Year-Long Cannabis Use for Medical Symptoms and Brain Activation During Cognitive Processes
Abstract
Question:
Is there an association between year-long cannabis use for medical symptoms and brain activation during cognitive processes implicated in cannabis use?
Findings:
In a cohort study of adults who newly obtained medical cannabis cards for symptoms of depression, anxiety, pain, or insomnia, functional magnetic resonance imaging measures during working memory, reward, and inhibitory control tasks did not differ statistically from baseline to 1 year and were not associated with changes in cannabis use frequency.
Meaning:
The absence of activation differences in this study suggests that adults using cannabis for medical symptoms over 1 year may not experience significant changes within reward, working memory, or inhibitory control domains.
Affiliations: McGovern Institute for Brain Research, MIT, Cambridge, Massachusetts; Harvard Medical School, Boston, Massachusetts; University of Pennsylvania School of Engineering and Applied Science, Philadelphia; Center for Addiction Medicine, Massachusetts General Hospital, Boston; Department of Otolaryngology–Head and Neck Surgery, Harvard Medical School, Boston, Massachusetts; Athinoula A. Martinos Center for Biomedical Imaging, Department of Radiology, Massachusetts General Hospital, Boston
License: Copyright 2024 Burdinski DCL et al. JAMA Network Open. CC BY 4.0 This is an open access article distributed under the terms of the CC-BY License.
Article links: DOI: 10.1001/jamanetworkopen.2024.34354 | PubMed: 39292455 | PMC: PMC11411392
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (16 KB)
Introduction
Accumulating evidence has shown that regular cannabis use can alter brain function, especially in networks that support working memory, cognitive control, and reward processing.1 Several prior reviews have described the functional impact of chronic cannabis use in both adults and adolescents,2,3,4 largely concluding that the domains of executive functioning and memory are most strongly affected by regular cannabis use.5,6 However, most of the evidence for brain changes with cannabis use is derived from between-group brain differences between those who use cannabis and those who do not, rather than from longitudinal changes at pre– and post–cannabis use time points, raising the question of whether preexisting differences between those who use cannabis and those who do not underlie observed changes. Longitudinal studies, such as the Adolescent Brain Cognitive Development Study,7 are under way. To date, however, few studies are focused on adults using cannabis to treat medical symptoms. Little is known about the effects of cannabis on the brain in medical populations, who may also experience illness-related cognitive weaknesses and may have different use patterns and age ranges compared with recreational users.
Delta-9-tetrahydrocannabinol (THC), the main psychoactive compound in cannabis, binds to endogenous cannabinoid CB1 receptors located in brain regions such as the hippocampus, amygdala, basal ganglia, prefrontal cortex, substantia nigra, and globus pallidus,8,9 making frontal-limbic neurocircuitry particularly susceptible to cannabis-related effects in the brain.10 Specifically, THC binding inhibits the release of neurotransmitters usually modulated through endocannabinoids.11 Many factors can modulate THC’s impact on the brain, including duration, frequency and quantity of use, age of initiation, potency, accompanying cannabidiol content, presence of cannabis use disorder (CUD), concurrent use of other substances, and sex and genetics.12
The question of how cannabis affects the brain is particularly relevant to those using cannabis to treat medical symptoms. Currently in the United States, 38 states and the District of Columbia have medical cannabis programs, and enrollment in medical cannabis programs increased 4.5-fold from 2016 to 2020.13 In Massachusetts, obtaining a medical cannabis card (MCC) gives patients access to tax-exempt cannabis purchases and additional medical dispensaries. However, evidence for the effectiveness of plant-based cannabis for any medical condition is sparse.14 In dispensaries, a myriad of products (eg, candies, gummies, smoked, vaped) are available to those using medically, and the neural effects of these products are unknown.
We sought to describe cognitive and brain-based associations with cannabis use in a longitudinal sample of participants obtaining MCCs and beginning to use cannabis for symptoms of anxiety, depression, pain, and insomnia. We previously published a pragmatic randomized clinical trial (RCT) of MCCs for medical symptoms to assess their effect on target symptoms when compared with a waitlist control group.15,16 In the current cohort study, we describe a longitudinal analysis of task-based functional magnetic resonance imaging (fMRI) data from the arm of the clinical trial in which participants were assigned to obtain MCCs immediately. We explore the extent to which cannabis was associated with brain activation during cognitive processes previously implicated in cannabis use, using neuroimaging tasks that probe working memory, reward processing, and inhibitory control. We hypothesized, based on previous literature,17,18,19 that 1 year of cannabis use would be associated with generally increased activation in brain regions underlying these processes, and that an increase in cannabis use frequency would be associated with this increased activation, with few associated differences in task performance over the study period.
Methods
Study Recruitment
The participants of this observational cohort study were recruited for a pragmatic, single-site, single-blind, RCT assessing patients seeking MCCs in the greater Boston area from July 1, 2017, to July 31, 2020 (NCT03224468).15,16 Participants were between the ages of 18 and 65 years and were seeking to obtain MCCs for the first time for depression, anxiety, pain, or insomnia symptoms, the most commonly reported symptoms in those seeking cannabis for symptom management. Exclusion criteria included daily cannabis use, CUD diagnosis at screening or baseline, cancer, psychosis, and current substance use disorders (except for mild or moderate alcohol use disorder and nicotine use disorder).
Study Protocol
Participants were randomized to either receive their MCC immediately or to delay acquisition by 12 weeks. Only the immediate MCC group received fMRI scans at baseline and at 1 year, and these data are presented here. Demographic variables were collected at baseline through self-report and included sex, age, race, ethnicity, years of education, the primary symptom for seeking MCCs, and handedness. Race and ethnicity were categorized as suggested by the National Institutes of Health for reporting and research purposes: for race, American Indian or Alaska Native, Black or African American, Asian, Native Hawaiian or Other Pacific Islander, and White; for ethnicity, Hispanic or Latino or not Hispanic or Latino. Due to limited numbers, Asian, Native Hawaiian or other Pacific Islander, and multiracial were collapsed into one category labeled other. Cannabis use metrics were collected at baseline and at 2, 4, 12, 24, and 52 weeks and included past-month frequency of cannabis use and a validated scale to assess CUD, the Cannabis Use Disorder Identification Test Revised (CUDIT-R). In addition, urinalysis for cannabis metabolites was conducted at the time of study visits. Structural and functional brain imaging data were collected in the MCC group (70 participants) (eFigure 1 in Supplement 1) at baseline and 1 year later. At baseline, imaging was also collected in an age- and sex-matched healthy control (HC) group (32 participants). fMRI tasks included a working memory task (N-back), a reward processing task (monetary incentive delay [MID]), and an inhibitory response task (stop signal task [SST]).20,21,22 The eMethods in Supplement 1 provide a description of the experimental paradigm (eFigures 2-4 in Supplement 1). Participants provided written informed consent and were financially compensated for their participation in the study. The clinical trial, as part of which data for this cohort study were collected, was approved by the Massachusetts General Brigham institutional review board. This report focuses on task-based functional imaging, which followed a pretest-posttest design with a control group at baseline. Clinical outcomes of the RCT are reported elsewhere.15,16 This study follows the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines.
Statistical Analysis
Demographic metrics were compared across 3 groups (HC participants, MCC participants imaged at both time points, and MCC participants who only received a scan at 1 time point) using a Kruskal-Wallis test for numerical variables, a Fisher exact test for categorical variables with less than 5 observations in a category, and a χ2 test for all other categorical variables. Cannabis metrics in the MCC participants who were imaged at both time points were compared across the 2 time points (baseline and 1 year) using a linear mixed-effects model with a participant-varying intercept to account for repeated measures and age and sex as covariates.
To analyze behavioral performance of tasks, differences between the HC and MCC groups at baseline and between the 2 time points of the MCC participants were assessed. For the N-back task, we analyzed reaction time and accuracy across the 2-back, 0-back, and combined stimuli using a linear regression model with age and sex as covariates. For the SST, we analyzed stop signal reaction time (SSRT; the inferred mean latency between the stop signal and response inhibition) via an additive multilevel linear model. Across-time models also included a participant-varying intercept to account for repeated measures. The MID task did not include a behavioral component, as response periods were dynamically updated throughout a run to maintain a similar accuracy across participants.
MRI data were preprocessed using version 23.0.1 of the fMRIPrep software, which included head-motion estimation, slice time correction, field map–based distortion correction, echo-planar imaging to T1 registration and resampling to both Montreal Neurological Institute volumetric and grayordinate space.23,24 The eMethods in Supplement 1 provide details on MRI acquisition and preprocessing.
Two general linear model analyses were conducted, one in volumetric and the other in grayordinate space, using the Python package Nilearn version 0.9.2.25,26 First-level linear regression modeling removed further noise and modeled typical task contrasts. Individual effect sizes for the contrasts were passed to a group-level linear regression model to assess group averages at a given time point, differences across groups and time points, and the role of changes in cannabis use frequency. Covariates included sex, age, and past-month cannabis use frequency, mean-centered for numerical variables. Whole brain activation was compared between MCC participants and a matched control group at baseline using a contrast between group-level intercepts. For the across-time analyses of the MCC group, observations were limited to those participants with imaging at both time points. We assessed whether, on average, there was a whole brain activation difference between baseline and 1 year in an individual, controlling for baseline cannabis use frequency to account for individual differences in use at the outset of the study. Furthermore, the association of the change in cannabis use frequency across time with whole brain activation at 1 year was assessed, adding the additional covariates of brain activation and cannabis use frequency at baseline. Of note, repeated measures in the across-time analysis were accounted for by including baseline values as covariates or by using a change score.
Quality control metrics derived from MRIQC (version 0.16.1) were used to exclude runs of lower quality prior to running group-level analyses, which led to varying sample sizes across tasks.27 The effect of quality control was assessed by running the analyses with varying exclusion criteria. Effect sizes at the group level were standardized for visualization purposes.28,29 The eMethods in Supplement 1 provide details on the general linear modeling approach and quality control metrics.
The statistical significance level was set to P < .05 for all analyses. Multiple comparisons were addressed by controlling the false-discovery rate (FDR) at .05 using the Benjamini-Hochberg procedure. Analyses were run in R version 4.3.3 (R Project for Statistical Computing) and Python version 3.9.13 (Python Software Foundation). Data analysis was conducted from August 2021 to April 2024.
Results
Participant Characteristics
Of 120 MCC participants in the parent trial, brain imaging data were collected in 70 as well as in 32 control participants (eFigure 1 in Supplement 1). A total of 35 MCC participants opted out of the MRI protocol, 11 were excluded due to having CUD at baseline, and 4 were lost to follow-up. At baseline, 57 MCC participants (38 female [66.7%]; 6 [10.5%] Black and 45 [78.9%] White participants; 1 [1.8%] Hispanic participant; median [IQR] age, 34.0 [24.0-51.0] years) and 32 control participants (22 female [68.8%]; 2 [6.2%] Black and 27 [84.4%] White participants; 3 [9.4%] Hispanic participants; median [IQR] age, 33.0 [24.8-38.2] years) were imaged. After 1 year, 54 MCC participants (37 female [68.5%]; 4 [7.4%] Black and 48 [88.9%] White participants; 1 [1.9%] Hispanic participant, median [IQR] age, 36.5 [25.0-51.0] years) were imaged. Of all MCC participants, 41 presented for imaging at both time points (28 female [68.3%]; 2 [4.9%] Black and 37 [90.2%] White participants; 1 [2.4%] Hispanic participant; median [IQR] age, 38.0 [26.0-51.0] years). MCC participants presenting for 1 scan, MCC participants presenting for 2 scans, and HC participants did not differ significantly in any of the characteristics assessed (Table). All cannabis use metrics, including CUDIT-R summed score, cannabis use frequency per month, and positive urine THC, were greater at 1 year than at baseline in the MCC group (eTable 1 in Supplement 1).
Table. : Characteristics of the Study Participants
| Characteristic | Participants, No. (%) | |||
|---|---|---|---|---|
| HC at Baseline (n = 32)zoi241023t1n1 | MCC | |||
| Baseline (n = 57)zoi241023t1n2 | 1 y (n =54)zoi241023t1n3 | Paired (n = 41)zoi241023t1n4 | ||
| Sex | ||||
| Female | 22 (68.8) | 38 (66.7) | 37 (68.5) | 28 (68.3) |
| Male | 10 (31.2) | 19 (33.3) | 17 (31.5) | 13 (31.7) |
| Age, median (IQR) | 33.0 (24.8-38.2) | 34.0 (24.0-51.0) | 36.5 (25.0-51.0) | 38.0 (26.0-51.0) |
| Race | ||||
| Black | 2 (6.2) | 6 (10.5) | 4 (7.4) | 2 (4.9) |
| Otherzoi241023t1n5 | 3 (9.4) | 6 (10.5) | 2 (3.7) | 2 (4.9) |
| White | 27 (84.4) | 45 (78.9) | 48 (88.9) | 37 (90.2) |
| Ethnicity | ||||
| Hispanic or Latino | 3 (9.4) | 1 (1.8) | 1 (1.9) | 1 (2.4) |
| Education, median (IQR), y | 17.5 (16.0-19.2) | 16.0 (16.0-18.0) | 16.5 (16.0-18.0) | 17.0 (16.0-18.0) |
| Primary symptom | ||||
| Depression or anxiety symptoms | NA | 27 (47.4) | 24 (44.4) | 19 (46.3) |
| Insomnia symptoms | NA | 12 (21.1) | 13 (24.1) | 9 (22.0) |
| Pain symptoms | NA | 18 (31.6) | 17 (31.5) | 13 (31.7) |
| Handedness | ||||
| Right-handed | 29 (90.6) | 49 (86.0) | 46 (85.2) | 35 (85.4) |
HC baseline corresponds to the imaging control group at baseline.
MCC baseline corresponds to the MCC group’s participants imaged at baseline.
MCC 1 year corresponds to the MCC group’s participants imaged at 1 year.
MCC paired corresponds to the MCC group’s participants imaged at both time points.
Other includes individuals identifying as Asian, Native Hawaiian or other Pacific Islander, and multiracial.
Abbreviations: HC, healthy control; MCC, medical cannabis card; NA, not applicable.
N-Back Task Behavioral and Imaging Results
At baseline, no performance differences in either accuracy or reaction time were observed between HC and MCC participants. MCC participants had a significantly faster mean (SD) 2-back reaction time at 1 year (540 [14] ms) compared with baseline (582 [13] ms) (β = −42.5; SE, 14.6; P = .04), and no difference in any of the other behavioral measures (eTable 2 in Supplement 1).
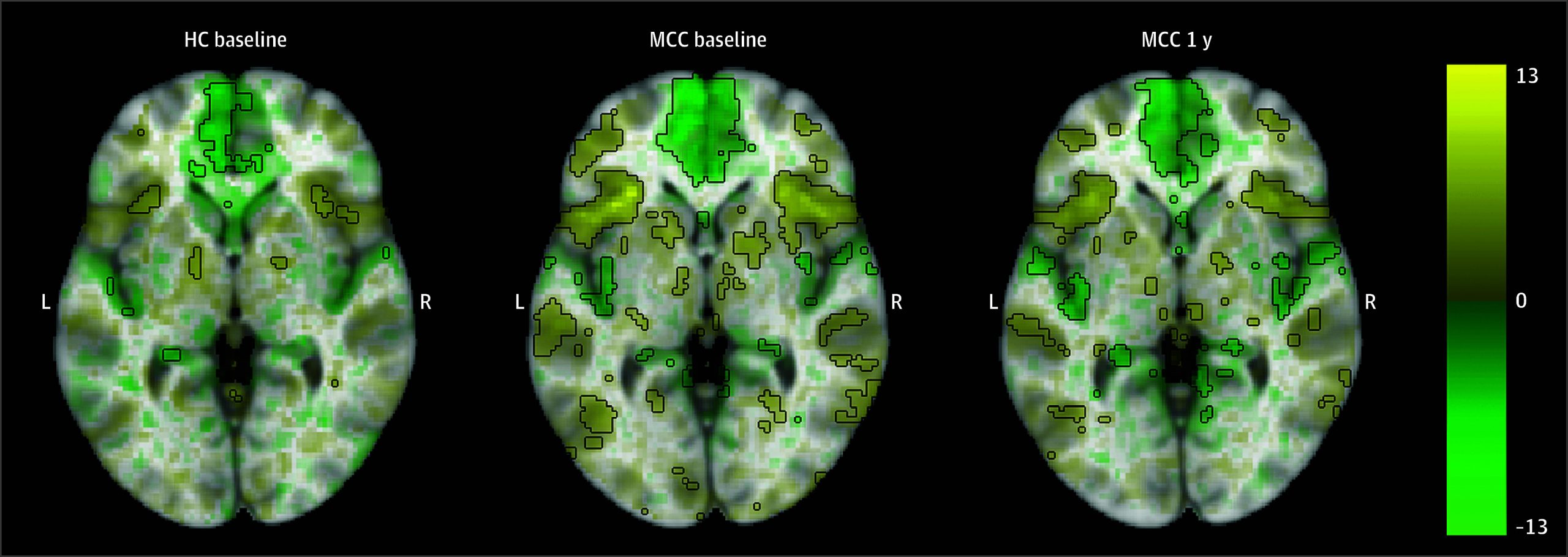
Activation in prefrontal and parietal cortical regions was observed for the 2-back vs 0-back image contrast in all groups, including MCC participants at baseline and 1 year as well as control participants (Figure 1; eAppendix 1 and eFigure 5 in Supplement 1). There were no significant differences in activation between the groups at baseline (22 HC participants and 40 MCC participants) or between the 2 time points of the MCC group (25 participants), and no associations between cannabis use frequency change and activation at 1 year for the MCC group were significant.
MID Task Imaging Results
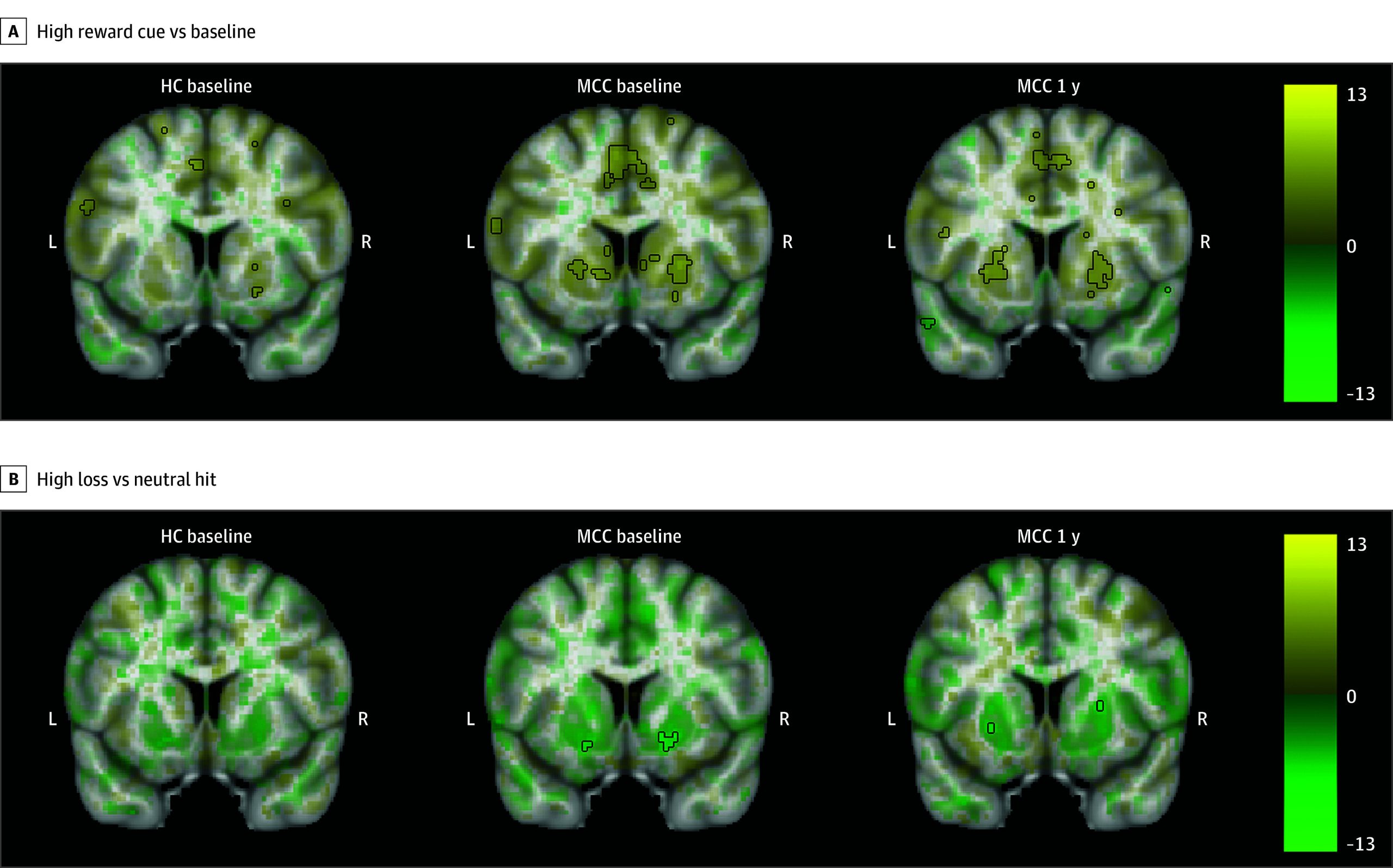
MCC participants at baseline and 1 year, as well as controls, showed activation in the bilateral basal ganglia during all cue contrasts, although activation only reached statistical significance during the high reward cue vs baseline contrast (Figure 2; eAppendix 1 and eFigures 6-8 in Supplement 1).30 During the reward vs missed reward feedback contrast, activation in the bilateral basal ganglia was observed, while during all other feedback contrasts, deactivation in the bilateral basal ganglia and insula was observed, although only significant in the high loss vs neutral hit contrast. No significant differences in activation were observed between the groups at baseline (23 HC participants and 35 MCC participants) or between the 2 time points of the MCC group (22 participants). Within the MCC group, there were no significant associations between brain activation at 1 year and cannabis use frequency changes.
SST Task Behavioral and Imaging Results
At baseline, HC participants had significantly faster mean (SD) SSRT (259 [43] milliseconds) compared with MCC participants (276 [40] milliseconds) (β = −16.2; SE, 7.7; P = .04), indicating better inhibitory control. MCC participants had a nonsignificant reduction in mean (SD) SSRT from baseline (276 [40] milliseconds) to 1 year (264 [43] milliseconds) (β = −10.0; SE, 5.1; P = .05) (eTable 3 in Supplement 1).
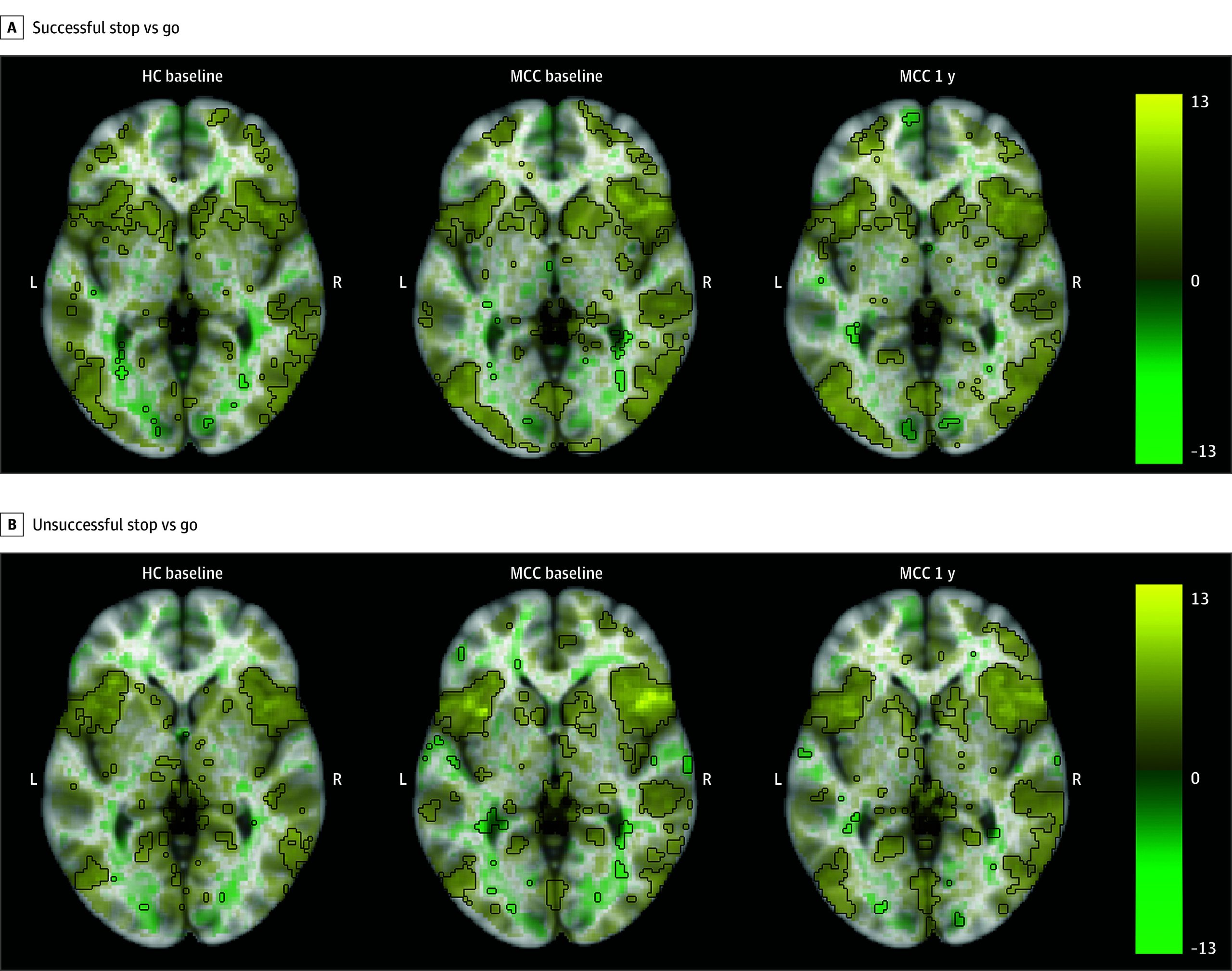
All groups showed activation in inhibitory control-related regions, including the right inferior frontal gyrus, frontal gyrus, and insula during the correct inhibition, incorrect inhibition, and successful inhibitory control contrasts (Figure 3; eAppendix 1 and eFigures 9-11 in Supplement 1). No differences in activation between the groups at baseline (25 HC participants and 40 MCC participants) or between the 2 time points of the MCC group (26 participants), and no associations of cannabis use frequency change with activation at 1 year for the MCC group were significant.
Effect of Quality Control on Neuroimaging Results
We note that we removed between 7 and 22 participants based on quality control metrics that were calculated separately for each run. Including additional participants in the analysis by relaxing the framewise displacement threshold from 0.2 to 0.3 or by including all participants regardless of quality control metrics did not significantly change the results (eAppendix 2 and eFigures 12-17 in Supplement 1).
Discussion
After year-long cannabis use for medical symptoms in adults who newly obtained MCCs, we did not observe functional differences between baseline and brain activation at 1 year during working memory, reward processing, or inhibitory control tasks, nor an association between changes in cannabis use frequency and brain activation at 1 year. Similarly, few significant changes in behavioral performance emerged. This suggests that cannabis use for medical purposes, within the snapshot of cognition captured by these tasks and within a mostly older, White, female, and generally well-educated population, did not have a significant association with brain activation or cognitive performance.
Prior studies have found that cannabis use, especially in adolescents, is associated with impairments in cognitive processes beyond acute intoxication.11,31,32 Such studies have largely been cross-sectional, have generally not focused on adults using cannabis for medical purposes, and have focused primarily on heavy cannabis use. Furthermore, conclusions of studies comparing individuals who use cannabis with those who do not often are limited by inherent group differences at the outset. Because participants in this study did not use cannabis heavily at baseline and obtained MCCs for the first time for their medical symptoms, the study was uniquely positioned to examine the brain before and after adults began to use cannabis regularly, a period that is difficult to capture. To our knowledge, this study is among the first to evaluate brain activation differences in an ecologically valid setting in those who began using cannabis for medical symptoms. Brain activation differences were also not found between HC participants and MCC participants at baseline, suggesting that the MCC group did not differ significantly at baseline from those who did not intend to use cannabis.
Memory is one of the most consistently reported processes that is affected by cannabis.33 Prior studies comparing those who use cannabis with those who do not have found significant changes in activation of frontal regions during the N-back task,34,35,36,37 although it should be noted that other studies did not report statistically significant differences.38,39 Reward-related activation has also been implicated in cannabis use, as prior studies have found significant changes in the activation of striatal regions during the MID task.40,41,42,43,44,45 Finally, inhibitory control activation differences have been reported,46,47 particularly in fronto-basal-ganglia circuits.48 However, these studies mainly consist of those who began using cannabis during adolescence or those who use cannabis frequently. Contrary to our initial hypothesis and this literature, our findings indicate that activation to working memory, reward processing, and inhibitory control tasks is largely unchanged in adults using cannabis to alleviate medical symptoms for 1 year. This study population may differ from previous studies of recreational cannabis in participants’ use patterns, motivations for use, age, or other factors.
Overall, the activation patterns of all 3 tasks were consistent with those observed in the literature. The working memory and inhibitory control tasks yielded statistically significant canonical activations in the control and MCC groups at baseline and in the MCC group at 1 year.20,48,49,50 The reward processing task also yielded canonical activations for the cue contrasts.30,50,51,52,53 Activations to feedback were consistent with previous studies, though the literature is less robust in regards to the feedback contrast.51,52,53 Of note, only 2 of the MID contrasts reached statistical significance. This suggests that the response to the task was more heterogeneous in this participant sample, and perhaps a larger sample or a differently designed reward task would have been needed to achieve more robust activation. It was recently noted that the MID task can have low reliability, which is consistent with our findings.54
Limitations
This study should be interpreted in light of its limitations. First, the sample was predominantly female, White, older, and well-educated, which may limit the generalizability of our findings. Future studies should thus focus on recruiting a more diverse sample. It is further possible that the lack of difference in task-based activation was due to limited power. We note that the maximal absolute effect size difference between the 2 time points in the MCC group was smaller than the maximal absolute effect size across the 2 time points for all 3 tasks. Thus, even if we did not detect a difference despite one existing, the change in brain activation after year-long cannabis use would be small. This study took place partly during the COVID-19 pandemic, which caused some participants to opt out of scanning procedures, reducing the sample size of those with neuroimaging data.
Importantly, adult-onset use of cannabis for medical symptoms after obtaining MCCs likely has different neural implications compared with recreational adolescent use. Moreover, comorbid conditions (eg, depression or pain) may influence the impact of cannabis on the brain. While our sample size was too small for a subgroup analysis of each of the symptoms for which participants sought MCCs, future studies should enroll sufficient participants to be able to discover the impact of cannabis within symptoms. Additionally, to emulate the system of medical cannabis in place, participants freely chose cannabis products at local dispensaries. Therefore, it is possible that doses of cannabinoids were too low to observe brain changes. Further research is warranted to understand how differences in product type, amounts, and patterns of use might affect the brain in cannabis users for medical symptoms. Additional limitations are presented in eAppendix 3 in Supplement 1.
Conclusions
In this cohort study of adults obtaining MCCs for medical symptoms, brain activation during working memory, reward processing, and inhibitory control tasks was not significantly different after year-long cannabis use and no association with changes in cannabis use frequency was noted. Our results suggest that adults who use cannabis, generally with light to moderate use patterns, for symptoms of pain, anxiety, depression, or poor sleep, experience few significant long-term neural associations in these areas of cognition.
References
- Cannabis effects on brain structure, function, and cognition: considerations for medical uses of cannabis and its derivatives.. Am J Drug Alcohol Abuse., 2019. [DOI | PubMed]
- Structural and functional imaging studies in chronic cannabis users: a systematic review of adolescent and adult findings.. PLoS One., 2013. [DOI | PubMed]
- Neuroimaging meta-analysis of cannabis use studies reveals convergent functional alterations in brain regions supporting cognitive control and reward processing.. J Psychopharmacol., 2018. [DOI | PubMed]
- Reduced responsiveness of the reward system is associated with tolerance to cannabis impairment in chronic users.. Addict Biol., 2021. [DOI | PubMed]
- Effects of cannabis on neurocognitive functioning: recent advances, neurodevelopmental influences, and sex differences.. Neuropsychol Rev., 2013. [DOI | PubMed]
- Considering cannabis: the effects of regular cannabis use on neurocognition in adolescents and young adults.. Curr Addict Rep., 2014. [DOI | PubMed]
- Introduction.. Dev Cogn Neurosci., 2018. [DOI | PubMed]
- Distribution of cannabinoid receptors in the central and peripheral nervous system.. Handb Exp Pharmacol., 2005. [DOI | PubMed]
- The molecular logic of endocannabinoid signalling.. Nat Rev Neurosci., 2003. [DOI | PubMed]
- Neuroimaging in cannabis use: a systematic review of the literature.. Psychol Med., 2010. [DOI | PubMed]
- Evidence on the acute and residual neurocognitive effects of cannabis use in adolescents and adults: a systematic meta-review of meta-analyses.. Addiction., 2022. [DOI | PubMed]
- Effects of cannabis use on human behavior, including cognition, motivation, and psychosis: a review.. JAMA Psychiatry., 2016. [DOI | PubMed]
- U.S. trends in registration for medical cannabis and reasons for use from 2016 to 2020: an observational study.. Ann Intern Med., 2022. [DOI | PubMed]
- 14National Academies of Sciences, Engineering, and Medicine; Health and Medicine Division; Board on Population Health and Public Health Practice; Committee on the Health Effects of Marijuana: An Evidence Review and Research Agenda. Therapeutic Effects of Cannabis and Cannabinoids. National Academies Press; 2017.
- Effect of medical marijuana card ownership on pain, insomnia, and affective disorder symptoms in adults: a randomized clinical trial.. JAMA Netw Open., 2022. [DOI | PubMed]
- Development of cannabis use disorder in medical cannabis users: a 9-month follow-up of a randomized clinical trial testing effects of medical cannabis card ownership.. Front Psychiatry., 2023. [DOI | PubMed]
- fMRI study of neural sensitization to hedonic stimuli in long-term, daily cannabis users.. Hum Brain Mapp., 2016. [DOI | PubMed]
- Abstinent adolescent marijuana users show altered fMRI response during spatial working memory.. Psychiatry Res., 2008. [DOI | PubMed]
- The impact of early-onset cannabis use on functional brain correlates of working memory.. Prog Neuropsychopharmacol Biol Psychiatry., 2010. [DOI | PubMed]
- N-back working memory paradigm: a meta-analysis of normative functional neuroimaging studies.. Hum Brain Mapp., 2005. [DOI | PubMed]
- FMRI visualization of brain activity during a monetary incentive delay task.. Neuroimage., 2000. [DOI | PubMed]
- Impulsivity and inhibitory control.. Psychol Sci., 1997. [DOI]
- The minimal preprocessing pipelines for the Human Connectome Project.. Neuroimage., 2013. [DOI | PubMed]
- fMRIPrep: a robust preprocessing pipeline for functional MRI.. Nat Methods., 2019. [DOI | PubMed]
- Statistical parametric maps in functional imaging: a general linear approach.. Hum Brain Mapp., 1994. [DOI]
- Machine learning for neuroimaging with scikit-learn.. Front Neuroinform., 2014. [DOI | PubMed]
- MRIQC: advancing the automatic prediction of image quality in MRI from unseen sites.. PLoS One., 2017. [DOI | PubMed]
- Standardized effect sizes and image-based meta-analytical approaches for fMRI data.. bioRxiv. Preprint posted online December 6, 2019. doi:. [DOI]
- Distribution theory for Glass’s estimator of effect size and related estimators.. J Educ Behav Stat., 1981. [DOI]
- The neural substrate of reward anticipation in health: a meta-analysis of fMRI findings in the Monetary Incentive Delay Task.. Neuropsychol Rev., 2018. [DOI | PubMed]
- Neural mechanisms of sensitivity to peer information in young adult cannabis users.. Cogn Affect Behav Neurosci., 2016. [DOI | PubMed]
- Variable activation in striatal subregions across components of a social influence task in young adult cannabis users.. Brain Behav., 2016. [DOI | PubMed]
- Impulsivity, attention, memory, and decision-making among adolescent marijuana users.. Psychopharmacology (Berl)., 2013. [DOI | PubMed]
- Effects of marijuana on visuospatial working memory: an fMRI study in young adults.. Psychopharmacology (Berl)., 2010. [DOI | PubMed]
- Fronto-striatal effective connectivity of working memory in adults with cannabis use disorder.. Psychiatry Res Neuroimaging., 2018. [DOI | PubMed]
- Prefrontal activity during working memory is modulated by the interaction of variation in CB1 and COX2 coding genes and correlates with frequency of cannabis use.. Cortex., 2016. [DOI | PubMed]
- Working overtime: altered functional connectivity in working memory following regular cannabis use in young adults.. Int J Ment Health Addict., 2021. [DOI]
- Effect of baseline cannabis use and working-memory network function on changes in cannabis use in heavy cannabis users: a prospective fMRI study.. Hum Brain Mapp., 2014. [DOI | PubMed]
- Relationship between working-memory network function and substance use: a 3-year longitudinal fMRI study in heavy cannabis users and controls.. Addict Biol., 2014. [DOI | PubMed]
- Chronic effects of cannabis use on the human reward system: an fMRI study.. Eur Neuropsychopharmacol., 2010. [DOI | PubMed]
- Attenuated reward activations associated with cannabis use in anxious/depressed individuals.. Transl Psychiatry., 2020. [DOI | PubMed]
- Association of marijuana use with blunted nucleus accumbens response to reward anticipation.. JAMA Psychiatry., 2016. [DOI | PubMed]
- Alterations of monetary reward and punishment processing in chronic cannabis users: an FMRI study.. PLoS One., 2015. [DOI | PubMed]
- Increased ventral striatal BOLD activity during non-drug reward anticipation in cannabis users.. Neuroimage., 2010. [DOI | PubMed]
- Pretreatment measures of brain structure and reward-processing brain function in cannabis dependence: an exploratory study of relationships with abstinence during behavioral treatment.. Drug Alcohol Depend., 2014. [DOI | PubMed]
- Parsing impulsivity in individuals with anxiety and depression who use cannabis.. Drug Alcohol Depend., 2020. [DOI | PubMed]
- Functional connectivity in inhibitory control networks and severity of cannabis use disorder.. Am J Drug Alcohol Abuse., 2013. [DOI | PubMed]
- Response inhibition in the stop-signal paradigm.. Trends Cogn Sci., 2008. [DOI | PubMed]
- A consensus guide to capturing the ability to inhibit actions and impulsive behaviors in the stop-signal task.. Elife., 2019. [DOI | PubMed]
- Baseline brain function in the preadolescents of the ABCD Study.. Nat Neurosci., 2021. [DOI | PubMed]
- The anticipation and outcome phases of reward and loss processing: a neuroimaging meta-analysis of the monetary incentive delay task.. Hum Brain Mapp., 2018. [DOI | PubMed]
- Dissociation of reward anticipation and outcome with event-related fMRI.. Neuroreport., 2001. [DOI | PubMed]
- Interactions between methodological and interindividual variability: how Monetary Incentive Delay (MID) task contrast maps vary and impact associations with behavior.. Brain Behav., 2021. [DOI | PubMed]
- Impact of analytic decisions on test-retest reliability of individual and group estimates in functional magnetic resonance imaging: a multiverse analysis using the monetary incentive delay task.. bioRxiv. Preprint posted online March 20, 2024. doi:. [DOI]
