Alternative Approaches and Plant‐Based Remedies for Livestock Health Management Among the Batswana of Southern Africa: A Review
Abstract
Due to limited access to, and the high cost of conventional veterinary services, Batswana communities often rely on ethnoveterinary practices for livestock health management. This review provides an in‐depth analysis on the ethnoveterinary uses, biological properties and safety assessment of plants utilised in livestock husbandry. A systematic literature search was conducted using scientific databases, focusing on articles published from 1997 to 2024. After generating the inventory of plants with ethnoveterinary data, further search was conducted to assess the documented biological activities, safety, and phytochemicals for the recorded plants. A total of 116 plants were documented as remedies for managing nine livestock conditions. The most cited health conditions were retained placenta (81 citations), diarrhoea (65), and wounds (44). The most prominent plants were Senna italica (10 citations), Terminalia sericea (8 citations), and Ziziphus mucronata (8 citations). Approximately 52% of the 116 plants with ethnoveterinary records have empirical data on their biological effect, safety, and phytochemicals. Antimicrobial screening was the most common assay conducted (36%), which dominantly used microbial strains such as Staphylococcus spp., Pseudomonas aeruginosa, and Escherichia coli. We established the vital role of ethnoveterinary practices in Batswana livestock management and the potential of plants in sustainable veterinary care.
Article type: Review Article
Keywords: animal health, antimicrobial, bioactivity, one health, phytochemicals
Affiliations: Indigenous Knowledge Systems Centre Faculty of Natural and Agricultural Sciences North‐West University Mmabatho South Africa; South African Research Chairs Initiative in Indigenous Knowledge‐Driven Medicinal Plants Utilisation and Conservation Strategies for Human, Animal, and Crop Health (IK‐Medplants4HAC), Faculty of Natural and Agricultural Sciences North‐West University Mmabatho South Africa; Agricultural Research Council – Vegetable, Industrial and Medicinal Plants Pretoria South Africa; Phytomedicine Programme, Department of Paraclinical Sciences Faculty of Veterinary Science University of Pretoria Onderstepoort South Africa; Unit For Environmental Sciences and Management, Faculty of Natural and Agriculture Sciences North‐West University Potchefstroom South Africa; School of Agriculture and Science College of Agriculture Engineering and Science University of KwaZulu‐Natal Durban South Africa
License: © 2026 The Author(s). Chemistry & Biodiversity published by Wiley‐VHCA AG. CC BY 4.0 This is an open access article under the terms of the http://creativecommons.org/licenses/by/4.0/ License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Article links: DOI: 10.1002/cbdv.202503248 | PubMed: 41996606 | PMC: PMC13090005
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (4.8 MB)
Introduction
Globally, livestock such as cattle, goats, sheep, chickens, and horses play crucial roles in human life by providing food, generating income, and supplying materials, while also symbolising wealth, and are linked to social standing and cultural heritage. Additionally, they contribute to tourism and employment opportunities [ref. 1, ref. 2, ref. 3]. Livestock husbandry is an integral part of the livelihoods of Batswana communities in southern Africa, providing economic support and cultural significance [ref. 4, ref. 5]. The Batswana are part of the Bantu‐speaking people and found across several countries in southern Africa [ref. 6, ref. 7]. Archaeological evidence suggests that livestock rearing took place from the later Stone Age in southern Africa [ref. 8], with historical link to east Africa [ref. 9]. Livestock have always been significant to the Bantu‐speaking agropastoral people of southern Africa, and still in present times, they remain important commodities used for wealth transfer and are valued in some cultures for their connections with ancestors [ref. 10]. The Batswana were selected as the focus of this review due to their wide geographical distribution across southern Africa, strong livestock‐based livelihoods, and well‐documented reliance on ethnoveterinary medicine [ref. 11]. Despite this, existing knowledge remains fragmented, necessitating a consolidated and critical synthesis. Livestock contribute greatly to food security in rural communities, provide invaluable ecological services, and are also used in traditional rituals [ref. 12].
However, limited access to conventional veterinary services, coupled with their high cost, has led to the widespread reliance on traditional methods for managing livestock health [ref. 13]. These methods, deeply rooted in indigenous knowledge systems, often involve the use of plant‐based remedies to manage a variety of livestock ailments [ref. 14]. Plant‐based remedies have long been recognised as an affordable and sustainable alternative to conventional veterinary medicine [ref. 15]. Among the Batswana, these remedies are employed to manage conditions ranging from reproductive disorders and gastrointestinal problems to respiratory infections and wounds.
Veterinary phytomedicine has long been practiced by indigenous communities worldwide. In sub‐Saharan Africa, its effectiveness has largely been based on oral traditions and practical use rather than formal documentation [ref. 16]. In contrast, other regions such as India have preserved records of traditional veterinary medicine in Ayurvedic texts [ref. 17]. These remedies are believed to have developed through trial and error or by observing animal self‐medication [ref. 17]. Many medicinal plants used in traditional veterinary practices contain bioactive compounds with antimicrobial, antioxidant, anti‐inflammatory, and anti‐parasitic properties, making them valuable for treating infections, wounds, and other livestock health issues [ref. 18]. Southern Africa, which is recognised as a biodiversity hotspot, harbors numerous plant species with potential for veterinary applications [ref. 19, ref. 20]. The secondary metabolites in these plants contribute to animal health and provide a cost‐effective alternative to synthetic drugs. They also help address critical challenges such as antimicrobial resistance and drug residues in animal products [ref. 21, ref. 22]. Despite the widespread use and cultural significance of these remedies, scientific documentation and validation/valorisation of their efficacy remain sparse [ref. 23, ref. 24].
Furthermore, understanding the pharmacological properties of these plants presents an opportunity to develop affordable and accessible veterinary products with proven efficacy and safety [ref. 25]. The current review entails an appraisal of the existing ethnoveterinary knowledge, biological activities, and phytochemical profile of plants used for managing livestock health among the Batswana in southern Africa. By highlighting the strengths and gaps in the current knowledge, the review aims to contribute to the increasing body of evidence supporting sustainable livestock management practices in southern Africa. Additionally, it identifies opportunities for future research into the pharmacological potential of traditional remedies, emphasising the importance of preserving, valorising, and integrating indigenous knowledge into contemporary veterinary medicine.
Methods
The review is based on published ethnoveterinary studies conducted amongst Batswana communities from January 1997 to June 2024. The systematic review is structured according to PRISMA guidelines [ref. 26]. Electronic databases such as Google Scholar, ScienceDirect, and Scopus were used to search for literature. Furthermore, published literature from dissertations, theses, and ethnobotanical books retrieved from the North‐West University online repositories were used in the review. Diverse keywords and phrases were used to access eligible articles. These included “medicinal plants for livestock, livestock health management, Batswana, indigenous knowledge, livestock management, southern Africa, and ethnoveterinary practices”. The Boolean operators of ‘AND’ and ‘OR’ were included to extend the search. Bibliographies of selected articles were also examined to identify further references that might have been omitted from the initial searches. The articles included in this review focused on Batswana communities in southern Africa and explicitly reported the use of ethnoveterinary medicine in managing livestock health care. The collected information included Latin and local names of the plants, plant parts, diseases or conditions treated, preparation methods and mode of administration, and the classification of livestock conditions. Publications were excluded if they focused on modern or non‐plant‐based veterinary practices, were conducted outside southern Africa, did not focus on Batswana communities, or lacked sufficient details on the ethnoveterinary practices and plant species used. Studies not available in English were also excluded. All scientific plant names were verified using the “Plants of the World Online | Kew Science” (https://powo.science.kew.org/).
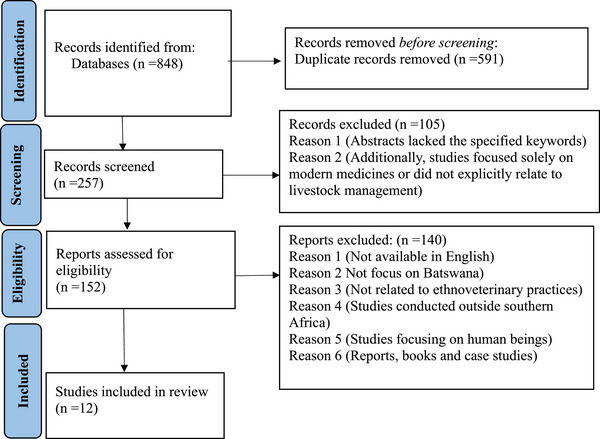
A total of 848 studies were recorded from various scientific databases (Figure 1), which included journal articles, theses, books, and dissertations on the ethnoveterinary studies conducted across southern Africa from 1997 until June 2024. During the screening phase, the titles and abstracts of all the articles were reviewed. A total of 591 duplicate articles were removed after applying the eligibility criteria. Following an additional individual screening of the remaining 257 studies, 105 articles were removed because their abstracts lacked the specified keywords, the studies focused solely on modern medicines, or did not explicitly relate to livestock management. A total of 140 articles were also excluded either because they are not available in English language, did not focus on Batswana, were not related to ethnoveterinary practices, such as those on modern or non‐plant‐based veterinary methods or the studies were conducted outside southern Africa, while the remaining 12 studies were eligible (Figure 1).
To assess the biological activity and safety assessment as well as the phytochemical profiles of plants identified as being used in Batswana ethnoveterinary medicine, a further literature search guided by the generated plant inventory and using the target biological activities as search keywords was undertaken. Journals, books and reports that focused on animal health were considered. The literature was searched using specific keywords on international databases such as Scopus, Web of Science and ScienceDirect.
Results and Discussion
Literature Search Output
In this review, the eligible studies covered two countries namely South Africa and Botswana. In terms of geographical distribution of the eligible ethnoveterinary studies, there were more studies among the Batswana communities in Botswana (58.33%) compared to eligible studies in South Africa (41.67%) (Table 1). Following a detailed analysis, 72.61% of the documented plants were from studies conducted in South Africa while 27.38% of the plants were from Botswana. Even though more studies were conducted in Botswana, the ethnoveterinary practices in South Africa contributed a higher portion in terms of diversity of plant species used for livestock health conditions. This could be attributed to several factors, such as ecological diversity in South Africa contributing to a broader range of medicinal plants, or more comprehensive documentation of plant species within South Africa [ref. 27].
TABLE 1: Overview of reviewed literature on ethnoveterinary plants used in livestock management by the Batswana in southern Africa.
| Author(s) | Title of the study | Country | No. of plants | No. of families | Voucher specimen deposited? | Participants | Livestock treated | Methodological framework |
|---|---|---|---|---|---|---|---|---|
| Chakale et al. [ref. 76] | Ethnoveterinary practices and ethnobotanical knowledge on plants used against cattle diseases among two communities in South Africa | South Africa | 64 | 32 | Yes | Community members | Cattle | Semi‐structured interview, field walk |
| Gabalebatse et al. [ref. 56] | Ethnoveterinary practices amongst livestock farmers in Ngamiland District, Botswana | Botswana | 11 | 9 | Unspecified | Farmers or cattle herders | Cattle | Structured questionnaires |
| Gabanakgosi et al. [ref. 131] | Ethnoveterinary medicine usage in family chickens in the selected four villages of Botswana | Botswana | 6 | 6 | Unspecified | Farmers | Chicken | Structured questionnaires |
| Getchell et al. [ref. 109] | Raising livestock in resource‐poor communities of the North West province of South Africa‐a participatory rural appraisal study | South Africa | 10 | 8 | Unspecified | Farmers | Cattle, sheep, goats and chicken | Questionnaire and focus group |
| Lechani et al. [ref. 53] | Participatory inventory of plant‐based ethnoveterinary medicine used to control internal parasites of goats in the Ngamiland region of Botswana | Botswana | 13 | 11 | Unspecified | Communal farmers | Goats | Structured questionnaires |
| Moichwanetse et al. [ref. 52] | Ethnoveterinary plants used for the treatment of retained placenta and associated diseases in cattle among Dinokana communities, North West Province, South Africa | South Africa | 25 | 18 | Yes | Farmers and herders | Cattle | Semi‐structuredinterview |
| Moreki et al. [ref. 62] | Potential use of ethnoveterinary medicine for retained placenta in cattle in Mogonono, Botswana | Botswana | 14 | 14 | Unspecified | Herd boys and stockowners | Cattle | Rapid Rural Techniques (RRA) |
| Moreki [ref. 171] | Small‐scale poultry production systems in Serowe‐Palapye sub‐district | Botswana | 5 | 3 | Unspecified | Poultry farmers | Poultry | Interviews, focus group, direct observation conference and a seminar |
| Ndou et al. [ref. 64] | Indigenous knowledge and use of medicinal plants for ethnoveterinary within the North West Province, South Africa | South Africa | 31 | 14 | Yes | Farmers, traditional healer, community members | Cattle, sheep, goats and chicken | Semi‐structured interview |
| Setlalekgomo and Setlalekgomo [ref. 63] | The use of ethnoveterinary medicine in goats in Lentsweletau village in Kweneng District of Botswana | Botswana | 13 | 12 | Unspecified | Farmers | Goats | Structured questionnaires |
| Setlalekgomo [ref. 55] | Snakebite management in cattle by farmers in Lentsweletau extension area of Kweneng District in Botswana | Botswana | 4 | 4 | Unspecified | Farmers and cattle herders | Cattle | Structured questionnaires |
| Van der Merwe et al. [ref. 91] | Use of ethnoveterinary medicinal plants in cattle by Setswana‐speaking people in the Madikwe area of the North West Province of South Africa | South Africa | 45 | 24 | Unspecified | Farmers, extension officers, traditional healers, Knowledge holders | Cattle | Rapid Rural Techniques (RRA), group interviews, observation and field walk |
The types of participants involved in each study has significant impact on the scope and depth of generated data [ref. 28]. Farmers (63.63%), community members (18.18%), traditional healers (18.18%), extension officers (9.09%), and knowledgeable elders (9.09%) provide first‐hand knowledge of plant usage in livestock health (Table 1). The expertise of farmers, community members, extension officers, knowledgeable elders, and traditional healers is largely derived from years of experience in livestock management, where they use traditional practices to address various health issues [ref. 29]. Such knowledge brings unique perspectives and practices to the preservation and application of indigenous health systems in managing livestock health. In many parts of Africa, these knowledge sources provide practical and accessible solutions for livestock health, serving as vital resources where modern veterinary services are lacking or not accessible [ref. 28].
Semi‐structured questionnaires (50%), rapid Rural Techniques (20%) and participatory research model (10%) were used to document the methodological framework of the studies in South Africa and Botswana (Table 1). The combination of semi‐structured questionnaires, rapid rural Appraisal (RRA) techniques, and participatory research approaches offers a comprehensive approach to studying ethnoveterinary practices [ref. 30, ref. 31, ref. 32]. These methods enable researchers to gather in‐depth, reliable data while fostering collaboration and inclusivity with Batswana communities [ref. 33]. The different methods provide an excellent opportunity to explore and experiment with various techniques, facilitating the collection of both qualitative and quantitative data [ref. 34]. The dual approach allows researchers to address contemporary theoretical issues surrounding the development, nature, and transmission of ethnobotanical knowledge [ref. 35].
Ethnoveterinary Status of Plant Species Used by the Batswana to Manage Livestock Health Conditions
Using the eligible literature, an analysis on the ethnoveterinary research that focused on the Batswana was conducted. Diverse aspect related to the identified plants and associated indigenous knowledge and practices are elaborated accordingly.
Diversity of Plant Species With Ethnoveterinary Records
A total of 116 plant species from 44 families were recorded as being used in the management of livestock health conditions in South Africa and Botswana (Table 2). Senna italica, Terminalia sericea, Ziziphus mucronata, Peltophorum africanum, Drimia sanguinea, and Aloe ferox were the most cited plant species, representing 37.06% of the generated plant inventory. The plants are reported to be used as multifunctional medicine for the treatment of various livestock diseases, including gastrointestinal infections, respiratory disorders, wound healing, and ectoparasitic infestations. These conditions are among the most frequently cited in ethnoveterinary studies, highlighting the broad‐spectrum use of these plant‐based remedies. Additionally, the health benefits of some of the most cited ethnoveterinary plant species has been demonstrated in other African countries such as Cameroon [ref. 36], Namibia [ref. 37, ref. 38], Ethiopia [ref. 39, ref. 40], and Zimbabwe [ref. 41]. The prevalence of Aloe species in the disease management of Zimbabwean poultry (e.g., wounds, diarrhoea, and ectoparasites) was held to be indicative of efficacy for the plant [ref. 42]. Geographical distribution, availability and health benefits of Aloe species (Aloe ferox, Aloe greatheadii, Aloe marlothii, Aloe vera, Aloe zebrina) in different African regions could be the contributing factor in their common usage for disease management or conditions such as wounds, constipation and retained placenta. The dominance of the Aloe genus illustrates its pharmacological potential, adaptability and broad‐spectrum efficacy as the plants are frequently praised for their anti‐inflammatory, and laxative properties [ref. 43]. The patterns of findings on species such as Aloe sp. align with the patterns observed in South Africa and Botswana, suggesting a shared reliance on specific taxa across different regions of Africa. The relatively high citation frequency of other commonly used plants such as Senna italica and Terminalia sericea demonstrates their perceived effectiveness and suggests their broad applicability in managing livestock conditions. In terms of popularity, plants with high citation frequencies, availability, and versatility in managing multiple conditions emerged as key species across the surveyed regions. About 42% of the recorded plants (Boophone disticha, Boscia albitrunca, Croton gratissimus, Entada elephantina, Gomphocarpus fruticosus, Grewia flava, Grewia flavescens, Hypoxis hemerocallidea and Vachellia karroo) were identified as the most popularly used plants based on their high citation number (3‐5), availability and/or uses (2‐9) in the management of multiple livestock conditions. The high frequency of citation for most used plants could indicate their effectiveness in managing diverse livestock diseases/conditions, considering that these practices in indigenous knowledge have often been refined over time.
TABLE 2: An inventory of ethnobotanical plants used in livestock management by Batswana across southern Africa. The botanical name and families were verified using the Plants of the World Online | Kew Science” (https://powo.science.kew.org/).
| Plant species | Family | Local name | Plant part(s) used | Preparation method | Administration mode | Conditions | References |
|---|---|---|---|---|---|---|---|
| Acokanthera oppositifolia(Lam.) Codd | Apocynaceae | Serekolo | Leaves | Decoction | Oral | Internal parasites | [ref. 53] |
| Acrotome inflata Benth. | Lamiaceae | Mogato | Leaves,Whole plant | Decoction, Burn | Oral, Topical | Cough, wounds | [ref. 76] |
| Roots | Infusion | Oral | Wounds, abscess in livestock | [ref. 64] | |||
| Aloe ferox Mill. | Asphodelaceae | Mokgwapha/ Sekgophana | Leaves | Chopped | Oral | NCD, coccidiosis and respiratory diseases | [ref. 131] |
| Infusion | Internal parasites | [ref. 53] | |||||
| Snakebite control | [ref. 55] | ||||||
| Unspecified | Unspecified | Diarrhoea, Cough | [ref. 63] | ||||
| Unspecified | Worms, Diarrhoea, Constipation | [ref. 109] | |||||
| Aloe greatheadii Schönland | Asphodelaceae | Kgopane e nyane | Leaves | Decoction, Infusion | Oral | Constipation, diarrhoea, retained placenta, ticks, abscesses, wounds, muscle pain | [ref. 76] |
| Decoction | Retained placenta, enhance blood circulation and treating diarrhoea | [ref. 52] | |||||
| Kgophane | Whole plant | Unspecified | Unspecified | Burns, general ailments, blood cleansing, internal parasites, eye infections | [ref. 91] | ||
| Aloe marlothii A. Berger | Asphodelaceae | Unspecified | Unspecified | Unspecified | Unspecified | Unspecified | [ref. 171] |
| Mokgopa | Leaves | Gallsickness, internal and external parasites, diarrhoea, constipation, retained placenta, dystocia, maggots | [ref. 91] | ||||
| Aloe vera (L.) Burm.f. | Asphodelaceae | Kgopane ya thaba | Leaves | Ground, decoction, infusion | Topical, oral | Abscess, wounds, retained placenta, diarrhoea | [ref. 76] |
| Decoction | Oral | Retained placenta, diarrhoea and gala | [ref. 52] | ||||
| Aloe zebrina Baker | Asphodelaceae | Kgophane | Leaves | Infusion, roosted, | Oral, topical | Ripening of abscess, fleas, Gastrointestinal parasites, gala | [ref. 64] |
| Whole plant | Unspecified | Unspecified | Burns, general ailments, blood cleansing, internal parasites, eye infections | [ref. 91] | |||
| Amaranthus blitum L. | Amaranthaceae | Modinakana | Leaves | Infusion | Oral | Blood cleansing, wounds | [ref. 64] |
| Amaranthus cruentus L. | Amaranthaceae | Setlepetlepe | Roots, leaves, whole plant | Poultice, ground | Oral, topical | Abscess, wounds, ear pain | [ref. 76] |
| Modinakana | Whole plant | Ground | Oral | Constipation | [ref. 76] | ||
| Ansellia africana Lindl. | Orchidaceae | Palamela | Unspecified | Unspecified | Unspecified | Diarrhoea | [ref. 56] |
| Roots | Decoction | Oral | Internal parasites | [ref. 53] | |||
| Aptosimum elongatum(Hiern) Engl. | Scrophulariaceae | Ditantanyane | Whole plant | Decoction | Oral | Arthralgia | [ref. 76] |
| Artemisia afra Jacq. ex Willd. | Asteraceae | Lengana | Leaves | Decoction, ground | Oral, topical | Cough, intestinal worms, arthralgia, ear pain | [ref. 76] |
| Infusion | Oral | Cough | [ref. 64] | ||||
| Asparagus africanus Lam. | Asparagaceae | Thokabotswaro | Roots, Stems | Infusion | Oral | Malnutrition | [ref. 76] |
| Asparagus laricinus Burch. | Asparagaceae | Lesitwane | Whole plant | Decoction | Oral | Muscle pain | [ref. 76] |
| Tubers | Unspecified | Unspecified | Sores, redwater, uterine infection, general ailments, umbilical cord inflammation | [ref. 91] | |||
| Mositwasitwane | Roots/nods | Retained placenta | [ref. 62] | ||||
| Asparagus nodulosus (Oberm.) J.‐P. Lebrun & Stork | Asparagaceae | Radipolwane/ polopolwane | Root | Decoction | Oral | Eye infection, retained placenta | [ref. 64] |
| Asparagus suaveolens Burch. | Asparagaceae | Motantanyane | Whole plant | Decoction | Oral | Dystocia | [ref. 76] |
| Lesitwane | Tubers | Unspecified | Unspecified | Sores, redwater, uterine infection, general ailments, umbilical cord inflammation | [ref. 91] | ||
| Babiana hypogaea Burch. | Iridaceae | Thuge | Leaves | Infusion | Oral | Abscess, muscle pain | [ref. 76] |
| Tuber | Diarrhoea | [ref. 64] | |||||
| Boerhavia diffusa L. | Nyctaginaceae | Moetapele | Leaves, Stems | Decoction | Topical | Eye infection, abscess, wounds | [ref. 76] |
| Boophone disticha Herb. | Amaryllidaceae | Leswama | Bulb | Decoction | OralOral | Fracture, post‐abortion, retained placenta | [ref. 64] |
| Lesoma/ Mathubadudifala | Leaves, Roots, Bulb | Constipation | [ref. 76] | ||||
| Lesoma (Legwama) | Roots, Leaves | Maceration | Retained placenta and wound healing | [ref. 52] | |||
| Matubadifala | Bulb scales | Unspecified | Unspecified | Abortion | [ref. 91] | ||
| Unspecified | Retained placenta, gall sickness | [ref. 109] | |||||
| Boscia albitrunca(Burch.) Gilg & Benedict | Capparaceae | Motlopi | Leaves, Roots | Decoction, ground | Oral | Internal parasites | [ref. 53] |
| Unspecified | [ref. 131] | ||||||
| Unspecified | Unspecified | Retained placenta | [ref. 62] | ||||
| Bark | Anthrax | [ref. 56] | |||||
| Eye diseases | [ref. 63] | ||||||
| Boscia foetida Schinz | Capparaceae | Mopipi | Leaves | Ground | Unspecified | Eye problems | [ref. 56] |
| Bulbine abyssinica A. Rich. | Asphodelaceae | Makgabenyane | Leaves | Ground | Topical | Abscess, wounds | [ref. 76] |
| Unspecified | Unspecified | Unspecified | Gall sickness, worms | [ref. 109] | |||
| Roots | Infusion | Oral | Blood cleansing, internal sores | [ref. 64] | |||
| Burkea africana Hook. | Fabaceae | Monato | Bark | Unspecified | Unspecified | Retained placenta | [ref. 62] |
| [ref. 63] | |||||||
| Cadaba aphylla (Thunb.) Wild | Capparaceae | Monnamontsho | Roots | Decoction | Oral | Blood cleansing | [ref. 64] |
| Cannabis sativa L. | Cannabaceae | Motekwane | Leaves | Decoction | Oral | Anthelmintic | [ref. 64] |
| Capsicum annuum L. | Solanaceae | Pherehere | Leaves/fruit | Chopped | Oral | Unspecified | [ref. 131] |
| Cassia abbreviata Oliv. | Fabaceae | Unspecified | Unspecified | Unspecified | Unspecified | Unspecified | [ref. 171] |
| Centella asiatica (L.) Urb. | Apiaceae | SetimamoleloSetimamolelo | Leaves, whole plant | Poultice, decoction | Topical, oral | Wound, abscess, eye infection, diarrhoea | [ref. 76] |
| Whole plant | Maceration | Oral | Retained placenta | [ref. 52] | |||
| Cleome gynandra L. | Cleomaceae | Rothwe | Flower, leaves, roots | Ground | Topical | Eye infection, ear problem, cough, constipation, intestinal worms | [ref. 76] |
| Colophospermum mopane (J. Kirk ex Benth.) J. Léonard | Fabaceae | Mophane | Bark, leaves | Infusion, decoction | Oral | Internal parasites | [ref. 53] |
| Combretum hereroenseSchinz | Combretaceae | Tsholakhudu | Leaves | Decoction | Oral | Cough, pains, dysentery, constipation | [ref. 76] |
| Combretum imberbe Wawra | Combretaceae | Unspecified | Unspecified | Unspecified | Unspecified | Fleas, mites, ticks | [ref. 171] |
| Croton gratissimus Burch | Euphorbiaceae | Moologa | Flower | Ground | Topical | Eye infection, ear problem | [ref. 76] |
| Leaves | Fertility enhancement | [ref. 64] | |||||
| Leaves, Roots | Unspecified | Unspecified | Pneumonia, fertility enhancement | [ref. 91] | |||
| Croton megalobotrys Müll.Arg. | Euphorbiaceae | Unspecified | Leaves | Unspecified | Topical | Lumpy skin | [ref. 56] |
| Cucumis myriocarpusNaudin | Cucurbitaceae | Monyaku | Fruit | Infusion | Oral | Vomiting, general malaise (gala) | [ref. 64] |
| Dichrostachys cinerea (L.)Wight & Arn. | Fabaceae | Moselesele | Bark | Poultice | Topical | Retained placenta, dystocia, fracture, arthralgia | [ref. 76] |
| Roots | Topical | Retained placenta, dystocia, fracture | [ref. 52] | ||||
| Dicoma galpinii F.C. Wilson | Asteraceae | Tlhlonya | Roots | Infusion | Oral | Diarrhoea, blood cleansing | [ref. 64] |
| Dicoma macrocephala DC. | Asteraceae | Tlhonya | Roots | Infusion | Oral | Diarrhoea | [ref. 76] |
| Diospyros lycioides Desf. | Ebenaceae | Motlhajwa/letlhajwa | Roots | Decoction | Oral | Snakebite control | [ref. 55] |
| Dracaena hyacinthoides (L.) Mabb. | Asparagaceae | Moshokelatsebe | Leaves, whole plant | Poultice, decoction | Topical, oral | Retained placenta, diarrhoea, constipation | [ref. 76] |
| Drimia sanguinea (Schinz) Jessop | Asparagaceae | Sekaname | Bulb | Infusion | Oral | Retained placenta, intestinal worms, constipation | [ref. 76] |
| Poultice | Oral | General ailments, general intestinal diseases, internal parasites, blood cleansing, gallsickness, heartwater, redwater, sores, retained placenta | [ref. 91] | ||||
| Roots | Retained placenta, uterus, blood cleaning | [ref. 52] | |||||
| Snakebite, heartwater | [ref. 64] | ||||||
| Unspecified | Unspecified | Foot rot | [ref. 63] | ||||
| Gallsickness, worms | [ref. 91] | ||||||
| Unspecified | |||||||
| Dysphania ambrosioides(L.) Mosyakin &Clemants | Amaranthaceae | Tlhatlhabadimo | Whole plant | Infusion | Oral | Cough, constipation | [ref. 76] |
| Ehretia rigida Druce | Boraginaceae | Morobe | Roots | Unspecified | Unspecified | Fractures | [ref. 91] |
| Elaeodendron transvaalense (Burtt Davy) R.H. Archer | Celastraceae | Mojelemane | Bark | Decoction | Oral | Diarrhoea | [ref. 76] |
| Unspecified | Unspecified | [ref. 91] | |||||
| Englerophytum magalismontanum (Sonder)T.D.Penn. | Sapotaceae | Motlatswa | Roots | Unspecified | Unspecified | Fertility enhancement | [ref. 91] |
| Entada burkei (Benth.) S.A. O’Donnell & G.P. Lewis | Fabaceae | Mositsane | Roots, Bark | Decoction, ground | Oral, topical | Cough, constipation, retained placenta, diarrhoea | [ref. 76] |
| Entada elephantina (Burch.) S.A. O’Donnell & G.P. Lewis | Fabaceae | MosetlhaneMositsaneBosetsana | Root‐stock | Unspecified | Unspecified | Diarrhoea, heartwater, coughing, pneumonia | [ref. 91] |
| Bulb | Retained placenta | [ref. 62] | |||||
| Roots | Poultice | Topical | Retained placenta, intestinal para sites, enhance blood circulation | [ref. 52] | |||
| Leaves | Decoction | Oral | Internal parasites | [ref. 53] | |||
| Rhizome | Infusion | Blood cleansing | [ref. 64] | ||||
| Euclea undulata Thunb. | Ebenaceae | Morobe | Leaves, bark, roots | Poultice, decoction | Topical, oral | Wounds, cough, constipation, retained placenta diarrhoea, arthralgia | [ref. 76] |
| Euphorbia balbisii Boiss. | Euphorbiaceae | Lwetsane | Leaves, Roots | Decoction | Oral | Diarrhoea, intestinal worms | [ref. 76] |
| Euphorbia inaequilateraSond. | Euphorbiaceae | Loetsane | Roots | Infusion | Unspecified | Eye problems | [ref. 56] |
| Euphorbia regis‐jubae Webb & Berthel. | Euphorbiaceae | Mosimama/Mosiama | Branches | Ground | Oral, topical | Snakebite control | [ref. 55] |
| Euphorbia serpens Kunth | Euphorbiaceae | Luetsane | Roots | Decoction | Oral | Blood cleansing | [ref. 64] |
| Gomphocarpus fruticosus (L.) W.T. Aiton | Apocynaceae | Motimola/ sebogamaswi | Whole plant | Infusion | Oral | Constipation, retained placenta, cough, bile reflux | [ref. 76] |
| Motimola | Maceration | Retained placenta, pain alleviation | [ref. 52] | ||||
| Sebogamashi | Roots | Decoction | Retained placenta, gala, respiratory diseases | [ref. 64] | |||
| Grewia flava DC. | Malvaceae | Moretlwa | Roots | Infusion | Oral | Diarrhoea, dystocia | [ref. 76] |
| Decoction | Diarrhoea | [ref. 64] | |||||
| Unspecified | Unspecified | Fertility enhancement | [ref. 91] | ||||
| Grewia flavescens Juss. | Malvaceae | Mokgompata | Unspecified | Unspecified | Unspecified | Diarrhoea | [ref. 56] |
| Mokgomphatha | Roots | Foot rot | [ref. 63] | ||||
| Motsotsojane | Leaves | Infusion | Oral | Pain, wounds, diarrhoea | [ref. 76] | ||
| Leaves, Roots | Internal parasites | [ref. 53] | |||||
| Harpagophytum procumbens (Burch.) DC.ex Meisn. | Pedaliaceae | Sengaparile | Roots | Unspecified | Unspecified | Mange | [ref. 63] |
| Lematla, Sengaparile | Fruit | Decoction, ground | Oral, topical | Retained placenta | [ref. 91] | ||
| Tuber, roots, leaves, fruit | Dystocia, pain after birth, abscess, fracture, muscle pain, retained placenta | [ref. 76] | |||||
| Helichrysum candolleanum H.Buek | Asteraceae | PhateyangakaPhate ya ngaka | Roots, leaves, fruit | Decoction | Oral | Retained placenta | [ref. 76] |
| Unspecified | Unspecified | Unspecified | Fowl pox, swelling of the head | [ref. 109] | |||
| Helichrysum paronychioides DC. | Asteraceae | Phateyangaka | Roots | Infusion | Oral | Cough, blood cleansing, pain, diarrhoea | [ref. 64] |
| Hermannia guerkeana K. Schum. | Malvaceae | Moreba | Roots | Unspecified | Unspecified | Retained placenta | [ref. 62] |
| Hypoxis hemerocallidea Fisch., C.A. Mey. & Avé‐Lall. | Hypoxidaceae | Maledu/Tshuku ya poo | Whole plant | Decoction | Oral | Cough, dystocia, arthralgia, constipation | [ref. 76] |
| Unspecified | Unspecified | Unspecified | Gall sickness | [ref. 109] | |||
| Corms | Fertility enhancement, general ailments, heartwater, abortion | [ref. 91] | |||||
| Bulb | Poultice | Topical | Retained placenta, anaemia | [ref. 52] | |||
| Hypoxis rigidula Baker | Hypoxidaceae | Tsuku‐ya‐poo | Corms | Unspecified | Unspecified | Fertility enhancement, general ailments, heartwater, abortion | [ref. 91] |
| Indigofera cryptantha Benth. ex Harv. | Fabaceae | Kofi | Roots | Decoction | Oral | Diarrhoea | [ref. 64] |
| Ipomoea oblongata E. Mey. ex‐Choisy | Convolvulaceae | Mokatelo | Roots | Decoction | Oral | Cough, wounds, muscle pain, diarrhoea | [ref. 76] |
| Jatropha zeyheri Sond. | Euphorbiaceae | Seswagadi | Roots | Maceration | Topical | Eye infections, constipation, retained placenta | [ref. 76] |
| Poultice | Retained placenta, blood cleansing and kidney stone | [ref. 52] | |||||
| Kleinia longiflora DC. | Asteraceae | MosimamaMosiama | Whole plant | Poultice | Topical | Eye infection | [ref. 76] |
| Ground | Fracture | [ref. 64] | |||||
| Lasiosiphon capitatus (Lam.) Burtt Davy | Thymelaeaceae | Mokaikai | Unspecified | Unspecified | Unspecified | Diarrhoea | [ref. 56] |
| Roots, leaves | Decoction, infusion | Oral | Internal parasites | [ref. 53] | |||
| Lippia scaberrima Sond. | Verbenaceae | Mosukutswane | Leaves | Decoction | Oral | Cough | [ref. 76] |
| Lycianthes biflora (Lour.) Bitter | Solanaceae | Makgonatsotlhe | Roots | Infusion | Oral, topical | Intestinal worms | [ref. 76] |
| Oral | Internal parasites | [ref. 53] | |||||
| Malva neglecta Wallr. | Malvaceae | Tikamotse | Leaves, flowers | Decoction | Oral | Constipation, wounds, abscess, cough | [ref. 76] |
| Malvastrum coromandelianum (L.) Garcke | Malvaceae | Thobega | Leaves | Decoction | Oral | Diarrhoea, abscess, wounds, ear pain | [ref. 76] |
| Mentha aquatica L. | Lamiaceae | Kgobedimetsing | Leaves | Decoction | Oral | Cough | [ref. 76] |
| Moringa oleifera Lam. | Moringaceae | Unspecified | Leaves | Ground | Oral | Unspecified | [ref. 131] |
| Whole plant | Unspecified | Unspecified | Cough | [ref. 63] | |||
| Nicotiana tabacum L. | Solanaceae | Motsoko | Leaves | Grounded | Oral | NCD, coccidiosis and respiratory diseases | [ref. 131] |
| Unspecified | Unspecified | Eye infections | [ref. 91] | ||||
| Tobacco | Worms, foaming from the mouth | [ref. 109] | |||||
| Internal parasites, eye diseases | [ref. 63] | ||||||
| Opuntia ficus‐indica (L.) Mill. | Cactaceae | Toorofeye | Leaves, stem, flowers | Decoction, Ground | Oral, topical | Diarrhoea, constipation, eye infections, retained placenta, abscess | [ref. 76] |
| Flower | Poultice | Topical | Retained placenta | [ref. 52] | |||
| Osyris lanceolata Hochst. & Steud. | Santalaceae | Mpera | Bulb | Maceration | Oral | Retained placenta, alleviation of pain, internal bleeding | [ref. 52] |
| Ozoroa paniculosa (Sond.) R. Fern. & A.Fern. | Anacardiaceae | MonokanaMonokane | Roots | Decoction | Oral | Cough, muscle pain | [ref. 76] |
| Unspecified | Unspecified | Retained placenta | [ref. 62] | ||||
| Bark, rootbark | Diarrhoea, redwater, sweating sickness | [ref. 91] | |||||
| Peltophorum africanum Sond. | Fabaceae | MosetlhaMosetlaUnspecified | Roots, bark | Decoction | Oral | Wounds, muscle pain, diarrhoea, constipation | [ref. 76] |
| Roots, leaves, bark | Internal parasites | [ref. 53] | |||||
| Leaves, Bark | Poultice | Topical | Retained placenta diarrhoea and removal of blood clots from the skin | [ref. 52] | |||
| Roots | Unspecified | Unspecified | Retained placenta | [ref. 62] | |||
| Bark, rootbark | Diarrhoea | [ref. 91] | |||||
| Unspecified | Unspecified | Fleas, mites, ticks | [ref. 171] | ||||
| Phyllanthus maderaspatensis L. | Phyllanthaceae | Mositwane | Whole plant | Ground, Decoction | Topical, Oral | Eye infection, constipation, diarrhoea | [ref. 76] |
| Phyllanthus parvulus var. garipensis (Müll.Arg.) Radcl.‐Sm. | Phyllanthaceae | Lentsane | Aerial parts | Unspecified | Unspecified | Eye infections | [ref. 91] |
| Phyllanthus parvulus Sond. | Phyllanthaceae | Lentsane | Aerial parts | Unspecified | Unspecified | Eye infections | [ref. 91] |
| Plumbago zeylanica L. | Plumbaginaceae | Masegomabe | Whole plant | Decoction | Oral | Cough, intestinal worms | [ref. 76] |
| Roots | Unspecified | Unspecified | Pneumonia | [ref. 91] | |||
| Portulaca oleracea L. | Portulacaceae | Selele | Whole plant | Decoction | Oral | Constipation, eye infection, muscle pain, wounds, intestinal worms | [ref. 76] |
| Pouzolzia mixta Solms | Urticaceae | Mongololo | Roots, leaves | Maceration, decoction, infusion | Oral | Retained placenta, diarrhoea, constipation | [ref. 76] |
| Unspecified | Unspecified | Retained placenta, bloat, vaginal discharge | [ref. 91] | ||||
| Roots | Poultice | Topical | Retained placenta, uterus cleansing | [ref. 52] | |||
| Unspecified | Unspecified | Retained placenta | [ref. 62] | ||||
| Rhoicissus tridentata (L.f.) Wild & R.B. Drumm. | Vitaceae | Ntagaraga | Tubers | Unspecified | Unspecified | Heartwater, redwater, internal parasites, general ailments, abortion | [ref. 91] |
| Ricinus communis L. | Euphorbiaceae | Mokhura | Leaves | Infusion | Oral | Constipation, eye infection | [ref. 76] |
| Seeds | Unspecified | Unspecified | Constipation, internal parasites | [ref. 91] | |||
| Scadoxus puniceus (L.)Friis & Nordal | Amaryllidaceae | Sekaname | Roots | Unspecified | Unspecified | Retained placenta | [ref. 62] |
| Schkuhria pinnata (Lam.)Kuntze ex Thell. | Asteraceae | SantlhokoSanthloko, Lefero | Whole plant | Ground | Topical | Eye infection, wounds, abscess | [ref. 76] |
| Aerial parts | Unspecified | Unspecified | Eye infections, pneumonia, diarrhoea, heartwater | [ref. 91] | |||
| Sclerocarya birrea Hochst. | Anacardiaceae | Morula | Barks | Unspecified | Unspecified | Diarrhoea, fracture | [ref. 91] |
| Searsia lancea (L.f.) F.A. Barkley | Anacardiaceae | MoshabelaMoshabele | Roots, leaves, stem | Poultice, infusion | Oral | Abscess, constipation, diarrhoea | [ref. 76] |
| Roots, bark | Unspecified | Unspecified | Diarrhoea, gallsickness | [ref. 91] | |||
| Searsia pyroides (Burch.) Moffett | Anacardiaceae | Bohitlha | Leaves | Decoction | Oral | Cough, dystocia, constipation, diarrhoea. intestinal worms, arthralgia | [ref. 76] |
| Roots | Poultice | Topical | Retained placenta | [ref. 52] | |||
| Securidaca longepedunculata Fresen. | Polygalaceae | Mmaba | Roots | Ground | Topical | Cough, dystocia, constipation, muscle pain | [ref. 76] |
| Seddera suffruticosaHallier f. | Convolvulaceae | Thobega | Roots | Unspecified | Unspecified | Fracture | [ref. 91] |
| Senecio consanguineus DC. | Asteraceae | Unspecified | Whole plant | Decoction | Oral | Cough, wounds, constipation | [ref. 76] |
| Senna italica Mill. | Fabaceae | Sebetebete/Sebete/Okatare | Leaves, bark | Decoction | Oral | Constipation, abscess, anthrax, aphosphorosis, lung diseases | [ref. 76] |
| Unspecified | Roots | Poultice | Topical | Retained placenta, pain alleviation | [ref. 52] | ||
| Unspecified | Unspecified | Gallsickness, general intestinal diseases, heartwater, anthrax, pneumonia | [ref. 91] | ||||
| Whole plant, roots | Diarrhoea, retained placenta | [ref. 64] | |||||
| Roots, Whole plant | Pasteurollosis, diphtheria | [ref. 63] | |||||
| Unspecified | Infusionchopped | Liver disease, gallsickness | [ref. 109] | ||||
| Calf diphtheria | [ref. 56] | ||||||
| Unspecified | [ref. 171] | ||||||
| Monyokololo | Gall sickness, worms | [ref. 109] | |||||
| Leaves/roots | Unspecified | [ref. 131] | |||||
| Senna tora (L.) Roxb. | Fabaceae | Mongepenpe | Whole plant | Poultice | Topical | Retained placenta, growth of scrotum | [ref. 52] |
| Sesamum eriocarpum (Decne.) Byng &Christenh. | Pedaliaceae | Makanangwane | Roots | Unspecified | Unspecified | Retained placenta | [ref. 62] |
| Tshetlho ya mibitlae mebedi | Whole plant | Poultice | Topical | [ref. 63] | |||
| Retained placenta, dystocia, general ailments | [ref. 91] | ||||||
| Retained placenta, flea eradication | [ref. 52] | ||||||
| Tshetlho ya mamitlwa a mabedi | Leaves, whole plantroots | Poultice, infusion | Topical, oral | Blackquarter, retained placenta, dystocia | [ref. 76] | ||
| Makanangwane | Unspecified | Unspecified | Retained placenta | [ref. 62] | |||
| Solanum campylacanthum Hochst. ex A. Rich. | Solanaceae | Tolwane enyaneTholwane e nyane | Roots, leaves | Infusion, maceration | Oral | Diarrhoea, eye infection | [ref. 76] |
| Roots | Decoction | Blood cleansing | [ref. 64] | ||||
| Solanum dimidiatum Raf. | Solanaceae | Mohato | Fruit sap | Unspecified | Unspecified | Diarrhoea | [ref. 91] |
| Solanum albidum Dunal | Solanaceae | Tolwana | Roots | Unspecified | Unspecified | Sores | [ref. 91] |
| Solanum lichtensteinii Willd. | Solanaceae | Tolwane | Whole plant | Poultice | Topical | Ticks | [ref. 76] |
| Flower, roots | Retained placenta | [ref. 52] | |||||
| Tholwane | Roots | Infusion | Oral | Blood cleansing, gastrointestinal parasites | [ref. 64] | ||
| Spirostachys africana Sond. | Euphorbiaceae | Morukuru | Bark | Unspecified | Unspecified | Retained placenta | [ref. 62] |
| [ref. 63] | |||||||
| Morekhure | Stem | Sweating sickness | [ref. 91] | ||||
| Tarchonanthus camphoratusHoutt. ex DC. | Asteraceae | Moologa | Leaves | Maceration | Oral | Retained placenta, wounds, dystocia | [ref. 52] |
| Tarchonanthus camphoratus L. | Asteraceae | Moologa | RootsLeaves | Infusion | Oral | Internal parasites | [ref. 53] |
| Mohatlha | Intestinal worms | [ref. 76] | |||||
| Cold | [ref. 64] | ||||||
| Terminalia sericea Burch.ex DC. | Combretaceae | Mogonono | Leaves, stem | Decoction | Oral | Cough | [ref. 76] |
| Roots | Poultice | Topical | Retained placenta, uterus cleansing | [ref. 52] | |||
| Unspecified | Unspecified | Diarrhoea | [ref. 63] | ||||
| [ref. 91] | |||||||
| [ref. 56] | |||||||
| Root bark | Retained placenta | [ref. 62] | |||||
| Leaves | [ref. 62] | ||||||
| Unspecified | Infusion | Oral | Internal parasites | [ref. 53] | |||
| Teucrium trifidum Retz. | Lamiaceae | Lethe la noga | Leaves, roots | Decoction | Oral | Cough, diarrhoea, constipation | [ref. 76] |
| Whole plant | Unspecified | Maintenance of pregnancy after abortion | [ref. 64] | ||||
| Thamnosma rhodesica (Baker f.) Mendonça | Rutaceae | Moralala | Whole plant | Unspecified | Unspecified | Contagious abortion | [ref. 63] |
| Thesium viridifolium Levyns | Santalaceae | Motlhogapele | Whole plant | Decoction | Oral | Diarrhoea | [ref. 64] |
| Tribulus terrestris L. | Zygophyllaceae | TshetlhoTsetlhoTshetlo | Leaves, whole plant | Ground | Oral | Arthralgia, | [ref. 76] |
| Whole plant | Poultice | Topical | Retained placenta, wound healing, dystocia | [ref. 52] | |||
| Unspecified | Unspecified | Retained placenta, bloat | [ref. 91] | ||||
| Triumfetta sonderii Ficalho & Hiern | Malvaceae | Mokuku | Rootbark | Unspecified | Unspecified | Retained placenta | [ref. 91] |
| Vachellia karroo (Hayne) Banfi & Galasso | Fabaceae | MookaMookana | Bark | Decoction | Oral | Lumpy skin disease | [ref. 76] |
| Bulb | Maceration | Retained placenta, bacterial infection | [ref. 52] | ||||
| Bark | Unspecified | Unspecified | Fractures, diarrhoea | [ref. 91] | |||
| Root, bark | Ground | Topical | Fracture | [ref. 64] | |||
| Vachellia tortilis (Forssk.) Galasso &Banfi | Fabaceae | Mosu | Branch tips | Unspecified | Unspecified | Diarrhoea | [ref. 91] |
| Vitex zeyheri Sond. ex Schauer | Lamiaceae | Mokwele | Leaves | Unspecified | Unspecified | Eye infections | [ref. 91] |
| Withania somnifera (L.) Dunal | Solanaceae | ModikasopeMokukwane | Roots | Infusion | Oral | Internal sores | [ref. 64] |
| Unspecified | Unspecified | Diarrhoea | [ref. 91] | ||||
| Ximenia americana L. | Olacaceae | Moretologana | Unspecified | Unspecified | Unspecified | Diarrhoea | [ref. 56] |
| Seretologa | Roots | Internal parasites | [ref. 91] | ||||
| Ziziphus mucronata Willd. | Rhamnaceae | Mokgalo/Sekgalo | Leaves | Poultice | Topical | Abscess | [ref. 64] |
| Roots | Retained placenta | [ref. 52] | |||||
| Decoction | Oral | Snakebite control | [ref. 55] | ||||
| Unspecified | Unspecified | Retained placenta | [ref. 62] | ||||
| Unspecified | Diarrhoea | [ref. 56] | |||||
| Roots, leaves | Fertility enhancement, sores, burns | [ref. 91] | |||||
| Decoction, ground | Oral, topical | Dystocia, diarrhoea, arthralgia, wounds, foot rot | [ref. 76] | ||||
| Decoction, infusion | Oral | Internal parasites | [ref. 53] | ||||
| Ziziphus oxyphyllaEdgew. | Rhamnaceae | Mokgalo fatshe | Roots | Decoction | Unspecified | Diarrhoea | [ref. 109] |
| Sekgalofatshe | Poultice | Topical | Retained placenta, increase stimulation for separating retained placenta | [ref. 52] | |||
| Ziziphus zeyherianaSond. | Rhamnaceae | Sekgalofatshe/Mokgalofatshe | Roots | Decoction | Oral | Blood cleansing, pain | [ref. 64] |
| Sekgalo‐fatshe | Leaves, branches | Unspecified | Unspecified | Diarrhoea, internal parasites, general ailments | [ref. 91] |
Distribution of Plant Families Used to Manage Livestock Health Conditions
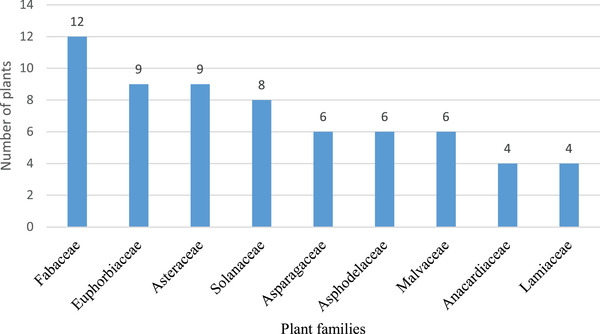
The recorded 116 plants were distributed within 44 families with the Fabaceae (12), Euphorbiaceae (9), Asteraceae (9), Solanaceae (8), Asparagaceae (6), Asphodelaceae (6) and Malvaceae (6) having the highest cited number of plants used to manage livestock conditions among Batswana people in southern Africa (Figure 2 and Table 2). Similarly, the high utilisation of the Fabaceae in managing different livestock conditions has been reported in ethnobotanical reviews or studies conducted in Africa [ref. 44, ref. 45, ref. 46]. The top 10 families comprised 56.89% of the total cited plants, while the remaining (42.24%) plants were represented within 34 other families. Furthermore, 84.09% of the families had relatively low representation averaging 1–4 plant species per family. The prevalent use of the Fabaceae family may likely be attributed to its broad distribution, high species richness, and diverse bioactive compounds known for their pharmacological properties [ref. 47, ref. 48, ref. 49]. This diversity reflects the broad spectrum of traditional plant knowledge across southern Africa, where various families are utilised for their specific benefits in livestock health management.
Pattern of Plant Parts Used, Preparation, and Route of Administration Methods
A total of 15 plant parts were used for treating livestock diseases among Batswana in southern Africa (Table 2). The most common plant parts used were roots (33%), leaves (26%), and whole plant (12%) (Figure 3A, Table S1). The popularity of roots as one of the most preferred plant parts has led to significant conservation challenges. The harvest of underground parts as a practice is often unsustainable, causing irreversible damage to plant populations and contributing to the risk of species decline which can lead to extinction [ref. 50, ref. 51]. The dominance of root usage in Batswana ethnoveterinary practices may be attributed to their belief in the strength and vitality that the earth imparts to these underground parts [ref. 52, ref. 53, ref. 54]. Roots and leaves are the most frequently used, reflecting traditional preferences for these accessible and widely applicable plant parts [ref. 13, ref. 55, ref. 56].
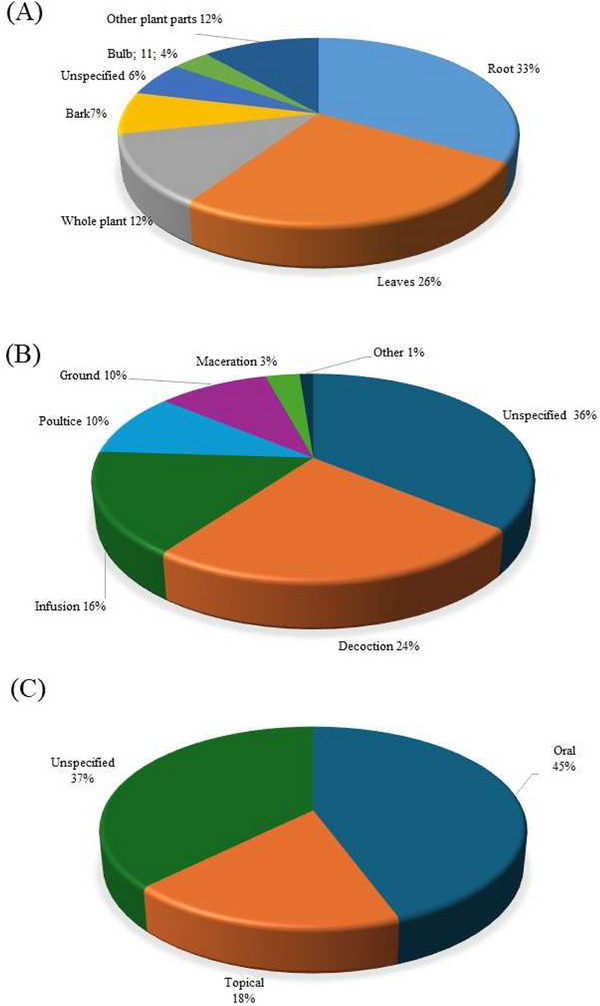
The methods of preparing medicinal plants for livestock conditions in southern Africa highlight a range of traditional techniques tailored to different health conditions [ref. 57]. Decoctions (24%) and infusions (16%) were the most common preparation methods used for medicinal plants among Batswana in southern Africa (Figure 3B). Decoction entails boiling the plant materials while infusion involves pouring cold/hot/warm water onto the plant material and allowing the mixture to steep and cool. Furthermore, poultices (10%) are primarily used for external treatments including wound care, skin infections and inflammation. This method involves crushing plant materials and applying them directly to the affected area. Grinding of the plant materials constituted 10% of the reported preparation methods. On the other hand, methods of preparation such as maceration (3.4%), burning (0.38%) and roasting (0.38%) were generally low (Table S2). It is important to highlight that a significant portion (36%) of the plant preparations do not have the specific method used. This could be due to traditional practices where the method is considered implicit or universally understood within the communities.
In southern African ethnoveterinary practices, the mode of administering medicinal plants varies significantly, with oral administration (44%) and topical applications (19%) being the most frequently cited practices (Figure 3C). Oral administration is favoured for treatments targeting internal ailments (e.g., organ damage, inflammation, infections), and topical applications is a localised practice used for conditions such as wounds and skin infections, offering a targeted approach for external relief [ref. 58]. The high percentage of unspecified administration (37%) suggests some flexibility in traditional practices, where the method may depend on the practitioner’s preference or the circumstances of each treatment. This distribution of administration routes highlights the adaptability and specificity of traditional livestock health condition treatment approaches [ref. 59].
Livestock Health Conditions Treated With Plants by the Batswana in Southern Africa
A total of 58 livestock conditions identified were categorised into 10 major groups (Table 3). The classification of the different diseases was based on the studies by Chakale et al. [ref. 60] and Ndou [ref. 61], with some slight modifications. Some of the dominant categories included, reproduction disorders (121), gastrointestinal problems (97), and skin problems (74). On the other hand, treatment of conditions such as eye problems and musculoskeletal systems were relatively lower in significance. This may reflect a lower prevalence of these issues or the possibility that such conditions are managed through other methods or external interventions beyond traditional plant‐based remedies. Retained placenta (81), diarrhoea (65), and wounds (44) were the most cited conditions managed within the livestock conditions (Table 3). Among these, retained placenta emerged as the most cited health condition. This underscores the importance of managing reproductive health in livestock, as issues such as retained placenta can significantly affect the productivity and reproductive efficiency of animals [ref. 15, ref. 52, ref. 62]. The frequent citation of gastrointestinal disorders shows the critical need for remedies to ensure digestive health, as poor digestion can lead to reduced nutrient absorption, weight loss, and lower productivity in livestock [ref. 56, ref. 63]. Skin problems were also prominent, with eight conditions cited, including wounds. The high number of references to wound treatment suggests that topical application of plant‐based remedies is a crucial aspect of traditional veterinary care [ref. 14, ref. 64]. The wound‐related problems may reflect the challenges posed by injuries sustained during grazing, handling, or attacks by predators, making wound care an essential aspect of livestock management [ref. 13].
TABLE 3: Livestock health conditions managed with medicinal plants by the Batswana in southern Africa.
| Category of the conditions | Conditions | Number of plants used |
|---|---|---|
| Eye problem | Eye infection | 19 |
| Conjunctivitis | 3 | |
| Blindness | 1 | |
| Fertility/reproduction disorders | Retained placenta | 81 |
| Dystocia | 11 | |
| Abortion | 10 | |
| Fertility problems | 4 | |
| Uterus cleansing | 3 | |
| Bloat | 2 | |
| Pain after birth | 1 | |
| Growth of scrotum | 1 | |
| Urinary infections | 1 | |
| Vaginal discharge | 1 | |
| Gastrointestinal problems | Diarrhoea | 65 |
| Constipation | 29 | |
| Bile reflux | 6 | |
| Gastrointestinal parasites | 2 | |
| Dysentery | 1 | |
| Kidney stone | 1 | |
| General system infection | Blood cleansing | 13 |
| Anthrax | 6 | |
| Black quarter | 6 | |
| Ear pain | 5 | |
| Enhance blood circulation | 2 | |
| Aphosphorosis | 1 | |
| Malnutrition | 1 | |
| Weating sickness | 1 | |
| Internal and external parasites | Internal parasites | 19 |
| Helminths | 13 | |
| Babesiosis | 6 | |
| Intestinal worms | 6 | |
| Anaemia | 3 | |
| Coccidiosis | 2 | |
| Newcastle Disease (NCD) | 2 | |
| Bacterial infection | 1 | |
| Musculoskeletal systems | Fracture | 13 |
| Pain | 12 | |
| Arthralgia | 7 | |
| Respiratory problems | Cough | 26 |
| Pneumonia | 3 | |
| Respiratory diseases | 3 | |
| Diphtheria | 2 | |
| Heart problem | 1 | |
| Lung diseases | 1 | |
| Skin problem | Wounds | 44 |
| Abscess | 16 | |
| Sores | 5 | |
| Foot rot | 4 | |
| Lumpy skin | 2 | |
| Pasteurollosis | 1 | |
| Mange | 1 | |
| Myiasis | 1 | |
| Snakebite | Snakebite control | 5 |
| Tick‐borne | Anaplasmosis | 23 |
| Cowdriosis | 8 | |
| Ticks | 6 | |
| Fleas | 4 | |
| Mites | 2 | |
| Heartwater | 2 | |
| Unspecified | Unspecified | 10 |
The widespread use of medicinal plants reflects both their accessibility and cultural significance, demonstrating how southern African communities have developed adaptive strategies for livestock disease management. Research underscores the potential of medicinal plants in ethnoveterinary practices, emphasising the need for systematic evaluations of their biological and pharmacological effects [ref. 65, ref. 66]. Further investigation into the efficacy and safety of these plant‐based treatments could enhance their application and facilitate their integration into sustainable livestock management practices. Additionally, systematic documentation and conservation efforts are essential to ensure the continued availability of these medicinal plant species for future generations [ref. 67].
Cultural Significance of Local Names for Plants Among the Batswana of Southern Africa
Among the Batswana communities in South Africa and Botswana, plants play a crucial role in ethnoveterinary medicine, with local names serving as key identifiers in traditional healing practices. These indigenous names encapsulate generations of botanical knowledge, reflecting the deep relationship between the people and their environment [ref. 68]. Local nomenclature provides valuable insights into plant characteristics, including their medicinal applications, ecological adaptations and distinctive morphological and sensory features such as size, shape, taste, smell and habitat [ref. 69].
The naming of medicinal plants among the Batswana is rooted in observation and cultural significance, with each name often describing a particular attribute or use of the plant. For instance, Senna italica (Sebete/Sebetebete) is named based on its use as a purgative to treat digestive disorders in livestock, while Hypoxis hemerocallidea (Tshuku ya poo) is recognised for its immune‐boosting properties and treatment of infections in cattle. Similarly, Grewia flava (Moretlwa) is applied to wounds due to its antibacterial effects, and Aloe ferox (Mokgwapha) is valued for alleviating respiratory infections in goats and cattle (Table 2).
The classification of medicinal plants among the Batswana communities in South Africa and Botswana is often inconsistent. In some instances, a single plant species may be identified by multiple local names within the same region. For example, Entada elephantina is known by three different names in various areas of South Africa and Botswana which are Mosetlhane, Mositsane and Bosetsana. Conversely, a single local name can be used to describe multiple plant species, leading to potential confusion in plant identification and application. For instance, the name Sekgalofatshe is associated with different species, including Ziziphus oxyphylla and Ziziphus zeyheriana.
Biological Activity, Safety Status and Phytochemicals of Medicinal Plants With Ethnoveterinary Records Among the Batswana of Southern Africa
After establishing the inventory of 116 plants used by the Batswana to manage their livestock, existing evidence on the biological effects, safety assessments and the phytochemicals of these botanicals were assessed. This was essential to identify plants with empirical data and potential for further research especially on their valorisation.
Biological Activity of Medicinal Plants With Ethnoveterinary Records
Herbal remedies are the oldest form of medication, generally used as multi‐target agents. As of 2024, approximately 3 780 plants have been recorded for medicinal purposes in South Africa [ref. 70]. However, there are no recent updates on the number of plants used in ethnoveterinary medicine [ref. 71]. Over 60 plants used by the Batswana people have been previously analysed for biological properties related to ethnoveterinary and their phytochemical composition (Tables 4, 5, 6, 7, 8, 9, Figures 4, 5, 6). Traditional medicine covers interdisciplinary research which involves observation, description and conducting experimental analysis of the identified medicinal plants for drug discovery. During the observation stages, plant part usage is crucial as medicinal plants have different kinds of bioactive compounds that accumulate in specific organs at different concentrations. As depicted in Figure 5, leaves were the most studied plant parts (46%) due to their availability, accessibility and plant conservation concerns, even though their frequency of use was 26% in the ethnoveterinary surveys reported (Figure 3A). Availability of plant materials and the complexities of bioactive compounds contribute to the high use of leaves [ref. 72]. In the documented ethnoveterinary surveys, roots were the most frequently used (33%) plant part by the Batswana communities, yet leaves accounted for 11% of the plant parts studied (lower than bark usage, 12%) in biological, safety and phytochemical analysis. The low use of seeds and fruits may be attributed to their seasonal availability [ref. 73]. Similar findings of limited usage of fruits and seeds were observed in the literature surveys.
TABLE 4: Summary of reported antimicrobial activity of plants used by Batswana for livestock health management.
| Plant species | Plant part(s) Used | Extraction solvent | Bioassay | Strains tested | Summary of findings | References |
|---|---|---|---|---|---|---|
| Acrotome inflata Benth | Fruit | Ethanol | Microplate serial dilutionAgar disk diffusion | Candida albicans | Fruit extracts were effective against Candida species with MIC = 1.25 mg/mL. In the disk diffusion assay, extracts had an inhibition zone of ±2 mm against C. albicans.Control: Fungazole and Ampicillin | [ref. 90] |
| Acrotome inflata Benth | Whole plant | Ethanol, water | Agar disk diffusion | Staphylococcus aureus, Pseudomonas aeruginosa | Ethanolic extracts (whole plant) with concentrations of 5 mg/mL were effective against P. aeruginosa (8.7 mm inhibition zone) strain, with 10 mg/mL and 20 mg/mL demonstrating an inhibition zone of ± 11 mm against B. subtilis.Control: Streptomycin and Penicillin G | [ref. 172] |
| Aloe ferox Mill. | Leaves | Methanol, water | Broth microdilution assay | Staphylococcus aureus, Escherichia coli, Enterococcus faecalis, Bacillus cereus, Bacillus pumilus, Pseudomonas aeruginosa, Klebsiella pneumoniae, Enterococcus cloacae | Methanol extracts were active against S. aureus and E. coli at 5 mg/mL MIC.Control: Tetracycline | [ref. 129] |
| Aloe marlothii A. Berger | Leaves | DCM, methanol, acetone, hexane, ethanol | Microplate serial dilution TLC Bioautography | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, Staphylococcus aureus, Mycobacterium aurum | All extracts had efficacy against the tested pathogens, with hexane extracts having the highest MIC value (2.5 mg/mL) against all the pathogens. Methanol extract also had an MIC value of 2.5 mg/mL against E. faecalis strain. Acetone, DCM and methanol extracts had significant activity against the test pathogens, with MIC values ranging from 0.028 mg/mL (acetone extract against S. aureus) and 0.625 mg/mL for methanol extracts against E. coli. Leaf extracts exhibit anti‐mycobacterial activity, MIC = 2.5 mg/mLControl: Gentamycin | [ref. 89, ref. 90] |
| Aloe zebrina | Leaves | DCM, hexane, acetone, methanol | Microplate serial dilution, bioautography | Escherichia coli, Enterococcus faecalis, Pseudomonas aeruginosa, Staphylococcus aureus | Hexane extracts showed the highest activity (2.5 mg/mL) against the tested pathogens. Acetone and methanol extracts showed good activity (0.039 mg/mL and 0.625 mg/mL) against S. aureus and E. coli, respectively.Control: Gentamycin | [ref. 89] |
| Ansellia africana Lindl. | Roots, stem | DCM, acetone, DCM‐methanol, water | Agar diffusion assay | Klebsiella pneumonia, Staphylococcus aureus, Mycobacterium smegmatis, Pseudomonas aeruginosa | Acetone root extracts inhibited growth of all studied strains, with the highest inhibition zone demonstrated against S. aureus (19.3 mm). Root and stem water extracts were not active against the test strains. Root and stem extracts from DCM were as only effective against K. pneumonia and S. aureus.Control: Ciprofloxacin | [ref. 173, ref. 174] |
| Artemisia afra Jacq. ex Willd. | Aerial parts | Ethanol extract | In vitroMicroplate serial dilution In vivo Oral administration of extract | Salmonella enterica subsp. enterica serovar Typhi, Salmonella enterica subsp. enterica serovar Enteritidis | In vitroExtracts inhibited S. enterica strains and the activity was recorded at 156 µg/mL, and more than 50% biofilm reduction for all the strains.In vivoExtracts had a significant reduction in bacterial load of rats show when tested at 200 and 300 mg/kg/bw. | [ref. 95] |
| Artemisia afra Jacq. ex Willd. | Leaves | Water, DCM | In vitroFirefly bioluminescence assay (via optical densitometry at 600 nm)In vivoOral administration of extract | Mycobacterium tuberculosis H34RvMycobacterium aurum | In vitroThe inhibitory activity of the DCM extract exhibited an IC50 = 270 mg/mL when tested against Mycobacterium aurum and an IC50 = 290 mg/mL for M. tuberculosis. The Mycobacterium aurum replication was inhibited by 200 µg/mL of water (>25%), methanol (<25%), and DCM extracts (41,4%). Control: Isoniazid (20 µg/mL).In vivoNo observed improvement of pulmonary burden and spleen burden, indicating no in vivo mycobacterial activity | [ref. 96] |
| Asparagus laricinus Burch. | Stem, leaves | Water | Agar dilution method | Staphylococcus aureus, Staphylococcus saprophyticus, Enterobacter cloacae | Leaf extracts exhibited antibacterial activity against S. aureus and B. subtilis (MIC = 1 mg/mL) as well as S. saprophyticus and E. cloacae (MIC = 0.125 mg/mL).Control: Chloramphenicol | [ref. 175] |
| Bulbine abyssinica A. Rich. | Leaves, rhizome, roots | Methanol | Agar well diffusion | Staphylococcus aureus, Escherichia coli | The leaf extracts and stems had the greater inhibition against S. aureus (inhibition zones of 19.33 and 15 mm respectively), than E. coli (inhibition zones of 13.67 and 14.67 mm, respectively). The roots had higher inhibition against E. coli (13.67 mm) than against S. aureus (12.67 mm)Negative control: DMSO | [ref. 176] |
| Bulbine abyssinica A. Rich. | Whole plant | Acetone, water | Agar well diffusion assay Microplate serial dilution assay | Pseudomonas aeruginosa, Staphylococcus aureus, Enterococcus faecalis, Klebsiella pneumonia, Serratia marcescens | The acetone and water extracts inhibited bacterial growth, particularly the inhibition zones for E. faecalis (35 and 41 mm respectively) are greater that the inhibition zone for the positive control (30.67 mm). The inhibition zones for extracts against P. aeruginosa, S. aureus, K. pneumonia, and S. marcescens strains were lower than the inhibition zone for the positive control. Control: Amoxicillin (0.0125 mg/mL) | [ref. 39] |
| Bulbine latifolia (L.f.) Spreng. | Leaves | Methanol | Microplate assayAntitubercular rapid radiometric assay | Citrobacter, Klebsiella pneumonia, Staphylococcus aureus, Candida albicans, Microsporum audouini, Mycobacterium smegmatis | The 10 mg/mL extract was effective against Citrobacter (MIC = 625 µg/mL), C. albicans (MIC = 625 µg/mL), and M. audouinii (MIC = 312.5 µg/mL).Controls: Nyastatin (fungi), Gentamycin (bacteria), and Ciprofloxacin & isoniazid (M. smegmatis) | [ref. 177] |
| Cannabis sativa L. | Leaves | Hexane, DCM, ethyl acetate, ethanol, water | Agar well diffusion assay | Bacillus cereus, Salmonella enterica | The extracts were effective against B. cereus stain (MIC = 2 mg/mL). | [ref. 178] |
| Cassia abbreviata Oliv. | Stem Bark | Ethanol, water | Agar disc diffusion | Escherichia coli,Staphylococcus aureus | Ethanol extracts showed no inhibitory activity against E. coli at all tested concentrations (1, 5, 10, 15, 20 mg/mL). Control: Ciprofloxacin (5 µg) | [ref. 179] |
| Cassia abbreviata Oliv. | Stem bark | Ethanol, water Soxhlet (cold ethanol, cold water, DCM, Trichloromethane (TCM): ethanol) | Agar well diffusion assay | Pseudomonas aeruginosa, Klebsiella pneumonia, Candida albicans | Water extract was active against P. aeruginosa (46.88 µg/mL). The TCM extract was active against K. pneumonia with an MIC = 46.88 µg/mL. The ethanol extracts showed activity against C. albicans with an MIC = 93.75 µg/mL.Controls: Ceftriaxone, Ciprofloxacin, Fluconazole | [ref. 92] |
| Cassia abbreviata Oliv. | Leaf, stem bark, root bark | Ethanol | Microplate serial dilution assay | Escherichia coli, Salmonella paratyphi, Klebsiella pneumoniae, Shigella sonnei, Enterobacter cloacae, Pseudomonas aeruginosa, Staphylococcus aureus, Enterococcus faecalis | Root bark extracts showed good activity in all test strains (0.31 ‐ 1.25 mg/mL). Stem bark extracts exhibited good activity with MIC values of 0.63 ‐ 1.25 mg/mL. The leaf extracts were least active against K. pneumoniae and E. faecalis (MIC = 2.50 mg/mL), and highly active against the other test strains (MIC = 0.31 ‐ 0.63 mg/mL). | [ref. 180] |
| Colophospermum mopane (J. Kirk ex Benth.) J. Léonard | Bark | Water, methanol | Microplate serial dilution | Staphylococcus aureus, Escherichia coli | Water (7.71 mg/mL) and methanol (5.99 mg/mL) extracts were active against S. aureus and E. coli (12.1 mg/mL and 7.86 mg/mL, respectively).Control: Gentamycin and Ampicillin | [ref. 181] |
| Colophospermum mopane (J.Kirk ex Benth.) J.Léonard | Bark, leaves | Water, ethanol | Disc agar diffusion assay | Staphylococcus aureus, Pseudomonas aeruginosa, Candida albicans | Leaf (20 mg/mL) and bark water extract (5 mg/mL &10 mg/mL) were effective against S. aureus and P. aeruginosa, respectively. All ethanol extracts were active against the tested bacterial strains.Control: Penicillin G | [ref. 172] |
| Combretum hereroense Schinz | Leaves | Methanol | Microplate serial dilution | Bacillus cereus, Escherichia coli, Klebsiella pneumoniae, Salmonella typhimurium, Staphylococcus aureus, Enterococcus faecalis, Pseudomonas aeruginosa | The extracts were active against all the test strains, with an average MIC of >1.75 mg/mL.Control: Ciprofloxacin (0.01 mg/mL) | [ref. 155] |
| Combretum hereroense Schinz | Leaves | Acetone, hexane, DCM, methanol | Microplate serial dilution | Candida albicans, Cryptococcus neoformans, Aspergillus fumigates, Sporothrix schenkii, Microsporum cannis | All extracts were highly active against the fungal strains after 24 and 48‐h periods. Extract activity was calculated at 0.39 mg/mL (acetone), 0.6 mg/mL (hexane), 0.67 mg/mL (DCM) and 0.24 mg/mL(methanol).Control: Amphotericin B | [ref. 182] |
| Combretum imberbe (Wawra) | Leaves | Acetone, hexane, DCM, methanol | Microplate serial dilution | Candida albicans, Cryptococcus neoformans, Aspergilllus fumigates, Sporothrix schenkii, Microsporum cannis | The extracts were active against all the test fungal strains after 24 and 48‐h period. Acetone and methanol extracts were active against C. albicans (>2.5 mg/mL) while DCM, acetone and hexane extracts were active against S. schenkii (2.5 mg/mL). Control: Amphotericin B | [ref. 182] |
| Combretum imberbe (Wawra) | Leaves | Methanol | Microplate serial dilution | Bacillus cereus, Escherichia coli, Klebsiella pneumoniae, Salmonella typhimurium, Staphylococcus aureus, Enterococcus faecalis, Pseudomonas aeruginosa, Staphylococcus epidermidis | The extracts were active against all the test strains, with an average MIC = 0.24 mg/mL.Control: Ciprofloxacin (0.01 mg/mL) | [ref. 155] |
| Croton gratissimus Burch. | Leaves | Methanol | Microplate serial dilutionAgar disc diffusion assay | Staphylococcus aureus, Staphylococcus epidermis, Staphylococcus aureus | The highest minimum inhibition zone of extracts was observed against for S. aureus (20 mm) and hospital isolate S. epidermidis (27 mm).Control: Cloxacillin | [ref. 86] |
| Croton gratissimus Burch. | Leaves | Ethanol | Microplate serial dilution | Candida albicans, Mycobacterium aurum | Extracts were effective against Mycobacterium aurum (2.5 mg/mL) and C. albicans (3.5 mg/mL).Control: Fungazole and Ampicillin | [ref. 90] |
| Dicerocaryum eriocarpum (Decne.) Abels(Syn: Sesamum eriocarpum (Decne.) Byng & Christenh.) | Roots | Ethanol | Microplate serial dilution | Candida albicans, Mycobacterium aurum | Root extracts were active against C. albicans (4.5 mg/mL) and Mycobacterium aurum (0.156 mg/mL).Control: Fungazole and Ampicillin | [ref. 90] |
| Dichrostachys cinerea (L.) Wight & Arn. | Roots | Ethanol | Microplate serial dilution | Candida albicans, Mycobacterium aurum | The extract showed activity against C. albicans at 2 mg/mL and Mycobacterium aurum at 0.156 mg/mL.Control: Fungazole and Ampicillin | [ref. 90] |
| Diospyros lycioides Desf. | Leaves | Hexane, acetone, ethyl acetate, methanol | Bioautography | Pseudomonas aeruginosa, Staphylococcus aureus, Enterococcus faecalis | Ethyl acetate and acetone extracts were active against across the test strains. Whereas the methanol and hexane extracts exhibited antibacterial activity against S. aureus and E. faecalis as well as E. faecalis, respectively. | [ref. 130] |
| Drimia sanguinea (Schinz.) Jessop | Bulb | Methanol, petroleum ether | Microplate serial dilution | Bacillus cereus, Candida albicans, Candida glabrata, Trichophyton tomsurans | The MIC value for methanol extracts against Candida albicans was the lowest at 1.56 mg/mL. | [ref. 88] |
| Elephantorrhiza elephantina (Burch) Skeels | Roots | Ethanol | Microplate serial dilutions | Candida albicans, Bacillus cereus, Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, Mycobacterium aurum | The extracts were active against all the test strains, with the highest MIC value of 3 mg/mL.Control: Fungazole and Ampicillin | [ref. 90] |
| Elephantorrhiza elephantina (Burch) Skeels | Rhizomes | Methanol, petroleum ether | Microplate serial dilution | Bacillus cereus, Candida albicans, Trichophyton tonsurans | Methanol extracts showed activity against B. cereus (20 mg/mL), Candida albicans (20 mg/mL), and T. tonsurans (10 mg/mL). Controls: Neomycin (antibacterial) and Amphotericin B (antifungal) | [ref. 88] |
| Euphorbia serpens Kunth | Shoots, roots | Ethanol, methanol, DCM, petroleum ether | Disc diffusion assay | Salmonella typhi, Streptococcus pneumoniae, Enterococcus faecalis, Aspergillus fumigatus,Candida albicans, Fusarium oxysporum | Ethanol extracts showed the strongest activity, except with the inhibition of E. faecalis, F. oxysporum and S. pneumoniae. Petroleum ether extracts were mainly effective against Candida albicans. | [ref. 183] |
| Gomphocarpus fruticosus (L.) W.T.Aiton | Aerial parts, fruits | Hexane, methanol | Microplate serial dilution assay | Staphylococcus aureusm Enterococcus faecalis, Klebsiella pneumoniae, Mycobacterium smegmatis | The antibacterial activity for aerial extracts was >250 µg/mL. Hexane fruit extracts had good activity against E. faecalis (125 µg/mL) and P. aeruginosa (31 µg/mL).Controls: Gentamicin, Rifampicin, Vancomycin | [ref. 184] |
| Grewia flava DC. | Twig, roots | Hexane, acetone, distilled water | Agar well diffusion methodMicroplate serial dilution assay | Pseudomonam aeruginosa, Staphylococcus aureus, Escherichia coli | Microbial inhibition from all the extracts was positive against all the test strains. Control: Chloramphenicol | [ref. 185] |
| Grewia flava DC. | Roots | Acetone | BioautographyMicroplate serial dilution assay | Candida albicans, Cryptococcus neoformans, Staphylococcus aureus, Proteus mirabilis, Moraxella catarrhalis, Klebsiella pneumoniaeBacillus cereus, Proteus vulgaris, Mycobacterium smegmatis, Mycoplasma hominis, Escherichia coli, Pseudomonas aeruginosa | Acetone extracts had the lowest average MIC value (247 µg/mL), hexane extracts have the highest average MIC value (923 µg/mL).Control: Amphotericin B, Vancomycin and Strepromycin | [ref. 51] |
| Helichrysum paronychioides DC. | Whole plant | Methanol, petroleum ether | Microplate serial dilution | Bacillus cereus, Candida albicans, Trichophyton tonsurans | The methanol extract had noteworthy antimicrobial activity against B. cereus and T. tonsurans (0.39 mg/mL). Petroleum ether extracts also exhibited good activity against S, flexneri (0.1 mg/mL).Controls: Neomycin (antibacterial) and Amphotericin B (antifungal) | [ref. 88] |
| Jatropha zeyheri Sond | RootsLeaves | Acetone, methanol, ethyl acetate | Microplate serial dilution assay | Escherichia coli, Pseudomonam aeruginosa, Enterobacter cloacae, Klebsiella pneumoniae, Serrattia marscens, Samonella spp., Staphylococcus aureus, Bacillus cereus, Bacillus pumilus | The extracts had clear inhibition zones for the test strains indicating activity. Methanol leaf extracts showed poor activity (>12.5 mg/mL) against E. coli and P. aeruginosa, similar to the activity of acetone leaf extract against S. marscens. Methanol root extracts also have the same activity MIC value (>12.5 mg/mL) against S. aureus, B. pumilus and A. calcaoceutical.Control: Neomycin, Metronidazole | [ref. 51] |
| Malva neglecta Wallr. | Leaves | Methanol, water | Disc agar diffusion assay, Well agar diffusion | Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, Proteus mirabilis, Staphylococcus aureus | Methanol extracts inhibited growth of S. aureus and K. pneumonia. | [ref. 186] |
| Malvastrum coromandelianum (L.) Garcke | Leaves | Methanol | Microplate serial dilution | Mycobacterium fortuitum, Mycobacterium smegmatis, Mycobacterium aurum | Extracts inhibited microbial growth of M. fortuitum and Mycobacterium aurum (0.63 mg/mL) as well as M. segmantis (0.31 mg/mL). Control: Streptomycin and Rifampicin | [ref. 126] |
| Malvastrum coromandelianum (L.) Garcke | Candida albicans | Ethyl acetate extract at 10 µg/mL inhibited Candida albicans (8 mm) while ethanol extract at 10 µg/mL inhibited Candida albicans (7 mm).Control: Fluconazole | [ref. 13] | |||
| Nicotiana tabacum L. | Leaves | Ethyl acetate | Agar well diffusion assay | Pseudomonam aeruginosa, Klebsiella pneumonia, Staphylococcus aureus, Salmonella enterica subsp. enteric serotype Typhi, Micrococcus sp., Proteus mirabilis, Klebsiella sp., Escherichia coli | Extract activity against S. aureus was 500 µg/mL, with an inhibition zone of approximately (160 mm). | [ref. 187] |
| Opuntia ficus‐indica (L) Mill | Fruits | Ethanol | Disc diffusion assay | Staphylococcus aureus, Bacillus cereus, Listeria monocytogenes, Escherichia coli, Salmonella typhi | The inhibition zones of 50 mg/mL were 17 mm against S. typhi and 35 mm for B. cereus. The lowest tested concentration (3.12 mg/mm) only inhibited B. cereus (3.12 mm)Control: Cyclohexane | [ref. 186] |
| Osyris lanceolata Hochst. & Steud. | Leaves | Methanol | Disc diffusion assay | Escherichia coli, Staphylococcus aureus | S. aureus inhibition for the smallest tested concentration (1 mg disc) was 6,0 mm. E. coli inhibition for the smallest tested concentration (1 mg disc) was 6,17 mm.Ampicillin and Ciprofloxacin | [ref. 188] |
| Osyris lanceolata Hochst. & Steud. | Bark | DCM, water, ethyl acetate | Microplate serial dilution | Proteus mirabilis, Klebsiella pneumonia, Salmonella typhi, Escherichia coli, Pseudomonas aeruginosa | The extracts showed some activity against the test strains (6.25 and 12.5 mg/mL). Control: Gentamicin | [ref. 189] |
| Ozoroa paniculosa var. paniculosa | Leaves | Acetone, hexane fractions, DCM, ethanol, butanol | Microplate serial dilution | Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, Enterococcus faecalis, Aspergilus fumigatus, Candida albicans, Cryptococcus neoformans | Water extracts activity against fungal strains ranged from 625 ‐ 2500 µg/mL. Extracts exhibited activity (1250 µg/mL) against E. coli and E. faecalis. The lowest activity (19 µg/mL) was observed in hexane and DCM fractions against S. albicans and E. faecalis.Control: Amphotericin B (0.78–6.25 µg/mL); gentamicin (0.39–1.56 µg/mL). | [ref. 190] |
| Peltophorum africanum Sond. | Stem, bark | Ethyl acetate | Well diffusion assay | Staphylococcus aureus, Pseudomonas aeruginosa, Aeromonas hydrophila, Shigella sonnei, Salmonella typhimurium, Aspergillus flavus, Candida albicans, Cryptococcus neoformans | The extracts were inactive against A. flavus at all tested concentrations. The extracts with MIC values above 2.5 mg/mL were Candida albicans (5 mg/mL) and C. neoformans (10 mg/mL).Control: Tetracycline and Amoxicillin | [ref. 189] |
| Rhus lancea L. F. | Leaves | Steam distillation | Disc diffusion assay | Acinobacter calcoaceticus, Citrobacter freundii, Clostridium perfringens, Clostridium sporogenes, Escherichia coli, Klebsiella pneumoniae, Proteus vulgaris, Pseudomonas aeruginosa, Salmonella typhi, Staphylococcus aureus, Yersinia enterocolitica, Candida albicans, Aspergillus flavus | Essential oil concentrations tested (10, 20, µg/mL) against K. pneumonia were reported to be resistant to the strain. Bacterial strains tested were susceptible to the different essential oil concentrations resulting in inhibition zones between 4.0 – 19.2 mm. Essential oil concentrations of 100 µg/mL had an inhibition zone of 74.2 mm, compared to the 76.2 mm inhibition zone of the control used. | [ref. 191] |
| Ricinus communis L. | Leaves | n‐Hexane, chloroform, DCM, ethyl acetate, acetone, ethanol, methanol | Microplate serial dilutionTLC‐bioautography | Staphylococcus aureus, Enterococcus faecalis, Escherichia coli, Pseudomonas aeruginosa | Hexane extracts showed good activity against the test strains (0.61 mg/mL). The hexane and ethanol extract had the best activity against P. aureginosa (0.13 mg/mL). The ethanol extracts showed activity (0.31 mg/mL) against the strains | [ref. 192] |
| Schkuhria pinnata (Lam.) Kuntze ex Thell. | Aerial parts | Hexane, DCM, acetone, ethyl acetate | Microplate serial dilution | Mycobacterium smegmatis | Extracts inhibited bacterial growth, with acetone extracts having good activity (0.27 mg/mL) compared with hexane extracts (2 mg/mL). | [ref. 120] |
| Searsia lancea (L.f.) F.A. Barkley | Leaves | Ethanol, acetone | Microplate serial dilution | Streptococcus agalactiae, Streptococcus uberis SUB 1 – 7, Streptococcus uberis ATCC 700407, Escherichia coli ECO 1 – 7, Escherichia coli ECO ATCC 25922 | Extracts were effective against the test strains with activity ranging between 0.06 and 0.2 mg/mL. | [ref. 93] |
| Securidaca longepedunculata Fresen. | Roots, bark | Ethanol | Disk diffusion assay | Candida albicans | The extract was active against Candida albicans. C. mycoderma and P. aeruginosa (Inhibition zones: 2.0 ± 0.3,3.0 ± 0.3 & 1.0 ±0.0 mm, respectively). Control: Fungazole and Ampicillin | [ref. 90] |
| Senna italica subsp. Arachoides Burch Lock | Roots | Acetone | Microplate serial dilution | Pseudomonas aeruginosa, Enterococcus faecalis, Escherichia coli, Staphylococcus aureus | The extract exhibited good activity against the test strains, with an average activity of 0.12 mg/mL. | [ref. 193] |
| Senna tora (L.) Roxb. | Leaves | Ethyl acetate | Agar well diffusion assay | Staphylococcus aureus, Enterococcus sp., Escherichia coli, Salmonella typhi, Pseudomonas aeruginosa, Haemophilus influenzae | Extracts were active against all strains. Extract concentrations of 62.5, 125 mg/mL, 250 mg/mL & 500 mg/mL, had smaller inhibition zones than the control.Control: Erythromycin | [ref. 135] |
| Tarchonanthus camphoratus L. | Leaves | Hexane, DCM, methanol | Disc diffusion assay | Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa, Proteus mirabilis, Salmonella typhi, Staphylococcus aureus, Bacillus spp., Candida albicans | Screening for activity revealed low inhibition (no or small inhibition zones) at 400 mg/mL concentration. Ethyl acetate extracts exhibited good activity (0.57 mg/mL) against Bacillus spp. and methanol extract gave the highest MIC activity against Candida albicans (2 mg/mL x 102).Control: Chloramphenicol and Nystatin | [ref. 194] |
| Terminalia sericea Burch. Ex DC | Leaves | Acetone, methanol | In vitroSerial dilution microplate assayIn vivoTopical application | Candida albicans, Cryptococcus neoformans, Microsporum canis, Shigella schenckii, Aspergillus fumigates | A 2 g/10 g dose (extract suspended in aqueous cream) administered to wounds inhibited infections after inoculation by test fungi. The in vivo studies showed good antifungal activity ranging from 0.02 ‐ 0.64 mg/mL.Control: Amphoterecin B | [ref. 94, ref. 97] |
| Terminalia sericea Burch. Ex DC | Leaves | Methanol | Staphylococcus aureus, Bacillus cereus, Staphylococcus epidermis, Enterococcus faecalis, Escherichia coli, Salmonella typhirium, Pseudomonas. aureginosa, Klebsiella pneumonia | Plant extracts were effective against all tested strains. The MIC values range from 0.6 to >3.0 mg/mL. The average MIC activity against the strains was >1.49 mg/mL. | [ref. 155] | |
| Vachellia karroo (Hayne) Banfi & Galasso | Leaves, roots | Chloroform, methanol, ethanol and ethyl acetate | Petri dish/ disc diffusion | Staphylococcus aureus, Escherichia coli, Klebsiella pneumonia, Salmonella typhi, Pseudomonas aeruginosa | Root extracts showed higher bacterial inhibition against all tested strains. | [ref. 195] |
| Vachellia nilotica (L.) P.J.H.Hurter & Mabb | Bark and leaves | Acetone, water | MIC Assay | Staphylococcus aureus, Streptococcus uberis, Streptococcus agalactiae, Streptococcus chromogenes, Streptococcus epidermidis, Escherichia coli, Klebsiella pneumonia, Pseudomonas aeruginosa, Proteus mirabilis, Enterobacter aerogenes | Acetone bark extract exhibited strong antibacterial activity against several Staphylococcus strains as well as P. aeruginosa, P. vulgaris and E. aerogenes strains.Control: Gentamycin | [ref. 134] |
| Ximenia americana L. | Bark, leaves, roots, stem | Water, methanol, chloroform | Cup‐plate agar diffusion, agar dilution method | Pseudomonas aeruginosa, Escherichia coli, Staphylococcus aureus, Candida albicans, | All methanol extracts had poor activity against the tested bacteria, MIC value range 0.21 ‐ > 72.75 mg/mL. S. aureus was most susceptible to methanol bark 0.62 mg/mL), water bark (1.62 mg/mL), methanol leaf (0.77 mg/mL) and water leaf extract (MIC = 1.99 mg/mL)Control: ampicillin, benzyl penicillin, cloxacillin, gentamicin (5, 10, 20 & 40 µg/mL).Methanol/water extracts (leaf, root, stem) were ineffective against Candida albicans. Controls: Clotrimazole (5 µg/mL) and Nystatin (25 µg/mL) | [ref. 158] |
| Ziziphus mucronata Willd. subsp. mucro nata | Leaves | Ethanol | Microplate serial dilution, Agar disc diffusion | Mycobacterium aurum, Bacillus cereus | Extracts were active against Mycobacterium aurum (0.625 mg/mL), and B. cereus (2 mg/mL).Control: Ampicillin | [ref. 90] |
| Ziziphus mucronata | Leaves | n‐Hexane, chloroform, DCM, ethyl acetate, acetone, ethanol, methanol | Microplate serial dilution, TLC‐bioautography | Staphylococcus aureus, Enterococcus faecalis, Escherichia coli, Pseudomonas aeruginosa | Hexane extracts showed good activity, the lowest average MIC value (0.50 mg/mL). hexane extracts had the best activity against E. coli (MIC = ). The highest activity (MIC = 1.05 mg/mL) for acetone and methanol extracts was against E. faecalis.Control: Not specified | [ref. 192] |
Abbreviations: Bw, body weight; DCM, dichloromethane; MIC, minimum inhibitory concentration; TCM, trichloromethane; TLC, thin layer chromatography
TABLE 5: Summary of analysis of anti‐parasitic activity of plants used by Batswana for livestock health management.
| Plant species | Plant part(s) | Extraction solvent | Bioassay and Application | Parasite species tested | Summary of findings | References |
|---|---|---|---|---|---|---|
| Acokanthera oppositifolia (Lam.) Codd | Leaves, twigs | Petroleum ether, DCM, ethanol, water | In vitro colourmetric assay | Caenorhabditis elegans | Petroleum ether and ethanol leaf extracts had high anthelmintic activity (MIC = 0.52 mg/mL) Control: 1 g/1 mL Levamisole | [ref. 122] |
| Aloe ferox Mill. | Leaves | Water | In vivo oral administration | Heterakis gallinarum | All the tested doses (50, 100, 200 and 400 mg/kg) reduced worm egg count (in vivo) after 14 days, with 100 and 400 mg/kg plant extracts exhibiting 99 and 100% egg count reduction. The 200 mg/kg dose resulted in the highest worm count reduction (± 85%) after slaughter.Control: Mebendazole | [ref. 108] |
| Aloe ferox Mill. | Leaves | Water | In vitro egg hatch assayLarval Development assayLarval mortality assay | Haemonchus contortus | All tested concentrations showed positive anthelmintic activity. The 7.5 mg/mL extract had the highest egg hatch inhibition (± 45%) and larval development inhibition percentage of ± 95%`. All tested concentrations (2.5, 5.0 & 7.5 mg/mL) exhibited high larval mortality concentration (99.1, 100 & 100 %) after 72 h.Control: 2.5, 5.0 & 7.5 mg/mL Thiambedazole | [ref. 196] |
| Aloe ferox Mill. | Leaves | Water (hot) | Egg hatch assayLarval development assay | Haemonchus contortus | Good egg hatch inhibition from concentration of 2.5 mg/mL (807%) onwards to 100% inhibition was achieved with 20 mg/mL extract (maximum concentration tested). The 1.25 mg/mL and 0.625 mg/mL extracts had the lowest inhibition (33.1% and 0.48%, respectively).Good larval development inhibition (98.9%) observed at the lowest tested concentration of 0.625 mg/mL.Control: Albendazole | [ref. 54] |
| Aloe marlothii A. Berger | Leaves | Acetone | Ehrlichia ruminantium | Plant extract with concentration of 50 mg/mL was effective against E. ruminantium | [ref. 197] | |
| Amaranthus blitum subsp. oleraceus (L.) Costea | Aerial parts | Ethanol | In vitro larvicidal assay | Trichinella spiralis | Extracts exhibited strong larvicidal activity at concentration of 1 mg/mL after 72 h, results show a 15% larval viability.Control: 20 µg/mL ABZ | [ref. 198] |
| Artemisia afra Jacq. ex Willd. | Leaves | Water (hot) | Adult worm inhibition | Schistosomula mansoni | Hexane and DCM extracts (100 µg/mL) inhibited 100% of the adult worms after 24, 48 and 72 h with a final IC50 value of 10.4 µg/mL (hexane) and 8.9 µg/mL (DCM) after 72 h.Control: praziquantel | [ref. 199] |
| Combretum hereroense Schinz | Leaves | Ethyl acetate, acetone, water | Microplate antischistosomal assay | Schistosoma haematobium,Caenorhabditis elegans var. Bristol | Acetone (80%–90% viability) and ethyl acetate (70%–80% viability) extracts were active against C. elegans at a concentration of 1 mg/mL. Water extracts were tested for antischistomosal activity, with no activity reported. | [ref. 200] |
| Dicerocaryum eriocarpum (Decne.) Abels(Syn: Sesamum eriocarpum (Decne.) Byng & Christenh.) | Leaves | Water | Egg hatch assayLarval development assay | Haemonchus contortus | The tested concentrations (2.5, 5, 7.5 mg/mL) were active against the tested parasite. The 7.5 mg/mL extract inhibited 100% egg hatching, 5 mg/mL and 7.5 mg/mL concentrations exhibited 100% larval development inhibition.Control: 2.5, 5.0 & 7.5 mg/mL Thiambedazole | [ref. 196] |
| Nicotiana tabacum L. | Leaves | Methanol | L. sericata development testCaenorhabditis elegans development testCtenocephalides felis adult oral testCtenocephalides felis adult contact test, Tick repellent/ knockdown contact test | Ctenocephalides felisLucilia seriataCaenorhabditis elegansRhipicephalus sanguineus (adult)Rhipicephalus sanguineus (larvae)Ixodes Ricinu | Minimum effective concentration for L. sericata larvae was >65 µg/mL, for C. felis concentration activity was >565 mg/mL, and for the percentage oral activity it was > 200 µg/mL.Controls: hiamethoxam, dicyclanil, ivermectin, and N, N‐diethyl‐m‐toluamide (DEET) | [ref. 110] |
| Sclerocarya birrea Hochst. | Bark | Methanol, water (cold/hot) | Larval mortality assay | Haemonchus, Oesophagostomum Trichostrongylus | Methanol extracts were effective against parasites. | [ref. 201] |
| Securidaca longepedunculata Fresen. | Roots | Water‐ethanol | In vitromortality assayIn vivoPostmortem worm countFaecal egg count | Haemonchus contortusHeligmosomoides polygyrus | In vitroThe extracts exhibited 75% mortality in the tested species (H. contortus), and 70% mortality of H. polygyrus.Control: LevamisoleIn vivoWorm count decreased by 80% after 500 mg/kg b.w. dose, worm count decreased by 88% after 1000 mg/kg b.w. (course duration: 4 days)Feacal egg count was increased after 4 consecutive days of treatmentControl: Pyrantel embonate | [ref. 202] |
| Vachellia nilotica (L.) P.J.H. Hurter & Mabb | Stem bark | Water | Trypanocidal activityLow Inoculation Long Incubation Test (LILIT)Long‐Term Viability Assay (LtVA) | Trypanosoma brucei brucei STIB 345 | Extracts had in vitro trypanocidal activity (MIC = 5 µg/mL)Stem extracts showed poor in vivo activity (MIC = 100 µg/mL) when 100–150 mg/kg body weight dose was administered orally.Control agent: diminazene aceturate (Berenil) | [ref. 203] |
| Ximenia americana L. var. microphylla Welw. ex Oliv. | Bark | Hydroethanol | Acaricidal adult immersion assay | Rhipicephalus microplus | Bark extract concentrations of 5 ‐ 80 mg/mL had acaricidal mortalities ranging from 13 ‐ 100%. | [ref. 204] |
| Thamnosma rhodesica (Baker f.) Mendonça | Roots | DCM | TLC‐BioautographyAntileishmanial assay | Leishmania major | Minimum amount for inhibiting fungal growth is 10 µg/mL. Control: Niastatin.Plant extracts inhibit reduce population of free‐living parasites (survival rate range from 30%–90%) and intracellular parasite (survival rate range 6.2% – 917 %).Control: Amphoterecin B | [ref. 205] |
Abbreviations: b.w, body weight; DCM, dichloromethane; MIC, minimum inhibitory concentration; TLC, thin layer chromatography.
TABLE 6: Summary of inflammation and pain study results of plants used by Batswana for livestock health management.
| Plant species | Plant part(s) | Extraction solvent | Biological assay | Summary of findings | References |
|---|---|---|---|---|---|
| Acokanthera oppositifolia (Lam.) Codd | Leaves, twigs | Petroleum ether, DCM, ethanol, water | COX 1 and 2 | Leaf petroleum ether, DCM and ethanolic extract showed good COX‐1 inhibition (98, 99, 97%). Leaf petroleum ether and DCM extracts exhibited good COX‐2 inhibition (85, 81%). | [ref. 122] |
| Aloe ferox Mill. | Leaves | Water | Extracts exhibited anti‐inflammatory and analgesic activities at a dose extract of 400 mg/kg. | [ref. 108] | |
| Bulbine abyssinica | Whole plant | Acetone, water | Protein denaturation method | Anti‐inflammatory activity of all extracts was observed 200 µg/mL dose. | [ref. 206] |
| Combretum hereroense Schinz | Leaves | Ethyl acetate, acetone, water | Plant extract exhibit in vitro inflammatory reaction at concentrations of 0.5 and 1 mg/ mL | [ref. 200] | |
| Malvastrum coromandelianum (L.) Garcke | Leaves | Methanol | 15‐LOX | Extracts and fractions were inhibitory to 15‐LOX, IC50 for the crude extract is 77.52 ±1.31 | [ref. 207] |
| Nicotiana tabacum L. | Seeds | Phytosterol isolates | COX 1 and 2 | Extract demonstrated inhibition of COX‐2 by down‐regulate the expression of COX‐2 mRNA and were ineffective against the expression COX‐1 mRNA. | [ref. 116] |
| Ozoroa paniculosa var. paniculosa | Leaves | Acetone | 15‐LOX | Maximum inhibition of >50% was observed when tested at 0.128 mg/mL extract concentration. | [ref. 208] |
| Portulaca oleracea L. | Aerial parts | Ethanol | Hind paw volume, reduction in cotton pellet weight, intraperitoneal administration | Time dependent positive anti‐inflammatory activity after 400 mg/kg dose, maximum hind paw volume increase of 67.1 ± 6.18 % after 240 mins. Percentage increase in cotton pellet weights in the control (86.69 ± 11.8%) after intraperitoneal administration.Control: diclofenac 4 mg/kg | [ref. 209] |
| Senna tora (L.) Roxb. | Leaves | Ethyl acetate | Heat induced haemolysis of RBC assay, BSA protein denaturation assay | Inhibition of RBC membrane hemolysis range of 31.058 ± 3.145% to 89.029 ± 1.186%, (IC50 = 28.309 µg/mL), tested dosage: 20 – 220 µg/mL. BSA denaturation inhibition range of 32.617 ± 0.890% to 91.731 ± 0.949% (IC50 = 22.980 µg/mL), tested dosage: 20–220 µg/mL.Control: ibuprofen 20–220 µg/mL. | [ref. 135] |
| Terminalia sericea Burch. Ex DC | Leaves | Acetone, methanol | In vivo, topical application | Wound healing activity observed is indicative of in vitro anti‐inflammatory activity (2 g/10 g dose) | [ref. 94, ref. 97] |
| Vachellia karroo (Hayne) Banfi & Galasso | Bark | Water | In vivo, administered via injection | Extracts had good analgesic and anti‐inflammatory activity in mice at doses of 100 and 200 mg/kg, comparable to the control usedControl: Indomethacin (10 mg/kg) | [ref. 123] |
| Vachellia tortilis (Forssk.) Galasso & Banfi | Seeds | Water | In vivo formalin induced paw lick test, acetic acid induces writhing test, hot plate test, tail flick method | Anti‐nociceptive activity (rats) when 100 and 200 mg/kg body weight administered orallyControl agents: Morphine sulphate + Naloxone, Morphine sulphate, Diclorofenac sodium | [ref. 117] |
Abbreviations: BSA, bovine serum albumin; COX, cyclooxygenase; DCM, dichlorormethane; LOX, 15‐lipoxygenae; NO, nitric acid; RBC, red blood cells.
TABLE 7: Summary of reported antioxidant activity of plants used by Batswana for livestock health management.
| Plant species | Plant part(s) | Extraction solvent | Biological assay | Summary of findings | References |
|---|---|---|---|---|---|
| Acokanthera oppositifolia (Lam.) Codd | Stem | Methanol | ABTS inhibition, DPPH scavenging | Extracts showed good ABTS inhibition (80 % inhibition) at concentration of 0.02 mg/mL. DPPH inhibition at 0.02 mg/mL was between 50%–60%. | [ref. 125] |
| Acokanthera oppositifolia (Lam.) Codd | Leaves | Acetone, ethyl acetate, chloroform, hexane, water | NO inhibition | In vitro analysis showed no NO‐inhibition at 10 mg/mL. | [ref. 126] |
| Acokanthera oppositifolia (Lam.) Codd | Leaves | Acetone, chloroform,, methanol | Hydroxyl free radical scavenging, Superoxide anion scavenging, Fe (III) reduction | Methanol extracts exhibited better Fe3+ reducing ability (IC50 = 0.234 mg/mL) than acetone (IC50 = 0.22 mg/mL) and chloroform (IC50 = 0.242 mg/mL). Superoxide anion scavenging activity was ± 78% for methanol extracts, ±66% for chloroform extracts and ±60% for acetone extracts. Acetone and chloroform extracts inhibit ±83% and methanol exhibit ±75% hydroxyl scavenging activity. | [ref. 17] |
| Aloe barbadensis Mill. | Leaves | Methanol | ORAC scavenging, HPS Scavenging, DPPH Scavenging | Extracts were found to have ORAC scavenging activity (TE ≈700 µmol/g), DPPH scavenging activity (TE ≈36 µmol/g), and HPS scavenging activity (TE ≈ 17 µmol/g). | [ref. 210] |
| Aloe ferox Mill. | Leaves | Methanol | DPPH scavenging, ABTS inhibition | Extracts reached 60% DPPH and 80% ABTS inhibition at 0.4 mg/mL. | [ref. 129] |
| Aloe ferox Mill. | Leaves | Methanol | ORAC scavenging, HPS Scavenging, DPPH Scavenging | Extracts exhibited ORAC scavenging activity (TE≈1000 µmol/g), DPPH scavenging activity (TE≈34 µmol/g), and HPS scavenging activity (TE≈37 µmol/g). | [ref. 210] |
| Aloe marlothii Mill. | Leaves | Methanol | ORAC scavenging, HPS Scavenging, DPPH Scavenging | Extracts showed ORAC, DPPH and HPS scavenging activity of ±1500 µmol/g, ± 55 mol/g, and ± 42 µmol/g, respectively. | [ref. 211] |
| Aloe marlothii A. Berger | Leaves | Methanol | DPPH scavenging | Extract concentration of 5 g/40 mL had DPPH scavenging activity of ±55 µmol/g. | [ref. 211] |
| Asparagus laricinus | Stem, leaves | Water | DPPH scavenging | Leaf and stem extract (2.5 mg/mL) exhibited 72% and 63% scavenging activity, respectively. | [ref. 135] |
| Cassia abbreviata Oliv. | Roots | Methanol | In vivo oral administration | Dosages of 200 and 500 mL/kg reduced oxidative stress in chickens and increased liver enzymes. | [ref. 137] |
| Diospyros lycioides | Leaves | Acetone, ethyl acetate, hexane, methanol | DPPH scavenging | Chromatograms elute in CEF had the highest number of compounds that with good DPPH scavenging activity; methanol extract had 4 bands, acetone extracts had 6 bands, ethyl acetate extract had 4 bands and hexane extracts had 1 band. The hexane extracts on the EMW eluted plate had no visible bands. All the extracts had 1 band on the BEA eluted plate, with similar Rf values (0.9 – 0.96). | [ref. 130] |
| Drimia sanguinea (Schinz.) Jessop | Bulb | Methanol | DPPH scavenging, B‐carotene linoleic acid | The extracts exhibited 64% DPPH scavenging activity (EC50 = 92.6 µg/mL) | [ref. 88] |
| Elephantorrhiza elephantina | Rhizomes | Methanol | DPPH scavenging | Extracts exhibited 84.7% antioxidant activity (EC50 = 5.8 µg/mL) | [ref. 88] |
| Euphorbia serpens Kunth | Shoots, roots | DCM, ethanol, methanol, petroleum ether | DPPH scavenging | All extracts exhibit antioxidant activity. Methanol shoot extracts had an IC50 value of 13.17 µg/mL, and DCM shoot extract had the highest IC50 value (553.26 µg/mL). | [ref. 130] |
| Grewia flava DC. | Twig, roots | Hexane, acetone, distilled water, methanol | DPPH scavenging, Ferric reducing, Metal Chelating | Methanol twig extracts were the most active at an IC50 of 14.50 µg/mL for DPPH scavenging activity, and distilled water twig extract had an IC50 value of 495 µg/mL. Metal chelation IC50 values were above 100 µg/mL, and ferric reducing power ranged from 637 ‐ 745 mg AAE/g. | [ref. 185] |
| Helichrysum paronychioides DC. | Whole plant | Methanol | DPPH scavenging | The extract exhibited 84.4% DPPH scavenging activity (EC50 = 20.1 µg/mL) | [ref. 88] |
| Malva neglecta Wallr. | Whole plant | Water‐methanol (30:70) | DPPH scavenging | The tested extract concentrations (0.0156 ‐ 1 mg/mL) exhibited antioxidant activity which increased depending on the concentration tested. The highest inhibition (70%) was observed at 1 mg/mL. | [ref. 212] |
| Ipomoea oblongata E. Mey. ex Choisy | Roots | Water, methanol, DCM | DPPH scavenging | The extract showed DDPH scavenging activity of 98% at 0.5 mg/mL tested extract concentration. | [ref. 213] |
| Moringa oleifera Lam. | Leaves | Methanol, fractions, diethyl ether, chloroform, ethyl acetate, water | In vitroDPPH scavenging, OH scavenging, NADH scavengingIn vivoOral ingestions, Rat liver dissection | In vitroExtract had 80% antioxidant activity; water residue had the lowest antioxidant activity of 22%. Crude extract showed strong reducing power (0.67 ASE/mL) and good DPPH scavenging activity (IC50 = 0.122 mg/mL).In vivoExtract had good ferrous ion chelating activity (0.45 mg/mL). | [ref. 132] |
| Ozoroa paniculosa var. paniculosa | Leaves | Acetone, fractions, hexane, DCM, ethanol, butanol, water | DPPH scavenging, ABTS Inhibition, OH‐ Scavenging | Extracts and ethanol fractions had the best DPPH scavenging activity (EC50 = 0.90 and 0.084 µg/mL), and water fractions had poor DPPH scavenging activity (EC50 = 663.47 µg/mL). Crude and ethanol extracts had good ABTS scavenging activity (EC50 = 0.99 and 1.60 µg/mL). | [ref. 190] |
| Portulaca oleracea L. | Whole plant | Water, ethanol | DPPH scavenging, ABTS inhibition | Antiradical activity for water extract showed an IC50 of 1.45 mg/g, while the 50% ethanol extract had an IC50 value of 0.36 mg/g. | [ref. 214] |
| Ricinus communis L. | Leaves | n‐hexane, chloroform, DCM, ethyl acetate, acetone, ethanol, methanol | DPPH scavenging | All extracts showed antioxidant activity on EMW chromatograms. | [ref. 192] |
| Senna tora (L.) Roxb. | Leaves | Ethyl acetate | DPPH scavenging, H2O2 scavenging | Extracts exhibited DPPH (33 ‐ 91%) and H2O2 (39 ‐ 99%) scavenging activity. | [ref. 139] |
| Tribulus terrestris L. | Fruit | Ethanol | NO inhibition | The extracts inhibited NO production at all test concentrations (50, 100, 200 µg/mL). | [ref. 153] |
| Vachellia karroo (Hayne) Banfi & Galasso | Leaves | Methanol‐water | ABTS+ scavenging, DPPH scavenging | The extracts demonstrated high DPPH scavenging activity (IC50 = 4.94 µg/mL) and ABTS+ (IC50 = 2.23 µg/mL) activity, with moderate FRAP scavenging activity (IC50 = 28 µg/mL), further supported by the TLC analysis and NMR. | [ref. 121] |
| Vachellia nilotica (L.) P.J.H. Hurter & Mabb | Leaves | Methanol‐water | DPPH scavenging | Extracts at 100 and 200 mg/mL exhibited DPPH scavenging activity. | [ref. 215] |
| Vachellia tortilis (Forssk.) Galasso & Banfi | Leaves, trunk bark | Methanol | DPPH scavenging | Leaf and trunk extract had good antioxidant activity (IC50 = 0.03 and 0.01 µg/mL). | [ref. 120] |
| Vachellia tortilis (Forssk.) Galasso & Banfi | Leaves | Methanol | DPPH scavenging | Low DPPH (IC50 = 70 µg/mL), ABTS + (IC50 = 52 µg/mL) and FRAP (IC50 = 97 µg/mL) scavenging activity was observed in extracts, further supported by the TLC analysis and NMR. | [ref. 121] |
| Ximenia americana L. var. microphylla Welwe. Ex Oliv. | Fruit, seeds | Ethanol | DPPH scavenging | Seeds showed significant DPPH radical scavenging activity at 200 µg/mL concentration. Both the red and yellow fruit flesh exhibited more than 90% of DPPH free radical scavenging activity at 200 µg/mL concentration. | [ref. 149] |
| Ximenia americana L. | Leaves | Methanol | DPPH scavenging | Extracts have a positive antioxidant activity, RC50 = 82.50 µg/mL | [ref. 216] |
| Ximenia americana L. | Pulp, seeds | Ethanol | DPPH scavenging | The red and yellow pulp extract had DPPH scavenging capacity percentage equal to/more than the ascorbic acid at the tested concentrations (160 and 200 µg/mL). The IC50 value for the yellow flesh was 102 µg/mL, and for the yellow seed it was 154 µg/mL. | [ref. 149] |
| Ziziphus mucronata | Leaves | n‐hexane, chloroform, DCM, ethyl acetate, acetone, ethanol, methanol | DPPH scavenging | Methanol extracts showed positive antioxidant activity on EMW chromatograms | [ref. 192] |
Abbreviations: ABTS+, 2,2’‐azino‐bis(3‐ethylbenzothiazoline‐6‐sulfonic acid); BEA = benzene: ethanol: ammonium hydroxide; CEF, chloroformethylacetate: formic acid; DCM, dichloromethane; DPPH, 2,2‐diphenyl‐1‐picrylhydrazyl; EMW, ethylacetate: methanol: water; FRAP, ferric reducing antioxidant power; H2O2, dihydrogen peroxide; HPS, NADH, nicotinamide adenine dinuclueotide hydrogen; NMR, nuclear magnetic resonance; NO, nitrous oxide; ORAC, oxygen radical absorbance capacity; TLC, thin layer chromatography.
TABLE 8: Summary of results from toxicity and safety studies of plants used by Batswana for livestock health management.
| Plant species | Plant part(s) | Extraction solvent | Summary of findings | References |
|---|---|---|---|---|
| Asparagus africanus Lam. | Roots | Water | In vivoNo acute oral toxicity was observed on all tested concentrations | [ref. 217] |
| Burkea africana Hook. | Stem bark | Ethanol | In vitroExtracts did not show oral toxicity at all tested concentrations.In vivoExtracts had a sedative effect at 2000 mg/kg. | [ref. 138] |
| Burkea africana Hook. | Stem bark | Ethanol | In vitroNo cytotoxicity observed at 2000 mg/kg. | [ref. 138] |
| Combretum imberbe (Wawra) | Leaves | Acetone | In vivoThe 10% extracts exhibited cytotoxicity and acute toxicity. | [ref. 94, ref. 97] |
| Cassia abbreviata Oliv. | Stem bark | Methanol | In vivoThe 200 and 500 mL/kg extract exhibited hepatotoxicity in indigenous chickens. | [ref. 137] |
| Combretum imberbe (Wawra) | Leaves | Acetone | In vitroExtracts showed some toxicity, with LC50 values ranging from 75.7–168.6 µg/mL ON Vero monkey cells | [ref. 94, ref. 97] |
| Cassine transvaalensis (Burtt Davy) CoddSyn: Elaeondedron transvaalense (Burtt Davy) R.H. Archer | Stem bark | Ethanol | In vitroIsolated compounds inhibited the growth of Vero cell lines. | [ref. 218] |
| Drimia sanguinea (Schinz.) Jessop | Bulb | Methanol, petroleum ether | In vitroMethanol extracts showed toxicity against Vero cells (LC50 = 0.015±0.01 µg/mL), and petroleum ether extracts had demonstrated low cytotoxicity (LC50 = 552.4± 48 µg/mL). | [ref. 88] |
| Elephantorrhiza elephantina (Burch) Skeels | Rhizome | Methanol, petroleum ether | In vitroMethanol extracts were toxic towards Vero cells (LC50 = 9.4±3.9 µg/mL) with petroleum ether extracts also showing some level of cytotoxicity with LC50 values of 173.4± 13 µg/mL. | [ref. 88] |
| Gomphocarpus fruticosus (L.) W.T. Aiton | Aerial parts | Methanol, DCM, chloroform, water | In vivoAcute oral toxicity of extracts had 0% mortality rate for 0.1 g/kg dose, 50% mortality rate for 0.2 g/kg dose, and the highest mortality rate was 0.3 and 0.35 g/kg doses with 83% mortality rate. | [ref. 219] |
| Helichrysum paronychioides DC. | Whole plant | Methanol, petroleum ether | In vitroMethanol and petroleum ether extracts exhibited low toxicity against Vero cells (LC50 = 24.6±0.4 and 50.2±1.8 µg/mL, respectively). | [ref. 88] |
| Malvastrum coromandelianum (L.) Garcke | Leaves | Methanol | In vitroIsolated compounds exhibited low cytotoxicity towards Vero cells. | [ref. 126] |
| Nicotiana tabacum L. | Seeds | Methanol, NaOH, HCl | In vivoIsolated compounds were found to be non‐toxic in mice. | [ref. 116] |
| Nicotiana tabacum L. | Leaves | Methanol, hexane | In vivoNo signs of sub‐acute toxicity were observed on mammalian models (rats). | [ref. 140] |
| Ozoroa paniculosa var. paniculosa | Leaves | Acetone | In vitroThere was some evidence of cytotoxicity (LC50 = 16.58 µg/mL) when extracts were studied against Vero cells. | [ref. 190] |
| Portulaca oleracea L. | Whole plants | Water, ethanol | In vitroHighest NCTC clone 929 cell viability (124.48%) after 72‐hour incubation with 100 µg/mL polysaccharide fraction, cell viability decreases sequentially with increase in tested concentration (250, 350, 500, 750, 1000 &1500 µg/mL) | [ref. 214] |
| Ricinus communis L. | Leaves | n‐Hexane, hexane, chloroform, DCM ethyl acetate, acetone, ethanol, methanol | In vitroNo observed toxicity on Vero kidney cells (LC50 = 131.8 µg/mL). | [ref. 192] |
| Securidaca longepedunculata Fresen. | Roots | Methanol | In vitroBrine shrimp assay revealed a dose dependent increase in mortality, LC50 = 74 µg/mL. | [ref. 202] |
| Senna tora (L.) Roxb. | Leaves | Ethyl acetate | In vitroModerately cytotoxic (LC50 = 35.246 µg/mL) was observed towards brine shrimp.In vivoLow acute oral toxicity (LD50 = 4263.906 mg/kg b. w.), body weight measurement does not indicate acute toxicity | [ref. 135] |
| Terminalia sericea Burch. Ex DC | Leaves | Acetone, methanol | In vitroExtracts exhibited low toxicity (LC50 = 75.7–168.6 µg/mL) when tested against Vero kidney cells. | [ref. 94, ref. 97] |
| Thesium spp. | Fruits | Ethanol | In vitroCell morphology was not affected, cell viability, cell viability of RAW 264.7 cells >98% after treatment with 50, 100 & 200 µg/mL of extract | [ref. 153] |
| Vachellia nilotica (L.) P.J.H.Hurter & Mabb | Bark, leaves | Acetone, water | In vitroWater and acetone extracts exhibit cytotoxicity (LC50 = 0.0032 and 0.0278 mg/mL) towards Vero kidney cells | [ref. 134] |
| Ziziphus mucronata | Leaves | Acetone, chloroform, DCM, ethanol, ethyl acetate, methanol, n‐hexane | In vitroNo observed toxicity towards Vero kidney cells with LC50 value of 131.8 µg/mL. | [ref. 192] |
Abbreviations: b.w., body weight; DCM, dichloromethane; HC, hydrochloric acid; MTT = 3‐ (4,5‐dimethylthiazol‐2‐yl)‐2,5‐diphenyl‐2H‐tetrazolium bromide; NaOH, sodium hydroxide; NCTC, National collection of type cultures
TABLE 9: Summary of bioactive phytochemicals and isolated compounds from plants used by Batswana for livestock health management.
| Species | Phytochemical analysis | Phytochemicals | Isolated compounds | References |
|---|---|---|---|---|
| Acokanthera oppositifolia (Lam.) Codd | Qualitative chemical assayQuantitative chemical assay (Spectrophotometry) | Flavanols, flavonoids, gallotannins, phenolics, polyphenols, proanthocyanidins, tannins | [ref. 17, ref. 122, ref. 126] | |
| Aloe ferox Mill. | Quantitative chemical assay (Spectrophotometry), HPLC, GC‐MS | Phenols, flavonoids, flavanols, proanthocyanidins, carotenoids | Lutein, beta‐carotein, capric acid, lauric acid, myristic acidpentadecyclic acid, palmitic acid, palmitoleic acid, cis‐7 hexadecenoic, margaric acid, stearic acid, oleic acid, vaccenic acid, linoleic acid,linolenic acid, arachidonic acid, behenic acid, tricosylic acid, lignoceric acid | [ref. 129, ref. 211] |
| Aloe marlothii A.Berger | GC‐MS | Carotenoids | Lutein, beta‐caroteinvitamin c, capric acidlauric acid, myristic acidpentadecyclic acid, palmitic acid, palmitoleic acid, cis‐7 hexadecenoic, margaric acid, stearic acid, oleic acid,vaccenic acid, linoleic acidlinolenic acid, arachidonic acid, behenic acid, tricosylic acid, lignoceric acid, fatty acids | [ref. 211] |
| Amaranthus blitum subsp. oleraceus (L.) Costea | GC‐MS | Phenol | Pyran based heterocycle,fatty acids, fatty ester, cyclododeca siloxane, alkyl siloxane, fatty acid, anyhydride | [ref. 220] |
| Ansellia africana Lindl. | Qualitative chemical assay | Saponin, alkaloids, tannins, terpenoids, steroids | [ref. 221] | |
| Artemisia afra Jacq. ex Willd. | HPLC | Luteolin, quercetin, chlorogenic acid, neochlorogenic acid, scopoletin | [ref. 199] | |
| Asparagus africanus Lam. | NMR, LC‐MS/Q‐TOF, Quantitative assay | Cardiac glycosides, proteins, flavonoids, tannins, saponins, steroids, terpenoids | Glucopyranoside, aspafricanol A, aspafricanol B, aspafricanol C, aspafricanol D, aspafricanol E, aspafricanene, aspafricanene A, acetylcaranine, 1,3,6,8‐naphthalenetetrol, stigmasterol,asparasaponin II, sarsasapogenin, prosopinine, glutinosonepandaroside C, cinncassiol C3 | [ref. 217, ref. 222, ref. 223] |
| Asparagus laricinus Burch. | Qualitative assay | Glycosides, steroids, flavonoids, saponins, tannins, phlobatannins, terpenoids, reducing sugars | [ref. 175] | |
| Bulbine abyssinica A. Rich. | Quantitative assay, qualitative assay | Tannins, phenolics, flavonoids, steroids, terpenoids, glycosides, saponins, alkaloids, proanthocyanins,condensed tannins | [ref. 176, ref. 206] | |
| Burkea africana Hook. | HPLC | Stigmasterol A, sitosterol B, catechin 4, glyceryl‐2‐stearate, glycerolmonostearate, epicatechin 5 | [ref. 138] | |
| Cadaba aphylla (Thunb) Wild | Qualitative assay, quantitative assay | Alkaloids, flavonoid, amino acids, proteins, anthraquinones, steroids, terpenoids, tannins, saponins, anthocyanins, coumarins, reducing sugars, oils and fats | [ref. 224] | |
| Cassia abbreviata Oliv. | Qualitative assay, GC‐MS, HPLC‐PDA—MS/MS, NMR | Alkaloids, saponinstannins, phenols, flavonoids, terpenoids, sterols, phenolsreducing sugars, total carbohydrates, cardiac glycosides, sterols, anthraquinone, coumarins, proanthosyanid, flavans | 2,3‐Dihydro‐5‐hydroxy‐8‐methoxy‐2‐ (4‐methoxyphenyl) chromen‐4‐one; 3,4‐Dihydro‐2‐(4‐hydroxyphenyl)‐ 4‐methoxy‐2H‐chromen‐7‐ol | [ref. 154, ref. 179, ref. 225, ref. 226] |
| Cassine transvaalensis (Burtt Davy) CoddSyn: Elaeondedron transvaalense (Burtt Davy) R.H. Archer | TLC chromatographySilica gel column chromatography | Triterpenoids | 3 –Oxo‐28‐hydroxylbetuli‐20(29)‐ene, 3,28‐dihydroxylbetuli‐20(29)‐ene | [ref. 86, ref. 218] |
| Centella asiatica (L.) Urb. | TLC, HPLC | Asiaticoside, Madecassoside, Asiatic acid | [ref. 227] | |
| Colophospermum mopane (J. Kirk ex Benth.) J. Léonard | Quantitative assay, NMR | Tannins, saponins, flavonoids, cardiac glycosides, sterols and steroids, alkaloids, coumarins,diterpenes | Dihydrogrindelic acid,Dihydrogrindelaldehyde, methyl labd‐13‐en‐15‐oate | [ref. 172] |
| Combretum hereroense Schinz | UPLC‐MS | Triterpene | [ref. 155] | |
| Combretum imberbe (Wawra) | TLC | Triterpene | [ref. 155] | |
| Diospyros lycioides Desf. | TLC, quantitative analysis, qualitative analysis | Flavonoids, phenolics, tannins, terpenoids, steroids | [ref. 130] | |
| Drimia sanguinea (Schinz.) Jessop | Quantitative assay, GC‐MS | Dotriacontane, benzothiazone,heptacosane, bumetrizole, phthalic acid (isomers), stigmasterol, hexanoic acid (derivatives), eicosanoic acid | [ref. 88] | |
| Elephantorrhiza elephantina (Burch) Skeels | Quantitative assay, GC‐MS, qualitative assay | Alkaloids, flavonoid, amino acids, proteins, anthraquinones,steroids, terpenoids, tannins, saponins, anthocyanins, coumarins, reducing sugars, oils and fats | Dotriacontane, benzothiazoneheptacosane, bumetrizole,phthalic acid (isomers), stigmasterol, hexanoic acid (derivatives), eicosanoic acid | [ref. 88, ref. 228, ref. 229] |
| Englerophytum magalismontanum (Sond.) T.D.Penn | Quantitative assay | Phenols | [ref. 228] | |
| Euclea undulata Thunb. | NMR | Naphthoquinones | Epicatechin, 7‐Methyl‐juglone, α‐amyrin‐3O‐β‐(5‐hydroxy), ferulic acid | [ref. 230] |
| Euphorbia regis‐jubae (Webb & Berth) | Affinity and gel exclusion chromatography, quantitative assay | Catechin | Catechin, perulic acid, caffeic acid, vanilla acid, Rutin, Syringic acid | [ref. 231] |
| Gomphocarpus fruticosus (L.) W.T.Aiton | GC‐MS, TLC, Qualitative assay | Flavanols, phenols, terpenoids | [ref. 112, ref. 184] | |
| Grewia flava DC. | GC‐MS | Alkaloids, flavonoids, saponins, steroids, glycosides, anthraquinones, tannins | Tetradecanoic acid, hexadecanoic acid methyl ester, n‐hexadecanoic acid, chlorpyrifos, phytol, methyl stearate, octadecanoic acideicosanoic acid methyl ester,disooctyl phthalate,tetratetracontane, tetracosanoic acid methyl ester, hentriacontane, octacosane, campesterol, stigmasterol, γ‐sitosterol, β‐amyrin, lupenone, lupeol, tarexerol | [ref. 185, ref. 232] |
| Harpagophytum procumbens (Burch.) DC. Ex Meisn. | HPLC‐DAD, GC‐MS, HPLC | Phenolics, polyphenols, tanninsflavonoids, flavanols | Rosmaric acid, harpagoside, arpagide | [ref. 233, ref. 234] |
| Helichrysum paronychioides DC. | Quantitative analysis, GC‐MS | Dotriacontane, benzothiazone, heptacosane, bumetrizole,phthalic acid (isomers), stigmasterol, hexanoic acid (derivatives), eicosanoic acid | [ref. 88] | |
| Hypoxis hemerocallidea Fisch., C.A. Mey. & Ave‐Lall. | Quantitative assay | Phenolics, tannins, gallotannins, flavonoids | [ref. 122] | |
| Ipomoea blongata E. Mey. ex Choisy | Qualitative assay, quantitative assay | Cardiac glycosides, steroids, triterpenoids, alkaloids, flavonoids, tannins | [ref. 129] | |
| Malva neglecta Wallr. | HPLC | Flavanoids, phenolic acids | Quercetin, kaempferol, gallic acid, chlorogenic acid, syringic acid, cinnamic acid | [ref. 212] |
| Malvastrum coromandelianum (L.) Garcke | NMR | Apigenin‐7‐О‐β‐6″ (p‐coumaroyl)‐glucopyranosideApigenin‐8‐C‐glucopyranoside (vitexin) | [ref. 126] | |
| Moringa oleifera Lam. | Qualitative assay, GC‐MS | Saponins, tannins, steroids, triterpenes, phlobatannins, flavonoids, glycosides, phytosterols | Brassicasterol, stigmasterol, campesterol, campesterol 2, 9,12‐octadecadienoic acid, gramisterol, Citrostadienol, beta‐sitosterol | [ref. 116, ref. 235] |
| Nicotiana tabacum L. | Quantitative assay, GC ‐MS | Alkaloids, anthraquinones,cardiac glycosides, flavonoid,saponins, steroids, tannins, terpenoids | (S)‐Nicotine, 2‐methyl‐4, ‐acetoxy‐tetrahydropyran5‐dihydrofuran, amitrole, citronellyl propionate, crotonaldehyde, isododecane, lavandulyl acetate, neophytadiene, pyridine,tetradecylaldehyde, trans‐phytol | [ref. 140, ref. 159] |
| Ozoroa paniculosa var. paniculosa Ozoroa paniculosa var. paniculosa (Sond.) R.Fern. & A.Fern | Qualitative methods | Phenolics, condensed tannins, proanthocyanidins, gallotannin, flavonoids, flavanols | [ref. 190] | |
| Peltophorum africanum Sond. | Qualitative and quantitative assays, TLC | Polyphenols, flavonoids, gallotannins, tannins | [ref. 203] | |
| Portulaca oleracea L. | Qualitative and quantitative assays | Flavonoids, organic acid, polyphenols | Acetic acid, butyric acid, caffeic acid, chlorogenic acid, cinnamic acid, citric acid, coumaric acid, ferulic acid, formic acid, gallic acid, genistein, isoquercitrin, kaempferol, lactic acid, luteolin, malic acid oxalic acid, propionic acid, quercetin, rutin, succinic acid, tartaric acid, umbeliferone | [ref. 214] |
| Rhoicissus tridentata subsp. Cuneifolia Rhoicissus tridentata (L.f.) Wind & R.B.Drumm. | Qualitative and quantitative assays | Anthraquinone, coumarins, flavonoids, glycosides, phenols,phlobatanins, phytosterolsresin, saponins, sterols and steroids, tannins, terpenoids, triterpenoids | [ref. 236] | |
| Rhus lancea L. f.Syn: Searsia lancea (L.f.) F.A.Barkley | Quantitative assay | Tannins | [ref. 93] | |
| Ricinus communis L. | GC‐MS, LC‐MS, NMR, Qualitative assay | Flavonoids, glycosides, phenolic compounds, tannins | 1‐O‐α‐D‐glucopyranosyl‐1,2‐eicosandiol, 2:3‐Glc‐campesterol, 5α,6β‐dihydroxysitosterol,6‐hydroxymethyllumazine8‐methylcaffeine, All‐trans‐retinyl linolate, Bufotenine O‐glucoside, germanicol cinnamate, medicagol, pomordol, N‐demethyl‐ricinine, phylloquinone, phytenic acid, pubescenol, pubesenolide, ricinine, ricinoleic acid, sodium oleate | [ref. 235, ref. 237] |
| Ricinus communis L. | Qualitative assay | Alkaloids, anthaquinescardiac glycosides, flavonoids, reduced sugars, steroids, tannins, terpenoids | [ref. 192] | |
| Schkuhria pinnata (Lam.) Kuntze ex Thell. | Qualitative and quantitave assay | Alkaloids, cardiac glycosides,flavonoids, phlabotannins,saponins, steroids, tannins | [ref. 238] | |
| Sclerocarya birrea Hochst. | Qualitative and quantitative assay | Alkaloids, flavonoids, glycosides, phenols, steroids, tannins | [ref. 201, ref. 220] | |
| Securidaca longepedunculata Fresen. | Qualitative analysis | Alkaloids, flavonoids, phenolic | [ref. 239] | |
| Senna italica Mill. | Quantitative analysis, GC‐MS | Alkaloids, anthocyanins, anthraquinones,catechins, flavonoids,leucoanthocyanins, tannins | (y)‐Sitosterol, (α)‐Tocopherol‐β‐D‐mannoside, 1,2‐enzenedicarboxylic acid, mono (2‐ ethylheptyl) ester, 12‐docosenamide, 1‐heptacosanol, lupeol, n‐tetracontane, oxirane, phytol, squalene, stigmasterol | [ref. 240, ref. 241] |
| Senna tora (L.) Roxb. | Quantitative assay, GC‐MS | Alkaloids, cardiac glycosides,flavonoids, reducing sugars,saponins, steroids, tannins, terpenoids | Carotene, cyclohexanedimethanol, hecamethyl cyclotrisiloxane, methyl stearate, neophytadiene, oxoalcohol, pentadecane, phenylethyl alcohol, pipecolic acid, tetradecane | [ref. 135, ref. 236] |
| Solanum incanum L. | Qualitative assay, C‐NMR, DEPT, H‐NMR, IR, UV | Phenolic acid, saponins, tannins | Steroid derivative SIE2 | [ref. 156, ref. 235] |
| Spirostachys africana Sond. | GC‐ToF‐MS | n‐Hexadecanoic acidnonadecane,2‐methyl | [ref. 235] | |
| Tarchonanthus camphoratus L. | Qualitative assays | Flavonoids, phenolic compounds, saponins, steroidstannins, terpenoid | [ref. 194] | |
| Terminalia sericea Burch. Ex DC | UPLC‐MS | Triterpene | [ref. 155] | |
| Thamnosma rhodesica (Baker f.) Mendonça | 3D NMR, C‐NMR, H‐NMR | Coumarins, furanocoumarins | 1‐hydroxy‐10‐methylacridone,byakangelicin, cnidili, gravacridonediol, imperatorinisopimpinellin, marmesin, rhodesiacridone, rutacridone, xanthotoxin | [ref. 205] |
| Tribulus terrestris L. | HPLC‐MS | Saponins | Astragaloside, hypericin, isomer of quercetin 3‐O‐arabinosyl galactoside, isoquercitrin, kaempferol 3‐ gentiobioside,microcephalin I, quercetin 3‐gentiobioside, quercetin‐3‐O‐(2,6‐α‐L‐dirhamnopyranosyl‐β‐D‐glucopyranoside), terrestrinin G, terrestrinin T, terrestrosin F, terrestrosin G, terrestrosin I, tribufuroside J, tribulosin, tribuluside A | [ref. 153] |
| Urginea sanguinea Schinz | C‐NMR, EL‐MS, FAB‐MS, H‐NMR | 3‐Hydroxy‐4‐methylbenzoic acid, 5a‐4,5‐dihydroscillaren A, n‐butanol fr, Phioroglucinol,Phloroglucinol, 1‐B‐D‐glucopyranoside (phiorin),Salicylic acid, Scillaren A, stigmasterol | [ref. 242] | |
| Vachellia karroo (Hayne) Banfi & Galasso | Quantitative Colometric phenolic assays | Phenolics | [ref. 243] | |
| Vachellia karroo (Hayne) Banfi & Galasso | GC‐MS | Phenolics | [ref. 195] | |
| Vachellia karroo (Hayne) Banfi & Galasso | TLC, H NMR, UHPLC‐qTOF‐MS | Baicalein, catechin, epicatechinepigallocatechin, kaempferol rutinoside, methyl gallate,myricetin rutinoside, quercetin, quercetin rutinoside, rutin | [ref. 121] | |
| Vachellia nilotica (L.) P.J.H.Hurter & Mabb | Anthraquinone, flavonoids, saponins, tannins | [ref. 244] | ||
| Vachellia tortilis (Forssk.) Galasso & Banfi | Qualitative analysis, H NMR, HP‐TLC, TLC, UHPLC‐qTOF‐MS | Alkaloid salts polyphenols, alkaloids, carotenoids, flavonoid, saponins, phenolic acids, phenols, saponin, sterols,tannins, triterpene, volatile oils | Baicalein, chrysoeriol glucopyranoside, cyanidin rhamnoside, dihydroacacipetalin, kaempferol, kaempferol rutinoside, luteolin glucoside, myricetin rutinoside | [ref. 120, ref. 121] |
| Withania somnifera (L.) Dunal | HPLC | Phenols | Withanolodine A, 12‐seoxywithastramonolide,Withanoside‐IV, Withaferin AWithanolide A, WithanonePhysalgulin‐D, Withastromonolide, 28‐hydroxywithanone | [ref. 119, ref. 245, ref. 246] |
| Ximenia americana L. var. microphylla Welwe. Ex Oliv. | Quantitative assay, FTIR, HP‐TLC, HP‐LC, GC‐MS | Alkaloids, fatty acids,flavonoids, glycosides, phenolics, phytosterols, saponins, tannins, terpenoids | 2,6,10,14‐Tetramethyloctadecane,chloracetamide‐n‐methanol,diethylhexylphthalate, elaidic acid methyl ester, nonadecane, oleic acid methyl ester, palmitic acid methyl ester, stearic acid methyl ester, ximeninic acid | [ref. 119, ref. 247] |
| Ziziphus oxyphylla EdgewSyn: Ziziphus acumi nata Royle | H‐ NMR, EI‐MS | Flavonoids, phenolic acids | Caffeic acid, catechin,ferulic acid, quercetin | [ref. 247] |
Abbreviations: CC, column chromatography; DEPT, distortionless enhancement by polarisation transfer; EI‐MS, electron ionisation‐mass spectrometry; FTIR, Fourier transform infrared; GC‐MS, gas chromatography‐mass spectrometry; HPLC, high performance liquid chromatography; IR, infrared; LC‐MS, liquid chromatography‐mass spectrometry; NMR, nuclear magnetic resonance; TLC, thin layer chromatography; UHPLC‐qTOF‐MA, ultra‐high performance liquid chromatography‐quadrupole time‐of‐flight mass spectrometer; UPLC, ultra performance liquid chromatography; UV, ultraviolet.
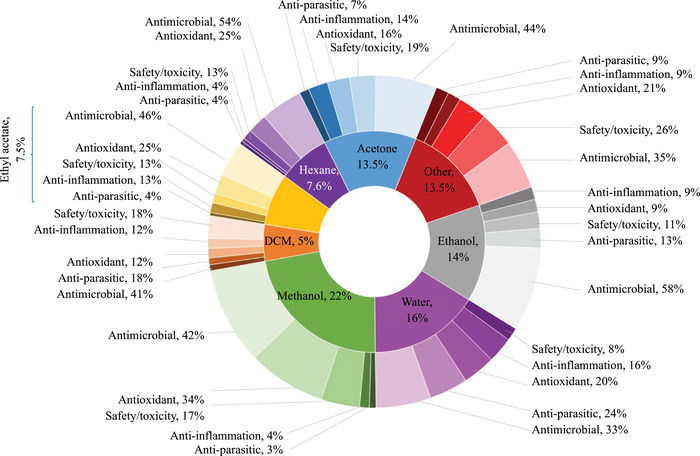
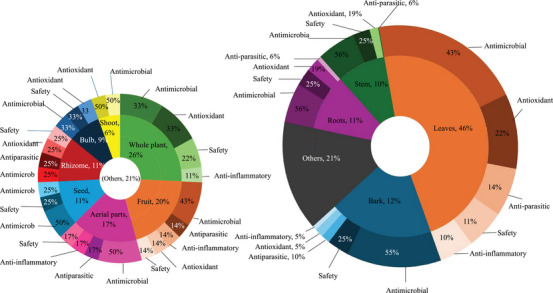
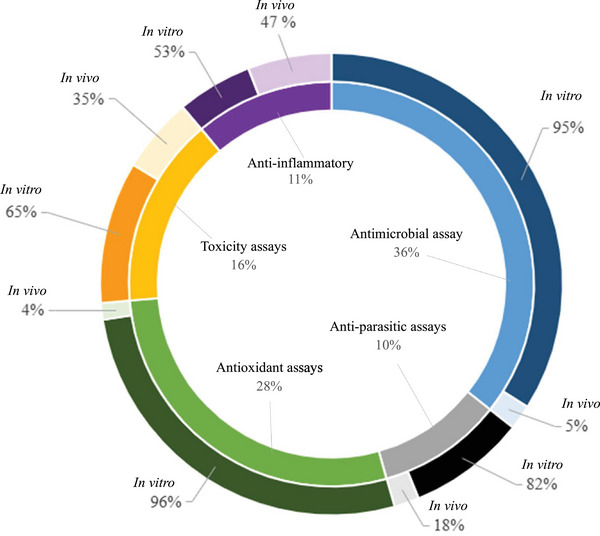
Extraction of bioactive compounds in different plant parts is highly dependent on the extraction solvents. The solvent is selected based on the polarity of targeted compounds, purpose of extraction, cost of solvent, safety of solvent, and potential energy demand for heating solvents [ref. 74, ref. 75]. The common use of water as an extractant can be attributed to its general use in traditional medicine to mimic ethnoveterinary practices involving maceration, decoction, poultice and infusions [ref. 76]. In the reviewed data (Figure 6), the use of water as an extractant was reported in 16% of the studies with methanol being the most prominent solvent used in majority of the studies (22%). Compounds with antimicrobial activity such as ketones, aldehydes and organic acids are extracted in high quantities using methanol as a solvent [ref. 77]. Ethanol (alcohol) is also widely used in traditional medicine as an alternative extractant to water in the reviewed literature and its use was calculated at 14%. Solvents reportedly used for plant extractions that had a low rate of use included petroleum ether (4%) and chloroform (4%) (Figure 4). The type of solvent used in plant extraction is also associated with the experimental analysis that is conducted, including biological activity, safety, phytochemical quantification, and bioactive compound isolation. Generally, biological activity (in vitro studies) in medicinal plants is investigated based on the ethnobotanical practices recorded by indigenous communities.
In the current review, the ethnoveterinary practices of medicinal plants used by the Batswana communities were assessed by examining the medicinal properties e.g., antimicrobial, antiparasitic, anti‐inflammatory, antioxidant, and toxicity of the plants. As a common trend in medicinal plant research, in vitro biological assays conducted accounted for 84% of the studies with in vivo assays contributing towards 16% of the research. Basically, plants that had noteworthy in vitro activity and have potential in drug discovery (interesting bioactive compounds) are further analysed in vivo, and such tests produce more accurate results than in vitro experiments. Unfavourable in vitro results may eliminate the need for further in vivo studies [ref. 78].
Antimicrobial Activity
Antimicrobial screening is the most common method applied for primary analysis of biological activity because it is quick, efficient and the resources required are usually readily affordable. Particularly, the serial dilution microplate assay is the most preferred method for evaluating the minimum inhibitory concentration (MIC) of plants against different test microbial strains, with more than 2 900 citations to date (accessed 08/02/2025) [ref. 79]. Following the recommended classification [ref. 80, ref. 81], extracts with MIC values > 0.32 mg/mL are regarded as exerting weak antimicrobial activity. However, higher MIC values may still be relevant for ethnopharmacological investigations [ref. 80]. The criteria for establishing activity of plant extracts are variable across difference sources, with noteworthy MIC values ranging from 8000 to ≤ 160 µg/mL [ref. 81]. MIC values < 0.1 mg/mL are also regarded as good by another author, while MIC values > 0.625 mg/mL was considered as weak [ref. 82, ref. 83]. For plant extracts used in traditional medicine, the MIC values of below 8 mg/mL are considered active [ref. 84]. Other authors considered a value as high as 12.5 mg/mL as active [ref. 85]. However, this review considers MIC as good/noteworthy when the activity is from <0.02 to 0.16 mg/mL [ref. 80]. Additionally, other in vitro assays used to determine antimicrobial activity included TLC bioautography, and agar disc diffusion [ref. 86, ref. 87, ref. 88]. Based on the results reported in Table 4, the serial dilution method was the most frequently applied technique followed by the agar disc diffusion assay. During the antimicrobial bioassays, microbial and fungal strains associated with animal gastrointestinal illnesses, skin infection, mastitis, and respiratory diseases amongst others were investigated in several studies, possibly due to the medicinal utilisation of the plants in ethnoveterinary medicine. The five most studied strains were Staphylococcus spp. (16.1%), Pseudomonas aeruginosa (11.9%), Escherichia coli (11.9%), Candida spp. (8.8%) and Klebsiella spp. (7.7%) (Figure 7). These pathogens are associated with most of the commonly reported ailments treated with medicinal plants. Aloe marlothii and Aloe zebrina extracts had excellent activity (MIC = 0.028 and 0.039 mg/mL) against E. coli [ref. 89, ref. 90]. This corroborates the perceived efficacy of the two Aloe species by the communities as they are reportedly used for diarrhoea and other infections [ref. 64, ref. 91]. Cassia abbreviata extracts had noteworthy activity against P.aeruginosa (MIC = 46.88 µg/mL), K. pneumoniae (MIC = 46.88 µg/mL), and good activity against C. albicans (MIC = 93.75 µg/mL) [ref. 92]. Dicerocaryum eriocarpum had good activity against Mycobacterium aurum (MIC = 0.156 mg/mL) but weak activity against C. albicans (MIC = 4.5 mg/mL) [ref. 90]. None of the conditions reported to be treated by D. eriocarpum are related to the tested pathogens [ref. 52, ref. 62, ref. 63, ref. 91], therefore the results do not support the traditional use. Searsia lancea had excellent activity (MIC range = 0.02–0.08 mg/mL) against the mastitis‐causing pathogens E. coli, Streptococcus agalactiae, Streptococcus dysgalactiae and Streptococcus uberis [ref. 93]. This may merit the use of S. lancea for treating diarrhoea resulting from one or more of the tested pathogens [ref. 91].
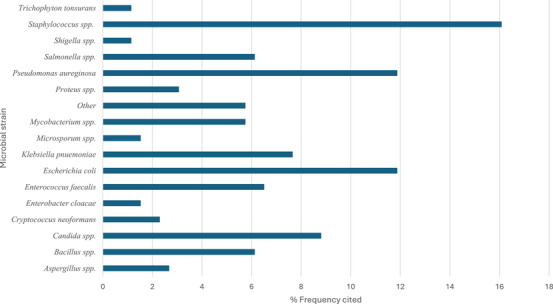
Diverse techniques are applied when observing the in vivo activity of medicinal plant extracts and these include visual examination of wound healing activity after topical application [ref. 94], faecal bacterial load [ref. 95], and determining secondary infection symptoms such as pulmonary burden after ingestion [ref. 96]. Study by Amoussa et al. [ref. 95] revealed that A. afra had notable in vitro activity (MIC range = 0.25–0.312 mg/mL) against non‐typhoidal Salmonella field isolates and ATCC strains and demonstrated the efficacy of the plant extracts against biofilm formation. From the same study, there is evidence of in vivo activity as the orally administered A. afra at 200 mg/kg/bw extracts completely eliminated the faecal bacterial load in mice after eight days [ref. 95]. Terminalia sericea was effective at reducing the induced wound associated with S. schenckii infection on mice, after excellent activity (MIC = 0.03 mg/mL) against S. schenckii was reported [ref. 97]. In vivo studies can also reveal additional information about the mode of action of the plant extract [ref. 96]. Additionally, these studies also confirm the potential of medicinal plant extracts to function as multitarget drugs [ref. 98]. A combination of in vivo and in vitro studies is important to further determine the efficacy of the plant extracts against the target strains. Artemisia afra is one plant that has been reported to have good antimicrobial activity in vitro against several strains of Salmonella spp. which is also supported by noteworthy in vivo activity [ref. 95]. However, an in vivo study revealed that the orally administered A. afra leaf water and DCM extracts had no antimycobacterial activity, despite the positive in vitro activity [ref. 96]. The phytochemicals associated with antimicrobial activities are terpenoids and phenolic compounds with hydroxyl groups which interrupt the bacterial cell membrane permeability [ref. 99]. The presence of such compounds in the aqueous bark extract of Ximenia americana [ref. 100] is consistent with the good antimicrobial activity reported against Bacillus subtilis (MIC = 0.20 mg/mL). It is presumed that the mode of action of most antimicrobial agents is by creating structural changes to the bacterial cell membrane which affects the integrity of the membrane [ref. 101]. Commonly, this results in lysis of the cells and leakage of the contents leading to cell death [ref. 98, ref. 101].
Antiparasitic Activity
Determining the antiparasitic activity of medicinal plants is of great significance in subtropical regions because the climatic conditions are breeding grounds for parasite infestations especially in animals [ref. 102]. In Batswana ethnoveterinary, examples of plants with notable in vitro antiparasitic activity includew Acokanthera oppositifolia, Aloe ferox, Aloe marlothii, Artemesia afra, Combretum hereroense, Dicerocaryum eriocarpum, Elephantorrhiza elephantina, Nicotiana tabacum, Sclerocarya birrea, Securidaca longepedunculata, Thamsosma rhodesica, and Ximenia americana (Table 5). In vitro assays such as egg hatch and parasite mortality assays are important in determining plant pesticidal activity [ref. 103]. However, in vitro studies usually target a single parasite, meanwhile multi‐parasitism is common in livestock [ref. 104]. On the other hand, in vivo assays typically involve topical application of plant extracts for external parasites, or oral ingestion for effective treatment of gastrointestinal parasites [ref. 105]. Parameters used to determine antiparasitic activity can reveal secondary effects, making them a suitable multitarget approach [ref. 106]. In vitro anthelminthic activity of Aloe ferox was reported [ref. 107] and in vivo studies supporting the activity of the extracts were conducted [ref. 54, ref. 108]. The authors observed that water extracts administered orally were able to reduce worm eggs and inhibit larval development [ref. 54, ref. 108]. As shown in Table 2, Nicotiana tabacum was cited twice in this report for use against parasites [ref. 63, ref. 109]. It has been tested against several parasites yielding positive results for in vitro and in vivo [ref. 110]. Ximenia americana had good activity (100% mortality) against Rhipicephalus microplus, however it has been reportedly used for internal parasites [ref. 91]. Fatty acids and fatty acid esters are believed to be responsible for nematocidal activity exhibited by plant extracts [ref. 111]. Phenolic compounds which are usually produced by plants to deter pests, also contribute to antiparasitic activity of plants [ref. 112]. Phytochemicals with proven nematocidal activity (e.g., tannins), have been isolated from plants used by the Batswana, including ganglion stimulants from Nicotiana tabacum leaves, which is similar to the active ingredient of levamisole (control agent) [ref. 113].
Anti‐Inflammatory Activity
Inflammation can be a result of physical injury, infection or other ailments [ref. 114, ref. 115]. In vitro activity is commonly determined by the inhibition of cyclooxygenase and lipoxygenase enzymes [ref. 116]. Anti‐inflammatory and other pain‐related assays had the highest rate (47%) of in vivo studies reported (Table 6). Physical evidence of pain and inflammation can be assessed visually in the tail‐flick assay and paw lick test [ref. 117], and by observing morphological changes during wound healing [ref. 118]. In vivo studies on Vachellia tortilis seed extracts exhibited noteworthy anti‐inflammatory response in mice with comparable activity (reaction time of 6.55 sec) in the group treated with diclofenac sodium (reaction time of 6.58 sec) and morphine sulfate (reaction time of 12.33 sec) [ref. 117]. The chemical profile of Vachellia tortilis revealed interesting phytochemicals such as myricetin rutinoside and luteolin glucoside which may be responsible for providing pain relief [ref. 119, ref. 120, ref. 121]. This supports the efficacy of V. tortilis for its traditional use of treating diarrhoea [ref. 91]. Decoction of Acokanthera oppositifolia leaves is used to expel parasites [ref. 53]. The extracts have excellent COX‐1 inhibition and COX‐2 inhibition activity, indicating good anti‐inflammatory activity with good in vitro nematocidal activity [ref. 122]. The anti‐inflammatory activity may be related to the presence of compounds which give the plant good antioxidant efficacy [ref. 123, ref. 124]. Vachellia karoo extracts had good anti‐inflammatory activity in mice which lasted up to 2 days after the treatment application. The 100 mg/kg dose produced 1.81 % inhibition, and the 200 mg/kg dose had 1.07% inhibition of inflammation, which is comparable to the inhibition of the positive control agent (1.38 %) used, indomethacin at 10 mg/kg [ref. 123]. The results support the use of V. karroo to treat fractures and skin diseases [ref. 64, ref. 76]
Antioxidant Activity
Plants with antioxidant activity are potential candidates for preventing certain inflammatory diseases [ref. 124]. Evidence of the antioxidant effect of the ethnoveterinary plants used by the Batswana have been assessed (Table 7). For instance, A. oppositifolia, which contains flavonoids, proanthocyanidins and quercetin equivalents did not exhibit in vitro NO‐ radical inhibition, but was active against ABTS free radicals, DPPH free radicals, hydroxyl free radical, superoxide anion scavenging activity and iron reducing activity [ref. 17, ref. 125, ref. 126].
In terms of the ethnoveterinary records, snake bites are one of the most common conditions treated by plants among the Batswana of southern Africa (Table 2). As the toxicity effect of snake venom is associated with inducing oxidative stress and inflammatory response [ref. 127, ref. 128], establishing the efficacy of medicinal plants used to treat snake bites becomes important. Some plants assessed for antioxidant activity are used to treat snake bite, with varying antioxidant activity reported. Aloe ferox extract exhibited notable DPPH (60%) and ABTS (80%) scavenging activity at the highest tested concentration of 0.4 mg/mL [ref. 129]. The DPPH scavenging activity of Diospyros lycioides was assessed using TLC and there were several bands in chromatograms eluted using three different systems indicating the presence of polar, non‐polar and neutral polarity with some antioxidant activity [ref. 130]. Moringa oleifera is reportedly used for cough and other unspecified conditions [ref. 63, ref. 131]. The extracts of M. oleifera had noteworthy in vitro DPPH, OH and NADH scavenging activity as well as in vivo ferrous ion chelating activity [ref. 132].
Toxicity and Safety of Medicinal Plants With Ethnoveterinary Records
Toxicity bioassays are necessary to determine the safety of herbal remedies in animals and humans [ref. 88]. The selectivity index (cytotoxicity) of plant extracts is important pecially in targeting specific cancer cell lines [ref. 133]. Cytotoxicity studies need to be conducted in parallel with the biological assays to confirm the efficacy for the plants or its general toxicity [ref. 134]. Some of the plants with ethnoveterinary records among the Batswana in southern Africa have been assessed for their safety with diverse responses (Table 8). As with other biological assays, in vitro cytotoxicity results will not always have the same results as acute toxicity tests. For instance, Senna tora leaves (ethyl acetate extracts) were reported to have moderate cytotoxicity (LC50 = 35.246 µg/mL) when using the brine shrimp assay, while no acute oral toxicity (LD50 = 4263.906 mg/kg b.w.) was observed in an in vivo assay using mammalian subjects [ref. 135].
Biomarkers such as glucose, urea, creatinine and alkaline phosphates are used to determine in vivo toxicity. These biomarkers usually indicate the degradation of vital organs. The impact on renal function, which is assessed by evaluating serum ureal and creatine concentrations in mice models is another indicator of the toxicity of Senna italica extracts [ref. 136]. In vivo assays account for 35% of the toxicity and safety assays reported (Figure 6). Some parameters used to determine acute oral toxicity are signs of hepatic damage [ref. 137], sedative or paralytic effect [ref. 138], loss of body weight [ref. 139], and mortality [ref. 94, ref. 113, ref. 140] reported on the subacute toxicity effect after topical application of plant extract. Mice treated with Nicotiana tabacum extract in a trial for activity against ticks (Rhipicephalus microplus) exhibited no signs of toxicity (i.e., skin reaction, intestinal distress, mortality) after 3 weeks [ref. 140]. The results indicate the efficacy and safety of N. tabacum for external parasites, leaving opportunity for investigating the efficacy and safety in terms of its traditional use in treating internal parasites [ref. 63, ref. 109, ref. 131]. The in vivo toxicity study revealing the safety of topically applied Terminalia sericea was part of a study investigating in vivo anti‐inflammatory activity [ref. 94]. This further supports the topical application of T. sericea (Table 2).
Profiling, Quantification and Identified Phytochemicals of Plants With Ethnoveterinary Records Among the Batswana of Southern Africa
Secondary metabolite production in plants is regulated by a range of factors, including biotic and abiotic stresses, physiological and developmental processes, chemical and mechanical elicitors, and cultivation and post‐harvest conditions. Despite the diversity of factors influencing their synthesis, plant secondary metabolism is predominantly directed towards the production of phenolics (≈45%), followed by terpenoids and steroids (≈27%), alkaloids (≈18%), and other compound classes (≈10%) [ref. 141]. From the current findings, a significant portion (51.72%) of the 116 plants with ethnoveterinary records have been profiled to establish their phytochemicals (Table 9). Phytochemical profiles varied among the identified species, with phenolics constituting the most abundant group, while terpenoids, steroids, and alkaloids were also present across the plant species. Generally, phytochemical analysis methods employed are chosen based on type of compounds sought, metabolites of interest, or the cost of analysis [ref. 142, ref. 143]. Chromatography methods are the most reported techniques used for phytochemical analysis (Figure 8). Earlier studies on phytochemical profiling primarily employed qualitative methods, such as colorimetric assays, which confirms the presence of phytochemical groups through precipitation, formation of foam or colour change in solution [ref. 144]. This method is usually applied at preliminary screening stages, as it provides a simple, quick and cost effective way of detecting phytochemicals without the need for sophisticated instrumentation [ref. 145].
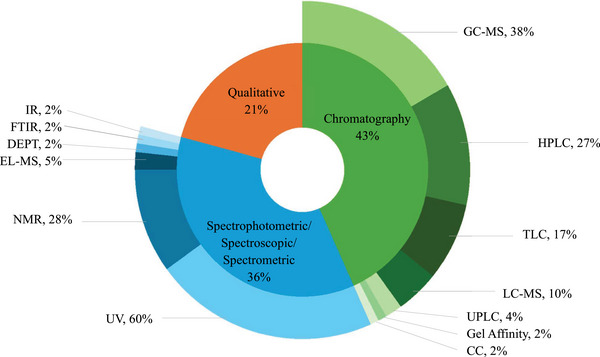
Gas chromatography is a relatively low cost method for identifying volatile organic compounds [ref. 146] and is the most commonly cited chromatography method due to volatility of phytochemicals such as terpenoids [ref. 147]. Ultraviolet light spectrophotometry is used to quantify phytochemicals that were detected using qualitative methods such as Sakowski test for steroids, Lieberman–Uchard’s test for triterpenes, Wanger’s test for alkaloid, ferric cyanide test for phenolic compounds and ferric chloride test for flavonoids [ref. 148].The phenolic compounds and flavonoids identified from Ximenia americana using the Folin–Ciocalteu and the Adom and Liu methods, respectively, have a wide range of biological activities [ref. 149]. Phenolic compound exhibit in vitro anthelmintic activity [ref. 150, ref. 151], which may contribute to the reported use to treat internal parasites (Table 2).
Chromatographic methods are used for separating, detection and quantification [ref. 152].Ultra‐performance liquid chromatography analysis of T. sericea extracts detected saponins which exhibited in vitro anti‐inflammatory activity on LPS treated RAW 264.7 cells [ref. 153]. The leaves, stem, roots and bark of T. sericea are used for several conditions including cough, internal parasites, diarrhoea and retained placenta (Table 2). The methanol extracts have in vitro anti‐microbial activity against Staphylococcus aureus, Bacillus cereus, Staphylococcus epidermis, Enterococcus faecalis, Escherichia coli, Salmonella typhirium, Pseudomonas aureginosa and Klebsiella pneumonia (average MIC ≥ 1.49 mg/mL), and the acetone and methanol extracts have in vivo wound healing activity observed, indicative of in vitro anti‐inflammatory activity (2 g/10 g dose) [ref. 97].
Spectrophotometric methods detect phytochemicals by detecting the functional groups present in a compound or the presence of conjugation within a compound [ref. 152]. The ─OH functional groups containing compounds identified from Cassia abbreviata, was identified as a flavan which was named 2,3‐dihydro‐5‐hydroxy‐8‐methoxy‐2‐(‐4‐methoxyphenyl)chromen‐4‐one [ref. 154]. The compound did not exhibit greater antiplasmodial activity (IC50 = 26.02 µg/mL) than the crude extract (IC50 = 13.31 µg/mL), or positive controls used, Chloroquine (IC50 = 0.026 µg/mL), mefloquine (IC50 = 0.03 µg/mL) and quinine (IC50 = 0.09 µg/mL) [ref. 154]. Despite the unspecified ethnoveterinary use of C. abbreviata (Table 2), flavans and other flavonoids exerted antioxidant, anti‐inflammatory and antimicrobial activities which contribute to treating several conditions [ref. 155, ref. 156].
The phytochemicals have a wide range of therapeutic properties, and the presence and concentration of specific phytochemicals can predict the bioactivity. The antioxidant activity of Ximenia afra leaf extracts can be attributed to the presence of procyanidins, and quercetin, compounds found to contribute to antioxidant activity [ref. 157]. The phytochemicals associated with antimicrobial activities are terpenoids and phenolic compounds with hydroxyl groups which interrupt the bacterial cell membrane permeability [ref. 99]. Therefore the presence of phenolic compounds and terpenoids (alkaloids, fatty acids, flavonoids, glycosides, phenolics, phytosterols, saponins, and tannins) in the aqueous bark extract of Ximenia americana [ref. 100] is consistent with the good antimicrobial activity reported [ref. 158].
The extensive chemical profiling for A. tortilis [ref. 121] revealed the presence of phytochemicals such as myricetin rutinoside and luteolin glucoside which may provide pain relief [ref. 119, ref. 120, ref. 121]. Fatty acid and fatty acid esters are believed to contribute to nematocidal activity of plant extracts [ref. 111]. Tannins and phenolic compounds are usually produced by plants for defense in response to biological attack [ref. 112, ref. 113]. This is evident in Nicotiana tabacum which has a high phenolic compound content [ref. 159] and has shown good antiparasitic activity. Qualitative analysis of Opuntia ficus‐indica extract, which has proven in vivo anthelmintic activity [ref. 160], has revealed the presence of polyphenols and phenolic compounds [ref. 161].
Clinical Trials of Plants With Ethnoveterinary Records
Clinical trials are required to determine long term toxicity, side effects and drug interactions and possible secondary benefits of phytotherapies [ref. 162, ref. 163, ref. 164]. A significant challenge in veterinary clinical trials is overcoming bias [ref. 165], which is compounded by the existing obstacles associated with clinical trials for phytotherapies. The complex nature of medicinal plants used in phytotherapies can cause inconsistencies and variations in phytochemical concentrations [ref. 166], therefore standardisation of protocols can be difficult [ref. 167]. The placebos used in phytotherapy clinical trials often pose an additional challenge as they fail to have the same colour, texture and smell, effectively not being an analogous compound. This brings to question the validity of the trial as a double‐blind study [ref. 168]. These challenges and more can limit the credibility of trial results, creating obstacles in regulating the industry [ref. 169]. Despite this, the clinical trial registry continues to be updated with new medicinal plant remedies in veterinary medicine (https://veterinaryclinicaltrials.org/studies/?term~recruiting_status = R, access 20/11/2024). A randomised, placebo‐controlled double‐blind clinical trial conducted on Cannabis sativa oil was reported to be beneficial in managing chronic pain by reducing inflammation and oxidative stress in canines [ref. 170]. There have been no reports to date of veterinary clinical trials for medicinal plants used by the Batswana people.
Conclusions
This review underscores the critical importance of traditional livestock husbandry practices among Batswana communities in southern Africa, where plant‐based remedies serve as essential tools for managing livestock health. The appraisal on ethnoveterinary knowledge uncovers the diversity of plants that the Batswana people use to treat a range of conditions such as parasitic infestations, wounds, infectious diseases and complications from animal bites. In the context of limited access to conventional veterinary care, these culturally rooted practices demonstrate the ingenuity and adaptability of indigenous knowledge systems in addressing diverse livestock health challenges. The continued reliance on these practices highlights their relevance not only as a means of sustaining livestock productivity but as a reflection of the deep connection between cultural heritage and indigenous medicinal plants. A comprehensive inventory of 116 plant species from 44 families was recorded, showcasing the remarkable biodiversity leveraged in treating nine major categories of livestock health conditions. Commonly used plant parts, such as roots and leaves, were prepared through methods such as decoctions and infusions, with oral and topical routes of administration being predominant. The presence of phytochemicals and the biological activities of the plants support the therapeutic potential of the plants. This provides empirical evidence to support ethnoveterinary knowledge. However, the presence of phytochemicals cannot be used as a sole determinant or factor for biological activity validation as other factors such as bioavailability can influence the efficacy and potency of plant extracts in vivo. Complementary in vivo studies and clinical trials can provide essential information on safety, pharmacological effect, and the correct dosage of ethnoveterinary medications, supporting the adoption of herbal remedies and their integration into modern veterinary health practices. Overall, the ethnoveterinary practices of the Batswana are of great benefit to underserved communities. The incorporation of plant‐based ethnoveterinary remedies into primary animal healthcare may be advantageous for rural communities given their dependence on livestock for survival.
Author Contributions
AOA and NAM conceptualised the study and edited the manuscript, TGM, NS and MVC sourced for the literature, conducted formal analysis and involved in the initial draft. JAA, SOA and LJM provided critical insight, edited the manuscript, and are involved in the supervision of the project. NAM, SOA and AOA secured the fundings for the project. All authors have read and agreed to the final version of the manuscript.
Funding
This work is based on the research supported by the South African National Department of Agriculture (DoA). A.O.A. received funding from the South African Research Chairs Initiative of the Department of Science, Technology and Innovation (DSTI)‐National Research Foundation (NRF) of South Africa (Grant No: RCHDI2411105279212). The financial support provided by the Higher Degree Committee of the Faculty of Natural and Agricultural Sciences (FNAS), North‐West University to TGM is sincerely appreciated. The opinions, findings, and conclusions or recommendations expressed are those of the authors alone; DoA and NRF accept no liability whatsoever in this regard.
Conflicts of Interest
The authors declare no conflict of interest.
Institutional Review Board Statement
This study was approved with reference number NWU‐01409‐23‐A9 by the Faculty of Natural and Agricultural Sciences Research Ethics Committee (FNASREC), North‐West University, South Africa.
Supplementary Materials
References
- Goodly Beasts, Beastly Goods: Cattle and Commodities in a South African Context,”. American Ethnologist, 1990
- 2 E. T. Khunoana and L. J. McGaw , “Ethnoveterinary Medicinal Plants Used in South Africa,” In Ethnoveterinary Medicine: Present and Future Concepts ed. L. McGaw and M. Abdalla (Springer, 2020), 211–250, 10.1007/978-3-030-32270-0_10.
- Swiss Ethnoveterinary Knowledge on Medicinal Plants–A Within‐Country Comparison of Italian Speaking Regions With North‐Western German Speaking Regions,”. Journal of Ethnobiology and Ethnomedicine, 2017. [PubMed]
- A Survey Analysis of Indigenous Goat Production in Communal Farming Systems of Botswana,”. Tropical Animal Health and Production, 2017. [PubMed]
- Foot‐and‐Mouth Disease and Market Access: Challenges for the Beef Industry in Southern Africa,”. Pastoralism, 2010. [DOI]
- The Genetic Legacy of the Expansion of Bantu‐Speaking Peoples in Africa,”. Nature, 2024. [DOI | PubMed]
- Genetic Substructure and Complex Demographic History of South African Bantu Speakers,”. Nature Communications, 2021. [DOI]
- ‘Where Goats Connect People’: Cultural Diffusion of Livestock not Food Production Amongst Southern African Hunter‐Gatherers During the Later Stone Age,”. Journal of Social Archaeology, 2017
- Male‐Biased Migration fpprom East Africa Introduced Pastoralism Into Southern Africa,”. BMC Biology, 2021. [DOI | PubMed]
- The Sociocultural Contexts and Meanings Associated With Livestock Keeping in Rural South Africa,”. African Journal of Range & Forage Science, 2013
- Batswana Ethnobotany: An Appraisal of the Uses of Botanicals for Diverse Purposes Among Local Communities in Botswana and South Africa,”. South African Journal of Botany, 2025
- Indigenous Village Chicken Production: A Tool for Poverty Alleviation, the Empowerment of Women, and Rural Development,”. Tropical Animal Health and Production, 2021
- Ethnoveterinary Use of Plants and Its Implication for Sustainable Livestock Management in Nepal,”. Frontiers in Veterinary Science, 2022. [DOI | PubMed]
- Exploration of Traditional Plant‐Based Medicines Used for Livestock Ailments in Northeastern Ethiopiaby,”. South African Journal of Botany, 2022
- 15 A. Zorloni , “Toward a Better Understanding of African Ethnoveterinary Medicine and Husbandry,” In Ethnoveterinary Medicine: Present and Future Concepts ed. L. McGaw and M. Abdalla (Springer, 2020), 151–172, 10.1007/978-3-030-32270-0_8.
- An Ethnoveterinary Survey of Medicinal Plants Used to Treat Poultry Diseases in Drylands of Zimbabwe,”. Ethnobotany Research and Applications, 2024
- Evaluation of Total Phenolic, Flavonoid Contents and Antioxidant Activity of Acokanthera oppositifolia and Leucaena leucocephala ,”. International Journal of Pharmacognosy and Phytochemical Research, 2015
- Ethnoveterinary Use of Southern African Plants and Scientific Evaluation of Their Medicinal Properties,”. Journal of Ethnopharmacology, 2008. [PubMed]
- Veterinary Drug Development From Indonesian Herbal Origin: Challenges and Opportunities,”. Indonesian Journal of Pharmaceutics, 2021
- Rates and Patterns of Habitat Loss Across South Africa’s Vegetation Biomes,”. South African Journal of Science, 2021. [DOI]
- Azithromycin Resistance in Escherichia coli and Salmonella from Food‐Producing Animals and Meat in Europe,”. Journal of Antimicrobial Chemotherapy, 2024. [PubMed]
- The Research of Antibacterial Properties of Decamethoxin, Decasan, Horosten,”. Journal of Education, Health and Sport, 2019
- Impact of Traditional Medicine Integration With Modern Healthcare in Africa,”. Newport International Journal of Scientific and Experimental Sciences, 2024
- A Review of the Traditional Use of Southern African Medicinal Plants for the Treatment of Inflammation and Inflammatory Pain,”. Journal of Ethnopharmacology, 2022. [PubMed]
- Ethnoveterinary Medicine in Africa,”. Journal of the International African Institute, 1992
- Preferred Reporting Items for Systematic Reviews and Meta‐Analyses: The PRISMA Statement,”. Annals of Internal Medicine, 2009. [PubMed]
- Ethnobotany at a Local Scale: Diversity of Knowledge of Medicinal Plants and Assessment of Plant Cultural Importance in the Polokwane Local Municipality, South Africa,”. Botany Letters, 2017
- 28 G. J. Martin , Ethnobotany: A Methods Manual (Routledge, 2010).
- Presence of Coliforms in Water, Poultry Mouth and Rectal Swabs From Selected Smallholder Poultry Farming Projects of Capricorn District, South Africa,”. Agricultural Science Digest, 2023
- A Review of PRA Methods for Livestock Research and Development,”. RRA Notes, 1994
- A Review of Veterinary Uses of Participatory Approaches and Methods Focussing on Experiences in Dryland Africa,”. Methods On The Move, 1999
- Participatory Epidemiology: Approaches, Methods, Experiences,”. The Veterinary Journal, 2012. [PubMed]
- The Origins and Practice of Participatory Rural Appraisal,”. World Development, 1994
- Qualitative and Mixed Methods Provide Unique Contributions to Outcomes Research,”. Circulation, 2009. [PubMed]
- Ethnoveterinary research and Development,”. Intermediate Technology, 1996
- 36 J. P. Dzoyem , R. T. Tchuenteu , K. Mbarawa , et al., “Ethnoveterinary Medicine and Medicinal Plants Used in the Treatment of Livestock Diseases in Cameroon,” In Ethnoveterinary Medicine: Present and Future Concepts ed. L. McGaw and M. Abdalla (Springer, 2020), 175–209, 10.1007/978-3-030-32270-0_8.
- An Ethnobotanical Study of Medicinal Plants Used to Treat Livestock Diseases in Onayena and Katima Mulilo, Namibia,”. South African Journal of Botany, 2014
- Wound Healing Plants in Mali, the Bamako Region. An Ethnobotanical Survey and Complement Fixation of Water Extracts From Selected Plants,”. Pharmaceutical Biology, 2002
- Ethnobotanical Study of Medicinal Plants Used by Local Communities of Minjar‐Shenkora District, North Shewa Zone of Amhara Region, Ethiopia,”. Journal of Medicinal Plants Studies, 2015
- A Review of Productive and Reproductive Characteristics of Indigenous Goats in Ethiopia,”. Livestock Research for Rural Development, 2015
- 41 P. Jambwa and E. T. Nyahangare , “Ethnoveterinary Medicine: A Zimbabwean Perspective,” In Ethnoveterinary Medicine: Present and Future Concepts ed. L. McGaw and M. Abdalla (Springer, 2020), 269–283, 10.1007/978-3-030-32270-0_12.
- A Review of Ethnoveterinary Medicines Used for Poultry Health Management in Zimbabwe,”. World’s Poultry Science Journal, 2023
- An Analysis of the Ethnoveterinary Medicinal Uses of the Genus Aloe L. for Animal Diseases in Africa,”. South African Journal of Botany, 2022
- Medicinal Plants of the Meinit Ethnic Group of Ethiopia: An Ethnobotanical Study,”. Journal of Ethnopharmacology, 2009. [PubMed]
- Medicinal Plants: An Invaluable, Dwindling Resource in Sub‐Saharan Africa,”. Journal of Ethnopharmacology, 2015. [PubMed]
- Medicinal Uses of the Fabaceae Family in Zimbabwe: A Review,”. Plants, 2023. [PubMed]
- 47 T. Sen and S. K. Samanta , “Medicinal Plants, Human Health and Biodiversity: A Broad Review,” In Biotechnological Applications of Biodiversity. Advances in Biochemical Engineering/Biotechnology ed. J. Mukherjee vol 147 (Springer, 2014): 59–110, 10.1007/10_2014_273.
- The Biological Approaches of Altering the Growth and Biochemical Properties of Medicinal Plants Under Salinity Stress,”. Applied Microbiology and Biotechnology, 2021. [PubMed]
- Ethnobotany, Phytochemistry, Pharmacology and Nutritional Potential of Medicinal Plants From Asteraceae family,”. The Journal of Materials Research and Technology, 2022
- Inventing a Herbal Tradition: The Complex Roots of the Current Popularity of Epilobium angustifolium in Eastern Europe,”. Journal of Ethnopharmacology, 2020. [PubMed]
- Ethnobotanical Knowledge of the Lay People of Blouberg Area (Pedi tribe), Limpopo Province, South Africa,”. Journal of Ethnobiology and Ethnomedicine, 2018. [PubMed]
- Ethno‐Veterinary Plants Used for the Treatment of Retained Placenta and Associated Diseases in Cattle among Dinokana Communities, North West Province, South Africa,”. South African Journal of Botany, 2020
- Participatory Inventory of Plant‐Based Ethnoveterinary Medicine Used to Control Internal Parasites of Goats in the Ngamiland Region of Botswana,”. South African Journal of Botany, 2024
- Ethnoveterinary Uses of Medicinal Plants: A Survey of Plants Used in the Ethnoveterinary Control of Gastro‐Intestinal Parasites of Goats in the Eastern Cape Province, South Africa,”. Pharmaceutical Biology, 2010. [PubMed]
- Snakebite Management in Cattle by Farmers in Lentsweletau Extension Area of Kweneng District in Botswana,”. International Journal of Innovative Research in Science, Engineering and Technology, 2015
- Ethno‐Veterinary Practices Amongst Livestock Farmers in Ngamiland District, Botswana,”. African Journal of Traditional, Complementary and Alternative Medicines, 2013
- 57 B. E. Wyk and N. Gericke , People’s Plants: A Guide to Useful Plants of Southern Africa (Briza Publications, 2000).
- An Ethnobotanical Study of Medicinal Plants Used in Kilte Awulaelo District, Tigray Region of Ethiopia,”. Journal of Ethnobiology and Ethnomedicine, 2013. [PubMed]
- Traditional Use and Safety of Herbal Medicines,”. Revista Brasileira de Farmacognosia, 2014
- Ethnoveterinary Knowledge and Biological Evaluation of Plants Used for Mitigating Cattle Diseases: A Critical Insight into the Trends and Patterns in South Africa,”. Frontiers in Veterinary Science, 2021. [DOI | PubMed]
- “A Study of Ethnoveterinary Medicine in the North West Province, South Africa.” Masters of Indigenous Knowledge Systems (MIKS) dissertation, North‐West University, South Africa. (. 2018
- Potential Use of Ethnoveterinary Medicine for Retained Placenta in Cattle in Mogonono, Botswana,”. Journal of Animal Production Advances, 2012
- The Use of Ethnoveterinary Medicine in Goats in Lentsweletau Village in Kweneng District of Botswana,”. Journal of Veterinary Advances, 2013
- Indigenous Knowledge and Use of Medicinal Plants for Ethnoveterinary Within the North West Province, South Africa,”. Frontiers in Veterinary Science, 2023. [DOI | PubMed]
- A Review of Ethnoveterinary Knowledge, Biological Activities and Secondary Metabolites of Medicinal Woody Plants Used for Managing Animal Health in South Africa,”. Veterinary Sciences, 2021. [PubMed]
- Livelihood Profiles and Adaptive Capacity to Manage Food Insecurity in Pastoral Communities in the Central Cattle Corridor of Uganda,”. Scientific African, 2022
- Commercially Important Medicinal Plants of South Africa: A Review,”. Journal of Chemistry, 2013
- Indigenous knowledge for Plant Species Diversity: A Case Study of Wild Plants’ Folk Names Used by the Mongolians in Ejina desert area, Inner Mongolia, P. R. China,”. Journal of Ethnobiology and Ethnomedicine, 2008. [DOI | PubMed]
- 69 Z. Leyew , Wild Plant Nomenclature and Traditional Botanical Knowledge among Three Ethnolinguistic Groups in North Western Ethiopia (African Books Collective, 2011).
- An Updated Inventory of Medicinal and Ritual Plants of Southern Africa,”. Journal of Ethnopharmacology, 2024. [PubMed]
- Ethnoveterinary Botanical Medicine in South Africa: A Review of Research from the Last Decade (2009 to 2019),”. Journal of Ethnopharmacology, 2020. [PubMed]
- Predictive Modeling for Sustainable High‐performance Concrete from Industrial Wastes: A Comparison and Optimization of Models Using Ensemble Learners,”. Journal of Cleaner Production, 2021
- Edible Seeds Medicinal Value, Therapeutic Applications and Functional Properties: A Review,”. International Journal of Pharmacy and Pharmaceutical Sciences, 2021
- A Standard Procedure for the Selection of Solvents for Natural Plant Extraction in the Early Stages of Process Development,”. Chemical Engineering & Technology, 2013
- Solvent Polarity Effects on Extraction Yield, Phenolic Content, and Antioxidant Properties of Malvaceae Family Seeds: A Comparative Study,”. New Zealand Journal of Botany, 2024
- Ethnoveterinary Practices and Ethnobotanical Knowledge on Plants Used Against Cattle Diseases Among Two Communities in South Africa,”. Plants, 2022. [PubMed]
- Bioactive Compounds, Antibacterial and Antioxidant Activities of Methanol Extract of Tamarindus Indica Linn,”. Scientific Reports, 2022. [PubMed]
- In Vitro Assays For Evaluating Phytate Degradation in Non‐Ruminants: Chances and Limitations,”. Journal of the Science of Food and Agriculture, 2021. [PubMed]
- Which Extractant Should be Used for the Screening and Isolation of Antimicrobial Components from Plants?,”. Journal of Ethnopharmacology, 1998. [PubMed]
- A Proposal towards a Rational Classification of the Antimicrobial Activity of Acetone Tree Leaf Extracts in a Search for New Antimicrobials,”. Planta Medica, 2021
- Antimicrobial Natural Product Research: A Review from a South African Perspective for the Years 2009–2016,”. Journal of Ethnopharmacology, 2017. [PubMed]
- Antibacterial Activity and Mode of Action of Acetone Crude Leaf Extracts of Under‐investigated Syzygium and Eugenia (Myrtaceae) s pecies on Multidrug Resistant Porcine Diarrhoeagenic Escherichia coli ,”. BMC Veterinary Research, 2019. [PubMed]
- Antibacterial and Antibiotic‐Modifying Activity of Methanol Extracts From Six Cameroonian Food Plants Against Multidrug‐Resistant Enteric Bacteria,”. BioMed Research International, 2017. [PubMed]
- A Comprehensive Review on Medicinal Plant Extracts as Antibacterial Agents: Factors, Mechanism Insights and Future prospects,”. Scientific African, 2024
- Phytochemical Content, Antibacterial, Antioxidant and Cytotoxic Activities, of Leaves Extracts of Eucalyptus globulus, Peltophorum africanum and Vangueria infausta ,”. Discover Agriculture, 2024
- Anti‐staphylococcal, Anti‐HIV and Cytotoxicity Studies of Four South African Medicinal Plants and Isolation of Bioactive Compounds From Cassine transvaalensis (Burtt. Davy) codd,”. BMC Complementary and Alternative Medicine, 2014. [PubMed]
- Awareness and Adoption of Green Computing in India,”. Sustainable Computing: Informatics and Systems, 2022
- Antimicrobial Activity, Antioxidant Potential, Cytotoxicity and Phytochemical Profiling of Four Plants Locally Used Against Skin Diseases,”. Plants, 2019. [PubMed]
- Extracts of Four Plant Species Used Traditionally to Treat Myiasis Influence Pupation Rate, Pupal Mass and Adult Blowfly Emergence of Lucilia cuprina and Chrysomya marginalis (Diptera: Calliphoridae),”. Journal of Ethnopharmacology, 2012
- Screening for Anti‐infective Properties of Selected Medicinal Plants From Botswana,”. African Journal of Plant Science and Biotechnology, 2011
- Use of Ethnoveterinary Medicinal Plants in Cattle by Setswana‐Speaking People in the Madikwe Area of the North West Province of South Africa,”. Journal of the South African Veterinary Association, 2001. [PubMed]
- Antimicrobial Activity and Potency of Cassia abbreviata Oliv Stem Bark Extracts,”. International Journal of Pharmaceutics, 2015
- In Vitro Antibiofilm and Quorum Sensing Inhibition Activities of Selected South African Plants With Efficacy Against Bovine Mastitis Pathogens,”. South African Journal of Botany, 2024
- The Use of a Rat Model to Evaluate the in vivo Toxicity and Wound Healing Activity of Selected Combretum and Terminalia (Combretaceae) species Extracts,”. Onderstepoort Journal of Veterinary Research, 2010
- Anti‐Salmonella Activity of Plant species in the Benin republic: Artemisia afra and Detarium senegalense with Promising in Vitro and in Vivo Activities,”. Biomedicine & Pharmacotherapy, 2023. [PubMed]
- Efficacy of Artemisia afra Phytotherapy in Experimental Tuberculosis,”. Tuberculosis, 2009. [PubMed]
- In Vivo Antifungal Effect of Combretum and Terminalia species Extracts on Cutaneous Wound Healing in Immunosuppressed Rats,”. Pharmaceutical Biology, 2010. [PubMed]
- Complex Interactions Between Phytochemicals. The Multi‐Target Therapeutic Concept of Phytotherapy,”. Current Drug Targets, 2011. [PubMed]
- Who Will be Smart Home Users? An Analysis of Adoption and Diffusion of Smart Homes,”. Technological Forecasting and Social Change, 2018
- Evaluation of Pharmacognostical and Phytochemical Studies for the Development of Quality Control Parameters for Stem Bark of Ximenia americana Linn. (Olacaceae),”. Ancient Science of Life, 2024
- Nutritional Status of Green Mussel Perna viridis at Tamil Nadu, Southwest Coast of India,”. Journal of Nutrition & Food Sciences, 2015. [DOI]
- Global Food Security: The Impact of Veterinary Parasites and Parasitologists,”. Veterinary Parasitology, 2013. [PubMed]
- Factors Affecting Utilisation of Indigenous Knowledge to Control Gastrointestinal Nematodes in Goats,”. Agriculture, 2021
- In vivo Anthelmintic Activity of Anogeissus leiocarpus Guill & Perr (Combretaceae) Against Nematodes in Naturally Infected Sheep,”. Parasitology Research, 2013. [PubMed]
- Efficacy of Strychnos spinosa (Lam.) and Solanum incanum L. Aqueous Fruit Extracts Against Cattle Ticks,”. Tropical Animal Health and Production, 2013. [PubMed]
- Photoreforming of Waste Polymers for Sustainable Hydrogen Fuel and Chemicals Feedstock: Waste to Energy,”. Chemical Reviews, 2023. [PubMed]
- Contamination of the Environment by Pathogenic Bacteria in a Livestock Farm in Limpopo Province, South Africa,”. Applied Ecology & Environmental Research, 2019
- Ethno‐Veterinary Control of Parasites, Management and Role of Village Chickens in Rural Households of Centane district in the Eastern Cape, South Africa,”. Tropical Animal Health and Production, 2009. [PubMed]
- Raising Livestock in Resource‐Poor Communities of the North West Province of South Africa‐A Participatory Rural Appraisal Study,”. Journal of the South African Veterinary Association, 2001
- Antiparasitic Properties of Leaf Extracts Derived From Selected Nicotiana Species and Nicotiana tabacum Varieties,”. Food and Chemical Toxicology, 2019. [PubMed]
- Phytochemical Evaluation, Antimicrobial Activity, and Determination of Bioactive Components From Leaves of Aegle marmelos ,”. BioMed Research International, 2014
- Metal–Organic Frameworks as Solid Catalysts for the Synthesis of Nitrogen‐Containing Heterocycles,”. Chemical Society Reviews, 2014. [PubMed]
- Over‐Expression of Tobacco UBC1 Encoding a Ubiquitin‐Conjugating Enzyme Increases Cadmium Tolerance by Activating the 20S/26S Proteasome and by Decreasing Cd Accumulation and Oxidative Stress in Tobacco (Nicotiana tabacum),”. Plant Molecular Biology, 2017. [PubMed]
- The Symptoms and Clinical Manifestations Observed in COVID‐19 Patients/Long COVID‐19 Symptoms That Parallel Toxoplasma gondii Infections,”. Journal of Neuroimmune Pharmacology, 2021. [PubMed]
- Chronic Inflammation and Cancer,”. Oncology, 2002
- Phytosterols Demonstrate Selective Inhibition of COX‐2: In‐Vivo and In‐Silico Studies of Nicotiana tabacum ,”. Bioorganic Chemistry, 2020. [PubMed]
- Curcumin Induces Apoptosis and Cell Cycle Arrest via the Activation of Reactive Oxygen Species–Independent Mitochondrial Apoptotic Pathway in Smad4 and p53 Mutated Colon Adenocarcinoma HT29 Cells,”. Nutrition Research, 2018. [PubMed]
- Evaluation of the Wound Healing Properties of South African Medicinal Plants Using Zebrafish and In Vitro Bioassays,”. Journal of Ethnopharmacology, 2022. [PubMed]
- Comparative Phytochemical Screening and Biological Evaluation of n‐Hexane and Water Extracts of Acacia tortilis ,”. Research In Pharmaceutical Biotechnology, 2012
- A Comparative Study of Phytochemical Profile and Antioxidant Activity of Sahelian Plants Used in the Treatment of Infectious Diseases in Northern Part of Burkina Faso: Acacia seyal Delile and Acacia tortilis (Forssk.) Hayne subsp. raddiana (Savi),”. Journal of Pharmacognosy and Phytotherapy, 2019
- Metabolomic Profiling of Antioxidant Compounds in Five Vachellia species,”. Molecules, 2021
- Processed Cranberry Bean (Phaseolus coccineus L.) Seed Flour for the African Diet,”. Canadian Journal of Plant Science, 2010
- Anti‐Inflammatory and Analgesic Activities of the Aqueous Extract of Acacia karroo Stem Bark in Experimental Animals,”. Basic & Clinical Pharmacology & Toxicology, 2008. [PubMed]
- Anti‐Inflammatory, Anticholinesterase and Antioxidant Activity of Leaf Extracts of Twelve Plants Used Traditionally to Alleviate Pain and Inflammation in South Africa,”. Journal of Ethnopharmacology, 2015. [PubMed]
- Antioxidant Properties of the Methanol Extracts of the Leaves And Stems of Celtis africana ,”. Records of Natural Products, 2009
- Green Synthesis of Silver Nanoparticle Using Oscillatoria sp. extract, its antibacterial, antibiofilm potential and cytotoxicity activity,”. Heliyon, 2019. [PubMed]
- Oxidative Stress and Antioxidant Defense in Detoxification Systems of Snake Venom‐induced Toxicity,”. Journal of Venomous Animals and Toxins Including Tropical Diseases, 2020
- Inflammation and Oxidative Stress in Snakebite Envenomation: A Brief Descriptive Review and Clinical Implications,”. Toxins, 2022. [PubMed]
- Antibacterial and Antioxidant Activities of some Selected Plants Used for the Treatment of Cattle Wounds in the Eastern Cape,”. African Journal of Biotechnology, 2012
- Antibacterial and Antimetastatic Potential of Diospyros lycioides Extract on Cervical Cancer Cells and Associated Pathogens,”. Evidence‐Based Complementary and Alternative Medicine, 2016
- Ethnoveterinary Medicine Usage in Family Chickens in the Selected Four Villages of Botswana,”. Journal of Veterinary Advances, 2012
- Review of Some Diseases of Dairy Animals and Treatment by Ethno‐Veterinary Medicines,”. Advancement in Medicinal Plant Research, 2023
- Antioxidant and Antibacterial Activities of Ethanol Extract and Flavonoids Isolated From Athrixia phylicoides ,”. Pretoria University of Pretoria (South Africa) (, 2010
- Selected Southern African Medicinal Plants with Low Cytotoxicity and Good Activity against Bovine Mastitis Pathogens,”. South African Journal of Botany, 2017
- Antioxidant, Analgesic, Anti‐Inflammatory, and Antipyretic Potentialities of Leaves of Artabotrys hexapetalus, Established by In Silico Analysis,”. Phytomedicine Plus, 2023
- Baba‐Moussa L: Phytochemical Screening, Antioxidant Activity, and Acute Toxicity Evaluation of Senna italica Extract Used in Traditional Medicine,”. Journal of Toxicology, 2023
- Effects of Cassia abbreviata Extract and Stocking Density on Growth Performance, Oxidative Stress and Liver Function of Indigenous Chickens,”. Tropical Animal Health and Production, 2019. [PubMed]
- Toxicological Evaluation of the Stem Bark of Burkea africana Hook.(Caesalpiniaceae) in Wistar Rats,”. The Nigerian Society of Pharmacognosy, 2019
- Editorial: Ethnoveterinary Practices in Livestock: Animal Production, Healthcare, and Livelihood Development,”. Frontiers in Veterinary Science, 2023. [DOI | PubMed]
- Cobalt Chloride Toxicity Elicited Hypertension and Cardiac Complication via Induction of Oxidative Stress and Upregulation of COX‐2/Bax Signaling Pathway,”. Human & Experimental Toxicology, 2019. [PubMed]
- Phytochemistry of Medicinal Plants,”. Journal of Pharmacognosy and Phytochemistry, 2013
- Mining Plant Metabolomes: Methods, Applications, and Perspectives,”. Plant Communications, 2021. [PubMed]
- 143 A. Bertoli , B. Ruffoni , L. Pistelli , and L. Pistelli , “Analytical Methods for the Extraction and Identification of Secondary Metabolite Production in ‘In Vitro’Plant Cell Cultures,” In Bio‐Farms for Nutraceuticals, Functional Food and Safety Control by Biosensors. Advances in Experimental Medicine and Biology ed. M. T. Giardi , G. Rea and B. Berra vol 698 (Springer, 2010), 250–266, 10.1007/978-1-4419-7347-4_19.
- Medicinal Plants Used in Jeddah, Saudi Arabia: Phytochemical Screening,”. Saudi Journal of Biological Sciences, 2021. [PubMed]
- Phytochemical Testing Methodologies and Principles for Preliminary Screening/Qualitative Testing,”. Asian Plant Research Journal, 2024
- Modern Developments in Gas Chromatography–mass Spectrometry‐Based Environmental Analysis,”. Journal of Chromatography, 2003. [PubMed]
- Comparative Genomics of Vancomycin‐Resistant Staphylococcus aureus Strains and Their Positions Within the Clade Most Commonly Associated With Methicillin‐Resistant S. aureus Hospital‐Acquired Infection in the United States,”. American Society for Microbiology, 2012. [DOI]
- Influence of Pinching and Plant Growth Regulators on Flowering, Yield and Economics of Fenugreek (Trigonella foenum‐graecum L.),”. Journal of Spices and Aromatic Crops, 2016
- Bioactive Composition, Free Radical Scavenging and Fatty Acid Profile of Ximenia americana Grown in Ethiopia,”. Heliyon, 2021. [PubMed]
- Anthelmintic Effect of Pterogyne nitens (Fabaceae) on Eggs and Larvae of Haemonchus contortus: Analyses of Structure‐Activity Relationships Based on Phenolic Compounds,”. Industrial Crops and Products, 2021
- Reducing Anthelmintic Inputs in Organic Farming: Are Small Ruminant Farmers Integrating Alternative Strategies to Control Gastrointestinal Nematodes?,”. Veterinary Parasitology, 2023. [PubMed]
- General Techniques Involved in Phytochemical Analysis,”. International Journal of Advanced Research in Chemical Science, 2015
- Anti‐Inflammatory Activity In Vitro, Extractive Process and HPLC‐MS Characterization of Total Saponins Extract From Tribulus terrestris L. fruits,”. Industrial Crops and Products, 2020
- Antiplasmodial Activity of Flavan Derivatives From Rootbark of Cassia abbreviata Oliv,”. Journal of Saudi Chemical Society, 2016
- Investigating Antimicrobial Compounds in South African Combretaceae species Using a Biochemometric Approach,”. Journal of Ethnopharmacology, 2021. [PubMed]
- Indigenous Uses, Phytochemical Analysis, and Anti‐Inflammatory Properties of Australian Tropical Medicinal Plants,”. Molecules, 2022. [PubMed]
- Eichhornia crassipes (Mart.) Solms: A Comprehensive Review of Its Chemical Composition, Traditional Use, and Value‐Added Products,”. Frontiers in Pharmacology, 2022. [PubMed]
- Antimicrobial Activity of Ximenia americana ,”. Fitoterapia, 2003. [PubMed]
- Phytochemical Analysis and Antimicrobial Activity of Bersama abyssinica fresen Against Multidrug‐Resistant Bacterial Uropathogens: Picolinyl Hydrazide Is a Major Compound,”. Journal of Herbs, Spices & Medicinal Plants, 2019
- Fucoxanthin as a Major Carotenoid in Isochrysis aff. Galbana: Characterization of Extraction for Commercial Application,”. Journal of the Korean Society for Applied Biological Chemistry, 2012
- Phytochemical Screening and Characterization of the Antioxidant, Anti‐Proliferative and Antibacterial Effects of Different Extracts of Opuntia ficus‐indica peel,”. Journal of King Saud University‐Science, 2022
- Clinical Effect of Autologous Platelet‐Rich Fibrin in the Treatment of Intra‐Bony Defects: A Controlled Clinical Trial,”. Journal of Clinical Periodontology, 2011. [PubMed]
- Codon Optimization Improves the Prediction of Xylose Metabolism from Gene Content in Budding Yeasts,”. Molecular Biology and Evolution, 2023
- Effects of Lower Limb Muscle Strengthening on Interface Pressure in Older Adults Undergoing Inelastic Compression: Randomized Controlled Clinical Trial,”. Phlebology, 2024
- Veterinary Clinical Trials Are on Trial,”. The Veterinary Record, 2017. [PubMed]
- An Insight into the Potential Mechanism of Bioactive Phytocompounds in the Wound Management,”. Pharmacognosy Reviews, 2023
- Challenges and Opportunities in the Advancement of Herbal Medicine: India’s Position and Role in a Global Context,”. Journal of Herbal Medicine, 2011
- Challenges and Guidelines for Clinical Trial of Herbal Drugs,”. Journal of Pharmacy and Bioallied Sciences, 2015. [PubMed]
- Challenges in Phytotherapy Research,”. Frontiers in Pharmacology, 2023. [DOI | PubMed]
- Biodeterioration of Stone Monuments: Studies on the Influence of Bioreceptivity on Cyanobacterial Biofilm Growth and on the Biocidal Efficacy of Essential Oils in Natural Hydrogel,”. Science of The Total Environment, 2023. [PubMed]
- Small‐Scale Poultry Production Systems in Serowe‐Palapye Sub‐District,”. Livestock Research for Rural Development, 1997
- Antipseudomonal Potential of Colophospermum mopane and Acrotome inflata, Medicinal Plants Indigenous to Namibia,”. African Journal of Pharmacy and Pharmacology, 2017
- Extracts of Ansellia africana and Platycarpha glomerata Exhibit Antibacterial Activities Against Some Respiratory Tract, Skin and Soft Tissue Infections Implicated Bacteria,”. South African Journal of Botany, 2018
- Phytochemical Analysis and Antibacterial Activity of the Kenyan Wild Orchids,”. Micro Environer, 2021
- Phytochemical Screening, Antibacterial and Antioxidant Activities of Asparagus laricinus Leaf and Stem Extracts,”. Bangladesh Journal of Pharmacology, 2014
- Comparative Phytochemical Analysis, Antioxidant and Antibacterial Activities in the Leaves, Underground Stems and Roots of Bulbine abyssinica ,”. Biomedical and Pharmacology Journal, 2022
- Antimicrobial Activity of Selected South African Medicinal Plants,”. BMC Complementary and Alternative, 2012
- Investigation on the Gastrointestinal Properties of Ethanolic Extract of Cannabis sativa Through In Vivo and In Vitro Approaches,”. Journal of Herbmed Pharmacology, 2023
- Phytochemical Analysis of Leaf, Stem Bark, and Root Extracts of Cassia abbreviata Grown in Zambia,”. Pharmacology & Pharmacy, 2022
- In Vitro Antibacterial Potential of Extracts of Sterculia africana, Acacia sieberiana, and Cassia abbreviata ssp. abbreviata Used by Yellow Baboons (Papio cynocephalus) for Possible Self‐Medication in Mikumi National Park, Tanzania,”. International Journal of Zoology, 2018
- Antibacterial Activity of Aqueous and Methanol Extracts of Selected species Used in Livestock Health Management,”. Pharmaceutical Biology, 2017. [PubMed]
- The Antifungal Activity of Twenty‐Four Southern African Combretum species (Combretaceae),”. South African Journal of Botany, 2007
- Chemical Composition, Antioxidant Potential, and Antimicrobial Activity of Nannorrhops ritchiana (Griff) Aitch,”. Archives of Advances in Biosciences, 2023. [DOI]
- Antibacterial Activity of Some African Medicinal Plants Used Traditionally Against Infectious Diseases,”. Pharmaceutical Biology, 2012. [PubMed]
- Grewia flava Twig Extracts: Phytochemical, Antioxidant, and Antimicrobial Evaluations,”. Bulletin of the National Research Centre, 2024
- Malva Neglecta: A Natural Inhibitor of Bacterial Growth and Biofilm Formation,”. Journal of Medicinal Plants Research, 2017
- Bioassay‐Guided Phytochemical Analyses and Antimicrobial Potentials of the Leaf Extract of Clematis hirsut a Perr. and Guill. Against Some Pathogenic Bacteria and Fungi,”. Infection and Drug Resistance, 2022. [PubMed]
- Assessment of Genetic Diversity in Crop Plants‐an Overview,”. Advances in Plants & Agriculture Research, 2017
- In Vitro Antibacterial Activity Of Plants Used as Herbal Tea in Tanzania,”. Journal of Complementary and Alternative Medical Research, 2016
- Cytotoxic, Antimicrobial, Antioxidant, Antilipoxygenase Activities and Phenolic Composition of Ozoroa and Searsia species (Anacardiaceae) Used in South African Traditional Medicine for Treating Diarrhoea,”. South African Journal of Botany, 2014
- Phytoconstituents and Biological Activities of Essential Oil from Rhus lancea L. F,”. African Journal of Biotechnology, 2008
- Antioxidant and antibacterial properties of Ziziphus mucronata and Ricinus communis leaves extracts,”. African Journal of Traditional, Complementary and Alternative Medicines, 2015
- Evaluation of the Antioxidant, Antibacterial, and Antiproliferative Activities of the Acetone Extract of the Roots of Senna italica (Fabaceae),”. African Journal of Traditional, Complementary and Alternative Medicines, 2010
- Antimicrobial Activity of Solvent Extracts from the Leaves of Tarchonanthus camphoratus (Asteraceae),”. Journal of Pharmacognosy and Phytochemistry, 2014
- Characterisation and Identification of Antibacterial Compound From Marine Actinobacteria: In Vitro and In Silico Analysis,”. Journal of Infection and Public Health, 2019. [PubMed]
- In Vitro Anthelmintic Activity of Seven Medicinal Plants Used to Control Livestock Internal Parasites in Chief Albert Luthuli Municipality, South Africa,”. Livestock Research for Rural Development, 2019
- Plastic Ingestion by Estuarine Mullet Mugil cephalus (Mugilidae) in an Urban Harbour, KwaZulu‐Natal, South Africa,”. African Journal of Marine Science, 2016
- Evaluating Anthelmintic, Anti‐Platelet, and Anti‐Coagulant Activities, and Identifying the Bioactive Phytochemicals of Amaranthus blitum L,”. BMC Complementary Medicine and Therapies, 2024. [PubMed]
- In Vitro Antischistosomal Activity of Artemisia annua and Artemisia afra Extracts,”. Phytomedicine Plus, 2022
- An Investigation on the Biological Activity of Combretum species,”. Journal of Ethnopharmacology, 2001. [PubMed]
- In Vitro Efficacy of Plant Extracts Against Gastrointestinal Nematodes in Goats,”. Tropical Animal Health and Production, 2021. [PubMed]
- Anthelmintic Activity of Securidaca longepedunculata (Family: Polygalaceae) Root Extract in Mice, In Vitro and In Vivo ,”. Asian Pacific Journal of Tropical Medicine, 2013. [PubMed]
- Evaluation of Medicinal Plants from Mali for Their In Vitro and In Vivo Trypanocidal Activity,”. Journal of Ethnopharmacology, 2006. [PubMed]
- Spatial Dynamics of the Girolando Breed in Brazil: Analysis of Genetic Integration and Environmental Factors,”. Tropical Animal Health and Production, 2020. [PubMed]
- Antileishmanial and Antifungal Acridone Derivatives from the Roots of Thamnosma rhodesica ,”. Phytochemistry, 2004. [PubMed]
- Preliminary Phytochemical Screening and Biological Activities of Bulbine abyssinica Used in the Folk Medicine in the Eastern Cape Province, South Africa,”. Evidence‐Based Complementary and Alternative Medicine, 2015
- Bioactive Constituents From Malvastrum coromandelianum (L.) Garcke Leaf Extracts,”. South African Journal of Botany, 2019
- In vivo effect of Selected Medicinal Plants Against Gastrointestinal Nematodes of Sheep,”. Tropical Animal Health and Production, 2014. [PubMed]
- Signaling by the TNF Receptor Superfamily and T Cell Homeostasis,”. Immunity, 2000. [PubMed]
- The Genus Rumex: Review of Traditional Uses, Phytochemistry and Pharmacology,”. Journal of Ethnopharmacology, 2015. [PubMed]
- Evolution of the Polyphenol and Terpene Content, Antioxidant Activity and Plant Morphology of Eight Different Fiber‐Type Cultivars of Cannabis sativa L. Cultivated at Three Sowing Densities,”. Plants, 2020
- Improvement of Plant Responses by Nanobiofertilizer: A Step Towards Sustainable Agriculture,”. Nanomaterials, 2022
- Ethno‐Medical Botany and Some Biological Activities of Ipomoea oblongata Collected in the Free State Province, South Africa,”. Journal of Biological Sciences, 2018
- Chemical Constituents and Bioactive Potential of Portulaca pilosa L vs. Portulaca oleracea L,”. Medicinal Chemistry Research, 2017
- Anticholinesterse and Antioxidant Investigations of Crude Extracts, Subsequent Fractions, Saponins and Flavonoids of Atriplex laciniata L.: Potential Effectiveness in Alzheimer’s and Other Neurological Disorders,”. Biological Research, 2015. [PubMed]
- Antioxidant Properties of Ximenia americana ,”. African Journal of Biotechnology, 2010
- Phytochemical Screening and Anti‐implantation Activity of Asparagus africanus Root Extract in Female Sprague–Dawley Rats,”. Revista Brasileira de Farmacognosia, 2019
- Cytotoxicity Activity of Isolated Compounds from Elaeodendron transvaalense Ethanol Extract,”. Journal of Medicinal Plants Research, 2010
- In Vitro and In Vivo Anticancer Activity of Gomphocarpus fruticosus Extracts and Development of LIPID‐Based Nanoparticles,” Namibia: University of Namibia (. 2019
- Green Synthesized Silver Nanoparticles: Optimization, Characterization, Antimicrobial Activity, and Cytotoxicity Study by Hemolysis Assay,”. Frontiers in Chemistry, 2022
- Phytochemical Analysis and Antibacterial Activity of the Kenyan Wild Orchids,”. Micro Environer, 2021
- Supercritical Water Gasification of Biomass: A State‐of‐the‐Art Review of Process Parameters, Reaction Mechanisms and Catalysis,”. Sustainable Energy & Fuels, 2019
- Chemical Constituents and Antioxidant Capacities of Asparagus africanus Lam.,”. Phytochemistry Letters, 2023
- Phytochemical Analysis in Roots and Tubers of Botswana Medicinal Plants‐Solanum aculeastrum Dunal, Elephantorrhiza elephentina, Cadaba aphylla (Thunb) and Adenia glauca ,”. Rasayan Journal of Chemistry, 2023
- A Proanthocyanidin‐Rich Extract From Cassia Abbreviata Exhibits Antioxidant and Hepatoprotective Activities In Vivo ,”. Journal of Ethnopharmacology, 2018. [PubMed]
- The Mechanism (s) of Action of Antioxidants: From Scavenging Reactive Oxygen/Nitrogen Species to Redox Signaling and the Generation of Bioactive Secondary Metabolites,”. Journal of Controlled Release, 2022
- Profiling of Centella asiatica (L.) Urban Extract,”. Malaysian Journal of Analytical Sciences, 2008
- Phytochemical Content, Antidiabetes, Anti‐inflammatory Antioxidant and Cytotoxic Activity of Leaf Extracts of Elephantorrhiza elephantina (Burch.) Skeels,”. South African Journal of Botany, 2020
- In Vitro Efficacy of Elephantorrhiza elephantina Root Extracts Against Adult Paramphistomum cervi in Goats,”. Parasite Epidemiology and Control, 2020. [PubMed]
- Variations in the Accumulation of Three Secondary Metabolites in Euclea undulata Thunb. Var. myrtina as a Function of Seasonal Changes,”. South African Journal of Botany, 2018
- Purification and Characterization of Aldose Reductase from Jerboa (Jaculus orientalis) and Evaluation of Its Inhibitory Activity by Euphorbia regis‐jubae (Webb & Berth) Extracts,”. Comparative Biochemistry and Physiology Part C: Toxicology & Pharmacology, 2021
- An Inventory of South African Medicinal Plants Used in the Management of Sexually Transmitted and Related Opportunistic Infections: An Appraisal and some Scientific Evidence (1990–2020),”. Plants, 2022. [PubMed]
- Antioxidant Activity and Phenolic Content of Betalain Extracts from Intact Plants and Hairy Root Cultures of the Red Beetroot Beta vulgaris Cv. Detroit Dark Red,”. Plant Foods for Human Nutrition, 2010. [PubMed]
- Antiarthritic Effects of a Root Extract from Harpagophytum procumbens DC: Novel Insights into the Molecular Mechanisms and Possible Bioactive Phytochemicals,”. Nutrients, 2020. [PubMed]
- Treatment of Brewery Industrial Wastewater and Generation of Sustainable Bioelectricity by Microbial Fuel Cell Inoculated with Locally Isolated Microorganisms,”. Journal of Water Process Engineering, 2021
- Phytochemical Profiles of Rhoicissus tridentata Harvested From the Slopes Elgon Sub‐region, Uganda,”. Research Square, 2023
- Identification of Antioxidant and Antimicrobial Compounds from the Oil Seed Crop Ricinus communis Using a Multiplatform Metabolite Profiling Approach,”. Industrial Crops and Products, 2018
- Phytochemical Investigation, Antioxidant and Antimycobacterial Activities of Schkuhria pinnata (Lam) Thell Extracts Against Mycobacterium smegmatis ,”. Journal of Evidence‐Based Integrative Medicine, 2019
- Comparative Study of Leaves and Root Bark From Securidaca longepedunculata Fresen (Polygalaceae): Phytochemistry and Antiplasmodial Activity,”. The Pharma Innovation Journal, 2018
- Sohounhloue DCK: Phytochemical Profile and Potential Pharmacological Properties of Leaves Extract of Senna italica Mill,”. American Journal of Pharmacological Sciences, 2021
- Effect of Seasonal Changes on the Quantity of Phytochemicals in the Leaves of Three Medicinal Plants From Limpopo Province, South Africa,”. Journal of Pharmacognosy and Phytotherapy, 2016
- Bufadienolides and Other Constituents of Urginea sanguinea ,”. Planta Medica, 1997. [PubMed]
- Proanthocyanidins and Other Phenolics in Acacia Leaves of Southern Africa,”. Animal Feed Science and Technology, 2001
- Preliminary Phytochemical and Elemental Analysis of Aqueous and Fractionated Pod Extracts of Acacia nilotica (Thorn mimosa),”. Veterinary Research Forum, 2014. [PubMed]
- Effect of Extraction Methods on Yield, Phytochemical Constituents and Antioxidant Activity of Withania somnifera ,”. Arabian Journal of Chemistry, 2017
- Specificity Protein 1‐dependent p53‐mediated Suppression of Human Manganese Superoxide Dismutase Gene Expression,”. Journal of Biological Chemistry, 2006. [PubMed]
- Isolation of Secondary Metabolites from Ziziphus oxyphylla Fruit,”. International Journal of Chemical and Biochemical Sciences, 2018
