Body weight and composition endpoints in cancer cachexia clinical trials: Systematic Review 4 of the cachexia endpoints series
Abstract
Significant variation exists in the outcomes used in cancer cachexia trials, including measures of body composition, which are often selected as primary or secondary endpoints. To date, there has been no review of the most commonly selected measures or their potential sensitivity to detect changes resulting from the interventions being examined. The aim of this systematic review is to assess the frequency and diversity of body composition measures that have been used in cancer cachexia trials. MEDLINE, Embase and Cochrane Library databases were systematically searched between January 1990 and June 2021. Eligible trials examined adults (≥18 years) who had received an intervention aiming to treat or attenuate the effects of cancer cachexia for >14 days. Trials were also of a prospective controlled design and included body weight or at least one anthropometric, bioelectrical or radiological endpoint pertaining to body composition, irrespective of the modality of intervention (e.g., pharmacological, nutritional, physical exercise and behavioural) or comparator. Trials with a sample size of <40 patients were excluded. Data extraction used Covidence software, and reporting followed the Preferred Reporting Items for Systematic Reviews and Meta‐Analyses guidance. This review was prospectively registered (PROSPERO: CRD42022276710). A total of 84 clinical trials, comprising 13 016 patients, were eligible for inclusion. Non‐small‐cell lung cancer and pancreatic cancer were studied most frequently. The majority of trial interventions were pharmacological (52%) or nutritional (34%) in nature. The most frequently reported endpoints were assessments of body weight (68 trials, n = 11 561) followed by bioimpedance analysis (BIA)‐based estimates (23 trials, n = 3140). Sixteen trials (n = 3052) included dual‐energy X‐ray absorptiometry (DEXA)‐based endpoints, and computed tomography (CT) body composition was included in eight trials (n = 841). Discrepancies were evident when comparing the efficacy of interventions using BIA‐based estimates of lean tissue mass against radiological assessment modalities. Body weight, BIA and DEXA‐based endpoints have been most frequently used in cancer cachexia trials. Although the optimal endpoints cannot be determined from this review, body weight, alongside measurements from radiological body composition analysis, would seem appropriate. The choice of radiological modality is likely to be dependent on the trial setting, population and intervention in question. CT and magnetic resonance imaging, which have the ability to accurately discriminate tissue types, are likely to be more sensitive and provide greater detail. Endpoints are of particular importance when aligned with the intervention’s mechanism of action and/or intended patient benefit.
Article type: Review Article
Keywords: body composition, cachexia, cancer cachexia, clinical trials
Affiliations: Clinical Surgery The University of Edinburgh, Royal Infirmary of Edinburgh Edinburgh UK; Improving Palliative, Aged and Chronic Care Through Clinical Research and Translation (IMPACCT) University of Technology Sydney Sydney Australia; Institute of Genetics and Cancer The University of Edinburgh, Western General Hospital Edinburgh UK; St Columba’s Hospice Care Edinburgh UK; Department of Oncology University of Alberta Edmonton Alberta Canada; Academic Unit of Surgery University of Glasgow, Glasgow Royal Infirmary Glasgow UK; Department of Medicine I, Medical Centre—University of Freiburg Faculty of Medicine University of Freiburg Freiburg Germany; Department of Clinical and Molecular Medicine, Faculty of Medicine and Health Sciences Norwegian University of Science and Technology Trondheim Norway; Department of Clinical Medicine, Clinical Nutrition Research Group UiT The Arctic University of Norway Tromsø Norway; Department of Oncology Oslo University Hospital Oslo Norway; Department of Nursing and Health Promotion, Faculty of Health Sciences Oslo Metropolitan University Oslo Norway; Department of Public Health and Nursing, Faculty of Medicine and Health Sciences Norwegian University of Science and Technology Trondheim Norway; Cancer Clinic St. Olav’s Hospital, Trondheim University Hospital Trondheim Norway; Cicely Saunders Institute of Palliative Care, Policy and Rehabilitation King’s College London London UK; Department of Nutrition, Institute of Basic Medical Sciences University of Oslo Oslo Norway; The Clinical Nutrition Outpatient Clinic, Section of Clinical Nutrition, Department of Clinical Service, Division of Cancer Medicine Oslo University Hospital Oslo Norway; Edinburgh Cancer Centre Western General Hospital Edinburgh UK
License: © 2024 The Authors. Journal of Cachexia, Sarcopenia and Muscle published by Wiley Periodicals LLC. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/jcsm.13478 | PubMed: 38738581 | PMC: PMC11154800
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.5 MB)
Introduction
Cancer cachexia is a complex multifactorial syndrome characterized by loss of muscle and body fat.ref. jcsm13478-bib-0001 These changes are strongly associated with poorer quality of life, increased morbidity and worse survival.ref. jcsm13478-bib-0002 The 2011 consensus definition for cancer cachexia provided diagnostic and staging criteria that have been instrumental in aiding cachexia trial design.ref. jcsm13478-bib-0001 At present, there is no similar consensus regarding endpoints, and significant variations remain amongst the clinical assessments used in cancer cachexia trials.
A comprehensive patient assessment of cachexia would consider changes in body composition, dietary intake, biomarkers of the pathophysiological drivers of cachexia, physical function and quality of life, and the influence on associated oncological outcomes. Depending on the mechanism of a given clinical trial intervention, particular weighting may be assigned to chosen measures within this broad range. Selected endpoints must be both sensitive enough to detect change and specific enough not to be readily influenced by other conditions or treatments. Furthermore, it is imperative that they convey clinical relevance.
Endpoints pertaining to body weight and composition are amongst the most frequently reported in cachexia trials and will be the focus of this review. Anthropometric measurements and electric bioimpedance analysis (BIA) are simple modalities that, although inexpensive and non‐invasive, are prone to confounders and provide finite levels of detail. Dual‐energy X‐ray absorptiometry (DEXA) is widely available and can provide estimates of regional/whole‐body fat or lean tissue mass. However, DEXA is unable to discriminate between different types of ‘lean tissue’ (e.g., skeletal muscle vs. organs) or anatomical locations (e.g., visceral vs. subcutaneous adipose tissue).ref. jcsm13478-bib-0003 While cachexia research has traditionally focused on the loss of muscle, it is now known that adipose tissue also plays an important role in cachexia pathophysiology, and different mechanisms underpin the loss of each tissue type.ref. jcsm13478-bib-0004 As such, the ability of modalities to distinguish between body tissue compartments is of increasing relevance. Computed tomography (CT) and magnetic resonance imaging (MRI) scans are considered the ‘gold‐standard’ assessment modalities for body composition owing to their specificity in discriminating tissue identities and their precision.ref. jcsm13478-bib-0005 Comparison of the two has shown high levels of agreement in assessments of muscle quantity and qualityref. jcsm13478-bib-0006; however, CT has been more frequently utilized in cachexia research owing to its more widespread use in routine clinical practice.
At present, it is not known what the best endpoints for cancer cachexia trials are. This may have resulted in sub‐optimal clinical trial design, which could have in turn hindered the development of effective therapies. An appraisal of the endpoints currently used would seem like a logical starting point. The aim of this systematic review is to assess the frequency and diversity of measures that have been used to assess body weight and body composition in cancer cachexia trials.
Methods
This systematic review is reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta‐Analyses (PRISMA) statement.ref. jcsm13478-bib-0007 The review protocol was prospectively registered at the International Prospective Register of Systematic Reviews: PROSPERO (CRD42022276710).ref. jcsm13478-bib-0008
This review will address assessments of body weight, alongside anthropometric, bioelectrical or radiological endpoints pertaining to body composition. It is one of a series of six that will comprehensively evaluate the endpoints examined in cancer cachexia trials. Given the breadth of outcome measures in the literature, these were categorized broadly under the following domains: physical function,ref. jcsm13478-bib-0009 quality of life, appetite and dietary intake, body weight and composition, oncological outcomes and biomarkers.
Search strategy
A systematic search of MEDLINE (Ovid), Embase (Ovid) and Cochrane Central Register of Controlled Trials databases was conducted by a senior research librarian (University of Oslo). All published studies from 1 January 1990 to 2 June 2021 were eligible. Search results were synthesized and managed using the web‐based systematic review software ‘Covidence’ (Veritas Health Innovations, Melbourne, Australia), and duplicates were removed. A detailed search strategy is outlined in Appendix A.
Study eligibility criteria
Prospective clinical trials that considered an intervention aiming to treat or attenuate the effects of cachexia in adult patients (≥18 years) with cancer were considered for eligibility. Inclusion was irrespective of the site of primary malignancy, modality of intervention (e.g., pharmacological, nutritional and physical exercise) or choice of comparator. Articles were excluded if they studied fewer than 40 patients and/or if the intervention lasted <14 days. Studies in which patients underwent surgery during the assessment period were excluded. All included full‐text articles were written in the English language.
Data selection and extraction
The titles and abstracts of the identified studies were independently reviewed by three authors (OD, TSS and BJAL). Those selected were subsequently subject to full‐text review (LRB and MSS). In instances of discrepancies between reviewers regarding an article’s inclusion, consensus was reached through consultation between reviewers or with the wider authorship group. A pre‐defined data extraction table was developed and pilot‐tested before relevant data points were extracted independently by the lead authors (LRB and MSS).
Relevant outcome measures
Endpoints considered by this review were those pertaining to assessments of body weight and other modalities that aim to assess changes in body composition. These shall be categorized as anthropometric (e.g., body weight, circumference or skinfold measurements), bioelectrical (e.g., BIA) or radiological (e.g., DEXA, CT or MRI) measure of body composition.
Assessment of methodology and risk of bias
The methodological quality of each study was independently assessed by four reviewers (JS, JM, OD and BJAL) using the modified Downs and Black checklist.ref. jcsm13478-bib-0010 This tool assesses several criteria including study design, internal and external validity, and reporting standards.
Data analysis
Study characteristics, patient details and disease demographics were reported descriptively. The aim of this review was to describe the body weight and composition outcomes used, rather than estimate treatment effects. As such, quantitative meta‐analysis was not performed. Furthermore, the heterogenous nature of the trials and interventions studied made meta‐analysis of treatment effects on each endpoint impractical. Analyses and visualization were conducted using RStudio Version 4.2.2 (R Foundation for Statistical Computing, Vienna, Austria) with packages including maps and tidyverse.
Results
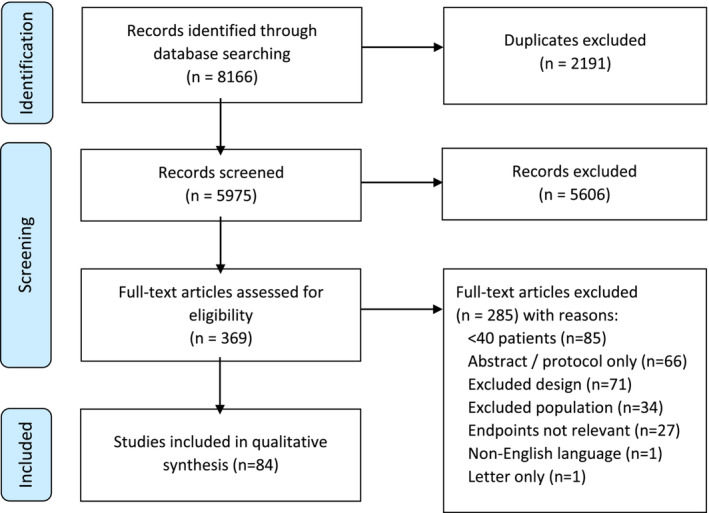
Overall, 8166 studies were identified following systematic searches of MEDLINE (Ovid), Embase (Ovid) and Cochrane Central Register of Controlled Trials databases (Appendix A). Following the removal of duplicates (n = 2191), further screening of the title and abstract for 5975 studies was performed. Of these, 5606 articles were excluded and 369 were retrieved for full‐text review. Following detailed screening against the chosen inclusion and exclusion criteria, 84 clinical trials were eligible for inclusion. The PRISMA flow chart is detailed in Figure jcsm13478-fig-0001.
Study characteristics
Between 1990 and 2021, a total of 84 prospective clinical trials (n = 13 016 participants) included body weight or measure(s) of body composition as an endpoint. While numerous primary tumour sites were considered, pancreatic cancer (n = 11 trials) and non‐small‐cell lung cancer (n = 10 trials) were the most frequently studied. Cohorts ranged in size, with the largest cohort being 979 patients studied by the ROMANA 1 and 2 trials.ref. jcsm13478-bib-0011 Pharmacological interventions (n = 43 trials) were most evaluated, followed by nutritional (n = 28 trials), multi‐modal (n = 9 trials) and exercise‐based modalities (n = 4 trials). The key characteristics of the included trials are detailed in Table jcsm13478-tbl-0001.
Table 1: Key characteristics of included clinical trials
| Author (reference) | Year | Sample size | Study design | Study quality | Primary cancer site | Intervention | Comparator | Body composition outcomesjcsm13478-note-0004 |
|---|---|---|---|---|---|---|---|---|
| Kardinal et al.ref. jcsm13478-bib-0012 | 1990 | 293 | RCT | 8 | Any malignancy (not brain) | Cyproheptadine(pharmacological) | Placebo | Body weightjcsm13478-note-0006 (primary) |
| Loprinzi et al.ref. jcsm13478-bib-0013 | 1990 | 133 | RCT | 9 | Any malignancy (not brain/breast/endometrial) | Megestrol acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0005,jcsm13478-note-0006(primary) |
| Feliu et al.ref. jcsm13478-bib-0014 | 1992 | 150 | RCT | 5 | Any malignancy (not hormone dependent) | Megestrol acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0005 (primary) |
| Downer et al.ref. jcsm13478-bib-0015 | 1993 | 60 | RCT | 1 | Any malignancy | Medroxyprogesterone acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0005 (primary)Mid‐arm circumferenceTriceps skinfold thickness |
| Loprinzi et al.ref. jcsm13478-bib-0016 | 1993 | 342 | Phase III RCT | 8 | Any malignancy (not breast/endometrial) | Megestrol acetate 1280 mg or megestrol acetate 800 mg(pharmacological) | Megestrol acetate 480 mg or megestrol acetate 160 mg | Body weightjcsm13478-note-0006 (primary) |
| Ovesen et al.ref. jcsm13478-bib-0017 | 1993 | 105 | RCT | 8 | Small‐cell‐lung/ovarian/breast | Nutritional counselling(nutritional) | Standard care | Body weightjcsm13478-note-0005 (primary)Arm muscle areaTriceps skinfold thickness |
| Goldberg et al.ref. jcsm13478-bib-0018 | 1995 | 70 | RCT | 8 | Any malignancy (not primary brain tumour) | Pentoxifylline(pharmacological) | Placebo | Body weightjcsm13478-note-0006 (primary) |
| Gebbia et al.ref. jcsm13478-bib-0019 | 1996 | 122 | RCT | 6 | Any malignancy (not hormone dependent) | Megestrol acetate 320 mg(pharmacological) | Megestrol acetate 160 mg | Body weight(primary) |
| Lissoni et al.ref. jcsm13478-bib-0020 | 1996 | 100 | RCT | 7 | Any solid tumour | Melatonin(pharmacological) | Standard care | Body weightjcsm13478-note-0005(primary) |
| Simons et al.ref. jcsm13478-bib-0021 | 1996 | 206 | RCT | 7 | Any malignancy (not hormone dependent) | Medroxyprogesterone acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0005(primary) |
| Beller et al.ref. jcsm13478-bib-0022 | 1997 | 240 | RCT | 4 | Any malignancy (not hormone dependent) | Megestrol acetate 480 mg or megestrol acetate 160 mg(pharmacological) | Placebo | Body weightjcsm13478-note-0005 (primary)Mid‐arm circumferenceMid‐arm fat and muscle areaTriceps skinfold thickness |
| Chen et al.ref. jcsm13478-bib-0023 | 1997 | 129 | RCT | 8 | Head and neck | Megestrol acetate or prepulside(pharmacological) | Placebo | Body weightjcsm13478-note-0005(primary) |
| Daneryd et al.ref. jcsm13478-bib-0024 | 1998 | 180 | RCT | 7 | Any malignancy | Indomethacin + erythropoietin(pharmacological) | Indomethacin | Lean body mass—DEXABody weightjcsm13478-note-0005 |
| De Conno et al.ref. jcsm13478-bib-0025 | 1998 | 42 | RCT | 6 | Any malignancy (not hormone dependent) | Megestrol acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0005(primary) |
| Vadell et al.ref. jcsm13478-bib-0026 | 1998 | 150 | RCT | 5 | Any malignancy | Megestrol acetate 480 mg or megestrol acetate 160 mg(pharmacological) | Placebo | Body weightjcsm13478-note-0005(primary)Mid‐arm circumferenceTriceps skinfold thickness |
| Loprinizi et al.ref. jcsm13478-bib-0027 | 1999 | 496 | RCT | 8 | Any malignancy (not breast/prostate/ovarian/endometrial) | Megestrol acetate or dexamethasone(pharmacological) | Fluoxymesterone | Body weightjcsm13478-note-0006 (primary) |
| McMillan et al.ref. jcsm13478-bib-0028 | 1999 | 73 | RCT | 7 | Gastrointestinal | Megestrol acetate + ibuprofen(pharmacological) | Megestrol acetate + placebo | Body weightjcsm13478-note-0005(primary)Mid‐arm circumferenceTriceps skinfold thicknessBiceps skinfold thickness |
| Westman et al.ref. jcsm13478-bib-0029 | 1999 | 255 | RCT | 7 | Other mixed | Megestrol acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0005 (primary) |
| Jatoi et al.ref. jcsm13478-bib-0030 | 2002 | 469 | RCT | 10 | Any malignancy (not brain/breast/ovarian/endometrial) | Megestrol acetate + dronabinol or megestrol acetate + placebo(pharmacological) | Dronabinol + placebo | Body weightjcsm13478-note-0006(primary) |
| Persson et al.ref. jcsm13478-bib-0031 | 2002 | 144 | RCT | 6 | Breast/colorectal/gastric/prostate | Individual nutritional counselling or individual and group nutritional counselling(nutritional) | Group nutritional counselling or standard care | Body weightjcsm13478-note-0006(primary) |
| Ulutin et al.ref. jcsm13478-bib-0032 | 2002 | 119 | RCT | 9 | NSCLC | Megestrol acetate 320 mg(pharmacological) | Megestrol acetate 160 mg | Body weight(increase vs. stable vs. decrease) |
| Bruera et al.ref. jcsm13478-bib-0033 | 2003 | 91 | RCT | 7 | Any malignancy | Fish oil capsules(nutritional) | Placebo | Lean body mass—BIABody weightjcsm13478-note-0005 Mid‐arm muscle circumferenceTriceps skinfold thicknessSubscapular skinfold thickness |
| Fearon et al.ref. jcsm13478-bib-0034 | 2003 | 200 | RCT | 8 | Pancreatic | n‐3 fatty acid and antioxidant‐enriched supplement(nutritional) | Supplement without n‐3 fatty acid and antioxidants | Lean body mass—BIABody weightjcsm13478-note-0005 |
| Isenring et al.ref. jcsm13478-bib-0035 | 2004 | 60 | RCT | 8 | Gastrointestinal/head and neck | Nutrition counselling and protocol(nutritional) | Standard care | Fat‐free mass—BIABody weightjcsm13478-note-0005 |
| Lundholm et al.ref. jcsm13478-bib-0036 | 2004 | 309 | RCT | 5 | Any solid tumour | Indomethacin + erythropoietin + nutritional support + home total parenteral nutrition(multi‐modal) | Indomethacin + erythropoietin | Fat mass—DEXALean body mass—DEXABody weightjcsm13478-note-0005 Mid‐arm muscle circumferenceTriceps skinfold thickness |
| Gonçalves Dias et al.ref. jcsm13478-bib-0037 | 2005 | 64 | Non‐randomized trial | 1 | Head and neck | Home enteral (nasogastric) feeding or oral diet + nutritional supplements(nutritional) | Oral diet | Body weightjcsm13478-note-0005 BMIMid‐arm circumferenceMid‐arm muscle areaTriceps skinfold thickness |
| Gordon et al.ref. jcsm13478-bib-0038 | 2005 | 50 | RCT | 10 | Pancreatic | Thalidomide(pharmacological) | Placebo | Body weightjcsm13478-note-0005(primary)Bone‐free arm muscle area |
| Fearon et al.ref. jcsm13478-bib-0039 | 2006 | 518 | RCT | 8 | Gastrointestinal/lung | EPA 2 g or EPA 4 g(pharmacological) | Placebo | Lean body mass—BIABody weightjcsm13478-note-0005 |
| Berk et al.ref. jcsm13478-bib-0040 | 2008 | 472 | RCT | 9 | Any solid tumour | Nutritional supplement(nutritional) | Placebo | Lean body mass—BIA(primary)Body weightjcsm13478-note-0006 Various skinfold thickness |
| Wiedenmann et al.ref. jcsm13478-bib-0041 | 2008 | 86 | Phase II RCT | 7 | Pancreatic | Infliximab 5 mg/kg or infliximab 3 mg/kg(pharmacological) | Placebo | Lean body mass—BIA(primary) |
| Beijer et al.ref. jcsm13478-bib-0042 | 2009 | 100 | RCT | 8 | Any malignancy | Adenosine 5′‐triphosphate(pharmacological) | Standard care | Triceps skinfold thickness(primary)Body weightjcsm13478-note-0005 Mid‐arm circumference |
| Mantovani et al.ref. jcsm13478-bib-0043 | 2010 | 332 | Phase III RCT | 7 | Any malignancy | Megestrol acetate or EPA‐enriched nutritional supplement or l‐carnitine or thalidomide(pharmacological) | Megestrol acetate + EPA‐enriched nutritional supplement + l‐carnitine + thalidomide | Lean body mass—DEXA(primary)Lean body mass—BIA(primary)Lean body mass—CT(primary) |
| Navari et al.ref. jcsm13478-bib-0044 | 2010 | 80 | RCT | 7 | Gastrointestinal/lung | Megestrol acetate + olanzapine(pharmacological) | Megestrol acetate | Body weightjcsm13478-note-0006(primary) |
| Baldwin et al.ref. jcsm13478-bib-0045 | 2011 | 358 | RCT | 8 | Gastrointestinal/NSCLC/mesothelioma | Nutritional supplement + nutritional counselling or nutritional supplement(nutritional) | Nutritional counselling or standard care | Body weightjcsm13478-note-0005 |
| Kraft et al.ref. jcsm13478-bib-0046 | 2012 | 72 | RCT | 10 | Pancreatic | l‐Carnitine supplement(nutritional) | Placebo | Fat mass—BIABMI |
| Macciò et al.ref. jcsm13478-bib-0047 | 2012 | 144 | Phase III RCT | 8 | Gynaecological | Megestrol acetate + l‐carnitine + celecoxib + antioxidants(pharmacological) | Megestrol acetate | Lean body mass—DEXA(primary) |
| Madeddu et al.ref. jcsm13478-bib-0048 | 2012 | 60 | Phase III RCT | 7 | Any malignancy | l‐Carnitine + celecoxib + megestrol acetate(pharmacological) | l‐Carnitine + celecoxib | Lean body mass—DEXA(primary)Lean body mass—CT(primary)Lean body mass—BIA(primary) |
| Silander et al.ref. jcsm13478-bib-0049 | 2012 | 134 | RCT | 6 | Head and neck | Prophylactic PEG(nutritional) | Standard care | Body weightjcsm13478-note-0006 (primary)BMI |
| Wen et al.ref. jcsm13478-bib-0050 | 2012 | 102 | RCT | 5 | Any malignancy | Megestrol acetate + thalidomide(pharmacological) | Megestrol acetate | Body weightjcsm13478-note-0005(primary) |
| Del Fabbro et al.ref. jcsm13478-bib-0051 | 2013 | 73 | RCT | 10 | Gastrointestinal/lung | Melatonin(pharmacological) | Placebo | Body weightjcsm13478-note-0005 (primary)Lean body mass—BIAFat‐free mass—BIA |
| Dobs et al.ref. jcsm13478-bib-0052 | 2013 | 159 | Phase II RCT | 8 | Other mixed | Enobosarm 1 mg or enobosarm 3 mg(pharmacological) | Placebo | Lean body mass—DEXA(primary)Body weightjcsm13478-note-0005 Fat mass—DEXA |
| Kanat et al.ref. jcsm13478-bib-0053 | 2013 | 69 | RCT | 8 | Any malignancy | Megestrol acetate + meloxicam or megestrol acetate + EPA‐enriched nutritional supplement(pharmacological) | Meloxicam + EPA‐enriched nutritional supplement | Body weightjcsm13478-note-0005 (primary)Lean body mass—BIA(primary)BMI |
| Poulsen et al.ref. jcsm13478-bib-0054 | 2013 | 61 | RCT | 5 | Oesophageal/gastric/gynaecological | Nutritional counselling(nutritional) | Standard care | Body weight(loss vs. maintenance)(primary)Fat mass—BIAFat‐free mass—BIA |
| Bourdel‐Marchasson et al.ref. jcsm13478-bib-0055 | 2014 | 336 | RCT | 10 | Other mixed | Nutritional counselling(nutritional) | Standard care | Body weightjcsm13478-note-0005 |
| Pottel et al.ref. jcsm13478-bib-0056 | 2014 | 85 | Exploratory RCT | 8 | Head and neck | Echium oil(nutritional) | Sunflower oil | Body weightjcsm13478-note-0006 (primary)Lean body mass—DEXALean body mass—BIAFat mass—DEXAFat mass—BIAFat‐free mass—BIA |
| Focan et al.ref. jcsm13478-bib-0057 | 2015 | 53 | RCT | 7 | Any malignancy | Dietetic and psychological mindfulness workshops(multi‐modal) | Standard care | BMI(primary)Body weightjcsm13478-note-0005 |
| Garcia et al.ref. jcsm13478-bib-0058 | 2015 | 82 | Phase II RCT | 7 | Any malignancy | Anamorelin 50 mg(pharmacological) | Placebo | Lean body mass—DEXA(primary)Appendicular LBM– DEXATotal body mass—DEXAFat mass—DEXA |
| Capozzi et al.ref. jcsm13478-bib-0059 | 2016 | 60 | Exploratory RCT | 8 | Head and neck | Early ‘lifestyle intervention’ (individualized exercise with education and support)(exercise) | Delayed ‘lifestyle intervention’ (individualized exercise with education and support) | Lean body mass—DEXAFat mass—DEXABMI |
| Kapoor et al.ref. jcsm13478-bib-0060 | 2016 | 63 | RCT | 8 | Any malignancy | Improved atta (nutritional supplement) + nutritional counselling(nutritional) | Nutritional counselling | Body weightjcsm13478-note-0005 Mid‐arm circumferenceVarious skinfold thickness |
| Mehrzad et al.ref. jcsm13478-bib-0061 | 2016 | 70 | RCT | 8 | Any malignancy (not brain) | Pentoxifylline(pharmacological) | Placebo | Body weightjcsm13478-note-0005 Mid‐arm circumference |
| Stewart Coats et al.ref. jcsm13478-bib-0062 | 2016 | 87 | Phase II RCT | 10 | NSCLC/colorectal | Espindolol 10 mg or espindolol 2.5 mg(pharmacological) | Placebo | Body weightjcsm13478-note-0005,jcsm13478-note-0006(primary)Lean body mass—DEXAFat mass—DEXA |
| Takayama et al.ref. jcsm13478-bib-0063 | 2016 | 181 | Phase II RCT | 8 | NSCLC | Anamorelin 100 mg or anamorelin 50 mg(pharmacological) | Placebo | Lean body mass—DEXA(primary)Lean body mass—BIAFat mass—DEXAFat mass—BIABody weightjcsm13478-note-0005 |
| Temel et al.ref. jcsm13478-bib-0011 | 2016 | 979 | Phase III RCT | 8 | NSCLC | Anamorelin(pharmacological) | Placebo | Lean body mass—DEXA(primary)Body weightjcsm13478-note-0005Total body mass—DEXAFat mass—DEXAAppendicular LBM—DEXA |
| Woo et al.ref. jcsm13478-bib-0064 | 2016 | 67 | Phase II RCT | 9 | Pancreatic | Pancreatic exocrine replacement therapy(nutritional) | Placebo | Body weightjcsm13478-note-0005 , jcsm13478-note-0006 (primary) |
| Currow et al.ref. jcsm13478-bib-0065 | 2017 | 513 | Phase III RCT | 8 | NSCLC | Anamorelin(pharmacological) | Placebo | Body weightjcsm13478-note-0005 |
| Jatoi et al.ref. jcsm13478-bib-0066 | 2017 | 302 | RCT | 8 | Any malignancy (not primary brain tumour) | Creatine monohydrate(nutritional) | Placebo | Body weightjcsm13478-note-0006 (primary) |
| Leedo et al.ref. jcsm13478-bib-0067 | 2017 | 40 | RCT | 8 | Lung | Home meal delivery(nutritional) | Standard care | Body weightjcsm13478-note-0005 |
| Sandmael et al.ref. jcsm13478-bib-0068 | 2017 | 41 | Pilot RCT | 9 | Head and neck | Exercise and nutrition intervention during radiotherapy treatment(multi‐modal) | Exercise and nutrition intervention after radiotherapy treatment | Skeletal muscle index—CTBody weightjcsm13478-note-0005 |
| Solheim et al.ref. jcsm13478-bib-0069 | 2017 | 46 | Phase II RCT | 8 | NSCLC/pancreatic | Exercise, celecoxib + nutritional supplements(multi‐modal) | Standard care | Body weightjcsm13478-note-0005,jcsm13478-note-0006Skeletal muscle area—CT |
| Werner et al.ref. jcsm13478-bib-0070 | 2017 | 60 | RCT | 7 | Pancreatic | Fish oil(nutritional) | Marine phospholipids | Body weightjcsm13478-note-0006 BMI |
| Ziętarska et al.ref. jcsm13478-bib-0071 | 2017 | 95 | RCT | 6 | Colorectal | Nutritional supplements(nutritional) | Standard care | BMI |
| Golan et al.ref. jcsm13478-bib-0072 | 2018 | 125 | Phase II RCT | 7 | Pancreatic | Anti‐myostatin antibody 300 mg or anti‐myostatin antibody 100 mg(pharmacological) | Placebo | Thigh muscle volume—CTSkeletal muscle area—CTAdipose tissue area—CTLean body mass—DEXAFat mass—DEXA |
| Katakami et al.ref. jcsm13478-bib-0073 | 2018 | 174 | Phase III RCT | 8 | NSCLC | Anamorelin(pharmacological) | Placebo | Lean body mass—DEXA(primary)Body weightjcsm13478-note-0005 |
| Kouchaki et al.ref. jcsm13478-bib-0074 | 2018 | 90 | Phase III RCT | 8 | Gastrointestinal | Megestrol acetate + celecoxib(pharmacological) | Megestrol acetate + placebo | Body weightjcsm13478-note-0005 , jcsm13478-note-0006 (primary) |
| Schink et al.ref. jcsm13478-bib-0075 | 2018 | 131 | Pilot non‐randomized trial | 9 | Any solid tumour | Whole‐body electromyostimulation + nutritional counselling(multi‐modal) | Nutritional counselling | Lean body mass—BIA(primary)Fat mass—BIABody weightjcsm13478-note-0005 |
| Uster et al.ref. jcsm13478-bib-0076 | 2018 | 58 | RCT | 9 | Gastrointestinal/lung | Exercise programme + nutritional counselling(multi‐modal) | Standard care | Body weightjcsm13478-note-0005 |
| Xie et al.ref. jcsm13478-bib-0077 | 2018 | 54 | RCT | 8 | Lung | Thalidomide + cinobufagin(pharmacological) | Cinobufagin | Body weightjcsm13478-note-0005 Mid‐arm circumference |
| Akita et al.ref. jcsm13478-bib-0078 | 2019 | 62 | RCT | 8 | Pancreatic | EPA‐enriched nutritional supplement(nutritional) | Standard care | Lean body mass—BIAFat mass—BIAPsoas muscle area—CTBMI |
| Britton et al.ref. jcsm13478-bib-0079 | 2019 | 307 | RCT | 7 | Head and neck | Psychological nutritional intervention(nutritional) | Standard care | Body weightjcsm13478-note-0006 |
| Cereda et al.ref. jcsm13478-bib-0080 | 2019 | 166 | RCT | 8 | Other mixed | Whey protein isolate supplement + nutritional counselling(nutritional) | Nutritional counselling | Fat‐free mass index—BIABody weightjcsm13478-note-0005 |
| Laviano et al.ref. jcsm13478-bib-0081 | 2019 | 55 | Pilot RCT | 8 | NSCLC | Targeted medical nutrition supplement(nutritional) | Isocaloric comparator drink | Skeletal muscle area—CTVisceral fat area—CTAppendicular LBM—DEXAFat mass—DEXABody weightjcsm13478-note-0005 |
| Obling et al.ref. jcsm13478-bib-0082 | 2019 | 47 | RCT | 7 | Gastrointestinal | Supplemental home parenteral nutrition and nutritional counselling(nutritional) | Nutritional counselling | Fat‐free mass—BIA(primary)Fat‐free mass index—BIA(primary) |
| Stuecher et al.ref. jcsm13478-bib-0083 | 2019 | 44 | RCT | 8 | Gastrointestinal | Walking exercise programme(exercise) | Standard care | Lean body mass—BIA |
| Wiskemann et al.ref. jcsm13478-bib-0084 | 2019 | 65 | RCT | 5 | Pancreatic | Supervised resistance training or home‐based resistance training(exercise) | Standard care | Body weightjcsm13478-note-0006 (primary) |
| Boulec et al.ref. jcsm13478-bib-0085 | 2020 | 111 | RCT | 7 | Any malignancy | Parenteral nutrition(nutritional) | Oral feeding | Body weightjcsm13478-note-0005 |
| Huang et al.ref. jcsm13478-bib-0086 | 2020 | 119 | RCT | 7 | Nasopharyngeal | Nutritional supplements(nutritional) | Standard care | Body weightjcsm13478-note-0005 |
| Kamel et al.ref. jcsm13478-bib-0087 | 2020 | 40 | RCT | 7 | Pancreatic | Resistance training(exercise) | Standard care | Appendicular LBM—DEXAFat mass—DEXA |
| Movahed et al.ref. jcsm13478-bib-0088 | 2020 | 100 | RCT | 8 | Oesophageal | Supplements ± enteral or parenteral nutrition ± pharmacotherapy + nutritional counselling(multi‐modal) | Nutritional counselling | Fat mass—BIAFat‐free mass index—BIABody weightjcsm13478-note-0005 BMI |
| Qiu et al.ref. jcsm13478-bib-0089 | 2020 | 96 | RCT | 6 | Oesophageal | Nutritional counselling(nutritional) | Standard care | BMI |
| Storck et al.ref. jcsm13478-bib-0090 | 2020 | 52 | RCT | 10 | Other mixed | Protein supplement + nutritional counselling + exercise programme(multi‐modal) | Standard care | Lean body mass—BIAFat mass—BIABMI |
| Currow et al.ref. jcsm13478-bib-0091 | 2021 | 190 | Phase III RCT | 6 | Any malignancy | Megestrol acetate or dexamethasone(pharmacological) | Placebo | Body weightjcsm13478-note-0005 |
| Hunter et al.ref. jcsm13478-bib-0092 | 2021 | 120 | Phase III RCT | 7 | Any solid tumour | Mirtazapine(pharmacological) | Placebo | Lean body mass—BIABody weightjcsm13478-note-0005 |
| Kutz et al.ref. jcsm13478-bib-0093 | 2021 | 58 | RCT | 7 | Head and neck | Nutritional counselling(nutritional) | Standard care | BMIFat‐free mass—BIA |
| Tobberup et al.ref. jcsm13478-bib-0094 | 2021 | 120 | Non‐randomized trial | 9 | NSCLC | Fish oil + nutritional counselling + exercise programme(multi‐modal) | Standard care(historical comparator) | Skeletal muscle area—CTBody weightjcsm13478-note-0005 |
Note: Sample sizes are reported as per ‘intention to treat’. Abbreviations: BIA, bioimpedance analysis; BMI, body mass index; CT, computed tomography; DEXA, dual‐energy X‐ray absorptiometry; EPA, eicosapentaenoic acid; LBM, lean body mass; NSCLC, non‐small‐cell lung cancer; PEG, percutaneous endoscopic gastrostomy; RCT, randomized controlled trial.
Endpoints that are bold underlined had a statistically significant difference between groups.
Endpoint expressed as change in absolute value from baseline.
Endpoint expressed as percentage change from baseline.
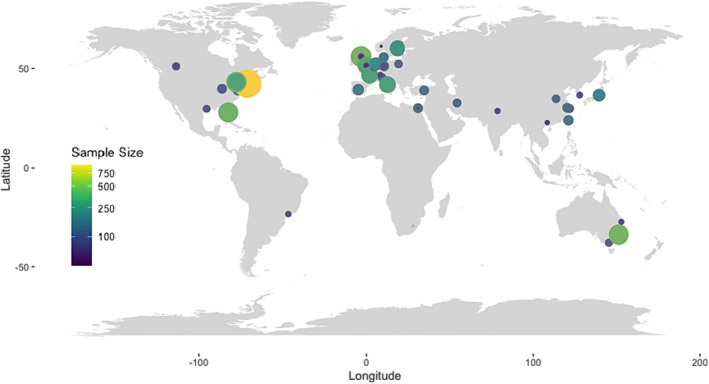
Figurejcsm13478-fig-0002 depicts the geographical distribution of the included cancer cachexia clinical trials. For multicentre or multinational trials (n = 12 studies), coordinates for the institution of the corresponding author were used. It was noted that limited research has been conducted in Eastern Europe, Africa or South America.
Temporal trends in body composition endpoint selection
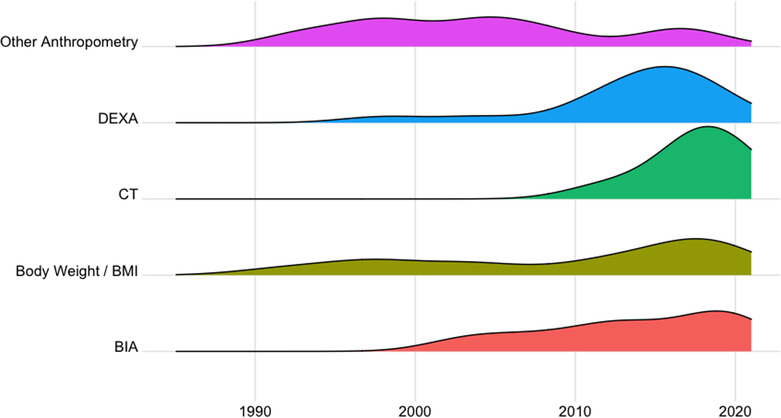
The relative use of body weight and body composition assessments over time is depicted in Figure jcsm13478-fig-0003. The proportion of trials that included assessments of body weight or body mass index (BMI) as an endpoint measure did not vary particularly over the time frame considered. Other anthropometric measures (e.g., skinfold thickness or arm circumference) have been less utilized in recent years with only three trials in the last decade reporting these endpoints. BIA has been used with relative consistency during the last 20 years, whereas DEXA‐based estimates of body composition were included in only two trials before 2010, when its use increased. The reporting of CT body composition analysis in clinical trials is more contemporary with only eight trials, all conducted within the last decade, having included this assessment modality.
Body weight and other anthropometric endpoints
Seventy‐five trials (n = 12 056 participants) measured body weight or other anthropometric endpoints pertaining to body composition (Table jcsm13478-tbl-0001 and Appendix B). Assessments of body weight (68 trials, n = 11 561 participants) were utilized in pharmacological (n = 37), nutritional (n = 22), multi‐modal (n = 8) and exercise‐based (n = 1) clinical trials. A body weight assessment was selected as the (co‐)primary endpoint in 32 (47.1%) of these trials and was a secondary/exploratory outcome for the other 36 (52.9%). Analyses were based on absolute change in body weight in 48 trials (70.6%) and percentage change from baseline in 16 trials (23.5%) with 4 trials considering both (5.9%). The ACT‐ONE trial analysed the rate (slope) of absolute and percentage weight change.ref. jcsm13478-bib-0062 Body weight was handled as an ordinal variable by three trialsref. jcsm13478-bib-0019, ref. jcsm13478-bib-0032, ref. jcsm13478-bib-0054 where comparison was drawn between proportions of weight‐gaining, weight‐stable and weight‐losing participants. Over one third of the trials that considered body weight (38.2%, n = 4735) noted significant differences between trial groups (Figure jcsm13478-fig-0004 and Table jcsm13478-tbl-0002). BMI was reported as an endpoint for 13 trials, most commonly in addition to body weight (6/13 trials). The majority of studies that chose BMI as an endpoint employed a nutritional intervention (61.5%). It was the primary endpoint in only one of these trials (7.7%). Fourteen trials included anthropometric measures of the arm as endpoints (n = 1901 participants). Eight of these evaluated pharmacological interventions (57.1%), five were nutritional (35.7%) and one was multi‐modal (7.1%). Measurements of mid‐arm circumference, and other derived upper arm measures such as muscle and fat areas, were selected as endpoints in 12 trials (n = 1324, Appendix B). Skinfold thickness was also used commonly as an endpoint (11 trials, n = 1727 participants). All of these 11 trials measured the triceps skinfold, and some also considered skinfold thickness at the biceps, subscapular skinfold and other sites. Triceps skinfold thickness was the primary endpoint for one trial, where arm anthropometry was used as a secondary/exploratory outcome for all other trials.
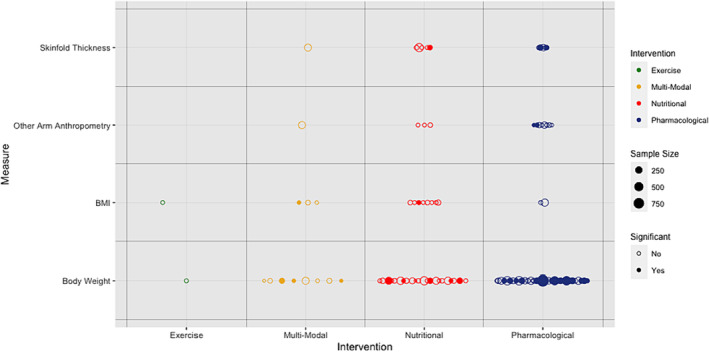
Table 2: Utilization of body composition endpoints
| Endpoint | No. of studies | Years of publication | Total sample size | Intervention type | Statistically significant results (between trial groups) | Intervention type |
|---|---|---|---|---|---|---|
| Anthropometric measures | ||||||
| Body weight | 68 | 1990–2021 | 11 561 | Pharmacological: 37Nutritional: 22Exercise: 1Multi‐modal: 8 | Yes: 26No: 42 | Pharmacological: 18Nutritional: 5Exercise/lifestyle:Multi‐modal: 3 |
| Skinfold thickness | 11 | 1993–2016 | 1727 | Pharmacological: 5Nutritional: 5Multi‐modal: 1 | Yes: 3No: 8 | Pharmacological: 2Nutritional: 1 |
| Other arm anthropometry | 12 | 1993–2018 | 1321 | Pharmacological: 8Nutritional: 3Multi‐modal: 1 | Yes: 2No: 10 | Pharmacological: 2 |
| Body mass index (BMI) | 13 | 2005–2021 | 917 | Pharmacological: 1Nutritional: 8Exercise: 1Multi‐modal: 3 | Yes: 2No: 11 | Nutritional: 1Multi‐modal: 1 |
| BIA body composition | ||||||
| Lean body mass | 16 | 2003–2021 | 2576 | Pharmacological: 8Nutritional: 5Exercise: 1Multi‐modal: 2 | Yes: 2No: 14 | Exercise: 1Multi‐modal: 1 |
| Fat mass | 8 | 2012–2020 | 744 | Pharmacological: 1Nutritional: 4Multi‐modal: 3 | Yes: 2No: 5 | Pharmacological: 1Nutritional: 1 |
| Fat‐free mass | 6 | 2004–2021 | 384 | Pharmacological: 1Nutritional: 5 | Yes: 1No: 5 | Nutritional: 1 |
| Fat‐free mass index | 3 | 2019–2020 | 313 | Nutritional: 2Multi‐modal: 1 | Yes: 2No: 1 | Nutritional: 2 |
| DEXA body composition | ||||||
| Lean body mass | 14 | 1998–2018 | 2957 | Pharmacological: 11Nutritional: 1Exercise: 1Multi‐modal: 1 | Yes: 8No: 6 | Pharmacological: 8 |
| Fat mass | 11 | 2004–2020 | 2162 | Pharmacological: 6Nutritional: 2Exercise: 2Multi‐modal: 1 | Yes: 2No: 9 | Pharmacological: 2 |
| Appendicular lean body mass | 4 | 2015–2020 | 1156 | Pharmacological: 2Nutritional: 1Exercise: 1 | Yes: 3No: 1 | Pharmacological: 2Exercise/lifestyle: 1 |
| Total body mass | 2 | 2015–2016 | 1061 | Pharmacological: 2 | Yes: 2 | Pharmacological: 2 |
| CT body composition | ||||||
| Estimated lean body mass (L3) | 2 | 2010–2012 | 392 | Pharmacological: 2 | Yes: 1No: 1 | Pharmacological: 1 |
| Skeletal muscle area (L3/L4/L5) | 4 | 2017–2021 | 346 | Pharmacological: 1Nutritional: 1Multi‐modal: 2 | No: 4 | N/A |
| Thigh muscle volume | 1 | 2018 | 125 | Pharmacological: 1 | No: 1 | N/A |
| Fat area (L4/L5) | 1 | 2018 | 125 | Pharmacological: 1 | No: 1 | N/A |
| Skeletal muscle index | 1 | 2017 | 41 | Multi‐modal: 1 | No: 2 | N/A |
| Psoas muscle area (L3) | 1 | 2019 | 62 | Nutritional: 1 | No: 1 | N/A |
| Visceral fat area | 1 | 2019 | 55 | Nutritional: 1 | No: 1 | N/A |
Note: Sample sizes are reported as per ‘intention to treat’. Abbreviations: BIA, bioimpedance analysis; BMI, body mass index; CT, computed tomography; DEXA, dual‐energy X‐ray absorptiometry; L3/L4/L5, third/fourth/fifth lumbar vertebral level.
All studies that included arm‐based anthropometric measurements also included body weight as an endpoint. Of these, 9 (64.3%) identified no statistically significant difference between groups using any selected outcome measure. Two trialsref. jcsm13478-bib-0017, ref. jcsm13478-bib-0042 identified statistically significant improvements in triceps skinfold thickness but no corresponding change in body weight. Conversely, McMillan et al.’s trial led to increased body weight (5.1 kg median difference between trial arms) and mid‐arm circumference measurement (1 cm median difference between trial arms), but no change in skinfold thickness measurements.ref. jcsm13478-bib-0028
Bioelectrical body composition endpoints
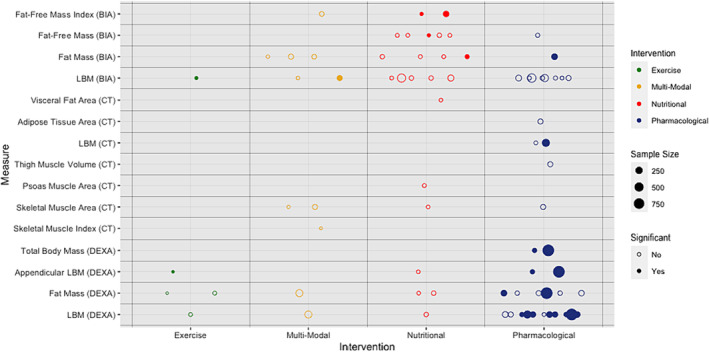
Endpoints based on assessment with BIA were used in 23 trials (n = 3140 participants, Table jcsm13478-tbl-0001 and Appendix C). The interventions tested were commonly nutritional (11 trials, 47.8%) or pharmacological (9 trials, 39.1%). BIA‐based endpoints were selected as a (co‐)primary endpoint in 6 trials (26.1%) and a secondary/exploratory endpoint in the remaining 17 (73.9%). The most frequently included endpoint was estimated whole‐body lean body mass (LBM) (16 trials, n = 2576 participants), with fat‐free mass (FFM) or fat‐free mass index (FFMI) calculated as alternative endpoints by 8 trials (n = 650 participants). Fat mass (FM) was estimated using BIA in eight trials (n = 744 participants).
Fourteen studies (60.9%) that used BIA did not detect statistically significant differences between trial groups with any of their selected endpoints (including those not BIA‐based) (Table jcsm13478-tbl-0002 and Figure jcsm13478-fig-0005). In two trials, improvements were identified in BIA estimates of LBM/FFM that were congruent with increased body weight.ref. jcsm13478-bib-0075, ref. jcsm13478-bib-0080 A statistically significant increase in body weight was identified in two trials of nutritional counsellingref. jcsm13478-bib-0035, ref. jcsm13478-bib-0054 but there were no accompanying changes in BIA estimates of LBM. Two of the seven trials (28.6%) that estimated FM using BIA demonstrated a statistically significant increase with their intervention that was congruent with body weight gain.ref. jcsm13478-bib-0046, ref. jcsm13478-bib-0063
Radiological body composition endpoints
DEXA was used in 16 trials (n = 3052) with LBM (n = 2957 participants) and FM (n = 2162) being the most frequently reported endpoints (Table jcsm13478-tbl-0001 and Appendix C). Appendicular lean mass was used as an alternative endpoint in two trialsref. jcsm13478-bib-0081, ref. jcsm13478-bib-0087 and alongside whole‐body LBM in another two trialsref. jcsm13478-bib-0058, ref. jcsm13478-bib-0065 (Figure jcsm13478-fig-0006). Pharmacological interventions were used for most trials that used DEXA (68.8%). DEXA‐based measures were used as the primary endpoint for 50% (n = 8) of these trials.
Five trials included in this review compared the effects of anamorelin against placebo (n = 1929 participants). Of these, four evaluated LBM using DEXA, and all identified a statistically significant increase compared to placebo with congruent increases in overall body weight.ref. jcsm13478-bib-0011, ref. jcsm13478-bib-0058, ref. jcsm13478-bib-0063, ref. jcsm13478-bib-0073 Of note, Takayama et al.’s relatively large (n = 181) placebo‐controlled trial identified significant improvements in body weight alongside increased FM using both DEXA and BIA, but only a significant improvement in LBM when measured with DEXA (mean difference vs. placebo: 1.15 kg [95% confidence interval—CI: 0.11–2.18]), not with BIA‐based estimates (mean difference vs. placebo: 0.78 kg [95% CI: −0.35 to 1.90]).ref. jcsm13478-bib-0063
Only eight trials included endpoints based on CT body composition (n = 841, Table jcsm13478-tbl-0001 and Appendix C), with two of these (25%) considering it a (co‐)primary endpoint. All but oneref. jcsm13478-bib-0043 of these had relatively small sample sizes (≤125 patients). Four measured the cross‐sectional area of skeletal muscle (n = 346) at the third (L3)ref. jcsm13478-bib-0068, ref. jcsm13478-bib-0069, ref. jcsm13478-bib-0094 or fourth/fifth lumbar vertebral level.ref. jcsm13478-bib-0072 One of these studies reported the L3 cross‐sectional area of muscle as normalized for height, termed skeletal muscle index (SMI).ref. jcsm13478-bib-0068 Others included the L3 cross‐sectional area of psoas majorref. jcsm13478-bib-0078 or derived estimates of LBM (kg) based on L3 muscularity.ref. jcsm13478-bib-0043, ref. jcsm13478-bib-0048 CT estimates of adipose tissue were also reported by two trials.ref. jcsm13478-bib-0072, ref. jcsm13478-bib-0081 Six of the included trials noted no significant differences for any of their selected endpoints.
The five‐arm phase III randomized controlled trial (RCT) (n = 332) by Mantovani et al.ref. jcsm13478-bib-0043 identified improved LBM using DEXA (mean difference: 2.1 kg) and CT estimates (mean difference: 2.6 kg) in one of the trial arms but detected no difference using BIA (mean difference: 1.2 kg, P = 0.609). Similarly, Madeddu et al.ref. jcsm13478-bib-0048 found improvements in LBM across both trial arms based on DEXA and CT estimates, but not with BIA.
Discussion
This systematic review summarizes the frequency and diversity of endpoints examining body weight and composition in cancer cachexia clinical trials. It is one of six systematic reviews being undertaken, with others considering physical function,ref. jcsm13478-bib-0009 quality of life, appetite and dietary intake, biomarkers and oncology/survival endpoints. Assessments of body weight were the most commonly reported endpoint, used by over 80% of the included trials. Other anthropometric measures, such as skinfold thickness measurements and arm circumference, were less frequently used, especially in more contemporary trials. BIA‐based estimates were often included but have been largely superseded by DEXA, especially in larger trials, and more recently by CT body composition analyses.
Body weight is the simplest and most widely available assessment that can indicate alterations to body composition and has long been regarded as a central tenet of cachexia. Included trials have used it to create various endpoints (e.g., absolute/percentage differences from baseline or comparison between weight‐stable, weight‐gaining and weight‐losing groups). Further study is likely required to establish consensus on which of these specific body weight endpoints is most informative. While BMI is likely a helpful baseline descriptor, given the relevance of obesity as a prognostic variable in patients with cachexia and/or sarcopenia,ref. jcsm13478-bib-0095, ref. jcsm13478-bib-0096, ref. jcsm13478-bib-0097 the use of BMI as an endpoint instead of, or alongside, body weight does not add value. Other simple anthropometric measures, such as triceps skinfold thickness or other arm anthropometry, were featured in earlier cachexia trials but have been used less in recent years. All of these methods pose practical advantages, such as low cost, widespread availability and ease of measurement, but provide limited information when compared with radiological methods.
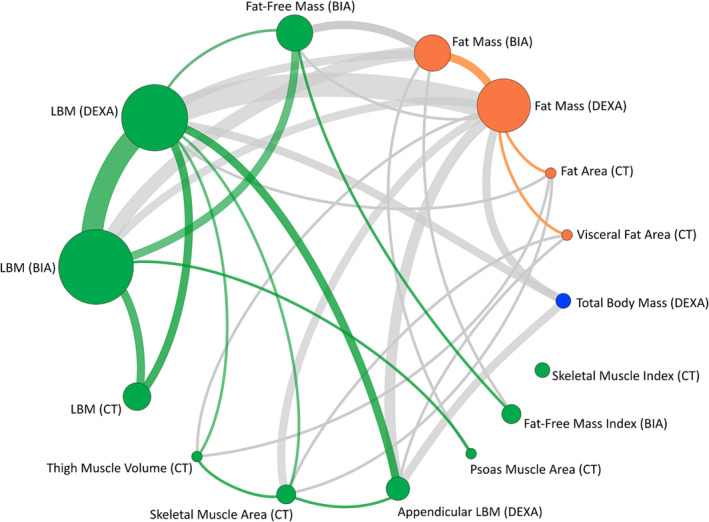
Estimates of body composition using BIA have been featured as chosen endpoints for a number of the included trials. BIA measures the electrical properties of tissues (resistance and reactance), and these values can be used in equations to approximate FM/FFM/LBM. Previous studies have shown that BIA is prone to overestimation for lean mass and underestimation of FMref. jcsm13478-bib-0098 and has poor agreement with comparable DEXAref. jcsm13478-bib-0099 and CT‐based measures.ref. jcsm13478-bib-0100 Indeed, some of the included studies identified significant differences between trial groups on DEXA and/or CT estimates of body composition that were not evident using BIA‐based estimates.ref. jcsm13478-bib-0043, ref. jcsm13478-bib-0048, ref. jcsm13478-bib-0063 However, it must be noted that BIA does have several practical advantages. It uses equipment that is portable and non‐invasive, making it perhaps a more attractive option for assessments in frail, incurable cachexia populations or in community‐based studies where assessments may take place in non‐clinical locations. As such, BIA may remain an appropriate option for particular trial settings despite its limitations.
DEXA has been widely used for assessment of lean and adipose tissue in cachexia trials to date, including amongst many of the larger studies included in this review.ref. jcsm13478-bib-0011, ref. jcsm13478-bib-0036, ref. jcsm13478-bib-0043 It does not routinely feature during the clinical staging pathway for patients with cancer but is an attractive research imaging adjunct owing to its modest cost, low radiation dose and short imaging time. While DEXA estimates of LBM have shown excellent correlation with both MRI and CT,ref. jcsm13478-bib-0101 there is some evidence that measures of adipose tissue using DEXA can tend towards underestimation.ref. jcsm13478-bib-0102 It also lacks the specificity to assess changes in individual muscle groups and is unable to identify myosteatosis, which appears to be an important prognostic feature on CTref. jcsm13478-bib-0103 and MRI body composition analysesref. jcsm13478-bib-0006 via observational studies.
CT body composition analysis was used in eight contemporary cancer cachexia trials (2010 onwards). Within the broader literature, a large number of observational studies have evaluated the impact of low muscle quantityref. jcsm13478-bib-0104 or radiodensityref. jcsm13478-bib-0103 as per CT body composition analysis, with consistently adverse prognostication noted for patients with cancer. While only one of the included trials identified significant differences between groups using CT‐based endpoints, it must be noted that similar negative findings were seen on other concurrent endpoints (including body weight, DEXA and BIA) in all but one study.ref. jcsm13478-bib-0069 This is therefore likely reflective of the efficacy of the interventions used, rather than the sensitivity of the endpoint measured. Furthermore, some of the trials that included CT‐based assessments used muscle groups or vertebral levels that have not yet been adequately validated to the same standard as the use of L3 cross‐sectional area.ref. jcsm13478-bib-0003 Other assessments, such as radiodensity or volumetric body composition analysis, remain unexplored in the cachexia trial setting. CT is a very pragmatic imaging modality for some patient groups, owing to its routine use in the clinical staging and follow‐up of cancer patients. However, when research imaging requirements are in excess of clinical need, the issue of associated ionizing radiation must be duly considered. Furthermore, trial timepoints may not align with the clinical pathway. Balancing this alongside finite resources and the need to limit patient burden can be challenging. Despite the dose reductions achieved with improved technology and focused scanning,ref. jcsm13478-bib-0105 the necessary radiation exposure from CT still often exceeds that of DEXA.ref. jcsm13478-bib-0106 As such, when clinical imaging is not available, other assessment modalities such as MRI may be more appropriate.
Non‐significant findings may result from an intervention that lacks efficacy, a trial that is inadequately powered relative to the chosen outcome or a measurement that lacks the precision to detect a true effect. With this in mind, limited inferences may be drawn regarding how a trial’s significant or non‐significant results may reflect on the selected endpoint(s), particularly if these are secondary endpoints that were not featured during sample size calculations. When comparing methods of body composition assessment, it is useful to consider several key measurement characteristics: reliability, validity, responsiveness to change, minimally important clinical difference and sensibility.
Reliability refers to the consistency and repeatability of a measurement when applied under similar conditions. One could consider intra‐rater reliability (agreement of measures by a single evaluator on different occasions) or inter‐rater reliability (agreement between different evaluators). Body weight would be expected to have excellent inter‐rater reliability, owing to ease of measurement. However, achieving intra‐rater reliability is dependent on consistent patient factors (e.g., clothing or fasting and hydration status) and use of the calibrated instruments. While good intra‐rater and inter‐rater reliability has been demonstrated for BIA, this also requires adherence to strict standardization of measurement conditions,ref. jcsm13478-bib-0107 which may be difficult to achieve in real‐world settings. DEXA and CT assessments of body composition may previously have been subject to lower levels of reliability, owing to their need for manual segmentation of anatomical features or regions of interest, but technological and software advancements have led to more reliable measurements now being obtained.ref. jcsm13478-bib-0108 Furthermore, CT performs well during tests of precision; the ability of a measurement technique to reproduce results when performed in an identical manner.ref. jcsm13478-bib-0109
Validity refers to a method’s accuracy in assessing what it is intended to measure. In the case of cachexia, researchers are likely to be interested in changes in quantities of muscle and/or fat. This may present an obvious limitation with body weight, as it cannot inform us regarding the alterations to body composition that have led to any change in weight. This limits its use in isolation. Furthermore, the potential for fluid accumulations (e.g., ascites/peripheral oedema/hydration status) to influence body weight, BIA or even DEXA could lead to these modalities providing less valid assessments.ref. jcsm13478-bib-0110 While most studies of CT body composition extrapolate single‐slice measurements to estimate whole‐body composition, it is not known whether wasting occurs uniformly throughout the body.ref. jcsm13478-bib-0109 Analyses conducted over larger regions of interest may yet improve the validity of CT body composition.ref. jcsm13478-bib-0111
Responsiveness to change is a measure’s ability to detect meaningful differences over time and is crucial for monitoring responses to trial interventions. Multiple factors can influence these parameters, and in the setting of cachexia clinical trials, it can be challenging to assess efficacy independent of confounders. While single axial slice CT (e.g., L3) and whole‐body measurements are known to be highly correlated,ref. jcsm13478-bib-0003 this may not hold true when assessing changes over time.ref. jcsm13478-bib-0109
A minimal clinically important difference (MCID) describes the smallest change that could be considered clinically significant. Such a metric may be determined with consideration of how changes in body composition endpoints relate to other outcomes. For example, what change in muscle mass is required to influence quality of life or survival? As has been observed with muscle mass and function,ref. jcsm13478-bib-0112 the relationships between endpoints may be non‐linear, and this must be acknowledged when considering responsiveness to change. The minimum body weight change that is considered clinically important for an individual’s health is more commonly studied in the field of obesity than in cachexia. Semaglutide (glucagon‐like peptide‐1 [GLP‐1] receptor agonist) was granted Food and Drug Administration (FDA) approval based on >5% weight loss being regarded as clinically meaningful.ref. jcsm13478-bib-0113 A >5% weight gain may be considered an equivalent MCID for treatment of cachexia, yet such precedent has not been set thus far.
Measurements should have sensibility (or interpretability) so they can be understood with ease. Body weight is meaningful and can be easily interpreted by clinicians, researchers and even patients. Conversely, bioelectrical estimates of body composition are less well known, and as such, the sensibility of these is limited. With improved consensus regarding effective assessment methods and endpoints, it should be anticipated that relevant stakeholders will become more informed regarding their chosen measurement techniques and how their findings relate to patients.
It is evident that no single assessment method currently fulfils all requirements. Rather, researchers should choose appropriate endpoints to align with their study aims and the cohort in question. What represents the ‘gold‐standard’ measure would be dependent on the choice of intervention and its underlying mechanism. Assessment of body weight alongside dietary intake may be reasonable when assessing an appetite stimulant and has the added advantage of having regulatory approval in the obesity arena. Similarly, assessing DEXA or CT‐based body composition would be sensible when trialling an exercise intervention aimed to improve lean mass. The practicalities, including cost and participant burden, mean that assessing these in clinical trials may be aspirational, despite a clear need.
A large volume of data has been compiled through each of the six reviews undertaken within this series. While there was a need to give a detailed appraisal within each of these, further work is ongoing to examine the relationships between these parameters. The findings presented are likely to have even greater value when considered in the context of other endpoints. For example, how do improvements in lean mass relate to physical function? The group aspires towards achieving a wider consensus alongside the identification and prioritization of areas for future research. Key strengths of this review include the broad search criteria and the robust methodological approach and appraisal process. However, the eligibility criteria could be considered a limitation, as balance was sought between the need to find trials of sufficient quality against having to appraise an impractical number of manuscripts. Although a specific time period was defined for the purposes of this work, it is accepted that trials published before 1990 may have yielded additional data, though this would pre‐date the use of most endpoints considered by this review. The sample size cut‐off was felt to be appropriate, as trials with <40 participants were expected to be insufficiently powered to assess changes in the endpoints being assessed. Furthermore, the minimum intervention time of 14 days was selected as interventions conducted for a shorter duration were felt unlikely to influence the disease course of cachexia. It should be acknowledged, however, that these restrictions may have precluded the inclusion of some informative trials. While the focus of this review was prospective, interventional trials, it is also acknowledged that many high‐quality studies of other designs may yet inform the process of establishing a consensus regarding the optimum endpoints for cachexia trials, and future review of these would also be informative.
Endpoints should be intrinsically linked to inclusion criteria and vice versa. Changes in body composition are a key feature of cancer cachexia and, therefore, by definition, often feature as a baseline descriptive criterion (and, thus, outcome measure) in cachexia intervention trials. The Global Leadership Initiative on Malnutrition (GLIM) consensus criteriaref. jcsm13478-bib-0114 were not designed to be used for the diagnosis of cachexia; however, they are intended to complement the existing cachexia literature, acknowledging that all patients with cachexia would meet their diagnosis of malnutrition. This group agreed that a body composition‐based phenotypical criterion (weight loss, low BMI or reduced muscle mass) and an aetiological criterion (reduced food intake/assimilation or inflammation) are required for a diagnosis of malnutrition. Their recommendation for methods of estimating low muscle mass was for DEXA or ‘corresponding standards using other body composition methods like BIA, CT or MRI’. The group also stated that anthropometric measures, such as arm muscle circumference, may be used as an alternative when radiological imaging is unavailable. Similarly, broad assessment methodology was proposed by the European consensus definition for sarcopenia,ref. jcsm13478-bib-0005 with DEXA, BIA, CT and MRI all listed as options for evaluating ‘muscle quantity or quality’. As shown in this present review, the range of modalities within the guidance is reflective of the heterogeneity within the existing literature.
Conclusions
Based on the findings presented herein, the use of body weight alongside a radiological modality for body composition analysis would seem like suitable endpoints for cancer cachexia trials. Thus far, body weight has been reported in a variety of ways, and further consensus is required regarding the specific body weight endpoint that should be used for future trials. The choice of radiological modality is likely to be dependent on the trial setting, population and intervention in question. When available, CT imaging is a well‐validated and often pragmatic option that provides good levels of detail regarding body composition. Through ongoing exploration of this, and other assessment methods such as MRI,ref. jcsm13478-bib-0110 further evidence is likely to emerge that will help standardize the appraisal of body composition. Endpoint heterogeneity in cancer cachexia clinical trials has greatly contributed to the lack of approved treatments by regulatory authorities.ref. jcsm13478-bib-0115 Moreover, discrepancies between clinicians, regulatory industries and patients’ perspectives regarding the most clinically relevant endpoints in cancer cachexia remain challenging. It is vital that consensus is achieved to ensure reporting consistency and maximize the efficacy of upcoming trials aiming to counteract the devastating effects of cancer cachexia.
Conflict of interest statement
LRB, MSS, MSY, VEB, DCM, AB, TRB, OD, RDD, MTF, CG, MJH, GJ, MM, JM, IOO, IP, JS, MRS, OMV and TSS have none to declare. JA has received lecture fees from Baxter and Danone. RJES has received personal fees for consultancy from Avidity Biosciences, Actimed, Faraday and Helsinn. BJAL has received personal fees for consultancy from Artelo, Actimed, Faraday, Kyowa Kirin and Toray.
| Database | Number of retrieved references for trials | Number of retrieved references for cohort/longitudinal studies |
|---|---|---|
| MEDLINE (Ovid) | 3812 | 1918 |
| Embase (Ovid) | 2033 | 2031 |
| Cochrane Central Register of Controlled Trials | 1923 | |
| Number of references before de‐duplication | 8166 | 3949 |
| Number of references after de‐duplication | 5998 | 3190 |
| # | Searches | Results |
|---|---|---|
| 1 | exp Neoplasms/or (neoplasm* or cancer* ortumor* ortumour* or oncol* or malign* or carcinom* or adenocarcinom* or adenoma or metasta*).ti,ab,kf. | 4 606 655 |
| 2 | Cachexia/ or Emaciation/ or Malnutrition/ or Starvation/ or Wasting syndrome/ or Thinness/ or Sarcopenia/ or Anorexia/or *Weight Loss/ | 63 432 |
| 3 | and/1‐2 | 9650 |
| 4 | ((cachexia or cachexic or anorexia or anorectic or emaciat* or malnutrition or underweight or starvation* or thiness or leanness or sarcopenia or wasting syndrome* or wasting disease* or weightloss* or ((appetite* or weight) adj2 (loss or loosing or losing))) adj4 (neoplasm* or cancer* ortumor* ortumour* or oncol* or malign* or carcinom* or adenocarcinom* or adenoma or metasta*)).ti,ab,kf. | 7231 |
| 5 | ((cachexia or cachexic or anorexia or anorectic or emaciat* or malnutrition or underweight or starvation* or thiness or leanness or sarcopenia or wasting syndrome* or wasting disease* or weightloss* or ((appetite* or weight) adj2 (loss or loosing or losing))) and (neoplasm* or cancer* ortumor* ortumour* or oncol* or malign* or carcinom* or adenocarcinom* or adenoma or metasta*)).ti. | 4253 |
| 6 | or/3‐5 | 13 924 |
| 7 | randomized controlled trial.pt. | 536 354 |
| 8 | controlled clinical trial.pt. | 94 265 |
| 9 | randomized.ab. | 525 221 |
| 10 | placebo.ab. | 219 320 |
| 11 | drug therapy.fs. | 2 343 029 |
| 12 | randomly.ab. | 360 557 |
| 13 | trial.ab. | 557 904 |
| 14 | groups.ab. | 2 213 680 |
| 15 | or/7‐14 | 5 047 938 |
| 16 | exp animals/not humans.sh. | 4 855 037 |
| 17 | 15 not 16 | 4 388 865 |
| 18 | 6 and 17 | 4078 |
| 19 | limit 18 to yr = ‘1990‐Current’ | 3812 |
| 20 | cohort studies/ or follow‐up studies/ or longitudinal studies/ or ‘national longitudinal study of adolescent health’/ or prospective studies/or retrospective studies/ | 2 168 707 |
| 21 | (cohort* or longitudinal or prospective* or retrospective*).tw. | 2 098 508 |
| 22 | or/20‐21 | 3 012 965 |
| 23 | and/6,22 | 3215 |
| 24 | limit 23 to yr = ‘1990‐Current’ | 3139 |
| 25 | 24 not 19 | 1918 |
| 26 | 19 or 24 | 5730 |
| # | Searches | Results |
|---|---|---|
| 1 | exp neoplasm/or (neoplasm* or cancer* ortumor* ortumour* or oncol* or malign* or carcinom* or adenocarcinom* or adenoma or metasta*).ti,ab,kw. | 6 400 926 |
| 2 | cachexia/ or emaciation/ or *malnutrition/ or starvation/ or wasting syndrome/ or *anorexia/ or sarcopenia/or *weight loss/ | 93 608 |
| 3 | and/1‐2 | 20 654 |
| 4 | ((cachexia or cachexic or anorexia or anorectic or emaciat* or malnutrition or underweight or starvation* or thiness or leanness or sarcopenia or wasting syndrome* or wasting disease* or weightloss* or ((appetite* or weight) adj2 (loss or loosing or losing))) adj3 (neoplasm* or cancer* ortumor* ortumour* or oncol* or malign* or carcinom* or adenocarcinom* or adenoma or metasta*)).ti,ab,kw. | 9798 |
| 5 | ((cachexia or cachexic or anorexia or anorectic or emaciat* or malnutrition or underweight or starvation* or thiness or leanness or sarcopenia or wasting syndrome* or wasting disease* or weightloss* or ((appetite* or weight) adj2 (loss or loosing or losing))) and (neoplasm* or cancer* ortumor* ortumour* or oncol* or malign* or carcinom* or adenocarcinom* or adenoma or metasta*)).ti. | 6437 |
| 6 | or/3‐5 | 24 964 |
| 7 | Randomized controlled trial/ | 666 248 |
| 8 | Controlled clinical trial/ | 463 928 |
| 9 | random$.ti,ab. | 1 691 555 |
| 10 | randomization/ | 91 413 |
| 11 | intermethod comparison/ | 272 763 |
| 12 | placebo.ti,ab. | 330 754 |
| 13 | (compare or compared or comparison).ti. | 571 937 |
| 14 | ((evaluated or evaluate or evaluating or assessed or assess) and (compare or compared or comparing or comparison)).ab. | 2 334 019 |
| 15 | (open adj label).ti,ab. | 88 467 |
| 16 | ((double or single or doubly or singly) adj (blind or blinded or blindly)).ti,ab. | 251 401 |
| 17 | double blind procedure/ | 187 924 |
| 18 | parallel group$1.ti,ab. | 27 750 |
| 19 | (crossover or cross over).ti,ab. | 112 863 |
| 20 | ((assign$ or match or matched or allocation) adj5 (alternate or group$1 or intervention$1 or patient$1 or subject$1 or participant$1)).ti,ab. | 359 989 |
| 21 | (assigned or allocated).ti,ab. | 424 479 |
| 22 | (controlled adj7 (study or design or trial)).ti,ab. | 385 967 |
| 23 | (volunteer or volunteers).ti,ab. | 264 717 |
| 24 | human experiment/ | 550 005 |
| 25 | trial.ti. | 340 818 |
| 26 | or/7‐25 | 5 516 795 |
| 27 | (random$ adj sampl$ adj7 (cross section$ or questionnaire$1 or survey$ or database$1)).ti,ab. not (comparative study/ or controlled study/or randomi?ed controlled.ti,ab. or randomly assigned.ti,ab.) | 8736 |
| 28 | Cross‐sectional study/not (randomized controlled trial/ or controlled clinical study/ or controlled study/or randomi?ed controlled.ti,ab. or control group$1.ti,ab.) | 273 554 |
| 29 | (((case adj control$) and random$) not randomi?ed controlled).ti,ab. | 18 641 |
| 30 | (Systematic review not (trial or study)).ti. | 179 350 |
| 31 | (nonrandom$ not random$).ti,ab. | 17 184 |
| 32 | Random field$.ti,ab. | 2525 |
| 33 | (random cluster adj3 sampl$).ti,ab. | 1368 |
| 34 | (review.ab. and review.pt.) not trial.ti. | 902 488 |
| 35 | we searched.ab. and (review.ti. or review.pt.) | 37 332 |
| 36 | update review.ab. | 116 |
| 37 | (databases adj4 searched).ab. | 43 700 |
| 38 | (rat or rats or mouse or mice or swine or porcine or murine or sheep or lambs or pigs or piglets or rabbit or rabbits or cat or cats or dog or dogs or cattle or bovine or monkey or monkeys or trout or marmoset$1).ti. and animal experiment/ | 1 113 420 |
| 39 | Animal experiment/not (human experiment/or human/) | 2 339 504 |
| 40 | or/27‐39 | 3 735 626 |
| 41 | 26 not 40 | 4 907 360 |
| 42 | and/6,41 | 3904 |
| 43 | limit 42 to yr = ‘1990‐Current’ | 3674 |
| 44 | limit 43 to conference abstracts | 1641 |
| 45 | 43 not 44 | 2033 |
| 46 | cohort analysis/ or follow up/ or longitudinal study/ or ‘national longitudinal study of adolescent health’/ or prospective study/or retrospective study/ | 3 499 550 |
| 47 | ((cohort adj (study or studies)) or cohort analy* or longitudinal).tw. | 705 575 |
| 48 | or/46‐47 | 3 722 608 |
| 49 | and/6,48 | 4417 |
| 50 | limit 49 to yr = ‘1990‐Current’ | 4387 |
| 51 | limit 50 to conference abstracts | 1639 |
| 52 | 50 not 51 | 2748 |
| 53 | 52 not 45 | 2031 |
| # | Searches | Results |
|---|---|---|
| 1 | [mh Neoplasms] | 82 548 |
| 2 | ((neoplasm* or cancer* or tumor* or tumour* or oncol* or malign* or carcinom* or adenocarcinom* or adenoma or metasta*)):ti,ab,kw (Word variations have been searched) | 232 559 |
| 3 | #1 or #2 | 241 300 |
| 4 | [mh Cachexia] or [mh ^Emaciation] or [mh ^Malnutrition] or [mh Starvation] or [mh ^‘Wasting syndrome’] or [mh Thinness] or [mh Sarcopenia] or [mh Anorexia] | 2665 |
| 5 | MeSH descriptor: [Weight Loss] this term only | 6360 |
| 6 | #3 and (#4 or #5) | 1017 |
| 7 | (((cachexia or cachexic or anorexia or anorectic or emaciat* or malnutrition or underweight or starvation* or thiness or leanness or sarcopenia or ‘wasting syndrom’ or ‘wasting syndromes’ or ‘wasting disease’ or ‘wasting diseases’ or weightloss* or ((appetite* or weight) near/2 (loss or loosing or losing))) near/3 (neoplasm* or cancer* or tumor* or tumour* or oncol* or malign* or carcinom* or adenocarcinom* or adenoma or metasta*))):ti,ab,kw | 1475 |
| 8 | (((cachexia or cachexic or anorexia or anorectic or emaciat* or malnutrition or underweight or starvation* or thiness or leanness or sarcopenia or ‘wasting syndrome’ or ‘wasting syndromes’ or ‘wasting disease’ or ‘wasting diseases’ or weightloss* or ((appetite* or weight) near/2 (loss or loosing or losing))) and (neoplasm* or cancer* or tumor* or tumour* or oncol* or malign* or carcinom* or adenocarcinom* or adenoma or metasta*))):ti (Word variations have been searched) | 758 |
| 9 | #6 or #7 or #8 with Publication Year from 1990 to 2021, in Trials | 2345 |
| Author (reference) | Year | Sample size | Study design | Study quality | Primary cancer site | Intervention | Comparator | Body composition outcomesjcsm13478-note-0010 |
|---|---|---|---|---|---|---|---|---|
| Body weight | ||||||||
| Kardinal et al.ref. jcsm13478-bib-0012 | 1990 | 293 | RCT | 8 | Any malignancy (not brain) | Cyproheptadine(pharmacological) | Placebo | Body weightjcsm13478-note-0012 (primary) |
| Loprinzi et al.ref. jcsm13478-bib-0013 | 1990 | 133 | RCT | 9 | Any malignancy (not brain/breast/endometrial) | Megestrol acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0011,jcsm13478-note-0012(primary) |
| Feliu et al.ref. jcsm13478-bib-0014 | 1992 | 150 | RCT | 5 | Any malignancy (not hormone dependent) | Megestrol acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0011 (primary) |
| Downer et al.ref. jcsm13478-bib-0015, jcsm13478-note-0009 | 1993 | 60 | RCT | 1 | Any malignancy | Medroxyprogesterone acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0011 (primary)Mid‐arm circumferenceTriceps skinfold thickness |
| Loprinzi et al.ref. jcsm13478-bib-0016 | 1993 | 342 | Phase III RCT | 8 | Any malignancy (not breast/endometrial) | Megestrol acetate 1280 mg or megestrol acetate 800 mg(pharmacological) | Megestrol acetate 480 mg or megestrol acetate 160 mg | Body weightjcsm13478-note-0012 (primary) |
| Ovesen et al.ref. jcsm13478-bib-0017, jcsm13478-note-0009 | 1993 | 105 | RCT | 8 | Small‐cell‐lung/ovarian/breast | Nutritional counselling(nutritional) | Standard care | Body weightjcsm13478-note-0011 (primary)Arm muscle areaTriceps skinfold thickness |
| Goldberg et al.ref. jcsm13478-bib-0018 | 1995 | 70 | RCT | 8 | Any malignancy (not primary brain tumour) | Pentoxifylline(pharmacological) | Placebo | Body weightjcsm13478-note-0012 (primary) |
| Gebbia et al.ref. jcsm13478-bib-0019 | 1996 | 122 | RCT | 6 | Any malignancy (not hormone dependent) | Megestrol acetate 320 mg(pharmacological) | Megestrol acetate 160 mg | Body weight(primary) |
| Lissoni et al.ref. jcsm13478-bib-0020 | 1996 | 100 | RCT | 7 | Any solid tumour | Melatonin(pharmacological) | Standard care | Body weightjcsm13478-note-0011(primary) |
| Simons et al.ref. jcsm13478-bib-0021 | 1996 | 206 | RCT | 7 | Any malignancy (not hormone dependent) | Medroxyprogesterone acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0011(primary) |
| Beller et al.ref. jcsm13478-bib-0022, jcsm13478-note-0009 | 1997 | 240 | RCT | 4 | Any malignancy (not hormone dependent) | Megestrol acetate 480 mg or megestrol acetate 160 mg(pharmacological) | Placebo | Body weightjcsm13478-note-0011 (primary)Mid‐arm circumferenceMid‐arm fat areaMid‐arm muscle areaTriceps skinfold thickness |
| Chen et al.ref. jcsm13478-bib-0023 | 1997 | 129 | RCT | 8 | Head and neck | Megestrol acetate or prepulside(pharmacological) | Placebo | Body weightjcsm13478-note-0011(primary) |
| Daneryd et al.ref. jcsm13478-bib-0024 | 1998 | 180 | RCT | 7 | Any malignancy | Indomethacin + erythropoietin(pharmacological) | Indomethacin | Lean body mass—DEXABody weightjcsm13478-note-0011 |
| De Conno et al.ref. jcsm13478-bib-0025 | 1998 | 42 | RCT | 6 | Any malignancy (not hormone dependent) | Megestrol acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0011(primary) |
| Vadell et al.ref. jcsm13478-bib-0026, jcsm13478-note-0009 | 1998 | 150 | RCT | 5 | Any malignancy | Megestrol acetate 480 mg or megestrol acetate 160 mg(pharmacological) | Placebo | Body weightjcsm13478-note-0011(primary)Mid‐arm circumferenceTriceps skinfold thickness |
| Loprinizi et al.ref. jcsm13478-bib-0027 | 1999 | 496 | RCT | 8 | Any malignancy (not breast/prostate/ovarian/endometrial) | Megestrol acetate or dexamethasone(pharmacological) | Fluoxymesterone | Body weightjcsm13478-note-0012 (primary) |
| McMillan et al.ref. jcsm13478-bib-0028, jcsm13478-note-0009 | 1999 | 73 | RCT | 7 | Gastrointestinal | Megestrol acetate + ibuprofen(pharmacological) | Megestrol acetate + placebo | Body weightjcsm13478-note-0011(primary)Mid‐arm circumferenceTriceps skinfold thicknessBiceps skinfold thickness |
| Westman et al.ref. jcsm13478-bib-0029 | 1999 | 255 | RCT | 7 | Other mixed | Megestrol acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0011 (primary) |
| Jatoi et al.ref. jcsm13478-bib-0030 | 2002 | 469 | RCT | 10 | Any malignancy (not brain/breast/ovarian/endometrial) | Megestrol acetate + dronabinol or megestrol acetate + placebo(pharmacological) | Dronabinol + placebo | Body weightjcsm13478-note-0012(primary) |
| Persson et al.ref. jcsm13478-bib-0031 | 2002 | 144 | RCT | 6 | Breast/colorectal/gastric/prostate | Individual nutritional counselling or individual and group nutritional counselling(nutritional) | Group nutritional counselling or standard care | Body weightjcsm13478-note-0012(primary) |
| Ulutin et al.ref. jcsm13478-bib-0032 | 2002 | 119 | RCT | 9 | NSCLC | Megestrol acetate 320 mg(pharmacological) | Megestrol acetate 160 mg | Body weight(increase vs. stable vs. decrease) |
| Bruera et al.ref. jcsm13478-bib-0033, jcsm13478-note-0009 | 2003 | 91 | RCT | 7 | Any malignancy | Fish oil capsules(nutritional) | Placebo | Lean body mass—BIABody weightjcsm13478-note-0011 Mid‐arm muscle circumferenceTriceps skinfold thicknessSubscapular skinfold thickness |
| Fearon et al.ref. jcsm13478-bib-0034 | 2003 | 200 | RCT | 8 | Pancreatic | n‐3 fatty acid and antioxidant‐enriched supplement(nutritional) | Supplement without n‐3 fatty acid and antioxidants | Lean body mass—BIABody weightjcsm13478-note-0011 |
| Isenring et al.ref. jcsm13478-bib-0035 | 2004 | 60 | RCT | 8 | Gastrointestinal/head and neck | Nutrition counselling and protocol(nutritional) | Standard care | Fat‐free mass—BIABody weightjcsm13478-note-0011 |
| Lundholm et al.ref. jcsm13478-bib-0036, jcsm13478-note-0009 | 2004 | 309 | RCT | 5 | Any solid tumour | Indomethacin + erythropoietin + nutritional support + home total parenteral nutrition(multi‐modal) | Indomethacin + erythropoietin | Fat mass—DEXALean body mass—DEXABody weightjcsm13478-note-0011 Mid‐arm muscle circumferenceTriceps skinfold thickness |
| Gonçalves Dias et al.ref. jcsm13478-bib-0037, jcsm13478-note-0009 | 2005 | 64 | Non‐randomized trial | 1 | Head and neck | Home enteral (nasogastric) feeding or oral diet + nutritional supplements(nutritional) | Oral diet | Body weightjcsm13478-note-0011 BMIMid‐arm circumferenceMid‐arm muscle areaTriceps skinfold thickness |
| Gordon et al.ref. jcsm13478-bib-0038, jcsm13478-note-0009 | 2005 | 50 | RCT | 10 | Pancreatic | Thalidomide(pharmacological) | Placebo | Body weightjcsm13478-note-0011(primary)Bone‐free arm muscle area |
| Fearon et al.ref. jcsm13478-bib-0039 | 2006 | 518 | RCT | 8 | Gastrointestinal/lung | EPA 2 g or EPA 4 g(pharmacological) | Placebo | Lean body mass—BIABody weightjcsm13478-note-0011 |
| Berk et al.ref. jcsm13478-bib-0040, jcsm13478-note-0009 | 2008 | 472 | RCT | 9 | Any solid tumour | Nutritional supplement(nutritional) | Placebo | Lean body mass—BIA(primary)Body weightjcsm13478-note-0012 Various skinfold thickness |
| Beijer et al.ref. jcsm13478-bib-0042, jcsm13478-note-0009 | 2009 | 100 | RCT | 8 | Any malignancy | Adenosine 5′‐triphosphate(pharmacological) | Standard care | Triceps skinfold thickness(primary)Body weightjcsm13478-note-0011 Mid‐arm circumference |
| Navari et al.ref. jcsm13478-bib-0044 | 2010 | 80 | RCT | 7 | Gastrointestinal/lung | Megestrol acetate + olanzapine(pharmacological) | Megestrol acetate | Body weightjcsm13478-note-0012(primary) |
| Baldwin et al.ref. jcsm13478-bib-0045 | 2011 | 358 | RCT | 8 | Gastrointestinal/NSCLC/mesothelioma | Nutritional supplement + nutritional counselling or nutritional supplement(nutritional) | Nutritional counselling or standard care | Body weightjcsm13478-note-0011 |
| Silander et al.ref. jcsm13478-bib-0049, jcsm13478-note-0009 | 2012 | 134 | RCT | 6 | Head and neck | Prophylactic PEG(nutritional) | Standard care | Body weightjcsm13478-note-0012 (primary)BMI |
| Wen et al.ref. jcsm13478-bib-0050 | 2012 | 102 | RCT | 5 | Any malignancy | Megestrol acetate + thalidomide(pharmacological) | Megestrol acetate | Body weightjcsm13478-note-0011(primary) |
| Del Fabbro et al.ref. jcsm13478-bib-0051 | 2013 | 73 | RCT | 10 | Gastrointestinal/lung | Melatonin(pharmacological) | Placebo | Body weightjcsm13478-note-0011 (primary)Lean body mass—BIAFat‐free mass—BIA |
| Dobs et al.ref. jcsm13478-bib-0052 | 2013 | 159 | Phase II RCT | 8 | Other mixed | Enobosarm 1 mg or enobosarm 3 mg(pharmacological) | Placebo | Lean body mass—DEXA(primary)Body weightjcsm13478-note-0011 Fat mass—DEXA |
| Kanat et al.ref. jcsm13478-bib-0053, jcsm13478-note-0009 | 2013 | 69 | RCT | 8 | Any malignancy | Megestrol acetate + meloxicam or megestrol acetate + EPA‐enriched nutritional supplement(pharmacological) | Meloxicam + EPA‐enriched nutritional supplement | Body weightjcsm13478-note-0011 (primary)Lean body mass—BIA(primary)BMI |
| Poulsen et al.ref. jcsm13478-bib-0054 | 2013 | 61 | RCT | 5 | Oesophageal/gastric/gynaecological | Nutritional counselling(nutritional) | Standard care | Body weight(loss vs. maintenance)(primary)Fat mass—BIAFat‐free mass—BIA |
| Bourdel‐Marchasson et al.ref. jcsm13478-bib-0055 | 2014 | 336 | RCT | 10 | Other mixed | Nutritional counselling(nutritional) | Standard care | Body weightjcsm13478-note-0011 |
| Pottel et al.ref. jcsm13478-bib-0056 | 2014 | 85 | Exploratory RCT | 8 | Head and neck | Echium oil(nutritional) | Sunflower oil | Body weightjcsm13478-note-0012 (primary)Lean body mass—DEXALean body mass—BIAFat mass—DEXAFat mass—BIAFat‐free mass—BIA |
| Focan et al.ref. jcsm13478-bib-0057, jcsm13478-note-0009 | 2015 | 53 | RCT | 7 | Any malignancy | Dietetic and psychological mindfulness workshops(multi‐modal) | Standard care | BMI(primary)Body weightjcsm13478-note-0011 |
| Kapoor et al.ref. jcsm13478-bib-0060, jcsm13478-note-0009 | 2016 | 63 | RCT | 8 | Any malignancy | Improved atta (nutritional supplement) + nutritional counselling(nutritional) | Nutritional counselling | Body weightjcsm13478-note-0011 Mid‐arm circumferenceVarious skinfold thickness |
| Mehrzad et al.ref. jcsm13478-bib-0061, jcsm13478-note-0009 | 2016 | 70 | RCT | 8 | Any malignancy (not brain) | Pentoxifylline(pharmacological) | Placebo | Body weightjcsm13478-note-0011 Mid‐arm circumference |
| Stewart Coats et al.ref. jcsm13478-bib-0062 | 2016 | 87 | Phase II RCT | 10 | NSCLC/colorectal | Espindolol 10 mg or espindolol 2.5 mg(pharmacological) | Placebo | Body weightjcsm13478-note-0011,jcsm13478-note-0012(primary)Lean body mass—DEXAFat mass—DEXA |
| Takayama et al.ref. jcsm13478-bib-0063 | 2016 | 181 | Phase II RCT | 8 | NSCLC | Anamorelin 100 mg or anamorelin 50 mg(pharmacological) | Placebo | Lean body mass—DEXA(primary)Lean body mass—BIAFat mass—DEXAFat mass—BIABody weightjcsm13478-note-0011 |
| Temel et al.ref. jcsm13478-bib-0011 | 2016 | 979 | Phase III RCT | 8 | NSCLC | Anamorelin(pharmacological) | Placebo | Lean body mass—DEXA(primary)Body weightjcsm13478-note-0011Total body mass—DEXAFat mass—DEXAAppendicular LBM—DEXA |
| Woo et al.ref. jcsm13478-bib-0064 | 2016 | 67 | Phase II RCT | 9 | Pancreatic | Pancreatic exocrine replacement therapy(nutritional) | Placebo | Body weightjcsm13478-note-0011 , jcsm13478-note-0012 (primary) |
| Currow et al.ref. jcsm13478-bib-0065 | 2017 | 513 | Phase III RCT | 8 | NSCLC | Anamorelin(pharmacological) | Placebo | Body weightjcsm13478-note-0011 |
| Jatoi et al.ref. jcsm13478-bib-0066 | 2017 | 300 | RCT | 8 | Any malignancy (not primary brain tumour) | Creatine monohydrate(nutritional) | Placebo | Body weightjcsm13478-note-0012 (primary) |
| Leedo et al.ref. jcsm13478-bib-0067 | 2017 | 40 | RCT | 8 | Lung | Home meal delivery(nutritional) | Standard care | Body weightjcsm13478-note-0011 |
| Sandmael et al.ref. jcsm13478-bib-0068 | 2017 | 41 | Pilot RCT | 9 | Head and neck | Exercise and nutrition intervention during radiotherapy treatment(multi‐modal) | Exercise and nutrition intervention after radiotherapy treatment | Skeletal muscle index—CTBody weightjcsm13478-note-0011 |
| Solheim et al.ref. jcsm13478-bib-0069 | 2017 | 46 | Phase II RCT | 8 | NSCLC/pancreatic | Exercise, celecoxib + nutritional supplements(multi‐modal) | Standard care | Body weightjcsm13478-note-0011,jcsm13478-note-0012Skeletal muscle area—CT |
| Werner et al.ref. jcsm13478-bib-0070 | 2017 | 60 | RCT | 7 | Pancreatic | Fish oil(nutritional) | Marine phospholipids | Body weightjcsm13478-note-0012 BMI |
| Katakami et al.ref. jcsm13478-bib-0073 | 2018 | 174 | Phase III RCT | 8 | NSCLC | Anamorelin(pharmacological) | Placebo | Lean body mass—DEXA(primary)Body weightjcsm13478-note-0011 |
| Kouchaki et al.ref. jcsm13478-bib-0074 | 2018 | 90 | Phase III RCT | 8 | Gastrointestinal | Megestrol acetate + celecoxib(pharmacological) | Megestrol acetate + placebo | Body weightjcsm13478-note-0011 , jcsm13478-note-0012 (primary) |
| Schink et al.ref. jcsm13478-bib-0075 | 2018 | 131 | Pilot non‐randomized trial | 9 | Any solid tumour | Whole‐body electromyostimulation + nutritional counselling(multi‐modal) | Nutritional counselling | Lean body mass—BIA(primary)Fat mass—BIABody weightjcsm13478-note-0011 |
| Uster et al.ref. jcsm13478-bib-0076 | 2018 | 58 | RCT | 9 | Gastrointestinal/lung | Exercise programme + nutritional counselling(multi‐modal) | Standard care | Body weightjcsm13478-note-0011 |
| Xie et al.ref. jcsm13478-bib-0077, jcsm13478-note-0009 | 2018 | 54 | RCT | 8 | Lung | Thalidomide + cinobufagin(pharmacological) | Cinobufagin | Body weightjcsm13478-note-0011 Mid‐arm circumference |
| Britton et al.ref. jcsm13478-bib-0079 | 2019 | 307 | RCT | 7 | Head and neck | Psychological nutritional intervention(nutritional) | Standard care | Body weightjcsm13478-note-0012 |
| Cereda et al.ref. jcsm13478-bib-0080 | 2019 | 166 | RCT | 8 | Other mixed | Whey protein isolate supplement + nutritional counselling(nutritional) | Nutritional counselling | Fat‐free mass index—BIABody weightjcsm13478-note-0011 |
| Laviano et al.ref. jcsm13478-bib-0081 | 2019 | 55 | Pilot RCT | 8 | NSCLC | Targeted medical nutrition supplement(nutritional) | Isocaloric comparator drink | Skeletal muscle area—CTVisceral fat area—CTAppendicular LBM—DEXAFat mass—DEXABody weightjcsm13478-note-0011 |
| Wiskemann et al.ref. jcsm13478-bib-0084 | 2019 | 65 | RCT | 5 | Pancreatic | Supervised resistance training or home‐based resistance training(exercise) | Standard care | Body weightjcsm13478-note-0012 (primary) |
| Boulec et al.ref. jcsm13478-bib-0085 | 2020 | 111 | RCT | 7 | Any malignancy | Parenteral nutrition(nutritional) | Oral feeding | Body weightjcsm13478-note-0011 |
| Huang et al.ref. jcsm13478-bib-0086 | 2020 | 119 | RCT | 7 | Nasopharyngeal | Nutritional supplements(nutritional) | Standard care | Body weightjcsm13478-note-0011 |
| Movahed et al.ref. jcsm13478-bib-0088 | 2020 | 100 | RCT | 8 | Oesophageal | Supplements ± enteral or parenteral nutrition ± pharmacotherapy + nutritional counselling(multi‐modal) | Nutritional counselling | Fat mass—BIAFat‐free mass index—BIABody weightjcsm13478-note-0011 BMI |
| Currow et al.ref. jcsm13478-bib-0091 | 2021 | 190 | Phase III RCT | 6 | Any malignancy | Megestrol acetate or dexamethasone(pharmacological) | Placebo | Body weightjcsm13478-note-0011 |
| Hunter et al.ref. jcsm13478-bib-0092 | 2021 | 120 | Phase III RCT | 7 | Any solid tumour | Mirtazapine(pharmacological) | Placebo | Lean body mass—BIABody weightjcsm13478-note-0011 |
| Tobberup et al.ref. jcsm13478-bib-0094 | 2021 | 120 | Non‐randomized trial | 9 | NSCLC | Fish oil + nutritional counselling + exercise programme(multi‐modal) | Standard care(historical comparator) | Skeletal muscle area—CTBody weightjcsm13478-note-0011 |
| BMI | ||||||||
| Gonçalves Dias et al.ref. jcsm13478-bib-0037, jcsm13478-note-0009 | 2005 | 64 | Non‐randomized trial | 1 | Head and neck | Home enteral (nasogastric) feeding or oral diet + nutritional supplements(nutritional) | Oral diet | Body weightjcsm13478-note-0011 BMIMid‐arm circumferenceMid‐arm muscle areaTriceps skinfold thickness |
| Kraft et al.ref. jcsm13478-bib-0046 | 2012 | 72 | RCT | 10 | Pancreatic | l‐Carnitine supplement(nutritional) | Placebo | Fat mass—BIABMI |
| Silander et al.ref. jcsm13478-bib-0049, jcsm13478-note-0009 | 2012 | 134 | RCT | 6 | Head and neck | Prophylactic PEG(nutritional) | Standard care | Body weightjcsm13478-note-0012 (primary)BMI |
| Kanat et al.ref. jcsm13478-bib-0053, jcsm13478-note-0009 | 2013 | 69 | RCT | 8 | Any malignancy | Megestrol acetate + meloxicam or megestrol acetate + EPA‐enriched nutritional supplement(pharmacological) | Meloxicam + EPA‐enriched nutritional supplement | Body weightjcsm13478-note-0011 (primary)Lean body mass—BIA(primary)BMI |
| Focan et al.ref. jcsm13478-bib-0057, jcsm13478-note-0009 | 2015 | 53 | RCT | 7 | Any malignancy | Dietetic and psychological mindfulness workshops(multi‐modal) | Standard care | BMI(primary)Body weightjcsm13478-note-0011 |
| Capozzi et al.ref. jcsm13478-bib-0059 | 2016 | 60 | Exploratory RCT | 8 | Head and neck | Early ‘lifestyle intervention’ (individualized exercise with education and support)(exercise) | Delayed ‘lifestyle intervention’ (individualized exercise with education and support) | Lean body mass—DEXAFat mass—DEXABMI |
| Werner et al.ref. jcsm13478-bib-0070 | 2017 | 60 | RCT | 7 | Pancreatic | Fish oil(nutritional) | Marine phospholipids | Body weightjcsm13478-note-0012 BMI |
| Ziętarska et al.ref. jcsm13478-bib-0071 | 2017 | 95 | RCT | 6 | Colorectal | Nutritional supplements(nutritional) | Standard care | BMI |
| Akita et al.ref. jcsm13478-bib-0078 | 2019 | 62 | RCT | 8 | Pancreatic | EPA‐enriched nutritional supplement(nutritional) | Standard care | Lean body mass—BIAFat mass—BIAPsoas muscle area—CTBMI |
| Movahed et al.ref. jcsm13478-bib-0088 | 2020 | 100 | RCT | 8 | Oesophageal | Supplements ± enteral or parenteral nutrition ± pharmacotherapy + nutritional counselling(multi‐modal) | Nutritional counselling | Fat mass—BIAFat‐free mass index—BIABody weightjcsm13478-note-0011 BMI |
| Qiu et al.ref. jcsm13478-bib-0089 | 2020 | 96 | RCT | 6 | Oesophageal | Nutritional counselling(nutritional) | Standard care | BMI |
| Storck et al.ref. jcsm13478-bib-0090 | 2020 | 52 | RCT | 10 | Other mixed | Protein supplement + nutritional counselling + exercise programme(multi‐modal) | Standard care | Lean body mass—BIAFat mass—BIABMI |
| Kutz et al.ref. jcsm13478-bib-0093 | 2021 | 58 | RCT | 7 | Head and neck | Nutritional counselling(nutritional) | Standard care | BMIFat‐free mass—BIA |
| Other anthropometry | ||||||||
| Downer et al.ref. jcsm13478-bib-0015, jcsm13478-note-0009 | 1993 | 60 | RCT | 1 | Any malignancy | Medroxyprogesterone acetate(pharmacological) | Placebo | Body weightjcsm13478-note-0011 (primary)Mid‐arm circumferenceTriceps skinfold thickness |
| Ovesen et al.ref. jcsm13478-bib-0017, jcsm13478-note-0009 | 1993 | 105 | RCT | 8 | Small‐cell‐lung/ovarian/breast | Nutritional counselling(nutritional) | Standard care | Body weightjcsm13478-note-0011 (primary)Arm muscle areaTriceps skinfold thickness |
| Beller et al.ref. jcsm13478-bib-0022, jcsm13478-note-0009 | 1997 | 240 | RCT | 4 | Any malignancy (not hormone dependent) | Megestrol acetate 480 mg or megestrol acetate 160 mg(pharmacological) | Placebo | Body weightjcsm13478-note-0011 (primary)Mid‐arm circumferenceMid‐arm fat areaMid‐arm muscle areaTriceps skinfold thickness |
| Vadell et al.ref. jcsm13478-bib-0026, jcsm13478-note-0009 | 1998 | 150 | RCT | 5 | Any malignancy | Megestrol acetate 480 mg or megestrol acetate 160 mg(pharmacological) | Placebo | Body weightjcsm13478-note-0011(primary)Mid‐arm circumferenceTriceps skinfold thickness |
| McMillan et al.ref. jcsm13478-bib-0028, jcsm13478-note-0009 | 1999 | 73 | RCT | 7 | Gastrointestinal | Megestrol acetate + ibuprofen(pharmacological) | Megestrol acetate + placebo | Body weightjcsm13478-note-0011(primary)Mid‐arm circumferenceTriceps skinfold thicknessBiceps skinfold thickness |
| Bruera et al.ref. jcsm13478-bib-0033, jcsm13478-note-0009 | 2003 | 91 | RCT | 7 | Any malignancy | Fish oil capsules(nutritional) | Placebo | Lean body mass—BIABody weightjcsm13478-note-0011 Mid‐arm muscle circumferenceTriceps skinfold thicknessSubscapular skinfold thickness |
| Gonçalves Dias et al.ref. jcsm13478-bib-0037, jcsm13478-note-0009 | 2005 | 64 | Non‐randomized trial | 1 | Head and neck | Home enteral (nasogastric) feeding or oral diet + nutritional supplements(nutritional) | Oral diet | Body weightjcsm13478-note-0011 BMIMid‐arm circumferenceMid‐arm muscle areaTriceps skinfold thickness |
| Lundholm et al.ref. jcsm13478-bib-0036, jcsm13478-note-0009 | 2004 | 309 | RCT | 5 | Any solid tumour | Indomethacin + erythropoietin + nutritional support + home total parenteral nutrition(multi‐modal) | Indomethacin + erythropoietin | Fat mass—DEXALean body mass—DEXABody weightjcsm13478-note-0011 Mid‐arm muscle circumferenceTriceps skinfold thickness |
| Gordon et al.ref. jcsm13478-bib-0038, jcsm13478-note-0009 | 2005 | 50 | RCT | 10 | Pancreatic | Thalidomide(pharmacological) | Placebo | Body weightjcsm13478-note-0011(primary)Bone‐free arm muscle area |
| Berk et al.ref. jcsm13478-bib-0040, jcsm13478-note-0009 | 2008 | 472 | RCT | 9 | Any solid tumour | Nutritional supplement(nutritional) | Placebo | Lean body mass—BIA(primary)Body weightjcsm13478-note-0012 Various skinfold thickness |
| Beijer et al.ref. jcsm13478-bib-0042, jcsm13478-note-0009 | 2009 | 100 | RCT | 8 | Any malignancy | Adenosine 5′‐triphosphate(pharmacological) | Standard care | Triceps skinfold thickness(primary)Body weightjcsm13478-note-0011 Mid‐arm circumference |
| Kapoor et al.ref. jcsm13478-bib-0060, jcsm13478-note-0009 | 2016 | 63 | RCT | 8 | Any malignancy | Improved atta (nutritional supplement) + nutritional counselling(nutritional) | Nutritional counselling | Body weightjcsm13478-note-0011 Mid‐arm circumferenceVarious skinfold thickness |
| Mehrzad et al.ref. jcsm13478-bib-0061, jcsm13478-note-0009 | 2016 | 70 | RCT | 8 | Any malignancy (not brain) | Pentoxifylline(pharmacological) | Placebo | Body weightjcsm13478-note-0011 Mid‐arm circumference |
| Xie et al.ref. jcsm13478-bib-0077, jcsm13478-note-0009 | 2018 | 54 | RCT | 8 | Lung | Thalidomide + cinobufagin(pharmacological) | Cinobufagin | Body weightjcsm13478-note-0011 Mid‐arm circumference |
Note: Sample sizes are reported as per ‘intention to treat’. Abbreviations: BIA, bioimpedance analysis; BMI, body mass index; CT, computed tomography; DEXA, dual‐energy X‐ray absorptiometry; EPA, eicosapentaenoic acid; NSCLC, non‐small‐cell lung cancer; PEG, percutaneous endoscopic gastrostomy; RCT, randomized controlled trial.
Considered more than one anthropometric estimate of body composition.
Endpoints that are bold underlined had a statistically significant difference between groups.
Endpoint expressed as change in absolute value from baseline.
Endpoint expressed as percentage change from baseline.
| Author (reference) | Year | Sample size | Study design | Quality score | Primary cancer site | Intervention | Comparator | Body composition outcomesjcsm13478-note-0015 |
|---|---|---|---|---|---|---|---|---|
| BIA body composition | ||||||||
| Bruera et al.ref. jcsm13478-bib-0033 | 2003 | 91 | RCT | 7 | Any malignancy | Fish oil capsules(nutritional) | Placebo | Lean body mass—BIABody weightMid‐arm muscle circumferenceTriceps skinfold thicknessSubscapular skinfold thickness |
| Fearon et al.ref. jcsm13478-bib-0034 | 2003 | 200 | RCT | 8 | Pancreatic | n‐3 fatty acid and antioxidant‐enriched supplement(nutritional) | Supplement without n‐3 fatty acid and antioxidants | Lean body mass—BIABody weight |
| Isenring et al.ref. jcsm13478-bib-0035 | 2004 | 60 | RCT | 8 | Gastrointestinal/head and neck | Nutrition counselling and protocol(nutritional) | Standard care | Fat‐free mass—BIABody weight |
| Fearon et al.ref. jcsm13478-bib-0039 | 2006 | 518 | RCT | 8 | Gastrointestinal/lung | EPA 2 g or EPA 4 g(pharmacological) | Placebo | Lean body mass—BIABody weight |
| Berk et al.ref. jcsm13478-bib-0040 | 2008 | 472 | RCT | 9 | Any solid tumour | Nutritional supplement(nutritional) | Placebo | Lean body mass—BIA(primary)Body weightVarious skinfold thickness |
| Wiedenmann et al.ref. jcsm13478-bib-0041 | 2008 | 86 | Phase II RCT | 7 | Pancreatic | Infliximab 5 mg/kg orinfliximab 3 mg/kg(pharmacological) | Placebo | Lean body mass—BIA(primary) |
| Mantovani et al.ref. jcsm13478-bib-0043, jcsm13478-note-0014 | 2010 | 332 | Phase III RCT | 7 | Any malignancy | Megestrol acetate or EPA‐enriched nutritional supplement or l‐carnitine or thalidomide(pharmacological) | Megestrol acetate + EPA‐enriched nutritional supplement + l‐carnitine + thalidomide | Lean body mass—DEXA(primary)Lean body mass—BIA(primary)Lean body mass—CT(primary) |
| Kraft et al.ref. jcsm13478-bib-0046 | 2012 | 72 | RCT | 10 | Pancreatic | l‐Carnitine supplement(nutritional) | Placebo | Fat mass—BIABMI |
| Madeddu et al.ref. jcsm13478-bib-0048, jcsm13478-note-0014 | 2012 | 60 | Phase III RCT | 7 | Any malignancy | l‐Carnitine + celecoxib + megestrol acetate(pharmacological) | l‐Carnitine + celecoxib | Lean body mass—DEXA(primary)Lean body mass—CT(primary)Lean body mass—BIA(primary) |
| Del Fabbro et al.ref. jcsm13478-bib-0051 | 2013 | 73 | RCT | 10 | Gastrointestinal/lung | Melatonin(pharmacological) | Placebo | Body weight(primary)Lean body mass—BIAFat‐free mass—BIA |
| Kanat et al.ref. jcsm13478-bib-0053 | 2013 | 69 | RCT | 8 | Any malignancy | Megestrol acetate + meloxicam or megestrol acetate + EPA‐enriched nutritional supplement(pharmacological) | Meloxicam + EPA‐enriched nutritional supplement | Body weight(primary)Lean body mass—BIA(primary)BMI |
| Poulsen et al.ref. jcsm13478-bib-0054 | 2013 | 61 | RCT | 5 | Oesophageal/gastric/gynaecological | Nutritional counselling(nutritional) | Standard care | Body weight(primary)Fat mass—BIAFat‐free mass—BIA |
| Pottel et al.ref. jcsm13478-bib-0056, jcsm13478-note-0014 | 2014 | 85 | Exploratory RCT | 8 | Head and neck | Echium oil(nutritional) | Sunflower oil | Body weight(primary)Lean body mass—DEXAFat mass—DEXALean body mass—BIAFat mass—BIAFat‐free mass—BIA |
| Takayama et al.ref. jcsm13478-bib-0063, jcsm13478-note-0014 | 2016 | 181 | Phase II RCT | 8 | NSCLC | Anamorelin 100 mg or anamorelin 50 mg(pharmacological) | Placebo | Lean body mass—DEXA(primary)Fat mass—DEXALean body mass—BIAFat mass—BIABody weight |
| Schink et al.ref. jcsm13478-bib-0075 | 2018 | 131 | Pilot non‐randomized trial | 9 | Any solid tumour | Whole‐body electromyostimulation + nutritional counselling(multi‐modal) | Nutritional counselling | Lean body mass—BIA(primary)Fat mass—BIABody weight |
| Akita et al.ref. jcsm13478-bib-0078, jcsm13478-note-0014 | 2019 | 62 | RCT | 8 | Pancreatic | EPA‐enriched nutritional supplement + nutritional guidance(nutritional) | Nutritional guidance | Lean body mass—BIAFat mass—BIAPsoas muscle area—CTBMI |
| Cereda et al.ref. jcsm13478-bib-0080 | 2019 | 166 | RCT | 8 | Other mixed | Whey protein isolate supplement + nutritional counselling(nutritional) | Nutritional counselling | Fat‐free mass index—BIABody weight |
| Obling et al.ref. jcsm13478-bib-0082 | 2019 | 47 | RCT | 7 | Gastrointestinal | Supplemental home parenteral nutrition and nutritional counselling(nutritional) | Nutritional counselling | Fat‐free mass—BIA(primary)Fat‐free mass index—BIA(primary) |
| Stuecher et al.ref. jcsm13478-bib-0083 | 2019 | 44 | RCT | 8 | Gastrointestinal | Walking exercise programme(exercise) | Standard care | Lean body mass—BIA |
| Movahed et al.ref. jcsm13478-bib-0088 | 2020 | 100 | RCT | 8 | Oesophageal | Supplements ± enteral or parenteral nutrition ± pharmacotherapy + nutritional counselling(multi‐modal) | Nutritional counselling | Fat mass—BIAFat‐free mass index—BIABody weightBMI |
| Storck et al.ref. jcsm13478-bib-0090 | 2020 | 52 | RCT | 10 | Other mixed | Whey protein supplement + nutritional counselling + exercise programme(multi‐modal) | Standard care | Lean body mass—BIAFat mass—BIABMI |
| Hunter et al.ref. jcsm13478-bib-0092 | 2021 | 120 | Phase III RCT | 7 | Any solid tumour | Mirtazapine(pharmacological) | Placebo | Lean body mass—BIABody weight |
| Kutz et al.ref. jcsm13478-bib-0093 | 2021 | 58 | RCT | 7 | Head and neck | Nutritional counselling(nutritional) | Standard care | BMIFat‐free mass—BIA |
| DEXA body composition | ||||||||
| Daneryd et al.ref. jcsm13478-bib-0024 | 1998 | 180 | RCT | 7 | Any malignancy | Indomethacin + erythropoietin(pharmacological) | Indomethacin | Lean body mass—DEXABody weight |
| Lundholm et al.ref. jcsm13478-bib-0036 | 2004 | 309 | RCT | 5 | Any solid tumour | Indomethacin + erythropoietin + nutritional support + home total parenteral nutrition(multi‐modal) | Indomethacin + erythropoietin | Fat mass—DEXALean body mass—DEXABody weightMid‐arm muscle circumferenceTriceps skinfold thickness |
| Mantovani et al.ref. jcsm13478-bib-0043, jcsm13478-note-0014 | 2010 | 332 | Phase II RCT | 9 | Any malignancy | Megestrol acetate or EPA‐enriched nutritional supplement or l‐carnitine or thalidomide(pharmacological) | Megestrol acetate + EPA‐enriched nutritional supplement + l‐carnitine + thalidomide | Lean body mass—DEXA(primary)Lean body mass—BIA(primary)Lean body mass—CT(primary) |
| Macciò et al.ref. jcsm13478-bib-0047 | 2012 | 144 | Phase III RCT | 8 | Gynaecological | Megestrol acetate + l‐carnitine + celecoxib + antioxidants(pharmacological) | Megestrol acetate | Lean body mass—DEXA(primary) |
| Madeddu et al.ref. jcsm13478-bib-0048, jcsm13478-note-0014 | 2012 | 60 | Phase III RCT | 7 | Any malignancy | l‐Carnitine + celecoxib + megestrol acetate(pharmacological) | l‐Carnitine + celecoxib | Lean body mass—DEXA(primary)Lean body mass—CT(primary)Lean body mass—BIA(primary) |
| Dobs et al.ref. jcsm13478-bib-0052 | 2013 | 159 | Phase II RCT | 8 | Other mixed | Enobosarm 1 mg or enobosarm 3 mg(pharmacological) | Placebo | Lean body mass—DEXA(primary)Body weightFat mass—DEXA |
| Pottel et al.ref. jcsm13478-bib-0056, jcsm13478-note-0014 | 2014 | 85 | Exploratory RCT | 8 | Head and neck | Echium oil(nutritional) | Sunflower oil | Body weight(primary)Lean body mass—DEXALean body mass—BIAFat mass—DEXAFat mass—BIAFat‐free mass—BIA |
| Garcia et al.ref. jcsm13478-bib-0058 | 2015 | 82 | Phase II RCT | 7 | Any malignancy | Anamorelin 50 mg(pharmacological) | Placebo | Lean body mass—DEXA(primary)Appendicular LBM—DEXATotal body mass—DEXAFat mass—DEXA |
| Capozzi et al.ref. jcsm13478-bib-0059 | 2016 | 60 | Exploratory RCT | 8 | Head and neck | Early ‘lifestyle intervention’ (individualized exercise with education and support)(exercise) | Delayed ‘lifestyle intervention’ (individualized exercise with education and support) | Lean body mass—DEXAFat mass—DEXABMI |
| Stewart Coats et al.ref. jcsm13478-bib-0062 | 2016 | 87 | Phase II RCT | 10 | NSCLC/colorectal | Espindolol 10 mg or espindolol 2.5 mg(pharmacological) | Placebo | Body weight(primary)Lean body mass—DEXAFat mass—DEXA |
| Takayama et al.ref. jcsm13478-bib-0063, jcsm13478-note-0014 | 2016 | 181 | Phase II RCT | 8 | NSCLC | Anamorelin 100 mg or anamorelin 50 mg(pharmacological) | Placebo | Lean body mass—DEXA(primary)Fat mass—DEXALean body mass—BIAFat mass—BIABody weight |
| Temel et al.ref. jcsm13478-bib-0011 | 2016 | 979 | Phase III RCT | 8 | NSCLC | Anamorelin(pharmacological) | Placebo | Lean body mass—DEXA(primary)Body weightTotal body mass—DEXAFat mass—DEXAAppendicular LBM—DEXA |
| Katakami et al.ref. jcsm13478-bib-0073 | 2018 | 174 | Phase III RCT | 8 | NSCLC | Anamorelin(pharmacological) | Placebo | Lean body mass—DEXA(primary)Body weight |
| Golan et al.ref. jcsm13478-bib-0072, jcsm13478-note-0014 | 2018 | 125 | Phase II RCT | 7 | Pancreatic | Anti‐myostatin antibody 300 mg or anti‐myostatin antibody 100 mg(pharmacological) | Placebo | Thigh muscle volume—CTSkeletal muscle area—CTAdipose tissue area—CTLean body mass—DEXAFat mass—DEXA |
| Laviano et al.ref. jcsm13478-bib-0081, jcsm13478-note-0014 | 2019 | 55 | Pilot RCT | 8 | NSCLC | Targeted medical nutrition supplement(nutritional) | Isocaloric comparator drink | Skeletal muscle area—CTVisceral fat area—CTAppendicular LBM—DEXAFat mass—DEXABody weight |
| Kamel et al.ref. jcsm13478-bib-0087 | 2020 | 40 | RCT | 7 | Pancreatic | Resistance training(exercise) | Standard care | Appendicular LBM—DEXAFat mass—DEXA |
| CT body composition | ||||||||
| Mantovani et al.ref. jcsm13478-bib-0043, jcsm13478-note-0014 | 2010 | 332 | Phase III RCT | 7 | Any malignancy | Megestrol acetate or EPA‐enriched nutritional supplement or l‐carnitine or thalidomide(pharmacological) | Megestrol acetate + EPA‐enriched nutritional supplement + l‐carnitine + thalidomide | Lean body mass—DEXA(primary)Lean body mass—BIA(primary)Lean body mass—CT(primary) |
| Madeddu et al.ref. jcsm13478-bib-0048, jcsm13478-note-0014 | 2012 | 60 | Phase III RCT | 7 | Any malignancy | l‐Carnitine + celecoxib + megestrol acetate(pharmacological) | l‐Carnitine + celecoxib | Lean body mass—DEXA(primary)Lean body mass—CT(primary)Lean body mass—BIA(primary) |
| Sandmael et al.ref. jcsm13478-bib-0068 | 2017 | 41 | Pilot RCT | 9 | Head and neck | Exercise and nutrition intervention during radiotherapy treatment(multi‐modal) | Exercise and nutrition intervention after radiotherapy treatment | Skeletal muscle index—CTBody weight |
| Solheim et al.ref. jcsm13478-bib-0069 | 2017 | 46 | Phase II RCT | 8 | NSCLC/pancreatic | Exercise, celecoxib + nutritional supplements(multi‐modal) | Standard care | Body weightSkeletal muscle area—CT |
| Golan et al.ref. jcsm13478-bib-0072, jcsm13478-note-0014 | 2018 | 125 | Phase II RCT | 7 | Pancreatic | Anti‐myostatin antibody 300 mg or anti‐myostatin antibody 100 mg(pharmacological) | Placebo | Thigh muscle volume—CTSkeletal muscle area—CTAdipose tissue area—CTLean body mass—DEXAFat mass—DEXA |
| Akita et al.ref. jcsm13478-bib-0078, jcsm13478-note-0014 | 2019 | 62 | RCT | 8 | Pancreatic | EPA‐enriched nutritional supplement + nutritional guidance(nutritional) | Nutritional guidance | Lean body mass—BIAFat mass—BIAPsoas muscle area—CTBMI |
| Laviano et al.ref. jcsm13478-bib-0081, jcsm13478-note-0014 | 2019 | 55 | Pilot RCT | 8 | NSCLC | Targeted medical nutrition supplement(nutritional) | Isocaloric comparator drink | Skeletal muscle area—CTVisceral fat area—CTAppendicular LBM—DEXAFat mass—DEXABody weight |
| Tobberup et al.ref. jcsm13478-bib-0094 | 2021 | 120 | Non‐randomized trial | 9 | NSCLC | Fish oil + nutritional counselling + exercise programme(multi‐modal) | Standard care(historical comparator) | Skeletal muscle area—CTBody weight |
Note: Sample sizes are reported as per ‘intention to treat’.
Abbreviations: BIA, bioimpedance analysis; BMI, body mass index; CT, computed tomography; DEXA, dual‐energy X‐ray absorptiometry; EPA, eicosapentaenoic acid; LBM, lean body mass; NSCLC, non‐small‐cell lung cancer; RCT, randomized controlled trial.
Considered more than one radiological or bioelectrical estimate of body composition.
Endpoints that are bold underlined had a statistically significant difference between groups.
References
- Definition and classification of cancer cachexia: an international consensus.. Lancet Oncol, 2011. [PubMed]
- Cancer‐associated cachexia.. Nat Rev Dis Primers, 2018. [PubMed]
- A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care.. Appl Physiol Nutr Metab, 2008. [PubMed]
- Cancer cachexia and fat–muscle physiology.. N Engl J Med, 2011. [PubMed]
- Sarcopenia: revised European consensus on definition and diagnosis.. Age Ageing, 2019. [PubMed]
- Body composition analysis using CT and MRI: intra‐individual intermodal comparison of muscle mass and myosteatosis.. Sci Rep, 2020. [PubMed]
- The PRISMA statement for reporting systematic reviews and meta‐analyses of studies that evaluate health care interventions: explanation and elaboration.. BMJ, 2009
- 8 Solheim T , Laird B , Skipworth RJE , Fallon M , Kaasa S , Dajani O . A systematic literature review examining endpoints for cancer cachexia trials. PROSPERO; 2022. Available from: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42022276710.
- Physical function endpoints in cancer cachexia clinical trials: Systematic Review 1 of the cachexia endpoints series.. J Cachexia Sarcopenia Muscle, 2023
- The feasibility of creating a checklist for the assessment of the methodological quality both of randomised and non‐randomised studies of health care interventions.. J Epidemiol Community Health, 1998. [PubMed]
- Anamorelin in patients with non‐small‐cell lung cancer and cachexia (ROMANA 1 and ROMANA 2): results from two randomised, double‐blind, phase 3 trials.. Lancet Oncol, 2016. [PubMed]
- A controlled trial of cyproheptadine in cancer patients with anorexia and/or cachexia.. Cancer, 1990. [PubMed]
- Controlled trial of megestrol acetate for the treatment of cancer anorexia and cachexia.. JNCI J Natl Cancer Inst, 1990. [PubMed]
- Usefulness of megestrol acetate in cancer cachexia and anorexia: a placebo‐controlled study.. Am J Clin Oncol, 1992. [PubMed]
- A double blind placebo controlled trial of medroxyprogesterone acetate (MPA) in cancer cachexia.. Br J Cancer, 1993. [PubMed]
- Phase III evaluation of four doses of megestrol acetate as therapy for patients with cancer anorexia and/or cachexia.. JCO, 1993
- Effect of dietary counseling on food intake, body weight, response rate, survival, and quality of life in cancer patients undergoing chemotherapy: a prospective, randomized study.. JCO, 1993
- Pentoxifylline for treatment of cancer anorexia and cachexia? A randomized, double‐blind, placebo‐controlled trial.. JCO, 1995
- Prospective randomised trial of two dose levels of megestrol acetate in the management of anorexia‐cachexia syndrome in patients with metastatic cancer.. Br J Cancer, 1996. [PubMed]
- Is there a role for melatonin in the treatment of neoplastic cachexia?. Eur J Cancer, 1996
- Effects of medroxyprogesterone acetate on appetite, weight, and quality of life in advanced‐stage non‐hormone‐sensitive cancer: a placebo‐controlled multicenter study.. JCO, 1996
- Improved quality of life with megestrol acetate in patients with endocrine‐insensitive advanced cancer: a randomised placebo‐controlled trial.. Ann Oncol, 1997. [PubMed]
- Effect of megestrol acetate and prepulsid on nutritional improvement in patients with head and neck cancers undergoing radiotherapy.. Radiother Oncol, 1997. [PubMed]
- Protection of metabolic and exercise capacity in unselected weight‐losing cancer patients following treatment with recombinant erythropoietin: a randomized prospective study.. Cancer Res, 1998. [PubMed]
- Megestrol acetate for anorexia in patients with far‐advanced cancer: a double‐blind controlled clinical trial.. Eur J Cancer, 1998. [PubMed]
- Anticachectic efficacy of megestrol acetate at different doses and versus placebo in patients with neoplastic cachexia.. Am J Clin Oncol, 1998. [PubMed]
- Randomized comparison of megestrol acetate versus dexamethasone versus fluoxymesterone for the treatment of cancer anorexia/cachexia.. JCO, 1999
- A prospective randomized study of megestrol acetate and ibuprofen in gastrointestinal cancer patients with weight loss.. Br J Cancer, 1999. [PubMed]
- Megestrol acetate in advanced, progressive, hormone‐insensitive cancer. Effects on the quality of life: a placebo‐controlled, randomised, multicentre trial.. Eur J Cancer, 1999. [PubMed]
- Dronabinol versus megestrol acetate versus combination therapy for cancer‐associated anorexia: a North Central Cancer Treatment Group study.. JCO, 2002
- A randomized study of nutritional support in patients with colorectal and gastric cancer.. Nutr Cancer, 2002. [PubMed]
- Megestrol acetate for cachexia and anorexia in advanced non‐small cell lung cancer: a randomized study comparing two different doses.. Tumori, 2002. [PubMed]
- Effect of fish oil on appetite and other symptoms in patients with advanced cancer and anorexia/cachexia: a double‐blind, placebo‐controlled study.. J Clin Oncol, 2003. [PubMed]
- Effect of a protein and energy dense n‐3 fatty acid enriched oral supplement on loss of weight and lean tissue in cancer cachexia: a randomised double blind trial.. Gut, 2003. [PubMed]
- Nutrition intervention is beneficial in oncology outpatients receiving radiotherapy to the gastrointestinal or head and neck area.. Br J Cancer, 2004. [PubMed]
- Palliative nutritional intervention in addition to cyclooxygenase and erythropoietin treatment for patients with malignant disease: effects on survival, metabolism, and function: a randomized prospective study.. Cancer, 2004. [PubMed]
- Nutritional intervention improves the caloric and proteic ingestion of head and neck cancer patients under radiotherapy.. Nutr Hosp, 2005. [PubMed]
- Thalidomide in the treatment of cancer cachexia: a randomised placebo controlled trial.. Gut, 2005. [PubMed]
- Double‐blind, placebo‐controlled, randomized study of eicosapentaenoic acid diester in patients with cancer cachexia.. JCO, 2006
- A randomized, double‐blind, placebo‐controlled trial of a β‐hydroxyl β‐methyl butyrate, glutamine, and arginine mixture for the treatment of cancer cachexia (RTOG 0122).. Support Care Cancer, 2008. [PubMed]
- A multicenter, phase II study of infliximab plus gemcitabine in pancreatic cancer cachexia.. J Support Oncol, 2008. [PubMed]
- Effect of adenosine 5′‐triphosphate infusions on the nutritional status and survival of preterminal cancer patients.. Anticancer Drugs, 2009. [PubMed]
- Randomized phase III clinical trial of five different arms of treatment in 332 patients with cancer cachexia.. Oncologist, 2010. [PubMed]
- Treatment of cancer‐related anorexia with olanzapine and megestrol acetate: a randomized trial.. Support Care Cancer, 2010. [PubMed]
- Simple nutritional intervention in patients with advanced cancers of the gastrointestinal tract, non‐small cell lung cancers or mesothelioma and weight loss receiving chemotherapy: a randomised controlled trial.. J Hum Nutr Diet, 2011. [PubMed]
- l‐Carnitine‐supplementation in advanced pancreatic cancer (CARPAN)—a randomized multicentre trial.. Nutr J, 2012. [PubMed]
- A randomized phase III clinical trial of a combined treatment for cachexia in patients with gynecological cancers: evaluating the impact on metabolic and inflammatory profiles and quality of life.. Gynecol Oncol, 2012. [PubMed]
- Randomized phase III clinical trial of a combined treatment with carnitine + celecoxib ± megestrol acetate for patients with cancer‐related anorexia/cachexia syndrome.. Clin Nutr, 2012. [PubMed]
- Impact of prophylactic percutaneous endoscopic gastrostomy on malnutrition and quality of life in patients with head and neck cancer—a randomized study.. Head Neck, 2012. [PubMed]
- Clinical studies on the treatment of cancer cachexia with megestrol acetate plus thalidomide.. Chemotherapy, 2012. [PubMed]
- Effects of melatonin on appetite and other symptoms in patients with advanced cancer and cachexia: a double‐blind placebo‐controlled trial.. JCO, 2013
- Effects of enobosarm on muscle wasting and physical function in patients with cancer: a double‐blind, randomised controlled phase 2 trial.. Lancet Oncol, 2013. [PubMed]
- Comparison of three different treatment modalities in the management of cancer cachexia.. Tumori, 2013. [PubMed]
- Randomized trial of the effects of individual nutritional counseling in cancer patients.. Clin Nutr, 2014. [PubMed]
- Nutritional advice in older patients at risk of malnutrition during treatment for chemotherapy: a two‐year randomized controlled trial.. PLoS ONE., 2014. [PubMed]
- Echium oil is not protective against weight loss in head and neck cancer patients undergoing curative radio(chemo)therapy: a randomised‐controlled trial.. BMC Complement Altern Med, 2014. [PubMed]
- Dietetic and psychological mindfulness workshops for the management of cachectic cancer patients. A randomized study.. Anticancer Res, 2015
- Anamorelin for patients with cancer cachexia: an integrated analysis of two phase 2, randomised, placebo‐controlled, double‐blind trials.. Lancet Oncol, 2015. [PubMed]
- Patient‐reported outcomes, body composition, and nutrition status in patients with head and neck cancer: results from an exploratory randomized controlled exercise trial.. Cancer, 2016. [PubMed]
- A prospective randomized controlled trial to study the impact of a nutrition‐sensitive intervention on adult women with cancer cachexia undergoing palliative care in India.. Integr Cancer Ther, 2017. [PubMed]
- Pentoxifylline treatment in patients with cancer cachexia: a double‐blind, randomized, placebo‐controlled clinical trial.. Adv Biomed Res, 2016. [PubMed]
- Espindolol for the treatment and prevention of cachexia in patients with stage III/IV non‐small cell lung cancer or colorectal cancer: a randomized, double‐blind, placebo‐controlled, international multicentre phase II study (the ACT‐ONE trial).. J Cachexia Sarcopenia Muscle, 2016. [PubMed]
- Anamorelin (ONO‐7643) in Japanese patients with non‐small cell lung cancer and cachexia: results of a randomized phase 2 trial.. Support Care Cancer, 2016. [PubMed]
- Efficacy of pancreatic exocrine replacement therapy for patients with unresectable pancreatic cancer in a randomized trial.. Pancreatology, 2016. [PubMed]
- ROMANA 3: a phase 3 safety extension study of anamorelin in advanced non‐small‐cell lung cancer (NSCLC) patients with cachexia.. Ann Oncol, 2017. [PubMed]
- A double‐blind, placebo‐controlled randomized trial of creatine for the cancer anorexia/weight loss syndrome (N02C4): an Alliance trial.. Ann Oncol, 2017. [PubMed]
- The effect of a home delivery meal service of energy‐ and protein‐rich meals on quality of life in malnourished outpatients suffering from lung cancer: a randomized controlled trial.. Nutr Cancer, 2017. [PubMed]
- Feasibility and preliminary effects of resistance training and nutritional supplements during versus after radiotherapy in patients with head and neck cancer: a pilot randomized trial.. Cancer, 2017. [PubMed]
- A randomized phase II feasibility trial of a multimodal intervention for the management of cachexia in lung and pancreatic cancer.. J Cachexia Sarcopenia Muscle, 2017. [PubMed]
- Dietary supplementation with n‐3‐fatty acids in patients with pancreatic cancer and cachexia: marine phospholipids versus fish oil—a randomized controlled double‐blind trial.. Lipids Health Dis, 2017. [PubMed]
- Chemotherapy‐related toxicity, nutritional status and quality of life in precachectic oncologic patients with, or without, high protein nutritional support. A prospective, randomized study.. Nutrients, 2017. [PubMed]
- LY2495655, an antimyostatin antibody, in pancreatic cancer: a randomized, phase 2 trial.. J Cachexia Sarcopenia Muscle, 2018. [PubMed]
- Anamorelin (ONO‐7643) for the treatment of patients with non‐small cell lung cancer and cachexia: results from a randomized, double‐blind, placebo‐controlled, multicenter study of Japanese patients (ONO‐7643‐04).. Cancer, 2018. [PubMed]
- Randomized double‐blind clinical trial of combined treatment with megestrol acetate plus celecoxib versus megestrol acetate alone in cachexia‐anorexia syndrome induced by GI cancers.. Support Care Cancer, 2018. [PubMed]
- Effects of whole‐body electromyostimulation combined with individualized nutritional support on body composition in patients with advanced cancer: a controlled pilot trial.. BMC Cancer, 2018. [PubMed]
- Effects of nutrition and physical exercise intervention in palliative cancer patients: a randomized controlled trial.. Clin Nutr, 2018. [PubMed]
- Clinical study on thalidomide combined with cinobufagin to treat lung cancer cachexia.. J Can Res Ther, 2018
- The utility of nutritional supportive care with an eicosapentaenoic acid (EPA)‐enriched nutrition agent during pre‐operative chemoradiotherapy for pancreatic cancer: prospective randomized control study.. Clin Nutr ESPEN, 2019. [PubMed]
- Eating As Treatment (EAT): a stepped‐wedge, randomized controlled trial of a health behavior change intervention provided by dietitians to improve nutrition in patients with head and neck cancer undergoing radiation therapy (TROG 12.03).. Int J Radiat Oncol*Biology*Phys, 2019
- Whey protein isolate supplementation improves body composition, muscle strength, and treatment tolerance in malnourished advanced cancer patients undergoing chemotherapy.. Cancer Med, 2019. [PubMed]
- Safety and tolerability of targeted medical nutrition for cachexia in non‐small‐cell lung cancer: a randomized, double‐blind, controlled pilot trial.. Controlled Pilot Trial Nutr Cancer, 2020. [PubMed]
- Home parenteral nutrition increases fat free mass in patients with incurable gastrointestinal cancer. Results of a randomized controlled trial.. Clin Nutr, 2019. [PubMed]
- Exercise improves functional capacity and lean body mass in patients with gastrointestinal cancer during chemotherapy: a single‐blind RCT.. Support Care Cancer, 2019. [PubMed]
- Progressive resistance training to impact physical fitness and body weight in pancreatic cancer patients: a randomized controlled trial.. Pancreas, 2019. [PubMed]
- Impact on health‐related quality of life of parenteral nutrition for patients with advanced cancer cachexia: results from a randomized controlled trial.. Oncologist, 2020. [PubMed]
- A prospective randomized controlled trial on the value of prophylactic oral nutritional supplementation in locally advanced nasopharyngeal carcinoma patients receiving chemo‐radiotherapy.. Oral Oncol, 2020. [PubMed]
- Resistance training impact on mobility, muscle strength and lean mass in pancreatic cancer cachexia: a randomized controlled trial.. Clin Rehabil, 2020. [PubMed]
- Effects of medical nutrition therapy compared with general nutritional advice on nutritional status and nutrition‐related complications in esophageal cancer patients receiving concurrent chemoradiation: a randomized controlled trial.. MNM, 2020
- Effect of whole‐course nutrition management on patients with esophageal cancer undergoing concurrent chemoradiotherapy: a randomized control trial.. Nutrition, 2020. [PubMed]
- Effect of a leucine‐rich supplement in combination with nutrition and physical exercise in advanced cancer patients: a randomized controlled intervention trial.. Clin Nutr, 2020. [PubMed]
- A randomised, double blind, placebo‐controlled trial of megestrol acetate or dexamethasone in treating symptomatic anorexia in people with advanced cancer.. Sci Rep, 2021. [PubMed]
- Mirtazapine in cancer‐associated anorexia and cachexia: a double‐blind placebo‐controlled randomized trial.. J Pain Symptom Manage, 2021. [PubMed]
- Quality of life, HPV‐status and phase angle predict survival in head and neck cancer patients under (chemo)radiotherapy undergoing nutritional intervention: results from the prospective randomized HEADNUT‐trial.. Radiother Oncol, 2022. [PubMed]
- Feasibility of a multimodal intervention on malnutrition in patients with lung cancer during primary anti‐neoplastic treatment.. Clin Nutr, 2021. [PubMed]
- Prevalence and prognostic value of sarcopenic obesity in patients with cancer: a systematic review and meta‐analysis.. Nutrition, 2022. [PubMed]
- Definition and diagnostic criteria for sarcopenic obesity: ESPEN and EASO consensus statement.. Clin Nutr, 2022. [PubMed]
- Diagnostic criteria for the classification of cancer‐associated weight loss.. JCO, 2015
- Reliability and agreement of various InBody body composition analyzers as compared to dual‐energy X‐ray absorptiometry in healthy men and women.. J Clin Densitom, 2020. [PubMed]
- Concordance between muscle mass assessed by bioelectrical impedance analysis and by dual energy X‐ray absorptiometry: a cross‐sectional study.. BMC Musculoskelet Disord, 2015. [PubMed]
- Measurement of body composition: agreement between methods of measurement by bioimpedance and computed tomography in patients with non‐small cell lung cancer.. Clin Nutr ESPEN, 2021. [PubMed]
- Pitfalls in the measurement of muscle mass: a need for a reference standard.. J Cachexia Sarcopenia Muscle, 2018. [PubMed]
- Whole‐body adipose tissue analysis: comparison of MRI, CT and dual energy X‐ray absorptiometry.. BJR, 2009. [PubMed]
- Myosteatosis and prognosis in cancer: systematic review and meta‐analysis.. Crit Rev Oncol Hematol, 2020. [PubMed]
- Prognostic value of sarcopenia in adults with solid tumours: a meta‐analysis and systematic review.. Eur J Cancer, 2016. [PubMed]
- International variation in radiation dose for computed tomography examinations: prospective cohort study.. BMJ, 2019
- Comparison of effective dose to children and adults from dual X‐ray absorptiometry examinations.. Bone, 2006. [PubMed]
- Reliability of multifrequency bioelectrical impedance analysis to quantify body composition in patients after musculoskeletal trauma.. Iowa Orthop J, 2022
- Evaluation of automated computed tomography segmentation to assess body composition and mortality associations in cancer patients.. J Cachexia Sarcopenia Muscle, 2020. [PubMed]
- Assessing dynamic change in muscle during treatment of patients with cancer: precision testing standards.. Clin Nutr, 2022. [PubMed]
- Imaging modalities for diagnosis and monitoring of cancer cachexia.. EJNMMI Res, 2021. [PubMed]
- Body composition from single versus multi‐slice abdominal computed tomography: concordance and associations with colorectal cancer survival.. J Cachexia Sarcopenia Muscle, 2022
- The relationship between muscle mass and function in cancer cachexia: smoke and mirrors?. Curr Opin Support Palliat Care, 2018. [PubMed]
- Once‐weekly semaglutide in adults with overweight or obesity.. N Engl J Med, 2021. [PubMed]
- GLIM criteria for the diagnosis of malnutrition—a consensus report from the global clinical nutrition community.. J Cachexia Sarcopenia Muscle, 2019. [PubMed]
- Request for regulatory guidance for cancer cachexia intervention trials.. J Cachexia Sarcopenia Muscle, 2015. [PubMed]
