis Outgrowth but Can Be Mitigated With Bisphosphonate Therapy.. JBMR Plus, 2023. [DOI | PubMed]
- A. Chatterjee, E. J. Rodger, M. R. Eccles. Epigenetic drivers of tumourigenesis and cancer metastasis.. Semin. Cancer Biol., 2018. [DOI | PubMed]
- I. Wessels, M. Maywald, L. Rink. Zinc as a Gatekeeper of Immune Function.. Nutrients, 2017. [DOI | PubMed]
- M. Maywald, I. Wessels, L. Rink. Zinc Signals and Immunity.. Int. J. Mol. Sci., 2017. [DOI | PubMed]
- M. Dardenne. Zinc and immune function.. Eur. J. Clin. Nutr., 2002. [DOI | PubMed]
- N. Z. Gammoh, L. Rink. Zinc in Infection and Inflammation.. Nutrients, 2017. [DOI | PubMed]
- B. Kim, W. W. Lee. Regulatory Role of Zinc in Immune Cell Signaling.. Mol. Cells, 2021. [DOI | PubMed]
- M. Jarosz, M. Olbert, G. Wyszogrodzka, K. Mlyniec, T. Librowski. Antioxidant and anti-inflammatory effects of zinc. Zinc-dependent NF-kappaB signaling.. Inflammopharmacology, 2017. [DOI | PubMed]
- B. Rolles, M. Maywald, L. Rink. Intracellular zinc during cell activation and zinc deficiency.. J. Trace Elem. Med. Biol., 2021. [DOI | PubMed]
- J. M. Rice, A. Zweifach, M. A. Lynes. Metallothionein regulates intracellular zinc signaling during CD4(+) T cell activation.. BMC Immunol., 2016. [DOI | PubMed]
- H. Haase, L. Rink. The immune system and the impact of zinc during aging.. Immun. Ageing, 2009. [DOI | PubMed]
- H. Haase, L. Rink. Functional significance of zinc-related signaling pathways in immune cells.. Annu. Rev. Nutr., 2009. [DOI | PubMed]
- M. Yu, W. W. Lee, D. Tomar, S. Pryshchep, M. Czesnikiewicz-Guzik, D. L. Lamar, G. Li, K. Singh, L. Tian, C. M. Weyand. Regulation of T cell receptor signaling by activation-induced zinc influx.. J. Exp. Med., 2011. [DOI | PubMed]
- S. A. Read, K. S. O’Connor, V. Suppiah, C. L. E. Ahlenstiel, S. Obeid, K. M. Cook, A. Cunningham, M. W. Douglas, P. J. Hogg, D. Booth, J. George, G. Ahlenstiel. Zinc is a potent and specific inhibitor of IFN-lambda3 signalling.. Nat. Commun., 2017. [DOI | PubMed]
- J. D. Browning, R. S. MacDonald, W. H. Thornton, B. L. O’Dell. Reduced food intake in zinc deficient rats is normalized by megestrol acetate but not by insulin-like growth factor-I.. J. Nutr., 1998. [DOI | PubMed]
- N. Kagara, N. Tanaka, S. Noguchi, T. Hirano. Zinc and its transporter ZIP10 are involved in invasive behavior of breast cancer cells.. Cancer Sci., 2007. [DOI | PubMed]
- S. Ben Abdallah, Y. Mhalla, I. Trabelsi, A. Sekma, R. Youssef, K. Bel Haj Ali, H. Ben Soltane, H. Yacoubi, M. A. Msolli, N. Stambouli. Twice-Daily Oral Zinc in the Treatment of Patients With Coronavirus Disease 2019: A Randomized Double-Blind Controlled Trial.. Clin. Infect. Dis., 2023. [DOI | PubMed]
- J. A. M. Rodriguez, M. Bifano, E. Roca Goma, C. M. Plasencia, A. O. Torralba, M. S. Font, P. R. Millan. Effect and Tolerability of a Nutritional Supplement Based on a Synergistic Combination of beta-Glucans and Selenium- and Zinc-Enriched Saccharomyces cerevisiae (ABB C1((R))) in Volunteers Receiving the Influenza or the COVID-19 Vaccine: A Randomized, Double-Blind, Placebo-Controlled Study.. Nutrients, 2021. [DOI | PubMed]
- A. K. Jayaraman, S. Jayaraman. Increased level of exogenous zinc induces cytotoxicity and up-regulates the expression of the ZnT-1 zinc transporter gene in pancreatic cancer cells.. J. Nutr. Biochem., 2011. [DOI | PubMed]
- R. G. Uzzo, P. L. Crispen, K. Golovine, P. Makhov, E. M. Horwitz, V. M. Kolenko. Diverse effects of zinc on NF-kappaB and AP-1 transcription factors: implications for prostate cancer progression.. Carcinogenesis, 2006. [DOI | PubMed]
- L. C. Lin, J. Que, L. K. Lin, F. C. Lin. Zinc supplementation to improve mucositis and dermatitis in patients after radiotherapy for head-and-neck cancers: a double-blind, randomized study.. Int. J. Radiat. Oncol. Biol. Phys., 2006. [DOI | PubMed]
- S. W. Suh, J. W. Chen, M. Motamedi, B. Bell, K. Listiak, N. F. Pons, G. Danscher, C. J. Frederickson. Evidence that synaptically-released zinc contributes to neuronal injury after traumatic brain injury.. Brain Res., 2000. [DOI | PubMed]
- L. M. Canzoniero, P. Manzerra, C. T. Sheline, D. W. Choi. Membrane-permeant chelators can attenuate Zn2+-induced cortical neuronal death.. Neuropharmacology, 2003. [DOI | PubMed]
- P. Dobsak, C. Courderot-Masuyer, M. Zeller, C. Vergely, A. Laubriet, M. Assem, J. C. Eicher, J. R. Teyssier, J. E. Wolf, L. Rochette. Antioxidative properties of pyruvate and protection of the ischemic rat heart during cardioplegia.. J. Cardiovasc. Pharmacol., 1999. [DOI | PubMed]
- G. W. Albers. Advances in intravenous thrombolytic therapy for treatment of acute stroke.. Neurology, 2001. [DOI | PubMed]
- W. F. Lim, M. I. Mohamad Yusof, L. K. Teh, M. Z. Salleh. Significant Decreased Expressions of CaN, VEGF, SLC39A6 and SFRP1 in MDA-MB-231 Xenograft Breast Tumor Mice Treated with Moringa oleifera Leaves and Seed Residue (MOLSr) Extracts.. Nutrients, 2020. [DOI | PubMed]
- P. N. Changizzadeh, S. K. R. Mukkamalla, V. A. Armenio. Combined checkpoint inhibitor therapy causing diabetic ketoacidosis in metastatic melanoma.. J. Immunother. Cancer, 2017. [DOI | PubMed]
- M. Li, Y. Zhang, U. Bharadwaj, Q. J. Zhai, C. H. Ahern, W. E. Fisher, F. C. Brunicardi, C. D. Logsdon, C. Chen, Q. Yao. Down-regulation of ZIP4 by RNA interference inhibits pancreatic cancer growth and increases the survival of nude mice with pancreatic cancer xenografts.. Clin. Cancer Res., 2009. [DOI | PubMed]
- X. Shi, J. Yang, M. Liu, Y. Zhang, Z. Zhou, W. Luo, K. M. Fung, C. Xu, M. S. Bronze, C. W. Houchen. Circular RNA ANAPC7 Inhibits Tumor Growth and Muscle Wasting via PHLPP2-AKT-TGF-beta Signaling Axis in Pancreatic Cancer.. Gastroenterology, 2022. [DOI | PubMed]
- D. M. Wu, T. Liu, S. H. Deng, R. Han, Y. Xu. SLC39A4 expression is associated with enhanced cell migration, cisplatin resistance, and poor survival in non-small cell lung cancer.. Sci. Rep., 2017. [DOI | PubMed]
- A. G. Golub, V. G. Bdzhola, N. V. Briukhovetska, A. O. Balanda, O. P. Kukharenko, I. M. Kotey, O. V. Ostrynska, S. M. Yarmoluk. Synthesis and biological evaluation of substituted (thieno[2,3-d]pyrimidin-4-ylthio)carboxylic acids as inhibitors of human protein kinase CK2.. Eur. J. Med. Chem., 2011. [DOI | PubMed]
- M. Pandurangan, G. Enkhtaivan, D. H. Kim. Anticancer studies of synthesized ZnO nanoparticles against human cervical carcinoma cells.. J. Photochem. Photobiol., B, 2016. [DOI | PubMed]
- R. Wahab, N. Kaushik, F. Khan, N. K. Kaushik, E. H. Choi, J. Musarrat, A. A. Al-Khedhairy. Self-Styled ZnO Nanostructures Promotes the Cancer Cell Damage and Supresses the Epithelial Phenotype of Glioblastoma.. Sci. Rep., 2016. [DOI | PubMed]
- N. Wiesmann, M. Kluenker, P. Demuth, W. Brenner, W. Tremel, J. Brieger. Zinc overload mediated by zinc oxide nanoparticles as innovative anti-tumor agent.. J. Trace Elem. Med. Biol., 2019. [DOI | PubMed]
- R. Yang, R. Wu, J. Mei, F. R. Hu, C. J. Lei. Zinc oxide nanoparticles promotes liver cancer cell apoptosis through inducing autophagy and promoting p53.. Eur. Rev. Med. Pharmacol. Sci., 2021. [DOI | PubMed]
- A. Padmanabhan, M. Kaushik, R. Niranjan, J. S. Richards, B. Ebright, G. D. Venkatasubbu. Zinc Oxide nanoparticles induce oxidative and proteotoxic stress in ovarian cancer cells and trigger apoptosis Independent of p53-mutation status.. Appl. Surf. Sci., 2019. [DOI | PubMed]
- M. R. Islam, J. Attia, L. Ali, M. McEvoy, S. Selim, D. Sibbritt, A. Akhter, S. Akter, R. Peel, O. Faruque. Zinc supplementation for improving glucose handling in pre-diabetes: A double blind randomized placebo controlled pilot study.. Diabetes Res. Clin. Pract., 2016. [DOI | PubMed]
- M. Foster, P. Petocz, S. Samman. Inflammation markers predict zinc transporter gene expression in women with type 2 diabetes mellitus.. J. Nutr. Biochem., 2013. [DOI | PubMed]
- E. B. Fung, T. Ahmad, D. W. Killilea, R. Hussain, A. Lal. Zinc supplementation improves markers of glucose homeostasis in thalassaemia.. Br. J. Hamaetol., 2020. [DOI]
- R. Bobat, H. Coovadia, C. Stephen, K. L. Naidoo, N. McKerrow, R. E. Black, W. J. Moss. Safety and efficacy of zinc supplementation for children with HIV-1 infection in South Africa: a randomised double-blind placebo-controlled trial.. Lancet, 2005. [DOI | PubMed]
- E. B. Fung, J. L. Kwiatkowski, J. N. Huang, G. Gildengorin, J. C. King, E. P. Vichinsky. Zinc supplementation improves bone density in patients with thalassemia: a double-blind, randomized, placebo-controlled trial.. Am. J. Clin. Nutr., 2013. [DOI | PubMed]
- S. M. Ribeiro, C. B. Braga, F. M. Peria, F. A. Domenici, E. Z. Martinez, O. Feres, J. J. Ribeiro da Rocha, S. F. de Carvalho da Cunha. Effect of Zinc Supplementation on Antioxidant Defenses and Oxidative Stress Markers in Patients Undergoing Chemotherapy for Colorectal Cancer: a Placebo-Controlled, Prospective Randomized Trial.. Biol. Trace Elem. Res., 2016. [DOI | PubMed]
- S. K. Roy, M. J. Hossain, W. Khatun, B. Chakraborty, S. Chowdhury, A. Begum, S. Mah-e-Muneer, S. Shafique, M. Khanam, R. Chowdhury. Zinc supplementation in children with cholera in Bangladesh: randomised controlled trial.. BMJ., 2008. [DOI | PubMed]
- M. R. Nazem, M. Asadi, M. Adelipour, N. Jabbari, A. Allameh. Zinc supplementation ameliorates type 2 diabetes markers through the enhancement of total antioxidant capacity in overweight patients.. Postgrad. Med. J., 2023. [DOI | PubMed]
- B. Bao, A. S. Prasad, F. W. Beck, J. T. Fitzgerald, D. Snell, G. W. Bao, T. Singh, L. J. Cardozo. Zinc decreases C-reactive protein, lipid peroxidation, and inflammatory cytokines in elderly subjects: a potential implication of zinc as an atheroprotective agent.. Am. J. Clin. Nutr., 2010. [DOI | PubMed]
- A. H. Faghfouri, B. Baradaran, A. Khabbazi, M. Abdoli Shadbad, S. Papi, E. Faghfuri, Y. Khajebishak, S. Rahmani, P. Tolou Hayat, B. Alipour. Regulation of NLRP3 inflammasome by zinc supplementation in Behcet’s disease patients: A double-blind, randomized placebo-controlled clinical trial.. Int. Immunopharmacol., 2022. [DOI | PubMed]
- A. H. Faghfouri, A. Khabbazi, B. Baradaran, Y. Khajebishak, E. Baghbani, S. Noorolyai, S. Rahmani, S. M. Seyyed Shoura, M. Alipour, B. Alipour. Immunomodulatory and clinical responses to zinc gluconate supplementation in patients with Behcet’s disease: A double-blind, randomized placebo-controlled clinical trial.. Clin. Nutr., 2022. [DOI | PubMed]
- J. Veenemans, P. Milligan, A. M. Prentice, L. R. Schouten, N. Inja, A. C. van der Heijden, L. C. de Boer, E. J. Jansen, A. E. Koopmans, W. T. Enthoven. Effect of supplementation with zinc and other micronutrients on malaria in Tanzanian children: a randomised trial.. PLoS Med., 2011. [DOI | PubMed]
- C. H. Guo, P. C. Chen, G. S. Hsu, C. L. Wang. Zinc supplementation alters plasma aluminum and selenium status of patients undergoing dialysis: a pilot study.. Nutrients, 2013. [DOI | PubMed]
- Y. L. Qiao, S. M. Dawsey, F. Kamangar, J. H. Fan, C. C. Abnet, X. D. Sun, L. L. Johnson, M. H. Gail, Z. W. Dong, B. Yu. Total and cancer mortality after supplementation with vitamins and minerals: follow-up of the Linxian General Population Nutrition Intervention Trial.. J. Natl. Cancer Inst., 2009. [DOI | PubMed]
- D. W. Fry, A. J. Kraker, A. McMichael, L. A. Ambroso, J. M. Nelson, W. R. Leopold, R. W. Connors, A. J. Bridges. A specific inhibitor of the epidermal growth factor receptor tyrosine kinase.. Science, 1994. [DOI | PubMed]
- M. Dardenne, J. M. Pleau, B. Nabarra, P. Lefrancier, M. Derrien, J. Choay, J. F. Bach. Contribution of zinc and other metals to the biological activity of the serum thymic factor.. Proc. Natl. Acad. Sci. U.S.A., 1982. [DOI | PubMed]
- R. Codd, N. Braich, J. Liu, C. Z. Soe, A. A. Pakchung. Zn(II)-dependent histone deacetylase inhibitors: suberoylanilide hydroxamic acid and trichostatin A.. Int. J. Biochem. Cell Biol., 2009. [DOI | PubMed]
- M. Tumolo, V. Ancona, D. De Paola, D. Losacco, C. Campanale, C. Massarelli, V. F. Uricchio. Chromium Pollution in European Water, Sources, Health Risk, and Remediation Strategies: An Overview.. Int. J. Environ. Res. Public Health, 2020. [DOI | PubMed]
- D. Rehder. The role of vanadium in biology.. Metallomics, 2015. [DOI | PubMed]
- P. Aisen, R. Aasa, A. G. Redfield. The chromium, manganese, and cobalt complexes of transferrin.. J. Biol. Chem., 1969. [DOI | PubMed]
- S. Okamoto, L. D. Eltis. The biological occurrence and trafficking of cobalt.. Metallomics, 2011. [DOI | PubMed]
- R. M. Pruss, R. L. Akeson, M. M. Racke, J. L. Wilburn. Agonist-activated cobalt uptake identifies divalent cation-permeable kainate receptors on neurons and glial cells.. Neuron, 1991. [DOI | PubMed]
- B. Seetharam, N. Li. Transcobalamin II and its cell surface receptor.. Vitam. Horm., 2000. [DOI | PubMed]
- T. J. McCorvie, D. Ferreira, W. W. Yue, D. S. Froese. The complex machinery of human cobalamin metabolism.. J. Inherit. Metab. Dis., 2023. [DOI | PubMed]
- J. Morrissey, I. R. Baxter, J. Lee, L. Li, B. Lahner, N. Grotz, J. Kaplan, D. E. Salt, M. L. Guerinot. The ferroportin metal efflux proteins function in iron and cobalt homeostasis in Arabidopsis.. Plant Cell, 2009. [DOI | PubMed]
- R. Onodera, S. Asakawa, R. Segawa, N. Mizuno, K. Ogasawara, M. Hiratsuka, N. Hirasawa. Zinc ions have a potential to attenuate both Ni ion uptake and Ni ion-induced inflammation.. Sci. Rep., 2018. [DOI | PubMed]
- F. W. Sunderman. A review of the metabolism and toxicology of nickel.. Ann. Clin. Lab. Sci., 1977. [PubMed]
- F. W. Sunderman, M. I. Decsy, M. D. McNeely. Nickel metabolism in health and disease.. Ann. N.Y. Acad. Sci., 1972. [DOI | PubMed]
- B. Zambelli, S. Ciurli. Nickel and human health.. Met. Ions Life Sci., 2013. [DOI | PubMed]
- A. Munoz, M. Costa. Elucidating the mechanisms of nickel compound uptake: a review of particulate and nano-nickel endocytosis and toxicity.. Toxicol. Appl. Pharmacol., 2012. [DOI | PubMed]
- W. Hagen. Cellular uptake of molybdenum and tungsten.. Coord. Chem. Rev., 2011. [DOI]
- R. R. Mendel. Cell biology of molybdenum.. Biofactors, 2009. [DOI | PubMed]
- R. R. Mendel, T. Kruse. Cell biology of molybdenum in plants and humans.. Biochim. Biophys. Acta, 2012. [DOI | PubMed]
- G. Schwarz, R. R. Mendel, M. W. Ribbe. Molybdenum cofactors, enzymes and pathways.. Nature, 2009. [DOI | PubMed]
- B. Mukherjee, B. Patra, S. Mahapatra, P. Banerjee, A. Tiwari, M. Chatterjee. Vanadium–an element of atypical biological significance.. Toxicol. Lett., 2004. [DOI | PubMed]
- J. M. Winter, B. S. Moore. Exploring the chemistry and biology of vanadium-dependent haloperoxidases.. J. Biol. Chem., 2009. [DOI | PubMed]
- S. Lewicki, R. Zdanowski, M. Krzyzowska, A. Lewicka, B. Debski, M. Niemcewicz, M. Goniewicz. The role of Chromium III in the organism and its possible use in diabetes and obesity treatment.. Ann. Agric. Environ. Med., 2014. [DOI | PubMed]
- A. Yamamoto, O. Wada, T. Ono. Distribution and chromium-binding capacity of a low-molecular-weight, chromium-binding substance in mice.. J. Inorg. Biochem., 1984. [DOI | PubMed]
- A. Yamamoto, O. Wada, S. Manabe. Evidence that chromium is an essential factor for biological activity of low-molecular-weight, chromium-binding substance.. Biochem. Biophys. Res. Commun., 1989. [DOI | PubMed]
- J. B. Vincent. Mechanisms of chromium action: low-molecular-weight chromium-binding substance.. J. Am. Coll. Nutr., 1999. [DOI | PubMed]
- M. J. Warren, E. Raux, H. L. Schubert, J. C. Escalante-Semerena. The biosynthesis of adenosylcobalamin (vitamin B12).. Nat. Prod. Rep., 2002. [DOI | PubMed]
- M. Kobayashi, S. Shimizu. Cobalt proteins.. Eur. J. Biochem., 1999. [DOI | PubMed]
- G. H. Fong, K. Takeda. Role and regulation of prolyl hydroxylase domain proteins.. Cell Death Differ., 2008. [DOI | PubMed]
- R. Hille, T. Nishino, F. Bittner. Molybdenum enzymes in higher organisms.. Coord. Chem. Rev., 2011. [DOI | PubMed]
- I. Martinez-Reyes, N. S. Chandel. Cancer metabolism: looking forward.. Nat. Rev. Cancer, 2021. [DOI | PubMed]
- S. Trevino, A. Diaz. Vanadium and insulin: Partners in metabolic regulation.. J. Inorg. Biochem., 2020. [DOI | PubMed]
- F. Chen, X. Shi. Intracellular signal transduction of cells in response to carcinogenic metals.. Crit. Rev. Oncol. Hematol., 2002. [DOI | PubMed]
- Y. Li, J. Lou, S. Hong, D. Hou, Y. Lv, Z. Guo, K. Wang, Y. Xu, Y. Zhai, H. Liu. The role of heavy metals in the development of colorectal cancer.. BMC Cancer, 2023. [DOI | PubMed]
- D. Mu, H. Tang, G. Teng, X. Li, Y. Zhang, G. Gao, D. Wang, L. Bai, X. Lian, M. Wen. Differences of genomic alterations and heavy metals in non-small cell lung cancer with different histological subtypes.. J. Cancer Res. Clin. Oncol., 2023. [DOI | PubMed]
- V. A. Ferretti, A. E. León. An Overview of Vanadium and Cell Signaling in Potential Cancer Treatments.. Inorganics, 2022. [DOI]
- Z. Fang, M. Zhao, H. Zhen, L. Chen, P. Shi, Z. Huang. Genotoxicity of tri- and hexavalent chromium compounds in vivo and their modes of action on DNA damage in vitro.. PLoS One, 2014. [DOI | PubMed]
- J. Singh, D. L. Carlisle, D. E. Pritchard, S. R. Patierno. Chromium-induced genotoxicity and apoptosis: relationship to chromium carcinogenesis (review).. Oncol. Rep., 1998. [DOI | PubMed]
- S. Granata, F. Vivarelli, C. Morosini, D. Canistro, M. Paolini, L. C. Fairclough. Toxicological Aspects Associated with Consumption from Electronic Nicotine Delivery System (ENDS): Focus on Heavy Metals Exposure and Cancer Risk.. Int. J. Mol. Sci., 2024. [DOI | PubMed]
- M. H. Ryu, J. H. Park, J. E. Park, J. Chung, C. H. Lee, H. R. Park. Cobalt chloride stimulates phosphoinositide 3-kinase/Akt signaling through the epidermal growth factor receptor in oral squamous cell carcinoma.. Biocell, 2010. [DOI | PubMed]
- B. Zambelli, V. N. Uversky, S. Ciurli. Nickel impact on human health: An intrinsic disorder perspective.. Biochim. Biophys. Acta, 2016. [DOI | PubMed]
- H. Chen, N. C. Giri, R. Zhang, K. Yamane, Y. Zhang, M. Maroney, M. Costa. Nickel ions inhibit histone demethylase JMJD1A and DNA repair enzyme ABH2 by replacing the ferrous iron in the catalytic centers.. J. Biol. Chem., 2010. [DOI | PubMed]
- J. C. Pessoa, S. Etcheverry, D. Gambino. Vanadium compounds in medicine.. Coord. Chem. Rev., 2015. [DOI | PubMed]
- O. Tsave, S. Petanidis, E. Kioseoglou, M. P. Yavropoulou, J. G. Yovos, D. Anestakis, A. Tsepa, A. Salifoglou. Role of Vanadium in Cellular and Molecular Immunology: Association with Immune-Related Inflammation and Pharmacotoxicology Mechanisms.. Oxid. Med. Cell Longev., 2016. [DOI | PubMed]
- F. Chen, L. M. Demers, V. Vallyathan, M. Ding, Y. Lu, V. Castranova, X. Shi. Vanadate induction of NF-kappaB involves IkappaB kinase beta and SAPK/ERK kinase 1 in macrophages.. J. Biol. Chem., 1999. [DOI | PubMed]
- L. M. Pierce, F. Alessandrini, J. J. Godleski, J. D. Paulauskis. Vanadium-induced chemokine mRNA expression and pulmonary inflammation.. Toxicol. Appl. Pharmacol., 1996. [DOI | PubMed]
- G. Elberg, J. Li, Y. Shechter. Vanadium activates or inhibits receptor and non-receptor protein tyrosine kinases in cell-free experiments, depending on its oxidation state. Possible role of endogenous vanadium in controlling cellular protein tyrosine kinase activity.. J. Biol. Chem., 1994. [DOI | PubMed]
- S. Trevino, A. Diaz, E. Sanchez-Lara, B. L. Sanchez-Gaytan, J. M. Perez-Aguilar, E. Gonzalez-Vergara. Vanadium in Biological Action: Chemical, Pharmacological Aspects, and Metabolic Implications in Diabetes Mellitus.. Biol. Trace Elem. Res., 2019. [DOI | PubMed]
- J. J. O’Shea, D. W. McVicar, T. L. Bailey, C. Burns, M. J. Smyth. Activation of human peripheral blood T lymphocytes by pharmacological induction of protein-tyrosine phosphorylation.. Proc. Natl. Acad. Sci. U.S.A., 1992. [DOI | PubMed]
- R. Shrivastava, R. K. Upreti, P. K. Seth, U. C. Chaturvedi. Effects of chromium on the immune system.. FEMS Immunol. Med. Microbiol., 2002. [DOI | PubMed]
- U. Glaser, D. Hochrainer, H. Kloppel, H. Kuhnen. Low level chromium (VI) inhalation effects on alveolar macrophages and immune functions in Wistar rats.. Arch. Toxicol., 1985. [DOI | PubMed]
- H. Lawrence, D. J. Deehan, J. P. Holland, S. A. Anjum, A. E. Mawdesley, J. A. Kirby, A. J. Tyson-Capper. Cobalt ions recruit inflammatory cells in vitro through human Toll-like receptor 4.. Biochem. Biophys. Rep., 2016. [DOI | PubMed]
- B. M. Devitt, J. M. Queally, M. Vioreanu, J. S. Butler, D. Murray, P. P. Doran, J. M. O’Byrne. Cobalt ions induce chemokine secretion in a variety of systemic cell lines.. Acta Orthop., 2010. [DOI | PubMed]
- L. Diez-Tercero, L. M. Delgado, E. Bosch-Rue, R. A. Perez. Evaluation of the immunomodulatory effects of cobalt, copper and magnesium ions in a pro inflammatory environment.. Sci. Rep., 2021. [DOI | PubMed]
- A. Cavani. Breaking tolerance to nickel.. Toxicology, 2005. [DOI | PubMed]
- A. Cavani, F. Nasorri, C. Ottaviani, S. Sebastiani, O. De Pita, G. Girolomoni. Human CD25+ regulatory T cells maintain immune tolerance to nickel in healthy, nonallergic individuals.. J. Immunol., 2003. [DOI | PubMed]
- C. Rundle, S. E. Jacob. Chelation Therapy for Nickel Allergy.. J. Dermatol. Nur. Assoc., 2017. [DOI]
- E. J. Baran. Vanadium detoxification: chemical and biochemical aspects.. Chem. Biodivers., 2008. [DOI | PubMed]
- M. M. Jones, M. A. Basinger. Chelate antidotes for sodium vanadate and vanadyl sulfate intoxication in mice.. J. Toxicol. Environ. Health, 1983. [DOI | PubMed]
- J. L. Domingo, J. M. Llobet, J. Corbella. Protection of mice against the lethal effects of sodium metavanadate: a quantitative comparison of a number of chelating agents.. Toxicol. Lett., 1985. [DOI | PubMed]
- J. L. Domingo, J. M. Llobet, J. M. Tomas, J. Corbella. Influence of chelating agents on the toxicity, distribution and excretion of vanadium in mice.. J. Appl. Toxicol., 1986. [DOI | PubMed]
- P. C. Wilkins, M. D. Johnson, A. A. Holder, D. C. Crans. Reduction of vanadium(V) by L-ascorbic acid at low and neutral pH: kinetic, mechanistic, and spectroscopic characterization.. Inorg. Chem., 2006. [DOI | PubMed]
- J. L. Domingo. Vanadium: a review of the reproductive and developmental toxicity.. Reprod. Toxicol., 1996. [DOI | PubMed]
- T. Scior, A. Guevara-Garcia, P. Bernard, Q. T. Do, D. Domeyer, S. Laufer. Are vanadium compounds drugable? Structures and effects of antidiabetic vanadium compounds: a critical review.. Mini Rev. Med. Chem., 2005. [DOI | PubMed]
- A. K. Srivastava, M. Z. Mehdi. Insulino-mimetic and anti-diabetic effects of vanadium compounds.. Diabet. Med., 2005. [DOI]
- I. Goldwaser, D. Gefel, E. Gershonov, M. Fridkin, Y. Shechter. Insulin-like effects of vanadium: basic and clinical implications.. J. Inorg. Biochem., 2000. [DOI | PubMed]
- K. H. Thompson. Vanadium and diabetes.. Biofactors, 1999. [DOI | PubMed]
- J. H. McNeill, V. G. Yuen, H. R. Hoveyda, C. Orvig. Bis(maltolato)oxovanadium(IV) is a potent insulin mimic.. J. Med. Chem., 1992. [DOI | PubMed]
- C. Orvig, P. Caravan, L. Gelmini, N. Glover, F. G. Herring, H. Li, J. H. McNeill, S. J. Rettig, I. A. Setyawati. Reaction chemistry of BMOV, bis(maltolato)oxovanadium(IV), a potent insulin mimetic agent.. J. Am. Chem. Soc., 1995. [DOI]
- S. Fujimoto, K. Fujii, H. Yasui, R. Matsushita, J. Takada, H. Sakurai. Long-Term Acting and Orally Active Vanadyl-Methylpicolinate Complex with Hypoglycemic Activity in Streptozotocin-Induced Diabetic Rats.. J. Clin. Biochem. Nutr., 1997. [DOI]
- D. A. Barrio, S. B. Etcheverry. Potential use of vanadium compounds in therapeutics.. Curr. Med. Chem., 2010. [DOI | PubMed]
- J. B. Vincent. The biochemistry of chromium.. J. Nutr., 2000. [DOI | PubMed]
- B. W. Morris, S. MacNeil, C. A. Hardisty, S. Heller, C. Burgin, T. A. Gray. Chromium homeostasis in patients with type II (NIDDM) diabetes.. J. Trace Elem. Med. Biol., 1999. [DOI | PubMed]
- R. A. Anderson. Chromium and diabetes.. Nutrition, 1999. [DOI | PubMed]
- W. Maret. Chromium Supplementation in Human Health, Metabolic Syndrome, and Diabetes.. Met. Ions Life Sci., 2019. [DOI]
- F. Zhao, D. Pan, N. Wang, H. Xia, H. Zhang, S. Wang, G. Sun. Effect of Chromium Supplementation on Blood Glucose and Lipid Levels in Patients with Type 2 Diabetes Mellitus: a Systematic Review and Meta-analysis.. Biol. Trace Elem. Res., 2022. [DOI | PubMed]
- A. Schaffner, X. Li, Y. Gomez-Llorente, E. Leandrou, A. Memou, N. Clemente, C. Yao, F. Afsari, L. Zhi, N. Pan. Vitamin B(12) modulates Parkinson’s disease LRRK2 kinase activity through allosteric regulation and confers neuroprotection.. Cell Res., 2019. [DOI | PubMed]
- S. Catalani, M. C. Rizzetti, A. Padovani, P. Apostoli. Neurotoxicity of cobalt.. Hum. Exp. Toxicol., 2012. [DOI | PubMed]
- A. P. Lan, J. Chen, Z. F. Chai, Y. Hu. The neurotoxicity of iron, copper and cobalt in Parkinson’s disease through ROS-mediated mechanisms.. Biometals, 2016. [DOI | PubMed]
- G. Genchi, A. Carocci, G. Lauria, M. S. Sinicropi, A. Catalano. Nickel: Human Health and Environmental Toxicology.. Int. J. Environ. Res. Public Health, 2020. [DOI | PubMed]
- K. Mechler, W. K. Mountford, G. F. Hoffmann, M. Ries. Ultra-orphan diseases: a quantitative analysis of the natural history of molybdenum cofactor deficiency.. Genet. Med., 2015. [DOI | PubMed]
- H. J. Lee, I. M. Adham, G. Schwarz, M. Kneussel, J. O. Sass, W. Engel, J. Reiss. Molybdenum cofactor-deficient mice resemble the phenotype of human patients.. Hum. Mol. Genet., 2002. [DOI | PubMed]
- A. Veldman, J. A. Santamaria-Araujo, S. Sollazzo, J. Pitt, R. Gianello, J. Yaplito-Lee, F. Wong, C. A. Ramsden, J. Reiss, I. Cook. Successful treatment of molybdenum cofactor deficiency type A with cPMP.. Pediatrics, 2010. [DOI | PubMed]
- E. Horak, F. W. Sunderman, B. Sarkar. Comparisons of antidotal efficacy of chelating drugs upon acute toxicity of Ni(II) in rats.. Res. Commun. Chem. Pathol. Pharmacol., 1976. [PubMed]
- F. W. Sunderman, F. W. Sunderman. Nickel poisoning. VIII. Dithiocarb: a new therapeutic agent for persons exposed to nickel carbonyl.. Am. J. Med. Sci., 1958. [DOI | PubMed]
- S. L. Benoit, A. A. Schmalstig, J. Glushka, S. E. Maier, A. S. Edison, R. J. Maier. Nickel chelation therapy as an approach to combat multi-drug resistant enteric pathogens.. Sci. Rep., 2019. [DOI | PubMed]
- S. L. Benoit, R. J. Maier. The nickel-chelator dimethylglyoxime inhibits human amyloid beta peptide in vitro aggregation.. Sci. Rep., 2021. [DOI | PubMed]
- S. Salehi, S. M. M. Moghaddama, M. Tarina, A. S. Saljooghia. Pharmaceutical Nickel(II) Chelation Properties of 3-Hydroxyflaven, Deferiprone and Maltol Metal Chelators: A Density Functional Study.. Phys. Chem. Res., 2020. [DOI]
- K. S. Kasprzak, B. A. Diwan, J. M. Rice. Iron accelerates while magnesium inhibits nickel-induced carcinogenesis in the rat kidney.. Toxicology, 1994. [DOI | PubMed]
- M. Jiang, R. Wu, D. Liu, X. Wang. Utilizing Ni(II) complex for metal drug-gel particles in cervical cancer treatment and designing novel drugs through machine learning methods.. Sci. Rep., 2024. [DOI | PubMed]
- O. Andersen. Principles and recent developments in chelation treatment of metal intoxication.. Chem. Rev., 1999. [DOI | PubMed]
- O. Andersen. Chemical and biological considerations in the treatment of metal intoxications by chelating agents.. Mini Rev. Med. Chem., 2004. [DOI | PubMed]
- H. V. Aposhian, R. M. Maiorino, D. Gonzalez-Ramirez, M. Zuniga-Charles, Z. Xu, K. M. Hurlbut, P. Junco-Munoz, R. C. Dart, M. M. Aposhian. Mobilization of heavy metals by newer, therapeutically useful chelating agents.. Toxicology, 1995. [DOI | PubMed]
- J. L. Domingo, M. Gomez, J. M. Llobet, J. Corbella. Chelating agents in the treatment of acute vanadyl sulphate intoxication in mice.. Toxicology, 1990. [DOI | PubMed]
- J. Li, G. Elberg, D. C. Crans, Y. Shechter. Evidence for the distinct vanadyl(+4)-dependent activating system for manifesting insulin-like effects.. Biochemistry, 1996. [DOI | PubMed]
- I. Goldwaser, J. Li, E. Gershonov, M. Armoni, E. Karnieli, M. Fridkin, Y. Shechter. L-Glutamic acid gamma-monohydroxamate. A potentiator of vanadium-evoked glucose metabolism in vitro and in vivo.. J. Biol. Chem., 1999. [DOI | PubMed]
- C. M. Krejsa, S. G. Nadler, J. M. Esselstyn, T. J. Kavanagh, J. A. Ledbetter, G. L. Schieven. Role of oxidative stress in the action of vanadium phosphotyrosine phosphatase inhibitors. Redox independent activation of NF-kappaB.. J. Biol. Chem., 1997. [DOI | PubMed]
- L. Amaral, T. Moniz, A. M. N. Silva, M. Rangel. Vanadium Compounds with Antidiabetic Potential.. Int. J. Mol. Sci., 2023. [DOI | PubMed]
- G. Boden, X. Chen, J. Ruiz, G. D. van Rossum, S. Turco. Effects of vanadyl sulfate on carbohydrate and lipid metabolism in patients with non-insulin-dependent diabetes mellitus.. Metabolism, 1996. [DOI | PubMed]
- E. Irving, A. W. Stoker. Vanadium Compounds as PTP Inhibitors.. Molecules, 2017. [DOI | PubMed]
- M. Blanusa, V. M. Varnai, M. Piasek, K. Kostial. Chelators as antidotes of metal toxicity: therapeutic and experimental aspects.. Curr. Med. Chem., 2005. [DOI | PubMed]
- W. Banner, M. Koch, D. M. Capin, S. B. Hopf, S. Chang, T. G. Tong. Experimental chelation therapy in chromium, lead, and boron intoxication with N-acetylcysteine and other compounds.. Toxicol. Appl. Pharmacol., 1986. [DOI | PubMed]
- J. R. Behari, S. K. Tandon. Chelation in metal intoxication. VIII. Removal of chromium from organs of potassium chromate administered rats.. Clin. Toxicol., 1980. [DOI | PubMed]
- I. R. Walpole, K. Johnston, R. Clarkson, G. Wilson, G. Bower. Acute chromium poisoning in a 2 year old child.. Aust. Paediatr. J., 1985. [DOI | PubMed]
- U. Korallus, C. Harzdorf, J. Lewalter. Experimental bases for ascorbic acid therapy of poisoning by hexavalent chromium compounds.. Int. Arch. Occup. Environ. Health, 1984. [DOI | PubMed]
- S. K. Tandon, L. Srivastava. Chelation in metal intoxication XVII: Antidotal efficacy of polyaminocarboxylic acids on acute chromate toxicity.. Arch. Toxicol., 1985. [DOI | PubMed]
- E. Molina-Jijon, G. Zarco-Marquez, O. N. Medina-Campos, Z. L. Zatarain-Barron, R. Hernandez-Pando, E. Pinzon, R. M. Zavaleta, E. Tapia, J. Pedraza-Chaverri. Deferoxamine pretreatment prevents Cr(VI)-induced nephrotoxicity and oxidant stress: role of Cr(VI) chelation.. Toxicology, 2012. [DOI | PubMed]
- T. J. McLoughlin, S. K. Tsivitse, J. A. Edwards, B. A. Aiken, F. X. Pizza. Deferoxamine reduces and nitric oxide synthase inhibition increases neutrophil-mediated myotube injury.. Cell Tissue Res., 2003. [DOI | PubMed]
- M. Duran, F. A. Beemer, C. van de Heiden, J. Korteland, P. K. de Bree, M. Brink, S. K. Wadman, I. Lombeck. Combined deficiency of xanthine oxidase and sulphite oxidase: a defect of molybdenum metabolism or transport?.. J. Inherit. Metab. Dis., 1978. [DOI | PubMed]
- S. Vasto, D. Baldassano, L. Sabatino, R. Caldarella, L. Di Rosa, S. Baldassano. The Role of Consumption of Molybdenum Biofortified Crops in Bone Homeostasis and Healthy Aging.. Nutrients, 2023. [DOI | PubMed]
- S. L. Schreiber. The Rise of Molecular Glues.. Cell, 2021. [DOI | PubMed]
- R. Rodriguez, K. M. Miller. Unravelling the genomic targets of small molecules using high-throughput sequencing.. Nat. Rev. Genet., 2014. [DOI | PubMed]
- L. Kruidenier, C. W. Chung, Z. Cheng, J. Liddle, K. Che, G. Joberty, M. Bantscheff, C. Bountra, A. Bridges, H. Diallo. Kruidenier et al. reply.. Nature, 2014. [DOI | PubMed]
- M. Parrot, H. Tajmouati, V. B. R. da Silva, B. R. Atwood, R. Fourcade, Y. Gaston-Mathe, N. Do Huu, Q. Perron. Integrating synthetic accessibility with AI-based generative drug design.. J. Cheminform, 2023. [DOI | PubMed]
- Q. Perron, O. Mirguet, H. Tajmouati, A. Skiredj, A. Rojas, A. Gohier, P. Ducrot, M. P. Bourguignon, P. Sansilvestri-Morel, N. Do Huu. Deep generative models for ligand-based de novo design applied to multi-parametric optimization.. J. Comput. Chem., 2022. [DOI | PubMed]
