A decision-space model explains context-specific decision-making
Abstract
Optimal decision-making requires consideration of internal and external contexts. Biased decision-making is a transdiagnostic symptom of neuropsychiatric disorders. We created a computational model demonstrating how the striosome compartment of the striatum constructs a context-dependent mathematical space for decision-making computations, and how the matrix compartment uses this space to define action value. The model explains multiple experimental results and unifies other theories like reward prediction error, roles of the direct versus indirect pathways, and roles of the striosome versus matrix, under one framework. We also found, through new analyses, that striosome and matrix neurons increase their synchrony during difficult tasks, caused by a necessary increase in dimensionality of the space. The model makes testable predictions about individual differences in disorder susceptibility, decision-making symptoms shared among neuropsychiatric disorders, and differences in neuropsychiatric disorder symptom presentation. The model provides new evidence for the central role that striosomes play in neuroeconomic and disorder-affected decision-making.
Affiliations: .Computational Science Program, University of Texas at El Paso, EI Paso, TX, USA; .Department of Biological Sciences, University of Texas at El Paso, El Paso, TX, USA; .Artificial Intelligence Laboratory, Department of Computer Science, Massachusetts Institute of Technology, Cambridge, MA, USA; .Ming Hsieh Department of Electrical and Computer Engineering, Viterbi School of Engineering, University of Southern California, Los Angeles, CA; .Department of Mathematics and Statistics, Old Dominion University, Norfolk, VA, USA; .National Institute on Drug Abuse, Baltimore, MD, USA; .Department of Psychiatry; Center for Translational Medicine and Pharmacology; Friedman Brain Institute; Icahn School of Medicine at Mount Sinai, NY, USA; .Lead author
License: CC BY 4.0 This work is licensed under a Creative Commons Attribution 4.0 International License, which allows reusers to distribute, remix, adapt, and build upon the material in any medium or format, so long as attribution is given to the creator. The license allows for commercial use.
Article links: DOI: 10.21203/rs.3.rs-5499511/v1 | PubMed: 39678348 | PMC: PMC11643335
Relevance: Moderate: mentioned 3+ times in text
Full text: PDF (7.5 MB)
Decision-making is altered in neuropsychiatric disorders affecting the basal ganglia1. A range of experimental evidence links, in particular, balances between the compartments of the striatum and connected brain regions to decision-making function and dysfunction2–6. Understanding these intricate interactions will be crucial for designing next-generation treatments.
Striatal neurons can be categorized via neurochemistry and connectivity into groups, including the striosome and matrix compartments4,7. Striosomal spiny projection neurons (sSPNs) make up ~10–15% of the striatum and matrix spiny projection neurons (mSPNs) another ~85–90%8,9 (for acronyms, see Table 1). New technologies, including recording and targeting methods and genetically engineered mice, have enabled important new discoveries about differential roles for striosomes and matrix in decision-making3,10–19, including in disorders2,4. Further, both sSPNs and mSPNs belong to either the direct pathway (dsSPNs and dmSPNs), identified by D1 receptor expression, or the indirect pathway (dsSPNs and isSPNs) identified by D2 receptor expression4,20. Notably, dsSPNs, to a greater extent than dmSPNs, project in turn to regions which influence midbrain dopamine release via a subcircuit that is conserved across species8,16,21–24 (Fig. 1a, Table 2). Thus, dsSPNs, isSPNs, dmSPNs, and imSPNs appear to have different physiological roles16,20, raising the possibility they each play a distinct functional role during decision-making.
Table 1:: Terminology
| Term | Definition |
|---|---|
| GPi | Globus pallidus internus |
| GPe | Globus pallidus externus |
| LHb | Lateral habenula |
| RMTg | Rostromedial tegmental nucleus |
| daSNC | dopaminergic neurons of the substantia nigra compacta |
| FSI | Striatal fast-spiking Interneuron |
| sSPN | Striosomal striatal projection neuron |
| mSPN | Matrix striatal projection neuron |
| dsSPN | Direct pathway sSPN |
| isSPN | Indirect pathway sSPN |
| dmSPN | Direct pathway mSPN |
| imSPN | Indirect pathway mSPN |
| BG | Basal ganglia |
| Decision-dimension | An axis of the coordinate system with which SPNs (dsSPNs, isPSNs, dmSPNs, imSPNs) process cortical activity. Subpopulations of SPNs (for each dsSPNs, isPSNs, dmSPNs, imSPNs) encode data along different decision-dimensions. Cortical activity is linearly mapped to this basis of decision-dimensions such that the activity of a single cortical neuron no longer is encoded by a single SPN. Certain decision-dimensions might correspond more predominantly, for instance, to reward level, cost level, or novelty level, as encoded across multiple cortical neurons. Decision- dimensions are modeled as the principal components of cortical activity. |
| Decision-space | The subspace produced by the decision-dimensions which are selected by the circuit to be used during a decision. Decision-space is formed when dopamine releases to mSPNs (dmSPNs and imSPNs), signaling that certain decision-dimensions are important and others are unimportant, and therefore can be excluded from the subspace. |
| Action value | Value assigned by the circuit to an action |
| Inaction value | Value assigned by the circuit for refraining from an action |
| Prediction error | The difference between expected and observed information along a data axis (for instance, reward prediction error, punishment prediction error, or novelty prediction error). |
| Circuit activity | The set of average activities of each circuit element during a decision |
| Advantage | The degree to which a circuit activity is preferred. Used in our analysis of the change in circuit activity between trials. |
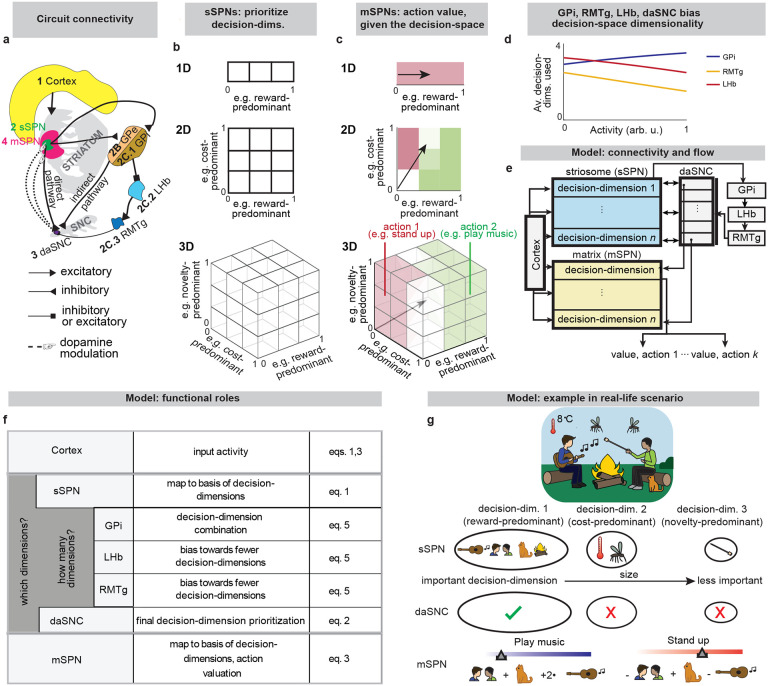
Table 2:: Evidence for the connectivity used in our model.
| Connectivity | Evidence |
|---|---|
| cortexis→SPN/mSPN | |
| cortex→FSI→sSPN/mSPN | |
| sSPN (more so than mSPN)→daSNC | |
| sSPN (more so than mSPN)→GPi→LHb→RMTg→daSNc | |
| sSPN (more so than mSPN)→GPe→daSNC | |
| daSNC→sSPN/mSPN |
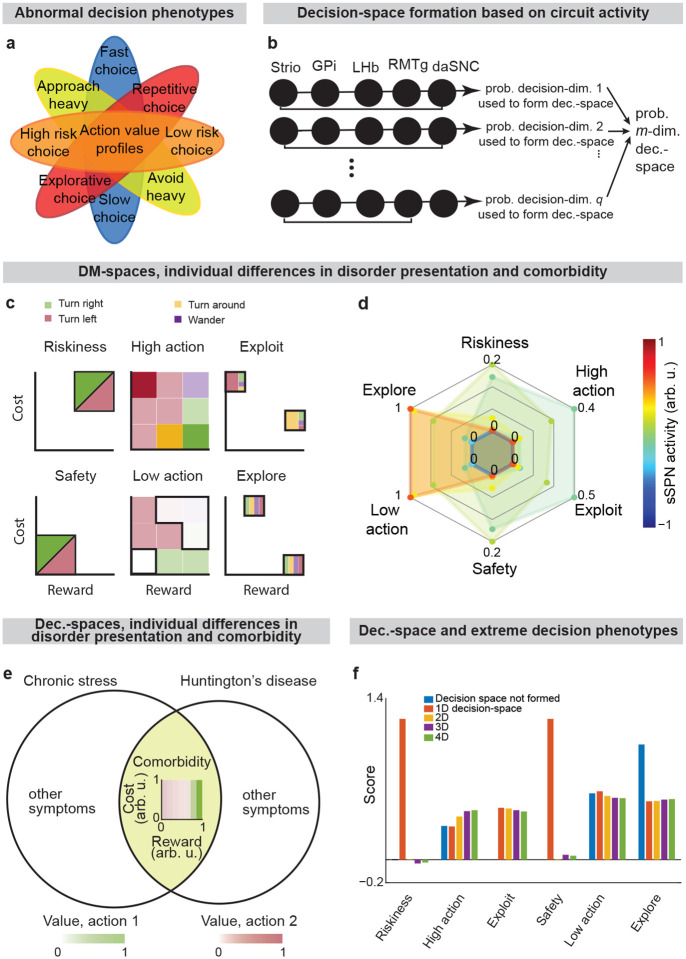
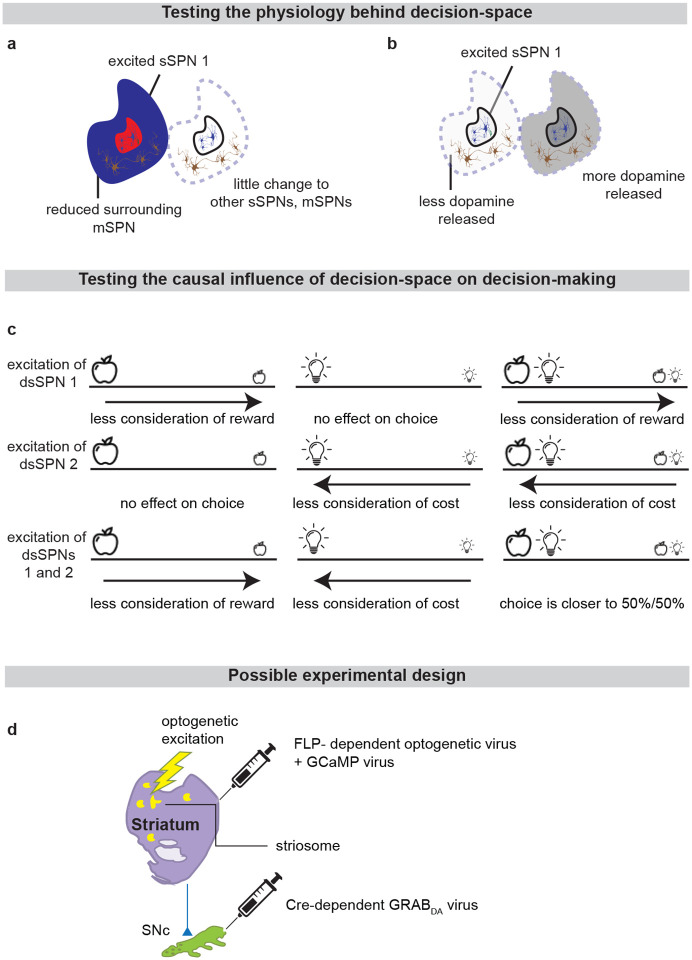
While a range of modeling works explore the roles of direct versus indirect pathway function25, they largely omit the important dimension of striosome versus matrix. The omission of striosomes from models of decision-making or basal ganglia function hinders the interpretation of important features of the striatum because it prevents an accurate depiction of striatal-daSNC interplay, which is primarily striosomal4,24,26 (Table 2). Further, disorders that differentially affect the direct versus indirect pathways have been found to also affect striosomes versus matrix differently2, suggesting that attention to all four compartments is necessary for an accurate understanding of disorder-affected decision-making. To close this gap, we formed a model that accounts for striosome versus matrix subdivisions, including the selective modulation of midbrain dopamine by striosomes (Fig. 1, Extended Data Fig. 1). From our physiological model arises the concept of a “decision-dimension,” our term for an axis along which the modeled circuit encodes information (for terminology, see Table 1). During a decision, decision-dimensions that are important based on the context are selected, forming a mathematical “decision-space.” We present evidence, via our analysis of the neural recordings and our models of findings from the experimental literature, for the core tenets of our model: that subpopulations of SPNs encode information along decision-dimensions, that a decision-space is formed, and that the decision-space adapts based on context (Figs. 2,3, Extended Data Figs. 2–5). Then we demonstrate the power of the model to explain a range of physiological and behavioral phenomena, including RPE and the roles of the indirect/direct pathway (Fig. 4, Extended Data Fig. 6). Finally, we speculate how the model might explain behavioral phenomena observed in psychiatry (Figs. 5,6, Extended Data Figs. 7,8) and suggest future experiments (Extended Data Fig. 9).
Results
Model description.
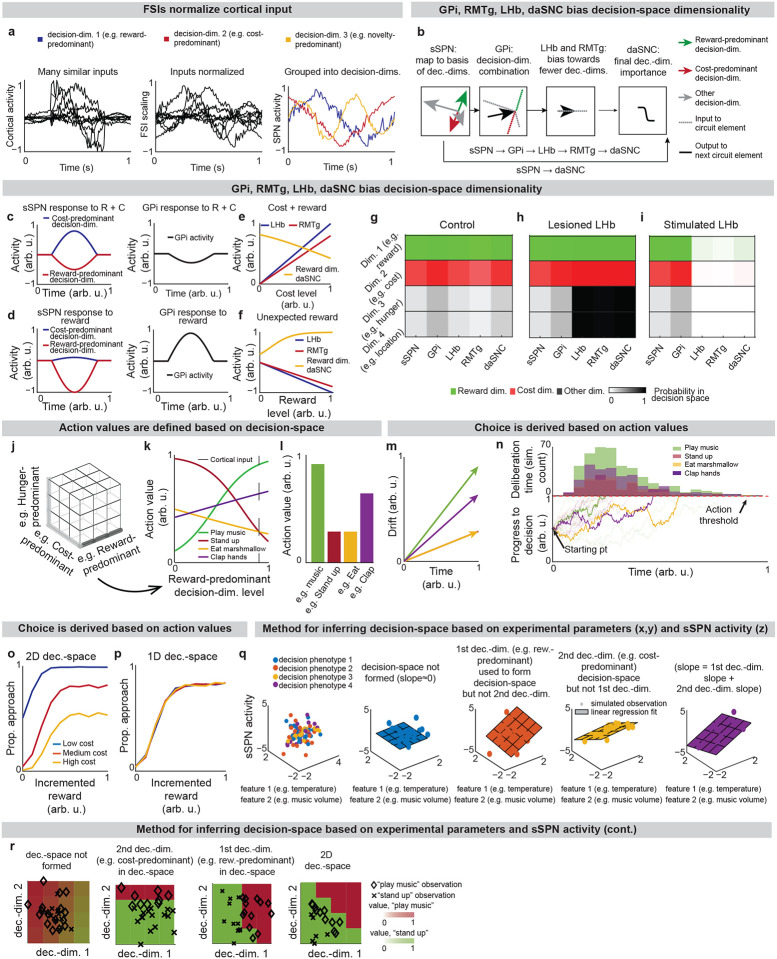
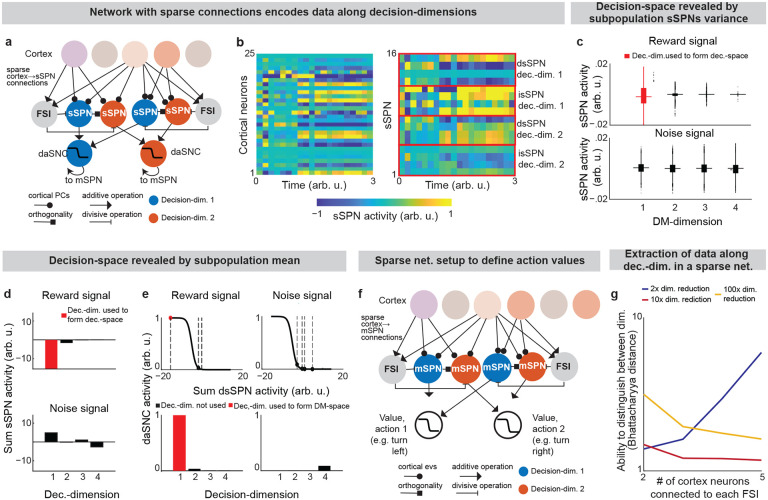
Our model describes how physiological interactions between elements of a striosome-centered circuit inform decision-making. (For extended reasoning behind our choice of circuit elements, see Supplementary Note 1.) Striatum-projecting cortical neurons that encode mixed information serve as the input to the modeled circuit. We assume for the purpose of our model that the information passed from the cortex to each striatal compartment is roughly similar (in actuality, there is much overlap with some differences, see Table 2 and Supplementary Note 2). The cortical neurons synapse on subpopulations of proximate SPNs which have been found to each encode distinct information27. During this process, fast spiking interneurons (FSIs) perform a normalization operation (Extended Data Fig. 1a). Mathematically, we represent this as a matrix dictating cortex-SPN connection weights for each pathway (direct or indirect) mapping cortical activity to the coordinate space of sSPNs, where it undergoes divisive normalization by FSI activity and a shift in activity to form sSPN activity :
Information from sSPNs is then passed to dopaminergic neurons of the substantia nigra compacta (daSNC). daSNC neurons, like sSPNs, have been shown to be organized topologically into subpopulations that each encode distinct information28. Experimental evidence suggests that signals are passed from sSPN subpopulations to daSNC neurons in three ways (see Table 2): A) dsSPN→daSNC. Subpopulations of dsSPNs inhibit daSNC subpopulations directly via dendrite bouquets26. Thus, in our model, each dsSPN subpopulation inhibits a corresponding daSNC subpopulation. B) isSPN→GPe→daSNC. isSPNs send signals to daSNC neurons via GPe16, which inhibit daSNC subpopulations. Thus, in our model, each isSPN subpopulation disinhibits a corresponding daSNC subpopulation. C) sSPN→GPi→LHb→RMTg→daSNC. GPi integrates signals from many sSPN subpopulations through synapses that release both GABA and glutamate29, and the LHb, when activated by GPi, powerfully inhibits multiple of the dopaminergic subpopulations via RMTg30,31. So, in our model, shifts in this pathway lead to a shift across all daSNC subpopulations. Mathematically, we represent the three circuits as daSNC combining activity from sSPNs ssSPN,P (with connection weights corresponding to each decision-dimension and pathway ) with RMTg activity and an additive shift :
In addition to sSPNs, there are subpopulations of mSPNs, termed matrisomes4, that densely surround sSPN subpopulations. In our model, we hypothesize that these sSPN and mSPN subpopulations communicate with one another via dopamine release from the corresponding daSNC subpopulation (there are other sSPN→mSPN connections which we do not model that play more local roles, see Supplementary Note 3). There are multiple groups of these functionally connected sSPNs, daSNCs, and mSPNs. In the model, when a daSNC subpopulation is active, dopamine is released to the corresponding sSPN and mSPN subpopulations, resulting in enhanced or inhibited mSPN reception of cortical signal among the subpopulations, as shown in experimental work32. Mathematically, mSPN activity is defined similarly to sSPN activity, but for a diagonal matrix corresponding to the dopamine release that probabilistically defines the decision-space, with :
mSPNs have been found to be primarily involved in motor functions, projecting to the GPi, SNr, and then to brainstem motor programs33. SPN activity (which is predominantly mSPN) has been shown to contribute to action selection and initiation34. The direct pathway has generally been implicated in promoting actions and the indirect pathway in preventing actions35. So, in our model, the output of the circuit is the definition of action values (the value of performing various actions, encoded by the direct pathway) and inaction values (the value of refraining from those actions, encoded by the indirect pathway) by mSPN signals on route to downstream regions. Mathematically, values (for each action/inaction , pathway ) are defined based on mSPN activity, internal coefficients , and priors (which are set to arbitrary values that are constant across analyses, see Common parameters, Methods):
Based on these values, actions are either performed or refrained from over time. We model this using a Merton process model where the first process to hit a threshold is enacted (direct pathway) or refrained from (indirect pathway). See Defining choice, Methods.
Thus, our model is constructed based on the anatomy and physiology of the striosome-centered circuit. The physiological description also produces a simple and convenient geometric interpretation. If we let each SPN and daSNC subpopulation encode the principal components of cortical activity, such as could be learned via a modified Oja’s rule36, then the columns of become orthogonal. So, each SPN subpopulation can be thought of as encoding information along an axis of Euclidean space. We term these axes “decision-dimensions” and have evidence that they correspond to constructs such as reward, cost, or novelty (discussed in more detail below). We suggest that when dopamine is released to SPNs, selectively enhancing reception of cortical signal, decision-dimensions are effectively prioritized. Therefore, cortex, striosomes, and dopamine work together to form a “decision-space”, only focusing on decision-dimensions that are necessary based on context (Figs. 1b,c). In this light, sSPN and daSNC, via pairs of connected subpopulations that process information in parallel, serve the functional role of selecting which decision-dimensions should receive high priority. In particular, dsSPNs determine which decision-dimensions to use in the direct pathway, and isSPNs which decision-dimensions to use in the indirect pathway. On the other hand, GPi, LHb, and RMTg, by prioritizing or deprioritizing all daSNC subpopulations together, determine how many decision-dimensions should be prioritized (Fig. 1d, Extended Data Figs. 1b–i).
Importantly, the advantage of this formation is not only conceptual, but practical for linking physiology to decision-making (Figs. 1e–g, Extended Data Figs. 1j–p). Distinct sSPN and/or daSNC subpopulations have been found to encode, for instance, reward12,14,15,28, cost12,14,15,28, or novelty37. Thus, we might imagine that decision-dimensions could correspond loosely to reward level, cost level, or novelty level. If this is the case, a logical prediction of the model arises: we would expect a low-dimensional decision-space to be formed during a simple choice (e.g. between two rewards) and a high-dimensional decision-space to be formed during a more difficult choice (e.g. between offers which each have benefits and costs that must be weighed in order to solve the problem). This hypothesis, if proven, would allow us to infer the decision-spaces of behaving rodents or humans simply by regressing sSPN activity on experimental parameters (for example, temperature or music volume), as we demonstrate using synthetic data (Extended Data Figs. 1q,r). For example, a significant correlation between sSPN activity and novelty level would indicate the existence of decision-dimension that corresponds roughly to novelty (Inferring decision-space from SPN activity and choice, Methods). We sought to determine if this hypothesis is supported by experimental physiological data collected during decision-making.
Support for the model: context-dependent sSPN physiology matches model predictions.
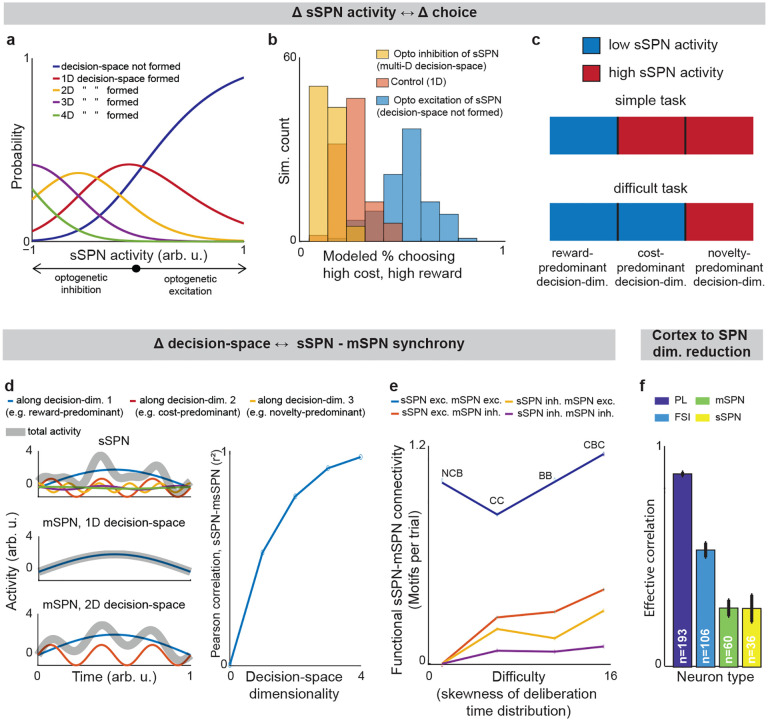
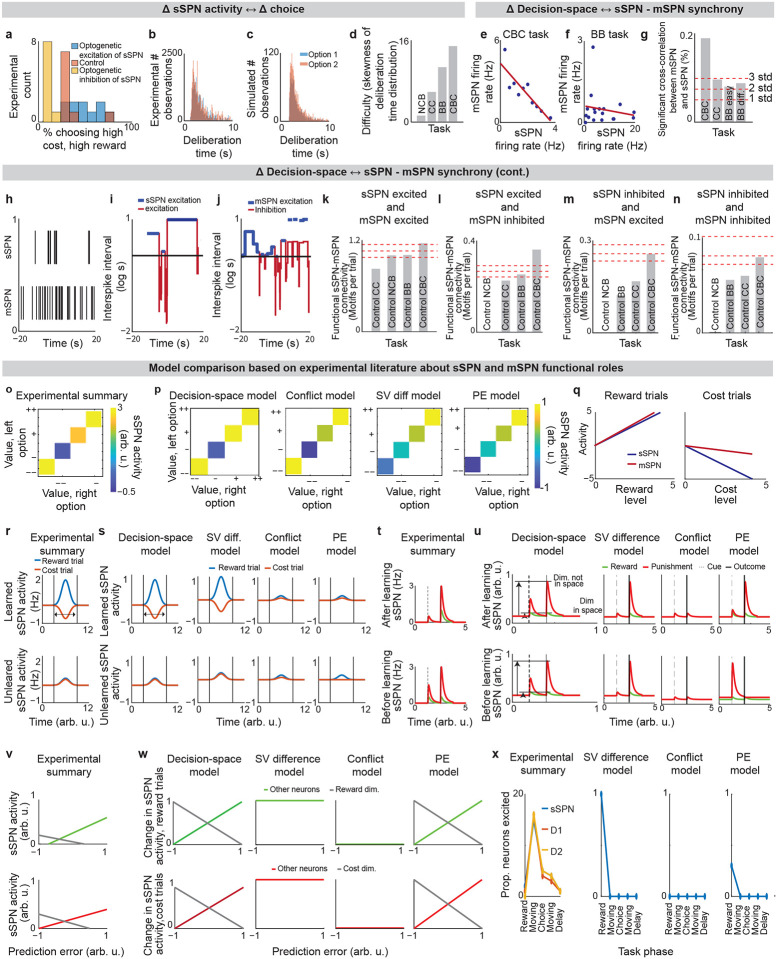
We began by asking whether the results of the physiological sSPN literature support a link between the decision-space (which our model postulates is driven by sSPN activity) and task difficulty (inferred from experimental inputs, for instance a simple task with reward only versus a difficult task with conflicting rewards and costs). We began with the experimental literature on sSPNs. In one experiment11, sSPNs were optogenetically stimulated (or inhibited) during a rodent conflict decision-making task. Per our model, this should cause inhibition (or disinhibition) of daSNC neurons, leading to reduced (or enhanced) dopamine release to SPNs, producing a lower- or (higher-) dimensional decision-space. Indeed, the stimulation led to choices indicative of decision-making using few informational dimensions, while inhibition led to choices indicative of decision-making using multiple informational dimensions (Figs. 2a,b, Extended Data Fig. 2a, Effect of sSPN activity on decision-space and Effect of decision-space on choice, Methods). A second experiment11 tested, reversely, the activity of striosomes during simple versus difficult tasks. As the model expects, striosomal activity during simple tasks resembled the levels observed during optogenetic excitation, whereas striosomal activity during difficult tasks resembled the levels observed during optogenetic inhibition (Fig. 2c, Extended Data Figs. 2b–d). In another study, striosome activity was lower among rodents that learned a difficult reversal learning task than among rodents that did not10. Thus, there appears to be a relationship between sSPN physiology and task difficulty in the direction expected by our model.
Support for the model: context-dependent sSPN-mSPN synchrony matches model predictions.
Importantly, the result above does not distinguish our model from the alternative, simpler explanation that sSPNs might collectively encode task difficulty. We could term such a model a “conflict model,” where sSPN activity tracks the overall conflict present in a task (see Table 3). To determine whether it was changes to sSPN subpopulations driving the overall change in sSPN activity (i.e. decision-space), rather than a general effect, we analyzed the paired activities of sSPNs during simple versus difficult tasks using the Corticostriosomal Circuit Stress Experimental database3. We hypothesized that a greater number of sSPNs and mSPNs would be functionally connected during difficult tasks. This prediction arises from the circuit connectivity in our model, where sSPNs are functionally connected to mSPNs via daSNC, and each additional prioritized subpopulation causes more sSPN→daSNC→mSPN modulation (Fig. 2d). This could be observed, for instance, as an increase in striosome and matrix correlation as decision-space increases. This hypothesis is also inspired by the observation that striosome and matrix activity has been found to roughly track one another over time in a difficult task13.
Table 3:: Criteria used in Tables 4–8 to test our decision-space model and compare it to alternative models of the circuit.
| Modeled Functional role of a circuit region. | Expectation per the model. |
|---|---|
| sSPNs influence the priority of decision-dimensions, thereby affecting decision-space.(decision-space model) | 1.A. During a high-dimensional decision-space, choice more closely aligns to experimental inputs (e.g. chocolate milk level, light brightness) during difficult tasks (e.g. consideration of both rewards and costs). |
| 1.B. A low-dimensional decision-space is often formed from decision-dimensions which are commonly used during a decision-making task, for instance information about reward in rodents that are trained to respond to reward cues. | |
| 1.C. Cortical data is mapped orthogonally and continuously onto SPNs. | |
| sSPNs encode conflict.(our conflict model) | 2.A. sSPN activity scales with conflict between two features like reward and cost. |
| 2.B. Changes to conflict, revealed by sSPN activity, alter choice. | |
| 2.C. Changes to sSPN signals are greatest when conflict is introduced. | |
| sSPNs encode subjective values.(our subjective value model) | 3.A. sSPN activity scales (possibly directly or inversely) with subjective value of stimuli, likely roughly tracking reward minus cost. |
| 3.B. Higher subjective value, reflected by sSPN activity, leads to increased selection of an offer. | |
| 3.C. Changes to sSPN activity are strongest at the time during scenarios where cues are associated with subjective values. | |
| sSPNs encode prediction errors.(our prediction error model) | 4.A. sSPN activity should change proportionally and continuously to the difference between expected and actual reward and cost at each time step. |
| 4.B. As a task is learned, the trend in sSPN activity over time changes as earlier cues become associated with later outcomes. | |
| sSPNs encode actions.(our actions model) | 5.A. Different neurons would encode different actions |
| 5.B. Activity of sSPN subpopulations should scale directly or inversely with the predisposition of action execution. | |
| 5.C. Changes to the signals of action-encoding subpopulation would be greatest prior to or during action execution. | |
| GPi during decision-space formation.(decision-space model) | 6.A. Changes to activity along a decision-dimension is reflected in GPi activity. |
| 6.B. A given GPi neuron encodes data across multiple decision-dimensions. | |
| 6.C. Activation of GPi causes more decision-dimensions to be incorporated into the decision-space while inactivation causes fewer decision-dimensions to be used. | |
| LHb and RMTg optimizing or modifying decision-space.(decision-space model) | 7.A. Active LHb (or RMTg) leads to choice reflective of reduction in dimensionality of the decision-space and vice versa. |
| 7.B. LHb (or RMTg) is active during times when lower-dimensional decision-spaces are beneficial to decision-making. | |
| Dopaminergic neurons of the SNc during decision-space formation.(decision-space model) | 8.A. There exist subpopulations of daSNC neurons that encode information along an orthogonal axis of information. |
| 8.B. Activity in one daSNC subpopulation only affects the subpopulation of SPNs corresponding to one decision-dimension. | |
| Direct and indirect pathways alter decision-space formation.(decision-space model) | 9.A. Higher dopamine leads to lower dimensionality of the direct pathway decision-space while lower dopamine leads to higher dimensionality, and vice versa for the indirect pathway. |
| 9.B. Direct pathway mSPNs promote actions while indirect pathway mSPNs aid in action suppression. | |
| 9.C. Subpopulations of mSPNs encode data along decision-dimensions orthogonally and continuously over time. |
To test this, we analyzed the synchrony between striosome neurons and matrix neurons during simple decisions (that forced choice between either two rewards or two costs) and during difficult decisions (that forced choice between offers that contained both rewards and costs together). We measured synchrony as the cross-correlation between sSPN activity and mSPN activity over the period of the task. To control for possible physiological differences across the phases of the rats’ movements, we also developed a custom Granger causality-based tool. Per both metrics, synchrony was significantly higher in the difficult task. Further, synchrony scaled with the difficulty with which the rats treated the task, as measured based on deliberation time (Fig. 2e, Extended Data Figs. 2e–n, Connected SPNs through cross-correlation and Connected SPNs through Granger causality, Methods). Thus, rather than sSPNs encoding conflict in their general level of activity, there appears to be an important relationship between sSPN and mSPN subpopulations during high-conflict tasks. Our analysis does not confirm that sSPNs have a causal effect on mSPNs. However, if this is assumed, the evidence suggests enhanced modulation of mSPN subpopulations by sSPN subpopulations during the difficult tasks, that is, a formation of a higher-dimensional decision-space.
Support for the model: Dimensionality reduction from the cortex to SPNs.
Notably, the synchrony analysis above demonstrates the use of subpopulations during context-dependent decision-making, but it does not test whether those subpopulations correspond to decision-dimensions. There is, however, evidence that SPNs encode what we term decision-dimensions. Experimental work has demonstrated that distinct SPN subpopulations encode different information, and that these subpopulations persist across days27. Further, our analysis suggests that dimensionality reduction occurs from the cortex to SPNs, as it does in our model during the mapping of information to a basis of decision-dimensions. Cortical neurons had the most coordinated activities over time (measured as effective correlation38), then FSIs, followed by sSPNs and mSPNs (Fig. 2f, Analyzing neural dimensionality reduction, Methods). This would suggest a higher-dimensional representation in cortex, where neurons encode similar information over time, than in the downstream regions, where information is compactly encoded in neurons that behave differently over time. Thus, it seems that cortex→SPN dimensionality reduction occurred during the tasks, like in our model the mapping from high-dimensional cortical information to a basis of SPN decision-dimensions.
Support for the model: Strong alignment to the experimental literature on SPNs compared to alternative models.
The analysis to this point is based on a selection of the experimental literature relating sSPN activity to mSPN activity and choice. We wished to test the model more broadly using a range of experimental results. To this end, we devised five tests that link sSPN activity to choice, each verifiable with the experimental literature, that might support or reject the decision-space model (Extended Data Figs. 2o–x, Table 3). As benchmarks, we also constructed, from roles commonly assigned to sSPNs, four alternative models in which sSPNs encode 1) conflict, 2) subjective value, 3) prediction error, or 4) actions. We found that while the alternative models each can be used to interpret a subset of the experimental evidence, only the decision-space model aligned to the breadth of it (Table 4). A selection of experimental studies on GPi, LHb, and daSNC also align with the decision-space model (see Tables 5–8), thus offering a new lens through which to interpret their functions.
Table 5:: Testing the alignment of the decision-space model to a selection of the experimental literature on GPi.
| Criterion | Weglage et al. (2022)73 (I) | Munte et al. (2017)78 (II) | Stephenson-Jones et. al (2016)79 (III) |
|---|---|---|---|
| 6.A | ∅ | ✓ | ∅ |
| 6.B | ∅ | ∅ | ✓ |
| 6.C | ✓ | ∅ | ∅ |
✓ — aligned with criterion
≈ — somewhat aligned to criterion
✘ — not aligned with criterion
∅ — experiment does not test criterion
6.A.II. Level of reward correlated with GPi activity. This may suggest that GPi activity scales up or down depending on the level of information along a reward-related decision-dimension.
6.B.III. Individual LHb-projecting GPi neurons were both excited by punishment-predicting cues and the punishment itself and were inhibited by rewards and their associated cues. This may suggest that information along two decision-dimensions, one reward-related and one-cost related, is encoded by the same GPi neurons. Further, the opposite response of GPi neurons to reward and cost lends support for our choice to differentially weight GPi inputs from sSPN subpopulations corresponding to different decision-dimensions.
6.C.I. An identified subpopulation of LHb-projecting GPi affected the profile of choices made, aligning with the functional role of the GPi in our model. Decreased activity of these neurons was associated with increased commitment to actions, which may correspond to effective formation of a decision-space. This would align with the hypothesis of the model that lower GPi activity leads to a higher-dimensional decision-space.
Inability to form a high-dimensional decision-space in disorders.
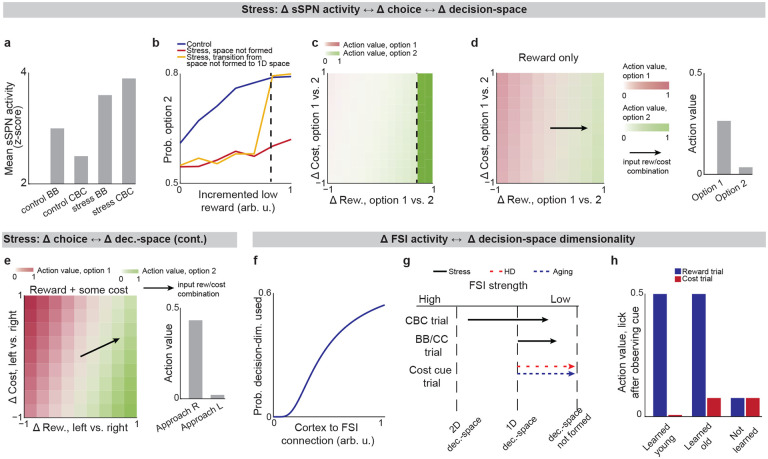
We next applied our model to the experimental literature on neuropsychiatric disorders, wondering if it could offer insight into disorders associated with sSPN changes. Interestingly, in an experimental study on chronic stress3, sSPNs were hyperactive compared to controls during the most difficult task but less affected during the simpler tasks (Fig. 3a). Meanwhile, the rodents were less adherent to reward level only in the difficult task, suggesting dysfunction in processing reward and cost together (Figs. 3b,c, Effect of decision-space on choice, Methods). Thus, we wondered if post-stress sSPN hyperactivity could cause altered choices due to a reduction in the dimensionality of the decision-space, similar to the model in Fig. 2b where optogenetic excitation reduced the decision-space in controls.
Our analysis supports this. Animals made decisions in the cost-benefit conflict task more quickly after stress, as if the task were less difficult (Extended Data Figs. 3a–d, Defining decision difficulty by task, Methods). Meanwhile, after stress, choices involving both reward and cost no longer had more functionally connected sSPNs and mSPNs than the simple tasks, suggesting a change to the decision-space as well as a general shift in activity. In fact, synchrony was similar across tasks and to the simple tasks for controls (Extended Data Figs. 3e–i). Thus, after stress, the rodents both showed both neural signatures aligned with a low-dimensional decision-space and choices aligned with processing of information in a low-dimensional way.
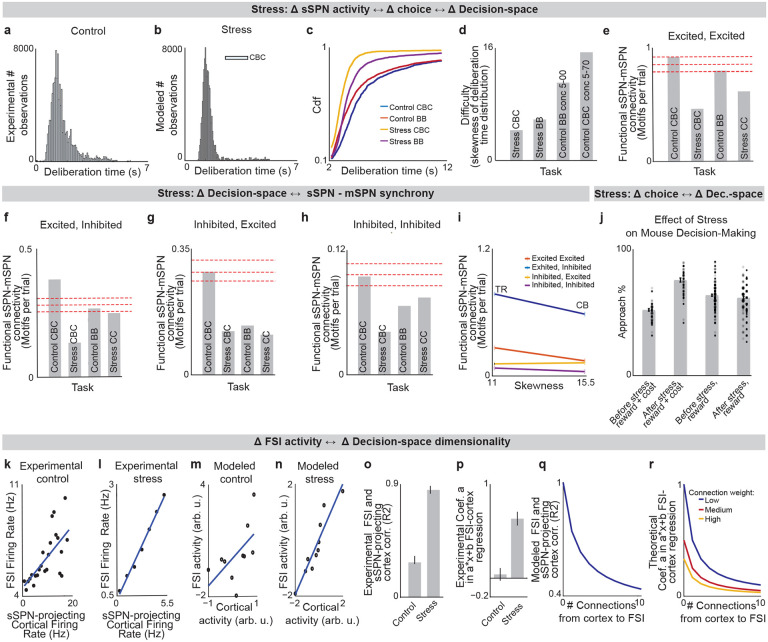
The inability to form a high-dimensional decision-space can also explain a counterintuitive finding that stress causes rodents to prefer a reward-cost combination over a reward presented without cost (Extended Data Fig. 3j, Changes to choice after adding cost to a reward offer, Methods). In classic economic theory, the addition of cost to a reward typically makes a good less attractive39 and thus our observation cannot be readily explained. In contrast, the decision-space model offers a simple explanation: cost can increase offer attractiveness in instances where cost level causes a transition from a default low-dimensional decision-space to a higher-dimensional decision-space, as we hypothesized is the case after stress in Figs. 3b,c. In these cases, the rules encoded by mSPNs can assign a higher value to accepting versus avoiding an offer when reward and cost are considered rather than only reward (Figs. 3d,e). In other cases, decisions are predicted to resemble those predicted by classic theory, for example in cases where either a one-dimensional (as in Fig. 3d) or two-dimensional (Fig. 3e) decision-space is used across cost levels.
Cortex→FSI connectivity is impacted by chronic stress3 (Extended Data Figs. 3k–r, Analyzed cortex-FSI connectivity and Modeled cortex-FSI connectivity after stress, Methods), leading to hyperactive sSPNs. Our model suggests that this causes the formation of a lower-dimensional decision-space (Figs. 3f,g), which would lead to lower variance and higher mean of SPN subpopulations (Extended Data Fig. 4). Notably, the cortex→FSI connection is also impacted in Huntington’s disease and aged rodents10, raising the possibility that an inability to form a high-dimensional decision-space during difficult decisions is a feature of multiple health conditions. Supporting this hypothesis, a model where Huntington’s disease and aged subjects use a lower-dimensional decision-space produces action values that follow the trend of experimental choice (Fig. 3h, Effect of decision-space on choice, Methods).
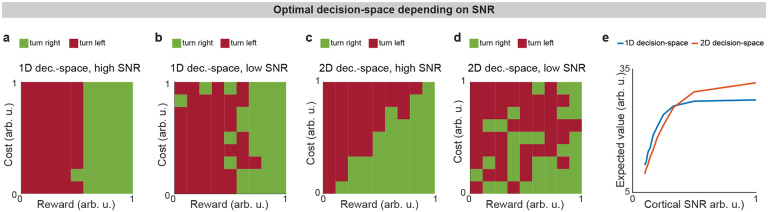
Interestingly, our model expects low dimensional decision-space to be beneficial in disorder conditions. A feature of chronic stress3 and schizophrenia40 is reduced cortical signal-to-noise ratio (SNR) and disrupted cortical signaling. In these conditions, a low dimensional decision-space is theoretically optimal because only the highest-priority decision-dimensions carry enough signal to outweigh the drawback of noise (Extended Data Fig. 5, Effect of cortical SNR on choice, Methods). Our analysis raises the possibility that FSIs help steer the circuit towards a helpful decision-space in disorders.
Functional roles of dsSPNs, isSPNs, dmSPNs, and imSPNs.
sSPNs and mSPNs have been found to be distributed between the direct and indirect pathways of the striatum24,41,42, with dopamine release differently affecting sSPNs versus mSPNs and also dSPNs versus iSPNs20,32,43. Various neuropsychiatric disorders are associated with disturbed sSPN versus mSPN2 and direct versus indirect pathway balances44. Thus, the compartments likely play distinct functional roles in decision-making, including in disorders.
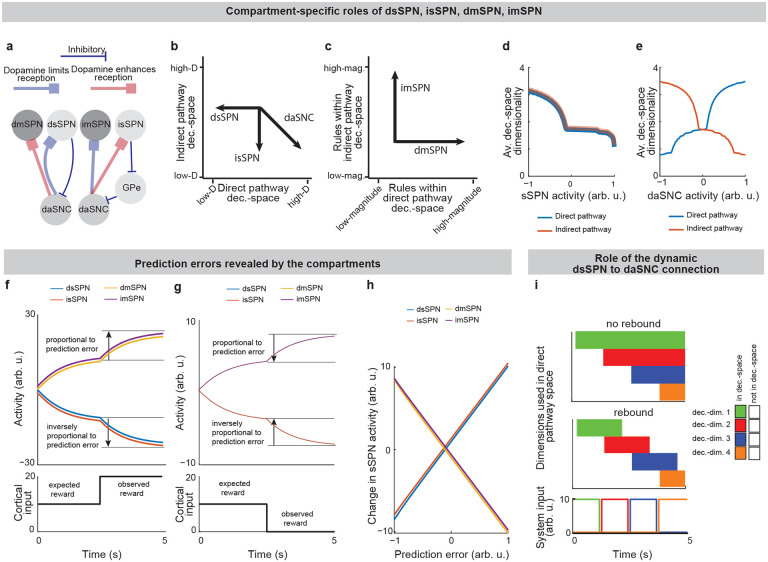
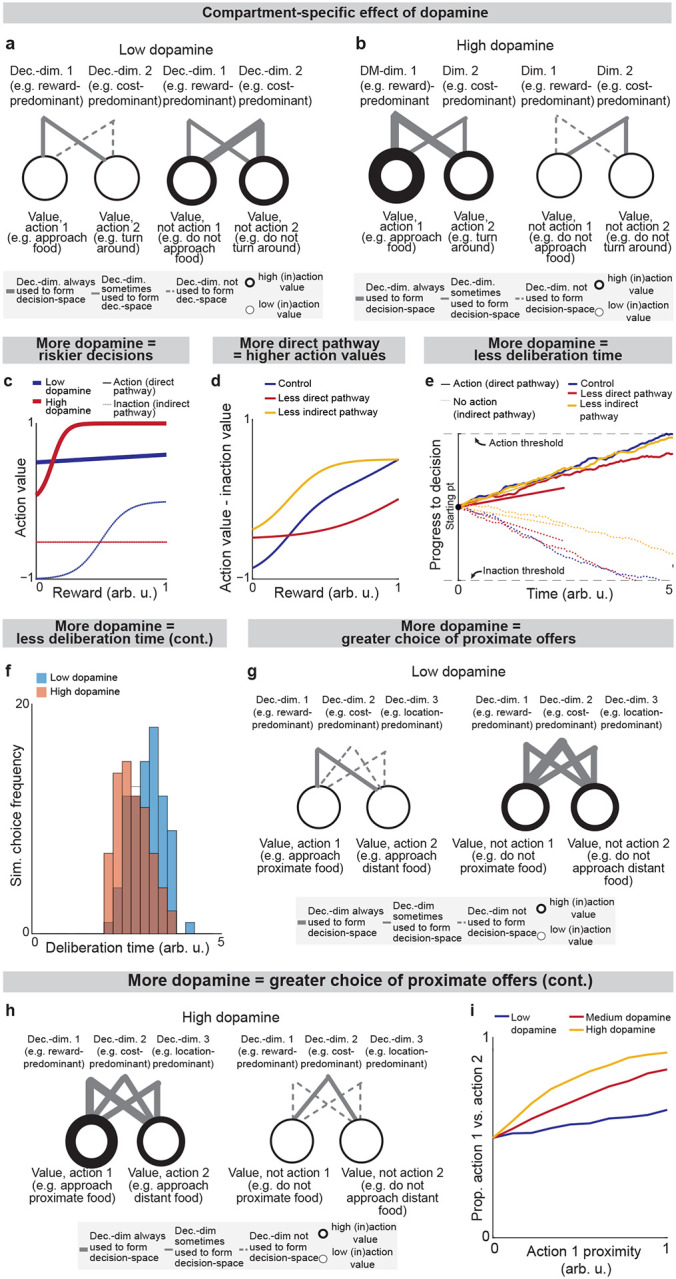
Because dsSPNs connect to dmSPNs through daSNC in our model, and isSPNs to imSPNs through GPe and daSNC, two decision-spaces are formed in parallel, one related to each pathway (Fig. 4a). Thus, based on the functional roles we assign the pathways, the circuit uses a direct pathway decision-space to determine whether to perform an action and an indirect pathway decision-space to determine whether to refrain from it. dsSPNs influence the direct pathway decision-space while isSPNs influence the indirect pathway decision-space, and dmSPNs promote actions while imSPNs discourage actions (Figs. 4a–e, Modeling time-variant input, Methods). The circuit uses these compartment-specific mechanisms to calculate which actions should be performed with one set of decision-dimensions and calculate which actions should not be performed with another. The answers to these questions might overlap. For instance, the direct-pathway and indirect-pathway space should provide divergent answers to the value of consuming cocaine based on the dimensions they prioritized; the former, focusing on reward, might assign it great value while the latter, focusing on cost, might assign great value to not consuming it. When balances between the direct versus indirect pathways and striosome versus matrix changes, this calculus changes. For example, it has been found that dopamine release is enhanced to sSPNs versus mSPNs after cocaine administration45 and simultaneously dSPNs are enhanced in the short-term and iSPNs over longer-term scales of time46. This might lead to a high-dimensional direct-pathway decision-space but pruning of ordinarily important decision-dimensions from the indirect pathway decision-space, producing a reduction of nuance in determining when to avoid actions and heightened impulsivity.
Indeed, the model’s interpretation of the two parallel decision-spaces offers an intuitive explanation for a range of experimental observations on the direct versus indirect pathway. For example, our model replicates the experimental observations that increased dopamine leads to riskier and quicker decisions and preference for nearby offers, in time or physical proximity, compared to distant offers (Extended Data Fig. 6, Table 8, Effect of dopamine on action/inaction values and Effect of decision-dimensions on choice, Methods).
Prediction error encoding is an emergent property of the model.
A range of experimental studies have shown that SPN activities track prediction errors12,47. This observation has led to hypotheses that SPNs encode a function related to prediction errors in a reinforcement learning framework12. The decision-space model offers a different explanation. In our model, the weights from cortical neurons to SPNs naturally separate cortical information by their associations. For instance, if a bell tends to sound when a subject drinks chocolate milk, both stimuli, even if they arrive from different cortical sources, will likely be mapped to the same reward-related decision-dimension as synapses adjust per Oja’s rule. Less reliable cues are expected to develop mappings with smaller weights. Therefore, the activities of reward-related SPNs may rise when cues predicting rewards appear and fall when cues predicting less reward appear. A sudden change to reward information, for instance a predictive cue, should thus lead to a sudden change in the activity of an SPN subpopulation related to reward, and this change in activity should resemble a prediction error (Figs. 4f–h). Thus, in contrast with the more traditional interpretation where SPNs internally encode a temporal difference value function, the decision-space model suggests that the mapping of cortical activity to the basis of striatal decision-dimensions is sufficient to track prediction error in many cases, without additional computational work performed by the SPNs.
Both interpretations can be used to explain much of the experimental evidence, although the interpretation of the decision-space model may more closely align to recent experimental data (Tables 3,4). Our model also may provide a functional rational for the observation that separate SPNs encode data along different informational axes12,14. In fact, our model expects more of these axes to be uncovered by future work. We might expect, for instance, a cue predictive of the novelty of an object to produce an immediate change in activity of an SPN subpopulation related to novelty.
How might the circuit respond in cases where new information diverges sharply from expectations? Our model predicts that in these cases, sSPNs will signal to daSNC that a decision-dimension should be reprioritized, effectively adding or removing the dimension from the decision-space. Interestingly, the circuit has an inherent physiological mechanism to quickly transition away from a decision-space that is no longer optimal. Experimental evidence has identified rebounds in daSNC activity21 and striatal dopamine release18 after sSPN optogenetic stimulation. Our model suggests that these observations are part of a system by which the circuit can rapidly de-prioritize a decision-dimension after a negative prediction error (e.g. less reward than expected). Thus, the circuit is able to quickly shift to a more helpful decision-space (Fig. 4i).
Discussion
We found evidence, through the experimental literature and our analysis of neural recordings, to support our hypothesis that modeled physiological patterns in SPN activity (the decision-space) can be used to predict patterns in decision-making, and vice versa. This supports our model of the roles of striosomes and matrix neurons of the direct and indirect pathways in context-dependent decision-making.
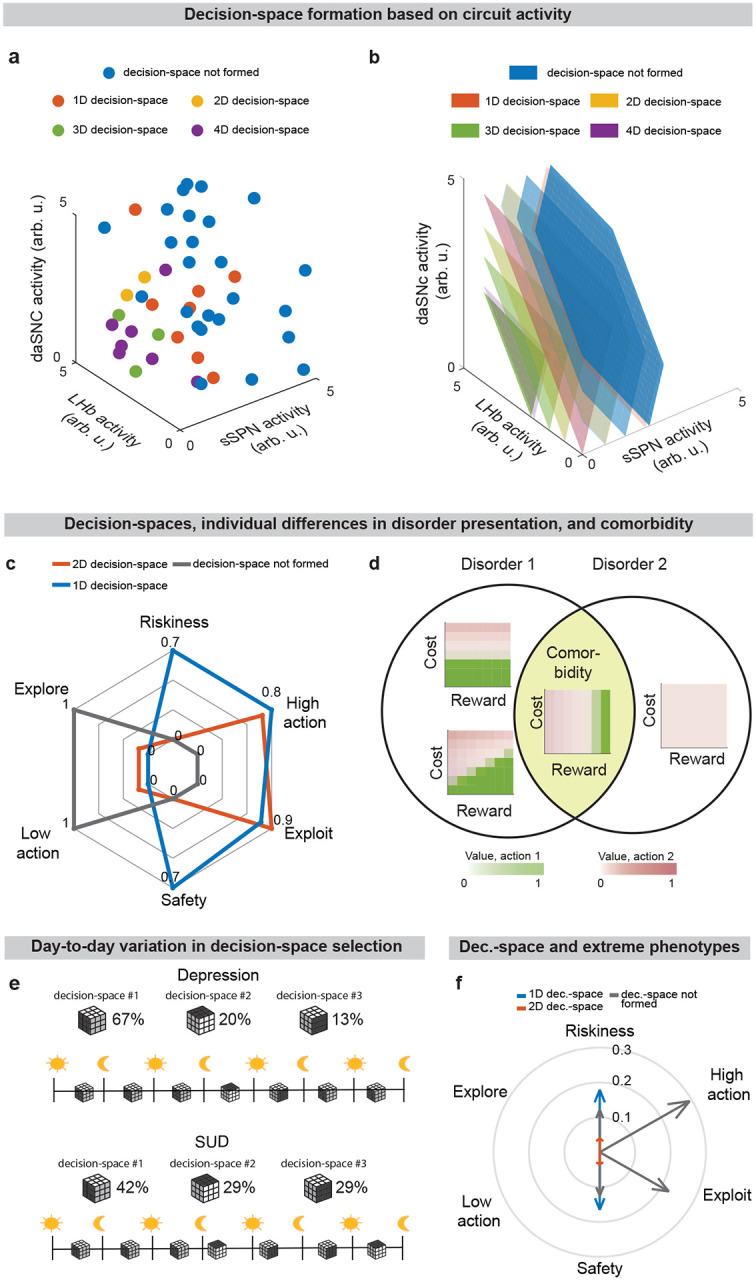
Due to the circuit’s important role in decision-making processes including in neuropsychiatric disorders, our model provides a framework with which to study decision-making phenomena commonly observed in psychiatry. An important prediction of our model is variance in context-dependent decisions, between individuals and over time (Figs. 5a–e, Extended Data Fig. 7a–e, Effects of sSPN, LHb, and daSNC activity on choice profiles, Methods). Individual differences in decision-making as a function of disorders, as seen in the experimental literature48–50, could arise in cases where there are slight differences in activity of the circuit we model, leading to similar decision-making phenotypes only when a similar decision-space is formed. Daily variance in decision-making, a common observation in pscyhology51, could arise from daily variance in circuit activity, causing daily variance in the decision-spaces formed most often. Further, differences in circuit activity may explain the established inter-individual differences in the severity of psychiatric disorder symptoms observed during decision-making52,53. Individual differences in disorder susceptibility could arise from reliance upon or avoidance of a decision-space that leads to extreme decision-making tendencies (e.g. extremely action-heavy, extremely risk-averse) when combined with abnormal action value rules in mSPNs (Fig. 5f and Extended Data Fig. 7f).
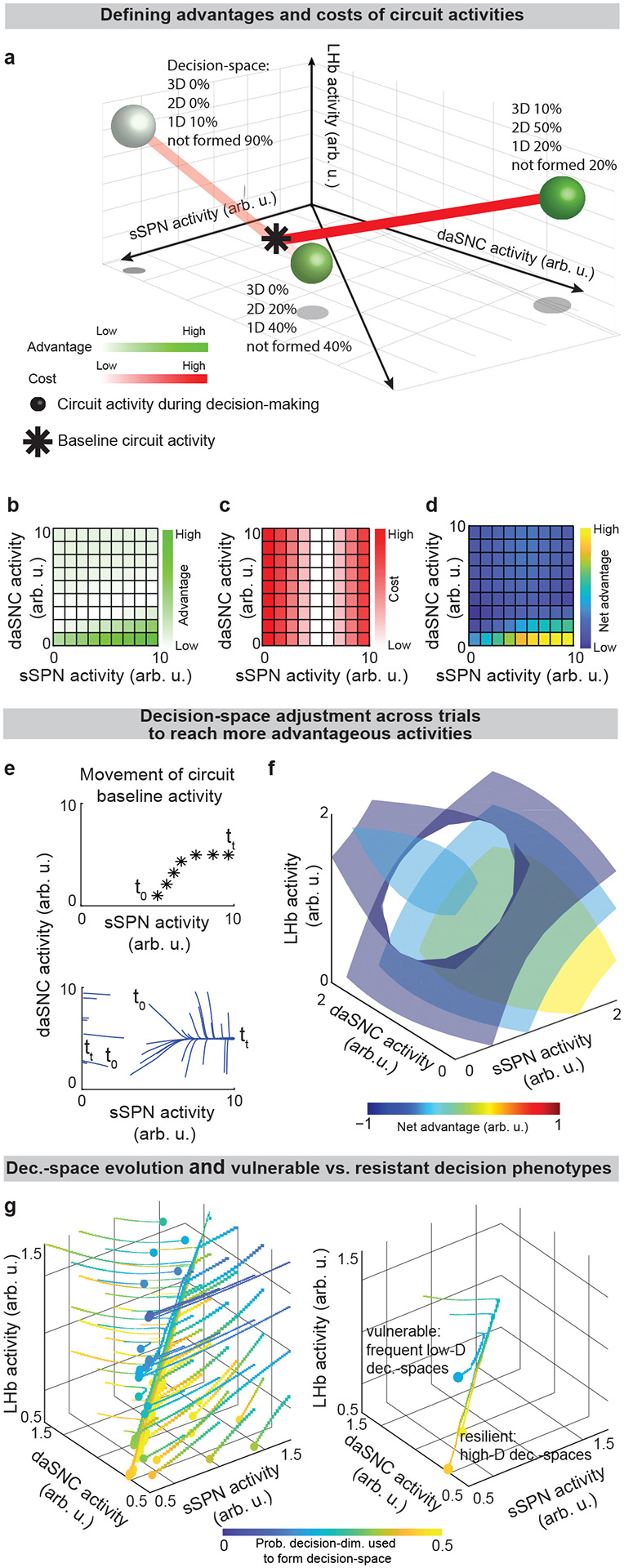
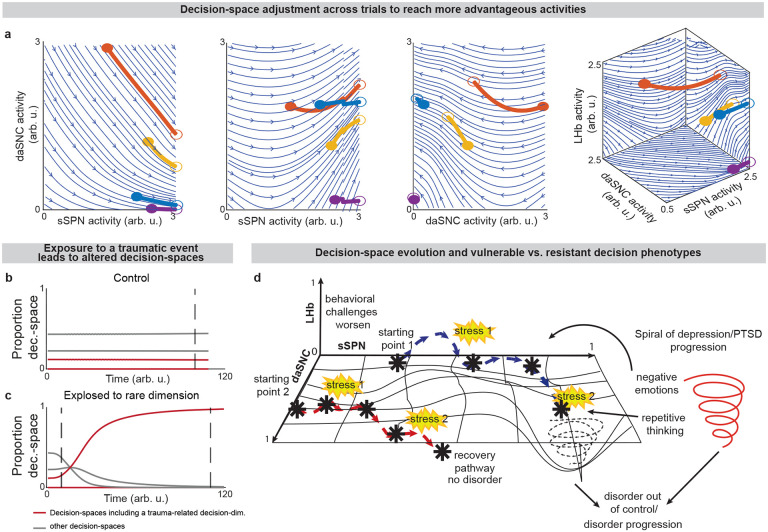
Further, our model serves as a framework for forming hypotheses about changes to the circuit across days and weeks, including during neuropsychiatric disorder progression. Our model expects the circuit to adapt between trials as it adjusts to more frequently form a preferred decision-space (Figs. 6a–f, Effect of initial circuit activity on future trials, Methods). So, vulnerability or resilience to disorders can be framed as an adaptation that is favorable (e.g., to adeptly form a high-dimensional decision-space) or maladaptive (e.g., to only form a low-dimensional decision-space, regardless of decision context). Depending on its initial activity, a modeled circuit can adapt to reach very different activity, leading to disposition to either a high- or low-dimensional decision-space (Fig. 6g and Extended Data Fig. 8a). Thus, differences in the circuit before exposure to a traumatic event, for instance, may explain why two subjects that encounter the same traumatic event do not always develop the aberrant decision-making symptoms of PTSD54. It may also shed light on the neural processes underlying incubation of fear55 and incubation of craving56, where disorders progress over the span of weeks or months, even when the traumatic event or addictive substance does not reappear (Extended Data Figs. 8b–d, Effect of altered advantage score on future trials, Methods). By representing the role of SPNs in a compartment-specific way, our model facilitates understanding of disorders that affect striosomes and matrix differentially.
Our model adds detail to the predictions made by a range of other models of basal ganglia function. For example, a range of other models consider how the direct and indirect pathways of the basal ganglia interact to moderate action selection25,57. Our model offers a simple explanation for several experimental phenomena that are used to fit other models by differentiating the roles of striosomes versus matrix (Extended Fig. 6). In particular, direct/indirect pathway models more commonly fit data from simple reward and/or cost tasks rather than highly difficult tasks, and our model allows for scaling to difficult tasks with many informational dimensions. Another class of model examines how RPE signals facilitate adaptability58–60. Our model does not preclude the possibility that SPNs and/or dopamine may encode an algorithm similar to those used in reinforcement learning. However, our model expects that such an algorithm would function within the framework of the decision-space, perhaps by encoding a separate reinforcement learning value function along each decision-dimension, instead of a traditional model that learns only the discounted sum of future rewards. Exploring this possibility would add depth to current studies of reinforcement learning in the basal ganglia. Our model can additionally be used to expand on other models that explore the role of basal ganglia pathways in performing dimensionality reduction61 or responding to events as sequences that influence each successive action62,63. In each of these cases, our model adds detail on the functional process by which the basal ganglia prepares for a decision based on context. It also clarifies the mechanisms by which the compartments of the striatum process information, led by striosomal influence on dopamine.
Our model carries several limitations, including its limited focus on a dorsomedial striosomal circuit and certain physiological assumptions (Supplemental Notes 1–4). We limit our focus to a specific circuit that has been implicated in decision-making, rather than attempt a unifying theory of basal ganglia function or decision-making encoding across the brain (Supplemental Note 1). While we demonstrate alignment to the existing experimental evidence in Figs. 2 and 3 and Tables 3–8, future experiments (outlined in Extended Data Fig. 9) will be required to confirm the several assumptions we make. Despite these limitations, our model has demonstrated success in relating neural activity to decision-making across a range of behavioral tasks and has the power to explain a range of phenomena, from neural processes to psychiatric observations.
Extended Data
Supplementary Materials
Table 6:: Testing the alignment of the decision-space model to a selection of the experimental literature on LHb and RMTg.
| Criterion | Matsumoto & Hikosaka (2007)80 (I) | Lee & Hikosaka (2022)81 (II) | Stopper & Floresco (2014)82 (III) | Vento et al. (2017)83 (IV) |
|---|---|---|---|---|
| 7.A | ∅ | ∅ | ≈ | ✓ |
| 7.B | ✓ | ✓ | ∅ | ∅ |
✓ — aligned with criterion
≈ — somewhat aligned to criterion
✘ — not aligned with criterion
∅ — experiment does not test criterion
7.A.III. LHb inactivation led subjects to change their choice during a probabilistic discounting task to accept a large, risky reward over a smaller, safe reward. As our model expects, LHb inactivation played an important role in affecting decisions that required multiple decision-dimensions. It is expected, however, that reduced LHb activity produces enhanced adherence to any decision-dimensions required to perform the task. The subjects with inactive LHb would appear to be incorporating fewer, not more, decision-dimensions into their choices. One possible explanation is that LHb inactivation led to an overwhelming increase in dimensionality of the decision-space that reduced focus on a few important decision-dimensions, such as reward.
7.A.IV. RMTg selectively altered decisions, primarily in response to cost. This may suggest that RMTg inactivation led to choices with less adherence to an important cost-related decision-dimension.
7.B.I. LHb activation led to suppression of dopaminergic signaling among daSNC neurons. LHb was active at times when it may not have been beneficial to construct a decision-space involving a reward-related decision-dimension (when no reward was presented) but inactive when it may have been beneficial to construct a decision-space using a reward-related decision-dimension (when reward was presented).
7.B.II. LHb was found to alter its activity depending on situational context. LHb was most active at times when it may not have been beneficial to construct a decision-space involving a reward-predominant decision-dimension (when it was indicated that minimal reward would be available or when less than expected reward was presented) and at times when evaluation of data along decision-dimensions may have been less necessary (the uncontrollable tasks).
Table 7:: Testing the alignment of the decision-space model to a selection of the experimental literature on daSNC.
| Criterion | Fiorillo et al. (2003)84 (I) | Matsumoto & Hikosaka (2009)85 (II) | Gan et al. (2010)86 (III) | Bromberg-Martin et al. (2010)87 (IV) | Kim et al. (2020)88 (V) | Long et al. (2024)89 (VI) |
|---|---|---|---|---|---|---|
| 8.A | ✓ | ✓ | ✓ | ✓ | ✓ | ∅ |
| 8.B | ∅ | ∅ | ∅ | ∅ | ∅ | ≈ |
✓ — aligned with criterion
≈ — somewhat aligned to criterion
✘ — not aligned with criterion
∅ — experiment does not test criterion
8.A.I. daSNC neurons responded differently during the cue and during the outcome period depending on the likelihood of a cue predicting a reward outcome. This may suggest that a subpopulation of daSNC neurons encodes information along a reward-related decision-dimension, and probabilistic inputs are reflected continuously over time as the information along the decision-dimension is updated.
8.A.II. Two daSNC populations responded very differently to rewarding or aversive stimuli and a third group was non-responsive. This may support the tenet of the decision-space model that different daSNC subpopulations correspond to different decision-dimensions, some of which might be related to reward information, some to cost information, and some to neither reward nor cost information.
8.A.III. The activities of recorded dopamine neurons showed more resemblance to reward levels than to overall utility. This may support the tenet of the decision-space model that different dopamine subpopulations correspond to different decision-dimensions, some of which are related to reward information, and that dopamine neurons encode data along decision-dimensions, not an overall value function.
8.A.IV. Subpopulations of daSNC neurons that encoded value were excited by rewarding information while salience neurons were excited by both rewarding and aversive cues. This may support the tenet of the decision-space model that different daSNC subpopulations correspond to different decision-dimensions, some of which are related to reward information and some to other information.
8.A.V. Dopamine changed in response to altered proximity to reward. This may support the architecture of the decision-space model, where changes to reward information are captured in an sSPN subpopulation related to reward, then passed to a corresponding daSNC subpopulation.
8.B.VI. VTA cells were optogenetically inhibited or excited as ventral striatal neurons were recorded. Several findings are particularly relevant to the decision-space model: 1) A subpopulation of the striatal neurons responded to reward, and the activities of these neurons correlated with the VTA neurons. 2) 8% of all SPNs (4% above control, both non-reward-responding and reward-responding) had altered activities when VTA was inhibited. 3) The physical location of the SPNs that had altered activities had significantly distinct locations. Finding 1 may support the decision-space model, where mSPNs and sSPNs receive somewhat similar cortical inputs and sSPNs influence daSNC activity. A proportion of the reward-responding SPNs may encode a reward-related decision-dimension (the others may encode other information about the task and the reward administration). Further, per Finding 2, only a proportion of SPNs were affected by the reward-induced daSNC activity, supporting the selective release of dopamine to a reward-related SPN subpopulation in the model. Per Finding 3, the SPNs that did change their activities had spatial organization, supporting the assumption of our model that SPN subpopulations corresponding to decision-dimensions are organized spatially. The experiments, however, were primarily conducted on VTA neurons, not daSNC neurons, and a decoder did not accurately discriminate inhibition from control trials based on SPN spiking. This might be because a reward-related decision-space would require the modulation of more than 8% of neurons. Per our model, a decision-space would be formed in cases where sufficient dopamine was released to modulate a larger percentage (but not all SPNs). Indeed, when VTA neurons were manipulated to release more dopamine than they ordinarily did during the head-fixed licking task, up to 37% SPNs responded and a decoder successfully linked firing rates to the task, suggesting the formation of a decision-space. It may be that a decision-space only forms in certain tasks, for instance perhaps in tasks that require action selection, a hypothesis that might be supported by the findings of Samejima et al.90 and Seo et al.91 Our model provides a framework for task-dependent dopamine release to be studied in future work.
References
- R. L. Aupperle, M. P. Paulus. Neural systems underlying approach and avoidance in anxiety disorders.. Dialogues in clinical neuroscience, 2010. [PubMed]
- J. R. Crittenden, A. M. Graybiel. Basal Ganglia disorders associated with imbalances in the striatal striosome and matrix compartments.. Front Neuroanat, 2011. [PubMed]
- A. Friedman. Chronic Stress Alters Striosome-Circuit Dynamics, Leading to Aberrant Decision-Making.. Cell, 2017. [PubMed]
- A. M. Graybiel, A. Matsushima. Striosomes and Matrisomes: Scaffolds for Dynamic Coupling of Volition and Action.. Annu. Rev. Neurosci., 2023. [PubMed]
- A. B. Nelson, A. C. Kreitzer. Reassessing models of basal ganglia function and dysfunction.. Annu Rev Neurosci, 2014. [PubMed]
- J. Cox, I. B. Witten. Striatal circuits for reward learning and decision-making.. Nat Rev Neurosci, 2019. [PubMed]
- A. M. Graybiel, C. W. Ragsdale. Histochemically distinct compartments in the striatum of human, monkeys, and cat demonstrated by acetylthiocholinesterase staining.. Proc Natl Acad Sci U S A, 1978. [PubMed]
- K. R. Brimblecombe, S. J. Cragg. The Striosome and Matrix Compartments of the Striatum: A Path through the Labyrinth from Neurochemistry toward Function.. ACS Chem Neurosci, 2017. [PubMed]
- C. R. Gerfen. Synaptic organization of the striatum.. J. Elec. Microsc. Tech., 1988
- A. Friedman. Striosomes Mediate Value-Based Learning Vulnerable in Age and Huntington’s Model.. Cell, 2020. [PubMed]
- A. Friedman. A Corticostriatal Path Targeting Striosomes Controls Decision-Making under Conflict.. Cell, 2015. [PubMed]
- B. Bloem. Multiplexed action-outcome representation by striatal striosome-matrix compartments detected with a mouse cost-benefit foraging task.. Nat Commun, 2022. [PubMed]
- M. Weglage. Complete representation of action space and value in all dorsal striatal pathways.. Cell Rep, 2021. [PubMed]
- X. Xiao. A Genetically Defined Compartmentalized Striatal Direct Pathway for Negative Reinforcement.. Cell, 2020. [PubMed]
- T. Yoshizawa, M. Ito, K. Doya. Reward-predictive neural activities in striatal striosome compartments.. eNeuro, 2018
- I. Lazaridis. Striosomes control dopamine via dual pathways paralleling canonical basal ganglia circuits.. Current Biology, 2024. [DOI]
- T. A. Jenrette, J. B. Logue, K. A. Horner. Lesions of the Patch Compartment of Dorsolateral Striatum Disrupt Stimulus-Response Learning.. Neuroscience, 2019. [PubMed]
- J. A. Nadel. Optogenetic stimulation of striatal patches modifies habit formation and inhibits dopamine release.. Sci Rep, 2021. [PubMed]
- J. A. Nadel, S. S. Pawelko, D. Copes-Finke, M. Neidhart, C. D. Howard. Lesion of striatal patches disrupts habitual behaviors and increases behavioral variability.. PLoS One, 2020. [PubMed]
- E. M. Prager. Dopamine Oppositely Modulates State Transitions in Striosome and Matrix Direct Pathway Striatal Spiny Neurons.. Neuron, 2020. [PubMed]
- R. C. Evans. Functional Dissection of Basal Ganglia Inhibitory Inputs onto Substantia Nigra Dopaminergic Neurons.. Cell Reports, 2020. [PubMed]
- S. Hong. Predominant Striatal Input to the Lateral Habenula in Macaques Comes from Striosomes.. Curr Biol, 2019. [PubMed]
- N. Rajakumar, K. Elisevich, B. A. Flumerfelt. Compartmental origin of the striato-entopeduncular projection in the rat.. J Comp Neurol, 1993. [PubMed]
- F. Fujiyama. Exclusive and common targets of neostriatofugal projections of rat striosome neurons: a single neuron-tracing study using a viral vector.. Eur J Neurosci, 2011. [PubMed]
- H. Schroll, F. H. Hamker. Computational models of basal-ganglia pathway functions: focus on functional neuroanatomy.. Front. Syst. Neurosci., 2013
- J. R. Crittenden. Striosome-dendron bouquets highlight a unique striatonigral circuit targeting dopamine-containing neurons.. Proceedings of the National Academy of Sciences of the United States of America, 2016. [PubMed]
- G. Barbera. Spatially Compact Neural Clusters in the Dorsal Striatum Encode Locomotion Relevant Information.. Neuron, 2016. [PubMed]
- M. Azcorra. Unique functional responses differentially map onto genetic subtypes of dopamine neurons.. Nat Neurosci, 2023. [PubMed]
- M. L. Wallace. Genetically Distinct Parallel Pathways in the Entopeduncular Nucleus for Limbic and Sensorimotor Output of the Basal Ganglia.. Neuron, 2017. [PubMed]
- G. R. Christoph, R. J. Leonzio, K. S. Wilcox. Stimulation of the lateral habenula inhibits dopamine-containing neurons in the substantia nigra and ventral tegmental area of the rat.. The Journal of neuroscience : the official journal of the Society for Neuroscience, 1986. [PubMed]
- S. Hong, T. C. Jhou, M. Smith, K. S. Saleem, O. Hikosaka. Negative Reward Signals from the Lateral Habenula to Dopamine Neurons Are Mediated by Rostromedial Tegmental Nucleus in Primates.. Journal of Neuroscience, 2011. [PubMed]
- A. K. Lahiri, M. D. Bevan. Dopaminergic Transmission Rapidly and Persistently Enhances Excitability of D1 Receptor-Expressing Striatal Projection Neurons.. Neuron, 2020. [PubMed]
- S. Grillner, B. Robertson. The basal ganglia downstream control of brainstem motor centres—an evolutionarily conserved strategy.. Current Opinion in Neurobiology, 2015. [PubMed]
- B. W. Balleine, M. R. Delgado, O. Hikosaka. The Role of the Dorsal Striatum in Reward and Decision-Making.
- H. J. Lee. Activation of Direct and Indirect Pathway Medium Spiny Neurons Drives Distinct Brain-wide Responses.. Neuron, 2016. [PubMed]
- T. D. Sanger. Optimal unsupervised learning in a single-layer linear feedforward neural network.. Neural Networks, 1989
- J. Kamiński. Novelty-Sensitive Dopaminergic Neurons in the Human Substantia Nigra Predict Success of Declarative Memory Formation.. Current Biology, 2018. [PubMed]
- S. Kim, A. K. Bera. Scalar Measures of Volatility and Dependence for the Multivariate Models with Applications to Asian Financial Markets.. JRFM, 2023
- 39.Classical Microeconomics. in On Classical Economics 48–78 (Yale University Press, 2017). doi:10.12987/9780300185669-005.
- G. Winterer, D. R. Weinberger. Genes, dopamine and cortical signal-to-noise ratio in schizophrenia.. Trends in Neurosciences, 2004. [PubMed]
- Y. Miyamoto, S. Katayama, N. Shigematsu, A. Nishi, T. Fukuda. Striosome-based map of the mouse striatum that is conformable to both cortical afferent topography and uneven distributions of dopamine D1 and D2 receptor-expressing cells.. Brain structure & function, 2018. [PubMed]
- O. Hikosaka. Direct and indirect pathways for choosing objects and actions.. Eur J Neurosci, 2019. [PubMed]
- M. Maltese, J. R. March, A. G. Bashaw, N. X. Tritsch. Dopamine differentially modulates the size of projection neuron ensembles in the intact and dopamine-depleted striatum.. Elife, 2021. [PubMed]
- P. Calabresi, B. Picconi, A. Tozzi, V. Ghiglieri, M. Di Filippo. Direct and indirect pathways of basal ganglia: a critical reappraisal.. Nat Neurosci, 2014. [PubMed]
- A. G. Salinas, M. I. Davis, D. M. Lovinger, Y. Mateo. Dopamine dynamics and cocaine sensitivity differ between striosome and matrix compartments of the striatum.. Neuropharmacology, 2016. [PubMed]
- Z. Luo, N. D. Volkow, N. Heintz, Y. Pan, C. Du. Acute Cocaine Induces Fast Activation of D1 Receptor and Progressive Deactivation of D2 Receptor Striatal Neurons: In Vivo Optical Microprobe [Ca2+]i. Imaging. J. Neurosci., 2011. [PubMed]
- K. Oyama, I. Hernádi, T. lijima, K.-I. Tsutsui. Reward Prediction Error Coding in Dorsal Striatal Neurons.. J. Neurosci., 2010. [PubMed]
- P. Cavedini. Decision-making heterogeneity in obsessive-compulsive disorder: ventromedial prefrontal cortex function predicts different treatment outcomes.. Neuropsychologia, 2002. [PubMed]
- S. E. Cohen, J. B. Zantvoord, B. N. Wezenberg, C. L. H. Bockting, G. A. Van Wingen. Magnetic resonance imaging for individual prediction of treatment response in major depressive disorder: a systematic review and meta-analysis.. Transl Psychiatry, 2021. [PubMed]
- S. Saperia. Modeling Effort-Based Decision Making: Individual Differences in Schizophrenia and Major Depressive Disorder. Biological Psychiatry:. Cognitive Neuroscience and Neuroimaging, 2023
- A. G. C. Wright, L. J. Simms. Stability and fluctuation of personality disorder features in daily life.. Journal of Abnormal Psychology, 2016. [PubMed]
- A. S. S. de Siqueira. Decision Making assessed by the lowa Gambling Task and Major Depressive Disorder A systematic review.. Dement Neuropsychol, 2018. [PubMed]
- J. Cousijn. Individual differences in decision making and reward processing predict changes in cannabis use: a prospective functional magnetic resonance imaging study.. Addict Biol, 2013. [PubMed]
- S. R. Horn, A. Feder. Understanding Resilience and Preventing and Treating PTSD.. Harv Rev Psychiatry, 2018. [PubMed]
- C. L. Pickens, S. A. Golden, T. Adams-Deutsch, S. G. Nair, Y. Shaham. Long-lasting incubation of conditioned fear in rats.. Biol. Psychiatry, 2009. [PubMed]
- I. Fredriksson. Orbitofrontal cortex and dorsal striatum functional connectivity predicts incubation of opioid craving after voluntary abstinence.. Proc. Natl. Acad. Sci. U.S.A., 2021. [PubMed]
- K. Gurney, T. J. Prescott, P. Redgrave. A computational model of action selection in the basal ganglia. II. Analysis and simulation of behaviour.. Biol Cybern, 2001. [PubMed]
- M. Möller, S. Manohar, R. Bogacz. Uncertainty-guided learning with scaled prediction errors in the basal ganglia.. PLoS Comput Biol, 2022. [PubMed]
- K. Morita, A. Kato. Striatal dopamine ramping may indicate flexible reinforcement learning with forgetting in the cortico-basal ganglia circuits.. Front. Neural Circuits, 2014
- W. Dabney. A distributional code for value in dopamine-based reinforcement learning.. Nature, 2020. [PubMed]
- I. Bar-Gad, G. Morris, H. Bergman. Information processing, dimensionality reduction and reinforcement learning in the basal ganglia.. Progress in Neurobiology, 2003. [PubMed]
- G. S. Berns, T. J. Sejnowski. A Computational Model of How the Basal Ganglia Produce Sequences.. Journal of Cognitive Neuroscience, 1998. [PubMed]
- E. Garr. Contributions of the basal ganglia to action sequence learning and performance.. Neuroscience & Biobehavioral Reviews, 2019. [PubMed]
- A. Reiner. Corticostriatal projection neurons – dichotomous types and dichotomous functions.. Front. Neuroanat., 2010
- J. L. Lanciego, N. Luquin, J. A. Obeso. Functional neuroanatomy of the basal ganglia.. Cold Spring Harb Perspect Med, 2012. [PubMed]
- A.-C. Martel, A. Galvan. Connectivity of the corticostriatal and thalamostriatal systems in normal and parkinsonian states: An update.. Neurobiology of Disease, 2022. [PubMed]
- F. Eblen, A. M. Graybiel. Highly restricted origin of prefrontal cortical inputs to striosomes in the macaque monkey.. J Neurosci, 1995. [PubMed]
- M. M. McGregor. Functionally Distinct Connectivity of Developmentally Targeted Striosome Neurons.. Cell Rep, 2019. [PubMed]
- C. R. Gerfen. The neostriatal mosaic: compartmentalization of corticostriatal input and striatonigral output systems.. Nature, 1984. [PubMed]
- J. B. Smith. Genetic-Based Dissection Unveils the Inputs and Outputs of Striatal Patch and Matrix Compartments.. Neuron, 2016. [PubMed]
- S. Hong, O. Hikosaka. Diverse sources of reward value signals in the basal ganglia nuclei transmitted to the lateral habenula in the monkey.. Front Hum Neurosci, 2013. [PubMed]
- S. Hong, O. Hikosaka. The Globus Pallidus Sends Reward-Related Signals to the Lateral Habenula.. Neuron, 2008. [PubMed]
- M. Weglage. Sst+ GPi Output Neurons Provide Direct Feedback to Key Nodes of the Basal Ganglia and Drive Behavioral Flexibility., 2022. [DOI]
- L. Gonçalves, C. Sego, M. Metzger. Differential projections from the lateral habenula to the rostromedial tegmental nucleus and ventral tegmental area in the rat.. J of Comparative Neurology, 2012
- T. C. Jhou, H. L. Fields, M. G. Baxter, C. B. Saper, P. C. Holland. The Rostromedial Tegmental Nucleus (RMTg), a GABAergic Afferent to Midbrain Dopamine Neurons, Encodes Aversive Stimuli and Inhibits Motor Responses.. Neuron, 2009. [PubMed]
- R. J. Smith, P. J. Vento, Y. S. Chao, C. H. Good, T. C. Jhou. Gene expression and neurochemical characterization of the rostromedial tegmental nucleus (RMTg) in rats and mice.. Brain Struct Funct, 2019. [PubMed]
- A. Jaquins-Gerstl, K. M. Nesbitt, A. C. Michael. In vivo evidence for the unique kinetics of evoked dopamine release in the patch and matrix compartments of the striatum.. Anal Bioanal Chem (, 2021. [DOI]
- T. F. Münte. The human globus pallidus internus is sensitive to rewards – Evidence from intracerebral recordings.. Brain Stimulation, 2017. [PubMed]
- M. Stephenson-Jones. A basal ganglia circuit for evaluating action outcomes.. Nature, 2016. [PubMed]
- M. Matsumoto, O. Hikosaka. Lateral habenula as a source of negative reward signals in dopamine neurons.. Nature, 2007. [PubMed]
- H. Lee, O. Hikosaka. Lateral habenula neurons signal step-by-step changes of reward prediction.. iScience, 2022. [PubMed]
- C. M. Stopper, S. B. Floresco. What’s better for me? Fundamental role for lateral habenula in promoting subjective decision biases.. Nat Neurosci, 2014. [PubMed]
- P. J. Vento, N. W. Burnham, C. S. Rowley, T. C. Jhou. Learning From One’s Mistakes: A Dual Role for the Rostromedial Tegmental Nucleus in the Encoding and Expression of Punished Reward Seeking.. Biological Psychiatry, 2017. [PubMed]
- C. D. Fiorillo, P. N. Tobler, W. Schultz. Discrete Coding of Reward Probability and Uncertainty by Dopamine Neurons.. Science, 2003. [PubMed]
- M. Matsumoto, O. Hikosaka. Two types of dopamine neuron distinctly convey positive and negative motivational signals.. Nature, 2009. [PubMed]
- J. O. Gan, M. E. Walton, P. E. M. Phillips. Dissociable cost and benefit encoding of future rewards by mesolimbic dopamine.. Nat Neurosci, 2010. [PubMed]
- E. S. Bromberg-Martin, M. Matsumoto, O. Hikosaka. Dopamine in Motivational Control: Rewarding, Aversive, and Alerting.. Neuron, 2010. [PubMed]
- H. R. Kim. A Unified Framework for Dopamine Signals across Timescales.. Cell, 2020. [PubMed]
- C. Long. Constraints on the subsecond modulation of striatal dynamics by physiological dopamine signaling.. Nat Neurosci, 2024. [PubMed]
- K. Samejima, Y. Ueda, K. Doya, M. Kimura. Representation of action-specific reward values in the striatum.. Science, 2005. [PubMed]
- M. Seo, E. Lee, B. B. Averbeck. Action Selection and Action Value in Frontal-Striatal Circuits.. Neuron, 2012. [PubMed]
- J. G. Parker. Diametric neural ensemble dynamics in parkinsonian and dyskinetic states.. Nature, 2018. [PubMed]
- J. Peak, B. Chieng, G. Hart, B. W. Balleine. Striatal direct and indirect pathway neurons differentially control the encoding and updating of goal-directed learning.. eLife, 2020. [PubMed]
- F. Sun. Next-generation GRAB sensors for monitoring dopaminergic activity in vivo.. Nat Methods, 2020. [PubMed]
