Does Cannabis Use Contribute to Schizophrenia? A Causation Analysis Based on Epidemiological Evidence
Abstract
Cannabis abuse has been linked to acute psychotic symptoms as well as to the development of schizophrenia. Although the association has been well described, causation has not yet been investigated. Therefore, we investigated whether cannabis or cannabinoid use is causal for the development of schizophrenia, conducting a systematic literature review according to the PRISM guidelines. Epidemiological studies and randomized clinical trials investigating the links between cannabis and psychosis-like events (PLE) and schizophrenia were identified (according to PRISM guidelines), and relevant studies were included in a Forest plot analysis. Confounder analysis was performed using a funnel plot, and the Hill causality criteria were used to estimate causation. A total of 18 studies fulfilled the search criteria; 10 studies were included in a forest plot. All studies reported an increased risk for PLE or schizophrenia, and nine of the ten studies, a significant increase; the overall OR was calculated to be 2.88 (CI 2.24 to 3.70), with a twofold-higher risk calculated for cannabis use during adolescence. Confounder effects were indicated by a funnel plot. The Hill criteria indicated a high likelihood for the contribution of cannabis to schizophrenia development. Cannabinoids likely contribute to chronic psychotic events and schizophrenia, especially if taken during adolescence. This effect likely increases with a high cannabis THC concentration and increased frequency of cannabis use, and is stronger in males than in females. This points to the possibility of a selective cannabis toxicity on synaptic plasticity in adolescence, as compared to adult cannabis use. Cannabis use should be regulated and discouraged, and prevention efforts should be strengthened, especially with reference to adolescence.
Article type: Review Article
Keywords: cannabinoids, psychosis, causality assessment, tetrahydrocannabinol, dihydrocannabidiol
Affiliations: Clinic of Anesthesiology, Surgical Intensive Care, Emergency Medicine and Pain Medicine, Main-Kinzig-Kliniken, Herzbachweg 14, D-63571 Gelnhausen, Germanylisarottmann0@gmail.com (E.R.); Institute of Occupational, Social and Environmental Medicine, Goethe-University Frankfurt/Main, Theodor-Stern-Kai 7, D-60590 Frankfurt/Main, Germany; tooba_91@hotmail.com; Paediatric Department, Epsom General Hospital, Epsom and St Helier University Hospitals NHS Trust, Dorking Road, Epsom KT18 7EG, Surrey, UK; Anesthesiological Clinic, St. Josef-Hospital Wiesbaden, Beethovenstraße 20, D-65189 Wiesbaden, Germany; bscheller@joho.de
License: © 2025 by the authors. CC BY 4.0 Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.3390/biom15030368 | PubMed: 40149904 | PMC: PMC11940535
Relevance: Relevant: mentioned in keywords or abstract
Full text: PDF (1.4 MB)
1. Introduction
Recreational cannabis use is illegal in most countries, but it is one of the most commonly used drugs, both in frequency and dosage, with estimated 228 million users in 2022 worldwide [ref. 1]. Until recently, the main cannabis varieties were dried flowers (marijuana) and resin (haschisch); however, high-tetrahydrocannabinol (THC) varieties like “Sinsemilla” and “Skunk” are marketed in many countries [ref. 2]. Besides recreational use, cannabinoids and cannabis flowers are licensed medications in many countries for a variety of diseases [ref. 3,ref. 4,ref. 5]. Despite its status as medication, evidence for its efficiency is scant, and this evidence has not been confirmed in meta-analyses, e.g., findings for spasmolysis in multiple sclerosis [ref. 6]. Side effects of cannabis and cannabinoids include acute psychotic states and psychosis-like events (PLE), decreased cognitive functions and attention, and lower concentration [ref. 7]. Long-term effects are less well-established; besides drug dependence, neurocognitive deficits, and morphological changes in brain architecture, PLE and schizophrenia have been postulated as direct consequences of cannabis abuse [ref. 7,ref. 8]. Among these effects, the chronic effects pose a much larger risk for the individual as well as creating a large burden of disease for society.
Schizophrenia is defined by the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5), as a combination of two or more symptoms, including delusions, hallucinations, disordered speech, and disturbed behavior [ref. 9]. Substance-use disorder is common among patients with schizophrenia [ref. 10,ref. 11], and its prevalence is estimated to be about 40 to 50% [ref. 12], with even higher values determined for cigarette smoking [ref. 12,ref. 13,ref. 14]. Impaired social functioning is a key criterion for the diagnosis of schizophrenia, and such impairments are highly correlated with deficits in interpersonal skills; additionally, affected individuals have deficits in neuropsychology and social cognition [ref. 15], as well as emotional recognition [ref. 16]. Khokha et al. [ref. 17] describe a high risk of development of substance-use disorders in schizophrenic patients, whereas usually drug abuse—especially cannabis abuse—is observed prior to the diagnosis of schizophrenia [ref. 11,ref. 18]. For this association a “neurobiological predisposition” has been proposed, as well as a “self-medication” hypothesis [ref. 12]. While psychotic symptoms are seen as predominant by some authors [ref. 10], others postulate more negative symptoms [ref. 12]. Case studies demonstrate a faster development of psychotic symptoms with earlier and/or more frequent cannabis use [ref. 19]. Early on, Smit et al. postulated that cannabis use nearly doubles the risk of the development of schizophrenia [ref. 20]. The strong correlation between cannabis use and the early onset of psychosis suggests that cannabis use could cause or accelerate the onset of schizophrenia.
No meta-analysis has been published yet for chronic psychotic cannabis reactions. In order to investigate whether cannabis abuse is a major contributor to the development of schizophrenia or merely indicates individuals with an increased risk to develop schizophrenia, we performed a meta-analysis of all relevant studies that calculated an OR for chronic PLE or schizophrenia. Using the Hill criteria for causality we studied whether the association was contributory/causal or merely temporal.
2. Materials and Methods
The databases PubMed, Cochrane Library, EMBASE, PsycINFO, Google Scholar, and the American and European Clinical Trial databases https://clinicaltrials.gov and https://www.clinicaltrialsregister.eu (both accessed on 13 July 2024) were searched following the Preferred Reporting Items for Systematic reviews and Meta-analyses (PRISM; this study has not been registered). The search term “(cannabis OR marijuana) AND (schizophrenia OR psychosis) AND (prevalence OR risk OR incidence OR association OR relationship)” was used, with and without the restrictions “RCT”, “observational study”, and “review”. Additionally, citations in reviews and full texts analyzed from original articles were screened for additional studies not identified in the original search. For the further analysis, only English-language publications were included.
This selection resulted in 371 articles, which were then screened for relevance according to titles and abstracts. According to their abstracts, 203 publications did not directly report observational studies or RCT; in 114 articles, schizophrenia or psychosis had not been diagnosed according to ICD or DSM criteria, control groups were lacking, or data sets were incomplete, resulting in the exclusion of 317 articles. The full texts of the remaining 54 articles were downloaded and analyzed by at least two independent reviewers. In sum, 36 additional articles were excluded since the inclusion/exclusion criteria were not met, and/or confounders were not listed. From these eighteen studies, odds ratios from ten studies were included in the forest plot analysis, and eight studies were excluded because of major overlap with another study, a misfit of psychosis criteria or the lack of a control group (Figure 1).
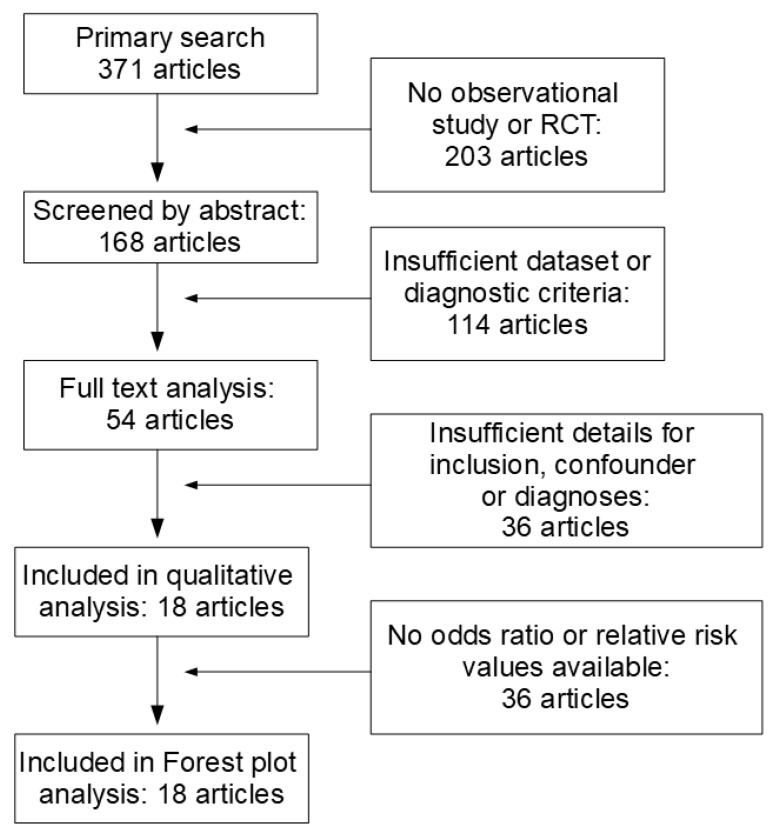
Inclusion criteria comprised the following:
- Observational studies, case-control studies, and RCT for the association of cannabis use and the development of psychotic or affective disorders (schizoaffective symptoms);
- Psychotic or affective symptoms diagnosed as specified by ICD or DSM criteria;
- Anamnestic prolonged cannabis use;
- Sufficient description of confounders, e.g., use of other drugs, preexisting disease, or medications.
Exclusion criteria comprised the following:
- Case reports or case series;
- Overlapping cohorts in multiple publications;
- No control group included;
- No calculation of odds ratio possible.
2.1. Data Extraction and Quality Evaluation
Data extraction was performed by at least two authors for each published article. From all publications, the following information was extracted: authors, publication year, type of study, country of origin, number of controls and patients, method to determine cannabis exposure (if possible, estimation of cumulative dose), diagnostic criterion (ICD, DSM), odds ratios, confidence intervals, and confounders.
To estimate the possibility of bias all included articles were assessed by the criteria of Moore et al. [ref. 21] for confounders in randomization (negative if no method is given), frequency of study deviations (number of incomplete data sets), diagnostic criteria (qualitative, semiquantitative), and outcome reporting; additionally, the GRADE criteria [ref. 22] were applied for risk of selection bias (e.g., participant selection, participating institutions), inconsistencies (i.e., deviation from comparable parameters, other studies), indirectness (e.g., selection of effects, diagnostic criteria), imprecision (i.e., qualitative reporting), and quality of results (completeness of datasets). A funnel plot analysis was also performed to estimate comparability of the included studies; a larger number of studies outside of the expected funnel indicates a substantial level of study heterogeneity.
2.2. Statistical Analysis
Based on 10 articles with a given odds ratio and confidence interval, a forest plot analysis was performed [ref. 23]. If no OR value was given in an article it was calculated from the relative risk values for the control and exposed groups, in addition to an estimated confidence interval. To identify additional confounders or other possible bias factors, a funnel plot was constructed [ref. 24].
3. Results
3.1. Study Characteristics
In total, we identified 18 articles investigating the relationship between cannabis use and schizophrenia (Table 1). The oldest article was from 1997, with a nearly identical distribution of publications for the years between 2004 to 2024. Study design, psychosis parameters, and follow-up were heterogenous. Only three studies were longitudinal; they included adolescents 14–16 years of age, and registered psychotic symptoms [ref. 25,ref. 26] or affective symptoms [ref. 27] with 6–10 years follow-up, Callaghan et al. [ref. 28] followed adolescents for only 1 year. Additionally, some cross-sectional studies were included in the analysis [ref. 29,ref. 30,ref. 31], as was the study of Power et al. [ref. 32] although this twin study focused on genetic factors. Sevy et al. [ref. 33] studied risk factors in schizophrenic patients, i.e., reverse causation. In order to estimate an overall risk for schizophrenia by cannabis use we analyzed these studies together despite the large study variability; however, the resulting forest plot estimate has to be discussed cautiously. On the other hand, we excluded studies if no odds ratio could be calculated or where there were variations in study protocols, as well as intervention studies with cannabidiol; the study from Hjorthoj et al. [ref. 34] was excluded, since this study analyzed the same cohort as the 2023 study [ref. 29]. Studies not included in the forest plot could provide data for the criterion-based causality analysis.
Table 1: Articles investigating the correlation between cannabis use and psychosis/schizophrenia.
| Reason for Inclusion/Exclusion | Focus of Study | Country, Participants | Type of Study | Study |
|---|---|---|---|---|
| Excluded; no odds ratio given | Acute psychotic events after 10 mg THC | U.S.A., 31 participants | RCT | Aghaei et al., 2024 [ref. 35] |
| Included | Development of psychosis after cannabis use in adolescence | Canada, 11,363 part., 12 to 24 y | Longitudinal cohort study | McDonald et al., 2024 [ref. 25] |
| Excluded; psychosis was no end-point | Impairment of driving after smoking cannabis | Canada, 31 older volunteers | Open label exposure study | Di Ciano et al., 2024 [ref. 36] |
| Excluded due to study protocol, no results yet | Decrease in psychosis by CDB | U.S.A., 120 patients | RCT | Dixon et al., 2023 [ref. 37] |
| Included | Correlation of schizophrenia diagnosis and cannabis use disorder | Denmark, 7,186,834 population, 16–49 y | Population study | Hjorthoj et al., 2023 [ref. 29] |
| Excluded; cohort identical to that of Hjorthoj et al., 2023 [ref. 29] | Population-attributable risk factor of cannabis for schizophrenia | Denmark, 6,907,859 population, 16–49 y | Population study | Hjorthoi et al., 2021 [ref. 34] |
| Included | Correlation of psychosis and cannabis use in psychiatric patients | EU, Brazil; 901 cases, 1237 control | Case control study | Di Forti et al., 2019 [ref. 38] |
| Excluded; no odds ratio for psychosis given | Effectiveness of cannabis-based medicine in cannabis reduction | London UK, 278 interv., 273 controls | RCT | Johnson et al., 2019 [ref. 39] |
| Included | Development of psychosis after cannabis use in adolescence | Northern Finland, 6534 patients 16 y | Longitudinal population study | Mustonen et al., 2018 [ref. 26] |
| Excluded; no THC applied | Antipsychotic activity of 1000 mg CDB | Europe, 43 interv., 45 controls | RCT | McGuire et al., 2017 [ref. 40] |
| Included; small cohort overlap with Di Forti et al., 2019 [ref. 38] | Association of psychosis and cannabis use in psychiatric patients | London, UK, 410 cases | Cohort study | Di Forti et al., 2014 [ref. 30] |
| Included; similar/identical genetics | Association of schizophrenia with cannabis use in twins | Australia, 2082 participants | Twin registry | Power et al., 2014 [ref. 32] |
| Excluded; no control group without drug abuse | Comparison of different drug-abuse personalities | UK, 160 users, 167 nonusers | Case control study | Barrowclough et al., 2013 [ref. 41] |
| Included; “Cannabis-related problems” over 1 year | Correlation of cannabis use with CRP | BC, Canada; 3339 users | Population study | Callaghan et al., 2012 [ref. 28] |
| Included | Risk factors and hallucinations in cannabis users | USA, 49 cases, 51 controls | Case control study | Sevy et al., 2010 [ref. 33] |
| Excluded; very low case number | Psychosis in drug abuse, 19 regular cannabis users | Australia; 881 adolescents 16 y | Cohort study | Hides et al., 2009 [ref. 42] |
| Included | Frequency of cannabis use in schizophrenic patients, 15–54 y | The Hague, The Netherlands | Cohort study | Veen et al., 2004 [ref. 31] |
| Included | Development of depression/anxiety in cannabis users | Australia, 1601 students 14–15 y | Longitudinal cohort study | Patton et al., 2002 [ref. 27] |
3.2. Study Quality
For all studies, the risk of bias was quantified by the criteria published by Moore et al. [ref. 21]. Figure 2 illustrates the results of this study; most studies, including seven of the ten studies selected for the forest plot, have some concern for bias, most often in the randomization domain, with the least concern being for missing data.
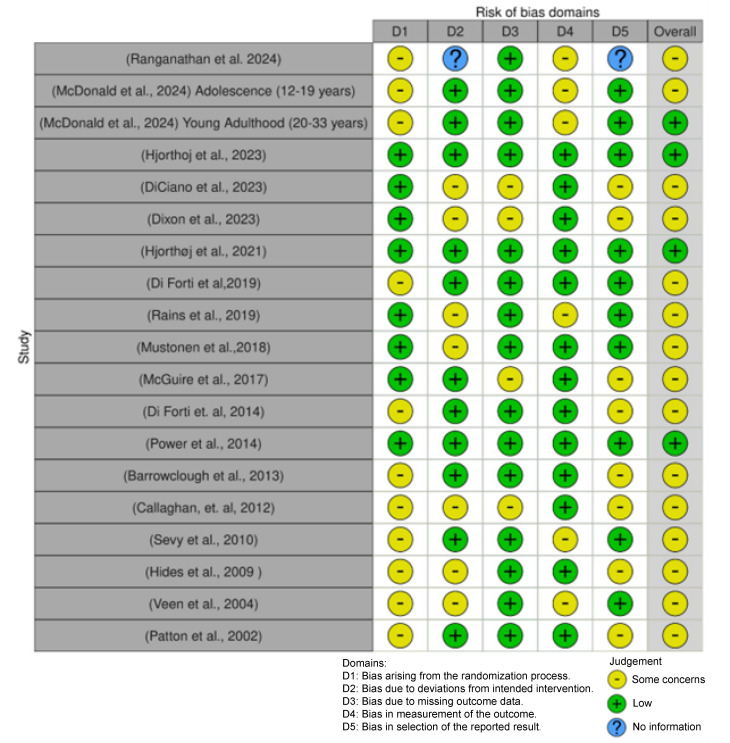
The ten studies included in the forest plot were assessed for their data quality using the GRADE criteria: “risk of bias”, “inconsistency”, “indirectness”, “imprecision” and “quality” [ref. 22]. Table 2 summarizes this analysis, with a low possibility of flaws indicated by a green symbol, a moderate concern identified with a yellow symbol, and insufficient information with a blue symbol.
Table 2: Study quality of studies included in the forest plot.
| Quality | Imprecision | Indirectness | Inconsistency | Risk of Bias | Study |
|---|---|---|---|---|---|
| Serious | Serious | Serious | Not serious | Not serious | McDonald et al., 2024 [ref. 25] |
| Moderate | Not serious | Not serious | Serious | Not serious | Hjorthoj et al., 2023 [ref. 29] |
| Not serious | Not serious | Serious | Serious | Serious | Di Forti et al., 2019 [ref. 38] |
| Not serious | Serious | Serious | Serious | Serious | Mustonen et al., 2018 [ref. 26] |
| Not serious | Not serious | Serious | Serious | Serious | Di Forti et al., 2014 [ref. 30] |
| Moderate | Serious | Not serious | Not serious | Not serious | Power et al., 2014 [ref. 32] |
| Moderate | Serious | Not serious | Not serious | Serious | Callaghan et al., 2012 [ref. 28] |
| Not serious | Serious | Serious | Not serious | Serious | Sevy et al., 2010 [ref. 33] |
| Moderate | Serious | Not serious | Not serious | Serious | Veen et al., 2004 [ref. 31] |
| Not serious | Serious | Serious | Serious | Serious | Patton et al., 2002 [ref. 27] |
Most studies were conducted in the US (4 studies), followed by the UK (3 studies), and then Australia, Canada, and Denmark (2 studies each); one study per country was performed in the other countries, or studies were performed in multiple countries. All studies were located in industrialized countries and included high-quality data. Despite their leading roles in cannabis research and decriminalization of cannabis use, and long-existing markets for medical cannabis, no study was performed in Israel [ref. 44] or Italy [ref. 45].
For the calculation of the forest plot (Figure 3) ten studies were included; since only three studies directly addressed causality of psychosis or affective disorders by cannabis, we combined RCT, case control studies, and cohort studies for this meta-analysis in order to approach the recommended number of studies for a forest plot. The relevant information for these 10 studies is summarized in Table 3.
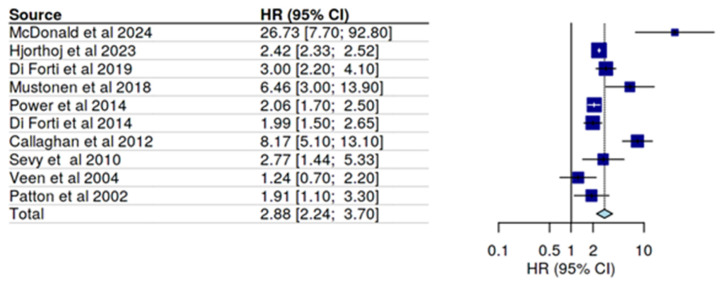
Table 3: Overview of the human studies for cannabis use and psychosis/schizophrenia included in the forest plot (Figure 3).
| Confounders | OR (CI) | Parameter | Criteria/Intervention | Country, Participants | Type of Study, Psychosis Criteria | Study |
|---|---|---|---|---|---|---|
| Sex (m > f), income, race, urban, cigarettes, alcohol, illicit drugs | 12–19 y:26.7 (7.7–92.8)>19 y:1.8 (0.6–5.4) | Physician or ED visit for psychotic disorder | Questionnaire, yes/no last year, Kaplan–Meier analysis | Canada,11,363 part., 12 to 24 y | Longitudinal cohort study, DSM/ICD | McDonald et al., 2024 [ref. 25] |
| Alcohol, drug abuse, psychiatric history | Male: 3.84 (3.43–4.29)female 1.81 (1.53–2.15) | Psychiatric hospitalization | Register study, PARF, incidence rates | Denmark, 7,186,834 population, 16–49 y | Population study, ICD | Hjorthoj et al., 2023 [ref. 29] |
| Prior psychosis, SES | 3.2 (2.2–4.1) | First episode of psychosis | Cannabis use, PARF | EU, Brazil; 901 cases, 1237 control | Case control, ICD-10 | Di Forti et al., 2019 [ref. 38] |
| Alcohol or other drugs, family history, SES | 6.5 (3.0–13.9) | Psychosis | Cannabis use | Northern Finland, 6534 patients, 16 y | Prospective population study, ICD | Mustonen et al., 2018 [ref. 26] |
| Age, ethnicity, SES, illicit drugs | 1.39 (1.11–1.68) | Psychosis | Cannabis use, | London, UK, 410 cases | Cohort study, ICD-10 | Di Forti 2014 [ref. 30] |
| SES, gender | 2 (1.7–2.5) | Schizophrenia | GWAS, logistic regression | Australia, 2082 part. | Twin registry, ICD | Power et al., 2014 [ref. 32] |
| Sex, age, education, income, SES | Male > female, young, income, marital state | Cannabis-use disorder | Cannabis use, logistic regression | BC, Canada, 3339 users | Population study, DSM | Callaghan et al., 2012 [ref. 28] |
| Gender, age, SES, education | 2.77 (1.44–5.33) | Schizophrenia | CUD/non-CUD | USA, 49 cases, 51 controls | Case control, DSM | Sevy et al., 2010 [ref. 33] |
| Age at milestones | 2.5 (1.1–5.0) males/female | Psychosis incidence | Drug use | The Hague, The Netherlands, 70 cases | Cohort study, ICD | Veen et al., 2004 [ref. 31] |
| Alcohol, tobacco, illicit drugs | Daily: ♂ 1.9 (0.93–3.8)♀ 8.6 (4.2–18) | Depression, anxiety | Cannabis use, frequency | Australia, 1601 stu dents 14–15 y | Longitudinal cohort study, DSM | Patton et al., 2002 [ref. 27] |
The forest plot resulted in a determination of an overall OR of 2.88 for the association of psychosis/schizophrenia and cannabis use. By far, the highest risk was calculated by McDonald et al. [ref. 25] who restricted the outcome parameter to ER visits and hospitalizations only, i.e., included in this calculation only severely psychotic events. If all psychotic episodes were included, the OR decreased to 11.2 (CI 4.6–27.3); since the study included a cohort of 11,363 adolescents their impact on the overall OR is high; however, calculation with the lower OR value only slightly lowered the overall OR. Patton et al., [ref. 27] with a low OR as compared to the other studies, used affective symptoms rather than psychosis; excluding this study would slightly increase the overall risk.
Five studies reported OR values separately for men, and four studies for women, allowing an estimate for sex differences. Not all OR values are comparable. McDonald et al. [ref. 25] additionally report an OR for men of 1.22, if women are used as reference. Other studies also found a higher OR for men than for women, with the exception of Patton et al. [ref. 27], indicating a higher OR for women for affective symptoms. More consistent are results for the age subgroups, with most studies including adults older than 18 y (legal age of adulthood). Two studies [ref. 25,ref. 26] included cannabis use in adolescence and found higher OR of 26.7 [ref. 25] and 6.5 [ref. 26], as compared to studies considering cannabis use later in life. This finding suggests an age-dependent effect of cannabis ingredients on the development of psychosis or schizophrenia.
Since research in cannabis effects may be hampered by a multitude of confounders, we performed a funnel plot analysis to search for unreported confounders. The symmetric distribution of data points in the funnel plot indicated no major misrepresentation; many symmetrical outliers indicate confounders like unequal selection and different end-point stringency, and may also represent the combination of case control studies, prospective studies, and cross-sectional cohort studies in this analysis.
3.3. Causality Criteria
Data were examined with the aim of discovering whether they—in addition to a timely correlation—also indicated a causal contribution of cannabis use to later development of psychosis and/or schizophrenia. Hill [ref. 46] proposed nine criteria for causality in epidemiological data, with causality being more likely with a rising number of positive criteria. This approach also is amenable for the cohort studies included in this analysis; its results are summarized in Table 4.
Table 4: Application of Hill criteria to epidemiological support for cannabis causality towards schizophrenia.
| Main Reasons | Causality Support | Criterion |
|---|---|---|
| All studies positive | Strong | Strength of association |
| All studies positive | High | Consistency |
| Different psychosis criteria between studies | Moderate | Specificity |
| Three longitudinal studies | High | Temporality |
| Dose-dependent effects if dose is quantified | High | Biological gradient—dose response |
| No pathophysiological model for psychosis or schizophrenia | Moderate | Plausibility |
| No positive criterion for psychosis | Moderate | Coherence |
| Unethical studies | Lacking | Experiment |
| Similarity with dopamine-induced psychosis | Moderate | Analogy |
Among the nine criteria, five support a causative role of cannabis for psychosis/schizophrenia development; however, experimental proof is lacking since conducting this experiment in humans would be unethical, and no animal model for psychosis exists. Also, plausibility, coherence, and analogy depend on objective clinical criteria in addition to patient reports; the former, however, do not exist for psychotic symptoms. Taken together, the criteria support the causative role of cannabis use/misuse in the development of psychosis.
4. Discussion
A connection between cannabis use and later development of psychotic experiences and schizophrenia has been found in a number of studies. Since correlation is no proof of causation, we tried to further investigate this connection with regard to pathophysiological causation.
The most stringent connection is a direct chronic psychotic reaction to cannabis or THC intake, i.e., the exogenous cannabinoid hypothesis [ref. 43], as has been well documented for acute cannabis effects [ref. 7,ref. 35], and even for low doses in human volunteers [ref. 35]. Adverse reactions reported in clinical trials do not mention psychosis. Psychotic cannabis reactions are comparable to those resulting from other hallucinogenic compounds, like serotonin [ref. 47]; whether these will later develop into schizophrenia—with symptoms occurring without an exogenous trigger—is unclear.
In 2008, Müller-Vahl and Emrich [ref. 48] proposed a “cannabinoid hypothesis” of schizophrenia based on the interplay of the activating dopamine receptor system with the modulating endocannabinoid receptor system, which is compatible with cannabis being a relevant factor in schizophrenia development. Accordingly, Murrie et al. [ref. 49] found a high rate of transition from acute psychosis to schizophrenia, i.e., from acute substance toxicity to disease induction, with cannabis users showing the highest rate among drug users; however, this study did not address the age of drug consumption or the age of disease onset.
Another hypothesis interpreted cannabis use as an indicator for individuals at high risk of developing schizophrenia; this hypothesis has been coined the “shared vulnerability” hypothesis [ref. 50]. Fischer et al. [ref. 51] associated altered risk-taking and risk perception in schizophrenic cannabis users; in this interpretation, cannabis users would underestimate risks; also, cannabis use would serve as an indicator rather than a contributing agent. Similarly, a genetic factor for both schizophrenia [ref. 52,ref. 53,ref. 54] and drug addiction [ref. 55,ref. 56] exists, although in both cases, genome-wide-association studies indicated a polygenetic contribution, with hundreds of genes involved for both schizophrenia and addiction. This makes accidental overlaps likely, and the 3–27 overlapping genes, as identified by Cheng et al. [ref. 57], most likely are arbitrary rather than causal.
The most intriguing result in our analysis is the large age effect (adolescent cannabis users) on chronic psychosis/schizophrenia development. Two studies [ref. 25,ref. 26] recruited adolescents of 14–16 years of age and prospectively studied the occurrence of PLE and schizophrenia over 10 years in relation to cannabis use; both found a high OR, of 26.7 (after adjusting for other risk factors, [ref. 25]) and 6.5 [ref. 26], whereas cannabis use in adulthood (legal age) had a much lower potency for PLE and schizophrenia. This age sensitivity could be explained by cannabinoid effects on pubertal brain development. Based on the small amount of available data, this phenomenon appears to be specific for PLE, since Patton et al. [ref. 27] could not confirm a similar effect for affective disorders like depression or anxiety.
Cannabinoid effects have been found to be dose-dependent. Whereas for acute toxic PLE, this is to be expected, for chronic effects this dose-dependency has not been proven. Contrastingly, Hides ([ref. 42]; data not included in analysis due to a very low case number) found the highest psychotic effects at an intermediate dose, and Di Forti et al. ([ref. 30]; data included from 2019 publication) found increasing OR with earlier drug intake, high THC content, and frequent cannabis use, all indicating a positive dose dependency.
Therefore, we speculate that cannabinoids have two major psychotic effects: acute psychotic sensations comparable to other hallucinogenic drugs and indicative of acute toxicity; and altered synaptic plasticity during adolescence.
The current evidence also supports a higher vulnerability in women, as compared to men [ref. 25,ref. 29].
Therefore, we propose the specific contribution of cannabinoids, especially THC, to the development of schizophrenia. The effect is likely larger when THC exposure occurs in adolescence, compared to prenatal exposure [ref. 58] or adult exposure (this analysis); it may be caused by changes in synaptic plasticity, and it possibly affects specific patient subgroups.
Limitations
Our hypothesis is based on the available epidemiological evidence; the contribution is strengthened by the coherence of data across different study designs and outcome criteria, as well as by a strong support for causality by the Hill criteria. The proposed hypothesis is in accordance with data for brain development, and the comparable effects from other hallucinogens. However, human evidence is limited both in the number of studies as well as their comparability; only two studies directly address causality in a prospective study design. Also, diagnostic criteria vary across studies, with some studies including patients with self-described psychosis-like experiences, while others only consider hospital treatment of schizophrenia. These differences result in a large heterogeneity. Both basic research and epidemiological studies are necessary, especially to confirm adolescence as a specifically critical exposure time, and to determine the impacts of THC content and duration of abuse as criteria for dose dependency.
5. Conclusions
Cannabinoids likely carry a specific contributing risk for the development of schizophrenia and PLE in later life, especially when consumed during adolescence. Preventing cannabis use in this life stage may have a large effect on reducing the burden of psychiatric diseases. Drug prevention programs for adolescents should be strengthened, and the availability of cannabis strictly regulated.
References
- Online World Drug Report 2024—Drug Market Patterns and Trends
- P. Casadio, C. Fernandes, R.M. Murray, M. Di Forti. Cannabis use in young people: The risk for schizophrenia. Neurosci. Biobehav. Rev., 2011. [DOI | PubMed]
- G.W. Hong, A. Sideris, S. Waldman, J.D.O. Stauffer, C.L. Wu. Legal and Regulatory Aspects of Medical Cannabis in the United States. Anesth. Analg., 2024. [DOI | PubMed]
- D.J. Kruger, J. Gerlach, J.S. Kruger, M.A. Mokbel, D.J. Clauw, K.F. Boehnke. Physicians’ Attitudes and Practices Regarding Cannabis and Recommending Medical Cannabis Use. Cannabis Cannabinoid Res., 2024. [DOI | PubMed]
- J. Troyer, K. Tanco. Review of the Use of Medicinal Cannabis Products in Palliative Care. Cancers, 2024. [DOI | PubMed]
- D. Joseph, J. Schulze. Cannabinoid Activity—Is There a Causal Connection to Spasmolysis in Clinical Studies?. Biomolecules, 2021. [DOI | PubMed]
- I. Urits, K. Charipova, K. Gress, N. Li, A.A. Berger, E.M. Cornett, H. Kassem, A.L. Ngo, A.D. Kaye, O. Viswanath. Adverse effects of recreational and medical cannabis. Psychopharmacol. Bull., 2021. [PubMed]
- O. Chan, A. Daudi, D. Ji, M. Wang, J.P. Steen, P. Parnian, C. Li, A. Xiong, W. Zhang, L.C. Lopes. Cannabis Use During Adolescence and Young Adulthood and Academic Achievement: A Systematic Review and Meta-Analysis. JAMA Pediatr., 2024. [DOI | PubMed]
- 9. American Psychiatric Association (APA) Diagnostic and Statistical Manual of Mental Disorders5th ed.American Psychiatric Association (APA)Washington, DC, USA201310.1176/appi.books.9780890425596
- A. Talamo, F. Centorino, L. Tondo, A. Dimitri, J. Hennenm, R.J. Baldessarini. Comorbid substance-use in schizophrenia: Relation to positive and negative symptoms. Schizophr. Res., 2006. [DOI | PubMed]
- J.M. Lappin, S. Darke, M. Farrell. Psychotogenic potential of prescribed drugs. Aust. N. Z. J. Psychiatry, 2023. [DOI | PubMed]
- P. Batel. Addiction and schizophrenia. Eur. Psychiatry, 2000. [DOI | PubMed]
- J.B. Ding, K. Hu. Cigarette Smoking and Schizophrenia: Etiology, Clinical, Pharmacological, and Treatment Implications. Schizophr. Res. Treat., 2021. [DOI | PubMed]
- G. Winterer. Why do patients with schizophrenia smoke?. Curr. Opin. Psychiatry, 2010. [DOI | PubMed]
- A.E. Pinkham, D.L. Penn. Neurocognitive and social cognitive predictors of interpersonal skill in schizophrenia. Psychiatry Res., 2006. [DOI | PubMed]
- K. Kucharska-Pietura, A. David, K. Masia, M. Phillips. Perception of facial and vocal affect by people with schizophrenia in early and late stages of illness. Br. J. Psychiat., 2005. [DOI | PubMed]
- J.Y. Khokhar, L.L. Dwiel, A.M. Henricks, W.T. Doucette, A.I. Green. The link between schizophrenia and substance use disorder: A unifying hypothesis. Schizophr. Res., 2018. [DOI | PubMed]
- D.-M. Ciucă Anghel, G.V. Nițescu, A.-T. Tiron, C.M. Guțu, D.L. Baconi. Understanding the Mechanisms of Action and Effects of Drugs of Abuse. Molecules, 2023. [DOI | PubMed]
- H. Abush, S. Ghose, E.A. Van Enkevort, B.A. Clementz, G.D. Pearlson, J.A. Sweeney, M.S. Keshavan, C.A. Tamminga, E.I. Ivleva. Associations between adolescent cannabis use and brain structure in psychosis. Psychiatry Res. Neuroimaging, 2018. [DOI | PubMed]
- F. Smit, L. Bolier, P. Cuijpers. Cannabis use and the risk of later schizophrenia: A review. Addiction, 2004. [DOI | PubMed]
- T.H.M. Moore, J.P.T. Higgins, K. Dwan. Ten tips for successful assessment of risk of bias in randomized trials using the RoB 2 tool: Early lessons from Cochrane. Cochrane Evid. Synth. Methods, 2023. [DOI]
- Y. Zhang, P. Alonso-Coello, G.H. Guyatta, J.J. Yepes-Nunez, E.A. Akl, G. Hazlewood, H. Pardo-Hernandez, I. Etxeandia-Ikobaltzeta, A. Qaseem, J.W. Williams. GRADE Guidelines: 19. Assessing the certainty of evidence in the importance of outcomes or values and preferences—Risk of bias and indirectness. J. Clin. Epidemiol., 2019. [DOI | PubMed]
- A.K. Akobeng. Understanding systematic reviews and meta-analysis. Arch. Dis. Child., 2005. [DOI | PubMed]
- J.-L. Tang, J.L.Y. Liu. Misleading funnel plot for detection of bias in meta-analysis. J. Clin. Epidemiol., 2000. [DOI | PubMed]
- A.J. McDonald, P. Kurdyak, J. Rehm, M. Roerecke, S.J. Bondy. Age-dependent association of cannabis use with risk of psychotic disorder. Psychol. Med., 2024. [DOI | PubMed]
- A. Mustonen, S. Niemelä, T. Nordström, G.K. Murray, P. Mäki, E. Jääskeläinen, J. Miettunen. Adolescent cannabis use, baseline prodromal symptoms and the risk of psychosis. Br. J. Psychiatry, 2018. [DOI | PubMed]
- G.C. Patton, C. Coffey, J.B. Carlin, L. Degenhardt, M. Lynskey, W. Hall. Cannabis use and mental health in young people: Cohort study. BMJ, 2002. [DOI | PubMed]
- R.C. Callaghan, M. Sanches, S.J. Kish. Quantity and frequency of cannabis use in relation to cannabis-use disorder and cannabis-related problems. Drug Alcohol. Depend., 2020. [DOI | PubMed]
- C. Hjorthøj, W. Compton, M. Starzer, D. Nordholm, E. Einstein, A. Erlangsen, M. Nordentoft, N.D. Volkow, B. Han. Association between cannabis use disorder and schizophrenia stronger in young males than in females. Psychol. Med., 2023. [DOI | PubMed]
- M. Di Forti, H. Sallis, F. Allegri, A. Trotta, L. Ferraro, S.A. Stilo, A. Marconi, C. La Cascia, T. Reis Marques, C. Pariante. Daily use, especially of high-potency cannabis, drives the earlier onset of psychosis in cannabis users. Schizophr. Bull., 2014. [DOI | PubMed]
- N.D. Veen, J.P. Selten, I. van der Tweel, W.G. Feller, H.W. Hoek, R.S. Kahn. Cannabis use and age at onset of schizophrenia. Am. J. Psychiatry, 2004. [DOI | PubMed]
- R.A. Power, K.J. Verweij, M. Zuhair, G.W. Montgomery, A.K. Henders, A.C. Heath, P.A. Madden, S.E. Medland, N.R. Wray, N.G. Martin. Genetic predisposition to schizophrenia associated with increased use of cannabis. Mol. Psychiatry, 2014. [DOI | PubMed]
- S. Sevy, D.G. Robinson, B. Napolitano, R.C. Patel, H. Gunduz-Bruce, R. Miller, J. McCormack, B.S. Lorell, J. Kane. Are cannabis use disorders associated with an earlier age at onset of psychosis? A study in first episode schizophrenia. Schizophr. Res., 2010. [DOI | PubMed]
- C. Hjorthøj, C.M. Posselt, M. Nordentoft. Development Over Time of the Population-Attributable Risk Fraction for Cannabis Use Disorder in Schizophrenia in Denmark. JAMA Psychiatry, 2021. [DOI | PubMed]
- A.M. Aghaei, L.U. Spillane, B. Pittman, L.T. Flynn, J.P. De Aquino, A.B. Nia, M. Ranganathan. Sex differences in the acute effects of oral THC: A randomized, placebo-controlled, crossover human laboratory study. Psychopharmacology, 2024. [DOI | PubMed]
- P. Di Ciano, T.K. Rajji, L. Hong, S. Zhao, P. Byrne, Y. Elzohairy, J.R. Brubacher, M. McGrath, B. Brands, S. Chen. Cannabis and Driving in Older Adults. JAMA Netw. Open., 2024. [DOI | PubMed]
- T. Dixon, K.S. Cadenhead. Cannabidiol versus placebo as adjunctive treatment in early psychosis: Study protocol for randomized controlled trial. Trials, 2023. [DOI | PubMed]
- M. Di Forti, D. Quattrone, T.P. Freeman, G. Tripoli, C. Gayer-Anderson, H. Quigley, V. Rodriguez, H.E. Jongsma, L. Ferraro, C. La Cascia. EU-GEI WP2 Group. The contribution of cannabis use to variation in the incidence of psychotic disorder across Europe (EU-GEI): A multicentre case-control study. Lancet Psychiatry, 2019. [DOI | PubMed]
- S. Johnson, L.S. Rains, S. Marwaha, J. Strang, T. Craig, T. Weaver, P. McCrone, M. King, D. Fowler, S. Pilling. A contingency management intervention to reduce cannabis use and time to relapse in early psychosis: The CIRCLE RCT. Health Technol. Assess., 2019. [DOI]
- P. McGuire, P. Robson, W.J. Cubala, D. Vasile, P.D. Morrison, R. Barron, A. Taylor, S. Wright. Cannabidiol (CBD) as an Adjunctive Therapy in Schizophrenia: A Multicenter Randomized Controlled Trial. Am. J. Psychiatry, 2018. [DOI | PubMed]
- C. Barrowclough, R. Emsley, E. Eisner, R. Beardmore, T. Wykes. Does change in cannabis use in established psychosis affect clinical outcome?. Schizophr. Bull., 2013. [DOI | PubMed]
- L. Hides, D.I. Lubman, J. Buckby, H.P. Yuen, E. Cosgrave, K. Baker, A.R. Yung. The association between early cannabis use and psychotic-like experiences in a community adolescent sample. Schizophr. Res., 2009. [DOI | PubMed]
- M. Ranganathan, P.D. Skosnik, D.C. D’Souza. Marijuana and Madness: Associations Between Cannabinoids and Psychosis. Biol. Psychiatry, 2016. [DOI | PubMed]
- F. Baratta, I. Pignata, L. Ravetto Enri, P. Brusa. Cannabis for Medical Use: Analysis of Recent Clinical Trials in View of Current Legislation. Front. Pharmacol., 2022. [DOI | PubMed]
- S.R. Sznitman. Trends in medical cannabis licensure, Israel, 2013–2018. Drug Alcohol Rev., 2020. [DOI | PubMed]
- A.B. Hill. The environment and disease: Association or causation?. J. R. Soc. Med., 1965. [DOI | PubMed]
- B.B. Quednow, M.A. Geyer, A.L. Halberstadt. Serotonin and schizophrenia. Handbook of Behavioral Neuroscience, 2020. [DOI]
- K.R. Müller-Vahl, H.M. Emrich. Cannabis and schizophrenia: Towards a cannabinoid hypothesis of schizophrenia. Expert. Rev. Neurother., 2008. [DOI | PubMed]
- B. Murrie, J. Lappin, M. Large, G. Sara. Transition of Substance-Induced, Brief, and Atypical Psychoses to Schizophrenia: A Systematic Review and Meta-analysis. Schizophr. Bull., 2020. [DOI | PubMed]
- C. Ksir, C.L. Hart. Cannabis and Psychosis: A Critical Overview of the Relationship. Curr. Psychiatry Rep., 2016. [DOI | PubMed]
- B.A. Fischer, R.P. McMahon, D.L. Kelly, H.J. Wehring, W.A. Meyer, S. Feldman, W.T. Carpenter, D.A. Gorelick. Risk-taking in schizophrenia and controls with and without cannabis dependence. Schizophr. Res., 2015. [DOI | PubMed]
- P. Harrison, D. Weinberger. Schizophrenia genes, gene expression, and neuropathology: On the matter of their convergence. Mol. Psychiatry, 2005. [DOI | PubMed]
- M.G. Henriksen, J. Nordgaard, L.B. Jansson. Genetics of Schizophrenia: Overview of Methods, Findings and Limitations. Front. Hum. Neurosci., 2017. [DOI | PubMed]
- P.F. Sullivan, S. Yao, J. Hjerling-Leffler. Schizophrenia genomics: Genetic complexity and functional insights. Nat. Rev. Neurosci., 2024. [DOI | PubMed]
- J. Cheron, A.D. Kerchove d’Exaerde. Drug addiction: From bench to bedside. Transl. Psychiatry, 2021. [DOI | PubMed]
- M.R. Greener, S.J. Storr. Conflicting theories on addiction aetiology and the strengths and limitations of substance use disorder disease modelling. Front. Mol. Neurosci., 2023. [DOI | PubMed]
- W. Cheng, N. Parker, N. Karadag, E. Koch, G. Hindley, R. Icick, A. Shadrin, K.S. O’Connell, T. Bjella, S. Bahrami. The relationship between cannabis use, schizophrenia, and bipolar disorder: A genetically informed study. Lancet Psychiatry, 2023. [DOI | PubMed]
- A.C. Huizink. Prenatal cannabis exposure and infant outcomes: Overview of studies. Prog. Neuro-Psychopharmacol. Biol. Psychiatry, 2014. [DOI]
