Integrating endocannabinoid signaling, CCK interneurons, and hippocampal circuit dynamics in behaving animals
Abstract
The brain’s endocannabinoid signaling system modulates a diverse range of physiological phenomena and is also involved in various psychiatric and neurological disorders. The basic components of the molecular machinery underlying endocannabinoid-mediated synaptic signaling have been known for decades. However, limitations associated with the short-lived nature of endocannabinoid lipid signals had made it challenging to determine the spatiotemporal specificity and dynamics of endocannabinoid signaling in vivo. Here, we discuss how novel technologies have recently enabled unprecedented insights into endocannabinoid signaling taking place at specific synapses in behaving animals. In this review, we primarily focus on cannabinoid-sensitive inhibition in the hippocampus in relation to place cell properties to illustrate the potential of these novel methodologies. In addition, we highlight implications of these approaches and insights for the unraveling of cannabinoid regulation of synapses in vivo in other brain circuits in both health and disease.
Affiliations: Department of Neurosurgery, Stanford University, Stanford, CA, USA; These authors contributed equally; Department of Neurology, Harvard Medical School, Boston, MA, USA; Rosamund Stone Zander Translational Neuroscience Center, Boston Children’s Hospital, Boston, MA, USA; F.M. Kirby Neurobiology Center, Harvard Medical School, Boston, MA, USA; Department of Neurology, Baylor College of Medicine, Houston, TX, USA; Department of Neuroscience, Columbia University, New York, NY, USA; Kavli Institute for Brain Sciences, Columbia University, New York, NY, USA; Mortimer B. Zuckerman Mind Brain Behavior Institute, Columbia University, New York, NY, USA
License: CC BY 4.0 This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Article links: DOI: 10.1016/j.neuron.2025.03.016 | PubMed: 40267911 | PMC: PMC12410895
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (1.9 MB)
INTRODUCTION
The brain by dry weight is composed mostly of lipids, and endocannabinoids (eCBs) form a major class of lipid-derived information-carrying molecules. eCBs have important roles in a wide range of neuronal phenomena, including appetite regulation, temperature regulation, pain perception, brain development, learning and memory, and motor functions. In addition, the eCB system is implicated in a large number of psychiatric and neurological disorders, including epilepsy, pain, autism spectrum disorders, addiction, anxiety, psychosis, Alzheimer’s disease, Huntington’s disease, and Parkinson’s disease.1–5 The major neuronal receptor for eCBs is the cannabinoid type-1 (CB1) receptor. Reflecting the widespread involvement of the eCB signaling system in a diversity of cognitive, sensory-motor, affective, and homeostatic brain functions and dysfunctions, the CB1 receptor is one of the most abundant G-protein-coupled receptors (GPCRs) in the brain.6
The basic elements and key mechanistic features of the eCB system have been established for over two decades, mostly based on biochemical, immunocytochemical, in vitro electrophysiological, and behavioral studies.1,6,7 It has also been well recognized that while the expression of CB1 receptors is widespread across numerous cortical and subcortical areas, CB1 receptors at the microscopic scale are selectively localized on the presynaptic axon terminals of specific subpopulations of GABAergic and glutamatergic cells.8–10 The two major eCB ligands in the brain are the neuromodulatory lipids 2-arachidonoylglycerol (2-AG) and N-arachidonoyl-ethanolamide (anandamide, AEA).11–13 2-AG is a full CB1 receptor agonist that is present at substantially higher concentrations (nmol g−1) than the partial agonist AEA (pmol g−1). Importantly, both eCBs are known to be highly labile molecules, due to the rapid action of specialized metabolic enzymes (see Box 1 for an introductory overview of the eCB signaling system).
In terms of synaptic actions of eCBs, in vitro electrophysiology studies carried out in culture systems and acute brain slices took advantage of the so-called depolarization-induced suppression of inhibition (DSI) and excitation (DSE) phenomena to reveal the cardinal properties of the eCB synaptic signaling system (Box 1).23–25 Namely, these research efforts showed that eCB signaling is a powerful, postsynaptic neuronal activity-dependent, retrograde process that results in a short-term depression of γ-aminobutyric acid (GABA) (in the case of DSI) or glutamate (DSE) release from nearby presynaptic axon terminals that express CB1 receptors. In DSI, a strong depolarization of the postsynaptic principal cell (typically to 0 mV for a second, delivered through the recording pipette by the experimenter) induces a transient (lasting several seconds) suppression of the incoming inhibitory synaptic events. Mechanistically, the strong depolarizing pulse activates voltage-gated calcium channels, leading to a prominent rise in intracellular calcium, which, in turn, activates postsynaptic membrane-bound enzymes that synthesize eCBs from their phospholipid precursors. The newly generated eCBs are then thought to be released from the activated postsynaptic neuron through a still-debated mechanism34,35 to retrogradely reach and activate the presynaptic CB1 receptors to decrease GABA release through the inhibition of calcium channels. Such in vitro studies also revealed that the basic features of DSE (e.g., its retrograde nature and CB1 antagonist sensitivity) are similar to those of DSI, although DSE in cortical circuits typically requires a longer postsynaptic depolarization (7–10 s) to induce it, at least with somatic single-cell recordings. Regardless of whether one considers DSI- or DSE-sensitive axon terminals, a key feature of the eCB signaling system is that CB1 receptors are heterogeneously expressed across cell types. For example, immunocytochemical studies, paired patch clamp recordings in acute slices, and pharmacological manipulations revealed that among GABAergic interneurons (INs), presynaptic CB1 receptor expression is highest in INs that co-express cholecystokinin (CCK). In particular, GABA release from perisomatically targeting basket cells (BCs) that express CB1 and CCK (CCKBCs) has been found to be exquisitely sensitive to eCBs in various brain regions, including the hippocampus, amygdala, and neocortex.8,36,37 Interestingly, eCBs and CB1 receptors are also involved in non-canonical signaling pathways. For example, 2-AG has been shown to potentiate GABAA receptor activity at low GABA concentrations independent of CB1 receptors,38,39 while CB1 receptors are also modulated by non-classical ligands, such as pregnenolone.40,41
Despite the availability of detailed knowledge about the molecules and neuronal compartments involved in the eCB signaling system, how this molecular signaling pathway actually functions in particular synaptic-cellular circuits in behaving animals has remained incompletely understood, raising many fundamental questions. For example, can eCBs be generated in postsynaptic cells in response to physiological neuronal activity during normal behaviors (e.g., locomotion), or do they require unnaturally large stimuli similar to the depolarizing pulses used in DSI experiments in acute brain slices? Which eCB species is actually generated postsynaptically during the continuously fluctuating neuronal activity that typically accompanies natural behaviors? And are the eCBs generated in sufficient quantities by the active postsynaptic cells during physiologically relevant brain states to reach CB1 receptors on the presynaptic GABAergic terminals that impinge on them? Does a DSI-like suppression of inhibitory synaptic events exist in vivo? What are the neuronal dynamics of the CB1-expressing CCKBCs during naturally occurring brain states? And, finally, can CB1 receptor-dependent control of GABA release influence neuronal coding properties? In the current review, we primarily focus on the CA1 circuit of the hippocampus to illustrate how neurotechnological advances in the last few years have enabled researchers to answer some of these long-standing, fundamental questions about the eCB system in the neuronal circuits of behaving animals. These recent results highlight exciting new opportunities for research efforts aimed at developing an integrated, multi-scale understanding of molecular-level eCB signaling pathways engaged during natural behaviors at specific synapses under normal conditions and in psychiatric and neurological disorders.
MOLECULAR TRACKING OF eCB FLUCTUATIONS IN THE HIPPOCAMPUS IN VIVO
Development and validation of a novel eCB sensor
As mentioned above, biochemical and electrophysiological assays have suggested that a key property of the eCB system is the lability of its ligands, with signaling occurring over a timescale of seconds and over distances spanning just tens of micrometers.23,42 However, the majority of conventional, “gold standard” approaches used to study this system typically lack the spatiotemporal specificity that it requires. For example, post-mortem biochemical analyses of eCBs, typically measured with liquid chromatography and mass spectrometry, cannot capture potentially rapid fluctuations of eCBs during behavior. Even in the case of seizures, which are expected to capture supraphysiological levels of eCBs and are often used for validation of activity dependence, studies have yielded inconsistent results.43–46 In vitro electrophysiological experiments, such as DSI, have provided more clues on the spatiotemporal dynamics of eCBs but are indirect measures that, until recently, lacked in vivo validation (see below). Thus, a fundamental barrier to answering some of the key questions posed above has been the lack of a highly adaptive molecular tool that functions at timescales relevant for fluctuating neuronal activity in vivo.
The recent development of the GPCR activation-based eCB2.0 sensor (GRABeCB2.0) has overcome this limitation by allowing the detection of endogenous eCBs with fast kinetics.47 GRABeCB2.0 consists of the CB1 receptor as its scaffold coupled to green fluorescent protein, such that a CB1 agonist increases the amount of fluorescence emitted (Figure 1A). GRABeCB2.0 has similar pharmacological properties to the native CB1 receptor, such as increased concentration-dependent activity with endogenous, phyto-, and synthetic agonists and reversal of fluorescence activity with antagonists and cannabidiol (CBD).48 Moreover, with the lack of an intracellular signaling domain, recruitment of downstream effector proteins has not been observed and should therefore not interfere with cellular physiology at reasonable expression levels (note that, as with any sensor, some degree of agonist buffering will occur, highlighting the importance of avoiding overexpression).47 Furthermore, GRABeCB2.0 has onset (1.6 s) and offset (11.2 s) kinetics on second-long time scales,47 which are well-suited to capture the dynamics of DSI and DSE and highlight its utility for the in vivo study of neuronal activity. The sensor shows high specificity for eCBs when tested for a variety of neurotransmitter and neuromodulator ligands and a robust fluorescence response at physiological eCB concentrations (half-maximal effective concentration or EC50 for 2-AG and AEA is in the micromolar and submicromolar range, respectively). Any potential limitations that may arise for certain applications, for example, in kinetics, sensitivity, or specificity, are likely addressable by improvements introduced with newer versions of the tool (e.g., versions of the eCB sensor that can differentiate between 2-AG and AEA).
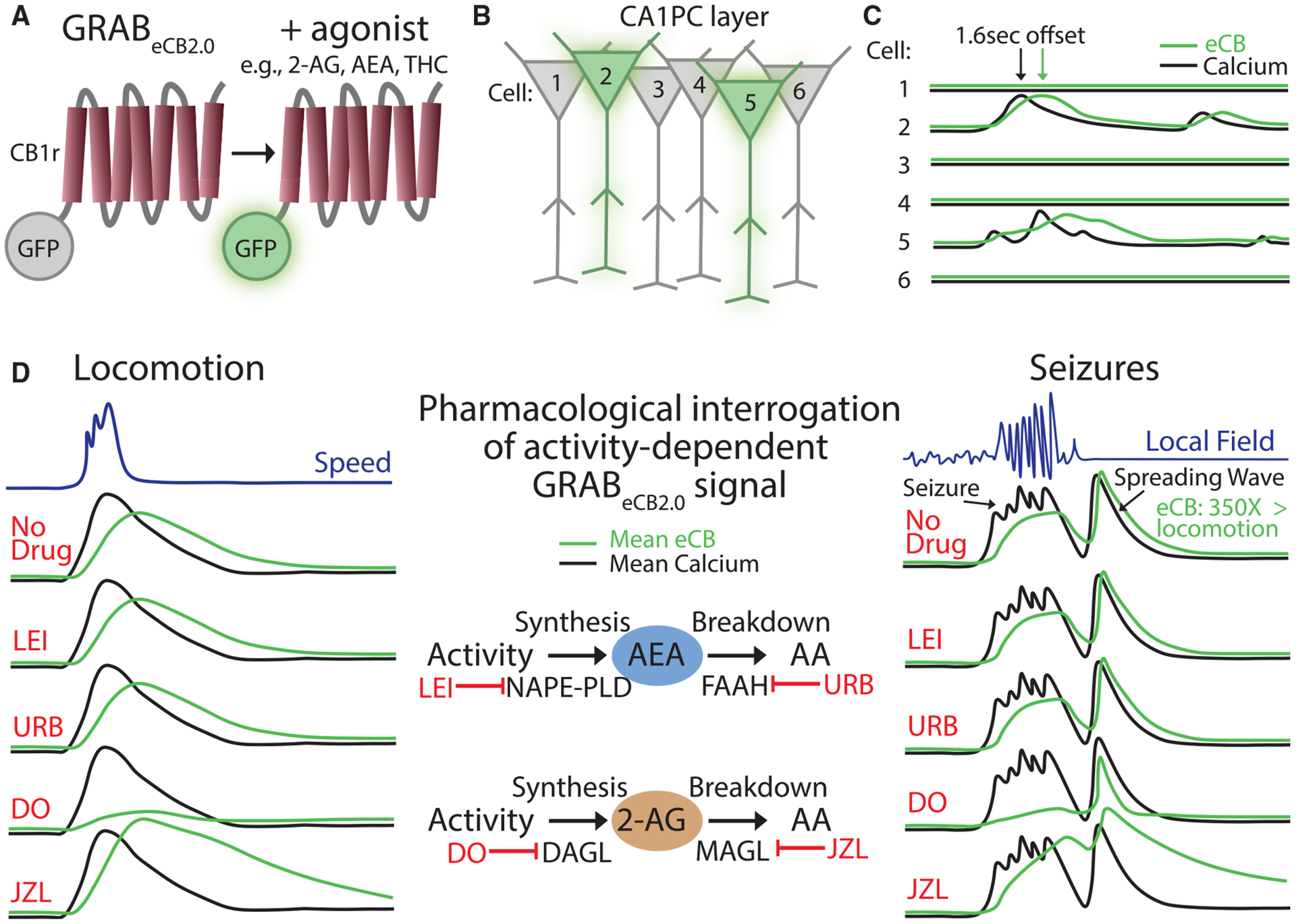
GRABeCB2.0 captures eCB elevations during electrical stimulation of cultured neurons, which are blocked by AM251, a CB1 receptor antagonist/inverse agonist. Interestingly, blocking the synthesis of 2-AG, but not AEA, successfully blocks the GRABeCB2.0 signal, suggesting that 2-AG primarily underlies this signal.47 Similar results were also obtained from slices of mouse striatum, including the observation that blocking 2-AG degradation slows the decay and that no signal change was observed in mutant mice lacking the 2-AG synthetic enzyme, DAGL.49 These results are consistent with prior in vitro work demonstrating a crucial role of 2-AG in short-term and long-term depression and motor control50,51 and establish GRABeCB2.0 as a useful tool for resolving 2-AG dynamics, setting the stage for its use in vivo.
Dynamics and spatiotemporal specificity of eCB signaling during physiological neuronal activity in vivo
With the substantial in vitro validation outlined above, this tool was ready to be applied in vivo, either with fiber photometry47,52 or two-photon imaging.3,47 The first question concerned the relative contribution of 2-AG versus AEA during physiological neural activity in the hippocampus. Utilization of two-photon imaging in awake, behaving mice revealed increases in GRABeCB2.0 activity in CA1 PCs, closely following the elevated calcium signal during locomotion with an approximately second-long delay3 (Figures 1B and 1C). Much like the in vitro work above, pharmacology was then used to dissect the contribution of AEA or 2-AG to this signal (Figure 1D). Blockade of 2-AG synthesis led to suppression of the GRABeCB2.0 signal in PCs, while blockade of 2-AG breakdown led to a robust increase with a prolonged decay. Notably, similar signal changes with enzymatic manipulation of 2-AG were observed with fiber photometry at ventral hippocampal to amygdala synapses.52 Enzyme inhibitors specific for AEA, on the other hand, resulted in no changes in GRABeCB2.0 signal.3 Therefore, these findings strongly suggest that 2-AG is the primary eCB generated in response to hippocampal neural activity in vivo.
The second question concerned the spatial specificity of the eCB signal carried by 2-AG in the CA1 in vivo. Importantly, the coupling between the calcium and the GRABeCB2.0 signals revealed spatially precise eCB signaling, as indicated by the observation that the eCB signal in a given neuron was more correlated to that cell’s calcium signal than that of other nearby cells. These results suggest a remarkable spatiotemporal specificity of eCB signaling, confined to when and where neural activity occurs. We will return to the topic of the spatiotemporal specificity of eCB dynamics when we discuss eCB signaling shaping perisomatic GABAergic inhibition as a function of hippocampal PC activity during spatial navigation (see below).
Utilization of GRABeCB for studying eCB signaling during pathological neuronal activity
Given the abundant expression of the eCB system at synapses throughout the brain, it is unsurprising that it is thought to be involved in a variety of significant psychiatric and neurological brain disorders, including autism, addiction, pain, psychosis, eating disorders, alcohol use disorder, epilepsy, and cannabis use disorder.1–5 Increasing evidence, backed by genetic studies, has suggested that the eCB system undergoes changes during various pathological processes, but the function and contribution of these changes to disease pathophysiology often remain unclear.53–58 Application of GRABeCB2.0 to study activity-dependent eCB dynamics offers a promising method for a direct readout of its engagement in models of neurological disease. For example, acute seizures were shown to lead to an over 100-fold increase in both the calcium and the GRABeCB2.0 signals in the CA1.3 Much like in the case of physiological activity, pharmacology revealed a crucial role of 2-AG in seizure-related GRABeCB2.0 signal (Figure 1D). This signal decayed back to baseline within a minute of seizure termination, which explains how prior studies using conventional biochemical methods that require removal of the brain were unable to capture this increase in 2-AG.43–46 Finally, the use of two-photon imaging to resolve the spatial patterns of seizure-related 2-AG increases revealed that the calcium and eCB activity of the entire CA1 PC network was engaged during the seizure, highlighting a loss of spatial specificity,3 and was followed by a traveling wave in both signals that was often larger than the seizure per se59 (Figure 1D). Thus, visualizing when and where eCBs are released in pathophysiology can lead to important insights, such as the potential role of spreading depolarization, which has now been consistently observed in a range of seizure models,60 in hijacking the molecular machinery that drives eCB synthesis.
CELLULAR DYNAMICS OF THE CB1 RECEPTOR-EXPRESSING INs ACROSS BRAIN STATES IN THE HIPPOCAMPUS OF BEHAVING ANIMALS
Genetic access to CB1 receptor-expressing hippocampal BCs
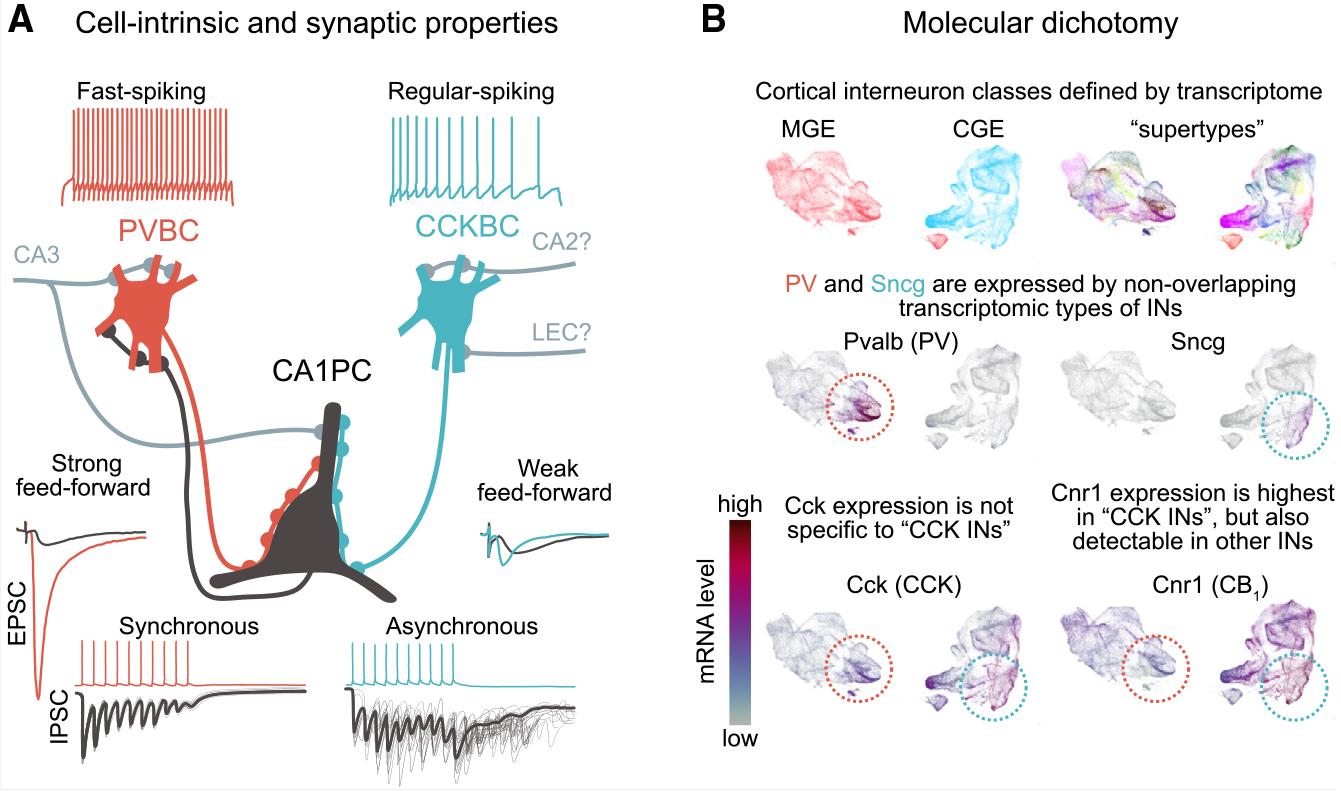
Despite being the most abundant GPCR in the brain, CB1 receptor expression is highly specific. In fact, although there are numerous IN types in the hippocampus, CB1 receptors are known to be predominantly present on the axons of CCK-expressing INs,8 whereas other prominent classes, including parvalbumin- (PV) and somatostatin-expressing, do not express CB1 or express it at much lower levels. However, due to the previous lack of CCK IN-specific molecular markers (e.g., CCK and CB1 receptors are expressed not only by CCK INs but also by PCs, albeit at a much lower level), genetic access to CCK INs has been limited (Box 2). Therefore, until recently, our knowledge of the CB1 receptor-expressing, perisomatically projecting CCKBCs in the hippocampal area CA1 in vivo has largely been based on a small number of recordings (from 7 cells in total) in anesthetized rats.61,62
One promising solution to this limitation was to combine single-cell resolution two-photon imaging of broad IN populations in vivo with post hoc immunocytochemical cell type identification.70,71 Acousto-optic deflection (AOD) microscopy-based three-dimensional (3D) in vivo calcium imaging of diverse GABAergic cells labeled in mice expressing Cre recombinase under the control of the vesicular GABA transporter (VGAT)70,71 provided key insights into CCK IN dynamics in awake, behaving animals (as discussed below). However, this approach did not distinguish CCK IN subtypes, including dendritically targeting CCK INs and CCKBCs with distinct eCB sensitivity and function (see below), and the two non-overlapping subtypes of CCKBCs that express either vasoactive intestinal polypeptide (VIP) or vesicular glutamate transporter type 3 (VGLUT3).72,73
A major step forward in studying specifically CCKBCs in vivo occurred with the development of a new mouse line that allows for specific, genetic targeting of this cell type.66 Using single-cell transcriptomics, the gamma synuclein (Sncg) gene was found to be selectively expressed in subsets of CCK cells74–76 and was used as a specific marker to target CCK INs in the Sncg-IRES2-Flp (Sncg-Flp) mouse line66 (Box 3). Importantly, electrophysiological recordings revealed that most neurons labeled by this technique exhibited an accommodating firing pattern66 (Box 3 image, panel A), and axons of the labeled neurons were restricted to the pyramidal layer, indicating that they constitute primarily CCKBCs (and not dendritically targeting CCK cells). In addition, Patch-sequencing experiments revealed that most CCKBCs labeled in the CA1 belonged to the VGLUT3 subtypes, with a non-overlapping minority expressing VIP. The development of the Sncg-Flp mouse line therefore allowed the first selective, population-level study of CCKBCs in vivo.
Activity dynamics of CB1 receptor-expressing CCKBCs in vivo
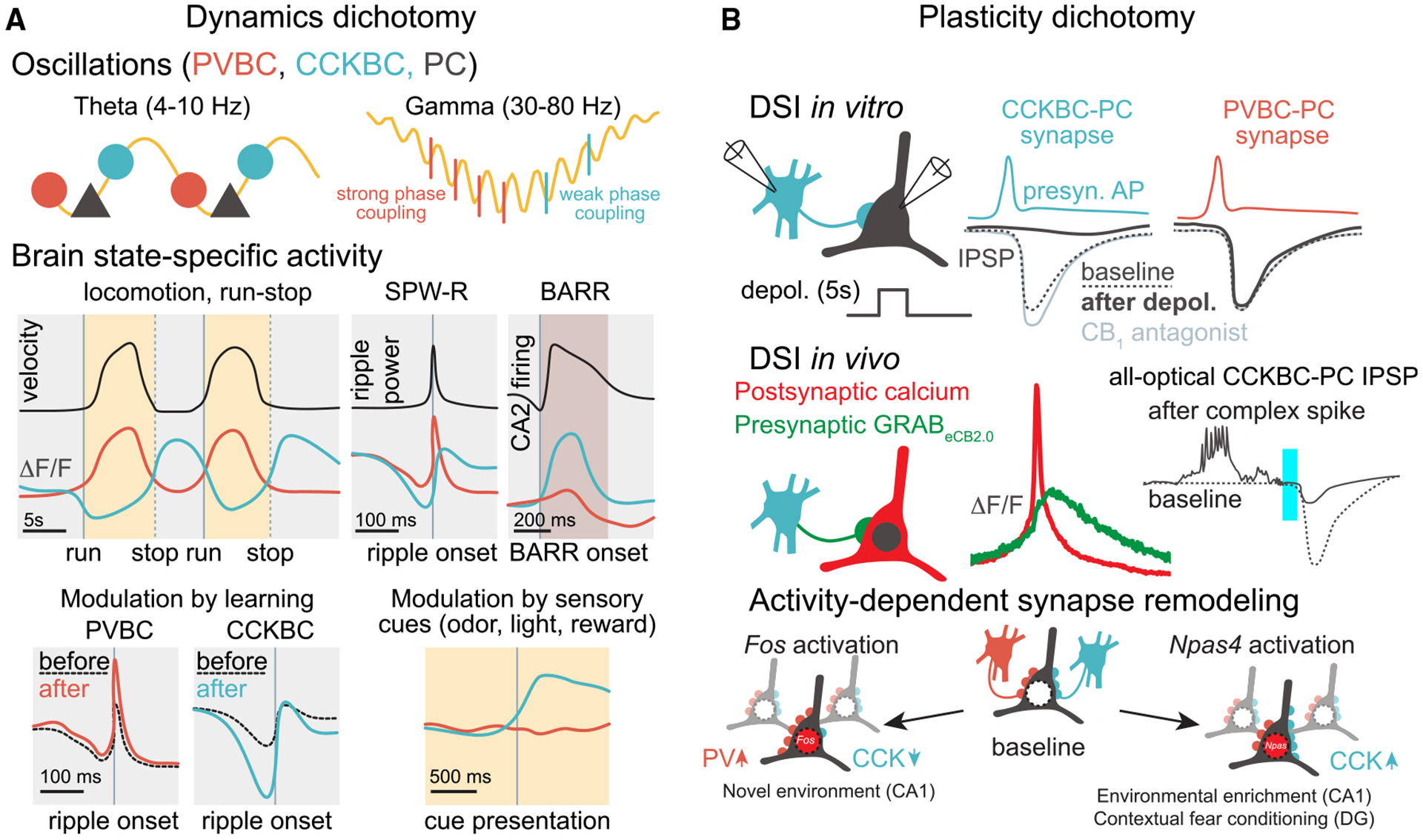
Previous immunocytochemical and in vitro electrophysiological studies have suggested that CCKBCs may be tuning network excitability in response to behavioral states,83 but direct evidence in support of this inference was lacking. In vivo two-photon calcium imaging of the Sncg-Flp mouse line found that CCKBCs are indeed robustly modulated by behaviorally relevant brain state transitions.66 Interestingly, CCKBC activity was found to be largely anticorrelated on second-long time scales with surrounding network activity, including that of PV-expressing BCs (PVBCs) that exhibit complementary properties to CCKBCs in many synaptic and single-cell electrophysiological properties (Box 3 image, panel A).77 In vivo imaging studies using the Sncg-Flp line further highlighted this dichotomy. For example, during theta oscillations (4–10 Hz), which correlate with an engaged brain state and occur when an animal is running, the activity of CCKBCs generally decreases while PVBC activity increases.66 Extracellular silicone probe recordings from optotagged Sncg/CCKBC units indicated that there was not a complete suppression of CCKBCs during theta oscillations, in agreement with studies using in vivo juxtacellular recordings that have shown that the recruitment of CCKBCs and PVBCs takes place during distinct phases of the locomotion-associated theta rhythm,61,84,85 revealing a temporal segregation of PVBC and CCKBC activities even on the timescale of tens of milliseconds. At the end of the locomotion epochs (i.e., when the animal stops running and enters a period of immobility), theta oscillations give way to a period of irregular, low-amplitude local field potential (LFP) activity. During this locomotion-to-immobility transition time, there was a rapid decline in PC and PVBC activity, whereas CCKBC activity rose to a maximal level.66 The latter transient, maximal CCKBC activity dynamic is referred to as the “run-stop response”70 (Figure 2A). A few seconds after the animals stop running, sharp wave ripples (SPW-Rs), which are ensemble network events that are linked to memory replay and consolidation,86 start to appear on the LFP trace. During SPW-Rs, PVBCs were strongly recruited, whereas CCKBCs were suppressed. Taken together, these in vivo studies showed that the neuronal dynamics of PVBCs and CCKBCs are strictly complementary and alternating across different brain states and time scales. We refer to this complementarity as “inverse scaling” below (Figure 2A).
Mechanisms of alternating sources of perisomatic inhibition in vivo
What may be the mechanistic underpinning of the inverse scaling between these two BC classes? As mentioned above, hippocampal PVBCs receive many excitatory inputs from both from local (CA1) and upstream (e.g., CA3) sources, which may explain why they can faithfully follow the overall activity levels within the PC populations. In comparison, the inversely correlated activity of CA1 CCKBCs with respect to the PVBCs predicts that this cell type may receive a unique pattern of excitatory and inhibitory inputs. Indeed, CCKBCs receive monosynaptic inhibition from PVBCs,66 which may explain the observed suppression during locomotion/theta oscillations and SPW-Rs during rest, when PVBC activity is high. In addition, CCK INs are innervated by local collaterals of theta-off/ripple-on (TORO) long-range projecting GABAergic cells, potentially contributing to the relative suppression of CCKBC activity during SPW-Rs.82
The excitatory inputs that activate CCKBCs remain poorly characterized. In general, CCKBCs receive weaker feedforward and feedback excitation and altogether fewer excitatory synapses compared with other INs, both in absolute numbers and relative to inhibitory synapses.83,87 The increase in CCKBC activity during the run-stop periods suggests that these cells play a crucial role in brain state transitions and potentially in currently incompletely understood spatial navigation-related circuit computations following the termination of locomotory bouts.66 What may drive CCKBC activity during the run-stop response? Is the run-stop response of CCKBCs purely due to the decrease in inhibition from PVBCs after the cessation of locomotion, or are there excitatory afferents to CCKBCs that may be driving it? Interestingly, the afferents within the CA1 that were particularly active at the time of the run-stop response were subsets of axons originating from the LEC and CA266 (Box 3).
LEC inputs can include both glutamatergic and long-distance GABAergic afferents and are known to transmit information on reward-related and contextual cues to CA1 PC distal dendrites88,89 and also modulate INs that likely include dendrite-targeting CCK cells90,91 that may suppress dendritic spikes.92 However, whether CCKBCs are specifically innervated by LEC afferents and how such an innervation may shape CCKBC run-stop responses remains unclear. Although not yet directly shown, the CA2 may also provide inputs responsible for modulating CCKBC activity during the run-stop response. In CA2, a distinct population of PCs has been reported to represent current animal location during immobility and sleep, in association with a previously unidentified hippocampus-wide network pattern.93 Therefore, it is possible that CA1 CCKBCs are part of an immobility-associated network activated during the run-stop response. Moreover, during non-rapid eye movement (NREM) sleep, CA2 PCs initiate another previously undefined network event, firing a sustained barrage of action potentials (BARR).94 These non-theta, non-SPW-R events recruit CA1 CCKBCs, just like the run-stop response (Figure 2A). At the same time, other CA1 neurons, including PCs and PVBCs, are relatively silent. Interestingly, PCs that increased their activity the most during learning and subsequent SPW-Rs were the ones that were the most silenced during such barrages, possibly by CCKBCs. This relationship between past PC activity and present inhibition (i.e., the most active cells were more strongly inhibited) is distinct from DSI, which causes the most active cells to be the least inhibited, on the time scale of seconds. Whether the circuit mechanisms and cognitive functions of the run-stop response and CA2-driven sleep barrages are similar remains to be investigated in future experiments.
Most cortical regions, unlike CA1, are not targets of direct synaptic pathways from LEC or CA2, which raises the question of whether the inverse scaling of CCKBC and PVBC activity is specific to CA1. The answer is likely no, as cell types molecularly related to CCKBCs have inversely scaled activity patterns in the neocortex as well. In the primary visual cortex (V1), Sncg-expressing IN types, the homologs of CA1 CCKBCs, are inversely modulated during distinct brain states compared with PV INs.95 The brain state-specific activity modulation of V1 INs was strongly correlated to transcriptomic similarity. Moreover, the activity pattern of the so-called sleep down-state active (DSA) INs in the cortex96 also resembles that of CCKBCs in that DSA IN activity is inversely correlated to PCs and other INs (additional similarities include the observation that DSAs are transiently recruited when overall network activity drops and they receive relatively weak feedback excitation from the local circuit compared with other INs). While the Nkx2.1-positive DSAs in the neocortex are reported to be neurogliaform cells, not BCs,96 CCKBCs belong to the same broad transcriptomic group of cortical INs (Id2) and may be similarly modulated by brain state.65 Finally, in the hippocampal CA3 circuit, calbindin-positive SATB1 transcription factor-negative INs represent a major subset of immobility active INs,71 which have the same developmental origin as the CGE-derived CCK INs. Why would transcriptomic similarity predict similarity in brain state modulation?95 In addition to developmental specification, another potential explanation is that transcriptomically similar cell types share similar neuromodulator receptor repertoires and second messenger pathways. Considering the expression of various neuromodulatory receptors by CCKBCs, it is possible that the run-stop response is at least partially driven by cholinergic, noradrenergic, serotonergic, and/or dopaminergic inputs.79,97 Recent developments in a variety of GRABeCB sensors for these neuromodulators will help to better understand the interactions of neuromodulatory, GABAergic, and glutamatergic inputs shaping eCB and CCKBC dynamics.98
HIPPOCAMPAL PLACE CELL PROPERTIES AND THE eCB SYSTEM
What may be the behavioral relevance of the eCB system in relation to CCKBC dynamics, and how can tools such as GRABeCB2.0 and the unprecedented in vivo access to the CB1 receptor-bearing CCKBCs offered by the Sncg-Flp mouse line make it possible to study the latter question? As an animal navigates its environment, place cells in the CA1 fire at specific locations, known as place fields, and they collectively form a neuronal representation of space referred to as a “cognitive map.”99 Various studies have implicated the eCB system in spatial navigation. For instance, the use of synthetic cannabinoids has been shown to alter spatial memory and spike-timing coordination, although no alterations in location-specific place cell firing were detected.100,101 A major but previously not directly testable hypothesis has been that eCB-mediated inhibition of CCKBCs is likely involved in modulating place cell properties.61 This has specifically been hypothesized to occur through DSI, during which, as described above, in vitro observations revealed that strong depolarization of PCs ultimately leads to eCB-mediated suppression of GABA release for several seconds. Accordingly, increased place cell firing at place fields99,102,103 may lead to decreased GABAergic inhibition from CCKBCs through a DSI-like mechanism and a relative amplification of the excitability of only the currently active place cells, while other PCs would continue to receive normal levels of CCKBC-mediated GABAergic inhibition. In support of this proposed mechanism, in vivo studies indeed have found decreased inhibition corresponding to increased place cell activity104–106 but without direct evidence for the role of eCBs in this process.
Recently, two-photon imaging in behaving mice investigating the relationship between postsynaptic calcium signals in place cells linked to increased GRABeCB2.0 fluorescence within the same regions of interest offered the long-elusive clues about the retrograde transfer of eCBs that could mediate DSI in vivo.107 Specifically, when animals were navigating a linear track lined with tactile cues, increased activity-dependent calcium signals in place cells at their place field locations were associated both with increased post- as well as presynaptic eCB signals at the same locations.107 These findings indicating the existence of place fields for eCB molecular signaling during spatial navigation strongly suggested the potential involvement of DSI in place cell coding but still fell short of showing any kind of modulation of inhibitory postsynaptic potentials (IPSPs) as a function of postsynaptic neuronal activity in vivo. Although the direct measurements of subthreshold, fast inhibitory activity in behaving animals seemed out of reach even as recently as a few years ago, advances in all-optical interrogation of circuit and cellular-synaptic properties using voltage imaging in combination with optogenetics108,109 now make it feasible to detect small hyperpolarizing voltage changes (i.e., IPSPs) in CA1 PCs not just in general but also in response to activation of specific subtypes of INs in vivo (Figure 2B). Previous studies using whole-cell recording in vivo have found that a particular PC firing pattern, plateau-driven complex spiking, plays an important role in synaptic plasticity and place cell formation,103,110 findings that recently have also been demonstrated using all-optical physiology.108,109 Importantly, plateau-driven complex spikes are large events associated with postsynaptic calcium entry and may therefore also lead to the synthesis and release of eCBs, which is consistent with the increased eCB signal at place fields. Indeed, the combination of voltage imaging and genetic access to CCKBCs provided by the Sncg-Flp mouse line demonstrated that CCKBC-evoked IPSPs in CA1 PCs were significantly smaller when preceded by plateau-driven complex spikes in the same PCs, in agreement with what would be expected from a DSI-like phenomenon107 (Figure 2B). Notably, mice lacking CB1 receptors in INs, which mostly affects CCK cells, displayed a widening of place fields. Furthermore, population-level encoding of the animal’s position was less accurate, indicating impaired place cell properties in the absence of eCB control of CCKBC synapses. These findings indicate that a DSI-driven phenomenon in vivo, relying on presynaptic CB1 receptors on GABAergic terminals, is crucial in regulating the precision of place cell firing and supporting spatial navigation.107
ADDITIONAL COGNITIVE FUNCTIONS OF CCKBCs AND THE eCB SYSTEM
As described above, CCKBCs and the eCB system play important roles in cognitive functions. For instance, the increased activity of CCKBCs during the run-stop response suggests their potential involvement in implementing brain state transitions via changes in local circuit dynamics, and in vivo evidence of DSI suggests a role of eCBs in behaviorally relevant neural activity related to spatial navigation. Studies in the CA3 of the hippocampus suggest that CCK cells may also be involved in other cognitive functions, such as memory consolidation and selective attention. Indeed, using AOD 3D calcium imaging, CCK cell dynamics suggested that they may play a significant role in modulating SPW-Rs in the CA3.71 Specifically, the magnitude of inhibition of CCK cells before the SPW-R was correlated with the duration of the SPW-R. Interestingly, it was the magnitude of PVBC response after the SPW-R that was associated with the duration of the SPW-R. This study also demonstrated that spatial learning induces changes in CA3 inhibitory network dynamics such that PVBCs and CCKBCs become more activated and inhibited, respectively, around SPW-Rs after learning,71 further showcasing the dichotomy of CCK and PV cell dynamics in vivo (Figure 2A). Given that the duration of SPW-Rs is likely to be related to memory performance,111 these results further highlight the potential roles of CCK INs in cognitive processes.
Importantly, the Vancura et al.71 study also reported that CCK cells were preferentially activated in response to sensory cues, such as reward, light, and odor. Although we do not yet have direct evidence for the role of these cue-responsive CCK cells, they may significantly contribute to selective attention. Schaffer collaterals, originating from CA3 PCs and terminating on CA1 PC dendrites in the radiatum, are thought to be important for encoding and consolidating memories by driving SPW-Rs.112–115 Interestingly, a recent study found that Schaffer collateral axons in the CA1 were activated by specific sensory cues, but their activation was excluded from SPW-Rs if the associated cues were not spatially relevant.116 SPW-Rs therefore are preferentially biased to behaviorally relevant information and actively suppress irrelevant stimuli. In other words, as an animal navigates its environment and encounters a barrage of sensory information, only the most relevant cues, such as those pertaining to the location of food or important landmarks, are encoded during SPW-Rs.116 Moreover, in a different study using a virtual reality maze, CA1 CCK INs showed a strong modulation of activity depending not only on reward but also on the animal’s interaction with the maze (i.e., cells were activated when the virtual reality was turned off, and therefore sensory information was not salient to the task).117 Given that non-salient cues were actively inhibited during SPW-Rs and that CCK cells display the strongest cue responses in the CA3 during active exploration71 (Figure 2A), it is possible that these cells are modulating and filtering the CA3-CA1 circuit based on cue salience,118 potentially further extending the cognitive impact of CCK INs.
The plasticity of CCKBC synapses may also play a role in contextual memory. The consolidation of contextual fear memory involves synaptic plasticity to facilitate the reactivation of unique neuronal ensembles representing specific memories—these ensembles are frequently called memory engrams.119 The role of inhibitory synapse remodeling in these processes remains the focus of inquiry. Distinct activity patterns of PCs result in the activation of distinct immediate-early gene networks (Box 3 image, panel B), characterized by the expression of either Fos or Npas4.120 Interestingly, Npas4 activation enhances inhibitory inputs from CCK INs in the dentate gyrus and CA1,120–122 while Fos activation leads to enhanced PV IN inputs and reduced CCK IN synapses through a mechanism involving neuropeptides encoded by the Scg2 gene.123 Such inhibitory plasticity, and the activity of CCK INs, is required for the emergence of selective engrams and for discrimination during recall.120,124 As the Fos and Npas4 regulators can be activated in different cells at the same time, these mechanisms likely contribute to the development of circuits preferentially innervated by either CCK or PV INs.
OUTSTANDING QUESTIONS
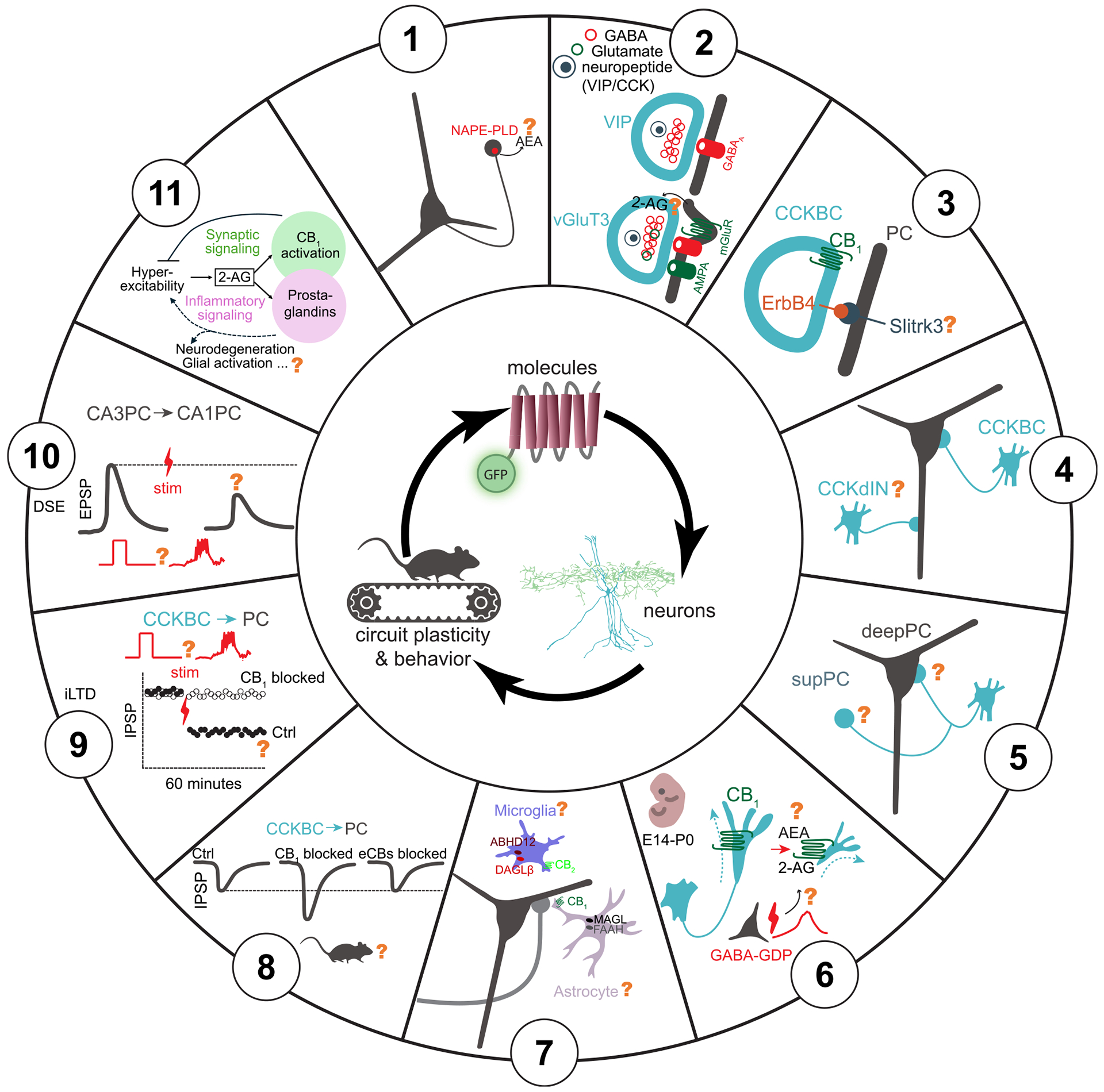
Finally, we briefly summarize what we believe are some of the most important questions (Figure 3) concerning the eCB system that the field can now begin to address by taking advantage of some of the new technical advances discussed above. We begin with questions related to molecules involved in eCB signaling, move to those pertaining to cellular circuits, and finally end with questions with the most behavioral and pathological relevance.
Dynamics of AEA
Previous measurements obtained in vivo from several groups utilizing GRABeCB2.0 have found a key role for 2-AG in the underlying phasic signal changes.3,47,49 It is still unclear how AEA dynamically fluctuates, both spatially and temporally, as it relates to ongoing neural activity. Since AEA levels are generally orders of magnitude lower than 2-AG, overcoming this challenge may come from the development of new sensors designed for higher AEA sensitivity (GRABeCB2.0 EC50 for AEA is between 200 and 800 nM). Another consideration is that AEA may have fundamentally distinct spatiotemporal dynamics. One proposed model for AEA function, which is a partial CB1 receptor agonist, is that it acts as a slow, tonic signal that sets the tone of the CB1 receptor, whereas the full agonist, 2-AG, fine-tunes CB1 receptor activity with high temporal precision.125–127 At perisomatic CCK synapses, this interaction may be more complex, since FAAH inhibitors that elevate AEA levels can regulate phasic 2-AG synthesis via transient receptor potential vanilloid type 1 (TRPV1) receptors, while CB1 receptors on dendritically targeting CCK INs are not regulated by FAAH inhibition.128 Thus, the temporal dynamics and synapse-specific functions of AEA are unclear. In addition to the potentially unique temporal dynamics of AEA, it is also possible that circuits that have a greater bias for AEA production have not been investigated yet. Within the hippocampus, for example, only the CA1 has been studied, but levels of the biosynthetic enzyme for AEA, NAPE-PLD, are higher in the CA3 and DG.129 At a subcellular level, it is important to note that NAPE-PLD is expressed in dentate granule cell axon terminals, which do not have CB1 receptors.130 Thus, AEA may not function as a traditional retrograde transmitter (i.e., from soma/dendrite to axon), and, as highlighted above, likely has other molecular targets than the CB1 receptor, such as the TRPV1 receptor. Thus, unlike 2-AG, which has significant in vitro data to formulate hypotheses for how it might act in vivo, a hypothesis for when and where AEA is synthesized, where it acts, and how long its activity persists is less straightforward.
Co-release of neuroactive substances from CCKBCs
A defining feature of CCK INs is the expression of the CCK neuropeptide, which is present in dense-core vesicles in these cells. In vitro, exogenously applied CCK has profound effects on inhibition, including a robust activation of PVBCs and the CCK receptor-mediated synthesis and subsequent release of eCBs.131,132 The eCBs depress GABA release from CCKBC terminals, amplifying perisomatic inhibition originating from PVBCs at the expense of CCKBCs.131,132 Whether CCK can be co-released with GABA from CCK INs during behavior remains to be established, likely aided by the recent availability of the GRABeCB sensor.133 In addition, as briefly mentioned above, a subset of CCK INs (including both CCKBCs and dendritically targeting CCK INs) co-express VGLUT3 (encoded by the Slc17a8 gene), and this subset appears to be mutually exclusive with VIP-expressing CCK INs based on immunohistochemistry.72,73 Further evidence indicates that VGLUT3-expressing CCKBCs constitute a distinct cell type where glutamate-GABA co-release can occur at their output synapses with potentially unique functional roles.73 Interestingly, the Sncg-Flp line mostly labels VGLUT3 CCKBCs (see above), and the role of ErbB4 in CCKBC wiring (see below) also appears to be specific to VGLUT3-expressing cells, as ErbB4 expression is only marginally present in VIP-expressing CCKBCs.134 In the amygdala, VGLUT3-containing CCKBCs form specialized, invaginated synaptic structures on postsynaptic PCs, rich in metabotropic glutamate and CCK receptors, and molecules involved in the downstream eCB-synthesizing enzymatic pathway.135 Thus, co-release of glutamate may trigger retrograde eCB signaling, forming a negative feedback loop to suppress GABA release after particularly high presynaptic activity at CCKBC synapses. Furthermore, in the hippocampus, co-released glutamate can also activate postsynaptic AMPA receptors at VGLUT3 CCKBC synapses, which may render these synapses paradoxically excitatory under pathological conditions such as in epilepsy.73 However, the in vivo existence and roles of CCK and glutamate co-release from CCKBCs remain an open question.
Role of ErbB4 in the integration and function of CCKBCs in hippocampal networks
ErbB4, a receptor tyrosine kinase, has been shown to be essential for the successful integration of CCKBCs and PVBCs into hippocampal networks,136 including the establishment and refinement of their excitatory inputs.6,134,137–141 Specifically, ErbB4 expression in CCKBCs is crucial for their synaptic connections with both PCs and PVBCs.134 A key question concerns the identity of the postsynaptic adhesion molecules that interact with ErbB4 to determine the specific targets of CCKBC synapses. One potential candidate is the transmembrane protein Slitrk3, recently shown to interact with ErbB4 and promote the formation of inhibitory synapses on PCs.142 Importantly, specific deletion of ErbB4 in VGLUT3 CCKBCs during development has been shown to alter inhibitory signaling onto PCs, reduce the power of theta oscillations during exploratory behavior, disrupt spatial coding by place cells, and selectively impair spatial learning and memory in adult mice.134 Many of these disruptions likely occur due to impaired eCB signaling. These findings are particularly intriguing, especially the role of VGLUT3 CCKBCs in modulating theta oscillations, a role previously mainly attributed to PVBCs. Given that, as discussed above, VGLUT3 CCKBCs can release both GABA and glutamate onto postsynaptic PCs,72,73,135,143,144 it is plausible that it is this co-release mechanism that endows CCKBCs with unique computational properties that could be crucial for modulating theta oscillations.
Functions of dendritically projecting CB1 receptor-expressing INs
Different compartments of PCs receive unique compositions of local and long-range inputs. Recent studies have shown that different dendritic compartments exhibit distinct activity and plasticity dynamics during place field formation.109,145,146 Although most attention has been focused on CCKBCs whose perisomatic synapses are implicated in shaping place cell properties during spatial navigation through a DSI-like mechanism (as discussed above), dendritically targeting CCK INs likely also impact information processing and may be regulated by the eCB system in distinct ways. Dendritically targeting CCK INs are composed of at least seven identified subtypes, distributed throughout all layers of CA1.147,148 There is much left to be discovered about most of these subtypes, as evidenced by the very small sample sizes (usually only 1–2) for in vivo recorded and identified cells for each of these dendritically projecting CCK IN subtypes. It is likely that dendritically targeting CCK INs have eCB signaling properties and in vivo activity dynamics that are distinct from CCKBCs. For example, CA1 Schaffer collateral-associated CCK INs that project to the stratum radiatum in the CA1 have been shown to display no or considerably weaker DSI, CB1 receptor-dependent tonic inhibition of GABA release, and metabotropic glutamate receptor activation-induced, CB1 receptor-mediated depression of GABA release. This is the case in spite of the prominent presence of all members of the eCB-synthesizing molecular machinery in the dendrites of postsynaptic principal cells and the expression of functional CB1 receptors in the axon terminals of these INs.10 Furthermore, molecularly identified CCK INs, which include both CCKBCs and dendritically targeting INs, exhibit a more heterogeneous velocity modulation profile compared with immobility active CCKBCs, suggesting that at least some dendritically projecting CCK INs are active during locomotion.70 Therefore, dendritically projecting CCK INs may have differential effects on behavioral readouts and eCB-mediated processes. Indeed, these cells are well-positioned to modulate PC activity in an input-specific manner, since different compartments of PCs receive distinct excitatory inputs from distinct intra- and extrahippocampal areas.147 Along these lines, dendritically projecting CCK INs have been suggested to be involved in gating dendritic spikes driven by LEC inputs.92
eCB modulation of perisomatic inhibition onto heterogeneous CA1 PC populations
As previously discussed, CCKBCs and PVBCs modulate CA1 PCs in opposite yet complementary ways.66 This dichotomy is exacerbated by their differential synaptic connectivity with respect to the heterogeneity present in the CA1 PC population itself, characterized by differences in PC soma positioning along the radial axis (from superficial to deep layers), distinct neurochemical markers, and long-range projection patterns.149–151 Surprisingly, it has been discovered that PVBCs exert approximately 3-fold stronger inhibition onto CA1 PCs located in the deep compared with superficial layer of the stratum pyramidale.152,153 Additionally, superficial PCs more frequently provide excitatory inputs to PVBCs than their deep counterparts. PVBC-to-PC inhibition also segregates along PC projection patterns. For example, PVBCs preferentially innervate PCs projecting to the amygdala compared with their prefrontal cortex-projecting neighbors but receive preferential excitation from PCs projecting to the prefrontal cortex and much less from the amygdala-projecting PCs.152 Whether such preferential innervation of PCs also applies to CCKBCs is not yet established, with one study finding no overt selectivity,152 whereas another report suggests that CCKBCs may primarily inhibit superficial CA1 PCs.153 Interestingly, CCKBCs do indeed show robust selectivity for postsynaptic principal cell populations in layer II of the medial entorhinal cortex.154 These observations raise multiple questions, for example, related to the additional computational properties potentially provided by eCB signaling in a selective manner to certain PC populations preferentially targeted by CCKBCs in neuronal circuits. Exploring such questions could provide valuable insights into the distinct roles of CCKBCs and PVBCs and may also yield novel molecular-genetic interventional tools to study their impact on learning and memory.
Neuronal activity-dependent roles of the eCB signaling system in the developing brain in vivo
The eCB signaling system plays a variety of crucial roles in the developing nervous system, from lineage segregation of stem cells and excitatory and inhibitory synapse positioning to the refinement of synaptic functions and the control of adult neurogenesis.155–157 Indeed, the highest levels of CB1 receptor expression occur as synaptic connectivity is established during embryonic (PCs) and early postnatal (GABAergic INs) development.155 Pharmacological interference with the CB1 receptor suggests that these receptors modulate synchronous GABAergic depolarizing network events, known as giant depolarizing potentials (GDPs), in the developing hippocampus.158 The emerging novel tools and approaches described in this review may also shed light on how the eCB-mediated activity-dependent feedback mechanisms shape spontaneous coincident neuronal population dynamics, including those linked to externally generated sensorimotor activity during early postnatal development.159
eCB signaling in glia
Although we have focused on the eCB system in neuronal pathways, growing evidence suggests that astrocytes and microglia play important roles related to eCB activity, function, and metabolism.160,161 Astrocytes, the most abundant cell type in the central nervous system, are classically known for their role in neuronal support, homeostasis, and synapse function.162,163 These cells express functional CB1 receptors, which lead to astroglia calcium increases that can subsequently spread along the cell and lead to the release of neuroactive substances referred to as “gliotransmission” (typically involving ATP and glutamate), as well as short- and long-term potentiation.164–169 Additionally, astrocytes express TRP channels and other GPCRs that may be modulated by cannabinoids.170–172 In terms of metabolism, astrocytes notably express MAGL and FAAH, enzymes that are responsible for 2-AG and AEA breakdown, respectively (Box 1), and are likely important for attenuating neuronal cannabinoid signaling at the so-called “tripartite” synapse involving pre- and postsynaptic structures and astrocytic processes.173–177 eCB signaling in astrocytes likely also plays a significant role in inflammation, based on studies suggesting that exogenously applied AEA and synthetic cannabinoid analogs can have anti-inflammatory effects.178–182 Along the same lines, inhibitors of MAGL have been shown to reduce lipopolysaccharide-induced inflammation.175,183 However, the exact pathways for these findings, as well as their implications in vivo, remain to be discovered. Furthermore, although there is some evidence that astrocytes may release 2-AG and AEA, it remains an open question whether these eCBs have functional relevance.184–187
Microglia, known for their immune functions in the brain, likely also contribute to eCB signaling and modulation. Unlike in astrocytes, however, the functionally relevant expression of CB1 receptors in these cells is not fully established.161,188,189 Interestingly, microglia do express Cnr2 transcripts, coding for CB2 receptors, at higher levels than neurons.190 Studies suggest that microglial CB2 receptors may facilitate communication between neurons and microglia and modulate glutamatergic neurotransmission.191–197 Furthermore, cannabinoid signaling, especially CB2 receptor expression, in microglia is activity dependent and likely contributes to modulation of neuroinflammation.198 Findings suggest reduced levels of infiltrating macrophages as well as a reduced proinflammatory drive upon CB2 receptor deletion.199 Modulation of eCB-mediated neuroinflammation likely also occurs through microglia expression of eCB synthesis and degradation enzymes, diacylglycerol lipase-β (DAGLβ) and α/β-hydrolase domain-containing 12 (ABHD12).200 Future studies can further explore these findings in vivo to dissect eCB-mediated microglial involvement in neuroinflammation and the modulation of neurotransmission.
Tonic inhibition of GABA release by CB1 receptors in vivo
In addition to their role in DSI-related activity-dependent short-term plasticity, CB1 receptors are known to also modulate GABA release from CCKBCs in a time-invariant manner.128,201 This occurs through their intrinsic, constitutive, ligand-free activity, most likely related to the ability of GPCRs to flip into an active conformation with some non-zero probability even in the absence of the ligand.128,201 This tonic regulation of GABA release by CB1 receptors (see also panel G in the Box 1 image) has been shown in in vitro studies to be a powerful regulator of the probability of release at inhibitory synapses of CCKBCs that can even lead to “silent GABAergic synapses.”10,28–30 At such “silent” GABAergic synapses, the probability of release is typically close to zero (i.e., a presynaptic action potential seems to evoke no postsynaptic responses in paired recordings in vitro), but it can be dramatically increased in the presence of an inverse agonist that blocks the constitutive GPCR activity.128,201 One promising indication that tonic eCB signaling alters neuronal activity during behavior comes from comparing neuronal activity between two visual regions with quantitatively different tonic eCB signaling.202 In the secondary visual cortex (V2), a relatively strong cannabinoid tone (observed in vitro) seems to coexist with relatively high spontaneous PC activity in vivo. By contrast, in primary visual cortex (V1), a lower cannabinoid tone and consequently stronger inhibition (assessed in vitro) seem to accompany lower spontaneous PC activity in vivo. Furthermore, treatment with the CB1 receptor antagonist/inverse agonist AM251 eliminates such differences in vivo,202 indicating that the cannabinoid tone may indeed modulate PC firing rates. Importantly, however, direct evidence for the existence of tonic inhibitory control of GABA release by CB1 receptors in vivo is still lacking, and it is also not known as to what degree it can depress GABA release in behaving animals. A related question is whether the selective presence of the tonic control of GABA release by CB1 receptors at perisomatically but not dendritically targeting CCK INs (see above) observed in vitro10,128,201,203 also applies to the in vivo situation. The importance of a better understanding of the in vivo status of the CB1 receptor-dependent tonic control of GABA release is underlined by the fact that it was found to be selectively disrupted in animal models of autism in acute hippocampal slices, without corresponding changes in DSI.204 These questions related to the CB1 receptor tonic activity in vivo can be explored in knock-out mouse models that lack the synthesizing enzyme for 2-AG (diacylglycerol lipase-α [DAGLα]−/−)16,17 or AEA (NAPE-PLD−/−).205
eCB-mediated longer-term plasticity of inhibition
In vitro experiments revealed that, on a longer time scale than DSI (on the order of minutes), cannabinoid-sensitive inhibition can also be tuned by long-term depression (referred to as iLTD). In CA1 PCs, theta-burst firing, designed to mimic in vivo patterns of place cell activity, triggered retrograde eCB signaling that persistently suppressed presynaptic inhibition.206 Interestingly, by contrast to DSI, which is expressed by a G-protein-mediated suppression of voltage-gated calcium channels and vesicle release, iLTD also requires presynaptic protein synthesis downstream to CB1 receptor activation.207,208 Whether iLTD-like long-term plasticity mechanisms contribute to place cell formation and maintenance during spatial navigation in the hippocampus in vivo remains to be investigated. Interestingly, a unique form of CA2 iLTD has been implicated in social memory formation in vivo.209
DSE during natural behaviors
While DSE has been demonstrated under in vitro conditions in a variety of brain circuits,25,210–214 it remains to be investigated if DSE exists in the intact brain in vivo during natural behaviors. A related and intriguing question is whether and how eCB signaling plays a role in behavioral timescale plasticity (BTSP). Intuitively, the large amplitude calcium channel- and NMDA receptor-mediated dendritic plateau potentials that induce BTSP should lead to eCB release and DSE at glutamatergic synapses. Therefore, DSE may preferentially suppress synapses that are active after dendritic plateaus and, in turn, regulate the shape of the bidirectional plasticity kernel of BTSP.215
Pathological eCB signaling in brain disorders
In agreement with the wide distribution and high density of CB1 receptors in a variety of reward, habit, and cognition-related circuits, eCB signaling has been implicated in a large number of neurological and psychiatric disorders, including epilepsy, pain, autism spectrum disorders, addiction, cannabis use disorder, eating disorders, anxiety, psychosis, aging, Alzheimer’s disease, schizophrenia, Huntington’s disease, Parkinson’s disease, and many others.1–5 Accordingly, key targets in the eCB system may provide therapeutic opportunities for such disorders. For instance, inhibition of 2-AG hydrolysis may be beneficial for certain epilepsies and neuroinflammatory diseases. 2-AG is primarily hydrolyzed by MAGL in presynaptic neurons and a smaller percentage by α/β-hydrolase domain-containing 6 (ABHD6) in postsynaptic neurons. Inhibition of ABHD6 has been shown to reduce excessive excitation during seizures by increasing 2-AG levels and allosterically increasing GABAA receptor activity.38,216–218 Beyond the canonical mechanisms of 2-AG-mediated control of synaptic function, 2-AG is also the starting point of another lipid signaling pathway. MAGL-mediated hydrolysis of 2-AG yields the cyclooxygenase-2 substrate arachidonic acid,219 and this eCB source of substrate plays a dominant role in brain prostaglandin production.220 In light of this finding, MAGL inhibitors have been tested in mouse disease models and demonstrated to ward off neurodegeneration in a model of Parkinson’s disease220 and neuropathological features in a model of Alzheimer’s disease.221 This dual role for synaptic and neuroinflammatory control by 2-AG is perhaps best exemplified during seizures, where activity-dependent production of 2-AG is hijacked by excessive neural activity to produce supraphysiological 2-AG levels. On one hand, dramatic 2-AG elevations can attenuate seizures via the CB1 receptor, which is consistent with work performed in a range of mouse models.222–225 Conversely, elevated 2-AG fuels prostaglandin signaling pathways,220 which were found to drive profound vasoconstriction after seizures, resulting in over an hour of severe local brain hypoxia.3 This work exemplifies the Janus-faced nature of 2-AG in some disease settings, given the potential benefit of synaptic signaling in restricting hyperexcitability and potential detriment of prostaglandin-mediated control of cerebral blood flow. In general, more work is needed to better understand how the multiple roles of 2-AG contribute to disease mechanisms. Utilizing biosensors to visualize the spatiotemporal dynamics of 2-AG and its downstream metabolites in different cell types, including microglia and astrocytes,216,226,227 alongside molecular profiling to determine enzyme and gene expression changes in disease could elucidate underlying mechanisms and therapeutic strategies. Moreover, MAGL inhibitors could prove to be a useful tool in augmenting 2-AG levels when and where they are locally produced, potentially leading to diminished side effects associated with CB1 receptor agonism, but can also restrict prostaglandin production and suppress the neuroinflammatory components of neurological and psychiatric disease.228 Utilization of biosensors has the potential to expand our knowledge about exogenously applied cannabinoids (exocannabinoids) as well. For example, the eCB sensor offers the possibility to visualize the modulation of presynaptic terminals by the psychoactive component of marijuana, delta-9-tetrahydrocannabinol (THC), that acts as a weak partial agonist at CB1 receptors, in various key reward- and cognition-related brain areas during behavior. Similarly, these new tools open novel opportunities to refine our understanding of how CBD, the first marijuana-derived compound to be approved for medicinal use in children with devastating forms of epileptic encephalopathy,229,230 may exert its beneficial effects in vivo.231 Finally, these approaches could now be employed to investigate exciting novel therapeutics with intracellular signaling-specific mechanisms of action, such as the possibility of inhibition of THC effects without producing behavioral effects per se for cannabis use disorder.41
CONCLUSION
As illustrated by the results highlighted in this review, tremendous progress has been made in gaining an integrative and translational understanding of the eCB system. The development of the GRABeCB sensor allowed for the in vivo imaging of eCBs at unprecedented spatiotemporal resolution and provided insights into their physiological roles. Identification of a specific marker (Sncg) for CCK- and CB1 receptor-expressing INs allowed for the first population-level study of these cells in awake, non-anesthetized animals, both in terms of activity monitoring and also by providing genetic access for their optogenetic and chemogenetic manipulations. The combination of the approaches represented by the eCB sensor and the Sncg transgenic mouse line then allowed for the first in vivo demonstration of DSI and showcased the importance of this phenomenon in place field formation. Finally, as the outstanding questions we have presented demonstrate, much remains to be discovered about the eCB system at the levels of molecules, cells, and behavior. There is an abundance of evidence pointing to the potential roles of the eCB system in various pathological states, but pinpointing therapeutic targets will require a multi-system approach that can begin to determine how disruptions of eCB molecular signaling pathways can lead to changes in CB1 receptor-expressing microcircuits with brain state-dependent behavioral consequences. For a glossary of the terms used in this review, see Box 4.
References
- I Soltesz, BE Alger, M Kano, SH Lee, DM Lovinger, T Ohno-Shosaku, M Watanabe. Weeding out bad waves: towards selective cannabinoid circuit control in epilepsy.. Nat. Rev. Neurosci, 2015. [DOI | PubMed]
- HV Curran, TP Freeman, C Mokrysz, DA Lewis, CJA Morgan, LH Parsons. Keep off the grass? Cannabis, cognition and addiction.. Nat. Rev. Neurosci, 2016. [DOI | PubMed]
- JS Farrell, R Colangeli, A Dong, AG George, K Addo-Osafo, PJ Kingsley, M Morena, MD Wolff, B Dudok, K He. In vivo endocannabinoid dynamics at the timescale of physiological and pathological neural activity.. Neuron, 2021. [DOI | PubMed]
- M Maccarrone, V Di Marzo, J Gertsch, U Grether, AC Howlett, T Hua, A Makriyannis, D Piomelli, N Ueda, M van der Stelt. Goods and Bads of the Endocannabinoid System as a Therapeutic Target: Lessons Learned after 30 Years.. Pharmacol. Rev, 2023. [DOI | PubMed]
- SA Herrlinger, BY Rao, MEC Paredes, AL Tuttman, H Arain, E Varol, JA Gogos, A Losonczy. Disorganized Inhibitory Dynamics and Functional Connectivity in Hippocampal area CA1 of 22q11.2 Deletion Mutant Mice. Preprint at. bioRxiv., 2024. [DOI]
- M Kano, T Ohno-Shosaku, Y Hashimotodani, M Uchigashima, M Watanabe. Endocannabinoid-mediated control of synaptic transmission.. Physiol. Rev, 2009. [DOI | PubMed]
- I Katona, TF Freund. Endocannabinoid signaling as a synaptic circuit breaker in neurological disease.. Nat. Med, 2008. [DOI | PubMed]
- I Katona, B Sperlágh, A Sík, A Käfalvi, ES Vizi, K Mackie, TF Freund. Presynaptically located CB1 cannabinoid receptors regulate GABA release from axon terminals of specific hippocampal interneurons.. J. Neurosci, 1999. [DOI | PubMed]
- I Katona, GM Urbán, M Wallace, C Ledent, KM Jung, D Piomelli, K Mackie, TF Freund. Molecular composition of the endocannabinoid system at glutamatergic synapses.. J. Neurosci, 2006. [DOI | PubMed]
- SH Lee, C Földy, I Soltesz. Distinct endocannabinoid control of GABA release at perisomatic and dendritic synapses in the hippocampus.. J. Neurosci, 2010. [DOI | PubMed]
- WA Devane, L Hanus, A Breuer, RG Pertwee, LA Stevenson, G Griffin, D Gibson, A Mandelbaum, A Etinger, R Mechoulam. Isolation and structure of a brain constituent that binds to the cannabinoid receptor.. Science, 1992. [DOI | PubMed]
- R Mechoulam, S Ben-Shabat, L Hanus, M Ligumsky, NE Kaminski, AR Schatz, A Gopher, S Almog, BR Martin, DR Compton. Identification of an endogenous 2-monoglyceride, present in canine gut, that binds to cannabinoid receptors.. Biochem. Pharmacol, 1995. [DOI | PubMed]
- T Sugiura, S Kondo, A Sukagawa, S Nakane, A Shinoda, K Itoh, A Yamashita, K Waku. 2-Arachidonoylglycerol: a possible endogenous cannabinoid receptor ligand in brain.. Biochem. Biophys. Res. Commun, 1995. [DOI | PubMed]
- N Stella, P Schweitzer, D Piomelli. A second endogenous cannabinoid that modulates long-term potentiation.. Nature, 1997. [DOI | PubMed]
- Y Hashimotodani, T Ohno-Shosaku, H Tsubokawa, H Ogata, K Emoto, T Maejima, K Araishi, HS Shin, M Kano. Phospholipase Cbeta serves as a coincidence detector through its Ca2+ dependency for triggering retrograde endocannabinoid signal.. Neuron, 2005. [DOI | PubMed]
- A Tanimura, M Yamazaki, Y Hashimotodani, M Uchigashima, S Kawata, M Abe, Y Kita, K Hashimoto, T Shimizu, M Watanabe. The endocannabinoid 2-arachidonoylglycerol produced by diacylglycerol lipase alpha mediates retrograde suppression of synaptic transmission.. Neuron, 2010. [DOI | PubMed]
- Y Gao, DV Vasilyev, MB Goncalves, FV Howell, C Hobbs, M Reisenberg, R Shen, MY Zhang, BW Strassle, P Lu. Loss of retrograde endocannabinoid signaling and reduced adult neurogenesis in diacylglycerol lipase knock-out mice.. J. Neurosci, 2010. [DOI | PubMed]
- J Suárez, O Ortíz, N Puente, FJ Bermúdez-Silva, E Blanco, P Fernández-Llebrez, P Grandes, FR de Fonseca, R Moratalla. Distribution of diacylglycerol lipase alpha, an endocannabinoid synthesizing enzyme, in the rat forebrain.. Neuroscience, 2011. [DOI | PubMed]
- T Yoshida, M Fukaya, M Uchigashima, E Miura, H Kamiya, M Kano, M Watanabe. Localization of diacylglycerol lipase-alpha around postsynaptic spine suggests close proximity between production site of an endocannabinoid, 2-arachidonoyl-glycerol, and presynaptic cannabinoid CB1 receptor.. J. Neurosci, 2006. [DOI | PubMed]
- Y Okamoto, J Morishita, K Tsuboi, T Tonai, N Ueda. Molecular characterization of a phospholipase D generating anandamide and its congeners.. J. Biol. Chem, 2004. [DOI | PubMed]
- M Egertová, BF Cravatt, MR Elphick. Comparative analysis of fatty acid amide hydrolase and cb1 cannabinoid receptor expression in the mouse brain: evidence of a widespread role for fatty acid amide hydrolase in regulation of endocannabinoid signaling.. Neuroscience, 2003. [DOI | PubMed]
- PJ Little, DR Compton, MR Johnson, LS Melvin, BR Martin. Pharmacology and stereoselectivity of structurally novel cannabinoids in mice.. J. Pharmacol. Exp. Ther, 1988. [DOI | PubMed]
- RI Wilson, RA Nicoll. Endogenous cannabinoids mediate retrograde signalling at hippocampal synapses.. Nature, 2001. [DOI | PubMed]
- T Ohno-Shosaku, T Maejima, M Kano. Endogenous cannabinoids mediate retrograde signals from depolarized postsynaptic neurons to presynaptic terminals.. Neuron, 2001. [DOI | PubMed]
- AC Kreitzer, WG Regehr. Retrograde inhibition of presynaptic calcium influx by endogenous cannabinoids at excitatory synapses onto Purkinje cells.. Neuron, 2001. [DOI | PubMed]
- TA Pitler, BE Alger. Depolarization-induced suppression of GABAergic inhibition in rat hippocampal pyramidal cells: G protein involvement in a presynaptic mechanism.. Neuron, 1994. [DOI | PubMed]
- AF Hoffman, CR Lupica. Mechanisms of cannabinoid inhibition of GABA(A) synaptic transmission in the hippocampus.. J. Neurosci, 2000. [DOI | PubMed]
- GG Szabó, N Lenkey, N Holderith, T Andrási, Z Nusser, N Hájos. Presynaptic calcium channel inhibition underlies CB1 cannabinoid receptor-mediated suppression of GABA release.. J. Neurosci, 2014. [DOI | PubMed]
- A Losonczy, AA Biró, Z Nusser. Persistently active cannabinoid receptors mute a subpopulation of hippocampal interneurons.. Proc. Natl. Acad. Sci. USA, 2004. [DOI | PubMed]
- A Neu, C Földy, I Soltesz. Postsynaptic origin of CB1-dependent tonic inhibition of GABA release at cholecystokinin-positive basket cell to pyramidal cell synapses in the CA1 region of the rat hippocampus.. J. Physiol, 2007. [DOI | PubMed]
- T Maejima, K Hashimoto, T Yoshida, A Aiba, M Kano. Presynaptic inhibition caused by retrograde signal from metabotropic glutamate to cannabinoid receptors.. Neuron, 2001. [DOI | PubMed]
- T Maejima, S Oka, Y Hashimotodani, T Ohno-Shosaku, A Aiba, D Wu, K Waku, T Sugiura, M Kano. Synaptically driven endocannabinoid release requires Ca2+-assisted metabotropic glutamate receptor subtype 1 to phospholipase Cbeta4 signaling cascade in the cerebellum.. J. Neurosci, 2005. [DOI | PubMed]
- SP Brown, SD Brenowitz, WG Regehr. Brief presynaptic bursts evoke synapse-specific retrograde inhibition mediated by endogenous cannabinoids.. Nat. Neurosci, 2003. [DOI | PubMed]
- E Albarran, Y Sun, Y Liu, K Raju, A Dong, Y Li, S Wang, TC Südhof, JB Ding. Postsynaptic synucleins mediate endocannabinoid signaling.. Nat. Neurosci, 2023. [DOI | PubMed]
- VM Straub, B Barti, ST Tandar, AF Stevens, N van Egmond, T van der Wel, N Zhu, J Rüegger, C van der Horst, LH Heitman. The endocannabinoid 2-arachidonoylglycerol is released and transported on demand via extracellular microvesicles.. Proc. Natl. Acad. Sci. USA, 2025. [DOI]
- R Tremblay, S Lee, B Rudy. GABAergic Interneurons in the Neocortex: From Cellular Properties to Circuits.. Neuron, 2016. [DOI | PubMed]
- N Hájos. Interneuron Types and Their Circuits in the Basolateral Amygdala.. Front. Neural Circuits, 2021. [DOI | PubMed]
- E Sigel, R Baur, I Rácz, J Marazzi, TG Smart, A Zimmer, J Gertsch. The major central endocannabinoid directly acts at GABA(A) receptors.. Proc. Natl. Acad. Sci. USA, 2011. [DOI | PubMed]
- T Golovko, R Min, N Lozovaya, C Falconer, N Yatsenko, T Tsintsadze, V Tsintsadze, C Ledent, RJ Harvey, D Belelli. Control of Inhibition by the Direct Action of Cannabinoids on GABAA Receptors.. Cereb. Cortex, 2015. [DOI | PubMed]
- M Vallée, S Vitiello, L Bellocchio, E Hébert-Chatelain, S Monlezun, E Martin-Garcia, F Kasanetz, GL Baillie, F Panin, A Cathala. Pregnenolone can protect the brain from cannabis intoxication.. Science, 2014. [DOI | PubMed]
- M Haney, M Vallée, S Fabre, S Collins Reed, M Zanese, G Campistron, CA Arout, RW Foltin, ZD Cooper, T Kearney-Ramos. Signaling-specific inhibition of the CB1 receptor for cannabis use disorder: phase 1 and phase 2a randomized trials.. Nat. Med, 2023. [DOI | PubMed]
- AA Zoerner, FM Gutzki, S Batkai, M May, C Rakers, S Engeli, J Jordan, D Tsikas. Quantification of endocannabinoids in biological systems by chromatography and mass spectrometry: a comprehensive review from an analytical and biological perspective.. Biochim. Biophys. Acta, 2011. [DOI | PubMed]
- S Guggenhuber, H Romo-Parra, L Bindila, J Leschik, E Lomazzo, F Remmers, T Zimmermann, R Lerner, M Klugmann, HC Pape, B Lutz. Impaired 2-AG Signaling in Hippocampal Glutamatergic Neurons: Aggravation of Anxiety-Like Behavior and Unaltered Seizure Susceptibility.. Int. J. Neuropsychopharmacol, 2015. [DOI | PubMed]
- MJ Wallace, RE Blair, KW Falenski, BR Martin, RJ DeLorenzo. The endogenous cannabinoid system regulates seizure frequency and duration in a model of temporal lobe epilepsy.. J. Pharmacol. Exp. Ther, 2003. [DOI | PubMed]
- G Marsicano, S Goodenough, K Monory, H Hermann, M Eder, A Cannich, SC Azad, MG Cascio, SO Gutiérrez, M van der Stelt. CB1 cannabinoid receptors and on-demand defense against excitotoxicity.. Science, 2003. [DOI | PubMed]
- R Lerner, J Post, S Loch, B Lutz, L Bindila. Targeting brain and peripheral plasticity of the lipidome in acute kainic acid-induced epileptic seizures in mice via quantitative mass spectrometry.. Biochim. Biophys. Acta Mol. Cell Biol. Lipids, 2017. [DOI | PubMed]
- A Dong, K He, B Dudok, JS Farrell, W Guan, DJ Liput, HL Puhl, R Cai, H Wang, J Duan. A fluorescent sensor for spatio-temporally resolved imaging of endocannabinoid dynamics in vivo.. Nat. Biotechnol, 2022. [DOI | PubMed]
- S Singh, D Sarroza, A English, M McGrory, A Dong, L Zweifel, BB Land, Y Li, MR Bruchas, N Stella. Pharmacological Characterization of the Endocannabinoid Sensor GRABeCB2.0.. Cannabis Cannabinoid Res., 2024. [DOI | PubMed]
- DJ Liput, HL Puhl, A Dong, K He, Y Li, DM Lovinger. 2-Arachidonoylglycerol mobilization following brief synaptic stimulation in the dorsal lateral striatum requires glutamatergic and cholinergic neurotransmission.. Neuropharmacology, 2022. [DOI | PubMed]
- GL Gerdeman, J Ronesi, DM Lovinger. Postsynaptic endocannabinoid release is critical to long-term depression in the striatum.. Nat. Neurosci, 2002. [DOI | PubMed]
- AC Kreitzer, RC Malenka. Endocannabinoid-mediated rescue of striatal LTD and motor deficits in Parkinson’s disease models.. Nature, 2007. [DOI | PubMed]
- V Kondev, M Najeed, F Yasmin, A Morgan, N Loomba, K Johnson, DN Adank, A Dong, E Delpire, Y Li. Endocannabinoid release at ventral hippocampal-amygdala synapses regulates stress-induced behavioral adaptation.. Cell Rep., 2023. [DOI | PubMed]
- C Armstrong, RJ Morgan, I Soltesz. Pursuing paradoxical proconvulsant prophylaxis for epileptogenesis.. Epilepsia, 2009. [DOI | PubMed]
- ME Hofmann, CJ Frazier. Marijuana, endocannabinoids, and epilepsy: potential and challenges for improved therapeutic intervention.. Exp. Neurol, 2013. [DOI | PubMed]
- KJ Iremonger, JI Wamsteeker Cusulin, JS Bains. Changing the tune: plasticity and adaptation of retrograde signals.. Trends Neurosci., 2013. [DOI | PubMed]
- S Ruehle, AA Rey, F Remmers, B Lutz. The endocannabinoid system in anxiety, fear memory and habituation.. J. Psychopharmacol, 2012. [DOI | PubMed]
- B Lutz, G Marsicano, R Maldonado, CJ Hillard. The endocannabinoid system in guarding against fear, anxiety and stress.. Nat. Rev. Neurosci, 2015. [DOI | PubMed]
- F Bellia, A Girella, E Annunzi, B Benatti, M Vismara, A Priori, F Festucci, F Fanti, D Compagnone, W Adriani. Selective alterations of endocannabinoid system genes expression in obsessive compulsive disorder.. Transl. Psychiatry, 2024. [DOI | PubMed]
- JS Farrell, R Colangeli, B Dudok, MD Wolff, SL Nguyen, J Jackson, CT Dickson, I Soltesz, GC Teskey. In vivo assessment of mechanisms underlying the neurovascular basis of postictal amnesia.. Sci. Rep, 2020. [DOI | PubMed]
- I Tamim, DY Chung, AL de Morais, ICM Loonen, T Qin, A Misra, F Schlunk, M Endres, SJ Schiff, C Ayata. Spreading depression as an innate antiseizure mechanism.. Nat. Commun, 2021. [DOI | PubMed]
- T Klausberger, LF Marton, J O’Neill, JHJ Huck, Y Dalezios, P Fuentealba, WY Suen, E Papp, T Kaneko, M Watanabe. Complementary roles of cholecystokinin- and parvalbumin-expressing GABAergic neurons in hippocampal network oscillations.. J. Neurosci, 2005. [DOI | PubMed]
- B Lasztóczi, JJ Tukker, P Somogyi, T Klausberger. Terminal field and firing selectivity of cholecystokinin-expressing interneurons in the hippocampal CA3 area.. J. Neurosci, 2011. [DOI | PubMed]
- H Taniguchi, M He, P Wu, S Kim, R Paik, K Sugino, D Kvitsiani, Y Fu, J Lu, Y Lin. A Resource of Cre Driver Lines for Genetic Targeting of GABAergic Neurons in Cerebral Cortex.. Neuron, 2011. [DOI | PubMed]
- T Klausberger, P Somogyi. Neuronal diversity and temporal dynamics: the unity of hippocampal circuit operations.. Science, 2008. [DOI | PubMed]
- R Machold, S Dellal, M Valero, H Zurita, I Kruglikov, JH Meng, JL Hanson, Y Hashikawa, B Schuman, G Buzsáki, B Rudy. Id2 GABAergic interneurons comprise a neglected fourth major group of cortical inhibitory cells.. eLife, 2023. [DOI | PubMed]
- B Dudok, PM Klein, E Hwaun, BR Lee, Z Yao, O Fong, JC Bowler, S Terada, FT Sparks, GG Szabo. Alternating sources of perisomatic inhibition during behavior.. Neuron, 2021. [DOI | PubMed]
- DKR Guerrero, K Balueva, U Barayeu, P Baracskay, I Gridchyn, M Nardin, CN Roth, PW Csicsvari. Hippocampal cholecystokinin-expressing interneurons regulate temporal coding and contextual learning. 2061.e2010.. Neuron, 2024. [DOI | PubMed]
- L Rovira-Esteban, O Gunduz-Cinar, O Bukalo, A Limoges, E Brockway, K Müller, L Fenno, YS Kim, C Ramakrishnan, T Andrási. Excitation of Diverse Classes of Cholecystokinin Interneurons in the Basal Amygdala Facilitates Fear Extinction. ENEURO.0220–19.2019.. eNeuro, 2019. [DOI]
- B Schuman, RP Machold, Y Hashikawa, J Fuzik, GJ Fishell, B Rudy. Four Unique Interneuron Populations Reside in Neocortical Layer 1.. J. Neurosci, 2019. [DOI | PubMed]
- T Geiller, B Vancura, S Terada, E Troullinou, S Chavlis, G Tsagkatakis, P Tsakalides, K Ócsai, P Poirazi, BJ Rózsa, A Losonczy. Large-Scale 3D Two-Photon Imaging of Molecularly Identified CA1 Interneuron Dynamics in Behaving Mice.. Neuron, 2020. [DOI | PubMed]
- B Vancura, T Geiller, A Grosmark, V Zhao, A Losonczy. Inhibitory control of sharp-wave ripple duration during learning in hippocampal recurrent networks.. Nat. Neurosci, 2023. [DOI | PubMed]
- J Somogyi, A Baude, Y Omori, H Shimizu, S El Mestikawy, M Fukaya, R Shigemoto, M Watanabe, P Somogyi. GABAergic basket cells expressing cholecystokinin contain vesicular glutamate transporter type 3 (VGLUT3) in their synaptic terminals in hippocampus and isocortex of the rat.. Eur. J. Neurosci, 2004. [DOI | PubMed]
- KA Pelkey, D Calvigioni, C Fang, G Vargish, T Ekins, K Auville, JC Wester, M Lai, C Mackenzie-Gray Scott, X Yuan. Paradoxical network excitation by glutamate release from VGluT3+ GABAergic interneurons.. eLife, 2020. [DOI | PubMed]
- NW Gouwens, SA Sorensen, F Baftizadeh, A Budzillo, BR Lee, T Jarsky, L Alfiler, K Baker, E Barkan, K Berry. Integrated Morphoelectric and Transcriptomic Classification of Cortical GABAergic Cells.. Cell, 2020. [DOI | PubMed]
- B Tasic, V Menon, TN Nguyen, TK Kim, T Jarsky, Z Yao, B Levi, LT Gray, SA Sorensen, T Dolbeare. Adult mouse cortical cell taxonomy revealed by single cell transcriptomics.. Nat. Neurosci, 2016. [DOI | PubMed]
- Z Yao, H Liu, F Xie, S Fischer, RS Adkins, AI Aldridge, SA Ament, A Bartlett, MM Behrens, K Van den Berge. A transcriptomic and epigenomic cell atlas of the mouse primary motor cortex.. Nature, 2021. [DOI | PubMed]
- C Armstrong, I Soltesz. Basket cell dichotomy in microcircuit function.. J. Physiol, 2012. [DOI | PubMed]
- V Miczán, K Kelemen, JR Glavinics, ZI László, B Barti, K Kenesei, M Kisfali, I Katona. NECAB1 and NECAB2 are Prevalent Calcium-Binding Proteins of CB1/CCK-Positive GABAergic Interneurons.. Cereb. Cortex, 2021. [DOI | PubMed]
- TF Freund, I Katona. Perisomatic inhibition.. Neuron, 2007. [DOI | PubMed]
- Z Yao, CTJ van Velthoven, M Kunst, M Zhang, D McMillen, C Lee, W Jung, J Goldy, A Abdelhak, M Aitken. A high-resolution transcriptomic and spatial atlas of cell types in the whole mouse brain.. Nature, 2023. [DOI | PubMed]
- G Katona, G Szalay, P Maák, A Kaszás, M Veress, D Hillier, B Chiovini, ES Vizi, B Roska, B Rózsa. Fast two-photon in vivo imaging with three-dimensional random-access scanning in large tissue volumes.. Nat. Methods, 2012. [DOI | PubMed]
- GG Szabo, JS Farrell, B Dudok, WH Hou, AL Ortiz, C Varga, P Moolchand, CI Gulsever, T Gschwind, J Dimidschstein. Ripple-selective GABAergic projection cells in the hippocampus.. Neuron, 2022. [DOI | PubMed]
- TF Freund. Interneuron Diversity series: Rhythm and mood in perisomatic inhibition.. Trends Neurosci., 2003. [DOI | PubMed]
- C Varga, P Golshani, I Soltesz. Frequency-invariant temporal ordering of interneuronal discharges during hippocampal oscillations in awake mice.. Proc. Natl. Acad. Sci. USA, 2012. [DOI | PubMed]
- C Varga, M Oijala, J Lish, GG Szabo, M Bezaire, I Marchionni, P Golshani, I Soltesz. Functional fission of parvalbumin interneuron classes during fast network events.. eLife, 2014. [DOI | PubMed]
- G Buzsáki. Hippocampal sharp wave-ripple: A cognitive biomarker for episodic memory and planning.. Hippocampus, 2015. [DOI | PubMed]
- F Mátyás, TF Freund, AI Gulyás. Convergence of excitatory and inhibitory inputs onto CCK-containing basket cells in the CA1 area of the rat hippocampus.. Eur. J. Neurosci, 2004. [DOI | PubMed]
- SS Deshmukh, JJ Knierim. Representation of non-spatial and spatial information in the lateral entorhinal cortex.. Front. Behav. Neurosci, 2011. [DOI | PubMed]
- JC Bowler, A Losonczy. Direct cortical inputs to hippocampal area CA1 transmit complementary signals for goal-directed navigation.. Neuron, 2023. [DOI | PubMed]
- J Basu, KV Srinivas, SK Cheung, H Taniguchi, ZJ Huang, SA Siegelbaum. A cortico-hippocampal learning rule shapes inhibitory microcircuit activity to enhance hippocampal information flow.. Neuron, 2013. [DOI | PubMed]
- J Basu, JD Zaremba, SK Cheung, FL Hitti, BV Zemelman, A Losonczy, SA Siegelbaum. Gating of hippocampal activity, plasticity, and memory by entorhinal cortex long-range inhibition.. Science, 2016. [DOI | PubMed]
- OM Bilash, S Chavlis, CD Johnson, P Poirazi, J Basu. Lateral entorhinal cortex inputs modulate hippocampal dendritic excitability by recruiting a local disinhibitory microcircuit.. Cell Rep., 2023. [DOI | PubMed]
- K Kay, M Sosa, JE Chung, MP Karlsson, MC Larkin, LM Frank. A hippocampal network for spatial coding during immobility and sleep.. Nature, 2016. [DOI | PubMed]
- LA Karaba, HL Robinson, RE Harvey, W Chen, A Fernandez-Ruiz, A Oliva. A hippocampal circuit mechanism to balance memory reactivation during sleep.. Science, 2024. [DOI | PubMed]
- S Bugeon, J Duffield, M Dipoppa, A Ritoux, I Prankerd, D Nicoloutsopoulos, D Orme, M Shinn, H Peng, H Forrest. A transcriptomic axis predicts state modulation of cortical interneurons.. Nature, 2022. [DOI | PubMed]
- M Valero, TJ Viney, R Machold, S Mederos, I Zutshi, B Schuman, Y Senzai, B Rudy, G Buzsáki. Sleep down state-active ID2/Nkx2.1 interneurons in the neocortex.. Nat. Neurosci, 2021. [DOI | PubMed]
- SJ Smith, M.v. von Zastrow. A Molecular Landscape of Mouse Hippocampal Neuromodulation.. FrontFront. Neural Circuits, 2022. [DOI]
- Z Wu, D Lin, Y Li. Pushing the frontiers: tools for monitoring neurotransmitters and neuromodulators.. Nat. Rev. Neurosci, 2022. [DOI | PubMed]
- J O’Keefe, J Dostrovsky. The hippocampus as a spatial map. Preliminary evidence from unit activity in the freely-moving rat.. Brain Res., 1971. [DOI | PubMed]
- D Robbe, G Buzsáki. Alteration of theta timescale dynamics of hippocampal place cells by a cannabinoid is associated with memory impairment.. J. Neurosci, 2009. [DOI | PubMed]
- D Robbe, SM Montgomery, A Thome, PE Rueda-Orozco, BL McNaughton, G Buzsaki. Cannabinoids reveal importance of spike timing coordination in hippocampal function.. Nat. Neurosci, 2006. [DOI | PubMed]
- F Dubruc, D Dupret, O Caillard. Self-tuning of inhibition by endocannabinoids shapes spike-time precision in CA1 pyramidal neurons.. J. Neurophysiol, 2013. [DOI | PubMed]
- KC Bittner, C Grienberger, SP Vaidya, AD Milstein, JJ Macklin, J Suh, S Tonegawa, JC Magee. Conjunctive input processing drives feature selectivity in hippocampal CA1 neurons.. Nat. Neurosci, 2015. [DOI | PubMed]
- T Geiller, S Sadeh, SV Rolotti, H Blockus, B Vancura, A Negrean, AJ Murray, B Rózsa, F Polleux, C Clopath, A Losonczy. Local circuit amplification of spatial selectivity in the hippocampus.. Nature, 2022. [DOI | PubMed]
- M Valero, A Navas-Olive, LM de la Prida, G Buzsáki. Inhibitory conductance controls place field dynamics in the hippocampus.. Cell Rep., 2022. [DOI | PubMed]
- SV Rolotti, MS Ahmed, M Szoboszlay, T Geiller, A Negrean, H Blockus, KC Gonzalez, FT Sparks, AS Solis Canales, AL Tuttman. Local feedback inhibition tightly controls rapid formation of hippocampal place fields.. Neuron, 2022. [DOI | PubMed]
- B Dudok, LZ Fan, JS Farrell, S Malhotra, J Homidan, DK Kim, C Wenardy, C Ramakrishnan, Y Li, K Deisseroth, I Soltesz. Retrograde endocannabinoid signaling at inhibitory synapses in vivo.. Science, 2024. [DOI | PubMed]
- LZ Fan, DK Kim, JH Jennings, H Tian, PY Wang, C Ramakrishnan, S Randles, Y Sun, E Thadhani, YS Kim. All-optical physiology resolves a synaptic basis for behavioral timescale plasticity.. Cell, 2023. [DOI | PubMed]
- KC Gonzalez, A Negrean, Z Liao, S Terada, G Zhang, S Lee, K Ócsai, BJ Rózsa, MZ Lin, F Polleux, A Losonczy. Synaptic basis of feature selectivity in hippocampal neurons.. Nature, 2025. [DOI | PubMed]
- J Epsztein, M Brecht, AK Lee. Intracellular determinants of hippocampal CA1 place and silent cell activity in a novel environment.. Neuron, 2011. [DOI | PubMed]
- A Fernández-Ruiz, A Oliva, E Fermino de Oliveira, F Rocha-Almeida, D Tingley, G Buzsáki. Long-duration hippocampal sharp wave ripples improve memory.. Science, 2019. [DOI | PubMed]
- J Csicsvari, H Hirase, A Mamiya, G Buzsáki. Ensemble patterns of hippocampal CA3-CA1 neurons during sharp wave-associated population events.. Neuron, 2000. [DOI | PubMed]
- K Diba, G Buzsáki. Forward and reverse hippocampal place-cell sequences during ripples.. Nat. Neurosci, 2007. [DOI | PubMed]
- T Nakashiba, DL Buhl, TJ McHugh, S Tonegawa. Hippocampal CA3 output is crucial for ripple-associated reactivation and consolidation of memory.. Neuron, 2009. [DOI | PubMed]
- T Nakashiba, JZ Young, TJ McHugh, DL Buhl, S Tonegawa. Transgenic inhibition of synaptic transmission reveals role of CA3 output in hippocampal learning.. Science, 2008. [DOI | PubMed]
- S Terada, T Geiller, Z Liao, J O’Hare, B Vancura, A Losonczy. Adaptive stimulus selection for consolidation in the hippocampus.. Nature, 2022. [DOI | PubMed]
- T Forro, T Klausberger. Differential behavior-related activity of distinct hippocampal interneuron types during odor-associated spatial navigation.. Neuron, 2023. [DOI | PubMed]
- Z Liao, S Terada, IG Raikov, D Hadjiabadi, M Szoboszlay, I Soltesz, A Losonczy. Inhibitory plasticity supports replay generalization in the hippocampus.. Nat. Neurosci, 2024. [DOI | PubMed]
- S Tonegawa, M Pignatelli, DS Roy, TJ Ryan. Memory engram storage and retrieval.. Curr. Opin. Neurobiol, 2015. [DOI | PubMed]
- X Sun, MJ Bernstein, M Meng, S Rao, AT Sørensen, L Yao, X Zhang, PO Anikeeva, Y Lin. Functionally Distinct Neuronal Ensembles within the Memory Engram.. Cell, 2020. [DOI | PubMed]
- AL Hartzell, KM Martyniuk, GS Brigidi, DA Heinz, NA Djaja, A Payne, BL Bloodgood. NPAS4 recruits CCK basket cell synapses and enhances cannabinoid-sensitive inhibition in the mouse hippocampus.. eLife, 2018. [DOI | PubMed]
- DVC Brito, J Kupke, R Sokolov, S Cambridge, M Both, CP Bengtson, A Rozov, AMM Oliveira. Biphasic Npas4 expression promotes inhibitory plasticity and suppression of fear memory consolidation in mice.. Mol. Psychiatry, 2024. [DOI | PubMed]
- EL Yap, NL Pettit, CP Davis, MA Nagy, DA Harmin, E Golden, O Dagliyan, C Lin, S Rudolph, N Sharma. Bidirectional perisomatic inhibitory plasticity of a Fos neuronal network.. Nature, 2021. [DOI | PubMed]
- DF Tomé, Y Zhang, T Aida, O Mosto, Y Lu, M Chen, S Sadeh, DS Roy, C Clopath. Dynamic and selective engrams emerge with memory consolidation.. Nat. Neurosci, 2024. [DOI | PubMed]
- M Ye, SK Monroe, SM Gay, ML Armstrong, DE Youngstrom, FL Urbina, SL Gupton, N Reisdorph, GH Diering. Coordinated Regulation of CB1 Cannabinoid Receptors and Anandamide Metabolism Stabilizes Network Activity during Homeostatic Downscaling. ENEURO.0276–22.2022.. eNeuro, 2022. [DOI]
- R Colangeli, M Morena, QJ Pittman, MN Hill, GC Teskey. Anandamide Signaling Augmentation Rescues Amygdala Synaptic Function and Comorbid Emotional Alterations in a Model of Epilepsy.. J. Neurosci, 2020. [DOI | PubMed]
- GN Petrie, G Balsevich, T Füzesi, RJ Aukema, WPF Driever, M van der Stelt, JS Bains, MN Hill. Disruption of tonic endocannabinoid signalling triggers cellular, behavioural and neuroendocrine responses consistent with a stress response.. Br. J. Pharmacol, 2023. [DOI | PubMed]
- SH Lee, M Ledri, B Tóth, I Marchionni, CM Henstridge, B Dudok, K Kenesei, L Barna, SI Szabó, T Renkecz. Multiple Forms of Endocannabinoid and Endovanilloid Signaling Regulate the Tonic Control of GABA Release.. J. Neurosci, 2015. [DOI | PubMed]
- P Rivera, S Arrabal, A Vargas, E Blanco, A Serrano, FJ Pavón, F Rodríguez de Fonseca, J Suárez. Localization of peroxisome proliferator-activated receptor alpha (PPARα) and N-acyl phosphatidylethanolamine phospholipase D (NAPE-PLD) in cells expressing the Ca(2+)-binding proteins calbindin, calretinin, and parvalbumin in the adult rat hippocampus.. Front. Neuroanat, 2014. [DOI | PubMed]
- R Nyilas, B Dudok, GM Urbán, K Mackie, M Watanabe, BF Cravatt, TF Freund, I Katona. Enzymatic machinery for endocannabinoid biosynthesis associated with calcium stores in glutamatergic axon terminals.. J. Neurosci, 2008. [DOI | PubMed]
- C Földy, SY Lee, J Szabadics, A Neu, I Soltesz. Cell type-specific gating of perisomatic inhibition by cholecystokinin.. Nat. Neurosci, 2007. [DOI | PubMed]
- SY Lee, C Földy, J Szabadics, I Soltesz. Cell-type-specific CCK2 receptor signaling underlies the cholecystokinin-mediated selective excitation of hippocampal parvalbumin-positive fast-spiking basket cells.. J. Neurosci, 2011. [DOI | PubMed]
- H Wang, T Qian, Y Zhao, Y Zhuo, C Wu, T Osakada, P Chen, Z Chen, H Ren, Y Yan. A tool kit of highly selective and sensitive genetically encoded neuropeptide sensors.. Science, 2023. [DOI | PubMed]
- I Del Pino, JR Brotons-Mas, A Marques-Smith, A Marighetto, A Frick, O Marín, B Rico. Abnormal wiring of CCK+ basket cells disrupts spatial information coding.. Nat. Neurosci, 2017. [DOI | PubMed]
- Y Omiya, M Uchigashima, K Konno, M Yamasaki, T Miyazaki, T Yoshida, I Kusumi, M Watanabe. VGluT3-expressing CCK-positive basket cells construct invaginating synapses enriched with endocannabinoid signaling proteins in particular cortical and cortex-like amygdaloid regions of mouse brains.. J. Neurosci, 2015. [DOI | PubMed]
- J Neddens, A Buonanno. Selective populations of hippocampal interneurons express ErbB4 and their number and distribution is altered in ErbB4 knockout mice.. Hippocampus, 2010. [DOI | PubMed]
- P Fazzari, AV Paternain, M Valiente, R Pla, R Luján, K Lloyd, J Lerma, O Marín, B Rico. Control of cortical GABA circuitry development by Nrg1 and ErbB4 signalling.. Nature, 2010. [DOI | PubMed]
- I Del Pino, C García-Frigola, N Dehorter, JR Brotons-Mas, E Alvarez-Salvado, M Martínez de Lagrán, G Ciceri, MV Gabaldón, D Moratal, M Dierssen. Erbb4 deletion from fast-spiking interneurons causes schizophrenia-like phenotypes.. Neuron, 2013. [DOI | PubMed]
- AK Ting, Y Chen, L Wen, DM Yin, C Shen, Y Tao, X Liu, WC Xiong, L Mei. Neuregulin 1 promotes excitatory synapse development and function in GABAergic interneurons.. J. Neurosci, 2011. [DOI | PubMed]
- JM Yang, J Zhang, XJ Chen, HY Geng, M Ye, NC Spitzer, JH Luo, SM Duan, XM Li. Development of GABA circuitry of fast-spiking basket interneurons in the medial prefrontal cortex of erbb4-mutant mice.. J. Neurosci, 2013. [DOI | PubMed]
- ME Cahill, C Remmers, KA Jones, Z Xie, RA Sweet, P Penzes. Neuregulin1 signaling promotes dendritic spine growth through kalirin.. J. Neurochem, 2013. [DOI | PubMed]
- B Luo, Z Liu, D Lin, W Chen, D Ren, Z Yu, M Xiong, C Zhao, E Fei, B Li. ErbB4 promotes inhibitory synapse formation by cell adhesion, independent of its kinase activity.. Transl. Psychiatry, 2021. [DOI | PubMed]
- C Fasano, J Rocchetti, K Pietrajtis, JF Zander, F Manseau, DY Sakae, M Marcus-Sells, L Ramet, LJ Morel, D Carrel. Regulation of the Hippocampal Network by VGLUT3-Positive CCK-GABAergic Basket Cells.. Front. Cell. Neurosci, 2017. [DOI | PubMed]
- MJ Stensrud, FA Chaudhry, TB Leergaard, JG Bjaalie, V Gundersen. Vesicular glutamate transporter-3 in the rodent brain: vesicular colocalization with vesicular gamma-aminobutyric acid transporter.. J. Comp. Neurol, 2013. [DOI | PubMed]
- MEJ Sheffield, DA Dombeck. Calcium transient prevalence across the dendritic arbour predicts place field properties.. Nature, 2015. [DOI | PubMed]
- JK O’Hare, KC Gonzalez, SA Herrlinger, Y Hirabayashi, VL Hewitt, H Blockus, M Szoboszlay, SV Rolotti, TC Geiller, A Negrean. Compartment-specific tuning of dendritic feature selectivity by intracellular Ca2+ release.. Science, 2022. [DOI | PubMed]
- SA Booker, I Vida. Morphological diversity and connectivity of hippocampal interneurons.. Cell Tissue Res., 2018. [DOI | PubMed]
- T Klausberger. GABAergic interneurons targeting dendrites of pyramidal cells in the CA1 area of the hippocampus.. Eur. J. Neurosci, 2009. [DOI | PubMed]
- KG Baimbridge, JJ Miller. Immunohistochemical localization of calcium-binding protein in the cerebellum, hippocampal formation and olfactory bulb of the rat.. Brain Res., 1982. [DOI | PubMed]
- L Slomianka, I Amrein, I Knuesel, JC Sørensen, DP Wolfer. Hippocampal pyramidal cells: the reemergence of cortical lamination.. Brain Struct. Funct, 2011. [DOI | PubMed]
- I Soltesz, A Losonczy. CA1 pyramidal cell diversity enabling parallel information processing in the hippocampus.. Nat. Neurosci, 2018. [DOI | PubMed]
- SH Lee, I Marchionni, M Bezaire, C Varga, N Danielson, M Lovett-Barron, A Losonczy, I Soltesz. Parvalbumin-positive basket cells differentiate among hippocampal pyramidal cells.. Neuron, 2014. [DOI | PubMed]
- M Valero, E Cid, RG Averkin, J Aguilar, A Sanchez-Aguilera, TJ Viney, D Gomez-Dominguez, E Bellistri, LM de la Prida. Determinants of different deep and superficial CA1 pyramidal cell dynamics during sharp-wave ripples.. Nat. Neurosci, 2015. [DOI | PubMed]
- C Varga, SY Lee, I Soltesz. Target-selective GABAergic control of entorhinal cortex output.. Nat. Neurosci, 2010. [DOI | PubMed]
- P Berghuis, AM Rajnicek, YM Morozov, RA Ross, J Mulder, GM Urbán, K Monory, G Marsicano, M Matteoli, A Canty. Hardwiring the brain: endocannabinoids shape neuronal connectivity.. Science, 2007. [DOI | PubMed]
- T Harkany, K Mackie, P Doherty. Wiring and firing neuronal networks: endocannabinoids take center stage.. Curr. Opin. Neurobiol, 2008. [DOI | PubMed]
- T Zimmermann, M Maroso, A Beer, S Baddenhausen, S Ludewig, W Fan, C Vennin, S Loch, B Berninger, C Hofmann. Neural stem cell lineage-specific cannabinoid type-1 receptor regulates neurogenesis and plasticity in the adult mouse hippocampus.. Cereb. Cortex, 2018. [DOI | PubMed]
- C Bernard, M Milh, YM Morozov, Y Ben-Ari, TF Freund, H Gozlan. Altering cannabinoid signaling during development disrupts neuronal activity.. Proc. Natl. Acad. Sci. USA, 2005. [DOI | PubMed]
- E Leprince, RF Dard, S Mortet, C Filippi, M Giorgi-Kurz, R Bourboulou, PP Lenck-Santini, MA Picardo, M Bocchio, A Baude, R Cossart. Extrinsic control of the early postnatal CA1 hippocampal circuits.. Neuron, 2023. [DOI | PubMed]
- A Eraso-Pichot, S Pouvreau, A Olivera-Pinto, P Gomez-Sotres, U Skupio, G Marsicano. Endocannabinoid signaling in astrocytes.. Glia, 2023. [DOI | PubMed]
- S Marinelli, MC Marrone, M Di Domenico, S Marinelli. Endocannabinoid signaling in microglia.. Glia, 2023. [DOI | PubMed]
- W Sun, A Cornwell, J Li, S Peng, MJ Osorio, N Aalling, S Wang, A Benraiss, N Lou, SA Goldman, M Nedergaard. SOX9 Is an Astrocyte-Specific Nuclear Marker in the Adult Brain Outside the Neurogenic Regions.. J. Neurosci, 2017. [DOI | PubMed]
- A Verkhratsky, R Zorec, V Parpura. Stratification of astrocytes in healthy and diseased brain.. Brain Pathol., 2017. [DOI | PubMed]
- M Navarrete, A Araque. Endocannabinoids mediate neuron-astrocyte communication.. Neuron, 2008. [DOI | PubMed]
- M Navarrete, A Araque. Endocannabinoids potentiate synaptic transmission through stimulation of astrocytes.. Neuron, 2010. [DOI | PubMed]
- A Gutiérrez-Rodríguez, I Bonilla-Del Río, N Puente, SM Gómez-Urquijo, CJ Fontaine, J Egaña-Huguet, I Elezgarai, S Ruehle, B Lutz, LM Robin. Localization of the cannabinoid type-1 receptor in subcellular astrocyte compartments of mutant mouse hippocampus.. Glia, 2018. [DOI | PubMed]
- J Han, P Kesner, M Metna-Laurent, T Duan, L Xu, F Georges, M Koehl, DN Abrous, J Mendizabal-Zubiaga, P Grandes. Acute cannabinoids impair working memory through astroglial CB1 receptor modulation of hippocampal LTD.. Cell, 2012. [DOI | PubMed]
- A Covelo, A Araque. Neuronal activity determines distinct gliotransmitter release from a single astrocyte.. eLife, 2018. [DOI | PubMed]
- M Gómez-Gonzalo, M Navarrete, G Perea, A Covelo, M Martín-Fernández, R Shigemoto, R Luján, A Araque. Endocannabinoids Induce Lateral Long-Term Potentiation of Transmitter Release by Stimulation of Gliotransmission.. Cereb. Cortex, 2015. [DOI | PubMed]
- C Muller, P Morales, PH Reggio. Cannabinoid Ligands Targeting TRP Channels.. Front. Mol. Neurosci, 2018. [DOI | PubMed]
- T Boczek, L Zylinska. Receptor-Dependent and Independent Regulation of Voltage-Gated Ca(2+) Channels and Ca(2+)-Permeable Channels by Endocannabinoids in the Brain.. Int. J. Mol. Sci, 2021. [DOI | PubMed]
- L Cristino, T Bisogno, V Di Marzo. Cannabinoids and the expanded endocannabinoid system in neurological disorders.. Nat. Rev. Neurol, 2020. [DOI | PubMed]
- Y Chen, X Liu, CR Vickstrom, MJ Liu, L Zhao, A Viader, BF Cravatt, QS Liu. Neuronal and Astrocytic Monoacylglycerol Lipase Limit the Spread of Endocannabinoid Signaling in the Cerebellum. ENEURO.0048–16.2016.. eNeuro, 2016. [DOI]
- X Liu, Y Chen, CR Vickstrom, Y Li, A Viader, BF Cravatt, QS Liu. Coordinated regulation of endocannabinoid-mediated retrograde synaptic suppression in the cerebellum by neuronal and astrocytic monoacylglycerol lipase.. Sci. Rep, 2016. [DOI | PubMed]
- A Viader, JL Blankman, P Zhong, X Liu, JE Schlosburg, CM Joslyn, QS Liu, AJ Tomarchio, AH Lichtman, DE Selley. Metabolic Interplay between Astrocytes and Neurons Regulates Endocannabinoid Action.. Cell Rep., 2015. [DOI | PubMed]
- C Benito, JP Romero, RM Tolón, D Clemente, F Docagne, CJ Hillard, C Guaza, J Romero. Cannabinoid CB1 and CB2 receptors and fatty acid amide hydrolase are specific markers of plaque cell subtypes in human multiple sclerosis.. J. Neurosci, 2007. [DOI | PubMed]
- S Kallendrusch, C Hobusch, A Ehrlich, S Ziebell, N Ueda, G Geisslinger, M Koch, F Dehghani. Site-specific and time-dependent activation of the endocannabinoid system after transection of long-range projections.. PLoS One, 2012. [DOI | PubMed]
- F Molina-Holgado, A Lledó, C Guaza. Anandamide suppresses nitric oxide and TNF-alpha responses to Theiler’s virus or endotoxin in astrocytes.. NeuroReport, 1997. [DOI | PubMed]
- F Molina-Holgado, E Molina-Holgado, C Guaza. The endogenous cannabinoid anandamide potentiates interleukin-6 production by astrocytes infected with Theiler’s murine encephalomyelitis virus by a receptor-mediated pathway.. FEBS Lett., 1998. [DOI | PubMed]
- WS Sheng, S Hu, X Min, GA Cabral, JR Lokensgard, PK Peterson. Synthetic cannabinoid WIN55,212–2 inhibits generation of inflammatory mediators by IL-1beta-stimulated human astrocytes.. Glia, 2005. [DOI | PubMed]
- F Molina-Holgado, E Molina-Holgado, C Guaza, NJ Rothwell. Role of CB1 and CB2 receptors in the inhibitory effects of cannabinoids on lipopolysaccharide-induced nitric oxide release in astrocyte cultures.. J. Neurosci. Res, 2002. [DOI | PubMed]
- KA Dudek, SEJ Paton, LB Binder, A Collignon, L Dion-Albert, A Cadoret, M Lebel, O Lavoie, J Bouchard, FN Kaufmann. Astrocytic cannabinoid receptor 1 promotes resilience by dampening stress-induced blood-brain barrier alterations.. Nat. Neurosci, 2025. [DOI]
- GF Grabner, TO Eichmann, B Wagner, Y Gao, A Farzi, U Taschler, FPW Radner, M Schweiger, A Lass, P Holzer. Deletion of Monoglyceride Lipase in Astrocytes Attenuates Lipopolysaccharide-induced Neuroinflammation.. J. Biol. Chem, 2016. [DOI | PubMed]
- L Walter, A Franklin, A Witting, T Moller, N Stella. Astrocytes in culture produce anandamide and other acylethanolamides.. J. Biol. Chem, 2002. [DOI | PubMed]
- L Walter, T Dinh, N Stella. ATP induces a rapid and pronounced increase in 2-arachidonoylglycerol production by astrocytes, a response limited by monoacylglycerol lipase.. J. Neurosci, 2004. [DOI | PubMed]
- L Walter, N Stella. Endothelin-1 increases 2-arachidonoyl glycerol (2-AG) production in astrocytes.. Glia, 2003. [DOI | PubMed]
- A Covelo, A Eraso-Pichot, I Fernández-Moncada, R Serrat, G Marsicano. CB1R-dependent regulation of astrocyte physiology and astrocyte-neuron interactions.. Neuropharmacology, 2021. [DOI | PubMed]
- L Walter, A Franklin, A Witting, C Wade, Y Xie, G Kunos, K Mackie, N Stella. Nonpsychotropic cannabinoid receptors regulate microglial cell migration.. J. Neurosci, 2003. [DOI | PubMed]
- Á Moreno-García, A Bernal-Chico, T Colomer, A Rodríguez-Antigüedad, C Matute, S Mato. Gene Expression Analysis of Astrocyte and Microglia Endocannabinoid Signaling during Autoimmune Demyelination.. Biomolecules, 2020. [DOI | PubMed]
- QR Liu, A Canseco-Alba, Y Liang, H Ishiguro, ES Onaivi. Low Basal CB2R in Dopamine Neurons and Microglia Influences Cannabinoid Tetrad Effects.. Int. J. Mol. Sci, 2020. [DOI | PubMed]
- Y Li, J Kim. Distinct roles of neuronal and microglial CB2 cannabinoid receptors in the mouse hippocampus.. Neuroscience, 2017. [DOI | PubMed]
- B Basilico, L Ferrucci, P Ratano, MT Golia, A Grimaldi, M Rosito, V Ferretti, I Reverte, C Sanchini, MC Marrone. Microglia control glutamatergic synapses in the adult mouse hippocampus.. Glia, 2022. [DOI | PubMed]
- J Cornell, S Salinas, HY Huang, M Zhou. Microglia regulation of synaptic plasticity and learning and memory.. Neural Regen. Res, 2022. [DOI | PubMed]
- PT Nguyen, LC Dorman, S Pan, ID Vainchtein, RT Han, H Nakao-Inoue, SE Taloma, JJ Barron, AB Molofsky, MA Kheirbek, AV Molofsky. Microglial Remodeling of the Extracellular Matrix Promotes Synapse Plasticity.. Cell, 2020. [DOI | PubMed]
- CN Parkhurst, G Yang, I Ninan, JN Savas, JR Yates, JJ Lafaille, BL Hempstead, DR Littman, WB Gan. Microglia promote learning-dependent synapse formation through brain-derived neurotrophic factor.. Cell, 2013. [DOI | PubMed]
- T Pfeiffer, E Avignone, UV Nägerl. Induction of hippocampal long-term potentiation increases the morphological dynamics of microglial processes and prolongs their contacts with dendritic spines.. Sci. Rep, 2016. [DOI | PubMed]
- J Kim, Y Li. Chronic activation of CB2 cannabinoid receptors in the hippocampus increases excitatory synaptic transmission.. J. Physiol, 2015. [DOI | PubMed]
- JC Ashton, M Glass. The cannabinoid CB2 receptor as a target for inflammation-dependent neurodegeneration.. Curr. Neuropharmacol, 2007. [DOI | PubMed]
- AC Schmöle, R Lundt, S Ternes, Ö Albayram, T Ulas, JL Schultze, D Bano, P Nicotera, J Alferink, A Zimmer. Cannabinoid receptor 2 deficiency results in reduced neuroinflammation in an Alzheimer’s disease mouse model.. Neurobiol. Aging, 2015. [DOI | PubMed]
- A Viader, D Ogasawara, CM Joslyn, M Sanchez-Alavez, S Mori, W Nguyen, B Conti, BF Cravatt. A chemical proteomic atlas of brain serine hydrolases identifies cell type-specific pathways regulating neuroinflammation.. eLife, 2016. [DOI | PubMed]
- B Barti, B Dudok, K Kenesei, M Zöldi, V Miczán, GY Balla, D Zala, M Tasso, C Sagheddu, M Kisfali. Presynaptic nanoscale components of retrograde synaptic signaling.. Sci. Adv, 2024. [DOI | PubMed]
- F Koukouli, M Montmerle, A Aguirre, M De Brito Van Velze, J Peixoto, V Choudhary, M Varilh, F Julio-Kalajzic, C Allene, P Mendéz. Visual-area-specific tonic modulation of GABA release by endocannabinoids sets the activity and coordination of neocortical principal neurons.. Cell Rep., 2022. [DOI | PubMed]
- B Dudok, L Barna, M Ledri, SI Szabó, E Szabadits, B Pintér, SG Woodhams, CM Henstridge, GY Balla, R Nyilas. Cell-specific STORM super-resolution imaging reveals nanoscale organization of cannabinoid signaling.. Nat. Neurosci, 2015. [DOI | PubMed]
- C Földy, RC Malenka, TC Südhof. Autism-associated neuroligin-3 mutations commonly disrupt tonic endocannabinoid signaling.. Neuron, 2013. [DOI | PubMed]
- K Tsuboi, Y Okamoto, N Ikematsu, M Inoue, Y Shimizu, T Uyama, J Wang, DG Deutsch, MP Burns, NM Ulloa. Enzymatic formation of N-acylethanolamines from N-acylethanolamine plasmalogen through N-acylphosphatidylethanolamine-hydrolyzing phospholipase D-dependent and -independent pathways.. Biochim. Biophys. Acta, 2011. [DOI | PubMed]
- TJ Younts, V Chevaleyre, PE Castillo. CA1 pyramidal cell theta-burst firing triggers endocannabinoid-mediated long-term depression at both somatic and dendritic inhibitory synapses.. J. Neurosci, 2013. [DOI | PubMed]
- TJ Younts, HR Monday, B Dudok, ME Klein, BA Jordan, I Katona, PE Castillo. Presynaptic Protein Synthesis Is Required for Long-Term Plasticity of GABA Release.. Neuron, 2016. [DOI | PubMed]
- HR Monday, M Bourdenx, BA Jordan, PE Castillo. CB1-receptor-mediated inhibitory LTD triggers presynaptic remodeling via protein synthesis and ubiquitination.. eLife, 2020. [DOI | PubMed]
- M Loisy, G Bouisset, S Lopez, M Muller, A Spitsyn, J Duval, RA Piskorowski, L Verret, V Chevaleyre. Sequential inhibitory plasticities in hippocampal area CA2 and social memory formation.. Neuron, 2022. [DOI | PubMed]
- T Ohno-Shosaku, H Tsubokawa, I Mizushima, N Yoneda, A Zimmer, M Kano. Presynaptic cannabinoid sensitivity is a major determinant of depolarization-induced retrograde suppression at hippocampal synapses.. J. Neurosci, 2002. [DOI | PubMed]
- K Chen, A Ratzliff, L Hilgenberg, A Gulyás, TF Freund, M Smith, TP Dinh, D Piomelli, K Mackie, I Soltesz. Long-term plasticity of endocannabinoid signaling induced by developmental febrile seizures.. Neuron, 2003. [DOI | PubMed]
- CQ Chiu, PE Castillo. Input-specific plasticity at excitatory synapses mediated by endocannabinoids in the dentate gyrus.. Neuropharmacology, 2008. [DOI | PubMed]
- T Jain, J Wager-Miller, K Mackie, A Straiker. Diacylglycerol lipasealpha (DAGLα) and DAGLβ cooperatively regulate the production of 2-arachidonoyl glycerol in autaptic hippocampal neurons.. Mol. Pharmacol, 2013. [DOI | PubMed]
- SA Kodirov, K Bonni, M Wehrmeister, B Lutz. Depolarization-initiated endogenous cannabinoid release and underlying retrograde neurotransmission in interneurons of amygdala.. Learn. Mem, 2021. [DOI | PubMed]
- AD Milstein, Y Li, KC Bittner, C Grienberger, I Soltesz, JC Magee, S Romani. Bidirectional synaptic plasticity rapidly modifies hippocampal representations.. eLife, 2021. [DOI | PubMed]
- WR Marrs, JL Blankman, EA Horne, A Thomazeau, YH Lin, J Coy, AL Bodor, GG Muccioli, SSJ Hu, G Woodruff. The serine hydrolase ABHD6 controls the accumulation and efficacy of 2-AG at cannabinoid receptors.. Nat. Neurosci, 2010. [DOI | PubMed]
- AV Naydenov, EA Horne, CS Cheah, K Swinney, KL Hsu, JK Cao, W Marrs, JL Blankman, S Tu, AE Cherry. ABHD6 blockade exerts antiepileptic activity in PTZ-induced seizures and in spontaneous seizures in R6/2 mice.. Neuron, 2014. [DOI | PubMed]
- JK Cao, J Kaplan, N Stella. ABHD6: Its Place in Endocannabinoid Signaling and Beyond.. Trends Pharmacol. Sci, 2019. [DOI | PubMed]
- JL Blankman, GM Simon, BF Cravatt. A comprehensive profile of brain enzymes that hydrolyze the endocannabinoid 2-arachidonoylglycerol.. Chem. Biol, 2007. [DOI | PubMed]
- DK Nomura, BE Morrison, JL Blankman, JZ Long, SG Kinsey, MCG Marcondes, AM Ward, YK Hahn, AH Lichtman, B Conti, BF Cravatt. Endocannabinoid hydrolysis generates brain prostaglandins that promote neuroinflammation.. Science, 2011. [DOI | PubMed]
- R Chen, J Zhang, Y Wu, D Wang, G Feng, YP Tang, Z Teng, C Chen. Monoacylglycerol lipase is a therapeutic target for Alzheimer’s disease.. Cell Rep., 2012. [DOI | PubMed]
- G Terrone, A Pauletti, A Salamone, M Rizzi, BR Villa, L Porcu, MJ Sheehan, E Guilmette, CR Butler, JR Piro. Inhibition of monoacylglycerol lipase terminates diazepam-resistant status epilepticus in mice and its effects are potentiated by a ketogenic diet.. Epilepsia, 2018. [DOI | PubMed]
- EL von Rüden, RM Bogdanovic, CT Wotjak, H Potschka. Inhibition of monoacylglycerol lipase mediates a cannabinoid 1-receptor dependent delay of kindling progression in mice.. Neurobiol. Dis, 2015. [DOI | PubMed]
- Y Sugaya, M Yamazaki, M Uchigashima, K Kobayashi, M Watanabe, K Sakimura, M Kano. Crucial Roles of the Endocannabinoid 2-Arachidonoylglycerol in the Suppression of Epileptic Seizures.. Cell Rep., 2016. [DOI | PubMed]
- DR Powell, JP Gay, N Wilganowski, D Doree, KV Savelieva, TH Lanthorn, R Read, P Vogel, GM Hansen, R Brommage. Diacylglycerol Lipase alpha Knockout Mice Demonstrate Metabolic and Behavioral Phenotypes Similar to Those of Cannabinoid Receptor 1 Knockout Mice.. Front. Endocrinol. (Lausanne), 2015. [DOI | PubMed]
- N Stella. Cannabinoid and cannabinoid-like receptors in microglia, astrocytes, and astrocytomas.. Glia, 2010. [DOI | PubMed]
- P Gómez-Sotres, U Skupio, T Dalla Tor, F Julio-Kalajzic, A Cannich, D Gisquet, I Bonilla-Del Rio, F Drago, N Puente, P Grandes. Olfactory bulb astrocytes link social transmission of stress to cognitive adaptation in male mice.. Nat. Commun, 2024. [DOI | PubMed]
- C Chen. Inhibiting degradation of 2-arachidonoylglycerol as a therapeutic strategy for neurodegenerative diseases.. Pharmacol. Ther, 2023. [DOI | PubMed]
- O Devinsky, JH Cross, L Laux, E Marsh, I Miller, R Nabbout, IE Scheffer, EA Thiele, S Wright. Trial of Cannabidiol for Drug-Resistant Seizures in the Dravet Syndrome.. N. Engl. J. Med, 2017. [DOI | PubMed]
- S Billakota, O Devinsky, E Marsh. Cannabinoid therapy in epilepsy.. Curr. Opin. Neurol, 2019. [DOI | PubMed]
- JS Farrell, I Soltesz. Plants come to mind: phytocannabinoids, endocannabinoids and the control of seizures.. Addiction, 2019. [DOI | PubMed]
