Effects of Dietary Pectin and Lactobacillus salivarius ATCC 11741 on Growth Performance, Immunocompetence, Gut Microbiota, Antioxidant Capacity, and Disease Resistance in Narrow-Clawed Crayfish, Postantacus leptodactylus
Abstract
The present study was conducted to clarify the effects of Lactobacillus salivarius (LS) ATCC 11741 and pectin (PE) on growth performance, digestive enzymes activity, gut microbiota composition, immune parameters, antioxidant defense as well as disease resistance against Aeromonas hydrophila in narrow-clawed crayfish, Postantacus leptodactylus. During 18 weeks trial feeding, 525 narrow-clawed crayfish juvenile (8.07 ± 0.1 g) fed with seven experimental diets including control (basal diet), LS1 (1 × 107 CFU/g), LS2 (1 × 109 CFU/g), PE1 (5 g/kg), PE2 (10 g/kg), LS1PE1 (1 × 107 CFU/g +5 g/kg), and LS2PE2 (1 × 109 CFU/g +10 g/kg). After 18 weeks, growth parameters (final weight, weight gain, and specific growth rate) and feed conversion rate were significantly improved in all treatments (P < 0.05). Besides, diets incorporated with LS1PE1 and LS2PE2 significantly increased the activity of amylase and protease enzymes compared to LS1, LS2, and control groups (P < 0.05). Microbiological analyses revealed that the total heterotrophic bacteria count (TVC) and lactic acid bacteria (LAB) of narrow-clawed crayfish fed diets containing LS1, LS2, LS1PE1, and LS2PE2 were higher than control group. The highest total haemocyte count (THC), large-granular (LGC) and semigranular cells (SGC) count, and hyaline count (HC) was obtained in LS1PE1 (P < 0.05). Similarly, higher immunity activity (lysozyme (LYZ), phenoloxidase (PO), nitroxidesynthetase (NOs), and alkaline phosphatase (AKP)) observed in the LS1PE1 treatment compared to the control group (P < 0.05). The glutathione peroxidase (GPx) and superoxide dismutase (SOD) activity remarkably enhanced in LS1PE1 and LS2PE2, while malondialdehyde (MDA) content reduced in these two treatments. In addition, specimens belonging to LS1, LS2, PE2, LS1PE1, and LS2PE2 groups presented higher resistance against A. hydrophila compared to the control group. In conclusion, feeding narrow-clawed crayfish with synbiotic had higher efficiency on growth parameters, immunocompetence, and disease resistance compared to single consumption of prebiotics and probiotics.
Affiliations: Biological Sciences Department, Faculty of Sciences, King Abdulaziz University, Jeddah, Saudi Arabia; Biology Department, Universitas Islam Negeri Alauddin, Indonesia; Assistant Professor, Rattanakosin International College of Creative Entrepreneurship, Rajamangala University of Technology Rattanakosin, Thailand; Department of Pharmacy, Al-Manara College for Medical Sciences, Maysan, Iraq; Medical Technical College, Al-Farahidi University, Iraq; Anesthesia Techniques, Al–Nisour University College, Iraq; Anesthesia Techniques Department, Al-Mustaqbal University College, Babylon, Iraq; Microbiology Research Group, College of Pharmacy, Al-Ayen University, Thi-Qar, Iraq; Department of Botany, Bahauddin Zakariya University, Multan, Pakistan; Public Health Department, Faculty of Health Science, University of Pembangunan Nasional Veteran Jakarta, Jakarta, Indonesia; Department of Fisheries, Faculty of Natural Resources, University of Tehran, Karaj, Iran; Department of Fisheries, Faculty of Natural Resources, Guilan University, Rasht, Iran
License: Copyright © 2022 Samyah Darwish Saddig Jastaniah et al. CC BY 4.0 This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1155/2022/1861761 | PubMed: 36860450 | PMC: PMC9973152
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (534 KB)
1. Introduction
Employing novel protein sources can reduce the risk of global food shortages due to war, drought, and unfair food distribution [ref. 1]. In this regard, narrow-clawed crayfish (Postantacus leptodactylus) is a high-quality aquaprotein that could help to address protein deficiency and global food insecurity [ref. 2–ref. 4]. In recent years, the reduction of the wild crayfish population, the limitation of settling in other resources as well as the high nutritional and commercial value have increased the focus on crayfish production under captivity conditions [ref. 5, ref. 6]. However, astaciculture is challenged by various problems such as a lower growth rate than tropical shrimp under cultured conditions, infectious diseases, and environmental stresses [ref. 2, ref. 7].
Studies on functional compounds like probiotics, prebiotics, and synbiotics that may enhance growth rate, immune responses, antioxidant defenses, and subsequently disease resistance in narrow-clawed crayfish have become a promising strategy in aquaculture [ref. 8].
Synbiotics are the combination of live microbial adjuncts (probiotics) and indigestible compounds (prebiotics) that beneficially affect the physiological activities of commercial species during the rearing period [ref. 9, ref. 10]. The use of synbiotics is an effective way to induce immunoregulation in aquatic animals, which in turn increases the disease resistance to infectious agents [ref. 11, ref. 12]. On the other hand, synbiotics are also known as growth promoters that can cover some production costs [ref. 13, ref. 14]. Other findings showed that diets containing synbiotics improve the resistance of fish against stressful events through the scavenging of reactive oxygen species (ROS) and altering the activity of antioxidant enzymes [ref. 15]. In astaciculture, Safari and Paolucci [ref. 8] also reported that galactooligosaccharide + Enterococcus faecalis in narrow-clawed crayfish were found to trigger growth performance, innate immune responses, and antioxidant enzyme activities.
Lactobacillus salivarius is a gram-positive bacterium that has been isolated from the breast milk and the cecum of animals [ref. 16, ref. 17]. So far, several studies have demonstrated the probiotic properties of L. salivarius including the production of natural antibiotics and short chain fatty acids, the reduction of gut pH, and the modulation of gut microbiota [ref. 16, ref. 18]. In animals, L. salivarius in the chicken diet improved growth performance, boosted immune responses, and attenuated the negative effects of stress [ref. 19]. Another study demonstrated that the colonization of L. salivarius in the gut of rats led to improved raffinose breakdown, nutrient absorption, and immune responses [ref. 20]. Despite the potential benefits of L. salivarius, there is no available information on the possible impacts of L. salivarius on aquatic animals.
Pectin is a natural prebiotic that is widely obtained from the skins and wastes of some fruits, such as apple and kiwifruit pomace, citrus, papaya, and banana peels, which is employed as a low-cost carbon source for probiotics [ref. 21, ref. 22]. Pharmacological findings have proven the therapeutic properties of pectin including lowering plasma cholesterol, against senescent, and anticancer and antidiarrhoeal properties in humans [ref. 23, ref. 24]. Furthermore, the recent findings revealed that low molecular weight and degree of methylation were useful for a wide range of probiotics [ref. 25]. Gómez et al. [ref. 26] reported that pectin in vitro increased joint populations of bifidobacteria and lactobacilli from 19% to 34% and 29%, respectively. In aquaculture, dietary pectin also has revealed beneficial effects on improved growth performance, innate immune responses, and disease resistance in different hosts such as Nile tilapia Oreochromis niloticus [ref. 22], zebrafish, Danio rerio [ref. 27] as well as antioxidant defense in common carp, Cyprinus carpio [ref. 28, ref. 29]. Besides, trials carried out by Kuo et al. [ref. 30] showed that a combination of Lactobacillus plantarum and pectin as carbon source provided a notable improvement in growth performance and immunocompetence in Litopenaeus vannamei. It seems that the combination of pectin and L. salivarius affected the physiological processes of the host more effectively than the single form of pro-/prebiotic, due to possible synergistic effects. Due to the increasing demand for narrow-clawed crayfish, the use of potential synbiotics can support P. leptodactylus production. Therefore, the present study was conducted to investigate the effects of pectin and L. salivarius in single and combined (synbiotic) forms on growth performance, immunohaemocyte, gut microbiota profile, antioxidant capacity, and disease resistance in narrow-clawed crayfish, P. leptodactylus.
2. Material and Methods
2.1. Preparation of the Experimental Diet
In this work, pectin was purchased from Sigma-Aldrich Inc. (P9135, obtained from citrus peel, galacturonic acid ≥ 74.0%). L. salivarius ATCC 11741 (IBRC-M 10865) was prepared from the Iranian Biological Resource Center (Tehran, Iran). The bacterium was cultured in Man Rogson Sharp (MRS; Merck, Germany) at 37°C for two overnight, then centrifuged at 4000 g for 20 minutes and washed twice with phosphate-buffered saline solution (PBS; pH 7.2). Afterward, it was suspended in PBS, and the desired concentrations including 1 × 107 and 1 × 109 CFU/g were adjusted. In this work, a basal diet was formulated based on Safari and Paolucci [ref. 8]. The feedstuffs used to prepare the experimental diets along with the biochemical composition of each diet are presented in Table 1. In summary, the powdered food ingredients were mixed well together (20 min), and then the homogenized mixture was pasted using water. Then, the pellets with a diameter of 2 mm were produced via an industrial meat grinder (National Meat Grinder MK-G20NR, Japan). The obtained pellets were stored at room temperature for 24 h, and stored in plastic bags in the refrigerator. Other experimental diets were made by adding dietary supplements including LS1 (L. salivarius, 1 × 107 CFU/g), LS2 (L. salivarius, 1 × 109 CFU/g), PE1 (Pectin, 5 g/kg), and PE2 (Pectin, 10 g/kg), LS1PE1 (L. salivarius; 1 × 107 CFU/g + Pectin, 5 g/kg) and LS2PE2 (L. salivarius, 1 × 109 CFU/g + Pectin, 10 g/kg), to the dough. Different levels of supplements in this study were selected based on the positive results of previous reports of pectin and other lactic acid bacteria on fish and shellfish [ref. 6, ref. 22, ref. 31, ref. 32]. Experimental diets were prepared biweekly to ensure that high L. salivarius values remained in the feeds for the duration of the trial [ref. 33]. Survival and number of colonies per each diet were confirmed using culture in tryptic soy agar (TSA; Merck, Germany) culture medium. This process was done twice a week [ref. 34] and the mean concentration of L. salivarius in supplemented diets during two weeks was based on CFU/g as follows: LS1: 0.88 × 107, LS2: 0.87 × 109, LS1PE1: 0.80 × 107, and LS2PE2: 0.75 × 109.
Table 1: Feedstuff and proximate analysis of the experimental diets.
| Ingredients | Diets (g/kg in dry basis) | ||||||
|---|---|---|---|---|---|---|---|
| Control | LS1 | LS2 | PE1 | PE2 | LS1PE1 | LS2PE2 | |
| Fishmeala | 147 | 147 | 147 | 147 | 147 | 147 | 147 |
| Wheat flourb | 289 | 289 | 289 | 289 | 289 | 289 | 289 |
| Soybean mealb | 175 | 175 | 175 | 175 | 175 | 175 | 175 |
| Corn glutenb | 112 | 112 | 112 | 112 | 112 | 112 | 112 |
| Starch mealb | 49 | 49 | 49 | 49 | 49 | 49 | 49 |
| Soybean oilb | 41 | 41 | 41 | 41 | 41 | 41 | 41 |
| Lectinb | 50 | 50 | 50 | 50 | 50 | 50 | 50 |
| Fish oilb | 42 | 42 | 42 | 42 | 42 | 42 | 42 |
| Cholesterolc | 5 | 5 | 5 | 5 | 5 | 5 | 5 |
| Glucosamined | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| Choline chloridec | 15 | 15 | 15 | 15 | 15 | 15 | 15 |
| Carboxymethyl cellulosed | 19.9 | 19.9 | 19.9 | 14.9 | 9.9 | 14.9 | 9.9 |
| Ytterbium oxided | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Vitamin Cc | 10 | 10 | 10 | 10 | 10 | 10 | 10 |
| Vitamin premixe | 20 | 20 | 20 | 20 | 20 | 20 | 20 |
| Mineral premixe | 15 | 15 | 15 | 15 | 15 | 15 | 15 |
| L. salivarius (CFU/g) | 1 × 107 | 1 × 109 | 1 × 107 | 1 × 109 | |||
| Pectin (g) | 0 | 0 | 0 | 5 | 10 | 5 | 10 |
| Chemical analysis of the experimental diets (g/kg dry matter basis) | |||||||
| (i) Dry matter | 872 | 872.02 | 872.03 | 877.20 | 883.3 | 877.22 | 883.33 |
| (ii) Crude protein | 382.3 | 382.3 | 382.3 | 382.65 | 382.95 | 382.65 | 382.95 |
| (iii) Crude lipid | 127 | 127 | 127 | 1271.15 | 127.32 | 127.15 | 127.32 |
| (iv) Ash | 37.9 | 37.92 | 37.93 | 36.60 | 36.90 | 36.62 | 36.93 |
| (v) Fiber | 30.4 | 30.40 | 30.4 | 32.7 | 35.4 | 32.70 | 35.5 |
| (vi) NFEf | 422.4 | 422.38 | 422.37 | 420.9 | 417.43 | 420.88 | 417.3 |
aPeygir Co., Gorgan, Iran. bBehparvar Aquafeed Co., Iran. cKimia Roshd Co., Iran. dSigma, Germany. eThe premix provided the following amounts per kg of feed: A:1000 IU; D3: 5000 IU; E: 20 mg; B5: 100 mg; B2: 20 mg; B6: 20 mg; B1: 20 mg; H: 1 mg; B9: 6 mg; B12: 1 mg; B4: 600 mg; C:50 mg; Mg: 350 mg; Fe: 13 mg; Co: 2.5 mg; Cu: 3 mg; Zn: 60 mg; Se: 0.3 mg; I: 1.5 mg; and Mn: 10 mg. Chinechin Co., Tehran, Iran cKimia Roshd Co., Iran. fNitrogen-free extracts (NFE) = 1000–(crude protein + crude lipid + ash + fiber).
2.2. Experimental Procedure
In this work, five hundred and fifty narrow-clawed crayfish juveniles with an average weight of (7 ± 0.20 g, mean ± SD) were collected from Aras Dam Lake (West Azerbaijan, Iran.) and transferred to a private farm in Rasht City (Iran). Adaptation was performed for 14 days in fiberglass tanks (1000 L) at DO: 6.5 ± 0.20 mg/L, pH: 7.28 ± 0.34, hardness: 143 ± 5.5 mg/L CaCO3, unionized ammonia (<0.05 mg/L), temperature: 23 ± 1°C, and specimens were fed with the basal diet, four times a day (7:00, 11:00, 16:00, and 21:00). This work was carried out in 7 experimental groups with 3 replications. For this, five hundred and twenty five narrow-clawed crayfish (8.07 ± 0.1 g, mean ± SD) were divided into 21 cubic polyethylene tanks (60 × 44 × 160 cm) at a density of 25 specimens per each tank. Animals belonging to the control group fed with the basal diet and other experimental groups received diets supplemented with probiotics or pectin including LS1, LS2, PE1, PE2, LS1PE1, and LS2PE2 four times a day based on apparent satiety for 18 weeks (July 2018 to December 2018). To calculate the eaten food, four hours after each meal, the unconsumed food was collected from each tank and dried in oven at 60. To prevent aggressive behaviors, plastic tubes (diameter: 4 cm and length: 11 cm) were placed in each tank. The rearing system was without water flow (static) and water quality was maintained by continuous aeration, removing uneaten food and particulate matter, and changing 30% of the water, daily. Also, physicochemical parameters were checked once a week.
2.3. Zootechnical Parameters and Feed Utilization
At the end of the feeding trial, the total biomass of each tank was weighed. During the rearing period, uneaten food was collected from each tank and dried. Finally, growth and nutritional parameters and survival rate were estimated based on the following formulas [ref. 35]:
Nf is the narrow-clawed crayfish number at final of trial feeding and Ni is the narrow-clawed crayfish number at initial of trial feeding.
2.4. Quantification of Digestive Enzymes
At the end of the 18th week, all animals in each tank were fasted for 24 hours and three crayfish were harvested from each replicate and dissected on ice plates. In the next step, the hepatopancreatic tissue was isolated, washed with distilled water, and gently dried using a towel. Afterward, samples were mixed with Tris-HCl buffer (W/V, 50 mM, pH: 7.0) and homogenized via an electric homogenizer (D 500). The homogenized mixture was centrifuged at 10000 g at 4°C for 25 min (Hermle Z36HK, Germany), and the supernatant was aliquoted and stored at −70°C [ref. 36]. Alpha-amylase activity was measured using starch as a substrate (0.3%) diluted in Na2HPO4 buffer (pH: 7.4). In this method, the reaction was stopped via dinitrosalicylic acid reagent, and the absorbance was recorded at 540 nm [ref. 37]. The level of alkaline protease activity was determined using 2% azo-casein solution in 50 mM Tris-HCl (pH: 9.0) as substrate. After incubation of the mixture at 25°C for 10 min, the reaction was stopped by 0.5 mL TCA (Trichloroacetic Acid). The samples were centrifuged at 6500 g for 5 min and the absorbance was recorded at 440 nm [ref. 38]. Lipase activity was measured based on the method described by Iijima et al. [ref. 39] and using p-nitrophenol myristate (Sigma N2502) as the substrate dissolved in 0.25 mM Tris-HCl (pH: 9.0) along with 0.25 mM 2-methoxyethanol and 5 mM sodium cholate solution. The reaction was terminated by the addition 0.7 mL of acetone/heptane (5 : 2 v/v), and the mixture was centrifuged (6000 g at 4°C for 5 min) and the absorbance of the supernatant was recorded at 405 nm.
2.5. Bacteriological Assay
To evaluate the gut microbiota, the skin surface of the individual was washed using distilled water and then disinfected with ethanol 70%. Thereafter, the animals were dissected aseptically using a sterile instrument, the gut was isolated, washed with PBS, and homogenized using an electric homogenizer (DI 18 Disperser). The obtained homogeneity was continuously diluted in PBS (pH: 7.2). In the next step, 100 μL of the sample was spread onto MRS and plate count agar (PCA; Merck Co) for the determination of lactic acid bacteria (LAB) and total viable heterotrophic bacteria count (TVC), respectively. The pellets were saved at 28°C for 48 h and TVC and LAB colonies were counted and reported in tissue CFU/g [ref. 40].
2.6. Hemolymph Collection
At the end of the 18th week, three starved narrow-clawed crayfish were caught from each tank, and hemolymph was drawn from the ventral sinus using a syringe with a needle 25 g. The obtained hemolymph was stored for two targets. One part was placed in a 2 mL Eppendorf containing Alsever buffer as anticoagulant agent (115 mmoL glucose, 336 mmoL NaCl, 27 mM sodium citrate, and 9 mM EDTA with pH: 7.0). 200 μL of a hemolymph-anticoagulant sample was used to estimate total haemocyte count (THC) and differential counts of haemocytes (DHC). The remaining part was immediately centrifuged at 1000 g for 5 min at 4°C and the supernatant was stored to assess immunity indicators [ref. 8].
2.6.1. Hemolymph Indices
THC (cells/mL) was counted via a hemocytometer (Neubauer, Germany) under a light microscope [ref. 41]. Assay DHC (cells/mL) was carried out by preparing the hemolymph smear, complete drying of smears exposed to airflow, fixing in 70% methanol for 10 min, and staining based on May-Grunwald and Giemsa technique [ref. 42]. Finally, the number of large granular, semigranular cell granular, and the hyaline cell was counted using a light microscope (Nikon E100; Nikon, Tokyo, Japan).
2.6.2. Hemolymph Immune Parameters
Plasma lysozyme (LYZ) activity was measured based on plasma capability in lysis of Micrococcus luteus using turbidity test [ref. 43], and recording OD at 450 nm. Phenoloxidase (PO) activity was determined spectrophotometrically (Pharmacia Biotech Ultrospec 2000) via the production of dopachrome from L-dihydroxy phenylalanine (LDOPA, Sigma), and reading OD at 490 nm [ref. 44]. Nitric oxide synthase (NOS) activity rate was calculated by the commercially available kit (Nanjing Jiancheng Bioengineering Institute, China) [ref. 45]. The activities of alkaline phosphatase (AKP) and acid phosphatase (ACP) were detected using the method described by Hao et al. [ref. 46]. In this method, disodium phenyl phosphate-4-aminoantipyrine-potassium ferricyanide and disodium phenyl phosphate were used as the substrate to estimate the activity of AKP and ACP, respectively.
2.6.3. Hemolymph Antioxidant Enzymes Activities and Malondialdehyde Content
Superoxide dismutase activity was estimated by a commercial kit (ZellBio GmbH, Germany) based on the protocol supplied by the manufacturer. In this method, 10 μL hemolymph supernatant was transferred to each well of the microplate (96-well). Afterward, 250 μL reagent 1, 10 μL reagent 2, and 10 μL distilled water were poured into each chamber. In the next step, the chromogenic matter was added to the related chambers. Finally, the color change was recorded with a microplate reader at 420 nm [ref. 6]. Catalase activity was detected using a commercial kit (ZellBio GmbH, Germany), which was used previously for crayfish [ref. 47]. The OD was estimated colorimetrically via the microplate reader at a wavelength of 405 nm. The glutathione peroxidase (GPx) was detected by quantifying the level of H2O2 in the presence of glutathione (GSH), according to the protocols recommended by the manufacturer (ZellBio GmbH, Germany). Malondialdehyde (MDA) content was estimated by reaction of thiobarbituric with malondialdehyde and maximum OD was recorded at 534 nm [ref. 47].
2.7. In Vivo Crayfish Infection Test
At the end of the feeding trial, the challenge was performed based on complete hygiene and quarantine principles. Aeromonas hydrophila (AH04) was purchased from the Faculty of Veterinary Medicine, University of Tehran. Stocks were grown on TSA medium at 37°C for 24 h. The grown bacteria were centrifuged at 10,000 × g and 4°C for 10 min. Pellets were washed twice using PBS. The desired dose was adjusted at 1 × 108 cells/mL using sequential dilution and based on previous results [ref. 8]. Crayfish were infected by injecting 20 μL into the ventral sinus [ref. 48, ref. 49]. The injected specimens (10 samples per aquarium) were transferred to a glass aquaria (V: 100 L water). The challenge period lasted for 5 days and infected animals were checked for mortality rate (MR).
2.8. Statistical Analysis
In this study, data analysis was carried out using SPSS software (version 22, (SPSS Inc., Chicago, USA). In the first step, homogeneity of variance and normality of the data were confirmed using Leaven’s and Kolmogorov-Smirnov tests, respectively. In the next step, differences between the results of experimental groups were evaluated via one-way ANOVA analysis. Besides, significant differences between the treatments were determined using Tukey’s test. The results were presented as mean ± SD.
3. Results
3.1. Growth Performance
The growth performance of P. leptodactylus fed with levels of LS and PE on the 18th week is indicated in Table 2. On the 18th week, the treated diets included LS, PE, and LSPE showed a significant increase in growth indices including FW, WG, and SGR compared to the control group (P < 0.05). Besides, the highest FW, WG, and SGR were obtained in animals fed with the diet containing LS1PE1, which showed a significant difference with those receiving diets LS1, LS2, and basal diet (P < 0.05). In contrast, an inverse pattern was revealed in FCR among different experimental groups. In this regard, the highest FCR was recorded in crayfish fed the control diet, which presented a significant difference compared to the treated groups (P < 0.05). Among the experimental groups, the lowest FCR was recorded in fish receiving the LS1PE1 diet. SR was also statistically similar among the experimental groups (P > 0.05).
Table 2: Growth performance indices of narrow-clawed crayfish (Postantacus leptodactylus) fed with basal diet and supplemented with LS1 (L. salivarius, 1 × 107 CFU/g), LS2 (L. salivarius, 1 × 109 CFU/g), PE1 (Pectin, 5 g/kg), and PE2 (Pectin, 10 g/kg), LS1PE1 (L. salivarius, 1 × 107 CFU/g + Pectin, 5 g/kg) and LS2PE2 (L. salivarius, 1 × 109 CFU/g + Pectin, 10 g/kg) for 18 weeks.
| Treatments | |||||||
|---|---|---|---|---|---|---|---|
| Parameters | Control | LS1 | LS2 | PE1 | PE2 | LS1PE1 | LS2PE2 |
| IW(g) | 8.11 ± 0.07a | 8.13 ± 0.12a | 8.15 ± 0.8a | 7.96 ± 0.07a | 8.10 ± 0.10a | 8.05 ± 0.09a | 8.01 ± 0.13a |
| FW (g) | 26.00 ± 1.50d | 31.83 ± 1.75c | 32.93 ± 2.00bc | 36.83 ± 2.56abc | 36.36 ± 2.12abc | 40.93 ± 1.90a | 38.16 ± 2.25ab |
| WG (g) | 17.88 ± 1.57d | 23.70 ± 1.81c | 24.79 ± 2.07bc | 28.86 ± 2.49abc | 28.26 ± 2.14abc | 32.89 ± 1.91a | 30.15 ± 2.26ab |
| SGR (%/d) | 0.92 ± 0.05d | 1.08 ± 0.06c | 1.10 ± 0.07bc | 1.21 ± 0.04abc | 1.19 ± 0.05abc | 1.29 ± 0.03a | 1.23 ± 0.04ab |
| FCR | 3.90 ± 0.32a | 2.98 ± 0.22b | 2.78 ± 0.19bc | 2.40 ± 0.21bcd | 2.46 ± 0.19bcd | 2.11 ± 0.15d | 2.30 ± 0.20cd |
| SR (%) | 77.33 ± 2.30a | 80 ± 4.00a | 78.66 ± 2.30a | 82 ± 2.30a | 80.25 ± 6.92a | 85.33 ± 2.30a | 81.33 ± 2.40a |
Note: Different letters in each column show significant differences among the experimental groups (P < 0.05). Values are shown as mean ± SD (n = 3). Abbreviations: IW: initial weight (g); FW: final weight (g); WG: weight gain (g); SGR: specific growth rate (%/d); FCR: feed conversation rate; SR: survival rate (%).
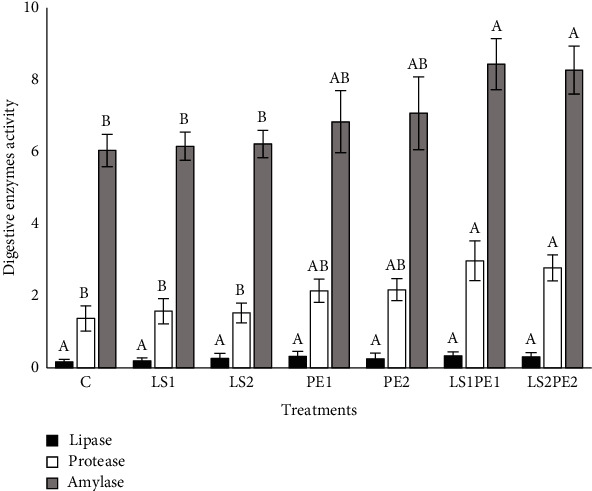
3.2. Digest Enzymes Activities
The digestive enzyme activities of the narrow-clawed crayfish after 18 weeks of feeding are presented in Figure 1. Feeding animals with diets incorporated with LS1PE1 and LS2PE2 significantly improved the activity of amylase and protease enzymes compared to ones fed with LS1, LS2, and basal diets (P < 0.05). However, lipase activity was not affected by supplemented diets (P > 0.05).
3.3. Microbiological Analysis
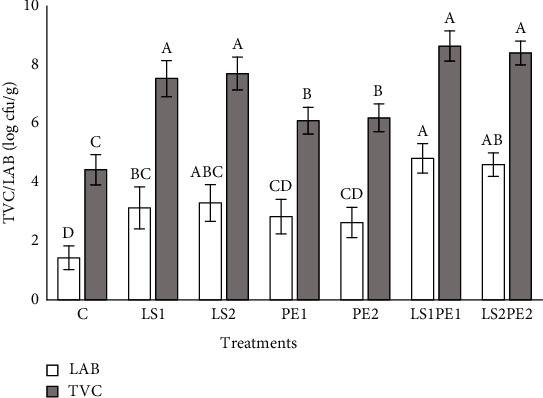
The microbiota composition of narrow-clawed crayfish after the 18 weeks feeding trial is shown in Figure 2. The highest TVC was obtained in the gut of animals fed with diets containing LS1, LS2, LS1PE1, and LS2PE2 in comparison with crayfish belonging to PE1, PE2, and control groups (P < 0.05). Moreover, crayfish fed with feeds containing LS1PE1, and LS2PE2 showed higher LAB counts compared to animals that received PE1, PE2, and basal diets (P < 0.05).
3.4. Hemolymph Indices
As indicated in Table 3, hemolymph indices in groups fed with different levels of probiotic, prebiotic, and synbiotic were significantly affected (P < 0.05). The THC count in response to the diet containing LS1PE1 indicated a significant difference compared to other groups, except for LS2PE2 (P < 0.05). HC count in all groups fed with probiotic, prebiotic, and synbiotic was significantly higher than narrow-clawed crayfish which belongs to the control group (P < 0.05). Also, feeding crayfish with LS1PE1 and LS2PE2 diets remarkably increased LGC compared to PE1, PE2, and control groups (P < 0.05). Administration of LS1, LS1PE1, and LS1PE1 resulted in a remarkable enhancement in SGC count in compared to PE1 and PE2, and control groups (P < 0.05).
Table 3: Hemolymph indices of narrow-clawed crayfish (Postantacus leptodactylus) fed with basal diet and supplemented with LS1 (L. salivarius, 1 × 107 CFU/g), LS2 (L. salivarius, 1 × 109 CFU/g), PE1 (Pectin, 5 g/kg), and PE2 (Pectin, 10 g/kg), LS1PE1 (L. salivarius, 1 × 107 CFU/g + Pectin, 5 g/kg) and LS2PE2 (L. salivarius, 1 × 109 CFU/g + Pectin, 10 g/kg) for 18 weeks.
| Treatments | |||||||
|---|---|---|---|---|---|---|---|
| Parameters | Control | LS1 | LS2 | PE1 | PE2 | LS1PE1 | LS2PE2 |
| THC (×105 cell mL-1) | 125.50 ± 3.50d | 149.16 ± 1.90bc | 148.60 ± 5.28bc | 143.36 ± 2.51c | 142.10 ± 2.74c | 159.96 ± 2.93a | 155.86 ± 3.09ab |
| HC (×105 cell mL-1) | 70.83 ± 3.32b | 81.33 ± 1.52a | 82.83 ± 3.40a | 83.50 ± 2.78a | 84.40 ± 1.90a | 86.96 ± 2.87a | 85.40 ± 2.42a |
| SGC (×105 cell mL-1) | 26.16 ± 1.25c | 32.40 ± 1.11a | 31.43 ± 0.86ab | 28.36 ± 1.28bc | 27.20 ± 1.11c | 34.50 ± 1.80a | 33.30 ± 2.02a |
| LGC (×105 cell mL-1) | 28.50 ± 1.50e | 35.43 ± 0.62abc | 34.33 ± 1.25bcd | 31.50 ± 1.51cde | 30.50 ± 1.32de | 38.50 ± 1.80a | 37.16 ± 1.75ab |
THC: total haemocyte count; LGC: large-granular count; SGC: semigranular count; HC: hyaline count. Values are presented as mean ± SD. Different superscripts within a row indicate significant differences at P < 0.05 (n = 3).
3.5. Immunological Responses
The effect of the lactobacillus, pectin, and synbiotic on immunity responses of narrow-clawed crayfish at the end of 18th week is exhibited in Table 4. Administration LS1PE1 significantly increased LYZ and PO activities compared to the control group (P < 0.05). Additionally, a peak NOS activity was recorded in crayfish fed with diets supplemented with PE1, PE2, LS1PE1, and LS2PE2 in comparison with the control group (P < 0.05). AKP activity in all treatments was remarkably elevated compared to control groups (P < 0.05). However, there was no significant difference in ACP between different treatments and the control group (P > 0.05).
Table 4: Nonspecific immunity responses of narrow-clawed crayfish (Postantacus leptodactylus) fed with basal diet and supplemented with LS1 (L. salivarius, 1 × 107 CFU/g), LS2 (L. salivarius, 1 × 109 CFU/g), PE1 (Pectin, 5 g/kg), and PE2 (Pectin, 10 g/kg), LS1PE1 (L. salivarius, 1 × 107 CFU/g + pectin, 5 g/kg) and LS2PE2 (L. salivarius, 1 × 109 CFU/g + pectin, 10 g/kg) for 18 weeks.
| Parameter | Treatments | ||||||
|---|---|---|---|---|---|---|---|
| Control | LS1 | LS2 | PE1 | PE2 | LS1PE1 | LS2PE2 | |
| LYZ (U mL−1) | 12.51 ± 1.30b | 15.14 ± 1.50ab | 15.06 ± 1.43ab | 14.75 ± 1.31ab | 14.45 ± 1.42ab | 17.26 ± 2.30a | 16.80 ± 1.70ab |
| PO (U mL−1) | 1.07 ± 0.10b | 1.35 ± 0.15ab | 1.29 ± 0.16ab | 1.27 ± 0.12ab | 1.18 ± 0.13ab | 1.58 ± 0.28a | 1.41 ± 0.17ab |
| NOS (U mL−1) | 10.06 ± 2.00c | 14.16 ± 1.25bc | 13.86 ± 1.10bc | 18.60 ± 1.42a | 17.96 ± 1.30ab | 16.93 ± 1.90ab | 16.43 ± 1.60ab |
| AKP (U/L) | 15.03 ± 1.55c | 16.93 ± 1.90ab | 17.30 ± 1.70ab | 19.26 ± 1.02ab | 18.93 ± 2.18ab | 21.93 ± 1.90a | 21.76 ± 2.25a |
| ACP (U/L) | 9.16 ± 1.25a | 9.06 ± 0.90a | 9.56 ± 1.15a | 10.10 ± 1.35a | 10.63 ± 1.82a | 11.33 ± 1.25a | 11.76 ± 1.12a |
LYZ: lysozyme; PO: phenol-oxidase; NOS: nitric oxide synthase; AKP: alkaline phosphatase; ACP: acid phosphatase. Values are presented as mean ± SD. Different superscripts within a row indicate significant differences at P < 0.05 (n = 3).
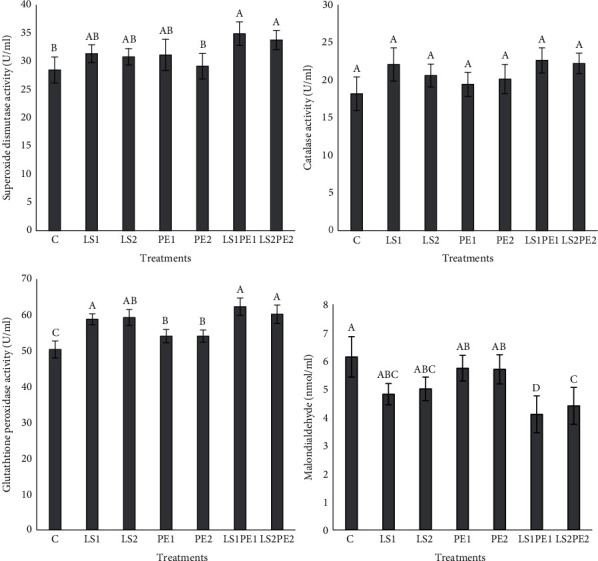
3.6. Antioxidant Defense
Alterations in the level of crayfish antioxidant enzymes in response to different levels of probiotics, pectin, and synbiotic are presented in Figure 3. SOD activity of narrow-clawed crayfish in LS1PE1 and LS2PE2 treatments was higher than PE2 and control groups (P < 0.05). GPx activity was remarkably increased in all treatments compared to the control group (P < 0.05). However, the level of CAT activity in experimental treatments was statistically similar (P > 0.05). The lowest MDA value was recorded in both LS1PE1 and LS2PE2 treatments.
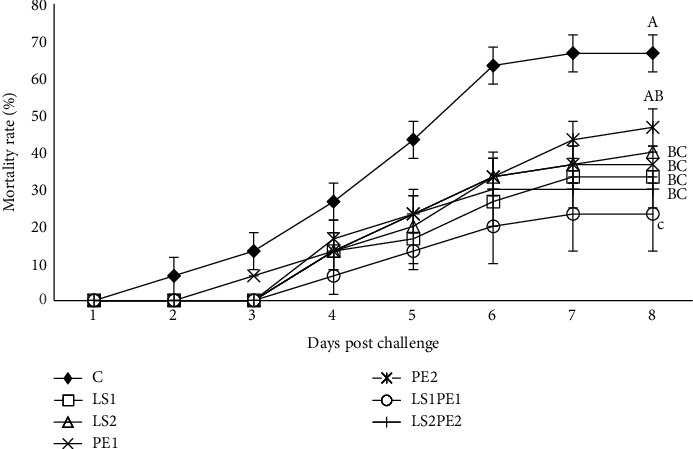
3.7. Disease Resistance
The mortality rates of crayfish infected with Aeromonas hydrophila are shown in Figure 4. Accordingly, the percentage of mortality rate (MR) in crayfish belonging to the control group was higher than those of the other groups on different days. At the end of the challenge period, the highest and lowest MR were recorded in the control (66.66%) and LS1PE1 (23.33%) groups, respectively. Experimental diets containing LS1, LS2, PE2, LS1PE1, and LS2PE2 significantly increased disease resistance compared to the control group.
4. Discussion
In modern aquaculture, there is great attention on developing feed additives to boost immune response and fish health, which in turn leads to improved disease resistance [ref. 10, ref. 50]. Dietary interventions with functional probiotics/prebiotics could exert beneficial effects on the host through influencing nutrition, gut bacterial ecosystem, metabolism, and immune function [ref. 50–ref. 52]. In the current study, growth performance and FCR in animals fed with LS or PE were beneficially altered at the end of the feeding trial. However, the highest growth indices and the lowest FCR were obtained in specimens treated by LS1PE1. Therefore, synbiotics had a greater impact than probiotics or prebiotics on growth performance and nutritional efficiency. In agreement with our results, the simultaneous employ of lactobacillus and prebiotics such as pectin + L. plantarum [ref. 30], oyster mushroom + L. plantarum [ref. 53], galactooligosaccharide + L. plantarum [ref. 54] in L. vannamei elevated growth indices and feed utilization in L. vannamei. These protective effects on host growth can be linked with releasing endogenous enzyme, producing metabolites from the fermentation process (vitamins, SCFs), and improving gut epithelial cells, which in turn facilitate the digestibility and absorption of nutrients [ref. 30, ref. 40]. Other studies proposed that the beneficial effects of synbiotics are related to the ability of prebiotics in enhancing the colonization of favorable bacteria [ref. 55, ref. 56]. During the recent decade, several researchers have confirmed the role of synbiotics on microbiota in crustaceans. For example, Safari et al. [ref. 7] and Safari and Paolucci [ref. 8] indicated the modulating effects of Enterococcus faecalis + xylooligosaccharide, Pediococcus acidilactici + mannanoligosaccharide, and E. faecalis + galactooligosaccharide on the gut microbiota of crayfish. These results are consistent with the findings of our study that in the presence of pectin, the rate of LAB colonization increased in the gut. The study implied that LS could use from pectin as a carbon source during the fermentation process, a hypothesis previously confirmed by Kuo et al. [ref. 30] in vitro and in vivo studies. On the other hand, the successful colonization of LAB can be an inducing factor for the secretion of intracellular and extracellular digestive enzymes [ref. 57]. In the current study, a notable improvement in amylase and protease enzymes activities was recorded in narrow-clawed crayfish fed LS1PE1 and LS2PE2 diets. Several works also have confirmed elevated digestive enzymes activities in crustaceans such as L. vannamei [ref. 58, ref. 59] and narrow-clawed crayfish [ref. 8] after feeding by synbiotic. However, more works are needed to elucidate the mechanism of PE + LS on the level of digestive enzymes synthesized by LAB or the host.
Despite the lack of a specific immune system in crustaceans, these animals benefit from two efficient cell-mediated mechanisms to trigger a nonspecific immune system and combat with invader agents. (1) A set of cellular responses are executed directly by immune cells, such as phagocytosis, nodule formation, and encapsulation of pathogens. (2) Humoral molecules secreted by the immune cells including antimicrobial peptides, proteins involved in hemolymph coagulation, and the prophenoloxidase system (proPO) [ref. 60]. These two mechanisms are associated with three types of haemocytes, including HCs, GCs, and SGCs [ref. 61]. It is generally accepted that the number of circulating haemocytes is a valid marker for assessing the health status of crustaceans [ref. 62]. In the current study, the dietary supplementation of LS or PE significantly increased THC and HC. Besides, LGC and SGC count are notable elevated in LS1, LS2 and LS1PE1, and LS2PE2. Similarly, the hemolymph indices were increased in A. leptodactylus fed with the Lactobacillus plantarum diet at 107 CFU/g [ref. 6]. Another study on A. leptodactylus also indicated that feed incorporated with synbiotic (Enterococcus faecalis 7.86 log CFU/g +10 g/kg xylooligosaccharide) remarkably increased THC, HC, LGC, and SGC counts [ref. 7]. Enhancing the circulating haemocyte count through diet manipulation could induce the activity of humoral factors. Phenoloxidase is the final product in the prophenoloxidase (proPO) system and its activation plays a vital role in the recognition and control of hemolymph infection [ref. 63]. Lysozyme acts as an antibacterial molecule in the hydrolysis of the bacterial peptidoglycan layer. Our findings revealed that supplementation of LS1PE1 elevated the PO and LYZ levels of narrow-clawed crayfish serum [ref. 64]. These results are in agreement with the immunity findings of L. vannamei after feeding with pectin and probiotics, synergistic effects between pectin and L. plantarum were also observed [ref. 30]. Alkaline phosphatase (AKP) is a type of lysosomal enzyme related to various key functions such as hydrolytic activities, antibacterial, and wound healing in all animals [ref. 65]. Acid phosphatase (ACP) is another known lysosomal enzyme in the degradation of microbial pathogens and is considered an indicator to assess the capability of macrophages to digest invader agents [ref. 63]. Crayfish fed with diets containing LS, PE, and LSPE illustrated a significant increase in AKP activity in serum when compared to control. Similarly, mannanoligosaccharide + Pediococcus acidilactici and xylooligosaccharide + E. faecalis, improved the activity of AKP in the hemolymph of crayfish [ref. 7]. Our findings demonstrated that LS or PE added to the crayfish diet beneficially regulated AKP activity in crayfish. Nitric oxide (NO) is a bactericidal molecule against extracellular and intracellular pathogens in the nonspecific immune systems of many organisms [ref. 66]. In the current study, NOs activity was remarkably influenced by diets probiotic (LS, LSPE). Similarly, a rise in NOs level was reported using dietary E. faecalis + galactooligosaccharide and E. faecalis + mannanoligosaccharidein in A. leptodactylus [ref. 8]. These results confirmed that the compounds generated in the presence of PE by LS can be useful for regulating the immunocompetence of crayfish via both the complementary and synergistic effects. Enhancing disease resistance against common pathogens is the end product of dietary supplements. In the current study, diets containing synbiotic (pectin + probiotic) elevated the survival rate of the narrow-clawed crayfish infected with A. hydrophila. Indeed, LS1PE1 administrated into diet protected narrow-clawed crayfish up to 44% against infection. The finding is consistent with the observers of other authors, who indicated the stimulating effects of synbiotics on the immunocompetence and resistance of shellfish and finfish species against biological stressors [ref. 56, ref. 67, ref. 68]. Increased colonization of lactic acid bacteria in the presence of pectin, the release of bacteriocins, and boosting of the immune defense elements including LYZ, AKP, NO, PO, and haemocytes are possible mechanisms to enhance disease resistance in LSPE groups.
Coping with oxidative stress in aquatic animals is accomplished through a set of antioxidant enzymes such as SOD, CAT, and GPx [ref. 69]. These enzymes play a vital role in maintaining cell integrity by scavenging reactive oxygen species (ROS) [ref. 70]. However, in harsh conditions, the host’s antioxidant capacity is not adequate to scavenge ROS, thus external resources are needed to boost antioxidant capacity [ref. 71, ref. 72]. Adding stimulant compounds to the aquafeed is one of the commonest ways to improve the antioxidant defense in fish. In this study, the dietary LSPE indicated high antioxidant capacity where SOD and GPx activities remarkably increased. Besides, MDA levels as a good marker for measuring oxidative stress (imbalance between antioxidant defense and ROS level) were significantly reduced in response to synbiotic diets [ref. 70]. These results may be because of the ability of pectin to boost antioxidant capacity or ROS scavenging, previously reported in carp by Hoseinifar et al. [ref. 73]. On the other hand, the beneficiary properties of probiotics on the antioxidant capacity may be associated to resist ROS, chelating ions, and producing compounds with antioxidant properties such as folate, butyrate, and glutathione [ref. 74]. Therefore, inclusion synbiotic in the narrow-clawed crayfish diet can enhance the antioxidant power of the crayfish and support the cells versus the harmful effects of oxidative stress.
5. Conclusion
In conclusion, the present findings indicated that the dietary L. salivarius or pectin could enhance growth performance and hemolymph indices. However, administration of synbiotic, especially LS1PE1, had better performance on digestive enzyme activity, modulation of gut microflora, nonspecific immune responses, antioxidant capacity, as well as disease resistance against Aeromonas hydrophila. Accordingly, dietary supplementation with LS1PE1 can be considered a beneficial feed additive in crayfish diet.
References
- J. A. Pradeepkiran. Aquaculture role in global food security with nutritional value: a review. Translational Animal Science, 2019. [DOI | PubMed]
- M. M. Harlıoğlu, A. Farhadi. Factors affecting the reproductive efficiency in crayfish: implications for aquaculture. Aquaculture Research, 2017. [DOI]
- A. Kouba, H. Niksirat, I. Kuklina, M. Buřič, P. Kozák. Ultraviolet light and semi-recirculating systems in artificial incubation of noble crayfish (Astacus astacus) eggs: opportunities and limitations. Aquaculture Research, 2012. [DOI]
- G. Vogt. Evaluation of the suitability of the parthenogenetic marbled crayfish for aquaculture: potential benefits versus conservation concerns. Hydrobiologia, 2021. [DOI]
- S. Nedaei, A. Noori, A. Valipour, A. A. Khanipour, S. H. Hoseinifar. Effects of dietary galactooligosaccharide enriched commercial prebiotic on growth performance, innate immune response, stress resistance, intestinal microbiota and digestive enzyme activity in narrow clawed crayfish (Astacus leptodactylus_ Eschscholtz, 1823). Aquaculture, 2019. [DOI]
- A. Valipour, S. Nedaei, A. Noori, A. A. Khanipour, S. H. Hoseinifar. Dietary _Lactobacillus plantarum_ affected on some immune parameters, air- exposure stress response, intestinal microbiota, digestive enzyme activity and performance of narrow clawed crayfish (Astacus leptodactylus_ , Eschscholtz). Aquaculture, 2019. [DOI]
- O. Safari, M. Paolucci, H. A. Motlagh. Effects of synbiotics on immunity and disease resistance of narrow-clawed crayfish, _Astacus leptodactylus leptodactylus_ (Eschscholtz, 1823). Fish & Shellfish Immunology, 2017. [DOI | PubMed]
- O. Safari, M. Paolucci. Modulation of growth performance, immunity, and disease resistance in narrow- clawed crayfish, _Astacus leptodactylus leptodactylus_ (Eschscholtz, 1823) upon synbiotic feeding. Aquaculture, 2017. [DOI]
- E. Ringø, S. K. Song. Application of dietary supplements (synbiotics and probiotics in combination with plant products and β-glucans) in aquaculture. Aquaculture Nutrition, 2016. [DOI]
- S. Yilmaz, E. Yilmaz, M. A. Dawood, E. Ringø, E. Ahmadifar, H. M. Abdel-Latif. Probiotics, prebiotics, and synbiotics used to control vibriosis in fish: a review. Aquaculture, 2022. [DOI]
- U. D. Butt, N. Lin, N. Akhter, T. Siddiqui, S. Li, B. Wu. Overview of the latest developments in the role of probiotics, prebiotics and synbiotics in shrimp aquaculture. Fish & Shellfish Immunology, 2021. [DOI | PubMed]
- S. H. Hoseinifar, E. Ringø, A. Shenavar Masouleh, M. Á. Esteban. Probiotic, prebiotic and synbiotic supplements in sturgeon aquaculture: a review. Reviews in Aquaculture, 2016. [DOI]
- T. G. Huynh, Y. L. Shiu, T. P. Nguyen, Q. P. Truong, J. C. Chen, C. H. Liu. Current applications, selection, and possible mechanisms of actions of synbiotics in improving the growth and health status in aquaculture: a review. Fish & Shellfish Immunology, 2017. [DOI | PubMed]
- M. F. Rohani, S. M. Islam, M. K. Hossain. Probiotics, prebiotics and synbiotics improved the functionality of aquafeed: upgrading growth, reproduction, immunity and disease resistance in fish. Fish & Shellfish Immunology, 2021. [DOI | PubMed]
- S. H. Hoseinifar, S. Yousefi, H. Van Doan. Oxidative stress and antioxidant defense in fish: the implications of probiotic, prebiotic, and synbiotics. Reviews in Fisheries Science & Aquaculture, 2021
- B. D. Chaves, M. M. Brashears, K. K. Nightingale. Applications and safety considerations of Lactobacillus salivarius as a probiotic in animal and human health. Journal of Applied Microbiology, 2017. [DOI | PubMed]
- X. Chen, M. Ishfaq, J. Wang. Effects of _Lactobacillus salivarius_ supplementation on the growth performance, liver function, meat quality, immune responses and _Salmonella_ Pullorum infection resistance of broilers challenged with aflatoxin B1. Poultry Science, 2022. [DOI | PubMed]
- S. Sureshkumar, S. K. Jung, D. Kim. Administration of L. salivarius expressing 3D8 scFv as a feed additive improved the growth performance, immune homeostasis, and gut microbiota of chickens. Animal Science Journal, 2020. [DOI | PubMed]
- J. Wang, M. Ishfaq, Y. Guo, C. Chen, J. Li. Assessment of probiotic properties of Lactobacillus salivarius isolated from chickens as feed additives. Frontiers in Veterinary Science, 2020. [DOI | PubMed]
- J. Xia, S. Jiang, L. Lv. Modulation of the immune response and metabolism in germ-free rats colonized by the probiotic Lactobacillus salivarius LI01. Applied Microbiology and Biotechnology, 2021. [DOI | PubMed]
- M. S. Elshahed, A. Miron, A. C. Aprotosoaie, M. A. Farag. Pectin in diet: interactions with the human microbiome, role in gut homeostasis, and nutrient-drug interactions. Carbohydrate Polymers, 2021. [DOI | PubMed]
- H. Van Doan, S. H. Hoseinifar, P. Elumalai. Effects of orange peels derived pectin on innate immune response, disease resistance and growth performance of Nile tilapia (Oreochromis niloticus) cultured under indoor biofloc system. Fish & Shellfish Immunology, 2018. [DOI | PubMed]
- R. Ciriminna, A. Fidalgo, F. Meneguzzo. Pectin: a long-neglected broad-Spectrum antibacterial. ChemMedChem, 2020. [DOI | PubMed]
- W. Zhang, P. Xu, H. Zhang. Pectin in cancer therapy: a review. Trends in Food Science & Technology, 2015. [DOI]
- D. Q. Li, J. Li, H. L. Dong. Pectin in biomedical and drug delivery applications: a review. International Journal of Biological Macromolecules, 2021. [DOI | PubMed]
- B. Gómez, B. Gullón, R. Yáñez, H. Schols, J. L. Alonso. Prebiotic potential of pectins and pectic oligosaccharides derived from lemon peel wastes and sugar beet pulp: a comparative evaluation. Journal of Functional Foods, 2016. [DOI]
- S. L. Edirisinghe, S. H. S. Dananjaya, C. Nikapitiya. Novel pectin isolated from _Spirulina maxima_ enhances the disease resistance and immune responses in zebrafish against _Edwardsiella piscicida_ and _Aeromonas hydrophila_. Fish & Shellfish Immunology, 2019. [DOI | PubMed]
- S. H. Hoseinifar, M. A. Jahazi, R. Mohseni. Dietary apple peel-derived pectin improved growth performance, antioxidant enzymes and immune response in common carp, _Cyprinus carpio_ (Linnaeus, 1758). Aquaculture, 2021. [DOI]
- S. M. Hosseini, S. H. Hoseinifar, M. Mazandarani, H. Paknejad, H. Van Doan, E. R. El-Haroun. The potential benefits of orange peels derived pectin on serum and skin mucus immune parameters, antioxidant defence and growth performance in common carp (Cyprinus carpio). Fish & Shellfish Immunology, 2020. [DOI | PubMed]
- H. W. Kuo, C. C. Chang, W. Cheng. Synbiotic combination of prebiotic, cacao pod husk pectin and probiotic, _Lactobacillus plantarum_ , improve the immunocompetence and growth of _Litopenaeus vannamei_. Fish & Shellfish Immunology, 2021. [DOI | PubMed]
- C. N. Zheng, W. Wang. Effects of Lactobacillus pentosus on the growth performance, digestive enzyme and disease resistance of white shrimp, Litopenaeus vannamei (Boone, 1931). Aquaculture Research, 2017. [DOI]
- X. Zheng, Y. Duan, H. Dong, J. Zhang. Effects of dietary _Lactobacillus plantarum_ in different treatments on growth performance and immune gene expression of white shrimp _Litopenaeus vannamei_ under normal condition and stress of acute low salinity. Fish & Shellfish Immunology, 2017. [DOI | PubMed]
- Y. Z. Sun, H. L. Yang, R. L. Ma, W. Y. Lin. Probiotic applications of two dominant gut _Bacillus_ strains with antagonistic activity improved the growth performance and immune responses of grouper _Epinephelus coioides_. Fish & Shellfish Immunology, 2010. [DOI | PubMed]
- M. M. Yamashita, S. A. Pereira, L. Cardoso. Probiotic dietary supplementation in Nile tilapia as prophylaxis against streptococcosis. Aquaculture Nutrition, 2017. [DOI]
- O. Safari, D. Shahsavani, M. Paolucci, M. Mehraban Sang Atash. The effects of dietary nucleotide content on the growth performance, digestibility and immune responses of juvenile narrow clawed crayfish, Astacus Leptodactylus leptodactylus Eschscholtz, 1823. Aquaculture Research, 2015. [DOI]
- M. Banaee, M. Akhlaghi, S. Soltanian, A. Gholamhosseini, H. Heidarieh, M. S. Fereidouni. Acute exposure to chlorpyrifos and glyphosate induces changes in hemolymph biochemical parameters in the crayfish, _Astacus leptodactylus_ (Eschscholtz, 1823). Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 2019. [DOI | PubMed]
- C. C. Worthington. Worthington Enzyme Manual Related Biochemical, 1991
- F. L. García-Carreño. Protease inhibition in theory and practice. Biotechnology Education, 1992
- N. Iijima, S. Tanaka, Y. Ota. Purification and characterization of bile salt-activated lipase from the hepatopancreas of red sea bream, Pagrus major. Fish Physiology and Biochemistry, 1998. [DOI]
- S. H. Hoseinifar, A. Mirvaghefi, M. A. Amoozegar, D. L. Merrifield, E. Ringø. In vitro selection of a synbiotic and in vivo evaluation on intestinal microbiota, performance and physiological response of rainbow trout (Oncorhynchus mykiss) fingerlings. Aquaculture Nutrition, 2017. [DOI]
- G. Jiang, R. Yu, M. Zhou. Modulatory effects of ammonia-N on the immune system of _Penaeus japonicus_ to virulence of white spot syndrome virus. Aquaculture, 2004. [DOI | PubMed]
- J. E. Hose, G. G. Martin, A. S. Gerard. A decapod hemocyte classification scheme integrating morphology, cytochemistry, and function. The Biological Bulletin, 1990. [DOI | PubMed]
- A. E. Ellis. Lysozyme Assay. Techniques in Fish Immunology, 1990
- J. Hernández-López, T. Gollas-Galván, F. Vargas-Albores. Activation of the prophenoloxidase system of the brown shrimp _Penaeus californiensis_ Holmes. Comparative Biochemistry and Physiology Part C: Pharmacology, Toxicology and Endocrinology, 1996. [DOI]
- M. Marzinzig, A. K. Nussler, J. Stadler. Improved methods to measure end products of nitric oxide in biological fluids: nitrite, nitrate, and _S_ -nitrosothiols. Nitric Oxide, 1997. [DOI | PubMed]
- K. Hao, J. Y. Liu, F. Ling. Effects of dietary administration of _Shewanella haliotis_ D4, _Bacillus cereus_ D7 and _Aeromonas bivalvium_ D15, single or combined, on the growth, innate immunity and disease resistance of shrimp, _Litopenaeus vannamei_. Aquaculture, 2014. [DOI]
- O. Rezaei, M. S. Mehrgan, H. Paknejad. Dietary garlic (Allium sativum) powder improved zootechnical performance, digestive enzymes activity, and innate immunity in narrow-clawed crayfish (Postantacus leptodactylus). Aquaculture Reports, 2022. [DOI]
- O. Safari, D. Shahsavani, M. Paolucci, M. M. S. Atash. Single or combined effects of fructo- and mannan oligosaccharide supplements on the growth performance, nutrient digestibility, immune responses and stress resistance of juvenile narrow clawed crayfish, _Astacus leptodactylus leptodactylus_ Eschscholtz, 1823. Aquaculture, 2014. [DOI]
- H. M. Sang, R. Fotedar. Dietary supplementation of mannan oligosaccharide improves the immune responses and survival of marron, _Cherax tenuimanus_ (Smith, 1912) when challenged with different stressors. Fish & Shellfish Immunology, 2009. [DOI | PubMed]
- E. Ringø, H. Van Doan, S. H. Lee. Probiotics, lactic acid bacteria and bacilli: interesting supplementation for aquaculture. Journal of Applied Microbiology, 2020. [DOI | PubMed]
- M. A. Dawood, S. Koshio, M. Á. Esteban. Beneficial roles of feed additives as immunostimulants in aquaculture: a review. Reviews in Aquaculture, 2018. [DOI]
- A. Wang, C. Ran, Y. Wang. Use of probiotics in aquaculture of China–a review of the past decade. Fish & Shellfish Immunology, 2019. [DOI | PubMed]
- E. Prabawati, S. Y. Hu, S. T. Chiu, R. Balantyne, Y. Risjani, C. H. Liu. A synbiotic containing prebiotic prepared from a by-product of king oyster mushroom, _Pleurotus eryngii_ and probiotic, _Lactobacillus plantarum_ incorporated in diet to improve the growth performance and health status of white shrimp, _Litopenaeus vannamei_. Fish & Shellfish Immunology, 2022. [DOI | PubMed]
- T. G. Huynh, A. C. Cheng, C. C. Chi, K. H. Chiu, C. H. Liu. A synbiotic improves the immunity of white shrimp, _Litopenaeus vannamei_ : metabolomic analysis reveal compelling evidence. Fish & Shellfish Immunology, 2018. [DOI | PubMed]
- S. H. Hoseinifar, M. Á. Esteban, A. Cuesta, Y. Z. Sun. Prebiotics and fish immune response: a review of current knowledge and future perspectives. Reviews in Fisheries Science & Aquaculture, 2015. [DOI]
- G. Mohammadi, M. Hafezieh, A. A. Karimi. The synergistic effects of plant polysaccharide and _Pediococcus acidilactici_ as a synbiotic additive on growth, antioxidant status, immune response, and resistance of Nile tilapia (Oreochromis niloticus) against _Aeromonas hydrophila_. Fish & Shellfish Immunology, 2022. [DOI | PubMed]
- M. A. Dawood, S. Koshio. Application of fermentation strategy in aquafeed for sustainable aquaculture. Reviews in Aquaculture, 2020. [DOI]
- T. G. Huynh, C. C. Chi, T. P. Nguyen, T. T. T. H. Tran, A. C. Cheng, C. H. Liu. Effects of synbiotic containing Lactobacillus plantarum 7–40 and galactooligosaccharide on the growth performance of white shrimp, Litopenaeus vannamei. Aquaculture Research, 2018. [DOI]
- Z. H. Zuo, B. J. Shang, Y. C. Shao, W. Y. Li, J. S. Sun. Screening of intestinal probiotics and the effects of feeding probiotics on the growth, immune, digestive enzyme activity and intestinal flora of _Litopenaeus vannamei_. Fish & Shellfish Immunology, 2019. [DOI | PubMed]
- Y. Bouallegui. A comprehensive review on crustaceans’ immune system with a focus on freshwater crayfish in relation to crayfish plague disease. Frontiers in Immunology, 2021. [DOI | PubMed]
- J. Rodrıguez, G. Le Moullac. State of the art of immunological tools and health control of penaeid shrimp. Aquaculture, 2000. [DOI]
- A. Tassanakajon, K. Somboonwiwat, P. Supungul, S. Tang. Discovery of immune molecules and their crucial functions in shrimp immunity. Fish & Shellfish Immunology, 2013. [DOI | PubMed]
- B. Zhang, Z. R. Shi, X. L. Wang, S. G. Deng. The effects of hairtail protein hydrolysate–Fe2+ complexes on growth and non-specific immune response of red swamp crayfish (Procambarus clarkii). Aquaculture International, 2016. [DOI]
- H. Li, T. E. Zhang, Q. Li. Influence of compound Chinese herbal medicine on nonspecific immunity of turbot Scophthalmus maximus. Journal of Dalian Ocean University, 2013
- Q. X. Chen, W. Z. Zheng, J. Y. Lin, Y. Shi, W. Z. Xie, H. M. Zhou. Effect of metal ions on the activity of green crab (Scylla serrata) alkaline phosphatase. The International Journal of Biochemistry & Cell Biology, 2000. [DOI | PubMed]
- T. Rodríguez-Ramos, Y. Carpio, J. Bolívar, L. Gómez, M. P. Estrada, C. Pendón. Nitric oxide synthase-dependent immune response against gram negative bacteria in a crustacean, _Litopenaeus vannamei_. Fish & Shellfish Immunology, 2016. [DOI | PubMed]
- G. El-Nobi, M. Hassanin, A. A. Khalil. Synbiotic effects of Saccharomyces cerevisiae, Mannan oligosaccharides, and β-glucan on innate immunity, antioxidant status, and disease resistance of Nile tilapia, Oreochromis niloticus. Antibiotics, 2021. [DOI | PubMed]
- W. Yao, X. Li, C. Zhang, J. Wang, Y. Cai, X. Leng. Effects of dietary synbiotics supplementation methods on growth, intestinal health, non-specific immunity and disease resistance of Pacific white shrimp, _Litopenaeus vannamei_. Fish & Shellfish Immunology, 2021. [DOI | PubMed]
- A. El-Gawad, A. Eman, A. M. El Asely, E. I. Soror, A. A. Abbass, B. Austin. Effect of dietary Moringa oleifera leaf on the immune response and control of Aeromonas hydrophila infection in Nile tilapia (Oreochromis niloticus) fry. Aquaculture International, 2020. [DOI]
- V. Aliko, M. Qirjo, E. Sula, V. Morina, C. Faggio. Antioxidant defense system, immune response and erythron profile modulation in gold fish, _Carassius auratus_ , after acute manganese treatment. Fish & Shellfish Immunology, 2018. [DOI | PubMed]
- H. S. Hamed, S. M. Ismal, C. Faggio. Effect of allicin on antioxidant defense system, and immune response after carbofuran exposure in Nile tilapia, Oreochromis niloticus. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 2021
- Y. Tang, L. Han, X. Chen, M. Xie, W. Kong, Z. Wu. Dietary supplementation of probiotic Bacillus subtilis affects antioxidant defenses and immune response in grass carp under Aeromonas hydrophila challenge. Probiotics and Antimicrobial Proteins, 2019. [DOI | PubMed]
- S. H. Hoseinifar, Y. Z. Sun, A. Wang, Z. Zhou. Probiotics as means of diseases control in aquaculture, a review of current knowledge and future perspectives. Frontiers in Microbiology, 2018. [DOI | PubMed]
- A. Amaretti, M. Di Nunzio, A. Pompei, S. Raimondi, M. Rossi, A. Bordoni. Antioxidant properties of potentially probiotic bacteria: in vitro and in vivo activities. Applied Microbiology and Biotechnology, 2013. [DOI | PubMed]
