The State of Knowledge of the Primary and Secondary Metabolites of the Iris Genus
Abstract
The genus Iris L. is one of the largest in the Iridaceae Juss. family, comprising more than 320 species. These plants are widespread across the Northern Hemisphere, particularly in temperate and subtropical climate zones. The greatest species diversity occurs in the Mediterranean region, as well as in Southwest and Central Asia. Throughout history, Iris species have had horticultural, cultural, and medicinal value. The rhizomes of several Iris species have long been used for treating various diseases and as a source of essential oils for the cosmetic industry. Phytochemical studies of plants have revealed a rich diversity of primary and secondary metabolites, including phenolic compounds, stilbenes, triterpenoids, quinones, amino acids, and organic acids. These specialised metabolites exhibit a wide range of biological activities, such as antitumor, antimicrobial, estrogenic, antioxidant, and other effects. Modern chromatographic and spectroscopic techniques have enabled precise structural characterisation and quantification of these compounds, thereby providing chemotaxonomic and pharmacological research. This review highlights the chemical diversity of key compounds in Iris species and examines the ecological and chemotaxonomic relationships underlying their distribution. This article aims to consolidate current phytochemical knowledge of the genus Iris and identify gaps in chemical analysis and ecological adaptation.
Article type: Review Article
Keywords: biogeography, chemotaxonomy, ecology, Iridaceae, pharmaceuticals
Affiliations: Pharmaceutical Chemistry Department National University of Pharmacy Kharkiv Ukraine; Pharmacognosy and Phytotherapy Group UCL School of Pharmacy London UK; Department of Pharmaceutical Biology Kiel University Kiel Germany; Department of Analytical and Toxicological Chemistry Lithuanian University of Health Sciences Kaunas Lithuania; Laboratory of Flora and Geobotany State Scientific Research Institute Nature Research Centre Vilnius Lithuania
License: © 2026 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202502203 | PubMed: 41503740 | PMC: PMC12780939
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (4.1 MB)
Introduction
The study of the diversity and resources of medicinal plants is currently a key topic in biodiversity research and conservation. Among them, plants of the genus Iris L. are an important component of plant diversity and value worldwide for their medicinal, ornamental, edible and technical applications.
The genus Iris is one of the largest in the Iridaceae family, which comprises about 1800 species distributed among 80 genera. Almost all Iris species are rhizomatous perennial herbs, often ephemeral, ranging in height from 5 to 200 cm. They display a wide variety of flower colours and possess significant ability for vegetative reproduction due to intensive rhizome branching.
Iris species are widespread throughout the Northern Hemisphere, particularly in temperate and subtropical regions. The largest species diversity occurs in the Mediterranean region, Southwest Asia and Central Asia. These plants usually grow in open, sunny habitats, although a few species are adapted to shaded or marshy areas. Many species exhibit high ecological plasticity and are classified as mesophytes, showing strong potential for successful cultivation and introduction to new environments.
Historically, Iris species have been cultivated as ornamental plants thanks to their striking floral morphology and bright colours. Additionally, essential oils extracted from the flowers have been used in perfumery, whereas the rhizomes have long been employed in traditional medicine [ref. 1]. The scientific name Iris was assigned by the Greek physician Hippocrates around the 4th century bce. The medicinal properties of Iris rhizomes were later documented by Dioscorides, Theophrastus, Aristotle and other classical physicians.
Ethnobotanical records highlight the use of Iris rhizomes as diaphoretic, expectorant and laxative agents, and in the treatment of liver and gallbladder disorders, cardiovascular diseases, venereal infections, ringworm, fever and oedema, as well as for blood purification [ref. 2]. The use of the underground parts of several Iris species was well established in traditional European medicine for centuries. The peeled and dried rhizomes of Iris × germanica, Iris forentina and Iris pallida, known collectively as Rhizoma iridis, were valued for their emetic, cathartic, stimulant, expectorant and errhine properties and were also used in tooth powders.
The rhizomes of Iris species are a rich source of secondary metabolites belonging to the classes of flavonoids, isoflavonoids, xanthones, stilbenes, simple phenolic compounds, triterpenoids, quinones and irons. These phytochemicals contribute to the diverse and beneficial properties of Iris extracts. The pharmacological activity of pure compounds and plant extracts from Iris species has been extensively described by Khatib [ref. 1], Singab [ref. 2] and Wang [ref. 3]. These plant extracts possess anti‐inflammatory, antioxidant, phytoestrogenic, antidiabetic, hepatoprotective, hypolipidemic, immunomodulating, antimutagenic, antimicrobial, antitumour, cytotoxic, antidepressant, anticholinesterase and molluscicidal effects. Recent studies have also highlighted the potential of Iris compounds and crude extracts against bacterial and viral infections, such as influenza H1N1 and enterovirus D68 [ref. 4]; amoebicidal activity against Acanthamoeba species [ref. 5], antiprotozoal potential [ref. 6] and cardioprotective activity [ref. 7]. Some Iris species have also been traditionally used as a natural dye for fabrics [ref. 8] and in perfumery [ref. 9].
Primary and secondary metabolites in Iris species include hundreds of compounds classified according to their biosynthetic origin. This review summarises the current state of research on the genus Iris, with focus on its phytochemical diversity and ecological significance. It also identifies promising future research directions, particularly in the discovery of natural products, conservation strategies and the sustainable use of Iris resources.
Materials and Methods
This review summarises data on the chemical constituents and biodiversity of species in the genus Iris. To compile all available information on the genus Iris in the public domain, keyword research was performed using the following search terms: Iris species, phytochemistry, secondary metabolites, primary metabolites and environmental effect. The databases included NCBI‐PubMed, Web of Knowledge, Google Scholar, ScienceDirect, Wiley Online Library, DOAJ and SpringerLink.
The taxonomy of Iris species was verified, and plant names were provided according to World Flora Online [ref. 10]. In cases of a change in taxonomic position, the species names used in the cited references are provided in synonymy (Table 1). We believe that a comprehensive analysis of the recent phytochemical profiling studies and the global distribution of species will enable natural product researchers to fully recognise the genus’s potential.
TABLE 1: A list of the analysed Iris taxa, along with their main synonyms and native range.
| Accepted name | Synonyms | Native range |
|---|---|---|
| Iris adriatica Trinajstic ex Mitic | Croatia | |
| Iris alberti Regel | Central Asia | |
| Iris aphylla L. | Iris biflora L., Iris hungarica Waldst. & Kit. | Central Europe, East Europe |
| Iris auranitica Dinsm. | Syria | |
| Iris barnumiae Foster & Baker | Southwest Asia | |
| Iris bostrensis Mouterde | Syria, Jordan | |
| Iris bracteata S.Watson | North America | |
| Iris brevicaulis Raf. | North America | |
| Iris bulleyana Dykes | East Asia | |
| Iris bungei Maxim. | East Asia | |
| Iris cathayensis Migo | East Asia | |
| Iris chrysographes Dykes | Iris dykesii Stapf | East Asia |
| Iris chrysophylla Howell | North America | |
| Iris clarkei Baker ex Hook.f. | East Asia | |
| Iris confusa Sealy | China | |
| Iris crocea Jacquem. ex R.C.Foster | Iris aurea Lindl. | Himalaya |
| Iris decora Wall. | Iris nepalensis D.Don. | South Asia |
| Iris delavayi Micheli | East Asia | |
| Iris dichotoma Pall. | East Asia | |
| Iris domestica (L.) Goldblatt & Mabb. | Belamcanda chinensis (L.) Redouté; Ixia chinensis L. | East Asia, South Asia |
| Iris douglasiana Herb. | North America | |
| Iris ensata Thunb. | Iris kaempferi Siebold ex Lem. | Asia |
| Iris florentina L. | Iris albicans Lange | Arabian Peninsula |
| Iris foetidissima L. | West Europe, North Africa | |
| Iris forrestii Dykes | East Asia | |
| Iris fulva Ker Gawl. | North America | |
| Iris × germanica L. | Iris × nepalensis Wall. ex Lindl.; Iris × sambucina L.; Iris × trojana A.Kern. ex Stapf | Balkan Peninsula |
| Iris gracilipes A.Gray | Japan | |
| Iris graminea L. | West Europe, Central Europe | |
| Iris halophila Pall. | Iris pallida Salisb. | East Europe, West Asia |
| Iris halophila var. sogdiana (Bunge) Skeels | Iris sogdiana Bunge | Central Asia |
| Iris haussknechtii Bornm. ex Baker | Iris kerneriana Asch. & Sint. ex Dykes | Turkey |
| Iris histrioides (G.F.Wilson) S.Arn. | Turkey | |
| Iris × hollandica H.R.Wehrh. | Hybridogenous species | |
| Iris hoogiana Dykes | Central Asia | |
| Iris hookeriana Foster | South Asia | |
| Iris humilis Georgi | Iris flavissima Pall. | Asia |
| Iris imbricata Lindl. | Caucasus, Iran | |
| Iris japonica Thunb. | East Asia | |
| Iris kashmiriana Baker | Himalaya | |
| Iris kemaonensis Wall. ex D.Don | Iris kumaonensis Auct.; Iris tigrina Jacquem. ex Baker | East Asia |
| Iris lactea Pall. | Asia | |
| Iris laevigata Fisch. | Iris phragmitetorum Hand.‐Mazz. | East Asia |
| Iris leptophylla Lingelsh. ex H.Limpr. | Iris sichuanensis Y.T.Zhao | East Asia |
| Iris loczyi Kanitz | Iris thianschanica (Maxim.) Vved. ex Woronow & Popov | Central Asia, South Asia |
| Iris lutescens Lam. | Iris longiflora Vest; Iris sordida Willd.; Iris subbiflora Brot. | West Europe |
| Iris marsica I.Ricci & Colas. | Italy | |
| Iris milesii Baker ex Foster | Central Asia, East Asia | |
| Iris missouriensis Nutt. | Iris montana Nutt. ex Dykes | North America |
| Iris nigricans Dinsm. | Palestine | |
| Iris oxypetala Bunge | Iris lactea subsp. chinensis (Fisch.) Kitag. | East Asia |
| Iris pallida Lam. | South Europe | |
| Iris paradoxa Steven | Iris medwedewii Fomin | Southwest Asia |
| Iris persica L. | Southwest Asia | |
| Iris planifolia (Mill.) T.Durand & Schinz | Iris alata Poir. | West Europe, North Africa |
| Iris postii Mouterde | Southwest Asia | |
| Iris potaninii Maxim. | Iris thorold Barker ex Hemsl | Asia |
| Iris pseudacorus L. | Europe, West Asia | |
| Iris pseudopumila Tineo | South Europe | |
| Iris pumila L. | Iris coerulea Spach | East Europe, West Asia |
| Iris reichenbachii Heuff. | South Europe | |
| Iris reticulata M.Bieb. | Xiphion reticulatum (M.Bieb.) Klatt | Southwest Asia |
| Iris revoluta Colas. | Italy | |
| Iris rossii Baker | East Asia | |
| Iris sanguinea Hornem. | East Asia | |
| Iris sanguinea var. sanguinea | East Asia | |
| Iris sanguinea var. tobataensis S.Akiyama & Iwashina | Japan | |
| Iris sari Schott ex Baker | Turkey | |
| Iris scariosa Willd. ex Link | Iris elongata Fisch. ex Baker | Asia |
| Iris schachtii Markgr. | Turkey | |
| Iris setina Colas. | Italy | |
| Iris setosa Pall. ex Link | North Europe, North Asia, North America | |
| Iris setosa var. hondoensis Honda | Japan | |
| Iris setosa var. nasuensis H.Hara | Japan | |
| Iris setosa var. setosa | North Europe, North Asia, North America | |
| Iris sibirica L. | Europe, West Asia | |
| Iris songarica Schrenk ex Fisch. & C.A.Mey. | Central Asia, South Asia | |
| Iris spuria L. | Xiphion spurium (L.) Alef. | Europe, Southwest Asia |
| Iris spuria subsp. carthaliniae (Fomin) B.Mathew | Iris carthaliniae Fomin | Caucasus |
| Iris suaveolens Boiss. & Reut. | South Europe, Turkey | |
| Iris susiana L. | Iris sofarana Foster | Southwest Asia |
| Iris taochia Woronow ex Grossh. | Turkey | |
| Iris tectorum Maxim. | China | |
| Iris tenuifolia Pall. | Iris acaulis Pall. | East Europe, Asia |
| Iris tingitana Boiss. & Reut. | Xiphion tingitanum (Boiss. & Reut.) Baker | North Africa |
| Iris unguicularis Poir. | North Africa, Southwest Asia | |
| Iris variegata L. | Europe | |
| Iris versicolor L. | North America | |
| Iris virginica L. | North America | |
| Iris wattii Baker ex Hook.f. | Central Asia, Southeast Asia |
Overview of Iris Taxonomy, Diversity and Distribution
The genus Iris (Fig. 1a‐c), which currently comprises around 320 species, is the largest in the Iridaceae Juss. family [ref. 10, ref. 11, ref. 12, ref. 13]. The circumscription and taxonomy of the genus Iris have long been, and continue to be, a matter of discussion. Although some authors have adopted a broad concept of the genus [ref. 10, ref. 13, ref. 14, ref. 15], others have divided it into as many as 25 narrowly defined genera [ref. 12, ref. 16]. In this study, we follow the broad definition of the genus Iris [ref. 10] (Figure 1).

Mathew [ref. 14] developed the most comprehensive and widely accepted system for the broadly circumscribed genus Iris. In this classification, the genus was divided into six subgenera: Iris L., Limniris (Tausch) Spach, Nepalensis (Dykes) G. H. M. Lawrence, Xiphium (Mill.) Spach, Scorpiris Spach and Hermodactyloides Spach. Subsequent authors have revised and modified this system [ref. 17], but the molecular studies have not supported the division [ref. 15, ref. 18, ref. 19, ref. 20, ref. 21]. Further comprehensive molecular studies are necessary to resolve the remaining taxonomic issues within the genus [ref. 13]. Moreover, natural hybridisation significantly contributes to the taxonomic complexity of the highly diverse genus [ref. 22, ref. 23].
The genus Iris is native to the Northern Hemisphere. Its most important centres of diversity are the Mediterranean region, Central Asia, East Asia and North America [ref. 10, ref. 11, ref. 13, ref. 15]. The Mediterranean region harbours the highest number of species, with around 140 [ref. 24]. A total of 57 species have been found in the mountains of the Central Asian diversity centre [ref. 13], 58 in the East Asian diversity centre [ref. 25] and 34 in North America [ref. 26].
Most Iris species occur in arid regions, where they grow in semi‐deserts and on rocky mountain slopes. However, many species found in temperate areas grow in mesic habitats, such as grasslands, forests and forest edges, or even in wetlands [ref. 13, ref. 23, ref. 24, ref. 26]. Iris species have been cultivated as ornamental plants for centuries, and tens of thousands of cultivars and hybrids have been developed to date [ref. 14, ref. 24].
Characteristics of Iris Species
The genus Iris comprises perennial herbs with rhizomes, corms or bulbs. Different taxonomists sometimes classify plants with corms or bulbs as separate genera, whereas others include them in the genus Iris [ref. 12, ref. 13, ref. 14, ref. 15, ref. 16]. The height of the plants varies greatly. The rhizome is usually thick, horizontal and creeping, although in some species, it is thin and very short. The stem is typically single, but in some species, several stems may occur in clusters, either simple or branched. The leaves are flat and sword‐shaped, with a wax coating and arranged in two rows. The stem usually bears few leaves or may even be leafless.
The flowers are large and may occur singly or in small clusters on long peduncles. The inflorescence is enclosed by one to three elongated, pointed, sometimes inflated, leaf‐like bracts. The perianth of the flower consists of six parts arranged in two rows: The three outer segments (sepals) spread out or droop downwards, whereas the three inner segments (petals) stand upright. The three styles in Iris flowers are large and resemble petals. These distinctive floral features make it easy to recognise whether a plant belongs to the genus Iris.
The flowers of Iris species are pollinated by insects, most commonly bees and bumblebees attracted by the nectar. The outer segments of the perianth serve as landing platforms for pollinators. Iris species typically flower from May to July, and the buds of the following year’s flowers are formed in the rhizome during summer. The fruit is a multi‐seeded syncarpous capsule that dehisces loculicidally along the midrib of each of the three carpels. The seeds are either flattened or nearly spherical, with a membranous or papery coat, and sometimes bear a distinct appendage.
Bioactive Compounds in Plants of the Iris Genus
Since the beginning of research on the Iris genus, several reviews have been published on the distribution of secondary metabolites in these plants [ref. 1, ref. 27, ref. 28]. One of the most comprehensive reviews of the phenolic compounds of the Iridaceae family was conducted in the 1980s by Williams et al. [ref. 29, ref. 30]. Later, in 1997, the same authors focused predominantly on flavonoids, isoflavones and xanthones, recording 17 glycosylflavones in 14 Iris species, with the aim of tracing chemical evolution within the genus [ref. 31].
In 1998, Donnelly and Boland [ref. 32] summarised data on more than 260 new isoflavonoid compounds isolated from plants, including species of the genus Iris, between 1991 and 1996. A detailed analysis of the chemical composition of Iris species was also published by Iwashina in the same year [ref. 33], describing the structure, distribution and functions of 46 different isoflavonoid aglycones from 18 Iris species, predominantly from China.
In 2005, French scientists [ref. 34] published a review on the distribution of isoflavones in plants. Among monocotyledonous taxa, Iris species exhibited the highest diversity of isoflavonoids, with 52 compounds recorded.
Chinese scientists [ref. 3] later summarised data on flavonoids and isoflavonoids of Iris species published between 1999 and 2008. By 2010, 56 isoflavones had been identified, 20 of which were new compounds (5 isoflavones and 15 isoflavone glycosides).
Between 2012 and 2015, Polish and Czech researchers [ref. 35, ref. 36] compiled information on the distribution of bioactive compounds and pharmacological activity of various Iris species. These plants were found to contain flavonoids, isoflavonoids, xanthone C‐glycosides, quinones and iridal‐type triterpenoids [ref. 37, ref. 38]. In 2016, Singab et al. [ref. 2] published a review of plants in the Iridaceae family, focusing on isoflavonoids, flavonoids, triterpenoids, quinones, xanthones and simple phenolics in the Iris genus. The authors described 58 isoflavones.
The most recent and comprehensive review of the chemical composition of Iris species was published in 2020 by Iwashina and Mizuno [ref. 39]. Between 1961 and 2020, 85 isoflavonoids, 84 flavonoid aglycones and glycosides, 6 benzenecarboxylic acids, 7 phenylpropanoids, 6 anthocyanidins, 9 aceto‐ and benzophenones, 6 stilbenes and their derivatives and 15 xanthones were isolated and identified from 90 taxa of the genus Iris, including flowers, leaves and rhizomes. This review covered approximately 32% of Iris species distributed worldwide. Chalcones, dihydrochalcones, aurones, bioflavonoids and neoflavonoids have not been reported in the Iris genus.
One of the most recent reviews by Khatib [ref. 1] focuses primarily on the ethnobotanical and ethnoveterinary uses of Iris species, as well as evaluating their pharmacological potential. It also describes the chemical composition of Iris extracts, including phenolic acids, flavonoids, alkaloids, primary metabolites and essential oil components.
In addition to general reviews, several studies have been devoted to the chemical composition and pharmacological potential of individual Iris species, particularly I. ensata [ref. 40], I. × germanica [ref. 27], I. kashmiriana [ref. 41] and I. domestica [ref. 42, ref. 43].
This present review summarises and integrates the findings of these authors while providing an updated overview of compounds identified in Iris species in recent years. Special attention is given to phenolic compounds, which are classified into six major classes: isoflavonoids and isoflavone glycosides; flavonoids and flavone glycosides; peltogynoids; coumaronochromones; rotenoids; and xanthones and phenol carboxylic acids. This classification is illustrated in Figure 2. Additionally, the review considers other classes of bioactive compound found in Iris plants, including terpenoids, amino acids, carboxylic acids, alkaloids and essential oils.
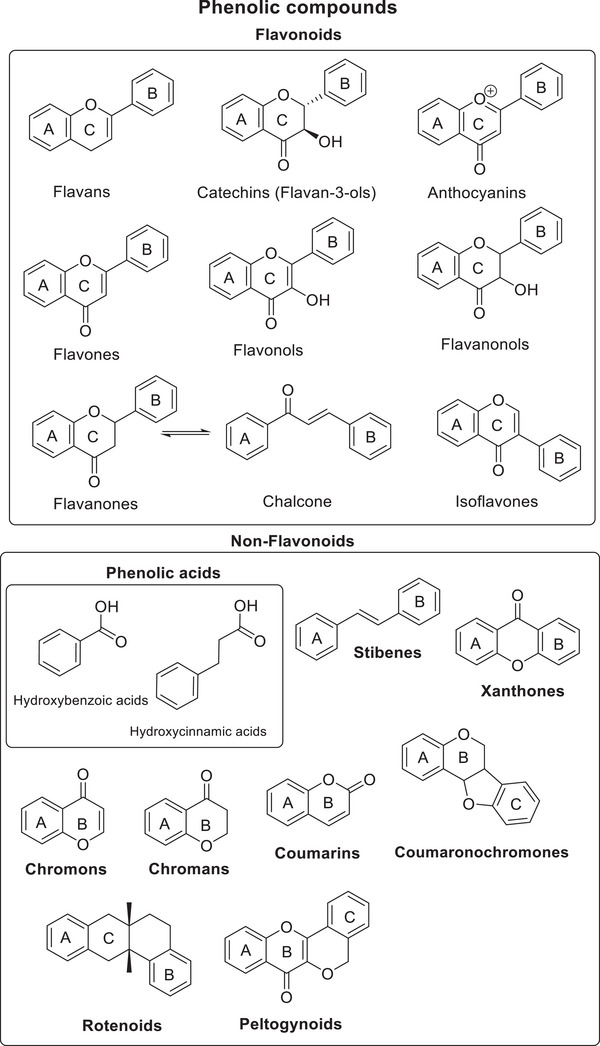
Isoflavones and flavones in Iris species typically contain simple O‐substituents, such as hydroxy‐, methoxy‐ and methylenedioxy groups, as well as sugar components. The substitution pattern differs between rings A and B: phenolic compounds most often exhibit 5,7‐di‐ or 5,6,7‐tri‐O‐substitution in ring A, whereas ring B often has 2′‐ or 4′‐O‐substitution, as well as 2′,3′‐ or 3′,4′‐di‐O‐substitution, and occasionally 3′,4′,5′‐tri‐O‐substitution. It is noteworthy that the aglycones of their glycosides rarely display 2′‐O‐ or 2′,3′‐di‐O‐substitution patterns.
In addition to typical flavonoids, Iris species also contain structurally modified phenolic compounds formed through cyclisation of flavonoid derivatives. These include peltoquinoids, coumaronochromones and protenoids. Similar to conventional flavonoids, they possess simple O‐substituents. However, a characteristic feature of these compounds is the presence of an oxygen‐containing ring condensed with rings B and C, typically involving substitution at the C‐2 or C‐3 position.
Isoflavonoids and Isoflavone Glycosides
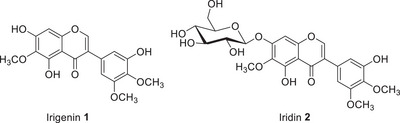
Plants of the genus Iris contain a wide range of compounds belonging to different classes and exhibiting diverse pharmacological activities. The underground organs predominantly accumulate isoflavones, xanthones, essential oils and triterpenoids, whereas the aerial parts are rich in flavonoids, polyphenolic acids and xanthones. The first isoflavonoids isolated from the rhizomes of Iris florentina were the aglycone irigenin 1 and its 7‐glucoside, iridin 2 (irigenin 7‐O‐β‐d‐glucopyranoside). Irigenin, detected in 23 Iris species [ref. 39], and iridin, identified in 16 species, are among the most widely distributed compounds within the genus (Figure 3).
Isoflavones and their glycosides are most often found in the rhizomes of Iris species and, to a lesser extent, in the leaves and flowers. Analysis of the distribution of phenolic compounds in the Iris genus indicates that I. × germanica has been the most extensively studied, followed by I. tectorum, I. florentina and I. dichotoma.
The most common isoflavones in this genus are tectorigenin and its glycosides, which have been identified in 20 species (Figure 4). Of these, six belong to subgenus Iris, four to subgenus Crossiris, six to subgenus Limniris, three to subgenus Xyridion and one to subgenus Parandanthopsis. Irilon has been reported in 12 species, and nigricin in 14 species. Tectorigenin and irigenin contain hydroxyl groups At Positions 5 and 7, as well as methoxy groups at Position 6, whereas nigricin and irilon feature a methylenedioxy group at the 6,7‐position of ring A.
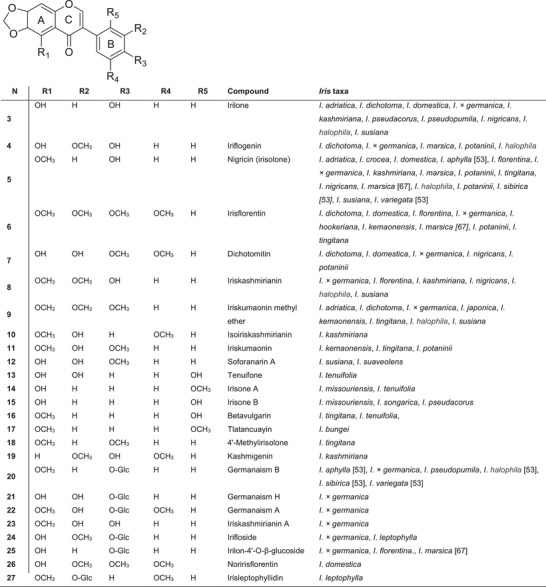
Isoflavonoids containing a methylenedioxy group in ring A are relatively rare and are unevenly distributed in nature. The Iridaceae family is the largest source of isoflavonoids, with more than 50 distinct compounds described. Figure 4 shows the most common isoflavones containing a methylenedioxy group. The largest number of isoflavonoids with a 6,7‐methylenedioxy group (32 out of 94 isoflavonoids) has been isolated from Iris species, most of which belong to the subgenus Iris. Irilone 3 was found in eight species of subgenus Iris, three species of subgenus Limniris and one species of subgenus Pardanthopsis. The highest number of isoflavonoids with a 6,7‐methylenedioxy group and their glycosides were isolated from the rhizomes of Iris potaninii (nine compounds) and I. × germanica (14 compounds). Among the most notable are iriflogenin 4, nigricin (or irisolone) 5, irisflorentin 6, dichotomitin 7, nigricanin (or iriskashmirianin) 8 and iriskumaonin methyl ether 9.
A distinctive compound of the genus Iris is irisolone 5 (also known as nigricin), which was first isolated from the rhizomes of I. florentina [ref. 44] and later from Iris nigricans [ref. 45]. In addition to nigricin, the isoflavonoids iriskashmirianin 8 and irilone 3, belonging to the 5,6,7‐trihydroxy group of isoflavones, were also isolated from the rhizomes of I. nigricans. Nigricin was found in nine species from subg. Iris, one from subg. Limniris and one from subg. Xyridion.
An example of an isoflavone found in Iris plants with hydroxy or methoxy substituents at the 3′,4′‐ or 3′,4′,5′‐tri‐O‐positions of ring B is sophoronarin A 12. This compound was first isolated from the methanol extract of the rhizomes of Iris susiana (subg. Iris) from Turkey, together with sophoronarin B [ref. 46]. Iristectorigenin B (5,7,4′‐trihydroxy‐6,4′‐dimethoxyisoflavone) was initially isolated from I. florentina rhizomes [ref. 44] and was subsequently identified in Iris pseudacorus, Iris spuria subsp. carthaliniae, I. tectorum and I. × germanica. Iristectorigenin A (5,7,3′‐trihydroxy‐6,4′‐dimethoxyisoflavone), which contains 3′,4′‐substituents in ring B, was found in eight Iris species.
Functional groups in the B ring are most commonly found at the C‐4′ position. However, some species of subgenus Limniris are characterised by isoflavonoids containing a hydroxyl or methoxyl group at the 2′‐position of the B ring. Species‐specific 6,7‐methylenedioxy isoflavonoids isolated from Iris tenuifolia include tenuifone 13 (5,2′,3′‐trihydroxy), tenuifodione, irisone B (5,2′‐dihydroxy‐) 15 and betavulgarin (6′‐hydroxy‐5‐methoxy‐) 16 [ref. 47]. Irilins A and B, along with 5,7‐dihydroxy‐6,2′‐dimethoxyisoflavone, were also identified in the rhizomes of Iris songarica.
A large number of unique isoflavonoids have been isolated from Iris species. Tlatancuyain (5,2′‐dimethoxy‐6,7‐methylenedioxyisoflavone) 17 was isolated from the rhizomes of Iris bungei [ref. 48]; irisolone (4′‐O‐[O‐β‐d‐glucopyranosyl‐(1→6)‐O‐β‐d‐glucopyranosyl‐(1→6)‐β‐d‐glucopyranoside) and iriskashmirianin (4′‐O‐[O‐β‐d‐glucopyranosyl‐(1→6)‐O‐β‐d‐glucopyranosyl‐(1→6)‐β‐d‐glucopyranoside) were isolated from the underground parts of I. florentina [ref. 49]. Additionally, 5,6‐dihydroxy‐7,8,3′,5′‐tetramethoxyisoflavone was identified in the rhizomes of I. pseudacorus [ref. 50] and has also been detected in Iris halophila, Iris aphylla, Iris sibirica, Iris variegata and I. × germanica [ref. 51, ref. 52]. Other common occurring isoflavones include tectoridin, irigenin, iristectorigenin B, irisolidone, irisolidone‐7‐O‐β‐d‐glucopyranoside and nigricin [ref. 53].
New isoflavones containing a methylenedioxy group at the 6,7‐position of ring A include the aglycone iriskashmirianin A 23 and the glycoside germanaism H 21, both first isolated from the rhizomes of I. × germanica [ref. 54]. In addition, eight known isoflavones were also isolated: irilone, iriskumaonin methyl ether, iriflogenin 4, irifloside 24, irilon‐4′‐O‐β‐glucopyranoside 25 and germanaisms A–B 20 and 22. Isoflavonoids with an oxygen‐containing functional group at the 2′‐position are uncommon in species of subgenus Iris, with I. × germanica being a notable exception. Noririsflorentin (5‐hydroxy‐6,7‐methylenedioxy‐3′,4′,5′‐trimethoxyisoflavone) 26 has been isolated only once, from I. domestica. Two unusual isoflavone monosaccharides have also been identified: irisleptophyllidin 27 from the rhizomes of Iris leptophylla [ref. 55] and 7,4′‐dimethoxy‐8,3′,5′‐trihydroxy‐6‐O‐β‐d‐glucopyranosylisoflavone [ref. 56], isolated from the underground parts of I. potaninii and structurally identified as 3′‐O‐ and 6‐O‐β‐d‐glucopyranosides, respectively.
From the ethyl acetate fraction of the rhizomes of I. spuria subsp. carthaliniae [ref. 57], three isoflavonoid glycosides were isolated for the first time: tectorigenin 4′‐glucosyl(1→6)‐glucoside, iristectorigenin B 7‐glucosyl(1→6)‐glucoside and 4′‐methyltectorigenin 7‐glucoside. Tectorigenin glucosides have also been found in the rhizomes of Iris confusa and I. dichotoma [ref. 58], as well as in the rhizomes of I. variegata, Iris milesii, I. tectorum, I. leptophylla, I. songarica, I. spuria, Iris crocea and Iris cathayensis [ref. 30]. Iristectorins A and B have been found in I. spuria, I. tectorum and I. dichotoma [ref. 58, ref. 59, ref. 60].
New mono‐ and diglycosides of germanaisms A–E and G–H, which differ in the position of the aglycone, as well as the only triglycoside of germanaism F, were isolated from the rhizomes of I. × germanica using column chromatography on silica gel. Notably, germanaism G [ref. 61, ref. 62] is 3′,4′‐di‐O‐β‐d‐glucopyranosylisoflavone and contains a 2‐methoxy‐4‐acetylphenyl glucose derivative at the C‐2 position of the 4′‐glucopyranoside. This rare phenyl glucose residue is exceedingly uncommon among isoflavone glycosides of the Iris genus. Germanaisms A, B and E also have been identified in Iris pseudopumila [ref. 63] as well as in I. leptophylla, Iris marsica and I. florentina, all belonging to subgenus Iris.
Other glycosides isolated from Iris species include iriskashmirianin 4′‐O‐β‐d‐glucoside, irilon 4′‐O‐β‐d‐glucoside, irisolidon 7‐O‐α‐d‐glucoside, irigenin 7‐O‐glucoside, irisolon‐4′‐O‐bioside and irilon‐bioside. The specific isoflavone muningin (6,4′‐dihydroxy‐5,7‐dimethoxyisoflavone) has only been identified in I. × germanica [ref. 64].
New isoflavonoid glucosides, including iridins B–C, tectoridin A and ampelopsinin A, along with one previously undescribed phenolic glucoside (diplostephioside B) and one novel phenolic compound (phenanthrenetriol A), were isolated from the rhizomes of I. domestica [ref. 65]. Isoflavanones, which are widespread in the Fabaceae and Vitaceae families, are rare in the genus Iris, with the only reported example being 2,3‐dihydroirigenin from I. halophila [ref. 66].
Although isoflavonoids are among the most studied secondary metabolites in the genus Iris, their structural diversity across subgenera remains incompletely characterised. Many of the identified compounds, especially in poorly studied ones, are promising for further analysis aimed at clarifying chemosystematic relationships within the genus.
Flavones and Glycosides of Flavones
As described by Iwashina [ref. 33] the main flavonoid components of Iris plants are C‐glycosylflavones (such as vitexin 40, isovitexin 41, isorientin 42, orientin 43 and swertisin 44) and O‐glycosylflavones, which occur mainly in the leaves and rhizomes of most species of Iris. Mono‐ and di‐C‐glycosides of apigenin, luteolin, quercetin and myricetin are also commonly present (Figure 5).
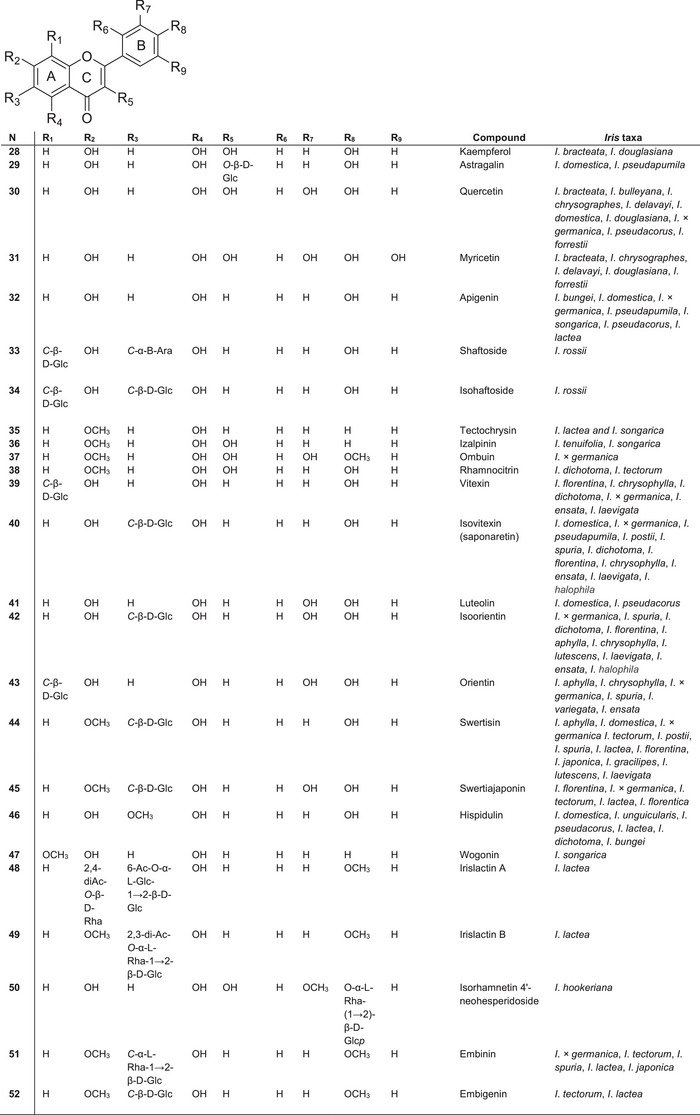
In contrast to 7‐ and 4′‐O‐glycosyl isoflavones, the sugar moieties in these compounds are typically attached at the C‐6 and C‐7 positions of the aglycone. Unlike isoflavones, flavones in Iris species predominantly occur as 6‐C‐glycosides. For example, isorhamnetin 4′‐neohesperidoside 50 and isovitexin 40 have been reported in species of subgenus Limniris (Iris rossii, I. ensata) and subgenus Iris (I. pseudopumila, I. florentina) [ref. 31, ref. 68, ref. 69]. Along with the flavon glycosides embinin 51 and embigenin 52, two unusual acetylated C‐glycosylflavone derivatives—irislactin A 48 and irislactin B 49—were isolated from the leaves of Iris lactea (subg. Limniris, sect. Limniris) [ref. 70]. These compounds differ in their substituents and the glucosylation pattern at the C‐6 and C‐7 positions of ring A.
Irislactin A 48 is an acetylated linear disaccharide, identified as 7‐O‐(β‐d‐2⁗,4⁗‐diacetylrhamnopyranosyl)‐6‐C‐[O‐(α‐l‐6‴‐acetylglucopyranosyl)‐1→2‐β‐d‐glucopyranoside] 5‐hydroxy‐4′‐methoxyflavone [ref. 71]. Irislactin B 49 is also an acylated linear disaccharide with a methoxyl group at C‐7 of ring A, that is, 6‐С‐[O‐(α‐l‐2‴,3‴‐diacetylrhamnopyranosyl)‐1→2‐β‐d‐glucopyranoside] of 5‐hydroxy‐7,4′‐dimethoxyflavone [ref. 72]. C‐glycosides of flavones (6‐С‐(O‐α‐l‐2‴‐acetylramnopyranosyl‐1→2‐β‐d‐glucopyranoside) 5‐hydroxy‐7,4′‐dimethoxyflavone and 6‐С‐(O‐α‐l‐3‴‐acetylramnopyranosyl‐1→2‐β‐d‐glucopyranoside) 5‐hydroxy‐7,4′‐dimethoxyflavone) were isolated from I. tectorum [ref. 73]. These compounds contain one fewer acetyl group than irislactin B. In I. rossii, the C‐glycosylflavone shaftoside 33 and isoshaftoside 34 were also identified [ref. 74].
The most unique flavonoid identified in the genus Iris is irisflavone D, which has 2′,6′‐di‐O‐substitution in the B‐ring, a feature that is rare among Iris flavonoids. This compound was first isolated from the rhizomes of I. bungei [ref. 75]. Irisflavone A was also isolated from the related species I. songarica. Flavones containing a methylenedioxy group at the C‐6 and C‐7 positions of ring A, such as kanzakiflavone‐1 and kanzakiflavone‐2, were isolated from the rhizomes of Iris unguicularis [ref. 76]. These two compounds differ in the position of hydroxy groups, occurring at 5,8‐OH and 5,4′‐OH, respectively.
Among the newly identified flavones, 5,2′‐dihydroxy‐3‐methoxy‐6,7‐methylenedioxyflavone, isolated from the rhizomes of I. × germanica, is notable for its 2′‐O‐substitution in ring B. This is another characteristic feature of Iris‐derived flavonoids. Between 1999 and 2008, only two new flavone aglycones were isolated: tectochrysin 35 from I. lactea and I. songarica [ref. 70] and wogonin 47 from I. songarica [ref. 77]. The novel flavonol izalpinin 36 was isolated from I. tenuifolia [ref. 47] and I. songarica [ref. 77].
The flavone ombuin 37 (4′,7‐dimethoxy‐3,3′,5‐trihydroxyflavone) was first isolated from the rhizomes of I. × germanica, marking its initial discovery within the Iridaceae family [ref. 64]. The flavonol rhamnocitrin 38 was isolated from the rhizomes of I. dichotoma and I. tectorum [ref. 78]. The latest investigation by Kai‐Dong Liu [ref. 59] described several new flavonoid glucosides, such as iridins B–C, tectoridin A and ampelopsinin A, along with one previously undescribed phenolic glucoside (diplostephioside B) and a new phenolic compound (phenanthrene triol A), isolated from the rhizomes of I. domestica.
The most recent analysis (2024) led to the isolation of five new flavonoids from the underground parts of I. tenuifolia, all bearing 2,3‐ and 3,4‐dihydroxy substituents on the phenyl ring [ref. 79].
Current knowledge of flavonoids in Iris species highlights the occurrence of both common and structurally unique flavones, especially in the form of C‐glycosides. However, there remains a significant gap in understanding the regulation of species‐specific flavone biosynthesis and their functional role in stress adaptation. These aspects could be clarified through targeted metabolomic and transcriptomic analysis. Due to the current lack of such data, these topics were not addressed in this review.
Flavanones and Dihydroflavonols
Flavanones are a prominent class of secondary metabolites in the Iris genus, notable for their structural diversity and chemotaxonomic significance. The flavanones identified across different Iris species exhibit variations in hydroxylation and methoxylation patterns, particularly on the A‐ and B‐rings (Figure 6). Several novel or rare compounds have been identified, including selenone 53 from I. × germanica [ref. 80], alpinone 54 from I. songarica rhizomes [ref. 77], 3,5,3′‐trihydroxy‐7,4′‐dimethoxyflavanonol 55 from I. potaninii [ref. 56] and aromadendrin 7‐methyl ether 56 from I. tectorum rhizomes [ref. 81]. The dihydroflavanonol alpinone 54 has been found in four species of subgenus Limniris (I. pseudacorus, I. crocea, I. tenuifolia and Iris loczyi) [ref. 39] but is absent in species of subgenus Iris. Dihydroechioidinin 57 was first reported in I. cathayensis rhizomes [ref. 82] and later detected in I. lactea and I. songarica [ref. 3].
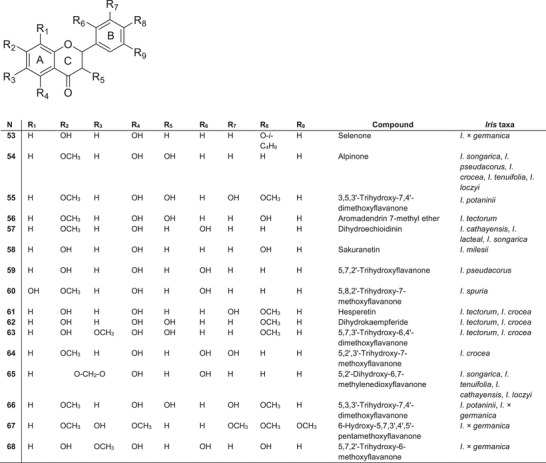
The flavanone sakuranetin 58 was uniquely isolated from I. milesii [ref. 83], whereas 5,7,2′‐trihydroxyflavanone 59 and 5,8,2′‐trihydroxy‐7‐methoxyflavanone 60 were identified in I. pseudacorus [ref. 84] and I. spuria [ref. 85], respectively. Hesperetin 61, dihydrokaempferide 62 and 5,7,3′‐trihydroxy‐6,4′‐dimethoxyflavanone 63 were reported in I. tectorum [ref. 78] and I. crocea [ref. 85]. Furthermore, Bhat et al. [ref. 86] identified 5,2′,3′‐trihydroxy‐7‐methoxyflavanone 64 in the methanol extracts of I. crocea rhizomes.
The presence of 5,2′‐dihydroxy‐6,7‐methylenedioxyflavanone 65 has been confirmed in four species of section Tenuifoliae: I. songarica [ref. 77], I. tenuifolia [ref. 47], I. cathayensis [ref. 82] and I. loczyi [ref. 87]. Flavanone 5,3,3′‐trihydroxy‐7,4′‐dimethoxyflavanone 66 was first isolated from I. potaninii rhizomes [ref. 56] and I. × germanica [ref. 64]. Additionally, I. × germanica rhizomes yielded 6‐hydroxy‐5,7,3′,4′,5′‐pentamethoxyflavanone 67 under oxygen deprivation conditions [ref. 80], as well as a novel flavanone, 5,7,2′‐trihydroxy‐6‐methoxyflavanone 68 [ref. 88].
Of particular interest are flavanones that have an oxidised substituent at the C‐2′ position of the B‐ring. Such compounds are rare in nature and are usually only found in a few plant families, such as Asteraceae, Iridaceae, Fabaceae and Moraceae. Flavanones in Iris species are characterised by a higher degree of structural substitution; however, their chemotaxonomic significance and pharmacological profiles remain poorly explored. The rare occurrence of oxygen‐containing substituents on the B‐ring requires further study, especially among taxa belonging to section Tenuifoliae.
Peltogynoids
Irisoids A–E 69–73 were isolated from the underground parts of I. bungei [ref. 48]. These compounds belong to the rare class of flavonoids known as peltoginoids, which contain an additional oxygen‐containing ring (Figure 7). In addition to I. bungei, irisoid A 69 has also been isolated from five Iris species belonging to the section Tenuifoliae, such as I. tenuifolia, I. songarica, I. cathayensis, I. loczyi and I. lactea [ref. 39], suggesting its potential chemotaxonomic significance as a marker compound for this group.
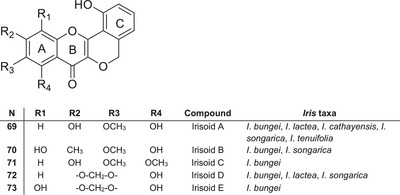
Analysis of the available data indicates that peltogynoids are restricted to a limited number of species within the section Tenuifoliae, highlighting their potential value as chemotaxonomic markers. However, due to the limited number of reports, further investigation is required to clarify their distribution, biosynthesis pathway and biological significance.
Coumaronochromones and Rotenoids
Iris species contain isoflavonoids and flavonoids, some of which have additional five‐ or six‐membered, oxygen‐containing heterocycles in their structure. Coumaronochromones, such as ayamenins A–E 74–78, have been isolated from the leaves of I. pseudacorus as stress‐induced metabolites [ref. 84, ref. 89] (Figure 8). Ayamenin B 75 and a new coumaronochromone, irisbungin (5,7,5′‐trihydroxy‐coumaronochromone) 80, were isolated from the roots of I. bungei [ref. 90]. Ayamenin A 74 has also been found in I. songarica [ref. 77] and I. lactea [ref. 91]. Pyranocoumaronochromones, such as lupinalbin 79 A and 5,7,3′‐trihydroxy‐6‐methoxycoumaronochromone 81, were identified in I. pseudacorus leaves treated with cupric chloride, which induces the production of fungitoxic stress metabolites [ref. 89]. These oxygen‐bridged flavonoid derivatives further expand the structural diversity to specialised metabolites in the Iris genus and may play roles in stress tolerance and species‐specific biological activity.
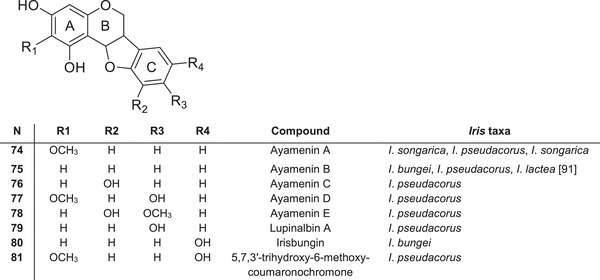
All these species belong to the subgenus Limniris. The global literature contains no evidence for the occurrence of coumaronochromones in plants belonging to other subgenera of the Iris genus. This suggests that coumaronochromones may be important chemotaxonomic markers for subgenus Limniris. In the rhizomes of I. crocea [ref. 85] and I. spuria [ref. 92], which belong to the subgenus Limniris ser. Spuriae, new 12α‐hydroxyrotenoids were identified: 1,11‐dihydroxy‐9,10‐methylenedioxy‐ 82, irispurinol (9,11,12‐trihydroxy‐10‐methoxy‐) 83 and 9‐methoxyirispurinol 84 (Figure 9). The new rotenoid crocetenone 85 (9,11‐dihydro‐10‐methoxy‐) was also isolated from the methanolic extract of I. crocea rhizomes [ref. 86]. These compounds, which are absent in other Iris taxa, may therefore represent a distinctive chemotaxonomic feature of ser. Spuriae within subg. Limniris.
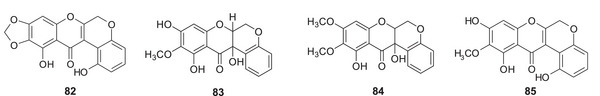
These rare flavonoid derivatives are mostly restricted to the subgenus Limniris, especially in species exposed to abiotic stress. Their presence suggests potential adaptive significance; however, their ecological functions and therapeutic potential remain largely speculative and should be addressed in future targeted bioactivity studies.
Anthocyanins
The variety of perianth colour in species of the Iris genus is largely determined by anthocyanins. These plants are characterised by the presence of both leucoanthocyanidins (leucocyanidin and leucodelphinidin) and a diverse array of anthocyanins (Figure 10). Common anthocyanin structures in Iris species include delphinidin 87, petunidin 86 and malvidin 90 derivatives, such as malvidin 3‐O‐(p‐coumaroylrutinoside)‐5‐glucosides, delphinidin 3‐O‐rutinoside‐5‐glucosides and peonidin 3‐O‐(p‐coumaroylrutinoside) (Figure 9). The most extensively studied anthocyanins have been isolated from I. ensata [ref. 93, ref. 94]. The aerial parts (flowers and leaves) of I. ensata, Iris laevigata, Iris setosa, Iris japonica and others contain malvidin derivatives, petunidin derivatives, peonidin, cyanidin and their glycosides [ref. 39]. Anthocyanins derived from pelargonidin have been identified in the flowers of I. dichotoma and I. domestica [ref. 95]. These compounds have not been reported in other Iris species.

Xanthones
The most common and well‐known C‐glycosylxanthone found in Iris species is mangiferin 92 (Figure 11). Mangiferin was first identified in the leaves of Mangifera indica L. (Anacardiaceae) [ref. 96] and has since been detected in several ferns and dicotyledonous plants across at least 28 genera in 19 families. Among monocotyledonous plants, mangiferin was initially discovered in the flowers of I. × germanica in 1963 and later in other Iris species [ref. 97].
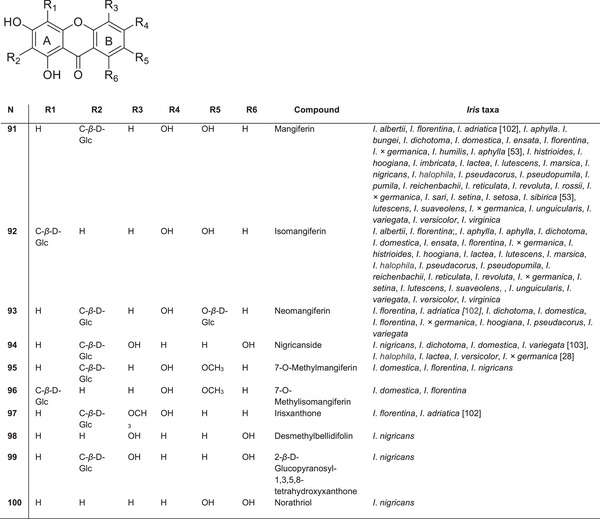
Mangiferin is widely distributed throughout the genus Iris. By the late 1980s, it had been identified in all 15 species of Iris from subgenus Iris [ref. 98]. It was also found in Iris humilis (sect. Psammiris) and Iris hoogiana (sect. Regelia). Within subg. Limniris, sect. Limniris, mangiferin is present in ser. Tripetalae, ser. Laevigatae, ser. Ensatae and ser. Unguiculares. In the subg. Hermodactyloides, mangiferin has been found in two species: I. histriodes and Iris reticulata [ref. 33, ref. 39].
In addition to mangiferin and its isomer isomangiferin 93, their derivatives, such as irixanthone, 5‐methyl ether, O‐methyl ether and O‐glucoside, are also commonly found. These compounds serve as important taxonomic markers for diploid species within the bearded Iris group. In 1973, Takemoto’s team first isolated irisxanthone from I. florentina [ref. 44]. Compared to mangiferin, irixanthone has a methoxy group at Position 5 and lacks a hydroxyl group at Position 7. Irisxanthone has also been identified in the leaves of I. florentina, I. adriatica and I. × germanica [ref. 39].
According to Iwashina and Mizuno [ref. 39], mangiferin has been identified in 47 species and subspecies of the Iris genus, whereas isomangiferin was found in 41. Among unusual C‐glucosyl xanthones, 6‐dehydrojacareubins were isolated as stress metabolites from I. × germanica leaves [ref. 80], but they have not been reported in other Iris species.
New xanthones, including the 1,3,5,8‐tetrahydroxy derivatives bellidifolin and desmethylbellidifolin, as well as nigricanside, were isolated from the rhizomes of I. nigricans [ref. 99]. Another compound, 2‐β‐d‐glucopyranosyl‐1,3,5,8‐tetrahydroxyxanthone, was also found. Mangiferin 7‐O‐β‐glucoside (neomangiferin) and nigricanside were also detected in I. dichotoma [ref. 58] and I. domestica [ref. 64], whereas 6′‐O‐acetyl mangiferin was found in the rhizomes of I. rossii [ref. 74]. Chinese researchers have indicated the chemotaxonomic significance of mangiferin for the Iridaceae family at multiple taxonomic levels, including tribe, subgenus, section and series [ref. 100]. For instance, the Irideae and Tigridae tribes can be distinguished from other Iridaceae tribes on their mangiferin content [ref. 31, ref. 101]. Overall, mangiferin and its derivatives are widely distributed across the Iris genus and demonstrate significant potential both as chemotaxonomic markers and as bioactive compounds with anti‐inflammatory, antimicrobial and antiviral properties.
Xanthones, especially mangiferin and its derivatives, are widely distributed across Iris species and hold both taxonomic and therapeutic significance. However, the impact of environmental factors on the accumulation of xanthones in Iris plants remains poorly understood and requires further study.
Benzophenones and Furans
Benzophenones serve as a key intermediate in xanthone biosynthesis and have been found in several Iris species. Iriflophenone is the most common form, occurring in I. adriatica [ref. 102], I. humilis, I. pumila, I. veriegata [ref. 103], I. florentina [ref. 104, ref. 105], I. halophila, I. spuria subsp. Carthaliniae, and I. versicolor [ref. 28], I. potaninii [ref. 56], I. scariosa [ref. 106] and I. domestica [ref. 107]. Other derivatives, such as 2,6,4′‐trihydroxy‐4‐methoxybenzophenone, have been identified in I. × germanica and I. halophila resinoids [ref. 66], as well as in I. adriatica rhizomes [ref. 102]. Additionally, 2,4′,6‐trihydroxy‐4‐methoxybenzophenone‐2‐O‐β‐d‐glucoside was isolated from I. × germanica rhizomes [ref. 64].
From I. domestica seeds, four new enamides, belamcandines A–D, were isolated and identified as 4a,8‐dihydroxy‐2,7‐dimethoxy‐1,4‐dioxo‐1,4,4a,9b‐tetrahydrodibenzofurans with two alkyl side chains at Positions 9 and 9b [ref. 108]. Subsequently, the sucrose derivatives belamcanoside A and B and the pyrrole derivative 4‐(2‐formyl‐5‐hydroxymethylpyrrol‐1‐yl) butyric acid were also isolated from the I. domestica seeds and I. spuria rhizomes [ref. 60, ref. 109], respectively. Overall, benzophenones and furan derivatives remain poorly studied. Given their restricted occurrence, they may serve as promising chemotaxonomic or pharmacological markers upon further investigation.
Quinones and Benzoquinones
One of the earliest reports of quinone isolation from Iris rhizomes was by Wong et al. [ref. 110]. They found 2‐hydroxy‐3‐octadecyl‐5‐methoxy‐1,4‐benzoquinone (irisoquin) and 3‐octadecyl‐5‐methoxy‐1,4‐benzoquinone (deoxyirisoquin) in Iris missouriensis. Irisoquin was later also found in the rhizomes of Iris kemaonensis [ref. 111] and in the seed oil of I. missouriensis [ref. 112]. Seven homologues, including irisquinone (pallason A), 2‐methoxy‐6‐[Z]‐pentadec‐8‐enyl‐1,4‐benzoquinone and 5‐hydroxy‐2‐methoxy‐6‐(pentadec‐7‐enyl)‐1,4‐benzoquinone, were found in the seed oils of I. pseudacorus, I. sibirica and I. missouriensis [ref. 112]. Pallason A, B (dihydroirisquinone) and C were isolated from I. lactea [ref. 113].
Several alkylated p‐benzoquinones have been reported in other species. For example, irisoquin A–F were identified in I. kemaonensis rhizomes [ref. 111], 3‐[(z)‐12′‐heptadecenyl]‐2‐hydroxy‐5‐methoxy‐1,4‐benzoquinone in the same species [ref. 114] and nepaloquinone A in I. × germanica rhizomes [ref. 115]. From I. bungei seeds and roots, novel derivatives such as belamcandaquinone N, 3‐hydroxyirisquinone, bungeiquinone and dihydrobungeiquinone were also isolated [ref. 116, ref. 117]. Other benzoquinones, including irisquin B, 3‐hydroxyirisquinone and 2‐acetoxy‐3,6‐dimethoxy‐1,4‐benzoquinone, were found in I. crocea, Iris suaveolens and I. × germanica, respectively [ref. 85, ref. 118, ref. 119].
Additionally, Tie et al. [ref. 120] described a new oligostilbene, vitisin A‐13‐O‐β‐d‐glucoside, alongside known oligostilbenes vitisin D, hopeaphenol and vitisin A, all from I. lactea seeds. The abundance of these quinones and oligostilbenes in rhizomes and seeds highlights their structural diversity, species specificity and potential roles in plant defence and stress adaptation, as well as their promising pharmacological applications.
Chromanes and Chromones
Chromane, also known as benzo‐γ‐dihydropyran, is a heterocyclic compound that serves as a structural scaffold for more complex molecules, including vitamin E (tocopherols and tocotrienols). For the first time, Dagvadorj’s working group in Mongolia [ref. 121] isolated five new compounds from the underground parts of I. tenuifolia, including an unusual macrolide, moniristenulide. Additionally, the chromone derivative 5,7‐dihydroxy‐6‐methoxychromone was identified in the rhizomes of I. unguicularis [ref. 122], further expanding the diversity of heterocyclic metabolites in the genus.
Chromanes and chromones are relatively rare in the Iris genus and have been reported in only a few taxa, often as minor components. Their limited distribution suggests potential chemosystematic significance, but comprehensive screening across more species is needed to clarify their taxonomic and biological relevance.
Benzenecarboxylic Acids, Ketones, Phenylpropanoids
Hydroxybenzoic acids are common precursors in the biosynthesis of flavonoids and isoflavonoids, particularly in Iris species. Among these, gallic acid, p‐hydroxybenzoic acid, protocatechuic acid, vanillic acid and syringic acid have been frequently identified. Hydroxycinnamic acids are also widely distributed and include ferulic acid, caffeic acid, p‐coumaric acid, trans‐cinnamic acid, chlorogenic acid, neochlorogenic acid and sinapic acid [ref. 28, ref. 29, ref. 30, ref. 31, ref. 32, ref. 33, ref. 34, ref. 35, ref. 36, ref. 37, ref. 38, ref. 39, ref. 40, ref. 41, ref. 42, ref. 43, ref. 44, ref. 45, ref. 46, ref. 47, ref. 48, ref. 49, ref. 50, ref. 51, ref. 52, ref. 53, ref. 54, ref. 55, ref. 56, ref. 57, ref. 58, ref. 59, ref. 60, ref. 61, ref. 62, ref. 63, ref. 64, ref. 65, ref. 66, ref. 67, ref. 68, ref. 69, ref. 70, ref. 71, ref. 72, ref. 73, ref. 74, ref. 75, ref. 76, ref. 77, ref. 78, ref. 79, ref. 80, ref. 81, ref. 82, ref. 83, ref. 84, ref. 85, ref. 86, ref. 87, ref. 88, ref. 89, ref. 90, ref. 91, ref. 92, ref. 93, ref. 94, ref. 95, ref. 96, ref. 97, ref. 98, ref. 99, ref. 100, ref. 101, ref. 102, ref. 103, ref. 104, ref. 105, ref. 106, ref. 107, ref. 108, ref. 109, ref. 110, ref. 111, ref. 112, ref. 113, ref. 114, ref. 115, ref. 116, ref. 117, ref. 118, ref. 119, ref. 120, ref. 121, ref. 122, ref. 123, ref. 124, ref. 125, ref. 126]. These phenolic acids contribute to the structural diversity of downstream secondary metabolites and may play roles in plant defence, stress response and pigmentation (Tables 2 and 3).
TABLE 2: The derivatives of phenolcarboxylic acids identified in Iris taxa.
| Compound | Iris taxa |
|---|---|
| Vanillic acid | I. bungei [ref. 90]; I. dichotoma, I. humilis, I. lactea, I. tenuifolia [ref. 125]; I. halophila, I. versicolor, I. lactea, I. spuria subsp. Carthaliniae, I. × germanica [ref. 28]; I. spuria [ ref. 60] |
| Vanillic acid 4‐O‐β‐d‐glucopyranoside | I. spuria [ref. 60] |
| Syringic acid | I. bungei [ref. 90]; I. humilis, I. lactea [ref. 125]; I. schachtii [ref. 126] |
| Glucosyringic acid | I. spuria [ref. 60] |
| Gallic acid | I. domestica [ref. 59]; I. pseudacorus, I. sibirica, I. × germanica, I. variegata [ref. 57]; I. aphylla [ref. 53]; I. lactea [ref. 125]; I. schachtii [ref. 126] |
| Protocatechuic acid | I. × germanica [ref. 127]; I. dichotoma, I. humilis, I. bungei, I. lactea, I. tenuifolia [ref. 125]; I. schachtii [ref. 126]; I. halophila, I. versicolor, I. lactea, I. spuria subsp. carthaliniae, I. × germanica [ref. 28] |
| 3‐Hydroxybenzoic acid | I. bungei, I. tenuifolia [ref. 125] |
| 4‐Hydroxy benzoic acid | I. × germanica [ref. 127]; I. dichotoma, I. humilis, I. bungei, I. lactea, I. tenuifolia [ref. 125]; I. schachtii [ref. 126]; I. halophila, I. versicolor [ref. 28]; I. domestica [ref. 59] |
| Benzoic acid | I. schachtii [ref. 126] |
| Caffeic acid | I. aphylla [ref. 124]; I. pseudacorus [ref. 123]; I. sibirica [ref. 52]; I. × germanica, I. variegata [ref. 53]; I. halophila [ref. 127] |
| Chlorogenic acid | I. aphylla [ref. 124]; I. pseudacorus, I. sibirica [ref. 52]; I. germanica [ref. 127] |
| Neochlorogenic acid | I. aphylla [ref. 124]; I. pseudacorus, I. sibirica, I. × germanica [ref. 52] |
| Ferulic acid | I. aphylla [ref. 124]; I. pseudacorus, I. sibirica [ref. 52]; I. × germanica [ref. 127]; I. dichotoma, I. humilis, I. lactea [ref. 125] I. halophila, I. versicolor, I. lactea, I. spuria subsp. carthaliniae, I. × germanica [ref. 28]; I. nigricans [ref. 45] |
| p‐Coumaric acid | I. pseudacorus, I. sibirica, I. × germanica [ref. 52]; I. humilis, I. lactea [ref. 125]; I. schachtii [ref. 126] |
| trans‐Cinnamic acid | I. pseudacorus, I. sibirica, I. × germanica, I. halophila [ref. 52]; I. dichotoma, I. humilis, I. bungei, I. lactea, I. tenuifolia [ref. 125]; I. schachtii [ref. 126]; I. halophila, I. versicolor, I. lactea, I. spuria subsp. carthaliniae, I. × germanica [ref. 28] |
| Sinapic acid | I. schachtii [ref. 126] |
| Shikimic acid | I. domestica [ref. 59] |
TABLE 3: Acetophenones derivatives identified in Iris taxa.
| 3‐Hydroxy‐5‐methoxyacetophenone | I. × germanica [ref. 88] |
|---|---|
| Apocynin (4‐hydroxy‐3‐methoxyacetophenone) | I. × germanica [ref. 119]; I. pallida [ref. 132]; I. suaveolens [ref. 129]; I. nigricans [ref. 45]; I. tectorum [ref. 133, ref. 134]; I. × germanica [ref. 64]; I. tectorum [ref. 135]; I. domestica [ref. 136]; I. pallida [ref. 132]; I. tingitana ([131]; I. crocea [ref. 86] |
| Androsin (4‐O‐β‐d‐glucopyranosyl‐acetovanillone) | I. postii [ref. 137]; I. tectorum [ref. 133]; I. × germanica [ref. 64]; I. postii [ref. 137]; I. tectorum [ref. 135] |
| Tectorusid | I. tectorum [ref. 134]; I. domestica [ref. 138] |
| Diapocynin | I. tectorum [ref. 134] |
| Apocynin‐4‐O‐β‐d‐(6′‐O‐syringyl) glucopyranoside | I. tectorum ([135] |
| Scrophenoside C‐7‐ethylether | I. tectorum [ref. 135] |
| Apocynin‐4‐O‐β‐d‐xylopyranoside | I. tectorum [ref. 139] |
The phenylpropanoid glycoside e‐coniferin has been identified in I. spuria [ref. 128]. In additional, an unusual phenylpropanoid containing an epoxy group, cis‐epoxyconiferyl alcohol, was isolated from the fresh rhizomes of I. suaveolens, alongside coniferaldehyde and p‐hydroxyacetophenol [ref. 129].
Acetovanillone (apocynin), its β‐d‐glucoside isorhizone and tectoroside were identified in the roots of I. tectorum [ref. 90]. Apocynin has also been found in I. × germanica, I. suaveolens, I. nigricans, I. susiana and I. tingitana [ref. 45, ref. 46, ref. 129, ref. 130, ref. 131]. Overall, benzenecarboxylic acids and phenylpropanoids are widespread in Iris species, though their ecological roles remain largely unexplored.
Phytosterols
Steroids are also present in the rhizomes of Iris species, though they are less abundant than phenolic compounds. Among the identified steroidal compounds, β‐sitosterol has been reported in I. tenuifolia [ref. 47], I. × germanica [ref. 64], I. lactea [ref. 91], I. susiana [ref. 140], I. suaveolens [ref. 118] and I. domestica [ref. 141]. Other steroids include daucosterol [ref. 64], stigmasterol and stigmasterol‐3‐O‐β‐d‐glucopyranoside [ref. 88] in I. × germanica, as well as 7‐β‐hydroxystigmast‐4‐en‐3‐one in I. suaveolens [ref. 118]. Isoarborinol and stigmasterol were isolated from the underground parts of I. confusa [ref. 6]. Daucosterol was isolated from I. domestica [ref. 141] and I. lactea [ref. 91]. Tristearoyl‐sn‐glycerol, 1‐margaroyl‐2‐lauroyl‐3‐palmitoyl‐sn‐glycerol and 1,2‐distearoyl‐sn‐glycerol were isolated from I. marsica [ref. 67]. Stigmasterol was isolated from I. susiana [ref. 140]. A complete overview of the phytosterols isolated or identified in Iris species is provided in Table 4.
TABLE 4: Stilbenes identified in Iris taxa.
| Compound | Iris taxa |
|---|---|
| Stigmasterol | I. × germanica [ref. 88]; I. susiana [ref. 46]; I. cathayensis [ref. 82]; I. songarica [ref. 142] |
| Stigmasterol‐3‐O‐β‐d‐glucopyranoside | I. × germanica [ref. 88] |
| β‐Sitosterol | I. susiana [ref. 46]; I. tenuifolia [ref. 47]; I. suaveolens [ref. 129]; I. lactea [ref. 91]; I. domestica [ref. 141]; I. adriatica [ref. 104]; I. leptophylla [ref. 55]; I. songarica [ref. 142]; I. tectorum [ref. 133]; I. × germanica [ref. 64]; I. florentina [ref. 44]; I. nigricans [ref. 45] |
| Dausterol | I. domestica [ref. 59]; I. lactea [ref. 91]; I. halophila var. sogdiana [ref. 106]; I. cathayensis [ref. 82]; I. leptophylla [ref. 55]; I. songarica [ref. 142]; I. tectorum [ref. 133]; I. × germanica [ref. 64] |
| Resveratrol | I. hookeriana [ref. 143]; I. dichotoma [ref. 58]; I. domestica [ref. 144]; I. halophila [ref. 145]; I. clarkei [ref. 146] |
| Resveratroloside (trans‐resveratrol 4′‐O‐β‐d‐glucuronide) | I. hookeriana [ref. 143] |
| trans‐Resveratrol 3‐O‐β‐glucoside | I. tingitana [ref. 60] |
| Resveratrol 3,4′‐O‐di‐β‐d‐glucopyranoside | I. postii [ref. 137] |
| Piceid | I. hookeriana [ref. 143]; I. × germanica [ref. 85] |
| Ampelopsin B | I. clarkei [ref. 146] |
| (+)‐α‐Viniferin | I. clarkei [ref. 146] |
| (−)‐trans‐ε‐Viniferin | I. halophila [ref. 145]; I. lactea [ref. 147]; I. postii [ref. 137] |
| trans‐ε‐Viniferin‐13b‐β‐d‐glucopyranose | I. lactea [ref. 147] |
| cis‐ε‐Viniferin‐11a,13b‐di‐O‐β‐d‐glucopyranoside | I. lactea [ref. 147] |
| γ‐2‐Viniferin | I. halophila [ref. 145] |
| Halophilol A | I. halophila [ref. 145] |
| Halophilol B | I. halophila [ref. 145] |
| Vatalbinoside C | I. lactea [ref. 147] |
| cis‐Vitisin B‐13b‐O‐β‐d‐glucopyranoside | I. lactea [ref. 147] |
| cis‐Vitisin B | I. lactea [ref. 147] |
| cis‐Vitisin C | I. lactea [ref. 147] |
| Vitisin A | I. lactea [ref. 147] |
| Vitisin B | I. lactea [ref. 147] |
| (−)‐Hopeaphenol | I. domestica [ref. 109] |
| 7β‐Hydroxystigmast‐4‐en‐3‐one | I. suaveolens [ref. 129] |
| Tingitanol A | I. tingitana [ref. 60] |
| Tingitanol B | I. tingitana [ref. 60] |
Some species in the Iris genus produce both monomeric and oligomeric stilbenes. Resveratrol has been identified in I. dichotoma [ref. 58], I. domestica [ref. 144], Iris hookeriana [ref. 143], I. halophila [ref. 145] and Iris clarkei [ref. 146]. The resveratrol glucosides piceid (3‐O‐β‐d‐glucoside of resveratrol) and resveratroloside were isolated from the underground organs of I. hookeriana [ref. 143], with piceid also detected in I. × germanica [ref. 85]. Two stilbenes containing a γ‐lactam unit [(+) and (−)] were isolated from the rhizomes of I. domestica [ref. 65]. Additionally, resveratrol‐3‐glucoside and two new stilbene glycosides, tingitanols A and B, were isolated from I. tingitana (subg. Xiphium) [ref. 60].
Monomeric stilbene halophylol A and the tetrameric stilbene halophylol B were found in the seeds of I. halophila (subg. Limniris, sect. Limniris), along with ε‐viniferin and γ‐2‐viniferin [ref. 145]. Two resveratrol‐type oligostilbenes, ampelopsin B and α‐viniferin, were isolated from the seeds of I. clarkei [ref. 146]. Ten stilbene oligomers, including vatalbinoside C, cis‐vitisin B‐13b‐O‐β‐d‐glucopyranoside, trans‐ε‐viniferin‐13b‐β‐d‐glucopyranose, cis‐ε‐viniferin‐11a,13b‐di‐O‐β‐d‐glucopyranoside, trans‐ε‐viniferin, cis‐vitisin B and C and vitisin A and B, were also found in I. lactea seeds [ref. 147]. Furthermore, trans‐ε‐viniferin and resveratrol 3,4′‐O‐di‐β‐d‐glucopyranoside were isolated from Iris postii [ref. 137], and the stilbenoid (−)‐hopeaphenol, a hydroxylated derivative of stilbene, was isolated from I. domestica seeds [ref. 109]. These are the only examples of the isolation of oligomeric stilbenes from the Iridaceae family.
Phytosterols have been sparsely identified in Iris species, mainly in rhizomes, and have not been comprehensively assessed. Comparative profiling across species and developmental stages may reveal taxon‐specific sterol patterns and support their evaluation as bioactive or chemotaxonomic markers.
Alkaloids
Data on the presence of alkaloids in Iris species are extremely limited. The notable exception is the study by Xie et al. [ref. 148], who investigated the ethanolic extract of I. × germanica rhizomes and identified several alkaloid compound structures. These included 1,2,3,4‐tetrahydro‐c‐carboline‐3‐carboxylic acid, S‐(−)‐methyl‐1,2,3,4‐tetrahydro‐9H‐pyrido[3,4‐b]indole‐3‐carboxylate, (1R,3R)‐methyl‐1‐methyl‐2,3,4,9‐tetrahydro‐1H‐pyrido[3,4‐b]indole‐3‐carboxylate, (1S,3R)‐methyl‐1‐methyl‐2,3,4,9‐tetrahydro‐1H‐pyrido‐[3,4‐b]indole‐3‐carboxylate, 4‐(9H‐c‐carbolin‐1‐yl)‐4‐oxobut‐2‐enoic acid methyl ester, 2‐(furan‐2‐yl)‐5‐(2,3,4‐trihydroxybutyl)‐1,4‐diazine, 3‐c‐d‐ribofuranosyluracil,6‐hydroxymethyl‐3‐pyridinol and 2‐amino‐1H‐imidazo[4,5‐b]pyrazine. The findings of the study represent the first detailed characterisation of alkaloids in Iris genus, particularly in I. × germanica. Given the scarcity of such data, further phytochemical investigations across additional Iris species are warranted to explore the potential chemotaxonomic relevance and potential pharmacological properties of their alkaloid profiles.
Terpenoids
Iridals constitute a distinct family of triterpenoids that are the primary lipid components of Iris rhizomes. These compounds are typically monocyclic, featuring a modified ring A with various degrees of unsaturation and/or hydroxylation, or they may occur as bicyclic C31‐triterpenoids. The latter class arises through cyclisation of the homofarnesyl side chain, initiated by the transfer of a methyl group from S‐adenosylmethionine to the terminal double bond. The resulting cycloiridals act as biosynthetic precursors of dihydroirones and irones [ref. 149, ref. 150]. Notably, no single terpenoid has been reported to occur in more than one Iris taxon. Therefore, the information in Table 5 is arranged according to species, from Iris taxa to the corresponding identified compounds.
TABLE 5: Iridal‐type triterpenoids identified in species of the genus Iris.
| Iris taxa | Triterpenoid | Refs. |
|---|---|---|
| I. delavayi | 6S,10R,11R)‐18,19‐Epoxy‐10‐deoxy‐17‐hydroxyiridal, (2(7)E,6S,10R,11R)‐10‐Deoxy‐17‐hydroxyirida, (2(7)E,6S, 10R, 11R)‐18,19‐Epoxy‐10‐deoxy‐17‐hydroxyiridal | [ref. 151] |
| I. confusa | Spirioiridoconfal A‐C, isobelamcandal, 17‐hydroxyl‐27‐ene‐iridal | [ref. 152] |
| I. forrestii | Forrestin A, forrestin B | [ref. 153] |
| I. foetidissima | 6R,10S,11S,14S,26R)‐26‐Hydroxy‐15‐methylidenespiroirid‐16‐en | [ref. 150] |
| I. tectorum | Iritectol G, Iritectol H, 19‐epiiritectol H | [ref. 154] |
| I. tectorum | Iritectorol A, iritectorol B, iridobelamal A, isoiridogermanal | [ref. 155] |
| I. tectorum | Iridotectoral A, iridotectoral B, 28‐deacetylbelamcandal, (6R,10S,11R)‐26‐x‐hydroxy‐(13R)‐oxaspiroirid‐16‐enal | [ref. 156] |
| I. tectorum | Iridotectoral C, Iridotectoral D | [ref. 157] |
| I. tectorum | Polycycloiridals A–D | [ref. 158] |
| I. tectorum | Spirioiridotectals A−F | [ref. 158] |
| I. domestica | Iridobelamal B | [ref. 157] |
| I. domestica | Iridobelamal A, 28‐deacetylbelamcandal, (6R,10S,11R)‐26‐x‐hydroxy‐(13R)‐oxaspiroirid‐16‐enal, 6R,10S,11S,14S,26R)‐26‐hydroxy‐15‐methylidene‐spiroirid‐16‐enal, 16‐O‐acetyl‐iso‐iridogermanal, isoiridogermanal | [ref. 156] |
| I. domestica | Polycycloiridals K–T | [ref. 159] |
| I. domestica | Belamchinenin A | [ref. 160] |
| I. domestica | Dibelamcandal A | [ref. 161] |
| I. domestica | 16‐Acetyliridal A, 16‐acetyliridal B | [ref. 162] |
| I. tenuifolia | Arborinone | [ref. 47] |
| I. variegata | 21‐Hydroxyiridal, 21‐hydroxy‐10‐deoxyiridal, 16‐hydroxyiridal, 22‐methyl‐γ‐cycloiridal, 23‐hydroxyiridal | [ref. 149] |
| I. × germanica | Isoiridogermanal, 16‐O‐acetylisoiridogermanal, α‐iridogermanal, γ‐iridogermanal, α‐dehydroiridogermanal, iridial, iriflorental, iripallidal, irisgermanicals A–C | [ref. 163, ref. 164] |
| I. × germanica | Irigermanal, iridogermanal | [ref. 165] |
| I. × germanica | Iristectoron K | [ref. 51] |
| I. crocea | Iridalglycoside 5b, iridalglycoside 7 | [ref. 85] |
| I. spuria | Iridalglycoside 5b/6c, iridalglycoside 7/8 | [ref. 85] |
| I. pseudacorus | Irispseudoacorins A–D, 16‐formyl‐isoiridogermananl, 3‐formyl‐isoiridogermananl, iridojaponals A‐B, isoiridogermananl; iridobelamal A; 17(R)‐hydroxyliridal | [ref. 166] |
| I. versicolor | 22,23‐Dihydro‐22‐methyleneiridal, 26‐hydroxyiridal, 6‐methylhept‐5‐en‐2‐one, (4RS)‐4‐hydroxy‐6,1O‐dimethylundeca‐5,9‐dien‐2‐one, 17ξ,26‐dihydroxyiridal | [ref. 167] |
| I. sibirica | Iridal, spirocyclic hemiacetal, 16ξ‐hydroxyiridal, 26‐hydroxyiridal, 17ξ,26‐dihydroxyiridal, 17ξ‐hydroxyiridal, 10‐deoxy‐l7ξ‐hydroxyirid | [ref. 167] |
| I. sibirica | (6R,10S, 11S)‐17,29‐Didehydroiridal | [ref. 168] |
Pentacyclic triterpenes, such as ursolic acid, betulin, betulonic acid and betulone, have been identified in the rhizomes of I. domestica [ref. 141]. Arborinone, a 3‐oxygenated pentacyclic triterpene, was isolated from I. tenuifolia [ref. 47]. Although most reviews of Iris phytochemistry emphasise isoflavonoids and xanthones, triterpenoids, particularly those occurring in the rhizomes, also present a promising but understudied class of compounds.
Terpenoids, especially triterpenoids and iridals, constitute major secondary metabolites in Iris rhizomes. Although some of them have demonstrated cytotoxic, anti‐inflammatory and antimicrobial activities, their structural diversity and biological functions remain largely unexplored. Comprehensive metabolomic and genomic investigations are therefore required to clarify their biosynthetic origins and assess their potential therapeutic applications.
Essential Oil
The industrial production of Iris essential oil for perfumery, at a concentration of about 0.1%–2.0%, began in 1912 at the Santa Maria Novella factory in France. Initial production volume was 10 kg per year. The cultivation of Iris species for industrial purposes, however, dates back to the mid‐19th century in Italy [ref. 169]. Today, only a few regions worldwide cultivate Iris species for use as a plant raw material in the perfume industry: I. pallida, I. × germanica and I. florentina in Tuscany and Florence (Italy); I. pallida in Landes (France); and I. × germanica in Marrakech, in the foothills of the Atlas Mountains (Morocco).
Orris oil, valued for its characteristic violet fragrance, is obtained from the rhizomes of I. florentina, I. × germanica and I. pallida. The crude extract appears as a semi‐solid yellow mass known as Iris concrete. Essential oil have also been identified in the rhizomes and leaves of several Iris species, including I. persica [ref. 170], I. variegata [ref. 149], I. bulleyana [ref. 171], I. aphylla [ref. 172], I. medwedewii, I. spuria subsp. carthaliniae [ref. 173], I. versicolor, I. graminea, I. halophila [ref. 174], I. haussknechtii, I. susiana [ref. 175], I. nigricans [ref. 176], I. taochia [ref. 177], I. planifolia [ref. 178], I. pseudopallida, I. illyrica and I. adriatica [ref. 179].
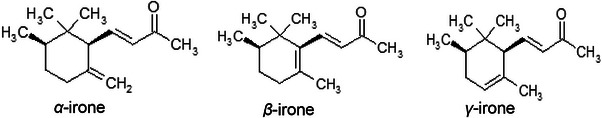
The essential oils of Iris species contain a complex mixture of volatile organic compounds, including monoterpenes, sesquiterpenes, diterpenes, triterpenes, fatty acids, aliphatic hydrocarbons and aldehydes. The monoterpene ketone α‐irone and the triterpenoid squalene have been identified in all analysed samples, making them potential chemotaxonomic and chemosystematic markers for the genus Iris [ref. 180]. α‐Irone, which accounts for 10%–20% of the essential oil and provides its characteristic violet scent, occurs in three structural and stereoisomeric forms: α‐, β‐ and γ‐irone (Figure 12).
Among the components of Iris essential oils, various norterpenoids, such as β‐ionone‐5,6‐epoxide, β‐ionone, trans‐2,6‐γ‐irone, β‐isometilionone and β‐damascenone, have been identified. Other constituents include neophytadiene, eugenol, α‐terpineol, germacrene D, terpinen‐4‐ol, hexahydrofarnesylacetone, farnesylacetone, phenylacetaldehyde, geranylacetone and 2‐methoxy‐4‐vinylphenol. Essential oils from Iris species are also characterised by a high proportion of myristic acid (45%–85%), as well as other fatty acids (caprylic, capric, lauric, palmitic etc.) and their esters.
Essential oils obtained from Iris species, especially from the rhizomes, contain unique aromatic compounds such as irones, which are of high value in perfumery and traditional medicine. However, the chemical composition of these oils varies considerably among species and is influenced by environmental conditions, developmental stage and extraction methods. Despite their long‐standing commercial significance, phytochemical profiling of Iris essential oils remains incomplete for many taxa. Comprehensive metabolomic studies employing standardised analytical protocols and broader taxonomic coverage are needed to fully elucidate their chemical diversity, biological activities and potential for industrial and pharmacological applications.
Organic and Amino Acids
Various organic acids have been identified in Iris species, predominantly in the aerial parts. The chemical composition of the carboxylic acids has been studied in the rhizomes of I. spuria subsp. carthaliniae and I. paradoxa [ref. 181], as well as in the leaves and rhizomes of I. graminea and I. halophila [ref. 182], and in the leaves of I. aphylla, I. × germanica, I. halophila and I. variegata [ref. 183]. Lactic acid was also found in I. marsica [ref. 67]. These compositions include both short‐ and long‐chain carboxylic acids, such as monocarboxylic, dicarboxylic and tricarboxylic acids, as well as saturated and unsaturated fatty acids.
For the first time, six previously undescribed long‐chain fatty acid esters and isoflavone glycosides were isolated from the rhizomes of I. domestica [ref. 184]. Asghar et al. [ref. 185] identified 11 compounds in the petroleum ether extract of I. × germanica rhizomes, with hexadecanoic and octadecenoic acid methyl esters being the most abundant. Another study [ref. 186] reported 23 compounds in I. × germanica rhizomes, including methyl esters of octadecanoic, palmitic and linolenic acids, as well as lauric, oleic and caprylic acids, among other derivatives. Additional analyses have been conducted on I. sibirica leaves and rhizomes [ref. 187], I. × germanica, Iris barnumiae, Iris bostrensis and Iris aurantica [ref. 188] and I. taochia [ref. 177]. Notably, myristic acid dominated in some species (61%–80%).
Typically, Iris leaves exhibit a higher content of unsaturated fatty acids (such as oleic, palmitoleic, linoleic and linolenic acids) than rhizomes, due to their elevated levels of polyunsaturated fatty acids and a lower proportion of saturated (e.g., palmitic, lauric and myristic) and monounsaturated fatty acids. Conversely, rhizomes, as a rule, accumulate more saturated fatty acids while containing less unsaturated ones.
Pyroglutamic acid was found in I. aphylla rhizomes, whereas alanine, valine, isoleucine, proline, serine, aspartic acid and pyroglutamic acid were found in I. variegata rhizome extract [ref. 189]. Phenylalanine, isoleucine and valine were identified in the rhizomes of I. marsica [ref. 67] and l‐tryptophan was present in the rhizomes of I. postii [ref. 137].
The diversity of carboxylic and amino acids across different species and organs of the Iris genus, as well as under various environmental conditions, highlights its value as a source of both primary and secondary metabolites. The organ‐specific distribution pattern, whereby rhizomes accumulate more saturated fatty acids and leaves are enriched in unsaturated forms, underlines the ecological and physiological specialisation of these plants. Continued exploration of Iris species phytochemistry reflects the ecological and physiological specialisation of these plants.
Organic and amino acids are essential primary metabolites in Iris species, contributing to plant physiology, stress responses and serving as precursors for secondary metabolism. However, their profiles have not been systematically analysed across species or organs, and their chemotaxonomic or ecological significance remains largely unexplored. Targeted metabolomic investigation could elucidate their potential roles in species adaptation, environmental interaction and metabolic specialisation within the genus.
The Effect of Environmental Factors
The composition of primary and secondary plant metabolites is significantly influenced by environmental factors. Various Iris species have been analysed depending on their habitat characteristics, including climate, light intensity, precipitation, soil composition and other ecological parameters. The greatest changes in metabolite profile, particularly in the concentration of isoflavones, hydroxycinnamic acids and xanthones, have been observed among Iris species.
Using I. variegata as an example [ref. 190], it was found that the accumulation of these compounds is highest in spring and decreases towards autumn. Plants grown under high light intensity exhibited elevated levels of chlorogenic acid and mangiferin glucoside, whereas caffeic acid, luteolin and naringenin showed the opposite trend. The developmental stage of the growing season strongly influenced metabolite profiles, with almost all secondary metabolites reaching their maximum concentrations in spring and declining progressively during summer and autumn until the end of the vegetative period.
Our previous study [ref. 52] demonstrated that the levels of phenolic compounds in Iris rhizomes from subgenus Limniris and Iris were significantly affected by soil nutrient composition. Specifically, the contents of phosphorus and potassium in the soil positively influenced phenolic accumulation, whereas nitrogen levels had no significant effect. Meteorological factors in Ukraine, Lithuania and Latvia had limited impact overall; however, sunshine duration correlated positively, whereas rainfall correlated negatively with phenolic content.
Experimental cultivation of Iris species under monocomponent mineral nutrition [ref. 191] further confirmed the sensitivity of their secondary metabolism. it is a positive impact on the biosynthesis and accumulation of specific isoflavonoids (e.g., tectoridin, nigricin d‐glucoside, genistin, irisectorigenin B, nigricin, irigenin, irisolidone) and the xanthone mangiferin was observed in rhizomes. Additionally, an increase in chlorogenic acid content was recorded in leaves. These findings indicate that Iris phenylpropanoid metabolism responds strongly to mineral nutrition, suggesting that cultivation strategies can be optimised to enhance the production of bioactive compounds.
Although Iris species display high ecological plasticity and adaptability to diverse habitats, the environmental factors of their metabolic biosynthesis remain poorly studied. Current evidence suggests that soil composition, climate factors and biotic stressors can significantly influence both the qualitative and quantitative profiles of secondary metabolites. However, comprehensive eco‐chemical correlation studies are still lacking. Future research should focus on evaluating the influence of environmental variables on Iris metabolite composition across different geographical regions to better understand species resilience and support the sustainable cultivation of plants with high pharmacological potential.
Perspectives and Future Directions
The genus Iris represents a diverse and important group within the Iridaceae family, characterised by a wide global distribution and a long history of ethnomedicinal use [ref. 192]. This review consolidates current data on the primary and secondary metabolites identified in more than 90 taxa, with particular emphasis on isoflavonoids, flavonoids, xanthones and rare compound classes, such as peltogynoids and coumaronochromones. Many of these metabolites exhibit potent pharmacological activities, including antimicrobial, antitumour, antioxidant, antiviral and anti‐inflammatory properties. These findings highlight Iris species as promising candidates for natural product‐based drug discovery.
The medicinal potential of Iris plants offers valuable opportunities for application in the pharmaceutical, food and cosmetic industries, provided that rigorous quality control of raw materials is maintained throughout cultivation, harvesting and processing [ref. 193]. However, not all species within the genus have been studied in detail. Continued efforts to isolate novel bioactive compounds and evaluate their pharmacological properties are warranted, as such studies may yield new therapeutic agents and expand industrial applications.
Despite considerable advances in the phytochemical characterisation of Iris species, significant knowledge gaps remain. Of the more than 300 known species, only about one‐third have been chemically characterised, and the majority of studies have focused on a limited number of taxa, primarily I. × germanica, I. tectorum and I. dichotoma. Moreover, the influence of ecological, geographical and environmental factors on metabolite profiles remains insufficiently explored. Future research employing advanced metabolomic, transcriptomic and genomic tools will be essential for elucidating biosynthetic pathways, identifying species‐specific chemical markers and understanding the ecological and evolutionary determinants of secondary metabolism within the genus.
Despite extensive phytochemical research, many Iris species remain poorly characterised, especially endemic and geographically restricted taxa. Furthermore, the effects of environmental and ecological factors on metabolite composition are still insufficiently studied. Future investigations should therefore focus on:
- expanding metabolomic profiling to include lesser‐known Iris species;
- elucidating the influence of ecological and geographical variation on metabolite accumulation of secondary metabolites;
- integrating omics‐based approaches (e.g., genomics, transcriptomics and metabolomics) to clarify biosynthetic pathways;
- exploring the pharmacological activities of both crude extracts and isolated constituents using relevant biological models.
Overall, Iris species emerge as promising plants for advancing the discovery of natural products and chemotaxonomy. Continued interdisciplinary efforts will pave the way for its sustainable use in medicine, cosmetics and biotechnology, while also helping to conserve plant biodiversity.
Author Contributions
Olha Mykhailenko: conceptualisation, data analysis, writing – original draft preparation, writing – review and editing. Liudas Ivanauskas: data analysis, writing – review and editing. Victoriya Georgiyants: data analysis, writing – original draft preparation, writing – review and editing. Zigmantas Gudžinskas: writing – original draft preparation, writing – review and editing. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Exploring the Use of Iris Species: Antioxidant Properties, Phytochemistry, Medicinal and Industrial Applications,”. Antioxidants, 2022. [PubMed]
- Shedding the Light on Iridaceae: Ethnobotany, Phytochemistry and Biological Activity,”. Industrial Crops and Products, 2016. [DOI]
- Flavonoids of the Genus Iris (Iridaceae),”. Mini—Reviews in Medicinal Chemistry, 2010. [DOI | PubMed]
- Anti‐Viral and Anti‐Inflammatory Isoflavonoids From Ukrainian Iris aphylla Rhizomes: Structure‐Activity Relationship Coupled With ChemGPS‐NP Analysis,”. Planta Medica, 2023. [DOI | PubMed]
- Iris setosa Pall. Ex Link Extract Reveals Amoebicidal Activity Against Acanthamoeba castellanii and Acanthamoeba polyphaga With Low Toxicity to Human Corneal Cells,”. Microorganisms, 2024. [PubMed]
- Correlation Between Secondary Metabolites of Iris confusa Sealy and Iris pseudacorus L. and Their Newly Explored Antiprotozoal Potentials,”. BMC Complementary Medicine and Therapies, 2023. [DOI | PubMed]
- Investigation of the Profile of Dry Extracts of Iris hungarica Leaves and Rhizome to Determine the Cardioprotective Activity in the Rat Model of Doxorubicin Cardiomyopathy,”. ScienceRise: Pharmaceutical Science, 2024. [DOI]
- Dyeing Potential of Linen Fabric With Iris Flower Extracts,”. ProEnvironment, 2018
- Identification of Floral Scent Profiles in Bearded Irises,”. Molecules (Basel, Switzerland), 2019. [DOI | PubMed]
- 10 POWO , “Iris Tourn. ex L.,” in Plants of the World Online (Kew Gardens, 2023), https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:326330‐2.
- Some Aspects of the ‘Juno Group’ of Irises,”. Annals of botany, 2000
- Can a Rainbow Consist of a Single Colour? A New Comprehensive Generic Arrangement of the ‘Iris sensu latissimo’ Clade (Iridaceae), Congruent With Morphology and Molecular Data,”. Phytotaxa, 2015. [DOI]
- The Genus Iris L. s. l. (Iridaceae) in the Mountains of Central Asia Biodiversity Hotspot,”. Plant Diversity of Central Asia, 2022
- 14 B. Mathew , ‘The Iris’, 2nd ed. (Batsford Ltd., 1989).
- Molecular Phylogenetics of the Juno Irises, Iris Subgenus Scorpiris (Iridaceae), Based on Six Plastid Markers,”. Botanical Journal of the Linnean Society, 2011. [DOI]
- Notes on Taxonomy and Nomenclature of Juno Irises (Juno, Iridaceae),”. Phytotaxa, 2018. [DOI]
- A New System of the Genus Iris (Iridaceae),”. Botanicheskiĭ Zhurnal, 2009
- Subgeneric Classification in Iris Re‐Examined Using Chloroplast Sequence Data,”. Taxon, 2011. [DOI]
- Sectional Relationships in the Eurasian Bearded Iris (subgen. Iris) Based on Phylogenetic Analyses of Sequence Data,”. Systematic Botany, 2017. [DOI]
- At Least 23 Genera Instead of One: The Case of Iris L. s.l. (Iridaceae),”. PLoS ONE, 2014. [DOI | PubMed]
- The Royal Irises (Iris subg. Iris Sect. Oncocyclus): Plastid and Low‐Copy Nuclea Data Contribute to an Understanding of Their Phylogenetic Relationships,”. Taxon, 2016. [DOI]
- Sectional Relationships in the Eurasian Bearded Iris (subgen. Iris) Based on Phylogenetic Analyses of Sequence Data,”. Systematic Botany, 2017. [DOI]
- Molecular Study of Selected Taxonomically Critical Taxa of the Genus Iris L. From the Broader Alpine‐Dinaric Area,”. Plants, 2020. [DOI | PubMed]
- Iris: A Significant Element of the Mediterranean Landscape,”. Annals of Botany, 2000
- Iris Linnaeus,”. FOC, 2000
- Iris L. In: Flora of North America North of Mexico. Vol. 26. Magnoliophyta: Liliidae: Liliales and Orchidales,”. Economic Botany, 2003
- A Review on Phytochemical and Therapeutic Potential of Iris germanica ,”. Journal of Pharmacy and Pharmacology, 2021. [DOI | PubMed]
- Phytochemical Composition and In Vitro Biological Activity of Iris spp. (Iridaceae): A New Source of Bioactive Constituents for the Inhibition of Oral Bacterial Biofilms,”. Antibiotics, 2020. [DOI | PubMed]
- Biflavonoids, Quinones and Xanthones as Rare Chemical Markers in the Family Iridaceae. Zeitschrift Für Naturforschung C,”. Phytochemistry, 1985
- Correlations Between Phenolic Patterns and Tribal Classification in the Family Iridaceae,”. Phytochemistry, 1986. [DOI]
- Flavonoid and Xanthone Patterns in Bearded Iris Species and the Pathway of Chemical Evolution in the Genus,”. Biochemical Systematics and Ecology, 1997. [DOI]
- Isoflavonoids and Related Compounds,”. Natural Product Reports, 1998. [DOI]
- Flavonoids of the Genus Iris; Structures, Distribution and Function: Review,”. Annals of the Tsukuba Botanical Garden, 1998
- Isoflavonoids in Non‐Leguminous Families: An Update,”. Natural Product Reports, 2005. [DOI | PubMed]
- Major Secondary Metabolites of Iris Spp.,”. Phytochemistry Reviews, 2013
- Secondary Metabolites of the Chosen Genus Iris Species,”. Acta Universitatis Agriculturae et Silviculturae Mendelianae Brunensis, 2012
- The Phytochemical Richness of the Iridaceae and Its Systematic Significance,”. Annals of Botany, 2000
- 38 J. Zhou , G. Xie , and X. Yan , Encyclopedia of Traditional Chinese Medicines: Molecular Structures, Pharmacological Activities, Natural Sources and Applications. Vol. 3: Isolated Compounds H‐M. (Springer, 2011).
- Flavonoids and Xanthones From the Genus Iris: Phytochemistry, Relationships With Flower Colors and Taxonomy, and Activities and Function,”. Natural Products Communications, 2020
- Iris ensata Thunb: Review on Its Chemistry, Morphology, Ethno Medical Uses, Phytochemistry and Pharmacological Activities,”. Asian Journal of Dental and Health Sciences, 2022. [DOI]
- Phytochemistry and Pharmacology of Iris kashmiriana ,”. Pharmacognosy Research, 2022. [DOI]
- Belamcandae Chinensis Rhizoma—A Review of Phytochemistry and Bioactivity,”. Fitoterapia, 2015. [PubMed]
- Belamcanda chinensis (L.) DC‐An Ethnopharmacological, Phytochemical and Pharmacological Review,”. Journal of Ethnopharmacology, 2016. [DOI | PubMed]
- The Constituents of Iris florentina L. (3). Structure of Irisxanthone, a New C‐Glycosylxanthone,”. Chemical and Pharmaceutical Bulletin, 1973. [DOI]
- New Isoflavones From Iris nigricans ,”. Journal of Natural Products, 1994. [DOI]
- Two New Isoflavanoids From the Rhizomes of Iris Soforana,”. Natural Product Research, 2004. [DOI | PubMed]
- A Benzil and Isoflavone From Iris tenuifolia ,”. Phytochemistry, 2008. [DOI | PubMed]
- Four New Flavones and a New Isoflavone From Iris bungei ,”. Journal of Natural Products, 2001. [DOI | PubMed]
- Isoflavone Constituents of the Underground Parts of Iris florentina and Their Inhibitory Activity on the Formation of Advanced Glycation End Products,”. Chemical & Pharmaceutical Bulletin, 2025. [DOI | PubMed]
- A New Isoflavone From Iris pseudacorus ,”. Chemistry of Natural Compounds, 2013. [DOI]
- Isoflavonoids From the Rhizomes of Iris hungarica and Antibacterial Activity of the Dry Rhizomes Extract,”. Ars Pharmaceutica, 2017. [DOI]
- Effect of Ecological Factors on the Accumulation of Phenolic Compounds in Iris Species From Latvia, Lithuania and Ukraine,”. Phytochemical Analysis, 2020. [DOI | PubMed]
- Qualitative and Quantitative Analysis of Ukrainian Iris Species: A Fresh Look on Their Antioxidant Content and Biological Activities,”. Molecules (Basel, Switzerland), 2020. [DOI | PubMed]
- New Isoflavones With Cytotoxic Activity From the Rhizomes of Iris germanica L.,”. Natural Product Research, 2013. [DOI | PubMed]
- New Isoflavonoid Glycosides From the Rhizomes of Iris leptophylla Lingelsh,”. Journal of Integrative Plant Biology, 2007. [DOI]
- New Isoflavones and Flavanol From Iris potaninii ,”. Chemical & Pharmaceutical Bulletin, 2002. [DOI | PubMed]
- Isoflavonoids and Flavone Glycosides From Rhizomes of Iris carthaliniae ,”. Phytochemistry, 1999. [DOI]
- Qualitative and Quantitative Evaluation of Phenolic Compounds in Iris dichotoma Pall,”. Phytochemical Analysis, 2012. [DOI | PubMed]
- Phenolic Constituents With Anti‐Inflammatory and Cytotoxic Activities From the Rhizomes of Iris domestica ,”. Phytochemistry, 2022. [DOI | PubMed]
- New Isoflavone Glycosides From Iris spuria L. (Calizona) Cultivated in Egypt,”. Journal of Natural Medicines, 2009. [DOI | PubMed]
- Isoflavonoid Glycosides From the Rhizomes of Iris germanica ,”. Chemical and Pharmaceutical Bulletin, 2002. [DOI | PubMed]
- Isoflavonoid Glycosides From the Rhizomes of Iris germanica ,”. Helvetica Chimica Acta, 2003. [DOI]
- Antioxidant Flavonoids and Isoflavonoids From Rhizomes of Iris pseudopumila ,”. Planta Medica, 2007. [DOI | PubMed]
- Chemical Constituents From Rhizomes of Iris germanica ,”. Zhongguo Zhong Yao Za Zhi = Zhongguo Zhongyao Zazhi = China Journal of Chinese Materia Medica, 2014. [PubMed]
- New Stilbenes and Phenolic Constituents From the Rhizomes of Belamcanda chinensis ,”. Journal of Asian Natural Products Research, 2022. [DOI | PubMed]
- Characterisation and Quantification of Flavonoids in Iris germanica L. and Iris pallida Lam. Resinoids From Morocco,”. Phytochemical Analysis, 2012. [DOI | PubMed]
- Isoflavones and Other Compounds From the Roots of Iris marsica I. Ricci E Colas. Collected From Majella National Park, Italy,”. Medicinal Chemistry, 2017. [DOI]
- Protection Against Neurodegenerative Diseases of Iris pseudopumila Extracts and Their Constituents,”. Fitoterapia, 2009. [DOI | PubMed]
- Phenolic Compounds From Iris rossii, and Their Chemotaxonomic and Systematic Significance,”. Biochemical Systematics and Ecology, 2012. [DOI]
- Chemical Constituents of Leaves of Iris lactea Pall. Var. Chinensis ,”. Chinese Journal of Pharmaceuticals, 2009
- Two New C‐Glycosylflavones From the Leaves of Iris lactea Var. Chinensis,”. Chinese Chemical Letters, 2008. [DOI]
- Measurement of Food Flavonoids by High‐Performance Liquid Chromatography: A Review,”. Journal of Agricultural and Food Chemistry, 2000. [DOI | PubMed]
- C‐Glycosylflavones From the Leaves of Iris tectorum Maxim,”. Acta Pharmaceutica Sinica B, 2012. [DOI]
- 6′‐O‐Acetyl Mangiferin From Iris rossii Baker Inhibits Lipid Accumulation Partly via AMPK Activation in Adipogenesis,”. Chemico‐Biological Interactions, 2019. [DOI | PubMed]
- Five New Peltogynoids From Underground Parts of Iris bungei: A Mongolian Medicinal Plant,”. Chemical and Pharmaceutical Bulletin, 2001. [DOI | PubMed]
- Studies on Constituents of Genus Iris. VIII. The Constituents of Iris unguicularis Poir. 2,”. Chemical and Pharmaceutical Bulletin, 1976. [DOI]
- Flavonoids From Iris songarica and Their Antioxidant and Estrogenic Activity,”. Planta Medica, 2008. [DOI | PubMed]
- Analysis of Flavonoids and Phenolic Acids in Iris tectorum by HPLC‐DAD‐ESI‐MSn,”. Chinese Journal of Natural Medicines, 2010. [DOI]
- Five New Flavonoids and Their Pharmacological Activities From Iris tenuifolia Pall,”. Fitoterapia, 2024. [DOI | PubMed]
- Towards the Characterization of Radicals Formed in Rhizomes of Iris germanica ,”. Phytochemistry, 1994. [DOI]
- Cytotoxic Effects of Compounds From Iris tectorum on Human Cancer Cell Lines,”. Journal of Ethnopharmacology, 2008. [DOI | PubMed]
- Chemical Constituents of Iris cathayensis Migo,”. Journal of China Pharmaceutical University, 2005
- Phenolic Constituents of Iris milesii Rhizomes,”. Phytochemistry, 1984. [DOI]
- Flavonoids Produced by Iris pseudacorus Leaves Treated With Cupric Chloride,”. Phytochemistry, 1991. [DOI]
- HPLC‐DAD‐ESI‐MS/MS Identification and Characterization of Major Constituents of Iris crocea, Iris germanica and Iris spuria Growing in Kashmir Himalayas, India,”. Journal of Analytical and Bioanalytical Techniques, 2014. [DOI]
- Crocetenone, a New Rotenoid With an Unusual Trans‐Fused Ring System From Iris crocea ,”. Natural Products Communications, 2015
- Studies on α‐Glucosidase Inhibition and Anti‐Glycation Potential of Iris loczyi and Iris unguicularis ,”. Life Sciences, 2013. [DOI | PubMed]
- New Constituents From the Rhizomes of Egyptian Iris germanica L,”. Molecules (Basel, Switzerland), 2012. [DOI | PubMed]
- Isoflavonoids Produced by Iris pseudacorus Leaves Treated With Cupric Chloride,”. Phytochemistry, 1991. [DOI]
- A New Coumaronochromone and Phenolic Constituents From the Leaves of Iris bungei Maxim,”. Biochemical Systematics and Ecology, 2009. [DOI]
- Chemical Constituents of Leaves of Iris lactea Pall, Van Chinensis,”. Chinese Journal of Pharmaceuticals, 2009
- Irispurinol, a 12a‐Hydroxyrotenoid From Iris spuria ,”. Phytochemistry, 1988. [DOI]
- Anthocyanin 5‐O‐Glucosyltransferase in Flowers of Iris ensata ,”. Plant Science, 2002. [DOI]
- Characterization of Anthocyanins in Flowers of Japanese Garden Iris, Iris ensata Thunb,”. Cytol, 2003. [DOI]
- Characterization of Anthocyanins in the Hybrid Progenies Derived From Iris dichotoma and I. domestica by HPLC‐DAD‐ESI/MS Analysis,”. Phytochemistry, 2018. [DOI | PubMed]
- Mango (Mangifera indica L.): A Magnificent Plant With Cancer Preventive and Anticancer Therapeutic Potential,”. Critical Reviews in Food Science and Nutrition, 2021. [DOI | PubMed]
- Mangiferin and Other Glycophenolics in Iris Species,”. Nature, 1963. [DOI]
- Mangiferin: A Review of Dietary Sources, Absorption, Metabolism, Bioavailability, and Safety,”. Critical Reviews in Food Science and Nutrition, 2023. [DOI | PubMed]
- A Xanthone C‐Glycoside From Iris nigricans ,”. Phytochemistry, 1995. [DOI]
- Characterization and Determination of the Major Constituents in Belamcandae Rhizoma by HPLC–DAD–ESI‐MSn,”. Journal of Pharmaceutical and Biomedical Analysis, 2011. [DOI | PubMed]
- Phylogeny and Classification of Iridaceae,”. Annals of the Missouri Botanical Garden, 1990. [DOI]
- Metabolic Profiling of Rhizomes of Native Populations of the Strictly Endemic Croatian Species Iris adriatica,”. Plant Biosyst, 2019. [DOI]
- Phytochemical Analysis and Total Antioxidant Capacity of Rhizome, Above‐Ground Vegetative Parts and Flower of Three Iris Species,”. Chemistry and Biodiversity, 2019
- Investigation of Secondary Metabolites and Their Bioactive Potential in Various Iris Species and Cultivars Grown Under Different Cultivation Conditions,”. ACS Omega, 2025. [DOI | PubMed]
- Chemical Constituents of the Underground Parts of Iris florentina and Their Cytotoxic Activity,”. Natural Products Communications, 2015
- Chemical Constituents From Iris scariosa and Iris halophila Var. Sogdiana ,”. Chinese Traditional and Herbal Drugs, 2013
- Phenolic Constituents of the Rhizomes of the Thai Medicinal Plant Belamcanda chinensis With Proliferative Activity for Two Breast Cancer Cell Lines,”. Journal of Natural Products, 2005. [DOI | PubMed]
- Belamcandones A‐D, Dioxotetrahydrodibenzofurans From Belamcanda chinensis ,”. Phytochemistry, 1995. [DOI]
- Phenolic Compounds From Belamcanda chinensis Seeds,”. Molecules (Basel, Switzerland), 2018. [DOI | PubMed]
- Isolation, Structural Elucidation, and Chemical Synthesis of 2‐Hydroxy‐3‐Octadecyl‐5‐Methoxy‐1,4‐Benzoquinone (irisoquin), a Cytotoxic Constituent of Iris missouriensis ,”. Journal of Pharmaceutical Sciences, 1985. [DOI | PubMed]
- Alkylated Benzoquinones From Iris kumaonensis ☆,”. Phytochemistry, 2002. [DOI | PubMed]
- Phenols and Quinones From Seeds of Different Iris Species,”. Helvetica Chimica Acta, 1992. [DOI]
- Method for Preparing High Purity Irisquinone, Pallason A . China Patent CN101575277B.. 2009
- A New Alkylated Benzoquinone From Rhizomes of Iris kumaonensis ,”. Natural Product Research, 2006. [DOI | PubMed]
- New Alkylated Benzoquinone From Iris nepalensis ,”. Natural Product Research, 2013. [DOI | PubMed]
- A New Belamcandaquinone From the Seeds of Iris bungei Maxim,”. Fitoterapia, 2011. [DOI | PubMed]
- Two New Quinones From Iris bungei ,”. Chemical and Pharmaceutical Bulletin, 2000. [DOI | PubMed]
- Antioxidant and Anticholinesterase Constituents From the Petroleum Ether and Chloroform Extracts of Iris suaveolens ,”. Phytotherapy Research, 2010. [PubMed]
- Secondary Metabolites Isolated From Iris germanica ,”. Records of Natural Products, 2009
- Isolation of Oligostilbenes From Iris lactea Pall. Var. Chinensis (Fisch.) Koidz and Their Anti‐Inflammatory Activities,”. RSC Advances, 2022. [DOI | PubMed]
- Chromane Derivatives From Underground Parts of Iris tenuifolia and Their In Vitro Antimicrobial, Cytotoxicity and Antiproliferative Evaluation,”. Molecules (Basel, Switzerland), 2021. [DOI | PubMed]
- New and Known Constituents From Iris unguicularis and Their Antioxidant Activity,”. Heterocycles, 2010. [DOI]
- Phenolic Compounds of the Genus Iris Plants (Iridaceae),”. Česká a slovenská farmacie, 2016. [DOI | PubMed]
- Hydroxycinnamic Acids in the Raw Material of Hybrid Bearded Irises ,”. Zaporozhye Medical Journal, 2020
- Screening for Phenolic Acids in Five Species of Iris Collected in Mongolia,”. Acta Chromatographica, 2008. [DOI]
- Biological Effects and Chemical Characterization of Iris schachtii Markgr. Extracts: A New Source of Bioactive Constituents,”. Food and Chemical Toxicology, 2018. [DOI | PubMed]
- Antimicrobial, Antioxidant, Antimutagenic Activities, and Phenolic Compounds of Iris germanica ,”. Industrial Crops and Products, 2014. [DOI]
- Stilbene Glucosides From the Bulbs of Iris tingitana ,”. Phytochemistry Letters, 2009. [DOI]
- Antioxidant and Anticholinesterase Constituents From the Petroleum Ether and Chloroform Extracts of Iris suaveolens ,”. Phytotherapy Research, 2011. [DOI | PubMed]
- Profiling of Isoflavonoids in Iris germanica Rhizome Extracts by Microprobe NMR and HPLC–PDA–MS Analysis,”. Fitoterapia, 2011. [DOI | PubMed]
- Two Isoflavonoids From the Fresh Bulbs of Iris tingitana ,”. Phytochemistry, 1980. [DOI]
- Etudes sur les Matières Végétales Volatiles XC. Présence d’ apocynine (acéto‐vanillone) dans L’huile Essentielle D’iris,”. Helvetica Chimica Acta, 1949. [DOI]
- Constituents of Rhizomes of Iris tectorum ,”. Chinese Journal of Natural Medicines, 2007
- Non‐Isoflavonoid Compounds of Iris tectorum ,”. Zhong Yao Cai = Zhongyaocai = Journal of Chinese Medicinal Materials, 2013. [PubMed]
- Apocynin Derivatives From Iris tectorum ,”. Journal of Asian Natural Products Research, 2017. [DOI | PubMed]
- Isoflavonoids From the Rhizomes of Belamcanda chinensis and Their Effects on Aldose Reductase and Sorbitol Accumulation in Streptozotocin Induced Diabetic Rat Tissues,”. Archives of Pharmacal Research, 2002. [DOI | PubMed]
- Phytochemistry and Biological Activities of Iris Species Growing in Iraqi Kurdistan and Phenolic Constituents of the Traditional Plant Iris postii ,”. Molecules (Basel, Switzerland), 2021. [DOI | PubMed]
- 138 H. Wagner , R. Bauer , D. Melchart , P.‐G. Xiao , and A. Staudinger , “Ch. Rhizoma Belamcandae Sinensis Shegan,” in Chromatographic Fingerprint Analysis of Herbal Medicines, 2nd ed. (Springer‐Verlag Wien, 2011), 10.1007/978-3-7091-0763-8. Ed. 2,.
- Rearranged Iridal‐Type Triterpenoids From Iris tectorum ,”. Fitoterapia, 2019. [DOI | PubMed]
- Two New Isoflavanoids From the Rhizomes of Iris soforana ,”. Natural Product Research, 2004. [DOI | PubMed]
- Chemical Constituents of the Ethyl Acetate Extract of Belamcanda chinensis (L.) DC Roots and Their Antitumor Activities,”. Molecules (Basel, Switzerland), 2012. [DOI | PubMed]
- Chemical Constituents of the Leaves of Iris songarica ,”. Journal of China Pharmaceutical University, 2006
- LC–MS Guided Isolation of Bioactive Principles From Iris hookeriana and Bioevaluation of Isolates for Antimicrobial and Antioxidant Activities,”. Drug Research, 2016. [PubMed]
- Quality Evaluation of Rhizoma Belamcandae (Belamcanda chinensis (L.) DC.) by Using High‐Performance Liquid Chromatography Coupled With Diode Array Detector and Mass Spectrometry,”. Journal of Chromatography A, 2009. [DOI | PubMed]
- Halophilols A and B, Two New Stilbenes From Iris halophila ,”. Planta Medica, 2003. [PubMed]
- Resveratrol‐Type Oligostilbenes From Iris clarkei Antagonize 20‐Hydroxyecdysone Action in the Drosophila Melanogaster BII Cell Line,”. Cellular and Molecular Life Sciences, 2000. [DOI | PubMed]
- Combined MS/MS‐NMR Annotation Guided Discovery of Iris lactea Var. Chinensis Seed as a Source of Viral Neuraminidase Inhibitory Polyphenols,”. Molecules (Basel, Switzerland), 2020. [DOI | PubMed]
- Alkaloids From the Rhizomes of Iris germanica ,”. Chemistry of Natural Compounds, 2017. [DOI]
- Analysis of the Iridals in Rhizome Extracts of Iris variegata Linn,”. Natural Product Research, 2005. [DOI | PubMed]
- Structure Determination of a New Spirobicyclic Triterpenoid From Iris foetidissima ,”. Helvetica Chimica Acta, 1990. [DOI]
- Chemical Diversity of Iridal‐Type Triterpenes in Iris delavayi Collected in Yunnan Province of China,”. Natural Products Communications, 2011
- New C16‐Noriridals and Formyl‐Monocycloiridals From the Rhizomes of Iris Pseudoacorus ,”. Fitoterapia, 2018. [DOI | PubMed]
- Two New Iridal‐Type Triterpenoids From Iris forrestii ,”. Journal of Asian Natural Products Research, 2019. [DOI | PubMed]
- Iridal‐Type Triterpenoids With Neuroprotective Activities From Iris tectorum ,”. Journal of Natural Products, 2014. [DOI | PubMed]
- Isolation and Structure Determination of Triterpenes From Iris tectorum ,”. Phytochemistry, 2007. [DOI | PubMed]
- Iridals From Iris tectorum and Belamcanda chinensis ,”. Phytochemistry, 2000. [DOI | PubMed]
- Protein Kinase C Activation by Iridal Type Triterpenoids,”. Biological & Pharmaceutical Bulletin, 2002. [DOI | PubMed]
- Polycycloiridals A–D, Four Iridal‐Type Triterpenoids With an α‐Terpineol Moiety From Iris tectorum ,”. Organic Letters, 2015. [DOI | PubMed]
- Iridal‐Type Triterpenoids With a Cyclopentane Unit From the Rhizomes of Belamcanda chinensis ,”. Journal of Natural Products, 2019. [DOI | PubMed]
- Belamchinenin A, an Unprecedented Tricyclic‐Fused Triterpenoid With Cytotoxicity From Belamcanda chinensis ,”. Organic & Biomolecular Chemistry, 2018. [DOI | PubMed]
- A New Dimeric Iridal Triterpenoid From Belamcanda chinensis With Significant Molluscicide Activity,”. Organic Letters, 2011. [DOI | PubMed]
- Molecular Networking‐Based Discovery of Long Chain Fatty Acid Bearing Iridal Triterpenoids With Neurite Outgrowth Promoting Activity From Iris domestica Rhizomes,”. Fitoterapia, 2025. [DOI | PubMed]
- Localization of Iridals in Iris germanica Rhizomes,”. Phytochemistry, 1996. [DOI]
- New Piscicidal Triterpenes From Iris germanica ,”. Chemical and Pharmaceutical Bulletin, 1995. [DOI]
- Irigermanal and Iridogermanal: Two New Triterpenoids From Rhizomes of Iris germanica L.,”. Journal of Organic Chemistry, 1982. [DOI]
- Iridal‐Type Triterpenoids With Anti‐HBV Activity From Iris confusa ,”. Fitoterapia, 2018. [DOI | PubMed]
- Isolation and Structure Determination of New Iridals From Iris sibirica and Iris versicolor ,”. Liebigs Annalen Der Chemie, 1992. [DOI]
- Isolation and Structure Determination of a New iridal From Iris sibirica ,”. Journal of Natural Products, 1995. [DOI]
- 169 J. B. Harborne and H. Baxter , Chemical Dictionary of Economic Plants (John Wiley & Sons, 2001).
- Chemical Composition and Antifungal Activity of Essential Oils From Flowers, Leaves, Rhizomes, and Bulbs of the Wild Iraqi Kurdish Plant Iris persica ,”. Natural Products Communications, 2017
- Chemical Composition and Biological Activities of Essential Oil From the Rhizomes of Iris bulleyana ,”. Agricultural Science in China, 2009. [DOI]
- Aromatic Compounds and Terpenoids of Iris hungarica ,”. Chemistry of Natural Compounds, 2014. [DOI]
- Constituents of Essential Oils From Azerbaijan Iris Medwedewii and I. carthaliniae Rhizomes,”. Chemistry of Natural Compounds, 2016. [DOI]
- Chemical Composition of the Essential Oil of Several Iris Species,”. Thai Journal of Pharmaceutical Sciences, 2020
- Composition of Volatiles From Three Iris Species of Turkey,”. Journal of Essential Oil Research, 2011
- Variation in Essential Oil Composition of Iris nigricans Dinsm. (Iridaceae) Endemic to Jordan at Different Flowering Stages,”. Arabian Journal of Chemistry, 2016. [DOI]
- Antioxidant Activity of the Aqueous Extract of Iris taochia and Identification of Its Chemical Constituents,”. Indian Journal of Pharmaceutical Sciences, 2018. [DOI]
- Free Radical Scavenging and Antibacterial Activity of Essential Oil and Solvent Extracts of Iris Planifolia (Mill) From Algeria,”. Journal of Medicinal Plants Research, 2012
- Gas Chromatography‐Mass Spectrometry Analysis of Volatile Organic Compounds From Three Endemic Iris Taxa: Headspace Solid‐Phase Microextraction vs. Hydrodistillation,”. Molecules, 2024. [PubMed]
- Composition of Volatile Oil of Iris pallida Lam. From Ukraine,”. Turkish Journal of Pharmaceutical Sciences, 2018. [PubMed]
- Gas Chromatography‐Mass Spectrometry Studies of the Component Composition of Carboxylic Acids of the Rhizomes of Iris medwedewii and Iris carthaliniae (Iridaceae),”. Česká a slovenská farmacie, 2017. [DOI | PubMed]
- Carboxylic Acids From Iris graminea and I. halophila ,”. Chemistry of Natural Compounds, 2018. [DOI]
- The Comparative Analysis of Carboxylic Acid Composition of Four Iris Species From Ukraine,”. Chemistry and Biodiversity, 2021. [DOI | PubMed]
- Long‐Chain Fatty Acid Acylated Derivatives of Isoflavone Glycosides From the Rhizomes of Iris domestica ,”. Phytochemistry, 2022. [DOI | PubMed]
- Gas Chromatography‐Mass Spectrometry (GC‐MS) Analysis of Petroleum Ether Extract (Oil) and Bio‐Assays of Crude Extract of Iris germanica ,”. International Journal of Genetics and Molecular Biology, 2011. [DOI]
- 186 T. K. Lim , “ Iris germanica ,” in Edible Medicinal and Non‐Medicinal Plants. Vol. 11 Modified Stems, Roots, Bulbs (Springer International Publishing, 2016).
- Fatty Acid Composition of Lipids of Iris sibirica ,”. Ceska A Slovenska Farmacie, 2017. [DOI]
- Chemical Analysis of Essential Oils of Some Syrian Wild Iris Species,”. American Journal of Biochemistry and Molecular Biology, 2013. [DOI]
- Comparative Investigation of Amino Acids Content in the Dry Extracts of Juno bucharica, Gladiolus Hybrid Zefir, Iris hungarica, Iris variegata and Crocus Sativus Raw Materials of Ukrainian Flora,”. Scientia Pharmaceutica, 2020. [DOI]
- Genetic and Environmental Factors Jointly Impact Leaf Phenolic Profiles of Iris variegata L.,”. Plants, 2021. [PubMed]
- Bioactive Constituents of Iris hybrida (Iridaceae): Processing Effect,”. Biomedical Chromatography, 2022. [DOI | PubMed]
- 192 C. P. Khare , Indian Medicinal Plants (Springer—Verlag, 2007), 10.1007/978-0-387-70638-2.
- Innovative GACP Approaches for Obtaining the Quality Iris Hybrida Leaves for the Pharmaceutical Industry,”. Chemistry and Biodiversity, 2022. [DOI | PubMed]
